CADTH Reimbursement Review
Azacitidine (Onureg)
Sponsor: Celgene Inc., a Bristol Myers Squibb Company
Therapeutic area: Acute myeloid leukemia
This multi-part report includes:
Clinical Review
Pharmacoeconomic Review
Clinical Review
Abbreviations
7 + 3
7 days of cytarabine plus 3 days of anthracycline therapy
AE
adverse event
AML
acute myeloid leukemia
ANC
absolute neutrophil count
ANCOVA
analysis of covariance
APL
acute promyelocytic leukemia
ATB-MPG
Alberta Tumour Board Myeloid Physician Group
BSC
best supportive care
CFA
confirmatory factor analysis
CI
confidence interval
CLL
chronic lymphocytic leukemia
CMML
chronic myelomonocytic leukemia
CR
complete remission
CRi
complete remission with incomplete blood count recovery
ECOG
Eastern Cooperative Oncology Group
ELN
European LeukemiaNet
EMA
European Medicines Agency
EQ-5D-3L
EuroQol 5-Dimensions 3-Levels questionnaire
EQ VAS
EuroQol VAS
FACIT-F
Functional Assessment of Chronic Illness Therapy–Fatigue
FLT3
FMS-like tyrosine kinase 3
H-DAC
Hematological Cancer Drug Advisory Committee
HR
hazard ratio
HRQoL
health-related quality of life
HSCT
hematopoietic stem cell transplantation
ICC
intraclass correlation coefficient
IPCW
inverse probability of censoring weighted
ITT
intention-to-treat
IWG
International Working Group
KM
Kaplan-Meier
LLSC
Leukemia and Lymphoma Society of Canada
LS
least squares
MDS
myelodysplastic syndrome
MID
minimally important difference
mITT
modified intention-to-treat
MMRM
mixed-effect model for repeated measures
MRD
minimal residual disease
OH-CCO
Ontario Health – Cancer Care Ontario
OS
overall survival
PINR
Physical Impairment Numeric Rating
QLQ-C30
Quality of Life Questionnaire Core 30
RCT
randomized controlled trial
RD
responder definition
RFS
relapse-free survival
SD
standard deviation
TEAE
treatment-emergent adverse event
Executive Summary
An overview of the submission details for the drug under review is provided in Table 1.
Item | Description |
|---|---|
Drug product | Azacitidine (Onureg), tablet 300 mg, oral |
Indication | Maintenance therapy in adult patients with AML who achieved CR or CRi following induction therapy with or without consolidation treatment, and who are not eligible for HSCT |
Reimbursement request | As per indication |
Health Canada approval status | NOC |
Health Canada review pathway | Priority review |
NOC date | January 4, 2021 |
Sponsor | Celgene Inc., a Bristol Myers Squibb company |
AML = acute myeloid leukemia; CR = complete remission; CRi = complete remission with incomplete blood count recovery; HSCT = hematopoietic stem cell transplantation; NOC = Notice of Compliance.
Introduction
Acute myeloid leukemia (AML) is the most common form of acute leukemia in adults. It is an aggressive hematological malignancy that affects the production of normal blood cells (i.e., clonal expansion of myeloid blasts in the peripheral blood, bone marrow, and/or other tissue), causing weakness, infection, bleeding, and other symptoms and complications.3,4 According to the most recent incidence statistic available for AML in Canada, 1,090 new cases were reported in 2016; recent mortality statistics indicate that 1,184 Canadians died from AML in 2017.5
Patients with AML who can tolerate aggressive treatment receive initial induction therapy followed by consolidation therapy, and approximately 50% of patients aged 60 years or younger are cured in this fashion.4,6,7 Allogeneic transplantation, which is potentially curative (the probability of long-term survival is 50%), is pursued as a consolidation strategy in patients in first remission with intermediate or poor cytogenetic risk who are eligible for transplantation.1 However, the procedure is associated with a high risk of morbidity and mortality,8 and reasons for transplantation ineligibility vary, including old age, comorbidities, and decreased organ function.1 For patients who are ineligible for transplantation, maintaining first remission to delay relapse and prolong survival is an important clinical goal.1 The clinical experts consulted by CADTH noted that there are currently no standard funded maintenance regimens specific for patients with AML who are in first remission by achieving either complete remission (CR) or complete remission with incomplete blood count recovery (CRi) following induction therapy with or without consolidation treatment and who are not eligible for transplantation. In the absence of proven treatment options for patients in this setting, a watch-and-wait approach or best supportive care (BSC) is recommended.1 Survival of patients with relapsed disease after induction or consolidation treatment is poor; if first remission lasts for less than 1 year, the expected median overall survival (OS) is 4 to 6 months.1 The clinical experts consulted by CADTH agreed that there is a need for effective treatments that prolong life, delay relapse, maintain or improve patient quality of life, have an acceptable safety profile, and have a convenient oral route of administration.
Oral azacitidine is a cytidine nucleoside analogue that incorporates into both DNA and RNA after phosphorylation to the nucleotide and produces anti-leukemic activity in cancers such as AML.1 On January 4, 2021, oral azacitidine was approved by Health Canada as maintenance therapy in adult patients with AML who achieved CR or CRi following induction therapy with or without consolidation treatment and who are not eligible for hematopoietic stem cell transplantation (HSCT).2 The requested reimbursement criteria for oral azacitidine are as per the Health Canada–approved indication. Oral azacitidine has no other Health Canada–approved indication and has not previously been reviewed by CADTH. The recommended starting dosage is 300 mg administered orally once daily on day 1 through day 14 of continuous 28-day cycles. The product monograph states that if the absolute neutrophil count (ANC) is less than 500 µL on day 1 of a cycle, oral azacitidine should not be administered and the start of the cycle should be delayed until the ANC is 500 µL or more.2
Stakeholder Perspectives
The information in this section is a summary of input provided by the patient groups who responded to CADTH’s call for patient input and from clinical expert(s) consulted by CADTH for the purpose of this review.
Patient Input
One patient group, the Leukemia and Lymphoma Society of Canada (LLSC), provided input for this review. The input was based on an online survey created by the LLSC to which a total of 29 patients with AML responded.
Respondents to the LLSC survey indicated a varying range of AML symptoms affecting their daily lives, including disruption to their social lives and relationships, causing detrimental effects on their health. Patient respondents reported being easily fatigued, unable to exercise or work, losing vision in 1 eye, nausea, bruising, numbness or body aches, and being immunocompromised. Many of these symptoms reportedly led to feelings of isolation and fear of relapse, with negative impacts on their psychological well-being.
According to the patient input received, patients expect the following key outcomes to be improved with any new drug or treatment: maintaining remission, fewer side effects, lower cost, and being accessible in their neighbourhoods. Additionally, the following factors were noted in choosing a new cancer treatment: physician recommendation, impact on disease, quality of life, closeness of home, and outpatient treatment. No survey respondents indicated that they had experience taking oral azacitidine.
Clinician Input
Input From Clinical Experts Consulted by CADTH
The following input was provided by 3 clinical specialists with expertise in the diagnosis and management of AML.
The clinical experts consulted by CADTH indicated that there are currently no standard funded maintenance regimens, and watch-and-wait or BSC are recommended for patients in the present target setting. The clinical experts identified an unmet need for effective therapies with acceptable toxicity profiles that delay relapse, prolong survival, maintain health-related quality of life (HRQoL), and potentially lead to a cure in patients with AML who are in their first remission and are not eligible for transplantation. They emphasized a need for a convenient oral route of administration to achieve high adherence and reduce hospital-based resource utilization. It was agreed that, while oral azacitidine would likely shift the current treatment paradigm, more experience with maintenance oral azacitidine therapy will be necessary to determine the potential impacts on the current treatment paradigm (e.g., potentially fewer patients requiring transplant, a reduction of cycles of consolidation chemotherapy, and refinement of the target population).
The clinical experts agreed that patients as selected per the inclusion and exclusion criteria of the QUAZAR AML-001 trial should be eligible for maintenance oral azacitidine therapy. While the clinical experts agreed that there is currently insufficient evidence to guide a recommendation on which patient subgroups would be best suited for (most likely to show a response to) oral azacitidine, the experts identified the following potential patient subgroups as having the highest risk of relapse and therefore being most in need of maintenance therapy: patients aged 65 years or older, patients with minimal residual disease (MRD)-positive status, patients who have not received consolidation chemotherapy, and patients with poor-risk karyotypes. In the opinion of the clinical experts, patient subgroups that would potentially benefit the least from oral azacitidine may include patients with an MDR-negative status, patients with low-risk features for relapse, patients who develop unacceptable toxicities, and patients lacking the social and/or medical support necessary to be safely treated with oral azacitidine.
The clinical experts agreed that patients would be identified as possible candidates for oral azacitidine treatment if they are considered ineligible for transplantation. The transplantation ineligibility status may be known in patients at the time of diagnosis or may develop over the course of their treatment. Clinical assessments to evaluate the response to treatment with oral azacitidine would include regular monitoring of blood counts (every 1 to 2 weeks) to determine if a patient maintains CR or CRi. If changes in blood count signal a potential relapse, a bone marrow examination may be required to determine if a patient is still in CR or CRi or has relapsed AML. The clinical experts indicated that the most clinically meaningful responses to treatment include prolonged OS and relapse-free survival (RFS) while maintaining or improving HRQoL and reducing symptom burdens. No increase in drug-related toxicities, such as infections, neutropenia, and thrombocytopenia, and a reduced risk of relapse were also noted as clinically meaningful outcomes.
In the opinion of the clinical experts, treatment with maintenance therapy of oral azacitidine should be discontinued if a patient experiences relapsed AML, has a markedly impaired performance status, or is intolerant to or experiences unacceptable toxicity from oral azacitidine. If AML recurs with at least 5% of blasts in the peripheral blood or bone marrow, oral azacitidine should be discontinued. There is currently insufficient evidence from the QUAZAR AML-001 trial to recommend dose escalation when AML recurs with blast levels between 5% and 15% in either the peripheral blood or bone marrow.
Clinician Group Input
Two clinician group inputs were provided, 1 from the Ontario Health–Cancer Care Ontario (OH-CCO) Hematological Cancer Drug Advisory Committee (H-DAC) and 1 from the Alberta Tumour Board Myeloid Physician Group (ATB-MPG). Overall, the views of the clinician groups were consistent with those of the clinical experts consulted by CADTH; all agreed that the most important treatment goals are prolongation of life and remission as reflected by the pivotal trial’s primary and key secondary end points of OS and RFS, respectively, as well as improvement in quality of life. Similar to the clinical experts consulted by CADTH, the clinicians from the OH-CCO H-DAC reported that they would generalize the QUAZAR AML-001 trial results to patients younger than 55 years of age. In addition, the clinician group also stated that pharmacokinetic data suggest that patients with myelodysplastic syndrome (MDS) who have progressed on subcutaneous azacitidine and subsequently received induction chemotherapy with or without consolidation and have achieved CR may also benefit from oral azacitidine if they are not eligible for transplantation. There was consensus among the clinical experts consulted by CADTH and the clinicians from both clinician groups that oral azacitidine would be a standard-of-care maintenance therapy for patients with AML who are in first CR and ineligible for HSCT.
Drug Program Input
Input from the drug programs identified factors pertaining to relevant comparators, prior therapies required for eligibility, therapy continuation criteria, dosing considerations, and generalizability that may affect their ability to implement a recommendation. The clinical experts consulted by CADTH weighed evidence from the QUAZAR AML-001 trial and other clinical considerations to provide responses to questions from the drug programs related to these factors, which can be found in Table 4.
Clinical Evidence
Pivotal Studies and Protocol-Selected Studies
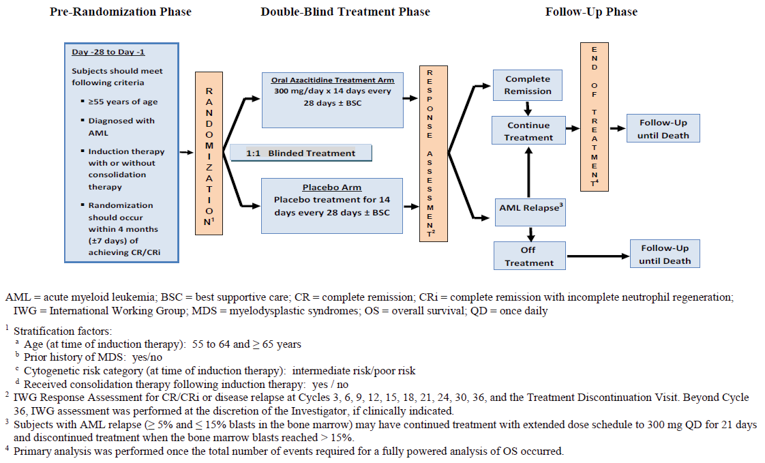
Description of Study
The QUAZAR AML-001 trial is an ongoing, international, multicentre, double-blind, placebo-controlled randomized phase III trial that compared the efficacy and safety of maintenance therapy with oral azacitidine plus BSC with placebo plus BSC in patients with AML in first CR. A total of 472 patients were randomized in a 1:1 ratio to receive maintenance oral azacitidine (300 mg tablets once daily for the first 14 days of each 28-day cycle) plus BSC or oral placebo (matching placebo tablets once daily for the first 14 days of each 28-day cycle) plus BSC. Randomization was stratified by age at time of induction therapy (55 to 64 years and ≥ 65 years), prior history of MDS or chronic myelomonocytic leukemia (CMML) (yes/no), cytogenetic risk category at time of induction therapy (intermediate risk/poor risk), and receipt of consolidation therapy following induction (yes/no). No crossover between the treatment groups was permitted. The primary outcome was OS, and the key secondary outcome was RFS. Other secondary end points included time to relapse and time to discontinuation from treatment. Two HRQoL measures, the EuroQol 5-Dimensions 3-Levels questionnaire (EQ-5D-3L) and the Physical Impairment Numeric Rating (PINR) scale, were included as secondary and exploratory outcomes, respectively. A symptom severity measure, the Functional Assessment of Chronic Illness Therapy–Fatigue (FACIT-F) scale was included as a secondary outcome.9
The QUAZAR AML-001 trial enrolled adults 55 years of age and older diagnosed with AML or AML secondary to prior MDS or CMML with intermediate- or poor-risk cytogenetics and who had achieved first remission (CR or CRi) following induction with or without consolidation chemotherapy. Patients were excluded if they were eligible for HSCT. At screening patients had to have an Eastern Cooperative Oncology Group Performance Status (ECOG PS) of 0 to 3 and adequate marrow function.10 The mean age of patients in the trial was 68.0 (standard deviation [SD] = 5.66) years of age. The oral azacitidine group had a lower proportion (versus placebo) of male patients (49.6% versus 54.3%) and patients enrolled in trial sites in Asia (2.5% versus 7.3%) and a higher proportion of White patients (90.8% versus 84.2%) and patients enrolled in trial sites in Europe (70.2% versus 62.8%). The majority of patients had an ECOG PS of 0 (48.7% versus 47.4%) or 1 (42.4% versus 45.3%) and intermediate cytogenetic risk (85.3% versus 86.8%) compared with patients given placebo. Most of the patients received 1 cycle (46.2% versus 43.6%) or 2 cycles (29.4% versus 32.9%) of consolidation therapy. The most common reason for transplantation ineligibility was age (64.7% versus 65.0%), followed by comorbidities (21.8% versus 21.4), and no available donor (15.5% versus 15.0%).9 The oral azacitidine group had a lower proportion of patients with a negative MRD status at randomization (55.9% versus 47.4%) and a lower proportion of patients who achieved CR after induction therapy with or without consolidation compared with placebo (78.6% versus 84.2%).9 The clinical experts consulted by CADTH noted that the differences observed in patient characteristics at baseline between the oral azacitidine and the placebo groups are unlikely to have had an impact on the treatment differences observed between the treatment groups.
A post-relapse dose escalation (300 mg once daily from 14 days to 21 days of the 28-day cycle) was planned for patients with disease relapse with blast levels of between 5% and 15% in either the peripheral blood or bone marrow.9 This dose escalation was used in the context of re-induction of remission, which is not consistent with the Health Canada indication and the requested reimbursement criteria, which are for oral azacitidine as maintenance therapy.11 The QUAZAR AML-001 trial was not designed to assess if dose escalation of oral azacitidine produces benefits for patients. Dose escalation has not been authorized by Health Canada as per the Health Canada product monograph.11
The primary analysis, which coincided with the final data cut-off date (July 15, 2019) was conducted once the total number of events (n = 330 deaths) occurred.9,10 The key secondary and other secondary end points were also planned to be assessed at the time of the final data cut-off date. The trial is ongoing, with an estimated completion date of December 31, 2021. Data from an updated data cut-off date (September 2020), which was not planned a priori in the statistical analysis plan, were not provided to CADTH by the sponsor at the time of this review.12,13
Efficacy Results
The key efficacy results from the QUAZAR AML-001 trial are summarized in Table 2. At the final data cut-off date (July 15, 2019) the median duration of follow-up for OS was ||| months in the oral azacitidine group and ||| months in the placebo group. Median OS was 24.7 (95% confidence interval [CI], 18.7 to 30.5) months in the oral azacitidine group compared with 14.8 (95% CI, 11.7 to 17.6) months in the placebo group, with a stratified hazard ratio (HR) of 0.69 (95% CI, 0.55 to 0.86; P = 0.0009) in favour of the oral azacitidine group. Testing of treatment-by-time interactions in the overall Cox proportional hazards model suggested deviation from the proportional hazards assumption. The results of sensitivity analyses for OS that were planned to be conducted if the proportional hazards assumption was violated suggested robustness and consistency of the primary OS result. The OS results for the subgroups of interest, as prespecified a priori in the protocol for this CADTH review, suggested that the treatment effect on OS for the subgroups was generally consistent with the primary analysis. The subgroup analysis by cycles of consolidation therapy suggested possible heterogeneity of treatment effect; however, a number of methodological issues limit the ability to interpret these results.9
The median RFS was 10.2 months (95% CI, 7.9 to 12.9) in the oral azacitidine group and 4.8 months (95% CI, 4.6 to 6.4) in the placebo group, with a stratified HR of 0.65 (95% CI, 0.52 to 0.81; P = 0.0001) in favour of the oral azacitidine group.9
As of the final analysis, the percentage of patients who had relapsed was 64.7% in the oral azacitidine group compared with 76.5% in placebo group. Median time to relapse was 10.2 months (95% CI, 8.3 to 13.4) in the oral azacitidine group and 4.9 months (95% CI, 4.6 to 6.4) in the placebo group. The relapse rate at 1 year was 0.53 (95% CI, 0.46 to 0.59) in the oral azacitidine group and 0.71 (95% CI, 0.65 to 0.77) in the placebo group.9
Most patients in both treatment groups had discontinued study treatment (81.1% in the oral azacitidine group and 88.9% in the placebo group) at the time of the final analysis. The median time to treatment discontinuation was 11.4 months (95% CI, 9.8 to 13.6) in the oral azacitidine group and 6.1 months (95% CI, 5.1 to 7.4) in the placebo group. The 1-year treatment discontinuation rate was |||||||||||||||||||||||||||||||| in the oral azacitidine group and |||||||||||||||||||||||||||||||| in the placebo group.9
Overall, there were no statistically significant or clinically meaningful differences between the oral azacitidine group and the placebo group in the observed mean changes from baseline at any post-baseline assessment for the EQ-5D-3L health utility index and EuroQol Visual Analogue Scale (EQ VAS), PINR scale, and FACIT-F scale. Results using analysis of covariance (ANCOVA) models and mixed-effect model for repeated measures (MMRM) analyses showed similar results overall, with no statistically significant or clinically meaningful differences in least squares (LS) mean changes evident between treatment groups across assessments for any of the 3 instruments (EQ-5D-3L, PINR scale, and FACIT-F scale). Furthermore, no statistically significant differences in the proportion of patients with clinically meaningful deterioration were evident between the oral azacitidine and placebo groups for the FACIT-F scale and the EQ-5D-3L health utility index and EQ VAS. The time to definitive deterioration was similar between the oral azacitidine and the placebo groups regardless of the instrument used (i.e., FACIT-F, EQ-5D-3L health utility index, or EQ VAS). Clinically meaningful deterioration and time to definitive deterioration were not reported for the PINR scale.14 All analyses performed on the HRQoL outcomes and symptom severity were noninferential.
Harms Results
Key harms reported in the QUAZAR AML-001 trial as of the final analysis (July 15, 2019, data cut-off date) are summarized in Table 2. Nearly all patients in both study groups (97.9% of patients in the oral azacitidine group and 96.6% in the placebo group) experienced at least 1 treatment-emergent adverse event (TEAE). The most commonly reported TEAEs in the oral azacitidine and the placebo groups were nausea (64.8% and 23.6%, respectively), vomiting (59.7% and 9.9%, respectively), diarrhea (50.4% and 21.5%, respectively), and neutropenia (44.5% and 26.2%, respectively). TEAEs led to discontinuation of study treatment in 13.1% of patients in the oral azacitidine group and 4.3% of patients in the placebo group. The percentage of patients experiencing serious TEAEs was 33.5% in the oral azacitidine group compared to 25.3% in the placebo group. The most common serious TEAEs reported in the oral azacitidine and the placebo groups were febrile neutropenia (6.8% and 3.9%, respectively), pneumonia (3.8% and 3.0%, respectively), and pyrexia (2.1% and 0.4%, respectively). As of the final analysis, 66.4% of patients in the oral azacitidine group and 73.1% of patients in the placebo group had died, with most deaths attributed to AML.9
The percentage of patients experiencing gastrointestinal TEAEs was 91.1% in the oral azacitidine group compared to 66.5% in the placebo group (Table 2). The most commonly reported gastrointestinal TEAEs in the oral azacitidine and the placebo groups were nausea (64.8% and 23.6%, respectively), vomiting (59.7% and 9.9%, respectively), diarrhea (50.4% and 21.5%, respectively), and constipation (38.6% and 24.0%, respectively).9
The percentage of patients experiencing myelosuppression TEAEs was |||| in the oral azacitidine group compared to |||| in the placebo group (Table 2). The most commonly reported myelosuppression event was neutropenia, which was reported in 44.5% of patients in the oral azacitidine group compared to 26.2% of patients in the placebo group.9
The percentage of patients experiencing infections was 62.3% in the oral azacitidine group and 52.8% in the placebo group (Table 2). The most commonly reported infections in the oral azacitidine group included upper respiratory tract infections (13.1% and 13.7% in the oral azacitidine and placebo groups, respectively), |||||||||| (|||||||||||||| respectively), and |||||||||||||||||| (|||||||||||||||||| respectively).9
The percentage of patients experiencing fatigue was 29.7% in the oral azacitidine group and 19.3% in the placebo group.9
The types and frequency of hemorrhagic events were similar across study groups. The most commonly reported events (oral azacitidine versus placebo) included |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||.9
Table 2: Summary of Key Results From Pivotal and Protocol-Selected Studies
Outcome | Oral azacitidine (N = 238) | Placebo (N = 234) |
|---|---|---|
Overall survival | ||
Data cut-off date | July 15, 2019 | |
Primary outcome: overall survival | ||
Median follow-up time for overall survival,a months | |||||||||||||| | |||||||| |
Median, months (95% CI)b | 24.7 (18.7 to 30.5) | 14.8 (11.7 to 17.6) |
Events (death), n (%) | 158 (66.4) | 171 (73.1) |
Censored, n (%) | |||||||||||||| | |||||||| |
Hazard ratio (95% CI)c | 0.69 (0.55 to 0.86) | |
P valued | 0.0009 | |
Survival probabilitye | ||
12 months (95% CI) | 0.73 (0.66 to 0.78) | 0.56 (0.49 to 0.62) |
24 months (95% CI) | 0.51 (0.44 to 0.57) | 0.37 (0.31 to 0.43) |
Relapse-free survival | ||
Median, months (95% CI)f | 10.2 (7.9 to 12.9) | 4.8 (4.6 to 6.4) |
Events (relapse or death) | 164 (68.9) | 181 (77.4) |
Relapse, n (%) | 154 (64.7) | 179 (76.5) |
Death, n (%) | |||||||||||||| | |||||||| |
Censored, n (%) | 74 (31.1) | 53 (22.6) |
Hazard ratio (95% CI)c | 0.65 (0.52 to 0.81) | |
P valued | 0.0001 | |
Relapse-free survival probabilityg | ||
6 months (95% CI) | 0.67 (0.61 to 0.73) | 0.45 (0.39 to 0.52) |
12 months (95% CI) | 0.45 (0.38 to 0.51) | 0.27 (0.22 to 0.34) |
24 months (95% CI) | |||||||||||||| | |||||||| |
Harms (safety population) | ||
N | 236 | 233 |
TEAEs, n (%) | 231 (97.9) | 225 (96.6) |
Nausea | 153 (64.8) | 55 (23.6) |
Vomiting | 141 (59.7) | 23 (9.9) |
Diarrhea | 119 (50.4) | 50 (21.5) |
Neutropenia | 105 (44.5) | 61 (26.2) |
Serious TEAEs, n (%) | 79 (33.5) | 59 (25.3) |
Febrile neutropenia | 16 (6.8) | 9 (3.9) |
Pneumonia | 9 (3.8) | 7 (3.0) |
Pyrexia | 5 (2.1) | 1 (0.4) |
Cellulitis | 4 (1.7) | 1 (0.4) |
Discontinued treatment due to TEAEs | 31 (13.1) | 10 (4.3) |
Deaths | 157 (66.0) | 171 (73.1) |
Notable harms | ||
Gastrointestinal events (any grade TEAE), n (%) | 215 (91.1) | 155 (66.5) |
Nausea | 153 (64.8) | 55 (23.6) |
Vomiting | 141 (59.7) | 23 (9.9) |
Diarrhea | 119 (50.4) | 50 (21.5) |
Constipation | 91 (38.6) | 56 (24.0) |
Myelosuppression event (any grade TEAE), n (%) | |||||||||||||| | |||||||| |
Neutropenia | 105 (44.5) | 61 (26.2) |
Thrombocytopenia | 79 (33.5) | 63 (27.0) |
Anemia | 48 (20.3) | 42 (18.0) |
Febrile neutropenia | 28 (11.9) | 18 (7.7) |
Infections (any grade TEAE), n (%) | 147 (62.3) | 123 (52.8) |
Upper respiratory tract infection | 31 (13.1) | 32 (13.7) |
Influenza | |||||||||||||| | |||||||| |
Nasopharyngitis | |||||||||||||| | |||||||| |
Urinary tract infection | |||||||||||||| | |||||||| |
Fatigue (any grade TEAE), n (%) | 70 (29.7) | 45 (19.3) |
Hemorrhagic events (any grade TEAE), n (%) | 51 (21.6) | 46 (19.7) |
Epistaxis | |||||||||||||| | |||||||| |
Contusion | |||||||||||||| | |||||||| |
Hematoma | |||||||||||||| | |||||||| |
Ecchymosis | |||||||||||||| | |||||||| |
CI = confidence interval; TEAE = treatment-emergent adverse event.
aMedian follow-up time for overall survival estimated by the reverse Kaplan-Meier method for all patients.
bMedian estimate of overall survival is from a Kaplan-Meier method.
cThe hazard ratio is from a Cox proportional hazards model stratified by age, cytogenetic risk category, and receipt of consolidation therapy or not.
dThe P value is 2-sided from a log-rank test stratified by age, cytogenetic risk category, and receipt of consolidation therapy or not.
eKaplan-Meier methods were used to estimate the 1-year and 2-year survival probabilities.
fMedian estimate of relapse-free survival is from a Kaplan-Meier method.
gKaplan-Meier methods were used to estimate the 1-year and 2-year survival probabilities.
Source: Clinical Study Report.9
Critical Appraisal
Testing of treatment-by-time interactions in the overall Cox proportional hazards model suggested deviation from the proportional hazards assumption. Sensitivity analyses were performed to adjust for nonproportional hazards. Results of these analyses were consistent with the results of the proportional hazards analysis, which supported the HR obtained in the primary analysis demonstrating that oral azacitidine improved patients’ OS relative to placebo.
A higher proportion of patients in the placebo group compared with the oral azacitidine group received subsequent systemic therapy and subsequent transplantation after discontinuing study treatment. Results from planned sensitivity analyses, including censoring for the use of subsequent therapy or subsequent transplantation, were consistent with the primary OS results. Given the consistent results obtained in the sensitivity analyses and the fact that the clinical experts consulted by CADTH anticipated limited impact of post-relapse therapy on survival, the potential of confounding effects on survival outcomes from subsequent treatment is expected to be low.
Only the primary end point, OS, and the key secondary end point, RFS, were included in the statistical hierarchy and adjusted for multiplicity. No adjustment for multiplicity was made for any other analyses of other outcomes in the trial, such as HRQoL and symptom severity.
Methodological issues limited the ability to interpret the results from subgroup analyses. The subgroup analyses were noninferential, wide CIs reflected uncertainty in the effect estimates, and small sample sizes limited the generalizability to a broader population.
The ability to interpret results from the EQ-5D-3L and FACIT-F scale is limited and results from the PINR scale are inconclusive, due to the noninferential analyses, the significant decline in patients available to provide assessments over time, and the use of an unestablished method to derive a minimally important difference (MID).
The standard dose of oral azacitidine in the trial would be the dose used in Canadian clinical practice and is in line with the Health Canada–approved dosing. Post-relapse dose escalation, which was attempted in the QUAZAR AML-001 trial to reinitiate remission, is not generalizable to clinical practice in Canada according to the clinical experts consulted by CADTH and has not been approved by Health Canada. The clinical experts noted that it is unlikely that dose escalation would significantly influence the clinical outcomes observed in the trial due to the small numbers of patients who received dose escalation relative to the study sample size.
Indirect Comparisons
No indirect treatment comparisons were included in the sponsor’s submission to CADTH or identified in the literature search.
Other Relevant Evidence
No long-term extension studies or other relevant studies were included in the sponsor’s submission to CADTH or identified in the literature search.
Conclusions
One phase III, double-blind, placebo-controlled randomized controlled trial (RCT) (QUAZAR AML-001) provided evidence regarding the efficacy and safety of maintenance therapy with oral azacitidine plus BSC compared with placebo plus BSC in patients with AML in first CR. Compared with patients treated with placebo, patients who were treated with oral azacitidine showed statistically significant improvements in OS, the primary end point, and RFS, the key secondary outcome. The differences in median OS and RFS between treatment groups (approximately 10 months and 5 months, respectively) were considered clinically meaningful by the clinical experts consulted by CADTH. Other secondary efficacy outcomes, time to relapse from CR or CRi and time to discontinuation from treatment, were also supportive of the observed OS and RFS gains. The level of fatigue and HRQoL over the assessment period was similar between the 2 study groups and oral azacitidine did not appear to significantly delay time to deterioration compared with placebo. However, results for HRQoL and symptom severity remain uncertain due to a number of important limitations. The toxicity profile of oral azacitidine was considered manageable by the clinical experts consulted by CADTH.
Introduction
Disease Background
Acute myeloid leukemia is the most common form of acute leukemia in adults. It is an aggressive hematological malignancy that affects the production of normal blood cells (i.e., clonal expansion of myeloid blasts in the peripheral blood, bone marrow, and/or other tissue), causing weakness, infection, bleeding, and other symptoms and complications.3,4,15 According to the most recent incidence statistics available for AML in Canada, 1,090 new cases of AML were reported in 2016.5 The most recent mortality statistics indicate that 1,184 Canadians died from AML in 2017.5 The median age of diagnosis has been estimated to be 65 years, with approximately one-third of diagnoses occurring in those aged 75 years or older.4,16
In Canada, the 5-year and 10-year estimated net survival rates for AML are 21% and 18%, respectively.17 Median survival of untreated AML has been estimated to range between 11 and 20 weeks.16 The prognosis of patients with AML is influenced by several factors, including age at diagnosis, cytogenetics, and MRD. Older people have worse prognoses; those diagnosed after the age of 85 have an estimated 5-year survival rate of 0.4%.1 Patients with AML are stratified into those with favourable, intermediate, and adverse risk, primarily mediated by the molecular genetic profile of the AML.6
AML is classified into 4 main disease subtypes by the WHO classification system: AML with recurrent genetic abnormalities (11% of cases), AML with myelodysplasia-related features (6% of cases), therapy-related AML (2% of cases), and AML not otherwise specified (81% of cases).16 Commonly associated mutations in AML include mutations in the FMS-like tyrosine kinase 3 (FLT3) gene and mutations in nucleophosmin 1 (NPM1) both of which are found in approximately 30% of patients with AML.6 Generally, 20% blasts in the marrow or blood is required for a diagnosis of AML; however, a diagnosis of AML may be made with a myeloid blast level of 20% with the following cytogenetic abnormalities: t(15;17), t(8;21), t(16;16), or inv(16).16 A diagnosis of AML is confirmed by bone marrow histology and ancillary tests such as cytogenetics and molecular testing.
Standards of Therapy
Although there are no overarching national Canadian guidelines for the management of AML, several international guidelines harmonize with practice in Canada.4,6,7 Different therapeutic approaches are pursued for patients who can tolerate aggressive therapy and those who cannot. For patients who are not candidates for intensive chemotherapy because of advanced age or frailty, treatment with low-intensity therapies without curative intent is a reasonable option.4
In patients who can tolerate aggressive treatment, the first step of treatment is induction therapy, with the goal of achieving a first CR. Standard induction treatment involves combination chemotherapy (7 days of cytarabine and 3 days of anthracycline therapy [7 + 3]). Targeted therapies are added to this backbone as indicated. There is evidence to support that the combination of gemtuzumab ozogamicin with 7 + 3 prolongs progression-free survival and OS in patients with AML,18 particularly those with a favourable or intermediate cytogenetic risk. Gemtuzumab ozogamicin was recently reviewed by CADTH and is recommended for reimbursement for adults with previously untreated, de novo CD33-positive AML, with the exception of those with acute promyelocytic leukemia (APL), and who have good performance status and favourable, intermediate, or unknown cytogenetics (ELN [European LeukemiaNet] 2017).19 For patients with an FLT3 mutation, combining midostaurin with standard remission induction (7 + 3) and consolidation chemotherapy is associated with an OS benefit. Midostaurin has been reviewed by CADTH and is funded in most jurisdictions in Canada for this indication.20 In patients with a higher risk of disease, a more aggressive regimen than the 7 + 3 regimen, such as FLAG (fludarabine plus high-dose cytarabine plus granulocyte colony-stimulating factor) with or without idarubicin may be considered. As was indicated by the clinical experts consulted by CADTH, if first remission is not achieved after 1 cycle of induction therapy, a second cycle of induction therapy is given. This may include a repeat of 7 + 3 chemotherapy or alternatively FLAG-Ida (FLAG plus idarubicin).16
The goal of consolidation therapy is to maintain first CR.1 Post-remission consolidation therapy involves up to 4 cycles of high-dose cytarabine.16 Approximately 50% of patients aged 60 years or younger are cured in this fashion.4,6,7 Allogeneic transplantation, which is potentially curative, is pursued as a consolidation strategy in first CR patients with intermediate or poor cytogenetic risk who are eligible for transplantation.1 The clinical experts consulted by CADTH noted that patients with favourable cytogenetic risk are not generally considered for transplantation given their positive outcomes and likelihood of being cured after consolidation therapy with standard chemotherapy treatment alone. Allogeneic transplantation for AML in first remission is associated with a long-term survival probability of 50%; however, the procedure is associated with a high risk of morbidity and mortality.8 Reasons for transplantation ineligibility include old age, comorbidities, and decreased organ function.1 The clinical experts consulted by CADTH noted that the stem cell donor option or a patient’s preference also determines transplant eligibility. Furthermore, according to the clinical experts, at times the optimal number of cycles of consolidation chemotherapy cannot be administered for similar reasons, as noted for transplantation ineligibility. The percentage of patients proceeding to transplantation is approximately 30% in those younger than 70 years of age and lower than 5% in those older than 70 years.1
For patients who are ineligible for transplantation, maintaining first CR to delay relapse and prolong survival is an important clinical goal, particularly in patients with intermediate or poor cytogenetic risk who have a higher risk of relapse.1 The clinical experts consulted by CADTH noted that there are currently no standard funded maintenance regimens specific for patients with AML who achieved CR or CRi following induction therapy with or without consolidation treatment and who are not eligible for transplantation. In the absence of proven treatment options in this setting, a watch-and-wait approach or BSC is recommended. Treatment options of relapsed AML are limited and commonly involve palliative chemotherapy.16 Survival of patients with relapsed disease after induction and/or consolidation treatment is poor (if first CR lasts 1 to 2 years, median OS is estimated at 1 year or more; if first CR lasts less than 1 year, expected median OS is 4 to 6 months).1 The clinical experts agreed that there is a need for effective treatments that prolong life, delay relapse, maintain or improve patient’s quality of life with an acceptable safety profile, and have a convenient oral route of administration.
The clinical experts noted that injectable azacitidine may be considered as maintenance therapy in patients who are ineligible for standard consolidation therapy. The use of injectable azacitidine for this particular clinical situation is considered off-label. Funding criteria for injectable azacitidine as maintenance therapy vary by jurisdiction and there is currently no standardized dose protocol. The product monograph for oral azacitidine discourages substituting oral azacitidine for IV or subcutaneous azacitidine.2 Consequently, injectable azacitidine as maintenance therapy is given to a small number of patients. Due to its infrequent use, injectable azacitidine was not included as relevant comparator in the protocol for this CADTH systematic literature review.
The clinical experts also noted that MRD monitoring is currently being studied in clinical practice of AML management, as it may help identify patients at high risk of an impending relapse of AML, providing an opportunity to intervene with therapeutics before full-blown relapse. However, MRD monitoring in AML is not widely available and is not yet part of formal standard of care.
Drug
Oral azacitidine is a formulation of the hypomethylating drug azacitidine, which is administered orally.1 It is a cytidine nucleoside analogue that incorporates into both DNA and RNA after phosphorylation to the nucleotide and produces anti-leukemic activity in cancers such as AML.1
On January 4, 2021, oral azacitidine was approved by Health Canada as maintenance therapy in adult patients with AML who achieved CR or CRi following induction therapy with or without consolidation treatment and who are not eligible for HSCT.2 The sponsor’s requested reimbursement criteria for oral azacitidine are as per the Health Canada–approved indication. Oral azacitidine received a priority review by Health Canada. Oral azacitidine has no other Health Canada–approved indication and has not previously been reviewed by CADTH.
Oral azacitidine is available as 200 mg and 300 mg tablets. The recommended starting dosage is 300 mg administered orally once daily on day 1 through day 14 of continuous 28-day cycles. The product monograph states that if the ANC is less than 500/µL on day 1 of a cycle, oral azacitidine should not be administered and the start of the cycle should be delayed until the ANC is 500/µL or more.2
Table 3: Key Characteristics of Oral Azacitidine
Characteristic | Oral azacitidine |
|---|---|
Mechanism of action | Incorporates into both DNA and RNA and produces anti-leukemic activity in cancers such as AML |
Indicationa | Maintenance therapy in adult patients with AML who achieved CR or CRi following induction therapy with or without consolidation treatment, and who are not eligible for HSCT |
Route of administration | Oral |
Recommended dosage | 300 mg administered orally once daily on day 1 through day 14 of continuous 28-day cycles |
Serious adverse effects or safety issues | Do not substitute oral azacitidine for IV or subcutaneous azacitidine; gastrointestinal toxicities were the most frequent adverse reactions and therefore provision of prophylactic anti-emetic therapy should be considered; hematologic toxicity was commonly reported and therefore complete blood counts should be monitored and dosage modified as recommended |
AML = acute myeloid leukemia; CR = complete remission; CRi = incomplete blood count recovery; HSCT = hematopoietic stem cell transplantation.
aHealth Canada–approved indication.
Source: Product monograph.2
Stakeholder Perspectives
Patient Group Input
This section was prepared by CADTH staff based on the input provided by patient groups.
About the Patient Group(s) and Information Gathered
One patient group, the LLSC, provided input for this review. The LLSC has the following mission statement: Cure leukemia, lymphoma, Hodgkin’s disease and myeloma, and improve the quality of life of patients and their families. This is done through research, patient support, and advocacy.
The patient input was collected through an online survey created by the LLSC, available in both English and French, during the period from December 7, 2020, to January 24, 2021. A total of 29 patients with AML responded: 18 females, 10 males, and 1 patient who did not identify their gender. Most patients were from Ontario (n = 13); the remaining patients were from Quebec and British Columbia (n = 6 each) and Alberta (n = 4). The majority of patients were aged between the ages of 55 years and 74 years (n = 18); 9 patients were younger than 55 years of age, while 2 were older than 75 years. Most patients were diagnosed with AML between 2015 and 2020, with the exception of 2 patients who were diagnosed in 2014 (n = 1) and 2021 (n = 1).
Disease Experience
Respondents in the LLSC survey indicated that a varying range of AML symptoms affected their daily lives, including their social lives and relationships, causing a number of detrimental effects on their health. Patient respondents reported being easily fatigued, unable to exercise or work, losing vision in an eye, nausea, bruising, numbness or body aches, and being immunocompromised. Many of these symptoms left patients feeling physically and/or socially isolated and in fear of relapse, leading to negative impacts on their psychological well-being. Some patient respondents indicated that physical activity helped alleviate their fatigue.
The following quotes illustrate patients’ experiences of living with AML:
“The initial year was absolutely overwhelming, in terms of treatment and expenses.”
“Everything in my life stopped cold turkey-employment, social life, relationships, etc. I made a complete personal 360 degree pivot to focus on my healing and living.”
“COVID and my compromised immune system has caused me to be very socially isolated. I haven’t seen some very important people in my life for almost 2 years at this point.”
The effects of AML are experienced by both patients and their caregivers. According to the survey, patients reported that caregivers or family members might feel stress, worry, sadness, insecurity, and fear of dying. Caregivers were also reported to express more tolerance toward the patients. One individual responded, “It affects those in your immediate household since they will most probably be your primary caregivers. However, anyone that is close to you will be affected as well.”
