CADTH Reimbursement Review
Human Insulin (Entuzity KwikPen)
Sponsor: Eli Lilly Canada Inc.
Therapeutic area: Diabetes mellitus
Clinical and Pharmacoeconomic Review
Abbreviations
AE
adverse event
BMI
body mass index
CFB
change from baseline
CI
confidence interval
FAS
full analysis set
FPG
fasting plasma glucose
GLP-1
glucagon-like peptide 1
LS
least squares
MMRM
mixed model for repeated measures
OAD
oral antihyperglycemic drug
PP
per protocol
RCT
randomized controlled trial
SAE
serious adverse event
SD
standard deviation
SGLT2
sodium-glucose cotransporter-2
SMBG
self-monitored blood glucose
T1DM
type 1 diabetes mellitus
T2DM
type 2 diabetes mellitus
TDD
total daily dose
TEAE
treatment-emergent adverse event
U-500R
Humulin R 500 units/mL KwikPen (also refers to Entuzity KwikPen)
Executive Summary
An overview of the submission details for the drug under review is provided in Table 1.
Item | Description |
|---|---|
Drug product | Human biosynthetic insulin (Entuzity KwikPen), 500 units/mL subcutaneous insulin injection, human biosynthetic, solution for injection |
Indication | Entuzity is indicated to improve glycemic control in adults and children with diabetes mellitus requiring more than 200 units of insulin per day |
Reimbursement request | As per indication |
Health Canada approval status | NOC |
Health Canada review pathway | Standard review |
NOC date | July 27, 2017 |
Sponsor | Eli Lilly Canada Inc. |
NOC = Notice of Compliance.
Introduction
Diabetes is a group of progressive metabolic disorders characterized by elevated plasma blood glucose levels1-3; in 2016, approximately 8.76% of the Canadian population had diabetes.4 There are 2 major types of diabetes: type 1 diabetes mellitus (T1DM) and type 2 diabetes mellitus (T2DM). Diabetes symptoms vary depending on how much the blood glucose is elevated. Some people, especially those in the early stage of T2DM, may not experience symptoms.5 In people with T1DM, symptoms tend to come on quickly and more severely.6 The most noticeable signs and symptoms of diabetes are increased thirst, frequent urination, hunger, unexplained weight loss, presence of ketones in the urine, fatigue, irritability, blurred vision, slow-healing sores, and frequent infections.1,2,5,6 T1DM can develop at any age, although it often appears during childhood or adolescence.6 T2DM, the more common type of diabetes, can develop at any age, although it is more common in people older than 40 years of age.5 Optimal glycemic control is the cornerstone of treatment for diabetes.1,2,5,6 Both fasting and postprandial plasma glucose levels correlate with the risk of complications and contribute to the measured glycated hemoglobin (hemoglobin A1C) value. Glycemic targets should be individualized based on the individual’s frailty or functional dependence and life expectancy. Currently available treatments for diabetes can be classified into 3 types: lifestyle changes (including diet and physical exercises); oral antihyperglycemic drugs (OADs) or other, such as metformin; and insulin. Among the 3 types of treatment methods, insulin is the most powerful in terms of glycemic control. However, because of the need for injections, insulin is the least convenient treatment.1,2,5,6 Patients with T1DM must use insulin from diagnosis because of an absolute lack of endogenous insulin.6 For T2DM, because the process is slow at the early stage of the disease, patients will try lifestyle modifications first and OADs next. However, when the condition becomes severe, patients with T2DM will rely on insulin for treatment as well.5
This is a tailored review submission of new formulation of a biosynthetic human regular insulin, Entuzity KwikPen. Entuzity KwikPen is the trade name for HUMULIN R 500 units/mL KwikPen (hereafter referred to as U-500R).7,8 U-500R is a biosynthetic human regular insulin at a concentration of 500 units/mL that has been investigated for the treatment of insulin-resistant patients with diabetes requiring high-dose insulin (daily doses > 200 units).7,8 HUMULIN R (100 units/mL [U-100]) was registered in Canada in 1983 (File # 9427-L13 to 181; NDS #HN-8242) and has a well-defined efficacy and safety profile. U-500 is a 5-times higher concentration version of HUMULIN R U-100.7,8 HUMULIN R U-500 regular insulin administered via vial and non-dedicated syringe was approved in the US in 1994 under New Drug Application (NDA) 18 to 780 and has been marketed since 1997.7,8 HUMULIN R U-500 has been available in Canada since 2002 in the non-dedicated syringe format through a special access program.7,8 Following a US FDA safety letter issued in October 2008 regarding concerns about medication errors observed with the U-500 vial and non-dedicated syringe, Eli Lilly and Company developed a dosing device for the HUMULIN R U-500 KwikPen that was approved in the US on December 29, 2015 (HUMULIN R U-500 United States Prescribing Information).7
The objective of this report is to review the potential benefits and harms of U-500R to improve glycemic control in adults and children with diabetes mellitus requiring more than 200 units of insulin per day, based on the clinical evidence submitted by the sponsor,9 which formed the basis of this submission.
Stakeholder Perspectives
The information in this section is a summary of input provided by the patient groups who responded to the call from CADTH for patient input, from a clinical expert consulted by CADTH for the purpose of this review, from a clinician group who responded to the CADTH call for clinician groups input, and from the public drug plans participating in CADTH drug reimbursement reviews.
Patient Input
Patient input was provided by Diabetes Canada, which is a national health charity representing Canadians living with diabetes or prediabetes. Diabetes Canada used an online survey conducted between January 29 and February 12, 2021, from people across Canada of all ages with T1DM or T2DM and their caregivers to gather information on patient input. The survey tool Diabetes Canada used consisted of a self-administered questionnaire of closed- and open-ended questions about respondents’ experience with diabetes and diabetes medications (with specific questions about the drug under review, U-500R), and expectations for new drug therapies in this country. A total of 48 people completed the survey: 26 with T1DM, 19 with T2DM, and 3 caregivers (1 for T1DM and 2 for T2DM).
The vast majority of patients expressed how challenging, preoccupying, time-consuming, and worrisome it is to live with diabetes. Patients characterize diabetes as a burden and a condition that must be dealt with 24/7 and 365 days a year with no breaks and no holidays or time off.
Keeping blood glucose at satisfactory level while avoiding low blood glucose is the most important outcomes for patients surveyed by Diabetes Canada. Other important outcomes for these patients are reducing complications. Ten patients (21%) reported having experience with the drug under review. In general, patients had good experiences with U-500R because it is easier to use, they do not have to measure blood glucose levels as frequently, and it has a quicker and longer blood glucose–controlling effect compared with regular U-100 insulin.
Clinician Input
The clinical expert consulted by CADTH indicated that in insulin-resistant patients who need large insulin doses, large volumes of insulin need to be injected to achieve these doses, and 2 or more injections at a time may be needed to reach the required dose if standard U-100 is used. Thus, patients requiring large doses of insulin 4 times per day may require 8 or more injections per day. For these patients, U-500R could be used as a monotherapy or in combination with other non-insulin treatments such as oral medications. U-500R is used to improve glycemic control for diabetes patients with high insulin resistance. Once a patient with T2DM requires insulin, other treatments such as lifestyle changes and medications have usually been tried. U-500R would be used as an alternative insulin and insulin regimen for patients requiring more than 200 units of insulin per day in either basal or basal-bolus regimens.
Clinician Group Input
One clinician group input on behalf of clinician members from the Diabetes Canada Professional Section was received. Views expressed by the clinician group were in agreement with the input of the clinical expert consulted by CADTH.
Drug Program Input
Input from drug programs explored the questions of generalizability to patients with T1DM, and the lack of comparators in the clinical studies. The responses to the questions are presented in Table 4.
Clinical Evidence
Description of the Study
The CADTH Clinical Review was based on a summary of clinical evidence provided by the sponsor with the CADTH tailored review process, focused on the pivotal B5K-US-IBHC (IBHC) study.9 IBHC was a 24-week, randomized, open-label, parallel-arm, noninferiority study in which patients were assigned to either thrice-daily or twice-daily U-500R dosing regimens in patients with T2DM requiring high-dose insulin therapy (201 units to 600 units per day). Subject randomization was stratified by site, baseline hemoglobin A1C, total daily dose (TDD), and pioglitazone use. The primary objective of this study was to compare the change in hemoglobin A1C from baseline after 24 weeks of treatment in 2 treat-to-target algorithms of U-500R (thrice daily and twice daily) in adult subjects with T2DM who did not achieve adequate glycemic control on high-dose U-100 insulins or insulin analogues with or without OADs.
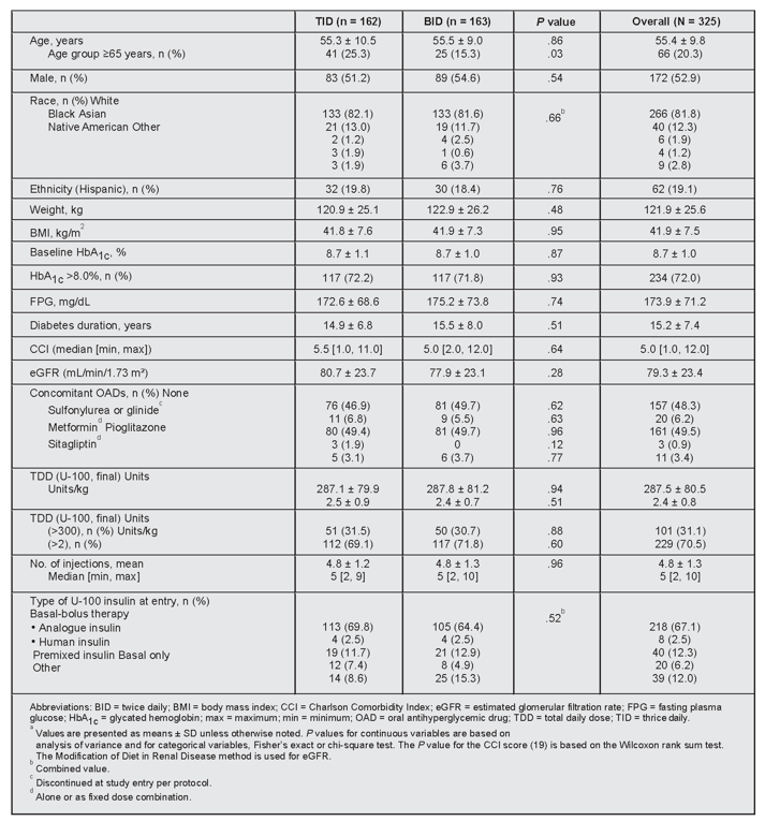
A total of 325 adult participants with T2DM (162 in the thrice-daily arm and 163 in the twice-daily arm) were randomized in the IBHC study. The mean age of study participants was 55.4 years (standard deviation [SD] = 9.8), and more than half (n = 172; 52.9%) were male. The main race groups reported were White (n = 266; 81.8%) and Black (n = 40; 12.3%); Hispanic was the main ethnic group (n = 62; 19.1%). Overall, 260 patients (80.0%) completed the study, with 132 of 162 patients (81.5%) completing the thrice-daily treatment and 128 of 163 patients (78.5%) completing the twice-daily treatment. The most frequent reasons for discontinuation included protocol violations (n = 27; 8.3%) and subject decision (n = 17; 5.2%). A total of 8 patients (2.5%) discontinued because of an adverse event (AE), and 1 (0.3%) because of death. There was no significant difference between the percentages of subjects who discontinued from the thrice-daily and twice-daily regimens for any reason. At baseline, patients were already on a mean of 287 units of insulin, a median of 5 injections per day (range = 2 to 10) and had a mean hemoglobin A1C level of 8.7% (SD = 1.0%). Patients were able to increase their insulin dose (51 units/day to 55 units/day) with fewer injections using U-500R, to achieve significantly improved glycemic control using treat-to-target algorithms.
Efficacy Results
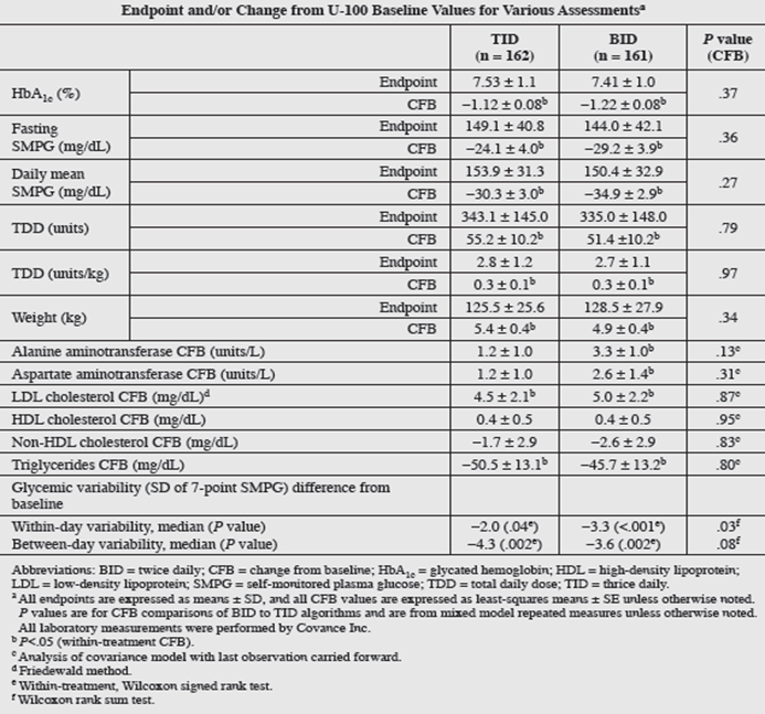
No difference in least squares (LS) mean of hemoglobin A1C at the end of 24 weeks of treatment was found between groups: thrice daily = 7.53% (SD = 1.1) and twice daily = 7.41% (SD = 1.0). However, the LS mean was reduced for hemoglobin A1C from baseline to 24 weeks of treatment with both the thrice-daily (1.12%; standard error [SE] = 0.08) and twice-daily (1.22%; SE = 0.08) regimens. The difference between the 2 treatment groups in change from baseline to the end of 24 weeks treatment was –0.10 with a 95% confidence interval (CI) of –0.33% to 0.12%. This 95% CI for the difference between the 2 treatment groups was within the predefined noninferiority margin of 0.4%. There was also a reduction in the number of injections per day, which declined by 2 and 3 in the thrice-daily and twice-daily groups, respectively, from baseline values of standard U-100 insulin treatment.
Harms Results
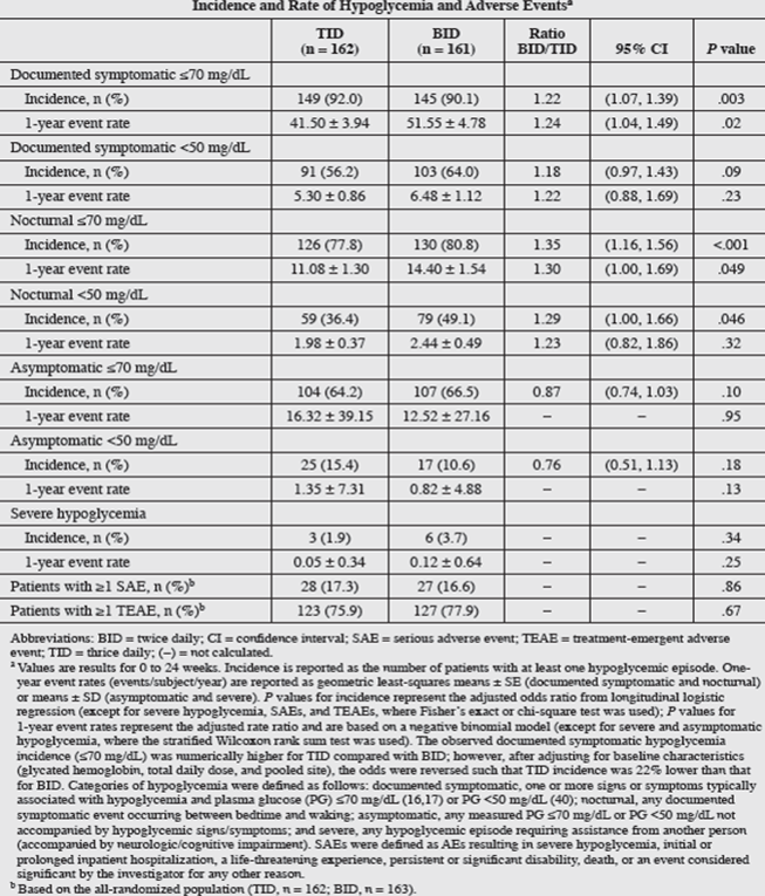
Serious adverse events (SAEs) during this trial were related to patients’ advanced diabetes disease state, as indicated by long diabetes duration and high pre-existing comorbidities at baseline. Incidences of SAEs and treatment-emergent AEs (TEAEs) were comparable between the thrice-daily and twice-daily groups. The only individual TEAEs that differed substantially between treatment groups were pain in extremity (thrice daily: n = 3, 1.9%; twice daily: n = 10, 6.1%; P = 0.049) and arthralgia (thrice daily: n = 7, 4.3%; twice daily: n = 1, 0.6%; P = 0.04). AEs requiring U-500R discontinuation (4 patients [2.5%] in each group) were also balanced. No AEs were recorded for dosing errors related to administration of U-500R via U-100 insulin syringes. Both treatment with U-500 and the titration algorithms were safe alternatives for patients who had failed glycemic control on high-dose/high-volume U-100 insulin therapy. The regimens showed no significant differences in severe hypoglycemia between the thrice-daily and twice-daily groups, although higher non-severe hypoglycemia and weight gain was observed in the twice-daily group. An increase in insulin dose (TDD increase of 41.4% for the thrice-daily group and 34.5% for the twice-daily group) from baseline to 24 weeks of treatment and weight gain accompanying reduction in hemoglobin A1C (0.47 kg per 1% reduction in hemoglobin A1C for the thrice-daily group and 1.31 kg per 1% reduction in hemoglobin A1C for the twice-daily group) were observed.
Table 2: Summary of Key Results From the Pivotal Study
Key result | Thrice-daily U-500 regimen | Twice-daily U-500 regimen | P valuea |
|---|---|---|---|
Screened, N | 467 | 467 | NR |
Randomized, N | 162 | 163 | NR |
Discontinued, N (%) | 30 (18.5) | 33 (20.5) | NR |
ITT, N | 162 | 161 | NR |
PP, N | 128 | 124 | NR |
Safety, N | 162 | 161 | NR |
TDD (units), mean (SD) | 343.1 (145.0) | 335.0 (148.0) | NR |
Hemoglobin A1C end point (%), mean (SD) | 7.53 (1.1) | 7.41 (1.0) | 0.37 |
Hemoglobin A1C CFB (%), LS mean (SE) | –1.12 (0.08)b | –1.22 (0.08)b | |
Fasting SMPG (mg/dL) end point, mean (SD) | 149.1 (40.8) | 144.0 (42.1) | 0.36 |
Fasting SMPG (mg/dL) CFB, LS mean (SE) | –24.1 (4.0)b | –29.2 (3.9)b | |
Weight (kg) end point, mean (SD) | 125.5 (25.6) | 128.5 (27.9) | 0.34 |
Weight (kg) CFB, LS mean (SE) | 5.4 (0.4)b | 4.9 (0.4)b | |
Glycemic variability (SD of 7-point SMPG) difference from baseline | |||
Within-day variability, median (P value) | –2.0 (0.04c) | –3.3 (< 0.01c) | 0.03d |
Between-day variability, median (P value) | –4.3 (0.002c) | –3.6 (0.002c) | 0.08d |
Patients with ≥ 1 SAE, n (%)e | 28 (17.3) | 27 (16.6) | NR |
Patients with ≥ 1 TEAE, n (%)e | 123 (75.9) | 127 (77.9) | NR |
Hypoglycemia | |||
Incidence, n (%) | 3 (1.9) | 6 (3.7) | NR |
1-year event rate, mean (SD) | 0.05 (0.34) | 0.12 (0.64) | NR |
Documented symptomatic < 50 mg/dL, n (%) | 91 (56.2) | 103 (64.0) | NR |
Nocturnal < 50 mg/dL, n (%) | 59 (36.4) | 79 (49.1) | NR |
Severe hypoglycemia, n (%) | 3 (1.9) | 6 (3.7) | NR |
CFB = change from baseline; hemoglobin A1C = glycated hemoglobin; ITT = intention to treat; LS = least squares; NR = not reported; PP = per protocol; SAE = severe adverse event; SMPG = self-monitored plasma glucose; TDD = total daily dose; TEAE = treatment-emergent adverse event.
aP values are reported as per sponsor’s submission.
bP < 0.05 (within-treatment CFB).
cWithin-treatment, Wilcoxon signed rank test.
dWilcoxon rank sum test.
eBased on total randomized population (thrice daily = 162, twice daily = 163).
