CADTH Health Technology Review
Cost-Effectiveness and Guidelines for Opioid Substitution Treatment
Rapid Review
Authors: Kwakye Peprah, Hannah Loshak
Abbreviations
AGREE II
Appraisal of Guidelines for Research and Evaluation II
CCSMH
Canadian Coalition for Seniors Mental Health
CEAC
cost-effectiveness acceptability curve
CRISM
Canadian Research Initiative in Substance Misuse
DAM
diacetylmorphine
GRADE
Grades of Recommendation Assessment, Development, and Evaluation
HDM
hydromorphone
ICER
incremental cost-effectiveness ratio
MMT
methadone maintenance treatment
NAOMI
North American Opiate Medication Initiative
OUD
opioid use disorder
QALY
quality-adjusted life-year
RCT
randomized controlled trial
RIOTT
Randomized Injectable Opiate Treatment Trial
SALOME
Study to Assess Long-term Opioid Maintenance Effectiveness
SROM
slow-release oral morphine
Key Messages
There was evidence indicating that in the treatment of opioid use disorder, injectable hydromorphone, or injectable methadone provided more benefit at less cost compared with injectable diacetylmorphine over a 6-month time horizon.
Evidence suggests that in the treatment of opioid use disorder, both injectable hydromorphone and injectable diacetylmorphine are likely to provide more benefit at less cost than methadone maintenance treatment. Treatment with injectable hydromorphone was more cost-effective than injectable diacetylmorphine in opioid use disorder patients who do not respond to or relapse from drug treatments.
The evidence is limited because observed data were collected during a short-term follow-up, and long-term cost-effectiveness outcomes were based on extrapolations beyond data from the actual studies.
One guideline provided a weak recommendation, supported by low-quality evidence, for using slow-release oral morphine in older adults with adequate renal function in whom buprenorphine and methadone maintenance have been ineffective to treat opioid use disorder or could not be tolerated. Another guideline recommends using injectable hydromorphone or injectable diacetylmorphine for individuals with severe opioid use disorders who relapsed previous treatments failed.
No relevant cost-effectiveness evidence or guidelines with recommendations regarding the use of oral hydromorphone, fentanyl patches, or fentanyl buccal tablets for opioid use disorder treatment were identified; therefore, no summary can be provided.
Context and Policy Issues
Opioid dependence has an enormous burden on individuals and society due to the association between opioid use disorder (OUD) and criminal activity, incarceration, deterioration in overall health and social functioning, quality of life, and overdose-related death.1-3 First-line treatment for most patients with OUD includes pharmacotherapy with an opioid agonist or antagonist and adjunct psychosocial treatment.4 Methadone maintenance treatment (MMT) and buprenorphine, for example, have been shown to be effective in improving physical and psychological health, decreasing drug use, infectious disease transmission, illegal activity, and death in many individuals.5,6 However, a subpopulation of individuals with severe OUD fail to benefit and continue to inject heroin regularly, necessitating alternative approaches with enhanced effectiveness.3,5,7 There is emerging evidence suggesting that individuals who do not respond to or relapse from the effect of these first-line drug treatments may benefit from supervised injectable opioids such as prescription diacetylmorphine (heroin),2,3 hydromorphone,1 or other oral alternatives.8
While the expanded OUD treatment options may lead to better outcomes, it comes with challenges such as the risk of adverse effects, restricted access, and increased resource pressure on health services with an associated cost.7 In 2017, CADTH produced a report summarizing abstracts of publications concerning the comparative clinical effectiveness, cost-effectiveness, and evidence-based recommendations for using the various formulations for use in OUDs.9 The objective of this Rapid Response report is to review current full-text evidence regarding the cost-effectiveness of various opioid agonist interventions for treating OUDs and summarize identified evidence-based guidelines recommendations for their use. The clinical effectiveness of opioid substitution treatment is being reviewed in a separate report.10
Research Questions
Q1: What is the cost-effectiveness of sustained-release oral morphine in opioid use disorder?
Q2: What is the cost-effectiveness of oral hydromorphone in opioid use disorder?
Q3: What is the cost-effectiveness of injectable hydromorphone or prescription diacetylmorphine in opioid use disorder?
Q4: What is the cost-effectiveness of fentanyl patches or fentanyl buccal tablets in opioid use disorder?
Q5: What are the evidence-based guidelines regarding the use of sustained-release oral morphine, oral hydromorphone, injectable hydromorphone, injectable prescription diacetylmorphine, fentanyl patches or fentanyl buccal tablets for opioid abuse treatment?
Methods
Literature Search Methods
A limited literature search was conducted by an information specialist on key resources including MEDLINE, the Cochrane Database of Systematic Reviews, the international HTA database, the websites of Canadian and major international health technology agencies, as well as a focused Internet search. The search strategy comprised controlled vocabularies, such as the National Library of Medicine’s MeSH (Medical Subject Headings), and keywords. The main search concepts were opioid substitution therapies. CADTH-developed search filters were applied to limit retrieval to economic studies and guidelines. Where possible, retrieval was limited to the human population. The search was also limited to English documents published between January 1, 2012, and November 4, 2021.
Selection Criteria and Methods
One reviewer screened citations and selected studies. In the first screening level, titles and abstracts were reviewed, and potentially relevant articles were retrieved and assessed for inclusion. The final selection of full-text articles was based on the inclusion criteria presented in Table 1.
Criteria | Description |
|---|---|
Population | Adults (18 years and older) requiring treatment for opioid use disorder |
Intervention | Q1 and Q5: Sustained release oral morphine (SROM) (also referred to as slow or extended-release oral morphine) Q2 and Q5: Oral hydromorphone (also referred to as immediate-release oral hydromorphone) Q3 and Q5: Injectable hydromorphone or prescription diacetylmorphine (also referred to as heroin-assisted therapy [HAT] or medical-grade heroin) Q4 and Q5: Fentanyl patches (also referred to as transdermal fentanyl) or fentanyl buccal tablets (also referred to as sublingual fentanyl) |
Comparator | Q1 to Q4: Placebo; standard of care (i.e., buprenorphine-naloxone, methadone); alternative interventions of interest (i.e., SROM, oral hydromorphone, injectable hydromorphone or prescription diacetylmorphine, fentanyl patches, or fentanyl buccal tablets) Q5: Not applicable |
Outcomes | Q1 to Q4: Cost-effectiveness outcomes (e.g., cost per quality-adjusted life-year, cost per health benefit gained) Q5: Recommendations regarding the appropriate use (including the role of witness ingestion, appropriateness as a substitute for standard of care, dosing regimens, settings of use) of the interventions of interest in adults with opioid use disorder |
Study designs | Economic Evaluations and Evidence-based Guidelines |
Exclusion Criteria
Articles were excluded if they did not meet the selection criteria outlined in Table 1, they were duplicate publications, or were published before 2012. Guidelines8,11 with relevant portions adapted into another guideline that is more recent and more comprehensive were omitted.
