CADTH Health Technology Review
Cost-Effectiveness of Treatment for Pediatric Immune Thrombocytopenia
Rapid Review
Authors: Khai Tran, Mê-Linh Lê
Abbreviations
EPAG
eltrombopag
ITC
indirect treatment comparison
ITP
immune thrombocytopenia
PSA
probabilistic sensitivity analyses
ROMI
romiplostim
TPO-R
thrombopoietin receptor
W&R
watch-and-rescue
WTP
willingness-to-pay
Key Messages
Limited evidence from 1 cost-consequence analysis study showed that eltrombopag was the preferred thrombopoietin receptor agonist over romiplostim (i.e., less expensive and more effective) for treatment of chronic immune thrombocytopenia in pediatric patients.
No cost-effectiveness studies of dapsone or rituximab were identified.
Context and Policy Issues
Immune thrombocytopenia (ITP) affects approximately 5 in 100,000 children per year, with a peak incidence occurring in children between 2 and 5 years of age.1,2 ITP is an autoimmune disorder characterized by accelerated destruction of platelets, a risk of increased bleeding caused by platelet deficiencies (i.e., platelet count drops below 10 to 20 × 109 platelets/L).3 Of the cases, 75% to 80% can be self-resolved within 6 months,3 with some resolving within a year after diagnosis.4 Approximately 20% to 30% of pediatric ITP patients could not have spontaneous remission and the disease became chronic.3 Most patients present with mild bruising, and about 3% had serious bleeding from the nose, mucosa, and gastrointestinal tract.5 Intracranial hemorrhage is the most serious complication of ITP, with the rates ranging from 0.17% to 0.6% among cases.1,6,7 The cause of ITP remains unclear, although it can be triggered by viral infection, environmental factors, or immune defect.8
There are various ITP therapies available for preventing bleeding episodes by increasing platelet counts. Three commonly used first-line treatments for ITP are glucocorticoids, IV immunoglobulin, and anti-D immunoglobulin.8 Glucocorticoids (e.g., prednisone or dexamethasone) have both rapid and delayed effects in increasing platelet counts, but most patients relapse after the steroids are discontinued.9 Prolonged use of glucocorticoids has been associated with adverse side effects of steroids.9 IV immunoglobulin has been shown to be superior to glucocorticoids or no treatment in early recovery of platelet counts within 24 hours of administration.9 The exact mechanism of action of immunoglobulin is not well understood, although evidence has suggested that immunoglobulin slows down the destruction of platelets by inhibiting macrophage-induced phagocytosis.9 Anti-D immunoglobulin is an antibody against the Rhesus (Rh)-positive blood type. It works by binding to Rh-positive red blood cells, the complex of which then binds to the Fc receptors on macrophages, which induces the destruction of red blood cells, thus sparing the removal of antibody-coated platelets.9 Anti-D antibody is ineffective in patients with Rh-negative blood types and is contraindicated in patients with anemia.9
In patients having inadequate response to first-line therapies and still being treated with ITP for more than 3 to 6 months, second-line drugs may be considered.10 They are rituximab, thrombopoietin receptor (TPO-R) agonists, and splenectomy.10 Splenectomy is highly effective for inducing remission ITP and does not require maintenance therapy.10 Although highly effective, splenectomy is associated with increased risk of infections, especially bacterial sepsis.10 It is recommended to delay splenectomy in children for at least 12 months after initial ITP diagnosis, because some patients can achieve remission regardless of the choice of therapy.11 Rituximab is a monoclonal antibody to CD20, which targets and destroys autoantibody-producing B lymphocytes.9 The initial response rate of rituximab in children with chromic ITP is 40% to 50%, and the response rate at 2 to 5 years follow-up was only 25%.12,13 TPO-R agonists such as eltrombopag (EPAG) and romiplostim (ROMI), which stimulate megakaryocytes to increase the production of platelets, are emerging effective drugs for treatment of chronic ITP.14 EPAG (brand name Revolade) has been approved by Health Canada for the treatment of ITP in adult and pediatric patients who have had an insufficient response to corticosteroids or immunoglobulins.15 ROMI (brand name Nplate) has also been approved by Health Canada for the treatment of ITP in adult patients, but not in children, with chronic ITP.16 The advantage of EPAG over ROMI is that EPAG is administered orally, while ROMI is administered weekly via injection.10 Evidence has shown that both EPAG and ROMI appear to be effective treatment options for children with chronic ITP, but the their long-term efficacy and safety remain unclear.17 Other drugs including mycophenolate mofetil, mercaptopurine, cyclosporin, and dapsone has been used to treat pediatric ITP, but the rate of efficacy of these drugs was generally low.10
Numerous studies have assessed the cost-effectiveness of various therapies in the treatment of ITP.18-25 However, these studies did not consider pediatric patients.
