CADTH Reimbursement Review
Daratumumab (Darzalex SC)
Sponsor: Janssen Inc.
Therapeutic area: Light chain amyloidosis
This multi-part report includes:
Clinical Review
Pharmacoeconomic Review
Clinical Review
Abbreviations
AE
adverse event
AL
light chain
ASCT
autologous stem cell transplant
CI
confidence interval
CMRG
Canadian Myeloma Research Group
CR
complete response
CyBorD
cyclophosphamide, bortezomib, and dexamethasone
DCyBorD
daratumumab in combination with cyclophosphamide, bortezomib, and dexamethasone
ECOG PS
Eastern Cooperative Oncology Group Performance Score
eCRF
electronic case report form
EORTC QLQ-C30
European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire
FLC
free light chain
HR
hazard ratio
HRQoL
health-related quality of life
IPCW
inverse probability of censoring weight
IRC
independent review committee
ITT
intention-to-treat
LS
least squares
MCS
mental component summary
MID
minimal important difference
MOD-PFS
major organ deterioration progression-free survival
MRD
minimal residual disease
NT-proBNP
N-terminal pro–B-type natriuretic peptide
OS
overall survival
PFS
progression-free survival
PR
partial response
SAE
serious adverse event
SC
subcutaneous
SF-36v2
Short Form (36) Health Survey version 2
SOC
system organ class
TEAE
treatment-emergent adverse event
ULN
upper limit of normal
VAS
visual analogue scale
VGPR
very good partial response
Executive Summary
An overview of the submission details for the drug under review is provided in Table 1.
Item | Description |
|---|---|
Drug product | Daratumumab (Darzalex), 1,800 mg/15 mL (120 mg/mL) for subcutaneous injection |
Indication | In combination with bortezomib, cyclophosphamide, and dexamethasone, for the treatment of adult patients with newly diagnosed light chain amyloidosis |
Reimbursement request | As per indication |
Health Canada approval status | NOC |
Health Canada review pathway | Other expedited pathway (Project ORBIS) |
NOC date | April 12, 2021 |
Sponsor | Janssen Inc. |
NOC = Notice of Compliance.
Introduction
Systemic immunoglobulin light chain (AL) amyloidosis is a rare disease characterized by the deposition of light chain amyloid fibrils produced by clonal CD38+ plasma cells. AL amyloidosis can affect multiple organs simultaneously and manifest with a range of non-specific presenting symptoms, such as unexplained heart failure, neuropathy, and hepatomegaly, making it difficult to diagnose at early stages. Patients diagnosed at advanced stages, particularly when heart involvement is present, are at high risk of death within a few months.1 Light chain amyloidosis is an incurable disease and survival is generally poor, particularly among patients with delayed diagnosis, later-stage disease, cardiac involvement, or multiple affected organs. Cardiac damage is a major determinant of survival. The median overall survival (OS) of AL amyloidosis has been estimated to range from 7.8 years for cardiac stage I to 5.8 years for cardiac stage IV disease.2 In Canada, based on real-world data from Alberta, the median OS from initiation of first-line treatment for AL amyloidosis was 5.2 years among patients diagnosed in 2012 onward.3
Although most patients are diagnosed at later stages of the disease after overt symptoms appear, biomarkers of organ involvement can be used to detect cardiac and renal amyloidosis. In these patients, immunofixation electrophoresis of the serum and urine and an immunoglobulin-free light chain assay (which assesses the concentration of kappa and lambda free light chains [FLCs] and their ratio in the serum) are carried out to detect precursor light chain protein. Where available, imaging with radio-iodinated serum amyloid P component can identify amyloid deposits in individuals with these syndromes, but this test is not widely available. Because other types of systemic amyloidosis can have clinical presentations that overlap those of AL amyloidosis, a tissue biopsy and histopathological analysis may be necessary to confirm a diagnosis of AL amyloidosis. The characteristics of the underlying clone and the extent and severity of organ involvement are then evaluated to guide therapeutic strategy.4,5
There is currently no formal Health Canada–approved therapy for AL amyloidosis in Canada. However, many of the approved therapies for multiple myeloma are used in AL amyloidosis. Historically, melphalan in the context of an autologous stem cell transplant (ASCT) has been the standard of care, but few patients are suitable for this treatment. Bortezomib-based triplet therapy (cyclophosphamide, bortezomib, and dexamethasone [CyBorD] and bortezomib, melphalan, and dexamethasone [BMDex]) are the established standard of care in AL amyloidosis.
Daratumumab, a human CD38-targeting antibody, in combination with CyBorD (DCyBorD), was recently approved in Canada (Health Canada Notice of Compliance issued April 12, 2021) for the treatment of adult patients with newly diagnosed AL amyloidosis through Project Orbis.6
The objective of this review was to evaluate the efficacy and safety of daratumumab subcutaneous (SC) injection in combination with CyBorD for the treatment of adult patients with newly diagnosed AL amyloidosis.
Stakeholder Perspectives
The information in this section is a summary of the input provided by the patient and clinician groups who responded to CADTH’s call for input and from a clinical expert consulted by CADTH for the purpose of this review.
Patient Input
Myeloma Canada provided input for the review of DCyBorD for the treatment of AL amyloidosis. Myeloma Canada conducted a survey of its patient and caregiver community via email and social media. The survey was available from July 12 to July 25, 2021. The survey received 40 responses, 12 of which were deemed eligible (from 7 patients currently receiving CyBorD, 3 patients currently receiving DCyBorD, and 2 patients waiting to receive treatment). All the patients surveyed rated access to effective treatments for AL amyloidosis as extremely important. The respondents who had treatment experience with CyBorD (n = 7) indicated that, of their expectations of a new treatment before taking CyBorD, “minimal side effects” was mentioned by most patients (n = 4), followed by “disease control” (3) and “improved quality of life” (1). All patients treated with CyBorD rated their experience with this treatment regimen as “somewhat tolerable,” “tolerable,” or “very tolerable.” Fatigue and neuropathy were cited as the least tolerable side effects of CyBorD. Patients who had been treated with DCyBorD rated their overall side effects as “somewhat tolerable,” “tolerable,” or “very tolerable.” Some patients found the side effects of bortezomib and cyclophosphamide intolerable, and 1 patient was treated with only daratumumab and dexamethasone. In terms of what is important to patients when it comes to treating their AL amyloidosis, the majority of responses described a strong desire for a good or better quality of life and being able to continue daily activities without debilitating side effects of treatment.
Clinician Input
Input From Clinical Experts Consulted by CADTH
The clinical experts consulted by CADTH noted that no treatments other than DCyBorD are currently approved or generally funded for the treatment of AL amyloidosis in Canada. However, CyBorD is used off-label and may be accessed through the manufacturer’s special access program for a limited treatment duration. In some provinces (e.g., Alberta) it is funded through a special agreement with provincial groups. DCyBorD is amenable to use for most patients with newly diagnosed AL amyloidosis. In terms of response assessment, the clinical experts noted that improved hematologic response, progression-free survival (PFS), and OS, as well as organ response and health-related quality of life (HRQoL), are considered clinically meaningful responses to treatment. With respect to frequency of assessment of treatment response, the clinical experts indicated that monthly assessments are common in Canada but decisions on adequate hematologic and organ responses are made 3 months and 6 months after initiation of treatment. The clinical experts indicated that daratumumab is a practice-changing regimen in a disease area where no approved or funded treatments exist.
Clinician Group Input
Clinician input was received from the Canadian Myeloma Research Group (CMRG) and the Ontario Health (Cancer Care Ontario) Hematology Cancer Drug Advisory Committee. The clinician group noted that the most important goals of any plasma cell– directed therapy is to achieve deep and rapid elimination of clonal plasma cells and the monoclonal protein product they secrete. Based on the data from the ANDROMEDA trial’s control arm, the deep responses necessary for the organ improvement and prolonged disease control are not optimal with CyBorD alone. Given the toxic effect of the amyloid light chain, it is vital to achieve rapid and deep responses and to have access to first-line therapies that produce deep and rapid responses. The clinician group noted that DCyBorD is a major breakthrough in AL amyloidosis, which, if not treated quickly and with deep responses, can lead to irreversible organ damage. The encouraging results of the ANDROMEDA trial suggest that, if DCyBorD is approved for this indication, it is likely fewer AL patients will require ASCT, with its attendant risk of morbidity and increased mortality. The clinician group also noted that, because DCyBorD is well tolerated with SC dosing of both daratumumab and bortezomib and produces minimal hematologic toxicity, virtually all newly diagnosed AL patients would be potential candidates. The rapid responses the drug can generate can be associated with rapid organ improvement. The clinician group also noted that the lack of access to daratumumab regimens for the current population of Canadian AL amyloidosis patients who have already received first-line therapy, and in whom daratumumab at relapse could well be life-saving or live-extending, is an important consideration and a particular concern for CMRG physicians. Although this is expected be a limited group of patients, the clinician group stated that these patients deserve the chance to receive daratumumab therapy at disease progression, given the limited range of other options.
Drug Program Input
The drug plans identified jurisdictional implementation issues related to considerations for the initiation and prescribing of therapy and generalizability. The clinical experts consulted by CADTH weighed evidence from the ANDROMEDA trial and other clinical considerations to provide responses to the Provincial Advisory Group’s implementation questions. Table 3 provides more details.
Clinical Evidence
Pivotal Studies and Protocol-Selected Studies
Description of Studies
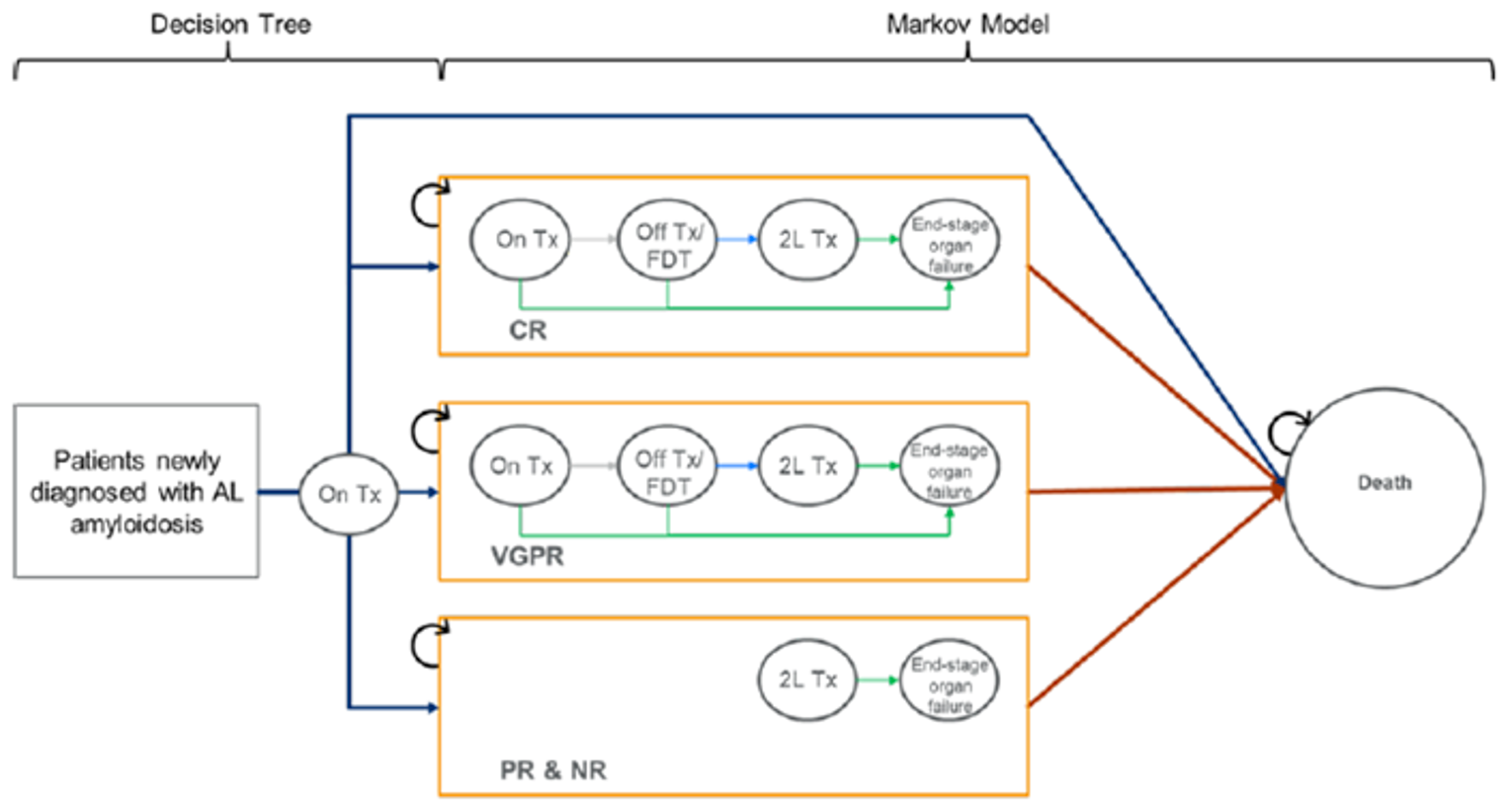
The ANDROMEDA trial is an ongoing, randomized, open-label, active-controlled, phase III, multi-centre trial designed to compare the efficacy of DCyBorD to CyBorD alone in the treatment of patients with newly diagnosed AL amyloidosis. Patients were stratified by cardiac stage based on the Mayo Clinical Cardiac Staging System (stages I, II, and IIIa), ASCT availability in the country of residence, and renal function (creatinine clearance ≥ 60 mL/min or < 60 mL/min) and were randomly assigned in a 1:1 ratio to receive either DCyBorD or CyBorD. The primary end point was a hematological complete response (CR) in the intention-to-treat (ITT) population. The key secondary efficacy end points were major organ deterioration progression-free survival (MOD-PFS), organ response rate, OS, overall hematologic response (CR, very good partial response [VGPR] or partial response [PR]), and time and duration of hematologic response. The HRQoL and medical resource utilization were also evaluated as exploratory end points.
A total of 388 patients were randomized to treatment with either DCyBorD (n = 195) or CyBorD (n = 193). The median age in the study population was 64 years (62 years and 64 years in the DCyBorD and CyBorD treatment arms, respectively). The median number of organs involved at baseline was 2 (range = 1 to 6) and 65.5% of patients had 2 or more organs involved. Cardiac and renal involvement were most common, affecting 71.4% and 59.0% of patients, respectively. Approximately one-third (36.6%) of patients were cardiac stage III at baseline. Of the 202 patients tested for t(11;14) (a translocation between chromosomes 11 and 14) at baseline, t(11;14) was present in 106 (DCyBorD: n = 51; CyBorD: n = 55).
Efficacy Results
At the primary analysis (data cut-off date: February 14, 2020; median follow-up of 11.4 months), 104 patients (53.3%) in the DCyBorD arm and 35 (18.1%) in the CyBorD arm had an independent review committee (IRC)-assessed hematologic CR (relative risk ratio = 2.9; 95% confidence interval [CI], 2.1 to 4.1; P < 0.001).
Hematologic CR rates across cardiac stages were consistent with results observed in the overall population of patients. The hematologic CR rate was higher in the DCyBorD arm compared to the CyBorD arm for all cardiac stages. The difference in hematologic CR rates between the 2 treatment arms increased by Mayo cardiac stage (DCyBorD versus CyBorD: 45% versus 28% for cardiac stage I; 54% versus 20% for stage II, and 58% versus 10% for stage III, respectively). Similarly, the magnitude of hematologic CR rates in the t(11;14) translocation subgroup was similar to that observed in the overall population. Patients in the DCyBorD arm had equally high rates of hematologic CR regardless of t(11;14) translocation, whereas lower hematologic CR rates were observed for patients with t(11;14) treated with CyBorD.
Among the responders, the median time to hematologic CR was 60 days (range = 8 to 299) in the DCyBorD arm and 85 days (range = 14 to 340) in the CyBorD arm. At the time of the primary analysis, the median duration of hematological CR had not been reached in either treatment arm (range = 0.85 to 17.5 months for DCyBorD and 0.03 to 18.4 months for CyBorD). Of the 104 patients who achieved hematological CR in the DCyBorD arm, 4 died while in hematological CR and no patients relapsed following hematological CR. Of the 35 patients who achieved hematological CR in the CyBorD arm, 2 died while in hematologic CR and 2 relapsed following hematologic CR. The hazard ratio (HR) for MOD-PFS for DCyBorD versus CyBorD was 0.58 (95% CI, 0.36 to 0.93).
Among patients who could be evaluated for cardiac response, 41.5% of the DCyBorD and 22.2% of the CyBorD arm had a cardiac response at 6 months. Among patients who could be evaluated for renal response, 53.0% of the DCyBorD arm and 23.9% of the CyBorD arm had a renal response at 6 months. In the updated analysis, the 12-month organ response rates in the DCyBorD and CyBorD arms were 57% and 28%, respectively, for cardiac response, and 57% and 27%, respectively, for renal response.
Median OS was not reached in either treatment arm.
Patient-Reported Outcomes
The median time to improvement for global health status as measured by the European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30 (EORTC QLQ-C30) was 7.8 months in the DCyBorD arm and 16.7 months in the CyBorD arm (HR = 1.53; 95% CI, 1.10 to 2.13). EORTC QLQ-30 global health status showed continued improvement in the DCyBorD arm after 6 cycles when patients were receiving daratumumab monotherapy.
Medical resource utilization was reported for |||| and |||| of patients in the DCyBorD and CyBorD arms, respectively. Use | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| The most common indication for use of medical services was ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Harms Results
Nearly all patients experienced at least 1 treatment-emergent adverse event (TEAE) (DCyBorD: 97.9%; CyBorD: 98.4%). In both treatment arms, TEAEs led to discontinuation of study treatment in approximately 4% of patients. A higher percentage of patients (43.0%) in the DCyBorD arm reported at least 1 serious adverse event (SAE, i.e., a serious TEAE) compared with the CyBorD arm (36.2%). The most commonly reported SAEs (≥ 5% in either treatment arm) were pneumonia (DCyBorD: 7.3%, CyBorD: 4.8%) and cardiac failure (DCyBorD: 6.2%, CyBorD: 4.8%). The incidence of several events was at least 2% higher in the DCyBorD arm, specifically pneumonia (DCyBorD: 7.3%; CyBorD: 4.8%), sepsis (3.1% and 0%, respectively), and cardiac arrest (3.6% and 1.6%, respectively).
Across all cycles, the incidence of any grade of neutropenia (DCyBorD: 10.9%; CyBorD: 6.4%) and grade 3 or 4 neutropenia (DCyBorD: 5.2%; CyBorD: 2.7%) was higher in the DCyBorD arm. The incidence of any grade of infection (DCyBorD: 65.8%; CyBorD: 53.7%), grade 3 or 4 infection (DCyBorD: 16.6%; CyBorD: 10.1%), and serious infection (DCyBorD: 16.1%; CyBorD: 8.5%) was higher in the DCyBorD arm. The most commonly reported infections of any grade (> 10% in either treatment arm) were upper respiratory tract infection (DCyBorD: 25.9%; CyBorD: 11.2%) and pneumonia (10.9% and 6.4%, respectively).
At the time of primary analysis, 27 patients (14.0%) in the DCyBorD arm and 28 patients (14.9%) in the CyBorD arm had died. One patient in the CyBorD arm died before receiving any treatment. A higher proportion of patients in the DCyBorD arm died due to an adverse event (AE) (11.9%) compared with the CyBorD arm (7.4%), and more patients in the CyBorD arm died due to progressive disease (1.0% and 4.8%, respectively) and “other” causes (1.0% and 2.7%, respectively. The most common AEs leading to death (≥ 2% in either treatment arm) were cardiac disorders, including cardiac arrest (DCyBorD: 3.1%; CyBorD: 1.6%), sudden death (3.1% and 1.6%, respectively), and cardiac failure (2.6% and 0.5%, respectively). All patients who died due to a cardiac disorder had cardiac involvement at baseline (DCyBorD: 14 of 14; CyBorD: 7 of 7).
Table 2: Summary of Key Results From the ANDROMEDA Trial — ITT Population
Outcome | DCyBorD | CyBorD |
|---|---|---|
Hematologic complete response | ||
Primary analysisa | ||
n (%) | 104 (53.3) | 35 (18.1) |
Relative risk ratio (95% CI),7 P value | 2.9 (2.1 to 4.1), < 0.001 | |
Updated analysisb | ||
n (%) | 115 (59.0) | 37 (19.2) |
Relative risk ratio (95% CI) | Not reported | |
Time to hematologic response | ||
Median, days (range) | 60 (8 to 299) | 85 (14 to 340) |
Hematologic progression-free survival | ||
n (%) | Not reported (17.4) | Not reported (24.9) |
Hazard ratio (95% CI) | 0.63 (0.40 to 0.97) | |
Organ response | ||
Primary analysisa | ||
Response at 6 months, % | ||
Cardiac response, n of n evaluable (%) | 49 of 118 (41.5) | 26 of 117 (22.2) |
Renal response, n of n evaluable (%) | 62 of 117 (53.0) | 27 of 113 (23.9) |
Updated analysisb | ||
Response at 12 months, % | ||
Cardiac response, n of n evaluable (%) | 67 of 118 (57) | 33 of 117 (28) |
Renal response, n of n evaluable (%) | 67 of 117 (57) | 31 of 113 (27) |
Major organ deterioration progression-free survival | ||
Hazard ratio (95% CI) | 0.58 (0.36 to 0.93) | |
Safety population, N | 193 | 188 |
Any treatment-emergent adverse event | 189 (97.9) | 185 (98.4) |
Serious treatment-emergent adverse events | 83 (43.0) | 68 (36.2) |
Treatment-emergent adverse events leading to discontinuation of study treatment | 8 (4.1) | 8 (4.3) |
Deaths | 27 (14.0) | 28 (14.9) |
CI = confidence interval; CyBorD = cyclophosphamide, bortezomib, and dexamethasone; DCyBorD = daratumumab in combination with cyclophosphamide, bortezomib, and dexamethasone, ITT = intention-to-treat.
aData cut-off: February 14, 2020; median follow-up of 11.4 months.
bMedian follow-up of 20.3 months.
Source: Clinical Study Reports for Darzalex8 and Kastritis et al. (2021).7,9
Critical Appraisal
The ANDROMEDA trial was an open-label study; patients and investigators were not blinded to study assignment, although patient blinding would not have been possible given the differences in the 2 study treatment regimens. Nonetheless, sources of bias that may result from the lack of blinding of patients and investigators to assigned study treatments cannot be ruled out. For example, patient knowledge of their assigned treatment may have affected some safety end points, and HRQoL in particular, and different concomitant supportive care may have been offered to patients in the 2 treatment arms. The primary end point of hematologic CR and organ response were laboratory-based objective measures, which were unlikely to be affected by the open-label design. The longer duration of therapy in the DCyBorD arm and the use of subsequent therapy in the trial are other possible sources of bias. Only the primary outcome of hematologic CR is unaffected by this possible bias and can therefore be considered valid. Use of subsequent therapy may affect other key secondary outcomes (VGPR, PR, and MOD-PFS). This issue is addressed in the primary analysis for MOD-PFS, which employed the inverse probability of censoring weight (IPCW) method to adjust estimates of a treatment effect in the presence of subsequent non–cross-resistant anti–plasma cell therapy, which still showed longer MOD-PFS in the DCyBorD arm. Missing data, including missing organ response assessments and patient attrition (26.6% in the DCyBorD and 35.2% in the CyBorD arm), although not unexpectedly high for a cancer trial setting, may also affect the internal validity of the evidence.
Although the ANDROMEDA trial included a heterogenous population of patients with AL amyloidosis, and a wide range of clinical presentations were well-represented, some groups of patients, including those with advanced cardiac stage, were excluded. The trial-based evidence regarding efficacy and safety of DCyBorD compared to CyBorD in these groups of patients is therefore limited. The comparator of the trial (CyBorD) is not approved for treatment of patients with AL amyloidosis in Canada. However, CyBorD is an appropriate comparator because it is standard of care for newly diagnosed AL amyloidosis in Canada. This is a special instance in which the drug regimen under review (i.e., DCyBorD) is the only Health Canada–approved treatment. The primary and key secondary outcomes and assessment schedule were also reflective of clinical practice. Based on input from the clinical experts consulted by CADTH and clinician groups, patients are assessed in clinical practice every 3 to 6 months. Formal response criteria have been established previously and were included in the ANDROMEDA trial.
Conclusions
Daratumumab plus CyBorD is the first approved treatment for AL amyloidosis in Canada, where no publicly funded therapies are currently available. AL amyloidosis is a rare condition and has a poor prognosis, especially in patients with severe organ involvement. Evidence from the ANDROMEDA trial showed that the addition of daratumumab to CyBorD significantly improved hematological response in newly diagnosed patients with AL amyloidosis without decrements in HRQoL or new safety concerns. The clinical benefit was supported by several secondary end points, including depth of response and organ response. The treatment benefit observed with DCyBorD was consistent in subgroups of patients with poor prognoses, notably patients with cardiac stage II or III and a t(11;14) cytogenic abnormality who respond less well to current treatments. Based on the superiority of DCyBorD to CyBorD alone, as demonstrated by short-term outcomes, current evidence suggests that DCyBorD can fill an important unmet treatment need in this patient population. Longer follow-up data are needed to establish the long-term benefit of adding daratumumab to CyBorD for the treatment of patients with AL amyloidosis.
Introduction
Disease Background
Systemic AL amyloidosis is a rare disease characterized by clonal proliferation of plasma cells with excessive production of monoclonal light chains that transform into misfolded amyloid protein fibrils. The special configuration of these amyloid fibrils makes them insoluble, causing deposition and aggregation in the extracellular matrix and resulting in cytotoxicity and organ dysfunction. AL deposits accumulate predominantly in the heart and kidneys but can affect other organs, including the peripheral nervous system, liver, gastrointestinal tract, and soft tissues.4 Because AL amyloidosis can affect multiple organs simultaneously, it can manifest as a range of non-specific symptoms, such as unexplained heart failure, neuropathy, and hepatomegaly, making it difficult to diagnose AL amyloidosis at early stages. Patients diagnosed at advanced stages, particularly when heart involvement is present, are at high risk of death within a few months.1 There are 2 known risk factors for AL amyloidosis. The first is a pre-existing monoclonal gammopathy, a condition in which abnormal antibodies produced by a small number of plasma cells in the bone marrow are found in the blood. The most common condition of monoclonal gammopathy is monoclonal gammopathy of undetermined significance. In such cases, the relative risk of developing AL amyloidosis is 8.8 compared with individuals without a monoclonal gammopathy of undetermined significance.4,10 The second is a particular single nucleotide polymorphism within the splice site of CCND1 encoding cyclin D1. The variant rs9344, which promotes a translocation between chromosomes 11 and 14 (t[11;14]), has been associated with a higher risk of AL amyloidosis in genetic association studies, suggesting a common genetic susceptibility for the disease.11 Recent data suggest that patients whose plasma cell clones harbour a t(11;14) abnormality may have worse outcomes when treated with common regimens.12,13
Given the rarity of AL amyloidosis, limited epidemiological data on the disease are available. The prevalence estimates were 8.8 to 15.5 individuals per million person-years before 2010, but these estimates have since increased to between 40 and 58 individuals per million person-years (in Europe), owing to improved therapies and OS of patients over the past decade.14,15 The prevalence of AL amyloidosis rises with age, doubling in individuals older than 65 years of age compared with those aged 35 to 54 years. The reported mean age at diagnosis is 63 years, and 55% of patients are men.16 Light chain amyloidosis is an incurable disease and survival is generally poor, particularly among patients with delayed diagnosis, later-stage disease, cardiac involvement, or multiple affected organs. Cardiac damage is a major determinant of survival. The most widely used staging system for AL amyloidosis, developed by the Mayo Clinic, stratifies patients into stages I to IV, based on severity of cardiac involvement (i.e., N-terminal pro–B-type natriuretic peptide [NT-proBNP] and cardiac troponin T levels) and the difference between involved amyloidogenic and uninvolved circulating FLC levels.2,17 All of these markers have been shown to be independent prognostic factors, with increased levels associated with a higher mortality risk.18 The estimated median OS ranges from 7.8 years for stage I to 5.8 years for stage IV.2 The estimated 5-year survival ranges from 59% for patients with stage I disease to 14% for patients with stage IV disease.2 In Canada, based on real-world data from Alberta, the median OS from initiation of first-line treatment for AL amyloidosis was 5.2 years among patients who were diagnosed in 2012 onwards.3
Although most patients are diagnosed at later stages of the disease after overt symptoms appear, cardiac and renal amyloidosis can be detected using the biomarkers of organ involvement (NT-proBNP and albuminuria) during the pre-symptomatic stage of the disease. A diagnosis of amyloidosis should be considered for patients presenting with symptoms such as heart failure with preserved ejection fraction, nephrotic-range proteinuria, peripheral neuropathy, hepatomegaly without imaging abnormalities, a monoclonal gammopathy, or atypical multiple myeloma. In these patients, immunofixation electrophoresis of the serum and urine and an immunoglobulin FLC assay (which assesses the concentration of kappa and lambda FLCs and their ratio in the serum) is carried out to detect precursor AL proteins. Imaging with radio-iodinated serum amyloid P component can identify amyloid deposits in individuals with these symptoms, but this test is not widely available. Because other types of systemic amyloidosis can have clinical presentations that overlap those of AL amyloidosis, a tissue biopsy and histopathological analysis may be necessary to confirm a diagnosis of AL amyloidosis. Once a diagnosis of AL amyloidosis is established, an evaluation of the characteristics of the underlying clone and the extent and severity of organ involvement become essential to guiding the therapeutic strategy.4,5
Standards of Therapy
The therapeutic options that form the basis of the systemic AL amyloidosis treatment paradigm are derived from experiences with multiple myeloma, as both diseases originate in malignant plasma cells. Because cardiac damage is a major determinant of survival, a primary goal of therapy is to improve cardiac function. In AL amyloidosis, therapy is aimed at targeting the B-cell clone responsible for producing aberrant clonal immunoglobulin proteins. A rapid and profound decrease in amyloid precursor proteins can reverse organ dysfunction. The type and intensity of treatment targeting B-cell disease are based on a risk assessment that takes into consideration the characteristics of the patient and levels of cardiac biomarkers. Close monitoring of clonal and organ responses guide the duration of therapy and changes in treatment regimen.4,5
According to the clinical experts consulted by CADTH and clinician groups for the purpose of this review, DCyBorD is the first Health Canada–approved therapy for AL amyloidosis, although it is currently only available through special access programs for a limited treatment duration. It is also funded in some provinces (e.g., Alberta) through special agreements with provincial groups. There is a long track record of using many of the approved therapies for multiple myeloma in AL amyloidosis. Historically, melphalan (either in low doses delivered orally or at high doses in the context of ASCT) has been the standard of care. However, only a minority of patients are suitable for treatment with melphalan, and the risk of transplant-related mortality is relatively high, even in selected patients. With the advent of novel therapies such as proteasome inhibitors, combinations of alkylator-steroid backbone therapy with drugs such as bortezomib have achieved considerably superior outcomes compared with older treatments. Specifically, CyBorD and bortezomib plus melphalan plus dexamethasone have been widely accepted treatments for AL amyloidosis. Bortezomib-based triplet therapy was established as the new standard of care in AL amyloidosis. Many jurisdictions in Canada have already adopted this regimen as the front-line treatment of choice, albeit with the substitution of cyclophosphamide for melphalan as the preferred alkylator (similar to the evolution of such therapy in multiple myeloma). Compared with melphalan, cyclophosphamide offers more predictable and less profound blood-count suppression, easier administration in the setting of renal compromise, and less-permanent damaging effects on the bone marrow, preserving the ability to collect stem cells in patients in the event ASCT becomes an option in the future. Consequently, CyBorD has become the most widely used off-label regimen for the front-line treatment of AL amyloidosis in Canada. If a deep remission is achieved, fixed-duration therapy may be administered for 6 to 12 cycles, after which the patient is monitored for relapse off-therapy. Treatment with ASCT is reserved for a minority of patients with limited (1 or 2) organ involvement, an excellent Karnofsky performance score, and no significant cardiac disease, orthostatic hypotension, factor X deficiency, or extensive gastrointestinal involvement who may not have achieved a sufficiently deep remission with CyBorD yet still meet all eligibility criteria.
There is no consensus second-line treatment for relapsed AL, nor are there any approved or funded regimens. Some patients may become eligible for transplant if their organs improve sufficiently during the first remission, but this is uncommon. More often, patients will be offered, and some will respond to, another course of CyBorD, although data for this approach are lacking. Consolidation of a response with ASCT, after re-induction with CyBorD (particularly if less than a CR is achieved) may be considered to try to deepen and/or prolong the response; however, this is a rare scenario. Lenalidomide plus dexamethasone may be used but has many disadvantages. Specifically, it may result in peripheral edema, may precipitate congestive heart failure in some patients, and is associated with considerable fatigue, which limits the ability to administer an adequate dose. Its main disadvantage is that it rarely produces deep remissions, particularly the CRs necessary to control AL production and further organ damage. Pomalidomide is a more potent immunomodulatory imide drug, but it is available or funded in myeloma only after failure of lenalidomide, is not specifically funded for AL, and therefore is difficult to procure. However, it is generally assumed to produce deeper remissions, is better tolerated than lenalidomide, and is preferred when patients can obtain it through private insurance.
The clinicians indicated that, because AL amyloidosis is eventually fatal in the majority of patients due to organ failure, virtually all patients with systemic AL amyloidosis would be expected to benefit from the addition of a monoclonal antibody to their treatment regimen, in both front-line and relapsed settings.
Drug
Daratumumab is a human immunoglobin G1 kappa monoclonal antibody that binds CD38-expressing cells with high affinity. A multifunctional glycoprotein ectoenzyme, CD38 is highly expressed on the cell surface of diverse hematologic malignancies, including clonal plasma cells that produce the amyloidogenic immunoglobulin AL. Daratumumab leads to rapid and sustained elimination of highly immunosuppressive subsets of CD38+ regulatory T cells, CD38+ myeloid-derived suppressor cells, and CD38+ regulatory B cells. The elimination of these immunosuppressive cells, modulation of CD38 enzymatic activity, and destruction of the malignant myeloma cells is thought to lead to the clonal expansion of CD8+ and CD4+ T cells.
