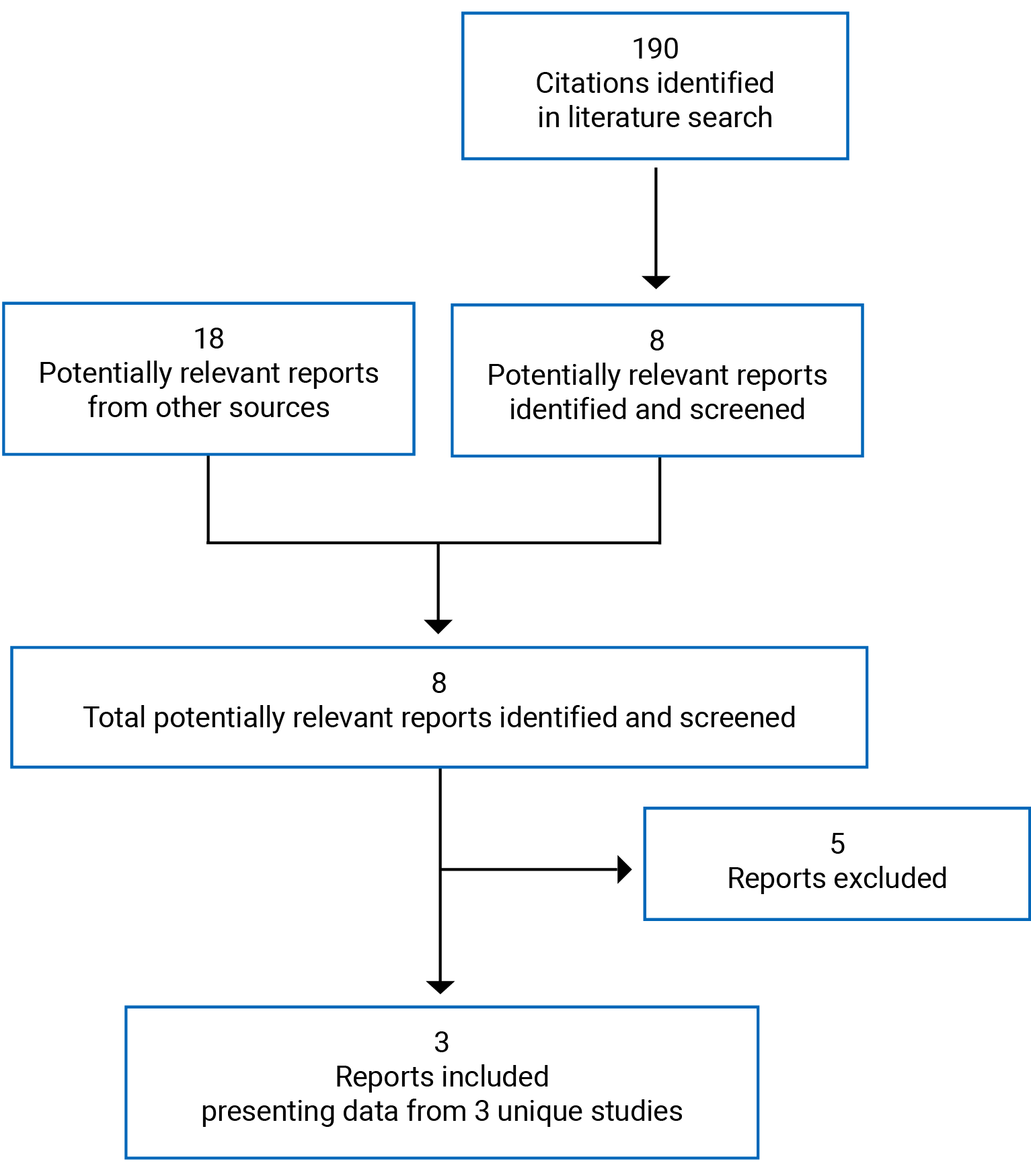
CADTH Reimbursement Review
Inclisiran (Leqvio)
Sponsor: Novartis Pharmaceuticals Canada Inc.
Therapeutic area: Primary hypercholesterolemia
This multi-part report includes:
Clinical Review
Pharmacoeconomic Review
Clinical Review
Abbreviations
AE
adverse event
ANCOVA
analysis of covariance
ApoB
apolipoprotein B
ASCVD
atherosclerotic cardiovascular disease
CCS
Canadian Cardiovascular Society
CHD
coronary heart disease
CHPA
Canadian Heart Patient Alliance
CI
confidence interval
CrI
credible interval
CV
cardiovascular
FH
familial hypercholesterolemia
GalNAc
triantennary N-acetylgalactosamine
HDL-C
high-density lipoprotein cholesterol
HeFH
heterozygous familial hypercholesterolemia
HoFH
homozygous familial hypercholesterolemia
HRQoL
health-related quality of life
IDMC
independent data monitoring committee
ITC
indirect treatment comparison
ITT
intention to treat
LDL
low-density lipoprotein
LDL-C
low-density lipoprotein cholesterol
LDLR
low-density lipoprotein receptor
LMT
lipid-modifying therapy
LSM
least squares mean
MACE
major adverse cardiovascular event
MAR
missing at random
MI
myocardial infarction
MMRM
mixed-effects model for repeated measures
MTD
maximally tolerated dose
nFH
non-familial hypercholesterolemia
NMA
network meta-analysis
PAD
peripheral artery disease
PCSK9
proprotein convertase subtilisin/kexin type 9
PMM
pattern-mixture model
RCT
randomized controlled trial
RNA
ribonucleic acid
SAE
serious adverse event
TEAE
treatment-emergent adverse event
WDAE
withdrawal due to adverse event
Executive Summary
An overview of the submission details for the drug under review is provided in Table 1.
Item | Description |
|---|---|
Drug product | Inclisiran (Leqvio), 284 mg in 1.5 mL (189 mg/mL) solution for SC injection |
Indication | As an adjunct to lifestyle changes, including diet, to further reduce low-density lipoprotein cholesterol (LDL-C) level in adults with the following conditions who are on a maximally tolerated dose of a statin, with or without other LDL-C-lowering therapies:
|
Reimbursement request | As an adjunct to diet and maximally tolerated statin therapy, with or without other lipid-lowering therapies, in adult patients with HeFH or clinical ASCVD, who require additional lowering of LDL-C |
Health Canada approval status | NOC |
Health Canada review pathway | Standard |
NOC date | July 26, 2021 |
Sponsor | Novartis Pharmaceuticals Canada Inc. |
ASCVD = atherosclerotic cardiovascular disease; HeFH = heterozygous familial hypercholesterolemia; LDL-C = low-density lipoprotein cholesterol; NOC = Notice of Compliance; SC = subcutaneous.
Introduction
Atherosclerotic cardiovascular (CV) diseases (ASCVDs) are a group of disorders of the heart and blood vessels. They are the leading cause of death globally, with an estimated 17.9 million deaths each year.1-3 CV diseases are generally associated with high blood-cholesterol levels (hypercholesterolemia), resulting in the buildup of cholesterol, specifically low-density lipoprotein (LDL) cholesterol (LDL-C), and fatty deposits inside the arteries leading to atherosclerosis.3-5
Symptoms of ASCVD depend on the atherosclerotic site and the specific condition; however, typical symptoms of underlying CV issues include pain or pressure, particularly in the chest and/or arms, shortness of breath, lightheadedness or dizziness, cold sweats, and fatigue. More severe manifestations of ASCVD as a result of hypercholesterolemia may include various CV events, such as myocardial infarction (MI) or stroke, which may be fatal.5
There are both genetic and acquired or behavioural causes of hypercholesterolemia. Primary hypercholesterolemia can be classified into 2 subtypes: familial hypercholesterolemia (FH) and non-familial hypercholesterolemia (nFH). FH can be further subdivided into heterozygous (HeFH) and homozygous (HoFH) disease, with HoFH being the more severe and rare form of the disease.6-9
The management of hypercholesterolemia is divided into primary and secondary prevention, where primary prevention is the effort to prevent or delay the onset of ASCVD, while secondary prevention refers to the effort to treat known ASCVD and to prevent or delay the onset of disease manifestations.10,11
Statins are the primary pharmacological intervention to achieve control of LDL-C in patients with hypercholesterolemia. Most patients with ASCVD should be initiated on the maximum dose of high-intensity statins (atorvastatin or rosuvastatin), with the goal of lowering LDL-C by at least 50%. In cases of primary prevention where the LDL goal is unmet with statin therapy alone, add-on ezetimibe or bile acid sequestrants (or both) is recommended.10,12,13 Ezetimibe is a cholesterol absorption inhibitor that blocks the absorption of dietary cholesterol and delivery to the liver, resulting in enhanced clearance of LDL-C and further reducing LDL-C by between 10% and 40% (average 20%).14 In patients with clinical ASCVD, if LDL goals are still not met, proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors (alirocumab and evolocumab) are available to patients meeting certain criteria as an adjunct treatment to diet, a maximally tolerated dose (MTD) of a statin, and ezetimibe.8,15
Inclisiran (Leqvio) is a small interfering ribonucleic acid (RNA) conjugated with triantennary N-acetylgalactosamine (GalNAc) to facilitate uptake by hepatocytes and selectively target the liver. Through RNA interference, inclisiran directs catalytic breakdown of PCSK9 messenger RNA, preventing the production of PCSK9 protein and increasing the number of LDL receptors (LDLRs) on hepatocyte surfaces, which results in increased LDL-C uptake and reduction of the LDL-C in circulation. Inclisiran is approved by Health Canada as an adjunct to lifestyle changes, including diet, to further reduce LDL-C levels in adults with HeFH or nFH with ASCVD who are on an MTD of a statin, with or without other LDL-C-lowering therapies.16
As per the product monograph, the effect of inclisiran on CV morbidity and mortality has not been determined.16
The objective of the current review is to perform a systematic review of the beneficial and harmful effects of inclisiran in adults with primary hypercholesterolemia (HeFH or nFH).
Stakeholder Perspectives
The information in this section is a summary of input provided by the patient groups that responded to CADTH’s call for patient input and from the clinical expert consulted by CADTH for the purpose of this review.
Patient Input
Two patient groups, the Canadian Heart Patient Alliance (CHPA) and the HeartLife Foundation, provided input for this review. The CHPA is a patient-led non-profit umbrella organization of patients, families, health professionals, and supporters dedicated to reducing CV disease and preventing early death due to cholesterol and other risk factors. Its focus is high cholesterol and other lipids, due to genetic and non-genetic factors, as the leading under-diagnosed and under-treated cause of CV disease and early death. The CHPA is the successor to the FH Canada Patient Network and collaborates with FH Canada, the Heart Healthy Program Prevention Clinic at St. Paul’s Hospital (Vancouver), and the Lipid Genetics Clinic at the London (Ontario) Health Sciences Centre’s University Hospital. The HeartLife Foundation is a patient-driven charity whose mission is to transform the quality of life of people living with heart failure by engaging, educating, and empowering a global community to create lasting solutions and build healthier lives.
The information provided by CHPA was gathered from a total of 262 individuals through an online survey (n = 254) and individual interviews (n = 8). The information provided by the HeartLife Foundation was gathered through discussions held with individual members across Canada. Members include both patients living with heart failure and their family caregivers. The discussions were held as informal group conversations (e.g., via Zoom sessions) or through phone calls with individuals.
About 25% of respondents to the CHPA survey reported regular physical symptoms related to their lipid levels, some minor and some significant, including headaches (like icy picks), chest pains, muscle pains in legs and ankles, shortness of breath, xanthomas (under the skin of the wrists, ankles, or elsewhere), weakness, fatigue, muscle loss, and neuropathy. About 20% also indicated that managing their cholesterol level and keeping it at target was an ongoing challenge; however, 20% said their high cholesterol or other lipid condition had little to no effect on their quality of life. Many reported they had changed their diet and exercise. However, some responses indicated that patients were not always aware of the impact of high cholesterol, in part because they were well managed on treatment and did not experience daily symptoms. Most respondents felt positive about their daily life and had accepted or adapted to living with high cholesterol, including those who have experienced a CV event or have stents. The 2 most frequently mentioned sources of anxiety were:
uncertainty regarding the effectiveness of the medications or the risk of a CV event in future
the impact on their children, whether their children had been diagnosed or were at risk.
The majority of respondents to the CHPA survey expressed multiple concerns, largely about treatment schedule, side effects, and the cost of current therapies. For respondents, the most important impact was knowing there was a treatment that could lower their cholesterol levels and keep them closer to target, thereby reducing the risk of further CV events. Public reimbursement for PCSK9 inhibitors in Canada is limited, and access for patients with uncontrolled LDL-C is highly restricted by provincial health benefit program reimbursement criteria.
Clinician Input
Input From the Clinical Expert Consulted by CADTH
According to advice obtained from the clinical expert consulted by CADTH for this review, many patients are unable to meet the pre-specified LDL-C thresholds, outlined in the Canadian Cardiovascular Society (CCS) Guidelines for the Management of Dyslipidemia for the Prevention of Cardiovascular Disease and the CCS Position Statement on FH, for current treatments, and there is an unmet need for additional treatment options that further reduce LDL-C levels. Statins are considered the standard of treatment for the prevention of ASCVD in high-risk patients and patients with FH or severe hypercholesterolemia; however, the clinical expert pointed out that up to 15% of patients are partially or completely intolerant to statins.
It was also emphasized by the clinical expert that the CCS guidelines should be used as a basis for recommendation in identifying and treating patients (in patients with HeFH without clinical ASCVD whose LDL-C remains above the target LDL-C level of at least 2.5 mmol/L or less than 50% reduction from baseline despite MTD statin therapy with or without ezetimibe therapy, or patients with HeFH and ASCVD whose LDL-C remains above at least 1.8 mmol/L despite MTD statin therapy, with or without ezetimibe), and that suitable patients should match the characteristics of the patients enrolled in the clinical trials included in this review. The clinical expert indicated that the patients least suitable for treatment with the drug under review were those with low-risk ASCVD, patients with low-risk severe hypercholesterolemia that is well controlled with statins, patients with ASCVD who are at their LDL-C goals with current therapies, patients with nFH above a certain age where the treatment would be for primary prevention, and patients with multiple comorbidities that limit lifespan.
The clinical expert stated that the percentage reduction in LDL-C and the absolute level of LDL-C achieved are outcomes used in clinical practice to determine response to treatment. The clinical expert indicated that treatment response should be assessed every 6 months, then yearly. The patients considered most likely to exhibit a response to treatment with inclisiran, according to the clinical expert, were those that achieved a 30% to 40% reduction in LDL-C from baseline levels (while on an optimized statin, with or without ezetimibe therapy) but require further lowering of LDL-C. The clinical expert stated that age, end-stage disease, and/or dementia are important factors that should be considered when deciding to discontinue treatment.
Clinician Group Input
A group of clinicians consisting of lipid specialists working in lipid clinics in British Columbia, including the Healthy Heart Program Prevention Clinic at St. Paul’s Hospital, the lipid clinic at Surrey Memorial Hospital, and the Victoria Lipid Clinic, provided input for this review.
The clinician group noted the tolerability of current treatments, compliance, ability to treat to targeted lipid levels, and accessibility as the current unmet needs in treating patients with HeFH and/or ASCVD. The clinician group described an ideal treatment option as 1 that would reduce levels of LDL-C, non-HDL-C, and apolipoprotein B (ApoB); reduce the risk of major adverse cardiovascular events (MACEs) and CV mortality, and be safe and well tolerated, with properties that promote adherence.
The clinician group noted that patients with the greatest unmet need for intervention are those with HeFH, patients with statin intolerance, and patients with ASCVD with other markers of high risk (e.g., multi-vessel disease, polyvascular disease, diabetes, elevated lipoprotein [a]). Finally, they noted the patients least suitable for treatment with the drug under review would be patients who do not have an indication for the therapy, patients who have achieved LDL targets on other therapies (a statin with or without ezetimibe), and patients who have not attempted a statin.
The clinician group noted that inclisiran may displace other PCSK9 inhibitors as an add-on to statins and ezetimibe if it is more accessible than current treatments and depending on the results of currently ongoing CV outcome trials. It may also fill a void if it is approved for secondary prevention in high-risk patients.
Drug Program Input
The questions the drug plans submitted to the clinical expert for clarification focused on the definitions of specific populations and the variability in treatment according to Canadian guidelines. The clinical expert consulted by CADTH for this review indicated that, overall, initiation criteria for inclisiran may follow that of other PCSK9 inhibitors, with patients first receiving MTD statins followed by ezetimibe, if they are within 20% of their LDL-C target, or PCSK9 if their distance to target is greater than 20%. The cut-point targets of 2.6 mmol/L (> 2.5 mmol/L) for HeFH and 1.8 mmol/L for patients with ASCVD are reflective of the guidelines for these populations. The drug plans were also concerned whether laboratory assessments were appropriate outcomes for assessing effectiveness in the real world. The clinical expert noted that LDL-C, ApoB, and non-HDL-C are guideline-recommended biomarkers for CV outcomes. It was also noted that inclisiran may follow the same initiation and renewal criteria as the currently available PCSK9 inhibitors, and that the occurrence of cardiac events would not warrant discontinuation. Lastly, the drug programs were concerned with whether or not inclisiran would be used in patients who do not have hypercholesterolemia or who had not had a prior heart attack or stroke (i.e., for primary prevention). The clinical expert noted that elevated LDL-C can be caused by other diseases, and that these should be addressed separately and are therefore not within the context of this review.
Clinical Evidence
Pivotal Studies and Protocol Selected Studies
Description of Studies
A total of 3 studies were included in this review: ORION-9, ORION-10, and ORION-11.17-19 The included studies were all phase III, double-blind, randomized controlled trials (RCTs) comparing inclisiran with placebo in patients with HeFH or ASCVD (or with an ASCVD risk equivalent, i.e., diabetes, FH, or a 20% or greater 10-year risk of a CV event as assessed by the Framingham Risk Score for CV disease or equivalent) who were receiving MTD statins or were statin-intolerant. Patients in the ORION-9 trial were adults (≥ 18 years) with a history of HeFH (with a diagnosis of HeFH by genetic testing or phenotypic Simon Broome criteria) and/or a documented history of untreated LDL-C of greater than 190 mg/dL and a family history of FH, or elevated cholesterol or early heart disease, which may indicate FH. Patients enrolled in the ORION-10 trial were adults (≥ 18 years) with a history of ASCVD, and patients enrolled in the ORION-11 trial were adults (≥ 18 years) with a history of ASCVD or with an ASCVD risk equivalent. In all 3 ORION studies, patients were randomized 1:1 to either inclisiran sodium 300 mg or placebo, in addition to MTD statin. The ORION-9, −10, and −11 trials enrolled 482, 1,561, and 1,617 patients, respectively. The studies were all 18 months in duration, with patients receiving 4 300 mg doses of inclisiran sodium (1 each on day 1, day 90, day 270, and day 450). The primary outcome of the ORION-9, −10, and −11 trials was the percentage change in LDL-C from baseline to day 510. In all trials, the co-primary end point was the average percentage change in LDL-C from baseline over the period after day 90 up to day 540, reflecting the start of the twice-yearly dosing regimen. Incidences of CV death, resuscitated cardiac arrest, non-fatal MI, non-fatal stroke (ischemic and hemorrhagic) were exploratory outcomes in the ORION trials within the composite outcome of MACEs, and total deaths was a secondary outcome reported as adverse events (AEs) in the ORION studies.
Baseline characteristics were well balanced across groups in each trial. In ORION-9, patients were mostly White (94.0%) with a median age of 56 years, and more than half of the patients were female (52.9%). CV risk factors were balanced between the treatment groups. Overall, 350 patients (72.6%) had an ASCVD risk equivalent and 132 (27.4%) had ASCVD. A total of 356 patients (73.9%) were treated with high-intensity statins at baseline, and just more than half were treated with ezetimibe. Partial or complete intolerance to statins was reported in 122 patients (25.3%).17 In ORION-10, patients were mostly White (85.7%) and male (69.4%), with a median age of 67 years. All patients had ASCVD, and most had coronary heart disease (CHD) (91.1%). A total of 1,084 patients (69.4%) were on high-intensity statins at baseline, and 156 patients (9.9%) were treated with ezetimibe. Partial or complete intolerance to statins was reported in 344 patients (22.0%).18
In ORION-11, patients were mostly White (98.1%) and male (71.7%), with a median age of 65 years. CV risk factors were balanced between the treatment groups; 1,414 (87.4%) had ASCVD and 203 (12.6%) had an ASCVD risk equivalent. The non-HeFH, ASCVD risk–equivalent population from ORION-11 was not of interest to this review, as these patients were not included in the funding request. Overall, 1,261 patients (78.0%) were on a high-intensity statin at baseline. A total of 114 patients (7.1%) were treated with ezetimibe. Partial or complete intolerance to statins was reported in 185 patients (11.4%).19
Efficacy Results
All-cause and CV-related mortality were assessed as AEs in the ORION-9, −10, and −11 trials, and were reported as the incidence of death within the safety population. In ORION-9, only 2 deaths occurred (0.4%), 1 in each treatment group.17 A total of 23 patients died during the ORION-10 study, 12 (1.5%) in the inclisiran group, and 11 (1.4%) in the placebo group.18 In total, 29 patients (1.8%) died during the ORION-11 study, 14 (1.7%) in the inclisiran group, and 15 (1.9%) in the placebo group.19 Most frequently, deaths were related to cardiac disorders as a system organ class, ranging from 1 patient (0.4%) to 7 patients (0.9%) in the ORION-9, −10, and −11 trials.17-19
Although not referred to as CV-related morbidity in the ORION trials, for the purposes of this review, the incidence of MACEs and its composite components were considered CV-related morbidity and it was an exploratory outcome of the ORION trials. No between-group comparisons were conducted in the ORION-9, −10, or −11 trials for this outcome. The incidence of MACEs in the inclisiran groups was consistently similar to or lower than in the placebo groups across all trials (4.1% versus 4.2%, 7.4% versus 10.2%, and 7.8% versus 10.3%, in ORION-9, −10, −11, respectively). Non-fatal MI was the most frequently occurring individual event across all trials, occurring in 3.7% versus 4.2%, 5.1% versus 8.2%, and 5.8% versus 8.5% of patients in the inclisiran and placebo groups of ORION-9, −10, and −11, respectively.17-19 No resuscitated cardiac arrests or stroke events occurred in the ORION-9 trial.17 Other CV-related morbidities of interest to this review, including hospitalizations and minimally invasive CV interventions, were not reported in the ORION trials.
The primary efficacy end point in the ORION-9, −10, and −11 trials was the percentage change in LDL-C from baseline to day 510. In all ORION trials, inclisiran reduced LDL-C levels from baseline to day 510: −41.15% in ORION-9 (95% confidence interval [CI], −44.52 to −37.77), −56.34% in ORION-10 (95% CI, −58.35 to −54.34), and −49.3% in ORION-11 (95% CI, −51.22 to −47.48), while the change from baseline LDL-C levels increased with placebo: 8.37% in ORION-9 (95% CI, 3.96 to 12.77), 1.30% in ORION-10 (95% CI, −1.24 to 3.83), and 4.2% in ORION-11 (95% CI, 1.62 to 6.69).17-19 Between-group differences were statistically significant in favour of inclisiran in all studies with differences from placebo of −49.52 (95% CI, −55.04 to −43.99) in ORION-9, −57.64 (95% CI, −60.86 to −54.43) in ORION-10, and −53.5 (95% CI, −56.66 to −50.35) in ORION-11 (P < 0.0001 for all).17-19 The clinical expert consulted by CADTH considered the between-group differences in LDL-C levels to be clinically meaningful.
Results for key secondary outcomes in the ORION trials of absolute change in LDL-C from baseline to day 510, time-adjusted change in LDL-C from baseline after day 90 up to day 540, and percentage change from baseline to day 510 in total cholesterol, ApoB, and non-HDL-C were consistent with the co-primary end points. For absolute change in LDL-C from baseline to day 510, inclisiran displayed a larger absolute reduction in LDL-C: −58.95 mg/dL in ORION-9 (95% CI, −64.75 to −53.15), −56.18 mg/dL in ORION-10 (95% CI, −58.47 to −53.90), and −50.91 mg/dL in ORION-11 (95% CI, −53.14 to −48.67). The between-group differences were statistically significant in favour of inclisiran in all studies: −68.89 mg/dL in ORION-9 (95% CI, −77.11 to −60.67), −54.12 mg/dL in ORION-10 (95% CI, −57.37 to −50.88), and −51.87 mg/dL in ORION-11 (95% CI, −55.01 to −48.72) (P < 0.0001 for all). In all trials, inclisiran was associated with greater absolute reductions in LDL-C from baseline after day 90 and up to day 540: −56.58 mg/dL (95% CI, −60.98 to −52.17) versus 6.17 mg/dL (95% CI, 1.72 to 10.62) in ORION-9, −53.66 mg/dL (95% CI, −55.41 to −51.92) versus −0.39 mg/dL (95% CI, −2.14 to 1.37) in ORION-10, and −48.63 mg/dL (95% CI, −50.37 to −46.89) versus 0.31 mg/dL (95% CI, −1.42 to 2.04) in ORION-11. The mean difference between inclisiran and placebo was statistically significant in all trials (P < 0.0001). Lastly, results for percentage change in total cholesterol, ApoB, and non-HDL-C showed greater percentage changes for the inclisiran groups in all studies, and the mean difference from placebo was statistically significant in all cases (P < 0.0001).17-19
Other outcomes of interest to this review, including health-related quality of life (HRQoL) and neurocognitive assessments, were not included in the ORION trials.
Harms Results
The incidence of treatment-emergent adverse events (TEAEs) was consistent between inclisiran- and placebo-treated patients as well as across trials, with patients experiencing at least 1 TEAE (76.8% versus 71.7%, 73.5% versus 74.8%, and 82.7% versus 81.5% in ORION-9, −10, and −11, respectively).17-19 There was no difference in the frequency of treatment-emergent serious adverse events (SAEs) between the treatment groups in ORION-9, −10, and −11. Treatment-emergent SAEs in ORION-9 occurred in 7.5% of inclisiran-treated patients and 13.8% of placebo-treated patients.17 In ORION-10 and −11, SAEs occurred in 22.4% and 22.3% of inclisiran-treated patients compared with 26.3% and 22.5% of placebo-treated patients.18,19 In ORION-9, 1.2% of patients in the inclisiran group withdrew due to an AE, while no patients in the placebo group withdrew due to AEs.17 The incidence of withdrawal due to adverse events (WDAEs) in ORION-10 and −11 were similar, with 2.4% and 2.8% of inclisiran-treated patients and 2.2% of placebo-treated patients in each trial withdrawing due to AEs, respectively.18,19
No difference in either neurologic events or neurocognitive disorders was observed between inclisiran and placebo in any of the ORION trials; however, the incidence was higher in all placebo groups. In all trials, fewer placebo-treated patients reported TEAEs at the injection site compared with those treated with inclisiran. Injection-site reactions were mild to moderate, and no severe reactions were seen across trials. There were no differences between inclisiran and placebo for other notable harms of hypersensitivity reactions, renal safety, or hepatic safety.17-19
Table 2: Summary Key Results From Pivotal and Protocol Selected Studies
Outcome | ORION-9 | ORION-10 | ORION-11 | |||
|---|---|---|---|---|---|---|
Inclisiran N = 242 | Placebo N = 240 | Inclisiran N = 781 | Placebo N = 780 | Inclisiran N = 810 | Placebo N = 807 | |
Mortality, n (%)a | ||||||
TEAEs with fatal outcome | 1 (0.4) | 1 (0.4) | 12 (1.5) | 11 (1.4) | 14 (1.7) | 15 (1.9) |
|||||||||||||||||||||||| | ||||| | ||||| | ||||| | ||||| | ||||| | || |
|||||||||||||||||||||||||||||||||||||||||||||||||||| | ||||| | ||||| | ||||| | ||||| | ||||| | || |
|||| | ||||| | ||||| | ||||| | ||||| | ||||| | || |
Incidence of MACEs (CV death, MI, stroke, resuscitated cardiac arrest, non-fatal MI, and non-fatal stroke), n (%)a | ||||||
MACE | 10 (4.1) | 10 (4.2) | 58 (7.4) | 79 (10.2) | 63 (7.8) | 83 (10.3) |
CV death | 1 (0.4) | 0 (0) | 7 (0.9) | 5 (0.6) | 9 (1.1) | 9 (1.1) |
Resuscitated cardiac arrest | NR | NR | 1 (0.1) | 1 (0.1) | 3 (0.4) | 0 (0) |
Non-fatal MI | 9 (3.7) | 10 (4.2) | 40 (5.1) | 64 (8.2) | 47 (5.8) | 68 (8.5) |
Stroke (ischemic or hemorrhagic) | NR | NR | 12 (1.5) | 10 (1.3) | 4 (0.5) | 8 (1.0) |
Percentage change in LDL-C (95% CI)b | ||||||
Percentage CFB in LDL-C to day 510 (observed values) | −41.15 (−44.52 to −37.77) | 8.37 (3.96 to 12.77) | −56.34 (−58.35 to −54.34) | 1.30 (−1.24 to 3.83) | −49.3 |||||||||||||| | 4.2 |||||||||| |
Difference from placebo | −49.52 (−55.04 to −43.99) P < 0.0001 | −57.64 (−60.86 to −54.43) P < 0.0001 | −53.5 (−56.66 to −50.35) P < 0.0001 | |||
Percentage CFB in LDL-C to day 510 (washout-imputed values) | −39.67 (−43.72 to −35.62) | 8.22 (4.27 to 12.16) | −51.28 (−53.76 to −48.81) | 0.96 (−1.48 to 3.40) | −45.8 (−48.16 to −43.48) | 4.0 (1.76 to 6.31) |
Difference from placebo | −47.89 (−53.52 to −42.26) P < 0.0001 | −52.24 (−55.65 to −48.83) P < 0.0001 | −49.9 (−53.07 to −46.64) P < 0.0001 | |||
Time-adjusted percentage CFB in LDL-C after day 90 to day 540 | −38.08 (−41.03 to −35.14) | 6.22 (3.26 to 9.17) | −51.27 (−53.00 to −49.54) | 2.51 (0.77 to 4.25) | −45.82 (−47.52 to −44.13) | 3.35 (1.65 to 5.05) |
Difference from placebo | −44.30 (−48.48 to 40.12) P < 0.0001 | −53.78 (−56.23 to 51.33) P < 0.0001 | −49.17 (−51.57 to 46.77) P < 0.0001 | |||
Absolute change in LDL-C (95% CI)b | ||||||
Absolute CFB in LDL-C to day 510 (PMM) | −58.95 (−64.75 to 53.15) | 9.94 (4.10 to 15.78) | −56.18 (−58.47 to 53.90) | −2.06 (−4.36 to 0.24) | −50.91 (−53.14 to 48.67) | 0.96 (−1.26 to 3.18) |
Difference from placebo | −68.89 (−77.11 to 60.67) P < 0.0001 | −54.12 (−57.37 to 50.88) P < 0.0001 | −51.87 (−55.01 to 48.72) P < 0.0001 | |||
||||||||||||||||||||||||||||||||||||||||||||||||||| | |||||||||||||| | |||||||||| | ||||||||||||||| | |||||||||| | |||||||||||||||| | |||||||||||| |
|||||||||||||||||||||||||||| | ||||||||||||||||| | |||||||||||||||||||||| | |||||||||||||||||||| | |||
Time-adjusted absolute CFB in LDL-C after day 90 to day 540 (PMM), mg/dL | −56.58 (−60.98 to 52.17) | 6.17 (1.72 to 10.62) | −53.66 (−55.41 to 51.92) | −0.39 (−2.14 to 1.37) | −48.63 (−50.37 to −46.89) | 0.31 (−1.42 to 2.04) |
Difference from placebo | −62.74 (−69.01 to −56.48) P < 0.0001 | −53.28 (−55.75 to −50.80) P < 0.0001 | −48.94 (−51.39 to −46.48) P < 0.0001 | |||
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | |||||||||||||| | |||||||||| | |||||||||||||| | |||||||||| | |||||||||||||||| | |||||||||||| |
|||||||||||||||||||||||||||| | |||||||||||||| | |||||||||||||||||| | |||||||||||||||| | |||
Percentage change in TC, ApoB, non-HDL-C (95% CI)b | ||||||
Percentage CFB to day 510 in TC (PMM) | −25.11 (−27.83 to −22.39) | 6.66 (3.96 to 9.36) | −33.56 (−35.09 to −32.03) | −0.42 (−1.95 to 1.11) | −28.00 (−29.40 to −26.60) | 1.79 (0.38 to 3.21) |
Difference from placebo | −31.77 (−35.59 to −27.94) P < 0.0001 | −33.13 (−35.30 to −30.97) P < 0.0001 | −29.79 (−31.78 to −27.81) P < 0.0001 | |||
||||||||||||||||||||||||||||||||||||||||||||| | |||||||||||||| | |||||||| | ||||||||||||||| | |||||||||| | |||||||||||||||| | ||||||||||| |
|||||||||||||||||||||||||||| | |||||||||||||||||| | ||||||||||||||||||||||| | ||||||||||||||||||||| | |||
Percentage CFB to day 510 in ApoB (PMM) | −33.14 (−35.91 to −30.36) | 2.93 (0.14 to 5.71) | −44.81 (−46.52 to −43.10) | −1.72 (−3.46 to 0.02) | −38.15 (−39.76 to −36.54) | 0.79 (−0.82 to 2.41) |
Difference from placebo | −36.06 (−39.99 to −32.14) P < 0.0001 | −43.09 (−45.50 to −40.67) P < 0.0001 | −38.94 (−41.21 to −36.67) P < 0.0001 | |||
||||||||||||||||||||||||||||||||||||||||||||||||| | |||||| |||||||||||||| | |||||| |||||||||||||||| | |||||| |||||||||||||||| | |||||| |||||||||||||||| | |||||| |||||||||||||||| | |||||| |||||||||||||||| |
|||||||||||||||||||||||||||| | |||||||||||||||||| |||||| | |||||||||||||||||| |||||| | |||||||||||||||||| |||||| | |||
Percentage CFB to day 510 in non-HDL-C (PMM) | −34.93 (−38.46 to −31.40) | 7.43 (3.93 to 10.92) | −47.41 (−49.44 to −45.38) | −0.05 (−2.08 to 1.99) | −41.16 (−43.09 to −39.24) | 2.15 (0.22 to 4.09) |
Difference from placebo | −42.36 (−47.32 to −37.40) P < 0.0001 | −47.36 (−50.25 to −44.47) P < 0.0001 | −43.32 (−46.04 to −40.60) P < 0.0001 | |||
||||||||||||||||||||||||||||||||||||||||||||||||||||| | |||||||||||||| | |||||||||| | ||||||||||||||| | |||||||||| | |||||||||||||||| | |||||||||| |
|||||||||||||||||||||||||||| | |||||||||||||| | |||||||||||||||||| | |||||||||||||||| | |||
Harms, n (%)a | ||||||
TEAEs | 185 (76.8) | 172 (71.7) | 574 (73.5) | 582 (74.8) | 671 (82.7) | 655 (81.5) |
SAEs | 18 (7.5) | 33 (13.8) | 175 (22.4) | 205 (26.3) | 181 (22.3) | 181 (22.5) |
WDAEs | 3 (1.2) | 0 (0) | 19 (2.4) | 17 (2.2) | 23 (2.8) | 18 (2.2) |
Notable harms, n (%)a | ||||||
|||||||||||||||||||||||| | |||||| | |||||| | |||||| | || | |||||| | ||||| |
Injection-site reactions | 41 (17.0) | 4 (1.7) | 47 (6.0) | 15 (1.9) | 62 (7.6) | 14 (1.7) |
|||||||||||||||||||||||||||||| | |||||| | || | |||||| | || | |||||| | |||||| |
|||||||||||||| | |||||| | |||||| | |||||| | || | |||||| | |||||| |
|||||||||||||||| | |||||| | |||||| | |||||| | |||||||| | |||||| | ||||| |
ApoB = apolipoprotein B; CFB = change from baseline; CI = confidence interval; CV = cardiovascular; HDL-C = high-density lipoprotein cholesterol; LDL-C = low-density lipoprotein cholesterol; LSM = least squares mean; MACE = major cardiovascular event; MI = myocardial infarction; MMRM = mixed-effects model for repeated measures; PMM = pattern-mixture model; SAE = serious adverse event; TEAE = treatment-emergent adverse event; TC = total cholesterol; WDAE = withdrawal due to adverse event.
aSafety population.
bIntention-to-treat population.
Source: Clinical Study Reports for ORION-9,17 ORION-10,18 and ORION-11.19
Critical Appraisal
ORION-9, ORION-10, and ORION-11 were all phase III, double-blind RCTs. There were no notable differences in baseline characteristics within the studies. Given the higher incidence of injection-site reactions in the inclisiran group, it may have been possible to reveal treatment assignment, and it is unclear what effect this would have had on the results; however, it is considered minimal, given the objective nature of the study outcomes. There were no apparent imbalances in discontinuations that may have led to unblinding. Screening failures and inclusion criteria were considered appropriate for the ORION trials, given the specified LDL-C cut points of 1.8 mmol/L and 2.6 mmol/L, which are aligned with current CCS guidelines. The included patient populations in the ORION studies were mostly reflective of the population for which reimbursement was requested; however, ORION-11 included a proportion of patients with ASCVD-risk equivalences (13%), which were not part of the reimbursement population requested by the sponsor. All ORION trials were placebo-controlled studies; therefore, they lacked comparison to a relevant treatment, increasing the risk of bias in the estimation of treatment effects.
Acceptable methods to account for multiplicity were used in all trials for the co-primary and key secondary efficacy end points. Other secondary and exploratory end points, including CV-related mortality and morbidity, which were of importance to this review, were not controlled for multiplicity; thus, they need to be interpreted with consideration of type I error. Given the large number of comparisons in the study, a statistically significant finding may be attributable to an inflated type I error.
The ORION trials were based on lipid and lipoprotein efficacy outcomes that are well recognized and widely accepted surrogate end points for CV-related outcomes. The clinical expert consulted by CADTH noted that the incremental improvements in LDL-C and the differences between the inclisiran and placebo groups are still clinically meaningful, given that patients are heavily treated with other therapies in both clinical trials and real-world settings. However, important outcomes, including reductions in CV-related morbidity (CV death, MI, stroke, resuscitated cardiac arrest, non-fatal MI, and non-fatal stroke) or the composite of MACEs as well as all-cause and CV-related mortality, were exploratory and not powered for statistical analysis; thus, the impact of inclisiran on these outcomes remains uncertain. Moreover, the duration of the studies (18 months) was not sufficient to measure these outcomes. No HRQoL or patient-reported outcomes were assessed in the ORION trials; therefore, the effect of inclisiran with respect to these outcomes remains unknown.
One outcome considered important to patients was a more appropriate dosing regimen. The majority of patients in the ORION trials completed the study ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. In comparison with current PCSK9 inhibitors, which require injections every 2 weeks, inclisiran is a twice-yearly injection administered by a health care professional. The clinical expert consulted by CADTH for this review indicated that this rate of injection may improve adherence relative to current PCSK9 inhibitors.
Indirect Treatment Comparisons
Description of Studies
The sponsor submitted an indirect treatment comparison (ITC) that compared the efficacy of inclisiran with relevant drug comparators in patients with HeFH or ASCVD (or an ASCVD risk equivalent). The objective of the sponsor-submitted report was to conduct a feasibility assessment through a systematic review of the literature and, if possible, to conduct an ITC evaluating the relative efficacy and safety of inclisiran versus relevant drug comparators, including ezetimibe and other PCSK9 inhibitors, in patients with HeFH or ASCVD (or an ASCVD risk equivalent).20
The sponsor-submitted ITC was informed by a systematic review of RCTs conducted in April 2020. Thirty-nine studies met the inclusion criteria of the review and feasibility assessment, and 22 studies were sub-selected for inclusion in the ITC based on network connectivity and homogeneity in study characteristics, patient characteristics, or outcomes that were likely modifiers of the relative treatment effects.20
The analyses were conducted using a network meta-analysis (NMA). A selection of both fixed and random effects was conducted for outcomes of interest. Random-effects analyses were selected as the base case, given the number of studies per node and observed heterogeneity in patient and trial characteristics. Three network scenarios were conducted: HeFH patients on an MTD statin, patients with ASCVD or an ASCVD risk equivalent on an MTD statin, and patients with ASCVD or an ASCVD risk equivalent who are intolerant to statins. Efficacy outcomes included the percentage, absolute, and time-adjusted change from baseline in LDL-C and percentage change from baseline in HDL-C, and safety outcomes included total discontinuations and discontinuations due to AEs.20
Efficacy Results
A total of 7 trials were included in the network for the HeFH population on MTD statins, 13 studies were included in the base-case network for the ASCVD and ASCVD risk–equivalent populations on MTD statins, where 1 closed loop was formed, and 7 trials were included in the network for ASCVD and ASCVD risk–equivalent populations intolerant to statins. In the HeFH population on MTD statins, there was no difference between inclisiran and alirocumab or evolocumab for any efficacy and safety outcomes. In the network for ASCVD and ASCVD risk–equivalent populations on an MTD statin, inclisiran was favoured over ezetimibe for efficacy outcomes related to LDL-C; however, there was no difference between inclisiran and alirocumab or evolocumab for any efficacy or safety outcomes. In the network for ASCVD and ASCVD risk–equivalent populations intolerant to statins, inclisiran was favoured over ezetimibe for efficacy outcomes related to LDL-C but not for safety outcomes. There was no difference between inclisiran and alirocumab or evolocumab in any efficacy or safety outcomes.
Critical Appraisal
There were several limitations with the key assumptions made in the NMA approach with regard to background statin use and the time of assessment of outcomes. This impacted the clinical and methodological heterogeneity, thus limiting the interpretability and generalizability of the results. Though not reported or accounted for, these assumptions likely impacted treatment effects and the results of each NMA and were a significant source of heterogeneity in the studies. It was assumed in the NMA that individual statins had similar efficacy as background therapy, regardless of dose, and that the use of statins would not bias the results of the NMA; however, based on discussions with the clinical expert consulted by CADTH, this was not considered a reasonable assumption. It was also assumed that differences in CV risk and severity would not impact the relative effects on LDL-C; therefore, no attempt was made to adjust for differences in baseline characteristics due to the number of studies and the inconsistent reporting of characteristics. The NMA used 24 weeks as the time of assessment, which was considered acceptable for lipid and lipoprotein outcomes. End-of-study values for safety were used and considered comparable if the duration of follow-up was 24 weeks or longer. Variations in trial length are bound to influence the number of patients withdrawing for various reasons and, given the time of assessment was 24 weeks, may undermine true treatment effects. Additionally, given the twice-yearly dosing regimen of inclisiran, a an assessment at 24 weeks may be insufficient to assess safety outcomes compared with the every-2-weeks dosing regimen of alirocumab and evolocumab.
Overall, the studies included in the NMA were believed to be statistically heterogeneous based on the considerable I2; however, it is unclear what the source of heterogeneity was. The observed heterogeneity was likely due to observed and unobserved differences in patient populations across the included studies, the data imputation analysis methods, and the specific background treatments allowed and/or delivered. Unidentified or unknown methodological or clinical (particularly treatment-effect modifiers) heterogeneity needs to be explored, as it is unclear if the transitivity assumption was appropriately met.
In general, all treatments were favoured over placebo for all outcomes in each network scenario; however, the results typically displayed exceedingly wide credible intervals (CrIs), challenging the precision of the results.
Other Relevant Evidence
Two additional relevant studies (ORION-4 and ORION-8) were noted in the sponsor submission and identified in the CADTH screening of clinical trial databases. At the time of this review, no results were available for either of the studies. As such, ORION-4 and ORION-8 were not included in the available evidence discussed earlier. ORION-4 aims to evaluate the efficacy of inclisiran on the number of patients with the major CV events of CHD, MI, fatal or non-fatal ischemic stroke, or urgent coronary revascularization procedure, or the composite of CHD death or MI, and the number of CV deaths in patients with ASCVD. ORION-8 is an extension study of the ORION-5, −9, −10, and −11 trials to evaluate the long-term efficacy, safety, and tolerability of inclisiran in patients with ASCVD, an ASCVD risk equivalent, HeFH, or HoFH who still had elevated LDL-C despite maximally tolerated LDL-C-lowering therapies. Results of these trials are expected to provide further evidence to better characterize the efficacy profile of inclisiran in pertinent clinical outcomes and provide long-term efficacy and safety data for inclisiran.
Conclusions
In all 3 ORION studies, inclisiran demonstrated a statistically significant and clinically meaningful change compared with placebo in the co-primary end points of change in LDL-C from baseline at day 510, and time-adjusted change from baseline after day 90 up to day 540 in patients with HeFH, ASCVD, and ASCVD or an ASCVD risk equivalent, respectively. Results of the key secondary end points were in line with the primary outcome. Together, the lipid and lipoprotein efficacy outcomes were appropriate, and are reflective of clinical practice in Canada. The combined percentage change and absolute change in LDL-C from baseline, as well as the ApoB and non-HDL-C measurements, are guideline-specific and relevant in diagnosing and treating HeFH and ASCVD. None of the ORION studies assessed HRQoL; thus, the effect of inclisiran on HRQoL is uncertain.
Overall, treatment with inclisiran was well tolerated over the study period. Inclisiran did not appear to be associated with more AEs or SAEs compared with placebo. Known AEs of interest, such as injection-site reactions, were more frequent in the inclisiran group; however, there were no concerns. Although the sponsor submitted an ITC evaluating inclisiran and appropriate comparators in the treatment of HeFH and ASCVD, the results of the indirect evidence for the outcomes evaluated were inconclusive on the efficacy and safety of inclisiran compared with relevant treatments. In addition, the sponsor-submitted ITC provides minimal value to assess efficacy compared with either ezetimibe or the other available PCSK9 inhibitor, as it did not evaluate clinical outcomes.
The ORION-9, −10, and −11 trials were not designed to compare between treatment groups for outcomes of critical importance to the review: mortality (all-cause and CV-related) and CV-related morbidity (resuscitated cardiac arrest, non-fatal MI, non-fatal stroke, and MACE); however, there was no apparent difference between inclisiran and placebo. The short follow-up duration of the ORION trials (18 months) did not allow for adequate evaluation of these outcomes. Longer follow-up data obtained from the ORION-8 trial, combined with results from the ORION-4 trial, will provide important insight into the efficacy of inclisiran in reducing CV-related morbidity and mortality.
Introduction
Disease Background
ASCVDs are a group of disorders of the heart and blood vessels. They are the leading cause of death globally, with an estimated 17.9 million deaths each year.1-3 CV diseases are generally associated with high blood-cholesterol levels (hypercholesterolemia) resulting in the buildup of cholesterol, specifically LDL-C, and fatty deposits inside the arteries leading to atherosclerosis.3-5 Changes in the endothelial cell lining of the arterial wall lead to an accumulation of lipoproteins and inflammatory cells, resulting in the formation of an atherosclerotic lesion or plaque, which narrows the arterial lumen reducing blood flow. ASCVDs consist of coronary artery disease, cerebrovascular disease, and peripheral artery disease (PAD).
Symptoms of ASCVD depend on the atherosclerotic site and the specific condition; however, typical symptoms of underlying CV issues include pain or pressure, particularly in the chest and/or arms, shortness of breath, lightheadedness or dizziness, cold sweats, and fatigue. More severe manifestations of ASCVD as a result of hypercholesterolemia may include various CV events such as MI or stroke, which may be fatal.5
There are both genetic and acquired or behavioural causes of hypercholesterolemia. Primary hypercholesterolemia can be classified into 2 subtypes: FH and nFH. FH is associated with a specific underlying genetic defect resulting in elevated LDL-C levels from birth that persist throughout life and can lead to the early development of atherosclerosis, while nFH is associated with several genetic interactions as well as dietary and lifestyle risk factors. FH can be further subdivided into HeFH and HoFH disease, with HoFH being the more severe and rare form of the disease.6-9 About 60% to 80% of people with FH have genetic mutations in 1 of 3 known genes: under-expression or alteration of LDLRs of hepatocytes, which encode clearance of LDL and account for approximately 85% of FH cases; defective ApoB resulting in a loss of ligand binding to LDLRs; and a gain-of-function mutation in PCSK9 resulting in more rapid degradation of LDLRs, which increases plasma LDL-C levels and which account for 5% and less than 2% of cases, respectively.21-24 The general incidence and prevalence of HeFH is likely underestimated, yet it is still believed to affect 30 million people worldwide, or 1 in 311 people, and may be higher in certain groups,25,26 particularly founder populations of French Canadians. There are an estimated 145,000 patients with FH in Canada.6,15,22,27,28
Hypercholesterolemia and ASCVD are diagnosed by a thorough physical examination, analysis of medical and family history and risk factors, and an array of laboratory tests (e.g., electrocardiogram, echocardiogram, stress tests, and cardiac catheterization) and imaging (CT, MRI). The exact cause of ASCVD remains unclear; however, there are numerous well-known risk factors that increase the chances of developing the disease. Although not the sole cause of ASCVD, elevated LDL-C is the major causal and most readily modifiable factor in developing the disease.29 Other risk factors include hypertension, smoking, diabetes, being overweight or obese, and a family history of CV disease.
