Drugs, Health Technologies, Health Systems
Reimbursement Review
Spesolimab (Spevigo)
Sponsor: Boehringer Ingelheim (Canada) Ltd.
Therapeutic area: Generalized pustular psoriasis (GPP)
This multi-part report includes:
Clinical Review
Pharmacoeconomic Review
Ethics Review
Clinical Review
Abbreviations
AE
adverse event
AESI
adverse event of special interest
AGEP
acute generalized exanthematous pustulosis
CDA-AMC
Canada’s Drug Agency
CFB
change from baseline
Cl
confidence interval
DLQI
Dermatology Life Quality Index
DRESS
drug reaction with eosinophilia and systemic symptoms
ERASPEN
European Rare and Severe Psoriasis Expert Network
FACIT-Fatigue
Functional Assessment of Chronic Illness Therapy–Fatigue
GPP
generalized pustular psoriasis
GPPASI
Generalized Pustular Psoriasis Area and Severity Index
GPPGA
Generalized Pustular Psoriasis Physician Global Assessment
GRADE
Grading of Recommendations Assessment, Development and Evaluation
HD
high dose
HR
hazard ratio
HRQoL
health-related quality of life
IL
interleukin
IL-36R
interleukin-36 receptor
ITT
intention to treat
LD
low dose
LOCF
last observation carried forward
MCP-Mod
multiple comparison procedure with modelling techniques
MD
medium dose
MID
minimal important difference
NRI
nonresponse imputation
OL
open label
OLE
open-label extension
PM
primary (censoring) method
PSS
Psoriasis Symptom Scale
RCT
randomized controlled trial
SAE
serious adverse event
SAPHO
synovitis, acne, pustulosis, hyperostosis, and osteitis
SC
subcutaneous
SD
standard deviation
SOC
standard of care
TNF
tumour necrosis factor
VAS
visual analogue scale
Executive Summary
An overview of the submission details for the drug under review is provided in Table 1.
Table 1: Background Information for Application Submitted for Review
Item | Description |
|---|---|
Drug product |
|
Sponsor | Boehringer Ingelheim |
Indication | For the treatment of GPP in adults and pediatric patients 12 years of age and older and weighing at least 40 kg. |
Reimbursement request |
|
Health Canada approval status | NOC |
Health Canada review pathway | Standard review |
NOC date | July 31, 2024 |
Recommended dose | For GPP flare treatment:
For GPP flare prevention:
|
GPP = generalized pustular psoriasis; GPPGA = Generalized Pustular Psoriasis Physician Global Assessment; NOC = Notice of Compliance; SC = subcutaneous.
Introduction
Generalized pustular psoriasis (GPP) is a rare, chronic, severe, and potentially life-threatening neutrophilic skin disease characterized by recurrent episodes (GPP flares) of widespread eruption of sterile, macroscopically visible pustules that occur frequently with or without systemic inflammation.1-3 Although GPP can present with chronic skin involvement (e.g., painful erythema, scaling) similar to psoriasis vulgaris, it has a distinct pathophysiology involving the dysregulation of the immune system leading to the activation of immune cells surrounding an abnormality in the interleukin-36 (IL-36) pathway.4 Flares are characterized by the sudden onset of rapidly disseminating cutaneous eruption and sterile pustules, crusts, and scales combined with systemic symptoms, such as fever and general malaise with fatigue.5 Systemic symptoms and extracutaneous manifestations such as arthritis, uveitis, neutrophilic cholangitis, acute respiratory distress syndrome, and cardiovascular septic shock often accompany significant flares.5,6 GPP onset can occur at any age, including childhood; the median age of diagnosis is approximately 50 years.7 Risk factors for GPP include mutations in IL-36, smoking, obesity, anxiety disorder, and recent systemic corticosteroid.8 There are limited epidemiological data in Canada for GPP due to the rarity of the disease. The prevalence estimate reported in Canada for 2023 based on population-based data (hospitalizations and visits to emergency departments and hospital-based and community-based outpatient clinics) was 2.8 to 5.4 per million individuals, with an incidence rate of 1.95 per million.9 During an acute flare, patients with GPP have an increased risk of morbidity and mortality due to the systemic involvement of the disease. Mortality rates reported ranged from 2% to 16%.
There is an urgent unmet need for treatments that resolve GPP flares and help achieve rapid and effective control of recurrent episodes in patients. At the time of this review, no published guidelines in Canada for the management and prevention of GPP flares have been identified, and spesolimab is the only treatment approved in Canada that is indicated for the treatment of GPP. The current treatment options that are used in clinical practice in Canada are indicated for psoriasis (e.g., plaque psoriasis) and used off-label for GPP. These include biologics targeting interleukins (e.g., brodalumab, guselkumab, ixekizumab, secukinumab, bimekizumab, risankizumab, and ustekinumab), biologics targeting tumour necrosis factor (TNF) alpha (e.g., adalimumab, certolizumab pegol, etanercept, golimumab, and infliximab) and nonbiologic systemic drugs (e.g., acitretin, cyclosporine, and methotrexate). As noted by the sponsor, even when flares are treated, approximately 74% to 83% of patients still experience residual symptoms (e.g., relapse of localized skin lesions) between flares and after treatment.10,11
Spesolimab is a humanized antagonistic immunoglobulin G1 antibody that blocks IL-36 signalling by binding to the interleukin-36 receptor (IL-36R). The binding of spesolimab to IL-36R prevents the subsequent activation of IL-36R by its ligands (IL-36 alpha, beta, and gamma) and downstream activation of proinflammatory and profibrotic pathways. Spesolimab is approved for the treatment of GPP in adults and pediatric patients aged 12 years and older and weighing at least 40 kg.12 The recommended dose of spesolimab solution for infusion to treat a GPP flare is a single dose of 900 mg (two 450 mg/7.5 mL vials) administered as an IV infusion. If flare symptoms persist, an additional 900 mg dose may be administered 1 week after the initial dose. The recommended dose of spesolimab for GPP flare prevention is a subcutaneous (SC) loading dose of 600 mg (four 150 mg injections) followed by 300 mg (two 150 mg injections) administered subcutaneously every 4 weeks. If a patient experiences a GPP flare while receiving spesolimab SC prevention treatment, the GPP flare may be treated with spesolimab IV. Four weeks after GPP flare treatment with spesolimab IV, spesolimab SC can be initiated or reinitiated at a dosage of 300 mg (two 150 mg injections) administered every 4 weeks. In this case, an SC loading dose is not required.
Spesolimab has not been previously reviewed by Canada’s Drug Agency (CDA-AMC). The objective of this report is to review and critically appraise the evidence submitted by the sponsor on the beneficial and harmful effects of spesolimab 900 mg (60 mg/mL) single-dose IV infusion, and spesolimab 150 mg/mL concentrate for SC injection for the treatment and prevention of flares in adult and pediatric patients aged 12 years and older and weighing at least 40 kg. The sponsor has requested reimbursement for the following indication: the prevention of GPP flares (SC administration) in adults and pediatric patients aged 12 years and older who have a history of GPP flares, and the treatment of GPP flares (IV administration) in patients with a Generalized Pustular Psoriasis Physician Global Assessment (GPPGA) total score of 2 or greater.
Perspectives of Patients, Clinicians, and Drug Programs
The information in this section is a summary of input provided by the patient and clinician groups that responded to CDA-AMC’s call for input and from the clinical experts consulted by CDA-AMC for this review.
Patient Input
Input was received from 1 patient group, Psoriasis Canada, a national psoriatic disease organization. Psoriasis Canada gathered information from a virtual GPP summit of 7 participants, including 2 patients with diagnosed GPP, and by conducting interviews with 3 patients and 1 caregiver and conducting a survey with 10 respondents who were interested in attending the GPP virtual summit but were not able to do so. Psoriasis Canada explained that the severity of flares and symptoms of GPP can vary across patients and experiences. Emergency department visits or inpatient care may be required, depending on the level of skin impacted and the degree of systemic involvement. More severe involvement can lead to serious complications, including heart failure, renal failure, and sepsis. Psoriasis Canada added that living with GPP, even without active flares, can present challenges. For example, people with this condition may experience poor self-image, difficulty with intimacy, disruptions in school and work life, burden on personal finances, stigma and discrimination, feelings of isolation, and difficulties accessing diagnosis, care, and treatment throughout different times in their lives. Psoriasis Canada explained that patients’ lives can be completely disrupted during GPP flares as a result of missing work, being bedridden, being hospitalized, and being dependent on caregivers during severe flares.
Psoriasis Canada noted that important treatment outcomes reported by patients with GPP are symptom reduction, reduced frequency and severity of flares, management of symptoms between flares, access to appropriate care and treatment, and the ability to control GPP to reduce the stress of worrying about the next flare.
Clinician Input
Input From the Clinical Experts Consulted by CDA-AMC
According to the clinical experts consulted during the review, there is an unmet need for new treatments that rapidly resolve flare symptoms during acute flares and also prevent GPP flares from reoccurring. Before the Health Canada approval of spesolimab, the treatment options used in clinical practice to resolve acute flares and prevent recurrent flares were treatments indicated for plaque psoriasis and used off-label for patients with GPP. The experts consulted noted examples of treatments that are used in the acute-flare setting, which include methotrexate, cyclosporine, and acitretin. Other treatments preferentially considered for patients presenting with life-threatening flares were fast-acting biologics indicated for psoriasis such as biologics that target IL-17 (e.g., secukinumab, ixekizumab, bimekizumab, and brodalumab) or TNF alpha (e.g., adalimumab, certolizumab pegol, and infliximab). For long-term control and flare prevention, the experts noted that oral therapies or biologics such as those that target IL-23 or IL-12, and IL-23 (e.g., risankizumab, guselkumab, ustekinumab) are considered.
The ideal treatment goal for acute flares is resolving flares; improving erythema, pustulation, and accompanying systemic symptoms (such as fever and arthritis); and preventing mortality. For patients with a history of recurrent GPP flares or with a risk of GPP flares, the ideal treatment goal is to limit flares and reduce pain, which will eventually lead to improved patient quality of life.
The experts anticipate that spesolimab will shift the current GPP treatment paradigm. According to the experts, spesolimab would be appropriate as first-line therapy for the treatment of flares and the prevention of flares in patients with a definitive diagnosis of GPP due to its unique mechanism of action, an IL-36 receptor inhibitor designed to treat and prevent GPP flares.
The experts highlighted that spesolimab will be appropriate for patients presenting with an acute flare of GPP. According to the experts, it would be best not to reserve this medication for those who are intolerant to other options or for whom other medications are contraindicated, given the rapid onset of GPP and the associated risks of undertreatment, including hospitalization, respiratory failure, septic shock, and death. For flare prevention, spesolimab would also be appropriate for patients for whom no modifiable trigger has been identified for the flare of GPP, given that flares can be associated with abrupt withdrawal of immunosuppressive medications such as prednisone or cyclosporine and other medications such as terbinafine or amoxicillin.
According to the clinical experts consulted, the evaluation of the response to therapy in clinical practice will be based on whether patients are being treated for an acute flare or if the goal is to prevent reoccurrences of flares. The experts noted that resolving erythema and pustulation, including skin pain and systemic symptoms, will be an ideal therapeutic outcome for acute flares. The experts noted that, given the spectrum and severity of GPP, a meaningful response requires near-complete resolution of the flare to eliminate the risk of severe complications that may require hospitalization and place the patient at increased risk of mortality. A reduction in mortality associated with GPP would also be considered a good measure of the success of a therapy on a population level. The experts indicated that spesolimab may be discontinued after the resolution of an acute flare, although if there is a history of recurrent flares or the patient is at a high risk of another GPP flare, spesolimab may be used after the acute flare to prevent the recurrence of flares. The experts expressed that a physician, such as a dermatologist with expertise in the diagnosis and experience in the treatment of GPP and other subtypes of psoriasis, is necessary for treating and managing GPP.
Clinician Group Input
One clinician group submitted input for this submission, the Origins Dermatology Centre, which services urban, rural, and Indigenous populations in an underserviced area and focuses on medical and general dermatology. One clinician, who was the author of this input, gathered information from literature resources and those with clinical experience and obtained input from experienced nurses.
According to the Origins Dermatology Centre input, treatment goals would include fast control of acute flares, controlling signs and symptoms (e.g., fever, malaise, pain, itch, swelling, pustules) and controlling and preventing systemic worsening or collapse as a part of the disease process. Long-term goals would include encouraging sustained responses, including preventing flares, keeping patients out of the hospital, disease control, improving quality of life impact, and creating a favourable and advantageous safety profile.
Origins Dermatology Centre explained that, in Canada, there were no current guidelines or approved therapies for the treatment of GPP until spesolimab became available. Current off-label systemic treatments (systemic immunosuppressants and biologic therapy) for plaque psoriasis have proven inadequate to control chronic and acute forms of GPP. Origins Dermatology Centre added that based on a survey reported by Strober et al. (2021),13 dermatologists treating GPP reported there are high rates of relapse with current off-label therapies, and treatments are slow to control flares. Further, most patients will relapse within 1 year of treatment. The clinician group also noted that the broad oral systemic immunosuppressants that are often used for this condition (e.g., methotrexate, cyclosporine) come with side effects such as cytopenia, liver and renal toxicity, and increased risk of infection, among others, which limit both their short-term and long-term use in this disease.
Origins Dermatology Centre stated there is an unmet need for studied, approved, safe, and effective targeted options for the treatment of patients living with GPP. The clinician group believed that the drug under review would be a first-line therapy for those diagnosed with GPP, noting that those experiencing active disease, flares, systemic symptoms, and hospitalization would be most in need of intervention.
According to the Origins Dermatology Centre, clinical response over time, disease progression, and adjunctive therapy use may be considered when deciding to discontinue treatment with spesolimab.
Based on the clinician group input, hospitals and IV infusion clinics are the appropriate setting for treatment with spesolimab and, once the diagnosis is confirmed, specialists in the field of dermatology, internal medicine, and emergency medicine could prescribe and monitor its use effectively.
Drug Program Input
Input was obtained from the drug programs participating in the CDA-AMC Reimbursement Review process. The following were identified as key factors that could potentially impact the implementation of a CDA-AMC recommendation for spesolimab: relevant comparators, considerations for initiation of therapy, considerations for continuation or renewal of therapy, considerations for discontinuation of therapy, considerations for prescribing therapy, generalizability, care provision issues, and system and economic issues.
Clinical Evidence
Systematic Review
Description of Studies
The systematic review included 2 pivotal studies (Effisayil 1 and Effisayil 2). The Effasyil 1 trial evaluated the use of spesolimab IV as a treatment for acute GPP flares, whereas the Effisayil 2 trial evaluated the use of spesolimab SC for the prevention of flares.
Effisayil 1 was a multicentre, randomized, placebo-controlled, double-blind, phase II trial designed to evaluate the efficacy, safety, and tolerability of spesolimab administered as a single-dose IV compared with placebo in adults (aged 18 to 75 years) with GPP presenting with an acute flare of moderate to severe intensity who had received a diagnosis of GPP, as per European Rare and Severe Psoriasis Expert Network (ERASPEN) criteria. Patients were randomized to treatment with spesolimab or placebo if they experienced a GPP flare of moderate to severe intensity, defined by the emergence of the following (inclusive): a GPPGA total score of 3 or greater, new or worsening pustules, a GPPGA pustulation subscore of 2 or greater, and erythema and the presence of pustules involving 5% or more of the patient’s body surface area. Patients were enrolled across 37 centres in 12 countries, none of which were in Canada. In total, 53 patients who presented with a GPP flare of moderate to severe intensity were randomized (2:1) to receive either spesolimab 900 mg single-dose IV (n = 35 patients) or placebo (n = 18 patients). The primary end point was the proportion of patients with a GPPGA pustulation subscore of 0 at the end of week 1. The key secondary end point assessed the proportion of patients with a GPPGA total score of 0 or 1 at the end of week 1. Other secondary end points of note for this review included change from baseline (CFB) in the pain visual analogue scale (VAS). The final database lock date was April 1, 2021. The mean age in the spesolimab group was 43.2 years versus 42.6 years in the placebo group. In total, 60.0% and 83.3% of participants were female in the spesolimab and placebo groups, respectively. Numerical differences were observed in both arms of the trial in race (Asian ethnicity: 45.7% for spesolimab and 72.2% for placebo; white: 54.3% for spesolimab and 27.8% for placebo), GPPGA pustulation subscore (score of 2: 17.1% for spesolimab versus 27.8% for placebo; score of 3: 45.7% for spesolimab versus 38.9% for placebo; score of 4: 37.1% for spesolimab versus 33.3% for placebo), and present or past occurrence of psoriasis (yes: 68.6% for spesolimab versus 77.8% for placebo).
Effisayil 2 was a multicentre, randomized, placebo-controlled, double-blind, phase IIb dose-finding study designed to evaluate the efficacy and safety of spesolimab SC for the prevention of GPP flares in adult and pediatric patients aged 12 years and older with a history of GPP. Three doses were evaluated: low (spesolimab 300 mg loading dose followed by maintenance treatment of 150 mg every 12 weeks as SC injections), medium (spesolimab 600 mg loading dose followed by maintenance treatment of 300 mg every 12 weeks as SC injections), and high (spesolimab 600 mg loading dose followed by 300 mg subcutaneously, administered every 4 weeks). Only the efficacy results for the high dose (HD) have been reported for this review, as the other doses were not included under the recommended dosage approved by Health Canada. Patients enrolled in the Effisayil 2 trial were required to have a known and documented history of GPP, to have experienced at least 2 GPP flares, and to have a GPPGA score of 0 or 1 (clear or almost clear) at randomization. Patients were also required to be between the ages of 12 and 75 years and to have a documented history of GPP, per the ERASPEN criteria. The study was conducted across 71 sites in 23 countries, with no sites in Canada. In total, 30 patients were randomized into the HD group and 31 patients into the placebo group. The primary hypothesis was a dose-finding assessment followed by the assessment of time to first GPP flare at week 48; a key secondary end point was the proportion of patients with at least 1 GPP flare at week 48. The final database lock date was January 13, 2023. Patients who were enrolled were between the ages of 14 and 75 years (8 patients were adolescents). The mean age at randomization in the spesolimab HD group was 40.2 years (standard deviation [SD] = 16.4 years) versus 39.5 years (SD = 14.0 years) for placebo; 70.0% versus 54.8% of patients were of Asian ethnicity, 30.0% versus 45.2% were white, 3.3% versus 9.7% were Hispanic or Latino, 60.0% versus 58.1% were female, and 40.0% versus 41.9% were male in the spesolimab HD and placebo groups, respectively. At baseline, the mean weight was 68.7 kg (SD = 22.9) versus 75.73 kg (SD = 23.92), and the mean body mass index was 25.6 kg/m2 (SD = 7.3) versus 26.9 kg/m2 (SD = 8.3) in the spesolimab HD and placebo groups, respectively. The proportion of patients who had a GPPGA pustulation score of 0 (clear) was 67.7% in both the spesolimab HD and placebo groups; the proportion of patients with a GPPGA pustulation score of 1 (almost clear) was 33.3% in the spesolimab HD group and 32.3% in the placebo group. The mean Dermatology Life Quality Index (DLQI) total score was 11.1 (SD = 6.9) in the spesolimab HD group versus 7.2 (SD = 5.6) in the placebo group. Numerical differences were observed in the spesolimab HD group compared with placebo for race (proportion of patients who were of Asian ethnicity: 70% for spesolimab HD versus 54.8% for placebo), concurrent plaque psoriasis (23.3% for spesolimab HD versus 32.3% for placebo), presence of potentially pathogenic IL36RN variation (23.3% for spesolimab HD versus 12.9% for placebo), and prior use of at least 1 biologic therapy (20% for spesolimab HD versus 29% for placebo).
Patients who completed treatment with spesolimab in either of the 2 Effisayil trials were permitted to participate in Effisayil ON, a long-term, open-label extension (OLE) trial; however, results were not available at the time of this review.
Efficacy Results
Effisayil 1 Trial
Proportion of Patients With a GPPGA Pustulation Subscore of 0 at Week 1: Clinical experts, patient groups, and other advocacy groups considered a GPPGA pustulation subscore of 0 (i.e., no visible pustules) to be a critical outcome for decision-making and deliberations. At the April 1, 2021, data cut-off date, the primary objective of the Effisayil 1 trial, the proportion of patients with a GPPGA pustulation subscore of 0, was met. More specifically, 54.3% of patients who received a single dose of spesolimab IV experienced an improvement in flare resolution (GPPGA pustulation subscore of 0) 1 week following treatment compared with 5.6% of patients who received placebo, corresponding to a risk difference of 48.7% (95% confidence interval [CI], 21.5% to 67.2%; P = 0.0004) in favour of spesolimab. Three sensitivity analyses were carried out on the primary end point, specifically using alternative methods to handle missing data, plus an analysis of additional estimands (where death or any use of escape medication before observing the week 1 primary end point was considered a nonresponse). Findings from all sensitivity analyses were consistent with the main analysis of the primary end point.
Proportion of Patients With a GPPGA Total Score of 0 or 1 at Week 1: A GPPGA total score of 0 or 1 (i.e., clear or almost clear skin) was also identified as an outcome of importance to the clinical experts and the patient and clinician groups. In total, 42.9% of patients who received a single dose of spesolimab IV achieved clear or almost clear skin (i.e., GPPGA total score of 0 or 1) after 1 week compared with 11.1% of patients in the placebo group, corresponding to a risk difference of 31.7% (95% CI, 2.2% to 52.7%; P = 0.0118) in favour of spesolimab. The sensitivity analyses that were conducted were consistent with the main analysis.
Change From Baseline in Pain VAS Score at Week 4: Pain VAS scores were identified as a clinically important patient-reported outcome. In total, 88.9% of patients in the placebo group were considered nonresponders in terms of pain VAS score compared with 42.9% in the spesolimab group due to the use of escape medication, open-label (OL) spesolimab at day 8, or rescue medication with spesolimab before week 4. There was a decrease in median CFB of −2.45 in the spesolimab group, representing a decrease in pain, whereas, in the placebo group, the median was not calculable due to the use of escape medication, OL spesolimab at day 8, or rescue medication with spesolimab before week 4.
Effisayil 2 Trial
By the January 13, 2023, data cut-off, confirmatory testing of the secondary objective had been conducted.
Time to First GPP Flare Up to Week 48: The time to first GPP flare was considered by the clinical experts and patient groups to be a critical outcome for decision-making and deliberations regarding the prevention of GPP flares in adults and pediatric patients aged 12 years and older. As per the analysis of time to first GPP flare following 48 weeks of treatment, the risk of GPP flare was lower among patients who received spesolimab SC relative to patients who received placebo, based on a hazard ratio (HR) of 0.157 (95% CI, 0.046 to 0.541; P = 0.0005). Four sensitivity analyses (to assess whether any use of rescue medication with spesolimab IV or investigator-prescribed treatment for GPP was considered treatment for a GPP flare, i.e., event or treatment failure) were conducted for the primary end point. Findings from all sensitivity analyses were consistent with the main analysis of the primary end point.
Occurrence of 1 or More GPP Flares Up to Week 48: The proportion of patients experiencing 1 or more flares was also considered by the clinical experts, patient groups, and other advocacy groups to be a critical outcome for decision-making and deliberations for GPP flare prevention in adults and pediatric patients aged 12 years and older. The key secondary end point was met by the January 13, 2023, data cut-off date. The estimated adjusted risk difference by week 48 was −9.0% (95% CI, −62.1% to −15.9%; superiority P = 0.0013) in favour of spesolimab HD over placebo.
Time to First Worsening of DLQI Up to Week 48: Health-related quality of life (HRQoL) was assessed based on the time to first worsening of DLQI up to 48 weeks following initiation of treatment with spesolimab SC. Of note, first worsening of DLQI was defined as a 4-point increase in total score from baseline. Use of rescue medication or investigator-prescribed treatment for GPP was also considered to be the onset of worsening HRQoL. The patient group input identified HRQoL as an outcome of importance. In total, in the spesolimab HD and placebo groups, 27 patients had a DLQI worsening up to week 48 of treatment. In total, 23% of patients in the spesolimab HD group reported DLQI worsening at up to week 48 compared with 65% in the placebo group. The estimated HR for risk of DLQI worsening up to 48 weeks was 0.259 (95% CI, 0.109 to 0.620). The estimated risk difference for DLQI worsening in the spesolimab HD versus placebo was −42.4% (95% CI, −64.3% to −20.4%) in favour of spesolimab HD.
Harms
Effisayil 1 Trial
In the Effisayil 1 trial, adverse events (AEs) were reported before the nonrandomized administration of spesolimab and up to week 1 (herein referred to as the week 1 analysis) and following any spesolimab up to week 12 in addition to the residual-effect period (herein referred to as the week 12 analysis). Of note, the week 12 analysis included AEs observed in patients following treatment with any spesolimab verum (double-blind or nonrandomized) up until 16 weeks after the last administration of spesolimab, end of study, or treatment in the extension trial, whichever was earlier.
Based on the week 1 analysis, the incidence rate for AEs of any grade was numerically higher in the spesolimab group (77.1%) compared with placebo (66.7%) before the administration of nonrandomized spesolimab. The most frequently reported AEs during week 1 were pustular psoriasis (37.1% in the spesolimab group versus 38.9% in the placebo group) and pyrexia (5.7% in the spesolimab group versus 22.2% in the placebo group). Overall, most AEs were mild (grade 1) or moderate (grade 2), while the AEs experienced by 2 patients (11.1%) in the placebo group and 6 patients (17.1%) in the spesolimab group were classified as severe (grade 3). The grade 3 AEs in the spesolimab group included anemia, pustular psoriasis, and arthritis during week 1. Based on the week 12 analysis (i.e., after receiving any spesolimab at randomization, receiving OL spesolimab on day 8, or receiving spesolimab as rescue treatment later), 91.4% of patients initially randomized to spesolimab and 93.8% of patients initially randomized to placebo on day 1 experienced at least 1 AE up to week 12. The most frequently reported AEs overall up to week 12 were pustular psoriasis (57.1% in the spesolimab group versus 43.8% in the placebo group), pyrexia (8.6% in the spesolimab group versus 12.5% in the placebo group), and vomiting (11.4% in the spesolimab group versus 6.3% in the placebo group).
For the week 1 analysis, the most frequently reported serious adverse event (SAE) overall was pustular psoriasis (11.4% in the spesolimab group versus 16.7% in the placebo group). All other SAEs (arthritis, drug-induced liver injury, drug reaction with eosinophilia and systemic symptoms [DRESS], and urinary tract infection) were experienced by only 1 patient in each category. After receiving spesolimab, the most frequently reported SAEs were pustular psoriasis and DRESS in 9 and 2 patients, respectively. One patient in the spesolimab group was reported to have experienced adverse events of special interest (AESIs) (drug-induced liver injury and DRESS) before the administration of nonrandomized spesolimab. After receiving any spesolimab (randomized or rescue), 1 patient who was initially randomized to placebo on day 1 experienced latent tuberculosis. No patient discontinuations due to AEs and deaths were reported in the study.
Effisayil 2 Trial
The proportion of patients experiencing any AEs was comparable in both groups (86.7% in the spesolimab HD group and 86.7% in the placebo group). The most frequently reported AEs (≥ 10% in either group) were pustular psoriasis (10.0% of patients receiving spesolimab HD versus 53.3% receiving placebo), psoriasis (13.3% for spesolimab HD versus 10.0% for placebo), and injection site erythema (16.7% for spesolimab HD versus 3.3% for placebo). Overall, most patients experienced AEs of mild (grade 1) or moderate (grade 2) intensity. Overall, the most frequently reported AE of the worst intensity (grade 3) was pustular psoriasis, reported in 9 patients (9.7%) in the combined spesolimab dose groups and 4 patients (13.3%) in the placebo group.
In total, 10% of patients who received spesolimab HD and 3.3% of patients who received placebo reported 1 or more SAEs during the randomized treatment period of the study. The most reported SAE was pustular psoriasis (3.2%) across all spesolimab groups (1 patient in each spesolimab dose group) compared with none in the placebo group. The SAEs reported in the spesolimab HD group included pustular psoriasis, breast cancer, and cholelithiasis (1 patient each). No AESIs were reported in the spesolimab HD group. AEs leading to study discontinuation occurred in 10% of patients treated with spesolimab HD, which included pustular psoriasis, psoriasis, and breast cancer (1 patient for each AE; 3.3%); no patients in the placebo group discontinued due to AEs. There were no reports of death during the study.
Critical Appraisal
The Effisayil 1 and Effisayil 2 trials were multicentre phase II and IIb randomized controlled trials (RCTs), respectively. The risk of bias related to randomization and treatment allocation concealment was considered low in both studies. There were numerical differences observed in some factors in both studies (Effisayil 1 trial: sex, race, GPPGA pustulation subscore, and present or past occurrence of psoriasis; Effisayil 2 trial: race, concurrent plaque psoriasis, IL36RN variation, GPPGA total score and prior use of at least 1 biologic therapy), possibly due to the small sample size, which was expected due to the rarity of the disease. The clinical experts consulted during the review did not anticipate that these noted differences would bias findings.
Both trials were double-blind, and steps were implemented to maintain blinding of patients and investigators before the data cut-offs. However, there is the potential that patients could have inferred the group to which they were assigned, evidenced by differences observed in the efficacy and harms in the spesolimab group relative to placebo. The presence and direction of any bias are uncertain. Statistical analyses for the primary outcome in the Effisayil 1 trial were based on the exact Suissa and Shuster z-pooled test, and analyses in the Effisayil 2 trial were based on the stratified Cochran–Mantel–Haenszel test using the intention-to-treat (ITT) population. Missing data were imputed as nonresponders (for binary outcomes), and the last observation carried forward (LOCF) method was used for continuous outcomes. The statistical tests implemented in both studies were considered appropriate. Sensitivity analyses conducted in both trials showed that missing data were unlikely to bias the results for the primary outcome. The outcomes investigated in both trials were generally accepted and aligned with clinical practice.
Concerning external validity, the characteristics of the patients enrolled in both trials were considered representative of patients in Canada. Most patients enrolled across the 2 trials were of Asian ethnicity or were white. There were no key patient groups excluded. Both trials excluded patients with different conditions, such as synovitis, acne, pustulosis, hyperostosis, and osteitis (SAPHO) syndrome, primary erythrodermic psoriasis vulgaris, and drug-triggered acute generalized exanthematous pustulosis (AGEP), which may impact the generalizability of the findings from both trials to those patient populations in current practice. However, the clinical experts consulted did not anticipate that these exclusion criteria would impact the generalizability of the findings to patients in current practice. In both trials, placebo was used as the comparator because there is no approved treatment for GPP (the current drugs used for the treatment of GPP in practice are indicated for plaque psoriasis and currently used off-label for GPP). The use of other treatment options that are prescribed for GPP (biologics and systemic modulating drugs for GPP and other conditions such as plaque psoriasis) was restricted in the randomized phase of both trials but allowed as rescue therapy in scenarios where patients experienced a flare recurrence or did not experience an improvement following treatment with spesolimab. These procedures were considered appropriate and aligned with the approved Health Canada product monograph. The experts anticipate that a few patients will require up to 2 doses of spesolimab IV in practice to ensure complete resolution of flares. The experts also noted that patients with concomitant comorbidities (such as plaque psoriasis) may require other medications to treat symptoms other than GPP flares. The treatment assessment duration in the trials was considered appropriate and reflective of clinical practice. The information available at the time of this review was too limited to draw a conclusion on the long-term efficacy and safety of spesolimab for patients living with GPP, as the OLE study that enrolled patients from the Effisayil 1 and Effisayil 2 trials is ongoing.
GRADE Summary of Findings and Certainty of the Evidence
For the pivotal studies and RCTs identified in the sponsor’s systematic review, the Grading of Recommendations Assessment, Development and Evaluation (GRADE) was used to assess the certainty of the evidence for outcomes considered most relevant to inform the deliberations of the CDA-AMC expert committee, and a final certainty rating was determined as outlined by the GRADE Working Group.14,15
Following the GRADE approach, evidence from RCTs started as high-certainty evidence and could be rated down for concerns related to study limitations (which refer to internal validity or risk of bias), indirectness, imprecision of effects, and publication bias.
When possible, certainty was rated in the context of an important (nontrivial) treatment effect; if this was not possible, certainty was rated in the context of the presence of any treatment effect (i.e., the clinical importance is unclear). In all cases, the target of the certainty-of-evidence assessment was based on the point estimate and where it was located relative to the threshold for a clinically important effect (when a threshold was available) or to the null. The target of the certainty-of-evidence assessment was the presence or absence of a clinically important effect based on the threshold informed by the clinical experts consulted by CDA-AMC for the following outcomes: the proportion of patients with a GPPGA pustulation subscore of 0, the proportion of patients with a GPPGA total score of 0 or 1, CFB in pain VAS scores, and time to worsening of the DLQI up to week 48. The clinical experts could not provide a clinically meaning threshold for time to first GPP flare, the proportion of patients with the occurrence of 1 or more GPP flares, or SAEs; thus, the null was used.
Results of GRADE Assessments
The GRADE assessments included an evaluation of the main outcomes considered important by clinicians, patient groups, and other advocacy groups. The selection of outcomes for the GRADE assessment was based on the sponsor’s summary of clinical evidence, consultation with clinical experts, and the input received from patient and clinician groups and public drug plans. The following list of outcomes was finalized in consultation with members of the CDA-AMC expert committee and was assessed using GRADE: the proportion of patients with a GPPGA pustulation subscore of 0, the proportion of patients with a GPPGA total score of 0 or 1, CFB in pain VAS scores, time to first GPP flare, the proportion of patients with the occurrence of 1 or more GPP flares, time to worsening in DLQI, and SAEs.
Table 2 and Table 3 present the GRADE findings for spesolimab versus placebo for the Effisayil 1 and Effisayil 2 trials, respectively.
Table 2: Summary of Findings for Spesolimab Versus Placebo for the Treatment of Acute GPP Flares in Adults (Effisayil 1 Trial)
Outcome and follow-up | Patients (studies), N | Relative effect (95% CI) | Absolute effects (95% CI) | Certainty | What happens | ||
|---|---|---|---|---|---|---|---|
Placebo | Spesolimab | Difference | |||||
GPPGA pustulation subscore | |||||||
Proportion of patients with a GPPGA pustulation subscore of 0 Follow-up: 1 week | 53 | NR | 6 per 100 | 54 per 100 | 49 more per 100 (from 22 more to 67 more) | Moderatea | Spesolimab (900 mg single-dose infusion) likely results in a clinically meaningful increase in the proportion of patients with a GPPGA pustulation subscore of 0 after 1 week of treatment when compared with placebo. |
GPPGA total score | |||||||
Proportion of patients with a GPPGA total score of 0 or 1 Follow-up: 1 week | 53 | NR | 11 per 100 | 43 per 100 | 32 more per 100 (from 2 more to 53 more) | Moderateb | Spesolimab (900 mg single-dose infusion) likely results in a clinically meaningful increase in the proportion of patients with a GPPGA total score of 0 or 1 after 1 week of treatment when compared with placebo. |
Pain VAS | |||||||
CFB in pain VAS Follow-up: 4 weeks | 50 | Patients with pain VAS response (responders):
Median (IQR):
| Very lowc | The effect of spesolimab (900 mg single-dose infusion) on CFB in the pain VAS is very uncertain when compared with placebo. | |||
Harms | |||||||
Proportion of patients with an SAE before receiving nonrandomized spesolimab Follow-up: 1 week | 53 (1 RCT) | NR | 16.7 per 100 | 14.3 per 100 (NR) | NR | Lowd | Spesolimab may result in little to no difference in the proportion of patients experiencing 1 or more SAEs after week 1 compared with placebo. |
CFB = change from baseline; CI = confidence interval; GPP = generalized pustular psoriasis; GPPGA = Generalized Pustular Psoriasis Physician Global Assessment; IQR = interquartile range; NR = not reported; RCT = randomized controlled trial; SAE = serious adverse event; VAS = visual analogue scale.
aThe clinical experts consulted suggested a conservative threshold of 15 to 20 patients per 100 as a clinically meaningful minimal important difference between groups due to the rare nature of GPP and the lack of available treatments in current settings. Rated down 1 level for imprecision. Although all values within the 95% CI were considered clinically important, the sample size is small, raising concerns for prognostic imbalance and a potential that the true effect is overestimated.
bThe clinical experts consulted suggested a conservative threshold of 15 to 20 patients per 100 as a clinically meaningful minimal important difference between groups due to the rare nature of GPP and the lack of available treatments in the current setting. Rated down 1 level for imprecision. The 95% CI included values that were considered not clinically meaningful by the clinical experts consulted.
cIn the absence of a threshold for clinical importance, the null was used. Rated down 1 level for serious imprecision due to noncalculable events in the placebo arm. Rated down 2 levels for risk of bias due to the use of escape medications, open-label spesolimab, or rescue medication in the placebo group, rendering the effect uninterpretable. Pain VAS is a subjective outcome, and there is a potential for bias due to reporting if the patients inferred what group they were in.
dRated down 2 levels for very serious imprecision. The effect may be unstable, as it is informed by few events.