Daratumumab SC was recently approved in the US and the European Union, having demonstrated comparable efficacy and tolerability to the IV formulation. The SC formulation minimizes the risk of volume overload that may be anticipated in patients with AL amyloidosis with cardiac and renal involvement and reduces the risks of infusion-related reactions. Daratumumab SC in combination with CyBorD for the treatment of adult patients with newly diagnosed AL amyloidosis was approved by Health Canada (a Notice of Compliance was issued April 12, 2021) through Project Orbis.6
Darzalex is supplied as a solution for SC injection at 1,800 mg/15 mL (120 mg/mL) and contains recombinant human hyaluronidase PH20, an enzyme used to increase the dispersion and absorption of co-administered daratumumab. According to the product monograph for Darzalex SC, the recommended SC dosage is 1,800 mg over approximately 3 to 5 minutes weekly (for a total of 8 doses) in weeks 1 through 8, every 2 weeks (a total of 8 doses) in weeks 9 to 24, and every 4 weeks from week 25 onward until disease progression or a maximum of 2 years.6
Stakeholder Perspectives
Patient Group Input
This section was prepared by CADTH staff based on the input provided by patient groups. Appendix 1 provides the full patient group input submitted to CADTH.
Myeloma Canada provided input for the review of DCyBorD for the treatment of AL amyloidosis. Myeloma Canada conducted a survey of its patient and caregiver community via email and social media. The survey was available from July 12 to July 25, 2021. The survey received 40 responses, 12 of which were deemed eligible (from 7 patients currently receiving CyBorD, 3 patients currently receiving DCyBorD, and 2 patients waiting to receive treatment). All patients surveyed rated access to effective treatments for AL amyloidosis as extremely important. The respondents who had treatment experience with CyBorD (n = 7) indicated that, of their expectations of a new treatment before taking CyBorD, “minimal side effects” was mentioned by most patients (n = 4), followed by “disease control” (3) and “improved quality of life” (1). All patients treated with CyBorD rated their experience with this treatment regimen as “somewhat tolerable,” “tolerable,” or “very tolerable.” Fatigue and neuropathy were cited as the least tolerable side effects of CyBorD. Patients who had been treated with combined daratumumab and CyBorD rated their overall side effects as “somewhat tolerable,” “tolerable,” or “very tolerable.” Some patients found the side effects of bortezomib and cyclophosphamide intolerable; 1 patient was treated only with daratumumab and dexamethasone. In terms of what is important to patients when it comes to treating their AL amyloidosis, the majority of patient responses described a strong desire for a good or better quality of life and being able to continue daily activities without debilitating side effects of treatment.
Clinician Input
Input From Clinical Experts Consulted by CADTH
All CADTH review teams include at least 1 clinical specialist with expertise regarding the diagnosis and management of the condition for which the drug is indicated. Clinical experts are a critical part of the review team and are involved in all phases of the review process (e.g., providing guidance on the development of the review protocol, assisting in the critical appraisal of clinical evidence, interpreting the clinical relevance of the results, and providing guidance on the potential place in therapy). The following input was provided by clinical specialists with expertise in the diagnosis and management of AL amyloidosis.
Unmet Needs
The clinical experts consulted by CADTH noted that patients with advanced organ damage (high-risk AL amyloidosis patients) tolerate treatment poorly and do not survive long enough to show any hematological or organ response. Early mortality is common, with 30% to 40% of patients dying within a year of diagnosis. The use of daratumumab SC will likely diminish the possibility of worsening heart failure due to volume overloading. The clinical impact and patient experience of being free from organ deterioration is very meaningful and any treatment that can prevent, delay or repair organ damage therefore meets an important treatment need.
Place in Therapy
Daratumumab SC limits the amount of infused fluid, making it more tolerable for patients with heart failure. With daratumumab treatment, the dosages of cyclophosphamide and bortezomib can be reduced, which can help reduce transient hypotension and autonomic neuropathy. Daratumumab will be used as approved for a duration of 2 years in combination with CyBorD in patients newly diagnosed with AL amyloidosis. Treatment with DCyBorD is appropriate for most patients with newly diagnosed AL amyloidosis. Currently no other treatments are approved or generally funded for the treatment of AL amyloidosis in Canada. Access to CyBorD may be available through the sponsor’s special access program, and it is funded in some provinces (e.g., Alberta), through a special agreement with provincial groups. Treatment with DCyBorD will be a practice-changing development.
Patient Population
The clinical experts consulted by CADTH indicated that DCyBorD is expected to lead to good and rapid response, and most patients with AL amyloidosis are likely to benefit from this treatment regimen. In addition, based on the ANDROMEDA trial data, DCyBorD can potentially overcome the effect of a t:(11;14) mutation on survival. Patients harbouring this mutation who generally have poorer prognoses were observed to response well when treated with DCyBorD. In terms of identifying patients best suited for treatment with daratumumab, the clinical expert indicated that early diagnosis is key in AL amyloidosis. The disease is still relatively underdiagnosed, and misdiagnoses of myeloma are common as biopsies are often not pursued in clinical settings. The clinical expert noted that the diagnosis of AL amyloidosis requires adequate infrastructure. Ongoing efforts are being made to increase awareness and facilitate diagnosis across Canada.
Assessing Response to Treatment
The clinical expert consulted by CADTH indicated that no biomarkers exist to predict treatment response. However, a difference between involved and uninvolved FLCs of less than 10 and involved FLCs of less than 20 at 1 month have been described as potential prognostic markers of better survival. In clinical practice, as in clinical trials, hematological response and organ response correlate with survival, and outcomes of deeper and faster responses are used to determine response to treatment. Improved hematologic response, PFS, and OS, as well as improved organ response and quality of life, are considered clinically meaningful responses to treatment. With respect to frequency of assessment of treatment response, the clinical experts indicated that monthly assessments are common in Canada, but decisions on adequate hematologic and organ responses are made 3 and 6 months after initiation of treatment.
Discontinuing Treatment
Treatment with daratumumab should be discontinued if it is not tolerated or in the event of either hematological or organ progression.
Prescribing Conditions
The clinical experts consulted by CADTH considered all cancer centres across Canada to be appropriate settings for providing daratumumab SC to patients with AL amyloidosis.
Additional Considerations
Daratumumab is a practice-changing regimen for a disease with no approved or funded treatments. To date, no long-term follow-up data on the DCyBorD regimen are available. However, deeper responses are associated with prolonged survival and, based on the ANDROMEDA trial, there is a high rate of deep response. At present, it is difficult to estimate the potential of DCyBorD as a curative treatment strategy. However, based on the available trial data, the regimen is expected to at least induce long-lasting response.
Clinician Group Input
This section was prepared by CADTH staff based on the input provided by clinician groups. Appendix 2 provides the full clinical group input submitted to CADTH.
Clinician input was received from the CMRG and the Ontario Health (Cancer Care Ontario) Hematology Cancer Drug Advisory Committee. The CMRG is a charitable organization whose membership consists of physicians specializing in plasma cell dyscrasias, including multiple myeloma and AL amyloidosis, at 22 major academic medical centres in Canada. Its main activities consist of conducting academic clinical trials to improve patient outcomes, maintaining the national plasma cell dyscrasias database, and generating consensus statements for the management of multiple myeloma and related plasma cell dyscrasias. The Ontario Health Hematology Cancer Drug Advisory Committee provides evidence-based clinical and health system guidance on drug-related issues in support of Cancer Care Ontario’s mandate, including the Provincial Drug Reimbursement Programs and the Systemic Treatment Program. The clinician groups noted that the most important goals of any plasma cell–directed therapy is to achieve deep and rapid elimination of clonal plasma cells and the monoclonal protein product they secrete. Patients with AL amyloidosis tolerate all anti–plasma cell treatments less well than myeloma patients due to the underlying organ damage intrinsic to this disease (with compromised kidneys, heart, and liver). Based on data from the ANDROMEDA trial’s control arm, the deep responses necessary for organ improvement and prolonged disease control are not optimal with CyBorD alone. Given the toxic effect of the amyloid AL, it is vital to achieve rapid and deep responses as early in the disease course as possible to limit ongoing damage to organs. It is therefore imperative to have access to first-line therapies that produce deep and rapid responses. The advent of DCyBorD is a major breakthrough in the treatment of a disease that, if not treated quickly and with deep responses, can lead to irreversible organ damage and result in significant morbidity and subsequent poor quality of life. The clinician groups noted that DCyBorD is intended for use as a first-line therapy. Given the pathophysiology of AL amyloidosis, there is no rationale or justification to try a less effective first-line therapy. In addition, the SC administration of daratumumab used in the ANDROMEDA trial reduces the potentially serious effect of volume overloading that can be seen in patients with cardiac and renal amyloid involvement.
The clinician groups indicated that adding daratumumab to the current standard of care of CyBorD will represent a major shift in the treatment of AL amyloidosis. This regimen would be widely adopted for patients with AL amyloidosis because of the favourable and pivotal results of ANDROMEDA study. Given DCyBorD’s effectiveness, it is likely fewer patients will require ASCT, with its attendant risk of morbidity and increased risk of mortality. The use of less effective yet expensive regimens, such as immunomodulatory imide drugs, would be delayed or perhaps be unnecessary. Because DCyBorD is well tolerated with SC dosing of both daratumumab and bortezomib and produces minimal hematologic toxicity, virtually all newly diagnosed patients would be potential candidates. The rapid responses that DCyBorD can generate can be associated with rapid organ improvement. The clinician group also noted that the lack of access to daratumumab regimens for the current population of Canadian patients with AL amyloidosis who have already received first-line therapy and in whom daratumumab at relapse could be life-saving or live-extending, is an important consideration and of particular concern to CMRG physicians. Although this is expected be a relatively small group of patients, the clinician groups believe that these patients deserve the chance to receive daratumumab therapy at progression, given the limited range of other treatment options.
Drug Program Input
The drug programs provide input on each drug being reviewed through CADTH’s reimbursement review processes by identifying issues that may affect their ability to implement a recommendation. The implementation questions and corresponding responses from the clinical experts consulted by CADTH, as well as comments or issues raised by the drug plans for consideration, are summarized in Table 3.
Table 3: Summary of Drug Plan Input and Clinical Expert Response
Drug program implementation questions | Clinical expert response |
|---|---|
Policy considerations for reimbursing the drug | |
Considerations for initiation of therapy | |
Only a small proportion of patients in the ANDROMEDA trial proceeded to ASCT (6.7% in the DCyBorD group and 10.6% in the CyBorD group). Please confirm that patients who proceed to ASCT are not eligible for daratumumab maintenance therapy post-ASCT. | Patients who undergo ASCT should be eligible for DCyBorD at relapse if they have adequate heart and kidney function. |
Would patients who complete 2 years of daratumumab maintenance therapy and subsequently relapse be eligible for re-treatment with DCyBorD followed by daratumumab maintenance? If so, what is the appropriate interval for re-treatment? | As the ANDROMEDA trial is ongoing, longer follow-up data will inform the duration of maintenance therapy and re-treatment at relapse. In the absence of long-term trial data, if a patient achieved a good response with the first round of DCyBorD treatment, they could be re-treated with the same regimen at relapse, but the re-treatment response could be shorter. |
Would patients who complete up to 6 cycles of DCyBorD followed by ASCT be eligible for re-treatment with DCyBorD and daratumumab maintenance? If so, what is the appropriate interval for re-treatment? | If a deep response is achieved with DCyBorD, patients may not need ASCT. If long-term data from the ANDROMEDIA trial show positive results, it could eliminate the need for ASCT for some patients. There are currently no data to inform on an appropriate interval for re-treatment. |
Considerations for prescribing of therapy | |
For patients unable to tolerate the SC formulation, would an IV equivalent for daratumumab be appropriate? | Daratumumab can be administered intravenously when SC administration is not possible or contraindicated. |
| For consideration by the CADTH pan-Canadian Oncology Drug Review Expert Review Committee. |
Special implementation issues | |
Generalizability | |
Please confirm that patients with advanced cardiac disease (Mayo stage IIIB or NYHA class IIIB or 4) would not be eligible for DCyBorD. | Patients with advanced cardiac disease (e.g., Mayo stage III or IV or NYHA class IIIB or IV) should be eligible for DCyBorD, and treatment with DCyBorD should not be limited by cardiac stage. If patients are suitable for CyBorD treatment, the addition of daratumumab is expected to lead to a better response without |
(continued) | causing significant toxicity. One of the clinical experts consulted by CADTH noted that access to NT-proBNP could be limited in some centres (which may only have access to B-type natriuretic peptide values) and as a result it may not be possible to determine the Mayo cardiac stage. In such situations, other risk scores may be used, and determination of cardiac involvement may also be partly based on clinical opinion. |
On a time-limited basis, should patients currently on CyBorD (or another regimen) but whose disease has not yet progressed be switched to DCyBorD? If yes, is there an appropriate time frame based on the number of cycles? On a time-limited basis, should patients who recently completed CyBorD but whose disease has not yet progressed be eligible for daratumumab maintenance? If yes, is there an appropriate time frame? | In clinical practice, response is assessed 3 months from initiation of treatment. If there is no response at this time, daratumumab can be added to CyBorD. Patients who achieve adequate response on CyBorD do not need to be treated with daratumumab as maintenance therapy. |
ASCT = autologous stem cell transplant; CyBorD = cyclophosphamide, bortezomib, and dexamethasone; DCyBorD = daratumumab in combination with cyclophosphamide, bortezomib, and dexamethasone; NT-proBNP = N-terminal pro–B-type natriuretic peptide; NYHA = New York Heart Association; SC = subcutaneous.
Clinical Evidence
The clinical evidence included in the CADTH review of daratumumab SC includes a systematic review outlining pivotal studies provided in the sponsor’s submission to CADTH as well as those studies that were selected according to an a priori protocol.
Systematic Review of Pivotal and Protocol-Selected Studies
Objectives
To evaluate the efficacy and safety of daratumumab SC in combination with bortezomib, cyclophosphamide, and dexamethasone for the treatment of adult patients with newly diagnosed AL amyloidosis.
Methods
Studies selected for inclusion in the systematic review included pivotal studies provided in the sponsor’s submission to CADTH and Health Canada, as well as those meeting the selection criteria presented in Table 4. Outcomes included in the CADTH review protocol reflect those considered to be important to patients, clinicians, and drug plans.
Table 4: Inclusion Criteria for the Systematic Review
Criteria | Description |
|---|---|
Population | Adult patients (≥ 18 years of age) with newly diagnosed AL amyloidosis Subgroups of interest:
|
Intervention | Daratumumab SC injection in combination with cyclophosphamide, bortezomib, and dexamethasone (DCyBorD) up to a maximum duration of 2 years |
Comparator | Cyclophosphamide, bortezomib, and dexamethasone (CyBorD) |
Outcomes | Efficacy outcomes:
Patient-reported outcomes:
Medical resource utilization
Harms outcomes:
Notable harms:
|
Study designs | Published and unpublished phase III and phase IV randomized controlled trials |
PFS = progression-free survival; vs. = versus.
The literature search for clinical studies was performed by an information specialist using a peer-reviewed search strategy according to the PRESS Peer Review of Electronic Search Strategies checklist.19
Published literature was identified by searching the following bibliographic databases: MEDLINE All (1946–) via Ovid and Embase (1974–) via Ovid. All Ovid searches were run simultaneously as a multi-file search. Duplicates were removed using Ovid deduplication for multi-file searches, followed by manual deduplication in Endnote. The search strategy comprised both controlled vocabulary, such as the National Library of Medicine’s MeSH (Medical Subject Headings), and keywords. The main search concepts were daratumumab combined with bortezomib, cyclophosphamide, dexamethasone, and amyloidosis. Clinical trials registries searched included the US National Institutes of Health’s clinicaltrials.gov, WHO’s International Clinical Trials Registry Platform search portal, Health Canada’s Clinical Trials Database, and the European Union Clinical Trials Register.
No filters were applied to limit the retrieval by study type. Retrieval was not limited by publication date or by language. Conference abstracts were excluded from the search results. Appendix 1 provides detailed search strategies.
The initial search was completed on August 11, 2021. Regular alerts updated the search until the meeting of the CADTH pan-Canadian Oncology Drug Review Expert Committee (pERC) on December 1, 2021.
Grey literature (literature that is not commercially published) was identified by searching relevant websites from the Grey Matters: A Practical Tool For Searching Health-Related Grey Literature resource.20 Included in this search were the websites of regulatory agencies (US FDA and European Medicines Agency). Google was used to search for additional internet-based materials. Appendix 1 provides more information on the grey literature search strategy.
Two CADTH clinical reviewers independently selected studies for inclusion in the review based on titles and abstracts, according to the predetermined protocol. Full-text articles of all citations considered potentially relevant by at least 1 reviewer were acquired. Reviewers independently made the final selection of studies to be included in the review, and differences were resolved through discussion.
A focused literature search for network meta-analyses dealing with amyloidosis or daratumumab combined with bortezomib, cyclophosphamide, and dexamethasone was run in MEDLINE All (1946) on August 12, 2021. No search limits were applied.
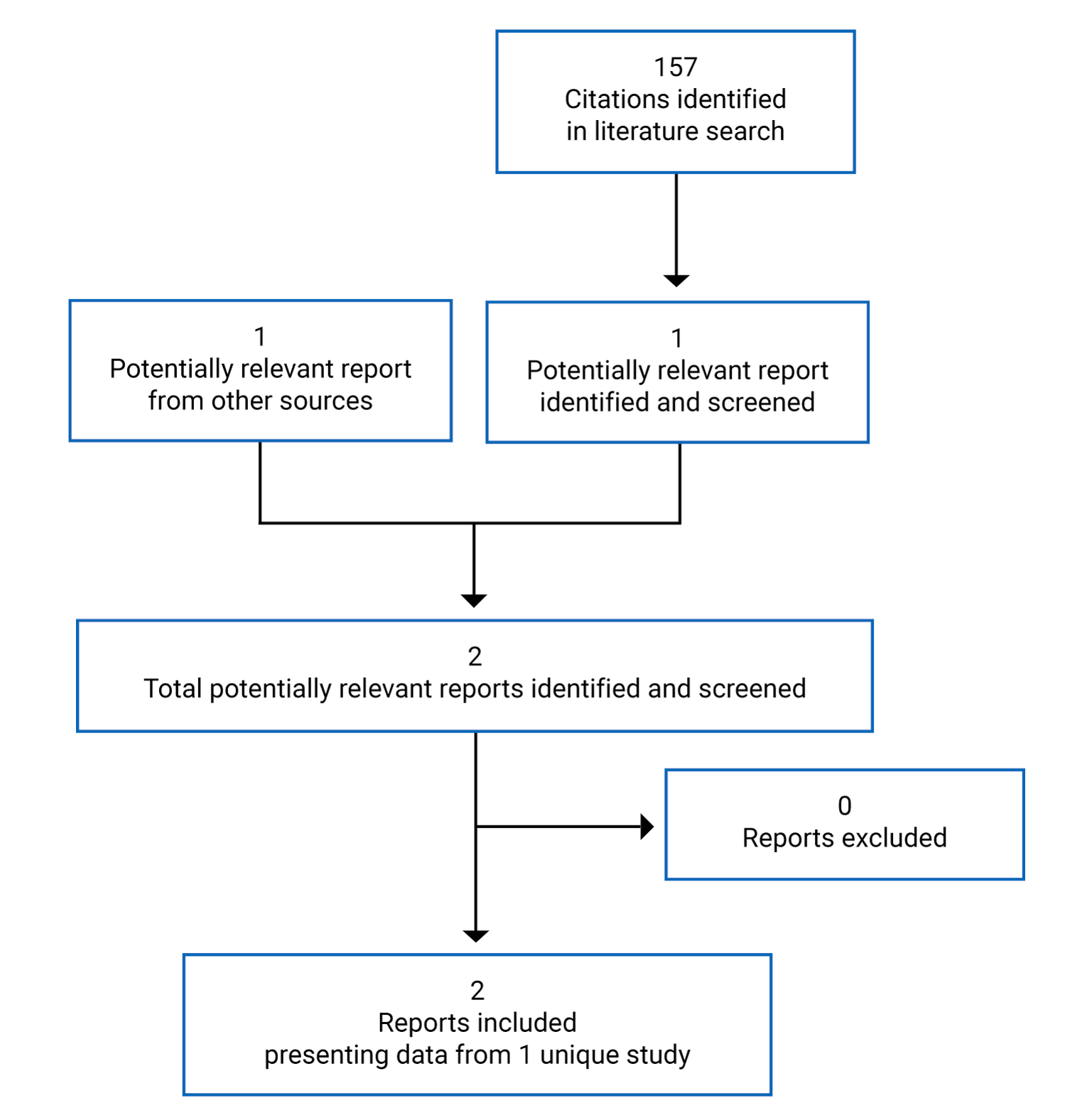
Findings From the Literature
A total of 157 studies were identified; 2 potentially relevant citations were retrieved for full-text screening. These citations reported on the ANDROMEDA trial and were retained (Figure 1). The included study is summarized in Table 5. Information relevant to this report was also derived from the submission to CADTH (Clinical Study Report and other technical documents).
Table 5: Details of the ANDROMEDA Study
Criteria | Description |
|---|---|
Design and population | |
Study design | Phase III, randomized, open-label, active-controlled, multi-centre |
Locations | 109 sites in 22 countries (Australia, Belgium, Brazil, Canada, China, Denmark, France, Germany, Greece, Hungary, Israel, Italy, Japan, Mexico, the Netherlands, Poland, Spain, South Korea, Sweden, Turkey, UK, and US) |
Study duration | October 10, 2017, to ongoing Patient enrolment: May 3, 2018, to August 15, 2019 |
Data cut-off date | February 14, 2020 (date of last observation for last patient recorded as part of the database for primary analysis) |
Number of patients randomized (randomization ratio) | N = 388 (1:1) |
Main inclusion criteria |
|
Main exclusion criteria |
|
(continued) |
|
Drugs | |
Comparator | CyBorD: Cyclophosphamide (1.3 mg/m2 orally or intravenously), bortezomib (1.3 mg/m2 of body surface area), dexamethasone (40 mg orally or intravenously once weekly), for 6 cycles of 28 days each |
Intervention | DCyBorD: Cyclophosphamide (1.3 mg/m2 orally or intravenously), bortezomib (1.3 mg/m2 of body surface area), dexamethasone (40 mg orally or intravenously once weekly), for 6 cycles of 28 days each plus Daratumumab 1,800 mg per 15 mL administered SC, weekly in cycles 1 and 2, every 2 weeks in cycles 3 through 6, and every 4 weeks thereafter until disease progression or the start of subsequent therapy, or for a maximum of 24 cycles from start of the trial |
Duration | |
Phase | |
Safety run-in (non-randomized) | 2 years (n = 28) daratumumab SC weekly in cycles 1 to 2, every 2 weeks in cycles 3 to 6, and every 4 weeks thereafter for up to 2 years; CyBorD was given weekly for 6 cycles; patients received a median of 16 (range = 1 to 23) treatment cycles |
Screening | Up to 28 days before treatment initiation |
Open-label | CyBorD: 24 weeks DCyBorD: 24 weeks for CyBorD and up to 24 months for daratumumab SC monotherapy |
Follow-up | Ongoing |
Outcomes | |
Primary end point | Hematologic CR assessed by an independent review committee |
Secondary and exploratory end points | Secondary end points:
Exploratory end points:
Safety:
|
Notes | |
Publications | Kastritis et al. (2021)7 |
AE = adverse event; AL = light chain; CR = complete response; CyBorD = cyclophosphamide, bortezomib, and dexamethasone; DCyBorD = daratumumab in combination with cyclophosphamide, bortezomib, and dexamethasone; EFS = event-free survival; MOD = major organ deterioration; NT-ProBNP = N-terminal pro–B-type natriuretic peptide; PFS = progression-free survival; SC = subcutaneous; TEAE = treatment-emergent adverse event; ULN = upper limit of normal; VGPR = very good partial response.
Source: Clinical Study Report for Darzalex.8
Description of the ANDROMEDA Trial
The ANDROMEDA trial is an ongoing, randomized, open-label, active-controlled, phase III, multi-centre trial designed to compare the efficacy of DCyBorD to CyBorD alone in the treatment of patients with newly diagnosed AL amyloidosis. The trial was conducted in 109 sites across 22 countries, with 6 sites in Canada, including British Columbia, Alberta, Ontario, and Quebec (n = 19). The randomized phase of the study consisted of a screening phase (up to 28 days before cycle 1, day 1), a treatment phase (from cycle 1, day 1 until study treatment discontinuation), a post-treatment observation phase, and a long-term follow-up phase. Of the 583 patients who were assessed for eligibility, 195 did not meet eligibility criteria; therefore, 388 patients were included in the study. The clinical cut-off date for the primary analysis was February 14, 2020. The trial was funded by Janssen Research and Development. An overview of the ANDROMEDA trial design is presented in Figure 2.
Randomization and treatment allocation: Patients were stratified by cardiac stage based on the Mayo Clinical Cardiac Staging System (stages I, II, and IIIa), ASCT availability (i.e., countries that typically offer transplant for patients with AL amyloidosis and countries that do not offer stem cell transplant for patients with AL amyloidosis), and renal function (creatinine clearance ≥ 60 mL/min or < 60 mL/min). Patients were randomly assigned in a 1:1 ratio to receive either DCyBorD or CyBorD alone, after balancing for cardiac stage, renal function, and availability of ASCT. The method of randomization was randomly permuted blocks. An interactive web-based randomization system was used to assign each patient a unique patient number.
Blinding: As ANDROMEDA was an open-label study, blinding procedures were not applicable. The IRC was blinded to patients’ study treatment assignment.
Study phases: A 2-year safety run-in phase (n = 28) was conducted before the randomized phase of the trial to evaluate the SC formulation of daratumumab in combination with CyBorD. Enrolment in the safety run-in cohort was staggered to allow at least 48 hours before administering daratumumab SC to the next enrolled participant and to allow enough time to assess any new safety information. In the randomized portion of the study, patients were randomized in a 1:1 ratio to CyBorD or DCyBorD treatment arms. All treatment cycles were 4 weeks (28 days) in length. In the CyBorD arm, the drug was administered for a maximum of 6 cycles (24 weeks). Patients randomized to the DCyBorD arm received daratumumab SC at a fixed dose of 1,800 mg. After cycle 6, patients continued to receive daratumumab as monotherapy on day 1 of subsequent 28-day cycles until disease progression, start of subsequent therapy, or a maximum of 2 years from the start of the treatment. In the CyBorD arm, data on AEs were collected up to 30 days after cycle 6 day 22.
Protocol Amendments
Three amendments were made to the protocol (on April 2018, January 2019, and October 2019). The changes included revisions to the protocol to better define requests for clarification from health authorities and to revise the AL amyloidosis response consensus criteria (e.g., clarified censoring criteria of data for some secondary end points, updated renal organ response criteria, excluded patients with hypersensitivity or contraindication to cyclophosphamide); text for identification of hepatitis B virus reactivation, testing, and management of patients was added in response to identification of a new important risk (hepatitis B reactivation); and aggregated (hematologic and organ) PFS was split into specific hematologic PFS which was moved to an exploratory end point and severity criteria for AEs were revised to align with version 4.03 of the National Cancer Institute’s Common Terminology Criteria for Adverse Events severity definitions.
Figure 2: ANDROMEDA Trial Design
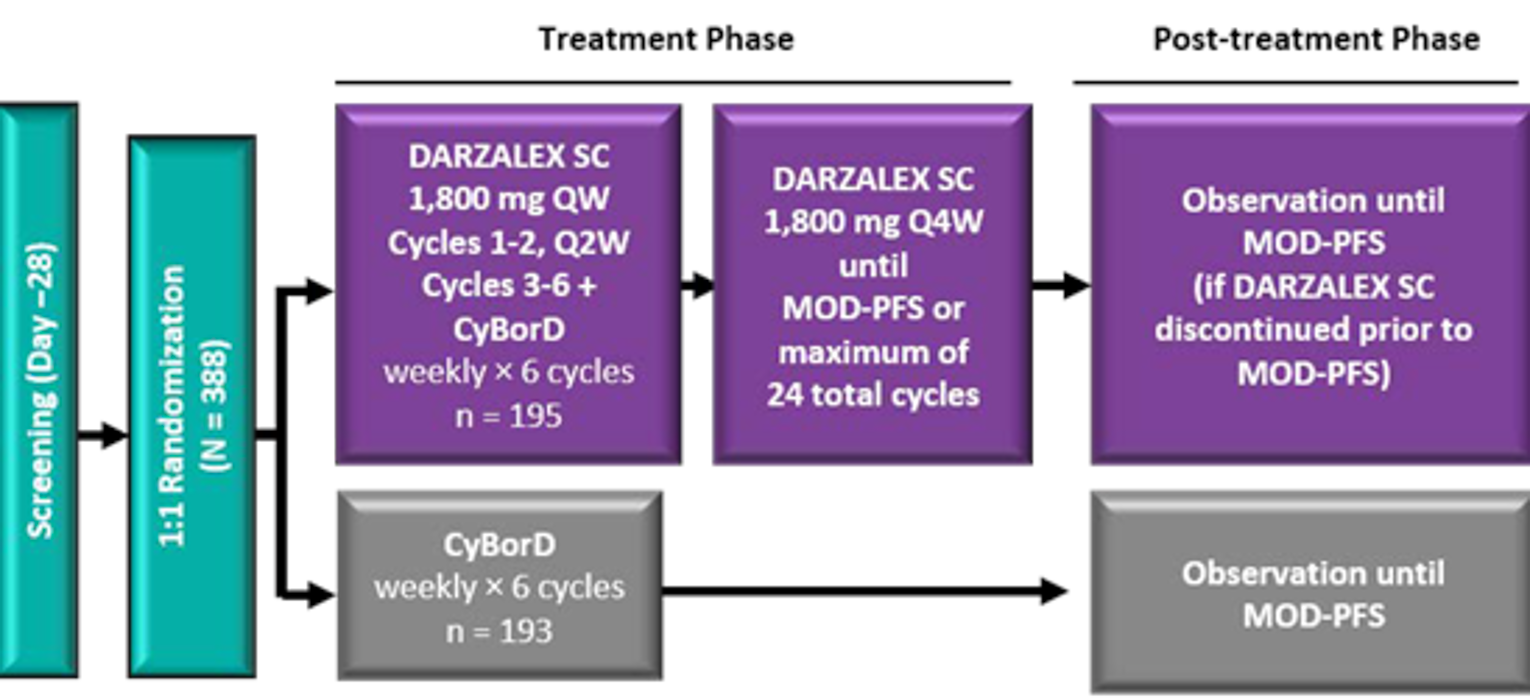
CyBorD = cyclophosphamide, bortezomib, and dexamethasone; MOD-PFS = major organ deterioration progression-free survival; Q1W = weekly; Q2W = every 2 weeks; Q4W = every 4 weeks; SC = subcutaneous.
Source: Clinical Study Report for Darzalex.8
Populations
Inclusion and Exclusion Criteria
Eligible patients had a confirmed histopathological diagnosis of AL amyloidosis, involvement in 1 or more organ(s), measurable hematologic disease (i.e., via serum FLC criteria or serum M-protein) and an Eastern Cooperative Oncology Group Performance Score (ECOG PS) score of 0 to 2. Patients were excluded if they had advanced stage IIIB disease on the European modification of the Mayo Cardiac Staging System, a previous or current diagnosis of symptomatic multiple myeloma, evidence of significant cardiovascular conditions or abnormal liver enzyme levels (i.e., alanine aminotransferase or aspartate aminotransferase > 2.5 times the upper limit of normal [ULN]), non-AL amyloidosis, a planned ASCT during the first 6 cycles of treatment, or received prior therapy for AL amyloidosis or multiple myeloma including anti-CD38 medications (with the exception of 160 mg of dexamethasone or an equivalent corticosteroid with maximum exposure before randomization).
Baseline Characteristics
At that time of the data cut-off date (February 14, 2020), 388 patients were randomized to treatment with either DCyBorD (n = 195) or CyBorD (n = 193) The median age in the study population was 64 years (62 years and 64 years in the DCyBorD and CyBorD treatment arms, respectively). The median time since amyloidosis diagnosis was 48 days in the DCyBorD arm and 43 days in the CyBorD arm. The median number of organs involved at baseline was 2 in both treatment arms (range = 1 to 6) and 65.5% of patients had 2 or more organs involved. Cardiac and renal involvement were most common, affecting 71.4% and 59.0% of patients, respectively. Most patients were either cardiac stage II (DCyBorD: 39.0%; CyBorD: 41.5%) or IIIA (DCyBorD: 35.9%; CyBorD: 33.2%) and renal stage I (DCyBorD: 55.4%; CyBorD: 52.3%) at baseline (Table 6). A total of 106 of the 202 patients tested at baseline had a t(11;14) mutation present (DCyBorD: 51 patients; CyBorD: 55 patients).
