CADTH Health Technology Review
Therapeutic Mattresses for Chronic Pain
Rapid Review
Authors: Angela M. Barbara, Aleksandra Grobelna
Abbreviations
AMSTAR 2
A MeaSurement Tool to Assess systematic Reviews 2
RCT
randomized controlled trial
SR
systematic review
Key Messages
One systematic review (that summarized 9 relevant studies) provided evidence on the use of mattresses for treating chronic low back pain.
Medium-firm mattresses may lead to less back pain and better sleep quality compared with firmer mattresses.
Mattresses with an air overlay may also improve pain and sleep quality in those with chronic low back pain.
Several limitations of the data informing this report were identified, which limits the certainty of the evidence.
Context and Policy Issues
Chronic pain is generally defined as pain lasting for 3 months or longer or persisting beyond the time needed for normal tissue healing.1 It can affect the quality of life of those living with chronic pain and can lead to sleeplessness, functional disability, and emotional distress.2 One in 5 people living in Canada has chronic pain; it is 1 of the most common reasons for individuals seek medical attention. The economic burden is substantial.2 An estimated annual direct cost of $7.2 billion is associated with managing chronic pain in Canada.3
Chronic pain can affect various parts of the body, such as the lower back, upper back, knee, leg, feet, shoulder, neck, and hip. Lower back pain appears to be the most predominant type, occurring in more than one-third of those who have chronic pain.4 The goal of treatment is to control pain, maintain function, maximize coping, and prevent disability. There are several non-pharmacological treatment options available for chronic pain, such as exercise, multidisciplinary rehabilitation, psychological therapies, and physical modalities.1 Specific types of mattresses may also be included in this list. The primary purpose of a mattress is to support the sleeper’s posture and maintain the body’s normal and neutral postural position by adjusting the normal spinal alignment according to the body curvature to distribute weight uniformly.5 Therapeutic mattresses refer to any mattress or bedding with design characteristics or features that are specifically designed for patient care to achieve patient outcomes (i.e., mattress used as an intervention for people with chronic pain). Companies and manufactures of mattresses may promote their particular types of mattresses as producing health benefits but there is insufficient research to support these claims.6,7
Previous CADTH reports on this topic include a 2014 Summary With Critical Appraisal on the use of mattresses for chronic back or neck pain8 and a 2016 Reference List on the use of therapeutic bedding for chronic pain.9 In the 2014 report,8 limited conclusive evidence was identified regarding the efficacy of specific mattress types for treatment of back and neck pain. One randomized controlled trial (RCT) was identified presenting evidence that firm mattresses may be the least effective treatment for lower back pain. Four guidelines were identified that found a lack of evidence to form a basis for mattress recommendations for the treatment of chronic back and neck pain of musculoskeletal origin. The 2016 CADTH report9 identified no relevant health technology assessments, systematic reviews (SRs), RCTs, or non-randomized studies that assessed the clinical effectiveness of therapeutic bedding for patients with neuropathic pain.
The objective of this report is to identify and review current evidence regarding the clinical effectiveness of therapeutic mattresses for the treatment of people with chronic pain.
Research Question
What is the clinical effectiveness of therapeutic mattresses for the management of people with chronic pain?
Methods
Literature Search Methods
A limited literature search was conducted by an information specialist on key resources including MEDLINE, CINAHL, the Cochrane Database of Systematic Reviews, the International HTA Database, the websites of Canadian and major international health technology agencies, as well as a focused internet search. The search strategy comprised both controlled vocabulary, such as the National Library of Medicine’s MeSH (Medical Subject Headings), and keywords. The main search concepts were therapeutic mattresses and chronic pain. No filters were applied to limit the retrieval by study type. Comments, newspaper articles, editorials, and letters were excluded. If possible, retrieval was limited to the human population. The search was also limited to English-language documents published between January 1, 2017, and February 18, 2022.
Selection Criteria and Methods
One reviewer screened citations and selected studies. In the first level of screening, titles and abstracts were reviewed and potentially relevant articles were retrieved and assessed for inclusion. The final selection of full-text articles was based on the inclusion criteria presented in Table 1.