Source: Clinical Study Report.9
Critical Appraisal
The overall design of the pivotal trial appears to be appropriate with respect to randomization and standardized assessment of the efficacy and safety outcomes. Based on the information available in the sponsor’s summary of the clinical evidence, the trial appeared to be generally well-balanced in terms of baseline demographics and disease characteristics. Improved glycemic control in this study was accompanied by increased insulin dose and weight gain.10-12 This patient population was likely at high risk for weight gain given their relatively poor glycemic control at baseline in conjunction with the good treatment response.12 Prior studies of U-500R therapy13-15 have reported mean weight changes of −0.3 kg to 6.8 kg. Concomitant use of metformin (approximately 50% of patients in the current study) or other non-insulin therapies may attenuate weight gain in patients with T2DM.10,16
This is a noninferior trial, with the primary objective to compare the hemoglobin A1C–lowering effect between thrice-daily and twice-daily regimens. The authors hypothesized that U-500R is superior to standard U-100 insulins or insulin analogues in terms of hemoglobin A1C–lowering effect, with or without OADs, for adult T2DM patients who require high doses of insulin, and made a before-after comparison component in the study. However, the primary objective of this study was to compare the change in hemoglobin A1C from baseline to 24 weeks of treatment in 2 treat-to-target algorithms (thrice daily versus twice daily). The results did show that there was a reduction in hemoglobin A1C from baseline to 24 weeks of treatment with both thrice-daily and twice-daily regimens. There was also a reduction in number of injections per day from baseline to after 24 weeks of treatment for both treatment groups.
A major limitation of the study submitted by the sponsor is that it made a comparison between thrice-daily and twice-daily regimens, with the assumption that U-500R is superior to standard U-100 insulins or insulin analogues in terms of hemoglobin A1C–lowering effect, with or without OADs, for adult T2DM patients who require high doses of insulin. They made a before-after comparison in the study and used change from baseline to end of 24 weeks of treatment as the outcome measure to demonstrate the hemoglobin A1C–lowering effect by U-500R. Although the superiority of U-500R to standard U-100 insulins or insulin analogues has been discussed in the literature,7,9 no evidence from randomized controlled trials (RCTs) with 2 parallel is available. Before-after comparison is not ideal to answer the question of superiority between U-500R and standard U-100 insulins or insulin analogues, especially in an open-label design. Patients enrolled into the trial may become aware of the importance of appropriate glycemia control, and health care providers may also intensify their monitoring effort during the trial. These may improve patient compliance resulting in better glycemia control, regardless of the drugs or methods of drug administration.
The clinical expert consulted for this review indicated that a comparison with standard of care using U-100 insulin regimens would be more informative clinically. For noninferiority between thrice-daily and twice-daily regimens, the authors found that the 95% CI for the difference between the 2 treatments in reduction of hemoglobin A1C from baseline to 24 weeks of treatment was within the predefined noninferiority margin of 0.4%. Based on these findings and the margin assumption, the authors considered that a noninferiority between the thrice-daily and twice-daily regimens was achieved in this study in terms of lowering hemoglobin A1C levels.9 However, defining a margin of equivalence can be very challenging and needs to be clearly described.17 The predefined margin of 0.4% is considered wide compared with the estimated point difference of 0.1% between the 2 groups. No clear explanation was provided about how and why this noninferiority margin was determined in the trial, and a simple comparison of the 95% CI of the difference with the predefined margin of noninferiority seems insufficient. Due to practical difficulties, this study was an open-label design. Because most of the outcomes were measured objectively, the lack of blindness or concealment of treatment allocation may have limited impact on these outcomes. However, some outcomes, such as AEs and symptoms of hypoglycemia, were assessed subjectively. Whether and to what extent the open-label design would bias the assessment of these subjective outcomes deserves consideration.
Overall, the proportion of patients who discontinued from the trial was high (20%) for a short-duration trial according to the clinical expert consulted by CADTH. Analysis for most outcomes was done by an intention-to-treat approach. However, the study did not mention if outcomes (especially hemoglobin A1C values) for discontinued participants were measured at the end of the study and included in the analysis. If the outcomes at the end of the study for all or a major proportion of discontinued patients were not measured, and therefore not included in the analysis, the results may be biased. However, there was no imbalance in discontinuations observed between treatment arms overall or for specific reasons of discontinuation.
Another limitation of this study was that it did not evaluate other combination therapy approaches, including newer OADs such as sodium-glucose cotransporter-2 (SGLT2) inhibitors18,19 or injectable glucagon-like peptide 1 (GLP-1) receptor agonists,18,20 which are typically accompanied by weight reduction. Participants who dropped out of the study after randomization were not replaced. Imputation for missing data was performed only for certain patients. The authors indicated that if the baseline data or all post-baseline observations for a given outcome were missing for a participant, then that participant’s data would not be included in the analysis of that outcome. However, exactly how many individual outcome measures were not included was not presented. No mention of handling multiplicity in the statistical analysis was made; however, only 1 comparison was made for the primary outcome. As a result, the lack of clarity on handling multiplicity in statistical analysis will not affect the results on the primary objective of the study.
There are also some data presentation issues. For example, total injections per day is an important outcome measure and should be presented clearly. However, only the number of reductions from baseline to end of 24 weeks of treatment was provided in the original report. Fasting glucose was expressed as mg/dL in the original study, not the standard units of mmol/L.
Despite the various generalizability issues, such as the different population profiles between US and Canada in terms of race and/or ethnic composition20-22 and the use of needles or syringes instead of the KwikPen, the baseline demographic and disease characteristics of the patients enrolled in the trial are generally representative of patients with high insulin resistance seen in clinical practice in Canada.
Economic Information
At a submitted price of $94.79 per package of two 3-mL prefilled pens, the cost per 100 units of U-500R insulin is $3.16. This cost per 100 units is less than that of prefilled pens and cartridges of most other human insulin or insulin analogue products available, but more than most of the less frequently used 10-mL vials. Assuming a mean dose of 339.1 units per day, the cost of U-500R insulin is $3,911 per patient per year.
The available clinical trial showed an increase in the number of units of insulin used per day compared to baseline, when patients previously using U-100 insulin were switched to U-500R (287.5 daily units versus 339.1 daily units). In analyses accounting for this change, U-500R insulin remained less expensive than most combinations of U-100 prandial and basal insulin analogue products (annual cost range: $4,277 to $6,026 per patient) but was more expensive than the combination of subsequent entry products Admelog-brand insulin lispro and Basaglar-brand insulin glargine (annual cost: $3,771 per patient) as well as combinations of human insulin (annual cost range: $2,577 to $3,415 per patient). The submitted price of U-500R insulin would need to be reduced by 3.6% for its annual cost to be equivalent to that of the least expensive combination of insulin analogues, and 14% or 34% to be equivalent to the least expensive combination of human insulins in cartridges or vials, respectively. For public plans that reimburse insulin needles, a small amount of additional savings may be realized due to the decreased number of injections required per day with the use of U-500R insulin.
These estimated costs or savings are uncertain due to a lack of comparative evidence between U-500R insulin and other concentrations of insulin. Additionally, these incremental costs or savings are based on publicly available list prices and may not reflect actual prices paid by Canadian public drug plans.
Conclusions
Based on the summary of clinical evidence submitted by the sponsor, for patients with T2DM who require greater than 200 units of insulin per day, U-500R reduced hemoglobin A1C levels within a 24-week period to a similar magnitude using a twice-daily or thrice-daily regimen. According to the clinical expert consulted for this review, U-500R fulfills a need for diabetes patients with severe insulin resistance, and it may replace basal-bolus insulin regimens in this patient population. Further, the clinical expert indicated that the findings of the clinical efficacy and AEs reported in the included trial were aligned with what would be expected in Canadian clinical practice. Issues with the submitted study that were identified include insufficiency in terms of predefined noninferiority margin and method used for the analysis of noninferiority, potential bias in subjective assessment of some outcomes because of the open-label design, lack of clarity on handling of missing information, and a large proportion of discontinued participants and lack of clarity on outcome measures in these discontinued participants. Although superiority of U-500R to standard of care using U-100 insulin for diabetes patients requiring high-dose insulin has been discussed in the literature, no evidence from RCTs making direct comparisons between U-500R and regular U-100 insulin in terms of efficacy and safety is available. The clinical expert consulted for this review indicated that a comparison with standard of care using U-100 insulin regimens would be more informative clinically. Whether diabetes patients requiring more than 200 units of insulin per day are willing to accept an increased insulin dose and weight gain by switching to U-500R deserves consideration.
At the submitted price and based on the mean dose at week 24 in the pivotal IBHC trial, the annual cost of U-500R insulin is $3,911 per patient per year. When the comparators are assumed to be dosed as at baseline in the pivotal IBHC trial, U-500R insulin is less expensive than combinations of originator brands of basal and prandial insulin analogues, but more expensive than combinations of human insulin or the 2 available subsequent entry insulin analogues. The submitted price of U-500R insulin would need to be reduced by 3.6% for its annual cost to be equivalent to that of the least expensive combination of insulin analogues, and 14% or 34% to be equivalent to the least expensive combination of human insulins in cartridges or vials, respectively. The costs and savings associated with the use of U-500R insulin are uncertain due to a lack of comparative clinical evidence to other insulin products.
Introduction
Disease Background
Diabetes refers to a group of diseases that affect how the patient’s body uses blood glucose.1-3 The underlying cause of diabetes varies by type1,2,5,6,23; all types of diabetes can lead to excess sugar in the blood, which can lead to serious health problems. Majority of chronic diabetes consists of 2 types: T1DM and T2DM.1,2,5,6 Potentially reversible diabetes conditions include prediabetes and gestational diabetes.1,2 Prediabetes occurs when blood glucose levels are higher than normal, but not high enough to be classified as diabetes. Prediabetes is often the precursor of diabetes unless appropriate measures are taken to prevent progression.1,2 Gestational diabetes occurs during pregnancy but it may resolve after the birth of the baby.24
T1DM can develop at any age, although it often appears during childhood or adolescence.6 T2DM, the more common type of diabetes, can develop at any age, although it is more common in people older than 40 years of age.5 T1DM occurs when the beta cells of the pancreas are destroyed by the immune system and can no longer produce insulin, which the body needs in adequate supply to help it function.6 There is no known way to prevent T1DM; 5% to 10% of people with diabetes have T1DM.1,2,6 T2DM occurs when the body does not make enough insulin and/or does not respond well to the insulin it makes.5 Approximately 90% to 95% of people with diabetes have T2DM.1,2,5 People are typically diagnosed with T2DM after the age of 40 years, although it is now also being seen in children and adolescents.5 This type of diabetes is associated with excess body weight and, in most people, is preventable by following a healthy meal plan, getting regular exercise, and maintaining a healthy weight.5 Diabetes symptoms vary depending on how much the blood glucose level is elevated.1,2 Some people, especially those with prediabetes or T2DM, may not experience symptoms.1,2,5 In T1DM, symptoms tend to come on quickly and be more severe.6 Some of the main signs and symptoms of T1DM and T2DM are increased thirst, frequent urination, extreme hunger, unexplained weight loss, presence of ketones in the urine (ketones are a by-product of the breakdown of muscle and fat that happens when there is not enough insulin available), fatigue, irritability, blurred vision, slow-healing sores, and frequent infections, such as gum or skin infections and vaginal infections. Cardiovascular and renal functions are often compromised in the late stage of diabetes.1,2,5,6
In 2016, 8.76% of Canadians were living with diabetes.4 This rate increased to 9% in 2019 and is projected to increase to 11% in 2029.25 Although the prevalence increases with age, more than 50% of Canadians diagnosed with diabetes (1.2 million) are of working age, between 25 years and 64 years of age.25 The overall prevalence is higher among males (7.2%) than females (6.4%). Newfoundland and Labrador, Nova Scotia, and Ontario have the highest age-standardized prevalence rates of diagnosed diabetes in Canada; Nunavut, Alberta, and Quebec have the lowest.25 Individuals with diabetes are more than 3 times more likely to be hospitalized with cardiovascular disease than individuals without diabetes, 12 times more likely to be hospitalized with end-stage renal disease, and almost 20 times more likely to be hospitalized with non-traumatic lower limb amputations. Diabetes was the primary cause of 34% of new cases of end-stage renal disease in 2009, creating a growing demand for renal replacement therapy (dialysis or transplant) in Canada. Because diabetes shares several risk factors with other chronic diseases, 36.5% of Canadian adults with diabetes also reported having 2 or more other serious chronic conditions (hypertension, heart disease, chronic obstructive pulmonary disease, mood disorder, and/or arthritis), and 12.5% reported having 3 or more.25 Nearly 40% of Canadian adults who reported having diabetes rated their health as “fair” or “poor” compared with 10% of the adult population without diabetes. Although only 3.1% of all deaths in Canada were attributed to diabetes in 2007, more than a quarter (29.9%) of individuals who died had diabetes in 2008 to 2009.25 Diabetes itself does not typically lead directly to death; it is the complications associated with diabetes that do lead to death. In every age group, individuals with diabetes experienced mortality rates at least 2 times higher than those without diabetes. This results in noticeable decreases in life expectancy and health-adjusted life expectancy. The most recent cost estimates available for this report are outdated by 11 years, which is a major information gap. Therefore, it is difficult to assess the real economic burden of diabetes. However, it is expected that costs will only continue to rise with the increasing prevalence of diabetes and its associated health care costs.25
Diagnosis of diabetes is made by both clinical symptoms and results of laboratory investigation of glycemia or glycemia metabolism; the establishment of a diabetes diagnosis should be made by a clinical specialist.1-3 The vast majority of diabetes fall into 2 broad etiopathogenetic categories. In the T1DM category, the cause is an absolute deficiency of insulin secretion.1,2,6 Individuals at increased risk of developing this type of diabetes can often be identified by serological evidence of an autoimmune pathologic process occurring in the pancreatic islets and by genetic markers.6 In the T2DM category, which is much more prevalent, the cause is a combination of resistance to insulin action and an inadequate compensatory insulin secretory response.1,2,5 In the latter category, a degree of hyperglycemia sufficient to cause pathologic and functional changes in various target tissues, but without clinical symptoms, may be present for a long period of time before diabetes is detected.5 During this asymptomatic period, it is possible to demonstrate an abnormality in carbohydrate metabolism by measurement of plasma glucose in the fasting state or after a challenge with an oral glucose load.
Assigning a type of diabetes to an individual often depends on the circumstances present at the time of diagnosis, and many diabetic individuals do not easily fit into a single class.1,2 For T1DM, the rate of beta cell destruction is quite variable, being rapid in some individuals (mainly infants and children) and slow in others (mainly adults).6 Some patients, particularly children and adolescents, may present with ketoacidosis as the first manifestation of the disease. Others have modest fasting hyperglycemia that can rapidly change to severe hyperglycemia and/or ketoacidosis in the presence of infection or other stress. Still others, particularly adults, may retain residual beta cell function sufficient to prevent ketoacidosis for many years; such individuals eventually become dependent on insulin for survival and are at risk for ketoacidosis. At this latter stage of the disease, there is little or no insulin secretion, as manifested by low or undetectable levels of plasma C peptide.6 Immune-mediated diabetes commonly occurs in childhood and adolescence, but it can occur at any age, even in the eighth and ninth decades of life. Some forms of T1DM have no known etiologies. Some of these patients have permanent insulinopenia and are prone to ketoacidosis but have no evidence of autoimmunity.6 Individuals with this form of diabetes suffer from episodic ketoacidosis and exhibit varying degrees of insulin deficiency between episodes. This form of diabetes is strongly inherited, lacks immunological evidence for beta cell autoimmunity, and is not associated with human leukocyte antigen. An absolute requirement for insulin replacement therapy in affected patients may come and go.6
T2DM, which accounts for 90% to 95% of those with diabetes, includes individuals who have insulin resistance and usually relative (rather than absolute) insulin deficiency.1,2,5 At least initially, and often throughout their lifetime, these individuals do not need insulin treatment to survive.5 There are probably many different causes of this form of diabetes. Although the specific etiologies are not known, autoimmune destruction of beta cells does not occur in T2DM.5 Most patients with this form of diabetes are obese, and obesity itself causes some degree of insulin resistance.5 Patients who are not obese by traditional weight criteria may have an increased percentage of body fat distributed predominantly in the abdominal region. Laboratory investigation tests for T1DM, T2DM, and prediabetes usually include a hemoglobin A1C test, random blood glucose test, fasting blood glucose test, and oral glucose tolerance test.3 The hemoglobin A1C test, which does not require fasting, indicates average blood glucose levels for the past 2 to 3 months.3 It measures the percentage of blood glucose attached to hemoglobin, the oxygen-carrying protein in red blood cells. The higher the blood glucose levels, the more hemoglobin with sugar attached. A hemoglobin A1C level of 6.5% or higher on 2 separate tests indicates diabetes; a hemoglobin A1C level between 6.1% and 6.4% indicates prediabetes. A level less than 6.1% is considered normal. For a random blood glucose test, a blood sample is taken at a random time. A random blood glucose level of 200 mg/dL — 11.1 mmol/L — or higher suggests diabetes. For a fasting blood glucose test, a blood sample is taken after an overnight fast. A fasting blood glucose level of 126 mg/dL (7 mmol/L) or higher on 2 separate occasions indicates diabetes. For an oral glucose tolerance test, a fasting blood glucose level is measured first then blood glucose levels are tested periodically for the next 2 hours after a drink of sugary liquid. A blood glucose level of more than 200 mg/dL (11.1 mmol/L) after 2 hours indicates diabetes; a reading between 140 mg/dL (7.8 mmol/L) and 199 (11.0 mmol/L) indicates prediabetes. If prediabetes is suspected, urine will be tested for the presence of a by-product produced when muscle and fat tissue are used for energy because the body does not have enough insulin to use the available glucose (ketones).3
Standards of Therapy
Optimal glycemic control is fundamental to the management of diabetes. There are 3 methods for glycemic control: lifestyle modifications, including both physical exercises and nutrition therapy; oral or other medications; and insulin.1,2
Healthy eating. Diabetes patients need to centre their diet on more fruits, vegetables, lean proteins, and whole grains — foods that are high in nutrition and fibre and low in fat and calories — and cut down on saturated fats, refined carbohydrates, and sweets.
Physical activity. Exercise helps lower blood glucose levels by moving sugar into cells, where it is used for energy. Exercise also increases sensitivity to insulin, which means the body needs less insulin to transport sugar to cells.
Oral or other medications. Some diabetes medications stimulate the pancreas to produce and release more insulin, whereas other medications inhibit the production and release of glucose, which means less insulin to transport sugar into cells. Still other medications block the action of stomach or intestinal enzymes that break down carbohydrates or make tissues more sensitive to insulin. Metformin is generally the first medication prescribed for T2DM.
Insulin. Insulin is often used in T2DM, when patients have profound metabolic disturbances, when they have other comorbidities that preclude use of oral or other medications, or when their endogenous insulin production is sufficiently reduced that they require exogenous insulin.5
Canadian guidelines recommend a personalized diabetes care, which should be organized around the person living with diabetes and their supports.1 The person with diabetes should be an active participant in their own care, be involved in shared-care decision-making, and self-manage to their full abilities, facilitated by a proactive, interprofessional team with training in diabetes and the ability to provide ongoing self-management education and support. This model should be organized within the context of the expanded chronic care model and delivered using as many of the components of the model as possible (in particular, self-management education and support, interprofessional team-based care with expansion of professional roles, collaboration with the primary care provider, and monitoring with medication adjustment and case management). Individualized self-management education interventions according to the type of diabetes and recommended therapy within the context of the individual’s ability for learning and change, culture, health beliefs and preferences, literacy level, socioeconomic status, and other health challenges should be considered.