Experiences With Currently Available Treatments
According to the survey, the majority of patients received chemotherapy and stem cell or bone marrow transplantation after diagnosis, while a small number of patients received drug or radiation therapy and 1 patient received chimeric antigen receptor T-cell therapy. The following treatment-associated side effects had a large impact on patients’ quality of life (in order of most frequently reported): hair loss; weakness; extreme fatigue; diarrhea; infections; anemia; mouth sores; nausea and vomiting; fever; low blood cell counts; tingling sensations; constipation; graft-versus-host disease; lung, heart, kidney or nerve problems, cough; rashes; shortness of breath; and psychological distress. Chemotherapy and transplantation reportedly had 1 of the largest negative impacts on quality of life, due to changes in physical activity, anxiety, problems in mental health and overall happiness, eating challenges, and social and educational development. Feelings of isolation during hospitalization for stem cell transplantations, was another common issue reported among respondents. Opportunistic infection could occur due to their immunocompromised state.
Comments from patients regarding their experiences with AML treatments included:
“Your whole world changes when you are diagnosed with AML. Suddenly, you confront your mortality. You feel extremely weak, you have to go into hospital for months, and you don't realize you MUST go into remission to have a stem cell transplant.”
“The Chemo, transplant, radiation and drug therapies are all hard on the body and short term, long term effects are tough as well. The worst part is that these therapies can cause other illnesses.”
Patient respondents reported a mix of both positive and negative experiences accessing treatments. Almost half of the patients reported generally positive experiences and support from medical staff. Six patients reported negative experiences, resulting from a long referral period with their physician, lack of treatment information, difficulty getting a room for consolidation therapy, having to receive treatment in another city or province, being away from their support systems as a result of moving, and a general lack of psychological support.
Improved Outcomes
Patients reported that they expect any new drug or treatment to produce the following key outcomes: maintaining remission, fewer side effects, lower cost, and being accessible in their neighbourhoods. Additionally, the following factors were noted in choosing a new cancer treatment: physician recommendation, impact on disease, quality of life, closeness to home, and outpatient treatment. The availability of supportive options, such as meditation, hypnosis, neuro-linguistic programming support, and awareness support (thoughts, emotions, and behaviours), were also mentioned.
Experience With Drug Under Review
No survey respondents indicated that they had experience taking oral azacitidine.
Clinician Input
Input From Clinical Experts Consulted by CADTH
All CADTH review teams include at least 1 clinical specialist with expertise in the diagnosis and management of the condition for which the drug is indicated. Clinical experts are a critical part of the review team and are involved in all phases of the review process, providing guidance on the development of the review protocol, assisting in the critical appraisal of clinical evidence, interpreting the clinical relevance of the results, and providing guidance on the potential place in therapy). The following input was provided by 3 clinical specialists with expertise in the diagnosis and management of AML.
Unmet Needs
The clinical experts consulted by CADTH noted that there are currently no standard funded maintenance regimens specific to patients with AML who have achieved CR or CRi following induction therapy with or without consolidation treatment and who are not eligible for HSCT. In the absence of proven treatment options in this setting, watch-and-wait or BSC is recommended. There was consensus among the clinical experts that there is an unmet need for effective therapies with an acceptable toxicity profile that delay relapse, prolong survival, maintain HRQoL and potentially lead to a cure in patients with AML who are in their first remission and not eligible for transplantation. They emphasized the need for a convenient oral route of administration to achieve high adherence and reduce hospital-based resource utilization. One clinical expert suggested that the benefit-risk profile of new therapies may be more beneficial for patients with high-risk features for relapse (e.g., MRD-positive or intermediate- or poor-cytogenic risk [ELN 2017]).
Place in Therapy
Oral azacitidine was intended to be used by patients with AML who achieved CR or CRi following induction therapy with or without consolidation treatment and who are not eligible for HSCT as per the QUAZAR AML-001 trial. While the clinical experts consulted by CADTH agreed that oral azacitidine would likely shift the current treatment paradigm, they also agreed that more experience with maintenance oral azacitidine therapy will be necessary to determine the potential impacts on the current treatment paradigm (e.g., potentially fewer patients requiring transplantation, a reduction in the number of cycles of consolidation chemotherapy, and refinement of the target population). The experts added that, given the lack of comparative data, the relative efficacy of hypomethylating drugs, such as oral azacitidine, compared with targeted therapy in subtypes of AML with certain mutations (e.g., midostaurin in the FLT3-ITD-mutated AML subtype) is currently unknown.
Patient Population
Overall, the clinical experts agreed that patients selected according to the inclusion and exclusion criteria of the QUAZAR AML-001 trial should be eligible for maintenance oral azacitidine therapy. While it was agreed that there is currently insufficient evidence to recommend which patient subgroups would be best suited for or most likely show a response to oral azacitidine, the clinical experts identified the following potential subgroups as having the highest risk of relapse and therefore being most in need of maintenance therapy: patients 65 years or older, patients with an MRD-positive status, patients who have not received consolidation chemotherapy, and patients with poor-risk karyotypes. Subgroups who may benefit the least from oral azacitidine include patients with an MDR-negative status, patients with low-risk features for relapse, patients who develop unacceptable toxicities, and patients lacking the social and/or medical support necessary to be safely treated with oral azacitidine (e.g., those with no family or friends, living in a remote community without medical support services, or an inability to understand the treatment risks and benefits). The clinical experts agreed that patients would be identified as possible candidates for oral azacitidine treatment if they are considered ineligible for transplantation. The transplantation ineligibility status may be known in patients at the time of diagnosis or may develop over the course of their treatment.
Assessing Response to Treatment
In the opinion of the clinical experts consulted by CADTH, clinical assessments to evaluate the response to treatment include regular monitoring of blood counts (every 1 to 2 weeks) to determine if a patient maintains CR or CRi. It was noted that 28-day treatment cycles would likely lead to drugs being dispensed 1 month at a time due to drug costs. If changes in a blood count signal a potential relapse (e.g., blood counts drop significantly below the normal range, particularly if they are trending downward) a bone marrow examination may be required to determine if a patient is still in CR or CRi or has relapsed AML. Bone marrow exams are usually not done unless a relapse is suspected. Some clinicians may routinely monitor MRD status using peripheral blood every 3 to 6 months; however, this is not currently part of standard of care.
The clinical experts indicated that the most clinically meaningful responses to treatment include prolonged OS and RFS while maintaining or improving HRQoL and reducing symptom burden. No increase of drug-related toxicities, such as infections, neutropenia, and thrombocytopenia, was also noted as a clinically meaningful outcome.
Discontinuing Treatment
In the opinion of the clinical experts consulted by CADTH, maintenance therapy with oral azacitidine should be discontinued if a patient experiences relapsed AML, has a markedly impaired performance status, or is intolerant to or experiences unacceptable toxicity from oral azacitidine. If AML recurs with blast counts of 5% or greater in the peripheral blood or bone marrow, oral azacitidine should be discontinued. The clinical experts agreed that there is currently insufficient evidence to recommend dose escalation as attempted in the QUAZAR AML-001 trial when AML recurs with blast levels reaching between 5% and 15% in either the peripheral blood or bone marrow. It was stated that the aim of dose escalation is to reinitiate remission, which is beyond the scope of the requested reimbursement criteria, which are for oral azacitidine maintenance therapy.
Prescribing Conditions
In the opinion of the clinical experts consulted by CADTH, oral azacitidine is a drug that is self-administered in a patient’s home. Monitoring of blood work and evaluation for toxicity from treatment, which may warrant dose modification, is required. The clinical experts agreed that patients should have access to a regional cancer clinic to ensure that treatment tolerance is confirmed, and that the disease has not relapsed.
Clinician Group Input
This section was prepared by CADTH staff based on the input provided by clinician groups.
Input was received from 2 clinician groups, the OH-CCO H-DAC and the ATB-MPG, on the reimbursement review of oral azacitidine (Onureg) for the maintenance therapy in adult patients with AML who achieved CR or CRi following induction therapy with or without consolidation treatment and who are not eligible for HSCT.
The clinicians from the OH-CCO H-DAC noted that they provide evidence-based clinical and health system guidance on drug-related issues in support of the OH-CCO mandate, which includes the Provincial Drug Reimbursement Programs and the Systemic Treatment Program. Information for this review was jointly discussed at an H-DAC meeting.
The ATB-MPG is a group of physicians who treat myeloid malignancies and acute leukemias within Alberta and functions within the Alberta Hematology Tumour Group. Members meet weekly as Edmonton and Calgary groups to discuss patient cases and upcoming and open clinical trials. They also meet quarterly to discuss clinical trials, the streamlining of patient care across the province, and improvements to the diagnosis, management, and follow-up of patients with myeloid diseases. The group meets annually to update Alberta provincial treatment guidelines. Information for this review was gathered through a review of publications and group discussions.
Unmet Needs
The ATB-MPG noted that a majority of new AML cases are diagnosed in patients who are elderly and/or comorbid. The group added that approximately 80% of patients are diagnosed with intermediate or high-risk AML. Although these patients will often achieve remission with induction chemotherapy, they will invariably relapse without further chemotherapy to consolidate and maintain remission. Allogeneic stem cell transplantation, which offers a potential cure, is standard-of-care post-remission therapy in patients who are fit and younger than 70 years old. The ATB-MPG noted that, because of the serious and prohibitive morbidity and mortality associated with transplantation in older or comorbid patients, many of these patients are not eligible for transplantation.
Both clinician groups agreed that there are currently no standard funded maintenance regimens specific for patients with AML who are in first remission following induction therapy with or without consolidation treatment and who are not eligible for transplantation. The clinicians from the OH-CCO H-DAC added that, in the absence of proven treatment options, patients are currently being followed by surveillance. Similarly, clinicians from the ATB-MPG noted that current options for care involve a palliative approach focused on transfusion support, hydroxyurea, treatment for infections, and general symptom-based care. The clinicians from ATB-MPG described the current treatment landscape for these patients as bleak and added that treatment options are needed to offer improved survival and quality of life for these patients, who generally only have months to live once they relapse.
The ATB-MPG added that, based on the results reported in the QUAZAR AML-001 trial, oral azacitidine appeared to be a promising option, potentially addressing a large unmet need in the AML disease space. The ATB-MPG noted that oral azacitidine was associated with a significant prolongation of remission and a significant improvement in OS compared with placebo, as well as being generally well tolerated. The group emphasized that oral azacitidine is an oral medication that offers important quality-of-life and feasibility advantages by avoiding the need for frequent visits to treatment centres for therapy. The ATB-MPG noted oral azacitidine is accessible in Alberta via compassionate access from the drug manufacturer and access is expected to be temporary and cumbersome to navigate and may not be available to all candidate patients. The ATB-MPG added that, given the non-curative nature of nontransplantation approaches and the elderly and/or comorbid nature of this target population, treatments that are easy and convenient to administer and well tolerated are urgently needed to prolong remission and survival without adversely affecting quality of life.
According to the clinicians from the OH-CCO H-DAC, the most important goals that an ideal treatment would address are to prolong life as reflected by the end point of OS and improve quality of life. The clinician group commented that maintenance oral azacitidine demonstrated an OS benefit in a patient population that has not seen the benefit of new therapies since 1973, when 7 + 3 became the standard of care. Similarly, the ATB-MPG noted that prolonging life and remission, as well as improving HRQoL, are the most important treatment goals for the target population. It was noted that improving quality of life includes reducing transfusion needs, hospital admissions, severity of symptoms, adverse events (AEs), and decreasing hospital visits to receive therapy. The latter is particularly important for patients who live far from treatment centres and/or are not independent (i.e., caregivers may struggle to manage the demands of intensive therapies).
Both clinician groups agreed that AML remains a highly “lethal” cancer with poor outcomes in patients who are not eligible for transplantation (including those who do not have an available donor). Therefore, these patients have the greatest unmet need for an intervention. The ATB-MPG noted that, given that the large majority of patients who are newly diagnosed with AML have intermediate- or high-risk disease and that a considerable proportion of these patients are not eligible for transplantation, these represent a substantial cohort of patients with AML who will invariably relapse with a prognosis that is measured in months. The group noted that oral azacitidine would fill a large therapeutic hole in the treatment landscape and has the potential to address a substantial unmet need.
Place in Therapy
With respect to how oral azacitidine would fit into the current treatment paradigm, both clinician groups noted that the treatment would be a standard-of-care maintenance therapy for patients with AML who are in first CR and ineligible for HSCT.
The ATB-MPG added that, because oral azacitidine offers the prospect of prolonged remission and survival, decreased disease-related complications, an acceptable safety profile, and a convenient oral route of administration with the potential for improved quality of life, it could shift the current treatment paradigm for patients for whom few other therapeutic options exist. The group noted that the use of oral azacitidine could result in a substantial reduction in the health care burden to patients, their caregivers, and the health care system as a whole.
The OH-CCO H-DAC clinicians commented that oral azacitidine would not affect the sequencing of available therapies for AML. The ATB-MPG added that no other treatment options are available to recommend for this select group of patients before initiating oral azacitidine. However, the ATB-MPG clinicians noted that patients may have received additional cycles of chemotherapy to consolidate their remission before starting oral azacitidine, as per the eligibility criteria of the QUAZAR AML-001 trial. The ATB-MPG also noted that, after receiving oral azacitidine, patients could be enrolled in a clinical trial if available or be given palliative supportive therapy (transfusion, hydroxy urea, or symptom-driven care) as second-line treatment.
Both clinician groups agreed that the patients best suited for treatment with oral azacitidine would be those who meet the QUAZAR AML-001 trial inclusion criteria; this would include patients who do not have an available donor. The OH-CCO H-DAC clinicians added that, although not included in the pivotal trial, maintenance oral azacitidine would be appropriate for patients younger than 55 years of age. Based on pharmacokinetic data, patients with MDS who have progressed on subcutaneous azacitidine, subsequently received induction chemotherapy with or without consolidation, achieve CR, and are transplantation ineligible, may also benefit from oral azacitidine.
According to the clinicians from OH-CCO’s H-DAC, the patients best suited for treatment with oral azacitidine would be identified through a diagnosis of AML, having received treatment with induction therapy with or without consolidation, having achieved CR, and being ineligible for transplantation. The clinicians added that all transplantation-ineligible patients in first CR would be equally suitable for oral azacitidine. The ATB-MPG clinicians noted that patients would be identified through clinician examination and assessment. Testing is widely available as this is an objectively diagnosed disease. The group added non-candidacy for allogeneic stem cell transplantation would involve formal consultation and assessment to confirm transplantation ineligibility. The ATB-MPG group noted that patients who were eligible for transplantation would be the least suitable for treatment with oral azacitidine. Additionally, treatment of patients who are too elderly or comorbid to be included in the clinical trial for oral azacitidine would be subject to a multifaceted clinical judgment by the treating physician. The group commented that patients who choose not to receive oral azacitidine therapy for personal or other reasons would also not be suitable.
Both clinician groups agreed that it would not be possible to identify patients who are most likely to exhibit a response to treatment with oral azacitidine at this time and there were no subgroups of patients who did not benefit from oral azacitidine in the pivotal trial. The ATB-MPG noted that potential identification of particular subpopulations that may experience greater benefit would require further long-term analyses supported by additional patient and clinical experience with oral azacitidine use.
Assessing Response to Treatment
The OH-CCO H-DAC clinicians noted that outcomes for these patients should be assessed according to usual clinical practice, and that oral azacitidine should be continued until overt clinical relapse. The clinicians added that bone marrow sampling is not necessarily required unless there is suspicion of relapse. According to the clinicians, a clinically meaningful response to treatment would be the absence of relapse. The clinicians commented that treatment response should be assessed through a complete blood count and bone marrow surveillance as per clinician judgment.
The ATB-MPG noted that the following outcomes would be used to determine patients’ response to treatment: bone marrow biopsy to assess disease response, improvement in cytopenia and transfusion needs, decrease in blast counts in peripheral blood, improvement in disease-related symptoms (infections, bleeding, and fatigue), improvement in quality of life (measured subjectively by patient-described experiences and objectively through quality-of-life scores), and improvement in functional status (due to improvements in cytopenias, decreased disease-related complications, and decreased need for hospital admission and outpatient visits for therapy administration, transfusion, or other support).
The clinicians added that these outcomes are aligned with those used in clinical trials, although bone marrow biopsies to assess relapse are more frequently performed in a clinical trial setting.
The ATB-MPG clinicians noted that patients who are undergoing initial therapy will have laboratory work performed weekly to assess transfusion needs and drug side effects. However, the group noted that this monitoring will decrease in frequency to monthly depending on individual patients’ transfusion needs, cytopenias, drug tolerability, and stability of treatment response. The group commented that improvement in cytopenias and transfusion requirements will be monitored as patients proceed through each cycle of treatment. The clinicians added that a marrow biopsy would be performed after 4 to 6 cycles and would act as a definitive indicator of disease response. According to the clinicians, once remission or maximal response is obtained, a repeat bone marrow biopsy would be indicated if there is clinical deterioration, significant cytopenias, or other clinical findings requiring reassessment of disease status.
Discontinuing Treatment
The OH-CCO H-DAC clinicians noted that disease relapse or treatment intolerance should be considered when deciding whether to discontinue treatment with oral azacitidine. The ATB-MPG added that assessment of disease progression as measured by peripheral blood indices and transfusion needs, and definitively by a bone marrow biopsy, is required to determine treatment discontinuation. Additionally, the ATB-MPG noted that treatment intolerance, which could include cytopenias complicated by recurrent severe infections, transfusion needs that do not improve after 4 to 6 cycles of therapy, intolerable adverse effects (i.e., gastrointestinal intolerance), or patient preference are factors to be considered by the treating physician for treatment discontinuation.
Prescribing Conditions
Both clinician groups agreed that outpatient cancer clinics in the community setting where regular laboratory and transfusion support are available are most appropriate for treatment with oral azacitidine as the drug can be taken at home.
Additional Considerations
No additional considerations were identified.
Drug Program Input
Drug programs provide input on each drug being reviewed through CADTH’s reimbursement review processes by identifying issues that may affect their ability to implement a recommendation.
The drug programs indicated that the standard approach for patients who have achieved CR or CRi after potential consolidation therapy is to “watch and wait” without any disease-targeting therapies. It was noted that the Health Canada product monograph2 for oral azacitidine indicates that oral azacitidine is not interchangeable with injectable azacitidine, which is available in generic form. As indicated in the product monograph, oral azacitidine is available as 200 mg and 300 mg tablets in a blister pack containing 7 tablets. The list price per tablet was described as extremely high. Furthermore, oral azacitidine would be supplied in 7-day blister packs and dose adjustments or extending days of treatment (i.e., from days 1 through 14 to days 1 through 21) would have a significant effect on treatment costs or risk of wastage. It was suggested that jurisdictions may wish to limit the quantity dispensed if part fills are mandated (e.g., in 7-day increments versus a full cycle) and that dispensed quantities should align with the timing of clinical assessments and blood work. The drug programs emphasized that, because the most commonly reported serious AE of oral azacitidine is febrile neutropenia, patients may require granulocyte colony-stimulating factor, which will add to the cost of patient treatment. It was noted that several drugs in the AML treatment space have been either reviewed or are currently under review by CADTH; midostaurin, gemtuzumab ozogamicin, and gilteritinib were previously reviewed, and azacitidine (injectable) with venetoclax, low-dose cytarabine with venetoclax, and daunorubicin and cytarabine are currently under review.
The implementation questions and corresponding responses from the clinical experts consulted by CADTH are summarized in Table 4.
Table 4: Summary of Drug Program Input and Clinical Expert Response
Drug program implementation questions | Clinical expert response |
|---|---|
Which induction/consolidation therapies were used in the QUAZAR trial? Which induction and/or consolidation therapies are appropriate and/or acceptable to be eligible for azacitidine maintenance? Would patients who are FLT3 mutation–positive for AML who received midostaurin in combination with induction and/or consolidation chemotherapy be eligible for oral azacitidine maintenance? | In the QUAZAR AML-001 trial, the most commonly received induction therapies were cytarabine, idarubicin, and daunorubicin. The most commonly received consolidation therapies were cytarabine, idarubicin, and daunorubicin. The clinical experts consulted by CADTH agreed that all standard prior therapies currently received by patients in Canada are acceptable to be eligible for oral azacitidine maintenance therapy. The most commonly received induction therapies used in Canada are standard-dose cytarabine with an anthracycline (i.e., 7 + 3) or FLAG (fludarabine + high-dose cytarabine + G-CSF) with or without idarubicin in patients with high-risk disease. The experts felt that it would be reasonable to offer oral azacitidine maintenance to patients who received induction therapy with gemtuzumab ozogamicin, which recently received a positive final pERC recommendation for patients with favourable, intermediate, or unknow risk AML.19 The most commonly received consolidating therapy is high-dose cytarabine. The clinical experts agreed that is would be reasonable to offer oral azacitidine maintenance therapy to patients who are FLT3 mutation–positive and who received midostaurin in combination with induction and/or consolidation chemotherapy. This patient group in first complete remission is at high risk of relapse and there is no biological rationale to assume that outcomes of oral azacitidine would be different in patients with FLT3-positive AML. |
How or when will patients be assessed for possible dose changes (including reductions and/or extending number of treatment days based on clinical response)? | The clinical experts noted that they anticipate patients will have regular clinical assessments and monitoring of blood work every 1 to 2 weeks at the beginning of treatment, moving to once a month at the start of every treatment cycle later on (i.e., the timing of moving assessments to once a month should be at the discretion of the treating physician but will likely occur after 3 to 4 cycles). There is currently insufficient evidence to extend the number of treatment days with oral azacitidine, as the QUAZAR AML-001 trial was not designed to assess the efficacy and safety of oral azacitidine maintenance therapy on an extended dosing schedule. |
The recommended starting dose is 300 mg orally daily on days 1 through 14 of a 28-day treatment cycle. In the QUAZAR AML-001 trial, patients who had evidence of relapse with blast levels ≥ 5% and ≤ 15% in either peripheral blood or bone marrow were eligible for an increase in the number of doses per cycle from 14 days to the first 21 days of each 28-day treatment cycle. Is the increased number of doses recommended for patients losing response, as it is not reflected in the product monograph? | The post-relapse dose escalation in the QUAZAR AML-001 trial explored whether oral azacitidine could be used to reinitiate remission, which is not consistent with the Health Canada indication or the CADTH reimbursement request, which is for azacitidine as maintenance therapy. As noted previously, the clinical experts agreed that they would not attempt dose escalation in clinical practice as there is currently insufficient evidence to generalize the QUAZAR AML-001 trial results to oral azacitidine used to reinitiate remission. |
Patients were excluded from QUAZAR AML-001 if they were candidates for HSCT at the time of study. Should the following patients be eligible for azacitidine maintenance?
| In the QUAZAR AML-001 trial some patients were eligible to undergo subsequent HSCT (6.3% of patients in the oral azacitidine group and 13.7% in the placebo group). The clinical experts consulted by CADTH noted that patients would be identified as possible candidates for oral azacitidine treatment if they are considered ineligible for transplantation. The transplantation ineligibility status may be known in patients at the time of diagnosis or may become apparent over the course of treatment. Occasionally a patient could be reconsidered eligible for HSCT if comorbidities improve or resolve while on maintenance oral azacitidine and could undergo HSCT before or after disease relapse. The QUAZAR AML-001 trial excluded patients younger than 55 years of age and patients with less-than-adequate organ function. The clinical experts consulted by CADTH felt that it would be reasonable to generalize the QUAZAR AML-001 trial results to patients younger than 55 years given the acceptable safety profile of oral azacitidine and the absence of a biological rationale to assume that outcomes of oral azacitidine would be different in younger patients with AML who otherwise meet the trial’s inclusion criteria. |
7 + 3 = 7 days of cytarabine and 3 days of anthracycline therapy; AML = acute myeloid leukemia; FLT3 = FMS-like tyrosine kinase 3; G-SCF = granulocyte colony-stimulating factor; HSCT = hematopoietic stem cell transplant; pERC = CADTH pan-Canadian Oncology Drug Review Expert Review Committee.
Clinical Evidence
The clinical evidence included in the review of oral azacitidine is presented in this section. The systematic review includes pivotal studies provided in the sponsor’s submission to CADTH and Health Canada, as well as those studies that were selected according to an a priori protocol. No indirect or other evidence was identified.
Systematic Review (Pivotal and Protocol-Selected Studies)
Objectives
To perform a systematic review of the beneficial and harmful effects of azacitidine tablets as maintenance therapy for the treatment of AML in adult patients who achieved CR or CRi following induction therapy with or without consolidation treatment and who are not eligible for HSCT.
Methods
Studies selected for inclusion in the systematic review included the pivotal study provided in the sponsor’s submission to CADTH and Health Canada, as well as those meeting the selection criteria presented in Table 5. Outcomes included in the CADTH review protocol reflect outcomes considered to be important to patients, clinicians, and drug plans.
Table 5: Inclusion Criteria for the Systematic Review
Criteria | Description |
|---|---|
Population | Adult patients with AML who achieved CR or CRi following induction therapy with or without consolidation treatment, and who are not eligible for HSCT Subgroups
|
Intervention | Azacitidine 300 mg orally once daily on day 1 through day 14 of repeated 28-day treatment cycles |
Comparator | Watch and wait BSC |
Outcomes | Efficacy outcomes
Harms outcomes TEAEs, AEs, SAEs, WDAEs, deaths due to AE Notable harms and harms of special interest
|
Study designs | Published and unpublished phase III and IV randomized controlled trials |
AE = adverse event; AML = acute myeloid leukemia; BSC = best supportive care; CMML = chronic myelomonocytic leukemia; CR = complete remission; CRi = complete remission with incomplete blood count recovery; ECOG PS = Eastern Cooperative Oncology Group Performance Status; ELN = European LeukemiaNet; HRQoL = health-related quality of life; HSCT = hematopoietic stem cell transplantation; MDS = myelodysplastic syndromes; MRD = measurable residual disease; OS = overall survival; RFS = relapse-free survival; SAE = serious adverse event; TEAE = treatment-emergent adverse event; WDAE = withdrawal due to adverse event.
aThe first edition of the ELN recommendations for diagnosis and management of AML in adults, published in 2010, as updated by the ELN 2017 recommendations.21 The ENL 2017 is a prognostic system for AML, which incorporates cytogenetic and molecular risks. It stratifies patients into the following 3 risk groups: favourable, intermediate, and adverse. Most patients in the favourable risk group can be cured with chemotherapy alone, while most patients in the intermediate and adverse risk groups are considered for HSCT.22
bThese outcomes were identified as being of particular importance to patients in the input received by CADTH from patient groups.
The literature search for clinical studies was performed by an information specialist using a peer-reviewed search strategy according to the Peer Review of Electronic Search Strategies checklist (https://www.cadth.ca/resources/finding-evidence/press).23
Published literature was identified by searching the following bibliographic databases: MEDLINE All (1946–) via Ovid and Embase (1974–) via Ovid. The search strategy comprised both controlled vocabulary, such as the National Library of Medicine’s MeSH (Medical Subject Headings), and keywords. The main search concepts were Onureg, oral azacitidine, and acute myeloid leukemia. Clinical trials registries searched included the US National Institutes of Health’s clinicaltrials.gov, WHO’s International Clinical Trials Registry Platform search portal, Health Canada’s Clinical Trials Database, and the European Union Clinical Trials Register.
Search filters were applied to limit retrieval to randomized controlled trials or controlled clinical trials. Retrieval was not limited by publication date or language. Conference abstracts were excluded from the search results. Appendix 1 provides detailed search strategies.
The initial search was completed on March 25, 2021. Regular alerts updated the search until the meeting of the CADTH pan-Canadian Oncology Drug Review Expert Committee (pERC) on August 11, 2021.
Grey literature (literature that is not commercially published) was identified by searching relevant websites from the Grey Matters: A Practical Tool For Searching Health-Related Grey Literature checklist (https://www.cadth.ca/grey-matters).24 Included in this search were the websites of regulatory agencies (US FDA and European Medicines Agency [EMA]). Google was used to search for additional internet-based materials. See Appendix 1 for more information on the grey literature search strategy.
These searches were supplemented by reviewing bibliographies of key papers and through contacts with appropriate experts. In addition, the sponsor of the drug was contacted for information regarding unpublished studies.
Two CADTH clinical reviewers independently selected studies for inclusion in the review based on titles and abstracts, according to the predetermined protocol. Full-text articles of all citations considered potentially relevant by at least 1 reviewer were acquired. Reviewers independently made the final selection of studies to be included in the review, and differences were resolved through discussion.
A focused literature search for network meta-analyses dealing with acute myeloid leukemia was run in MEDLINE All (1946–) on March 26, 2021. No language or date limits were applied.
Findings From the Literature
Six reports presenting data from 1 unique study were identified from the literature for inclusion in the systematic review (Figure 1). The included study is summarized in Table 6. A list of excluded studies is presented in Appendix 2.
Table 6: Details of the Included Study
Detail | QUAZAR AML-001 |
|---|---|
Design and population | |
Study design | Phase III, ongoing, double-blind, placebo-controlled, multi-centre RCT |
Locations | Patients randomized at 147 sites across 23 countries:
|
Patient enrolment dates | First patient enrolled: May 10, 2013 Enrolment end date: Sponsor noted that the final patient enrolment date cannot presently be confirmed12 |
Data cut-off dates | Interim futility analysis: December 31, 2015 Final data cut for primary outcome measure: July 15, 2019 Updated data cut: September 202025 Estimated study completion date (last patient’s last visit): December 31, 2021 |
Randomized (N) | 472 patients
|
Inclusion criteria |
|
Exclusion criteria |
|
Exclusion criteria (continued) |
|
Drugs | |
Intervention | Azacitidine: 300 mg administered orally once day for 14 days of each 28-day treatment cycle. Treatment should continue until disease relapse (> 15% blasts in the bone marrow or peripheral blood), or unacceptable toxicity |
Comparator(s) | Placebo: Placebo orally once daily for 14 days of each 28-day treatment cycle. Treatment should continue until disease relapse (> 15% blasts in the bone marrow or peripheral blood) or unacceptable toxicity |
Best supportive care | Best supportive care used in combination with either oral azacitidine or placebo as judged necessary; best supportive care included but was not limited to treatment with transfusions of packed red blood cells or whole blood, single-donor or pooled-donor platelet transfusions, use of ESAs and other red blood cell hematopoietic growth factors, antibiotic, antiviral and/or antifungal therapy, G-CSFs, and nutritional support |
Outcomes | |
Primary end point | OS |
Secondary and exploratory end Points | Secondary
Exploratory
|
Secondary and exploratory end Points (continued) | Safety
|
Notes | |
Publicationsb | Wei et al., 2020 – Final analysis results (data cut-off date: July 15, 2019) Clinicaltrials.gov (NCT01757535)13 |
AE = adverse event; AML = acute myeloid leukemia; CML = chronic myeloid leukemia; CMML = Chronic myelomonocytic leukemia; CR = complete remission; CRi = complete remission with incomplete blood count recovery; ECOG = Eastern Cooperative Oncology Group; EQ-5D-3L = EuroQol 5-Dimensions 3-Levels questionnaire; ESAs = erythropoiesis-stimulating agent; FACIT-F = Functional Assessment of Chronic Illness Therapy–Fatigue; FAB M3 = French-American-British classification of AML, acute promyelocytic leukemia (M3); G-CSF = granulocyte colony-stimulating factor; HMA = hypomethylating agent; HRQoL = health-related quality of life; MDS = myelodysplastic syndrome; MRD = minimal residual disease; OS = overall survival; PINR = Physical Impairment Numeric Rating; RCT = randomized controlled trial; RFS = relapse-free survival; TEAE = treatment-emergent adverse event.
aSpecific biomarkers were not specified a priori in the statistical analysis plan.10
bFour additional reports were included: the Clinical Study Report9 and the health-related quality of life report14 from the submission to CADTH, the Health Canada reviewer report11 received as requests for additional information made to the sponsor, and the FDA report.26
Description of Studies
The QUAZAR AML-001 trial is an ongoing, international, multi-centre, double-blind, placebo-controlled, randomized phase III trial that compared the efficacy and safety of maintenance therapy with oral azacitidine plus BSC compared with placebo plus BSC in patients with AML in first CR. The primary objective of the trial was to compare OS between maintenance therapy with oral azacitidine and placebo in patients with AML, aged 55 years or older who had achieved first CR or CRi after induction with intensive chemotherapy with or without consolidation chemotherapy.10 Patients in this international trial were randomized at 147 sites across 23 countries, which are listed in Table 6. The majority of sites were in North America and Europe, including 5 sites in Canada.9 According to the sponsor, | Canadian patients were randomized in the QUAZAR AML-001 trial.27
A total of 472 patients were randomized (using an interactive voice response system) in a 1:1 ratio to receive maintenance oral azacitidine plus BSC or oral placebo plus BSC. Enrolment began on May 10, 2013; the enrolment end date has not been determined.12 Best supportive care used in combination with either oral azacitidine or placebo was administered as needed. The BSC products were recorded as concomitant medications, therapies, or procedures. A description of BSC is provided in Table 6.10 Randomization was stratified by age at the time of induction therapy (55 to 64 years and ≥ 65 years), prior history of MDS or CMML (yes/no), cytogenetic risk category at time of induction therapy (intermediate/poor risk), and receipt of consolidation therapy following induction (yes/no). The study design is depicted in Figure 6.
The study consisted of 3 phases: pre-randomization (screening), treatment, and follow-up. No crossover between treatment groups was allowed at any point during the study and blinding was upheld until study closure and database lock.10 During the follow-up phase, patients were monitored for survival every month for the first year and then every 3 months. An extension phase was added to the trial as protocol amendment 2 to ensure drug access for patients who continued to benefit from azacitidine as well as for the collection of survival data after study closure.10 Data from the extension phase are not included in this review.
Response assessment, according to AML response criteria defined by the International Working Group (IWG) for maintaining CR or CRi was planned to occur every 3 cycles starting at cycle 3 until cycle 24, at cycles 30 and 36, and at the treatment discontinuation visit. After cycle 36, response assessments occurred at the investigator’s discretion.9 Relapse was defined as the appearance of a blast level of at least 5% in peripheral blood or bone marrow.11 A post-relapse dose escalation (300 mg once daily from 14 days to 21 days of the 28-day cycle) was planned for patients with disease relapse with a blast level of between 5% and 15% in either the peripheral blood or bone marrow.9 This dose escalation was used in the context of re-induction of remission, which is not consistent with the Health Canada indication and the requested reimbursement criteria, which are for oral azacitidine as maintenance therapy.11 The QUAZAR AML-001 trial was not designed to assess if dose escalation of oral azacitidine produces benefits for patients. Dose escalation has not been authorized by Health Canada according to the Health Canada product monograph.11
The primary analysis, which coincided with the final data cut-off date (July 15, 2019) was conducted once the total number of events (n = 330 deaths) occurred.9,10 The key secondary and other secondary end points were also planned to be assessed at the final data cut-off date. An updated analysis, which was not planned a priori in the statistical analysis plan, occurred based on a September 2020 data cut-off date. Data from this updated data cut-off date have not been provided by the sponsor to CADTH at the time of this review.12 The trial is still ongoing with an estimated completion date of December 31, 2021.13 The QUAZAR AML-001 trial was sponsored by Celgene (a wholly owned subsidiary of Bristol Myers Squibb).10
Populations
Inclusion and Exclusion Criteria
The key inclusion and exclusion criteria used in the QUAZAR AML-001 trial are described in Table 6. Briefly, the trial enrolled adults, 55 years of age and older diagnosed with AML or AML secondary to prior MDS or CMML, with intermediate- or poor-risk cytogenetics, and who had achieved first remission (CR or CRi) following induction with or without consolidation chemotherapy. Patients were excluded if they were considered eligible for HSCT. Transplantation eligibility criteria were not predefined in the trial protocol. At screening patients had to have an ECOG PS of 0 to 3 and adequate marrow function (based on area under the curve for ANC ≥ 0.5 × 109/L and a platelet count ≥ 20 × 109/L).10
Baseline Characteristics
The baseline characteristics of patients who comprised the intention-to-treat (ITT) population of the QUAZAR AML-001 trial are summarized in Table 7. In the ITT population the mean age was 68.0 (SD = 5.66). The oral azacitidine group had a lower proportion (versus placebo) of male patients (49.6% versus 54.3%) and patients enrolled in trial sites in Asia (2.5% versus 7.3); and a higher proportion of White patients (90.8% versus 84.2%) and patients enrolled in trial sites in Europe (70.2% versus 62.8%). A total of 90.9% of patients had de novo AML, and the most common initial AML classification was AML not otherwise specified (62.2% versus 62.0%). The majority of patients had an ECOG PS of 0 (48.7% versus 47.4%) or 1 (42.4% versus 45.3%) and intermediate cytogenetic risk (85.3% versus 86.8%). Subsequent to induction therapy 80.1% of patients received consolidation therapy, with most of these patients receiving 1 cycle (46.2% versus 43.6%) or 2 cycles (29.4% versus 32.9%). The most common reason for transplantation ineligibility was age (64.7% versus 65.0%), followed by comorbidities (21.8% versus 21.4%), and no available donor (15.5% versus 15.0%). The oral azacitidine group had a lower proportion of patients with a negative MRD status at randomization compared with the placebo group (55.9% versus 47.4%) and of patients who achieved CR after induction therapy with or without consolidation (78.6% versus 84.2%).9 The clinical experts consulted by CADTH noted that the differences observed in patient characteristics at baseline between the oral azacitidine and the placebo groups are unlikely to have had an impact on the treatment differences observed between the treatment groups.
Table 7: Summary of Baseline Characteristics — Intention-to-Treat Population
Characteristic | QUAZAR AML-001 | ||
|---|---|---|---|
Oral azacitidine N = 238 | Placebo N = 234 | Total N = 472 | |
Age (years) | |||
Mean (SD) | 67.9 (5.72) | 68.0 (5.62) | 67.9 (5.66) |
Median (minimum to maximum) | 68.0 (55 to 86) | 68.0 (55 to 82) | 68.0 (55 to 86) |
Age (years), n (%) | |||
≥ 55 to < 65 | 66 (27.7) | 68 (29.1) | 134 (28.4) |
≥ 65 to < 75 | 144 (60.5) | 142 (60.7) | 286 (60.6) |
≥ 75 | |||||||||||||| | |||||||| | |||||||||||||| |
≥ 85 | |||||||||||||| | |||||||| | |||||||||||||| |
Sex, n (%) | |||
Male | 118 (49.6) | 127 (54.3) | 245 (51.9) |
Female | 120 (50.4) | 107 (45.7) | 227 (48.1) |
Race, n (%) | |||
White | 216 (90.8) | 197 (84.2) | 413 (87.5) |
Black or African-American | 2 (0.8) | 6 (2.6) | 8 (1.7) |
Asian | 6 (2.5) | 20 (8.5) | 26 (5.5) |
Other | 12 (5.0) | 11 (4.7) | 23 (4.9) |
Missing | 2 (0.8) | 0 | 2 (0.4) |
Geographical region,a n (%) | |||
North America | 37 (15.5) | 42 (17.9) | 79 (16.7) |
Europe | 167 (70.2) | 147 (62.8) | 314 (66.5) |
Asia | 6 (2.5) | 17 (7.3) | 23 (4.9) |
Australia | 26 (10.9) | 23 (9.8) | 49 (10.4) |
South America | 2 (0.8) | 5 (2.1) | 7 (1.5) |
Initial AML classification, n (%) | |||
AML with recurrent genetic abnormalities | 39 (16.4) | 46 (19.7) | 85 (18.0) |
AML with myelodysplasia-related changes | 49 (20.6) | 42 (17.9) | 91 (19.3) |
Therapy-related myeloid neoplasms | 2 (0.8) | 0 | 2 (0.4) |
AML, not otherwise specified | 148 (62.2) | 145 (62.0) | 293 (62.1) |
Missing | 0 | 1 (0.4) | 1 (0.2) |
Type of AML, n (%) | |||
Primary (de novo) | 213 (89.5) | 216 (92.3) | 429 (90.9) |
Secondary | 25 (10.5) | 18 (7.7) | 43 (9.1) |
Time since original AML diagnosis (months) to randomization | |||
Mean (SD) | 4.37 (1.273) | 4.30 (1.246) | 4.33 (1.259) |
Median (range) | 4.21 (1.5 to 9.2) | 4.17 (1.4 to 10.9) | 4.21 (1.4 to 10.9) |
Prior history of MDS/CMML, n (%) | |||
Yes | 22 (9.2) | 17 (7.3) | 39 (8.3) |
Primary | 20 (8.4) | 17 (7.3) | 37 (7.8) |
Secondary | 0 | 0 | 0 |
Missing | 2 (0.8) | 0 | 2 (0.4) |
ECOG PS, n (%) | |||
Grade 0 | 116 (48.7) | 111 (47.4) | 227 (48.1) |
Grade 1 | 101 (42.4) | 106 (45.3) | 207 (43.9) |
Grade 2 | 21 (8.8) | 15 (6.4) | 36 (7.6) |
Grade 3 | 0 | 2 (0.9) | 2 (0.4) |
Cytogenetic risk category at diagnosis, n (%) | |||
Intermediate | 203 (85.3) | 203 (86.8) | 406 (86.0) |
Poor | 35 (14.7) | 31 (13.2) | 66 (14.0) |
MRD status at randomization,b n (%) | |||
Negative | 133 (55.9) | 111 (47.4) | 244 (51.7) |
Positive | 103 (43.3) | 116 (49.6) | 219 (46.4) |
Missing | 2 (0.8) | 7 (3.0) | 9 (1.9) |
Reason ineligible for transplantation,c n (%) | |||
Age | 154 (64.7) | 152 (65.0) | 306 (64.8) |
Comorbidities | 52 (21.8) | 50 (21.4) | 102 (21.6) |
Performance status | 14 (5.9) | 9 (3.8) | 23 (4.9) |
Not acceptable or available donor | 37 (15.5) | 35 (15.0) | 72 (15.3) |
Patient decision | 19 (8.0) | 32 (13.7) | 51 (10.8) |
Unfavourable cytogenetics | 6 (2.5) | 10 (4.3) | 16 (3.4) |
Other | 28 (11.8) | 21 (9.0) | 49 (10.4) |
Received consolidation therapy following induction therapy | |||
Yes | 186 (78.2) | 192 (82.1) | 378 (80.1) |
1 cycle | 110 (46.2) | 102 (43.6) | 212 (44.9) |
2 cycles | 70 (29.4) | 77 (32.9) | 147 (31.1) |
3 cycles | 6 (2.5) | 13 (5.6) | 19 (4.0) |
4 cycles | 0 | 0 | 0 |
No | 52 (21.8) | 42 (17.9) | 94 (19.9) |
Response achieved after induction therapy (with or without consolidation therapy), n (%) | |||
CR | 187 (78.6) | 197 (84.2) | 384 (81.4) |
CRi | 51 (21.4) | 37 (15.8) | 88 (18.6) |
CR or CRi status at randomization,d n (%) | |||
CR | 183 (76.9) | 177 (75.6) | 360 (76.3) |
CRi | 50 (21.0) | 44 (18.8) | 94 (19.9) |
Not in CR or CRi | 5 (2.1) | 11 (4.7) | 16 (3.4) |
Missing | 0 | 2 (0.9) | 2 (0.4) |
Time from start of induction therapy to randomization (months) | |||
N | |||||||||||||| | |||||||| | |||||||||||||| |
Mean (SD) | |||||||||||||| | |||||||| | |||||||||||||| |
Median (range) | 3.98 (1.4 to 8.8) | 4.01 (1.3 to 15.1) | 3.98 (1.3 to 15.1) |
Time from induction therapy to first achieving CR or CRi to (days) | |||
N | |||||||||||||| | |||||||| | |||||||||||||| |
Mean (SD) | |||||||||||||| | |||||||| | |||||||||||||| |
Median (range) | 36.0 (13 to 242) | 35.0 (14 to 455) | 35.0 (13 to 455) |
Time since first achieving CR or CRi to randomization (days) | |||
N | |||||||||||||| | |||||||| | |||||||||||||| |
Mean (SD) | |||||||||||||| | |||||||| | |||||||||||||| |
Median (range) | 84.5 (7 to 154) | 86.0 (7 to 263) | 85.0 (7 to 263) |
Bone marrow blasts (%) | |||
N | |||||||||||||| | |||||||| | |||||||||||||| |
Mean (SD) | |||||||||||||| | |||||||| | |||||||||||||| |
Median (range) | 2.00 (0.0 to 5.0) | 2.00 (0.0 to 6.5) | 2.00 (0.0 to 6.5) |
Peripheral blood blasts (%) | |||
N | |||||||||||||| | |||||||| | |||||||||||||| |
Mean (SD) | |||||||||||||| | |||||||| | |||||||||||||| |
Median (range) | |||||||||||||| | |||||||| | |||||||||||||| |
AML = acute myeloid leukemia; CMML = chronic myelomonocytic leukemia; CR = complete remission; CRi = complete remission with incomplete blood count recovery; ECOG = Eastern Cooperative Oncology Group; MDS = myelodysplastic syndrome; MRD = minimal residual disease; SD = standard deviation.
