CADTH Health Technology Review
Treat and Release for Patients Requiring Emergency Medical Services
Rapid Review
Authors: Calvin Young, Jennifer Horton
Abbreviations
AMSTAR 2
A MeaSurement Tool to Assess systematic Reviews 2
CI
confidence interval
HTA
health technology assessment
MCS
mental component summary
MFES
Modified Falls Efficacy Scale
OR
odds ratio
PCS
physical component summary
QALY
quality-adjusted life-year
RCT
randomized controlled trial
SD
standard deviation
SF-12
Short Form Health Survey (12 items)
Key Messages
“Treat and release” and “treat and refer” protocols or practices refer to the onsite treatment of patients by responding emergency medical services personnel that does not involve transporting patients to health care facilities for additional assessment and treatment. The goal of these protocols is to allow patients to be released from care or to be referred directly to non-emergency services by emergency medical services personnel when appropriate, diverting patients from emergency departments.
One health technology assessment that included a relevant randomized controlled trial and economic evaluation and 2 non-randomized studies were identified for inclusion. These studies examined treat and release or treat and refer protocols for treating hypoglycemia and exertional heat stroke, and for attending to older people following a fall.
Overall, the clinical evidence summarized in this report suggests that treat and release protocols are as good as, or better than, usual care (i.e., onsite treatment of immediate medical care followed by transportation to health care facilities). Across most reported outcomes, there were no significant differences between patients who received care using treat and release or treat and refer protocols, and those who received usual care; however, there were some instances where the use of these protocols was associated with improvements in some clinical outcomes, such as patient satisfaction, risk for future falls or fractures, and some measures of repeat access to health care services.
Findings related to the cost-effectiveness of treat and refer protocols were inconclusive because of the limited generalizability of the findings from the included economic evaluation. The economic evaluation estimated that implementing a treat and refer protocol for older patients who experienced a fall did not result in significant changes to health care resource utilization and did not generate improved health-related quality of life compared to usual care.
No evidence-based guidelines regarding the use of treat and release protocols for patients requiring emergency medical services were identified.
Context and Policy Issues
The health care systems of many countries around the world are facing ever-increasing emergency department overcrowding.1,2 In Canada, the number of emergency department visits per year has grown over time and is out of proportion to population growth.3,4 The growing burden of chronic disease and population aging, shortages of health care providers, an increasing number of visits by patients with serious or complex health conditions, and high volumes of low-acuity presentations (i.e., patients with a relatively low need for urgent medical care) are some of the suspected causes of this major global health care issue.5,6 Consequences of emergency department overcrowding include increased stress among care providers,6 increased length of hospital stay,7 increased violence toward staff,6,8 increased risk of patient readmission,9 reduced patient satisfaction,10 increased risk of medication errors,11 and increased risk of mortality and poor health outcomes.2,5,9,12
One strategy proposed to help alleviate emergency department overcrowding has been the development and implementation of treat and release protocols or policies that provide emergency medical services personnel with more flexibility in how they provide care for patients. Emergency medical services personnel act as the first point of contact for many patients who access emergency health care services. Their role is typically to administer immediate medical care followed by transportation of the patient to emergency departments for further assessment and treatment. However, treat and release or treat and refer protocols enable emergency medical services personnel to assess, treat, and discharge patients at the scene or to refer them directly to specialized, non-emergency health care services.13 While the intention of these policies is not to divert all patients seen by emergency medical services personnel, these policies encourage first responders to consider alternative options for patients, when appropriate, particularly for patients with low-acuity conditions such as minor trauma, hypoglycemia, minor epistaxis, and non-threatening exacerbations of chronic conditions or pain syndromes.13-16
In 2014, CADTH assessed the literature regarding treat and release protocols for patients requiring emergency medical services;17 however, little evidence was identified at that time, thus limiting the conclusions that could be drawn.17 The objective of the current report is to review the literature published since the 2014 CADTH report17 regarding the clinical and cost-effectiveness of treat and release protocols for patients requiring emergency medical services. Additionally, evidence-based guidelines regarding the use of treat and release protocols for patients requiring emergency medical services will be reviewed.
Research Questions
What is the clinical effectiveness of treat and release protocols for patients requiring emergency medical services?
What is the cost-effectiveness of treat and release protocols for patients requiring emergency medical services?
What are the evidence-based guidelines regarding the use of treat and release protocols for patients requiring emergency medical services?
Methods
Literature Search Methods
A limited literature search was conducted by an information specialist on key resources including MEDLINE, the Cochrane Database of Systematic Reviews, the international HTA database, the websites of Canadian and major international health technology agencies, as well as a focused internet search. The search strategy comprised both controlled vocabulary, such as the National Library of Medicine’s MeSH (Medical Subject Headings), and keywords. The main search concepts were emergency medical services and treat and release programs. Search filters were applied to limit retrieval to health technology assessments (HTAs), systematic reviews, meta-analyses, or network meta-analyses; any types of clinical trials or observational studies; economic studies; and guidelines. Where possible, retrieval was limited to the human population. The search was also limited to English-language documents published between January 1, 2014 and March 29, 2021.
Selection Criteria and Methods
One reviewer screened citations and selected studies. In the first level of screening, titles and abstracts were reviewed and potentially relevant articles were retrieved and assessed for inclusion. The final selection of full-text articles was based on the inclusion criteria presented in Table 1.
Criteria | Description |
Population | Patients with lower acuity who contact emergency medical services; for example, those who have:
|
Intervention | Treat and release protocols or policies; treat and refer (e.g., “Assess/Treat/Refer”) protocols or policies; the use of emergency care practitioners with referral and on-scene discharge capabilities; treat and no transport; treatment or transport refusal; assess and refer; assess and release; assess, treat, and release |
Comparator | Transfer to emergency department for all patients (e.g., treat and transfer); standard of care (if the patient requests hospital transport, they have to be transported; i.e., up to the patient’s discretion) |
Outcomes | Q1: Clinical effectiveness (e.g., safety, risks, number of hospitalizations, emergency department overcrowding) Q2: Cost-effectiveness (e.g., quality-adjusted life-years, incremental cost-effectiveness ratios) Q3: Recommendations regarding the use of treat and release protocols in the emergency medical service; recommendations regarding the appropriate patients for treat and release protocol |
Study designs | HTAs, SRs, RCTs, non-randomized studies, economic evaluations, and evidence-based guidelines |
Q = question; HTA = health technology assessment; RCT = randomized controlled trial; SR = systematic review.
Exclusion Criteria
Articles were excluded if they did not meet the selection criteria outlined in Table 1, they were duplicate publications, they had been included in 2014 CADTH report17 on treat and release protocols, or they were published before 2014. Systematic reviews in which all relevant studies were captured in other more recent or more comprehensive systematic reviews were excluded. Primary studies retrieved by the search were excluded if they were captured in 1 or more included systematic review. Guidelines with unclear methodology were also excluded.
Critical Appraisal of Individual Studies
The included publications were critically appraised by 1 reviewer using the following tools as a guide: A MeaSurement Tool to Assess systematic Reviews 2 (AMSTAR 2)18 for systematic reviews, the Downs and Black checklist19 for randomized and non-randomized studies, and the Drummond checklist20 for economic evaluations. Summary scores were not calculated for the included studies; rather, the strengths and limitations of each included publication were described narratively.
Summary of Evidence
Quantity of Research Available
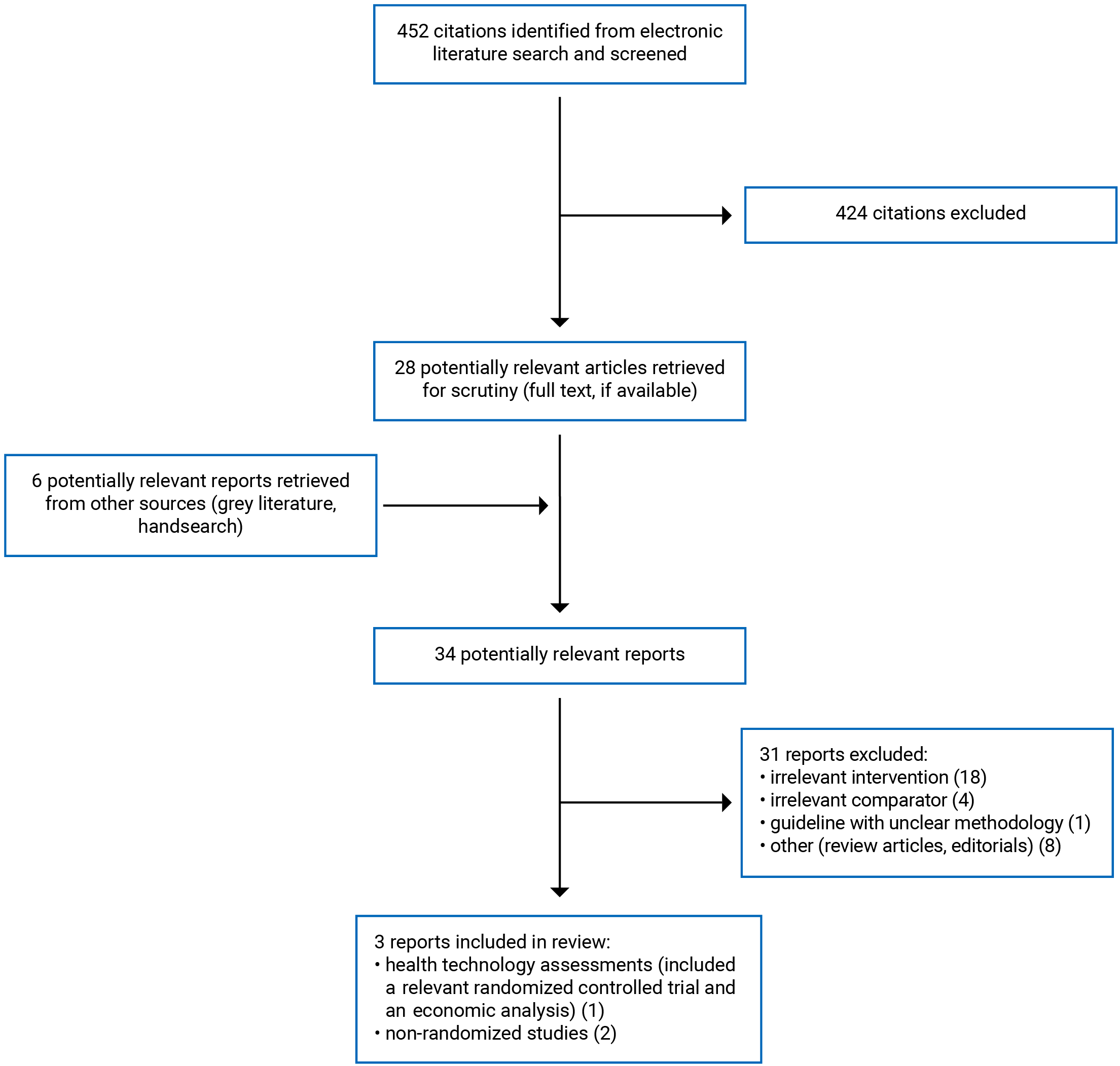
A total of 452 citations were identified in the literature search. Following the screening of titles and abstracts, 424 citations were excluded and 28 potentially relevant reports from the electronic search were retrieved for full-text review. Six potentially relevant publications were retrieved from the grey literature search for full-text review. Of these potentially relevant articles, 31 publications were excluded for various reasons and 3 publications met the inclusion criteria and were included in this report. These comprised 1 HTA21 (that included a systematic review, a randomized controlled trial [RCT], and an economic evaluation) and 2 non-randomized studies.22,23 Appendix 1 presents the PRISMA24 flow chart of the study selection. Additional references of potential interest are provided in Appendix 5.
