CADTH Reimbursement Review
Somatrogon (Ngenla)
Sponsor: Pfizer Canada ULC
Therapeutic area: Growth hormone deficiency
This multi-part report includes:
Clinical Review
Pharmacoeconomic Review
Clinical Review
Abbreviations
ANCOVA
analysis of covariance
CI
confidence interval
DCOA
Dyad Clinical Outcome Assessment
DCOA 1
Dyad Clinical Outcome Assessment 1
DCOA 2
Dyad Clinical Outcome Assessment 2
GH
growth hormone
GHD
growth hormone deficiency
hGH
human growth hormone
HRQoL
health-related quality of life
ICC
intraclass correlation coefficient
IGF-I
insulin-like growth factor I
IRT
interactive response technology
IWRS
Interactive Web Response system
LIG-GHD
Life Interference Questionnaire for Growth Hormone Deficiency
LIQ-GHD-Pediatric
Life Interference Questionnaire for Growth Hormone Deficiency–Pediatric
LSM
least squares mean
LT-OLE
long-term open-label extension
MID
minimal important difference
MNAR
missing not at random
OAT
Observer Assessment Tool
OLE
open-label extension
PAT
Participant Assessment Tool
PGI-S: IDA
Patient Global Impression–Severity: Impact on Daily Activities
PP
per-protocol
PRO
patient-reported outcome
QoLISSY
Quality of Life in Short Stature Youth
rhGH
recombinant human growth hormone
SC
subcutaneous
SD
standard deviation
SDS
standard deviation score
SGA
small for gestational age
Executive Summary
An overview of the submission details for the drug under review is provided in Table 1.
Item | Description |
|---|---|
Drug product | Somatrogon (Ngenla), 24 mg/1.2 mL (20 mg/mL) as a pre-filled pen or 60 mg/1.2 mL (50 mg/mL) as a pre-filled pen for SC injection |
Indication | The long-term treatment of pediatric patients who have growth failure due to an inadequate secretion of endogenous growth hormone (growth hormone deficiency) |
Reimbursement request | Long-term treatment of pediatric patients who have growth hormone deficiency |
Health Canada approval status | NOC |
Health Canada review pathway | Standard |
NOC date | October 26, 2021 |
Sponsor | Pfizer Canada ULC |
NOC = Notice of Compliance; SC = subcutaneous.
Introduction
Growth hormone (GH) has been available for management of the short stature associated with growth hormone deficiency (GHD) for more than 60 years.1 Recombinant human growth hormone (rhGH) is the primary treatment for GHD, as well as for the associated abnormalities in body composition, metabolic profile, exercise capacity, and quality of life. Challenges to effective treatment include difficulty in establishing a firm diagnosis of GHD and variable responsiveness to GH within the population diagnosed with GHD.2
GHD is a clinical diagnosis based on auxologic features (i.e., a comparison of a child’s growth pattern to established norms) and confirmed by biochemical testing. If GHD is congenital and complete, the diagnosis is relatively easy to confirm. Affected children present with early growth failure, delayed bone age, the central distribution of body fat, and generally low serum concentrations of GH, insulin-like growth factor I (IGF-I), and insulin-like growth factor-binding protein 3, the major binding protein of circulating IGF-I. IGF-I is a protein that is typically produced by the liver in response to GH stimulation. Both GH and IGF-I form part of the somatotropic axis, which is markedly active at the onset of puberty, and responsible for whole body growth and development.3,4 Infants with GHD are prone to hypoglycemia, prolonged jaundice, micropenis (in males), and giant cell hepatitis. For children with milder manifestations of GHD, it may be more difficult to establish the diagnosis. Nonetheless, once the diagnosis is confirmed, such children can and should be treated with GH until linear growth ceases.5
The prevalence of GHD is estimated to be between 1 in 3,500 children and 1 in 4,000 children in the UK.6 According to a research paper published in 2012, approximately 20,000 children in the US have been diagnosed with GHD.7 Currently, there are no published data on the prevalence and incidence of GHD in Canada.
The drug under review is somatrogon, available as a single patient use, multi-dose, disposable pre-filled pen for subcutaneous (SC) injection, in 2 dose strengths: 24 mg in 1.2 mL sterile solution (20 mg/mL) or 60 mg in 1.2 mL sterile solution (50 mg/mL).8 The indication for somatrogon is long-term treatment of pediatric patients who have growth failure due to an inadequate secretion of endogenous GH (GHD).9 The sponsor reimbursement request is long-term treatment of pediatric patients who have GHD.8
The objective of this report is to perform a systematic review of the beneficial and harmful effects of somatrogon for the long-term treatment of pediatric patients who have growth failure due to an inadequate secretion of endogenous GH (GHD).
Stakeholder Perspectives
The information in this section is a summary of input provided by the clinical expert consulted by CADTH for the purpose of this review.
Clinician Input
Input From Clinical Expert Consulted by CADTH
The most important goals that the ideal treatment would address would be optimizing final adult height, restoring metabolic functions associated with GHD, and optimizing quality of life. Some children also have additional co-existing pituitary hormone deficiencies such as thyroid hormone deficiency, cortisol deficiency, and gonadotropin deficiency. These hormone deficiencies should also be appropriately replaced to optimize growth. GH is an important counter-regulatory hormone in the regulation of blood glucose. GH also helps improve muscular tone and has anabolic effects on bone. While not the primary indications for replacing GH, these additional benefits are appreciated by patients and prescribing physicians.
Adherence is a major limitation to experiencing the full benefits of rhGH therapy. Currently, rhGH formulations are given as SC injections on a daily or near-daily basis (6 days per week). These injections must be given throughout childhood and adolescence. This daily schedule can be inconvenient when patients want to leave their home for any reason (e.g., travelling, visiting, camping) because they have to think about how to transport and store the drug, remember to bring the accompanying supplies (e.g., needles, pen tips, alcohol swabs), and disrupt the activities that they are doing. Furthermore, some patients find the injections painful or anxiety-provoking. These nightly injections cause stress on the families from having to chase after their children and find them and then hold them down for their injections. An ideal rhGH treatment would provide benefits on growth and metabolic outcomes while minimizing pain and anxiety. At the moment, the unmet needs with current rhGH formulations pertain to suboptimal adherence due to the anxiety and pain of injections, the frequency of injections, and the inconvenience in storing and handling injections, or simply forgetting to administer the injection.
Somatrogon could be used as first-line treatment for pediatric GHD. Currently, there is no evidence available for somatrogon in patients who are younger than 3 years of age, so if GHD was diagnosed in infancy or early childhood, then the child would start with the daily rhGH formulations and could be switched to the once-weekly formulation after the age of 3 years.
The clinical expert consulted by CADTH indicated that patients who have been using rhGH daily are likely to derive similar benefit from somatrogon in terms of impact on growth. In addition, a patient’s quality of life may significantly improve with a switch from daily injections to once-weekly injections.
A positive change in height velocity that results in an increase in height standard deviation score (SDS) indicates a favourable response to treatment. An inadequate response after the initiation of rhGH therapy in patients with GHD is often defined by 1 or more of the following criteria: a change in height velocity of less than 2 cm per year, a height velocity SDS of less than 0, or a change in height SDS of less than 0.3 per year during the first 6 months to 12 months of therapy. With height being the major outcome of interest, treatment response should be monitored every 3 months to 4 months in younger children who are expected to grow more rapidly and then every 6 months in the elementary school-aged child who grows less rapidly, and then every 4 months to 6 months in the pubertal-aged child.
RhGH is generally very safe and well tolerated. Prescribers and nurses discuss potential side effects and adverse effects with their patients and their caregivers. In the rare instance where a patient might develop a slipped capital femoral epiphysis or pseudotumour cerebri, GH therapy is paused to allow for treatment or resolution of the adverse event. In cases of glucose intolerance or significantly high IGF-I levels, the dose of GH may need to be reduced. Reasons to stop treatment before growth is complete would be if the patient and caregiver do not adhere to treatment advice, for example, by neglecting to attend appointments, adjusting doses on their own, or refusing to follow through on recommended laboratory monitoring. Generally, in these cases, the prescriber would first try to determine barriers to care before discontinuing therapy altogether.
Somatrogon should be prescribed only by pediatric endocrinologists who have access to the resources needed to be able to diagnose GHD properly and to endocrine nurses who are knowledgeable in GHD and would be able to support patients who require treatment.
Drug Program Input
Input was obtained from the drug programs that participate in the CADTH reimbursement review processes. The following were identified as key factors that could impact the implementation of a CADTH recommendation for somatrogon.
When should somatrogon be stopped? The clinical expert consulted by CADTH was of the opinion that when growth plates are fused, rhGH therapy becomes contraindicated. Other considerations could be bone age older than 14 years in girls or bone age older than 16 years in boys or height velocity of less than 2 cm per year.
Would somatrogon be prescribed off label? The clinical expert consulted by CADTH noted that patients with other non-GHD indications may request somatrogon to enable less frequent injections. It is unlikely that somatrogon would be covered by private insurance if it were prescribed off label. They felt that of all the indications, adult GHD may be the one most considered for off-label prescription of somatrogon.
Clinical Evidence
Pivotal Studies and Protocol Selected Studies
Description of Studies
Two phase III studies (CP-4-006 and CP-4-009) were included in the systematic review.
CP-4-006 (N = 224) was an open-label, multi-centre, randomized, active-controlled, parallel group study evaluating the efficacy and safety of weekly somatrogon to daily Genotropin. Patients enrolled in the CP-4-006 study were prepubertal children with GHD and ranged in age from 3 years to younger than 11 years for girls and younger than 12 years for boys. CP-4-009 was a 12-month, open-label, multi-centre, randomized, active-controlled, parallel group study conducted in Japan that compared the efficacy and safety of weekly somatrogon to daily Genotropin in Japanese prepubertal children with GHD who ranged in age from 3 years to younger than 11 years for girls and younger than 12 years for boys. In both studies, patients were randomized in a 1:1 method to receive weekly SC doses of somatrogon or daily SC of Genotropin for 12 months. In both studies, following the completion of the 12-month treatment period, eligible patients were enrolled in a single-arm, open-label extension (OLE) treatment phase with somatrogon. The OLE treatment phase of study CP-4-009 (CP-4-009-Japan-OLE) is summarized in the Other Relevant Evidence section of this report. The OLE treatment phase of study CP-4-006 was not available at the time of writing this report.
The primary efficacy outcome of the CP-4-006 and CP-4-009 trials was annualized height velocity after 12 months of treatment. The secondary efficacy outcomes of both studies were annualized height velocity at 6 months, a change in height SDS at 6 months and 12 months, and a change in bone maturation at 12 months.
Efficacy Results
In study CP-4-006, the primary objective was to demonstrate that annual (12 months) height velocity from weekly somatrogon administration is noninferior to daily Genotropin administration in children with GHD. The primary objective in study CP-4-009 was to demonstrate that annual (12 months) height velocity from weekly somatrogon administration is comparable to daily Genotropin administration in children with GHD. Noninferiority in study CP-4-006 was concluded if the lower bound of the 2-sided 95% CI for the mean treatment difference between somatrogon and Genotropin in annualized height velocity after 12 months of treatment was –1.8 cm per year or more. Comparability in study CP-4-009 was concluded if the mean treatment difference of somatrogon minus Genotropin in annualized height velocity after 12 months of treatment was –1.8 cm per year or more. In study CP-4-006, the least squares mean (LSM) treatment difference for the mean height velocity after 12 months of treatment was 0.33 cm per year (95% confidence interval [CI], –0.24 to 0.89). The lower bound of the 2-sided 95% CI for mean height velocity was greater than the pre-specified noninferiority margin of –1.8 cm per year, indicating that weekly somatrogon administration was noninferior to daily Genotropin administration. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. In study CP-4-009, the treatment difference (somatrogon minus Genotropin) in LSM height velocity (cm per year) was 1.79 cm per year with a 2-sided 95% CI (0.97 to 2.61). Since the point estimate of 1.79 cm per year was greater than the pre-established margin of –1.8 cm per year, weekly somatrogon was concluded to be comparable to daily Genotropin.
The LSM treatment difference for annualized height velocity at 6 months was |||| cm per year (95% CI, |||||||||||||) in CP-4-006 and |||| cm per year (95% CI, |||||||||||||) in study CP-4-009. The LSM treatment difference for change in height SDS from baseline to 6 months was |||| (95% CI, |||||||||||||) in CP-4-006 and |||| (95% CI, |||||||||||||) in study CP-4-009. The LSM treatment difference for change in height SDS from baseline to 12 months was 0.05 (95% CI, –0.06 to 0.16) in CP-4-006 and |||| (95% CI, |||||||||||||) in study CP-4-009. The statistical significance of these results cannot be interpreted due to the lack of reporting of P values.
In both studies, advancement in bone age ||||||||||| exceed advancement in chronological age; mean bone maturation (defined as the ratio of bone age to chronologic age) at 12 months was ||||||||||||| in both treatment groups, |||||||||||||||||||||||||||||||||||||| was reported.
Health-related quality of life (HRQoL) was assessed in CP-4-006 using the Quality of Life in Short Stature Youth (QoLISSY) questionnaire, which assesses the impact of short stature on children from the perspectives of both the patients and their parents. In addition, the number of successful injection attempts by patients using the somatrogon multi-dose, pre-filled pen was evaluated based on the Observer Assessment Tool (OAT) and Participant Assessment Tool (PAT). The OAT was used to record the observer’s assessment of an administration with the device after the patient or caregiver injected somatrogon. The PAT was used to record all of the patients’ or caregivers’ injections of somatrogon and was completed by the actual user of the pen or parent/legal guardian. However, the analyses for QoLISSY were performed in selected locations only and the analyses for other patient-reported outcome (PRO) responses were reported for the somatrogon treatment group only; analyses were conducted in observed case patients, there were substantial amounts of missing data that would introduce significant biases, and no minimal important difference (MID) was identified in the literature for QoLISSY, OAT, and PAT. Hence, the effect of these assessments in support of somatrogon are highly uncertain.
Harms Results
In CP-4-006, 87.2% and 84.3% of the patients in the somatrogon group and Genotropin group, respectively, reported at least 1 AE; in CP-4-009, |||% and ||||% of the patients in the somatrogon group and Genotropin group, respectively, reported at least 1 AE. In both studies, the most commonly occurring adverse events were injection site pain, nasopharyngitis, pyrexia, and headache. No deaths were reported in either of the studies. In CP-4-006, the most notable harm reported was an injection-related event that was reported in ||||% and ||||% of patients in the somatrogon treatment group and Genotropin treatment group, respectively. In CP-4-009, the most common notable harm reported was an injection-related event that was reported in 72.7% of patients in the somatrogon treatment group. The clinical expert did express concern that the injection site reaction in somatrogon was higher compared to Genotropin. As somatrogon is a once-weekly injection and Genotropin is a once-daily injection, these analyses may warrant further explanation. The long-term safety concerns from the CP-4-006 study remain unknown.
Table 2: Summary of Key Results From Pivotal and Protocol Selected Studies
Outcome measures | CP-4-006 | CP-4-009 | ||
|---|---|---|---|---|
Somatrogon N = 109 | Genotropin N = 115 | Somatrogon N = 22 | Genotropin N = 22 | |
Annual HV at 12 months (full analysis set) | ||||
n (%) | 108 (99.0) | 113 (98.2) | 22 (100) | 21 (95.45) |
LSM estimate | 10.10 | 9.78 | 9.654 | 7.868 |
Treatment mean difference (95% CI) | 0.33 (–0.24 to 0.89)a | 1.786 (0.966 to 2.605)b | ||
P value | NR | NR | ||
Annual HV at 6 months (full analysis set) | ||||
n (%) | ||||||||||||| | ||||||||||||| | 22 (100) | 21 (95.45) |
LSM estimate | 10.59 | 10.04 | |||||| | ||||| |
Treatment mean difference (95% CI)c | ||||||||||||| | ||||||||||||| | ||
P value | ||||||||||||| | ||||||||||||| | ||
Change in height SDS at 6 months (full analysis set) | ||||
n (%) | 108 (99.0) | 114 (99.1) | 22 (100) | 21 (95.45) |
LSM estimate | 0.54 | 0.48 | 0.576 | 0.312 |
Treatment mean difference (95% CI)c | ||||||||||||| | ||||||||||||| | ||
P value | ||||||||||||| | ||||||||||||| | ||
Change in height SDS at 12 months (full analysis set) | ||||
n (%) | 108 (99.0) | 113 (98.2) | 22 (100) | 21 (95.45) |
LSM estimate | 0.92 | 0.87 | 0.939 | 0.523 |
Treatment mean difference (95% CI)c | 0.05 (–0.06 to 0.16) | ||||||||||||| | ||
P value | NR | NR | ||
Harms, n (%) (safety analysis set) | ||||
AEs | 95 (87.2) | 97 (84.3) | 22 (100.0) | ||||||||||||| |
SAEs | 3 (2.8) | 2 (1.7) | 2 (9.1) | 2 (9.1) |
WDAEs (from study treatment) | 1 (0.9) | 0 | 0 | 1 (4.5) |
Deaths | 0 | 0 | 0 | 0 |
Notable harms, n (%) (safety analysis population) | ||||
Injection-related events | 47 (43.1) | 29 (25.2) | 16 (72.7) | 3 (13.6) |
Glucose intolerance/insulin resistance | ||||||||||||| | ||||||||||||| | ||||||||||||| | ||||||||||||| |
Excess IGF-I levels | ||||||||||||| | ||||||||||||| | ||||||||||||| | ||||||||||||| |
Malignancies | ||||||||||||| | ||||||||||||| | ||||||||||||| | ||||||||||||| |
Benign intracranial hypertension | ||||||||||||| | ||||||||||||| | ||||||||||||| | ||||||||||||| |
Slipped capital femoral epiphysis | ||||||||||||| | ||||||||||||| | ||||||||||||| | ||||||||||||| |
Scoliosis | ||||||||||||| | ||||||||||||| | ||||||||||||| | ||||||||||||| |
Tonsillar hypertrophy | ||||||||||||| | ||||||||||||| | ||||||||||||| | ||||||||||||| |
Carpal tunnel | ||||||||||||| | ||||||||||||| | ||||||||||||| | ||||||||||||| |
Peripheral edema | ||||||||||||| | ||||||||||||| | ||||||||||||| | ||||||||||||| |
AE = adverse event; CI = confidence interval; HV = height velocity; IGF-I = insulin-like growth factor I; LSM = least squares mean; NR = not reported; SAE = serious adverse event; SDS = standard deviation score; WDAE = withdrawal due to adverse event.
aNoninferiority was concluded if the lower bound of the 2-sided 95% CI was –1.8 or more.
bComparability was concluded if the point estimate of the LSM treatment difference was –1.8 or more.
cCalculated as somatrogon minus Genotropin.
Source: Clinical Study Report for CP-4-006 (2020)10 and Clinical Study Report for CP-4-009 (2020).11
Critical Appraisal
Internal Validity
The clinical expert noted that the commonly used treatment for GHD in Canada is Humatrope. Both the CP-4-006 and CP-4-009 studies had only Genotropin as the active comparator, despite other approved comparators being available for treatment in Canada. No direct or indirect evidence against GHD used in clinical practice within Canada was identified; hence, the comparative efficacy and safety of somatrogon against other regimens such as somatropin are unknown.
No justification for the comparability criteria was provided in study CP-4-009. While study CP-4-009 met the pre-specified criteria for comparability, this should not be confused with the unequivocal demonstration of equivalence, noninferiority, or superiority.
The clinical expert consulted by CADTH noted that the imbalance in age and sex in study CP-4-009 could influence the efficacy results in favour of somatrogon. However, the difference in age would not impact outcomes observed within the first year, but would impact the final adult height.
The primary outcomes of CP-4-006 and CP-4-009 looked to establish the noninferiority and comparability of somatrogon with Genotropin, respectively. Once noninferiority was established, analyses of the secondary efficacy outcomes were conducted. However, these secondary end points were not part of a hierarchical statistical testing plan and were not controlled for multiplicity, and P values have not been reported. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||, and superiority was not tested in CP-4-009.
CP-4-006 reported HRQoL and other PRO responses. However, these tools were administered only in selected locations with a very small sample size; as the study was open label, this could bias the results of the efficacy outcomes. Moreover, only a complete case analysis was carried out for this data with different subsets of patients at each time point. This would be subjected to an increased risk of bias due to the complete case analysis approach. No MID was identified from literature for the QoLISSY questionnaire or the OAT and PAT instruments.
External Validity
The clinical expert noted that based on baseline demographic and disease characteristics, the study population was fairly generalizable to Canadian patients; however, the clinical expert was of the opinion that the age for inclusion within both studies (i.e., ≥ 3 years) was not reflective of Canadian practice since patients with GHD would be identified in infancy. It was also noted that in CP-4-006, 20% of the cohort was Asian, which is a higher level than that seen in Canada. The clinical expert also commented on how indigenous peoples who are treated in Canada are underestimated in both studies. The clinical expert also noted that the proportion of patients with a peak GH level of greater than 7 ng/dL was higher than what is observed in Canadian clinical practice.
GH therapies are long-term therapies, and even though the primary end point of each of the 2 studies was met, in the absence of long-term comparative efficacy and safety results, interpreting the long-term clinical meaningfulness of somatrogon is limited.
Indirect Comparisons
No indirect comparisons were identified or submitted by the sponsor.
Other Relevant Evidence
Description of C0311002 (Study 002)
The C0311002 study (N = 87) was a randomized, open-label, multi-centre, 2-period crossover study that enrolled children with GHD who ranged in age from 3 years to younger than 18 years. Patients were randomized in a 1:1 ratio to 1 of 2 sequences (sequence 1 or sequence 2). Patients randomized to sequence 1 received treatment with once-daily somatropin for 12 weeks, followed by 12 weeks of treatment with once-weekly somatrogon. Patients randomized to sequence 2 received treatment with once-weekly somatrogon for 12 weeks, followed by 12 weeks of treatment with once-daily somatropin.
The primary objective of C0311002 was to evaluate the treatment burden of a weekly injection of somatrogon and a daily injection of somatropin (Genotropin). Secondary objectives included an evaluation of patient and caregiver self-assessments of treatment experience, and an evaluation of the psychometric properties of the Dyad Clinical Outcome Assessment (DCOA) questionnaires.
Efficacy Results
In the C0311002 study, all of the domains of the Dyad Clinical Outcome Assessment 1 (DCOA 1) questionnaire were associated with numerically greater overall scores during treatment with Genotropin than during treatment with somatrogon, with 2 exceptions: the injection signs and symptoms domain (from patients ≥ 8 years) and the assessment of signs domain (from caregivers for children < 8 years). The reported overall score for these 2 domains did not suggest a preference for either treatment based on the reported overall scores. The primary end point of C0311002 demonstrated that the treatment burden of the once-weekly somatrogon injection schedule, as evaluated by the patient life interference questionnaire, was lower than that of the once-daily Genotropin injection schedule. The LSM for the total score of overall life interference was lower for the once-weekly somatrogon injection schedule than for the once-daily Genotropin injection schedule. The mean treatment difference (95% CI; P value) between somatrogon and Genotropin was –15.49 (95% CI, –19.71 to –11.27; P < 0.0001).
The results of the Dyad Clinical Outcome Assessment 2 (DCOA 2) questionnaire showed that the overall proportion of patients who responded to the questionnaire indicating preference for somatrogon was greater than the proportion of patients indicating preference for Genotropin. The proportion of patients who preferred somatrogon and Genotropin in terms of injecting the medicine was ||||% and ||||%, respectively.
Harms Results
Thirty-eight (44.2%) patients in the Genotropin group and 47 (54.0%) patients in the somatrogon group reported at least 1 adverse event during treatment. The most frequently reported adverse event was injection site pain. One patient stopped treatment due to adverse events, which occurred during treatment with somatrogon as a result of injection site pain. With regard to notable harms for this review, injection-related events were reported by ||||% and ||||% of patients during treatment with Genotropin and somatrogon, respectively.
Critical Appraisal
The primary objective of C0311002 evaluated the treatment experience and patient preference for treatment with once-weekly somatrogon compared to once-daily Genotropin using subjective PROs within an open-label study design, which has potential for significant bias in the results. Evidence of reliability was demonstrated; however, there was no evidence of validity or responsiveness, and an MID was not identified from the literature. Additionally, results for the DCOA 1 questionnaire included P values, but the statistical tests were not controlled for multiplicity and, consequently, were at risk of type I error. The other secondary outcomes were reported descriptively. Both of these factors and the lack of an established MID make it difficult to determine the clinical meaningfulness of the results.
C0311002 had concerns of generalizability to the Canadian patient population and the lack of an appropriate assessment period.
Description of CP-4-009-Japan-OLE
The CP-4-009 long-term open-label extension (LT-OLE) (N = 42) evaluated the long-term efficacy and safety of somatrogon in a single-arm trial in Japanese prepubertal children. Patients who were treated with Genotropin and completed 12 months of treatment during the CP-4-009 main study were switched to a somatrogon dosage of 0.66 mg/kg per week and somatrogon-treated patients who completed 12 months of treatment during the main study continued to receive somatrogon with the same mg/kg per week dosage in the OLE treatment phase. The OLE phase would continue until the marketing registration of somatrogon in Japan.
Efficacy Results
The efficacy outcomes reported were annualized height velocity at 24 months and bone maturation at 24 months. These efficacy outcomes were not part of a pre-specified statistical testing plan. The mean (SD) change from baseline of the open-label phase at month 24 for annualized height velocity in the somatrogon group (N = |||||||||) was ||||||||||||||| and in the Genotropin group (N = |||||||||) was |||||||||||||||. The mean (standard deviation [SD]) change from baseline at month 24 for height SDS in the somatrogon group (N = |||||||||) was |||||| |||||||| and in the Genotropin group (N = |||||||||) was |||||||||||||||. The mean (SD) change from baseline of the open-label phase at month 24 for bone maturation in the somatrogon group (N = |||||||||) was ||||||||||||||| and in the Genotropin group (N = |||||||||) was |||||||||||||||.
Harms Results
The most commonly reported adverse event was |||||||||% in the somatrogon group and ||||% in the Genotropin-followed-by-somatrogon treatment group. |||||||| patient reported ||||||||| as a serious treatment-related adverse event in the Genotropin-followed-by-somatrogon treatment group. There were no reports of any deaths or patients who stopped treatment due to adverse events.
Critical Appraisal
CP-4-009-OLE was conducted to evaluate the long-term efficacy and safety of once-weekly somatrogon. The efficacy results of the OLE phase were selectively reported. The analyses were not part of a statistical testing plan and, hence, the effect of somatrogon at the data cut-off date of March 13, 2020, is considered uncertain. No efficacy analyses for HRQoL or other PRO measures have been conducted; hence, the long-term effect of somatrogon on HRQoL is unknown. CP-4-009-OLE was conducted exclusively in Japanese prepubertal children and did not include any Canadian patients; this is not reflective of Canadian clinical practice. Hence, CP-4-009-OLE has noted generalizability issues. The CP-4-009-OLE study excluded patients younger than 3 years of age, so this also leads to a Canadian generalizability issue as the clinical expert consulted by CADTH stated that in clinical practice, patients with GHD are seen as early as in their infancy.
Conclusions
Two phase III randomized controlled trials were included in the CADTH systematic review of somatrogon for long-term treatment of pediatric patients who have growth failure due to an inadequate secretion of endogenous GH. Both studies demonstrated that for the primary efficacy outcome of annualized height velocity at 12 months, treatment with somatrogon was noninferior (CP-4-006) or comparable (CP-4-009) to Genotropin. |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| Other relevant outcomes such as HRQoL and other PRO responses were not assessed in study CP-4-009 and were not properly assessed in study CP-4-006; as a result, the effect of somatrogon on HRQoL is uncertain. The results of the long-term open-label phase for CP-4-006 were not available; hence, interpretation of the sustained response of treatment to somatrogon is unknown. Key evidence gaps include absence of adherence analyses, limited evidence on switchover from somatrogon to somatropin or vice versa, and limited interpretation of HRQoL and other PRO responses.
The key safety issues with somatrogon were related to injections, with a low number of serious adverse events being reported in the somatrogon and Genotropin treatment groups in both studies. A higher number of patients in the somatrogon treatment group experienced injection site pain, nasopharyngitis, pyrexia, and headache. Conclusions regarding the long-term safety of somatrogon cannot be made in the absence of corresponding data.
Introduction
Disease Background
GH has been available for management of the short stature associated with GHD for more than 60 years.1 Recombinant human growth hormone is the primary treatment for GHD as well as the associated abnormalities in body composition, metabolic profile, exercise capacity, and quality of life. Challenges to effective treatment include difficulty in establishing a firm diagnosis of GHD and variable responsiveness to GH within the population diagnosed with GHD.2 GH therapy is also prescribed for several other specific indications in children and adolescents, including idiopathic short stature and short stature associated with small for gestational age (SGA), chronic kidney disease, Turner syndrome, Prader-Willi syndrome, mutations in the SHOX gene, and Noonan syndrome.
GH affects many of the metabolic processes carried out by somatic cells. Linear growth and skeletal development are tightly regulated processes that are highly dependent on GH signalling and action.12 The best known effect is the effect of increasing body mass. GH increases total body protein content, decreases total body fat content, and increases fat deposition in the liver. Physiologic concentrations of GH also have beneficial effects on the plasma lipid profile (i.e., decreases serum low-density lipoprotein and increases high-density lipoprotein).13,14 The effects on fat are due to stimulation of lipolysis and reciprocal antagonism of the lipogenic action of insulin in peripheral fat stores. GH also increases bone mass by stimulating skeletal IGF-I synthesis and causing the proliferation of pre-chondrocytes, the hypertrophy of osteoblasts, bone remodelling, and net mineralization.15 GH stimulates cartilage growth; this is most evident as a widening of the epiphyseal plate and is associated with an increase in amino acid incorporation into cartilage and bone.16
GHD is a clinical diagnosis based on auxologic features (i.e., a comparison of a child’s growth pattern to established norms) and confirmed by biochemical testing. If GHD is congenital and complete, the diagnosis is relatively easy to confirm. Affected children present with early growth failure, delayed bone age, the central distribution of body fat, and generally low serum concentrations of GH, IGF-I, and insulin-like growth factor-binding protein 3, the major binding protein of circulating IGF-I. IGF-I is a protein that is typically produced by the liver in response to GH stimulation. Both GH and IGF-I form part of the somatotropic axis, which is markedly active at the onset of puberty and responsible for whole body growth and development.3,4 Infants with GHD are prone to hypoglycemia, prolonged jaundice, micropenis (in males), and giant cell hepatitis. For children with milder manifestations of GHD, it may be more difficult to establish the diagnosis. Nonetheless, once the diagnosis is confirmed, such children can and should be treated with GH until linear growth ceases.5
The prevalence of GHD is estimated to be between 1 in 3,500 children and 1 in 4,000 children in the UK.6 According to a research paper published in 2012, approximately 20,000 children in the US have been diagnosed with GHD.7 Currently, there are no published data on the prevalence and incidence of GHD in Canada.
Standards of Therapy
Once the diagnosis GHD is established in children, GH therapy is the recommended course of treatment. Therapy should be started as soon as possible to enhance growth velocity and normalize final adult height.17,18 Patients prescribed GH treatment are injected subcutaneously on a daily basis, 6 to 7 times per week.9,19,20 Various studies indicated that GH is effective in children with GHD and, if started in early childhood, will normalize final height.17
The clinical expert consulted by CADTH indicated that the most important goals that the ideal treatment would address would be optimizing final adult height, restoring metabolic functions associated with GHD, and optimizing quality of life. The clinical expert also noted that GHD is diagnosed based on multiple clinical, auxological, and laboratory and radiographic data. Once the diagnosis of GHD is made, rhGH is prescribed. RhGH for the long-term treatment of pediatric GHD is the only indication for which public health drug plans cover costs. The starting dosage is around 0.18 mg/kg per week, administered once daily as an SC injection, for 6 to 7 days per week. (One day is provided as a break for patients and parents or guardians. For those who experience hypoglycemia from multiple pituitary hormone deficiencies that include GHD, it is common practice to dose daily).
