CADTH Reimbursement Review
Risperidone for Extended-Release Injectable Suspension (Perseris)
Sponsor: HLS Therapeutics Inc.
Therapeutic area: Schizophrenia, adults
Clinical and Pharmacoeconomic Review
Abbreviations
AAP
atypical antipsychotic
AE
adverse event
BIA
budget impact analysis
CGI-I
Clinical Global Impression–Improvement
CGI-S
Clinical Global Impression–Severity of Illness
CI
confidence interval
ER
extended release
EQ-5D-5L
EuroQol 5-Dimensions 5-Levels questionnaire
HRQoL
health-related quality of life
IAM
Institute for Advancements in Mental Health
IM
intramuscular
ITT
intention to treat
LAI
long-acting injectable
LSM
least squares mean
LSMD
least squares mean difference
MAR
missing at random
MMRM
mixed-effects model for repeated measures
MSQ
Medication Satisfaction Questionnaire
PANSS
Positive and Negative Syndrome Scale
POM
Preference of Medicine questionnaire
SAE
serious adverse event
SC
subcutaneous
SD
standard deviation
SE
standard error
SWN-S
Subjective Well-Being Under Neuroleptic Treatment — Short Form
TEAE
treatment-emergent adverse event
Executive Summary
An overview of the submission details for the drug under review is provided in Table 1.
Item | Description |
|---|---|
Drug product | Risperidone for extended-release injectable suspension (Perseris), powder for suspension, 90 mg or 120 mg subcutaneous injection |
Indication | Treatment of schizophrenia in adults |
Reimbursement request | As per indication |
Health Canada approval status | NOC |
Health Canada review pathway | Standard |
NOC date | November 19, 2020 |
Sponsor | HLS Therapeutics Inc. |
NOC = Notice of Compliance.
Introduction
Schizophrenia is a severe and chronic psychiatric disease and represents a heterogeneous group of disorders that may differentially affect presentation, course, treatment response, and outcome.1-3 The onset of schizophrenia usually occurs during middle to late adolescence or early adulthood and is diagnosed by specific signs and symptoms that prevent reality-based judgment.4,5 Schizophrenia is associated with hallucinations, delusions, cognitive impairment, disorganized thoughts, social withdrawal, and amotivation.6 Patients with schizophrenia are at an increased risk for numerous other medical illnesses, suicide, substance abuse, homelessness, and unemployment.7,8 In 2016, the prevalence of schizophrenia in Canada was estimated to be approximately 0.95% of the population: 1.08% in males and 0.82% in females.9 The incidence of schizophrenia in Canada was estimated to be approximately 49 per 100,000 in 2016, with 58 cases per 100,000 in males and 41 cases per 100,000 in females.9 Despite its low prevalence, its health, social, and economic burden is tremendous, not only for patients, but also for families, caregivers, and the wider society.10
Antipsychotic medications that target the characteristic symptoms of schizophrenia form the cornerstone of treatment.6,11,12 The choice of antipsychotic medication should be made by the patient and physician together.2 Following resolution of positive symptoms of an acute episode of schizophrenia, patients should be offered maintenance treatment and antipsychotic medication for up to 5 years or longer.2
Oral medications, including a low-to-moderate dosage of antipsychotic monotherapy represent first-line treatment.13,14 One major obstacle to the effective treatment of schizophrenia is nonadherence to medication, resulting in cycles of relapse.15,16 The primary indication for long-acting injectable (LAI) antipsychotic drugs is for patients with schizophrenia who have a history of good response to an oral antipsychotic but who adhered poorly to the medication, leading to relapse.17
In Canada, existing long-term atypical antipsychotic (AAP) medications may be given as an intramuscular (IM) injection every 2 weeks to every 3 months, depending on the medication.14 The existing AAP LAIs available in Canada include risperidone LAI (IM every 2 weeks),18 aripiprazole LAI (IM monthly),19 paliperidone LAI (IM monthly),20 and paliperidone LAI (IM every 3 months).21
Risperidone for extended release (ER) injectable suspension (risperidone ER [Perseris]), powder for suspension, 90 mg or 120 mg subcutaneous (SC) injection, was approved by Health Canada in November 2020.22 It is indicated for the treatment of schizophrenia in adults.22 Like other AAPs, the exact mechanism of risperidone ER is unclear. The Health Canada–recommended dose is 90 mg or 120 mg once monthly by SC injection. It does not require a loading dose. Risperidone ER 90 mg corresponds to 3 mg/day oral risperidone, and risperidone ER 120 mg corresponds to 4 mg/day oral risperidone.
The objective of this report is to review the beneficial and harmful effects of risperidone for ER injectable suspension powder for suspension, 90 mg or 120 mg SC injection, in the treatment of schizophrenia in adults.
Stakeholder Perspectives
The information in this section is a summary of the input provided by the patient groups that responded to CADTH’s call for patient input and from the clinical expert consulted by CADTH for the purpose of this review.
Patient Input
Patient group input was provided by the Institute for Advancements in Mental Health (IAM) and was obtained based on IAM’s 40-year history of serving adults with schizophrenia. Its submission also draws some information from a survey of members of IAM’s client network that was conducted in 2018. Respondents to that survey self-described as: personally diagnosed (12%), caregiver (50%), family member or friend of someone diagnosed (63%), or working in social services (18%).
Respondents indicated that many patients experience symptoms of psychosis, which have a significant impact on day-to-day functioning. Patient experiences vary widely but typically involve some levels of cognitive impairment, delusions, and hallucinations. A large number of patients also experience a lack of insight into their illness, which often impacts their ability and motivation to access treatment and supports. This symptom can cause significant strain in relationships, including those with caregivers and family members, ultimately leading to social isolation and a lack of supports for the individual with the illness.
Patients indicated the most common side effects of antipsychotic drugs were drowsiness (reported by 58%), dry mouth (50%), restlessness (50%), dizziness, muscle stiffness, constipation, and anxiety.
Twenty-three percent of patients identified the cost of medications as a significant barrier to access; 63% of respondents indicated that it is difficult to pay for health care bills, including for medication, visits to specialists, counselling, and so forth; 20% of patients indicated that not having their preferred medication covered by public drug programs is a challenge.
Patients expect new, quick, simple, convenient, and affordable access to a wide range of treatments and medications to suit their unique needs, which can improve adherence and allow for the highest level of daily functioning and symptom reduction while managing side effects.
Clinician Input
Input From the Clinical Expert Consulted by CADTH
The clinical expert indicated that current treatments do not treat the underlying pathophysiology, which is not really understood. Medications have burdensome side effects which, in some cases, are life-threatening (diabetes, neuroleptic malignant syndrome) or irreversible (tardive dyskinesia). Antipsychotic drugs treat only 1 of 3 symptom domains—they do not treat negative and cognitive symptoms. Treatments for refractory disorders are few and have severe adverse effects or inconvenience.
The clinical expert consulted by CADTH for this review anticipated that risperidone ER would initially be used as second-line treatment; it is a convenient LAI that does not require concomitant oral medication to initiate. This may allow earlier hospital discharge or easier community initiations. Since it is based on a familiar drug, clinicians could be quick to adopt it.
The clinical expert indicated that adult patients with well-diagnosed schizophrenia or schizoaffective disorder who have responded to oral risperidone would be the ideal candidates for risperidone ER. Geriatric and pediatric patients would not be good candidates, given the lack of data on these groups and the risk of stroke and increased mortality in older adults. Patients with appropriately diagnosed treatment-resistant illness would be unlikely to benefit.
The clinical expert indicated that, most precisely, a 20% reduction in the positive symptoms on a valid psychosis rating scale, e.g., Positive and Negative Syndrome Scale (PANSS) or Brief Psychiatric Rating Scale, is the most reliable way to confirm response to antipsychotic medication. However, such scales are often not used in clinical practice. Therefore, the routine mental status exam, which thoroughly assesses hallucination, delusions, and disorganized thought and behaviour, is appropriate. Clinically meaningful improvement was usually defined as a 20% reduction in the positive symptoms on a valid psychosis rating scale (e.g., PANSS). In addition, it would also involve improved function, as manifested by more appropriate social interactions, greater consistency in activities of daily living, and a reduction in the risk of self-harm or aggression.
The clinical expert indicated that in the acute phase in a community setting, treatment response should be assessed at least twice a week, which may be done through virtual or in-person visits combined with collateral input. If the patient is in the hospital and at risk of aggression or suicide, a daily assessment by a physician or nurse is necessary. Once a patient is in the stabilization phase and in the community, an assessment once every 1 or 2 weeks is adequate. In the maintenance phase, once a month or even every 3 months can be adequate.
The clinical expert indicated that if a patient has been symptom-free and has had good functional recovery for 2 or more years, discontinuation can be considered.
The clinical expert indicated that risperidone ER could be initiated in hospital or in the community, typically in an acute psychiatry unit or a community or tertiary mental health program. Family physicians who are familiar with antipsychotic medication could also initiate the treatment for patients with mild exacerbations who have demonstrated tolerability to risperidone oral medication.
Clinician Group Input
No clinician group input was received for this review.
Drug Program Input
Drug programs identified several key issues related to implementation, as follows:
First, the drug programs requested clarification on whether risperidone ER would be used for a patient who failed either oral therapy or conventional LAIs. The clinical expert consulted by CADTH for this review indicated that risperidone ER should be offered to any patient who might benefit from the drug, not only those who have failed oral therapy due to nonadherence.
Second, the drug programs asked whether there are special concerns with the use of risperidone ER (use of the Atrigel delivery system) in pregnant patients. The clinical expert indicated that risperidone ER has not undergone adequate study in pregnant patients to determine whether it is safe, especially in the first trimester of pregnancy. The benefits may outweigh the risks of risperidone ER for certain patients, and close monitoring would be necessary if it is prescribed to a pregnant patient.
Third, the drug programs wondered whether or not a washout period for patients currently on oral therapies would be required. The clinical expert indicated that patients who undergo a switch of antipsychotic usually have a “cross taper” in which the first medication is gradually reduced and discontinued over several weeks, while the second is gradually increased. Although a washout is not typically necessary for oral medications, cross-tapering with this injection technology has not been studied.
Fourth, the drug programs requested clarification on whether risperidone ER would be used for the prevention of relapse and for maintenance. The clinical expert indicated that risperidone ER was shown in the regulatory trial to be effective for acute exacerbation of schizophrenia. Given experience with other risperidone and paliperidone LAIs, it is justified to assume that risperidone ER would be effective for maintenance therapy, but a long-term study is required to confirm this.
Fifth, the drug programs requested clarification of the definitions of “treatment resistant” or “refractory disease.” The clinical expert indicated that treatment-resistant schizophrenia is diagnosed in patients who do not have at least a 20% reduction in positive symptoms in response to either of 2 medication trials of different antipsychotic drugs at adequate dose and duration. These patients should receive clozapine, whenever possible. A minority of patients also fail to meet the response criteria for clozapine and those patients are considered refractory.
Clinical Evidence
Pivotal Studies
Description of Studies Submitted by the Sponsor
The CADTH clinical review was based on a summary of clinical evidence provided by the sponsor in accordance with the CADTH tailored review process. One phase III, randomized, double-blind, placebo-controlled study (Study 09 to 0010) that was performed at 33 centres in the US was included in the sponsor’s summary of the clinical evidence. The objective of Study 09 to 0010 was to evaluate the efficacy and safety of risperidone ER compared with placebo in patients (N = 354) aged 18 to 55 years with moderate-to-severe schizophrenia in an acute exacerbation phase. The study was conducted on an inpatient basis in a hospital setting. Patients were randomized to 1 of 3 treatment groups: risperidone ER 90 mg SC, risperidone ER 120 mg SC, or placebo injection SC for 8 weeks. The primary outcome was the change from baseline in PANSS total score at end-of-treatment and the secondary outcome was change from baseline to end of treatment on the Clinical Global Impression–Severity of Illness (CGI-S).
Baseline demographic characteristics were generally balanced across treatment arms. The majority of patients included in the study were Black (> 70%) and male (> 73.5%). The mean age ranged from 40.5 to 42.4 years across the 3 groups. The baseline disease characteristics were not summarized in the sponsor’s summary of clinical evidence.
Efficacy Results
The PANSS total score change from baseline at week 8 (primary outcome) demonstrated an improvement in the risperidone ER 90 mg, risperidone 120 mg group, and placebo group (least squares mean [LSM] of −15.37 [standard error [SE] = 1.22], −16.46 [SE = 1.20], and −9.22 [SE = 1.22] in the risperidone ER 90 mg, risperidone ER 120 mg, and placebo groups, respectively). Compared with placebo, both risperidone ER 90 mg and risperidone ER 120 mg demonstrated a statistically significant improvement (risperidone ER minus placebo LSM difference [LSMD] of −6.15 [95% confidence interval [CI], −9.98 to −2.31; P = 0.0004] for risperidone ER 90 mg and −7.24 [95% CI, −11.05 to −3.43; P < 0.0001] for risperidone ER 120 mg) (Table 2). It is uncertain whether the difference between the risperidone ER treatment group and placebo group was clinically meaningful. The CADTH clinical expert consulted for this review indicated that a 20% improvement in the PANSS total score is usually considered a clinical meaningful response to treatment in schizophrenia patients.
In terms of change from baseline in Clinical Global Impression (CGI-S) score (the secondary outcome), both risperidone ER groups (90 mg and 120 mg) and the placebo group demonstrated an improvement at the end of the study (LSM of −0.87 [SE = 0.07], −0.91 [SE = 0.07], and −0.52 [SE = 0.07] in the risperidone ER 90 mg and 120 mg groups and placebo group, respectively). Compared with placebo, both risperidone ER 90 mg and risperidone ER 120 mg also demonstrated a statistically significant improvement (LSMD of −0.35 [95% CI, −0.56 to −0.14; P = 0.0002] for risperidone ER 90 mg minus placebo and −0.40 [95% CI, −0.60 to −0.19; P < 0.0001] for risperidone ER 120 mg minus placebo) (Table 2). For the CGI-S, neither the change from baseline for either risperidone ER treatment group, nor the treatment-group difference between the risperidone ER groups and placebo, met the minimal important difference (i.e., a reduction of 1 point in the CGI-S). Therefore, the clinical significance of the observed findings in CGI-S is unclear.
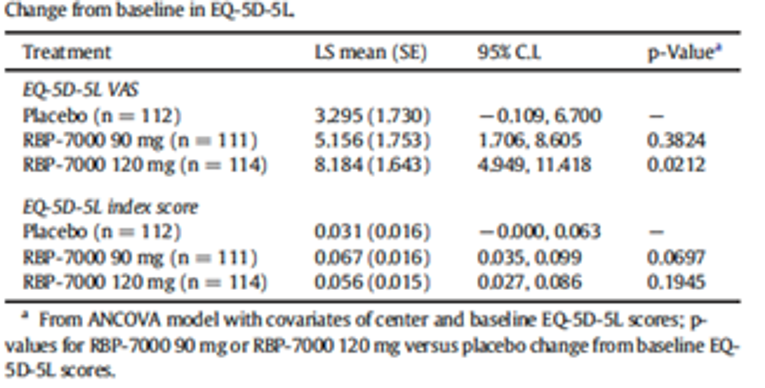
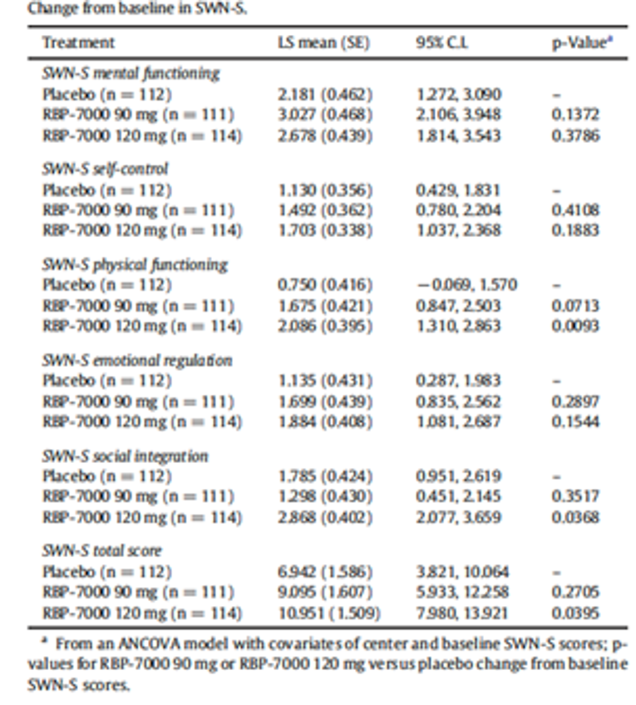
Health-related quality of life (HRQoL) was assessed using the EuroQol 5-Dimensions 5-Levels questionnaire (EQ-5D-5L). Patients’ subjective well-being was estimated with the Subjective Well-Being Under Neuroleptic Treatment–Short Form (SWN-S), and patients’ satisfaction with medication was examined with the Medication Satisfaction Questionnaire (MSQ). However, HRQoL was assessed as an exploratory outcome in Study 09 to 0010; therefore, no conclusions can be made.
Harms Results
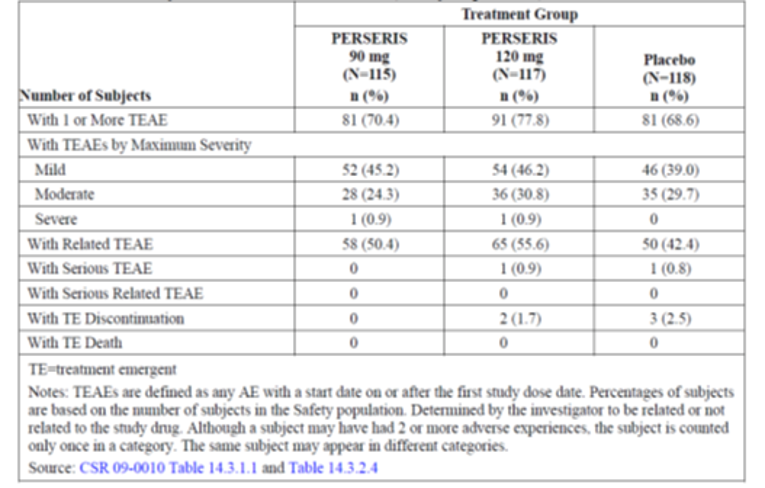
The proportion of the patients who experienced at least 1 treatment-emergent adverse event (TEAE) was reportedly higher for the risperidone ER 120 mg group (77.8%) compared with the risperidone ER 90 mg (70.4%) and placebo groups (68.6%). Overall, the most frequently reported TEAEs that occurred at higher rates in the risperidone ER groups compared with the placebo group were weight gain (13%, 12.8%, and 3.4% in the risperidone ER 90 mg and 120 mg groups and the placebo group, respectively) and somnolence (5.2%, 4.3%, and 0% in the risperidone ER 90 mg and 120 mg groups and placebo group, respectively).
There were no deaths reported during the treatment periods. The incidence of serious TEAEs was infrequent (0%, 0.9%, and 0.8% in the risperidone ER 90 mg and 120 mg groups and the placebo group, respectively). The proportion of patients who withdrew due to adverse events (AEs) was reportedly low (0%, 1.7%, and 2.5% in the risperidone ER 90 mg and 120 mg groups and placebo group, respectively). Regarding the AEs of special interest for this review, more patients (13%) in the risperidone ER groups experienced weight gain compared with patients who received placebo (3.4%), which was an expected AE that has been reported in all other AAP drugs.
Treatment with risperidone ER (90 mg and 120 mg) over an 8-week treatment period was generally well tolerated in adult patients with acute schizophrenia. There were no new safety signals compared with the known safety profile of oral LAI risperidone products.
Table 2: Summary of Key Results From Pivotal Study
Outcomes | Study 09-001023 | ||
|---|---|---|---|
Risperidone ER 90 mg (N = 116a) | Risperidone ER 120 mg (N = 119a) | Placebo (N = 119a) | |
PANSS total score (ITT, MMRM)b | |||
Number of patients contributing to the analysis (%) | 111 (95.7) | 114 (95.8) | 112 (94.1) |
At baseline, mean (SD)c | 95.5 (9.23) | 94.9 (8.09) | 94.1 (8.89) |
At specified end-of-treatment time point, mean (SD)c | 75.8 (16.35) | 73.1 (14.81) | 81.5 (18.35) |
Change from baseline, LSM (SE) | −15.37 (1.22) | −16.46 (1.20) | −9.22 (1.22) |
Treatment group difference vs. placebo control LSMD (95% CI) | −6.15 (−9.98 to –2.31) | −7.24 (−11.05 to –3.43) | NA |
P valued | P = 0.0004 | P < 0.0001 | NA |
CGS-I (ITT)b | |||
Number of patients contributing to the analysis (%) | 111 (95.7) | 114 (95.8) | 112 (94.1) |
At baseline, mean (SD) | 4.8 (0.58) | 4.8 (0.48) | 4.8 (0.59) |
At specified end-of-treatment time point, mean (SD)d | 3.7 (0.92) | 3.6 (0.88) | 4.2 (1.06) |
Change from baseline, MMRM, LSM (SE) | −0.87 (0.07) | −0.91 (0.07) | −0.52 (0.07) |
Treatment group difference vs. placebo (95% CI) | −0.35 (−0.56 to −0.14) | −0.40 (−0.60 to −0.19) | NA |
P valued | P = 0.0002 | P < 0.0001 | NA |
Harms, n (%) (safety population) | |||
AEs | 81 (70.4) | 91 (77.8) | 81 (68.6) |
SAEs | 0 (0) | 1 (0.9) | 1 (0.8) |
WDAEs | 0 (0) | 2 (1.7) | 3 (2.5) |
Deaths | 0 | 0 | 0 |
Notable harms | |||
Injection-site reactions | There were no meaningful differences across the treatment groups in the proportions of patients with injection-site reactionse | ||
Weight gain | 15 (13) | 15 (12.8) | 4 (3.4) |
Akathisia | 3 (2.6) | 8 (6.8) | 5 (4.2) |
AE = adverse event; CGI-S = Clinical Global Impression–Severity of Illness; CI = confidence interval; ITT = intention to treat; LSM = least squares mean; LSMD = least squares mean difference; MMRM = mixed-effects model for repeated measures; NA = not applicable; PANSS = Positive and Negative Syndrome Scale; SAE = serious adverse event; SD = standard deviation; SE = standard error; WDAE = withdrawal due to adverse event.
aNumber of patients randomized was added by CADTH.
bITT population analysis. The primary efficacy end point was analyzed using an MMRM, with the difference from baseline in PANSS total score as the dependent variable, considering all available observations of PANSS total score at various visits. The MMRM included terms for treatment (placebo and RBP-7000 90 mg and 120 mg, i.e., risperidone ER [Perseris]), baseline PANSS total score, visit (5, 6, 8, and 9), and treatment-by-visit interaction as fixed effects.
cA comparison of each dose group with placebo was performed using the Dunnett procedure for controlling type I error at 2.5% (1-sided), and dosage groups that were significantly superior to placebo were identified. The superiority of RBP-7000 (i.e., risperidone ER [Perseris]) over placebo was demonstrated if 1 or both of the 1-sided P values for testing the null hypothesis or the secondary null hypothesis satisfied P < 0.0135.
dNot reported in the sponsor’s summary of evidence. Data were extracted from the CADTH Clinical Study Report.
eData were not presented in the sponsor’s summary of evidence.