Criteria | Description |
|---|---|
Population | People (any age) with chronic pain |
Intervention | Therapeutic mattresses (e.g., specialty mattresses designed to improve health outcomes) |
Comparator | Standard mattresses |
Outcomes | Clinical effectiveness (e.g., pain, physical function, sleep quality, sleep latency, emotional and psychological functioning [e.g., anxiety, depression], health-related quality of life, changes in use of pharmacotherapy, safety [e.g., adverse events, hospitalizations]) |
Study designs | Health technology assessments, systematic reviews, randomized controlled trials, non-randomized studies |
Exclusion Criteria
Articles were excluded if they did not meet the selection criteria outlined in Table 1, were duplicate publications, or were published before 2017.
Critical Appraisal of Individual Studies
The included publication was critically appraised by 1 reviewer using A MeaSurement Tool to Assess systematic Reviews 2 (AMSTAR 2).10 Summary scores were not calculated for the included studies; rather, the strengths and limitations of the included publication were described narratively.
Summary of Evidence
Quantity of Research Available
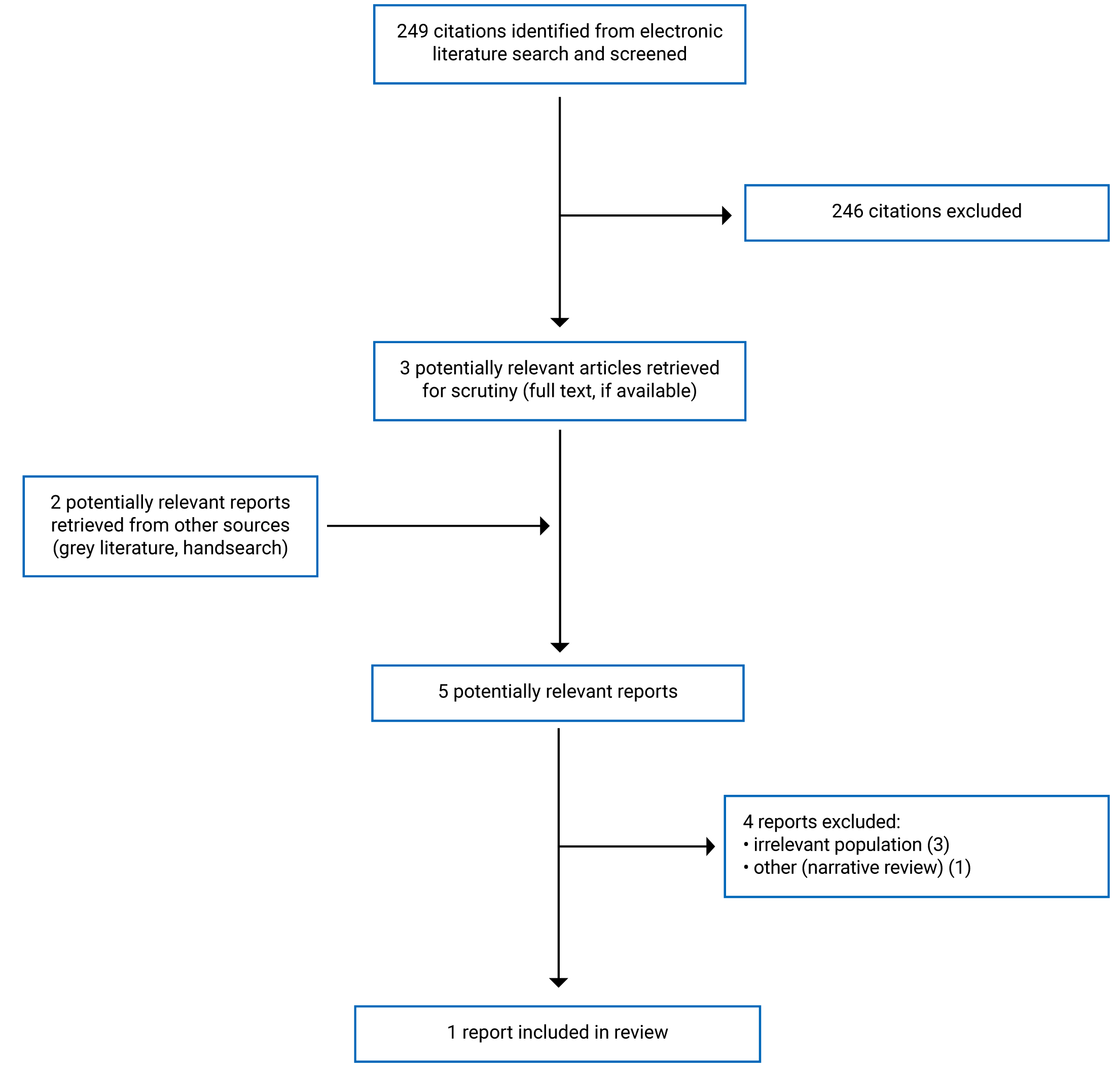
A total of 249 citations were identified in the literature search. Following screening of titles and abstracts, 246 citations were excluded and 3 potentially relevant reports from the electronic search were retrieved for full-text review. Two potentially relevant publications were retrieved from the grey literature search for full-text review. Of these 5 potentially relevant articles, 4 publications were excluded for various reasons, and 1 publication met the inclusion criteria and was included in this report. This comprised 1 SR. Appendix 1 presents the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow chart11 of the study selection.