Note: Time interval in days was calculated as the difference between the randomization date and the date of interest (e.g., date of original AML diagnosis) plus 1 day. Time interval presented in month is transformed from days to months by using conversion formula: months = days/30.4375.
aNorth America includes Canada, Mexico, and the US; Asia includes South Korea and Taiwan; Australia includes Australia; Europe includes Austria, Belgium, Czech Republic, Finland, France, Germany, Ireland, Israel, Italy, Lithuania, Poland, Portugal, the Russian Federation, Spain, and Turkey; South America includes Brazil.
bDuring the screening period.
cA patient may have had more than 1 reason.
dCR or CRi at randomization was programmatically derived based on IWG AML response criteria using bone marrow data collected during screening, and ANC and platelet counts closest to randomization date. For a subject with a bone marrow blast level of less than 5%, and both ANC less than 1.0 × 109/L and platelet count less than 100 × 109/L, the subject was considered not in CR or CRi.
Source: Clinical Study Report.9
All patients in the ITT population received at least 1 induction therapy for AML before enrolment into the trial, and the proportions and types of therapies were balanced across groups as summarized in Table 8. The most commonly received induction therapy (oral azacitidine versus placebo) was cytarabine (99.6% versus 99.1%) followed by idarubicin (54.2% versus 55.6% placebo), and daunorubicin (33.2% versus 32.9%).9
Table 8: Prior Induction Therapies for AML Reported for More Than 1.0% of Patients — Intention-to-Treat Population
ATC dictionary-level preferred namea | QUAZAR AML-001 | ||
|---|---|---|---|
Oral azacitidine N = 238 | Placebo N = 234 | Total N = 472 | |
Patient with at least 1 prior induction therapy for AML, n (%) | 238 (100.0) | 234 (100.0) | 472 (100.0) |
Antineoplastic and immunomodulating agents | 238 (100.0) | 234 (100.0) | 472 (100.0) |
|||||||||| | |||||| | |||||| | |||||| |
|||||||||| | |||||| | |||||| | |||||| |
|||||||||||||| | |||||| | |||||| | |||||| |
|||||||||||| | |||||| | |||||| | |||||| |
|||||||||||| | |||||| | |||||| | |||||| |
|||||||||| | |||||| | |||||| | |||||| |
|||||||||||||||||||| | |||||| | |||||| | |||||| |
|||||||||||||||||||||||||||||||| | |||||| | |||||| | |||||| |
|||||||||||||||||||||||||||||| | |||||| | |||||| | |||||| |
|||||||||||||||||||||||||||||||| | |||||| | |||||| | |||||| |
|||||||| | |||||| | |||||| | |||||| |
||||||||||||||||||||||| | |||||| | |||||| | |||||| |
AML = acute myeloid leukemia; ATC = Anatomical Therapeutic Chemical.
aCoded using WHODrug version March 2019. A subject with multiple occurrences of a drug class or drug is counted only once in the specific ATC classification or preferred name, respectively.
Source: Clinical Study Report.9
In the ITT population, most of the patients received at least 1 consolidation therapy for AML before enrolment into the trial, and the proportions and types of therapies were balanced across groups as summarized in Table 9. The most commonly received consolidation therapy was cytarabine (78.2% oral azacitidine; 81.6% placebo) followed by idarubicin (21.4% oral azacitidine; 18.8% placebo), and daunorubicin (9.2% oral azacitidine; 6.4% placebo). Most of the patients received 1 cycle (46.2% oral azacitidine; 43.6% placebo) or 2 cycles (29.4% oral azacitidine; 32.9% placebo) (Table 9).9
Table 9: Prior Consolidation Therapies — Intention-to-Treat Population
ATC dictionary-level preferred namea | QUAZAR AML-001 | ||
|---|---|---|---|
Oral azacitidine N = 238 | Placebo N = 234 | Total N = 472 | |
Patients with at least 1 prior consolidation therapy for AML, n (%) | 186 (78.2) | 192 (82.1) | 378 (80.1) |
Antineoplastic and immunomodulating agents | 186 (78.2) | 192 (82.1) | 378 (80.1) |
|||||||||| | |||||| | |||||| | |||||| |
|||||||||| | |||||| | |||||| | |||||| |
|||||||||||||| | |||||| | |||||| | |||||| |
|||||||||||| | |||||| | |||||| | |||||| |
|||||||||||| | |||||| | |||||| | |||||| |
|||||||||| | |||||| | |||||| | |||||| |
|||||||||||||||||||||||||||||| | |||||| | |||||| | |||||| |
|||||||||| | |||||| | |||||| | |||||| |
|||||||||||||||||||||||||||||||||||||||||||| | |||||| | |||||| | |||||| |
|||||||||| | |||||| | |||||| | |||||| |
|||||||||| | |||||| | |||||| | |||||| |
|||||||||||| | |||||| | |||||| | |||||| |
|||||||||||||||||||||||||||||||| | |||||| | |||||| | |||||| |
|||||||||||| | |||||| | |||||| | |||||| |
|||||||||||||||||||||||||||||||| | |||||| | |||||| | |||||| |
|||||||| | |||||| | |||||| | |||||| |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||| | |||||| | |||||| | |||||| |
|||||||||||||||||| | |||||| | |||||| | |||||| |
AML = acute myeloid leukemia; ATC = Anatomical Therapeutic Chemical.
aCoded using WHODrug version March 2019. A patient with multiple occurrences of a drug class or drug is counted only once in the specific ATC classification or preferred name, respectively.
Source: Clinical Study Report.9
Interventions
Patients randomized in the QUAZAR AML-001 trial were allocated to either the oral azacitidine plus BSC group or the placebo plus BSC group. Treatments and doses are described in Table 10.
Table 10: Treatment Regimens in the QUAZAR AML-001 Trial
Regimen | Oral azacitidine | Placebo |
|---|---|---|
Dose | Oral azacitidine: 300 mg administered orally (tablets) once daily for the first 14 days of each 28-day treatment cycle BSC: Used as needed; BSC includes (but is not limited to):
| Placebo: Matching placebo tablets administered orally once daily for the first 14 days of each 28-day treatment cycle BSC: Used as needed; includes (but is not limited to):
|
Treatment discontinuation | Treatment was discontinued when the following criteria were met:
| Treatment was discontinued when the following criteria were met:
|
AE = adverse event; BSC = best supportive care; CR = complete remission; CRi = complete remission with incomplete blood count recovery; G-CSFs = granulocyte colony-stimulating factors; pRBC = packed red blood cell; RBC = red blood cell.
Source: Wei et al. (2020).10
Dose Escalation in Patients With Disease Relapse
A dose escalation (300 mg once daily from 14 days to 21 days of the 28-day cycle) was planned for all patients with disease relapse with blasts between 5% and 15% either in the peripheral blood or bone marrow at the discretion of the investigator and under the following criteria10:
For patients currently on blinded treatment with oral azacitidine 300 mg or placebo once daily for 14 days: Escalate schedule to oral azacitidine 300 mg or placebo once daily for 21 days
For patients currently on blinded treatment of oral azacitidine 200 mg or placebo once daily for 14 days in the event that the dosage had previously been reduced: Escalate schedule as a first step to 21 days once daily and then escalate dosage to oral azacitidine or placebo to 300 mg once daily for 21 days
For patients currently on blinded treatment with oral azacitidine 200 mg or placebo once daily for 7 days: Escalate schedule as a first step, to oral azacitidine 200 mg or placebo once daily for 14 days and then escalate dosage to oral azacitidine 300 mg or placebo once daily for 14 days.
Dose Modifications
Dose interruptions, delays, or modifications were permitted per guidelines of version 4.0 of the National Cancer Institute Common Terminology Criteria for Adverse Events for the grading of severity.10 A maximum of 1 dose reduction (to a daily dose of 200 mg) was permitted. One treatment schedule modification was allowed from 14 to 7 days (21 days to 14 days for patient on dose escalation). Patients could not receive less than 200 mg or be on a treatment schedule for less than 7 days. Once a dose or schedule modification occurred, titration back up to the original dose or schedule was permitted.10
Concomitant Medication
Best supportive care was documented as concomitant medication in the trial. The following BSC medications were allowed (but not limited to): red blood cell and platelet transfusions, use of an erythropoiesis-stimulating drug, antibiotic, antiviral, and antifungal therapy, nutritional support, and granulocyte colony-stimulating factor for patients experiencing neutropenic infections.10
Outcomes
A list of the efficacy end points identified in the CADTH review protocol that were assessed in the clinical trials included in this review is provided in Table 11 and summarized in the following section. A detailed discussion and critical appraisal of the HRQoL and symptom severity outcomes is provided in Appendix 4.
Table 11: Summary of Outcomes of Interest Identified in the CADTH Review Protocol
Outcome measure | QUAZAR AML-001 |
|---|---|
Overall survival | Primary |
Relapse-free survival | Key secondary |
Time to relapse | Additional secondary |
Time to discontinuation from treatment | Additional secondary |
Health-related quality of life | |
EQ-5D-3L | Additional secondary |
PINR scale | Exploratory |
Symptom severity | |
FACIT-F | Additional secondary |
Measure of health care resource utilization | |
Hospitalization | Additional secondary |
Days of hospitalizations | Additional secondary |
Days of hospitalizations per person-year of exposure | Additional secondary |
Relative risk of hospitalizations | Additional secondary |
Safety | |
Type, frequency, severity, and relationship of AEs to study treatments | Safety |
EQ-5D-3L = EuroQol 5-Dimensions 3-Levels questionnaire; FACIT-F = Functional Assessment of Chronic Illness Therapy–Fatigue; PINR = Physical Impairment Numeric Rating.
Source: Wei et al. (2020).10
Overall Survival
Overall survival was the primary outcome of the QUAZAR AML-001 trial and was defined as the time from the date of randomization until the date of death due to any cause.10
Relapse-Free Survival
Relapse-free survival was designated the key secondary outcome and defined for patients who achieved CR or CRi and measured as the time from the date of randomization until the date of documented relapsed disease or death by any cause, whichever occurred first.10 Disease relapse for the analysis of RFS was determined according to the IWG AML response criteria: relapse after CR or CRi was defined as reappearance of leukemic blasts in the peripheral blood of 5% or more in the bone marrow not attributable to any other cause (e.g., bone marrow regeneration after consolidation therapy). In the setting of recent treatment, if there were no circulating blasts and the bone marrow contained 5% to 20% blasts, a repeat bone marrow sample taken at least 1 week later was necessary to distinguish relapse from regeneration. A separate central review (blinded to patients’ treatment assignment) was used to confirm all clinical laboratory value assessments.10 A sensitivity analysis was conducted to assess RFS based on investigator-assessed response.10
Time to Relapse From CR or CRi
Time to relapse from CR or CRi was designated an additional secondary outcome and was defined as the interval from the date of randomization to the date of documented relapse after CR or CRi, as determined according to the IWG’s AML response criteria.10
Time to Discontinuation From Treatment
Time to discontinuation from treatment was designated an additional secondary outcome and was defined as the interval from the date of randomization to the date of discontinuation from investigational product.10
Health-Related Quality of Life
The HRQoL outcomes measured in the trial included the EQ-5D-3L instrument and the PINR scale. The EQ-5D-3L instrument was an additional secondary outcome, whereas the PINR scale was an exploratory outcome.10 The primary analysis of the EQ-5D-3L assessed the treatment-effect difference between oral azacitidine and placebo on the mean change from baseline. The secondary analyses of the EQ-5D-3L questionnaire evaluated the proportion of patients with a clinically meaningful deterioration at a given post-baseline assessment visit, as well as the time to a definitive deterioration (defined as a clinically meaningful deterioration for at least 2 consecutive assessment visits).14 The sponsor defined a clinically meaningful deterioration as a decline of at least 0.10 and 11 points on the EQ-5D-3L health utility index and the EQ VAS, respectively.14 The EQ-5D-3L was assessed on day 1 of each 28-day cycle until discontinuation of study treatment and at the end-of-treatment visit.14 A detailed discussion and critical appraisal of the HRQoL measures are provided in Appendix 4.
The EQ-5D-3L is a standardized, patient self-administered questionnaire that consists of 2 parts:
Health utility index: A descriptive section comprising 5 dimensions. Each of the 5 dimensions (anxiety/depression, pain/discomfort, mobility, self-care, usual activities) has 3 response levels reflecting health and well-being (no problems, moderate problems, and extreme problems).14 The health utility index generates a population preference–weighted score in that it reflects societal preferences for the health states.
EQ VAS: A visual analogue scale that provides a quantitative measure by recording a respondent’s self-rated health on a 100-point scale between 100 (“best imaginable health state”) and 0 (“worst imaginable health state”).14 The higher the score the better the imaginable health state. The EQ VAS captures the individual’s value or judgment of his or her present health state.
The validity of the EQ-5D-3L questionnaire was assessed in 168 patients with AML in China.28 Known-group and discriminative validities were assessed and both demonstrated an ability to distinguish groups based on various baseline factors. Construct validity was assessed through the convergent approach, indicating overall good validity. Test-retest reliability was assessed using the intraclass correlation coefficient (ICC), which showed good reproducibility. No relevant studies assessing responsiveness were found. An MID was not identified in the literature for patients with AML. The sponsor referenced studies in the published literature that reported MIDs ranging from 0.06 to 0.12 for the EQ-5D-3L health index score and from 6 to 11 for the EQ VAS in cancer patients.29,30 Other reported MIDs for the health index score range from 0.033 to 0.074 in the general population.31 The sponsor’s submission noted that, although the terms MID and RD have gradually differentiated over time, these terms are still being used interchangeably in the literature.14 The sponsor therefore used the same cut-off values for the RD as for the MID. In addition, the sponsor presented the entire distribution of responses (cumulative distribution frequency curves of patients experiencing different degrees of change at cycles 3, 6, 12, 18, and 24).32
The analysis of the PINR scale assessed the treatment-effect difference between oral azacitidine and placebo on the mean change from baseline.32 The PINR scale was assessed on day 1 of each 28-day cycle until discontinuation of the study treatment and at the end-of-treatment visit.14 The PINR scale is a self-administered patient questionnaire for evaluating the level of physical impairment, difficulty completing outdoor physical tasks, and difficulty completing indoor tasks over the past 24 hours.14 The PINR scale consists of 3 questions, each reflecting the level of impairment and difficulty (0 = “no impairment or not difficulty” and 10 = “worse impairment or extremely difficult”). The higher the score, the greater the difficulty or impairment respondents experienced. Items were scored separately (i.e., each item was scored and not combined into a single overall score). The PINR scale was assessed on day 1 of each 28-day cycle and at the end-of-treatment visit.14
No study was found that assessed the psychometric properties related to validity or reliability for the PINR scale in patients with AML or leukemia. No studies of the validity or reliability of the PINR scale were provided in the sponsor’s submission to CADTH. An MID for the PINR scale was not identified in the literature for AML or leukemia patients. The sponsor’s submission noted that neither the MID nor the responder definition (RD) for the PINR scale were available in the literature.14 The sponsor’s submission reported that half of the SD at baseline from all ITT patients was used as an approximation for the MID.
Symptom Severity
Symptom severity was assessed in the trial using version 4 of the FACIT-F scale and was designated an additional secondary outcome.10 The primary analysis of the FACIT-F scale assessed the treatment-effect difference between oral azacitidine and placebo in the mean change from baseline; the secondary analyses of the FACIT-F scale evaluated the proportion of patients with a clinically meaningful deterioration at a given post-baseline assessment visit and the time to a definitive deterioration.14 Assessments of the FACIT-F scale occurred on day 1 of each 28-day cycle until discontinuation of the study treatment and at the end-of-treatment visit.14 A detailed discussion and critical appraisal of the FACIT-F scale is provided in Appendix 4.
The FACIT-F scale is a standardized, self-administered patient questionnaire for evaluating the level of fatigue in an individual during usual daily activities over the past week.14 The FACIT-F scale consists of a 5-point Likert scale (0 = not at all fatigued to 4 = very much fatigued) and a summary score is calculated based on the FACIT-F scoring guidelines. A higher score corresponds to a lower level of fatigue (scores range from 0 to 52).14
No study assessing the psychometric properties related to the validity or reliability of the FACIT-F scale in patients with AML was found. One study, which assessed the content validity of the FACIT-F scale in 40 patients in the first line or relapsed or refractory chronic lymphocytic leukemia (CLL) setting in the US, found that interviewed patients understood the terminology and agreed with response options.33 The same study investigated structural validity, construct validity, known-groups validity, and internal consistency using data from a phase III RCT (ASCEND)34 in 263 patients with relapsed or refractory CLL. An assessment of structural validity using confirmatory factor analysis (CFA) supported the unidimensionality of the FACIT-F scale.33 Construct validity, as assessed through the convergent approach, indicated overall good validity. Known-groups validity was supported by differences between groups based on various baseline factors. Internal consistency reliability was assessed using Cronbach alpha. The FACIT-F total, symptom, and impact subscales demonstrated good internal consistency.33 No relevant studies were found to assess responsiveness. An MID was not identified in the literature in patients with AML. The sponsor referenced studies in the published literature that reported MIDs ranging from 3 to 4 points in cancer patients.35,36 It was noted in the sponsor’s submission that, although the terms MID and responder definition have gradually differentiated over time, these terms are still being used interchangeably in the literature.14 In consequence, the sponsor used the same cut-off values for the responder definition as for the MID for the FACIT-F scale. In addition, the sponsor presented the entire distribution of responses (cumulative distribution frequency curves of patient experiencing different degrees of change at cycles 3, 6, 12, 18, and 24).14
Measures of Health Care Resource Utilization
Health care resource utilization was designated an additional secondary outcome. Health care resource use was recorded in specifically designed case report forms adapted from the universal billing (UB) 90 billing form and/or through routine study activities. Resource utilization data collected included, but was not limited to, hospitalizations, medications, clinic visits, medical and diagnostic procedures, and treatment for AEs. Health care resource utilization data were collected until the last dose of study treatment or the date of last study visit, whichever occurred later.10
Transfusion Independence
This outcome was not assessed in the QUAZAR AML-001 trial.
Safety
The QUAZAR AML-001 trial included assessment of the following safety parameters: TEAEs (summarized by worst severity grade, system organ class, and preferred term), TEAEs leading to death or to discontinuation from treatment, TEAEs leading to dose modification, AEs classified as grade 3 or grade 4 by the National Cancer Institute Common Terminology Criteria for Adverse Events (Version 4.0), AEs related to investigational product, serious AEs, time to discontinuation from the study due to AE, and deaths.10
All AEs were documented by the investigator from the date a patient signed the informed consent document to 28 days after the last dose of investigational drug or until the date of the last study visit, whichever occurred later.10
The following parameters were monitored: a patient’s clinical signs and symptoms, laboratory, pathological, radiological or surgical findings, physical examination findings; and results of any other appropriate test or procedure.10
Statistical Analysis
Details related to statical analyses of efficacy end points are summarized in Table 12.
Sample Size Determination
The trial sample size was determined based on the comparison of the primary end point of OS between the 2 treatment groups, with approximately 460 patients planned for randomization. The data cut-off for the primary analysis of OS was planned for the time point at which 330 deaths were observed across both treatment groups. If at the time of the primary analysis the average OS HR was 0.70, the study would have 90% power to demonstrate a statistically significant difference in OS at a 1-sided alpha of 0.025. No rationale was provided in the sponsor’s submission for assuming an OS HR of 0.70. An assumption of exponential OS distribution with a constant hazard rate was made with median OS values of 22.9 months and 16 months for the oral azacitidine and placebo groups, respectively.10 No rationale was provided in the sponsor’s submission for assuming a median OS of 22.9 months. However, with regards to the assumption of a median OS of 16 months, the sponsor’s submission referenced 2 studies: Baer et al. (2011)37 reported outcomes for 610 patients aged 60 years or older with untreated de novo and secondary AML who received induction intensive chemotherapy, and Baer et al. (2010)38 provided a historical comparison of patients aged 60 or older with de novo AML who either received 1 course or multiple courses of post-remission consolidation chemotherapy.
Interim Analysis
An interim analysis for futility after 30% of the events (99 deaths) had occurred was planned. The pre-specified futility boundary of a z value of less than −1.9796, using gamma(−10) to correspond to a conditional power level of approximately 18%, was calculated using a beta-spending function.9 At the data cut-off date of December 31, 2015, at which point 104 deaths had occurred, the interim analysis was conducted. The data monitoring committee reviewed unblinded results27 and determined that the HR of oral azacitidine versus placebo remained within the pre-specified futility boundary (the z score was 3.4883 and the P value was 0.0538 based on stratified log-rank test)27 and that no safety concerns were observed.10 Therefore, the data monitoring committee recommended that the study could proceed as planned.10 As the purpose of the interim analysis was to stop the trial for futility, no alpha adjustment was applied in the final analysis.27
Primary Outcome
The difference in OS between the 2 study groups was compared using a log-rank test to assess superiority of oral azacitidine, at a 5% (2-sided) significance level and stratified by age at the time of induction therapy, prior history of MDS, cytogenetic risk category, and receipt of consolidation therapy following induction therapy. If the minimum number of patients in a stratum was less than 16, then only the 3 stratification variables that resulted in the largest minimum stratum size were used for analysis purposes. The HRs and 95% CIs were estimated from a stratified Cox proportional hazards regression model. The proportional hazards assumption was assessed by a time-dependent Cox model with interaction of treatment and time. A Kaplan–Meier (KM) plot of OS was presented by treatment group, unadjusted for stratification variables. Medians and 25th and 75th percentiles with corresponding 95% CIs as well as the numerical difference (and 95% CI difference) between groups were presented. KM estimates of probabilities and corresponding 95% CIs at several time points (i.e., 1 year and 2 year) were also reported. Reasons for censoring included losses to follow-up, withdrawal of consent, or alive at the data cut-off date.10
Several sensitivity analyses for OS were planned as follows:
Investigation of potentially confounding effects of subsequent therapies10:
censoring for the use of any subsequent therapy (including post-treatment transplantation) for AML
censoring for the use of disease-modifying subsequent AML therapy, defined as any subsequent AML therapy that is not hydroxycarbamide
censoring for post-treatment transplantation
a Cox proportional hazard model with adjustment for covariates that included, but was not limited to, subsequent AML therapy as a time-varying covariate and treatment-by-subsequent AML therapy interaction, and baseline characteristics (such as age, ECOG score, cytogenetic risk status, and CR or CRi status at randomization)
inverse probability of censoring weighted (IPCW) method
regression-based imputation analysis method that allows for inferences about the treatment effect in the presence of confounding due to additional therapy received subsequent to the study treatment.
Additional analyses were planned if the proportional hazards assumption was violated. As the proportional hazard assumption did not hold, the following analyses were performed:
restricted mean survival time
a piecewise Cox proportional hazard model with interval time points of 3 or less, 3 to 6, 6 to 12, and greater than 12 months
a generalized Wilcoxon test in the presence of nonproportional and early OS separation
Not censoring for withdrawal of consent
Primary analysis on a modified intention-to-treat population (mITT) population (defined in the section that follows).
Secondary Outcomes
Relapse-free survival, the key secondary outcome in the QUAZAR AML-001 trial, was planned to be analyzed in the same manner as OS. If superiority of oral azacitidine was demonstrated for OS at the 2-sided 0.05 significance level, then RFS was formally tested for statistical significance in the primary analysis. The P value from the stratified log-rank test was the confirmatory P value. This hierarchical testing scheme ensured that the overall alpha level at 0.05 across the OS and RFS end points was preserved.10 Reasons for censoring included no documented relapse and still alive, lost to follow-up, or withdrawal of consent without documented relapse. A date of documented relapse was censored if subsequent therapy for AML was initiated before the date of relapse or if the interval between the date of relapse and the previous bone marrow assessment date or the randomizing date (if no post-baseline bone marrow assessment), was more than 200 days.
Several sensitivity analyses for RFS were planned as follows10:
Primary analysis was performed on the mITT population (defined in the section that follows)
The RFS definition was replaced with the documented relapse based on investigator-assessed response
Censoring was based on EMA guidance, with the following censoring rules26:
for patients who experienced documented relapse or death, define outcome as event and event date as the earliest of documented relapse or death
for patients without documented relapse or death, define outcome as censoring and event date as the date of last bone marrow assessment or date of randomization if no post-baseline bone marrow assessment.
Time to relapse was a descriptive end point that was calculated using a competing risk analysis whereby death without documented relapse was considered a competing risk for relapse. The median time to relapse was calculated using an unstratified KM analysis. A cumulative incidence function, as described by Kalbfleisch and Prentice (1980),39 was calculated and displayed graphically for each treatment group. Censoring rules similar to those used for RFS were applied.10
Time to discontinuation from treatment was a descriptive end point that was calculated using a competing risk model with the following competing events10:
disease relapse
adverse event(s)
eligibility for bone marrow or stem cell transplantation
withdrawal of consent, lost to follow-up, or protocol violation or other event
death.
Cumulative incidence functions for each specific reason for discontinuation from treatment were calculated and displayed graphically for each study group.10 The median time to discontinuation was calculated based on an unstratified KM analysis. The CI for the difference in median times between treatment groups was obtained using the Kosorok method. Treatment discontinuation rates were estimated using KM methods. The CIs for the difference in treatment discontinuation rates were obtained using a Greenwood variance estimate.9 The reason for censoring is ongoing in treatment at the time of study closure.10
All analyses performed on the HRQoL outcomes (EQ-5D-3L and PINR scale) and symptom severity outcome (FACIT-F scale) were based on the July 15, 2019, data cut-off, considered descriptive (i.e., noninferential) in nature, and unadjusted for multiplicity.9,14 The HRQoL-evaluable population (defined as all ITT patients who had a valid [i.e., non-missing] assessment at baseline and at least 1 valid post-baseline assessment) was used to analyze all HRQoL and symptom severity outcomes, with the exception of the calculation of adherence and completion rates, which were based on the ITT population. Statistical tests were conducted at a 2-sided 0.05 level of significance and noninferential P values were presented.14 Point estimates of treatment difference and associated 95% CIs were presented for all end points.14 Missing data at a given assessment visit were not imputed. Missing items for a measure at a given assessment visit were handled according to the scoring guidance of the respective measure.14
In the HRQoL-evaluable population, descriptive statistics (mean SD, median, first quartile, third quartile, minimum, and maximum) were used to summarize observed scores at each assessment visit and the change from baseline.14 For the FACIT-F scale, the EQ-5D-3L instrument (health utility index and EQ VAS) and each of 3 PINR questions, within-group changes from baseline at each post-baseline assessment visit were tested using paired 1-sample t-tests, and between-group differences in changes from baseline were tested using independent 2-sample t-tests.14 Additionally, ANCOVA models were used to assess change from baseline to each post-baseline assessment visit, including covariates (treatment, baseline score, and trial randomization stratification factors). The treatment differences between treatment groups were evaluated using differences in LS mean changes (95% CIs and P values). Plots were generated for each treatment group showing the mean changes (and standard errors) from baseline across all post-baseline assessment visits that had 25 patients or more in each treatment group.14 Furthermore, an MMRM analysis was used to analyze the FACIT-F scale, the EQ-5D-3L instrument (health utility index and EQ VAS), and each of 3 PINR questions using the HRQoL-evaluable population but excluding the end-of-treatment visit. The MMRM analysis controlled for the baseline HRQoL score and other pre-selected covariates of interest. The MMRM analysis was used to assess the overall LS means for change from baseline at each post-baseline assessment visit as well as to calculate the treatment differences between treatment groups using differences in LS mean change (95% CI and P value) at each post-baseline assessment visit. Mixed-effect models with a random intercept or slope were used to assess treatment differences in change scores over time between treatment groups.
To determine if the between-group difference was considered clinically meaningful in the MMRM analysis, the sponsor defined the following MIDs9:
FACIT-F scale: change from baseline of 3 points
EQ-5D-3L health utility index: change from baseline of 0.10 points
EQ VAS: change from baseline of 11 points
PINR scale, physical impairment: change from baseline of 1.12
PINR scale, difficulty completing outdoor physical tasks: change from baseline of 1.59
PINR scale, difficulty completing indoor tasks: change from baseline of 1.28.
As the MID for the PINR scale has not been described in the literature, the sponsor used half of a SD (0.5 × SD) at baseline from all ITT patients as an approximation for the MID.14
To compare the proportions of patients with clinically meaningful change between treatment groups, Cochran-Mantel-Haenszel tests were performed (an odds ratio and a 95% CI with an associated P value were provided). This analysis was performed for the EQ-5D-3L and FACTI-F scale but not for the PINR scale. The sponsor defined a change of 3 points or greater as a clinically meaningful change at the individual level for the FACIT-F scale. For the EQ-5D-3L, a change of 0.10 points or greater from baseline was defined as a clinically meaningful improvement, whereas a change of 0.08 points or greater was used to define clinically meaningful worsening. A change of 11 points or more from baseline was used to define clinically meaningful change in the EQ VAS.14 Specifically, each patient was categorized into 3 response levels at each post-baseline visit: improvement (i.e., change from baseline ≥ RD), no change (i.e., −RD < change from baseline < RD), and deterioration (i.e., change from baseline ≤ −RD).
Time to definitive deterioration (defined as having 2 consecutive clinically meaningful deteriorations) was assessed using the KM product-limited method to estimate survival distribution functions. For the median time to deterioration, a 2-sided 95% CI was estimated. Treatment groups were compared using a non-stratified log-rank test. The Cox proportional hazards regression analysis (adjusting for treatment group [oral azacitidine versus placebo], baseline score, and stratification factors) was used to calculate HRs, 95% CIs and P values. Patients who never experienced a clinically meaningful deterioration were censored at the time of the last assessment visit. Sensitivity analysis was performed with death as an event.14
Measures of Health Care Resource Utilization
Summaries of hospitalizations were specified a priori in the statistical analysis plan and included number of events, total number of days hospitalized, rate of events, days of hospitalized per person-year of exposure, and associated relative risk of hospitalization with the corresponding 95% CI.10 How missing data were handled was not reported.
Subgroup Analyses
For each subgroup, the HR and 95% CI were calculated from an unstratified Cox proportional hazards model.10 Subgroup analyses were planned a priori in the statistical analysis plan for the following groups of patients10:
age at induction therapy (< 65, ≥ 65, ≥ 75 years)
sex (male, female)
race (White, Asian, Black, or other)
CR or CRi status at randomization (CR, CRi)
CR or CRi status at first achieving response (CR, CRi)
CR or CRi status at randomization and use of consolidation (CR with consolidation, CR without consolidation, CRi with consolidation, and CRi without consolidation)
prior history of MDS or CMML (yes, no)
cytogenetic risk category at induction therapy (intermediate, poor)
MRD status at randomization (positive, negative)
CR or CRi status at randomization and MRD status at randomization (CR with MRD-positive, CR with MRD-negative, CRi with MRD-positive, and CRi with MRD-negative)
consolidation therapy following induction (yes, no)
consolidation therapy following induction (1 or 2 cycles, 3 or 4 cycles)
geographic region (North America, Europe, Asia, Australia)
ECOG PS (0 or 1, 2 or 3)
WHO AML classification (AML with myelodysplasia-related changes, AML with recurrent genetic abnormalities, AML not otherwise specified)
types of first-line subsequent therapy:
high-intensity and low-intensity chemotherapy
hypomethylating drug monotherapy and non-hypomethylating subsequent therapy
injectable azacitidine monotherapy and other subsequent therapy (excluding decitabine monotherapy).
In addition, OS and RFS were analyzed for the subset of patients whose doses were escalated to 300 mg × 21 days per 28-day cycle to explore the efficacy of oral azacitidine in this patient group. This subgroup analysis was also planned a priori in the statistical analysis plan.
The following subgroups, planned a priori in the statistical analyses plan, aligned with the subgroups prespecified in the protocol for this CADTH review: age, ECOG PS, prior history of MDS or CMML, cytogenetic risk, consolidation therapy following induction (yes, no), consolidation therapy following induction (1 or 2 cycles, 3 or 4 cycles), CR or CRi status at randomization (CR, CRi), MRD status at randomization. Only those subgroups identified in the CADTH review protocol are reported in the efficacy section below.
Multiplicity
Apart from the prespecified hierarchical testing of OS and RFS, no adjustments for multiplicity were performed to control the type I error rate.10
Dose Escalation
A post-relapse dose escalation (300 mg once daily from 14 days to 21 days of the 28-day cycle) was planned for all patients with disease relapse with blasts of between 5% and 15%, in either the peripheral blood or bone marrow.10 Blinding was upheld until study closure; therefore, all patients, irrespective of the study group they belonged to, could be selected for dose escalation. Because dose escalation was only implemented post-relapse (relapse was defined in the trial as the appearance of relapse was defined in the trial as the appearance of blast counts of ≥ 5% in the peripheral blood or bone marrow) the key secondary outcomes of RFS and the secondary outcome of time to relapse were not affected by dose escalation. However, the primary analysis of OS included all patients in the ITT population, whether they had received dose escalation or not. The OS outcome and the secondary outcome of time to discontinuation from treatment were not adjusted for the impact of patients who received a dose escalation. Subgroup analyses for OS and RFS were planned a priori according to the statistical analysis plan for the subset of patients who had experienced dose escalation in each treatment group. In addition, overall response and best response (using the IWG’s response criterion for AML) were planned a priori to be assessed in each treatment group in patients who experienced dose escalation.10 Overall response was defined as CR plus CRi plus partial response, and best response categories included CR, CRi, and partial response.9 Safety in the subset of patients who received a dose escalation was also assessed.9,10
Amendments
The study protocol of the QUAZAR AML-001 trial was amended twice (Amendment 1: December 29, 2015; Amendment 2 on November 8, 2018).10 Amendment 1 included changes to inclusion criteria and the schedule of examinations. Amendment 2 implemented an extension phase in which patients who were receiving oral azacitidine and demonstrated clinical benefit after the end of study were allowed to continue receiving oral azacitidine until they met the study discontinuation criteria or oral azacitidine became commercially available. As well, patients who were discontinued from study treatment (irrespective of randomization group) and continued in the follow-up phase were followed up for survival for at least another 12 months, until death, withdrawal of consent, or study closure, or they were lost to follow-up.9 Data from the extension phase are not included in this review.
Table 12: Statistical Analysis of Efficacy End Points
End point | Statistical model | Adjustment factors | Sensitivity analyses |
|---|---|---|---|
OS (primary outcome) Definition: the number of days from the date of randomization until the date of death from any cause and calculated as (date of death − date of randomization + 1) | KM survival method to estimate median, 25th and 75th percentiles, and 2-sided 95% CIs; numerical difference in the median, 25th and 75th percentiles, and 95% CIs of the difference between treatment groups was estimated; P values were calculated using a 2-sided stratified and unstratified log-rank test HRs and 95% CIs for the difference between treatment groups (null hypothesis: HR = 1) was derived using stratified Cox proportional hazard regression model. Survival and its 95% CI were assessed at pre-specified time intervals of 1 year and 2 years | Stratification factors:a
| Censoring:
Same statistical methods as those used for OS Additional analyses to assess confounding effects from subsequent treatments:
Proportional hazards assumption:
|
RFSb (key secondary outcome) Definition: time from the date of randomization to the date of documented relapsec or death from any cause, whichever occurred first | KM survival method to estimate median, 25th and 75th percentiles, and 2-sided 95% CIs; numerical difference in the median, 25th and 75th percentiles, and 95% CIs of the difference between treatment groups was estimated; P values were calculated using a 2-sided stratified and unstratified log-rank test HRs and 95% CIs for the difference between treatment groups (null hypothesis: HR = 1) is derived using stratified Cox proportional hazard regression model RFS and its 95% CI were assessed at pre-specified time intervals of 1 year and 2 years | Stratification factors:a
| Censoring based on EMA guidance RFS based on investigator-assessed response Primary analysis performed on mITT population |
Time to relapse (secondary outcome) Definition: interval from the date of randomization to the date of documented relapse after CR or CRi, defined according to IWG response criteria for AMLd | Competing risk analysis Cumulative incidence function (Kalbfleisch and Prentice [1980])39 Median time to relapse (KM analysis) | Not stratified | None |
Time to discontinuation from treatment (secondary outcome) Definition: interval from the date of randomization to the date of discontinuation from investigational product | Competing risk model Cumulative incidence functions; median time to discontinuation (unstratified KM analysis and difference, Kosorok method) Treatment discontinuation rates (KM methods and difference using a Greenwood variance estimate) | Not stratified | None |
EQ-5D-3L instrument (secondary outcome) | Statistical tests conducted at a 2-sided 0.05 level of significance with noninferential P values Analysis of observed changes from baseline (within-group changes from baseline: 1-sample t-test; between-group difference in changes from baseline: 2-sample t-tests) ANCOVA model to assess change from baseline (treatment difference: LS mean changes [95% CI and P value]) MMRM analysis to assess overall LS mean for change from baseline and difference in LS mean between treatment groups (95% CI and P value) Comparison of patients with clinically meaningful changes between treatment groups (CMH test) Time to definitive deterioration (KM product-limited method to estimate survival distribution functions) | ANCOVA model included covariates (treatment, baseline score, and trial randomization stratification factors) MMRM analysis controlled for the baseline HRQoL score and other pre-selected covariates of interest | For the time to definitive deterioration analysis a sensitivity analysis was performed with death as an event |
FACIT-F (secondary outcome) | Statistical tests were conducted at a 2-sided 0.05 level of significance with noninferential P values. Analysis of observed changes from baseline (within-group changes from baseline: 1-sample t-test; between-group difference in changes from baseline: 2-sample t-tests) ANCOVA model to assess change from baseline (treatment difference: LS mean changes [95% CI and P value]) MMRM analysis to assess overall LS mean for change from baseline and difference in LS mean between treatment groups (95% CI and P value) Comparison of patients with clinically meaningful changes between treatment groups (CMH test) Time to definitive deterioration (KM product-limited method to estimate survival distribution functions) | ANCOVA model included covariates (treatment, baseline score, and trial randomization stratification factors). The MMRM analysis controlled for the baseline HRQoL score and other pre-selected covariates of interest | For the time to definitive deterioration analysis a sensitivity analysis was performed with death as an event |
PINR (exploratory outcome) | Statistical tests were conducted at a 2-sided 0.05 level of significance with noninferential P values. Analysis of observed changes from baseline (within-group changes from baseline: 1-sample t-test; between-group difference in changes from baseline: 2-sample t-tests) ANCOVA model to assess change from baseline (treatment difference: LS mean changes [95% CI and P value]) MMRM analysis to assess overall LS mean for change from baseline and difference in LS mean between treatment groups (95% CI and P value) | ANCOVA model included covariates (treatment, baseline score, and trial randomization stratification factors) MMRM analysis controlled for the baseline HRQoL score and other pre-selected covariates of interest | None |
AML = acute myeloid leukemia; ANCOVA = analysis of covariance; CI = confidence interval; CMH = Cochran-Mantel-Haenszel; CR = complete remission; CRi = complete remission with incomplete blood count recovery; EMA = European Medicines Agency; EQ-5D-3L = EuroQol 5-Dimensions 3-Levels questionnaire; FACIT-F = Functional Assessment of Chronic Illness–Fatigue; HR = hazard ratio; HRQoL = health-related quality of life; IPCW = inverse probability of censoring weighted method; KM = Kaplan–Meier; mITT = modified intention-to-treat; MMRM = mixed-effect model for repeated measures; LS = least squares; OS = overall survival; PINR = Physical Impairment Numeric Rating; RFS = relapse-free survival.
aPrimary history of MDS was not included as a factor in the stratified analyses as the minimum number of patients in that stratum was lower than 16 (irrespective of treatment group).
bFormal statistical analyses for RFS will only be made if oral azacitidine is in favour of placebo for OS at the 2-sided 0.05 significance level. The P value from the stratified log-rank test is the confirmatory P value.
cDocumented relapse was defined as the earliest date of any of the following: 5% or greater bone marrow blasts from the central pathology report; the appearance of greater than 0% blasts in the peripheral blood with a later bone marrow confirmation (bone marrow blasts 5% or greater) within 100 days; or at least 2 peripheral blasts of at least 5% within 30 days.
dResponse according to International Working Group criteria for AML (Table 24, Appendix 3).