Sources: Effisayil 1 Clinical Study Report.16 Details included in the table are from the sponsor’s summary of clinical evidence.17
Table 3: Summary of Findings for Spesolimab HD Versus Placebo for the Prevention of GPP Flares in Adults and Pediatric Patients Aged 12 Years and Older (Effisayil 2 Trial)
Outcome and follow-up | Patients (studies), N | Relative effect (95% CI) | Absolute effects (95% CI) | Certainty | What happens | ||
|---|---|---|---|---|---|---|---|
Placebo | Spesolimab HD | Difference | |||||
Time to first GPP flare | |||||||
Time to first GPP flare (weeks) Follow-up: 48 weeks | 61 | Patients with GPP flares:
Median (95% CI) weeks to first flare:
HR: 0.157 (95% CI, 0.046 to 0.541) | Moderatea | Spesolimab 600 mg loading dose followed by 300 mg SC every 4 weeks likely results in a clinically meaningful increase in the time to first GPP flare compared with placebo. | |||
Occurrence of ≥ 1 GPP flares | |||||||
Probability of GPP flare occurrence Follow-up: 48 weeks | 61 | NR | 51.6 per 100 | 12.7 per 100 (95% CI, 5 to 28.9) | 39 fewer per 100 (95% CI, 62.1 to 15.9 fewer) | Moderatea | Spesolimab 600 mg loading dose followed by 300 mg SC every 4 weeks likely results in a clinically meaningful reduction in the proportion of patients having a flare event up to 48 weeks of treatment compared with placebo. |
Time to first DLQI worsening | |||||||
Time to first 4-point worsening of DLQI Follow-up: 48 weeks | 61 | Patients with DLQI worsening:
Median weeks to first DLQI worsening:
HR: 0.259 (95% CI, 0.109 to 0.620) | Lowb | Spesolimab 600 mg loading dose followed by 300 mg SC every 4 weeks may result in a clinically meaningful reduction in the proportion of patients with worsening of at least 4 points in the DLQI at week 48 when compared with placebo. | |||
Harms | |||||||
SAEs Follow-up: 48 weeks | 60 (1 RCT) | NR | 3.3 per 100 | 10 per 100 (NR) | NR | Lowc | Spesolimab 600 mg loading dose followed by 300 mg SC every 4 weeks may result in increase in SAEs when compared with placebo. The clinical relevance of the increase is uncertain. |
CI = confidence interval; DLQI = Dermatology Life Quality Index; GPP = generalized pustular psoriasis; HD = high dose; HR = hazard ratio; NE = not evaluable; NR = not reported; RCT = randomized controlled trial; SAE = serious adverse event; SC = subcutaneous.
aThe clinical experts could not provide a clinically important threshold, so the null was used. Rated down 1 level for imprecision. Although all values within the 95% CI did not include the null, the sample size was considered small, raising concern for prognostic imbalance and a potential that the true effect is overestimated. According to the clinical experts, the estimated between-group differences were clinically important.
bA 15% to 20% threshold was used as per clinical expert input. Rated down 1 level for imprecision. Although all values within the 95% CI were considered clinically important, the sample size is small, raising concerns for prognostic imbalance and a potential that the true effect is overestimated. Rated down 1 level for risk of bias. There is a risk that patients may have detected the treatment to which they were assigned due to differences in efficacy between groups, and the outcome is subjective. According to the clinical experts, the estimated between-group difference was clinically important. Due to prior failure of the statistical hierarchy (for Psoriasis Symptom Scale), results for DLQI are considered as supportive evidence.
cRated down 2 levels for very serious imprecision. The effect may be unstable, as it is informed by few events.
Sources: Effisayil 2 Clinical Study Report.18 Details included in the table are from the sponsor’s summary of clinical evidence.17
Conclusions
GPP is a rare, chronic, severe, and potentially life-threatening disease. At the time of this review, spesolimab is the only treatment approved in Canada with an indication for the treatment of GPP. As such, input from patients and clinicians on this review emphasized a need for new treatments that effectively treat and prevent flares. Two pivotal RCTs provided evidence for the efficacy and safety of spesolimab compared with placebo for the treatment and prevention of GPP flares (Effisayil 1 and Effisayil 2, respectively).
In the Effisayil 1 trial, patients receiving spesolimab IV infusions for acute GPP flares likely experienced better resolution of their flares with no visible pustules after 1 week of treatment compared with placebo. Similarly, patients who received spesolimab IV infusions may have experienced clear or almost clear skin following treatment after 1 week compared with placebo. A reduction in pain was considered an important patient-reported outcome for acute flare treatment. Compared with placebo, patients with GPP with acute flares may have experienced improvement in pain after receiving spesolimab, based on the CFB in the pain VAS.
Regarding the use of spesolimab for the prevention of flares assessed in the Effisayil 2 trial, treatment with spesolimab SC (loading dose of 600 mg followed by 300 mg of spesolimab every 4 weeks) up to 48 weeks likely results in a clinically meaningful increase in the time to first GPP flare compared with placebo for patients with GPP aged 12 years and older. Similarly, compared with placebo, patients treated with spesolimab were less likely to experience a GPP flare. HRQoL was measured using a 4-point increase from baseline in DLQI scores, indicative of worsening in HRQoL. Findings showed that patients receiving spesolimab may have had a lower risk of experiencing a worsening of their symptoms compared with placebo. However, given the hierarchical testing in the statistical analysis plan, the DLQI was not tested, and only a nominal P value was provided; thus, the findings should be interpreted as supportive. Potential sources of uncertainty for the key outcomes assessed in both trials include the small sample sizes, which raises concerns for potential prognostic imbalances between groups and the potential overestimation of the treatment effects; the potential risk of bias, particularly for subjective outcomes such as patient-reported outcomes and AEs; and the risk of bias for CFB in pain VAS, given that most patients in the placebo group received rescue medications or OL spesolimab. However, the direction of bias is uncertain.
The frequency of AEs reported was generally high due to the severity of GPP but was comparable across groups in both trials. Additionally, there were no deaths reported in either of the pivotal trials. The safety data reviewed in this report were available up to week 1 and up to week 12 for spesolimab IV in the Effisayil 1 trial, and up to 48 weeks for spesolimab SC in the Effisayil 2 trial. Overall, there were no new concerns regarding the safety profile of spesolimab in the acute setting. While the safety data available for spesolimab in the preventive setting did not raise any concerns, the evidence informing the long-term safety of spesolimab SC for patients living with GPP is ongoing, with limited information available at the time of this review.
Introduction
The objective of this report is to review and critically appraise the evidence submitted by the sponsor on the beneficial and harmful effects of spesolimab for injection, 900 mg (60 mg/mL) concentrate for solution for single-dose IV infusion and 150 mg/mL solution for SC injection, for the treatment and prevention of flares in adult and pediatric patients aged 12 years and older.
Disease Background
Contents within this section have been informed by materials submitted by the sponsor and clinical expert input. The following was summarized and validated by the review team.
GPP is a rare, chronic, severe, and potentially life-threatening neutrophilic skin disease characterized by recurrent episodes (GPP flares) of widespread eruption of sterile, macroscopically visible pustules that occur frequently with or without systemic inflammation.1-3
Although GPP can present with chronic skin involvement (e.g., painful erythema, scaling) similar to psoriasis vulgaris, also known as plaque psoriasis, they are distinct, with differing genetic, histological, and clinical features. GPP is largely caused by a dysregulation of the immune system that leads to the activation of keratinocytes, neutrophils, macrophages, dendritic cells, and T cells, all surrounding an abnormality in the IL-36 pathway.4 There is a link between a mutation in the IL-36 encoding genes and an elevated incidence of GPP. Mutations in IL36RN encoding IL-36R alpha were most common in patients with GPP who are of European (34.7%) and East Asian (28.8%) descent.19 Other risk factors associated with the development of GPP include smoking, obesity, anxiety disorder, and recent systemic corticosteroids.8 Although GPP onset can occur at any age, including childhood, the median age of diagnosis is around 50 years.7 Flares are characterized by the sudden onset of rapidly disseminating cutaneous eruption and sterile pustules, crusts, and scales combined with systemic symptoms, such as fever and general malaise with fatigue.5 Systemic symptoms and extracutaneous manifestations such as arthritis, uveitis, neutrophilic cholangitis, acute respiratory distress syndrome, and cardiovascular septic shock often accompany significant flares.5,6 Mortality rates attributable to GPP and its treatments, such as systemic corticosteroids, have been reported to be 2% to 16%.6
Based on Canadian population-based data published in 2023 for hospitalizations, visits to emergency departments, and visits to outpatient clinics that are based in hospitals and communities, the prevalence rate for GPP was reported to be 2.8 to 5.4 cases per million individuals, and the incidence rate was reported to be 1.95 per million individuals. The prevalence and incidence rates were the highest in Alberta, at 5.4 per million individuals and 3.7 per million individuals, respectively, due to 100% reporting in that province.9
According to the 2017 European consensus statement published by the ERASPEN, GPP should be diagnosed only when the condition has relapsed at least once or persists for more than 3 months. However, the statement did note there is an unmet need for the development of a new score to measure GPP severity.1
Standards of Therapy
Contents within this section have been informed by materials submitted by the sponsor and clinical expert input. The following was summarized and validated by the CDA-AMC review team.
There are no established guidelines globally for managing GPP, and current treatment options rely heavily on the approaches and options that are available for plaque psoriasis.3,4,20-22 Guidelines for the management of GPP established in the US and Japan are for GPP management in general; they do not provide guidance specifically for treating or preventing flare episodes in GPP and lack targeted biological options. As such, treatments used for GPP are associated with slow resolution of GPP flares, and the complete clearance of pustules and skin is not always achieved.10
In Canada, there are no guidelines for the management and prevention of GPP flares, and only spesolimab is currently approved for treating adults with GPP. Current treatment options are indicated for other types of psoriasis (e.g., plaque psoriasis) and are used off-label for GPP. These include biologics targeting ILs (e.g., brodalumab, guselkumab, ixekizumab, secukinumab, bimekizumab, risankizumab, and ustekinumab), biologics targeting TNF alpha (e.g., adalimumab, certolizumab pegol, etanercept, golimumab, and infliximab) and nonbiologic systemic drugs (e.g., acitretin, cyclosporine, and methotrexate). None of these therapies has been proven effective in treating GPP, as GPP has a distinct pathogenesis centred around IL-36. Studies have shown that even when flares are treated, about 74% to 83% of patients with GPP still suffer from residual symptoms (e.g., relapse of localized skin lesions) between flares and following treatment.10,11 When experiencing a flare, patients with GPP are still burdened by significant morbidity that may require frequent or long-term hospitalization and can be life-threatening.
There is an urgent unmet need for a treatment option that prevents the occurrence of GPP flares and helps achieve rapid and effective control of GPP flare episodes in patients. Spesolimab is a first-in-class IL-36R antibody specifically indicated for treating and preventing GPP flares in Canada that meets the needs of patients with GPP.
Drug Under Review
Key characteristics of spesolimab are summarized in Table 4 for the treatment of GPP, including the treatment of GPP in adults and pediatric patients aged 12 years and older and weighing at least 40 kg.
Spesolimab is a humanized antagonistic immunoglobulin G1 antibody that blocks IL-36 signalling by binding to IL-36R. The binding of spesolimab to IL-36R prevents the subsequent activation of IL-36R by its ligands (IL-36 alpha, beta, and gamma) and downstream activation of proinflammatory and profibrotic pathways. Spesolimab for injection includes a 150 mg/mL solution for SC injection and a 450 mg/7.5 mL (60 mg/mL) concentrate for solution for IV infusion.
The recommended dose of spesolimab for GPP flare prevention in adults and adolescents aged 12 years and older is an SC loading dose of 600 mg (four 150 mg injections), followed by 300 mg (two 150 mg injections) administered subcutaneously every 4 weeks. The recommended dose of spesolimab solution for infusion to treat a GPP flare is a single dose of 900 mg (2 × 450 mg/7.5 mL vials) administered as an IV infusion. If flare symptoms persist, an additional 900 mg dose may be administered 1 week after the initial dose. Spesolimab has not been previously reviewed by CDA-AMC. The sponsor’s requested reimbursement for spesolimab SC is for the prevention of GPP flares in adults and pediatric patients aged 12 years and older who have a history of GPP flares, and for IV use for the treatment of GPP flares in patients with a GPPGA total score of 2 or greater.
Spesolimab IV has been reviewed by Health Canada for the treatment of flares in adult patients with GPP and received a Notice of Compliance on July 31, 2024, for the treatment of GPP in adults and pediatric patients aged 12 years and older and weighing at least 40 kg. Spesolimab (IV and SC formulations) has been reviewed by the FDA and approved for treating GPP in adults and pediatric patients aged 12 years and older and weighing at least 40 kg. Spesolimab IV received a conditional marketing authorization from the European Medicines Agency for use in adults to treat flare-ups (recurrence or worsening) of GPP, an inflammatory skin disease causing pustules (pus-filled lesions) to appear over large areas of skin.
Table 4: Key Characteristics of Spesolimab
Characteristic | Spesolimab |
|---|---|
Mechanism of action | Spesolimab binds to IL-36R, preventing the subsequent activation of IL-36R by its ligands and downstream activation of proinflammatory and profibrotic pathways. |
Indicationa | For the treatment of GPP in adults and pediatric patients aged 12 years and older and weighing at least 40 kg. |
Route of administration | IV, SC |
Recommended dose | For GPP flare prevention in adults and adolescents aged 12 years and older, the recommended dose is an SC loading dose of 600 mg (four 150 mg injections) followed by 300 mg (two 150 mg injections) administered subcutaneously every 4 weeks. The recommended dose for infusion to treat a GPP flare is a single dose of 900 mg (2 × 450 mg/7.5 mL vials) administered as an IV infusion. |
Serious adverse effects or safety issues |
|
Other |
|
GPP = generalized pustular psoriasis; IL-36R = interleukin-36 receptor; SC = subcutaneous.
aHealth Canada–approved indication.
Source: Product monograph for spesolimab.12
Perspectives of Patients, Clinicians, and Drug Programs
Patient Group Input
This section was prepared by the CDA-AMC review team based on the input provided by patient groups. The full original patient input received by CDA-AMC has been included in the Perspectives of Patients, Clinicians, and Drug Programs section of this report.
Input was received from 1 patient group, Psoriasis Canada, formed by the recent consolidation of the Canadian Psoriasis Network (CPN) and the Canadian Association of Psoriasis Patients (CAPP) as a single, national, psoriatic disease organization. Both CPN and CAPP were originally formed as national not-for-profits. CPN was created to enhance the quality of life of people with psoriasis and psoriatic arthritis; CAPP was formed to better serve the needs of people living with psoriasis and psoriatic arthritis in Canada.
Psoriasis Canada gathered information via a virtual GPP summit of 7 participants, including 2 patients with diagnosed GPP, a survey of 10 people who were interested in attending the GPP virtual summit but were unavailable to attend live, and interviews with 3 patients and 1 caregiver.
Psoriasis Canada explained that the severity of flares and symptoms of GPP can vary across patients and experiences. Emergency department visits or inpatient care may be required, depending on the level of skin impacted and the degree of systemic involvement. More severe involvement can lead to serious complications, including heart failure, renal failure, and sepsis.
According to Psoriasis Canada, 1 patient with GPP described their experience with the disease as “survival,” including spending 3 days in the hospital on IV fluids and IV antibiotics in a life-threatening condition with pustules covering 70% of their body, resulting in losing all that skin at once and having to miss almost a month of work. Psoriasis Canada added that living with GPP, even in the absence of active flares, can present challenges. For instance, people with this condition may experience poor self-image, difficulty with intimacy, disruptions in school and work life, a burden on personal finances, stigma and discrimination, feelings of isolation, and difficulties accessing diagnosis, care, and treatment throughout different times in their lives.
Based on the patient group input, patients with GPP reported challenges of daily life. For example, these patients prefer to wear clothes that provide full coverage to have protection against the sun or contact with work equipment and to cover the affected skin in public, which results in being questioned about wearing these kinds of clothes. Additionally, patients with GPP experience unique challenges related to the rarity and potential severity of the condition, such as misdiagnosis; “having to go through layers” to get an appointment with a dermatologist; and receiving ineffective treatments with a family physician before obtaining a referral. Psoriasis Canada explained that patients’ lives can be completely disrupted during GPP flares, including missing work, being bedridden, being hospitalized, and being almost entirely dependent on caregivers during severe flares. According to Psoriasis Canada, the impact of the disease can also be significant between flares, as patients reported they must diligently manage their physical and mental health between flares because of concern that anything can trigger another flare. Based on the input, patients reported their concerns about being dependent on their current job and changing jobs in the future. Psoriasis Canada explained that GPP can also negatively affect relationships, and patients expressed frustration with having to repeatedly explain their condition to others.
Psoriasis Canada stated that according to the respondents, the current treatments for GPP are inadequate, unaffordable, or have side effects. According to Psoriasis Canada, patients with GPP described frustration in finding an effective treatment that was covered by insurance, accessing dermatologists with relevant expertise, paying for some treatments, asking their family physician for a referral, and asking their dermatologist to consider other treatment options.
Psoriasis Canada noted that important treatment outcomes reported by patients with GPP are symptom reduction, reduced frequency and severity of flares, management of symptoms between flares, accessibility, and the capability of controlling disease. Psoriasis Canada clarified that among the respondents, 3 patients with GPP had experience with spesolimab through a trial. Psoriasis Canada mentioned that 1 patient reported improvement in their condition that has changed their life; another patient highlighted that 30 hours after starting the treatment, the blisters on their lower legs were gone and the treatment worked; and the third patient reported they are “still really good” several months later, but had stress at the beginning of the new treatment which was “definitely worth it,” since they experienced no side effects and no flares, were sleeping better, were able to move, were less of a burden on their spouse and, even between flares, it “was never this good without the treatment.” Based on the patient group input, the third patient added that a maintenance dose may be appropriate for them because it is difficult to determine when an “attack” is going to occur and how long to wait, or how severe the symptoms are, before going for treatment again.
Clinician Input
Input From the Clinical Experts Consulted by CDA-AMC
All Reimbursement Review teams include at least 1 clinical specialist with expertise in the diagnosis and management of the condition for which the drug is indicated. Clinical experts are a critical part of the review team and are involved in all phases of the review process (e.g., providing guidance on the development of the review protocol, assisting in the critical appraisal of clinical evidence, interpreting the clinical relevance of the results, and providing guidance on the potential place in therapy). In addition, as part of the review of spesolimab, a panel of 4 clinical experts from across Canada was convened to characterize unmet therapeutic needs, assist in identifying and communicating situations where there are gaps in the evidence that could be addressed through the collection of additional data, promote the early identification of potential implementation challenges, gain further insight into the clinical management of patients living with GPP, and explore the potential place in therapy of the drug (e.g., potential reimbursement conditions). A summary of this panel discussion follows.
Unmet Needs
GPP is a rare and life-threatening disease characterized by a cutaneous eruption that may involve systemic symptoms or concomitant plaque psoriasis. GPP may cause high fever and malaise and significant extracutaneous effects such as cholangitis, acute renal failure, and pneumonitis. Compared with chronic plaque psoriasis, acute flares of GPP last several weeks, and patients may require hospitalization. Given the severity of flares and the systemic inflammation accompanying them, the experts highlighted that topical therapy is insufficient and oral therapy is necessary for the rapid control of symptoms and the reduction of morbidity and mortality. Before the Health Canada approval of spesolimab, the first-line options available to patients with GPP included conventional systemic treatments such as methotrexate, cyclosporine, and acitretin. Unfortunately, these treatment options do not adequately treat flares, and their onset of action is slow (typically weeks), leading to a significant risk of further clinical deterioration for patients and associated morbidity. Some of the current treatment options have limitations. For instance, some are contraindicated for specific groups of patients (acitretin), can result in a rebound flare (corticosteroids), are limited to 1 to 2 years of use, and are not compatible in patients with hepatic and renal dysfunction. Therefore, there is an unmet need for treatments that rapidly resolve symptoms during acute flares and prevent recurrent GPP flares.
GPP flares can also lead to patient mortality (mortality rates of 2% to 16% have been reported). According to the experts consulted, a treatment that is known to prevent patient death, treat systemic symptoms (and the possibility of organ damage), and rapidly treat the cutaneous symptoms of a flare, including the resolution of pustulosis, would be ideal in the acute setting, particularly if the safety profile is better than that of the off-label drugs commonly used in practice. Following the resolution and satisfactory management of an acute flare, an ideal treatment goal in the prevention setting would be to prevent or minimize subsequent flares, allowing patients to maintain employment, maintain independence, and reduce the burden on caregivers. The experts noted that current treatment options do not necessarily modify GPP’s pathophysiology or cure the disease. Thus, patients presenting with a flare are likely to have another flare in the future if they are not closely monitored or placed on ongoing therapy for flare prevention. According to the experts, not all patients respond adequately to available treatments, including conventional suppressive therapies like cyclosporine, and alternative biologics are not always effective. There exists a significant proportion of patients who continue to require hospitalization or admission to an intensive care unit or die as a result of a flare. According to the experts, the resolution of systemic symptoms such as fever and arthritis is an important treatment goal for the treatment of flares, in addition to improvement of the erythema and pustulation during a flare. For patients with a history of recurrent GPP flares or at risk of GPP flares, limiting or preventing flares is important, leading ultimately to a reduction in pain, an improvement in quality of life, and an improvement in the patient’s ability to maintain employment.
Place in Therapy
The experts noted that although GPP may be characterized as a subset of plaque psoriasis, the pathophysiology differs, with IL-36 being a key cytokine. There are currently no consensus guidelines for treating and managing GPP in Canada. However, the experts noted that clinicians will likely use the ERASPEN guidelines to define GPP flares in practice, and the GPPGA scoring system will likely be adopted to assess patient response to ongoing treatment.
Spesolimab will likely cause a shift in the current treatment paradigm. According to the experts, spesolimab would be appropriate as first-line therapy for the treatment of flares and the prevention of flares in patients with a definitive diagnosis of GPP due to its unique mechanism of action (targeting IL-36) and rapid onset of action, with a manageable safety profile compared with conventional treatments like cyclosporine, acitretin, and infliximab, which have been associated with AEs. The experts indicated it would be inappropriate to recommend that patients try other treatments before initiating spesolimab, given that patients with acute GPP flares are at increased risk of mortality. Awaiting any failure of other conventional therapies will delay targeted treatment and thus increase a patient’s risk of death. In addition, the current therapies are used off-label, and there is limited data on their efficacy and safety in patients with GPP.
Regarding using spesolimab for the long-term management of flares in patients with known flare reoccurrence, the clinical experts believe that spesolimab could be considered a first-line therapy due to its mechanism of action and efficacy, as reported in the Effisayil 2 trial. With limited evidence available, the experts were unable to make conclusive statements regarding how current off-label biologics compare with spesolimab with respect to the prevention of recurrent GPP flares. According to the experts, if patients with known GPP flares were currently receiving treatment with a biologic to prevent flare recurrence and there was no clinical indication to switch them, they would continue to monitor these patients before initiating spesolimab in this setting.
The experts believed that spesolimab could potentially be used in combination with other conventional off-label therapies for GPP, if these conventional therapies had different mechanisms of action that could help relieve flare symptoms in patients with inadequate flare resolution following initial management with spesolimab. The experts highlighted that if, for instance, 2 doses of spesolimab did not adequately resolve a GPP flare in a given patient, a second therapy might be considered to alleviate flare symptoms in a chronic situation. In other scenarios where a patient has comorbidities (e.g., plaque psoriasis), concomitant therapies to relieve patients of these other symptoms would also be considered.
Patient Population
According to the clinical experts, spesolimab will be appropriate for any patient with a diagnosis of acute flare due to GPP. Given the rapid onset of GPP and the associated risks of undertreatment, hospitalization, respiratory failure, and septic shock, the experts noted it would be inappropriate to reserve this medication only for patients who previously did not experience a response to other options or for whom other medications are contraindicated. For flare prevention, spesolimab would also be appropriate for patients for whom no modifiable trigger was identified for the flare of GPP, given that flares can be associated with abrupt withdrawal of immunosuppressive medications such as prednisone and cyclosporine, and other medications such as terbinafine and amoxicillin.
According to the experts, diagnosing GPP is not clinically challenging; however, there is a potential for misdiagnosis. Patients may be diagnosed at onset as having an infection (e.g., candidiasis or herpes zoster), an AGEP hypersensitivity reaction, or having another autoinflammatory disease (subcorneal pustular dermatosis or immunoglobulin A pemphigus). Given the overexpression of IL-36 in other pustular autoinflammatory disorders and AGEP, patients misdiagnosed as having GPP may also demonstrate clinical improvement with spesolimab similar to patients with GPP. The experts did not express any concerns regarding overdiagnosis or underdiagnosis of GPP in practice. A GPP diagnosis is made during an acute flare presentation and based on physician assessments. The clinical experts noted that in addition to the initial clinical examination, an additional culture of purulent fluid and a biopsy of the lesions may help identify patients with GPP and rule out mimicking conditions. However, an additional fluid culture and biopsy may not be feasible when patients present with an acute flare because these are time-consuming tests to perform.
According to the clinical experts consulted, the patients who would be least suitable for treatment with spesolimab, especially for preventing GPP flares, would include those who do not respond to initial treatment or patients with an allergy to components of spesolimab. The experts expressed that a patient having an acute GPP flare who shows no response to initial therapy with spesolimab, and no resolution of their flare, would likely not be considered for re-treatment with spesolimab. They would also not be considered for long-term treatment with spesolimab for flare prevention. However, the experts highlighted that if a patient’s condition demonstrates suboptimal response following initial treatment, spesolimab may still be considered due to issues associated with accessing other conventional off-label therapies in current practice. The presence of an IL-36 mutation may be a key factor in identifying patients who will respond to spesolimab. The experts noted that, given that some patients in the trials did not achieve flare resolution or flare prevention, it is unclear whether there is a clear patient demographic that may not respond well to spesolimab. In the opinion of the experts, patients in whom interferon, TNF, or IL-17 play a larger role in their GPP flares are likely patients whose condition would not respond well to spesolimab.
Assessing the Response to Treatment
According to the experts consulted, outcomes that will be considered to determine whether patients are responding to treatment in the acute-flare setting will include the resolution of erythema and pustulation and the resolution of skin pain and systemic symptoms (fever and the associated organ damage). In addition, patient response to changes in the pain VAS assessments is valuable, as a reduction in pain is expected along with any improvement in pustulation and erythema measured by the GPPGA scoring system. The experts noted that the outcomes used in clinical practice align closely with those used in the Effisayil clinical trials. The experts noted that given the spectrum and severity of GPP, a meaningful response requires complete or near-complete resolution of the flare to eliminate the risk of severe complications that may require hospitalization and place the patient at increased risk of mortality. The response magnitude may vary across patients based on disease severity and other factors or comorbidities.
In the flare-prevention setting, the clinical experts noted that assessing the failure of spesolimab treatment will depend on several factors, such as number of prior flares, severity of flares, time to flares, and number of flares in a given time frame. For instance, the experts noted that if a patient has 1 flare per year following treatment with spesolimab, and that is a great reduction from their previous number of flares, the experts would consider this treatment a success. However, if a patient is having 1 flare a year with current spesolimab SC treatment, and their baseline before treatment was 1 flare in 10 years, treatment with spesolimab may be considered a failure. Thus, defining treatment success or failure in the recurrent-flare setting depends on patient history and baseline flare-related values. According to 1 expert, a patient having 2 flares a year will be fundamentally defined as experiencing a treatment failure, and this patient will require either adjuvant therapy or a change of therapy.
The experts indicated that managing GPP depends on the knowledge of factors such as identifiable triggers for acute flares, time between flares, patient history of having flares (i.e., number of prior flares), and time interval between flares. After a patient is initially treated for an acute flare, the experts said they would continue to monitor the patient for subsequent flares or triggers (for instance, monthly for 3 to 6 months following initial treatment, and then maybe every 3 months after that for perhaps 1 or 2 years) before a decision to initiate preventive therapy is considered. According to the experts, any patient who experiences a GPP flare once a year following initial treatment of an acute flare, for example, will be considered for preventive therapy with spesolimab due to the increased mortality rates associated with flares. The experts also noted that patients who have had at least 2 prior flares are at high risk of having a third flare, especially in a situation where a trigger is known and depending on flare severity. These patients would also be considered for preventive therapy with spesolimab.
According to the clinical experts consulted, patients with an acute flare admitted to the hospital would be assessed daily. For patients who have milder flares or known disease courses, outcomes would generally be evaluated weekly or monthly in an outpatient setting. Once patients are on stable therapy to prevent flares, the expert indicated that patients would generally be monitored every 3 to 6 months. In the experts’ opinion, a reduction of mortality associated with GPP would also be considered a good measure of the success of a therapy on a population level. The experts added that, given the dramatic difference in clinical findings between patients who respond to treatment and those who do not, this is not likely to vary across physicians.
Discontinuing Treatment
Treatment discontinuation with spesolimab will be considered based on the phase of the treatment. According to the experts, the primary factor to be considered is whether or not a patient responds after a complete treatment course with spesolimab. The experts noted that treatment failure would be measured clinically via the persistence of pustular lesions or the development of additional systemic symptoms, including cardiopulmonary failure, renal failure, peripheral edema, and cholangitis. Treatment failure could also be measured by the patient’s need for additional therapies to manage systemic symptoms, including the addition of immunosuppressive therapies.
In an acute flare phase, the experts expressed that 2 treatments of spesolimab IV infusion with no improvement in signs and symptoms of GPP would warrant treatment discontinuation. In 1 expert’s opinion, treatment discontinuation will occur if a patient presents with 2 flares within a year, and considerations will be made for the addition of an adjuvant treatment for the GPP flare or a switch to an alternative drug. Treatment would also be discontinued if a patient experienced a severe drug reaction, such as an anaphylactic reaction to receiving spesolimab.
Prescribing Considerations
According to the clinical experts, spesolimab may be administered in an inpatient or outpatient setting (e.g., community infusion centres or specialty clinics with infusion capabilities) that allows for active monitoring of patients during infusions, depending on the severity of the flare and the experience of the treating clinician. For long-term flare prevention, the experts recommended that patients be treated in an outpatient setting. A specialist such as a dermatologist with expertise in diagnosing and treating GPP and other psoriasis subtypes would be necessary in this setting.
Clinician Group Input
This section was prepared by the CDA-AMC review team based on the input provided by clinician groups. The full original clinician group input received by CDA-AMC has been included in the Perspectives of Patients, Clinicians, and Drug Programs section of this report.
For this submission, input was received from 1 clinician group from Origins Dermatology Centre, which services urban, rural, and Indigenous populations in an underserviced area with a focus on medical and general dermatology. One clinician, who was the author of this input, gathered information from literature resources and those with clinical experience and obtained input from experienced nurses.
According to the Origins Dermatology Centre, treatment goals would include fast control of acute flares, controlling signs and symptoms (e.g., fever, malaise, pain, itch, swelling, pustules), and controlling and preventing systemic worsening or collapse as a part of the disease process. Long-term goals would include encouraging sustained responses, including preventing flares, keeping patients out of the hospital, disease control, improving quality of life impact, and an advantageous safety profile.
The Origins Dermatology Centre input explained that there are no approved therapies or current guidelines for the treatment of GPP. Current off-label systemic treatments (systemic immunosuppressants and biologic therapy) for plaque psoriasis have proven inadequate in controlling both chronic and acute forms of GPP. The Origins Dermatology Centre added that based on a survey reported by Strober et al. (2021),13 dermatologists treating GPP reported there are high rates of relapse with current off-label therapies, treatments are slow to control flares, and most patients will relapse within 1 year of treatment. Furthermore, the broad oral systemic immunosuppressants that are often used for this condition (e.g., methotrexate, cyclosporine) come with side effects such as cytopenia, liver and renal toxicity, and increased risk of infection, among others, which limit both their short- and long-term use in this disease.
The Origins Dermatology Centre input stated there is an unmet need for studied, approved, safe, and effective targeted options for treatment of patients with GPP. The clinician group believed that the drug under review would be a first-line therapy for those diagnosed with GPP.
According to the Origins Dermatology Centre input, clinical response over time, disease progression, and adjunctive therapy may be considered when deciding to discontinue treatment with spesolimab.
Based on the clinician group input, hospitals and IV infusion clinics are the best setting for treatment with spesolimab and, once the diagnosis is confirmed, specialists in the field of dermatology, internal medicine, and emergency medicine could prescribe and monitor effectively.
Drug Program Input
The drug programs participating in the CDA-AMC Reimbursement Review process provide input on each drug being reviewed by identifying issues that may impact their ability to implement a recommendation. The implementation questions and corresponding responses from the clinical experts consulted by CDA-AMC are summarized in Table 5.
Table 5: Summary of Drug Program Input and Clinical Expert Response
Drug program implementation questions | Clinical expert response |
|---|---|
Relevant comparators | |
The sponsor states there are reimbursed treatments that are currently used off-label in Canadian practice. These would include biologic drugs and nonbiologic systemic drugs. However, the Health Canada drug product monographs for methotrexate and cyclosporine do not define the type of psoriasis indicated, other than stating these drugs are for severe, disabling psoriasis. This could be interpreted to mean they are indicated for severe psoriasis of any subtype, including GPP, which is a severe form of psoriasis. Similarly, the Health Canada drug product monograph for acitretin notes it is indicated for severe psoriasis, including pustular types. 1. Would you agree there are no off-label comparators used in Canada for the treatment or prevention of GPP flares suitable for comparison with spesolimab? 2. If you disagree, which medication(s) do you feel would be a suitable comparator? | 1. The clinical experts highlighted that no other medications have been studied specifically for treating and preventing GPP flares. Conventional therapies are indicated for other diseases and are used off-label for GPP. The experts noted that although the product monographs for methotrexate and cyclosporine highlight potential use in psoriasis vulgaris, that type of psoriasis does not encompass GPP, and there is no direct evidence of the efficacy of these drugs in patients with GPP. 2. The experts cited the availability of evidence supporting the use of off-label biologics in GPP. The experts considered the following to be suitable comparators: biologics targeting IL (e.g., brodalumab, guselkumab, ixekizumab, secukinumab, bimekizumab, ustekinumab), biologics targeting TNF alpha (adalimumab, certolizumab pegol, etanercept, infliximab), and nonbiologic systemic drugs (acitretin, cyclosporine, methotrexate). |
Many of the therapies historically used to treat GPP are reimbursed for plaque psoriasis and include biologics that target IL and TNF alpha, in addition to nonbiologic medications such as acitretin, cyclosporine, and methotrexate. Access to public funding is limited, given the reimbursed indication for restricted biologic therapies is plaque psoriasis. | Comment from the drug programs to inform CDEC deliberations. |
Considerations for initiation of therapy | |
There is a 2017 European consensus statement by ERASPEN defining diagnostic criteria; however, there is a lack of consensus among experts in Canada, resulting in a diagnosis relying on expert examination. The reimbursement request includes the use of a GPPGA scoring system, requiring patients presenting with acute flares to have a GPPGA total score of 2 or greater. 1. Is the GPPGA currently used in clinical practice? 2. Are there potential barriers to asking for this score? 3. Should any other scores, such DLQI, be obtained as a baseline? | 1. The clinical experts highlighted that GPPGA assessments are currently not used in clinical practice for GPP. The experts noted the following barriers to using the GPPGA: limited time allotted for individual patient appointments and lack of familiarity with this scoring system. 2. The experts anticipate that clinicians will adopt the GPPGA scoring system if this is a prerequisite for patient access to treatment. 3. The experts indicated that the DLQI and PSS are ancillary scores that can support GPPGA scores in assessing treatment success. However, these outcomes will not supersede GPPGA. For example, 1 of the clinical experts indicated that the DLQI scores may be considered less relevant, given that the majority of patients with GPP will be managed in the setting of an acute flare. Considering the severe and life-threatening nature of an acute flare, the clinical expert suggested that the patient is unlikely to have clearly defined opinions related to how their skin condition has impacted their daily life over the last 7 days. |
The requested indication is for the treatment and prevention of flares in both adult and pediatric patients aged ≥ 12 years; however, patients younger than 18 years were excluded from the Effisayil 1 trial. In addition, patients weighing less than 40 kg were excluded from the preventive treatment study (Effisayil 2 trial).
| Despite the weight limitations outlined in the studies, the experts consulted did not anticipate safety concerns with using spesolimab to treat patients with GPP aged 12 years and older. The experts cited evidence on the biologics used to treat psoriasis and atopic dermatitis that show no clinically meaningful differences in safety between adolescents and adults. |
The Effisayil trials had a multitude of exclusions with regard to comorbidities, including the following conditions:
1. Do you feel the conditions listed as exclusions in the Effisayil trials would be contraindications to the use of spesolimab? 2. If not all conditions, which ones in particular would you consider to be contraindications? | 1. The clinical experts consulted were not concerned that the comorbidities listed as exclusion criteria in both trials (Effisayil 1 and Effisayil 2) would impact spesolimab use in patients with GPP in current practice. According to the experts, GPP is a life-threatening condition; thus, treatment will be chosen based on the potential risk of death from acute flares. The experts stated that some patients in their practice have died from using methotrexate, but they have not reported cases of mortality associated with the use of an IL-23 inhibitor. 2. According to the experts, patients with severe active infections such as TB, viral hepatitis, or systemic bacterial or fungal infections would not be suitable candidates for spesolimab. There is also evidence showing that spesolimab may cause liver injury. Therefore, patients with severe, progressive, or uncontrolled hepatic disease would be contraindicated. Patients with heart failure would also be contraindicated due to the volume of fluid that is infused during spesolimab treatment. The expert noted that GPP flare onset is progressive and nonresponsive to traditional therapies. Thus, treating physicians must weigh the risk of death due to GPP vs. the risk of medication administration. In this scenario, the only absolute contraindication would be anaphylaxis caused by spesolimab. |
There are no guidelines for the treatment of GPP, and all traditionally used treatments are considered by the sponsor to be off-label.
|
|
With regard to the need to establish baseline characteristics for assessing response to treatment, Consider aligning spesolimab with those biologic drugs previously given a positive CDEC recommendation for the indication of plaque psoriasis. For example, PASI vs. GPPASI and DLQI or other comparable outcomes. | Comment from the drug programs to inform CDEC deliberations. |
Considerations for continuation or renewal of therapy | |
It is unclear what outcomes would be useful for assessing continuation of therapy, as many were used in the studies. 1. Which outcomes would be applicable in real-world practice settings (e.g., GPPGA, GPPASI 75, absence or reduction of flares, PSS, DLQI)? 2. How should a clinically meaningful response be defined using objective parameters? | 1. According to the experts, any treatment should be geared toward achieving a GPPGA score of 0 or at least a complete resolution of erythema and pustulation. The experts further stated that desquamation may continue for longer periods and thus may not be a good marker of treatment success. 2. Both experts considered reducing or eliminating flares an important outcome for long-term treatment. One expert said they would objectively define a clinically meaningful response as the absolute reduction of flares (i.e., flares per year before spesolimab minus flares per year with spesolimab) or fewer than 2 flares per year. PSS and DLQI results were considered valuable assessments but would not supersede the clinical assessment. |
Consider aligning with biologic drugs previously given a positive CDEC recommendation for the indication of plaque psoriasis with regard to requiring assessment using the PASI or GPPASI, DLQI, or other comparable measures. | Comment from the drug programs to inform CDEC deliberations. |
Considerations for discontinuation of therapy | |
The trial treatment was discontinued once the investigator-prescribed treatment for GPP was initiated, with the exception of treatments such as topical steroids, methotrexate, cyclosporine, and retinoids during flare treatment periods (4 weeks post IV day 1).
|
|
Consider aligning with biologics previously given a positive CDEC recommendation for the indication of plaque psoriasis with regard to requiring assessment using the PASI or GPPASI, DLQI, or other comparable measures. | Comment from the drug programs to inform CDEC deliberations. |
Considerations for prescribing of therapy | |
As per the drug product monograph, treatment with spesolimab should be initiated by physicians experienced in the management of patients with inflammatory skin diseases. This is a rare disease, with an estimated prevalence in Canada of 2.77 cases per million individuals. Depending on the amount of specialized training required, accessing an experienced physician may be challenging in some areas. | Comment from the drug programs to inform CDEC deliberations. |
At this time, most therapies used for GPP are considered off-label, with only SOC being publicly available.
| Both experts consulted highlighted that there are patients in practice who have concurrent psoriasis that also appears to be prone to GPP flares. According to the experts, these patients will require other therapies in addition to spesolimab to control the totality of the psoriasis. The experts anticipate that a portion of patients treated with spesolimab will require adjuvant or combination therapy with biologics or oral drugs geared at treating psoriasis, some of which will include biologics targeting IL (e.g., brodalumab, guselkumab, ixekizumab, secukinumab, bimekizumab, risankizumab, and ustekinumab), biologic drugs targeting TNF alpha (adalimumab, certolizumab pegol, etanercept, golimumab, infliximab); nonbiologic systemic drugs (acitretin, cyclosporine, methotrexate), and phototherapy. The experts also noted that in the event of treatment failure with spesolimab, medications, including acitretin, cyclosporine, and antipsoriasis biologics will be started to manage an acute flare. The experts noted that for GPP flare prevention, they do not anticipate any medications to be used in combination. |
Generalizability | |
Can patients with GPP who are currently in remission on an off-label biologic drug transition to spesolimab? | According to the clinical experts, patients could be transitioned from an off-label biologic to spesolimab; however, the experts do not anticipate this scenario would happen frequently. Both experts noted that if patients were doing well on an off-label biologic and were currently in remission, they would not advise transitioning patients to spesolimab except in the event of a flare. |
Care provision issues | |
Spesolimab IV requires access to a hospital or infusion clinic for administration by a trained health care professional. Administering spesolimab SC requires patient or caregiver training. | Comment from the drug programs to inform CDEC deliberations. |
Anti-TB medication should be considered before initiating spesolimab in patients with latent TB or a history of TB for whom an adequate course of treatment cannot be confirmed. | Comment from the drug programs to inform CDEC deliberations. |
System and economic issues | |
The BIA was developed to encompass the full Health Canada indication rather than the requested deviation to the indication. This may affect the actual impact. | Comment from the drug programs to inform CDEC deliberations. |
The sponsor noted that it is offering a patient support program that will limit the impact on publicly funded health care resources during the treatment of flares with spesolimab IV and noted that the patient support program will also be available for preventive treatment with spesolimab SC. The program limitations remain undefined. | Comment from the drug programs to inform CDEC deliberations. |
AGEP = acute generalized exanthematous pustulosis; BIA = budget impact analysis; ALT = alanine aminotransferase; AST = aspartate aminotransferase; CDEC = Canadian Drug Expert Committee; DLQI = Dermatology Life Quality Index; ERASPEN = European Rare and Severe Psoriasis Expert Network; GPP = generalized pustular psoriasis; GPPASI = Generalized Pustular Psoriasis Area and Severity Index; GPPASI 75 = 75% reduction in the Generalized Pustular Psoriasis Area and Severity Index score; GPPGA = Generalized Pustular Psoriasis Physician Global Assessment; IL = interleukin; PSS = Psoriasis Symptom Scale; SC = subcutaneous; SOC = standard of care; SAPHO = synovitis, acne, pustulosis, hyperostosis, and osteitis; TB = tuberculosis; TNF = tumour necrosis factor; ULN = upper limit of normal.