Additional references of potential interest are provided in Appendix 5.
Summary of Study Characteristics
One SR with a narrative synthesis7 was included in this report.
The SR7 had broader inclusion criteria considered than the present review. Specifically, in addition to studies in populations with chronic low back pain, the SR included studies of different type of mattresses in asymptomatic populations for the prevention of low back pain and promotion of sleep quality and correct spinal column alignment. Only the characteristics and results of the subset of relevant studies in patients with chronic pain7 were described in this report.
The details regarding the characteristics of the included SR are provided in Appendix 2.
Study Design
The SR7 investigated the association of different mattresses with sleep quality and low back pain and provided a narrative synthesis. The databases searched were Science Direct, PsycINFO, Embase, PubMed, Google Scholar, Web of Science, Scopus, and Cochrane Collaboration. The date range covered by the search was 2000 to 2019. The 9 relevant primary studies were 2 RCTs and 7 single-case non-randomized studies (3 controlled trials, 2 non-randomized controlled trials, 2 before-and-after or pretest-posttest studies). The relevant primary studies were published between 2000 and 2010.
Country of Origin
The authors of the SR7 were from Italy. The countries in which the primary studies were conducted were not reported in the SR.
Patient Population
The SR7 presented data on a total of 808 adults with chronic low back pain. The number of participants in each study ranged from 19 to 313. The ages of the participants in the individual studies were not reported. The proportion of females in 2 non-randomized studies by Jacobson et al. was 51%. This detail was not reported for the other studies. The duration of pain experienced by participants in the RCT by Kovacs et al. was a minimum of 3 months; this information was not reported for the other primary studies.7
Interventions and Comparators
The 9 relevant studies in the SR7 assessed different combinations of interventions and comparators. Most of the relevant primary studies described the design of the mattress intervention according to its firmness.
The 2 RCTs in the SR7 evaluated the effects of different types of therapeutic mattresses on back pain. The double-blind multi-centre RCT by Kovacs et al. included in the SR7 compared medium-firm mattresses and firm mattresses. Mattress firmness was scored based on the scale developed by the European Committee for Standardization that ranged from 0 (maximum firmness) to 10 (minimum firmness). The medium-firm mattress was rated as 5.6 and the firm mattress was rated as 2.3. The other relevant studies in the SR assessed mattress firmness on the basis of subjective evaluations.7
The single-blind RCT by Berghodt et al. included in the SR7 assessed 3 types of mattresses: waterbed (Akva), body-conforming foam mattress (Tempur), and an adjustable airbed mattress with firmness control (Futon Innovation).