Hypercholesterolemia can be defined as LDL-C greater than 190 mg/dL (> 4.9 mmol/L) or greater than 160 mg/dL (> 4.1 mmol/L) with 1 major risk factor, or greater than 130 mg/dL (> 3.4 mmol/L) with 2 CV risk factors.22 In general, LDL-C levels in adult patients with HeFH are greater than 4.9 mmol/L. A clinical diagnosis of FH is made based on high plasma levels of LDL-C, family history of hypercholesterolemia, a history of premature ASCVD, and the presence of tendon xanthomas.15,30 Left untreated, 50% of men with FH will experience an MI by the age of 50, and 30% of women by the age of 60.24 A reduction of 1 mmol/L (approximately 38.67 mg/dL) in LDL-C is estimated to reduce the relative risk of ASCVD by 20% to 22%.31
Multiple diagnostic criteria for FH exist; however, the CCS recommends the proposed criteria developed by FH Canada (Figure 1).8,27,32 A diagnosis of FH should be considered in patients with a baseline LDL-C of 5 mmol/L or greater for patients who are at least 40 years of age (or LDL-C ≥ 4.0 mmol/L for age < 18 years, or LDL-C ≥ 4.5 mmol/L for age ≥ 18 years and < 40 years). The presence of 1 or more major criteria (DNA mutation, tendon xanthomas, LDL-C ≥ 8.5 mmol/L) establishes a diagnosis of definite FH.27 Genetic testing is not necessary for diagnosis, and approximately 30% of patients with a definitive diagnosis of HeFH do not display a monogenic variant.33 The clinical expert consulted by CADTH for this review indicated that genetic testing through next-generation sequencing is not routinely available in Canadian provinces but is available in Quebec.
The 2021 dyslipidemia guidelines in Canada recommend both non-high-density lipoprotein cholesterol (non-HDL-C) and ApoB as lipid-screening parameters to improve risk stratification and management of ASCVD. In addition to LDL-C levels, both non-HDL-C and ApoB levels provide an additional assessment of the total concentration of atherogenic particles. Non-HDL-C and ApoB levels are both predictors of CV event risk and benefit from lipid-lowering therapy. Non-HDL-C is routinely reported across Canada at no additional cost, based on the simple calculation of total cholesterol minus HDL-C, and ApoB testing is available as an insured laboratory test in all provinces except Ontario.11
Figure 1: Proposed Canadian Definition of FH
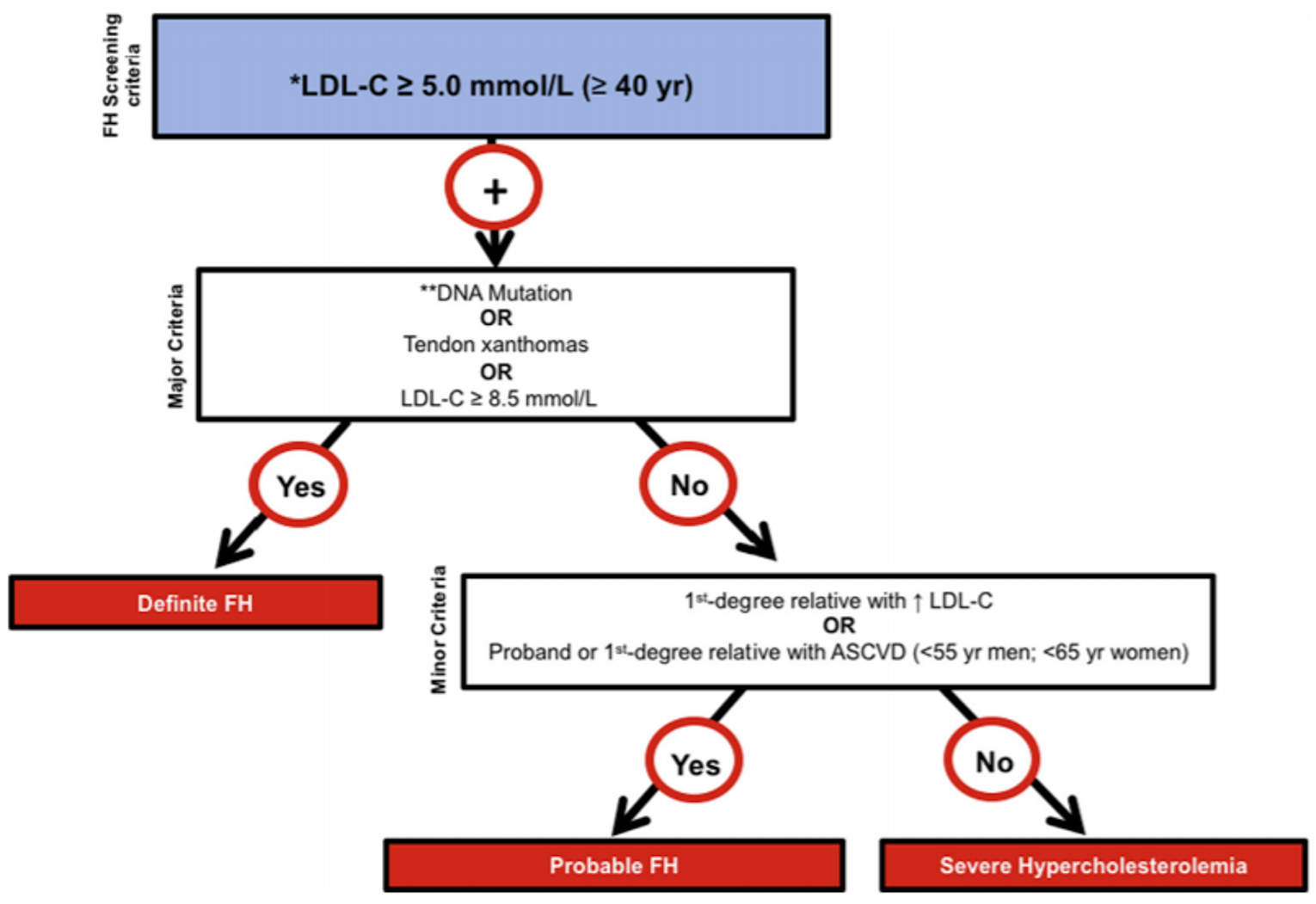
ASCVD = atherosclerotic cardiovascular disease; APOB = apolipoprotein B gene; FH = familial hypercholesterolemia; LDL-C = low-density lipoprotein cholesterol; LDLR = low-density lipoprotein receptor gene; PCSK9 = proprotein convertase subtilisin/kexin type 9 gene.
* Secondary causes of high LDL-C should be ruled out (severe or untreated hypothyroidism, nephrotic syndrome, hepatic disease [biliary cirrhosis], and medication, especially antiretroviral drugs).
** Causal DNA mutation refers to the presence of a known FH-causing variant in the LDLR, APOB, or PCSK9 gene based on the presence of the variant in ClinVar database, Human Gene Mutation Database, or Western Database of Lipid Variants in the proband or a first-degree relative.
Source: Ruel et al.34
Standards of Therapy
The management of hypercholesterolemia is divided into primary and secondary prevention, where primary prevention is the effort to prevent or delay the onset of ASCVD, while secondary prevention refers to the effort to treat known ASCVD.10 The condition of FH is associated with premature death and complications due to accelerated development of ASCVD.35
Early detection of FH is important to reduce the risk of CV events and, as such, initial non-pharmacological interventions for hypercholesterolemia include diet and lifestyle modifications. The lowering of plasma cholesterol levels is known to reduce CV events, CHD mortality, and all-cause mortality.36 Recommended lifestyle modifications, as per the CCS guidelines on the diagnosis and treatment of dyslipidemias, include weight control, reducing the amount of fat to less than 30% of daily calories, consuming 10 g to 20 g of fibre per day, and increased physical activity. Additional lifestyle changes include smoking cessation and limiting alcohol intake.8,12 Lifestyle and diet changes alone are unlikely to achieve LDL-C goals (estimated reduction in LDL-C concentration of 10% to 15%), and most patients will require pharmacological intervention.8,14,37
In addition to the recommended lifestyle changes, statins are the primary pharmacological intervention to achieve control of LDL-C in patients with hypercholesterolemia. Most patients with ASCVD should be initiated on the maximum dose of high-intensity statins (atorvastatin or rosuvastatin) with the goal of lowering LDL-C by at least 50%. Lower-intensity statins (reduced dose of atorvastatin or rosuvastatin, or simvastatin or pravastatin), which lower LDL-C by approximately 30%, should be used only in the elderly or frail or those unable to tolerate high-intensity statins.10 In cases of primary prevention where the LDL goal is unmet with statin therapy alone, add-on ezetimibe or bile acid sequestrants (or both) is recommended.10,12,13 Ezetimibe is a cholesterol absorption inhibitor that blocks the absorption of dietary cholesterol and its delivery to the liver, resulting in enhanced clearance of LDL-C, further reducing LDL-C between 10% to 40% (average 20%).14
In patients with ASCVD, if LDL goals are still not met, PCSK9 inhibitors (alirocumab and evolocumab) are available to patients meeting certain criteria as an adjunct treatment to diet, MTD, and ezetimibe.8,15 In patients with HeFH without ASCVD, Canadian guidelines recommend initiation of a PCSK9 inhibitor to lower LDL-C in patients whose LDL-C remains above the target (i.e., LDL-C ≥ 2.5 mmol/L or < 50% reduction from baseline, or ApoB ≥ 0.85 mg/dL or non-HDL-C ≥ 3.2 mmol/L) despite MTD statin therapy with or without ezetimibe therapy. In patients with HeFH and established ASCVD, PCSK9 inhibitors are to be initiated in those whose LDL-C remains above the threshold of 1.8 mmol/L or greater (or ApoB ≥ 0.7 mg/dL or non-HDL-C ≥ 2.4 mmol/L) despite MTD statin therapy, with or without ezetimibe.11 The addition of PCSK9 inhibitors can lower LDL-C up to 60% for those on statin therapy.22 Reimbursement for both alirocumab and evolocumab are variable in Canada, with most provinces funding them as limited-use options.
Drug
Inclisiran (Leqvio) is a small interfering RNA conjugated with GalNAc to facilitate uptake by hepatocytes and selectively target the liver. Through RNA interference, inclisiran directs catalytic breakdown of PCSK9 messenger RNA, preventing the production of PCSK9 protein and increasing the amount of LDLR on hepatocyte surfaces, which results in increased LDL-C uptake and reduction of the LDL-C in circulation (Figure 2).
Figure 2: Inclisiran Mechanism of Action
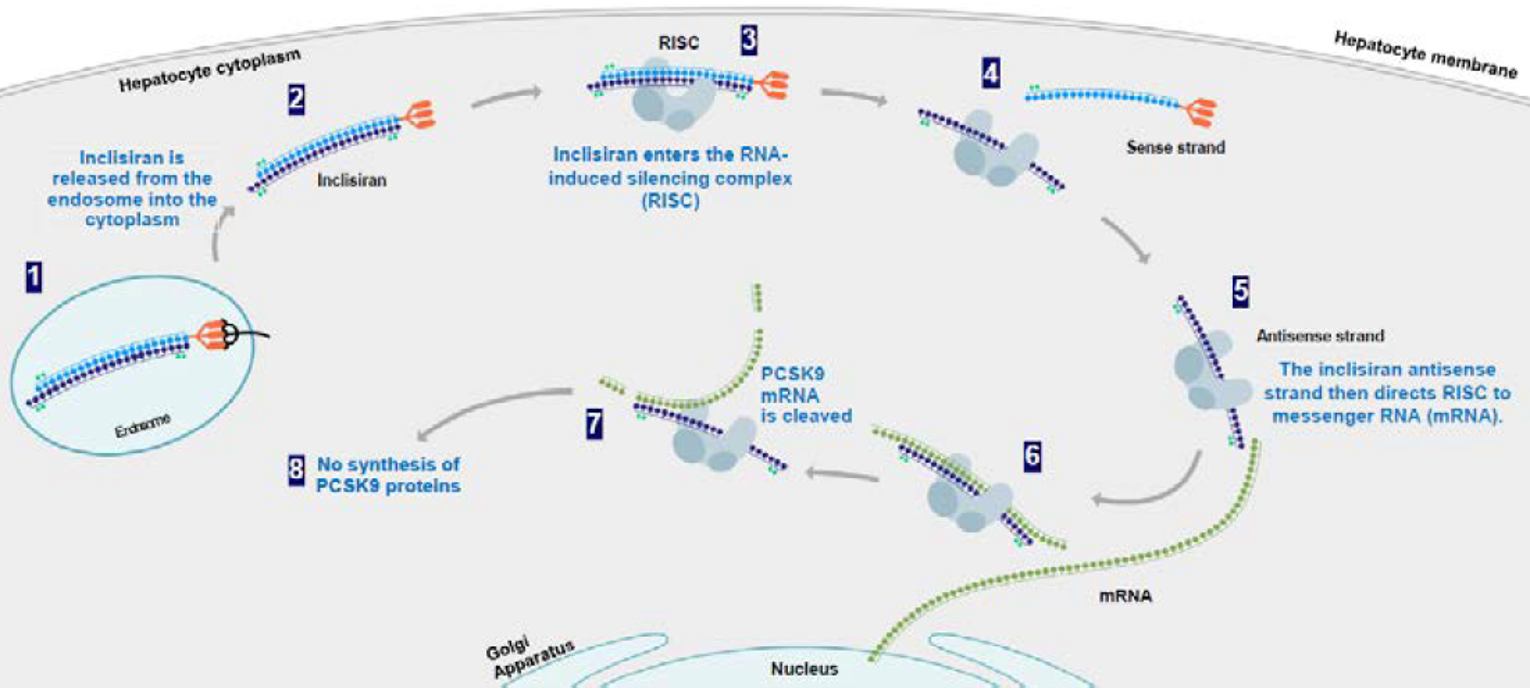
ASGPR = asialoglycoprotein receptor; GalNAc = triantennary N-acetylgalactosamine; mRNA = messenger ribonucleic acid; PCSK9 = proprotein convertase subtilisin/kexin type 9; RISC = RNA-induced silencing complex; RNA = ribonucleic acid.
Source: Sponsor submission.16
Inclisiran is administered subcutaneously by a health care professional (doctor, nurse, pharmacist) in a single 1.5 mL pre-filled syringe (equivalent to 300 mg inclisiran sodium). The recommended dose of inclisiran is 284 mg, initially and again at 3 months, followed by every 6 months thereafter.16
Inclisiran is indicated by Health Canada as an adjunct to lifestyle changes, including diet, to further reduce LDL-C levels in adults with HeFH or nFH with ASCVD who are on an MTD statin, with or without other LDL-C-lowering therapies.16 The Health Canada Notice of Compliance was granted on July 26, 2021. The European Medicines Agency granted marketing authorization to inclisiran on December 11, 2020 for the treatment of adults with hypercholesterolemia or mixed dyslipidemia. The FDA issued a complete response letter for inclisiran stating that it could not approve the new drug application due to unresolved facility inspection–related conditions at the third-party manufacturing facility. Resubmission to the FDA was conducted in July 2021, and was approved by the FDA in December 2021.38
The sponsor is requesting that inclisiran be reimbursed as an adjunct to diet and maximally tolerated statin therapy, with or without other lipid-lowering therapies, in adult patients with HeFH or clinical ASCVD who require additional lowering of LDL-C.
Table 3: Key Characteristics of Inclisiran, PCSK9 Inhibitors, Statins, and Ezetimibe
Characteristics | Inclisiran (Leqvio) | PCSK9 inhibitors | Statins | Ezetimibe |
|---|---|---|---|---|
Mechanism of action | Double-stranded siRNA conjugated with triantennary GalNAc to facilitate uptake by hepatocytes. Inclisiran directs breakdown of mRNA for PCSK9, increasing LDL-C receptor recycling and expression on the hepatocellular surface, increasing LDL-C uptake, and lowering LDL-C levels in circulation | Inhibits PCSK9, increases LDL-C receptor density | Inhibits cholesterol synthesis through inhibition of HMG-CoA reductase | Reduces cholesterol absorption by inhibiting the intestinal Niemann-Pick like 1 transporter |
Indication | As an adjunct to lifestyle changes, including diet, to further reduce LDL-C level in adults with the following conditions who are on a maximally tolerated dose of a statin, with or without other LDL-C-lowering therapies:
|
| All:
Many statins also have CV indications, such as reducing the risk of coronary events in patients with/without clinically evident CHD, reducing the risk of major cardiovascular events in patients with CHD who have undergone a PCI, and slowing the progression of coronary atherosclerosis in patients with CHD | CAD, hypercholesterolemia |
Route of administration | SC | SC | Oral | Oral |
Recommended dose | 284 mg administered as a single SC injection given initially, again at 3 months, then every 6 months thereafter |
| Various | 10 mg once daily |
Serious adverse effects or safety issues | Contraindicated in patients who are hypersensitive to this drug or any ingredient in the formulation | Hypersensitivity reactions |
|
|
ASCVD = atherosclerotic cardiovascular disease; CAD = coronary artery disease; CHD = coronary heart disease; CV = cardiovascular; GalNAc = triantennary N-acetylgalactosamine; HeFH = heterozygous familial hypercholesterolemia; HMG-CoA = 3-hydroxy-3-methylglutaryl coenzyme-A; HoFN = homozygous familial hypercholesterolemia; LDL-C = low-density lipoprotein cholesterol; mRNA = messenger ribonucleic acid; nFH = non-familial hypercholesterolemia; PCSK9 = proprotein convertase subtilisin/kexin type 9; q.2.w. = every 2 weeks; SC = subcutaneous; siRNA = small interfering ribonucleic acid.
Source: Sponsor submission,39 inclisiran product monograph,16 evolocumab Clinical Study Report.40
Stakeholder Perspectives
Patient Group Input
This section was prepared by CADTH staff based on the input provided by patient groups.
About the Patient Groups and Information Gathered
Two patient groups, the CHPA and the HeartLife Foundation, provided input for this review. The CHPA is a patient-led non-profit umbrella organization of patients, families, health professionals, and supporters dedicated to reducing CV disease and preventing early death due to cholesterol and other risk factors. Its focus is high cholesterol and other lipids, due to genetic and non-genetic factors, as the leading under-diagnosed and under-treated cause of CV disease and early death. The CHPA is the successor to the FH Canada Patient Network and collaborates with FH Canada, the Heart Healthy Prevention Program at St. Paul’s Hospital in British Columbia, and the Lipid Genetics Clinic at the London (Ontario) Health Sciences Centre’s University Hospital. The HeartLife Foundation is a patient-driven charity whose mission is to transform the quality of life of people living with heart failure by engaging, educating, and empowering a global community to create lasting solutions and build healthier lives.
The information provided by CHPA was gathered from a total of 262 individuals through an online survey (n = 254) and individual interviews (n = 8). The web-based English survey was conducted through Survey Monkey from March 18 to April 3, 2021, with a 95% completion rate. The 8 individual interviews were conducted by telephone or Zoom calls, with 6 calls in English and 2 in French. Request for participation in the survey was distributed mainly through the CHPA mailing list, the St. Paul’s Hospital Healthy Heart Program FH patient list, and various social media channels. About 95% of respondents identified themselves as residing in Canada: 61% in British Columbia, 19% in Ontario, 8% in Quebec, 3% in Alberta, and 4% in Manitoba, Saskatchewan, and Yukon. The remaining 5% were residing internationally. The online survey was directed to patients and family members affected by “high lipids,” including HeFH and ASCVD. Overall, 51% of survey respondents indicated they or a family member were affected by HeFH. A small proportion of patients identified as having HoFH (4%), symptoms but not diagnosed with FH (9%), a family history of FH (5%), ASCVD (6%), or other lipid disorders (7%). A total of 13% said they were awaiting diagnosis, unsure, or had multiple indications. The mean age of the patients (not respondents) was 59 years, and the median age was 61 years.
The information provided by the HeartLife Foundation was gathered through discussions held with their members across Canada. Members include both patients living with heart failure and their family caregivers. The discussions were held as informal group conversations (e.g., via Zoom sessions) or through phone calls with individuals. Information was gathered from a total of individuals who were between the ages of 35 and 70.
Disease Experience
As part of the online survey by CHPA, participants were asked an open-ended question regarding the impact of living with high cholesterol, high lipids, or CV disease. Approximately 95% of respondents had received or were still receiving treatment for their condition, with about 90% reporting their LDL-C level as well managed. Of note, CHPA mentioned that these respondents are probably better managed than most patients. About 80% of patient respondents were part of a CV program and received regular follow-up care by a specialist or their family physician. The responses were categorized under several non-exclusive themes:
Symptoms: About 25% reported regular physical symptoms related to their lipid levels or condition, some minor and some significant, including headaches (like icy picks), chest pains, muscle pains in legs and ankles, shortness of breath, xanthomas (under the skin in wrists, ankles, or elsewhere), weakness, fatigue, muscle loss, and neuropathy.
Managing cholesterol: About 20% also indicated that managing their cholesterol level and keeping it at target was an ongoing challenge. As 1 patient stated, “Spent 15 years trying to bring my cholesterol down with diet and exercise with little success and living in fear of having a stroke or heart attack.”
Lifestyle impact: About 20% said their high cholesterol or lipid condition had little or no effect on their quality of life. Many reported they had changed their diet and exercise. However, some responses indicated that patients were not always aware of the impact of high cholesterol, in part because they were well managed on treatment and did not experience daily symptoms. According to 1 patient, “The condition never affected my work, school or social life. It affected my family life by altering our diet.”
CV events: 75% of the FH patients reported having had at least 1 CV event or intervention, such as heart attack, stroke, angioplasty, bypass surgery, and/or stent insertion. Many have had multiple events requiring multiple interventions. Many expressed anxieties about the “unpredictability” of a CV event and the potential for severe consequences.
Psychological and family impact: Most respondents felt positive about their daily life and have accepted or adapted to living with high cholesterol, including those who have experienced a CV event or have stents. The 2 most frequently mentioned sources of anxiety were:
future uncertainty of the medications not working or the risk of a CV event
the impact on their children, whether diagnosed or at risk. According to 1 patient with FH, “It is a relatively symptom free genetic disorder. My only concerns have been related to life expectancy due to the increased risk of heart failure or stroke. I am also concerned with my 2 young children and whether they will develop the condition as they grow older.”
The HeartLife Foundation emphasized that the negative effects of ASCVD on Canadian families and the health system are significant, and that more needs to be done to address CV disease and improve health outcomes for patients.
Experience With Treatment
CHPA collected respondents’ perceptions of their treatments from the open-ended question as well as from 2 rating scales measuring the effectiveness and side effects experienced from about 13 treatments, including drug therapies, diet, and apheresis. Almost all patients on treatment were also following a low-fat diet and exercising. However, most respondents reported they were not well managed using diet or nutritional supplements alone. The majority (about 94%) of respondents had taken or were currently taking statins. In 55% of respondents, statins were reported to have worked well or very well, whereas 18% of respondents reported their cholesterol levels were not managed by statins. Severe or very severe adverse effects from statins were experienced by 25% of respondents.
About 2-thirds of respondents had an additional drug (often ezetimibe) added and about 40% to 50% had switched or added a PCSK9 inhibitor to their treatment regimen. The responses were similar, with approximately 80% who received a PCSK9 inhibitor (Repatha or Praluent) reporting that these therapies worked well or very well; 12% reported that their cholesterol levels were not managed by PCSK9 inhibitors. Between 83% (Repatha) and 95% (Praluent) of patients reported none or very few adverse effects, with 10% (Repatha) reporting some “severe” adverse effects. Less than 10% indicated that none of the drug regimens were effective. The following perspectives on treatment were shared by patients:
“At present, no drug alone or in combination has been successful in managing my cholesterol level and statins have caused severe side effects. The PCSK9 inhibiter, while tolerable without adverse side effects, has shown a small measurable decrease in cholesterol levels but not significant enough to be called successful.”
“My body does not tolerate the many statins I have used and causes significant elevations to my CK and liver levels. Hence, I have been taking PCSK9 along with low doses of Crestor, along with ezetimibe to aggressively manage the LDL levels. These drug use over the years have caused many side effects such as muscle and joint pain, low libido, sleep problems, and headaches over the years. They have led to anxiety and depression. The physical symptoms have made it difficult to engage in regular sports, exercise and personal and intimate relationships in order to enjoy a high-quality life.”
According to the HeartLife Foundation, a large proportion of the population treated with statins cannot achieve LDL-C goals. In addition, patient adherence to currently available and publicly reimbursed therapeutic options is recognized as poor. Public reimbursement for PCSK9 inhibitors in Canada is limited, and access for patients with uncontrolled LDL-C is highly restricted by provincial health benefit program reimbursement criteria.
Improved Outcomes
The majority of respondents to the CHPA survey felt they were well managed on their current therapy but also expressed multiple concerns, largely about treatment schedule, side effects, and cost.
Overall, 10% of respondents knew much or very much about inclisiran (Leqvio). Among those who did know, the response was mostly positive, with inclisiran viewed as an alternative to statins or injections of PCSK9 inhibitors every 2 weeks. Those taking statins expressed hope it would be more effective in controlling cholesterol. For those on PCSK9 inhibitors, the impact on quality of life was considered as the positive aspect. Additionally, several respondents expressed the hope that the cost of twice-yearly injections would be less than the cost of injections administered every 2 weeks. Some expressed caution based on the lack of evidence. The following perspectives on inclisiran were shared by patients:
“I hope there is a drug that could keep us patients in the ‘normal’ range. I also find an injection every 2 weeks impedes my ability to travel or be away when delivery is expected. It is not an easy drug to commit too. I would prefer to go back to a daily pill so I can make personal plans easier.
“I think it’s amazing, especially with patients who may forget to take daily medication or need health care providers. Because I work as a pilot, the daily statins is more reliable, than 2-week injections. But bi-annual injections would be great since I won’t have to remember to carry pills with me all the time between home and travel.”
“It effectively lowers LDL [but] we need to see outcome data with regards decrease HEART ATTACKS, strokes, cardiac death & total mortality.”
The survey also presented a number of paired “trade-off” decisions comparing a daily pill, self-injection every 2 weeks, and twice-yearly injections by a health care provider. Overall, respondents clearly preferred the treatment modality that was most effective (with the fewest side effects), whether pill or injection. When treatments are considered equally effective, there is a small preference for a daily pill over a self-injection every 2 weeks, but a preference for a twice-yearly injection administered by a health care provider over the daily pill. Finally, respondents (76%) preferred a twice-yearly injection administered by a health care provider to a self-injection every 2 weeks.
Additional feedback was received from 10 patients who had experience with inclisiran through clinical trials or an early access program. Four resided in Canada. All were diagnosed with ASCVD or HeFH and reported they had been on maximum statin doses without achieving target cholesterol levels. All had received inclisiran while continuing their statin regimen, and all had experienced a significant reduction in LDL levels with no adverse effects related to inclisiran. For respondents, the most important impact was knowing there was a treatment that could lower their cholesterol levels and keep them closer to target, thereby reducing the risk of further CV events. According to 1 respondent, “I feel good that I am doing something to prevent further lipid build-up and reduce risk of hardening of arteries.”
The HeartLife Foundation stated that the outcome that Canadians deserve is the ability to reduce the long-term ASCVD health consequences in high-risk patients caused by uncontrolled or poorly managed LDL-C levels. The merits of a twice-yearly dosage schedule will reduce pill burden and make it significantly easier for patients to take this drug, resulting in greater adherence and improved control of LDL-C. Many members had expressed significant interest in reducing the number of pills taken and moving toward a twice-yearly medication.
Additional Details
CHPA noted it was surprising and very gratifying to compare responses from their most recent survey to 1 that was conducted 6 years ago. Only 25% of respondents in 2015 reported that their LDL cholesterol levels were kept at desired levels, compared with 90% in the present survey. Among those not at target in 2015, about half had experienced challenges getting the statin dosage correct or access to a supplemental medication. One of the main differences in the respondent populations was the greater number of patients prescribed a PCSK9 inhibitor in the most recent survey. According to CHPA, while these respondents do not represent all Canadians living with high cholesterol, they give a clear picture of what can happen when patients receive an appropriate diagnosis, education, support, and the right medication. This patient group emphasizes that it would be extremely beneficial to introduce an alternative therapy that would be available to those not well managed on current therapies, with immediate access to those not at target, despite a maximum statin dosage.
The HeartLife Foundation noted that the number of people who suffer serious health consequences caused by ASCVD, heart attack, and ischemic strokes, and the number of people that ultimately die from the effects of CV disease, is immense. Risk factors for ASCVD need to be addressed as aggressively as possible with the tools and interventions that are available. According to the HeartLife Foundation, the product under review is 1 of these valuable interventions that should be widely available to Canadians.
Clinician Input
Input From the Clinical Expert Consulted by CADTH
All CADTH review teams include at least 1 clinical specialist with expertise regarding the diagnosis and management of the condition for which the drug is indicated. Clinical experts are a critical part of the review team and are involved in all phases of the review process (e.g., providing guidance on the development of the review protocol, assisting in the critical appraisal of clinical evidence, interpreting the clinical relevance of the results, and providing guidance on the potential place in therapy). The following input was provided by 1 clinical specialist with expertise in the diagnosis and management of HeFH and ASCVD.
Unmet Needs
In the CCS Guidelines for the Management of Dyslipidemia for the Prevention of Cardiovascular Disease and CCS Position Statement on FH, strict LDL-C thresholds have been selected based on the available evidence according to the risk of ASCVD and absolute level of LDL-C, non-HDL-C, or ApoB for the initiation of statins, ezetimibe, and PCSK9 inhibitors. Statins are considered the standard of treatment for the prevention of ASCVD in high-risk patients and patients with FH or severe hypercholesterolemia. The clinical expert pointed out that up to 15% of patients are partially or completely intolerant to statins, meaning that despite maximally tolerated statin therapy (here, the dose can be 0 mg), the patient’s LDL-C level still exceeds the threshold as described in the CCS guidelines. In these cases, ezetimibe is recommended but might be insufficient to treat optimally, following which PCSK9 inhibitors are considered. The clinical expert noted that treatment with PCSK9 inhibitors has been shown to reduce MACEs (CV death, non-fatal MI, and strokes), and there is a trend toward reduction in CV mortality. Patients unable to meet the pre-specified LDL-C thresholds with current treatments have unmet need for additional treatment options.
Place in Therapy
Decreasing atherogenic lipoprotein levels directly addresses the pathophysiology of ASCVD and modifies the natural course of the disease. The overall aim of treatment is to reduce MACEs. After lifestyle modifications, statins plus or minus ezetimibe remain the first-line approach and must be optimized before a PCSK9 inhibitor is considered. The clinical expert consulted by CADTH explained that PCSK9 inhibitors complement statin therapy and should be considered only if the patient has not reached the appropriate guideline-mandated goals of therapy. Rarely, PCSK9 inhibitors may be used alone.
Patient Population
The clinical expert also emphasized that the CCS guidelines should be used as a basis for identifying and treating patients. The clinical expert consulted by CADTH did express, however, that the characteristics of appropriate patients should match the clinical study data (i.e., age, presence of CV disease or severe hypercholesterolemia), and that stage of disease may be relevant in patients who are elderly or have end-stage disease. In the case of inclisiran, the indications would be similar to those expressed for PCSK9 inhibitors in the CCS Guidelines for the Management of Dyslipidemia for the Prevention of Cardiovascular Disease and CCS Position Statement on FH.
Lastly, the patients noted by the clinical expert as least suitable for treatment with the drug under review were those with low-risk ASCVD, those with low-risk severe hypercholesterolemia well controlled with statins, patients with ASCVD at LDL-C goals with current therapies, patients above a certain age taking the drug for primary prevention, and patients with multiple comorbidities that limit lifespan.
Assessing Response to Treatment
In line with the CCS guidelines highlighted previously, the clinical expert stated that a reduction from baseline of 50% in LDL-C and in the absolute level of LDL-C achieved below the target (LDL-C < 2.5 mmol/L for HeFH, or LDL-C < 1.8 mmol/L for ASCVD) are relevant outcomes used in clinical practice to determine response. The clinical expert indicated that treatment response should be assessed every 6 months, then yearly.
The clinical expert considered the patients most likely to exhibit a response to treatment were those that achieved a 30% to 40% reduction in LDL-C from baseline levels while on an optimized statin, with or without ezetimibe therapy.
Discontinuing Treatment
The clinical expert stated that age, end-stage disease, and/or dementia are important factors that should be considered when deciding to discontinue treatment.
Prescribing Conditions
The clinical expert noted that treatment settings would initially be specialized lipid clinics, with diagnoses and treatment confirmed by cardiologists and endocrinologists; however, throughout treatment and follow-up, treatment would migrate to internal medicine and general practice. The clinical expert also noted that in patients with (suspected) FH, genetic testing should be considered to confirm the diagnosis.
Clinician Group Input
This section was prepared by CADTH staff based on the input provided by clinician groups.
The clinician group that provided input for this project was an informal group consisting of lipid specialists and physicians working in lipid clinics in British Columbia, including the Healthy Heart Program Prevention Clinic at St. Paul’s Hospital, the Surrey Lipid Clinic at Surrey Memorial Hospital, and the Victoria Lipid Clinic. This group frequently communicates to share best practices, collaborate on research and educational projects, and meets through various forums, including advisory boards, conferences, and continuing medical education events.
The information for this submission was based on a review of relevant literature and publications as well as background knowledge in the area.
Unmet Needs
The clinician group noted the following unmet needs:
Tolerability of current treatments: Many patients have side effects to statins and ezetimibe, leading to therapy discontinuation. Therefore, therapies with lower rates of perceived side effects are needed.
Compliance: Drugs that optimize patient adherence to treatment are needed. It is well known that daily dosing regimens (of current treatments) are seldom fully adhered to.
Ability to treat to target: Despite existing therapies, many patients do not reach their guideline-recommended lipid target. This issue is increasing in importance because the latest version of many guidelines (including the 2020 CCS lipid guidelines) recommends treating LDL-C to even lower levels in high-risk patients. Add-on therapies are therefore needed to allow patients to reach their lipid targets.
Accessibility: Due to the high cost of PCSK9 inhibitors and the lack of coverage for the ASCVD indication in all Canadian provinces, therapies with greater accessibility to the large population of patients that would benefit because of their high CV risk are needed. Additionally, many patients with FH who have a high lifelong CV risk may require access to alternatives to PCSK9 inhibitors.
The clinician group described an ideal treatment option as 1 that would reduce LDL-C, non-HDL-C, and ApoB; reduce the risk of MACEs and CV mortality, and be safe and well tolerated, with properties that promote adherence.
Place in Therapy
The clinician group noted that current Health Canada–approved treatments include statins, ezetimibe, and PCSK9 monoclonal antibodies, in addition to dietary therapy consisting of reduced intake of saturated fat and dietary cholesterol. All of these therapies are routinely used in clinical practice and endorsed in the 2021 CCS dyslipidemia guidelines.
The clinician group noted that statins are likely to remain the cornerstone of therapy for these patients, given the amount of data supporting them and, as such, inclisiran would most likely be used after a patient has already been optimized (taking an MTD statin or no statin in fully statin-intolerant patients). Alternative LDL-C-lowering add-on drugs (e.g., ezetimibe) may sometimes be effective if LDL-C levels are close to optimal levels whereas, when residual LDL-C remains high, the most appropriate statin add-on would have to be more potent (e.g., PCSK9 inhibitor or inclisiran). Therefore, inclisiran would most likely be used as an add-on to MTDs of statins (and/or ezetimibe) in patients who require additional lipid lowering.
Inclisiran is a first-in-class small interfering RNA that works by inhibiting PCSK9 synthesis, thereby diminishing liver excretion into circulation and subsequently increasing the expression of hepatic LDLRs. The clinician group noted that inclisiran may displace other PCSK9 inhibitors as an add-on to statins and ezetimibe if it is more accessible than current treatments and depending on the results of currently ongoing CV outcome trials. It may also fill a void if it is approved for secondary prevention in high-risk patients.
Patient Population
The clinician group noted that patients with the greatest unmet need for intervention are those with HeFH, patients with statin intolerance, and patients with ASCVD with other markers of high risk (e.g., recent MI, coronary artery bypass graft, multi-vessel disease, polyvascular disease, diabetes, elevated lipoprotein[a]).
Based on the available data, the clinician group noted that inclisiran would be suited to all patients who require additional lowering of LDL. Patients would be identified for treatment based on their diagnosis (FH or ASCVD) and the results of lipid testing (LDL-C, non-HDL-C, ApoB), which is widely available and already used in clinical practice. In terms of patients who could benefit most from treatment with inclisiran, the clinician group noted that patients with either lifetime or short-term high risk, in particular (e.g., patients requiring secondary ASCVD prevention, patients with FH, and patients with high risk, such as those with diabetes mellitus or a high Framingham Risk Score). The clinician group also noted that asymptomatic patients should be treated in specific circumstances, such as patients with FH, patients with documented atherosclerotic disease who have not yet had a clinical event, and other guideline-recommended groups considered to have high CV risk amenable to the benefits of LDL lowering (e.g., patients with a high Framingham Risk Score, diabetes mellitus, or chronic kidney disease).
Finally, they noted that the patients least suitable for treatment with the drug under review would be patients who do not have an indication for the therapy, patients who have achieved LDL targets on other therapies (statin with or without ezetimibe), and patients who have not attempted a statin.
Assessing Response to Treatment
The clinician group noted that LDL-C, non-HDL-C, and ApoB measurements are all included in typical Canadian clinical practice and are endorsed by national guideline recommendations. A reduction in LDL-C or non-HDL-C of at least 30% would be considered a clinically meaningful response to treatment, and treatment response should be assessed at least every 6 months when beginning therapy. Treatment response could then possibly be assessed every year, once the patient is on stable treatment.
Additionally, with regard to identifying patients who are most likely to exhibit a response to treatment with inclisiran, because the response to inclisiran is fairly uniform, the issue of non-response is likely not particularly relevant.
Discontinuing Treatment
The clinician group noted that the factors to consider when deciding whether to discontinue treatment would include a lack of response (which is expected to be very rare), treatment intolerability, or if another treatment option were to become more accessible or available (e.g., a PCSK9 inhibitor).
Prescribing Conditions
The clinician group noted that the settings that would be appropriate for treatment with inclisiran include specialty clinics, community settings, and hospital or community outpatient clinics and that, in theory, inclisiran should be able to be used appropriately by both primary care and specialist physicians. In practice, however, FH is infrequently diagnosed in primary care, so identification of these patients may require a specialist who is familiar with this. Many patients with ASCVD are followed by specialists (e.g., internist, cardiologist), and it is expected that this would be the most likely scenario in which inclisiran would be initiated.
Drug Program Input
The drug programs provide input on each drug being reviewed through CADTH’s reimbursement review processes by identifying issues that may impact their ability to implement a recommendation. The implementation questions and corresponding responses from the clinical expert consulted by CADTH are summarized in Table 4.
Table 4: Summary of Drug Plan Input and Clinical Expert Response
Drug program implementation questions | Clinical expert response |
|---|---|
Is there a discrepancy between the Canadian guidelines for FH with established ASCVD (LDL-C target of < 2.0 mmol/L or < 1.8 mmol/L in those with a recent acute coronary syndrome and established coronary disease) and the enrolment criteria for the ORION studies (baseline LDL-C > 2.6 mmol/L in ORION-9 and −11, and > 1.8 mmol/L in ORION-10)? | The cut points for entry into the ORION trials are based on the guidelines. Entry criteria for the ODYSSEY (alirocumab) and FOURIER (evolocumab) trials were also LDL-C > 1.8 mmol/L. Subgroup analyses of the ODYSSEY trial suggested greater benefit in patients with LDL-C > 2.6 mmol/L; however, this was not the case for the FOURIER trial. For primary prevention of HeFH, 2.6 mmol/L (> 2.5 mmol/L) is a reasonable threshold for considering escalation of therapy; while, in ASCVD, 1.8 mmol/L is considered a reasonable threshold for escalation of therapy. |
There is a discrepancy in the definition of adherence to MTD of statins used by jurisdictions. What is considered “adherent” to MTD statins? | Optimization of statin treatment generally occurs over a 3-month period. If patients do not achieve threshold LDL-C levels after 3 months, then application for reimbursement of PCSK9 inhibitors is warranted. Three months is also an appropriate follow-up for appointments with physicians in this population. Private insurance companies and INESSS consistently use a 40% reduction in LDL-C from baseline over 3 months as evidence of therapy (proof of efficacy) with statins and/or PCSK9 inhibitors. As a general rule, this is considered reasonable. |
Can laboratory outcomes be confirmed as effective in the real world? | Yes. LDL-C is still used as a surrogate biomarker for CV outcomes; thus, clinical trials are designed to achieve certain thresholds. Guidelines suggest non-HDL-C can also be measured, which encompasses atherogenic lipoproteins and ApoB; however, 90% of hypercholesterolemia is driven by LDL-C levels. In the blood, triglyceride-rich lipoproteins also carry cholesterol (also known as non-LDL-C [total cholesterol minus HDL-C]) and increase CV risk. Guidelines are recommending this measure in patients with diabetes, metabolic syndrome, and so forth, and is equivalent to ApoB. |
Are the initiation criteria for inclisiran consistent with other PCSK9 inhibitors for HeFH? Should ezetimibe or other non-statin lipid-lowering therapies be used before starting inclisiran? | Initiation criteria would be in line with other PCSK9 inhibitors. If a patient is within 20% of the CCS guideline threshold (LDL-C < 2.5 mmol/L for HeFH without clinical ASCVD, or LDL-C < 1.8 mmol/L for patients with HeFH and ASCVD) on MTD statin, then initiation of ezetimibe is warranted first; however, if the patient is not within 20% of the designated threshold, then PCSK9 inhibitors would be initiated. |
Are the renewal criteria for inclisiran consistent with other PCSK9 inhibitors? | Treatment would be considered for discontinuation in the elderly, those with very severe end-stage kidney or heart failure where lipid therapy will not alter outcomes. Otherwise, patients are expected to be on treatment for a long time; potentially lifelong. |
In the case of primary prevention, would an event (heart attack or stroke) be considered treatment failure and warrant discontinuation of treatment with inclisiran? | No. During the course of a clinical trial, patients may still experience 2 or more clinical events (e.g., stroke, MI, need for revascularization). Subgroup analyses of other trials for PCSK9 inhibitors (FOURIER and ODYSSEY) showed that PCSK9 inhibitors reduced the total number of events. Thus, even with an event, therapy should be continued. |
Should inclisiran be initiated in patients with hypercholesterolemia who have had a prior heart attack or stroke (i.e., for secondary prevention)? | Yes. This refers to the ASCVD indication for inclisiran and should therefore be initiated in these patients. |
Is it expected that inclisiran would be used in patients who do not have hypercholesterolemia or did not have a prior heart attack or stroke (i.e., for primary prevention)? | Inclisiran should not be used in patients who do not have hypercholesterolemia or did not have a prior heart attack or stroke (i.e., for primary prevention). Some very rare disorders display elevated LDL-C caused by other diseases (e.g., hypothyroidism, nephrotic syndrome, severe liver disease); however, the underlying disease should be addressed first, and these are not relevant to this review. |
ApoB = apolipoprotein B; ASCVD = atherosclerotic cardiovascular disease; CCS = Canadian Cardiovascular Society; CV = cardiovascular; FH = familial hypercholesterolemia; HDL-C = high-density lipoprotein cholesterol; HeFH = heterozygous familial hypercholesterolemia; INESSS = Institut national d’excellence en santé et en services sociaux; LDL-C = low-density lipoprotein cholesterol; MI = myocardial infarction; MTD = maximally tolerated dose; PCSK9 = proprotein convertase subtilisin/kexin type 9.
Clinical Evidence
The clinical evidence included in the review of inclisiran (Leqvio) is presented in 3 sections. The first section, the systematic review, includes pivotal studies provided in the sponsor’s submission to CADTH and Health Canada, as well as those studies that were selected according to an a priori protocol. The second section includes indirect evidence from the sponsor and indirect evidence selected from the literature that met the selection criteria specified in the review. The third section includes sponsor-submitted long-term extension studies and additional relevant studies that were considered to address important gaps in the evidence included in the systematic review.
Systematic Review (Pivotal and Protocol Selected Studies)
Objectives
To perform a systematic review of the beneficial and harmful effects of inclisiran in adults with primary hypercholesterolemia (HeFH and nFH).
Methods
Studies selected for inclusion in the systematic review included pivotal studies provided in the sponsor’s submission to CADTH and Health Canada, as well as those meeting the selection criteria presented in Table 5. Outcomes included in the CADTH review protocol reflect outcomes considered to be important to patients, clinicians, and drug plans.
Of note, the systematic review protocol presented in Table 5 was established before the granting of a Notice of Compliance from Health Canada.
Table 5: Inclusion Criteria for the Systematic Review
Criteria | Description |
|---|---|
Population | As an adjunct to diet and maximally tolerated statin therapy, with or without other lipid‐lowering therapies, in adult patients with HeFH or clinical ASCVD who require additional lowering of LDL‐C Subgroups:
|
Intervention | Inclisiran 284 mg SC injection (initially, at 3 months, then every 6 months thereafter) alone or in combination with a statin, a statin ± other lipid-lowering therapies, or other lipid-lowering therapies alone |
Comparator | HeFH:
ASCVD:
|
Outcomes | Efficacy outcomes:
Harms outcomes:
|
Study designs | Published and unpublished phase III and IV RCTs |
AE = adverse event; ApoB = apolipoprotein B; ASCVD = atherosclerotic cardiovascular disease; CK = creatinine kinase; CV = cardiovascular; HDL-C = high-density lipoprotein cholesterol; HeFH = heterozygous familial hypercholesterolemia; HRQoL = health-related quality of life; LDL-C = low-density lipoprotein cholesterol; MI = myocardial infarction; PCI = percutaneous coronary intervention; PCSK9 = proprotein convertase subtilisin/kexin type 9; RCT = randomized controlled trial; SAE = serious adverse event; SC = subcutaneous; WDAE = withdrawal due to adverse event.
The literature search for clinical studies was performed by an information specialist using a peer-reviewed search strategy according to the PRESS Peer Review of Electronic Search Strategies checklist.41
Published literature was identified by searching the following bibliographic databases: MEDLINE All (1946‒) through Ovid and Embase (1974‒) through Ovid. The search strategy comprised both controlled vocabulary, such as the National Library of Medicine’s MeSH (Medical Subject Headings), and keywords. The main search concept was inclisiran. Clinical trials registries were searched: the US National Institutes of Health’s clinicaltrials.gov, the WHO’s International Clinical Trials Registry Platform (ICTRP) search portal, Health Canada’s Clinical Trials Database, and the European Union Clinical Trials Register.
No filters were applied to limit the retrieval by study type. Retrieval was not limited by publication date or by language. Conference abstracts were excluded from the search results. See Appendix 1 for the detailed search strategies.
The initial search was completed on April 15, 2021. Regular alerts updated the search until the meeting of the CADTH Canadian Drug Expert Committee on August 18, 2021.
Grey literature (literature that is not commercially published) was identified by searching relevant websites from the Grey Matters: A Practical Tool for Searching Health-Related Grey Literature checklist.42 Included in this search were the websites of regulatory agencies (FDA and European Medicines Agency). Google was used to search for additional internet-based materials. See Appendix 1 for more information on the grey literature search strategy.
Two CADTH clinical reviewers independently selected studies for inclusion in the review based on titles and abstracts, according to the predetermined protocol. Full-text articles of all citations considered potentially relevant by at least 1 reviewer were acquired. Reviewers independently made the final selection of studies to be included in the review, and differences were resolved through discussion.
Findings From the Literature
A total of 3 studies were identified from the literature for inclusion in the systematic review (Figure 3).17-19 The included studies are summarized in Table 6. A list of excluded studies is presented in Appendix 2.
