CADTH Health Technology Review
Intranasal Glucagon for the Treatment of Hypoglycemia
Rapid Review
Authors: Khai Tran, Danielle MacDougall
Abbreviations
AE
adverse event
CI
confidence interval
IM
intramuscular
IN
intranasal
MA
meta-analysis
OR
odds ratio
RCT
randomized controlled trial
SC
subcutaneous
SR
systematic review
Key Messages
Intranasal glucagon was equally effective compared with glucagon given by intramuscular or subcutaneous injection to treat experimentally insulin-induced hypoglycemia in both adults and children with type 1 diabetes.
The overall rates of adverse events were similar between groups treated with intranasal glucagon or intramuscular or subcutaneous glucagon. Nausea and vomiting were more frequent in the groups treated with intramuscular or subcutaneous glucagon, whereas eye and nose symptoms occurred more frequently in the intranasal glucagon group.
There was no evidence identified that compared the clinical effectiveness of intranasal glucagon with placebo or no treatment in people treated with insulin who received treatment for hypoglycemia.
No evidence was found about the cost-effectiveness of intranasal glucagon and no evidence-based guidelines were found about the use of intranasal glucagon for the treatment of hypoglycemia in people who receive treatment with insulin.
Context and Policy Issues
Hypoglycemia is a condition in which the blood glucose level is less than 70 mg/dL (3.9 mmol/L).1 There are 3 levels of hypoglycemia severity.2 Level 1 hypoglycemia is defined as a blood glucose level between 54 mg/dL and 70 mg/dL, level 2 hypoglycemia is defined as a blood glucose level less than 54 mg/dL, and level 3 is severe hypoglycemia with cognitive impairment requiring external assistance for recovery.2 The main causes of hypoglycemia are drugs used to control diabetes, such as insulin, or agents that promote the secretion of insulin.3 Symptoms of hypoglycemia can be mild, moderate, or severe.1 Untreated hypoglycemia can lead to seizures, coma, and death.1 People with mild or moderate symptoms of hypoglycemia, or conscious individuals with severe hypoglycemia, can be treated with oral ingestion of carbohydrates.3 Individuals with severe hypoglycemia, who may not be able to ingest carbohydrates due to disorientation or unconsciousness, need external support to administer glucose or glucagon.1
Glucagon is a hormone secreted by the alpha cells of the pancreas in response to hypoglycemia which increases blood glucose levels by breaking down glycogen or through glucose synthesis.4 However, individuals with diabetes have a deficient glucagon secretory response to insulin-induced hypoglycemia.4 Thus, exogenous glucagon is the preferred treatment for severe hypoglycemia.3 Glucagon is generally available as intravascular, intramuscular (IM), and subcutaneous (SC) injection for the treatment of severe hypoglycemia in individuals with diabetes.3 The Canadian Diabetes Association recommends 1 mg of IM or SC glucagon be prescribed to resolve hypoglycemia in individuals with diabetes who are unconscious.3 However, administration of IM or SC glucagon is complex, and a trained caregiver is needed to reconstitute the glucagon from a dry powder form because glucagon is not stable in its solution form.5
Intranasal (IN) formulation of glucagon (Baqsimi, 3 mg) has recently become available and has been approved by Health Canada for the treatment of severe hypoglycemia in children (4 years and older) and adults with diabetes.6 Recently, CADTH produced 2 formulary review reports on the clinical effectiveness7 and cost-effectiveness8 of glucagon nasal powder (Baqsimi) for the treatment of severe hypoglycemia in individuals with diabetes receiving treatment with insulin.
The aim of this report is to summarize new evidence published since the previous CADTH formulary reviews7,8 about the clinical effectiveness and cost-effectiveness of IN glucagon compared with placebo, no treatment, or alternative interventions for the treatment of hypoglycemia in people receiving treatment with insulin. Additionally, this report aims to summarize recommendations from evidence-based guidelines regarding the use of IN glucagon for hypoglycemia in people receiving treatment with insulin.