Table 6: Baseline Demographic and Disease Characteristics — ITT Population
Characteristic | DCyBorD (N = 195) | CyBorD (N = 193) |
|---|---|---|
Age (years), median (range) | 62 (34 to 87) | 64 (35 to 86) |
Sex, n (%) | ||
Female | 87 (44.6) | 76 (39.4) |
Male | 108 (55.4) | 117 (60.6) |
Race, n (%) | ||
American-Indian or Alaska Native | 1 (0.5) | 2 (1.0) |
Asian | 30 (15.4) | 34 (17.6) |
Black or African American | 6 (3.1) | 7 (3.6) |
Native Hawaiian or other Pacific Islander | 0 | 1 (0.5) |
White | 151 (77.4) | 143 (74.1) |
Multiple | 0 | 1 (0.5) |
Unknown | 7 (3.6) | 5 (2.6) |
ECOG PS, n (%) | ||
0 | 90 (46.2) | 71 (36.8) |
1 | 86 (44.1) | 106 (54.9) |
2 | 19 (9.7) | 16 (8.3) |
Median time since amyloidosis diagnosis, days (range) | 48 (8 to 1,611) | 43 (5 to 1,102) |
AL isotype, n (%) | ||
Lambda | 158 (81.0) | 149 (77.2) |
Kappa | 37 (19.0) | 44 (22.8) |
Involved organs, median (range) | 2 (1 to 5) | 2 (1 to 6) |
Distribution, n (%) | ||
Heart | 140 (71.8) | 137 (71.0) |
Kidney | 115 (59.0) | 114 (59.1) |
Liver | 15 (7.7) | 16 (8.3) |
Other | 127 (65.1) | 124 (64.2) |
Cardiac stage, n (%) | ||
I | 47 (24.1) | 43 (22.3) |
II | 76 (39.0) | 80 (41.5) |
IIIA | 70 (35.9) | 64 (33.2) |
IIIB | 2 (1.0) | 6 (3.1) |
Renal stage, n/total n (%) | ||
I | 107/193 (55.4) | 101/193 (52.3) |
II | 67/193 (34.7) | 74/193 (38.3) |
III | 19/193 (9.8) | 18/193 (9.3) |
Creatinine clearance, n (%) | ||
< 60 mL/min | 69 (35.4) | 62 (32.1) |
≥ 60 mL/min | 126 (64.6) | 131 (67.9) |
AL = light chain; CyBorD = cyclophosphamide, bortezomib, and dexamethasone; DCyBorD = daratumumab in combination with cyclophosphamide, bortezomib, and dexamethasone; ECOG PS = Eastern Cooperative Oncology Group Performance Score; ITT = intention to treat.
Source: Clinical Report for Darzalex.8
Interventions
All patients in both treatment arms received SC bortezomib at a dose of 1.3 mg/m2 of body surface area, cyclophosphamide at a dose of 300 mg/m2 orally or intravenously (500 mg maximum weekly dose), and dexamethasone at a dose of 40 mg orally or intravenously once weekly for 6 cycles of 28 days each. Patients who were assigned to the DCyBorD arm received 1,800 mg of daratumumab per 15 mL administered subcutaneously, co-formulated with recombinant human hyaluronidase PH20, weekly in cycle 1 and cycle 2, every 2 weeks in cycles 3 through 6, and every 4 weeks thereafter until disease progression, start of subsequent therapy, or for a maximum of 24 cycles (2 years) from the start of the trial, whichever occurred first. Patients receiving daratumumab were given pre-infusion and post-infusion medications to prevent infusion-related reactions. Pre-infusion medications administered 1 hour to 3 hours before each SC daratumumab administration included dexamethasone or long-lasting corticosteroids, antipyretics (e.g., acetaminophen), antihistamines (e.g., diphenhydramine or equivalent), and montelukast (a leukotriene inhibitor). Post-infusion medications included low-dose methylprednisolone (≤ 20 mg) or equivalent, the day after the daratumumab SC infusion. Additional post-infusion medications were administered based on patient’s risk factors. For example, patients with a high risk of respiratory complications, including asthma and chronic obstructive pulmonary disease, could receive antihistamines, leukotriene inhibitors, short-acting beta-2 adrenergic receptor agonists, or long-lasting bronchodilators. Dose modifications to daratumumab were not allowed in response to toxicity. Dose delay was the only permitted method of managing toxicity for daratumumab.
Outcomes
The end points identified in the CADTH review protocol that were assessed in the clinical trials included in this review are detailed in the following section. A detailed discussion and critical appraisal of the outcome measures are provided in Appendix 2.
Efficacy
The primary end point was a hematological CR in the ITT population at the time of the data cut-off date for primary analyses (February 14, 2020). The response had to be confirmed by a subsequent assessment, during or after the trial treatment by the IRC, which consisted of 3 experts in AL amyloidosis. Efficacy outcomes are defined in Table 7.
Disease evaluations were performed on the scheduled assessment day (± 5 days) every 4 weeks during cycles 1 through 6 and every 8 weeks from cycle 7 and beyond, on the scheduled assessment and included:
serum immunofixation, serum M-protein quantitation by electrophoresis, serum FLC assay, 24-hour urine M-protein quantitation by electrophoresis, and urine immunofixation for assessment of hematologic response
assessment of cardiac biomarkers (NT-proBNP, troponin T, and high sensitivity troponin T), New York Heart Association classification, and ejection fraction to monitor cardiac response
assessment of estimated glomerular filtration rate and 24-hour proteinuria for kidney response
assessment of alkaline phosphatase for hepatic response
bone marrow aspirate for assessing minimal residual disease by next generation sequencing.
Evaluation of disease response and progression were conducted in accordance with the consensus guidelines for AL amyloidosis and renal response and progression criteria by Palladini (2014).21,22 All assessments, with the exception of New York Heart Association classification, were performed by a central laboratory. Clarifications to these criteria were implemented during the conduct of the study based on emerging literature and guidance from the study steering committee and IRC.
According to the study protocol, patients who started subsequent therapy before meeting the MOD-PFS end point were to continue disease assessments until disease progression or MOD-PFS was observed. The IRC provided independent determination of progressive disease and response to ensure consistent evaluation across all patients in the study.
Patient-Reported Outcomes
Medical resource utilization and HRQoL were evaluated electronically. Physical functioning, symptom improvement, functional improvement, health utility, and HRQoL were measured using the Short Form (36) Health Survey version 2 (SF-36v2), the EORTC QLQ-C30 with supplemental symptom items, and the EQ-5D-5L. Scoring for each instrument was based on instrument-developer guidelines. No imputation was performed for missing data. The EORTC QLQ-30 is a patient-reported, cancer-specific quality-of-life questionnaire using 4- and 7-point Likert scales. The EQ-5D-5L is a preference-based measure of HRQoL consisting of 5 domains (mobility, self-care, usual activities, pain and/or discomfort, and anxiety and/or depression). The SF-36v2 is an 8-dimension patient-reported generic instrument that measures functional health and well-being. Improvement in HRQoL was defined as change from baseline in the EORTC QLQ-C30 global health status score. Improvement in mental functioning was defined as the change from baseline in the SF-36v2 mental health component summary (MCS). Although these instruments are used extensively in evaluations of other cancers, there is limited evidence supporting adequate validity, reliability, and responsiveness in patients with AL amyloidosis, and no minimally important difference (MID) has been defined for this patient population (Appendix 2).
Medical resource utilization associated with protocol-driven medical encounters and safety monitoring was collected in an electronic case report form (eCRF) by the investigator and study-site personnel. All hospitalizations and reason for hospitalizations were recorded. The medical encounters summary (yes or no) question in the eCFR was answered at each disease assessment (cycles 1 to 6 every 28 days, cycle 7 and after, and during the post-treatment observation period every 8 weeks), and every 16 weeks during the long-term follow-up phase of the study. If the patient required any additional encounters other than those mandated per protocol since the last disease evaluation visit, a medical encounters eCRF page was required to be completed.
Safety
AEs were recorded based on the definition of the International Council of Harmonisation criteria. Assessments included clinical laboratory tests (hematology, serum chemistry, and urinalysis), vital sign measurements, physical examinations, electrocardiograms, and ECOG PS. Serious adverse events were defined based on International Council of Harmonisation and European Union guidelines on pharmacovigilance for medicinal products for human use.
AEs were either reported by the patient (or, when appropriate, by a caregiver, surrogate, or the patient's legally acceptable representative) from the time a signed and dated informed consent was obtained until 30 days following the last dose of study treatment, withdrawal of consent for study participation by subject, or start of subsequent therapy, whichever came first. All AEs were described and recorded on the patient’s source document and eCRF, which included the date of onset, seriousness, severity, outcome, action taken, and relationship as evaluated by the investigator. All AEs were followed to satisfactory resolution or a clinically stable end point. In the event of an SAE or unexpected AE, the medical monitor was notified as specified in the protocol.
Statistical Analysis
Sample Size Calculations
The sample size for the ANDROMEDA trial was based on the alternative hypothesis of a 15% improvement in hematologic CR. Taking a hematologic CR rate estimated to be 25% for the CyBorD arm and adding a 15% improvement translated to a hematologic CR rate of 40% for the DCyBorD arm. Approximately 360 patients (180 per arm) would provide more than 85% power to detect a 15% improvement in hematologic CR using a likelihood ratio test with a 2-sided alpha of 0.05. Analysis of the hematologic CR rate at 6 months was performed similarly to the primary end point of hematologic CR rate.
The post-treatment observation phase was to continue until approximately 200 MOD-PFS events had been observed. Therefore, the trial was to achieve an approximately 80% power to detect a 33% reduction in the risk of hematologic progression, major organ deterioration, need for subsequent non–cross-resistant anti–plasma cell therapy use for suboptimal hematologic response, resistant amyloidosis–related organ dysfunction, or death (HR for DCyBorD versus CyBorD] = 0.67), with a log-rank test (2-sided alpha = 0.05).
Analysis Populations
Efficacy analyses were performed in the ITT population, which included all the patients who underwent randomization.
The safety population comprised patients who received at least 1 dose of trial treatment.
Analyses of Outcomes
The primary comparison of the 2 randomized treatments was made with respect to hematologic CR based on an IRC assessment using the Cochran-Mantel-Haenszel chi-square test in the ITT population stratified by cardiac risk, countries that typically offer transplant for patients with AL amyloidosis, and renal function. All binary secondary end points were analyzed using the Cochran-Mantel-Haenszel chi-square test. If the between-group difference for the primary end point of hematologic CR was significant, the major secondary end point of MOD-PFS and OS were tested (in that order) using a hierarchical testing approach that controls the type I error.23
For time-to-event end points, Kaplan-Meier estimates were obtained, along with a stratified log-rank test (as with analysis of the primary efficacy outcome) comparing the 2 treatment arms. Median and corresponding 95% CIs were obtained from the Kaplan-Meier estimates. Cox’s regression was applied to obtain the HR estimate and the corresponding 95% CI. As the treatment paradigm for AL amyloidosis entails a treatment switch when hematologic response is suboptimal or in the event of worsening organ function (which commonly occurs before disease progression and may affect the evaluation of the MOD-PFS), the primary analysis for MOD-PFS employed the IPCW method to adjust estimates of a treatment effect in the presence of subsequent non–cross-resistant anti–plasma cell therapy. Details of statistical analyses for efficacy end points are outlined in Table 7.
Table 7: Outcome Definitions and Statistical Analysis of End Points
End point | Definition or assessment | Analysis methods | Included in hierarchy of statistical testing |
|---|---|---|---|
Primary end point | |||
Hematologic CR rate | The proportion of patients who achieved a hematological CR (assessed by an IRC) according to the consensus guidelines for AL amyloidosis (an iFLC of less than the ULN range with a negative serum and urine immunofixation); if the iFLC was less than the ULN, normalization of the uninvolved FLC and FLC ratio were not required | A stratified Cochran-Mantel-Haenszel test was used to test treatment difference in the proportion of patients who achieved a hematologic CR in the ITT population, at the time of data cut-off (primary analyses: February 14, 2020) Sensitivity analyses:
| Yes (1) |
Secondary end points | |||
Hematologic VGPR-or-better rate | The proportion of patients who achieve a confirmed hematologic CR or VGPR, according to the consensus guidelines for AL amyloidosis, during or after the study treatment | Analyzed similarly as the primary end point hematologic CR rate (based on IRC-assessed response) | No |
Organ response rate | Organ response rate was defined as the proportion of response-evaluable patients who achieved organ response:
| Descriptive statistics Stratified Cochran-Mantel-Haenszel test | No |
MOD-PFS | A composite end point of clinically observable end points defined from randomization to any one of the following events, whichever came first:
| The primary analysis of MOD-PFS is based on IRC assessment and employed the IPCW method to adjust estimates of a treatment effect in the presence of subsequent non–cross-resistant anti–plasma cell therapy Sensitivity analyses:
| Yes (2) |
MOD-EFS | Time to hematologic progression, major organ deterioration, initiation of subsequent non–cross-resistant anti–plasma cell therapy, or death, whichever occurred first | MOD-EFS was evaluated in support of MOD-PFS, as it reflects clinical outcome in accordance with the AL amyloidosis treatment paradigm in which subsequent treatment is initiated in patients with insufficient hematological response in the absence of hematologic progressive disease | No |
OS | Time from the date of randomization to the date of the patient’s death; patients who are lost to follow-up were censored at the time of lost to follow-up; patients who were still alive at the clinical cut-off date for the analysis were censored at the last known alive date | Unstratified log-rank test, unstratified Cox regression model, and Kaplan-Meier analysis | Yes (3) |
Other efficacy end points | |||
Hematologic PFS | The time from the date of randomization to the date of first documentation of hematologic disease progression, according to central laboratory results and judged by international consensus guidelines, or death due to any cause, whichever occurs first; for patients still alive and who had not yet progressed, data were censored at the last disease assessment | Stratified log-rank test, stratified Cox regression model, and Kaplan-Meier analysis | No |
Time to hematologic response | Includes time to hematologic CR, time to VGPR or better, and time to PR-or-better response:
| Descriptive statistics | No |
Time to organ response | The time between the date of randomization and the first efficacy evaluation at which the patient had each organ response | Descriptive statistics | No |
Duration of hematologic response | Includes duration of hematologic CR, duration of VGPR-or-better response, and duration of PR-or-better response:
| Kaplan-Meier analysis | No |
MRD | MRD status was assessed in patients who achieved hematologic CR based on next generation sequencing or similar technologies | Descriptive statistics | No |
AL = light chain; CR = complete response; FLC = free light chain; iFLC = involved free light chain; IPCW = inverse probability of censoring weight; IRC = independent review committee; ITT = intention-to-treat; NT-proBNP = N-terminal pro–B-type natriuretic peptide; MOD-EFS = major organ deterioration event-free survival; MOD-PFS = major organ deterioration progression-free survival; MRD = minimal residual disease; NYHA = New York Heart Association; PR = partial response; ULN = upper limit of normal; VGPR = very good partial response.
Source: Clinical Overview for Darzalex (section 2.5).24
Two interim analyses were conducted. An independent data monitoring committee assessed the results of the interim analyses. The primary end point of hematologic CR and secondary efficacy end points were adjudicated by an IRC. The first interim analysis was a safety analysis that occurred after the first 30 patients completed at least 1 cycle of treatment. The purpose of this analysis was to have a comprehensive evaluation of safety (AEs were assessed). The second interim analysis assessed safety and efficacy and occurred after 180 patients were treated for at least 6 cycles. The purpose of the second interim analysis was to evaluate cumulative interim safety and efficacy. Both futility and efficacy stopping rules were built into this interim analysis. The study would be stopped due to futility if the hematologic CR rate in the DCyBorD arm was the same or worse than the hematologic CR rate in the CyBorD arm. The study would be stopped due to efficacy if the significance level at this interim analysis to establish the superiority of DCyBorD over CyBorD was less than or equal to 0.0001 (2-sided). The protocol-specified primary analysis for hematologic CR was performed after all patients had completed 6 cycles of treatment. The nominal significance level for primary analysis was 0.0499 (2-sided) after adjusting for the efficacy interim analysis. Hierarchical testing between hematologic CR and MOD-PFS was performed at this analysis. If hematologic CR was significant, then MOD-PFS was tested at P = 0.00136 at this interim analysis for this end point using the O’Brien-Fleming spending function.
An updated analysis of efficacy and safety was performed after a median follow-up of 20.3 months.9
Sensitivity Analyses
Pre-planned sensitivity analyses of the primary end point of hematologic CR were performed. This included assessment of hematologic CR by an investigator (confirmed by a subsequent assessment at a subsequent visit) and hematologic response as determined by a computerized algorithm (negative serum and urine immunofixation and involved FLCs less than the ULN). Statistical analyses were performed using an approach similar to that of the main analysis.
Subgroup Analyses
The primary end point of IRC-assessed hematologic CR rate was assessed in pre-planned subgroups, including cardiac stage and t(11;14) translocation, which were subgroups of interest identified in the CADTH systematic review protocol.
Results
Patient Disposition
At the time of data cut-off date (February 14, 2020), 388 patients were randomized to receive treatment with either DCyBorD (n = 195) or CyBorD (n = 193); a total of 193 patients were treated with DCyBorD and 188 were treated with CyBorD. Of these patients, 52 (26.6%) in the DCyBorD arm and 68 (35.2%) in the CyBorD arm discontinued study treatment. The main reasons for discontinuation were death (DCyBorD: 10.3%; CyBorD: 7.4%), and subsequent therapy for AL amyloidosis (DCyBorD: 5.3%; CyBorD: 12.2%). Two patients (1%) in the DCyBorD arm and 11 (5.8%) in the CyBorD arm discontinued study treatment due to hematologic progression or major organ deterioration (Table 8).
Protocol Deviations
All protocol deviations of eligibility criteria and those deviations that could affect patient safety or primary end points were considered major protocol deviations. Overall, 4.4% of patients had major protocol deviations; none of these were considered to affect patient safety or data integrity. Three patients (0.8%) received a disallowed concomitant treatment and 5 (1.3%) entered the study but did not satisfy eligibility criteria. One patient entered but did not satisfy criteria (QT interval by Fridericia’s formula [QTcF] > 500 ms during the screening period) and also received disallowed concomitant treatment (doxycycline for > 14 days within first 6 cycles of therapy). The other protocol deviations included: bortezomib administration after expiration date (no safety issues reported) (n = 2); elevated total bilirubin on cycle 1, day 1 (n = 1); failure to complete hepatitis B virus screening before cycle 1, day 1 (n = 3); failure to meet the randomization time window (n = 1); missing laboratory testing for at least 2 consecutive visits (n = 1); and missing more than 2 disease evaluations (n = 1).
Table 8: Patient Disposition in the ADROMEDA Study
Disposition | DCyBorD | CyBorD |
|---|---|---|
Screened, N | 583 | |
Randomized, N | 195 | 193 |
Received allocated intervention | 193 | 188 |
Did not receive allocated intervention | 2 | 5 |
Patients who discontinued treatment, N (%) | 52 (26.6) | 68 (35.2) |
Reason for discontinuation, N (%) | ||
Adverse events | 8 (4.1) | 8 (4.2) |
Death | 20 (10.3) | 14 (7.4) |
Physician decision | 1 (0.5) | 1 (0.5) |
Patient decision | 3 (1.5) | 7 (3.7) |
Received subsequent therapy for AL amyloidosis | 5 (5.3) | 23 (12.2) |
Progressive disease | 2 (1.0) | 11 (5.8) |
Received autologous stem cell transplant | 12 (6.2) | 3 (1.5) |
Other | 1 (0.5) | 1 (0.5) |
Patients who discontinued studya | 31 (15.9) | 41 (21.2) |
Reason for discontinuation, N (%)b | ||
Death | 27 (13.8) | 27 (14.0) |
Lost to follow-up | 0 | 1 (0.5) |
Withdrawal by patient | 4 (2.1) | 13 (6.7) |
Intention-to-treat analysis, N | 195 | 193 |
Excluded from safety analysis | 2 | 5 |
AL = light chain; CyBorD = cyclophosphamide, bortezomib, and dexamethasone; DCyBorD = daratumumab in combination with cyclophosphamide, bortezomib, and dexamethasone.
aPercentages are based on number of a patients randomized.
bPercentages are based on number of patients treated.
Source: Clinical Overview for Darzalex (section 2.5).24
Exposure to Study Treatments
The median treatment duration was 9.6 months in the DCyBorD arm and 5.3 months in the CyBorD arm (as daratumumab was continued beyond the initial 6 cycles of CyBorD, the treatment duration was expected to be longer in the DCyBorD arm). The median number of treatment cycles received was 11 (range = 1 to 23) for the DCyBorD arm and 6 (range = 1 to 6) for the CyBorD arm. Overall, 381 patients received at least 1 administration of study treatment (DCyBorD: 193; CyBorD: 188). During the first 2 cycles, a similar percentage of patients in both treatment arms received study treatment. From cycle 3 onward, more patients in the CyBorD arm discontinued study treatment compared with patients in the DCyBorD arm; 121 patients (64.4%) in the CyBorD and 159 patients (82.4%) in the DCyBorD arm completed 6 cycles of trial treatment. In the DCyBorD arm, 149 patients (77.2%) continued single-drug daratumumab SC after completing the first 6 treatment cycles. At the time of the primary analysis, 141 of 195 patients (72.3%) in the DCyBorD arm were continuing to receive daratumumab.
The median total doses (exposures) of cyclophosphamide (mg/m2), bortezomib (mg/m2), and dexamethasone (mg) were similar in the 2 treatment arms during cycles 1 and 2 and slightly higher in the DCyBorD arm during cycles 3 through 6, likely due to more patients in the CyBorD arm discontinuing study treatment from cycle 3 onward. The extent of exposure to individual study drugs during the first 6 cycles was similar between the 2 treatment arms. The median total dose of cyclophosphamide ranged from 1,022.9 mg/m2 to 1,077.7 mg/m2 in the CyBorD arm and 1,025.3 mg/m2 to 1,041.6 mg/m2 in the DCyBorD arm across 6 cycles. The protocol-specified dose of cyclophosphamide was 1,200 mg/m2 per cycle (with a maximum weekly dose of 500 mg). The median total dose of bortezomib ranged from 5.1 mg/m2 to 5.2 mg/m2 in the CyBorD arm and was 5.1 mg/m2 in the DCyBorD arm across 6 cycles. The protocol-specified dose of bortezomib was 5.2 mg/m2 per cycle. The median total dose of dexamethasone was 160 mg per cycle for all cycles in both treatment arms. The protocol-specified dose of steroid required per cycle was 160 mg. The median relative dose intensities for cyclophosphamide, bortezomib, and dexamethasone were consistent across the treatment arms (cyclophosphamide: 85.8% versus 86.1% in the DCyBorD and CyBorD arms, respectively; bortezomib: 96.6% versus 97.4% respectively; dexamethasone: 100% in each arm). The median relative dose intensity for daratumumab was 100%. In the DCyBorD and CyBorD treatment arms, dose reductions were carried out for cyclophosphamide (17.6% and 13.8%, respectively), bortezomib (25.9% and 19.7%, respectively), and dexamethasone (27.5% and 27.7%, respectively).
When assessed across all cycles, a higher percentage of patients in the DCyBorD arm (43.5%) had cycle delays compared with the CyBorD arm (21.8%) due to continuation of daratumumab SC beyond cycle 6. The percentage of patients with treatment delays was similar between treatment arms during cycles 1 through 6 (DCyBorD: 25.4%; CyBorD: 21.8%). Reasons for treatment delays included AEs and “other.” The majority of “other” reasons for cycle delays in the DCyBorD arm were administrative. Other reasons included “stem cell collection.” Dose delay and dose skipping of daratumumab SC occurred in 6.7% and 25.9% of patients respectively, in the DCyBorD arm.
Updated analysis: At the updated analyses performed at a median follow-up of 20.3 months, 40% of patients in the DCyBorD arm were still receiving daratumumab monotherapy. The median duration of study treatment was 18.5 months (range = 0.03 to 23.7) in the DCyBorD arm, and 5.3 months (range = 0.03 to 7.3) in the CyBorD arm. The percentages of patients in the DCyBorD and CyBorD treatment arms who received more than 3 cycles of treatment were 86% and 80%, respectively.9
Concomitant Medications
The most commonly used therapeutic classes (≥ 50% of patients) of concomitant medications, as reported until the earlier of 30 days post-treatment or the start of subsequent therapy, were antivirals for systemic use (76.1%), antibacterials for systemic use (71.1%), and drugs for acid-related disorders (52.5%). Therapeutic classes of concomitant medications with at least a 10% difference in use between the treatment arms were antibacterials for systemic use (DCyBorD: 78.8%; CyBorD: 63.3%); macrolides, lincosamides, and streptogramins (19.2% and 5.9%, respectively); drugs for acid-related disorders (57.5% and 47.3%, respectively); and analgesics (49.7% and 36.7%, respectively).
Subsequent Therapy
Patients with suboptimal hematologic response or worsening organ function were permitted to start subsequent non–cross-resistant anti–plasma cell therapy before developing hematologic progression after 3 cycles of treatment. Twenty patients (10.4%) in the DCyBorD arm and 90 patients (47.9%) in the CyBorD arm received subsequent therapy, including cross-resistant as well as non–cross-resistant anti–plasma cell therapy and ASCT. Of those who received subsequent therapy in the CyBorD arm, 48 of the 90 patients (53.3%) received daratumumab intravenously as subsequent therapy. Most patients who initiated subsequent therapy (either for suboptimal response or as consolidation therapy) in the DCyBorD arm (13 of 20 patients [65%]), received ASCT compared with the CyBorD arm (20 of 90 patients [22.2%]).
Efficacy
Only those efficacy outcomes and analyses of subgroups identified in the review protocol are reported below. The primary efficacy analyses are based on a median follow-up of 11.4 months (data cut-off date: February 15, 2020)7,8 and the updated analyses are based on a median follow-up of 20.3 months.9
Hematologic Response
At the primary analysis, 104 patients (53.3%) in the DCyBorD arm and 35 (18.1%) in the CyBorD arm had an IRC-assessed hematologic CR (relative risk ratio = 2.9; 95% CI, 2.1 to 4.1; P < 0.001) (Table 9). Landmark analysis of hematologic CR at 6 months was consistent with hematologic CR; 49.7% versus 14.0% of patients in the DCyBorD versus CyBorD treatment arms, respectively, had hematologic CR at 6 months (relative risk ratio = 3.5; 95% CI, 2.4 to 5.2). The hematologic CR rates at 12 months were 28.2% in the DCyBorD arm and 7.3% in the CyBorD arm. The percentage of patients who had a VGPR or better was 78.5% in the DCyBorD arm and 49.2% in the CyBorD arm (relative risk ratio = 1.6; 95% CI, 1.4 to 1.9). The results of sensitivity analyses using the investigator-assessed hematologic CR and computerized algorithm-derived hematologic CR were consistent with the IRC-assessed hematologic CR (investigator-assessed: 53.3% versus 17.1%, respectively; computerized algorithm: 53.3% versus 16.6%, respectively).
At the updated analysis, the hematologic CR rates were 59.0% in the DCyBorD arm and 19.2% in the CyBorD arm, and the rates of hematologic overall response were 92% (≥ VGPR: 79%) in the DCyBorD arm and 77% (≥ VGPR: 50%) in the CyBorD arm. The rates of hematologic overall response were 91.8% in the DCyBorD arm (≥ VGPR: 78.5%) and 76.7% (≥ VGPR: 49.2%) in the CyBorD arm (Table 9).
Table 9: Summary of Hematologic Responses
Response | DCyBorD (N = 195) | CyBorD (N = 193) |
|---|---|---|
Primary analysis (median follow-up: 11.4 months) | ||
Any response | ||
n (%) | 179 (91.8) | 148 (76.7) |
Complete response | ||
n (%) | 104 (53.3) | 35 (18.1) |
Relative risk ratio (95% CI),7 P value | 2.9 (2.1 to 4.1), < 0.001 | |
Very good partial response or better, n (%) | 153 (78.5) | 95 (49.2) |
Very good partial response, n (%) | 49 (25.1) | 60 (31.1) |
Partial response, n (%) | 26 (13.3) | 53 (27.5) |
No response, n (%) | 8 (4.1) | 38 (19.7) |
Progressive disease, n (%) | 0 | 0 |
Non-evaluable, n (%) | 8 (4.1) | 7 (3.6) |
Updated analysis (median follow-up: 20.3 months) | ||
Any response | ||
n (%) | 179 (92) | 149 (77) |
Complete response | ||
n (%) | 115 (59) | 37 (19) |
Relative risk ratio (95% CI), P value | Not reported | |
Very good partial response or better, n (%) | 154 (79) | 96 (50) |
Very good partial response, n (%) | 39 (20) | 60 (31) |
PR, n (%) | 25 (13) | 50 (26) |
CI = confidence interval; CyBorD = cyclophosphamide, bortezomib, and dexamethasone; DCyBorD = daratumumab in combination with cyclophosphamide, bortezomib, and dexamethasone.
Source: Kastritis et al. (2021),7 Clinical Study Report for Darzalex,8 and Kastritis et al. (2021).9
Subgroup Analyses
Hematologic CR rates across cardiac stages were consistent with the results observed in the overall population of patients. The hematologic CR rate was higher in the DCyBorD arm compared to the CyBorD arm for all cardiac stages. The difference between the hematologic CR rates in the 2 treatment arms increased by Mayo cardiac stage (DCyBorD versus CyBorD: 45% versus 28% for cardiac stage I; 54% versus 20% for stage II, and 58% versus 10% for stage III, respectively). Similarly, the magnitude of hematologic CR rates in patients with a t(11;14) mutation was similar to that observed in the overall population. Patients in the DCyBorD arm had equally high rates of hematologic CR regardless of t(11;14), whereas lower hematologic CR rates were observed for patients with a t(11;14) mutation treated with CyBorD (Table 10).
Table 10: Subgroup Analysis of Hematologic Complete Response Rate
Subgroup | Patients with a response/total number of patients (%) | |
|---|---|---|
DCyBorD (N = 195) | CyBorD (N = 193) | |
Cardiac stage at baseline | ||
I | 21/47 (45) | 12/43 (28) |
II | 41/76 (54) | 16/80 (20) |
IIIA or IIIB | 42/72 (58) | 7/70 (10) |
t(11;14) translocation | ||
Yes | 28/51 (55) | 7/55 (13) |
No | 23/44 (52) | 13/52 (25) |
CyBorD = cyclophosphamide, bortezomib, and dexamethasone; DCyBorD = daratumumab in combination with cyclophosphamide, bortezomib, and dexamethasone.
Source: Clinical Study Report for Darzalex.8
Time to Hematologic Response
Among responders, the median time to hematologic CR was 60 days (range = 8 to 299 days) in the DCyBorD arm and 85 days (range = 14 to 340 days) in the CyBorD arm. Among patients who achieved at least a VGPR, the median time to VGPR or better was 17 days (range = 5 to 336 days) in the DCyBorD arm and 25 days (range = 8 to 171 days) in the CyBorD arm.
Duration of Hematologic Response
At the primary analysis, the median duration of hematological CR had not been reached in either treatment arm (range = 0.85 to 17.5 months for DCyBorD; 0.03 to 18.4 months for CyBorD). Of the 104 patients who achieved hematological CR in the DCyBorD arm, 4 patients died while in hematological CR and no patients relapsed following hematological CR. Of the 35 patients who achieved hematological CR in the CyBorD arm, 2 died while in hematologic CR and 2 relapsed following hematologic CR. Similarly, among patients who achieved a VGPR-or-better or PR-or-better status, the median duration of VGPR or better and duration of PR or better in both treatment arms was not reached, as most responders continued to respond without hematologic progression.
Hematologic Progression-Free Survival
At the time of primary analysis, 17.4% of patients in the DCyBorD arm and 24.9% of patients in the CyBorD arm had hematologic progression based on IRC assessment or had died. At 6 months, the hematologic PFS was similar between the 2 treatment arms (DCyBorD: 86.9%; CyBorD: 85.7%). At 12 months, a higher hematologic PFS rate was observed for patients in the DCyBorD arm compared to patients in the CyBorD arm (81.5% versus 73.3%). The HR for hematologic PFS was 0.63 (95% CI, 0.40 to 0.97; P = 0.036).
Organ Response
Of the 277 patients with baseline cardiac involvement (DCyBorD: 140 [71.8%]; CyBorD: 137 [71.0%]), 235 patients (DCyBorD: 118 [60.5%]; CyBorD: 117 [60.6%]), were evaluated for cardiac response. Of patients who could be evaluated for cardiac response, 41.5% of the patients in the DCyBorD arm and 22.2% of the patients in the CyBorD arm had a cardiac response at 6 months. Among patients who could be evaluated for renal response (DCyBorD 113 [59%]; CyBorD n = 117 [61%]), 53.0% of the patients in the DCyBorD arm and 23.9% of the patients in the CyBorD arm had a renal response at 6 months. In the updated analysis, 12-month organ response rates in the DCyBorD and CyBorD arms were 57% and 28% respectively, for cardiac response, and 57% and 27%, respectively, for renal response (Table 11).
Table 11: Summary of Cardiac and Renal Responses
Response | DCyBorD | CyBorD |
|---|---|---|
Primary analysis (median follow-up: 11.4 months) | ||
Response at 6 months | ||
Cardiac response, n of n evaluable (%) | 49 of 118 (41.5) | 26 of 117 (22.2) |
Renal response, n of n evaluable (%) | 62 of 117 (53.0) | 27 of 113 (23.9) |
Updated analysis (median follow-up: 20.3 months)a | ||
Response at 12 months, % | ||
Cardiac response, n of n evaluable (%) | 67 of 118 (57) | 33 of 117 (28) |
Renal response, n of n evaluable (%) | 67 of 117 (57) | 31 of 113 (27) |
CyBorD = cyclophosphamide, bortezomib, and dexamethasone; DCyBorD = daratumumab in combination with cyclophosphamide, bortezomib, and dexamethasone.
aNumber of patients who could be evaluated.
Source: Clinical Study Report for Darzalex8 and Kastritis et al. (2021).9
Time to Organ Response
For the primary analyses, 59 patients in the DCyBorD arm and 41 patients in the CyBorD arm were considered cardiac responders. The median time to cardiac response based on IRC assessment was 3.0 months in the DCyBorD arm and 3.8 months in the CyBorD arm (without censoring for subsequent non–cross-resistant anti–plasma cell therapy). For renal response, 83 patients in the DCyBorD arm and 45 patients in the CyBorD arm were considered responders. The median times to renal response based on IRC assessment were 1.2 months in the DCyBorD arm and 2.2 months in the CyBorD arm (without censoring subsequent non–cross-resistant anti–plasma cell therapy).
MOD-PFS
The HR for MOD-PFS (DCyBorD versus CyBorD) based on the primary IPCW analysis was 0.580 (95% CI, 0.36 to 0.93). The nominal P value was 0.0211 and did not cross the pre-specified stopping boundary (P = 0.00136) at the interim analysis for MOD-PFS based on approximately 43.5% of the required number of events for the final analysis (Figure 3). The median MOD-PFS was not reached in either treatment arm. The 18-month MOD-PFS rate was estimated at 79.3% in the DCyBorD arm and 59.8% in the CyBorD arm.