Critical Appraisal of Individual Studies
The included publications were critically appraised by 1 reviewer using the following tools as a guide: the Drummond checklist12 for economic evaluations and the Appraisal of Guidelines for Research and Evaluation (AGREE) II instrument13 for guidelines. Summary scores were not calculated for the included studies; instead, the strengths and limitations of each included publication were described narratively.
Summary of Evidence
Quantity of Research Available
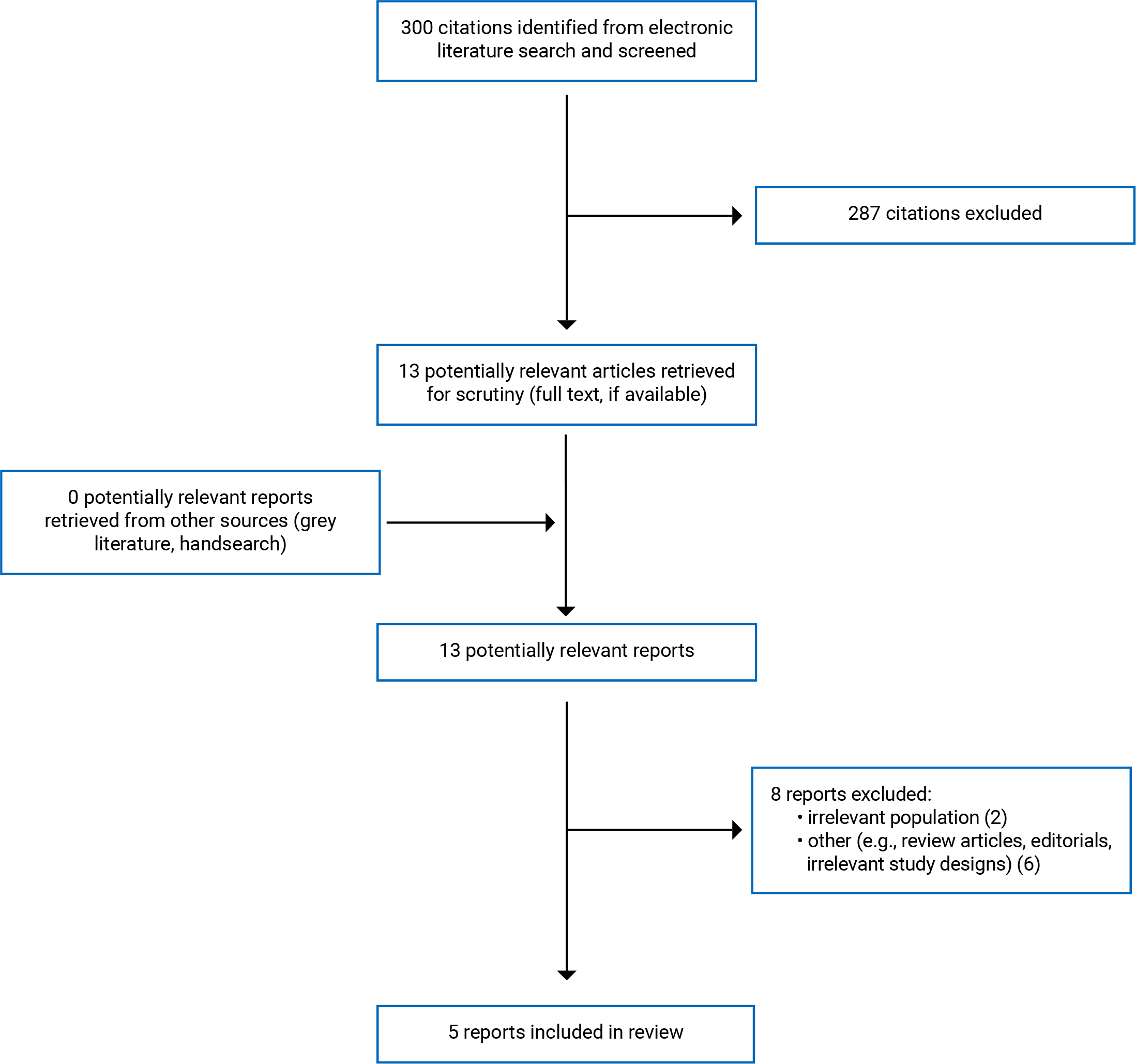
A total of 300 citations were identified in the literature search. After screening titles and abstracts, 287 citations were excluded, and 13 potentially relevant reports from the electronic search were retrieved for full-text review. No potentially relevant publications were retrieved from the grey literature search for full-text review. Of the 13 articles, 8 publications were excluded for various reasons, and 5 papers that met the inclusion criteria were included in this report. These comprised 3 economic evaluations7,14,15 and 2 evidence-based guidelines.16,17 Appendix 1 presents the PRISMA18 flow chart of the study selection.
Summary of Study Characteristics
Additional details regarding the characteristics of included publications are provided in Appendix 2.