While on treatment, routine monitoring consists of clinic visits. The frequency of visits depends on the age of the child (infants are seen every 3 months while school-aged children are seen every 4 months to 6 months). During the visits, the child’s growth is measured, puberty is assessed, side effects are reviewed, adherence and treatment satisfaction are explored, and dosing adjustments are made. Approximately every 3 months to 6 months, laboratory measurements are taken of IGF-I, thyroid function, any other pituitary hormone deficiencies, and glucose tolerance. A bone age estimation may be done once per year.
Treatment with rhGH for GHD is generally continued until growth is completed (when fused epiphyses are observed) or near complete (growth of less than 2 cm per year or a bone age of > 14 years in girls or > 16 years in boys). Once growth is completed, the dosage is reduced to adult GHD prescribing recommendations until the age of 18 years.
Drug
Somatrogon is a modified long-acting analogue of human growth hormone (hGH). It is a new molecular entity with receptor-binding properties and a mechanism of action analogous to hGH.10 Somatrogon binds to the GH receptor and initiates a signal transduction cascade, culminating in changes in growth and metabolism. Consistent with GH signalling, somatrogon binding leads to the activation of the STAT5B signalling pathway and increases the serum concentration of IGF-I.8
The drug under review is somatrogon, available as a single patient use, multi-dose, disposable pre-filled pen for SC injection, in 2 dose strengths: 24 mg in 1.2 mL sterile solution (20 mg/mL) or 60 mg in 1.2 mL sterile solution (50 mg/mL).8 The Health Canada indication for somatrogon is long-term treatment of pediatric patients who have growth failure due to an inadequate secretion of endogenous GH (GHD).9 The sponsor reimbursement request is long-term treatment of pediatric patients who have GHD.8
Health Canada’s recommended dosage is 0.66 mg/kg body weight administered once weekly by SC injection. If a patient’s growth rate fails to increase in the first year, assess for treatment adherence and other causes of growth failure (e.g., hypothyroidism, undernutrition, advanced bone age); discontinuation of somatrogon treatment should be considered. Treatment with somatrogon should be discontinued when there is evidence of closure of the epiphyseal growth plates.9
Somatrogon is administered using a delivery device (a pre-filled pen). Injection sites are to be rotated successively (left upper arm, right upper arm, left buttock, right buttock, left thigh, right thigh, left abdomen, and right abdomen) such that the same injection site is used only after all other injection sites have been rotated.9 Table 3 presents key characteristics of currently available GH therapies.
Table 3: Key Characteristics of Somatrogon, Genotropin, and Humatrope
Characteristic | Somatrogon | Genotropin | Humatrope |
|---|---|---|---|
Mechanism of action | Binds to the GH receptor initiating signal transduction cascade, resulting in changes in growth and metabolism. Somatrogon binding leads to activation of the STAT5B signalling pathway and increases the serum concentration of IGF-I. | Polypeptide hormone of recombinant DNA origin. The amino acid sequence of the product is identical to that of human GH of pituitary origin. Stimulates linear growth in children with GH deficiency | Stimulates linear growth in pediatric patients who lack adequate normal endogenous GH and in children with short stature in association with Turner syndrome, idiopathic short stature, SHOX deficiency, and failure to catch up in height after small for gestational age birth. Treating pediatric patients with GH deficiency and patients with Turner syndrome with Humatrope produces an increased growth rate and IGF-I concentrations similar to those seen in therapy with human GH of pituitary origin. |
Indicationa | Long-term treatment of pediatric patients who have growth failure due to an inadequate secretion of endogenous GH (GH deficiency) | Long-term treatment of children who have growth failure due to an inadequate secretion of endogenous GH | Long-term treatment of pediatric patients who have growth failure due to an inadequate secretion of normal endogenous GH and whose epiphyses are not closed |
Route of administration | SC | SC | SC |
Recommended dosageb | 0.66 mg/kg per week | 0.16 mg/kg body weight per week to 0.24 mg/kg body weight per week Divided into 6 to 7 doses | 0.18 mg/kg per week (daily equivalent dose of 0.026 mg/kg) Maximum: 0.3 mg/kg per week (daily equivalent dose of 0.043 mg/kg) Divide into equal doses given on 3 alternate days or 6 to 7 times per week or daily |
Serious adverse effects or safety issues | Contraindicated in patients with active malignancy and in patients with closed or fused epiphyses | Contraindicated in patients with PWS who have uncontrolled diabetes, or active psychosis, or active cancer | Not be used for growth promotion in pediatric patients with closed epiphyses |
GH = growth hormone; IGF-I = insulin-like growth factor I; PWS = Prader-Willi syndrome; SC = subcutaneous.
aHealth Canada–approved indication.
bDosage for patients with GH deficiency.
Table 4: Key Characteristics of Nutropin AQ, Omnitrope, Saizen
Characteristic | Nutropin/Nutropin AQ | Omnitrope | Saizen |
|---|---|---|---|
Mechanism of action | Human GH produced by recombinant DNA technology. The amino acid sequence of the somatropin protein is identical to that of pituitary-derived human GH. The treatment of children who lack adequate secretion of endogenous GH results in an increase in growth rate and an increase in IGF-I. | Human GH produced by recombinant DNA technology. The amino acid sequence of the somatropin protein is identical to that of pituitary-derived human GH. The treatment of pediatric patients who have GHD results in linear growth and normalizes concentrations of IGF-I. | Polypeptide hormone consisting of 191 amino acid residues; its structure is identical to that of GH extracted from human pituitary glands. It is produced by recombinant DNA technology in a mammalian cell expression system. It provides an exogenous supply of human GH for those patients lacking the ability to produce adequate endogenous supplies. |
Indicationa | Long-term treatment of children who have growth failure due to GH inadequacy | Long-term treatment of children who have growth failure due to an inadequate secretion of endogenous GH | Long-term treatment of children who have growth failure due to inadequate secretion of normal endogenous GH |
Route of administration | SC | SC | SC |
Recommended dosageb | 0.3 mg/kg per week (~ 0.90 IU/kg per week) Divided daily doses | 0.16 mg/kg body weight per week to 0.24 mg/kg body weight per week Divided into 6 to 7 doses | 0.2 mg/kg body weight per week. Increased to 0.27 mg/kg per week if insufficient response to treatment Weekly |
Serious adverse effects or safety issues | Not be used for growth promotion in pediatric patients with closed epiphyses | Not be used when there is any evidence of neoplastic activity and in pediatric patients with closed epiphyses | Not be used for growth promotion in pediatric patients with closed epiphyses or patients with active neoplasia |
GH = growth hormone; GHD = growth hormone deficiency; IGF-I = insulin-like growth factor I; SC = subcutaneous.
aHealth Canada–approved indication.
bDosage for patients with GHD.
Stakeholder Perspectives
Patient Group Input
This section was prepared by CADTH staff based on the input provided by patient groups.
No patient input was received by CADTH for this submission.
Clinician Input
Input From Clinical Experts Consulted by CADTH
All CADTH review teams include at least 1 clinical specialist with expertise regarding the diagnosis and management of the condition for which the drug is indicated. Clinical experts are a critical part of the review team and are involved in all phases of the review process (e.g., providing guidance on the development of the review protocol, assisting in the critical appraisal of clinical evidence, interpreting the clinical relevance of the results, providing guidance on the potential place in therapy). The following input was provided by 1 clinical specialist with expertise in the diagnosis and management of long-term treatment of pediatric patients who have growth failure due to an inadequate secretion of endogenous GH.
Unmet Needs
The most important goals that ideal treatment would address would be optimizing final adult height, restoring metabolic functions associated with GHD, and optimizing quality of life. Final adult height depends on several factors, some of which are intrinsic to the patient while others are modifiable. Taller parental height, younger age at initiation of treatment, later onset of puberty, and greater magnitude of GHD are associated positively with final adult height. Longer duration of GH treatment, consistent adherence to therapy, and higher doses of GH therapy are also positively associated with final adult height and are modifiable variables. Some children also have additional co-existing pituitary hormone deficiencies such as thyroid hormone deficiency, cortisol deficiency, and gonadotropin deficiency. These hormone deficiencies should also be appropriately replaced to optimize growth.
GH also affects other metabolic processes. GH is an important counter-regulatory hormone in the regulation of blood glucose. When the level of blood glucose falls, GH increases to promote the breakdown of glycogen and the endogenous formation of new glucose. Thus, 1 of the reasons to treat a child with GHD using rhGH is to prevent hypoglycemia. GH also helps improve muscular tone and has anabolic effects on bone. While not the primary indications for replacing GH, these additional benefits are appreciated by patients and prescribing physicians.
Adherence is a major limitation to experiencing the full benefits of rhGH therapy. Currently, rhGH formulations are given as SC injections on a daily or near-daily basis (6 days per week). These injections must be given throughout childhood and adolescence, amounting to approximately 18 years of therapy. This daily schedule can be inconvenient when patients want to leave their home for any reason (e.g., travelling, visiting, camping) because they have to think about how to transport and store the drug, remember to bring the accompanying supplies (e.g., needles, pen tips, alcohol swabs), and disrupt the activities that they are doing. Furthermore, some patients find the injections painful or anxiety-provoking. Clinicians have heard from caregivers of their children running away from them whenever it comes time for their rhGH injections. These nightly injections cause stress on families from having to chase after their children and find them and then hold them down for their injections. An ideal rhGH treatment would provide proven benefits on growth and metabolic outcomes while minimizing pain and anxiety.
At the moment, the unmet needs with current rhGH formulations pertain to suboptimal adherence due to anxiety and pain of injections, frequency of injections, and inconvenience in storing and handling injections, or simply forgetting to administer the injection. Ideally, GH would be given as a pill rather than an injection. Over the years, GH manufacturers have provided options that look less like needles and syringes. They offer pens and pods that attempt to mitigate the anxiety provoked by seeing a syringe. However, the needles in these pens and pods still cause pain, and the possibility of pain continues to be a source of anxiety. Additional work can be put into finding ways to reduce anxiety before injections. At most children’s hospitals, there are child life specialists who can help children with their fear of needles, but the demand currently outnumbers the availability of these specialists. In other conditions where options for longer-lasting formulations are available (e.g., intramuscular Lupron administered once every 3 months compared to monthly intramuscular Lupron for pubertal suppression, weekly intramuscular testosterone compared to daily transdermal testosterone for hormone replacement in hypogonadism), patients often choose the less frequent option to reduce the episodes of anxiety, for convenience, or both. Thus, a longer-acting, less frequently injected formulation of rhGH could address suboptimal adherence from anxiety, pain, and inconvenience. There are also some rhGH formulations that are pre-mixed and ready to load and others that do not require refrigeration.
Place in Therapy
Somatrogon could be used as first-line treatment for pediatric GHD. Currently, there is no evidence available for somatrogon in patients younger than 3 years of age, so if GHD was diagnosed in infancy or early childhood, then the child would start with the daily rhGH formulations and could be switched to the once-weekly formulation after the age of 3. There are some unanswered questions about somatrogon, though — specifically, at what point to measure and then how to use information from IGF-I levels to adjust the dose.
Patient Population
The clinical experts indicated that patients who would be best suited for treatment with somatrogon are pediatric patients who have a confirmed diagnosis of GHD and are at least 3 years of age. Somatrogon can be initiated with pediatric patients with GHD who are treatment naive or have previously received GH replacement therapy. Treatment with rhGH is very long (lasting throughout childhood and adolescence), and if there is an option available that is associated with less pain and less anxiety for children, it would not be ethical to limit access to that option. In addition, most children with GHD have additional pituitary hormone deficiencies: thyroid hormone deficiency (replaced with thyroid hormone in tablet form), cortisol deficiency (replaced by hydrocortisone taken by mouth 3 times per day), diabetes insipidus (treated with by Ddavp [desmopressin acetate injection] taken by mouth 2 times to 3 times per day), and hypogonadism (in adolescence, testosterone is replaced with intramuscular injections every 1 week to 4 weeks while estrogen is replaced either via a daily tablet or a transdermal patch with progesterone replacement by mouth for 10 days to 12 days per month). Children with GHD may also have comorbidities such as cerebral palsy or blindness. Thus, strategies to reduce the burden of care are essential.
A patient’s quality of life may improve with a switch to once-weekly injections. GH therapy requires a long commitment (childhood through adolescence). Adherence decreases over time while treatment fatigue increases. Patients younger than 3 years of age, patients with any of the contraindications or relative contraindications listed in the somatrogon monograph, and patients with an allergy to somatrogon would be least suited for treatment with somatrogon.
Assessing Response to Treatment
There is no single measure of what constitutes a meaningful response to treatment. Consensus guidelines describe seeing improvements in height velocity SDS of 0.3 to 0.5 SD in the first year and catch-up growth that places the child closer to the child’s mid-parental target height. However, any increase in height might be construed by a patient as being positive. A positive change in height velocity that results in an increase in height SDS indicates a favourable response to treatment. An inadequate response after the initiation of rhGH therapy in patients with GHD is often defined by 1 or more of the following criteria: a change in height velocity of less than 2 cm per year, height velocity SDS of less than 0, or a change in height SDS of less than 0.3 per year during the first 6 months to 12 months of therapy. With height being the major outcome of interest, treatment response should be monitored every 3 months to 4 months in younger children who are expected to grow more rapidly and then every 6 months in the elementary school-aged child who grows less rapidly, and then every 4 to 6 months in the pubertal-aged child who again grows more rapidly.
Discontinuing Treatment
Those with GHD should receive GH replacement not only to grow, but also to gain the metabolic benefits of GH. They should not discontinue therapy if possible. The dose of GH may be reduced after the completion of growth to an adult GHD dose. Afterwards, the individual may decide whether to continue with GH replacement as an adult, though it is generally recommended to do so if the adult truly has GHD. In situations where an adverse event happens, GH injections may need to be temporarily stopped until resolution of the adverse outcome. For example, in the case of active malignancy or within a year of treatment, GH therapy should be paused. If the patient develops a slipped capital femoral epiphysis or pseudotumour cerebri, GH therapy is paused to allow for treatment or resolution of the adverse event. In cases of glucose intolerance or significantly high IGF-I levels, the dose of GH may need to be reduced. An additional consideration to stop treatment would be if the patient and caregiver do not adhere to treatment advice, for example, by neglecting to attend appointments, adjusting doses on their own, or refusing to follow through on recommended laboratory monitoring. Generally, in these cases, the prescriber would first try to determine barriers to care before discontinuing therapy altogether.
Prescribing Conditions
Somatrogon should be prescribed only by pediatric endocrinologists who have access to the resources needed to be able to diagnose GHD properly and to endocrine nurses who are knowledgeable in GHD, and would be able to support patients who require treatment. Patients should be monitored in pediatric endocrinology clinics. Those who live far from pediatric endocrinology clinics should have a pediatrician who can collaborate with a pediatric endocrinologist to monitor growth and side effects.
Additional Considerations
Rarely, there are patients who appear to have true GHD even though their stimulated GH peak exceeds traditional cut-offs. The combination of other clinical data (e.g., significant short stature, poor height velocity, delayed bone age, low IGF-I, abnormal pituitary MRI findings) can indicate GHD irrespective of the GH level. These patients may require GH therapy for adequate growth — thus, excessive emphasis should not be placed on only the results of the GH stimulation test when making a diagnosis of GHD or making recommendations for treatment under the indication of GHD.
Clinician Group Input
This section was prepared by CADTH staff based on the input provided by clinician groups.
No clinician group input was identified by CADTH for this submission.
Drug Program Input
The drug programs provide input on each drug being reviewed through CADTH’s reimbursement review processes by identifying issues that may impact their ability to implement a recommendation. The implementation questions and corresponding responses from the clinical experts consulted by CADTH are summarized in Table 5.
Table 5: Summary of Drug Plan Input and Clinical Expert Response
Drug program implementation questions | Clinical expert response |
|---|---|
1. At what age should somatrogon be stopped? | When growth plates are fused, rGH therapy becomes contraindicated. Afterwards, rGH therapy is continued but at a lower adult GHD dose. After 18 years of age, it becomes the decision of each individual whether to continue with rGH therapy. Some public plans require demonstration of persistent GHD (i.e., measurement of IGF-I off rGH or another GH stimulation test). |
2. What criteria should be taken into consideration when assessing whether treatment should be stopped? Would such criteria depend on growth velocity or fusion of growth plates? | Fusion of the growth plates would be a consideration for stopping somatrogon for the indication of childhood GHD. Other considerations could be bone age > 14 years in girls and bone age > 16 years in boys or height velocity of less than 2 cm per year. |
3. There could be indication creep. Do you anticipate that somatrogon will be prescribed off label in patients with chronic renal failure, Turner syndrome, idiopathic short stature, Prader-Willi syndrome, or adult GHD? | Patients with other non-GHD indications may request somatrogon to enable less frequent injections, but I do not think that the practical uptake will happen soon. Other than for the indication of chronic renal insufficiency (prescribed by nephrologists) that may be covered by public plans, the other non-GHD indications are not covered by public insurance plans. Coverage of rGH for these indications would be accomplished through private insurance, compassionate donations from the drug manufacturer, or out of pocket. It is unlikely that somatrogon would be covered by private insurance if it were prescribed off label. I think that of all the indications, adult GHD may be the one most considered for off-label prescription of somatrogon, but if being prescribed off-label, it may not be covered as readily. |
GH = growth hormone; GHD = growth hormone deficiency; IGF-I = insulin-like growth factor I; rGH = recombinant growth hormone.
Clinical Evidence Selection
The clinical evidence included in the review of somatrogon is presented in 3 sections. The first section, the systematic review, includes pivotal studies provided in the sponsor’s submission to CADTH and Health Canada, as well as those studies that were selected according to an a priori protocol. The second section includes indirect evidence from the sponsor (if submitted) and indirect evidence selected from the literature that met the selection criteria specified in the review. The third section includes sponsor-submitted long-term extension studies and additional relevant studies that were considered to address important gaps in the evidence included in the systematic review.
Systematic Review (Pivotal and Protocol Selected Studies)
Objectives
To perform a systematic review of the beneficial and harmful effects of somatrogon (20 mg/mL or 50 mg/mL pre-filled pen for SC injection) for the long-term treatment of pediatric patients who have growth failure due to an inadequate secretion of endogenous GH.
Methods
Studies selected for inclusion in the systematic review will include pivotal studies provided in the sponsor’s submission to CADTH and Health Canada, as well as those meeting the selection criteria presented in Table 6. Outcomes included in the CADTH review protocol reflect outcomes considered to be important to patients, clinicians, and drug plans.
Of note, the systematic review protocol presented in Table 6 was established before the granting of a Notice of Compliance from Health Canada.
Table 6: Inclusion Criteria for the Systematic Review
Criteria | Description |
|---|---|
Patient population | Patients aged 3 years to < 18 years of age diagnosed with GHD due to an inadequate secretion of endogenous GH Subgroups:
|
Intervention | Somatrogon 0.66 mg/kg body weight administered once weekly by SC injection |
Comparators |
|
Outcomes | Efficacy outcomes:
Harms outcomes:
|
Study design | For example, published and unpublished phase III and phase IV RCTs |
AE = adverse event; GH = growth hormone; GHD = growth hormone deficiency; IGF-I = insulin-like growth factor I; LIS = life interference score; OAT = Observer Assessment Tool; PAT = Participant Assessment Tool; PRO = patient-reported outcome; QoLISSY = Quality of Life in Short Stature Youth; RCT = randomized controlled trial; SAE = serious adverse event; SC = subcutaneous; vs. = versus; WDAE = withdrawal due to adverse event.
The literature search for clinical studies was performed by an information specialist using a peer-reviewed search strategy according to the PRESS Peer Review of Electronic Search Strategies checklist.24
Published literature was identified by searching the following bibliographic databases: MEDLINE All (1946–) via Ovid and Embase (1974–) via Ovid. All Ovid searches were run simultaneously as a multi-file search. Duplicates were removed using Ovid deduplication for multi-file searches, followed by manual deduplication in Endnote. The search strategy consisted of both controlled vocabulary, such as the US National Library of Medicine’s MeSH (Medical Subject Headings), and keywords. The main search concept was somatrogon. Clinical trials registries were searched: the US National Institutes of Health’s ClinicalTrials.gov, the WHO’s International Clinical Trials Registry Platform search portal, Health Canada’s Clinical Trials Database, and the European Union Clinical Trials Register.
No filters were applied to limit the retrieval by study type. Retrieval was not limited by publication date or by language. Conference abstracts were excluded from the search results. See Appendix 1 for the detailed search strategies.
The initial search was completed on June 25, 2021. Regular alerts updated the search until the meeting of the CADTH Canadian Drug Expert Committee on October 27, 2021.
Grey literature (literature that is not commercially published) was identified by searching relevant websites from the Grey Matters: A Practical Tool For Searching Health-Related Grey Literature reference.25 Included in this search were the websites of regulatory agencies (the US FDA and European Medicines Agency). Google was used to search for additional internet-based materials. See Appendix 1 for more information on the grey literature search strategy.
These searches were supplemented by reviewing bibliographies of key papers and through contacts with appropriate experts. In addition, the manufacturer of the drug was contacted for information regarding unpublished studies.
Two CADTH clinical reviewers independently selected studies for inclusion in the review based on titles and abstracts, according to the predetermined protocol. Full-text articles of all citations considered potentially relevant by at least 1 reviewer were acquired. Reviewers independently made the final selection of studies to be included in the review, and differences were resolved through discussion.
Findings From the Literature
A total of 2 studies were identified from the literature for inclusion in the systematic review (Figure 1). The included studies are summarized in Table 7. A list of excluded studies is presented in Appendix 2.
Table 7: Study Characteristics
Characteristics | CP-4-006 | CP-4-009 |
|---|---|---|
Designs and populations | ||
Study design | Open-label, multi-centre, randomized, active-controlled, parallel group study with LT-OLE | Open-label, multi-centre, randomized, active-controlled, parallel group study with LT-OLE |
Locations | Western Europe, Israel, Australia, New Zealand, Canada, US, Greece, Turkey, and others | Japan |
Patient enrolment dates | April 19, 2017 | December 7, 2017 |
Randomized (N) | 228 | 44 |
Inclusion criteria |
Inclusion in the LT-OLE:
|
Inclusion in the LT-OLE:
|
Exclusion criteria |
Exclusion during the LT-OLE:
|
Exclusion during the LT-OLE:
|
Intervention | Somatrogon 0.66 mg/kg once a week through pre-filled pen for SC injection | Somatrogon administered in 3 stepwise escalating doses (0.25 mg/kg per week, 0.48 mg/kg per week, and 0.66 mg/kg per week), each for 2 weeks sequentially. Then for the remaining 46 weeks, patients received somatrogon at a dose of 0.66 mg/kg per week. |
Comparator(s) | Genotropin 0.034 mg/kg once a day through pre-filled pen for SC injection | Genotropin 0.025 mg/kg once a day through pre-filled pen for SC injection |
Duration | ||
Phase | ||
Screening | 12 weeks | 4 weeks |
Open label | 12 months | 12 months |
LT-OLE | Until marketing approval | Until marketing registration in Japan |
Outcomes | ||
Primary end point | Annualized HV after 12 months of treatment | Annualized HV after 12 months of treatment |
Secondary and exploratory end points | Secondary
| Secondary
OLE period
|
(continued) | Other
OLE period
• Harms | — |
Notes | ||
Publications | None | None |
Ab = antibody; ADHD = attention-deficit/hyperactivity disorder; BA = bone age; BM = bone maturation; BMI = body mass index; CA = chronological age; GH = growth hormone; GHD = growth hormone deficiency; GHRP-2 = growth hormone-releasing peptide 2; hGH = human growth hormone; HV = height velocity; IGF-I = insulin-like growth factor I; IGFBP-3 = insulin-like growth factor-binding protein 3; LT-OLE = long-term open-label extension; MHD = multiple hormonal deficiency; OAT = Observer Assessment Tool; OLE = open-label extension; PAT = Participant Assessment Tool; PK/PD = pharmacokinetic/pharmacodynamic; QoL = quality of life; QoLISSY = Quality of Life in Short Stature Youth; rhGH = recombinant human growth hormone; SAE = serious adverse event; SC = subcutaneous; SD = standard deviation; SDS = standard deviation score; SGA = small for gestational age; TB = tuberculosis.
Source: Clinical Study Report for CP-4-006 (2020)10 and Clinical Study Report for CP-4-009 (2020).11
Description of Studies
Two phase III randomized controlled trials, CP-4-006 (C0311009) (N = 224) and CP-4-009 (C0311010) (N = 44) were included in the CADTH systematic review. The details of the 2 trials are provided in Table 7. In CP-4-006, a total of 84 study sites randomized 228 patients of which 224 received at least 1 dose of study drug. In CP-4-009, a total of 24 study sites randomized patients; ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. For CP-4-006, the last patient’s last visit was on August 23, 2019. For CP-4-009, the data cut-off date was March 13, 2020. The main body of the review report presents data of the randomized phase for both studies.
CP-4-006 was an open-label, multi-centre, randomized, active-controlled, parallel group study evaluating the efficacy and safety of weekly somatrogon to daily GH (Genotropin). The study had 2 parts: a main study phase and a long-term open-label phase. The main study phase had a screening period of up to 12 weeks, wherein patients who met the inclusion criteria were randomized in a 1:1 method to receive weekly SC doses of somatrogon or daily SC doses of Genotropin for 12 months. At the end of the screening period, patients were administered a starting dosage of 0.66 mg/kg per week of somatrogon or a starting dosage of 0.034 mg/kg per day of Genotropin (equivalent to 0.24 mg/kg per week divided equally into 7 daily injections, which aligned with Health Canada–approved guidelines). The doses of somatrogon and Genotropin were adjusted every 3 months based on the patient’s body weight and decreased for safety reasons based on predefined dose-adjustment criteria (this was based on the severity of adverse events or repeated, elevated levels of IGF-I SDS). Genotropin was administered as an SC injection in the evening or during bedtime hours once daily, using pre-filled cartridges with the Genotropin Pen delivery device, into the upper arms, buttocks, thighs, or abdomen (8 locations). Injection sites were to be rotated successively (left upper arm, right upper arm, left buttock, right buttock, left thigh, right thigh, left abdomen, and right abdomen), using the same injection site only after all other injection sites had been rotated. Both somatrogon and Genotropin were to be stored, refrigerated at 2°C to 8°C.10
Patients were centrally randomized by geographical region, peak GH levels at screening, and chronological age. Patients were randomized through an interactive web response system (IWRS). Details of the randomization stratification were as follows:
geographical region (region 1 — Western Europe, Israel, Australia, New Zealand, Canada, and the US; region 2 — Central and Eastern Europe, Greece, Turkey, Latin America, and Asia, except for India and Vietnam; region 3 — India and Vietnam)
GH peak levels at screening — 3 ng/mL or less, more than 3 ng/mL to a maximum of 7 ng/mL, and more than 7 ng/mL to a maximum of 10 ng/mL
chronological age of 3 years to 7 years and 0 days, and older than 7 years and 0 days.
In the LT-OLE phase, patients who received somatrogon during the main study continued with the same dosage of somatrogon (mg/kg per week) during the LT-OLE phase. Patients who received Genotropin during the main study were switched to somatrogon and began treatment with a dosage of 0.66 mg/kg per week no less than 1 day after stopping Genotropin treatment. During the entire study (main study and LT-OLE phase), the doses of somatrogon and Genotropin (main study only) were adjusted every 3 months based on the patient’s body weight and may have been decreased for safety reasons, based on predefined dose-adjustment criteria (based on the severity of adverse events or repeated, elevated levels of IGF-I SDS). Patients were free to discontinue from the study at any time. Patients who had withdrawn from the study during the 12 months of treatment post−first dose were not replaced. Patients who had withdrawn after successfully completing screening, but before first dose, were replaced. The main study lasted 15 months (a 12-month treatment period, up to 12 weeks of screening, and a 1-month post-dosing end-of-study follow-up for patients who discontinued the study early or did not continue in the LT-OLE phase). The LT-OLE phase was to continue until marketing approval.
CP-4-009 was a 12-month, open-label, multi-centre, randomized, active-controlled, parallel group study conducted in Japan. It compared the efficacy and safety of weekly somatrogon to daily Genotropin in Japanese prepubertal children with GHD. The study had 2 parts: a main study phase and a long-term open-label phase. In the main study, after a screening period of 4 weeks, patients meeting the inclusion criteria were randomized in a 1:1 ratio to receive either weekly somatrogon SC injections for 12 months or daily Genotropin SC injections (0.025 mg/kg per day, which is equivalent to 0.175 mg/kg per week, divided equally into 7 daily injections over a week) for 12 months. The randomization was not stratified. Somatrogon was administered in 3 stepwise escalating doses (0.25 mg/kg per week, 0.48 mg/kg per week, and 0.66 mg/kg per week), each for 2 weeks sequentially. Then for the remaining 46 weeks, patients continued to receive somatrogon at a dosage of 0.66 mg/kg per week. The doses of somatrogon and Genotropin were adjusted every 3 months based on the patient’s body weight and decreased for safety reasons based on predefined dose-adjustment criteria (based on the severity of adverse events or repeated, elevated levels of IGF-I SDS). Patients were free to discontinue from the study at any time. Patients who had withdrawn from the study before completing 12 months of treatment were not replaced. Both somatrogon and Genotropin were injected subcutaneously using a pen device. Following the completion of the 12-month treatment period, eligible patients were enrolled in a single-arm OLE treatment phase with somatrogon. Eligible Genotropin-treated patients who completed 12 months of treatment during the main study were switched to a somatrogon dosage of 0.66 mg/kg per week and somatrogon-treated patients who completed 12 months of treatment during the main study continued to receive somatrogon with the same dosage in the OLE phase. The OLE phase was to continue until the marketing registration of somatrogon in Japan. The LT-OLE phase has been summarized in the Other Relevant Evidence section of this report.
Populations
Inclusion and Exclusion Criteria
CP-4-006 included prepubertal children who ranged in age from 3 years to younger than 11 years for girls or younger than 12 years for boys, with either isolated GHD or GH insufficiency as part of multiple pituitary hormone deficiency. Patients were treatment naive to other rhGH therapy. Patients included in the study had to have impaired height and height velocity, which were defined as annualized height velocity below the 25th percentile for chronological age (height velocity < –0.7 SDS) and sex, according to the sponsor’s calculator; the interval between 2 height measurements should have been at least 6 months but should not have exceeded 18 months before inclusion. The baseline IGF-I level had to have been at least 1 SD below the mean IGF-I level standardized for age and sex (IGF-I SDS ≤ –1). Key exclusion criteria included children with a prior history of leukemia, lymphoma, sarcoma, or any other form of cancer and history of radiation or chemotherapy. Patients requiring glucocorticoid therapy (e.g., for asthma) who were taking chronically a dose greater than 400 mcg per day of inhaled budesonide or equivalent were excluded. Patients with chromosomal abnormalities including Turner syndrome, Laron syndrome, Noonan syndrome, Prader-Willi syndrome, Russell-Silver syndrome, SHOX mutations/deletions, and skeletal dysplasias were also excluded. Detailed inclusion and exclusion criteria are shown in Table 7. For patients to move into the LT-OLE phase, patients had to complete 12 months of treatment on the main study, with adequate compliance to protocol and checked as per a review of diary cards.
CP-4-009 included prepubertal Japanese children who ranged in age from 3 years to younger than 10 years for girls or younger than 11 years for boys. Other key inclusion and exclusion criteria were similar to that of study CP-4-006.