The aim of this report is to summarize the evidence regarding the cost-effectiveness of dapsone, rituximab, and TPO-R agonists for pediatric patients with ITP.
Research Questions
What is the cost-effectiveness of dapsone for pediatric patients with ITP?
What is the cost-effectiveness of rituximab for pediatric patients with ITP?
What is the cost-effectiveness of TPO-R agonists for pediatric patients with ITP?
Methods
Literature Search Methods
A limited literature search was conducted by an information specialist on key resources including MEDLINE, Embase, the Cochrane Database of Systematic Reviews, the International HTA Database, the websites of Canadian and major international health technology agencies, as well as a focused internet search. The search strategy consisted of both controlled vocabulary, such as the National Library of Medicine’s MeSH (Medical Subject Headings), and keywords. The main search concepts were thrombocytopenia and specific treatments (dapsone, rituximab, and thrombopoietin receptor agonists). CADTH-developed search filters were applied to limit retrieval to economic studies. Comments, newspaper articles, editorials, letters, and conference abstracts were excluded. Where possible, retrieval was limited to the human population. The search was completed on March 28, 2022 and limited to English-language documents published since January 1, 2012.
Selection Criteria and Methods
One reviewer screened citations and selected studies. In the first level of screening, titles and abstracts were reviewed and potentially relevant articles were retrieved and assessed for inclusion. The final selection of full-text articles was based on the inclusion criteria presented in Table 1.
Criteria | Description |
|---|---|
Population | Pediatric patients (< 18 years of age) with immune thrombocytopenia |
Intervention | Q1: Dapsone Q2: Rituximab Q3: Thrombopoietin receptor agonists (e.g., romiplostim, eltrombopag) |
Comparator | Q1: Rituximab, thrombopoietin receptor agonists Q2: Dapsone, thrombopoietin receptor agonists Q3: Dapsone, rituximab, alternative thrombopoietin receptor agonists Q1 to Q3: Placebo, IV immunoglobulin, splenectomy |
Outcomes | Cost-effectiveness (e.g., cost per quality-adjusted life-year gained) |
Study designs | Economic evaluations |
Exclusion Criteria
Articles were excluded if they did not meet the selection criteria outlined in Table 1, or they were published before 2012.
Critical Appraisal of Individual Studies
The included publication was critically appraised by 1 reviewer using the Drummond checklist26 for economic evaluations. Summary scores were not calculated for the included study; rather, the strengths and limitations were described narratively.
