CADTH Health Technology Review
Urethral Inserts for the Management of Adult Male Urinary Incontinence
Rapid Review
Authors: Srabani Banerjee, Nina Frey
Key Messages
No evidence was identified regarding the clinical effectiveness of male urethral inserts for the management of male urinary incontinence.
No evidence was identified regarding the cost-effectiveness of male urethral inserts for the management of male urinary incontinence.
Context and Policy Issues
Urinary incontinence refers to the involuntary leakage of urine.1 With increasing age, urinary incontinence can occur in both men and women, and affects the quality of life. This condition is associated with social and economic impacts.2 For men aged more than 65 years, the prevalence of urinary incontinence has been reported to range between 11% and 34%.1 The prevalence of incontinence increases with age. In Canada, according to a 2014 report, the estimates for the prevalence of incontinence in men were 6.4%, 11.6, and 18.7% in the age ranges 65 to 74 years, 75 to 84 years, and greater than 85 years, respectively.3 There are various types of urinary incontinence; these include urge urinary incontinence, stress urinary incontinence, mixed incontinence (combination of urge urinary incontinence and stress urinary incontinence), overflow incontinence, post-void dribbling, functional incontinence, and incontinence after prostrate treatment.1 Treatment and management options for urinary incontinence include pelvic floor muscle exercise, pharmacotherapy, urethral inserts, catheters, and surgical interventions.1-3 Generally, if conservative treatments (such as exercises) are not effective, other less conservative treatment modalities may be considered.3 Pharmacotherapy may be effective but may be associated with side effects.1,3 Surgical interventions are invasive and may be associated with complications.2,3 As urethral inserts may be less invasive, there is growing interest in this management option for controlling urinary incontinence. The Contino is an example of a self-administered urethral insert to control urinary incontinence in male adults. It is inserted into the distal portion of the male urethra, inhibiting the flow of urine, and can be removed before urination and reinserted after urination.4 The Contino is made from medical-grade polymer and has been tested for biocompatibility.4
The purpose of this report is to summarize the evidence regarding the clinical effectiveness and cost-effectiveness of male urethral inserts for the management of male urinary incontinence.
Research Questions
What is the clinical effectiveness of male urethral inserts for the management of male urinary incontinence?
What is the cost-effectiveness of male urethral inserts for the management of male urinary incontinence?
Methods
Literature Search Methods
A limited literature search was conducted by an information specialist on key resources including MEDLINE, the Cochrane Database of Systematic Reviews, the international HTA database, the websites of Canadian and major international health technology agencies, as well as a focused internet search. The search strategy comprised both controlled vocabulary, such as the National Library of Medicine’s MeSH (Medical Subject Headings), and keywords. The main search concept was urethral insert. No search filters were applied to limit retrieval. No language or date limits were applied.
Selection Criteria and Methods
One reviewer screened citations and selected studies. In the first level of screening, titles and abstracts were reviewed and potentially relevant articles were retrieved and assessed for inclusion. The final selection of full-text articles was based on the inclusion criteria presented in Table 1.
Criteria | Description |
Population | Adult males aged ≥ 18 years with urinary incontinence for any reason (e.g., post-surgery [radical prostatectomy], sphincteric incompetence, hyperactive bladder) |
Intervention | Urethral insert or blocking device (e.g., brand name Contino) for male urinary incontinence |
Comparator | Penile clamp; external or intermittent catheter or catheter in general No comparator (safety studies only) |
Outcomes | Q1: Clinical effectiveness (e.g., quality of life, bladder leakage); safety Q2: Cost-effectiveness |
Study designs | HTAs, SRs, RCTs, non-randomized studies, and economic evaluation |
HTAs = health technology reviews; Q = question; RCTs = randomized controlled trials; SRs = systematic reviews.
Exclusion Criteria
Articles were excluded if they did not meet the selection criteria outlined in Table 1.
Critical Appraisal of Individual Studies
Not applicable, as no relevant studies were identified.