Baseline Characteristics
The baseline characteristics for CP-4-006 and CP-4-009 are given in Table 8 and Table 9. In general, the baseline characteristics were similar between treatment groups within each of the included studies; however, there are a few differences to note. There was an imbalance between treatment groups in terms of age and sex in both studies, and mean (SD) and median (range) height and weight in CP-4-009. In CP-4-006, in the somatrogon treatment group, 60.6% of the patients were 7 years or older and 39.4% of the patients ranged in age from more than 3 years to a maximum of 7 years. In the Genotropin treatment group, 59.1% of the patients were 7 years or older and 40.9% of the patients ranged in age from more than 3 years to a maximum of 7 years. In CP-4-009 in the somatrogon treatment group, ||||||||% of the patients were |||||||| years or older and ||||||||% of the patients ranged in age from more than |||||||| years to a maximum of |||||||| years. In the Genotropin treatment group, ||||||||% of the patients were |||||||| years or older and ||||||||% of the patients ranged in age from more than |||||||| years to a maximum of |||||||| years. In CP-4-006, 75.2% of the patients were male in the somatrogon treatment group and 68.7% of the patients were male in the Genotropin treatment group. In CP-4-009, 40.9% of the patients were male in the somatrogon treatment group and 54.5% of the patients were male in the Genotropin treatment group. In CP-4-009, the mean (SD) height (cm) was |||||||| and |||||||| in the somatrogon treatment group and Genotropin treatment group, respectively. Similarly, the mean (SD) weight was 14.49 (3.33) kg and 17.87 (4.85) kg in the somatrogon treatment group and Genotropin treatment group, respectively.
In CP-4-006, |||||||| of the patients in the somatrogon treatment group and |||||||| of the patients in the Genotropin treatment group had any prior medication. The most frequently reported prior treatments were |||||||| and ||||||||. In the somatrogon treatment group, ||||||||% of the patients had a prior |||||||| and in the Genotropin treatment group, ||||||||% of the patients had a prior ||||||||. In CP-4-009, ||||||||% of the patients in the somatrogon treatment group and ||||||||% of the patients in the Genotropin treatment group had any prior medication. The most frequently reported prior treatments ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.
Table 8: Summary of Baseline Characteristics — Safety Analysis Population
Characteristic | CP-4-006 | CP-4-009 | ||
|---|---|---|---|---|
Somatrogon (N = 109) | Genotropin (N = 115) | Somatrogon (N = 22) | Genotropin (N = 22) | |
Age, years | ||||
n (%) | 109 (100) | 115 (100) | 22 (100) | 22 (100) |
Mean (SD) | 7.83 (2.66) | 7.61 (2.37) | 5.28 (1.83) | 6.78 (2.34) |
Median (range) | 7.92 (3.01 to 11.96) | 7.84 (3.05 to 11.85) | ||||||||||||||||||| | ||||||||||||||||||| |
> 3 years to ≤ 7 years, n (%) | 43 (39.4) | 47 (40.9) | ||||||||||||||||||| | ||||||||||||||||||| |
> 7 years, n (%) | 66 (60.6) | 68 (59.1) | ||||||||||||||||||| | ||||||||||||||||||| |
Sex, n (%) | ||||
Male | 82 (75.2) | 79 (68.7) | 9 (40.9) | 12 (54.5) |
Female | 27 (24.8) | 36 (31.3) | 13 (59.1) | 10 (45.5) |
Race, n (%) | ||||
White | 81 (74.3) | 86 (74.8) | 0 | 0 |
Black or African-American | 0 | 2 (1.7) | 0 | 0 |
Asian | 24 (22.0) | 21 (18.3) | 22 (100.0) | 22 (100.0) |
American Indian or Alaska Native | 1 (0.9) | 0 | 0 | 0 |
Native Hawaiian or Other Pacific Islander | 0 | 1 (0.9) | 0 | 0 |
Other | 3 (2.8) | 5 (4.3) | 0 | 0 |
Unknown | 0 | 0 | 0 | 0 |
Multiracial | 0 | 0 | 0 | 0 |
Not reported | 0 | 0 | 0 | 0 |
Ethnicity, n (%) | ||||
Hispanic or Latino | 11 (10.1) | 13 (11.3) | NR | NR |
Non-Hispanic or non-Latino | 98 (89.9) | 102 (88.7) | NR | NR |
Height (cm) | ||||
n (%) | 109 (100) | 115 (100) | 22 (100.0) | 22 (100.0) |
Mean (SD) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
Median (range) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
Weight (kg) | ||||
n (%) | 109 (100) | 115 (100) | 22 (100.0) | 22 (100.0) |
Mean (SD) | ||||||||||||||||||| | ||||||||||||||||||| | 14.49 (3.33) | 17.87 (4.85) |
Median (range) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
BMI (kg/m2) | ||||
n (%) | 109 (100) | 115 (100) | 22 (100.0) | 22 (100.0) |
Mean (SD) | ||||||||||||||||||| | ||||||||||||||||||| | 15.27 (1.30) | 15.89 (1.09) |
Median (range) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
BMI = body mass index; NR = not reported; SD = standard deviation.
Source: Clinical Study Report for CP-4-006 (2020)10 and Clinical Study Report for CP-4-009 (2020).11
Table 9: Summary of Other Characteristics — Safety Analysis Population
Characteristic | CP-4-006 | CP-4-009 | ||
|---|---|---|---|---|
Somatrogon (N = 109) | Genotropin (N = 115) | Somatrogon (N = 22) | Genotropin (N = 22) | |
Region, n (%) | ||||
Region 1 (Western Europe, Israel, Greece, Australia, New Zealand, Canada, and the US) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
Region 2 (Central and Eastern Europe, Turkey, Latin America, and Asia, except for India and Vietnam) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
Region 3 (India and Vietnam) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
Peak GH level group, n (%) | ||||
≤ 3 ng/mL | 22 (20.18) | 21 (18.26) | ||||||||||||||||||| | ||||||||||||||||||| |
> 3 ng/mL to ≤ 7 ng/mL | 53 (48.62) | 56 (48.70) | ||||||||||||||||||| | ||||||||||||||||||| |
> 7 ng/mL | 34 (31.19) | 38 (33.04) | ||||||||||||||||||| | ||||||||||||||||||| |
Peak GH (ng/dL) | ||||
n (%) | 109 (100) | 115 (100) | 22 (100.0) | 22 (100.0) |
Mean (SD) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
Median (range) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
Peak GH level | ||||
Low | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
High | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
Father’s height (cm) | ||||
n (%) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
Mean (SD) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
Median (range) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
Mother’s height (cm) | ||||
n (%) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
Mean (SD) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
Median (range) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
Target height, males (cm)a | ||||
n (%) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
Mean (SD) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
Median (range) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
Target height, females (cm)b | ||||
n (%) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
Mean (SD) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
Median (range) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
Bone agec | ||||
n (%) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
Mean (SD) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
Median (range) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
Bone maturationd | ||||
n (%) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
Mean (SD) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
Median (range) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
Height (SDS) | ||||
n (%) | 109 (100) | 115 (100) | ||||||||||||||||||| | ||||||||||||||||||| |
Mean (SD) | –2.94 (1.29) | –2.78 (1.27) | ||||||||||||||||||| | ||||||||||||||||||| |
Median (range) | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| |
GH = growth hormone; SD = standard deviation; SDS = standard deviation score.
aTarget height: Males (cm) = (father’s height [cm] + mother’s height [cm] + 13) ÷ 2.
bTarget height: Females (cm) = (father’s height [cm] + mother’s height [cm] − 13) ÷ 2.
cBone age determination with the method of Tanner-Whitehouse 2, using a central bone age reader.
dBone maturation is calculated as bone age divided by chronological age.
Source: Clinical Study Report for CP-4-006 (2020)10 and Clinical Study Report for CP-4-009 (2020).11
Interventions
In CP-4-006, somatrogon was provided as a solution for injection, containing 20 mg/mL or 50 mg/mL in a multi-dose, disposable pre-filled pen. The formulation included citrate, histidine, sodium chloride, metacresol, poloxamer 188, and pH = 6.6. Genotropin was provided in a 2-chamber cartridge for administration with the Genotropin Pen delivery device (Goquick Pen delivery device). The Genotropin Pen was used for daily (evening or bedtime) SC administration of Genotropin. Training on drug administration was provided for patients and parents or a legal guardian. The device was intended to assist self-injecting adult and pediatric patients, health care professionals, and caregivers with the daily SC injection, primarily self-administered at home or in a health care environment. The starting dose for somatrogon administration was 0.66 mg/kg per week. The starting dose regimen for Genotropin was 0.034 mg/kg per day (or 0.24 mg/kg per week divided equally into 7 injections over a week). If a patient on Genotropin missed a dose, they could resume the medication with the next scheduled dose. In case the prescribed dose could not be fully set for a single injection on a pen, the patient was instructed to split the dose into 2 injections. The partial dosing could occur in 2 cases:
Do 2 injections using 1 pen — In the event that the prescribed dose was higher than the maximum dose, which could be selected according to the pen amount, the patient was instructed to subtract the dose already received from the prescribed dose and set the pen accordingly.
Split dose between 2 pens, the current pen and a new pen — This could happen when a complete dose could not be fully administered from the pen in use; in this case, the patient was instructed to subtract the dose already delivered from the prescribed dose and set the new pen accordingly.
Whether from the same pen or from a new pen, it was important with the second injection that the patient replace the needle and rotate the injection. The doses of somatrogon and Genotropin (main study only) were assessed every 3 months based on a patient’s body weight. Doses were determined by the IWRS and included an automatic rounding to the closest pen increment (0.2 increments in 20 mg/mL pens and 0.5 increments in 50 mg/mL pens). Doses were decreased for safety reasons according to the predefined, dose-adjustment criteria (which were based on the severity of adverse events or repeated, elevated levels of IGF-I SDS). For patients on somatrogon, the dose was decreased based on 2 repeated day-4 levels of IGF-I greater than 2.0 SDS. For patients on Genotropin, the dose was decreased based on repeated IGF-I levels greater than 2.0 SDS. If a patient had an IGF-I level greater than 2.0 SDS, they were to return for an unscheduled visit within 4 weeks to 6 weeks after the greater than 2.0 SDS result, on day 4 post dose for somatrogon-treated patients or any day for Genotropin-treated patients. If their IGF-I level was still greater than 2.0 SDS, the most recent dose was to be reduced by 15% (i.e., to 0.56 mg/kg per week for somatrogon and 29 mcg/kg per day for Genotropin). The patient was to be treated with the new dose for at least 4 weeks before a subsequent IGF-I determination could result in a further dose modification. At the time of the next visit, if the IGF-I was still greater than 2.0 SDS, the dose was to be reduced by an additional 15% to 0.48 mg/kg per week for somatrogon and to 24.7 mcg/kg per day for Genotropin. If the IGF-I was still greater than 2.0 SDS following 2 dose reductions (at least 4 weeks after the second dose reduction), the course of treatment would be decided on an individual basis. If a patient on somatrogon treatment missed a dose by not more than 72 hours, then the patient had to take a full dose as soon as they remembered. If the dose was more than 72 hours late, the patient could not take a dose for the whole week and would continue taking the study medication on the regular day the following week.
In CP-4-009, somatrogon was provided as a solution for injection, containing 20 mg/mL or 50 mg/mL in a multi-dose, disposable pre-filled pen. The device used in this study had not been approved or certified in Japan. The formulation included citrate, histidine, sodium chloride, metacresol, poloxamer 188, and pH = 6.6. Genotropin was provided in a 2-chamber cartridge for administration with the Genotropin Pen delivery device (Goquick Pen delivery device). If a patient on somatrogon treatment missed a dose for not more than 72 hours, then the patient had to take a full dose as soon as they remembered. If the dose was more than 72 hours late, the patient could not take a dose for the whole week and would continue taking the study medication on the regular day the following week. The starting dosage regimen for Genotropin was 0.025 mg/kg per day (or 0.175 mg/kg per week divided equally to 7 injections over a week). If a patient on Genotropin missed a dose, they could resume the medication with the next scheduled dose. In case the prescribed dose could not be fully set for a single injection on a pen, the patient was instructed to split the dose into 2 injections. The partial dosing could occur in 2 cases:
Do 2 injections using 1 pen — In the event that the prescribed dose was higher than the maximum dose (24 mg pen is 12 mg and the 60 mg pen is 30 mg), which could be selected according to the pen amount, the patient was instructed to subtract the dose already received from the prescribed dose and set the pen accordingly.
Split dose between 2 pens, the current pen and a new pen — This could happen when a complete dose could not be fully administered from the pen in use; in this case, the patient was instructed to subtract the dose already delivered from the prescribed dose and set the new pen accordingly.
Whether from the same pen or from a new pen, it was important with the second injection that the patient replace the needle and rotate the injection. The doses of somatrogon and Genotropin (main study only) were assessed every 3 months based on a patient’s body weight. Doses were determined by the interactive response technology (IRT) and included an automatic rounding to the closest pen increment (0.2 increments in 20 mg/mL pens and 0.5 increments in 50 mg/mL pens). Doses were decreased for safety reasons according to the predefined, dose-adjustment criteria (which were based on the severity of adverse events or repeated, elevated levels of IGF-I SDS). For patients on somatrogon, the dose was decreased based on 2 repeated day-4 levels of IGF-I greater than 2.0 SDS. For patients on Genotropin, the dose was decreased based on repeated IGF-I levels greater than 2.0 SDS. If a patient had an IGF-I level greater than 2.0 SDS, they were to return for an unscheduled visit within 4 weeks to 6 weeks after the greater than 2.0 SDS result, on day 4 post dose for somatrogon-treated patients or any day for Genotropin-treated patients. If their IGF-I level was still greater than 2.0 SDS, the most recent dose was to be reduced by 15% (i.e., to 0.56 mg/kg per week for somatrogon and 21 mcg/kg per day for Genotropin). The patient was to be treated with the new dose for at least 4 weeks before a subsequent IGF-I determination could result in a further dose modification. At the time of the next visit, if the IGF-I was still greater than 2.0 SDS, the dose was to be reduced by an additional 15% to 0.48 mg/kg per week for somatrogon and to 18 mcg/kg per day for Genotropin. If the IGF-I was still greater than 2.0 SDS following 2 dose reductions (at least 4 weeks after the second dose reduction), the course of treatment would be decided on an individual basis.
In CP-4-006, in the somatrogon treatment group, |||||||||% of the patients received concomitant medications and in the Genotropin treatment group, |||||||||% of the patients received concomitant medications. The most frequent medications included ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. In CP-4-009, in the somatrogon treatment group, |||||||||% of the patients received concomitant medications and in the Genotropin treatment group, |||||||||% of the patients received concomitant medications. The most frequently reported concomitant medication included |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.
Outcomes
A list of efficacy end points identified in the CADTH review protocol that were assessed in the clinical trials included in this review is provided in Table 10. These end points are further summarized as follows. A detailed discussion and critical appraisal of the outcome measures is provided in Appendix 3.
Table 10: Summary of Outcomes of Interest Identified in the CADTH Review Protocol
Outcome measure | CP-4-006 | CP-4-009 |
|---|---|---|
Height velocity | Primary | Primary |
Auxologic response | Secondary | Secondary |
Serum biomarker levels | Secondary | Secondary |
HRQoL | Other | NR |
Absent or delayed sexual development during puberty | NR | NR |
Other PRO response | Other | NR |
HRQoL = health-related quality of life; NR = not reported; PRO = patient-reported outcome.
Source: Clinical Study Report for CP-4-006 (2020)10 and Clinical Study Report for CP-4-009 (2020).11
Height Velocity
The primary efficacy outcome evaluated the annual height velocity in cm per year at 12 months. This was based on the difference between the heights at 12 months and baseline. Height was to be measured using a calibrated wall-mounted stadiometer and this process was to be conducted at the same time of the day for each visit, preferably in the morning. To ensure the consistency of results, ideally the same auxologist would perform the measurements for each patient at each visit. Three independent readings were recorded for each visit.
Auxologic Response
Auxologic responses were the secondary outcomes, evaluated as annualized height velocity at 6 months, a change in height SDS at 6 months and 12 months, and a change in bone maturation at 12 months. Annualized heigh velocity at 6 months was estimated based on the difference between the heights at 6 months and baseline. Changes in height SDS at 6 months and 12 months were determined from the age and sex standards listed in the US’s 2000 Centers for Disease Control and Prevention growth charts in CP-4-006 and from the age and gender standards listed in the national survey in year 2000 in CP-4-009. Change in bone maturation was estimated at 12 months. Bone maturation was calculated as bone age divided by chronological age. The bone age was to be determined by an X-ray according to the Greulich-Pyle method.10,11
Serum Biomarker Levels
Clinical evaluations of IGF-I and IGF-I SDS were performed at every study visit during the main study except visit 3, visit 6a, visit 6b, visit 8a, and visit 9. Descriptive statistics were used to summarize the results of serum biomarker levels.
Health-Related Quality of Life
CP-4-006 used the QoLISSY questionnaire to assess HRQoL. The questionnaire was designed for self-reported use by adolescents between the ages of 8 years and 18 years and observer-reported use by parents of children between the ages of 4 years and 18 years for assessment of HRQoL. Items from 3 core domains (physical, social, and emotional) are summed for a 22-item total score. The core domains are accompanied by 28 additional items reflecting coping (10 items), beliefs (4 items), and treatment (14 items). Items are answered using a 5-point Likert type scale; subscale scores are transformed to a score from 0 to 100 where higher values represent a higher HRQoL. The 3 dimensions of the QoLISSY questionnaire were calculated individually and as a combined core total score based on the QoLISSY scoring manual. The core score is calculated as the sum of the means of these 3 dimensions and divided by 3. MID is not estimated for QoLISSY. HRQoL using the QoLISSY was not reported in the CP-4-009 study. The QoLISSY questionnaire was ||||||||||||||||||||||||||| completed for the following countries using a validated translated tool: US, Australia, New Zealand, Belarus, Russia, Ukraine, UK, and Spain.
Absent or Delayed Sexual Development During Puberty
Absent or delayed sexual development during puberty was not reported as an outcome in either study but was identified by CADTH as an outcome of interest.
Other Patient-Reported Outcome Responses
In CP-4-006, the OAT was used to record the observer’s assessment of the use of the device by patients or caregivers to administer somatrogon by injection. The observer recorded whether an injection attempt was successful or not based on the question from the OAT, “…did the user successfully inject into an acceptable injection site without physical assistance?” The injection attempt was considered successful if the observer answered “yes” to the questions for all attempts on the form. Of note, the instructions for the OAT advised the observer as follows: “|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.” This introduces substantial subjectivity into the assessment of a successful injection administration.9
The PAT was also used to record the patient’s or caregiver’s injections of somatrogon, based on an assessment by the user of the pen or by the parent or legal guardian. The following questions were used to determine a successful injection attempt via the PAT: “Did the dose window show ‘0’ when you finished your injection?” and “Do you believe that a full dose was injected?” The injection attempt was considered successful if the subject answered yes to both questions for all attempts on the form.10 Other PRO responses were not reported in the CP-4-009 study.
Evidence of MID, validity, reliability, and responsiveness of the OAT and PAT were not identified during this review.
Statistical Analysis
CP-4-006 aimed to establish noninferiority of somatrogon compared to Genotropin by a noninferiority margin of −1.8 cm per year. Noninferiority was concluded if the lower bound of the 2-sided 95% CI for the mean treatment difference between somatrogon and Genotropin in the primary efficacy end point was greater than or equal to –1.8 cm per year or more. In CP-4-006, an estimated sample size of 100 patients per treatment group were required to achieve at least 80% power for the noninferiority test between the treatment groups with a 2-sided 0.05 level of significance and assumed 10% dropout rate for each treatment group. The between-patient SD of annual growth rate of 2.5 cm per year in both treatment groups was assumed. The true mean treatment difference in the primary efficacy end point was assumed to be –0.8 cm per year. It was planned that 110 patients would be randomized to each treatment group for a total of 220 patients.
The rationale for the choice of the noninferiority margin was based on:
Historical data from MacGillivray et al. (1996)26 and Wilton and Gunnarsson (1988)27 height velocity response for the first year of daily GH ranged from 10.2 cm per year (SD = 2.5) to 11.4 cm per year (SD = 2.5). Using the SD of 2.5 from these references, a noninferiority margin of –1.8 cm per year was within 1 SD of the expected results, and approximately 23% of the reference treatment response distribution would be below this value.
Assuming the height velocity response for daily GH treatment is 11.5 cm per year in the first year, a margin of –1.8 cm per year would show that 84% of the growth rate from the reference daily GH treatment effect on the approved active control is retained.
Other studies of long-acting GH compared to daily GH have used noninferiority margins of –1.8 cm to –2.0 cm per year as used in a phase III pivotal trial (NCT02781727). The use of –1.8 cm per year is the more conservative value based on the precedent set with these other studies.
If the condition for noninferiority was met, an assessment of superiority was conducted on the primary end point. Superiority would be achieved if the lower bound of the 2-sided 95% CI for the mean height velocity difference of somatrogon minus Genotropin is greater than or equal to 0 cm per year or more. Due to the sequential tests, no correction for multiple comparisons was performed.
CP-4-009 aimed to establish comparability of somatrogon to Genotropin by a comparability margin of −1.8 cm per year. Comparability was concluded if the mean treatment difference between somatrogon and Genotropin in the primary efficacy end point was greater than or equal to –1.8 cm per year or more. In CP-4-009, an estimated sample size of 20 patients per treatment group was required to achieve at least 88% power to detect that the observed difference between the treatment groups was greater than –1.8 cm per year. The between-patient SD of annual growth rate of 2.5 cm per year in both treatment groups was assumed. The true mean treatment difference in the primary efficacy end point was assumed to be –0.8 cm per year.
In CP-4-009, statistical hypothesis testing for superiority was not planned.
For both studies, in the primary efficacy analysis of annualized height velocity at 12 months, the CI for the difference of means between the 2 treatments was derived from an analysis of covariance (ANCOVA) model using multiple imputations assuming missing not at random (MNAR) on the full analysis set population and imputing the missing results. The ANCOVA model included classification terms for treatment, age group (3 years to 7 years and 0 days or > 7 years and 0 days), sex (male or female), peak GH levels (≤ 3 ng/mL, > 3 to ≤ 7 ng/mL, and > 7 to ≤ 10 ng/mL), and region (region 1: Western Europe, Israel, Australia, New Zealand, Canada, and the US; region 2: Central and Eastern Europe, Greece, Turkey, Latin America, and Asia, except for India and Vietnam; region 3: India and Vietnam). The model also included baseline height SDS as a covariate. The determination of noninferiority was based on LSM for the 2 treatments from the ANCOVA model and the 95% CI of the differences between the treatments. If the primary end point of noninferiority of somatrogon in comparison to Genotropin was met, an assessment of superiority of somatrogon over Genotropin at 12 months was to be conducted. Superiority was achieved if the lower bound of the 2-sided 95% CI for the mean height velocity difference between somatrogon and Genotropin was 0 cm per year or more. The MNAR and fully conditional specification method using multiple imputations was applied for the primary efficacy outcome.
For both studies, a similar ANCOVA model that was used for the primary end point was used to analyze the auxologic responses (i.e., annualized height velocity at 6 months, and a change in height SDS at 6 months and 12 months), which were the secondary end points. LSM estimates for the 2 treatments and the 95% CI of the difference between the treatments will be presented. For bone maturation, descriptive statistics (including univariate 95% CI) were reported.
In CP-4-006, for serum biomarker levels, descriptive statistics were reported for observed factors and change from baseline for the biochemical end points at each visit. The number and percent of patients who had IGF-I SDS greater than 2 was summarized at each visit. The number and percent of patients who achieved IGF-I normalization (defined as IGF-I SDS between –0.5 and 1.5, inclusive) were summarized. The number and percent of patients who had IGF-I SDS greater than 2.0 were summarized at each visit. This was not reported in the CP-4-009 study.
In CP-4-006, HRQoL was analyzed using the QoLISSY tool. The QoLISSY questionnaire had 3 dimensions (physical, social, and emotional) that would be calculated individually and as a combined core total score based on the QoLISSY scoring manual. Scores for individual dimensions were transformed from raw scores to a scale of 0 to 100. The core score was calculated as the sum of the means of these 3 dimensions and divided by 3. This tool was not used in CP-4-009.
In CP-4-006, other PROs were summarized using descriptive statistics for the OAT and the PAT. The number and percentage of successful single injections for PAT was to be summarized overall. The number and percentage of successful single injections for OAT was to be summarized overall. The number and percentage of successful single injections for PAT and OAT was summarized by age group, sex, and race. The number and percentage of the number of attempts required to achieve a success was summarized for PAT and OAT. The OAT and PAT were performed only in the US. This tool was not used in CP-4-009.
Missing Data
In both studies, for the primary and secondary end point analyses performed by ANCOVA, multiple imputations assuming MNAR were used to impute missing results. The imputation was by treatment group. The imputation model included randomization stratification factors and baseline height SDS. A total of 100 imputed datasets were created and the seed was set using the database lock date. An ANCOVA model was used to calculate the LSM and 95% CI of the treatment difference for each imputed set. The number of imputed datasets was increased if necessary. For somatrogon, values below the limit of quantification at the baseline visit were treated as 0.
Sensitivity Analysis
The ANCOVA-based primary efficacy analysis was performed using the per-protocol (PP) set.
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Subgroup Analysis
Although several subgroups were identified by the sponsor, only those subgroups identified in the protocol are presented (i.e., age, sex, risk of GHD, and prior use of GH therapy).
Analysis populations
The safety analysis set included all patients who had received at least 1 dose of study treatment. Patients were analyzed according to actual treatment received.
The efficacy analyses were based on the full analysis set, defined as patients who were randomized and had received at least 1 dose of study medication. Patients were analyzed according to randomized treatment group.
The PP set consisted of all randomized patients who did not have any major protocol deviations. Patients who had major protocol deviations were identified in a blinded review.
Results
Patient Disposition
Patient disposition and protocol violations for CP-4-006 and CP-4-009 are presented in Table 11. In CP-4-006, 0.9% of the patients discontinued in each of the treatment groups. In CP-4-009, 4.5% of the patients in the Genotropin treatment group discontinued from treatment. In the somatrogon treatment group, |||||||||% of the patients were continued over to the LT-OLE phase, and |||||||||% of the patients in the Genotropin treatment group continued over to the LT-OLE phase. Protocol violations were documented in both studies. In CP-4-006, |||||||||% and |||||||||% of patients reported major protocol violations in the somatrogon treatment group and Genotropin treatment group, respectively. In the somatrogon treatment group, |||||||||% of patients reported drug and dosing administration-related protocol violations while in the Genotropin treatment group, |||||||||% of patients reported the same. Patients with a major protocol violation were excluded from the PP set. In CP-4-009, |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.
Patient disposition | CP-4-006 | CP-4-009 | ||
|---|---|---|---|---|
Somatrogon | Genotropin | Somatrogon | Genotropin | |
Screened, N | 536 | 65 | ||
Randomized and received at least 1 dose of study drug | 224 | 44 | ||
Randomized, n (%) | 109 (100) | 115 (100) | 22 (100) | 22 (100) |
Discontinued from study, n (%) | 1 (0.9) | 1 (0.9) | 0 | 1 (4.5) |
Reason for discontinuation, n (%) | ||||
Adverse events | 1 (0.9) | 0 | 0 | 1 (4.5) |
Lost to follow-up | 0 | 0 | 0 | 0 |
Withdrawal by parent/guardian or patient | 0 | 1 (0.9) | 0 | 0 |
Death | 0 | 0 | 0 | 0 |
Completed, rolled over to OLE, n (%) | ||||||||| | ||||||||| | ||||||||| | ||||||||| |
Completed, not rolled over to OLE, n (%) | ||||||||| | ||||||||| | ||||||||| | ||||||||| |
FAS, n (%) | ||||||||| | ||||||||| | ||||||||| | ||||||||| |
PP, n (%) | ||||||||| | ||||||||| | ||||||||| | ||||||||| |
SAS, n (%) | ||||||||| | ||||||||| | ||||||||| | ||||||||| |
FAS = full analysis set; PP = per-protocol; OLE = open-label extension; SAS = safety analysis set.
Source: Clinical Study Report for CP-4-006 (2020)10 and Clinical Study Report for CP-4-009 (2020).11
Exposure to Study Treatments
The extent of exposure was evaluated in the safety analysis set. The extent of exposure in the study is presented in Table 12. In CP-4-006, the main study lasted up to 15 months (a 12-month treatment period, up to 12 weeks of screening, and a 1-month post-dosing end-of-study follow-up for patients who discontinued the study early or did not continue in the LT-OLE phase). In CP-4-009, the total duration of patient participation in the main study lasted up to 14 months (up to 5 weeks of screening, 12 months of active treatment, and 1 month of follow-up).
Table 12: Extent of Exposure — Safety Analysis Set
Duration of treatment (days) | CP-4-006 | CP-4-009 | ||
|---|---|---|---|---|
Somatrogon N = 109 | Genotropin N = 115 | Somatrogon N = 22 | Genotropin N = 22 | |
n (%) | 109 (100) | 115 (100) | 22 (100) | 22 (100) |
Mean (SD) | ||||||||| | ||||||||| | ||||||||| | ||||||||| |
Median (range) | ||||||||| | ||||||||| | ||||||||| | ||||||||| |
Dose reduction due to IGF-I SDS > 2, n (%) | ||||||||| | ||||||||| | ||||||||| | ||||||||| |
IGF-I = insulin-like growth factor I; SD = standard deviation; SDS = standard deviation score.
Note: Extent of exposure is only for the main study phase.
Source: Clinical Study Report for CP-4-006 (2020)10 and Clinical Study Report for CP-4-009 (2020).11
Efficacy
Only those efficacy outcomes and analyses of subgroups identified in the review protocol are reported as follows. Absent or delayed sexual development during puberty was not an efficacy outcome analyzed in either CP-4-006 or CP-4-009.
Height Velocity
In study CP-4-006, the LSM treatment difference for the mean height velocity after 12 months of treatment was 0.33 cm per year (95% CI, –0.24 to 0.89). The lower bound of the 2-sided 95% CI for mean height velocity was greater than the pre-specified noninferiority margin of –1.8 cm per year, indicating that weekly somatrogon administration is noninferior to daily Genotropin administration. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. In study CP-4-009, the treatment difference (somatrogon minus Genotropin) in LSM height velocity (cm per year) was 1.79 cm per year with a 2-sided 95% CI (0.97 to 2.61). Since the point estimate of 1.79 cm per year was greater than the pre-established margin of –1.8 cm per year, it was concluded that weekly somatrogon was comparable to daily Genotropin. The results of the primary efficacy outcome are presented in Table 13. |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.
Table 13: Annual Height Velocity at 12 Months — Full Analysis Set
Annual HV | CP-4-006 | CP-4-009 | ||
|---|---|---|---|---|
Somatrogon N = 109 | Genotropin N = 115 | Somatrogon N = 22 | Genotropin N = 22 | |
Observed end point | ||||
n | 108 | 113 | 22 | 21 |
Mean (SD) | ||||||||| | ||||||||| | ||||||||| | ||||||||| |
Median (range) | ||||||||| | ||||||||| | ||||||||| | ||||||||| |
ANCOVAa | ||||
LSM estimate | 10.10 | 9.78 | 9.654 | 7.868 |
Treatment mean difference (95% CI) | 0.33 (–0.24 to 0.89)b | 1.786 (0.966 to 2.605)c | ||
P value | NR | NR | ||
ANCOVA = analysis of covariance; CI = confidence interval; GH = growth hormone; HV = height velocity; LSM = least squares mean; NR = not reported; SD = standard deviation.
aANCOVA model with classification terms for treatment, age group, sex, peak GH levels, and region.
bNoninferiority was concluded if the lower bound of the 2-sided 95% CI was –1.8 or more. Superiority will be achieved if the lower bound of the 2-sided 95% CI is 0 cm per year or more.
cComparability was concluded if the point estimate of the LSM treatment difference was –1.8 or more.