Summary of Study Characteristics
One HTA21 (that included a systematic review, an RCT, and an economic evaluation) and 2 non-randomized studies22,23 were identified for inclusion in this review. Detailed study characteristics are available in Appendix 2, Table 2, Table 3, and Table 4.
The systematic review that was included in the HTA21 had objectives and inclusion criteria that were broader than the current report (i.e., wider in scope). Specifically, the systematic review21 included studies of any interventions or enhanced practices applied by emergency medical services that aimed to reduce demand for emergency departments, rather than just treat and release protocols. The systematic review also examined a wider range of relevant comparators than those considered relevant to the current report (i.e., any comparator or no comparator was deemed eligible for inclusion in the review). None of the studies included in the systematic review evaluated the comparison of interest for this report, namely treat and release or treat and refer protocols or policies compared to standard of care (i.e., transportation of patients to hospitals).
Study Design
The RCT, which was conducted as part of the HTA,21 was performed at 25 ambulance stations across 3 study sites. Randomization was done using a cluster design (using ambulance stations as units of clustering) as the intervention involved providing additional training to paramedics that could not be switched on and off as needed for a patient-level RCT. Participants were enrolled between March 2011 and June 2012. The follow-up duration was 6 months.
The 2 non-randomized studies were retrospective cohort studies. Sinclair et al. (2019)22 included data from patients treated between January 1, 2011 and December 31, 2011, while Sloan et al. (2015)23 included data from patients treated between 2005 to 2012.
The economic evaluation was conducted as a cost-utility analysis alongside the included RCT as part of the HTA.21 The evaluation used a 6-month time horizon from the perspective of the UK National Health Service and Personal Social Services. Effectiveness inputs, which were used to derive utility values, and use of health care and social services resources were obtained from routine hospital records and patient-completed questionnaires that were collected during the RCT. Costs were calculated using published unit costs from various sources (e.g., National Health Service Reference Costs 2011 to 2012 and the Unit Costs of Health and Social Care 2013).
Country of Origin
The RCT and economic evaluation that were conducted as part of the HTA were authored by a group based in the UK.21 The non-randomized studies were conducted in Canada22 and the US.23
Patient Population
The RCT21 enrolled 4,704 people, aged 65 years and older (mean age, 82.35 years), who were attended by a study paramedic following an emergency call to the ambulance service after a fall without priority symptoms (i.e., serious conditions in addition to the fall). The proportion of female participants within the study population was 62.9%. Because the RCT provided the clinical inputs to the economic evaluation, the participants in the RCT were the same participants in the economic evaluation.21
The non-randomized study by Sinclair et al. (2019) 22 included data from 791 adults (18 years of age and older), with or without a history of diabetes, who were assessed by paramedics for hypoglycemia within the city of Ottawa, Ontario, Canada. Individuals who were younger than 18 years of age, required supraglottic airways or intubation, had absent vital signs, were terminally ill, or required palliative care were excluded. The mean age of study participants was 56.2 years and the proportion of female participants was 47.7%. The Sloan et al. (2015)23 non-randomized study included 32 patients from the Indianapolis half-marathon between 2005 and 2012 who had developed exertional heat stroke, which was defined as having a core temperature of higher than 102 °F and an altered mental status. The age and sex or gender of participants were not reported.
Interventions and Comparators
In the RCT,21 patients received emergency care from paramedics who used a clinical protocol that enabled them to assess and refer patients directly to community-based falls services when appropriate (i.e., a treat and refer protocol) or to convey patients to the emergency department as needed. Or, patients received emergency care from paramedics who did not receive additional training on the novel clinical protocol and were asked to continue their usual practice (which comprised assessment of injury or other conditions requiring immediate care followed by conveyance to the emergency department unless the patient refused). The economic evaluation21 assessed the cost-effectiveness of these 2 paramedic assessment strategies.
Participants of the non-randomized study by Sinclair et al. (2019)22 received paramedic assessment and treatment for hypoglycemia at the scene followed by either patient release due to refusal of transport to a hospital (i.e., treat and release group) or transport to hospital (i.e., standard of care group). The intervention investigated in the Sloan et al.23 non-randomized study was an exertional heat stroke protocol that enabled emergency medical services at the Indianapolis half-marathon to treat patients onsite using cooling tubs and IV saline infusion. Patients who reached the cooling target (core temperature of approximately 102 °F) and had a return-to-normal mental status could be discharged by the emergency medical services staff. Patients who reached the cooling target and still had altered mental status or abnormal vital signs were transported by emergency medical services to the hospital. Participants in the control group of the non-randomized study by Sloan et al. (2015)23 were immediately transported to the nearest hospital for the offsite treatment of exertional heat stroke (i.e., usual care). Patients were assigned to the treat and release or usual care groups, depending on their location (i.e., section of the half-marathon course) at the time they required medical attention. Specifically, patients who experienced emergent conditions within 1 mile of the finish line or after race completion were treated onsite and all other patients were transported to the nearest hospital for offsite treatment.
Outcomes
The included RCT21 and 2 non-randomized studies22,23 reported on various measures of clinical effectiveness:
repeat access to health care services,21,22 which included the proportion of patients with further emergency calls (i.e., calls to paramedic services) or prehospital service,21,22 emergency department attendance,21,22 and emergency department or hospital admission;21-23 additionally, the authors of the RCT21 reported on the mean number of further emergency department attendances per patient, the mean number of further emergency service calls per patient (i.e., calls to paramedic services), and the mean duration of subsequent inpatient episodes (nights in hospital) following initial emergency medical services assessment
emergency department conveyance by emergency medical services21
hospital admissions following initial assessment by emergency medical services23
referral to non-emergency department services,21 including the proportion of patients who were referred to falls services by the emergency service crew following the initial assessment21 and the proportion of patients who were left at the scene without referral following the initial assessment21
quality of life, which was measured in the RCT21 using the Short Form Health Survey, 12 items (SF-12). The SF-12 is a validated tool that evaluates 8 health concepts: physical functioning, role-physical, bodily pain, general health, energy/fatigue, social functioning, role-emotional, and mental health.25 The tool generates 2 separate summary scores (i.e., physical component summary [PCS] scores and mental component summary [MCS] scores).25,26 Total scores range from 0 to 100, with higher scores indicating increased quality of life26
patient satisfaction, measured in the RCT21 using the Quality of Care Monitor (QCM), which is a validated questionnaire that can be used to measure patient perceptions of quality of care in both inpatients and outpatients.27 The QCM includes 2 subscales: the QCM Technical and the QCM Interpersonal. No information on the scoring of the QCM was available in the RCT21
fear of falling21 was evaluated by the authors of the RCT21 using the Modified Falls Efficacy Scale (MFES). The MFES is a validated 14-item questionnaire that asks users to score each item between 1 (not confident at all) and 10 (completely confident) using a visual analogue scale.28,29 Total scores are the average of scores for each of the 14 items and thus range between 0 (less confidence and more fear of falling) and 10 (more confidence and less fear of falling)28
adverse events,21 which included the proportion of patients who experienced further fractures21 and the proportion of patients who had an emergency service call,21 attended the emergency department,21 had an emergency admission,21 or who died within 2 days following initial emergency medical services assessment.21
In the RCT,21 data for all outcomes were adjusted for patient characteristics (i.e., age and gender), ambulance site, distance to the emergency department from the point of assessment, recruitment point (time since the start of the study), seasonality, and whether or not the index call was made out of general practitioner hours. Unadjusted data were presented, with statistical comparisons from the adjusted data.
The economic evaluation21 reported on costs of the intervention and comparator (reported in £), quality-adjusted life-years (QALYs), which were derived from SF-12 scores measured in the RCT, and measures of cost-effectiveness (i.e., cost per QALY gained).