Summary of Evidence
Quantity of Research Available
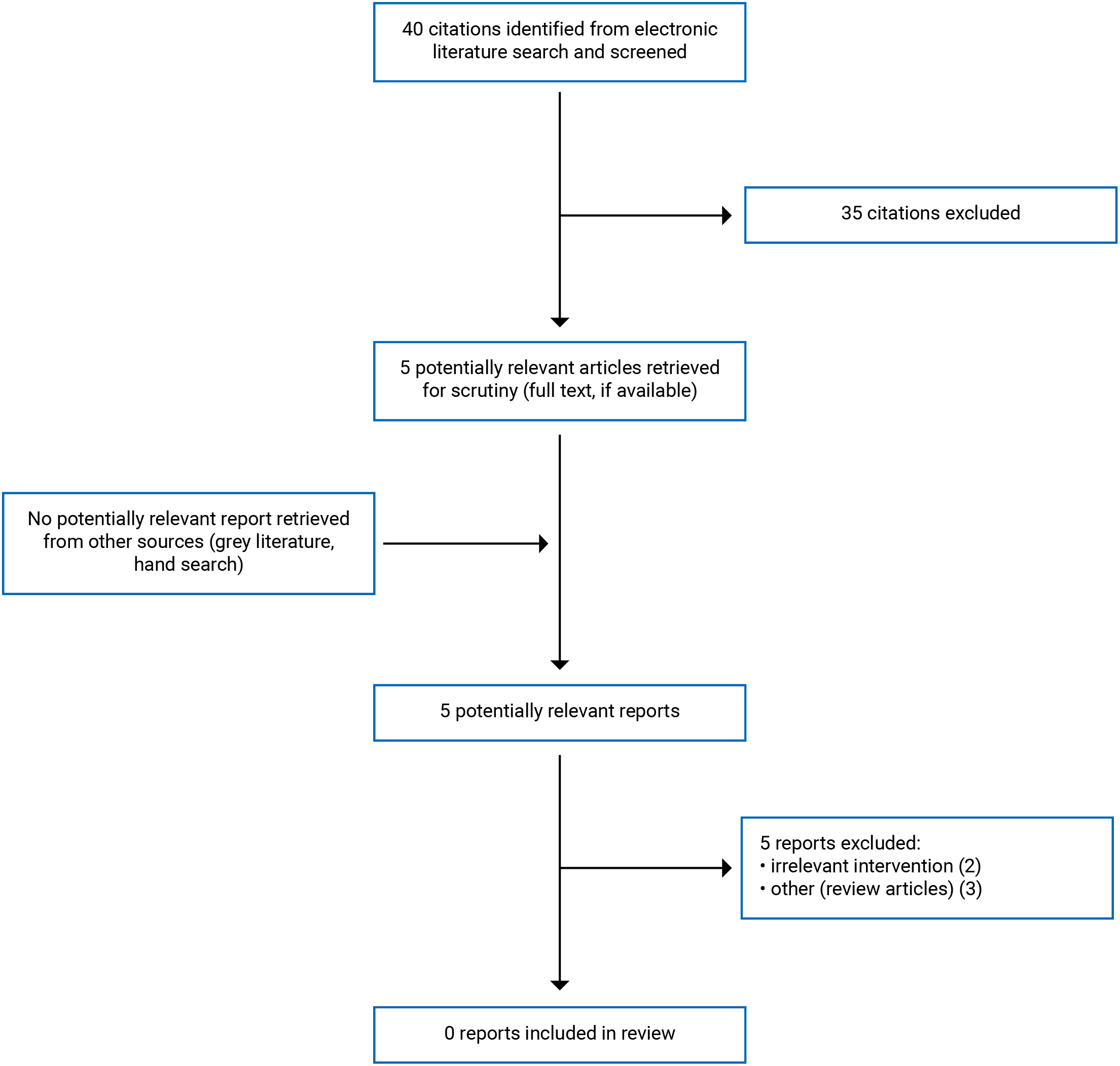
A total of 40 citations were identified in the literature search. Following the screening of titles and abstracts, 35 citations were excluded and 5 potentially relevant reports from the electronic search were retrieved for full-text review. No potentially relevant publications were retrieved from the grey literature search for full-text review. Of these potentially relevant articles, 5 publications were excluded for various reasons, and no publication met the inclusion criteria and was not included in this report. Appendix 1 presents the PRISMA5 flow chart of the study selection.