Summary of Evidence
Quantity of Research Available
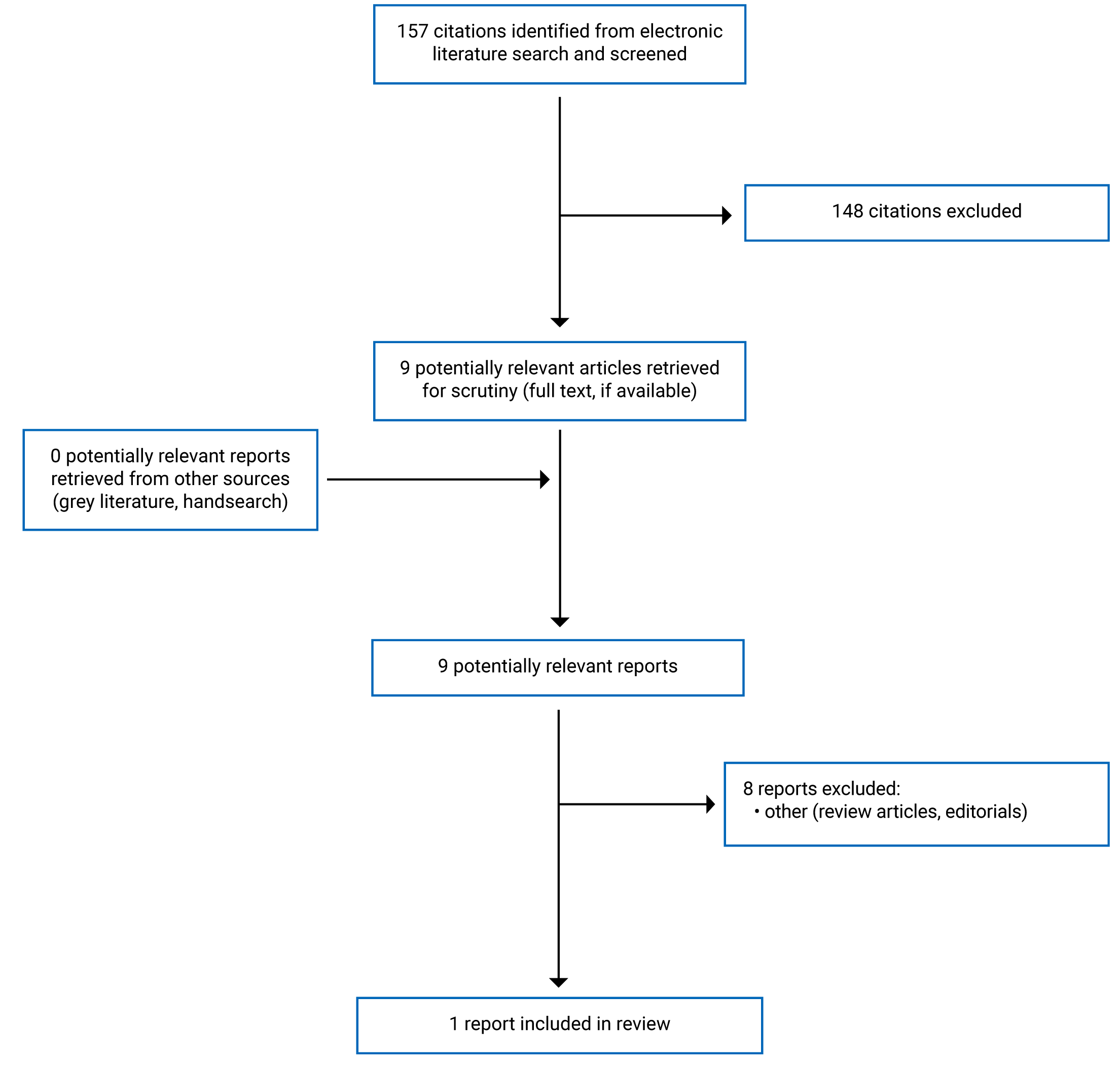
A total of 157 citations were identified in the literature search. Following screening of titles and abstracts, 148 citations were excluded and 9 potentially relevant reports from the electronic search were retrieved for full-text review. No potentially relevant publications were retrieved from the grey literature search for full-text review. Of these potentially relevant articles, 8 publications were excluded for various reasons, and 1 publication (i.e., economic evaluation) met the inclusion criteria and was included in this report. Appendix 1 presents the PRISMA27 flow chart of the study selection.
Summary of Study Characteristics
Additional details regarding the characteristics of the included publication are provided in Appendix 2.
Study Design
The economic evaluation study by Tremblay et al. (2018)28 used a cost-consequence model to assess the cost-effectiveness in response to different treatment outcomes. The clinical data were obtained from 2 randomized placebo-controlled trials.29,30 The indirect treatment comparison (ITC) was used to indirectly compared the clinical data of the comparators. All cost data were from US-based sources and US health care inflation of 3.6% was applied when there were no up-to-date data. No currency conversion was required. The analyses were conducted from a general US payer’s perspective over a 26-week time horizon. As the model horizon was less than 1 year, discounting was not applied.