Study Design
All the 3 included economic evaluations7,14,15 were based on randomized controlled trials. One each used data from the Study to Assess Long-term Opioid Maintenance Effectiveness (SALOME),14 the Randomized Injectable Opiate Treatment Trial (RIOTT),7 and the North American Opiate Medication Initiative (NAOMI)15 trials. Each study evaluated estimated incremental costs, quality-adjusted life-years (QALYs) and cost-effectiveness ratios from a societal perspective, considering costs borne by the health care,7,14,15 social services,7,14,15 and criminal justice systems,7,14,15 as well as out-of-pocket costs borne by society.14,15 QALYs were calculated using the Euroqol EQ-5D tool as a utility measure. The considered time horizons for analyses were 26 weeks,7,14 1 year,15 5 years,15 10 years,15 and a lifetime.14,15 Lifetime analysis was based on average additional years lived following entry into the model of 14.515 to 14.9,14 17.5,14 and 15.4515 to 18.414 for patients treated with MMT, injectable hydromorphone (HDM), and injectable diacetylmorphine (DAM), respectively. All the 3 economic evaluations7,14,15 sourced clinical and costs data from the respective RCTs, administrative sources, and the published literature. A decision-analytic approach was used in all the economic evaluations, with 2 studies14,15 stating that a semi-Markov model was applied. In contrast, 1 study7 did not specify the model used in the analysis.
Two evidence-based guidelines16,17 were included in this report. The Canadian Coalition for Seniors Mental Health developed 1 of the guidelines,16 and the Canadian Research Initiative in Substance Misuse developed the other guideline.17 Both guidelines16,17 were developed using evidence from relevant literature identified through systematic literature searches. The recommendations were developed through consensus. The quality of evidence and the strength of recommendations were rated and reported according to the Grades of Recommendation Assessment, Development, and Evaluation (GRADE) tool.16,17 The evidence quality was ranked as high, moderate, low, and very low. A high rate indicates strong confidence that the actual and estimated effects are close. A very low rank means very little confidence in the effect estimate because the true and estimated effects are likely to be substantially different.17 A moderate rating signifies moderate confidence because the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. A low-quality rank denotes a limited confidence because the true and estimated effects may be significantly different. In 1 of the guidelines,16 the low and very low scores were joined together and reported as low.
Country of Origin
Two of the 3 economic evaluations were conducted in Canada,14,15 and 1 was conducted in the UK.7 Both of the included evidence-based guidelines16,17 were developed in Canada.
Patient Population
Patients who participated in the trials that formed the basis for the included economic evaluations were adults who had received previous treatments with opioid agonists, including MMT but were still injecting street heroin (diacetylmorphine) as at the time of enrolment into the various studies.7,14,15 Sample sizes of the source trials were 251 (for NAOMI),2,15 127 (for RIOTT),7 and 202 (for SALOME).14 The mean age of the patients was between 37 and 40 years old, and most were male (≤ 61%).7,14,15 The mean duration for which the patients had been injecting drugs was between 13.7 and 16.5 years.7,14,15 In 1 study,7 eligible patients had been on conventional oral MMT for at least 6 months but continued to inject street heroin regularly (≥ 50% of days in the preceding 3 months). Another study15 required that patients should have had at least 2 previous opioid substitution treatment attempts and been out of treatment for at least 6 months before trial entry.
The target population of 1 of the guidelines was older adults (i.e., ≥ 65 years) with OUD.16 In the other guideline,17 the target population was adults with severe OUD relapsed or unsuccessful on a previous oral opioid agonist treatment, or whose circumstances and risks indicated that they might benefit from injectable opioid agonist treatment. Both guidelines16,17 were intended for use by health care professionals in Canada.
Interventions and Comparators
One economic evaluation14 was based on the SALOME trial that randomized patients to treatment with injectable hydromorphone (HDM) or injectable diacetylmorphine (DAM). Doses were presented in diacetylmorphine equivalents up to 400 mg per dose.1 A patient could receive up to 3 doses per day, but not exceeding 1,000 mg per day.1
One economic evaluation was based on the RIOTT trial in which patients were randomly assigned to DAM, injectable methadone, or optimized oral methadone (oral methadone).7 The DAM was given twice daily titrated on an individual basis to a typical stabilizing daily dose of between 300 mg/day and 600 mg/day, to a maximum of 900 mg/day.7 Injectable methadone was administered once daily, titrated individually to a maximum of 200 mg/day.7
Another economic evaluation15 was based on the NAOMI trial in which patients were randomly allocated to injection-assisted treatment (55%) or methadone maintenance alone (45%). Patients in the injection group were randomly assigned to receive hydromorphone HDM or DAM.2 The doses of the individual drugs were not specified.
Pharmacological interventions considered within 1 of the guidelines16 included buprenorphine maintenance, buprenorphine-naloxone, MMT, naltrexone alone, slow-release oral morphine (SROM). The other guideline17 focused on DAM and HDM.
Outcomes
For economic evaluations,7,14,15 reported outcomes were costs and QALYs gained, and incremental cost-effectiveness ratios (ICER). One of the studies assessed cost-effectiveness at specified willingness-to-pay thresholds using cost-effectiveness acceptability curves (CEAC). For guidelines,16,17 major outcomes considered by the guideline panels included a reduction in illicit heroin or any street opioid use, premature death, nonfatal overdose, blood-borne infectious diseases, involvement in sex work and criminal activity, as well as improvement in treatment retention rates, overall health, and social functioning.
Summary of Critical Appraisal
Economic Evaluation
The 3 included economic evaluations7,14,15 stated research objectives and their economic importance, and provided the perspective and time horizons used in analyses. Each of the studies7,14,15 compared relevant alternatives, described the rationale for choosing them and reported incremental effects due to their use. Two of the economic evaluations14,15 stated and justified the type of analysis and modelling approach used in the evaluation were described and justified. Although authors of the other study7 reported that their analysis used a decision-making approach, the evaluation model and its parameters were not adequately described, and its selection was not justified. All the included economic evaluations7,14,15 reported the sources of data for effectiveness estimates. The supporting RCTs were adequately referenced, and the population of interest was described for each evaluation. The studies7,14,15 stated the primary outcome measure and reported methods of estimating quantities, unit cost, and value of benefits.
Two economic evaluations14,15 applied discount rates to adjust for inflation, although no justification was provided for the chosen rates. One study7 did not adjust for inflation in the analysis, although it was based on cost data from a study conducted at least 3 years earlier. Therefore, the reported cost of providing the OUD treatments in that study7 may have been underestimated. In 1 of the economic evaluations,14 the lifetime analysis extrapolated costs and outcomes data from multiple sources and applied an indirect comparison approach. However, the methods of data synthesis were not provided. The major outcomes in all the included economic evaluations7,14,15were presented in both disaggregated and aggregated forms, and the approaches to sensitivity analysis were described clearly. For each economic evaluation, the discussion of the results considered the study's limitations, and the conclusions reflected the evidence used to derive them.