The non-randomized studies in the SR7 were single-case studies that each evaluated 1 type of therapeutic mattress compared with a standard mattress. The 2010 non-randomized study by Jacobson et al. included in the SR7 assessed individually prescribed mattresses compared with participants’ own mattresses. Participants served as their own control and assessed their own mattress during the 3-week “pretest” and the prescribed mattress during the 12-week intervention stage. Similarly, in the other 4 non-randomized studies by Jacobson et al. included in the SR,7 participants evaluated the clinical effectiveness of new mattresses with intermediate firmness (medium or mid-firm) compared with their own personal mattresses. In these studies, the medium-firm surface of the mattresses was based on the following components: foam-encased Bonnell spring unit, densified fibre pad, super-soft foam, damask cover, semi-flex foundation, and slick fibred. The mattresses had the same sizes as mattresses previously used by study participants.7
Two non-randomized studies in the SR7 evaluated mattresses with air overlay systems. The study by Monsein et al. assessed a spring bed with air topper; the study by Price et al. used an air-filled mattress low-pressure fixed overlay (Repose). The study by Monsein et al.7 used an A-B-A design, in which baseline data were obtained from participants’ experience on their own mattress (A), then the airbed intervention was installed and assessed for 28 days (B), and finally the participants’ own bed was reinstalled and evaluated for another 14 days (A). An A-B design was used in the study by Price et al.,7 in which participants provided data on their experience with their own bedding (A) and then provided data using the overlay mattress at 4 weeks (B).
The length of time that participants were assigned to sleep on the mattress interventions ranged from 28 days, 4 weeks, or 1 month (most studies) to 6 months (1 study).
Outcomes
The clinical effectiveness outcomes reported in the SR7 were pain, sleep quality, and disability. Eight studies (2 RCTs and 6 non-randomized studies) assessed self-reported pain. Seven non-randomized studies assessed self-reported sleep quality and 1 RCT evaluated self-reported disability. Pain and sleep quality were assessed via visual analogue scales in 8 studies. The other subjective measures included a Danish questionnaire named COBRA (not further defined in the SR) to measure pain symptom levels, Short Form (36) Health Survey, and the Epworth Daytime Sleepiness Scale. The Spanish version of the Roland-Morris Disability Questionnaire was used to measure degree of disability.
For each study in the SR,7 outcome data (e.g., visual analogue scale scores, effect sizes, P values) for each study were not reported. Rather, study conclusions were presented.
Summary of Critical Appraisal
An overview of the critical appraisal of the included publication is summarized in the following text. Additional details regarding the strengths and limitations of the included SR are provided in Appendix 3.
Systematic Reviews
The SR7 had a number of strengths. The objective and inclusion criteria were clearly stated. A literature search was conducted using multiple databases, the selection of articles was described and a flow chart presented, and a table of the included primary studies was given. Providing details of the literature search strategy increases the reproducibility of the review. Heterogeneity across studies was considered and described. The SR received no funding, and the authors reported no conflicts of interest.7
The SR7 also had limitations. It was not reported whether a protocol had been published before the conduct of the review; therefore, it is unknown whether any significant protocol deviations occurred that may have affected the interpretation of the findings. The SR did not report searching the grey literature or trial registries, which limits the breadth of literature found. Although study selection was done by 2 reviewers, it was unclear how data extraction was done. Therefore, the potential for errors in data extraction is unknown. A list of excluded studies was not presented in the review. In the absence of justifications for excluding studies, it is unclear if the selection process captured all the relevant studies.7 A brief description of the included studies was provided (e.g., study design, aim, materials and methods, conclusion); however, the level of detail varied across studies and was lacking or insufficient for some studies (e.g., complete description of the population, full details of the mattress intervention). Insufficient detail on any study characteristics may limit how the results from this SR can be applied to other contexts.
The authors of the SR classified the level of evidence based on the study design of the primary studies.7 SRs with or without meta-analyses were classified as evidence I, RCTs were classified as evidence II, and controlled clinical trials without randomization were classified as evidence III. However, the classification is uncertain; for example, the RCT by Kovacs et al. was classified as evidence I, and it is unclear whether the Kovacs et al. study was misclassified as an SR or if the RCT was assigned a higher level of evidence for unreported reasons. The RCT by Bergholdt et al. was classified as evidence II, and the remaining 7 studies were evidence III. Study design alone does not indicate level of study quality or risk of bias, nor does it indicate certainty in evidence for the outcomes measured. Without risk of bias assessment using a validated tool, it is not possible to know to what extent biases may have affected the findings of the relevant studies and how to properly interpret the conclusions (which may underestimate or overestimate the effects).
Summary of Findings
The main findings from the included SR7 are summarized in the following sections and in Appendix 4.