Research Questions
What is the clinical effectiveness of IN glucagon versus placebo or no treatment for the treatment of hypoglycemia in people who receive treatment with insulin?
What is the clinical effectiveness of IN glucagon versus alternative interventions for the treatment of hypoglycemia in people who receive treatment with insulin?
What is the cost-effectiveness of IN glucagon versus placebo or no treatment for the treatment of hypoglycemia in people who receive treatment with insulin?
What is the cost-effectiveness of IN glucagon versus alternative interventions for the treatment of hypoglycemia in people who receive treatment with insulin?
What are the evidence-based guidelines regarding the use of IN glucagon for the treatment of hypoglycemia in people who receive treatment with insulin?
Methods
Literature Search Methods
The literature search strategy used in this report is an update of 1 developed for a previous CADTH report.7 For the current report, a limited literature search was conducted by an information specialist on key resources including MEDLINE, Embase, the Cochrane Database of Systematic Reviews, the International HTA Database, Canadian and major international health technology agencies, as well as a focused internet search. No filters were applied to limit the retrieval by study type. The initial search was not limited by language or publication dates. For the current report, database searches were rerun on May 11, 2022, to capture any articles published or made available since the initial search date. The search of major health technology agencies was also updated to include documents published since August 2019.
Selection Criteria and Methods
One reviewer screened citations and selected studies. In the first level of screening, titles and abstracts were reviewed and potentially relevant articles were retrieved and assessed for inclusion. The final selection of full-text articles was based on the inclusion criteria presented in Table 1.
Criteria | Description |
|---|---|
Population | People of any age who receive treatment with insulin |
Intervention | Glucagon nasal powder |
Comparator | Q1 and Q3: placebo, no treatment Q2 and Q4: alternative interventions (i.e., glucagon [subcutaneous, intramuscular], intravascular dextrose, oral glucose, oral carbohydrates) Q5: not applicable |
Outcomes | Q1 and Q2: clinical effectiveness (e.g., resolution of hypoglycemic episodes, time to dose administration, quality of life, safety [e.g., adverse events]) Q3 and Q4: cost-effectiveness (e.g., cost per quality-adjust life-year gained, incremental cost-effectiveness ratios) Q5: recommendations regarding the use of intranasal glucagon for the treatment of hypoglycemic episodes |
Study designs | Health technology assessments, systematic reviews, randomized controlled trials, non-randomized studies, economic evaluations, evidence-based guidelines |
Q = question.
Exclusion Criteria
Articles were excluded if they did not meet the selection criteria outlined in Table 1, they were duplicate publications, or were published before 2019. Primary studies retrieved by the search were excluded if they were included in the previous CADTH report.7 Systematic review with all included studies found in the previous CADTH report7 was also excluded.
Critical Appraisal of Individual Studies
The included publication was critically appraised by 1 reviewer using A MeaSurement Tool to Assess Systematic Reviews 2 (AMSTAR 2).9 Summary scores were not calculated; rather, the strengths and limitations of the systematic review were described narratively.