Both fasting and postprandial plasma glucose levels correlate with the risk of complications and contribute to the measured hemoglobin A1C values.1 Intensive glucose control with lowering hemoglobin A1C values to 7.0% or less in both T1DM and T2DM provides strong benefits for microvascular complications and, if achieved early in the disease with avoidance of hypoglycemia and glucose variability as part of a multifactorial treatment approach, likely provide a significant cardiovascular benefit. More intensive glucose control, hemoglobin A1C of 6.5% or less, may be sought in people with a shorter duration of diabetes and longer life expectancy, especially in those patients who are on treatment with antihyperglycemic agents with a low risk of hypoglycemia. A hemoglobin A1C target of 8.5% or less may be more appropriate in people with T1DM and T2DM with limited life expectancy, higher level of functional dependency, and a history of repeated severe hypoglycemia with hypoglycemia unawareness. Figure 1 displays the recommended targets for glycemic control.1
Figure 1: Recommended Targets for Glycemic Control
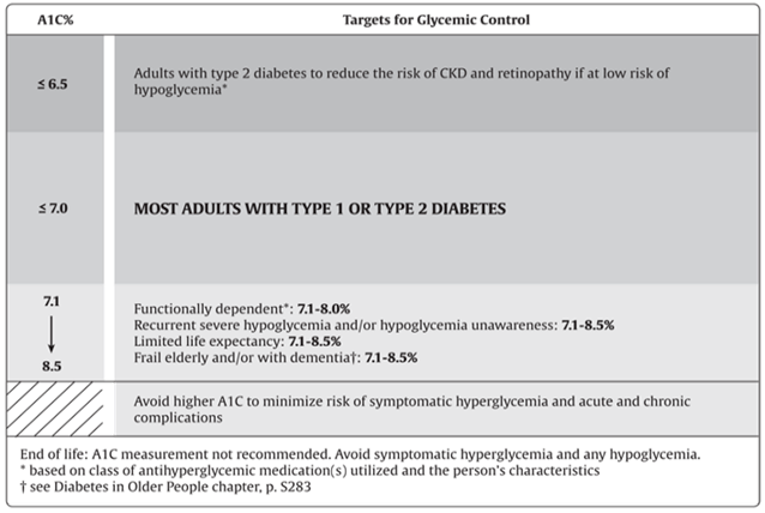
Source: Reprinted from Canadian Journal of Diabetes, 42(Suppl 1), Imran SA, Agarwal G, Bajaj HS, Ross S, Targets for glycemic control, p. S42-S46, Copyright 2018, with permission from Elsevier. https://www.sciencedirect.com/journal/canadian-journal-of-diabetes.26
Since glucose can be self-measured but facilities in a clinic or a pharmacy is needed for hemoglobin A1C test, and since glucose measures current blood glucose level while hemoglobin A1C test measures average blood glucose level over the past 3 months, these 2 measures can be complementary for monitoring blood glucose levels for diabetes patients.
Drug
The focus of this CADTH review, Entuzity (insulin injection, human biosynthetic U-500R), is a concentrated human insulin formulation.7,8 U-500R is produced by recombinant DNA technology in a non-disease–producing special laboratory strain of E. coli that has been genetically altered by the addition of the human gene for insulin production. U-500R is a concentrated human insulin indicated to improve glycemic control in adults and children with diabetes mellitus requiring more than 200 units of insulin per day.7
U-500R is reserved for the treatment of patients with diabetes requiring total daily doses of more than 200 units of insulin (basal and/or bolus). Each Entuzity KwikPen contains 1,500 units of insulin and can deliver from 5 units to 300 units per injection.
Table 3: Key Characteristics of U-500R
Key characteristics | Description |
|---|---|
Mechanism of action | Concentrated human insulin formulation |
Indicationa | To improve glycemic control in adults and children with diabetes requiring more than 200 units of insulin per day |
Route of administration | Subcutaneous |
Recommended dose | Solution for injection 500 units/mL in a 3-mL disposable prefilled pen (1,500 units of insulin) |
Serious adverse effects or safety issues | Hypoglycemia; hypokalemia; headache; confusion; drowsiness; fatigue; anxiety; blurred vision; diplopia; numbness of the lips, nose, or fingers; coma; seizure; or neurologic impairment, mostly caused by overdose. Adjustments in drug dosage, meal patterns, or exercise may be needed. |
Other | Entuzity is a clear, colourless solution. It is important to always examine the appearance of the cartridge in the prefilled pen. It should not be used if it has become viscous (thickened) or cloudy or if it has formed a deposit of solid particles on the wall of the cartridge. |
aHealth Canada–approved indication.
Source: Sponsor’s product monograph.7
Stakeholder Perspectives
Patient Group Input
This section was prepared by CADTH staff based on the input provided by patient group.
About Diabetes Canada and the Information Gathered
This patient input was prepared and submitted by Diabetes Canada. Diabetes Canada is a national health charity representing Canadians living with diabetes or prediabetes. The priorities of Diabetes Canada’s mission are diabetes prevention, care, and cure. Diabetes Canada helps to better manage diabetes by putting practical, evidence-based tools into the hands of health care providers.
Diabetes Canada used an online survey conducted between January 29 and February 12, 2021, from people across Canada of all ages with T1DM or T2DM and their caregivers to gather information on patient input and presented in this report to CADTH. The survey tool they used consisted of a self-administered questionnaire of closed- and open-ended questions about respondents’ experiences with diabetes and diabetes medications (with specific questions about the drug under review, U-500R), and expectations for new drug therapies in this country. It was advertised through Diabetes Canada’s social media channels (Facebook, Twitter, Instagram, and LinkedIn), by email through a monthly newsletter to members of the Diabetes Canada Professional Section, and through a post on the health care professional discussion platform TimedRight. A total of 48 people completed the survey: 26 with T1DM, 19 with T2DM, and 3 caregivers (1 for T1DM and 2 for T2DM). Respondents resided in 8 provinces (Newfoundland, Nova Scotia, New Brunswick, Ontario, Manitoba, Saskatchewan, Alberta, and British Columbia), with the most representation from Ontario (n = 21) and British Columbia (n = 12). Respondents’ ages ranged from younger than 18 years (n = 2) to 75 years and older (n = 3), with 79% of people reporting being at least 35 years old and the largest concentration of respondents (n = 14) falling in the 35-year to 44-year grouping. Approximately 27% of respondents (n = 13) reported living with diabetes for 10 years or less (5 had it for less than 1 year), 15 with diabetes for 11 years to 20 years, and the largest proportion (42%, n = 20) had diabetes for more than 20 years.
Disease Experience
Long-term high blood glucose could damage blood vessels and nerves, resulting in blindness, heart disease, kidney problems, and lower limb amputations, while low blood glucose could precipitate an acute crisis, such as confusion, coma, and/or seizure that, in addition to being dangerous, may also contribute to a motor vehicle, workplace, or other type of accident. As a result, appropriate management of blood glucose is the most important and yet most challenging issue for diabetes care, for both T1DM and T2DM, to keep glucose levels within a target range to minimize symptoms and prevent or delay complications.
The vast majority of patients expressed how challenging, preoccupying, time-consuming, and worrisome it is to live with diabetes. Patients characterize diabetes as a burden, a condition that must be dealt with 24/7 and 365 days a year with no breaks and no holidays or time off. Testing, medication adjustment, and equipment set-up and use all take lots of time. Planning meals, making food choices, and counting carbohydrates can be difficult. Patients often feel unwell, particularly when blood sugars are variable. Patients felt that living with complications is painful, upsetting, and bothersome, both physically and mentally. One patient expressed:
It makes life much more difficult. I don’t feel well most of the time, weight gain is a big problem, my feet are causing walking to be very difficult. Cost is a factor. Its very inconvenient having to give myself injections 3 times a day. Some of the tools offered for checking blood without using the test strips are too expensive for me. In my opinion it would be nice if the Government was more helpful with the costs of diabetes.
Experiences With Currently Available Treatments
A total of 45 patients reported having experience with antihyperglycemic agents to manage their diabetes. The oral and injectable medications being taken at the time of survey completion included GLP-1 receptor agonists, DPP-4 inhibitors plus metformin, SGLT2 inhibitors, sulfonylureas, metformin, and insulin. For those who had experience with insulin, the following types were used: glargine or glargine biosimilar, glargine U300 or other long-acting, intermediate-acting, short-acting, and rapid-acting formulations.
Twenty-nine percent of patients reported being “very satisfied” with the medication they are currently taking and 47% reported being “somewhat satisfied.” Another 13% said they were “neither satisfied nor dissatisfied” with their medication, while 11% said they were “somewhat dissatisfied” or “very dissatisfied.” When asked what they like about their medications, respondents cited the following: effectiveness and reasonable control of blood glucose, efficiency or speed with which it works, freedom to carry and use medication anywhere, and works with a pump.
Patients reported the following dislikes about their medications:
having to inject
having to take big doses
the length of time it takes the medications to work
fatigue caused by the medications
multiple daily injections
having to take medications daily
the size of tablets
the high cost
the frequency of monitoring that accompanies medication use
side effects.
The most frequently reported side effects included yeast infections, urinary tract infections, weight gain, fatigue, hypoglycemia, arrhythmias, stomach upset, diarrhea, loss of appetite, bruising around injection site, thirst, light headedness, and depression and “brain fog.” Many felt their current medication regimen is helping them better meet fasting blood glucose target levels and those measured upon waking and after meals, as well as their target hemoglobin A1C levels. Many felt that the medications they are taking now were neither better nor worse than ones previously taken with respect to weight maintenance or loss and problems like gastrointestinal issues, thirst or dehydration, and infections. Most respondents said they do not have any issues obtaining their medications, although some pointed out that they are costly or not covered by their health insurance, which are significant barriers. One person said, “Medication is so expensive so sometimes I have to conserve insulin use.”
Twelve patients provided answers to the question about what was very important when choosing pharmacotherapy for diabetes management. The most frequently cited considerations were:
keeping blood glucose at a satisfactory level during the day or after meals: 91%
keeping blood glucose at a satisfactory level upon waking or after fasting: 91%
avoiding low blood glucose overnight: 79%
avoiding low blood glucose during the day: 75%
avoiding weight gain or reducing weight: 63%
reducing risk of heart problems: 63%
reducing high blood pressure: 61%
avoiding fluid retention: 61%
avoiding gastrointestinal side effects: 56%
avoiding urinary tract and/or yeast infections: 56%
Improved Outcomes
The patients’ expectations for improved outcomes can be summarized as:
to reduce or to remove medication, with advance medical care, or a cure, plain and simple
to improve the delivery of insulin or other medication, such as faster working insulin or easy insulin delivery with less injections or not having to take needles, or to use better pump technology
to reduce side effects
to improve tracking devices for all children
to get more information about new happenings with diabetes treatments.
Experience With Drug Under Review
Ten patients (21%) reported having experience with the drug under review, U-500R. In total, 7 patients (1 with T1DM, 6 with T2DM, all older than 35 years and all with diabetes for at least 11 years) said that they currently take U-500R along with other diabetes medications, 3 only take U-500R (1 with T1DM, 2 with T2DM, all older than 45 years and all with diabetes for more than 20 years).
In general, patients had good experiences with U-500R because it has a quicker response, is easier to use, there is no need to measure blood glucose levels as frequently, and it has a longer blood glucose–controlling effect. One patient said, “I love how it controls my glucose levels without the need to increase volume. Checking my blood sugar levels several times a day. You notice the medication working. Less spikes.”
Additional Information
The Diabetes Canada 2018 Clinical Practice Guidelines for the Prevention and Management of Diabetes in Canada highlights the importance of personalized care. This includes careful consideration of medication selection, route of administration (oral, injection, infusion), frequency in monitoring blood glucose and adjusting dosage, benefits and risks that the patient experiences and/or tolerates, and lifestyle changes the patient is willing or able to make. These patient survey responses reinforce the message that different people with diabetes require different medications and/or treatment modalities to help effectively manage their disease. The unique clinical profile, preferences, and tolerance of therapy should direct prescribers to the most appropriate choice or combination of treatments for disease management. Health care providers should be supported in prescribing evidence-based therapies, and patients should have access through public and private drug plans to a range of treatments that will allow them to optimize their health outcomes. Although current therapies have generally led to improvements for many people with diabetes in blood glucose and hemoglobin A1C control, respondents hope for additional affordable agents that they can access equitably, in a timely manner, and with good results to help them lead a normal life. U-500R may help people achieve better glycemic control, which could potentially improve lives and save millions in direct health care costs. For this reason, U-500R should be an option for people living with diabetes.
Clinician Input
All CADTH review teams include at least 1 clinical specialist with expertise regarding the diagnosis and management of the condition for which the drug is indicated. Clinical experts are a critical part of the review team and are involved in all phases of the review process (e.g., providing guidance on the development of the review protocol, assisting in the critical appraisal of clinical evidence, interpreting the clinical relevance of the results, and providing guidance on the potential place in therapy). The following input was provided by 1 clinical specialist in the diagnosis and management of diabetes.
Unmet Needs
The clinical expert indicated that for diabetes patients required to use 200 units or more per day, large volumes of insulin need to be injected at each dose, and often 2 or more injections are needed at a time to reach the required dose. Patients may be on basal-only insulin regimens or on basal-bolus regimens.
Place in Therapy
The clinical expert consulted by CADTH for this review considered that U-500R should be reserved for diabetes patients with high insulin resistance who require more than 200 units of insulin per day. U-500R could be used as a monotherapy or in combination with other non-insulin medications. U-500R is used to improve glycemic control for diabetes patients with high insulin resistance. Other treatments, such as lifestyle changes and medications, have usually been tried for T2DM before insulin is started. U-500R would be used as an alternative insulin and insulin regimen for patients requiring more than 200 units of insulin per day in either basal or basal-bolus regimens.
Patient Population
The clinical expert consulted by CADTH for this review considered that U-500R should benefit diabetes patients with high insulin resistance who require more than 200 units of insulin per day, especially those who had difficulty controlling their blood glucose using the standard U-100 formulary. The clinical expert indicated that although the pivotal trial was conducted in adult patients (18 years to 75 years of age) affected by T2DM, there is no reason to believe that this new formulary of drug cannot be used in children or patients older than 75 years of age, if there is a need to administer high-dose insulin to control their blood glucose level.
Assessing Response to Treatment
The clinical expert consulted by CADTH for this review considered that blood glucose levels should be monitored by measurement of hemoglobin A1C level every 3 months and, when stable, at 6-month intervals. The aim is to ensure that overall glycemic control (hemoglobin A1C) is maintained or improved, while minimizing or reducing the risk of hypoglycemia compared to other insulins.
Discontinuing Treatment
The original treatment plan should be discontinued if the patient experiences increased hypoglycemia despite dose adjustments; different insulin preparations or different regimens should be used.
Prescribing Conditions
The clinical expert consulted by CADTH indicated that at the level of the large doses of insulin used in U-500R, a practitioner with expertise in diabetes may need to participate in the assessment and decision-making to transition from standard insulin to U-500R. However, U-500R could be prescribed by primary care physicians as well.
Clinician Group Input
This section was prepared by CADTH staff based on the input provided by clinician groups.
One clinician group input on behalf of clinician members from the Diabetes Canada Professional Section was received on the reimbursement review of U500-R.
Diabetes Canada is a national health charity that represents Canadians living with diabetes or prediabetes. The clinicians noted the priorities of their mission as diabetes prevention, care, and cure. They aim to provide evidence-based tools to health care providers. The professional members from Diabetes Canada, herein noted as the Diabetes Canada Professional Section, comprises clinicians and health care professionals, researchers, academics, and students working or studying in the field of diabetes. The Professional Section was informed to provide an opportunity for members to connect and collaborate, the most up-to-date information on the prevention and management of diabetes in Canada, and access to resources and tools which help support practice.
Unmet Needs
The clinical members from the Diabetes Canada Professional Section noted that T2DM is a disorder of insulin resistance (IR) and relative insulin deficiency. To minimize the development of diabetes-related complications, the clinicians note that achieving and maintaining near-normal blood glucose levels is an important goal in the majority of patients. The clinical members added that non-pharmacological treatments include behavioural interventions, such as good nutrition, regular physical activity, and healthy body weight, should be an important part of the management. For pharmacological options, the clinical members noted that there are a number of pharmacological options which address both IR and the relative insulin deficiency. They also added that the current treatment paradigm requires both non-pharmacological and pharmacological treatments to achieve glycemic and other targets. Insulin was noted by the clinical members as an available pharmacological option that can be used by patients throughout their diabetes journey. However, the clinical members added that some patients, despite the use of multiple therapies, require very high doses of insulin per day due to their IR, leading to suboptimal glycemic control.
The treatment goals as noted by the clinical members were to lessen diabetes complications, independent of the treatment’s glucose-lowering properties. If that is not possible, the clinical members note that an ideal treatment would lower glucose levels with minimal hypoglycemia and decrease patient burden (e.g., fewer injections and greater comfort).
The clinical members added that patients living with T2DM who are currently requiring more than 200 units per day to achieve glycemic targets represents the population with the greatest unmet need for U-500R.
Place in Therapy
The clinical members noted that the drug under review (U-500R) would replace the existing insulin therapies for appropriate patients because it possesses both basal and bolus properties. They also added that the use of U-500R would allow patients to use fewer injections of insulin and simplify the regimen (i.e., the patients would use 1 type of insulin instead of 2 different kinds). The clinical members noted that U-500R can be used in combination with other antihyperglycemic therapies, similar to what is currently occurring with other insulins. The clinical members also provided the following quotation to demonstrate the role of U-500R in the current treatment paradigm:
I am a clinical endocrinologist. I started using Humalog U-500R when it was still special order through Health Canada Special Access Program and only available in vials and administered with syringes. My first patient had type B severe insulin resistance syndrome and…maxed out on 1,800 units of insulin per day. That patient has been on insulin for eight years now after receiving his treatment. With the 1,800 units/day and the vial and syringe method, the patient was still only taking four injections per day. Subsequently…I started using U-500R on significantly insulin resistance patients (none with type B), first through the Health Canada Special Access Program and subsequently in the form of U-500R when it became commercially available.
When asked whether or not it would be appropriate to recommend that patients try other treatments before initiating treatment with the drug under review, the clinical members noted that it would be appropriate for patients to be placed on the usual insulin therapies (100 units/mL, 200 units/mL, or 300 units/mL concentrations) first. They added that patients should have insulin requirements greater than 200 units per day. In the case of T2DM, the clinical members added that not only should patients also be on metformin, a GLP-1 receptor agonist, and an SGLT2 inhibitor, they should be maintained on these medications, if tolerated, to improve their insulin sensitivity and/or lower insulin requirements. The clinical members added that access to all these medications may not be possible depending on provincial formularies. The clinical members noted that only after being placed on the usual insulin therapies and accompanying therapies, if glycemic control remains inadequate or the number of injections and/or volume intolerable, would U-500R be appropriate.
The group of clinical members also added that U-500R would not affect the sequencing of other therapies and it would be a substitute for existing insulin therapy for patients requiring more than 200 units per day who are not achieving glycemic targets.
Patient Population
When asked which patients would be best suited for treatment with the drug under review, the clinical members noted that patients requiring more than 200 units per day of insulin and who are continuing to struggle to achieve glycemic targets or find the volume or insulin and/or number of injections intolerable would be best suited for treatment with U-500R. They added that if access is available and there are no contraindications, these patients should be placed on metformin, a GLP-1 receptor, and a SGLT2 inhibitor, and if they still require more than 200 units per day of insulin, U-500R should be used instead. Additionally, the clinical members noted that patients who are living with obesity or weigh more than 100 kg and require more than 2 units/kg per day of insulin could also benefit from U-500R. The clinical members group reiterated that patients best suited for treatment are those identified as requiring more than 200 units of insulin per day despite the concomitant use of metformin, a GLP-1 receptor, and a SGLT2 inhibitor (assuming there is access to these and no contraindication).
The clinical members added that the least suitable patients are those requiring less than 200 units per day of insulin.
Assessing Response to Treatment
The clinical members noted that response to treatment can be assessed through patients requiring fewer insulin injections, reporting greater injection site comfort, and demonstrating improved hemoglobin A1C levels. Similarly, the clinical members noted that a clinically meaningful response to treatment would be the requirement of fewer insulin injections, greater injection site comfort, and improved hemoglobin A1C levels. The clinical members noted that treatment response should be assessed every 3 months for hemoglobin A1C and, when stable, at 6-month intervals.
Discontinuing Treatment
The clinical members noted that treatment can be discontinued if the patient is experiencing increased hypoglycemia despite dose adjustments.
Prescribing Conditions
The most appropriate setting for treatment with U-500R was noted by the clinical members as any outpatient setting.
The clinical members noted that U-500R should be initiated and monitored by a practitioner with expertise in diabetes management. They also added that health care providers who prescribe and manage patients on U-500R comment that dosing can be “tricky,” given than U-500R has unique kinetic properties and is generally given without basal insulin. Additionally, the clinical members added that patients who take U-500R should be followed by a care team that includes diabetes educators who can provide additional teaching, training, and support to ensure safety and optimal outcomes on the medication.
Additional Considerations
The clinical members added that practitioners have generally found better results in their patients on U-500R, along with improved convenience with the medication and lower overall cost to the patient.