Outcomes assessed in the systematic review,21 which did not identify any primary studies relevant to the current report, are described in Appendix 2, Table 2
Summary of Critical Appraisal
Additional details regarding the strengths and limitations of the included publications are provided in Appendix 3, Table 5, Table 6, and Table 7.
Systematic Review
The systematic review21 was considered to be of high methodological quality based on the assessment using AMSTAR 2.18 The review had clearly defined objectives and eligibility criteria, literature searches were conducted in multiple databases (i.e., 18 electronic databases, including both traditional and grey literature sources), and provided a description of key search terms, search strategies, and search restrictions (e.g., only studies published in English between 1990 and 2013 were eligible for inclusion). Additionally, a flow chart of study selection and a list of studies excluded after full-text review were provided. These features increase the reproducibility of the systematic review. The review methods were established before conducting the review and protocols were registered with PROSPERO and the All Wales Systematic Review Register, decreasing the risk for selective reporting and increasing transparency in the review process. The methods for article selection and data extraction were well-documented and were conducted involving multiple reviewers, decreasing the likelihood for inconsistency in these processes. The review authors declared potential conflicts of interest and reported sources of funding, both of which were considered unlikely to have influenced the findings of the review.
As for methodological limitations, the authors of the systematic reviews provided no justification for the selection of eligible study designs and did not report on the sources of funding for the included primary studies. Additionally, it was unclear if quality assessment was conducted in duplicate.
Randomized Controlled Trial
The included RCT21 had clearly described objectives, intervention, comparator, main outcomes, participant eligibility criteria, and findings. The characteristics of participants — such as age, sex, and distance to the emergency department — were described for both intervention and control groups. While the authors did not report on the statistical significance of any between-group differences for these baseline characteristics, the 2 groups appeared to be balanced, suggesting randomization was successful. Additional methodological strengths were that patients in the intervention and control groups were recruited over the same period of time (between March 14, 2011 and June 30, 2012); outcome measures were valid; estimates of random variability (e.g., standard deviations [SDs] and confidence intervals [CIs]), and actual P values, were reported; the trial statistician was blinded to participant allocations; the length of follow-up between the intervention and control groups was consistent; a power calculation was used to inform the number of participants required; and study participants, care providers, and care settings appeared to be representative of those of interest. The study authors declared potential conflicts of interest and reported sources of funding, both of which were considered unlikely to have influenced the findings of the study.
One methodological limitation of the RCT21 was that it was not possible to blind paramedics, trial managers, and site researchers because of the nature of the intervention. As a result, there was a risk for bias in either direction depending on the perceptions of those involved in the trial. Additionally, a substantial proportion of study participants (i.e., 72.2% [3,397 of 4,704]) did not respond to 1-month questionnaires that assessed quality of life and fear of falling. Although questionnaire response rates did not significantly vary across treatment groups, non-responders were statistically significantly older than those who responded to the questionnaires (82.74 years versus 81.33 years respectively, P < 0.001). The results of this RCT21 have unclear generalizability to Canadian settings given the RCT involved patients who received care from 3 ambulance services in the UK.
Non-Randomized Studies
Both non-randomized studies22,23 had clearly described objectives, interventions, comparators, participant eligibility criteria, and findings. While the authors of the Sinclair et al. (2019)22 study provided a clear description of main outcomes and patient characteristics (e.g., age, sex, medical history, current medications, vital signs), Sloan et al.23 had poorly described main outcomes and did not report several important patient characteristics, such as age and sex or gender. In both studies,22,23 the intervention assignment was not randomized. In the study by Sinclair et al.,22 participants who refused transportation to the hospital were designated the intervention group. There were significant differences between those who were transported to the hospital compared to those who refused transport with respect to age, sex, history of diabetes, use of insulin therapy or corticosteroids, location of paramedic assessment, and drug and/or alcohol dependencies. Similarly, participants in the Sloan et al.23 study were assigned to intervention and control groups based on their location (i.e., section of the half-marathon course; patients who experienced emergent conditions within 1 mile of the finish line or after race completion were treated onsite) at the time they required medical attention. As a result, both non-randomized studies22,23 were at a high risk of bias due to confounding. Additional methodological limitations included the fact that adverse events related to the treatment protocols may have been missed, neither study22,23 included a power calculation, and the authors of the study by Sloan et al.23 did not report the statistical significance of the main findings and did not explicitly state potential conflicts of interest. Finally, the generalizability of these studies to Canadian settings was unclear, as 1 was conducted in the US23 and the other was conducted at a single geographic location in Canada.22
Methodological strengths common to both studies22,23 included:
No participants were lost to follow-up because of the nature of the studies.
Patients in the intervention and control groups were recruited over the same periods of time.
The study participants, care providers, and settings appeared to be representative to those of interest (increasing external validity).
The study authors disclosed their sources of funding, which were considered unlikely to have influenced the findings of the studies.
Compliance with the intervention was considered reliable in the Sinclair et al.22 study, as participants were assigned to the intervention and comparator group based on the actual intervention they received (i.e., patients who were discharged at the scene were analyzed in the treat and release group; patients who were transported to hospital were analyzed in the standard of care group). Conversely, compliance with the intervention was unclear in the Sloan et al.,23 study as there were no data regarding the level of compliance with the treat and release protocol that the study aimed to evaluate.
Economic Evaluation
The economic evaluation21 included clear descriptions of the research questions, objectives, economic importance of the research questions, time horizons (i.e., 6 months), treatment strategies being compared, rationale for choosing alternative interventions, and the perspective of the analysis. The sources of effectiveness estimates (i.e., the RCT separately described in the current report) were provided and were described in sufficient detail, including measures used to derive health utility values and treatment cost estimates. The primary outcome of the economic evaluation was stated, characteristics of study participants from which effectiveness estimates were drawn were provided, and quantities of resource use were reported separately from their unit costs. These methodological strengths increased confidence in the reporting of methods used by the authors. The authors of the economic evaluation21 described the approach to sensitivity analyses, justified the decision to not apply a discount rate, reported incremental analyses, provided an answer to the study question, summarized the findings with conclusions accompanied by appropriate caveats, declared potential conflicts of interest, and disclosed sources of funding (which were considered unlikely to have influenced the findings of the study).
Methodological limitations of the economic evaluation21 included:
No description of currency price adjustments for inflation was provided.
The estimates of intervention effectiveness estimates were taken from a single RCT, rather than from a synthesis or meta-analysis of estimates from multiple sources or studies.
Sensitivity analyses were only based on the upper and lower bounds of the 95% CIs for both net costs and net QALYs.
The generalizability of the findings to Canadian settings was unclear, as the analysis was conducted in the UK from the perspective of the UK National Health Service and Personal Social Services, and any changes in the cost of implementing the intervention or any associated health care resource uses may vary greatly.
Summary of Findings
The overall findings of the included studies are subsequently highlighted. Detailed summaries of the main findings and authors’ conclusions are available in Appendix 4.
Clinical Effectiveness of Treat and Release Protocols for Patients Requiring Emergency Medical Services
Evidence regarding the clinical effectiveness of treat and release protocols for patients requiring emergency medical services was available from 1 RCT21 and 2 non-randomized studies.22,23
Repeat Access to Health Care Services
Patients Who Experienced a Fall
The authors of the RCT21 reported several measures of repeat access to health care services in patients who were treated for falls using a treat and refer protocol and those who were treated using usual care. At 1-month follow-up, a significantly smaller proportion of participants treated using the treat and refer protocol had further emergency service calls (i.e., calls to paramedic services) following initial assessment compared to those in the usual care group after adjusting for patient characteristics (i.e., age and gender), ambulance site, distance to the emergency department from the point of assessment, recruitment point (time since the start of the study), seasonality, and whether or not the index call was made out of general practitioner hours (18.5% and 21.8%, respectively; adjusted P = 0.006). Additionally, patients treated with the treat and refer protocol had significantly fewer further emergency service calls 1 month after the initial assessment compared to those who received usual care, whether further emergency service calls were expressed as “mean calls per patient” or “mean calls per patient per day at risk” (i.e., when days in hospital or after death were excluded). At 6-month follow-up, compared to those in the usual care group, patients treated using the treat and refer protocol had significantly fewer further emergency service calls (expressed as calls per patient per day at risk) and fewer further emergency department attendances.
There were no statistically significant between-group differences at 1-month or 6-month follow-ups for all other measures of repeat access to health care services, including the proportion of patients with further emergency admission, the proportion of patients with further emergency department attendance, the mean number of further emergency department attendances per patient per day at risk, the mean duration of subsequent inpatient episodes (nights in hospital), and a composite outcome that combined the proportion of patients with further emergency service calls, emergency department attendance, emergency admission, or death.
Patients With Hypoglycemia
The non-randomized study by Sinclair et al.22 reported measures of repeat access to health care services in patients with hypoglycemia who were treated by paramedics but refused emergency medical services transportation to the hospital compared to those who were transported to the hospital. For all outcomes reported in this study (which were recorded within 72 hours of initial hypoglycemic assessment and treatment), there were no statistically significant differences between those who refused hospital transport (i.e., were treated at the scene and then released) and those who were transported to the hospital for further assessment, including the proportion of patients who had repeat access to either prehospital or emergency department care, repeat access to prehospital care alone, repeat access to prehospital care and were transported to the emergency department, repeat access to prehospital care and were transported to the emergency department and admitted, repeat access to emergency department care (not transported by paramedics), repeat access to emergency department care (not transported by paramedics) and were admitted, repeat access to prehospital care due to hypoglycemia, repeat access to emergency department care (not transported by paramedics) due to hypoglycemia.