Summary of Evidence
Quantity of Research Available
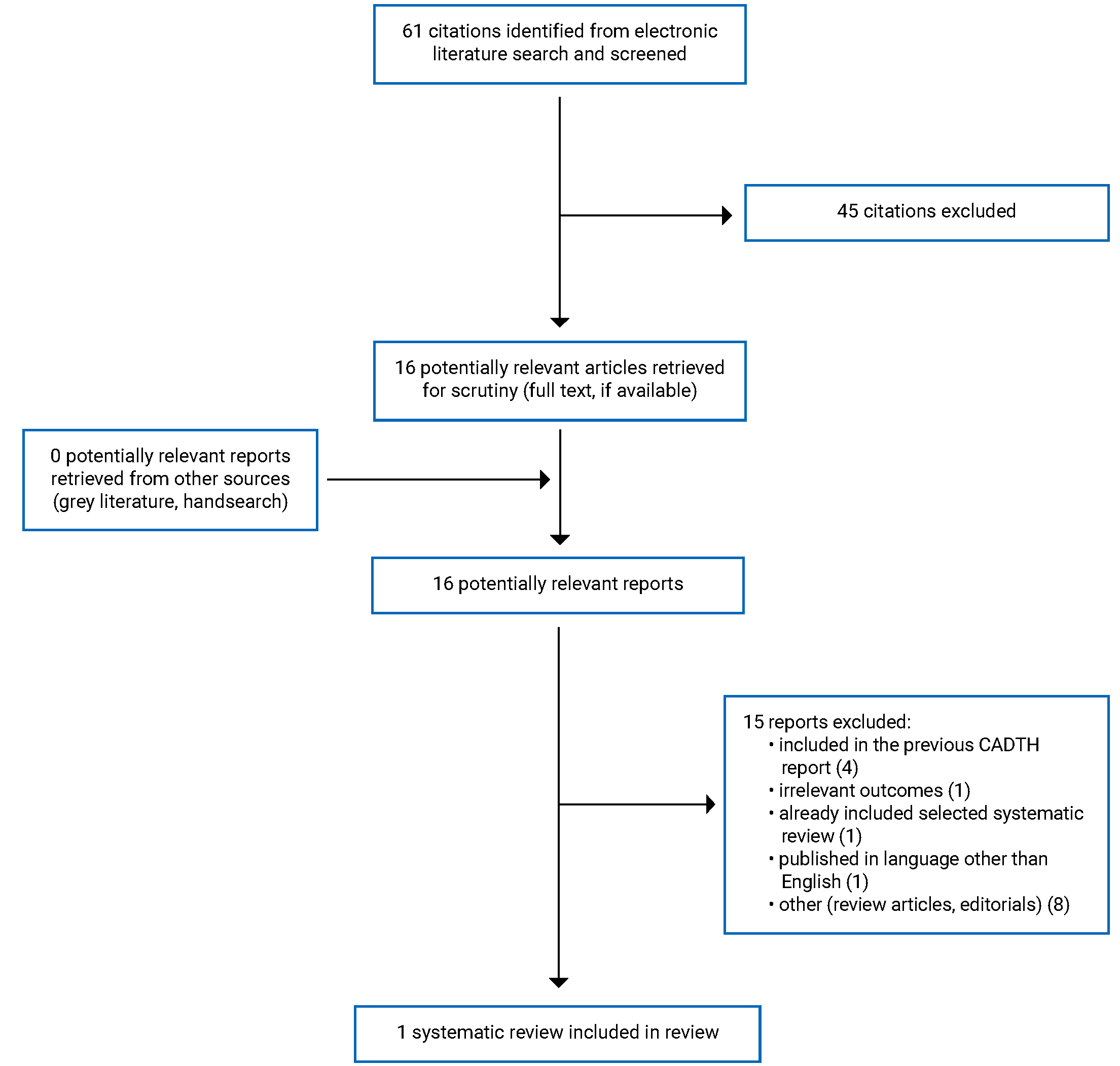
A total of 61 citations were identified in the literature search. Following screening of titles and abstracts, 45 citations were excluded and 16 potentially relevant reports from the electronic search were retrieved for full-text review. No potentially relevant publications were retrieved from the grey literature search for full-text review. Of 16 potentially relevant articles, 15 publications were excluded for various reasons, and 1 systematic review (SR) with a meta-analysis (MA) met the inclusion criteria and was included in this report. Appendix 1 presents the PRISMA10 flow chart of the study selection. Additional references of potential interest are provided in Appendix 5.
Summary of Study Characteristics
The detailed characteristics of the included SR11 are provided in Appendix 2 (Table 2).
Study Design
The SR by Pontiroli and Tagliabue (2020)11 included data of 8 randomized controlled trials (RCTs) in an MA. Two of the trials had a parallel design and 6 were crossover studies. The RCTs were published between 1989 and 2019. All trials were conducted in hospital settings.