Clinical Evidence
The objective of this Clinical Review Report is to review and critically appraise the clinical evidence submitted by the sponsor on the beneficial and harmful effects of spesolimab 900 mg (60 mg/mL) single-dose IV infusion and spesolimab 150 mg/mL concentrate solution for SC injection for the treatment and prevention of flares in adult and pediatric patients aged 12 years and older.
A summary of the clinical evidence included by the sponsor in the review of spesolimab is presented in 2 sections, with the CDA-AMC critical appraisal of the evidence included at the end of each section. The first section, the systematic review, includes the pivotal studies and RCTs selected according to the sponsor’s systematic review protocol. The CDA-AMC assessment of the certainty of the evidence in this first section using the GRADE approach follows the critical appraisal of the evidence.
Included Studies
Clinical evidence from the following is included in the CDA-AMC review and appraised in this document:
the 2 pivotal RCTs identified in the systematic review.
Systematic Review
Contents within this section have been informed by materials submitted by the sponsor. The following has been summarized and validated by the CDA-AMC review team.
Description of Studies
Characteristics of the included studies are summarized in Table 6. The trial designs are shown in Figure 1, Figure 2, and Figure 3.
Table 6: Details of Studies Included in the Systematic Review
Detail | Effisayil 1 trial | Effisayil 2 trial |
|---|---|---|
Designs and populations | ||
Study design | Phase II, randomized, placebo-controlled, double-blind, parallel-group study | Phase IIb, randomized, placebo-controlled, double-blind, parallel-group, dose-finding study |
Locations | 37 centres in 12 countries (China, France, Germany, Japan, Malaysia, Singapore, South Korea, Switzerland, Taiwan, Thailand, Tunisia, US) | 71 centres in 23 countries (Argentina, Belgium, Chile, China, France, Germany, Greece, Italy, Japan, Korea, Netherlands, Philippines, Republic of Malaysia, Mexico, Russian Federation, South Africa, Spain, Taiwan, Thailand, Tunisia, Turkey, US, Vietnam) |
Patient enrolment dates |
|
|
Randomized (N) | N = 53:
| N = 123:
|
Inclusion criteria | Criteria for enrolment in the trial:
In addition, patients were required to have previous or current evidence of fever, asthenia, myalgia, elevated CRP, and/or leukocytosis with peripheral blood neutrophilia (above the ULN) and 1 of the following:
Criteria for initiation of randomized treatment: Treatment was initiated immediately (at visit 2) for patients who experienced a GPP flare of moderate to severe intensity, defined by the emergence of the following (inclusive):
|
|
Exclusion criteria |
|
|
Drugs | ||
Intervention |
|
|
Comparator | Placebo (solution for IV infusion) | Placebo (SC injection) |
Rescue therapies |
|
|
Study duration | ||
Screening phase | In the 6 months before starting treatment (screening at visit 1 before day 1) | In the 12 weeks before starting treatment (visit 1 before day 1) |
Treatment phase | On day 1 | 48 weeks |
Follow-up phase | 12 to 28 weeks after treatment (depending on the timing of the last dose and participation in OLE triala) | 16 weeks after the last dose of treatment (i.e., at week 44) |
Outcomes | ||
Primary end point | Proportion of patients with GPPGA pustulation subscore of 0 (i.e., no visible pustules) at week 1 | Time to first GPP flare (defined by an increase in the GPPGA score of ≥ 2 from baseline and a score of ≥ 2 in the pustular component of the GPPGA) up to week 48. |
Secondary and exploratory end points | Key secondary:
Secondary:
Exploratory:
Safety end points:
| Key secondary:
Secondary:
Exploratory:
Safety:
|
Publication status | ||
Publications | Bachelez et al. (2021)23 EudraCT 2017 to 004231 to 3724 NCT0378279225 | Morita et al. (2023)26 EudraCT 2018 to 003081 to 1427 NCT0439983728 |
AE = adverse event; AGEP = acute generalized exanthematous pustulosis; ALT = alanine aminotransferase; AST = aspartate aminotransferase; BI 655130 = spesolimab; CFB = change from baseline; CGI-I = Clinical Global Impression–Improvement scale; CRP = C-reactive protein; DLQI = Dermatology Life Quality Index; ECG = electrocardiogram; ERASPEN = European Rare and Severe Psoriasis Expert Network; FACIT = Functional Assessment of Chronic Illness Therapy; GPP = generalized pustular psoriasis; GPPASI = Generalized Pustular Psoriasis Area and Severity Index; GPPASI 50 = 50% reduction in Generalized Pustular Psoriasis Area and Severity Index score; GPPASI 75 = 75% reduction in Generalized Pustular Psoriasis Area and Severity Index score; GPPGA = Generalized Pustular Psoriasis Physician Global Assessment; JDA = Japanese Dermatological Association; OL = open label; OLE = open-label extension; PGI-C = Patient Global Impression of Change; PGI-S = Patient Global Impression–Severity; PSS = Psoriasis Symptom Scale; q.4.w. = every 4 weeks; q.12.w. = every 12 weeks; SAE = serious adverse event; SAPHO = synovitis, acne, pustulosis, hyperostosis, and osteitis syndrome; SC = subcutaneous; SF-36 = Short Form (36) Health Survey; SOC = standard of care; TB = tuberculosis; TEAE = treatment-emergent adverse event; TPSS = Target Plaque Severity Score; ULN = upper limit of normal; VAS = visual analogue scale; WBC = white blood cell; WPAI = Work Productivity and Activity Impairment.
aPatients who achieved clinical improvement and completed the trial were offered to roll over into the OLE trial. If eligible, they were followed until week 12; otherwise, they were followed for 16 weeks (i.e., corresponding to the residual-effect period) or up to 28 weeks, depending on whether they received rescue treatment with OL spesolimab.
Sources: Bachelez et al. (2021),23 Morita et al. (2023),26 and Effisayil 116 and Effisayil 218 Clinical Study Reports. Details included in the table are from the sponsor’s summary of clinical evidence.17
Two pivotal studies (Effisayil 1 and Effisayil 2) were included in the sponsor’s systematic review to support the reimbursement request.
Effisayil 1 was a multicentre, randomized, placebo-controlled, double-blind, phase II trial designed to evaluate the efficacy, safety, and tolerability of spesolimab single-dose IV compared with placebo in adult patients with GPP presenting with an acute flare of moderate to severe intensity. Patients aged 18 to 75 years who had received a diagnosis of GPP as per the ERASPEN criteria were enrolled across 37 centres in 12 countries. There were no sites in Canada. Of the 85 patients screened, 53 who presented with a GPP flare of moderate to severe intensity were randomized (2:1) to receive either spesolimab 900 mg single-dose IV (n = 35) or placebo (n = 18). Randomization was performed using blocking and stratified by Asian versus non-Asian ethnicity. The trial’s primary end point was the proportion of patients with a GPPGA pustulation subscore of 0 at the end of week 1. The key secondary end point assessed the proportion of patients with a GPPGA total score of 0 or 1 at the end of week 1. Other secondary end points included the proportion of patients with a 75% or greater reduction in the Generalized Pustular Psoriasis Area and Severity Index (GPPASI) at week 4, CFB in pain VAS score at week 4, the CFB in Psoriasis Symptom Scale (PSS) score at week 4, and the CFB in the Functional Assessment of Chronic Illness Therapy–Fatigue (FACIT-Fatigue) score at week 4. The Effisayil 1 trial comprised 3 phases: a screening period of up to 6 months, a single-dose treatment on day 1, and a follow-up period from 12 to 28 weeks. Follow-up duration varied across patients and depended on whether they needed rescue treatment with OL spesolimab after week 1 (day 8) through week 12. The final database lock date was April 1, 2021.17
Effisayil 2 was a multicentre, randomized, placebo-controlled, double-blind, phase IIb dose-finding study designed to evaluate the efficacy and safety of 3 different doses of spesolimab SC (low, medium, and high) for the prevention of GPP flares in adult and pediatric patients aged 12 years and older with a history of GPP. The patients enrolled had experienced at least 2 GPP flares and had a GPPGA score of 0 or 1 (clear or almost clear) at randomization, were aged between 12 and 75 years, and had a known and documented history of GPP, as per the ERASPEN criteria. The study was conducted across 71 sites in 23 countries, with no sites in Canada. Of the 157 patients screened, 123 were randomized (1:1:1:1) to receive low-dose (LD) spesolimab (n = 31), medium-dose (MD) spesolimab (n = 31), spesolimab HD (n = 30), or placebo (n = 31). Randomization was stratified by concomitant use of systemic GPP medications at randomization (yes versus no) and 2 blocking factors: region (Japan versus non-Japan) and population (adults versus adolescents). The primary objective of the Effisayil 1 trial was to demonstrate a non-flat dose-response curve and evaluate the dose-response relationship for 3 spesolimab SC dosing regimens (with each regimen consisting of a single loading dose and a separate maintenance SC dosing regimen) versus placebo on the primary end point, the time to the first GPP flare onset up to week 48. This objective was met at the data cut-off for this analysis. The secondary objective was to demonstrate the superiority of spesolimab HD (300 mg every 4 weeks) and spesolimab MD (300 mg every 12 weeks) versus placebo on the primary end point, time to first GPP flare onset up to week 48, as well as the key secondary end point, the occurrence of at least 1 GPP flare up to week 48. Other secondary end points included time to first worsening of PSS, DLQI, and sustained remission as per GPPGA score, measured up to week 48. The Effisayil 2 trial also comprised 3 phases: a screening period of 3 months, a randomized treatment phase involving a single loading dose administered on day 1, and a separate SC dosing regimen maintenance dose over 48 weeks. A follow-up period of 16 weeks after the last dose of the trial medication (i.e., the residual-effect period) was designed for patients who did not qualify or declined to participate in the OLE trial. While 3 dosage regimens were studied in the Effisayil 2 trial, only the efficacy findings from the spesolimab HD group are reviewed for the reimbursement request, given that the Health Canada recommended dosage of spesolimab for GPP flare prevention is a 600 mg loading dose of spesolimab SC, followed by 300 mg administered subcutaneously every 4 weeks (spesolimab HD). Baseline disease and demographic characteristics and patient disposition are presented for all groups assessed in the trial. The final database lock date was January 13, 2023.17
Patients who completed treatment with spesolimab in either of the 2 Effisayil trials were permitted to participate in the Effisayil ON long-term OLE trial.29
Populations
Inclusion and Exclusion Criteria
Effisayil 1 Trial
The key inclusion criterion for enrolment in the Effisayil 1 trial was a diagnosis of GPP as per the ERASPEN criteria. Patients were also required to have 1 of the following: a GPPGA score of 0 or 1, an acute flare of moderate intensity, or a first episode of acute GPP flare of moderate-severe intensity with evidence of at least 1 systemic symptom (i.e., fever, asthenia, myalgia, elevated C-reactive protein, neutrophilic leukocytosis), and a retrospective diagnosis of GPP. Patients were randomized to treatment with spesolimab or placebo if they experienced a GPP flare of moderate to severe intensity, defined by the emergence of the following (inclusive): a GPPGA total score of 3 or greater, new or worsening pustules, a GPPGA pustulation subscore of 2 or greater, and erythema and the presence of pustules involving 5% or more of the patient’s body surface area. Additionally, patients who had been receiving background treatment with retinoids, methotrexate, cyclosporine, or other restricted medications that could interfere with the study drug had to discontinue treatment before receiving the first dose of the study medication.17
The key exclusion criteria were patients who presented with other types of psoriasis or conditions that could interfere with treatment (e.g., plaque psoriasis without the presence of pustules or with pustules that were restricted to psoriatic plaques, drug-triggered AGEP, or SAPHO syndrome). Additionally, patients with GPP flares of life-threatening severity were excluded.17
Effisayil 2 Trial
The key inclusion criteria included a known and documented history of GPP as per the ERASPEN criteria, with a history of at least 2 GPP flares and a GPPGA score of 0 or 1 at both screening and randomization. Additionally, patients not receiving concurrent GPP treatment at randomization were required to have experienced 2 moderate to severe GPP flares in the past year before randomization, with at least 1 flare showing evidence of systemic symptoms (e.g., fever, elevated C-reactive protein, elevated white blood cell count, asthenia, myalgia). For those not on concomitant GPP treatment at random assignment but who had been on such treatment up to 12 weeks before, it was necessary to have a history of flaring during treatment or following dose reduction or treatment discontinuation. Furthermore, patients undergoing background treatment with retinoids, methotrexate, cyclosporine, or other restricted medications that could interfere with treatment were required to discontinue these drugs before receiving the first dose of the study medication. These patients also had to have a history of flaring while on concurrent treatment for GPP or after a dose reduction or discontinuation of concomitant medication. The key exclusion criterion was the presence of concomitant illnesses that could interfere with treatment (e.g., SAPHO syndrome, primary erythrodermic psoriasis vulgaris, and drug-triggered AGEP).17
Figure 1: Effisayil 1 Trial Study Design
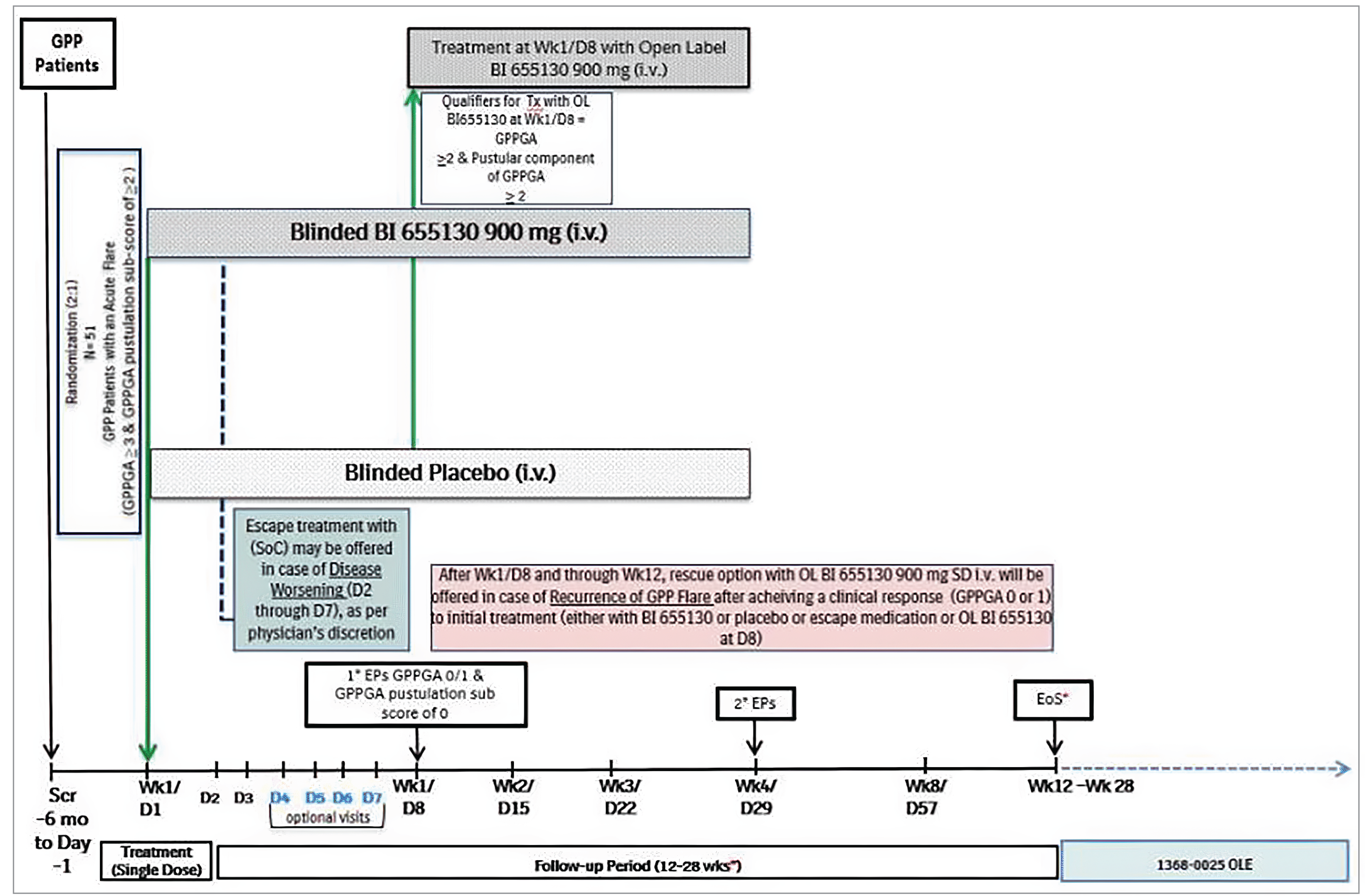
BI 655130 = spesolimab; D = day; EoS = end of study; EP = end point; GPP = generalized pustular psoriasis; GPPGA = Generalized Pustular Psoriasis Physician Global Assessment; OL = open label; OLE = open-label extension; Scr = screening; SD = single dose; SOC = standard of care; Tx = treatment; Wk = week.
Notes: Days 2 to 7: Escape treatment (SOC) could be offered in case of disease worsening defined as worsening of clinical status or GPP skin and/or systemic symptoms as defined by the investigator.
After day 8 to week 12: Only 1 rescue dose with OL spesolimab was permitted if a patient who had previously achieved a response to initial treatment (GPPGA score of 0 or 1) with either spesolimab or placebo at day 1, or escape medication or OL spesolimab at day 8, subsequently experienced a recurrence of a GPP flare (≥ 2-point increase in the GPPGA score and a score of ≥ 2 in the pustular component of the GPPGA). Subsequent flares were to be treated with SOC, per physician’s choice.
Patients who did not require rescue treatment with OL spesolimab were to be followed until week 12 (visit 14 or EoS) before entering OLE trial 1368 to 0025.
Patients who received rescue treatment with OL spesolimab between weeks 2 and 6 were to be followed until week 12 (visit 14 or EoS) before entering the OLE trial. If at visit 14 they qualified to enter the OLE trial, then visit 14 was considered the EoS for these patients. If not, then patients were to have an additional 10 weeks of follow-up and an EoS at visit 16 (week 16 to 28).
Patients who received rescue treatment with OL spesolimab between weeks 7 and 12 were to be followed for an additional 6 weeks and were to have a response evaluation at visit 15 (week 13 to 18); these patients did not have a visit 14. If, at visit 15, patients qualified to enter the OLE trial, then visit 15 was to be considered EoS for these patients; if not, the patients were to have an additional 10 weeks of follow-up and an EoS at visit 16 (week 16 to 28).
Patients who did not qualify to enter the OLE trial were to be followed for 16 weeks (EoS at visit 16, week 16 to 28) after the last dose of trial medication, which was the latest time point for trial medication given during the study (i.e., the latest of day 1 or day 8 if OL spesolimab or rescue with OL spesolimab was given).
Sources: Bachelez et al. (2021)23 and Effisayil 1 Clinical Study Report.16
Figure 2: Effisayil 2 Trial Study Design
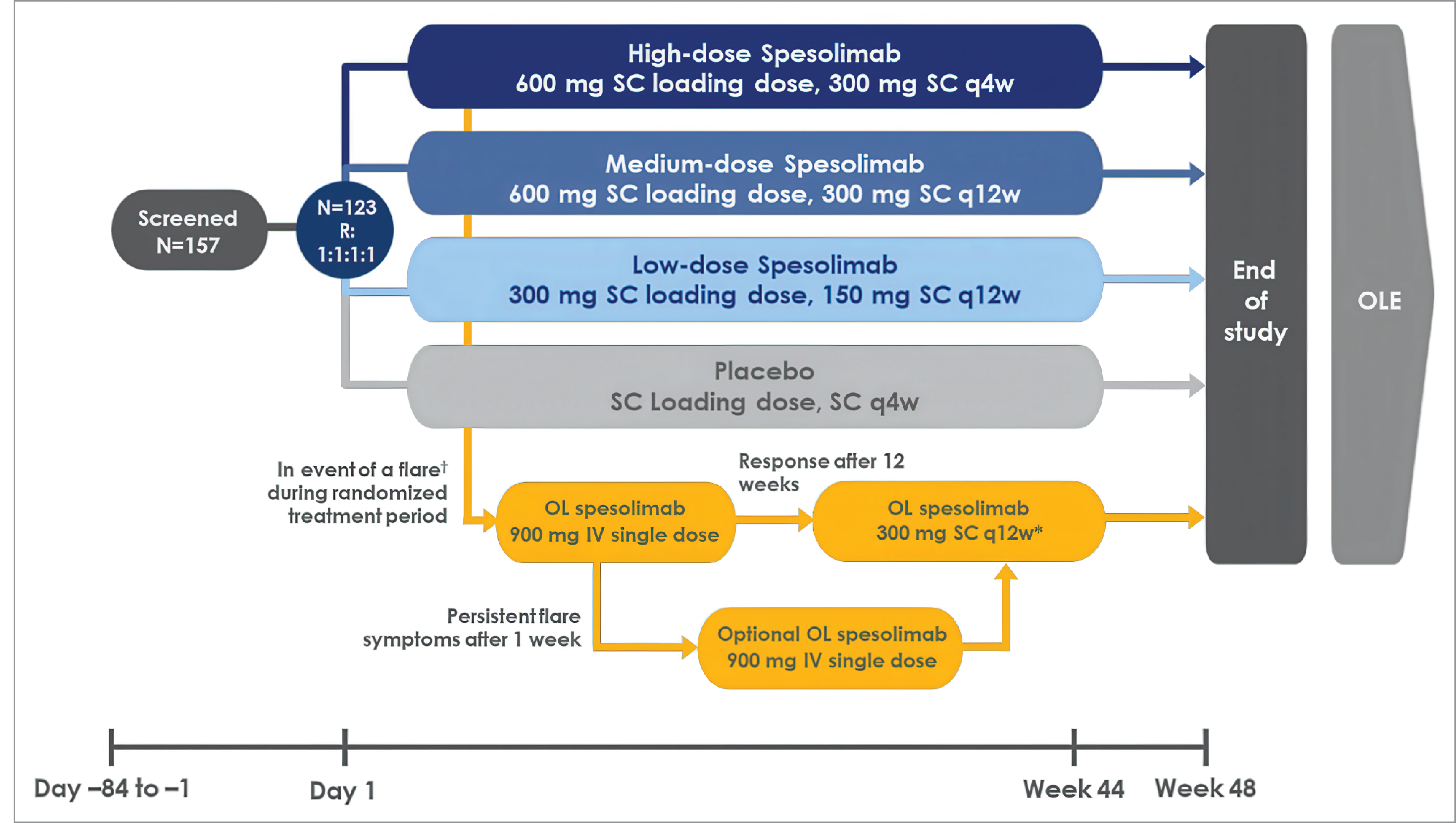
GPPGA = Generalized Pustular Psoriasis Physician Global Assessment; OL = open label; OLE = open-label extension; q4w = every 4 weeks; q12w = every 12 weeks; R = randomization; SC = subcutaneous.
†Defined as an increase in GPPGA score by ≥ 2 from baseline and a score of ≥ 2 in the GPPGA pustular component. In the event of persistent flare symptoms, defined as either a GPPGA score of ≥ 3 and GPPGA pustular component score of ≥ 2 at day 1 (where the total GPPGA was ≥ 2 and the GPPGA pustular component was ≥ 2 at day 8), or a GPPGA score of 2 and a GPPGA pustular component score of ≥ 2 at day 1 (where the GPPGA pustular component score was ≥ 2 at day 8), a patient would receive an additional rescue treatment with an OL IV dose of 900 mg of spesolimab after 1 week.
*Throughout the maintenance treatment, an SC injection of OL spesolimab 300 mg was given every 12 weeks. If the patient’s GPPGA total score increased by ≥ 1 (with or without the presence or new appearance of pustules), or if there was a ≥ 1 increase in the GPPGA pustular component score, the investigator could treat the patient with intensified maintenance therapy with OL spesolimab SC 300 mg every 4 weeks.
Sources: Morita et al. (2023)26 and Effisayil 2 Clinical Study Report.18
Interventions
Effisayil 1 Trial
Patients were randomized 1:1 to receive 1 of the following treatment options:
spesolimab 900 mg (single-dose IV infusion)
placebo (solution for IV infusion).
Treatment completion was defined as patients receiving the whole prepared dose on day 1.
Figure 3: Effisayil 2 Trial Study Design (in the Event a Patient Experiences Their First GPP Flare)
@ = the day of administration of rescue treatment: day 1. All subsequent study days were counted from this day 1 except for ‘Last dose’ for the maintenance treatment, EoS1 and EoS2; * = dosing days; BI 655130 = spesolimab; D = day; EoS = end of study; F = flare; GPP = generalized pustular psoriasis; GPPGA = Generalized Pustular Psoriasis Physician Global Assessment; OL = open label; OLE = open-label extension; q12wks = every 12 weeks; R = randomization; s.c. = subcutaneous; SOC = standard of care; V = visit; W = week.
1First GPP flare defined as an increase in GPPGA score by ≥ 2 from baseline and a score of ≥ 2 in the pustular component of the GPPGA.
2“Week” represented the end of each week (e.g., end of week 1 = day 8, end of week 2 = day 15, end of week 3 = day 22, end of week 4 = day 29).
3Investigator could treat the patient with intensified maintenance therapy with OL spesolimab SC 300 mg every 4 weeks if the patient met protocol-specified criteria.
4Patients who received rescue treatment with spesolimab up to week 34 (day 239) of randomization started the OL SC maintenance treatment 12 weeks later following the response of the GPP flare. The day of administration of rescue treatment = day 1. All subsequent study days were counted from this day 1, except for “last dose” for the maintenance treatment, EoS1, and EoS2.
5EoS1: Visit 14 was recorded as the EoS visit (i.e., EoS1) for patients who qualified and agreed to enter the OLE trial (1368 to 0025). Visit 14 was also recorded as an EoS visit for patients who prematurely discontinued with the last dose of treatment up to and including day 232 and who agreed to complete all remaining study visits up to week 48 from randomization. Since these patients discontinued prematurely, they did not qualify to enter the OLE trial.
6EoS2 was applicable for patients who did not qualify for or did not agree to enter the OLE trial (1368 to 0025) at week 48. EoS2 was also applicable for patients who prematurely discontinued with the last dose of treatment after day 232. Since these patients discontinued prematurely, they did not qualify to enter the OLE trial.
Sources: Morita et al. (2023)26 and Effisayil 2 Clinical Study Report.18
Escape treatment was permitted for patients experiencing a deterioration of the disease necessitating immediate intervention during week 1, as well as for those experiencing disease worsening who did not meet the criteria for receiving rescue medication with OL spesolimab after week 1. Typically, this would involve a standard of care (SOC) treatment chosen by the treating physician during and after the first week for patients who do not qualify for rescue medication with OL spesolimab. At day 8, patients were eligible for a single dose of 900 mg of OL spesolimab IV if they still had an unresolved flare (i.e., a GPPGA total score and GPPGA pustular component score ≥ 2) after receiving the initial dose of spesolimab IV and if they had not received escape treatment. After day 8 and through week 12, only 1 dose of OL single-dose 900 mg spesolimab IV was permitted for patients in the case of a recurrence of a GPP flare, provided they had previously achieved a clinical response (i.e., GPPGA of 0 or 1) to spesolimab, placebo, or SOC. Subsequent flares were to be treated with SOC, according to the physician’s choice.17
In case of safety concerns (e.g., systemic hypersensitivity or infusion reactions), investigators could slow down or temporarily stop the infusion. However, the infusion duration could not exceed 180 minutes. In instances where anaphylactic reactions were suspected to be caused by the study medication, the investigator was permitted to discontinue treatment with spesolimab prematurely. No additional treatment was planned. However, if AEs required treatment, the investigator could use symptomatic therapy. All treatments were administered in a hospital setting during the trial.
Follow-up duration varied across participants and depended on whether patients needed rescue treatment with OL spesolimab after week 1 (day 8) and through week 12. Patients transitioning to the OLE trial had a 12-week follow-up period, while patients who needed rescue medication with spesolimab (administered at weeks 7 and 12) had a total follow-up of 18 weeks (due to the implementation of a 6-week extension of follow-up after the intervention was administered). For patients who did not participate in the OLE, the follow-up period lasted 16 weeks after the last spesolimab dose, and the total follow-up period was up to 28 weeks (encompassing the randomized treatment on day 1, OL spesolimab on day 8, or rescue medication after day 8).17
Concomitant Medications
Patients were permitted to take stable doses of concomitant therapies for chronic conditions, and neither the condition nor the treatment were judged to exclude the patient from participation in the trial. Patients could receive medications and study interventions unless they were listed as prohibited or alternative treatments. Methotrexate, cyclosporine, and retinoids had to be discontinued before receiving the first dose of spesolimab or placebo and were not allowed after visit 2; systemic corticosteroids were not allowed 30 days before visit 2 and through the end of study visit; inhaled corticosteroids to treat asthma and corticosteroid drops administered in the eye or ear were allowed. Other restricted medications included biologics, phototherapy, topical corticosteroids, systemic immunomodulating treatments (e.g., corticosteroids, cyclophosphamide), tofacitinib, apremilast, and other systemic psoriasis treatments (e.g., fumarates, any other drug known to possibly have any benefit in the treatment of psoriasis) photochemotherapy (e.g., psoralen plus UV A [PUVA]), IL-36R inhibitors, and investigational products for psoriasis.17
Effisayil 2 Trial
Patients were randomized (1:1:1:1) to receive 1 of the following treatment options:
Spesolimab LD: Spesolimab 300 mg loading dose followed by maintenance treatment of 150 mg every 12 weeks as SC injections.
Spesolimab MD: Spesolimab 600 mg loading dose followed by maintenance treatment of 300 mg every 12 weeks as SC injections.
Spesolimab HD: Spesolimab 600 mg loading dose followed by maintenance treatment of 300 mg every 4 weeks as SC injections.
Placebo: SC injections.
During the randomized maintenance period, patients experiencing a first GPP flare (i.e., an increase from baseline in the overall GPPGA score by 2 or more points from baseline and a score of 2 or more in the pustular component of the GPPGA) were permitted to receive either a rescue treatment (spesolimab 900 mg IV infusion) or investigator-prescribed SOC if the patient did not qualify for rescue treatment. If flare symptoms persisted after a week, patients were allowed to receive another rescue treatment with an OL spesolimab IV dose of 900 mg. OL spesolimab SC doses of 300 mg every 12 weeks (with possible escalation of frequency to spesolimab SC 300 mg every 4 weeks) were offered to patients whose GPP flare responded to rescue treatment after 12 weeks.17
Unblinding by the investigator or a designee through the interactive response technology system was permitted in emergencies. To maintain the blinding for treatment, all patients received the blinded treatments every 4 weeks. Treatment was discontinued upon the initiation of investigator-prescribed SOC, except for specific treatments such as topical corticosteroids, methotrexate, cyclosporine, and retinoids during the OL flare treatment period (occurring 4 weeks following an IV dose of rescue treatment with OL spesolimab at day 1). These specific treatments were also allowed during the OL maintenance treatment period in response to a GPP flare. In instances where anaphylactic reactions occurred that were suspected to be caused by the study medication, the investigator had the authority to discontinue treatment with spesolimab prematurely. No additional treatment was planned; however, if AEs required treatment, the investigator could use symptomatic therapy.17
A follow-up period of 16 weeks after the last dose of the trial medication (i.e., the residual-effect period) was designed for patients who did not qualify for or declined to participate in the OLE trial. If a patient experienced an initial GPP flare during the randomized maintenance treatment period, a rescue treatment was initiated, followed by a maintenance treatment. Treatment involved an OL spesolimab IV dose of 900 mg followed by 300 mg of OL spesolimab SC every 12 weeks if there was a response after 12 weeks. Patients also had the option to escalate to 300 mg SC every 4 weeks if there was an increase in the pustular component of the GPPGA of 1 or greater, or an increase in the GPPGA total score from any of the previous OL visits. In the event of persistent flare symptoms, patients could receive another rescue treatment with an OL spesolimab IV dose of 900 mg after 1 week. In the randomized maintenance treatment phase, if a patient received any SOC prescribed by the investigator due to the worsening of GPP disease, they were required to discontinue the trial treatment, with some exceptions.17
Concomitant Medications
Aside from the study interventions, concomitant medication use was permitted if these medications did not interfere with the investigational medicinal product or other trial treatment, according to the investigator, and unless they were specifically listed as prohibited medications or alternative treatments. Restricted medications included biologics, phototherapy, topical corticosteroids, systemic immunomodulating treatments (e.g., corticosteroids and cyclophosphamide), tofacitinib, apremilast, and other systemic psoriasis treatments (e.g., fumarates and any other drug known to possibly have any benefit in the treatment of psoriasis) photochemotherapy (e.g., PUVA), IL-36R inhibitors, or investigational products for psoriasis.
Outcomes
A list of efficacy end points assessed in this Clinical Review Report is provided in Table 7, followed by descriptions of the outcome measures. The summarized end points are based on outcomes included in the sponsor’s summary of clinical evidence and any outcomes identified as important to this review according to the clinical experts consulted by CDA-AMC and the input from the patient and clinician groups and public drug plans. The clinical experts consulted generally considered a GPPGA pustulation subscore of 0 (no visible pustules) and a GPPGA total score of 0 or 1 (clear or almost clear) as clinically important outcomes to assess treatment efficacy for patients presenting with acute GPP flares and considered the duration of the Effisayil 1 trial to have been appropriate to treat flares. Time to first GPP flare (defined as an increase in the GPPGA score of 2 or more from baseline and a score of 2 or greater in the GPPGA pustular component) and the proportion of patients with 1 or more GPP flares up to week 48 was also considered critical for flare prevention, and 48 weeks for the Effisayil 2 trial’s duration was considered appropriate. Pain VAS was considered an important patient-reported outcome for acute flare treatment because it aligns with changes in the GPPGA pustulation subscore during treatment. The DLQI questionnaire was considered an important HRQoL outcome to supplement other outcomes for flare prevention due to the non–disease specific list of questions measuring other aspects of the patient’s quality of life. Using the same considerations, the review team selected end points considered most relevant to inform expert committee deliberations and finalized this list of end points in consultation with members of the expert committee. The following outcomes were considered: the proportion of patients with a GPPGA pustulation subscore of 0, the proportion of patients with a GPPGA total score of 0 or 1, CFB in pain VAS scores, time to the first GPP flare, the proportion of patients with the occurrence of 1 or more GPP flares, time to worsening of DLQI, and SAEs.