Country of Origin
The economic evaluation study28 was conducted by authors from US.
Patient Population
Patients considered in the economic evaluation study28 were children with chronic ITP who had insufficient response to corticosteroids, immunoglobulins, or splenectomy from 2 randomized placebo-controlled trials.29,30 The 2 studies had some notable differences in baseline characteristics including ethnic origins, the amount of time since diagnosis, and proportion of male patients.
Interventions and Comparators
EPAG was the intervention, while ROMI and watch-and-rescue (W&R) were selected as comparators. W&R treatment was based on the placebo groups of the respective trials. Those patients would receive rescue therapy as needed.
Outcomes
The outcomes in the economic evaluation study28 were incremental cost-effectiveness ratio (ICER), which was calculated as the incremental cost per responder, per severe bleeding event avoided, per bleeding event avoided, and per patient.
Summary of Critical Appraisal
Additional details regarding the strengths and limitations of included economic evaluation study28 are provided in Appendix 3.
The included economic evaluation study28 clearly stated the objective, the economic importance of the research question, the rational for choosing the alternative comparators (i.e., EPAG versus ROMI and EPAG versus W&R), and the type of economic evaluation (i.e., cost-consequence analysis) that was conducted. The model was constructed from a general US payer’s perspective with a time horizon of 26 weeks. For data collection, the study clearly stated the source of effectiveness estimates with details of the design and findings (i.e., from 2 randomized placebo-controlled trials.29,30) The ITC technique was used to compare efficacy data between 2 trials, although there were some notable differences in baseline characteristics between trials that may affect the findings. The study clearly stated the outcome measures for economic evaluation (i.e., incremental cost per responder, incremental cost per severe bleeding event avoided, incremental cost per bleeding event avoided, and incremental cost per patient). A decision-tree model was presented with all parameters employed in the analysis. For the analysis and interpretation of results, the study clearly stated the time horizon of costs and benefits, statistical tests and confidence intervals, justification for the choice of variables for sensitivity analysis, and the ranges over which the variables were varied. Discounting was not applicable as the time horizon was less than 1 year. The study reported incremental analysis and presented major outcomes in a disaggregated as well as aggregated form. Probabilistic sensitivity analyses (PSA) were used to address uncertainty in the analysis, with a wide range of willingness-to-pay (WTP) thresholds. The conclusion in the study was based on the data reported and was accompanied by the appropriate caveats. Overall, the study was of moderate methodological quality with respect to the study design, data collection, and analysis and interpretation of results.
Summary of Findings
Appendix 4 presents the main study findings and authors’ conclusions.
Cost-Effectiveness of Dapsone for Pediatric Patients with ITP
No cost-effectiveness studies of dapsone versus rituximab or other TPO-R agonists were identified; therefore, no summary can be provided.
Cost-Effectiveness of Rituximab for Pediatric Patients with ITP
No cost-effectiveness studies of rituximab versus dapsone or other TPO-R agonists were identified; therefore, no summary can be provided.
Cost-Effectiveness of TPO-R Agonists for Pediatric Patients With ITP
For overall response (i.e., platelet counts ≥ 50 × 109/L without rescue medication use in the preceding 4 weeks), patients in the EPAG group had higher response rates compared to those in the ROMI group (difference of 2.3%) and W&R group (difference of 53.9%). There were fewer incidence in severe bleeding, overall bleeding, use of rescue medication in the EPAG group compared with ROMI and W&R groups. For total costs, EPAG was estimated to cost US$34,506 less than ROMI, and US$33,830 more than W&R. EPAG’s lower cost compared to ROMI was largely due to lower drug costs, administration costs, costs of severe bleeding, costs of moderate bleeding, and costs of adverse events.