Country of Origin
The SR was conducted by authors from Italy.11
Patient Population
Patient populations in the included studies of the SR11 were either adults or children with type 1 diabetes. The mean age of the adults ranged between 30 years to 35 years, and the mean age of the children ranged between 9 years to 13 years. The mean blood glucose levels of the adults at baseline ranged between 36 mg/dL to 72 mg/dL, and the mean blood glucose levels of the children at baseline ranged between 38 mg/dL to 59 mg/dL.
Interventions and Comparators
The SR11 included RCTs comparing IN glucagon with IM or SC glucagon in adults or children with hypoglycemia. Hypoglycemia was not defined in the SR. Seven of the included RCTs had hypoglycemia that was intentionally induced by insulin injection; 1 RCT evaluated inpatients with hypoglycemia when 2 conditions were simultaneously present (i.e., glucose levels < 70 mg/dL and subjective feeling of hypoglycemia).
Outcomes
The outcomes considered in the SR11 were response rate to glucagon (i.e., increase in blood glucose) and adverse events (AEs). The level of increase in glucose that represented a response to glucagon treatment was not defined.
Summary of Critical Appraisal
Additional details regarding the strengths and limitations of the included SR11 are provided in Appendix 3 (Table 3).
The SR11 clearly described its objective, inclusion criteria, and selection of the study designs. The review authors partially used a comprehensive literature search strategy (used at least 2 databases, provided keywords and search strategy). The authors did not report if they searched the reference lists or bibliographies of included studies, searched the trial registries, searched for grey literature, or included content experts in the field. Details of the literature search strategy were not provided, which limits the reproducibility of the review. The report did not state if a research protocol was established before conducting the review, without which there is a potentially increased risk of reporting bias. The review authors did not report whether study selection and data extraction were performed in duplicate. Therefore, it is unclear whether a fully systematic approach was taken in study selection and data extraction. Performing these activities in duplicate reduces the likelihood of missing relevant studies and making errors in data extraction. The SR did not provide a list of excluded studies, the reasons for exclusion, or the sources of funding for the included studies. Not having a list of excluded studies or the justification for their exclusion makes it impossible to assess whether any relevant articles were excluded or the reasons that was done.
The characteristics of the included studies were not described in adequate detail. Providing sufficient details of the included studies allows assessing the heterogeneity among the studies and whether subgroup analyses are needed to explore the sources of heterogeneity. However, the authors of the review performed a meta-regression analysis with covariates such as number of participants in each study, age, and fasting glucose level before administration of glucagon. The authors of the SR assessed the methodological quality of the included studies using the risk-of-bias assessment tool for nonrandomized studies (RoBANs) tool and evaluated publication bias using the funnel plot method. However, RoBANs may not be appropriate for RCTs because it was designed for nonrandomized studies. For the MA, the review authors used appropriate methods for statistical combination of the results. Treatment outcomes were expressed as odds ratios (ORs) with 95% confidence intervals (CIs) and were pooled into an overall OR using a random-effects model. The review authors did not assess the potential impact of the risk of bias in individual studies on the results of the MA. However, most of the included studies had no or low risk of bias, according to the authors’ assessment. The review authors declared that they had no conflicts of interest, and the study was supported by a grant from the Ministry of Health in Italy. Overall, the methodological quality of the included SR was limited.
Summary of Findings
Appendix 4 presents the main study findings of the SR.11 The findings are presented by outcome, which are response rate (Table 4) and AEs (Table 5).
Clinical Effectiveness of IN Glucagon Versus Placebo or No Treatment for the Treatment of Hypoglycemia in People Who Receive Treatment With Insulin
No studies comparing IN glucagon versus placebo or no treatment for the treatment of hypoglycemia in people who receive treatment with insulin were identified; therefore, no summary can be provided.