Table 7: Outcomes Summarized From the Studies Included in the Systematic Review
Outcome measure | Time point | Hierarchy |
|---|---|---|
Effisayil 1 trial | ||
Proportion of patients with a GPPGA pustulation subscore of 0 | At week 1 | Primarya |
Proportion of patients with a GPPGA total score of 0 or 1 | At week 1 | Key secondarya |
CFB in VAS score | At week 4 | Secondarya |
Effisayil 2 trial | ||
Time to first GPP flare | Up to week 48 | Primarya (secondary objective)b |
Proportion of patients with the occurrence of ≥ 1 GPP flare | Up to week 48 | Key secondarya |
Time to first worsening of DLQI | Up to week 48 | Secondarya |
CFB = change from baseline; DLQI = Dermatology Life Quality Index; FACIT = Functional Assessment of Chronic Illness Therapy; GPP = generalized pustular psoriasis; GPPGA = Generalized Pustular Psoriasis Physician Global Assessment; VAS = visual analogue scale.
aStatistical testing for these end points was adjusted for multiple comparisons (i.e., hierarchal testing). Of note is that the order of the outcomes presented in this table represents the order in which the hierarchical testing was performed.
bThe secondary objective was to demonstrate superiority vs. placebo for high-dose spesolimab (300 mg every 4 weeks) and medium-dose spesolimab (300 mg every 12 weeks) on the primary end point, the time to the onset of the first GPP flare up to week 48 and, for the key secondary end point, the occurrence of at least 1 GPP flare up to week 48.
Sources: Effisayil 116 and Effisayil 218 Clinical Study Reports.
Effisayil 1 Trial
Generalized Pustular Psoriasis Physician Global Assessment
The proportion of patients with a GPPGA pustulation subscore of 0 and a GPPGA total score of 0 or 1 at week 1 were the primary and key secondary end points, respectively, of the Effisayil 1 trial.
The GPPGA relies on a clinical assessment of the skin presentation of the patient with GPP. It is a modified Physician’s Global Assessment, a physician’s assessment of psoriatic lesions adapted to evaluate patients with GPP. The investigator scored 3 components: erythema, pustules, and scaling of all GPP lesions from 0 (clear) to 4 (severe). Each of the 3 components was graded separately and averaged, and the final GPPGA was determined from the composite score. A lower score indicated lesser severity, with 0 being clear and 1 being almost clear.30 A score of 2 indicates mild disease, defined by bright red erythema and discrete, grouped, moderate-density pustules (noncoalescent) and predominantly fine scaling or crusting; a score of 3 indicates moderate disease (meaning bright red erythema, with high-density pustules with some coalescence, and moderate scaling or crusting covering most or all lesions), and a score of 4 indicates severe disease (meaning deep fiery red erythema, very high density pustules with pustular lakes and severe scaling or crusting covering most or all lesions).
The GPPGA scale is a validated measure, with intrarater and interrater analyses confirming GPPGA as a suitable study end point for GPP clinical trials. In a validation study involving 20 dermatologists, intrarater reliability was deemed excellent (0.87 to 0.90), while interrater reliability among 26 dermatologists ranged from excellent (0.76 to 0.82) for absolute agreement and erythema and scaling, to good for pustulation (0.69).31 Additionally, psychometric analyses conducted on the GPPGA total score using data from week 1 of the Effisayil 1 study showed good test-retest reliability for both the GPPGA total score and pustulation subscore (intraclass correlation coefficient of 0.70 and 0.91, respectively), along with good evidence of convergent validity.32 These analyses demonstrated that these scores could effectively detect changes over time in symptom and disease severity. The psychometric validation of GPPGA total score, GPPGA pustulation subscore, and GPPASI total score was assessed in a study by Burden et al. (2023)30 using data from the Effisayil 1 study. The authors concluded that these outcomes could detect changes in symptom and disease severity. A statistical significance was observed for the least squares mean change scores from baseline between the improved and not improved patient subgroups assessed for the scores for 3 corresponding patient-reported questionnaires: Clinical Global Impression–Improvement, DLQI item 1, and the EQ-5D pain/discomfort domain. Findings from the anchor-based analysis showed a range of clinically meaningful changes in scores from baseline, as follows. The change in score from baseline ranged from −1.34 to −1.56 for the GPPGA total score, −2.11 to −2.30 for the GPPGA pustulation subscore, and −10.82 to −12.65 for the GPPASI total score. The authors concluded that mean reductions of −1.4, −2.2, and −12.0 suggest a clinically meaningful threshold for the GPPGA total score, GPPGA pustulation subscore, and GPPASI total score, respectively.32
Pain Visual Analogue Scale
The CFB in the pain VAS score at week 4 was a secondary end point of the Effisayil 1 trial. The pain VAS is a unidimensional measure of pain intensity.33 It is a continuous VAS comprising a horizontal or vertical line, usually 10 cm (100 mm) in length, anchored by word descriptors at each end (no pain, very severe pain). The pain VAS was to be self-completed by the respondent. The respondent was asked to place a vertical (|) mark on the horizontal line to indicate the severity of the pain. Using a ruler, the score was determined by measuring the distance (mm) on the 10 cm line between the no pain anchor and the patient’s mark, providing a range of scores from 0 to 100. A higher score indicated greater pain intensity, while a negative CFB signified an improvement from baseline.
Data from the Effisayil 1 trial was used to inform the psychometric properties of the pain VAS scale in a GPP population. The results support this measure’s reliability, validity, and ability to detect changes in symptom severity in patients with GPP.32 No established minimal important differences (MIDs) were identified for the pain VAS scale in GPP populations.
Safety
The main safety variables in the Effisayil 1 trial included adverse events of special interest (AESIs), treatment-emergent AEs (TEAE), SAEs, AEs leading to discontinuation, and AEs resulting in a fatal outcome. The severity of AEs was graded according to Rheumatology Common Toxicity Criteria version 2.0. The proportion of patients experiencing an AESI was monitored throughout the study and included the following, as prespecified in the protocol: hepatic injuries, systemic hypersensitivity reaction (including infusion reactions and anaphylactic reactions), severe infections, and opportunistic and Mycobacterium tuberculosis infections.17
Effisayil 2 Trial
Generalized Pustular Psoriasis Physician Global Assessment
The primary end point of the Effisayil 2 study was time to first GPP flare, while the key secondary end point was the proportion of patients experiencing at least 1 GPP flare. A GPP flare in the Effisayil 2 trial was defined as an increase from baseline of 2 or more points in the GPPGA total score and a score of 2 or greater in the pustular component of the GPPGA. The use of rescue medication or investigator-prescribed SOC for GPP worsening indicated the onset of a GPP flare.17
Dermatology Quality of Life Index
The time to first worsening in the DLQI was a secondary end point of the Effisayil 2 study. Worsening of the DLQI was defined as a 4-point increase in total score from baseline. Intake of rescue medication, or investigator-prescribed SOC, was considered to be the onset of a worsening. The DLQI is a patient-administered, 10-question quality of life questionnaire covering 6 domains, including symptoms and feelings, daily activities, leisure, work and school, personal relationships, and treatment. The DLQI has a 1-week recall period. Response categories include not relevant (score of 0), not at all (score of 0), a little (score of 1), a lot (score of 2), and very much (score of 3). Question 7 in the DQLI questionnaire was a yes or no question (“Over the last week, has your skin prevented you from working or studying?”), where yes was given a score of 3. DLQI total score was calculated by summing the scores of each question, resulting in a range of 0 to 30, where 0 to 1 equalled no effect on a patient’s life, 2 to 5 equalled a small effect, 6 to 10 equalled a moderate effect, 11 to 20 equalled a very large effect, and 21 to 30 equalled an extremely large effect on a patient’s life. The higher the score, the more the quality of life was impaired. In this trial, a change in score of 4 points was considered the MID, aligning with the recommendation for general inflammatory skin conditions where a change in DLQI score of at least 4 points is considered clinically important.34
Safety
The main safety variables in the Effisayil 2 trial included the following treatment-emergent AEs, SAEs, AEs leading to discontinuation, and AEs resulting in a fatal outcome. The severity of AEs was graded according to Rheumatology Common Toxicity Criteria version 2.0. The proportion of patients experiencing an AESI was monitored throughout the study and included the following, as prespecified in the protocol: hepatic injuries, systemic hypersensitivity reaction (including infusion reactions and anaphylactic reaction), severe infections, and opportunistic and Mycobacterium tuberculosis infections.17
Statistical Analysis
Definitions of estimand and imputation methods for the Effisayil 1 and Effisayil 2 trials are presented in Table 8.
Sample Size and Power Calculation
Effisayil 1 Trial
Power calculations for sample size assessment in the Effisayil 1 trial were based on the primary and key secondary end points, and the correlation of the 2 end points was set to be 0.65, which was obtained from efficacy data of the proof-of-concept study.35
The superiority of spesolimab relative to placebo was assessed for 2 outcomes: the proportion of patients achieving a GPPGA pustulation subscore of 0 at week 1 (primary end point) and the proportion of patients achieving a GPPGA total score of 0 or 1 at week 1 (secondary end point). Confirmation of efficacy was reported if the null hypothesis for the primary end point was rejected. Subsequently, a treatment effect on the key secondary end point was to be tested. In a scenario where the null hypothesis tests for both outcomes (primary and key secondary) were rejected, testing of the treatment effect for the other 4 secondary end points was designed hierarchically based on the following assumptions: at least 60% of patients would have a GPPGA pustulation subscore of 0, at least 60% of patients would have a GPPGA total score of 0 or 1 after 1 week following treatment with spesolimab, and only 10% of patients treated with placebo would present a response to each of the primary and key secondary end points at week 1. Assuming that the expected response rates for the primary and key secondary end points were 0.55 for spesolimab and 0.1 for placebo, the overall power to achieve both end points was approximately 87.7%.17
Therefore, assuming that the expected response rates for both the primary and key secondary end points were 0.6 for spesolimab and 0.1 for placebo, a total sample size of 51 patients was established, and randomization was set at a 2:1 ratio to provide 93.9% power to detect a clinically relevant difference between the spesolimab and placebo groups, respectively, at a 1-sided type I error of 0.025.17
Table 8: Definitions of Estimand and Imputation Methods in the Effisayil 1 and Effisayil 2 Trials
Analyses | Definition |
|---|---|
Effisayil 1 trial | |
EN | Nonresponse was assigned to patients with any of the following before observing the end point: death, any use of escape medication, OL spesolimab use on day 8, and rescue medication with spesolimab. |
EN-PC | EN with the addition of censoring any data after the use of restricted medication for other purposes. |
EC | Any data after the following was censored: death, any use of escape medication, OL spesolimab use on day 8, rescue medication with spesolimab. |
Imputation methods for the Effisayil 1 trial | |
NRI | If data were available from the visits both before and after the visit with a missing outcome, imputation as a success was performed when both neighbouring visits also represented a success and there was no use of escape medication, OL spesolimab on day 8, or rescue medication with spesolimab within this imputation period; otherwise, imputation was performed as a failure to achieve a response (nonresponse imputation). |
LOCF | The missing outcome was imputed as the last available value (including baseline) before the missing outcome and before the occurrence of any applicable intercurrent event (i.e., death, the use of escape medication, OL spesolimab on day 8, or rescue medication with spesolimab). |
BRI | All missing values were imputed based on the best response observed at visits conducted before withdrawal or occurrence of missing data. If no nonmissing data were available, then the missing value was imputed as a nonresponse. |
Mixed-effect model for repeated measures | |
EM | If < 3 patients had missing data on an end point at week 1, then a list of all possible treatment differences was generated whereby each of the potential responses (response, nonresponse) was imputed for each patient in an exhaustive manner. The primary end point analysis was repeated for each possible combination of imputed values on missing responses and the results were summarized in a single table. |
Imputation methods for the Effisayil 2 trial | |
EM | Use of investigator-prescribed SOC for GPP or use of OL spesolimab IV for GPP flare treatment regarded as an event or treatment failure. |
EMR | Use of rescue medication with spesolimab IV, or investigator−prescribed SOC is considered as GPP flare; data after use of restricted medication for other indications is censored. |
ET | Only use of rescue medication with spesolimab IV is censored. |
MI | Method using sequential logistic regression method. |
PM | Censoring is made at the earliest date of EoS; day 351 if no intercurrent event. |
SM | Censoring is made at the last assessment before 2 or more consecutive missing assessments. |
EoS = end of study; GPP = generalized pustular psoriasis; LOCF = last observation carried forward; NRI = nonresponse imputation: OL = open label; SOC = standard of care.
Sources: Effisayil 116 and Effisayil 218 Clinical Study Reports.
Effisayil 2 Trial
Power calculations for sample size in the Effisayil 2 trial were based on the dose-finding analysis (primary objective) and the secondary objective, i.e., the superiority of spesolimab HD (300 mg every 4 weeks) and spesolimab MD (300 mg every 12 weeks) versus placebo on the primary end point, time to first GPP flare onset up to week 48, as well as the key secondary end point, the occurrence of at least 1 GPP flare up to week 48. The effect of each dose of spesolimab in preventing a GPP flare in patients with a history of GPP flares was compared with placebo. The following assumptions were outlined: spesolimab HD and MD were assumed to be closely effective, while the lowest dose of spesolimab was assumed to be a subtherapeutic dose. Given that most patients who were enrolled in the Effisayil 2 trial had withdrawn from prior GPP medication, the overall GPP flare rate was expected to be higher for patients within the first 3 months of the maintenance treatment period (withdrawal of previous medication was considered a potential trigger for the onset of a GPP flare event).17
The following assumptions were made in the base scenario. The overall HR between spesolimab HD (300 mg every 4 weeks) and placebo was 0.1, the HR between spesolimab MD (300 mg every 12 weeks) and placebo was 0.15, and the HR between spesolimab LD (150 mg every 12 weeks) and placebo was 0.3. A piecewise exponential distribution assumption was established (i.e., first 3 months versus post 3 months) for the time to first GPP flare in each arm of the trial. The analysis anticipated that within the first 3 months of treatment with spesolimab HD, spesolimab MD, and spesolimab LD, approximately 1.7%, 2.6%, and 5.1% of patients, respectively, would experience a first GPP flare per month under the base scenario. In contrast, the rate of first flare on placebo within the first 3 months was expected to be higher, i.e., approximately 17.1% per month under the base scenario. After 3 months, the overall rate of first flare was expected to decline and stabilize for the remainder of the trial. The expected rate of first flare after 3 months was 0.3% per month for patients on spesolimab HD, 0.4% per month for patients on spesolimab MD, 0.9% per month for patients on spesolimab LD, and 3% per month for patients on placebo under the base scenario.17
A total sample size of 120 patients, randomized 1:1:1:1, would provide sufficient power to test a clinically relevant difference between the different doses of spesolimab against placebo at a 1-sided type I error of 0.05. The power of the test was driven by the number of events truly observed at the time of the primary analysis. The primary analysis was scheduled when the last patient completed the trial or when early discontinuation occurred after the 48-week treatment. The target number of events was 27. The probability of trial success was defined as the probability of obtaining a significant test for a non-flat dose-response curve, and the probability of success was approximately 99.5% for the base scenario.17
Statistical Testing
Effisayil 1 Trial
Binary end points (proportion of patients with a GPPGA pustulation subscore of 0, GPPGA total score of 0 or 1, and a 75% reduction in the GPPASI) were estimated using the Wilson method and analyses were conducted on the ITT population using an exact Suissa and Shuster z-pooled test at a 1-sided alpha of 0.025 (the 2-sided P value was reported by doubling the 1-sided P value of < 0.1). The 95% CIs around risk differences were estimated using the Chan and Zhang method.17
Secondary end points with continuous data (CFB in the pain VAS, PSS, and FACIT-Fatigue scores) were estimated using the modified Hodges-Lehmann estimate of the median difference and 95% CIs. Treatment groups were compared using the Wilcoxon rank testing method, and a 1-sided P value was calculated. Worst-case ranks were assigned to those who had died; had prior use of escape medication, OL spesolimab on day 8, or rescue medication with spesolimab; and to patients with missing data at week 4 for other reasons. The best possible baseline values for the pain VAS and PSS were 0 (the lowest value) and 52 (the highest value) for the FACIT-Fatigue scale. The worst possible postbaseline values were 100 for the pain VAS (the highest value), 16 for the PSS (the highest value), and 0 (the lowest value) for the FACIT-Fatigue scale. Therefore, the maximum value for the worst possible CFB (i.e., the worst possible postbaseline value minus the best possible baseline value) was 100 for pain VAS, 16 for PSS, and −52 for the FACIT-Fatigue scale. Continuous data were presented using descriptive statistics. Of note, all analyses were unadjusted. No interim analysis was conducted. Formal statistical hypothesis testing was performed at an overall 1-sided alpha level of 0.025. The analyses of the secondary end points and further end points that were not included in the hierarchical testing strategy were considered exploratory in nature.17
Data Imputation Methods: Several approaches were implemented to assess the impact of missing data on the efficacy end points defined in the Effisayil 1 trial for the primary and key secondary outcomes. Missing data imputations were performed using all available on-treatment data observed up to the respective analysis cut-off date. Imputations for binary outcomes were planned only at the binary level, i.e., the GPPGA total score itself was not imputed, but the binary end points derived based on these scores were imputed unless otherwise specified. For the binary efficacy end points for a randomized dose at day 1, the following primary imputation strategy (nonresponse imputation [NRI]) was performed after the implementation of the estimand concept: for end points that are measured at multiple visits, if there are available data at the visits both before and after the visit with a missing outcome, then impute as a success only if both neighbouring visits also represent a success. Otherwise, it was imputed as a failure to achieve a response (i.e., NRI).
Continuous efficacy end points for a randomized dose at day 1 were also analyzed.17 For the primary estimand of continuous end points, death or any use of escape medication, OL spesolimab at day 8, or any rescue medication with spesolimab before observing the end point was considered a nonresponse. The outcome of nonresponse itself was not a missing value but considered the worst possible outcome of the corresponding continuous end point. The LOCF was the primary imputation strategy to account for missing values for continuous end points following the implementation of the estimand concept. Missing data were imputed as the last available value (including baseline) before the missing outcome. The Wilcoxon rank test was used to estimate the treatment effect and assess continuous secondary end points at week 1 or week 4. Worst-case ranks were assigned to patients who had died or had used prior escape medication, OL spesolimab at day 8, or rescue medication with spesolimab.17
Sensitivity Analyses: Three sensitivity analyses were carried out on the primary end point as follows:17
Sensitivity analysis 1 (EN-BRI): Included the ITT population while excluding values after the use of escape medication, OL spesolimab at day 8, or rescue medication with spesolimab (i.e., nonresponse). The best response imputation strategy was applied.
Sensitivity analysis 2 (EN-PC-NRI): Involved the ITT population, excluding values after the use of escape medication, OL spesolimab at day 8, or rescue medication with spesolimab (i.e., nonresponse). Nonresponse and data after the use of restricted medication for other purposes were excluded, and the NRI strategy for any missing data was applied.
Sensitivity analysis 3 (EN-NRI): Focused on the per-protocol population, excluding values after the use of escape medication, OL spesolimab at day 8, or rescue medication with spesolimab (i.e., nonresponse). The NRI strategy for any missing data was applied.
Effisayil 2 Trial
The primary end point (time to first GPP flare) for the Effisayil 2 trial was estimated using the Kaplan-Meier method. The treatment difference between the spesolimab and placebo groups was estimated using the Cox regression model, and the 1-sided P value was calculated using the log-rank test. Comparisons between groups were stratified using systemic GPP medications at randomization. The estimates from the Cox regression model were then used for the non-flat dose-response assessment using the multiple comparison procedure with modelling techniques (MCP-Mod).17
The secondary end point (proportion of patients with ≥ 1 GPP flare) analysis was estimated using the Cochran-Mantel-Haenszel test, stratified by the use of systemic GPP medication at randomization (yes or no). A truncated Hochberg procedure was used to control for multiplicity at an overall 1-sided alpha level of 0.025, in the event that both doses were statistically significant during testing for the primary end point. If only 1 dose of spesolimab was statistically significant during testing, the dose would be retested at a 1-sided alpha of 0.00625. If neither dose was statistically significant and superior to placebo, no further testing in the hierarchical sequence would be conducted. The difference between groups and associated CIs were presented using a Mantel-Haenszel–type weighted average of differences using weights proposed by Greenland and Robins.36 The time-to-event secondary end point (time to worsening of PSS score and DLQI score) analyses were estimated similarly to the primary end point, except for the dose-response assessment, which applied the MCP-Mod.17
Formal testing of all trial end points in the testing hierarchy (primary, key secondary, and secondary end points) was performed at a 1-sided alpha level of 0.025. An interim analysis of the OL spesolimab IV flare treatment and subsequent maintenance treatment periods was planned to support the initial administration of spesolimab for GPP flare treatment; no unblinding of the randomized SC treatment was required.17
Data Imputation Methods: During the randomized maintenance treatment period, the GPPGA and PSS were to be assessed every 4 weeks, while DLQI was assessed at weeks 4, 8, 12, 24, 36, and 48. Patients were to make unscheduled clinical visits if GPP flares were suspected between the protocol-specified scheduled visits.17
For time-to-event end points, the primary method (PM) censored patients without an event but with at least 1 postrandomization assessment at the last contact date in the planned randomized maintenance treatment period. Patients with no postrandomization assessment and no intercurrent event were excluded from the analysis (i.e., censored at the date of randomization or the first dose, whichever was later). As a sensitivity analysis method (SM), missed postrandomization assessments were considered.
For binary end points (e.g., the key secondary end point and the last secondary end point), once a treatment failure occurred, it was assigned to all subsequent visits. A sequential logistic regression with multiple imputation (MI) methods was used for patients without documented treatment failure but had missing assessments. The imputation was done at the end point level (i.e., responder [response to treatment] or treatment failure).17
Sensitivity Analyses: Several sensitivity analyses were conducted for the primary and key secondary end points.
Sensitivity analysis 1 (EM-SM): Included the ITT population using the primary estimand (EM) where any use of rescue medication with spesolimab IV or investigator-prescribed SOC was considered a GPP flare (i.e., event or treatment failure). Censoring was applied at the last assessment before 2 or more consecutive missing assessments.
Sensitivity analysis 2 (EM-PM): The same estimand and censoring method was used as the primary analysis but focused on the per-protocol population.
Sensitivity analysis 3 (EMR-PM): Included the ITT population using a secondary estimand (EMR) where any use of rescue medication with spesolimab IV or investigator-prescribed SOC was considered a GPP flare (i.e., event or treatment failure) and data after use of restricted medication for indications other than GPP were censored. The censoring method is consistent with the primary analysis.
Sensitivity analysis 4 (ET-PM): Included the ITT population using a secondary estimand (ET) where only the use of rescue medication with spesolimab IV was censored. The censoring method is consistent with the primary analysis.
Analysis Populations
Results are reported for the enrolled set, ITT (randomized set), per-protocol set, and safety analysis set populations for the Effisayil 1 and Effisayil 2 trials. The analysis sets are summarized in Table 9.
Table 9: Analysis Populations of the Effisayil 1 and Effisayil 2 Trials in the Randomized Set
Study | Population | Definition | Application |
|---|---|---|---|
Effisayil 1 | ES | All patients who signed informed consent. | This set was used for the analyses of patient disposition. |
RS (ITT) | All randomized patients. | This was the main set for the analyses of efficacy end points. It was also used for the analyses of demographics, baseline characteristics, concomitant medication, and DNA sequencing results. | |
SAS (safety) | All patients who were randomized and received at least 1 dose of the study drug. | This set was the main analysis set for safety. | |
PPS (PP) | All patients in the RS who adhered to the CTP without any iPDs which led to exclusion from the PPS. | This set was used for sensitivity analyses on the primary and key secondary efficacy end points. | |
Effisayil 2 | ES | All enrolled patients. | This set was used for disposition data. |
RS (ITT) | All randomized patients. | This was the main analysis set for presentation of efficacy. | |
PPS (PP) | All patients in the ITT who adhered to the CTP without any IPVs. | This set was used for the sensitivity analyses on the primary and key secondary end points. | |
SAS (safety) | All patients who were randomized and received at least 1 dose of study drug. | This set was used for the safety analyses. |
CTP = clinical trial protocol; ES = enrolled set; iPD = important protocol deviation; IPV = important protocol violation; ITT = intention to treat; PP = per protocol; PPS = per-protocol set; RS = randomized set; SAS = safety analysis set.
Sources: Effisayil 116 and Effisayil 218 Clinical Study Reports.
As described previously, safety analyses were performed on the safety analysis set. Different analysis periods were taken into account, as follows:
Before nonrandomized administration of spesolimab up to week 1 (week 1 analysis): Included AEs starting or worsening from the start of treatment to day 8 or end of study, whichever was earlier.
After any spesolimab administration up to week 12 (week 12 analysis): Included only patients who received any spesolimab verum (double-blind or nonrandomized). Events were included until 16 weeks after the last spesolimab administration (i.e., up to week 12 in addition to the residual-effect period), end of study, or treatment in the extension trial, whichever was earlier.
Results
Patient Disposition
Effisayil 1 Trial
A summary of patient disposition in the Effisayil 1 trial is presented in Table 10.
In the Effisayil 1 trial, of the 85 patients screened, 53 patients (62%) who presented with a GPP flare of moderate to severe intensity were randomized. The most common reason for not entering patients in the trial was “other” (17 patients), with the main reason being that a global recruitment target was achieved before the patients experienced a flare. The second most common reason was “failure to meet randomization criteria” (13 patients), with the main reason being that inclusion and/or exclusion criteria had not been met (e.g., patient did not develop a flare within 6 months). A total of 39 patients, 27 (77.1%) in the spesolimab group and 12 (66.7%) in the placebo group, rolled over into the OLE trial.17
Table 10: Summary of Patient Disposition From the Effisayil 1 Trial (Treatment Phase and Follow-Up Period)
Patient disposition | Spesolimab | Placebo |
|---|---|---|
Screened, N | 85 | |
Not entered or randomized, N | 32 | |
Randomized, N | 35 | 18 |
Treated n (%) | 35 (100.0) | 18 (100.0) |
Received OL treatment with spesolimab on day 8 | 12 (34.3) | 15 (83.3) |
Received rescue treatment with spesolimab | 4 (11.4) | 2 (11.1) |
Discontinued study, n (%) | 3 (8.6) | 1 (5.6) |
Reason for discontinuation,a n (%) | ||
Withdrawal by patient | 2 (5.7) | 1 (5.6) |
Other | 1 (2.9) | 0 |
Completed trial, n (%) | 32 (91.4) | 17 (94.4) |
Continued in the extension study, n (%) | 27 (77.1) | 12 (66.7) |
ITT, n (%) | 35 (100) | 18 (100) |
PP, n (%) | 34 (97.1) | 18 (100) |
Safetyn (%) | 35 (100) | 18 (100) |
ITT = intention to treat; OL = open label; PP = per protocol.
aThe Effisayil 1 trial included patients who prematurely discontinued the trial, corresponding to those who did not complete the follow-up period (i.e., the planned observation period).
Sources: Effisayil 1 Clinical Study Report.16 Details included in the table are from the sponsor’s summary of clinical evidence.17
Effisayil 2 Trial
A summary of patient disposition in the Effisayil 2 trial is presented in Table 11.
In the Effisayil 2 trial, of the 157 patients screened, 123 (78%) were randomized. Of the randomized patients, 111 (90.2%) completed the trial as planned: 26 patients (86.7%) in the spesolimab HD group versus 30 patients (96.8%) in the placebo group. Of the 123 patients who were randomized, 93 (75.6%) continued in the extension trial, including 20 patients (66.7%) in the spesolimab HD group versus 26 patients (83.9%) in the placebo group.17
Table 11: Summary of Patient Disposition From the Effisayil 2 Trial (Treatment Phase)
Patient disposition | Spesolimab SC (loading dose 300 mg, 150 mg q.12.w.) (LD) (N = 31) | Spesolimab SC (loading dose 600 mg, 300 mg q.12.w.) (MD) (N = 31) | Spesolimab SC (loading dose 600 mg, 300 mg q.4.w.) (HD) (N = 30) | Placebo (N = 31) |
|---|---|---|---|---|
Screened, N | 157 | |||
Not entered or randomized, N | 34 | |||
Randomized, N | 31 | 31 | 30 | 31 |
Treated, n (%) | 31 (100.0) | 31 (100.0) | 30 (100.0) | 31 (100.0) |
Completed 48-week randomized period without flare | 23 (74.2) | 20 (64.5) | 21 (70.0) | 14 (45.2) |
Switched to OL spesolimab IV for flare | 7 (22.6) | 8 (25.8) | 2 (6.7) | 15 (48.4) |
Completed flare treatment period (12 weeks) | 6 (19.4) | 8 (25.8) | 2 (6.7) | 14 (45.2) |
Started OL SC for maintenance | 2 (6.5) | 5 (16.1) | 1 (3.3) | 12 (38.7) |
Discontinued from flare treatment period | 1 (3.2) | 0 | 0 (0.0) | 1 (3.2) |
Discontinued randomized period for reasons other than OL spesolimab IV for flare | 1 (3.2) | 3 (9.7) | 7 (23.3) | 2 (6.5) |
Adverse event | 0 | 1 (3.2) | 3 (10.0) | 0 |
Lack off efficacy | 0 | 1 (3.2) | 0 | 0 |
Withdrawal by patient | 0 | 1 (3.2) | 1 (3.3) | 0 |
Other | 1 (3.2) | 0 | 3 (10.0) | 2 (6.5) |
Discontinued study, n (%) | 4 (12.9) | 3 (9.7) | 4 (13.3) | 1 (3.2) |
Reason for discontinuation, n (%) | — | — | — | — |
Withdrawal by patient | 2 (6.5) | 1 (3.2) | 1 (3.3) | 1 (3.2) |
Other | 2 (6.5) | 2 (6.5) | 3 (10.0) | 0 |
Completed trial, n (%) | 27 (87.1) | 28 (90.3) | 26 (86.7) | 30 (96.8) |
Continued in the extension study | 24 (77.4) | 23 (74.2) | 20 (66.7) | 26 (83.9) |
ITT, n (%) | 31 (100) | 31 (100) | 30 (100) | 31 (100) |
Safety,a n (%) | 32 (103) | 31 (100) | 30 (100) | 29 (93.5) |
PP, n (%) | 31 (100) | 30 (96.8) | 29 (96.7) | 31 (100) |
HD = high dose; ITT = intention to treat; LD = low dose; MD = medium dose; OL = open label; PP = per protocol; q.4.w. = every 4 weeks; q.12.w. = every 12 weeks; SC = subcutaneous.
aIn the Effisayil 2 trial, a patient randomly assigned to the placebo group who accidentally received a single dose of spesolimab 150 mg on day 1 was assigned to the spesolimab LD group for the analyses of exposure and safety (i.e., the safety analysis set includes 30 patients in the placebo group and 32 patients in the spesolimab LD group for these analyses).
Sources: Effisayil 2 Clinical Study Report.18 Details included in the table are from the sponsor’s summary of clinical evidence.17
Baseline Characteristics
The baseline characteristics outlined in Table 12 are limited to the most relevant to this review or that were felt to affect the outcomes or interpretation of the study results.
Effisayil 1 Trial
The mean age in the spesolimab group was 43.2 years versus 42.6 years in the placebo group. In total, 60.0% and 83.3% of participants were female in the spesolimab and placebo groups, respectively. Most patients were of Asian ethnicity (spesolimab: 45.7%; placebo: 72.2%) or white (spesolimab: 54.3%; placebo: 27.8%). Overall, most patients across both treatment groups had a GPPGA total score of 3 (spesolimab: 80.0%; placebo: 83.3%) and a GPPGA pustulation subscore of 3 (spesolimab: 45.7%; placebo: 38.9%) or 4 (spesolimab: 37.1%; placebo: 33.3%). The mean GPPASI total score at baseline was 27.79 (SD = 13.44) in the spesolimab group and 24.06 (SD = 15.21) in the placebo group. The mean pain VAS score at baseline was 76.4 (SD = 16.8) in the spesolimab group and 64.6 (SD = 27.6) in the placebo group; for the PSS score, the mean score was 10.4 (SD = 3.6) in the spesolimab group and 10.3 (SD = 3.1) in the placebo group; and for the FACIT-Fatigue score, the mean was 18.1 (SD = 14.2) in the spesolimab group and 19.0 (SD = 14.9) in the placebo group at baseline. A C-reactive protein level greater than the upper limit of normal (> 10 mg/L) at baseline was reported for 20 patients (57.1%) in the spesolimab group and 13 patients (72.2%) in the placebo group.17
Effisayil 2 Trial
Baseline characteristics for all 3 doses evaluated in the Effisayil 2 trial are presented in Table 12. Key baseline characteristics for the HD (spesolimab 600 mg loading dose followed by 300 mg subcutaneously, administered every 4 weeks) group versus placebo were as follows. The patients enrolled were aged 14 to 75 years (8 patients were adolescents); the mean age at randomization in the spesolimab HD group was 40.2 (SD = 16.4) years versus 39.5 (SD = 14.0) years for placebo. A total of 70.0% versus 54.8% of patients were of Asian ethnicity, 30.0% versus 45.2% were white, 60.0% versus 58.1% were female, and 40.0% versus 41.9% were male in the spesolimab HD and placebo groups, respectively. The mean weight was 68.7 kg (SD = 22.9) versus 75.73 kg (SD = 23.92), and the mean body mass index was 25.6 kg/m2 (SD = 7.3) versus 26.9 kg/m2 (SD = 8.3) in the spesolimab HD and placebo groups, respectively. The proportion of patients who had a GPPGA pustulation score of 0 (clear) was 67.7% in both groups; the proportion of patients with a GPPGA pustulation score of 1 (almost clear) was 33.3% in the spesolimab HD group and 32.3% in the placebo group. At baseline, the mean DLQI total score was 11.1 (SD = 6.9) in the spesolimab HD group versus 7.2 (SD = 5.6) in the placebo group. Numerical differences were observed with spesolimab HD compared with placebo for race (proportion of patients who were of Asian ethnicity was 70% in the spesolimab HD group versus 54.8% in the placebo group), concurrent plaque psoriasis (23.3% in the spesolimab HD group versus 32.3% in the placebo group), presence of potentially pathogenic IL36RN variation (23.3% in the spesolimab HD group versus 12.9% in the placebo group), GPPGA total score (score of 0: 10% in the spesolimab HD group versus 12.9% in the placebo group; score of 1: 90% in the spesolimab HD group versus 87.1% in the placebo group), DLQI score (6.9% in the spesolimab HD group versus 5.6% in the placebo group), and prior use of at least 1 biologic therapy (20% in the spesolimab HD group versus 29% in the placebo group). The mean weight in the placebo group was 75.73 kg (SD = 23.92), which was higher than in the spesolimab groups combined (71.77 kg; SD = 23.21).17
Table 12: Summary of Baseline Characteristics From the Effisayil 1 and Effisayil 2 Trials
Characteristic | Effisayil 1 trial | Effisayil 2 triala | ||||
|---|---|---|---|---|---|---|
Spesolimab (900 mg single-dose IV) (N = 35) | Placebo (N = 18) | Spesolimab SC 150 mg q.12.w. (LD) (N = 31) | Spesolimab SC 300 mg q.12.w. (MD) (N = 31) | Spesolimab SC 300 mg q.4.w. (HD) (N = 30) | Placebo (N = 31) | |
Sex, n (%) | ||||||
Male | 14 (40.0) | 3 (16.7) | 11 (35.5) | 11 (35.5) | 12 (40.0) | 13 (41.9) |
Female | 21 (60.0) | 15 (83.3) | 20 (64.5) | 20 (64.5) | 18 (60.0) | 18 (58.1) |
Race, n (%) | ||||||
Asian | 16 (45.7) | 13 (72.2) | 20 (64.5) | 21 (67.7) | 21 (70.0) | 17 (54.8) |
Hispanic or Latino | ███ | ███ | █████ | ███ | █████ | █████ |
White | 19 (54.3) | 5 (27.8) | 11 (35.5) | 10 (32.3) | 9 (30.0) | 14 (45.2) |
Age (years) | ||||||
Mean (SD) | 43.2 (12.1) | 42.6 (8.4) | 38.9 (16.5) | 42.9 (16.7) | 40.2 (16.4) | 39.5 (14.0) |
Age categories, n (%) | ||||||
Adolescents (aged ≥ 12 to < 18 years) | NA | NA | 2 (6.5) | 2 (6.5) | 2 (6.7) | 2 (6.5) |
Adults (≥ 18 years) | NA | NA | 29 (93.5) | 29 (93.5) | 28 (93.3) | 29 (93.5) |
BMI (kg/m2) | ||||||
Mean (SD) | 27.36 (7.64) | 26.29 (9.62) | 26.85 (7.22) | 27.38 (8.76) | 25.61 (7.25) | 26.85 (8.28) |
Smoking status, n (%) | ||||||
Never | ██ ██████ | ██ ███ | ██ | ██ | ██ | ██ |
Former | █████ | ██████ | ██ | ██ | ██ | ██ |
Current | ██████ | ██████ | ██ | ██ | ██ | ██ |
Time since first diagnosis, n (%) | ||||||
≤ 1 year | ██ ██████ | ██████ | 5 (16.1) | 4 (12.9) | 4 (13.3) | 3 (9.7) |
> 1 to ≤ 5 years | ██████ | ██████ | 6 (19.4) | 9 (29.0) | 9 (30.0) | 10 (32.3) |
> 5 to ≤ 10 years | ██████ | ██████ | 6 (19.4) | 8 (25.8) | 8 (26.7) | 7 (22.6) |
> 10 years | ██ ██████ | ██████ | 14 (45.2) | 10 (32.3) | 9 (30.0) | 11 (35.5) |
Average number of flares per year | ||||||
Mean (SD) | ███ █████ | ███ ██ | 2.7 (2.3) | 1.9 (0.9) | 2.4 (1.9) | 2.4 (1.2) |
Concomitant use of systemic GPP medication at randomization, n (%) | ||||||
Yes | NA | NA | 25 (80.6) | 23 (74.2) | 22 (73.3) | 22 (71.0) |
Concurrent plaque psoriasis,b n (%) | ||||||
Yes | NA | NA | 10 (32.3) | 7 (22.6) | 7 (23.3) | 10 (32.3) |
GPPGA total score, n (%)c | ||||||
0 | NA | NA | 2 (6.5) | 8 (25.8) | 3 (10.0) | 4 (12.9) |
1 | NA | NA | 29 (93.5) | 23 (74.2) | 27 (90.0) | 27 (87.1) |
3 | 28 (80.0) | 15 (83.3) | NA | NA | NA | NA |
4 | 7 (20.0) | 3 (16.7) | NA | NA | NA | NA |
GPPGA pustulation subscore, n (%) | ||||||
0 | NA | NA | 23 (74.2) | 24 (77.4) | 20 (66.7) | 21 (67.7) |
1 | NA | NA | 8 (25.8) | 7 (22.6) | 10 (33.3) | 10 (32.3) |
2 | 6 (17.1) | 5 (27.8) | NA | NA | NA | NA |
3 | 16 (45.7) | 7 (38.9) | NA | NA | NA | NA |
4 | 13 (37.1) | 6 (33.3) | NA | NA | NA | NA |
GPPASI total score | ||||||
Mean (SD) | 27.789 (13.436) | 24.056 (15.209) | 3.03 (3.48) | 3.12 (4.16) | 3.92 (4.42) | 3.11 (2.81) |
Pain VAS score | ||||||
Mean (SD) | 76.4 (16.8) | 64.6 (27.6) | NA | NA | NA | NA |
PSS total score | ||||||
Number of patients contributing to the analysis | 35 | 18 | 31 | 31 | 29 | 31 |
Mean (SD) | 10.4 (3.6) | 10.3 (3.1) | 4.1 (3.8) | 3.9 (2.9) | 5.3 (3.8) | 3.6 (2.9) |
DLQI score | ||||||
Number of patients contributing to the analysis | 35 | 18 | 30 | 31 | 29 | 31 |
Mean (SD) | 19.1 (7.1) | 19.6 (7.1) | 7.6 (6.7) | 6.6 (5.6) | 11.1 (6.9) | 7.2 (5.6) |
FACIT-Fatigue score | ||||||
Mean (SD) | 18.1 (14.2) | 19.0 (14.9) | NA | NA | NA | NA |
JDA GPP Severity Index, n (%) | ||||||
Mild | 9 (25.7) | 5 (27.8) | ██ ███████ | ██ ███████ | ██ ██████ | ██ ███ |
Moderate | 19 (54.3) | 8 (44.4) | █████ | █████ | █████ | █████ |
Severe | 4 (11.4) | 4 (22.2) | █████ | █████ | █████ | █████ |
Missing | 3 (8.6) | 1 (5.6) | █████ | █████ | █████ | █████ |
Presence of potentially pathogenic IL36RN variation | ||||||
No, n (%) | 24 (68.6) | 12 (66.7) | 17 (54.8) | 15 (48.4) | 19 (63.3) | 22 (71.0) |
Yes, n (%) | 5 (14.3) | 2 (11.1) | 7 (22.6) | 10 (32.3) | 7 (23.3) | 4 (12.9) |
DNA sequencing not done, n (%) | 6 (17.1) | 4 (22.2) | 7 (22.6) | 6 (19.4) | 4 (13.3) | 5 (16.1) |
Present or past occurrence of psoriasis | ||||||
Number of patients contributing to the analysis | ██ | ██ | NA | NA | NA | NA |
No, n (%) | █████ | █████ | 8 (25.8) | 12 (38.7) | 14 (46.7) | 8 (25.8) |
Yes, n (%) | █████ | █████ | 23 (74.2) | 19 (61.3) | 16 (53.3) | 22 (71.0) |
Unknown, n (%) | █████ | █████ | 0 | 0 | 0 | 1 (3.2) |
Prior use of at least 1 biologic therapy | ||||||
n (%) | NA | NA | 5 (16.1%) | 8 (25.8%) | 6 (20.0%) | 9 (29.0%) |
BMI = body mass index; DLQI = Dermatology Life Quality Index; FACIT-Fatigue = Functional Assessment of Chronic Illness Therapy–Fatigue; GPP = generalized pustular psoriasis; GPPASI = Generalized Pustular Psoriasis Area and Severity Index; GPPGA = Generalized Pustular Psoriasis Physician Global Assessment; HD = high dose; ITT = intention to treat; JDA = Japanese Dermatological Association; LD = low dose; MD = medium dose; NA = not applicable; PSS = Psoriasis Symptom Scale; q.4.w. = every 4 weeks; q.12.w. = every 12 weeks; SC = subcutaneous; SD = standard deviation; VAS = visual analogue scale.
aThe baseline characteristics for all 3 doses — low (spesolimab 300 mg loading dose followed by maintenance treatment of 150 mg q.12.w. as SC injections), medium (spesolimab 600 mg loading dose followed by maintenance treatment of 300 mg q.12.w. as SC injections), and high (spesolimab 600 mg loading dose followed by 300 mg subcutaneously, administered every 4 weeks) — are presented for the Effisayil 2 trial. Only the efficacy results for spesolimab HD have been reported for this review, as the other doses are not included under the recommended dosage approved by Health Canada.
bThe presence of concurrent plaque psoriasis was based on the investigator’s clinical investigation at enrolment.
cBaseline characteristics for the Effisayil 1 trial reflect the characteristics measured at randomization. In the Effisayil 1 trial, patients were required to have a GPPGA total score of at least 3 and, for the Effisayil 2 trial, patients were required to have a score of 0 or 1.