Clinical Effectiveness of Therapeutic Mattresses
The findings for this report are informed by 9 primary studies summarized in 1 SR.7 Numerical data and P values from the primary studies were not reported in the SR, which limits the interpretation of the results.
Pain
The SR7 included 8 primary studies that assessed low back pain.
The RCT by Kovacs et al. included in the SR7 found that improvement was observed using both mattresses. Although participants using the medium-firm mattress reported a higher level of pain reduction, the difference between groups was reported as non-significant. The RCT by Berghodt et al. included in the SR7 reported that water mattresses and foam mattresses had a more positive influence on low back pain compared with firm mattresses.
The SR7 reported that the 2010 non-randomized study by Jacobson et al. found significant decrease in back pain, back stiffness, and shoulder pain with individually prescribed mattresses compared with participants’ own mattresses. Three non-randomized studies by Jacobson et al. in the SR7 found that sleeping on medium-firm mattresses resulted in pain improvement. The 2009 study by Jacobson et al. in the SR7 reported that this benefit was found independently from initial sleep level, age, weight, height, and body mass index. Improvement also appeared to progressively increase between the first and the fourth week of using the new medium-firm mattresses.
Participants had significantly less pain sleeping on the adjustable airbed in the non-randomized study by Monsein et al. included in the SR.7 There was also statistically significant pain improvement with the use of a new low-pressure inflatable overlay mattress in the non-randomized study by Price et al. reported in the SR.7
Sleep Quality
The SR7 included 7 primary studies that reported on sleep quality.
The SR7 reported that there was significant improvement in sleep quality with individually prescribed mattresses compared with participants’ own mattresses in the 2010 non-randomized study by Jacobson et al.
Four non-randomized studies by Jacobson et al. summarized in the SR7 found that the use of medium-firm mattresses resulted in improved sleep compared with participants’ own standard mattresses. One non-randomized study (Jacobson et al. [2008]) in the SR7 reported that prompt replacement of mattress system components improved quality of sleep. The SR reported significant improvement of sleep quality of 55% with mid-firm mattress compared with participants’ own mattresses was reported in the 2002 study by Jacobson et al.7 At a 5-month to 6-month follow-up from the initial experimental phase of the 2008 study by Jacobson et al. in the SR,7 individuals completed an additional sleep assessment to complete the evaluation and reported that the positive effects had lasted over time.
Two non-randomized studies in the SR7 reported that, compared with participants’ own standard mattresses, beds with air overlays resulted in significant improvement in sleep quality in people with chronic back pain.
Disability
One primary study in the SR7 reported on disability. Results of the RCT by Kovacs et al. in the SR7 showed that, although improvement was observed using both medium-firm and firm mattresses, participants using a medium-firm mattress reported a higher level of improved disability.
Limitations
The evidence in this report the same limitations of the included SR.7 The relevant primary studies in the SR7 were limited by sample sizes: two-thirds of relevant studies involved fewer than 100 participants. The duration of the interventions in the included studies were short, ranging from 28 days to 90 days. One study had a follow-up of 5 months to 6 months, but no studies had long-term follow-up. Thus, the long-term effectiveness of the mattresses remains unclear. The RCT by Kovacs et al. was double-blinded and the RCT by Bergholdt et al. was single-blinded. However, the 7 non-randomized studies were unblinded, which increased the risk of performance bias and detection bas. The non-randomized studies were also limited by the lack of a control group. Most studies, except for the Kovacs et al. study, assessed mattress firmness on the basis of subjective evaluations. Although the studies used reproducible grading scales to assess back pain and sleep quality, the data were based on subjective ratings of participants’ outcomes rather than on objective measurements. Risk of bias in primary studies was not assessed in the SR.7 Any quality issues from the primary studies causes uncertainty in the findings presented in the SR. The countries in which the included studies were conducted were not reported. There was considerable heterogeneity among the primary studies identified in the SR7 in terms of interventions (mattress designs), comparators, and duration of intervention. This heterogeneity makes it difficult to make a definitive conclusion regarding the clinical effectiveness of therapeutic mattresses.