Source: The sponsor’s submission,24 Clinical Study Report,23 and Nasser (2016).25
Critical Appraisal
The overall design of the included study (Study 09 to 0010) was appropriate with respect to randomization, blinding, allocation concealment, and standardized assessment of the primary outcomes. Based on the information available in the sponsor’s summary of the clinical evidence, the baseline demographic characteristics were generally well balanced across treatment arms in the pivotal study.
The proportion of patients who discontinued from the trials was relatively high (i.e., 22.4% to 29.4% across the treatment groups) for an 8-week study. Although the discontinuation rates were similarly high between the groups and not differential, this could potentially lead to an imbalance in baseline characteristics as the trial progressed, and efficacy data at 8 weeks were not available for a large number of patients. It is uncertain how the missing data could have impacted the study results. The mixed-effects model for repeated measures (MMRM) analysis that was used assumes the data are missing at random (MAR), which is often not the case in clinical trials. Although a pattern sensitivity analysis suggests the MAR assumption was not violated and supported the primary efficacy analysis,23 this approach also makes several assumptions to estimate the factors in the dropout pattern, which are unclear. Moreover, although a multiple-imputation approach was also used, this is valid only if the data missing from the dropouts were truly MAR. As a result, it is unclear what impact missing data may have had on the efficacy results observed.
The study was conducted in a distinct study population: patients were adults younger than 55 years of age; the majority of patients were male and Black with a PANSS total score of between 80 and 120, which may not fully represent the characteristics of Canadian patients with schizophrenia.
The 8-week duration of the double-blind randomized controlled trial was considered short to assess the long-term maintenance effect of treatment. However, the clinical expert consulted by CADTH for this review considered 8 weeks to be an adequate duration to demonstrate a treatment effect in clinical trials for acute exacerbation of schizophrenia. Nevertheless, the CADTH clinical expert indicated that while further studies are needed to assess the long-term efficacy (e.g., relapse, remission, hospitalization, and so forth) and safety of risperidone ER, other formulations of risperidone (oral and LAI drugs) have been available and used in Canadian clinical practice for a long time; therefore, a physician would likely have minimal concerns regarding the long-term efficacy and safety of risperidone ER.
Study 09 to 0010 was a placebo-controlled study. There was no direct or indirect treatment comparison evidence included in the sponsor’s submission that compared risperidone ER with oral risperidone or risperidone LAI (IM every 2 weeks) or other relevant AAP LAIs currently marketed in Canada. The sponsor indicated that the efficacy of risperidone ER was similar to that observed for oral risperidone26,27 and risperidone LAI.28,29 According to the expert consulted by CADTH for this review, it is generally accepted that all LAIs are of similar efficacy, and a lack of comparative data would be unlikely to influence the prescribing of risperidone ER, since the efficacy and safety profile of oral risperidone and risperidone LAIs has been well established, although the novel technology may make some clinicians cautious.
Economic Information
The sponsor submitted a cost comparison evaluating the annual cost of risperidone ER versus risperidone LAI, risperidone tablets, aripiprazole LAI, and paliperidone LAI products. No evidence was submitted to support the sponsor’s assumption of clinical similarity among LAI AAP comparators, nor was evidence submitted to support implicit assumptions of dose-equivalency between LAI AAPs. Therefore, any conclusions regarding incremental costs or savings associated with the use of risperidone ER are uncertain.
At the submitted prices of $456.18 (90 mg dose) and $608.22 (120 mg dose), the annual cost of risperidone ER is $5,474 to $7,299 per patient. This annual cost is more expensive than that of risperidone tablets ($349 to $524 per patient annually), but within the range of other LAI AAPs ($3,815 to $8,877 per patient annually). A small amount of savings in drug administration may also be seen when risperidone ER is compared with risperidone LAI due to its less frequent dosing (monthly versus every 2 weeks). However, the highest available dose of each LAI AAP comparator is the most frequently used. In order for the cost of the highest recommended dose of risperidone ER to equal that of the least expensive comparator at its highest recommended dose (aripiprazole LAI), the price of risperidone ER 120 mg would need to be reduced by 25%. These incremental costs (or savings) are based on publicly available list prices and may not reflect actual prices paid by Canadian public drug plans.
Conclusions
Based on the summary of clinical evidence submitted by the sponsor, risperidone ER 90 mg and 120 mg (SC once monthly) showed statistically significant improvements in schizophrenia symptoms compared with placebo after 8 weeks, as measured by PANSS total scores and CGI-S scores; however, given that improvements in these outcomes were also observed in the placebo group, the clinical importance of these results is uncertain. The findings pertaining to the potential impact of risperidone ER on patient HRQoL were not conclusive due to the trial’s methodological limitations. The safety of the once-monthly formulation appears to be consistent with the safety profile of risperidone (both oral and LAI every 2 weeks). Key evidence gaps include the short duration of the trial; risperidone ER is intended to be used as a chronic treatment and longer trials comparing it with the existing oral risperidone or LAI AAP drugs available in Canada for the maintenance treatment of schizophrenia are needed to adequately assess the long-term outcomes, including mortality, relapse, remission, and hospitalization.
At the submitted price, the annual cost of risperidone ER is $5,474 to $7,299 per patient, which is more expensive than risperidone tablets ($349 to $524 per patient per year), but within the range of other LAI AAPs ($3,815 to $8,877 per patient annually). However, the highest available dose of each LAI AAP comparator is the most frequently used. In order for the cost of the highest recommended dose of risperidone ER to equal that of the least expensive comparator at its highest recommended dose (aripiprazole LAI), the price of risperidone ER 120 mg would need to be reduced by 25%. No evidence was submitted to support the sponsor’s assumption of clinical similarity between LAI AAP comparators, nor any evidence to support implicit assumptions of dose-equivalency between LAI AAPs; thus, any conclusions regarding incremental costs or savings associated with the reimbursement of risperidone ER are uncertain, particularly when long-term clinical outcomes are considered. Additionally, these incremental costs or savings are based on publicly available list prices and may not reflect actual prices paid by Canadian public drug plans.
Introduction
Disease Background
Schizophrenia is a major severe, chronic, and disabling psychiatric syndrome that represents a heterogeneous group of disorders and may differentially affect presentation, course, treatment response, and outcome.1-3 The onset of schizophrenia typically occurs during middle to late adolescence or early adulthood and is diagnosed by specific signs and symptoms that prevent reality-based judgment.4,5 The most recent updated diagnostic criteria for schizophrenia is defined in the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5).4, 30 Schizophrenia is associated with hallucinations, delusions, cognitive impairment, disorganized thoughts, social withdrawal, and amotivation.6 Schizophrenia symptoms are often categorized as being either positive or negative in nature.11 Positive symptoms reflect a distortion or abundance of normal functions (e.g., delusion, conceptual disorganization, hallucinatory behaviour); negative symptoms reflect a loss or restriction of normal functioning (e.g., blunted affect, emotional withdrawal, poor rapport).12,31,32 Patients with schizophrenia are at an increased risk for suicide, substance abuse, homelessness, and unemployment.7,8
Global Burden of Disease studies reported that the age-standardized point prevalence of schizophrenia was 0.28% in 2016, with little variation across countries or regions.33 The prevalence of schizophrenia in Canada has been estimated to be approximately 0.95% of the population in 2016, with 1.08% in males and 0.82% in females.9 The incidence of schizophrenia in Canada has been estimated to be approximately 49 per 100,000 in 2016, with 58 per 100,000 in males and 41 per 100,000 in females.9 In 2018, schizophrenia was 1 of the top 20 causes of disability worldwide.30 Despite its low prevalence, its health, social, and economic burden is tremendous, not only for patients, but also for families, caregivers, and the wider society.10
Standards of Therapy
Antipsychotic medications form the cornerstone of treatment for schizophrenia, as they target its characteristic symptoms.6,11,12 The underlying principles for the administration of pharmacotherapy include the individualization of medication (including patient preferences), simple medication regimens, appropriate dosing, attention to side effect profiles, regular evaluation of responses (including AEs), and short- and long-term clinical efficacy, safety, and tolerability.8 The choice of antipsychotic medication should be made by the patient and physician together, taking into account the views of a caregiver, where appropriate.2 According to the clinical expert consulted by CADTH, the medications are given in oral and parenteral formulations and treat the positive symptoms of psychosis. The negative and cognitive symptoms, which are the most impairing for long-term function, have no approved medication treatments.
Based on input from the clinical expert consulted by CADTH for this review, the ideal medication for schizophrenia would reduce both positive and negative symptoms, would have a rapid onset of action, would have minimal adverse effects, and would be given once daily by mouth or monthly (or less often) as an injectable treatment. It would also be associated with improved daily function (such as improved social and occupational activities) and quality of life.
Existing antipsychotic therapies fall into 1 of 2 classes. The typical antipsychotic drugs (also known as conventional antipsychotic drugs or neuroleptics) are the first-generation antipsychotic class. These drugs have antagonistic activity at dopamine D2 receptors34 and are associated with an increased incidence of extrapyramidal symptom side effects.8 The second-generation or AAP drugs have antagonistic activity at both D2 receptors and serotonin (5-HT2a) receptors. The risk of the incidence of extrapyramidal symptoms appears reduced with AAPs; however, differences between typical antipsychotic drugs and AAP drugs can be variable in this respect.35 Both typical antipsychotic drugs and AAP classes are considered to be equally effective in the treatment of positive symptoms. AAPs appear to be more effective in the treatment of negative symptoms8; however, AAPs have an increased risk of weight gain and metabolic side effects.8
Risperidone, aripiprazole, and paliperidone are AAPs.
One major obstacle to the effective treatment of schizophrenia is nonadherence to medication, resulting in cycles of relapse.15,16 Studies have found that continuing treatment with LAI antipsychotic preparations in clinical practice outperforms oral antipsychotic medication in preventing rehospitalization.13
LAI antipsychotic drugs are usually used for patients with schizophrenia who have a history of good response to an oral antipsychotic but who have poor adherence to the medication.17 The American Psychiatric Association practice guidelines (2021) suggest that patients receive treatment with an LAI antipsychotic medication if they prefer such treatment or if they have a history of poor or uncertain adherence.30 The Canadian schizophrenia guidelines (2017) recommend that LAIs be used earlier in the course of treatment and should not be restricted to only those patients who have difficulty with adherence.2
In Canada, existing long-term AAP medications may be given as an IM injection every 2 weeks to every 3 months, depending on the medication (Table 3).14 The existing AAP LAIs available in Canada include risperidone LAI (IM monthly),18 aripiprazole LAI (IM monthly),19 paliperidone LAI (IM monthly),20 and paliperidone LAI (IM every 3 months)21 (Table 3). Canadian schizophrenia guidelines recommend that, after a change of antipsychotic medication in response to acute exacerbation of schizophrenia, the medication should be continued for at least 4 weeks, unless there are significant safety issues. If a partial response is observed at 4 weeks, the medication should be reassessed at 8 weeks, unless there are significant safety concerns.2 Following resolution of positive symptoms of an acute episode of schizophrenia, patients should be offered antipsychotic maintenance treatment for up to 5 years or longer.2
Drug
Risperidone for ER injectable suspension (risperidone ER [Perseris]), powder for suspension, 90 mg or 120 mg SC injection, was approved by Health Canada in November 2020.22 It is indicated for the treatment of schizophrenia in adults.22 Like other AAPs, the exact mechanism of risperidone ER is unclear. The effect on schizophrenia could be mediated through a combination of D2 and 5-HT2 receptor antagonism. In addition, risperidone has a high affinity to alpha-adrenergic receptors. Antagonism at receptors other than D2 and 5-HT2 may explain some of the other effects of risperidone. Risperidone does not bind to dopamine D1 and has no affinity (when tested at concentrations > 10−5 M) for muscarinic cholinergic receptors.22
The Health Canada–recommended dose is 90 mg or 120 mg once monthly by abdominal SC injection. Based on average plasma concentrations of risperidone and total active moiety, risperidone ER 90 mg corresponds to 3 mg/day oral risperidone and risperidone ER 120 mg corresponds to 4 mg/day oral risperidone. No loading dose is required. Patients who are on stable oral risperidone doses lower than 3 mg/day or higher than 4 mg/day may not be candidates for risperidone ER.22 Similar to oral risperidone, risperidone LAI monthly, and other AAP LAIs, the main safety concern is increased mortality in elderly patients with dementia. Health Canada has not authorized an indication for risperidone ER for pediatric use.22 Risperidone ER should be administered by a health care professional only.
Risperidone tablets have been approved for the treatment of schizophrenia in Canada since 1993.36 Risperidone LAI for IM administration every 2 weeks has been approved for the treatment of schizophrenia in Canada since 2004.37 Oral risperidone and risperidone LAI IM every 2 weeks were not previously reviewed by CADTH. Risperidone ER is a new formulation of risperidone for once-monthly SC administration.
The objective of this report is to review the beneficial and harmful effects of risperidone for ER injectable suspension powder, 90 mg or 120 mg SC injection, for the treatment of schizophrenia in adults.
Table 3: Key Characteristics of Risperidone and Other LAIs
Characteristic | Risperidone ER (Perseris)22 | Risperidone LAI (Risperdal Consta)18 | Oral Risperidone38 | Aripiprazole (Abilify Maintena, IM)19 | Paliperidone palmitate (Invega Sustenna, IM monthly)20 | Paliperidone palmitate (Invega Trinza, IM every 3 months)21 |
|---|---|---|---|---|---|---|
Mechanism of action | Unclear. The effect on schizophrenia could be mediated through a combination of D2 and 5-HT2 receptor antagonism. In addition, risperidone has a high affinity to alpha-adrenergic receptors. Antagonism at receptors other than D2 and 5-HT2 may explain some of the other effects of risperidone. Risperidone does not bind to dopamine D1 and has no affinity (when tested at concentrations > 10−5 M) for muscarinic cholinergic receptors. | Unclear. Effects may be mediated through a dose-dependent combination of partial agonist activity at D2 and 5-HT1a receptors and antagonist activity at 5-HT2a receptors. | Unknown. However, effects on schizophrenia may be mediated through a combination of D2 and 5-HT2a receptor antagonism. Antagonism at receptors other than D and 5-HT2a may explain some of the other effects. | |||
Indicationa | For the treatment of schizophrenia in adults. | For the management of schizophrenia. | For the acute treatment and maintenance treatment of schizophrenia and related psychotic disorders. | For the maintenance treatment of schizophrenia in stabilized adult patients. | For the treatment of schizophrenia. | For the treatment of schizophrenia in adult patients. Paliperidone palmitate IM every 3 months is to be used only after paliperidone palmitate IM monthly has been established as adequate treatment for at least 4 months. |
Route of administration | SC injection | IM injection | Oral | IM injection | IM injection | IM injection |
Recommended dose | Initial dose: Risperidone ER at a dose of 90 mg or 120 mg once monthly by SC injection; do not administer a loading dose. | 25 mg every 2 weeks. | On a schedule of either once a day or twice a day, generally beginning with 1 mg to 2 mg per day. The dose should be adjusted gradually over several days, based on clinical response to a target dose of 4 mg to 6 mg per day. Some patients may benefit from lower initial doses and/or a slower adjustment schedule. | 400 mg once monthly. | Initial dose: 150 mg on day 1 and 100 mg on day 8 (1 week later). Monthly maintenance dose: 75 mg (can be adjusted from 50 mg to 150 mg). | Initial dose: The equivalent of 3.5-fold of a 1-month dose of paliperidone palmitate. Following the initial dose, it should be administered every 3 months. If needed, dose adjustment can be made every 3 months in increments within a range of 175 mg to 525 mg, based on individual patient tolerability and/or efficacy. |
Serious side effects and safety issues | Increased mortality in elderly patients with dementia. | |||||
5-HT2 = serotonin type 2; D2 = dopamine type 2; ER = extended release; IM = intramuscular; LAI = long-acting injectable; M = molar concentration; SC = subcutaneous.
aHealth Canada indication.
Stakeholder Perspectives
Patient Group Input
This section was prepared by CADTH staff based on the input provided by patient groups.
About the Patient Groups and Information Gathered
One patient group, IAM (formerly the Schizophrenia Society of Ontario), responded to the call for patient input for this reimbursement review. IAM is a connector, collaborator, and solution-driven organization, supporting, innovating, and driving change for better mental health. IAM innovates in mental health with a focus on returning solutions back to communities through partnership and collaboration. Historically serving those impacted by psychotic illnesses such as schizophrenia and psychosis, IAM serves anyone impacted by serious mental illness and their families, caregivers and supports, and communities. IAM’s direct services include 1-on-one and group counselling using cognitive behavioural therapy-based interventions, information and system navigation, training for front-line workers, community education, and more.
This submission was based on IAM’s 40-year history of serving adults with schizophrenia and other psychotic illnesses and their families, service providers, and communities. Much of it came directly from IAM’s work with clients and the expertise of IAM front-line staff. IAM’s clients are primarily adults from the southern and central regions of Ontario, a number of clients from northern regions of Ontario that are served virtually, and health service and social service providers who work with individuals with psychotic illnesses, including schizophrenia. In addition, IAM also drew some information from a survey of the IAM client network that was conducted by Ipsos in 2018. In the Ipsos survey, respondents self-described as: personally diagnosed (12%), a caregiver (50%), a family member or friend of someone diagnosed (63%), or working in social services (18%). About 77% of survey respondents were born in Canada, with 83% describing themselves as White. A total of 55% of respondents came from the Greater Toronto Area, while 20% came from southwestern Ontario, 14% came from Eastern Ontario, 5% from Central Ontario, and 4% from Northern Ontario. An additional 2% came from outside Ontario.
The funding information for the patient group is included in the patient group input submission published on the CADTH website.
Disease Experience
The IAM patient input submission included individuals with schizophrenia and other related illnesses with psychosis as a main feature. It indicated that many of the patients experience symptoms of psychosis without having a formal diagnosis. Often, their symptoms have a significant impact on day-to-day functioning. The patient’s experiences vary widely but typically involve some levels of cognitive impairment, delusions, and hallucinations. The cognitive impairment can range from mild to severe but is typically strongest in the cognitive domains of working and episodic memory, attention, processing speed, problem-solving, and social cognition. A large number of patients also experience anosognosia, a lack of insight into their illness, which often impacts their ability and motivation to access treatment and supports. This symptom can cause significant strain in relationships, including those with caregivers and family members, ultimately leading to social isolation and a lack of supports for the individual with the illness.
Experience With Treatment
In the survey of the IAM client network, 55% of respondents were taking LAI medications or had taken them before, while 98% were taking or had taken oral medications to manage their schizophrenia or psychosis symptoms. The most common side effects they reported were drowsiness (58%), dry mouth (50%), restlessness (50%), dizziness (45%), muscle stiffness (45%), constipation (43%), and anxiety (43%).
Of those taking LAI medications, the most common stated benefits were convenience and not needing to remember to take it every day. The difficulties that were stated most frequently were pain at the injection site and frequent travel to clinics.
While 63% of IAM’s respondents said they paid for their medications with government insurance, 23% paid out of pocket for their medications. A total of 63% of respondents agreed it is difficult to pay for health care bills, including medication, visits to specialists, counselling, and so forth.
Twenty-three percent of respondents identified the cost of medications as a significant challenge to access. Other challenges identified included: preferred medication not being covered by public drug programs (20%) and the wait times for approvals of certain medications under the Ontario Public Drug Program Exceptional Access Program and Health Canada Special Access Program (13%).
Patients expect new, quick, simple, convenient, and affordable access to a wide range of treatments and medications to suit their unique needs and that can improve adherence and allow for the highest level of daily functioning and symptom reduction while managing side effects.