Guidelines
The included guidelines16,17 had clear objectives to answer specific health questions in well-described populations. They were developed based on evidence from a systematic review of relevant literature. Although for 1 guideline16 the criteria for selecting the evidence and provision of advice or tools on applying the recommendations were unclear, each of the guidelines16,17 demonstrated strengths in all the 6 domains in the AGREE II instrument.13 Overall, both guidelines16,17 ranked positively regarding scores for scope and purpose, stakeholder involvement, development rigour, presentation clarity, applicability, and editorial independence. Thus, they provide essential details, such as an explicit link between recommendations and supporting evidence, criteria for selecting evidence, strengths and limitations of the body of evidence, and methods for formulating recommendations. Both guidelines16,17 were externally peer-reviewed and had input from a broad base of stakeholders, including intended professional users and targeted populations representatives. Furthermore, each guideline16,17 provided information about facilitators, barriers, and potential resource implications for applying the recommendations and stated monitoring approaches to ensure the recommendations were up to date.
Additional details regarding the strengths and limitations of included publications are provided in Appendix 3.
Summary of Findings
Appendix 4 presents the main study findings and authors’ conclusions.
Cost-Effectiveness of Sustained Release Oral Morphine in Opioid Use Disorder
No relevant evidence regarding the cost-effectiveness of SROM for OUD was identified; therefore, no summary can be provided.
Cost-Effectiveness of Oral Hydromorphone in Opioid Use Disorder
No relevant evidence regarding the cost-effectiveness of oral hydromorphone for OUD was identified; therefore, no summary can be provided.
Cost-Effectiveness of Injectable Hydromorphone or Prescription Diacetylmorphine in Opioid Use Disorder
One economic evaluation14 found that in the short term (up to 6 months), treatment with HDM dominated (i.e., providing more benefit at less cost than) DAM in OUD patients who were refractory to treatments (ICER HDM versus DAM = 6,683,925 CN$/QALY). The outcomes were not discussed in terms of decision-makers’ willingness-to-pay threshold. In analysis extrapolating data over a lifetime, the study14 found that compared with MMT, the probability of providing more benefit at less cost in treating OUD was higher with HDM (67%) and DAM (75%).
One economic evaluation7 with 26 weeks’ time horizon showed that at a willingness-to-pay threshold of £30,000 per QALY, both DAM and injectable methadone dominated oral methadone. At the same willingness-to-pay level, injectable methadone was more likely to be more cost-effective than DAM (80% versus 20%).7 Sensitivity analysis showed that the relative cost-effectiveness was sensitive to the price of DAM. However, the DAM price needed to fall considerably (≥ 84%) before being more cost-effective than injectable methadone.7 However, DAM had a probability of being more cost-effective than injectable methadone at a higher willingness-to-pay threshold (i.e., ≥ £70,000).
One economic evaluation15 that compared DAM and MMT over 1-year, 5-year, 10-year, and lifetime horizons found that DAM was a dominant strategy over MMT to treat OUD in each of the time horizons. Sensitivity analysis showed that DAM's cost-effective probability was 76% and 95% at a willingness-to-pay thresholds of $0 per QALY gained and $100,000 per QALY gained, respectively.
Cost-Effectiveness of Fentanyl Patches or Fentanyl Buccal Tablets in Opioid Use Disorder >
No relevant evidence regarding the cost-effectiveness of fentanyl patches or fentanyl buccal tablets for OUD was identified; therefore, no summary can be provided.
Guidelines
Sustained-Release Oral Morphine
The CCSMH guideline16 recommends that SROM be considered with caution for treating OUDs in older adults with adequate renal function in whom buprenorphine and methadone maintenance have been ineffective or could not be tolerated. The recommendation is weak and is supported by low-quality evidence from 1 guideline, 1 consensus statement of an International Expert Panel, and 1 animal study.16 According to the recommendation, the treatment should be initiated by first starting the patient on supervised short-acting morphine before transitioning to maintenance with the long-acting, 24-hour SROM formulation.
Injectable Hydromorphone and Injectable Prescription Diacetylmorphine
The CRISM guideline17 recommends that for patients who are determined to be likely to benefit from injectable opioid agonist treatment, both DAM and HDM are acceptable treatment options. The recommendation was rated as strong, although supported by low-quality evidence from 2 systematic reviews and 1 RCT.17 According to the authors, the strong rating was based on expert consensus, substantial clinical experience in British Columbia, reduced risk of adverse events for HDM compared with DAM, and the lack of regulatory and supply barriers affecting access to HDM.17 The CRISM guideline also recommends that treatment with injectable opioid agonists be provided on an open-ended basis, with decisions to transition to oral opioid agonists made collaboratively with the patient.17 The recommendation was rated as strong despite being supported by low-quality evidence because it aligns with the WHO's recommendation that opioid agonists be provided as open-ended treatment for OUD, and the open-ended approach potentially reduces a patient’s risk of exposure to fentanyl-contaminated illicit opioid use.17
No evidence-based guidelines with recommendations regarding the use of oral hydromorphone, fentanyl patches, or fentanyl buccal tablets for OUD treatment were identified; therefore, no summary can be provided.
Limitations
A key limitation for the included cost-effectiveness studies7,14,15 is that the actual observed data came from trials with short-term follow-up duration. Although 2 out of the 3 economic evaluations14,15 provided longer-term outcomes, they were based on extrapolations beyond the supporting studies, and 1 study14 indirectly compared interventions from multiple sources without giving details about the data synthesis approach. Thus, there is some uncertainty about the reported long-term cost-effectiveness outcomes. Further, the long-term evaluations were limited to comparisons between MMT and either HDM14 or DAM.14,15 Therefore, it is unknown if the reported short-term cost-effectiveness dominance of HDM14 and injectable methadone7 over DAM could be replicated in analysis with a longer time horizon. One of the economic evaluations7 was based on a study conducted in the UK with the cost data denominated in the British pound sterling and analysis not adjusted for inflation. Therefore, in addition to a likely underestimation of the costs, the generalizability of the reported findings in the Canadian context is unknown. Also, besides methadone, none of the economic evaluations considered other important oral opioid agonist interventions such as slow-release morphine or buprenorphine preparations.