Drug Program Input
The drug programs provide input on each drug being reviewed through CADTH’s reimbursement review processes by identifying issues that may impact their ability to implement a recommendation. The drug program input for U-500R was led by the New Brunswick Drug Program, with participation by other jurisdictions in Canada. Drug program input on implementation questions and corresponding responses from the clinical expert consulted by CADTH are summarized in Table 4.
Table 4: Summary of Drug Plan Input and Clinical Expert Response
Drug program implementation questions | Clinical expert response |
|---|---|
For this product, is it sufficient to only demonstrate efficacy in reducing hemoglobin A1C levels vs. comparing hemoglobin A1C reduction using a basal-bolus insulin regimen? | No. The clinical question of relevance is whether this product performs better than routine care with other insulins with respect to glycemic control, hypoglycemic events, and weight gain. The pivotal study did not address this. |
The treat-to-target study excluded T1DM. Is there evidence to support safety and efficacy in the T1DM population? | It is likely that U-500R will be used mostly in patients with T2DM. |
T1DM = type 1 diabetes mellitus; T2DM = type 2 diabetes mellitus.
Sponsor’s Summary of the Clinical Evidence
Note that the clinical evidence summarized in this section was prepared by the sponsor in accordance with the CADTH tailored review process and has not been modified by CADTH.
Pivotal Study
Eli Lily Canada reached out CADTH for guidance on the correct way to complete the following table because U-500R has been available in Canada for more than 10 years and internationally. On March 2nd, 2021, CADTH advised that we focus on the pivotal Phase IV trial, or IBHC study (Hood RC et al. 2015)a that is referenced in the approved product monograph for U-500R (Personal communication from CADTH, March 2nd, 2021).
Table 5: Details of Included Study
Criteria | Description |
|---|---|
Study design | 24-week, open-label, multicenter (US only) RCT |
Population | Patients 18–75 years with T2DM, HbA1c 7.5–12.0% and BMI ≥25 kg/m2 Ongoing treatment (≥3 months) with high-dose U-100 insulin (201–600 U/day) with or without other OADs at stable doses |
Location | 43 investigative sites in US and Puerto Rico |
Randomization (N) | 325, 162 TID 163 BID |
Inclusion criteria | T2DM; age 18-75 years, inclusive; BMI ≥25 kg/m2; HbA1c 7.5% to 12.0%, inclusive; current U-100 users on between >200 and ≤600 units/day for ≥3 months; concomitant medications may have included metformin, DPP-4 inhibitors approved for use with insulin at the time of study entry, pioglitazone, and/or SUs/glinides at a stable dose for ≥3 months |
Exclusion criteria | History of >1 episode of severe hypoglycemia within the prior 6 months; current continuous subcutaneous (SC) insulin infusion therapy; use of U-500R insulin in prior 3 months; use of rosiglitazone, once- or twice-daily glucagon-like peptide-1 receptor agonist, pramlintide, or other injectable or oral antihyperglycemic therapy not listed in inclusion criteria in the prior 3 months; use of once-weekly GLP-1 receptor agonist in the prior 4 months; use of weight loss drugs in the prior 3 months; bariatric surgery; malignancy; cardiac disease with functional status New York Heart Association (NYHA) Class III or IV. |
Intervention | 4-week screening period followed by randomization to U-500 TID or BID dosing 24-week treatment period (12-week intensified dose-titration phase and 12-week maintenance dose-titration phase) |
Comparator | TID versus BID |
Phase | IV |
Run-in | Baseline 4 weeks, treatment 24 weeks |
Double-blind | Open-labeled |
Follow-up | N/A |
Primary end point | Change of HbA1c from baseline to the end of 24 weeks treatment both between treatments and within treatment |
Secondary and exploratory end points | Change in FPG from baseline to the end of 24 weeks between TID and BID; change from baseline 7-point SMBG profiles; glycemic variability as measured by within-group and between-group SD of SMBG values; time to reach HbA1c targets between TID and BID; TDD between TID and BID at 24 week values; reduction in volume of insulin from baseline to 24-week values for both between treatment groups and within treatment; death; AE; SAE; TEAE; WDAE; and hypoglycemia |
Publication | Hood RC et al. Two treatment approaches for human regular U-500 insulin in patients with type 2 diabetes not achieving adequate glycemic control on high-dose U-100 insulin therapy with or without oral agents: a randomized, titration-to-target clinical trial. Endocr Pract. 2015;21(7):782-793) |
AE=adverse events; BID=twice daily; BMI=body mass index; DPP-4=dipeptidyl peptidase-4; FPG=fasting plasma glucose; GLP-1=glucagon-like peptide-1; HbA1c=glycated hemoglobin; NYHA=New York Heart Association; OAD=oral anti-hyperglycemic drug; RCT=randomized controlled trial; SAE=serious adverse events; SMBG= self-monitoring blood glucose; SC=subcutaneous; SUs=sulfonylureas; T2DM=type 2 diabetes mellitus; TDD=total daily dose; TID= thrice daily; US=United States; WDAE=withdrawal due to adverse events
Source: Clinical Study Reporta
Description of Study
This was a Phase IV, US only, multicenter, randomized, open-label, 2-arm, parallel, noninferiority study in insulin resistant subjects with T2DM who had inadequate glycemic control on between >200 and ≤600 units/day of U-100 insulins/analogues with or without OADs, which assessed efficacy and safety of initiating and intensifying U-500R insulin using 2 different treatment algorithms (TID versus BID).a
Population
Inclusion and Exclusion Criteriaa
The study population was adult patients (18–75 years) affected by T2DM with HbA1c 7.5–12.0% and body mass index (BMI) ≥25 kg/m2 at baseline and ongoing treatment with high-dose U-100 insulin (201–600 U/day) with or without other OADs (including metformin, dipeptidyl peptidase-4 inhibitors approved for use with insulin at the time of study entry, pioglitazone, and/or sulfonylureas /glinides) at stable doses for ≥ 3 months. Exclusion criteria included a history of >1 episode of severe hypoglycemia within the prior 6 months, current continuous subcutaneous insulin infusion therapy, use of U-500R insulin in prior 3 months, use of rosiglitazone, once- or twice-daily glucagon-like peptide-1 receptor agonist, pramlintide, or other injectable or oral antihyperglycemic therapy not listed in inclusion criteria in the prior 3 months, use of once-weekly glucagon-like peptide-1 receptor agonist in the prior 4 months, use of weight loss drugs in the prior 3 months, bariatric surgery, malignancy, and cardiac disease with functional status New York Heart Association Class III or IV.
Baseline Characteristicsa
The 2 treatment groups were comparable, with no significant differences at baseline for the below parameters (Figure 2), apart from the ≥65 years age group. Further analysis of this age class imbalance suggested that age (≥65 and <65 years) did not impact the primary endpoint or documented symptomatic hypoglycemia rate (≤70 mg/dL). Concomitant medications not listed included statins (68.9%), antithrombotic medications (53.5%), and β-blockers (22.5%), which were comparable between the two groups. Ranges for weight, BMI, and number of injections were 58 to 201 kg, 25 to 67 kg/m2, and 2 to 10, respectively.
Interventions
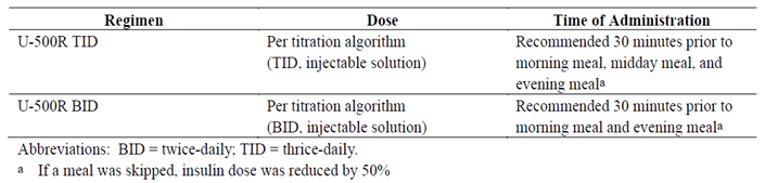
This study involved a comparison of U-500R administered subcutaneously TID with U-500R administered subcutaneously BID.a
0.5-mL U-100 syringes were used for administration for all U-500R doses ≤250 units. All U-500R doses >250 units were administered using 1.0-mL U-100 syringes.
Site personnel instructed subjects on the proper use of U-100 insulin syringes with U-500R vials and related dose conversions that were required.a
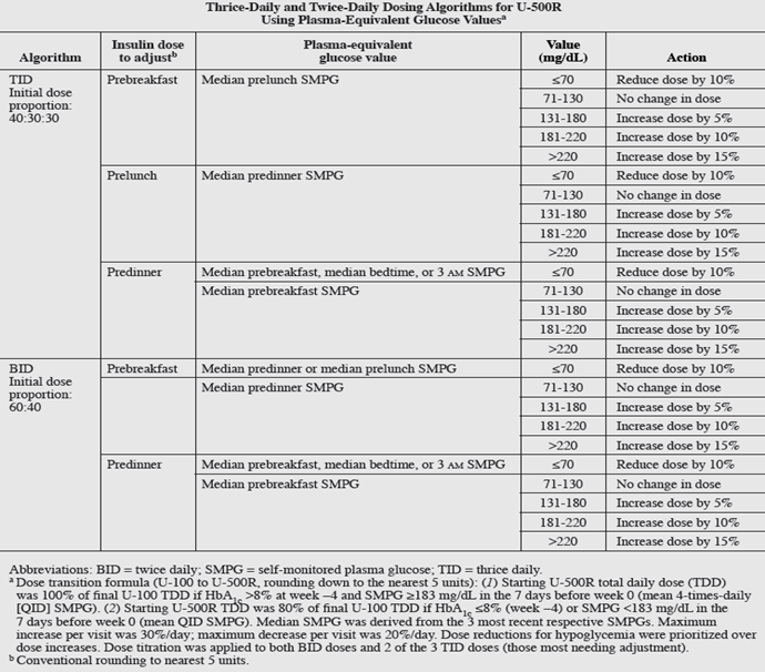
The study design included an initial 4-week screening period, during which actual U-100 insulin doses were verified and adjusted per investigators’ judgment. Assignment to TID or BID treatments was performed using a computer- generated random sequence with an interactive voice response system. Randomization was stratified by investigator site, baseline HbA1c (≤8.0% or >8.0%), total daily insulin dose (TDD, ≤300 or >300 units), and pioglitazone use. The 24-week treatment period consisted of a 12-week intensified dose-titration phase (weekly visits × 6, then every 2 weeks × 3) and subsequent 12-week maintenance dose-titration phase (visits every 3 weeks × 4). U-500R replaced U-100 insulins (predominantly basal-bolus analogue therapy) following 1:1 randomization at doses determined by the transition formula described in Initial dosing proportions were 40:30:30 (TID, breakfast:lunch:dinner) and 60:40 (BID, breakfast:dinner). The titration-to-target algorithms specified a +5 to +15% adjustment per dose as needed (−10% for median post-dose or 3 am hypoglycemia [≤70 mg/dL]) to achieve fasting/premeal self-monitored plasma glucose (SMPG) targets of 71 to 130 mg/dL. Both BID doses but only 2 of the 3 TID doses were titrated (those most needing adjustment, prioritizing dosage reductions for hypoglycemia over increases) to equalize the potential daily adjustments per algorithm at each visit (30% maximum increase; −20% maximum decrease). Algorithms were developed using input from expert protocol consultants, published reviews, and simulations using a systems physiology model of diabetes (developed in the Entelos Physiolab Modeler) based on pharmacokinetic/pharmacodynamics studies and steady-state simulations of U-500R.
Injections of U-500R were recommended 30 minutes before meals. Patients were instructed to perform SMPG 4 times daily: before each meal and at bedtime (additionally at 3 am within 48 hours of dosage increases). Seven-point SMPG profiles (pre- and 2-hours-postmeal and 3 am) were obtained on 3 nonconsecutive days just prior to weeks 0 (randomization), 12, and 24. Adverse events (AEs) and hypoglycemia were monitored throughout the study.
Patients were advised to reduce their U-500R dose by half for skipped meals and contact investigators if they experienced SMPG <50 mg/dL or severe hypoglycemia.a
Rescue therapy was initiated in the following circumstances:
If HbA1c values at Week 6 and Week 12 were both >10.5% AND the Week 12 HbA1c value was not at least 0.4% less than the Week 6 HbA1c value.
If HbA1c values at Week 12 and Week 18 were both >10.5% AND the Week 18 Hb1Ac value was not at least 0.4% less than the Week 12 HbA1c value.
Subjects who received TID U-500R were rescued by the addition of a bedtime U-500R dose (increase to 4-times daily therapy). Subjects who received BID U-500R were rescued by the addition of a pre-midday meal U-500R dose (increase to TID therapy). A suggested starting dose for the rescue would have been 10% of TDD, but this may have been modified based on investigator judgment. Every attempt was made to keep these subjects in the study. Change in treatment regimen was recorded in the electronic case report form.
One subject randomly assigned to BID treatment was inappropriately rescued at Visit 14. Prior to Visit 15 subject appropriately resumed BID assigned dosing. This subject completed the study and was included in the primary analysis, but excluded from per protocol (PP) population.a
Outcomes
A list of efficacy endpoints identified in the CADTH review protocol that were assessed in the clinical trial included in this review is provided in the pivotal trial.a These endpoints are further summarized below.
Primary Efficacy Results:a
The change in % HbA1c from baseline to the 24-week endpoint was analyzed using mixed model for repeated measures (MMRM), with treatment as fixed effect. There was a statistically significant LS Mean reduction in HbA1c from baseline to 24 weeks (p<0.001) with both TID (1.12%; 7.53% [24-week value]) and BID (1.22%; 7.41% [24-week value]) regimens. The LS mean difference between treatments (TID minus BID) in change from baseline to the end of 24 weeks was -0.10% (95% CI: [ -0.33%, 0.12%]). This 95% CI for the difference between the 2 treatments is within the pre-defined noninferiority margin of 0.4%. These results demonstrate that TID and BID achieved clinically equivalent (mutually noninferior) HbA1c lowering (1.1%-1.2% after 24 weeks).
Secondary Efficacy Results:a
A summary and analysis of subjects who reached the various HbA1c target values at endpoint but were not already at these targets at randomization showed no statistically significant differences between treatment groups at any time point.
There was no statistically significant change in FPG from baseline to the end of 24 weeks for TID (-6.7 mg/dL) or BID (-8.9 mg/dL) treatment groups.
No statistically significant differences between treatment groups were observed for the change from baseline 7-point SMBG profiles. The LS mean reduction in daily mean SMBG at 24 weeks was statistically significant (p<0.05) for both TID (30.3 mg/dL) and BID (34.9 mg/dL). The LS mean fasting SMBG for both treatments significantly decreased (p<0.05) from baseline to 24 weeks (TID 24.1 mg/dL; BID 29.2 mg/dL). This was in contrast to the lack of statistical reduction in FPG from baseline to 24 weeks.
Glycemic variability as measured by within-group and between-group SD of SMBG values also improved with TID and BID U-500R dosing as compared to U-100 insulin therapy at baseline.
No significant differences in time to reach HbA1c targets between TID and BID as measured by probability to reach target HbA1c using either Cox regression survival analysis or non-parametric log-rank test.
There were no statistically significant differences in TDD between treatment at 24 week values (mean: 343.1 and 335.0 units; for TID and BID, respectively). The Mean TDD ± SD change from first U-500R dose was 100.4 ± 66.0 and 86.0 ± 75.7 units for TID and BID, respectively. The LS Mean change from baseline U-100 TDD at randomization to the 24-week value of U-500R TDD was 55.2 and 51.4 units (both, p<0.05) for TID and BID, respectively.
There was a significant reduction in volume of insulin from baseline to 24-week values for both treatment groups but no difference between treatments (LS Mean TID: 2.19 mL; p<0.05 and BID: 2.20 mL; p<0.05). The LS Mean number of injections/day declined by 2 and 3 for TID and BID (both p<0.05), respectively, from baseline values for U-100 insulins (p< 0.001, BID vs. TID).
The adjustments to endpoint dose proportions (% [SD]: 45[10]:30[7]:27[9] for TID and 62[9]:38[9] for BID) from baseline (40:30:30 [TID]; 60:40 [BID]) were statistically different for breakfast and dinner for both treatments. The study team, however, judged that the endpoint proportions were sufficiently similar numerically to the initial dosing proportions to support recommending clinical application of the original U-500R dosing proportions.
Statistically significant (p<0.05) improvement in HbA1c (LS Mean range [-1.0% to -1.4%]) with no treatment-by-subgroup interactions (overall or by week) was observed in both the ≤2 and >2 units/kg and ≤300 and >300 units TDD subgroups. Statistically significant (p<0.05) improvement in HbA1c (LS Mean range [-1.1% to -1.3%]) with no treatment-by-subgroup interactions (overall or by week) was observed in both the metformin and no-OAD subgroups. The comparison of age groups (≥65 and <65 years) suggested that age did not impact the change in HbA1c from baseline to 24 week values.
Clinical Relevance: In a group of patients unable to achieve glycemic targets on high daily doses of 100 unit/mL insulins, U-500R given BID or TID reduced the number of daily injections, and significantly improved HbA1c, with a corresponding increase in insulin dose and weight.a
Statistical Analysis
Primary Outcome(s) of the Studies
Power Calculation:a
325 subjects were randomized (with 1:1 randomization ratio of U-500R insulin TID and BID), 260 subjects completed the 24-week treatment (with a predicted 35% screen-fail rate and 20% dropout rate). The 260 completers provided 66.4% chance to show TID and BID are equivalent, 14.4% chance to show TID is noninferior to BID but not vice-versa, 2.5% chance to show TID is superior to BID, 14.4% chance to show BID is noninferior to TID but not vice-versa, and 2.5% to show BID is superior to TID by assuming the difference in HbA1c change between TID and BID were 0%.
Primary Efficacy:a
The primary efficacy measure was to compare the TID versus BID algorithm. The result was based on a 2-sided 95% confidence interval (CI) for the mean difference in HbA1c changes from baseline (randomization) to end of study between TID dosing algorithm and BID dosing algorithm. The outcome of the primary efficacy analysis was classified according to 7 exclusive categories:
TID is superior to BID; TID is superior to BID while BID is noninferior to TID; TID is noninferior to BID; TID is noninferior to BID and BID is noninferior to TID (that is, the difference between TID and BID is not clinically meaningful [TID and BID dosing algorithms are clinically equivalent.]); BID is noninferior to TID; BID is superior to TID while TID is noninferior to BID; BID is superior to TID.
The noninferiority margin (δ) of 0.4% was used to compared treatments. Superiority was declared if the lower limit of the 2-sided 95% CI on the comparison of treatments was >0. Superiority implied noninferiority. If TID was noninferior to BID and BID were noninferior to TID, this indicated clinical equivalence between the 2 algorithms.
The repeated-subject effect models the within-subject errors that used an unstructured variance-covariance matrix. If the analysis failed to converge, the following variance-covariance matrix was used (in order) until the model converges: heterogeneous compound symmetry, compound symmetry, and first-order autoregressive.
Supporting analyses were conducted using a fixed-effect analysis of covariance (ANCOVA) that used last observation carried forward as endpoint for the full analysis set (FAS) population; the ANCOVA model included the following fixed effects: baseline HbA1c and treatment. A similar MMRM approach was conducted for the PP population (subjects included in the FAS) population who had completed the Week 24 visit without significant protocol violations) at Week 24.
Secondary Efficacy:a
The following measurements and the corresponding change from baseline at visits where the corresponding measurements were scheduled for collection was analyzed using a MMRM model similar to that used in the primary analysis for the FAS population: FPG, body weight, TDD, 7-point SMBG, time to reach HbA1c targets/values between treatment groups.
Treatment comparisons for proportion of subjects with HbA1c ≤6.5%; <7.0%; <7.5%; and <8.0% (LOCF) were analyzed using a logistic regression including treatment effect and baseline HbA1c value in the model.
Time to reach HbA1c targets were compared by using a Cox model. Baseline HbA1c and treatment were used as fixed effects.
Data Imputation Methods:a
If the baseline or all post-baseline observations were missing for a subject for a given outcome, then that subject will not be included in the analysis of that outcome.
Body weight was measured twice at each scheduled visit and the average of the 2 measurements was used for analyses that included descriptive statistics.
Missed values in the EQ-5D-5L and the VAS for Injection Site Pain were not imputed. Missed values in the TRIM-D were handled as described in the survey.
Subjects who dropped out of the study after randomization were not replaced. No imputations were made for data that were missing except for the following cases: 1) baseline and endpoint whenever the LOCF was mentioned; 2) or stated otherwise; 3) missing month and/or day date in subject’s birth date, AE starting, or AE ending date per Lilly standard for TEAE determination or SAE summary.
Subgroup Analyses:a
TDD: Analyses were conducted for units TDD subgroups (≤300 units and >300 units) and units/kg TDD subgroups (≤2 units/kg and >2 units/kg) by adding subgroup as a main effect and interaction term of treatment-by-subgroup while removing pooling site for the following parameters of interest:
HbA1c change from baseline (CFB); Adjusted 30-day rate of total hypoglycemic events (≤70 mg /dL, <50 mg/dL); Percentage of subjects with total hypoglycemic events; Body weight (absolute and percentage) in CFB.
