CADTH Health Technology Review
Biologics in Plaque Psoriasis
Environmental Scan
Authors: Peter Dyrda, Sirjana Pant
Abbreviations
BSA
body surface area
CAF
Canadian Armed Forces
CDEC
CADTH Canadian Drug Expert Committee
CSC
Correctional Services Canada
DLQI
Dermatology Life Quality Index
ES
Environmental Scan
FPT
federal, provincial, and territorial
IL
interleukin
LOI
letter of intent
MTX
methotrexate
NIHB
Non-Insured Health Benefits
NOC
Notice of Compliance
ODB
Ontario Drug Benefit
PASI
Psoriasis Area and Severity Index
pCPA
pan-Canadian Pharmaceutical Alliance
PLA
product listing agreement
PsO
plaque psoriasis
TNF
tumour necrosis factor
VAC
Veterans Affairs Canada
Key Messages
Health Canada has approved 11 biologics for the treatment of adults with moderate-to-severe plaque psoriasis (PsO). These biologics can be divided into 2 groups based on mechanisms of action and market authorization dates:
Old-generation biologics (5): include anti–tumour necrosis factor (TNF) agents (etanercept, adalimumab, infliximab, and certolizumab pegol) and an anti-interleukin (IL)-12/IL-23 inhibitor (ustekinumab) which were approved in Canada before 2010.
New-generation biologics (6): include anti-IL-17 inhibitors (secukinumab, ixekizumab, and brodalumab) and anti-IL-23 inhibitors (guselkumab, tildrakizumab, and risankizumab) which were approved in Canada in 2015 or later.
Patent protection has expired for infliximab, certolizumab, and ustekinumab. There is no valid data protection status for all 5 older generation biologics. However, only 3 of the old-generation biologics have biosimilar versions (adalimumab, etanercept, and infliximab) that are available in the Canadian market.
Biosimilar versions for adalimumab and etanercept were marketed for PsO approximately 3 years to 4 years after their initial Notice of Compliance was issued, respectively. This delay can be attributed to various factors, including litigation and global agreements between manufacturers.
Despite the expiry of data and patent protection for both ustekinumab and certolizumab, no biosimilar versions are available in Canada. Data protection for both biologics expired more than 4 years ago, and the patents have expired in 2021. A lack of a biosimilar entrant in Canada could be attributed to various factors, including clinical trial development based on exclusivity timelines in the US, where data protection is 4 years longer for biologics versus Canada.
CADTH has reviewed 3 of 5 of the old-generation biologics and all 6 of the new-generation biologics; all drugs received similar CADTH Canadian Drug Expert Committee (CDEC) recommendations. The clinical programs of most of the new-generation biologics included direct evidence demonstrating superiority or statistically significantly higher efficacy outcomes compared with the active comparator of the old-generation biologics. The clinical trials for new-generation biologics also incorporated more stringent primary outcome measures.
The old-generation biologics predated the pan-Canadian Pharmaceutical Alliance (pCPA) process (except for certolizumab), which could imply disparate product listing agreements across public drug plans for these drugs. The only biologics not included on any public formularies are certolizumab, guselkumab, and tildrakizumab (although tildrakizumab has yet to begin pCPA negotiations). Listing status for biologics fell under restricted benefit, but differed in terms of active (e.g., review through special authorization forms) versus passive (e.g., Limited Use codes in Ontario) procedures across public drug plans. Moreover, 3 public drug plans employed 2-tiered formularies (i.e., Alberta, Manitoba, and Correctional Services Canada) which required a trial of new-generation biologics or old-generation biosimilars before reimbursement of old-generation originators.
Utilization patterns of old- versus new-generation biologics within the Ontario Public Drug Programs demonstrated that a significant proportion of new patients were treated with old-generation biologics (54% in 2019 and 37% in 2020) despite the availability of multiple new-generation biologics.
In conclusion, formulary management is warranted for biologics for PsO given the significant utilization of old-generation originators in the current context of delayed marketing of their biosimilar versions for PsO and their reimbursement predating the pCPA process. New-generation biologics underwent pCPA negotiations and direct evidence was submitted that demonstrated superiority versus old-generation biologic active comparators, which may ultimately prove to be a greater value for patients and payers.
Executive Summary
Background: Plaque psoriasis (PsO) is a prevalent chronic inflammatory condition associated with lowered quality of life. Moderate-to-severe PsO that does not respond to first-line treatment is treated with biologic medications, which can be divided into 2 groups based on market authorization dates and mechanisms of action: old-generation biologics (etanercept, adalimumab, infliximab, certolizumab, and ustekinumab) and new-generation biologics (secukinumab, ixekizumab, brodalumab, guselkumab, tildrakizumab, and risankizumab).
Policy issue: Given the significant expenditures on biologics and changing dynamics with regards to loss of exclusivity and new-generation biologic entrants, there is a need to assess the current state of reimbursement of biologics used to treat adults with moderate-to-severe PsO.
Objectives: To assess the regulatory, exclusivity, CADTH review, and reimbursement status of biologics (originators and biosimilars) used for PsO that are relevant to Canadian federal, provincial, and territorial (FPT) public drug plans, and assess the difference in utilization patterns between old- and new-generation biologics for a sample of new patients initiating biologic therapy from 1 public drug plan.
Approach: A grey literature search was conducted on key resources, including websites of Health Canada’s drug product database, patent register, and data protection register; CADTH website; Canadian public drug plan formulary databases; and the Ontario public drug plan utilization database, to inform the Environmental Scan (ES).
Findings: Health Canada has approved 11 biologics for PsO (5 old generation and 6 new generation). Of the 5 old-generation drugs, only 2 have registered patents (i.e., adalimumab and etanercept), and none have valid data protection status. Despite this exclusivity status, there are only 3 biosimilar versions available for old-generation biologics (adalimumab, infliximab, and etanercept), and these were marketed for PsO years after the initial market authorization. Furthermore, despite the expiry of data and patent protection for both ustekinumab and certolizumab, no biosimilar versions are available in Canada. All biologics received comparable CADTH Canadian Drug Expert Committee (CDEC) recommendations; however, clinical programs of new-generation biologics included direct evidence demonstrating superiority or statistically significantly higher efficacy outcomes compared with the active comparator of old-generation biologics. The pan-Canadian Pharmaceutical Alliance (pCPA) process has resulted in letters of intent (LOIs) for biologics for PsO except for guselkumab, certolizumab, and tildrakizumab. However, 4 of 5 old-generation biologics predated pCPA, thus PLAs across public drug plans may be disparate for these drugs. Biologics are reimbursed diversely across public drug plans, with active versus passive forms of restricted benefit status and within a tiered formulary in some cases (i.e., Alberta, Manitoba, and CSC). Utilization patterns of old- versus new-generation biologics within the Ontario Public Drug Programs demonstrated that a significant proportion of new patients were treated with old-generation biologics (54.4% in 2019 and 36.8% in 2020).
Implications for policy-makers: Significant utilization remains for old-generation originators despite the availability of new-generation biologics which have direct comparative evidence to support superiority. These old-generation originators also largely predated the pCPA process. Therefore, it is possible that payers have not had optimal value for biologics for PsO because old-generation originators have been reimbursed beyond their loss of exclusivity while also having a lower likelihood of product listing agreements (PLAs) compared with new-generation biologics. To optimize value in this class, formulary management strategies could be employed, such as pursuing pCPA negotiations for old-generation originators that do not have biosimilar versions and/or establishing tiered formularies to prioritize the reimbursement of old-generation biosimilars and new-generation originators.
Disease and Treatments
Plaque psoriasis (PsO) is a chronic inflammatory condition in which inflamed scaly patches of skin develop on the body which are associated with itching, redness, pain, discomfort, and lowered quality of life. PsO can develop on any part of the body, but most often occurs on the elbows, knees, scalp, and trunk. PsO is recognized as a multisystemic disease because it is associated with serious comorbidities including psoriatic arthritis, cardiovascular disease, metabolic syndrome, obesity, inflammatory bowel disease, and depression. PsO is the most common form of psoriasis, affecting approximately 90% of patients. It is estimated that 1% to 3% of people in Canada are affected by psoriasis.1-4
Disease severity, relevant comorbidities, patient preference (including cost and convenience), efficacy, and evaluation of individual patient response determine the type of treatment.1,3 Tools used to evaluate the severity and extent of psoriasis and associated impact on quality of life include Psoriasis Area and Severity Index (PASI), body surface area (BSA), and Dermatology Life Quality Index (DLQI), among others.5 First-line treatments include phototherapy, topical steroids, topical vitamin D, phototherapy, and oral systemic anti-inflammatory medications such as methotrexate, acitretin, and cyclosporine. Moderate-to-severe disease in adults that does not respond to first-line treatment is treated with biologic medications. Older generation biologics for PsO include anti–tumour necrosis factor (TNF) agents (etanercept, adalimumab, infliximab, and certolizumab pegol) and an anti-interleukin (IL)-12/IL-23 inhibitor (ustekinumab). More recently, a new generation of biologics have been approved for use for PsO. These include anti-IL-17 inhibitors (secukinumab, ixekizumab, brodalumab) and anti-IL-23 inhibitors (guselkumab, tildrakizumab, and risankizumab).1
Certain populations require special consideration in choosing the appropriate biologics therapy for PsO. For example, only 4 biologics are approved for the pediatric population (etanercept, ustekinumab, ixekizumab, and secukinumab).6-30 Studies have indicated that there are sex differences with regards to severity of PsO symptoms, prescribing patterns, and treatment outcomes.31-34 Although there are limited data on the use of biologics for PsO in pregnancy, a review by Ferreira et al. (2020) indicated that older generation biologics had lower risk compared with newer generation biologics.35 A systematic review by Sandhu et al. (2020) of biologic treatment in an elderly population with PsO recommended closer monitoring and prudent screening because serious adverse events and discontinuation due to adverse events were more common.36 PsO is also a lifelong condition and relapse is common after treatment discontinuation. As such, multiple treatment options may be needed throughout the life cycle of the disease.37
Objectives
Given the emergence of newer generation biologics and biosimilar options for older generation biologics in PsO, an ES was warranted for this treatment class. This ES provides regulatory, health technology assessment, and formulary status of biologics (originators and biosimilars) used in PsO relevant to Canadian FPT public drug plans. This scan also assesses the difference in use between old- and new-generation biologics for a sample population from 1 public drug plan.
The objectives of this ES are to provide a summary of the following for biologics for the treatment of moderate-to-severe PsO:
Regulatory status: including date of NOC, first marketed date, and indication with regards to PsO
Exclusivity status: data protection and patent expiry dates
CADTH review status: studies assessed and reasons for recommendation
Reimbursement status: listing status and coverage criteria across FPT public drug plans
Utilization patterns: market share of biologics for new patients for a public drug plan.
Methods
This ES provides information on regulatory status, reimbursement status, CADTH reviews, and utilization data on biologics and biosimilars used in patients with moderate-to-severe PsO. The components of the information presented in this scan are presented in Table 1. In this report, the terms “drugs” or “biologics” are used to refer to originators and their biosimilar versions (if available) as listed in Table 1.
Table 1: Components for Literature Screening and Information Gathering
Component | Description |
|---|---|
Population | Patients with moderate-to-severe PsO |
Intervention | Biologic (and biosimilars, if available) used in PsO:
|
Settings | Canadian publicly funded drug plans Provincial and territorial plans:
|
Federal plans:
| |
Types of information |
|
CDR = CADTH Common Drug Review; PsO = plaque psoriasis
aNunavut and the Northwest Territories follow the coverage category and criteria of the Non-Insured Health Benefits (NIHB) program.38,39
Literature Search
A grey literature search was conducted on key resources, including the websites of Health Canada’s drug product database, patent register, and data protection register; CADTH website (CADTH Common Drug Review [CDR] records); Canadian public drug plan formulary databases; and National Prescription Drug Utilization Information System (NPDUIS) database. No bibliographic literature searches were performed. The databases were searched between June 10, 2021, and August 18, 2021.
Some information presented in this report was not available in the public domain and was obtained through personal communication with members of the CADTH Formulary Working Group Health Technology Assessment FWG-HTA committee.40 In these cases, permission was obtained to publish this information in this report, and all details obtained through personal communication were referenced accordingly. Information from 4 federal public drug plans was included: Non-Insured Health Benefits (NIHB), CSC, Veterans Affairs Canada (VAC), and Canadian Armed Forces (CAF). Publicly reimbursed medications for residents of Nunavut and the Northwest Territories follow the coverage category and reimbursement criteria of the NIHB program.38,39
Utilization Patterns
Utilization data of old- versus new-generation biologics for new users was available for Ontario. Utilization data on biologics and biosimilars used in the treatment of PsO for the Ontario Public Drug Programs was provided by Reformulary Group.41 Eligible claims made to the Ontario Drug Benefit (ODB) Program during the period from April 1, 2017, to March 31, 2021, were included in the analysis. For this study, a market definition for biologics used to treat PsO that included all Drug Identification Numbers (DINs) was developed using the Reformulary drug data warehouse. Many public programs in Canada also define the drug products on their formulary using Product Identification Numbers (PINs); therefore, Reformulary drug data warehouse was used to identify the PINs and these were included in the market definition. Most biologics used to treat PsO are also indicated to treat other inflammatory conditions, so a reference drug list including drugs typically used in the management of each indication or use was also created. Limited Use codes in the claims data, where available, were used in determining the indication for each user in the ODB Program, and no further inference of indication or use was carried out.
Exclusions
Private payers and Quebec’s public drug program, the Régie de l’assurance maladie du Québec (RAMQ), were excluded. The biologics and biosimilars included in this report are also indicated for medical conditions other than PsO; however, relevant information related only to the PsO indication are presented in this report. In addition to coverage criteria for the specific drugs, other relevant formulary policies are presented in this report, such as policies on the use of biosimilar drugs and biosimilar switching. Although the clinical basis and economic basis for a CADTH recommendation for the biologics or biosimilars for PsO are presented, the ES did not assess the comparative clinical effectiveness or the relative cost-effectiveness of biologics or biosimilars drugs used in the treatment of patients with PsO. Thus, any conclusions or recommendations about the value of these medications or their place in therapy were outside of the scope of the ES.
Consultations
A stakeholder consultation was held between July 19, 2021, and July 30, 2021, to gather feedback on the scope of the ES. Feedback was received from 2 manufacturers and 1 joint patient input submission. Authors reviewed the feedback and duly revised the analysis and report.
Findings
Objective 1: Regulatory Status
Health Canada has approved 11 biologics for treatment of chronic moderate-to-severe PsO. Although all 11 biologics are indicated for the adult population, only 4 are indicated for pediatrics (etanercept, ustekinumab, ixekizumab, and secukinumab).6-30 Of the 11 biologics for PsO, 3 have biosimilar versions (adalimumab, etanercept, and infliximab) available in the Canadian market. Health Canada has approved 6, 2, and 4 biosimilar versions of adalimumab, etanercept, and infliximab, respectively. Among the 6 adalimumab biosimilars approved, 5 are currently marketed in Canada. Similarly, 3 of 5 biosimilar versions of infliximab are currently marketed in Canada. Both biosimilars of etanercept are marketed in Canada.6-11,13-15,22-28,42 Appendix 1 presents regulatory information, including manufacturer, date of NOC, first marketed date, and indication with regards to PsO for each drug.