Sources: Effisayil 1 Clinical Study Report16 and Effisayil 2 Clinical Study Report.18
Table 13: Patient Exposure to Study Treatments in the Effisayil 1 Trial (SAS)
Patient exposure | Spesolimab (900 mg single-dose IV) (N = 35) | Placebo (N = 18) |
|---|---|---|
Total dose (mg) | ||
Mean (SD) | █████ ██████ | ██████ |
Median (range) | █████ ████ ██ ████ | ██████ |
Duration of infusion, minutes | ||
Mean (SD) | ████ ██████ | █████ ██████ |
Median (range) | ████ ███ ██ ████ | █████ ███ |
Adherence,a n (%) | ||
80% to 100% of planned volume infused | ██ █████ | ██ █████ |
SAS = safety analysis set; SD = standard deviation.
aTreatment adherence to the randomized study treatment on day 1 was characterized by dose intensity (%), which was defined as the total volume (in millimetres) of spesolimab or placebo that the patient received divided by the total volume of spesolimab or placebo the patient should have received (100 mL) multiplied by 100.
Source: Effisayil 1 Clinical Study Report.16
Table 14: Patient Exposure to Randomized Study Treatments — Effisayil 2 Trial (Treatment Phase, SAS)
Patient exposure | Spesolimab SC 300 mg q.4.w. (HD) (N = 30) | Placebo (N = 30) |
|---|---|---|
Total SC dose per patient (mg) | ||
Mean (SD) | ██████ ████████ | ██████ |
Median (range) | ██████ ████ ██ █████ | ██████ |
Duration of exposure (weeks) | ||
Mean (SD) | ████ ██████ | ████ ██████ |
Median (range) | ████ ████ ██ ███ | ████ ████ ██ |
Adherencea | ||
% of planned total dose, mean (SD) | ████ █████ | ██ |
HD = high dose; NA = not applicable; q.4.w. = every 4 weeks; SAS = safety analysis set; SC = subcutaneous; SD = standard deviation.
aTreatment compliance with the randomized study treatment was characterized by dose intensity (%), which was defined as the total dose of spesolimab (in milligrams) that the patient received divided by the total dose of spesolimab the patient should have received multiplied by 100.
Source: Effisayil 2 Clinical Study Report.18
Exposure to Study Treatments
Exposure to randomized study treatments in the Effisayil 1 and Effisayil 2 trials is summarized in Table 13 and Table 14.
██ ███ ███████████ ██████ ████████ ██████████ ██ ██████████ ████████ █ ████ ████ ████ ██ █████ ██████ ███ ███ ████ ████ ████████ ████████ ███ ████ ██████ ███████ ██ ███ ██████████ ██████ ████ █████ ██ ████████ █████████ ██ ████████ ████████ ██ ██ ███████ ██ ████ ████ ███ ████████ █████ ███ ██ ████ ██ ███ ███████ ████ ██████ ███ ███████ ███ ███ ███████ ███ ███ ███████████ ██████ ████████ ██████████ ██ ██████████ ██ ████████ █ ████ ████ ████ ██ ██████ ██ █████████ ██ █████████ ███ ████ ████ ████████ ██ ████████ ███ ████ ██████ █████ ███ ████████ ████████ ██ ██████████ ███ ████ ██████ █████ ███ ████████ ████████ ██ ██████████ ███ ████ ██████ █████ ███ ████████ ████████ ██ ██ ██████████ ███ ████ █████ ██████ ███ ████████ ████████ ██ ████████ ██████████ ███ ██████████ ███████████ █████████ ███████ ███ ████ ████ ██ ███ █████ ████████████ ██████████ ████ ███ █████ ██████ ██ ███ ████████ █████ ████ ██████ ███ █ ███████ ███ ████ ███████ ██ █████████ █████████ ███ ████████ ██████ ███ ███████ ██████████ █████████ ███████
Concomitant Medications and Co-Interventions
Effisayil 1 Trial
The most common types of concomitant medications and the number of patients who used escape medication and spesolimab rescue medication during the trial are summarized in Table 15. Escape medications were used by a total of 11 patients (17.1% of patients in the spesolimab group versus 27.8% in the placebo group), while OL spesolimab on day 8 was used by a total of 27 patients (34.3% in the spesolimab, group versus 83.3% in the placebo group), and rescue treatment with spesolimab was used by 6 patients (11.4% in the spesolimab group versus 11.1% in the placebo group).17
Effisayil 2 Trial
The most common types of concomitant medications and rescue treatment used during the treatment and maintenance phases are summarized in Table 16.
██████ ███ ██████████ ███████████ ███████ █████ ██ ████████ ████████ ██ █████ █ ███████████ ███████████ ███ ████ ██████████ ████ ███████████ ███████████ ████ ███████████ ██████ ██ ██████████ ██ █████ ██ ████████ ███ ██████████ ██████ ██ ██████████ ██ █ ██ █████████ ███████████████████████ ███ ███ ████████████ ██ █ ████████ ██ █████ ███ ██████████. In total, 32 patients received spesolimab IV rescue treatment after experiencing a first GPP flare during the randomized period, thus entering the flare treatment period. ██████ ████ ████ ██ ███████████ ██ ██████ ████ ███ ████████████ ██ ██ █████████ ███ █ ██████ ████ █████ ███ ██ ██████ ███ ████████████ ██ ███ ███ ████ ██████ ████ ██ ███████████ ██████████ ████████████ ██████ ███ █████ █████████ ██████ ███ ███████████████ ████████. A total of 30 patients completed the 12-week flare treatment period, and 20 patients received an OL maintenance dose (300 mg spesolimab SC injection every 12 weeks) lasting up to week 48.17
Table 15: Concomitant Medications — Effisayil 1 Trial (ITT)
Received concomitant medication,a n (%) | Spesolimab (900 mg single-dose IV) (N = 35) | Placebo (N = 18) |
|---|---|---|
Rescue use within the treatment phaseb | ||
Patients with ≥ 1 OL spesolimab on day 8 | 12 (34.3) | 15 (83.3) |
Patients with ≥ 1 escape medication (SOC) | 6 (17.1) | 5 (27.8) |
Patients with ≥ 1 spesolimab rescue medication | 4 (11.4) | 2 (11.1) |
Other medication starting in the treatment phase | ||
Patients with ≥ 1 concomitant medication, n (%) | ██ ██████ | ██ ██████ |
Most commonc concomitant medication, n (%) | ||
███████████ | ██ ██████ | ██ ██████ |
██████████ | █████ | █████ |
███████████ ████ █████████ ███████ █████ ████ ████████ | █████ | █████ |
█████ ████ | █████ | █████ |
████████████ ██████ | █████ | █████ |
█████████████ | █████ | █████ |
█████████████ | █████ | █████ |
████████████ | █████ | █████ |
GPP = generalized pustular psoriasis; ITT = intention to treat; OL = open label; SOC = standard of care.
aConcomitant medications include all medications ongoing at the start of randomized trial treatment and not only medications for GPP.
bThe treatment phase is defined as the period between the start of spesolimab or placebo administration and the end of the residual-effect period (16 weeks after the placebo dose or the last dose of spesolimab).
cFrequency ≥ 10% of all randomized patients in the study group.
Source: Effisayil 1 Clinical Study Report.16
Table 16: Concomitant Medications — Effisayil 2 Trial (ITT)
Received concomitant medication, n (%) | Spesolimab SC 300 mg q.4.w. (HD) | Placebo |
|---|---|---|
Rescue use | ||
Patients with ≥ 1 investigator-prescribed SOC (discontinued from the study) | 1 (3.3) | 1 (3.2) |
Patients with ≥ 1 spesolimab IV (entering OL flare treatment period) | 2 (6.7) | 15 (48.4) |
Patients with ≥ 1 spesolimab SC post flare (entering OL maintenance period) | 1 (3.3) | 12 (38.7) |
Other | ||
Patients with ≥ 1 concomitant medication, n (%) | ██ ██████ | ██ ██████ |
Most commona concomitant medication, n (%) | ||
████████████ | █████ | █████ |
████████ ███████ | █████ | █████ |
████████ ███████ ████ ██ ████████ ████████ | █████ | █████ |
███████████ | █████ | █████ |
███████████ | █████ | █████ |
GPP = generalized pustular psoriasis; HD = high dose; ITT = intention to treat; OL = open label; q.4.w. = every 4 weeks; SC = subcutaneous; SOC = standard of care.
aFrequency ≥ 10% of all randomized patients in the study group.
Source: Effisayil 2 Clinical Study Report.18
Efficacy
Effisayil 1 Trial
Proportion of Patients With a GPPGA Pustulation Subscore of 0 at Week 1 (Primary End Point)
The primary objective of the Effisayil 1 trial was met at the April 1, 2021, data cut-off date. In the spesolimab group, 54.3% of patients achieved a GPPGA pustulation subscore of 0 (i.e., no visible pustules) at week 1 compared with 5.6% in the placebo group. The risk difference between spesolimab and placebo was 48.7% (95% CI, 21.5% to 67.2%; superiority P value was 0.0004).17 Table 17 summarizes the proportion of patients with a GPPGA pustulation subscore of 0 at week 1.
Sensitivity Analyses: Three sensitivity analyses were carried out on the primary end point. Findings from all sensitivity analyses were consistent with the main analysis of the primary end point. The risk difference between spesolimab and placebo for the proportion of patients with a GPPGA pustulation subscore of 0 at week 1 was █████ ████ ███ █████ ██ ███████ █████ ████ ███ █████ ██ ███████ ███ █████ ████ ███ █████ ██ ██████ for sensitivity analysis 1 (EN-BRI), sensitivity analysis 2 (EN-PC-NRI), and sensitivity analysis 3 (EN-NRI), respectively.17
Additional Analyses: Additional analyses conducted for patients who received OL spesolimab on day 8 for their unresolved flares or who still had flare symptoms were as follows. Twelve of 35 patients randomized to spesolimab who experienced persistent flare symptoms on day 8 received a second dose of spesolimab on day 8. Of these 12 patients, 41.7% (5 patients) achieved pustular clearance, and 16.7% (2 patients) achieved a GPPGA total score of 0 or 1 at week 2 (i.e., 1 week after the second dose administration).
Proportion of Patients With a GPPGA Total Score of 0 or 1 at Week 1 (Key Secondary End Point)
The key secondary objective of the Effisayil 1 trial was met by the April 1, 2021, data cut-off date. In total, 42.9% of patients in the spesolimab group had achieved a GPPGA total score of 0 or 1 (i.e., clear or almost clear skin) at week 1 compared with 11.1% in the placebo group. The risk difference between the spesolimab and placebo groups was 31.7% (95% CI, 2.2% to 52.7%; superiority P value was 0.0118).17 Table 17 summarizes the proportion of patients with a GPPGA total score of 0 or 1 at week 1.
Sensitivity Analyses: In the Effisayil 1 trial, 3 sensitivity analyses were conducted for the key secondary end point, mirroring the primary analyses. Findings from all sensitivity analyses were consistent with the main analysis of the key secondary end point. The risk difference between spesolimab and placebo for the proportion of patients with a GPPGA pustulation subscore of 0 or 1 at week 1 ███ █████ ████ ███ ████ ██ ███████ █████ ████ ███ ████ ██ ███████ ███ █████ ████ ███ ████ ██ ██████ ███ ███████████ ████████ 1 (EN-BRI), sensitivity analysis 2 (EM-PC-NRI), and sensitivity analysis 3 (EN-NRI), respectively.17
Table 17: Key Efficacy Outcomes — Effisayil 1 Trial (ITT)
Outcome measure | Spesolimab (900 mg single-dose IV) (N = 35) | Placeboa (N = 18) |
|---|---|---|
Primary end point: GPPGA pustulation subscore of 0 at week 1 | ||
Number of responders, n (%) | 19 (54.3) | 1 (5.6) |
95% CIb (%) | 38.2 to 69.5 | 1.0 to 25.8 |
Risk difference, % (95% CI)c | 48.7 (21.5 to 67.2) | NA |
P valued | 0.0004 | NA |
Key secondary end point: GPPGA total score of 0 or 1 at week 1 | ||
Number of responders, n (%) | 15 (42.9) | 2 (11.1) |
95% CIb (%) | 28.0 to 59.1 | 3.1 to 32.8 |
Risk difference and 95% CIc (%) | 31.7 (2.2 to 52.7) | NA |
P valued | 0.0118 | NA |
Secondary end point: CFB in pain VAS at week 4 | ||
Number of failures, n (%) | ██ ██████ | ██ ██████ |
Median (IQR) | ██████ ████████ ███ | ██ ████ |
Estimate of difference, median (95% CI)e | ██ ████ | ██ ████ |
P valuef | ██████ | ██ ████ |
CFB = change from baseline; CI = confidence interval; GPPGA = Generalized Pustular Psoriasis Physician Global Assessment; IQR = interquartile range; ITT = intention to treat; NA = not applicable; OL = open label; VAS = visual analogue scale.
aFor the placebo group, more than 50% of the patients were classified as nonresponders, and a median absolute CFB could not be calculated at this time point. This classification was attributed to the large number of patients in both groups who used rescue medication, OL spesolimab on day 8, or spesolimab rescue medication. Additionally, nonresponses were ranked as the worst values in the analysis under the primary estimand for continuous end points.
bCalculated using the Wilson CI method.
cCalculated using the Chan and Zhang method.
dCalculated using the Suissa and Shuster z-pooled test (1-sided P value).
eEstimated using the modified Hodges-Lehmann method: The median difference using this method was not calculated once the overall nonresponse rate was > 30%.
fBased on Wilcoxon rank testing (1-sided P value).
Source: Effisayil 1 Clinical Study Report.17
CFB in Pain VAS Score at Week 4 (Secondary End Point)
By the April 1, 2021, data cut-off date, 88.9% of patients in the placebo group were nonresponders in the pain VAS score CFB assessment compared with 42.9% in the spesolimab group due to the use of escape medication, OL spesolimab at day 8, or rescue medication with spesolimab before week 4. There was a change in the median absolute CFB of −22.45 (IQR, −70.41 to nonresponse) in the spesolimab group, representing a decrease in pain, whereas, in the placebo group, the median was not calculable due to the use of escape medication, OL spesolimab at day 8, or rescue medication with spesolimab before week 4. A treatment difference compared with placebo was noncalculable for the absolute CFB in the pain VAS score at week 4 (P value = 0.0012).17 Table 17 summarizes the absolute CFB in the pain VAS score at week 4.
Effisayil 2 Trial
Only the results for the spesolimab 300 mg SC every 4 weeks dosage are presented in this section.
Time to First GPP Flare Up to Week 48 (Primary End Point)
The secondary hypothesis of the Effisayil 2 trial was also met by the January 13, 2023, data cut-off. The estimated HR was 0.157 (95% CI, 0.046 to 0.541; superiority P value = 0.0005). Table 18 summarizes the time to first GPP flare onset up to week 48.
Sensitivity Analyses: ████████ ████ ███ ███████████ ████████ █████ █████ ███ █████ ████████ ████ █████ ███████ █████ ██ ████████ ███ ██ ████████ ███ ████ █████ ████ ███ █████ ██ ████████ ███ ██████ ████████ ████ █████ ████ ███ █████ ██ ████████ ███ ███ █████ ████████ ████ █████ ████ ███ █████ ██ ███████ ███ ██████████ ██ ████████ ██ ███████ ████ ██████████ ████ ███ ███████ █████████
Occurrence of 1 or More GPP Flares Up to Week 48 (Key Secondary End Point)
The key secondary end point was met by the January 13, 2023, data cut-off date. The estimated adjusted risk difference by week 48 was −39.0% (95% CI, −62.1% to −15.9%; superiority P value = 0.0013) in favour of spesolimab HD over placebo. An allocated alpha of 0.0063 was applied in the analysis.17 Table 18 summarizes the occurrence of at least 1 GPP flare up to week 48.
Time to First Worsening of DLQI Up to Week 48 (Secondary End Point)
By the January 13, 2023, data cut-off, 23% of patients in the spesolimab HD group reported DLQI worsening at up to week 48 compared with the placebo group (65%). The estimated HR for risk of DLQI worsening over 48 weeks was 0.259 (95% CI, 0.109 to 0.620).17 The estimated risk difference for the probability of DLQI worsening in the spesolimab HD versus placebo was −42.4% (95% CI, −64.3% to −20.4%). Table 18 summarizes the time to first worsening of the DLQI score up to week 48.
Table 18: Key Efficacy Outcomes Up to Week 48 — Effisayil 2 Trial (ITT)
Outcome measure | Spesolimab SC 300 mg q.4.w. (HD) (N = 30) | Placebo (N = 31) |
|---|---|---|
Time to first GPP flare | ||
Patients with GPP flares, n (%) | 3 (10.0) | 16 (51.6) |
Meeting GPP flare criteria,a n (%) | 2 (6.7) | 15 (48.4) |
Probability of event,b Kaplan-Meier estimate, % (95% CI) | 10.0 (3.3 to 27.9) | 51.6 (35.6 to 69.8) |
Median time to event, weeks (95% CI) | NC (NC to NC) | 37.3 (4.0 to NC) |
HR (95% CI)b for time to the first flare vs. placebo | 0.157 (0.046 to 0.541) | NA |
P valuec | 0.0005 | NA |
Occurrence of ≥ 1 GPP flare | ||
Proportion of patients with at least 1 GPP flare,d % (95% CI) | 12.7 (5.0 to 28.9) | 51.6 (34.8 to 68.0) |
Risk difference for GPP flare occurrence in the spesolimab HD group vs. placebo (95% CI)e | −39.0 (−62.1 to −15.9) | NA |
P value (1-sided) | 0.0013 | NA |
Time to first worsening of DLQI | ||
Patients with DLQI worsening,f n (%) | 7 (23.3) | 20 (64.5) |
Probability of event,g % (95% CI) | 24.7 (12.6 to 45.1) | 64.5 (48.1 to 80.6) |
Median time to event, weeks (95% CI) | NC (NC to NC) | 16.0 (4.0 to NC) |
HR (95% CI)b | 0.259 (0.109 to 0.620) | NA |
P valuec | 0.0010h | NA |
Risk difference for DLQI worsening in the spesolimab HD group vs. placebo (95% CI)e | −42.4 (−64.3 to −20.4) | NA |
P value (1-sided) | < 0.0001 | NA |
CI = confidence interval; DLQI = Dermatology Life Quality Index; GPP = generalized pustular psoriasis; GPPGA = Generalized Pustular Psoriasis Physician Global Assessment; HD = high dose; HR = hazard ratio; ITT = intention to treat; NA = not applicable; NC = not calculable; q.4.w. = every 4 weeks; SC = subcutaneous; SOC = standard of care.
aGPP flare criterion: Increase in GPPGA score by ≥ 2 from baseline and a GPPGA pustulation subscore of ≥ 2 up to week 48.
bHR was compared with the placebo using the Cox regression model stratified by the use of systemic GPP medications at randomization.
cLog-rank test stratified by the use of systemic GPP medications at randomization. Proportion of patients before imputation.
dThe use of spesolimab IV treatment as a rescue medication or investigator-prescribed SOC to treat GPP worsening were considered as onset of GPP flare.
eCochran-Mantel-Haenszel test after multiple imputation, stratified by the use of systemic GPP medications at randomization.
fDLQI worsening criterion: 4-point increase in total score from baseline up to week 48. The use of spesolimab IV treatment as a rescue medication or investigator-prescribed SOC to treat GPP worsening were considered as onset of DLQI worsening.
gProbability of event was calculated using Kaplan-Meier estimates.
hNominal P value.
Source: Effisayil 2 Clinical Study Report.17
Harms
Effisayil 1 Trial
For the Effisayil 1 trial, AEs occurring between the start of treatment and the end of the residual-effect period (i.e., 16 weeks after the placebo or the last spesolimab dose) were reported. Key safety data for the Effisayil 1 trial were reported before the nonrandomized administration of spesolimab (week 1) and week 12 (i.e., after administration of any spesolimab, including only patients who received any spesolimab verum, double-blind or nonrandomized). AEs were reported until 16 weeks after the last spesolimab administration, end of study, or treatment in the extension trial, whichever was earlier, and are presented in Table 19.17
Adverse Events
Following the initial dose of spesolimab up to week 1 (day 8 or end of study), 77.1% of patients randomized to spesolimab and 66.7% of patients randomized to placebo reported at least 1 AE. The most frequently reported AEs during week 1 were pustular psoriasis (37.1% in the spesolimab group versus 38.9% in the placebo group) and pyrexia (5.7% in the spesolimab group versus 22.2% in the placebo group). The frequencies of all infections and infestations were numerically lower in the placebo group (5.6%) versus the spesolimab group (17.1%). All infections were categorized as mild or moderate intensity, except for 1 event (urinary tract infection) in the spesolimab group that was reported as serious. One patient in the spesolimab group was reported as having had a grade 4 life-threatening AE, which was DRESS syndrome.17
In total, 91.4% of patients initially randomized to spesolimab and 93.8% of patients initially randomized to placebo on day 1 experienced at least 1 AE. The most frequently reported AEs overall up to week 12 were pustular psoriasis (57.1% in the spesolimab group versus 43.8% in the placebo group), pyrexia (8.6% in the spesolimab group versus 12.5% in the placebo group), and vomiting (11.4% in the spesolimab group versus 6.3% in the placebo group). Overall, AE frequencies in the spesolimab group were comparable for patients after the first dose of spesolimab was administered and following up to 3 doses of spesolimab (including OL spesolimab on day 8 and/or rescue spesolimab after that).17
Serious Adverse Events
In total, 14.3% of patients treated with spesolimab and 16.7% of patients treated with placebo experienced at least 1 SAE up to day 7. The most frequently reported SAE was pustular psoriasis (11.4% in the spesolimab group versus 16.7% in the placebo group).17 After receiving any spesolimab, 25.7% of patients who were initially randomized to spesolimab on day 1 and 25.0% of patients who were initially randomized to placebo on day 1 experienced at least 1 SAE up to week 12. The most frequently reported SAEs were pustular psoriasis (14.3% in the spesolimab group versus 25% in the placebo group) and DRESS (2.9% in the spesolimab group versus 0 in the placebo group).17
Adverse Events of Special Interest
At week 1, 1 patient in the spesolimab group was reported as having experienced drug-induced liver injury and DRESS. At week 12, 1 patient who was initially randomized to spesolimab on day 1 experienced drug-induced liver injury and DRESS, and 1 patient who was initially randomized to placebo on day 1 experienced latent tuberculosis.17
Withdrawals Due to Adverse Events
There were no patient discontinuations due to AEs reported in the Effisayil 1 study.
Mortality
No deaths were reported in the Effisayil 1 study.
Table 19: Key Harms Data for the Effisayil 1 Trial (Safety Analysis Set)
AEs | Before nonrandomized spesolimab up to week 1a | After any spesolimab up to week 12b,c | ||
|---|---|---|---|---|
Spesolimab 900 mg single-dose IV (N = 35) | Placebo | Spesolimab 900 mg single-dose IV (N = 35) | Placebo (N = 16) | |
Most common AEs (> 10% of patients in either group), n (%)d | ||||
Patients with any AE | 27 (77.1) | 12 (66.7) | 32 (91.4) | 15 (93.8) |
Nervous system disorders | 5 (14.3) | 3 (16.7) | 6 (17.1) | 1 (6.3) |
Dizziness | 0 | 2 (11.1) | 0 | 0 |
████████ | █████ | █████ | █████ | █████ |
Skin and subcutaneous tissue disorders | 22 (62.9) | 9 (50.0) | 23 (65.7) | 10 (62.5) |
████████ █████████ | █████ | █████ | █████ | █████ |
████████ | █████ | █████ | █████ | █████ |
████████ | █████ | █████ | █████ | █████ |
General disorders and administration site conditions | 9 (25.7) | 5 (27.8) | 10 (28.6) | 3 (18.8) |
Pyrexia | 2 (5.7) | 4 (22.2) | 3 (8.6) | 2 (12.5) |
Gastrointestinal disorders | 6 (17.1) | 1 (5.6) | 9 (25.7) | 4 (25.0) |
████████ | █████ | █████ | █████ | █████ |
█████████ | █████ | █████ | █████ | █████ |
██████ | █████ | █████ | █████ | █████ |
Musculoskeletal and connective tissue disorders | 6 (17.1) | 3 (16.7) | 7 (20.0) | 4 (25.0) |
████ ██ █████████ | █████ | █████ | █████ | █████ |
Investigations | 7 (20.0) | 2 (11.1) | 7 (20.0) | 0 |
████ █████████ | █████ | █████ | █████ | █████ |
Infections and infestations | 12 (34.3) | 1 (5.6) | 20 (57.1) | 4 (25.0) |
Blood and lymphatic system disorders | 3 (8.6) | 2 (11.1) | 4 (11.4) | 0 |
Metabolism and nutrition disorders | 4 (11.4) | 2 (11.1) | 4 (11.4) | 0 |
SAEs, n (%)d | ||||
Patients with any SAE | 5 (14.3) | 3 (16.7) | 9 (25.7) | 4 (25.0) |
Resulted in death | 0 | 0 | 0 | 0 |
████████ ██ █████████ ███████████████ | █████ | █████ | █████ | █████ |
Arthritis | 1 (2.9) | 0 | 1 (2.9) | 0 |
Drug-induced liver injury | 1 (2.9) | 0 | 1 (2.9) | 0 |
Drug reaction with eosinophilia and systemic symptoms | 1 (2.9) | 0 | 2 (5.7) | 0 |
████████ █████████ | █████ | █████ | █████ | █████ |
Urinary tract infection | 1 (2.9) | 0 | 1 (2.9) | 0 |
AESIs,e n (%)d | ||||
Patients with any AESI | 1 (2.9) | 0 | 1 (2.9) | 1 (6.3) |
Drug-induced liver injuryf | 1 (2.9) | 0 | 1 (2.9) | 0 |
Drug reaction with eosinophilia and systemic symptoms | 1 (2.9) | 0 | 1 (2.9) | 0 |
Latent tuberculosis | 0 | 0 | 0 | 1 (6.3) |
Discontinuation due to AEs | ||||
Patients who discontinued | 0 | 0 | 0 | 0 |
Deaths | ||||
Patients who died | 0 | 0 | 0 | 0 |
AE = adverse event; AESI = adverse event of special interest; EoS = end of study; SAE = serious adverse event.
aIncluding AEs starting or worsening from the start of treatment to day 8 or EoS, whichever was earlier.
bPost any spesolimab: Including only patients who received any spesolimab verum (double-blind or nonrandomized): events are included until 16 weeks after last spesolimab administration, EoS, or treatment in the extension trial, whichever was earlier.
cEvents are included until 16 weeks after last spesolimab administration, EoS, or treatment in the extension trial.
dPercentages are calculated using total number of patients in the treatment class as the denominator.
eThe following were considered AESIs in the Effisayil 1 trial: hepatic injury, infusion reactions (including anaphylactic reactions), severe infections, and opportunistic and Mycobacterium tuberculosis infections.
fDrug-induced hepatic injury was reflected by an increase in aminotransferase levels and was considered to be a systemic symptom of drug reaction with eosinophilia and systemic symptoms.
Sources: Effisayil 1 Clinical Study Report.16 Details included in the table are from the sponsor’s summary of clinical evidence.17
Effisayil 2 Trial
Safety analyses in the Effisayil 2 study were conducted on the safety analysis set, which included all randomized patients who received at least 1 dose of the study drug. Key harms data for the spesolimab HD group (spesolimab 600 mg loading dose followed by 300 mg administered subcutaneously every 4 weeks) and for the Effisayil 2 trial are presented in Table 20.17
Adverse Events
The proportion of patients experiencing any AEs was comparable between the spesolimab and placebo groups. In total, 86.7% of patients in the spesolimab HD group and 86.7% in the placebo group reported 1 or more AEs. The most frequently reported AEs (≥ 10% in either group) were pustular psoriasis (10.0% of patients receiving spesolimab HD versus 53.3% receiving placebo), psoriasis (13.3% for spesolimab HD versus 10.0% for placebo), and injection site erythema (16.7% for spesolimab HD versus 3.3% for placebo).
Overall, most patients experienced AEs of mild (grade 1) or moderate (grade 2) intensity. A grade 3 AE was reported in 3 patients (10.0%) in the spesolimab HD group and 6 patients (20.0%) in the placebo group. Pustular psoriasis was the most frequently reported AE of grade 3, reported in 1 patient in the spesolimab HD group and 4 patients (13.3%) in the placebo group.17
Serious Adverse Events
In total, 10% of patients in the spesolimab HD and 3.3% in the placebo group reported 1 or more SAEs during the randomized treatment period. The most common SAE was pustular psoriasis (3.2%) in the spesolimab group (1 patient in each spesolimab dose group) compared with 0 in the placebo group. SAEs reported in the spesolimab HD group included pustular psoriasis, breast cancer, and cholelithiasis (1 patient each).17
Adverse Events of Special Interest
AESIs were not reported in the spesolimab HD group.17
Withdrawals Due to Adverse Events
AEs leading to study discontinuation were reported in 10% of patients treated with spesolimab HD. One patient withdrew due to pustular psoriasis. No patients in the placebo group discontinued treatment due to AEs.17
Mortality
No deaths were reported during the Effisayil 2 study.
Table 20: Key Harms for the Effisayil 2 Trial in the Randomized Maintenance Treatment Period (Safety Analysis Set)
AEs | Spesolimab SC 300 mg q.4.w. (HD) (N = 30) | Placebo (N = 30a) |
|---|---|---|
Most common AEs,b n (%) | ||
Patients with any AE | 26 (86.7) | 26 (86.7) |
Arthralgia | 3 (10.0) | 1 (3.3) |
COVID-19 | 3 (10.0) | 1 (3.3) |
Injection site erythema | 5 (16.7) | 1 (3.3) |
Pustular psoriasis | 3 (10.0) | 16 (53.3) |
Psoriasis | 4 (13.3) | 3 (10.0) |
Upper respiratory tract infection | 0 | 4 (13.3) |
Urinary tract infection | 4 (13.3) | 0 |
Blood creatine phosphokinase increased | 0 | 2 (6.7) |
SAEs, n (%) | ||
Patients with any SAE | 3 (10.0) | 1 (3.3) |
████████ ██ █████████ ██ | █████ | █████ |
Other medically important | 0 | 0 |
Breast cancer | 1 (3.3) | 0 |
Cholelithiasis | 1 (3.3) | 0 |
Multiple sclerosis | 0 | 1 (3.3) |
Pustular psoriasis | 1(3.3) | 0 |
AESIs,c n (%) | ||
Patients with any AESI | 0 | 0 |
Discontinuation due to AEs, n (%) | ||
Patients who discontinued | 3 (10.0) | 0 |
██████ ██████ | █████ | █████ |
█████████ | █████ | █████ |
█████████ ███████████ | █████ | █████ |
████████ █████████ | █████ | █████ |
Deaths, n (%) | ||
Patients who died | 0 | 0 |
AE = adverse event; AESI = adverse event of special interest; HD = high dose; q.4.w. = every 4 weeks; SAE = serious adverse event; SAS = safety analysis set; SC = subcutaneous.
aA patient randomly assigned to the placebo group who accidentally received a single dose of spesolimab 150 mg on day 1 was assigned to the spesolimab low-dose group for the analyses of exposure and safety (i.e., the SAS includes 30 patients in the placebo group and 32 patients in the spesolimab low-dose group for these analyses).
bFrequency ≥ 10% of all randomized patients in the study group.
cThe following were considered AESIs in the Effisayil 2 trial: hepatic injury, systemic hypersensitivity reactions (including infusion reactions and anaphylactic reactions), and peripheral neuropathy.
Sources: Effisayil 2 Clinical Study Report.18 Details included in the table are from the sponsor’s summary of clinical evidence.17
Critical Appraisal
Internal Validity
Effisayil 1 and Effisayil 2 were multicentre phase II and IIb RCTs (respectively) that employed similar study procedures and statistical methods. Patients were allocated to treatments using interactive response technology, and randomization was stratified by blocking and key factors. In the Effisayil 2 trial, randomization was stratified by region (Japan versus non-Japan), population (adults versus adolescents), and Asian versus non-Asian ethnicity. In the Effisayil 1 trial, randomization was stratified by concomitant use of systemic GPP medications at randomization (yes versus no). In the Effisayil 1 trial, numerical differences were observed in both arms of the trial in sex (males: 40.0% in the spesolimab group versus 16.7% in the placebo group; females: 60.0% in the spesolimab group versus 83.3% in the placebo group), race (Asian ethnicity: 45.7% in the spesolimab group versus 72.2% in the placebo group; white: 54.3% in the spesolimab group versus 27.8% in the placebo group), GPPGA pustulation subscore (score of 2: 17.1% in the spesolimab group versus 27.8% in the placebo group; score of 3: 45.7% in the spesolimab group versus 38.9% in the placebo group; score of 4: 37.1% in the spesolimab group versus 33.3% in the placebo group), and present or past occurrence of psoriasis (████ █████ ██████████ ██ █████ ███████). In the Effisayil 2 trial, numerical differences were observed in the spesolimab HD group compared with placebo for race, concurrent plaque psoriasis, IL36RN variation, and prior use of at least 1 biologic therapy. Due to the small sample size in both trials, it is more likely that prognostic balance was not achieved, as evidenced by the numerical imbalances in patient characteristics at baseline. Due to the rarity of GPP and the challenges associated with enrolling patients, a larger sample size was unlikely feasible.