Clinical Effectiveness of IN Glucagon Versus Alternative Interventions for the Treatment of Hypoglycemia in People Who Receive Treatment With Insulin
One SR11 was identified comparing IN glucagon with IM or SC glucagon in adults and children with type 1 diabetes.
Response Rate
The SR11 included data of 8 RCTs in an MA investigating the response rate of IN glucagon versus IM or SC glucagon in adults and children with type 1 diabetes. Of 8 RCTs, 4 reported some failures in both groups, and the pooled OR for response was 0.61 (95% CI, 0.13 to 2.82; P = 0.96; I2 = 30%), showing no significant difference between the IN glucagon and IM or SC glucagon groups. Because the remaining 4 studies reported 100% effectiveness in both groups (i.e., failures were null with both treatments), a simulation was made by adding 1 failure for each arm to allow for the calculation of ORs. A pooled analysis of all 8 RCTs again showed no significant difference in response rate between the IN glucagon and IM or SC glucagon groups (OR = 0.80; 95% CI, 0.28 to 2.32; P = 1.00; I2 = 47%).
Meta-regression analysis showed that the effect of IN and IM or SC glucagon was independent on the size of the study, age of participants, and blood glucose levels at baseline. Subgroup analysis revealed no statistically significant differences in the response rates between IN and IM or SC glucagon among adult participants (OR = 0.80; 95% CI, 0.18 to 3.45) or among children (OR = 0.76; 95% CI, 0.13 to 4.32).
Adverse Events
The authors of the review stated that a systematic analysis of AEs was not possible because the included studies had different ways of reporting AEs. Incidence of nausea and vomiting was lower in the IN group than the IM or SC glucagon group. Local side effects, such as eye symptoms and nasal symptoms, were more frequent in the IN group than in the IM or SC glucagon group. Other side effects were similar with both groups.
Cost-Effectiveness of IN Glucagon Versus Placebo or No Treatment for the Treatment of Hypoglycemia in People Who Receive Treatment With Insulin
No studies comparing the cost-effectiveness of IN glucagon versus placebo or no treatment for the treatment of hypoglycemia in people who receive treatment with insulin were identified; therefore, no summary can be provided.
Cost-Effectiveness of IN Glucagon Versus Alternative Interventions for the Treatment of Hypoglycemia in People Who Receive Treatment With Insulin
No studies comparing the cost-effectiveness of IN glucagon versus alternative interventions for the treatment of hypoglycemia in people who receive treatment with insulin were identified; therefore, no summary can be provided.
Guidelines
No evidence-based guidelines regarding the use of IN glucagon for the treatment of hypoglycemia in people who receive treatment with insulin; therefore, no summary of recommendations can be provided.
Limitations
The included SR11 had several limitations. First, 5 of 8 trials had small sample sizes, ranging from 3 to 15, and most studies were conducted from few centres. The second limitation was that 7 of 8 trials used insulin to artificially induce and control hypoglycemia, but not severe hypoglycemia. Hypoglycemia induction at the study site might not represent hypoglycemia in the real world, where people may be disoriented or unconscious when they experience severe hypoglycemia. Therefore, the evidence from the SR on the use of IN glucagon for the treatment of hypoglycemia may have limited generalizability to a real-world population. The third limitation was that most of the included studies were crossover in design and lacked study blinding, meaning the possibility of performance and detection biases could not be ruled out. The fourth limitation was that this SR only focused on studies that included individuals with type 1 diabetes, making it unclear if IN glucagon is effective in individuals with type 2 diabetes. However, a Japanese study12 that was included in the previous CADTH report7 showed that IN glucagon was noninferior to IM glucagon for successful treatment of insulin-induced hypoglycemia in individuals with either type 1 or type 2 diabetes. The fifth limitation of this SR was that the AEs in the IN glucagon groups were not systematically investigated or clearly reported. A summary of AEs for treatment with IN glucagon compared with IM glucagon is available in the previous CADTH report.7
This report could not identify any studies that compared IN glucagon with placebo or no treatment, nor any studies on cost-effectiveness of IN glucagon. Also, no evidence-based guidelines were identified on the use of IN glucagon for the treatment of hypoglycemia in people who receive treatment with insulin.