Mortality
Patients Who Experienced a Fall
The RCT21 found that the proportion of patients who died of any cause following emergency medical services assessment did not significantly differ from patients who were in the treat and refer group and those who were in the usual care group 1 month (6.1% versus 6.0%; adjusted P = 0.960) and 6 months post-assessment (19.2% versus 18.5%; adjusted P = 0.094).
Patients With Heat Stroke
The authors of the non-randomized study by Sloan et al.,23 which compared outcomes between patients who were treated for exertional heat stroke using an onsite treatment protocol (i.e., a treat and release protocol) administered by emergency medical services and those who were immediately transported to a hospital for treatment noted that no patients in either treatment group died from exertional heat stroke during the study period.
Emergency Department Conveyance
Patients Who Experienced a Fall
As reported by the authors of the RCT,21 the proportion of patients who were conveyed to the emergency department following assessment and treatment for a fall by emergency medical services was 65.2% and 62.7% in the treat and refer and usual care groups, respectively. This between-group difference was not statistically significant (adjusted odds ratio [OR] = 1.082; 95% CI, 0.958 to 1.223; adjusted P = 0.205).
Hospital Admissions
Patients With Heat Stroke
The authors of the non-randomized study by Sloan et al.23 noted that 41% of participants who were treated onsite by emergency medical services for exertional heat stroke were admitted to the hospital compared to 60% of those who were immediately transported and treated offsite. The statistical significance of this finding was not reported.
Referral to Non-emergency Department Services
Patients Who Experienced a Fall
The authors of the RCT21 reported that 8.4% of treat and refer group participants were referred to falls services by the emergency services crew following initial assessment, compared to 1.1% of participants in the usual care group. The between-group difference was statistically significant (adjusted OR = 51.730; 95% CI, 16.46 to 162.54; adjusted P < 0.001). Additionally, a significantly smaller proportion of patients in the treat and refer group were left at the scene without referral following emergency medical services assessment compared to those treated using usual care (22.6% versus 30.3%; adjusted OR = 0.686; 95% CI, 0.600 to 0.784; adjusted P < 0.001).
Quality of Life
Patients Who Experienced a Fall
The authors of the RCT21 did not observe any statistically significant differences in mean SF-12 MCS scores or mean SF-12 PCS scores at 1-month and 6-month follow-ups.
Patient Satisfaction
Patients Who Experienced a Fall
Patient satisfaction was assessed in the RCT21 using mean QCM scores. Compared to those in the usual care group, patients who were treated with the treat and refer protocol had mean QCM Interpersonal scores that were significantly higher 1 month after the initial assessment (unadjusted scores: 68.92 [SD = 8.66] versus 68.04 [SD = 9.12]; adjusted P < 0.001). There were no significant between-group differences in mean QCM technical scores, which were 62.82 (SD = 7.98) and 63.21 (SD = 8.16) in the treat and refer and usual care groups, respectively (adjusted P = 0.506), 1 month after initial assessment.
Fear of Falling
Patients Who Experienced a Fall
There were no statistically significant differences between those who were treated using the treat and refer protocol compared to those treated with usual care for mean MFES scores at 1 or 6 months of follow-up in the RCT.21
Adverse Events
Patients Who Experienced a Fall
The authors of the RCT21 reported the proportion of study participants who experienced various adverse events. There were no significant between-group differences in the proportion of study participants who reported further fractures at 1-month follow-up; however, a statistically significantly higher proportion of those in the usual care group had further fractures at 6-month follow-up compared to those in the treat and refer group (9.8% versus 9.5%; adjusted OR = 1.449 [95% CI, 1.076 to 1.952]; adjusted P = 0.015). The proportion of patients who reported at least 1 fall following initial assessment was significantly higher in the usual care group compared to the treat and refer group at 1-month follow-up (69.4% versus 66.5%; adjusted OR = 0.723 [95% CI, 0.544 to 0.961]; adjusted P = 0.025). Statistical significance of between-group differences in additional adverse events within 2 days following initial assessment was not reported, including the proportion of patients who experienced an emergency service call, attendance at the emergency department, emergency admission, and death.
Cost-Effectiveness of Treat and Release Protocols for Patients Requiring Emergency Medical Services
Evidence regarding the cost-effectiveness of treat and release protocols for patients requiring emergency medical services was available from 1 economic evaluation.21
Measures of Cost-Effectiveness
Patients Who Experienced a Fall
The results of the economic evaluation21 suggested that the incremental cost of the intervention was £17.30 (95% CI, –£475.01 to £509.40) per patient and that it resulted in incremental QALYs of –0.0026 (95% CI, –0.0066 to 0.0014). Therefore, usual care was less costly and more effective (i.e., usual care dominated the treat and refer intervention). The authors of the economic evaluation21 stressed the uncertainty in these results, as there were no statistically significant differences in QALYs generated between the 2 groups, and suggested that further investigation was warranted.
Guidelines
No relevant evidence-based guidelines regarding the use of treat and release protocols for patients requiring emergency medical services were identified; therefore, no summary can be provided.
Limitations
Treat and release protocols are highly individualized and context-specific. The clinical or cost-effectiveness of 1 protocol may not accurately reflect the effectiveness of all treat and release protocols as an intervention class.
Outcomes experienced by participants of the Sloan et al.23 study were recorded until patients were discharged from care, which was typically several hours. Similarly, the authors of the Sinclair et al.22 study reported outcomes up to 72 hours after the initial hypoglycemia assessment. While the RCT21 had a substantially longer follow-up duration (i.e., 6 months), the long-term clinical effectiveness of treat and release protocols is uncertain.
The authors of the RCT21 did not describe minimal clinically important difference values for any outcomes measured using questionnaires, tools, or scales (e.g., patient satisfaction assessed using mean QCM scores). It was unclear if any of the reported statistically significant differences in outcomes assessed using these measures translate into clinically meaningful differences.
The studies identified for inclusion of this review investigated treat and release or treat and refer protocols for patients who had experienced a fall, were hypoglycemic, or were having exertional heat stroke. The effectiveness of treat and release protocols in other patient populations — such as those with minor trauma that does not need further assessment for internal injury, acute conditions (e.g., minor epistaxis), influenza symptoms without prostration or altered consciousness, non-threatening exacerbations of chronic conditions or pain syndromes, and palliative care patients — is unknown.
None of the included studies enrolled participants younger than 18 years of age. The clinical and cost-effectiveness of treat and release protocols for children and adolescents requiring emergency medical services is unclear.
Conclusions and Implications for Decision- or Policy-Making
One HTA21 (that included a relevant RCT and economic evaluation) and 2 non-randomized studies22,23 that addressed the clinical effectiveness and cost-effectiveness of treat and release protocols for patients requiring emergency medical services were included in this review. No evidence-based guidelines regarding the use of treat and release protocols for patients requiring emergency medical services were identified.
The evidence summarized in this report revealed mixed findings regarding the clinical effectiveness of treat and release protocols for patients accessing emergency medical services. For most outcomes, including mortality,21,23 emergency department conveyance,21 hospital admissions,23 quality of life,21 and fear of falling,21 there were no statistically significant differences between participants who were treated using various treat and release or treat and refer protocols and those who received usual care (i.e., transportation to hospital); however, participants of the treat and refer group of the RCT21 demonstrated statistically significant improvements in certain clinical outcomes compared to those in the usual care group. Specifically, treat and refer patients reported fewer further emergency service calls or emergency department attendances per patient, greater patient satisfaction, and fewer further fractures or falls. There were no instances where patients treated using usual care reported statistically significantly improved outcomes compared to those treated using treat and release protocols.
The cost-effectiveness of treat and release protocols was unclear. The authors of the economic evaluation21 included in this report concluded that, while the treat and release intervention was relatively low cost (i.e., net cost of £17.30 per patient), it did not result in improved health-related quality of life and there were no statistically significant differences in the number of QALYs generated between the 2 treatment groups.
Similar to the conclusions made in the 2014 CADTH report,17 the evidence regarding the clinical effectiveness and cost-effectiveness of treat and release protocols remains inconclusive. The limitations of the included literature,21-23 such as the variable quality of included primary studies and the risk of bias due to confounding, should be considered when interpreting the findings of this report. Future studies that assess the clinical effectiveness of treat and release protocols for other patient populations (e.g., those with minor trauma and non-threatening exacerbations of chronic conditions) would help to better define the role of “treat and release protocols as tools for emergency medical services personnel. Additionally, cost-effectiveness analyses of treat and release protocols from the perspective of Canadian health care payers are warranted.
References
1.Yarmohammadian MH, Rezaei F, Haghshenas A, Tavakoli N. Overcrowding in emergency departments: A review of strategies to decrease future challenges. J Res Med Sci. 2017;22:23. PubMed
2.Rasouli HR, Esfahani AA, Nobakht M, et al. Outcomes of Crowding in Emergency Departments; a Systematic Review. Arch Acad Emerg Med. 2019;7(1):e52. PubMed
3.Rowe BH, McRae A, Rosychuk RJ. Temporal trends in emergency department volumes and crowding metrics in a western Canadian province: a population-based, administrative data study. BMC Health Serv Res. 2020;20(1):356. PubMed
4.Health Quality Ontario. Under Pressure: Emergency department performance in Ontario. Toronto, ON: Queen's Printer for Ontario; 2016: https://www.hqontario.ca/portals/0/Documents/system-performance/under-pressure-report-en.pdf. Accessed 2021 Apr 27.