There were several reporting issues in the SR.7 There was a lack of (or inconsistent) detail about the included studies, such as study populations (e.g., age, sex, assessment of pain, duration of pain), settings (e.g., residential, institutional), interventions (e.g., mattress support systems), and findings (e.g., outcome data, effect sizes, P values). Therefore, generalizability of the findings to the Canadian context are unknown.
This report is also limited in terms of population and outcomes. The SR7 specifically addressed low back pain and sleep quality. The effectiveness of therapeutic mattresses on other types of chronic pain is unknown. The SR7 also did not report physical function, sleep latency, emotional and psychological functioning, health-related quality of life, changes in use of pharmacotherapy, or safety.
Conclusions and Implications for Decision- or Policy-Making
This rapid review was conducted to examine the clinical effectiveness of therapeutic mattresses for the management of people with chronic pain. Two previous CADTH reports from 20148 and 20169 also addressed this topic. The 2014 report8 found a lack of evidence to inform the choice of mattress to positively influence chronic back and neck pain. This conclusion was based on the review of 1 RCT (Bergholdt et al., included in the SR of this report) and 4 evidence-based guidelines. The 2016 CADTH report9 did not identify any relevant publications that assessed the clinical effectiveness of therapeutic bedding for patients with neuropathic pain. The current report includes 1 SR7 that narratively summarized 9 primary studies.
The SR7 reported that several types of mattresses demonstrated beneficial effects in study participants with chronic low back pain. Mattresses with intermediate firmness were better at improving disability compared with firm mattresses. Water mattresses and foam mattresses were also better than firm mattresses for low back pain. Medium-firm mattresses and mattresses with an air overlay system provided more effective pain relief and better sleep quality compared with standard mattresses. There was also evidence that a prescribed individualized bedding system reduced pain and improved sleep. However, the limitations of the SR7 should be considered when interpreting these findings, including the unknown quality of the primary studies in the SR, heterogeneity in the interventions and comparisons, and limited reporting of study details (e.g., study characteristics and findings).
More robust research using well-designed, high-quality studies is required to support the clinical effectiveness and subsequent decision-making regarding the use of therapeutic mattresses for people with chronic pain.
References
1.Skelly A, Chou R, Dettori J, et al. Noninvasive nonpharmacological treatment for chronic pain: a systematic review. Rockville (MD): Agency for Healthcare Research and Quality (US); 2018: https://www.ncbi.nlm.nih.gov/books/NBK556229/pdf/Bookshelf_NBK556229.pdf. Accessed 2022 Mar 21.
2.Canadian Pain Task Force. Chronic pain in Canada: laying a foundation for action. Ottawa (ON): Health Canada; 2019: https://physiotherapy.ca/sites/default/files/canadian_pain_task_force_june_2019_report_en.pdf. Accessed 2022 Mar 21.
3.Hogan ME TA, Katz J, Shah V, Krahn M. Incremental health care costs for chronic pain in Ontario, Canada: A population-based matched cohort study of adolescents and adults using administrative data. Pain. 2016;157(8):1626-1633. PubMed
4.Schopflocher D TP, Jovey R. The prevalence of chronic pain in Canada. Pain Research and Management. 2011;16(6):445-450. PubMed
5.Lopez-Torres M, Porcar R, Solaz J, Romero T. Objective firmness, average pressure and subjective perception in mattresses for the elderly. Appl Ergon. 2008;39(1):123-130. PubMed
6.Shields N, Capper J, Polak T, Taylor N. Are cervical pillows effective in reducing neck pain? NZJ Physiother. 2006;34.
7.Caggiari G TG, Toro G, Jannelli E, Monteleone G, Puddu L. What type of mattress should be chosen to avoid back pain and improve sleep quality? Review of the literature J Orthop Traumatol. 2021;22(1):51. PubMed
8.Mattresses for chronic back or neck pain: a review of the clinical effectiveness and guidelines. (CADTH rapid response report: summary with critical appraisal). Ottawa (ON): CADTH; 2014: https://www.cadth.ca/media/pdf/htis/nov-2014/RC0553%20Mattresses%20for%20Chronic%20Pain%20Final.pdf. Accessed 2022 Mar 21.
9.Therapeutic Bedding for Chronic Pain: Clinical Effectiveness. Ottawa, ON: CADTH;2016.