The approval timeline in Figure 1 indicates the date of NOC for the PsO indication. Some drugs received NOC for the PsO indication after receiving their first NOC (for indications other than PsO). Some drugs received NOC for the PsO indication after receiving their first NOC (for indications other than PsO). Humira, Cimzia, Enbrel, Brenzys, Erelzi, and Remicade received their NOC for PsO in 2008 (versus 2004 for first NOC), 2018 (versus 2009 for first NOC), 2005 (versus 2000 for first NOC), 2020 (versus 2016 for first NOC), 2020 (versus 2017 for first NOC), and 2006 (versus 2001 for first NOC)., respectively. Old-generation biologics were defined as those with a first NOC before 2010, whereas new-generation biologics were defined as those with a first NOC in 2015 or later. Of the 5 old-generation biologics, 3 have biosimilar versions (infliximab, etanercept, and adalimumab) and 2 do not (certolizumab pegol and ustekinumab).
Figure 1: Health Canada Approval Timeline for Biologics by NOC Date for PsO Indication
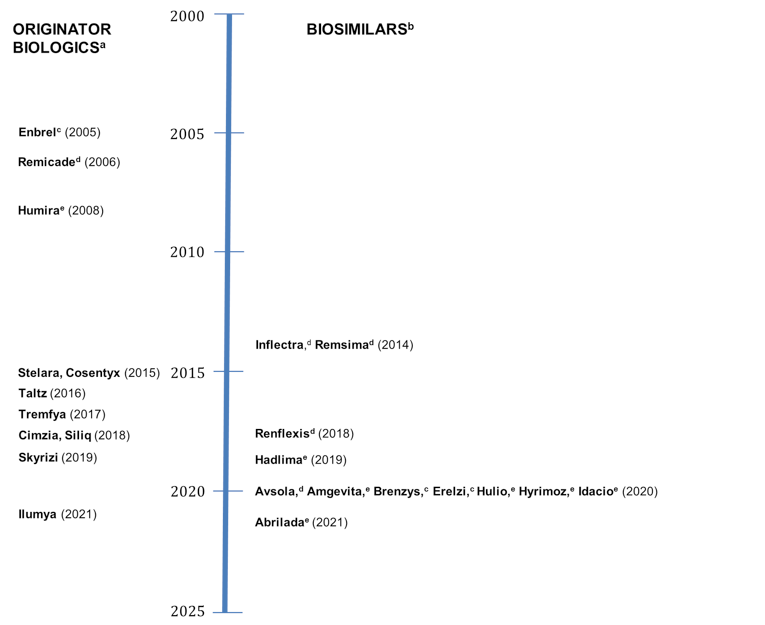
NOC = Notice of Compliance; PsO = plaque psoriasis.
a Originator biologics: Enbrel (etanercept), Remicade (infliximab), Humira (adalimumab), Stelara (ustekinumab), Cimzia (certolizumab pegol), Cosentyx (secukinumab), Siliq (brodalumab), Tremfya (guselkumab), Skyrizi (risankizumab), Ilumya (tildrakizumab), Taltz (ixekizumab).
b Biosimilars: Infliximab: Inflectra, Remsima, Renflexis, Avsola, Etanercept: Erelzi, Brenzys.
c Etanercept.
d Infliximab.
e Adalimumab.
Source: Information was collected from product monographs.6-30,42-51
Objective 2: Exclusivity Status
Exclusivity status is a function of patent and data protection. Patent protection is a 20-year period offered to innovative drugs from the date of filing that can be applied in various manners (e.g., chemical, change in use). Data protection regulations in Canada are governed by regulations under the Food and Drug Regulations published in 2006.52,53 These regulations provide data protection for an 8-year term with a possibility of adding 6 more months for submissions that include pediatric studies. During this time, only the owner or generator of preclinical and clinical trial data can use these data to obtain marketing authorization for drugs, effectively preventing a second-entry manufacturer from filing a submission for a copy of that innovative drug. Data protection begins from the time of issuance of NOC by Health Canada and when the drug is added to the Health Canada's Register of Innovative Drugs.52,53 Data protection for biologics in the US is 12 years from approval.54
Patent protection is still valid for most biologics except for 3 (infliximab, certolizumab, and ustekinumab); which had patent expiries as of October 14, 2021. The other 8 biologics (adalimumab, etanercept, guselkumab, risankizumab, tildrakizumab, brodalumab, ixekizumab, and secukinumab) had existing registered patents in Canada. Of these 8 biologics, the last patent to expire is for ixekizumab in 2033.55
Of the 11 biologics approved for use in PsO, all 5 old-generation biologics have had their data protection status expire or have predated the enactment of the data protection regulations of 2006. Among the 6 new-generation biologics for PsO, data protection will expire for secukinumab in 2023, followed by ixekizumab in 2024, guselkumab in 2025, brodalumab in 2026, risankizumab in 2027, and tildrakizumab in 2029.56 Table 2 provides information on the status of data protection and patent for biologics used for PsO.
Biosimilars are approved for adalimumab, infliximab, and etanercept, despite registered patents for originators of both adalimumab and etanercept. The approval of these biologics predated data protection regulations and thus data protection does not apply. However, it has been more than 8 years since their respective NOC dates for PsO indications (Figure 1). There appears to be delays between the NOC approval and marketed date for the first adalimumab biosimilar, and between the first NOC and the approval of the PsO indication for the etanercept biosimilars (Appendix 1). These delays are likely the result of litigation and/or agreements between the originator and biosimilar manufacturers.57,58 These delays represent approximately 3 years (time from NOC to marketed date for Hadlima) to 4 years (time from initial NOC to NOC for PsO for Brenzys) of foregone potential market authorization time for biosimilar versions (Appendix 1). Despite the expiry of data and patent protection for both ustekinumab and certolizumab, no biosimilar versions are available in Canada. Because the US market presents the largest opportunity for biosimilar developers,59 global development timelines for biosimilars may be based around US exclusivity timelines. The fact that the US applies a longer period of data exclusivity for biologics versus Canada54 may explain some of the previous delays and the future delays that may occur for biosimilar launches in Canada.
Table 2: Status of Data Protection and Patent Expiry
Generic name | Brand name (biologics) | Data protection expiry date | Data protection ended (yes/no) | Patent end date (for longest filed) | Patent expired (yes/no) |
|---|---|---|---|---|---|
Adalimumab | Humira | NAa | NAa | November 11, 2031 | No |
Brodalumab | Siliq | March 6, 2026 | No | January 12, 2031 | No |
Certolizumab pegol | Cimzia | August 12, 2017 | Yes | June 5, 2021 | Yes |
Etanercept | Enbrel | NAa | NAa | February 27, 2023 | No |
Guselkumab | Tremfya | November 10, 2025 | No | December 28, 2026 | No |
Infliximab | Remicade | NAa | NAa | August 1, 2017b | NA |
Ixekizumab | Taltz | November 25, 2024 | No | March 1, 2033 | No |
Risankizumab | Skyrizi | April 17, 2027 | No | November 2, 2031 | No |
Secukinumab | Cosentyx | August 27, 2023 | No | October 7, 2031 | No |
Tildrakizumab | Ilumya | May 19, 2029 | No | February 21, 2028 | No |
Ustekinumab | Stelara | December 12, 2016 | Yes | August 7, 2021 | Yes |
NA = not available.
aNOCs for adalimumab, etanercept, and infliximab were issued before the enactment of the data protection regulations in 2006.10,14,25,52
bThe longest patent that was filed for the originator infliximab was found to be infringed by the biosimilar launch. The only other patent filed for infliximab expired March 18, 2012.60
Source: Dates for data protection expiry and patent end date are from Health Canada databases.55,56,61
Objective 3: CADTH Review Status
CADTH has reviewed 5 of the 16 old-generation biologics for PsO (originators and biosimilars). Biosimilars approved after 2019 were not reviewed due to a policy change by CADTH to no longer review files completed after June 1 of that year.62 The 5 old-generation biologics reviewed by CADTH included 3 originators (Humira, Cimzia, and Stelara) and 2 infliximab biosimilars (Inflectra and Renflexis). These drugs were reviewed for chronic moderate-to-severe PsO; all were recommended to be listed with criteria and conditions.63-67 Enbrel and Remicade were approved by Health Canada for PsO before CADTH reviewed new indications for drugs.68 Biosimilars for etanercept (Brenzys and Erelzi) were reviewed by CADTH, but not for PsO.69,70 All 6 new-generation biologics (guselkumab, risankizumab, tildrakizumab, brodalumab, ixekizumab, and secukinumab) were reviewed by CADTH. These drugs were reviewed for chronic moderate-to-severe PsO and were recommended to be listed with criteria and conditions.71-76 Table 3 provides an overview of the CADTH review status and CDEC recommendations of the biologics for PsO; a summary of the evidence and clinical and/or economic rationale for the recommendations by CDEC are presented in Appendix 2.
With the exception of the biosimilar reviews and the review of Humira, both placebo- and active-controlled trials informed the CDEC recommendations (in addition to any indirect comparison). Direct comparisons demonstrating superiority or statistically significantly higher efficacy outcomes compared with the active comparator were available for ustekinumab (compared with etanercept), guselkumab (compared with adalimumab and ustekinumab), risankizumab (compared with adalimumab and ustekinumab), brodalumab (compared with ustekinumab), ixekizumab (compared with etanercept), and secukinumab (compared with etanercept). Direct comparisons of tildrakizumab with etanercept, and certolizumab with etanercept were also available, but no statistically significant differences were identified between the comparators for relevant efficacy outcomes. There is additional evidence of direct comparisons between these biologics from post-market studies conducted after the CADTH review. However, these studies are outside the scope of this ES. Indirect comparisons were also used to inform the CDR recommendation for certolizumab, risankizumab, tildrakizumab, and ixekizumab. Figure 3 presents a diagrammatic representation of evidence of direct comparison between biologics that informed the CDR recommendation.
Primary end points for the registration studies for biologics changed over time (Appendix 2). Earlier approvals were based on primary end points of achieving a 75% or greater reduction in PASI score (i.e., PASI 75), whereas more recent studies used primary end points that required the achievement of PASI 90 (e.g., guselkumab and risankizumab).
Table 3: CADTH Review Status and CDEC Recommendation for PsO
Generic name | Brand name | CADTH review (yes/no) | CDEC recommendation for PsO (date of publication) |
|---|---|---|---|
Adalimumab | Humira | Yes | List with clinical criteria and/or conditions (Oct. 16, 2008) |
Amgevita (biosimilar) | No | NAa | |
Hadlima (biosimilar) | No | NAa | |
Hulio (biosimilar) | No | NAa | |
Hyrimoz (biosimilar) | No | NAa | |
Idacio (biosimilar) | No | NAa | |
Abrilada (biosimilar) | No | NAa | |
Certolizumab pegol | Cimzia | Yes | Reimburse with clinical criteria and/or conditions (Nov. 20, 2019) |
Etanercept | Enbrel | No | NAb |
Brenzys (biosimilar) | No | NAc | |
Erelzi (biosimilar) | No | NAd | |
Infliximab | Remicade | No | NAb |
Avsola (biosimilar) | No | NAa | |
Inflectra (biosimilar) | Yes | List with criteria/condition (Dec. 19, 2014) | |
Omvyence (biosimilar) | No | NAa | |
Remsima (biosimilar) | Withdrawn | NA (Withdrawn) | |
Renflexis (biosimilar) | Yes | Reimburse with clinical criteria and/or conditions (Feb. 20, 2018) | |
Ustekinumab | Stelara | Yes | List with clinical criteria and/or conditions (Jun. 17, 2009) |
Guselkumab | Tremfya | Yes | Reimburse with clinical criteria and/or conditions (Feb. 21, 2018) |
Risankizumab | Skyrizi | Yes | Reimburse with clinical criteria and/or conditions (May 28, 2019) |
Tildrakizumab | Ilumya | Yes | Reimburse with clinical criteria and/or conditions (Jun. 21, 2021) |
Brodalumab | Siliq | Yes | Reimburse with clinical criteria and/or conditions (Jun. 20, 2018) |
Ixekizumab | Taltz | Yes | Reimburse with clinical criteria and/or conditions (Oct. 25, 2016) |
Secukinumab | Cosentyx | Yes | List with criteria/condition (Oct. 28, 2015) |
CDEC = CADTH Canadian Drug Expert Committee; NA = not applicable; PsO = plaque psoriasis.
aAs of June 1, 2019, CADTH stopped any work on any biosimilar reviews that would have been completed after June 1, 2019.62
b(Interim) CDR began in 200268
cReviewed by CDR in October 2016, but not for PsO.70
dReviewed by CDR in July 2017, but not for PsO. However, the reason for recommendation notes that trials conducted in patients with moderate-to-severe chronic plaque-type psoriasis demonstrating the pharmacokinetics, efficacy, safety, and immunogenicity of Erelzi are similar to those of the reference products.69
Source: Data were extracted from published CADTH reports.62-77
Figure 2: Network Diagram Representing Evidence of Direct Comparisons Between Biologics That Informed the CDR Recommendation
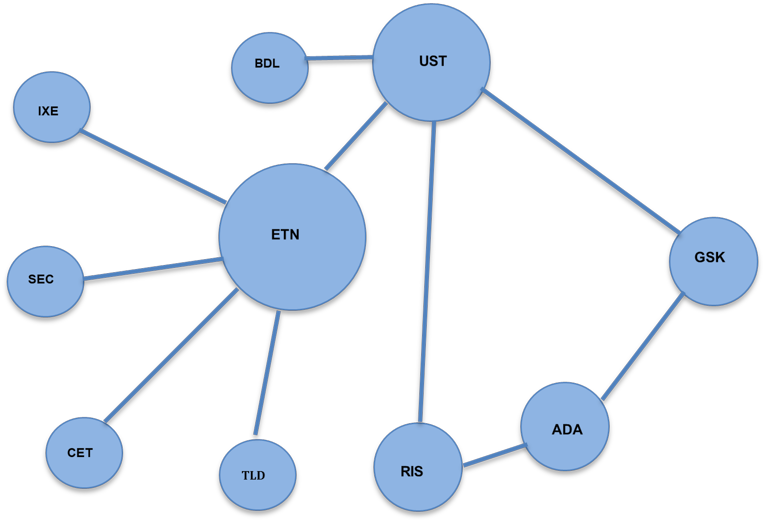
ADA = adalimumab; BDL = brodalumab; CET = certolizumab pegol; CDR = CADTH Common Drug Review; ETN = etanercept; GSK = guselkumab; IXE = ixekizumab; RIS = risankizumab; SEC = secukinumab; TLD = tildrakizumab; UST = ustekinumab.
Objective 4: Reimbursement Status
Negotiated Agreements
The pCPA was formed in 2010 for public drug plans to work together in when entering into negotiations with manufacturers for pharmaceuticals, from which, if successful, an LOI was created. The LOI lists the terms and conditions for funding a drug and are used to create a PLA between each participating member jurisdiction and the manufacturer.78 The following biologics have undergone pCPA negotiations for PsO that resulted in a LOI: Hadlima, Idacio, Hyrimoz, Amgevita, Hulio, Avsola, Inflectra, Skyrizi, Siliq, Taltz, and Cosentyx.79 Other drugs may have predated pCPA, failed to achieve an LOI, or have yet to begin negotiations. Information on the pCPA negotiation status of biologics for PsO are presented in Table 4.