Improved Outcomes
The patient group indicated that the right medication allows for the highest level of daily functioning while managing side effects, which is often achieved through a trial-and-error process of trying several different medications and dosages to find what works best. This process can make it difficult for patients to adhere to treatment and is exacerbated by additional challenges, such as difficulty accessing psychiatrists, obtaining prescriptions, and understanding medication options; the cost of medications; and the wait times to access medications through public drug programs. When the right combination of therapy and medication is determined, individuals may still experience relapse and may require extensive supports to adhere to the treatment plan. This trial-and-error practice of finding the right medication for each individual would be improved by having quick, simple, and affordable access to a wide range of treatments and medications to suit unique needs.
Experience With the Drug Under Review
The patient group indicated its patients did not have experience with risperidone ER (Perseris).
Additional Information
The patient group indicated that the greater the variety and affordability of medications on the market, the more treatment adherence we are likely to see among patients with schizophrenia and psychosis and, by extension, greater levels of recovery. IAM advocates for a wide selection of reimbursable medications in the Canadian marketplace.
Clinician Input
Input From the Clinical Expert Consulted by CADTH
All CADTH review teams include at least 1 clinical specialist with expertise regarding the diagnosis and management of schizophrenia in adults. Clinical experts are a critical part of the review team and are involved in all phases of the review process (e.g., providing guidance on the development of the review protocol, assisting in the critical appraisal of clinical evidence, interpreting the clinical relevance of the results, and providing guidance on the potential place in therapy). The following input was provided by 1 clinical specialist with expertise in the diagnosis and management of schizophrenia in adults.
Unmet Needs
The clinical expert consulted by CADTH for this review indicated that antipsychotic medications do not reliably improve psychosocial function. Current treatments do not treat the underlying pathophysiology, which is not well understood. Medications have burdensome side effects which, in some cases, are life-threatening (diabetes, neuroleptic malignant syndrome) or irreversible (tardive dyskinesia). Antipsychotic drugs treat only 1 of 3 symptom domains—they do not treat negative and cognitive symptoms. Treatments for refractory disorders are few and have severe adverse effects or are inconvenient.
Place in Therapy
The clinical expert did not consider risperidone ER to be a breakthrough treatment. Its mechanism of action is the same as all other second-generation antipsychotic drugs, and the side effects are not anticipated to be different from other forms of risperidone and paliperidone. The clinical expert does not anticipate that risperidone ER would be used as a first-line treatment; it might initially be used as a second-line treatment. It appears to be a convenient LAI that does not require concomitant oral medication to initiate, which may allow earlier hospital discharge or easier community initiations. Since it is based on a familiar drug, clinicians could be quick to adopt it.
The clinical expert stated that the patient should have been given oral risperidone to confirm the lack of an allergy to the molecule. Although failure of adherence to oral medication could be a requirement before giving an LAI, patient preference should also be considered. LAIs reduce rehospitalization; therefore, they can result in an overall reduction in health resource utilization, so there should not be large barriers to their use.
Patient Population
The clinical expert indicated that adult patients with well-diagnosed schizophrenia or schizoaffective disorder who have responded to oral risperidone would be the ideal candidates. The lack of significant extrapyramidal symptoms on oral risperidone would suggest tolerability of the ER version. Patients with tardive dyskinesia would need a careful risk-benefit analysis, as would those with elevated prolactin. Geriatric and pediatric patients would not be good candidates, given the lack of data in these groups and the risk of stroke in older adults.
The suitable patients would be identified using a thorough psychiatric history, mental status exam, and focused physical exam. Laboratory testing would play a role, as patients with hyperglycemia or a metabolic disorder might be given an alternative antipsychotic. Comorbid mental and addictive disorders may complicate treatment and play a role in treatment selection. Given that most patients who receive LAI antipsychotic drugs have a history of poor adherence, limited insight, and relapses, collateral information is crucial in obtaining an adequate history. Family involvement, when available, is important in assessing patients and engaging them in care. Without these efforts, appropriate selection is impeded and treatment may be unsuccessful.
Patients with appropriately diagnosed treatment-resistant illness would be unlikely to benefit; whenever possible, they should receive clozapine. Patients with an allergy to or a history of poor tolerance of risperidone or paliperidone are not suitable. Finally, patients with atypical forms of psychosis or drug-induced psychosis often require antipsychotic therapy, although these conditions are not studied adequately and Health Canada does not usually approve medications for them. Likewise, pediatric and geriatric patients have not been enrolled in trials to allow assessment of the appropriateness of risperidone ER in those populations.
There exists no reliable a priori method to identify patients who will respond to a given antipsychotic, especially among those who are naive to such treatment. However, patients whose disorder has not responded to 2 adequate antipsychotic treatment trials would have a low likelihood of benefiting from risperidone ER. Those whose apparent poor treatment response can be linked to medication nonadherence would be good candidates for risperidone ER, assuming there are no contraindications to risperidone. Patients with comorbid mental and addictive disorders will require a thorough assessment of those conditions, often once their psychosis is adequately treated. This may influence the decision on whether to prescribe an LAI.
Assessing Response to Treatment
The clinical expert indicated that, most precisely, a 20% reduction of positive symptoms on a valid psychosis rating scale, such as the PANSS or Brief Psychiatric Rating Scale, is the most reliable way to confirm response to antipsychotic medication; however, such scales are often not used in clinical practice. Therefore, the routine mental status exam, which thoroughly assesses hallucination, delusions, and disorganized thought and behaviour, is appropriate. Documented improvement of such findings over an 8-week course of therapy would indicate a response to treatment, along with collateral input from caregivers, when available, indicating reduced behavioural signs of psychosis. Adherence to treatment must also be assessed; with LAI medication, this is less difficult to monitor.
A clinically meaningful improvement was usually defined as a 20% reduction of positive symptoms on a valid psychosis rating scale, (e.g., PANSS). In addition, it would also involve improved function, as manifested by more appropriate social interactions, greater consistency in activities of daily living, and a reduction in the risk of self-harm or aggression. The definition depends in part on the patient’s baseline function and the treatment setting. If a patient is in hospital, readiness for transition to the community is an important milestone. If they are in the community, readiness to engage in rehabilitation or even to return to work or school are relevant milestones.
In the acute phase in a community setting, treatment response should be assessed at least twice a week, which may be done through virtual or in-person visits combined with collateral input. If in hospital, if the patient is at risk of aggression or suicide, daily assessment by a physician or nurse is necessary. Once a patient is in the stabilization phase and in the community, assessment once every 1 or 2 weeks is adequate. In the maintenance phase, once a month or even every 3 months can be adequate. An assessment of adverse effects must be done regularly, as well; this includes administering involuntary movement exams, measuring weight and waist circumference, assessing fasting glucose and lipids, and determining whether prolactin is elevated.
Discontinuing Treatment
The clinical expert indicated that patients will often ask that an antipsychotic medication be discontinued prematurely because of adverse effects or because they lack insight into ongoing symptoms and risk of relapse. If a patient has been symptom-free and had good functional recovery for 2 or more years, discontinuation can be considered. In cases where multiple relapses have occurred or the acute episodes have led to a high risk of suicide or aggression, a longer symptom-free interval is necessary before discontinuation. If psychiatric comorbidity or a substance-use disorder is present, the decision will have to take this into account. Adverse effects may require rethinking treatment; these effects include weight gain and metabolic syndrome, involuntary movements, and hyperprolactinemia with sexual dysfunction or galactorrhea. A feasible alternative medication will have to be available in those cases; otherwise, efforts to manage and minimize adverse effects will be necessary. Finally, patients who develop a resistant disorder will require a switch to clozapine, the only medication indicated for treatment-resistant psychosis.
Prescribing Conditions
The clinical expert indicated that risperidone ER could be initiated in hospital or in the community, typically in an acute psychiatry unit or a community or tertiary mental health program. Family physicians who are familiar with antipsychotic medication could also initiate treatment for patients with mild exacerbations who have demonstrated tolerability to risperidone oral medication.
For mild cases of schizophrenia with a low risk of behavioural and psychosocial disruptions, a family physician can manage antipsychotic treatment, including LAIs. For hospitalized patients, in cases with a high risk of self-harm or aggression or with concurrent addiction or psychiatric comorbidity, a psychiatrist should assess and prescribe treatment in the acute and stabilization phases. In the maintenance phase, such patients could be transferred to general practice with the availability of psychiatric consultation.
Clinician Group Input
No clinician group input was received for this review.
Drug Program Input
The drug programs provide input on each drug being reviewed through CADTH’s reimbursement review processes by identifying issues that may impact their ability to implement a recommendation. The key implementation questions and corresponding responses from the clinical expert consulted by CADTH are summarized in Table 4.
Table 4: Summary of Drug Plan Input and Clinical Expert Response
Drug program implementation questions | Clinical expert response |
|---|---|
Most jurisdictions require that previous treatment failure or intolerance to oral therapy and conventional LAIs before approving reimbursement of a new treatment. Would risperidone ER be used for a patient who had failed oral therapy or conventional LAIs? | The use of this medication should not be restricted to patients who failed oral therapy for nonadherence, as some patients may prefer an LAI and, given evidence for reduced risk of hospitalization with LAIs, clinicians should offer this treatment to any patients who might benefit. On the other hand, experience in Canada with risperidone ER is limited, so prescribers may hesitate to use it as first-line therapy. |
Are there special concerns with the use of risperidone ER (use of the Atrigel delivery system) in pregnant patients? | Risperidone ER has not undergone adequate study in pregnant women to determine whether it is safe, especially in the first trimester of pregnancy. The benefits may outweigh the risks of risperidone ER for certain patients, and close monitoring would be necessary if it were prescribed to a pregnant patient. The disadvantage of an LAI in this situation is that it cannot be rapidly discontinued in the event of adverse effects. |
Does there have to be a washout period for patients currently on oral therapies? If so, does the clinical expert have any concerns? | Patients who undergo a switch of antipsychotic usually have a “cross taper” in which the first medication is gradually reduced and discontinued over several weeks while the second is gradually increased. A washout is not typically necessary and, if a patient has no effective antipsychotic on board for a substantial period, increased symptoms or relapse may happen. This would usually involve oral medication as opposed to an LAI; the latter would be started after a patient has taken an oral form of the medication and the prescriber is satisfied that the patient is not allergic or is tolerating it well. Initiating risperidone ER in a patient on a different LAI antipsychotic (within 120 days before study23) has not been studied. |
Would risperidone ER be used for the prevention of relapse and for maintenance? The pivotal study was in patients with acute exacerbations. Are there data on the prevention of relapse and on maintenance? | Risperidone ER was shown in the regulatory trial to be effective for acute exacerbation of schizophrenia. Given experience with other risperidone and paliperidone LAIs, it is justified to assume that risperidone ER will be effective for maintenance therapy, but the sponsor and distributor should undertake a long-term study to confirm this. |
How should “treatment-resistant” or “refractory disease” be defined? Based on what parameters? | Treatment-resistant schizophrenia is diagnosed in patients who do not have at least a 20% reduction in positive symptoms in response to either of 2 medication trials of different antipsychotic drugs at adequate dose and duration. These patients should receive clozapine, whenever possible. A significant minority of patients also fail to meet response criteria for clozapine, and those patients are considered refractory. Unfortunately, no medication is approved for that form of the disorder. |
ER = extended release; IM = intramuscular injection; LAI = long-acting injectable.
In addition to the questions presented in Table 4, the public drug plans noted that the LAI antipsychotic drugs previously reviewed by CADTH include aripiprazole IM, paliperidone IM monthly (Invega Sustenna), and paliperidone IM (Invega Trinza). Furthermore, the drug programs indicated that the discontinuation criteria for AAP drugs are typically not specified. The drug plans also noted that risperidone ER may offer some advantage over other LAIs, since it reaches therapeutic plasma concentrations on day 1, requiring no loading dose or supplemental oral dosing. They also noted that the product monograph does specify that tolerability must be established with oral risperidone; risperidone ER is available as an SC injection, which may be less painful than an IM injection but still has to be administered by a health care professional. Finally, the drug programs noted economic issues, including the existence of negotiated prices for other LAI AAPs, the potential for a reduction in injection-related costs with the use of risperidone ER compared with risperidone LAI every 2 weeks, the high indirect costs associated with schizophrenia such as unemployment, and the importance of adherence to therapy in a condition characterized by anosognosia (lack of insight by the patient into their condition).
Clinical Evidence
Sponsor’s Summary of the Clinical Evidence
Note that the clinical evidence summarized in this section was prepared by the sponsor in accordance with the CADTH tailored review process and has not been modified by CADTH.
Table 5: Details of Included Studies
Details | Study 09-0010 |
|---|---|
Study design and population | |
Study design | phase III, randomized, double-blind, placebo-controlled study |
Locations | 33 centers in the US |
Randomized (N) | 354 |
Inclusion criteria |
|
Exclusion criteria |
|
Drugs | |
Intervention | Risperidone ER 90 mg or 120 mg subcutaneous injections on Days 1 and 29 in the lower quadrant of the abdomen rotating right and left |
Comparator(s) | Subcutaneous injection of placebo on Days 1 and 29 in the lower quadrant of the abdomen rotating right and left |
Duration | |
Phase | |
Run-in | Screening visit 3-8 days before start of double-blind treatment |
Double-blind | 57 days |
Follow-up | 1 week |
Outcomes | |
Primary End Point | Change From Baseline to End of Treatment in the Positive and Negative Syndrome Scale (PANSS) Total Score |
Secondary and exploratory end points |
Tertiary:
Exploratory: The change in individual PANSS components from baseline to Day 57, including: positive scale score, negative scale score, general psychopathology score |
Notes | |
Publications | Nasser AF, et al, (2016),25 Isitt JJ, et al., (2016),39 |
CGI-S = Clinical Global Impression - Severity Scale; EQ-5D-5L = EuroQol-5 dimension-5 level; LAI = long-acting injectable.
MSQ = Medication Satisfaction Questionnaire; PANSS = Positive and Negative Syndrome Scale; SWN-S = Subjective Well-being under Neuroleptic Treatment – Short Version.
Source: The sponsor’s submission.24
Description of Studies
The efficacy for risperidone ER was demonstrated in a phase III, randomized, double-blind, placebo-controlled clinical study (Study 09 to 0010) that was performed at 33 centres in the US.23 The study was designed to evaluate the efficacy, safety, and tolerability of risperidone ER (90 mg and 120 mg) compared with placebo in patients experiencing acute exacerbations of schizophrenia. The primary objective of this study was to assess the efficacy of risperidone ER (90 mg and 120 mg) compared with placebo on the symptoms of acute schizophrenia over an 8-week treatment period using the change from baseline (defined as the last no missing value before the first dose of risperidone ER or placebo on day 1) to day 57 in the PANSS total score, which is the sum of all 30 PANSS items.
The diagnosis of schizophrenia was defined by the Diagnostic and Statistical Manual for Mental Disorders, Fourth Edition, Text Revision (DSM-IV-TR) criteria. Subjects were required to have a PANSS total score of 80 to 120, inclusive (moderate to severely ill), at visit 1 (the initial screening visit, which occurred 3 to 8 days before the start of double-blind treatment) without an improvement in the PANSS total score of ≥ 20% between visit 1 and visit 3 (day 1 of double-blind treatment/pre-dose).
This was an inpatient study that started with the initial screening at visit 1 and continued through visit 9 (end of study [EOS]/early termination [ET]). At the time of screening, the patients could be inpatients or outpatients and either on treatment or currently not on any antipsychotic treatment. At visit 1 (screening visit), every patient received a 0.25 mg tablet of oral risperidone and a 0.25 mg tablet of oral risperidone on the following day to assess medication tolerability (e.g., acute hypersensitivity reaction). Patients were placed into an inpatient setting, if not already hospitalized, and tapered off their current oral antipsychotic medication over a period of 3 to 8 days. Patients were then randomized to receive 2 doses of risperidone ER (90 mg or 120 mg) or placebo at 28-day intervals (on day 1 and day 29). A total of 538 patients were screened for study participation at 33 centres in the US, of which 354 patients were randomly assigned to treatment.
Figure 1: Study 09 to 0010: Screening, Treatment and Follow-Up Visits
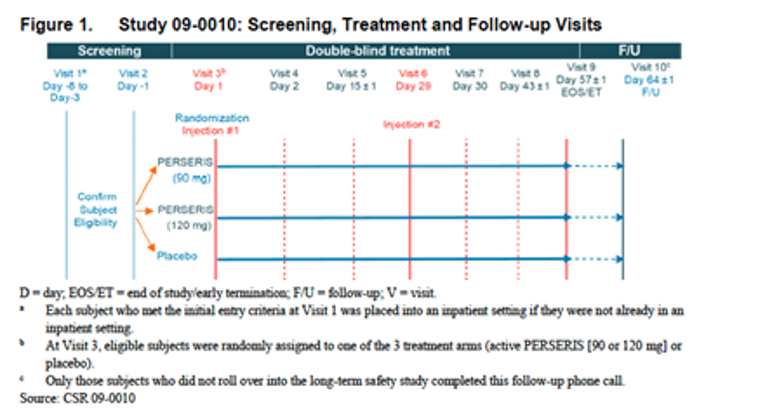
Source: Clinical Study Report.23
Populations
Inclusion Criteria
The population selected for the placebo-controlled study was adult subjects (ages 18 to 55 years) with a confirmed diagnosis of schizophrenia who had a total PANSS score of 80 to 120, inclusive, and who were in an acute exacerbation phase of illness. Specifically, subjects had a PANSS total score between 80 and 120, inclusive, at Visit 1, and a score of ≥ 4 on at least 2 of the following 4 items of the PANSS positive subscale: hallucinatory behaviour, delusions, conceptual disorganization or suspiciousness. The diagnosis was determined to be valid by the SAFER interview (i.e., State, Assessability, Face, Ecological, and Rule). An acute exacerbation was demonstrated by all of the following:
recent marked deterioration in baseline (usual, not acute) status, as evidenced by impairment in functional capabilities secondary to schizophrenia symptoms.
start of exacerbation occurring ≤ 8 weeks before the screening visit (Visit 1).
subject would have benefited from psychiatric hospitalization or continued hospitalization (if hospitalized before screening) for symptoms of schizophrenia.
Exclusion Criteria
The most relevant of these criteria in relation to efficacy were the following:
Subjects who had improvement in their PANSS total score of 20% or greater between Visit 1 (the initial screening visit) and Day 1 (first injection).
Subjects who had been hospitalised for more than 14 days in the current episode before study screening (Visit 1).
Subjects who received any risperidone or 9-hydroxyrisperidone LAI formulation or other LAI antipsychotic within the 120 days before study screening (Visit 1).
Subjects taking daily oral risperidone at a dose ≥ 6 mg/day.
Subjects taking mood stabilizers, or inducers or inhibitors of cytochrome P450 (CYP) 2D6 within 5 half-lives of the medication or 30 days for fluoxetine and medications containing fluoxetine before Day − 1 (Visit 2). Medications, which in the opinion of the investigator in conjunction with the medical monitor could have been expected to significantly interfere with the metabolism or excretion of risperidone and/or 9-hydroxyrisperidone, could have been associated with a significant drug interaction with risperidone, could have posed a significant risk to subjects’ participation in the study or potentially confounded the assessment of the primary outcome measure of risperidone ER were also restricted.
Subjects with treatment-resistant schizophrenia, as judged by the investigator, who had been treated with antipsychotics for adequate durations and with adequate dosages. This also included subjects who had been treated at any time with clozapine for treatment-resistant schizophrenia and subjects with documented failure of 2 or more antipsychotic medications or had a documented failure to risperidone/9-hydroxyrisperidone.
Baseline Characteristics
Table 6: Summary of Baseline Characteristics — Study 09 to 0010 (Safety Population)
Characteristics | Risperidone ER 90 mg (N=116) | Risperidone ER 120 mg (N=119) | Placebo (N=119) |
|---|---|---|---|
Gender, n (%) | |||
Male | 95 (82.6) | 86 (73.5) | 87 (73.7) |
Female | 20 (17.4) | 31 (26.5) | 31 (26.3) |
Age (Years) | |||
Mean (SD) | 40.5 (9.41) | 40.6 (9.45) | 42.4 (9.07) |
Min, Max | 19, 55 | 18, 54 | 20, 55 |
Race, n (%) | |||
White | 28 (24.3) | 30 (25.6) | 27 (22.9) |
Black or African American | 83 (72.2) | 83 (70.9) | 88 (74.6) |
Asian | 1 (0.9) | 3 (2.6) | 1 (0.8) |
Native Hawaiian or Other Pacific Islander | 1 (0.9) | 1 (0.9) | 1 (0.8) |
Other | 2 (1.7) | 0 | 1 (0.8) |
Weight (kg) | |||
Mean (SD) | 90.6 (18.90) | 89.0 (20.46) | 91.8 (22.89) |
Median | 88.30 | 83.60 | 87.45 |
Min, Max | 52.2, 136.8 | 51.7, 161.9 | 51.7, 180.5 |
BMI (kg/m2) | |||
Mean (SD) | 29.6 (5.94) | 29.4 (6.66) | 30.7 (7.29) |
Median | 29.210 | 27.550 | 29.640 |
Min, Max | 18.55, 49.95 | 17.70, 57.08 | 17.89, 55.51 |
ER = extended release; N = number of patients randomized, added by CADTH; SD = standard deviation; Max = maximum; Min = minimum.
Source: CSR 09 to 0010 Table 9.1.3.2.23
The study population was representative of the expected population (patients with schizophrenia) for risperidone ER. The treatment groups were similar across most demographic and baseline characteristics for the Safety population. However, the proportion of male subjects was higher for the risperidone ER 90-mg group compared with the risperidone ER 120-mg and placebo groups. Notably, most subjects in each group in this study were black or African American (71% to 75% per group). The treatment groups were similar in most demographic and baseline characteristics for the ITT and the PP populations (Table 6).