Table 6: Details of Included Studies
Detail | ORION-9 | ORION-10 | ORION-11 |
|---|---|---|---|
Designs and populations | |||
Study design | Double-blind, phase III RCT | Double-blind, phase III RCT | Double-blind, phase III RCT |
Locations | US, Europe, South Africa, Canada | US | Europe, South Africa |
Patient enrolment dates | December 12, 2017 to August 27, 2019 | December 2, 2017 to September 10, 2019 | November 1, 2017 to July 31, 2019 |
Randomized (N) | N = 482 | N = 1,561 | N = 1,617 |
Inclusion criteria | History of HeFH with a diagnosis of HeFH by genetic testing and/or a documented history of untreated LDL-C of > 4.9 mmol/L (190 mg/dL) and a family history of FH, elevated cholesterol, or early heart disease, which may indicate FH Serum LDL-C ≥ 2.6 mmol/L (≥ 100 mg/dL) at screening | History of ASCVD (CHD, CVD, or PAD) Serum LDL-C ≥ 1.8 mmol/L (≥ 70 mg/dL) No current or planned renal dialysis or renal transplantation | History of ASCVD (CHD, CVD, or PAD) or an ASCVD risk equivalent (type 2 diabetes, FH) and patients whose 10-year risk of a CV event (as assessed by a Framingham Risk Score or equivalent) has a target LDL-C of < 2.6 mmol/L (< 100 mg/dL) Serum LDL-C ≥ 1.8 mmol/L (≥ 70 mg/dL) for patients with ASCVD or ≥ 2.6 mmol/L (≥ 100 mg/dL) for patients with an ASCVD risk equivalent at screening |
| |||
Exclusion criteria |
| ||
(continued) |
| ||
Drugs | |||
Intervention | Inclisiran sodium 300 mg/1.5 mL solution (equivalent to 284 mg inclisiran) for SC injection administered initially, at 3 months, and every 6 months thereafter | Inclisiran sodium 300 mg/1.5 mL solution (equivalent to 284 mg inclisiran) or pre-filled syringes for SC injection administered initially, at 3 months, and every 6 months thereafter | Inclisiran sodium 300 mg/1.5 mL solution per vial (equivalent to 284 mg inclisiran) for SC injection administered initially, at 3 months, and every 6 months thereafter |
Comparator(s) | Matching placebo (sterile normal saline [0.9% sodium chloride in water for injection]) in vials for SC injection | Matching placebo (sterile normal saline [0.9% sodium chloride in water for injection]) for SC injection in vials and pre-filled syringes | Matching placebo (sterile normal saline [0.9% sodium chloride in water for injection]) in vials for SC injection |
Duration | |||
Phase | |||
Screening | 14 days before first dose of study drug | ||
Double-blind | 540 days (18 months) | ||
Follow-up | Day 540 (90 days post final dose) | ||
Outcomes | |||
Primary end point |
| ||
Secondary and exploratory end points | Secondary:
| ||
(continued) |
Proportion of patients in each group who attain global lipid targets for their level of ASCVD risk Exploratory:
Response of LDL-C reduction by underlying causal mutations of HeFH (ORION-9 only) | ||
Notes | |||
Publications | Raal et al. (2020)35 | Ray et al. (2020)43 | Ray et al. (2020)43 |
AE = adverse event; ALT = alanine aminotransferase; ApoB = apolipoprotein B; ASCVD = atherosclerotic cardiovascular disease; AST = aspartate aminotransferase; CHD = coronary heart disease; CVD = cardiovascular disease; DBP = diastolic blood pressure; eGFR = estimated glomerular filtration rate; FH = familial hypercholesterolemia; HDL-C = high-density lipoprotein cholesterol; HeFH = heterozygous familial hypercholesterolemia; LDL-C = low-density lipoprotein cholesterol; MACE = major cardiovascular event; MI = myocardial infarction; MTD = maximally tolerated dose; NYHA = New York Heart Association; PAD = peripheral artery disease; PCSK9 = proprotein convertase subtilisin/kexin type 9; RCT = randomized controlled trial; SBP = systolic blood pressure; SC = subcutaneous; TBIL = total bilirubin; TC = total cholesterol; ULN = upper limit of normal.
Source: Clinical Study Reports for ORION-9,17 ORION-10,18 and ORION-11.19
Description of Studies
A total of 3 studies were included in this review: ORION-9, ORION-10, and ORION-11.17-19 The included studies were all phase III, double-blind RCTs comparing inclisiran with placebo in patients with HeFH or ASCVD (and/or an ASCVD risk equivalent) who were receiving MTD statins or were statin-intolerant. All 3 studies were funded by the sponsor.
ORION-9 was a phase III, double-blind, multinational, randomized, placebo-controlled trial designed to assess the efficacy, safety, and tolerability of inclisiran 284 mg versus placebo in patients with HeFH and elevated LDL-C despite maximum tolerated dose of LDL-C-lowering therapies. ORION-9 was conducted in 47 centres in 8 countries: Canada, Czech Republic, Denmark, Netherlands, South Africa, Spain, Sweden, and the US. Three Canadian study centres were included in the ORION-9 study, which enrolled 23 patients.17,35
ORION-10 was a phase III, double-blind, multi-centre, randomized, placebo-controlled trial designed to assess the efficacy, safety, and tolerability of inclisiran 284 mg versus placebo in patients with ASCVD (CHD, CV disease, PAD) and elevated LDL-C despite an MTD of LDL-C-lowering therapies. ORION-10 was conducted in 146 sites across the US. No Canadian study sites or patients were included in the ORION-10 study.18,43
ORION-11 was an international, multi-centre, phase III, placebo-controlled, double-blind, randomized study to determine the efficacy, safety, and tolerability in patients with ASCVD or an ASCVD risk equivalent (e.g., diabetes and FH) and elevated LDL-C despite MTD statin therapy. The study was conducted within 72 centres in European countries and South Africa. No Canadian study sites or patients were included in the ORION-11 study.19,43
In all 3 ORION studies, patients were randomized 1:1 to either inclisiran sodium 300 mg or placebo on top of MTD statin. The ORION-9, −10, and −11 trials enrolled 482, 1,561, and 1,617 patients, respectively. The studies were all 18 months in duration, with patients receiving a 300 mg dose of inclisiran sodium on 4 days: day 1, day 90, day 270, and day 450. The ORION-9, −10, and −11 trials shared an identical study design, which is displayed in Figure 4.17-19
Figure 4: Schematic Overview of the ORION-9, ORION-10, and ORION-11 Trial Schema
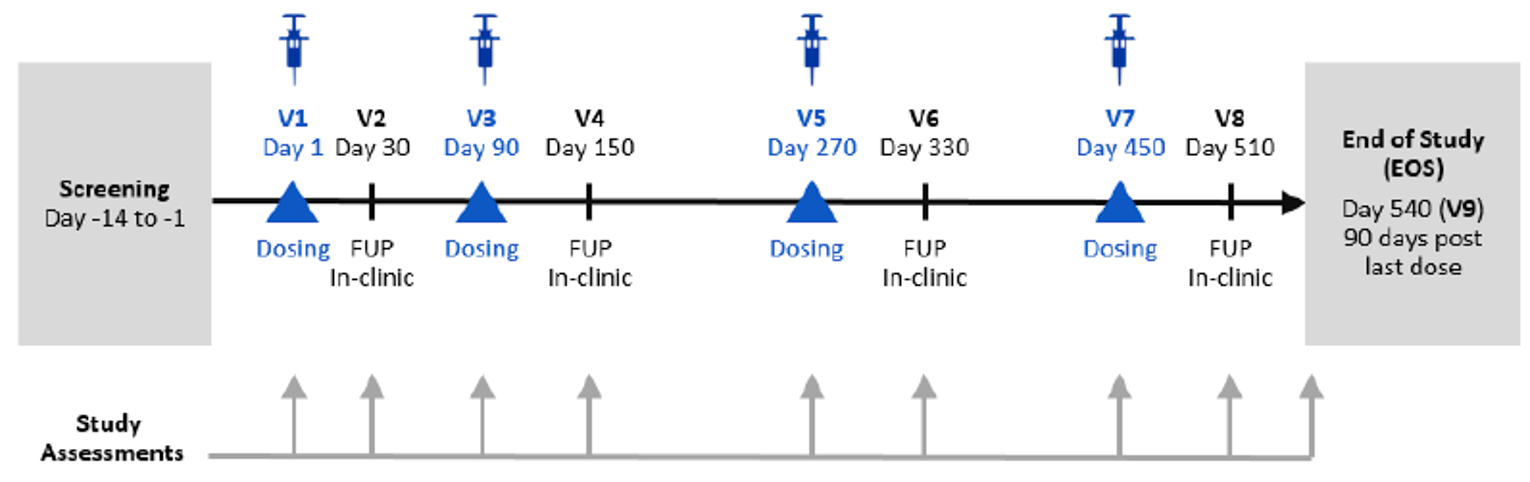
EOS = end of study; FUP = follow up; V = visit.
Source: Clinical Study Reports for ORION-9,17 ORION-10,18 and ORION-11.19
Key eligibility criteria for the ORION-9, −10, and −11 studies are summarized in Table 6. Briefly, eligible patients in ORION-9 included adults (≥ 18 years) with a history of HeFH and/or a documented history of untreated high LDL-C (> 190 mg/dL), and a family history of FH, elevated cholesterol, or early heart disease, which may indicate FH. Patients eligible for ORION-10 were adults (≥ 18 years) with a history of ASCVD. Patients eligible for ORION-11 were adults (≥ 18 years) with a history of ASCVD or an ASCVD risk equivalent.17-19
The minimum follow-up and duration of all 3 ORION trials was 18 months. The last observation and data cut-off (end of study) for the ORION-9, −10, and −11 trials was at visit 9 (day 540), which was 90 days following the last dose of the study drug at visit 8 (day 450).17-19
Populations
Inclusion and Exclusion Criteria
The inclusion and exclusion criteria for the 3 ORION trials are summarized in Table 6. Overall, the 3 trials had similar inclusion criteria, with slight differences specific to the included population and disease of interest. In the ORION-9 trial, eligible patients were required to have a family history of HeFH, elevated cholesterol, or early heart disease, or HeFH diagnosed by genetic testing or phenotypic Simon Broome criteria and/or a documented history of untreated high LDL-C (> 190 mg/dL).17 In contrast, the ORION-10 study enrolled patients with a history of ASCVD, including CHD, CV disease, or PAD.18 Lastly, the ORION-11 trial included patients with a history of ASCVD (CHD, CV disease, or PAD) or an ASCVD risk equivalent (e.g., type II diabetes, or FH).19 All trials required patients to be receiving the MTD of a statin, defined as the maximum dose that can be taken on a regular basis without AEs; for those not receiving a statin, documented evidence of intolerance to all doses of at least 2 different statins was required.17-19
The trials shared the same exclusion criteria. Patients were ineligible if they had New York Heart Association (NYHA) class IV heart failure or a last known left ventricular ejection fraction of less than 25%, cardiac arrhythmia within 3 months before randomization that was not controlled by medication or through ablation, or an MACE within 3 months before randomization.17-19
Baseline Characteristics
Baseline characteristics for each included trial are summarized in Table 7. Baseline characteristics were well balanced across groups in each trial. Patients enrolled in the ORION-10 and −11 trials were more similar than those enrolled in the ORION-9 trial.
In ORION-9, approximately half of the included patients (227; 47.1%) were male, and the median age was 56 years old.17 The majority of patients in ORION-10 and −11 were male (69.4% and 71.7%, respectively), and the mean age was 65 to 67 years.18,19 The majority of patients in all 3 trials were White (range = 83.6 to 98.6%).17-19 The mean and median age of patients in the ORION-9 trial were approximately 10 years younger than those included in the ORION-10 and −11 trials, and the majority of patients were 65 years of age or older in the ORION-10 (n = 931; 59.6%) and ORION −11 (n = 884; 54.7%) trials compared with the ORION-9 trial, where the majority were younger than 65 years (n = 374; 77.6%).17-19 Across all 3 trials, the majority of patients also had renal impairment (63.5%, 76.4%, and 71.1% in ORION-9, −10, and −11, respectively).17-19
In ORION-9, 350 patients (72.6%) had an ASCVD risk equivalent, of which 238 (93.2%) had HeFH, while 132 (27.4%) had ASCVD. Half of the patients had baseline LDL-C levels below, and half above, 138.5 mg/dL, with a mean of 151.4 mg/dL and 154.7 mg/dL in the inclisiran and placebo groups, respectively.35 Conversely, in ORION-10, all patients had ASCVD, and in ORION-11, 1,414 (87.5%) had ASCVD and 203 (12.6%) had an ASCVD risk equivalent, of which only 11 (5.4%) had HeFH. In these trials, patients were evenly balanced for mean baseline LDL-C levels, with a mean LDL-C level of 104.5 mg/dL and 107.2 mg/dL in the inclisiran groups, and 104.8 mg/dL and 103.7 mg/dL in the placebo groups.43 The ASCVD risk–equivalent population of ORION-11 was not of interest to this review, as this population was not included in the reimbursement request. Of those with ASCVD, the proportion of patients with |||||||||| was |||||||||| in the ORION-9 trial, occurring in |||||||||| of patients in the placebo group and |||||||||| in the inclisiran group. Conversely, the ORION-10 and −11 trials reported |||||||||||||||||||| of patients with |||||||||||| ranging from ||||||||||, CV disease ranging from ||||||||||||||||, and |||||||||| ranging from |||||||||||||||||| compared with |||||||||||||||||||||||||||| in ORION-9. Lower rates of |||||||||||||||||||||||| hypertension were seen in the ORION-9 trial (|||||||||| 42.1%, ||||||||||||||), compared with the ORION-10 and −11 trials (|||||||||||||||||||||||||| 90.6%, and 80.5%, respectively).17-19
In ORION-9, 356 patients (73.9%) were on a high-intensity statin at baseline, and 135 patients (55.8%) and 120 patients (50.0%) in the inclisiran and placebo groups, respectively, were treated with ezetimibe.17,35 In ORION-10, 1,084 patients (69.4%) were on high-intensity statins at baseline, and a total of 80 (10.2%) and 74 patients (9.5%) in the inclisiran and placebo groups, respectively, were treated with ezetimibe.18,43 In ORION-11, 1,261 patients (78.0%) were on a high-intensity statin at baseline based on the FDA feedback on September 13, 2019, where simvastatin 40 mg was considered a moderate-intensity statin. A total of 52 (6.4%) and 62 patients (7.7%) in the inclisiran and placebo groups, respectively, were treated with ezetimibe.19,43
In the ORION-9, −10, and −11 trials, 122 (25.3%), 344 (22.0%), and 185 patients (11.4%) were partially or completely intolerant to statins, respectively.
Table 7: Summary of Baseline Characteristics (ITT Populations)
Characteristic | ORION-9 | ORION-10 | ORION-11 | |||
|---|---|---|---|---|---|---|
Inclisiran (N = 242) | Placebo (N = 240) | Inclisiran (N = 781) | Placebo (N = 780) | Inclisiran (N = 810) | Placebo (N = 807) | |
Age (years) | ||||||
Mean (SD) | 54.4 (12.48) | 55.0 (11.81) | 66.4 (8.90) | 65.7 (8.89) | 64.8 (8.29) | 64.8 (8.68) |
Median (range) | 56.0 (22 to 79) | 56.0 (21 to 80) | 67.0 (35 to 90) | 66.0 (39 to 89) | 66.0 (20 to 88) | 65.0 (34 to 87) |
18 to < 65, n (%) | 189 (78.1) | 185 (77.1) | 297 (38.0) | 333 (42.7) | 367 (45.3) | 366 (45.4) |
≥ 65, n (%) | 53 (21.9) | 55 (22.9) | 484 (62.0) | 447 (57.3) | 443 (54.7) | 441 (54.6) |
Sex, n (%) | ||||||
Male | 112 (46.3) | 115 (47.9) | 535 (68.5) | 548 (70.3) | 579 (71.5) | 581 (72.0) |
Female | 130 (53.7) | 125 (52.1) | 246 (31.5) | 232 (29.7) | 231 (28.5) | 226 (28.0) |
Weight (kg), mean (SD) | 83.7 (18.55) | 84.2 (18.27) | 92.2 (19.60) | 93.6 (21.57) | 85.8 (16.24) | 87.6 (18.19) |
BMI (kg/m2), mean (SD) | 29.0 (5.68) | 28.8 (5.09) | 31.5 (6.25) | 31.8 (6.44) | 29.7 (4.79) | 30.2 (5.15) |
Race, n (%) | ||||||
White | 226 (93.4) | 227 (94.6) | 653 (83.6) | 685 (87.8) | 791 (97.7) | 796 (98.6) |
Black or African American | 8 (3.3) | 7 (2.9) | 110 (14.1) | 87 (11.2) | 12 (1.5) | 8 (1.0) |
Asian | 7 (2.9) | 5 (2.1) | 9 (1.2) | 1 (0.1) | 6 (0.7) | 2 (0.2) |
Other | 1 (0.4) | 1 (0.4) | 9 (1.2) | 7 (0.9) | 1 (0.1) | 1 (0.1) |
Smoking status, n (%) | ||||||
Current | 28 (11.6) | 28 (11.7) | 123 (15.7) | 111 (14.2) | 160 (19.8) | 132 (16.4) |
||||||| | ||||||| | |||||||| | |||||||| | ||||||| | |||||||| | ||||||| |
||||||| | |||||||| | ||||||||| | |||||||| | |||||||| | |||||||| | |||||| |
||||||||||||||||||||||||||||||||||||||| | ||||||
|||||||||||| | ||||||| | ||||||| | |||||||| | |||||||| | ||||||| | ||||||| |
|||||||||||| | ||||||| | ||||||| | |||||||| | ||||||||| | ||||||| | ||||||| |
|||||||||||| | ||||||| | ||||||| | |||||||| | |||||||| | ||||||| | ||||||| |
|||||||||||||||| | ||||||| | ||||||| | |||||||| | ||||||||| | ||||||| | ||||||| |
|||||||||||||||||||| | ||||||| | ||||||| | |||||||| | |||||||| | ||||||| | ||||||| |
|||||||||||||||| | ||||||| | ||||||| | |||||||| | ||||||||| | ||||||| | ||||||| |
Baseline GFR (mL/min/1.73 m2) | ||||||
Mean (SD) | 86.3 (20.41) | 83.8 (19.33) | 75.6 (22.27) | 76.1 (22.05) | 80.0 (19.23) | 79.1 (19.54) |
|||||||||||||||||| | |||||||||||||||| | |||||||||||||||| | |||||||||||||| | |||||||||||||| | |||||||||||||| | |||||||||||| |
Hypertension, n (%) | ||||||
Yes | 102 (42.1) | 101 (42.1) | 714 (91.4) | 701 (89.9) | 640 (79.0) | 661 (81.9) |
No | 140 (57.9) | 139 (57.9) | 67 (8.6) | 79 (10.1) | 170 (21.0) | 146 (18.1) |
ASCVD type, n (%) | ||||||
ASCVD | 59 (24.4) | 73 (30.4) | 781 (100) | 780 (100) | 712 (87.9) | 702 (87.0) |
ASCVD risk equivalent | 183 (75.6) | 167 (69.9) | NA | NA | 98 (12.1) | 105 (13.0) |
Prior or current CHD, n (%) | ||||||
||||| | |||||| | |||| | |||||| | ||||||||| | |||||||||||||||||||| | |||||||||||||||||| |
||||||||||| | |||||||||||| | |||||||||| | |||||| | |||||||||||||||||||| | |||||||||||||||| | |||||||||||||||| |
|||||| | ||||||||||||| | |||||||||||||||||| | |||||| | |||||||||||||||||| | |||||||||||||||| | ||||||||||||||||||| |
|| | |||||| | |||||||| | |||| | ||| | |||||| | |||| |
|||||||||||||||||||||||||||||||||||||||||||||||| | ||||||
|||| | || | |||||| | |||||| | |||||| | ||||||||| | |||||||| |
|||||||||||||||||||||||||| | |||||| | |||||||| | |||||| | ||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||| |
||| | ||||||||| | ||||||||||| | |||||||| | |||||| | |||||||||| | |||||| |
||||||||||||||||||||||||||||||||||||||||||| | ||||||
|||| | || | || | |||||| | ||||| | |||||| | |||||| |
|| | |||||| | |||||||| | |||||| | |||||| | |||||||| | |||||| |
LMT use | ||||||
|||||||||||||| | ||||||| | |||||||| | ||||||| | ||||||| | ||||||| | |||||| |
Statin use at baseline, n (%) | 219 (90.5) | 217 (90.4) | 701 (89.8) | 692 (88.7) | 766 (94.6) | 766 (94.9) |
High intensity | 185 (76.4) | 171 (71.3) | 538 (68.9) | 546 (70.0) | 633 (78.1) | 628 (77.8) |
Moderate intensity | 30 (12.4) | 40 (16.7) | 152 (19.5) | 140 (17.9) | 123 (15.2) | 128 (15.9) |
Low intensity | 5 (2.1) | 4 (1.7) | 6 (0.8) | 6 (0.8) | 3 (0.4) | 3 (0.4) |
Statins only, n (%) | 102 (42.1) | 86 (35.8) | 508 (65.0) | 481 (61.7) | 688 (84.9) | 686 (85.0) |
Other LMT, n (%) | 127 (52.5) | 140 (58.3) | 240 (30.7) | 249 (31.9) | 96 (11.9) | 95 (11.8) |
Other LMT only, n (%) | 10 (4.1) | 9 (3.8) | 47 (6.0) | 38 (4.9) | 18 (2.2) | 15 (1.9) |
ASCVD = atherosclerotic cardiovascular disease; BMI = body mass index; CHD = coronary heart disease; CVD = cerebrovascular disease; GFR = glomerular filtration rate; ITT = intention to treat; LMT = lipid-modifying therapy; NA = not applicable; PAD = peripheral artery disease; SD = standard deviation.
aValues for ORION-11 are for ASCVD patients only (N = 702).
Source: Clinical Study Reports for ORION-9,17 ORION-10,18 and ORION-11.19,43
Interventions
Treatments Administered
ORION-9, −10, and −11 were all double-blind RCTs. Patients were randomized 1:1 by automated interactive response technology to receive either inclisiran sodium subcutaneous injection as the investigational product in all 3 ORION trials, delivered at a dose of 284 mg or placebo as normal sterile saline (0.9% sodium chloride in water for injection). Patients were administered inclisiran or placebo as a single subcutaneous injection in the abdomen for a total of 4 doses, on study days 1, 90, 270, and 450.17-19
Inclisiran was provided as a unit dose of 300 mg/1.5 mL per 2 mL glass vial, and placebo was provided in a matching glass vial, both manufactured by Alcami (formerly AAI Pharma) prepared and administered by a pharmacist or qualified clinical study site staff. Inclisiran was also administered as a 2.25 mL pre-filled syringe for doses 3 and 4 in ORION-10.17-19
Treatment allocation was stratified by country and by current use of statins or other lipid-modify therapies (LMTs) in block sizes of 4. The principal investigators were authorized to unblind a patient through interactive response technology in the event of an emergency or AE for which it was necessary to know the study drug to determine an appropriate course of therapy. The investigator was advised not to reveal the study treatment assignment to any other site or sponsor personnel.17-19
Patients were required to fast for at least 8 hours for all visits for fasting lipids and glucose blood samples, refrain from unaccustomed strenuous physical exercise for 48 hours before the screening and any study visit until the follow-up had been completed, and were not permitted to donate blood at any time during the study.17-19
Prior and Concomitant Therapy
Eligible prior and concomitant medications for all 3 ORION trials included statins and other lipid-lowering therapies (e.g., ezetimibe); patients were required to have been on a stable dose for a minimum of 30 days before screening. Patients receiving statins were to be receiving the MTD. Hormone replacement therapy, prescription medications to treat pre-existing medical conditions such as diabetes or hypertension, and prescription and non-prescription medications to treat AEs at the discretion of the investigator were also permitted.17-19
All of the trials prohibited adding any medications that are prescribed to lower LDL-C during the trial. This included statins, ezetimibe, lomitapide, mipomersen, niacin, colesevelam, bile acid absorption inhibitors, monoclonal antibodies directed toward PCSK9, and any medications taken for the purpose of lipid lowering, including over-the-counter and herbal therapies.17-19
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Outcomes
The efficacy end points identified in the CADTH review protocol that were assessed in the clinical trials included in this review are listed and summarized in Table 8.
Table 8: Summary of Outcomes of Interest Identified in the CADTH Review Protocol
Outcome measure | ORION-9, ORION-10, ORION-11 efficacy end points by type |
|---|---|
Percentage change in LDL-C from baseline to day 510 | Primary |
Time-adjusted percentage change in LDL-C from baseline after day 90 and up to day 540 | Primary |
Absolute change in LDL-C from baseline to day 510 | Key secondary |
Time-adjusted absolute change in LDL-C from baseline after day 90 and up to day 540 | Key secondary |
Percentage change from baseline to day 510 in PCSK9, TC, ApoB, and non-HDL-C | Key secondary |
Maximum percentage change in LDL-C | Secondary |
Absolute change from baseline to day 510 in PCSK9, total cholesterol, ApoB, and non-HDL-C from baseline | Secondary |
Absolute and percentage change in LDL-C from baseline to each assessment time up to day 540 | Secondary |
Individual responsiveness, defined as the number of patients reaching on-treatment LDL-C levels of < 25 mg/dL, < 50 mg/dL, < 70 mg/dL, and < 100 mg/dL at day 510 | Secondary |
Proportion of patients in each group with an LDL-C reduction from baseline of 50% or greater | Secondary |
Absolute and percentage change in other lipids, lipoproteins, apolipoproteins, and PCSK9 from baseline at each subsequent visit to day 540 | Secondary |
Proportion of patients in each group who attain global lipid targets for their level of ASCVD risk | Secondary |
Incidence of cardiovascular death, resuscitated cardiac arrest, non-fatal MI, and non-fatal stroke (ischemic and hemorrhagic) | Exploratory |
Proportion of patients in each group with any LDL-C reduction from baseline at any visit (responders) | Exploratory |
Response of LDL-C reduction by underlying causal mutations of HeFHa | Exploratory |
ApoB = apolipoprotein B; ASCVD = atherosclerotic cardiovascular disease; HDL-C = high-density lipoprotein cholesterol; HeFH = heterozygous familial hypercholesterolemia; LDL-C = low-density lipoprotein cholesterol; MI = myocardial infarction; PCSK9 = proprotein convertase subtilisin/kexin type 9; TC = total cholesterol.
aORION-9 only.
Source: Clinical Study Reports for ORION-9,17 ORION-10,18 and ORION-11.19
Patients were in a fasted state for all efficacy laboratory assessments. The parameters assessed were total cholesterol, triglycerides, LDL-C, HDL-C, non-HDL-C, very low–density lipoprotein cholesterol, apolipoprotein A1, ApoB, lipoprotein (a), high-sensitivity C-reactive protein, and PCSK9. All laboratory assessments and assays were performed by a central laboratory, with the exception of urinalysis.17-19
The primary outcome of the ORION-9, −10, and −11 trials was the percentage change in LDL-C from baseline to day 510. All trials had a co-primary end point of time-adjusted percentage change in LDL-C from baseline after day 90 and up to day 540, defined as the average percentage change in LDL-C from baseline over the period after day 90 and up to day 540, reflecting the start of the twice-yearly dosing regimen.17-19
Key secondary efficacy end points of the ORION trials included:
absolute change in LDL-C from baseline to day 510
time-adjusted absolute change in LDL-C from baseline after day 90 and up to day 540
percentage change from baseline to day 510 in PCSK9, total cholesterol, ApoB, and non-HDL-C
Key secondary end points were not tested if either 1 of the co-primary efficacy end points’ null hypotheses failed to be rejected.17-19
Other secondary outcomes were consistent across the ORION trials and included all of the following:
maximum percentage change in LDL-C
absolute and percentage change in LDL-C from baseline to each assessment time, up to day 540
individual responsiveness (defined as the number of patients reaching on-treatment LDL-C levels of less than 25 mg/dL, less than 50 mg/dL, less than 70 mg/dL, and less than 100 mg/dL at day 510)
the proportion of patients in each group with a reduction in LDL-C from baseline of 50% or greater
absolute change and percentage change in other lipids, lipoproteins, and apolipoproteins
PCSK9 change from baseline at each subsequent visit to day 540
the proportion of patients in each group who attained global lipid targets for their level of ASCVD risk
Exploratory end points included incidence of CV death, resuscitated cardiac arrest, non-fatal MI, and non-fatal stroke (ischemic and hemorrhagic), and proportion of patients in each group with any LDL-C reduction from baseline at any visit (responders).17-19 In ORION-9, an additional exploratory outcome consisted of response of LDL-C reduction by underlying causal mutations of HeFH.17
Blood samples were taken at scheduled time points to determine LDL-C concentrations. Additional aliquots of plasma and serum were collected at each time point and stored for additional analyses, including future analysis of biomarkers of CV risk. Plasma samples were analyzed using a validated enzyme-linked immunosorbent assay to determine PCSK9 protein concentration.17-19
Safety and tolerability of inclisiran was a secondary end point of the study and was measured by AEs, SAEs, vital signs, clinical laboratory values, electrocardiogram measurements, and formation and characterization of anti-drug antibodies. Adverse events, defined as any untoward medical occurrence in a patient administered a medicinal product where the AE does not necessarily have a causal relationship with the treatment, were coded using the Medical Dictionary for Regulatory Activities (MedDRA) v20.1. The severity of AEs was assessed by the investigator on a 3-point scale of mild, moderate, or severe.17-19
No patient-reported HRQoL outcomes were assessed in the ORION trials.17-19
Statistical Analysis
Sample Size and Power Calculation
The sample size calculations for the ORION-9, −10, and −11 trials were identical and were based on the observed results of the phase II study. Calculations were performed under the assumption that the difference in change from baseline between the active-dose group and the placebo group for LDL-C would be no less than 30 mg/dL, with a standard deviation of 20 mg/dL.17-19
Assuming a 5% dropout rate, the sample size was approximately 380 patients in ORION-9, and 1,425 patients in both ORION-10 and −11 who were evaluable for efficacy across the placebo and inclisiran dose groups. This sample size of at least 380, 1,425, and 1,425 evaluable patients, respectively, provided more than 90% power to detect a 30% reduction of LDL-C levels in the inclisiran group compared with the placebo group at a 1-sided significance level of 0.025.17-19 Due to faster-than-expected enrolment in the ORION-9 trial, the actual enrolment was 482 patients.17 ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.44
Interim and Final Analyses
No interim analyses of efficacy were performed for ORION-9, ORION-10, or ORION-11. An independent data monitoring committee (IDMC) reviewed safety data after the first 40 patients received the first subcutaneous injection of inclisiran or placebo and completed 1 month of follow-up. In all ORION trials, an IDMC reviewed safety data 90 days after the first 40 patients received the first injection of inclisiran or placebo. Thereafter, the IDMC reviewed safety data approximately every 3 months until the end of study, unless requested otherwise by the IDMC.17-19
Statistical and Analytical Plans
In all cases, the statistical analysis plan was finalized before database lock; however, changes were requested by the FDA. For ORION-10 and −11, an addendum and changes to the statistical analysis plan were requested by the FDA as well as other changes in analyses after the statistical analysis plan was finalized, the database locked, and the study unblinded.17-19
All study-collected data were summarized by treatment group using descriptive statistics, graphs, and/or raw data listings. Categorical variables were summarized using counts and percentages. Percentages were based on the number of patients in the analysis set for whom there were non-missing data, unless otherwise specified. Continuous variables, including changes from day 1, were summarized using descriptive statistics. All P values were 2-sided and rounded to 3 decimal places. All P values rounded to 0.000 were presented as “< 0.001” and P values that round to 1.000 were presented as “1.000.”17-19
Absolute change and percentage change from baseline were calculated as follows17-19:
Absolute change = (value at day χ) minus baseline value
Percentage change = (absolute change ÷ baseline value) × 100%
Primary Efficacy End Point Analyses
The intention-to-treat (ITT) population was the primary population for the efficacy analysis in all ORION trials. The statistical hypothesis for the co-primary end points was that the difference between patients treated with inclisiran and placebo in the least squares mean (LSM) percentage change and mean time-adjusted change in LDL-C from baseline at day 510 was 0. The co-primary end points were controlled for multiplicity using the family-wise type I error rate which was controlled at a 1-sided alpha significance level of 0.05 by using a nested testing procedure. A sequential testing procedure was used to assess the percentage change in LDL-C from baseline to day 510. If the null hypothesis was rejected at a 2-sided alpha significance level of 0.05 and superiority of inclisiran over placebo was claimed, then the time-adjusted percentage change in LDL-C from baseline after day 90 and up to day 540 was tested, also at a 2-sided alpha significance level of 0.05.17-19
The primary end points used a reflexive LDL-C measurement approach. LDL-C was calculated based on the Friedewald formula. If the calculated LDL-C was less than 40 mg/dL (1.0 mmol/L), or triglycerides were greater than 400 mg/dL (4.5 mmol/L), or calculated LDL-C was missing, directly measured (using ultracentrifugation) LDL-C was used.17-19
A multiple imputation washout model was used to impute missing data for the percentage change in the LDL cholesterol level from baseline to day 510. The washout model can be thought of as a modified control-based pattern-mixture model (PMM) that was used to explore the possibility of data missing not at random for patients who discontinued the study early. Percentage change for each visit was calculated after the missing data imputation was performed.17-19 In ORION-11, the multiple imputation washout model was modified such that patients in the inclisiran group who received all 4 doses of the study drug who had the day 510 value missing and had evaluable data at day 540, had their intermittent missing day 510 values imputed assuming the missing data were missing at random (MAR).19
The primary analysis was based on an analysis of covariance (ANCOVA) model on each multiply imputed dataset (100 total). The model included the fixed effect of treatment and baseline LDL-C as a covariate. Treatment effects from these analyses were then combined using the Rubin method.45 The difference in the LSM between treatment groups and the corresponding 2-sided 95% CI were provided for hypothesis testing.17-19
For the co-primary end point of time-adjusted percentage change in the LDL cholesterol level between day 90 and day 540, a control-based PMM was used to impute any missing values. A mixed-effects model for repeated measures (MMRM) was used for data obtained during all visits on each multiply imputed dataset. The model included fixed effects for treatment, visit, baseline LDL-C, and the interaction between treatment and visit. Linear combinations of the estimated means after day 90 and up to day 540 were used to compare treatment effects. Note that day 90 was excluded from the MMRM.17-19
The PMM was used to explore the possibility of data missing not at random for patients who discontinued the study. For patients who discontinued the study without any further follow-up data, missing values after study discontinuation were imputed under the assumption that their outcome would be similar to those in the placebo group. For patients who did not discontinue the study, missing values were imputed based on the MAR assumption. Multiple imputations were used to account for uncertainty in the imputation process and results from the imputed datasets were combined using the Rubin method as with the primary end point.17-19
Sensitivity Analyses
Sensitivity analyses of the co-primary end points was conducted to support the robustness of the conclusions. Three sensitivity analyses were conducted for the percentage change in LDL-C from baseline to day 510, as follows17-19:
A control-based PMM using the same imputed datasets and MMRM that was used for the second co-primary efficacy end point.
An MMRM was used that included fixed effects for treatment, visit, baseline value, and interaction between treatment and visit. The ORION-11 study included the current use of statins or other LMT as fixed effects, as well.
An ANCOVA model was used to assess the impact of country (ORION-9 and ORION-11) as fixed effects and use of statins or other LMT (ORION-9). The model used the modified ITT population, including the fixed effect of treatment group and baseline LDL-C, as a covariate (ORION-9 and ORION-10).
For time-adjusted percentage change in LDL-C from baseline to day 90 and up to day 540, sensitivity analyses included17-19:
An MMRM was used that assumed data are MAR and included fixed effects for treatment, visit, baseline value, interaction between treatment and visit in ORION-9, and −10. The current use of statins or other LMTs was also included as a fixed effect in ORION-11.
A second MMRM assessed the impact of country (ORION-9 and ORION-11) and current use of statins (ORION-9 and ORION-10) or other LMT as fixed effects.
A 2-sample t-test was performed to test the treatment difference between inclisiran and placebo for time-adjusted percentage change, which was calculated by taking the arithmetic mean of the percentage change in LDL-C from baseline at each visit after day 90 through day 540.
Secondary Efficacy End Point Analyses
Analyses of the secondary end points were performed only after the analyses of the 2 primary end points were completed and the null hypotheses were rejected. The key secondary end points of this study were17-19:
absolute change in LDL-C from baseline to day 510
time-adjusted absolute change in LDL-C from baseline after day 90 and up to day 540
percentage change from baseline to day 510 in PCSK9, total cholesterol, ApoB, and non-HDL-C.
Key secondary efficacy end points were not tested if either 1 of the co-primary efficacy end points’ null hypotheses failed to be rejected. Key secondary end points were controlled for multiplicity using the Hochberg procedure,46 which was applied to control the family-wise type I error rate at 2-sided alpha significance level of 0.05. Missing values were imputed using the control-based PMM on LDL-C, PCSK9, total cholesterol, ApoB, and non-HDL-C; absolute change or percentage change from baseline was calculated based on imputed data before any analysis was performed.17-19
The absolute change in LDL-C from baseline to day 510 and percentage change from baseline to day 510 in PCSK9, total cholesterol, ApoB, and non-HDL-C were analyzed using an MMRM with covariates. The time-adjusted absolute change in LDL-C from baseline after day 90 and up to day 540 was analyzed similarly to the analysis of the time-adjusted percentage change in LDL-C from baseline after day 90 and up to day 540. In ORION-11, as 94.7% of patients were currently using statins or other LMT at baseline, a revised model that excluded statins and other LMTs as a covariate was performed. This change did not affect the placebo-adjusted change from baseline in any of the analyses; therefore, key secondary end points using the PMM excluded statins and other LMTs as a covariate.19 An MMRM without multiple imputation was used as a sensitivity analysis for the key secondary end points.17-19
The 2-sided 95% CI for LSMs was provided for continuous variables at a single point using an ANCOVA model or using MMRM methods for variables measured over time. The odds ratio and 95% CI were provided for binary variables using logistic regression models. Nominal P values were provided when applicable. Descriptive and graphical summaries by treatment group were also presented.17-19
Exploratory Efficacy End Point Analyses
Major CV events were defined as the composite of CV death, resuscitated cardiac arrest, non-fatal MI, and stroke (ischemic or hemorrhagic). The number and percentage of patients with an MACE or any of the individual events were presented by treatment group. Events were identified using predefined MedDRA terms, including fatal SAEs in cardiac disorders and fatal SAEs in general-disorder system organ classes, including preferred terms for death, sudden cardiac death, cardiac death, and apparent death.17-19
For the proportion of patients in each group with any LDL-C reduction from baseline at any visit (responders), the number and percentage of patients with any LDL-C reduction at any visit were presented by treatment group. The odds ratio and 95% CI for the odds ratio were provided using a logistic regression model. The number and percentage of patients with any LDL-C reduction was also summarized at each visit.17-19
In ORION-9, the absolute change and percentage change in LDL-C and PCSK9 from baseline to day 510 were summarized descriptively by HeFH mutation type and treatment group.17 The results of that analysis were not included in this report.
Safety Analyses
Safety analyses were based on observed values. Any missing safety data, including laboratory data, were not imputed. For common TEAEs, serious TEAEs, and TEAEs leading to the withdrawal of the study drug, risk ratios along with 95% CIs were presented to compare treatment groups with respect to risk.17-19
Subgroup Analyses
Analyses of efficacy and safety were performed for the following pre-specified subgroups of interest in all 3 ORION trials using MMRM to account for missing data: gender (male, female), age (< 65 versus ≥ 65 years, and < 75 versus ≥ 75 years,), BMI (≤ the median versus > the median, and quartiles), race (White, Black, other), baseline statin use (yes, no), baseline statin intensity (high intensity, not high intensity), other LMT (any statin with other LMT, other LMT but no statin, no LMT), baseline triglyceride level (< 200 mg/dL versus ≥ 200 mg/dL, and ≤ the median versus > the median), metabolic disease (diabetes, metabolic syndrome without diabetes, neither), renal impairment by eGFR categories (eGFR in mL/min/1.73 m2 = ≥ 15 to < 30, ≥ 30 to < 60, ≥ 60 to < 90, and ≥ 90), history of allergy (yes, no), baseline LDL-C (≤ the median versus > the median, and quartiles), and post-baseline LDL-C (≤ 25 at any time point versus > 25 mg/dL at all time points).17-19
Additional subgroups of phenotype (FH, nFH), and baseline ezetimibe use (yes, no) were assessed in ORION-9.17 ASCVD status (ASCVD, ASCVD risk equivalent), study centre (North America, Europe, South Africa) were evaluated in ORION-9 and ORION-11, but not ORION-10.17,19 The ORION-10 study also included a subgroup of ethnicity (Hispanic, non-Hispanic).18
The subgroups identified in the review protocol included prior CV event (MI, stroke, unstable angina, hospitalization), concomitant treatments (statin, ezetimibe, other LMT), baseline LDL-C level, and patients who are not candidates for or are intolerant to statins.
Analysis Populations
The following analysis populations were defined in ORION-9, ORION-10, and ORION-1117-19:
ITT population: This included all patients who were randomized into the study. The ITT population was used for the analysis of the primary and secondary end points.
Full analysis set: This consisted of all patients who were randomized into the study, took any study drug, and had at least 1 post-treatment lipid data measured.
Modified ITT: This consisted of all randomized patients who received at least 1 dose of the study drug and had both the baseline and the day 510 follow-up LDL-C assessment.
Safety population: This comprised all patients who received at least 1 dose of the study drug. This was the primary population for the safety analyses.
The ORION-10 trial also included a pharmacokinetic analysis population18 that is not described in this report.
Protocol Amendments and Deviations
A total of 4 global protocol amendments occurred in ORION-9. Any potentially relevant changes were made before the enrolment of patients; therefore, there was no impact on the conduct of the study or on the results of the statistical analyses. A total of 4 global protocol amendments also occurred in ORION-10, and 2 global protocol amendments |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| for ORION-11.17-19 The amendments had no major impact on the studies, as the enrolment and statistical analyses were not completed before the amendments, and changes made to the statistical analysis plan did not impact the study conduct.
In ORION-9, 1 protocol deviation occurred in 11.4% of patients. The most common protocol deviations were prohibited concomitant medication or change in baseline statin or dose of other lipid-lowering therapy (3.7% versus 5.8% for the inclisiran and placebo groups, respectively) and inclusion and/or exclusion criteria violation (3.3% each in the inclisiran and placebo groups).17 In ORION-10, 1 protocol deviation occurred in 22.1% of patients. The most common protocol deviations were prohibited concomitant medication or change in baseline statin or dose of other lipid-lowering therapy (11.9% versus 12.2%) and mis-dosing for any reason other than patient safety or withdrawal (3.0% versus 5.4%). Inclusion and exclusion criteria violations were low and similar in patients taking placebo (2.6%) and patients taking inclisiran (4.4%).18 In ORION-11, 1 protocol deviation occurred in 15.2% of patients. The most common protocol deviations were prohibited concomitant medication or change in baseline statin or dose of other lipid-lowering therapy (8.0% versus 10.0%) and mis-dosing for any reason other than patient safety or withdrawal (3.3% versus 2.7%). Inclusion and exclusion criteria violations were low and similar in patients taking placebo (1.9%) and patients taking inclisiran (1.4%).19
Results
Patient Disposition
The ORION-9, −10, and −11 trials shared a similar study design and patient enrolment. Table 9 summarizes the patient flow of the ORION trials. In ORION-9, a total of 617 patients were screened and 482 were randomized. The most common reason for screening failure was “inclusion/exclusion criteria not met” ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. A total of 2,329 patients were randomized in ORION-10, of which 1,561 were randomized. In ORION-11, a total of 2,381 patients were screened and 1,617 were randomized. The primary reason for screening failure in ORION-10 and −11 was patient not meeting inclusion or exclusion criteria, ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. The proportion of patients who failed screening varied across trials but was more similar in the ORION-10 and −11 trials (32.98% and 32.09%) compared with ORION-9 (21.88%).17-19
Across all studies, 89% of patients or more completed the study (i.e., had a day 540 end-of-study visit). The discontinuation rate was similar across studies, with ORION-10 having the greatest proportion of patients discontinuing treatment. In all studies, the primary reason for discontinuation was withdrawal of consent. The proportion of patients discontinuing due to death was similar in the ORION-10 and −11 trials but was lower in the ORION-9 trial.17-19
Detail | ORION-9 | ORION-10 | ORION-11 | |||
|---|---|---|---|---|---|---|
Inclisiran | Placebo | Inclisiran | Placebo | Inclisiran | Placebo | |
Screened, N | 617 | 2,329 | 2,381 | |||
Screen failures | 135 | 768 | 764 | |||
Randomized, N (%) | 242 | 240 | 781 | 780 | 810 | 807 |
Completed study,a N (%) | 235 (97.1) | 231 (96.3) | 721 (92.3) | 694 (89.0) | 772 (95.3) | 770 (95.4) |
Reason for discontinuation, n (%) | ||||||
Withdrew consent | 0 (0) | 4 (1.7) | 24 (3.1) | 34 (4.4) | 13 (1.6) | 17 (2.1) |
Lost to follow-up | 1 (0.4) | 2 (0.8) | 10 (1.3) | 24 (3.1) | 6 (0.7) | 3 (0.4) |
Death | 1 (0.4) | 1 (0.4) | 12 (1.5) | 11 (1.4) | 14 (1.7) | 15 (1.9) |
Adverse event | 0 (0) | 0 (0) | 8 (1.0) | 5 (0.6) | 4 (0.5) | 0 (0) |
Initiation of protocol-prohibited PCSK9 inhibitor | 0 (0) | 1 (0.4) | 0 (0) | 9 (1.2) | 0 (0) | 0 (0) |
Physician decision | 0 (0) | 0 (0) | 1 (0.1) | 0 (0) | 1 (0.1) | 1 (0.1) |
Other | 5 (2.1) | 1 (0.4) | 5 (0.6) | 3 (0.4) | 0 (0) | 1 (0.1) |
ITT, n | 242 | 240 | 781 | 780 | 810 | 807 |
FAS, n | 241 | 239 | 767 | 768 | 803 | 800 |
Safety, n | 241 | 240 | 781 | 778 | 811 | 804 |
FAS = full analysis set; ITT = intention to treat; PSCK9 = proprotein convertase subtilisin/kexin type 9.
aRefers to patients who completed the day 540 visit.
Source: Clinical Study Reports for ORION-9,17 ORION-10,18 and ORION-11.19
Exposure to Study Treatments
All study drugs were administered by staff at the clinical study site. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.17-19
Table 10: Study Exposure (Safety Population)
Category | ORION-9 | ORION-10 | ORION-11 | |||
|---|---|---|---|---|---|---|
Inclisiran (N = 241) | Placebo (N = 240) | Inclisiran (N = 781) | Placebo (N = 778) | Inclisiran (N = 811) | Placebo (N = 804) | |
Number of study doses administered, n (%) | ||||||
|||| ||| | ||||| | ||| | ||||| | ||||| | ||||| | ||||| |
|||| ||| | ||||| | ||| | ||||| | |||||| | ||||| | ||||| |
|||| ||| | ||||| | ||| | ||||| | ||||| | ||||| | ||||| |
|||| ||| | |||||||| | |||||||| | |||||| | ||||||| | |||||| | |||||| |
SD = standard deviation.
aPatients could be counted in multiple dosing visits.
This table has been redacted at the request of the sponsor. The rest of the rows have therefore been deleted.
Source: Clinical Study Reports for ORION-9,17 ORION-10,18 and ORION-11.19
Efficacy
Only those efficacy outcomes and analyses of subgroups identified in the review protocol are reported subsequently. See Appendix 3 for detailed sensitivity and subgroup efficacy data.
Mortality (All-Cause and CV-Related)
All-cause and CV-related mortality were measured as AEs in the ORION-9, −10, and −11 trials. The list of deaths as AEs in the ORION trials is summarized in Table 11.
In ORION-9, only 2 deaths occurred (0.4%), 1 in each trial arm.17 A total of 23 patients died during the ORION-10 study, 12 (1.5%) in the inclisiran group, and 11 (1.4%) in the placebo group.18 In total, 29 patients (1.8%) died during the ORION-11 study, 14 (1.7%) in the inclisiran group, and 15 (1.9%) in the placebo group.19 |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. No deaths were attributed to the study drug in any of the studies.17-19
Table 11: Deaths (Safety Population)
Death outcomes | ORION-9 | ORION-10 | ORION-11 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
Inclisiran (N = 241) | Placebo (N = 240) | Inclisiran (N = 781) | Placebo (N = 778) | Inclisiran (N = 811) | Placebo (N = 804) | |||||||
n (%) | E | n (%) | E | n (%) | E | n (%) | E | n (%) | E | n (%) | E | |
TEAEs with fatal outcome, n (%) | 1 (0.4) | ||||| | 1 (0.4) | ||||| | 12 (1.5) | |||| | 11 (1.4) | |||| | 14 (1.7) | ||||| | 15 (1.9) | ||||| |
|||||||||||||||||||| | |||| | ||||| | |||| | ||||| | |||| | |||| | |||| | ||||| | |||||| | ||||| | |||| | ||||| |
|||||||||||||| | |||| | ||||| | ||| | ||||| | ||||| | |||| | ||||| | ||||| | ||||| | ||||| | ||||| | ||||| |
|||||||| | ||||| | ||||| | ||||| | ||||| | ||||| | |||| | ||||| | ||||| | ||| | ||||| | |||| | ||||| |
CAD = coronary artery disease; E = event count; MI = myocardial infarction; NR = not reported; SOC = system organ class; TEAE = treatment-emergent adverse event.
The rest of the data in this table has been redacted at the request of the sponsor, so the redacted rows have been deleted.