Conclusions and Implications for Decision- or Policy-Making
This report identified 1 SR11 that compared IN glucagon with injectable (IM or SC) glucagon for treatment of hypoglycemia in people with type 1 diabetes.
The findings of the SR showed that the use of IN glucagon for treatment of experimentally insulin-induced hypoglycemia had a similar response rate compared with IM or SC glucagon in conscious populations of both adults and children with type 1 diabetes. The overall incidence of AEs was similar between IN and IM or IS glucagon groups. Nausea and vomiting were less frequent with IN glucagon treatment, whereas local side effects such as eye and nasal symptoms were more frequent with IN glucagon treatment compared with IM or SC glucagon treatment. The clinical effectiveness of IN glucagon relative to IM or SC glucagon may have limited generalizability in real-world situations where people with severe hypoglycemia may require external assistance for recovery. Well-designed studies are needed to investigate the effectiveness of IN glucagon in real-world conditions, including emergency situations. Future trials focusing on the elderly population with type 1 or type 2 diabetes who have high risk of severe hypoglycemia is also needed. There is a similar need for economic evaluations investigating the cost-effectiveness of IN glucagon for treatment of hypoglycemia in people with diabetes treated with insulin.
References
1.American Diabetes Association. Hypoglycemia (low blood sugar). (Blood glucose testing and management) 2022; https://www.diabetes.org/healthy-living/medication-treatments/blood-glucose-testing-and-control/hypoglycemia. Accessed 2022 May 18.
2.Members of the International Hypoglycaemia Study Group. Glucose Concentrations of Less Than 3.0 mmol/L (54 mg/dL) Should Be Reported in Clinical Trials: A Joint Position Statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care. 2017;40(1):155-157. PubMed
3.Yale JF, Paty B, Senior PA. Hypoglycemia. Can J Diabetes. 2018;42 Suppl 1:S104-s108. PubMed
4.Fanelli CG, Porcellati F, Rossetti P, Bolli GB. Glucagon: the effects of its excess and deficiency on insulin action. Nutr Metab Cardiovasc Dis. 2006;16 Suppl 1:S28-34. PubMed
5.Gelenter MD, Smith KJ, Liao SY, et al. The peptide hormone glucagon forms amyloid fibrils with two coexisting beta-strand conformations. Nat Struct Mol Biol. 2019;26(7):592-598. PubMed
6.Baqsimi (glucagon): 3 mg per device, intranasal powder [product mongraph]. Toronto (ON): Eli Lilly Canada Inc.; 2021 Sep 28: https://pi.lilly.com/ca/baqsimi-ca-pm.pdf. Accessed 2022 May 19.
7.Drug Reimbursement Review clinical guidance report: glucagon nasal powder (Baqsimi) for the treatment of severe hypoglycemic reactions which may occur in management of insulin treated patients with diabetes mellitus, when impaired consciousness precludes oral carbohydrates. Ottawa (ON): CADTH; 2020: https://www.cadth.ca/sites/default/files/cdr/clinical/sr0626-baqsimi-clinical-review-report.pdf. Accessed 2022 May 16.
8.Drug Reimbursement Review pharmacoeconomic report: glucagon nasal powder (Baqsimi) for the treatment of severe hypoglycemic reactions which may occur in management of insulin treated patients with diabetes mellitus, when impaired consciousness precludes oral carbohydrates. Ottawa (ON): CADTH; 2020: https://www.cadth.ca/sites/default/files/cdr/pharmacoeconomic/sr0626-baqsimi-pharmacoeconomic-review-report.pdf. Accessed 2022 May 16.