Source: Clinical Study Report for CP-4-006 (2020)10 and Clinical Study Report for CP-4-009 (2020).11
Auxologic Response
The secondary efficacy outcome was the mean annualized height velocity at 6 months. In CP-4-006, the treatment mean difference was ||||||||||||||||||||||||||||||||||||||. In CP-4-009, the treatment mean difference was |||||||||||||||||||||||||||||||| in favour of somatrogon. The results of the mean annualized height velocity at 6 months are presented in Table 14.
Change in height SDS was evaluated at 6 months and 12 months. In CP-4-006, the treatment mean difference between the somatrogon treatment group and Genotropin treatment group at 6 months was ||||||||||||||||||||||||||||||||. In CP-4-009, the treatment mean difference between the somatrogon treatment group and Genotropin treatment group at 6 months was |||||||||||||||||||||||||||||| in favour of somatrogon. In CP-4-006, the treatment mean difference between the somatrogon and Genotropin treatment groups at 12 months was 0.05 (95% CI, –0.06 to 0.16). In CP-4-009, the treatment mean difference between the somatrogon and Genotropin treatment groups at 12 months was ||||||||||||||||||||||||||||||||| in favour of somatrogon. The results of a change in height SDS at 6 months and 12 months are presented in Table 15 and Table 16, respectively.
Change from baseline in bone maturation was evaluated at 12 months and was similar between the treatment groups of somatrogon and Genotropin. In CP-4-006, the mean (SD) change from baseline was 0.05 (0.09) and 0.06 (0.10) in the somatrogon group and Genotropin group, respectively. |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. The results are presented in Table 17.
Table 14: Annual Height Velocity at 6 Months — Full Analysis Set
Annual HV | CP-4-006 | CP-4-009 | ||
|---|---|---|---|---|
Somatrogon N = 109 | Genotropin N = 115 | Somatrogon N = 22 | Genotropin N = 22 | |
Observed end point | ||||
n (%) | 108 (99.1) | 114 (99.1) | 22 (100) | 21 (95.4) |
Mean (SD) | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
Median (range) | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
ANCOVAa | ||||
LSM estimate | 10.59 | 10.04 | |||||||||||||||||| | |||||||||||||||||| |
Treatment mean difference (95% CI)b | |||||||||||||||||| | |||||||||||||||||| | ||
P value | NR | NR | ||
ANCOVA = analysis of covariance; CI = confidence interval; GH = growth hormone; HV = height velocity; LSM = least squares mean; NR = not reported; SD = standard deviation.
aANCOVA model with classification terms for treatment, age group, sex, peak GH levels, and region.
bCalculated as somatrogon minus Genotropin.
Source: Clinical Study Report for CP-4-006 (2020)10 and Clinical Study Report for CP-4-009 (2020).11
Table 15: Change in Height Standard Deviation Score at 6 Months — Full Analysis Set
Height SDS | CP-4-006 | CP-4-009 | ||
|---|---|---|---|---|
Somatrogon N = 109 | Genotropin N = 115 | Somatrogon N = 22 | Genotropin N = 22 | |
Observed end point | ||||
n (%) | 108 (99.1) | 114 (99.1) | 22 (100) | 21 (95.4) |
Mean (SD) | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
Median (range) | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
ANCOVAa | ||||
LSM estimate | 0.54 | 0.48 | 0.576 | 0.312 |
Treatment mean difference (95% CI)b | |||||||||||||||||| | |||||||||||||||||| | ||
P value | NR | NR | ||
ANCOVA = analysis of covariance; CI = confidence interval; GH = growth hormone; LSM = least squares mean; NR = not reported; SD = standard deviation; SDS = standard deviation score.
aANCOVA model with classification terms for treatment, age group, sex, peak GH levels, and region.
bCalculated as somatrogon minus Genotropin.
Source: Clinical Study Report for CP-4-006 (2020)10 and Clinical Study Report for CP-4-009 (2020).11
Table 16: Change in Height Standard Deviation Score at 12 Months — Full Analysis Set
Height SDS | CP-4-006 | CP-4-009 | ||
|---|---|---|---|---|
Somatrogon N = 109 | Genotropin N = 115 | Somatrogon N = 22 | Genotropin N = 22 | |
Observed end point | ||||
n (%) | 108 (99.1) | 113 (98.3) | 22 (100) | 21 (95.4) |
Mean (SD) | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
Median (range) | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
ANCOVAa | ||||
LSM estimate | 0.92 | 0.87 | 0.939 | 0.523 |
Treatment mean difference (95% CI)b | 0.05 (–0.06 to 0.16) | |||||||||||||||||| | ||
P value | NR | NR | ||
ANCOVA = analysis of covariance; CI = confidence interval; GH = growth hormone; LSM = least squares mean; NR = not reported; SD = standard deviation; SDS = standard deviation score.
aANCOVA model with classification terms for treatment, age group, sex, peak GH levels, and region.
bCalculated as somatrogon minus Genotropin.
Source: Clinical Study Report for CP-4-006 (2020)10 and Clinical Study Report for CP-4-009 (2020).11
Table 17: Change in Bone Maturation at 12 Months — Full Analysis Set
Bone maturation | CP-4-006 | CP-4-009 | ||
|---|---|---|---|---|
Somatrogon N = 109 | Genotropin N = 115 | Somatrogon N = 22 | Genotropin N = 22 | |
Observed baseline | ||||
n (%) | 104 (95.4) | 102 (88.7) | 22 (100) | 21 (95.4) |
Mean (SD) | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
Median (range) | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
95% CI | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
Change from baseline | ||||
n (%) | 104 (95.4) | 102 (88.7) | 22 (100) | 21 (95.4) |
Mean (SD) | 0.05 (0.09) | 0.06 (0.10) | |||||||||||||||||| | |||||||||||||||||| |
Median (range) | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
95% CI | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
CI = confidence interval; SD = standard deviation.
Note: Bone maturation is calculated as bone age divided by chronological age.
Source: Clinical Study Report for CP-4-006 (2020)10 and Clinical Study Report for CP-4-009 (2020).11
Serum Biomarker Levels
For the purposes of this review, serum biomarker levels were evaluated using IGF-I levels and IGF-I SDS. Results were reported as change from baseline at 6 months and 12 months. The mean absolute IGF-I and IGF-I SDS values were reported only in the CP-4-006 study. At 6 months, the mean (SD) change from baseline in IGF-I levels was ||||||||||||| in the somatrogon treatment group and ||||||||||||| in the Genotropin treatment group. At 12 months, the mean (SD) change from baseline in IGF-I levels was ||||||||||||| in the somatrogon treatment group and ||||||||||||| in the Genotropin treatment group. At 6 months, the mean (SD) change from baseline in IGF-I SDS was ||||||||||||| in the somatrogon treatment group and ||||||||||||| in the Genotropin treatment group. At 12 months, the mean (SD) change from baseline in IGF-I levels was ||||||||||||| in the somatrogon treatment group and ||||||||||||| in the Genotropin treatment group. Results of the serum biomarker levels are presented in Table 18 and Table 19.
Table 18: Summary of Mean Absolute IGF-I Level — Full Analysis Set (CP-4-006)
IGF-I (ng/mL) level | Somatrogon N = 109 | Genotropin N = 115 |
|---|---|---|
Observed (baseline) | ||
n (%) | ||||||||||||| | ||||||||||||| |
Mean (SD) | ||||||||||||| | ||||||||||||| |
Median (range) | ||||||||||||| | ||||||||||||| |
Observed (6 months) | ||
n (%) | ||||||||||||| | ||||||||||||| |
Mean (SD) | ||||||||||||| | ||||||||||||| |
Median (range) | ||||||||||||| | ||||||||||||| |
Change from baseline (6 months) | ||
n (%) | ||||||||||||| | ||||||||||||| |
Mean (SD) | ||||||||||||| | ||||||||||||| |
Median (range) | ||||||||||||| | ||||||||||||| |
Observed (12 months) | ||
n (%) | ||||||||||||| | ||||||||||||| |
Mean (SD) | ||||||||||||| | ||||||||||||| |
Median (range) | ||||||||||||| | ||||||||||||| |
Change from baseline (12 months) | ||
n (%) | ||||||||||||| | ||||||||||||| |
Mean (SD) | ||||||||||||| | ||||||||||||| |
Median (range) | ||||||||||||| | ||||||||||||| |
IGF-I = insulin-like growth factor I; SD = standard deviation.
Source: Clinical Study Report for CP-4-006 (2020).10
Table 19: Summary of IGF-I Standard Deviation Score — Full Analysis Set (CP-4-006)
IGF-I SDS | Somatrogon N = 109 | Genotropin N = 115 |
|---|---|---|
Observed (baseline) | ||
n (%) | ||||||||||||| | ||||||||||||| |
Mean (SD) | ||||||||||||| | ||||||||||||| |
Median (range) | ||||||||||||| | ||||||||||||| |
Observed (6 months) | ||
n (%) | ||||||||||||| | ||||||||||||| |
Mean (SD) | ||||||||||||| | ||||||||||||| |
Median (range) | ||||||||||||| | ||||||||||||| |
Change from baseline (6 months) | ||
n (%) | ||||||||||||| | ||||||||||||| |
Mean (SD) | ||||||||||||| | ||||||||||||| |
Median (range) | ||||||||||||| | ||||||||||||| |
Observed (12 months) | ||
n (%) | ||||||||||||| | ||||||||||||| |
Mean (SD) | ||||||||||||| | ||||||||||||| |
Median (range) | ||||||||||||| | ||||||||||||| |
Change from baseline (12 months) | ||
n (%) | ||||||||||||| | ||||||||||||| |
Mean (SD) | ||||||||||||| | ||||||||||||| |
Median (range) | ||||||||||||| | ||||||||||||| |
IGF-1 = insulin-like growth factor I; SD = standard deviation; SDS = standard deviation score.
Source: Clinical Study Report for CP-4-006 (2020).10
Health-Related Quality of Life
The HRQoL was measured in CP-4-006 using the QoLISSY instrument. No MID was identified from literature for the QoLISSY instrument. The mean (SD) change from baseline in the total score of the QoLISSY tool was ||||||||||||| in the somatrogon treatment group and ||||||||||||| in the Genotropin treatment group. The results of the change from baseline in the 3 domains of the QoLISSY tool are presented in Table 20.
Table 20: Summary of QoLISSY Core Total Score — Full Analysis Set (CP-4-006)
QoLISSY | Somatrogon N = 109 | Genotropin N = 115 |
|---|---|---|
Total baseline | ||
n (%) | ||||||||||||| | ||||||||||||| |
Mean (SD) | ||||||||||||| | ||||||||||||| |
Median (range) | ||||||||||||| | ||||||||||||| |
Total observed (12 months) | ||
n (%) | ||||||||||||| | ||||||||||||| |
Mean (SD) | ||||||||||||| | ||||||||||||| |
Median (range) | ||||||||||||| | ||||||||||||| |
Total change from baseline (12 months) | ||
n (%) | ||||||||||||| | ||||||||||||| |
Mean (SD) | ||||||||||||| | ||||||||||||| |
Median (range) | ||||||||||||| | ||||||||||||| |
Physical baseline | ||
n (%) | ||||||||||||| | ||||||||||||| |
Mean (SD) | ||||||||||||| | ||||||||||||| |
Median (range) | ||||||||||||| | ||||||||||||| |
Physical observed (12 months) | ||
n (%) | ||||||||||||| | ||||||||||||| |
Mean (SD) | ||||||||||||| | ||||||||||||| |
Median (range) | ||||||||||||| | ||||||||||||| |
Physical change from baseline (12 months) | ||
n (%) | ||||||||||||| | ||||||||||||| |
Mean (SD) | ||||||||||||| | ||||||||||||| |
Median (range) | ||||||||||||| | ||||||||||||| |
Social baseline | ||
n (%) | ||||||||||||| | ||||||||||||| |
Mean (SD) | ||||||||||||| | ||||||||||||| |
Median (range) | ||||||||||||| | ||||||||||||| |
Social observed (12 months) | ||
n (%) | ||||||||||||| | ||||||||||||| |
Mean (SD) | ||||||||||||| | ||||||||||||| |
Median (range) | ||||||||||||| | ||||||||||||| |
Social change from baseline (12 months) | ||
n (%) | ||||||||||||| | ||||||||||||| |
Mean (SD) | ||||||||||||| | ||||||||||||| |
Median (range) | ||||||||||||| | ||||||||||||| |
Emotional baseline | ||
n (%) | ||||||||||||| | ||||||||||||| |
Mean (SD) | ||||||||||||| | ||||||||||||| |
Median (range) | ||||||||||||| | ||||||||||||| |
Emotional observed (12 months) | ||
n (%) | ||||||||||||| | ||||||||||||| |
Mean (SD) | ||||||||||||| | ||||||||||||| |
Median (range) | ||||||||||||| | ||||||||||||| |
Emotional change from baseline (12 months) | ||
n (%) | ||||||||||||| | ||||||||||||| |
Mean (SD) | ||||||||||||| | ||||||||||||| |
Median (range) | ||||||||||||| | ||||||||||||| |
QoLISSY = Quality of Life in Short Stature Youth; SD = standard deviation.
Note: Calculated by the sum of the means of the 3 dimensions (physical, social, and emotional) and divided by 3.
Source: Clinical Study Report for CP-4-006 (2020).10
Other Patient-Reported Outcome Responses
In CP-4-006, other PRO responses were measured only in the somatrogon treatment group. The PRO instruments used were the PAT and OAT. These analyses were performed only in the US. The somatrogon injections were evaluated by subgroups (age, sex, and race). For the PAT, in the group of those patients ranging in age from 3 years to less than 7 years, ||||||||% of the administered injections were successful. And ||||||||% of the administered injections in the group of patients older than 7 years of age were successful. As well, ||||||||% of injections in male patients were successfully administered as compared to ||||||||% of injections in female patients. Lastly, ||||||||% of the somatrogon injections were successfully administered in patients who were Asian as compared ||||||||% of injections in patients who were White.
For the OAT, the somatrogon injection was successfully administered in |||||||| of all patient subgroups.
Sensitivity Analysis
To account for missing data, several sensitivity analyses were conducted for the primary efficacy end point. The sensitivity analysis results were aligned with the main analyses and established noninferiority of somatrogon over Genotropin in CP-4-006 and comparability of somatrogon to Genotropin in CP-4-009.
Subgroups
Subgroup analyses conducted in both studies that were identified as being of interest in the CADTH review protocol focused on age (prepubertal versus pubertal children) and sex (male versus female). Results were reported for the primary efficacy outcome and the secondary outcomes of annual height velocity at 6 months and annual height velocity SDS at 6 months and 12 months.
Harms
Only those harms identified in the review protocol are reported as follows. See Table 21 for detailed harms data. Information about harms in the CP-4-009-OLE phase has been reported in the Other Relevant Evidence section of this report.
Adverse Events
In CP-4-006, 87.2% and 84.3% of patients in the somatrogon group and Genotropin group, respectively, reported at least 1 adverse event. The most commonly occurring adverse events were injection site pain (39.4% and 25.2% of patients in the somatrogon group and Genotropin group, respectively), nasopharyngitis (22.9% and 25.2% of patients in the somatrogon group and Genotropin group, respectively), pyrexia (16.5% and 13.9% of patients in the somatrogon group and Genotropin group, respectively), and headache (16.5% and 21.7% of patients in the somatrogon group and Genotropin group, respectively).
In CP-4-009, ||||||||% and ||||||||% of the patients in the somatrogon group and Genotropin group, respectively, reported at least 1 adverse event. The most commonly occurring adverse events were injection site pain (72.7% and 13.6% of patients in the somatrogon group and Genotropin group, respectively), nasopharyngitis (54.5% and 50.0% of patients in the somatrogon group and Genotropin group, respectively), pyrexia (18.2% and 13.6% of patients in the somatrogon group and Genotropin group, respectively), and headache (9.1% and 15.8% of patients in the somatrogon group and Genotropin group, respectively).
Serious Adverse Events
In CP-4-006, 2.8% and 1.7% of patients in the somatrogon group and Genotropin group, respectively, reported at least 1 serious adverse event. The most commonly occurring serious adverse events were under the system organ class of infections and infestation (2.8% and 0.9% of patients in the somatrogon group and Genotropin group, respectively). In the Genotropin treatment group, 0.9% of patients reported ureterolithiasis as a serious adverse event.
In CP-4-009, 9.1% of patients in both the somatrogon group and Genotropin group reported at least 1 serious adverse event. In the somatrogon treatment group, 4.5% of patients reported a serious adverse event under the system organ class of infections and infestations. As well, 4.5% of patients in the somatrogon treatment group reported hypoparathyroidism and febrile convulsion as a serious adverse event. In the Genotropin treatment group, 4.5% of patients reported craniopharyngioma and asthma as a serious adverse event.
Withdrawals Due to Adverse Events
In CP-4-006, 1 patient permanently withdrew from the study due to an injection site erythema and injection site induration in the somatrogon treatment group and no patients permanently discontinued from the study due to an adverse event in the Genotropin group.
In CP-4-009, only 1 patient withdrew from the study due to a craniopharyngioma, in the Genotropin treatment group.
Mortality
Neither study had any mortalities.
Notable Harms
The notable harms identified in the CADTH review protocol included the following: injection-related events, glucose intolerance and/or insulin resistance, excess IGF-I levels, malignancies, benign intracranial hypertension, slipped capital femoral epiphysis, scoliosis, tonsillar hypertrophy, carpal tunnel, and peripheral edema. In CP-4-006, of the identified notable harms, 43.1% of patients and 25.2% of patients in the somatrogon group and Genotropin group, respectively, reported experiencing an injection-related event. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. In CP-4-009, of the identified notable harms, 72.7% of patients in the somatrogon group reported experiencing an injection-related event. One patient in the Genotropin treatment group reported for malignancies under the system organ class of neoplasms benign, malignant, and unspecified (including cysts and polyps). ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 21: Summary of Harms — Safety Analysis Set
Harms | CP-4-006 | CP-4-009 | ||
|---|---|---|---|---|
Somatrogon N = 109 | Genotropin N = 115 | Somatrogon N = 22 | Genotropin N = 22 | |
Patients with ≥ 1 AE | ||||
n (%) | 95 (87.2) | 97 (84.3) | 22 (100.0) | |||||||| |
Most common AEs,a n (%) | ||||
Injection site pain, n | 43 (39.4) | 29 (25.2) | 16 (72.7) | 3 (13.6) |
Nasopharyngitis | 25 (22.9) | 29 (25.2) | 12 (54.5) | 11 (50.0) |
Pyrexia | 18 (16.5) | 16 (13.9) | 4 (18.2) | 3 (13.6) |
Headache | 18 (16.5) | 25 (21.7) | |||||||| | |||||||| |
Patients with ≥ 1 SAE | ||||
n (%) | 3 (2.8) | 2 (1.7) | 2 (9.1) | 2 (9.1) |
Most common SAEs, n (%) | ||||
Infections and infestations | 3 (2.8) | 1 (0.9) | 1 (4.5) | 0 |
Ureterolithiasis | 0 | 1 (0.9) | 0 | 0 |
Hypoparathyroidism | 0 | 0 | 1 (4.5) | 0 |
Craniopharyngioma | 0 | 0 | 0 | 1 (4.5) |
Febrile convulsion | 0 | 0 | 1 (4.5) | 0 |
Asthma | 0 | 0 | 0 | 1 (4.5) |
Patients who stopped treatment due to AEs | ||||
n (%) | 1 (0.9) | 0 | 0 | 1 (4.5) |
Injection site erythema and injection site induration, n (%) | 1 (0.9) | 0 | 0 | 0 |
Craniopharyngioma, n (%) | 0 | 0 | 0 | 1 (4.5) |
Deaths | ||||
n (%) | 0 | 0 | 0 | 0 |
Notable harms, n (%) | ||||
Injection-related events | 47 (43.1) | 29 (25.2) | 16 (72.7) | 3 (13.6) |
Glucose intolerance/insulin resistance | |||||||| | |||||||| | |||||||| | |||||||| |
Excess IGF-I levels | |||||||| | |||||||| | |||||||| | |||||||| |
Malignancies | |||||||| | |||||||| | |||||||| | |||||||| |
Benign intracranial hypertension | |||||||| | |||||||| | |||||||| | |||||||| |
Slipped capital femoral epiphysis | |||||||| | |||||||| | |||||||| | |||||||| |
Scoliosis | |||||||| | |||||||| | |||||||| | |||||||| |
Tonsillar hypertrophy | |||||||| | |||||||| | |||||||| | |||||||| |
Carpal tunnel | |||||||| | |||||||| | |||||||| | |||||||| |
Peripheral edema | |||||||| | |||||||| | |||||||| | |||||||| |
AE = adverse event; IGF-I = insulin-like growth factor I; SAE = serious adverse event.
Note: Main study phase only.
aReported for 2% or more of patients in any treatment group.
Source: Clinical Study Report for CP-4-006 (2020)10 and Clinical Study Report for CP-4-009 (2020).11
Critical Appraisal
Internal Validity
CP-4-006 and CP-4-009 were both open-label studies; however, the studies used an IWRS or IRT to randomize patients.
Randomization was stratified by age group, sex, peak GH levels, and region. In both studies, baseline characteristics were mostly well balanced, except for age, sex, and mean (SD) and median (range) height and weight in CP-4-009. There were low dropout rates in both studies, which limited selection bias. The clinical expert consulted by CADTH was of the opinion that an imbalance in age and sex in study CP-4-009 could influence the efficacy results in favour of somatrogon. The clinical expert noted that in CP-4-009, the difference in age would not impact outcomes observed within the first year, but it could impact the final adult height. The concomitant medications used in both studies were reasonable to expect from a Canadian clinical perspective.
The clinical expert consulted by CADTH expressed that the commonly used treatment for GHD in Canada is Humatrope. Both CP-4-006 and CP-4-009 had only Genotropin as the active comparator, despite other approved comparators being available for treatment in Canada. No direct or indirect evidence against GHD used in clinical practice within Canada was identified; hence, the comparative efficacy and safety of somatrogon against other regimens such as somatropin are unknown.
No justification for the comparability criteria was provided in study CP-4-009. While study CP-4-009 met the pre-specified criteria for comparability, this should not be confused with the unequivocal demonstration of equivalence, noninferiority, or superiority.
The chosen noninferiority margin of –1.8 cm per year or more was supported by historical studies,26,27 1 of which had a higher dosing of somatropin than that used in CP-4-006 and CP-4-009. In MacGillivray et al. (1996),26 the total weekly dose of somatropin was 0.3 mg/kg and in Wilton and Gunnarsson (1988),27 the doses ranged from 0.3 IU/kg per week to 0.8 IU/kg per week.
The primary outcomes of CP-4-006 and CP-4-009 looked to establish noninferiority and comparability of somatrogon with Genotropin, respectively. Once noninferiority was established, analyses of the secondary efficacy outcomes was conducted. However, these end points were not part of a hierarchical statistical testing plan and were not controlled for multiplicity, and no P values were reported.
CP-4-006 reported HRQoL and other PRO responses; however, these tools were administered only in selected locations with a very small sample size and as the study was open label, this could have biased the results of the efficacy outcomes in favour of somatrogon. Moreover, only a complete case analysis was carried out for this data, with different subsets of patients at each time point. As it is not a true full analysis set population, both outcomes would be subjected to an increased risk of bias due to the complete case analysis approach. No MID was identified from literature for the QoLISSY questionnaire or the OAT and PAT instruments.
The clinical expert consulted by CADTH stressed that adherence to treatment was of concern. Given the absence of patient input, CADTH is unable to determine the rate of adherence for somatrogon. Since the PAT and OAT were conducted only in the US with very small patient populations, the results can be viewed only as supportive evidence.
In CP-4-006, multiple sensitivity analyses were conducted to account for missing data for the primary efficacy outcome, and they were supportive of the primary efficacy outcome. In CP-4-009, a sensitivity analysis was performed using the PP population and was supportive for the primary efficacy outcome. However, the clinical expert consulted by CADTH was of the opinion that the sample size of CP-4-006 was very small, thereby limiting the interpretation of overall results.
The clinical expert consulted by CADTH was of the opinion that the safety results were satisfactory, and they were comfortable with the overall results. However, a concern regarding injection site reaction was flagged. Given that somatrogon was administered once weekly and Genotropin was administered daily, a higher number of injection-related adverse events in somatrogon could not be explained.
External Validity
The clinical expert noted that, based on baseline demographic and disease characteristics, the study population was fairly generalizable to Canadian patients; however, they were of the opinion that the age for inclusion in both studies (i.e., ≥ 3 years) was not reflective of Canadian practice, since patients with GHD would be identified and treated in infancy. They also noted that in CP-4-006, 20% of the cohort was Asian, which is a higher level than that seen in Canada. The clinical expert also commented on how the numbers of indigenous peoples who are treated in Canada are underestimated in both studies. The clinical expert also noted that the proportion of patients with a peak GH level of greater than 7 ng/dL was higher than what is observed in Canadian clinical practice. The dosing of Genotropin was aligned with Health Canada–approved dosing.
Most of the outcomes assessed in both studies were relevant to clinical practice and results were clinically meaningful. The experts were satisfied with the timing of the assessment of the primary and secondary efficacy end points. According to the clinical expert consulted for this review, the HRQoL outcomes are not typically used in clinical practice as far as routine management of prepubertal children with GHD is concerned.
The clinical expert consulted by CADTH was of the opinion that GH therapies are long-term therapies, and even though the primary end point of each of the 2 studies was met, in the absence of long-term comparative efficacy and safety results, interpreting the long-term clinical meaningfulness of somatrogon is limited.
Indirect Evidence
A focused literature search for network meta-analyses dealing with somatrogon and GHD was run in MEDLINE All (1946 − ) on June 25, 2021. No limits were applied.
No indirect evidence was submitted by the sponsor or identified in our literature search that would match the inclusion and exclusion criteria of this review.
Other Relevant Evidence
This section includes submitted long-term extension studies and additional relevant studies included in the sponsor’s submission to CADTH that were considered to address important gaps in the evidence included in the systematic review.
Patient Perception of Treatment Burden Study (C0311002)
Study C0311002 (study 002) was included as a study relevant to this review for the evaluation of the patient perception of somatrogon, which was not assessed in the studies included in the systematic review. Study 002 was a randomized, open-label, multi-centre, 2-period crossover study of children aged 3 years to less than 18 years with GHD. The primary objective of this study was to evaluate the treatment burden of a weekly injection of somatrogon and a daily injection of somatropin (Genotropin). Secondary objectives included an evaluation of patient and caregiver self-assessments of treatment experience, and an evaluation of the psychometric properties of the DCOA questionnaires. The latter is not reviewed in this summary.
Methods
The study included a 30-day screening period, a 24-week treatment period, and a follow-up phone call 4 weeks after the last clinic visit. Patients were required to be stable on treatment with daily somatropin for a minimum of 3 months before enrolment.
Key inclusion and exclusion criteria for study 002 has been presented in Table 22. To be included in study 002, patients were required to be from 3 years to less than 18 years of age with isolated GHD or GH insufficiency as part of multiple pituitary hormone deficiencies, to have an IGF-I SDS less than 2, and to be on treatment with 1 of the following formulations of somatropin for at least 3 months: Genotropin Pen, Genotropin Goquick Pen, Humatropen (US only), or Omnitrope Pen (US only). Patients were excluded from study 002 if they had a history of cancer, radiation or chemotherapy, or diabetes mellitus. Patients were also excluded if they had psychosocial dwarfism, were born SGA, had other causes of short stature, had chromosomal abnormalities, had a history of exposure to long-acting hGH preparation, had treatment with regularly scheduled medications other than the somatropin formulations listed in the inclusion criteria, or had closed epiphyses.
Table 22: Study 002 Key Inclusion and Exclusion Criteria
Inclusion criteria | Exclusion criteria |
|---|---|
|
|
GH = growth hormone; GHD = growth hormone deficiency; hGH = human growth hormone; ICF = informed consent form; IGF-I = insulin-like growth factor I; SDS = standard deviation score.
Source: Study 002 Clinical Study Report (2020).28
Patients were randomized in a 1:1 ratio to 1 of 2 sequences as illustrated in Figure 2. Randomization was stratified by region (the US or EU) and the type of Genotropin injection device used (Genotropin Pen or Genotropin Goquick). Patients randomized to sequence 1 received treatment with once-daily somatropin for 12 weeks followed by 12 weeks of treatment with once-weekly somatrogon. Patients randomized to sequence 2 received treatment with once-weekly somatrogon for 12 weeks followed by 12 weeks of treatment with once-daily somatropin.
Study visits occurred at baseline and at week 6, week 12, week 18, and week 24, with a follow-up phone call at week 28. Patients and caregivers were asked to complete the DCOA questionnaires at the end of each 12-week treatment period (DCOA 1 at week 12 and DCOA 2 at week 24). The DCOA questionnaires are described in the outcomes section below.
Figure 2: Study Design for Study 002
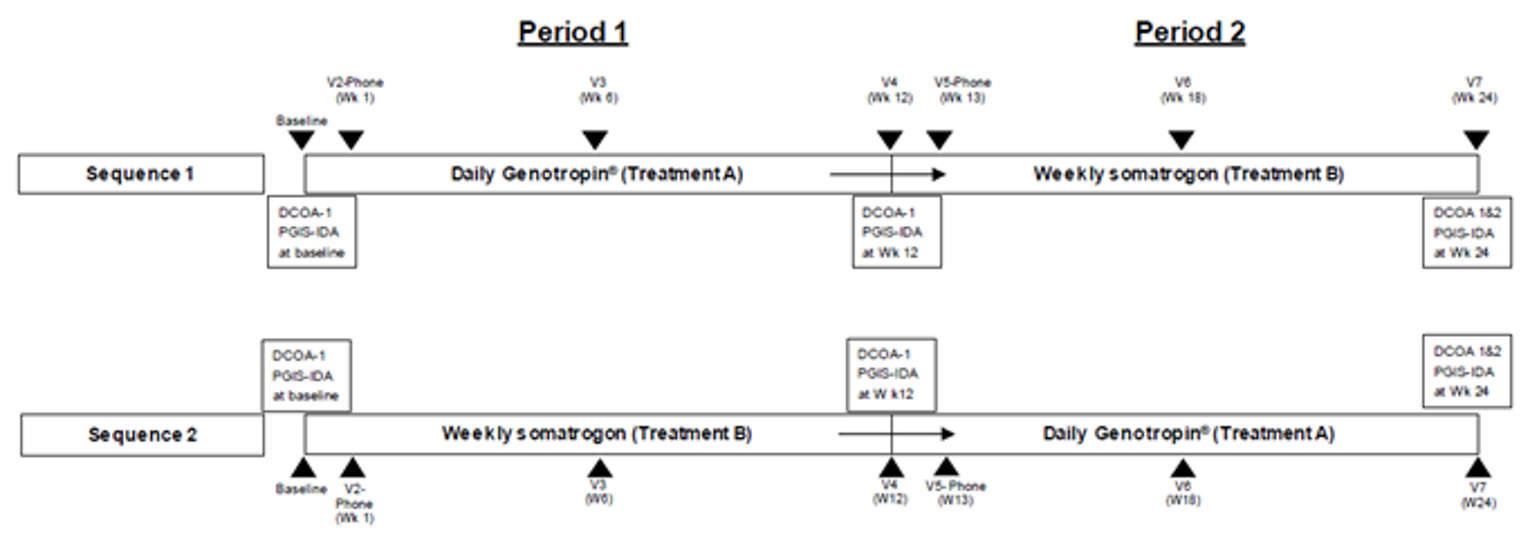
DCOA-1 = Dyad Clinical Outcome Assessment 1; DCOA-2 = Dyad Clinical Outcome Assessment 2; PGIS-IDA = Patient Global Impression–Severity: Impact on Daily Activities; v = visit; w = week; wk = week.