Source: Clinical Study Reports for ORION-9,17 ORION-10,18 and ORION-11.19
CV-Related Morbidity
Incidence of Resuscitated Cardiac Arrest, Non-Fatal MI, and Stroke
CV-related morbidity outcomes, including the incidence of resuscitated cardiac arrest, non-fatal MI, and stroke, were included as a compound end point of MACE and was an exploratory outcome in the ORION-9, −10, and −11 trials. Incidences of events are summarized in Table 12. Non-fatal MI was the most frequently occurring individual event across all trials, occurring in 3.7% versus 4.2%, 5.1% versus 8.2%, and 5.8% versus 8.5% of patients in the inclisiran and placebo groups of ORION-9, −10, and −11, respectively; however, there were no apparent differences between groups. No resuscitated cardiac arrest or stroke events occurred in the ORION-9 trial,17 and results were similar between groups in ORION-10 and ORION-11.18,19 The overall incidence of MACEs in the inclisiran groups was consistently similar to or lower than in placebo groups across all trials (4.1% versus 4.2%, 7.4% versus 10.2%, and 7.8% versus 10.3%, in ORION-9, −10, −11, respectively).17-19
Table 12: Incidence of MACEs (CV Death, MI, Stroke, Resuscitated Cardiac Arrest, Non-Fatal MI), and Non-Fatal Stroke (Ischemic and Hemorrhagic) — Safety Population
Category | ORION-9 | ORION-10 | ORION-11 | |||
|---|---|---|---|---|---|---|
Inclisiran (N = 241) | Placebo (N = 240) | Inclisiran (N = 781) | Placebo (N = 778) | Inclisiran (N = 811) | Placebo (N = 804) | |
MACEa | 10 (4.1) | 10 (4.2) | 58 (7.4) | 79 (10.2) | 63 (7.8) | 83 (10.3) |
CV death | 1 (0.4) | 0 (0) | 7 (0.9) | 5 (0.6) | 9 (1.1) | 9 (1.1) |
Resuscitated cardiac arrest | 0 (0) | 0 (0) | 1 (0.1) | 1 (0.1) | 3 (0.4) | 0 (0) |
Non-fatal MI | 9 (3.7) | 10 (4.2) | 40 (5.1) | 64 (8.2) | 47 (5.8) | 68 (8.5) |
Stroke (ischemic or hemorrhagic) | 0 (0) | 0 (0) | 12 (1.5) | 10 (1.3) | 4 (0.5) | 8 (1.0) |
CV = cardiovascular; MACE = major cardiovascular event; MI = myocardial infarction.
Note: Strokes are only those that are non-fatal.
aMACE is defined as the composite of CV death, resuscitated cardiac arrest, non-fatal MI, and stroke (ischemic or hemorrhagic).
Source: Clinical Study Reports for ORION-9,17 ORION-10,18 and ORION-11.19
Hospitalizations and minimally invasive CV interventions were not reported in ORION-9, ORION-10, or ORION-11.
Percentage and Absolute Change in LDL-C
Percentage Change in LDL-C From Baseline to Day 510
The primary efficacy end point of the ORION-9, −10, and −11 trials was the percentage change in LDL-C from baseline to day 510, which is summarized in Table 13. In all ORION trials, inclisiran reduced LDL-C levels from baseline to day 510: −41.15% in ORION-9 (95% CI, −44.52 to −37.77), −56.34% in ORION-10 (95% CI, −58.35 to −54.34), and −49.3% in ORION-11 (||||||||||||||||||||||||||||||), while the change from baseline LDL-C levels increased with placebo: 8.37% in ORION-9 (95% CI, 3.96 to 12.77), 1.30% in ORION-10 (95% CI, −1.24 to 3.83), and 4.2% in ORION-11 (||||||||||||||||||||||).17-19 The washout-imputed values, which took missing data into consideration, were similar, albeit the LSM percentage change from baseline was slightly smaller than the observed values.
In all trials, the LSM difference from placebo in percentage change in LDL-C from baseline favoured inclisiran for both the observed and washout-imputed values. The observed values were −49.52 in ORION-9 (95% CI, −55.04 to −43.99), −57.64 in ORION-10 (95% CI, −60.86 to −54.43), and −53.5 in ORION-11 (95% CI, −56.66 to −50.35). The washout-imputed values were −47.89 in ORION-9 (95% CI, −53.52 to −42.26), −52.24 in ORION-10 (95% CI, −55.65 to −48.83), and −49.9 in ORION-11 (95% CI, −53.07 to −46.64). All P values were less than 0.0001,17-19 with observed values, not accounting for missing data, showing a larger percentage reduction in LDL-C from baseline (Table 13).
Sensitivity Analyses
Results of the 3 additional sensitivity analyses (PMM, MMRM, and an ANCOVA with country and current use of statins or other LMT as fixed effects) for percentage change in LDL-C from baseline to day 510 yielded results similar to the primary end point, with inclisiran demonstrating a reduction in LDL-C and placebo showing an increase in LDL-C levels. The between-group difference was consistent with the base-case scenario and the results were statistically significant (P < 0.0001).17-19 Detailed outcome data for the sensitivity analyses are summarized in Table 29, Table 30, and Table 32 in Appendix 3.
Subgroup Analysis
Subgroup analyses were provided for the co-primary outcome of the percentage change in LDL-C from baseline to day 510 for the ORION-9, −10, and −11 trials. Results of the subgroup analyses for subgroups of interest, including baseline LDL-C, baseline statin treatment (on treatment versus not on treatment), statin intensity (high versus not high), LMT use (any statin or other LMT versus no statin or LMT), were consistent with the overall patient population and there was no evidence for effect modification within subgroups, with the percentage change from baseline ranging from −42.7% to −55.7% in ORION-9, −69.8% to −45.9% in ORION-10, and −60.8% to −38.2% in ORION-11.17-19 In the ORION-9 and −11 trials, subgroups based on risk category (patients with ASCVD or an ASCVD risk equivalent) were reported. In both trials, the subgroup results were consistent with the overall population, with an LSM percentage difference in LDL-C of −53.6% (95% CI, −64.8 to −42.3) in ORION-9, and −53.3% (95% CI, −56.6 to −50.1) in ORION-11 for patients with ASCVD, and −47.4% (95% CI, −53.7 to 41.1) and −47.2% (95% CI, −56.1 to −38.3) for the ASCVD risk equivalent subgroup in ORION-9 and −11, respectively.17,19 The population in ORION-11 with an ASCVD risk equivalent was considered not of interest, as the majority of the population was not included in the funding request.
Comprehensive forest plots summarizing the results of all subgroups evaluated in the ORION-9, ORION-10, and ORION-11 trials for percentage change in LDL-C from baseline at day 510 are provided in Figure 8, Figure 10, and Figure 12 in Appendix 3.
Time-Adjusted Percentage Change in LDL-C From Baseline After Day 90 and Up to Day 540
The time-adjusted percentage change in LDL-C from baseline after day 90 and up to day 540 was the co-primary end point of the ORION-9, −10, and −11 trials. Results are summarized in Table 13. Inclisiran showed greater reductions in LDL-C compared with placebo in all 3 trials: −38.08% (95% CI, −41.03 to −35.14) versus 6.22% (95% CI, 3.26 to 9.17), in ORION-9, −51.27% (95% CI, −53.00 to −49.54) versus 2.51% (95% CI, 0.77 to 4.25) in ORION-10, and −45.82% (95% CI, −47.52 to −44.13) versus 3.35% (95% CI, 1.65 to 5.05) in ORION-11.17-19
The LSM difference from placebo also favoured inclisiran in all trials: −44.30% (95% CI, −48.48 to −40.12) in ORION-9, −53.78% (95% CI, −56.23 to −51.33) in ORION-10, and −49.17% (95% CI, −51.57 to −46.77) in ORION-11 (P < 0.0001 for all).17-19
Sensitivity Analyses
The results of the sensitivity analyses for time-adjusted percentage change in LDL-C from baseline after day 90 and up to day 540 were consistent with the overall population.17-19 Detailed outcome data for the sensitivity analyses are summarized in Table 29, Table 31, and Table 32 in Appendix 3.
Subgroup Analysis
Subgroup analyses were provided for the co-primary outcome of the time-adjusted percentage change in LDL-C after day 90 up to day 540 for the ORION-9, −10, and −11 trials. Results of the subgroup analyses of interest to this review, including baseline LDL-C, baseline statin treatment, statin intensity, and LMT use, were consistent with the overall patient population, ranging from −59.7% to −33.0% in ORION-9, −67.0% to −44.9% in ORION-10, and –58.7% to −35.4% in ORION-11.17-19
In the ORION-9 and −11 trials, subgroups based on risk category (patients with ASCVD or an ASCVD risk equivalent) were reported. In both trials, subgroup results were consistent with the overall population, with an LSM percentage difference in LDL-C of −47.9% (95% CI, −56.2 to −39.6) and −50.9% (95% CI, −53.4 to −48.8) for the ASCVD populations in ORION-9 and −11, respectively, and −43.7% (95% CI, −48.5 to −38.9) and −42.3% (95% CI, −49.2 to −35.3) for the ASCVD risk–equivalent populations in ORION-9 and −11, respectively.17,19 The ASCVD risk–equivalent population in ORION-11 was considered not of interest, as the majority of the population was not included in the funding request.
A comprehensive forest plot summarizing the results of all subgroups evaluated in the ORION-9, ORION-10, and ORION-11 trials for time-adjusted percentage change in LDL-C from baseline after day 90 and up to day 540 is provided in Figure 9, Figure 11, and Figure 13 in Appendix 3.
Absolute Change in LDL-C From Baseline to Day 510
Absolute change in LDL-C from baseline to day 510 was a key secondary outcome of the ORION trials, and the results are summarized in Table 13. The absolute reduction in LDL-C in the inclisiran groups was −58.95 mg/dL (95% CI, −64.75 to −53.15) in ORION-9, −56.18 mg/dL (95% CI, −58.47 to −53.90) in ORION-10, and −50.91 mg/dL (95% CI, −53.14 to −48.67) in ORION-11 while, in the placebo groups, the reduction was 9.94 mg/dL (95% CI, 4.10 to 15.78) in ORION-9, −2.06 mg/dL (95% CI, −4.36 to 0.24) in ORION-10, and 0.96 mg/dL (95% CI, −1.26 to 3.18) in ORION-11. The LSM difference from placebo favoured inclisiran in all trials; −68.89 mg/dL (95% CI, −77.11 to −60.67) in ORION-9, −54.12 mg/dL (95% CI, −57.37 to −50.88) in ORION-10, and −51.87 mg/dL (95% CI, −55.01 to −48.72) in ORION-11 (P < 0.0001 for all). ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.17-19
Time-Adjusted Absolute Change in LDL-C From Baseline After Day 90 and Up to Day 540
The time-adjusted absolute change from baseline in LDL-C after day 90 up to day 540 was a secondary efficacy outcome of the ORION-9, −10, and −11 trials, and is summarized in Table 13. In the time-adjusted analyses for all trials, inclisiran demonstrated greater reductions in LDL-C compared with placebo: −56.58 mg/dL (95% CI, −60.98 to −52.17) versus 6.17 mg/dL (95% CI, 1.72 to 10.62) in ORION-9, −53.66 mg/dL (95% CI, −55.41 to −51.92) versus −0.39 mg/dL (95% CI, −2.14 to 1.37) in ORION-10, and −48.63 mg/dL (95% CI, −50.37 to −46.89) versus 0.31 mg/dL (95% CI, −1.42 to 2.04) in ORION-11. Compared with placebo, the time-adjusted absolute change from baseline after day 90 and up to day 540 ranged from −62.74 mg/dL (95% CI, −69.01 to −6.48) to −48.94 mg/dL (95% CI, −51.39 to −46.48) (P < 0.0001 for all). ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.17-19
Global Lipid LDL-C Targets
At day 510, 158 (65.3%), 651 (83.4%), and 661 (81.6%) of inclisiran-treated patients in the ORION-9, −10, and −11 trials reached an LDL-C level of less than 100 mg/dL compared with 21 (8.8%), 387 (49.6%), and 425 (52.7%) placebo-treated patients, respectively. Similarly, 99 (40.9%), 581 (74.4%), and 564 (69.6%) of inclisiran-treated patients in the ORION-9, −10, and −11 trials reached LDL-C level of less than 70 mg/dL compared with 3 (1.3%), 119 (15.3%), and 104 (12.9%) placebo-treated patients, respectively. Note that some patients were counted under multiple categories.17-19
In the ORION-9 study, a total of 159 (66.0%) inclisiran-treated patients achieved a 50% or greater reduction in LDL-C from baseline at any visit over the course of the study compared with 9 (3.8%) placebo-treated patients.17 In ORION-10, and ORION-11, 701 (91.4%) and 658 (81.9%) inclisiran-treated patients reached a 50% or greater reduction in LDL-C from baseline compared with 50 (6.5%) and 47 (5.9%) placebo-treated patients.18,19
In ORION-9, at any visit, 186 (77.2%) inclisiran-treated patients achieved their corresponding LDL-C targets (LDL-C < 100 mg/dL or < 70 mg/dL) compared with 44 (18.4%) placebo-treated patients. In ORION-10, 722 (94.1%) inclisiran-treated patients achieved their LDL-C targets at any visit compared with 277 (36.1%) placebo-treated patients. In ORION-11, 741 (92.4%) inclisiran-treated patients achieved their corresponding LDL-C targets at any visit compared with 335 (41.9%) placebo-treated patients. In patients with ASCVD, at day 510, 31 (52.5%), 581 (84.1%), and 522 (81.7%) inclisiran-treated patients achieved their LDL-C target (LDL-C < 70 mg/dL) in ORION-9, −10, and −11, respectively, compared with 1 (1.4%), 119 (17.9%), and 103 (16.0%) placebo-treated patients, respectively.17-19 In patients with HeFH (ORION-9), 115 (66.9%) inclisiran-treated patients and 14 (8.9%) placebo-treated patients achieved their LDL-C target (LDL-C < 100 mg/dL) at day 510.17,19
Table 13: Percentage, Absolute, and Time-Adjusted Change in LDL-C — ITT Population
Detail | ORION-9 | ORION-10 | ORION-11 | ||||||
|---|---|---|---|---|---|---|---|---|---|
Inclisiran (N = 242) | Placebo (N = 240) | Difference from placebo | Inclisiran (N = 781) | Placebo (N = 780) | Difference from placebo | Inclisiran (N = 810) | Placebo (N = 807) | Difference from placebo | |
Percentage change in LDL-C (LSM, 95% CI) | |||||||||
Percentage CFB in LDL-C to day 510 (observed values) | −41.15 (−44.52 to −37.77) | 8.37 (3.96 to 12.77) | −49.52 (−55.04 to −43.99) P < 0.0001 | −56.34 (−58.35 to −54.34) | 1.30 (−1.24 to 3.83) | −57.64 (−60.86 to −54.43) P < 0.0001 | −49.3 |||||||||||||||||| | 4.2 |||||||||||| | −53.5 (−56.66 to −50.35) P < 0.0001 |
Percentage CFB in LDL-C to day 510 (washout-imputed values)a,b | −39.67 (−43.72 to −35.62) | 8.22 (4.27 to 12.16) | −47.89 (−53.52 to −42.26) P < 0.0001 | −51.28 (−53.76 to −48.81) | 0.96 (−1.48 to 3.40) | −52.24 (−55.65 to −48.83) P < 0.0001 | −45.8 (−48.16 to −43.48) | 4.0 (1.76 to 6.31) | −49.9 (−53.07 to −46.64) P < 0.0001 |
Absolute change in LDL-C (mg/dL; LSM, 95% CI) | |||||||||
Absolute CFB in LDL-C to day 510 (PMM)a | −58.95 (−64.75 to −53.15) | 9.94 (4.10 to 15.78) | −68.89 (−77.11 to −60.67) P < 0.0001 | −56.18 (−58.47 to −53.90) | −2.06 (−4.36 to 0.24) | −54.12 (−57.37 to −50.88) P < 0.0001 | −50.91 (−53.14 to −48.67) | 0.96 (−1.26 to 3.18) | −51.87 (−55.01 to −48.72) P < 0.0001 |
|||||||||||||||||||||||||||| |||||||||||||||||||||||||| | ||||||||||||| | |||||||||||||| | |||||||||||||| | |||||||||||||||||| | |||||||||||||| | |||||||||||||| | |||||||||||||||||| | |||||||||||||| | |||||||||||||| |
Time-adjusted change in LDL-C (LSM, 95% CI) | |||||||||
Time-adjusted percentage CFB in LDL-C after day 90 to day 540d | −38.08 (−41.03 to −35.14) | 6.22 (3.26 to 9.17) | −44.30 (−48.48 to −40.12) P < 0.0001 | −51.27 (−53.00 to −49.54) | 2.51 (0.77 to 4.25) | −53.78 (−56.23 to −51.33) P < 0.0001 | −45.82 (−47.52 to −44.13) | 3.35 (1.65 to 5.05) | −49.17 (−51.57 to −46.77) P < 0.0001 |
Time-adjusted absolute CFB in LDL-C after day 90 to day 540 (PMM), mg/dL | −56.58 (−60.98 to −52.17) | 6.17 (1.72 to 10.62) | −62.74 (−69.01 to −56.48) P < 0.0001 | −53.66 (−55.41 to −51.92) | −0.39 (−2.14 to 1.37) | −53.28 (−55.75 to −50.80) P < 0.0001 | −48.63 (−50.37 to −46.89) | 0.31 (−1.42 to 2.04) | −48.94 (−51.39 to −46.48) P < 0.0001 |
|||||||||||||||||||||| ||||||||||||||||||| |||||||||||||||||||| | ||||||||||||| | |||||||||||||| | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||| | |||||||||||||||||| | |||||||||||||||||| | |||||||||||||| | |||||||||||||||||| |
CFB = change from baseline; CI = confidence interval; ITT = intention to treat; LDL-C = low-density lipoprotein cholesterol; LSM = least squares mean; MAR = missing at random; MMRM = mixed-effects model for repeated measures; PMM = pattern-mixture model.
aA control-based PMM was used for missing data imputation with 100 total imputed datasets. An MMRM on each of the 100 datasets was performed by including fixed effects for treatment, visit, interaction between treatment and visit, and baseline LDL-C as a covariate.
bORION-11 used a modified washout model to adjust for missing data, such that patients in the inclisiran group who received all 4 doses of the study drug, had the day 510 value missing, and had evaluable data at day 540, had their intermittent missing day 510 values imputed assuming the missing data were MAR.
cAn MMRM analysis that assumes missing data are MAR was performed.
dA control-based PMM was used for missing data imputation with 100 total imputed datasets. An MMRM on each of the 100 datasets was performed by including fixed effects for treatment, visit, interaction between treatment and visit, and baseline LDL-C as a covariate. A linear combination of the estimated means after day 90 and up to day 540 was used to compare treatment groups. Treatment effects from the 100 analyses were combined using the Rubin method.
eAn MMRM analysis that assumes missing data are MAR was performed. The model included fixed effects for treatment, visit, interaction between treatment and visit, and baseline LDL-C as a covariate. The restricted maximum likelihood estimation approach was used with the covariance structure set as unstructured. A linear combination of the estimated means after day 90 and up to day 540 was used to compare treatment groups.
Source: Clinical Study Reports for ORION-9,17 ORION-10,18 and ORION-11.19
Health-Related Quality of Life
No patient-reported outcomes or HRQoL outcomes were evaluated in the included trials.
Neurocognitive Assessments
Neurocognitive testing was not conducted in the ORION trials.
Change in Laboratory Parameters
Change in laboratory parameters, including the percentage change in total cholesterol, ApoB, non-HDL-C, and triglycerides from baseline to day 510, were key secondary outcomes in the ORION-9, −10, and −11 trials. Results are summarized in Table 14. In ORION-9, −10, and −11, the percentage reduction in total cholesterol using PMM for inclisiran was −25.11 (95% CI, −27.83 to −22.39), −33.56 (95% CI, −35.09 to −32.03), and −28.00 (95% CI, −29.40 to −26.60), respectively, with a mean difference from placebo of −31.77 (95% CI, −35.59 to −27.94; P < 0.0001), −33.13 (95% CI, −35.30 to −30.97; P < 0.0001), and −29.79 (95% CI, −31.78 to −27.81; P < 0.0001), respectively.17-19 ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. In ORION-9, −10, and −11, the percentage reduction in ApoB using PMM for inclisiran was −33.14 (95% CI, −35.91 to −30.36), −44.81 (95% CI, −46.52 to −43.10) and −38.15 (95% CI, −39.76 to −36.54), respectively, with mean differences from placebo of −36.06 (95% CI, −39.99 to −32.14; P < 0.0001), −43.09 (95% CI, −45.50 to −40.67; P < 0.0001), and −38.94 (95% CI, −41.21 to −36.67; P < 0.0001), respectively.17-19 ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. In ORION-9, −10, and −11, the percentage reduction in non-HDL-C for inclisiran using PMM was −34.93 (95% CI, −38.46 to −31.40), −47.41 (95% CI, −49.44 to −45.38), and −41.16 (95% CI, −43.09 to −39.24), respectively, with mean differences from placebo of −42.36 (95% CI, −47.32 to −37.40; P < 0.0001), −47.36 (95% CI, −50.25 to −44.47; P < 0.0001), and −43.32 (95% CI, −46.04 to −40.60; P < 0.0001), respectively.17-19 ||||||||||||||||||||||||||||||||||||||||||||||||||||||. The percentage change in triglyceride levels was greater in the inclisiran groups compared with the placebo groups in all ORION trials, with a placebo-adjusted difference of 11.8%, 12.6%, and 7.0% in ORION-9, −10, and −11, respectively. Triglyceride levels were not key secondary end points and did not take into account the multiplicity of testing and the Hochberg procedure therefore, statistical significance was not assessed.35,43
Table 14: Percentage Change in TC, ApoB, Non-HDL-C, and Triglycerides (ITT Population)
Category | ORION-9 | ORION-10 | ORION-11 | ||||||
|---|---|---|---|---|---|---|---|---|---|
Inclisiran (N = 242) | Placebo (N = 240) | Difference from placebo | Inclisiran (N = 781) | Placebo (N = 780) | Difference from placebo | Inclisiran (N = 810) | Placebo (N = 807) | Difference from placebo | |
Percentage change in total cholesterol (LSM, 95% CI) | |||||||||
Percentage CFB to day 510 in TC (PMMa) | −25.11 (−27.83 to −22.39) | 6.66 (3.96 to 9.36) | −31.77 (−35.59 to −27.94) P < 0.0001 | −33.56 (−35.09 to −32.03) | −0.42 (−1.95 to 1.11) | −33.13 (−35.30 to −30.97) P < 0.0001 | −28.00 (−29.40 to −26.60) | 1.79 (0.38 to 3.21) | −29.79 (−31.78 to −27.81) P < 0.0001 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | |||||||||||||||||||| | |||||||||||| | ||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||| | ||||||||||||||||| | |||||||||||||||||| | ||||||||||| | ||||||||||||||||||| |
Percentage change in apolipoprotein B (LSM, 95% CI) | |||||||||
Percentage CFB to day 510 in ApoB (PMMa) | −33.14 (−35.91 to −30.36) | 2.93 (0.14 to 5.71) | −36.06 (−39.99 to −32.14) P < 0.0001 | −44.81 (−46.52 to −43.10) | −1.72 (−3.46 to 0.02) | −43.09 (−45.50 to −40.67) P < 0.0001 | −38.15 (−39.76 to −36.54) | 0.79 (−0.82 to 2.41) | −38.94 (−41.21 to −36.67) P < 0.0001 |
|||||||||||||||||||||||||||||||||||||||||||||||||||| | |||||||||||||||||||| | |||||||||||| | |||||||||||||||||||| | ||||||||||||||||||| | ||||||||||||||||| | ||||||||||||||||| | |||||||||||||||||| | |||||||||||||| | |||||||||||||||||| |
Percentage change in non-HDL-C (LSM, 95% CI) | |||||||||
Percentage CFB to day 510 in non-HDL-C (PMMa) | −34.93 (−38.46 to −31.40) | 7.43 (3.93 to 10.92) | −42.36 (−47.32 to −37.40) P < 0.0001 | −47.41 (−49.44 to −45.38) | −0.05 (−2.08 to 1.99) | −47.36 (−50.25 to −44.47) P < 0.0001 | −41.16 (−43.09 to −39.24) | 2.15 (0.22 to 4.09) | −43.32 (−46.04 to −40.60) P < 0.0001 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | |||||||||||||||||||| | ||||||||||||| | |||||||||||||||||||| | ||||||||||||||||||| | |||||||||||||||| | ||||||||||||||||| | |||||||||||||||||| | |||||||||| | ||||||||||||||||||| |
Percentage change in triglycerides (median) | |||||||||
Percentage CFB to day 510 in triglycerides (observed)c | −11.1 | −0.7 | −11.8 | −14.9 | −2.3 | −12.6 | −12.0 | −5.0 | −7.0 |
ApoB = apolipoprotein B; CFB = change from baseline; CI = confidence interval; HDL-C = high-density lipoprotein cholesterol; ITT = intention to treat; LDL-C = low-density lipoprotein cholesterol; LSM = least squares mean; MAR = missing at random; MMRM = mixed-effects model for repeated measures; PMM = pattern-mixture model; TC = total cholesterol.
aA control-based PMM was used for missing data imputation with 100 total imputed datasets. An MMRM on each of the 100 datasets was performed by including fixed effects for treatment, visit, interaction between treatment and visit, and baseline LDL-C as a covariate.
bAn MMRM analysis that assumed missing data are MAR was performed.
cCIs and P values are not shown for percentage change in triglycerides, as multiplicity of testing and the Hochberg procedure were not taken into account.
Source: Clinical Study Reports for ORION-9,17 ORION-10,18 and ORION-11.19
Harms
Only those harms identified in the review protocol are reported subsequently. Table 15 contains detailed harms data.
Treatment-Emergent Adverse Events
The incidence of TEAEs was consistent between inclisiran- and placebo-treated patients as well as across trials, with patients in both groups experiencing at least 1 TEAE (inclisiran versus placebo): 76.8% versus 71.7%, 73.5% versus 74.8%, and 82.7% versus 81.5% in ORION-9, −10, and −11, respectively.17-19
The incidence and type of common TEAEs were similar in the placebo and inclisiran groups in all trials; however, the most common TEAEs varied across the 3 ORION trials. In ORION-9, the most common TEAEs in the inclisiran and placebo groups were nasopharyngitis (11.6% versus 8.3%), influenza (5.4% versus 8.8%), upper respiratory tract infection (6.6% versus 6.7%), and back pain (7.1% versus 4.2%).17 The most common TEAEs in ORION-10 were diabetes mellitus (15.4% versus 13.9%), hypertension (5.4% versus 5.4%), back pain (5.0% versus 5.0%), bronchitis (5.9% versus 3.9%), and dyspnea (5.0% versus 4.2%).18 In ORION-11, the most common TEAEs were diabetes mellitus (10.9% versus 11.7%), nasopharyngitis (11.2% versus 11.2%), hypertension (6.5% versus 6.7%), and upper respiratory tract infection (6.4% versus 6.1%).19 ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.17-19
Serious Adverse Events
Treatment-emergent SAEs in ORION-9 occurred in 18 (7.5%) inclisiran-treated patients and 33 (13.8%) placebo-treated patients.17 The incidence of SAEs was higher in ORION-10 and −11, occurring in 175 (22.4%) and 181 (22.3%) inclisiran-treated patients, respectively, compared with 205 (26.3%) and 181 (22.5%) placebo-treated patients.18,19 There was no difference in the frequency of treatment-emergent SAEs between the treatment groups in ORION-9, ORION-10, and ORION-11. Treatment-emergent SAEs in ORION-9 included mostly CV events; the most common events included unstable angina (1.0%) and myocardial ischemia (0.8%).17 The most common SAEs in ORION-10 were CV-related, with 2.4%, 1.7%, and 1.7% of patients experiencing coronary artery disease, congestive cardiac failure, or acute MI, respectively.18 As with the 2 other ORION trials, the most common SAEs in ORION-11 were also CV-related and included angina pectoris (1.7%), acute MI (1.4%), and unstable angina (1.4%).19 |||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.17-19
Withdrawal Due to Adverse Events
In ORION-9, 3 patients (1.2%) in the inclisiran group withdrew due to an AE. No patients in the placebo group withdrew due to an AE. |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.17
The incidence of WDAEs in ORION-10 was similar across groups, occurring in 19 inclisiran-treated patients (2.4%) and 17 placebo-treated patients (2.2%). |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.18
The incidence of WDAEs in ORION-11 was similar to ORION-10, with 23 (2.8%) and 18 (2.2%) inclisiran- and placebo-treated patients, respectively, experiencing TEAEs that led to withdrawal. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||19
Notable Harms
Neurologic Events
Neurologic events and neurocognitive disorders are summarized in Table 15. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||.17-19
Injection-Site Reactions
Injection-site reactions are summarized in Table 15. In all trials, fewer placebo-treated patients reported TEAEs at the injection site than those treated with inclisiran. The incidence of TEAEs at the injection site for inclisiran- and placebo-treated patients was 41 (17.0%) versus 4 (1.7%) in ORION-9, 47 (6.0%) versus 15 (1.9%) in ORION-10, and 62 (7.6%) versus 14 (1.7%) in ORION-11. Injection-site reactions were mild to moderate, and no severe reactions were seen across trials. One patient withdrew from the study due to injection-site reactions in the ORION-9 and ORION-10 inclisiran groups, while 2 patients in the ORION-11 inclisiran group withdrew from the study due to injection-site reactions. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.
Hypersensitivity Reactions
Hypersensitivity reactions are summarized in Table 15. TEAEs related to hypersensitivity did not differ between groups in all 3 trials. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.17-19
Renal and Hepatic Safety
Renal and hepatic safety is summarized in Table 15. Renal events were not different between inclisiran and placebo groups across trials, |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.17-19
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||17-19
Clinical Laboratory Evaluations
The incidence of clinically significant creatinine kinase changes was recorded as part of the clinical laboratory evaluation in ORION-9, −10, and −11. A detailed summary of clinically significant creatinine kinase changes at the end of the study is provided in Table 35 in Appendix 3. |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.19
Table 15: Harms (Safety Population)
Harms | ORION-9 | ORION-10 | ORION-11 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
Inclisiran (N = 241) | Placebo (N = 240) | Inclisiran (N = 781) | Placebo (N = 778) | Inclisiran (N = 811) | Placebo (N = 804) | |||||||
n (%) | E | n (%) | E | n (%) | E | n (%) | E | n (%) | E | n (%) | E | |
TEAEs | ||||||||||||
Patients with ≥ 1 TEAE | 185 (76.8) | 663 | 172 (71.7) | 588 | 574 (73.5) | 2,559 | 582 (74.8) | 2,639 | 671 (82.7) | 2,893 | 655 (81.5) | 2,605 |
Most common TEAEsa | ||||||||||||
Nasopharyngitis | 28 (11.6) | 36 | 20 (8.3) | 21 | 21 (2.7) | ||| | 24 (3.1) | ||| | 91 (11.2) | 105 | 90 (11.2) | 110 |
Injection-site reaction | 22 (9.1) | 37 | 0 (0) | 0 | |||| | ||| | ||| | | | |||| | ||| | ||| | | |
Back pain | 17 (7.1) | 19 | 10 (4.2) | 11 | 39 (5.0) | 42 | 39 (5.0) | 41 | 27 (3.3) | ||| | 28 (3.5) | ||| |
Upper respiratory tract infection | 16 (6.6) | 19 | 16 (6.7) | 22 | 37 (4.7) | ||| | 38 (4.9) | ||| | 52 (6.4) | 59 | 49 (6.1) | 57 |
Influenza | 13 (5.4) | 15 | 21 (8.8) | 24 | ||||| | ||||| | ||||| | | | 19 (2.3) | ||||| | 20 (2.5) | ||| |
Bronchitis | 9 (3.7) | ||||| | 4 (1.7) | | | 46 (5.9) | 54 | 30 (3.9) | 38 | 23 (2.8) | ||||| | 16 (2.0) | ||| |
Hypertension | 9 (3.7) | | | 8 (3.3) | | | 42 (5.4) | 43 | 42 (5.4) | 43 | 53 (6.5) | 60 | 54 (6.7) | 59 |
Arthralgia | 9 (3.7) | | | 7 (2.9) | | | 35 (4.5) | ||| | 33 (4.2) | ||| | 47 (5.8) | 56 | 32 (4.0) | 37 |
Diabetes mellitus | ||||| | | | ||||| | | | 120 (15.4) | 125 | 108 (13.9) | 113 | 88 (10.9) | 101 | 94 (11.7) | 100 |
Osteoarthritis | ||||| | | | ||||| | | | |||| | ||||| | |||| | ||| | 32 (3.9) | 37 | 40 (5.0) | 43 |
Dyspnea | ||||| | | | ||||| | | | 39 (5.0) | 41 | 33 (4.2) | 36 | |||| | ||| | ||| | | |
SAEs | ||||||||||||
Patients with ≥ 1 SAE | 18 (7.5) | 23 | 33 (13.8) | 48 | 175 (22.4) | 339 | 205 (26.3) | 422 | 181 (22.3) | 283 | 181 (22.5) | 293 |
Most common SAEsb | ||||||||||||
CAD | ||||| | | | ||||| | | | 15 (1.9) | 15 | 22 (2.8) | 25 | 8 (1.0) | 8 | 11 (1.4) | 15 |
Cardiac failure congestive | ||||| | | | ||||| | | | 7 (0.9) | 7 | 20 (2.6) | 30 | ||||| | | | ||||| | | |
Acute MI | 2 (0.8) | 2 | 1 (0.4) | 1 | 14 (1.8) | 15 | 12 (1.5) | 13 | 5 (0.6) | 5 | 18 (2.2) | 21 |
Unstable angina | 1 (0.4) | 1 | 4 (1.7) | 4 | 4 (0.5) | 5 | 10 (1.3) | 12 | 11 (1.4) | 12 | 11 (1.4) | 11 |
Myocardial ischemia | 1 (0.4) | 1 | 3 (1.3) | 3 | ||||| | ||||| | ||||| | | | ||| | | | ||||| | | |
Pneumonia | | | | | | | | | 11 (1.4) | 12 | 9 (1.2) | 9 | 9 (1.1) | 9 | 7 (0.9) | 7 |
Non-cardiac chest pain | | | | | | | | | 10 (1.3) | 10 | 9 (1.2) | 9 | 4 (0.5) | 4 | 8 (1.0) | 8 |
Atrial fibrillation | | | | | | | | | 10 (1.3) | 11 | 8 (1.0) | 9 | 10 (1.2) | 11 | 6 (0.7) | 7 |
COPD | | | | | | | | | 8 (1.0) | 10 | 8 (1.0) | 10 | | | | | | | | |
Angina pectoris | | | | | | | | | | | | | | | | | 14 (1.7) | 14 | 13 (1.6) | 14 |
Occlusive PAD | | | | | | | | | | | | | | | | | 7 (0.9) | 7 | 8 (1.0) | 9 |
WDAEs | ||||||||||||
Incidence of WDAEs (SOC) | 3 (1.2) | | | 0 (0) | | | 19 (2.4) | | | 17 (2.2) | | | 23 (2.8) | | | 18 (2.2) | | |
Notable harms | ||||||||||||
|||||||||||||||||||||||||||||||||||||||||| | | | | | | | | | | | | | | | | | | | | | | | | |
Patients with at least 1 TEAE at the injection siteb | 41 (17.0) | | | 4 (1.7) | | | 47 (6.0) | | | | 15 (1.9) | | | | 62 (7.6) | | | | 14 (1.7) | | | |
ALT = alanine aminotransferase; AST = aspartate aminotransferase; CAD = coronary artery disease; COPD = chronic obstructive pulmonary disease; E = event count; GFR = glomerular filtration rate; MI = myocardial infarction; NR = not reported; PAD = peripheral artery disease; SAE = serious adverse event; SOC = system organ class; TEAE = treatment-emergent adverse event; WDAE = withdrawal due to adverse event.
aOccurring in ≥ 5% of patients in 1 group.
bOccurring in ≥ 1% of patients in 1 group.
cOccurring in ≥ 0.5% of patients in 1 group.
dClinically relevant injection-site reactions.
Source: Clinical Study Reports for ORION-9,17 ORION-10,18 and ORION-11.19
Critical Appraisal
Internal Validity
ORION-9, ORION-10, and ORION-11 were all phase III, double-blind RCTs. Appropriate methods for randomization (using interactive response technology), treatment allocation (stratified by country and current use of statins or other LMTs in block sizes of 4), and maintenance of blinding to treatment assignment were used in all 3 trials reducing selection, performance, and detection biases. |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. There were no notable differences in baseline characteristics within the studies and there was no imbalance in discontinuations, suggesting that attrition bias was limited. Moreover, unblinding was permitted only in the event of an emergency or AE for which it was necessary to know the study drug to determine an appropriate course of therapy. Injection-site reactions are known complications of PCSK9 inhibitors and were more frequent in the inclisiran groups across trials, which could have revealed treatment assignment; overall, injection-site reactions were not common, and it is unclear what effect this would have had on the results and unblinding.
The primary analyses of the ORION trials were conducted in the ITT population. Low dropout rates were seen in all trials; however, the total number of missing data were not reported for any outcomes in the trials and, therefore, the extent of missing data in each group, at various time points and for each key outcome, is unknown. Efforts were made to reduce the amount of missing data, including diligent follow-up. Missing data were imputed in the co-primary end point of percentage change in LDL-C from baseline to day 510 using a multiple imputation washout model on actual values with baseline and observed efficacy measures, and current use of statins or other LMTs as covariates. Missing values in the inclisiran group were imputed under the assumption that their outcome was similar to the outcomes in the placebo group, with similar background characteristics. This method may be subject to bias, resulting in greater treatment effects in favour of inclisiran. For patients in the inclisiran group, only missing day 510 values were imputed. For patients in the placebo group, missing values over all visits after early termination were imputed based on the MAR assumption. Again, this may impact the direction of treatment effect in favour of inclisiran over time. Results of the co-primary and secondary outcomes were consistently large, with minimal differences between observed and imputed values. Numerous sensitivity analyses, with and without multiple imputations, were also employed. Therefore, it is unlikely that missing data would have impacted the LSM percentage change in LDL-C.
The pre-specified power and sample size calculation for all ORION trials was identical and was based on the difference in change from baseline in LDL-C between the inclisiran and placebo groups being no less than 30 mg/dL, with a standard deviation of 20 mg/dL; however, the enrolled populations were much higher than what was defined by the power and sample size calculation. |||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. The additional patients enrolled in the ORION trials could have led to the overpowering of study results, whereby the higher number of patients enrolled may increase the probability of seeing minuscule differences between groups. However, overpowering is unlikely to have affected the results of the ORION studies, given the large differences in efficacy observed between inclisiran and placebo and, given the request by the FDA, any ethical or resource-allocation issues are of no concern for the ORION trials. The higher sample sizes contributed additional safety information for all trials; however, secondary outcomes and CV-related events considered of interest to this review were not accounted for.
Acceptable methods to account for multiplicity were used in all trials. The co-primary efficacy outcome was controlled for multiplicity using the family-wise error rate using a nested testing procedure first on the percentage change in LDL-C from baseline to day 510, and then on the time-adjusted percentage change in LDL-C from baseline after day 90 and up to day 540. The Hochberg procedure was applied for key secondary end points.18,19,46 Other secondary end points and exploratory end points were not controlled for multiplicity or missing data, including the composite outcome of MACE, which was considered of clinical importance to this review. The proportion of patients reaching global lipid targets (LDL-C < 100 mg/dL or < 70 mg/dL) were secondary end points that were also not controlled for multiplicity or missing data. The sponsor’s evaluation of these outcomes was conducted on the ITT population; however, there was a discrepancy between the number of patients in the ITT population and the reported number of patients at day 510, with the proportion of patients missing from the analysis ranging from 5% to 15% across trials. The resulting missing patients inflated the proportions of patients achieving global lipid targets in both the inclisiran and placebo arms.11,29,47
Subgroup analyses for efficacy and safety were pre-specified in the statistical analysis plan, and missing data were accounted for using the MMRM method but were not adjusted for multiplicity. CIs for most subgroup analyses suggested precision; however, those subgroups with a lower number of patients had wider, more imprecise CIs. Subgroups based on risk status (ASCVD or an ASCVD risk equivalent) were conducted for ORION-9 and ORION-11; however, the ASCVD risk–equivalent subgroup in ORION-11 was not considered of interest, given that it was mostly made up of patients who were not part of the population for which reimbursement was requested by the sponsor (only 11 patients had HeFH and the rest had an ASCVD risk equivalent).
External Validity
The ORION-9, −10, and −11 trials aimed to enrol patients with ASCVD and/or an ASCVD risk equivalent and within specific cut-offs for serum LDL-C and triglyceride. |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. Screening failures and inclusion criteria were considered appropriate for the ORION-10 and −11 trials, given the specified LDL-C cut points of 1.8 mmol/L and 2.6 mmol/L, which are aligned with current CCS guidelines; therefore, the threat of sampling bias is estimated to be low, given the eligibility criteria. The included patient populations in the ORION studies were mostly reflective of the funding request; however, ORION-11 included a proportion of patients with ASCVD risk equivalents (13%), which were not part of the reimbursement population requested by the sponsor.
With the exception of ORION-10, the ORION trials were multinational; however, ORION-9 was the only study that enrolled Canadian patients (n = 23). The proportion of patients receiving high-intensity statins at baseline was as expected for patients with HeFH in ORION-9 (73.9%), as well as for the patients with ASCVD in ORION-10 (69.4%) and −11 (78.0%); however, the clinical expert believed that more patients with HeFH would be receiving ezetimibe compared with what was seen in ORION-9 (52.3%). Partial or complete intolerance to statins at baseline ranged from 11.4% to 25.3%, which was in line with the 15% to 20% proportion estimated by the clinical expert. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. The inclusion criteria in the ORION-9, −10, and −11 studies excluded patients if they had previously received or were currently receiving treatment with PCSK9 monoclonal antibodies within 90 days of screening, and the baseline characteristics of patients in the ORION trials did not include the proportion of patients who had prior treatment with PCSK9 inhibitors. The product monograph for inclisiran states that when transitioning from a PCSK9 inhibitor to inclisiran, the last dose of PCSK9 should be delivered and then, at the next scheduled date, inclisiran can be administered; however, the efficacy of switching from other PCSK9 inhibitors to inclisiran in terms of reducing LDL-C, CV-related morbidity, and mortality, remains uncertain. Despite this and, according to the clinical expert consulted by CADTH, the baseline demographic and medical characteristics of the ORION trials reflect the HeFH and ASCVD populations that are expected to use inclisiran in Canada.
All of the ORION studies were placebo-controlled trials and did not include an active comparator. This allows for adequate evaluation of the treatment effect of inclisiran; however, it may overestimate the treatment effects. Demonstration of effectiveness compared with another PCSK9 inhibitor would have allowed for better interpretation of efficacy results. Despite this, the clinical expert consulted by CADTH noted that the incremental improvements in LDL-C and differences between inclisiran and the placebo groups are still clinically meaningful given that, in both the clinical trial and real-world settings, patients with HeFH and/or ASCVD are heavily treated with background medications (i.e., statins, ezetimibe, blood pressure medication, diabetes medications); therefore, the differences seen were still notable. Given there were no direct comparisons between inclisiran and alirocumab or evolocumab, the 2 available PCSK9 inhibitors available in Canada, the sponsor provided an ITC to address this gap.
The outcomes used to inform on the efficacy of inclisiran were chosen based on validated laboratory assessments of lipids. They are considered widely accepted surrogates for clinically relevant outcomes and are reflective of and important in guiding treatment decisions in Canadian clinical practice. The duration of the trial (18 months) was also considered appropriate for assessing these outcomes over time, given that the trials for the PCSK9 inhibitors alirocumab and evolocumab were 12 to 24 weeks in duration and that effects on lipids are rapidly seen.11 However, the included studies were not designed to assess important CV-related outcomes, including reductions in MACEs and all-cause and CV-related mortality, as these outcomes were exploratory and not powered for statistical analysis. Across trials, there were no differences between groups and, therefore, there was no cause for concern for these outcomes. However, the impact of inclisiran on these outcomes remains uncertain, and the duration of the trial was considered too short for assessing reductions in these outcomes. Moreover, the product monograph for inclisiran states that the effect of inclisiran on CV morbidity and mortality has not been determined.16
No HRQoL or patient-reported outcomes were assessed in the ORION trials and, therefore, the effect of inclisiran with respect to these outcomes remains unknown.
Indirect Evidence
Objectives and Methods for the Summary of Indirect Evidence
Due to the lack of direct evidence comparing inclisiran with other existing therapies as monotherapy, or as add-on therapy in the treatment of adult patients with HeFH or ASCVD, the sponsor submitted an ITC that was used to inform the pharmacoeconomic model.20 The objective of this section is to summarize and critically appraise the methods and findings of the sponsor-submitted ITC comparing inclisiran with relevant drug comparators for the treatment of adult patients with HeFH or ASCVD with high LDL-C that is uncontrolled by MTD statins, with or without ezetimibe, or those with uncontrolled LDL-C who are statin-intolerant or contraindicated, with or without ezetimibe.
A focused literature search for NMAs dealing with hypercholesterolemia was run in MEDLINE All (1946–) and Embase (1974–) on April 14, 2021. No limits were applied to the search. Retrieval was not limited by publication date or by language. Articles were screened by 1 researcher for ITCs that met the patient, intervention, comparator, and outcome criteria listed in Table 5. In addition, the sponsor-submitted ITC was reviewed.
The literature search for NMAs identified 103 articles; however, no studies evaluated the efficacy and safety of inclisiran in patients with HeFH or ASCVD against relevant comparators.
Description of Indirect Comparison(s)
The sponsor submitted an ITC that compared the efficacy of inclisiran with relevant drug comparators in patients with HeFH or ASCVD (or an ASCVD risk equivalent). The sponsor first conducted a systematic literature review to evaluate various efficacy, safety, and HRQoL outcomes to assess the feasibility of conducting an NMA. Thirty-nine studies met the inclusion criteria of the systematic review and feasibility assessment, and 22 studies were sub-selected for inclusion in the NMA based on network connectivity and whether there were any differences in the study, patient, or outcome characteristics that were likely modifiers of the relative treatment effects.20 Table 16 summarizes the available selection criteria specific to the ITC and NMA, as well as the methods for study selection for the systematic literature review.
Table 16: Study Selection Criteria and Methods for the NMA
Detail | Sponsor-submitted NMA |
|---|---|
Population | Adults (≥ 18 years) with HeFH or ASCVD who are:
|
Intervention | Inclisiran 284 mg |
Comparatora |
|
Outcome |
|
Study design | RCTs |
Publication characteristics | Full-text, peer-reviewed publications, conference abstracts and presentations, SLRs, and sponsor data on file |
Exclusion criteria | Trials involving patients with HoFH or those using a low-intensity background statin or with no prior statin treatment (unless intolerant or contraindicated) and non-RCTs with < 12 weeks’ follow-up and < 10 patients per group were excluded; editorials, press releases, expert opinion, letters, and case studies were also excluded |
Databases searched | The following databases were to be searched: Ovid MEDLINE, Embase (Ovid), Cochrane Central Register of Controlled Trials, PubMed, Conference Proceedings Citation Index – Science. Hand searches of clinical trial registries and conferences were also conducted. |
Selection process | Titles and abstracts were screened using the Covidence online screening tool followed by an independent review of records by 2 researchers. Full-text citations were reviewed independently by 2 reviewers according to the predefined inclusion criteria. |
Data extraction process | Two independent researchers imported extracted data into the predefined extraction forms. A single researcher collated the data from both researchers to identify discrepancies, and a third researcher was involved to resolve discrepancies. |
Quality assessment | The Cochrane risk-of-bias assessment tool was used to assess the quality of included RCTs |
AE = adverse event; ASCVD = atherosclerotic cardiovascular disease; CFB = change from baseline; HDL-C = high-density lipoprotein cholesterol; HeFH = heterozygous familial hypercholesterolemia; HoFH = homozygous familial hypercholesterolemia; LDL-C = low-density lipoprotein cholesterol; MTD = maximally tolerated dose; NMA = network meta-analysis; q.2.w. = every 2 weeks; RCT = randomized controlled trial; SC = subcutaneous; SLR = systematic literature review.
aOnly statins are used in the MTD statin population and uncontrolled LDL-C ± ezetimibe population.
Source: Sponsor-submitted NMA.20
ITC Methods
Objectives
The objective of the sponsor-submitted report was to conduct a feasibility assessment through a systematic literature review and, if possible, to conduct an ITC evaluating the relative efficacy and safety of inclisiran versus relevant drug comparators, including ezetimibe and other PCSK9 inhibitors, in patients with HeFH or ASCVD (or who had an ASCVD risk equivalent).20
Study Selection Methods
The sponsor-submitted NMA was informed by a systematic review of RCTs conducted in April 2020. The sponsor provided the protocol and plan for analyses in a separate report. Briefly, eligible publications were full-text, peer-reviewed RCTs. The planned method for identifying citations was through searches of MEDLINE, Embase, and the Cochrane Central Register of Controlled Trials databases, among others. Study selection and data extraction were planned to be conducted by 2 independent reviewers with discrepancies resolved by discussion using the Covidence online screening tool.20 Assessment of the quality of included studies was planned using the Cochrane risk-of-bias assessment tool; however, no quality assessment was included in the NMA report.