Analysis Populations
The primary and secondary efficacy end points were analyzed using the ITT population as defined in Table 13.10 The EQ-5D-3L instrument, the PINR scale, and the FACIT-F scale were analyzed using the HRQoL-evaluable population as defined in Table 13. In sensitivity analyses, OS and RFS were analyzed using the mITT population as defined in Table 13.14 Analyses of safety were performed using the safety population (Table 13).10
Table 13: Analysis Populations in QUAZAR AML-001
Analysis population | Description |
|---|---|
ITT | All patients who are randomized, regardless of whether they received study treatment or not |
mITT | Patients who have met the inclusion/exclusion criteria and experienced no protocol violations (as defined below) and have received a minimum of 1 cycle of treatment. Protocol violations:
|
HRQoL-evaluable population | All patients in the ITT population who had a valid (i.e., non-missing) assessment at baseline and at least 1 valid post-baseline assessment |
Safety population | All randomized patients who have received at least 1 dose of study drug |
CR = complete remission; CRi = complete remission with incomplete blood count recovery; HRQoL = health-related quality of life; ITT = intention-to-treat; mITT = modified intention-to-treat.
Results
Patient Disposition
Details of patient disposition in the QUAZAR AML-001 trial are summarized in Table 14. A total of 555 patients were screened and of those 85.0% (N = 472) were randomized to receive oral azacitidine and BSC (N = 238) or placebo and BSC (N = 234). All patients who were not randomized failed to meet the trial eligibility criteria. A total of 469 randomized patients (84.5%) were treated: 236 of 238 (99.2%) in the oral azacitidine group and 233 of 234 (99.6%) in the placebo group. In the oral azacitidine group, 193 patients (81.1%) had discontinued treatment as of the final analysis data cut-off date. In the placebo group, 208 patients (88.9%) had discontinued placebo as of the final analysis.9
As of the final analysis, 347 patients (74%) had terminated the study: 167 patients (70.2%) in the azacitidine group and 180 patients (76.9%) in the placebo group. The main reason for termination was death, and the percentages of patients who died were 66.0% in the oral azacitidine group and 73.1% in the placebo group. In the oral azacitidine group, 10 patients (4.2%) discontinued the study due to withdrawal of consent and no patient discontinued due to being lost to follow-up. In the placebo group, 8 patients (3.4%) discontinued due to withdrawal of consent and 1 (0.4%) was lost to follow-up. At the time of the final data cut-off date, 18.9% of patients in the oral azacitidine group and 11.1% of patients in the placebo group were ongoing on study treatment, respectively.9
Table 14: Patient Disposition — ITT Population (Data Cut-Off Date: July 15, 2019)
Disposition | QUAZAR AML-001 | |
|---|---|---|
Oral azacitidine | Placebo | |
Screened, n | 555 | |
Randomized, na | 238 | 234 |
Treateda (safety population), n | 236 | 233 |
With ongoing treatment, n (%) | 45 (18.9) | 26 (11.1) |
Discontinued from treatment phase, n (%) | 193 (81.1) | 208 (88.9) |
Reason for discontinuation from treatment phase, n (%) | ||
Disease relapse | 143 (60.1) | 180 (76.9) |
Adverse event | 29 (12.2) | 11 (4.7) |
Withdrew consent | 9 (3.8) | 13 (5.6) |
Otherb | |||||| | |||||| |
Physician decisionc | |||||| | |||||| |
Death | |||||| | |||||| |
Lost to follow-up | |||||| | |||||| |
Protocol violation | |||||| | |||||| |
Patients in follow-up after discontinuation from treatment phase | 26 (10.9) | 28 (12.0) |
Patients who discontinued from the study n (%) | 167 (70.2) | 180 (76.9) |
Reason for discontinuation from the study n (%) | ||
Death | 157 (66.0) | 171 (73.1) |
Withdrew consent | 10 (4.2) | 8 (3.4) |
Lost to follow-up | 0 | 1 (0.4) |
ITT population | 238 | 234 |
mITT populationd | 223 | 217 |
HRQoL-evaluable population | ||
FACIT-F | |||||| | |||||| |
EQ-5D-3L | |||||| | |||||| |
PINR | ||
Physical impairment | |||||| | |||||| |
Difficulty completing outdoor tasks | |||||| | |||||| |
Difficultly completing indoor tasks | |||||| | |||||| |
Safety, Ne | 236 | 233 |
EQ-5D-3L = EuroQol 5-Dimensions 3-Levels questionnaire; FACIT-F = Functional Assessment of Chronic Illness Therapy–Fatigue; HRQoL = health-related quality of life; ITT = intention-to-treat; mITT = modified intention-to-treat; PINR = Physical Impairment Numeric Rating.
aTwo eligibility criteria were not met by a least 2% of patients screened: inclusion criterion No. 4 (achieved first CR or CRi status within 4 months ± 7 days before randomization; 47 patients, 8.5%) and inclusion criterion No. 10 (can understand and voluntarily sign an informed consent document before any study-related procedures/assessments being conducted; 12 patients, 2.2%).
bOther reasons for discontinuation were physician decision other than eligible for transplantation, patient decision, psychological reasons, and offered another clinical trial.
cPatients become eligible for allogeneic bone marrow or stem cell transplantation during the treatment phase.
dIn the oral azacitidine group, 10 patients (4.2%) were excluded from the mITT population because they had not received at least 1 cycle of study treatment, 5 patients (2.1%) did not achieve CR or CRi at randomization, and 1 (0.4%) had at least 1 eligibility criteria violation. In the placebo group, 3 patients (1.3%) were excluded from the mITT population because they did not receive at last 1 cycle of study treatment, 13 patients (5.6%) did not achieve CR or CRi at randomization, and 1 (0.4%) had at least 1 eligibility criteria violation.
eTwo patients (0.8%) in the oral azacitidine group (disease relapse; withdrew consent) and 1 (0.4%) in the placebo group (withdrew consent) did not receive study treatment and were excluded from the safety population.
Protocol violations were reported for |||||||||| patients randomized to the oral azacitidine group and |||||||||| patients in the placebo group. Patients may have had more than 1 violation. The most commonly reported violations (oral azacitidine versus placebo) included |||||||||||||||||||||||||||||||||| |||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||| (|||| |||||| ||||), |||||||||||||||||||||||||||||||||||||||||||| | |||||||||||||||||||||| No violations that substantially affected the internal validity of the study were reported.
Exposure to Study Treatments
Exposure to oral azacitidine and placebo as of the final analysis is summarized in Table 15. The median duration of treatment with oral azacitidine of 11.6 months (range = 0.5 to 74.3) was close to twice that of the treatment duration with placebo of 5.7 months (range = 0.7 to 68.5). The median dose intensity and median relative dose intensity of treatments were similar between groups, indicating high treatment adherence in both study groups. The median dose intensity (Table 15) for azacitidine was |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||, compared to |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| with placebo. The median relative dose intensities (Table 15) for oral azacitidine and placebo were |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| respectively. The median overall adherence (Table 15) was |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||| in both study groups.9,10
Table 15: Exposure to Study Treatment — Safety Set (Data Cut-Off Date: July 15, 2019)
Treatment exposure | QUAZAR AML-001 | |
|---|---|---|
Oral azacitidine | Placebo | |
Treated, n | 236 | 233 |
Duration of treatment (months)a | ||
Mean (SD) | |||||| | |||||| |
Median (range) | 11.6 (0.5 to 74.3) | 5.7 (0.7 to 68.5) |
Dose intensity (mg/day)b | ||
Mean (SD) | |||||| | |||||| |
Median | |||||| | |||||| |
Range | |||||| | |||||| |
Relative dose intensity (%)c | ||
Mean (SD) | |||||| | |||||| |
Median | |||||| | |||||| |
Range | |||||| | |||||| |
≤ 75%, n (%) | |||||| | |||||| |
> 75% to ≤ 85%, n (%) | |||||| | |||||| |
> 85% to ≤ 100%, n (%) | |||||| | |||||| |
> 100%, n (%) | |||||| | |||||| |
Overall adherence (%)d | ||
Mean (SD) | |||||| | |||||| |
Median | |||||| | |||||| |
Range | |||||| | |||||| |
SD = standard deviation.
aTreatment duration in months is defined as (treatment end date − first dose date + 1)/30.4375. Treatment end date is the last dose date + 14 days (the prescribed rest period of each cycle), or the death date, whichever is earlier.
bDose intensity is the cumulative dose divided by the treatment duration, with treatment duration defined as the date of first dose through last dose date + 14 days, or date of death, whichever is earlier (i.e., the last cycle’s end date).
cRelative dose intensity is the ratio of dose intensity to the planned dose intensity (300 mg/day × 14 days/28 days = 150 mg/day for all patients).
dThe adherence rate (%) for each subject was computed as 100 times the cumulative dose (in milligrams) taken over the period divided by the intended cumulative dose (in milligrams) that should have been taken over the same period.
Source: Clinical Study Report.9
Dose Modifications (Interruption, Reduction, Escalation)
As of the final analysis (July 15, 2019, data cut-off date), 102 patients (43.2%) in the oral azacitidine group and 40 patients (17.2%) in the placebo group of the safety population required a study treatment interruption due to a TEAE. The most common TEAE leading to dose interruption was neutropenia.10 More patients in the oral azacitidine group compared with the placebo group required dose reductions. One or 2 dose reductions, respectively, were experienced by |||||||||||||||||| in the oral azacitidine group and |||||||||||||||| in the placebo group. A higher proportion of patients in the oral azacitidine group compared with the placebo group experienced a dose reduction due to a TEAE (n = 37 [15.7%] and n = 6 [2.6%] in the oral azacitidine and placebo groups, respectively). The most common TEAE leading to dose reduction was neutropenia.10 Information regarding dose delays was not reported and not available at this time as confirmed with the sponsor.12
More dose escalations were reported for the oral azacitidine group compared to the placebo group in the ITT population (N = 51 [21.4%] and N = 40 [17.1%] in the oral azacitidine and placebo groups, respectively). The most common reason for dose escalation was |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||9
Concomitant Medication
Concomitant medications, as of the July 15, 2021, data cut-off date, were generally similarly administered across treatment groups and reported for |||||||||||||||||||||||||||||||| in the ITT population. Medications for |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| were those most commonly used. The most frequently reported concomitant medications included |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||9
Subsequent Treatments
The proportion of patients receiving subsequent AML therapies was lower in the oral azacitidine group compared with the placebo group (Table 16). At least 1 subsequent therapy was received by 137 patients (57.6%) in the oral azacitidine group compared to 170 patients (72.6%) in the placebo group. The percentage of patients who received subsequent intensive chemotherapy was 29.0% in the oral azacitidine group compared to 37.6% in the placebo group. The most common subsequent systemic treatments (reported in > 10% in either study group; oral azacitidine versus placebo) received by patients included |||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||9
The proportion of patients receiving subsequent transplantation was 6.3% in the oral azacitidine group compared with 13.7% the placebo group (Table 16).9
Table 16: Subsequent AML Therapies — Intention-to-Treat Population
ATC dictionary-level preferred name,a n (%) | QUAZAR AML-001 | ||
|---|---|---|---|
Oral azacitidine (N = 238) | Placebo (N = 234) | Total (N = 472) | |
Patients with at least 1 subsequent AML therapy | 137 (57.6) | 170 (72.6) | 307 (65.0) |
Type of therapy | |||
Intensive chemotherapy | 69 (29.0) | 88 (37.6) | 157 (33.3) |
Low-intensity therapy | 94 (39.5) | 110 (47.0) | 204 (43.2) |
|||| | |||||| | |||||| | |||||| |
|||||| | |||||| | |||||| | |||||| |
Subsequent AML therapies reported for ≥ 10% of patients in either treatment group | |||
-|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | |||||| | |||||| | |||||| |
|||||||||| | |||||| | |||||| | |||||| |
|||||||||||| | |||||| | |||||| | |||||| |
|||||||||||| | |||||| | |||||| | |||||| |
|||||||||||||||||||| | |||||| | |||||| | |||||| |
|||||||||| | |||||| | |||||| | |||||| |
Subsequent transplantation | 15 (6.3) | 32 (13.7) | 47 (10) |
AML = acute myeloid leukemia; ATC = Anatomical Therapeutic Chemical.
Note: Subsequent AML therapy is defined as any therapy collected on the case report form for subsequent therapy for AML.
aCoded using WHODrug version March 2019. A subject with multiple occurrences of a drug class or drug is counted only once in the specific ATC classification or preferred name, respectively.
bInjectable azacitidine.27
Source: Clinical Study Report.9
Efficacy
Only those efficacy outcomes and analyses of subgroups identified in the review protocol are reported below. Refer to Appendix 3 for detailed efficacy data.
Overall Survival
The OS results of the QUAZAR AML-001 trial for the oral azacitidine and the placebo groups are summarized in Table 17.
As of the final analysis (July 15, 2019, data cut-off date), 329 death events occurred across both study groups. The median duration of follow-up for OS for all patients was 41.2 months, ||| months for patients in the oral azacitidine group, and ||| months for those in the placebo group. The median OS was 24.7 months (95% CI, 18.7 to 30.5) in the oral azacitidine group compared to 14.8 months (95% CI, 11.7 to 17.6) in the placebo group, with a stratified HR of 0.69 (95% CI, 0.55 to 0.86; P = 0.0009) in favour of the oral azacitidine group. The KM curves are depicted in Figure 3. The survival curves converged at the end of the follow-up period. The survival probabilities of patients surviving to 12 and 24 months were 0.73 (95% CI, 0.66 to 0.78) and 0.51 (95% CI, 0.44 to 0.57), respectively, in the oral azacitidine group, and 0.56 (95% CI, 0.49 to 0.62) and 0.37 (95% CI, 0.31 to 0.43), respectively, in the placebo group. The sensitivity analyses of OS using the mITT and ITT populations produced consistent results. The sensitivity analysis performed to assess the impact of censoring patients who withdrew consent from survival follow-up was also consistent with the OS results for the primary analysis.9
Graphical assessment and testing of treatment-by-time interactions in the overall Cox proportional hazards model suggested deviation from the proportional hazard assumption. The results of pre-specified sensitivity analyses for OS (using a piecewise Cox model, general Wilcoxon test, and restricted mean survival time) that were planned to be conducted if the proportional hazards assumption was violated suggested the robustness and consistency of the primary OS results (Table 25, Table 26, Table 27, and Table 28 in Appendix 3).
Table 17: Summary of Primary and Secondary End Points in the QUAZAR AML-001 Trial (Data Cut-Off Date: July 15, 2019)
Efficacy outcome in ITT population | Oral azacitidine (N = 238) | Placebo (N = 234) | Difference (95% CI) |
|---|---|---|---|
Primary outcome: OS | |||
Median follow-up time for OS,a months | |||||| | |||||| | |||||| |
Median OS, months (95% CI)b | 24.7 (18.7 to 30.5) | 14.8 (11.7 to 17.6) | 9.9 (4.6 to 15.3) |
Events (death), n (%) | 158 (66.4) | 171 (73.1) | NR |
Censored, n (%) | |||||| | |||||| | NR |
Hazard ratio (95% CI)c | 0.69 (0.55 to 0.86) | NA | |
P valued | 0.0009 | NR | |
Survival probabilitye | |||
12 months, (95% CI) | 0.73 (0.66 to 0.78) | 0.56 (0.49 to 0.62) | 0.17 (0.08 to 0.26) |
24 months, (95% CI) | 0.51 (0.44 to 0.57) | 0.37 (0.31 to 0.43) | 0.14 (0.05 to 0.23) |
Secondary outcome: RFS | |||
Median, months (95% CI)f | 10.2 (7.9 to 12.9) | 4.8 (4.6 to 6.4) | 5.3 (3.1 to 7.5) |
Events (relapse or death) | 164 (68.9) | 181 (77.4) | NR |
Relapse, n (%) | 154 (64.7) | 179 (76.5) | NR |
Death, n (%) | |||||| | |||||| | NR |
Censored, n (%) | 74 (31.1) | 53 (22.6) | NR |
Hazard ratio (95% CI)c | 0.65 (0.52 to 0.81) | NA | |
P valued | 0.0001 | NR | |
RFS probabilityg | |||
6 months (95% CI) | 0.67 (0.61 to 0.73) | 0.45 (0.39 to 0.52) | 0.22 (0.13 to 0.31) |
12 months (95% CI) | 0.45 (0.38 to 0.51) | 0.27 (0.22 to 0.34) | 0.18 (0.09 to 0.26) |
|||||||||||||||||||||| | |||||| | |||||| | |||||| |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | |||
|||||||||||||||||||||||||||||||| | |||||| | |||||| | |||||| |
|||||||||||||| | |||
|||||||||||||||||||||| | |||||| | |||||| | |||||| |
|||||||||||||||||||||| | |||||| | |||||| | |||||| |
|||||||||||||||||||||| | |||||| | |||||| | |||||| |
Secondary outcome: Time to discontinuation from treatment | |||
Median, months (95% CI)j | 11.4 (9.8 to 13.6) | 6.1 (5.1 to 7.4) | |||||| |
Treatment discontinuation ratek | |||
|||||||||||||||||||||| | |||||| | |||||| | |||||| |
|||||||||||||||||||||| | |||||||||||||||||| | |||||| | |||||| |
|||||||||||||||||||||| | |||||||||||||||||| | |||||||||||||| | |||||| |
CI = confidence interval; CR = complete remission; CRi = complete remission with incomplete blood count recovery; ITT = intention-to-treat; NA = not applicable; NR = not reported; OS = overall survival; RFS = relapse-free survival.
aMedian follow-up time for OS estimated by the reverse Kaplan-Meier method for all patients.
bMedian estimate of OS is from a Kaplan-Meier method. Difference was calculated as oral azacitidine minus placebo. The CI for the difference was derived using the Kosorok method.
cThe hazard ratio is from a Cox proportional hazards model stratified by age, cytogenetic risk category, and receipt of consolidation therapy or not.
dThe P value is 2-sided from a log-rank test stratified by age, cytogenetic risk category, and receipt of consolidation therapy or not.
eKaplan-Meier methods are used to estimate the 1-year and 2-year survival probabilities. The CIs for the difference in the 1-year and 2-year survival probabilities were derived using the Greenwood variance estimate.
fMedian estimate of RFS is from a Kaplan-Meier method. Difference was calculated as oral azacitidine minus placebo. The CI for the difference was derived using the Kosorok method.
gKaplan-Meier methods are used to estimate the 1-year and 2-year survival probabilities. The CIs for the difference in the 1-year and 2-year survival probabilities were derived using the Greenwood variance estimate.
hUnstratified Kaplan-Meier analysis.
iEstimates of relapse rate are based on the cumulative incidence function from a competing risk analysis with death as a competing risk for relapse from CR or CRi.
jMedian estimate of time to discontinuation is from an unstratified Kaplan-Meier analysis. Difference was calculated as oral azacitidine minus placebo. The CI for the difference was derived using the Kosorok method.
kKaplan-Meier methods were used to estimate treatment discontinuation rates. The CIs for the difference were derived using the Greenwood variance estimate.
Source: Clinical Study Report.9
Planned sensitivity analyses, which censored for the use of subsequent therapy or subsequent transplantation, were conducted to assess potential confounding effects on the survival analyses from subsequent treatments (Table 18). The results of these sensitivity analyses were consistent with the primary OS results. Given that censoring for patients in each treatment group was greater than 80% in the analyses censoring for subsequent therapy, the ability to interpret these sensitive analyses remains limited. Additional sensitivity analyses for OS (a Cox proportional hazard model with covariates adjustment, IPCW method, and regression-based imputation analysis) were performed to further assess potential biases from subsequent treatments. The results of these sensitivity analyses also suggested consistency with the OS results for the ITT population (Table 29, Table 30, Table 31, Table 32, and Table 33 in Appendix 3).
Table 18: Summary of Prespecified Sensitivity Analyses of OS for Subsequent Therapy
Sensitivity analyses | Hazard ratio (95% CI) (oral azacitidine vs. placebo) | P valuea |
|---|---|---|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||| | |||||| | |||||| |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | |||||| | |||||| |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | |||||| | |||||| |
CI = confidence interval; OS = overall survival; vs = versus.
aThe hazard ratio is from a Cox proportional hazards model stratified by age, cytogenetic risk category, and receipt of consolidation therapy or not. The nominal P value is 2-sided from a log-rank test, stratified as described for the hazard ratio.
bExcluded because hydroxycarbamide is unlikely to lead to a significant effect in prolonging survival.
Source: Clinical Study Report.9
The OS results by subgroups of interest, as specified a priori in the protocol for this CADTH review, are summarized in Figure 4.
The treatment effect on OS was consistent with the primary analysis across patient subgroups, except for the subgroup of 3 or 4 cycles of consolidation therapy. The sample size of this subgroup was small (6 patients in the oral azacitidine group and 13 patients in the placebo group). Several other subgroups (≥ 75, prior history of MDS or CMML [yes], poor cytogenetic risk status at induction, and CRi at randomization) had relatively small sample sizes (< 50 patients in either group).9 The wide CIs in subgroups reflected uncertainty in the effect estimates.
Figure 4: Forest Plot of OS by Prespecified Subgroups — ITT Population (July 15, 2019, Data Cut-Off Date)
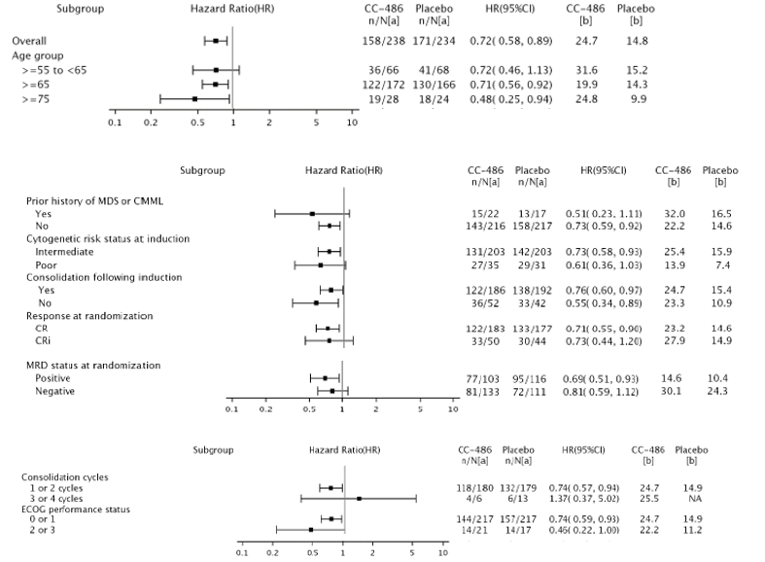
CC-486 = oral azacitidine; CI = confidence interval; CMML = chronic myelomonocytic leukemia; CR = complete remission; CRi = complete remission with incomplete blood count recovery; ECOG = Eastern Cooperative Oncology Group; HR = hazard ratio; ITT = intention-to-treat; MDS = myelodysplastic syndromes; MRD = minimal residual disease; OS = overall survival.
a Number of events per number of patients.
b Median OS in months.
Source: Clinical Study Report.9
Exploratory OS results in the subset of patients who had dose escalation are summarized in Table 35 in Appendix 3.
Relapse-Free Survival
The RFS results of the QUAZAR AML-001 trial for the oral azacitidine and the placebo groups are summarized in Table 17. Median RFS was longer in the oral azacitidine group compared with the placebo group. As of the final analysis (July 15, 2019, data cut-off date), median RFS was 10.2 (95% CI, 7.9 to 12.9) months in the oral azacitidine group and 4.8 (95% CI, 4.6 to 6.4) months in the placebo group, with a stratified HR of 0.65 (95% CI, 0.52 to 0.81; P = 0.0001) in favour of the oral azacitidine group. The KM curves are depicted in Figure 5. The survival curves converged at the end of the follow-up period. The probabilities of RFS at 12 and 24 months were 0.53 (95% CI, 0.46 to 0.59) and |||||||||||||||||||||||||||||||||| respectively, in the oral azacitidine group, and 0.72 (95% CI, 0.65 to 0.77) and 0.81 (95% CI, 0.76 to 0.86), respectively, in the placebo group. The results of the RFS sensitivity analysis using the mITT population were consistent with the RFS results for the ITT population.9

|||||||||||||||||||||||||||||||||||||||||||||||||| | 9
Note: This figure has been redacted at the sponsor’s request.
Planned sensitivity analyses, using censoring rules based on EMA guidance, which accounts for all events regardless of when they occurred, were consistent with the primary analysis of RFS. The median RFS values were 10.2 months for the oral azacitidine group and 4.8 months for the placebo group, with a stratified HR of 0.66 (95% CI, 0.54 to 0.81; P = 0.0001). For this analysis, |||| of patients in the oral azacitidine group and |||| of patients in the placebo group were censored.9
In addition, a sensitivity analysis was conducted using investigator-assessed response according to the IWG’s AML criteria. The results for this analysis were consistent with the primary analysis of RFS. Median RFS was |||||||||||||||||||||||||||||||| months in the oral azacitidine group and |||||||||||||||||||||||||||| months in the placebo group, with a stratified HR of |||||||||||||||||||||||||||||||||||||| |||||| in favour of the oral azacitidine group.9
Exploratory RFS results in the subset of patients who had dose escalation are summarized in Table 35 in Appendix 3.
Time to Relapse From CR or CRi
The results for time to relapse for oral azacitidine and the placebo groups are summarized in Table 17.
As of the final analysis (July 15, 2019, data cut-off date), the percentage of patients who had relapsed was |||| in the oral azacitidine group compared with |||| in the placebo group. Median time to relapse was |||||||||||||||||||||||||||||| months in the oral azacitidine group and |||||||||||||||||||||||||||| months in the placebo group. The 1-year and 2-year relapse rates were |||||||||||||| |||||||||||||| and |||||||||||||||||||||||||||||||||| respectively, in the oral azacitidine group and |||||||||||||||||||||||||||||||| and |||||||||| |||||||||||||||||| respectively, in the placebo group. These reported relapse rates were based on the cumulative incidence function from a competing risk analysis with death as a competing risk for relapse from CR or CRi. Figure 11 in Appendix 3 provides the cumulative incidence distribution of relapse for each treatment group.9
Time to Discontinuation From Treatment
The results for time to discontinuation from treatment for the oral azacitidine and the placebo groups are summarized in Table 17.
As of the final analysis (July 15, 2019, data cut-off date), most patients in both treatment groups had discontinued study treatment (81.1% in the oral azacitidine group and 88.9% in the placebo group). The median time to treatment discontinuation was 11.4 (95% CI, 9.8 to 13.6) months in the oral azacitidine group and 6.1 (95% CI, 5.1 to 7.4) months in the placebo group. The 1-year treatment discontinuation rate was |||||||||||||||||||||||||||||||| in the oral azacitidine group and |||||||||||||||||||||||||||||||| in the placebo group.9
Results for time to discontinuation from treatment were also reported using competing risks. As of the final analysis (July 15, 2019, data cut-off date) 143 patients (60.1%) in the oral azacitidine group and 180 patients (76.9%) in the placebo group had discontinued treatment due to disease relapse. Additional reasons for treatment discontinuation, representing the competing risks, are described in Table 34 in Appendix 3. The median time to treatment discontinuation due to disease relapse was 14.6 months (95% CI, 11.3 to 20.1) in the oral azacitidine group and 6.9 months (95% CI, 5.3 to 7.9) in the placebo group (Appendix 3).9
Health-Related Quality of Life
EQ-5D-3L
Adherence rates for the EQ-5D-3L were highest at baseline (> 95%) and similar between the oral azacitidine and the placebo groups over all post-baseline assessments (> 85%), except for the end-of-treatment visit (|||||||||||||||||||| in the oral azacitidine group and placebo groups, respectively). However, the completion rates declined over time in both study groups. After cycle 12 less than 50% of patients were still available for completion in the oral azacitidine group while the number of patients available for completion declined more rapidly in the placebo group, with less than 50% of patients available beyond cycle 6.14
The baseline characteristics based on the HRQoL-evaluable population were balanced between study groups (see Table 36 in Appendix 3 for baseline characteristics). Baseline scores were balanced between treatment groups (Table 19).9
The observed mean changes from baseline for the EQ-5D-3L health utility index and the EQ VAS are summarized in Figure 6 and Figure 7, respectively.
Analyses performed on the HRQoL outcomes were noninferential. Overall, no statistically significant or clinically meaningful differences were observed between the oral azacitidine group and the placebo groups based on the mean changes from baseline at any post-baseline assessment for the EQ-5D-3L health utility index and EQ VAS. The results generated by the ANCOVA models were consistent with the summary statistics of observed scores. The MMRM analyses showed results similar to those from the ANCOVA models, with no statistically significant or clinically meaningful differences observed in LS mean changes between treatment groups at any assessment time point for the EQ-5D-3L health utility index and EQ VAS.
The proportions of patients experiencing clinically meaningful deterioration over time by treatment group are summarized in Figure 9.
Overall, no statistically significant differences were observed in the proportion of patients with clinically meaningful deterioration between the oral azacitidine and placebo groups for the EQ-5D-3L health utility index and EQ VAS across all post-baseline visits, except for 1 visit (cycle 19, day 1) for which statistical significance was detected. However, the sponsor’s submission noted that, given the lack of adjusting for multiple testing, the statistical significance may be due to chance.14
Times to definitive deterioration for the EQ-5D-3L health utility index and EQ VAS are summarized in Figure 10. The time to definitive deterioration was similar between the oral azacitidine and the placebo groups. For the EQ-5D-3L health utility index and EQ VAS, respectively, the median times to definitive deterioration were |||| weeks in the oral azacitidine group versus |||| weeks for the placebo group (||||||||||||||||||||||||||||||||||||||||||||||||||||||) and “not yet reached” for the oral azacitidine group versus |||| weeks for the placebo group (||||||||||||||||||||||||||||||||||||||||||||||||||||). Sensitivity analyses in which deaths were censored showed similar results. Given that both treatment groups had greater than 65% and greater than 70% censored patients in the time to definitive deterioration analysis and its sensitivity analysis, respectively, the ability to interpret the time to definitive deterioration analyses remains limited.
PINR
Adherence and completion rates for the PINR scale were not reported.14 Overall, baseline characteristics based on the HRQoL-evaluable population were balanced between study groups (Table 36 in Appendix 3 provides baseline characteristics). Baseline scores were balanced between treatment groups (Table 19).
The summary statistics of observed mean changes from baseline for the FACIT-F scale are summarized in Figure 8. Overall, no statistically significant or clinically meaningful differences were observed between the oral azacitidine group and the placebo group based on the mean changes from baseline at any post-baseline assessment for the PINR scale (i.e., level of physical impairment, level of difficulty completing outdoor physical tasks, and level of difficulty completing indoor tasks). The results produced by the ANCOVA models were consistent with the summary statistics of observed scores. The MMRM analyses showed results similar to those of the ANCOVA models, with overall no statistically significant or clinically meaningful differences observed in LS mean changes between treatment groups at any assessment time point for the PINR scale.
The proportion of patients experiencing clinically meaningful deterioration and time to definitive deterioration were not reported for the PINR scale.14
Symptom Severity
FACIT-F Scale
Adherence rates for the FACIT-F scale were highest at baseline (> 95%) and similar between the oral azacitidine and placebo groups over all post-baseline assessments (> 85%), except for the end-of-treatment visit (69.6% and 63.3% in the oral azacitidine group and placebo groups, respectively). However, the completion rates declined over time in both study groups. After cycle 12 less than 50% of patients were still available for completion in the oral azacitidine group while the number of patients available for completion declined more rapidly in the placebo group, with less than 50% of patients available beyond cycle 6.
Overall, baseline characteristics based on the HRQoL-evaluable population were balanced between study groups (see Table 36 in Appendix 3 for baseline characteristics). Baseline scores were balanced between treatment groups (Table 19).
The summary statistics of observed mean changes from baseline for the FACIT-F scale are summarized in Figure 6. Analyses performed were noninferential. Overall, no statistically significant or clinically meaningful differences were observed between the oral azacitidine group and the placebo group based on the mean changes from baseline at any post-baseline assessment for the FACIT-F scale. The results using the ANCOVA models were consistent with the summary statistics of observed scores. The MMRM analyses showed results similar to those of the ANCOVA models; overall, no statistically significant or clinically meaningful differences were observed in LS mean changes between treatment groups at any assessment time point for the FACIT-F scale.
The proportions of patients experiencing clinically meaningful deterioration over time by treatment group are summarized in Figure 9. Overall, no statistically significant differences were observed in the proportion of patients with clinically meaningful deterioration between the oral azacitidine and placebo groups for the FACIT-F scale across all post-baseline visits, except for 1 visit (cycle 29, day 1) for which statistical significance was detected. However, the sponsor’s submission noted that, given the lack of adjusting for multiple testing, the statistical significance found may be due to chance.14
Time to definitive deterioration for the FACIT-F scale is summarized in Figure 10. The time to definitive deterioration was similar between the oral azacitidine and the placebo groups. The median time to definitive deterioration was ||| weeks for the oral azacitidine group and ||| weeks for the placebo group (||||||||||||||||||||||||||||||||||||||||||||||||||||).14 Sensitivity analyses in which deaths were censored showed similar results. Given that in both treatment groups there were more than 47% and more than 48% of censored patients in the time to definitive deterioration analysis and its sensitivity analysis, respectively, the ability to interpret the time to definitive deterioration analyses remains limited.
Table 19: Mean HRQoL Domain Scores at Baseline by Treatment Group for the HRQoL-Evaluable Population
HRQoL domain | Oral azacitidine (N = 225) | Placebo (N = 219) | Overall (N = 444) |
|---|---|---|---|
FACITa | |||
Scale Mean (SD) | |||||| | |||||||| | |||||||| |
EQ-5D-3La | |||
Health utility index, mean (SD) | |||||| | |||||||| | |||||||| |
VAS | |||||||||| | |||||||||| | |||||||||| |
PINRa | |||
Physical impairment | |||| | |||| | |||| |
Difficulty completing outdoor physical tasks | |||| | |||| | |||| |
Difficulty completing indoor task | |||| | |||| | |||| |
EQ-5D-3L = EuroQol 5-Dimensions 3-Levels questionnaire; FACIT-F = Functional Assessment of Chronic Illness Therapy–Fatigue; HRQoL = health-related quality of life; NA = not applicable; PINR = physical impairment numeric rating; SD = standard deviation; VAS = Visual Analogue Scale.
Note: HRQoL-evaluable population comprised patients with a non-missing assessment at baseline and at least 1 non-missing post-baseline assessment based on each specific HRQoL measure
aHigher scores indicate lower levels of fatigue for FACIT-F, better health for the EQ-5D-3L, and higher level of physical impairment for PINR items.
Source: Clinical Study Report.14
Figure 6: Results of HRQoL Analyses: Observed Mean Change From Baseline by Visit and Treatment Group — HRQoL-Evaluable Populationa
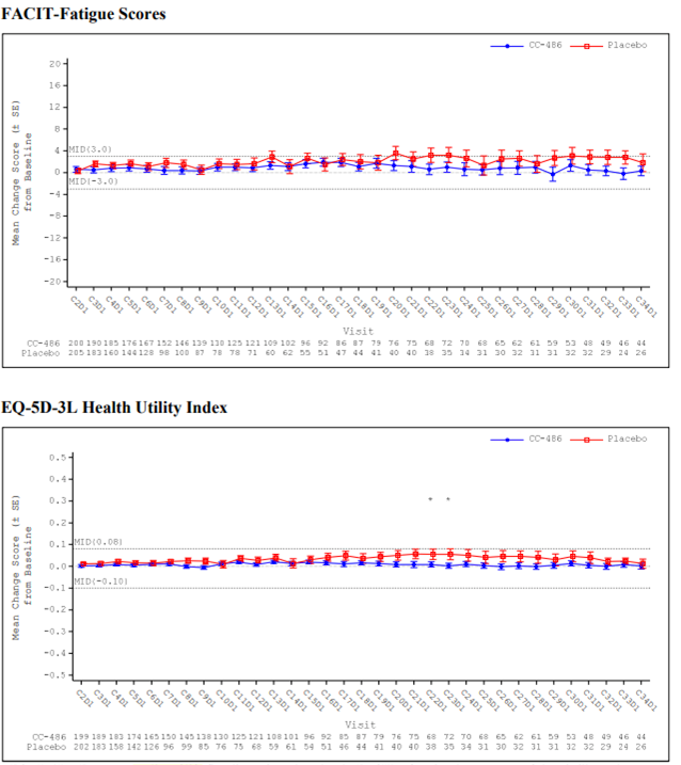
CC-486 = oral azacitidine; EQ-5D-3L = EuroQol 5-Dimensions 3-Levels questionnaire; FACIT = Functional Assessment of Chronic Illness Therapy; HRQoL = health-related quality of life; MID = minimally important difference; SE = standard error.
Note: Data are reported up to the cycle with an n of at least 25 in both treatment groups. A positive change indicates an improvement from baseline for FACIT-Fatigue and EQ-5D-3L.
a Intention-to-treat patients with a valid (i.e., non-missing) HRQoL assessment at baseline and at least 1 valid post-baseline assessment.
* Indicates a statistically significant difference at P < 0.05 in mean change from baseline between treatment groups at a specific post-baseline assessment visit based on a 2-sample, 2-sided t-test.
Source: Clinical Study Report.9
Figure 7: Observed Mean Change From Baseline in EQ VAS by Visit and Treatment Group — HRQoL-Evaluable Population (July 15, 2019, Data Cut-Off Date)
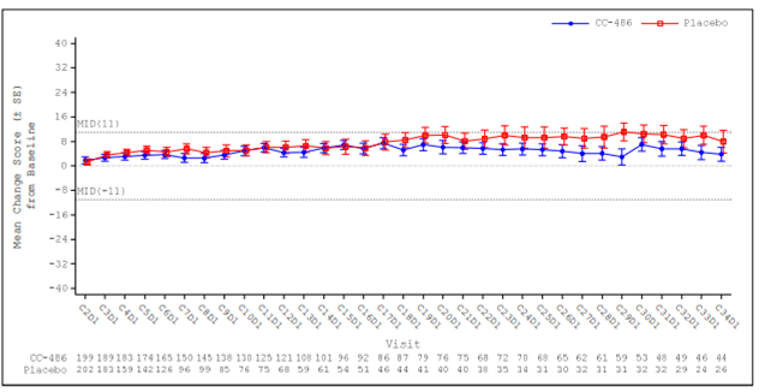
C = cycle; CC-486 = oral azacitidine; D = day; EQ VAS = EuroQol Visual Analogue Scale; HRQoL = health-related quality of life; SE = standard error.
Note: HRQoL-evaluable population were ITT patients with a non-missing assessment at baseline and at least 1 post-baseline visit based on the EQ VAS. Data are reported up to the cycle with an n or least 25 in both treatment groups. A positive change indicated an improvement from baseline. Dashed lines indicate clinically meaningful thresholds of improvement and worsening at the group level for the EQ VAS.
Source: Clinical Study Report.14
Figure 8: Observed Mean Change From Baseline in PINR Levels by Visit and Treatment Group — HRQoL-Evaluable Population (July 15, 2019, Data Cut-Off Date)
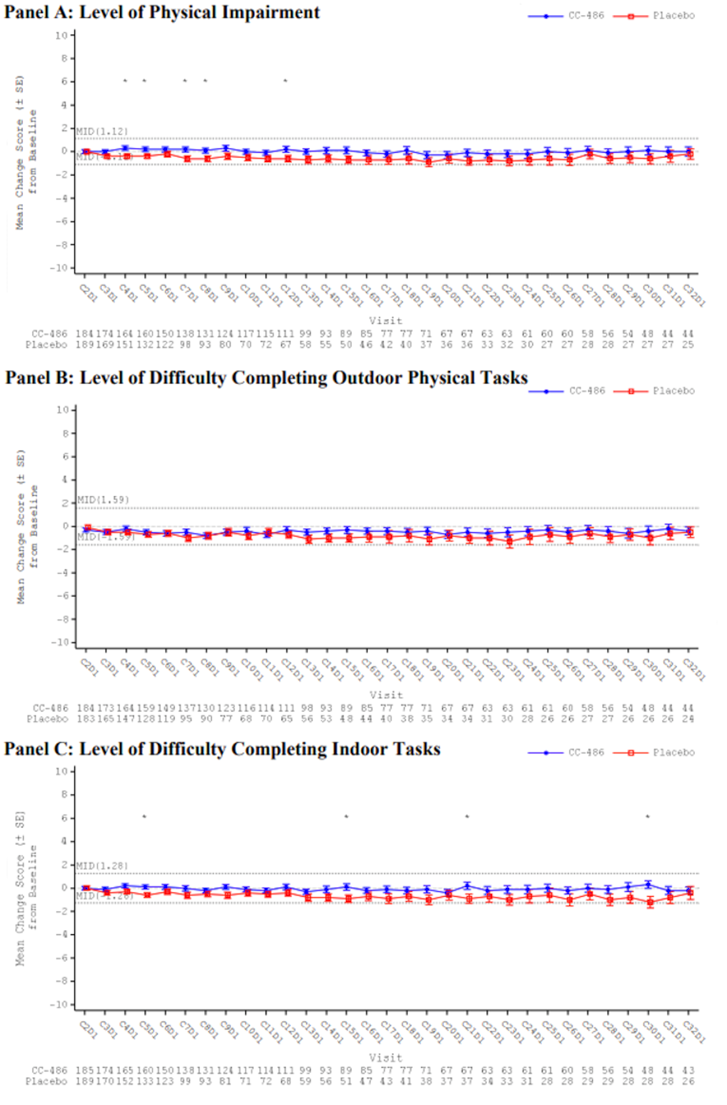
C = cycle; CC-486 = oral azacitidine; D = day; HRQoL = health-related quality of life; ITT = intention-to-treat; MID = minimally important difference; PINR = Physical Impairment Numeric Rating; SE = standard error.