Both trials were double-blind, where patients and study investigators involved in patient care or outcome assessment were blinded to treatments. Patients received a combination of matching active and placebo prefilled syringe solutions for injection to ensure that each treatment group received the same number and timing of injections to match the dosing regimens; as such, there is likely a low risk of bias in the measurement of the outcomes. However, there is the potential that patients could have inferred the group to which they were assigned due to differences observed in efficacy and harms in the spesolimab group relative to placebo, specifically in the Effisayil 2 trial. Knowledge of group assignment would increase the risk of bias, particularly for subjective outcomes (e.g., DLQI worsening [Effisayil 2 trial] and pain VAS [Effisayil 1 trial] and subjective harms); however, the presence and direction of any bias are uncertain.
In the Effisayil 1 trial, statistical analyses to test the superiority of spesolimab relative to placebo for the primary binary end point (GPPGA pustulation subscore of 0) and the key secondary end point (GPPGA total score of 0 or 1 at week 1) were conducted on the ITT population using an exact Suissa and Shuster z-pooled test. The between-group treatment effect was estimated at a 1-sided alpha level of 0.025 based on the stratification factors used at randomization. The CDA-AMC review team considered the statistical method and the method used to derive the 95% CIs around the risk difference (Chan and Zhang method) to be appropriate. The primary and key secondary outcomes were tested hierarchically, and familywise type I error was controlled at a 2-sided P value of less than 0.05 (1-sided alpha level of 0.025). Various approaches were used to assess the impact of missing data on the efficacy end points, depending on the type of end point. For binary end points, missing values were considered nonresponders, which was considered a conservative assumption of missing data. For continuous end points, missing data were primarily imputed using the LOCF method, which may not represent the true trajectory of the outcome (as it requires the value to be constant over time, from the time the data are missing to the time point analyzed). All randomized patients were treated and no patients prematurely discontinued study medication. Drug discontinuation rates during the planned observation period were low in the trial: 8.6% (n = 3) in the spesolimab group versus 5.6% (n = 1) in the placebo group. Statistical significance was achieved for the primary outcome, as the point estimate and corresponding 95% CIs did not include any values that were considered not clinically meaningful, according to the clinical experts’ opinion. The results of the sensitivity analyses to evaluate various methods of imputing missing data were consistent with the primary analysis, indicating that missing data were unlikely to bias the results for the primary outcome.
The statistical methods used to assess the hypothesis tests were considered appropriate. Spesolimab was favoured in the analysis, but there was uncertainty due to the small sample size, which increased the risk that prognostic balance was not achieved. A fixed sequence statistical hierarchy was implemented to control the familywise type I error rate (1-sided alpha level of 0.025) for the primary and key secondary outcomes in the Effisayil 2 study. Although more patients in the spesolimab HD group (13.3%; n = 4) discontinued treatment prematurely compared with the placebo group (3.2%; n = 1), no pattern was observed regarding the reason for discontinuations. Multiple imputation methods were applied to assess key secondary end points and the secondary binary end point. For binary end points, the primary imputation strategy for missing values was nonresponse imputation (which assumed that dropouts were nonresponders regardless of actual response status at the time of dropout), which is considered a conservative approach in evaluating the drug effect on the outcomes. For continuous end points, missing data were primarily imputed using the LOCF method (which is limited, as it may lead to the overestimation of treatment effects for participants for whom LOCF was applied). Sensitivity analyses were conducted to evaluate the robustness of the primary end point using different methods and analyses of population sets, and the findings were consistent with the primary analyses. The sensitivity analyses to evaluate missing data were also consistent with the primary analysis, indicating that missing data are unlikely to bias the results for the primary outcome.
Several patient-reported outcomes were assessed in both trials; however, only the pain VAS score and DLQI worsening were considered important for this review. The proportion of patients who experienced a decrease in the pain VAS from baseline to week 4 was not calculable in the placebo group due to the use of escape medication, OL spesolimab at day 8, or rescue medication with spesolimab before week 4. Therefore, these results were considered inconclusive. Additional analyses were conducted to assess the proportion of patients who did not experience a drop of 30 points or more in the pain VAS from baseline. However, the rationale for using a 30-point drop in the pain VAS scores from baseline was not provided and the review team was unable to draw definitive conclusions as to how spesolimab impacts the pain VAS in the absence of a valid MID of −30 points or more. The experts also considered the DLQI to be an important HRQoL assessment tool in flare prevention, given its non–disease-specific approach to assessing HRQoL in patients with dermatological conditions. The experts indicated they would consider using this questionnaire to assess changes in HRQoL in the presence of recurrent GPP flares.
The key outcomes evaluating spesolimab for the acute treatment of GPP flares in the Effisayil 1 trial were assessed at week 1, and the key outcomes for evaluating spesolimab in the prevention of GPP flares in the Effasayil 2 trial were assessed at 48 weeks. Both time frames were considered appropriate by the clinical experts consulted for this review.
External Validity
The inclusion criteria in the Effisayil trials differed due to the treatment setting. Both trials adequately captured patients with GPP, according to the experts; given the rarity of the disease, the potential for a small sample size of patients in both settings was expected. The majority of patients enrolled across the 2 trials were of Asian ethnicity or white and there were many unsuccessful screenings reported overall; most of the patients who were screened out did not meet the randomization criteria (32 patients in the Effisayil 1 trial and 34 patients in the Effisayil 2 trial). There were no trial sites in Canada. The clinical experts considered the inclusion and exclusion criteria in both trials to be generally acceptable and representative of patients in Canada. According to the experts, no key patient groups were excluded.
Several exclusion criteria were applied to both trials; notably, patients with SAPHO syndrome, primary erythrodermic psoriasis vulgaris, or drug-triggered AGEP were excluded from both. The clinical experts consulted did not anticipate that these exclusion criteria would impact the generalizability of the findings to patients in current practice. Acute flares are severe and associated with a risk of mortality. The experts indicated that a clinical decision on whether to administer spesolimab to a patient with an acute flare and presenting with any 1 of the outlined exclusion criteria would depend on the beneficial effects of spesolimab in resolving the flare and the potential risk of patient death from the acute flare.
Both trials were placebo-controlled, which was reasonable, given the absence of approved treatments specifically indicated for the treatment of GPP. Although the experts highlighted that the biologics indicated for plaque psoriasis are currently often used in both the acute and preventive settings to treat GPP, conclusions about the efficacy and harms of spesolimab compared with these off-label treatments could not be drawn in the absence of data from head-to-head trials in the setting of GPP flare treatment and prevention.
The concomitant medication use reported in both trials was aligned with clinical practice in Canada. Other treatment options (biologics and systemic modulating drugs for GPP and other conditions, such as plaque psoriasis) were restricted in the randomized phase of both trials. However, the use of methotrexate that was reported in the placebo group may contribute to the efficacy of placebo, thereby reducing the magnitude of the between-group effect (i.e., bias against spesolimab). The trial procedures were considered appropriate and reflective of clinical practice (i.e., the use of current treatment options as escape medication to treat flares and the additional use of spesolimab for patients who still had an unresolved flare (i.e., a GPPGA total score and GPPGA pustular component score ≥ 2 after receiving the initial dose of spesolimab IV). However, the experts noted that patients with comorbidities (plaque psoriasis) may require concomitant medications to treat both their symptoms and GPP flares. The use of spesolimab for the acute treatment of flares is aligned with the product monograph and with the current and future anticipated use in clinical practice. The duration of treatment assessment in both trials was considered appropriate and reflective of clinical practice.
The outcomes investigated in both trials were appropriate. The impact of spesolimab on CFB in the pain VAS was considered the most important patient-reported outcome measure that was investigated, as patients who respond to treatment and experience improvement in their flares (no pustules, i.e., a GPPGA pustulation subscore of 0) will experience less pain overall. The experts also considered the DLQI to be an important HRQoL assessment tool in flare prevention, given its non–disease-specific approach to assessing HRQoL in patients with dermatological conditions. The experts indicated they would consider using this questionnaire to assess changes in HRQoL in the presence of recurrent GPP flares. Mortality was also considered an important outcome for patients with GPP. Due to the severity of acute flares, patients may be at high risk of death from inflammation and potential organ failure. There were no deaths reported in any study group in either trial. As such, the review team was unable to draw conclusions on whether spesolimab reduces the risk of mortality in patients experiencing acute flares or at risk of flare recurrence.
Patients who completed treatment with spesolimab in the Effisayil 1 or Effisayil 2 trials were permitted to participate in the Effisayil ON long-term OLE trial, which is ongoing. The results from this study were not available at the time of this review. As such, limited information was available during this review to conclude on the long-term efficacy and safety of spesolimab for patients living with GPP.
GRADE Summary of Findings and Certainty of the Evidence
Methods for Assessing the Certainty of the Evidence
For the pivotal studies and RCTs identified in the sponsor’s systematic review, GRADE was used to assess the certainty of the evidence for those outcomes considered most relevant to inform the deliberations of the CDA-AMC expert committee, and a final certainty rating was determined as outlined by the GRADE Working Group.14,15
High certainty: We are very confident that the true effect lies close to that of the estimate of the effect.
Moderate certainty: We are moderately confident in the effect estimate. The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. We use the word “likely” for evidence of moderate certainty (e.g., “X intervention likely results in Y outcome”).
Low certainty: Our confidence in the effect estimate is limited. The true effect may be substantially different from the estimate of the effect. We use the word “may” for evidence of low certainty (e.g., “X intervention may result in Y outcome”).
Very low certainty: We have very little confidence in the effect estimate. The true effect is likely to be substantially different from the estimate of effect. We describe evidence of very low certainty as “very uncertain.”
Following the GRADE approach, the evidence from the RCTs started as high-certainty evidence and could be rated down for concerns related to study limitations (which refer to internal validity or risk of bias), indirectness, imprecision of effects, and publication bias.
When possible, certainty was rated in the context of an important (nontrivial) treatment effect; if this was not possible, certainty was rated in the context of the presence of any treatment effect (i.e., the clinical importance is unclear). In all cases, the target of the certainty-of-evidence assessment was based on the point estimate and where it was located relative to the threshold for a clinically important effect (when a threshold was available) or to the null. The target of the certainty-of-evidence assessment was the presence or absence of a clinically important effect based on the threshold informed by the clinical experts consulted by CDA-AMC for the following outcomes: the proportion of patients with a GPPGA pustulation subscore of 0, the proportion of patients with a GPPGA total score of 0 or 1, CFB in pain VAS scores, and time to worsening of the DLQI up to week 48. The clinical experts could not provide a clinically meaning threshold for time to first GPP flare, the proportion of patients with the occurrence of 1 or more GPP flares, and SAEs; thus, the null was used. The selection of outcomes for the GRADE assessment was based on the sponsor’s summary of clinical evidence, consultation with clinical experts, and input received from patient and clinician groups and public drug plans.
The following list of outcomes was finalized in consultation with expert committee members and was assessed using GRADE: the proportion of patients with a GPPGA pustulation subscore of 0, the proportion of patients with a GPPGA total score of 0 or 1, CFB in pain VAS scores, time to first GPP flare, the proportion of patients with the occurrence of 1 or more GPP flares, time to worsening of the DLQI up to week 48, and SAEs.
Results of GRADE Assessments
Table 2 and Table 3 present the GRADE findings for spesolimab versus placebo for the Effisayil 1 and Effisayil 2 trials, respectively.
Long-Term Extension Studies
Contents within this section have been informed by materials submitted by the sponsor. The following was summarized and validated by the CDA-AMC review team.
The long-term extension trial (Effisayil ON) to assess the efficacy and safety of long-term exposure to spesolimab IV and SC treatment (up to 252 weeks) for patients who were enrolled in the Effisayil 1 and Effisayil 2 trials is ongoing, with limited information available during this review.
Studies Addressing Gaps in the Systematic Review Evidence
No studies were submitted to address gaps in the systematic review.
Discussion
Summary of Available Evidence
Two pivotal studies (Effisayil 1 and Effisayil 2) were included in the systematic review. Effisayil 1 was a multicentre, randomized, placebo-controlled, double-blind, phase II trial evaluating the efficacy, safety, and tolerability of spesolimab single-dose IV compared with placebo in adults (ages 18 to 75 years) presenting with an acute flare of moderate to severe intensity who had received a diagnosis of GPP as per the ERASPEN criteria. The primary end point in the Effasayil 1 trial was the proportion of patients with a GPPGA pustulation subscore of 0 at the end of week 1. The key secondary end point assessed the proportion of patients with a GPPGA total score of 0 or 1 at the end of week 1. Other secondary end points included the CFB in the pain VAS score at week 4. The mean age in the spesolimab group was 43.2 years versus 42.6 years in the placebo group. In total, 60.0% and 83.3% of participants were female and 40% versus 16.7% were male in the spesolimab and placebo groups, respectively. Most patients were of Asian ethnicity (spesolimab: 45.7%; placebo: 72.2%) or white (spesolimab: 54.3%; placebo: 27.8%). Overall, most patients included in the trial had GPP that was moderately severe, i.e., had a GPPGA total score of 3 (spesolimab: 80.0%; placebo: 83.3%) and a GPPGA pustulation subscore of 3 (spesolimab: 45.7%; placebo: 38.9%) or 4 (spesolimab: 37.1%; placebo: 33.3%).
Effisayil 2 was a multicentre, randomized, placebo-controlled, double-blind, phase IIb dose-finding study evaluating the efficacy and safety of spesolimab SC for the prevention of GPP flares in patients with a history of GPP. The patients enrolled had experienced at least 2 GPP flares and had a GPPGA score of 0 or 1 (clear or almost clear) at randomization; they were aged between 12 and 75 years and had a known and documented history of GPP per the ERASPEN criteria. Only efficacy findings from the spesolimab HD group were reviewed for this reimbursement request. The primary end point tested after the dose-finding objective was time to first GPP flare up to week 48. The key secondary end point was the proportion of patients with at least 1 GPP flare up to week 48. At baseline, patients were aged between 14 and 75 years (8 patients were adolescents). The mean age at randomization in the spesolimab HD group was 40.2 years (SD = 16.4) versus 39.5 years (SD = 14.0) for placebo; 70.0% versus 54.8% were of Asian ethnicity, 30.0% versus 45.2% were white, 60.0% versus 58.1% were female, and 40.0% versus 41.9% were male in the spesolimab HD and placebo groups, respectively. The proportion of patients who had a GPPGA pustulation score of 0 (clear) was 67.7% in both groups, and the proportion of patients who had a GPPGA pustulation score of 1 (almost clear) was 33.3% in the spesolimab HD group versus 32.3% in the placebo group. Numerical differences were observed in the spesolimab HD group compared with placebo for race (the proportion of patients who were of Asian ethnicity was 70% in the spesolimab HD group versus 54.8% in the placebo group), concurrent plaque psoriasis (23.3% for spesolimab HD versus 32.3% for placebo), presence of potentially pathogenic IL36RN variation (23.3% for spesolimab HD versus 12.9% for placebo), and prior use of at least 1 biologic therapy (20% for spesolimab HD versus 29% for placebo). Patients who completed treatment with spesolimab in the Effisayil 1 or Effisayil 2 trial were permitted to participate in the Effisayil ON long-term OLE trial.
Interpretation of Results
Efficacy
GPP is a rare, chronic, severe, and potentially life-threatening disease. GPP is distinct from plaque psoriasis due to its unique genetic inflammatory pathway involving IL-36 and its histology and unique clinical presentation. GPP flares are associated with the risk of mortality in patients (up to 16% of patients die from flares). Thus, there is a need for treatments that effectively treat and prevent GPP flares. There is a lack of consensus guidelines for managing and treating GPP flares in Canada, and spesolimab is the only treatment specifically indicated for the treatment of GPP. As such, treatment choices may vary across jurisdictions, and current treatment options include treatments that are approved for plaque psoriasis but used off-label for GPP. The experts consulted indicated that current treatment options are often ineffective in managing GPP, given that their mechanisms of action do not target the IL-36 pathway, a key component in the pathophysiology of GPP. Current treatment options also have other limitations, for instance, some are contraindicated for certain patients (e.g., acitretin is contraindicated in women of childbearing potential), are not recommended for long-term use (corticosteroids), or may cause rebound (corticosteroids). The following treatment goals were important for acute flares: mortality prevention, rapid resolution of systemic symptoms, improved erythema and pustulation, and favourable safety profile. In consultation with the clinical experts on this review, long-term goals for patients with a history of flares who are also considered at risk of a GPP flare recurrence primarily include limiting or preventing flares and reducing pain, which may reduce disease burden and improve patient quality of life.
The Effisayil 1 trial provided evidence to support the use of spesolimab IV for the treatment of acute flares in adults with GPP, and Effisayil 2 provided evidence to support the use of subcutaneous spesolimab (loading dose of 600 mg followed by 300 mg of spesolimab every 4 weeks) for the prevention of GPP flares in adults and pediatric patients aged 12 years and older who had previously experienced at least 2 flares. Due to the rarity of GPP, a small sample size was expected in both trials. The certainty of evidence was assessed using GRADE for the following outcomes of interest for this review: the proportion of patients with a GPPGA pustulation subscore of 0, the proportion of patients with a GPPGA total score of 0 or 1, CFB in pain VAS scores, time to first GPP flare, the proportion of patients with the occurrence of 1 or more GPP flares, time to worsening of DLQI, and SAEs. The clinical experts consulted generally considered a GPPGA pustulation subscore of 0 (no visible pustules) and a GPPGA total score of 0 or 1 (clear or almost clear skin) as clinically important outcomes for patients presenting with acute GPP flares to assess treatment efficacy, and considered that the duration of the trial was appropriate to treat flares. Time to first GPP flare (defined as an increase in GPPGA score of 2 or more from baseline and a score of 2 or more in the pustular component of the GPPGA) and the proportion of patients with 1 or more GPP flares up to week 48 (defined as an increase in the GPPGA score of 2 or more points from baseline and a score of 2 or more in the pustular component of the GPPGA) were also considered critical for flare prevention, and 48 weeks for the duration of the Effisayil 33 trial was considered appropriate. The CFB in the pain VAS was considered an important patient-reported outcome for acute flare treatment because it aligns with changes in the GPPGA pustulation subscore during treatment.
The clinical experts could not provide a threshold for a clinically meaningful between-group difference in flare prevention. According to the experts, the most important outcomes to assess whether a treatment is impactful (treatment success or failure) in the prevention setting depends on key factors such as prior history of flares, triggers, the number of acute flares per year while on or not on treatment, flare severity (requiring hospitalization or admission to an intensive care unit), and time between consecutive flares (few months or years in between). Sources of uncertainties identified in both trials included the small sample size, raising concerns for potential prognostic imbalances (as evidenced by imbalances in patient disease and demographic characteristics across the groups in both trials) and the potential for overestimating the true treatment effect, and the potential for bias in the measurement of subjective patient-reported outcomes (pain VAS and DLQI). The patient populations enrolled in both trials were considered generalizable to the Canadian population, and key exclusion criteria will not impact the use of spesolimab in clinical practice. The Effisayil ON long-term trial is ongoing, with limited information available during this review. As such, the efficacy and harms of spesolimab relative to any comparator when used longer term is unknown.
The primary objective in the Effisayil 1 trial was met, supporting the superiority of single-dose spesolimab IV over placebo in treating adult patients with GPP flares. The proportion of patients presenting with no visible pustules (GPPGA pustulation subscore of 0) was higher in the spesolimab IV group (54.3%) versus the placebo group (5.6%). The experts considered a threshold of 15% to 20% as a clinically meaningful between-group difference for this outcome, including the proportion of patients reporting clear or almost clear skin and pain VAS scores. The point estimate for the difference between groups exceeded the threshold that the experts determined, and the 95% CI intervals did not include trivial effects, favouring spesolimab over placebo. The certainty of evidence was considered moderate due to imprecision (the wide CIs and potential for treatment overestimation due to the small sample size). Sensitivity analyses were consistent with the primary analyses, which is indicative of the robustness of the primary analysis using various estimands and methods of imputing missing data.
The key secondary outcome in the Effisayil 1 trial, the proportion of patients with a GPPGA total score of 0 or 1 at week 1 (i.e., clear or almost clear skin), also supported the clinical benefit of spesolimab IV over placebo for treating acute flares in adult patients with GPP. More patients (42.9%) who received spesolimab IV experienced clear or almost clear skin at week 1 compared with placebo (11.1%), which was considered clinically meaningful. The estimated between-group difference exceeded the experts’ suggested threshold for a clinically important effect, favouring spesolimab over placebo, and the CIs included estimates considered trivial by the experts. The certainty of the evidence was considered moderate, as the evidence was rated down due to imprecision.
The patient input received during the review highlighted that GPP substantially impacts patient quality of life, self-image, and school and work life, and places a financial strain on patients and caregivers. The CFB in the pain VAS, which was identified as an important patient-reported outcome in the Effisayil 1 trial, was tested hierarchically and controlled for type I error. The pain VAS is a unidimensional measure of pain intensity that is indicating using a horizontal or vertical line. A higher score indicates greater pain intensity, while a negative CFB signified an improvement from baseline. Statistical significance favoured spesolimab over placebo, as a change in the median absolute CFB of −22.45 was observed in the spesolimab group, suggesting a decrease in pain and an improvement from baseline. In the placebo group, the median was not calculable due to the use of escape medication, OL spesolimab at day 8, or rescue medication with spesolimab before week 4. The limitations identified include the potential for treatment effect overestimation due to the small sample size and the potential for reporting bias due to the subjective nature of this outcome, possibly impacting estimates in favour of spesolimab. In the absence of a calculable risk difference between the 2 groups in the trial population because the patients in the placebo group who were included in the analysis at week 4 received either rescue medication (SOC) or spesolimab at day 8, the review team was unable to make conclusive statements as to whether spesolimab impacted the pain VAS scores following treatment.
Subsequent analyses conducted on patients who received OL spesolimab on day 8 for unresolved flares or patients in the Effisayil 1 trial who still had flare symptoms showed that patients receiving spesolimab achieved pustular clearance. In total, 12 of the 35 patients randomized to spesolimab with persistent flare symptoms on day 8 received a second dose of spesolimab on day 8. Of these 12 patients, 41.7% (5 patients) achieved pustular clearance, and 16.7% (2 patients) achieved a GPPGA total score of 0 or 1 at week 2 (i.e., 1 week after the second dose administration). Of note, these subsequent analyses were not included in the hierarchical testing procedure and not conducted in the randomized set. No definitive conclusions could be drawn from the evidence; therefore, results should be considered supportive of the primary analyses.
As previously described, the Effisayil 2 trial evaluated the use of spesolimab for the prevention of GPP flares. The primary end point of the second objective of the trial was the time to first GPP flare, measured up to 48 weeks following treatment with spesolimab. Time to first GPP flare was defined as an increase in the GPPGA score of 2 or more points from baseline, with a pustular component score of 2 or greater. In the Effisayil 2 trial, GPP was evaluated according to the ERASPEN criteria, and a flare was defined according to the GPPGA scoring system. The experts highlighted that the ERASPEN criteria used in the trials was appropriate and consistent with clinical practice. However, the GPPGA scoring system is currently not used in clinical practice. The GPPGA relies on a physician’s clinical assessment of the skin presentation of the patient. It is scored using 3 components: erythema, pustules, and scaling of all GPP lesions. A score of 2 indicates mild disease, defined by bright red erythema and discrete, grouped, moderate-density pustules (noncoalescent) and predominantly fine scaling or crusting; a score of 3 indicates moderate disease (meaning bright red erythema with high-density pustules with some coalescence, and moderate scaling or crusting covering most or all lesions), and a score of 4 indicates severe disease (meaning deep fiery red erythema, very high density pustules with pustular lakes, and severe scaling or crusting covering most or all lesions). These definitions were consistent with the definition of a flare used in clinical practice. As per the analysis of time to first GPP flare following 48 weeks of treatment, the risk of GPP flare was lower among patients who received spesolimab SC relative to patients who received placebo, based on an HR of 0.157 (95% CI, 0.046 to 0.541; P = 0.0005). Fewer patients in the spesolimab group (10%) experienced a flare (defined as an increase in GPPGA score by ≥ 2 from baseline and a score of ≥ 2 in the pustular component of the GPPGA) compared with the placebo group (51.6%). Further, the CDA-AMC assessment using the GRADE framework suggested that spesolimab 300 mg SC every 4 weeks likely results in a clinically meaningful increase in the time to first GPP flare compared with placebo. Of note, the occurrence of GPP flares was consistently described as unpredictable during this review. The clinical experts consulted by CDA-AMC as well as the input from the sponsor’s submission highlighted that it is difficult to predict when a patient’s next GPP flare will happen, and the occurrence or severity of a flare does not necessarily correspond to the likelihood of the timing of the next flare. As such, although this outcome was the primary end point of the Effsayil 2 trial, the time to the next flare was not considered to be as important as the frequency or recurrence of GPP flares.
The occurrence of 1 or more GPP flares up to week 48 was a key secondary end point in the Effisayil 2 trial. The certainty-of-evidence assessment showed that spesolimab likely results in a clinically meaningful reduction in the proportion of patients having a flare event up to week 48 (risk difference = −39.0%; 95% CI, −62.1% to −15.9%). Overall, the clinical experts considered that any treatment that prevents recurring flares would benefit patients. The clinical experts elaborated that evaluating treatment success in the flare-prevention setting depends on an interplay of factors, including the patient’s baseline results and history in terms of the number of prior flares, severity of flares, triggers (if known), time between each flare, and number of flares per period. For instance, the experts noted that if a patient experiences a reduction in the number of flares compared with baseline following treatment with spesolimab, they would consider this a treatment success.
The patient group input also highlighted the need for new treatments that manage and reduce the frequency and severity of flares and minimize the symptoms and impact that patients experience between flares. Although several patient-reported outcomes were investigated in the Effisayil 2 trial, the DLQI questionnaire was considered very important due to the list of questions specific to dermatological conditions that measure other aspects of a patient’s quality of life. Time to first worsening of DLQI was a secondary end point in the Effisayil 2 trial, defined as a 4-point increase in total score from baseline. Intake of rescue medication or investigator-prescribed treatment for GPP was also considered an onset of a worsening. Findings from the trial suggest that patients who received spesolimab may have had a lower risk of experiencing a 4-point increase in the total DLQI score from baseline over week 48 compared with placebo (HR = 0.259; 95% CI, 0.109 to 0.620). The CDA-AMC assessment using the GRADE framework suggested that spesolimab SC 300 mg every 4 weeks may result in a clinically meaningful decrease in DLQI worsening by 4 points compared with placebo, indicating that patients who received spesolimab may not have experienced a negative impact in their quality of life in the trial when compared with placebo. However, given that statistical significance could not be established in the analysis, results should be interpreted as supportive of the overall effect of spesolimab on HRQoL in the flare-prevention setting.
As previously described, current treatment options for acute flare treatment and flare prevention before the availability of spesolimab include various treatments that are indicated for other diseases but are used off-label for patients with GPP. As such, they were not considered suitable comparators for assessing the efficacy and safety of spesolimab in the Effisayil 1 and Effisayil 2 trials, and the use of placebo-controlled trials was considered reasonable. However, it should be noted that common SOC therapies were not permitted as concomitant therapies. These included methotrexate, cyclosporine, retinoids, systemic corticosteroids, and treatments used for other types of psoriasis. As such, the use of placebo in the trial is not aligned with current SOC. The trial procedures implemented in the Effisayil 1 and Effisayil 2 trials were considered appropriate, and spesolimab was used in the trials as it would be in clinical practice. Although the Effisayil 1 and Effisayil 2 trials enrolled only patients with GPP, patients with GPP often present with other comorbidities, such as plaque psoriasis. The clinical experts noted that when faced with potentially life-threatening GPP flares in practice, clinicians would consider using other treatments, weighing the benefits and risks of the decision. Therefore, how these additional treatments may impact the efficacy of spesolimab or its safety profile in real-world practice is a source of uncertainty.
There was also limited information available to draw a conclusion on the long-term efficacy and safety of spesolimab for patients with GPP. This gap in the evidence is particularly relevant to the use of spesolimab for the prevention of flares. The long-term extension study (Effisayil ON), which is ongoing, was designed to assess the efficacy and safety of long-term exposure (up to 252 weeks) of spesolimab treatment (IV and SC) for patients who were enrolled in the Effisayil 1 and Effisayil 2 trials, with limited information available during this review.
Harms
Overall, the safety profile of spesolimab was considered manageable, and no new safety concerns were identified.
In the Effisayil 1 trial, AEs were reported at week 1 (i.e., before the nonrandomized administration of spesolimab) and week 12 (i.e., after any spesolimab administration). The week 12 analysis of AEs included only patients who received any spesolimab verum (double-blind or nonrandomized); AEs were included until 16 weeks after the last spesolimab administration, end of study, or treatment in the extension trial, whichever was earlier. The AEs reported were numerically higher in the spesolimab group versus placebo at week 1 and comparable in both groups at week 12 (week 1: 77.1% in the spesolimab group versus 66.7% for placebo; week 12: 91.4% in the spesolimab group versus 93.8% for placebo), which could be attributable to the severity of acute GPP flares. However, no new signals were identified. The AEs most commonly reported at week 1 were pustular psoriasis (37.1% in the spesolimab group versus 38.9% in the placebo group) and pyrexia (5.7% in the spesolimab group versus 22.2% in the placebo group). In the Effisayil 2 trial, the proportion and incidence rates of patients experiencing any AEs were comparable between the spesolimab HD group and placebo group (86.7% in the spesolimab HD group versus 86.7% in the placebo group). The most frequently reported AEs were pustular psoriasis (10% in the spesolimab HD group versus 53.3% in the placebo group), psoriasis (13.3% in the spesolimab HD group versus 10.0% in the placebo group), and injection site erythema (16.7% in the spesolimab HD group versus 3.3% in the placebo group). Although pustular psoriasis was frequently reported in both groups, which could be indicative of disease worsening, the experts consulted during the review did not consider this event to be a new signal. Upon further evaluation of individual patient reports of pustular psoriasis, the sponsor noted that some occurrences commenced before patients began receiving spesolimab while others were reported during the first week of treatment, suggesting they were related to the current GPP flare.
In the Effisayil 1 trial, the most frequently reported SAE overall was pustular psoriasis (11.4% in the spesolimab group versus 16.7% in the placebo group). After receiving any spesolimab, the most frequently reported SAEs were pustular psoriasis (14.3% in the spesolimab group versus 25% in the placebo group), and DRESS was reported in 2.9% of patients in the spesolimab group versus in 0 patients in the placebo group. In the Effisayil 2 trial, 10% of patients in the spesolimab HD group and 3.3% in the placebo group reported 1 or more SAEs during the randomized treatment period. The SAEs reported in the spesolimab HD group included pustular psoriasis, breast cancer, and cholelithiasis (1 patient each). There is uncertainty as to whether spesolimab increases the risk of SAEs due to the duration of the trials and the small number of events reported in both groups across trials. The AESIs reported were generally similar across trials. In the Effisayil 1 trial, 1 patient in the spesolimab group was reported as having experienced AESIs (drug-induced liver injury and DRESS) before the administration of nonrandomized spesolimab. One patient initially randomized to placebo on day 1 experienced latent tuberculosis after any spesolimab was administered. The clinical experts consulted considered this AESI to be manageable, with no new concern identified. In the Effisayil 2 trial, no AESIs occurred in the spesolimab HD group.
There were no reports of treatment discontinuations due to AEs in the Effisayil 1 trial. In the Effisayil 2 trial, AEs leading to study discontinuation occurred in 10% of patients receiving spesolimab HD and included pustular psoriasis (3.3%), psoriasis, and breast cancer (1 patient each; rate of 3.3% for each AE).
Mortality was also considered an important outcome for patients with GPP. Due to the severity of acute flares, patients may be at a high risk of death from inflammation and potential organ failure. In terms of safety, no deaths were reported. As such, no conclusions could be drawn about whether spesolimab has any effect on mortality.
Conclusion
GPP is a rare, chronic, severe, and potentially life-threatening disease. At the time of this review, spesolimab is the only treatment approved in Canada with an indication for the treatment of GPP. As such, the input from patients and clinicians on this review emphasized a need for new treatments that effectively treat and prevent flares. Two pivotal RCTs provided evidence for the efficacy and safety of spesolimab compared with placebo for the treatment and prevention of GPP flares (Effisayil 1 and Effisayil 2, respectively).
In the Effisayil 1 trial, patients receiving spesolimab IV infusions for acute GPP flares likely experienced better resolution of their flares with no visible pustules after 1 week of treatment compared with placebo. Similarly, patients receiving spesolimab IV infusions may have experienced clear or almost clear skin following treatment after 1 week compared with placebo. A reduction in pain was considered an important patient-reported outcome for acute flare treatment. Compared with placebo, patients with GPP with acute flares may have experienced an improvement in pain after receiving spesolimab based on the CFB in the pain VAS.
Regarding the use of spesolimab for the prevention of flares that was assessed in the Effisayil 2 trial, treatment with spesolimab SC (loading dose of 600 mg followed by 300 mg spesolimab every 4 weeks) up to 48 weeks likely results in a clinically meaningful increase in the time to first GPP flare compared with placebo for patients with GPP aged 12 years and older. Similarly, compared with placebo, patients treated with spesolimab were less likely to experience a GPP flare. HRQoL was measured using a 4-point increase from baseline in DLQI scores, indicative of a worsening in HRQoL. Findings showed that patients receiving spesolimab may have had a lower risk of experiencing a worsening of their symptoms compared with placebo. However, given the hierarchical testing in the statistical analysis plan, DLQI scores were not obtained, and only a nominal P value was provided; thus, the findings should be interpreted as supportive. Potential sources of uncertainty for the key outcomes assessed in both trials include the small sample sizes, which raise concerns for potential prognostic imbalances between groups and the potential overestimation of the treatment effects; potential risk of bias, particularly for subjective outcomes such as patient-reported outcomes and AEs; and the risk of bias for CFB in pain VAS, given that most patients in the placebo group received rescue medications or OL spesolimab. However, the direction of bias is uncertain.
The frequency of the AEs reported was generally high due to the severity of GPP, but comparable across groups in both trials. Additionally, there were no deaths reported in either of the pivotal trials. Safety data reviewed in this report were available up to week 1 and up to week 12 for spesolimab IV in the Effisayil 1 trial, and up to 48 weeks for spesolimab SC in the Effisayil 2 trial. Overall, there were no new concerns regarding the safety profile of spesolimab in the acute setting. While the safety data available for spesolimab in the preventive setting did not raise any concerns, the evidence informing the long-term safety of spesolimab SC for patients living with GPP is ongoing, with limited information available at the time of this review.
References
1.Navarini AA, Burden AD, Capon F, et al. European consensus statement on phenotypes of pustular psoriasis. J Eur Acad Dermatol Venereol. 2017;31(11):1792-1799. PubMed
2.Takeichi T, Akiyama M. Generalized Pustular Psoriasis: Clinical Management and Update on Autoinflammatory Aspects. Am J Clin Dermatol. 2020;21(2):227-236. PubMed
3.Fujita H, Terui T, Hayama K, et al. Japanese guidelines for the management and treatment of generalized pustular psoriasis: The new pathogenesis and treatment of GPP. J Dermatol. 2018;45(11):1235-1270. PubMed
4.Gooderham MJ, Van Voorhees AS, Lebwohl mg. An update on generalized pustular psoriasis. Expert Rev Clin Immunol. 2019;15(9):907-919. PubMed
5.Bachelez H. Pustular Psoriasis: The Dawn of a New Era. Acta Derm Venereol. 2020;100(3):adv00034. PubMed
6.Choon SE, Navarini AA, Pinter A. Clinical Course and Characteristics of Generalized Pustular Psoriasis. Am J Clin Dermatol. 2022;23(Suppl 1):21-29. PubMed
7.Rivera-Díaz R, Daudén E, Carrascosa JM, Cueva P, Puig L. Generalized Pustular Psoriasis: A Review on Clinical Characteristics, Diagnosis, and Treatment. Dermatol Ther (Heidelb). 2023;13(3):673-688. PubMed
8.Mastacouris N, Feda A, Strunk A, Garg A. Risk factors for generalized pustular psoriasis: A case-control study. J Am Acad Dermatol. 2023;89(4):846-848. PubMed
9.Tarride JE, Prajapati VH, Lynde C, Blackhouse G. The burden associated with generalized pustular psoriasis: A Canadian population-based study of inpatient care, emergency departments, and hospital- or community-based outpatient clinics. JAAD Int. 2023;12:90-98. PubMed
10.Kharawala S, Golembesky AK, Bohn RL, Esser D. The clinical, humanistic, and economic burden of generalized pustular psoriasis: a structured review. Expert Rev Clin Immunol. 2020;16(3):239-252. PubMed
11.Strober B, Leman J, Mockenhaupt M, et al. Unmet Educational Needs and Clinical Practice Gaps in the Management of Generalized Pustular Psoriasis: Global Perspectives from the Front Line. Dermatol Ther (Heidelb). 2022;12(2):381-393. PubMed
12.Spevigo (spesolimab): Spesolimab injection solution for subcutaneous injection 150 mg/1 mL (150 mg/mL); Spesolimab for injection concentrate for solution for intravenous infusion 450 mg/7.5 mL (60 mg/mL) [product monograph]. Burlington (ON): Boehringer Ingelheim (Canada) Ltd.; 2024.
13.Strober B, Kotowsky N, Medeiros R, et al. Unmet Medical Needs in the Treatment and Management of Generalized Pustular Psoriasis Flares: Evidence from a Survey of Corrona Registry Dermatologists. Dermatol Ther (Heidelb). 2021;11(2):529-541. PubMed
14.Balshem H, Helfand M, Schünemann HJ, et al. GRADE guidelines: 3. Rating the quality of evidence. J Clin Epidemiol. 2011;64(4):401-406. PubMed
15.Santesso N, Glenton C, Dahm P, et al. GRADE guidelines 26: informative statements to communicate the findings of systematic reviews of interventions. J Clin Epidemiol. 2020;119:126-135. PubMed
16.Clinical Study Report: c31523813-01. Effisayil™ 1: Multi-center, double-blind, randomized, placebo-controlled, Phase II study to evaluate efficacy, safety and tolerability of a single intravenous dose of BI 655130 in patients with Generalized Pustular Psoriasis (GPP) presenting with an acute flare of moderate to severe intensity [sponsor-supplied reference]. Ingelheim (DE): Boehringer Ingelheim, Ltd; 2021.