Table 17 summarizes the predefined study selection criteria for the systematic review in the sponsor-submitted ITC and NMA. The list of comparators and outcomes of interest included in the literature review was broader than that of the NMA; otherwise, the patient population was similar. No limitations on publication language were applied. The eligible patient population for the review included adults with HeFH or ASCVD with inadequate LDL-C control on MTD statins or who are statin-intolerant or contraindicated, which was identical to the NMA population. Multiple networks were constructed based on the HeFH, ASCVD, and ASCVD risk–equivalent population. The outcomes of interest to the NMA were percentage and absolute change in LDL-C at 24 weeks, total discontinuations and discontinuations due to AEs at 24 weeks, and percentage change in HDL-C at 24 weeks.20
Table 17: PICOS Criteria for the SLR to Identify Trials for the Sponsor-Submitted NMA
PICOS component | Inclusion criteria | Exclusion criteria |
|---|---|---|
Population |
|
|
AE = adverse event; AIDS = AIDS; ASCVD = atherosclerotic cardiovascular disease; CFB = change from baseline; CV = cardiovascular; FH = familial hypercholesterolemia; HDL-C = high-density lipoprotein cholesterol; HeFH = heterozygous familial hypercholesterolemia; HoFH = homozygous familial hypercholesterolemia; HRQoL = health-related quality of life; hs-CRP = high-sensitivity C-reactive protein; HTA = health technology assessment; LDL-C = low-density lipoprotein cholesterol; LMT = lipid-modifying therapy; MTD = maximally tolerated dose; NR = not reported; NYHA = New York Heart Association; PCSK9 = proprotein convertase subtilisin/kexin type 9; PICOS = population, intervention, comparison, outcomes, and study; RCT = randomized controlled trial; SAE = serious adverse event; SLR = systematic literature review; TC = total cholesterol; TRAE = treatment-related adverse event; VLDL-C = very low–density lipoprotein cholesterol.
The rest of the data in this table has been redacted at the request of the sponsor, so the redacted rows have been deleted.
Source: Sponsor-submitted SLR protocol.20
Feasibility Assessment
A feasibility assessment was conducted to review the studies identified in the systematic literature review, which included the following criteria:
determination of a connected network comparing the treatments of interest regarding the outcomes of interest
differences in the study, patient, or outcome characteristics across comparisons that are likely modifiers of the relative treatment effects.
The primary outcomes of interest for the NMA were percentage, absolute, and time-adjusted change from baseline in LDL-C; percentage change from baseline in HDL-C; total discontinuations; and discontinuations due to AEs.20 Several study design, patient, and intervention characteristics were identified a priori as potential treatment-effect modifiers. Key assumptions and recommendations from the feasibility assessment for the approach used in the sponsor-submitted NMA are summarized in Table 18 and included the following:
Background Ezetimibe and Statins
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. In the NMA, it was assumed that individual statins (atorvastatin, rosuvastatin, and simvastatin) had similar efficacy as background therapy, regardless of dose, and would not bias the results of the NMA. |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.20
CV Risk and Severity
Studies included in the feasibility assessment were inconsistent in their reporting of CV risk and severity, and definitions of risk equivalent varied across trials. Populations in the ORION trials included patients considered to have an ASCVD “risk equivalent,” defined by the presence of type 2 diabetes, FH, or a 20% or greater 10-year risk of a CV event as assessed by the Framingham Risk Score for CV disease or equivalent. |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.20
Treatment Dosing
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||20
Time of Assessment and Follow-Up
Follow-up for the ORION trials were 540 days; however, other PCSK9 inhibitors had much shorter durations of follow-up. Twenty-four weeks was selected as the preferred base-case time point for multiple reasons. |||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||| | ||||||||||||||||||||||||||||| | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | |||||| ||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. For safety outcomes, results were presented at end of study and, given the variation in follow-up, end-of-study outcomes were considered comparable if the duration of follow-up was 24 weeks or longer. |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||.20
Table 18: Summary of Main Assumptions and Recommendations in the Sponsor-Submitted NMA
Potential effect modifier | Assumptions | Recommendation |
|---|---|---|
Population characteristics | ||
Background ezetimibe | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |
Background statins | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||, it was assumed that individual statins (e.g., atorvastatin, rosuvastatin, simvastatin) would have similar efficacy as background therapy, regardless of the specific statin and dosage. |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| Small proportion of statin-intolerant patients in the ORION trials (ORION-10 [22%], ORION-11 [12%], ORION-9 [25%]) would not bias the NMA. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||| |||||||||||||||||||||||||||| |
CV risk and severity | Differences in CV risk and severity of patients within each population strata of interest (i.e., HeFH and ASCVD or an ASCVD risk equivalent) would not impact the relative effects observed for efficacy outcomes focused on changes in LDL-C ||||||||||||||||||. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||| |
Treatment characteristics | ||
Inclisiran | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||| | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |
Alirocumab | |||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||| | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||| | |||||||||||||||||||||||||||||||||||||||| |
Evolocumab | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | ||||||||||||||||||||| | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |
Ezetimibe | ||||||||||||| | |||||||||||||||||||||||||||||||||||||||||||| |
Placebo | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||| | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||| |
Outcome characteristics | ||
Time points | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | 24 weeks (||||||||||||||||||||||||||) was selected as the preferred time point of interest for the base case. |||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |
For safety outcomes of interest, results are presented at end of study; therefore, given the variation in follow-up, end-of-study outcomes were considered comparable if the duration of follow-up was 24 weeks or longer. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | |
ASCVD = atherosclerotic cardiovascular disease; CV = cardiovascular; FH = familial hypercholesterolemia; HeFH = heterozygous familial hypercholesterolemia; ITT = intention to treat; LDL-C = low-density lipoprotein cholesterol; MTD = maximally tolerated dose; NMA = network meta-analysis; PCSK9 = proprotein convertase subtilisin/kexin type 9; q.2.w. = every 2 weeks; q.m. = every month; SA = sensitivity analysis.
Source: Sponsor-submitted NMA.20
ITC Analysis Methods
The NMA methods are briefly summarized in Table 19. The analyses were conducted within a Bayesian framework. A selection of both fixed- and random-effects analyses was conducted for outcomes of interest. Random-effects analyses were selected as the base case, given the number of studies per node and the observed heterogeneity in patient and trial characteristics, ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||.20
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 19: ITC Analysis Methods
Characteristics | Sponsor-submitted NMA |
|---|---|
ITC methods | |||||||||||||||||||||||||||||||||||||||||||||||||| |
Priors | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |
Assessment of model fit | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |
Assessment of consistency | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |
Assessment of convergence | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |
Outcomes | Outcomes included percentage and absolute change in LDL-C from baseline to 24 weeks, total discontinuations and discontinuations due to AEs at 24 weeks, and percentage change in HDL-C at 24 weeks |
Follow-up time points | 24-week follow-up was chosen as the base case |
Construction of nodes | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |
Sensitivity analyses | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |
Subgroup analysis | |||| |
Methods for pairwise meta-analysis | | |
AE = adverse event; ASCVD = atherosclerotic cardiovascular disease; DIC = deviance information criterion; FE = fixed effects; HDL-C = high-density lipoprotein cholesterol; HeFH = heterozygous familial hypercholesterolemia; ITC = indirect treatment comparison; ITT = intention to treat; LDL-C = low-density lipoprotein cholesterol; MTD = maximally tolerated dose; NMA = network meta-analysis; NR = not reported; q.2.w. = every 2 weeks; q.m. = every month; RE = random effect; SA = sensitivity analysis; SD = standard deviation.
Source: Sponsor-submitted NMA.20
Results of the ITC
Summary of Included Studies
The results of the systematic review were included in a separate report. |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Results
HeFH Populations
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
The network for the HeFH population on MTD statins is shown in Figure 5. A total of 7 trials were included in the network. ||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Figure 5: Network Diagram for HeFH Populations on MTD Statin
HeFH = heterozygous familial hypercholesterolemia; LDL-C = low-density lipoprotein cholesterol; MTD = maximally tolerated dose.
Note: Interventions and placebo arms are in addition to background statin with or without other lipid-lowering therapy; no network is feasible for statin-intolerant patients.
Red text indicates the trial was excluded in a sensitivity analysis.
* Subgroup data for patients with HeFH were used in the analysis.
Source: Sponsor-submitted network meta-analysis.20
Base-Case Results
Base-case results for the percentage change in LDL-C at 24 weeks, absolute change in LDL-C at 24 weeks, total discontinuations at 24 weeks or greater, discontinuations due to AEs at 24 weeks or greater, and percentage change in HDL-C at 24 weeks in the HeFH populations on MTD statin are summarized in Table 20.
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 20: HeFH MTD Random-Effects NMA Results
Comparator | Random-effects difference (95% CrI) |
|---|---|
Percentage change in LDL-C at 24 weeks (mean difference) | |
|||||||||||||||||||||||||| | ||||| |
|||||||||||||||||||||||||||| | |||||||||| |
|||||||||||||||||||||| | |||||||| |
|||||||||||||||||||||||||||| | |||||||||| |
|||||||||||||||||||||||||||||| | |||||||||| |
AE = adverse event; CrI = credible interval; HDL-C = high-density lipoprotein cholesterol; HeFH = heterozygous familial hypercholesterolemia; LDL-C = low-density lipoprotein cholesterol; MTD = maximally tolerated dose; NMA = network meta-analysis.
The rest of the data in this table has been redacted at the request of the sponsor, so the redacted rows have been deleted.
Source: Sponsor-submitted NMA.20
Sensitivity Analysis
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 21: HeFH MTD — Sensitivity Analysis Results for Difference in Percentage and Absolute Change in LDL-C
Comparator | Difference in CFB (95% CrI) | |
|---|---|---|
Percentage change in LDL-C | Absolute change in LDL-C | |
|||||||||| | ||
|||||||||| | |||||| | |||||||||| |
|||||||||||| | |||||| | |||||||||||| |
|||||| | |||||||||||| | |||||||||||||||| |
CFB = change from baseline; CrI = credible interval; HeFH = heterozygous familial hypercholesterolemia; LDL-C = low-density lipoprotein cholesterol; MTD = maximally tolerated dose; NR = not reported; SA = sensitivity analysis.
The rest of the data in this table has been redacted at the request of the sponsor, so the redacted rows have been deleted.
Source: Sponsor-submitted network meta-analysis.20
ASCVD and ASCVD Risk–Equivalent Populations on MTD Statins
The network for the ASCVD and ASCVD risk–equivalent populations on MTD statins is displayed in Figure 6. A total of 13 studies were included in the base-case network. |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Figure 6: Network Diagram for ASCVD and ASCVD Risk–Equivalent Populations on MTD Statin
ASCVD = atherosclerotic cardiovascular disease; MTD = maximally tolerated dose.
Note: Interventions and placebo arms are in addition to background statin with or without other lipid-lowering therapy.
Red text indicates the trial was excluded in a sensitivity analysis.
Grey text indicates the trial was included only in a sensitivity analysis.
Source: Sponsor-submitted network meta-analysis.20
Base-Case Results
Base-case results for the percentage change in LDL-C at 24 weeks, absolute change in LDL-C at 24 weeks, total discontinuations at 24 weeks or greater, discontinuations due to AEs at 24 weeks or greater, and the percentage change in HDL-C at 24 weeks in the ASCVD and ASCVD risk–equivalent populations on MTD statins is summarized in Table 22.
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 22: Random-Effects NMA Results for ASCVD and ASCVD Risk–Equivalent Populations on MTD Statins
Comparator | Random-effects difference (95% CrI) |
|---|---|
Percentage change in LDL-C at 24 weeks (mean difference) | |
||||||||||||||||||||||| | |||||||||| |
||||||||||||||||||||||||| | |||||||| |
|||||||||||||||||||||||||| | |||||||||| |
|||||||||||||||||||||||||| | |||||||||| |
AE = adverse event; ASCVD = atherosclerotic cardiovascular disease; CrI = credible interval; HDL-C = high-density lipoprotein cholesterol; LDL-C = low-density lipoprotein cholesterol; MTD = maximally tolerated dose; NMA = network meta-analysis.
The rest of the data in this table has been redacted at the request of the sponsor, so the redacted rows have been deleted.
Source: Sponsor-submitted NMA.20
Sensitivity Analysis
The results of the sensitivity analyses, for percentage and absolute change in LDL-C at 24 weeks, are summarized in Table 23.
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 23: Sensitivity Analysis of Random-Effects NMA Results for the ASCVD and ASCVD Risk–Equivalent Populations on MTD Statins
Comparator | Difference in CFB (95% CrI) | |
|---|---|---|
Percentage change in LDL-C | Absolute change in LDL-C | |
|||||||||||||||||||||||||||||||||||||||||||||||||| | ||
|||||||||| | |||||||||| | |||||||| |
|||||||||||| | |||||| | |||||||| |
|||||||||| | |||||||||||||||| | |||||||||||| |
|||||| | |||||||||||||||| | |||||||||||||| |
ASCVD = atherosclerotic cardiovascular disease; CFB = change from baseline; CrI = credible interval; LDL-C = low-density lipoprotein cholesterol; MTD = maximally tolerated dose; NMA = network meta-analysis; NR = not reported; SA = sensitivity analysis.
The rest of the data in this table has been redacted at the request of the sponsor, so the redacted rows have been deleted.
Source: Sponsor-submitted NMA.20
ASCVD and ASCVD Risk–Equivalent Populations Intolerant to Statins
A total of 7 trials were included in the network for ASCVD and ASCVD risk–equivalent populations intolerant to statins (Figure 7). There were no closed loops.
Figure 7: Network Diagram for ASCVD and ASCVD Risk–Equivalent Populations Intolerant to Statins
ASCVD = atherosclerotic cardiovascular disease.
Note: Interventions and placebo arms are in addition to background statin with or without other lipid-lowering therapy.
Grey text indicates the trial was included only in a sensitivity analysis.
* Subgroup data for statin-intolerant patients to be used in the analysis.
Source: Sponsor-submitted network meta-analysis.20
Base-Case Results
Table 24 summarizes the base-case results for the percentage change in LDL-C at 24 weeks, absolute change in LDL-C at 24 weeks, total discontinuations at 24 weeks or greater, discontinuations due to AEs at 24 weeks or greater, and percentage change in HDL-C at 24 weeks in the ASCVD and ASCVD risk–equivalent populations on MTD statins.
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||
Table 24: Random- and Fixed-Effects NMA Results for ASCVD and ASCVD Risk–Equivalent Populations Intolerant to Statins
Comparator | Random- or fixed-effects differencea (95% CrI) |
|---|---|
Percentage change in LDL-C at 24 weeks (mean difference) | |
|||||||||||||||||||||||| | |||||||| |
||||||||||||||||||||||| | ||||| |
|||||||||||||||||||||||||||| | |||||| |
||||||||||||||||||| | |||||| |
AE = adverse event; ASCVD = atherosclerotic cardiovascular disease; CrI = credible interval; HDL-C = high-density lipoprotein cholesterol; LDL-C = low-density lipoprotein cholesterol; NMA = network meta-analysis.
The rest of the data in this table has been redacted at the request of the sponsor, so the redacted rows have been deleted.
aFixed-effects analysis were conducted for the total discontinuations and discontinuations due to AEs at ≥ 24 weeks.
Source: Sponsor-submitted NMA.20
Sensitivity Analysis
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 25: Sensitivity Analysis Random-Effects NMA Results for the ASCVD and ASCVD Risk–Equivalent Populations Intolerant to Statins
Comparator | Difference in CFB (95% CrI) | |
|---|---|---|
Percentage change in LDL-C | Absolute change in LDL-C | |
|||||||||| | ||
|||||||||| | |||||||||| | |||||||| |
|||||||||||| | |||||||||| | |||||| |
|||||||||| | |||||||||| | |||||||||||| |
|||||| | |||||||||||||| | |||||||||||||| |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | ||
|||||||||| | |||||||| | |||| |
|||||||||||| | |||||||| | |||| |
|||||||||| | |||||||||| | |||||||||| |
|||||| | |||||||||||||| | |||||||||||||| |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | ||
|||||||||| | |||||||| | |||||||||||||| |
|||||||||||| | |||||||| | |||||||||||||| |
|||||||||| | |||||||||| | |||||||||||||| |
|||||| | |||||||||||||| | |||||||||||||| |
ASCVD = atherosclerotic cardiovascular disease; CFB = change from baseline; CrI = credible interval; LDL-C = low-density lipoprotein cholesterol; NMA = network meta-analysis; NR = not reported; SA = sensitivity analysis.
Source: Sponsor-submitted NMA.20
Critical Appraisal of the ITC
The NMA was based on an adequately conducted systematic literature search that included planned searches of multiple databases, conference proceedings, and clinical trial registries as well as regulatory and health technology assessment agency websites. Screening was conducted based on standard methods, with studies selected independently in duplicate according to pre-specified criteria. Although planned, no quality assessment of the included studies was reported in the sponsor’s NMA; however, a full quality assessment was included in the systematic literature review.
The population, interventions, and outcomes of the sponsor-submitted systematic review were relevant to Canadian clinical practice. The eligible studies included adults with HeFH or nFH (ASCVD or an ASCVD risk equivalent) whose LDL-C is inadequately controlled by MTD statin or who are statin-intolerant. The interventions included in the review were broader than the NMA and included icosapent ethyl, which is not publicly funded, and bempedoic acid, which is not available in Canada at the time of the report; however, no studies were included in the NMA that evaluated these treatments. Dosing regimens of included interventions reflected clinical practice. The outcomes included in the systematic review were also broader than those selected for the NMA. Outcomes were relevant and appropriate to the treatment for HeFH and ASCVD; however, outcomes important to patients and of critical importance to this review, including reduction in MACEs or other CV events, were not considered or included as part of the PICOS (population, intervention, comparison, outcomes, and study) criteria for the NMA.
A feasibility assessment was conducted to determine the ability to conduct an NMA based on network connectivity and differences in the study, patient, and outcome characteristics that were provided in the accompanying systematic literature review, and the reason for the exclusion of studies was provided in the NMA report. It was noted that visual inspection of study heterogeneity was conducted for treatment-effect modifiers, including background statin use, definition of CV risk and severity, and time points for assessment. As noted by the clinical expert consulted by CADTH, the treatment-effect modifiers of age, sex, and baseline LDL-C levels are the main drivers of differences in this population; however, they were not included as treatment-effect modifiers in the NMA. There were several limitations with the key assumptions made in the feasibility assessment with regard to background statin use and the time of assessment of outcomes, impacting clinical and methodological heterogeneity.
The NMA aimed to include only studies with patients who were receiving MTD statins as background therapy (with or without ezetimibe); however, a definition of MTD from each included study was not provided, and only the proportions of patients treated with low-, moderate-, or high-intensity statins were provided in the systematic literature review, which may result in unknown heterogeneity across populations. Moreover, the ORION trials (−9, −10, and −11) included a small proportion of patients who were intolerant to statins (25%, 22%, and 11%, respectively); however, the proportion of statin-intolerant patients in other trials was not noted; thus, heterogeneity may exist. To note, the results of sensitivity analyses for network scenario 2 excluded patients from ORION-10 and ORION-11 and were consistent with the base-case results; therefore, it is unlikely that these patients had a significant impact on the results. It was also assumed that individual moderate- and high-intensity statins (atorvastatin, rosuvastatin, and simvastatin) had similar efficacy as background therapy, regardless of dose, and would not bias the results of the NMA. It was considered reasonable to assume that background statin therapy that followed treatment guidelines was used in all clinical trials and would be well balanced, and that differences in treatment effect would likely be minimal; however, based on discussions with the clinical expert consulted by CADTH, it is unclear what effect different dosages of moderate- or high-intensity statins might have. Moreover, it is worth noting that inclisiran would be given only following treatment with an MTD statin, but it was unclear if this was the case in the included studies. It was also assumed that differences in CV risk and severity would not impact the relative effects on LDL-C and, therefore, no attempt was made to adjust for differences in baseline characteristics due to the number of studies and inconsistent reporting of characteristics.
The ORION trials had the longest follow-up of all included trials, and the duration of follow-up varied significantly across trials, from 12 weeks to 18 months (approximately 77 weeks), resulting in heterogeneity across the included studies. The NMA used 24 weeks as the time of assessment, which may underestimate and bias the results of trials with a longer duration. While 24 weeks is likely appropriate to assess lipid-related outcomes, including LDL-C and HDL-C changes, it may not be sufficient to assess safety outcomes. Given the variation in trial follow-up and duration, the authors considered end-of-study values for safety outcomes comparable if the duration of follow-up was 24 weeks or longer. As a result of the longer duration of the ORION trials, it is likely that more total discontinuations and discontinuations due to AEs were recorded purely based on trial length. This assumption is also likely to result in variation of events, favouring trials with shorter duration and differences in dosing regimen. Given the twice-yearly dosing regimen of inclisiran, a 24-week time of assessment may be insufficient to assess safety outcomes compared with the every-2-weeks dosing regimen of alirocumab and evolocumab, hence why the end-of-study time point was used for comparisons involving inclisiran.
Both fixed- and random-effects models were conducted. Random effects were considered most appropriate, given the number of studies per node and the heterogeneity observed. Given the small number of included studies, fixed-effects NMAs were conducted only for safety outcomes in the population of patients with ASCVD or an ASCVD risk equivalent who were intolerant to a statin. Model fit using a deviance information criterion was assessed; however, no results on model convergence and fit were reported. Data from the ORION-10 and ORION-11 studies were pooled for the ASCVD analyses, which was acceptable, given the observable similarities in the included populations; however, the method of pooling was not specified. It was noted that statistical heterogeneity for each pairwise comparison was high, with varying significant and non-significant P values for Q, and I2 values ranging from 0% to more than 80%. The authors also assessed global statistical heterogeneity through tau, which considered heterogeneity as moderate. The Cochrane handbook for the systematic review of interventions indicates that an I2 value of 75% or higher represents considerable heterogeneity, with the caveat that it is dependent on the magnitude and direction of effects and strength of evidence for heterogeneity.49 Overall, the studies included in the NMA were believed to be statistically heterogeneous based on the considerable range of I2 values; however, it was considered uncertain due to the small number of studies included in the analysis. Moreover, it is unclear what was the source of heterogeneity, as it was not explored. The authors relied on visual inspection of heterogeneity based on the statistical tests and concluded that the observed heterogeneity is likely due to observed and unobserved differences in patient populations across the included studies, data imputation analysis methods, and the specific background treatments allowed and/or delivered. Unidentified or unknown clinical (particularly treatment-effect modifiers) or methodological heterogeneity need to be explored through additional subgroup analyses or meta-regression, as it is unclear if the transitivity assumption was appropriately met.
In the HeFH network, 1 trial was noted to be based on a subgroup of patients with HeFH (ODYSSEY Long-term), resulting in broken randomization for the comparison of this study with others, which may bias the results of the alirocumab and placebo comparison. For all outcomes except total discontinuations at 24 weeks or greater, all treatments were generally favoured over placebo, yet there was no difference between PCKS9 inhibitors. Additionally, results for all outcomes except total discontinuations at 24 weeks or greater displayed exceedingly wide CrIs, leading to imprecise estimates of treatment effect. In the ASCVD and ASCVD risk–equivalent population on MTD statins, randomization was preserved across studies, and a closed loop was formed, allowing for both direct and indirect comparisons, which demonstrated no inconsistency. With the exception of safety outcomes, CrIs were wide, resulting in uncertainty across results. In the ASCVD and ASCVD risk–equivalent population intolerant to statins, subgroup data were used for the ORION trials, which resulted in broken randomization from the ORION trials. For all outcomes, mostly all treatments were favoured over placebo; however, the CrI for each outcome except total discontinuations at 24 weeks or greater were exceedingly wide, resulting in imprecision of treatment effects. It is unclear what caused the wide CrIs for outcomes in the NMAs; however, it is believed to be due to study heterogeneity, and low sample sizes for certain outcomes. Sensitivity analyses were appropriately conducted to evaluate differences between the ORION trials and comparator PSCK9 inhibitor trials, as well as the impact of excluding outlier comparator trials. The results of the sensitivity analyses were consistent with the base-case analyses.
In general, the 3 network scenarios made several clinical and methodological assumptions concerning the included populations, which limit the ability to interpret the generalizability of the results. Key assumptions included equivalence of background statins, regardless of individual statin or dose, and that background ezetimibe is not an effect modifier. Also, there was no adjustment of baseline characteristics for CV risk and severity, and the time of assessment for efficacy and safety outcomes was limited, given the variation of study durations. Though not reported or accounted for, these assumptions likely impacted treatment effects and the results of each NMA. Despite the limitations, there was no difference reported between inclisiran and other PCSK9 inhibitors in the efficacy and safety outcomes evaluated, and the results of most outcomes in all network scenarios displayed exceedingly wide CrIs, further challenging the precision of the results.
Other Relevant Evidence
This section includes submitted long-term extension studies and additional relevant studies included in the sponsor’s submission to CADTH that were considered to address important gaps in the evidence included in the systematic review.
Two additional relevant studies (ORION-4 and ORION-8) were noted in the sponsor submission and identified in the CADTH screening of clinical trial databases. At the time of this review, results were not available for either study. As such, ORION-4 and ORION-8 were not included in the available evidence discussed earlier. The 2 studies are described briefly in this section to provide complete information. Results of these trials are expected to provide further evidence to better characterize the efficacy profile of inclisiran in pertinent clinical outcomes, as well as provide long-term efficacy and safety data for inclisiran.
ORION-4 is a double-blind randomized, placebo-controlled trial investigating the effects of inclisiran on clinical outcomes in patients with ASCVD. The trial is to enrol approximately 15,000 patients in the UK and US who are 55 years or older with a history or evidence of prior MI, ischemic stroke, or PAD. Patients will be randomized in a 1:1 ratio to inclisiran sodium 300 mg or matching placebo, administered as a subcutaneous injection on the day of randomization, at 3 months, and then every 6 months thereafter. The primary end point of ORION-4 is the number of patients with a MACE, defined as time-to-first occurrence of CHD, MI, fatal or non-fatal ischemic stroke, or urgent coronary revascularization procedure. Secondary outcomes included the number of patients with a composite of CHD death or MI, and the number of patients with CV death. The planned median follow-up is 5 years, with an estimated primary completion date (i.e., final data collection date for the primary outcome measure) of July 2026, according to clinicaltrials.gov.50
ORION-8 is an open-label long-term extension study of patients previously enrolled in 1 of 4 phase III trials: ORION-5, ORION-9, ORION-10, or ORION-11. The purpose of this study is to evaluate the long-term efficacy, safety, and tolerability of inclisiran in patients with ASCVD, an ASCVD risk equivalent, HeFH, or HoFH, who still had elevated LDL-C despite maximally tolerated LDL-C-lowering therapies. ||||||||||||||||||||||||||||||||||||||||||||||||||||. Included patients are adults 18 years and older who had received the last dose of the study drug; completed the final ORION-5, −9, −10, or −11 study visit; and who are currently on the lipid-lowering therapies (e.g., statin and/or ezetimibe) from the previous study with no planned medication or dose change during ORION-8 participation. In this extension study, patients receive inclisiran 300 mg subcutaneous injections on day 1 (except for ORION-5 patients) and day 90, then every 180 days to day 990. On day 1 of the trial, patients who received blinded placebo in the previous feeder study are administered blinded inclisiran, whereas those who previously received blinded placebo are administered blinded inclisiran. The primary end point of ORION-8 is the proportion of patients reaching on-treatment target LDL-C level of less than 70 mg/dL (< 1.8 mmol/L) or less than 100 mg/dL (< 2.6 mmol/L), the global lipid target for their level of ASCVD risk. Secondary end points include the effect of inclisiran on LDL-C, total cholesterol, triglycerides, and HDL-C levels from baseline. The planned time frame of evaluation is 1,080 days, with an estimated primary completion date of August 2023, according to clinicaltrials.gov.51
Discussion
Summary of Available Evidence
Three double-blind RCTs (ORION-9, ORION-10, and ORION-11) were included in this review. All studies were 540 days (18 months) in duration. ORION-9 included adult patients with established HeFH or ASCVD, ORION-10 included adult patients with ASCVD, and ORION-11 included patients with ASCVD and ASCVD risk equivalents. A total of 482, 1,561, and 1,617 patients were enrolled in ORION-9, ORION-10, and ORION-11, respectively. In all 3 ORION trials, patients were randomized to 284 mg inclisiran or matching placebo. The co-primary end points of the ORION trials were the percentage change in LDL-C from baseline to day 510, and the time-adjusted percentage change in LDL-C from baseline after day 90 and up to day 540. Key secondary outcomes included the absolute change in LDL-C from baseline to day 510, the time-adjusted percentage change from baseline after day 90 and up to day 540, and the percentage change from baseline to day 510 in PCSK9, total cholesterol, ApoB, and non-HDL-C.
Baseline characteristics of the ORION trials were balanced between groups, and generally applicable to the Canadian population. The ORION-9 trial enrolled mostly White patients (94.0%) with a median age of 56 years and a relatively even ratio of males and females (47.1% male, 52.9% female) with either ASCVD (27.4%) or an ASCVD risk equivalent (72.6%), of which 93.2% were HeFH. A total of 73.9% of patients were on high-intensity statins at baseline, with 25.3% either partially or completely intolerant to statins, and 52.3% were treated with ezetimibe. The ORION-10 trial enrolled mostly White (85.7%) males (69.4%) with a median age of 67 years, all with ASCVD (91.1% with CHD). Approximately 2-thirds (69.4%) of patients were on a high-intensity statin at baseline, with 22.0% partially or completely intolerant. A total of 9.9% of patients were treated with ezetimibe. ORION-11 enrolled patients with ASCVD (87.4%) or an ASCVD risk equivalent (12.6%). Patients were mostly White (98.1%) males (71.7%) with a median age of 65 years. A total of 78% of patients were receiving high-intensity statins, while 11.4% were considered partially or completely intolerant, and 7.1% of patients were treated with ezetimibe.
Interpretation of Results
Efficacy
All 3 ORION trials shared the same key primary, secondary, and exploratory outcomes. Clinically important outcomes relevant to this review of CV-related morbidity (i.e., MACEs) and mortality were secondary outcomes of the ORION trials, with no between-group comparisons conducted. The reported incidence of MACEs and death outcomes in the ORION trials were minimal and there were no apparent differences between groups; however, it remains uncertain whether the reductions in percentage and absolute change in LDL-C observed with inclisiran will translate into a reduction in clinically significant CV morbidity. According to guidelines, a reduction of 1 mmol/L (approximately 38.67 mg/dL) in LDL-C is estimated to reduce the relative risk of ASCVD by 20% to 22%.31 Between-group differences in the ORION-9, −10, and −11 trials of −68.89 mg/dL, −54.12 mg/dL, and −51.87 mg/dL indicate that long-term inclisiran use may result in a reduction in CV-related events. The currently ongoing ORION-4 trial will aim to address this important gap; however, results are not available yet. Further details on the ORION-4 trial are outlined in the Other Relevant Evidence section.
In all cases, inclisiran was superior to placebo for the co-primary outcome of percentage change in LDL-C from baseline to day 510, and time-adjusted percentage change in LDL-C from baseline after day 90 up to day 540. In managing FH, according to CCS guidelines, further treatment should be implemented in patients who have not achieved a 50% reduction in LDL-C levels on MTD statin.11 The observed and washout-imputed percentage change in LDL-C with inclisiran was −41.15% and −39.67% in ORION-9, −56.34% and −51.28% in ORION-10, and −49.3% and −45.8% in ORION-11, which were considered clinically meaningful reductions in LDL-C, per the clinical expert consulted by CADTH, considering the patient populations and use of background statins. In both cases, inclisiran was associated with a statistically significant difference compared with placebo. Given that the observed values for percentage change in LDL-C from baseline were higher than washout-imputed values, there is a risk of bias in these results; however, the results were similar when accounting for missing values. Moreover, the co-primary end point of time-adjusted percentage change in LDL-C from baseline after day 90 and up to day 540 was consistent with the percentage change in LDL-C from baseline to day 510.
In patients with HeFH, baseline LDL-C levels are generally higher than in those without FH, so percentage change from baseline alone is not sufficient to determine clinical efficacy and must be supplemented with the absolute change in LDL-C from baseline. As mentioned earlier, inclisiran was associated with statistically significantly higher absolute changes from baseline in the ORION trials compared with placebo. The mean difference compared with placebo was higher in the ORION-9 trial than in the ORION-10 and −11 trials, as these were mostly patients with HeFH with higher baseline levels of LDL-C.
Along with LDL-C, measuring ApoB and non-HDL-C levels is in line with current guidance.11 Results for ApoB and non-HDL-C outcomes were similar to the results for LDL-C, with inclisiran resulting in greater changes from baseline than placebo in all trials; however, the change in LDL-C was more pronounced than change in non-HDL-C or ApoB. The clinical expert consulted by CADTH indicated that clinicians must determine which biomarker to select for clinical decision-making.
In addition to percentage and absolute reductions in LDL-C, reaching global lipid targets are an important aspect of treating patients with HeFH and ASCVD. In patients with HeFH (or with an ASCVD risk equivalent in ORION-9 and ORION-11), 66.9% to 77.6% of inclisiran-treated patients achieved a target LDL-C of less than 100 mg/dL (2.6 mmol/L) compared with 8.9% to 30.5% of placebo-treated patients in ORION-9 and −11 at day 510. Conversely, 52.5% to 84.1% of inclisiran-treated patients with ASCVD reached a target LDL-C of less than 70 mg/dL (1.8 mmol/L) compared with 1.4% to 17.9% of placebo-treated patients in ORION-9, −10, and −11; however, it is worth noting that 5% to 15% of patients were missing from the analyses. The results from the ASCVD risk–equivalent population in ORION-11 should be interpreted with caution, given that the population was not entirely of interest, as only 11 patients had HeFH. Interestingly, 18.4% to 41.9% of placebo-treated patients were able to achieve target LDL-C levels at any visit compared with 77.2% to 94.1% of inclisiran-treated patients. Although greater than 50% more patients achieved target LDL-C levels with inclisiran compared with placebo, it is unclear why such a high proportion of placebo-treated patients were able to achieve target LDL-C levels; however, it may be related to background statin and/or ezetimibe treatment.
Although not an outcome of the study but an outcome important to patients, the twice-yearly dosing regimen provides a more manageable dosing and administration schedule compared with the PCSK9 inhibitors available; alirocumab and evolocumab, which require injection 26 times per year. The clinical expert consulted by CADTH also noted that this dosing regimen aligns with routine patient follow-up in these populations, which would also improve adherence to treatment, as evidenced by the high proportion of patients completing the 18-month studies (89.0% to 97.1%), and |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
The sponsor-submitted ITC compared the efficacy and safety of inclisiran and relevant drug comparators (evolocumab and alirocumab) for the treatment of adult patients with HeFH or ASCVD with high LDL-C uncontrolled by MTD statins with or without ezetimibe, or those who are statin-intolerant or contraindicated, with or without ezetimibe, with uncontrolled LDL-C. The results of the ITC suggest there was no difference in efficacy between inclisiran and alirocumab or evolocumab. The considerable clinical, methodological, and statistical heterogeneity, coupled with the wide CrIs in each network scenario for comparisons between inclisiran and placebo, PCSK9 inhibitors, and ezetimibe, resulted in significant uncertainty in the comparative efficacy of inclisiran. In addition, important outcomes of all-cause, CV-related mortality, and CV-related morbidity (MACE) outcomes were not included in the analysis.
Harms
The incidence of harms reported in the ORION trials was well balanced between the inclisiran and placebo groups, with an occurrence of TEAEs ranging from 71.7% to 82.7%. No important or consistent differences in SAEs, WDAEs, or most notable harms were evident between the placebo and inclisiran groups across trials, except for harms related to administration (such as injection-site reactions), which were higher in the inclisiran arm in ORION-9, −10, and −11 (17.0%, 6.0%, and 7.6%, respectively) compared with placebo (1.7%, 1.9%, and 1.7%, respectively). The clinical expert involved in this review did not identify any specific safety concerns with inclisiran; however, despite the 18-month trial duration, there is still limited evidence regarding the long-term safety of inclisiran. The currently ongoing ORION-8 study aims to address this gap and provide additional long-term safety data for inclisiran use over 3 years.
The safety of inclisiran compared with other relevant treatments for HeFH and/or ASCVD could not be assessed based on the use of placebo as a comparator in the ORION studies; therefore, the sponsor-submitted ITC attempted to determine the safety of inclisiran and other PCSK9 inhibitors through the outcomes of total discontinuations and discontinuations due to AEs. The time of assessment for efficacy and safety outcomes in the NMA was 24 weeks, which was deemed sufficient for lipid and lipoprotein assessments for currently available PCSK9 inhibitors. It was assumed that end-of-study safety outcomes were considered comparable despite the large variation in study duration across trials, which may bias the results in favour of trials with shorter duration, as the frequency of events may change over time, depending on dosing, administration, and adherence differences between inclisiran and other treatments.
In general, there was no difference between inclisiran and ezetimibe, alirocumab, or evolocumab across network scenarios for safety outcomes. Comparative safety results for inclisiran were generally associated with wide CrIs, particularly for discontinuations due to AEs at 24 weeks or greater, which is potentially a result of smaller populations or event counts in these groups. However, given the limitations described for the efficacy analyses, particularly the follow-up time assumptions, the results for comparative harms may not be valid.
Conclusions
In all 3 ORION studies, inclisiran demonstrated a statistically significant and clinically meaningful change compared with placebo in the co-primary end points of change in LDL-C from baseline at day 510, and time-adjusted change from baseline after day 90 up to day 540 in patients with HeFH, ASCVD, and ASCVD or an ASCVD risk equivalent, respectively. Results of the key secondary end points were in line with the primary outcome. Together, the lipid and lipoprotein efficacy outcomes were appropriate, and are reflective of clinical practice in Canada. Combined, the percentage change and absolute change in LDL-C from baseline, as well as the ApoB and non-HDL-C measurements, are guideline-specific and relevant in diagnosing and treating HeFH and ASCVD. None of the ORION studies assessed HRQoL; hence, the effect of inclisiran on HRQoL is uncertain.
Overall, treatment with inclisiran was well tolerated over the study period. Inclisiran did not appear to be associated with more AEs, or SAEs compared with placebo. Known AEs of interest, such as injection-site reactions, were more frequent in the inclisiran group; however, there were no concerns. Although the sponsor submitted an ITC evaluating inclisiran and appropriate comparators in the treatment of HeFH and ASCVD, the results of the indirect evidence were inconclusive on the comparative efficacy and safety of inclisiran and relevant treatments for the outcomes evaluated. In addition, the sponsor-submitted ITC provides minimal value to assess comparative efficacy with the other available PCSK9 inhibitor or ezetimibe, as it did not evaluate clinical outcomes.
ORION-9, −10, and −11 trials were not designed to compare between treatment groups for outcomes of critical importance to the review: mortality (all-cause and CV-related) and CV-related morbidity (resuscitated cardiac arrest, non-fatal MI and stroke, and MACE); however, there was no apparent difference between inclisiran and placebo. The short follow-up duration of the ORION trials (18 months) did not allow for adequate evaluation of these outcomes. Longer follow-up obtained from the ORION-8 trial, combined with results from the ORION-4 trial, will provide important insight into the efficacy of inclisiran in reducing CV-related morbidity and mortality.
References
1.Virani SS, Alonso A, Benjamin EJ, et al. Heart disease and stroke statistics-2020 update: a report from the American Heart Association. Circulation. 2020;141(9):e139-e596. PubMed
2.Roth GA, Johnson C, Abajobir A, et al. Global, regional, and national burden of cardiovascular diseases for 10 causes, 1990 to 2015. J Am Coll Cardiol. 2017;70(1):1-25. PubMed
3.World Health Organization. Cardiovascular diseases (CVDs). 2021; https://www.who.int/en/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds). Accessed 2021 Aug 4.
4.American Heart Association. What is cardiovascular disease? 2017; https://www.heart.org/en/health-topics/consumer-healthcare/what-is-cardiovascular-disease. Accessed 2021 Aug 4.
5.Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019;139(25):e1082-e1143. PubMed
6.Nordestgaard BG, Chapman MJ, Humphries SE, et al. Familial hypercholesterolaemia is underdiagnosed and undertreated in the general population: guidance for clinicians to prevent coronary heart disease: consensus statement of the European Atherosclerosis Society. Eur Heart J. 2013;34(45):3478-3490a. PubMed
7.Sniderman AD, Tsimikas S, Fazio S. The severe hypercholesterolemia phenotype: clinical diagnosis, management, and emerging therapies. J Am Coll Cardiol. 2014;63(19):1935-1947. PubMed
8.Brunham LR, Ruel I, Aljenedil S, et al. Canadian Cardiovascular Society position statement on familial hypercholesterolemia: update 2018. Can J Cardiol. 2018;34(12):1553-1563. PubMed
9.Marbach JA, McKeon JL, Ross JL, Duffy D. Novel treatments for familial hypercholesterolemia: pharmacogenetics at work. Pharmacotherapy. 2014;34(9):961-972. PubMed
10.Atherosclerotic Cardiovascular Disease (ASCVD) secondary prevention guideline. Washington (DC): Kaiser Permanente; 2020: https://wa.kaiserpermanente.org/static/pdf/public/guidelines/ascvd-secondary.pdf. Accessed 2021 Aug 4.
11.Pearson GJ, Thanassoulis G, Anderson TJ, et al. 2021 Canadian Cardiovascular Society guidelines for the management of dyslipidemia for the prevention of cardiovascular disease in the adult. Can J Cardiol. 2021. PubMed
12.Anderson TJ, Grégoire J, Pearson GJ, et al. 2016 Canadian Cardiovascular Society guidelines for the management of dyslipidemia for the prevention of cardiovascular disease in the adult. Can J Cardiol. 2016;32(11):1263-1282. PubMed
13.Watts GF, Gidding S, Wierzbicki AS, et al. Integrated guidance on the care of familial hypercholesterolemia from the International FH Foundation. J Clin Lipidol. 2014;8(2):148-172. PubMed
14.Lambert CT, Sandesara P, Isiadinso I, et al. Current treatment of familial hypercholesterolaemia. Eur Cardiol. 2014;9(2):76-81. PubMed
15.Alonso R, Perez de Isla L, Muñiz-Grijalvo O, Diaz-Diaz JL, Mata P. Familial hypercholesterolaemia diagnosis and management. Eur Cardiol. 2018;13(1):14-20. PubMed
16.Leqvio (inclisiran): 284 mg in 1.5 mL (189 mg/mL) solution for subcutaneous injection [product monograph]. Dorval (QC): Novartis Pharmaceuticals Canada Inc; 2021 Jul 23.
17.Clinical Study Report: MDCO-PCS-17-03. A placebo-controlled, double-blind, randomized trial to evaluate the effect of 300 mg of inclisiran sodium given as subcutaneous injections in subjects with heterozygous familial hypercholesterolemia (HEFH) and elevated low density lipoprotein cholesterol (LDL-C) [internal sponsor's report]. Parsippany (NJ): The Medicines Company; 2019 Dec 2.
18.Clinical Study Report: MDCO-PCS-17-04. A placebo-controlled, double-blind, randomized trial to evaluate the effect of 300 mg of inclisiran sodium given as subcutaneous injections in subjects with atherosclerotic cardiovascular disease (ASCVD) and elevated low density lipoprotein cholesterol (LDL-C) [internal sponsor's report]. Parsippany (NJ): The Medicines Company; 2019 Dec 5.
19.Clinical Study Report: MDCO-PCS-17-08. A placebo-controlled, double-blind, randomized trial to evaluate the effect of 300 mg of inclisiran sodium given as subcutaneous injections in subjects with atherosclerotic cardiovascular disease (ASCVD) or ASCVD-risk equivalents and elevated low-density lipoprotein cholesterol (LDL-C) [internal sponsor's report]. Parsippany (NJ): The Medicines Company; 2019 Nov 20.
20.Network Meta-analysis to support global submission for Inclisiran [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Leqvio (inclisiran), 284 mg in 1.5 mL (189 mg/mL) solution for subcutaneous injection. Dorval (QC): Novartis Pharmaceuticals Canada Inc; 2021.
21.Berberich AJ, Hegele RA. The complex molecular genetics of familial hypercholesterolaemia. Nat Rev Cardiol. 2019;16(1):9-20. PubMed
22.Ibrahim MA, Asuka E, Jialal I. Hypercholesterolemia [updated 2021 Apr 3]. StatPearls. StatPearls Publishing: Treasure Island (FL); 2020: https://www.ncbi.nlm.nih.gov/books/NBK459188/. Accessed 2021 Aug 4.
23.Obradovic M, Zaric B, Sudar-Milovanovic E, et al. PCSK9 and hypercholesterolemia: therapeutic approach. Curr Drug Targets. 2018;19(9):1058-1067. PubMed
24.Centers for Disease Control and Prevention. Familial hypercholesterolemia. 2020; https://www.cdc.gov/genomics/disease/fh/FH.htm#:~:text=How%20do%20I%20know%20if,heart%20disease%20or%20heart%20attacks. Accessed 2021 Aug 4.
25.Hu P, Dharmayat KI, Stevens CAT, et al. Prevalence of familial hypercholesterolemia among the general population and patients with atherosclerotic cardiovascular disease: a systematic review and meta-analysis. Circulation. 2020:1742-1759. PubMed
26.Beheshti SO, Madsen CM, Varbo A, Nordestgaard BG. Worldwide prevalence of familial hypercholesterolemia: meta-analyses of 11 million subjects. J Am Coll Cardiol. 2020;75(20):2553-2566. PubMed
27.Brunham LR, Ruel I, Khoury E, et al. Familial hypercholesterolemia in Canada: initial results from the FH Canada national registry. Atherosclerosis. 2018;277:419-424. PubMed
28.Turgeon RD, Barry AR, Pearson GJ. Familial hypercholesterolemia. Can Fam Physician. 2016;62(1):32. PubMed
29.Ference BA, Ginsberg HN, Graham I, et al. Low-density lipoproteins cause atherosclerotic cardiovascular disease. 1. Evidence from genetic, epidemiologic, and clinical studies. A consensus statement from the European Atherosclerosis Society Consensus Panel. Eur Heart J. 2017;38(32):2459-2472. PubMed
30.Gidding SS, Champagne MA, de Ferranti SD, et al. The agenda for familial hypercholesterolemia: a scientific statement from the American Heart Association. Circulation. 2015;132(22):2167-2192. PubMed
31.Silverman MG, Ference BA, Im K, et al. Association between lowering LDL-C and cardiovascular risk reduction among different therapeutic interventions: a systematic review and meta-analysis. JAMA. 2016;316(12):1289-1297. PubMed
32.Genest J, Hegele RA, Bergeron J, et al. Canadian Cardiovascular Society position statement on familial hypercholesterolemia. Can J Cardiol. 2014;30(12):1471-1481. PubMed
33.Talmud PJ, Shah S, Whittall R, et al. Use of low-density lipoprotein cholesterol gene score to distinguish patients with polygenic and monogenic familial hypercholesterolaemia: a case-control study. Lancet. 2013;381(9874):1293-1301. PubMed
34.Ruel I, Brisson D, Aljenedil S, et al. Simplified Canadian definition for familial hypercholesterolemia. Can J Cardiol. 2018;34(9):1210-1214. PubMed
35.Raal FJ, Kallend D, Ray KK, et al. Inclisiran for the treatment of heterozygous familial hypercholesterolemia. N Engl J Med. 2020;382(16):1520-1530. PubMed
36.Rosenson RS, Durrington P. Familial hypercholesterolemia in adults: treatment. In: Freeman MW, ed. UpToDate. Waltham (MA): UpToDate; 2020: https://www.uptodate.com. Accessed 2021 Apr 6.
37.Neefjes LA, Ten Kate GJ, Rossi A, et al. CT coronary plaque burden in asymptomatic patients with familial hypercholesterolaemia. Heart. 2011;97(14):1151-1157. PubMed
38.Novartis. Novartis receives complete response letter from U.S. FDA for inclisiran. 2020; https://www.novartis.com/news/media-releases/novartis-receives-complete-response-letter-from-us-fda-inclisiran. Accessed 2021 Aug 4.
39.Drug Reimbursement Review sponsor submission: Leqvio (inclisiran), 284 mg in 1.5 mL (189 mg/mL) solution for subcutaneous injection [internal sponsor's package]. Dorval (QC): Novartis Pharmaceuticals Canada Inc; 2021 Mar 17.
40.CADTH common drug review: clinical review report (resubmission) of evolocumab (Repatha- Amgen Canada Inc). Ottawa (ON): CADTH; 2017: https://cadth.ca/sites/default/files/cdr/clinical/SR0515_Repatha_Resubmission_CL_Report.pdf. Accessed 2021 Aug 4.
41.McGowan J, Sampson M, Salzwedel DM, Cogo E, Foerster V, Lefebvre C. PRESS Peer Review of Electronic Search Strategies: 2015 guideline statement. J Clin Epidemiol. 2016;75:40-46. PubMed
42.Grey matters: a practical tool for searching health-related grey literature. Ottawa (ON): CADTH; 2019: https://www.cadth.ca/grey-matters. Accessed 2020 Apr 1.
43.Ray KK, Wright RS, Kallend D, et al. Two phase 3 trials of inclisiran in patients with elevated LDL cholesterol. N Engl J Med. 2020;382(16):1507-1519. PubMed
44.Novartis Pharmaceuticals Canada Inc. response to May 10, 2021 DRR request for additional information regarding Leqvio (inclisiran) DRR review [internal additional sponsor's information]. Dorval (QC): Novartis Pharmaceuticals Canada Inc; 2021.
45.Rubin D. Multiple imputation for nonresponse in surveys. Toronto (ON): John Wiley & Sons; 1987: https://onlinelibrary.wiley.com/doi/pdf/10.1002/9780470316696.fmatter. Accessed 2021 Aug 4.