Summary statistics were provided for all subgroups. For HbA1c and body weight analyses, an MMRM similar to that used in the primary analysis was used, replacing baseline TDD with TDD subgroup listed above (>2.0 and ≤2.0 units/kg, or baseline TDD >300 and ≤300 units, or Week 24 TDD >300 and ≤300 units). A negative binomial regression model, similar to that used in the primary analysis for hypoglycemia, was used to analyze the 30-day adjusted rate of total hypoglycemic events. A plot for each treatment and 24-week values were generated separately for the TDD subgroups (>2.0 and ≤2.0 units/kg, or baseline TDD >300 and ≤300 units, or Week 24 TDD >300 and ≤300 units).a
Additional subgroup analyses on metformin usage and non-OAD usage were conducted on Week 24 CFB in HbA1c (%), TDD (units/kg), CFB in body weight (kg), total hypoglycemia 1-year rate, nocturnal hypoglycemia 1-year rate, and non-nocturnal hypoglycemia 1-year rate. For this analysis, all hypoglycemia rates used plasma glucose ≤70 mg/dL as cutoff. These subgroup analyses used similar statistical methods as described for TDD subgroup analyses.
Sensitivity Analyses:a
A Sensitivity analyses for the primary objective of change in HbA1c with TID and BID groups were performed using MMRM on the PP population and the FAS population. The PP population LS mean change in HbA1c from baseline to 24-week value was 1.14% and 1.27% for the TID and BID groups, respectively. The LS mean difference between treatments (TID minus BID) in change from baseline at end of 24 weeks was -0.12% (95% CI: [-0.36%, 0.11%]). The CS population LS mean change in HbA1c from baseline to 24-week value was 1.13% and 1.26% for the TID and BID groups, respectively. The LS mean difference between treatments (TID minus BID) in change from baseline at end of 24 weeks was -0.13% (95% CI: [-0.36%, 0.10%]). Last observation carried forward analysis showed similar results as the MMRM results and led to the same conclusions. These results are consistent with those of the FAS population indicating that TID and BID achieved clinically equivalent HbA1c lowering.
All analyses used the 2-sided α-level of 0.05 and corresponding 95% CIs with no adjustments for multiple comparisons.
Analysis Populationsa
The FAS was used for all analyses. The FAS consisted of all patients who received at least one dose of study drug according to the randomization assignment. Supportive analyses were performed using the PP set – all patients from the full analysis set who completed week 24 without significant protocol violation.
Sponsor’s Summary of the Results
Patient Disposition
325 subjects were randomized. Overall, 260 subjects (80.0%) completed the study, with 132 subjects (81.5%) completing the TID treatment, and 128 subjects (78.5%) completing the BID treatment. The most frequent reasons for discontinuation included protocol violations (27 subjects discontinued [8.3%]) and subject decision (17 subjects discontinued [5.2%]). A total of 8 subjects (2.5%) discontinued because of an AE, and 1 subject (0.3%) died. There was no significant difference between the percentages of subjects who discontinued from TID and BID for any reason.a
U-500R TID | U-500R BID | |
|---|---|---|
Screened, N | 467 | 467 |
Randomized, N | 162 | 163 |
Discontinued, N (%) | 30 (18.5%) | 33 (20.5%) |
Reason for discontinuation, N (%) | ||
Adverse events | 4 (2.5%) | 4 (2.5%) |
Death | 0 (0%) | 1 (0.6%) |
Protocol violation | 14 (8.6%) | 13 (8.1% |
Lost to follow-up | 1 (0.6% | 3 (1.9%) |
Physician decision | 5 (3.1%) | 1 (0.6%) |
Sponsor decision | 0 (0%) | 2 (1.2%) |
Subject decision | 6 (3.7%) | 9 (5.6%) |
ITT, N | 162 | 161 |
PP, N | 128 | 124 |
Safety, N | 162 | 161 |
ITT = intention to treat; PP = Per Protocol
Source: Clinical Study Reporta
Exposure to Study Treatments
Study Treatments
Study treatments are summarized in the study overview above, and further details can be found in the Clinical Study Report.a The mean exposure to study drug in IBHC was 0.42 subject-years for the TID group and 0.42 subject-years for the BID group.a
Concomitant Medications
Exposure to concomitant medications in the study may be found in the Clinical Study Reports.a
Efficacy
Note: CADTH prefers confidence interval values on top of the p-values, however, given that U-500R was not studied by the sponsor but by a third party, we do not have the Clinical Study Reports that might contain this level of detail.
The change in % HbA1c from baseline to the 24-week endpoint was analyzed using MMRM, with treatment as fixed effect. There was a statistically significant LS Mean reduction in HbA1c from baseline to 24 weeks (p<0.001) with both TID (1.12%; 7.53% [24-week value]) and BID (1.22%; 7.41% [24-week value]) regimens. The LS mean difference between treatments (TID minus BID) in change from baseline to the end of 24 weeks was -0.10% (95% CI: [ -0.33%, 0.12%]). This 95% CI for the difference between the 2 treatments is completely within the pre-defined noninferiority margin of 0.4%. These results demonstrate that TID and BID achieved clinically equivalent (mutually noninferior) HbA1c lowering (1.1%-1.2% after 24 weeks).a
This study provides the first randomized clinical trial data rigorously assessing TID and BID titration-to-target dosing regimens for U-500R in patients with T2DM and severe insulin resistance on high-dose U-100 insulin. The U-500R treatment regimens were found to be equivalent for HbA1c reduction over 24 weeks. The formula for transitioning from U-100 TDD to U-500R, reducing the dose by 20% for lower baseline HbA1c or SMBG values, was effective, and study results support its clinical application. Additionally, both titration algorithms were efficacious and safe alternatives to high-dose/high-volume U-100 insulin therapy, regardless of baseline TDD, and both significantly reduced the number of daily injections from baseline to the end of 24 weeks. These results provide clinicians with a practical framework for initiation and titration of U-500R using either a TID or BID regimen and detailed information on potential clinical outcomes with use of U-500R in severely insulin resistance high-dose insulin-treated patients with suboptimally controlled T2DM.a
Harms
Both treatment with U-500 and titration algorithms were safe alternatives for patients who had failed glycemic control on high-dose/high-volume U-100 insulin therapy. The regimens showed no significant differences in severe hypoglycemia and small differences in non-severe hypoglycemia with weight gain associated with higher insulin TDD.
Adverse event | TID (N = 162) | BID (N = 161) |
|---|---|---|
Overall, n (%) | 123 (75.9%) | 127 (77.9%) |
Infections/Infestations | 71 (43.8%) | 74 (45.4%) |
Gastrointestinal | 36 (22.2%) | 41 (25.2%) |
Respiratory | 28 (17.3%) | 41 (25.2% |
General | 31 (19.1%) | 34 (20.9%) |
Musculoskeletal | 32 (19.8%) | 33 (20.2%) |
Nervous System/Headache | 32 (19.8%) | 25 (15.3%) |
Cardiac disorders | 6 (3.7%) | 6 (3.7%) |
Infections/infestations | 5 (3.1%) | 6 (3.7%) |
Respiratory | 3 (1.9%) | 8 (4.9%) |
(n = number of patients with event)
Source: Clinical Study Report1
There were no statistically significant differences in AEs or SAEs between the TID and BID treatment arms. Of the 162 patients randomized to the TID group, 123 (75.9%) had at least 1 TEAE and 28 (17.3%) had at least 1 SAE. Of the 163 patients randomized to the BID group, 127 (77.9%) had at least 1 TEAE and 27 (16.6%) had at least 1 SAE. Four patients (2.5%) in each treatment group discontinued due to AEs. One patient (0.6%) in the BID group died during the study (no deaths in the TID group). The incidence of SAEs as well as TEAEs was well-balanced between treatment groups.a
Overall, 10 subjects (6.2%) in the TID group and 18 subjects (11.0%) in the BID group reported TEAEs possibly related to study drug. The most frequently reported TEAEs possibly related to study drug in the TID group were hypoglycemia (3 patients, 1.9%), and abnormal weight gain (3 patients, 1.9%). The most frequently reported TEAEs possibly related to study drug in the BID group were hypoglycemia (6 patients, 3.7%), and edema peripheral (3 patients, 1.8%). There were no notable differences between treatment groups in the reporting of TEAEs possibly related to study drug.a
Serious Adverse Events
SAEs during this trial were related to patients’ advanced diabetes disease state, as indicated by diabetes duration, pre-existing comorbidities, and relatively high Charlson Comorbidity Index scores at baseline and likely poor glycemic control over time.a
A total of 55 subjects (16.9%) reported at least 1 SAE, with no difference between the treatment algorithms in the overall reporting of SAEs (TID treatment group: 28 subjects [17.3%]; BID treatment group: 27 subjects [16.6%]). The highest incidence of SAEs was in the following SOCs: cardiac disorders (3.7%); infections and infestations (3.4%); and respiratory, thoracic, and mediastinal disorders (3.4%). Rates were generally similar between the treatment groups. The most frequently reported SAEs in the TID group were acute myocardial infarction (3 subjects, 1.9%) and hypoglycemia (3 subjects, 1.9%). The most frequently reported SAEs by PT in the BID group were hypoglycemia (6 subjects, 3.7%), cardiac failure congestive (3 subjects, 1.8%), and pneumonia (3 subjects, 1.8%).a
Withdrawals Due to Adverse Events
Adverse events leading to discontinuation (2.5% of subjects [n=4] for each group) were well-balanced between treatments. No single AE leading to discontinuation was reported by more than 1 subject in either treatment group.a
Adverse Events of Special Interest: Hypoglycemia
Overall hypoglycemia incidence and rates in the TID and BID-assigned groups were similar except for (1) incidence and rates of documented symptomatic and nocturnal hypoglycemia (≤70 mg/dL) and (2) incidence of nocturnal hypoglycemia (<50 mg/dL), which were all lower for TID. Severe hypoglycemia incidence did not differ significantly between the 2 arms (1.85% [TID], 3.73% [BID]; P = .34), which was further corroborated by a Bayesian analysis estimating relative risk probabilities for BID versus TID (>1-fold risk [88% probability], >2-fold [59%], and >3-fold [38%]). Severe hypoglycemia rates were too low to be analyzed using negative binomial or recurrent-event analyses; for these comparisons, the stratified Wilcoxon rank sum test was used, which showed no significant difference between the TID and BID arms (P = .25).a
One BID-assigned patient suffered presumed prolonged severe hypoglycemia with seizure and coma/irreversible brain injury 2 days after the week-21 visit (detailed medical records were unavailable; time of day could not be determined); death occurred 2.5 weeks later. Prior to the last visit, this 72-year-old patient, who had a history of coronary artery bypass/stent placement, recorded 6 hypoglycemic plasma glucose values over the course of the study (2 at <50 mg/dL [41 and 49 mg/dL, week 21 both at 3 am]). Additional postrandomization 3 am SMPGs and the week 12 7-point SMPG profiles were not recorded, and all HbA1c values were >7%. The patient lived alone at the time of the event. An unplanned Data Monitoring Committee review for safety assessment was convened in February 2014 following the study death; no protocol changes were recommended.a
Adverse Events of Special Interest: Weight Gain
Weight gain, common with conventional insulin therapy, is expected with U-500 therapy. Increase in body weight from randomization was similar for TID and BID (P = 0.34). Weight gain per 1% reduction in HbA1c was 0.47 kg (TID) and 1.31 kg (BID) (simple linear regression analysis).a
Sponsor’s References
a. Hood RC, Arakaki RF, Wysham C, Li YG, Settles JA, Jackson JA. Two treatment approaches for human regular U-500 insulin in patients with type 2 diabetes not achieving adequate glycemic control on high-dose U-100 insulin therapy with or without oral agents: a randomized, titration-to-target clinical trial. [published correction appears in Endocr Pract. 2016;22(7):905]. Endocr Pract. 2015;21(7):782-793.
CADTH Critical Appraisal of the Clinical Evidence
CADTH conducted a critical appraisal of the clinical study for U-500R based on the summary of the evidence provided by the sponsor.
Internal Validity
The body of evidence included in the sponsor’s summary was based on publications presented by the sponsor. Therefore, outcomes and results may be subject to potential reporting bias.
Based on information available in the summary of evidence submitted by the sponsor, a number of methodological strengths of study design were demonstrated. This was a prospective, multi-centre, open-label, randomized, 2-arm comparison noninferiority phase IV trial.9 The inclusion and exclusion criteria were reasonable and justifiable. The study protocol and treatments were appropriate and are well described. Appropriate randomization procedures were described clearly in the trial. To reduce bias and achieve balance in the allocation of participants to treatment arms, randomization was stratified by site, baseline hemoglobin A1C levels (≤ 8.0% or > 8.0%), total daily insulin dose (≤ 300 units or > 300 units), and pioglitazone use. The efficacy and safety outcome measures were well-defined and clearly presented. An intention-to-treat approach was used in the main statistical analyses, which was appropriate. The analysis population, subgroup analysis, and sensitivity analysis were well-defined and appropriate. Detailed interpretations for all analysis results, including primary outcome, secondary outcomes, study quality measures, and safety indicators were provided. Overall, the design, implementation, analysis, and interpretation features as described minimize the risk of performance bias and detection bias. Because different treatment algorithms were used, this study was an open-label design. However, because the main outcomes were measured objectively, the lack of blindness or concealment of treatment allocation may have limited impact on these study findings. However, some other outcomes, such as AEs and symptoms of hypoglycemia, were assessed subjectively. Whether and to what extent the open-label design would bias the assessment of these outcomes deserve considerations.
Baseline demographic characteristics, degree of insulin resistance, and history of insulin and concomitant drug uses were generally well-balanced between the 2 treatment arms. The only observed difference was from the subgroup aged 65 and older. Further analysis of this age class suggested that imbalance in age (≥ 65 and < 65 years) did not impact the primary outcome or documented symptomatic hypoglycemia rate (≤ 70 mg/dL) or not-listed concomitant medications, including statins, antithrombotic medications, and beta-blockers.
The duration of a 4-week screening plus lead-in period and a 24-week treatment period was considered to be the minimum duration needed in a clinical trial for diabetes and of adequate duration to demonstrate a treatment effect by the clinical expert consulted by CADTH. However, longer follow-up could be more informative to assess the durability of the glycemic control benefits and to assess the impact on cardiovascular health, renal function, or survival.
Imputation for missing data was performed only for certain patients. Patients who dropped out of the study after randomization were not replaced. The authors indicated that if the baseline data or all post-baseline observations were missing for a participant for a given outcome, then that participant’s data would not be included in the analysis of that outcome. However, exactly how many individual outcome measures were missed was not presented. No mention of the handling multiplicity in statistical analysis was made; however, only 1 comparison was made for the primary outcome. As a result, the lack of clarity on handling multiplicity in the statistical analysis had no impact on the primary objective of this study. According to the clinical expert consulted by CADTH, the overall proportion of patients who discontinued from the trial was high (20%) for a short-duration study. Analysis for most outcomes was done by an intention-to-treat approach, which is appropriate. However, the study did not discuss if outcomes (especially hemoglobin A1C values) at the end of the study in discontinued subjects were measured and included in the analysis. If the outcomes at the end of the study for all or a major proportion of discontinued patients were not measured, and therefore not included in the analysis, the results may be biased. However, there was no imbalance in discontinuation between treatment arms observed overall or for specific reasons of discontinuation.
The reduction in hemoglobin A1C levels from baseline to the end of 24 weeks of treatment was the primary efficacy outcome assessed in the study.9 There was a statistically significant reduction in hemoglobin A1C levels from baseline to the end of treatment (P < 0.001) in both the thrice-daily (1.12%) and twice-daily (1.22%) regimens, but there was no between-treatment (thrice daily minus twice daily) difference in change from baseline to the end of 24 weeks (–0.10%; 95% CI, –0.33% to 0.12%). This 95% CI for the difference between the 2 treatments is within the predefined noninferiority margin of 0.4%. Based on these findings and the margin assumption, the authors considered that noninferiority between the thrice-daily and twice-daily regimens was achieved in this study in terms of lowering hemoglobin A1C levels.9 However, defining a margin of equivalence is challenging and often arbitrary.17 The predefined margin of 0.4% is wide compared with the estimated between-group difference of 0.1%. No clear explanation was provided about how and why this noninferiority margin was determined in the trial, and the analysis to help draw a conclusion on noninferiority seemed insufficient. However, the clinical expert consulted by CADTH indicated that the reduction in hemoglobin A1C levels suggests a clinically relevant change.
In summary, for the submitted study, baseline characteristics were generally well-balanced, execution of the study was well implemented, and data analysis was generally appropriate. A main concern of this study was the large proportion of discontinuations. If the outcomes at the end of the study for all or a major proportion of discontinued patients were not measured, and therefore not included in the analysis, the results may be biased. The choice of noninferiority margin and the methods used by the study team to analyze noninferiority equivalence were not considered the most stringent. Some outcomes, such as AEs and symptoms of hypoglycemia, were assessed subjectively. Whether and to what extent the open-label design would bias the assessment of these outcomes deserve consideration.
External Validity
The study submitted by the sponsor was conducted in the US from February 2013 to May 2014. There are some differences in terms of racial and/or ethnic composition and methods of drug delivery between the US and Canada. The study submitted by the sponsor included adult patients (18 years to 75 years of age) affected by T2DM. However, the target of this drug was diabetes patients with high insulin resistance, with no restriction on age- or type-specific indication. The main deficit appears to be a lack of a randomized comparison to conventional care for the target population. The clinical expert consulted by CADTH suggested that a thrice-daily regimen versus twice-daily regimen is not the most relevant clinical question. CADTH staff carefully assessed the impact of these factors on the generalizability of the drug under review to Canadian diabetes patients:
Racial and/or ethnic composition. There are differences in the racial and/or ethnic composition between the US and Canada, with larger numbers of Black and Hispanic people in the US,21 whereas there is a large Asian population in Canada.22 There are some racial and/or ethnic differences in pathoetiology and pathology of diabetes, with an increased risk of developing diabetes and increased insulin resistance at the same BMI in some Asian populations, such as in India, compared with other racial and/or ethnic groups.23 However, according to the clinical expert consulted by CADTH, racial and/or ethnic concentrations should not impact drug efficacy once someone requires more than 200 units per day.
Type of diabetes. The study submitted by the sponsor enrolled patients affected by T2DM with high insulin resistance. Generalizability to T1DM may be limited.
Drug delivery method. In the study submitted by the sponsor, needles and syringes were used to administer insulin. However, pens with prefilled insulin cartridges are used to deliver the drug under review in Canada. Compared with needles and syringes, pens with prefilled insulin cartridges are more convenient for patients to use and can deliver insulin with higher precision. Because these were not in the pivotal study, results that are affected by adherence may not reflect clinical practice use.
Comparison with standard care. This trial was designed to compare thrice-daily and twice-daily regimens. This design assumes that to administer U-500R, thrice daily and twice daily are the only 2 options and they are equivalent in terms of efficacy and safety profile. The clinical expert consulted by CADTH suggested that thrice daily versus twice daily U-500R is not the relevant clinical question. Instead, the comparison should be made with standard of care using U-100 insulin.
In summary, the baseline demographic and disease characteristics of the patients enrolled in the trial are generally representative of patients with T2DM and high insulin resistance seen in clinical practice in Canada, with the exception of race and/or ethnic composition. The use of needles or syringes in the study instead of the pens with prefilled insulin cartridges that are used in Canada may limit the generalizability of outcomes that are influenced by adherence and/or dose precision because the clinical assumption is that adherence and dose precision is better with the use of the U-500R pen.7 A noticeable limitation of the study submitted by the sponsor is that it made a comparison only between thrice daily and twice daily to administer U-500R. The clinical expert consulted for this review indicated that a comparison with standard of care using U-100 insulin would be more informative clinically.
Economic Review
The current review is for biosynthetic human insulin U-500R (Entuzity) for adults and children with diabetes mellitus requiring more than 200 units of insulin per day.
Economic Information
Summary of Sponsor’s Economic Information
The sponsor submitted a cost comparison of 500 units/mL human biosynthetic insulin (U-500R insulin, Entuzity) compared with other available human and analogue insulins used for the treatment of diabetes mellitus in patients requiring more than 200 units of insulin per day.27 Table 8 includes publicly available individual and weighted average costs per unit for each included brand of insulin calculated using IQVIA public claims data,28 as well as the incremental cost of each comparator’s weighted average cost compared to that of U-500R insulin.