Source: Study 002 Clinical Study Report (2020).28
Populations
A summary of baseline characteristics for patients included in study 002 is provided in Table 23. Overall, the mean age of patients was 10.7 years (SD = 3.5 years). The majority of patients were male (82.8%) and White (93.1%), |||||||||||||||||||||||||||||||| lived in ||||. The mean height and weight of patients was |||||| cm (SD = |||||| cm) and |||||| kg (SD = |||||| kg), respectively. Most patients were using |||||||||||| before study start (|||||| using the Genotropin Pen and |||||| were using the Genotropin Goquick Pen). The remainder had been using |||||||||||||||||| or ||||||||||||||||||||||||.
In general, the treatment groups were well balanced by the baseline characteristics presented. Differences to note include the proportion of patients who were male (sequence 1 = 79.1% versus sequence 2 = 86.4%), and those with prior experience with the |||||||||||||||||||||||| (sequence 1 = |||||| versus sequence 2 = ||||||) and |||||||||||| (sequence 1 = |||||| versus sequence 2 = ||||||).
Table 23: Baseline Characteristics of Patients Enrolled in Study 002
Characteristics | Genotropin, then somatrogon N = 43 | Somatrogon, then Genotropin N = 44 | Total N = 87 |
|---|---|---|---|
Age (years) | |||
< 8, n (%) | 8 (18.6) | 10 (22.7) | 18 (20.7) |
8 to < 18, n (%) | 35 (81.4) | 34 (77.3) | 69 (79.3) |
Mean (SD) | 10.8 (3.4) | 10.7 (3.7) | 10.7 (3.5) |
Median (range) | 12 (4 to 16) | 11 (3 to 17) | 11 (3 to 17) |
Sex, n (%) | |||
Male | 34 (79.1) | 38 (86.4) | 72 (82.8) |
Female | 9 (20.9) | 6 (13.6) | 15 (17.2) |
Race, n (%) | |||
Asian | 0 | 1 (2.3) | 1 (1.1) |
Black or African-American | 3 (7.0) | 1 (2.3) | 4 (4.6) |
White | 39 (90.7) | 42 (95.5) | 81 (93.1) |
Not reported | 1 (2.3) | 0 | 1 (1.1) |
Ethnicity, n (%) | |||
Hispanic or Latino | 3 (7.0) | 2 (4.5) | 5 (5.7) |
Non-Hispanic or non-Latino | 39 (90.7) | 42 (95.5) | 81 (93.1) |
Not reported | 1 (2.3) | 0 | 1 (1.1) |
Region, n (%) | |||
US | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
Europe (Bulgaria, Czech Republic, Slovakia, UK) | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
Country, n (%) | |||
Bulgaria | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
Czech Republic | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
Slovakia | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
UK | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
US | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
Height (cm) | |||
Mean (SD) | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
Median (range) | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
Weight (kg) | |||
Mean (SD) | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
Median (range) | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
BMI (kg/m2) | |||
Mean (SD) | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
Median (range) | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
Type of growth hormone injection pen used before study start, n (%) | |||
Genotropin Goquick Pen | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
Genotropin Pen | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
Humatropen | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
Omnitrope Pen | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||||||| |
BMI = body mass index; SD = standard deviation.
Source: Study 002 Clinical Study Report (2020).28
Interventions
The interventions used in study 002 were somatrogon and Genotropin. More specifically, somatrogon 60 mg/1.2 mL solution for injection was available as a multi-dose disposable pre-filled pen for SC self-injection. Somatrogon was administered once weekly at approximately the same time on a regularly scheduled day of the week.
Commercially available Genotropin was the comparator in study 002. The product varied by region. For patients in the US, Genotropin was available as a 5 mg dose or 12 mg dose 2-chamber cartridge administered using the corresponding Genotropin Pen 5 and Genotropin Pen 12 delivery devices. In the EU region, Genotropin was available as a 5.3 mg dose or 12 mg dose 2-chamber cartridge administered using the Genotropin Pen 5.3 and Genotropin Pen 12 delivery devices. These doses were also available for administration using the Genotropin Goquick pre-filled pen. Genotropin was administered daily by SC administration at the same time of day as they were injecting their daily GH at the time of screening.
Interventions were administered at the clinical site at the start of 2 treatment periods, then self-administered at home for the rest of the study. There were no washout periods as the treatments were required to be taken continually.
Outcomes
Dyad Clinical Outcome Assessment and Patient Life Interference Questionnaire
The DCOA was designed to measure the experience of patients taking rhGH GHD injections. The intention of the DCOA is to demonstrate significant benefit of a weekly injection compared to a daily injection in terms of adherence and acceptance. As this study assessed GH injections in children, the patient and caregiver answer the questions together (a dyadic approach). The DCOA comprises 2 main aspects — an assessment of treatment burden and preference. Treatment burden includes concepts of patient life interference, injection signs and symptoms, pen ease of use, ease and convenience of injection schedule, and caregiver life interference. Preference includes concepts of missed injection, patient satisfaction, and willingness to continue. The 9 items of the DCOA 1 questionnaire and corresponding number of questions and evaluation scale are summarized as follows:
pen ease of use (5 questions, 5-point scale each)
ease of the injection schedule (1 question, 5-point scale)
convenience of the injection schedule (1 question, 7-point scale)
satisfaction with overall treatment experience (1 question, 5-point scale)
willingness to continue injection schedule (1 question, 5-point scale)
injection signs and symptoms (from patients aged 8 years and older) (4 questions, 0 to 10 scale each)
assessment of signs (from caregivers for children aged < 8 years) (2 questions, 0 to 10 scale each)
caregiver life interference, including family life interference (13 questions, 5-point scale each)
missed injections (1 question, enter number of daily administrations missed (0 to 31), or number of weekly administrations missed (0 to 5)).
Treatment burden was assessed as the difference in mean overall life interference total scores between the weekly injection schedule and daily injection schedule as determined by the patient life interference questionnaire (which is part of DCOA 1) completed by the patient/caregiver dyad at baseline and after each treatment schedule experience. The patient life interference questionnaire outcome consisted of 7 questions, including 5 questions related to life interference (with daily activities, social activities, recreation/leisure activities, spending the night away from home, and with travel), 1 question about life interference due to changes to life routine, and 1 question about life interference due to the bother of GH injections. Questions were answered using a 5-point scale that ranged from “never” (1) to “always” (5). A total score was reported, which ranged from 7 to 35, where a lower score for life interference was considered to be a better outcome.
The DCOA 2 questionnaire was completed at week 24 and evaluated the proportion of patient/caregiver dyads that selected the weekly injection schedule compared to the daily injection schedule. The DCOA 2 questionnaire was assessed by the following domains and corresponding number of questions:
choice of injection pen (1 question)
preferred injection schedule (1 question)
convenience of injection schedule (1 question)
easier to follow (1 question)
pen ease of use (4 questions)
patient life interference (6 questions)
caregiver life interference, including family life interference (11 questions)
benefit relating to the injection schedule (1 question)
intention to comply (4 questions).
A recently published field study demonstrated evidence of validity and reliability of the DCOA.29 Internal consistency reliability demonstrated for all domains (Cronbach’s alpha ≥ 0.70), with the exception of the injection signs reported by caregiver domain (alpha = 0.653) and the satisfaction and willingness to continue domain (alpha = 0.589). Test-retest reliability was assessed and demonstrated across domains based on an intraclass correlation coefficient (ICC) of 0.70 or more. Some evidence of construct validity was reported in the overall sample (including children, adolescents, and adults). Concurrent validity was not demonstrated in the subgroup for children. Evidence of responsiveness or an MID was not identified. Additional information about this outcome is available in Appendix 3.
Patient Global Impression–Severity: Impact on Daily Activities
The Patient Global Impression–Severity: Impact on Daily Activities (PGI-S: IDA) was based on a single question, ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. Raw scores were transformed onto a scale from 0 to 100, where lower scores represent less impact on daily activities.
Statistical Analysis
The primary end point in study 002 was treatment burden, measured as the difference between weekly injection schedule and the daily injection schedule based on the mean overall life interference total scores.28 The primary end point was analyzed using a linear mixed-effects model. Sequence, period, and treatment were included as fixed effects, and subject within sequence and within-subject error were included as random effects in the model.
Treatment experience using the DCOA questionnaires and the PGI-S: IDA questionnaire were secondary end points in study 002. Treatment experience via the DCOA 1 questionnaire was assessed using the difference in mean scores between the experience with weekly injections and daily injections, based on each variable of the DCOA 1 questionnaire. Treatment experience via the DCOA 2 questionnaire was assessed based on the proportion of patient/caregiver dyads that selected the weekly injection schedule compared to the daily injection schedule. Lastly, the PGI-S: IDA was reported at baseline and at the end of week 12 and week 24.28
Patient Disposition
A total of 87 patients were included in this study, with 43 patients randomized to sequence 1 (Genotropin first) and 44 patients randomized to sequence 2 (somatrogon first). Overall, 2 patients randomized to sequence 2 discontinued from the study. One patient discontinued due to adverse events while receiving somatrogon and 1 patient discontinued due to a protocol violation while receiving Genotropin.
Exposure to Study Treatments
The duration of treatment was a mean of ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.
Efficacy
The mean (SD) of the total scores for the overall life interference questionnaire was a score of |||||||||||||||||||||||||||||| during treatment with Genotropin and ||||||||||||||||||||||| during treatment with somatrogon. The reported difference in overall scores was –15.49 (95% CI, –19.71 to –11.27; P < 0.001) in favour of treatment with somatrogon, indicating a lower treatment burden during treatment with somatrogon.
Table 24: Overall Life Interference Total Scores — Full Analysis Set
Outcome measure | During Genotropin treatment N = 86 | During somatrogon treatment N = 87 |
|---|---|---|
N | 85 | 82 |
Mean (SD) | |||||||||||||||||| | |||||||||||||||||| |
Median (range) | |||||||||||||||||| | |||||||||||||||||| |
Model-based mean (95% CI)a | 24.1 (20.6 to 27.7) | 8.6 (5.1 to 12.2) |
Somatrogon vs. Genotropin | ||
Difference in overall scores (95% CI)a | –15.5 (–19.7 to –11.3) | — |
P value | < 0.0001 | — |
CI = confidence interval; SD = standard deviation; vs. = versus.
aResults based on a linear mixed-effects model including sequence, period, and treatment as fixed effects, and subject within sequence and within-subject error as random effects.
Source: Study 002 Clinical Study Report (2020).28
The results of the DCOA 1 questionnaire presented as total scores for each domain are summarized in Table 25. All of the domains of the DCOA 1 questionnaire were associated with numerically greater overall scores during treatment with Genotropin than during treatment with somatrogon, with 2 exceptions: the injection signs and symptoms domain (from patients aged 8 years and older) and the assessment of signs domain (from caregivers for children < 8 years old). The reported overall score for these 2 domains did not suggest a preference for either treatment based on the reported overall scores.
Table 25: DCOA 1 Questionnaire, Total Scoresa — Full Analysis Set
Question | Total, N | Overall score (for drug received during any period), Mean (SD) | Treatment group difference (somatrogon vs. Genotropin) | |
|---|---|---|---|---|
Mean difference (95% CI) | P valueb | |||
Pen ease of use | ||||
During Genotropin treatment | ||||||||||| | ||||||||||| | |||||||||||||||||||||| | ||||||||||| |
During somatrogon treatment | ||||||||||| | ||||||||||| | |||||||||||||||||||||| | — |
Ease of the injection schedule | ||||
During Genotropin treatment | ||||||||||| | ||||||||||| | |||||||||||||||||||||| | ||||||||||| |
During somatrogon treatment | ||||||||||| | ||||||||||| | — | — |
Convenience of the injection schedule | ||||
During Genotropin treatment | ||||||||||| | ||||||||||| | |||||||||||||||||||||| | ||||||||||| |
During somatrogon treatment | ||||||||||| | ||||||||||| | — | — |
Satisfaction with overall treatment experience | ||||
During Genotropin treatment | ||||||||||| | ||||||||||| | |||||||||||||||||||||| | ||||||||||| |
During somatrogon treatment | ||||||||||| | ||||||||||| | — | — |
Willingness to continue the injection schedule | ||||
During Genotropin treatment | ||||||||||| | ||||||||||| | |||||||||||||||||||||| | ||||||||||| |
During somatrogon treatment | ||||||||||| | ||||||||||| | — | — |
Injection signs and symptoms (from patients ≥ 8 years) | ||||
During Genotropin treatment | ||||||||||| | ||||||||||| | |||||||||||||||||||||| | ||||||||||| |
During somatrogon treatment | ||||||||||| | ||||||||||| | — | — |
Assessment of signs (from caregivers for children < 8 years old) | ||||
During Genotropin treatment | ||||||||||| | ||||||||||| | |||||||||||||||||||||| | ||||||||||| |
During somatrogon treatment | ||||||||||| | ||||||||||| | — | — |
Caregiver life interference, including family life interference | ||||
During Genotropin treatment | ||||||||||| | ||||||||||| | |||||||||||||||||||||| | ||||||||||| |
During somatrogon treatment | ||||||||||| | ||||||||||| | — | — |
Missed injections | ||||
During Genotropin treatment | ||||||||||| | ||||||||||| | |||||||||||||||||||||| | ||||||||||| |
During somatrogon treatment | ||||||||||| | ||||||||||| | — | — |
CI = confidence interval; DCOA 1 = Dyad Clinical Outcome Assessment 1; SD = standard deviation; vs. = versus.
aResults based on a linear mixed-effects model including sequence, period, and treatment as fixed effects, and subject within sequence and within-subject error as random effects.
bThe P value has not been adjusted for multiple testing (i.e., the type I error rate has not been controlled).
Source: Study 002 Clinical Study Report (2020).28
The results of the DCOA 2 questionnaire are summarized in Table 26. Overall, the proportion of patients who responded to the questionnaire indicating preference for somatrogon was greater than the proportion of patients indicating preference for Genotropin. Of note, between 27.4% and 64.3% of patients indicated no preference in response to the 4 items included in the domain addressing the pen ease of use. Further, the proportion of patients who preferred somatrogon and Genotropin in terms of injecting the medicine was 36.9% and 33.3%, respectively.
Table 26: DCOA 2 Questionnaire — Full Analysis Set
Question | Total, N | Preferred treatment | ||
|---|---|---|---|---|
Somatrogon, n (%) | Genotropin, n (%) | No preference, n (%) | ||
Choice of injection pen: Which pen would you choose? | 84 | 74 (88.1) | 10 (11.9) | 0 |
Preferred injection schedule: Which injection schedule do you prefer overall? | 84 | 77 (91.7) | 6 (7.1) | 1 (1.2) |
Convenience of injection schedule: Which injection schedule was more convenient overall? | 84 | 80 (95.2) | 4 (4.8) | 0 |
Easier to follow: Which injection schedule was easier to follow overall? | 84 | 72 (85.7) | 8 (9.5) | 4 (4.8) |
Pen ease of use: Which pen is easier to use? (4 questions) | ||||
Preparing the injection | 84 | 54 (64.3) | 7 (8.3) | 23 (27.4) |
Setting the dose | 84 | 38 (45.2) | 14 (16.7) | 32 (38.1) |
Injecting the medicine | 84 | 31 (36.9) | 28 (33.3) | 25 (29.8) |
Storing the pen | 84 | 26 (31.0) | 4 (4.8) | 54 (64.3) |
Patient life interference: Which injection schedule interfered less? (5 questions) | ||||
With patient’s daily activities | 84 | 66 (78.6) | 3 (3.6) | 15 (17.9) |
With patient’s social activities | 84 | 68 (81.0) | 2 (2.4) | 14 (16.7) |
With patient’s recreation/leisure | 84 | 67 (79.8) | 3 (3.6) | 14 (16.7) |
With patient’s night away from home | 84 | 73 (86.9) | 3 (3.6) | 8 (9.5) |
With patient’s travel | 84 | 70 (83.3) | 3 (3.6) | 11 (13.1) |
Caregiver life interference: Which injection schedule interfered less? (5 questions) | ||||
With caregiver’s daily activities | 84 | 67 (79.8) | 2 (2.4) | 15 (17.9) |
With caregiver’s social activities | 84 | 68 (81.0) | 2 (2.4) | 14 (16.7) |
With caregiver’s recreation/leisure | 84 | 69 (82.1) | 2 (2.4) | 13 (15.5) |
With caregiver’s night away from home | 84 | 72 (85.7) | 1 (1.2) | 11 (13.1) |
With caregiver’s travel | 84 | 72 (85.7) | 2 (2.4) | 10 (11.9) |
Family life interference: Which injection schedule interfered less? (5 questions) | ||||
With family daily activities | 84 | 61 (72.6) | 1 (1.2) | 22 (26.2) |
With family social activities | 84 | 62 (73.8) | 1 (1.2) | 21 (25.0) |
With family recreation/leisure | 84 | 64 (76.2) | 1 (1.2) | 19 (22.6) |
With family night away from home | 84 | 65 (77.4) | 1 (1.2) | 18 (21.4) |
With family travel | 84 | 67 (79.8) | 1 (1.2) | 16 (19.0) |
Intention to comply: Which schedule would be more likely to be followed? | 84 | 57 (67.9) | 5 (6.0) | 22 (26.2) |
DCOA 2 = Dyad Clinical Outcome Assessment 2.
Source: Study 002 Clinical Study Report (2020).28
As presented in Table 27, the overall mean PGI-S: IDA score was 20.6 (SD = 19.9) for patients while being treated with Genotropin and 5.9 (SD = 8.8) for patients while being treated with somatrogon. The between-groups difference was –14.6 (95% CI, –18.7 to –10.4; P < 0.0001).
Table 27: Patient Global Impression and Impact on Daily Activities — Full Analysis Set
Outcome | During Genotropin treatment N = 86 | During somatrogon treatment N = 87 |
|---|---|---|
Overall score | ||
N | 85 | 82 |
Mean (SD) | ||||||||||| | ||||||||||| |
Median (range) | ||||||||||| | ||||||||||| |
Model-based mean (95% CI)a | 20.6 (17.3 to 24.0) | 6.1 (2.7 to 9.5) |
Somatrogon vs. Genotropin | ||
Difference in overall scores (95% CI)a | –14.6 (–18.7 to –10.4) | — |
P valueb | < 0.0001 | — |
CI = confidence interval; SD = standard deviation; vs. = versus.
aResults based on a linear mixed-effects model including sequence, period, and treatment as fixed effects, and subject within sequence and within-subject error as random effects.
bThe P value has not been adjusted for multiple testing (i.e., the type I error rate has not been controlled).
Source: Study 002 Clinical Study Report (2020).28
Harms
A summary of harms reported during study 002 is presented in Table 28. Adverse events were reported by 44.2% of patients during treatment with Genotropin and 54.0% of patients during treatment with somatrogon. The most frequently reported adverse event was injection site pain, followed by injection site hematoma, nasopharyngitis, and headache. No serious adverse events or deaths were reported during study 002. One patient stopped treatment due to adverse events, which occurred during treatment with somatrogon as a result of injection site pain. With regard to notable harms for this review, injection-related events were reported by ||||||||||||||||||| and |||||||||| of patients during Genotropin treatment and during somatrogon treatment, respectively. Additionally, |||||||||||||||||||||| reported excess IGF-I levels during somatrogon treatment.
Outcome | During Genotropin treatment N = 86 | During somatrogon treatment N = 87 |
|---|---|---|
Patients with ≥ 1 AEa | ||
n (%) | 38 (44.2) | 47 (54.0) |
Nasopharyngitis | 5 (5.8) | 6 (6.9) |
Injection site pain | 11 (12.8) | 13 (14.9) |
Injection site hematoma | 8 (9.3) | 4 (4.6) |
Headache | 5 (5.8) | 6 (6.9) |
Patients with ≥ 1 SAE | ||
n (%) | 0 | 0 |
Patients who stopped treatment due to AEs | ||
n (%) | 0 | 1 (1.1) |
Injection site pain | 0 | 1 (1.1) |
Deaths | ||
n (%) | 0 | 0 |
Notable harms, n (%) | ||
Injection-related eventsb | 27 (31.4) | 25 (28.7) |
Glucose intolerance/insulin resistance | ||||||||||| | ||||||||||| |
Excess IGF-I levels | ||||||||||| | ||||||||||| |
Malignancies | ||||||||||| | ||||||||||| |
Benign intracranial hypertension | ||||||||||| | ||||||||||| |
Slipped capital femoral epiphysis | ||||||||||| | ||||||||||| |
Scoliosis | ||||||||||| | ||||||||||| |
Tonsillar hypertrophy | ||||||||||| | ||||||||||| |
Carpal tunnel | ||||||||||| | ||||||||||| |
Peripheral edema | ||||||||||| | ||||||||||| |
AD = adverse event; IGF-I = insulin-like growth factor I; SAE = serious adverse event.
a5% or more.
bIncludes injection site pain, injection site hematoma, injection site swelling, injection site bruising, administration site pain, injection site erythema, injection site hemorrhage, administration site edema, application site pruritus, and injection site reaction.
Source: Study 002 Clinical Study Report (2020).28
Critical Appraisal
Internal Validity
Study 002 was conducted to evaluate the treatment experience and patient preference for treatment with once-weekly somatrogon compared to once-daily Genotropin. This was evaluated using subjective PROs within an open-label study design, which has potential for significant bias in the results. Further, the primary and secondary end points were evaluated using outcomes derived from the DCOA questionnaires, which were developed by the sponsor and underwent an evaluation of the psychometric properties of the tool as part of study 002. Evidence of reliability was demonstrated; however, there was no evidence of validity or responsiveness. An MID was not identified from the literature. Additionally, results for the DCOA 1 questionnaire included P values, but the statistical tests were not controlled for multiplicity and, consequently, were at risk of type I error. The other secondary outcomes were reported descriptively. Both of these factors and the lack of an established MID make it difficult to determine the clinical meaningfulness of the results. Lastly, the assessment of patient preference and treatment burden following 12 weeks of treatment with somatrogon is likely to overestimate the results compared to what is expected with long-term treatment in clinical practice as a result of diminishing excitement or expectations for a new treatment option over time.
External Validity
Study 002 did not include any patients living in Canada and the majority of included patients were White. This demographic does not reflect the diversity of patients seen in clinical practice in Canada. Study 002 also excluded patients younger than 3 years of age, which was noted as a generalizability issue for an important population living with GHD (those younger than 3 years old), per feedback from the clinical expert on this review. Lastly, the preference and treatment burden of somatrogon was assessed based on a period of 12 weeks, which does not reflect use in clinical practice where patients require long-term treatment with GH therapy.
CP-4-009 – Long-Term Open-Label Extension Study
The CP-4-009 LT-OLE evaluated the long-term efficacy and safety of somatrogon in a single-arm trial.
Methods
Eligible patients treated with Genotropin who completed 12 months of treatment during the CP-4-009 main study were switched to a somatrogon dose of 0.66 mg/kg per week and somatrogon-treated patients who completed 12 months of treatment during the main study continued to receive somatrogon with the same mg/kg per week dose in the OLE phase. The OLE phase would continue until the marketing registration of somatrogon in Japan.
Populations
Pediatric patients with GHD who completed the main study (12 months of treatment) with adequate compliance and adherence to the visit schedule and without major protocol deviation according to the main study protocol were eligible to enter the OLE phase.
Interventions
Patients who had received Genotropin for 12 months during the main study were switched to receive a dose of 0.66 mg/kg per week of somatrogon in the OLE phase (no less than 1 day after cessation of the Genotropin treatment).
Outcomes
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Patient Disposition
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 29: Extent of Exposure, Safety Analysis Set — CP-4-009-OLE
Duration of treatment (days) | Somatrogon N = 22 | Genotropin to somatrogona N = 19 |
|---|---|---|
n (%) | ||||||||||| | ||||||||||| |
Mean (SD) | ||||||||||| | ||||||||||| |
Median (range) | ||||||||||| | ||||||||||| |
Dose reduction due to IGF-I SDS > 2, n (%) | ||||||||||| | ||||||||||| |
IGF-I = insulin-like growth factor I; SD = standard deviation; SDS = standard deviation score.
aGenotropin, then switched to somatrogon.
Source: Clinical Study Report for CP-4-009 (2020).11
Efficacy
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Harms
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 30: Summary of Harms — CP-4-009-OLE
Harms | Somatrogon N = 22 | Genotropin N = 19 |
|---|---|---|
Patients with ≥ 1 AE | ||
n (%) | ||||||||||| | ||||||||||| |
Most common AEs, n (%) | ||
Injection site pain | ||||||||||| | ||||||||||| |
Nasopharyngitis | ||||||||||| | ||||||||||| |
Pyrexia | ||||||||||| | ||||||||||| |
Headache | ||||||||||| | ||||||||||| |
Patients with ≥ 1 SAE | ||
n (%) | ||||||||||| | ||||||||||| |
Most common SAEs, n (%) | ||||||||||| | ||||||||||| |
Infections and infestations | ||||||||||| | ||||||||||| |
Upper respiratory tract inflammation | ||||||||||| | ||||||||||| |
Patients who stopped treatment due to AEs | ||
n (%) | ||||||||||| | ||||||||||| |
Deaths | ||
n (%) | ||||||||||| | ||||||||||| |
Notable harms, n (%) | ||
Injection related events | ||||||||||| | ||||||||||| |
Glucose intolerance/insulin resistance | ||||||||||| | ||||||||||| |
Excess IGF-I levels | ||||||||||| | ||||||||||| |
Malignancies | ||||||||||| | ||||||||||| |
Benign intracranial hypertension | ||||||||||| | ||||||||||| |
Slipped capital femoral epiphysis | ||||||||||| | ||||||||||| |
Scoliosis | ||||||||||| | ||||||||||| |
Tonsillar hypertrophy | ||||||||||| | ||||||||||| |
Carpal tunnel | ||||||||||| | ||||||||||| |
Peripheral edema | ||||||||||| | ||||||||||| |
AE = adverse event; SAE = serious adverse event.
Source: Clinical Study Report for CP-4-009 (2020).11
Critical Appraisal
Internal Validity
CP-4-009-OLE was conducted to evaluate the long-term efficacy and safety of once-weekly somatrogon. Patients who had completed the main study of CP-4-009 and were eligible for the long-term open-label phase, and had received daily Genotropin in the main study, switched over to once-weekly somatrogon. The efficacy results of the OLE phase are selectively reported. The analyses are not part of a statistical testing plan and, hence, the effect of somatrogon at the data cut-off date of March 13, 2020, is considered uncertain. No efficacy analyses for HRQoL or other PRO measures were conducted; hence, the long-term effect of somatrogon on HRQoL is unknown.
External Validity
CP-4-009-OLE was conducted exclusively in Japanese prepubertal children and did not include any Canadian patients. This is not reflective of Canadian clinical practice. Hence, CP-4-009-OLE has noted generalizability issues. The CP-4-009-OLE study excluded patients younger than 3 years of age; this also leads to a Canadian generalizability issue, as the clinical expert consulted by CADTH stated that in clinical practice, patients with GHD are seen as early as in their infancy.
Discussion
Summary of Available Evidence
Two phase III studies were included in the systematic review. The CP-4-006 and CP-4-009 studies included prepubertal children with GHD.
CP-4-006 was an open-label, multi-centre, randomized, active-controlled, parallel group study evaluating the efficacy and safety of weekly somatrogon to daily GH (Genotropin). Patients were randomized in a 1:1 method to receive weekly SC doses of somatrogon or daily SC doses of Genotropin for 12 months. Following the completion of the 12-month treatment period, in both studies, eligible patients were enrolled in a single-arm OLE treatment phase with somatrogon. C0311002 was a randomized, open-label, multi-centre, 2-period crossover study.
The primary objective of CP-4-006 was to establish noninferiority of somatrogon compared to Genotropin. The primary objective of CP-4-009 was to establish comparability of somatrogon to Genotropin. The secondary objectives of both studies were annualized height velocity at 6 months, a change in height SDS at 6 months and 12 months, and a change in bone maturation at 12 months. The primary objective of C0311002 was to evaluate the treatment burden of a weekly injection of somatrogon and a daily injection of somatropin (Genotropin). Secondary objectives included an evaluation of patient and caregiver self-assessments of treatment experience and an evaluation of the psychometric properties of the DCOA questionnaires.
CP-4-006 and CP-4-009 used appropriate randomization techniques and the primary and secondary end points were considered clinically meaningful. Patient dropout was low in both studies and missing data were accounted for appropriately. The main limitations were a lack of multiplicity controls for all end points, a lack of rationale for a noninferiority margin, concerns with age of inclusion of patients in the study, a lack of interpretation of HRQoL outcomes and no reported MID. C0311002 had concerns of generalizability to the Canadian patient population and lacked an appropriate assessment period.
In addition to CP-4-006 and CP-4-009, evidence from C0311002 — which included prepubertal and pubertal children with GHD and assessed the treatment burden of a weekly injection of somatrogon and a daily injection of somatropin (Genotropin) — was also included in the Other Relevant Evidence section of this report. In addition, the OLE phase of CP-4-009 was also included in the Other Relevant Evidence section.
Interpretation of Results
Efficacy
In CP-4-006 and CP-4-009, the primary end point was to establish the noninferiority of once-weekly somatrogon as compared to daily Genotropin. Noninferiority was concluded if the lower bound of the 2-sided 95% CI for the mean treatment difference between somatrogon and Genotropin in the primary efficacy end point was –1.8 cm per year or more. The studies statistically demonstrated that somatrogon was not inferior to Genotropin (CP-4-006) and was comparable to Genotropin (CP-4-009). |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. In CP-4-009, the CI of the primary efficacy outcome did not cross the null; however, due to the small sample size and imbalance in baseline characteristics, the true treatment effect cannot be established with certainty.
In CP-4-006 and CP-4-009, the secondary efficacy outcomes were annualized height velocity at 6 months, a change in height SDS at 6 months and 12 months, and a change in bone maturation at 12 months. In annualized height velocity at 6 months and a change in height SDS at 6 months and 12 months, the between–treatment group difference was higher for somatrogon. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. P values for these analyses were not reported; hence, the statistical significance of these results in favour of 1 treatment cannot be interpreted.
In CP-4-006, other efficacy outcomes that were described using descriptive statistics were serum biomarker levels (IGF-I and IGF-I SDS), HRQoL, and other PRO responses. CP-4-009 did not report on any of these outcomes. HRQoL and other PRO responses had no MID identified in the literature. All the analyses were done in observed case patients and there were substantial amounts of missing data, which would introduce significant biases. The analyses for HRQoL and other PRO responses (only in the somatrogon treatment group) were performed in selected locations only. Hence, the effects of these assessments in support of somatrogon are highly uncertain.
Results from the long-term open-label phase for CP-4-006 were not available at the time of writing this report. In the absence of the long-term open-label phase results, it is uncertain whether the response of somatrogon was sustained. For CP-4-009, the sample size of the population in the long term open-label phase is very small to make a conclusive assessment of the long-term response of somatrogon.
In C0311002, evidence of the reliability of the DCOA 1 questionnaire was demonstrated; however, evidence of validity was inconsistent and there was no evidence of responsiveness or an MID. Additionally, results for the DCOA 1 questionnaire included P values, but the statistical tests were not controlled for multiplicity and, consequently, were at risk of type I error. The other secondary outcomes were reported descriptively. Both of these factors and the lack of an established MID make it difficult to determine the clinical meaningfulness of the results. Lastly, the assessment of patient preference and treatment burden following 12 weeks of treatment with somatrogon is likely to overestimate the results compared to what is expected with long-term treatment in clinical practice.