Note: HRQoL-evaluable population were ITT patients with a non-missing assessment at baseline and at least 1 post-baseline visit based on the PINR item. Data were reported up to the cycle with an n of at least 25 in both treatment groups. A positive change indicated a deterioration from baseline. Dashed lines indicate clinically meaningful thresholds of improvement and worsening at the group level for the PINR item, defined as 0.5 × standard deviation.
* Indicates a statistically significant difference at P < 0.05 in mean change from baseline between treatment groups at a specific post-baseline assessment visit based on a 2-sample, 2-sided t-test.
Source: Clinical Study Report.14
Figure 9: Proportion of Patients Experiencing Clinically Meaningful Deterioration Over Time by Treatment Group — HRQoL-Evaluable Population (July 15, 2019, Data Cut-Off Date)
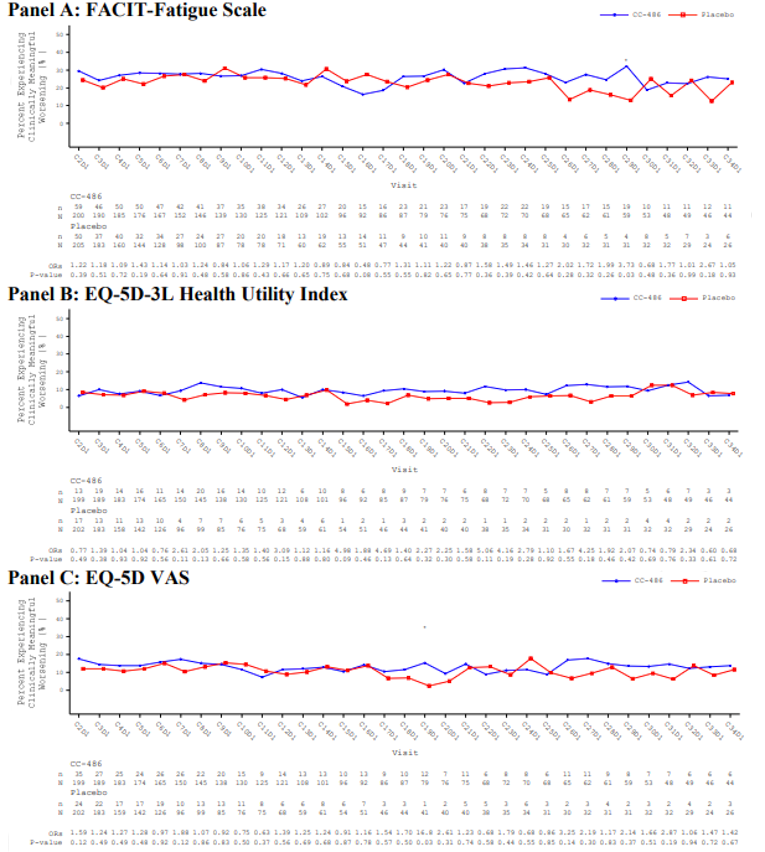
C = cycle; CC-486 = oral azacitidine; D = day; EQ-5D-3L = EuroQol 5-Dimensions 3-Levels; EQ-5D VAS = EuroQol 5-Dimensions Visual Analogue Scale. FACIT = Functional Assessment of Chronic Illness Therapy; HRQoL = health-related quality of life; OR = odds ratio.
Note: HRQoL-evaluable population comprised ITT patients with a non-missing assessment at baseline and at least 1 post-baseline visit based on the HRQoL measure. Data were reported up to the cycle with an n of at least 25 in both treatment groups. A clinically meaningful deterioration was defined as at least 3, 0.10, and 11 points worsening for the FACIT-Fatigue, EQ-5D-3L health utility index, and EQ-5D VAS, respectively. The denominator (N) at a given visit was based on the number of the HRQoL-evaluable patients who had valid non-missing scores at that visit. The ORs (and P value) were estimated using Cochran-Mantel-Haenszel tests, stratified by randomization stratification factors, to compare the odds of experiencing clinically meaningful deterioration between CC-486 versus placebo.
* Indicates a statistically significant difference at P < 0.05 in the proportion of patients with a deterioration between treatment groups (CC-486 versus placebo) at a specific post-baseline assessment visit based on a Cochran-Mantel-Haenszel test, stratified by randomization stratification factors
Source: Clinical Study Report.14
Figure 10: Kaplan-Meier Plot of Time to Definitive Deterioration in HRQoL Measures When Treating Death as an Event — HRQoL-Evaluable Population (Data Cut-Off Date: July 15, 2019)
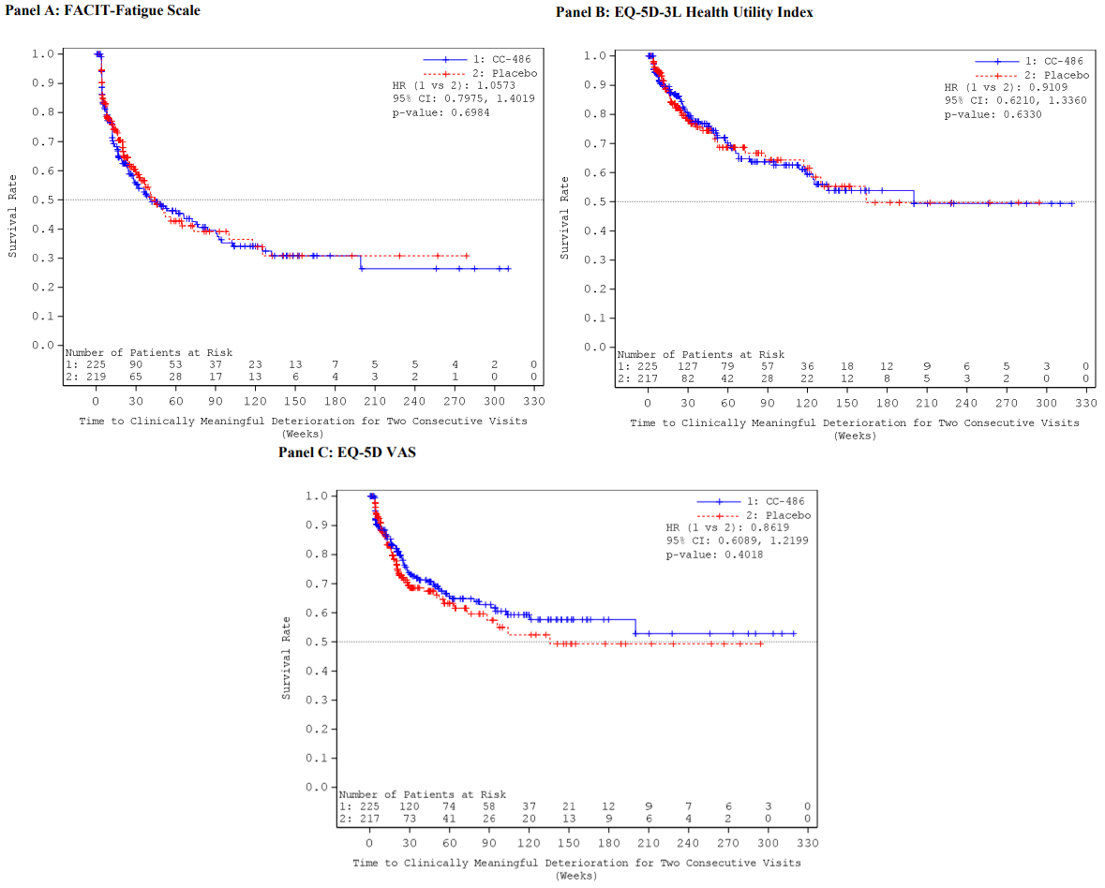
CI = confidence interval; CC-486 = oral azacitidine; EQ-5D-3L = EuroQol 5-Dimensions 3-Levels questionnaire; FACIT = Functional Assessment of Chronic Illness Therapy; HRQoL = health-related quality of life; HR = hazard ratio; EQ-5D VAS = EuroQol 5-Dimensions Visual Analogue Scale.
Note: HRQoL-evaluable population was intention-to-treat patients with a non-missing assessment at baseline and at least 1 post-baseline visit based on the HRQoL measure. Time to definitive HRQoL deterioration was defined as time to a clinically meaningful HRQoL deterioration sustained for at least 2 consecutive assessment visits. The HR was estimated from a Cox regression analysis with treatment group (CC-486 versus placebo), baseline domain score, and stratification factors (age group, prior history of myelodysplastic syndrome, cytogenetic risk category at time of induction therapy, and consolidation therapy following induction therapy) as covariates in the model.
Source: Clinical Study Report.14
Transfusion Independence
The outcome transfusion independence was specified in the CADTH review protocol but was not evaluated in the trial.
Health Care Resource Utilization
The total numbers of hospitalizations were 173 in the oral azacitidine group and 151 in the placebo group; hospitalization events after adjusting for duration of study drug exposure were 0.48 per person-year for the oral azacitidine group compared with 0.64 per person-year for the placebo group (relative risk = 0.740; 95% CI, 0.595 to 0.920; P = 0.0068) (Table 20).9 Analyses were noninferential.
A total of 2,872 hospitalization days were reported for the oral azacitidine group and 3,139 hospitalization days were reported for the placebo group. The number of days of hospitalization per person-year (exposure-adjusted rate) was lower in the oral azacitidine group compared with the placebo group: 7.89 days per person-year in the oral azacitidine group versus 13.36 days per person-year in the placebo group (relative risk = 0.591; 95% CI, 0.562 to 0.621; P < 0.0001) (Table 20).9 Analyses were noninferential.
Table 20: Hospitalizations — Safety Population (Data Cut-Off Date: July 15, 2019)
Hospitalizations | Oral azacitidine (N = 236) | Placebo (N = 233) |
|---|---|---|
Total person-years exposure (years) | 363.8 | 234.9 |
Number of patients hospitalized | 108 (45.8) | 118 (50.6) |
Number of hospital events | 173 | 151 |
Rate per person-year (2-sided 95% CI)a | 0.48 (0.41 to 0.55) | 0.64 (0.54 to 0.75) |
Relative risk (oral azacitidine vs. placebo) (2-sided 95% CI)b | 0.740 (0.595 to 0.920) | |
Two-sided P valueb | 0.0068 | |
Number of days hospitalized | 2,872 | 3,139 |
Rate per person-year (2-sided 95% CI)a | 7.89 (7.61 to 8.19) | 13.36 (12.90 to 13.84) |
Relative risk (oral azacitidine vs. placebo) (2-sided 95% CI)b | 0.591 (0.562 to 0.621) | |
Two-sided P valueb | < 0.0001 | |
CI = confidence interval.
aThe 95% CI for the per person-year rate of exposure is based on the exact method.
bThe 95% CI for the relative risk estimate and associated nominal P value testing that the relative risk is equal to 1 are based on asymptotic methods.
Source: Clinical Study Report.9
Harms
Only those harms identified in the CADTH review protocol are reported below. Detailed harms data are presented in Table 21. Safety in the subset of patients who had dose escalation was also assessed (Table 35 in Appendix 3).
Adverse Events
Nearly all patients in both study groups experienced at least 1 TEAE (97.9% of patients in the oral azacitidine group and 96.6% in the placebo group). The most commonly reported TEAEs in the oral azacitidine and the placebo groups, respectively, were nausea (64.8% and 23.6%), vomiting (59.7% and 9.9%), diarrhea (50.4% and 21.5%), neutropenia (44.5% and 26.2%), constipation (38.6% and 24.0%), thrombocytopenia (33.5% and 27.0%), and fatigue (29.7% and 19.3%).
Grade 3 or 4 TEAEs occurred in 71.6% of patients in the oral azacitidine group and 63.1% of patients in the placebo group (Table 21). The most commonly reported grade 3 or 4 TEAE in both groups was neutropenia. The percentage of patients experiencing neutropenia was 41.1% in the oral azacitidine group and 23.6% in the placebo group. Other grade 3 or 4 TEAEs occurring across both treatment groups included thrombocytopenia (22.5% and 21.5% in the oral azacitidine and placebo groups, respectively), anemia (14.0% and 12.9% in the oral azacitidine and placebo groups, respectively), febrile neutropenia (11.4% and 7.7% in the oral azacitidine and placebo groups, respectively), and leukopenia (7.6% and 6.0% in the oral azacitidine and placebo groups, respectively).9
Serious Adverse Events
The percentage of patients experiencing serious TEAEs was 33.5% in the oral azacitidine group compared to 25.3% in the placebo group (Table 21). The most common serious TEAEs were febrile neutropenia, which occurred in 6.8% of patients in the oral azacitidine group and 3.9% of patients in the placebo group; pneumonia, in 3.8% of patients in the oral azacitidine group and 3.0% of patients in the placebo group; and pyrexia, in 2.1% of patients in oral azacitidine group and 0.4% of patient in the placebo group.9
Withdrawals Due to Adverse Events
Withdrawals specifically due to AEs were not reported in the QUAZAR AML-001 trial. Adverse events led to discontinuation of study treatment in 13.1% patients in the oral azacitidine group and 4.3% patients in the placebo group (Table 21). The most commonly occurring TEAE leading to treatment discontinuation (reported in more than 1 patient in either group) included nausea (n = 5 [2.1%] and n = 0 [0%] in the oral azacitidine and placebo groups, respectively), diarrhea (n = 4 [1.7%] and n = 0 [0%] in the oral azacitidine and placebo groups, respectively), vomiting (n = 3 [1.3%] and n = 0 [0%] in the oral azacitidine and placebo groups, respectively), and thrombocytopenia (n = 1 [0.4%] and n = 2 [0.9%] in the oral azacitidine and placebo groups, respectively).9
Mortality
On-treatment deaths were reported for |||||||||| patients in the oral azacitidine group and |||||||||| patients in the placebo group. On-treatment death included those occurring on or after the first dose date and within 28 days after the date of last study treatment. Most on-treatment deaths were due to the |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| Post-treatment deaths were reported among |||||||||||| patients in the oral azacitidine group and |||||||||||| patients in the placebo group. Post-treatment deaths included those occurring beyond 28 days after the date of last study treatment. Most post-treatment deaths were due to the |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||).9
Overall, TEAEs leading to death occurred relatively rarely in both treatment groups (n = 9 [3.8%] and n = 4 [1.7%] in the oral azacitidine and placebo groups, respectively). The most commonly occurring TEAEs leading to death (reported in more than 1 patient in either group) included cerebral hemorrhage (n = 2 [0.8%] and n = [0.4%] in the oral azacitidine and placebo groups, respectively) and multiple organ dysfunction syndrome (n = 1 [0.4%] and n = 2 [0.9%] in the oral azacitidine and placebo groups, respectively). None of the TEAEs leading to death were considered treatment-related.9
Notable Harms
Notable harms specified in the protocol included gastrointestinal toxicities, hematologic toxicities, infections, fatigue, and bleeding.
Gastrointestinal Toxicities
The percentages of patients experiencing gastrointestinal TEAEs were 91.1% in the oral azacitidine group and 66.5% in the placebo group (Table 21). The most commonly reported gastrointestinal TEAEs in the oral azacitidine and placebo groups, respectively, included nausea (64.8% and 23.6%), vomiting (59.7% and 9.9), diarrhea (50.4% and 21.5%), and constipation (38.6% and 24.0). The percentage of patients experiencing gastrointestinal TEAEs of grade 3 or 4 was 16.9% in the oral azacitidine group compared with 6.4% in the placebo group. The most commonly reported grade 3 or 4 gastrointestinal TEAEs in the oral azacitidine group (versus placebo) were diarrhea (5.1% versus 1.3%), vomiting (3.0% versus 0.0%), nausea (2.5% versus 0.4), and constipation (1.3% versus 0%). Serious gastrointestinal TEAEs most commonly reported in the oral azacitidine group (versus placebo) included diarrhea (1.3% versus 0.0%), vomiting (0.8% versus 0.0%) gastroenteritis (0.8% versus 0.0%), and gastritis (0.8% versus 0.0%).9
Hematologic Toxicities
The percentage of patients experiencing myelosuppression TEAEs was |||| in the oral azacitidine group and |||| in the placebo group (Table 21). The most commonly reported myelosuppression event was neutropenia (44.5% and 26.2% in the oral azacitidine and placebo groups, respectively). Other myelosuppression TEAEs included thrombocytopenia (33.5% and 27.0% in the oral azacitidine and placebo groups, respectively), anemia (20.3 and 18.0% in the oral azacitidine and placebo groups, respectively), and febrile neutropenia (11.9% and 7.7% in the oral azacitidine and placebo groups, respectively). The percentage of patients experiencing grade 3 or 4 myelosuppression events was |||| in the oral azacitidine group and |||| in the placebo group; those most commonly reported (oral azacitidine versus placebo) included neutropenia (41.1% versus 23.6%), thrombocytopenia (22.5% versus 21.5%), and anemia (14.0% versus 12.9%). The percentage of patients experiencing serious myelosuppression events was |||| in the oral azacitidine group and |||| in the placebo group; those most commonly reported included |||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||9
Infections
The most commonly reported infections in the oral azacitidine group included upper respiratory tract infections (13.1% and 13.7% in the oral azacitidine and placebo groups, respectively), |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||). The percentage of patients experiencing grade 3 or 4 infections and serious infections was |||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. Grade 3 or 4 infections and serious infections most commonly reported in the oral azacitidine group (oral azacitidine versus placebo) included: |||||||||||| |||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||9
Fatigue
The percentage of patients experiencing fatigue TEAEs was 29.7% in the oral azacitidine group and 19.3% in the placebo group (Table 21). Fatigue of grade 3 or 4 or serious fatigue occurred rarely in patients in the trial. Fatigue of grade 3 or 4 was experienced by 3.0% and 0.9% of patients in the oral azacitidine group and placebo groups, respectively, and |||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||.9
Bleeding
The types and frequency of hemorrhagic events was similar across study groups (Table 21). The most commonly reported events in both groups included |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| Grade 3 or 4 hemorrhagic events and serious hemorrhagic events occurred rarely in both groups (|||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| ||||||||||||||||||||||||||||||||||||||||||||||||||9
Table 21: Summary of Harms — Safety Population (Data Cut-Off Date: July 15, 2019)
Harms | Oral azacitidine N = 236 | Placebo N = 233 |
|---|---|---|
Patients with at least 1 TEAE | ||
n (%) | 231 (97.9) | 225 (96.6) |
Most common events,a n (%) | ||
Nausea | 153 (64.8) | 55 (23.6) |
Vomiting | 141 (59.7) | 23 (9.9) |
Diarrhea | 119 (50.4) | 50 (21.5) |
Neutropenia | 105 (44.5) | 61 (26.2) |
Constipation | 91 (38.6) | 56 (24.0) |
Thrombocytopenia | 79 (33.5) | 63 (27.0) |
Fatigue | 70 (29.7) | 45 (19.3) |
Anemia | 48 (20.3) | 42 (18.0) |
Asthenia | 44 (18.6) | 13 (5.6) |
Pyrexia | 36 (15.3) | 44 (18.9) |
Arthralgia | 32 (13.6) | 24 (10.3) |
Abdominal pain | 31 (13.1) | 16 (6.9) |
Upper respiratory tract infection | 31 (13.1) | 32 (13.7) |
Decreased appetite | 30 (12.7) | 15 (6.4) |
Cough | 29 (12.3) | 39 (16.7) |
Febrile neutropenia | 28 (11.9) | 18 (7.7) |
Back pain | 28 (11.9) | 23 (9.9) |
Leukopenia | 25 (10.6) | 19 (8.2) |
Pain in extremity | 25 (10.6) | 12 (5.2) |
Dizziness | 25 (10.6) | 21 (9.0) |
Headache | 23 (9.7) | 26 (11.2) |
Peripheral edema | 21 (8.9) | 24 (10.3) |
Patients with at least 1 grade 3 or 4 TEAE | ||
n (%) | 169 (71.6) | 147 (63.1) |
Most common events,b n (%) | ||
Neutropenia | 97 (41.1) | 55 (23.6) |
Thrombocytopenia | 53 (22.5) | 50 (21.5) |
Anemia | 33 (14.0) | 30 (12.9) |
Febrile neutropenia | 27 (11.4) | 18 (7.7) |
Leukopenia | 18 (7.6) | 14 (6.0) |
Diarrhea | 12 (5.1) | 3 (1.3) |
|||||||||||||| | |||| | |||| |
|||||||||||| | |||| | |||| |
Fatigue | 7 (3.0) | 2 (0.9) |
Vomiting | 7 (3.0) | 0 |
Nausea | 6 (2.5) | 1 (0.4) |
|||||||||||||| | |||| | |||| |
|||||||||||||||||||||||||||||| | |||| | |||| |
|||||||| | |||| | |||| |
|||||||| | |||| | |||| |
Patients with at least 1 serious TEAE | ||
n (%) | 79 (33.5) | 59 (25.3) |
Most common events, n (%)c | ||
Febrile neutropenia | 16 (6.8) | 9 (3.9) |
Pneumonia | 9 (3.8) | 7 (3.0) |
Pyrexia | 5 (2.1) | 1 (0.4) |
Cellulitis | 4 (1.7) | 1 (0.4) |
Sepsis | 4 (1.7) | 5 (2.1) |
Influenza | 3 (1.3) | 0 |
Diarrhea | 3 (1.3) | 0 |
Back pain | 3 (1.3) | 0 |
Atrial fibrillation | 3 (1.3) | 0 |
Cholecystitis | 3 (1.3) | 2 (0.9) |
Anemia | 2 (0.8) | 3 (1.3) |
Thrombocytopenia | 2 (0.8) | 3 (1.3) |
Patients who stopped treatment due to TEAEs | ||
n (%) | 31 (13.1) | 10 (4.3) |
Most common events,d n (%) | ||
Nausea | 5 (2.1) | 0 (0) |
Diarrhea | 4 (1.7) | 0 (0) |
Vomiting | 3 (1.3) | 0 (0) |
Thrombocytopenia | 1 (0.4) | 2 (0.9) |
Abdominal pain upper | 2 (0.8) | 0 (0) |
Fatigue | 2 (0.8) | 0 (0) |
Deaths | ||
|||||||||||||| | |||| | |||| |
|||||||||||| | |||| | |||| |
Due to TEAEs,g n (%) | 9 (3.8) | 4 (1.7) |
Most common events,d n (%) | ||
Cerebral hemorrhage | 2 (0.8) | 1 (0.4) |
Multiple organ dysfunction syndrome | 1 (0.4) | 2 (0.9) |
Notable harms | ||
Gastrointestinal events | ||
Gastrointestinal events (any grade TEAEs), n (%)b | 215 (91.1) | 155 (66.5) |
Most common events, n (%)b | ||
Nausea | 153 (64.8) | 55 (23.6) |
Vomiting | 141 (59.7) | 23 (9.9) |
Diarrhea | 119 (50.4) | 50 (21.5) |
Constipation | 91 (38.6) | 56 (24.0) |
Abdominal pain | 31 (13.1) | 16 (6.9) |
Gastrointestinal events (grade 3 or 4 TEAEs), n (%) | 40 (16.9) | 15 (6.4) |
Gastrointestinal events (serious TEAEs), n (%) | 18 (7.6) | 8 (3.4) |
Myelosuppression events | ||
Myelosuppression event (any grade TEAEs), n (%) | |||| | |||| |
Most common events,a n (%) | |||| | |||| |
Neutropenia | 105 (44.5) | 61 (26.2) |
Thrombocytopenia | 79 (33.5) | 63 (27.0) |
Anemia | 48 (20.3) | 42 (18.0) |
Febrile neutropenia | 28 (11.9) | 18 (7.7) |
Leukopenia | 25 (10.6) | 19 (8.2) |
Myelosuppression event (grade 3 or 4 TEAEs), n (%) | |||| | |||| |
Myelosuppression event (serious TEAEs), n (%) | |||| | |||| |
Infections | ||
Infections (any grade TEAEs), n (%) | 147 (62.3) | 123 (52.8) |
Most common events,h n (%) | ||
Upper respiratory tract infection | 31 (13.1) | 32 (13.7) |
Influenza | 18 (7.6) | 7 (3.0) |
Nasopharyngitis | 17 (7.2) | 16 (6.9) |
Urinary tract infection | 17 (7.2) | 13 (5.6) |
Pneumonia | 14 (5.9) | 11 (4.7) |
Bronchitis | 13 (5.5) | 9 (3.9) |
Rhinitis | 12 (5.1) | 4 (1.7) |
Infections (any grade 3 or 4 TEAEs), n (%) | |||| | |||| |
Infections (serious TEAEs), n (%) | |||| | |||| |
Fatigue | ||
Fatigue (any grade TEAEs), n (%) | 70 (29.7) | 45 (19.3) |
Fatigue (grade 3 or 4 TEAEs), n (%) | 7 (3.0) | 2 (0.9) |
Fatigue (serious TEAEs), n (%) | |||| | |||| |
Hemorrhagic events | ||
Hemorrhagic events (any grade TEAEs), n (%) | |||| | |||| |
Most common events,i n (%) | ||
|||||||| | |||| | |||| |
|||||||||| | |||| | |||| |
|||||||||| | |||| | |||| |
|||||||||||| | |||| | |||| |
|||||||||||||||||| | |||| | |||| |
|||||||||||| | |||| | |||| |
|||||||||||||| | |||| | |||| |
Hemorrhagic events (any grade 3 or 4 TEAEs), n (%) | |||| | |||| |
Hemorrhagic events (serious TEAEs), n (%) | |||| | |||| |
TEAE = treatment-emergent adverse event.
Note: Coded using Medical Dictionary for Regulatory Activities version 22.0. A subject is counted only once for multiple events within preferred term/system organ class. TEAEs include adverse events that started between first dose date and the date 28 days after the last dose date of study treatment.
aFrequency of greater than 10% of patients in either treatment group.
bFrequency of at least 2% of patients in the oral azacitidine group.
cFrequency of at least 1% of patients in either study group.
dMore than 1 patient in either study group.
eDeaths occurring on or after the first dose date and within 28 days after the date of last study treatment.
fDeaths occurring beyond 28 days after the date of last study treatment.
gNone considered treatment-related.
hFrequency of greater than 5% of patients in either study group.
iFrequency of at least 1.5% of patients in either study group.
Source: Clinical Study Report.9
Critical Appraisal
Internal Validity
Baseline characteristics: A stratified randomization procedure was used based on known prognostic factors to minimize potential imbalances between the study groups that could lead to biased results. While imbalances were noted for a number of baseline characteristics, they were unlikely to influence clinical outcomes, according to the clinical experts consulted by CADTH.
Double-blind design: The QUAZAR AML-001 trial had a double-blind design. However, it is possible that some side effects of interest, such as gastrointestinal or myelosuppression events, allowed for the possible detection of the intervention being received for some participants. If trial investigators and/or patients were aware of the intervention assignment, this may have affected behaviour (such as initiation of subsequent treatment or adherence to treatment) or perceived HRQoL and symptom severity. The fact that a separate central review (blinded to patients’ treatment assignment) was used to confirm all clinical laboratory value assessments may have reduced the potential for bias arising from knowledge of treatments. Overall, the magnitude and direction of this bias remain unclear.
Statistical hierarchy and adjustment for multiplicity: Apart from the prespecified sequential testing of OS and RFS, no adjustments for multiplicity were performed to control the type I error rate; therefore, the analyses of the other secondary outcomes, such as HRQoL and symptom severity, were at risk of type I error.
Subgroup analysis: Methodological issues limited the ability to interpret the results from subgroup analyses. The subgroup analyses were noninferential, wide CIs reflected uncertainty in the effect estimates, and small sample sizes limited the generalizability to a broader population.
Proportional hazards: The analyses of OS and RFS were based on a stratified Cox proportional hazards model by which the HR between treatments is assumed to be unchanged over the duration of the comparison. The KM survival curves for OS initially diverged then converged at the end of the follow-up period, with a downward trend in survival. The clinical experts consulted by CADTH noted that the curves appeared to reflect their expectations of patients’ survival in clinical practice, i.e., patients with intermediate or poor cytogenic risk who are not eligible for transplantation (transplantation being the only potential cure in this setting) will relapse after some time. A pre-specified survival model that incorporated a treatment-by-time interaction suggested that the proportional hazards assumption did not hold. This affects the validity of the Cox regression models used to analyze OS in the trial. Prespecified sensitivity analyses were performed to adjust for nonproportional hazards such as restricted mean survival time, piecewise Cox regression, and generalized Wilcoxon tests. Results of these analyses were consistent with the results of the proportional hazards analysis, which supported the HRs obtained in the primary analysis demonstrating that oral azacitidine improved survival for patients relative to placebo. The graphical representation of the KM curves for RFS initially diverged and then converged, also suggesting a nonproportional hazard. However, no sensitivity analyses were pre-specified or conducted to evaluate the proportional hazards assumption.
Subsequent treatment: A higher proportion of patients in the placebo group compared with the oral azacitidine group received subsequent systemic therapy after discontinuing study treatment (57.6% in the oral azacitidine group versus 72.6% in the placebo group). Intensive chemotherapy and low-intensity therapy, respectively, were received by 29.0% and 39.5% of patients in the oral azacitidine group and by 37.6% and 47.0% of patients in the placebo group. Subsequent transplantation (implemented after discontinuation of study treatment)27 was received by a small number of patients, 15 (6.3%) patients in the oral azacitidine group and 32 (13.7%) patients in the placebo group. Subsequent treatments could confound the assessment of OS if patients experienced additional clinical benefit. Planned sensitivity analyses, which censored for the use of subsequent therapy or subsequent transplantation, were conducted to assess potential confounding effects on the survival analyses from subsequent treatments or transplantation. The results of these sensitivity analyses were consistent with the primary OS results. However, given that greater than 80% of patients were censored in each treatment group in the analyses censoring for subsequent therapy, the ability to interpret these sensitive analyses remains limited. Additional sensitivity analyses for OS (a Cox proportional hazards model with covariates adjustment, the IPCW method, and regression-based imputation analysis) were performed to further assess potential biases from subsequent therapies. The results of these sensitivity analyses also suggested consistency with the primary OS results. The clinical experts consulted by CADTH noted that post-relapse salvage therapies in this setting are unlikely to have a significant impact on survival outcomes. It was further noted that the number of patients who would receive subsequent transplantation in clinical practice would be small and that there was no clinical rationale as to why the proportion of patients who went on to receive subsequent transplantation. Given the consistent results obtained in the sensitivity analyses and the anticipated limited impact of post-relapse therapy on survival, the potential of confounding effects on survival outcomes from subsequent treatment is expected to be low.
Censoring of RFS: The study provided 2 definitions of RFS based on different censoring rules. In the analysis of RFS using the primary definition, patients who received subsequent anticancer therapy before the date of relapse were censored in the analysis (overall,|| patients in the trial were censored for having a relapse event after initiating follow-up therapy: | patients in the oral azacitidine group and | patients in the placebo group).9 This may lead to biased results through informative censoring. The study conducted a sensitivity analysis of RFS based on EMA guidance that did not consider the receipt of subsequent therapy. The RFS results based on the sensitivity analysis were similar and supported the results of the primary definition. Given the consistent results obtained in the analyses using EMA guidance and the relatively small proportion of patients censored for having a relapse after initiating follow-up therapy in either study group, the potential for informative censoring bias affecting RFS is low.
Exposure to study: The median duration of 11.6 months (range = 0.5 to 74.3) of treatment with oral azacitidine was approximately twice as long as the treatment duration of 5.7 months (range = 0.7 to 68.5) with placebo. Given that most patients in the placebo group discontinued treatment beyond cycle 5 or 6, a safety comparison between the study groups across all cycles may be biased against the oral azacitidine group. The oral azacitidine group had a higher proportion of patients remaining on study treatment as well as longer exposure to study treatment compared with the placebo group; therefore, patients in the oral azacitidine group were more likely to report AEs relative to the placebo group during the treatment phase.
HRQoL and symptom severity assessments: The interpretation of results for the EQ-5D-3L and FACIT-F scale (i.e., the ability to assess trends over time and to make comparisons across treatment groups) at later cycles is limited by the significant decline in patients available to provide assessment over time (less than 50% of patients were available in the placebo group beyond cycle 6 and less than 50% of patients were available in the oral azacitidine group beyond cycle 13). In addition, selection bias over time should be considered when interpreting results, as the long-term survivors tended to be the healthier patients. Given that the analyses of HRQoL and severity of symptom outcomes were based on the HRQoL-evaluable population and not on the ITT population, they do not capture the experience of all patients randomized in each treatment group. Furthermore, multiple statistical testing was conducted with no adjustments for multiplicity to control the type I error rate.
The EQ-5D-3L has been validated in patients with AML in China, achieving overall good validity and reliability (responsiveness was not assessed), whereas ranges for MIDs have been reported for cancer patients but not specially for patients with AML. The sponsor used a conservative approach to defining MIDs (i.e., chose relative high cut-off values from the MID ranges reported in the literature) for the EQ-5D-3L health utility index and EQ VAS scale (i.e., relatively high cut-off values compared with the MID ranges reported in the literature). The PINR scale has not been validated nor have MIDs been reported in the literature. The sponsor’s submission stated that half of the standard deviation at baseline from all ITT patients was used as an approximation for MID. However, a distribution-based approach to estimate the MID internally from the trial data is not an established method; rather a triangulation with anchor-based approach would be required. It is therefore unclear if the MID used in the trial is appropriate and reflective of a clinically meaningful change in outcomes in patients with AML. The FACIT-F scale has been validated in patients with CCL and showed overall good validity and reliability (responsiveness was not assessed), and MIDs have been reported in the literature for cancer patients but not specially for patients with AML. The sponsor defined the MID by choosing a cut-off value within the range of reported values in the literature. Overall, these methodological issues limit the ability to interpret the results from the EQ-5D-3L and the FACIT-F scale and render results from the PINR scale inconclusive.
External Validity
The QUAZAR AML-001 trial was an international, multicentre trial. Although the majority of patients in the trial were enrolled in trial sites in Europe, according to the clinical experts consulted by CADTH, the enrolled population was consistent with the population expected to be treated for AML in first remission in Canadian clinical practice. The clinical experts consulted by CADTH agreed that no different treatment effect would be expected based on different disease-management practices across countries. They noted that few patients in the trial had received prior types of therapy that are not standard of care in Canada. While a small number of patients in both study groups received consolidation therapies that are not offered in Canada, the clinical experts indicated that this is unlikely to have affected the study outcomes. The clinical experts felt that the QUAZAR AML-001 trial results would be generalizable to patients who have received gemtuzumab ozogamicin as induction therapy. Gemtuzumab ozogamicin was recently reviewed by CADTH and is recommended for reimbursement for adults with previously untreated, de novo CD33-positive AML, with the exception of patients with APL, who have good performance status and favourable, intermediate, or unknown cytogenetics (ELN 2017).19 The most common reason for transplantation ineligibility reported in the trial was age (for 64.8% of patients). The clinical experts pointed out that, since the inception of the QUAZAR AML-001 trial, practice has shifted away from using a certain age cut-off as a single criterion for transplantation ineligibility. However, given that older age, more comorbidities, and worse ECOG PS correlate, overall, it was felt that the patients selected for transplantation ineligibility were reflective of Canadian clinical practice. In the trial most patients received 1 cycle (44.9%) followed by 2 cycles (31.1%) of prior consolidation therapy. The experts noted that fewer patients in Canadian clinical practice would be expected to only receive 1 cycle and more patients would receive 2 cycles of prior consolidation therapy.
Relevance of trial efficacy outcomes: The primary outcome in the QUAZAR AML-001 trial was OS and the key secondary outcome was RFS. According to the clinical experts consulted by CADTH, both are clinically meaningful end points for patients with AML in their first remission and both are used to guide treatment selection in clinical practice. Upon AML relapse, treatment options are limited and administered primarily with palliative intent. Extending the period patients remain in remission is therefore important, as AML relapse is associated with a higher burden of symptoms, a decrease in quality of life, and shorter time to death. The primary and key secondary outcomes were supported by secondary outcomes that included time to relapse from CR or CRi and time to discontinuation from treatment.
Excluded patient subgroups: The trial excluded patients aged less than 55 and patients with less-than-adequate organ function. The clinical experts consulted by CADTH felt that it would be reasonable to generalize the QUAZAR AML-001 trial results to patients younger than 55 years, given the acceptable safety profile of oral azacitidine and the lack of a biological rationale to assume that outcomes of oral azacitidine would be different in younger patients with AML who otherwise meet the trial’s inclusion criteria. As well, given the acceptable safety profile of oral azacitidine the clinical experts agreed that it would be reasonable to leave it up to the discretion of the treating physician to apply some flexibility in the terms of using oral azacitidine in patients with slightly lower laboratory parameters than those outlined in the trial.
Dose escalation: One design feature of the QUAZAR AML-001 trial was to allow post-relapse dose escalation for patients who relapsed with 5% to 15% of blasts in the bone marrow. The dosing schedule was extended from 14 to 21 days out of a period of 28 days at the investigator’s discretion. The dose escalation allowed for an exploration of whether oral azacitidine could be used to reinitiate remission, which is not consistent with the Health Canada indication or the reimbursement request for azacitidine as maintenance therapy. The percentage of patients that had relapsed was |||| in the oral azacitidine group compared with |||| in the placebo group (with a relapse defined as a blast count ≥ 5% in the peripheral blood or bone marrow). Dose escalation was implemented in 51 patients in the oral azacitidine group and 40 patients in the placebo group. The Health Canada product monograph does not recommend dose escalation as Health Canada concluded that there is insufficient evidence at this time to guide a recommendation (i.e., the trial was not designed to assess efficacy in patients who received dose escalation).11 The clinical experts consulted by CADTH agreed that they would not attempt dose escalation in clinical practice as there is currently insufficient evidence to generalize the QUAZAR AML-001 trial results to oral azacitidine used to reinitiate remission. They further noted that it is unlikely that dose escalation would significantly influence OS outcomes observed in the trial due to the small numbers of patients who received dose escalation relative to the study sample size. Relapse-free survival was not affected by dose escalation as dose escalation was initiated after disease relapse, which was defined according to the IWG’s ALM response criteria: reappearance in the peripheral blood of 5% or greater blasts in the bone marrow.
Treatment discontinuation threshold: Treatment was continued until disease relapse (> 15% blasts in the bone marrow or peripheral blood), or unacceptable toxicity.10 The clinical experts consulted by CADTH noted that a 15% threshold is not a clinically established definition for relapse but appears to be an arbitrary choice in the trial to explore dose escalation in patients with bone marrow blast levels between 5% and 15%. The CADTH experts agreed that treatment with oral azacitidine would be discontinued in clinical practice at the time of relapse, as defined as reappearance of leukemic blasts in the peripheral blood or 5% or greater blasts in the bone marrow. An initially planned sensitivity analysis of RFS based on relapse using a 16% bone marrow blast threshold was not performed as it was determined that the interpretation of such analysis would be inconclusive given that not all patients with bone marrow blast levels between 5% and 15% received a dose escalation, and not all patients with dose escalation had marrow blast levels below 15%.9
Concomitant medications received by patients in the trial appeared reflective of the medications patients would receive in Canadian clinical practice, according to the clinical experts consulted by CADTH.
Placebo plus BSC is an appropriate comparator in Canadian clinical practice. The clinical experts by CADTH agreed that there is currently no active alternative treatment used in Canada in the target setting.
Indirect Evidence
No indirect treatment comparisons were included in the sponsor’s submission to CADTH or identified in the literature search.
Other Relevant Evidence
No long-term extension studies or other relevant studies were included in the sponsor’s submission to CADTH or identified from the literature.
Discussion
Summary of Available Evidence
The CADTH systematic review included 1 phase III RCT of oral azacitidine as maintenance therapy for the treatment of AML in adult patients who achieved CR or CRi following induction therapy with or without consolidation treatment and who are not eligible for HSCT. The QUAZAR AML-001 trial (N = 472) is an ongoing, international, multicentre, double-blind, placebo-controlled RCT that compares the efficacy and safety of maintenance therapy with oral azacitidine plus BSC with placebo plus BSC in patients with AML in first CR. Randomization was centrally performed in a 1:1 ratio and stratified by age at time of induction therapy, prior history of MDS or CMML, cytogenetic risk category at time of induction therapy, and receipt of consolidation therapy following induction. No crossover between the treatment groups was permitted. The primary outcome was OS, and the key secondary outcome was RFS. Other secondary end points included time to relapse, time to discontinuation from treatment, HRQoL, and symptoms.9
The QUAZAR AML-001 trial enrolled adults diagnosed with AML or AML secondary to prior MDS or CMML and intermediate- or poor-risk cytogenetics and who had achieved first remission (CR or CRi) following induction with or without consolidation chemotherapy. The majority of patients had an ECOG PS of 0 or 1, an intermediate cytogenetic risk, and had received 1 or 2 cycles of consolidation therapy. The most common reason for transplantation ineligibility was age, followed by comorbidities.9
No indirect treatment comparisons or other evidence were included in the sponsor’s submission to CADTH or identified in the literature search.