46.Hochberg Y. A sharper Bonferroni procedure for multiple tests of significance. Biometrika. 1988;75(4):800-802.
47.Cholesterol Treatment Trialists’ (CTT) Collaboration, Baigent C, Blackwell L, et al. Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170,000 participants in 26 randomised trials. Lancet. 2010;376(9753):1670-1681. PubMed
48.Dias S, Welton NJ, Sutton AJ, Caldwell DM, Lu G, Ades AE. NICE DSU technical support document 4: inconsistency in networks of evidence based on randomised controlled trials. NICE Decision Support Unit technical support documents. London (GB): National Institute for Health and Care Excellence (NICE); 2014.
49.Cochrane handbook for systematic reviews of interventions. In: Higgins JPT, Thomas J, Chandler J, et al., eds. London (GB): Cochrane; 2021: https://training.cochrane.org/handbook. Accessed 2021 Aug 4.
50.University of Oxford. NCT03705234: A randomized trial assessing the effects of inclisiran on clinical outcomes among people with cardiovascular disease (ORION-4). ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2021: https://clinicaltrials.gov/ct2/show/record/NCT03705234. Accessed 2021 May 13.
51.Novartis. NCT03814187: Trial to assess the effect of long term dosing of inclisiran in subjects with high CV risk and elevated LDL-C (ORION-8). ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2020: https://clinicaltrials.gov/ct2/show/record/NCT03814187?view=record. Accessed 2021 May 13.
Appendix 1: Literature Search Strategy
Note that this appendix has not been copy-edited.
Clinical Literature Search
Overview
Interface: Ovid
Databases:
Embase 1974 to present
MEDLINE Daily and MEDLINE 1946 to present
MEDLINE In-Process & Other Non-Indexed Citations
Note: Patient headings have been customized for each database. Duplicates between databases were removed in Ovid.
Date of Search: April 15, 2021
Alerts: Biweekly search updates until project completion.
Study Types: No filters were applied to limit the retrieval by study type.
Limits:
No date or language limits were used
Conference abstracts were excluded
Table 26: Clinical Literature Search
Syntax guide | Detail |
|---|---|
/ | At the end of a phrase, searches the phrase as a subject heading |
MeSH | Medical Subject Heading |
exp | Explode a subject heading |
* | Before a word, indicates that the marked subject heading is a primary topic; or, after a word, a truncation symbol (wildcard) to retrieve plurals or varying endings |
.ti | Title |
.ot | Original title |
.ab | Abstract |
.hw | Heading word; usually includes subject headings and controlled vocabulary |
.kf | Author keyword heading word (MEDLINE) |
.kw | Author keyword (Embase) |
.dq | Candidate term word (Embase) |
.rn | Registry number |
.nm | Name of substance word (MEDLINE) |
medall | Ovid database code: MEDLINE All, 1946 to present, updated daily |
oemezd | Ovid database code; Embase, 1974 to present, updated daily |
Table 27: Multi-Database Strategy
# | Searches | Results |
|---|---|---|
1 | (inclisiran* or Leqvio* or ALN PCS* or ALNPCS* or aln 60212 or aln60212 or UOW2C71PG5).ti,ab,kf,ot,hw,rn,nm. | 399 |
2 | 1 use medall | 118 |
3 | *inclisiran/ | 88 |
4 | (inclisiran* or Leqvio* or ALN PCS* or ALNPCS* or aln 60212 or aln60212).ti,ab,kw,dq. | 293 |
5 | 3 or 4 | 305 |
6 | 5 use oemezd | 207 |
7 | 6 not (conference review or conference abstract).pt. | 176 |
8 | 2 or 7 | 294 |
9 | remove duplicates from 8 | 187 |
Clinical Trials Registries
ClinicalTrials.gov
Produced by the US National Library of Medicine. Targeted search used to capture registered clinical trials.
[Search -- Studies with results | Leqvio OR inclisiran]
WHO ICTRP
International Clinical Trials Registry Platform, produced by the World Health Organization. Targeted search used to capture registered clinical trials.
[Search terms -- Leqvio OR inclisiran]
Health Canada’s Clinical Trials Database
Produced by Health Canada. Targeted search used to capture registered clinical trials.
[Search terms -- Leqvio OR inclisiran]
EU Clinical Trials Register
European Union Clinical Trials Register, produced by the European Union. Targeted search used to capture registered clinical trials.
[Search terms -- Leqvio OR inclisiran]
Grey Literature
Search dates: April 6 to April 9, 2021
Keywords: Leqvio, inclisiran, hypercholesterolemia
Limits: None
Updated: Search updated prior to the completion of stakeholder feedback period
Relevant websites from the following sections of the CADTH grey literature checklist Grey Matters: A Practical Tool for Searching Health-Related Grey Literature were searched:
Health Technology Assessment Agencies
Health Economics
Clinical Practice Guidelines
Drug and Device Regulatory Approvals
Advisories and Warnings
Drug Class Reviews
Clinical Trials Registries
Databases (free)
Health Statistics
Internet Search.
Appendix 2: Excluded Studies
Note that this appendix has not been copy-edited.
Reference | Reason for exclusion |
|---|---|
Landmesser U, Haghikia A, Leiter LA, et al. Effect of inclisiran, the small-interfering RNA against proprotein convertase subtilisin/kexin type 9, on platelets, immune cells, and immunological biomarkers: a pre-specified analysis from ORION-1. Cardiovasc Res. 2021 Jan 01;117(1):284-291. | Study design (not a phase III or phase IV RCT) |
Leiter LA, Teoh H, Kallend D, et al. Inclisiran Lowers LDL-C and PCSK9 Irrespective of Diabetes Status: The ORION-1 Randomized Clinical Trial. Diabetes Care. 2019 01;42(1):173-176. | Study design (not a phase III or phase IV RCT) |
Ray KK, Stoekenbroek RM, Kallend D, et al. Effect of 1 or 2 Doses of Inclisiran on Low-Density Lipoprotein Cholesterol Levels: One-Year Follow-up of the ORION-1 Randomized Clinical Trial. JAMA Cardiol. 2019 11 01;4(11):1067-1075. | Study design (not a phase III or phase IV RCT) |
Ray KK, Stoekenbroek RM, Kallend D, et al. Effect of an siRNA Therapeutic Targeting PCSK9 on Atherogenic Lipoproteins: Prespecified Secondary End Points in ORION 1. Circulation. 2018 09 25;138(13):1304-1316. | Study design (not a phase III or phase IV RCT) |
Ray KK, Landmesser U, Leiter LA, et al. Inclisiran in Patients at High Cardiovascular Risk with Elevated LDL Cholesterol. N Engl J Med. 2017 04 13;376(15):1430-1440. | Study design (not a phase III or phase IV RCT) |
LDL-C = low-density lipoprotein cholesterol; PCSK9 = proprotein convertase subtilisin/kexin type 9; RCT = randomized controlled trial; siRNA = small interfering ribonucleic acid.
Appendix 3: Detailed Outcome Data
Note that this appendix has not been copy-edited.
ORION-9 Sensitivity Analyses
Table 29: Percentage Change in LDL-C From Baseline to Day 510 Sensitivity Analyses — ORION-9 (ITT Population)
Sensitivity analysis | Inclisiran (N = 242) | Placebo (N = 240) | Mean difference from placebo |
|---|---|---|---|
Control-Based PMM,a LSM (95% CI) | −39.71 (−43.69 to −35.73) | 8.27 (4.32 to 12.23) | −47.98 (−53.59 to −42.38) P < 0.0001 |
MMRM,b LSM (95% CI) | −40.76 (−44.63 to −36.88) | 8.06 (4.16 to 11.96) | −48.82 (−54.32 to −43.32) P < 0.0001 |
ANCOVA from multiple imputation washout model, including country and current use of statin/LMT,c LSM (95% CI) | −40.05 (−47.40 to −32.69) | 7.82 (0.09 to 15.55) | −47.86 (−55.45 to −40.28) P < 0.0001 |
ANCOVA = analysis of covariance; CI = confidence interval; ITT = intention to treat; LDL-C = low-density lipoprotein cholesterol; LMT = lipid-modifying therapy; LSM = least squares mean; MAR = missing at random; MMRM = mixed-effects model for repeated measures; PMM = pattern-mixture model.
aA control-based PMM was used for missing data imputation with 100 total imputed datasets. An MMRM on each of the 100 datasets was performed by including fixed effects for treatment, visit, interaction between treatment and visit, and baseline LDL-C as a covariate.
bAn MMRM analysis that assumes missing data are MAR was performed.
cA multiple imputation washout model was used for missing data imputation with 100 total imputed datasets.
Analysis of covariance (ANCOVA) on each of the 100 datasets was performed by including fixed effects for treatment, current use of statins or other LMTs (y/n), country, interaction between treatment and country, and baseline LDL-C as a covariate.
Treatment effects from the 100 analyses were combined using the Ruben method.
Source: ORION-9 Clinical Study Report.17
Table 30: Time-Adjusted Percentage Change in LDL-C From Baseline After Day 90 and Up to Day 540 Sensitivity Analyses — ORION-9 (ITT Population)
Sensitivity analysis | Inclisiran (N = 242) | Placebo (N = 240) | Mean difference from placebo |
|---|---|---|---|
MMRM,a LSM (95% CI) | −38.49 (−41.40 to −35.59) | 6.27 (3.34 to 9.20) | −44.76 (−48.89 to −40.64) P < 0.0001 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||| | |||||||||||||||||||||||||||||||||||| | |||||||||||||||||||||||||||||||||||||||||||||||||||||||| | |||||||||||||||||||||||||||||||||||||||||||||||||||||||| |
Two-sample t-test,c LSM (95% CI) | −38.01 (−40.61 to −35.41) | 6.14 (2.86 to 9.43) | −44.15 (−48.34 to −39.96) P < 0.0001 |
CI = confidence interval; ITT = intention to treat; LDL-C = low-density lipoprotein cholesterol; LMT = lipid-modifying therapy; LSM = least squares mean; MAR = missing at random; MMRM = mixed-effects model for repeated measures; PMM = pattern-mixture model.
aAn MMRM analysis that assumes missing data are MAR was performed. The model included fixed effects for treatment, visit, interaction between treatment and visit, and baseline LDL-C as a covariate.
bA control-based PMM was used for missing data imputation with 100 total imputed datasets. An MMRM on each of the 100 datasets was performed by including fixed effects for treatment, visit, interaction between treatment and visit, current use of statins or other LMTs (y/n), country, interaction between treatment and country, and baseline LDL-C as a covariate.
cThe time-adjusted percentage change was calculated by taking the arithmetic mean of calculated percentage change in LDL-C from baseline at each visit after day 90 through day 540.
Source: ORION-9 Clinical Study Report.17
ORION-10 Sensitivity Analyses
Table 31: Percentage Change in LDL-C From Baseline to Day 510 Sensitivity Analyses — ORION-10 (ITT Population)
Sensitivity analysis | Inclisiran (N = 781) | Placebo (N = 780) | Mean difference from placebo |
|---|---|---|---|
Control-based PMM,a LSM (95% CI) | −53.45 (−55.77 to −51.12) | 1.01 (−1.32 to 3.35) | −54.46 (−57.77 to −51.15) P < 0.0001 |
MMRM,b LSM (95% CI) | −56.17 (−58.36 to −53.98) | 1.07 (−1.15 to 3.29) | −57.24 (−60.36 to −54.13) P < 0.0001 |
ANCOVA from multiple imputation washout model, including current use of statin/LMT,c LSM (95% CI) | −45.49 (−49.31 to −41.67) | 6.78 (2.99 to 10.56) | −52.27 (−55.66 to −48.87) P < 0.0001 |
ANCOVA = analysis of covariance; CI = confidence interval; ITT = intention to treat; LDL-C = low-density lipoprotein cholesterol; LMT = lipid-modifying therapy; LSM = least squares mean; MAR = missing at random; MMRM = mixed-effects model for repeated measures; PMM = pattern-mixture model.
aA control-based PMM was used for missing data imputation with 100 total imputed datasets. An MMRM on each of the 100 datasets was performed by including fixed effects for treatment, visit, interaction between treatment and visit, and baseline LDL-C as a covariate.
bAn MMRM analysis that assumes missing data are MAR was performed.
cA multiple imputation washout model was used for missing data imputation with 100 total imputed datasets.
ANCOVA on each of the 100 datasets was performed by including fixed effects for treatment, current use of statins or other LMTs (y/n), and baseline LDL-C as a covariate.
Treatment effects from the 100 analyses were combined using the Rubin method.
Source: ORION-10 Clinical Study Report.18
Table 32: Time-Adjusted Percentage Change in LDL-C From Baseline Between Day 90 and Day 540 Sensitivity Analyses — ORION-10 (ITT Population)
Sensitivity analysis | Inclisiran (N = 781) | Placebo (N = 780) | Mean difference from placebo |
|---|---|---|---|
MMRM,a LSM (95% CI) | −53.15 (−54.79 to −51.50) | 2.72 (1.06 to 4.39) | −55.87 (−58.21 to −53.53) P < 0.0001 |
Control-based PMM, including current use of statin/LMT,b LSM (95% CI) | −46.33 (−48.91 to −43.76) | 7.47 (4.89 to 10.05) | −53.80 (−56.23 to −51.37) P < 0.0001 |
Two-sample t-test,c LSM (95% CI) | −51.25 (−52.89 to −49.62) | 2.50 (0.63 to 4.37) | −53.75 (−56.24 to −51.27) P < 0.0001 |
CI = confidence interval; ITT = intention to treat; LDL-C = low-density lipoprotein cholesterol; LMT = lipid-modifying therapy; LSM = least squares mean; MAR = missing at random; MMRM = mixed-effects model for repeated measures; PMM = pattern-mixture model.
aAn MMRM analysis that assumes missing data are MAR was performed. The model included fixed effects for treatment, visit, interaction between treatment and visit, and baseline LDL-C as a covariate.
bA control-based PMM was used for missing data imputation with 100 total imputed datasets. An MMRM on each of the 100 datasets was performed by including fixed effects for treatment, visit, interaction between treatment and visit, current use of statins or other LMTs (y/n), and baseline LDL-C as a covariate.
cThe time-adjusted percentage change was calculated by taking the arithmetic mean of calculated percentage change in LDL-C from baseline at each visit after day 90 through day 540.
Source: ORION-10 Clinical Study Report.18
ORION-11 Sensitivity Analyses
Table 33: Percentage Change in LDL-C From Baseline to Day 510 Sensitivity Analyses — ORION-11 (ITT Population)
Sensitivity analysis | Inclisiran (N = 810) | Placebo (N = 807) | Mean difference from placebo |
|---|---|---|---|
Control-based PMM,a LSM (95% CI) | −47.73 (−49.93 to −45.53) | 4.09 (1.88 to 6.31) | −51.82 (−54.94 to −48.70) P < 0.0001 |
MMRM,b LSM (95% CI) | −48.81 (−50.98 to −46.64) | 3.87 (1.71 to 6.03) | −52.68 (−55.74 to −49.62) P < 0.0001 |
ANCOVA from multiple imputation washout model, including country,c LSM (95% CI) | −47.95 (−51.87 to −44.02) | 1.93 (−1.84 to 5.71) | −49.88 (−55.30 to −44.46) P < 0.0001 |
ANCOVA = analysis of covariance; CI = confidence interval; ITT = intention to treat; LDL-C = low-density lipoprotein cholesterol; LSM = least squares mean; MAR = missing at random; MMRM = mixed-effects model for repeated measures; PMM = pattern-mixture model.
aA control-based PMM was used for missing data imputation with 100 total imputed datasets. An MMRM on each of the 100 datasets was performed by including fixed effects for treatment, visit, interaction between treatment and visit, and baseline LDL-C as a covariate. A linear contrast at day 510 was used to compare treatment groups. Treatment effects from the 100 analyses were combined using the Rubin method.
bAn MMRM analysis that assumes missing data are MAR was performed. The model included fixed effects for treatment, visit, interaction between treatment and visit, and baseline LDL-C as a covariate. The restricted maximum likelihood estimation approach was used with the covariance structure set as unstructured. A linear contrast at day 510 was used to compare treatment groups.
cA multiple imputation washout model was used for missing data imputation with 100 total imputed datasets. This modified model assumed missing day 510 MAR for inclisiran patients if they received all 4 doses and had data observed at day 540.
ANCOVA on each of the 100 datasets was performed by including fixed effects for treatment, country, interaction between treatment and country, and baseline LDL-C as a covariate. Treatment effects from the 100 analyses were combined using Rubin method.
Source: ORION-11 Clinical Study Report.19
Table 34: Time-Adjusted Percentage Change in LDL-C From Baseline Between Day 90 and Day 540 Sensitivity Analyses — ORION-11 (ITT Population)
Sensitivity analysis | Inclisiran (N = 810) | Placebo (N = 807) | Mean difference from placebo |
|---|---|---|---|
MMRM,a LSM (95% CI) | −46.58 (−48.25 to −44.90) | 3.35 (1.67 to 5.02) | −49.92 (−52.29 to −47.55) P < 0.0001 |
Control-based PMM, including country,b LSM (95% CI) | −47.35 (−50.20 to −44.50) | 4.05 (1.26 to 6.83) | −51.39 (−55.37 to −47.42) P < 0.0001 |
Two-sample t-test,c LSM (95% CI) | −45.97 (−47.48 to −44.47) | 3.50 (1.60 to 5.40) | −49.47 (−51.90 to −47.05) P < 0.0001 |
CI = confidence interval; ITT = intention to treat; LDL-C = low-density lipoprotein cholesterol; LSM = least squares mean; MAR = missing at random; MMRM = mixed-effects model for repeated measures; PMM = pattern-mixture model.
aAn MMRM analysis that assumes missing data were MAR was performed. The model included fixed effects for treatment, visit, interaction between treatment and visit, and baseline LDL-C as a covariate.
bA control-based PMM was used for missing data imputation with 100 total imputed datasets.
cThe time-adjusted percentage change was calculated by taking the arithmetic mean of calculated percentage change in LDL-C from baseline at each visit after day 90 through day 540.
Source: ORION-11 Clinical Study Report.19
Subgroup Analysis
Figure 8: Forest Plot of Treatment Differences in Percentage Change From Baseline in LDL-C at Day 510 — Subgroup Analyses (ORION-9 ITT Population)
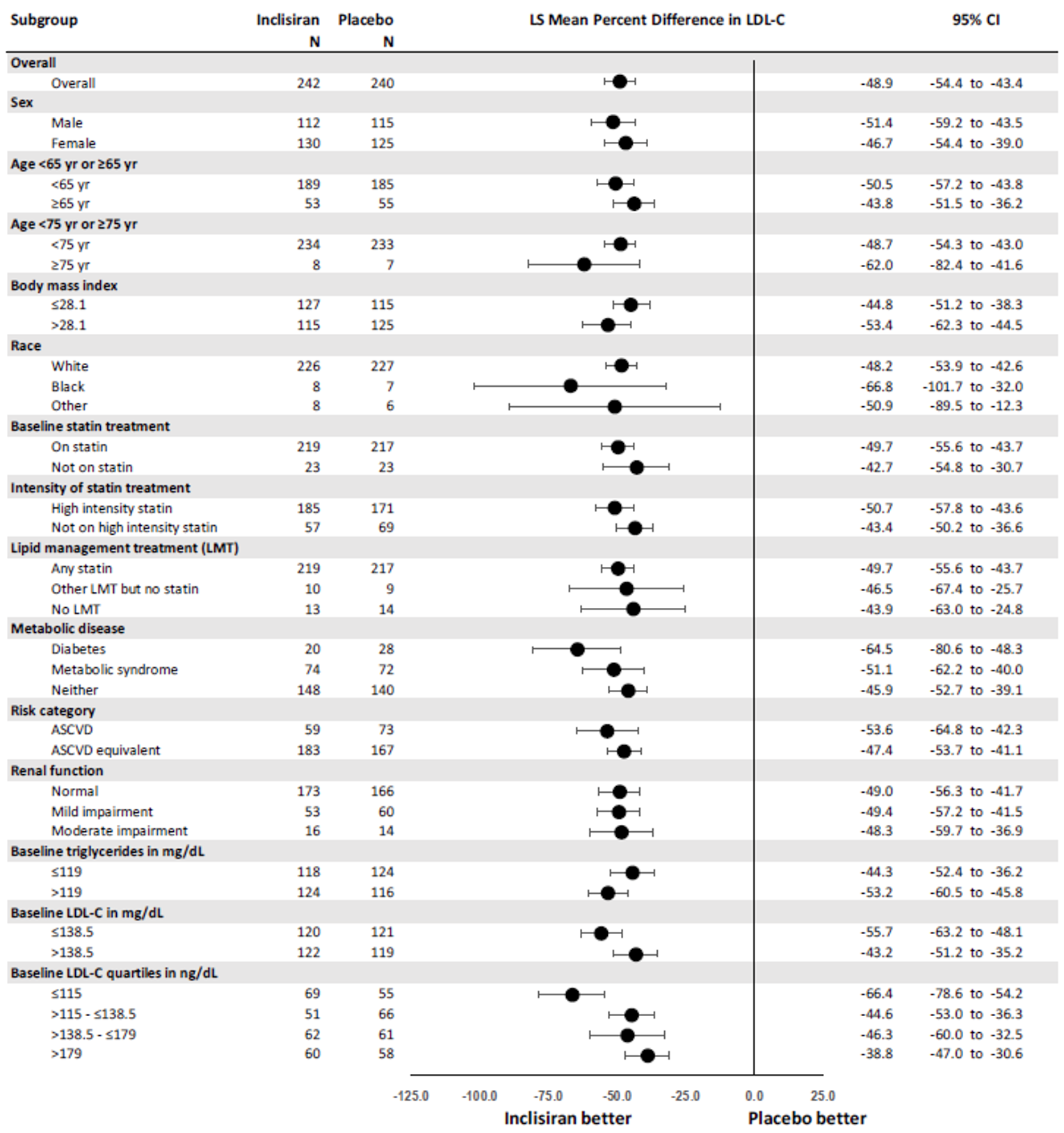
ASCVD = atherosclerotic cardiovascular disease; CI = confidence interval; LDL-C = low-density lipoprotein cholesterol; LMT = lipid-modifying therapy; LS = least squares; yr = years.
Source: ORION-9 Clinical Study Report.17
Figure 9: Forest Plot of Treatment Differences in Time-Adjusted LDL-C After Day 90 and Up to Day 540 — Subgroup Analyses (ORION-9)
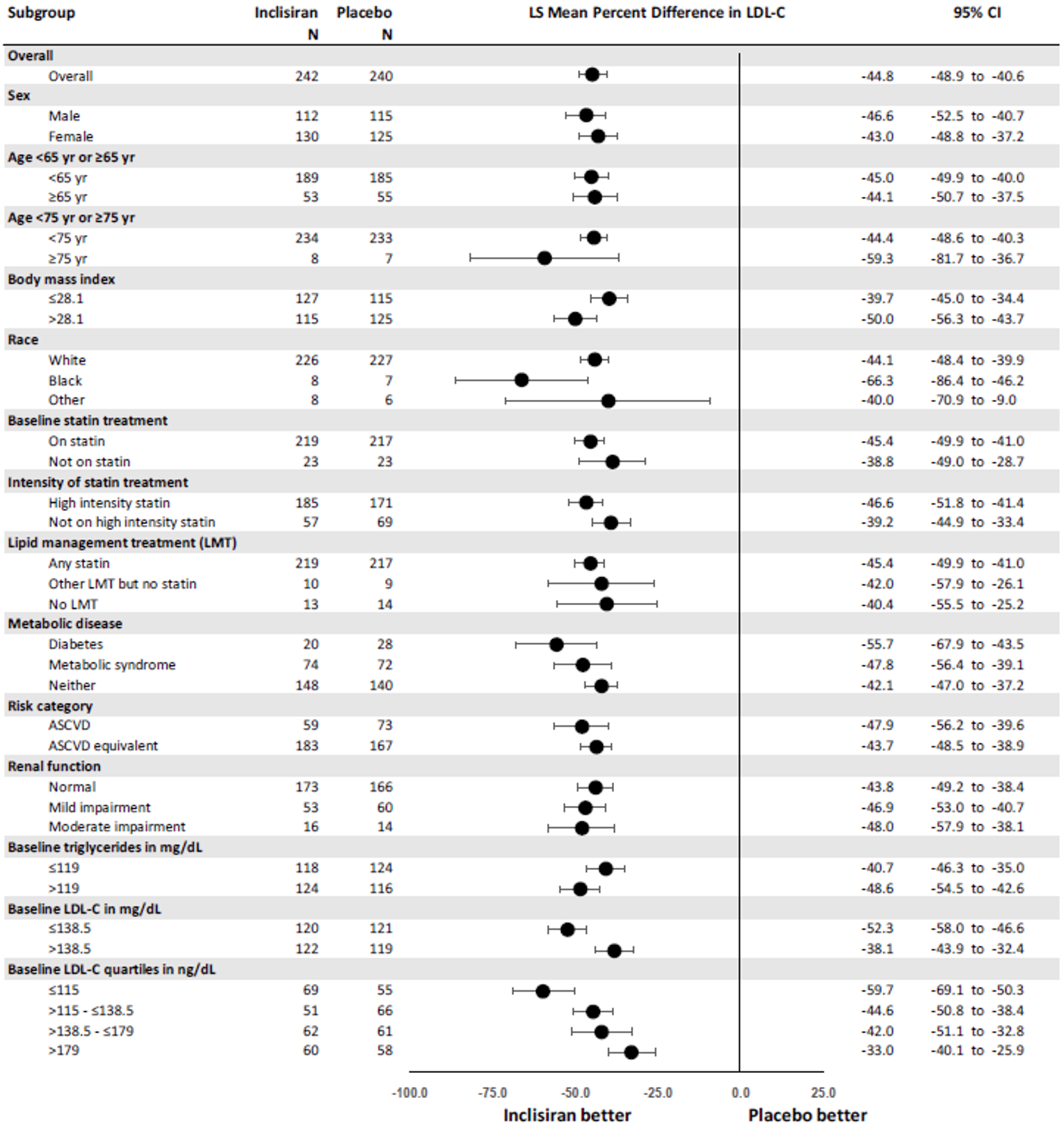
ASCVD = atherosclerotic cardiovascular disease; CI = confidence interval; LDL-C = low-density lipoprotein cholesterol; LMT = lipid-modifying therapy; LS = least squares; yr = years.
Source: ORION-9 Clinical Study Report.17
Figure 10: Forest Plot of Treatment Differences in Percentage Change From Baseline in LDL-C at Day 510 — Subgroup Analyses (ORION-10 ITT Population)
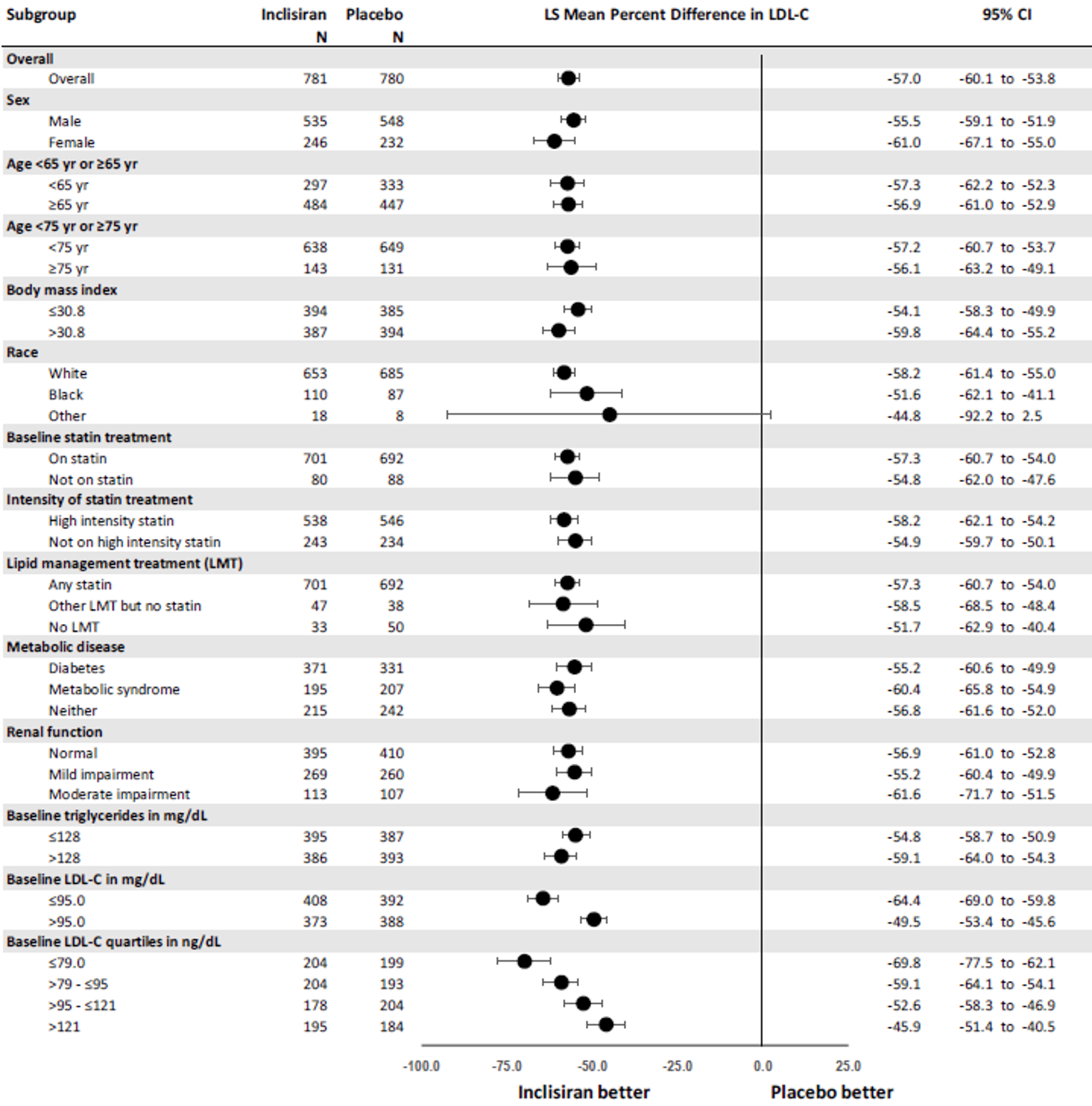
CI = confidence interval; LDL-C = low-density lipoprotein cholesterol; LMT = lipid-modifying therapy; LS = least squares; yr = year.
Source: ORION-10 Clinical Study Report.18
Figure 11: Forest Plot of Treatment Differences in Time-Adjusted LDL-C After Day 90 and Up to Day 540 — Subgroup Analyses (ORION-10 ITT Population)
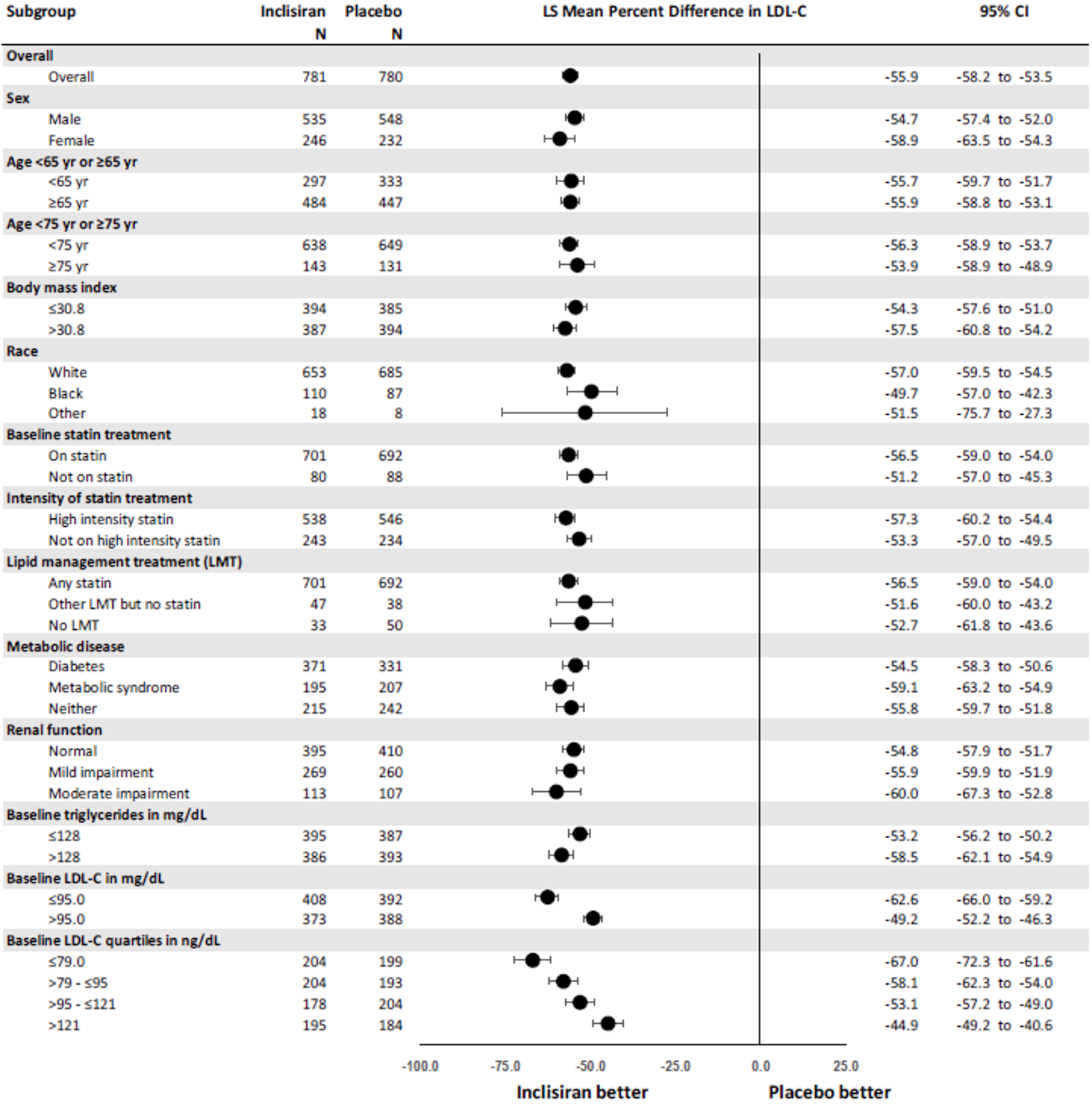
CI = confidence interval; LDL-C = low-density lipoprotein cholesterol; LMT = lipid-modifying therapy; LS = least squares; yr = year.
Source: ORION-10 Clinical Study Report.18
Figure 12: Forest Plot of Treatment Differences in Percentage Change in LDL-C From Baseline to Day 510 — Subgroup Analyses (ORION-11 ITT Population)
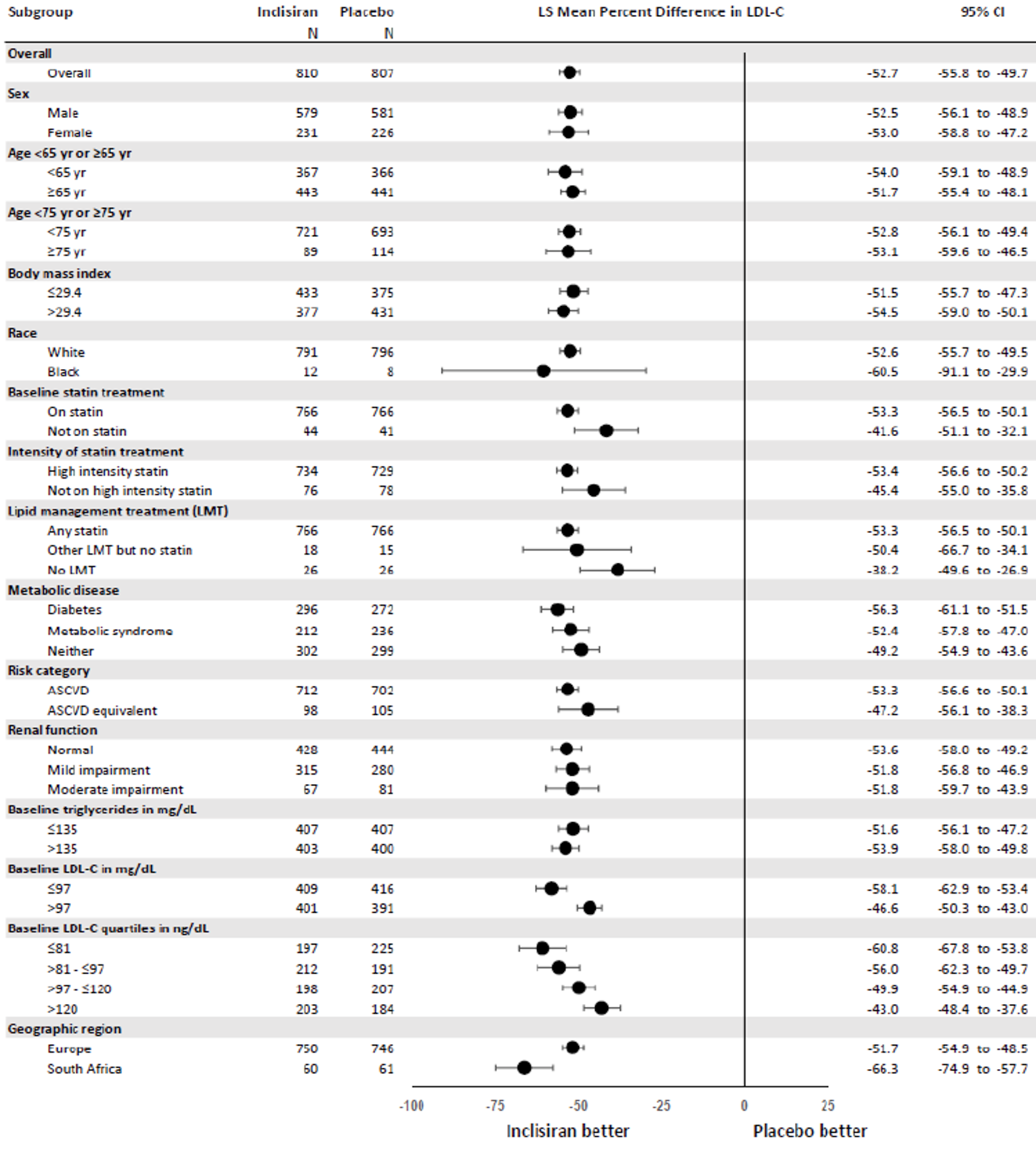
ASCVD = atherosclerotic cardiovascular disease; CI = confidence interval; LDL-C = low-density lipoprotein cholesterol; LMT = lipid-modifying therapy; LS = least squares; yr = year.
Source: ORION-11 Clinical Study Report amendment.19
Figure 13: Forest Plot of Treatment Differences in Time-Adjusted Percentage Change in LDL-C After Day 90 and Up to Day 540 — Subgroup Analyses (ORION-11 ITT Population)
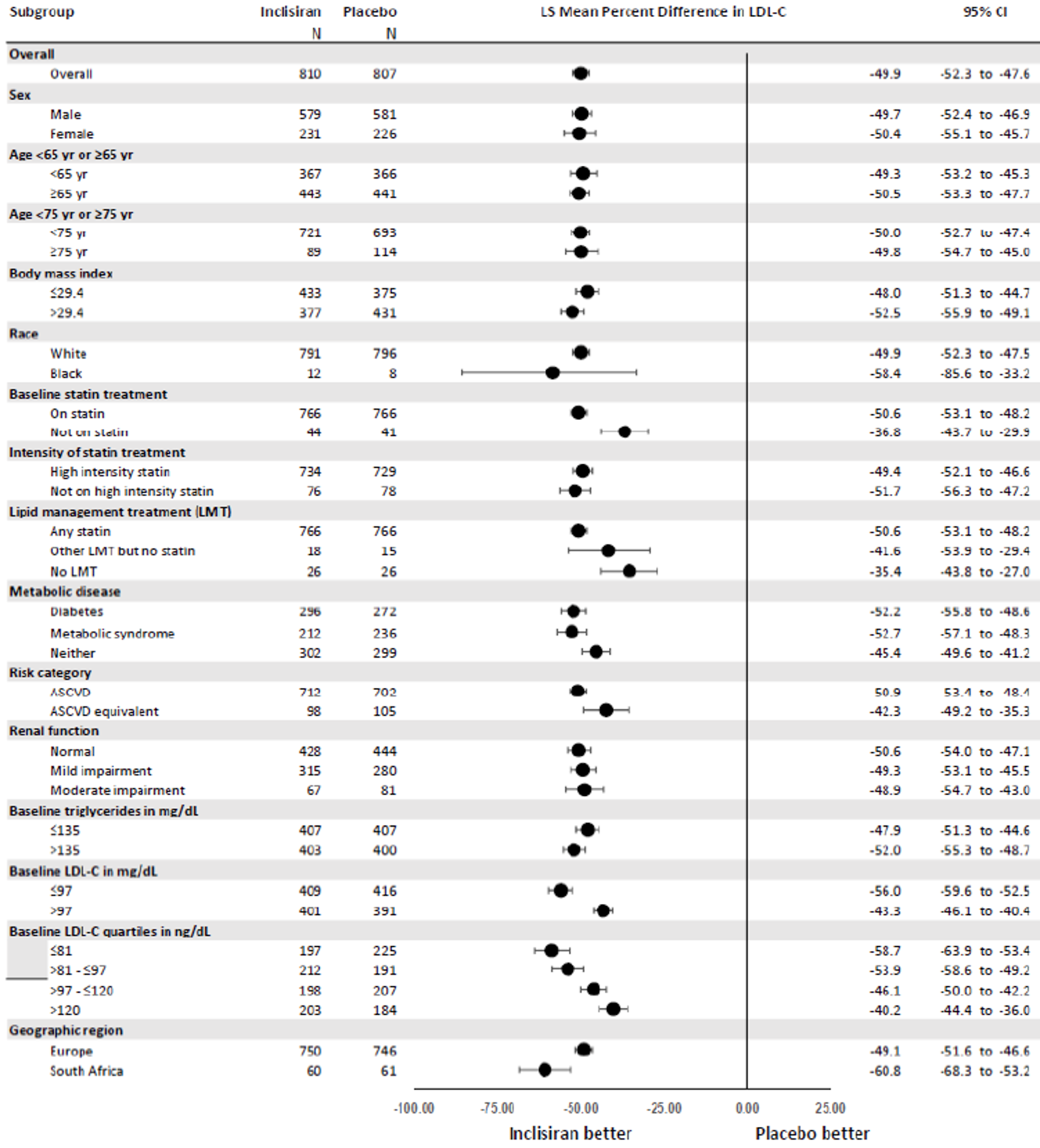
CI = confidence interval; LDL-C = low-density lipoprotein cholesterol; LMT = lipid-modifying therapy; LS = least squares; yr = year.
Source: ORION-11 Clinical Study Report amendment.19
Table 35: Incidence of Clinically Significant Creatinine Kinase — ORION-9, -10, and -11 (Safety Population)
Category | ORION-9 | ORION-10 | ORION-11 | |||
|---|---|---|---|---|---|---|
Inclisiran (N = 241) | Placebo (N = 240) | Inclisiran (N = 781) | Placebo (N = 778) | Inclisiran (N = 811) | Placebo (N = 804) | |
Creatine kinase (U/L), n (%) | ||||||
> 5 × ULN | 4 (1.7) | 5 (2.1) | 10 (1.3) | 8 (1.0) | 10 (1.2) | 9 (1.1) |
||||||||||| | ||||||||||| | ||||||||||| | ||||||||||| | ||||||||||| | ||||||||||| | ||||||||||| |
||||||||||| | ||||||||||| | ||||||||||| | ||||||||||| | ||||||||||| | ||||||||||| | ||||||||||| |
||||||||||| | ||||||||||| | ||||||||||| | ||||||||||| | ||||||||||| | ||||||||||| | ||||||||||| |
ULN = upper limit of normal.
Note: Clinically significant criteria are met when both of the following occur:
• post-baseline values meet the thresholds, and
• baseline values or any prior post-baseline values do not meet the thresholds.
The worst post-baseline value will be utilized in the analyses.
Source: Clinical Study Reports for ORION-9,17 ORION-10,18 and ORION-11.19
Pharmacoeconomic Review
Abbreviations
ACS
acute coronary syndrome
ASCVD
atherosclerotic cardiovascular disease
BIA
budget impact analysis
CTTC
Cholesterol Treatment Trialists’ Collaboration
CUA
cost-utility analysis
HeFH
heterozygous familial hypercholesterolemia
ICER
incremental cost-effectiveness ratio
LDL-C
low-density lipoprotein cholesterol
NMA
network meta-analysis
PCSK9
proprotein convertase subtilisin/kexin type 9
QALY
quality-adjusted life-year
RR
rate ratio
SoC
standard of care
WTP
willingness to pay
Executive Summary
The executive summary comprises 2 tables (Table 1 and Table 2) and a conclusion.
Item | Description |
|---|---|
Drug product | Inclisiran, solution for subcutaneous injection: 284 mg per 1.5 mL pre-filled syringe (189 mg/mL) |
Submitted price | Inclisiran, 284 mg: $2,839.28 per pre-filled syringe |
Indication | Proposed: As an adjunct to lifestyle changes, including diet, to further reduce low-density lipoprotein cholesterol (LDL-C) level in adults with the following conditions who are on a maximally tolerated dose of a statin, with or without other LDL-C-lowering therapies:
|
Health Canada approval status | Under review (pre-NOC) |
Health Canada review pathway | Standard |
NOC date | July 26, 2021 |
Reimbursement request | As an adjunct to diet and maximally tolerated statin therapy, with or without other lipid-lowering therapies, in adult patients with heterozygous familial hypercholesterolemia or clinical atherosclerotic cardiovascular disease who require additional lowering of LDL-C |
Sponsor | Novartis Pharmaceuticals Canada Inc. |
Submission history | Previously reviewed: No |
LDL-C = low-density lipoprotein cholesterol; NOC = Notice of Compliance.
Table 2: Summary of Economic Evaluation
Component | Description |
|---|---|
Type of economic evaluation | Cost-utility analysis Markov model |
Target populations | Adult patients with heterozygous familial hypercholesterolemia (HeFH) or clinical atherosclerotic cardiovascular disease (ASCVD) who require additional lowering of low-density lipoprotein cholesterol (LDL‐C) despite maximally tolerated statin therapy |
Treatments | Inclisiran + standard of care (SoC; defined as maximally tolerated statins with or without ezetimibe) |
Comparator | ASCVD patients: SoC HeFH patients: SoC, evolocumab + SoC; alirocumab + SoC |
Perspective | Canadian publicly funded health care payer |
Outcome | QALYs, life-years |
Time horizon | Lifetime (40 years) |
Key data source | The impact of treatment on LDL-C was informed by network meta-analyses for inclisiran (ORION-9, ORION-10, ORION-11), evolocumab, and alirocumab. SoC was assumed to have no effect on LDL-C level. |
Submitted results | ASCVD subgroup: ICER for inclisiran plus SoC vs. SoC alone = $79,726 per QALY (incremental cost, $64,017; incremental QALYs, 0.80). HeFH subgroup: Sequential ICER of inclisiran plus SoC vs. SoC alone = $175,172 per QALY (incremental cost, $97,621; incremental QALYs, 0.56). Evolocumab and alirocumab were dominated or extendedly dominated by inclisiran and SoC. |
Key limitations | The effect of inclisiran on cardiovascular outcomes is highly uncertain. The predicted survival benefit for patients treated with inclisiran has not been shown in clinical trials. The sponsor’s model used a surrogate outcome, LDL-C, to approximate the relationship between treatment and cardiovascular risk. The comparative clinical effectiveness of inclisiran to other PCSK9 inhibitors is highly uncertain. There have been no head-to-head trials of inclisiran vs. other PCSK9 inhibitors, and there is substantial uncertainty in the results of the sponsor’s network meta-analyses. The sponsor considers relative, but not absolute, changes in LDL-C levels. The clinical expert consulted by CADTH for this review indicated that absolute changes may be a more relevant measure of effect for patients with HeFH. The baseline risk of cardiovascular events in the modelled population may not reflect risk in the Canadian population. Inclisiran was assumed to maintain consistent treatment effectiveness over the model's 40-year analysis horizon. The long-term effectiveness of inclisiran has not been assessed beyond 18 months of treatment in clinical trials. The sponsor employed poor modelling practices in their model, preventing CADTH from fully validating the model and its findings. |
CADTH reanalysis results | In the CADTH reanalyses, in light of the high level of uncertainty in the comparative clinical evidence, the effectiveness inputs are informed by direct evidence from the ORION-9 (HeFH subgroup) and ORION-10 (ASCVD subgroup) trials, with a pairwise comparison of inclisiran plus SoC vs. SoC alone. In addition, a similar relationship was assumed between LDL-C reduction and cardiovascular risk as observed with evolocumab in the FOURIER trial. CADTH was unable to address: the inability to reflect the effect of inclisiran on absolute changes in LDL-C in the HeFH subgroup, uncertainty regarding the baseline risk of cardiovascular events, and uncertainty regarding the long-term clinical effectiveness of inclisiran. Based on the CADTH reanalyses, inclisiran plus SoC remained more costly and more effective than SoC alone in both the ASCVD and HeFH subgroups:
A price reduction of 83% would be required for inclisiran to be considered optimal at a WTP threshold of $50,000 per QALY in the ASCVD subgroup, while a price reduction of 91% would be required for inclisiran to be considered optimal in the HeFH subgroup. |
ASCVD = atherosclerotic cardiovascular disease; HeFH = heterozygous familial hypercholesterolemia; ICER = incremental cost-effectiveness ratio; LDL-C = low-density lipoprotein cholesterol; PCSK9 = proprotein convertase subtilisin/kexin type 9; QALY = quality-adjusted life-year; SoC = standard of care; WTP = willingness to pay.