Summary of Findings
As no relevant evidence on the clinical effectiveness and cost-effectiveness of urethral inserts for the management of adult male urinary incontinence was identified, a summary of evidence cannot be provided.
Limitations
To our knowledge, there appears to be lack of relevant published evidence regarding the clinical effectiveness and cost-effectiveness of urethral inserts for the management of adult male urinary incontinence.
Conclusions and Implications for Decision- or Policy-Making
No evidence was identified regarding the clinical effectiveness or the cost-effectiveness of male urethral inserts for the management of male urinary incontinence.
One unpublished, prospective, open-label, single arm study (NCT04165408)4 was identified; this study investigated the safety and clinical efficacy of Contino for the control of urinary incontinence. Of note, as no published report of this study was available, this study was not included in the main text and a critical appraisal of the study was not conducted. The estimated study completion date was December 30, 2020.4 This study included 25 male adults, with evidence of sphincteric incompetence, as assessed by the investigator.4 Some results of this study were available from the 2020 Canadian Urological Association (CUA) meeting abstracts collection.6 The study duration was 30 days and 15 patients completed the study. With the use of the Contino device, there was a reduction in the International Consultation on Incontinence Questionnaire – Short Form (ICIQ-SF) score (mean [standard deviation]) from 16.5 (3.7) at baseline to 12.2 (5.5) at the end of the study;6 the higher the ICIQ-SF score, the greater the severity of urinary incontinence.7 Adverse events with use of Contino were generally mild and not serious. Another prospective, open-label, single arm study (the COMFORT STUDY: NCT03605459) investigated the safety and clinical efficacy of the Comfort Plug for the control of urinary incontinence.8 This study included 30 male adults with evidence of moderate to severe urinary incontinence, as assessed by the investigator; study duration was 30 days. The study was completed on April 3, 2018, but no results have been published. Considering the lack of relevant published evidence regarding the use of urethral inserts for the management of urinary incontinence in male adults, definitive conclusions are not possible.
Comparative studies involving larger patient numbers and investigating long-term effects and economic evaluations are needed to understand the clinical effectiveness and cost-effectiveness of urethral inserts, relative to alternatives, for the control of adult male urinary incontinence.
References
1.Clemens JQ. Urinary incontinence in men. In: Post TW, ed. UpToDate. Waltham (MA): UpToDate; 2019. Accessed 2021 Apr 14.
2.Natali AN, Fontanella CG, Carniel EL. Biomechanical analysis of the interaction phenomena between artificial urinary sphincter and urethral duct. Int J Numer Method Biomed Eng. 2020;36(3):e3308. PubMed
3.Cameron Institute. Incontinence: The Canadian perspective. Peterborough (ON): The Canadian Continence Foundation; 2014 Dec: https://www.canadiancontinence.ca/pdfs/en-incontinence-a-canadian-perspective-2014.pdf. Accessed 2021 Apr 6.
4.CMX Research. NCT04165408: Trial to assess the safety and clinical performance of Contino® in preventing urinary incontinence. ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2019 Nov: https://www.clinicaltrials.gov/ct2/show/NCT04165408 Accessed 2021 Apr 5.
5.Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol. 2009;62(10):e1-e34. PubMed
6.Elterman DS, Casey R, De los Reyes TJ, et al. The Contino® urethral insert in the treatment of male stress urinary incontinence. Canadian Urological Association Journal 2020 CUA Abstracts.Vol 14(Issue 6):S59-S60. https://cuaj.ca/index.php/journal/article/view/6732/4463. Accessed 2021 Mar 31.
7.Karmakar D, Mostafa A, Abdel-Fattah M. A new validated score for detecting patient-reported success on postoperative ICIQ-SF: a novel two-stage analysis from two large RCT cohorts. Int Urogynecol J. 2017;28(1):95-100. PubMed
8.CMX Research. NCT03605459: Trial to assess the efficacy of the Comfort Plug™in preventing urinary incontinence (COMFORT STUDY). ClinicalTrials.gov. Bethesda (MD) U. S. National Library of Medicine; 2018 Jul: https://clinicaltrials.gov/ct2/show/study/NCT03605459. Accessed 2021 Apr 5.
Appendix 1: Selection of Included Studies
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up to date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third-party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.
Questions or requests for information about this report can be directed to Requests@CADTH.ca