Figure 3: Inverse Probability Weighted Kaplan-Meier Plot of MOD-PFS Based on IRC Assessment — ITT Population
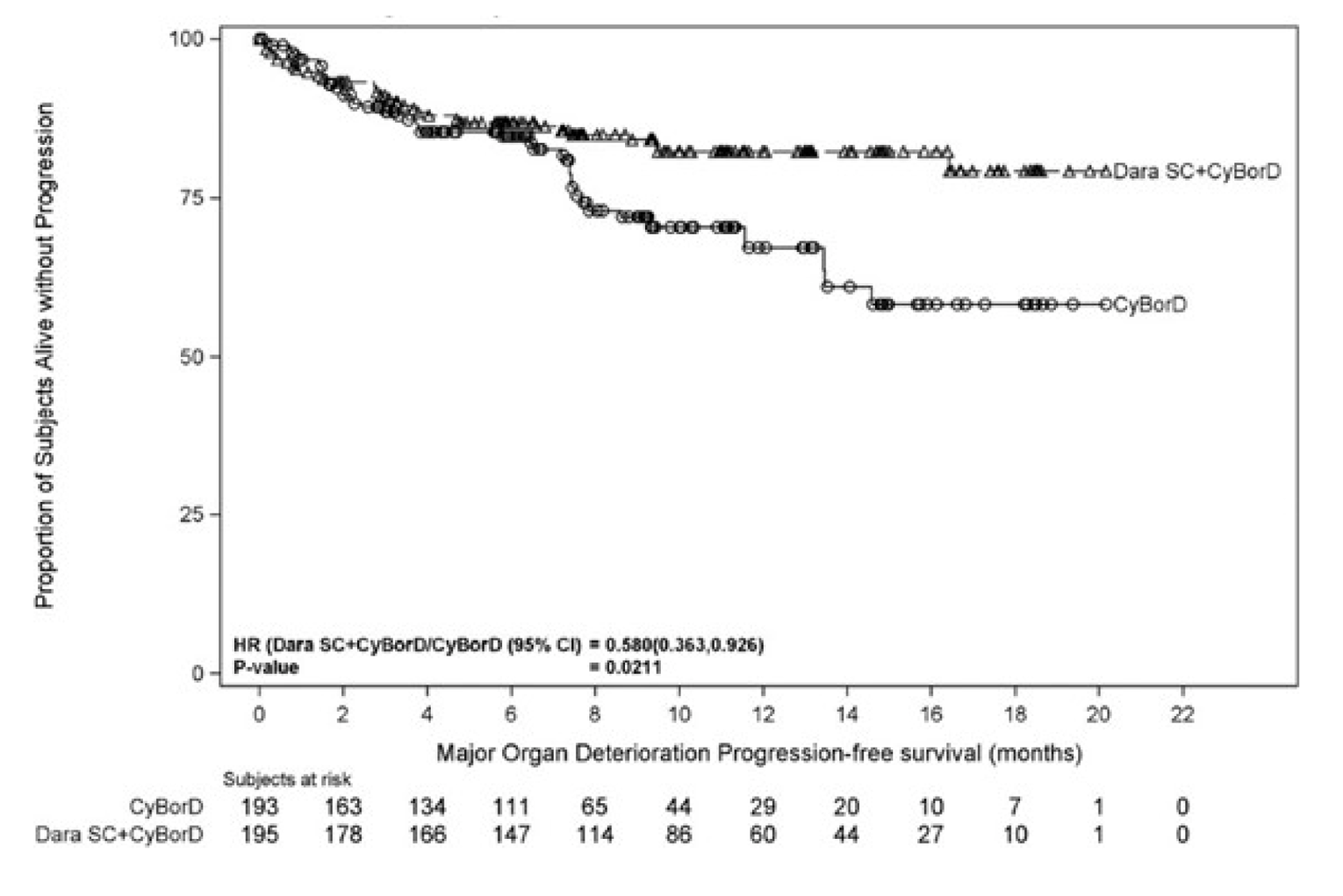
CI = confidence interval; CyBorD = bortezomib, cyclophosphamide, and dexamethasone; dara = daratumumab; IRC = independent review committee; ITT = intention-to-treat; MOD-PFS = major organ deterioration progression-free survival; SC = subcutaneous.
Source: Clinical Study Report for Darzalex.8
MOD-EFS
The median MOD-EFS was not reached in the DCyBorD arm and was 8.8 months in the CyBorD arm. The HR for EFS was 0.39 (95% CI, 0.27 to 0.56).
MOD-OS
Survival free from MOD, hematologic progression, or subsequent treatment was longer in the DCyBorD versus the CyBorD arm; the HR for MOD, hematologic progression, subsequent treatment, or death was 0.39 (95% CI, 0.27 to 0.56).
Overall Survival
At the time of the data cut-off, OS data were not yet sufficiently mature to compare treatment groups. A total of 56 deaths occurred during follow-up, including 27 (13.8%) in the DCyBorD arm and 29 (15.0%) in the CyBorD arm (HR = 0.90; 95% CI 0.53 to 1.53; P = 0.7055). The median OS was not reached in either treatment arm, with estimated 18-month OS rates of 85.6% in the DCyBorD arm and 76.9% in the CyBorD arm (Figure 4).
Figure 4: Weighted Kaplan-Meier Plot of Overall Survival — ITT Population
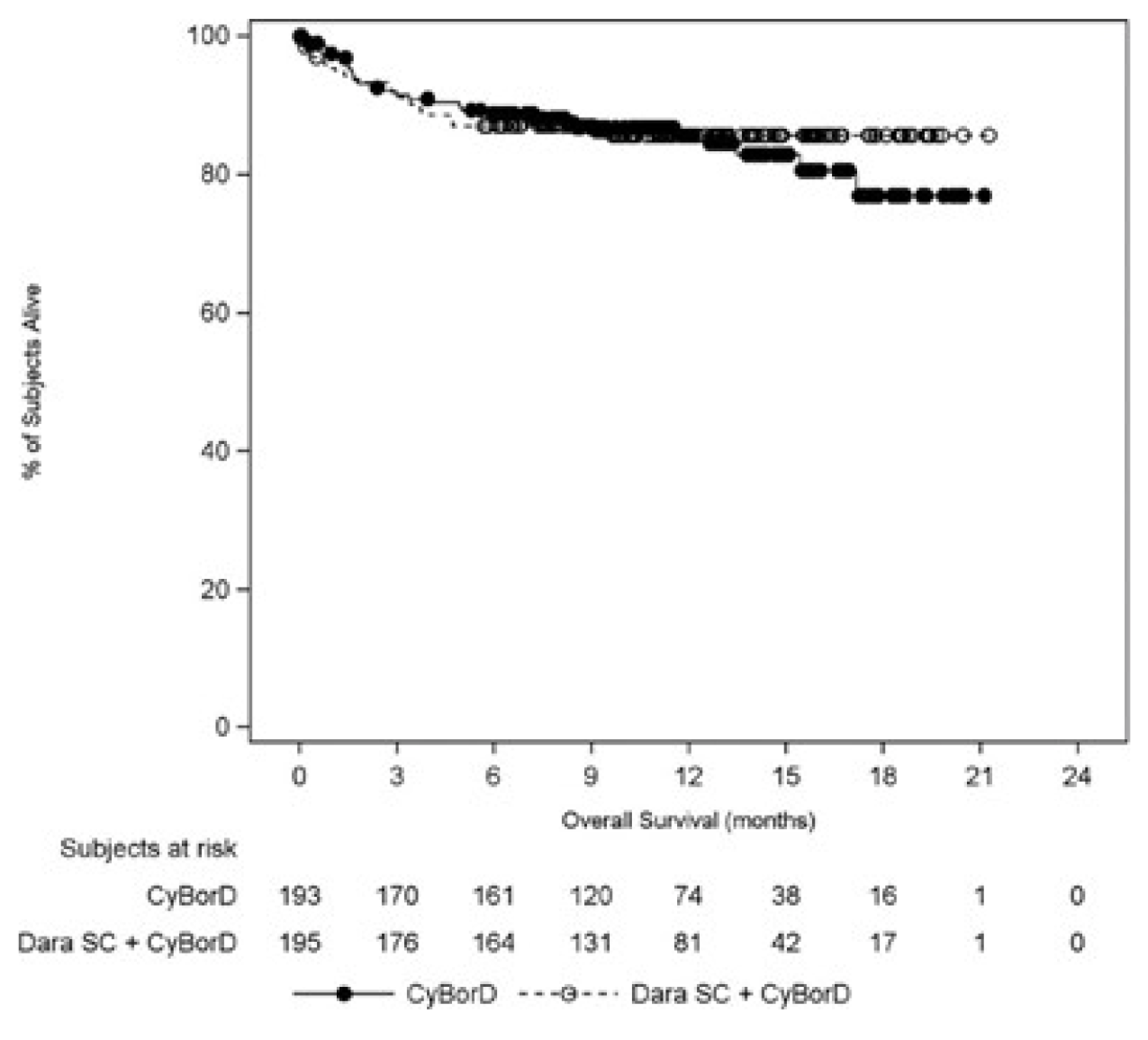
CyBorD = cyclophosphamide, bortezomib, and dexamethasone; dara = daratumumab; ITT = intention-to-treat; SC = subcutaneous.
Source: Clinical Study Report for Darzalex.8
Minimal Residual Disease
Minimal residual disease (MRD) status was evaluated in patients who achieved hematologic CR to explore this end point as a surrogate for hematological PFS and OS, or as a biomarker for relapse. Overall, 84 patients (67 in the DCyBorD arm and 17 in the CyBorD arm) had bone marrow MRD samples obtained. The MRD negativity rate was 3-fold higher in the DCyBorD arm compared to the CyBorD arm; in the ITT population, the MRD negativity rate was 6.7% in the DCyBorD arm and 2.1% in the CyBorD arm at the 10-5 threshold (Table 12).
Table 12: Summary of MRD Negativity Rate in Bone Marrow — ITT Population
MRD negativity rate [threshold] | DCyBorD (N = 195) | CyBorD (N = 193) |
|---|---|---|
MRD negativity rate [10-4], n (%) | 34 (17.4) | 7 (3.6) |
95% CI of MRD negativity rate | 12.4 to 23.5 | 1.5 to 7.3 |
MRD negativity rate [10-5], n (%) | 13 (6.7) | 4 (2.1) |
95% CI of MRD negativity rate | 3.6 to 11.1 | 0.6 to 5.2 |
MRD negativity rate [10-6], n (%) | 5 (2.6) | 1 (0.5) |
95% CI of MRD negativity rate | 0.8 to 5.9 | < 0.1 to 2.9 |
CI = confidence interval; CyBorD = bortezomib, cyclophosphamide, and dexamethasone; DCyBorD = daratumumab in combination with cyclophosphamide, bortezomib, and dexamethasone; MRD = minimal residual disease.
Source: Clinical Study Report for Darzalex.8
Health-Related Quality of Life
EORTC QLQ-C30
The median time to improvement for global health status was 7.8 months in the DCyBorD arm and 16.7 months in the CyBorD arm (HR = 1.53; 95% CI, 1.10 to 2.13). Scores for EORTC QLQ-30 global health status showed continued improvement in the DCyBorD arm after 6 cycles when patients were receiving daratumumab monotherapy (Figure 5). There was no difference in the median time to worsening between treatment arms. The mean changes from baseline in global health status were higher in the DCyBorD arm, with the greatest difference between the 2 arms occurring at week 12 (least squares [LS] mean change of −1.57 [95% CI, −4.65 to 1.50] for DCyBorD and −6.06 [95% CI, −9.14 to −2.98] for DCyBorD; P = 0.0314) and week 16 (LS mean change of −2.56 [95% CI, −5.68 to 0.55] for DCyBorD and −7.24 [95% CI, −10.38 to −4.11] for CyBorD; P = 0.0274) (Figure 6).
Figure 5: Mean EORTC QLQ-C30 Global Health Status Scores Over Time — ITT Population
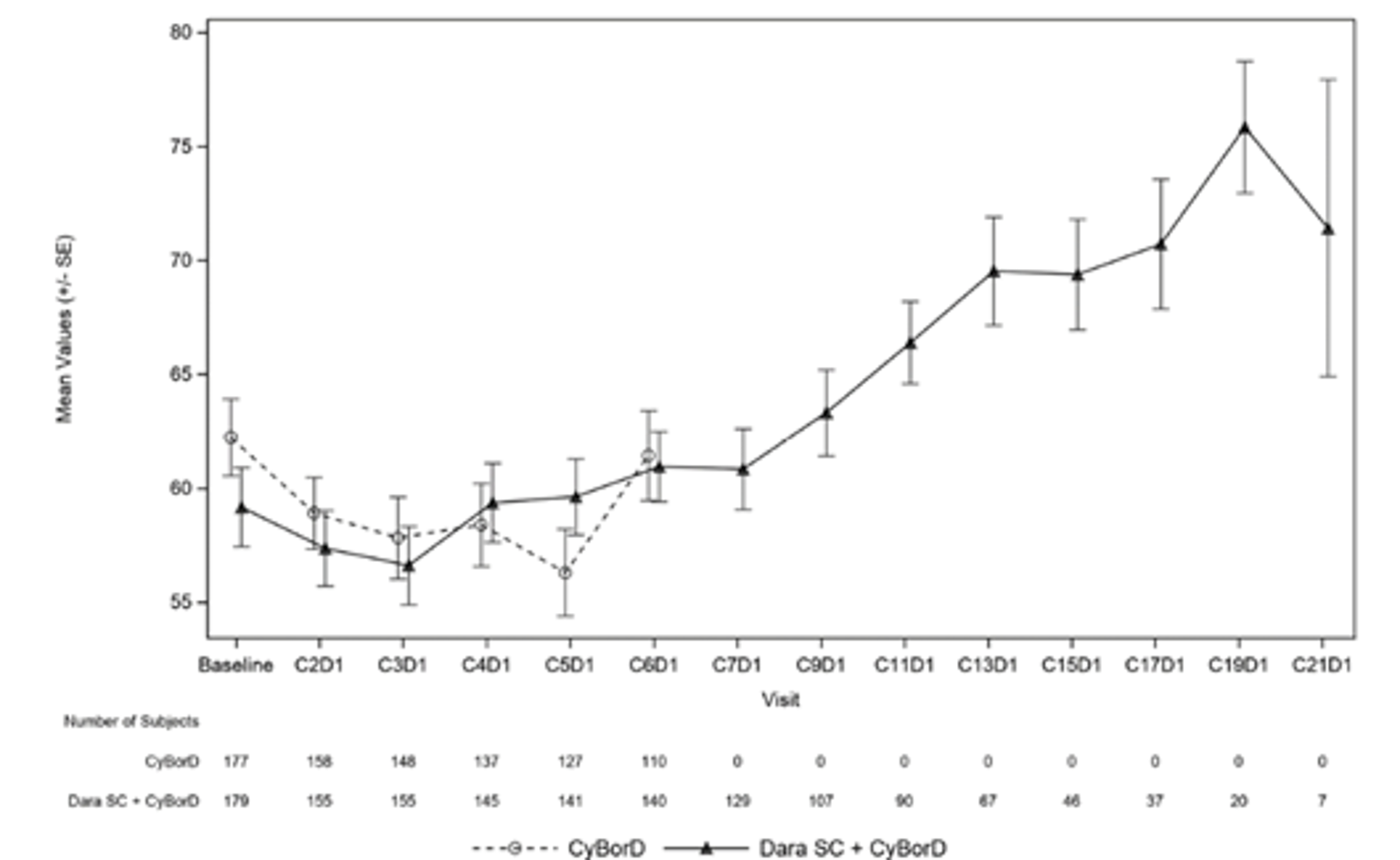
CyBorD = cyclophosphamide, bortezomib, and dexamethasone; dara = daratumumab; EORTC QLQ-C30 = European Organisation for Research and Treatment Cancer Quality of Life Questionnaire Core 30 version 3.0; ITT = intention-to-treat; SC = subcutaneous; SE = standard error.
Source: Clinical Study Report for Darzalex.8
Figure 6: Mean Change in EORTC QLQ-C30 Global Health Status Scores Over Time — ITT Population
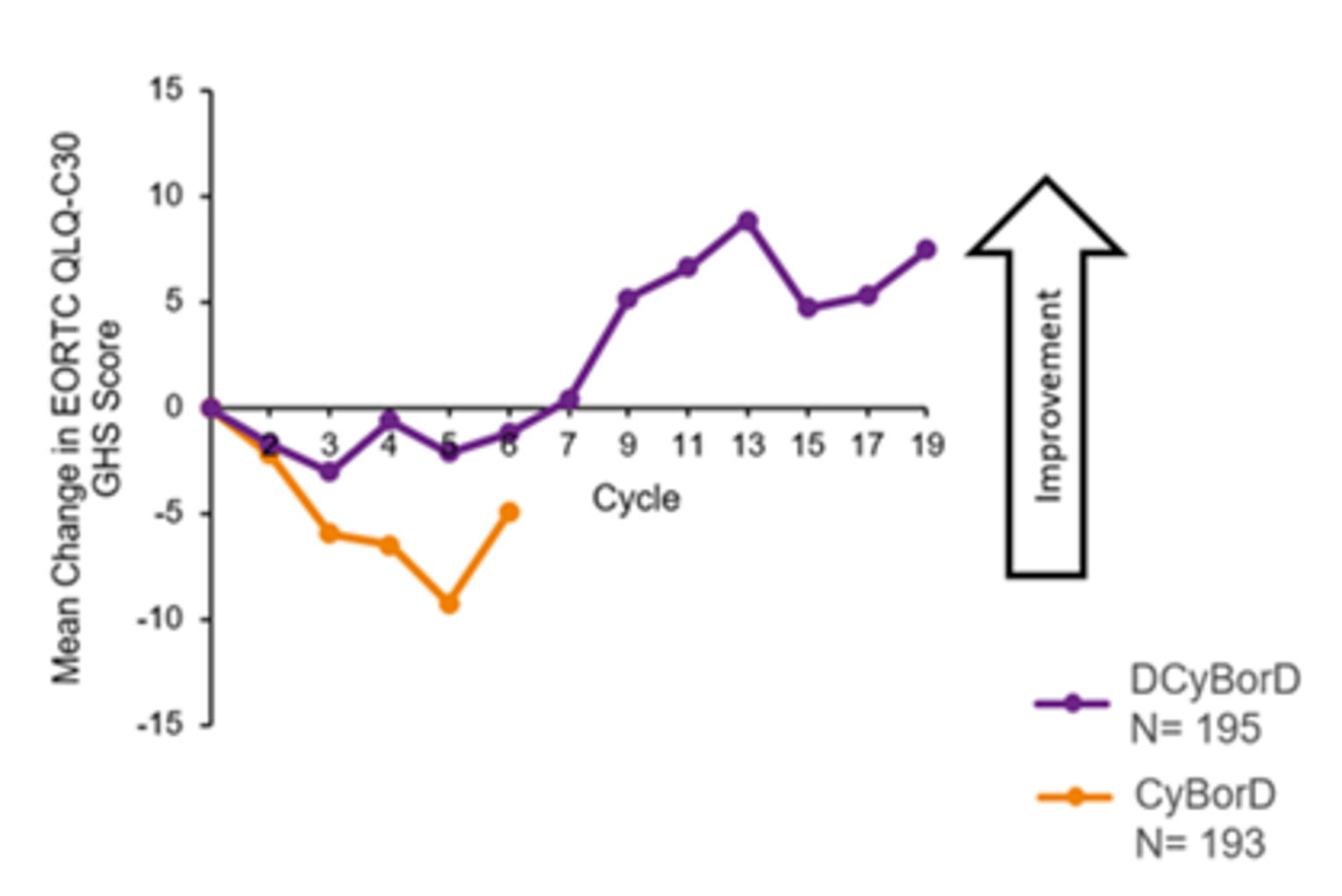
CyBorD = cyclophosphamide, bortezomib, and dexamethasone; DCyBorD = daratumumab in combination with cyclophosphamide, bortezomib, and dexamethasone; EORTC QLQ-C30 = European Organisation for Research and Treatment Cancer Quality of Life Questionnaire Core 30 version 3.0; GHS = global health status; ITT = intention-to-treat.
Source: Clinical Study Report for Darzalex.8
EQ-5D-5L
For median time to improvement, the utility value median was 9.26 months for the DCyBorD arm and 11.79 months for the CyBorD arm, and the visual analogue scale (VAS) median was 10.05 months for the DCyBorD arm and not evaluable in the CyBorD arm at the time of the primary analysis. Median time to worsening in utility value was 4.90 months for the DCyBorD arm and 4.27 months for the CyBorD arm, and the VAS median was 4.14 months for the DCyBorD arm and 3.38 months for the CyBorD arm. There was a statistically significant difference in the mean change from baseline at week 16 for the EQ-5D-5L utility value and the VAS between the 2 treatment arms (utility value LS mean change of 0.00 [95% CI, −0.032 to 0.033] for DCyBorD and −0.056 [95% CI, −0.089 to −0.023] for CyBorD; P = 0.0104 and VAS LS mean change of −1.46 [95% CI to −4.34 to 1.42] for DCyBorD and −6.07 [95% CI, −8.97 to −3.17] for CyBorD; P = 0.0178). For patients in the DCyBorD arm who continued treatment with daratumumab monotherapy after cycle 6, overall HRQoL continued to improve after cycle 6 (Figure 7).
SF-36v2
There was no difference in the time to improvement and time to worsening of patients’ mental health as assessed by the MCS of the SF-36v2. The median time to improvement in the MCS was 7.4 months in the DCyBorD arm and 7.5 months in the CyBorD arm. The median time to worsening was 3.8 and 4.6 months for the DCyBorD and CyBorD arms, respectively. Patients in the DCyBorD arm reported no considerable changes from baseline in their mental health during the first 6 cycles of treatment, and patients in the CyBorD arm reported a worsening over time. The largest numeric difference between the 2 treatment arms occurred at week 16 (LS mean change of −0.11 [95% CI, −1.73 to 1.52] for DCyBorD and −2.95 [95% CI, −4.59 to −1.31] for CyBorD; P = 0.0101). After cycle 6, MCS continued to improve in the DCyBorD arm for patients receiving daratumumab monotherapy (Figure 8).
Figure 7: Mean EQ-5D-5L VAS Scores Over Time — ITT Population
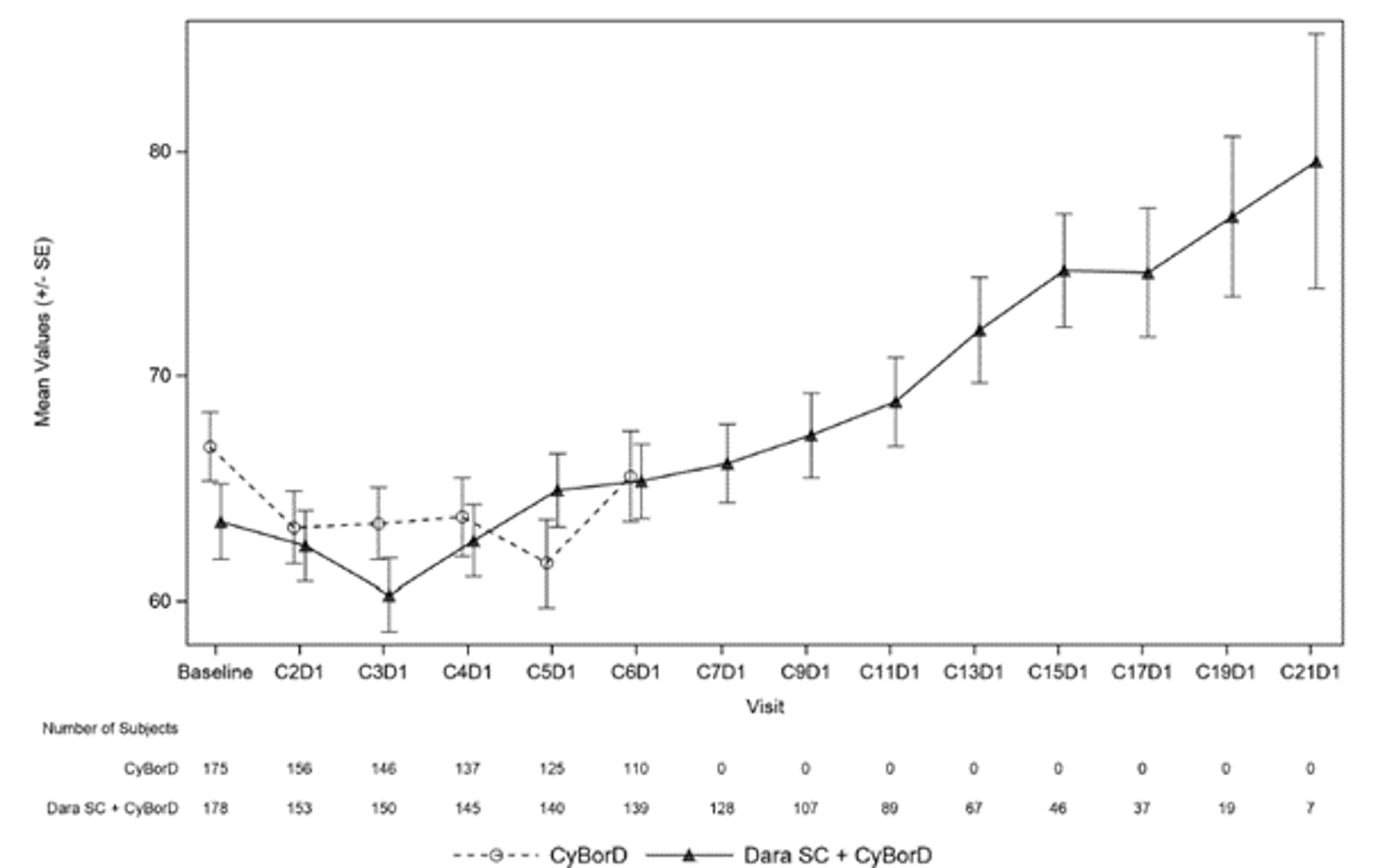
C = cycle; CyBorD = cyclophosphamide, bortezomib, and dexamethasone; D = day; dara = daratumumab; ITT = intention-to-treat; SC = subcutaneous; SE = standard error; VAS = visual analogue scale.
Source: Clinical Study Report for Darzalex.8
Figure 8: Mean SF-36v2 Mental Component Summary Scores Over Time — ITT Population
C = cycle; CyBorD = cyclophosphamide, bortezomib, and dexamethasone; D = day; dara = daratumumab; ITT = intention-to-treat; MCS = mental component summary; SE = standard error; SF-36v2 = Short Form (36) Health Survey version 2.
Source: Clinical Study Report for Darzalex.8
Health Care Resource Utilization
Medical resource utilization was reported for |||| and |||| of patients in the DCyBorD and CyBorD arms, respectively. Use ||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| The most common indication for use of medical services was ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| (Table 13). However, the ability to directly compare health care resource utilization between the 2 treatment arms is limited due to the difference in treatment duration by study arm ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.
Table 13: Medical Resource Utilization — Safety Analysis Set [Redacted]
|||||||||||||||||||||||||||||| | |||||||||||||||||||||||||||||| | |||||||||||||||||||||||||||||| |
|---|---|---|
|||||||||||||||||||||||||||||| | |||||||||||||||||||||||||||||| | |||||||||||||||||||||||||||||| |
|||||||||||||||||||||||||||||| | |||||||||||||||||||||||||||||| | |||||||||||||||||||||||||||||| |
Note: Table 13 has been redacted.
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Source: Additional information provided by the sponsor.
Harms
Adverse Events
Nearly all patients experienced at least 1 TEAE (DCyBorD: 97.9%; CyBorD:98.4%). In both treatment arms, TEAEs led to discontinuation of study treatment in approximately 4% of patients (Table 14). The incidence of grade 3 or grade 4 TEAEs overall was similar between treatment arms (DCyBorD: 58.5%; CyBorD: 57.4%). The most commonly reported (> 5% in either treatment arm) grade 3 or grade 4 TEAEs, all of which except syncope were reported at a least a 2% higher incidence in the DCyBorD arm. No grade 3 or higher treatment-emergent infusion-related reactions or injection-site reactions related to daratumumab SC were reported. The incidence of grade 3 or grade 4 TEAEs was similar between treatment arms during cycle 1 and cycle 2 (DCyBorD: 34.2%, CyBorD: 31.9%) and cycle 3 to cycle 6 (43.5% and 44.2%, respectively). In the DCyBorD arm, 18.1% of patients had grade 3 or 4 TEAEs from cycle 7 onward. The most commonly reported (≥ 2%) grade 3 or grade 4 TEAEs from cycle 7 onward were in the system organ class (SOC) of blood and lymphatic system disorders (4.7%); infections and infestations (4.0%); cardiac disorders (2.7%); and respiratory, thoracic, and mediastinal disorders (2.7%).
A higher percentage of patients (43.0%) in the DCyBorD arm reported at least 1 SAE (i.e., a serious TEAE) compared with the CyBorD arm (36.2%). The most commonly reported (≥ 5% in either treatment arm) SAEs were pneumonia (DCyBorD: 7.3%, CyBorD: 4.8%) and cardiac failure (DCyBorD: 6.2%, CyBorD: 4.8%). The incidence of the following was at least 2% higher in the DCyBorD arm: pneumonia (DCyBorD: 7.3%; CyBorD: 4.8%), sepsis (3.1% and 0%, respectively), and cardiac arrest (3.6% and 1.6%, respectively). Fluid overload was reported with at least 2% higher incidence in the CyBorD arm (DCyBorD: 0.5%; CyBorD: 2.7%). Overall, the incidence of serious TEAEs in the renal (DCyBorD: 3.6%; CyBorD: 2.7%), cardiac (15.5% and 13.3%, respectively), and hepatic (0.5% and 0%, respectively) SOCs was similar (< 5% difference) between treatment arms.
Table 14: Summary of Treatment-Emergent Adverse Events — Safety Population
Treatment-emergent adverse events | DCyBorD (N = 193) n (%) | CyBorD (N = 188) n (%) |
|---|---|---|
Any TEAE | 189 (97.9) | 185 (98.4) |
Maximum toxicity grade | ||
Grade 1 | 8 (4.1) | 10 (5.3) |
Grade 2 | 62 (32.1) | 61 (32.4) |
Grade 3 | 79 (40.9) | 83 (44.1) |
Grade 4 | 18 (9.3) | 16 (8.5) |
Grade 5 | 22 (11.4) | 15 (8.0) |
Any serious TEAE | 83 (43.0) | 68 (36.2) |
TEAE leading to discontinuation of study treatment | 8 (4.1) | 8 (4.3) |
CyBorD = cyclophosphamide, bortezomib, and dexamethasone; DCyBorD = daratumumab in combination with cyclophosphamide, bortezomib, and dexamethasone; TEAE = treatment-emergent adverse event.
Source: Clinical Study Report for Darzalex.8
The following events were reported at an incidence rate of 25% or higher in each treatment arm: peripheral edema (DCyBorD: 35.8%; CyBorD: 36.2%), diarrhea (DCyBorD: 35.8%; CyBorD: 30.3%), constipation (DCyBorD: 34.2%; CyBorD: 28.7%), peripheral sensory neuropathy (DCyBorD: 31.1%; CyBorD: 19.7%), fatigue (DCyBorD: 26.9%; CyBorD: 28.2%), nausea (DCyBorD: 26.9%; CyBorD: 27.7%), upper respiratory tract infection (DCyBorD: 25.9%; CyBorD: 11.2%), and insomnia (DCyBorD: 23.8%; CyBorD: 25.0%). The incidence rates of upper respiratory tract infection, peripheral sensory neuropathy, diarrhea, and constipation were at least 5% higher in the DCyBorD arm (Table 15).
Table 15: Most Commonly Reported (> 10%) Treatment-Emergent Adverse Events by Preferred Term — Safety Population
Preferred term | DCyBorD (N = 193) n (%) | CyBorD (N = 188) n (%) |
|---|---|---|
Patients with 1 or more TEAEs, n (%) | 189 (97.9) | 185 (98.4) |
Diarrhea | 69 (35.8) | 57 (30.3) |
Peripheral edema | 69 (35.8) | 68 (36.2) |
Constipation | 66 (34.2) | 54 (28.7) |
Peripheral sensory neuropathy | 60 (31.1) | 37 (19.7) |
Fatigue | 52 (26.9) | 53 (28.2) |
Nausea | 52 (26.9) | 52 (27.7) |
Upper respiratory tract infection | 50 (25.9) | 21 (11.2) |
Anemia | 47 (24.4) | 44 (23.4) |
Insomnia | 46 (23.8) | 47 (25.0) |
Dyspnea | 44 (22.8) | 32 (17.0) |
Lymphopenia | 36 (18.7) | 28 (14.9) |
Thrombocytopenia | 33 (17.1) | 22 (11.7) |
Cough | 32 (16.6) | 19 (10.1) |
Asthenia | 31 (16.1) | 20 (10.6) |
Dizziness | 29 (15.0) | 26 (13.8) |
Hypotension | 27 (14.0) | 21 (11.2) |
Vomiting | 26 (13.5) | 21 (11.2) |
Headache | 25 (13.0) | 18 (9.6) |
Pyrexia | 25 (13.0) | 16 (8.5) |
Hypokalemia | 24 (12.4) | 28 (14.9) |
Back pain | 23 (11.9) | 11 (5.9) |
Neutropenia | 21 (10.9) | 12 (6.4) |
Pneumonia | 21 (10.9) | 12 (6.4) |
Arthralgia | 20 (10.4) | 9 (4.8) |
Decreased appetite | 19 (9.8) | 23 (12.2) |
Injection-site erythema | 18 (9.3) | 21 (11.2) |
CyBorD = cyclophosphamide, bortezomib, and dexamethasone; DCyBorD = daratumumab in combination with cyclophosphamide, bortezomib, and dexamethasone; TEAE = treatment-emergent adverse event.