Conclusions
Inclisiran plus standard of care (SoC) reduces low-density lipoprotein cholesterol (LDL-C) compared with SoC in patients with atherosclerotic cardiovascular disease (ASCVD) or heterozygous familial hypercholesterolemia (HeFH), although the comparative effects of inclisiran relative to other proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors (i.e., evolocumab, alirocumab) and its long-term effects are highly uncertain. There is no direct clinical evidence to substantiate whether the reduction in LDL-C with inclisiran treatment reduces the risk of cardiovascular events.
CADTH undertook reanalyses to address limitations in the sponsor’s submission, including assuming a similar relationship between LDL-C reduction and cardiovascular risk as observed with evolocumab in its cardiovascular outcomes trial (FOURIER trial), by adopting hazard ratios from FOURIER that were observed with evolocumab treatment. CADTH was unable to address: the inability to reflect absolute changes in LDL-C; uncertainty regarding the baseline risk of cardiovascular events; and uncertainty regarding the long-term clinical effectiveness of inclisiran. In addition, CADTH could not address the uncertainty with the results of the sponsor’s network meta-analyses (NMA). As such, CADTH reanalyses focused on the cost-effectiveness of inclisiran plus SoC relative to SoC alone in HeFH and ASCVD patients, using direct evidence from the ORION-9 and ORION-10 trials, respectively.
In the CADTH reanalyses, inclisiran plus SoC was more costly and more effective than SoC alone in both subgroups, which is aligned with the sponsor’s submission. In the ASCVD subgroup, inclisiran plus SoC was associated with an incremental cost-effectiveness ratio (ICER) of $366,650 per quality-adjusted life-year (QALY) relative to SoC, while the ICER for inclisiran plus SoC was $626,458 per QALY relative to SoC in the HeFH subgroup. In both subgroups, the cost of inclisiran was a key driver of the ICER. In the ASCVD subgroup, an 83% price reduction would be required for inclisiran to be considered optimal at a willingness-to-pay (WTP) threshold of $50,000, and a 91% price reduction would be required for it to be considered optimal in the HeFH subgroup. This price reduction may be an underestimate, given the cost-effectiveness is reliant on achieving a reduction in cardiovascular risk and in maintaining long-term treatment benefit, neither of which has been shown in clinical trials.
Whether inclisiran reduces the risk of cardiovascular events is uncertain, and the CADTH base-case results are highly sensitive to the use of LDL-C as a surrogate marker for cardiovascular outcomes. While there was no difference in cardiovascular outcomes between inclisiran and placebo in the ORION clinical trials, these trials were not designed or powered to adequately assess cardiovascular outcomes. In the CADTH reanalyses, based on the input of the clinical expert consulted by CADTH for this review, the reduction in cardiovascular risk for each 1 mmol/L reduction in LDL-C with inclisiran treatment was modelled based on the reduction in risk observed with evolocumab in its cardiovascular outcomes trial. The notable increase in the ICER in both subgroups with this change highlights the uncertainty associated with the use of LDL-C as a surrogate outcome in the model. As such, CADTH’s estimate of the incremental effectiveness (and therefore cost-effectiveness) of inclisiran remains highly uncertain.
Stakeholder Input Relevant to the Economic Review
This section is a summary of the feedback received from the patient groups, registered clinicians, and drug plans that participated in the CADTH review process.
Patient input was received from the Canadian Heart Patient Alliance and the HeartLife Foundation, through interviews and online surveys, including from patients with “high lipids,” HeFH, and ASCVD. Patients described symptoms of high cholesterol including headaches, chest pains, muscle pains, shortness of breath, xanthomas, weakness, fatigue, muscle loss, and neuropathy. Some participants noted that their high cholesterol or high lipid condition had little or no effect on their quality of life; however, some noted that they experienced anxiety related to the “unpredictability” of a cardiovascular event and the potential for severe consequences, concerns about medications not working, and the impact on their children (e.g., through diagnosis or risk). Patients reported experience with drug therapies (statins, proprotein convertase subtilisin/kexin type 9 [PCSK9] inhibitors), diet and/or exercise, and apheresis. Most respondents reported they were not well managed on diet or nutritional supplements alone and reported currently or previously taking statins. Statins were described by many as working “well or very well;” however, many stated that their cholesterol levels were not managed by statins and some reported experiencing “severe” or “very severe” adverse effects while taking statins. Patients described PCSK9 inhibitors as working well to reduce their cholesterol levels with few adverse effects, and some reported a positive impact of PCSK9 inhibitors on their quality of life. Patients who had experience with inclisiran described a significant reduction in low-density lipoprotein cholesterol (LDL-C) levels with no adverse effects. Patients noted that a biannual injection would be preferred over treatments that require more frequent administration.
Input from a group of lipid specialists and physicians working in lipid clinics in British Columbia noted that many patients do not reach their guideline-recommended lipid target. Clinicians described an ideal treatment as 1 that would reduce levels of LDL-C, non-high-density lipoprotein cholesterol, and apolipoprotein B, reduce the risk of major adverse cardiovascular events, be safe and well-tolerated, and have properties that promote adherence (e.g., non-daily administration). Clinicians indicated that inclisiran would most likely be used after statin therapy had been optimized (with or without ezetimibe) or for those who are intolerant to statins. Clinicians noted that patients with either a lifetime or short-term high risk of a cardiovascular event (e.g., patients requiring secondary ASCVD prevention, those with familial hypercholesterolemia, and those with diabetes or a high Framingham Risk score) would be most suited for inclisiran. The clinician group noted that inclisiran may be considered for patients with documented atherosclerotic disease who have not had a clinical event, as well as other guideline-recommended groups at high risk of a cardiovascular event who would potentially benefit from LDL-C lowering (e.g., those with a high Framingham Risk score, diabetes mellitus, or chronic kidney disease).
CADTH participating drug plans noted that comparators for inclisiran include PCSK9 inhibitors (evolocumab, alirocumab), which are reimbursed only for the HeFH population, as well as ezetimibe and icosapent ethyl. The plans noted that inclisiran may be considered for patients taking a maximally tolerated dose of statins, defined as atorvastatin 80 mg/day or rosuvastatin 40 mg/day. The plans noted that the administration of inclisiran by a health care professional will be associated with administration costs.
Several of these concerns were addressed in the sponsor’s model:
Inclisiran was assumed to be used after statin therapy (with or without ezetimibe) or for those who are intolerant to statins.
Evolocumab and alirocumab were included as comparators in the HeFH population; however, there were important limitations with the sponsor’s NMAs, and the interpretation of the findings is unclear. A proportion of patients in each subgroup were assumed to receive ezetimibe as part of SoC. Icosapent ethyl was not included as a comparator.
Inclisiran administration costs were included in the model but were underestimated.
CADTH was unable to address the following concerns raised from stakeholder input:
The sponsor’s model does not consider the effect of inclisiran on non-high-density lipoprotein cholesterol or apolipoprotein B.
Patient-reported symptoms of high cholesterol (e.g., headaches, chest pains, muscle pains) were not included in the sponsor’s model.
Economic Review
The current review is for inclisiran for patients with HeFH or non-familial ASCVD.
Economic Evaluation
Summary of Sponsor’s Economic Evaluation
Overview
The sponsor submitted a cost-utility analysis (CUA) of inclisiran plus SoC in patients with HeFH or non-familial ASCVD.1 The cost-effectiveness of inclisiran plus SoC was compared with SoC in ASCVD patients, and to SoC, alirocumab plus SoC, and evolocumab plus SoC in HeFH patients. The sponsor assumed that SoC comprised maximally tolerated statin therapy with or without ezetimibe (7% of patients were assumed to receive ezetimibe).1 The sponsor’s reimbursement request is for inclisiran as an adjunct to diet and maximally tolerated statin therapy, with or without other lipid‐lowering therapies, in adult patients with HeFH or clinical ASCVD who require additional lowering of LDL‐C.1 Two subgroup analyses that aligned with the reimbursement request were submitted (i.e., for patients with HeFH or ASCVD). Patients with ASCVD were assumed to include those with an acute coronary syndrome (ACS) for 0 to 1 or 1 to 2 years, “other” coronary heart disease, ischemic stroke, or peripheral artery disease, while patients with HeFH included primary and secondary prevention.
Inclisiran is available as a pre-filled 284 mg/1.5 mL syringe (189 mg/mL) for administration as a subcutaneous injection by a health care professional.2 The recommended dosage for inclisiran is 284 mg administered initially, at month 3, and subsequently every 6 months.2 The annual cost of inclisiran is $5,679 ($8,518 in the initial year) based on a unit cost of $2,839 per syringe. The annual cost of SoC was estimated by the sponsor to be $58 per patient, while the annual cost for both evolocumab and alirocumab was estimated to be $6,661 per patient.
The clinical outcomes were QALYs and life-years. The sponsor adopted a lifetime horizon (40 years) using yearly cycles and undertook the analysis from the perspective of the publicly funded health care payer. Costs and clinical outcomes were discounted at a rate of 1.5% per year.
Model Structure
The sponsor submitted a Markov model with 15 health states, including 3 initial health states (stable, 0 to 1 years following a previous cardiovascular event, 1 to 2 years following a previous event), 10 post-cardiovascular event health states: post unstable angina (0 to 1 years, 1 to 2 years), stable (i.e., > 2 years after an event), post non-fatal myocardial infarction (0 to 1 years, 1 to 2 years, stable), post non-fatal stroke (0 to 1 years, 1 to 2 years, stable), and revascularization (stable), and 2 death states (cardiovascular, non-cardiovascular) (Figure 1). At cohort entry, patients were distributed between the 3 initial health states based on time since a prior cardiovascular event (stable, 0 to 1 years post event, 1 to 2 years post event). Patients receiving treatment as primary prevention in the HeFH subgroup and ASCVD patients who had experienced a previous event at least 2 years earlier or for whom the risk of a subsequent event was assumed to not vary by time since event (i.e., peripheral artery disease, stroke) entered the model in the stable state. From an initial health state, patients could experience a non-fatal cardiovascular event (unstable angina, myocardial infarction, stroke, revascularization) or a fatal event (cardiovascular death, non-cardiovascular death). Following a non-fatal cardiovascular event, patients progressed through subsequent health states based on time since the event and remained at risk of a subsequent event (fatal or non-fatal). Movement through the model was based on time since a previous cardiovascular event as well as the risk of experiencing a subsequent event.
Model Inputs
The baseline patient characteristics in the model (age, prevalence of diabetes and average LDL-C) were aligned with those of the ORION trials (ORION-9, ORION-10, ORION-11), depending on the patient subgroup.1 Baseline characteristics for patients with HeFH were based on the HeFH subgroup from ORION-9 (52.36 years, 58% female, 7% diabetes, LDL-C of 4.09 mmol/L), while the characteristics of patients with ASCVD were based on ASCVD subgroups from ORION-10 and/or ORION-11 (64.75 years, 34% female, 38% diabetes, LDL-C of 3.47 mmol/L).1 20% of patients in both subgroups were assumed to be intolerant to statins. At baseline, patients with HeFH were assumed to be receiving treatment for primary prevention (72.6% of patients) or secondary prevention (27.4%) based on the distribution in ORION-9. The distribution of patients within the ASCVD subgroup was based on the alirocumab submission to the National Institute for Health and Care Excellence3 (ACS in the previous 0 to 1 years, 8.7% of patients; ACS in the previous 1 to 2 years, 0.9%; “other” coronary heart disease, 62.3%; ischemic stroke, 19.2%; peripheral artery disease, 8.9%). Baseline cardiovascular risk for ASCVD patients was based on the Clinical Practice Research Database (CPRD) Aurum cohort; baseline cardiovascular risk for HeFH patients was based on the CPRD cohort and a retrospective cohort involving 131 patients with confirmed HeFH in the Netherlands.4 Baseline cardiovascular event risks were adjusted to reflect the average level of LDL-C within the population being modelled by assuming a log-linear relationship between LDL-C and the probability of experiencing a cardiovascular event.5 The annual probability of a cardiovascular event was based on data from the CPRD analysis for ASCVD patients and on Mohrschladt4 for HeFH patients. The rate of non-cardiovascular death was derived by subtracting the rate of cardiovascular death from the annual age- and sex-stratified rate of all-cause death; the source of all-cause death rates was not cited by the sponsor.
Treatment effect in the sponsor’s model was based on a surrogate outcome for cardiovascular risk (i.e., percentage reduction in LDL-C at 24 weeks). The sponsor modelled the relationship between LDL-C reduction and cardiovascular risk based on an NMA of data from statin trials comparing the effects of statins on cardiovascular risk.6 From this NMA, the sponsor adopted the following rate ratios (RRs) for each 1.0 mmol/L reduction in LDL-C: a cardiovascular mortality RR of 0.84 (95% CI, 0.80 to 0.88), a non-fatal myocardial infarction RR of 0.73 (95% CI, 0.70 to 0.76), an ischemic stroke RR of 0.79 (95% CI, 0.74 to 0.85), an any stroke RR of 0.81 (95% CI, 0.77 to 0.86), and a revascularization RR of 0.75 (95% CI, 0.72 to 0.78). In the absence of head-to-head data, the sponsor conducted de novo NMAs to derive the comparative percentage reduction in LDL-C at 24 weeks for inclisiran, alirocumab, evolocumab, and ezetimibe.7 For patients with ASCVD, the sponsor assumed that the risk of cardiovascular death, myocardial infarction, or stroke would increase by 50% for subsequent events. The probability of treatment discontinuation was assumed to be equivalent across PCSK9 inhibitors (2%) and was based on the number of patients who discontinued alirocumab and evolocumab in a single-centre retrospective cohort.7
The sponsor estimated age- and gender-adjusted EuroQol 5-Dimensions questionnaire (EQ-5D) utility values for individuals with no history of cardiovascular disease,8 and estimated baseline utility values for each health state by multiplying these utilities by “utility multipliers” for each cardiovascular event.9 Post-cardiovascular event disutilities (unstable angina, non-fatal stroke, non-fatal myocardial infarction) were applied multiplicatively to the health state utilities. Patients were assumed to experience acute disutility in the first year after an event, after which they would experience a chronic post-event utility. The source of the post-event utility multipliers was not provided by the sponsor. No adverse events were considered in the sponsor’s model.
The model included drug costs (acquisition, administration), as well as costs related to the management of cardiovascular events. Drug acquisition costs for inclisiran were based on the sponsor’s submitted price, while the price of alirocumab, evolocumab, statins, and ezetimibe was obtained from the Ontario Drug Benefit formulary.10 The relative use of individual statins and statin doses as part of SoC was obtained from IQVIA.11 Administration costs were included only for inclisiran, which the sponsor assumed would be administered by a nurse and would incur the cost of 10 minutes of nursing time.12 Costs associated with cardiovascular events (myocardial infarction, unstable angina, stroke, revascularization) were obtained from the literature.13-17 The cost of cardiovascular death was based on a generic 1-month end-of-life cost in Canada.15
Summary of Sponsor’s Economic Evaluation Results
The sponsor-submitted probabilistic analyses aligned with the reimbursement request, stratified by patient subgroup (ASCVD, HeFH). The sponsor’s cost-effectiveness analysis was based on 500 probabilistic iterations, for which findings are presented subsequently. Additional results from the sponsor’s submitted economic evaluation base case are presented in Appendix 3. The submitted analyses were based on the publicly available prices of the comparator treatments.
Base-Case Results
Among ASCVD patients, inclisiran plus SoC was associated with an incremental cost of $64,017 and 0.80 additional QALYs compared with SoC over a 40-year horizon, resulting in an ICER of $79,726 per QALY (Table 3). At a WTP threshold of $50,000 per QALY, the probability of inclisiran plus SoC being considered the most likely cost-effective intervention was 0%.
The drug costs associated with inclisiran were key drivers of the ICER (incremental cost of $68,019 versus SoC) (Appendix 3, Table 13). At the end of the 40-year time horizon, 0% of patients receiving inclisiran plus SoC or SoC alone remained alive.
Table 3: Summary of the Sponsor’s Economic Evaluation Results — ASCVD Patients
Drug | Total costs ($) | Incremental costs ($) | Total QALYs | Incremental QALYs | ICER vs. SoC ($/QALY) |
|---|---|---|---|---|---|
SoC | 20,897 | Reference | 8.60 | Reference | Reference |
Inclisiran + SoC | 84,915 | 64,017 | 9.41 | 0.80 | 79,726 |
ASCVD = atherosclerotic cardiovascular disease; ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year; SoC = standard of care.
Source: Sponsor’s pharmacoeconomic submission.1
Among HeFH patients, inclisiran plus SoC was associated with an incremental cost of $97,621 and 0.56 additional QALYs compared with SoC over a 40-year horizon, resulting in an ICER of $176,866 per QALY versus SoC alone (Table 4). Evolocumab and alirocumab were dominated or extendedly dominated by inclisiran and SoC. At a WTP of $50,000 per QALY, the probability of inclisiran plus SoC being considered the most likely cost-effective intervention was 0%.
The drug costs associated with inclisiran were key drivers of the ICER (incremental cost of $100,655 versus SoC) (Appendix 3, Table 14). At the end of the 40-year time horizon, 0% of patients remained alive across treatments.
Table 4: Summary of the Sponsor’s Economic Evaluation Results — HeFH Patients
Drug | Total costs ($) | Total QALYs | Sequential ICER ($/QALY) |
|---|---|---|---|
SoC | 12,574 | 17.15 | Reference |
Inclisiran + SoC | 110,196 | 17.70 | 175,172 vs. SoC |
Evolocumab + SoC | 124,351 | 17.78 | 176,866 vs. inclisiran + SoC |
HeFH = heterozygous familial hypercholesterolemia; ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year; SoC = standard of care; vs. = versus.
Note: Only treatments that are on the efficiency frontier are reported in the main body. Full results are reported in Appendix 3.
Source: Sponsor’s pharmacoeconomic submission.1
Sensitivity and Scenario Analysis Results
The sponsor assessed the impact of several model parameters in probabilistic scenario analyses, including adopting alternative discount rates, time horizons, and discontinuation rates; assuming that the RR for major vascular events varies over time; adopting an alternative distribution of subgroups within the ACSVD population; assuming alternative LDL-C thresholds; adopting alternative stroke costs; adopting a societal perspective; and including evolocumab and alirocumab as comparators for ACSVD patients. Scenarios were also provided in which the HeFH subgroup was further divided by those taking inclisiran as primary or secondary prevention and in which the ASCVD subgroup was divided by the type of precipitating cardiovascular event (ACS for 0 to 1 or 1 to 2 years, coronary heart disease, ischemic stroke, peripheral artery disease).
Among ASCVD patients, most scenarios had no meaningful effect on the ICER, with the exception of increasing the LDL-C threshold to 3.4 mmol/L (ICER of $43,583 versus SoC). Among HeFH patients, most scenarios similarly had no meaningful impact on the ICER, with the following exception: When the cost-effectiveness was analyzed separately for those taking inclisiran for primary or secondary prevention, the ICER for primary prevention was $305,791 per QALY gained versus SoC, while the ICER for secondary prevention was $66,833 per QALY gained.
CADTH Appraisal of the Sponsor’s Economic Evaluation
The effect of inclisiran on cardiovascular outcomes is highly uncertain. In the sponsor’s submission, the treatment effect of inclisiran on cardiovascular events and mortality was estimated based on the surrogate outcome of LDL-C. The relationship between LDL-C reduction and cardiovascular risk was based on that observed with statin treatment in the Cholesterol Treatment Trialists’ Collaboration (CTTC) 2019 meta-analyses.6 Previous CADTH submissions have similarly used this approach.18,19 However, in the 2017 CADTH review of evolocumab,18 it was noted that the reduction in cardiovascular outcomes predicted on the basis of the relationship between statins and cardiovascular risk in the CTTC meta-analyses may not be appropriate for modelling the relationship between PCSK9 inhibitors and cardiovascular risk. Notably, the use of the relationship between LDL-C and cardiovascular risk for statins in the evolocumab submission overestimated the effectiveness of evolocumab compared with that observed in clinical trials.20 Specifically, in the evolocumab submission,18 the sponsor’s model predicted a 50% reduction in the risk of cardiovascular events with evolocumab treatment, while in the FOURIER cardiovascular outcomes trial,20 the risk of cardiovascular events with evolocumab was reduced by 15% to 20%, despite a 59% reduction in LDL-C from baseline. Additionally, in the FOURIER trial, there was no statistically significant difference in cardiovascular or all-cause death between those who received evolocumab or placebo on a background of statin treatment.20
The true relationship between LDL-C reduction and cardiovascular risk with the use of inclisiran is unknown. In the sponsor’s submission, the use of LDL-C as a surrogate outcome predicts a survival benefit for inclisiran compared with SoC in both the HeFH and ASCVD subgroups, which has not been shown in clinical studies. A cardiovascular outcomes trial (ORION-4) is currently underway to assess the effect of inclisiran on cardiovascular outcomes. In the absence of such evidence, the clinical expert consulted by CADTH for this review indicated it is reasonable to expect that the relationship between LDL-C reduction and the risk of cardiovascular events for inclisiran to be similar to that observed in the FOURIER trial20 for evolocumab.
The survival benefit predicted by the sponsor’s model with the use of inclisiran has not been shown in clinical trials and may be overestimated. In the CADTH base case, the observed hazard ratios from the FOURIER cardiovascular outcomes trial20 were used to model the relationship between LDL-C reduction and the risk of a cardiovascular event. This approach is described in Appendix 4.
The comparative clinical effectiveness of inclisiran versus other PCSK9 inhibitors is highly uncertain. There have been no head-to-head trials of inclisiran versus other PCSK9 inhibitors (i.e., evolocumab, alirocumab) in either the HeFH or ASCVD populations. The sponsor conducted NMAs to estimate the relative effectiveness of inclisiran, evolocumab, alirocumab, and SoC in patients with HeFH. For the ASCVD subgroup, although the sponsor’s base case compared only inclisiran and SoC, the sponsor similarly based the relative effectiveness of inclisiran on estimates derived from the NMA. As noted in the CADTH Clinical Report, the interpretation of the findings from the sponsor’s NMAs is limited by several methodological and clinical issues. Namely, the NMAs assumed equivalence of background statins regardless of individual statin or dose, assumed that background ezetimibe is not an effect modifier, did not adjust for baseline characteristics for cardiovascular risk and/or severity, and included a limited time of assessment (24 weeks). Consequently, the NMA is not sufficient to conclude whether inclisiran is superior, inferior, or equivalently effective versus other comparators. The incremental QALYs predicted by the model based on the NMA results should therefore be interpreted with a higher degree of uncertainty than is reflected in the probabilistic analysis when comparing these treatments in all subgroups.
In light of the high level of uncertainty associated with the comparative clinical evidence, the CADTH reanalyses compared inclisiran plus SoC with SoC alone and are informed by direct evidence from the ORION-9 (HeFH subgroup) and ORION-10 (ASCVD subgroup) trials. The cost-effectiveness of inclisiran compared with evolocumab and alirocumab was explored in scenario analyses.
The sponsor’s model does not consider absolute changes in LDL-C. Effectiveness in the sponsor’s model is based solely on the percentage change in LDL-C from baseline. The clinical expert consulted by CADTH indicated that, for patients with HeFH, absolute change is considered a more relevant measure of effect, noting that, for example, a patient with a baseline LDL-C level of 8 mmol/L who had a 50% reduction would still be considered to have a high LDL-C level. As such, clinical effectiveness should consider absolute, as well as relative, changes in LDL-C in this subgroup. As noted in the CADTH Clinical Review, the mean difference in absolute LDL-C in ORION-9 at day 510 between inclisiran and placebo was −68.89 mg/dL (−1.78 mmol/L); however, baseline LDL-C levels were not provided. In ORION-9 at day 510, 65.3% of patients had reached the LDL-C target level of less than 100 mg/dL (2.6 mmol/L), while 40.9% of patients had reached a target LDL-C level of less than 70 mg/dL (1.8 mmol/L).
CADTH was unable to address this limitation owing to the structure of the sponsor’s model.
The baseline risk of cardiovascular events is uncertain. In the absence of Canadian-specific data, the sponsor utilized data from the UK to inform the baseline cardiovascular risks for ASCVD patients from the CPRD database and data from the UK (CRPD) and the Netherlands4 to estimate the baseline risk for HeFH patients. As noted in previous CADTH reviews,18 transition probabilities obtained from CPRD might not be applicable to Canadian ASCVD patients. Additionally, the Netherlands cohort4 included patients from a single university-based lipid centre who were taking statins for primary (62%) or secondary (38%) prevention of cardiovascular events. As noted by the clinical expert consulted by CADTH for this review, the risk of a cardiovascular event for patients with HeFH varies depending on whether the patient has definite, probable, or possible HeFH; genetic testing to confirm definite familial hypercholesterolemia is not routinely available in clinical practice in Canada. It is thus unclear whether the baseline risk in the model approximates that of the eligible Canadian population. If the baseline risk is overestimated, this may overestimate the absolute benefit of inclisiran.
CADTH was unable to address this limitation owing to a lack of clinical data.
Uncertainty regarding long-term clinical effectiveness. The sponsor’s pharmacoeconomic submission assumed that the effects of inclisiran were consistent over the lifetime analysis horizon (40 years), such that patients on treatment were assumed to maintain the initial reduction in LDL-C levels for the entire analysis horizon. CADTH notes that the majority of QALYs are accumulated after the trial horizon and were derived on the basis of extrapolated trial findings rather than an observed benefit. Additionally, the sponsor used a data cut-off of 24 weeks to inform the effectiveness parameter in the model, despite the availability of longer-term data (up to day 510 in ORION-9 and ORION-10). ORION-8, a long-term extension study involving patients who completed ORION-9, ORION-10, or ORION-11, is currently ongoing. The clinical expert consulted by CADTH indicated that, once initiated, PCSK9 treatment is anticipated to be continued indefinitely, with some exceptions (i.e., elderly patients, patients with end-stage kidney disease or heart failure). The clinical expert noted there was no evidence to suggest waning effectiveness in other PCSK9 inhibitors. However, the long-term effectiveness of inclisiran, as well as the true rate of injection-site reactions, is unknown in clinical practice and may differ from evolocumab and alirocumab owing to a different mechanism of action.
CADTH was unable to explore the impact of treatment waning owing to the structure of the sponsor’s model.
Poor modelling practices were employed. The model includes numerous IFERROR statements, which lead to situations in which the parameter value is overwritten with an alternative value without alerting the user to the automatized overwriting. The systematic use of IFERROR statements makes thorough auditing of the sponsor’s model difficult, as it remains unclear whether errors have been overridden. For some model parameters (e.g., health state costs), the sponsor arbitrarily incorporated uncertainty as plus or minus 15% of the mean value, which does not reflect the true uncertainty around the model’s parameters possible values.
CADTH was unable to fully validate the model and notes that the results presented should be treated with a degree of caution.
Additional limitations were identified but were not considered to be key limitations:
Costs related to inclisiran administration were underestimated. The sponsor’s submission states that 10 minutes of nursing time was included per inclisiran injection; however, in the model, this cost was applied once per year. Based on the frequency of inclisiran injection, this cost should be applied 3 times in the first year and twice yearly after that.
In the CADTH base case, administration costs for inclisiran were applied based on the recommended dosing schedule.2
Additionally, the following key assumptions were made by the sponsor and have been appraised by CADTH (Table 5).
Table 5: Key Assumptions of the Submitted Economic Evaluation
Sponsor’s key assumption | CADTH comment |
|---|---|
Patients enrolled in the ORION clinical trials were assumed to be representative of patients in Canada who would be eligible for inclisiran. | Reasonable. CADTH notes that the ORION-9 trial includes patients with HeFH, ORION-10 includes patients with ASCVD, and ORION-11 includes patients with ASCVD as well as ASCVD risk-equivalent patients. As such, ORION-9 and ORION-10 are most relevant to the HeFH and ASCVD subgroups. The clinical expert consulted by CADTH for this review indicated that the characteristics of the patients in ORION-9 and ORION-10 are in line with Canadian patients with HeFH and ASCVD who would be eligible for inclisiran. |
Baseline distribution of patients with ASCVD was based on the alirocumab submission to NICE3:
| Uncertain. The clinical expert consulted by CADTH indicated that the relevance of the modelled distribution to the Canadian population is unclear. In particular, the proportion of patients with ACS in the previous 1 to 2 years was noted to be lower than expected. While the sponsor provided additional weights based on the proportion of patients in Canada with coronary heart disease, peripheral artery disease, and cerebrovascular disease,21 these weights were not qualitatively different from those employed in the sponsor’s base case owing to a series of assumptions to move from coronary heart disease, peripheral artery disease, and cerebrovascular disease to the modelled health states. CADTH was unable to assess the impact of this assumption owing to a lack of additional data about the relative proportion of patients in each ASCVD state. |
Within the HeFH subgroup, 72.6% of patients were assumed to be receiving a PCSK9 for primary prevention, based on the distribution in ORION-9. | Reasonable. The clinical expert noted that the sponsor’s assumption that the majority of HeFH patients would be receiving PCSK9 inhibitors as primary prevention was generally aligned with clinical practice and is similar to the ORION-9 patient population. |
Time since a cardiovascular event is categorized in the model as 0 to 1 year post event, 1 to 2 years post event, and > 2 years (stable). | Reasonable. The clinical expert consulted by CADTH indicated that the risk of a subsequent event is highest in the first year after a coronary event, while after 2 years, patients are considered to have chronic stable coronary artery disease. |
The minimum LDL-C level required for treatment intensification (i.e., addition of a PCSK9 inhibitor) was assumed to be 1.8 mmol/L. | Reasonable. The clinical expert consulted by CADTH indicated that 1.8 mmol/L is used as a threshold for treatment intensification for patients with established ASCVD (including patients with HeFH taking inclisiran for secondary prevention). |
The distribution of stains and doses of statins as part of SoC was based on claims data from IQVIA Pharmastat.11 The included statins were atorvastatin, fluvastatin, lovastatin, pravastatin, rosuvastatin, and simvastatin. | Inappropriate, although unlikely to have an important effect on the ICER owing to the low cost of statins. The use of claims data does not account for statins used outside the scope of the current submission (e.g., ASCVD risk–equivalent conditions). The distribution of statins in the sponsor’s submission is not aligned with that from the ORION trials. The clinical expert consulted by CADTH indicated that high-dose atorvastatin (40 mg or 80 mg daily) or rosuvastatin (20 mg or 40 mg daily) are preferred for this indication; for patients intolerant to these, lower doses or less potent statins may be considered. In the CADTH submission of alirocumab,19 SoC was assumed to comprise atorvastatin (40 mg or 80 mg daily) or rosuvastatin (20 mg or 40 mg daily) or simvastatin (80 mg daily), while SoC was assumed to comprise rosuvastatin (5 mg or 40 mg daily) in the CADTH submission of evolocumab.18 |
SoC was assumed to have no effect on LDL-C levels in patients who had previously received maximally tolerated statin therapy. | Reasonable. The clinical expert consulted by CADTH for this review indicated that additional statin treatment would not likely result in a further reduction of LDL-C among patients who require additional lowering of LDL-C despite prior treatment with maximally tolerated statin therapy. |
Adverse events were not included in the sponsor’s model. | Uncertain. The sponsor justified the exclusion of adverse events from the model based on previous CADTH submissions of evolocumab18 and alirocumab19 and because of similar rates of adverse events between inclisiran and placebo in the ORION trials. The clinical expert consulted by CADTH noted, however, that the mechanism of action is different for inclisiran compared with other PCSK9 inhibitors (silencing RNA vs. antibodies) and that adverse event profiles may differ across PCSK9 inhibitors. Injection-site reactions in the ORION trials, although generally mild to moderate, were more common among patients who received inclisiran compared with placebo. |
ACS = acute coronary syndrome; ASCVD = atherosclerotic cardiovascular disease; HeFH = heterozygous familial hypercholesterolemia; ICER = incremental cost-effectiveness ratio; LDL-C = low-density lipoprotein cholesterol; NICE = National Institute for Health and Care Excellence; PCSK9 = proprotein convertase subtilisin/kexin type 9; RNA = ribonucleic acid; SoC = standard of care.
CADTH Reanalyses of the Economic Evaluation
Several limitations with the sponsor’s submission could not be adequately addressed. CADTH could not address the lack of comparative clinical data for inclisiran versus evolocumab or alirocumab. Given the limitations with the sponsor’s NMA identified in the CADTH Clinical Review, the CADTH base case consists of pairwise comparisons of inclisiran versus SoC in both the HeFH and ASCVD subgroups and is informed by direct evidence from the ORION-9 and ORION-10 trials, respectively.
CADTH was also unable to address the effect of inclisiran on absolute LDL-C levels, the uncertainty regarding the baseline risk of cardiovascular events, and the uncertainty regarding long-term clinical effectiveness. CADTH could not fully validate the sponsor’s model owing to a lack of transparency and the poor modelling practices employed. While the effect of inclisiran on cardiovascular outcomes is uncertain owing to a lack of data, CADTH undertook reanalyses assuming a similar relationship between LDL-C reduction and cardiovascular risk as observed with evolocumab, adopting a similar approach to that employed in the CADTH review of evolocumab.18
CADTH undertook reanalyses that addressed limitations within the model, as summarized in Table 6. The CADTH base case was derived by making changes in model parameter values and assumptions, in consultation with clinical experts.
Table 6: CADTH Revisions to the Submitted Economic Evaluation
Stepped analysis | Sponsor’s value or assumption | CADTH value or assumptiona |
|---|---|---|
Corrections to sponsor’s base case | ||
None | — | — |
Changes to derive the CADTH base case | ||
1. Effectiveness estimates. | Based on sponsor-provided NMAs. | Based on direct trial evidence (HeFH data from ORION-9, ASCVD data from ORION-10). |
2. Cardiovascular risk. | The relationship between LDL-C reduction and cardiovascular risk was based on that observed with statin treatment in the CTTC meta-analyses.6 | The relationship between LDL-C reduction and cardiovascular risk was based on observed hazard ratios from the FOURIER cardiovascular outcomes trial (see Appendix 4 for additional information). |
3. Administration costs | Applied once yearly. | Applied 3 times in the first year and twice in each subsequent year. |
CADTH base case | Reanalysis 1 + 2 + 3 | |
ASCVD = atherosclerotic cardiovascular disease; CTTC = Cholesterol Treatment Trialists’ Collaboration; HeFH = heterozygous familial hypercholesterolemia; LDL-C = low-density lipoprotein cholesterol.
aApplied to both the ASCVD and HeFH subgroups, unless otherwise noted.
Base-Case Results — ASCVD Subgroup
CADTH undertook a stepped analysis, incorporating each change proposed in Table 6 into the sponsor’s base case for the ASCVD subgroup to highlight the impact of each change (Table 7; disaggregated results are presented in Appendix 4, Table 16).
In the CADTH base case, inclisiran plus SoC was associated with higher costs (incremental costs = $58,286) and higher QALYs (incremental QALYs = 0.16) than SoC alone over a 40-year horizon. The ICER for inclisiran plus SoC was $366,650 per QALY versus SoC alone. There is a 0% probability that inclisiran plus SoC is optimal compared with SoC alone at a WTP threshold of $50,000 per QALY. The incremental QALYs gained with inclisiran in the first 2 years of treatment was 0.003 (about 1 day), indicating that the majority of the incremental benefits were accrued in the post-trial period and were derived on the basis of extrapolated trial findings rather than observed benefit (while the median treatment duration in ORION-10 was 536 days, the analysis horizon in the sponsor’s model could not accommodate partial years). Drug acquisition costs for inclisiran are key drivers of the ICER, representing 105% of incremental costs (Table 16).
Table 7: Summary of the CADTH Reanalysis Results — ASCVD Subgroup
Stepped analysis | Drug | Total costs ($) | Total QALYs | ICER ($/QALYs) |
|---|---|---|---|---|
Sponsor’s base case | SoC | 20,897 | 8.60 | Reference |
Inclisiran + SoC | 84,915 | 9.41 | 79,726 | |
CADTH reanalysis 1 | SoC | 20,889 | 8.60 | Reference |
Inclisiran + SoC | 84,763 | 9.34 | 87,049 | |
CADTH reanalysis 2 | SoC | 21,177 | 8.29 | Reference |
Inclisiran + SoC | 79,230 | 8.48 | 334,790 | |
CADTH reanalysis 3 | SoC | 20,889 | 8.60 | Reference |
Inclisiran + SoC | 84,982 | 9.41 | 79,805 | |
CADTH base case (1 + 2 + 3) | SoC | 21,119 | 8.29 | Reference |
Inclisiran + SoC | 79,405 | 8.45 | 366,650 |
ASCVD = atherosclerotic cardiovascular disease; ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year; SoC = standard of care.
Note: Reanalyses are based on publicly available prices of the comparator treatments.
Base-Case Results — HeFH Subgroup
CADTH undertook a stepped analysis, incorporating each change proposed in Table 6 into the sponsor’s base case for the HeFH subgroup to highlight the impact of each change (Table 8; disaggregated results are presented in Appendix 4, Table 17).
In CADTH’s base case, inclisiran plus SoC was associated with higher costs (incremental costs = $95,943) and higher QALYs (incremental QALYs = 0.13) than SoC over a 40-year horizon in the HeFH subgroup. The ICER for inclisiran was $626,458 per QALY. There is a 0% probability that inclisiran plus SoC is optimal compared with SoC alone at a WTP threshold of $50,000 per QALY. The incremental QALYs with inclisiran plus SoC in the first 2 years of treatment was 0.002 (less than 1 day), indicating that the majority of the incremental benefits were derived on the basis of extrapolated trial findings rather than observed benefit. Drug acquisition costs for inclisiran are key drivers of the ICER, representing 102% of incremental costs (Table 17).
Table 8: Summary of the CADTH Reanalysis Results — HeFH Subgroup
Stepped analysis | Drug | Total costs ($) | Total QALYs | Sequential ICER ($/QALYs) |
|---|---|---|---|---|
Sponsor’s base case | SoC | 12,574 | 17.15 | Reference |
Inclisiran + SoC | 110,196 | 17.70 | 175,172 | |
Alirocumab + SoC | 124,293 | 17.74 | Extended dominance | |
Evolocumab + SoC | 124,351 | 17.78 | 176,886 | |
CADTH reanalysis 1 | SoC | 12,574 | 17.15 | Reference |
Inclisiran + SoC | 110,189 | 17.69 | 180.278 | |
CADTH reanalysis 2 | SoC | 12,813 | 17.14 | Reference |
Inclisiran + SoC | 108,706 | 17.29 | Extended dominance | |
Evolocumab + SoC | 121,973 | 17.32 | 598,625 | |
Alirocumab + SoC | 122,088 | 17.31 | Dominated | |
CADTH reanalysis 3 | SoC | 12,574 | 17.15 | Reference |
Inclisiran + SoC | 110,305 | 17.70 | 175,368 | |
Alirocumab + SoC | 124,293 | 17.74 | Extended dominance | |
Evolocumab + SoC | 124,351 | 17.78 | 175,497 | |
CADTH base case (1 + 2 + 3) | SoC | 12,813 | 17.14 | Reference |
Inclisiran + SoC | 108,756 | 17.29 | 626,458 |
HeFH = heterozygous familial hypercholesterolemia; ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year; SoC = standard of care.
Note: Reanalyses are based on the publicly available prices of the comparator treatments.
Scenario Analysis Results
A price reduction analysis was performed based on the sponsor’s base case and CADTH’s reanalysis for the ASCVD population (Table 9) and the HeFH population (Table 10). For the HeFH subgroup, only the pairwise comparison against SoC for the sponsor’s base case is reported for comparability to the CADTH reanalysis. The results presented in Table 9 indicate that a price reduction of 83% is required to make inclisiran cost-effective at a WTP of $50,000 compared with SoC in the ASCVD subgroup. In the HeFH subgroup, a 91% price reduction would be required to make inclisiran cost-effective compared with SoC (Table 10). As noted previously, the limitations within the clinical evidence and the sponsor’s use of poor modelling practices mean that the price reduction estimates are uncertain and may underestimate the true price reduction needed for inclisiran to reach this WTP threshold.
Table 9: CADTH Price Reduction Analyses — ASCVD Subgroup
Analysis | ICERs for inclisiran vs. SoC ($/QALY) | |
|---|---|---|
Price reduction | Sponsor’s base case | CADTH reanalysis |
No price reduction | 79,726 | 366,650 |
10% | 71,199 | 329,947 |
20% | 62,891 | 291,512 |
30% | 54,364 | 252,412 |
40% | 46,162 | 217,277 |
50% | NA | 173,237 |
60% | NA | 134,295 |
70% | NA | 96,927 |
80% | NA | 58,900 |
83% | NA | 47,248 |
ASCVD = atherosclerotic cardiovascular disease; ICER = incremental cost-effectiveness ratio; NA = not applicable; SoC = standard of care.
Table 10: CADTH Price Reduction Analyses — HeFH Subgroup
Analysis | ICERs for inclisiran vs. SoC ($/QALY) | |
|---|---|---|
Price reduction | Sponsor’s base case | CADTH reanalysis |
No price reduction | 175,172 | 626,457 |
10% | 159,041 | 562,426 |
20% | 139,558 | 498,395 |
30% | 120,007 | 434,363 |
40% | 104,174 | 370,332 |
50% | 86,597 | 300,342 |
60% | 68,300 | 238,728 |
70% | 49,123 | 174,215 |
80% | NA | 112,877 |
90% | NA | 50,526 |
91% | NA | 43,745 |
HeFH = heterozygous familial hypercholesterolemia; ICER = incremental cost-effectiveness ratio; NA = not applicable; SoC = standard of care.
Several scenario and sensitivity analyses were conducted on the CADTH base case for the ASCVD and HeFH subgroups (Table 18). These scenario analyses explored the impact of the following model parameters and assumptions: the relationship between LDL-C reduction and cardiovascular risk, adopting alternative inclisiran effect estimates, including evolocumab and alirocumab as comparators, and adopting a 20-year analysis horizon.
Of these, the ICER was most notably reduced in both subgroups when the relationship between LDL-C reduction and cardiovascular risk was based on RRs from the CTTC meta-analysis6 (Table 19, Table 20). This highlights that the use of a surrogate outcome, LDL-C, is a key driver in the model. As noted previously, the CTTC meta-analysis6 assessed the relationship between statins and cardiovascular risk and may not be appropriate for modelling the relationship between PCSK9 inhibitors and cardiovascular risk.
Issues for Consideration
CADTH notes that no PCSK9 inhibitors are currently reimbursed by public drug plans in Canada for the treatment of ASCVD. While evolocumab and alirocumab received conditional recommendations from the CADTH Canadian Drug Expert Committee,22,23 negotiations with the Pan-Canadian Pharmaceutical Alliance concluded without agreement on either drug. As such, should inclisiran become reimbursed, it would be the first PCSK9 inhibitor publicly reimbursed for patients with ASCVD in Canada. This is in contrast with the HeFH population, where evolocumab and alirocumab are both currently publicly reimbursed. CADTH notes that the estimated budget impact of reimbursing inclisiran is considerably higher for the ASCVD subgroup and is likely above the threshold considered affordable by the public drug plans.
Genetic testing to determine whether a patient has possible, probable, or definite familial hypercholesterolemia (i.e., HeFH or homozygous familial hypercholesterolemia) is not widely available in clinical practice, according to the clinical expert consulted by CADTH for this review and as noted in the 2018 Canadian Cardiovascular Society’s position statement on familial hypercholesterolemia.24
The patient input received for this review notes that some patients may prefer treatment that can be administered less frequently. Inclisiran is a twice-yearly injection, compared with bimonthly or monthly injections for evolocumab and alirocumab, and daily administration for statins.
Overall Conclusions
Inclisiran reduces LDL-C compared with SoC in patients with ASCVD or HeFH, although the comparative effects of inclisiran relative to other PCSK9 inhibitors (i.e., evolocumab, alirocumab) are highly uncertain owing to a lack of direct comparative evidence and limitations within the sponsor’s NMAs. Whether the observed reduction in LDL-C in ASCVD and HeFH patients in the ORION trials results in reduced cardiovascular events is highly uncertain. The long-term effects of inclisiran on LDL-C and cardiovascular risk are highly uncertain, and the effects of inclisiran on health-related quality of life in patients with ASCVD or HeFH are unknown, as health-related quality of life was not assessed in the ORION trials.
CADTH undertook analyses to address limitations in the sponsor’s submission, including assuming that the relationship between LDL-C reduction and cardiovascular risk is similar to that observed with other PCSK9 inhibitors (i.e., evolocumab). CADTH was unable to address the high level of uncertainty associated with the comparative clinical evidence for inclisiran versus other PCSK9 inhibitors; as such, the CADTH reanalyses comprised a pairwise analysis of inclisiran plus SoC compared with SoC alone. These analyses were informed by direct evidence from the ORION-9 and ORION-10 trials for the HeFH subgroup and the ASCVD subgroups, respectively. CADTH was unable to address the absolute changes in LDL-C, the uncertainty regarding the baseline risk of cardiovascular events, and the uncertainty regarding long-term clinical effectiveness.
In the CADTH base case, inclisiran plus SoC is not cost-effective compared with SoC alone at a WTP threshold of $50,000 in either the ASCVD or HeFH subgroups. Specifically, inclisiran plus SoC was associated with an ICER of $366,650 per QALY gained compared with SoC among ASCVD patients (incremental costs = $58,286; incremental QALYs = 0.16), and $626,458 per QALY compared with SoC among HeFH patients (incremental costs = $95,943; incremental QALYs = 0.15). Inclisiran was considered optimal at a WTP of $50,000 in 0% of replications in either subgroup. The key driver of the ICER in both subgroups is the cost of inclisiran acquisition, and price reduction analyses suggest that, in the ASCVD subgroup, an 83% price reduction would be required for inclisiran to be considered optimal at a WTP threshold of $50,000, while a 91% price reduction would be required for it to be considered optimal in the HeFH subgroup. This price reduction may be conservative given that the cost-effectiveness estimate is reliant on achieving a reduction in cardiovascular risk and in maintaining long-term treatment benefit, which have not been shown in clinical trials.
A key driver of the model is the use of a surrogate outcome, LDL-C, to approximate the effect of inclisiran on the risk of cardiovascular events. Data from the submitted pivotal trials (ORION-9, ORION,10, ORION-11) were not powered to show that inclisiran reduces the risk of cardiovascular events, and the CADTH base-case results are highly sensitive to the use of LDL-C as a surrogate marker. The approach of using LDL-C reduction as a surrogate marker for cardiovascular risk has been adopted in previous CADTH submissions of PCSK9 inhibitors18,19; however, this approach has been criticized as potentially overestimating the reduction in risk when compared with clinical trials designed to assess the effect of treatment on cardiovascular outcomes.18 The results of ORION-4, the ongoing cardiovascular outcomes trial of inclisiran, are not yet available. In the absence of such data, CADTH was unable to validate the survival benefit of inclisiran use predicted by the sponsor’s model. Based on the input of the clinical expert consulted by CADTH for this review, in the CADTH reanalyses, the reduction in cardiovascular risk due to LDL-C lowering was based on the risk reduction observed with evolocumab treatment in the FOURIER trial, a cardiovascular outcomes trial designed and powered to assess the effect of evolocumab on cardiovascular outcomes. The notable increase in the ICER in both subgroups with this change highlights the uncertainty associated with the use of LDL-C lowering as a surrogate outcome in the model. As such, the CADTH base-case results may be more uncertain than suggested by the probabilistic analyses.
Overall, it is highly unlikely that inclisiran would be considered cost-effective at a $50,000 per QALY threshold, even if a substantial price reduction were obtained.
References
1.Pharmacoeconomic evaluation [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Leqvio (inclisiran), 284 mg in 1.5 mL (189 mg/mL) solution for subcutaneous injection. Dorval (QC): Novartis Pharmaceuticals Canada Inc; 2021 Mar 17.
2.Leqvio (inclisiran): 284 mg in 1.5 mL (189 mg/mL) solution for subcutaneous injection [product monograph]. Dorval (QC): Novartis Pharmaceuticals Canada Inc; 2021 Jul 23.
3.National Institute for Health and Care Excellence. Alirocumab for treating primary hypercholesterolaemia and mixed dyslipidaemia (Technology appraisal guidance TA393) 2016; https://www.nice.org.uk/guidance/ta393. Accessed 2021 Apr 13.