Harms
In CP-4-006, 87.2% and 84.3% of the patients in the somatrogon group and Genotropin group, respectively, and in CP-4-009, ||||||||% and ||||||||% of the patients in the somatrogon group and Genotropin group, respectively, reported at least 1 adverse event. The most commonly occurring adverse events were injection site pain, nasopharyngitis, pyrexia, and headache. No deaths were reported in any of the studies. The clinical expert consulted by CADTH was impressed with the safety profile of somatrogon. The notable harms identified in the CADTH review protocol included the following: injection-related events, glucose intolerance and/or insulin resistance, excess IGF-I levels, malignancies, benign intracranial hypertension, slipped capital femoral epiphysis, scoliosis, tonsillar hypertrophy, carpal tunnel, and peripheral edema. In CP-4-006, of the identified notable harms, 43.1% and 25.2% of patients in the somatrogon group and Genotropin group, respectively, reported experiencing an injection-related event. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. In CP-4-009, of the identified notable harms, 72.7% of patients in the somatrogon group reported experiencing an injection-related event. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. The clinical expert did express concern that the injection site reaction in somatrogon was higher compared to Genotropin. As somatrogon is a once-weekly injection and Genotropin is a once-daily injection, these analyses may warrant further explanation.
In C0311002, 44.2% of patients during treatment with Genotropin and 54.0% of patients during treatment with somatrogon reported at least 1 adverse event.
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.
Conclusions
Two phase III randomized controlled trials were included in the CADTH systematic review of somatrogon for long-term treatment of pediatric patients who have growth failure due to an inadequate secretion of endogenous GH. Both studies demonstrated that for the primary efficacy outcome of annualized height velocity at 12 months, treatment with somatrogon was noninferior (CP-4-006) or comparable (CP-4-009) to Genotropin. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. Other relevant outcomes such as HRQoL and other PRO responses were not assessed in study CP-4-009 and were not properly assessed in study CP-4-006; as a result, the effect of somatrogon on HRQoL is uncertain. The results of the long-term open label phase for CP-4-006 were not available; hence, interpretation of sustained response of treatment to somatrogon is unknown. Key evidence gaps include the absence of adherence analyses, limited evidence on switchover from somatrogon to somatropin or vice versa, limited interpretation of HRQoL, and other PRO responses.
The key safety issues with somatrogon were related to injections, with a low number of serious adverse events being reported in the somatrogon treatment group and Genotropin treatment group in both studies. A higher number of patients in the somatrogon treatment group experienced injection site pain, nasopharyngitis, pyrexia, and headache. Conclusions regarding the long-term safety of somatrogon cannot be made in the absence of corresponding data.
References
1.Raben MS. Treatment of a pituitary dwarf with human growth hormone. J Clin Endocrinol Metab. 1958;18(8):901-903. PubMed
2.Decker R, Nygren A, Kristrom B, et al. Different thresholds of tissue-specific dose-responses to growth hormone in short prepubertal children. BMC Endocr Disord. 2012;12:26. PubMed
3.Chandrashekar V, Zaczek D, Bartke A. The consequences of altered somatotropic system on reproduction. Biol Reprod. 2004;71(1):17-27. PubMed
4.Silva JR, Figueiredo JR, van den Hurk R. Involvement of growth hormone (GH) and insulin-like growth factor (IGF) system in ovarian folliculogenesis. Theriogenology. 2009;71(8):1193-1208. PubMed
5.Tanner JM, Davies PS. Clinical longitudinal standards for height and height velocity for North American children. J Pediatr. 1985;107(3):317-329. PubMed
6.National Institute for Clinical Excellence. Human growth hormone (somatropin) for the treatment of growth failure in children. (Technology appraisal guidance TA188). http://www.nice.org.uk/guidance/TA188. Accessed 2021 Aug 30.
7.Saenger P. Biosimilar growth hormone. Indian J Pediatr. 2012;79(1):92-98. PubMed
8.Drug Reimbursement Review sponsor submission: Ngenla (somatrogon), 24 mg/1.2 mL (20 mg/mL), 60 mg/1.2 mL (50 mg/mL), prefilled pen for subcutaneous injection [internal sponsor's package]. Kirkland (QC): Pfizer Canada ULC; 2021 Jun 3.
9.Ngenla (somatrogon): 24 mg/1.2 mL (20 mg/mL), 60 mg/1.2 mL (50 mg/mL), prefilled pen for subcutaneous injection [product monograph]. Kirkland (QC): Pfizer Canada ULC; 2021 Oct 26.
10.Clinical Study Report: CP-4-006 (C0311009). A phase 3, open-label, randomized, multicenter, 12 months, efficacy and safety study of weekly MOD-4023 compared to daily Genotropin therapy in pre-pubertal children with growth hormone deficiency [internal sponsor's report]. New York (NY): Pfizer Inc; 2020 Aug 24.
11.Clinical Study Report: CP-4-009 (C0311010). A phase 3, open-label, randomized, multicenter, 12-month, efficacy and safety study of weekly MOD-4023 compared to daily Genotropin therapy in Japanese pre-pubertal children with growth hormone deficiency [internal sponsor's report]. New York (NY): Pfizer Inc; 2020 Dec 10.
12.Ahmed SF, Farquharson C. The effect of GH and IGF1 on linear growth and skeletal development and their modulation by SOCS proteins. J Endocrinol. 2010;206(3):249-259. PubMed
13.August GP, Lippe BM, Blethen SL, et al. Growth hormone treatment in the United States: demographic and diagnostic features of 2331 children. J Pediatr. 1990;116(6):899-903. PubMed
14.Grimberg A, Feemster KA, Pati S, et al. Medically underserved girls receive less evaluation for short stature. Pediatrics. 2011;127(4):696-702. PubMed
15.Grimberg A, Huerta-Saenz L, Grundmeier R, et al. Gender bias in U.S. pediatric growth hormone treatment. Sci Rep. 2015;5:11099. PubMed
16.Karlberg J. A biologically-oriented mathematical model (ICP) for human growth. Acta Paediatr Scand Suppl. 1989;350:70-94. PubMed
17.Hardin DS. Treatment of short stature and growth hormone deficiency in children with somatotropin (rDNA origin). Biologics. 2008;2(4):655-661. PubMed
18.Stanley T. Diagnosis of growth hormone deficiency in childhood. Curr Opin Endocrinol Diabetes Obes. 2012;19(1):47-52. PubMed
19.Humatrope (somatropin for injection): 5 mg vial; 6, 12, 24 mg cartridges [product monograph]. Toronto (ON): Eli Lilly Canada Inc; 2020 Jun 19: https://pdf.hres.ca/dpd_pm/00056950.PDF. Accessed 2021 Aug 9.
20.Genotropin GoQuickTM (somatropin [rDNA origin] for injection): 5 mg, 5.3 mg, 12 mg pre-filled pen, GoQuick, lyophilized powder for reconstitution; Genotropin MiniQuick (somatropin [rDNA origin] for injection: 02 mg, 04 mg, 06 mg, 08 mg, 1.0 mg, 1.2 mg, 1.4 mg, 1.8 mg, and 2.0 mg prefilled syringe, MiniQuick, lyophilized powder for reconstitution [product monograph]. Kirkland (QC): Pfizer Canada ULC; 2020 Jun 22: https://pdf.hres.ca/dpd_pm/00056686.PDF. Accessed 2021 Aug 9.
21.Nutropin (somatropin): 10 mg/vial lyophilized powder for injection; Nutropin AQ (somatropin): 10 mg/2 mL vial injection solution; Nutropin AQ PEN Cartridge (somatropin): 10 mg/2 mL pen cartridge injection solution; Nutropin AQ NuSpin (somatropin): 5 (5 mg/2 mL), 10 (10 mg/2 mL), 20 (20 mg/2 mL) injection solution, NuSpin injection device prefilled with cartridge [product monograph]. Mississauga (ON): Hoffmann-La Roche Limited 2012 Dec 21: https://pdf.hres.ca/dpd_pm/00018659.PDF. Accessed 2021 Aug 9.
22.Omnitrope (somatropin [rDNA origin] for injection): 5.8 mg/vial lyophilized powder for solution; 5 mg/1.5 mL, 10 mg/1.5 mL solution for injection [product monograph]. Boucherville (QC): Sandoz Canada Inc; 2015 May 8: https://pdf.hres.ca/dpd_pm/00030421.PDF. Accessed 2021 Aug 9.
23.Saizen (somatropin): 5 mg/vial somatropin for injection, lyophilized powder for reconstitution; Saizen (somatropin): 6 mg (5.83 mg/mL), 12 mg (8 mg/mL), 20 mg (8 mg/mL) solution for injection in a cartridge [product monograph]. Mississauga (ON): EMD Serono, a Division of EMD Inc., Canada; 2020 Jun 8: https://pdf.hres.ca/dpd_pm/00056674.PDF. Accessed 2021 Aug 9.
24.McGowan J, Sampson M, Salzwedel DM, Cogo E, Foerster V, Lefebvre C. PRESS Peer Review of Electronic Search Strategies: 2015 guideline statement. J Clin Epidemiol. 2016;75:40-46. PubMed
25.Grey matters: a practical tool for searching health-related grey literature. Ottawa (ON): CADTH; 2019: https://www.cadth.ca/grey-matters. Accessed 2021 Jun 22.
26.MacGillivray MH, Baptista J, Johanson A. Outcome of a four-year randomized study of daily versus three times weekly somatropin treatment in prepubertal naive growth hormone-deficient children. Genentech Study Group. J Clin Endocrinol Metab. 1996;81(5):1806-1809. PubMed
27.Wilton P, Gunnarsson R. Clinical experience with Genotropin in growth hormone deficient children. Acta Paediatr Scand Suppl. 1988;343:95-101. PubMed
28.Clinical Study Report: C031102. A phase 3, randomized, multicenter, open-label, crossover study assessing subject perception of treatment burden with use of weekly growth hormone (somatrogon) versus daily growth hormone (Genotropin) injections in children with growth hormone deficiency [internal sponsor's report]. Collegeville (PA): Pfizer Inc; 2020 Mar 24.
29.Turner-Bowker DM, Yaworsky A, Palladino A, et al. Development and psychometric evaluation of the Life Interference Questionnaire for Growth Hormone Deficiency (LIQ-GHD) to assess growth hormone injection burden in children and adults. Patient. 2020;13(3):289-306. PubMed
30.Clinical Study Report: CP-4-004. Safety and dose finding study of different MOD-4023 dose levels compared to daily r-hGH therapy in pre-pubertal growth hormone deficient children [internal sponsor's report]. New York (NY): Pfizer Inc; 2020 May 19.
31.Bloemeke J, Silva N, Bullinger M, Witt S, Dorr HG, Quitmann J. Psychometric properties of the quality of life in short statured youth (QoLISSY) questionnaire within the course of growth hormone treatment. Health Qual Life Outcomes. 2019;17(1):49. PubMed
32.Bullinger M, Quitmann J, Power M, et al. Assessing the quality of life of health-referred children and adolescents with short stature: development and psychometric testing of the QoLISSY instrument. Health Qual Life Outcomes. 2013;11:76. PubMed
Appendix 1: Literature Search Strategy
Note that this appendix has not been copy-edited.
Clinical Literature Search
Overview
Interface: Ovid
Databases:
MEDLINE All (1946-present)
Embase (1974-present)
Note: Subject headings and search fields have been customized for each database. Duplicates between databases were removed in Ovid.
Date of search: June 25, 2021
Alerts: Weekly search updates until project completion
Search filters applied: No filters were applied to limit the retrieval by study type.
Limits:
Publication date limit: none
Language limit: none
Conference abstracts: excluded
Syntax | Description |
|---|---|
/ | At the end of a phrase, searches the phrase as a subject heading |
MeSH | Medical Subject Heading |
exp | Explode a subject heading |
* | Before a word, indicates that the marked subject heading is a primary topic; or, after a word, a truncation symbol (wildcard) to retrieve plurals or varying endings |
# | Truncation symbol for one character |
adj# | Requires terms to be adjacent to each other within # number of words (in any order) |
.ti | Title |
.ot | Original title |
.ab | Abstract |
.hw | Heading word; usually includes subject headings and controlled vocabulary |
.kf | Author keyword heading word (MEDLINE) |
.kw | Author keyword (Embase) |
.dq | Candidate term word (Embase) |
.pt | Publication type |
.mp | Mapped term |
.rn | Registry number |
.nm | Name of substance word (MEDLINE) |
medall | Ovid database code: MEDLINE All, 1946 to present, updated daily |
oemezd | Ovid database code; Embase, 1974 to present, updated daily |
Multi-Database Strategy
(somatrogon* or Ngenla* or Lagova* or mod 4023 or mod4023 or "pf 06836922" or pf06836922 or pf 6836922 or pf6836922 or mod-401 or mod401 or 6D848RA61B).ti,ab,kf,ot,hw,nm,rn.
((carboxy-terminal peptide* or C-terminal peptide* or CTP) adj5 (hgh or rhgh or human growth hormone*)).ti,ab,kf,ot,hw,nm,rn.
exp growth hormone/ and (carboxy-terminal peptide* or C-terminal peptide* or CTP).ti,ab,kf,ot,hw,nm,rn.
or/1-3
4 use medall
*somatrogon/ or (somatrogon* or Ngenla* or Lagova* or mod 4023 or mod4023 or "pf 06836922" or pf06836922 or pf 6836922 or pf6836922 or mod-401 or mod401).ti,ab,kw,dq.
((carboxy-terminal peptide* or C-terminal peptide* or CTP) adj5 (hgh or rhgh or human growth hormone*)).ti,ab,kw,dq.
exp growth hormone derivative/ and (carboxy-terminal peptide* or C-terminal peptide* or CTP).ti,ab,kw,dq.
or/6-8
9 use oemezd
10 not (conference review or conference abstract).pt.
5 or 11
remove duplicates from 12
Clinical Trials Registries
ClinicalTrials.gov
Produced by the U.S. National Library of Medicine. Targeted search used to capture registered clinical trials.
[Search terms – somatrogon, growth hormone deficiency]
WHO ICTRP
International Clinical Trials Registry Platform, produced by the World Health Organization. Targeted search used to capture registered clinical trials.
[Search terms – somatrogon, growth hormone deficiency]
Health Canada’s Clinical Trials Database
Produced by Health Canada. Targeted search used to capture registered clinical trials.
[Search terms – somatrogon, growth hormone deficiency]
EU Clinical Trials Register
European Union Clinical Trials Register, produced by the European Union. Targeted search used to capture registered clinical trials.
[Search terms – somatrogon, growth hormone deficiency]
Grey Literature
Search dates: June 21-25, 2021
Keywords: somatrogon, growth hormone deficiency
Limits: none
Updated: Search updated prior to the completion of stakeholder feedback period.
Relevant websites from the following sections of the CADTH grey literature checklist Grey Matters: A Practical Tool for Searching Health-Related Grey Literature were searched:
Health Technology Assessment Agencies
Health Economics
Clinical Practice Guidelines
Drug and Device Regulatory Approvals
Advisories and Warnings
Drug Class Reviews
Clinical Trials Registries
Databases (free)
Internet Search
Open Access Journals
Appendix 2: Excluded Studies
Reference | Reason for exclusion |
|---|---|
Clinical Study Report: CP-4-004. Safety and dose finding study of different MOD-4023 dose levels compared to daily r-hGH therapy in pre-pubertal growth hormone deficient children [internal sponsor’s report]. New York (NY): Pfizer Inc; 2020 May 19.30 | Phase II study |
Note: This table has not been copy-edited.
Appendix 3: Description and Appraisal of Outcome Measures
Note that this appendix has not been copy-edited.
Aim
To describe the following outcome measures and review their measurement properties (validity, reliability, responsiveness to change, and MID):
QoLISSY
OAT and PAT
Patient life interference questionnaire (LIQ-GHD)
The QoLISSY, OAT, and PAT were included as other outcomes in the pivotal trial. The patient life interference questionnaire or LIQ-GHD was evaluated as a primary and secondary outcome in study 002, which was summarized under Other Relevant Evidence.
Findings
Table 33: Summary of Outcome Measures and Their Measurement Properties
Outcome measure | Type | Conclusions about measurement properties | MID |
|---|---|---|---|
QoLISSY | Self-reported/observer-reported assessment of HRQoL in in children and adolescents with ISS and GHD. Items from 3 core domains (physical, social, and emotional) are summed for a 22-item total score. The core domains are accompanied by 28 additional items reflecting coping (10 items), beliefs (4 items), and treatment (14 items). Items are answered using a 5-point Likert type scale; subscale scores are transformed to a score from 0 to 100 where higher values represent a higher HRQoL. | Reliability: Internal consistency reliability was demonstrated (Cronbach’s alpha ≥ 0.70). Validity: Acceptable content validity (known-groups approach) and construct validity (confirmatory factor analysis approach) were reported for the core domains of the QoLISSY, although associated with uncertainty based on a secondary evaluation of validity that yielded mixed results. Responsiveness: The QoLISSY was unable to demonstrate adequate responsiveness over 12 months. | Evidence of a MID was not identified. |
OAT and PAT | Questionnaires used to record the observer (OAT) or patient/caregiver (PAT) assessment of successful administration of injection of study drug. | Evidence of validity, reliability, and responsiveness of the OAT and PAT were not identified during this review. | Evidence of a MID was not identified. |
Patient life interference questionnaire (LIQ-GHD) | PRO questionnaire/COA of the treatment burden associated with growth hormone injections in adults/children, respectively Consists of 9 domains, each with varying number of items. Items were evaluated using either a 5-point Likert type scale, 5-point verbal response scale, or 11-point numeric rating scale. Questions were answered based on a 4-week or 1-week recall period. | Reliability: Internal consistency reliability demonstrated for all domains (Cronbach’s alpha ≥ 0.70), with the exception of injection signs reported by caregiver domain (alpha = 0.653) and satisfaction and willingness to continue domain (alpha = 0.589).
Test-retest reliability was assessed and demonstrated across domains in the overall sample based on an ICC ≥ 0.70.
Validity: Some evidence of construct validity was reported in the overall sample (including children, adolescents, and adults). Concurrent validity was not demonstrated in the subgroup for children. Responsiveness: Evidence of responsiveness was not identified. | Evidence of a MID was not identified. |
COA = clinical outcome assessment; ISS = idiopathic short stature; GHD = growth hormone deficiency; ICC = intraclass correlation coefficient; LIQ-GHD = Life Interference Questionnaire for Growth Hormone Deficiency; MID = minimal important difference; OAT = Observer Assessment Tool; PAT = Participant Assessment Tool; PRO = patient-reported outcome; QoLISSY = Quality of Life in Short Stature Youth.
Quality of Life in Short Stature Youth
The QoLISSY questionnaire is an instrument developed by Pfizer that is used to obtain a self-reported assessment of HRQoL in children and adolescents with idiopathic short stature and GHD. The questionnaire was designed for self-reported use by adolescents between the age of 8 and 18 years and observer-reported use by parents of children between the age of 4 and 18 years. The QoLISSY includes 3 core domains, namely the 6-item physical domain, 8-item social domain, and 8-item emotional domain. The 3 core domains are summed for a 22-item total score. The core domains are accompanied by 28 additional items reflecting 3 predictors of quality of life: coping (10 items), beliefs (4 items), and treatment (14 items). Additionally, there is a version of the QoLISSY for parents that contains 2 supplementary domains about the parent’s worries about their child’s future (5 items) and the impact of the child’s condition on the parent’s well-being (11 items).
Each of the items are answered on a 5-point Likert type scale, which ranges from “not at all/never” (1) to “extremely/always” (5) for positively worded items. Scoring is reversed for negatively worded item, where “not at all/never” = 5 and “extremely/always” = 1. Of note, items are positively or negatively worded. Negatively worded items need to be reversed so that a higher sub scale score reflects higher quality of life. All subscale scores are transformed from raw scores to 0 to 100 scores with higher values representing a higher quality of life.31
The study describing the original development and psychometric assessment of the QoLISSY reported acceptable internal consistency reliability of the QoLISSY demonstrated in patients with diagnosed short stature (GHD or ISS) based on a Cronbach’s alpha of 0.70 or more.32 Evidence of acceptable content and construct validity assessed using a known groups approach and confirmatory factor analysis, respectively, were also reported for the core domains of the QoLISSY.32 The reliability, validity and responsiveness to change of the QoLISSY was also evaluated in the context of treatment for GHD by Bloemeke et al. (2019).31 This assessment was based on a prospective observational study, conducted in a sample of children and adolescents with idiopathic GHD and SGA. Patients were treated with hGH treatment over 1 year (12 months) and HRQoL in this population was compared to untreated children and adolescents with ISS. Some evidence of content validity was demonstrated for the QoLISSY. More specifically, moderate positive correlations (r = 0.28 to 0.46) were found between disease-specific and generic HRQoL instruments for patient reports, and weak to moderate positive correlations (r = 0.13 to 0.41) were found between disease-specific and generic HRQoL instruments for parent reports. Construct validity was assessed using a known-groups approach; however, results were mixed as the QoLISSY was unable to distinguish between severity level of short stature. Internal consistency reliability was assessed by Cronbach’s alpha, and was satisfactory (Cronbach’s alpha ≥ 0.70) in all scales except coping in the child-report (Cronbach’s alpha < 0.70). Responsiveness over 12 months was assessed using a repeated measures multivariate analyses of covariance. Responsiveness was not demonstrated based on differences in HRQoL that were not statistically significant over time.
Evidence of a MID was not identified for the QoLISSY during this review. It was noted that addition evidence of the use of the QoLISSY over time is required to determine the clinical meaningfulness of changes in HRQoL scores.31
Observer Assessment Tool and Participant Assessment Tool
The OAT and PAT questionnaires were used to evaluate the number of successful injection attempts by patients using the somatrogon pre-filled pen.10
The OAT was used to record the observer’s assessment of the use of the device by patients or caregivers to administer somatrogon by injection. The observer recorded whether an injection attempt was successful or not based on the question from the OAT “…did the user successfully inject into an acceptable injection site without physical assistant?” The injection attempt was considered successful if the observer answered “yes” to the questions for all attempts on the form. Of note, the instructions for the OAT instruct the observer as follows: “If the user is able to successfully administer the dose but requires your help to complete the injection, document the specifics below, including asking the user if after the injection they are now confident that they will be able to inject unassisted at home.” This introduces substantial subjectivity in the assessment of a successful injection administration.10
The PAT was also used to record the patient’s or caregiver’s injections of somatrogon, based on an assessment by the user of the pen or parent/legal guardian. The following questions were used to determine a successful injection attempt via the PAT: “Did the dose window show ‘0’ when you finished your injection?” and “Do you believe that a full dose was injected?” The injection attempt was considered successful if the subject answered “yes” to both questions for all attempts on the form.10
No evidence of validation of the OAT or PAT were identified for this review.
Patient Life Interference Questionnaire
The Life Interference Questionnaire for Growth Hormone Deficiency (LIQ-GHD) is a questionnaire designed to evaluate the treatment burden associated with GH injections in children and their caregivers, as well as adults.29 The development and psychometric evaluation of the LIQ-GHD was conducted and supported by Pfizer. It is available as 2 versions, the LIQ-GHD Adult and LIQ-GHD-Pediatric. The pediatric version is the focus of the discussion herein.
The LIQ-GHD-Pediatric is a clinical outcome assessment intended for children and adolescents between the age of 3 and 17. The LIQ-GHD-Pediatric was designed for use by the patient and caregiver in a “dyadic administration” approach. It is composed of questions regarding the severity of injection symptoms, which are only administered to children and adolescents who are able to reliably self-report. It also includes observer-reported outcomes completed by the caregiver regarding the caregiver experience, experience of the family, and observed behaviours of the child.29 More specifically, the LIQ-GHD includes 9 domains with a varying number of items, described as follows:
pen ease of use, 5 items
ease of injection schedule, 2 items
patient life interference, 5 or 7 items
satisfaction and willingness to continue, 2 items
missed injections, 2 items
injection signs and symptoms (patient reported), 4 items
injection signs (caregiver reported), 2 items
caregiver life interference, 5 or 7 items
family life interference, 5 or 6 items
Items that assess the ease of treatment and treatment satisfaction use a 5-point Likert type scale for responses that ranges from ‘easy’ or ‘satisfied’ to ‘difficult’ or ‘dissatisfied.’ Items that assess the frequency of experience used a 5-point verbal response scale, and items assessing sign or symptom severity used an 11-point numeric rating scale. The items about missed injections were reported as the number of missed injections, and the reason for missed injections using multiple choice options. Most of the items were based on a 4-week recall period, with the exception of injection signs and symptoms (patient reported) and Injection Signs (caregiver reported), which were based on a 7-day recall period.29
The psychometric properties of the LIQ-GHD were evaluated in a sample of 224 participants with GHD through an online observational study.29 A total of 70 children (age 3 to 11 years)/caregiver dyads, 79 adolescents (age 12 to 17 years)/caregiver dyads, and 75 adults (25 years or older). Floor effects were reported, but no ceiling effects were observed. In the overall sample, internal consistency reliability was demonstrated for all domains, based on a Cronbach’s alpha of 0.70 or more, with the exception of injection signs reported by caregiver domain (alpha = 0.653) and satisfaction and willingness to continue domain (alpha = 0.589). In the child/caregiver dyad subgroup, internal consistency reliability results were consistent with the overall population. In the adolescent/caregiver dyad subgroup, the satisfaction and willingness to continue domain was the only domain that did not meet the 0.70 threshold for internal consistency reliability. Test-retest reliability was also assessed and reported using ICCs, and demonstrated across domains in the overall sample based on an ICC of 0.70 or more. Results for test-retest reliability were similar when analyzed by subgroup, with the exception of the ease of injection schedule domain (ICC = 0.679) and satisfaction and willingness to continue domain (ICC = 0.697) in children.
Construct validity was assessed using a known groups approach for the patient life interference domain and ease of injection schedule domain. Five known groups were analyzed with differences between groups anticipated in the overall population; however, no significant differences were observed between groups defined by self-rating of overall health or severity of GHD. It was noted that analyses in the subgroups (i.e., children or adolescents) were limited due to a small sample size. Concurrent validity was assessed by testing a priori hypotheses about the strength of correlation with the Self-Injection Assessment Questionnaire (SIAQ); however, concurrent validity was not observed in the child/caregiver dyad subgroup.29
Evidence of a MID was not identified in the literature during this review.
Pharmacoeconomic Review
Abbreviations
BIA
budget impact analysis
GH
growth hormone
GHD
growth hormone deficiency
HSDS
height standard deviation score
HV
height velocity
ICER
incremental cost-effectiveness ratio
LAGH
long-acting growth hormone
QALY
quality-adjusted life-year
SAGH
short-acting growth hormone
SC
subcutaneous
Executive Summary
The executive summary comprises 2 tables (Table 1 and Table 2) and a conclusion.
Item | Description |
|---|---|
Drug product | Somatrogon (Ngenla), 24 mg/1.2 mL (20 mg/mL) as a pre-filled pen or 60 mg/1.2 mL (50 mg/mL) as a pre-filled pen for subcutaneous injection |
Submitted price | Somatrogon, 24 mg/1.2 mL pre-filled pen: $345.84 Somatrogon, 60 mg/1.2 mL pre-filled pen: $864.60 |
Indication | Proposed: For the long-term treatment of pediatric patients who have growth failure due to an inadequate secretion of endogenous growth hormone (growth hormone deficiency) |
Health Canada approval status | NOC |
Health Canada review pathway | Standard review pathway |
NOC date | October 26, 2021 |
Reimbursement request | Long-term treatment of pediatric patients who have growth hormone deficiency |
Sponsor | Pfizer Canada ULC |
Submission history | Previously reviewed: No |
NOC = Notice of Compliance.
Table 2: Summary of Economic Evaluation
Component | Description |
|---|---|
Type of economic evaluation | Cost-utility analysis Markov model |
Target population(s) | Prepubertal children (ranging from 3 years to 11 years for girls and 12 years for boys) with either isolated GHD or GH insufficiency associated with multiple pituitary hormone deficiencies and who are treatment naive |
Treatment | Somatrogon (once-weekly injection) |
Comparator | Somatropin (once-daily injection, average cost of all branded somatropin products weighted by market share) |
Perspective | Canadian publicly funded health care payer |
Outcome | QALYs |
Time horizon | Maximum of 15 years (i.e., until the age of 18; total length of time horizon depends on age of treatment initiation) |
Key data source | CP-4-006, a phase III, 24-week, multi-centre, randomized crossover trial |
Submitted results | Somatrogon was dominant; it was associated with more QALYs (a gain of 0.28) and was less costly (a saving of $6,369) compared with somatropin |
Key limitations |
|
|
|
GH = growth hormone; GHD = growth hormone deficiency; ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year; vs. = versus.
Conclusions
The clinical evidence submitted by the sponsor demonstrated that in pediatric patients with growth hormone deficiency (GHD), treatment with somatrogon was noninferior to Genotropin based on the clinically meaningful treatment difference in the pivotal trial (CP-4-006) primary end point (i.e., a change in height of ≥ –1.8 cm per year was met).
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| for the long-term treatment of growth failure due to an inadequate secretion of endogenous growth hormone (GH). The clinical efficacy of somatrogon beyond 12 months is unknown.
CADTH’s base-case reanalysis included assuming equal height velocities (HVs) for both somatrogon and somatropin, and revising the dose for all somatropin products to align with the dose most commonly used in Canadian clinical practice. In the CADTH base case, somatrogon was associated with an incremental cost-effectiveness ratio (ICER) of $107,714 per QALY gained (incremental costs of $27,005 and an incremental benefit of 0.25 QALYs) compared with somatropin. A price reduction of at least 11% would be required for somatrogon to be considered cost-effective at a willingness-to-pay threshold of $50,000 per QALY gained.
The cost-effectiveness of somatrogon is primarily driven by drug acquisition costs associated with various somatropin products. These costs are affected by dosing and wastage assumptions, as well as the magnitude of the quality-of-life benefit related to weekly rather than daily injections associated with somatrogon.
Stakeholder Input Relevant to the Economic Review
This section is a summary of the feedback received from drug plans that participated in the CADTH review process (specifically, information that pertains to the economic submission). Neither patient input nor clinician input was received.
Feedback from the drug plans indicated that somatropin (Genotropin) is reimbursed by all public drug plans for GHD, either through drug plan listings or separate programs. Drug plans indicated that additional biosimilar products for somatropin may become available, which may decrease its costs. Drug plans had concerns about the anticipated budget impact of reimbursing somatrogon due to the possibility of indication creep with other conditions such as patients with chronic renal failure, Turner syndrome, idiopathic short stature, Prader-Willi syndrome, and adult-onset conditions. Drug plans reported that although somatrogon injections are weekly, the most common adverse effects of somatrogon are injection site reactions, most of which do not result in treatment discontinuation. Drug plans also noted that there are some negotiated prices for comparators, and that somatrogon is more expensive than some somatropin options based on their public list prices. Finally, drug wastage may be an issue with GH products, including somatrogon.
Several of these concerns were addressed in the sponsor’s model:
The sponsor’s base-case analysis compared somatrogon to a mix of somatropin treatments (Genotropin, Humatrope, Norditropin Nordiflex, Nutropin AQ, Omnitrope, Saizen).
The sponsor captured various types of wastage that may be associated with GH products, including last-dose wastage, device-setting wastage, storage wastage, preparation wastage, and adherence wastage.
In addition, CADTH addressed some of these concerns as follows:
The cost-effectiveness of somatrogon compared to Genotropin only was conducted in a scenario analysis to address drug plan feedback, as Genotropin is reimbursed by all public drug plans for the given indication and has a list price lower than that of somatrogon.