Source: Clinical Study Report for Darzalex.8
A higher percentage of patients in the DCyBorD arm (43.0%) reported at least 1 SAE (i.e., a serious TEAE) compared with the CyBorD arm (36.2%). The most commonly reported (≥ 5% in either treatment arm) SAEs were pneumonia (DCyBorD: 7.3%, CyBorD: 4.8%) and cardiac failure (DCyBorD: 6.2%, CyBorD: 4.8%).
Discontinuation Due to Adverse Events
The incidence of any-grade (DCyBorD: 4.1%; CyBorD: 4.3%) and grade 3 or grade 4 (DCyBorD: 3.1%; CyBorD: 2.7%) TEAEs leading to discontinuation of all study treatment was low and similar between the 2 treatment arms. Most of these events were grade 3 or grade 4. No preferred term led to treatment discontinuation in 3 or more patients in either treatment arm. There was a similar discontinuation rate of individual CyBorD components between treatment arms. Eight of the 9 patients in the DCyBorD arm who discontinued daratumumab due to TEAE did so during cycle 1 through cycle 6.
Deaths
At the time of primary analysis, 27 patients (14.0%) in the DCyBorD arm and 28 patients (14.9%) in the CyBorD arm died (Table 16). One patient in the CyBorD arm died before receiving any treatment. A higher proportion of patients in the DCyBorD arm reportedly died due to an AE (11.9%) compared with the CyBorD arm (7.4%), and more patients in the CyBorD arm reportedly died due to progressive disease (1.0% and 4.8%, respectively) and “other” causes (1.0 and 2.7%, respectively). Of the 2 deaths reported as “other” in the DCyBorD arm, 1 was due to an unknown cause, and 1 was due to complications of amyloidosis-macroglossia and increased risk of aspiration. Of the 5 deaths reported as “other” in the CyBorD arm, 1 was due to an unknown cause, 1 was due to pneumonia, 1 was due to aggravation of underlying AL amyloidosis (heart failure), 1 was a consequence of AL amyloidosis (the patient had cardiac involvement at baseline), and 1 was likely caused by septic shock with a respiratory origin.
Due to the design of the trial (i.e., longer treatment duration in the DCyBorD arm) and higher rates of early treatment discontinuation in the CyBorD arm, categorization of certain death events differed between treatment arms. For patients in the CyBorD arm who died more than 30 days after the last treatment dose, fatal events were not categorized as grade 5 TEAEs, but rather as disease progression or other. The incidence of all deaths (DCyBorD: 6.7%; CyBorD: 6.9%) and deaths due to AEs (6.2% and 6.4%, respectively) was similar between treatment arms within 60 days of the first study treatment dose, suggesting that the difference in treatment duration affected the reported differences between treatment arms. Deaths due to AEs within 30 days of the last study treatment were reported for 10.4% of patients in the DCyBorD arm and 7.4% of patients in the CyBorD arm (Table 16).
The most common AEs (≥ 2% in either treatment arm) leading to death were cardiac disorders (cardiac arrest [DCyBorD: 3.1%; CyBorD: 1.6%], sudden death [3.1% and 1.6%, respectively], and cardiac failure [2.6% and 0.5%, respectively]). All patients who died due to cardiac disorders had cardiac involvement at baseline (DCyBorD: 14 or 14; CyBorD: 7 of 7). The majority of all deaths due to TEAEs across all SOCs (21 of 22 patients in the DCyBorD arm and 15 of 15 patients in the CyBorD arm) occurred in patients with cardiac involvement at baseline. The 1 patient treated with DCyBorD who died without having baseline cardiac involvement was reported as having baseline renal involvement (stage III) and died of grade 5 sepsis during cycle 1. No patients who were Mayo cardiac stage I at baseline died due to a TEAE. In both treatment arms, a higher proportion of patients who died were stage III at baseline (DCyBorD: 20.8%; CyBorD: 16.4%) compared with those who were stage II at baseline (9.3% and 5.1%, respectively). These data are consistent with OS by cardiac stage at baseline. Overall, deaths were primarily due to AL amyloidosis–related cardiomyopathy, reported either as TEAEs or disease progression, in both treatment arms.
Table 16: Summary of Death and Cause of Death — Safety Population
Deaths | DCyBorD (N = 193) n (%) | CyBorD (N = 188) n (%) | Total (N = 381) |
|---|---|---|---|
Patients who died during study | |||
n (%) | 27 (14.0) | 28 (14.9) | 55 (14.4) |
Primary cause of death | |||
Adverse event | 23 (11.9) | 14 (7.4) | 37 (9.7) |
At least 1 relateda | 6 (3.1) | 2 (1.1) | 8 (2.1) |
Adverse event(s) unrelated | 17 (8.8) | 12 (6.4) | 29 (7.6) |
Progressive disease | 2 (1.0) | 9 (4.8) | 11 (2.9) |
Other | 2 (1.0) | 5 (2.7) | 7 (1.8) |
Patients who died within 30 days of last study treatment dose | |||
n (%) | 21 (10.9) | 17 (9.0) | 38 (10.0) |
Primary cause of death | |||
Adverse event | 20 (10.4%) | 14 (7.4%) | 34 (8.9%) |
At least 1 relateda | 6 (3.1%) | 2 (1.1%) | 8 (2.1%) |
Adverse event(s) unrelated | 14 (7.3%) | 12 (6.4%) | 26 (6.8%) |
Progressive disease | 1 (0.5%) | 3 (1.6%) | 4 (1.0%) |
Other | 0 | 0 | 0 |
Patients who died after 30 days of last study treatment dose | |||
n (%) | 13 (6.7) | 13 (6.9) | 26 (6.8) |
Primary cause of death | |||
Adverse event | 12 (6.2%) | 12 (6.4%) | 24 (6.3%) |
At least 1 relateda | 5 (2.6%) | 2 (1.1%) | 7 (1.8%) |
Adverse event(s) unrelated | 7 (3.6%) | 10 (5.3%) | 17 (4.5%) |
Progressive disease | 1 (0.5%) | 1 (0.5%) | 2 (0.5%) |
Other | 0 | 0 | 0 |
CyBorD = cyclophosphamide, bortezomib, and dexamethasone; DCyBorD = daratumumab in combination with cyclophosphamide, bortezomib, and dexamethasone.
aIncludes adverse events that were related to at least 1 of the 4 components of study treatment: cyclophosphamide, bortezomib, dexamethasone and daratumumab.
Source: Clinical Study Report for Darzalex.8
Notable Harms
Infusion-Related Reactions
Systemic reactions to daratumumab administration occurred in 14 patients (7.3%). All reactions were of grade 1 or grade 2, manageable, and did not lead to treatment discontinuation. The majority of patients (86%) had a reaction at the first daratumumab administration, with a median time to onset of 1.3 hours (range = 0.2 to 7.3). Local injection-site reactions related to daratumumab occurred in 21 patients (10.9%), all of which were grade 1 or grade 2.
Neutropenia
Across all cycles, the incidence of any grade of neutropenia (DCyBorD: 10.9%; CyBorD: 6.4%) and grade 3 or grade 4 neutropenia (DCyBorD: 5.2%; CyBorD: 2.7%) was higher in the DCyBorD arm. Serious neutropenia was reported in 1 patient in the DCyBorD arm and no patients in the CyBorD arm. In the DCyBorD arm, the incidence of any grade of neutropenia (cycle 1 and cycle 2: 6.7%; cycle 3 through cycle 6: 7.9%; cycle 7 and onward: 2.7%) and grade 3 or grade 4 neutropenia (cycle 1 and cycle 2: 2.6%; cycle 3 through cycle 6: 2.3%; cycle 7 and onward: 1.3%) decreased from cycle 7 onward. Neutropenia was not reported as leading to treatment discontinuation or death in any patient.
Infections
The incidence of any grade of infection (DCyBorD: 65.8%; CyBorD: 53.7%), grade 3 or grade 4 infection (DCyBorD: 16.6%; CyBorD: 10.1%), and serious infection (DCyBorD:16.1%; CyBorD: 8.5%) was higher in the DCyBorD arm. The most commonly reported (> 10% in either treatment arm) any-grade infections were upper respiratory tract infection (DCyBorD: 25.9%; CyBorD: 11.2%) and pneumonia (10.9% and 6.4%, respectively). The most commonly reported grade 3 or grade 4 event in either treatment arm was pneumonia (DCyBorD 7.8%; CyBorD: 4.3%). Infections were generally manageable, and the treatment discontinuation rate was low in both treatment arms (DCyBorD: 1.6%; CyBorD: 0.5%). Two patients in the DCyBorD arm (cases of sepsis) and 1 patient in the CyBorD arm (a case of septic shock) experienced fatal infections. Fatal sepsis was considered related to daratumumab SC and the components of the CyBorD regimen for 1 of the 2 patients in the DCyBorD arm. Both patients who died of sepsis in this arm had organ involvement (cardiac or renal) at baseline. The median time to first infection was 9.7 weeks in the DCyBorD arm and 8.1 weeks in the CyBorD arm. The incidence of any grade of infection was similar between treatment arms during cycle 1 and cycle 2 (DCyBorD: 29.5%, CyBorD: 26.6%) and cycle 3 through cycle 6 (DCyBorD: 50.3%; CyBorD: 45.4%). A similar trend was observed for grade 3 or 4 infections in cycles 1 and 2 (DCyBorD: 4.7%; CyBorD: 3.7%) and cycle 3 through cycle 6 (DCyBorD: 11.3% and CyBorD: 7.4%). The incidence of both any grade of infection (39.6%) and grade 3 or grade 4 infections (4.0%) in the DCyBorD arm was lower from cycle 7 onward compared with cycle 3 through cycle 6.
The incidences of any-grade opportunistic infections were 11.9% in the DCyBorD arm and 8.5% in the CyBorD arm. Grade 3 or grade 4 TEAEs of opportunistic infections were low in both treatment arms (DCyBorD: 1.6%; CyBorD: 1.1%).
Cardiac Toxicity
The incidence of any grade of cardiac disorders was higher in the DCyBorD arm (32.6%) compared with the CyBorD arm (21.8%). The most commonly reported (≥ 5% in either treatment arm) cardiac TEAEs included cardiac failure or cardiac failure congestive combined (DCyBorD: 8.8%; CyBorD: 7.4%), atrial fibrillation (DCyBorD: 5.7%; CyBorD: 2.1%), and palpitations (DCyBorD: 5.7%; CyBorD: 3.2%); a difference in the incidence of 2% or greater between treatment arms was observed for each of these preferred terms. The incidence of any grade of TEAEs in the SOC of cardiac disorders increased with the severity of the baseline cardiac stage in both treatment arms (DCyBorD: 6.5% stage I, 37.3% stage II, and 44.4% stage III; CyBorD: 4.8% stage I, 17.7% stage II, and 37.3% stage III). This trend was also observed for grade 3 or grade 4 cardiac TEAEs (DCyBorD: 0% stage I, 12.0% stage II, 18.1% stage III; CyBorD: 2.4% stage I, 6.3% stage II, and 17.9% stage III).
The incidence of any grade of cardiac events was similar between treatment arms during cycle 1 and cycle 2 (DCyBorD: 17.1%, CyBorD: 12.8%) and cycle 3 through cycle 6 (DCyBorD: 17.5%; CyBorD: 14.7%). A similar trend was observed for grade 3 or 4 cardiac events in cycle 1 and cycle 2 (DCyBorD: 5.7%; CyBorD: 3.7%), and cycle 3 through cycle 6 (DCyBorD: 6.2%; CyBorD: 7.4%). The incidence of grade 3 or grade 4 cardiac events (2.7%) and any grade of cardiac events (8.1%) in the DCyBorD arm was less from cycle 7 onward compared with cycle 3 through cycle 6.
All patients in the DCyBorD arm who experienced serious (30 of 30) or fatal (14 of 14) cardiac TEAEs had amyloidosis–related cardiomyopathy at baseline. A similar trend was observed for serious (24 of 25) and fatal (7 of 7) cardiac TEAEs in the CyBorD arm. A similar proportion of patients died of any cause in both treatment arms (DCyBorD:14.0%; CyBorD: 14.9%). A case-level review confirmed that most deaths in both treatment arms were primarily due to AL amyloidosis–related cardiomyopathy and were either reported as TEAEs in the DCyBorD arm or as disease progression or other in the CyBorD arm.
Renal Toxicity
The incidence of any-grade (DCyBorD: 21.2%; CyBorD: 18.1%) and grade 3 or grade 4 (DCyBorD: 5.7%; CyBorD: 6.4%) renal and urinary disorders was similar between treatment arms. The most commonly reported (≥ 2% in either treatment arm) grade 3 or grade 4 renal and urinary disorders were acute kidney injury (DCyBorD: 2.1%; CyBorD: 1.6%), chronic kidney disease (DCyBorD: 2.1%; CyBorD: 1.1%), and renal impairment (DCyBorD: 0%; CyBorD: 2.1%).
Critical Appraisal
Internal Validity
Given that AL amyloidosis is a rare disease, the sample size of the trial was acceptable. Statistical power calculations were reported, and the target sample size (n = 360) was achieved. Randomization was stratified and stratification was based on relevant prognostic factors that included cardiac stage, availability of transplantation in the local country, and renal function. Baseline demographic and disease characteristics were generally balanced between the 2 treatment arms.
The ANDROMEDA trial was an open-label study; patients and investigators were not blinded to study assignment, although patient blinding would not have been possible given the differences in the 2 study treatment regimens. Nonetheless, sources of bias that may result from lack of blinding of patients and investigators to assigned study treatments cannot be ruled out. For example, patient’s knowledge of their assigned treatment may have affected some safety end points, and HRQoL in particular, and different concomitant supportive care may have been offered to patients in the 2 treatment arms. The primary end point of hematologic CR and organ response were laboratory-based objective measures, which were unlikely to be affected by the open-label design. Hematologic response was assessed by an IRC, which further helped reduce bias related to subjective outcome assessment by individual investigators.
Data were immature for time-to-event outcomes, and median MOD-PFS and OS were not reached in either treatment arm. Given that the ANDROMEDA trial is ongoing, future analyses will provide more information with respect to time-to-event outcomes. For organ response time-to-event analyses, censoring due to start of a subsequent therapy (non–cross-resistant anti–plasma cell therapy) before progressive disease could bias the results. However, sensitivity analyses without censoring for subsequent therapy yielded results similar to those of the primary analysis.
Missing data and attrition in the trial may also affect the internal validity of the evidence. This includes missing assessments for organ response. Cardiac response could not be evaluated in 15% of patients with cardiac involvement at baseline. If data were not missing at random (i.e., if organ response assessment is missing for patients with weaker response in 1 treatment arm) this could bias the results. Moreover, throughout the trial, 26.6% and 35.2% of patients in the DCyBorD and CyBorD arms, respectively, discontinued trial treatment. Although this is not an unexpectedly high attrition rate in a cancer trial, the risk of attrition bias cannot be ruled out.
Longer duration of therapy in the DCyBorD arm and use of subsequent therapy in the trial is another possible source of bias. Based on a clinical practice treatment paradigm for AL amyloidosis that entails a treatment switch when hematologic response is suboptimal or in cases of worsening of organ function, which commonly occurs before disease progression, patients with suboptimal hematologic response or worsening organ function were permitted to start subsequent non–cross-resistant anti–plasma cell therapy before developing hematologic progression after 3 cycles of trial treatment. Only the primary outcome of hematologic CR is unaffected by this possible bias and can therefore be considered valid. Use of subsequent therapy may affect other key secondary outcomes, including VGPR, PR, and MOD-PFS. This issue is addressed in the primary analysis for MOD-PFS, which employed IPCW to adjust estimates of a treatment effect in the presence of subsequent non–cross-resistant anti–plasma cell therapy, which still showed longer MOD-PFS in the DCyBorD arm.
Although HRQoL assessment in the trial is important, some limitations should be noted. This end point was exploratory, and no adjustments were made for multiplicity. Three different instruments were used to assess HRQoL: the EORTC QLQ-C30, EQ-5D-5L, and SF-36v2. Only the SF-36v2 had been previously evaluated for content validity in the AL amyloidosis setting; there is no evidence available with respect to the validity, reliability, responsiveness, and MID of these instruments in patients with AL amyloidosis. Although these instruments have been used in trials of multiple myeloma and the 2 diseases share characteristics, AL amyloidosis has distinct clinical features. Notably, the organ and tissue impairment that is a hallmark of AL amyloidosis is not a key feature of multiple myeloma. As such, physical and functional impairment experienced by patients with AL amyloidosis with different organ involvement may be different from those of patients with multiple myeloma. Another limitation of HRQoL assessment is related to the open-label design of the study. Patient-reported outcomes are susceptible to bias from a lack of blinding of patients to their treatment. In addition, the different treatment durations in the 2 arms could affect the comparability of results. For example, the mean changes in the EORTC QLQ-C30 and EQ-5D-5L scores were markedly different between the 2 treatment arms at cycle 16, which is after the end of trial treatment in the CyBorD arm. It is possible that the improvement in HRQoL observed in the DCyBorD arm resulted from continued daratumumab monotherapy, and this is supported by the clinical data. However, the absence of information regarding validity and reliability in this patient population and differences in treatment duration affect the robustness of the HRQoL results.
External Validity
The ANDROMEDA trial included a heterogenous population of patients with AL amyloidosis and the wide range of clinical presentations were well-represented. The clinical experts consulted by CADTH and clinician groups indicated that the characteristics of the patient population, including prognostic factors such as cardiac stage, were reflective of the real-world clinical setting. However, the clinicians also noted that nearly all patients with AL amyloidosis would be suitable for treatment with DCyBorD, including those with advanced cardiac stage. Because patients with advanced cardiac stage disease and those with an ECOG PS greater than 2 were excluded from the study, the trial evidence regarding efficacy and safety of DCyBorD compared to CyBorD in these groups of patients is limited.
The comparator of the trial (CyBorD) is not approved for treatment of patients with AL amyloidosis in Canada. However, CyBorD is an appropriate comparator because it is standard of care for newly diagnosed AL amyloidosis in Canada. This is a special instance in which the drug regimen in review (i.e., DCyBorD) is the only Health Canada–approved treatment.
The primary and key secondary outcomes and assessment schedule were also reflective of clinical practice. Based on input from the clinical expert consulted by CADTH and clinician groups, in clinical practice, patients are assessed every 3 to 6 months. Formal response criteria have been established previously and were included in the ANDROMEDA trial. Standard response assessments in AL amyloidosis in clinical practice consist of 2 components: the hematologic or clonal response (as measured by serum and urine protein electrophoresis and immunofixation and serum FLC testing) and the organ response (as measured by levels of NT-proBNP for cardiac response, a combination of renal function and proteinuria for renal response, and the alkaline phosphatase level for liver involvement). Hematologic responses are the first to occur, and patients with deep hematologic responses are typically the first to experience subsequent organ responses. These are captured by the primary and secondary outcomes in the trial.
Indirect Evidence
No indirect evidence was submitted by the sponsor.
Discussion
Summary of Available Evidence
The ANDROMEDA trial that forms the evidence base for this review was a randomized, open-label, active-controlled, phase III trial that compared the efficacy of daratumumab SC in combination with CyBorD to CyBorD alone in the treatment of patients with newly diagnosed AL amyloidosis. The primary end point was hematologic CR and the key secondary end points were MOD-PFS and organ response rate. A total of 388 patients in 22 countries, including 19 patients from Canada, were randomized to treatment with either DCyBorD (n = 195) or CyBorD (n = 193). The median age was 64 years, 47.2% of patients were 65 years of age or older, and 58% of patients were male. The median time since initial AL amyloidosis diagnosis to randomization was 43 days. A median number of 2 organs were involved at baseline; 71.4% of patients had cardiac involvement (23.2% stage I, 40.2% stage II, 34.5% stage IIIA, and 2.1% stage IIIB) and 59.0% had renal involvement (53.9% stage I, 36.5% stage II, and 9.6% stage III). The median follow-up period was 11.9 months, with longer follow-up data presented for updated analyses after a median follow-up of 20.3 months.
Interpretation of Results
Efficacy
In the ANDROMEDA trial, DCyBorD resulted in a significantly higher frequency of hematologic CR compared to CyBorD alone in patients with newly diagnosed AL amyloidosis (53.3% versus 18.1%; relative risk ratio = 2.9; 95% CI, 2.1 to 4.1; P < 0.001). Hematologic CR was deeper and occurred more rapidly in the DCyBorD arm. A higher 6-month hematologic CR rate was observed in the DCyBorD arm compared with the CyBorD arm (49.7% versus 14.0%, respectively). Among responders (≥ VGPR), the median time to response (≥ VGPR) was short in both treatment arms; 17 days in the DCyBorD arm and 25 days in the CyBorD arm), but hematologic CR was reached more rapidly in the DCyBorD arm (median of 60 days) compared with the CyBorD arm (median of 85 days). Given the importance of rapid and deep hematologic response, these results are noteworthy. Based on key secondary outcomes, including MOD-PFS, organ response, and time to organ response, the results support and substantiate superior treatment effects of DCyBorD compared to CyBorD alone with respect to hematological response. The MRD negativity rate at the 10-5 threshold was 3-fold higher in the DCyBorD arm compared with the CyBorD arm, suggesting that the higher hematologic response observed with DCyBorD was accompanied by a deeper response.
Although data were immature as of the data cut-off date for primary analyses for some time-to-event end points, the higher cardiac and renal organ response rates observed in patients treated with DCyBorD provide further supportive evidence for the superiority of DCyBorD. Organ response is expected to follow hematologic response. Longer follow-up data from recent updated analyses with a median follow-up of 20.3 months show a sustained benefit of adding daratumumab to CyBorD, one that significantly improves hematologic response, which deepens over time, along with a doubling of the rates of cardiac and renal response. The CMRG consulted by CADTH noted that the AL amyloid fibrils can regress from the deposits when their production by plasma cells is halted. Consequently, improvement in organ function can be seen relatively quickly in some patients, but maximal responses may not be documented for months or even years after a deep hematologic response is achieved. Because AL amyloidosis is characterized by irreversible organ damage, a rapid and deep response is highly desired, and evidence from the ANDROMEDA trial suggests that DCyBorD achieves this treatment goal.
The results of the subgroup analysis, with respect to efficacy in patients with poor prognoses are also notable. The hematologic CR rate was equally high in patients treated with DCyBorD, regardless of poor prognostic factors like Mayo cardiac stage III, and t(11;14) mutation, whereas lower hematologic CR rates were observed for patients in the CyBorD arm with similarly poor prognoses. As the clinical expert consulted by CADTH and clinician groups noted, the addition of daratumumab to CyBorD fills an important unmet need in a particularly vulnerable subgroup of patients. While AL amyloidosis is eventually fatal in the majority of patients due to organ failure, those who most desperately need a deep and rapid response are those with extensive (stage II and III) cardiac involvement. The ANDROMEDA data demonstrate that the addition of daratumumab markedly improves outcomes for the subgroup of patients at particularly high risk of disease progression compared with CyBorD alone. In addition, as the CMRG noted, there has been prior concern regarding the use of bortezomib in patients with AL amyloidosis possessing the t(11;14) cytogenic abnormality, given the observation that both myeloma and AL amyloidosis patients with this cytogenetic subtype respond less well to this drug. Given the critical role of bortezomib in AL amyloidosis treatment until now, the t(11;14) abnormality is considered an adverse prognostic factor in AL patients. Results of the ANDROMEDA trial suggest that the addition of daratumumab to CyBorD may offer an improved treatment opportunity for these patients.
Relevant questions remain regarding the level of sustained response after 2 years of daratumumab monotherapy post-CyBorD and the optimal duration of long-term maintenance treatment and re-treatment with daratumumab. It is possible that prolonged exposure to anti-CD38 treatment could result in resistance or relapse and a lack of response to further treatment with anti-CD38 targeted therapy. These questions cannot be addressed by the available evidence from the ANDROMEDA trial, and future follow-up data from the ongoing trial may help to answer these questions.
With respect to quality of life, which is an important aspect of treatment desired by patients as noted by the patient group, the evidence from the ANDROMEDA study suggests that DCyBorD treatment has a positive impact on HRQoL. Although HRQoL scores were largely similar in the 2 treatment arms during the first 6 cycles of treatment, patients in the DCyBorD arm reported continued improvements in HRQoL following cycle 6, with notable differences at week 16 observed for overall global health status and fatigue. Although the different durations of treatment in the 2 treatment arms limits the value of comparing outcomes, the sustained improvement in HRQoL, consistent across HRQoL scores, suggests that daratumumab monotherapy following DCyBorD treatment can lead to clinical improvements without decrements in HRQoL over the treatment period.
Harms
The AE profile of DCyBorD was consistent with the known safety profiles of daratumumab, cyclophosphamide, bortezomib, and dexamethasone alone. Across all cycles, the incidence of any-grade (DCyBorD: 10.9%; CyBorD: 6.4%) and grade 3 or 4 (5.2% and 2.7%, respectively) neutropenia was higher in the DCyBorD arm. Serious neutropenia was reported in 1 patient in the DCyBorD arm and no patients in the CyBorD arm. These trends were consistent with hematology laboratory toxicity results and current prescribing information that daratumumab may increase neutropenia induced by background therapy. In the DCyBorD arm, the incidence of any-grade and grade 3 or grade 4 neutropenia decreased from cycle 7 onward. While infections (including serious infections) were reported more commonly in the DCyBorD arm compared with the CyBorD arm, they rarely led to treatment discontinuation or death in patients treated with DCyBorD. The median time to first infection was 9.7 weeks for patients treated with DCyBorD and 8.1 weeks for those treated with CyBorD, suggesting that the addition of daratumumab SC to the CyBorD regimen did not substantially accelerate the time to first infection. Additionally, low use of immunostimulants was reported in both treatment arms (DCyBorD: 5.2%; CyBorD: 3.2%). These data suggest that neutropenia did not lead to clinically consequential AEs in the majority of patient treated with DCyBorD. The safety profile of daratumumab is consistent with previous trials involving daratumumab in clinical trials in similar disease settings, notably multiple myeloma.25 The clinical experts consulted by CADTH and clinician groups also noted that neutropenia and increased risk of infection are well-known AEs associated with daratumumab therapy and are generally monitored and well managed in clinical practice.
There is substantial heterogeneity in the manifestations of AL amyloidosis–related cardiomyopathy. Although the incidence of certain cardiac TEAEs was higher in the DCyBorD arm, the overall data suggest that the observed cardiac AEs in both treatment arms are likely related to underlying AL amyloidosis–related cardiomyopathy. Serious and fatal cardiac events in both treatment arms were reported almost exclusively in patients who had cardiac involvement at baseline. Exposure-adjusted incidence rates for cardiac events in the trial indicated that there were no clinically meaningful differences in cardiac TEAEs between the treatment arms. The incidence of the cardiac disorder SOC was lower in the DCyBorD arm compared with the CyBorD arm for any of grade TEAE (4.07 versus 5.47, respectively) and grade 3 or grade 4 TEAEs (1.23 versus 2.25, respectively). Similarly, exposure-adjusted incidence rates in the DCyBorD arm compared with the CyBorD arm were lower for cardiac failure of any grade (0.88 and 1.25, respectively) and grade 3 or grade 4 cardiac failure (0.54 and 0.61, respectively), and for congestive cardiac failure of any grade and grade 3 or grade 4 (0.11 and 0.49, respectively). The clinical experts consulted by CADTH confirmed that cardiac-related AEs are expected to be at least partly related to cardiac involvement from the underlying disease.
A low and similar percentage of patients in both treatment arms reported any grade of TEAE (DCyBorD: 4.1%; CyBorD: 4.3%) and grade 3 or grade 4 TEAEs (3.1% and 2.7%, respectively) leading to treatment discontinuation. The most commonly reported (≥ 1% in either treatment arm) any-grade TEAEs leading to treatment discontinuation were cardiac failure and fatigue. Individual components of the CyBorD regimen were discontinued at a similar incidence between treatment arms, suggesting that daratumumab SC did not affect the toxicity of background therapy.
Conclusions
Daratumumab plus CyBorD is the first approved treatment for AL amyloidosis in Canada, where there are currently no publicly funded therapies. AL amyloidosis is a rare condition and has a poor prognosis, especially in patients with severe organ involvement. The evidence from the ANDROMEDA trial showed that the addition of daratumumab to CyBorD significantly improved hematological response in newly diagnosed patients with AL amyloidosis without decrements in HRQoL and no new safety concerns. The clinical benefit was supported by several secondary end points, including depth of response and organ response. The treatment benefit observed with DCyBorD was consistent in patients with poor prognoses, notably those with cardiac stage II or stage III disease and t(11;14) cytogenic abnormality, who respond less well to current treatments. Based on superiority of DCyBorD to CyBorD alone, the evidence from short-term outcomes suggests that DCyBorD can fill an important unmet treatment need in this patient population. Longer follow-up data are needed to establish the long-term benefit of adding daratumumab to CyBorD for the treatment of patients with AL amyloidosis.
References
1.Palladini G, Milani P, Merlini G. Management of AL amyloidosis in 2020. Hematology. 2020;2020(1):363-371. PubMed
2.Kumar S, Dispenzieri A, Lacy MQ, et al. Revised prognostic staging system for light chain amyloidosis incorporating cardiac biomarkers and serum free light chain measurements. J Clin Oncol. 2012;30(9):989-995. PubMed
3.Drug Reimbursement Review sponsor submission: Darzalex SC (daratumumab), 120mg/mL solution for subcutaneous injection [internal sponsor's package]. Toronto (ON): Janssen Inc.; 2021 Jul 28.
4.Merlini G, Dispenzieri A, Sanchorawala V, et al. Systemic immunoglobulin light chain amyloidosis. Nat Rev Dis Primers. 2018;4(1):38. PubMed
5.Palladini G, Milani P, Merlini G. Management of AL amyloidosis in 2020. Blood. 2020;136(23):2620-2627. PubMed
6.Darzalex (daratumumab): 1800 mg/15mL (120mg/ML) solution for subcutaneous injection [product monograph]. Toronto (ON): Janssen Inc.; 2021 Apr 12.
7.Kastritis E, Palladini G, Minnema MC, et al. Daratumumab-based treatment for immunoglobulin light-chain amyloidosis. N Engl J Med. 2021;385(1):46-58. PubMed
8.Clinical Study Report: JNJ-54767414AMY3001. A randomized phase 3 study to evaluate the efficacy and safety of daratumumab in combination with cyclophosphamide, bortezomib and dexamethasone (CyBorD) compared with CuBorD in newly diagnosed systemic AL amyloidosis [internal sponsor's report]. Toronto (ON): Janssen Research & Development; 2020 Aug 18.
9.Kastritis E, Sanchorawala V, Palladini G, et al. Subcutaneous daratumumab plus bortezomib, cyclophosphamide, and dexamethasone in patients with newly diagnosed light chain amyloidosis: updated results from the phase 3 ANDROMEDA study. ASCO Annual Meeting; 2021.
10.Kyle RA, Larson DR, Therneau TM, et al. Long-term follow-up of monoclonal gammopathy of undetermined significance. N Engl J Med. 2018;378(3):241-249. PubMed
11.Meziane I, Huhn S, Filho M, et al. Genome-wide association study of clinical parameters in immunoglobulin light chain amyloidosis in three patient cohorts. Haematologica. 2017;102(10):e411-e414. PubMed
12.Dumas B, Yameen H, Sarosiek S, Sloan JM, Sanchorawala V. Presence of t(11;14) in AL amyloidosis as a marker of response when treated with a bortezomib-based regimen. Amyloid. 2020;27(4):244-249. PubMed
13.Kobayashi H, Abe Y, Miura D, et al. Prevalence and clinical implications of t(11;14) in patients with amyloid light-chain amyloidosis with or without concurrent multiple myeloma. Jpn J Clin Oncol. 2019;49(2):195-198. PubMed
14.Hemminki K, Li X, Forsti A, Sundquist J, Sundquist K. Incidence and survival in non-hereditary amyloidosis in Sweden. BMC Public Health. 2012;12:974. PubMed
15.Pinney JH, Smith CJ, Taube JB, et al. Systemic amyloidosis in England: an epidemiological study. Br J Haematol. 2013;161(4):525-532. PubMed
16.Quock TP, Yan T, Chang E, Guthrie S, Broder MS. Epidemiology of AL amyloidosis: a real-world study using US claims data. Blood Adv. 2018;2(10):1046-1053. PubMed
17.Wechalekar AD, Schonland SO, Kastritis E, et al. A European collaborative study of treatment outcomes in 346 patients with cardiac stage III AL amyloidosis. Blood. 2013;121(17):3420-3427. PubMed
18.Palladini G, Dispenzieri A, Gertz MA, et al. New criteria for response to treatment in immunoglobulin light chain amyloidosis based on free light chain measurement and cardiac biomarkers: impact on survival outcomes. J Clin Oncol. 2012;30(36):4541-4549. PubMed
19.McGowan J, Sampson M, Salzwedel DM, Cogo E, Foerster V, Lefebvre C. PRESS Peer Review of Electronic Search Strategies: 2015 guideline statement. J Clin Epidemiol. 2016;75:40-46. PubMed
20.Grey matters: a practical tool for searching health-related grey literature. Ottawa (ON): CADTH; 2019: https://www.cadth.ca/grey-matters. Accessed 2021 Nov 4.
21.Comenzo RL, Reece D, Palladini G, et al. Consensus guidelines for the conduct and reporting of clinical trials in systemic light-chain amyloidosis. Leukemia. 2012;26(11):2317-2325. PubMed
22.Palladini G, Hegenbart U, Milani P, et al. A staging system for renal outcome and early markers of renal response to chemotherapy in AL amyloidosis. Blood. 2014;124(15):2325-2332. PubMed
23.Tang DI, Geller NL. Closed testing procedures for group sequential clinical trials with multiple endpoints. Biometrics. 1999;55(4):1188-1192. PubMed
24.Clinical Overview: JNJ-54767414. Daratumuman subcutaneous administration for the treatment of subjects with AL amyloidosis. Janssen Research & Development; 2020 Aug 25.