Table 4: Overview of the pCPA Negotiation Status of Biologics
Generic name | Brand name | Status for PsO indication (date negotiation concluded) | Status of other indications (date negotiation concluded) |
|---|---|---|---|
Adalimumab | Humira | Concluded without agreement (2020-04-16)a | Ulcerative colitis: Concluded with LOI (2017-05-23) Hidradenitis suppurativa: Concluded with LOI (2017-10-23) |
Amgevita (biosimilar) | Concluded with LOI (2021-01-20) | Rheumatoid arthritis, polyarticular juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn disease, ulcerative colitis, hidradenitis suppurativa, and uveitis: Concluded with LOI (2021-01-20) | |
Hadlima (biosimilar) | Concluded with LOI (2021-01-22) | Rheumatoid arthritis, polyarticular juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn disease, ulcerative colitis, hidradenitis suppurativa, and uveitis: Concluded with LOI (2021-01-22) | |
Hulio (biosimilar) | Concluded with LOI (2021-01-20) | Rheumatoid arthritis, polyarticular juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn disease, ulcerative colitis, hidradenitis suppurativa, and uveitis: Concluded with LOI (2021-01-20) | |
Hyrimoz (biosimilar) | Concluded with LOI (2021-01-20) | Rheumatoid arthritis, polyarticular juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn disease, ulcerative colitis, hidradenitis suppurativa, and uveitis: Concluded with LOI (2021-01-20) | |
Idacio (biosimilar) | Concluded with LOI (2021-01-26) | Rheumatoid arthritis, polyarticular juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn disease, ulcerative colitis, hidradenitis suppurativa, and uveitis: Concluded with LOI (2021-01-26) | |
Certolizumab pegol | Cimzia | Concluded without agreement (2021-05-07) | Ankylosing spondylitis: Concluded with LOI (2016-01-06) |
Etanercept | Enbrel | NA | NA |
Brenzys (biosimilar) | NA | Rheumatoid arthritis and ankylosing spondylitis: Concluded with LOI (2017-06-19) | |
Erelzi (biosimilar) | NA | Ankylosing spondylitis, polyarticular juvenile idiopathic arthritis, and rheumatoid arthritis: Concluded with LOI (2017-10-17) | |
Infliximab | Remicade | NA | Rheumatoid arthritis, ulcerative colitis, and Crohn disease: Concluded with LOI (2015-02-23) |
Inflectra (biosimilar) | Concluded with LOI (2016-10-31) | Ankylosing spondylitis, psoriatic arthritis, Rheumatoid arthritis, Crohn disease, and ulcerative colitis: Concluded with LOI (2016-10-31) | |
Renflexis (biosimilar) | NA | Rheumatoid arthritis, psoriatic arthritis, and ankylosing spondylitis: Concluded with LOI (2018-07-27) | |
Avsola (biosimilar) | Concluded with LOI (2020-11-02) | Ankylosing spondylitis, psoriatic arthritis, rheumatoid arthritis, Crohn disease, and ulcerative colitis: Concluded with LOI (2020-11-02) | |
Ustekinumab | Stelara | NA | Ulcerative colitis: Concluded without agreement (2021-07-28) Crohn disease: Concluded without agreement (2019-03-29) Psoriatic arthritis: Negotiations were not pursued (2014-11-27) |
Guselkumab | Tremfya | Concluded without agreement (2019-06-21) | NA |
Risankizumab | Skyrizi | Concluded with LOI (2019-11-28) | NA |
Tildrakizumab | Ilumya | Under consideration for negotiation | NA |
Brodalumab | Siliq | Concluded with LOI (2018-12-14) | NA |
Ixekizumab | Taltz | Concluded with LOI (2017-12-29) | Ankylosing spondylitis: Concluded without agreement (2021-03-19) Psoriatic arthritis: Concluded with LOI (2018-11-08) |
Secukinumab | Cosentyx | Concluded with LOI (2016-06-08) | Ankylosing spondylitis: Concluded with LOI (2017-12-19) Psoriatic arthritis: Concluded with LOI (2017-12-19) |
LOI = Letter of Intent; NA = not applicable (negotiations not held); pCPA = pan-Canadian Pharmaceutical Alliance; PsO = plaque psoriasis.
aFor Humira (citrate-free) and negotiated for multiple indications (details of indications not specified).
Source: Data are from LOI timelines published on the pCPA website.78,79
Formulary Listing Status
The current process for formulary listings begins with an LOI which leads to PLAs with individual drug plans. However, not all drugs achieve the LOI stage; when they do, not all LOIs lead to successful PLAs with jurisdictions. Some jurisdictions can choose to not participate in a negotiation, or formulary listings may take longer if the drug is not a priority. Moreover, 4 of 5 old-generation biologics predated the pCPA process, which means that PLAs may be disparate across public drug plans.80 For biologics for PsO, certolizumab, guselkumab, and tildrakizumab were not covered by any of the 14 public drug plans (tildrakizumab has only recently been marketed in Canada). Other drugs that are not covered include risankizumab by CSC and CAF; brodalumab by Alberta, British Columbia, Yukon, VAC, and CSC; ixekizumab by Yukon and CSC; and secukinumab by CSC.20,38,39,71,81-97 Moreover, some drug plans do not cover (or have plans to stop coverage for) originators for which a biosimilar version is available. As such, Alberta, British Columbia, and New Brunswick do not provide coverage for Humira, Enbrel, and Remicade; Ontario and CSC do not provide coverage for Remicade; and Newfoundland and Labrador, NIHB, and CSC do not cover Enbrel.81,83,84,88,89,92-97 Conversely, some drug plans do not cover biosimilar versions that are available; for example, Nova Scotia, Prince Edward Island, CAF, and VAC do not cover any of the adalimumab biosimilars, and Nova Scotia and Prince Edward Island do not cover any of the etanercept biosimilars.82,85-87,90,91
Tiered Formularies
Some drug plans have a 2-tiered biologics formulary (i.e., Alberta, Manitoba, and CSC). The policy requires patients to trial and fail the first tier of biologics to be eligible for coverage of the second tier. Alberta requires patients to be refractory or intolerant to at least 3 other biologics (adalimumab, etanercept, infliximab, ixekizumab, risankizumab, and secukinumab) before reimbursing ustekinumab. Manitoba’s policy applies to new patients (biologic naive) and existing patients who have previously been trialled and deemed unresponsive to biologic therapy. Patients must fail to respond to more than 2 Tier 1 drugs to be eligible for coverage for Tier 2 drugs. Tier 1 drugs include biosimilar versions of infliximab (Avsola, Inflectra, Renflexis) and etanercept (Erelzi and Brenzys), and originator biologics Cosentyx, Humira, Siliq, Skyrizi, and Taltz. Tier 2 drugs include originator biologics Enbrel, Remicade, and Stelara.98-100 CSC also requires existing patients deemed unresponsive to biologic therapy to switch to a Tier 1 biologic. Once all options on Tier 1 are exhausted, options on Tier 2 list can be made available. Tier 1 drugs include biosimilar versions of etanercept (Brenzys, Erelzi), adalimumab (Hadlima, Hyrimoz, Idacio), and infliximab (Inflectra, Renflexis). Tier 2 drugs include Stelara and Humira.84
Table 5 provides an overview of the listing status of biologics for PsO in public drug plans.
Table 5: Overview of Listing Status of Biologics for PsO in Public Drug Plans
Generic name | Brand name | AB | BC | SK | MB | ON | NB | NS | NL | PE | YT | NIHBa | VACb | CSC | CAF |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Adalimumab | Humira | NAB | NAB | EDS | EDS | LU | NAB | ESD | SA | SA | EDS | LU/PA | SA | Tier 2 | SA |
Amgevita (biosimilar) | SA | LCD/SA | EDS | EDS | LU | SA | NAB | SA | NAB | EDS | LU/PA | SA | NAB | NAB | |
Hadlima (biosimilar) | SA | LCD/SA | EDS | EDS | LU | SA | NAB | SA | NAB | NAB | LU/PA | NAB | CM | NAB | |
Hulio (biosimilar) | SA | LCD/SA | EDS | EDS | LU | SA | NAB | SA | NAB | EDS | LU/PA | SA | NAB | NAB | |
Hyrimoz (biosimilar) | SA | LCD/SA | EDS | EDS | LU | SA | NAB | SA | NAB | NAB | LU/PA | SA | CM | NAB | |
Idacio (biosimilar) | SA | LCD/SA | EDS | EDS | LU | SA | NAB | SA | NAB | NAB | LU/PA | SA | CM | NAB | |
Certolizumab | Cimzia | NAB | |||||||||||||
Etanercept | Enbrel | NAB | NAB | EDS | Tier 2 | LU | NAB | ESD | NAB | SA | EDS | NAB | SA | NAB | SA |
Brenzys (biosimilar) | SA | LCD/SA | EDS | EDS | LU | SA | NAB | SA | NAB | EDS | NAB | NAB | CM | SA | |
Erelzi (biosimilar) | SA | LCD/SA | EDS | EDS | LU | SA | NAB | SA | NAB | EDS | LU/PA | SA | CM | SA | |
Infliximab | Remicade | NAB | NAB | EDS | Tier 2 | NAB | NAB | ESD | SA | SA | EDS | LU/PA | SA | NAB | SA |
Inlfectra (biosimilar) | SA | LCD/SA | EDS | EDS | LU | SA | ESD | SA | SA | EDS | LU/PA | SA | CM | SA | |
Renflexis (biosimilar) | SA | LCD/SA | EDS | EDS | LU | SA | ESD | SA | SA | NAB | LU/PA | SA | CM | SA | |
Avsola (biosimilar) | SA | LCD/SA | EDS | EDS | LU | SA | NAB | SA | NAB | NAB | LU/PA | SA | NAB | NAB | |
Ustekinumab | Stelara | Tier 2 | LCD/SA | EDS | Tier 2 | LU | SA | ESD | SA | SA | EDS | LU/PA | SA | Tier 2 | SA |
Guselkumab | Tremfya | NAB | |||||||||||||
Risankizumab | Skyrizi | SA | LCD/SA | EDS | EDS | LU | SA | ESD | SA | SA | EDS | LU/PA | SA | NAB | NAB |
Tildrakizumab | Ilumya | NABc | |||||||||||||
Brodalumab | Siliq | NAB | NAB | EDS | EDS | LU | SA | ESD | SA | SA | NAB | LU/PA | NAB | NAB | SA |
Ixekizumab | Taltz | SA | LCD/SA | EDS | EDS | LU | SA | ESD | SA | SA | NAB | LU/PA | SA | NAB | SA |
Secukinumab | Cosentyx | SA | LCD/SA | EDS | EDS | LU | SA | ESD | SA | SA | EDS | LU/PA | SA | NAB | SA |
AB = Alberta; BC = British Columbia; CAF = Canadian Armed Forces; CM = Criteria Medicine; CSC = Correctional Services Canada; EDS = Exception Drug Status; ESD = Exception Status Drug; LU = Limited Use; MB = Manitoba; NAB = not a benefit; NB = New Brunswick; NIHB = Non-Insured Health Benefits; NL = Newfoundland and Labrador; NS = Nova Scotia; ON = Ontario; PA = Prior Approval; PE = Prince Edward Island; SA = Special Authorization; SK = Saskatchewan; UR = under review; VAC = Veterans Affairs Canada; YT = Yukon.
Note: Tier 2 refers to reimbursement only after tier 1 drugs have been trialled. Shaded cells indicate drugs that are not listed.
aAlso applicable to Nunavut and the Northwest Territories.38,39
bInformation is based on a personal communication with the jurisdictional representative (Anne Bastarache: personal communication, July 2021).
cTildrakizumab is not yet marketed in Canada and was only recently reviewed by CDR.20,71
Source: Listing statuses are from payer formulary websites.,81-97
Type of Listing Status
Public drug plans list prescription medicines according to specific coverage categories that can be broadly classified as restricted benefits or unrestricted benefits. “Unrestricted benefit” refers to drugs with usage that is not limited by clinical criteria requiring authorization. Depending on the public drug plan, this type of formulary benefit status (coverage category) is referred to as open benefit, full benefit, or regular benefit. “Restricted benefit” refers to drugs with usage limited by specific clinical criteria or to a defined patient subgroup. Depending on the public drug plan, this type of formulary benefit status is categorized under Special Authorization, Exceptional Access Program, Exceptional Drug Status (EDS), Limited Use, Limited Coverage Drug, or Prior Authorization.101 The “restricted benefit” categories can be further classified by the following reimbursement processes:
Restricted Benefit–Active: Applicable to the following coverage categories for biologics for PsO: Special Authorization or Limited Coverage Drug (British Columbia), Special Authorization (Alberta, New Brunswick, Newfoundland and Labrador, Prince Edward Island, VAC, CAF), EDS (Saskatchewan, Manitoba, Nova Scotia, Yukon), or limited use or prior authorization (NIHB). Application for public reimbursement with the required clinical details must be made by the authorized prescriber using established processes (e.g., use of specific authorization forms). Each request is subject to a medication review by staff responsible for claims adjudication for the public drug plan.82,83,85-91,93-97
Restricted Benefit–Passive: Applicable to the following coverage categories for biologics for PsO: Limited Use (Ontario) and benefit with criteria medications (CSC). In comparison with Restricted Benefit–Active, the use of specific authorization forms and a medication review is not a requirement. Rather, a Limited Use code (Ontario) or a Reason for Use code (CSC) must be specified in the prescription.84,92
Coverage Criteria
All biologics for PsO were categorized as a “restricted benefit”; therefore, the drugs were reimbursed under specific medical circumstances in which patients were required to meet specific clinical criteria for drugs to be eligible for reimbursement. These clinical criteria were established by each drug plan’s formulary review committee. In general, initial coverage criteria for biologics in PsO was some form of the following: objective diagnosis of PsO (including 1 or more of the following: PASI score > 10, DLQI > 10, BSA > 10%, and significant involvement of the face, palms of the hands, soles of the feet, or genital region) and failure to respond to, contraindication to, or intolerant of MTX and cyclosporine, and phototherapy (unless unable to access) before being considered for coverage with biologics for PsO.20,38,39,71,81-97
Appendix 3 provides a summary of coverage criteria, and Appendix 4 provides information on approved dose and duration of therapy for biologics for PsO across public drug plans.
Objective 5: Utilization Patterns
Data obtained from ODB was used to calculate the proportion of new claimants (biologic-naive patients) who initiated treatment with an old- or new-generation biologic in 2019 and 2020. The proportion of patients reimbursed for old-generation biologics decreased from 54.4% in 2019 to 36.8% in 2020, whereas the proportion of patients reimbursed for new-generation biologics increased from 45.6% in 2019 to 63.2% in 2020.
Figure 3: Market Share of New Users (Claimants) for Old-Generation Versus New-Generation Biologics for PsO (ODB 2019 and 2020)
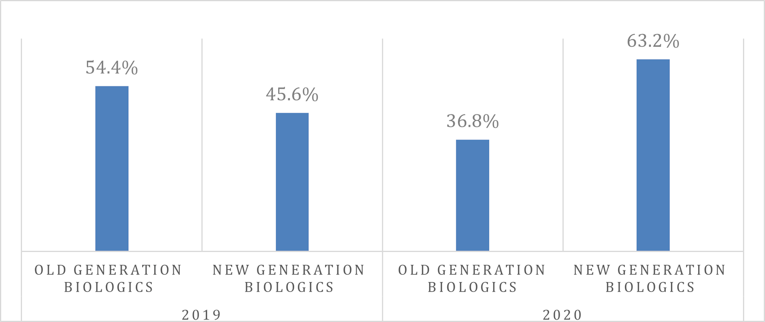
ODB = Ontario Drug Benefit; PsO = plaque psoriasis.
Note: Old-generation biologics = adalimumab, infliximab, etanercept, ustekinumab, and certolizumab; New-generation biologics = secukinumab, ixekizumab, risankizumab, and brodalumab.