Table 8: Units Costs by Brand of Insulin
Drug and comparator | 3-mL prefilled disposable pen cost per unit | 3-mL reusable pen cartridge cost per unit | 10-mL vial cost per unit | Weighted average cost per unita | Weighted average unit cost vs. U-500R insulinb |
|---|---|---|---|---|---|
Biosynthetic Human Insulin U-500R (Entuzity) | $0.0316 | NA | NA | $0.0316 | Reference |
Basal human insulins | |||||
Insulin isophane U-100 (Humulin N) | $0.0325 | $0.0325 | $0.0249 | $0.0307 | –$0.0009 |
Insulin isopane U-100 (Novolin ge NPH) | NA | $0.0325 | $0.0248 | $0.0318 | $0.0002 |
Long-acting (basal) insulin analogues | |||||
Insulin degludec U-100 (Tresiba) | $0.0743 | NA | NA | $0.0743 | $0.0427 |
Insulin degludec U-200 (Tresiba) | $0.0743 | NA | NA | $0.0743 | $0.0427 |
Insulin glargine U-100 (Basaglar) | $0.0464 | $0.0464 | NA | $0.0464 | $0.0148 |
Insulin glargine U-100 (Lantus) | $0.0619 | $0.0619 | $0.0617 | $0.0619 | $0.0303 |
Insulin glargine U-300 (Toujeo) | $0.0587 | NA | NA | $0.0587 | $0.0271 |
Insulin detemir U-100 (Levemir) | $0.0743 | $0.0736 | NA | $0.0740 | $0.0424 |
Prandial human insulins | |||||
Regular human insulin U-100 (Humulin R) | NA | $0.0325 | $0.0249 | $0.0303 | –$0.0013 |
Regular human insulin U-100 (Novolin ge Toronto) | NA | $0.0318 | $0.0243 | $0.0304 | –$0.0012 |
Rapid-acting (prandial) insulin analogues | |||||
Insulin aspart U-100 (NovoRapid) | $0.0425 | $0.0408 | $0.0302 | $0.0387 | $0.0071 |
Insulin glulisine U-100 (Apidra) | $0.0354 | $0.0351 | $0.0266 | $0.0353 | $0.0037 |
Insulin lispro U-100 (Humalog) | $0.0398 | $0.0400 | $0.0302 | $0.0387 | $0.0071 |
Insulin lispro U-200 (Humalog) | $0.0370 | NA | NA | $0.0370 | $0.0054 |
Rapid-acting analogue insulin weighted averagea | $0.0407 | $0.0400 | $0.0299 | $0.0393 | $0.0077 |
NA = not applicable; vs. = versus.
aWeighted average per-unit cost based on national (excluding Quebec) IQVIA market shares for formulations within each comparator and within the rapid-acting insulin analogue class.
bWeighted average cost of comparator minus the weighted average cost of U-500R insulin. Negative values indicate U-500R insulin is more expensive on a unit-to-unit basis.
Source: Adapted from Sponsor’s Submission Template, Table 5.27
The sponsor assumed that basal and prandial insulins would each account for 50% of daily units and that all insulins would be used at a daily dose consistent with that of U-500R insulin at week 24 of the IBHC randomized trial,9 or 339.1 units per day. Daily costs, annual costs, and annual costs relative to U-500R insulin are reported in Table 10 for basal and prandial regimens considered by the sponsor as well as an additional analysis considering the potential savings related to reduced use of insulin needles with U-500R insulin.
The sponsor reported that the use of U-500R insulin would be associated with an incremental cost of $62 to $136 per patient per year compared to human insulins and savings of $1,392 to $3,119 per patient per year when compared to insulin analogue regimens (composed of a basal insulin and the weighted average cost of prandial insulins) when only drug acquisition costs are considered. When needle costs are included, the use of U-500R insulin would be associated with a savings of $105 to $3,360 per patient per year for patients using more than 200 units of insulin per day.
Table 9: Summary of the Sponsor’s Submitted Cost Comparison for Insulin Regimens
Drug and comparator | Total insulin units per day | Basal units per day | Prandial units per day | Weighted average daily cost | Weighted average annual cost | Annual cost difference relative to U-500R insulina | Annual cost difference relative to U-500R insulin including needle costsa,b |
|---|---|---|---|---|---|---|---|
Biosynthetic Human Insulin U-500R (Entuzity) | 339.1 | NA | NA | $10.72 | $3,911 | Reference | Reference |
Basal human insulin combinations | |||||||
Humulin N and Humulin R | 339.1 | 169.55 | 169.55 | $10.34 | $3,775 | –$136 | $105 |
Novolin ge NPH and Novolin ge Toronto | 339.1 | 169.55 | 169.55 | $10.55 | $3,849 | –$62 | $179 |
Insulin analogue combinations | |||||||
Insulin degludec U-100 (Tresiba) and RAI Average | 339.1 | 169.55 | 169.55 | $19.26 | $7,030 | $3,119 | $3,360 |
Insulin degludec U-200 (Tresiba) and RAI Average | 339.1 | 169.55 | 169.55 | $19.26 | $7,030 | $3,119 | $3,360 |
Insulin glargine U-100 (Basaglar) and RAI Average | 339.1 | 169.55 | 169.55 | $14.53 | $5,304 | $1,392 | $1,633 |
Insulin glargine U-100 (Lantus) and RAI Average | 339.1 | 169.55 | 169.55 | $17.16 | $6,263 | $2,352 | $2,592 |
Insulin glargine U-300 (Toujeo) and RAI Average | 339.1 | 169.55 | 169.55 | $16.62 | $6,065 | $2,154 | $2,394 |
Insulin detemir U-100 (Levemir) and RAI Average | 339.1 | 169.55 | 169.55 | $19.21 | $7,012 | $3,100 | $3,341 |
NA = not applicable; RAI = rapid-acting insulin analogues.
aComparator combination minus U-500R insulin; negative values indicate U-500-R insulin is more expensive than the comparator combination.
bNeedle costs include 3 injections daily for U-500R insulin ($361.24 per year) and 5 injections daily for all other insulins ($602.07 per year), assuming a cost of $0.3299 per injection for the cost of pen needles as reported by the sponsor for BD Ultra-Fine Nano pen needles on the Saskatchewan formulary.
Source: Adapted from Sponsor’s Submission Template, Table 6.27 Weighted average per-unit cost based on national (excluding Quebec) IQVIA market shares for formulations within each comparator and within the rapid-acting insulin analogue class.
CADTH Appraisal of the Sponsor’s Economic Information
CADTH identified several key limitations to the sponsor’s analysis that have notable implications on the economic analysis:
Assumption of clinical similarity is uncertain: U-500R insulin was not compared to U-100 insulin in a clinical trial nor did the pivotal trial include patients with T1D1. Instead, patients with T2D2 who had been using between 201 and 600 units of insulin per day for at least 3 months were randomized in the IBHC trial to U-500R insulin either twice or 3 times daily. Over the 24 weeks of the trial, glycemic control was improved from baseline in both groups, while patients in both groups experienced a moderate weight gain and an increase in the mean daily number of units of insulin used. Additionally, patients in the twice-daily group experienced higher rates of hypoglycemia than those in the 3 times daily group.
CADTH was unable to address this limitation in reanalyses because the primary assumption underlying a cost comparison is clinical similarity between comparators; as such, the estimated cost savings are uncertain.
Total daily dose of insulin increased during the U-500R clinical trial: In the pivotal IBHC trial,9 patients who had used high-dose U-100 insulin for at least 3 months were randomized to U-500R insulin twice or 3 times daily for 24 weeks. At baseline, patients in the IBHC trial were using a mean total daily dose of 287.5 units of U-100 insulin, or 2.4 U/kg. At the end of 24 weeks, the mean total daily dose of U-500R insulin was 339.1 units, with this change due to both an increased mean dose per weight to 2.7 U/kg, and a mean increase in weight of 4.9 kg. Patients in the trial were not new to insulin use and had been using high-dose insulin for at least 3 months before randomization; therefore, in the absence of a randomized trial comparing U-500R insulin to U-100 insulin, this suggests that the sponsor’s 1:1 unit replacement between U-500R insulin and U-100 insulin is not an appropriate representation of the available data.
CADTH reanalyses considered the mean daily dose at baseline from the IBHC trial for U-100 comparators and the mean daily dose at 24 weeks for U-500R insulin. U-200 insulin and U-300 insulin were not considered for this reanalysis due to an absence of data on relative units administered per day. CADTH recalculated the weighted average cost of rapid-acting (prandial) insulin analogues to remove comparators with concentrations greater than U-100 and to incorporate Ontario Drug Benefit (ODB) list prices as of April 2021 and the market shares of each rapid-acting insulin analogue in the fourth quarter of 2020, the last period for which IQVIA Pharmastat claims data were available at the time of the analysis.
Missing comparator: The sponsor excluded insulin lispro U-100 (Admelog) because no public price was available at the time of their analysis. The ODB Formulary has since listed Admelog at $45.00 per package of five 3-mL prefilled pens or cartridges, and $22.70 per 10-mL vial (see Table 14).29 Although its current use may not be widespread, the list price of Admelog is substantially less expensive than the other basal insulin analogues, and thus may represent savings to public payers relative to the current comparators. Thus, using an overall weighted average to estimate the daily and annual cost of all prandial insulin analogues may not represent relative costs in the future.
CADTH reanalyses included Ademelog-brand insulin lispro and compared the costs of all combinations of U-100 basal and prandial insulin analogues to that of U-500R insulin.
List prices may not reflect costs paid by public plans: The sponsor’s analysis and the CADTH analysis consider publicly available list prices for the included insulin comparators and do not reflect any confidential pricing agreements that may be in place. The existence of such agreements would lower or eliminate the savings associated with the reimbursement of U-500R insulin at the submitted price.
CADTH was unable to address this limitation in reanalyses.
Price of insulin needles is overestimated and not always applicable: The sponsor used the list price of BD Ultra-Fine Nano pen needles (pseudo-DIN: 97799527) on the Saskatchewan formulary to represent the cost per unit of all insulin needles within the analysis. At $0.3299 per needle (updated to $0.3456 in April 2021), the BD Ultra-Fine Nano list price represents 1 of the most expensive needles listed in the Saskatchewan formulary,30 and thus is likely to inflate the savings associated with the need for fewer injections with the use of U-500R insulin relative to U-100 insulins. Other available pen needle list prices on the formulary range from $0.1999 to $0.3905 per unit, with most reimbursed options costing around $0.28 per unit. Additionally, not all public health care payers reimburse insulin needles, or may do so with annual caps or grants limiting the amount qualifying patients may be reimbursed for, thus complicating the applicability of such analyses.31
CADTH reanalyses consider the potential savings associated with a reduced use of insulin needles (i.e., omitting insulin costs), and assumed a mean cost of $0.2800 per needle.
CADTH Reanalyses of the Economic Information
CADTH reanalyses considered the annual cost of U-500R insulin compared with available U-100 insulin regimens when considering:
The total daily dose of insulin units used by patients in the IHBC trial at baseline of U-100 insulin comparators and at week 24 for U-500R insulin.
A comparison of the weighted average cost of each basal (long-acting) insulin analogue in combination with each prandial (rapid-acting) insulin analogue comparator rather than using an overall weighted average of rapid-acting insulin analogues. Fourth quarter 2020 IQVIA Pharmastat claims data were used to estimate the weighted average cost of each brand of insulin analogue.
The inclusion of insulin lispro U-100 (Admelog).
When compared with basal and prandial human insulins at the mean baseline dose in the IHBC trial, the use of U-500R insulin at the mean dose reported for week 24 resulted in increased costs of between $496 and $1,334 per patient per year, depending on the brand and format used (see Table 10).
Table 10: CADTH Cost Comparison of U-500R Insulin Compared to U-100 Human Insulin Combinations
Insulin combination | Total insulin units per day | Basal units per day | Prandial units per day | Weighted average daily cost ($) | Weighted average annual cost ($) | Annual cost difference relative to U-500R insulin ($) |
|---|---|---|---|---|---|---|
Biosynthetic Human Insulin U-500R (Entuzity) | 339.1 | NA | NA | $10.71 | $3,911 | Reference |
Human insulin combinations | ||||||
Humulin N and Humulin R vials | 287.5 | 143.8 | 143.8 | 7.15 | 2,611 | –1,300 |
Humulin N and Humulin R pens/cartridges | 287.5 | 143.8 | 143.8 | 9.36 | 3,415 | –496 |
Novolin ge NPH and Novolin ge Toronto vials | 287.5 | 143.8 | 143.8 | 7.06 | 2,577 | –1,334 |
Novolin ge NPH and Novolin ge Toronto pens and cartridges | 287.5 | 143.8 | 143.8 | 9.25 | 3,375 | –535 |
NA = not applicable.
In the CADTH reanalyses, U-500R insulin remains cost saving relative to most combinations of U-100 insulin analogues when ODB list prices and doses from baseline and week 24 from the IHBC trial were considered. Annual savings associated with the use of U-500R insulin range from $366 to $2,043 per patient, with the only exception being the combination of insulin glargine (Basaglar) and insulin lispro (Admelog), 2 products released after the expiry of the patent for the originator brands. This combination is $140 per patient per year less expensive than U-500R insulin (see Table 11).
The relative cost of U-500R insulin compared to individual formulations of included comparators on a unit-to-unit basis can be found in Appendix 1, Table 13.
Table 11: CADTH Cost Comparison of Annual Cost of U-500R Insulin ($3,911) Compared With U-100 Insulin Analogue Combinations
U-100 prandial insulin analogues | |||||
|---|---|---|---|---|---|
U-100 basal insulin analogues | Insulin Aspart (NovoRapid) | Insulin glulisine (Apidra) | Insulin lispro (Admelog) | Insulin lispro (Humalog) | |
$2,126 | $1,841 | $1,335 | $2,053 | ||
Insulin degludec (Tresiba) | $3,900 | Cost: $6,026 Inc.: $2,115 | Cost: $5,741 Inc.: $1,831 | Cost: $5,235 Inc.: $1,324 | Cost: $5,953 Inc.: $2,043 |
Insulin detemir (Levemir) | $3,880 | Cost: $6,006 Inc.: $2,095 | Cost: $5,721 Inc.: $1,810 | Cost: $5,215 Inc.: $1,304 | Cost: $5,933 Inc.: $2,022 |
Insulin glargine (Basaglar) | $2,436 | Cost: $4,561 Inc.: $651 | Cost: $4,277 Inc.: $366 | Cost: $3,771 Inc.: –$140 | Cost: $4,489 Inc.: $578 |
Insulin glargine (Lantus) | $3,248 | Cost: $5,373 Inc.: $1,462 | Cost: $5,089 Inc.: $1,178 | Cost: $4,582 Inc.: $671 | Cost: $5,301 Inc.: $1,390 |
inc. = incremental cost; U-100 = 100 units/mL; U-500R = 500 units/mL regular insulin (Entuzity).
Note: Weighted average cost by formulation is based on fourth quarter 2020 Pharmastat claims data. Annual costs assume 287.5 daily units of insulin as reported at baseline in the IHBC trial. Analysis assumes 50% of daily units are basal insulin and 50% are prandial, thus the annual cost of each individual component assumes 143.75 units per day. The annual cost of U-500R insulin used to calculate incremental costs for each insulin analogue combination is $3,911 and assumes the use of 339.1 units daily as reported at week 24 of the IHBC trial. All analyses use publicly available list prices.
CADTH conducted an analysis estimating the percentage reduction to the submitted price of U-500R insulin that would be required for the annual cost of U-500R to be equivalent to that of the least expensive alternatives under the CADTH reanalysis assumptions. The submitted price of U-500R insulin would need to be reduced by 3.6% for its annual cost to equal that of the least expensive insulin analogue combination, and by 13.7% and 34.1% to equal that of the least expensive human insulin combination in pens or cartridges and vials, respectively (see Table 12).
Table 12: CADTH Price Reduction Analyses
Scenario | Current list price of U-500R ($) | Reduction needed for U-500R (%) | Reduced price ($) | Savingsa ($) |
|---|---|---|---|---|
Price reduction required to equal least expensive insulin analogue combination, insulin glargine (Basaglar) and insulin lispro (Admelog) | 94.79 per 2 × 3 mL | 3.6% | 91.38 | 141 |
Price reduction required to equal least expensive human insulin combinations available in pens or cartridges, Novolin ge NPH and Novolin ge Toronto | 94.79 per 2 × 3 mL | 13.7% | 81.80 | 536 |
Price reduction required to equal least expensive human insulin combination overall, Novolin ge NPH and Novolin ge Toronto vials | 94.79 per 2 × 3 mL | 34.1% | 62.47 | 1,334 |
aSavings from the sponsor list price per patient per year.
Finally, U-500R insulin is recommended to be administered twice or 3 times daily and is likely to reduce the number of daily injections required by patients using more than 200 units of daily insulin. Assuming a cost of $0.28 per insulin pen needle and an average of 5 daily injections of lower concentration insulin, for plans that reimburse insulin needles, U-500R insulin would be associated with a cost savings of $204 per patient per year for those administering it 3 times daily and $307 per patient per year for those administering it twice daily. Such savings would be increased for patients currently using more than 5 injections per day.
Discussion
Summary of Available Evidence
The CADTH Clinical Review was based on a summary of clinical evidence provided by the sponsor and focused on the clinical study that is referenced in the approved product monograph for U-500R.
All the evidence provided in the submission is based on results from a pivotal trial conducted in the US using U-500 insulin administered by needle and syringe.9 One pharmacokinetics and pharmacodynamics study was also submitted by the sponsor.32 After consulting with the clinical expert, CADTH decided to focus the critical appraisal and the discussion on the pivotal trial,9 with references to the pharmacokinetics and pharmacodynamics study32 only if such a reference is needed.
The results of this study suggested that switching to U-500R for diabetes patients requiring more than 200 units of insulin per day may result in a hemoglobin A1C decrease and a reduction in number of injections per day from baseline to 24 weeks of treatment for both the thrice-daily and twice-daily treatment groups. However, insulin dose and weight increased from baseline to 24 weeks of treatment for both the thrice-daily and twice-daily groups; the rate of non-severe hypoglycemia was higher in the twice-daily group than in the thrice-daily group. The design and execution of the trial appear to be appropriate with respect to randomization and standardized assessment of the efficacy and safety outcomes. However, a number of issues need to be considered in the interpretation of the results.
This was a noninferior trial, with the primary objective to compare the hemoglobin A1C–lowering effect between a thrice-daily and a twice-daily regimen. A major limitation is that it compared the thrice-daily and twice-daily regimens, with the assumption that U-500R is superior to standard U-100 insulins or insulin analogues in terms of hemoglobin A1C–lowering effect, with or without OADs, for adult T2DM patients who require high doses of insulin. The sponsor made a before-after comparison in the study and used change from baseline to end of 24 weeks of treatment as the outcome measure to demonstrate the hemoglobin A1C–lowering effect of U-500R. Although the superiority of U-500R to standard U-100 insulins or insulin analogues has been discussed in the literature,7,9 no evidence from a parallel 2-arm RCT superiority study that directly compares the efficacy and safety of U-500R and standard of care using U-100 insulins or insulin analogues is available. Before-after comparison is not ideal to determine superiority between U-500R and standard of care using U-100 insulins or insulin analogues, especially in an open-label design. Of special importance in controlling glycemia for diabetes patients are awareness and compliance. Patients enrolled in the trial may become aware of the importance of appropriate glycemia control and health care providers may intensify their monitoring effort during the trial. All these may result in improvement in patient’s compliance and glycemia control, regardless of the drugs or methods of drug administrations. The clinical expert consulted for this review indicated that a comparison with standard of care using U-100 insulin regimens would be more informative clinically. For noninferiority, the authors found that the 95% CI for the difference in reduction of hemoglobin A1C levels from baseline to 24 weeks of treatment between the 2 treatments was within the predefined noninferiority margin of 0.4%. Based on these findings and the margin assumption, the authors considered that a noninferiority between the thrice-daily and twice-daily regimens was achieved in this study for hemoglobin A1C lowering.9 However, defining a margin of equivalence is most challenging and often arbitrary.17 The predefined margin of 0.4% is considered wide compared with the estimated point difference of 0.1% between the 2 groups. No clear explanation was provided about how and why this noninferiority margin was determined in the trial, and a simple comparison of the 95% CI of the difference with the predefined margin of noninferiority seems insufficient. Because of practical difficulties, this study was an open-label design and because most of the outcomes were measured objectively, the lack of blindness or concealment of treatment allocation may have limited the impact on these outcomes. However, some outcomes, such as AEs and symptoms of hypoglycemia, were assessed subjectively. Whether and to what extent the open-label design would bias the assessment of these subjective outcomes deserves consideration. Overall, the proportion of patients who discontinued from the trial was high (20%) for a short-duration study according to the clinical expert consulted by CADTH. The study did not mention if outcomes (especially hemoglobin A1C values) at the end of the study for discontinued participants were measured and included in the analysis. If the outcomes at the end of the study for all or a major proportion of discontinued patients were not measured, and therefore not included in the analysis, the results may be biased. However, no imbalance in discontinuations between the treatment arms was observed overall or for specific reasons of discontinuation. Another limitation of this study was that it did not evaluate other combination therapy approaches, including newer OADs such as SGLT2 inhibitors18,19 or injectable GLP-1 receptor agonists,18,20 which are typically accompanied by weight reduction. Participants who dropped out of the study after randomization were not replaced. Imputation for missing data was performed only for certain patients. The authors indicated that if baseline data or all post-baseline observations were missing for a participant for a given outcome, then that participant would not be included in the analysis of that outcome. However, exactly how many individual outcome measures were missed was not reported. There was no mention of handling multiplicity in the statistical analysis; however, only 1 comparison was made for the primary outcome. As a result, the lack of clarity on handling multiplicity in the statistical analysis will not affect the results on the primary objective of the study. There are also some data presentation issues. For example, total injections per day is an important outcome measure and it should be presented more clearly. However, only the number of reductions from baseline to the end of 24 weeks of treatment was provided in the original report. Fasting glucose was expressed as mg/dL in the original study, not the standard units of mmol/L.