9.Shea BJ, Reeves BC, Wells G, et al. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ. 2017;358:j4008. PubMed
10.Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol. 2009;62(10):e1-e34. PubMed
11.Pontiroli AE, Tagliabue E. Intranasal versus injectable glucagon for hypoglycemia in type 1 diabetes: systematic review and meta-analysis. Acta Diabetol. 2020;57(6):743-749. PubMed
12.Matsuhisa M, Takita Y, Nasu R, Nagai Y, Ohwaki K, Nagashima H. Nasal glucagon as a viable alternative for treating insulin-induced hypoglycaemia in Japanese patients with type 1 or type 2 diabetes: A phase 3 randomized crossover study. Diabetes Obes Metab. 2020;22(7):1167-1175. PubMed
Appendix 1: Selection of Included Studies
Appendix 2: Characteristics of Included Publications
Note that this appendix has not been copy-edited.
Table 2: Characteristics of the Included Systematic Review
Study citation, country, funding source | Study designs and numbers of primary studies included | Population characteristics | Intervention and comparator(s) | Clinical outcomes, length of follow-up |
|---|---|---|---|---|
Pontiroli and Tagliabue (2020)11 Italy Funding: from Ministry of Health, Italy | SR with MA Total: 8 RCTs (N = 285) included in MA Year of publication: 1989 to 2019 Sample size: 3 to 75 Quality assessment tool: RoBANs. | Adults and children with type I diabetes Mean age of adults: 30 years to 35 years Mean age of children: 9 years to 13 years Gender or sex: NR Mean blood glucose of adults at baseline: 36 mg/dl to 72 mg/dl Mean blood glucose of adults at baseline: 38 mg/dl to 59 mg/dl | IN glucagon (N = 254) vs. IM/IS glucagon (N = 213)a Seven studies had hypoglycemia that was intentionally induced by insulin injection; one study evaluated inpatients having hypoglycemia when 2 conditions simultaneously present: glucose levels < 70 mg/dl and subjective feeling of hypoglycemia. | Outcomes:
Follow-up: NR |
AE = adverse event; IM = intramuscular; IN = intranasal; MA = meta-analysis; NR = not reported; OR = odds ratio; RCT = randomized controlled trial; RoBANs = risk-of-bias assessment tool for nonrandomized studies; SC = subcutaneous; SR = systematic review.
aSix trials were crossover in design. The numbers of participants in those trials were counted twice.
bFour trials had 100% effective (i.e., no failure). A simulation was made by adding 1 failure for each arm to calculate ORs.
Appendix 3: Critical Appraisal of Included Publications
Note that this appendix has not been copy-edited.
Table 3: Strengths and Limitations of the Systematic Review Using AMSTAR 29
Strengths | Limitations |
|---|---|
Pontiroli and Tagliabue (2020)11 | |
The research question or objective and the inclusion criteria for the review clearly include the components of PICO. For meta-analysis, the review authors use appropriate methods for statistical combination of results. Treatment outcomes were expressed as ORs with 95% CI and pooled into an overall OR using a random-effects model. The review authors provided a discussion of the heterogeneity observed in the results. A meta-regression analysis was performed to explore the potential effect of patients of study characteristics on the pooled estimate of efficacy. The covariates considered for the meta-regression analysis were number of participants of each study, age, and fasting of glucose at baseline. As most of the included studies had no or low RoB, according to the authors’ assessment, assessment of the potential impact of RoB in individual studies on the results of the meta-analysis was not performed. The review authors evaluated the existence of a potential publication bias using the Funnel plot technique. The review authors reported the source of funding (i.e., Ministry of Health, Italy) and declared that there were no conflicts of interest. | The report of the review did not contain any statement indicating the review methods were established before the conduct of the review. The review authors partially used a comprehensive literature search strategy. The review authors did not report whether study selection and data extraction were performed in duplicate. Therefore, it is unclear whether a fully systematic approach was taken in study selection and data extraction. A list of excluded studies and the reasons for exclusion were not provided. Therefore, it was not possible to assess whether any relevant articles were excluded and if so, for what reasons. The characteristics of the included studies were not described in adequate detail. Although the quality of the reports and RoB were assessed using RoBANs, the tool was designed to assess to RoB of nonrandomized studies. The review authors did not report the sources of funding for the included studies. This is potentially a concern because funding received from industry can introduce bias in favour of the intervention. |
AMSTAR 2 = A MeaSurement Tool to Assess systematic Reviews 2; CI = confidence interval; OR = odds ratio; PICO = population, intervention, comparator, and outcome; RoB = risk of bias; RoBANs = risk-of-bias assessment tool for nonrandomized studies.