Source: Utilization analysis provided by Reformulary Group.41
Discussion and Implications for Policy-Making
Biosimilar delays. Formulary management has not traditionally been required for drug plans because expenditures were attenuated through the uptake of generic medications after the loss of exclusivity for originator drugs (i.e., expiry of data protection and/or patents). However, this drug life cycle has not been achieved for the class of biologics for several reasons, including that biosimilars have not been deemed bioequivalent to their originators.102 In addition to the challenges of adopting biosimilars into clinical practice, this ES uncovered significant delays in the marketing of biosimilars. In the cases of ustekinumab and certolizumab, there were no biosimilar versions under review for market authorization despite the expiry of patents and data protection. Although the market authorizations for infliximab, adalimumab, and etanercept were in the early 2000s, and thus predated data protection regulations, it would have been expected that a second-entry manufacturer would file a submission for a copy of these drugs in the early 2010s (if the standard generic life cycle was applicable). However, the PsO indication for biosimilars of adalimumab and etanercept were not marketed until 2021 and 2020, respectively. Notably, initial NOCs for adalimumab and etanercept were valid for some biosimilar versions as early as 2018 and 2016, respectively. It appears that these delays are driven by factors external to the Canadian context, such as global agreements among manufacturers58 and global development timelines for biosimilars more closely aligned to the exclusivity timelines of the US.59 This begs the policy question for how Canadian payers can address affordability of biologics in the context of delayed or unavailable biosimilar competitors. Formulary management strategies could be employed, such as pursuing pCPA negotiations for old-generation originators that do not have biosimilar versions and/or establishing tiered formularies to prioritize the reimbursement of old-generation biosimilars
Value of new-generation biologics. New-generation biologics have stronger evidence packages compared with old-generation biologics. CDEC recommendations for all 6 new-generation biologics were based on randomized controlled trials that compared the drug with placebo and an active comparator, and demonstrated superiority or non-inferiority over the old-generation comparators. Moreover, some of the new-generation biologics had clinical trial programs that included more stringent primary outcomes versus clinical trial programs for old-generation biologics. New-generation biologics may also have better value for public payers compared with old-generation biologics due to their inclusion within the pCPA negotiation process. Because reimbursement for old-generation biologics predated the pCPA process, it is feasible that PLAs for these drugs are disparate across public drug plans.80 This is further supported by the existence of tiered formularies by several drug plans. Therefore, it is possible that public payers may be paying less and getting better outcomes for new-generation versus old-generation biologics for PsO.
Policy options. Public payers may choose to seek greater value from old-generation biologics based on the results of this ES. This can be achieved by undertaking pCPA negotiations for old-generation originators for which biosimilar versions are not available. Conversely, public payers may choose to adopt tiered formularies (such as those implemented by Alberta, Manitoba, and CSC) to ensure optimal use of the most valuable therapeutic options. There may also be a rationale to justify conducting similar scans for other disease areas treated by biologics.
Conclusion
Formulary management is warranted for biologics for PsO given the significant utilization of old-generation originators in the current context of delayed marketing of their biosimilar versions and their reimbursement predating the pCPA process. Conversely, new-generation biologics underwent pCPA negotiations and provided direct evidence that demonstrated superiority compared with old-generation biologics. Therefore, it is likely that payers have not had optimal value for biologics in PsO because old-generation originators have been reimbursed beyond loss of exclusivity at a lower likelihood of having a PLA compared with superior new-generation biologics. To optimize value in this class, formulary management strategies could be employed, such as pursuing pCPA negotiations for old-generation originators that do not have biosimilar versions and/or establishing tiered formularies to prioritize the reimbursement of old-generation biosimilars and new-generation originators.
References
1.Feldman SR. Treatment of psoriasis in adults. In: Post T, ed. UpToDate. Waltham (MA): UpToDate; 2021: https://www.uptodate.com/contents/treatment-of-psoriasis-in-adults. Accessed 2021 Jun 8.
2.Canadian Psoriasis Network. About psoriasis. 2021; https://www.canadianpsoriasisnetwork.com/psoriasis/. Accessed 2021 Jun 18.
3.Canadian Psoriasis Network. Plaque psoriasis. 2021; https://www.canadianpsoriasisnetwork.com/psoriasis/forms-of-psoriasis/. Accessed 2021 Jun 18.
4.Canadian Psoriasis Network. Conditions associated with psoriasis. 2021; https://www.canadianpsoriasisnetwork.com/psoriasis/conditions/. Accessed 2021 Jun 18.
5.Oakley A, Jones C, Gupta M. Assessment of psoriasis. DermNet NZ 2014; https://dermnetnz.org/topics/psoriasis/. Accessed 2021 Jun 18.
6.Health Canada. Product Information for Abrilada. 2021; https://health-products.canada.ca/dpd-bdpp/info.do?lang=en&code=100029. Accessed 2021 Aug 15.
7.Health Canada. Product information for Amgevita. 2021; https://health-products.canada.ca/dpd-bdpp/info.do?lang=en&code=94649. Accessed 2021 Jun 10.
8.Health Canada. Product information for Hadlima. 2021; https://health-products.canada.ca/dpd-bdpp/info.do?lang=en&code=96243. Accessed 2021 Jun 10.
9.Health Canada. Product information for Hulio. 2021; https://health-products.canada.ca/dpd-bdpp/info.do?lang=en&code=99197. Accessed 2021 Jun 10.
10.Health Canada. Product information for Humira. 2021; https://health-products.canada.ca/dpd-bdpp/info.do?lang=en&code=74379. Accessed 2021 Jun 10.
11.Health Canada. Product information for Idacio. 2021; https://health-products.canada.ca/dpd-bdpp/info.do?lang=en&code=99222. Accessed 2021 Jun 10.
12.Health Canada. Product information for Cimzia. 2021; https://health-products.canada.ca/dpd-bdpp/info.do?lang=en&code=81737. Accessed 2021 Jun 10.
13.Health Canada. Product information for Brenzys. 2021; https://health-products.canada.ca/dpd-bdpp/info.do?lang=en&code=94214. Accessed 2021 Jun 10.
14.Health Canada. Product information for Enbrel. 2021; https://health-products.canada.ca/dpd-bdpp/info.do?lang=en&code=66735. Accessed 2021 Jun 10.
15.Health Canada. Product information for Erelzi. 2021; https://health-products.canada.ca/dpd-bdpp/info.do?lang=en&code=95072. Accessed 2021 Jun 10.
16.Health Canada. Product information for Stelara. 2021; https://health-products.canada.ca/dpd-bdpp/info.do?lang=en&code=80608. Accessed 2021 Jun 22.
17.Health Canada. Product information for Tremfya. 2021; https://health-products.canada.ca/dpd-bdpp/info.do?lang=en&code=95860. Accessed 2021 Jun 10.
18.Health Canada. Product information for Tremfya One-Press. 2021; https://health-products.canada.ca/dpd-bdpp/info.do?lang=en&code=97748. Accessed 2021 Jun 22.
19.Health Canada. Product information for Siliq. 2021; https://health-products.canada.ca/dpd-bdpp/dispatch-repartition.do. Accessed 2021 Jun 22.
20.Health Canada. Product information for Ilumya. 2021; https://health-products.canada.ca/dpd-bdpp/info.do?lang=en&code=100544. Accessed 2021 Jun 22.
21.Health Canada. Product information for Skyrizi. 2021; https://health-products.canada.ca/dpd-bdpp/info.do?lang=en&code=97761. Accessed 2021 Jun 22.
22.Health Canada. Product information for Avsola. 2021; https://health-products.canada.ca/dpd-bdpp/info.do?lang=en&code=98681. Accessed 2021 Jun 22.
23.Health Canada. Product information for Inflectra. 2021; https://health-products.canada.ca/dpd-bdpp/info.do?lang=en&code=90410. Accessed 2021 Jun 22.
24.Health Canada. Product information for Omvyence. 2021; https://health-products.canada.ca/dpd-bdpp/info.do?lang=en&code=99953. Accessed 2021 Jun 22.
25.Health Canada. Product information for Remicade. 2021; https://health-products.canada.ca/dpd-bdpp/info.do?lang=en&code=67959. Accessed 2021 Jun 22.
26.Health Canada. Product information for Remsima. 2021; https://health-products.canada.ca/dpd-bdpp/info.do?lang=en&code=90411. Accessed 2021 Jun 22.
27.Health Canada. Product information for Remsima SC. 2021; https://health-products.canada.ca/dpd-bdpp/info.do?lang=en&code=100076. Accessed 2021 Jun 22.
28.Health Canada. Product information for Renflexis. 2021; https://health-products.canada.ca/dpd-bdpp/info.do?lang=en&code=95936. Accessed 2021 Jun 22.
29.Health Canada. Product information for Taltz. 2021; https://health-products.canada.ca/dpd-bdpp/info.do?lang=en&code=94193. Accessed 2021 Jun 22.
30.Health Canada. Product information for Cosentyx. 2021; https://health-products.canada.ca/dpd-bdpp/info.do?lang=en&code=92243. Accessed 2021 Jun 22.
31.Murer C, Sgier D, Mettler SK, et al. Gender differences in psoriasis: a Swiss online psoriasis survey. Arch Dermatol Res. 2021;313(2):89-94. PubMed
32.Colombo D, Cassano N, Bellia G, A. VG. Gender medicine and psoriasis. World J Dermatol. 2014;3(3):36-44.
33.Hägg D, Eriksson M, Sundström A, Schmitt-Egenolf M. The higher proportion of men with psoriasis treated with biologics may be explained by more severe disease in men. PLoS One. 2013;8(5):e63619. PubMed
34.Maul JT, Augustin M, Sorbe C, et al. Association of sex and systemic therapy treatment outcomes in psoriasis: a two-country, multicentre, prospective, noninterventional registry study. Br J Dermatol. 2021;[online ahead of print]. PubMed
35.Ferreira C, Azevedo A, Nogueira M, Torres T. Management of psoriasis in pregnancy - a review of the evidence to date. Drugs Context. 2020;9. PubMed
36.Sandhu VK, Ighani A, Fleming P, Lynde CW. Biologic treatment in elderly patients with psoriasis: a systematic review. J Cutan Med Surg. 2020;24(2):174-186. PubMed
37.Huang YW, Tsai TF. Remission duration and long-term outcomes in patients with moderate-to-severe psoriasis treated by biologics or tofacitinib in controlled clinical trials: a 15-year single-center experience. Dermatol Ther (Heidelb). 2019;9(3):553-569. PubMed
38.Government of the Northwest Territories Health and Social Services. Extended health benefits for specified disease conditions program. 2019; https://www.hss.gov.nt.ca/en/services/supplementary-health-benefits/extended-health-benefits-specified-disease-conditions. Accessed 2021 Jun 22.
39.Government of Nunavut. EHB full coverage plan. 2020; https://www.gov.nu.ca/health/information/ehb-full-coverage-plan. Accessed 2021 Jun 22.
40.Pharmaceutical Advisory Committee Formulary Working Group for health technology assessments. Ottawa (ON): CADTH; 2021: https://www.cadth.ca/collaboration-and-outreach/advisory-bodies/dpac-fwg-hta. Accessed 2021 Jun 22.
41.Reformulary Group. About us. 2021; https://reformulary.com/about-us. Accessed 2021 Jul 8.
42.Health Canada. Product information for Hyrimoz. 2021; https://health-products.canada.ca/dpd-bdpp/info.do?lang=en&code=99460. Accessed 2021 Jun 10.
43.Health Canada. Notice of compliance (NOC) for Enbrel. 2005; https://health-products.canada.ca/noc-ac/info.do?lang=en&no=7665. Accessed 2021 Aug 14.
44.Health Canada. Notice of Compliance (NOC) for Remicade. 2006; https://health-products.canada.ca/noc-ac/info.do?lang=en&no=8015. Accessed 2021 Aug 14.
45.Health Canada. Notice of Compliance (NOC) for Humira. 2008; https://health-products.canada.ca/noc-ac/info.do?lang=en&no=9310. Accessed 2021 Aug 14.
46.Health Canada. Notice of Compliance (NOC) for Stelara. 2015; https://health-products.canada.ca/noc-ac/info.do?lang=en&no=17584. Accessed 2021 Aug 14.
47.Health Canada. Notice of Compliance (NOC) for Cimzia. 2018; https://health-products.canada.ca/noc-ac/info.do?lang=en&no=21220. Accessed 2021 Aug 14.
48.Health Canada. Notice of Compliance (NOC) for Brenzys. 2020; https://health-products.canada.ca/noc-ac/info.do?lang=en&no=24091. Accessed 2021 Aug 14.
49.Health Canada. Notice of Compliance (NOC) for Erelzi. 2020; https://health-products.canada.ca/noc-ac/info.do?lang=en&no=23793. Accessed 2021 Aug 14.
50.Health Canada. Notice of Compliance (NOC) for Taltz. 2021; https://health-products.canada.ca/noc-ac/info.do?lang=en&no=25547. Accessed 2021 Aug 14.
51.Health Canada. Notice of Compliance (NOC) for Cosentyx. 2021; https://health-products.canada.ca/noc-ac/info.do?lang=en&no=25044. Accessed 2021 Aug 14.
52.Kendall M, Hamill D. A Decade of Data Protection for Innovative Drugs in Canada: Issues, Limitations, and Time for a Reassessment. Biotechnology Law Report. 2016;35(6). PubMed
53.Guidance document: data protection under C.08.004.1 of the Food and Drug Regulations. Ottawa (ON): Health Canada; 2011: https://www.canada.ca/en/health-canada/services/drugs-health-products/drug-products/applications-submissions/guidance-documents/guidance-document-data-protection-under-08-004-1-food-drug-regulations.html. Accessed 2021 Jul 20.
54.Reducing data protection for biologics would slow medical progress and chill R&D investment in the U.S. Washington (DC): PhRMA; 2015: http://phrma-docs.phrma.org/sites/default/files/pdf/data-exclusivity-for-biologics-fact-sheet.pdf. Accessed 2021 Oct 19.
55.Health Canada. Patent register. 2021; https://pr-rdb.hc-sc.gc.ca/pr-rdb/index-eng.jsp. Accessed 2021 Jun 22.
56.Register of innovative drugs. Ottawa (ON): Health Canada; 2021: https://www.canada.ca/en/health-canada/services/drugs-health-products/drug-products/applications-submissions/register-innovative-drugs.html. Accessed 2021 Jun 22.
57.Mathias T. AbbVie, Samsung Bioepis in deal; Humira biosimilar U.S. release in 2023. Reuters 2018; https://www.reuters.com/article/us-abbvie-biogen-idUSKCN1HC1SP. Accessed 2021 Aug 10.
58.Courage N, Shivakumar S. The biosimilar landscape in Canada. Lexology 2018; https://www.lexology.com/library/detail.aspx?g=bc06ea65-d2bc-4ffe-bf04-9b497b10fce0. Accessed 2021 Oct 14.
59.Blackstone EA, Joseph PF. The economics of biosimilars. Am Health Drug Benefits. 2013;6(8):469-478. PubMed
60.Lee K, Wojtyra U. New Use Patent for Janssen’s REMICADE valid and infringed by Hospira’s biosimilar INFLECTRA. Smart & Biggar 2018; https://www.smartbiggar.ca/insights/publication/new-use-patent-for-janssen-s-remicade-valid-and-infringed-by-hospira-s-biosimilar-inflectra. Accessed 2021 Oct 13.