CADTH was unable to address the following concerns raised from stakeholder input:
Adverse events such as injection site reactions associated with weekly or daily injections were not included in the sponsor’s model.
Off-label use of somatrogon or indication creep resulting from patients with other non-GHD indications could not be addressed by the sponsor’s submitted budget impact analysis (BIA).
The cost-effectiveness of somatrogon in comparison with the introduction of additional biosimilar somatropin products, should they become available, is unknown.
Economic Review
The current review is for somatrogon (Ngenla) for the long-term treatment of pediatric patients who have GHD.
Economic Evaluation
Summary of Sponsor’s Economic Evaluation
Overview
The sponsor submitted a cost-utility analysis assessing somatrogon versus somatropin for the long-term treatment of prepubertal children (ranging from 3 years to 11 years for girls and 12 years for boys) with either isolated GHD or GH insufficiency associated with multiple pituitary hormone deficiencies and who are treatment naive. While the proposed Health Canada indication states somatrogon is indicated for the long-term treatment of pediatric patients who have growth failure due to an inadequate secretion of endogenous GH, the modelled population was aligned with the trial population from the pivotal trial, CP-4-006.
The recommended dosage for somatrogon is 0.66 mg/kg administered once weekly by subcutaneous (SC) injection.1 At a submitted cost of $345.84 for a 24 mg/1.2 mL pre-filled pen for SC injection, or $864.60 for a 60 mg/1.2 mL pre-filled pen for SC injection, the annual cost of somatrogon is approximately $9,684, assuming a patient weight of 19.30 kg and considering wastage of unused product. The recommended dosage for somatropin varies by brand (Genotropin, Humatrope, Norditropin Nordiflex, Nutropin AQ, Omnitrope, Saizen) and ranges from 0.16 mg/kg per week to 0.30 mg/kg per week for an average annual cost ranging from $4,584 to $14,709, depending on the brand and assuming a patient weight of 19.30 kg as well as considering wastage of unused product. In their base case, somatropin was a single comparator reflected as a mix of somatropin products that were assumed to have equivalent efficacy and safety. The total drug acquisition cost of somatropin was an average cost weighted by the market shares of the various somatropin products. The sponsor’s model also considered several forms of wastage, such as device-setting wastage, storage wastage, and preparation wastage, depending on the device.2
The economic analysis was conducted from the perspective of the public health care payer over a maximum time horizon of 15 years, with the time horizon varying by a patient’s age at treatment initiation and the time horizon terminating at 18 years of age.2 Costs and clinical outcomes (i.e., quality-adjusted life-years [QALYs]) were discounted at a rate of 1.5% per annum. The model was stratified by the age of treatment initiation such that the overall results were weighted by the anticipated distribution of patients who initiated treatment at each age.
Model Structure
A Markov model structure was developed to capture the long-term costs and effects of treatment for isolated GHD or GH insufficiency. The disease course was modelled via 2 health states: alive on-treatment and alive off-treatment (Figure 1 in Appendix 3). Each model cycle was 1 year in duration.2 Patients entered the model alive on-treatment, between the ages of 3 years and 12 years, and received either a weekly SC injection of somatrogon or a daily SC injection of somatropin. Patients were assumed to remain on their assigned treatment until they were taken off treatment at the age of 18 years unless they discontinued treatment due to a lack of response in the first model cycle. Over the course of yearly cycles, patients experienced gains in height as determined by HV, which were converted to a height standard deviation score (HSDS), and accrued treatment-related costs and clinical benefits attributed to gains in height.2
Model Inputs
The patient cohort consisted of pediatric patients with isolated GHD or GH insufficiency whose baseline characteristics mainly reflected the pivotal trial CP-4-006. Approximately 71.9% of the population was assumed to be male and the age range of patients who could initiate treatment in the model was 3 years to 12 years, with a mean age of 7.7 years, reflective of CP-4-006. The proportion of patients assumed to initiate treatment by age was derived from CP-4-006 and used to weight model results. In each model cycle, patient weight was calculated via body mass index data for the general pediatric population at each age, and this was further assumed to be applicable to the target population.
Efficacy data for somatrogon and somatropin were based on the primary efficacy end point in CP-4-006, the annualized HV (i.e., change in height in cm per year) at the end of 12 months.3 This was used to derive the gain in height after the first model cycle. Yearly HV data for year 2 onwards (subsequent model cycles) were derived from a German study by Ranke and Lindberg (2010)4 that compared observed and expected growth over the first 2 years of GH treatment in prepubertal children. Specifically, changes in the HSDS that were observed from year 1 to year 2 by age in the Ranke study were used to derive the yearly HV from year 2 onwards for somatrogon and somatropin by calculating an average percentage of decrease in HSDS gain; these were assumed to apply across age bands over time. Additionally, the sponsor assumed that approximately 20.4% of patients would discontinue treatment after year 1, based on a database analysis of claims from the US.2 All other patients were assumed to continue treatment for the remainder of the time horizon. For each year that patients were alive and on treatment, an annual treatment-specific adherence rate was assumed. In year 1 of treatment, adherence with somatrogon was assumed to be 5% higher than somatropin due to its weekly administration schedule, followed by an identical annual relative rate of decline in treatment adherence for both treatments. In the economic model, it was further assumed that poor treatment adherence would lead to a relative reduction in HV, based on a study by Maggio et al. (2018).5
Health state utility values were derived from a study that examined the influence of short stature, via HSDS, on health-related quality of life among adults from the general adult population in the UK via the EQ-5D Three-Level questionnaire.6 Utility values from this study were the basis for a linear interpolation that was conducted to derive utility scores by HSDS in the economic model for each 0.01 increase in HSDS, rather than the 0.5 increments estimated by Christensen et al. (2007).2 Based on this linear interpolation, the utility by HSDS was applied to patients at each model cycle. An additional benefit associated with weekly injections for somatrogon, in comparison with the daily injections with somatropin, was applied to patients who received somatrogon, based on a study by Boye et al. (2011).7
Costs captured in the economic model included drug acquisition costs, those associated with health care resource use,8-10 and monitoring costs.8-10 Included in the drug acquisition costs were various forms of potential drug wastage associated with injectable GH therapies, including last-dose wastage at a threshold of 10%, preparation wastage, storage wastage, and device-setting wastage. The sponsor calculated drug costs based on the price per mg basis rather than by dosage form (i.e., pen, cartridge, or device).2
Summary of Sponsor’s Economic Evaluation Results
The sponsor presented probabilistic analyses for their base case based on 5,000 iterations. The deterministic results were similar to the probabilistic results. The results presented are the overall results, which are weighted by the anticipated distribution of patients who initiated treatment at each age from ages 3 to 12.
Base-Case Results
In the sponsor’s probabilistic base case, somatrogon was found to be less costly (–$6,661 incremental costs) and more effective (0.28 incremental QALYs) than somatropin over the modelled time horizon. As a result, somatrogon was dominant when compared to somatropin.2 The sponsor’s model predicted an average total height gain of 2.28 cm for somatrogon compared with somatropin. The model results indicated that approximately 7% of the incremental benefit of somatrogon compared with somatropin was derived from the 12-month observed trial period, with the remaining benefit from the period for which there is no available data.2
Table 3: Summary of the Sponsor’s Economic Evaluation Results
Drug | Total costs ($) | Incremental costs ($) | Total QALYs | Incremental QALYs | ICER vs. reference ($/QALY) |
|---|---|---|---|---|---|
Somatropin | 147,126 | Reference | 7.44 | Reference | Reference |
Somatrogon | 153,787 | 6,661 | 7.72 | 0.28 | Dominant |
ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year; vs. = versus.
Source: Sponsor’s Pharmacoeconomic Submission (2021).2
Sensitivity and Scenario Analysis Results
The sponsor conducted several sensitivity and scenario analyses. These included varying the time horizon to account for a maximum age of treatment of 19 years and 16 years, varying the discount rate to 0% and 3%, assuming no product wastage, assuming no adherence wastage, changing the proportion of patients who discontinued GH therapy after year 1 to 10% and 30%, varying the age of treatment initiation among patients (between ages 3 years to 7 years, and between ages 8 years to 12 years), exploring the impact of using the lowest and highest costs of somatropin within its price range, using the lowest and highest doses for somatropin, changing the source of data for HV adjustment by adherence, and assuming no adherence benefit for somatrogon across the time horizon. The sponsor’s base-case results revealed that somatrogon remained dominant in all scenarios, except in the scenario for which the dose of somatropin was set to 0.16 mg/kg per week (ICER range = $141,220) and the scenario for which Genotropin captured the entire market share distribution (ICER range = $116,714).2
CADTH Appraisal of the Sponsor’s Economic Evaluation
CADTH identified several key limitations to the sponsor’s analysis that have notable implications on the economic analysis.
Somatrogon is noninferior to somatropin: In the submitted economic model, yearly HV in year 1 was based on the primary efficacy end point in the pivotal trial for both somatrogon and somatropin (i.e., Genotropin). For the remaining years of treatment in the model (year 2 onwards), the sponsor predicted age-specific yearly changes in HV for patients who received somatrogon and somatropin using data from Ranke and Lindberg (2010), a study that compared observed and expected growth in response to GH treatment in prepubertal children with growth disorders (including severe or less severe GHD, Turner syndrome, or short children who were born small for gestational age). The sponsor’s model included greater yearly HVs for patients treated with somatrogon than for patients treated with somatropin; however, the CADTH clinical review concluded that the available evidence demonstrated treatment with somatrogon was noninferior to Genotropin, ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. As a result, there is no clinical evidence to suggest that |||||||||||||||||||||||||||||||||||||||||||||||| with regards to gains in height over the period for which there is observed data, nor is there any long-term comparative efficacy data. The sponsor’s prediction of an additional gain in height with somatrogon in comparison with somatropin for patients fully adherent to treatment over the modelled time horizon biased results in favour of somatrogon.
CADTH addressed this limitation by assuming equal HVs, including standard errors, for patients who received somatrogon and somatropin over the entire model time horizon.
The magnitude of benefit associated with weekly somatrogon injections in comparison with daily somatropin injections is uncertain: The sponsor assumed that there was an incremental utility benefit (0.023 per year) associated with weekly injections of somatrogon compared with daily injections of somatropin. The clinical expert consulted by CADTH for this review noted that the assumption that a weekly injection compared to a daily injection would be associated with an improvement in a patient’s quality of life was reasonable, since any change or reduction in the number of injections is important to patients. However, there remains uncertainty associated with the magnitude of the added utility benefit assumed by the sponsor, as this has yet to be assessed in the context of children with GHD or GH insufficiency. The utility increment used in the sponsor’s submission was based on a study that assessed utilities and disutilities associated with injectable treatments for type 2 diabetes.7 Should the magnitude of benefit with weekly injections be smaller than that assumed by the sponsor, somatrogon would be less cost-effective (i.e., ICER would increase) given a resulting smaller gain in incremental QALYs over somatropin.
CADTH was unable to address this limitation. In a scenario analysis, CADTH explored the impact of no additional utility benefit with weekly versus daily treatment administration for patients treated with somatrogon.
Dosing for somatropin does not align with the dose commonly used in Canadian clinical practice: In the economic model, the sponsor assumed that the average administered dose for all somatropin products was 0.24 mg/kg per week. The clinical expert consulted by CADTH indicated that the average dose applied in the sponsor’s base case was higher than the average dose commonly used in Canadian clinical practice, which is typically 0.18 mg/kg per week, and further noted that very few patients are administered a higher dose. The average administered dose in Canadian clinical practice as indicated by CADTH’s clinical expert is supported by results from the Canadian cohort of the multinational phase IV prospective observational Genetics and Neuroendocrinology of Short Stature International Study of children with various causes of short stature, including patients with GHD who were treated with GH.11 Deal et al. (2018) reported that the average somatropin dose administered to children with GHD within the Canadian cohort over an approximate 5-year period was 0.18 mg/kg per week and further indicated that Canadian mean GH doses were lower than those administered in the US or globally. CADTH’s clinical expert further indicated that the conclusions from the CADTH clinical review relating to the noninferiority of somatrogon compared with somatropin in CP-4-006 would still be applicable to patients receiving this lower dose. As drug costs are a key driver of model results, the sponsor’s assumption of a higher dose for all somatropin products overestimated the total costs associated with somatropin, biasing cost-effectiveness results in favour of somatrogon.
To align with the dosing expected in Canadian clinical practice, the CADTH base case altered the dosing for somatropin treatments to 0.18 mg/kg per week, as administered in Deal et al. (2018). CADTH notes that this value was held constant in the CADTH reanalyses, as the clinical expert consulted by CADTH noted that there was limited variability in this dose.
Uncertainty in the market share distribution of various branded somatropin treatments: In the economic model, the sponsor compared somatrogon to somatropin, which consisted of a mix of various GH treatments (i.e., Genotropin, Humatrope, Norditropin Nordiflex, Nutropin AQ, Omnitrope, and Saizen). The sponsor calculated drug costs for somatropin treatments by incorporating the market share distribution of the GH treatments by brand and by size and/or dose form within each brand. However, there is uncertainty around the market share distribution assumed for each somatropin product. The sponsor’s estimates are not indication specific, and the clinical expert consulted by CADTH noted there are likely to be differences between the distribution of brands of somatropin for pediatric GHD or GH insufficiency in comparison with the overall GH market for all indications. As drug costs are a key driver of model results, there is potential for the assumed market share distribution to significantly affect the cost associated with somatropin and the resulting cost-effectiveness of somatrogon.
CADTH was unable to address the limitation. In a scenario analysis, CADTH compared the cost-effectiveness of somatrogon to the least costly somatropin product (Genotropin) to align with the comparator in the sponsor’s clinical submission and to address drug plan feedback.
There is uncertainty with the use of utility scores by HSDS: In the economic model, the sponsor assumed a linear relationship between health-related quality of life and height, based on a study that assessed EQ-5D Three-Level utility scores by HSDS.6 This study found that mean EQ-5D scores were lower in adults with a height shorter than that of the average person, with the EQ-5D score decreasing as the HSDS became further removed from the average. The sponsor further conducted a linear interpolation analysis using the Christensen et al. (2007) study to predict patient utility by HSDS, such that patient utility scores could be measured in 0.1 HSDS increments rather than increments of 0.5. The applicability of these results to the sponsor’s submitted model and patients with GHD or GH insufficiency is associated with some uncertainty, including the generalizability of these values in adults to children with GHD or GH insufficiency, as well as the validity of the linear interpolation. As there were only minor differences in final height predicted by the model, the use of these utility values has minimal impact on model results.
CADTH was unable to address this limitation.
Relative treatment adherence rates for patients on somatrogon and somatropin are uncertain: In the economic model, the sponsor has assumed patients on somatrogon would have greater adherence than patients receiving treatment with somatropin. The clinical expert consulted by CADTH noted that the sponsor’s assumption of a 5% increase in adherence at baseline appeared reasonable in the absence of available data, and that the difference may even be greater than predicted in the sponsor’s model in subsequent years. In the absence of adherence data comparing the 2 treatments, there remains uncertainty related to the treatment adherence rates applied in the economic model. Given poorer adherence rates were assumed to affect yearly HV, this assumption did favour somatrogon.
CADTH could not address this limitation. Two different adherence assumptions were tested in scenario analyses, 1 with equal treatment adherence at baseline and another with greater treatment adherence with somatrogon than that assumed by the sponsor.
Additional limitations were identified but were not considered to be key limitations. These limitations are outlined subsequently.
Treatment discontinuation after the first year of treatment does not align with clinical expectations of discontinuation from GH treatment: In their base case, the sponsor assumed 20.4% of patients would discontinue any GH treatment following year 1 due to lack of efficacy, according to a 2005 study.12 In contrast, a much lower rate of discontinuation was reported in CP-4-006 for each treatment group (0.9%). The clinical expert consulted by CADTH indicated that the sponsor’s estimate was much higher than expected in practice and the proportion of patients reported to have discontinued treatment in CP-4-006 due to a lack of efficacy was much lower than expected in Canadian clinical practice, as clinical trials are conducted in tightly controlled environments relative to the practice setting. Further, the expert noted that discontinuation rates of GH treatments among children in Canada are unlikely to be high after the first year of treatment.
In a scenario analysis, CADTH arbitrarily assumed a lower discontinuation rate of 10%.
Drug cost calculations may be slightly underestimated or overestimated due to the sponsor’s approach to calculating drug costs and uncertainty with wastage assumptions: In the economic model, drug costs were calculated on a per mg basis rather than by dose form or device type (i.e., pen, vial, or cartridge), which may have led to some underestimation and overestimation of drug costs, depending on the product. As GH treatments are typically dispensed by dose form or device type rather than by mg, costs should have been calculated based on the full device amount (e.g., rounding up to nearest full pen). Additionally, the sponsor assumed various types of wastage, which appear to be appropriate. However, there remains uncertainty around wastage assumptions related to storage wastage, preparation wastage, and last-dose wastage, as these wastage assumptions could not be validated by the clinical expert consulted by CADTH.
CADTH was unable to address these limitations. In a scenario analysis, CADTH explored the impact of assuming no wastage.
The sponsor inappropriately included probability distributions for drug costs of somatropin: The sponsor’s model included probabilistic distributions for drug costs of somatropin. Drug costs are fixed and should not be varied across model runs.
CADTH corrected the sponsor’s base case by fixing the drug costs for somatropin. All subsequent reanalyses included this change.
Additionally, the following key assumptions were made by the sponsor and have been appraised by CADTH (refer to Table 4).
Table 4: Key Assumptions of the Submitted Economic Evaluation (Not Noted as Limitations to the Submission)
Sponsor’s key assumption | CADTH comment |
|---|---|
A time horizon of a maximum of 15 years (i.e., until the age of 18 or adulthood) was adopted in the sponsor’s base case. | Appropriate. According to the CADTH Guidelines for the Economic Evaluation of Health Technologies, the time horizon selected in the economic model should be long enough to capture the costs and effects of treatment. This treatment is administered in childhood and is expected to continue until the age of 18 at the latest, as the disease is expected to resolve by then. |
The maximal treatment duration in the model is 15 years. | Likely appropriate. CADTH’s clinical expert indicated that treatment is typically continued until full bone maturity is reached; however, some physicians may continue to prescribe growth hormone for some time shortly afterwards. The latter has not been assessed in the sponsor’s economic submission, nor by CADTH. |
In the model, patients can receive treatment between ages 3 and 18. The oldest age to initiate treatment is 12 years for males and 11 years for females. | Likely appropriate. CADTH’s clinical expert noted that the typical age range of treatment is from 2 years to 3 years until full bone maturity is reached, which is expected to be 18 years at the latest. |
Devices with less than 10% of somatropin remaining will be wasted. | Likely appropriate. The clinical expert consulted by CADTH noted that some drug wastage remaining in the cartridge is expected, although the exact amount of product wastage is unknown. Patients are typically dispensed 3 months’ worth of medication at a time, regardless of adherence. |
The various somatropin brands in the model are assumed to have similar effectiveness and safety. A similar dose is assumed for all somatropin brands. | Appropriate. |
HV is assumed to be significantly correlated with treatment adherence. A study by Maggio et al. (2018) was used to estimate reduction in HV associated with different adherence levels. Similar impact was assumed with somatrogon and somatropin for a given adherence rate. | Appropriate. The clinical expert consulted by CADTH indicated that in patients with GHD, HV and final height is affected by treatment adherence. |
Adverse events were assumed to be similar for patients who received somatrogon and somatropin and, therefore, were not modelled. | Likely appropriate. The CADTH clinical review noted that in CP-4-006, a higher number of patients in the somatrogon treatment group experienced injection site pain, nasopharyngitis, pyrexia, and headache. Despite this, key safety issues with somatrogon were reported to be minimal. |
No difference in mortality with somatrogon in comparison with somatropin was assumed. | Appropriate. |
GHD = growth hormone deficiency; HV = height velocity.
CADTH Reanalyses of the Economic Evaluation
Base-Case Results
The CADTH base case was derived by making changes in model parameter values and assumptions, in consultation with a clinical expert. CADTH undertook the reanalyses outlined in Table 5 to address, where possible, the limitations with the sponsor’s submitted economic model. CADTH was unable to address the limitations related to the uncertainty with the magnitude of clinical benefit associated with weekly somatrogon injections compared with daily somatropin injections over the time horizon, issues regarding the applicability of the utility scores by HSDS used in the model, and the uncertainty in the market share distribution of various branded somatropin treatments and product size within brands.
Table 5: CADTH Revisions to the Submitted Economic Evaluation
Stepped analysis | Sponsor’s value or assumption | CADTH value or assumption |
|---|---|---|
Corrections to sponsor’s base case | ||
Distribution of somatropin costs | Somatropin costs were assumed to vary probabilistically. | Somatropin costs were assumed to be fixed, with the list price used in each model simulation. |
Changes to derive the CADTH base case | ||
1. Dosing for somatropin treatments | 0.24 mg/kg per week, varied probabilistically | 0.18 mg/kg per week, assumed to be fixed |
2. Comparative efficacy (i.e., height velocity) | Greater height velocity assumed for somatrogon compared to somatropin over the years of treatment | Equal height velocities for both treatment groups (values for somatrogon applied to somatropin) |
CADTH base case | Reanalyses 1 + 2 | |
The results for the stepwise analyses can be found in Table 6. Somatrogon was dominant in all stepped analyses except when the dose assumed with somatropin was reduced. Results from the probabilistic CADTH base case found that somatrogon was associated with incremental costs of $27,005 and an incremental benefit of 0.25 QALYs compared with somatropin over the lifetime time horizon. The ICER for somatrogon versus somatropin was $107,714 per QALY gained. Somatrogon had a 1% probability of being cost-effective at a willingness-to-pay threshold of $50,000 per QALY. CADTH also noted that the cost-effectiveness of somatrogon varied by age category, with the deterministic ICER ranging from $78,029 to $127,276 per QALY gained, depending on the age of treatment initiation (refer to Table 12 in Appendix 4).
Table 6: Summary of the Stepped Analysis of the CADTH Reanalysis Results
Stepped analysis | Drug | Total costs ($) | Total QALYs | ICER ($/QALYs) |
|---|---|---|---|---|
Sponsor’s base case | Somatropin | 153,567 | 7.44 | Reference |
Somatrogon | 147,198 | 7.72 | Dominant | |
Sponsor’s corrected base case | Somatropin | 153,710 | 7.44 | Reference |
Somatrogon | 147,056 | 7.74 | Dominant | |
CADTH reanalysis 1 | Somatropin | 119,221 | 7.45 | Reference |
Somatrogon | 147,106 | 7.72 | 114,709 | |
CADTH reanalysis 2 | Somatropin | 154,917 | 7.47 | Reference |
Somatrogon | 147,167 | 7.72 | Dominant | |
CADTH base case (reanalyses 1 and 2) | Somatropin | 120,129 | 7.47 | Reference |
Somatrogon | 147,134 | 7.72 | 107,714 |
ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year.
Scenario Analysis Results
CADTH undertook a series of price reduction analyses on the price of somatrogon based on the CADTH base case. Proportional price reductions were applied to both sizes of somatrogon available. In the CADTH base case, somatrogon may be cost-effective at a willingness-to-pay threshold of $50,000 per QALY with a price reduction of 11%. The small price reduction needed was likely attributed to drug acquisition costs as they are the sole source of incremental costs in the model.
Table 7: CADTH Price Reduction Analyses
Analysis | ICERs for somatrogon vs. somatropin ($/QALY) | |
|---|---|---|
Price reduction | Sponsor base case | CADTH reanalysis |
No price reduction | Dominant | 107,714 |
10% | Dominant | 51,431 |
11% | Dominant | 47,269 |
20% | Dominant | Dominant |
ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year; vs. = versus.
CADTH also undertook several scenario analyses to determine the impact of alternative assumptions on the cost-effectiveness of somatrogon versus somatropin. The analyses included the following:
examining the cost-effectiveness of somatrogon compared to Genotropin only (i.e., lowest priced option)
assuming a somatropin dosage of 0.24 mg/kg per week (or 0.034 mg/kg per day)
applying the assumption that there is no drug wastage of any kind (i.e., no last-dose wastage, no device-setting wastage, no storage wastage, no preparation wastage, and no adherence wastage)
removing the incremental benefit associated with reduced administration frequency with somatrogon
assuming an equal distribution of large and small pen sizes will be administered to patients who receive somatrogon
applying the assumption that there is a 10% treatment discontinuation rate after the first year of treatment
assuming equal treatment adherence for both somatrogon and somatropin based on the adherence values for somatropin
assuming treatment adherence of 80% for somatropin in years 4 onwards and 90% for somatrogon in years 2 onwards (i.e., improved adherence in the long-term with somatrogon).
The results of these analyses are presented in Table 13 in Appendix 4. Results were most sensitive to the scenario that only considered a comparison to Genotropin (ICER = $186,120 per QALY gained) and the assumption that there is no utility benefit associated with weekly injections of somatrogon (ICER = $368,381 per QALY gained). Assuming equal treatment adherence for both treatments based on values for somatropin resulted in an ICER of $131,880 per QALY gained. Higher adherence in years 4-plus and in years 2-plus for somatropin and somatrogon, respectively, resulted in an ICER of $104,319 per QALY gained.
Issues for Consideration
Potential off-label use of somatrogon: The sponsor is requesting that somatrogon be reimbursed for the long-term treatment of pediatric patients who have GHD. However, it is possible that somatrogon will be used off label for other conditions that other somatropin products are also indicated for, such as idiopathic short stature, SHOX-containing gene deficiency, small for gestational age, and in children with growth failure due to chronic renal failure. The potential for off-label use of somatrogon may be associated with considerable costs to the drug plans. However, it should be noted that the possibility of off-label use is not unique to somatrogon and applies to the other available somatropin products on the market in Canada.
Stopping rules: In practice, treatment with GH therapies is expected to be continued until target height has been achieved. In the submitted model, patients on treatment were not assumed to achieve their target height until age 18 and were assumed to be on treatment until then. No other stopping rules were assessed by the sponsor nor by CADTH.
Use in patients beyond the trial population is uncertain: The sponsor’s submitted economic evaluation considered pediatric patients between the ages of 3 years and 12 years who were treatment naive. The cost-effectiveness in patients younger than 3 years of age, or initiation in patients above the age of 12, including adults, has not been assessed in the sponsor’s submission and could not be addressed by CADTH. Such patients may be treated in clinical practice. Further, the cost-effectiveness of somatrogon in patients with prior exposure to somatropin has not been assessed in the sponsor’s pharmacoeconomic evaluation and could not be addressed by CADTH.
Overall Conclusions
The clinical evidence submitted by the sponsor demonstrated that in pediatric patients with GHD, treatment with somatrogon was noninferior to Genotropin based on the clinically meaningful treatment difference in the primary end point of trial CP-4-006 (i.e., a ≥ –1.8 cm per year change in height was met). ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| for the long-term treatment of growth failure due to an inadequate secretion of endogenous GH. The clinical efficacy of somatrogon beyond 12 months is unknown.
CADTH identified several major limitations with the submitted economic evaluation beyond those related to the clinical evidence. These included issues related to the uncertainty in the magnitude of benefit associated with a weekly injection of somatrogon in comparison with daily injections of somatropin, an overestimation of the dose for all somatropin treatments, uncertainty in the market share distribution for the various brands of somatrogon, uncertainty with the utility values by HSDS applied in the model, and uncertainty in the relative treatment adherence with somatrogon in comparison with somatropin.
CADTH’s base-case reanalysis included assuming equal HVs for both somatrogon and somatropin, and revising the dosage for all somatropin products to align with the dosage most commonly used in Canadian clinical practice. In the CADTH base case, somatrogon was associated with an ICER of $107,714 per QALY gained (incremental costs of $27,005 and an incremental benefit of 0.25 QALYs) compared with somatropin. A price reduction of at least 11% would be required for somatrogon to be considered cost-effective at a willingness-to-pay threshold of $50,000 per QALY gained.
The cost-effectiveness of somatrogon is primarily driven by drug acquisition costs related to somatropin, which is affected by particular brands, dose and wastage assumptions, and the magnitude of benefit related to weekly rather than daily injections associated with somatrogon.
CADTH was unable to address important limitations related to the magnitude of the quality-of-life benefit associated with weekly somatrogon injections compared with daily somatropin injections over the time horizon, generalizability of utility scores by HSDS identified by the sponsor to the target population, and the uncertainty in the market share distribution of the various branded somatropin treatments.
References
1.Ngenla (somatrogon): 24 mg/1.2 mL (20 mg/mL), 60 mg/1.2 mL (50 mg/mL), prefilled pen for subcutaneous injection [product monograph]. Kirkland (QC): Pfizer Canada ULC; 2021 Oct 26.
2.Pharmacoeconomic evaluation [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Ngenla (somatrogon), 24 mg/1.2 mL (20 mg/mL), 60 mg/1.2 mL (50 mg/mL), prefilled pen for subcutaneous injection. Kirkland (QC): Pfizer Canada ULC; 2021 Jun 3.
3.Clinical Study Report: CP-4-006 (C0311009). A phase 3, open-label, randomized, multicenter, 12 months, efficacy and safety study of weekly MOD-4023 compared to daily Genotropin therapy in pre-pubertal children with growth hormone deficiency [internal sponsor's report]. New York (NY): Pfizer Inc; 2020 Aug 24.
4.Ranke MB, Lindberg A. Observed and predicted growth responses in prepubertal children with growth disorders: guidance of growth hormone treatment by empirical variables. J Clin Endocrinol Metab. 2010;95(3):1229-1237. PubMed
5.Maggio MC, Vergara B, Porcelli P, Corsello G. Improvement of treatment adherence with growth hormone by easypod™ device: experience of an Italian centre. Ital J Pediatr. 2018;44(1):113. PubMed
6.Christensen TL, Djurhuus CB, Clayton P, Christiansen JS. An evaluation of the relationship between adult height and health-related quality of life in the general UK population. Clin Endocrinol (Oxf). 2007;67(3):407-412. PubMed
7.Boye KS, Matza LS, Walter KN, Van Brunt K, Palsgrove AC, Tynan A. Utilities and disutilities for attributes of injectable treatments for type 2 diabetes. Eur J Health Econ. 2011;12(3):219-230. PubMed
8.Schedule of benefits for laboratory services: effective July 1, 2020. Toronto (ON): Ontario Ministry of Health; 2020: https://www.health.gov.on.ca/en/pro/programs/ohip/sob/lab/lab_mn2020.pdf. Accessed 2021 Oct 5.
9.Schedule of benefits for physician services under the Health Insurance Act: effective April 1, 2020. Toronto (ON): Ontario Ministry of Health; 2020: https://www.health.gov.on.ca/en/pro/programs/ohip/sob/physserv/sob_master20200306.pdf. Accessed 2021 Oct 5.
10.Hôpital Notre-Dame. Non-OHIP insured tests. 2017; http://www.ndh.on.ca/wp-content/uploads/2018/01/uninsured-lab-tests.pdf. Accessed 2021 Oct 5.
11.Deal C, Kirsch S, Chanoine J-P, et al. Growth hormone treatment of Canadian children: results from the GeNeSIS phase IV prospective observational study. CMAJ Open. 2018;6(3):E372-E383. PubMed
12.Joshi AV, Munro V, Russell MW. Cost-utility of somatropin (rDNA origin) in the treatment of growth hormone deficiency in children. Curr Med Res Opin. 2006;22(2):351-357. PubMed
13.Exceptional Access Program (EAP). Toronto (ON): Ontario Ministry of Health; Ontario Ministry of Long-Term Care; 2021: https://www.health.gov.on.ca/en/pro/programs/drugs/odbf/odbf_except_access.aspx. Accessed 2021 Oct 5.
14.Saskatchewan Drug Plan: search formulary. 2021; https://formulary.drugplan.ehealthsask.ca/SearchFormulary. Accessed 2021 Oct 5.