10.Shea BJ, Reeves BC, Wells G, et al. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ. 2017;358:j4008. PubMed
11.Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol. 2009;62(10):e1-e34. PubMed
Appendix 1: Selection of Included Studies
Appendix 2: Characteristics of Included Publication
Note that this appendix has not been copy-edited.
Table 2: Characteristics of Included Systematic Review
Study citation, country, funding source(s) | Review objectives, last search dates, numbers of primary studies included | Population characteristics | Intervention | Comparators | Outcomes, length of follow-up |
|---|---|---|---|---|---|
Caggiari et al. (2021)7 Italy Funding source: No funding | Review objective: To evaluate available studies to understand which mattresses can effectively reduce back pain or prevent its onset Search dates: 2000 to 2019 Number of relevant studies: 9 (2 RCT, 7 NRS) | Included population: Adults with chronic low back pain Excluded population: Athletes, children; hospitalized adults; patients with ulcer, asthma, other pathologies unrelated to pain Sample size of relevant primary studies: 19 to 313 Age: 18 years and older % female: 50.8 (reported for 2 NRS only) | Firm mattress Medium-firm mattress Technologies-based mattress Mattress with air overlay system | Medium-firm mattress Waterbed mattress Memory foam mattress Own standard mattress | Outcomes:
Follow-up:
|
NR = not reported; NRS = non-randomized study; RCT = randomized controlled trial; SF = Short Form; SR = systematic review; VAS = Visual Analogue Scale.
aCOBRA not further defined.
Appendix 3: Critical Appraisal of Included Publication
Note that this appendix has not been copy-edited.
Table 3: Strengths and Limitations of Systematic Reviews Using AMSTAR 210
Strengths | Limitations |
|---|---|
Caggiari et al. (2021)7 | |
• The research question and inclusion criteria for the review included the components of PICO • Comprehensive literature search strategy and detailed methods were described • The search was conducted in multiple databases and key search terms were provided • Study selection was performed in duplicate, and discrepancy between evaluations were resolved by involving a third reviewer to analyze the controversy • The level of evidence in individual studies was classified based on study methodology • Authors acknowledged the heterogeneity observed in the included studies • Authors reported that no funding was received and declared that they had no conflicts of interest | • Unclear whether review methods were established before the conduct of the review • Authors did not provide justification for eligible study designs • Unclear if data extraction was performed in duplicate • Details on study design, methods, and sample characteristics of included studies inconsistently reported (and lacking for some studies) • Authors did not provide a list of excluded studies with justifications for exclusion • Risk of bias in individual studies was not assessed • Authors did not investigate publication bias (small study bias) • Sources of funding for individual studies included in the review were not reported |
AMSTAR 2 = A MeaSurement Tool to Assess systematic Reviews 2.
Appendix 4: Main Study Findings
Note that this appendix has not been copy-edited.
Table 4: Summary of Study Findings by Outcome — Pain
Intervention | Comparison(s) | Study citation and study design | Pain |
|---|---|---|---|
Firm mattress | Medium-firm mattress | Caggiari et al. (2021)7 SR (1 RCT) | Kovacs et al. (2003) VAS pain score in 313 patients with chronic low back pain
|
Firm mattress | Waterbed mattress and memory foam mattress | Caggiari et al. (2021)7 SR (1 RCT) | Bergholdt et al. (2008) Danish COBRA questionnaire in 160 people with chronic low back pain
|
Individually prescribed (technologies-based) mattress | Own standard mattress | Caggiari et al. (2021)7 SR (1 NRS) | Jacobson et al. (2010) VAS pain score in 27 patients with back pain, back stiffness, and shoulder pain
|
Medium-firm mattress | Own standard mattress | Caggiari et al. (2021)7 SR (3 NRS) | Jacobson et al. (2009) VAS pain score in 59 people with back stiffness
|
Jacobson et al. (2006) VAS pain score in 59 people with back stiffness
| |||
Jacobson et al. (2002) VAS pain score in 22 people with shoulder pain, low back pain, and spine stiffness
| |||
Mattress with air overlay | Own standard mattress | Caggiari et al. (2021)7 SR (1 A-B-A design, 1 A-B prospective pilot study) | Monsein et al. (2000) VAS score and SF-36 Health Survey in 90 people with symptomatic chronic low back pain
|
Price et al. (2003) VAS score in 19 patients with chronic back pain
|
NRS = non-randomized study; RCT = randomized controlled trial; SF = Short Form; SR = systematic review; VAS = Visual Analogue Scale.