61.Lexchin J. Increase in Drug Spending in Canada Due to Extension of Data Protection for Biologics: A Descriptive Study. Healthc Policy. 2019;14(3):10-18. PubMed
62.CADTH pharmaceutical reviews update — issue 8. Ottawa (ON): CADTH; 2019: https://www.cadth.ca/cadth-pharmaceutical-reviews-update-issue-8. Accessed 2021 Jun 20.
63.CADTH Canadian Drug Expert Committee (CDEC) final recommendation: infliximab (Renflexis - Merck Canada Inc). 2018 Feb 20. https://cadth.ca/infliximab-3. Accessed 2021 Jun 28.
64.CADTH Canadian Drug Expert Committee (CDEC) final recommendation: certolizumab pegol (Cimzia - UCB Canada Inc). Ottawa (ON): CADTH; 2018 Nov 20: https://cadth.ca/certolizumab-pegol-2. Accessed 2021 Jun 28.
65.CADTH Canadian Drug Expert Committee (CDEC) final recommendation: infliximab (Inflectra - Hospira Healthcare Corp). Ottawa (ON): CADTH; 2014 Dec 19: https://cadth.ca/infliximab-1. Accessed 2021 Jun 28.
66.CADTH Canadian Drug Expert Committee (CDEC) final recommendation: adalimumab (Humira - Abbott Laboratories Ltd). 2008 Oct 16. https://cadth.ca/adalimumab-3. Accessed 2021 Jun 28.
67.CADTH Canadian Drug Expert Committee (CDEC) final recommendation: ustekinumab (Stelara - Janssen-Ortho Inc). 2009 Jun 17. https://cadth.ca/ustekinumab. Accessed 2021 Jun 28.
68.MacPhail E, Shea B. An inside look at the early history of the CADTH Common Drug Review in Canada. Ottawa (ON): CADTH; 2017: https://www.cadth.ca/sites/default/files/pdf/early_history_of_CDR.pdf. Accessed 2021 Jun 20.
69.CADTH Canadian Drug Expert Committee (CDEC) final recommendation: etanercept (Erelzi - Sandoz Canada Inc). Ottawa (ON): CADTH; 2017 Jul 25: https://cadth.ca/etanercept-0. Accessed 2021 Jun 28.
70.CADTH Canadian Drug Expert Committee (CDEC) final recommendation: etanercept (Brenzys - Merck Canada Inc). Ottawa (ON): CADTH; 2016 Oct 25: https://cadth.ca/etanercept. Accessed 2021 Jun 28.
71.CADTH Canadian Drug Expert Committee (CDEC) final recommendation: tildrakizumab (Ilumya - Sun Pharma Canada). Ottawa (ON): CADTH; 2021 Jun 21: https://cadth.ca/tildrakizumab. Accessed 2021 Jun 28.
72.CADTH Canadian Drug Expert Committee (CDEC) final recommendation: risankizumab (Skyrizi - AbbVie Corporation). Ottawa (ON): CADTH; 2019 May 28: https://cadth.ca/risankizumab. Accessed 2021 Jun 28.
73.CADTH Canadian Drug Expert Committee (CDEC) final recommendation: brodalumab (Siliq - Valeant Canada LP). Ottawa (ON): CADTH; 2018 Jun 20: https://cadth.ca/brodalumab. Accessed 2021 Jun 28.
74.CADTH Canadian Drug Expert Committee (CDEC) final recommendation: guselkumab (Tremfya - Janssen Inc). Ottawa (ON): CADTH; 2018 Feb 21: https://cadth.ca/guselkumab. Accessed 2021 Jun 28.
75.CADTH Canadian Drug Expert Committee (CDEC) final recommendation: ixekizumab (Taltz - Eli Lilly Canada Inc). Ottawa (ON): CADTH; 2016 Oct 25: https://cadth.ca/ixekizumab. Accessed 2021 Jun 28.
76.CADTH Canadian Drug Expert Committee (CDEC) final recommendation: secukinumab (Cosentyx - Novartis Pharmaceuticals Canada Inc). 2015 Oct 28. https://cadth.ca/secukinumab. Accessed 2021 Jun 28.
77.CADTH Canadian Drug Expert Committee (CDEC) final recommendation: infliximab (Remsima - Fresenius Kabi Canada). Ottawa (ON): CADTH; 2014 May 30: https://cadth.ca/infliximab-0. Accessed 2021 Jun 28.
78.pan-Canadian Pharmaceutical Alliance. FAQs. 2021; https://www.pcpacanada.ca/faq#:~:text=The%20pCPA%20enters%20into%20negotiations,member%20jurisdiction%20and%20the%20manufacturer.https://www.pcpacanada.ca/faq#:~:text=The%20pCPA%20enters%20into%20negotiations,member%20jurisdiction%20and%20the%20manufacturer. Accessed 2021 Jun 22.
79.pan-Canadian Pharmaceutical Alliance. Brand name drug negotiations status. 2021; https://www.pcpacanada.ca/negotiations. Accessed 2021 Jun 22.
80.Morgan SG, Friesen MK, Thomson PA, Daw JR. Use of product listing agreements by Canadian provincial drug benefit plans. Healthc Policy. 2013;8(4):45-55. PubMed
81.Government of Alberta. Interactive Drug Benefit List (iDBL). 2021; https://www.ab.bluecross.ca/dbl/publications.php. Accessed 2021 Jun 22.
82.Express Scripts Canada. Drug benefit list. 2020; https://nihb.express-scripts.ca/dbl. Accessed 2021 Aug 18.
83.New Brunswick drug plans formulary. Frederiction (NB): Government of New Brunswick; 2021: https://www2.gnb.ca/content/dam/gnb/Departments/h-s/pdf/en/NBDrugPlan/NewBrunswickDrugPlansFormulary.pdf. Accessed 2021 Jun 22.
84.Correctional Service Canada. National drug formulary. 2021. Accessed 2021 Aug 18.
85.Veterans Affairs Canada. Drug formulary search form. 2019; https://www.veterans.gc.ca/eng/financial-support/medical-costs/treatment-benefits/poc10/search. Accessed 2021 Jun 22.
86.National Defence and the Canadian Armed Forces. Drug benefit list. 2019; http://www.cmp-cpm.forces.gc.ca/hs/en/drug-benefit-list/index.asp. Accessed 2021 Aug 18.
87.Formulary. Halifax (NS): Nova Scotia Department of Health; 2021: https://novascotia.ca/dhw/pharmacare/documents/formulary.pdf. Accessed 2021 Jun 22.
88.British Columbia Ministry of Health. Special authority drug list. 2021; https://www2.gov.bc.ca/gov/content/health/practitioner-professional-resources/pharmacare/prescribers/special-authority#Druglist. Accessed 2021 Jun 25.
89.Special authority request: adalimumab / etanercept / infliximab / risankizumab / secukinumab / ustekinumab / ixekizumab for the treatment of moderate to severe plaque psoriasis. Victoria (BC): British Columbia Ministry of Health; 2021: https://www2.gov.bc.ca/assets/gov/health/forms/5380fil.pdf. Accessed 2021 Jun 21.
90.Part 3 exception drug status (EDS). Winnipeg (MB): Manitoba Health; 2021: https://www.gov.mb.ca/health/mdbif/docs/edsnotice.pdf. Accessed 2021 Aug 18.
91.PEI Pharmacare formulary. Charlottetown (PEI): Government of Prince Edward Island; 2021: https://www.princeedwardisland.ca/sites/default/files/publications/pei_pharmacare_formulary.pdf. Accessed 2021 Jun 22.
92.Government of Ontario. Ontario Drug Benefit formulary search. 2021; https://www.formulary.health.gov.on.ca/formulary/. Accessed 2021 Jun 22.
93.Saskatchewan formulary bulletin #200: update to the 62nd edition of the Saskatchewan formulary. Regina (SK): Government of Saskatchewan; 2021: https://formulary.drugplan.ehealthsask.ca/Bulletins/Bulletin-0200-Jun-2021.pdf. Accessed 2021 Jun 22.
94.Government of Saskatchewan. Saskatchewan drug plan. 2021; https://formulary.drugplan.ehealthsask.ca/SearchFormulary/BG/682840. Accessed 2021 Jun 22.
95.Appendix A: exception drug status program. Regina (SK): Government of Saskatchewan; 2021: https://formulary.drugplan.ehealthsask.ca/PDFs/APPENDIXA.pdf. Accessed 2021 Jun 25.
96.Newfoundland and Labrador Health and Community Services. NLPDP drug product database. 2021; https://www.health.gov.nl.ca/health/prescription/newformulary.asp. Accessed 2021 Aug 18.
97.Government of Yukon. Yukon drug formulary. 2021: https://ihs.gov.yk.ca/drugs/f?p=161:9000. Accessed 2021 Jun 22.
98.Notice – tiered biologics reimbursement policy. Winnipeg (MB): Manitoba Health; 2018: https://www.gov.mb.ca/health/pharmacare/profdocs/notice_tiered_biologics.pdf. Accessed 2021 Jun 25.
99.Tiered biologics reimbursement policy by specialty. Winnipeg (MB): Manitoba Health; 2021: https://www.gov.mb.ca/health/pharmacare/profdocs/tiered_biologics_specialty.pdf. Accessed 2021 Jun 21.
100.Tiered biologics reimbursement policy overview. Winnipeg (MB): Manitoba Health; 2021: https://www.gov.mb.ca/health/pharmacare/profdocs/tiered_biologics_overview.pdf. Accessed 2021 Jun 25.
101.Alignment among public formularies in Canada part 1: general overview. Ottawa (ON): Patented Medicine Prices Review Board; 2019: http://www.pmprb-cepmb.gc.ca/view.asp?ccid=1327#a1. Accessed 2021 Jun 18.
102.Biosimilar biologic drugs in Canada: fact sheet. Ottawa (ON): Health Canada; 2019: https://www.canada.ca/content/dam/hc-sc/migration/hc-sc/dhp-mps/alt_formats/pdf/brgtherap/applic-demande/guides/Fact-Sheet-EN-2019-08-23.pdf. Accessed 2021 Oct 14.
103.Health Canada. Notice of Compliance (NOC) for Omvyence. 2019; https://health-products.canada.ca/noc-ac/info.do?lang=en&no=24929. Accessed 2021 Jul 21.
104.Health Canada. Drug Product Database (DPD) - terminology. 2021; https://www.canada.ca/en/health-canada/services/drugs-health-products/drug-products/drug-product-database/terminology.html. Accessed 2021 Jul 20.
Appendix 1: Regulatory Information
Note that this appendix has not been copy-edited.
Figure 4: Health Canada Approval Timeline for Biologics and Biosimilars by First NOC Date
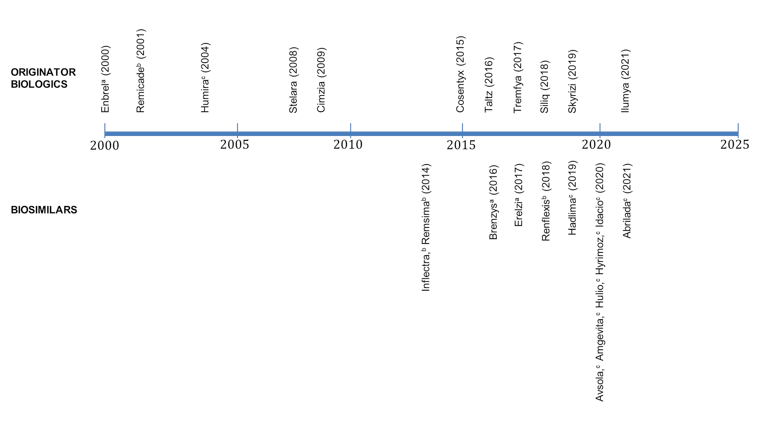
NOC = Notice of Compliance.
Note: Biologics include Enbrel (etanercept), Remicade (infliximab), Humira (adalimumab), Stelara (ustekinumab), Cimzia (Certolizumab pegol), Cosentyx (Secukinumab), Siliq (Brodalumab), Tremfya (Guselkumab), Skyrizi (Risankizumab), Ilumya (Tildrakizumab), and Taltz (Ixekizumab). Biosimilars include Infliximab: Inflectra, Remsima, Renflexis; Etanercept: Erelzi, Brenzys; Adalimumab: Hadlima, Hulio, Hyrimoz, Idacio, Amgevita, Abrilada, and Avsola.
a Etanercept.
b Infliximab.
c Adalimumab.
Table 6: Regulatory Information for Biologics in PsO
Generic name | Brand name | Manufacturer | First NOC date | NOC date for PsO (if different from first NOC date) | Marketed date | Indication for PsO |
|---|---|---|---|---|---|---|
Adalimumab | HUMIRA | AbbVie Corporation | 24 September 2004 | 23 January 2008 | 24 September 2004 | For the treatment of adult patients with chronic moderate-to-severe PsO who are candidates for systemic therapy. For patients with chronic moderate PsO, adalimumab should be used after phototherapy has been shown to be ineffective or inappropriate |
AMGEVITA (biosimilar) | Amgen Canada Inc. | 04 November 2020 | NA | 19 February 2021 | ||
HADLIMA (biosimilar) | Samsung Bioepis Co., Ltd. | 08 May 2018 | NA | 19 February 2021 | ||
HULIO (biosimilar) | BGP Pharma ULC | 24 November 2020 | NA | 16 February 2021 | ||
HYRIMOZ (biosimilar) | Sandoz Canada Inc. | 04 November 2020 | NA | 15 February 2021 | ||
IDACIO (biosimilar) | Fresenius Kabi Canada Ltd. | 30 October 2020 | NA | 16 February 2021 | ||
ABRILDA (biosimilar) | Pfizer Canada ULC | 09 April 2021 | NA | Approved but not marketedb | ||
Certolizumab pegol | CIMZIA | UCB Canada Inc. | 12 August 2009 | 16 August 2018 | 31 August 2009 | For the treatment of adult patients with moderate-to-severe PsO who are candidates for systemic therapy. |
Etanercept | ENBREL | Amgen Canada Inc. | 01 December 2000 | 20 December 2005 | 14 March 2001 | For the treatment of adult patients with chronic moderate-to-severe PsO who are candidates for systemic therapy or phototherapy For the treatment of pediatric patients ages 4 to 17 years with chronic severe PsO who are candidates for systemic therapy or phototherapy. |
BRENZYS (biosimilar) | Samsung Bioepis Co., Ltd. | 31 August 2016 | 19 August 2020 | 23 September 2016 | ||
ERELZI (biosimilar) | Sandoz Canada Inc. | 06 April 2017 | 09 June 2020 | 04 August 2017 | ||
Infliximab | REMICADEa | Janssen Inc. | 06 June 2001 | 07 June 2006 | 14 June 2001 | For the treatment of adult patients with chronic moderate-to-severe PsO who are candidates for systemic therapy. For patients with chronic moderate PsO, infliximab should be used after phototherapy has been shown to be ineffective or inappropriate. |
AVSOLA (biosimilar) | Amgen Canada Inc. | 12 March 2020 | NA | 01 June 2020 | ||
INFLECTRA (biosimilar) | Celltrion Healthcare Co., Ltd. | 15 January 2014 | NA | 04 September 2014 | ||
REMSIMA (biosimilar) | Celltrion Healthcare Co., Ltd. | 15 January 2014 | NA | Approved but not marketedb | ||
RENFLEXIS (biosimilar) | Samsung Bioepis Co., Ltd. | 01 December 2017 | NA | 22 March 2018 | ||
Ustekinumab | STELARA | Janssen Inc. | 12 December 2008 | n/a | 05 January 2009 | For the treatment of adult patients with chronic moderate-to-severe PsO who are candidates for phototherapy or systemic therapy. For the treatment of chronic moderate-to-severe PsO in pediatric patients (children and adolescents) from 6 to 17 years of age, who are inadequately controlled by, or are intolerant to, other systemic therapies or phototherapies. |
Guselkumab | TREMFYA | Janssen Inc. | 10 November 2017 | n/a | 27 November 2017 | For the treatment of adult patients with moderate-to-severe PsO who are candidates for systemic therapy or phototherapy. |
TREMFYA One-Press | Janssen Inc. | 18 April 2019 | n/a | 20 May 2019 | ||
Risankizumab | SKYRIZI | AbbVie Corporation | 17 April 2019 | n/a | 13 May 2019 | For the treatment of adult patients with moderate-to-severe PsO who are candidates for systemic therapy or phototherapy. |
Tildrakizumab | ILUMYA | Sun Pharma Global FZE | 19 May 2021 | n/a | Approved but not marketedb | for the treatment of adult patients with moderate to-severe PsO who are candidates for systemic therapy or phototherapy. |
Brodalumab | SILIQ | Bausch Health, Canada Inc. | 06 March 2018 | n/a | 05 July 2018 | For the treatment of moderate-to-severe PsO in adult patients who are candidates for systemic therapy or phototherapy. |
Ixekizumab | TALTZ | Eli Lilly Canada Inc. | 25 May 2016 | 01 April 2021 (pediatric indication) | 11 August 2016 | For the treatment of adult patients with moderate-to-severe PsO who are candidates for systemic therapy or phototherapy. For the treatment of pediatric patients from 6 to less than 18 years of age with moderate-to-severe PsO who are candidates for systemic therapy or phototherapy. |
Secukinumab | COSENTYX | Novartis Pharmaceuticals Canada Inc. | 17 February 2015 | 20 January 2021 (pediatric indication) | 10 April 2015 | For the treatment of moderate-to-severe PsO in adult patients who are candidates for systemic therapy or phototherapy. For the treatment of severe PsO in pediatric patients 12 to less than 18 years of age who are candidates for systemic therapy or phototherapy and have a body weight ≥ 50 kg. |
NA = not applicable; NOC = Notice of Compliance; PsO = plaque psoriasis.