5.Morley C, Unwin M, Peterson GM, Stankovich J, Kinsman L. Emergency department crowding: A systematic review of causes, consequences and solutions. PLoS One. 2018;13(8):e0203316. PubMed
6.Bond K, Ospina MB, Blitz S, et al. Frequency, determinants and impact of overcrowding in emergency departments in Canada: a national survey. Healthc Q. 2007;10(4):32-40. PubMed
7.Nippak PM, Isaac WW, Ikeda-Douglas CJ, Marion AM, VandenBroek M. Is there a relation between emergency department and inpatient lengths of stay? Can J Rural Med. 2014;19(1):12-20. PubMed
8.Medley DB, Morris JE, Stone CK, Song J, Delmas T, Thakrar K. An association between occupancy rates in the emergency department and rates of violence toward staff. J Emerg Med. 2012;43(4):736-744. PubMed
9.Guttmann A, Schull MJ, Vermeulen MJ, Stukel TA. Association between waiting times and short term mortality and hospital admission after departure from emergency department: population based cohort study from Ontario, Canada. BMJ. 2011;342:d2983. PubMed
10.Tekwani KL, Kerem Y, Mistry CD, Sayger BM, Kulstad EB. Emergency Department Crowding is Associated with Reduced Satisfaction Scores in Patients Discharged from the Emergency Department. West J Emerg Med. 2013;14(1):11-15. PubMed
11.Kulstad EB, Sikka R, Sweis RT, Kelley KM, Rzechula KH. ED overcrowding is associated with an increased frequency of medication errors. Am J Emerg Med. 2010;28(3):304-309. PubMed
12.Jo S, Jeong T, Jin YH, Lee JB, Yoon J, Park B. ED crowding is associated with inpatient mortality among critically ill patients admitted via the ED: post hoc analysis from a retrospective study. Am J Emerg Med. 2015;33(12):1725-1731. PubMed
13.Lefevre N. Assess, treat and refer initiatives and trends in EMS: a review of literature. Renfrew, ON: Community Paramedic; 2011: https://www.communityparamedic.ca/media/download_gallery/ssess%20Treat%20and%20Refer%20Lit%20Review%20-%20June%202011.pdf. Accessed 2021 Apr 27.
14.Minhas R, Vogelaar G, Wang D, et al. A prehospital treat-and-release protocol for supraventricular tachycardia. CJEM, Can. 2015;17(4):395-402. PubMed
15.Feldman MJ, Lukins JL, Verbeek PR, Burgess RJ, Schwartz B. Use of treat-and-release medical directives for paramedics at a mass gathering. Prehosp Emerg Care. 2005;9(2):213-217. PubMed
16.Snooks H, Kearsley N, Dale J, Halter M, Redhead J, Cheung WY. Towards primary care for non-serious 999 callers: results of a controlled study of “Treat and Refer” protocols for ambulance crews. Qual Saf Healthcare 2004;13(6):435-443. PubMed
17.Emergency Medical Service “Treat and Release” Protocols: A Review of Clinical and Cost-Effectiveness, Safety, and Guidelines. Ottawa, ON: CADTH; 2014: https://www.cadth.ca/sites/default/files/pdf/htis/jun-2014/RC0557%20Treat%20and%20Release%20Final.pdf. Accessed 2021 Apr 27.
18.Shea BJ, Reeves BC, Wells G, et al. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ. 2017;358:j4008. PubMed
19.Downs SH, Black N. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J Epidemiol Community Health. 1998;52(6):377-384. PubMed
20.Higgins JPT, Green S, editors. Figure 15.5.a: Drummond checklist (Drummond 1996). Cochrane handbook for systematic reviews of interventions. London (GB): The Cochrane Collaboration; 2011: http://handbook-5-1.cochrane.org/chapter_15/figure_15_5_a_drummond_checklist_drummond_1996.htm. Accessed 2021 Apr 27.
21.Snooks HA, Anthony R, Chatters R, et al. Support and Assessment for Fall Emergency Referrals (SAFER) 2: a cluster randomised trial and systematic review of clinical effectiveness and cost-effectiveness of new protocols for emergency ambulance paramedics to assess older people following a fall with referral to community-based care when appropriate. Health Technol Assess. 2017;21(13):1-218. PubMed
22.Sinclair JE, Austin M, Froats M, et al. Characteristics, Prehospital Management, and Outcomes in Patients Assessed for Hypoglycemia: Repeat Access to Prehospital or Emergency Care. Prehosp Emerg Care. 2019;23(3):364-376. PubMed
23.Sloan BK, Kraft EM, Clark D, Schmeissing SW, Byrne BC, Rusyniak DE. On-site treatment of exertional heat stroke. Am J Sports Med. 2015;43(4):823-829. PubMed
24.Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol. 2009;62(10):e1-e34. PubMed
25.Ware J, Jr., Kosinski M, Keller SD. A 12-Item Short-Form Health Survey: construction of scales and preliminary tests of reliability and validity. Med Care. 1996;34(3):220-233. PubMed
26.Younsi M. Health-Related Quality-of-Life Measures: Evidence from Tunisian Population Using the SF-12 Health Survey. Value Health Reg Issues 2015;7:54-66. PubMed
27.CAREY RG, SEIBERT JH. A Patient Survey System to Measure Quality Improvement: Questionnaire Reliability and Validity. Med Care. 1993;31(9):834-845. PubMed
28.Okoye EC, Akosile CO, Maruf FA, Onwuakagba IU, Urama ST. Validation of Igbo version of the modified falls efficacy scale among community-dwelling older adults: a validation study. Health Qual Life Outcomes 2020;18(1):294. PubMed
29.Hill KD, Schwarz JA, Kalogeropoulos AJ, Gibson SJ. Fear of falling revisited. Arch Phys Med Rehabil. 1996;77(10):1025-1029. PubMed
Appendix 1: Selection of Included Studies
Appendix 2: Characteristics of Included Publications
Table 2: Characteristics of Included Health Technology Assessment and Systematic Review
Study citation, country, funding source | Objectives, study designs, and numbers of primary studies included | Population characteristics | Intervention and comparator(s) | Clinical outcomes, length of follow-up |
Snooks et al. (2017)21 UK Funding source: The Health Technology Assessment Programme of the National Institute for Health Research | Objective: The objective of the systematic review conducted as part of the HTA was to evaluate, within emergency medical services, the effectiveness of interventions that aim to reduce demand for emergency departments for older people who fall. Study design: A systematic review of primary studies of any design. In addition to the systematic review, the HTA included an RCT, an economic evaluation, and a qualitative study. The RCT and economic evaluation are included and described separately within this report. Number of included studies: A total of 12 studies (i.e., 2 RCTs, 9 cohort studies, and 1 qualitative study) were included. Based on the description of these studies in the systematic review, none were considered relevant the current report. Quality assessment tool: The Scottish Intercollegiate Guidelines Network checklist and the summary criteria of Walsh and Downe | Studies of older people (≥ 60 years of age) who had fallen at home or in the community and called for an emergency ambulance were eligible. Studies of people who had fallen from higher than standing, sustained a fall while participating in sports, or those whose falls were identified by home-based technology (e.g., motion sensors) which alert emergency medical services, were excluded. | Intervention: Any interventions or enhanced practices applied by emergency medical services. Only primary studies that examined treat and release protocols or policies, or other treat and refer practices that met the intervention definition applied in the current report were considered relevant. Comparators: Any comparator was eligible for the systematic review. Only primary studies that used transfer to emergency for all patients or standard of care were considered relevant to the current report. | Clinical outcomes:
Only outcomes related to clinical effectiveness were considered relevant to the current report. Length of follow-up: Varied by individual study |
HTA = health technology assessment; RCT = randomized controlled trial.
Table 3: Characteristics of Included Primary Clinical Studies
Study citation, country, funding source | Objective and study design | Population characteristics | Intervention and comparator(s) | Clinical outcomes, length of follow-up |
RCTs | ||||
Snooks et al. (2017)21 UK Funding source: The Health Technology Assessment Programme of the National Institute for Health Research Note: The citation is for an HTA, which included the relevant RCT described here. | Objective: To assess the benefits of a clinical protocol that enables paramedics to assess and refer older people to community-based falls services directly Study design: Cluster RCT. The study was conducted at 25 ambulance stations across 3 study sites. This RCT was conducted as part of an HTA. | Inclusion criteria: People (aged ≥ 65 years) who were residents in the catchment area of participating falls services and were attended by a study paramedic following an emergency call to the ambulance service, which was coded by a dispatcher as a fall without priority symptoms. Excluded: No specific exclusion criteria were listed. Number of participants: 4,704 (2,420 in the treat and refer group; 2,284 in the usual care group) Mean age: 82.54 (SD = 7.97) years in the treat and refer group; 82.14 (SD = 8.11) years in the usual care group Sex: 61.2% female in the treat and refer group; 64.7% female in the usual care group | Intervention: Paramedics used a clinical protocol for the care of older people who had fallen. The protocol enabled paramedics to assess and refer patients directly to community-based falls services, when appropriate. When patients met certain criteria, they were transferred to the emergency department. Comparators: Paramedics assigned to the control group did not receive training in the clinical protocol and were asked to continue their usual practice, which comprised assessment of injury or other conditions requiring immediate care followed by conveyance to the emergency department unless the patient refused. | Clinical outcomes:
Follow-up: 6 months |
Non-randomized studies | ||||
Sinclair et al. (2019)22 Canada Funding source: A University of Ottawa, Department of Emergency Medicine academic grant | Objective: To assess the characteristics and outcomes of patients with hypoglycemia who were treated by paramedics but refused EMS transportation to the hospital vs. those who were transported to the hospital. Study design: Retrospective cohort study. The study was conducted at a single geographic site. | Inclusion criteria: Adults (≥ 18 years of age) with or without a history of diabetes who were assessed by paramedics for hypoglycemia Excluded: Those who were younger than 18 years of age, required supraglottic airways or intubation, had absent vital signs, or those who were terminally ill or a palliative care patient Number of participants: 791 (235 in the no-transport group; 556 in the transport group) Mean age: 52.7 (SD = 20.0) years in the no-transport group; 57.7 (SD = 22.6) years in the transport group Sex: 37.0% female in the no-transport group; 52.2% female in the transport group | Intervention: Paramedic assessment and treatment of hypoglycemia (prehospital glucose reading < 4.0 mmol/L) at the scene followed by patient release due to refusal of transport to hospital Comparators: Paramedic assessment and treatment of hypoglycemia (prehospital glucose reading < 4.0 mmol/L) at the scene followed by transport to hospital | Clinical outcomes:
Follow-up: 72 hours. |
Sloan et al. (2015)23 US Funding source: The National Institute on Drug Abuse of the National Institutes of Health | Objective: To assess the outcomes of patients who developed exertional heat stroke at a half-marathon and who were either treated using an onsite protocol or were transferred to hospitals Study design: Retrospective cohort study. The study was conducted at a single geographic site. | Inclusion criteria: Patients who developed exertional heat stroke, defined as a core temperature > 102 °F and altered mental status, at the Indianapolis half-marathon between 2005 and 2012 were eligible for inclusion. Excluded: No specific exclusion criteria were listed. Number of participants: 32 (22 in the onsite treatment group; 10 in the offsite treatment group). Mean age: NR Sex: NR | Intervention: An onsite exertional heat stroke treatment protocol. Upon arrival at the cooling tent, patients had their core temperature taken, an IV line placed, and blood drawn for sodium and glucose measurements. Patients who had core temperatures ≥ 104 °F were submersed in a cooling tub and given an IV saline infusion. Patients were removed from the tub when core temperatures were approximately 102 °F or when their mental status returned to normal. Patients who reached the cooling target and still had altered mental status or abnormal vital signs were transported by EMS to the hospital. Patients who reached the cooling targets and had normal mental status and vital signs were discharged. Comparators: Immediate transport to the nearest hospital for offsite treatment of exertional heat stroke | Clinical outcomes:
Follow-up: NR |
EMS = emergency medical services; HTA = health technology assessment; MFES = Modified Falls Efficacy Scale; NR = not reported; QCM = Quality of Care Monitor; RCT = randomized controlled trial; SD = standard deviation; SF-12 = Short Form Health Survey-12 items; vs. = versus.