Interventions
At Visit 1, every subject received a 0.25-mg tablet of oral risperidone and a 0.25-mg tablet of oral risperidone on the following day. The 0.25-mg oral dose on these 2 days was given to confirm the subject did not have any intolerable reaction to risperidone before receiving a long-acting injection of risperidone. Subjects who were not able to tolerate these 2 doses of risperidone were not allowed to continue in the study.
Subjects were placed into an inpatient setting, if not already hospitalized, and tapered off of their current oral antipsychotic medication. Following the screening period, eligible subjects remained in an inpatient setting to continue into the double-blind treatment period. At Visit 3, which was the start of the 8-week (56-day), double-blind treatment period, subjects were randomly assigned to receive either an injection of risperidone ER (90 mg or 120 mg) or placebo on Days 1 and 29. No oral risperidone was permitted during the double-blind treatment period.
Active study treatment:
Subjects received 2 single, blinded SC doses of 90 mg risperidone ER on Day 1 (Visit 3) and Day 29 (Visit 6).
Subjects received 2 single, blinded SC doses of 120 mg risperidone ER on Day 1 (Visit 3) and Day 29 (Visit 6).
Placebo treatment:
Subjects received 2 single, blinded SC doses of placebo on Day 1 (Visit 3) and Day 29 (Visit 6).
Placebo was supplied as just the ATRIGEL Delivery System with a sterile 18-gauge, 1-inch needle.
Concomitant therapies were defined as all medications the subject received during the course of the study (including prescribed and over-the-counter medications, herbal preparations and vitamins, and nutritional supplements). Subjects continued to take any previously prescribed non-antipsychotic medications during the study, as directed by the Investigator or medically qualified subinvestigator. Non-antipsychotic medications could also have been initiated during the study at the discretion of the Investigator or medically qualified subinvestigator, excluding those defined as exclusionary in the protocol.
Only unblinded personnel prepared the study drug. The final product (risperidone ER, RBP-700, Perseris) for each dose level consisted of a 2-syringe mixing system (Syringe A and Syringe B). Syringe A contained the ATRIGEL Delivery System, and Syringe B contained the active risperidone in a 90 mg or 120 mg dosage. Prior to administration, the syringes were coupled and the product was mixed by pushing the contents back and forth between the 2 syringes to provide a homogenous suspension. The syringes were uncoupled and a sterile 18-gauge, 1-inch needle was affixed to Syringe A for injection.
Criteria for Rescue Medication Use
If at any time during the subject’s participation in the study, it is deemed necessary to provide rescue medication, whether due to breakthrough symptoms or the SC injection of risperidone ER not maintaining the subject for 28 days, it was at the judgment of the principal investigator or medically qualified Sub-Investigator.
Concomitant use of benzodiazepines and antiparkinsonian medication could be used to treat and alleviate suffering and/or control behaviour of study subjects who may have experienced exacerbations of psychotic symptoms and/or side effects during the study treatment period. Benzodiazepines could not be administered within the 8-hour period before efficacy scale (PANSS, CGI-S) assessment.
Treatment of Agitation and Anxiety Related to Acute Exacerbation Of Psychosis: Concomitant use of oral lorazepam in divided doses for treatment of agitation and/or anxiety due to exacerbated psychotic state in subjects was allowed. Lorazepam could have been used on an as-needed basis only, to be given in divided doses of 1 to 2 mg and were not to exceed a total dose per day of 8.0 mg/24 hours allowed starting Day –7 through Day − 1. A protocol for tapering lorazepam after 1 week was provided.
Treatment of Insomnia: Concomitant use of oral zoldipem once daily at doses of 5 to 10 mg, was not to exceed 10 mg/24hrs total dose, throughout the study protocol. Zoldipem should not be used within 8 hours before rating symptoms using efficacy scales.
Treatment of Extrapyramidal Symptoms (EPS): In the event of newly emergent EPS during the treatment period, concomitant use of antiparkinsonian medications was allowed on an as-needed basis only. Concomitant use of oral anticholinergic medications including benztropine mesylate at doses of 1 to 2 mg was not to exceed 6.0 mg/24hour period. These medications were not used within 8 hours before ratings symptoms using efficacy or EPS scales.
Stopping Criteria for the Intervention
The principal investigator or medically qualified Sub-Investigator could choose to withdraw a subject from the study for any of the following reasons:
Protocol deviation that might compromise data integrity, protocol compliance, or subject safety,
An adverse event was reported that compromised or potentially compromised subject safety,
The subject requested to be discontinued from the study (i.e., subject declined further study participation).
If pregnancy in a subject was confirmed at any time during the study, the subject was immediately discontinued from the study.
Outcomes
The primary efficacy end point in this study was the change in Positive and Negative Syndrome Scale (PANSS) total score from baseline to Day 57. Sensitivity analyses using the Per Protocol population and adjusting for site and treatment duration were also produced focusing on Day 57. The secondary efficacy end point was the change in Clinical Global Impression-Severity of Illness (CGI-S) score from baseline to Day 57.
Health economics and outcomes assessments conducted prospectively during the study included the following:
health-related quality of life (QoL) using the EQ-5D-5L;
subjective well-being using the SWN-S;
subject satisfaction with medication using the MSQ.
The PANSS is a validated, multi-item inventory, composed of 5 subscales to evaluate positive symptoms, negative symptoms, disorganized thoughts, uncontrolled hostility/excitement and anxiety/depression. This medical scale was designed to measure symptom severity among subjects with schizophrenia, using a 30-item, 7-point rating scheme comprising adaptations of 18 items from the Brief Psychiatric Rating Scale (BPRS) and 12 items from the Psychopathology Rating Schedule (PRS). Each item on the PANSS is accompanied by a complete definition as well as detailed anchoring criteria for all 7 rating points, which represent increasing levels of psychopathology: 1 = absent, 2 = minimal, 3 = mild, 4 = moderate, 5 = moderate severe, 6 = severe and 7 = extreme. The theoretical constructs of positive and negative symptoms in patients with schizophrenia are measured by the positive and negative subscales and their use is designed to promote uniformity and reliability in the theoretical constructs of the symptoms. The general psychopathology subscale measures nonspecific symptoms such as somatic concern, anxiety, guilt feelings, disorientation and poor impulse control among others. The PANSS is scored by summation of ratings across items, such that the potential ranges are 7 to 49 for both the positive and negative scales and 16 to 112 for the general psychopathology scale. Decreased scores over time represent improvement. The PANSS range for assuring stability is a PANSS general psychopathology scale score of 70 or less, with no score of 4 or more in the positive subscale.
The CGI-S scale was the secondary efficacy parameter in the phase III Study. The subject’s clinical status was based on the answer to the following question: “Considering your total clinical experience with this particular population, how mentally ill is the patient at this time?”. Response choices included the following: 0 = not assessed; 1 = normal, not at all ill; 2 = borderline mentally ill; 3 = mildly ill; 4 = moderately ill; 5 = markedly ill; 6 = severely ill and 7 = among the most extremely ill patients. Decreased scores over time represent improvement.
The EQ-5D-5L is a standardised, patient-reported, generic instrument for measuring health outcome. It provides a simple descriptive profile and a single index value for health status. The instrument consists of the EQ-5D-5L descriptive system and the EuroQol (EQ) visual analogue scale (VAS). The descriptive system consists of 5 dimensions (mobility, self-care, usual activities, pain/discomfort and anxiety/depression), with 5 levels of severity within each particular EuroQol dimension (EQ-5D). The EQ VAS records the respondent’s self-rated health on a 20-cm vertical, VAS with end points labelled “the best health you can imagine” and “the worst health you can imagine”. This information can be used as a quantitative measure of health as judged by the individual respondents. The index score was calculated using the EQ-5D-5L United States of America (US) value set that was published by the EuroQol group. The maximum value is 1 and the minimum value is − 0.109, with scores anchored at 0 for health states equivalent to death. Higher scores represent better health states. The EQ-5D has been positively evaluated for construct validity and appropriateness for use in studies involving subjects with schizophrenia.
The SWN-S scale is a 20-item (10 positive and 10 negative), self-report instrument constructed to evaluate “subjective well-being under neuroleptics”. The short form SWN-S was constructed based on an item analysis of 212 patients with schizophrenia who were medicated with antipsychotics. The scale is reported to show good internal consistency and construct validity in patients with schizophrenia. Each item is scored on a Likert scale with 6 response categories (ranging from “not at all” to “very much”). Each item is scored 1 to 6, with a minimum total score of 20 (indicating low subjective well-being) and a maximum total score of 120 (indicating good subjective well-being). The SWN-S has 5 subscales: mental functioning, self-control, emotional regulation, physical functioning and social integration. Since items 1, 4, 6, 9, 10, 11, 12, 14, 16 and 17 are negatively stated items on the questionnaire, they were reversed for analysis as follows: 6 = 1, 5 = 2, 4 = 3, 3 = 4, 2 = 5 and 1 = 6.
The MSQ is a single-item questionnaire that evaluates satisfaction with antipsychotic medication in schizophrenia patients. The single item is scored from 1 being “Extremely dissatisfied” to 7 being “Extremely satisfied”. The levels in the MSQ are as follows: 1 = Extremely dissatisfied; 2 = Very dissatisfied; 3 = Somewhat dissatisfied; 4 = Neither satisfied nor dissatisfied; 5 = Somewhat satisfied; 6 = Very satisfied; 7 = Extremely satisfied.
Statistical Analysis
Primary Outcome of the Study
Power Calculation
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Statistical Test or Model
Primary Efficacy Analysis: The primary efficacy analysis was performed using a mixed-effects model for repeated measures (MMRM), with change from baseline in total PANSS score as the dependent variable, considering all available observations of total PANSS score at various visits. The MMRM model included terms for treatment (RBP-7000 [90 mg], RBP-7000[120 mg], placebo), baseline total PANSS score, visit (5, 6, 8, and 9), and treatment-by-visit interaction as fixed effects. The unstructured covariance matrix was used to model the within-subject variance-covariance matrix. Based on the multivariate response variable across visits, the least squares means (LSMs) and its corresponding standard error (SE) were presented for each treatment group, as well as the treatment difference LSMs, 95% confidence limits of the treatment difference, and P values. Comparison of each dose group with placebo was performed using Dunnett’s procedure for controlling type I error at 2.5% (1-sided), and dose groups that were significantly superior to placebo were identified. Superiority of risperidone ER over placebo was demonstrated if 1 or both of the 1-sided P values for testing H01 or H02 satisfied P value < 0.0135.
Subgroup Analyses
No subgroup analysis was performed.
Sensitivity Analyses
A sensitivity analysis was conducted to investigate the impact of missing data assumptions. The analysis consisted of 2 separate components, a pattern mixture model and a multiple-imputation approach.
||||||||||||||||||||||||||||||||||||||||||||||||||
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Secondary Outcomes of the Study
The key secondary efficacy analysis was performed for the ITT population. The analysis of the secondary efficacy variable was conducted using the same model as the primary efficacy variable with CGI-S scores in place of PANSS scores in the primary efficacy analysis. At each visit, LSM and LSM SE for each treatment group were presented, as well as the treatment difference LS mean, 95% CIs of the treatment difference and P values.
Safety Analyses
Safety parameters were summarized descriptively; no statistical comparisons across the treatment groups were performed.
Analysis Populations
Intent-to-Treat population: The ITT population contained all randomized subjects who received at least 1 dose of risperidone ER or placebo, and had data recorded for at least 1 postbaseline PANSS total score such that the change from baseline could be calculated. Subjects were classified according to their randomized treatment.
Per-protocol population: The PP population contained all subjects in the ITT population who did not experience any major protocol violations. A major protocol violation was defined as a protocol violation that could have affected the integrity of the data as determined by the sponsor. Protocol violations were identified and documented through programmatic checks of study data, as well as through review of selected subject data listings before database lock. Subjects were classified according to randomized treatment.
Safety population: The Safety population contained all randomized subjects who received at least 1 dose of risperidone ER or placebo during the double-blind treatment period. Subjects were classified according to treatment received. If there was any doubt whether a subject was treated or not, it was assumed they were treated for the purpose of analysis.
Sponsor’s Summary of the Results
Patient Disposition
Disposition | Risperidone ER 90 mg | Risperidone ER 120 mg | Placebo |
|---|---|---|---|
Screened, N | 538 | ||
Randomized, N | 116 | 119 | 119 |
Discontinued, n (%) | 26 (22.4) | 34 (28.6) | 35 (29.4) |
Reason for discontinuation, n (%) | |||
Withdrew Consent, | 20 (17.2) | 25 (21.0) | 21 (17.6) |
Withdrawn by investigator, | 3 (2.6) | 4 (3.4) | 5 (4.2) |
Insufficient clinical response, | 2 (1.7) | 0 | 4 (3.4) |
Adverse Event, | 0 | 2(1.7) | 3 (2.5) |
Protocol deviation | 0 | 3 (2.5) | 1 (0.8) |
Lost to follow-up | 1 (0.9) | 0 | 1 (0.8) |
ITT, N (%) | 111(95.7) | 114 (95.8) | 112 (94.1) |
PP, N (%) | 96 (82.8) | 96 (80.7) | 99 (83.2) |
Safety, N (%) | 115(99.1) | 117 (98.3) | 118 (99.2) |
ER = extended release; ITT = intention to treat, PP = per protocol.
A total of 538 patients were screened for study participation at 33 centres in the US, of which 354 subjects were randomly assigned to treatment. Four subjects were randomized incorrectly; these subjects were withdrawn from the study and did not receive study drug. Including these 4 subjects, there were 188 subjects who did not meet the entry criteria. Most subjects completed this study (73.2%). Withdrawal of consent was the most common reason for study discontinuation (18.6%) and occurred in similar proportions for each treatment group (Table 7). There were no meaningful differences across the treatment groups in the proportions of subjects who were withdrawn at each visit.
There were no patients whose treatment was prematurely unblinded before database lock. The most common protocol deviations leading to exclusion from the per-protocol population were the use of prohibited medications and the randomization of subjects who did not meet the inclusion/exclusion criteria. The protocol deviations that occurred during this study were not believed to have impacted the conclusions of the study results. The primary efficacy analysis was repeated using the PP population.
Exposure to Study Treatments
Study Treatments
Overall, 84.6% of subjects received both injections during the double-blind treatment period and were, therefore, considered treatment compliant. It should be noted that risperidone ER was administered by a health care practitioner in the inpatient facility. Thus, unless a subject was discontinued from the study at a dosing visit, the scheduled dose was administered. The proportions of subjects considered compliant with treatment in the risperidone ER 90 mg (87.8%), 120 mg (82.1%) and placebo (83.9%) group were similar.
Concomitant Medications
Most subjects in all treatment groups reported using drug therapy (for any indication) before and after randomization, and there were no clinically relevant differences across the treatment groups with respect to the use of any prior or concomitant medications. Concomitant medications were defined as those taken any time during study drug administration (Days 1 to 57). Per protocol, subjects were not to receive supplemental antipsychotics during the risperidone ER treatment period. The use of nervous system medications was prevalent, but there were no marked differences across the groups. The most frequently reported (> 5% in any treatment group) nervous system medications are summarized in Table 8.
Table 8: Summary of the Most Frequently Reported Concomitant Nervous System Medication ||||||||||||||||||||||||||||||||||||||||||
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| |
|---|---|---|
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| |
Table 8 was removed upon request from the sponsor because it contained confidential information.
Efficacy
Positive and Negative Syndrome Scale Total Score (PANSS Score)
Superiority of both risperidone ER (RBP-7000) 90 mg and 120 mg over placebo was demonstrated based on the MMRM analysis of change from baseline in PANSS total scores for the ITT Population (LSM: − 6.148; 95% CI, − 9.982 to − 2.314; P value: 0.0004 and LSM: − 7.237; 95% CI, − 11.045 to − 3.429; P value: < 0.0001 for the 90 and 120 mg groups, respectively) (Figure 2).
Figure 2: PANSS Total Score Change From Baseline Repeated Measures End Point for Visits 5, 6, 8, and 9 (ITT Population)
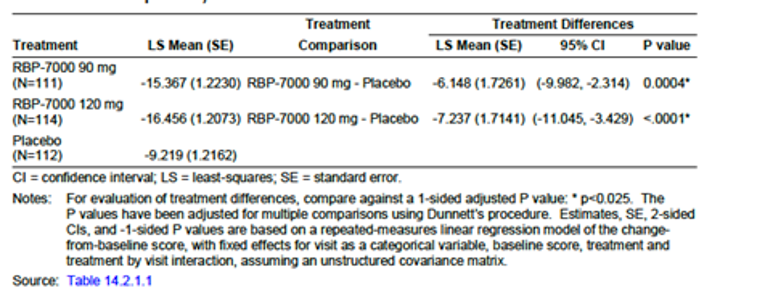
Source: CSR.23
The arithmetic mean PANSS total scores at baseline for the ITT population were similar for each treatment group (95.5, 94.9, and 94.1 points for the 90 mg, 120 mg, and placebo groups, respectively). The arithmetic mean PANSS total scores at baseline for the Per-Protocol Population were also similar for each treatment group. Sensitivity analysis results were consistent with the primary analysis. The MMRM assumption of missing at random (MAR) was tested and was found not to have been violated.
The PANSS measurements demonstrate efficacy as early as 2 weeks after dosing (first measurement of the study, see Figure 3).
Figure 3: Change From Baseline in PANSS Total Score by Visit and Dose Group — MMRM Analysis (ITT Population)
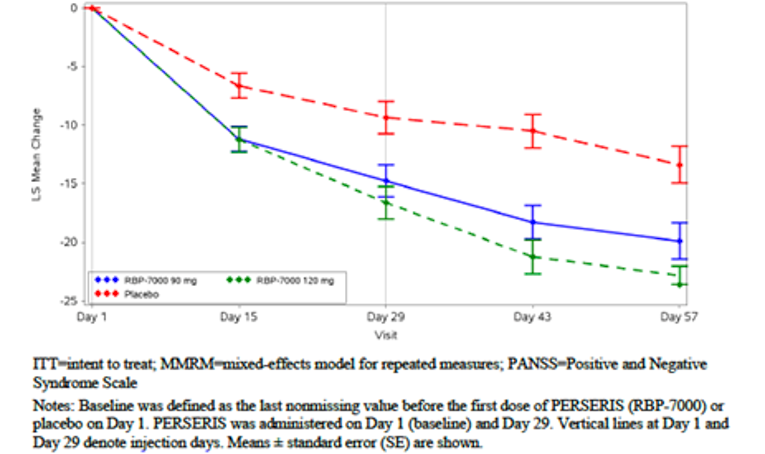
Source: Sponsor’s submission.24
Clinical Global Impression–Severity of Illness (CGI-S) Score
Superiority of both risperidone ER (RBP-7000) 90 mg and 120 mg over placebo was demonstrated based on the MMRM analysis of change from baseline in CGI-S scores for the ITT Population (LSM: − 0.350; 95% CI, − 0.557 to − 0.143; P value: 0.0002 and LSM: − 0.396; 95% CI, − 0.602 to − 0.190; P value: < 0.0001 for the 90 mg and 120 mg groups, respectively) (See Figure 4).
Figure 4: CGI-S Score Change From Baseline Repeated Measures End Point for Visits 5, 6, 8, and 9 (ITT Population)
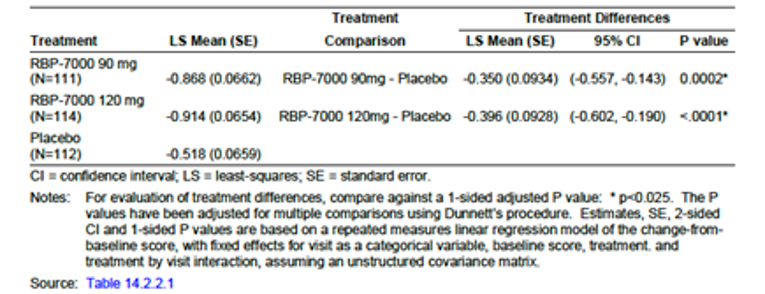
Source: CSR.23
Additional information on EQ-5D-5L, SWN-S and MSQ scales is presented in Appendix 4.
Study 13 to 0005, which provides evidence up to 52 weeks is presented in Appendix 5.
Harms
Safety Evaluation Plan
Safety parameters included adverse events (AE), serious adverse events (SAEs), discontinuations from study due to AEs (WDAEs), local injection-site tolerability (i.e., injection-site reactions), subject reported injection-site pain, concomitant medications, changes in clinical laboratory results, vital sign measurements, 12-lead electrocardiograms, physical examination results, body weight, height, body mass index (BMI) and abdominal fat measurement (waist-to-hip ratio). Extrapyramidal symptoms (EPS) were assessed using the following scales: the Abnormal Involuntary Movement Scale (AIMS), the Simpson-Angus Scale (SAS) and the Barnes Akathisia Rating Scale (BARS); note that this was sometimes abbreviated “BAS” in study reports. Suicidality was assessed using the Columbia-Suicide Severity Rating Scale(C-SSRS).
Overview of Safety
The proportion of subjects with 1 or more treatment-emergent adverse events (TEAEs) was higher for the risperidone ER 120 mg group (77.8%) compared with the risperidone ER 90 mg (70.4%) and placebo groups (68.6%). The incidence of TEAEs that were reported as related to study drug was higher for the 120 mg (55.6%) and 90 mg (50.4%) groups compared with the placebo (42.4%) group. No subject died during the study. Serious adverse events (SAEs) were reported for 1 subject each in the risperidone ER (RBP-7000) 120 mg (chest pain) and placebo (dyspepsia) treatment groups and no subjects in the risperidone ER (RBP-7000) 90 mg group, neither of which were reported as related to study drug. Five subjects were discontinued from the study as the result of AEs, including 2 subjects (1.7%) in the risperidone ER 120-mg group (groin pain and paranoia) and 3 subjects (2.5%) in the placebo group (psychotic disorder in 2 subjects and haematuria in 1 subject) (Figure 5).