Note: Shaded cells indicate biosimilar versions.
aOmvyence (infliximab, Janssen Inc.) was approved on 29 December 2020 by Health Canada. This product is not a biosimilar. The NOC submission was made for an ‘Additional Product Name’ for Remicade. The product is approved by Health Canada but not yet marketed in Canada.24,103
b“Approved” refers to an active Drug Identification number (DIN) for a product that has been reviewed and authorized for sale in Canada but has not yet been marketed in Canada. ‘Marketed’ refers to an active DIN for a product that is currently being sold in Canada.104
Appendix 2: CADTH Reviews of Biologics in PsO
Table 7: Summary of CDEC Recommendations for Biologics in PsO
Drug recommendation | Clinical rationale | Economic rationale | |
|---|---|---|---|
Study details | Clinical reason for recommendation | ||
HUMIRA (adalimumab) Recommended to be listed for patients with severe, debilitating psoriasis who meet all of the following criteria 1. BSA > 10% and/ or significant involvement of the face, hands, feet or genital regions; 2. Failure to respond to, contraindication to, or intolerant of MTX and cyclosporine; 3. Failure to respond to, intolerant to, or unable to access phototherapy. Response to adalimumab must be assessed after 16 weeks, and therapy continued only in patients who have responded to therapy. Potential criteria for defining response are achievement of a ≥ 75% reduction in PASI score, or ≥ 50% reduction in PASI with a ≥ 5 point improvement in the DLQI or a significant reduction in BSA involved, with consideration of important regions such as face, hands, feet, or genital region. Ongoing coverage for adalimumab maintenance therapy should only be provided for responders, as noted above, and for a dose not exceeding 40 mg every 2 weeks | Three phase III RCTs [N = 1,232] RCT 1: double-blind, placebo-controlled (patients naive to anti-TNF agents) RCT 2: double-blind, placebo-controlled (also included patient who had previously used biologic therapy) RCT 3: double-blind, placebo (patients naive to MTX and anti-TNF agents) and active comparator (methotrexate) For all 3 RCTs Primary Outcome: Proportion of patients achieving ≥ 75% reduction in PASI score, measured at 12 to 16 weeks of therapy. Population: Adult patients with moderate-to-severe PsO or active psoriasis despite receiving topical therapies. | Adalimumab vs. placebo: Adalimumab achieved statistically significantly higher PASI 75 and PASI 100 response rated compared to placebo and MTX and improved measure of QoL. Results of a subgroup analysis showed that the PASI 75 and PASI 100 response rates were similar in the group that had received recent prior systemic therapies and phototherapy (received in the last prior 12 months), compared with those who had not received prior systemic therapies and phototherapy. | At recommended maintenance doses, the annual cost of adalimumab ($18,191) is lower than etanercept ($20,377), efalizumab ($21,420), infliximab ($25,220) and alefacept (29,976). |
CIMZIA (Certolizumab pegol) Recommended to be reimbursed for the treatment of adult patients with moderate-to-severe PsO who are candidates for systemic therapy if the following conditions are met: Initiation Criteria: Adult patients with moderate-to-severe PsO who are candidates for systemic therapy. Discontinuation Criteria: Treatment should be discontinued if a response to treatment with certolizumab pegol has not been demonstrated by 16 weeks. Response to treatment defined as achievement of at least a 75% reduction in PASI 75. Prescribing Conditions: Patient should be under the care of a dermatologist. Pricing Conditions: The drug plan cost of treatment with certolizumab pegol should result in cost-savings compared with the drug plan cost of treatment with the least costly alternative biologic therapy reimbursed for the treatment of moderate-to-severe PsO. | Three phase III RCTs CIMPASI [N = 234] and CIMPASI-2 [N = 227]: multi-centre, double-blind placebo-controlled initial treatment period, followed by a dose-blind maintenance treatment period. Co-primary outcomes: Proportion of patients achieving ≥ 75% reduction in PASI score, and PGA responder rates (clear or almost clear, with at least 2-category improvement), measured at 16 weeks of therapy. CIMPACT [N = 559]: multi-centre, double-blind, parallel-group RCT with a double-blind placebo but open-label active-controlled (etanercept) initial treatment period, followed by a double-blind placebo-controlled maintenance period. Primary Outcome: Proportion of patients achieving ≥ 75% reduction in PASI score, measured at week 12. (due to comparison with etanercept) For all 3 RCTs Population: moderate-to-severe PsO, defined as BSA ≥ 10% or more, PASI ≥ 12, and static PGA ≥ 3 | Certolizumab vs. placebo: Certolizumab was associated with a statistically significant and clinically meaningful improvement in skin clearance (PASI 75) and PGA response (clear or almost clear; i.e., score of 0 or 1) compared with placebo in the induction period (12 or 16 weeks). Indirect treatment comparison (ITC) Results suggest that use of certolizumab is associated with superior PASI responses to placebo over short-term induction (16 week) treatment periods. However, with the exception of etanercept, there is no direct evidence comparing CZP with other available biologics. No evidence to suggest that certolizumab has any therapeutic advantage compared with other treatments that are currently reimbursed to treat moderate-to-severe PsO; therefore, certolizumab does not address any unmet need that is not currently met by other available treatments. | At the submitted price of $664.51 per pre-filled syringe or autoinjector, the annual cost of certolizumab is $19,271 to $34,555 in the first year and $17,277 to $34,555 thereafter. Based on ITC, etanercept was associated with the lowest cost and lowest QALYs, followed by brodalumab, infliximab and then guselkumab. Certolizumab 200 mg and 400 mg were dominated by infliximab and ixekizumab, respectively. At a WTP threshold of $50,000 per QALY gained, certolizumab 200 mg and 400 mg had 0% probability of being cost-effective.a |
INFLECTRA (infliximab, biosimilar) Recommended to be reimbursed in accordance with the Health Canada–approved indications for the treatment of PsO, if the following condition are met:
| Two RCTs PLANET-RA [N = 606] phase III, double-blind, multi-centre, multinational, parallel-group clinical equivalence study; comparing efficacy and safety of Inflectra with Remicade Population: patients with active RA who had an inadequate response to treatment with MTX Primary end point: Proportion of patients with an ACR 20 response at week 30. Therapeutic equivalence of clinical response according to ACR20 criteria would be demonstrated if the 95% CI for the treatment difference was within ± 15%. PLANET-AS [N = 250] was a phase I, randomized, double-blind, multi-centre, multinational, parallel-group study designed to compare the PK, safety, and efficacy of Inflectra and Remicade. Population: patients with active AS. Primary end point: demonstrate PK equivalence at a steady state of area under the concentration-time curve and observed maximum steady state serum concentration between Inflectra and Remicade between weeks 22 and 30. Equivalence was demonstrated if the 90% CIs lay within the equivalence margin of 80% to 125%. | Inflectra vs. Remicade Inflectra and Remicade have similar efficacy, safety, and PK profiles in patients with RA and AS. Extrapolation of the data from RA and AS to PsO is supported by the similar pathophysiology of these conditions and the identical dosage regimen for infliximab for these indications. | At the submitted price, for PsO, for the first year of treatment (using Saskatchewan Formulary costs), Inflectra (5 mg/kg, $20,800) is less costly than Remicade (5 mg/kg, $31,232), etanercept ($25,001 to $25,008), and ustekinumab ($22,966), but is more costly than adalimumab ($20,730). |
RENFLEXIS (infliximab, biosimilar) Recommended to be reimbursed in accordance with the Health Canada–approved indications for the treatment of PsO (among other indication), if the following criterion and condition are met: Criterion:
Condition:
| Two RCTs Study SB2-G11-NHV [N = 159], phase I 3-arm, single-blind study, comparing Renflexis with EU-Remicade and US-Remicade. Primary end point: PK profile (AUCinf, AUClast, Cmax), considered to be met if the 90% CI for the geometric mean was within the equivalence margin of 80% to 125% Study SB2-G31-RA [N = 584], phase III double-blind, multinational study, comparing efficacy, safety, immunogenicity, and PK of Renflexis with EU-Remicade Primary end point: ACR 20 response at Week 30. The primary end point was considered met if the 95% CI of the adjusted treatment difference was within the equivalence margin of −15% to 15%. For both RCTs Population: patients with moderate-to-severe RA despite MTX therapy | Renflexis vs. Remicade Renflexis has similar PK, efficacy, safety, and immunogenicity as the reference product, Remicade. Extrapolation of the data from RA to PsO is reasonable given the demonstrated similarities between Renflexis and Remicade in the included trials, and the role that tumour necrosis factor alpha (TNF-alpha) drugs play in these indications. | At a submitted price ($525.00 per 100 mg vial of lyophilized powder)), Renflexis is less costly than Remicade based on the OPDP price ($987.56 per 100 mg vial of lyophilized powder) for use in accordance with the Health Canada–approved indications. Renflexis is the same price as Inflectra (infliximab biosimilar), based on the OPDP price ($525.00 per 100 mg vial of lyophilized powder). |
STELARA (ustekinumab) Recommended to be listed for patients with severe, debilitating psoriasis who meet all of the following criteria:
Ustekinumab 45 mg should initially be given at weeks 0, 4 and 16. Response must be assessed before a fourth dose and further doses provided only for responders. Potential criteria for defining response are achievement of a ≥ 75% reduction in PASI score, or ≥ 50% reduction in PASI with a ≥ 5 point improvement in the DLQI or a significant reduction in BSA involved, with consideration of important regions such as face, hands, feet, or genital region | Three phase III RCTs [N = 2,899] PHOENIX 1: double blind, placebo-controlled PHOENIX 2: double blind, placebo-controlled ACCEPT: open-label, accessor blinded, active-controlled (etanercept) For all 3 RCTs Primary outcome: Proportion of patients achieving ≥ 75% reduction in PASI score, measured at 12 weeks of therapy. Population: chronic moderate-to-severe PsO | Ustekinumab vs. placebo Two double-blind RCTs ustekinumab achieved statistically significantly higher PASI 75 and PASI 100 response rated and improved measures of quality of life compared with placebo. Ustekinumab vs. etanercept In 1 RCT ustekinumab achieved a statistically significantly higher PASI 75 and PASI 100 response rates compared with etanercept In 2 RCTS patients were assessed after a third dose and all non-responders were discontinued before receiving a fourth dose. There is no data from trials on how non-responders after a third dose respond to a fourth dose. | At the submitted price of $4,200 per vial, 6 doses per year, in the first year, the annual cost of ustekinumab is $25,200; cost of treatment in subsequent years ranges from $16,800 to $21,000 (for 4 to 5 doses per year), which is similar to etanercept ($25,134 first year, $20,421 thereafter) and adalimumab ($18,574 first year, $17,887 thereafter), less expensive compared to infliximab ($40,740 first year, $25,220 thereafter), and more expensive compared to alefacept after the fist year ($29, 976 first year, $14,988 thereafter). |
TREMFYA (guselkumab) Recommended to be reimbursed for adult patients with moderate-to-severe PsO who are candidates for systemic therapy or phototherapy, with the following criteria and condition: Clinical Criteria:
Conditions:
| Three phase III RCTs VOYAGE-1 [N = 837] and VOYAGE-2 [N = 992]: multicentre, double-blind, parallel-group, placebo and active (adalimumab) controlled trials Co-primary outcome: Proportion of patients achieving an IGA score of 0 (cleared) or 1 (minimal) disease and the proportion of patients achieving PASI 90 response. Non-inferiority and superiority of guselkumab to adalimumab evaluated (secondary outcome) measured at 16 weeks of therapy. NAVIGATE [N = 268]): multicentre, double-blind, parallel-group, active (ustekinumab) controlled trial Primary outcome: number of visits during which patients achieved an IGA score of 0 or 1 and ≥ 2 grade improvement from week 16 during weeks 28 to week 40 of therapy. For all 3 RCTs Population: adults with moderate-to-severe PsO | Guselkumab vs. placebo guselkumab achieved statistically and clinically significant improvements in HRQoL as measured by the DLQI compared with placebo after 16 weeks of treatment. Guselkumab vs. placebo and adalimumab Guselkumab was shown to be superior to placebo and to adalimumab in achieving a higher proportion of patients with an IGA score of 0 or 1 (i.e., cleared or minimal disease) and a PASI 90 response at week 16. The safety profile of guselkumab was similar to that of adalimumab. Guselkumab vs. ustekinumab Patients with an inadequate response to ustekinumab, who were switched to guselkumab, had a statistically significantly higher number of health care provider visits in which they achieved an IGA score of 0 or 1 and at least a 2-grade improvement compared with patients who continued ustekinumab. Results of the indirect comparison is confidential. | Guselkumab is available as a 100 mg/mL pre-filled syringe at a price of $3,060. At the recommended dose of 100 mg to be given as a SC injection at week 0 and week 4, followed by maintenance dosing every 8 weeks thereafter, the first-year cost of guselkumab is $21,418 with the price changing to $19,943 annually thereafter. At the submitted price guselkumab was not cost-effective treatment option for moderate-to-severe PsO. Guselkumab was dominated by ixekizumab, and the incremental cost per QALY gained for guselkumab vs. infliximab was $1.6 million. The probability that guselkumab was cost-effective at a WTP threshold of $50,000 per QALY was 11.9%.a |
SKYRIZI (risankizumab) Recommended to be listed for the treatment of adult patients with moderate-to-severe PsO who are candidates for systemic therapy or phototherapy if the following conditions are met: Conditions for Reimbursement