25.Dimopoulos MA, Terpos E, Boccadoro M, et al. Daratumumab plus pomalidomide and dexamethasone versus pomalidomide and dexamethasone alone in previously treated multiple myeloma (APOLLO): an open-label, randomised, phase 3 trial. Lancet Oncol. 2021;22(6):801-812. PubMed
26.Kvam AK, Fayers PM, Wisloff F. Responsiveness and minimal important score differences in quality-of-life questionnaires: a comparison of the EORTC QLQ-C30 cancer-specific questionnaire to the generic utility questionnaires EQ-5D and 15D in patients with multiple myeloma. Eur J Haematol. 2011;87(4):330-337. PubMed
27.McClure NS, Sayah FA, Xie F, Luo N, Johnson JA. Instrument-defined estimates of the minimally important difference for EQ-5D-5L index scores. Value Health. 2017;20(4):644-650. PubMed
28.EORTC QLC-C30 scoring manual. Brussels (Belgium): European Organisation for Research and Treatment of Cancer (EORTC); 2001: https://www.eortc.org/app/uploads/sites/2/2018/02/SCmanual.pdf. Accessed August 26, 2021.
29.Giesinger JM, Kieffer JM, Fayers PM, et al. Replication and validation of higher order models demonstrated that a summary score for the EORTC QLQ-C30 is robust. J Clin Epidemiol. 2016;69:79-88. PubMed
30.Aaronson NK, Ahmedzai S, Bergman B, et al. The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst. 1993;85(5):365-376. PubMed
31.Luo N, Fones CS, Lim SE, Xie F, Thumboo J, Li SC. The European Organization for Research and Treatment of Cancer Quality of Life Questionnaire (EORTC QLQ-c30): validation of English version in Singapore. Qual Life Res. 2005;14(4):1181-1186. PubMed
32.Davda J, Kibet H, Achieng E, Atundo L, Komen T. Assessing the acceptability, reliability, and validity of the EORTC Quality of Life Questionnaire (QLQ-C30) in Kenyan cancer patients: a cross-sectional study. J Patient Report Outcomes. 2021;5(1):4. PubMed
33.Snyder CF, Blackford AL, Sussman J, et al. Identifying changes in scores on the EORTC-QLQ-C30 representing a change in patients' supportive care needs. Qual Life Res. 2015;24(5):1207-1216. PubMed
34.Bedard G, Zeng L, Zhang L, et al. Minimal important differences in the EORTC QLQ-C30 in patients with advanced cancer. Asia Pac J Clin Oncol. 2014;10(2):109-117. PubMed
35.Kvam AK, Fayers P, Wisloff F. What changes in health-related quality of life matter to multiple myeloma patients? A prospective study. Eur J Haematol. 2010;84(4):345-353. PubMed
36.Osborne TR, Ramsenthaler C, Siegert RJ, Edmonds PM, Schey SA, Higginson IJ. What issues matter most to people with multiple myeloma and how well are we measuring them? A systematic review of quality of life tools. Eur J Haematol. 2012;89(6):437-457. PubMed
37.Kontodimopoulos N, Samartzis A, Papadopoulos AA, Niakas D. Reliability and validity of the Greek QLQ-C30 and QLQ-MY20 for measuring quality of life in patients with multiple myeloma. Sci World J. 2012;2012:842867-842867. PubMed
38.Oemar M, Janssen B. EQ-5D-5L user guide. Basic information on how to use the EQ-5D-5L instrument. Rotterdam (Netherlands): EuroQol Group; 2013: https://www.unmc.edu/centric/_documents/EQ-5D-5L.pdf. Accessed 2021 Nov 4.
39.EQ-5D-5L. Available modes of administration. 2021; https://euroqol.org/eq-5d-instruments/eq-5d-5l-available-modes-of-administration/. Accessed August 26, 2021.
40.2014 Alberta population norms for EQ-5D-5L. Calgary (AB): Health Quality Council of Alberta; 2014: https://d10k7k7mywg42z.cloudfront.net/assets/542f01f2edb2f37083002e54/2014_EQ_5D_5L_report_FINALFINAL.pdf. Accessed 2021 Nov 4.
41.Pickard AS, Neary MP, Cella D. Estimation of minimally important differences in EQ-5D utility and VAS scores in cancer. Health Qual Life Outcomes. 2007;5:70-70. PubMed
42.15D instrument. Welcome to the homepage of the 15D: the health-related quality of life (HRQoL) instrument. http://www.15d-instrument.net/15d/. Accessed 2021 Oct 15.
43.Maruish ME. User's manual for the SF-36v2 health survey; 3rd edition. Lincoln (RD): Quality Metric Incorporated; 2011.
44.Ware JE, Sherbourne CD. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care. 1992;30(6):473-483. PubMed
45.Jenkinson C, Stewart-Brown S, Petersen S, Paice C. Assessment of the SF-36 version 2 in the United Kingdom. J Epidemiol Community Health. 1999;53:46-50. PubMed
46.The Short-Form-36 health survey (Rand Corporation and John E. Ware Jr., 1990, revised 1996). Excerpt from Ian McDowell Measuring health: a guide to rating scales and questionnaires. New York (NY): Oxford University Press; 2006: http://www.med.uottawa.ca/courses/CMED6203/Index_notes/SF36%20fn%20.pdf. Accessed 2021 Apr 26.
47.White MK, Bayliss MS, Guthrie SD, Raymond KP, Rizio AA, McCausland KL. Content validation of the SF-36v2 R health survey with AL amyloidosis patients. J Patient Report Outcomes. 2017;1(1):13. PubMed
48.Möller A, Sartipy U. Long-term health-related quality of life following surgery for lung cancer. Eur J Cardiothorac Surg. 2012;41(2):362-367. PubMed
49.Zhang L, Su Y, Hua Y, et al. Validation of EORTC QLQ-LC43 for Chinese patients with lung cancer. Lung Cancer. 2014;85(1):94-98. PubMed
50.Bergman B, Aaronson NK, Ahmedzai S, Kaasa S, Sullivan M. The EORTC QLQ-LC13: a modular supplement to the EORTC Core Quality of Life Questionnaire (QLQ-C30) for use in lung cancer clinical trials. EORTC Study Group on Quality of Life. Eur J Cancer. 1994;30a(5):635-642. PubMed
51.Chiu CC, Lee KT, Wang JJ, et al. Preoperative health-related quality of life predicts minimal clinically important difference and survival after surgical resection of hepatocellular carcinoma. J Clin Med. 2019;8(5). PubMed
52.Jayadevappa R, Malkowicz SB, Wittink M, Wein AJ, Chhatre S. Comparison of distribution- and anchor-based approaches to infer changes in health-related quality of life of prostate cancer survivors. Health Serv Res. 2012;47(5):1902-1925. PubMed
Appendix 1: Literature Search Strategy
Note this appendix has not been copy-edited.
Clinical Literature Search
Overview
Interface: Ovid
Databases
MEDLINE All (1946-present)
Embase (1974-present)
Note: Subject headings and search fields have been customized for each database. Duplicates between databases were removed in Ovid.
Date of search: August 11, 2021
Alerts: Bi-weekly search updates until project completion
Search filters applied: No filters were applied to limit the retrieval by study type
Limits
No publication date limits
No language limits
Conference abstracts: excluded
Syntax | Description |
|---|---|
/ | At the end of a phrase, searches the phrase as a subject heading |
MeSH | Medical Subject Heading |
exp | Explode a subject heading |
* | Before a word, indicates that the marked subject heading is a primary topic; or, after a word, a truncation symbol (wildcard) to retrieve plurals or varying endings |
# | Truncation symbol for one character |
? | Truncation symbol for one or no characters only |
adj# | Requires terms to be adjacent to each other within # number of words (in any order) |
.ti | Title |
.ot | Original title |
.ab | Abstract |
.hw | Heading word; usually includes subject headings and controlled vocabulary |
.kf | Author keyword heading word (MEDLINE) |
.kw | Author keyword (Embase); keyword (CDSR) |
.dq | Candidate term word (Embase) |
.pt | Publication type |
.mp | Mapped term |
.rn | Registry number |
.nm | Name of substance word (MEDLINE) |
.yr | Publication year |
.jw | Journal title word (MEDLINE) |
.jx | Journal title word (Embase) |
freq=# | Requires terms to occur # number of times in the specified fields |
medall | Ovid database code: MEDLINE All, 1946 to present, updated daily |
oemezd | Ovid database code; Embase, 1974 to present, updated daily |
Multi-Database Strategy
(DARA or daratumumab* or darzalex* or HuMax-CD38 or HuMaxCD38 or HuMax-CD 38 or JNJ 54767414 or JNJ54767414 or 4Z63YK6E0E).ti,ab,ot,kf,hw,rn,nm.
cyclophosphamide/
(alkyroxan* or carloxan* or ciclolen* or cicloxal* or cyclo-cell or cycloblastin* or ciclofosfamid* or ciclophosphamid* or clafen* or claphen* or cyclophosphamid* or cyclophosphan* or cyclophosphoramid* or cyclostin* or cyklofosfamid* or cytophosphan* or endoxan* or enduxan* or endoxon* or genoxal* or ledoxan* or ledoxina* or lyophilized cytoxan* or mitoxan* or neosar* or neosan* or noristan* or procytox* or semdoxan* or sendoxan* or syklofosfamid* or B-518 or B518 or HSDB-3047 or HSDB3047 or NSC-26271 or NSC26271 or SK-20501 or SK20501 or zyklophosphamid* or 6UXW23996M or 8N3DW7272P).ti,ab,ot,kf,hw,rn,nm.
2 or 3
bortezomib/
(bortezomib* or velcade* or bomib* or borcade* or bortega* or bortero* or bortesum* or egybort* or exfucikanet* or imozet* or mibor* or neomib* or nyubortez* or velkeyd* or zegomib* or zuricade* or bortecad* or chemobort* or HSDB-7666 or HSDB7666 or LDP-341 or LDP341 or MG-341 or MG341 or MLN-341 or MLN341 or BXCL-101 or BXCL101 or NSC-681239 or NSC681239 or PS-341 or PS341 or 69G8BD63PP).ti,ab,ot,kf,hw,rn,nm.
5 or 6
dexamethasone/
(adrecort* or adrenocot* or "aeroseb-D" or "aeroseb-dex" or aflucoson* or alfalyl* or anaflogistico* or aphtasolon* or arcodexan* or artrosone* or auxiron* or azium* or bidexol* or "bisu DS" or calonat* or cebedex* or colofoam* or corsona* or corsone* or cortastat* or cortidex* or cortidexason* or cortidrona* or cortidrone* or cortisumman* or dacortina fuerte* or dacortine fuerte* or dalalone* or danasone* or "de-sone la" or decacortin* or decadeltoson* or decaderm* or decadion* or decadron* or cecaesadril* or decagel* or decaject* or decalix* or decamethason* or decasone* or decaspray* or decasterolone* or decdan* or declione* or decofluor* or dectancyl* or dekacort* or delladec* or deltafluoren* or dergramin* or deronil* or desacort* or desadrene* or desalark* or desametason* or desamethason* or desameton* or deseronil* or desigdron* or "dex-ide" or dexa mamallet* or dexa-cortidelt* or dexa-cortisyl* or dexa-scheroson* or "dexa-sine" or dexacen or dexachel* or dexacort* or dexacortal* or dexacorten* or dexacortin* or dexacortisyl* or dexadabroson* or dexadecadrol* or dexadrol* or dexadeltone* or dexafarma* or dexagel* or dexagen* or dexahelvacort* or dexakorti* or dexalien* or dexalocal* or dexalona* or dexamecortin* or dexameson* or dexametason* or dexameth* or dexamonozon* or dexan or dexapolcort* or dexapos or dexapot* or dexaprol* or dexascheroson* or dexascherozon* or dexason or dexinolon* or dexinoral* or dexionil* or dexmethson* or dexona or dexone or DexPak or dextelan* or dextrasone* or dextenza* or dezone* or dibasona* or dinormon* or dxm or dxms or esacortene* or "ex s1" or exadion* or firmalone* or fluormethyl prednisolone* or fluormethylprednisolon* or fluormone* or fluorocort* or fluorodelta* or fortecortin* or gammacorten* or grosodexon* or hexadecadiol* or hexadecadrol* or hexadiol* or hexadrol* or "HL-dex" or isnacort* or isoptodex* or "isopto-dex" or isoptomaxidex* or "lokalison F" or loverine* or luxazone* or marvidone* or maxidex* or mediamethasone* or megacortin* or mephaseson* or metasolon* or methazon* or methazonion* or methylfluorprednisolone* or metisone lafi or mexasone* or mexidex* or millicorten* or mymethasone* or neoforderx* or nisomethasona* or novocort* or "ocu-trol" or "oftan-dexa" or opticorten* or opticortinol* or oradexan* or oradexon* or orgadrone* or ozurdex* or pidexon* or policort* or posurdex* or "predni F" or "prednisolon F" or "prednisolone F" or prodexona* or prodexone* or sanamethasone* or santeson* or sawasone* or solurex* or spoloven* or sterasone* or "sunia Sol D" or superprednol* or thilodexine* or triamcimetil* or turbinaire* or vexamet* or visumetazone* or visumethazone* or AI3-50934 or AI350934 or CCRIS-7067 or CCRIS7067 or DXMS or HSDB-3053 or HSDB3053 or MK-125 or MK125 or NSC-34521 or NSC34521 or ISV-305 or ISV305 or OTO-104 or OTO104 or 7S5I7G3JQL).ti,ab,ot,kf,hw,rn,nm.
8 or 9
(Cyclophosphamide-Bortezomib-Dexamethasone or CyBorD*).ti,ab,kf.
4 and 7 and 10
11 or 12
1 and 13
DCyBorD.ti,ab,kf.
14 or 15
16 use medall
exp amyloidosis/
(amyloidosis or amyloidoma* or paraamyloidosis or familial Mediterranean fever or Muckle Wells syndrome).ti,ab,kf.
(amyloid* adj4 (fibril* or neuropath* or angiopath* or polyneuropath* or light-chain or ATTR or ATTRm or ATTRwt or hereditary or senile or systemic or dialysis-related or wild-type or familial or plasma* or plasmacytic or plasmocytic or plasmocyte or tumor or tumour or tumors or tumours or cancer* or neoplas* or malignan*)).ti,ab,kf.
(protein adj3 (misfold* or transthyretin)).ti,ab,kf.
18 or 19 or 20 or 21
1 and 22
23 use medall
17 or 24
*daratumumab/
(DARA or daratumumab* or darzalex* or HuMax-CD38 or HuMax-CD 38 or HuMaxCD38 or JNJ 54767414 or JNJ54767414).ti,ab,kw,dq.
26 or 27
*Cyclophosphamide/
(alkyroxan* or carloxan* or ciclolen* or cicloxal* or cyclo-cell or cycloblastin* or ciclofosfamid* or ciclophosphamid* or clafen* or claphen* or cyclophosphamid* or cyclophosphan* or cyclophosphoramid* or cyclostin* or cyklofosfamid* or cytophosphan* or endoxan* or enduxan* or endoxon* or genoxal* or ledoxan* or ledoxina* or lyophilized cytoxan* or mitoxan* or neosar* or neosan* or noristan* or procytox* or semdoxan* or sendoxan* or syklofosfamid* or B-518 or B518 or HSDB-3047 or HSDB3047 or NSC-26271 or NSC26271 or SK-20501 or SK20501 or zyklophosphamid*).ti,ab,kw,dq.
29 or 30
*bortezomib/
(bortezomib* or velcade* or bomib* or borcade* or bortega* or bortero* or bortesum* or egybort* or exfucikanet* or imozet* or mibor* or neomib* or nyubortez* or velkeyd* or zegomib* or zuricade* or bortecad* or chemobort* or HSDB-7666 or HSDB7666 or LDP-341 or LDP341 or MG-341 or MG341 or MLN-341 or MLN341 or BXCL-101 or BXCL101 or NSC-681239 or NSC681239 or PS-341 or PS341).ti,ab,kw,dq.
32 or 33
*dexamethasone/
(adrecort* or adrenocot* or "aeroseb-D" or "aeroseb-dex" or aflucoson* or alfalyl* or anaflogistico* or aphtasolon* or arcodexan* or artrosone* or auxiron* or azium* or bidexol* or "bisu DS" or calonat* or cebedex* or colofoam* or corsona* or corsone* or cortastat* or cortidex* or cortidexason* or cortidrona* or cortidrone* or cortisumman* or dacortina fuerte* or dacortine fuerte* or dalalone* or danasone* or "de-sone la" or decacortin* or decadeltoson* or decaderm* or decadion* or decadron* or cecaesadril* or decagel* or decaject* or decalix* or decamethason* or decasone* or decaspray* or decasterolone* or decdan* or declione* or decofluor* or dectancyl* or dekacort* or delladec* or deltafluoren* or dergramin* or deronil* or desacort* or desadrene* or desalark* or desametason* or desamethason* or desameton* or deseronil* or desigdron* or "dex-ide" or dexa mamallet* or dexa-cortidelt* or dexa-cortisyl* or dexa-scheroson* or "dexa-sine" or dexacen or dexachel* or dexacort* or dexacortal* or dexacorten* or dexacortin* or dexacortisyl* or dexadabroson* or dexadecadrol* or dexadrol* or dexadeltone* or dexafarma* or dexagel* or dexagen* or dexahelvacort* or dexakorti* or dexalien* or dexalocal* or dexalona* or dexamecortin* or dexameson* or dexametason* or dexameth* or dexamonozon* or dexan or dexapolcort* or dexapos or dexapot* or dexaprol* or dexascheroson* or dexascherozon* or dexason or dexinolon* or dexinoral* or dexionil* or dexmethson* or dexona or dexone or DexPak or dextelan* or dextrasone* or dextenza* or dezone* or dibasona* or dinormon* or dxm or dxms or esacortene* or "ex s1" or exadion* or firmalone* or fluormethyl prednisolone* or fluormethylprednisolon* or fluormone* or fluorocort* or fluorodelta* or fortecortin* or gammacorten* or grosodexon* or hexadecadiol* or hexadecadrol* or hexadiol* or hexadrol* or "HL-dex" or isnacort* or isoptodex* or "isopto-dex" or isoptomaxidex* or "lokalison F" or loverine* or luxazone* or marvidone* or maxidex* or mediamethasone* or megacortin* or mephaseson* or metasolon* or methazon* or methazonion* or methylfluorprednisolone* or metisone lafi or mexasone* or mexidex* or millicorten* or mymethasone* or neoforderx* or nisomethasona* or novocort* or "ocu-trol" or "oftan-dexa" or opticorten* or opticortinol* or oradexan* or oradexon* or orgadrone* or ozurdex* or pidexon* or policort* or posurdex* or "predni F" or "prednisolon F" or "prednisolone F" or prodexona* or prodexone* or sanamethasone* or santeson* or sawasone* or solurex* or spoloven* or sterasone* or "sunia Sol D" or superprednol* or thilodexine* or triamcimetil* or turbinaire* or vexamet* or visumetazone* or visumethazone* or AI3-50934 or AI350934 or CCRIS-7067 or CCRIS7067 or DXMS or HSDB-3053 or HSDB3053 or MK-125 or MK125 or NSC-34521 or NSC34521 or ISV-305 or ISV305 or OTO-104 or OTO104).ti,ab,kw,dq.
35 or 36
31 and 34 and 37
(Cyclophosphamide-Bortezomib-Dexamethasone or CyBorD*).ti,ab,kw,dq.
38 or 39
28 and 40
41 use oemezd
(conference abstract or conference review).pt.
42 not 43
exp amyloidosis/
(amyloidosis or amyloidoma* or paraamyloidosis or familial Mediterranean fever or Muckle Wells syndrome).ti,ab,kw,dq.
(amyloid* adj4 (fibril* or neuropath* or angiopath* or polyneuropath* or light-chain or ATTR or ATTRm or ATTRwt or hereditary or senile or systemic or dialysis-related or wild-type or familial or plasma* or plasmacytic or plasmocytic or plasmocyte or tumor or tumour or tumors or tumours or cancer* or neoplas* or malignan*)).ti,ab,kw,dq.
(protein adj3 (misfold* or transthyretin)).ti,ab,kw,dq.
45 or 46 or 47 or 48
28 and 49
50 not 43
51 use oemezd
44 or 52
25 or 53
remove duplicates from 54
Clinical Trials Registries
ClinicalTrials.gov
Produced by the US National Library of Medicine. Targeted search used to capture registered clinical trials.
Search -- Studies with results: amyloidosis AND daratumumab
WHO ICTRP
International Clinical Trials Registry Platform, produced by the World Health Organization. Targeted search used to capture registered clinical trials.
Search terms – amyloidosis AND daratumumab
Health Canada’s Clinical Trials Database
Produced by Health Canada. Targeted search used to capture registered clinical trials.
Search terms – amyloidosis AND daratumumab
EU Clinical Trials Register
European Union Clinical Trials Register, produced by the European Union. Targeted search used to capture registered clinical trials.
Search terms – amyloidosis AND daratumumab
Grey Literature
Search dates: July 30 to August 5, 2021
Keywords: amyloidosis AND daratumumab
Limits: No publication date limits; no date limits
Updated: Search updated prior to the completion of stakeholder feedback period
Relevant websites from the following sections of the CADTH grey literature checklist Grey Matters: A Practical Tool for Searching Health-Related Grey Literature were searched:
Health Technology Assessment Agencies
Health Economics
Clinical Practice Guidelines
Drug and Device Regulatory Approvals
Advisories and Warnings
Drug Class Reviews
Clinical Trials Registries
Databases (free)
Health Statistics
Internet Search
Open Access Journals
Appendix 2: Description and Appraisal of Outcome Measures
Note this appendix has not been copy-edited.
Aim
To describe the following outcome measures and review their measurement properties (validity, reliability, responsiveness to change, and MID):
European Organisation for the Research and Treatment of Cancer Quality of Life Questionnaire – core 30 items (EORTC QLQ-C30)
European Quality of Life Scale – 5 Dimensions – 5 Levels (EQ-5D-5L)
SF-36v2 health survey
Findings
Table 18: Summary of Outcome Measures and Their Measurement Properties
Outcome measure | Type | Conclusions about measurement properties | MID |
|---|---|---|---|
EORTC QLQ-C30 | 30-item, patient-reported, cancer-specific quality of life questionnaire using 4- and 7-point Likert scales | Validity, reliability, and Responsiveness: Evidence of validity and reliability in populations with cancer.
| Patients with cancer:
Patients with MM:
Not identified in AL amyloidosis. The Global QoL scale had statistically significant standardized response means (SRM) in MM patients who improved (SRM 0.32, P < 0.01) and deteriorated (SRM 0.57, P < 0.01).26 |
EQ-5D-5L | EQ-5D-5L index: Generic, preference-based measure of HRQoL consisting of 5 domains: mobility, self-care, usual activities, pain/discomfort, and anxiety/depression. Scores range from 0-1 with higher scores indicating better health status. EQ VAS: Generic, preference-based measure of HRQoL presented as a scale from 0-100 with 0 anchored as the worst possible health state and 100 as the best. | Validity, reliability, and responsiveness: Evidence of validity and reliability in diverse patient populations.
| Index score in the general Canadian population:
Patients with MM:
Not identified in AL amyloidosis. |
SF-36v2 | Patient-reported generic instrument that measures functional health and well-being in patients using 8 dimensions:
| The SF-36 has been previously validated for a variety of health states and diseases. Content validity has been evaluated in patients with AL amyloidosis. No evidence for reliability and responsiveness in AL amyloidosis | General population MID:
Not identified in AL amyloidosis. |
EORTC QLQ-C30 = European Organisation for the Research and Treatment of Cancer Quality of Life Questionnaire Core 30; EORTC QLQ-CR29 = European Organisation for the Research and Treatment of Cancer Quality of Life Questionnaire – colorectal cancer module 29 items; EQ-5D-3L = European Quality of Life Scale – 5 Dimensions – 3 Levels; MID = minimal important difference. MCS = mental summary component.
European Organisation for the Research and Treatment of Cancer Quality of Life Questionnaire Core 30 Version 3.0.
The European Organisation for the Research and Treatment of Cancer Quality of Life Questionnaire Core 30 (EORTC QLQ-C30) is a multidimensional instrument commonly used to evaluate health-related quality-of-life outcomes in patients and was originally designed to gather information on specific issues in cancer patients and assess changes in response to treatment.28,29 The first 2 versions of the questionnaire were tested in lung cancer patients from 13 different countries to assess its reliability and validity.30 The current version (version 3.0) is composed of 30 questions which are scored in multi-item 5 functional scales (physical functioning, role functioning, cognitive functioning, emotional functioning, and social functioning), a global health status / QoL scale and a 9-item symptom scale (fatigue, nausea and vomiting, pain, dyspnea, appetite loss, insomnia, constipation, diarrhea, and financial difficulties) that assesses commonly reported symptoms in patients. This questionnaire has also been adapted for specific disease areas and allows the incorporation of supplementary modules to gather more information related to the specific disease.28
The EORTC QLQ-C30 uses a 1-week recall period to assess function and symptoms. Most questions have 4 response options (“not at all,” “a little,” “quite a bit,” “very much”) with scores on these items ranging from 1 to 4. For the 2 items that form the global QoL scale, the response format is a 7-point Likert-type scale with anchors at 1 = “very poor” and 7 = “excellent.” Scale measurements can be transformed to achieve standardized scores ranging from 0 to 100. Raw scores could also be reported alongside the transformed scores for clarity in interpretation. Higher functioning scores and lower symptom scores indicate better quality of life. According to the EORTC QLQ-C30 scoring algorithm, if there are missing items for a scale (i.e., the participant did not provide a response), the score for the scale can still be computed if there are responses for at least one-half of the items. In calculating the scale score, the missing items are simply ignored — an approach that assumes that the missing items have values equal to the average of those items for what the respondent completed.28
Validity and Reliability
The validity and reliability of the EORTC QLQ-C30 has been verified in many different disease conditions and cancers. A few examples include:
A cross-sectional conducted in a convenience sample of cancer patients in Singapore.31 This study assessed internal consistency reliability by calculating Cronbach alpha for all QLQ-C30 scales. The Cronbach alpha derived from the study was at least 0.70 for 6 of the 9 assessed QLQ-C30 scales; cognitive functioning, physical functioning, and nausea and vomiting had a Cronbach alpha ranging from 0.19 to 0.68.
A cross-sectional study conducted in Kenya that aimed to evaluate the psychometric properties of the EORTC QLQ-C30, using the English or Kiswahili version in 100 patients with cancer.32 The investigators also assessed the internal consistency of each scale of the questionnaire using Cronbach alpha coefficients. With the exception of the cognitive function scale, all of the scales had a Cronbach alpha of 0.70 of greater.
Minimal Important Difference
A Canadian study conducted in 2015 estimated the MIDs of EORTC QLQ-C30 scales using data from 193 patients newly diagnosed with breast and colorectal cancers.33 MIDs associated with a statistically significant improvement in supportive care needs ranged from 10 to 32 points for ‘improvement’ while those for ‘worsening’ ranged from 9 to 21 points. The range for unchanged supportive care needs was from 1-point worsening to 16-point improvement in EORTC QLQ-C30 score. Conclusions made by the authors suggest that a 10-point change in EORTC QLQ-C30 score represented changes in supportive care needs, and therefore, should be considered for clinical use.
Another Canadian study conducted in 2014, estimated the MID for EORTC QLQ-C30 in 369 patients with advanced cancer who completed the questionnaire at baseline and 1-month post-radiation.34 The most common cancer type was breast cancer, followed by lung, prostate, gastrointestinal, renal cell, and other cancers. MID was estimated using both anchor- and distribution-based methods for improvement and deterioration. Two anchors of overall health and overall QoL were used, both taken directly from the EORTC QLQ-C30 (questions 29 and 30) where patients rated their overall health and QoL themselves. Improvement and deterioration were categorized as an increase or decrease by 2 units to account for the natural fluctuation of patient scoring. With these 2 anchors, the estimated MIDs across all EORTC QLQ-C30 scales ranged from 9.1 units to 23.5 units for improvement, and from 7.2 units to 13.5 units for deterioration. Distribution-based estimates were closest to 0.5 SD.
Responsiveness and MID for Patients With AL Amyloidosis and Multiple Myeloma
The EORTC QLQ-C30 has been validated across countries around the world and in various conditions. However, the EORTC QLQ-C30 has not been validated in patients with AL amyloidosis and therefore its validity, reliability, and responsiveness to change have not been evaluated in the patient population of interest. No literature on the MID specific to AL amyloidosis patients was found.
Kvam et al., in 2010 conducted a prospective study, which tested the responsiveness of the EORTC QLQ-30 and estimated the MID in a group of 239 patients diagnosed with multiple myeloma. Patients completed questionnaires at inclusion (T1) and 3 months later (T2) (followed by a structured interview). The MID was estimated based of the mean score change (T2-T1) for patients who responded either ‘improved,’ ‘deteriorated’ or ‘unchanged’ during the interviews.35 A receiver-operating characteristic (ROC) curve method in addition to the effect size calculation using standard deviations at baseline were used to derive the MIDs which resulted in slight variations. Patients considered as ‘improved’ had an MID that ranged from 6 to 15 as opposed to 9 to 17 (on a scale from 0 to 100) for ‘deteriorated.’ MIDs derived from the ROC analysis ranged between 7 to 17 were considered clinically significant. MIDs derived using the effect size method suggested that values in the range between 5 to 6 represented a small change whereas values in the range between 11 to 15 represented a medium change. MID calculations obtained from both the ROC’s analysis and the effect size method suggest that changes in the instrument ranging from 6 to 17 were considered important in patients with multiple myeloma and thus the authors concluded that changes below 6, even if statistically significant, may not be important in this patient population.
Validity in Multiple Myeloma Patients
A systematic review conducted by Osborne et al. (2012)36 evaluated the content validity of HRQoL tools in terms of their ability to capture all issues important to patients with MM. A Cronbach alpha of 0.54 to 0.89 was identified for all subscales (0.49 for social functioning in 1 group) for the EORTC QLQ-C30 instrument. The construct validity for the EORTC QLQ-C30 instrument was evaluated. The authors noted that all subscales were impaired when compared to the general population. A significant improvement in some scales was observed with increasing time post-hematopoietic stem cell transplant. In total, 67% and 43% of patients in these studies scored below the 10th percentile for physical functioning and global quality-of-life subscales. The functional subscales and the global quality of life observed in the studies evaluated were lower in myeloma patients compared to the general hematology population. Other subscales such as pain, fatigue, physical and global quality of life were able to discriminate between patients that improved and those with stable/deteriorated. Apart from the diarrhea, the rest of the subscales could differentiate between patients with different performance status and response status. Other observations made in the study include a significant difference in global quality of life or global health status in treatment arms in trials. Non-overlapping scales were observed after scale structure was verified. The criterion validity was also evaluated and no correlation of any subscale with the SEIQoL-Index (individual quality-of-life scale with patient-nominated domains) was observed which suggested independence.
Reliability Assessed in Multiple Myeloma Patients
Kontodimopoulos et al. (2012) assessed the reliability of the Greek version of the EORTC QLQ-C30 in a sample of 89 patients with myeloma recruited from 2 hospitals in Athens. The Greek version of the SF-36 instrument was used as a gold standard. The hypothesized scale structure, Cronbach alpha, and construct validity were assessed (using Spearman correlations). The Cronbach alpha obtained was > 0.70 for all scales except in cognitive functioning. The global health status/QoL obtained was 0.92. The scores obtained for functional scales were PF = 0.80, RP = 0.9, EF = 0.84, CF = 0.57, SF = 0.77. Scores obtained from symptom scales included fatigue = 0.89, nausea and vomiting = 0.74, pain = 0.8. No scores were reported for dyspnea, insomnia, appetite loss, constipation, diarrhea, and financial difficulties. They study also observed significant higher item-scale correlations between the items and their hypothesized scales than with the competing scales.37
EQ-5D Questionnaire
The EQ-5D questionnaire is a generic instrument standardized to capture utility-based measures of HRQoL in patients across diverse health conditions. The first version, the EQ-5D-3L was introduced in the 1990s and uses 5 dimensions to describe health status (mobility, self-care, usual activities, pain/discomfort and anxiety/depression) and respondents can further describe them based on 3 levels of severity (no problems, some or moderate problems, and extreme problems).38-40 The most recent version, a 5-level version (EQ-5D-5L) was introduced in 2005 to address limitations reportedly observed in the EQ-5D-3L questionnaire (issues associated with sensitivity and ceiling effects). It is available in more than 130 languages and can be administered via different modes (paper, smart phones, tablets, and laptops or desktops) to patients. The EQ-5D-5L differs from the EQ-5D-3L questionnaire in the following aspects:
The most severe label for the mobility dimension was changed from “confined to bed” to “unable to walk about,” enhancing its applicability and increasing the sensitivity of the mobility dimension.
The instructions for the EQ VAS task were simplified, making the task easier to complete and easier to score.39
The questionnaire is composed of 2 parts:
The first part includes the descriptive EQ-5D system: It uses 5 levels (L1=no problems, L2=slight problems, L3= moderate problems, L4= severe problems, and L5= extreme problems) to describe patient responses to the 5 dimensions (this produces 3,125 unique health states possible for each patient) as opposed to the EQ-5D-3L (243 possible health states that can be achieved for a patient). Digits from the 5 dimensions are further combined to obtain a 5-digit number which describes the patient’s health state. For example, 11111 translates to no problems on the any dimension.38
The numerical values assigned to the levels for each dimension reflect ranked order categories of functioning. In terms of measurement properties, these are ordinal data, which do not have interval properties, therefore, should not summed or averaged. For example, to produce an individual dimension “score.” Results can then be captured as a digit number or single index value representing an aggregated value from the 5 dimensions. Index values are usually country specific (considering patient and population preferences) and differ based on the scoring algorithm used. These scores are used to derive the quality adjusted life years needed to perform economic evaluations. The range of index scores will differ according to the scoring algorithm used; however, in all scoring algorithms of the EQ-5D-5L, a score of 0 represents the health state “dead” and 1.0 reflects “perfect health.” Negative scores are also possible for those health states that society (not the individual patient) considers to be “worse than dead.”38,40
The second part of the questionnaire consists of a 20 cm EQ VAS. The end points of this scale range from 0 to 100 corresponding to “worst imaginable health state” and best imaginable health state” respectively allowing respondents to self-rate their health that day by marking a point on the VAS scale.38,40
The EQ-5D-5L has been validated across countries around the world for various disease conditions. However, the EQ-5D-5L has not been validated in patients with AL amyloidosis therefore its validity, reliability, and responsiveness to change have not been evaluated in the patient population of interest.