Treatment with risperidone ER (Perseris, 90 mg and 120 mg) over an 8-week treatment period was generally well tolerated in adult subjects with acute schizophrenia. The most frequently reported treatment-emergent AEs (TEAEs) occurring at higher rates in the risperidone ER treatment groups compared with the placebo group were weight gain and somnolence. The most frequently reported TEAEs assessed as related to risperidone ER treatment included weight gain and akathisia.
Laboratory data showed a possible association of risperidone ER treatment with elevated prolactin based on higher mean changes from baseline and higher incidences of markedly abnormal postbaseline laboratory values in the risperidone ER treatment groups compared with the placebo group. This was more pronounced in female subjects than male subjects and in the risperidone ER 120 mg treatment group. No other relevant treatment group differences were noted for any laboratory parameter.
Weight data showed a possible association of risperidone ER treatment with weight gain based on higher mean changes from baseline and higher incidence of markedly abnormal weight gain in the risperidone ER treatment groups compared with the placebo group.
Analyses of vital sign, ECG, BARS, AIMS, SAS, and C-SSRS assessments did not reveal any clinically relevant effect of risperidone ER treatment. There were no trends in vital signs, ECGs, or clinical laboratory evaluations that were considered clinically significant.
The incidence of TEAEs occurring in ≥ 5% of subjects is summarized in Figure 6. There were no clinically relevant differences across the treatment groups in the proportions of subjects with TEAEs for the most frequently reported (> 20% of subjects in either risperidone ER group system organ classes, including: gastrointestinal disorders (29.6%, 26.5%, and 29.7% for the 90 mg, 120 mg, and placebo groups, respectively), general disorders and administration site conditions (27.0%, 31.6%, and 35.6%, respectively), and nervous system disorder (29.6%, 27.4%, and 31.4%, respectively). Akathisia occurred in a higher proportion of subjects in the risperidone ER 120 mg group (6.8%) compared with the risperidone ER 90 mg (2.6%) and placebo (4.2%) groups.
The proportions of subjects with weight increased and somnolence were similar between the risperidone ER (90 and 120 mg) groups, but higher compared with the placebo group. Otherwise, there were no clinically relevant differences across the treatment groups in the proportions of subjects with the most frequently reported TEAEs.
Figure 6: Summary of Subjects With Treatment-Emergent Adverse Events Occurring in at Least 5% of Subjects (Safety Population)
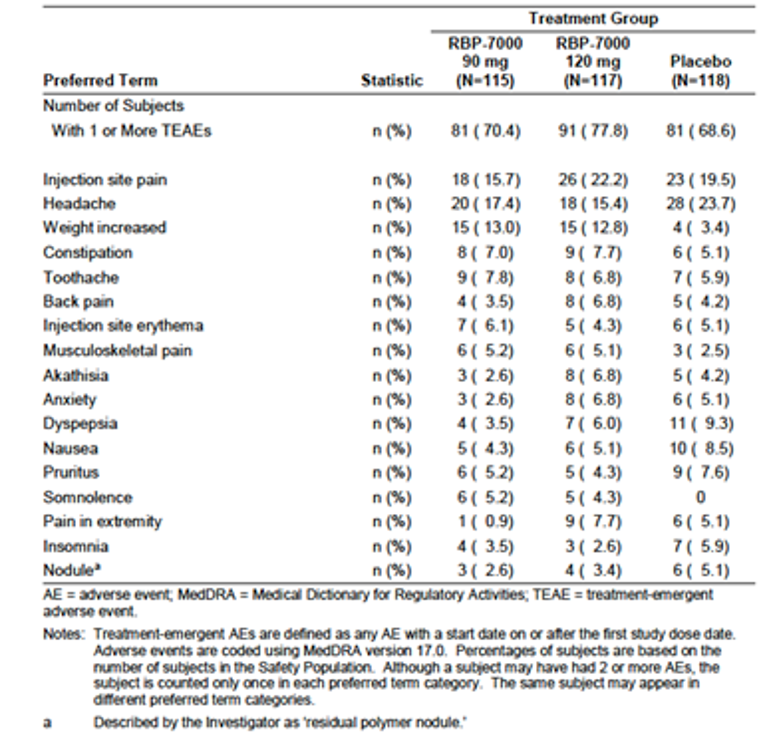
Source: CSR.23
Serious Adverse Events
Most TEAEs were mild or moderate in severity. There were 2 subjects who experienced SAEs during study conduct, including mild chest pain in 1 subject in the risperidone ER (RBP-7000) 120 mg group and moderate dyspepsia in 1 subject in the placebo group. Both of these SAEs were assessed by the investigator as not related to study drug.
Withdrawal Due to Adverse Events
There were 5 subjects with TEAEs leading to study discontinuation, including 2 (1.7%) subjects in the risperidone ER (RBP-7000) 120 mg group (groin pain and paranoia) and 3 (2.5%) subjects in the placebo group (psychotic disorder in 2 subjects and hematuria) (Figure 7).
Figure 7: Summary of Subjects With TEAEs Leading to Discontinuation From the Study (Safety Population)
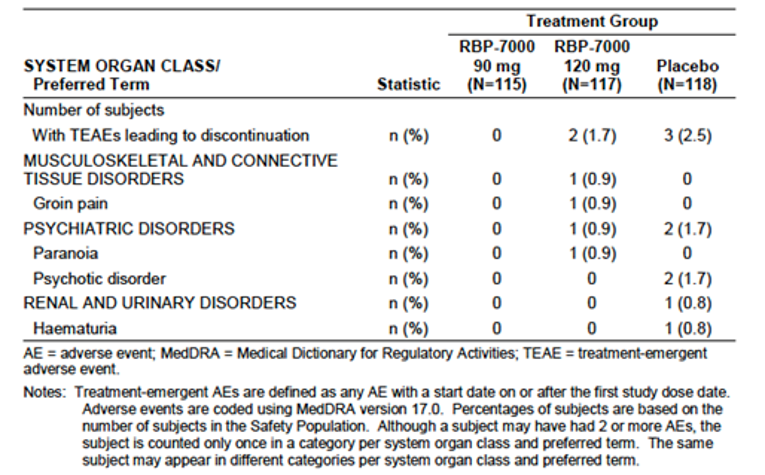
Source: CSR.23
Adverse Events of Special Interest
The majority of subjects did not have injection-site reactions (pain, tenderness, erythema/redness, or induration/swelling) immediately after dosing on either Day 1 or Day 29. Of the injection-site reactions observed, most were graded of mild intensity and there were no meaningful differences across the treatment groups in the proportions of subjects with injection-site reactions. In general, there were no meaningful differences in the pattern of injection-site reactions following the first injection compared with the second injection. Injection-site pain resolved by 12 hours post-dose for approximately half of the subjects in all treatment groups following both injections. For approximately half of the subjects with injection-site tenderness, symptoms persisted until about Day 5 for the risperidone ER (RBP-7000) groups and about Day 2 for the placebo group following the injection on Day 1 and until about Day 6 for all treatment groups following the injection on Day 29. Injection-site erythema occurred in few subjects and persisted until about Days 4 or 5 for all treatment groups following both injections. Injection-site induration/swelling also occurred in few subjects but appeared to persist throughout the observation period (7 days) and occurred in fewer subjects following the second injection on Day 29.
The mean subject-reported injection-site pain VAS scores were similar for all treatment groups following both injections. Mean VAS scores at 5 minutes post-dose were approximately 50% lower than those at 1-minute post-dose; mean scores were similar at 30 minutes and 60 minutes post-dose and were approximately 80% lower than those at 1-minute post-dose.
The proportions of subjects with injection-site stinging/burning were similar for the risperidone ER (RBP-7000) 90 mg and 120 mg groups at all time points following both injections and were generally lower compared with the placebo group.
CADTH’s Critical Appraisal of the Clinical Evidence
CADTH conducted a critical appraisal of the clinical study for risperidone for ER injectable suspension (risperidone ER) based on the summary of the evidence provided by the sponsor.
Internal Validity
Based on information available in the summary of evidence submitted by the sponsor, the study design demonstrated a number of methodological strengths. Study 09 to 001023 was a prospective, multi-centre, double-blind, randomized, parallel, placebo-controlled trial. Appropriate randomization and allocation concealment procedures (i.e., randomization of patients was performed using an interactive web response system)25 were clearly described. Comparison of each dose group with placebo was performed using a Dunnett procedure for controlling type I error at 2.5% (1-sided) for PANSS total score and CGI-S.23,25 However, several potential limitations are discussed subsequently.
The study was conducted at 33 study sites in the US. Randomization to treatment arm was not stratified by study centre. However, the study was conducted only in the US; no significant clinical practice discrepancy was expected between the 33 study sites in the US.
The proportion of patients who discontinued from the trials was relatively high (i.e., 22.4% to 29.4% across the treatment groups). Although the discontinuation rates were similarly high between the groups and not differential, it could result in an imbalance in characteristics as the trial progressed, and efficacy data at 8 weeks were not available for a large number of patients. The primary efficacy analysis was performed using an MMRM, with change from baseline in total PANSS score as the dependent variable, considering all available observations of total PANSS score at various visits. The MMRM model included terms for treatment (risperidone ER 90 mg, risperidone ER 120 mg, placebo), baseline total PANSS score, visit (5, 6, 8, and 9), and treatment-by-visit interaction as fixed effects. The unstructured covariance matrix was used to model the within-subject variance-covariance matrix. The MMRM analysis that was used assumes the data are MAR, which is often not the case in clinical trials. Although they did a pattern analysis that suggests the MAR assumption was not violated and which supported the primary efficacy analysis,23 this approach also makes several assumptions to estimate the factors in the dropout pattern, which are unclear. Moreover, although a multiple-imputation approach was also used, this is only valid if the data missing from the dropouts were truly MAR. As a result, it is unclear what impact the missing data may have had on the efficacy results observed.
The ITT population was defined as all randomized patients who received at least 1 dose of the study drug or placebo and who had at least 1 post-baseline PANSS total score, which was technically not a true ITT analysis but, rather, a modified ITT population. The ITT population included 96% of the randomized patients in both risperidone ER dose groups and 94% of the randomized patients in the placebo group; therefore, it is unclear if any bias would have been introduced in the efficacy analysis.
A total of 15.4% of patients were considered not compliant with the study drug (i.e., 84.6% of patients were compliant with the study drug) and, therefore, it is possible that the observed effects for the treatment arms may be an underestimation of the efficacy of the therapy, as not all were using the medication as intended. Overall, the results of the per-protocol population analysis were consistent with the results of the ITT population analysis.23 According to the study protocol, the use of oral risperidone was not allowed during the 8-week trial. However, it was noticed that the percentage of patients who used oral risperidone during the trial was higher in the risperidone ER groups than in the placebo group (11.3%, 8.5%, and 6.8% in the risperidone ER 90 mg, risperidone ER 120 mg, and placebo groups, respectively) (Table 8). Although the difference between treatment groups with respect to using oral risperidone was small, the higher use of oral risperidone in the treatments arms would bias the results in favour of the risperidone ER groups; however, the impact on the efficacy results cannot be fully determined.
The change from baseline in the PANSS total score at week 8 was 15.4, 16.5, and 9.2 in the risperidone ER 90 mg and 120 mg groups and placebo group, respectively. It was suggested that an absolute reduction of 15 points in the total PANSS score corresponds to “minimally improved” on the CGI-Improvement score, and a reduction in the CGI–Severity of Illness score by 1 severity step.40 However, these clinical improvement estimates were sensitive to baseline severity of illness to the extent that participants with a lower baseline severity of illness required smaller reductions in the PANSS to produce a particular improvement in the CGI. For this reason, it has been suggested that change in PANSS score has limited usefulness as a primary outcome, due to variability in baseline symptom intensity.41,42 Although change in PANSS score appeared to meet the minimal important improvement (i.e., a reduction of 15 points in PANSS score) in the risperidone groups, patients in the placebo group also demonstrated a reduction in the PANSS and CGI-S during the 8 weeks, which could reflect a potential placebo or supportive care response, or natural fluctuations of the disease. Therefore, it is uncertain whether the differences between treatment groups were clinically meaningful (risperidone ER minus placebo: LSM of −6.148 and −7.237 for the 90 mg group compared with the placebo group and the 120 mg group compared with the placebo group, respectively).
In terms of CGI-S, neither the change from baseline for each risperidone ER treatment group (0.87 and 0.92 in the risperidone ER 90 mg and 120 mg groups, respectively), nor the treatment-group difference between either the risperidone ER group and the placebo group (0.35 and 0.40 in risperidone 90 mg compared with placebo and risperidone ER 120 mg compared with placebo, respectively), met the minimal important difference (i.e., a 1-point reduction in the CGI-S). Therefore, the clinical significance of the observed findings in CGI-S is unclear.
One of the goals of the treatment of schizophrenia is to maintain or restore HRQoL. The EQ-5D-5L, SWN-S, and MSQ were assessed in the pivotal study. However, these HRQoL and patient satisfaction outcomes were designed as tertiary outcomes and based on power calculations for the primary end point (total PANSS). Moreover, these end points were not controlled for multiplicity and are at risk of a type I error and should be viewed as supportive evidence only for the overall effect of risperidone ER in patients with schizophrenia. Therefore, the validity of the findings of HRQoL outcomes remains uncertain.
In addition, more patients in the risperidone ER groups than in the placebo groups reported weight gain (13.0%, 12.8%, and 3.4% in the risperidone ER 90 mg and 120 mg groups and placebo group, respectively) and somnolence (5.2%, 4.3%, and 0% in the risperidone ER 90 mg and 120 mg groups and placebo group, respectively). This could have unblinded the patients and resulted in bias in the PANSS or CGI scores, which are subjective in nature, as patients may have reported differently knowing they were on the drug. Also, it likely contributed to the losses to follow-up, which have been discussed previously.
External Validity
The study was conducted in a distinct study population: the patients enrolled in the study were adults younger than 55 years of age and the majority were male and Black. This study population does not fully represent the characteristics of Canadian patients with schizophrenia. The doses of risperidone ER were aligned with the approved doses in Canada; the clinical expert indicated that dosing in certain subpopulations, such as Asian or Indigenous Canadians, could raise questions. In addition, the study was conducted in an inpatient population, which is not reflective of the anticipated use of risperidone ER in Canada, which will largely be administered to outpatients. However, the clinical expert consulted for this review indicated there should not be a major concern on the overall generalizability of the findings in the pivotal study to a Canadian clinical practice setting, given the extent of knowledge of risperidone (oral and LAI) and other LAIs. The Health Canada reviewer report also indicated that the efficacy of risperidone ER is unlikely affected by age.43
One of the inclusion criteria for Study 09 to 0010 was a PANSS total score of between 80 and 120; this may not reflect the broad range of symptom severity in practice. The sponsor indicated that the reason for setting the criteria for the PANSS total score at greater than 80 was to avoid flooring effects, that is, when the level of symptoms is too modest to allow detection of a therapeutic response. The reason for setting the criteria for the PANSS total score to less than 120 was to avoid a marked regression to the mean phenomenon, which may negatively affect response detection. It is not clear if the findings can be generalized to those patients with a PANSS score of less than 80 or greater than 120. The clinical expert indicated that patients with a PANSS score greater than 120 are rare in routine clinical settings. Those with a score of less than 80, however, ought not to differ greatly. The most relevant PANSS subscale is positive; the negative symptoms, if contributing to an elevated total PANSS total score, may not be as relevant, as they respond less well to antipsychotic drugs.
The duration of the double-blind randomized controlled trial was relatively short (i.e., 8 weeks). The CADTH clinical expert consulted for this review considered 8 weeks to be an adequate duration to demonstrate a treatment effect in clinical trials for acute exacerbation of schizophrenia. However, risperidone ER is intended to be used to treat a chronic condition (schizophrenia); considering the irregular and unpredictable fluctuation of the symptoms of the occurrence of schizophrenia, a longer duration (e.g., 1 year) of risperidone ER treatment would provide stronger evidence of long-term, clinically important treatment effects in this context. A 52-week, multi-centre, phase II, open-label, single-arm study on risperidone ER 120 mg was conducted in adults with schizophrenia,44,45 which was included in the sponsor’s submission.24 Briefly, a total of 500 patients was included in the open-label study. The primary objective of the open-label study was to evaluate the long-term safety and tolerability of risperidone ER in adults with schizophrenia.44,45 However, only 92 patients (18%)44,45 were from the pivotal study.23 A total of 408 patients (82%) were de novo participants with stable schizophrenia. Furthermore, due to the limitations resulting from its open-label and single-arm design, the findings of the extension should be interpreted with caution. The CADTH clinical expert indicated that while further studies with high-quality designs are needed to actually assess the long-term efficacy (e.g., relapse, remission, hospitalization, and so forth) and safety of risperidone ER, risperidone has been available in the market for a long time (oral tablets since 199336 and LAI drugs since 2004),37 and its long-term efficacy and safety profile has been well established; therefore, concerns regarding the long-term efficacy and safety of risperidone ER would be minimal.
Study 09 to 0010 was a placebo-controlled study; no direct or indirect treatment comparison evidence was included in the sponsor’s submission that compared risperidone ER with oral risperidone, risperidone LAI (IM every 2 weeks), and other relevant AAP LAIs currently marketed in Canada.
Economic Review
The current review is for risperidone ER (Perseris) for the treatment of schizophrenia in adults.
Economic Information
Summary of Sponsor’s Economic Information
The sponsor submitted a cost comparison for the annual drug acquisition costs associated with risperidone monthly injections (risperidone ER [Perseris]) compared with risperidone injections every 2 weeks (risperidone LAI [Risperdal Consta]), risperidone tablets (generic), and other non-risperidone LAI AAPs for the treatment of schizophrenia in adults (Table 9).
The sponsor’s analysis reports that risperidone ER has an annual cost of $5,474 to $7,299 per patient per year which, when considering similar dosage strengths, is substantially more than the annual cost of treatment with risperidone tablets, less than the annual cost of risperidone injections every 2 weeks, and priced similarly to aripiprazole and paliperidone LAIs.
Table 9: Sponsor’s Submitted Drug Acquisition Cost Comparison
Generic name (brand name) | Strength | Dosage form | Price ($) | Recommended dosage regimen | Annual drug cost ($) | Difference in annual cost ($) |
|---|---|---|---|---|---|---|
Perseris | 90 mg | Extended-release injectable suspension | 456.1800 | 90 mg or 120 mg monthly | 5,474.1600 | Reference |
120 mg | 608.2200 | 7,298.6400 | Reference | |||
Comparators | ||||||
Risperdal Consta (risperidone) | 12.5 mg | Prolonged-release injectable suspension | 75.4100 | 25 mg to 50 mg every 2 weeks | — | — |
25 mg | 156.0900 | 4,058.3400 | 1,415.82 | |||
37.5 mg | 234.1600 | 6,088.1600 | −614.00 | |||
50 mg | 312.2000 | 8,117.2000 | −818.56 | |||
Risperidone | 0.25 mg | Tablets | 0.1036 | 4 mg to 16 mga per day | 349.4510 to 1,397.8040a | 5,124.71 to 5,900.84a |
0.5 mg | 0.1735 | |||||
1 mg | 0.2397 | |||||
2 mg | 0.4795 | |||||
3 mg | 0.7180 | |||||
4 mg | 0.9574 | |||||
Other non-risperidone atypical antipsychotic LAI for the treatment of schizophrenia | ||||||
Abilify Maintena (aripiprazole) | 300 mg | Prolonged-release injectable suspension | 456.1800 | 300 mg or 400 mg monthly | 5,474.1600 | 0 |
400 mg | 456.1800 | 5,474.1600 | 0 | |||
Invega Sustenna (paliperidone) | 50 mg | Prolonged-release injectable suspension | 304.1000 | 50 mg to 150 mg monthly | 3,649.2000 | 1,824.96b |
75 mg | 456.1800 | 5,474.1600 | 0 | |||
100 mg | 456.1800 | 5,474.1600 | 0 | |||
150 mg | 608.2200 | 7,298.6400 | 0 | |||
Invega Trinza (paliperidone) | 175 mg | Prolonged-release injectable suspension | 912.3000 | 175 mg to 525 mg every 3 months | 3,649.2000 | 1,824.96b |
263 mg | 1,368.5400 | 5,474.1600 | 0 | |||
350 mg | 1,368.5400 | 5,474.1600 | 0 | |||
525 mg | 1,824.6600 | 7,298.6400 | 0 | |||
LAI = long-acting injectable.
Note: Adapted from sponsor’s tailored review submission (Table 10).46
Pricing source: Ontario Drug Benefit Formulary/Comparative Drug Index.
Recommended dosage regime source: Approved product monographs.
aIt was unclear whether the sponsor intended to include a dose range of 4 mg to 6 mg for risperidone tablets, as stated in the “recommended dosage regimen” column of Table 10 of its submission, or 4 mg to 16 mg, as calculated in the “annual drug cost” column of the same table. According to the product monograph,38 16 mg is the highest daily dose at which safety has been assessed, while 4 mg to 6 mg daily is recommended as the target dose range.
bCorrected from $1,764.96 in sponsor’s submission.
Additionally, the sponsor considered the cost of risperidone ER compared with risperidone LAI (every 2 weeks) when the cost of administering the injections is included (Table 10). When monthly risperidone ER is compared with risperidone LAI every 2 weeks, there may be additional savings due to differing annual injection frequency (i.e., 12 versus 26 injections).