Interpretation of Results
Efficacy
The QUAZAR AML-001 trial met its primary end point, demonstrating statistically significant improvement in OS in favour of the oral azacitidine group compared with the placebo group. The results of prespecified sensitivity analyses for OS, which were conducted to adjust for nonproportional hazards and to assess potential biases from subsequent treatments, suggested the robustness and consistency of the primary OS results, indicating that oral azacitidine resulted in improved survival for patients. Results for subgroups of interest, as specified in the protocol for this CADTH systematic literature review, suggested that the OS benefit was consistently favourable across pre-specified subgroups of patients (except for the subcategory “3 or 4 cycles of prior consolidation therapy”). However, given that the trial was not designed to detect differences in treatment effects across subgroups, no conclusions can be drawn from the subgroup results. Results for the key secondary outcome, RFS, were supportive of the OS results and demonstrated a statistically significant improvement in favour of oral azacitidine. Other secondary efficacy outcomes, time to relapse from CR or CRi and time to discontinuation from treatment, were also supportive of the observed OS gain. According to the clinical experts CADTH consulted for this review, OS and RFS are both clinically meaningful end points for patients with AML in their first remission and are used to guide treatment selection in clinical practice. Upon AML relapse, treatment options are limited and administered primarily with palliative intent. Extending the period patients remain in remission is important as AML relapse is associated with a higher burden of symptoms, a decrease in quality of life, and shorter time to death. This view was echoed by the input provided by the patient advocacy group and the registered clinician input, both of which highlighted prolongation of life and maintenance of remission as important treatment goals for patients. The clinical experts further noted that the improvements in OS and RFS of the magnitude observed in the QUAZAR AML-001 trial (absolute differences for OS and RFS were approximately 10 months and 5 months, respectively) are of clinical importance in a patient population for which there is currently no standard maintenance treatment. Given that most patients in this setting relapse after a few months, the benefits observed with oral azacitidine over placebo were considered clinically meaningful.
Input received by the patient advocacy group, the registered clinicians, and the clinical experts consulted by CADTH highlighted HRQoL as an important outcome and treatment goal for patients. The level of fatigue and HRQoL over the assessment period was similar between the 2 study groups and oral azacitidine did not appear to significantly delay time to deterioration compared with placebo. The clinical experts consulted by CADTH considered this to be reasonable in this setting, where patients’ HRQoL is expected to be relatively high and stable. However, given several important limitations, including the noninferential analyses and the significant decline in patients available to provide assessment over time, the interpretation of results for the EQ-5D-3L and FACIT-F scale is limited.
Although patients in the QUAZAR AML-001 trial were considered representative of patients in Canadian clinical practice, the clinical experts consulted by CADTH noted that it would be reasonable to generalize the QUAZAR AML-001 trial results to patients younger than 55 years, who were excluded from the trial. The Health Canada indication does not have an age restriction. As well, given the acceptable safety profile of oral azacitidine, the clinical experts felt that it would be reasonable to leave it to the discretion of the treating physician to apply flexibility in the terms of using oral azacitidine in patients with slightly lower laboratory parameters than those outlined in the trial. Gemtuzumab ozogamicin was recently reviewed by CADTH and is recommended for reimbursement for adults with previously untreated, de novo CD33-positive AML (except APL) who have good performance status and favourable, intermediate, or unknown cytogenetics (ELN 2017).19 The clinical experts agreed that the trial results would be generalizable to patients who have received gemtuzumab ozogamicin as induction therapy.
The standard dose of oral azacitidine in the trial would be the dose used in Canadian clinical practice and is in line with the Health Canada–approved dosing. Dose escalation, which was attempted in the QUAZAR AML-001 trial to reinitiate remission, is not generalizable to clinical practice in Canada according to the clinical experts consulted by CADTH and has not been approved by Health Canada. The clinical experts further noted that it is unlikely that dose escalation would significantly influence the clinical outcomes observed in the trial due to the small numbers of patients who received dose escalation relative to the study sample size.
Harms
The median duration of treatment with oral azacitidine was about twice that of the treatment duration with placebo, which should be considered when reviewing the incidence of TEAEs. Almost all patients in both study groups experienced at least 1 TEAE. Differences in the proportion of patients experiencing grade 3 and 4 TEAEs and serious TEAEs were driven mainly by gastrointestinal toxicities and myelosuppression events. The most commonly reported TEAEs in the oral azacitidine and the placebo groups were nausea, vomiting, diarrhea, and neutropenia. The most commonly reported grade 3 or 4 and serious TEAEs, respectively, were neutropenia and febrile neutropenia. The clinical experts consulted by CADTH noted that most TEAEs associated with oral azacitidine could be managed with dose modifications and BSC, and, while treatment discontinuation due to TEAEs was relatively rare, it occurred more frequently in the oral azacitidine group. From the review of notable harms, it appeared that toxicities from oral azacitidine were seen mostly in the gastrointestinal system and as myelosuppression events. Few deaths were due to TEAEs overall, and no TEAE leading to death was considered treatment-related. The clinical experts consulted by CADTH agreed that the TEAEs observed with oral azacitidine were acceptable overall and could be adequately managed in clinical practice.
Conclusions
One phase III, double-blind, placebo-controlled RCT (QUAZAR AML-001) provided evidence regarding the efficacy and safety of maintenance therapy with oral azacitidine plus BSC compared with placebo plus BSC in patients with AML in first CR. Compared to placebo, patients treated with oral azacitidine showed statistically significant improvements in OS, the primary end point, and RFS, the key secondary outcome. The differences in median OS and RFS between treatment groups (approximately 10 months and 5 months, respectively) were considered clinically meaningful by the clinical experts consulted by CADTH. Other secondary efficacy outcomes, time to relapse from CR or CRi, and time to discontinuation from treatment were also supportive of the observed OS and RFS gains. The level of fatigue and HRQoL over the assessment period was similar between the 2 study groups, and oral azacitidine did not appear to significantly delay time to deterioration compared with placebo. However, these results remain uncertain due to a number of important limitations. The toxicity profile of oral azacitidine was considered manageable by the clinical experts consulted by CADTH.9
References
1.Drug Reimbursement Review sponsor submission: Onureg (azacitidine): 200 mg, 300 mg tablets [internal sponsor's package]. St. Laurent (QC): Celgene; 2021 Mar 1.
2.PrOnureg® (azacitidine): 200 mg, 300 mg tablets [product monograph]. St. Laurent (QC): Celgene; 2021 Jan 4.
3.Cogle CR, Scott BL, Garcia-Manero G. Oral azacitidine (Cc-486) for the treatment ofmyelodysplastic syndromes and acutemyeloid leukemia. Oncologist. 2015;20(12):1404-1412. PubMed
4.Acute myeloid leukemia (Version 3.2021). NCCN Clinical practice guidelines in oncology. Plymouth Meeting (PA): National Comprehensive Cancer Network; 2021.
5.Acute myelogenous leukemia statistics. Toronto (ON): Canadian Cancer Society; 2021: https://www.cancer.ca/en/cancer-information/cancer-type/leukemia-acute-myelogenous-aml/statistics/?region=on. Accessed 2021 May 12.
6.Döhner H, Estey E, Grimwade D, et al. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood. 2017;129(4):424-447. PubMed
7.Fey MF, Buske C. Acute myeloblastic leukaemias in adult patients: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2013;24(Suppl 6):vi138-143. PubMed
8.Koreth J, Schlenk R, Kopecky KJ, et al. Allogeneic stem cell transplantation for acute myeloid leukemia in first complete remission: systematic review and meta-analysis of prospective clinical trials. JAMA. 2009;301(22):2349-2361. PubMed
9.Clinical study report CC-486-AML-001. A phase 3, randomized, double-blind, placebo controlled study to compare efficiency and safety of oral azacitidine plus best supportive care versus best supportive care as maintenance therapy in subjects with acute myeloid leukemia in complete remission [internal sponsor's report]. St. Laurent (QC): Celgene; 2020 Jan 8.
10.Wei AH, Dohner H, Pocock C, et al. Oral azacitidine maintenance therapy for acute myeloid leukemia in first remission. N Engl J Med. 2020;383(26):2526-2537. PubMed
11.Health Canada reviewer's report: Onureg (azacitidine) [internal sponsor's report]. St. Laurent (QC): Celgene; 2020 Dec 31.
12.Celgene response to May 18, 2021 request for additional information regarding Onureg review [internal additional sponsor's information]. St. Laurent (QC): Celgene; 2021.
13.Celgene. NCT01757535: Efficacy of oral azacitidine plus best supportive care as maintenance therapy in subjects with acute myeloid leukemia in complete remission (QUAZAR AML-001). ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2012: https://clinicaltrials.gov/ct2/show/NCT01757535. Accessed 2021 Apr 22.
14.Study CC-486-AML-001. Patient-reported outcome/ health-realted quality of life report: oral azacitidine (CC-486) [internal sponsor's report]. St. Laurent (QC): Celgene; 2019 Dec 16.
15.Kolitz JE. Overview of acute myeloid leukemia in adults. In: Post TW, ed. UpToDate. Waltham (MA): UpToDate; 2021: https://www.uptodate.com. Accessed 2021 May 11.
16.Acute Myeloid Leukemia. Clinical practice guideline LYHE-006. Version 6. Edmonton (AB): Alberta Health Services; 2019: https://www.albertahealthservices.ca/assets/info/hp/cancer/if-hp-cancer-guide-lyhe006-aml.pdf Accessed 2021 May 12.
17.Canadian Cancer Statistics Advisory Committee. Canadian Cancer Statistics 2019. Toronto (ON): Canadian Cancer Society; 2019: https://cdn.cancer.ca/-/media/files/research/cancer-statistics/2020-statistics/canadian-cancer-statistics/res-cancerstatistics-canadiancancerstatistics-2019-en.pdf?rev=82dc3652fe3648988b9174ad4b397a24&hash=6D3186DF3AC76787C58EE95D1712033C. Accessed 2021 May 12.
18.Hills RK, Castaigne S, Appelbaum FR, et al. Addition of gemtuzumab ozogamicin to induction chemotherapy in adult patients with acute myeloid leukaemia: a meta-analysis of individual patient data from randomised controlled trials. Lancet Oncol. 2014;15(9):986-996. PubMed
19.Gemtuzumab ozogamicin (Mylotarg) for acute myeloid leukemia. Drug reimbursement review. Ottawa (ON): CADTH; 2020: https://cadth.ca/gemtuzumab-ozogamicin-mylotarg-acute-myeloid-leukemia-details. Accessed 2021 May 12.
20.Provincial funding summary: Rydapt (midostaurin) for acute myeloid leukemia (pCODR 10108). Ottawa (ON): CADTH; 2020: https://www.cadth.ca/sites/default/files/pcodr/pcodr_profund_midostaurin_rydapt_aml.pdf. Accessed 2021 May 7.
21.Döhner H, Estey E, Grimwade D, et al. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood. 2017;129(4):424-447. PubMed
22.Hansen DK, Kim J, Thompson Z, et al. ELN 2017 genetic risk stratification predicts survival of acute myeloid leukemia patients receiving allogeneic hematopoietic stem cell transplantation. Transplant Cell Ther. 2021;27(3):256.e1-256.e7. PubMed
23.McGowan J, Sampson M, Salzwedel DM, Cogo E, Foerster V, Lefebvre C. PRESS Peer Review of Electronic Search Strategies: 2015 guideline statement. J Clin Epidemiol. 2016;75:40-46. PubMed
24.Grey matters: a practical tool for searching health-related grey literature. Ottawa (ON): CADTH; 2019: https://www.cadth.ca/grey-matters. Accessed 2021 Mar 16.
25.Celgene response to April 7, 2021 request for additional information regarding Onureg review [internal additional sponsor's information]. St. Laurent (QC): Celgene; 2021.
26.Center for Drug Evaluation Research. Multidiscipline review. Onureg (azacitidine): 200 mg, 300 mg tablets. Company: Celgene. Application No.: NDA214120. Approval date: Sept 1, 2020. Silver Spring (MD): U.S. Food and Drug Administration (FDA); 2020 Sep 1: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2020/214120Orig1s000MultidisciplineR.pdf. Accessed 2021 Apr 28.
27.Celgene response to May 28, 2021 request for additional information regarding Onureg review [internal additional sponsor's information]. St. Laurent (QC): Celgene; 2021.
28.Yu H, Zeng X, Sui M, et al. A head-to-head comparison of measurement properties of the EQ-5D-3L and EQ-5D-5L in acute myeloid leukemia patients. Qual Life Res. 2021;30(3):855-866. PubMed
29.Pickard AS, Neary MP, Cella D. Estimation of minimally important differences in EQ-5D utility and VAS scores in cancer. Health Qual Life Outcomes. 2007;5:70-70. PubMed
30.Kvam AK, Fayers PM, Wisloff F. Responsiveness and minimal important score differences in quality-of-life questionnaires: a comparison of the EORTC QLQ-C30 cancer-specific questionnaire to the generic utility questionnaires EQ-5D and 15D in patients with multiple myeloma. Eur J Haematol. 2011;87(4):330-337. PubMed
31.Sinnott PL, Joyce VR, Barnett PG. Preference measurement in economic analysis. Guidebook. Menlo Park (CA): Health Economics Research Center; 2007: https://www.herc.research.va.gov/files/BOOK_419.pdf. Accessed 2021 Apr 30.
32.Protocol CC-486-AML-001. Patient-reported outcomes/quality of life: statistical analysis plan [internal sponsor's report]. St. Laurent (QC): Celgene; 2019 Jul 26.
33.Eek D, Ivanescu C, Corredoira L, Meyers O, Cella D. Content validity and psychometric evaluation of the Functional Assessment of Chronic Illness Therapy-Fatigue scale in patients with chronic lymphocytic leukemia. J Patient Rep Outcomes. 2021;5(1):27. PubMed
34.Ghia P, Pluta A, Wach M, et al. ASCEND: Phase III, randomized trial of acalabrutinib versus idelalisib plus rituximab or bendamustine plus rituximab in relapsed or refractory chronic lymphocytic leukemia. J Clin Oncol. 2020;38(25):2849-2861. PubMed
35.Cella D, Eton DT, Lai JS, Peterman AH, Merkel DE. Combining anchor and distribution-based methods to derive minimal clinically important differences on the Functional Assessment of Cancer Therapy (FACT) anemia and fatigue scales. J Pain Symptom Manage. 2002;24(6):547-561. PubMed
36.Patrick DL, Gagnon DD, Zagari MJ, Mathijs R, Sweetenham J. Assessing the clinical significance of health-related quality of life (HrQOL) improvements in anaemic cancer patients receiving epoetin alfa. Eur J Cancer. 2003;39(3):335-345. PubMed
37.Baer MR, George SL, Sanford BL, et al. Escalation of daunorubicin and addition of etoposide in the ADE regimen in acute myeloid leukemia patients aged 60 years and older: Cancer and Leukemia Group B Study 9720. Leukemia. 2011;25(5):800-807. PubMed
38.Baer MR, George SL, Sanford BL, et al. Treatment of older patients with de novo acute myeloid leukemia (AML) with one or more postremission chemotherapy courses: analysis of four CALGB studies. J Clin Oncol. 2010;28(15 Suppl):6531-6531.
39.Kalbfleisch JD, Prentice RL. The statistical analysis of failure time data. New York (NY): John Wiley & Sons; 1980.
40.Luo X, Li M, Wu C, et al. A proposed approach for analyzing post-study therapy effect in survival analysis. J Biopharm Stat. 2016;26(4):790-800. PubMed
41.Pappa V, Girkas K, Sambani C, et al. Azacitidine in a patient with newly diagnosed acute myeloid leukemia and poor risk cytogenetics: outcomes of prolonged therapy. Leuk Res. 2012;36(4):e78-80. PubMed
42.Wehmeyer J, Zaiss M, Losem C, et al. Impact of performance status and transfusion dependency on outcome of patients with myelodysplastic syndrome, acute myeloid leukemia and chronic myelomonocytic leukemia treated with azacitidine (PIAZA study). Eur J Haematol. 2018;101(6):766-773. PubMed
43.Huls G, Chitu DA, Havelange V, et al. Azacitidine maintenance after intensive chemotherapy improves DFS in older AML patients. Blood. 2019;133(13):1457-1464. PubMed
44.Jehn U, Knuppel W, Wilmanns W. Intensive maintenance treatment in acute myelogenous leukemia (AML): single institution experience of a multicenter randomized trial. Onkologie. 1988;11(1):13-17. PubMed
45.Jehn U, Zittoun R. AML-6 study of the value of cyclic alternating chemotherapy during remission in acute myelocytic leukemia. Onkologie. 1985;8(2):94-95. PubMed
46.Rees JK, Gray R, Hayhoe FG. The ninth British Medical Research Council trial for the treatment of acute myeloid leukaemia. Haematol Blood Transfus. 1987;30:35-37. PubMed
47.Rees JK, Gray RG. Remission induction and postremission therapy in acute myelogenous leukemia: British MRC Study. Haematol Blood Transfus. 1990;33:243-248. PubMed
48.Dumas PY, Bertoli S, Berard E, et al. Azacitidine or intensive chemotherapy for older patients with secondary or therapy-related acute myeloid leukemia. Oncotarget. 2017;8(45):79126-79136. PubMed
49.Seymour JF, Dohner H, Butrym A, et al. Azacitidine improves clinical outcomes in older patients with acute myeloid leukaemia with myelodysplasia-related changes compared with conventional care regimens. BMC Cancer. 2017;17(1):852. PubMed
50.Seymour JF, Dohner H, Minden MD, et al. Incidence rates of treatment-emergent adverse events and related hospitalization are reduced with azacitidine compared with conventional care regimens in older patients with acute myeloid leukemia. Leuk Lymphoma. 2017;58(6):1412-1423. PubMed
51.Roboz GJ, Montesinos P, Selleslag D, et al. Design of the randomized, phase III, QUAZAR AML maintenance trial of CC-486 (oral azacitidine) maintenance therapy in acute myeloid leukemia. Fut Oncol. 2016;12(3):293-302. PubMed
52.Jehn U, Zittoun R, Lowenberg B. Study design of the AML 6 and 7 trial for treatment of acute myelogenous leukemia: cyclic alternating chemotherapy during remission and remission duration and survival in elderly patients. [German]. Onkologie. 1985;8(3):160-164. PubMed
53.Benefits of AML maintenance therapy extend to quality of life and hospitalization. Oncologist. 2021;26(Suppl 1):S11-S12. PubMed
54.Derissen EJ, Beijnen JH, Schellens JH. Concise drug review: azacitidine and decitabine. Oncologist. 2013;18(5):619-624. PubMed
55.Pan J, Altman D, Wilde L. Measurable residual disease-guided treatment to prevent relapse in acute myeloid leukemia and myelodysplastic syndrome. Front Oncol. 2020;10(576924). PubMed
56.Lowder JN, Taverna P, Issa JP. Will next-generation agents deliver on the promise of epigenetic hypomethylation therapy? Epigenomics. 2015;7(7):1083-1088. PubMed
57.Wei AH. Maintenance therapy for AML: are we there yet? Blood. 2019;133(13):1390-1392. PubMed
58.Sekeres MA, Watts J, Radinoff A, et al. Randomized phase 2 trial of pevonedistat plus azacitidine versus azacitidine for higher-risk MDS/CMML or low-blast AML. Leukemia. 2021;35(7):2119-2124. PubMed
59.Dinardo CD. Hypomethylating agents and venetoclax in acute myeloid leukemia. Clin Adv Hematol Oncol. 2021;19(2):82-83. PubMed
60.Protocol CC-486-AML-001. A phase 3, randomized, double-blind, placebo-controlled study to compare efficacy and safety of oral azacitidine plus best supportive care versus best supportive care as maintenance therapy in subjects with acute myeloid leukemia in complete remission [internal sponsor's report]. St. Laurent (QC): Celgene; 2018 Nov 8.
61.Norman GR, Sloan JA, Wyrwich KW. Interpretation of changes in health-related quality of life: the remarkable universality of half a standard deviation. Med Care. 2003;41(5):582-592. PubMed
62.Norman GR, Sloan JA, Wyrwich KW. The truly remarkable universality of half a standard deviation: confirmation through another look. Expert Rev Pharmacoecon Outcomes Res. 2004;4(5):581-585. PubMed
63.van Reenen M, Oppe M. EQ-5D-3L user guide: basic information on how to use the EQ-5D-3L instrument. Rotterdam (NL): EuroQol Research Foundation; 2015 Apr. .
64.FACIT-F: Functional Assessment of Chronic Illness Therapy – Fatigue. Munich (DE): FACIT.org: https://www.facit.org/measures/FACIT-F. Accessed 2021 Apr 26.
Appendix 1: Literature Search Strategy
Note that this appendix has not been copy-edited.
Clinical Literature Search
Overview
Interface: Ovid
Databases:
MEDLINE All (1946 to present)
Embase (1974 to present)
Note: Subject headings and search fields have been customized for each database. Duplicates between databases were removed in Ovid.
Date of search: March 25, 2021
Alerts: Bi-weekly search updates until project completion
Search filters applied: randomized controlled trials; controlled clinical trials
Limits: Conference abstracts: excluded
Syntax | Description |
|---|---|
/ | At the end of a phrase, searches the phrase as a subject heading |
exp | Explode a subject heading |
* | Before a word, indicates that the marked subject heading is a primary topic; or, after a word, a truncation symbol (wildcard) to retrieve plurals or varying endings |
? | Truncation symbol for 1 or no characters only |
adj# | Requires terms to be adjacent to each other within # number of words (in any order) |
.ti | Title |
.ab | Abstract |
.ot | Original title |
.hw | Heading word; usually includes subject headings and controlled vocabulary |
.kf | Author keyword heading word (MEDLINE) |
.kw | Author keyword (Embase) |
.pt | Publication type |
.rn | Registry number |
.dq | Candidate term word (Embase) |
medall | Ovid database code: MEDLINE All, 1946 to present, updated daily |
oemezd | Ovid database code; Embase, 1974 to present, updated daily |
Multi-Database Strategy
Search Strategy
Azacitidine/
(Onureg* or azacitidine* or azacytidin* or Vidaza* or Ladakamycin* or Mylosar* or 5-AZCR or 5AZCR or “BRN 0620461” or BRN0620461 or CCRIS 60 or CCRIS60 or EINECS 206-280-2 or EINECS 2062802 or EINECS2062802 or HSDB 6879 or HSDB6879 or NCI C01569 or NCIC01569 or NSC 102816 or NSC102816 or CC-486 or CC486 or U 18496 or U18496 or WR 183027 or WR183027 or M801H13NRU).ti,ab,ot,kf,hw,nm,rn.
1 or 2
exp Leukemia, Myeloid, acute/
(acute adj3 (granulocytic or myeloblastic or myelocytic or myelogenous or myeloid or nonlymphoblastic or non-lymphoblastic or nonlymphocytic or non-lymphcytic or megakaryocytic or monocytic or myelomonocytic or basophilic or eosinophilic or erythroblastic or megakaryoblastic or promyelocytic) adj3 leuk?emia*).ti,ab,kf.
(erythroleuk?emia* or mast cell leuk?emia* or promyelocytic leuk?emia*).ti,ab,kf.
(AML or ANLL).ti,ab,kf.
or/4-7
3 and 8
9 use medall
*Azacitidine/
(Onureg* or azacitidin* or azacytidin* or Vidaza* or Ladakamycin* or Mylosar* or 5-AZCR or 5AZCR or “BRN 0620461” or BRN0620461 or CCRIS 60 or CCRIS60 or EINECS 206-280-2 or EINECS 2062802 or EINECS2062802 or HSDB 6879 or HSDB6879 or NCI C01569 or NCIC01569 or NSC 102816 or NSC102816 or CC-486 or CC486 or U 18496 or U18496 or WR 183027 or WR183027).ti,ab,kw,dq.
11 or 12
exp Acute myeloid leukemia/
(acute adj3 (granulocytic or myeloblastic or myelocytic or myelogenous or myeloid or nonlymphoblastic or non-lymphoblastic or nonlymphocytic or non-lymphcytic or megakaryocytic or monocytic or myelomonocytic or basophilic or eosinophilic or erythroblastic or megakaryoblastic or promyelocytic) adj3 leuk?emia*).ti,ab,kw,dq.
(erythroleuk?emia* or mast cell leuk?emia* or promyelocytic leuk?emia*).ti,ab,kw,dq.
(AML or ANLL).ti,ab,kw,dq.
or/14-17
13 and 18
19 use oemezd
20 not (conference abstract or conference review).pt.
10 or 21
(Randomized Controlled Trial or Controlled Clinical Trial or Pragmatic Clinical Trial or Equivalence Trial or Clinical Trial, phase III).pt.
Randomized Controlled Trial/
exp Randomized Controlled Trials as Topic/
“Randomized Controlled Trial (topic)”/
Controlled Clinical Trial/
exp Controlled Clinical Trials as Topic/
“Controlled Clinical Trial (topic)”/
Randomization/
Random Allocation/
Double-Blind Method/
Double Blind Procedure/
Double-Blind Studies/
Single-Blind Method/
Single Blind Procedure/
Single-Blind Studies/
Placebos/
Placebo/
Control Groups/
Control Group/
(random* or sham or placebo*).ti,ab,hw,kf,kw.
((singl* or doubl*) adj (blind* or dumm* or mask*)).ti,ab,hw,kf,kw.
((tripl* or trebl*) adj (blind* or dumm* or mask*)).ti,ab,hw,kf,kw.
(control* adj3 (study or studies or trial* or group*)).ti,ab,kf,kw.
(Nonrandom* or non random* or non-random* or quasi-random* or quasirandom*).ti,ab,hw,kf,kw.
allocated.ti,ab,hw.
((open label or open-label) adj5 (study or studies or trial*)).ti,ab,hw,kf,kw.
((equivalence or superiority or non-inferiority or noninferiority) adj3 (study or studies or trial*)).ti,ab,hw,kf,kw.
(pragmatic study or pragmatic studies).ti,ab,hw,kf,kw.
((pragmatic or practical) adj3 trial*).ti,ab,hw,kf,kw.
((quasiexperimental or quasi-experimental) adj3 (study or studies or trial*)).ti,ab,hw,kf,kw.
(phase adj3 (III or “3”) adj3 (study or studies or trial*)).ti,hw,kf,kw.
or/23-53
22 and 54
remove duplicates from 55
Clinical Trials Registries
ClinicalTrials.gov
Produced by the US National Library of Medicine. Targeted search used to capture registered clinical trials.
[Search – Onureg/oral azacitidine AND acute myeloid leukemia]
WHO ICTRP
International Clinical Trials Registry Platform, produced by the WHO. Targeted search used to capture registered clinical trials.
[Search terms – Onureg/oral azacitidine AND acute myeloid leukemia]
Health Canada’s Clinical Trials Database
Produced by Health Canada. Targeted search used to capture registered clinical trials.
[Search terms – Onureg/oral azacitidine AND acute myeloid leukemia]
EU Clinical Trials Register
European Union Clinical Trials Register, produced by the European Union. Targeted search used to capture registered clinical trials.
[Search terms – Onureg/oral azacitidine AND acute myeloid leukemia]
Grey Literature
Search dates: March 15 to 17, 2021
Keywords: Onureg/oral azacitidine AND acute myeloid leukemia
Limits: None
Updated: Search updated before the completion of stakeholder feedback period
Relevant websites from the following sections of the CADTH grey literature checklist Grey Matters: A Practical Tool For Searching Health-Related Grey Literature (https://www.cadth.ca/grey-matters) were searched:
Health technology assessment agencies
Health economics
Clinical practice guidelines
Drug and device regulatory approvals
Advisories and warnings
Drug class reviews
Clinical trials registries
Databases (free)
Appendix 2: Excluded Studies
Note that this appendix has not been copy-edited.
Reference | Reason for exclusion |
|---|---|
Pappa, V., et al., Azacitidine in a patient with newly diagnosed acute myeloid leukemia and poor risk cytogenetics: outcomes of prolonged therapy. Leukemia Research, 2012. 36(4): p. e78-80.41 | Study design |
Wehmeyer, J., et al., Impact of performance status and transfusion dependency on outcome of patients with myelodysplastic syndrome, acute myeloid leukemia and chronic myelomonocytic leukemia treated with azacitidine (PIAZA study). European Journal of Haematology, 2018. 101(6): p. 766-773.42 | Study design |
Huls, G., et al., Azacitidine maintenance after intensive chemotherapy improves DFS in older AML patients. Blood, 2019. 133(13): p. 1457-1464.43 | Intervention |
Jehn, U., W. Knuppel, and W. Wilmanns, Intensive maintenance treatment in acute myelogenous leukemia (AML): Single institution experience of a multicenter randomized trial. Onkologie, 1988. 11(1): p. 13-17.44 | Intervention |
Jehn, U. and R. Zittoun, [AML-6 study of the value of cyclic alternating chemotherapy during remission in acute myelocytic leukemia]. Onkologie, 1985. 8(2): p. 94-5.45 | Intervention |
Rees, J.K., R. Gray, and F.G. Hayhoe, The ninth British Medical Research Council trial for the treatment of acute myeloid leukaemia. Haematology & Blood Transfusion, 1987. 30: p. 35-7.46 | Intervention |
Rees, J.K. and R.G. Gray, Remission induction and postremission therapy in acute myelogenous leukemia: British MRC Study. Haematology & Blood Transfusion, 1990. 33: p. 243-8.47 | Intervention |
Dumas, P.Y., et al., Azacitidine or intensive chemotherapy for older patients with secondary or therapy-related acute myeloid leukemia. Oncotarget, 2017. 8(45): p. 79126-79136.48 | Study population |
Seymour, J.F., et al., Azacitidine improves clinical outcomes in older patients with acute myeloid leukaemia with myelodysplasia-related changes compared with conventional care regimens. BMC Cancer, 2017. 17(1): p. 852.49 | Study population |
Seymour, J.F., et al., Incidence rates of treatment-emergent adverse events and related hospitalization are reduced with azacitidine compared with conventional care regimens in older patients with acute myeloid leukemia. Leukemia & Lymphoma, 2017. 58(6): p. 1412-1423.50 | Study population |
Roboz, G.J., et al., Design of the randomized, phase III, QUAZAR AML maintenance trial of CC-486 (oral azacitidine) maintenance therapy in acute myeloid leukemia. Future Oncology, 2016. 12(3): p. 293-302.51 | No report outcomes reported |
Jehn, U., R. Zittoun, and B. Lowenberg, Study design of the AML 6 and 7 trial for treatment of acute myelogenous leukemia: Cyclic alternating chemotherapy during remission and remission duration and survival in elderly patients. [German]. Onkologie, 1985. 8(3): p. 160-164.52 | Not English |
Anonymous, Benefits of AML Maintenance Therapy Extend to Quality of Life and Hospitalization. Oncologist, 2021. 26(S1): p. S11-S12.53 | Review article |
Derissen, E.J., J.H. Beijnen, and J.H. Schellens, Concise drug review: azacitidine and decitabine. Oncologist, 2013. 18(5): p. 619-2454 | Review article |
Pan, J., D. Altman, and L. Wilde, Measurable Residual Disease-Guided Treatment to Prevent Relapse in Acute Myeloid Leukemia and Myelodysplastic Syndrome. Frontiers in Oncology, 2020. 10 (no pagination)(576924).55 | Review article |
Lowder, J.N., P. Taverna, and J.P. Issa, Will next-generation agents deliver on the promise of epigenetic hypomethylation therapy? Epigenomics, 2015. 7(7): p. 1083-8.56 | Editorial |
Wei, A.H., Maintenance therapy for AML: Are we there yet? Blood, 2019. 133(13): p. 1390-1392.57 | Editorial |
Sekeres, M.A., et al., Randomized phase 2 trial of pevonedistat plus azacitidine versus azacitidine for higher-risk MDS/CMML or low-blast AML. Leukemia, 2021. 22: p. 22.58 | Editorial |
Dinardo, C.D., Hypomethylating agents and venetoclax in acute myeloid leukemia. Clinical Advances in Hematology and Oncology, 2021. 19(2): p. 82-83.59 | Interview/Q&A |
Appendix 3: Detailed Outcome Data
Note that this appendix has not been copy-edited.
Table 24: Hematologic Response According to International Working Group AML Response Criteria
Category | Definition |
|---|---|
Morphologic CR | The following conditions should be met:
|
Morphologic complete remission with CRi | Defined as a morphologic complete remission but the ANC count may be < 1,000/μL or the platelet count may be < 100,000/μL |
CRc | Defined as morphologic complete remission with a reversion to a normal karyotype |
Relapse-free survival | Defined for patients who achieve CR or CRi and is measured from the date of attaining leukemia free state until the date of AML relapse or death from any cause, whichever occurs first |
Disease relapse | Relapse after CR or CRi is defined as reappearance of leukemic blasts in the peripheral blood or ≥ 5% blasts in the bone marrow not attributable to any other cause (e.g., bone marrow regeneration after consolidation therapy). In the setting of recent treatment, if there are no circulating blasts and the bone marrow contains 5% to 20% blasts, a repeat bone marrow performed at least a week later is necessary to distinguish relapse from regeneration |
Source: Sponsor’s submission.60
Prespecified Sensitivity Analyses of OS to Evaluate Proportional Hazard Assumption:
Table 25: Proportional Hazard Test of Cox Model With the Interaction Between Treatment and Time-Dependent Variable for Overall Survival — ITT Population (Data Cut-Off Date: July 15, 2019)
Variables | Statistics | Oral azacitidine (N = 238) | Placebo (N = 234) |
|---|---|---|---|
Stratified modela Interaction between Treatment and Time | P value | |||| | |
Covariates in the model include treatment and Interaction between treatment and time-dependent variable.
aThe Cox regression model is stratified by age, cytogenetic risk category, and received consolidation therapy or not.
Source: Clinical Study Report.9
Prespecified SAs to Be Performed if Proportional Hazards Assumption Does Not Hold:
Table 26: Summary of Piecewise Cox Model for Overall Survival — ITT Population (Data Cut-Off Date: July 15, 2019)
Model periods | Oral azacitidine (N = 238) | Placebo (N = 234) |
|---|---|---|
Piecewise Cox modela | ||
Oral azacitidine vs. placebo in interval ≤ 3 months | ||
P value | |||| | |
HR (95% CI) | |||| | |
Oral azacitidine vs. placebo in interval 3 to ≤ 6 months | ||
P value | |||| | |
HR (95% CI) | |||| | |
Oral azacitidine vs. placebo in interval 6 to ≤ 12 months | ||
P value | |||| | |
HR (95% CI) | |||| | |
Oral azacitidine vs. placebo in interval > 12 months | ||
P value | |||| | |
HR (95% CI) | |||| | |
CI = confidence interval; HR = hazard ratio.
aBased on stratified piecewise Cox regression model. Stratification factors include age, cytogenetic risk category, and received consolidation therapy or not.
Source: Clinical Study Report.9
Table 27: Summary of General Wilcoxon Test for Overall Survival — ITT Population (Data Cut-Off Date: July 15, 2019)
Log-rank tests: Oral azacitidine vs. placebo | Statistics | Oral azacitidine (N = 238) | Placebo (N = 234) |
|---|---|---|---|
General Wilcoxon Testa | P value | |||| | |
aThe P value was estimated by a general Wilcoxon test comparing the survival curves between the groups of oral azacitidine and placebo. No covariates or stratification factors were included in these tests.
Source: Clinical Study Report.9
Table 28: Summary of Restricted Mean Survival Time for Overall Survival — ITT Population (Data Cut-Off Date: July 15, 2019)
Mean survival time | Oral azacitidine (N = 238) | Placebo (N = 234) |
|---|---|---|
RMST OS (months) on tau = 62.46 months (95% CI)a | |||| | |||| |
Difference on RMST OS (oral azacitidine-placebo) (months) (95% CI) | |||| | |
Ratio on RMST OS (oral azacitidine/ placebo) (95% CI) | |||| | |
CI = confidence interval; OS = overall survival; RMST = restricted mean survival time.
aTau = 62.46 is the minimum of the 2 arms' maximum observed event time.
Source: Clinical Study Report.9
Additional OS Sensitivity Analyses to Address Potential Issues Due to Subsequent Therapies
Table 29: Overall Survival Adjusted for Selected Baseline Factors — ITT Population (Data Cut-Off Date: July 15, 2019)
Variable | Summary of stepwise selectiona | Final modelb | |||
|---|---|---|---|---|---|
Score chi-square | P value | Hazard ratio | 95% CI | P value | |
N = 461 [c] | |||| | |||| | |||| | |||| | |||| |
Treatment (oral azacitidine vs. placebo) | |||| | |||| | |||| | |||| | |||| |
Minimal residual disease status (negative vs. positive) | |||| | |||| | |||| | |||| | |||| |
Central cytogenetic risk status (intermediate vs. poor) | |||| | |||| | |||| | |||| | |||| |
Age (years) | |||| | |||| | |||| | |||| | |||| |
Baseline ANC (log) | |||| | |||| | |||| | |||| | |||| |
First response type (CR vs. CRi) | |||| | |||| | |||| | |||| | |||| |
ANC = absolute neutrophil count; CI = confidence interval; CR = complete remission; CRi = complete remission with incomplete blood count recovery; ECOG = Eastern Cooperative Oncology Group; MDS = myelodysplastic syndrome; NA = not applicable.
aStepwise selection procedure was employed to select covariates into a Cox model that included treatment. The significance level for selecting a covariate into the model was set to 0.25; the significance level for retaining a covariate in the model was set to 0.15. Covariates that were considered but which did not meet the criterion for selection into the model were ECOG classification (0/1 vs 2/3), sex (male, female), prior history of MDS (yes, no), baseline PLAT (log), Time Since first CR or CRi at Rand (days), Response Status at Baseline (CR, CRi, not CR or CRi), Prior Consolidation Therapy (yes, no), and number of cycles of consolidation. Region met the criterion for removal from the model after being selected into the model. Covariates are displayed in the order in which they were selected into the model.
bThe hazard ratio, 95% confidence intervals and P value were estimated from a Cox model that included treatment and the covariates selected through the stepwise procedure described in footnote a.
cSubjects with a missing value for any covariate included in the final model are excluded from the analysis.
Source: Clinical Study Report.9
Table 30: Overall Survival Adjusted for Subsequent Therapy Status and Selected Baseline Factors (Subsequent Therapy as Time-Varying) — ITT Population (Data Cut-Off Date: July 15, 2019)
Variable | Model with subsequent therapya N = 472 | Model with subsequent therapy and baseline covariatesb (N = 461c) | ||||
|---|---|---|---|---|---|---|
HR | 95% CI | P value | HR | 95% CI | P value | |
Treatment (oral azacitidine vs. placebo) | |||| | |||| | |||| | |||| | |||| | |||| |
Subsequent therapy (YES vs NO) | |||| | |||| | |||| | |||| | |||| | |||| |
Treatment x subsequent therapy interaction | |||| | |||| | |||| | |||| | |||| | |||| |
Minimal residual disease status (negative vs positive) | |||| | |||| | |||| | |||| | |||| | |||| |
Central Cytogenetic risk status (intermediate vs poor) | |||| | |||| | |||| | |||| | |||| | |||| |
Age (years) | |||| | |||| | |||| | |||| | |||| | |||| |
Baseline ANC (log) | |||| | |||| | |||| | |||| | |||| | |||| |
First Response type (CR vs. CRi) | |||| | |||| | |||| | |||| | |||| | |||| |
ANC = absolute neutrophil count; CI = confidence interval; CR = complete remission; CRi = complete remission with incomplete blood count recovery; HR = hazard ratio; NA = not applicable.
aThe hazard ratio, 95% confidence intervals, and P values were estimated from a Cox model that included terms for treatment, subsequent therapy status as a time-varying covariate, and the interaction between treatment and subsequent therapy.
bBaseline covariates included in the multivariate Cox model were selected through the stepwise selection procedure. Subsequent therapy status is included in the model as a time-varying covariate.
cSubjects with a missing value for any covariate included in the model are excluded from the analysis.
Source: Clinical Study Report.9
Table 31: Overall Survival Adjusted for Subsequent Therapy Status and Selected Baseline Factors (Subsequent Therapy Status as Binary) — ITT Population (Data Cut-Off Date: July 15, 2019)
Variable | Model with subsequent therapya N = 472 | Model with subsequent therapy and baseline covariatesb (N = 461c) | ||||
|---|---|---|---|---|---|---|
HR | 95% CI | P value | HR | 95% CI | P value | |
Treatment (oral azacitidine vs. placebo) | |||| | |||| | |||| | |||| | |||| | |||| |
Subsequent therapy (YES vs NO) | |||| | |||| | |||| | |||| | |||| | |||| |
Minimal residual disease status (negative vs positive) | |||| | |||| | |||| | |||| | |||| | |||| |
Central cytogenetic risk status (intermediate vs poor) | |||| | |||| | |||| | |||| | |||| | |||| |
Age (years) | |||| | |||| | |||| | |||| | |||| | |||| |
Baseline ANC (log) | |||| | |||| | |||| | |||| | |||| | |||| |
First response type (CR vs. CRi) | |||| | |||| | |||| | |||| | |||| | |||| |
ANC = absolute neutrophil count; CI = confidence interval; CR = complete remission; CRi = complete remission with incomplete blood count recovery; HR = hazard ratio; NA = not applicable.
aThe hazard ratio, 95% confidence intervals, and P values were estimated from a Cox model that included terms for treatment, subsequent therapy status.
bBaseline covariates included in the multivariate Cox model were selected through the stepwise selection procedure. Subsequent therapy status is included in the model as a binary covariate.
cSubjects with a missing value for any covariate included in the model are excluded from the analysis.