No relevant cost-effectiveness evidence or guidelines with recommendations regarding the use of oral hydromorphone, fentanyl patches, or fentanyl buccal tablets for OUD treatment were identified; therefore, no summary can be provided.
Conclusions and Implications for Decision- or Policy-Making
Three included economic evaluations7,14,15 based on data from RCTs, and 2 evidence-based guidelines16,17 were included in this Rapid Response report. Results of cost-effectiveness analyses from the studies showed that in the short term (up to 6 months), HDM provided more benefit than DAM at less cost,14 and injectable methadone had a greater probability of being more effective than DAM (80% versus 20%).7 Two of the economic evaluations showed that MMT had a lower likelihood of being more cost-effective for the treatment of OUD compared with HDM14 or DAM14,15 in a lifetime analysis14,15 or over shorter time horizons (i.e., 1-year, 5-year, or 10-years horizons).15 There is some uncertainty about the reported long-term cost-effectiveness outcomes due to data extrapolations14,15 and lack of clarity about how data from multiple sources were synthesized.14 Further, the long-term evaluations were limited to comparisons between MMT and either HDM14 or DAM.14,15 Thus, it is unknown if the reported short-term cost-effectiveness dominance of HDM14 or injectable methadone7 over DAM could be replicated in analysis with a longer time horizon.
One guideline16 recommends using SROM in older adults with adequate renal function in whom buprenorphine and methadone maintenance have been ineffective to treat OUD or could not be tolerated. Another guideline recommends using injectable opioid agonists for individuals with severe, treatment-refractory OUD engaging in illicit injection opioid use, adding that HDM and DAM are acceptable treatment options.
No relevant cost-effectiveness evidence or guidelines with recommendations regarding the use of oral hydromorphone, fentanyl patches, or fentanyl buccal tablets for OUD treatment were identified; therefore, no summary can be provided.
Given the limitations discussed here and elsewhere in this report, future economic evaluations comparing the various opioid agonist therapies for OUD over the short- and long-terms are needed. Similarly, there is a need for evidence-based recommendations regarding the use of different opioid agonist treatment options for OUD.
References
1.Oviedo-Joekes E, Guh D, Brissette S, et al. Hydromorphone Compared With Diacetylmorphine for Long-term Opioid Dependence: A Randomized Clinical Trial. JAMA Psychiatry. 2016;73(5):447-455. PubMed
2.Oviedo-Joekes E, Nosyk B, Brissette S, et al. The North American Opiate Medication Initiative (NAOMI): profile of participants in North America's first trial of heroin-assisted treatment. J Urban Health. 2008;85(6):812-825. PubMed
3.Ferri M, Davoli M, Perucci CA. Heroin maintenance for chronic heroin-dependent individuals. Cochrane Database Syst Rev. 2011(12):CD003410. PubMed
4.Saxon A, Strain E, Peavy K. Approach to treating opioid use disorder. In: Post TW, ed. UptoDate. Waltham (MA): UptoDate; 2021: www.uptodate.com. Accessed 2021 Dec 6.
5.Mattick RP, Breen C, Kimber J, Davoli M. Buprenorphine maintenance versus placebo or methadone maintenance for opioid dependence. Cochrane Database Syst Rev. 2014(2):CD002207. PubMed
6.Sordo L, Barrio G, Bravo MJ, et al. Mortality risk during and after opioid substitution treatment: systematic review and meta-analysis of cohort studies. BMJ. 2017;357:j1550. PubMed
7.Byford S, Barrett B, Metrebian N, et al. Cost-effectiveness of injectable opioid treatment v. oral methadone for chronic heroin addiction. Br J Psychiatry. 2013;203(5):341-349. PubMed
8.Bruneau J, Ahamad K, Goyer ME, et al. Management of opioid use disorders: a national clinical practice guideline. CMAJ. 2018;190(9):E247-E257. PubMed
9.Sustained release oral morphine, injectable hydromorphone, and prescription diacetylmorphine for opioid use disorder: clinical and cost-effectiveness, and guidelines. (CADTH Rapid response report: summary of abstracts). Ottawa (ON): CADTH; 2017: https://www.cadth.ca/sites/default/files/pdf/htis/2017/RB1083%20-%20Opioid%20Substitution%20Treatment%20Final.pdf. Accessed 2021 Dec 6.
10.Clinical effectiveness of opioid substitution treatment. (CADTH Rapid response report: summary with clinical appraisal). Ottawa (ON): CADTH; 2021: https://www.cadth.ca/. Accessed 2021 Dec 6.
11.A guideline for the clinical management of opioid use disorder. Vancouver (BC): British Columbia Centre on Substance Use; 2017: https://www.bccsu.ca/wp-content/uploads/2017/06/BC-OUD-Guidelines_June2017.pdf. Accessed 2021 Nov 19.
12.Higgins JPT, Green S, editors. Figure 15.5.a: Drummond checklist (Drummond 1996). Cochrane handbook for systematic reviews of interventions. London (GB): The Cochrane Collaboration; 2011: http://handbook-5-1.cochrane.org/chapter_15/figure_15_5_a_drummond_checklist_drummond_1996.htm. Accessed 2021 Dec 6.
13.Agree Next Steps Consortium. The AGREE II Instrument. Hamilton (ON): AGREE Enterprise; 2017: https://www.agreetrust.org/wp-content/uploads/2017/12/AGREE-II-Users-Manual-and-23-item-Instrument-2009-Update-2017.pdf. Accessed 2021 Dec 6.