Cost-effectiveness analysis using ICER revealed that EPAG was dominant (i.e., less expensive, and more effective) over ROMI when assessing incremental cost per responder, per severe bleeding event avoided, per bleeding event avoided, and per patient. Uncertainty in the results was assessed via PSA, whose results were consistent with base case findings. The PSA results showed that the probabilities that EPAG being cost-effective for treating children with chronic IPT were close or equal to 100% at all WTP thresholds ranging from US$25,000 to US$10,000,000, from a general US payer’s perspective. Compared with W&R, EPAG had higher total cost but had improved clinical outcomes (e.g., overall response, bleeding, and mortality). Regarding cost per severe bleeding avoided, EPAG had an ICER of US$354,197. The PSA results showed that EPAG had an ICER of 0.8% under a WTP threshold of US$100,000.
Limitations
One limitation of the economic evaluation study28 was that there was no head-to-head trials that compared EPAG and ROMI to assess their relative efficacy and safety. The ITC technique with some inherent limitations was used to adjust the ROMI efficacy data to match the EPAG data. The technique can be biased by both observed and unobserved differences in baseline characteristics between trials. Another limitation was that this study could not include rituximab and splenectomy, the 2 common treatments for chronic ITP, as comparators, due to unavailable data. The study used a relatively short time horizon in the model, and did not consider the indirect costs (i.e., societal perspective) in the model. Health-related quality of life was not included in the analyses. As the model was constructed from a US payer’s perspective, the findings of the study may not be applicable to the Canadian context. No evidence regarding the cost-effectiveness of dapsone or rituximab was identified.
Conclusions and Implications for Decision- or Policy-Making
This report identified 1 economic evaluation study28 using a cost-consequence model to compare the cost-effectiveness of EPAG versus ROMI in pediatric patients with chronic ITP. The study found that EPAG was dominant over ROMI (less expensive and more effective) when assessing cost per responder, cost per severe bleeding event avoided, cost per overall bleeding event avoided, and cost per patient. This was largely driven by lower drug and administrative costs and fewer severe bleeding outcomes. No cost-effectiveness studies of dapsone or rituximab for pediatric patients with ITP were identified. Future studies are needed to verify the cost-effectiveness of EPAG using different perspectives and longer time horizons, and to evaluate the cost-effectiveness of other ITP therapies including dapsone and rituximab.
References
1.Witmer CM, Lambert MP, O'Brien SH, Neunert C. Multicenter cohort study comparing U.S. management of inpatient pediatric immune thrombocytopenia to current treatment guidelines. Pediatr Blood Cancer. 2016;63(7):1227-1231. PubMed
2.Terrell DR, Beebe LA, Vesely SK, Neas BR, Segal JB, George JN. The incidence of immune thrombocytopenic purpura in children and adults: A critical review of published reports. Am J Hematol. 2010;85(3):174-180. PubMed
3.Neunert CE, Buchanan GR, Imbach P, et al. Bleeding manifestations and management of children with persistent and chronic immune thrombocytopenia: data from the Intercontinental Cooperative ITP Study Group (ICIS). Blood. 2013;121(22):4457-4462. PubMed
4.Imbach P, Kühne T, Müller D, et al. Childhood ITP: 12 months follow-up data from the prospective registry I of the Intercontinental Childhood ITP Study Group (ICIS). Pediatr Blood Cancer. 2006;46(3):351-356. PubMed
5.Neunert CE, Buchanan GR, Imbach P, et al. Severe hemorrhage in children with newly diagnosed immune thrombocytopenic purpura. Blood. 2008;112(10):4003-4008. PubMed
6.Kühne T, Buchanan GR, Zimmerman S, et al. A prospective comparative study of 2540 infants and children with newly diagnosed idiopathic thrombocytopenic purpura (ITP) from the Intercontinental Childhood ITP Study Group. J Pediatr. 2003;143(5):605-608. PubMed
7.Kime C, Klima J, Rose MJ, O'Brien SH. Patterns of inpatient care for newly diagnosed immune thrombocytopenia in US children's hospitals. Pediatrics. 2013;131(5):880-885. PubMed
8.Ma I, Sandhu AT. Immune thrombocytopenia. Hospital Medicine Clinics. 2017;6(1):53-66.