Source: Clinical Study Report.9
Table 32: Overall Survival With IPCW [a] Adjustment for Subsequent Therapy — ITT Population (Data Cut-Off Date: July 15, 2019)
Variable | Univariate model | Multivariate modela | ||||
|---|---|---|---|---|---|---|
HR | 95% CI | P value | HR | 95% CI | P value | |
N = 472 [b] | ||||||
Treatment (oral azacitidine vs. placebo) | |||| | |||| | |||| | |||| | |||| | |||| |
Minimal Residual Disease Status (Negative vs. positive) | |||| | |||| | |||| | |||| | |||| | |||| |
Central Cytogenetic risk status (intermediate vs. poor) | |||| | |||| | |||| | |||| | |||| | |||| |
Age (years) | |||| | |||| | |||| | |||| | |||| | |||| |
Baseline ANC (log) | |||| | |||| | |||| | |||| | |||| | |||| |
First Response type (CR vs. CRi) | |||| | |||| | |||| | |||| | |||| | |||| |
ANC = absolute neutrophil count; CR = complete remission; CRi = complete remission with incomplete blood count recovery; HR = hazard ratio; IPCW = inverse probability of censoring weighted; NA = not applicable.
aBaseline covariates included in the multivariate model were selected through the stepwise selection procedure.
bSubjects with missing values for continuous covariates were assigned the mean value for the entire ITT population; missing values for centrally assessed cytogenetic risk was imputed by the cytogenetic risk status in an interactive voice response system.
Source: Clinical Study Report.9
Table 33: Summary of Overall Survival Adjusted for Effects of Subsequent Therapy: CC-486 Versus Placebo — ITT Population (Data Cut-Off Date: July 15, 2019)
Summary | Oral azacitidine (N = 193) | Placebo (N = 208) |
|---|---|---|
Event n (%) | 158 (81.9) | 171 (82.2) |
Censored n (%) | |||| | |||| |
Median OS (95% CI)a | |||| | |||| |
25th Percentile OS (95% CI)a | |||| | |||| |
75th Percentile OS (95% CI)a | |||| | |||| |
HR (oral azacitidine vs. placebo) (95% CI)b | |||| | |
Log-rank test: P valuec | |||| | |
1-year survival estimate (95% CI) | |||||||||||||||||||| | |||||||||||||||||||| |
2-year survival estimate (95% CI) | |||||||||||||||||||| | |||||||||||||||||||| |
CI = confidence interval; HR = hazard ratio; OS = overall survival.
Note: Overall survival estimates have been adjusted according to the imputation method proposed by Luo et al. in 2016.40 This is a regression-based imputation method that provides an adjusted estimate of the original treatment effect when confounding is present due to additional therapy received subsequent to the randomized study treatment.
aMedian, 25th, and 75th percentile estimates of OS are from a Kaplan–Meier analysis.
bThe hazard ratio is from a Cox proportional hazards model.
cThe P value is 2-sided from a log-rank test.
Source: Clinical Study Report.9

||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||9
Note: This figure has been redacted at the sponsor’s request.
Table 34: Summary of Time to Discontinuation From Treatment Due to Disease Relapse — ITT Population (Data Cut-Off Date: July 15, 2019)
Parameter | Oral azacitidine (N = 238) | Placebo (N = 234) |
|---|---|---|
Subjects with treatment discontinued due to disease relapse - n (%) | 143 (60.1) | 180 (76.9) |
Subjects with treatment discontinued due to adverse event – n (%) | 29 (12.2) | 11 (4.7) |
Subjects with treatment discontinued due to eligibility for bone marrow or stem cell transplant – n (%) | 6 (2.5) | 0 |
Subjects with treatment discontinued due to withdrawal of consent/lost to follow-up/protocol violation/other – n (%) | |||| | |||| |
Subjects with treatment discontinued due to death – n (%) | 1 (0.4) | 2 (0.9) |
Censored – n (%) | 45 (18.9) | 26 (11.1) |
Median time to discontinued due to disease relapse (months) (95% CI)a | |||| | |||| |
6-month treatment discontinuation due to disease relapse rate estimate (95% CI)b | |||| | |||| |
1-year treatment discontinuation due to disease relapse rate estimate (95% CI)b | |||| | |||| |
2-year treatment discontinuation due to disease relapse rate estimate (95% CI)b | |||| | |||| |
CI = confidence interval; ITT = intention-to-treat.
Note: Time to discontinuation from treatment is defined as the interval (in months) from the date of randomization to the date of discontinuation from study drug.
aUnstratified Kaplan–Meier analysis
bEstimates of treatment discontinuation due to disease relapse rate is based on the cumulative incidence function from a competing risk analysis with treatment discontinuation due to other reasons as competing risk.
Source: Clinical Study Report.9
Table 35: Efficacy and Safety Results During Dose Escalation Period — ITT Population With Dose Escalation (Data Cut-Off Date: July 15, 2019)
Result | Oral azacitidine (N = 51) | Placebo (N = 40) | ||
|---|---|---|---|---|
Evaluable patients,a n (%) [95%CI]c | 43 (84.3) [71.4 to 93.0] | 35 (87.5) [73.2 to 95.8] | ||
Not evaluable patients,a n (%) [95%CI]c | 8 (15.7) [7.0 to 28.6] | 5 (12.5) [4.2 to 26.8] | ||
Response | ||||
Overall response (CR + CRi)b, n (%) [95%CI]c | 10 (23.3) [11.8 to 38.6] | 4 (11.4) [3.2 to 26.7] | ||
Best response, n (%) [95%CI]c | ||||
CR | |||| | |||| | ||
CRi | |||| | |||| | ||
RFS | ||||
Event, n (%) | |||| | |||| | ||
Censored, n (%) | |||| | |||| | ||
Median RFS, months (95% CI) | |||| | |||| | ||
Hazard Ratio (95% CI)d | |||| | |||
OS | ||||
Events, (%) | |||| | |||| | ||
Censored, n (%) | |||| | |||| | ||
Median OS, months (95% CI) | 22.8 (17.7 to 30.1) | 14.6 (11.6 to 22.3) | ||
Hazard Ratio (95% CI)e | 0.66 (0.42 to 1.04) | |||
Treatment exposure | ||||
Treatment duration, median, months (range)f | |||| |||| | |||| |||| | ||
First occurrenceg of TEAEs by dose schedule period | ||||
300 mg × 14 days | 300 mg × 21 days | 300 mg × 14 days | 300 mg × 21 days | |
Febrile neutropenia, n (%) | |||| | |||| | |||| | |||| |
Thrombocytopenia, n (%) | |||| | |||| | |||| | |||| |
Anemia, n (%) | |||| | |||| | |||| | |||| |
CI = confidence interval; CR = complete remission; CRi = complete remission with incomplete blood count recovery; Event = relapse or death; IWG = International Working Group; ITT = intention-to-treat; KM = Kaplan–Meier; NE = not estimable; PR = partial Remission; RFS = relapse-free survival.
Note: Dose escalation is defined as subject with at least 1 assigned dose of 300 mg x 21 days. Dose escalation period is defined from the first dose date of dose escalation until treatment end date, which is the earlier of (last dose date + 7 days) and death date.
aSubject is considered not evaluable if the last bone marrow blast on or before the first dose date of dose escalation is < 5%, otherwise, the subject is considered evaluable. Percentage is calculated in the patients within each treatment arm.
bPatients achieving respective response. Percentage is calculated in the evaluable patients within each treatment arm. The response was derived programmatically based on the IWG AML response criterion using clinical data.
c95% confidence intervals are calculated using the exact binomial test.
dThe hazard ratio is from a Cox proportional hazards model stratified by age, cytogenetic risk category, and receipt of consolidation therapy or not.
eThe hazard ratio is from a Cox proportional hazards model.
fTreatment duration in months is defined as (treatment end date — first dose date of 300 mg × 21 days + 1)/30.4375, where treatment end date is last dose date + 7 days (the prescribed rest period for 300 mg × 21 days), or the death date, whichever is earlier.
gFrequency ≥ 10% of patients in either study group.
Source: Clinical Study Report9
Table 36: Baseline Disease Characteristics in the HRQoL-Evaluable Population (Data Cut-Off Date: July 15, 2019)
Characteristic | Oral azacitidine (N = 225) | Placebo (N = 219) |
|---|---|---|
WHO AML classification, n (%) | ||
AML with recurrent genetic abnormalities | |||| | |||| |
AML with myelodysplasia-related changes | |||| | |||| |
Therapy-related myeloid neoplasm | |||| | |||| |
AML not specified | |||| | |||| |
Missing | |||| | |||| |
Type of AML, n (%) | ||
Primary | |||| | |||| |
Secondary | |||| | |||| |
Response status (CR or CRi) after induction therapy (with or without consolidation therapy), n (%) | ||
CR | |||| | |||| |
CRi | |||| | |||| |
Prior history of MDS or CMML, n (%) | ||
Yes | |||| | |||| |
Primary | |||| | |||| |
Secondary | |||| | |||| |
Cytogenetic risk category at time of induction therapy, n (%) | ||
Intermediate | |||| | |||| |
Poor | |||| | |||| |
Consolidation therapy following induction, n (%) | ||
Yes | |||| | |||| |
1 cycle | |||| | |||| |
2 cycles | |||| | |||| |
3 cycles | |||| | |||| |
4 cycles | |||| | |||| |
No | |||| | |||| |
ECOG PS, n (%) | ||
0 | |||| | |||| |
1 | |||| | |||| |
2 | |||| | |||| |
3 | |||| | |||| |
Minimal residual disease status from central pathology report, n (%) | ||
Positive | |||| | |||| |
Negative | |||| | |||| |
Missing | |||| | |||| |
Bone marrow blast (%) | ||
N | |||| | |||| |
Mean (SD) | |||| | |||| |
Median | |||| | |||| |
First quartile to third quartile | |||| | |||| |
Minimum to maximum | |||| | |||| |
Time from initial AML diagnosis to randomization (months) | ||
N | |||| | |||| |
Mean (SD) | |||| | |||| |
Median | |||| | |||| |
Q1, Q3 | |||| | |||| |
Minimum to maximum | |||| | |||| |
Time from start of induction therapy to randomization (months) | ||
N * | |||| | |||| |
Mean (SD) | |||| | |||| |
Median | |||| | |||| |
First quartile to third quartile | |||| | |||| |
Minimum to maximum | |||| | |||| |
AML = acute myeloid leukemia; CR = complete remission; CRi = complete remission with incomplete blood count recovery; CMML = chronic myelomonocytic leukemia; ECOG = Eastern Cooperative Oncology Group; FACIT = Functional Assessment of Chronic Illness Therapy; HRQoL = health-related quality of life; MDS = myelodysplastic syndrome; SD = standard deviation.
HRQoL-evaluable population were ITT subjects with a non-missing assessment at baseline and ≥ 1 non-missing post-baseline assessment based on FACIT-F scale.
*One subject in the CC-486 group and 2 subjects from the placebo group with missing start time of induction therapy.
Source: Clinical Study Report.9
Appendix 4: Description and Appraisal of Outcome Measures
Note that this appendix has not been copy-edited.
To describe the following outcome measures and review their measurement properties (validity, reliability, responsiveness to change, and MID):
EQ-5D-3L
FACIT-F
Physical Impairment Numeric Rating (PINR) Scale
Findings
Table 37: Summary of Outcome Measures and Their Measurement Properties
Outcome measure | Type | Conclusions about measurement properties | MID |
|---|---|---|---|
EQ-5D-3L | Generic, utility-based measure of HRQoL, consisting of an index score (with 5 dimensions: mobility, self-care, usual activities, pain/ discomfort, and anxiety/depression) and a VAS. | Validity:
Reliability: Test–retest reliability assessed using ICC, good reproducibility shown. Responsiveness: No relevant studies found. Ceiling and floor effect: Moderate to high ceiling effect across domains, no floor effect observed. | Not identified in the literature in patients with AML. MID ranged from 0.06 to 0.12 for EQ-5D-3L index score, and from 6 to 11 for EQ VAS in cancer patients29,30; other reported MIDs range from 0.033 to 0.074.31 |
FACIT-F | 13-item patient-reported measure of fatigue that assesses self-reported fatigue and its impact on daily activities and function based on 5-point Likert-type scales. Consists of 5-item symptom subscale and an 8-item impact subscale. | Psychometric properties assessed in patients in CLL. Validity:
| Not identified in the literature for AML or leukemia patients. MIDs reported for other conditions range from 3 to 4 points for the total score.35,36 |
FACIT-F (continued) | Known-group and discriminative validity was assessed. Both approaches to validity assessment showed to distinguish groups based on various baseline factors. Reliability:
Responsiveness: No relevant studies found. Ceiling and floor effect: Some ceiling effects were observed in the study population, however; no floor effect observed. | ||
PINR Scalea | 3-item self-administered tool used to measure the level of physical impairment, difficulty completing outdoor physical tasks, and difficulty completing indoor tasks over the past 24 hours, based on an 11-point numerical scale. | Validity: No relevant studies found. Reliability: No relevant studies found. Responsiveness: No relevant studies found. Ceiling and floor effect: No relevant studies found. | Not identified in the literature for AML or leukemia patients. As noted in the sponsor’s submission, the MID the PINR scale is not available in the literature, therefore a half of the standard deviation at baseline from all ITT patients was used and considered an approximation for MID.61,62 |
AUROC = area under the receiver operating characteristics curve; CFA = confirmatory factor analysis; CLL = chronic lymphocytic leukemia; EQ-5D-3L = EuroQol 5-Dimensions 3-Levels; FACIT-F = Functional Assessment of Chronic Illness Therapy–Fatigue; HRQoL = health-related quality of life; ICC = intraclass correlation coefficient; MID = minimal important difference; PINR = Physical Impairment Numeric Rating; SAP = statistical analysis plan; VAS = Visual Analogue Scale.
aExploratory patient-reported outcome.
EQ-5D-3L
Description
The EQ-5D is a generic, utility-based measure of HRQoL. The EQ-5D is a 2-part questionnaire, consisting of the EQ-5D descriptive system and the EQ VAS.63
For the descriptive system of the EQ-5D, respondents are asked to indicate their health status that day (i.e., a 1-day recall) on 5 dimensions (mobility, self-care, usual activities, pain/ discomfort, and anxiety/depression). The 3-level version of the EQ-5D (i.e., the EQ-5D-3L) was used in the QUAZAR AML-001 trial, which has response options for each of the 5 dimensions that reflect 3 possible levels of functioning.
Level 1: No problems
Level 2: Some problems
Level 3: Extreme problems
The rating on each dimension is combined to create a descriptive health profile (referred to as the health state description) that is a vector of the levels. For example, an individual with no health problems on any dimension would have a health profile of 11111, while a person with extreme problems on all dimensions would have a health profile of 33333. The numerical values assigned to the levels 1, 2, or 3 for each dimension reflect rank order categories of function. There are 243 unique health profiles or vectors that exist for the EQ-5D-3L.63
Scoring
Index Scores
The health profile (health state description or vector) defined by the EQ-5D-3L questionnaire is used to create an overall index score. To create the EQ-5D-3L index score, a scoring algorithm (a mathematical equation termed a utility function) is applied to the vector. Various scoring algorithms for the EQ-5D-3L have been derived by determining the societal preferences for its 243 health states (i.e., by assessing how much value society places on each health state) using techniques such as the standard gamble or TTO. In all scoring algorithms of the EQ-5D-3L, a score of 0 represents the health state “dead” and 1.0 reflects “perfect health.” Negative scores are also possible for those health states that society (not the individual patient) considers to be “worse than dead.” In the QUAZAR AML-001 trial, the EQ-5D-3L health utility index scores were derived according to published guidance for the primary analyses, using Canadian population sample weights, which yields health utility scores ranging from −0.309 to 1.0.
Visual Analogue Scale Scores
The EQ-5D VAS is a distinct component of the EQ-5D questionnaire. The VAS score is determined by asking respondents to rate their health that day on a vertical line, with anchors (end points) labelled “Worst imaginable health state” at 0 and “Best imaginable health state” at 100. While the EQ-5D index score reflects societal preferences for the health state, the VAS captures the individual’s own value or judgment of his or her present health state. The EQ-5D VAS scores are not used to create utility scores but provide complementary information to the EQ-5D-3L index score.
Psychometric properties
Validity
One recent study compared the psychometric properties of EQ-5D-3L and EQ-5D-5L in 168 patients with AML in China.28 Patients completed 2 rounds of interviews within 2 to 3 days using the 2 versions of the EQ-5D, administered by interviewers. Following this, the ceiling effect was measured by the proportion of ‘no problems’ in each dimension of the descriptive system and the proportion of patients who reported full health state (11111). Similarly, the floor effect was measured by the number and percentage of the worst level in each dimension and across all dimensions (33333). At least 15% of respondents reporting lowest or highest level was considered as having a floor or ceiling effect, respectively. The 3L version showed ceiling effect (35.1% respondents reported full health), with mobility, self-care, usual activities domains showing the highest ceiling effect, followed by pain/discomfort and anxiety/depression. No floor effect was observed in EQ-5D-3L.28
Convergent validity was assessed by comparing EQ-5D-3L with Functional Assessment of Cancer Therapy- General (FACT-G); weak to moderate correlations were found between similar dimensions of EQ-5D-3L and the subscales of FACT-G (all 5 dimensions of EQ-5D-3L and the physical well-being, social/family well-being, emotional well-being, and functional well-being dimensions of FCT-G) (Spearman’s rank correlation coefficient ranged from −0.09 to −0.46 across dimensions). However, the correlation between total score of FACT-G and EQ-5D-3L index was strong (r = 0.60).28
Known-group validity was assessed by defining the groups by self-reported health status (excellent, very good, good, fair, and poor), ECOG status (0 to 3 + ), number of complications, risk category (favourable, intermediate, poor/adverse) and anxiety (mild to severe), depression (normal to moderate), and social support levels (low to high). The respondents who had lower ECOG performance score, higher self-rated health status, no complication, lower risk, less anxiety, milder depression, or higher social support, tended to have a higher EQ-5D index score (P < 0.05, data not presented).28
The ability of the EQ-5D-3L to discriminate was assessed using the area under the receiver operating characteristics curve (AUROC), with 3 external indicators of health status (i.e., self-reported status, ECOG status, and risk category) dichotomized in all possible ways. EQ-5D-3L showed a good distinction between all dichotomous configurations, with AUROC ranging from 0.77 to 0.91.28
Reliability
Authors of the above study also assessed the test–retest reliability using the ICC for the EQ-5D-3L index scores and using the weighted Cohen kappa coefficient for the 5-dimensional responses. The weighted kappa coefficient for EQ-5D-3L ranged from 0.59 for ‘Anxiety/depression’ and ‘Self-care’ to 0.69 for ‘Pain/discomfort’; indicating moderate to substantial agreement. The ICC for the index score were 0.77, which also indicated good reproducibility.28
Studies evaluating the responsiveness of the instrument was not found.
MID
Studies evaluating the MID of EQ-5D-3L specifically in patients with leukemia were not identified from the literature search. A number of hematology/oncology studies were referenced by the sponsor from the published literature, which showed several MID ranges based on different algorithms used. For example, for the EQ-5D-3L health utility index score, 1 study estimated an MID of 0.09 to 0.12 in cancer patients using an algorithm based on the UK population weights, and 0.06 to 0.09 based on United States (US) weights.29 In another study, the sponsor noted a MID of 0.08 for improvement and 0.10 for deterioration based on the data from patients with multiple myeloma.30 The sponsor also highlighted that for the EQ-5D VAS, an MID of 6 to 11 was reported based on patients with cancers.29 Ultimately, the sponsor used an MID of −0.10 for the EQ-5D-3L health utility index and −11 for EQ VAS for statistical analysis in the QUAZAR AML-001 trial. Other reported MIDs for the index score of the scale have ranged from 0.033 to 0.074 in the general population.31
FACIT-F
Description
The Functional Assessment of Chronic Illness Therapy (FACIT) – Fatigue Version 4 is a 13-item patient-reported measure of fatigue that assesses self-reported fatigue and its impact on daily activities and function. It consists of a 5-item symptom subscale and an 8-item impact subscale. It has a 7-day recall period.64
Scoring
Items of FACIT-F are rated on a 5-point Likert-type scale, i.e., 0 to 4 response scale, with anchors ranging from “Not at all” to “Very much so.” For scoring, all items are summed to create a single fatigue score with a range from 0 to 52. Items are reverse scored when appropriate to provide a scale in which higher scores represent better functioning or less fatigue. For the FACIT-F total and 2 subscales, the higher the score the better the QoL.64
Psychometric properties
Validity
No study was found assessing the psychometric properties of FACIT-F in patients with AML. One recent study assessed the psychometric properties of the FACIT-F in patients with CLL.33 Content validity was evaluated using cognitive debriefing (qualitative) interviews conducted with 40 patients with CLL in the first-line or relapsed or refractory setting (self-reported ECOG PS score ≤ 2). Patients were asked to complete and review the instrument with respect to instructions (clarity, difficulty understanding), individual items, response options and the questionnaire as a whole (missing and redundant items). Interviewed patients considered the FACIT-F items reflective of their CLL experience, understood most terminologies well and agreed with response options.33
Additionally, authors of the above study investigated structural validity, internal consistency, construct and known-groups validity using data from a phase III RCT (ASCEND trial) in 263 patients with relapsed or refractory CLL (who had previously been treated with at least 1 systemic therapy and had an ECOG PS score ≤ 2). Confirmatory factor analysis was employed to evaluate the latent structure (i.e., underlying subscales) of the FACIT-F instrument. Results based on fit statistics of the CFA models (root mean square error of approximation, standardized root mean square residual, and comparative fit index) and factor loadings (> 0.3) suggested unidimensionality of the FACIT-F scale, including the rationale to combine the 2 individual subscales.33
Construct validity was examined by exploring associations between the FACIT-F and the European Organization for the Research and Treatment of Cancer Quality of Life Questionnaire (QLQ-C30) (for convergent validity) and EQ VAS (for divergent validity), using Spearman’s rank correlation coefficients. As hypothesized, strong correlations were found between the FACIT-F total and subscale scores with the EQ VAS and relevant QLQ-C30 scales (global health status, physical function, role function and fatigue scale scores, all Spearman r ≥ 0.5), demonstrating convergent validity. Weak correlations (Spearman r < 0.30) were observed between the FACIT-F scales, and the QLQ-C30 insomnia, constipation and diarrhea scales, as hypothesized, indicating no relationship between fatigue and these symptoms, supporting divergent validity.33
Known-groups validity was assessed by comparing FACIT-F scores between groups expected to differ in their level of fatigue. Groups were based on ECOG PS score (0 [fully active] vs 1 or 2 [restricted activity but still ambulatory and capable of all self-care]), hemoglobin level (≥ 110 g/L [no/mild anemia] vs < 110 g/L [moderate/severe anemia]) and constitutional symptoms (night sweats, fever, unexplained weight loss, significant fatigue [none versus ≥ 1 symptom]). As hypothesized, there were significant differences between groups defined by baseline ECOG PS score, hemoglobin level, and constitutional symptoms (data not presented here).33
The presence of floor effects (> 25% of patients scoring ‘worst possible health state’) and ceiling effects (> 25% of patients scoring “best possible health state”) was assessed. Some ceiling effects were observed in the study population: the proportion of patients who answered “not at all” (indicating that they did not have the symptom or impact) was above 25% for 8 subscales. However, the authors reported that only 1 of the 5 symptom subscale items showed a ceiling effect; whereas 7 of the 8 impact subscale items showed a ceiling effect, indicating that there was low impact of fatigue on activities. No floor effects were observed.33
Reliability
Internal consistency reliability was assessed using the Cronbach alpha, which was greater than 0.85 for the FACIT-F symptom subscale, the impact subscale and the total scale, demonstrating good internal consistency.33
MID
Studies evaluating the MID of FACIT-F in patients with leukemia were not identified by the literature search. Based on findings of 2 studies of cancer patients, the sponsor reported the MID for FACIT-F in the range of 3 to 4 points.35,36 Notably, the sponsor used an MID of −3 for worsening in the FACIT-F scale for statistical analysis in the QUAZAR AML-001 trial.
PINR Scale
Description
The PINR scale is a 3-item self-administered tool asking patients to rate their level of physical impairment, difficulty completing outdoor physical tasks, and difficulty completing indoor tasks over the past 24 hours.
Scoring
Items of PINR are rated on an 11-point numerical scale, i.e., 0 to 10, with anchors ranging from “no impairment or no difficulty” to “worse impairment and extremely difficult” The higher the score, the more difficulty or impairment experienced over the past 24 hours. Items were scored separately (i.e., each item was scored and not combined into 1 overall score).
Psychometric properties
Validity
No study was found assessing the psychometric properties related to validity for PINR in patients with AML or leukemia. No studies of the validity of the PINR were provided in the Sponsor’s submission to CADTH.
Reliability
No study was found assessing the psychometric properties related to reliability for PINR in patients with AML or leukemia.
No studies of the reliability of the PINR were provided in the Sponsor’s submission to CADTH.
MID
A MID for PINR was not identified in the literature for AML or leukemia patients. It was noted in sponsor’s submission that the MID for the PINR scale is not available in the literature. As a result, a half of the standard deviation at baseline from all ITT patients was used and considered an approximation for MID for statistical analysis in the QUAZAR AML-001 trial.61,62
Pharmacoeconomic Review
Abbreviations
AE
adverse event
AML
acute myeloid leukemia
BSC
best supportive care
CR
complete remission
CRi
complete remission with incomplete blood count recovery
HSCT
hematopoietic stem cell transplant
ICER
incremental cost-effectiveness ratio
KM
Kaplan-Meier
OS
overall survival
PSM
partitioned survival model
QALY
quality-adjusted life-year
RFS
relapse-free survival
Executive Summary
Item | Description |
|---|---|
Drug product | Azacitidine (Onureg), tablets |
Submitted price | Azacitidine, 200 mg and 300 mg tablets: $952 per 200 mg $1,428 per 300 mg |
Indication | Health Canada indication as per product monograph: maintenance therapy in adult patients with AML who achieved CR or CRi following induction therapy with or without consolidation treatment and who are not eligible for HSCT |
Health Canada approval status | NOC |
Health Canada review pathway | Priority review |
NOC date | January 5, 2021 |
Reimbursement request | As per indication |
Sponsor | Celgene Inc., a Bristol Myers Squibb Company |
Submission history | Previously reviewed: No |
AML = acute myeloid leukemia; CR = complete remission; CRi = complete remission with incomplete blood count recovery; HSCT = hematopoietic stem cell transplant; NOC = Notice of Compliance.
Table 2: Summary of Economic Evaluation
Component | Description |
|---|---|
Type of economic evaluation | Cost-utility analysis Partitioned survival model |
Target population(s) | Adult patients with AML who achieved CR or CRi following induction therapy with or without consolidation treatment and who are not eligible for HSCT; reimbursement request aligns with Health Canada–approved indication |
Treatment(s) | Oral azacitidine 200 mg or 300 mg (Onureg) in combination with best supportive care |
Comparator(s) | Best supportive care (i.e., no active therapy), which includes red blood cell and platelet transfusions; use of an erythropoiesis-stimulating agent; antibiotic, antiviral, and/or antifungal therapy; nutritional support; and/or granulocyte colony-stimulating factor for patients experiencing neutropenic infections. |
Perspective | Canadian publicly funded health care payer |
Outcome(s) | QALYs, LYs |
Time horizon | Lifetime (20 years) |
Key data source | RFS, OS, and treatment duration: QUAZAR AML-001 trial (data cut-off: July 15, 2019) |
Submitted results | ICER for azacitidine + BSC compared with BSC: of $348,988 per QALY (incremental costs: $316,994; incremental QALYs: 0.91) |
Key limitations |
|
CADTH reanalysis results |
|
AML = acute myeloid leukemia; BSC = best supportive care; CR = complete remission; CRi = complete remission with incomplete blood count recovery; HSCT = hematopoietic stem cell transplant; ICER = incremental cost-effectiveness ratio; LY = life-year; OS = overall survival; RFS = relapse-free survival; QALY = quality-adjusted life-year.
Conclusions
Evidence from the CADTH Clinical Review and the QUAZAR AML-001 trial suggested that oral azacitidine led to a statistically significant improvement in relapse-free survival (RFS) and overall survival (OS) among patients with acute myeloid leukemia (AML) who were in remission after chemotherapy when compared to best supportive care (BSC).
CADTH attempted to address limitations identified in the sponsor’s base case by considering dose extension in the calculation of oral azacitidine costs in a revised base case. Both the sponsor and CADTH base cases suggest that oral azacitidine is not cost-effective compared to BSC at conventionally accepted willingness-to-pay thresholds. The CADTH base case suggests that the incremental cost-effectiveness ratio (ICER) for oral azacitidine plus BSC is $355,554 per quality-adjusted life-year (QALY) compared with BSC alone. A price reduction of at least 85% would be required to reach a willingness-to-pay threshold of $50,000 per QALY.
The cost-effectiveness of oral azacitidine was highly sensitive to statistical models used to fit RFS and OS data. Other key drivers included health utility values and the treatment duration. Findings from the CADTH reanalyses likely underestimate the true value of the ICER and the price reduction, as CADTH could not address the critical limitation regarding the implied post-relapse survival benefit of oral azacitidine due to constraints introduced by the submitted model structure. Oral azacitidine is not likely to be cost-effective at conventional thresholds without a large reduction in price.
Stakeholder Input Relevant to the Economic Review
This section is a summary of the feedback received from the patient groups, registered clinicians, and drug plans that participated in the CADTH review process (specifically, information that pertains to the economic submission).
One patient group, the Leukemia and Lymphoma Society of Canada, provided input for the review of oral azacitidine for the treatment of AML via an online survey created by the society. Patients stated that a range of AML symptoms negatively affects their physical, psychological, and social well-being. Patients reported that fatigue, nausea, numbness or body aches, being immunocompromised, losing vision in an eye, among other symptoms, contributed to being unable to participate in daily life, work, or exercise. Most patients noted that they received chemotherapy and a stem cell or bone marrow transplant after diagnosis, while others had received drug or radiation therapy and 1 patient had received chimeric antigen receptor T-cell therapy. Altogether, patients reported poor experiences with available AML treatments, particularly with chemotherapy and stem cell transplants, and expressed that various treatment-related side effects on their physical health, mental health, and social and educational development had negative impacts on their quality of life. None of the patients reported having experience with oral azacitidine; however, patients expressed hope that any new AML drug or treatment would help maintain remission, result in fewer side effects, be offered at a lower cost, and be accessible in their neighbourhoods. Additional social and emotional support was also desired by patients. Last, patients noted the negative impacts of AML on their caregivers, such as stress, worry, and sadness.
Registered clinician input described an unmet need for AML patients previously treated with induction therapy, with or without consolidation therapy, who are in complete remission (CR) or complete remission with incomplete blood count recovery (CRi) and ineligible for a transplant. Registered clinicians stated that there are no other maintenance treatments available for the highly lethal cancer with poor outcomes, as patients are typically followed by surveillance thereafter. Registered clinicians further noted that maintenance azacitidine has been the first treatment to demonstrate an OS benefit in the decades since treatment with 7 days of cytarabine plus 3 days of daunorubicin became the standard of care. Registered clinicians indicated that oral azacitidine may be continued in the absence of an overt clinical relapse and may be discontinued upon disease progression or treatment intolerance. The clinician group further highlighted the ease of use of oral azacitidine as it can be administered at home.
The drug plans highlighted considerations for the implementation of oral azacitidine that are relevant to the economic analysis. Drug plan input noted several factors that may affect patient treatment costs. As the list price per tablet is high, dose adjustments or modifications and days of extended treatment may have a significant impact on treatment costs and contribute to a higher risk of drug wastage, even if the drug were to be supplied in 7-day blister packs. Additionally, drug plans noted costs associated with granulocyte colony-stimulating factor, which is required to treat febrile neutropenia, a possible adverse effect of oral azacitidine.
Several of these concerns were addressed in the sponsor’s model:
Clinical benefits (i.e., RFS and OS) and adverse events (AEs) associated with oral azacitidine, including neutropenia, thrombocytopenia, anemia, febrile neutropenia, diarrhea, and leukopenia, were considered.
The sponsor submitted a scenario analysis from a societal perspective to account for lost productivity attributed to each health state. CADTH reported a societal perspective as 1 of the scenario analyses.
Drug plans expressed a concern about the cost of granulocyte colony-stimulating factor. The sponsor considered this treatment as part of BSC and assumed no differences in BSC between patients treated with oral azacitidine and those on no active therapy.
CADTH addressed some of these concerns as follows:
Drug plans raised concern about the impact of dose adjustments, days of extended treatment, and potential drug wastage on treatment costs. CADTH considered dose extension and drug wastage in its revised base case.
Registered clinicians indicated that oral azacitidine may be continued in the absence of relapse and may be discontinued upon disease progression or treatment intolerance. CADTH performed scenario analyses by replacing the mean number of treatment cycles with its lower and upper bounds.
Economic Review
The current review is for oral azacitidine (Onureg) for adult patients with AML who achieved CR or CRi following induction therapy with or without consolidation treatment and who are not eligible for hematopoietic stem cell transplant (HSCT).
Economic Evaluation
Summary of Sponsor’s Economic Evaluation
Overview
The sponsor submitted a cost-utility analysis comparing costs and outcomes for oral azacitidine in combination with BSC (oral azacitidine) with no active therapy combined with BSC (no active therapy) for adults with AML who achieved CR or CRi following induction therapy with or without consolidation treatment, and who are not eligible for HSCT. The modelled population was in line with the reimbursement request and Health Canada–approved indication.
Oral azacitidine is available as 200 mg and 300 mg tablets. The recommended dosage is 300 mg once daily for 14 days every 28-day treatment cycle. The per-cycle cost of oral azacitidine 300 mg was estimated to be $19,992. The sponsor focused on oral azacitidine 300 mg and the 14-day dosing schedule in its base case. No drug costs were assigned to the comparator of no active therapy. The sponsor assumed 100% compliance in its calculations.
The clinical outcome was QALYs and life-years. The economic analysis was undertaken over a lifetime horizon of 20 years from the perspective of a Canadian publicly funded health care system. Costs and QALYs were discounted at a rate of 1.5% per annum.
Model Structure
The sponsor submitted a partitioned survival model (PSM) with 3 health states: RFS, relapse, and death (Appendix 3, Figure 1). The proportion of patients who were relapse-free, who experienced relapse, or who were dead at any time over the model horizon was derived using joint curves and the log-normal distribution. All patients entered the RFS and were assumed to receive treatments until more than 15% of blasts were observed in peripheral blood or bone marrow or until unacceptable toxicity occurred. Patients could discontinue treatment but remain in the RFS health state. At the end of each cycle, the proportion of patients who relapsed or died was derived based on the area under the survival curves. Specifically, OS was partitioned to estimate the proportion of patients in the death state, while OS and RFS curves were used to estimate the proportion of patients in the relapse-free health state. The difference between the OS and RFS curves was partitioned at each time point to estimate the proportion of patients in the relapse health state. Relapse was defined as the earliest date of any of the following: 5% or more bone marrow blasts from the central pathology report; the appearance of greater than 0% blasts in the peripheral blood with a later bone marrow confirmation (bone marrow blast ≥ 5%) within 100 days; or at least 2 peripheral blasts of 5% or more within 30 days.
Model Inputs
The modelled population reflected the baseline patient characteristics of the QUAZAR AML-001 trial,1 an ongoing phase III, international, multicentre, randomized, double-blind, placebo-controlled study comparing the efficacy and safety of oral azacitidine plus BSC versus placebo plus BSC as maintenance therapy among patients with AML who are in CR or CRi after intensive chemotherapy. The submitted model assumed a mean age of 67.9 years, a mean weight of ||||| kg, and an average height of |||| cm.
The RFS and OS curves for oral azacitidine and no active therapy were generated using patient-level data from the QUAZAR AML-001 trial (data cut-off: July 15, 2019). The sponsor predicted OS and RFS curves using a joint curve model, which fitted a log-normal model to individual patient–level data using a treatment indicator (azacitidine versus no active therapy) as a covariate. The log-normal distribution was selected for both RFS and OS data based on clinical validity and statistical fit.
The model accounted for grade 3 or 4 AEs occurring in at least 5% of patients in the treatment arm of the QUAZAR AML-001 trial population. The proportion of patients receiving a subsequent therapy, and the mix of subsequent therapies, was informed by the QUAZAR AML-001 trial and was validated by clinical experts. The sponsor modelled HSCT as part of subsequent therapy and assumed that the proportions of receiving HSCT were 6.3% for patients receiving oral azacitidine and 13.7% for patients on no active therapy, as reported in the QUAZAR AML-001 trial.
State-specific health utility values were based on a UK study2 that used a time trade-off methodology to elicit health state utilities for AML from 210 members of the UK general population. The sponsor did not use health utility values measured in the QUAZAR AML-001 trial because the trial did not capture data on health-related quality of life for patients beyond the treatment period and into relapse. Utility decrements were included in the model to capture the impact of AEs and HSCT on health-related quality of life.
Costs included those associated with drugs (acquisition and premedication), disease management, subsequent therapy, AEs, and end-of-life care. The cost of oral azacitidine was based on sponsor data on file and a mean time on treatment of |||| cycles reported in the QUAZAR AML-001 trial. Subsequent treatment costs were estimated based on the proportion of patients receiving each subsequent therapy, per-cycle drug acquisition, treatment administration costs obtained from the published sources, and the estimated treatment duration. The model also considered disease management costs for physician and nurse visits, laboratory tests, chemistry and liver panels, blood transfusions, and bone marrow aspirates and biopsies. The frequency of resource use for each health state and treatment arm was informed by clinical experts. The proportion of patients receiving red blood cell and platelet transfusions in relapse was informed by the QUAZAR AML-001 trial. Costs of BSC were included in the model to capture ongoing disease management costs. It was assumed that there were no differences in BSC between patients receiving oral azacitidine and those on no active therapy. Costs related to HSCT were based on the average cost per procedure in Ontario, as reported by Ontario Health. Unit costs for each AE were obtained from the Ontario Case Costing Initiative database and the 2020 Ontario Schedule of Benefits, Physician Services. The cost of end-of-life care was applied as a one-off cost to patients who transitioned to the “death” health state; the costs were based on a study by Bekelman et al. (2016),3 which reported hospital costs for the 30 days preceding death for cancer patients in Canada (Ontario) and 6 other developed countries.
Summary of Sponsor’s Economic Evaluation Results
All analyses were run probabilistically with 5,000 iterations, with the deterministic and probabilistic results being comparable. The probabilistic findings are presented here. No drug costs were assigned to patients in the group receiving no active therapy.4
Base-Case Results
In the sponsor’s base-case analysis, oral azacitidine was associated with an ICER of $348,988 per QALY compared to no active therapy over a lifetime horizon (Table 3). At a willingness-to-pay threshold of $50,000 per QALY, the probability of oral azacitidine being cost-effective was 0% compared to no active therapy.4
The main driver of the incremental costs was drug cost (94.1%), followed by HSCT costs (2.9%) and disease management costs (1.2%). At the end of the model time horizon of 20 years, 1.9% of patients in the model were still alive. A breakdown of the sponsor-submitted results for the base-case population by trial duration and extrapolation period shows that 38.2% of the expected QALY gains come from the time beyond the trial period.
Table 3: Summary of Sponsor’s Economic Evaluation
Drug | Total costs ($) | Incremental costs ($) | Total QALYs | Incremental QALYs | ICER oral azacitidine + BSC vs. no active therapy + BSC ($ per QALY) |
|---|---|---|---|---|---|
No active therapy + BSC | 72,568 | Ref. | 1.75 | Reference | Reference |
Oral azacitidine + BSC | 389,562 | 316,994 | 2.66 | 0.91 | 348,988 |
BSC = best supportive care; ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year; vs. = versus.
Source: Sponsor’s pharmacoeconomic submission.4
Additional results from the sponsor’s submitted economic evaluation base case are presented in Appendix 3.
Sensitivity and Scenario Analysis Results
The sponsor performed scenario analyses by varying discount rates, reducing the time horizon to 15 years, using a societal perspective, considering dose extension or reduction, using alternative health utility values, and using alternative parametric survival models to predict RFS and OS curves. Treatment efficacy was the most influential driver of the cost-effectiveness results. Compared to no active therapy, the estimated ICERs of oral azacitidine ranged between $247,532 (assuming the lower bound of hazard ratios for RFS and OS) and $1,090,868 (assuming the upper bound of hazard ratios for RFS and OS) per QALY. Other key determinants included time on treatment and health utility values.
CADTH Appraisal of the Sponsor’s Economic Evaluation
CADTH identified several key limitations to the sponsor's analysis that have notable implications on the economic analysis:
Highly uncertain comparative effectiveness estimates: Results from the sponsor’s model suggested that oral azacitidine was associated with longer survival after relapse. While the QUAZAR AML-001 trial showed a significant impact of oral azacitidine on RFS and OS, there was no clear mechanism by which oral azacitidine would continue to provide clinical benefit after relapse. The sponsor’s use of a PSM, while methodologically appropriate for the decision question, introduces structural assumptions about the relationship between RFS and OS that could not be adjusted for in reanalysis. These assumptions may produce a post-relapse survival bias that favours azacitidine. Due to the structural independence between OS and RFS end points assumed in a PSM, extrapolations for each end point may reflect within-trial trends in the rates of relapse and death.
CADTH asked the sponsor to provide additional evidence to support the implied post-relapse benefit (0.2 incremental life-years; 0.1 incremental QALYs). The CADTH Clinical Review team and clinical experts consulted by CADTH examined this evidence and concluded that it was not sufficient to explain the post-relapse survival difference observed in the sponsor’s model. CADTH was unable to determine the extent to which the implied post-relapse benefit was due to the effect of treatment versus structural bias within the PSM, and could not address this in reanalysis. CADTH performed a scenario analysis in which alternate distributions for OS and RFS were explored.
Additional uncertainty around comparative effectiveness was contributed by the sponsor’s use of a joint curve model with log-normal distribution to predict RFS and OS data during the trial and after the trial. Upon visual inspection, the predicted RFS and OS data slightly overestimated the Kaplan-Meier (KM) curves observed in the QUAZAR AML-001 trial; this overestimate may reduce the ICER of oral azacitidine.