Table 10: Summary of Comparative Treatment Costs
Generic name (brand name) | Cost per administration | Annual cost of administration | Difference in drug acquisition costs per year | Difference in total health care costs per year | Difference in total costs per year |
|---|---|---|---|---|---|
Risperidone ER (Perseris) | $6.75a | $81.00 | At usual dose: −$614.00 At maximum dose: −$818.56 | −$94.98 | At usual dose: −$708.98 At maximum dose: −$913.54 |
Risperidone LAI (Risperdal Consta) | $175.98b |
ER = extended release; LAI = long-acting injectable.
Note: Adapted from Tables 13 and 14 of the sponsor’s tailored review submission,46 which compare the cost of 90 mg and 120 mg monthly risperidone ER with 37.5 mg and 50 mg risperidone LAI every 2 weeks, respectively. The cost of 25 mg risperidone LAI every 2 weeks was not included in this comparison by the sponsor.
aOntario Schedule of Benefits for Physician Services, Code G373, intramuscular injection, sole reason for visit.47
bAnnual cost of administration every 2 weeks was corrected by CADTH to avoid rounding to a 364-day year. Cost reported by the sponsor was $175.50 annually.
CADTH Appraisal of the Sponsor’s Economic Information
CADTH identified several key limitations of the sponsor’s analysis that have notable implications on the economic analysis:
Assumption of similar clinical efficacy: The sponsor’s cost comparison assumes there is similar clinical efficacy between risperidone ER and its comparators. The clinical trial (Study 09 to 0010)25 compared risperidone ER with placebo and no indirect evidence was submitted comparing risperidone ER with other LAI AAPs. Therefore, given the lack of evidence regarding clinical similarity, the appropriateness of conducting a cost comparison is unknown.
CADTH was unable to address this limitation in reanalysis.
Assumption of equivalent doses across comparators: The sponsor compared the cost of 90 mg and 120 mg monthly risperidone with comparators at doses that are most closely aligned with the cost of each risperidone ER dose. For example, the sponsor compared the annual cost of 120 mg monthly risperidone only with the cost of 150 mg monthly paliperidone, rather than with the other available doses. In doing so, the sponsor implicitly assumes these doses are equally effective without providing evidence for that assumption.
CADTH compared both the cost of 90 mg and 120 mg risperidone ER with each comparator dose, and then conducted a price-reduction analysis to determine the amount the submitted price of risperidone ER would need to be reduced for its lowest and highest recommended doses to be equivalent in cost to the least expensive comparator at the lowest and highest recommended doses.
Confidential pricing agreements: While the submitted price for risperidone injections every 2 weeks is similar to the publicly available list prices of other LAI AAPs, as indicated in the drug program input submitted for this review (see Stakeholder Perspectives section), these list prices are higher than the price paid by jurisdictional drug plans due to confidential pricing agreements. Therefore, the submitted price of risperidone ER likely requires a price reduction to avoid incurring additional costs relative to its comparators.
CADTH was unable to address this limitation in reanalyses, as the negotiated prices of comparators are unknown.
Updated prices available: Ontario Drug Benefit Formulary list prices have changed for risperidone LAI and both paliperidone LAIs since the sponsor conducted its analysis.
CADTH applied updated Ontario Drug Benefit Formulary list prices (accessed April 2021) in reanalyses.
Daily dose of risperidone tablets: It is unclear whether the sponsor’s calculation of the annual cost of risperidone tablets, which used a range of 4 mg to 16 mg daily, was intentional or an error. According to the product monograph,38 while the safety of doses up to 16 mg daily has been established, doses above 10 mg have not been shown to be more effective than lower doses and are associated with more extrapyramidal symptoms and other AEs. The recommended target dose range is 4 mg to 6 mg daily.
CADTH used a recommended daily dose of 4 mg to 6 mg for risperidone tablets in reanalyses.
CADTH Reanalyses of the Economic Information
The CADTH reanalysis was derived by making changes in values and assumptions in consultation with clinical experts. The changes made included updating Ontario Drug Benefit list prices, correcting the recommended daily dose of risperidone tablets to 4 mg to 6 mg, and calculating the incremental and relative cost of each dose of comparators to each dose of risperidone ER rather than assuming equivalent doses.
At the submitted price, the annual cost of risperidone ER ($5,474 to $7,299 per patient per year) is more expensive than risperidone tablets ($349 to $524 per patient annually). However, it is within the range of other LAI AAPs ($3,815 to $8,877 per patient annually) at publicly available list prices, although less expensive doses of some LAI comparators exist ($3,815 per patient annually for the lowest recommended dose of both paliperidone LAIs and $4,439 per patient annually for the lowest recommended dose of risperidone LAI) (Table 11).
When compared with risperidone LAI administered every 2 weeks, as described by the sponsor, the use of monthly injections of risperidone ER is associated with savings of approximately 14 administrations per year (a total of 26.07 injections administered every 2 weeks compared with 12 monthly doses). The amount of savings associated with less frequent injections will depend on the jurisdictional cost per injection and whether the injection is administered as part of a consultation appointment or as the sole reason for the visit. Using Ontario as an example, and assuming the injections are the sole reason for visits,47 this reduced frequency of injection leads to an average savings of $94.98 per patient per year (Table 10).
Table 11: CADTH Cost Comparison Table — New Formulation of Existing Drug
Drug or comparator | Strength or concentration | Dosage form | Price ($) | Recommended maintenance dose | Average annual drug cost ($) | Difference in annual drug costs ($) (%) compared with 90 mg risperidone | Difference in annual drug costs ($) (%) compared with 120 mg risperidone |
|---|---|---|---|---|---|---|---|
New formulation | |||||||
Risperidone (Perseris) | 150 mg/mL | 90 mg 120 mg powder for injection | 456.1800a 608.2200a | 90 mg or 120 mg monthly | 5,474 7,299 | Reference | Reference |
Reference formulations | |||||||
Risperidone (Risperdal Consta) | 6.25 mg/mL 12.5 mg/mL 18.75 mg/mL 25 mg/mL | 12.5 mg 25 mg 37.5 mg 50 mg powder for injection | 84.4600b 170.2600 255.3700 340.5000 | 25 mg to 50 mg | 4,439 6,658 8,877 | −1,035 (−18.9%) 1,184 (21.6%) 3,403 (62%) | −2,860 (−39%) −641 (−9%) 1,579 (22%) |
Risperidone (generics) | 0.25 mg 0.5 mg 1 mg 2 mg 3 mg 4 mg | Tablet | 0.1036 0.1735 0.2397 0.4795 0.7180 0.9574 | 4 mg to 6 mg per day | 349 to 524 | −5,125 (−94%) to −4,950 (−90%) | −6,949 (−95%) to −6,774 (−93%) |
Other long-acting atypical antipsychotic injectables | |||||||
Aripiprazole (Abilify Maintena) | 200 mg/mL | 300 mg 400 mg powder for injection | 456.1800 456.1800 | 300 mg to 400 mg monthly | 5,474 5,474 | 0 (0%) 0 (0%) | −1,924 (−25%) −1,924 (−25%) |
Paliperidone (Invega Sustenna) | 100 mg/mL | 50 mg 75 mg 100 mg 150 mg pre-filled syringe | 317.9100 476.8700 476.8700 635.8300 | 50 mg to 150 mg monthly | 3,815 5,722 5,722 7,630 | −1,659 (−30%) 248 (4%) 248 (4%) 2,156 (39%) | −3,484 (−48%) −1,576 (−22%) −1,576 (−22%) 331 (4%) |
Paliperidone (Invega Trinza) | 200 mg/mL | 175 mg 263 mg 350 mg 525 mg pre-filled syringe | 953.7300 1,430.6100 1,430.6100 1,907.4900 | 175 mg to 525 mg every 3 months | 3,815 5,722 5,722 7,630 | −1,659 (−30%) 248 (4%) 248 (4%) 2,156 (39%) | −3,484 (−48%) −1,576 (−22%) −1,576 (−22%) 331 (4%) |
Note: All prices are from the Ontario Drug Benefit Formulary, accessed April 202148 unless otherwise specified. Reanalyses are based on publicly available prices of the comparator treatments. All calculations assume a 365-day year.
Percent relative difference calculated as: (comparator cost divided by risperidone ER cost minus 1) × 100%.
aSponsor’s submitted price.46
bSaskatchewan Formulary, accessed April 2021.49
Price-Reduction Analyses
The annual cost of risperidone ER is within the range of other available LAI AAPs at publicly available list prices. However, the lowest recommended doses of risperidone LAI, paliperidone LAI monthly, and paliperidone LAI every 3 months, are less expensive than the lowest recommended dose of risperidone ER.
As there was no submitted evidence on the relative safety and efficacy of risperidone ER and its comparators, uncertainty exists regarding comparative clinical effects and dose equivalencies; therefore, a cost comparison between the lowest recommended doses of each comparator may be appropriate.50 In order for the annual cost of the lowest recommended dose of risperidone ER to equal that of the lowest recommended dose of risperidone LAI or that of the lowest recommended dose of paliperidone LAI (monthly or every 3 months), the submitted price of risperidone ER would need to be reduced by 18% and 30%, respectively (Table 12).
However, as reported in the IQVIA Pharmastat Plus database,51 between 47% (risperidone LAI) and 73% (aripiprazole LAI) of publicly reimbursed claims in 2020 were reimbursed for the highest available dose of each comparator. At current list prices, the highest dose of risperidone ER is less expensive than risperidone LAI and both types of paliperidone LAI, but more expensive than aripiprazole. In order for the cost of the highest recommended dose of risperidone ER to equal that of the highest recommended dose of aripiprazole LAI, the price of risperidone ER 120 mg would need to be reduced by 25%.
Table 12: CADTH Price Reduction Analyses
Scenario | Current list price ($) | Reduction needed (%) | Reduced price ($) | Savingsa ($) |
|---|---|---|---|---|
Price reduction required for 90 mg risperidone ER to equal the lowest recommended dose of risperidone LAI | 456.18 | 18.1 | 369.96 | 1,035 |
Price reduction required for 90 mg risperidone ER to equal the lowest recommended dose of risperidone LAI when administration is includedb | 456.18 | 17.2 | 377.81 | 940 |
Price reduction required for 90 mg risperidone ER to equal the lowest recommended dose of a monthly injectable AAP (paliperidone LAI monthly) | 456.18 | 30.3 | 317.91 | 1,659 |
Price reduction required for 120 mg risperidone ER to equal the highest recommended dose of aripiprazole LAI | 608.22 | 25 | 456.18 | 1,827 |
AAP = atypical antipsychotic; ER = extended release; LAI = long-acting injectable.
aSavings from the sponsor’s list price per patient per year.
bAdministration assumes a fee of $6.75 per injection every month for risperidone ER and every 2 weeks for risperidone LAI, based on the Ontario Schedule of Benefits for Physician Services, Code G373.47
Issues for Consideration
Lack of loading dose or cross-tapering requirement: As with other LAIs, patients initiating risperidone ER require an established tolerance to the oral form of their medication; however, risperidone ER does not require concomitant use with an oral antipsychotic therapy. In contrast, concomitant use of an oral antipsychotic drug is required during the initial 2 to 3 weeks of therapy with risperidone LAI every 2 weeks and aripiprazole LAI. Risperidone ER also does not require an additional injection as a loading dose, while it is required when initiating paliperidone LAI monthly injections or as an alternate to concomitant oral therapy when initiating aripiprazole LAI.18-20,22 This benefit eliminates the costs associated with cross-tapering and loading doses and may be an advantage in reducing the monitoring intensity required for outpatients or the length of stay required for inpatients when initiating LAI AAPs.
Risperidone every 2 weeks injection patent expiration: The patent for Risperdal Consta (patent 240536) expired on April 19, 2021, according to the Health Canada Patent Register database.52 As such, it is possible that generic versions of the risperidone every 2 weeks LAI will soon become available on the Canadian market. This would change the relative cost of treatment between LAI risperidone options and the economic and budgetary impact of risperidone ER’s introduction to the market. Assuming a generic product becomes available at 25% of the current Risperdal Consta list price, the annual cost of treatment with the generic would be $1,110 to $2,219 per patient, which is $3,255 to $6,189 less expensive per patient than risperidone ER, depending on the relative doses.
Doses above those recommended: The clinical expert consulted by CADTH indicated that, for some patients, the dose of long-acting AAPs may be increased above, or the interval between injections may be decreased below, what is recommended in the product monographs. For example, some patients may receive aripiprazole or paliperidone every 3 weeks instead of monthly. Should the interval of use for risperidone ER also be reduced in some patients, or the dose increased, calculated incremental savings or costs will be increased.
Discussion
Summary of Available Evidence
The CADTH clinical review was based on a summary of clinical evidence provided by the sponsor in accordance with the CADTH tailored review process. One phase III, randomized, double-blind, placebo-controlled study (Study 09 to 0010) that was performed at 33 centres in the US was included in the sponsor’s summary of the clinical evidence. The objective of Study 09 to 0010 was to evaluate the efficacy and safety of risperidone ER compared with placebo in patients (N = 354) aged 18 to 55 years with moderate-to-severe schizophrenia in an acute exacerbation phase. The study was conducted on an inpatient basis in a hospital setting. Patients were randomized to 1 of 3 treatment groups: risperidone ER 90 mg SC, risperidone ER 120 mg SC, or placebo injection SC for 8 weeks. The primary outcome was the change from baseline in PANSS total score at end-of-treatment, and the secondary outcome was change from baseline to end of treatment on the CGI-S.
Baseline demographic characteristics were generally well balanced across treatment arms in the pivotal study. The majority of patients included in the study were Black (> 70%) and male (> 73.5%).
Interpretation of Results
Efficacy
In terms of the PANSS total score (the primary outcome), both risperidone ER dose regimens (90 mg and 120 mg SC) demonstrated an improvement at the end of the study (week 8). An improvement in the placebo group was also observed, which could reflect a potential placebo or supportive care response or natural fluctuations of the disease. Compared with placebo, both risperidone ER 90 mg and risperidone ER 120 mg demonstrated a statistically significant improvement (risperidone ER minus placebo LSMD of −6.15; 95% CI, − 9.98 to – 2.31; P = 0.0004 for risperidone ER 90 mg and an LSMD of −7.24; 95% CI, −11.05 to −3.43; P < 0.0001 for risperidone ER 120 mg, respectively).40 The CADTH clinical expert consulted for this review indicated that PANSS was most commonly used in clinical research, but not commonly used in routine clinical practice. Usually, an improvement of 20% in the PANSS positive scale score was considered clinical meaningful. Therefore, the clinical importance of the difference between risperidone ER groups and placebo remains uncertain.
In terms of CGI-S score (the secondary outcome), both risperidone ER dose regimens (90 mg and 120 mg SC) and placebo demonstrated an improvement at the end of the study. Compared with placebo, both the risperidone ER 90 mg and 120 mg groups also demonstrated a statistically significant improvement. However, neither of the risperidone ER groups changes from baseline nor the treatment difference between risperidone ER and placebo reached the minimal clinically important difference estimated for the CGI-S (i.e., a 1-point decrease53).
One of the goals of the treatment of schizophrenia is to maintain or restore HRQoL. The EQ-5D-5L, SWN-S, and MSQ were assessed in the pivotal study. However, HRQoL was assessed only as a tertiary outcome in Study 09 to 0010; therefore, no conclusions can be made.
Harms
The proportion of the patients who experienced at least 1 TEAE was higher for the risperidone ER 120 mg group (77.8%) compared with the risperidone ER 90 mg (70.4%) and placebo groups (68.6%). The incidence of the TEAEs that were reported as related to the study drug was higher for the 120 mg (55.6%) and 90 mg (50.4%) groups compared with the placebo (42.4%) group. The most frequently reported TEAEs occurring at higher rates in the risperidone ER groups compared with the placebo group were weight gain (13%, 12.8%, and 3.4% in the risperidone ER 90 mg and 120 mg groups and placebo group, respectively) and somnolence (5.2%, 4.3%, and 0% in the risperidone ER 90 mg and 120 mg groups and placebo group, respectively).
There were no deaths reported during the study. The incidence of serious TEAEs was infrequent (0%, 0.9%, and 0.8% in the risperidone ER 90 mg and 120 mg groups and placebo group, respectively). The proportion of patients who withdrew due to AEs was reportedly low (0%, 1.7%, and 2.5% in the risperidone ER 90 mg and 120 mg groups and placebo group, respectively). Regarding the AEs of special interest for this review, more patients in the risperidone ER groups (13%) experienced weight gain compared with patients in the placebo group (3.4%), which was an expected AE, reported with all other AAP drugs.
Overall, treatment with risperidone ER (90 mg and 120 mg) over an 8-week treatment period was generally well tolerated in adult patients with acute schizophrenia. There were no new safety signals compared with the known safety profile of oral risperidone products, except for the expected local injection-site reactions that are observed with all LAI antipsychotic drugs.24 The clinical expert consulted for this review indicated that the AEs reported in the included trial might be similar to those observed with risperidone (oral and LAIs) and paliperidone in clinical practice. The finding of no significant extrapyramidal side effects with active treatment may differ from a clinician’s experience of oral risperidone, especially at higher doses.
Other Considerations
Other formulations of risperidone are currently approved in Canada, including a tablet (Risperdal) for once-daily oral administration and an LAI formulation (Risperdal Consta) for IM SC administration every 2 weeks. Currently, the reimbursement status of risperidone LAI is “restricted benefit with specified criteria” (e.g., special authorization, exception drug status, limited-use benefit) in 10 jurisdictions, and it has full benefit status in 4 jurisdictions. No reimbursement status for oral risperidone and other relevant LAIs was provided by the sponsor.
In addition, other existing LAIs for schizophrenia are administered intramuscularly and require oral supplementation or loading doses that are given either the same day or requiring additional office visits. The sponsor indicated that risperidone ER reached therapeutically relevant plasma concentrations on the first day of dosing and required no loading dose or supplemental oral risperidone dosing. Risperidone ER is administered subcutaneously, which may cause less pain, although no evidence addressing this issue was provided in the sponsor’s summary of clinical evidence for this review.
Cost
At the submitted price of $456.18 and $608.22 for the 90 mg and 120 mg doses, respectively, the annual cost of risperidone ER is $5,474 to $7,299 per patient. This annual cost is more expensive than that of risperidone tablets ($349 to $524 per patient annually), but within the range of other LAI AAPs ($3,815 to $8,877 per patient annually). However, the highest available dose of each LAI AAP comparator is the most frequently used. In order for the cost of the highest recommended dose of risperidone ER to equal that of the least expensive comparator at its highest recommended dose (aripiprazole LAI), the price of risperidone ER 120 mg would need to be reduced by 25%.
No evidence was submitted to support the sponsor’s assumption of clinical similarity between LAI AAP comparators, nor evidence to support implicit assumptions of dose-equivalency between LAI AAPs. As such, any conclusions regarding incremental costs or savings associated with the reimbursement of risperidone ER are uncertain. Additionally, these incremental costs or savings are based on publicly available list prices and may not reflect actual prices paid by Canadian public drug plans.
Conclusions
Based on the summary of clinical evidence submitted by the sponsor, risperidone ER 90 mg and 120 mg (SC once monthly) showed statistically significant improvements in schizophrenia symptoms compared with placebo after 8 weeks, as measured by PANSS total scores and CGI-S scores; however, given that improvements in these outcomes were also observed in the placebo group, the clinical importance of these results is uncertain. Findings pertaining to the potential impact of risperidone ER on patient HRQoL were not conclusive, due to its methodological limitations. The safety of the once-monthly formulation appears to be consistent with the safety profile of risperidone (both oral and LAI every 2 weeks). Key evidence gaps include the short duration of the trial. Risperidone ER is intended to be used as a chronic treatment, and longer trials comparing it with the existing oral risperidone or LAI AAP drugs available in Canada for the maintenance treatment of schizophrenia are needed to adequately assess long-term outcomes, including mortality, relapse, remission, and hospitalization.
At the submitted price, the annual cost of risperidone ER is $5,474 to $7,299 per patient, which is more expensive than that of risperidone tablets ($349 to $524 per patient per year), but within the range of other LAI AAPs ($3,815 to $8,877 per patient annually). However, the highest available dose of each LAI AAP comparator is the most frequently used. In order for the cost of the highest recommended dose of risperidone ER to equal that of the least expensive comparator at its highest recommended dose (aripiprazole LAI), the price of risperidone ER 120 mg would need to be reduced by 25%. No evidence was submitted to support the sponsor’s assumption of clinical similarity between LAI AAP comparators, nor evidence to support implicit assumptions of dose-equivalency between LAI AAPs; thus, any conclusions regarding incremental costs or savings associated with the reimbursement of risperidone ER are uncertain, particularly when long-term clinical outcomes are considered. Additionally, these incremental costs or savings are based on publicly available list prices and may not reflect actual prices paid by Canadian public drug plans.
References
1.Mc Glanaghy E, Turner D, Davis GA, et al. A network meta-analysis of psychological interventions for schizophrenia and psychosis: Impact on symptoms. Schizophr Res. 2021;228:447-459. PubMed
2.Remington G, Addington D, Honer W, Ismail Z, Raedler T, Teehan M. Guidelines for the pharmacotherapy of schizophrenia in adults. Can J Psychiatry. 2017;62(9):604-616. PubMed
3.Jablensky A, Sartorius N, Ernberg G, et al. Schizophrenia: manifestations, incidence and course in different cultures. A World Health Organization ten-country study. Psychol Med Monogr Suppl. 1992;20:1-97. PubMed
4.American Psychiatric Association. Diagnostic and statistical manual of mental disorders (DSM-5). 5th ed. Washington (DC): American Psychiatric Publishing; 2013.