Table 4: Characteristics of Included Economic Evaluation
Study citation, country, funding source | Type of analysis, time horizon, perspective | Population characteristics | Intervention and comparator(s) | Approach | Source of clinical, cost, and utility data used in analysis | Main assumptions |
Snooks et al. (2017)21 UK Funding source: The Health Technology Assessment Programme of the National Institute for Health Research Note: The citation is for an HTA, which included the relevant economic evaluation described here. | Analysis: Cost-utility analysis that was conducted alongside an RCT. This economic evaluation was conducted as part of a HTA. Time horizon: Health outcomes were measured over a 6-month period. Perspective: The UK National Health Service and Personal Social Services | Data from 4,704 participants of an RCT were included in the analysis. 2,420 patients were allocated to the treat and refer group and 2,284 to the usual care group. The RCT enrolled people aged ≥ 65 years who were residents in the catchment area of participating falls services and were attended by a study paramedic following an emergency call to the ambulance service, which was coded by a dispatcher as a fall without priority symptoms. Mean age: 82.5 years in the treat and refer group; 82.1 years in the usual care group Sex: 61.2% female in the treat and refer group; 64.7% female in the usual care group | Intervention: Paramedics used a clinical protocol for the care of older people who had fallen. The protocol enabled paramedics to assess and refer patients directly to community-based falls services, when appropriate. When patients met certain criteria, they were transferred to the emergency department. Comparators: Paramedics assigned to the control group did not receive training in the clinical protocol and were asked to continue their usual practice, which comprised assessment of injury or other conditions requiring immediate care followed by conveyance to the emergency department unless the patient refused. | Trial-based approach was used. | The costs of implementing the intervention were estimated using data collected from financial reports and documents, relevant information logged as a part of routine practice, and resource utilization recording sheets, together with reference to patient records and discussions with relevant finance staff. Data on participants’ use of health services and social services were collected from paramedic records, routine hospital records, and patient-completed questionnaires. Resource uses were estimated from various health system records. Costs were calculated using various published unit costs, such as the PSSRU Unit Costs of Health and Social Care and the National Health Service Reference Costs. Utility values were derived from SF-12 scores reported by patients during the RCT. | Missing SF-6D scores (derived from SF-12 scores), which were used to calculate utility values, were imputed by regression from all available values of that score at other data points. |
HTA = health technology assessment; PSSRU = Personal Social Services Research Unit; RCT = randomized controlled trial; SF-6D = Short Form Health Survey-6 Dimensions; SF-12 = Short Form Health Survey-12 items.
Appendix 3: Critical Appraisal of Included Publications
Table 5: Strengths and Limitations of Systematic Reviews Using AMSTAR 218
Strengths | Limitations |
Snooks et al. (2017)21 | |
|
|
AMSTAR 2 = A MeaSurement Tool to Assess systematic Reviews 2; NIHR = National Institute for Health Research.
Table 6: Strengths and Limitations of Clinical Studies Using the Downs and Black Checklist19
Strengths | Limitations |
Randomized controlled trials | |
Snooks et al. (2017)21 | |
|
|
Non-randomized studies | |
Sinclair et al. (2019)22 | |
|
|
Sloan et al. (2015)23 | |
|
|
NIHR = National Institute for Health Research.
Table 7: Strengths and Limitations of Economic Evaluation Using the Drummond Checklist20
Strengths | Limitations |
Snooks et al. (2017)21 | |
Study design
Data collection
Analysis and interpretation of results
Miscellaneous
|
|
CI = confidence interval; NIHR = National Institute for Health Research; QALY = quality-adjusted life-year; RCT = randomized controlled trial.
Appendix 4: Main Study Findings and Authors’ Conclusions
Summary of Findings of Included Randomized Controlled Trials
Snooks et al. (2017)21
Main Study Findings
Cluster randomized controlled trial (RCT) that investigated the effectiveness of a clinical protocol enabling paramedics to assess and refer older people to community-based falls services directly (N = 2,420) compared to usual care, which comprised the assessment of injury or other conditions requiring immediate care followed by conveyance to the emergency department unless the patient refused (N = 2,284).
Summary of Relevant Findings
Note: Between-group comparisons for all outcomes were adjusted for various covariates, including ambulance site, patient age, distance to the emergency department from the point of assessment, recruitment point (time since the start of the study), seasonality, patient gender, and whether or not the index call was made out of general practitioner hours.
Repeat Access to Health Care Services
Proportion of patients with further emergency service call, emergency department attendance, emergency admission, or death following initial assessment (primary outcome)
Treat and refer group at 1 month: 36.4% (870 of 2,391)
Usual care group at 1 month: 37.2% (843 of 2,264)
Adjusted odds ratio (OR) = 0.956 (95% confidence interval [CI], 0.848 to 1.077)
Adjusted P = 0.461
Treat and refer group at 6 months: 71.1% (1,701 of 2,391)
Usual care group at 6 months: 70.3% (1,592 of 2,264)
Adjusted OR = 1.018 (95% CI, 0.895 to 1.157)
Adjusted P = 0.789
Proportion of patients with further emergency admission following initial assessment
Treat and refer group at 1 month: 21.6% (517 of 2,391)
Usual care group at 1 month: 21.0% (475 of 2,264)
Adjusted OR = 1.039 (95% CI, 0.903 to 1.196)
Adjusted P = 0.595
Treat and refer group at 6 months: 48.2% (1,153 of 2,391)
Usual care group at 6 months: 47.9% (1,084 of 2,264)
Adjusted OR = 1.001 (95% CI, 0.891 to 1.125)
Adjusted P = 0.984
Proportion of patients with further emergency department attendance following initial assessment
Treat and refer group at 1 month: 19.4% (463 of 2,391)
Usual care group at 1 month: 18.5% (418 of 2,264)
Adjusted OR = 1.067 (95% CI, 0.920 to 1.237)
Adjusted P = 0.392
Treat and refer group at 6 months: 45.1% (1,079 of 2,391)
Usual care group at 6 months: 45.1% (1,021 of 2,264)
Adjusted OR = 0.999 (95% CI, 0.888 to 1.123)
Adjusted P = 0.986
Mean number of further emergency department attendances per patient following initial assessment
Treat and refer group at 1 month (N = 2,197): 0.2631 (standard deviation [SD] = 0.6162)
Usual care group at 1 month (N = 2,093): 0.2609 (SD = 0.7954)
Adjusted multiplicative event ratio = 1.104 (95% CI, 0.943 to 1.293)
Adjusted P = 0.219
Treat and refer group at 6 months (N = 2,380): 0.844 (SD = 1.392)
Usual care group at 6 months (N = 2,257): 0.913 (SD = 2.738)
Adjusted multiplicative event ratio = 0.810 (95% CI, 0.722 to 0.909)
Adjusted P < 0.001
Mean number of further emergency department attendances per patient per day at risk following initial assessment
Treat and refer group at 1 month (N = 2,197): 0.0236 (SD = 0.1018)
Usual care group at 1 month (N = 2,093): 0.0223 (SD = 0.0833)
Adjusted additive group effect = 0.0011 (95% CI, – 0.0045 to 0.0066)
Adjusted P = 0.710
Adjusted additive group effect (log-transformed data) = 0.0436 (95% CI, –0.0609 to 0.1481)
Adjusted P = 0.413
Treat and refer group at 6 months (N = 2,380): 0.0169 (SD = 0.0907)
Usual care group at 6 months (N = 2,257): 0.0144 (SD = 0.0686)
Adjusted additive group effect = 0.0025 (95% CI, –0.0021 to 0.0071)
Adjusted P = 0.292
Adjusted additive group effect (log-transformed data) = –0.0163 (95% CI, –0.1024 to 0.0699)
Adjusted P = 0.711
Proportion of patients with further emergency service calls following initial assessment
Treat and refer group at 1 month: 18.5% (442 of 2,391)
Usual care group at 1 month: 21.8% (493 of 2,264)