CADTH addressed this limitation by replacing a joint curve model with KM curves with tails in a scenario analysis. Prior to 65 months, OS and RFS data were based on the KM data from the QUAZAR AML-001 trial; data were then predicted from parametric survival models.
Exclusion of dose extension in the calculation of oral azacitidine cost: The sponsor’s model used RFS and OS data derived from the intention-to-treat analysis of the QUAZAR AML-001 trial, which considered dose adjustment (|||% for oral azacitidine group and |||% for placebo group); however, the model did not capture dose extension in the drug cost calculation. This likely overestimates the clinical benefit but underestimates the cost of oral azacitidine and the resulting ICER because more patients in the oral azacitidine group (21.4%) required dose extension compared to those receiving no active therapy (17.1%). This concern is supported by the sponsor’s exploratory analysis of a subset of patients who received a dose extension, suggesting a change in survival (OS hazard ratio, ||| versus |||) and relapse benefits (RFS hazard ratio, ||| versus |||) of oral azacitidine compared to the ITT analysis.
CADTH addresses this limitation by considering dose extension in drug cost calculations in its revised base case. The percentages of patients receiving dose extension and time spent on dose extension were based on the QUAZAR AML-001 trial. Dose reduction was not included because Canadian pharmacies are likely to dispense the entire quantity of oral medication for each treatment cycle.
Inappropriate assumption on the proportion of patients receiving HSCT: The sponsor assumed that 6.3% of patients treated with oral azacitidine and 13.7% of patients on no active therapy received HSCT. Clinical experts consulted by CADTH raised a concern about this assumption and suggested that the proportion of patients who received HSCT post-relapse was unlikely to depend on the type of initial treatments (oral azacitidine or no active therapy). Assigning the difference in post-relapse HSCT rates may underestimate ICER, given the smaller proportion of patients treated with oral azacitidine receiving HSCT compared to those receiving no active therapy.
CADTH addressed this limitation by assuming the same post-relapse HSCT rates for patients in the oral azacitidine and no active treatment groups in its scenario analysis.
An additional limitation was identified but was not considered to be key:
Underestimated end-of-life care cost: The sponsor obtained the costs of end-of-life care from a study conducted by Bekelman and colleagues, which estimated the Canadian terminal care expenditure from acute care stay, intensive care unit stay, emergency room visits, and chemotherapy administration incurred during the 30 days preceding death. These cost data are likely to underestimate the cost of end-of-life care as they exclude outpatient service costs, which account for 71.5% of the total cost of terminal cancer care patients.5 Low end-of-life care costs would increase the ICER because more patients with no active therapy transitioned to death compared to those treated with oral azacitidine.
CADTH addressed this limitation by using an alternative end-of-life cost5 that captures all publicly funded health services provided to terminal care patients in its reanalysis.
Key assumptions made by the sponsor and appraised by CADTH are provided in Table 4.
Table 4: Key Assumptions of the Submitted Economic Evaluation (Not Noted as Limitations to the Submission)
Sponsor’s key assumption | CADTH comment |
|---|---|
Patients who experienced relapse and required subsequent therapies were assumed to receive a single line of subsequent therapy after relapse. | The clinical experts consulted by CADTH disagreed with this assumption. It is possible that patients may require more than 1 line of subsequent treatments. For example, patients who experience relapse post-transplant are likely to receive another line of therapy. CADTH could not assess the impact of this limitation. This assumption is expected to have a minimal impact on the estimated ICER, given that the same type of subsequent therapies was applied to both treatment groups. |
Subsequent therapies could affect OS but not the health-related quality of life. | Acceptable. CADTH noted that the effect of subsequent therapies on patient survival would have been captured in the OS curves and that this effect might confound post-relapse survival observed in both treatment groups. Additionally, the impact of subsequent therapies on health-related quality of life could have been accounted for in a relapse-specific health utility. CADTH was unable to assess the effects of this assumption given the submitted model structure. |
Patients who received decitabine as a subsequent therapy in the QUAZAR AML-001 trial were assumed to receive low-dose cytarabine in the model. | Acceptable. The sponsor indicated that decitabine is not listed on Canadian public formularies and is rarely used in clinical practice for relapsed or refractory AML. |
HSCT was considered as a subsequent therapy. The model accounted for its impact on survival through OS curves and considered the cost and utility decrement associated with HSCT. | Acceptable. CADTH noted that the effect of HSCT might confound post-relapse survival observed in both treatment groups. CADTH was unable to assess the impact of this assumption given the submitted model structure. |
The model assumed a 100% compliance of oral azacitidine. | Acceptable. Canadian pharmacies are likely to dispense the full quantity of oral medications for each treatment cycle. |
The sponsor’s model obtained health utility values from a study conducted by Joshi et al. (2019)2 that estimated health utility values from AML patients from the general UK population using a composite time trade-off methodology. | Acceptable. CADTH was concerned that the health utility values generated from the UK population might not reflect the health utility of the general Canadian population. CADTH assessed the impact of this assumption by using an alternative data source for health utility values in a scenario analysis. |
Patients receiving oral azacitidine required a chemotherapy management fee and 5 days of ondansetron premedication. | Inappropriate. The clinical experts consulted by CADTH suggested that a chemotherapy management fee is not necessary for an oral oncology treatment, and for each treatment cycle patients will require ondansetron 8 mg once daily for each day of oral azacitidine, i.e., 14 days per cycle. CADTH removed the chemotherapy management fee and used a revised dosage regimen for ondansetron in its revised base case. |
AML = acute myeloid leukemia; BSC = best supportive care; HSCT = hematopoietic stem cell transplant; RFS = relapse-free survival; OS = overall survival.
CADTH Reanalyses of the Economic Evaluation
Base-Case Results
CADTH could not adequately address some limitations (i.e., inability to account for the impact of subsequent treatments, including HSCT, on post-relapse survival and OS) of the sponsor’s model due to the model structure. The CADTH base case was derived by considering dose extension in the calculation of oral azacitidine costs. Dose reduction was not included because Canadian pharmacies are likely to dispense the full quantity of oral medications for each treatment cycle. In addition, CADTH removed a management fee for oral chemotherapy and calculated the per-cycle cost of ondansetron based on 14 days of ondansetron 8 mg once daily.
Table 5 details the change made to derive the CADTH base case, and the summary results of the CADTH base case are presented in Table 6. Additional results are shown in Appendix 4 (Table 11). Results from the CADTH base case suggested that oral azacitidine was associated with higher costs ($322,869) and increased QALYs (0.91), for an ICER of $355,456 per QALY. The estimated ICER was slightly higher than that reported in the sponsor’s base case due to increased oral azacitidine costs when considering dose extension and the longer duration of ondansetron premedication. The probability that oral azacitidine is cost-effective was zero at the willingness to value of $50,000 per QALY. Approximately 39% of the QALY gains and 2% of incremental costs came from the extrapolation of outcomes beyond the trial follow-up period.
Table 5: CADTH Revisions to the Submitted Economic Evaluation
Stepped analysis | Sponsor’s value or assumption | CADTH value or assumption |
|---|---|---|
Correctionsa to sponsor’s base case | ||
None | None | None |
Changes to derive the CADTH base case | ||
1. The sponsor’s model did not account for dose extension in the cost calculation, but the effects of dose extension were captured in the RFS and OS estimates | Percentage of patients receiving dose extension = 0 Percentage of time spent on dose extension = 0 | Percentage of patients receiving dose extension = 21.7 Percentage of time spent on dose extension = 17.2 |
2. Chemotherapy management fee for an oral treatment | Patients receiving oral azacitidine required 1 chemotherapy management fee ($25.75) | $0 |
3. Oral ondansetron required before oral azacitidine | Patients receiving oral azacitidine required 5 days of ondansetron 8 mg, twice a day | Patients receiving oral azacitidine required 14 days of ondansetron 8 mg, taken once a day |
CADTH base case | — | 1 + 2 + 3 |
AML = acute myeloid leukemia; OS = overall survival; RFS = relapse-free survival.
Table 6: Summary of the Stepped Analysis of CADTH Reanalysis Results
Stepped analysis | Drug | Total costs ($) | Total QALYs | ICER ($ per QALY) |
|---|---|---|---|---|
Sponsor’s base case | No active therapy + BSC | 72,568 | 1.75 | Reference |
Oral azacitidine + BSC | 389,562 | 2.66 | 348,988 | |
CADTH reanalysis 1 | No active therapy + BSC | 72,568 | 1.75 | Reference |
Oral azacitidine + BSC | 395,526 | 2.66 | 355,554 | |
CADTH reanalysis 2 | No active therapy + BSC | 72,568 | 1.75 | Reference |
Oral azacitidine + BSC | 389,149 | 2.66 | 348,532 | |
CADTH reanalysis 3 | No active therapy + BSC | 72,568 | 1.75 | Reference |
Oral azacitidine + BSC | 391,005 | 2.66 | 350,576 | |
CADTH base case | No active therapy + BSC | 72,559 | 1.75 | Reference |
Oral azacitidine + BSC | 395,429 | 2.66 | 355,456 |
BSC = best supportive care; ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year.
Scenario Analysis Results
Based on the CADTH base case, scenario analyses were conducted. These analyses explored the impact of the following model parameters and assumptions: survival distributions to extrapolate RFS and OS data; health utility values; costs of end-of-life care; time on treatment duration; the proportion of patients receiving post-relapse HSCT; and a perspective of analysis.
Results from scenario analyses (Appendix 4, Table 11) demonstrated that the cost-effectiveness findings were sensitive to how RFS and OS data were incorporated into the economic model. In a scenario in which KM curves were used to estimate RFS and OS data during the trial period and parametric survival models were used to predict long-term RFS and OS data, the ICER increased from $355,456 to $543,359 per QALY. The ICERs were also influenced by health utility values and the treatment duration. Using alternative health utility values from Tremblay et al. (2020),6 which reported a lower utility value for the RFS state but a slightly higher utility value for the relapse state, increased the ICER to $384,118. As expected, assuming a similar proportion of patients receiving post-relapse HSCT for the groups receiving oral azacitidine and no active therapy and extending the duration of treatment for oral azacitidine increased the estimated ICERs. Cost-effectiveness findings were found to be robust to the changes in the costs of end-of-life care when considered from a societal perspective.
CADTH undertook a price reduction analysis based on the sponsor's base case and CADTH’s base case (Table 7). The results show that a price reduction of 85% is required for oral azacitidine to be considered cost-effective at a willingness-to-pay threshold of $50,000 per QALY. Given the uncertainty surrounding the existence of a post-relapse survival benefit, the price reduction is likely underestimated.
Table 7: CADTH Price Reduction Analyses
Price reduction analysis | ICERs for oral azacitidine + BSC vs. no active therapy + BSC ($ per QALY) | |
|---|---|---|
Sponsor base case | CADTH reanalysis | |
No price reduction | 348,988 | 355,456 |
10% | 313,681 | 319,493 |
20% | 278,375 | 283,530 |
30% | 243,068 | 247,567 |
40% | 207,762 | 211,604 |
50% | 172,456 | 175,641 |
60% | 137,149 | 139,678 |
70% | 101,843 | 103,715 |
80% | 66,537 | 67,752 |
85% | 48,883 | 49,771 |
BSC = best supportive care; ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year; vs. = versus.
Issues for Consideration
Subcutaneous azacitidine has been approved by Health Canada for adults with AML with 20% to 30% blasts and multi-lineage dysplasia who are not eligible for stem cell transplant. Although the subcutaneous formulation is less expensive than the oral formulation, clinical experts consulted by CADTH advised that the 2 formulations are not interchangeable; clinicians are unlikely to substitute oral azacitidine with subcutaneous azacitidine.
The oral route of administration may limit access to treatment and introduce financial barriers for patients in jurisdictions that do not fund oral medications through the same mechanisms as IV oncology regimens.
Drug plans participating in this review raised concerns that oral azacitidine may be used in patients with FLT3 mutation–positive AML who received midostaurin in combination with induction and/or consolidation therapy. Plans identified the potential for indication creep. The cost-effectiveness of oral azacitidine in this population is unknown.
Overall Conclusions
Evidence from the CADTH Clinical Review and the QUAZAR AML-001 trial results suggested that oral azacitidine led to significant improvement in RFS and OS among patients with AML who were in remission after chemotherapy. Survival models used to estimate the RFS and OS drove the incremental difference in QALYs, while the time on oral azacitidine was the key driver of the incremental costs between oral azacitidine and no active therapy.
CADTH identified several limitations within the sponsor’s submission: the lack of clinical evidence supporting post-relapse survival benefits of oral azacitidine shown in the economic analysis; the lack of fit of survival models used to estimate RFS and OS data; the exclusion of dose extension from the calculation of oral azacitidine costs; and the assumption on the rates of post-relapse HSCT. CADTH attempted to address these limitations by performing the following reanalyses: considering dose extension in the calculation of oral azacitidine costs, removing a management fee for oral chemotherapy, using an alternative regimen for ondansetron premedication, using alternate survival distributions to extrapolate RFS and OS data, applying alternate health utility values, assigning alternative end-of-life care costs, varying of time on treatment duration, changing the proportion of patients receiving post-relapse HSCT, and adopting a societal perspective. Both the sponsor’s and CADTH’s base cases suggested that oral azacitidine was associated with higher costs and improved QALYs but was not cost-effective compared to no active therapy. At the submitted price, the probability that oral azacitidine is cost-effective was 0%. A price reduction of at least 85% would be required to make oral azacitidine a cost-effective option at a willingness-to-pay threshold of $50,000 per QALY, although this is likely an underestimate.
The cost-effectiveness of oral azacitidine was highly sensitive to statistical approaches used to fit RFS and OS data. If the KM curves used to reflect RFS and OS data during the trial period and survival models were used to predict the long-term RFS and OS data, the ICER would increase substantially from $355,456 to $543,359 per QALY. Other key drivers included health utility values and the treatment duration. Using a lower utility value for the RFS state but a slightly higher utility value for the relapse state increased the ICER from $355,456 to $384,118 per QALY. Moreover, if the type of maintenance therapy in the first remission was assumed to have no impact on the proportion of patients receiving post-relapse HCST, the ICER would rise to $366,705 per QALY. Cost-effectiveness findings were robust to the changes in the cost of end-of-life care and the use of a societal perspective.
The CADTH estimates of cost-effectiveness are likely biased in favour of oral azacitidine, as the reanalysis was unable to address the critical limitation regarding the unproven post-relapse survival benefit due to constraints introduced by the submitted model structure. As such, oral azacitidine is unlikely to be cost-effective at a conventional threshold without a large reduction in price.
References
1.Wei AH, Döhner H, Pocock C, et al. Oral azacitidine maintenance therapy for acute myeloid leukemia in first remission. N Engl J Med. 2020;383(26):2526-2537. PubMed
2.Joshi N, Hensen M, Patel S, Xu W, Lasch K, Stolk E. Health state utilities for acute myeloid leukaemia: a time trade-off study. Pharmacoeconomics. 2019;37(1):85-92. PubMed
3.Bekelman JE, Halpern SD, Blankart CR, et al. Comparison of site of death, health care utilization, and hospital expenditures for patients dying with cancer in 7 developed countries. JAMA. 2016;315(3):272-283. PubMed
4.Pharmacoeconomic evaluation [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Onureg (azacitidine): 200 mg, 300 mg tablets. St. Laurent (QC): Celgene; 2021.
5.Yu M, Guerriere DN, Coyte PC. Societal costs of home and hospital end-of-life care for palliative care patients in Ontario, Canada. Health Soc Care Community. 2015;23(6):605-618. PubMed
6.Tremblay G, Cariou C, Recher C, et al. Cost-effectiveness of midostaurin in the treatment of newly diagnosed FLT3-mutated acute myeloid leukemia in France. Eur J Health Econ. 2020;21(4):543-555. PubMed
7.PrOnureg®(azacitidine): 200 mg, 300 mg oral tablets [product monograph]. St. Laurent (QC): Celgene; 2021 Jan 4.
8.Budget Impact Analysis [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Onureg (azacitidine): 200 mg, 300 mg tablets. St. Laurent (QC): Celgene; 2021.
9.Chronic myelogenous leukemia statistics. Toronto (ON): Canadian Cancer Society: https://www.cancer.ca/en/cancer-information/cancer-type/leukemia-chronic-myelogenous-cml/statistics/?region=on#:~:text=Incidence%20and%20mortality&text=The%20most%20recent%20incidence%20statistics,diagnosed%20with%20chronic%20myelogenous%20leukemia. Accessed 2021 May 28.
Appendix 1: Cost Comparison Table
Note this appendix has not been copy-edited.
The comparators presented in Table 8 have been deemed to be appropriate based on feedback from clinical expert(s) and drug plans. Comparators may be recommended (appropriate) practice or actual practice. Existing product listing agreements are not reflected in the table and as such, the table may not represent the actual costs to public drug plans.
Table 8: CADTH Cost Comparison Table for Acute Myeloid Leukemia
Treatment | Strength or concentration | Form | Price | Recommended dosage | Cost per 28-day cycle | Average annual cost |
|---|---|---|---|---|---|---|
Azacitidine tablets (Onureg) | 200 mg | Tablet | 952.0000 per tableta | 300 mg daily, Days 1 through Day 14 b | 19,992 | 260,610 |
300 mg | 1,428.0000 per tableta |
aSponsor-submitted price.
bEvery 28 days as per oral azacitidine product monograph.7
Appendix 2: Submission Quality
Note this appendix has not been copy-edited.
Description | Yes/No | Commentsa |
|---|---|---|
Population is relevant, with no critical intervention missing, and no relevant outcome missing. | Yes | Baseline characteristics of patients in the QUAZAR AML-001 trial are generalizable to Canadian patients with AML. |
Model has been adequately programmed and has sufficient face validity. | Yes | The submitted model is transparent and has no programmatic errors. |
Model structure is adequate for decision problem. | Yes | Acceptable. A partitioned survival model is commonly used in oncology submissions; however, the model structure may produce a post-relapse survival bias in favour of oral azacitidine. |
Data incorporation into the model has been done adequately (e.g., parameters for probabilistic analysis). | No | See CADTH appraisal section. |
Parameter and structural uncertainty were adequately assessed; analyses were adequate to inform the decision problem. | Yes | Acceptable. |
The submission was well organized and complete; the information was easy to locate (clear and transparent reporting; technical documentation available in enough details). | Yes | The economic report is well organized and easy to navigate. |
AML = acute myeloid leukemia.
Appendix 3: Additional Information on the Submitted Economic Evaluation
Note this appendix has not been copy-edited.
Figure 2: Observed and Predicted Overall Survival Data — Oral Azacitidine
Source: Sponsor’s pharmacoeconomic submission.4
Figure 3: Observed and Predicted Overall Survival Data — No Active Therapy
Source: Sponsor’s pharmacoeconomic submission.4
Figure 4: Observed and Predicted Relapse-Free Survival — Oral Azacitidine
Source: Sponsor’s pharmacoeconomic submission.4
Figure 5: Observed and Predicted Relapse-Free Survival — No Active Therapy
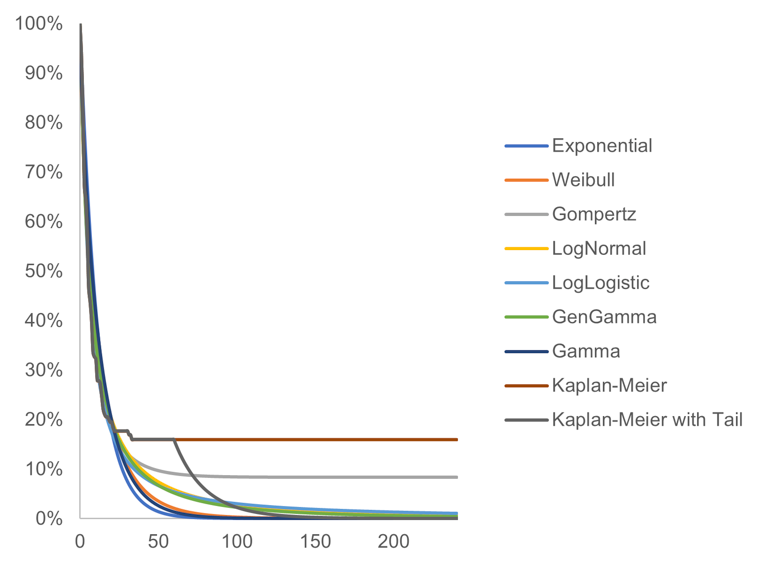
Source: Sponsor’s pharmacoeconomic submission.4
Appendix 4: Additional Details on the CADTH Reanalyses and Sensitivity Analyses of the Economic Evaluation
Note this appendix has not been copy-edited.
Detailed Results of CADTH Base Case
Table 10: Disaggregated Summary of CADTH’s Economic Evaluation Results
Parameter | Oral azacitidine + BSC | No active therapy + BSC | Incremental |
|---|---|---|---|
Discounted LYs | |||
Total | 3.57 | 2.47 | 1.11 |
By health state | |||
RFS: on treatment | 0.86 | 0.00 | 0.86 |
RFS: off treatment | 1.36 | 1.32 | 0.04 |
Relapse | 1.35 | 1.15 | 0.20 |
Discounted QALYs | |||
Total | 2.66 | 1.75 | 0.91 |
By health state | |||
RFS: on treatment | 0.77 | 0.00 | 0.77 |
RFS: off treatment | 1.21 | 1.17 | 0.04 |
Relapse | 0.69 | 0.58 | 0.10 |
AE | -0.008 | -0.006 | 0.00 |
HSCT | -0.001 | -0.002 | 0.00 |
Discounted costs ($) | |||
Total | 395,429 | 72,559 | 322,869 |
Acquisition | 326,661 | 0 | 326,661 |
Administration | 1,222 | 0 | 1,222 |
Disease management | 41,042 | 34,541 | 6,501 |
AEs | 1,020 | 708 | 312 |
Subsequent therapy | 2,848 | 4,572 | -1,725 |
HSCT | 8,318 | 18,108 | -9,789 |
End-of-life care | 10,615 | 10,941 | -326 |
ICER ($ per QALY) | 355,456 | ||
AE = adverse event; BSC = best supportive care; HSCT = hematopoietic stem cell transplant; ICER = incremental cost-effectiveness ratio; LY = life-year; QALY = quality-adjusted life-year; RFS = relapse-free survival.
Scenario Analyses
Based on CADTH’s base case, a series of scenario analyses were conducted (Table 11).
Table 11: Summary of CADTH Scenario Analyses
Drug | Total costs ($) | Total QALYs | ICERs ($ per QALY) |
|---|---|---|---|
Sponsor’s base case | |||
No active therapy + BSC | 72,568 | 1.75 | Reference |
Oral azacitidine + BSC | 389,562 | 2.66 | 348,988 |
CADTH’s base case | |||
No active therapy + BSC | 72,559 | 1.75 | Reference |
Oral azacitidine + BSC | 395,429 | 2.66 | 355,456 |
CADTH’s scenario analyses | |||
CADTH's scenario analysis 1: Alternative parametric survival models (KM OS curves with log-normal tail) | |||
No active therapy + BSC | 83,225 | 1.91 | Reference |
Oral azacitidine + BSC | 394,588 | 2.59 | 458,956 |
CADTH's scenario analysis 2: Alternative parametric survival models (KM RFS curves with generalized Gamma tail for oral azacitidine and Weibull tail for no active therapy) | |||
No active therapy + BSC | 70,351 | 1.80 | Reference |
Oral azacitidine + BSC | 399,230 | 2.63 | 400,275 |
CADTH's scenario analysis 3: Alternative parametric survival models (KM RFS and OS curves with tails) | |||
No active therapy + BSC | 79,559 | 1.98 | Reference |
Oral azacitidine + BSC | 397,234 | 2.56 | 543,359 |
CADTH's scenario analysis 4: Alternative parametric survival models (KM OS curves with generalized Gamma tail) | |||
No active therapy + BSC | 92,385 | 2.10 | Reference |
Oral azacitidine + BSC | 394,192 | 2.57 | 632,042 |
CADTH's scenario analysis 5: Alternative parametric survival models (KM RFS curves with log logistic tail) | |||
No active therapy + BSC | 73,803 | 1.73 | Reference |
Oral azacitidine + BSC | 397,087 | 2.63 | 360,261 |
CADTH's scenario analysis 6: Assuming the same proportion of post-relapse HSCT for oral azacitidine and no active therapy groups | |||
No active therapy + BSC | 62,778 | 1.75 | Reference |
Oral azacitidine + BSC | 395,429 | 2.66 | 366,705 |
CADTH's scenario analysis 7: Alternative health utility values (based on Tremblay et al. [2020])6 | |||
No active therapy + BSC | 72,559 | 1.69 | Reference |
Oral azacitidine + BSC | 395,429 | 2.53 | 384,118 |
CADTH's scenario analysis 8: Reducing time on treatment for oral azacitidine | |||
No active therapy + BSC | 72,559 | 1.75 | Reference |
Oral azacitidine + BSC | 286,204 | 2.66 | 235,208 |
CADTH's scenario analysis 9: Increasing time on treatment for oral azacitidine | |||
No active therapy + BSC | 72,559 | 1.75 | Reference |
Oral azacitidine + BSC | 483,281 | 2.66 | 452,175 |
CADTH's scenario analysis 10: Using alternative end-of-life care costs | |||
No active therapy + BSC | 78,131 | 1.75 | Reference |
Oral azacitidine + BSC | 400,835 | 2.66 | 355,273 |
CADTH's scenario analysis 11: Considering both dose extension and dose reduction for the calculation of oral azacitidine cost | |||
No active therapy + BSC | 72,559 | 1.75 | Reference |
Oral azacitidine + BSC | 393,460 | 2.66 | 353,289 |
CADTH's scenario analysis 12: Adopting a societal perspective | |||
No active therapy + BSC | 77,433 | 1.75 | Reference |
Oral azacitidine + BSC | 401,855 | 2.66 | 357,165 |
BSC = best supportive care; ICER = incremental cost-effectiveness ratio; KM = Kaplan-Meier; OS = overall survival; QALY = quality-adjusted life-year; RFS = relapse-free survival.
Appendix 5: Submitted Budget Impact Analysis and CADTH Appraisal
Note this appendix has not been copy-edited.
Table 12: Summary of Key Take-Aways
Key Take-Aways of the Budget Impact Analysis |
|---|
|
Summary of Sponsor’s Budget Impact Analysis
The sponsor assessed the budget impact of the introduction of oral azacitidine for patients with AML who achieve CR or CRi following induction therapy, with or without consolidation treatment, and who are not eligible for HSCT, from the drug plan perspective in the Canadian setting (excluding Quebec), over a 3-year time horizon.8 The sponsor only included drug acquisition costs. In the reference scenario, the sponsor assumed that patients would no active therapy (i.e., BSC), which was associated with no costs. In the new drug scenario, oral azacitidine was assumed to displace market shares of BSC.8
The sponsor estimated the eligible population size using an epidemiological approach, by leveraging data from the QUAZAR trial, multiple sources in the literature, and assumptions based on clinical expert input.8
Key inputs to the Budget Impact Analysis are documented in Table 13.
Figure 6: Sponsor’s Estimation of the Size of the Eligible Population
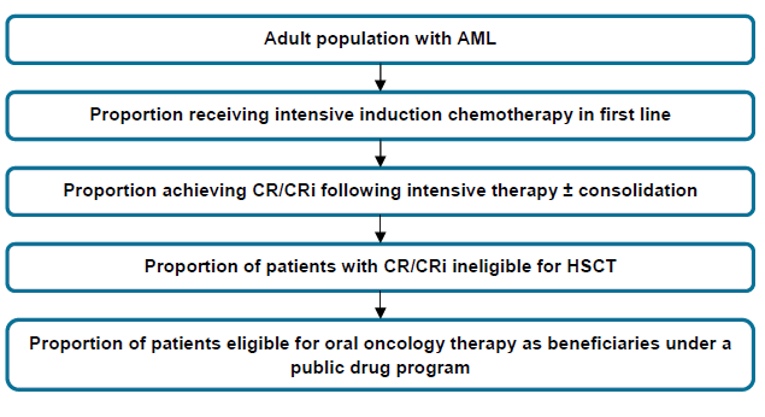
AML = acute myeloid leukemia; CR = complete remission; CRi= complete remission with incomplete blood count recovery; HSCT = hematopoietic stem cell transplant.
Source: Sponsor’s submitted budget impact analysis report.8
Table 13: Summary of Key Model Parameters
Parameter | Sponsor’s estimate (reported as year 1 / year 2 / year 3 if appropriate) |
|---|---|
Target population | |
Adult population with AML | |
Proportion receiving intensive induction chemotherapy in first line | 48% |
Proportion achieving CR/CRi following intensive therapy ± consolidation | 89% |
Proportion of patients with CR/CRi ineligible for HSCT | 46% |
Proportion of patients eligible for oral oncology therapy as beneficiaries under a public drug program | 100% |
Number of patients eligible for drug under review | 206 / 214 / 222 |
Market uptake (3 years) | |
Uptake (reference scenario) No active therapya | 100% / 100% / 100% |
Uptake (new drug scenario) Oral azacitidine No active therapya | 27% / 40% / 47% 73% / 60% / 53% |
Cost of treatment (per patient) | |
Cost of treatment, per year Oral azacitidine No active therapya | $251,899 $0 |
BSC = best supportive care; CR = complete remission; CRi = complete remission with incomplete blood count recovery; HSCT = hematopoietic stem cell transplant.
aNo active therapy represents patients receiving only BSC, and BSC is assumed to be utilized the same between those in no active therapy and in the oral azacitidine group.
Summary of the Sponsor’s Budget Impact Analysis Results
Results of the sponsor’s base-case analysis reflected the drug program perspective and revealed that the introduction of oral azacitidine in patients with AML who achieved CR or CRi following induction therapy with or without consolidation treatment, and who are not eligible for hematopoietic stem cell transplant, would result in an incremental budget impact of $14,713,983 in year 1, $22,931,743 in year 2, $27,797,095 in year 3, for a total budget impact of $65,442,821 over the 3-year time horizon.8
CADTH Appraisal of the Sponsor’s Budget Impact Analysis
CADTH identified several key limitations to the sponsor’s analysis that have notable implications on the results of the Budget Impact Analysis:
Uncertainty in the estimated eligible population size: The sponsor undertook an epidemiological approach to estimate the size of the population eligible for oral azacitidine which resulted in a total number of 215, 223 and 232 patients in Years 1, 2, and 3, respectively. The clinical experts consulted by CADTH indicated that while these numbers appeared to be reasonable, they noted several areas of uncertainty with the estimates and assumptions used to derive the eligible population. CADTH’ clinical experts noted uncertainty with the identified total incident AML population in the baseline year (i.e., 1,040 newly diagnosed patients in 2021), which was based on an incidence rate of 0.0044% for AML. The experts stated that this total appeared to be lower than they expected, and they anticipated that this total was likely 3 times the number of newly diagnosed patients with chronic myelogenous leukemia (CML). Among the patients newly diagnosed, the sponsor assumed that 48% would receive intensive induction chemotherapy in the first-line setting, however, 2 of CADTH’s clinical experts noted that this estimate likely varied across provinces while 1 expert felt this proportion was higher (approximately 70%). Among the proportion of patients who had achieved CR/CRi, the sponsor assumed 46% would be ineligible for stem cell transplant. However, CADTH’s clinical experts stated that this appeared to be an overestimate; they noted that a lower proportion of patients were ineligible for transplant (30%). The sponsor further assumed that full public coverage for oral therapies would be offered to the entire indicated population, across all provinces. CADTH’s clinical experts indicated that this was also an overestimate due to the fact that the funding criteria for oral therapy differed across provinces, and therefore not all patients would receive full coverage. Lastly, the sponsor used an incident approach and did not consider prevalence statistics as part of their methodology to estimate the market size, which would include the proportion of patients who are currently being treated for the condition and eligible for the treatment.
CADTH addressed this limitation by changing total number of incident AML patients to 3 times the number of newly diagnosed patients with CML based on a Canadian estimate of newly diagnosed patients with CML in 2016. As 600 newly diagnosed patients with CML were reported in 2016,9 this resulted in a total of 1,800 newly diagnosed AML patients which were included in the CADTH base case. CADTH further changed the proportion of patients ineligible for stem cell transplant to 30% in the base case. To assess the uncertainty with other epidemiological inputs used to derive the total eligible population, CADTH conducted the following scenario analyses to assess the impact of these inputs on the budget impact : increased the total incident AML population by 25%; increased the total incident AML population by 50%; increased the proportion of patients eligible to receive intensive induction chemotherapy in the first-line setting to 70%; and arbitrarily assumed that fewer patients (70%) would be able to receive full public coverage for oral therapies across Canada.
Median treatment time on treatment may underestimate drug costs associated with oral azacitidine: In the submitted pharmacoeconomic analysis, the sponsor adjusted total drug acquisition costs for oral azacitidine based on the mean time on treatment (i.e., | cycles). However, in the submitted BIA, the sponsor adjusted total drug costs based on the median time on treatment (i.e., 12.6 treatment cycles), which underestimated the total drug costs associated with oral azacitidine. To align with CADTH’s pharmacoeconomic base case, the treatment duration was changed ||||||||||||||||||||||||.
CADTH addressed this limitation by changing the time on treatment with oral azacitidine ||||||||||||||||||||||||||||||||||||. In a scenario analysis, CADTH examined the impact of the median time on treatment with oral azacitidine (12.6 treatment cycles).
Underestimation of drug costs for oral azacitidine due to exclusion of dose extension: In the submitted BIA, the sponsor did not consider dose extension as part of the total drug acquisition costs for oral azacitidine. In the QUAZAR trial, 21.7% of patients who received oral azacitidine received an extended dose. While the product monograph for oral azacitidine does not list any specifications for administering an extended dose among the indicated population, CADTH’s clinical experts affirmed that dose extension is relevant to Canadian clinical practice and that the proportion of patients who received an extended dose of oral azacitidine in the trial reflected their expectations of those who would receive an extended dose in clinical practice. Additionally, the sponsor assumed that if dose extension were applicable, 21.6% of patients would receive an extended dose for a median duration of | treatment cycles. However, to align with the CADTH pharmacoeconomic analysis base case, the proportion of patients who received an extended dose extension was revised to 21.7%, and a duration of | treatment cycles was assumed for patients who received an extended dose.
CADTH addressed this limitation by permitting dose extension to the proportion of patients who received an extended dose in the QUAZAR trial (21.7%) ||||||||||||||||||||||||||||||||||||||||||||||||||||||||.
Exclusion of relevant costs under the drug program perspective underestimated total expenditure associated with oral azacitidine: The sponsor only considered drug acquisition costs and excluded several other costs covered by public drug programs, which underestimated the total expenditure associated with the introduction of oral azacitidine. Specifically, the sponsor excluded pharmacy fees, costs of premedication and concomitant medications, outpatient drug costs to treat AEs, costs of subsequent therapy and costs of hematopoietic stem cell transplant, which are relevant under a public drug program perspective.
CADTH addressed this limitation by incorporating the aforementioned relevant costs under the drug plan perspective.
Proportion of patients receiving hematopoietic stem cell transplant (HSCT) is uncertain: In the submitted BIA, the sponsor assumed the proportion of patients who would receive HSCT in the treatment and comparator group aligned with the proportion of patients who received HSCT in the QUAZAR trial (6.3% and 13.7%, respectively). The clinical experts consulted by CADTH indicated that proportion of patients who actually receive HSCT is dependent on donor availability, and that this proportion is typically higher than that reported in the trial, which appeared to be lower than expected for the oral azacitidine group. Further, the experts noted that the same proportion of patients from both groups would be expected to receive HSCT.
CADTH addressed the limitation by assuming the same proportion of patients from both groups would receive HSCT based on the proportion in the oral azacitidine group (6.3%) to align with CADTH’s pharmacoeconomic analysis.
CADTH Reanalyses of the BIA
A table noting the changes made to the sponsor’s BIA as part of the CADTH reanalysis is available in Table 14.
Table 14: CADTH Revisions to the Submitted Budget Impact Analysis
Stepped analysis | Sponsor’s value or assumption | CADTH value or assumption |
|---|---|---|
Corrections to sponsor’s base case | ||
None | None | None |
Changes to derive the CADTH base case | ||
1. Approach to derive market size | Starting AML patient population in baseline year = 1,040 newly diagnosed patients Proportion of patients in CR or CRi who are ineligible for stem cell transplant = 46% | Starting AML patient population in baseline year = 1,800 newly diagnosed patients Proportion of patients in CR or CRi who are ineligible for stem cell transplant = 30% |
2. Treatment duration | Median time on treatment = 12.60 treatment cycles | Time on treatment = 19.68 treatment cycles |
3. Dose extension for oral azacitidine (and number of cycles on extended dose) | Dose extension excluded Proportion of patients who received a dose extension = 21.6% | Dose extension permitted, with 3.4 cycles on extended dose. Proportion of patients who received a dose extension revised to 21.7% |
4. Relevant costs under the drug program perspective (i.e., premedications, concomitant medications, adverse event drugs, pharmacy fees, subsequent therapy and HSCT) | Excluded | Included |
5. Proportion of patients receiving hematopoietic stem cell transplant (HSCT) | Oral azacitidine = 6.3% No active therapy = 13.7% | Oral azacitidine = 6.3% No active therapy = 6.3% |
CADTH base case | Reanalysis 1 + 2 + 3 | |
HSCT = hematopoietic stem cell transplant.
Applying these changes in Table 17 resulted in an increase in the budget impact under the drug plan perspective to $100,647,777 over 3 years. The results of the CADTH stepwise reanalyses are presented in summary format in Table 15, and a more detailed breakdown is presented in Table 16.
Table 15: Summary of the CADTH Reanalyses of the Budget Impact Analysis
Stepped analysis | Three-year total |
|---|---|
Submitted base case | $64,231,597 |
CADTH reanalysis 1 | $72,241,880 |
CADTH reanalysis 2 | $85,859,516 |
CADTH reanalysis 3 | 66,112,155 |
CADTH reanalysis 4 | $65,278,356 |
CADTH reanalysis 5 | $64,231,597 |
CADTH base case | $100,647,777 |
CADTH also conducted additional scenario analyses to address the remaining uncertainty regarding the potential size of the eligible population:
Explored the impact of an (a) 25% increase and (b) 50% increase in the total number of newly diagnosed patients with AML based on the sponsor’s estimated 1,040 total incident AML cases.
Increased the proportion of patients eligible to receive intensive induction chemotherapy in the first-line setting to 70%.
Applied the arbitrary assumption that fewer patients (70%) would be able to receive full public coverage for oral therapies across Canada rather than all patients.
Applied the median time on treatment (i.e., 12.6 treatment cycles) to calculate drug costs of oral azacitidine.
Applied an 85% price reduction on the submitted price of oral azacitidine to reflect the point at which the ICER was cost-effective within the $50,000 per QALY threshold.
Table 16: Detailed Breakdown of the CADTH Reanalyses of the Budget Impact Analysis
Stepped analysis | Scenario | Year 0 (current situation) | Year 1 | Year 2 | Year 3 | Three-year total |
|---|---|---|---|---|---|---|
Submitted base case | Reference | 0 | 0 | 0 | 0 | 0 |
New drug | 0 | 14,441,655 | 22,507,319 | 27,282,622 | 64,231,597 | |
Budget impact | 0 | 14,441,655 | 22,507,319 | 27,282,622 | 64,231,597 | |
CADTH base case | Reference | 5,673,352 | 5,894,613 | 6,124,503 | 6,363,359 | 18,382,475 |
New drug | 5,673,352 | 22,993,278 | 42,387,273 | 53,649,701 | 119,030,252 | |
Budget impact | 0 | 17,098,665 | 36,262,769 | 47,286,342 | 100,647,777 | |
CADTH scenario analysis 1a | Reference | 5,673,352 | 7,368,266 | 7,655,629 | 7,954,198 | 22,978,094 |
New drug | 5,673,352 | 28,739,710 | 52,980,133 | 67,056,977 | 148,776,820 | |
Budget impact | 0 | 21,371,444 | 45,324,504 | 59,102,778 | 125,798,726 | |
CADTH scenario analysis 1b | Reference | 5,673,352 | 8,841,920 | 9,186,755 | 9,545,038 | 27,573,712 |
New drug | 5,673,352 | 34,487,652 | 63,576,159 | 80,468,372 | 178,532,184 | |
Budget impact | 0 | 25,645,732 | 54,389,405 | 70,923,334 | 150,958,471 | |
CADTH scenario analysis 2 | Reference | 13,237,822 | 13,754,097 | 14,290,507 | 14,847,837 | 42,892,442 |
New drug | 13,237,822 | 53,650,982 | 98,903,636 | 125,182,636 | 277,737,254 | |
Budget impact | 0 | 39,896,885 | 84,613,129 | 110,334,799 | 234,844,812 | |
CADTH scenario analysis 3 | Reference | 3,971,347 | 4,126,229 | 4,287,152 | 4,454,351 | 12,867,732 |
New drug | 3,971,347 | 16,095,295 | 29,671,091 | 37,554,791 | 83,321,176 | |
Budget impact | 0 | 11,969,065 | 25,383,939 | 33,100,440 | 70,453,444 | |
CADTH scenario 4 | Reference | 5,673,352 | 5,894,613 | 6,124,503 | 6,363,359 | 18,382,475 |
New drug | 5,673,352 | 22,906,061 | 32,636,845 | 38,500,736 | 94,043,642 | |
Budget impact | 0 | 17,011,448 | 26,512,342 | 32,137,377 | 75,661,167 | |
CADTH scenario 5 | Reference | 5,673,352 | 5,894,613 | 6,124,503 | 6,363,359 | 18,382,475 |
New drug | 5,673,352 | 7,857,127 | 10,647,347 | 12,352,923 | 30,857,397 | |
Budget impact | 0 | 1,962,514 | 4,522,844 | 5,989,564 | 12,474,922 |
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up-to-date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.