Appendix 4: Main Study Findings by Outcomes
Note that this appendix has not been copy-edited.
Table 4: Summary of Findings by Outcome — Response Rate
Study citation, study design, and patient model | Study findings |
|---|---|
IN glucagon vs. IM or SC glucagon | |
Pontiroli and Tagliabue (2020)11 SR with MA Adults and children with type I diabetes | Analysis derived from published data (total 4 RCTs in the analysis): OR (95% CI) = 0.61 (0.13 to 2.82); P = 0.960; I2 = 30% A simulation analysis was made by adding 1 failure for each arm for the remaining 4 RCTs (total 8 RCTs in the analysis): OR (95% CI) = 0.80 (0.28 to 2.32); P = 1.00; I2 = 47% Meta-regression analysis:
Subgroup analysis:
|
CI = confidence interval; IM = intramuscular; IN = intranasal; MA = meta-analysis; OR = odds ratio; RCT = randomized controlled trial; SC = subcutaneous; SR = systematic review; vs. = versus.
Table 5: Summary of Findings by Outcome — Adverse Events
Study citation, study design, and patient model | Study findings |
|---|---|
IN glucagon vs. IM or SC glucagon | |
Pontiroli and Tagliabue (2020)11 SR with MA Adults and children with type I diabetes | Incidence of nausea, vomiting, and headache was lower with IN than with IM or SC glucagon. Local side effects such as nasal irritation and ocular lacrimation were more frequent with IN glucagon. |
IM = intramuscular; IN = intranasal; MA = meta-analysis; SC = subcutaneous; SR = systematic review; vs. = versus.
Appendix 5: References of Potential Interest
Note that this appendix has not been copy-edited.
Previous CADTH Reports
Drug Reimbursement Review clinical guidance report: glucagon nasal powder (Baqsimi) for the treatment of severe hypoglycemic reactions which may occur in management of insulin treated patients with diabetes mellitus, when impaired consciousness precludes oral carbohydrates. Ottawa (ON): CADTH; 2020: https://www.cadth.ca/sites/default/files/cdr/clinical/sr0626-baqsimi-clinical-review-report.pdf. Accessed 2022 May 16.
Drug Reimbursement Review pharmacoeconomic report: glucagon nasal powder (Baqsimi) for the treatment of severe hypoglycemic reactions which may occur in management of insulin treated patients with diabetes mellitus, when impaired consciousness precludes oral carbohydrates. Ottawa (ON): CADTH; 2020: https://www.cadth.ca/sites/default/files/cdr/pharmacoeconomic/sr0626-baqsimi-pharmacoeconomic-review-report.pdf. Accessed 2022 May 16.
Additional References
Osumili B, Artime E, Mitchell B, et al. Cost of Severe Hypoglycemia and Budget Impact with Nasal Glucagon in Patients with Diabetes in Spain. Diabetes Ther. 2022;13(4):775-794. PubMed
Singh-Franco D, Moreau C, Levin AD, Rosa D, Johnson M. Efficacy and Usability of Intranasal Glucagon for the Management of Hypoglycemia in Patients With Diabetes: A Systematic Review. Clin Ther. 2020;42(9):e177-e208. PubMed
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up-to-date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third-party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.
Questions or requests for information about this report can be directed to Requests@CADTH.ca