9.Bussel JB. Immune thrombocytopenia (ITP) in children: Initial management. In: Post TW, ed. UpToDate. Waltham (MA): UpToDate; 2022: http://www.uptodate.com. Accessed 2022 Apr 07.
10.Bussel JB. Immune thrombocytopenia (ITP) in children: Management of chronic disease. In: Post TW, ed. UpToDate. Waltham (MA): UpToDate; 2022: http://www.uptodate.com. Accessed 2022 Apr 07.
11.Neunert C, Lim W, Crowther M, Cohen A, Solberg L, Jr., Crowther MA. The American Society of Hematology 2011 evidence-based practice guideline for immune thrombocytopenia. Blood. 2011;117(16):4190-4207. PubMed
12.Parodi E, Rivetti E, Amendola G, et al. Long-term follow-up analysis after rituximab therapy in children with refractory symptomatic ITP: identification of factors predictive of a sustained response. Br J Haematol. 2009;144(4):552-558. PubMed
13.Patel VL, Mahévas M, Lee SY, et al. Outcomes 5 years after response to rituximab therapy in children and adults with immune thrombocytopenia. Blood. 2012;119(25):5989-5995. PubMed
14.Kuter DJ. The biology of thrombopoietin and thrombopoietin receptor agonists. Int J Hematol. 2013;98(1):10-23. PubMed
15.Revolade (eltrombopag): film-coated tablets, 25 mg and 50 mg (as eltrombopag olamine), oral use [product monograph]. Dorval (QC): Novartis Pharmaceuticals Canada Inc.; 2021: https://www.ask.novartispharma.ca/download.htm?res=revolade_scrip_e.pdf&resTitleId=1090. Accessed 2022 Apr 07.
16.Nplate (romiplostim): lyophilized powder for solution for injection, 250 μg/0.5 mL and 500 μg/1 mL [product monograph]. Mississauga (ON): Amgen Canada Inc.; 2018: https://pdf.hres.ca/dpd_pm/00046105.PDF. Accessed 2022 Apr 07.
17.Lassandro G, Palladino V, Vecchio GCD, et al. Thrombopoietin receptor agonists in children with immune thrombocytopenia: a new therapeutic era. Endocr Metab Immune Disord Drug Targets. 2021;21(3):397-406. PubMed
18.Goshua G, Sinha P, Kunst N, Pischel L, Lee AI, Cuker A. Cost-effectiveness of second-line therapies in adults with chronic immune thrombocytopenia. Am J Hematol. 2022;11:11. PubMed
19.Kaur MN, Arnold DM, Heddle NM, et al. Cost-effectiveness of eltrombopag vs intravenous immunoglobulin for the perioperative management of immune thrombocytopenia. Blood Adv. 2022;6(3):785-792. PubMed
20.Tremblay G, Dolph M, Roy AN, Said Q, Forsythe A. The cost-effectiveness of eltrombopag for the treatment of immune thrombocytopenia in the United States. Clin Ther. 2020;42(5):860-872.e868. PubMed
21.Allen R, Bryden P, Grotzinger KM, Stapelkamp C, Woods B. Cost-effectiveness of eltrombopag versus romiplostim for the treatment of chronic immune thrombocytopenia in England and Wales. Value Health. 2016;19(5):614-622. PubMed
22.Kikuchi K, Miyakawa Y, Ikeda S, Sato Y, Takebayashi T. Cost-effectiveness of adding rituximab to splenectomy and romiplostim for treating steroid-resistant idiopathic thrombocytopenic purpura in adults. BMC Health Serv Res. 2015;15:2. PubMed
23.Fust K, Parthan A, Li X, et al. Cost per response analysis of strategies for chronic immune thrombocytopenia. Am J Manag Care. 2018;24(8 Spec No.):SP294-SP302.