To assess the need for U-500R in Canada, CADTH conducted a study to estimate the overall utilization of insulin as well as the proportion of patients using more than 200 units of insulin per day for public drug beneficiaries in Canadian jurisdictions.8 The total number of public drug plan beneficiaries receiving insulin gradually increased year by year from 2014 to 2017, as did the number of units claimed.8 However, the proportion of insulin beneficiaries receiving 201 to 600 units per day decreased over the same time period, from 3.1% of all insulin beneficiaries in 2014 to 2.8% in 2017, while the proportion of units claimed by patients receiving 201 to 600 units per day rose from approximately 12.0% of all insulin units reimbursed in 2014 to 13.7% in 2016, before falling again to 13.2% in 2017.8 In 2017, in Canadian provincial jurisdictions covered by public drug plans, approximately 8,600 (2.8%) diabetes patients used an average of 201 to 600 units per day.8
Interpretation of Results
Efficacy
At recruitment in the pivotal trial, study participants had been treated with regular U-100 human insulin for at least 3 months.9 Overall, the use of U-500R for 24 weeks improved glycemic control from baseline in both the thrice-daily and twice-daily groups. In addition, the number of injections in both the thrice-daily and twice-daily groups were reduced from baseline to the end of treatment.
The primary efficacy outcome assessed in the study was the comparison of reduction in hemoglobin A1C levels from baseline to the end of 24 weeks of treatment between the thrice-daily and twice-daily groups.9 There was a reduction in hemoglobin A1C levels from baseline to the end of treatment (P < 0.001) in both the thrice-daily (1.12%) and twice-daily (1.22%) regimens but there was no between-treatment (thrice daily minus twice daily) difference in change from baseline to the end of 24 weeks (–0.10%; 95% CI, –0.33% to 0.12%). However, as discussed previously, a before-after comparison is not the ideal method to assess efficacy. Improvement in glycemic control may not necessarily be attributed to switching to U-500R during the trial from U-100 insulin at baseline but may be the result of improvement of patient’s compliance during the trial. Even if we can assume a true reduction in hemoglobin A1C levels by switching to U-500R, a simple comparison between the 95% CIs for the difference between the 2 treatments and the predefined noninferiority margin may be insufficient. As discussed previously, a margin of 0.4% against a point estimation of 0.1% difference between the 2 treatment groups is considered wide, the underlying reasons of choosing this noninferiority margin were not explained explicitly, and the methods used to analyze the noninferiority equivalence in this trial could not be considered stringent.
Some people living with T2DM have marked insulin resistance, either due to an underlying syndrome (e.g., certain lipodystrophies) or due to obesity. Those individuals require high doses of insulin to overcome that resistance and decrease their blood glucose levels. Those doses often exceed 200 units per day. The usual concentration of insulin (100 units/mL) is equivalent to injecting more than 2 mL of liquid under the skin. Multiple injections at the same time are needed to administer the dose; therefore, the amount of liquid under the skin is uncomfortable and, most concerning, the absorption of the insulin is not as predictable because of the large volume. For these patients, U-500R, which provides insulin in a 500 units/mL concentration, allows self-administration of much larger doses of insulin in a smaller volume. In theory, U-500R may work better for these patients than standard of care using U-100 insulin. However, to demonstrate the superiority of U-500R to U-100 insulin, a direct comparison with a parallel 2-arm RCT is needed.
Ideally, a diabetes treatment should appropriately control glucose levels and lessen diabetes complications. To observe long-term diabetes complications, a follow-up period much longer than 24 weeks is needed. If this ideal goal is not possible within the time window of a typical clinical trial, then an alternative goal of the treatment should be to lower glucose levels with minimal hypoglycemia and decreased patient burden (e.g., fewer injections and greater comfort). The pivotal trial submitted by the sponsor achieved the alternative goal with decreased hemoglobin A1C levels and reduced number of injections per day from baseline to the end of treatment in both the thrice-daily and twice-daily groups.
Harms
The clinical expert noticed that the incidence of hypoglycemia was higher in the twice-daily arm. However, this may not be a major concern because the reported SAEs and TEAEs were similar between the 2 arms. In the pivotal trial, no major difference in SAEs or TEAEs between the thrice-daily and twice-daily treatment arms was observed.9 Four patients (2.5%) in each treatment group discontinued due to AEs. One patient (0.6%) in the twice-daily group died during the study (no deaths occurred in the thrice-daily group). The incidences of SAEs and TEAEs were well-balanced between treatment groups. Overall, 10 participants (6.2%) in the thrice-daily group and 18 participants (11.0%) in the twice-daily group reported TEAEs possibly related to the study drug. The most frequently reported TEAEs possibly related to the study drug in the thrice-daily group were hypoglycemia (3 patients, 1.9%), and abnormal weight gain (3 patients, 1.9%). The most frequently reported TEAEs possibly related to the study drug in the twice-daily group were hypoglycemia (6 patients, 3.7%), and edema peripheral (3 patients, 1.8%). There were no notable differences between treatment groups in the reporting of TEAEs possibly related to the study drug. SAEs in this trial were related to patients’ advanced diabetes disease state, as indicated by long diabetes duration and high pre-existing comorbidities at baseline and likely poor glycemic control over time. AEs leading to discontinuation were well-balanced between treatments.
In addition to AEs in general, this study assessed 2 AEs of special interest: hypoglycemia and weight gain. Incidences of documented symptomatic hypoglycemia, nocturnal hypoglycemia (≤ 70 mg/dL), and nocturnal hypoglycemia (< 50 mg/dL) were higher in the twice-daily group than in thrice-daily group. However, the incidence of severe hypoglycemia did not differ significantly between the 2 arms, which was further corroborated by a Bayesian analysis estimating relative risk probabilities for twice daily versus thrice daily. Severe hypoglycemia rates were too low to be analyzed using negative binomial or recurrent-event analyses; for these comparisons, the stratified Wilcoxon rank sum test was used, which showed no significant difference between the thrice-daily and twice-daily arms. Additional post-randomization 3 a.m. SMPGs and the 7-point SMPG profiles at week 12 were not recorded, and all hemoglobin A1C values were greater than 7%.
One death was reported in the study. An unplanned Data Monitoring Committee review for safety assessment was convened in February 2014 following the study death; however, no protocol changes were recommended. Increase in body weight from randomization was similar for the thrice-daily and twice-daily groups. Weight gain per 1% reduction in hemoglobin A1C levels was 0.47 kg (thrice daily) and 1.31 kg (twice daily) by simple linear regression analysis.9 The use of U-500R was accompanied by weight gain. This patient population was likely at high risk for weight gain given the relatively poor glycemic control at baseline in conjunction with the good treatment response.10-12 Prior studies of U-500R therapy have reported mean weight changes of −0.3 kg to 6.8 kg.13-15 Concomitant use of metformin (approximately 50% in the current study) may attenuate weight gain in patients with T2DM.12,16 The clinical expert and clinician group consulted by CADTH considered that the reported rates of AEs and other safety concerns, including death, hypoglycemia, and weight gain, are expected in diabetes patients seen in clinical practice in Canada.
Other Considerations
U-500R would replace the existing insulin therapies for appropriate patients since it possesses both basal and bolus properties. It would allow patients to use fewer injections of insulin and simplify the regimen to replace 2 different kinds of insulin (basal and bolus) with a single type. It can be used in combination with other OADs, as is currently occurring with other insulins. Practitioners consulted by CADTH have generally found better results in their patients on U-500R, improved convenience with the medication, and lower overall cost to the patient (compared to a program with insulin analogues).
Cost
At a submitted price of $94.79 per package of two 3-mL pre-filled pens, the cost per 100 units of U-500R insulin is $3.16. This cost per 100 units is less than that of prefilled pens and cartridges of most other available human insulin or insulin analogue products, but more than most of the less frequently used 10-mL vials. Assuming a mean dose of 339.1 units per day, the cost of U-500R insulin is $3,911 per patient per year.
The available clinical trial showed an increase in the number of units of insulin used per day compared to baseline when patients who were previously using U-100 insulin were switched to U-500R (287.5 daily units versus 339.1 daily units). In analyses accounting for this increase, U-500R insulin remained less expensive than most combinations of U-100 prandial and basal insulin analogue products (range: $4,277 to $6,026 per patient per year). However, it was more expensive than the combination of Admelog-brand insulin lispro and Basaglar-brand insulin glargine ($3,771 per patient per year), as well as combinations of human insulin ($2,577 to $3,415 per patient per year). The submitted price of U-500R insulin would need to be reduced by 3.6% for its annual cost to be equivalent to that of the least expensive combination of insulin analogues, and 14% or 34% to be equivalent to the least expensive combination of human insulins in cartridges or vials, respectively. For public plans that reimburse insulin needles, a small additional saving may be realized due to the decreased number of injections required per day with the use of U-500R insulin.
These estimated costs or savings are uncertain due to a lack of comparative evidence between U-500R insulin and other concentrations of insulin. Additionally, these incremental costs or savings are based on publicly available list prices and may not reflect actual prices paid by Canadian public drug plans.
Conclusions
Based on the summary of clinical evidence submitted by the sponsor, for patients with T2DM who require greater than 200 units of insulin per day, U-500R reduced hemoglobin A1C levels within a 24-week period in a similar magnitude using a twice-daily or thrice-daily regimen. According to the clinical expert consulted on this review, U-500R fulfills a need for diabetes patients with severe insulin resistance and may replace basal-bolus insulin regimens in this patient population. Further, the clinical expert indicated that the findings of the clinical efficacy and AEs reported in the included trial were aligned with what would be expected in Canadian clinical practice. Issues with the submitted study that were identified include insufficiencies in terms of predefined noninferiority margin and methods used for the analysis of noninferiority, potential bias in the subjective assessment of some outcomes with the open-label design, a lack of clarity on handling of missing information, and large proportion of discontinued participants and a lack of clarity on outcome measures for these discontinued participants. Although superiority of U-500R to standard of care using U-100 insulin for diabetes patients requiring high-dose insulin has been discussed in the literature, no evidence from RCTs that make direct comparisons between U-500R and regular U-100 insulin in terms of efficacy and safety is available. The clinical expert consulted for this review indicated that a comparison with standard of care using a U-100 insulin regimen would be more clinically informative. Whether diabetes patients requiring more than 200 units of insulin per day are willing to accept an increased insulin dose and weight gain by switching to U-500R deserves consideration.
At the submitted price and based on the mean dose at week 24 in the pivotal IBHC trial, the annual cost of U-500R insulin is $3,911 per patient per year. When comparators are assumed to be dosed as at baseline in the pivotal IBHC trial, U-500R insulin is less expensive than combinations of originator brands of basal and prandial insulin analogues, but more expensive than combinations of human insulin or the 2 available subsequent entry insulin analogues. The submitted price of U-500R insulin would need to be reduced by 3.6% for its annual cost to be equivalent to that of the least expensive combination of insulin analogues, and 14% or 34% to be equivalent to the least expensive combination of human insulins in cartridges or vials, respectively. The costs and savings associated with the use of U-500R insulin are uncertain due to a lack of comparative clinical evidence to other insulin products.
References
1.Diabetes Canada Clinical Practice Guidelines Expert Committee. Diabetes Canada 2018 clinical practice guidelines for the prevention and management of Diabetes in Canada. Toronto (ON): Diabetes Canada; 2018 (updated 2020): http://guidelines.diabetes.ca/cpg. Accessed 2021 Mar 6.
2.American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care. 2012;35(Suppl 1):S64-71. PubMed
3.Sacks DB, Arnold M, Bakris GL, et al. Executive summary: guidelines and recommendations for laboratory analysis in the diagnosis and management of diabetes mellitus. Clin Chem. 2011;57(6):793-798. PubMed
4.Canadian Chronic Disease Surveillance System (CCDSS). Public Health infobase data tool 2018 edition. Ottawa (ON): Public Health Agency of Canada; 2019: https://health-infobase.canada.ca/ccdss/data-tool/. Accessed 2021 Mar 6.
5.Nolan CJ, Damm P, Prentki M. Type 2 diabetes across generations: from pathophysiology to prevention and management. Lancet. 2011;378(9786):169-181. PubMed
6.DiMeglio LA, Evans-Molina C, Oram RA. Type 1 diabetes. Lancet. 2018;391(10138):2449-2462. PubMed
7.EntuzityTM KwikPen: 500 units/mL solution for injection [product monograph]. Toronto (ON): Eli Lilly Canada; 2020 Aug 17.
8.Rojas-Fernandez C, Verbrugghe S, de Leseleuc L, Grobelna A. Concentrated U-500 insulin regular for injection: pharmacology and prospective utilization. Ottawa: CADTH; 2019: https://cadth.ca/sites/default/files/pdf/he0009-entuzity-U-500-technology-review.pdf. Accessed 2021 Apr 3.
9.Hood RC, Arakaki RF, Wysham C, Li YG, Settles JA, Jackson JA. Two treatment approaches for human regular U-500 insulin in patients with type 2 diabetes not achieving adequate glycemic control on high-dose U-100 insulin therapy with or without oral agents: a randomized, titration-to-target clinical trial. Endocr Pract. 2015;21(7):782-793. PubMed
10.Garber AJ, Abrahamson MJ, Barzilay JI, et al. American Association of Clinical Endocrinologists' comprehensive diabetes management algorithm 2013 consensus statement--executive summary. Endocr Pract. 2013;19(3):536-557. PubMed
11.Wallia A, Molitch ME. Insulin therapy for type 2 diabetes mellitus. JAMA. 2014;311(22):2315-2325. PubMed
12.Yki-Järvinen H. Combination therapies with insulin in type 2 diabetes. Diabetes Care. 2001;24(4):758-767. PubMed
13.Dailey AM, Williams S, Taneja D, Tannock LR. Clinical efficacy and patient satisfaction with U-500 insulin use. Diabetes Res Clin Pract. 2010;88(3):259-264. PubMed
14.Distiller LA, Nortje H, Wellmann H, Amod A, Lombard L. A 24-week, prospective, randomized, open-label, treat-to-target pilot study of obese type 2 diabetes patients with severe insulin resistance to assess the addition of exenatide on the efficacy of u-500 regular insulin plus metformin. Endocr Pract. 2014;20(11):1143-1150. PubMed
15.Quinn SL, Lansang MC, Mina D. Safety and effectiveness of U-500 insulin therapy in patients with insulin-resistant type 2 diabetes mellitus. Pharmacotherapy. 2011;31(7):695-702. PubMed
16.Standards of medical care in diabetes--2014. Diabetes Care. 2014;37(Suppl 1):S14-80. PubMed
17.Althunian TA, de Boer A, Groenwold RHH, Klungel OH. Defining the noninferiority margin and analysing noninferiority: an overview. Br J Clin Pharmacol. 2017;83(8):1636-1642. PubMed
18.Fonseca V, Gill J, Zhou R, Leahy J. An analysis of early insulin glargine added to metformin with or without sulfonylurea: impact on glycaemic control and hypoglycaemia. Diabetes Obes Metab. 2011;13(9):814-822. PubMed
19.Wilding JP, Norwood P, T'Joen C, Bastien A, List JF, Fiedorek FT. A study of dapagliflozin in patients with type 2 diabetes receiving high doses of insulin plus insulin sensitizers: applicability of a novel insulin-independent treatment. Diabetes Care. 2009;32(9):1656-1662. PubMed
20.Balena R, Hensley IE, Miller S, Barnett AH. Combination therapy with GLP-1 receptor agonists and basal insulin: a systematic review of the literature. Diabetes Obes Metab. 2013;15(6):485-502. PubMed
21.QuickFacts table. Suitland (MD): United States Census Bureau; 2019: https://www.census.gov/quickfacts/fact/table/US#. Accessed 2021 Mar 6.
22.Ontario [province] and Canada [country]: census profile, 2016 census. (Catalogue no. 98-316-X2016001). Ottawa (ON): Statistics Canada; 2017: https://www12.statcan.gc.ca/census-recensement/2016/dp-pd/prof/details/Page.cfm?Lang=E&Geo1=PR&Code1=35&Geo2=PR&Code2=01&SearchText=Ontario&SearchType=Begins&SearchPR=01&B1=All&GeoLevel=PR&GeoCode=35&type=0. Accessed 2021 Mar 6.
23.Zabaneh D, Chambers JC, Elliott P, Scott J, Balding DJ, Kooner JS. Heritability and genetic correlations of insulin resistance and component phenotypes in Asian Indian families using a multivariate analysis. Diabetologia. 2009;52(12):2585-2589. PubMed
24.McIntyre HD, Catalano P, Zhang C, Desoye G, Mathiesen ER, Damm P. Gestational diabetes mellitus. Nat Rev Dis Primers. 2019;5(1):47. PubMed
25.Diabetes in Canada. Toronto (ON): Diabetes Canada; 2019: https://www.diabetes.ca/DiabetesCanadaWebsite/media/About-Diabetes/Diabetes%20Charter/2019-Backgrounder-Canada.pdf. Accessed 2021 Mar 6.
26.Imran SA, Agarwal G, Bajaj HS, Ross S. Targets for Glycemic Control. Can J Diabetes. 2018;42(Suppl 1):S42-s46. PubMed
27.Drug Reimbursement Review sponsor submission: Entuzity KwikPen (insulin injection, human biosynthetic) 500 units/mL solution for injection [internal sponsor's package]. Toronto (ON): Eli LIlly Canada; 2021 Jan 27.
28.DeltaPA. [Ottawa (ON)]: IQVIA; 2021: https://www.iqvia.com/. Accessed 2021 Apr 10.
29.Ontario Ministry of Health, Ontario Ministry of Long-Term Care. Ontario drug benefit formulary/comparative drug index. 2021; https://www.formulary.health.gov.on.ca/formulary/. Accessed 2021 Apr 13.
30.Saskatchewan Drug Plan: search formulary. 2021; https://formulary.drugplan.ehealthsask.ca/SearchFormulary. Accessed 2021 Apr 18.
31.Interprofessional Diabetes Education and Advocacy (IDEA) Group. Diabetes resource manual. Toronto (ON): Diabetes Canada; 2018: https://diabetes.ca/diabetescanadawebsite/media/managing-my-diabetes/tools%20and%20resources/diabetes-resource-manual.pdf. Accessed 2021 Apr 13.
32.de la Peña A, Riddle M, Morrow LA, et al. Pharmacokinetics and pharmacodynamics of high-dose human regular U-500 insulin versus human regular U-100 insulin in healthy obese subjects. Diabetes Care. 2011;34(12):2496-2501. PubMed
33.Budget Impact Analysis [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Entuzity KwikPen (insulin injection, human biosynthetic) 500 units/mL solution for injection. Toronto (ON): Eli LIlly Canada; 2021 Jan 27.
34.Table: 17-10-0005-01. Population estimates on July 1st, by age and sex. Ottawa (ON): Statistics Canada; 2020: https://www150.statcan.gc.ca/t1/tbl1/en/tv.action?pid=1710000501. Accessed 2021 Feb 22.