15.Budget Impact Analysis [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Ngenla (somatrogon), 24 mg/1.2 mL (20 mg/mL), 60 mg/1.2 mL (50 mg/mL), prefilled pen for subcutaneous injection. Kirkland (QC) Pfizer Canada ULC; 2021 Jun 3.
16.Stochholm K, Gravholt CH, Laursen T, et al. Incidence of GH deficiency - a nationwide study. Eur J Endocrinol. 2006;155(1):61-71. PubMed
Appendix 1: Cost Comparison Table
Note that this appendix has not been copy-edited.
The comparators presented in Table 8 have been deemed to be appropriate based on feedback from clinical expert(s). Comparators may be recommended (appropriate) practice or actual practice. Existing Product Listing Agreements are not reflected in the table and as such, the table may not represent the actual costs to public drug plans.
Table 8: CADTH Cost Comparison Table for Human Growth Hormones to Treat Growth Hormone Deficiency
Treatment | Strength / concentration | Form | Price ($) | Recommended dosage | Daily cost ($) | Annual cost ($) |
|---|---|---|---|---|---|---|
Somatrogon (Ngenla) | 24 mg/1.2 mL (20 mg/mL) | Pre-filled pen | 345.8400a | 0.66 mg/kg weekly | 26.53 | 9,684 |
60 mg/1.2 mL (50 mg/mL) | 864.6000a | |||||
Daily growth hormone treatment | ||||||
Somatropin (Genotropin) | 5.3 mg 12 mg | Pre-filled pen | 147.8700 334.8000 | 0.16 to 0.24 mg/kg per week, in 6 to 7 doses | 12.56 to 18.64 | 4,584 to 6,802 |
0.6 mg 0.8 mg 1.0 mg 1.2 mg 1.4 mg 1.6 mg 1.8 mg 2.0 mg | Single use pre-filled syringe | 16.7400 22.3200 27.9000 33.4800 39.0600 44.6400 50.2200 55.8000 | 12.30 to 18.42 | 4,492 to 6,724 | ||
Somatropin (Humatrope) | 5 mg | Vial | 233.3500 | 0.18 to 0.30 mg/kg per week in 3 to 7 doses | 19.82 to 65.21 | 8,634 to 14,234 |
6 mg / 3.15 mL 12 mg / 3.15 mL 24 mg / 3.15 mL | Cartridge | 288.4200 576.8400 1,153.6800 | 24.50 to 40.30 | 8,941 to 14,709 | ||
Somatropin (Norditropin) | 5 mg / 1.5 mL 10 mg / 1.5 mL 15 mg / 1.5 mL | Pre-filled pen | 194.7000 389.4000 584.1000 | Up to 0.3 mg/kg per week, divided in daily doses | Up to 33.07 | Up to 12,071 |
Somatropin (Nutropin AQ) | 5 mg / 2 mL 10 mg / 2 mL 20 mg / 2mL | Cartridge | 203.7100 407.4200 814.8400 | Up to 0.3 mg/kg per week, divided in daily doses | Up to 34.60 | Up to $12,630 |
Somatropin (Omnitrope) | 5 mg 10 mg | Vial | 155.8000 311.6000 | 0.18 to 0.25 mg/kg per week | 15.79 to 21.77 | 5,765 to 7,946 |
15 mg / 1.5 mL | Cartridge | 467.4000b | 16.65 to 21.77 | 6,076 to 7,945 | ||
Somatropin (Saizen) | 3.33 mg 5 mg 8.8 mg | Vial | 149.2500 224.0500 358.4700 | 0.20 to 0.27 mg/kg per week in 3 to 7 doses | 25.17 to 33.76 | 9,186 to 12,322 |
6 mg (5.83 mg/mL) 12 mg (8 mg/mL) 20 mg (8 mg/mL) | Cartridge | 268.8300 537.6600 896.1000 | 24.99 to 33.81 | 9,122 to 12,342 | ||
Note: All prices are from the Ontario Exceptional Access Program13 (accessed July 2021), unless otherwise indicated, and do not include dispensing fees. Weight-based doses use an average weight of 19.30 kg as per CP-4-006. Annual costs based on number vials, pens, cartridges, or syringes needed in a calendar year rounded up to the nearest whole vial. Daily costs based on annual costs divided by 365. No other wastage costs have been considered.
aSponsor’s submitted price.2
bSaskatchewan Formulary.14
Appendix 2: Submission Quality
Note that this appendix has not been copy-edited.
Description | Yes/No | Comments |
|---|---|---|
Population is relevant, with no critical intervention missing, and no relevant outcome missing | Yes | No comment |
Model has been adequately programmed and has sufficient face validity | Yes | No comment |
Model structure is adequate for decision problem | Yes | No comment |
Data incorporation into the model has been done adequately (e.g., parameters for probabilistic analysis) | No | Sponsor included parameter distributions around drug costs which is not appropriate. Such costs should be fixed. |
Parameter and structural uncertainty were adequately assessed; analyses were adequate to inform the decision problem | Yes | No comment |
The submission was well organized and complete; the information was easy to locate (clear and transparent reporting; technical documentation available in enough details) | Yes | No comment |
Appendix 3: Additional Information on the Submitted Economic Evaluation
Note that this appendix has not been copy-edited.
Table 10: Disaggregated Summary of Sponsor’s Economic Evaluation Results by Patient Age of Treatment Initiation (Deterministic)
Parameter | Proportion of patients by age of treatment initiation | Somatrogon | Somatropin (reference) | Incremental |
|---|---|---|---|---|
Discounted LYs | ||||
Totala | NA | 9.34 | 9.34 | 0 |
Age 3 | 2.4% | 13.44 | 13.44 | 0 |
Age 4 | 4.8% | 12.64 | 12.64 | 0 |
Age 5 | 7.9% | 11.82 | 11.82 | 0 |
Age 6 | 11.3% | 10.99 | 10.99 | 0 |
Age 7 | 13.7% | 10.15 | 10.15 | 0 |
Age 8 | 18.6% | 9.29 | 9.29 | 0 |
Age 9 | 16.4% | 8.42 | 8.42 | 0 |
Age 10 | 12.4% | 7.54 | 7.54 | 0 |
Age 11 | 8.0% | 6.65 | 6.65 | 0 |
Age 12 | 4.4% | 5.74 | 5.74 | 0 |
Discounted QALYs | ||||
Totala | NA | 7.72 | 7.44 | 0.28 |
Age 3 | 2.4% | 11.07 | 10.61 | 0.46 |
Age 4 | 4.8% | 10.50 | 10.12 | 0.38 |
Age 5 | 7.9% | 9.80 | 9.45 | 0.35 |
Age 6 | 11.3% | 9.10 | 8.80 | 0.31 |
Age 7 | 13.7% | 8.42 | 8.15 | 0.27 |
Age 8 | 18.6% | 7.70 | 7.42 | 0.28 |
Age 9 | 16.4% | 6.98 | 6.74 | 0.24 |
Age 10 | 12.4% | 6.22 | 6.00 | 0.22 |
Age 11 | 8.0% | 5.42 | 5.17 | 0.25 |
Age 12 | 4.4% | 4.56 | 4.32 | 0.24 |
Discounted costs ($) | ||||
Totala | NA | 147,198 | 153,567 | –6,369 |
Age 3 | 2.4% | 176,668 | 179,975 | –3,307 |
Age 4 | 4.8% | 175,143 | 179,661 | –4,518 |
Age 5 | 7.9% | 169,193 | 174,538 | –5,345 |
Age 6 | 11.3% | 162,428 | 169,446 | –7,018 |
Age 7 | 13.7% | 156,453 | 165,353 | –8,900 |
Age 8 | 18.6% | 148,931 | 154,988 | –6,056 |
Age 9 | 16.4% | 141,587 | 148,400 | –6,814 |
Age 10 | 12.4% | 130,967 | 137,124 | –6,158 |
Age 11 | 8.0% | 118,410 | 123,252 | –4,842 |
Age 12 | 4.4% | 104,220 | 109,609 | –5,389 |
ICER ($/QALY) | Dominantb | |||
ICER = incremental cost-effectiveness ratio; LY = life-year; NA = not applicable; QALY = quality-adjusted life-year.
aTotal weighted by proportion of patients within age distribution.
bSponsor’s probabilistic base case.
Appendix 4: Additional Details on the CADTH Reanalyses and Sensitivity Analyses of the Economic Evaluation
Note that this appendix has not been copy-edited.
Detailed Results of CADTH Base Case
Table 11: Disaggregated Summary of CADTH’s Economic Evaluation Results (Deterministic)
Treatment | Component | Value | Incremental (vs. reference) |
|---|---|---|---|
Discounted LYs | |||
Somatropin | Total | 9.34 | NA |
Somatrogon | Total | 9.34 | 0 |
Discounted QALYs | |||
Somatropin | Total | 7.47 | NA |
Somatrogon | Total | 7.72 | 0.25 |
Discounted costs ($) | |||
Somatropin | Drug acquisition | 90,618 | NA |
Monitoring | 4,697 | NA | |
Wastage | 24,705 | NA | |
Last-dose wastage | 26 | NA | |
Device-setting wastage | 2,269 | NA | |
Storage wastage | 2,243 | NA | |
Preparation wastage | 593 | NA | |
Adherence wastage | 19,575 | NA | |
Total | 120,020 | NA | |
Somatrogon | Drug acquisition | 121,020 | 30,402 |
Monitoring | 4,697 | 0 | |
Wastage | 21,414 | –3,290 | |
Last-dose wastage | 99 | 73 | |
Device-setting wastage | 1,174 | –1,095 | |
Storage wastage | 932 | –1,310 | |
Preparation wastage | 0 | –593 | |
Adherence wastage | 19,209 | –365 | |
Total | 147,131 | 27,112 | |
ICER vs. reference ($/QALY) | |||
Somatropin | Reference | ||
Somatrogon | 107,714 | ||
ICER = incremental cost-effectiveness ratio; LY = life-year; NA = not applicable; QALY = quality-adjusted life-year; vs. = versus.
aCADTH’s probabilistic ICER.
Table 12: Disaggregated Summary of CADTH’s Economic Evaluation Results by Age of Treatment Initiation (Deterministic)
Parameter | Proportion of patients by age of treatment initiation | Somatrogon | Somatropin (reference) | Incremental |
|---|---|---|---|---|
Discounted LYs | ||||
Totala (weighted by proportion of patients within age distribution) | NA | 9.34 | 9.34 | 0 |
Age 3 | 2.4% | 13.44 | 13.44 | 0 |
Age 4 | 4.8% | 12.64 | 12.64 | 0 |
Age 5 | 7.9% | 11.82 | 11.82 | 0 |
Age 6 | 11.3% | 10.99 | 10.99 | 0 |
Age 7 | 13.7% | 10.15 | 10.15 | 0 |
Age 8 | 18.6% | 9.29 | 9.29 | 0 |
Age 9 | 16.4% | 8.42 | 8.42 | 0 |
Age 10 | 12.4% | 7.54 | 7.54 | 0 |
Age 11 | 8.0% | 6.65 | 6.65 | 0 |
Age 12 | 4.4% | 5.74 | 5.74 | 0 |
Discounted QALYs | ||||
Totala (weighted by proportion of patients within age distribution) | NA | 7.72 | 7.47 | 0.25 |
Age 3 | 2.4% | 11.07 | 10.65 | 0.42 |
Age 4 | 4.8% | 10.50 | 10.14 | 0.36 |
Age 5 | 7.9% | 9.80 | 9.47 | 0.33 |
Age 6 | 11.3% | 9.10 | 8.81 | 0.29 |
Age 7 | 13.7% | 8.42 | 8.16 | 0.26 |
Age 8 | 18.6% | 7.70 | 7.46 | 0.24 |
Age 9 | 16.4% | 6.98 | 6.78 | 0.21 |
Age 10 | 12.4% | 6.22 | 6.03 | 0.19 |
Age 11 | 8.0% | 5.42 | 5.22 | 0.20 |
Age 12 | 4.4% | 4.56 | 4.37 | 0.19 |
Discounted costs ($) | ||||
Totala (weighted by proportion of patients within age distribution) | NA | 120,020 | 147,131 | 33,050 |
Age 3 | 2.4% | 176,586 | 143,537 | 33,050 |
Age 4 | 4.8% | 175,063 | 142,475 | 32,588 |
Age 5 | 7.9% | 169,115 | 137,528 | 31,587 |
Age 6 | 11.3% | 162,353 | 132,789 | 29,565 |
Age 7 | 13.7% | 156,381 | 128,704 | 27,677 |
Age 8 | 18.6% | 148,864 | 121,593 | 27,270 |
Age 9 | 16.4% | 141,523 | 115,624 | 25,899 |
Age 10 | 12.4% | 130,909 | 106,281 | 24,628 |
Age 11 | 8.0% | 118,359 | 95,082 | 23,277 |
Age 12 | 4.4% | 104,176 | 84,661 | 19,516 |
ICER ($/QALY) | 107,714b | |||
ICER = incremental cost-effectiveness ratio; LY = life-year; QALY = quality-adjusted life-year.
aTotal weighted by proportion of patients within age distribution.
bCADTH’s probabilistic base case.
Scenario Analyses
Table 13: Scenario Analyses for Somatrogon vs. Somatropin
Scenario analysis | Drug | Total costs ($) | Total QALYs | ICER ($/QALY) |
|---|---|---|---|---|
CADTH base case | Somatropina | 120,129 | 7.47 | Reference |
Somatrogon | 147,134 | 7.72 | 107,714 | |
Somatrogon vs. Genotropin (least expensive somatropin only) | Somatropina | 100,344 | 7.47 | Reference |
Somatrogon | 147,057 | 7.72 | 186,120 | |
Dosing = 0.24 mg/kg per week | Somatropina | 154,785 | 7.47 | Dominant |
Somatrogona | 147,009 | 7.73 | Reference | |
No wastage | Somatropina | 95,790 | 7.47 | Reference |
Somatrogon | 126,382 | 7.72 | 123,538 | |
Utility benefit associated with weekly injections set to 0 | Somatropina | 120,130 | 7.47 | Reference |
Somatrogon | 147,124 | 7.54 | 368,381 | |
Equal distribution of sizes of somatrogon pen | Somatropina | 120,059 | 7.47 | Reference |
Somatrogon | 146,491 | 7.72 | 107,528 | |
Treatment discontinuation rate of 10% | Somatropina | 134,430 | 7.49 | Reference |
Somatrogon | 164,877 | 7.77 | 110,863 | |
Equal treatment adherence rates for somatrogon and somatropin | Somatropina | 120,034 | 7.47 | Reference |
Somatrogon | 143,698 | 7.65 | 131,880 | |
Assume 80% adherence in year 4+ with somatropin and 90% adherence in year 2 onwards with somatrogon based on clinical expectations | Somatropina | 119,876 | 7.47 | Reference |
Somatrogon | 148,610 | 7.74 | 104,319 |
ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year; vs. = versus.
aReference product is the least costly comparator.
Appendix 5: Submitted Budget Impact Analysis and CADTH Appraisal
Note that this appendix has not been copy-edited.
Table 14: Summary of Key Take-Aways
Key take-aways of the budget impact analysis |
|---|
|
Summary of Sponsor’s Budget Impact Analysis
The sponsor assessed the budget impact of the introduction of somatrogon compared with somatropin for pediatric patients with GHD, from the perspective of the public drug plan in the Canadian setting (excluding Quebec) over a 3-year time horizon.15 The sponsor’s submission only considered drug acquisition costs. In the reference scenario, the sponsor assumed that patients would be eligible to receive various brands of somatropin (i.e., Genotropin, Humatrope, Norditropin NordiFlex, Nutropin AQ, Omnitrope, Saizen). In the new drug scenario, somatrogon was assumed to displace market shares of the various brands of somatropin.15
The sponsor estimated the eligible population size using an epidemiological approach by leveraging data from multiple sources in the literature and assumptions based on internal data. Specifically, the eligible population for treatment was derived starting with an estimate of the Canadian population less than 18 years of age. Based on this subpopulation, the sponsor applied an estimated prevalence of GHD diagnosed in Canadian children, which was assumed to be 22.23 per 100,000 among individuals 0 to 19 years of age in 2020,15 and based on an incidence rate of 2.15 per 100,000, 10% of cases were assumed to be newly diagnosed cases in a given year.16 Following this step, the sponsor assumed that 75% of children between ages 3 and 16, and 35% of children at the age of 17 years were assumed to be on GHD treatment, respectively, based on internal data sources. The proportion of children with GHD or GH insufficiency covered by public drug programs was further assumed to be 52%.15
The sponsor’s BIA also included the following key assumptions:
The market share distributions in the reference and new drug scenarios were derived from a claims database that includes claims for all GH indications. Based on these data, the sponsor assumed that the market share distribution across all GH indications are applicable to GHD in children.15
In the reference scenario, the sponsor projected that market shares of short-acting GH brands would change over the 3-year time horizon, such that 20% of the market share for Humatrope from year 1 would be displaced equally between Genotropin and Norditropin Nordiflex in year 2 and year 3. The sponsor further assumed that another new long-acting growth hormone (LAGH) would be introduced to the market in year 2, such that it would capture 19% of the overall market share in year 2 and 30% in year 3.15
In the new drug scenario, the sponsor anticipated that somatrogon would capture 19% of the market in year 1. The sponsor further assumed that another new LAGH would be introduced to the market in year 2 such that overall market share of the 2 LAGHs would be 30% (split as 85% somatrogon and 15% for the other LAGH). In year 3, it was anticipated that the 2 LAGHs would capture 50% of the overall market share relative to other GH treatment (split as 64% somatrogon and 36% for the other LAGH).15
Key inputs to the BIA are documented in Table 15:
Table 15: Summary of Key Model Parameters
Parameter | Sponsor’s estimate (reported as baseline/year 1/year 2/year 3 if appropriate) |
|---|---|
Target population | |
Population by age Total 0 to < 18 0 to 2 3 to 16 17 to < 18 | 5,760,700 912,500 4,507,400 340,800 |
Prevalence of GHD diagnoses in children | 22.23 per 100,000 |
Incidence of GHD in children | 2.15 per 100,000 |
Percentage of children on GH treatment by age group: 0 to 2 3 to 16 17 to < 18 | 0% 75% 34% |
Percentage of incident patients/ total patients treated | 10% |
Proportion of children with CHD covered by public drug programs | 52% |
Number of patients eligible for drug under review | 404 / 412 / 420 / 427 |
Market uptake (3 years) | |
Uptake (reference scenario) Somatrogon Genotropin Humatrope Norditropin Nordiflex Nutropin AQ Nuspin Omnitrope Saizen | 0% / 0% / 0% 6.8% / 9.4% / 10.8% 48.3% / 31.3% / 21.7% 6.4% / 9.1% / 10.5% 3.2% / 2.6% / 2.3% 8.1% / 6.6% / 5.7% 27.2% / 22.0% / 19.0% |
Uptake (new drug scenario) Somatrogon Genotropin Humatrope Norditropin Nordiflex Nutropin AQ Nuspin Omnitrope Saizen | 19% / 31% / 50% 5.5% / 8.1% / 7.7% 39.1% / 27.1% / 15.5% 5.2% / 7.8% / 7.5% 2.6% / 2.3% / 1.6% 6.6% / 5.7% / 4.1% 22.0% / 19.0% / 13.6% |
Cost of treatment (per patient) | |
Cost of treatment over 28 days Somatrogon Genotropin Humatropea Norditropin Nordiflex Nutropin AQ Nuspin Omnitrope Saizen | $734.22 $512.62 $857.50 $715.47 $748.58 $572.52 $823.23 |
GH = growth hormone; GHD = growth hormone deficiency.
aBased on the 24 mg cartridge.
Summary of the Sponsor’s Budget Impact Analysis Results
Results of the sponsor’s base-case analysis under the drug plan perspective estimated that the introduction of somatrogon in patients with GHD would result in a cost savings of $9,984 in year 1, an incremental budget impact of $6,003 in year 2 and $28,717 in year 3, for a total budget impact of $24,736 over the 3-year time horizon.
CADTH Appraisal of the Sponsor’s Budget Impact Analysis
CADTH identified several key limitations to the sponsor’s analysis that have notable implications on the results of the BIA:
Dosing for somatropin does not align with dose commonly used in Canadian clinical practice: In the submitted BIA, the sponsor assumed that the average administered dose for all somatropin products was 0.034 mg/kg per day (or 0.24 mg/kg per week). The clinical expert consulted by CADTH for this review indicated that the average dose applied in the sponsor’s base case was higher than the average dose commonly used in Canadian clinical practice. In Canadian clinical practice, a lower daily dose of 0.0257 mg/kg (or 0.18 mg/kg per week) is typically prescribed and administered. This is aligned with the findings from a study by Deal et al., which assessed GH treatment patterns in Canadian children.11 The use of a higher dose led to an overestimation of the costs associated with somatropin, underestimating the total budget impact associated with the reimbursement of somatrogon.
CADTH addressed this limitation by changing the dose for all somatropin products to 0.0257 mg/kg per day in the base-case analysis.
Adjustment of drug costs by patient compliance rates likely underestimated total drug costs: In the submitted BIA, the sponsor assumed that the average treatment compliance to daily short-acting growth hormones (SAGHs) was 85%, and 90% for LAGHs. These rates were used to adjust drug costs to reflect patient compliance. While patient compliance may not be 100%, drug plans are likely to incur the costs of treatment with GHs regardless of whether patients use the drug or not and ignores the potential for drug wastage. This underestimated total drug costs associated with both SAGHs and LAGHs.
CADTH addressed this limitation by changing the average treatment compliance rate to 100% in order to capture all drug acquisition costs. Also of note, the sponsor’s BIA did not account for other forms of wastage that were captured in the submitted pharmacoeconomic model. This introduces some uncertainty with the sponsor’s submitted BIA.
The estimated population eligible and assumed to be treated is likely underestimated: The sponsor assumed that 75% of the indicated population between the ages of 3 and 16 would be on a GH based on various assumptions and published literature. The clinical expert consulted by CADTH noted that the proportion of children with GHD in this age group was underestimated and likely to be higher for the given indication. Additionally, the sponsor assumed that 34% of 17-year-olds with the indication (who were still eligible for treatment) were assumed to be on GH treatment based on the sponsor’s internal data source. The clinical expert consulted by CADTH indicated that this proportion was likely underestimated and that nearly half of patients eligible this age will be prescribed GHD treatment. Finally, the sponsor assumed that the proportion of children with GHD assumed to be covered by public drug programs was 52% based on the sponsor’s internal data. The clinical expert consulted by CADTH indicated that this proportion was also underestimated and should be higher as all provinces and territories publicly fund GH treatments for children with GHD.
CADTH addressed this limitation by revising the proportion of children receiving GH treatments between ages 3 and 16 years to 90%; revised the proportion of children aged 17 years receiving GH treatments to 50%; and revised the proportion of patients with GHD covered by public drug programs to 100%.
The market share distribution of the various brands of somatropin treatments in the reference scenario is uncertain: The market share distribution of the various brands of somatropin products in the reference scenario of the sponsor’s submitted BIA was obtained from the 2020 IQVIA Canada Public Claims for Growth Hormone Products. The database includes claims for all GH indications, and they are not specific to the indication of pediatric patients with GHD or GH insufficiency. The clinical expert consulted by CADTH indicated that based on their experience in clinical practice, the market share distribution in the reference scenario did not align with their expectations of the distribution of somatropin products for the indication under review. Further, the sponsor assumed various trends for the comparator treatments in the reference scenario, over the 3-year time horizon. Specifically, it was assumed that Humatrope would lose 20% of its market shares equally between Genotropin and Norditropin Nordiflex in year 2 and year 3. However, there is uncertainty around the projected trends between various somatropin products in the reference scenario. Given differences in costs between the various brands of somatropin, the budget impact is likely to vary should the distribution vary substantially from that assumed by the sponsor in their base case.
CADTH could not address this limitation.
The inclusion of a second LAGH that is not listed or does not have a positive listing recommendation from the CADTH Canadian Drug Expert Committee is inappropriate: The sponsor assumed that a second LAGH would be introduced in the market, capturing 19% of the overall market share in year 2, and 30% in year 3 of the reference scenario, as well as market share in the new drug scenario. The comparators included in the BIA should be aligned with the submitted pharmacoeconomic model and should reflect drugs that are either presently listed by CADTH participating drug plans or be drugs that have received positive listing recommendations from the CADTH Canadian Drug Expert Committee. As there are no other LAGHs that meet these criteria, the inclusion of this hypothetical LAGH is inappropriate. The inclusion of a hypothetical LAGH entering the market underestimated the incremental budget impact of somatrogon in the sponsor’s base case.
CADTH addressed this limitation by removing the additional LAGH from the CADTH base-case analysis.
The anticipated uptake of somatrogon in the new drug scenario is uncertain: In the sponsor’s submitted BIA, somatrogon was forecasted to capture 19% of the GH market shares in year 1. In year 2, the sponsor assumed that a second LAGH would also be introduced, such that both LAGHs (i.e., somatrogon and other LAGH) would account for 30% of the overall market share of GH products, with somatrogon comprising the majority (85%) of shares between the 2 LAGHs. In year 3, the sponsor anticipated that both LAGHs would comprise 50% of the overall market share. The clinical expert consulted by CADTH noted that for the indication of interest, it was reasonable to assume that somatrogon would capture market share from various somatropin products, but importantly, noted that a more gradual than rapid uptake was likely to occur among patients, and therefore it is unlikely that somatrogon will capture greater than 50% of the overall market share by year 3. However, the expert did note that 80% of patients on GH were likely to switch to a LAGH product like somatrogon eventually. Additionally, the clinical expert anticipated the products which somatrogon would displace to be uncertain. It was anticipated that somatrogon may either displace all comparator treatments proportionally, or compete more with Norditropin Nordiflex or Saizen, the commonly used treatments in their practice. However, this was noted to be an area of uncertainty.
CADTH conducted a scenario analysis where somatrogon captured a greater market share, with 50% market share in year 2 and 80% market share in year 3.
CADTH Reanalyses of the Budget Impact Analysis
A table noting the changes made to the sponsor’s BIA as part of the CADTH reanalysis is available in Table 16.
Table 16: CADTH Revisions to the Submitted Budget Impact Analysis
Stepped analysis | Sponsor’s value or assumption | CADTH value or assumption |
|---|---|---|
Corrections to sponsor’s base case | ||
None | — | — |
Changes to derive the CADTH base case | ||
1. Daily dose of somatropin | • 0.034 mg/kg per day (or 0.24 mg/kg per week) | • 0.0257 mg/kg per day (or 0.18 mg/kg per week) |
2. Treatment compliance | • Average treatment compliance to daily SAGH = 85% • Average treatment compliance to daily LAGH = 90% | • Average treatment compliance to daily SAGH = 100% • Average treatment compliance to daily LAGH = 100% |
3. Market share estimates in the reference and new drug scenario | • Included additional LAGH product in the reference and new drug scenario in Years 2 and 3 | • Excluded additional LAGH product entirely from reference and new drug scenarios |
4. Population size |
|
|
CADTH base case | Reanalyses 1 + 2 + 3 + 4 | |
LAGH = long-acting growth hormone; SAGH = short-acting growth hormone.
aChanges to derive the CADTH base case under the drug plan perspective.
Applying the changes in Table 16 resulted in an increase in the budget impact under the drug plan perspective to $1,965,211 over the 3-year time horizon. The results of the CADTH stepwise reanalyses are presented in summary format in Table 17 and a more detailed breakdown is presented in Table 18.
Table 17: Summary of the CADTH Reanalyses of the Budget Impact Analysis
Stepped analysis | 3-year total |
|---|---|
Submitted base case | $24,736 |
CADTH reanalysis 1 | $453,969 |
CADTH reanalysis 2 | –$87,635 |
CADTH reanalysis 3 | $78,181 |
CADTH reanalysis 4 | $56,753 |
CADTH base case | $1,965,211 |
Table 18: Detailed Breakdown of the CADTH Reanalyses of the Budget Impact Analysis
Stepped analysis | Scenario | Year 0 (current situation) | Year 1 | Year 2 | Year 3 | 3-year total |
|---|---|---|---|---|---|---|
Submitted base case | Reference | $3,490,661 | $3,558,338 | $3,527,406 | $3,534,957 | $10,620,701 |
New drug | $3,490,661 | $3,548,353 | $3,533,410 | $3,563,674 | $10,645,437 | |
Budget impact | $0 | –$9,984 | $6,003 | $28,717 | $24,736 | |
CADTH base case | Reference | $7,223,596 | $7,264,682 | $7,180,401 | $7,128,515 | $21,573,598 |
New drug | $7,223,596 | $7,582,596 | $7,758,013 | $8,198,200 | $23,538,809 | |
Budget impact | $0 | $317,914 | $577,612 | $1,069,685 | $1,965,211 |
CADTH conducted the following additional scenario analyses from the drug plan perspective (Scenarios 1 to 5, Table 19):
Assumed an average treatment compliance rate of 85% for SAGH and 90% for LAGH.
Assumed the highest possible dose for a given somatropin product, at 0.24 mg/kg per week (or 0.042 mg/kg per day).
Applied a price reduction of 12%, the price at which somatrogon is cost-effective at a $50,000 per QALY willingness-to-pay threshold under CADTH base-case assumptions in the pharmacoeconomic evaluation.
Assumed that 52% of children receive public coverage.
Applied the assumption that somatrogon captured a greater market share, with 50% market share in year 2 and 80% market share in year 3.
Somatrogon was only associated with budgetary savings in the scenario which assumed a larger dose for somatropin. Somatrogon was associated with a substantial increase in the 3-year total budget impact in comparison with the sponsor’s base case in all other scenarios.
Table 19: CADTH Scenario Analyses
Stepped analysis | Budget impact | Year 0 (current situation) | Year 1 | Year 2 | Year 3 | 3-year total |
|---|---|---|---|---|---|---|
CADTH scenario analysis 1 | Reference | $6,140,057 | $6,174,980 | $5,744,321 | $6,059,238 | 17,978,539 |
New drug | $6,140,057 | $6,530,117 | $6,730,898 | $7,200,167 | 20,461,181 | |
Budget impact | $0 | $355,137 | $986,577 | $1,140,929 | 2,482,643 | |
CADTH scenario analysis 2 | Reference | $9,551,199 | $9,605,524 | $9,494,086 | $9,425,481 | $28,525,091 |
New drug | $9,551,199 | $9,478,678 | $9,377,593 | $9,346,683 | $28,202,953 | |
Budget impact | $0 | –$126,846 | –$116,493 | –$78,798 | –$322,138 | |
CADTH scenario analysis 3 | Reference | $7,223,596 | $7,264,682 | $7,180,401 | $7,128,515 | $21,573,598 |
New drug | $7,223,596 | $7,378,717 | $7,430,054 | $7,641,870 | $22,450,640 | |
Budget impact | $0 | $114,035 | $249,653 | $513,354 | $877,042 | |
CADTH scenario analysis 4 | Reference | $3,756,270 | $3,777,635 | $3,733,809 | $3,706,828 | $11,218,271 |
New drug | $3,756,270 | $3,942,950 | $4,034,167 | $,263,064 | $12,240,181 | |
Budget impact | $0 | $165,315 | $300,358 | $556,236 | $1,021,910 | |
CADTH scenario analysis 5 | Reference | 7,223,596 | $7,264,682 | $7,180,401 | $7,128,515 | $21,573,598 |
New drug | 7,223,596 | $7,582,596 | $8,143,088 | $8,840,011 | $24,565,695 | |
Budget impact | $0 | $317,914 | $962,687 | $1,711,496 | $2,992,096 |
Note: All scenario analyses are conducted based on the CADTH base case undertaken from the drug program plan perspective.
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up-to-date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.