24.Augusto M, Gouveia M, Borges M, Campioni M. Cost-effectiveness of romiplostim for the treatment of chronic immune thrombocytopenia In Portugal. Value Health. 2014;17(7):A532. PubMed
25.Parrondo J, Grande C, Ibanez J, Palau J, Paramo JA, Villa G. [Economic evaluation of thrombopoietin receptor agonists in the treatment of chronic primary immune thrombocytopenia]. Farm Hosp. 2013;37(3):182-191. PubMed
26.Higgins JPT, Green S, editors. Figure 15.5.a: Drummond checklist (Drummond 1996). Cochrane handbook for systematic reviews of interventions. London (GB): The Cochrane Collaboration; 2011: http://handbook-5-1.cochrane.org/chapter_15/figure_15_5_a_drummond_checklist_drummond_1996.htm. Accessed 2022 Apr 07.
27.Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol. 2009;62(10):e1-e34. PubMed
28.Tremblay G, Dolph M, Bhor M, et al. Cost-consequence model comparing eltrombopag and romiplostim in pediatric patients with chronic immune thrombocytopenia. ClinicoEcon. 2018;10:715-721. PubMed
29.Grainger JD, Locatelli F, Chotsampancharoen T, et al. Eltrombopag for children with chronic immune thrombocytopenia (PETIT2): a randomised, multicentre, placebo-controlled trial. Lancet. 2015;386(10004):1649-1658. PubMed
30.Tarantino MD, Bussel JB, Blanchette VS, et al. Romiplostim in children with immune thrombocytopenia: a phase 3, randomised, double-blind, placebo-controlled study. Lancet. 2016;388(10039):45-54. PubMed
Appendix 1: Selection of Included Studies
Appendix 2: Characteristics of Included Publications
Table 2: Characteristics of Included Economic Evaluation
Study citation country, funding source | Type of analysis, time horizon, perspective | Population characteristics | Intervention and comparator(s) | Approach | Source of clinical, cost, and utility data used in analysis | Main assumptions |
|---|---|---|---|---|---|---|
Tremblay et al. (2018)28 US Novartis Pharmaceuticals | Cost-consequence analysis Time horizon: 26 weeks Perspective: US payer’s perspective Discount rate: NA | Children with chronic ITP who had insufficient response to corticosteroids, immunoglobulins, or splenectomy from 2 trials:
| EPAG vs. ROMI vs. W&R (placebo) EPAG (oral): 37.5 mg/day (25 mg/day for East Asians) for patients weighing less than 27 kg and 50 mg/day (25 mg/day for East Asians) for those weighing 27 kg. The dose was adjusted to 75 mg/day on the basis of individual platelet count. ROMI (SC injection): Dose adjusted weekly from 1 µg/kg to 10 µg/kg as weekly SC injection to target platelet counts of 50 to 200 x 109/L | The ITC technique was used to compare efficacy data between 2 trials. ICER was estimated as ratio of the difference in cost and difference in event. Cost outcomes: cost per responder, cost per bleeding event, cost per person. PSA were used to address uncertainty in the analysis, with the WTP thresholds ranging from $25,000 to $10,000,000. | Clinical data were from the EPAG trial and the ROMI trial. Severe bleeding (WHO grade 3 to 5) was the primary efficacy endpoint. Secondary efficacy endpoints were moderate bleeding and platelet response (platelet counts ≥ 50 × 109/L without rescue medication use in the preceding 4 weeks). All cost data were from US-based sources and US healthcare inflation (3.6%). No conversion needed. Total cost for each treatment included costs of drugs, administration, bleeding events, routine care, rescue medications, AEs (all grades), and mortality | Assumed that both clinical trials had similar baseline characteristics. |
AE = adverse event; EPAG = eltrombopag; ICER = incremental cost-effectiveness ratio; ITC = indirect treatment comparison; ITP = immune thrombocytopenia; NA = not applicable; PSA = probabilistic sensitivity analysis; ROMI = romiplostim; SC = subcutaneous; vs. = versus; W&R = watch-and-rescue.
Note: This table has not been copy-edited.
Appendix 3: Critical Appraisal of Included Publications
Note that this appendix has not been copy-edited.