14.Bansback N, Guh D, Oviedo-Joekes E, et al. Cost-effectiveness of hydromorphone for severe opioid use disorder: findings from the SALOME randomized clinical trial. Addiction. 2018;113(7):1264-1273. PubMed
15.Nosyk B, Guh DP, Bansback NJ, et al. Cost-effectiveness of diacetylmorphine versus methadone for chronic opioid dependence refractory to treatment. CMAJ. 2012;184(6):E317-328. PubMed
16.Rieb LM, Samaan Z, Furlan AD, et al. Canadian Guidelines on Opioid Use Disorder Among Older Adults. Can Geriatr J. 2020;23(1):123-134. PubMed
17.Fairbairn N, Ross J, Trew M, et al. Injectable opioid agonist treatment for opioid use disorder: a national clinical guideline. CMAJ. 2019;191(38):E1049-E1056. PubMed
18.Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol. 2009;62(10):e1-e34. PubMed
Appendix 1: Selection of Included Studies
Appendix 2: Characteristics of Included Publications
Table 2: Characteristics of Included Economic Evaluations
Study citation country, funding source | Type of analysis, time horizon, perspective | Population characteristics | Intervention and comparator(s) | Approach | Source of clinical, cost, and utility data used in the analysis | Main assumptions |
|---|---|---|---|---|---|---|
Bansback et al., 201814 Canada Funded by: Canadian Institutes of Health Research, Providence Health Care, The InnerChange Foundation, Providence Health Care Research Institute, St. Paul’s Hospital Foundation, and Vancouver Coastal Health. | Estimates of incremental costs, QALYs and cost-effectiveness ratio from a societal perspective. Time horizons were 6 months and a lifetime for direct and indirect comparisons, respectfully. | Adult long-term street opioid injection users (n = 202) participating in the SALOME trial. The patients had at least 2 previous treatment attempts, including 1 with methadone (or other opioid substitution therapy) Age, mean (SD):a 45.17 (10.19) years for HDM and 43.50 (9.03) years for DAM Male, n (%):a 67 (67.0) for HDM and 73 (71.6) for DAM Years injecting street heroin (DAM), mean (SD): a 15.56 (9.45) for HDM and 15.34 (9.29) for DAM Times attempted MMT in the prior 5 years; mean (SD):a 2.77 (2.14) | Injectable HDM vs. DAM (directly) and methadone maintenance treatment (indirectly). | The direct comparisons used patient-level data in a within-trial analysis, and the indirect analysis used a decision-analytical cohort model to evaluate a lifetime extrapolated costs and outcomes | For the within-trial analysis, clinical and costs data, including medication costs, human resources, and overheads, were sourced from the SALOME trial. Drug costs were derived from the Active Pharmaceutical Ingredient (API) price and associated costs of production based on the reported in the SALOME trial. For the decision-analytical model (i.e., lifetime analysis) clinical data external data, including patient-level data from the SALOME and NAOMI trials and the literature to extrapolate costs and QALYs | The base-case analysis assumed a similar cohort of adults who have severe opioid use disorder with demographic and other clinical characteristics as participants in the SALOME trial. The mortality rate was the same in the hydromorphone and diacetylmorphine states A 50-year time horizon, which assumed relapse occurred only in a state without engagement with care Life-time analysis was based on average additional years lived following entry into the model of 14.9, 17.5, and 18.4 for patients treated with MMT, HDM, and DAM, respectively Change in duration of successive diacetylmorphine and hydromorphone episodes (regarding transition probabilities to abstinence or relapse) was equivalent to methadone maintenance therapy |
Byford et al., 20137 The UK Funded by the Community Fund (Big Lottery Research section), through Action on Addiction | Cost-effectiveness was assessed as ICERs, QALYs, and estimates from CEAC at defined willingness-to-pay levels. The time horizon was 26 weeks, and the analysis was based on a broad societal perspective that included health, social services, and the criminal justice sector. | Adult patients with chronic heroin addiction (n = 127) who have been on conventional oral methadone maintenance treatment for at least 6 months but continued to inject street heroin regularly (≥ 50% of days in preceding 3 months) Mean (SD) age: 37.2 (6.5) years Age, mean (SD): 37.2 (6.5) years Male, n (%): 93 (73) Years injecting drugs, mean (SD) 13.7 (7.8) years | Injectable heroin vs. injectable methadone or optimized oral methadone | Economic analysis used an unnamed, pre-specified decision-making approach. Joint distribution of incremental mean costs and effects for the treatments under comparison was generated to explore the probability that each was the optimal choice about a decision-maker’s willingness-to-pay for an additional QALY | The clinical and costs data were derived from the RIOTT trial supplemented with external sources (e.g., the British National Formulary and treatment-specific pharmacy cost weighted by time spent ordering, preparing, and managing the distribution of each of the treatments). | The baseline characteristics of the participants in the RIOTT trial were assumed to be representative of the larger population of adult chronic heroin users. The cost of random urine tests was estimated assuming bi-weekly tests |
Nosyk et al., 201215 Canada Funded by: Canadian Institutes of Health Research, Canada Foundation for Innovation, The Canada Research Chairs Program, The University of British Columbia, Providence Health Care, Université de Montréal, Centre de recherche et d’aide pour narcomanes, The Government of Quebec, Vancouver Coastal Health and The BC Centre for Disease Control | Incremental cost-effectiveness ratio, interpreted as the incremental cost per QALY gained, were assessed from a broad societal perspective, including health care, criminal justice systems, and out-of-pocket costs borne by society. Time horizons were 1, 5 and 10 years, as well as the lifetime of the hypothetical cohort | Adult patients (≥ 25 years, n = 251) with regular opioid injection use who participated in the NAOMI trial. They should have had ≥ 2 previous opioid substitution treatment attempts and been out of treatment for at least 6 months before trial entry. Age, mean (SD):b 39.7 (8.6) years Male, n (%):b 154 (61.4) Years injecting drugs, mean (SD):b 16.5 (9.85) Number of previous drug treatments, median (IQR):b 7 (4 to 14) | Injectable diacetyl-morphine vs. methadone maintenance treatment | A decision-analytical model that applied a semi-Markov cohort model | The clinical and costs data were derived from the NAOMI trial supplemented with the literature– preferably published data on Canadian cohorts or local data sources. | A hypothetical cohort of patients assigned the baseline characteristics of the participants in the NAOMI trial and assumed to represent the population in North American that would be reached by diacetylmorphine treatment. It was assumed that all patients were in their third treatment attempt. Life-time analysis was based on average additional years lived following entry into the model of 14.54 and 15.45 for patients treated with MMT and DAM, respectively |
CEAC = cost-effectiveness acceptability curve; DAM = diacetylmorphine; HDM = hydromorphone; ICER = incremental cost-effectiveness ratio; MMT = methadone maintenance treatment; NAOMI = North American Opiate Medication Initiative; NR = not reported; QALYs = quality-adjusted life-years; RCT = randomized controlled trial; RIOTT = Randomized Injectable Opiate Treatment Trial; SALOME = Study to Assess Long-term Opioid Maintenance Effectiveness; SD = standard deviation.