35.Diabetes in Canada: highlights from the Canadian Chronic Disease Surveillance System. Ottawa (ON): Public Health Agency of Canada; 2017: https://www.canada.ca/content/dam/phac-aspc/documents/services/publications/diseases-conditions/diabetes-canada-highlights-chronic-disease-surveillance-system/diabetes-in-canada-eng.pdf. Accessed 2021 Apr 3.
36.Diabetes Canada. Assess your risk of developing diabetes: risk factors for type 2 diabetes. 2021: https://diabetes.ca/type-2-risks/risk-factors---assessments. Accessed 2021 Apr 15.
37.Food security and diabetes: position statement. Toronto (ON): Diabetes Canada: https://www.diabetes.ca/advocacy---policies/our-policy-positions/food-security-and-diabetes. Accessed 2021 Apr 15.
Appendix 1: Additional Economic Information
Note that this appendix has not been copy-edited.
Additional Details on the CADTH Reanalyses and Additional Analyses
Table 13: CADTH Cost Comparison Table of Insulin Products
Drug / comparator | Strength | Dosage form | Price per package ($) | Price per 100 units ($) | Difference in costs ($) per 100 U compared to U-500R |
|---|---|---|---|---|---|
Treatment class | |||||
Biosynthetic Human Insulin (Entuzity) | 500 U/mL | 2 × 3 mL pre-filled pens | 94.7900 | 3.1597 | Reference |
Basal human insulins | |||||
Insulin isophane (Humulin N) | 100 U/mL | 5 × 3 mL pre-filled pens | 48.8100 | 3.2540 | 0.0943 |
10 mL vial | 24.8800 | 2.4880 | −0.6717 | ||
Novolin ge NPH | 100 U/mL | 5 × 3 mL pre-filled pens | 48.8200 | 3.2547 | 0.0950 |
10 mL vial | 24.8300 | 2.4830 | −0.6767 | ||
Long-acting insulin analogues | |||||
Insulin degludec (Tresiba) | 100 U/mL | 5 × 3 mL pre-filled pens | 111.5000 | 7.4333 | 4.2736 |
200 U/mL | 3 × 3 mL pre-filled pens | 133.8000 | 7.4333 | 4.2736 | |
Insulin glargine (Basaglar) | 100 U/mL | 5 × 3 mL pre-filled pens or cartridges | 69.6375 | 4.6425 | 1.4828 |
Insulin glargine (Lantus) | 100 U/mL | 5 × 3 mL pre-filled pens or cartridges | 92.8500 | 6.1900 | 3.0303 |
10 mL vial | 61.6900 | 6.1680 | 3.0083 | ||
Insulin glargine (Toujeo) | 300 U/mL | 1.5 mL pre-filled pens 3 mL pre-filled pens | 26.4300 52.8666 | 5.8733 5.8733 | 2.7136 2.7136 |
Insulin detemir (Levemir) | 100 U/mL | 5 × 3 mL pre-filled pens 5 × 3 mL pre-filled cartridges | 111.5000 110.4100 | 7.4333 7.3607 | 4.2736 4.2010 |
Prandial human insulins | |||||
Regular human insulin (Humulin R) | 100 U/mL | 5 × 3 mL pre-filled cartridges | 48.8100 | 3.2540 | 0.0943 |
10 mL vial | 24.8800 | 2.4888 | −0.6717 | ||
Regular human insulin (Novolin ge Toronto) | 100 U/mL | 5 × 3 mL pre-filled cartridges | 47.6800 | 3.1787 | 0.0190 |
10 mL vial | 24.2800 | 2.4280 | −0.7317 | ||
Rapid-acting insulin analogues | |||||
Insulin aspart (NovoRapid) | 100 U/mL | 5 × 3 mL pre-filled pens 5 × 3 mL pre-filled cartridges | 63.7500 61.2300 | 4.2500 4.0820 | 1.0903 0.9223 |
10 mL vial | 30.1900 | 3.0190 | −0.1407 | ||
Insulin glulisine (Apidra) | 100 U/mL | 5 × 3 mL pre-filled pens 5 × 3 mL pre-filled cartridges | 53.1500 52.6500 | 3.5433 3.5100 | 0.3836 0.3503 |
10 mL vial | 25.5800 | 2.5580 | −0.6017 | ||
Insulin lispro (Humalog) | 100 U/mL | 5 × 3 mL pre-filled pens 5 × 3 mL pre-filled cartridges | 59.6300 60.0600 | 3.9753 4.0040 | 0.8156 0.8443 |
10 mL vial | 30.2300 | 3.0230 | −0.1367 | ||
Insulin lispro (Humalog) | 200 U/mL | 5 × 3 mL pre-filled pens | 111.0000 | 3.7000 | 0.5403 |
Insulin lispro (Admelog) | 100 U/mL | 5 × 3 mL pre-filled pens or cartridges | 45.0000 | 3.0000 | −0.1597 |
10 mL vial | 22.7000 | 2.2700 | −0.8897 | ||
Prices are Ontario Drug Benefit Formulary list prices (accessed March 2021) unless otherwise indicated. Note: Reanalyses are based on publicly available prices of the comparator treatments.
Appendix 2: Submitted BIA and CADTH Appraisal
Table 14: Key Take-Aways of the Budget Impact Analysis
Key Take-Aways of the BIA |
|---|
|
Summary of Sponsor’s BIA
In the submitted budget impact analysis (BIA),33 the sponsor assessed the expected budget impact of reimbursing U-500R insulin for patients with diabetes mellitus requiring more than 200 units of insulin per day. The BIA was from the perspective of a public drug plan, over a 3-year time horizon, and included only drug acquisition costs; markups and dispensing fees were not included for the pan-Canadian analysis. Comparators included basal and prandial human and analogue insulins, with each of the basal and prandial insulin categories assumed to make up 50% of units used each day. Patients were assumed to require 339.1 units of insulin per day, consistent with the mean total daily dose reported at Week 24 of the IBHC trial.
Data for the model were obtained from various sources including: the IQVIA Delta PA database,28 IQVIA RxDynamics, Statistics Canada,34 the Public Health Agency of Canada (PHAC),35 a previous CADTH technology review on U-500R insulin,8 the IBHC trial,9 the ODB formulary,29 and the sponsor’s internal market research.
Key inputs to the BIA are documented in Table 15. Other key assumptions include:
U-500R insulin is assumed to replace other insulins on a 1:1 unit basis
Table 15: Summary of Key Model Parameters
Parameter | Reference Scenario | New Drug Scenario | ||
|---|---|---|---|---|
Target Population: Patients with diabetes mellitus requiring more than 200 units of insulin per day. | ||||
Number of plan beneficiaries | 17,947,900a | |||
Annual population growth rate | 1.53%b | |||
Prevalence of diabetes | 8.10%c | |||
Annual incidence of diabetes | 0.59%c | |||
Patients treated with insulin | 12.43%c | |||
Insulin-treated patients administered > 200 units/day | 2.8%d | |||
Mean number of insulin units per patient per day | 339.1 unitse | |||
Proportion of units (basal/prandial) | 50% / 50% | |||
Number of patients eligible for drug under review | 5,434 / 5,808 / 6,182 | |||
Market Uptake (3 years) | ||||
Basal insulinsf | ||||
Human biosynthetic insulin (Entuzity) | 0% / 0% / 0% | 8.00% / 15.00% / 20.00% | ||
Human biosynthetic insulin (Humulin R)g | 0.61% / 0.37% / 0.18% | 0.56% / 0.31% / 0.14% | ||
Human biosynthetic insulin (Novolin ge Toronto)g | 0% / 0% / 0% | 0% / 0% / 0% | ||
Insulin glargine (Lantus) | 46.29% / 41.79% / 38.16% | 42.59% / 35.53% / 30.53% | ||
Insulin glargine (Basaglar) | 3.33% / 3.65% / 3.91% | 3.06% / 3.10% / 3.13% | ||
Insulin glargine (Toujeo) | 0% / 0% / 0% | 0% / 0% / 0% | ||
Insulin detemir (Levemir) | 4.83% / 3.73% / 2.85% | 4.44% / 3.17% / 2.28% | ||
Insulin degludec (Tresiba) | 44.95% / 50.46% / 54.90% | 41.35% / 42.89% / 43.92% | ||
Prandial insulinsf | ||||
Human biosynthetic insulin (Entuzity) | 0% / 0% / 0% | 8.00% / 15.00% / 20.00% | ||
Insulin isophane (Humulin N)b | 0.26% / 0% / 0% | 0.24% / 0% / 0% | ||
Insulin isophane (Novolin NPH)b | 5.37% / 4.57% / 3.86% | 4.94% / 3.88% / 3.09% | ||
Insulin aspart (NovoRapid) | 44.52% / 45.08% / 45.48% | 40.96% / 38.32% / 36.38% | ||
Insulin glulisine (Apidra) | 5.55% / 5.53% / 5.48% | 5.11% / 4.70% / 4.39% | ||
Insulin lispro (Humalog) | 44.30% / 44.82% / 45.18% | 40.75% / 38.09% / 36.14% | ||
Cost of treatment (per unit of insulin, weighted average of different forms where applicable) | ||||
Basal insulins | ||||
Human biosynthetic insulin (Entuzity) | $0.0316 | |||
Human biosynthetic insulin (Humulin R)g | $0.0307 | |||
Human biosynthetic insulin (Novolin ge Toronto)g | $0.0301 | |||
Insulin glargine (Lantus) | $0.0619 | |||
Insulin glargine (Basaglar) | $0.0464 | |||
Insulin glargine (Toujeo) | $0.0587 | |||
Insulin detemir (Levemir) | $0.0739 | |||
Insulin degludec (Tresiba) | $0.0743 | |||
Prandial insulins | ||||
Human biosynthetic insulin (Entuzity) | $0.0316 | |||
Insulin isophane (Humulin N)g | $0.0312 | |||
Insulin isophane (Novolin NPH)g | $0.0320 | |||
Insulin aspart (NovoRapid) | $0.0405 | |||
Insulin glulisine (Apidra) | $0.0351 | |||
Insulin lispro (Humalog) | $0.0386 | |||
aCited as IQVIA RxDynamics data.
bStatistics Canada.34
cPublic Health Agency of Canada 2017.35
dCADTH 2019.8
eIBHC trial.9
fWhile market shares within each category total 100%, prandial and basal insulins are each assumed to make up 50% of daily insulin units administered, with U-500R insulin replacing an equal number of units of each. Additionally, market shares reported here are based on Alberta inputs rather than a weighted average of included jurisdictions due to an error in the sponsor’s model.
gThis is an error in the sponsor’s submission; Humulin R and Novolin ge Toronto are short-acting insulins and thus should be considered prandial, while Humulin N and Novolin NPH are intermediate-acting insulins and thus should be considered basal.
Summary of the Sponsor’s BIA Results
Results of the sponsor’s base case suggest that the incremental budget impact, excluding markups and dispensing fees, associated with the reimbursement of U-500R insulin for patients with diabetes mellitus requiring more than 200 units per day would be a savings of $1,158,273 in Year 1, $2,356,921 in Year 2, and $3,384,928 in Year 3, for a 3-year cumulative budgetary savings of $6,900,122.
CADTH Appraisal of the Sponsor’s BIA
CADTH identified the following key limitations to the sponsor’s analysis that have notable implications on the results of the BIA:
Errors in the submission: The sponsor’s model inappropriately categorized human insulin isophane as prandial, while regular human biosynthetic insulin was categorized as basal. Additionally, although the model appeared to be programmed correctly, the market share sheet of the model inappropriately used Alberta market shares for each insulin product to populate the pan-Canadian (excluding Quebec) analysis, rather than the weighted average market share of included jurisdictions as intended by the sponsor.
CADTH corrected these errors by reassigning the human insulins to the correct categories and manually correcting the market share inputs for each product to the weighted average of all included jurisdictional market shares, overriding an unknown program flaw.
Underestimated daily usage of U-500R insulin: As described in the cost comparison above, patients in the IBHC trial9 used a daily mean of 287.5 units of U-100 insulin at baseline and 339.1 units of U-500R insulin at Week 24. This data does not support the sponsor’s assumption that U-500R insulin will replace U-100 insulin on a 1:1 basis. The sponsor’s model was not flexible enough to allow for differential unit assumptions between comparators, however by inflating the cost of U-500R insulin by 17.9% (339.1/287.5), the budgetary impact of assuming increased units can be simulated.
CADTH increased the unit cost of U-500R insulin by 17.9% to simulate an increase in the number of daily units used by patients receiving U-500R insulin.
Underestimated prevalence of diabetes in beneficiary population: The sponsor’s model estimates the number of patients using over 200 units of insulin per day by applying the prevalence of diabetes in the general Canadian population to the population of beneficiaries of Canadian public drug plans. However, beneficiaries of public drug plans tend to be those who are over 65 years of age, those with high annual medical expenditures, those of limited financial resources, and, in the case of NIHB, those who are indigenous. All of these populations have a higher prevalence of diabetes than the overall Canadian population,36,37 and thus the sponsor’s estimate of the number of people with diabetes, and who use insulin, and who use over 200 units of insulin per day is underestimated.
CADTH reanalyses used the number of insulin beneficiaries reported in 2017 in a previous CADTH report on U-500R insulin,8 applied the sponsor’s annual growth rate of 1.53% to estimate 2021 beneficiaries, and then assumed 2.8% of insulin beneficiaries use over 200 units per day.
Newly introduced comparator: Insulin lispro U-100 (Admelog) recently became available in Canada and is currently listed on the ODB Formulary at a price of $22.70 per 10 mL vial or $45.00 per package of five 3 mL prefilled pens or cartridges. Admelog is significantly less expensive than the other available insulin lispro U-100 product (Humalog); as patients who would otherwise have used Humalog begin using Admelog, the modelled budgetary impact of U-500R insulin will change depending on the uptake of the Admelog brand. There are insufficient claims data currently available to predict how the market share of each available brand of insulin lispro will change, however Basaglar-brand insulin glargine had approximately half as much market share as the originator brand Lantus within the BIA.
In the absence of claims data, and to explore the potential impact of a less expensive insulin lispro on the market, CADTH conducted a scenario analysis where 67% of insulin lispro within the BIA was priced at the current list price of Humalog and the other 33% at that of Admelog.
Insulin injection supplies excluded: While BIA submissions are intended to be from the perspective of a public drug plan and thus typically do not include non-drug resource costs, insulin needles are sometimes funded by provincial formularies and thus changes in their relative use between comparators could be considered to directly impact public drug plan budgets.
The model was not flexible enough to include the cost of insulin needles for jurisdictions which reimburse them through their drug plan formularies. Jurisdictions which do reimburse needles for injecting insulin could expect to see a small amount of additional savings as a result of reimbursing U-500R insulin for patients for whom it would reduce the number of insulin needles required per day, likely by at least 2 needles per patient per day. The extent of such savings would depend on each jurisdictions’ cost per needle, see CADTH reanalysis section of the cost comparison in the main body of this report for estimates.
CADTH Reanalyses of the BIA
CADTH revised the sponsor’s base case by: correcting the errors in the model, increasing the number of public drug plan beneficiaries who use insulin, and increasing the cost of U-500R insulin to simulate an increase in daily units of insulin when switching from other insulins to U-500R insulin. Table 16 outlines the parameters used by the sponsor in comparison to those used by CADTH. Corrected market shares for each of the comparators described in Table 16 can be found in Table 17.
Table 16: CADTH Revisions to the Submitted Budget Impact Analysis
Stepped analysis | Sponsor’s value or assumption | CADTH value or assumption |
|---|---|---|
Correctionsa to sponsor’s base case | ||
1. Human biosynthetic insulins appropriately categorized | Humulin R and Novolin ge Toronto are basal Humulin N and Novolin NPH are prandial | Humulin R and Novolin ge Toronto are prandial Humulin N and Novolin NPH are basal |
2. Comparator market share | Pan-Canadian model uses Alberta market shares for each insulin product | Pan-Canadian model uses weighted average market shares across all included jurisdictions for each insulin product |
Changes to derive the CADTH base case | ||
1. Number of plan beneficiaries with diabetes using > 200 units insulin per day (reference year 2021) | 17,947,900 plan beneficiaries × 8.1% DM prevalence × 12.43% insulin treated × 2.8% over 200 units = 5,060 eligible patients in 2021 | 313,972 insulin beneficiaries in 2017 × 2.8% using 201 to 600 units daily = 8,644 eligible patients in 2017 Inflated with 1.53% annual population growth rate = 9,185 eligible patients in 2021 |
2. U-500R insulin does not replace U-100 on a 1:1 unit basis | U-500R insulin was assumed to replace U-100 insulin on a 1:1 unit basis | The daily cost of U-500R insulin was inflated by 17.3% (339/289) to reflect the increase in daily units seen in the IBHC trialb |
CADTH base case | — | 1 + 2 |
aCorrections are minor errors that are not identified as limitations. In this case, the sponsor’s analysis incorrectly categorized the human insulins and incorrectly used Alberta market shares for the pan-Canadian (excluding Quebec) analysis rather than a weighted average of all included jurisdictions.
bThe sponsor’s model did not allow for alteration to the relative number of units used per day between comparators. Inflating the per-unit cost of U-500R insulin proportional to the increased total daily dose seen in the trial allowed for the impact of this assumption to be tested indirectly.
Table 17: CADTH Corrections to Sponsor’s Insulin Market Shares
Parameter | Reference Scenario (reported as year 1 / year 2 / year 3) | New Drug Scenario (reported as year 1 / year 2 / year 3) |
|---|---|---|
Market uptake (3 years) | ||
Basal insulinsa | ||
Human biosynthetic insulin (Entuzity) | 0% / 0% / 0% | 8% / 15% / 20% |
Human biosynthetic insulin (Humulin N) | 3.12% / 2.53% / 2.10% | 2.87% / 2.15% / 1.68% |
Human biosynthetic insulin (Novolin ge NPH) | 2.22% / 1.70% / 1.33% | 2.04% / 1.44% / 1.06% |
Insulin glargine (Lantus) | 37.25% / 34.13% / 31.62% | 34.27% / 29.01 / 25.29% |
Insulin glargine (Basaglar) | 14.96% / 16.44% / 17.61% | 13.76% / 13.97% / 14.09% |
Insulin glargine (Toujeo) | 7.42% / 7.97% / 8.40% | 6.83% / 6.78% / 6.72% |
Insulin detemir (Levemir) | 4.71% / 3.96% / 3.35% | 4.34% / 3.37% / 2.68% |
Insulin degludec (Tresiba) | 30.32% / 33.27% / 35.59 | 27.90% / 28.28% / 28.47% |
Prandial insulinsa | ||
Human biosynthetic insulin (Entuzity) | 0% / 0% / 0% | 8% / 15% / 20% |
Insulin isophane (Humulin R) | 2.83% / 2.41% / 2/11% | 2.60% / 2.05% / 1.68% |
Insulin isophane (Novolin ge Toronto) | 0.67% / 0.43% / 0.21% | 0.62% / 0.37% / 0.17% |
Insulin aspart (NovoRapid) | 42.85% / 42.92% / 42.94% | 39.43% / 36.48% / 34.35% |
Insulin glulisine (Apidra) | 8.75% / 8.89% / 9.00% | 8.05% / 7.56% / 7.20% |
Insulin lispro (Humalog) | 44.89% / 45.35% / 45.74% | 41.30% / 38.55% / 36.39% |
aWhile market shares within each category total 100%, prandial and basal insulins are each assumed to make up 50% of daily insulin units administered, with Entuzity replacing an equal number of units of each.
Applying these changes led to an estimated budgetary savings of $1,239,397 in Year 1, $2,464,406 in Year 2, and $3,451,832 in Year 3, for a 3-year total budgetary savings of $7,155,636. The results of the CADTH stepped reanalysis are presented in summary format in Table 18. Additionally, CADTH included a scenario analysis around the CADTH base case assuming the dispensing fees and markups allowed by ODB, as well as a scenario where 33% of insulin lispro was assumed to be priced at the ODB list price of Admelog.
Table 18: Summary of the CADTH Reanalyses of the Budget Impact Analysis
Stepped analysis | Three-year total |
|---|---|
Submitted base case | –$6,900,122 |
Corrected submitted base case | –$6,000,221 |
CADTH reanalysis 1: Increased Population of Beneficiaries with Diabetes | –$10,185,288 |
CADTH reanalysis 2: U-500R insulin cost increased to reflect increased units used | –$4,215,723 |
CADTH base case | –$7,155,636 |
CADTH Scenario A: Ontario mark-up and dispensing fees assumed for Canada | –$7,728,087 |
CADTH Scenario B: 33% of insulin lispro U-100 (Humalog) assumed be reimbursed at insulin lispro U-100 (Admelog) list price | –$6,784,152 |
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up-to-date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.