Adjusted OR = 0.815 (95% CI, 0.705 to 0.943)
Adjusted P = 0.006
Treat and refer group at 6 months: 43.7% (1,046 of 2,391)
Usual care group at 6 months: 46.2% (1,046 of 2,264)
Adjusted OR = 0.899 (95% CI, 0.799 to 1.011)
Adjusted P = 0.076
Mean number of further emergency service calls per patient following initial assessment
Treat and refer group at 1 month (N = 2,197): 0.2981 (SD = 0.7758)
Usual care group at 1 month (N = 2,093): 0.3378 (SD = 0.7823)
Adjusted multiplicative event ratio = 0.883 (95% CI, 0.780 to 1.000)
Adjusted P = 0.049
Treat and refer group at 6 months (N = 2,380): 1.136 (SD = 2.506)
Usual care group at 6 months (N = 2,257): 1.251 (SD = 2.672)
Adjusted multiplicative event ratio = 0.931 (95% CI, 0.860 to 1.007)
Adjusted P = 0.076
Mean number of further emergency service calls per patient per day at risk following initial assessment
Treat and refer group at 1 month (N = 2,197): 0.0204 (SD = 0.0641)
Usual care group at 1 month (N = 2,093): 0.0245 (SD = 0.0814)
Adjusted additive group effect = –0.0040 (95% CI, –0.0083 to 0.0003)
Adjusted P = 0.071
Adjusted additive group effect (log-transformed data) = –0.1354 (95% CI, –0.2418 to –0.0290)
Adjusted P = 0.013
Treat and refer group at 6 months (N = 2,380): 0.0125 (SD = 0.0363)
Usual care group at 6 months (N = 2,257): 0.0172 (SD = 0.0599)
Adjusted additive group effect = –0.0045 (95% CI, –0.0073 to –0.0017)
Adjusted P = 0.002
Adjusted additive group effect (log-transformed data) = –0.1183 (95% CI, –0.2079 to –0.0286)
Adjusted P = 0.010
Mean duration of subsequent inpatient episodes (nights in hospital) following initial assessment
Treat and refer group at 1 month (N = 2,391): 2.25 (SD = 6.14)
Usual care group at 1 month (N = 2,264): 2.10 (SD = 6.05)
Adjusted additive group effect = 0.141 (95% CI, –0.207 to 0.490)
Adjusted P = 0.426
Treat and refer group at 6 months (N = 2,391): 11.18 (SD = 22.80)
Usual care group at 6 months (N = 2,264): 11.62 (SD = 23.52)
Adjusted additive group effect = –0.563 (95% CI, –1.884 to 0.757)
Adjusted P = 0.403
Mortality
Proportion of patients who died (of any cause) following initial assessment
Treat and refer group at 1 month: 6.1% (147 of 2,391)
Usual care group at 1 month: 6.0% (136 of 2,264)
Adjusted OR = 0.994 (95% CI, 0.780 to 1.266)
Adjusted P = 0.960
Treat and refer group at 6 months: 19.2% (458 of 2,391)
Usual care group at 6 months: 18.5% (419 of 2,264)
Adjusted OR = 1.187 (95% CI, 0.971 to 1.451)
Adjusted P = 0.094
Emergency Department Conveyance
Proportion of patients who were conveyed to the emergency department following initial assessment
Treat and refer group: 65.2% (1,579 of 2,420)
Usual care group: 62.7% (1,431 of 2,284)
Adjusted OR = 1.082 (95% CI, 0.958 to 1.223)
Adjusted P = 0.205
Referral to Non-Emergency Department Services
Proportion of patients who were referred to falls service by emergency service crew following initial assessment
Treat and refer group: 8.4% (204 of 2,420)
Usual care group: 1.1% (26 of 2,284)
Adjusted OR = 51.730 (95% CI, 16.46 to 162.54)
Adjusted P < 0.001
Proportion of patients who were left at the scene without referral following initial assessment
Treat and refer group: 22.6% (547 of 2,420)
Usual care group: 30.3% (692 of 2,284)
Adjusted OR = 0.686 (95% CI, 0.600 to 0.784)
Adjusted P < 0.001
Adverse Events
Proportion of patients who experienced further reported fractures following initial assessment
Treat and refer group at 1 month: 4.1% (98 of 2,391)
Usual care group at 1 month: 4.0% (91 of 2,264)
Adjusted OR = 1.002 (95% CI, 0.744 to 1.351)
Adjusted P = 0.987
Treat and refer group at 6 months: 9.5% (228 of 2,391)
Usual care group at 6 months: 9.8% (222 of 2,264)
Adjusted OR = 1.449 (95% CI, 1.076 to 1.952)
Adjusted P = 0.015
Proportion of patients who reported ≥ 1 further falls following initial assessment
Treat and refer group at 1 month: 66.5% (413 of 621)
Usual care group at 1 month: 69.4% (409 of 589)
Adjusted OR = 0.723 (95% CI, 0.544 to 0.961)
Adjusted P = 0.025
Proportion of patients who had an emergency service call within 2 days following initial assessment
Treat and refer group: 4.2% (101 of 2,420)
Usual care group: 5.1% (117 of 2,284)
Adjusted P = not reported (NR)
Proportion of patients who attended the emergency department within 2 days following initial assessment
Treat and refer group: 3.2% (78 of 2,420)
Usual care group: 4.0% (92 of 2,284)
Adjusted P = NR
Proportion of patients who had an emergency admission within 2 days following initial assessment
Treat and refer group: 5.5% (133 of 2,420)
Usual care group: 4.8% (109 of 2,284)
Adjusted P = NR
Proportion of patients who died within 2 days following initial assessment
Treat and refer group: 0.8% (19 of 2,420)
Usual care group: 0.7% (16 of 2,284)
Adjusted P = NR
Quality of Life
Mean Short Form Health Survey-12 items mental component summary (MCS) scores following initial assessment
Treat and refer group at 1 month (N = 447): 39.80 (SD = 12.47)
Usual care group at 1 month (N = 410): 38.89 (SD = 12.16)
Adjusted additive group effect = 0.902 (95% CI, –0.744 to 2.547)
Adjusted P = 0.282
Treat and refer group at 6 months (N = 258): 43.21 (SD = 12.57)
Usual care group at 6 months (N = 241): 42.82 (SD = 12.28)
Adjusted additive group effect = 0.463 (95% CI, –1.717 to 2.643)
Adjusted P = 0.677
Mean SF-12 physical component summary (PCS) scores following initial assessment
Treat and refer group at 1 month (N = 447): 29.07 (SD = 9.97)
Usual care group at 1 month (N = 410): 29.40 (SD = 10.28)
Adjusted additive group effect = –0.495 (95% CI, –1.847 to 0.856)
Adjusted P = 0.472
Treat and refer group at 1 month (N = 258): 30.44 (SD = 11.33)
Usual care group at 1 month (N = 241): 31.88 (SD = 11.67)
Adjusted additive group effect = –1.300 (95% CI, –3.282 to 0.682)
Adjusted P = 0.198
Patient Satisfaction
Mean Quality of Care Monitor (QCM) technical scores following initial assessment
Treat and refer group at 1 month (N = 563): 62.82 (SD = 7.98)
Usual care group at 1 month (N = 551): 63.21 (SD = 8.16)
Adjusted additive group effect = –0.320 (95% CI, –1.265 to 0.625)
Adjusted P = 0.506
Mean QCM interpersonal scores following initial assessment
QCM interpersonal scores group at 1 month (N = 563): 68.92 (SD = 8.66)
Usual care group at 1 month (N = 551): 68.04 (SD = 9.12)
Adjusted additive group effect = 3.132 (95% CI, 1.587 to 4.678)
Adjusted P < 0.001
Fear of Falling
Mean fall-specific MFES scores following initial assessment
Treat and refer group at 1 month (N = 634): 3.714 (SD = 3.040)
Usual care group at 1 month (N = 600): 3.815 (SD = 3.117)
Adjusted additive group effect = –0.055 (95% CI, –0.385 to 0.275)
Adjusted P = 0.743
Treat and refer group at 6 months (N = 341): 4.547 (SD = 3.328)
Usual care group at 6 months (N = 310): 4.792 (SD = 3.393)
Adjusted additive group effect = –0.230 (95% CI, –0.729 to 0.270)
Adjusted P = 0.368
Authors’ Conclusion
“The complex SAFER 2 trial intervention, with a protocol for paramedics to assess older people who had fallen and refer those without need for immediate clinical care to community-based falls services, was inexpensive and safe. We did not find any effect on our primary outcome, although, when broken down into its components, there was a small reduction in the occurrence and rate of further emergency service calls. We did not find any evidence of improved quality of life, although some aspects of satisfaction were higher in the intervention group. Referral to falls services was lower than expected and variable between paramedics, although fairly consistent between sites. Fewer patients were left at scene in the intervention group by their attending ambulance crews without ongoing care than in the control group; however, other processes of care were unaltered (p. 100).”21
Summary of Findings of Included Non-Randomized Studies
Sinclair et al. (2019)22
Main Study Findings
Retrospective cohort study that assessed the characteristics and outcomes of patients with hypoglycemia who were treated by paramedics but refused emergency medical services transportation to the hospital (N = 235) versus those who were transported to the hospital (N = 556).