Note that this table has not been copy-edited.
aSourced from an article on the SALOME trial, Oviedo-Joekes et al., 20161
bSourced from a report on the NAOMI trial, Oviedo-Joekes et al., 20082
Table 3: Characteristics of Included Guidelines
Intended users, target population | Intervention and practice considered a | Major outcomes considered a | Evidence collection, selection, and synthesis | Evidence quality assessment | Recommendations development and evaluation | Guideline validation |
|---|---|---|---|---|---|---|
The CCSMH Canadian Guidelines on Opioid Use Disorder Among Older Adults – Rieb et al., 202016 | ||||||
Intended users: Health care workers and policy-makers working with older adults within the Canadian context Target population: Older adults (≥ 65 years) with OUD | Preventive measures to reduce the risk of developing OUD in older adults or opioid overdose in older adults with or at risk for an OUD Measures to screen for and assess OUD in older adults OUD treatments for older adults
| Effectiveness of preventive, screening and assessment measures, or interventions to curb opioid poisonings and the development of OUD in older adults | A systematic literature search for relevant studies, published 2008 to 2018, related to opioid use and opioid use disorder in adults and older adults | Guidelines were evaluated using AGREE II Recommendations were created and assessed using the GRADE method. | Existing guidelines were customized for older adults and the Canadian context using the ADAPTE collaboration process with information supplemented by evidence from current literature. Guideline working group members drafted recommendations with supporting evidence, An individual with a lived experience provided views and preferences of the target population. The wording of each final recommendation was determined through full (100%) consensus. The GRADE system evaluated each recommendation The quality of the evidence was scored as high, moderate, low, or very low.a The strength of each recommendation was rated as strong or weak. | The final recommendations were externally peer-reviewed, and most external reviewers’ comments were incorporated in the guidelines after discussion. |
The CRISM National Injectable Opioid Agonist Treatment Guideline – Fairbairn et al., 201917 | ||||||
Intended Users: Canadian health professionals Target Population: Adults with severe OUD who inject opioids for whom previous oral opioid agonist treatment was unsuccessful or whose circumstances and risks indicate that they may benefit from injectable opioid agonist treatment | Injectable opioid agonist treatment for the clinical management of severe OUD in adults | Reduction in illicit heroin or any street opioid use, criminal activity, and involvement in sex work. Improvement in treatment retention rates, overall health, and social functioning, premature death, nonfatal overdose, blood-borne infectious diseases, violence, and arrest. | A systematic literature search for relevant studies in multiple databases and reference lists, up to August 1, 2018 It was supplemented with grey literature searches, and engagement with international researchers and other experts in the field were conducted for existing guidelines on using injectable opioid agonist to treat OUD | Evidence sources were ranked such that SR and meta-analyses were given the most weight, followed in order by RCTs, quasi-experimental studies, observational studies, and expert opinion | Draft recommendations were developed and graded using the GRADE approach. Differences in opinion or interpretation about the recommendations or the evidence review were resolved through an iterative consensus process facilitated by the guideline review committee through teleconference or direct communication. A final decision was reached for all cases without the need for arbitration. | The draft recommendations and evidence were review by the guideline review committee. Revisions were confirmed as needed by the guideline review committee co-chairs and medical writer feedback. External reviews and input for the final draft were provided by the National Injectable Opioid Agonist Treatment Operational Guidance Review Committee, 10 international experts, individuals with lived OUD experience, and 1 family member affected by OUD |
AGREE II = Appraisal of Guidelines for Research and Evaluation II; CCSMH = Canadian Coalition for Seniors Mental Health; CRISM = Canadian Research Initiative in Substance Misuse; GRADE = Grades of Recommendation Assessment, Development, and Evaluation; OUD = opioid use disorder; SR = systematic review; SROM = slow-release oral morphine.
Note that this table has not been copy-edited.
aWhere a guideline discusses several interventions and outcomes, this report will focus on the relevant section related to the use of sustained-release oral morphine, oral hydromorphone, injectable hydromorphone, injectable prescription diacetylmorphine, fentanyl patches or fentanyl buccal tablets, as applicable, for opioid abuse treatment.
Appendix 3: Critical Appraisal of Included Publications
Note that this appendix has not been copy-edited.
Table 4: Strengths and Limitations of Economic Evaluations Using the Drummond Checklist12
Strengths | Limitations |
|---|---|
Bansback et al., 201814 | |
Study design
Data collection
Analysis and interpretation
|
|
Byford et al., 20137 | |
Study design
Data collection
Analysis and interpretation
|
|
Nosyk et al., 201215 | |
Study design
Data collection
Analysis and interpretation
|
|
OUD = opioid use disorder.
Table 5: Strengths and Limitations of Guidelines Using AGREE II13
Item | Rieb et al., 202016 | Fairbairn et al., 201917 |
|---|---|---|
Domain 1: Scope and Purpose | ||
1. The overall objective(s) of the guideline is (are) specifically described. | Yes | Yes |
2. The health question(s) covered by the guideline is (are) specifically described. | Yes | Yes |
3. The population (patients, public, etc.) to whom the guideline is meant to apply is specifically described. | Yes | Yes |
Domain 2: Stakeholder Involvement | ||
4. The guideline development group includes individuals from all relevant professional groups. | Yes | Yes |
5. The views and preferences of the target population (patients, public, etc.) have been sought. | Yes | Yes |
6. The target users of the guideline are clearly defined. | Yes | Yes |
Domain 3: Rigour of Development | ||
7. Systematic methods were used to search for evidence. | Yes | Yes |
8. The criteria for selecting the evidence are clearly described. | Unclear | Yes |
9. The strengths and limitations of the body of evidence are clearly described. | Yes | Yes |
10. The methods for formulating the recommendations are clearly described. | Yes | Yes |
11. The health benefits, side effects, and risks have been considered in formulating the recommendations. | Yes | Yes |
12. There is an explicit link between the recommendations and the supporting evidence. | Yes | Yes |
13. The guideline has been externally reviewed by experts before its publication. | Yes | Yes |
14. A procedure for updating the guideline is provided. | Yes | Yes |
Domain 4: Clarity of Presentation | ||
15. The recommendations are specific and unambiguous. | Yes | Yes |
16. The different options for management of the condition or health issue are clearly presented. | Yes | Yes |
17. Key recommendations are easily identifiable. | Yes | Yes |
Domain 5: Applicability | ||
18. The guideline describes facilitators and barriers to its application. | Yes | Yes |
19. The guideline provides advice and/or tools on how the recommendations can be put into practice. | Unclear | Yes |
20. The potential resource implications of applying the recommendations have been considered. | Yes | Yes |
21. The guideline presents monitoring and/or auditing criteria. | Yes | Yes |
Domain 6: Editorial Independence | ||
22. The views of the funding body have not influenced the content of the guideline. | Yes | Yes |
23. Competing interests of guideline development group members have been recorded and addressed. | Yes | Yes |
AGREE II = Appraisal of Guidelines for Research and Evaluation II.