Note: Numerical data and P values from primary studies were not reported in the SR.
Table 5: Summary of Study Findings by Outcome — Sleep Quality
Intervention | Comparison | Study citation and study design | Sleep quality |
|---|---|---|---|
Individually prescribed (technologies-based) mattress | Own standard mattress | Caggiari et al. (2021)7 SR (1 NRS) | Jacobson et al. (2010) VAS pain score in 27 patients with back pain
|
Medium-firm mattress | Own standard mattress | Caggiari et al. (2021)7 SR (4 NRS) | Jacobson et al. (2009) Evaluation of sleep quality in 59 people with back stiffness
|
Jacobson et al. (2008) VAS pain score in 59 people with low back, shoulder, and spine stiffness
| |||
Jacobson et al. (2006) VAS pain score in 59 people with back stiffness
| |||
Jacobson et al. (2002) VAS pain score in 22 people with disturbed sleep, shoulder pain, low back pain, and spine stiffness
| |||
Mattress with air overlay | Own standard mattress | Caggiari et al. (2021)7 SR (1 A-B-A design, 1 A-B prospective pilot study) | Monsein et al. (2000) VAS score and Epworth Daytime Sleepiness Scale in 90 people with symptomatic chronic low back pain
|
Price et al. (2003) VAS score in 19 patients with chronic back pain
|
RCT = randomized controlled trial; SR = systematic review; VAS = Visual Analogue Scale.
Note: Numerical data and P values from primary studies were not reported in the SR.
Table 6: Summary of Study Conclusions by Outcome — Disability
Intervention | Comparison | Study citation and study design | Pain |
|---|---|---|---|
Firm mattress | Medium-firm mattress | Caggiari et al. (2021)7 SR (1 RCT) | Kovacs et al. (2003) Roland-Morris questionnaire in 313 patients with chronic low back pain
|
Note: Numerical data and P values from primary studies were not reported in the SR.
Appendix 5: References of Potential Interest
Previous CADTH Reports
Mattresses or overlays used in palliative end-of-life care: clinical evidence and guidelines. (CADTH rapid response report: summary of abstracts). Ottawa (ON): CADTH; 2019: https://www.cadth.ca/sites/default/files/pdf/htis/2019/RB1304%20Pressure%20Relief%20Mattresses%20Final.pdf. Accessed 2022 Mar 21.
Lateral rotation mattresses for the prevention and treatment of pressure injuries: clinical effectiveness and guidelines. (CADTH rapid response report: summary of abstracts). Ottawa (ON): CADTH; 2019: https://www.cadth.ca/sites/default/files/pdf/htis/2019/RB1392%20Lateral%20Rotation%20Mattresses%20Final.pdf. Accessed 2022 Mar 21.
Mobility beds for patients with dementia: clinical effectiveness and cost-effectiveness. (CADTH rapid response report: summary of abstracts). Ottawa (ON): CADTH; 2017: https://www.cadth.ca/sites/default/files/pdf/htis/2017/RB1058%20Mattresses%20for%20Dementia%20Final.pdf. Accessed 2022 Mar 21.
Therapeutic bedding for chronic pain: clinical effectiveness. (CADTH rapid response report: reference list). Ottawa (ON): CADTH; 2016: https://www.cadth.ca/sites/default/files/pdf/htis/june-2016/RA0851%20Therapeutic%20Bedding%20Final.pdf. Accessed 2022 Mar 21.
Mattresses for chronic back or neck pain: a review of the clinical effectiveness and guidelines. (CADTH rapid response report: summary with critical appraisal). Ottawa (ON): CADTH; 2014: https://www.cadth.ca/media/pdf/htis/nov-2014/RC0553%20Mattresses%20for%20Chronic%20Pain%20Final.pdf. Accessed 2022 Mar 21.
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up-to-date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third-party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.
Questions or requests for information about this report can be directed to Requests@CADTH.ca