5.Lewis DA, Lieberman JA. Catching up on schizophrenia: natural history and neurobiology. Neuron. 2000;28(2):325-334. PubMed
6.A report on mental illnesses in Canada. Ottawa (ON): Health Canada; 2002: https://www.phac-aspc.gc.ca/publicat/miic-mmac/pdf/men_ill_e.pdf. Accessed 2021 May 6.
7.Clinical practice guidelines. Treatment of schizophrenia. Can J Psychiatry. 2005;50(13 Suppl 1):7s-57s. PubMed
8.Kasper S, Lerman MN, McQuade RD, et al. Efficacy and safety of aripiprazole vs. haloperidol for long-term maintenance treatment following acute relapse of schizophrenia. Int J Neuropsychopharmacol. 2003;6(4):325-337. PubMed
9.Government of Canada. Canadian Chronic Disease Surveillance System (CCDSS). 2018; https://health-infobase.canada.ca/ccdss/data-tool/. Accessed 2021 May 6.
10.Chong HY, Teoh SL, Wu DB, Kotirum S, Chiou CF, Chaiyakunapruk N. Global economic burden of schizophrenia: a systematic review. Neuropsychiatr Dis Treat. 2016;12:357-373. PubMed
11.Diagnostic and statistical manual of mental disorders: DSM-IV-TR. Washington (DC): American Psychiatric Association; 2000.
12.O'Carroll R. Cognitive impairment in schizophrenia. Adv Psychiatr Treat. 2000;6(3):161-168.
13.Barnes TR, Drake R, Paton C, et al. Evidence-based guidelines for the pharmacological treatment of schizophrenia: Updated recommendations from the British Association for Psychopharmacology. J Psychopharmacol. 2020;34(1):3-78. PubMed
14.Schizophrenia Care in the Community for adults. Toronto (ON): Health Quality Ontario; 2018: https://www.hqontario.ca/Portals/0/documents/evidence/quality-standards/qs-schizophrenia-community-clinician-guide-en.pdf. Accessed 2021 May 6.
15.Ayuso-Gutiérrez JL, del Río Vega JM. Factors influencing relapse in the long-term course of schizophrenia. Schizophr Res. 1997;28(2-3):199-206. PubMed
16.Cramer JA, Rosenheck R. Compliance with medication regimens for mental and physical disorders. Psychiatr Serv. 1998;49(2):196-201. PubMed
17.Lauriello J, Campbell AR. Pharmacotherapy for schizophrenia: Long-acting injectable antipsychotic drugs. In: Post TW, ed. UpToDate. Waltham (MA): UpToDate; 2021: www.uptodate.com. Accessed 2021 May 6.
18.Risperdal Consta® (risperidone powder for injectable prolonged-release suspension): 12.5 mg, 25 mg, 37.5 mg and 50 mg [product monograph]. Toronto (ON): Janssen Inc.; 2020 Dec 18: https://pdf.hres.ca/dpd_pm/00059290.PDF. Accessed 2021 May 6.
19.Abilify Maintena® (aripiprazole for prolonged release injectable suspension): 300 mg and 400 mg vial, 300 mg and 400 mg dual chamber syringe [product monograph]. Saint-Laurent (QC): Otsuka Canada Pharmaceutical Inc.; 2020 Dec 21: https://pdf.hres.ca/dpd_pm/00059334.PDF. Accessed 2021 May 6.
20.Invega Sustenna (paliperidone palmitate prolonged-release injectable suspension): pre-filled syringes containing 50 mg/0.5 mL, 75 mg/0.75 mL, 100 mg/1 mL, and 150 mg/1.5 mL [product monograph]. Toronto (ON): Janssen Inc.; 2020 Dec 17: https://pdf.hres.ca/dpd_pm/00059297.PDF. Accessed 2021 May 6.
21.Invega Trinza (paliperidone palmitate prolonged-release injectable suspension): pre-filled syringes containing 175 mg/0.875 mL, 263 mg/1.315 mL, 350 mg/1.75 mL, and 525 mg/2.625 mL (3 months) [product monograph]. Toronto (ON): Janssen Inc.; 2020 Dec 17: https://pdf.hres.ca/dpd_pm/00059281.PDF. Accessed 2021 May 6.
22.Perseris (risperidone for extended-release injectable suspension): powder for suspension, 90 mg or 120 mg subcutaneous injection [product monograph]. Etobicoke (ON): HLS Therapeutics Inc.; 2020.
23.Clinical Study Report: RB-US-09-0010. A phase 3, randomized, double-blind, placebo-controlled, multicenter study to evaluate the efficacy, safety and tolerability of RBP-7000 (90 mg and 120 mg) as a treatment in subjects with acute schizophrenia over 8 weeks (2 subcutaneous doses) [internal sponsor's report]. Richmond (VA): Reckitt Benckiser Pharmaceuticals Inc.; 2015 Jun 16.
24.Drug Reimbursement Review sponsor submission: Perseris (risperidone for extended-release injectable suspension 90 mg and 120 mg) [internal sponsor's package]. Etobicoke (ON): HLS Therapeutics Inc.; 2021 Jan 29.
25.Nasser AF, Henderson DC, Fava M, et al. Efficacy, safety, and tolerability of RBP-7000 once-monthly risperidone for the treatment of acute schizophrenia: an 8-Week, randomized, double-blind, placebo-controlled, multicenter phase 3 study. J Clin Psychopharmacol. 2016;36(2):130-140. PubMed
26.Chouinard G, Jones B, Remington G, et al. A Canadian multicenter placebo-controlled study of fixed doses of risperidone and haloperidol in the treatment of chronic schizophrenic patients. J Clin Psychopharmacol. 1993;13(1):25-40. PubMed
27.Marder SR, Meibach RC. Risperidone in the treatment of schizophrenia. Am J Psychiatry. 1994;151(6):825-835. PubMed
28.Kane JM, Eerdekens M, Lindenmayer JP, Keith SJ, Lesem M, Karcher K. Long-acting injectable risperidone: efficacy and safety of the first long-acting atypical antipsychotic. Am J Psychiatry. 2003;160(6):1125-1132. PubMed
29.Chue P, Eerdekens M, Augustyns I, et al. Comparative efficacy and safety of long-acting risperidone and risperidone oral tablets. Eur Neuropsychopharmacol. 2005;15(1):111-117. PubMed
30.The American Psychiatric Association practice guideline for the treatment of patients with schizophrenia. 3rd ed. Washington (DC): American Psychiatric Association; 2021: https://psychiatryonline.org/doi/pdf/10.1176/appi.books.9780890424841 Accessed 2021 May 6.
31.Kay SR, Fiszbein A, Opler LA. The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr Bull. 1987;13(2):261-276. PubMed
32.Kay SR, Opler LA, Lindenmayer JP. Reliability and validity of the positive and negative syndrome scale for schizophrenics. Psychiatry Res. 1988;23(1):99-110. PubMed
33.Charlson FJ, Ferrari AJ, Santomauro DF, et al. Global epidemiology and burden of schizophrenia: findings from the Global Burden of Disease Study 2016. Schizophr Bull. 2018;44(6):1195-1203. PubMed
34.Marder SR, McQuade RD, Stock E, et al. Aripiprazole in the treatment of schizophrenia: safety and tolerability in short-term, placebo-controlled trials. Schizophr Res. 2003;61(2-3):123-136. PubMed
35.Lehman AF, Kreyenbuhl J, Buchanan RW, et al. The Schizophrenia Patient Outcomes Research Team (PORT): updated treatment recommendations 2003. Schizophr Bull. 2004;30(2):193-217. PubMed
36.Health Canada. Notices of compliance: prescription products for human use. January - December, 1993. 1993; https://www.canada.ca/content/dam/hc-sc/migration/hc-sc/dhp-mps/alt_formats/hpfb-dgpsa/txt/prodpharma/pre93et-eng.txt. Accessed 2021 Apr 27.
37.Health Canada. Notice of compliance (NOC) for Risperdal Consta (risperidone powder for injectable prolonged-release suspension). 2004 Jul 16; https://health-products.canada.ca/noc-ac/info.do?lang=en&no=4799. Accessed 2021 Apr 26.
38.Risperdal® (risperidone): tablets house standard 0.25, 0.5, 1, 2, 3 and 4 mg risperidone tartrate oral solution risperidone 1 mg/mL [product monograph]. Toronto (ON): Janssen Inc.; 2018 Sep 10: https://pdf.hres.ca/dpd_pm/00047330.PDF. Accessed 2021 May 6.
39.Isitt JJ, Nadipelli VR, Kouassi A, Fava M, Heidbreder C. Health-related quality of life in acute schizophrenia patients treated with RBP-7000 once monthly risperidone: An 8-week, randomized, double-blind, placebo-controlled, multicenter phase 3 study. Schizophr Res. 2016;174(1-3):126-131. PubMed
40.Leucht S, Kane JM, Etschel E, Kissling W, Hamann J, Engel RR. Linking the PANSS, BPRS, and CGI: clinical implications. Neuropsychopharmacology. 2006;31(10):2318-2325. PubMed
41.Andreasen NC, Carpenter WT, Jr., Kane JM, Lasser RA, Marder SR, Weinberger DR. Remission in schizophrenia: proposed criteria and rationale for consensus. Am J Psychiatry. 2005;162(3):441-449. PubMed
42.van Os J, Burns T, Cavallaro R, et al. Standardized remission criteria in schizophrenia. Acta Psychiatr Scand. 2006;113(2):91-95. PubMed
43.Health Canada reviewer's report: Perseris (risperidone for extended-release injectable suspension) [internal sponsor's report]. Etobicoke (ON): HLS Therapeutics Inc.; 2020.
44.Andorn A, Graham J, Csernansky J, et al. Monthly extended-release risperidone (RBP-7000) in the treatment of schizophrenia: results from the phase 3 program. J Clin Psychopharmacol. 2019;39(5):428-433. PubMed
45.Laffont CM, Gomeni R, Zheng B, Heidbreder C, Fudala PJ, Nasser AF. Population pharmacokinetics and prediction of dopamine D2 receptor occupancy after multiple doses of RBP-7000, a new sustained-release formulation of risperidone, in schizophrenia patients on stable oral risperidone treatment. Clin Pharmacokinet. 2014;53(6):533-543. PubMed
46.Pharmacoeconomic evaluation [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Perseris (risperidone for extended-release injectable suspension 90 mg and 120 mg). Etobicoke (ON): HLS Therapeutics Inc.; 2021.
47.Schedule of benefits for physician services under the Health Insurance Act: effective April 1, 2020. Toronto (ON): Ontario Ministry of Health; 2020: https://www.health.gov.on.ca/en/pro/programs/ohip/sob/physserv/sob_master20200306.pdf. Accessed 2021 Apr 05.
48.Ontario Ministry of Health, Ontario Ministry of Long-Term Care. Ontario drug benefit formulary/comparative drug index. 2020; https://www.formulary.health.gov.on.ca/formulary/. Accessed 2021 Apr 08.
49.Saskatchewan Drug Plan: search formulary. 2021; https://formulary.drugplan.ehealthsask.ca/SearchFormulary. Accessed 2021 Apr 15.
50.Rothe PH, Heres S, Leucht S. Dose equivalents for second generation long-acting injectable antipsychotics: The minimum effective dose method. Schizophr Res. 2018;193:23-28. PubMed
51.PharmaStat Plus. [Ottawa (ON)]: IQVIA; 2021: https://www.iqvia.com/. Accessed 2021 Apr 26.
52.Patent Register Database. 2021; https://pr-rdb.hc-sc.gc.ca/pr-rdb/index-eng.jsp. Accessed 2021 Apr 08.
53.Rabinowitz J, Mehnert A, Eerdekens M. To what extent do the PANSS and CGI-S overlap? J Clin Psychopharmacol. 2006;26(3):303-307. PubMed
54.Peralta V, Cuesta MJ. Psychometric properties of the positive and negative syndrome scale (PANSS) in schizophrenia. Psychiatry Res. 1994;53(1):31-40. PubMed
55.Marder SR, Davis JM, Chouinard G. The effects of risperidone on the five dimensions of schizophrenia derived by factor analysis: combined results of the North American trials. J Clin Psychiatry. 1997;58(12):538-546. PubMed
56.Bell MD, Lysaker PH, Milstein RM, Beam-Goulet JL. Concurrent validity of the cognitive component of schizophrenia: relationship of PANSS scores to neuropsychological assessments. Psychiatry Res. 1994;54(1):51-58. PubMed
57.Bell MD, Lysaker PH, Beam-Goulet JL, Milstein RM, Lindenmayer JP. Five-component model of schizophrenia: assessing the factorial invariance of the positive and negative syndrome scale. Psychiatry Res. 1994;52(3):295-303. PubMed
58.Lançon C, Auquier P, Nayt G, Reine G. Stability of the five-factor structure of the Positive and Negative Syndrome Scale (PANSS). Schizophr Res. 2000;42(3):231-239. PubMed
59.Lindenmayer JP, Bernstein-Hyman R, Grochowski S. A new five factor model of schizophrenia. Psychiatr Q. 1994;65(4):299-322. PubMed
60.Guy W. ECDEU assessment manual for psychopharmacology. Revised 1976. Rockville (MD): U.S. Department of Health, Education and Welfare; 1976.
61.Haro JM, Kamath SA, Ochoa S, et al. The Clinical Global Impression-Schizophrenia scale: a simple instrument to measure the diversity of symptoms present in schizophrenia. Acta Psychiatr Scand Suppl. 2003(416):16-23. PubMed
62.Budget Impact Analysis [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Perseris (risperidone for extended-release injectable suspension 90 mg and 120 mg). Etobicoke (ON): HLS Therapeutics Inc.; 2021.
63.HLS Therapeutics response to April 14 2021 request for additional information regarding Perseris review: Request for clarification of BIA methods [internal additional sponsor's information]. Dorval (QC): HLS Therapeutics; 2021.
64.Audit Training: CompuScript, Key Things to Know. https://caelearning.customerportal.iqvia.com/cs/a001_wrap_up_key_things_to_know.html. Accessed 2021 Apr 26.
65.Younis IR, Gopalakrishnan M, Mathis M, et al. Association of End Point Definition and Randomized Clinical Trial Duration in Clinical Trials of Schizophrenia Medications. JAMA Psychiatry. 2020;77(10):1064-1071. PubMed
66.Dhanda R, Varghese D, Nadipelli VR, et al. Patient-reported outcomes in schizophrenia patients treated with once-monthly extended-release risperidone in a long-term clinical study. Patient Prefer Adherence. 2019;13:1037-1050. PubMed
Appendix 1: Description and Appraisal of Outcome Measures
Note that this appendix has not been copy-edited.
Aim
To describe the following outcome measures summarized in Table 13 and review their measurement properties including validity, reliability, responsiveness to change, and clinical relevance (i.e., minimal important difference).
Table 13: Outcome Measures Included in Study 09-0010
Outcome measure | Study 09-0010 |
|---|---|
PANSS | Primary |
CGI-S | Secondary |
CGI-S = Clinical Global Impression–Severity of Illness; PANSS = Positive and Negative Syndrome Scale.
Source: Sponsor’ submission,24 Clinical Study Report.23
Findings
The primary and secondary outcome measures are briefly summarized in Table 14.
Table 14: Summary of Outcome Measures and Their Measurement Properties
Instrument | Type | Conclusions about measurement properties | MID |
|---|---|---|---|
PANSS | 30-item rating scale (3 subscales; a score 1 indicating the absence of symptoms and a score of 7 indicating extremely severe symptoms): positive symptoms, negative symptoms, and general psychopathology. | Validity: Scores on all subscales were reported to exhibit a normal distribution. The range of scores was less than the potential range suggesting a lack of ceiling effect. Internal consistency was demonstrated for the positive (alpha = 0.73), negative (alpha = 0.83) and the general psychopathology (alpha = 0.79) subscales. Reliability: Test-retest reliability: Pearson correlation coefficients were 0.80, 0.68, and 0.60 for the positive, negative, and general psychopathology subscales, respectively.31 Positive and negative scales showed good inter-rater reliability; interclass correlation coefficients (ICC) of 0.72 and 0.80, respectively. Inter-rater reliability was moderate for the general psychopathology scale; ICC = 0.56. | Unclear, depends on baseline severity40 Usually, a 15-point reduction or a 20% reduction in the PANSS total score were considered as clinical improvement (similar to 1 point of CGI-S reduction)40,53 |
CGI | 3-item scale: Severity of illness (CGI-S), global improvement (CGI-I) and efficacy index (CGI-E). CGI-S and CGI-I are rated from a 1 (normal or very much improved) to 7 (extremely ill or very much worse) scale and are considered separately. | There is little information regarding its reliability or validity. In a study53 based on a predefined measures of clinical improvement were a 20% reduction in the PANSS score and a 1-point decrease on the CGI-S, CGI-S scores from the pooled trials corresponded to the following mean PANSS scores: 1 (normal) = PANSS 55.5, 2 (borderline ill) = PANSS 67.0, 3 (mildly ill) = PANSS 79.6, 4 (moderately ill )= PANSS 92.4, and 5 (markedly ill) = PANSS 99.7. | 1 point53 |
CGI-E = Clinical Global Impression Efficacy; CGI-S = Clinical Global Impression–Severity of Illness; MID = minimal important difference; PANSS = Positive and Negative Syndrome Scale.
Positive and Negative Syndrome Scale
The PANSS was developed as a 30-item rating scale, which adapted 18 items from the Brief Psychiatric Rating Scale and 12 items from the Psychopathology Rating Schedule (PRS). The PANSS requires a 30- to 40-minute patient interview to gather information on which to assess the patient with regard to the presence and severity of psychopathology in the previous week. The PANSS instrument provides a complete definition of each item as well as detailed anchoring criteria for each of the 7 rating points: 1 = absent, 2 = minimal, 3 = mild, 4 = moderate, 5 = moderate-severe, 6 = severe, 7 = extreme. A score of 1 indicates the absence of symptoms and a score of 7 indicates extremely severe symptoms. In the 30-item scale, 7 items are related to positive symptoms (i.e., delusions, conceptual disorganization, hallucinatory behaviour, excitement, grandiosity, suspiciousness and hostility), 7 items to negative symptoms (i.e., blunted affect, emotional withdrawal, poor rapport, passive/apathetic social withdrawal, difficulty in abstract thinking, lack of spontaneity and flow of conversation and stereotyped thinking), and 16 items (i.e., somatic concern, anxiety, guilt feelings, tension, mannerisms and posturing, depression, motor retardation, uncooperativeness, unusual thought content, disorientation, poor attention, lack of judgment and insight, disturbance of volition, poor impulse control, preoccupation and active social avoidance) to general psychopathology.31,32 The General Psychopathology scale is considered an adjunct to the positive and negative syndrome assessment since it provides a separate but parallel measure of schizophrenia severity that can serve as a point of reference for interpreting the positive and negative scores.31 Finally, a composite scale may be derived by subtracting the negative from the positive score. This scale expresses the direction and magnitude of difference between positive and negative syndromes. This score may reflect the degree of predominance of 1 syndrome over the other based on the score (positive or negative).
In clinical trials, changes from baseline in the PANSS total score, as well those for the positive and negative subscales, are typically used as study end points. The PANSS total is scored by summing ratings across the items in 3 subscales; the potential ranges are 7 to 49 for both the positive and negative subscales and 16 to 112 for the General Psychopathology subscale. Thus, the total range of possible scores for the 3 subscales is from 30 to 210. The General Psychopathology subscale is usually not rated individually, but it is captured in the total score. The range of scores for the composite scale is from −42 to 42, which may be used to characterize whether positive or negative symptoms predominate and is not a part of the PANSS total score.
Validity
Kay et al. reported on psychometric testing of the PANSS in 101 inpatients with schizophrenia.31 Scores on all subscales were reported to exhibit a normal distribution, suggesting suitability for parametric statistical analysis. Further, the range of scores was less than the potential range suggesting a lack of ceiling effect. Internal consistency was demonstrated for the positive (alpha = 0.73), negative (alpha = 0.83) and the general psychopathology (alpha = 0.79) subscales.
Reliability
Test-retest reliability was assessed 3 to 6 months later on a cohort of 15 patients who remained hospitalized; Pearson correlation coefficients were 0.80, 0.68, and 0.60 for the positive, negative, and general psychopathology subscales, respectively.31 Peralta and Cuesta reported on the inter-rater reliability of the PANSS from a sample of 100 consecutively admitted patients with schizophrenia.54 Positive and negative scales showed good inter-rater reliability; the interclass correlation coefficients (ICCs) were 0.72 and 0.80, respectively. Inter-rater reliability was moderate for the general psychopathology scale (ICC = 0.56).