Table 3: Strengths and Limitations of Economic Evaluation Using the Drummond Checklist26
Strengths | Limitations |
|---|---|
Tremblay et al. (2018)28 | |
Study design | |
|
|
Data Collection | |
| |
Analysis and interpretation of results | |
| |
EPAG = eltrombopag; ICER = incremental cost-effectiveness ratio; ITC = indirect treatment comparison; ITP = immune thrombocytopenia; PSA = probabilistic sensitivity analysis; ROMI = romiplostim; W&R = watch-and-rescue.
Appendix 4: Main Study Findings and Authors’ Conclusions
Note that this appendix has not been copy-edited.
Summary of Findings of Included Economic Evaluation
Tremblay et al. (2018)28
Main Study Findings
Cost-consequence analysis of EPAG vs. ROMI or EPAG vs. W&R
Efficacy Outcomes
Response
Change (EPAG – ROMI): 2.3%
Change (EPAG – W&R): 53.9%
Severe bleeding (WHO 3 to 5)
Change (EPAG – ROMI): -22.1%
Change (EPAG – W&R): -9.6%
Moderate bleeding only (WHO 2)
Change (EPAG – ROMI): -15.7%
Change (EPAG – W&R): -8.3%
Use of rescue medication
Change (EPAG – ROMI): -1.9%
Change (EPAG – W&R): -5.1%
Mortality (derived from severe bleeding)
Change (EPAG – ROMI): -0.79%
Change (EPAG – W&R): -0.34%
Costs (US$)
Drug costs
Change (EPAG – ROMI): -22,194
Change (EPAG – W&R): 40,178
Administrative costs
Change (EPAG – ROMI): -1,955
Change (EPAG – W&R): -889
Rescue medication costs
Change (EPAG – ROMI): 414
Change (EPAG – W&R): -1,201
Cost of severe bleeding (WHO 3 to 5)
Change (EPAG – ROMI): -9,837
Change (EPAG – W&R): -4,259
Cost of moderate bleeding (WHO 2)
Change (EPAG – ROMI): -354
Change (EPAG – W&R): -183
AEs costs
Change (EPAG – ROMI): -151
Change (EPAG – W&R): 373
Mortality costs
Change (EPAG – ROMI): -437
Change (EPAG – W&R): -189
Total costs
Change (EPAG – ROMI): -34,506
Change (EPAG – W&R): 33,830
ICER
Incremental cost per responder
EPAG/ROMI: Dominant
EPAG/W&R: US$62,749
Incremental cost per severe bleeding avoided (WHO 3 to 5)
EPAG/ROMI: Dominant
EPAG/W&R: US$354,197
Incremental cost per bleeding event avoided (WHO 2 to 5)
EPAG/ROMI: Dominant
EPAG/W&R: US$189,303
Incremental cost per patient
EPAG/ROMI: Dominant
EPAG/W&R: US$62,749
PSA Results — ICER for Costs per Severe Bleeding Avoided
At WTP of US$25,000: EPAG was 97.5% and 0.3% cost-effective compared with ROMI and W&R, respectively.
At WTP of US$50,000: EPAG was 98.3% and 0.5% cost-effective compared with ROMI and W&R, respectively.
At WTP of US$100,000: EPAG was 99.2% and 0.8% cost-effective compared with ROMI and W&R, respectively.
At WTP of US$150,000: EPAG was 99.6% and 1.5% cost-effective compared with ROMI and W&R, respectively.
At WTP of US$250,000: EPAG was 99.9% and 14.6% cost-effective compared with ROMI and W&R, respectively.
At WTP of US$500,000: EPAG was 100% and 79.8% cost-effective compared with ROMI and W&R, respectively.
At WTP of US$1,000,000: EPAG was 100% and 98.8% cost-effective compared with ROMI and W&R, respectively.
At WTP of US$10,000,000: EPAG was 100% and 99.7% cost-effective compared with ROMI and W&R, respectively.
Authors’ Conclusion
“EPAG was the preferred TPO-R agonist to treat chronic ITP when indirectly compared to ROMI, largely driven by its favorable severe bleeding outcomes and lower drug and administration costs.”28 (p. 715)
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up-to-date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third-party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.
Questions or requests for information about this report can be directed to Requests@CADTH.ca