Summary of Relevant Findings
Repeat access to Health Care Services
Proportion of patients who had repeat access to prehospital or emergency department care within 72 hours of initial hypoglycemic assessment and treatment
No-transport group: 3.8% (9 of 235)
Transport group: 6.1% (34 of 556)
P = 0.232
Proportion of patients who had repeat access to prehospital care within 72 hours of initial hypoglycemic assessment and treatment
No-transport group: 2.1% (5 of 235)
Transport group: 4.1% (23 of 556)
P = 0.208
Proportion of patients who had repeat access to prehospital care and were transported to the emergency department within 72 hours of initial hypoglycemic assessment and treatment
No-transport group: 2.1% (5 of 235)
Transport group: 3.2% (18 of 556)
P = 0.492
Proportion of patients who had repeat access to prehospital care and were transported to the emergency department and admitted within 72 hours of initial hypoglycemic assessment and treatment
No-transport group: 0.4% (1 of 235)
Transport group: 0.4% (2 of 556)
P = 1.000
Proportion of patients who had repeat access to emergency department care (not transported by paramedics) within 72 hours of initial hypoglycemic assessment and treatment
No-transport group: 1.7% (4 of 235)
Transport group: 2.3% (13 of 556)
P = 0.789
Proportion of patients who had repeat access to emergency department care (not transported by paramedics) and were admitted within 72 hours of initial hypoglycemic assessment and treatment
No-transport group: 0.4% (1 of 235)
Transport group: 0.2% (1 of 556)
P = 0.506
Proportion of patients who had repeat access to prehospital care because of hypoglycemia within 72 hours of initial hypoglycemic assessment and treatment
No-transport group: 0.8% (2 of 235)
Transport group: 0.7% (4 of 556)
P = 1.000
Proportion of patients who had repeat access to emergency department care (not transported by paramedics) because of hypoglycemia within 72 hours of initial hypoglycemic assessment and treatment
No-transport group: 0.4% (1 of 235)
Transport group: 0.2% (1 of 556)
P = 0.506
Authors’ Conclusions
“Our study revealed that most patients are currently transported to the emergency department following a prehospital hypoglycemic event, and that patients on insulin were less likely to experience any repeat access to paramedics and/or the [emergency department] after their initial treatment of hypoglycemia by paramedics, regardless of transport. These findings suggest there may be a place for a treat-and-release strategy that could greatly influence paramedic practice and permit the safe out-of-hospital management of hypoglycemia for many patients. Future research should explore the high-risk factors associated with other potential adverse events in this cohort of patients (e.g., admission to hospital) (p.375).”22
Sloan et al. (2015)23
Main Study Findings
Retrospective cohort study that assessed the outcomes of patients who developed exertional heat stroke at a half-marathon and who were either treated using an onsite protocol (N = 22) or were immediately transferred to local hospitals for treatment (N = 10).
Summary of Relevant Findings
Hospital Admissions
Proportion of patients who were admitted to the hospital
Onsite treatment group: 41% (9 of 22)
Offsite treatment group: 60% (6 of 10)
P = NR
Mortality
Proportion of patients who died from exertional heat stroke
Onsite treatment group: 0% (0 of 22)
Offsite treatment group: 0% (0 of 10)
P = NR
Authors’ Conclusions
“Onsite treatment of exertional heat stroke is safe and effective and may decrease the need to transport patients to the hospital. A similar approach could be adapted for football practice, large mass gathering events in the heat, military training, and for firefighters. Future studies are needed to help predict what race conditions and medical factors further increase the risk for athletes developing heatstroke (p. 829).”23
Summary of Findings of Included Economic Evaluation
Snooks et al. (2017)21
Main Study Findings
Cost-utility analysis that evaluated the cost-effectiveness of a clinical protocol that enabled paramedics to assess and refer older people to community-based falls services directly (N = 2,420) versus usual care, which comprised assessment of injury or other conditions requiring immediate care followed by conveyance to the emergency department unless the patient refused (N = 2,284).
Summary of Relevant Findings
Health Care Resource Use
Mean cost of index call
Treat and refer group at 1 month (N = 2,391): £2,242.19
Usual care group 1 month (N = 2,264): £2,069.15
Treat and refer group at 6 months (N = 2,391): £2,829.55
Usual care group at 6 months (N = 2,264): £2,606.97
Mean routine costs
Treat and refer group at 1 month (N = 2,391): £1,041.17
Usual care group at 1 month (N = 2,264): £993.55
Treat and refer group at 6 months (N = 2,391): £4,308.19
Usual care group at 6 months (N = 2,264): £4,467.95
Mean self-reported costs
Treat and refer group at 1 month (N = 2,391): £456.66
Usual care group at 1 month (N = 2,264): £451.67
Treat and refer group at 6 months (N = 2,391): £1,678.66
Usual care group at 6 months (N = 2,264): £1,586.85
Mean total costs
Treat and refer group at 1 month (N = 2,391): £3,740.02
Usual care group at 1 month (N = 2,264): £3,514.37
Treat and refer group at 6 months (N = 2,391): £8,816.41
Usual care group at 6 months (N = 2,264): £8,661.77
Quality-Adjusted Life-Year
Mean quality-adjusted life-years (QALYs) (derived from Short Form Health Survey- 6 Dimensions [SF-6D] utilities)
Treat and refer group at 6 months (N = 2,375): 0.2093 (standard deviation [SD] = 0.0709)
Usual care group at 6 months (N = 2,264): 0.2133 (SD = 0.0713)
Additive group effect = –0.0026 (95% CI, –0.0066 to 0.0014)
P = 0.202
Cost-Effectiveness
Incremental cost of the intervention = £17.30 (95% CI, –£475.01 to £509.40)
Incremental QALY = –0.0026 (95% CI, –0.0066 to 0.0014)
Cost per QALY gained: Usual care is dominant
“In summary, as there is no difference between groups in relation to resource use, the net cost of the intervention is £17.30. There is also no statistically significant difference in the number of QALYs generated between the two groups (p. 72).”21
Authors’ Conclusion
“The relative cost-effectiveness of the SAFER 2 trial intervention is inconclusive, and further investigation is warranted to establish whether or not it represents value for money. It has resulted in important difference in the number of subsequent emergency service calls at 1 month and [emergency department] attendances at 6 months, but no difference between intervention and usual care in relation to QALYs gained (p. 72).”21
Appendix 5: References of Potential Interest
Health Technology Assessments
Alternative Interventions
1.Guo B, Corabian P, Yan C, et al. Community paramedicine: Program characteristics and evaluation. Edmonton (AB): Institute of Health Economics; 2017: https://www.ihe.ca/advanced-search/community-paramedicine-program-characteristics-and-evaluation Accessed 2021 Apr 27.
Systematic Reviews
Alternative Interventions
2.van Vuuren J, Thomas B, Agarwal G, et al. Reshaping healthcare delivery for elderly patients: the role of community paramedicine; a systematic review. BMC Health Serv Res. 2021 Jan 06;21(1):29. PubMed
No Comparator
3.Greene JA, Deveau BJ, Dol JS, Butler MB. Incidence of mortality due to rebound toxicity after 'treat and release' practices in prehospital opioid overdose care: a systematic review. Emerg Med J. 2019 Apr;36(4):219-224. PubMed
Non-Randomized Studies
Alternative Comparators
4.Krumperman K, Weiss S, Fullerton L. Two Types of Prehospital Systems Interventions that Triage Low-Acuity Patients to Alternative Sites of Care. South Med J. 2015 Jun;108(7):381-386. PubMed
No Comparator
5.Minhas R, Vogelaar G, Wang D, et al. A prehospital treat-and-release protocol for supraventricular tachycardia. CJEM, Can. 2015 Jul;17(4):395-402. PubMed
Guidelines and Recommendations – Unclear Methodology
6.National Model EMS Clinical Guidelines. Falls Church (VA): National Association of State EMS Officials; 2019: https://nasemso.org/wp-content/uploads/National-Model-EMS-Clinical-Guidelines-2017-PDF-Version-2.2.pdf Accessed 2021 Apr 27.
Qualitative Studies
7.Power B, Bury G. A survey of general practitioner's opinion on the proposal to introduce 'treat and referral' into the Irish emergency medical service. Ir J Med Sci. 2020 Nov;189(4):1457-1463. PubMed
8.Power B, Bury G, Ryan J. Stakeholder opinion on the proposal to introduce 'treat and referral' into the Irish emergency medical service. BMC Emerg Med. 2019 Dec 21;19(1):81. PubMed
Review Articles
9.Kolinsky D, Keim SM, Cohn BG, et al. Is a Prehospital Treat and Release Protocol for Opioid Overdose Safe? J Emerg Med. 2017 Jan;52(1):52-58. PubMed
Additional References
10.Long D, Lord B. Widening the conversation: Paramedic involvement in interprofessional care. Prog Palliat Care. 2021 Jan; 29(2):57-58. https://www.tandfonline.com/doi/abs/10.1080/09699260.2020.1852656 Accessed 2021 Apr 27.
11.Rostykus P, Kennel J, Adair K, et al. Variability in the Treatment of Prehospital Hypoglycemia: A Structured Review of EMS Protocols in the United States. Prehosp Emerg Care. 2016 Mar; 20(4):524-530. https://www.tandfonline.com/doi/full/10.3109/10903127.2015.1128031 Accessed 2021 Apr 27. PubMed
12.Washington University School of Medicine in St. Louis Department of Emergency Medicine. Treat and Release After Naloxone Following Opiate Overdose. 2014. https://emergencymedicine.wustl.edu/items/treat-and-release-after-naloxone-following-opiate-overdose/ Accessed 2021 Apr 27.
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up to date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third-party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.
Questions or requests for information about this report can be directed to Requests@CADTH.ca