17.Sponsor’s clinical summary report: Spevigo (spesolimab) for the treatment and prevention of flares in adult and pediatric patients (≥12 years) with generealized pustular psoriasis in Canada [internal sponsor’s report]. In: Drug Reimbursement Review sponsor submission: SPEVIGO® (spesolimab) Solution for subcutaneous injection 150 mg/1 mL (150 mg/mL) and Concentrate for solution for intravenous infusion 450 mg/7.5 mL (60 mg/mL). Burlington (ON): Boehringer Ingelheim (Canada) Ltd.; 2024.
18.Clinical Study Report: c39219513-01. Effisayil™ 2: Multi-center, randomized, parallel group, double blind, placebo controlled, Phase IIb dose-finding study to evaluate efficacy and safety of BI 655130 (Spesolimab) compared with placebo in preventing generalized pustular psoriasis (GPP) flares in patients with history of GPP [sponsor-supplied reference]. Ingelheim (DE): Boehringer Ingelheim, Ltd; 2023.
19.Twelves S, Mostafa A, Dand N, et al. Clinical and genetic differences between pustular psoriasis subtypes. J Allergy Clin Immunol. 2019;143(3):1021-1026. PubMed
20.Menter A, Strober BE, Kaplan DH, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with biologics. J Am Acad Dermatol. 2019;80(4):1029-1072. PubMed
21.Menter A, Strober BE, Kaplan DH, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with systemic non-biologics therapies. J Am Acad Dermatol. 2020;82(6):1445-1486. PubMed
22.Robinson A, Van Voorhees AS, Hsu S, et al. Treatment of pustular psoriasis: from the Medical Board of the National Psoriasis Foundation. J Am Acad Dermatol. 2012;67(2):279-288. PubMed
23.Bachelez H, Choon SE, Marrakchi S, et al. Trial of Spesolimab for Generalized Pustular Psoriasis. N Engl J Med. 2021;385(26):2431-2440. PubMed
24.Boehringer Ingelheim. 2017-004231-37: Effisayil™ 1: Multi-center, double-blind, randomized, placebo-controlled, Phase II study to evaluate efficacy, safety and tolerability of a single intravenous dose of spesolimab (BI 655130) in patients with Generalized Pustular Psoriasis (GPP) presenting with an acute flare of moderate to severe intensity [sponsor-supplied reference]. Amsterdam (NL): EU Clinical Trials Register; 2022: https://www.clinicaltrialsregister.eu/ctr-search/trial/2017-004231-37/results.
25.Boehringer Ingelheim. NCT03782792: Effisayil™ 1: A Study to Test Spesolimab (BI 655130) in Patients With a Flare-up of a Skin Disease Called Generalized Pustular Psoriasis [sponsor-supplied reference]. ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2018: https://www.clinicaltrials.gov/study/NCT03782792.
26.Morita A, Strober B, Burden AD, et al. Efficacy and safety of subcutaneous spesolimab for the prevention of generalised pustular psoriasis flares (Effisayil 2): an international, multicentre, randomised, placebo-controlled trial. Lancet. 2023;402(10412):1541-1551. PubMed
27.Boehringer Ingelheim. 2018-003081-14: Effisayil™ 2: Multi-center, randomized, parallel group, double blind, placebo controlled, Phase IIb dose-finding study to evaluate efficacy and safety of BI 655130 (Spesolimab) compared with placebo in preventing generalized pustular psoriasis (GPP) flares in patients with history of GPP [sponsor-supplied reference]. Amsterdam (NL): EU Clinical Trials Register; 2023: https://www.clinicaltrialsregister.eu/ctr-search/trial/2018-003081-14/results.
28.Boehringer Ingelheim. NCT04399837: A Study to Test Whether BI 655130 (Spesolimab) Prevents Flare-ups in Patients With Generalized Pustular Psoriasis [sponsor-supplied reference]. ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2020: https://clinicaltrials.gov/study/NCT04399837.
29.Boehringer Ingelheim. NCT03886246: Effisayil™ ON: A Study to Test Long-term Treatment With Spesolimab in People With Generalized Pustular Psoriasis Who Took Part in a Previous Study [sponsor-supplied reference]. ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2019: https://clinicaltrials.gov/study/NCT03886246.
30.Burden AD, Bissonnette R, Lebwohl mg, et al. Psychometric validation of the generalized pustular psoriasis physician global assessment (GPPGA) and generalized pustular psoriasis area and severity index (GPPASI). J Eur Acad Dermatol Venereol. 2023;37(7):1327-1335. PubMed
31.Burden AD, Bachelez H, Choon SE, et al. Validation of the Generalized Pustular Psoriasis Physician Global Assessment (GPPGA) scale for assessing severity of GPP [sponsor-supplied reference]. American Academy of Dermatology. 2022.
32.Burden AD, Bissonnette R, Anatchkova M, et al. Psychometric validation of the Psoriasis Symptom Scale, Functional Assessment of Chronic Illness Therapy-Fatigue and pain-Visual Analogue Scale in patients with generalized pustular psoriasis. J Eur Acad Dermatol Venereol. 2024;38(7):1383-1390. PubMed
33.Flytstrom I, Stenberg B, Svensson A, Bergbrant IM. Patients’ visual analogue scale: a useful method for assessing psoriasis severity. Acta Derm Venereol. 2012;92(4):347-348. PubMed
34.DLQI instructions for use and scoring: information & conditions concerning use: clinical use of the DLQI [sponsor-supplied reference]. 1993. Accessed 2018 Jun 11.
35.Boehringer Ingelheim. NCT02978690: Spesolimab (BI 655130) Single Dose in Generalized Pustular Psoriasis [sponsor-supplied reference]. ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2016: https://classic.clinicaltrials.gov/ct2/show/NCT02978690.
36.Greenland S, Robins JM. Estimation of a common effect parameter from sparse follow-up data. Biometrics. 1985;41(1):55-68. PubMed
Pharmacoeconomic Review
Abbreviations
AE
adverse event
BAC
best available care
BIA
budget impact analysis
CDA-AMC
Canada’s Drug Agency
GPP
generalized pustular psoriasis
GPPGA
Generalized Pustular Psoriasis Physician Global Assessment
ICER
incremental cost-effectiveness ratio
QALY
quality-adjusted life-year
SC
subcutaneous
Executive Summary
The executive summary comprises 2 tables (Table 1 and Table 2) and a conclusion.
Item | Description |
|---|---|
Drug product |
|
Indication | For the treatment of GPP in adults and pediatric patients 12 years of age and older and weighing at least 40 kg. |
Health Canada approval status | NOC |
Health Canada review pathway | Standard review |
NOC date | July 31, 2024 |
Reimbursement request |
|
Sponsor | Boehringer Ingelheim (Canada) Ltd. |
Submission history | Previously reviewed: No |
GPP = generalized pustular psoriasis; GPPGA = Generalized Pustular Psoriasis Physician Global Assessment; NOC = Notice of Compliance; SC = subcutaneous.
Table 2: Summary of Economic Evaluation
Component | Description |
|---|---|
Type of economic evaluation |
|
Target populations | The treatment of GPP, including treatment of flares with a GPPGA total score of ≥ 2, and prevention of flares in adults and pediatric patients aged 12 years and older. |
Treatment | Spesolimab |
Dose regimen |
|
Submitted price | Spesolimab:
|
Submitted treatment cost |
|
Comparator | No treatment |
Perspective | Canadian publicly funded health care payer |
Outcomes | QALYs, LYs |
Time horizon | Lifetime (69 years) |
Key data sources |
|
Submitted results | ICER = $280,172 per QALY gained (incremental costs: $1,962,159; incremental QALYs: 7.31). |
Key limitations |
|
CDA-AMC reanalysis results |
|
BAC = best available care; CDA-AMC = Canada’s Drug Agency; GPP = generalized pustular psoriasis; GPPGA = Generalized Pustular Psoriasis Global Assessment; ICER = incremental cost-effectiveness ratio; LY = life-year; QALY = quality-adjusted life-year; SC = subcutaneous.
aThe sponsor assumed the same cost for preventive spesolimab treatment in the first and subsequent years.
Conclusions
Based on the Canada’s Drug Agency (CDA-AMC) Clinical Review, and based on the results of the Effisayil 1 trial, spesolimab results in a meaningful difference for the treatment of acute flares in achieving a Generalized Pustular Psoriasis Physician Global Assessment (GPPGA) pustulation subscore of 0 after 1 week of treatment compared with placebo (54.3% of patients on spesolimab versus 5.6% of patients on placebo), with moderate certainty. In the flare-prevention setting, CDA-AMC’s Clinical Review reported that based on the results of the Effisayil 2 trial, spesolimab results in a meaningful reduction in the proportion of patients experiencing a generalized pustular psoriasis (GPP) flare in 48 weeks compared with placebo (12.7% of patients on spesolimab versus 51.6% of patients on placebo), with moderate certainty. The CDA-AMC Clinical Review noted that the use of placebo as defined in the trials is not aligned with the current treatment options used in clinical practice. The Clinical Review additionally reported there is limited information available to draw a conclusion on the long-term efficacy and safety of spesolimab for GPP. There is no direct or indirect evidence comparing spesolimab with the treatments used in clinical practice in Canada.
CDA-AMC was able to address some limitations associated with the sponsor’s analysis, including applying the flare-related mortality estimate to the appropriate group of patients and including the administration costs associated with spesolimab IV. The results of the CDA-AMC base case are similar to the sponsor’s analysis. In the CDA-AMC base case, spesolimab is associated with an incremental cost-effectiveness ratio (ICER) of $431,569 per quality-adjusted life-year (QALY) gained compared with no treatment. The incremental cost is driven by treatment acquisition of spesolimab for preventive therapy, which makes up approximately 90% of the total cost for spesolimab. Higher life expectancy associated with spesolimab (5.69 incremental life-years) as a result of experiencing fewer flares drove the increase in QALYs. The majority of the benefit (99%) was incurred within 48 weeks (beyond which no clinical information is available for spesolimab). Cost-effectiveness is therefore heavily influenced by the long-term survival benefit for which the evidence is uncertain due to a lack of long-term information. For spesolimab to be considered cost-effective compared with no treatment at a threshold of $50,000 per QALY gained, the cost per dose of spesolimab for the treatment of acute flares would need to be $4,500, and the annual per-patient cost of spesolimab for preventive therapy would be $21,067 in the first year and $19,567 in subsequent years, reflecting a price reduction of approximately 79%.
CDA-AMC was unable to resolve several key limitations in the sponsor’s analysis. First, the review team was unable to resolve the lack of a comparison between spesolimab and the best care available in clinical practice in Canada, given the lack of comparative efficacy data. As such, the cost-effectiveness of spesolimab compared with current clinical practice remains unknown. Additionally, CDA-AMC was unable to resolve the limitations associated with the model structure, specifically, the use of adjuvant therapies and acute treatment changes within 24 to 48 hours. Finally, CDA-AMC was unable to resolve uncertainty in the duration of the treatment effect for spesolimab for the prevention of GPP flares; therefore, the reported ICER is dependent on the treatment benefit observed in the 48-week trial persisting over the 69-year time horizon. As a result of these limitations remaining unresolved, the reanalysis performed by CDA-AMC is highly uncertain.
Input Relevant to the Economic Review
This section is a summary of the feedback received from the patient groups, clinician groups, and drug plans that participated in the CDA-AMC review process.
Patient input was received from the Canadian Psoriasis Network and the Canadian Association of Psoriasis Patients, recently consolidated as Psoriasis Canada. The input was gathered from 3 sources of information: a GPP-focused virtual summit where 2 participants had a formal GPP diagnosis and 1 participant had symptoms aligned with GPP but no formal diagnosis, survey responses from 10 patients who could not attend the summit, and 4 interviews (3 patients with experience with the drug under review and 1 caregiver). The summit participants all resided in Canada. Three of the 4 patients interviewed were from Canada. Patient input for this submission was derived from people who live with GPP. Patients with GPP reported unpredictable flares involving painful pustules. The patient input indicated that GPP negatively impacted patients’ quality of life, including impacts on personal relationships, challenges at work requiring hands-on contact, and mental health. Patients said they experienced challenges getting referrals to dermatologists with experience in GPP and accessing treatments that treat GPP flares effectively and minimize symptoms between flares. Three patients indicated experience with treatment with spesolimab, and all reported improvements in pustules.
Clinician input was received from the Origins Dermatology Centre. Clinicians indicated challenges with the current pathway of care for patients with GPP due to the lack of guidelines and approved treatments, limited number of specialists, issues related to hospital and emergency department access (the issue magnified in rural communities), and difficulty arranging off-label systemic therapy. Current treatments for GPP include off-label systemic immunosuppressants and biologic therapy for plaque psoriasis. The clinicians reported that most patients with GPP relapse within 1 year of treatment. Furthermore, broad oral systemic immunosuppressants often lead to side effects, including cytopenia, liver and renal toxicity, and increased risk of infection. The clinicians noted that spesolimab targets the inflammatory pathway unique to GPP and should be used as first-line therapy for patients with GPP to address the unmet needs of resolving and preventing flares. The clinicians also noted that treatment goals should include fast control of acute flares, control of symptoms (i.e., pain, itching, swelling, and pustules), prevention of systemic disease (i.e., cardiac, renal, arthritis, and sepsis), and long-term prevention of flares. The clinician input indicated that the discontinuation of spesolimab should be based on clinical response over time and disease progression.
The participating drug plans sought clarification on whether using methotrexate, cyclosporine, and acitretin to treat GPP is considered off-label use, since the product monographs for these drugs state they are indicated for severe psoriasis and do not specify the type of psoriasis. They also expressed concerns on whether the exclusion criteria in the Effisayil trials (i.e., children excluded from the Effisayil 1 trial, patients weighing less than 40 kg excluded from the Effisayil 2 trial, and exclusions due to comorbidities such as primary plaque psoriasis vulgaris, hepatic disease, and congestive heart disease) would be contraindications to spesolimab. Additionally, they sought clarification on defining a clinically meaningful response and when to discontinue spesolimab. Lastly, the drug plans noted that the sponsor’s patient support program, created by the sponsor to limit the impact on publicly funded health care resources, requires further definition.
Several of these concerns were addressed in the sponsor’s model:
The submitted model accounted for long-term flare prevention.
The submitted model defined achieving a GPPGA subscore of 0 as a meaningful response to treatment.
In addition, CDA-AMC addressed some of these concerns as follows:
CDA-AMC compared spesolimab with best available care (BAC) by incorporating the costs and GPP flare probability associated with BAC into the model as a scenario analysis. However, the efficacy of BAC in treating acute flares and BAC-related adverse events (AEs) could not be assessed due to the lack of data.
CDA-AMC was unable to address the following concerns raised by the input:
CDA-AMC was not able to resolve issues around limited specialists, issues related to hospital or emergency department access (the issue magnified in rural communities), and difficulty arranging off-label systemic therapy.
Economic Review
The current review is for spesolimab (Spevigo) for the prevention of GPP flares in adults and pediatric patients aged 12 years and older who have a history of GPP flares, and the treatment of flares that have a GPPGA total score of 2 or greater.
Economic Evaluation
Summary of Sponsor’s Economic Evaluation
Overview
The sponsor submitted a cost-utility analysis of spesolimab for the treatment and prevention of GPP flares compared with no treatment.
The modelled population receiving treatment for acute flares with a GPPGA score of 2 or greater and the prevention of GPP flares consisted of adults and pediatric patients aged 12 years and older, with a history of GPP, and was in line with the reimbursement request.
The recommended dosing of spesolimab for GPP flare prevention is a loading dose of 600 mg followed by 300 mg every 4 weeks by subcutaneous (SC) injection.1 The recommended dosing for the treatment of flares is 900 mg by IV infusion.1 If flare symptoms persist, an additional treatment with 900 mg may be administered by IV infusion 1 week after the initial treatment. Spesolimab for GPP flare prevention is provided in 150 mg prefilled syringes in a package of 2 syringes of the same size. Spesolimab for flare treatment is provided in 450 mg vials in a package of 2 vials of the same size. The sponsor-submitted costs for spesolimab were $3,650.00 per 150 mg syringe and $10,950 per 450 mg vial. The annual cost of preventive treatment with spesolimab was estimated to be $95,229 per patient per year as assumed by the sponsor. The cost of spesolimab per treatment for a flare was $21,900 per dose as assumed by the sponsor, and patients could receive up to 2 doses per flare ($43,800 per patient per flare). The sponsor did not include administration costs for spesolimab, which were assumed to be covered by the sponsor for spesolimab IV and self-administered for spesolimab SC. In the model, spesolimab was compared with no treatment; therefore, no comparator treatment costs were included in the model.
The clinical outcomes of interest reported in the analysis were QALYs and life-years over a 69-year time horizon. Discounting of 1.5% per year was applied to both costs and health-related outcomes, and a 1-week cycle length was used. The base-case perspective was that of the Canadian publicly funded health care payer.
Model Structure
The sponsor submitted a Markov cohort-level model consisting of 8 health states.2 Among these, 2 states modelled the no-treatment patients (GPP flare-free and GPP flare), 4 states modelled patients treated with spesolimab (first GPP flare among patients previously flare-free, second GPP flare among patients previously flare-free, first GPP flare, and second GPP flare), and the persistent-flare state modelled patients from both treatment arms experiencing a persistent GPP flare. GPP flare-free states were defined as having a GPPGA subscore of 0. GPP flare states were defined as a GPPGA total score of 2 or greater. The GPP flare re-treatment states (second GPP flare-free and second GPP flare states) were tunnelling states to allow patients treated with spesolimab who received IV treatment for their first acute flare, became flare-free, and experienced a second acute flare to receive IV treatment again. The sponsor’s model structure is presented in Figure 1.
The patient flow was described as follows: patients entered the model in the GPP flare-free state according to their respective treatment arm. During each model cycle, patients could remain flare-free or transition to a GPP flare state. Patients in a flare state could remain in that state for a maximum of 2 weeks and then transition to the persistent GPP flare state for up to 11 additional weeks or return to the second flare-free state at each cycle. Patients treated with spesolimab could transition from the second flare-free state to the second GPP flare state to receive IV re-treatment for a second acute flare. Patients in a second acute-flare state could return to the second GPP flare-free state or remain in the second GPP flare state for a maximum of 2 weeks. Patients who experienced an acute flare after 2 weeks in the second GPP flare state transitioned to the persistent GPP flare state for a maximum of 11 weeks. Following the second nonresponse to treatment with spesolimab, patients transitioned from the persistent-flare state to the no-treatment health state (i.e., patients stopped treatment and did not receive spesolimab for the prevention or treatment of acute flares). Death was the absorbing state.
Model Inputs
The baseline characteristics for the model’s cohort were derived from the Effisayil 1 and Effisayil 2 trials. Effisayil 1 was a phase II trial to evaluate the efficacy and safety of spesolimab for the treatment of GPP flares. Fifty-three patients were enrolled in the trial, and eligible patients were between the ages of 18 and 75 years. Effisayil 2 was a phase IIb trial to evaluate the efficacy and safety of spesolimab for the prevention of GPP flares. Eligible patients were between the ages of 12 to 75 years, where adolescents aged 12 to 17 years made up 6.5% of the trial population of 31 patients. Both trials’ inclusion criteria required a diagnosis of GPP according to the European Rare and Severe Psoriasis Expert Network (ERASPEN), which involves physicians observing the presence of primary sterile visible pustules on nonacral skin, and that patients enrolled were not on concomitant GPP treatment. The average age was 41 years, the proportion of female participants was 63.2%, and the mean weight was 72.04 kg. GPP flare scoring was based on the modified GPPGA subscore used in the Effisayil 1 and Effisayil 2 trials and ranged from 0 (no visible pustules) to 4 (severe pustulation).2
Treatment efficacy was based on data directly observed in the Effisayil 1 and Effisayil 2 trials. Transition probabilities for the prevention of GPP flares were estimated based on the number of patients receiving spesolimab in the Effisayil 2 trial and experiencing a GPP flare. Transition probabilities for returning to GPP flare-free states after spesolimab treatment for acute GPP flares at week 1 and week 2 were based on results from the Effisayil 1 trial. Transition probabilities for patients receiving no treatment were based on results from the respective placebo arms of the Effisayil 2 (i.e., probability of a GPP flare) and Effsayil 1 (i.e., probability of returning to flare-free state) trials.3,4 Transition probabilities for the persistent GPP flare substates for both treatment arms were based on direct data from an observational study from the Central and Eastern Europe GPP Expert Network.5 The discontinuation rate for spesolimab due to AEs could occur in the first 8 weeks of the model and was based on the Effisayil 2 trial.
The mortality rate was sourced from age-dependent life expectancy tables from Statistics Canada.6 An excess mortality rate of 5.3% per flare was applied to patients experiencing flares and sourced from a retrospective public claims database study in Brazil.7
Utility scores were estimated based on the EQ-5D-5L data collected in the Effisayil 1 and Effisayil 2 trials. The EQ-5D-5L data from the Effisayil 1 trial estimated utilities for the GPP flare and persistent GPP flare states, mapped using UK tariffs. The EQ-5D-5L data from the Effisayil 2 trial estimated utilities for the GPP flare-free states, mapped using the Canadian time trade-off values.8
The sponsor’s model included all grade 3 to grade 4 AEs reported, informed by results of the Effisayil 1 and Effisayil 2 trials. A disutility for each AE occurred for 4 weeks and was obtained from multiple sources of published literature, including Perwitasari et al. (2021),9 Beusterien et al. (2010),10 Sullivan and Ghushchyan (2006),11 Ahmad et al. (2023),12 and Pataky et al. (2014).13 Age- and sex-adjusted disutilities due to aging in the Canadian population were based on regression coefficients reported in the literature.14
The sponsor included costs related to drug acquisition, medical costs associated with flare management, follow-ups, and AEs. The details of drug acquisition costs were previously outlined. The weighted average cost of managing an acute GPP flare included resource use in the outpatient setting, emergency department, and inpatient setting.15 The weighted average cost of managing a persistent GPP flare included resource use in the outpatient and inpatient settings based on expert opinion. The flare-free management cost included follow-up every 6 months, based on expert opinion. Flare, persistent flare, and flare-free management costs were sourced from Canadian literature, including Tarride et al. (2023)16 and the Ontario Schedule of Benefits: Physician Services.17 Resource use and expenses related to AE management were sourced from the Ontario Schedule of Benefits: Physician Services17 and the Canadian Institute for Health Information.18 Drug reaction with eosinophilia and systemic symptoms (DRESS) was associated with additional stays in an intensive care unit.18,19
Summary of Sponsor’s Economic Evaluation Results
The sponsor’s base-case analysis was run probabilistically (5,000 iterations). Sensitivity and scenario analyses were conducted deterministically. The base-case probabilistic and deterministic results were similar. The probabilistic findings are presented subsequently.
Base-Case Results
Spesolimab was associated with an incremental life-year gain of 9.23, an incremental QALY gain of 7.31, and an incremental cost of $1,962,159, resulting in an ICER of $280,172 per QALY gained compared with no treatment (Table 3).
The results showed that the additional QALYs associated with spesolimab were mainly attributed to the reduction in mortality (due to experiencing fewer flares) for patients treated with spesolimab. The increased costs for patients treated with spesolimab were driven by the drug acquisition cost for the prevention of flares. The results showed that approximately 99% of the total incremental life-years, 99% of the total incremental QALYs, and 96% of the total incremental costs were accrued during the post-trial period (i.e., beyond 48 weeks).
Table 3: Summary of the Sponsor’s Economic Evaluation Results
Drug | Total costs ($) | Incremental costs ($) | Total QALYs | Incremental QALYs | ICER vs. no treatment ($/QALY) |
|---|---|---|---|---|---|
No treatment | 262,754 | Reference | 12.86 | Reference | Reference |
Spesolimab | 2,224,913 | 1,962,159 | 20.16 | 7.31 | 280,172 |
ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year; vs. = versus.
Source: Sponsor’s pharmacoeconomic submission.2
Additional results from the sponsor’s submitted economic evaluation base case are presented in Appendix 3.
Sensitivity and Scenario Analysis Results
The sponsor evaluated several scenarios altering the time horizon, applying discount rates for costs and QALYs, applying the discontinuation of preventive treatment with spesolimab for the entire time horizon, applying the Dermatology Life Quality Index and EQ-5D-3L utility values from the Effisayil 1 trial, not allowing re-treatment following nonresponse, and including administration costs. Reducing the time horizon from 69 years to 1, 5, and 10 years had the most significant impact on the results (the ICER increasing by 98%, 74%, and 43%, respectively). Not allowing spesolimab re-treatment after the failure of 1 acute treatment resulted in an ICER of $251,873 per QALY gained compared with no treatment.
The sponsor also conducted a scenario analysis from a societal perspective that considered the indirect costs associated with productivity loss and travel costs. In this analysis, relative to no treatment, the ICER was $261,136 per QALY gained. This result was similar to the sponsor’s base-case analysis using a health care payer perspective.
Appraisal of the Sponsor’s Economic Evaluation
CDA-AMC identified several key limitations to the sponsor’s analysis that have notable implications on the economic analysis:
The treatments used in clinical practice in Canada were not included as comparators. In the sponsor’s submitted model, spesolimab was compared with no treatment, which is in line with evidence from the Effisayil 1 and Effisayil 2 trials. However, in clinical practice, despite the lack of available GPP-specific treatments, the majority of patients would be treated with BAC, which includes the off-label use of plaque psoriasis treatments (i.e., biologic drugs targeting interleukins, biologic drugs targeting tumour necrosis factor alpha, and nonbiologic systemic drugs). The clinical experts consulted by CDA-AMC noted that patients being treated with BAC would likely experience fewer flares compared with patients receiving no treatment. Furthermore, the clinical experts noted that if spesolimab treatment fails to prevent or treat GPP flares, patients will likely discontinue spesolimab and return to treatment with BAC instead of receiving no treatment. The sponsor’s submitted model has likely overestimated the incremental benefit of spesolimab in clinical practice in Canada by comparing it with no treatment rather than BAC.
CDA-AMC was unable to address this limitation due to a lack of clinical data comparing spesolimab with BAC. CDA-AMC notes that the cost-effectiveness of spesolimab compared with treatments currently being used for the prevention and treatment of GPP flares is unknown.
In a scenario analysis, CDA-AMC applied the annual risk of GPP flares while on BAC, based on a retrospective chart-review study conducted in Canada.20 In this study, patients had an average of 0.5 flares per year. The annual rate of flares from the study was converted to a weekly rate before calculating the weekly probability using the same method the sponsor used to calculate the weekly probability using the rate obtained from the Effisayil 2 trial. Due to a lack of data, the efficacy of BAC treatment for acute flares and BAC-related AEs was not included. In addition to modifying the weekly probability of GPP flares for the comparator group, CDA-AMC included the average annual cost of BAC for preventive therapy and the average cost of BAC per GPP flare from the sponsor’s budget impact analysis (BIA). CDA-AMC recognizes that this study included only 15 patients from 4 participating centres in Canada; however, this scenario analysis aimed to explore the uncertainty around the cost-effectiveness of spesolimab compared with BAC.
The flare-related mortality is likely overestimated. In the submitted model, the sponsor applied an excess mortality rate of 5.3% each time a patient experienced a GPP flare. The sponsor based the excess mortality estimate on a public claims database study from Brazil, where 5.3% of patients with GPP admitted to the hospital had a fatal outcome during hospitalization.7 Furthermore, the sponsor assumed that of the patients experiencing a flare, 36.3% are treated in an inpatient setting, 9.4% are treated in an emergency department setting, and the remaining patients are treated in an outpatient setting, based on a US study.15 The clinical experts consulted by CDA-AMC agreed that the majority of patients with GPP manage flares in an outpatient setting, and that patients who are most likely to die as a result of their GPP flare would be treated in hospital. By applying the flare-related mortality rate to all patients experiencing a flare in the model, the sponsor has applied the excess mortality rate to patients being treated for flares in both an outpatient and hospital setting. As a result, the sponsor has likely overestimated the number of patients dying from GPP in the submitted model.
In the reanalysis, CDA-AMC applied the flare-related mortality rate of 5.3% to the proportion of patients experiencing a flare in a hospital setting, conservatively applying the risk to patients treated in the emergency department setting as well as the inpatient setting (45.6% of patients).
The model structure is not in line with clinical practice. The sponsor’s submitted model uses a 1-week model cycle and assumes that spesolimab is provided as monotherapy for the prevention and treatment of GPP flares compared with no treatment, based on the results from the Effisayil 1 trial. If treatment with spesolimab IV for acute flares fails to return the patient’s GPPGA pustulation subscore back to 0 in the first week, the sponsor assumes 75% of patients will receive a second dose of spesolimab in the second week. Should the second spesolimab treatment fail as well, the patient enters the persistent-flare health state for up to 11 weeks, where no treatment is provided, before returning to the flare-free state and continuing preventive therapy with spesolimab. The sponsor assumed that all patients are re-treated with spesolimab for a second acute flare after the spesolimab treatment was unsuccessful for the first flare. The clinical experts consulted by CDA-AMC note that the sponsor’s assumptions on spesolimab monotherapy, acute treatment, re-treatment, and the persistent-flare state are not in line with clinical practice. First, the clinical experts noted that initially, patients would start with monotherapy in the preventive setting but, if GPP symptoms occur, adjuvant therapy with a different mechanism of action would likely be added. In the acute-flare setting, patients are offered monotherapy initially, but if the patient worsens within 24 to 48 hours, they would be given a second dose of spesolimab or adjuvant treatment rather than waiting a week for additional treatment. Hence, the use of a 1-week cycle length and the use of spesolimab as a monotherapy only did not reflect clinical practice in Canada. Second, the clinical experts noted that if 2 spesolimab IV treatments fail to return the patient’s GPPGA pustulation subscore back to 0, when patients return to the flare-free state, they would not be given spesolimab for preventive therapy. Treatment failure with spesolimab for the first acute flare would likely indicate that the drug is not effective for the patient, and the clinical experts noted that another therapy would be provided in the preventive setting and for subsequent flares. Finally, the clinical experts noted that a persistent-flare state with no additional treatment is highly unlikely to occur in clinical practice. All patients experiencing an acute flare would be given treatment to resolve the GPP flare.
CDA-AMC is not able to address limitations that would require extensive reprogramming and so was unable to address the limitations of the sponsor’s model structure. The sponsor provided a scenario analysis where re-treatment is not allowed after spesolimab fails to treat the first acute flare, which resulted in an approximate $30,000 decrease in the ICER per QALY gained.
The long-term treatment effect of spesolimab is unknown. The sponsor assumed that the treatment effect of spesolimab would persist indefinitely, such that the same risk of GPP flare observed during the 48 weeks of the Effisayil 2 trial would persist for up to 69 years while on treatment. Given that 99% of the incremental QALYs and life-years predicted by the sponsor’s model to be gained with spesolimab were derived based on extrapolated findings rather than observed benefits, the lack of long-term data introduces considerable uncertainty into the analysis.
It was not possible to assess the impact of treatment effect waning in the submitted model due to the model structure and a lack of alternative data. As such, CDA-AMC maintained the sponsor’s assumption regarding the treatment effects persisting over a lifetime time horizon; however, as a result, the clinical outcomes (including QALYs and life-years) for spesolimab may be overestimated in both the sponsor’s and CDA-AMC’s base-case analysis.
Administration costs were not included for spesolimab IV treatment. The IV administration of spesolimab for acute flares would take place in a hospital or infusion clinic and be carried out by a trained health care professional. The sponsor assumed that administration costs would be covered by the sponsor through a patient support program. However, if spesolimab is recommended for public reimbursement, it is unclear whether the sponsor would continue this program and, if so, to what degree. The drug program input indicated the need to further clarify the parameters of this program with the sponsor if spesolimab is recommended for reimbursement. If the patient support program covers administration costs, costs to the health care system will be lower.
In its reanalysis, CDA-AMC included the costs of administering spesolimab IV.
Additional limitations were identified but were not considered to be key limitations. These limitations are outlined subsequently.
The cost of spesolimab was underestimated. The sponsor applied a weekly drug cost for spesolimab that equated to the cost of a 300 mg dose every 4 weeks for preventive treatment with spesolimab. However, the sponsor did not include the cost of the loading dose (600 mg) of spesolimab that is specified in the product monograph.21
CDA-AMC corrected the sponsor’s base case by adding the additional drug acquisition cost associated with the loading dose to the first cycle of the model.
The ICER calculation for the probabilistic results was inaccurate. The sponsor calculated the probabilistic ICER by averaging the ICER values from each iteration of the probabilistic analysis. However, the appropriate way to estimate the ICER in a probabilistic analysis involves first calculating the mean costs and QALYs for each treatment arm and then using these mean values to determine the ICER.
CDA-AMC corrected the sponsor’s results by recalculating the ICER using the differences in mean costs and QALYs for spesolimab versus no treatment, based on the probabilistic results.
Additionally, the following key assumptions were made by the sponsor and appraised by CDA-AMC (Table 4).
Table 4: Key Assumptions of the Submitted Economic Evaluation (Not Noted as Limitations to the Submission)
Sponsor’s key assumption | CDA-AMC comment |
|---|---|
Patients not experiencing a GPP flare are assumed to have a mortality rate that is aligned with the general population. | Inappropriate. Severe GPP flares lead to complications such as heart failure, renal failure, and sepsis.2,22 The clinical experts consulted by CDA-AMC noted that patients with GPP who are not experiencing a flare likely have a higher mortality rate than the general Canadian population. However, this had a limited impact on the overall results. |
Assumptions regarding persistent flares in the sponsor’s pharmacoeconomic report note that patients who continue to experience acute flares after 2 weeks will transition into the persistent-flare state for a maximum of 11 weeks. However, in the model, a proportion of patients transition into the persistent-flare state after the first week and patients who experience an acute flare for 2 weeks move into the second week of the persistent-flare state. | Inappropriate. A proportion of patients do not experience an acute flare for a full 2 weeks before entering persistent-flare states and it would not be possible for these patients to experience a full 11 weeks of persistent flare. This reduces the time in the persistent-flare states and was not modelled in the method proposed by the sponsor. However, this had a limited impact on the overall results. |
CDA-AMC = Canada’s Drug Agency; GPP = generalized pustular psoriasis.
CDA-AMC Reanalyses of the Economic Evaluation
Base-Case Results
The CDA-AMC base case was derived by making changes in model parameter values and assumptions, in consultation with clinical experts. These changes (summarized in Table 5) included correcting the probabilistic ICER, including the cost of the spesolimab loading dose, modifying the flare-related mortality risk, and including administration costs.
Table 5: CDA-AMC Revisions to the Submitted Economic Evaluation
Stepped analysis | Sponsor’s value or assumption | CDA-AMC value or assumption |
|---|---|---|
Corrections to sponsor’s base case | ||
1. ICER calculation for probabilistic results | Calculated by averaging the ICERs from the probabilistic analysis | Calculated using the mean costs and QALYs from the probabilistic analysis |
2. Cost of spesolimab loading dose | Not included | Included |
Changes to derive the CDA-AMC base case | ||
1. GPP flare-related mortality | 5.3% mortality risk applied to all patients experiencing a GPP flare | 5.3% mortality risk applied to hospital inpatients and those visiting an emergency department during a GPP flare (45.6%) |
2. Spesolimab administration costs | Not included | Included |
CDA-AMC base case | ― | 1 + 2 |
CDA-AMC = Canada’s Drug Agency; GPP = generalized pustular psoriasis; ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year.
The CDA-AMC base-case analysis found that spesolimab was associated with 4.60 incremental QALYs (and 5.69 incremental life-years) at an additional cost of $1,986,465. Therefore, the ICER for spesolimab was $431,569 per QALY gained compared with no treatment. A summary of the stepped analysis and base-case analysis results can be found in Table 6. More than 99% of the incremental QALYs and incremental life-years were accrued beyond the time for which CDA-AMC has trial data (i.e., 48 weeks).
Spesolimab has a 0% probability of being cost-effective at a willingness-to-pay threshold of $50,000 per QALY gained. CDA-AMC’s reanalysis was driven by spesolimab preventive treatment acquisition cost, which made up approximately 90% of the total cost for spesolimab.
Table 6: Summary of the Stepped Analysis of the CDA-AMC Reanalysis Results
Stepped analysis | Drug | Total costs ($) | Total QALYs | ICER ($/QALY) |
|---|---|---|---|---|
Sponsor’s base case | No treatment | 263,150 | 12.73 | Reference |
Spesolimab | 2,232,064 | 20.09 | 267,619 | |
Sponsor’s corrected base case | No treatment | 263,150 | 12.73 | Reference |
Spesolimab | 2,239,364 | 20.09 | 268,611 | |
CDA-AMC reanalysis 1 | No treatment | 370,187 | 17.33 | Reference |
Spesolimab | 2,359,129 | 21.94 | 431,758 | |
CDA-AMC reanalysis 2 | No treatment | 263,150 | 12.73 | Reference |
Spesolimab | 2,232,501 | 20.09 | 267,679 | |
CDA-AMC base case (1 plus 2) | No treatment | 370,187 | 17.33 | Reference |
Spesolimab | 2,366,890 | 21.94 | 433,443 | |
CDA-AMC base case (1 plus 2) (probabilistic) | No treatment | 369,915 | 17.42 | Reference |
Spesolimab | 2,356,380 | 22.02 | 431,569 |
CDA-AMC = Canada’s Drug Agency; ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year.
Note: The CDA-AMC reanalysis is based on publicly available prices of the comparator treatments. The results of all steps are presented deterministically unless otherwise indicated, while the cumulative CDA-AMC base case is always presented both deterministically and probabilistically.
Scenario Analysis Results
CDA-AMC undertook price reduction analyses based on the sponsor base case and the CDA-AMC base-case results (Table 7). At a willingness-to-pay threshold of $50,000 per QALY gained, the CDA-AMC base case suggests that a 79% price reduction for spesolimab would be required for spesolimab to be considered cost-effective relative to no treatment (similar to the sponsor’s results). The cost per dose of spesolimab IV for the treatment of an acute flare would be $4,500 at a 79% price reduction, and the annual cost of spesolimab for preventive therapy would be $21,067 in the first year and $19,567 in subsequent years at a 79% price reduction. In the CDA-AMC base case, spesolimab is associated with a gain of 5.69 life-years compared with no treatment.