4.Mohrschladt MF, Westendorp RG, Gevers Leuven JA, Smelt AH. Cardiovascular disease and mortality in statin-treated patients with familial hypercholesterolemia. Atherosclerosis. 2004;172(2):329-335. PubMed
5.Cholesterol Treatment Trialists’ (CTT) Collaboration, Mihaylova B, Emberson J, et al. The effects of lowering LDL cholesterol with statin therapy in people at low risk of vascular disease: meta-analysis of individual data from 27 randomised trials. Lancet. 2012;380(9841):581-590. PubMed
6.Armitage J, Baigent C, Barnes E, et al. Efficacy and safety of statin therapy in older people: a meta-analysis of individual participant data from 28 randomised controlled trials. Lancet. 2019;393(10170):407-415. PubMed
7.Stoekenbroek RM, Hartgers ML, Rutte R, de Wijer DD, Stroes ESG, Hovingh GK. PCSK9 inhibitors in clinical practice: delivering on the promise? Atherosclerosis. 2018;270:205-210. PubMed
8.Ara R, Brazier JE. Populating an economic model with health state utility values: moving toward better practice. Value Health. 2010(5):509-518. PubMed
9.National Institute for Health and Care Excellence. Alirocumab for treating primary hypercholesterolaemia and mixed dyslipidaemia. Pt 3: evidence. (Technology appraisal guidance TA393) 2016; https://www.nice.org.uk/guidance/ta393/chapter/3-Evidence#ergs-revised-exploratory-analyses. Accessed 2021 Apr 28.
10.Ontario Ministry of Health, Ontario Ministry of Long-Term Care. Ontario drug benefit formulary/comparative drug index. 2020; https://www.formulary.health.gov.on.ca/formulary/. Accessed 2021 May 31.
11.Compuscript national TRx ext units from Jan 2019-Nov 2020. Ottawa (ON): IQVIA 2020.
12.Government of Canada. Summary: registered nurse (RN) in Canada. 2021.
13.Third-line pharmacotherapy for type 2 diabetes — update. (CADTH optimal use report vol.3, no1b). Ottawa (ON): CADTH; 2013.
14.Padwal RS, So H, Wood PW, et al. Cost-effectiveness of home blood pressure telemonitoring and case management in the secondary prevention of cerebrovascular disease in Canada. J Clin Hypertens (Greenwich). 2019;21(2):159-168. PubMed
15.Tanuseputro P, Wodchis WP, Fowler R, et al. The health care cost of dying: a population-based retrospective cohort study of the last year of life in Ontario, Canada. PLoS One. 2015;10(3):e0121759. PubMed
16.Tran DT, Welsh RC, Ohinmaa A, Thanh NX, Kaul P. Resource use and burden of hospitalization, outpatient, physician, and drug costs in short- and long-term care after acute myocardial infarction. Can J Cardiol. 2018;34(10):1298-1306. PubMed
17.Zhang Z. Cost-effectiveness of proprotein convertase subtilisin/kexin type 9 inhibitors: an economic evaluation (unpublished master's thesis). Calgary (AB): University of Calgary; 2017: https://prism.ucalgary.ca/handle/11023/3697. Accessed 2021 Apr 21.
18.CADTH common drug review: pharmacoeconomic review report (resubmission) of evolocumab (Repatha- Amgen Canada Inc). Ottawa (ON): CADTH; 2017: https://cadth.ca/sites/default/files/cdr/pharmacoeconomic/SR0515_Repatha_Resubmission_PE_Report.pdf. Accessed 2021 Aug 4.
19.CADTH common drug review: pharmacoeconomic review report of alirocumab (Praluent - Sanofi‐Aventis Canada Inc.). Ottawa (ON): CADTH; 2016: https://cadth.ca/sites/default/files/cdr/pharmacoeconomic/SR0469_Praluent_PE_Report.pdf Accessed 2021 Aug 4.
20.Sabatine MS, Giugliano RP, Keech AC, et al. Evolocumab and clinical outcomes in patients with cardiovascular disease. N Engl J Med. 2017;376(18):1713-1722. PubMed
21.Blais C, Rochette L, Ouellet S, Huynh T. Complex evolution of epidemiology of vascular diseases, including increased disease burden: from 2000 to 2015. Can J Cardiol. 2020;36(5):740-746. PubMed
22.CADTH Canadian Drug Expert Committee (CDEC) final recommendation: alirocumab (Praluent - Sanofi‐Aventis Canada Inc.). Ottawa (ON): CADTH; 2016 Jul 20: https://www.cadth.ca/sites/default/files/cdr/complete/SR0469_complete_Praluent_Jul-20-16.pdf. Accessed 2021 May 26.
23.CADTH Canadian Drug Expert Committee (CDEC) final recommendation: evolocumab (Repatha- Amgen Canada Inc). Ottawa (ON): CADTH; 2017 Nov 22: https://www.cadth.ca/sites/default/files/cdr/complete/SR0515_Repatha_Resubmission_complete_Nov_24_17.pdf. Accessed 2021 Mar 26.
24.Brunham LR, Ruel I, Aljenedil S, et al. Canadian Cardiovascular Society position statement on familial hypercholesterolemia: update 2018. Can J Cardiol. 2018;34(12):1553-1563. PubMed
25.Drug Reimbursement Review sponsor submission: Leqvio (inclisiran), 284 mg in 1.5 mL (189 mg/mL) solution for subcutaneous injection [internal sponsor's package]. Dorval (QC): Novartis Pharmaceuticals Canada Inc; 2021 Mar 17.
26.CADTH common drug review: pharmacoeconomic review report of icosapent ethyl (Vascepa- HLS Therapeutics). 2020: https://www.cadth.ca/sites/default/files/cdr/pharmacoeconomic/sr0619-vascepa-pharmacoeconomic-review-report.pdf. Accessed 2021 May 4.
27.Budget Impact Analysis [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Leqvio (inclisiran), 284 mg in 1.5 mL (189 mg/mL) solution for subcutaneous injection. Dorval (QC): Novartis Pharmaceuticals Canada Inc; 2021 Mar 17.
28.PharmaStat. Ottawa (ON): IQVIA; 2021: https://www.iqvia.com/. Accessed 2021 Mar 31.
29.Statistics Canada. Projected population, by projection scenario, age and sex, as of July 1 (x 1,000). 2019.
30.Akioyamen LE, Genest J, Shan SD, et al. Estimating the prevalence of heterozygous familial hypercholesterolaemia: a systematic review and meta-analysis. BMJ Open. 2017;7(9):e016461. PubMed
31.de Ferranti SD, Rodday AM, Mendelson MM, Wong JB, Leslie LK, Sheldrick RC. Prevalence of familial hypercholesterolemia in the 1999 to 2012 United States National Health and Nutrition Examination Surveys (NHANES). Circulation. 2016;133(11):1067-1072. PubMed
32.Brunham LR, Cermakova L, Lee T, et al. Contemporary trends in the management and outcomes of patients with familial hypercholesterolemia in Canada: a prospective observational study. Can J Cardiol. 2017;33(3):385-392. PubMed
33.Chen G, Farris MS, Cowling T, et al. Treatment and low-density lipoprotein cholesterol management in patients diagnosed with clinical atherosclerotic cardiovascular disease in Alberta. Can J Cardiol. 2019;35(7):884-891. PubMed
34.American College of Cardiology. ASCVD patients face barriers reaching guideline-recommended cholesterol goals. 2019; https://www.acc.org/latest-in-cardiology/articles/2019/11/09/08/42/nov11-5am-ascvd-patients-face-barriers-aha-2019. Accessed 2021 Feb.
35.Dinh T, Sutherland G. Understanding the gap: a pan-Canadian analysis of prescription drug insurance coverage. Ottawa (ON): The Conference Board of Canada; 2017: https://www.conferenceboard.ca/e-library/abstract.aspx?did=9326. Accessed 2021 Jun 10.
36.Repatha – hypercholestérolémie familiale homozygote et hétérozygote Montreal (QC): INESSS; 2016: https://www.inesss.qc.ca/fileadmin/doc/INESSS/Inscription_medicaments/Avis_au_ministre/Aout_2016/Repatha_HFHo_et_HFHe_2016_06.pdf. Accessed 2021 May 25.
Appendix 1: Cost Comparison Table
Note that this appendix has not been copy-edited.
The comparators presented in the following table have been deemed to be appropriate based on feedback from clinical expert(s). Comparators may be recommended (appropriate) practice or actual practice. Existing Product Listing Agreements are not reflected in the table and as such, the table may not represent the actual costs to public drug plans.
Table 11: CADTH Cost Comparison Table for the Treatment of Primary Hyperlipidemia for Patients With HeFH or ASCVD
Treatment | Strength / concentration | Form | Price ($) | Recommended dosage | Daily cost ($) | Annual cost ($) |
|---|---|---|---|---|---|---|
Inclisiran | 189 mg/mL | Single-use, pre-filled syringe (284 mg/1.5 mL) | 2,839.2800a | 284 mg administered initially, at month 3, and subsequently every 6 months | Initial year: 23.34 Subsequent years: 15.56 | Initial year: 8,518 Subsequent years: 5,679 |
Anti-PCSK9 monoclonal antibody | ||||||
Evolocumab (Repatha) | 120 mg/mL 140 mg/mL | Single-use pre-filled cartridge (420 mg/3.5 mL) Single-use pre-filled syringe or autoinjector (140 mg/1 mL) | 564.5500 (per cartridge) 260.5650 per autoinjector or pen | 140 mg every 2 weeks or 420 mg monthly | 18.56 | 6,775 |
Alirocumab (Praluent) | 75 mg/mL 150 mg/mL | Single-use pre-filled pen or syringe | 256.2100 per pen or syringe | 75 mg once every 2 weeks or 300 mg once every 4 weeks | 18.25 | 6,661 |
HMG-CoA reductase inhibitors (statins) | ||||||
Atorvastatin calcium (Lipitor, generics) | 10 mg 20 mg 40 mg 80 mg | Tab | 0.1743 0.2179 0.2342 0.2342 | 10 to 80 mg daily | 0.17 to 0.23 | 64 to 85 |
Fluvastatin sodium | 20 mg 40 mg | Cap | 0.6882 0.9671 | 20 to 80 mg daily | 0.69 to 1.93 | 251 to 706 |
Fluvastatin sodium (Lescol XL) | 80 mg | Tab | 1.6225 | 80 mg daily | 1.60 | 592 |
Lovastatin | 20 mg 40 mg | Tab | 0.4919 0.8985 | 20 to 80 mg daily | 0.49 to 0.90 | 180 to 656 |
Pravastatin sodium | 10 mg 20 mg 40 mg | Tab | 0.2916 0.3440 0.4143 | 20 to 80 mg daily | 0.34 to 0.83 | 126 to 302 |
Rosuvastatin calcium (Crestor, generics) | 5 mg 10 mg 20 mg 40 mg | Tab | 0.1284 0.1354 0.1692 0.1990 | 10 to 40 mg daily | 0.14 to 0.20 | 49 to 73 |
Simvastatin (Zocor, generics) | 5 mg 10 mg 20 mg 40 mg 80 mg | Tab | 0.1023 0.2023 0.2501 0.2501 0.2501 | 10 to 40 mg daily | 0.20 to 0.25 | 74 to 91 |
Cholesterol absorption inhibitor | ||||||
Ezetimibe (Ezetrol) | 10 mg | Tab | 0.1811 | 10 mg daily | 0.18 | 66 |
Lipid-regulating agent | ||||||
Icosapent ethyl (Vascepa) | 1 g | Cap | 2.4500b | 2 g twice daily | 9.80 | 3,577 |
Fibrates | ||||||
Bezafibrate (Bezalip, generic) | 400 mg | Tab | 1.7460 | 400 mg daily | 1.75 | 637 |
Fenofibrate (generic) | 67 mg 100 mg 200 mg | Cap | 0.5479 0.6105 0.2723 | 67 to 200 mg daily | 0.27 to 2.4 | 99 to 891 |
Fenofibrate (Lipidil EZ) | 48 mg 145 mg | Tab | 0.3560 0.5489 | 48 to 145 mg daily | 0.36 to 0.55 | 130 to 200 |
Gemfibrozil (generic) | 300 mg | Cap | 0.1340 | 600 mg twice daily | 0.27 | 49 |
Micro-coated fenofibrate (Lipidil Supra, generics) | 160 mg | Tab | 0.3116 | 160 mg daily | 0.31 | 114 |
PCSK9 = proprotein convertase subtilisin/kexin type 9.
Note: All prices are from the Ontario Drug Benefit Formulary (accessed May 2021), unless otherwise indicated, and do not include dispensing fees.
aSponsor’s submitted price.25
bCADTH Pharmacoeconomic report for icosapent ethyl, sponsor’s submitted price.26
Appendix 2: Submission Quality
Note that this appendix has not been copy-edited.
Description | Yes/No | Comments |
|---|---|---|
Population is relevant, with no critical intervention missing, and no relevant outcome missing | Yes | |
Model has been adequately programmed and has sufficient face validity | No | Poor modelling practices were employed (see main text). Owing to the poor modelling practices, CADTH was unable to fully validate the sponsor’s model. |
Model structure is adequate for decision problem | Yes | |
Data incorporation into the model has been done adequately (e.g., parameters for probabilistic analysis) | Yes | |
Parameter and structural uncertainty were adequately assessed; analyses were adequate to inform the decision problem | Yes | |
The submission was well organized and complete; the information was easy to locate (clear and transparent reporting; technical documentation available in enough details) | Yes |
Appendix 3: Additional Information on the Submitted Economic Evaluation
Detailed Results of the Sponsor’s Base Case
Table 13: Disaggregated Summary of the Sponsor’s Economic Evaluation Results — ASCVD Subgroup
Parameter | Inclisiran + SoC | SoC | Incremental |
|---|---|---|---|
Discounted LYs | |||
Total | 13.57 | 12.47 | 1.10 |
Discounted QALYs | |||
Total | 9.41 | 8.60 | 0.80 |
By Health State | |||
Initial | 6.57 | 5.34 | 1.23 |
Post Unstable Angina | 0.51 | 0.60 | −0.09 |
Post Myocardial Infarction | 0.57 | 0.71 | −0.15 |
Post Stroke | 0.57 | 0.66 | −0.10 |
Post Revascularization | 1.20 | 1.30 | −0.10 |
Discounted costs ($) | |||
Total | 84,915 | 20,897 | 64,017 |
Drug Acquisition | 68,747 | 728 | 68,019 |
SoC | 793 | 728 | 64 |
Intervention | 67,881 | 0 | 67,881 |
Administration | 73 | 0 | 73 |
Cardiovascular Events | 16,168 | 20,169 | −4,001 |
ICER ($/QALY) | 79,726 | ||
ASCVD = atherosclerotic cardiovascular disease; ICER = incremental cost-effectiveness ratio; LY = life-year; QALY = quality-adjusted life-year; SoC = standard of care; vs. = versus.
Source: Sponsor’s pharmacoeconomic submission.1
Table 14: Disaggregated Summary of the Sponsor’s Economic Evaluation Results — HeFH Subgroup
Treatment | Component | Value | Incremental (vs. SoC) | Incremental (sequential) |
|---|---|---|---|---|
Discounted LYs | ||||
SoC | Total | 21.91 | NA | NA |
Inclisiran + SoC | Total | 22.59 | 0.68 | NA |
Alirocumab + SoC | Total | 22.64 | 0.73 | 0.05 |
Evolocumab + SoC | Total | 22.69 | 0.78 | 0.05 |
Discounted QALYs | ||||
SoC | Initial Health State | 14.58 | NA | NA |
Post Unstable Angina | 0.95 | NA | NA | |
Post MI | 0.75 | NA | NA | |
Post Stroke | 0.38 | NA | NA | |
Post Revascularization | 0.50 | NA | NA | |
Total | 17.15 | NA | NA | |
Inclisiran + SoC | Initial Health State | 15.64 | 1.06 | NA |
Post Unstable Angina | 0.78 | −0.17 | NA | |
Post MI | 0.55 | −0.2 | NA | |
Post Stroke | 0.28 | −0.1 | NA | |
Post Revascularization | 0.45 | −0.05 | NA | |
Total | 17.70 | 0.55 | NA | |
Alirocumab + SoC | Initial Health State | 15.72 | 1.14 | 0.08 |
Post Unstable Angina | 0.77 | −0.18 | −0.01 | |
Post MI | 0.54 | −0.21 | −0.01 | |
Post Stroke | 0.27 | −0.11 | −0.01 | |
Post Revascularization | 0.45 | −0.05 | 0 | |
Total | 17.74 | 0.59 | 0.04 | |
Evolocumab + SoC | Initial Health State | 15.81 | 1.23 | 0.09 |
Post Unstable Angina | 0.75 | −0.2 | −0.02 | |
Post MI | 0.52 | −0.23 | −0.02 | |
Post Stroke | 0.26 | −0.12 | −0.01 | |
Post Revascularization | 0.44 | −0.06 | −0.01 | |
Total | 17.78 | 0.63 | 0.04 | |
Discounted costs ($) | ||||
SoC | Drug Acquisition | 1,280 | NA | NA |
SoC | 1,280 | NA | NA | |
Intervention | 0 | NA | NA | |
Administration | 0 | NA | NA | |
Cardiovascular Events | 11,294 | NA | NA | |
Total | 12,574 | NA | NA | |
Inclisiran + SoC | Drug Acquisition | 101,935 | 100,655 | NA |
SoC | 1,319 | 39 | NA | |
Intervention | 100,616 | 100,616 | NA | |
Administration | 110 | 110 | NA | |
Cardiovascular Events | 8,151 | −3,143 | NA | |
Total | 110,196 | 97,622 | NA | |
Alirocumab + SoC | Drug Acquisition | 116,355 | 115,075 | 14,420 |
SoC | 1,322 | 42 | 3 | |
Intervention | 115,033 | 115,033 | 14,417 | |
Administration | 0 | 0 | −110 | |
Cardiovascular Events | 7,938 | −3,356 | −213 | |
Total | 124,294 | 111,720 | 14,098 | |
Evolocumab + SoC | Drug Acquisition | 116,622 | 115,342 | 267 |
SoC | 1,325 | 45 | 3 | |
Intervention | 115,297 | 115,297 | 264 | |
Administration | 0 | 0 | 0 | |
Cardiovascular Events | 7,729 | −3,565 | −209 | |
Total | 124,351 | 111,777 | 57 | |
Treatment | ICER vs. reference ($) | Sequential ICER ($) | ||
SoC | Reference | Reference | ||
Inclisiran + SoC | 175,172 | 175,172 | ||
Alirocumab + SoC | 187,049 | Extended dominance | ||
Evolocumab + SoC | 175,384 | 176,886 | ||
HeFH = heterozygous familial hypercholesterolemia; ICER = incremental cost-effectiveness ratio; MI = myocardial infarction; LY = life-year; QALY = quality-adjusted life-year; SoC = standard of care; vs. = versus.
Source: Sponsor’s pharmacoeconomic submission.1
Appendix 4: Additional Details on the CADTH Reanalyses and Sensitivity Analyses of the Economic Evaluation
Note that this appendix has not been copy-edited.
Relationship Between LDL-C and Cardiovascular Outcomes
In the sponsor’s model,1 the RRs of PCSK9 inhibitors for each cardiovascular event was calculated based on RR per 1 mmol/L LDL-C reduction. As such, the observed hazard ratios (HRs) from the FOURIER trial could not be directly used in the model. The following approach was used to convert the observed FOURIER HRs to RR per 1 mmol/L reduction in LDL-C, consistent with the approach used in the CADTH Common Drug Review reanalysis of the evolocumab submission.18
An absolute LDL reduction of 1.45 mmol/L (56.18 mg/dL) was observed in ORION-10, while an absolution reduction of 1.53 mmol/L (58.95 mg/dL) was observed in ORION-9. These values were applied for the ASCVD and HeFH populations, respectively. In order for the observed HRs from the FOURIER trial to be used in the economic model, the HR for each cardiovascular event was converted to RR per 1 mmol LDL-C reduction by taking the inverse power of the absolute LDL-C reduction.
To convert the observed HR from FOURIER to RR per mmol/L, the following equations were used:
Total RR = RR per mmol/L to the power of absolute LDL-C reduction from inclisiran
For example: RR revascularization (ASCVD subgroup) = 0.78 ^ (1/1.45) = 0.84.
The converted RRs used in the CADTH reanalysis are shown in Table 15.
Table 15: Relative Risk per 1 mmol/L LDL-C Reduction Based on the FOURIER Trial
Parameter | Original model values;a | Observed HR from FOURIER trial; | Converted RR per mmol/L; | |
|---|---|---|---|---|
ASCVD population | HeFH population | |||
Revascularization | 0.75 (0.72 to 0.78) | 0.78 (0.71 to 0.86) | 0.84 (0.79 to 0.90) | 0.85 (0.80 to 0.91) |
Unstable angina | 0.73 (0.70 to 0.76) | NA | 0.84 (0.79 to 0.90)b | 0.85 (0.80 to 0.91)b |
Non-fatal MI | 0.73 (0.70 to 0.76) | 0.73 (0.65 to 0.82) | 0.81 (0.74 to 0.87) | 0.81 (0.75 to 0.88) |
Ischemic stroke | 0.79 (0.74 to 0.85) | 0.75 (0.62 to 0.92) | 0.82 (0.72 to 0.94) | 0.83 (0.73 to 0.95) |
Cardiovascular death | 0.84 (0.80 to 0.88) | 1.05 (0.88 to 1.25) | 1.00c | 1.00c |
ASCVD = atherosclerotic cardiovascular disease; CI = confidence interval; HeFH = heterozygous familial hypercholesterolemia; HR = hazard ratio; NR = not reported; RR = rate ratio.
aOriginal model values1 were based on the Cholesterol Treatment Trialists’ Collaboration’s 2019 meta-analysis,6 excluding trials that exclusively enrolled patients with heart failure or undergoing renal dialysis.
bUnstable angina was not an outcome in the FOURIER trial. The clinical expert consulted by CADTH for this review indicated that the risk of revascularization approximates that of unstable angina, although it is noted that there is no standardized definition of unstable angina.
cThe RR for cardiovascular death was set at 1 in the model, consistent with the approach adopted in the CADTH reanalysis of the evolocumab submission.18 CADTH notes that this assumption reduces the uncertainty associated with this parameter.
Detailed Results of CADTH Base Case
Table 16: Disaggregated Summary of CADTH’s Economic Evaluation Results — ASCVD Subgroup
Parameter | Inclisiran + SoC | SoC | Incremental |
|---|---|---|---|
Discounted LYs | |||
Total | 12.14 | 11.99 | 0.15 |
Discounted QALYs | |||
Total | 8.45 | — | 0.16 |
By Health State | |||
Initial | 5.62 | 5.07 | 0.55 |
Post Unstable Angina | 0.54 | 0.60 | −0.06 |
Post Myocardial Infarction | 0.57 | 0.71 | −0.13 |
Post Stroke | 0.50 | 0.63 | −0.12 |
Post Revascularization | 1.22 | 1.30 | −0.08 |
Discounted costs ($) | |||
Total | 79,405 | 21,119 | 58,286 |
Drug Acquisition | |||
SoC | 709 | 700 | 9 |
Intervention | 61,172 | 0 | 61,172 |
Administration | 131 | 0 | 131 |
Cardiovascular Events | 17,393 | 20,419 | −3,027 |
ICER ($/QALY) | 366,650 | ||
ASCVD = atherosclerotic cardiovascular disease; ICER = incremental cost-effectiveness ratio; LY = life-year; QALY = quality-adjusted life-year; SoC = standard of care.
Table 17: Disaggregated Summary of CADTH’s Economic Evaluation Results — HeFH Subgroup
Parameter | Inclisiran + SoC | SoC | Incremental |
|---|---|---|---|
Discounted LYs | |||
Total | 22.05 | 21.92 | 0.13 |
Discounted QALYs | |||
Total | 17.29 | 17.14 | 0.15 |
Initial | 14.93 | 14.35 | 0.58 |
Post Unstable Angina | 0.94 | 1.05 | −0.11 |
Post Myocardial Infarction | 0.62 | 0.79 | −0.18 |
Post Stroke | 0.30 | 0.40 | −0.10 |
Post Revascularization | 0.51 | 0.55 | −0.04 |
Discounted costs ($) | |||
Total | 108,756 | 12,813 | 95,943 |
Drug Acquisition | |||
SoC | 1,288 | 1,281 | 8 |
Intervention | 98,065 | 0 | 98,065 |
Drug Administration | 213 | 0 | 213 |
Cardiovascular Events | 9,189 | 11,533 | −2,343 |
ICER ($/QALY) | 626,458 | ||
HeFH = heterozygous familial hypercholesterolemia; ICER = incremental cost-effectiveness ratio; LY = life-year; NA = not applicable; QALY = quality-adjusted life-year; SoC = standard of care; vs. = versus.
Scenario Analyses
Table 18: CADTH Scenario Analyses
Parameters | CADTH Base Case | CADTH Scenario |
|---|---|---|
Scenario Analyses | ||
1. Relationship between LDL-C reduction and cardiovascular risk | Based on observed data from the FOURIER cardiovascular outcomes trial. | Based on CTTC meta-analyses for the relationship between the use of statins and cardiovascular risk, as provided in the sponsor’s submission.1 |
2. Effectiveness estimates (LDL-C reduction) | Based on direct evidence (HeFH: ORION-9; ASCVD: ORION-10), washout-imputed values. | Based on direct evidence (HeFH: ORION-9; ASCVD: ORION-10), observed values. |
3. Effectiveness estimates (LDL-C reduction) | Based on direct evidence (HeFH: ORION-9; ASCVD: ORION-10), washout-imputed values. | Based on indirect evidence from the sponsor’s NMA. |
4. Comparators | SoC | SoC |
5. Horizon | 40 years (lifetime) | 20 years |
ASCVD = atherosclerotic cardiovascular disease; CTTC = Cholesterol Treatment Trialists’ Collaboration; HeFH = heterozygous familial hypercholesterolemia; LDL-C = low-density lipoprotein cholesterol; SoC = standard of care.
Table 19: CADTH Scenario Analyses Results — ASCVD Subgroup
Drug | Total Costs ($) | Total QALYs | ICER ($/QALY) |
|---|---|---|---|
CADTH base case | |||
SoC | 21,119 | 8.29 | Reference |
Inclisiran + SoC | 79,405 | 8.45 | 366,650 |
Scenario 1: Relationship between LDL-C reduction and cardiovascular risk | |||
SoC | 20,820 | 8.59 | Reference |
Inclisiran + SoC | 84,706 | 9.32 | 87,461 |
Scenario 2: Effectiveness estimates based on observed values, ORION-10 | |||
SoC | 21,109 | 8.29 | Reference |
Inclisiran + SoC | 79,424 | 8.45 | 366,326 |
Scenario 3: Effectiveness estimates based on indirect evidence | |||
SoC | 21,145 | 8.29 | Reference |
Inclisiran + SoC | 79,448 | 8.45 | 363,766 |
Scenario 4: Treatment comparators | |||
SoC | 21,165 | 8.29 | Reference |
Inclisiran + SoC | 79,481 | 8.45 | Extended dominance |
Alirocumab + SoC | 86,515 | 8.47 | Extended dominance |
Evolocumab + SoC | 87,440 | 8.49 | 336,296 |
Scenario 5: 20-year analysis horizon | |||
SoC | 18,433 | 7.81 | Reference |
Inclisiran + SoC | 73,908 | 7.93 | 463,146 |
ASCVD = atherosclerotic cardiovascular disease; ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year; SoC = standard of care.
Note: Reanalyses are based on publicly available prices of the comparator treatments.
Table 20: CADTH Scenario Analyses Results — HeFH Subgroup
Drug | Total Costs ($) | Total QALYs | ICER ($/QALY) |
|---|---|---|---|
CADTH base case | |||
SoC | 12,813 | 17.14 | Reference |
Inclisiran + SoC | 108,756 | 17.29 | 626,458 |
Scenario 1: Relationship between LDL-C reduction and cardiovascular risk | |||
SoC | 12,574 | 17.15 | Reference |
Inclisiran + SoC | 110,298 | 17.69 | 180,480 |
Scenario 2: Effectiveness estimates based on observed values | |||
SoC | 12,811 | 17.15 | Reference |
Inclisiran + SoC | 108,791 | 17.30 | 625,228 |
Scenario 3: Effectiveness estimates based on indirect evidence | |||
SoC | 12,766 | 17.14 | Reference |
Inclisiran + SoC | 108,713 | 17.29 | 625,539 |
Scenario 4: Treatment comparators | |||
SoC | 12,813 | 17.14 | Reference |
Inclisiran + SoC | 108,756 | 17.29 | Extended dominance |
Alirocumab + SoC | 122,088 | 17.31 | Extended dominance |
Evolocumab + SoC | 123,875 | 17.32 | 609,114 |
Scenario 5: 20-year analysis horizon | |||
SoC | 7,911 | 12.45 | Reference |
Inclisiran + SoC | 83,205 | 12.51 | 1,327,732 |
HeFH = heterozygous familial hypercholesterolemia; ICER = incremental cost-effectiveness ratio; LDL-C = low-density lipoprotein cholesterol; QALY = quality-adjusted life-year; SoC = standard of care; vs. = versus.
Note: Reanalyses are based on publicly available prices of the comparator treatments.
Exploratory Scenario Analyses
CADTH conducted exploratory price reduction analyses, in which evolocumab plus SoC and alirocumab plus SoC were included as comparators. These analyses utilize effectiveness data from the sponsor’s NMA. These treatments were not included in the CADTH base case owing to limitations with the interpretation of NMA’s findings, as noted in the CADTH Clinical Review. As such, the results of this exploratory price reduction analysis should be interpreted with caution.
Table 21: CADTH Exploratory Price Reduction Analyses
Analysis | ICERs for inclisiran vs. evolocumab, alirocumab, and SoC ($/QALY) | |
|---|---|---|
Price reduction | ASCVD subgroupa | HeFH subgroupa |
No price reduction | WTP < 336,296: SoC WTP > 336,296: evolocumab + SoC | WTP < 609,114: SoC WTP > 609,114: evolocumab + SoC |
10% | WTP < 329,187: SoC WTP 329,187 to 394,436: inclisiran + SoC WTP ≥ 394,436: evolocumab + SoC | WTP < 562,426: SoC WTP 562,426 to 854,426: inclisiran + SoC WTP ≥ 854,140: evolocumab + SoC |
20% | WTP < 290,589: SoC WTP 290,589 to 565,797: inclisiran + SoC WTP ≥ 565,797: evolocumab + SoC | WTP < 498,395: SoC WTP 498,395 to 1,190,191: inclisiran + SoC WTP ≥ 1,190,191: evolocumab + SoC |
30% | WTP < 251,992: SoC WTP 251,992 to 737,158: inclisiran + SoC WTP ≥ 737,158: evolocumab + SoC | WTP < 434,363: SoC WTP 434,363 to 1,526,242: inclisiran + SoC WTP ≥ 1,526,242: evolocumab + SoC |
40% | WTP < 213,394: SoC WTP 213,394 to 908,520: inclisiran + SoC WTP ≥ 908,520: evolocumab + SoC | WTP < 370,332: SoC WTP 370,332 to 1,862,293: inclisiran + SoC WTP ≥ 1,862,293: evolocumab + SoC |
50% | WTP < 174,796: SoC WTP 174,796 to 1,079,881: inclisiran + SoC WTP ≥ 1,079,881: evolocumab + SoC | WTP < 306,300: SoC WTP 306,300 to 2,198,344: inclisiran + SoC WTP ≥ 2,198,344: evolocumab + SoC |
60% | WTP < 136,198: SoC WTP 136,198 to 1,251,242: inclisiran + SoC WTP ≥ 1,251,242: evolocumab + SoC | WTP < 242,268: SoC WTP 242,268 to 2,534,394: inclisiran + SoC WTP ≥ 2,534,394: evolocumab + SoC |
70% | WTP < 97,600: SoC WTP 97,600 to 1,422,603: inclisiran + SoC WTP ≥ 1,422,603: evolocumab + SoC | WTP < 178,237: SoC WTP 178,237 to 2,870,445: inclisiran + SoC WTP ≥ 2,870,445: evolocumab + SoC |
80% | WTP < 59,003: SoC WTP 59,003 to 1,593,964: inclisiran + SoC WTP ≥ 1,593,964: evolocumab + SoC | WTP < 114,205: SoC WTP 114,205 to 3,206,496: inclisiran + SoC WTP ≥ 3,206,496: evolocumab + SoC |
83% | WTP < 47,423: SoC WTP 47,423 to 1,645,373: inclisiran + SoC WTP ≥ 1,645,373: evolocumab + SoC | WTP < 94,996: SoC WTP 94,996 to 3,307,312: inclisiran + SoC WTP ≥ 3,307,312: evolocumab + SoC |
90% | NA | WTP < 50,174: SoC WTP 50,174 to 3,542,547: inclisiran + SoC WTP ≥ 3,542,547: evolocumab + SoC |
91% | NA | WTP < 43,771: SoC WTP 43,771 to 3,576,152: inclisiran + SoC WTP ≥ 3,576,152: evolocumab + SoC |
ASCVD = atherosclerotic cardiovascular disease; HeFH = heterozygous familial hypercholesterolemia; ICER = incremental cost-effectiveness ratio; NA = not applicable; SoC = standard of care; WTP = willingness to pay.
aAlirocumab was subject to extended dominance in all analyses.
Appendix 5: Submitted BIA and CADTH Appraisal
Table 22: Summary of Key Takeaways
Key Takeaways of the BIA |
|---|
|
Summary of Sponsor’s BIA
The submitted budget impact analysis27 (BIA) assessed the expected budgetary impact of reimbursing of inclisiran as an adjunct to diet and maximally tolerated statin therapy, with or without other lipid-lowering therapies, in adult patients with HeFH or ASCVD, who require additional lowering of LDL‐C. The sponsor-submitted subgroup analyses for patients with HeFH and for those with ASCVD, as well as an estimate of the budgetary impact for the full reimbursement request. The BIA was undertaken from the perspective of the Canadian public drug plans over a 3-year time horizon, and the sponsor’s pan-Canadian estimates reflect the aggregated results from provincial budgets (excluding Quebec), as well as the Non-Insured Health Benefits Program. Key inputs to the BIA are documented in Table 23.
The sponsor used an epidemiologic approach to estimate the number of eligible patients with HeFH and ASCVD (Table 23), assuming a prevalence of 0.4% for HeFH and 8.55% for ASCVD. The sponsor assumed that the proportion of patients with diagnosed HeFH would increase each year, from 15% before reimbursement of inclisiran to 47.5% of patients in the third year. The sponsor assumed that between 23% and 100% of patients would be eligible for public payer coverage based on the percentage of statins units covered by public payers in each province using data from IQVIA Pharmastat.28 Market share for evolocumab and alirocumab was based on the number of units for each from IQVIA Pharmastat,28 all of which were assumed to be used in the HeFH subgroup. The sponsor assumed that the use of statins and ezetimibe would not be affected by the reimbursement of inclisiran. Population growth was based on Statistics Canada’s annual growth-projection scenario (medium growth).29
The sponsor’s submission considered a reference scenario in which HeFH patients were assumed to receive evolocumab or alirocumab on a background of statins with or without ezetimibe, while patients in the ASCVD subgroup were assumed to receive statins with or without ezetimibe. In both subgroups, 20% of patients were assumed to be intolerant to statins and were assumed to receive no treatment (18.6%) or ezetimibe alone (1.4%) as SoC. In the new drug scenario, the uptake of inclisiran in the ASCVD population was assumed by the sponsor to be |||| in year 1, ||||% in year 2, and ||||% in year 3, which was intended to reflect a scenario involving a “population health agreement” for high-risk patients (LDL-C > | mmol/L).27 Uptake in the HeFH group was jurisdiction-specific and was assumed to differ between PCSK9-naïve patients and those switching from evolocumab or alirocumab.
Table 23: Summary of Key Model Parameters
Parameter | Sponsor’s estimate | |
|---|---|---|
Target population Prevalence Diagnosed (year 1 / year 2 / year 3) Treated with a lipid-lowering therapy Uncontrolled LDL Public coverage | HeFH 32.5% / 40% / 47.5%a 84.7%32 64.2%32,b Jurisdiction-specificc | ASCVD 8.55%33 NA 66%33 67%34,b Jurisdiction-specificc |
Number of patients eligible for drug under review (year 1 / year 2 / year 3) HeFH ASCVD | 11,281 / 14,058 / 16,900 603,336 / 610,885 / 618,456 | |
Market uptake (3 years); year 1 / year 2 / year 3 | ||
Uptake (reference scenario) HeFH population Inclisiran Evolocumab Alirocumab ASCVD population Inclisiran | 0% / 0% / 0% Jurisdiction-specificd Jurisdiction-specificd 0% / 0% / 0% | |
Uptake (new drug scenario) HeFH population Inclisiran Evolocumab Alirocumab ASCVD population Inclisiran | Jurisdiction and subgroup-specifice Jurisdiction-specificf Jurisdiction-specificf |||||% / |||||% / |||||%g | |
Annual cost of treatment (per patient) | ||
Inclisiran Evolocumab Alirocumab Statin therapy Ezetimibe | First year: $8,518; subsequent years: $5,679 $6,661 $6,661 $68 $66 | |
ASCVD = atherosclerotic cardiovascular disease; HeFH = heterozygous familial hypercholesterolemia; LDL = low-density lipoprotein; NA = not applicable; Y = year.
aThe sponsor assumed that 25% of patients would be diagnosed in the baseline year, with a subsequent increase of 7.5% per year after the reimbursement of inclisiran.
bThe proportion of patients whose LDL-C was not reduced to ≤ 1.8 mmol/L with statin treatment.
cEstimated based on 2019 IQVIA Pharmastat data for percentage of statins units covered by the public payer in each province.
dProjected market uptake for each comparator in the reference scenario was based on jurisdiction-specific historic claims data.
eIn the HeFH population, the sponsor assumed that inclisiran will have different uptake among patients adding inclisiran to a statin +/− ezetimibe background and among those switching from another PCSK9 inhibitor; this uptake was additionally assumed to vary between provinces.
fAssumed by the sponsor to vary by province.
gInclisiran market uptake in the ASCVD population was assumed by the sponsor to reflect a scenario in which a “population health agreement” for high-risk patients (with LDL-C level over ||||||||) is in place.
Summary of the Sponsor’s BIA Results
The sponsor estimated the net 3-year budget impact of introducing inclisiran for the HeFH patient subgroup to be $11,259,046 (year 1: $ 2,736,049; year 2: $ 4,243,906; year 3: $ 4,279,091). the net 3-year impact of introducing inclisiran for the ASCVD subgroup was estimated to be $1,543,765,514 (year 1: $287,790,912; year 2: $564,904,165; year 3: $691,070,437). The combined budget impact was projected by the sponsor to be $1,555,024,560 over 3 years.
CADTH Appraisal of the Sponsor’s BIA
CADTH identified several key limitations to the sponsor’s analysis that have notable implications on the results of the BIA:
The number of patients eligible for public drug coverage of inclisiran is underestimated. The sponsor estimated the proportion of patients eligible for public drug plan coverage by use IQVIA claims data for statins. There are 2 main issues with this approach. First, it is more appropriate to use the proportion of patients eligible for public coverage, rather than based on claims data, as the market size will be determined by all eligible patients for public coverage. Should inclisiran be reimbursed by public plans, it is assumed that all eligible patients for this treatment would enrol for public coverage. Second, there is uncertainty about the indication for statin claims, as the use of claims data does not account for statins used outside the scope of the current submission (e.g., ASCVD risk–equivalent conditions). Notably, the distribution of statins in the sponsor’s submission is not aligned with that from the ORION trials, where the majority of patients were taking high-intensity statins at baseline.
In CADTH’s reanalysis, the proportion of patients eligible for public drug plan coverage35 was used to determine the market size for inclisiran, based on the age distribution of ASCVD patients in the ORION-10 trial. Owing to the structure of the sponsor’s model, CADTH was unable to separately model the proportion of ASCVD and HeFH patients eligible for public drug plan coverage. Given that the starting age of HeFH patients in the cohort was 52.36 years based on the ORION-9 trial population,1 it is likely that the CADTH base case overestimates the proportion of HeFH patients eligible for public coverage. However, as HeFH patients account for approximately 2% of patients in the sponsor’s BIA, the effect on the budgetary impact is likely minimal.
The market uptake of inclisiran is uncertain. In the HeFH subgroup, the market uptake of inclisiran was based on the sponsor’s internal assumptions and on jurisdiction-specific use of evolocumab and alirocumab, and the sponsor assumed that the uptake of inclisiran will differ between those adding a PCSK9 inhibitor for the first time (i.e., adding inclisiran onto a statin +/− ezetimibe) and among those current taking a PCSK9 (i.e., switching from evolocumab or alirocumab). These assumptions were not justified by the sponsor. Among new PCSK9 users in the HeFH subgroup, the sponsor’s assumed uptake across jurisdictions in the first year of the BIA ranged from 1.5% to 4.8%, while uptake ranged from 3.6% to 11.6% in the third year. Among those switching from another PCSK9 inhibitor, uptake ranged from 10.8% to 29.6% in year 1 and from 0% to 29.6% in year 3 across jurisdictions. For the ASCVD subgroup, the sponsor adopted a new drug scenario in which inclisiran uptake would reflect a “population health agreement” with the objective to treat ||||% of eligible ASCVD patients with an LDL-C level of more than |||| mmol/L after 3 years.27 The sponsor assumed this would represent uptake of ||||% in year 1, ||||% in year 2, and ||||% in year 3 of the total eligible ASCVD population. In this population, uptake was assumed to not vary by jurisdiction, in contrast with the HeFH population. In both subgroups, the clinical expert consulted by CADTH indicated that the projected uptake of inclisiran is uncertain and is likely to be driven by marketing and patient preference (e.g., for the number of injections per year).
CADTH explored the impact of adopting higher market uptake of inclisiran the HeFH and ASCVD subgroups in scenario analyses.
Additional limitations were identified, but were not considered to be key limitations.
Misalignment of model inputs between the sponsor-submitted economic analysis and BIA. The annual cost of statin treatment was not aligned between the BIA and the cost-effectiveness analysis submitted by the sponsor. The sponsor’s BIA submission notes that the proportion of patients taking each statin and dose was consistent with the CUA; as such, the annual cost of statin therapy should be equal across the BIA and CUA. Because PCSK9 inhibitors were assumed to be used on a background of statin therapy, the impact of this misalignment was minimal.
In the CADTH base case, the annual cost of statin therapy was assumed to be $54 per patient, consistent with the value incorporated by the sponsor in the CUA
CADTH Reanalyses of the BIA
CADTH revised the sponsor’s base case by changing the percentage of patients eligible for public drug plan coverage, changing the proportion of HeFH patients who are diagnosed, and changing the annual cost of statin therapy (Table 24).
Table 24: CADTH Revisions to the Submitted Budget Impact Analysis
Stepped analysis | Sponsor’s value or assumption | CADTH value or assumption |
|---|---|---|
Corrections to sponsor’s base case | ||
None | — | — |
Changes to derive the CADTH base case | ||
1. Percentage of patients eligible for public drug plan coverage | Based on IQVIA Pharmastat data for the percentage of statins units covered by public payers in each province. | Based on the percentage of patients eligible for enrolment in public drug plans.35 |
2. Diagnosis of HeFH | The sponsor assumed that 25% of patients would be diagnosed in the baseline year, with a subsequent increase of 7.5% per year after the reimbursement of inclisiran. | Assumed to remain constant each year (25%), on the basis of clinical expert input that the proportion of patients diagnosed is not expected to increase with the introduction of inclisiran. |
3. Alignment of parameters in the BIA with the CUA. | Annual cost of statin treatment, per patient: $68. | Annual cost of statin treatment, per patient: $54. |
CADTH base case | 1 + 2 + 3 | |
BIA = budget impact analysis; CUA = cost-utility analysis; HeFH = heterozygous familial hypercholesterolemia.
The results of the CADTH step-wise reanalysis are presented in summary format in Table 25 and Table 26 (a more detailed breakdown is presented in Table 27.
In the CADTH reanalysis, the 3-year budget impact of reimbursing inclisiran is estimated to be $1,967,545,205 for the full reimbursement request (Table 25). The impact is driven by the ASCVD subgroup, in which the 3-year budget impact is projected to be $1,962,723,725 (year 1: $365,897,253; year 2: $718,192,585; year 3: $878,633,888). The 3-year budget impact of introducing inclisiran for the HeFH subgroup is estimated to be $4,821,480 (year 1: $2,305,280; year 2: $2,250,286; year 3: $265,914).
Table 25: Summary of the CADTH Reanalyses of the BIA — By Patient Subgroup
Stepped analysis | Three-year total ($) |
|---|---|
Submitted base case | 1,555,024,560 |
ASCVD patients | 1,543,765,514 |
HeFH patients | 11,259,046 |
CADTH base case | 1,967,545,205 |
ASCVD patients | 1,962,723,725 |
HeFH patients | 4,821,480 |
ASCVD = atherosclerotic cardiovascular disease; BIA = budget impact analysis; HeFH = heterozygous familial hypercholesterolemia.
Table 26: Summary of the CADTH Reanalyses of the BIA — Full Reimbursement Request
Stepped analysis | Three-year total ($)a |
|---|---|
Submitted base case | 1,555,024,560 |
CADTH reanalysis 1 | 1,977,966,674 |
CADTH reanalysis 2 | 1,546,279,243 |
CADTH reanalysis 3 | 1,555,024,560 |
CADTH base case | 1,967,545,205 |
BIA = budget impact analysis.
aCombined ASCVD and HeFH subgroups.
CADTH also conducted additional scenario analyses to address remaining uncertainty, using the CADTH base case. Results are provided in Table 27.
Adopting a higher prevalence of HeFH (0.46%36).
Assuming that the proportion of HeFH patients diagnosed increases with the introduction of inclisiran (32.5% / 40% / 47.5%27).
Adopting a 10% higher prevalence of ASCVD (9.4%).
Assuming 10% higher uptake of inclisiran (HeFH: jurisdiction-specific; ASCVD: 6.2% / 14.0% / 19.0%).
Table 27: Detailed Breakdown of the CADTH Reanalyses of the BIAa
Stepped analysis | Scenario | Year 0 (current situation) | Year 1 | Year 2 | Year 3 | Three-year total |
|---|---|---|---|---|---|---|
Submitted base case | Reference | 68,422,492 | 79,965,796 | 91,611,763 | 103,262,932 | 274,840,491 |
New drug | 68,422,492 | 370,492,757 | 660,759,833 | 798,612,460 | 1,829,865,050 | |
Budget impact | 0 | 290,526,961 | 569,148,071 | 695,349,528 | 1,555,024,560 | |
CADTH base case | Reference | 69,633,477 | 81,033,111 | 92,536,007 | 104,042,612 | 277,611,730 |
New drug | 69,633,477 | 449,235,644 | 812,978,878 | 982,942,414 | 2,245,156,935 | |
Budget impact | 0 | 368,202,533 | 720,442,871 | 878,899,801 | 1,967,545,205 | |
CADTH scenario analysis: 83% price reduction | Reference | 69,633,477 | 81,033,111 | 92,536,007 | 104,042,612 | 277,611,730 |
New drug | 69,633,477 | 139,827,344 | 205,752,317 | 238,516,615 | 584,096,277 | |
Budget impact | 0 | 58,794,233 | 113,216,310 | 134,474,003 | 306,484,546 | |
CADTH sensitivity analysis 1: Increased prevalence of HeFH | Reference | 69,711,700 | 81,112,368 | 92,616,253 | 104,123,856 | 277,852,478 |
New drug | 69,711,700 | 449,825,244 | 813,861,933 | 983,890,442 | 2,247,577,618 | |
Budget impact | 0 | 368,712,875 | 721,245,679 | 879,766,586 | 1,969,725,141 | |
CADTH sensitivity analysis 2: Increased proportion of HeFH diagnosed | Reference | 69,633,477 | 81,191,626 | 92,856,993 | 104,530,074 | 278,578,693 |
New drug | 69,633,477 | 450,414,844 | 816,851,324 | 989,279,199 | 2,256,545,367 | |
Budget impact | 0 | 369,223,218 | 723,994,331 | 884,749,125 | 1,977,966,674 | |
CADTH sensitivity analysis 3: Increased prevalence of ASCVD | Reference | 73,238,112 | 84,685,427 | 96,233,903 | 107,786,451 | 288,705,781 |
New drug | 73,238,112 | 489,263,710 | 888,076,037 | 1,074,035,820 | 2,451,375,567 | |
Budget impact | 0 | 404,578,283 | 791,842,134 | 966,249,369 | 2,162,669,786 | |
CADTH sensitivity analysis 4: Increased uptake of inclisiran | Reference | 69,633,477 | 81,033,111 | 92,536,007 | 104,042,612 | 277,611,730 |
New drug | 69,633,477 | 488,669,449 | 886,136,607 | 1,068,161,568 | 2,442,967,624 | |
Budget impact | 0 | 407,636,338 | 793,600,600 | 964,118,956 | 2,165,355,894 |
ASCVD = atherosclerotic cardiovascular disease; BIA = budget impact analysis; HeFH = heterozygous familial hypercholesterolemia.
aCombined HeFH and ASCVD subgroups.
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up-to-date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third-party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.