| Four phase III RCTS UltIMMA-1 [N = 506]; and UltIMMA-2 [N = 491]: multi-centre, double-blind, double-dummy, placebo- and active comparator (ustekinumab) -controlled studies. IMMhance [N = 507]: multi-centre, double-blind, placebo-controlled trial. IMMvent [N = 605]: multi-centre, double-blind, double-dummy, active-controlled (adalimumab) For all 4 RCTs Co-primary outcomes: PASI 90 and sPGA of clear or almost clear, measured at 16 weeks of therapy. Population: moderate-to-severe PsO (defined as BSA ≥ 10%, PASI ≥ 12, and sPGA] ≥ 3). Patients were randomized to double-blind treatment in blocks and stratified by body weight (≤ 100 kg vs. > 100 kg) and prior exposure to anti-TNF. | Risankizumab vs. placebo, ustekinumab, and adalimumab Risankizumab was associated with a statistically significant and clinically meaningful improvement in skin clearance (PASI 90) and HRQoL compared with placebo, ustekinumab, and adalimumab in the induction period (16 weeks). In UltIMMA-1 and UltIMMA-2, the benefit of risankizumab over ustekinumab for PASI 90 was maintained up to week 52. In the IMMvent trial, patients who did not exhibit an adequate response to adalimumab during the induction period had a higher rate of achieving PASI 90 response after switching to risankizumab for 28 weeks, compared with those continuing adalimumab. Risankizumab vs. other biologics Two ITCs suggest that use of risankizumab is associated with similar PASI 75 and PASI 90 responses to ixekizumab, brodalumab, and guselkumab, and was superior to placebo, apremilast, etanercept, adalimumab, ustekinumab, infliximab, and secukinumab in patients with moderate-to-severe chronic PsO over short-term induction (10 to 16 weeks) treatment periods. | At the submitted price of $2,467.50 per pre-filled syringe, the annual treatment cost per patient is $24,675 in the first year and $21,385 in all subsequent years. At the submitted price, risankizumab is not cost-effective at a WTP threshold of $50,000 per QALY. Given the uncertainty regarding the comparative efficacy of risankizumab compared with other biologics that may be reimbursed for the treatment of moderate-to-severe PsO and the limitations of the cost-utility analysis model, there is insufficient evidence to justify a cost premium over the least expensive biologic drug reimbursed for the treatment of moderate-to-severe PsO. Risankizumab was more effective and more costly than etanercept and brodalumab — resulting in an ICUR for risankizumab of $2,370,521 |
However, there is no direct evidence comparing risankizumab with the other available interleukin (IL)-23 inhibitor, guselkumab, or any of the 3 available IL-17 inhibitors (brodalumab, secukinumab, ixekizumab). Furthermore, the relative efficacy and safety of risankizumab in comparison with biologics other than ustekinumab and adalimumab beyond a short-term induction period remains unknown. Finally, HRQoL was not evaluated in the ITCs. | per QALY compared with brodalumab. At a WTP threshold of $50,000 per QALY, the price of risankizumab would need to be reduced by at least 26% to be considered cost-effective.a,b | ||
ILUMYA (tildrakizumab) Recommended to be reimbursed for the treatment of adult patients with moderate-to-severe PsO who are candidates for systemic therapy or phototherapy if the following conditions are met: Initiation: Eligibility for tildrakizumab should be based on the criteria used by each of the public drug plans for reimbursement of other biologics for moderate-to-severe PsO. Renewal: Treatment with tildrakizumab may be renewed for patients who exhibit a response to treatment after 12 to 16 weeks. A response to treatment is defined as an achievement of at least a 75% reduction in the PASI score (PASI 75). Prescribing:
Pricing: The drug plan cost of tildrakizumab should not exceed the drug plan cost of treatment with the least costly biologic therapy reimbursed for the treatment of moderate-to-severe PsO. | Two phase III RCT P010 or reSURFACE 1 [N = 772]: multicentre double-blind, placebo -controlled P011 or reSURFACE 2 [N = 1,090]: multicentre double-blind, placebo- and active (etanercept)-controlled For both RCTs Co-primary outcomes: Proportion of patients who achieved at least a 75% improvement in the PASI score from baseline to week 12, and the proportion of patients with a PGA score of clear or minimal with at least a 2-grade reduction from baseline for tildrakizumab 200 mg and 100 mg doses vs. placebo. Population: adults with moderate-to-severe PsO who are candidates for phototherapy or systemic therapy. | Tildrakizumab vs. placebo: Tildrakizumab was associated with statistically significant and clinically meaningful improvements vs. placebo at week 12 for the co-primary outcomes: the PASI 75 and the PGA response. A significantly higher percentage of patients responded to treatment with tildrakizumab (difference in PASI 75 response at week 12: 58% [95% CI, 51% to 64%] and 56% [95% CI, 48% to 62%] in Study P010 and Study P011, respectively; P < 0.001 for each), compared with placebo. The difference in the proportion of patients who exhibited a PGA score of clear or minimal with at least a 2-grade reduction from baseline at week 12 was 51% (95% CI, 44% to 57%; P < 0.001) in Study P010 and 50% (95% CI, 43% to 57%; P < 0.001) in Study P011. Tildrakizumab vs. etanercept: Study P011 included direct evidence for tildrakizumab compared with etanercept. There was no statistically significant difference between tildrakizumab 100 mg and etanercept (absolute difference in proportions: 7%; 95% CI, –0.5% to 15%; P = 0.066) in the PGA score at week 12. | At the submitted price of $4,935 per single-dose pre-filled 100 mg syringe of tildrakizumab, the expected annual cost of treatment with tildrakizumab is $24,675 per patient in the first year, and $21,385 in subsequent years. Based on publicly available prices of other products (annual per patient cost ranging from $16,023 to $39,080), the annual cost of tildrakizumab is higher than some of the other biologic products that are currently reimbursed by public drug plans but is lower than some other biologic treatments. Tildrakizumab is not cost-effective at WTP of $50,000 per QALY. Several biologic drugs provide better efficacy in terms of response at a lower total cost. For example, adalimumab, brodalumab, and infliximab have a better efficacy than tildrakizumab, at a lower total cost. |
Tildrakizumab vs. other biologics: The network meta-analysis (NMA) submitted by the sponsor suggested that tildrakizumab may be less effective than interleukin (IL)- 17 inhibitors (ixekizumab, brodalumab, secukinumab), other IL-23 inhibitors (guselkumab, risankizumab), and infliximab, but may be more effective than etanercept or apremilast. Tildrakizumab appeared to be similar to adalimumab, certolizumab, and ustekinumab | A price reduction of at least 20% would be required for tildrakizumab to be cost-effective at a WTP threshold of $50,000 per QALY. | ||
SILIQ (brodalumab) Recommended to be reimbursed for the treatment of moderate-to-severe PsO in adult patients who are candidates for systemic therapy or phototherapy, if the following criteria and conditions are met: Criteria
Condition
| Three phase III RCT AMAGINE-1 [N = 661]: double-blind, parallel-group, placebo-controlled AMAGINE-2 [N = 1,831]: double-blind, parallel-group, double-dummy, active-controlled (ustekinumab) trials AMAGINE-3 [N = 1,881]: double-blind, parallel-group, double-dummy, active-controlled (ustekinumab) trials For all 3 RCTs Primary outcome: Proportion of patients with PASI 75 and sPGA success (score of 0 or 1), measured at week 12 For AMAGINE-2 and -3 Primary outcome: Superiority of brodalumab to ustekinumab measured as proportion of patients with PASI 100 at week 12 of therapy. For all 3 RCTs Population: adults with moderate-to-severe PsO | Brodalumab vs. placebo: In AMAGINE-1, AMAGINE-2, and AMAGINE-3, brodalumab demonstrated statistically significant and clinically important improvements in skin clearance and dermatological symptoms (measured by the PASI and sPGA) over the short-term induction phase (12 weeks) compared with placebo. Brodalumab vs. placebo At 12 weeks, brodalumab was statistically superior to ustekinumab in achieving a PASI 100 response in, AMAGINE-2, and AMAGINE-3 and a PASI 75 response in AMAGINE-3. The Health Canada–approved product monograph states that discontinuation of therapy with brodalumab should be considered if an adequate response has not been achieved after 12 weeks to 16 weeks of treatment because continuing treatment beyond 16 weeks in patients who have not achieved an adequate response is not likely to result in greater success. Limitations in trial design in the AMAGINE clinical trial program, as well as the short duration of the trials in the context of proposed lifelong treatment, lead to uncertainty regarding the long-term clinical safety and effectiveness of brodalumab compared with ustekinumab. | At the submitted price, brodalumab (210 mg at weeks 0, 1, and 2, and every 2 weeks thereafter; $18,060 in year 1, $16,770 subsequent years) is less costly than biologics for the treatment of PsO based on publicly available list prices. Brodalumab was more effective and costly compared with adalimumab, resulting in an incremental cost per QALY gained for BDL of $42,981. At a WTP threshold of $50,000 per QALY gained, brodalumab had a 70.5% probability of being cost-effectivea,b However, because of the lack of comparative effectiveness data vs. all relevant biologic treatments and the inability of CDR to consider the confidential negotiated prices of other treatments, the true cost-effectiveness of brodalumab is uncertain and likely less favourable than the values estimated using publicly available prices. |
There is no direct evidence related to the comparative efficacy and safety of brodalumab vs. other biologic therapies, and the available indirect treatment comparisons (ITCs) are associated with substantial uncertainty due to heterogeneity of the included trials, particularly due to large differences in the placebo response. Therefore, no evidence is available to clearly support a higher cost for brodalumab compared with other biologic therapies. | |||
TALTZ (ixekizumab) Recommended to be reimbursed for patients with moderate-to-severe PsO with the following criteria and condition. Criteria
Condition
| Three phase III RCTs UNCOVER-1 [N = 1,296] double-blind, placebo- controlled UNCOVER-2 [N = 1,224] double-blind, placebo-controlled and active -controlled (etanercept) UNCOVER-3 [N = 1,346] double-blind, placebo-controlled, and active-controlled (etanercept) For all 3 RCTs Primary Outcome: Proportions of patients with at least a 2-point improvement in the sPGA, with a score of 0 or 1; and at least a PASI 75 score. Population: patients with moderate-to-severe PsO | Ixekizumab vs. placebo: UNCOVER-1, UNCOVER-2, and UNCOVER-3 demonstrated that ixekizumab is superior to placebo in improving PGA and PASI 75 scores after 12 weeks of treatment, and ixekizumab was associated with improvements in HRQoL and function compared with placebo in each of these studies. Ixekizumab vs. etanercept UNCOVER-2 and UNCOVER-3 demonstrated that ixekizumab is superior to etanercept improving PGA and PASI 75 scores after 12 weeks of treatment. The safety profile of ixekizumab is similar to that of etanercept. Ixekizumab vs. other biologics Indirect comparison suggests that ixekizumab is at least as efficacious in treating moderate-to-severe PsO as other interleukin inhibitors (specifically, secukinumab and ustekinumab) and TNF inhibitors, with no consistent differences between the safety profile of ixekizumab and the other drugs. | At the submitted price for ixekizumab of $1,519 per 80 mg/1 mL prefilled pen or syringe, and the recommended dose of 160 mg at week 0, 80 mg every 2 weeks until week 12, and 80 mg every 4 weeks thereafter, ixekizumab costs $27,342 in the first year and $19,747 in subsequent years. SEB infliximab remained the most cost-effective option when all comparators were assessed simultaneously, and the CDR base-case ICURs for ixekizumab vs. SEB infliximab were $360,307 and $393,762 per QALY for the mixed and biologic-experienced populations, respectively. Depending upon prior biologic experience, price reductions of 22% to 24% are required for the ICUR to fall below $100,000 per QALY vs. SEB infliximab, and 27% to 28% for the ICUR to fall below $50,000 per QALY.a Therefore, at the submitted price of $1,519 per 80 mg/1 mL prefilled pen or prefilled syringe, ixekizumab is not considered to be a cost-effective treatment option for PsO. |
COSENTYX (secukinumab) Recommended to be listed for the treatment of moderate-to-severe PsO in adult patients who are candidates for systemic therapy or phototherapy, if the following clinical criterion and condition are met: Clinical criterion
Condition:
| Four phase III RCTS
For all 4 RCTs Primary Outcomes: PASI 75 response and IGA mod 2011 0 or 1 response at week 12. Population: Adult patients with moderate-to-severe chronic PsO, inadequately controlled by topical treatments, phototherapy, or previous systemic therapy | Secukinumab vs. placebo Secukinumab 300 mg was superior to placebo for the proportion of patients achieving a PASI 75 response. Secukinumab vs. etanercept Secukinumab 300 mg was superior to etanercept 50 mg twice weekly for the proportion of patients achieving a PASI 75 response. | ICUR for secukinumab compared with SoC ranges from $82,534 to $122,365 per QALY. a Based on current list prices for comparators, secukinumab 300 mg is more costly in the first year than adalimumab and ustekinumab ($20,730 and $22,966, respectively) and less costly than etanercept, and infliximab-Remicade and SEB-infliximab (range: $25,297 to $39,502). In subsequent years, secukinumab 300 mg is less costly than other biologics (range: $19,249 to $32,096) Note: The manufacturer submitted a reduced price during the embargo period (price information is confidential) for 2 150 mg/1.0 mL pre-filled syringes or pens, a (% reduction is confidential) price reduction from the original submitted price of $1,645. |
ACR = American College of Rheumatology; AS = ankylosing spondylitis; BSA = body surface area; CDR = CADTH Common Drug Review; CI = Confidence Interval; DLQI = Dermatology Life Quality Index; HRQoL = health-related quality of life; IGA = Investigator Global Assessment; ITC = Indirect treatment comparisons; IUCR = incremental cost-utility ratio; MTX = methotrexate; OPDP = Ontario Public Drug Plan; PASI = Psoriasis Area Severity Index; PGA = Physician Global Assessment; PK = pharmacokinetic; PsO = plaque psoriasis; QALY = quality-adjusted life-year; RA = rheumatoid arthritis; RCT = randomized controlled trials; SC = subcutaneous; SEB = subsequent entry biologic SoC = Standard of Care; sPGA = static Physician Global Assessment; TNF = tumour necrosis factor; WTP = willingness-to-pay.
a Based on CDR’s reanalyses to account for limitations in the manufacturer’s economic model.
bThe results should be interested with caution as CADTH reanalysis was not able to address all the limitations of the manufacturer submitted economic analysis.
Note that this table has not been copy-edited.