Appendix 4: Main Study Findings and Authors’ Conclusions
Note that this appendix has not been copy-edited.
Table 6: Summary of Findings of Included Economic Evaluations
Main study findings | Authors’ conclusion |
|---|---|
Bansback et al., 201814 | |
Within-trial analysis – injectable HDM vs. injectable heroin (DAM); time horizon– 6 months
Lifetime analysis – HDM vs. DAM vs. MMT
|
|
Byford et al., 20137 | |
DAM vs. Oral methadone vs. Injectable methadone; time horizon– 26 weeks
A CEAC plot showed that at the usual willingness-to-pay threshold of £30,000 per QALY:
DAM had a probability of being more cost-effective than injectable methadone at higher willingness-to-pay levels ≥ £70,000 | “Our results do not support the continuing provision of oral methadone maintenance treatment alone for chronic refractory heroin addiction, despite the relatively low treatment costs in comparison to injectable alternatives. However, policymakers will need to compensate clinics for providing a more expensive service that generates cost savings primarily for the criminal justice sector. The choice of which injectable treatment to provide is less clear. There is currently evidence to suggest superior effectiveness of supervised injectable heroin but at a cost that policy makers may find unacceptable.”7 P.384 |
Nosyk et al., 201215 | |
MMT vs. DAM; time horizons– 1, 5, 10 years, and lifetime At 1 year
At 5 years
At 10 years
Lifetime
Sensitivity analysis showed that DAM's cost-effective probability was 76% at a willingness-to-pay threshold of $0 per QALY gained and 95% at a $100 000 per QALY gained. | “Using mathematical modelling to extrapolate results from the North American Opiate Medication Initiative, we found that a treatment strategy featuring diacetylmorphine may be more effective and less costly than methadone maintenance treatment among people with chronic opioid dependence refractory to treatment. Our model indicated that diacetylmorphine would decrease societal costs, largely by reducing costs associated with crime, and would increase both the duration and quality of life of treatment recipients.” P.E326 |
CEAC = Cost-effectiveness acceptability curve; CI = confidence intervals; CN$ = Canadian dollars; Crl = credible interval DAM = diacetylmorphine; HDM = hydromorphone; ICER = incremental cost-effectiveness ratio; MMT = methadone maintenance treatment; OUD = opioid use disorder; QALY = quality-adjusted life-years; SD = standard deviation; vs. = versus.
Table 7: Summary of Recommendations in Included Guidelines
Recommendations and supporting evidence | Quality of evidence | Strength of recommendations |
|---|---|---|
Rated on GRADE scale | ||
The CCSMH Canadian Guidelines on Opioid Use Disorder Among Older Adults – Rieb et al., 202016 | ||
“Opioid withdrawal management should only be offered in the context of connection to long-term addiction treatment.” P.126 Supporting evidence came from 12 publications, including at least 4 previous guidelines and 1 systematic review | Moderate (i.e., moderated confidence because the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different) | Strong |
“If renal function is adequate, daily witnessed ingestion of SROM may be considered with caution for those older adults in whom buprenorphine and methadone maintenance have been ineffective or could not be tolerated. Careful supervision of initiation onto short-acting morphine first is recommended, prior to transition to maintenance with the long-acting 24-hour formulation.” P.126-127 Supporting evidence came from 3 publications– 1 guideline, 1 consensus statement of an International Expert Panel, and 1 animal study. | Low (i.e., limited confidence because the true effect may be substantially different from the estimate of the effect) | Weak |
The CRISM National Injectable Opioid Agonist Treatment Guideline – Fairbairn et al., 201917 | ||
“Injectable opioid agonist treatment should be considered for individuals with severe, treatment-refractory opioid use disorder and ongoing illicit injection opioid use.” P. E1052 Supporting evidence came from 2 SRs (each with meta-analyses) and 1 RCT | Moderate (i.e., as defined above) | Conditionala |
“For patients who are determined to be likely to benefit from injectable opioid agonist treatment, both diacetylmorphine and hydromorphone are acceptable treatment options.” P. E1052 Supporting evidence came from 2 SRs (each with meta-analyses) and 1 RCT | Low (i.e., as defined above) | Strongb |
“Injectable opioid agonist treatment should be provided as an open-ended treatment, with decisions to transition to oral opioid agonist treatment made collaboratively with the patient.” P. E1054 | Low (i.e., as defined above) | Strongc |
GRDAE = Grading of Recommendations Assessment, Development and Evaluation; RCT = randomized controlled trial; SR = systematic review.
aRated conditional by authors because some patients would find the attendance requirements for injectable opioid agonist treatment onerous or otherwise not have their needs met by injectable opioid agonist treatment.
bRated strong authors based on expert consensus, substantial clinical experience in British Columbia, reduced risk of adverse events for hydromorphone compared with diacetylmorphine (heroin), and the lack of regulatory and supply barriers affecting access to hydromorphone.
cRated strong by authors despite the low quality of evidence, owing to the risk associated with fentanyl-contaminated illicit opioid use and its alignment with a recommendation from the WHO that opioid agonist treatment be provided as an open-ended treatment.
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up to date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third-party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.
Questions or requests for information about this report can be directed to Requests@CADTH.ca