More recently, a number of investigators have conducted a principal component analysis to expand the identification of discrete dimensions of schizophrenia beyond the focus on positive and negative symptoms. A number of similar 5-factor models including most or all of the original PANSS items have been proposed and tested for reliability and validity.55-59 One such model was proposed by Marder et al. and categorizes all original PANSS items into 5 dimensions; positive symptoms (8 items), negative symptoms (7 items), disorganized thought (7 items), uncontrolled hostility/excitement (4 items), and anxiety/depression (4 items).55
Clinical Relevance
However, in a comparison of PANSS to the Clinical Global Impressions (CGI) scale, it was suggested that an absolute reduction of 15 in the total PANSS score corresponds to “minimally improved” on the CGI-Improvement score, and a reduction of the CGI–Severity of Illness score by 1 severity step.40 In comparison, a reduction of 33 in the total PANSS score corresponds to “much improved” on the CGI-Improvement score. However, the above estimates were sensitive to baseline severity of illness to the extent that participants with a lower baseline severity of illness required smaller reductions in the PANSS to produce a particular improvement in the CGI. For this reason, it has been suggested that change in PANSS score has limited usefulness as a primary outcome, due to variability in baseline symptom intensity.41,42 Rather, standardized remission criteria, which may be suitable for use in clinical practice and clinical trials, has been proposed. Specifically, a score of ≤ 3 on 8 PANSS items (P1, P2, P3, N1, N4, N6, G5 and G9) for a period of at least 6 months is considered to represent remission of disease.41,42 in addition, similar to 1-point decrease on the CGI-S, a 20% reduction in the PANSS total score was considered as clinical improvement.53
Clinical Global Impression (CGI)
The CGI scale is a 3-item scale used to assess overall severity and response to treatment of mental disorders.60 It is not specific to schizophrenia, although efforts to adapt the scale to this disorder have been undertaken.61 The usual CGI scale items include severity of illness (CGI-S) at the time of the assessment on a 7-point scale (1 = normal; 7 = extremely ill), global improvement (CGI-Improvement) relative to baseline on a 7-point scale (1 = very much improved; 7 = very much worse), and an efficacy index which incorporates the clinician’s assessment of therapeutic effect in relation to adverse effects in a 4 point x 4 point grid rating scale (0 = marked improvement and no AEs; 4 = unchanged or worse, and AEs outweigh the therapeutic events).60 The difficulty of combing the 2 concepts of efficacy and AEs has led to criticism of this last item.61 However, there is no total score for the CGI, rather scores on the individual items are considered separately.
Validity, Reliability, Responsiveness, and Clinical Relevance
As the CGI is quick to administer it is suited to clinical settings; however, there is little information regarding its reliability or validity. Rabinowitz et al. sought to validate the CGI-S through a comparison of PANSS and CGI-S scores from 7 trials of risperidone in schizophrenia.53 CGI-S scores from the pooled trials corresponded to the following mean PANSS scores; 1 (normal) = PANSS 55.5, 2 (borderline ill) = PANSS 67.0, 3 (mildly ill) = PANSS 79.6, 4 (moderately ill) = PANSS 92.4, and 5 (markedly ill) = PANSS 99.7. Predefined measures of clinical improvement were a 20% reduction in the PANSS score and a 1-point decrease on the CGI-S. The sensitivities and specificities for the CGI-S to detect this level of improvement in the 7 trials ranged from 64.5% to 89.6% and 65.7% to 82.8%, respectively. From this assessment it appears that the CGI-S and PANSS are correlated and exhibit substantial agreement in detecting change.
Appendix 2: Additional Economic Information
Note that this appendix has not been copy-edited.
Additional Details on the Sponsor’s Submission
No additional information from the sponsor’s submitted pharmacoeconomic evaluation was considered in the review of risperidone ER injections.
Additional Details on the CADTH Reanalyses and Additional Analyses
CADTH did not conduct any additional pharmacoeconomic analyses in the review of risperidone ER injections.
Appendix 3: Submitted Budget Impact Analysis and CADTH Appraisal
Note that this appendix has not been copy-edited.
Table 15: Key Takeaways of the Budget Impact Analysis
Key Takeaways of the Budget Impact Analysis |
|---|
|
Summary of Sponsor’s Budget Impact Analysis
The sponsor submitted a claims-based budget impact analysis (BIA),62 assessing the expected budgetary impact of introducing risperidone ER for the treatment of schizophrenia over a 3-year time horizon from the perspective of a Canadian drug plan payer, including drug acquisition costs as well as dispensing fees and markups.
Data from the model were obtained from the IQVIA CompuScript Database,62,63 the applicable product monographs,18-22 and assumptions. The included IQVIA data estimated the number of units of each comparator sold within each jurisdiction, and the sponsor then estimated the number of patients using each comparator by dividing the number of units by the comparator’s recommended frequency of administration.
Key inputs to the BIA are documented in Table 16. Other assumptions made by the sponsor include:
All patients use the included comparators at product monograph recommended intervals.
Year 1 will start halfway through 2021.
For patients receiving risperidone ER, |||||||% would be comprised of patients who would otherwise have received aripiprazole monthly injections, |% would be patients who would otherwise have received paliperidone monthly injections, and |% would be patients who would otherwise have received risperidone injections every 2 weeks. |||||||% were assumed to receive risperidone ER who would otherwise receive paliperidone LAI every 3 months.
Table 16: Summary of Key Model Parameters
Parameter | Sponsor’s estimate |
|---|---|
Target population: Adult patients with schizophrenia for whom risperidone ER are being considered. | |
Proportion of units reimbursed by public drug plansa | 80% |
Number of units per yearb / Number of doses per yearc Aripiprazole (Abilify Maintena) Paliperidone palmitate (Invega Sustenna) Risperidone (Risperdal Consta) | Year 0 102,187 / 12 164,033 / 12 67,308 / 26 |
Annual growth rated Aripiprazole (Abilify Maintena) Paliperidone palmitate (Invega Sustenna) Risperidone (Risperdal Consta) | Overall: 9.7% 18.9% 8.3% −8.1% |
Number of patients eligible for drug under review | 27,242 / 29,711 / 32,179 |
Market uptake, number of patients (3 years) | |
Uptake (reference scenario) Aripiprazole (Abilify Maintena) Paliperidone palmitate (Invega Sustenna) Risperidone (Risperdal Consta) | 10,120 / 11,725 / 13,329 14,787 / 15,905 / 17,023 2,335 / 2,081 / 1,827 |
Uptake (new drug scenario) Risperidone (Perseris) Aripiprazole (Abilify Maintena) Paliperidone palmitate (Invega Sustenna) Risperidone (Risperdal Consta) | ||||||| ||||||| ||||||| ||||||| |
Cost of treatment (per patient per year)e | |
Risperidone (Perseris) Aripiprazole (Abilify Maintena) Paliperidone palmitate (Invega Sustenna) Risperidone (Risperdal Consta) | $||||||| $6,112 $7,087 $7,636 |
Note: Number of estimated patients reported in sponsor’s BIA report did not match the number of estimated patients within the BIA Excel model. Where discrepancies exist, figures from the Excel model are reported.62
aCited as an estimation from HLS Therapeutics within the submitted BIA report.62,63
bTotal number of units reported as sold in Canada, excluding Quebec, in an unspecified IQVIA database, multiplied by the 80% of units estimated to have been publicly reimbursed.62
cNumber of doses per year as recommended in applicable product monographs.18-20,22
dAnnual growth rate is based on a linear extrapolation of sales over the 6 years prior to year 0.62
e|||||||% of patients using risperidone ER were assumed to be on the 120 mg dose, while ||||||| % were assumed to be using the 90 mg dose. Other comparator pricing was based on actual sales divided by the number of units dispensed and multiplied by the recommended number of doses per year.62
Summary of the Sponsor’s Budget Impact Analysis Results
Results of the sponsor’s base case suggest that the incremental budget impact associated with the reimbursement of risperidone ER would be a savings of $93,029 in year 1, $481,279 in year 2, and $1,429,812 in year 3, for a cumulative 3-year budgetary savings of $2,004,121 when including dispensing fees and markups.
CADTH Appraisal of the Sponsor’s Budget Impact Analysis
CADTH identified the following key limitations to the sponsor’s analysis that have notable implications on the results of the BIA:
Modelled population is not limited to patients with schizophrenia: The claims included in the BIA represent all claims of the included comparators and are not limited to those reimbursed for schizophrenia. The included comparators are all indicated for schizophrenia, but are also indicated for schizoaffective disorder18,20 or bipolar disorder.18,19 Upon request, the sponsor clarified that risperidone ER’s single indication for schizophrenia is accounted for in its estimated market uptake.63 However, as the annual cost of each comparator is based on the total sales of each product, there is uncertainty around whether the overall distribution and growth rate of doses within the dataset represents the distribution of doses used by patients with schizophrenia. Therefore, the relative costs of the comparators are uncertain.
CADTH was unable to adjust for this limitation in reanalyses.
Market share projection unlikely to represent current growth: The sponsor estimated the annual growth rate for each comparator by conducting a linear extrapolation of the costs reimbursed over the 6 years prior to year 0 for each comparator starting in 2015. However, 2015 was the year aripiprazole LAI was reimbursed by public formularies; therefore, 2015 to 2017 include a large growth in market share for aripiprazole and subsequent displacement of other LAI products. This growth and displacement have not been sustained in recent years, and no longer represent current growth patterns.
CADTH reanalyses used the 3 years prior to year 0 to conduct linear extrapolation to predict the market growth for each comparator.
Comparator’s dispensing fees and markups were double counted: The sponsor calculated the annual cost of comparator products by dividing the annual sales figures by the number of units dispensed, then multiplying by the number of units used per patient per year. They then added the jurisdictionally appropriate markup and dispensing fees to calculate the total cost of each comparator within the reference and new drug scenarios. However, the IQVIA CompuScript Database already includes dispensing fees and markups within its sales data,64 therefore the sponsor’s BIA double counted these costs for all comparators. Additionally, as the dataset includes claims not reimbursed by public plans, the costs of comparators within the dataset may include claims made under wholesale or private plan list prices in addition to those using list prices set by public plans.
CADTH removed the additional dispensing fees and markups added by the sponsor for aripiprazole LAI, paliperidone LAI, and risperidone LAI in reanalyses. CADTH was unable to adjust for other pricing uncertainty of the comparators.
Uptake of risperidone ER is uncertain: The sponsor assumed that ||| patients would begin using risperidone monthly injections in the first year of its availability, rising to | in year 2 and ||||||| in year 3, corresponding to |||||||%, |||||||%, and |||||||% of estimated patients, respectively. Upon request, the sponsor clarified these estimates were based on internal forecasts. Additionally, the sponsor estimated that |% of risperidone ER patients would use the 90 mg monthly dose, with the remainder using the 120 mg dose. As these doses have different pricing, and dose equivalency between comparators is uncertain, changes in the assumption of the proportion of patients who will use each dose affects the overall budget impact.
CADTH explored the impact of assuming | 25% of patients would use the 90 mg monthly dose of risperidone ER. CADTH was unable to properly explore the impact of altering the uptake of risperidone ER due to inflexibility in the model programming, which led to negative numbers of patients using risperidone LAI (Risperidone Consta) when uptake of risperidone ER was increased.
Proportion of units reimbursed by public plans uncertain: The sponsor estimated that 80% of claims for the included AAPs would be reimbursed by public plans.62 Upon request, the sponsor clarified that this estimate was based on internal HLS Therapeutics data indicating that |||||||% of patients using Clozaril (clozapine, the brand distributed by HLS Therapeutics) for treatment-resistant schizophrenia were covered by public plans. As clinical expert opinion elicited by the sponsor indicated that patients eligible for risperidone ER would be ||||||| to have access to private insurance plans, an assumption of 80% public coverage was made.63 However, according to the IQVIA Pharmastat Plus database,51 approximately 85% of claims in 2020 for aripiprazole LAI and paliperidone LAI either monthly or quarterly were publicly reimbursed, while 92% of risperidone LAI claims were publicly reimbursed. Therefore, in patient with schizophrenia, there is uncertainty around the proportion of claims for risperidone ER that will be covered by public drug plans.
CADTH reanalysis assumed that 85% of claims for the included long-acting injectables would be reimbursed by public drug plans.
Confidential pricing agreements: While the submitted price of the every 2 weeks risperidone LAI is similar to the publicly available list prices of other LAI AAPs, as indicated in the Provincial Advisory Group input submitted for this review (see drug program input in the Stakeholder Perspectives section), confidential pricing agreements exist which lower the annual cost of the comparator products to the jurisdictional drug plans. The submitted price of risperidone ER would thus also have to be lowered to avoid incurring additional costs relative to its comparators.
CADTH explored reanalysis scenarios where all comparators were 10% and 25% less expensive than estimated by the sponsor.
Poor reporting: The submitted BIA report conflicted with the submitted BIA model in a number of instances, and lacked sufficient detail to explain methodology, sources, and assumptions used within the BIA model. For example, while the BIA report states that the number of patients within each comparator and year of the analysis was retrieved from an IQVIA database, the IQVIA database in fact estimated the number of units which were sold, and the sponsor estimated the number of patients by dividing the reported number of units by the recommended dose frequency for each comparator.
Uncertainty in the methodology and rationale used increases uncertainty in the BIA results.
CADTH Reanalyses of the Budget Impact Analysis
CADTH revised the sponsor’s base case by adjusting annual comparator growth rate based on 3-years of prior data, removing additional dispensing fees and markups from the IQVIA CompuScript sales data, and increasing the proportion of claims reimbursed by public drug plans. Table 17 outlines the parameters used by the sponsor in comparison to those used by CADTH.
Table 17: CADTH Revisions to the Submitted Budget Impact Analysis
Stepped analysis | Sponsor’s value or assumption | CADTH value or assumption |
|---|---|---|
Corrections to sponsor’s base case | ||
None | None | None |
Changes to derive the CADTH base case | ||
1. Annual growth of comparators based on previous 3 years | Aripiprazole (Abilify Maintena) 18.9% Paliperidone (Invega Sustenna) 8.3% Risperidone (Risperdal Consta) −8.1% Overall 9.7% | Aripiprazole (Abilify Maintena) 17.9% Paliperidone (Invega Sustenna) 4.6% Risperidone (Risperdal Consta) −8.4% Overall 7.2% |
2. Extra fees and markups removed | Fees and markups added to CompuScript sales data for comparators | Fees and markups only added to risperidone ER estimates as CompuScript data for comparators already included them |
3. Proportion of claims reimbursed by public plans | 80% | 85% |
CADTH base case | 1 + 2 + 3 | |
ER = extended release.
Applying these changes resulted in an updated BIA with budgetary savings of $1,171 in year 1, $32,179 in year 2, and $331,555 in year 3, for a 3-year cumulative budgetary savings of $298,205. The results of the CADTH step-wise reanalysis are presented in summary format in Table 18.
Additionally, CADTH included scenarios decreasing the proportion of patients using the 90 mg monthly strength of risperidone ER and reducing the cost of comparators to reflect uncertainty in the actual costs paid by public drug plans. The incremental budgetary savings associated with the reimbursement of risperidone ER are eliminated when fewer than 28% of patients use the 90 mg dose, or if all comparator costs are reduced by 1.9%.
Table 18: Summary of the CADTH Reanalyses of the Budget Impact Analysis
Stepped analysis | Three-year total ($) |
|---|---|
Submitted base case | −2,004,121 |
CADTH reanalysis 1: Annual growth rate based on past 3 years | −1,770,074 |
CADTH reanalysis 2: Extra fees and markups removed | −496,385 |
CADTH reanalysis 3: 85% public coverage | −2,018,091 |
CADTH base case | −298,205 |
CADTH Scenario A: Only 25% of patients use lower risperidone ER dose | 126,946 |
CADTH Scenario B: All comparators cost 10% less | 1,312,779 |
CADTH Scenario C: All comparators cost 25% less | 3,729,256 |
ER = extended release.
Appendix 4: Additional Information on EQ-5D-5L, SWN-S, and MSQ Scales
Note that this appendix has not been copy-edited.
In the pivotal phase III study (Study 09-0010), the tertiary objectives were to assess HRQoL, satisfaction with medication, and medicine preference. HRQoL was measured using the EQ-5D-5L, subjective well-being using the SWN-S, patient satisfaction with their medication using the MSQ, and patient and caregiver medication preference (if the caregiver was available) were measured using the Preference of Medicine (POM) questionnaire. The instruments to measure these aspects were administered to patients prior to clinical assessments at screening, baseline, and end of treatment. All questionnaires have been studied psychometrically; their measurement properties have been well published and determined to be appropriate for use in the schizophrenia setting.
The duration of the phase III (Study 09-0010) efficacy study (8 weeks) in schizophrenia was of sufficient duration to observe HRQoL; the study showed significantly greater improvements in HRQoL and overall well-being in patients randomized to risperidone ER compared to placebo. The results reported by Isitt et al.39 show that the HRQoL effect was more pronounced in the risperidone ER 120 mg group. In addition, patient satisfaction improved significantly and patient preference for their medicine favoured risperidone ER 90 mg and 120 mg versus placebo.39
It is further emphasized that 8 weeks is the generally accepted duration for a pivotal trial in schizophrenia for evaluating safety, and is well within the recommendation for such trials, given the ethical need to consider treatment of patients enrolled in the placebo group.65 Furthermore, The onset of an effect of risperidone ER on the reduction in PANSS total scores and CGI-S scores is evident as early as 2 weeks after the first injection.
EQ-5D-5L Results
The EQ-5D-5L VAS increased significantly in the risperidone ER 120 mg group compared to placebo (P = 0.0212).
SWN-S Results
In risperidone ER 120 mg, patients reported significant improvements in SWN-S physical functioning (P = 0.0093), social integration (P = 0.0368), and total score (P = 0.0395).
MSQ Results
Patients receiving risperidone ER reported significant increases in medication satisfaction over the course of the trial. Subject satisfaction with their medication at screening was 28.8% in risperidone ER 90 mg versus 33.9% in placebo, at week 4 improved to 64.0% in risperidone ER mg versus 54.5% in placebo, and at EOS improved to 73.0% in risperidone ER compared to only 50.9% in Placebo (p = 0.0009).
The relationship of patients' satisfaction in risperidone ER 120 mg was similar to that of risperidone ER 90 mg however, the proportion of patients satisfied with their medication by week 4 increased by a larger proportion (69.3% in risperidone ER 120 mg versus 54.5% in placebo [P = 0.0125]). At EOS the increase in medication satisfaction in the risperidone ER 120 mg group continued to 73.77% versus 50.9% in Placebo (P = 0.0006).
POM Results
In the POM dichotomous analysis, 33% of placebo patients rated their POM as “Slightly Better” or “Much Better” compared with 63.1% for risperidone ER 90 mg (p b 0.0001) and 45.6% for risperidone ER 120 mg (P = 0.0619), respectively. In contrast, 59.8% of placebo patients rated their POM as “About the same or worse” compared with 32.4% for risperidone ER 90 mg and 51.8% for risperidone ER 120 mg, respectively.
The authors conclude that significantly greater improvements in HRQoL and overall well-being were demonstrated in patients randomized to risperidone ER compared with placebo. The effect was more pronounced in the risperidone ER 120 mg group. Patient satisfaction improved significantly and patient preference for their medicine favoured risperidone ER 90 mg and 120 mg versus placebo.39
Appendix 5: Findings of Study 13-0005
Note that this appendix has not been copy-edited.
Study 13-0005 was a phase III, open-label, long-term safety and tolerability study of risperidone ER SC injections in patients diagnosed with schizophrenia (DSM-IV-TR) with a PANSS score of ≤ 70 (de novo patients) and in patients who completed the double-blind, placebo-controlled, efficacy Study 09-0010, conducted in patients with acute schizophrenia (“rollover” patients). All patients were assigned the monthly 120-mg dose of risperidone ER, which was subject to a one-time down-titration to 90 mg Perseris for tolerability, at the investigator’s discretion.
The primary objective of this study was to assess the long-term safety and tolerability of risperidone ER SC injections in patients with schizophrenia.
The secondary objective of this study was to continue collecting clinical outcome data with risperidone ER SC injections in patients with schizophrenia using the PANSS and CGI-S scale.
The tertiary objective of this study was to continue collecting health economic and patient-reported outcome data with Perseris SC injections in patients with schizophrenia using the EQ-5D-5L, Short Form (36) Health Survey version 2 (SF-36v2), SWN-S, MSQ, POM, and health care resource utilization (HCRU).
PANSS Scores24,44
Results of this trial show that in patients receiving Perseris 120 mg administered monthly over 12 months, mean PANSS scores remained stable among de novo participants (mean [standard deviation] [SD] change from baseline to EOS of −0.4 [SD = 8.7] and continued to improve in rollover participants: the rollover placebo group had mean (± SD) values of 72.9 (± 20.99) at baseline and 59.2 (± 20.14) at EOS. The rollover risperidone ER 90-mg group had mean (± SD) values of 76.4 (± 15.99) at baseline and 60.9 (± 12.04) at EOS. The rollover risperidone ER 120 mg group had mean (± SD) values of 71.0 (± 13.93) at baseline and 59.9 (± 13.23) at EOS.
CGI-S Scores24,44
For both de novo patients and rollover patients, CGI-S scores remained stable throughout the study with mean (± SD) values of 3.4 (± 0.71) at baseline and 3.2 (± 0.66) at EOS for all patients combined.
Health Economics and Outcomes Research Results66
Results from the overall analyses of the EQ-5D-5L, SF-36v2, and SWN-S remained stable from active baseline to EOS. Risperidone ER sustained patients’ well-being, as measured by the SWN-S, which is suggestive of consistent adherence to medication.
Approximately 81% of the patients reported satisfaction with the treatment at EOS compared with 66% at baseline. Along with satisfaction with treatment, patients also reported a strong preference to the treatment, as measured by the MSQ and POM, with 72% of the patients preferring current medication to their most recent pre-study antipsychotic.
The high Physical Component Summary and Mental Component Summary scores and low psychiatric re-admission rate of 5% is indicative of good long-term stability in patients using Perseris. Study patients also recorded a lower number of emergency room visits and hospital admissions, and shorter length of stay compared with what is reported in literature on patients with schizophrenia.
This study also attests to the long-term sustainability of improvements seen in HRQoL after 8 weeks of treatment with risperidone ER in the previously completed double-blind study.
Safety
Risperidone ER 90 mg and 120 mg were demonstrated to be safe and well tolerated when administered up to 1 year to individuals with schizophrenia. There were no new safety signals compared with the known safety profile of oral risperidone products, except for the expected local injection-site reactions that are observed with all LAI antipsychotic drugs. Local injection-site reactions were not assessed as serious by the investigator and led to discontinuation in only 2 patients across the entire development program.44
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up-to-date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third-party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.