Minimal Important Difference
No literature presenting information on the MID specific to patients with AL amyloidosis was found.
Pickard et al. estimated a range of MIDs for the EQ-5D instrument in a retrospective study using a cross-sectional sample of 543 cancer patients (presenting with advanced stage 3 or 4, cancer of the bladder, brain, breast (females), colon/rectum, head/neck, liver/pancreas, kidney, lung, lymphoma, ovary (females), and prostate), of which 50 had diagnosis of lung cancer.41 Utility Index scores were derived based on UK and US- population specific preference value sets and the VAS scores were estimated based on anchor-based (using ECOG performance status (PS) ratings and FACT-G total score-based quintiles) and distribution-based methods. The country specific MID estimates for all cancers including lung cancer obtained from the study were as follows
UK-Specific Utility Scores
PS estimates: 0.10 to 0.12 (for all cancers including lung cancer)
FACT-G quintiles estimates: 0.09 to 0.10 for all cancers and 0.07 to 0.08 for lung cancer
US-Specific Utility Scores
PS estimates: 0.07 to 0.09 (for all cancers including lung cancer)
FACT-G quintiles estimates: 0.06 to 0.07 for all cancers and 0.05 to 0.06 for lung cancers
They observed similar MIDs for VAS scores for all cancers including lung cancer which ranged from 8 to 12 (PS) and from 7 to 10 using the FACT-G quintiles ancho-based method.
Responsiveness and MID in Patients With Multiple Myeloma
Kvam et al. conducted a comparative study to evaluate the responsiveness of the cancer-specific EORTC QLQ-C30 instrument, with the EQ-5D and 15D generic instruments and to estimate the minimal score differences in sample of 260 patients with multiple myeloma.26 Patients were asked to complete questions at the beginning of the study (period T1) and after a 3-month follow-up period (T2) accompanied by a structured interview performed by an investigator. Patients were asked to rate their health status as either improved, unchanged, or deteriorated during the follow-up period. Questionnaires were administered in a fixed order: EORTC QLQ-C30, EQ-5D, and 15D, respectively. The 15D is a self-administered questionnaire that measures patient-reported outcomes in adults (age 16+ years). The health status is measured across 15 dimensions of health (breathing, mental function, communication, vision, mobility, usual activities, discomfort and symptoms, depression, vitality, and sexual activity hearing, eating, eliminating, sleeping, distress), each having 5 levels. The respondents choose the dimensions and levels which are combined into a single index measure. The 15D scores are generated based on a set of utility weights and scores are scaled on a 0 to 1 scale.42
The responsiveness and MIDs for these instruments were derived from the mean score changes obtained from the 2 periods (T2-T2). Standardized scores were used to assess instrument responsiveness using the Wilcoxon test for paired differences. Patients with significantly higher T2 scores in all 3 questionnaires administered were reported as ‘improved’ (P < 0.01). For patients that reported ‘improved’ states of health, the EQ-5D was the most responsive questionnaire (standardized response means [SRM] of 0.43), followed by the 15D (SRM 0.37) and the global QoL scale of the EORTC QLQ-C30 (SRM 0.32). Scores were negligible in patients that responded as being ‘unchanged’ while for those reportedly having a ‘deteriorated’ state, lower scores were seen at T2. Scores obtained from the 15D questionnaire showed no statistically significant difference between scores obtained at T2 and T1. The MIDs derived for patients labelled as ‘improved’ were 8, 0.08, and 0.03 as opposed to 12, 0.10 and 0.02 for patients labelled as ‘deteriorated’ in the EORTC QLQ-C30, EQ-5D and 15D instruments, respectively. The investigators concluded that all 3 questionnaires administered in the study showed acceptable responsiveness in patients who improved, however, they do not recommend the use of 15D questionnaires to assess quality of life in patients diagnosed with multiple myeloma given that the questionnaire did not respond optimally in patients who had reported “deteriorated” in the study.
Short Form (36) Health Survey Version 2
The SF-36 is a generic questionnaire that captures self-reported health outcomes in patients across different disease areas. It has been widely used in clinical trials to study the impact of chronic disease on health-related quality of life.43 Two versions of the instrument exist: The original SF-3644 and the SF-36 version 2 (SF-36 v2).43,45 The SF-36 v2 contains minor changes to the original survey in the following areas: instructions (reduced ambiguity), questions and answers (better layout), item-level response choices (increased), cultural/language comparability (increased), and elimination of a response option from the items in the mental health and vitality subscales.43,45
The SF-36v2 health survey questionnaire consists of 36 items which represent 8 subscales: physical functioning (PF; 10 items), role-physical (RP; role limitations due to physical problems [4 items]), bodily pain (BP; 2 items), general health perceptions (GH; 5 items), vitality (VT; 4 items), social functioning (SF; 2 items), role-emotional (RE; role limitations due to emotional problems [3 items]), and mental health (MH; 5 items). The second question of the survey is a single item not used for scoring the 8 scales, but instead used to estimate the general health from a cross-sectional standpoint.46 The recall period is 4 weeks and each response to an item option is presented on a 3- to 6-point, Likert-like scale.43,45 Each item is converted to a score ranging from 0 to 100 where a higher value indicates a more favourable health state and item scores are averaged together to create the 8 subscale scores.
The questionnaire also provides 2 component summaries: the physical component summary (PCS) and the mental component summary (MCS). These are created by aggregating the 8 subscales according to a scoring algorithm. The first 4 subscales (PF, RP, BP, and GH) belong to the PCS while the next 4 subscales (VT, SF, RE, and MH) make up the MCS. Like the individual items, the PCS, MCS, and 8 subscale scores are each measured from 0 to 100. The subscale and summary scores (PCS and MCS) are T-scores standardized to a reference population with a mean of 50 and standard deviation of 10.43 Thus, a score of 50 on any scale would be at the average or norm of the reference and a score 10 points lower (i.e., 40) would be 1 standard deviation below the norm.
Content Validity in AL Amyloidosis Patients
White et al. (2017) evaluated the content validity of SF-36v2 questionnaires in patients with AL amyloidosis. Qualitative interviews were administered to patients and physicians that targeted 3 phases of the qualitative research: concept elicitation interviews among physicians, concept elicitation interviews among patients, and cognitive debriefing interviews among patients.47 Results obtained by the authors showed that physicians tend to discuss the importance of physical functioning, general health, mental/emotional health sleep, fatigue, and work impact for AL amyloidosis while patients discussed the social, physical, role, and emotional impacts of AL amyloidosis and various treatments during the interviews. The authors also highlight that physician seldom used patient-reported outcome measures of health quality related quality of life as a standard in routine practice. Their results suggest that cognitive debriefing interviews support the relevance of concepts measured by the instrument. The authors observed that instructions provided by the instruments were comprehensive and understandable without change as reported by patients while the response choice and recall period provided in the questionnaire were pertinent/suitable for patients with AL amyloidosis. SF-36v2 health survey has been validated across countries around the world and in various conditions. However, no literature was identified that assessed the reliability and responsiveness of the SF-36v2 questionnaire in patients with AL amyloidosis.
No literature was identified that presented information related to MIDs in patients with AL amyloidosis.
Möller and Sartipy (2011) conducted a prospective, population-based, cohort study of Swedish patients who had lung surgery and assessed HRQoL using the Swedish version of the SF-36.48 Overall, 166 patients completed the questionnaire before as well as 6 months and 2 years after lung surgery. In total, 69.9% of patients were reported as having stage I to III tumours, 23.5% had received adjuvant chemotherapy, and 4.2% had adjuvant radiotherapy. After 2 years, the patients were compared to an age- and sex-matched reference population. Mean (SD) PCS scores decreased from baseline to 6 months post-operation and were similar at 2 years (47.4 [10.3], 41.1 [10.5], and 41.4 [11.6], respectively). The patient PCS score was lower at 2 years compared to the reference population (44.0 [3.9]; P = 0.013). Mean (SD) MCS scores increased for the same 3 timepoints from 40.5 [13.8] to 44.6 [12.6] at 6 months to 46.9 [12.2] at 2 years but were also lower than the reference population (50.6 [1.7]; P = 0.01).
In a 2014 study by Zhang et al., 317 Chinese patients with lung cancer were given the SF-36 and the European Organization for Research and Treatment of Cancer Quality of Life Questionnaire (EORTC QLQ) Core-30 (C30) and supplemental Lung Cancer-13 module (LC13).49 While the C30 measures HRQoL in relation to cancer, the LC13 focuses on symptoms and side effects specific to lung cancer and treatment, and both instruments have been validated.49,50 The 2 EORTC QLQs administered together were denoted as the LC43.49 Pearson’s correlation coefficients were calculated between the SF-36 and LC43 and were found to be highest among similar subscales such as 0.675 for PF on either questionnaire, 0.621 for PF of the SF-36 and role functioning of the LC43, 0.567 for both pain subscales, and 0.537 for SF on either questionnaire. Most of the other correlation coefficients were less than 0.5 which could be due to the SF-36 being a measure of general health and not being disease-specific like the LC43.
Chiu et al. assessed 369 patients in Taiwan who received surgical resection for hepatocellular carcinoma to estimate a MID for the SF-36.51 Using a distribution-based method (one-half SD), they suggested 3.6 points for the PCS and 4.2 points for the MCS. In a separate study of 528 prostate cancer survivors, Jayadevappa estimated MIDs for individual items of the SF-36 using both distribution-based methods (one-half and one-third SD) and anchor-based methods (baseline global health and patient-reported symptoms).52 Mean MIDs using distribution-based methods ranged from 7.0 to 16.1 points while mean estimates from anchor-based methods ranged from 5.0 to 12.4 points. The low and high ends of the ranges were for mental health and role physical items, respectively.
Pharmacoeconomic Review
Abbreviations
AE
adverse event
AL
light chain
BIA
budget impact analysis
CR
complete response
CyBorD
cyclophosphamide, bortezomib, and dexamethasone
DCyBorD
daratumumab in combination with cyclophosphamide, bortezomib, and dexamethasone
ICER
incremental cost-effectiveness ratio
IPD
individual patient data
MOD-PFS
major organ deterioration progression-free survival
NR
no response
OS
overall survival
PR
partial response
QALY
quality-adjusted life-year
RDI
relative dose intensity
SC
subcutaneous
VGPR
very good partial response
WTP
willingness to pay
Executive Summary
The executive summary comprises 2 tables (Table 1 and Table 2) and a conclusion.
Item | Description |
|---|---|
Drug product | Daratumumab (Darzalex SC), 1,800 mg/15 mL (120 mg/mL) solution for subcutaneous injection |
Submitted price | $7,310.00 per 1,800 mg vial |
Indication | In combination with bortezomib, cyclophosphamide, and dexamethasone for the treatment of adult patients with newly diagnosed light chain amyloidosis |
Health Canada approval status | NOC |
Health Canada review pathway | Priority review (Project ORBIS) |
NOC date | April 12, 2021 |
Reimbursement request | As per indication |
Sponsor | Janssen Inc. |
Submission history | Previously reviewed: No |
NOC = Notice of Compliance; SC = subcutaneous.
Table 2: Summary of Economic Evaluation
Component | Description |
|---|---|
Type of economic evaluation | Cost-utility analysis Decision tree followed by Markov model |
Target population | Adult patients with newly diagnosed AL amyloidosis |
Treatments | Daratumumab SC in combination with bortezomib, cyclophosphamide, and dexamethasone |
Comparators | CyBorD |
Perspective | Canadian publicly funded health care payer |
Outcomes | QALYs, life-years |
Time horizon | Lifetime (35 years) |
Key data source |
|
Submitted results | ICER = $70,201 per QALY for DCyBorD vs. CyBorD (incremental QALYs: 1.79; incremental costs: $125,956) |
Key limitations |
|
CADTH reanalysis results |
|
AL = light chain; CyBorD = cyclophosphamide, bortezomib, and dexamethasone; DCyBorD = daratumumab in combination with cyclophosphamide, bortezomib, and dexamethasone; ICER = incremental cost-effectiveness ratio; OS = overall survival; QALY = quality-adjusted life-year; RDI = relative dose intensity; SC = subcutaneous; vs. = versus.
Conclusions
The CADTH Clinical Review noted that, in the ANDROMEDA trial, daratumumab subcutaneous (SC) in combination with cyclophosphamide, bortezomib, and dexamethasone (DCyBorD) resulted in a significantly higher frequency of hematologic complete response (CR) compared with cyclophosphamide, bortezomib, and dexamethasone (CyBorD) alone, in patients with newly diagnosed light chain (AL) amyloidosis. Hematologic CR was deeper and occurred more rapidly in the DCyBorD arm. Results based on key secondary outcomes — including major organ deterioration progression-free survival (MOD-PFS), organ response, and time to organ response — further demonstrate the superior treatment effects of DCyBorD compared to CyBorD alone. The clinical experts consulted by CADTH for the clinical review noted that the importance of a rapid and deep hematologic response means these results are noteworthy.
CADTH identified several limitations involving the end-stage organ failure health state, use of relative dose intensities (RDIs), overall survival (OS) results, and treatment duration. Key survival parameters within the sponsor’s model were derived from a retrospective cohort study, which cannot control for unobserved characteristics that may be meaningful to survival and had a low number of long-term observations. As part of the reanalysis, CADTH assumed RDIs of 100% for all drugs, but was not able to address any of the other identified limitations. Based on the CADTH base case, DCyBorD was associated with an incremental cost-effectiveness ratio (ICER) of $67,484 per quality-adjusted life-year (QALY) compared to CyBorD, and the probability of cost-effectiveness at a $50,000 willingness-to-pay (WTP) threshold was 31.6%. A price reduction of 21% would be required to achieve cost-effectiveness at this threshold. The CADTH reanalysis results were similar to those of the sponsor’s submitted base case.
Uncertainties surrounding treatment duration and the end-stage organ failure health state remain in the analysis. The scenario involving maximum treatment duration resulted in an ICER of $88,004 per QALY and the scenario assuming half of patients with end-stage disease would require hemodialysis resulted in an ICER of $80,954 per QALY. This analysis should be interpreted with this sensitivity in mind, as they suggest that incremental costs may be underestimated and biased in favour of DCyBorD. Given the presence of uncertain estimates of both incremental costs and incremental QALYs, an additional price reduction may be warranted.
Stakeholder Input Relevant to the Economic Review
This section is a summary of the feedback received from the patient groups, registered clinicians, and drug plans that participated in the CADTH review process.
Myeloma Canada, a non-profit organization focused on research and support for patients with multiple myeloma, provided patient input in response to CADTH’s call. Myeloma Canada distributed a survey via email and social media in collaboration with the Leukemia & Lymphoma Society of Canada and received 12 eligible responses from Canadian patients with AL amyloidosis. The most important treatment goals for patients were disease control, improved quality of life, and prolonged life. The patients currently receiving CyBorD noted that side effects ranged from “somewhat intolerable” to “very tolerable,” with fatigue and neuropathy being the least tolerable side effects. Patients on CyBorD indicated that cardiac failure and neuropathy were the most important side effects to avoid. Three patients had experience with DCyBorD, with all describing the treatment as effective at controlling their AL amyloidosis. Overall, side effects received ratings between “somewhat tolerable” and “very tolerable,” with neutropenia, breathing difficulties, and cardiac failure considered the least tolerable side effects. One patient reported being treated only with daratumumab and dexamethasone as the side effects of bortezomib and cyclophosphamide were intolerable.
CADTH received registered clinician input from 2 clinician groups for this review: the Canadian Myeloma Research Group and the Ontario Health (Cancer Care Ontario) Hematology Cancer Drug Advisory Committee (OH-CCO). Clinicians noted that, given the rarity of AL amyloidosis, there are currently no formal Health Canada–approved therapies. Treatment patterns are similar to those for patients with multiple myeloma, with CyBorD emerging as the standard of care for front-line treatment of AL amyloidosis. Autologous stem cell transplant may be considered in a minority of patients, and lenalidomide plus dexamethasone may be used for second-line therapy, although there are significant side effects. The drug under review, DCyBorD, is expected to become the standard first-line therapy for patients with AL amyloidosis, and clinicians advocated for its use in second-line settings in patients who have already relapsed.
Drug plan input was received for this review. Drug plans were interested in the possibility of re-treatment with DCyBorD in patients who had already received the regimen but had relapsed. The plans asked if an equivalent IV dosing for daratumumab would be appropriate for these patients. The plans also inquired about whether patients currently being treated with CyBorD should switch to DCyBorD as soon as it becomes available. Regarding implementation, the plans noted that red blood cell genotyping is recommended before the initiation of therapy, and that variation in the dosing frequency of regimens containing daratumumab could lead to errors. Generic bortezomib is available. Finally, the plans expressed concern about the large budget impact for a relatively small number of patients.
Two of these concerns were addressed in the sponsor’s model:
The price for generic bortezomib was included in the sponsor’s model.
Patients were assumed to eventually progress to second-line therapy consisting of lenalidomide and dexamethasone.
In addition, CADTH was able to address these concerns as follows:
The price for generic bortezomib was included in CADTH’s cost comparison table (Table 8).
CADTH was unable to address the following concern raised from stakeholder input:
Neuropathy was not included as an adverse event in the sponsor’s model.
Economic Review
The current review is for daratumumab (Darzalex SC) in combination with bortezomib, cyclophosphamide, and dexamethasone for the treatment of adult patients with newly diagnosed AL amyloidosis.
Economic Evaluation
Summary of Sponsor’s Economic Evaluation
Overview
The sponsor submitted a cost-utility analysis of DCyBorD compared to CyBorD, for the treatment of adult patients with newly diagnosed AL amyloidosis. The modelled population aligned with the Health Canada indication and reimbursement request.1
Daratumumab is a monoclonal antibody that is available in both IV and SC formulations. As many patients with AL amyloidosis are at risk of fluid volume overload, the SC formulation is used in this population, at a cost of $7,310.00 for each vial of 1,800 mg/15 mL (120 mg/mL) in solution.2 Daratumumab is administered in 28-day cycles, with the recommended dose being weekly in cycles 1 and 2, biweekly in cycles 3 through 6, and once in cycles 7 and beyond. Daratumumab should be given until disease progression or for a maximum of 24 cycles (i.e., 2 years).1 Daratumumab is given in combination with CyBorD, which is the current standard of care for AL amyloidosis. Although CyBorD is used off-label in this population, treatment patterns and dosing are similar to those for patients with multiple myeloma. The recommended dosages for the CyBorD regimen according to the ANDROMEDA trial3 are as follows: 1.3 mg/m2 of bortezomib subcutaneously, 300 mg/m2 of oral cyclophosphamide, and 40 mg of oral dexamethasone once weekly. CyBorD can be given for a maximum of 6 cycles (28 days each). Thus, patients on the DCyBorD regimen could potentially receive daratumumab monotherapy for an additional 18 cycles after completion of the CyBorD regimen. Given the variable dosing of daratumumab, the 28-day cost also varies for DCyBorD. In the sponsor’s model, the cost per 28-day cycle of DCyBorD was calculated to be $31,888 in cycles 1 and 2, $17,268 in cycles 3 to 6, and $7,310 (the cost of 1 vial of daratumumab SC) in cycles 7 and beyond.
The comparator in this analysis was the CyBorD regimen, which has the same recommended doses as described previously. CyBorD is given for a maximum of 6 cycles, with a 28-day cycle cost of $2,648, according to the sponsor’s model. Wastage was assumed to occur for the components of the CyBorD regimen; however, as the dose of daratumumab is the same as the vial size (1,800 mg), wastage was not relevant for this drug.
The submitted model reported both QALYs and life-years over a lifetime horizon (35 years). The base-case analysis was conducted from the perspective of the Canadian public health care system, with discounting of 1.5% per annum applied to both costs and outcomes.
Model Structure
The sponsor’s model consisted of a decision tree followed by a Markov model (Figure 1). A hypothetical cohort of 1,000 patients enter the model on treatment, and the decision tree is used to stratify patients by their hematologic response to treatment: complete response (CR), very good partial response (VGPR), partial response (PR) and/or no response (NR), or dead. This distribution is informed by the ANDROMEDA trial.3 Patients are assessed for their response after 3 cycles (months) of first-line treatment, after which point they exit the decision tree and enter the Markov model, through which they progress in a linear manner. Patients achieving a CR or VGPR are assumed to continue therapy for another 3 cycles (to complete the maximum 6 cycles of CyBorD) and then move to an “off-treatment/fixed daratumumab treatment (FDT)” health state. Patients in this state who are in the DCyBorD arm can continue daratumumab monotherapy for an additional 18 cycles (to a maximum of 24) while patients in the CyBorD arm do not receive chemotherapy and are monitored for disease progression. Upon disease progression, patients will transition to the “second-line treatment” state and will receive chemotherapy as second-line treatment, and can progress further to “end-stage organ failure.” Patients can remain in this health state or die, as well as die at any of the earlier states. Patients achieving a PR or NR are assumed not to have responded and transition immediately to the second-line treatment health state; they do not continue on DCyBorD or CyBorD. These patients can then progress to end-stage organ failure and/or death. The cycle length is 28 days with a half-cycle correction applied.
Model Inputs
The model assumed a hypothetical cohort of 1,000 patients with baseline characteristics aligned with the population enrolled in the ANDROMEDA trial,3 which is consistent with the Health Canada indication and reimbursement request. This phase III, open-label trial enrolled 388 adult patients from 22 countries, and all patients had a histopathologic diagnosis of systemic AL amyloidosis and measurable hematologic disease.3 The mean age of the population was 63.1 years and 58% of patients were male.4
Comparative data for this model were informed by the ANDROMEDA trial, in which patients on DCyBorD and CyBorD achieved different levels of hematologic response. Data from ANDROMEDA outlined patients’ hematologic response after 3 cycles of treatment and informed the distribution of CR, VGPR, PR or NR, and patients who died in the decision tree.3 The model assumed that hematologic response dictated the transition probabilities elsewhere, regardless of treatment received. As mentioned, patients achieving CR or VGPR would stay on first-line treatment for 3 cycles after the decision tree, after which point they could transition to second-line treatment, end-stage organ failure, or death. The transition probabilities from first-line treatment to end-stage organ failure were calculated using individual patient data (IPD) from ANDROMEDA for the MOD-PFS outcome, which included end-stage cardiac and/or renal failure, and hematologic progressive disease.4 The transition probabilities between first-line and second-line treatment were based on IPD of time to subsequent non–cross resistant anti–plasma cell therapy in ANDROMEDA stratified by CR or VGPR responses.4 The transition probabilities between second-line treatment and end-stage organ failure were assumed to be the same as the transitions from first-line treatment to end-stage organ failure.
Patients could experience death from any state, and the transition to death was governed by OS data that overrode the other transition probabilities. The OS data from ANDROMEDA were not mature enough for extrapolation, leading the sponsor to obtain OS data from the published literature. Kaplan-Meier OS curves from Kastritis et al. (2020), a retrospective study from Greece, were extrapolated and used to estimate the OS of patients achieving CR, VGPR, and PR or NR.5 Based on model-fit statistics and visual assessment, an exponential model was used for CR, a log-logistic model was used for VGPR, and a Weibull model was used for the blended PR and NR data.2 These data informed the rate of death by hematologic response, and the health state distribution of these deaths was based on IPD from ANDROMEDA for the deaths observed during the OS follow-up (11.4 months).4 For all patients, OS data were capped by the general population mortality risk, derived from Statistics Canada reports and weighted for sex and age.6
Dosing for daratumumab and CyBorD has already been described and is consistent with the ANDROMEDA trial. All patients in the DCyBorD arm who remained alive and progression-free were assumed to continue daratumumab monotherapy for 24 cycles (i.e., no discontinuation for reasons other than progression). All patients in the CyBorD arm who remained alive and progression-free were assumed to receive 6 cycles of treatment. For both arms, patients achieving PR or NR were assumed to discontinue treatment at 3 months (the decision tree cut-off) and transition to second-line treatment.
Health-related quality-of-life data were collected from ANDROMEDA through the EQ-5D-5L questionnaire. The EQ-5D utilities for CR and for PR or NR were 0.759 and 0.732, respectively, and were derived directly from the collected data. As the utility value for VGPR did not meet face validity, it was calculated as the average of the utility values for CR and PR (0.750).2 Utility decrements for the health states of second-line treatment and end-stage organ failure were applied on a recurring per-cycle basis for as long as the patient remained in the health state. A utility decrement of 0.067 for patients in the second-line treatment health state was applied, calculated as the difference between the mean baseline utility score and the mean utility value associated with “progressive disease” from IPD from ANDROMEDA.2 For patients with end-stage organ failure, a health-related quality-of-life estimate from the published literature on patients with advanced chronic heart failure was used to calculate a utility decrement of 0.231.7 An additional decrement of 0.1 derived from a review of chronic kidney disease was applied to patients on hemodialysis, which the base case assumed to be 100% of patients with end-stage organ failure.8 The model considered adverse events (AEs), including cardiac failure, diarrhea, edema, hypokalemia, lymphopenia, neutropenia, pneumonia, and syncope, the frequencies of which were derived from ANDROMEDA for the DCyBorD and CyBorD arms.4 Utility decrements ranging from 0.0039 for syncope to 0.2 for pneumonia were derived from the published literature and were applied for the duration of the AE according to the Ontario Case Costing Initiative.2,9
Drug acquisition costs were calculated based on the unit drug costs and dosage received, with RDIs from ANDROMEDA applied to the calculation. In the DCyBorD arm, RDIs of 96.9%, 92.9%, 84.7%, and 94.4% were applied for daratumumab, bortezomib, cyclophosphamide, and dexamethasone, respectively. In the CyBorD arm, RDIs of 95.1%, 85.4%, and 96.0% were applied for bortezomib, cyclophosphamide, and dexamethasone, respectively.4 Administration costs of $55.18 and $51.70 were applied per SC injection of daratumumab and bortezomib, respectively, based on published data on nursing and overhead costs.2 A pharmacy dispensing fee of $10.09 per cycle was applied for the oral drugs cyclophosphamide and dexamethasone. Co-medication costs of $73.36 and $71.12 were applied to patients on DCyBorD and CyBorD, respectively, based on the co-medications received by patients in ANDROMEDA, which included acyclovir, diphenhydramine, dexamethasone, montelukast, methylprednisolone, and paracetamol.4 Unit costs for these products were derived from the Ontario Drug Benefit Formulary.10 Disease monitoring costs were applied to patients in the first-line and daratumumab monotherapy treatment states, and consisted of hematologist visits and assessments and routine laboratory tests. The frequency of this disease monitoring was derived from ANDROMEDA, and the unit costs were derived from the Ontario Schedule of Benefits for physician and laboratory services, as well as the published literature.4,11,12 The costs per cycle for disease monitoring were $424.51, $178.08, and $127.35 for patients on first-line, daratumumab monotherapy, and off-treatment, respectively. Costs associated with AEs ranging from $417.41 for edema to $9,858.48 for hypokalemia were derived from the Ontario Case Costing Initiative and published sources and were included as a 1-time cost weighted by the proportion of patients experiencing each AE.2,9 Patients progressing to second-line therapy were assumed to receive lenalidomide and dexamethasone, and incurred a 1-time cost for subsequent therapy of $53,451.41 based on 6 cycles of second-line therapy.13 End-stage organ failure was associated with a per-cycle cost of $5,492.16, based on 12 sessions of hemodialysis costing $457.68 each.14 In addition, health care resource use costs were included for patients requiring emergency room, inpatient, or outpatient visits, the unit costs of which were $187.08, $6,363.50, and $80.25, respectively, based on published reports.2 The frequency of this health care resource use was based on an unpublished burden-of-illness study commissioned by the sponsor and another published Canadian source.2,15 Finally, patients transitioning into the death state were assumed to incur a 1-time end-of-life-care cost of $5,650.66, which was based on the end-of-life costs for patients with heart failure.15
Summary of Sponsor’s Economic Evaluation Results
The sponsor submitted probabilistic analyses for the base-case and scenario analyses based on 3,000 simulations. The probabilistic findings are presented in Table 3. Probabilistic results were higher than deterministic results.
Base-Case Results
Over a lifetime horizon, DCyBorD was associated with incremental costs of $125,956 and QALYs of 1.79 in comparison with CyBorD, resulting in an ICER of $70,201 per QALY (Table 3). In the sponsor’s base case, DCyBorD had a 31.5% probability of being cost-effective at a WTP threshold of $50,000 per QALY.
Table 3: Summary of the Sponsor’s Economic Evaluation Results
Drug | Total costs ($) | Incremental costs ($) | Total QALYs | Incremental QALYs | ICER vs. reference ($ per QALY) |
|---|---|---|---|---|---|
CyBorD | 209,732 | Reference | 3.76 | Reference | Reference |
DCyBorD | 335,682 | 125,956 | 5.56 | 1.79 | 70,201 |
CyBorD = cyclophosphamide, bortezomib, and dexamethasone; DCyBorD = daratumumab in combination with cyclophosphamide, bortezomib, and dexamethasone; ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year; vs. = versus.
Source: Sponsor’s pharmacoeconomic submission.2
Sensitivity and Scenario Analysis Results
The sponsor conducted a number of scenario analyses involving discount rates, a shortened time horizon, alternative OS extrapolations, a 6-month exit from the decision tree, treatment duration, and mortality assumptions. The analysis involving a 20-year time horizon resulted in an ICER of $84,205. The analysis that assumed all patients received the maximum cycles of treatment (24 for daratumumab and 6 for CyBorD) resulted in an ICER of $93,002. The ICER resulting from the 6-month exit from the decision tree was $56,206 as the OS data in this scenario were derived from a different source.16 The scenario analyses involving alternative OS extrapolations had little effect on the ICER, as did the other scenarios involving the mortality distribution, utilities, and drug wastage.
CADTH Appraisal of the Sponsor’s Economic Evaluation
CADTH identified several key limitations to the sponsor’s analysis that have notable implications on the economic analysis:
Uncertainty surrounding the end-stage organ failure health state in this population: The sponsor made several assumptions while deriving the transition probabilities, costs, and utilities associated with end-stage organ failure that are not all aligned with one another and are associated with uncertainty. First, the transition probabilities to the end-stage organ failure health state from first- and second-line treatment were calculated based on the MOD-PFS outcome, which included end-stage cardiac and/or renal failure, and hematologic progressive disease. In its report the sponsor commented that “ideally, the transition probabilities would be based strictly on events pertaining to cardiac or renal failure … Although a potential limitation of using MOD-PFS is the risk of overestimating the transition probabilities to ‘End-stage Organ Failure,’ this was considered a simplistic assumption implemented due to data immaturity.”2 Although CADTH appreciates that the low number of events makes the incorporation of MOD-PFS methodologically challenging, this assumption appears to benefit the sponsor because the rate of hematologic progressive disease is higher in the CyBorD arm.4 This likely biases the results in favour of DCyBorD because the transition probabilities to end-stage organ failure would be inflated in the CyBorD arm. Furthermore, the clinical experts consulted by CADTH for this review noted that MOD-PFS is a novel outcome that has not yet been validated in other studies or by real-world evidence. Thus, use of this outcome to calculate transition probabilities is associated with uncertainty.
Second, the utility decrement of 0.231 applied to end-stage organ failure was based on a publication pertaining to advanced chronic heart failure.7 As this paper was focused on heart failure specifically, this utility decrement may not necessarily reflect the reduced quality of life associated with other aspects of end-stage organ failure, such as renal failure or hematologic progressive disease, all of which were used to define the transition probability into this state.
Third, the sponsor has assumed that 100% of patients in the end-stage organ failure health state will be on hemodialysis, which is associated with a recurring per-cycle cost of $5,492.16. This assumption does not align with the prior 2 assumptions, in which the transition into this health state is estimated by the rate of both cardiac and renal failure, and the utility is modelled after that of chronic heart failure. This assumption also does not align with the view of the clinical experts, who described 3 main types of end-stage organ failure that could be expected in this disease: cardiac involvement only, kidney involvement only, and end-stage failure of both organs (cardiorenal syndrome). While it may be reasonable to assume that 100% of patients with kidney involvement will be on hemodialysis, there is a proportion of patients with cardiac involvement alone that would not be expected to receive hemodialysis. The clinical experts were unable to provide estimates for the proportion of patients with end-stage cardiac involvement only for use in reanalysis. Consequently, the total costs of CyBorD are likely overestimated, creating a bias that favours DCyBorD.
CADTH was unable to address the uncertainty resulting from these assumptions in reanalysis. CADTH tested alternative assumptions about costs and utilities pertaining to end-stage organ failure in scenario analyses.
Uncertain generalizability of OS results: While most of the inputs in the model were based on the ANDROMEDA trial, OS data from a retrospective study were used to inform the probability of survival beyond 3 months based on hematologic response.5 CADTH requested that the sponsor provide a model in which OS data were derived directly from ANDROMEDA, but this was deemed infeasible due to the immaturity of the OS data. At the time of the primary analysis, 86% of patients were still alive at a median follow-up of 11.4 months, and long plateaus were observed in the Kaplan-Meier data.4 CADTH agreed with the sponsor that any extrapolation of such data would be implausible and associated with a high degree of uncertainty.
The CADTH Clinical Review evaluated the similarity between the Kastritis et al. (2020) and ANDROMEDA populations.4,5 This appraisal noted that the baseline characteristics between the 2 populations were generally similar, except for some potentially important differences in the proportion of patients within each cardiac stage. The sponsor emphasized that the Kastritis et al. (2020) population included more patients classified as Mayo stage IIIB compared with the ANDROMEDA study, suggesting this was a conservative assumption for OS; however, a clinical reviewer noticed a higher proportion of Mayo stage IIIA in Kastritis et al. (2020) compared to ANDROMEDA (34.5% versus 18%).4,5 Because all-cause mortality is expected to vary with cardiac stage, the extent to which these differences in baseline Mayo stage may have affected OS is uncertain. The clinical experts consulted by CADTH for this review agreed with the assessment that the use of the retrospective study was appropriate.