Appendix 3: Coverage Criteria for Biologics in PsO
Table 8: Summary Coverage Criteria for Originators and Biosimilars Across Public Drug Plans in PsO
Drug plan | Drugs covered | Initial coverage criteria | Renewal criteria | Notes |
|---|---|---|---|---|
AB | Adalimumab Etanercept Infliximab Ustekinumab Risankizumab Ixekizumab | FOR adalimumab, etanercept, Infliximab, ustekinumab, Risankizumab and Ixekizumab
FOR Ustekinumab ONLY
FOR Risankizumab and Ixekizumab ONLY
| Confirmation of response required including
For ongoing coverage, a reassessment every 12 months is required confirming response to therapy (See Renewal Criteria above) |
Relevant biosimilar policy:
|
BC | Adalimumab Etanercept Infliximab Ustekinumab Risankizumab Ixekizumab Secukinumab |
| First Renewal after the initial trial of biologic: Achievement of PASI > 75 from the baseline biologic naive PASI score Subsequent Renewals for Maintenance Therapy: Maintenance of PASI > 50 from the baseline biologic naive PASI score. | Switching to other biologics
Relevant biosimilar policy:
|
SK | Adalimumab Etanercept Infliximab Ustekinumab Risankizumab Brodalumab Ixekizumab Secukinumab |
| Confirmation of response required. | For infliximab ONLY:
Relevant biosimilar policy:
|
MB | Adalimumab Etanercept Infliximab Ustekinumab Risankizumab Brodalumab Ixekizumab Secukinumab |
Manitoba tiered biologics policy (new patients): Patients must fail to respond to more than 2 Tier 1 drugs to be eligible for coverage for Tier 2 drugs. Tier 1 drugs include biosimilars versions of infliximab (Avsola, Inflectra, Renflexis), and etanercept (Erelzi and Brenzys), and originator biologics Cosentyx (secukinumab) Humira (adalimumab) Siliq (brodalumab) Skyrizi (risankizumab) and Taltz (ixekizumab). Tier 2 drugs include originator biologics Enbrel (etanercept) Remicade (infliximab) Stelara (ustekinumab) | Confirmation of response required including
| For adalimumab, ONLY: Treatment should be discontinued if there is no improvement after 12 weeks of treatment Relevant biosimilar policy: Etanercept- and infliximab-naive patients were approved for coverage of biosimilar versions, only. Patients stable on the reference biologics continue to be approved for the coverage of the originator biologic, with the option to switch to a biosimilar version. If previously trialled and deemed unresponsive to therapy, patients are not permitted to switch from 1 biosimilar to another (of the same drug) or between a biosimilar and a biologics (of the same drug). Switching between originator biologics and biosimilar products is not permitted, if a patient was previously trialled on the originator drug or biosimilar and was deemed unresponsive to therapy.81,96,98 SK does not consider infliximab biosimilars and biologics to be interchangeable. |
ON | Adalimumab Etanercept Infliximab Ustekinumab Risankizumab Brodalumab Ixekizumab Secukinumab |
| Confirmation of response required (after 3 months):
| Treatment should be discontinued if there is no improvement after 12 weeks of treatment. If the patient has not responded adequately after 12 weeks of treatment at the Health Canada–approved dose, higher doses are not recommended, and the physician should consider switching to an alternative biologic agent. |
NB | Adalimumab Etanercept Infliximab Ustekinumab Risankizumab Brodalumab Ixekizumab Secukinumab | For Adalimumab ONLY Refractory, intolerant or have contraindications to conventional therapy. For etanercept, infliximab, ustekinumab, risankizumab, brodalumab, ixekizumab, secukinumab
| Confirmation of continued response required. | Combined use of more than 1 biologic drug will not be reimbursed Relevant biosimilar policy: Controlled switch policy of ALL patients to biosimilar version for adalimumab, infliximab and etanercept. Adult patients currently on the originator drug for the treatment of POS must switch to the biosimilar prior the expiry of their current SA approval, or November 30, 2021, whichever occurs first, to maintain coverage. For patients who are unable to switch for medical reasons, an SA request for exceptional coverage of the originator biologic may be submitted. Exceptional requests are reviewed on a case-by-case basis. |
NS | Adalimumab Etanercept Infliximab Ustekinumab Risankizumab Brodalumab Ixekizumab Secukinumab |
| Confirmation of response required including
| Concurrent use of biologics not reimbursed For adalimumab, etanercept, infliximab, risankizumab, brodalumab and ixekizumab ONLY: Treatment should be discontinued if there is no improvement after 12 weeks of treatment Relevant biosimilar policy: Infliximab-naive patients were approved for coverage of biosimilar versions, only. Patients stable on the reference biologics continue to be approved for the coverage of the originator biologic, with the option to switch to a biosimilar version. |
NL | Adalimumab Etanercept Infliximab Ustekinumab Risankizumab Brodalumab Ixekizumab Secukinumab |
| Confirmation of response required including
| Combined use of more than 1 biologic DMARD will not be reimbursed For infliximab ONLY: Patients will not be permitted to switch from Inflectra, Renflexis or Avsola to another infliximab product or vice versa, if previously trialled and deemed unresponsive to therapy. Relevant biosimilar policy: Etanercept- and infliximab- and adalimumab-naive patients were approved for coverage of biosimilar versions, only. Patients stable on the reference biologics continue to be approved for the coverage of the originator biologic, with the option to switch to a biosimilar version. If previously trialled and deemed unresponsive to therapy, patients are not permitted to switch from 1 biosimilar to another (of the same drug) or between a biosimilar and a biologics (of the same drug). |
PE | Adalimumab Etanercept Infliximab Ustekinumab Risankizumab Brodalumab Ixekizumab Secukinumab |
| Confirmation of continued response is required. | Combined use of more than 1 biologic DMARD will not be reimbursed. |
YT | Adalimumab Etanercept Infliximab Ustekinumab Risankizumab Secukinumab |
| FOR adalimumab, etanercept, ustekinumab, risankizumab and secukinumab Confirmation of response (yearly) required
Information not available for infliximab | Relevant biosimilar policy:
|
NIHB | Adalimumab Etanercept Infliximab Ustekinumab Risankizumab Brodalumab Ixekizumab Secukinumab |
| FOR adalimumab, ustekinumab, brodalumab, ixekizumab, and secukinumab ONLY Confirmation of response required including
Information not available infliximab and risankizumab | — |
CSC | Adalimumab Ustekinumab |
CSC’s Tiered Biologics Policy (existing patients): Patients deemed unresponsive to biologic therapy are required to switch to biosimilar/biologic therapy on a CSC defined list (Tier 1). Once all options on Tier 1 are exhausted, options on Tier 2 list can be made available. Tier 1 drugs include biosimilars versions of etanercept (Brenzys, Erelzi), adalimumab (Hadlima, Hyrimoz, Idacio) and infliximab (Inflectra, Renflexis). Tier 2 drugs include originator biologics Stelara (ustekinumab) and Humira (adalimumab). | For adalimumab and ustekinumab ONLY Confirmation of continued response is required
For etanercept and infliximab ONLY Confirmation of continued response (after 3 months) required including
| Relevant biosimilar policy: Etanercept- and infliximab- and adalimumab-naive patients were approved for coverage of biosimilar versions, only. Patients stable on the reference biologics continue to be approved for the coverage of the originator biologic, with the option to switch to a biosimilar version. If due to exceptional individual circumstances, it is deemed that a patient initiating treatment must receive the biologic (reference) drug; this request may be made via the Non-Formulary Request Process with accompanying rationale. |
CAF | Adalimumab Etanercept Infliximab Ustekinumab Brodalumab Ixekizumab Secukinumab |
| Information not available- | Relevant biosimilar policy: Infliximab-naive patients were approved for coverage of biosimilar versions, only. Patients stable on the reference biologics continue to be approved for the coverage of the originator biologic, with the option to switch to a biosimilar version. |
VACk | Adalimumab Etanercept Infliximab Ixekizumab Risankizumab Ustekinumab |
| Confirmation of continued response is required
| VAC encourages lower cost biosimilar use for naive-patients. However, the originator biologic will be approved if rationale is provided by the prescriber as to why the originator biologic should be used instead of the lower cost biosimilar. For treatment-experienced patients, the VAC SA criteria allow for the continuation with the originator biologic. The VAC SA criteria for biosimilars does not currently include mandatory switching. |
AB = Alberta; BC = British Columbia; BSA = body surface area; CAF = Canadian Armed Forces; CSC = Correctional Services Canada; DLQI = Dermatology Life Quality Index; MB = Manitoba; MTX = methotrexate; NB = New Brunswick; NIHB = Non-insured health benefit; NL = Newfoundland and Labrador; NS = Nova Scotia; PASI = Psoriasis Area Severity Index; PsO = plaque psoriasis; SA = Special Authorization; SK = Saskatchewan; VAC = Veterans Affairs Canada; YT = Yukon
aPASI and DLQI scores are required for all requests for PsO including those requests for patients that have significant involvement of the face, palms, soles of feet or genital region.
bRefractory' is defined as lack of effect at the recommended doses and for duration of treatments specified above. 'Intolerant' is defined as demonstrating serious adverse effects or contraindications to treatments as defined in product monographs.
cMTX at ≥ 20 mg (PO, SC, or IM) weekly (≥ 15 mg if patient is ≥ 65 years) for > 12 weeks.
dPatients who experience gastrointestinal intolerance to PO MTX must have a trial of parenteral MTX before being accepted as refractory.
e6 weeks treatment.
f4mg/Kg daily for 3 months
g15 to 30mg per week.
h12-week trial of parenteral MTX.
i12 week trial of cyclosporine.
jMTX at ≥ 20 mg (PO, SC, or IM) weekly (≥ 15 mg if patient is ≥ 65 years) for > 8 weeks.
kInformation is based on a personal communication with the jurisdictional representative (Anne Bastarache: personal communication, July 2021).
Note that this table has not been copy-edited.
Appendix 4: Approved Dose and Duration of Therapy
Note that this appendix has not been copy-edited.
Table 9: Dose and Duration of Therapy for Biologics in PsO
Drug dose and duration of therapy | Jurisdiction |
|---|---|
Adalimumab | |
Initial dose: 80 mg, then 40 mg at week 1, then 40 mg every 2 weeks | ABa, BC, ON, NB, NS, NL, PEI, NIHB |
Initial dose: 40 mg every 2 weeks | CSC |
Initial approval period: For a total of 9 doses or 16 weeks or 4 months | AB, BCb, SK, MB, NB, NS, NL, PEI, CSC, NIHB, VAC |
Initial approval period: 1 year | ON, YTa |
Renewal dose: 40 mg every 2 weeks | AB, BC, NS, PEI, YT, CSC |
Renewal approval period: 1 year | AB, BCb, ON, NL, PEI, CSC, VAC |
Etanercept | |
Initial dose: 100 mg per week (or 50 mg biweekly) for 12 weeks | ABa, BC, ON, NB, NS, NL, PEI, CSC |
Initial dose: 50 mg per week | YT |
Initial approval period: 12 weeks or 3 months | AB, BCb, MB, SK, NS, NL, PEI |
Initial approval period: 16 weeks | SK, NB, VAC |
Initial approval period: 1 year | ON, YTa, CSC |
Renewal dose: up to 50 mg per week | AB, ON, NB, NS, NL, PEI, YT, CSC |
Renewal dose: up to 50 mg weekly to twice weekly | BC |
Renewal approval period: 1 year | AB, BCb, ON, NL, PEI, YT, CSC, VAC |
Renewal approval period: long-term (not specified) | NB |
Infliximab | |
Initial dose: 5 mg/kg at 0, 2 and 6 weeks | ABc, BC, ON, NS, NL, PEI, CSC |
Initial approval period: 3 doses | AB, BCd |
Initial approval period 12 Weeks | NS, NL, CSC |
Initial approval period 16 Weeks | SK, MB, NB, PEI, VAC |
Initial approval period: 1 year | ON, YTa |
Renewal dose: 5 mg/kg every 8 weeks | AB, BC, ON, NS, NL, PEI, CSC |
Renewal approval period: 1 year | AB, BC, ON, NL, PEI, CSC, VAC |
Renewal approval period: long-term (not specified) | NB |
Ustekinumab | |
initial dose: < 100 kg: 45 mg at week 0, 4 and 16; > 100 kg: 90 mg at week 0, 4 and 16 | ABc, BC, ON, PEI, NIHB, CSC |
Initial dose: 45 mg at week 0, 4 and 16 | NS |
Initial dose: 90 mg at week 0, 4 and 16 | NB, NL |
Initial approval period: 3 doses or 16 weeks | AB, BC, SK, NB, NS, NL, PEI, YTa, NIHB, CSC, VAC |
Initial approval period: 3 months | MB |
Initial approval period: 1 year | ON, YTa |
Renewal dose: < 100 kg: 45 mg every 12 weeks; > 100 kg: 90 mg every 12 weeks | AB, BC, ON, YT |
Renewal dose: 90 mg every 12 weeks | NB, NL, PEI |
Renewal dose: 45 mg every 12 weeks | NS, CSC |
Renewal approval period: 1 Year | AB, BC, NB, NS, NL, ON, PEI, YT, CSC, VAC |
Risankizumab | |
Initial dose 150 mg at weeks 0, 4 and 16 | AB, BC, NL, YT |
Initial dose: 150 mg at weeks 0 and 4, and then every 12 weeks | ON, NB, PEI, NIHB |
Initial approval period 16 Weeks | AB, BC, SK, MB, NS, NB, NLe, PEI, YTa, NIHB, VAC |
Initial approval period: 1 year | ON, NL |
Renewal dose: 150 mg every 12 weeks | AB, BC, PEI, YT, NIHB |
Renewal approval period: 1 year | AB, BC, NB, PEI, YT, VAC |
Brodalumab | |
Initial dose: 210 mg at week 0, 1, 2, then, 210 mg every 2 weeks | ON, NIHB, NB, NS, NL, PEI |
Initial approval period: 12 weeks | NS, NIHB |
Initial approval period: 16 weeks | SK, MB, NB, NL, PEI |
Initial approval period: 1 year | ON |
Renewal dose: 210 mg every 2 weeks | ON, NB, NS, NL, PEI |
Renewal approval period: 1 year | NB, NS, NL, PEI |
Ixekizumab | |
Initial dose: 160 mg at week 0, then 80 mg at weeks 2, 4, 6, 8, 10 and 12 weeks | ABa, BC, ON, NB, NS, NL, PEI, NIHB |
Initial approval period: 12 weeks | AB, BCb, SK, MB, NB, NS, NL, PEI, NIHB |
Initial approval period: 16 weeks | VAC |
Initial approval period: 1 year | ON |
Renewal dose: 80 mg every 4 weeks | AB, BC, ON, NB, NS, NL, PEI, NIHB |
Renewal approval period: 1 year | AB, BC, NB, NS, PEI, VAC |
Secukinumab | |
Initial dose: 300 mg at week 0, 1, 2, 3 and 4, then 300 mg monthly | BC, SK, ON, NB, NS, NL, PEI, YT, NIHB |
Initial approval period: 12 weeks | BCf, SK, MB, NB, NS, NL, NIHB |
Initial approval period: 16 weeks | PEI, |
Initial approval period: 1 year | ON, YT |
Renewal dose: 300 mg monthly for 1 year | BC, NB, NS, NL, PEI, YT, NIHB |
Renewal approval period: 1 year | BCf, NB, NL, PEI, YT |
AB = Alberta; BC = British Columbia; CAF = Canadian Armed Forces; CSC = Correctional Services Canada; MB = Manitoba; NB = New Brunswick; NIHB = Non-Insured Health benefit; NL = Newfoundland and Labrador; NS = Nova Scotia; PE = Prince Edward Island; SK = Saskatchewan; VAC = Veterans Affairs Canada; YT = Yukon.
aFor AB, limited to 1-month supply per prescription. For YT, limited to 1-month supply per prescription for the first 4 months.
bMaximum 28-day supply per fill.
cLimited to 1 dose per prescription.
dMaximum of 56 days per fill for infliximab. One infusion (dose) usually provides treatment for 56 days or less.
eReassessment required at 12 weeks to confirm response.
fMaximum of 30 days per fill.
Acknowledgement: Tarry Ahuja
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up-to-date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third-party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
Redactions: Confidential information in this document has been redacted at the request of the sponsor in accordance with the CADTH Drug Reimbursement Review Confidentiality Guidelines.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.