The use of the Kastritis et al. study nevertheless contributes meaningful uncertainty to the economic evaluation. While the study cohort was similar in observed characteristics to those of the ANDROMEDA population, unobserved differences may yet exist and play an unknown role in survival. The retrospective nature of the study design means that unobserved characteristics could not be controlled for as they would have been if long-term survival data had been available from the ANDROMEDA trial or another randomized trial. CADTH also noted the limits of the retrospective study’s sample size (N = 277), which resulted in a low number of observed events beyond 4 years. The inability to account for the potential effect of unobserved characteristics and the small number of observations adds meaningful uncertainty to the pharmacoeconomic model’s estimated QALYs, and therefore the cost-effectiveness results.
CADTH was unable to address this uncertainty in reanalysis. CADTH tested an alternative OS extrapolation for CR patients in a scenario analysis.
RDI does not correlate well with drug costs: The sponsor included mean RDIs ranging from 84.7% to 96.9% for every drug in the treatment regimens and multiplied these values by the respective drug acquisition costs. This approach is problematic because RDI can be influenced by many different things. For example, the dose received by a patient may differ from the full planned dose of the drug due to dose delays, missed doses, dose reductions to manage toxicity, or subsequent dose re-escalation. Each of these reasons have differing impacts on drug costs. Furthermore, it is unclear how these assumptions interact with considerations about vial size and wastage, which were incorporated into the sponsor’s calculations of the per-cycle drug costs. Last, the RDI was assumed to be 100% in the budget impact analysis (BIA). Overall, without explicitly modelling dose delays and reductions for the patient population, this method of multiplying RDI by drug acquisition costs is associated with uncertainty about the true drug cost incurred by payers.
As part of the base case, CADTH assumed an RDI of 100% for all treatments.
Uncertainty surrounding the duration of DCyBorD treatment in clinical practice: The sponsor’s base case assumed that patients would be treated for an average of 15.0 months with daratumumab (received concurrently with the first 6 cycles of CyBorD). However, updated results of the ANDROMEDA trial stated that, as of November 2020, the median treatment duration was 18.5 months for the DCyBorD arm.17 Thus, it seems probable that the drug acquisition costs for daratumumab monotherapy (i.e., in the off-treatment/fixed daratumumab treatment state) are underestimated in the sponsor’s model. This biases the results in favour of DCyBorD because the drug acquisition costs of daratumumab are underestimated.
CADTH conducted a scenario analysis assuming the maximum treatment duration for both comparators (i.e., 24 cycles for DCyBorD and 6 cycles for CyBorD).
An additional limitation was identified but not considered to be a key limitation. In its probabilistic analysis, the sponsor used standard deviations instead of standard errors to characterize uncertainty for various parameters, including age, weight, body surface area, and treatment duration. This error produced large fluctuations in these parameters, resulting in an improper characterization of parameter uncertainty, ultimately leading to probabilistic results that were consistently higher than the deterministic results. CADTH corrected this by converting the standard deviations used by the sponsor to standard errors.
Additionally, the following key assumptions were made by the sponsor and have been appraised by CADTH (Table 4).
Table 4: Key Assumptions of the Submitted Economic Evaluation Not Noted as Limitations to the Submission
Sponsor’s key assumption | CADTH comment |
|---|---|
Treatment response is assessed at 3 months, after which patients could switch therapy if not responding. | Appropriate. The use of a 3-month exit from the decision tree aligned with clinical expert opinion. |
Hematologic response is correlated with OS and is treatment-independent. | Appropriate. This assumption was validated by the clinical experts and is supported by published literature.5,16 |
Transition probabilities for second-line treatment to end-stage organ failure were assumed to be equivalent to those for first-line treatment to end-stage organ failure. | Uncertain. The small number of MOD-PFS events at the ANDROMEDA clinical cut-off date made it difficult to calculate transition probabilities between the second-line treatment and end-stage organ failure health states. |
The utility value for VGPR is the mean of the utility values for CR and PR. | Uncertain. The utility value for VGPR was calculated as such because the value derived from the EQ-5D estimates in ANDROMEDA were lower than the values for PR and NR. This lack of face validity suggests that there were issues in the collection of EQ-5D data in the trial, which has an uncertain influence on the final values obtained. |
A pharmacy dispensing fee of $10.09 was applied to each SC administration and monthly for oral drugs. | Uncertain. Dispensing fees and coverage differ by jurisdiction and, as such, this fee may not be reflective of the Canadian publicly funded health care payer perspective. |
No patients were assumed to receive organ transplant or implantation of a cardiac assist device. | Uncertain. While the clinical expert noted that organ transplants are rare in this population, patients with cardiac arrythmias or other cardiac failure could require a cardiac assist device. |
CR = complete response; MOD-PFS = major organ deterioration progression-free survival; NR = no response; OS = overall survival; PR = partial response; SC = subcutaneous; VGPR = very good partial response.
CADTH Reanalyses of the Economic Evaluation
Base-Case Results
The CADTH base case was derived by making 1 change, detailed in Table 5, which involved assuming 100% RDI for all drugs included in the model.
Table 5: CADTH Revisions to the Submitted Economic Evaluation
Stepped analysis | Sponsor’s value or assumption | CADTH value or assumption |
|---|---|---|
Corrections to sponsor’s base case | ||
1. Uncertainty around various parameters derived from ANDROMEDA | Standard deviations used to characterize uncertainty | Standard errors used to characterize uncertainties |
Changes to derive the CADTH base case | ||
1. Relative dose intensity | DCyBorD
CyBorD
| 100% for all drugs |
CADTH base case | — | Reanalysis 1 |
CyBorD = cyclophosphamide, bortezomib, and dexamethasone; DCyBorD = daratumumab in combination with cyclophosphamide, bortezomib, and dexamethasone.
In the CADTH base case, DCyBorD was associated with estimated total costs of $345,453 and total QALYs of 5.81 compared with total costs and QALYs of $215,550 and 3.89, respectively, for patients receiving CyBorD. The ICER for DCyBorD compared to CyBorD was $67,484 per QALY, and the probability of cost-effectiveness at a $50,000 per QALY WTP threshold of was 31.6%. A detailed breakdown of the disaggregate results is available in Appendix 4, Table 11. In the CADTH base case, 95% of incremental QALYs were generated during the time period beyond the length of the ANDROMEDA trial (20.4 months).
Table 6: Summary of the Stepped Analysis of the CADTH Reanalysis Results (Deterministic)
Stepped analysis | Drug | Total costs ($) | Total QALYs | ICER ($/QALY) |
|---|---|---|---|---|
Sponsor’s corrected base case | CyBorD | 222,128 | 3.74 | Reference |
DCyBorD | 350,076 | 5.72 | 64,595 | |
CADTH reanalysis 1 (RDI) and base case | CyBorD | 222,143 | 3.74 | Reference |
DCyBorD | 350,096 | 5.72 | 64,598 |
CyBorD = cyclophosphamide, bortezomib, and dexamethasone; DCyBorD = daratumumab in combination with cyclophosphamide, bortezomib, and dexamethasone; ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year; RDI = relative dose intensity.
Note: The CADTH correction to the sponsor’s base case resulted in a reduction in the probabilistic ICER. This change should be interpreted as a statistical artifact.
Scenario Analysis Results
CADTH undertook price-reduction analyses based on the sponsor’s and CADTH’s base case. Based on the CADTH base case, a price reduction of 21% would be necessary to achieve cost-effectiveness at a WTP threshold of $50,000 per QALY (Table 7).
Table 7: CADTH Price-Reduction Analyses
Analysis | ICERs for DCyBorD vs. CyBorD | |
|---|---|---|
Price reduction | Sponsor base case | CADTH reanalysis |
No price reduction | $70,201 | $67,484 |
10% | $61,439 | $59,189 |
20% | $52,678 | $50,911 |
21% | $51,802 | $49,756 |
24% | $49,173 | $47,499 |
CyBorD = cyclophosphamide, bortezomib, and dexamethasone; DCyBorD = daratumumab in combination with cyclophosphamide, bortezomib, and dexamethasone; ICER = incremental cost-effectiveness ratio; vs. = versus.
CADTH undertook a series of exploratory analyses to determine the impact of alternative assumptions on the cost-effectiveness of DCyBorD, as outlined as follows:
The maximum treatment duration was assumed for DCyBorD (i.e., 24 cycles) and CyBorD (i.e., 6 cycles).
Assumed that half of patients in end-stage organ failure would require hemodialysis.
Assumed a utility decrement of 0.041 for patients in end-stage organ failure based on the difference between the mean baseline utility value in ANDROMEDA (0.731) and a utility value estimate for hemodialysis patients (0.69).4,8
Extrapolated OS data for CR patients using a Gompertz function.
Pharmacy dispensing fees were excluded.
Combined analyses 1 and 2.
The results of these analyses are presented in Appendix 4, Table 12. The scenario analysis involving the maximum treatment duration for both comparators had the largest effect on the ICER, which was calculated to be $88,004 per QALY. The scenario analysis in which half of patients in end-stage organ failure were assumed to require hemodialysis resulted in an ICER of $80,954 per QALY. The scenario analysis combining these 2 changes resulted in an ICER of $100,036 per QALY. Other scenarios tested had minimal impact on the ICER.
Issues for Consideration
Clinical experts consulted by CADTH indicated patients might be re-treated with daratumumab in a relapsed setting if they had previously experienced a positive response. This suggestion was also noted by the clinical input received, in which clinicians indicated a desire to use DCyBorD or daratumumab add-on therapy in any patient with relapsed AL amyloidosis. The effectiveness of such treatment or re-treatment in a relapsed setting, and therefore the cost-effectiveness, is unknown, but will lead to increased cost to drug programs as a result of this potential scope creep.
Overall Conclusions
The CADTH Clinical Review noted that, in the ANDROMEDA trial, daratumumab SC in combination with CyBorD resulted in significantly higher frequency of hematologic CR than CyBorD alone in patients with newly diagnosed AL amyloidosis. Hematologic CR was deeper and occurred more rapidly in the DCyBorD arm. The results based on key secondary outcomes, including MOD-PFS, organ response, and time to organ response, further demonstrate superior treatment effects of DCyBorD compared to CyBorD alone. According to the clinical experts consulted for the review, the importance of a rapid and deep hematologic response means these results are noteworthy. These results are incorporated into the pharmacoeconomic model by means of a decision tree, which separates patients by hematologic response. Patients with CR are assumed to have a low mortality, and downstream effects such as end-stage organ failure and hemodialysis are avoided.
CADTH identified several limitations with the sponsor’s analysis involving uncertainty about the end-stage organ failure health state, use of RDIs, OS results, and treatment duration. As part of the reanalysis, CADTH assumed RDIs of 100% for all drugs. Based on the CADTH base case, DCyBorD was associated with an ICER of $67,484 per QALY compared to CyBorD, and the probability of cost-effectiveness at a $50,000 WTP threshold was 31.6%. A price reduction of 21% would be required to achieve cost-effectiveness at this threshold. The CADTH reanalysis results were similar to those of the sponsor’s submitted base case.
Uncertainties surrounding treatment duration and the end-stage organ failure health state remain in the analysis. The scenario involving maximum treatment duration resulted in an ICER of $88,004 per QALY and the scenario assuming half of patients with end-stage disease would require hemodialysis resulted in an ICER of $80,954 per QALY. A scenario analyses combining these 2 changes resulted in an ICER of $100,036 per QALY. The results of this analysis should be interpreted with this sensitivity in mind because they suggest that incremental costs may be underestimated and biased in favour of DCyBorD. Thus, CADTH concludes that the reanalyzed ICER is likely underestimated, with the true ICER lying somewhere between the reanalysis and these scenario analyses. Although other scenarios involving OS modelling for CR patients, the utility of end-stage organ failure, and pharmacy dispensing fees were explored, all had minimal effect on the overall ICER.
The evaluation of the cost-effectiveness of DCyBorD was based on survival estimates derived from a retrospective cohort study conducted in Greece.5 CADTH’s review of this evidence found that the observed characteristics of this cohort were similar to those of ANDROMEDA trial participants, and concluded that it was an appropriate source from which to derive long-term survival estimates. However, the study design’s inability to control for unobserved characteristics and the small number of long-term observations in the retrospective study contribute additional uncertainty to CADTH’s estimate of the incremental effectiveness and, consequently, the ICER. This uncertainty could not be addressed or estimated through reanalysis. The presence of meaningful uncertainty in both the costs and long-term outcomes suggests that an additional price reduction may be warranted.
References
1.Darzalex (daratumumab for injection): 20 mg/mL concentrate for solution for infusion; antineoplastic, monoclonal antibody [product monograph]. Toronto (ON): Janssen Inc.; 2021 Jun 30.
2.Pharmacoeconomic evaluation [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Darzalex SC (daratumumab), 120mg/mL solution for subcutaneous injection. Toronto (ON): Janssen Inc.; 2021 Jul 28.
3.Kastritis E, Palladini G, Minnema M, et al. Daratumumab-based treatment for immunoglobulin light-chain amyloidosis. NEJM. 2021;385(1):46-58. PubMed
4.Clinical Study Report: JNJ-54767414AMY3001. A randomized phase 3 study to evaluate the efficacy and safety of daratumumab in combination with cyclophosphamide, bortezomib and dexamethasone (CyBorD) compared with CuBorD in newly diagnosed systemic AL amyloidosis [internal sponsor's report]. Toronto (ON): Janssen Research & Development; 2020 Aug 18.
5.Kastritis E, Fotiou D, Theodorakakou F, et al. Timing and impact of a deep response in the outcome of patients with systemic light chain (AL) amyloidosis. Amyloid. 2020;28(1):3-11. PubMed
6.Table: 13-10-0114-01. Life expectancy and other elements of the life table, Canada, all provinces except Prince Edward Island. Ottawa (ON): Statistics Canada; 2020: https://www150.statcan.gc.ca/t1/tbl1/en/tv.action?pid=1310011401. Accessed 2021 Sep 16.
7.Emin A, Rogers C, Banner N, UKCTA SG. Quality of life of advanced chronic heart failure: medical care, mechanical circulatory support and transplantation. Eur J Cardiothorac Surg. 2016;50(2):269-273. PubMed
8.Wyld M, Morton R, Hayen A, Howard K, Webster A. A systematic review and meta-analysis of utility-based quality of life in chronic kidney disease treatments. PLoS Med. 2012;9(9):e1001307. PubMed
9.Ontario Case Costing Initiative (OCCI). Toronto (ON): Ontario Health and Long-Term Care; 2017: https://data.ontario.ca/dataset/ontario-case-costing-initiative-occi. Accessed 2021 Sep 16.
10.Ontario Ministry of Health, Ontario Ministry of Long-Term Care. Ontario drug benefit formulary/comparative drug index. 2021; https://www.formulary.health.gov.on.ca/formulary/. Accessed 2021 Sep 16.
11.Schedule of benefits for laboratory services: effective July 1, 2020. Toronto (ON): Ontario Ministry of Health; 2020: https://www.health.gov.on.ca/en/pro/programs/ohip/sob/lab/lab_mn2020.pdf. Accessed 2021 Sep 16.
12.Schedule of benefits for physician services under the Health Insurance Act: effective April 1, 2020. Toronto (ON): Ontario Ministry of Health; 2020: https://www.health.gov.on.ca/en/pro/programs/ohip/sob/physserv/sob_master20200306.pdf. Accessed 2021 Sep 16.
13.Sanchorawala V, Wright D, Rosenzweig M, Finn K, Fennessey S. Lenalidomide and dexamethasone in the treatment of AL amyloidosis: results of a phase 2 trial. Blood. 2007;109(2):492-496. PubMed
14.Ferguson T, Whitlock R, Bamforth R, Beaudry A, Darcel J. Cost-utility of dialysis in Canada: hemodialysis, peritoneal dialysis, and nondialysis treatment of kidney failure. Kidney Med. 2021;3(1):20-30. PubMed
15.Kaul P, McAlister F, Ezekowitz J, Bakal J, Curtis L. Resource use in the last 6 months of life among patients with heart failure in Canada. Arch Intern Med. 2011;171(3):211-217. PubMed
16.Palladini G, Dispenzieri A, Gertz M, Kumar A, Wechalekar A. New criteria for response to treatment in immunoglobulin light chain amyloidosis based on free light chain measurement and cardiac biomarkers: impact on survival outcomes. J Clin Oncol. 2012;30(36):4541-4549. PubMed
17.Kastritis E, Sanchorawala S, Merlini G. Subcutaneous daratumumab + bortezomib, cyclophosphamide, and dexamethasone (VCd) in patients with newly diagnosed light chain (AL) amyloidosis: updated results from the phase 3 ANDROMEDA study. J Clin Oncol. 2021;15_suppl:8003.
18.Apo-dexamethasone (dexamethasone tables USP): 0.5 mg and 4 mg; corticosteroid [product monograph]. Weston (ON): Apotex Inc.; 2020 May 26.
19.Bortezomib for injection: 3.5 mg / vial bortezomib, as the mannitol boronic ester; antineoplastic agent [product monograph]. Mississauga (ON): Dr. Reddy's Laboratories Canada Inc.; 2020 May 19.
20.Procytox (cyclophosphamide tablets USP): 25 mg, 50 mg; cyclophosphamide for injection: 200 mg, 500 mg, 1000 mg, 2000 mg (powder for injection) per vial; antineoplastic agent [product monograph]. Mississauga (ON): Baxter Corporation; 2012 Sep 7.
21.DeltaPA. [Ottawa (ON)]: IQVIA; 2021: https://www.iqvia.com/. Accessed 2021 Sep 16.
22.Statistics Canada. Table 17-10-0005-01. Population estimates on July 1st, by age and sex. 2021; https://www150.statcan.gc.ca/t1/tbl1/en/cv.action?pid=1710000501. Accessed May 25, 2021.
23.Procedures for CADTH reimbursement reviews. September 2021. Ottawa (ON): CADTH; 2021: https://cadth.ca/sites/default/files/Drug_Review_Process/CADTH_Drug_Reimbursement_Review_Procedures.pdf. Accessed 2021 Sep 16.
Appendix 1: Cost Comparison Table
Note this appendix has not been copy-edited.
The comparators presented in the following table have been deemed to be appropriate based on feedback from clinical experts. Comparators may be recommended (appropriate) practice or actual practice. Existing product listing agreements are not reflected in the table and as such, the table may not represent the actual costs to public drug plans.
Table 8: CADTH Cost Comparison Table for Newly Diagnosed AL Amyloidosis
Treatment | Strength/ concentration | Form (vial size if single-use) | Price | Recommended dosage | Daily cost | 28-day cycle cost |
|---|---|---|---|---|---|---|
Daratumumab (Darzalex SC) | 120 mg/mL | 1,800 mg Solution for SC injection | $7,310.0000a | Cycles 1 and 2: 1,800 mg 4 times per 28 days Cycles 3 to 6: 1,800 mg twice per 28 days Cycle 7+: 1,800 mg once per 28 days | Cycles 1 and 2: $1,044.29 Cycles 3 to 6: $522.14 Cycle 7+: $261.07 | Cycles 1 and 2: $29,240 Cycles 3 to6: $14,620 Cycle 7+: $7,310 |
Cyclophosphamide | 25 mg 50 mg | Tablet Tablet | $0.3545 $0.4773 | 300 mg/m2 4 times per 28 days | $0.80 | $22 |
Bortezomib (generic) | 2.5 mg/mL | 3.5 mg Powder for SC injection | $654.3100b | 1.3 mg/m2 4 times per 28 days | $93.47 | $2,617 |
Dexamethasone | 0.5 mg 4 mg | Tablet Tablet | $0.1564 $0.3046 | 40 mg 4 times per 28 days | $0.44 | $12 |
DCyBorD regimen (CyBorD for cycles 1 to 6) | — | Cycles 1 and 2: $1,138.99 Cycles 3 to 6: $616.85 Cycle 7+: $261.07 | Cycles 1 and 2: $31,892 Cycle 3 to 6: $17,272 Cycle 7+: $7,310 | |||
CyBorD regimen (maximum 6 cycles) | — | $94.71 | $2,652 | |||
CyBorD = cyclophosphamide, bortezomib, and dexamethasone; DCyBorD = daratumumab in combination with cyclophosphamide, bortezomib, and dexamethasone; SC = subcutaneous.
Note: All prices are from the Ontario Drug Benefit Formulary (accessed September 16, 2021),10 unless otherwise indicated, and do not include dispensing fees. Dosing information according to the ANDROMEDA trial,3 from which a mean body surface area of 1.84 m2 was used, with vial sizes from the respective product monographs.18-20
aSponsor submitted price.2
bPrice from IQVIA DeltaPA database.21
Appendix 2: Submission Quality
Description | Yes/No | Comments |
|---|---|---|
Population is relevant, with no critical intervention missing, and no relevant outcome missing | Yes | No comment. |
Model has been adequately programmed and has sufficient face validity | Yes | No comment. |
Model structure is adequate for decision problem | Yes | No comment. |
Data incorporation into the model has been done adequately (e.g., parameters for probabilistic analysis) | No | The probabilistic results were consistently higher than the deterministic results in all reanalyses and scenario analyses, suggesting some parameters may be skewed despite CADTH reanalysis. |
Parameter and structural uncertainty were adequately assessed; analyses were adequate to inform the decision problem | No | The sponsor incorrectly used standard deviations rather than standard errors in their characterization of the uncertainty around various inputs from the ANDROMEDA trial such as patient age, weight, body surface area, and treatment duration. This led to large fluctuations in these parameters and the probabilistic ICER did not reflect the deterministic due to some parameters being capped at a maximum value (e.g., number of treatment cycles). |
The submission was well organized and complete; the information was easy to locate (clear and transparent reporting; technical documentation available in enough details) | Yes | No comment. |
ICER = incremental cost-effectiveness ratio.
Note: This table has not been copy-edited.
Appendix 3: Additional Information on the Submitted Economic Evaluation
Note this appendix has not been copy-edited.
Table 10: Detailed Results of the Sponsor’s Base Case
Parameter | DCyBorD | CyBorD | Incremental |
|---|---|---|---|
Discounted LYs | |||
Total LYs | 7.94 | 5.88 | 2.06 |
1L Tx | 0.41 | 0.35 | 0.06 |
Off Tx/FDT | 4.58 | 2.00 | 2.58 |
2L Tx | 2.13 | 2.10 | 0.03 |
End-Stage Organ Failure | 0.81 | 1.43 | −0.62 |
Discounted QALYs | |||
Total QALYs | 5.56 | 3.76 | 1.79 |
1L Tx | 0.31 | 0.26 | 0.05 |
Off Tx/FDT | 3.47 | 1.51 | 1.96 |
2L Tx | 1.45 | 1.41 | 0.03 |
End-Stage Organ Failure | 0.33 | 0.58 | −0.24 |
AE Disutility | −0.0006 | −0.0003 | −0.0002 |
Discounted costs ($) | |||
Total costs | 335,688 | 209,732 | 125,956 |
Total 1L drug therapy | 171,230 | 10,311 | 160,918 |
Total 1L drug administration | 2,388 | 884 | 1,504 |
Co-medication | 839 | 278 | 560 |
Health care resource use | 61,095 | 50,406 | 10,688 |
Adverse event management | 2,269 | 1,660 | 609 |
1L disease monitoring | 10,147 | 4,968 | 5,179 |
Subsequent therapy costs | 24,081 | 33,260 | −9,179 |
End-stage organ failure | 58,041 | 102,229 | −44,188 |
End-of-life costs | 5,599 | 5,736 | −137 |
ICER ($ per QALY) | 70,201 | ||
1L = first-line; 2L = second-line; AE = adverse event; CyBorD = cyclophosphamide, bortezomib, and dexamethasone; DCyBorD = daratumumab in combination with CyBorD; FDT = fixed daratumumab treatment; ICER = incremental cost-effectiveness ratio; LY = life-year; QALY = quality-adjusted life year; Tx = treatment.
Appendix 4: Additional Details on the CADTH Reanalyses and Sensitivity Analyses of the Economic Evaluation
Note this appendix has not been copy-edited.
Detailed Results of CADTH Base Case
Table 11: Disaggregated Summary of CADTH’s Economic Evaluation Results
Parameter | DCyBorD | CyBorD | Incremental |
|---|---|---|---|
Discounted LYs | |||
Total LYs | 8.25 | 6.06 | 2.20 |
1L Tx | 0.41 | 0.35 | 0.06 |
Off Tx/FDT | 4.77 | 2.06 | 2.71 |
2L Tx | 2.27 | 2.17 | 0.10 |
End-Stage Organ Failure | 0.80 | 1.47 | -0.67 |
Discounted QALYs | |||
Total QALYs | 5.81 | 3.89 | 1.92 |
1L Tx | 0.31 | 0.26 | 0.05 |
Off Tx/FDT | 3.62 | 1.56 | 2.06 |
2L Tx | 1.56 | 1.47 | 0.09 |
End-Stage Organ Failure | 0.33 | 0.60 | -0.27 |
AE Disutility | -0.0006 | -0.0003 | -0.0003 |
Discounted costs ($) | |||
Total costs | 345,453 | 215,550 | 129,902 |
Total 1L drug therapy | 177,972 | 10,796 | 167,176 |
Total 1L drug administration | 2,456 | 927 | 1,529 |
Co-medication | 901 | 290 | 611 |
Health care resource use | 63,374 | 51,905 | 11,469 |
Adverse event management | 2,265 | 1,663 | 602 |
1L disease monitoring | 10,541 | 5,153 | 5,388 |
Subsequent therapy costs | 24,850 | 33,493 | -8,643 |
End-stage organ failure | 57,481 | 105,579 | -48,098 |
End-of-life costs | 5,613 | 5,744 | -131 |
ICER ($/QALY) | 67,484 | ||
1L = first-line; 2L = second-line; AE = adverse event; CyBorD = cyclophosphamide, bortezomib, and dexamethasone; DCyBorD = daratumumab in combination with CyBorD; FDT = fixed daratumumab treatment; ICER = incremental cost-effectiveness ratio; LY = life-year; QALY = quality-adjusted life year; Tx = treatment.
Scenario Analyses
Table 12: Summary of Scenario Analyses Conducted on CADTH Base Case
Scenario | Drug | Total costs ($) | Total QALYs | ICER ($/QALY) |
|---|---|---|---|---|
CADTH base case | CyBorD | 215,550 | 3.89 | Reference |
DCyBorD | 345,453 | 5.81 | 67,484 | |
1. Maximum treatment duration assumed for both comparators | CyBorD | 216,349 | 3.86 | Reference |
DCyBorD | 383,958 | 5.77 | 88,004 | |
2. Only half of patients in end-stage organ failure would require hemodialysis | CyBorD | 163,461 | 3.96 | Reference |
DCyBorD | 316,939 | 5.86 | 80,954 | |
3. Alternate utility decrement for patients in end-stage organ failure | CyBorD | 215,019 | 4.16 | Reference |
DCyBorD | 346,355 | 5.96 | 72,653 | |
4. Extrapolated OS data for CR patients using a Gompertz function | CyBorD | 214,278 | 3.81 | Ref. |
DCyBorD | 342,828 | 5.57 | 73,277 | |
5. Pharmacy dispensing fees were excluded | CyBorD | 215,545 | 3.87 | Ref. |
DCyBorD | 345,218 | 5.78 | 67,811 | |
6. Analyses 1 + 2 (treatment duration and hemodialysis) | CyBorD | 163,685 | 3.93 | Ref. |
DCyBorD | 353,085 | 5.82 | 100,036 |
CR = complete response; CyBorD = cyclophosphamide, bortezomib, and dexamethasone; DCyBorD = daratumumab in combination with CyBorD; OS = overall survival.
Appendix 5: Submitted BIA and CADTH Appraisal
Note this appendix has not been copy-edited.
Table 13: Summary of Key Take-Aways
Key take-aways of the BIA |
|---|
|
Summary of Sponsor’s BIA
The submitted BIA assessed the introduction of DCyBorD for the treatment of AL amyloidosis. The analysis was taken from the perspective of the Canadian public drug plans using an epidemiology-based approach, with only drug acquisition costs considered. A 3-year time horizon was used, from 2023 to 2025, with 2022 as a base year. The population size was derived using the estimated incidence of AL amyloidosis.
The reference scenario of the BIA included CyBorD, the current standard of care for AL amyloidosis. The new drug scenario included DCyBorD and CyBorD. Key inputs to the BIA are documented in Table 14.
Table 14: Summary of Key Model Parameters
Parameter | Sponsor’s estimate (reported as year 1 / year 2 / year 3 if appropriate) |
|---|---|
Target population | |
Adult population in Canada (2020) | 30,730,00922 |
Annual incidence of AL amyloidosis | 7.16 per 1,000,0002 |
Patients without severe cardiac involvement | 90% (assumption) |
Patients not intending to get an ASCT | 95% (assumption) |
Number of patients eligible for drug under review | 196 / 396 / 401 |
Market uptake (3 years) | |
Uptake (reference scenario) | |
CyBorD | 100% / 100% / 100% |
Uptake (new drug scenario) | |
DCyBorD | ||||||||% / ||||||||% / ||||||||% |
CyBorD | ||||||||% / ||||||||% / ||||||||% |
Cost of treatment (per patient) | |
Cost of annual treatment per patient (year 1) | |
DCyBorD | $179,061 |
CyBorD | $10,384 |
Cost of annual treatment per patient (year 2) | |
DCyBorD | $52,267 |
CyBorD | $0 |
AL = light chain; ASCT = autologous stem cell transplant; CyBorD = cyclophosphamide, bortezomib, and dexamethasone; DCyBorD = daratumumab in combination with cyclophosphamide, bortezomib, and dexamethasone.
Summary of the Sponsor’s BIA Results
The estimated budget impact of funding DCyBorD for the treatment of patients with AL amyloidosis was $33,229,279 in year 1, $43,863,552 in year 2, and $44,476,136 in year 3 for a 3-year total of $121,568,967.
CADTH Appraisal of the Sponsor’s BIA
CADTH identified a key limitation to the sponsor’s analysis that has notable implications on the results of the BIA:
Inappropriate characterization of eligible patient population: The sponsor’s BIA included patients in Quebec, Yukon, and the Northwest Territories, which does not align with CADTH’s submission guidelines.23 Furthermore, the sponsor failed to include the NIHB population.
CADTH corrected the eligible population by excluding the patients from Quebec, Yukon, and the Northwest Territories and calculating and including the NIHB population.
An additional limitation was identified but was not considered to be a key limitation. The sponsor made a programming error in the reference scenario of their BIA, in which they rounded up patients to whole numbers in year 1, but not years 2 and 3. CADTH corrected this by assuming no rounding of patients in any year of the new and reference scenarios.
CADTH Reanalyses of the BIA
CADTH corrected the programming inconsistency in the sponsor’s base case as well as the derivation of the population size. CADTH did not undertake additional reanalyses as there were only a few inputs involved in deriving the population size and market shares which were considered valid by the clinical expert.
Table 15: CADTH Revisions to the Submitted BIA
Stepped analysis | Sponsor’s value or assumption | CADTH value or assumption |
|---|---|---|
Corrections to sponsor’s base case | ||
1. Programming error in reference scenario | Inconsistent patient rounding | No rounding of patients |
2. Derivation of population | Included Quebec, Northwest Territories, Yukon, but excluded NIHB | Correct population including NIHB as per the CADTH submission guidelines |
Changes to derive the CADTH base case | ||
None | — | — |
CADTH base case | Correction 1 + 2 | |
BIA = budget impact analysis; NIHB = non-insured health benefits.
The results of the CADTH stepwise reanalysis are presented in summary format in Table 16 and a more detailed breakdown is presented in Table 17. Based on the CADTH base case, the budget impact of the reimbursement of DCyBorD for the treatment of newly diagnosed AL amyloidosis is expected to be $25,887,024 in year 1, $34,273,444 in year 2, and $34,756,670 in year 3, with a 3-year total of $94,917,168. A scenario analysis was conducted in which it was assumed that daratumumab would also be available for those currently receiving CyBorD, a possibility suggested by the sponsor and the clinical input. The estimated 3-year budget impact as a result of this scenario analysis was $112,421,426.
Table 16: Summary of the CADTH Reanalyses of the BIA
Stepped analysis | Three-year total |
|---|---|
Submitted base case | $121,568,967 |
Submitted base case (corrected) and CADTH base case | $94,917,168 |
BIA = budget impact analysis.
Table 17: Detailed Breakdown of the CADTH Reanalyses of the BIA
Stepped analysis | Scenario | Year 0 (current situation) | Year 1 | Year 2 | Year 3 | Three-year total |
|---|---|---|---|---|---|---|
Submitted base case (corrected) and CADTH base case | Reference | $1,571,539 | $1,593,698 | $1,616,169 | $1,638,957 | $4,848,823 |
New drug | $1,571,539 | $27,480,722 | $35,889,613 | $36,395,657 | $99,765,992 | |
Budget impact | $0 | $25,887,024 | $34,273,444 | $34,756,670 | $94,917,168 | |
CADTH scenario analysis 1: daratumumab add-on therapy for existing patients | Reference | $1,571,539 | $1,593,698 | $1,616,169 | $1,638,957 | $4,848,823 |
New drug | $1,571,539 | $41,030,038 | $39,844,555 | $36,395,657 | $117,270,249 | |
Budget impact | $0 | $39,436,340 | $38,228,386 | $34,756,700 | $112,421,426 | |
CADTH scenario analysis 2: price reduction of 21% from PE model | Reference | $1,571,539 | $1,593,698 | $1,616,169 | $1,638,957 | $4,848,823 |
New drug | $1,571,539 | $22,062,062 | $28,710,053 | $29,114,864 | $79,886,979 | |
Budget impact | $0 | $20,468,364 | $27,093,884 | $27,475,908 | $75,038,155 |
BIA = budget impact analysis; PE = pharmacoeconomic.
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up-to-date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.