Table 7: CDA-AMC Price Reduction Analyses
Price reduction analysis | Unit drug cost per mg ($) | ICERs for spesolimab vs. no treatment ($/QALY) | |
|---|---|---|---|
Sponsor base case | CDA-AMC reanalysis | ||
No price reduction | 24 | 269,333 | 431,569 |
10% | 22 | 240,458 | 383,367 |
20% | 19 | 211,582 | 335,176 |
30% | 17 | 182,707 | 286,984 |
40% | 15 | 153,832 | 238,792 |
50% | 12 | 124,957 | 190,601 |
60% | 10 | 96,081 | 142,409 |
70% | 7 | 67,206 | 94,217 |
80% | 5 | 38,331 | 46,026 |
90% | 2 | 9,456 | Dominant |
100% | 0 | Dominant | Dominant |
CDA-AMC = Canada’s Drug Agency; ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year; vs. = versus.
aCalculated using the probabilistic results of the sponsor’s corrected base-case analysis.
CDA-AMC conducted a scenario analysis to explore the impact of including a BAC-related flare probability and treatment costs to estimate the cost-effectiveness of spesolimab compared with BAC. This scenario analysis found that spesolimab was associated with an incremental QALY gain of 3.00 (3.72 incremental life-years) at an additional cost of $1,686,215. The ICER for spesolimab was $561,330 per QALY gained compared with BAC. It is important to note that the efficacy of BAC in treating acute flares and BAC-related AEs could not be included due to a lack of data. This scenario analysis demonstrates that the cost-effectiveness of spesolimab is sensitive to the number of flares in each treatment group due to the mortality benefit of avoiding flares.
Issues for Consideration
The clinician and drug plan input received as part of this review noted the lack of guidelines and approved treatments and challenges in arranging off-label systemic therapy. If spesolimab is reimbursed, it will be the first drug indicated for the treatment of GPP flares. The clinical experts consulted by CDA-AMC noted that off-label drugs will still be used by patients with GPP that is effectively controlled and may be used adjuvant to spesolimab. The challenges of accessing and affording off-label drugs will likely persist.
The sponsor indicated it would provide a patient support program that will cover the costs of IV infusions. If the participating drug plans cannot access the sponsor’s patient support program, these costs will be incurred by the public health care payer. The drug plans noted in the feedback they provided as part of this review that the patient support program requires further definition.
Preventive therapy with spesolimab, which is administered subcutaneously, may be considered to be potentially equity-enhancing for patients who have limited access to infusion centres or a limited ability to access acute dermatology care.
Overall Conclusions
Based on the CDA-AMC Clinical Review and the results of the Effisayil 1 trial, spesolimab results in a meaningful difference for the treatment of acute flares in achieving a GPPGA pustulation subscore of 0 after 1 week of treatment compared with placebo (54.3% of patients on spesolimab versus 5.6% of patients on placebo) with moderate certainty. CDA-AMC’s Clinical Review reported that, in the flare-prevention setting and based on the results of the Effisayil 2 trial, spesolimab results in a meaningful reduction in the proportion of patients experiencing a GPP flare in 48 weeks compared with placebo (12.7% of patients on spesolimab versus 51.6% of patients on placebo) with moderate certainty. The Clinical Review also noted that the use of placebo as defined in the trials is not aligned with the current treatment options used in clinical practice. Additionally, the Clinical Review reported there is limited information available to conclude on the long-term efficacy and safety of spesolimab for GPP. There is no direct or indirect evidence comparing spesolimab with the treatments used in clinical practice in Canada, which include methotrexate, cyclosporine, retinoids, systemic corticosteroids, and treatments used for other types of psoriasis.
CDA-AMC was able to address some limitations associated with the sponsor’s analysis, including applying the flare-related mortality estimate to the appropriate group of patients and including administration costs associated with spesolimab IV. The results of the CDA-AMC base case are similar to the sponsor’s analysis. In the CDA-AMC base case, spesolimab is associated with 4.60 incremental QALYs at an additional cost of $1,986,465, resulting in an ICER of $431,569 per QALY gained compared with no treatment. The incremental cost is driven by the acquisition of spesolimab for preventive therapy, which makes up approximately 90% of the total cost for spesolimab. The higher life expectancy associated with spesolimab (5.69 incremental life-years) as a result of experiencing fewer flares drove the increase in QALYs. The majority of the benefit (99%) was incurred after 48 weeks (beyond which no clinical information is available for spesolimab). Cost-effectiveness is therefore heavily influenced by the long-term survival benefit for which the evidence is uncertain due to a lack of long-term information. For spesolimab to be considered cost-effective compared with no treatment at a threshold of $50,000 per QALY gained, the cost per dose of spesolimab for the treatment of acute flares would need to be $4,500, and the annual per-patient cost of spesolimab for preventive therapy would be $21,067 in the first year and $19,567 in subsequent years, reflecting a price reduction of approximately 79%.
CDA-AMC was unable to resolve several key limitations in the sponsor’s analysis. First, the review team was unable to resolve the lack of a comparison between spesolimab and the best care available in clinical practice in Canada, given the lack of comparative efficacy data. As such, the cost-effectiveness of spesolimab compared with current clinical practice remains unknown. Additionally, the review team was unable to resolve the limitations associated with the model structure, specifically, the use of adjuvant therapies and acute treatment changes within 24 to 48 hours. Finally, CDA-AMC was unable to resolve uncertainty in the duration of the treatment effect for spesolimab for the prevention of GPP flares; therefore, the reported ICER is dependent on the treatment benefit observed in the 48-week trial persisting over the 69-year time horizon. Because these limitations remain unresolved, the reanalysis performed by CDA-AMC is highly uncertain.
References
1.Spevigo (spesolimab for injection): Solution for subcutaneous injection 150 mg/1 mL (150 mg/mL); Concentrate for solution for intravenous infusion 450 mg/7.5 mL (60 mg/mL) [draft product monograph]. Burlington (ON): Boehringer Ingelheim (Canada) Ltd.; 2023 Aug 1.
2.Pharmacoeconomic evaluation [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Spevigo® (spesolimab for injection), solution for subcutanenous injection (150 mg/1mL) and concentration for solution for intravenous infusion (450 mg/7.5 mL [60 mg/mL]). Burlington (ON): Boehringer Ingelheim (Canada) Ltd.; 2024.
3.Bachelez H, Choon SE, Marrakchi S, et al. Trial of Spesolimab for Generalized Pustular Psoriasis. N Engl J Med. 2021;385(26):2431-2440. PubMed
4.Morita A, Strober B, Burden AD, et al. Efficacy and safety of subcutaneous spesolimab for the prevention of generalised pustular psoriasis flares (Effisayil 2): an international, multicentre, randomised, placebo-controlled trial. Lancet. 2023;402(10412):1541-1551. PubMed
5.Wolf P, Ceovic R, Conrad C, et al. Characteristics and management of generalized pustular psoriasis (GPP): Experience from the Central and Eastern Europe (CEE) GPP Expert Network. J Eur Acad Dermatol Venereol. 2024. PubMed
6.Table 17-10-0005-01 Population estimates on July 1st, by age and sex [sponsor-supplied reference]. Ottawa (ON): Statistics Canada; 2023. Accessed 2023 Oct.
7.Duarte GV, Esteves de Carvalho AV, Romiti R, et al. Generalized pustular psoriasis in Brazil: A public claims database study. JAAD Int. 2022;6:61-67. PubMed
8.Xie F, Pullenayegum E, Gaebel K, et al. A Time Trade-off-derived Value Set of the EQ-5D-5L for Canada. Med Care. 2016;54(1):98-105. PubMed
9.Perwitasari DA, Febriana SA, Tristiana RS. Quality of Life of Drug Reaction with Eosinophilia and Systemic Symptom (DRESS) and Stevens-Johnson Syndrome (SJS) and/or Toxic Epidermal Necrolysis (TEN) Patients. Patient Prefer Adherence. 2021;15:329-335. PubMed
10.Beusterien KM, Davies J, Leach M, et al. Population preference values for treatment outcomes in chronic lymphocytic leukaemia: a cross-sectional utility study. Health Qual Life Outcomes. 2010;8:50. PubMed
11.Sullivan PW, Ghushchyan V. Preference-Based EQ-5D index scores for chronic conditions in the United States. Med Decis Making. 2006;26(4):410-420. PubMed
12.Ahmad H, Campbell JA, van der Mei I, et al. Estimating the disutility of relapse in relapsing-remitting and secondary progressive multiple sclerosis using the EQ-5D-5L, AQoL-8D, EQ-5D-5L-psychosocial, and SF-6D: implications for health economic evaluation models. Qual Life Res. 2023;32(12):3373-3387. PubMed
13.Pataky R, Phillips N, Peacock S, Coldman AJ. Cost-effectiveness of population-based mammography screening strategies by age range and frequency. Journal of Cancer Policy. 2014;2(4):97-102.
14.Guertin JR, Feeny D, Tarride JE. Age- and sex-specific Canadian utility norms, based on the 2013-2014 Canadian Community Health Survey. CMAJ. 2018;190(6):E155-E161. PubMed
15.Zema CL, Valdecantos WC, Weiss J, Krebs B, Menter AM. Understanding Flares in Patients With Generalized Pustular Psoriasis Documented in US Electronic Health Records. JAMA Dermatol. 2022;158(10):1142-1148. PubMed
16.Tarride JE, Prajapati VH, Lynde C, Blackhouse G. The burden associated with generalized pustular psoriasis: A Canadian population-based study of inpatient care, emergency departments, and hospital- or community-based outpatient clinics. JAAD Int. 2023;12:90-98. PubMed
17.Schedule of benefits for physician services under the Health Insurance Act: (June 29, 2023 (effective July 24, 2023)) [sponsor-supplied reference]. Toronto (ON): Ontario Ministry of Health; 2023: https://www.health.gov.on.ca/en/pro/programs/ohip/sob/physserv/sob_master.pdf.
18.Canadian Institute for Health Information. Patient Cost Estimator [sponsor-supplied reference]. 2021; https://www.cihi.ca/en/patient-cost-estimator.
19.Wolfson AR, Zhou L, Li Y, Phadke NA, Chow OA, Blumenthal KG. Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) Syndrome Identified in the Electronic Health Record Allergy Module. J Allergy Clin Immunol Pract. 2019;7(2):633-640. PubMed
20.Boehringer Ingelheim Group of Companies. Canadian Pustular Psoriasis Study (CAPPS): Examining the history, clinical characteristics, disease burden, treatments and healthcare resource utilization for Generalized Pustular Psoriasis (GPP) flare in Canada [sponsor-supplied reference]. 2023.
21.Spevigo® (spesolimab for injection) [product monograph; sponsor-supplied reference]. Boehringer Ingelheim, Ltd. Accessed 2023 Aug 1.
22.Choon SE, Lai NM, Mohammad NA, Nanu NM, Tey KE, Chew SF. Clinical profile, morbidity, and outcome of adult-onset generalized pustular psoriasis: analysis of 102 cases seen in a tertiary hospital in Johor, Malaysia. Int J Dermatol. 2014;53(6):676-684. PubMed
23.Budget Impact Analysis [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Spevigo® (spesolimab for injection), solution for subcutanenous injection (150 mg/1mL) and concentration for solution for intravenous infusion (450 mg/7.5 mL [60 mg/mL]). Burlington (ON): Boehringer Ingelheim (Canada) Ltd.; 2024.
24.First Nations and Inuit Health Branch Non-Insured Health Benefits Program: Annual Report 2021/2022 [sponsor-supplied reference]. Ottawa (ON): Indigenous Services Canada; 2022.
25.Understanding the Gap 2.0 A Pan-Canadian Analysis of Prescription Drug Insurance Coverage [sponsor-supplied reference]. Ottawa (ON): The Conference Board of Canada; 2022.
26.Dietitians of Canada. The World Health Organization (WHO) Growth Charts for Canada [sponsor-supplied reference]. 2019; https://www.dietitians.ca/Secondary-Pages/Public/Who-Growth-Charts.aspx. Accessed 2024 Jan 24.
27.Annual Demographic Estimates: Canada, Provinces and Territories 2023. Ottawa (ON): Statistics Canada; 2024: https://publications.gc.ca/collections/collection_2024/statcan/91-215-x2023002-eng.pdf. Accessed 2024 Aug 15.
28.Saskatchewan Drug Plan and Extended Benefits Branch. Drug Cost Assistance. https://www.saskatchewan.ca/residents/health/prescription-drug-plans-and-health-coverage/extended-benefits-and-drug-plan/drug-cost-assistance. Accessed 2024 Sep 4.
29.Ontario Ministry of Health. Get help with high prescription drug costs. 2024; https://www.ontario.ca/page/get-help-high-prescription-drug-costs. Accessed 2024 Sep 4.
30.Government of British Columbia. Exceptional funding of EDRDs. 2024; https://www2.gov.bc.ca/gov/content/health/health-drug-coverage/pharmacare-for-bc-residents/what-we-cover/exceptional-funding-edrd. Accessed 2024 Sep 4.
31.Ontario Ministry of Health. Learn about OHIP+. 2024; https://www.ontario.ca/page/learn-about-ohip-plus. Accessed 2024 Sep 4.
32.Pan-Canadian Prescription Drug Data Landscape. Ottawa (ON): Canadian Institute for Health Information (CIHI); 2024: https://www.cihi.ca/sites/default/files/document/pan-canadian-prescription-drug-data-landscape-report-en.pdf. Accessed 2024 Aug 15.
Appendix 1: Cost Comparison Table
Please note that this appendix has not been copy-edited.
The comparators presented in the following table have been deemed to be appropriate based on feedback from clinical experts. Comparators may be recommended (appropriate) practice or actual practice. Existing Product Listing Agreements are not reflected in the table and, as such, the table may not represent the actual costs to public drug plans.
Table 8: CDA-AMC Cost Comparison Table for the Treatment and Prevention of Flares Due to Generalized Pustular Psoriasis
Treatment | Strength or concentration | Form | Price | Recommended dosage | One-time cost and daily cost ($) | One-time cost and annual cost ($) |
|---|---|---|---|---|---|---|
Spesolimab (Spevigo) | 60 mg/mL | 450 mg vial, concentrate for solution for IV infusion (2 vials per package) | 21,900.0000a | Treatment of flares: 900 mg single dose, if flare persists, an additional 900 mg dose may be administered 1 week after the initial dose | 21,900.00b | If 1 dose administered: 21,900b If 2 doses administered: 43,800 |
Spesolimab (Spevigo) | 150 mg/mL | 150 mg prefilled syringe, concentrate for solution for subcutaneous injection (2 syringes per package) | 7,300.0000a | Prevention of flares: 600 mg loading dose, followed by 300 mg maintenance every 4 weeks | First year: 280.70 Subsequent years: 260.71 | First year: 102,526c Subsequent years: 95,226c |
CDA-AMC = Canada’s Drug Agency.
aSponsor’s submitted price.
bCost includes a single dose of 900 mg and does not include any additional dose due to persistent flares.
cAssumes a 1-time 600 mg loading dose followed by 12.045 doses of 300 mg. Subsequent years assume 13.045 doses of 300 mg administered in a year.
Table 9: CDA-AMC Cost Comparison Table for Treatments Used Off-Label for the Treatment of Generalized Pustular Psoriasis Flares
Treatment | Strength or concentration | Form | Price | Recommended dosage | Daily cost ($) | Cost per flare ($) |
|---|---|---|---|---|---|---|
Acitretin (Mint-Acitretin) | 10 mg 25 mg | Capsule | 1.2965 2.7700 | Adult: 25 mg daily, maximum dose of 50 mg dailya Adolescent: 0.5 mg/kg to 1 mg/kg per dayb | 0.21 to 0.42 | 78 to 155 |
Apremilastc (Auro-Apremilast) | 30 mg | Tablet | 18.7237 | 30 mg twice daily | 2.87 | 1,049 |
Betamethasone dipropionate (Diprosone) | 0.05% W/W | Ointment | 0.1980 | Thin film applied to cover affected area once to twice daily | 0.03 | 10 |
Calcipotriol (Dovonex) | 50 mcg/g | Ointment | 1.1507 | Adults (aged > 14 years): 5 mg/weekd Adolescents aged 11 to 14 years: 3.75 mg/weekd | 1.26 | 460 |
Cyclosporine (Sandoz Cyclosporine) | 25 mg 50 mg 100 mg | Capsule | 0.7870 1.5350 3.0720 | 2 mg/kg/day, maximum 5 mg/kg/day | Adult: 0.35 to 0.88 Adolescent: 0.24 to 0.59 | Adult: 129 to 323 Adolescent: 86 to 215 |
Guselkumabc (Tremfya) | 100 mg/mL | 1 mL Prefilled syringe or autoinjector | 3,059.7400 | 100 mg at week 0 and 4 | 16.75 | 6,119 |
Infliximab (Renflexis biosimilar) | 5 mg/mL | 20 mL Vial | 493.0000 | 5 mg/kg initial dose followed by additional dose at 2 and 6 weeks | Adult: 16.20 Adolescent: 12.15 | Adult: 5,916 Adolescent: 4,437 |
Ixekizumab (Taltz) | 80 mg/mL | 1 mL Prefilled syringe or autoinjector | 1,865.1400 | Adults and adolescents > 50 kg: 160 mg at week 0, then 80 mg at weeks 2, 4, 6, 8, 10, and 12 Adolescents 25 kg to 50 kg: 80 mg < 25 kg: 40 mg | 40.85 | 1,491 |
Methotrexate - PO (Apo-Methotrexate) | 2.5 mg | Tablet | 0.2513 | 10 mg to 25 mg per week | 0.01 to 0.03 | 4 to 10 |
Secukinumab (Cosentyx) | 150 mg/mL | 1 mL Prefilled syringe | 934.0400 | Adult: 300 mg at weeks 0, 1, 2, 3, and 4 Adolescent:
| Adult: 25.57 Adolescent: 12.79 | Adult: 9,340 Adolescent: 4,670 |
Ustekinumab (Jamteki) | 90 mg/mL | 0.5 mL 1 mL Prefilled syringe | 2,755.8840 |
| 15.09 | 5,512 |
CDA-AMC = Canada’s Drug Agency; PO = per os (by mouth); SC = subcutaneous.
Note: All prices are from the Ontario Drug Benefit Formulary (accessed August 2024) unless otherwise indicated. Prices assume an adult patient weight of 73.7 kg and an adolescent patient weight of 51.8 kg, and wastage of excess medication in vials, if applicable. Doses for capsules were rounded to the nearest capsule size, if applicable. Assumes a 4-week treatment duration unless a dosing schedule is otherwise specified.
Adult and adolescent dosing specified, when different. Daily and annual costs for adults and adolescents are also indicated when different.
aAlternative dosing provided by clinical expert consulted by CDA-AMC.
bSponsor assumed doing and confirmed by clinical experts consulted by CDA-AMC. The product monograph does not indicate this dosing for pediatrics.
cTreatment used for adults only.
dAssumption of 50 g per dose for adolescents and 100 g per dose for adults.
Table 10: CDA-AMC Cost Comparison Table for Treatments Used Off-Label for the Prevention of Generalized Pustular Psoriasis Flares
Treatment | Strength or concentration | Form | Price | Recommended dosage | Daily cost ($) | Annual cost ($) |
|---|---|---|---|---|---|---|
Acitretin (Mint-Acitretin) | 10 mg 25 mg | Capsule | 1.2965 2.7700 | Adult: 25 mg per day Adolescent: 0.5 mg/kg/day to 1 mg/kg/daya | 2.77 | 1,012 |
Anakinrab (Kineret) | 150 mg/1 mL | Prefilled syringe | 54.6186c | 100 mg daily | 54.62 | 19,949 |
Apremilastb (Auro-Apremilast) | 30 mg | Tablet | 18.7237 | 30 mg twice daily | 37.45 | 13,678 |
Betamethasone Dipropionate (Diprosone) | 0.05% W/W | Ointment | 0.1980 | Thin film applied to cover affected area once to twice dailyd | 0.35 | 129 |
Cyclosporine (Sandoz Cyclosporine) | 25 mg 50 mg 100 mg | Capsule | 0.7870 1.5350 3.0720 | 2 mg/kg/day, maximum 5 mg/kg/day | Adult: 4.61 to 11.54 Adolescent: 3.07 to 7.68 | Adult: 1,683 to 4,214 Adolescent: 1,122 to 2,805 |
Etanercept (Rymti) | 50 mg/1 mL | Prefilled syringe | 236.1800 | Adult: 50 mg once weekly Adolescent: 0.8 mg/kg per week | 33.74 | 12,324 |
Guselkumabb (Tremfya) | 100 mg/mL | 1 mL Prefilled syringe or autoinjector | 3,059.7400 | 100 mg every 8 weeks | 54.64 | 19,957 |
Infliximab (Renflexis) | 5 mg/mL | 20 mL Vial | 493.0000 | 5 mg/kg every 8 weeks | Adult: 246.50 Adolescent: 26.41 | Adult: 90,034 Adolescent: 9,647 |
Ixekizumab (Taltz) | 80 mg/mL | 1 mL Prefilled syringe or autoinjector | 1,865.1400 | Adult: 80 mg every 4 weeks Adolescent:
| 66.61 | 24,330 |
Methotrexate PO (Apo-Methotrexate) | 2.5 mg | Tablet | 0.2513 | 10 mg to 25 mg per week | 0.14 to 0.36 | 52 to 131 |
Secukinumab (Cosentyx) | 150 mg/mL | 1 mL Prefilled syringe | 934.0400 | 300 mg every 4 weeks; alternatively, may be increased to 300 mg every 2 weekse Adolescent:
| Adult: 66.72 to 133.43 Adolescent: 33.36 | Adult: 24,368 to 48,737 Adolescent: 12,184 |
Ustekinumab (Jamteki) | 90 mg/mL | 0.5 mL 1 mL Prefilled syringe | 2,755.8840 |
| Adult: 32.81 Adolescent: 18.75 | Adult: 11,983 Adolescent: 6,848 |
CDA-AMC = Canada’s Drug Agency; PO = per os (by mouth); SC = subcutaneous.
Note: All prices are from the Ontario Drug Benefit Formulary (accessed August 2024) unless otherwise indicated. Assume an adult patient weight of 73.7 kg and an adolescent patient weight of 51.8 kg, and wastage of excess medication in vials, if applicable. Doses for capsules were rounded to the nearest capsule size, if applicable.
Note: Adult and adolescent dosing specified, when different. Daily and annual costs for adults and adolescents are also indicated when different.
aSponsor assumed doing and confirmed by clinical experts consulted by CDA-AMC. The product monograph does not indicate this dosing for pediatrics.
bTreatment used for adults only.
cSaskatchewan formulary (August 2024).
dAssumption of 50 g per dose.
eAlternative dosing provided by clinical expert consulted by CDA-AMC.
Appendix 2: Submission Quality
Please note that this appendix has not been copy-edited.
Description | Yes or No | Comments |
|---|---|---|
Population is relevant, with no critical intervention missing, and no relevant outcome missing. | No | Refer to key limitation: Treatments used in clinical practice in Canada were not included as comparators. |
Model has been adequately programmed and has sufficient face validity. | Yes | No comment. |
Model structure is adequate for decision problem. | No | Refer to the key limitation: The model structure is not in line with clinical practice. |
Data incorporation into the model has been done adequately (e.g., parameters for probabilistic analysis). | Yes | No comment. |
Parameter and structural uncertainty were adequately assessed; analyses were adequate to inform the decision problem. | Yes | No comment. |
The submission was well organized and complete; the information was easy to locate (clear and transparent reporting; technical documentation available in enough detail). | No | Refer to Table 4. In addition, the population data in the BIA report was copied in error and did not align with population data used in the BIA model. |
BIA = budget impact analysis.
Appendix 3: Additional Information on the Submitted Economic Evaluation
Please note that this appendix has not been copy-edited.
GPP = generalized pustular psoriasis; GPPGA = Generalized Pustular Psoriasis Physician Global Assessment; SC = subcutaneous injection.
*Up to 2 doses.
**Up to 3 months in “persistent GPP flare” health state.
Source: Sponsor’s pharmacoeconomic submission.2
Detailed Results of the Sponsor’s Base Case
Table 12: Disaggregated Summary of the Sponsor’s Economic Evaluation Results
Parameter | Spesolimab | No treatment |
|---|---|---|
Discounted LYs | ||
Total | 25.53 | 16.30 |
By health state | — | — |
Flare-free | 25.15 | 15.27 |
GPP flare | 0.17 | 0.43 |
Persistent flare | 0.21 | 0.60 |
Discounted QALYs | ||
Total | 20.16 | 12.86 |
By health state | — | — |
Flare-free | 19.99 | 12.32 |
GPP flare | 0.06 | 0.17 |
Persistent flare | 0.13 | 0.39 |
Discounted costs ($) | ||
Total | 2,224,913 | 262,754 |
Acquisition cost, acute | 71,466 | 0 |
Acquisition cost, prevention | 2,033,904 | 0 |
Administration | 0 | 0 |
Medical costs | 94,694 | 246,992 |
Adverse events cost | 24,848 | 15,761 |
GPP = generalized pustular psoriasis; LY = life-year; QALY = quality-adjusted life-year.
Source: Sponsor’s pharmacoeconomic submission.2
Appendix 4: Additional Details on the CDA-AMC Reanalyses and Sensitivity Analyses of the Economic Evaluation
Detailed Results of the CDA-AMC Base Case
Please note that this appendix has not been copy-edited.
Table 13: Disaggregated Summary of the CDA-AMC Economic Evaluation Results
Parameter | Spesolimab | No treatment |
|---|---|---|
Discounted LYs | ||
Total | 28.04 | 22.35 |
By health state | — | — |
Flare-free | 27.54 | 20.90 |
GPP flare | 0.22 | 0.60 |
Persistent flare | 0.28 | 0.86 |
Discounted QALYs | ||
Total | 22.02 | 17.42 |
By health state | — | — |
Flare-free | 21.79 | 16.66 |
GPP flare | 0.08 | 0.23 |
Persistent flare | 0.18 | 0.55 |
Discounted costs ($) | ||
Total | 2,356,380 | 369,915 |
Acquisition cost, acute | 75,264 | 0 |
Acquisition cost, prevention | 2,130,189 | 0 |
Administration | 454 | 0 |
Medical costs | 123,134 | 348,272 |
Adverse events cost | 27,339 | 21,642 |
CDA-AMC = Canada’s Drug Agency; GPP = generalized pustular psoriasis; LY = life-year; QALY = quality-adjusted life-year.
Scenario Analyses
Table 14: Results of the CDA-AMC Scenario Analysis Comparing Spesolimab With Best Available Care
Drug | Total costs ($) | Incremental costs ($) | Total QALYs | Incremental QALYs | ICER vs. BAC ($/QALY) |
|---|---|---|---|---|---|
No treatment | 768,388 | Reference | 19.36 | Reference | Reference |
Spesolimab | 2,454,602 | 1,686,215 | 22.36 | 3.00 | 561,330 |
BAC = best available care; CDA-AMC = Canada’ Drug Agency; ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year; vs. = versus.
Appendix 5: Submitted Budget Impact Analysis and CDA-AMC Appraisal
Please note that this appendix has not been copy-edited.
Table 15: Summary of Key Takeaways
Key takeaways of the budget impact analysis |
|---|
|
CDA-AMC = Canada’s Drug Agency; GPP = generalized pustular psoriasis.
Summary of Sponsor’s Budget Impact Analysis
The sponsor submitted a budget impact analysis (BIA) to estimate the incremental 3-year budget impact of spesolimab for the treatment of GPP flares in adult patients and adolescent patients aged 12 years and older.23 The BIA was undertaken from the perspective of a Canadian public payer over a 3-year time horizon using an epidemiological approach. The sponsor compared a reference scenario in which patients were treated with BAC, consisting of off-label biologic and nonbiologic treatments, with a new-drug scenario in which spesolimab was reimbursed.
Data for the model were obtained from various sources, including published literature,6,16,24 the sponsor’s internal data, and assumptions. Key inputs to the BIA are documented in Table 16.
Key assumptions included:
The proportions of patients on each BAC treatment were obtained from the Canadian Pustular Psoriasis Study (CAPPS) for the treatment and prevention of GPP flares in adults.20 The sponsor assumed the same proportion of adolescent patients on BAC, except for drugs not used in pediatric patients. The sponsor assumed that 15% of patients receiving BAC were not receiving treatment and did not incur costs.
The treatment of acute GPP flares was assumed to last for a mean duration of 4 weeks.
The sponsor assumed all treatments administered in the hospital would be included in the hospital budget and not paid by the public payer. Treatments in an outpatient setting were publicly covered for patients enrolled in a public drug plan.
The sponsor assumed that 81% of flares would have a GPPGA total score of 2 or greater.5
Summary of the Sponsor’s BIA Results
The sponsor estimated that reimbursing spesolimab for the treatment and prevention of GPP flares would result in incremental costs of $474,881 in year 1, $1,953,552 in year 2, and $4,115,625 in year 3, for a 3-year total cost of $6,544,058.
The majority of the estimated budget impact was associated with the prevention of GPP flares, where the sponsor estimated that reimbursing spesolimab for the prevention of GPP flares would result in incremental costs of $464,536 in year 1, $1,929,187 in year 2, and $4,071,542 in year 3, for a total of $6,465,266 over 3 years. The remaining estimated budget impact was associated with reimbursing spesolimab for the treatment of GPP flares, resulting in an incremental cost of $78,792 over 3 years.
Table 16: Summary of Key Model Parameters
Parameter | Sponsor’s estimate |
|---|---|
Target population | |
Pan-Canadian population, aged 12 years and older (excluding Quebec) | 27,560,615 |
Prevalence of GPP | 2.77 per million16 |
Incidence of GPP | 1.95 per million16 |
Coverage rate for < 65 years of agea | 24.75%25 |
Coverage rate for ≥ 65 years of agea | 88.32%25 |
Number of patients eligible for spesolimab treatment | 53 / 75 / 97 |
Annual number of GPP flares per patient Spesolimab BAC | 0.11 0.78 |
Market uptake (3 years) | |
Acute flare treatment uptake (reference scenario) BAC | ███% / ███% / ███% |
Flare prevention uptake (reference scenario) BAC | ███% / ███% / ███% |
Acute flare treatment uptake (new-drug scenario) Spesolimab BAC | ██% / ██% / ██% ██% / ██% / ██% |
Flare prevention uptake (new-drug scenario) Spesolimab BAC | ██% / ██% / ██% ██% / ██% / ██% |
Cost of treatment (per patient, per year) | |
Spesolimab IV for acute flare treatment Spesolimab SC for flare prevention | $29,409b First year: $102,200 Subsequent years: $94,900 |
BAC for acute flare treatment BAC, for flare prevention | $2,684c $17,754c |
BAC = best available care; GPP = generalized pustular psoriasis; SC = subcutaneous injection.
Note: To calculate weight-based drug doses, the mean adult patient weight was assumed to be 73.7 kg, sourced from the Effisayil 1 trial. The mean adolescent patient weight was assumed to be 51.8 kg, sourced from the Canadian growth chart.26
aProvince specific age-specific coverage rates were also provided.
bAssuming 65.7% of patients received 1 dose of 900 mg and 34.3% received 2 doses of 900 mg.
cAssumed 5.7% of the eligible population for SC prevention were adolescents and 94.3% were adults.
Source: Sponsor’s budget impact analysis submission.23
CDA-AMC Appraisal of the Sponsor’s BIA
CDA-AMC identified several key limitations to the sponsor’s analysis that have notable implications on the results of the BIA:
The use of both prevalence and incidence rates overestimated the eligible population: In the base year of the analysis, the sponsor added the prevalent and incident populations with GPP together to estimate the number of patients eligible for treatment with spesolimab. For years 1 to 3 of the analysis, the sponsor added the base year’s incident population to the prior year’s total eligible population. Given that the prevalence rate reflects the number of people with GPP at a given point in time (i.e., accounts for people entering and leaving the prevalent cohort through new diagnoses and death), the sponsor has overestimated the number of people with GPP in the BIA. In fact, in this approach, the sponsor has modelled a situation in which the prevalence of GPP is increasing steadily over time. A more appropriate method of estimating the eligible population would be using a strictly prevalence-based approach and accounting for the population growth rate of the Canadian population.
In the reanalysis, CDA-AMC used a prevalence-based approach by applying the Canadian annual growth rate to the pan-Canadian population each year of the BIA, then applying the prevalence rate to the estimated population. Statistics Canada estimated the growth rate to be 1.8% in 2021 to 2022, and 2.9% in 2022 to 2023, which include infants and children under 12 years old.27 To take a conservative approach, a 1.8% growth rate was used in the CDA-AMC reanalysis.
The prevalence of GPP was likely underestimated: The sponsor assumed that the GPP prevalence rate was 2.77 per million based on a Canadian population study using data from the Canadian Institute for Health Information (CIHI) that sought to identify people with GPP in hospital- and community-based outpatient clinics.16 This study excluded physician claims data and acknowledged that reporting visits to emergency departments and hospital- and community-based outpatient clinics is not mandatory in all jurisdictions in Canada. In Alberta, reporting of emergency department and community clinics is mandatory; in a sensitivity analysis considering only the Alberta population the prevalence of GPP was estimated to be 5.44 per million individuals.16 Additionally, this study noted that GPP burden studies in other countries that included physician claims data resulted in higher GPP prevalence estimates. Therefore, the GPP prevalence rate used by the sponsor likely underestimated the prevalent population with GPP in Canada.
In reanalysis, CDA-AMC used the prevalence rate of 5.44 per million individuals based on Alberta’s data from the same Canadian population study sourced by the sponsor. Clinical experts consulted by CDA-AMC agreed that this prevalence was more reflective of the number of patients with GPP in Canada This prevalence rate may still be an underestimate due to the exclusion of physician claims data.
The market share estimates do not align with clinical expectations: The sponsor assumed that spesolimab would take market share from BAC based on internal estimates. In the acute setting, the sponsor estimated that spesolimab would capture ██% in year 1, ██% in year 2, and ██% in year 3. In the prevention setting, the sponsor estimated that spesolimab would capture ██% in year 1, ██% in year 2, and ██% in year 3. Clinical experts consulted by CDA-AMC note that should spesolimab be reimbursed, it would be the only approved medication for GPP, and that spesolimab would be considered used as a first-line therapy. Hence, clinical experts expect a more rapid adoption compared with the sponsor’s estimates in the BIA, especially in the acute flare treatment setting. The sponsor submitted the survey responses that determined the market mix. The survey results supported the feedback from clinical experts consulted by CDA-AMC on the rapid adoption of spesolimab treatment for acute flares. In the preventive setting, clinical experts consulted by CDA-AMC noted that patients would switch from BAC to spesolimab at the time of experiencing a flare. There are patients currently well managed with BAC who would not switch to spesolimab as preventive therapy. Based on the number of patients treated for GPP flares in Ontario, clinical experts estimated that spesolimab will have a higher market uptake than the sponsor’s estimated in the preventive setting, but slower adoption compared with spesolimab for the treatment of acute flares.
In reanalysis, CDA-AMC revised the spesolimab market share of 85%, 90%, and 95% in years 1, 2, and 3, respectively. A scenario analysis was conducted to explore the increased spesolimab market uptake of 20%, 50%, and 55% in the preventive setting, based on clinical expert feedback.
The annual number of flares for patients treated with BAC was derived from results of the Effisayil 2 trial placebo group: The sponsor assumed that patients would experience 0.78 flares per patient per year if being treated with BAC in the BIA. However, the placebo group in the Effisayil 2 trial was not being treated with BAC. In the sponsor’s BIA, the cost of BAC was included, but the efficacy of no treatment was used to represent the efficacy of BAC. Clinical experts consulted by CDA-AMC noted that patients being treated with BAC would likely experience fewer flares compared with patients on no treatment. Therefore, using the efficacy of no treatment in the BIA in place of the efficacy of BAC likely resulted in an overestimation of annual flares experienced by patients treated with BAC, and an underestimation of the total budget impact of spesolimab.
CDA-AMC did not address this limitation. It is expected to have minimal impact on the results of the BIA.
The coverage rate did not consider the rare disease program: In the sponsor’s submitted BIA model, the coverage rate estimate was based on the proportion of patients eligible and enrolled in the public drug plan. While the public coverage estimates account for high public coverage of adults 65 years of age and older, additional public funding programs are likely to cover remaining patients. These programs may include high cost drug coverage (e.g., Saskatchewan’s Special Support Program,28 and Ontario’s Trillium drug program29), rare disease programs (e.g., British Columbia’s Expensive Drugs for Rare Diseases program30), and youth coverage programs (e.g., Ontario’s OHIP+ for people 24 years and younger without private coverage31) may cover the cost of spesolimab.32 The sponsor’s approach to estimating the public coverage did not account for coverage under other public funding programs. This may have underestimated the proportion of patients eligible for spesolimab, resulting in an underestimate of the budget impact.
In a scenario analysis, CDA-AMC used the Canadian estimate on the proportion of the population eligible for the public drug plan for those under 65 years of age (i.e., 64%, based on the sponsor’s estimated eligible population).
Markups and dispensing fees were included in the BIA: The BIA included markups and dispensing fees that were province specific. Pharmacy/wholesale markups and dispensing fees may have different criteria depending on the drug plan and subject to change. Hence, the BIA base case should not include these estimates
In the reanalysis, CDA-AMC excluded markups and dispensing fees.
The basket of BAC is not in line with clinical practice: The sponsor estimated the proportion of patients using each drug in BAC based on the retrospective chart-review study by CAPPS and clinical expert opinion consulted by the sponsor.20 Clinical experts consulted by CDA-AMC note that several of the drugs in the list of BAC do not align with Canadian clinical practice. For instance, oral corticosteroids and ibuprofen would not be considered GPP treatments.
CDA-AMC did not address this limitation in the BIA. It is expected to have minimal impact on the results of the BIA.
CDA-AMC Reanalyses of the BIA
CDA-AMC revised the sponsor’s submitted analysis by removing the incident rate, including the Canadian annual growth rate, revising the prevalence rate, and revising the acute setting market share of spesolimab (Table 17).
Table 17: CDA-AMC Revisions to the Submitted Budget Impact Analysis
Stepped analysis | Sponsor’s value or assumption | CDA-AMC value or assumption |
|---|---|---|
Changes to derive the CDA-AMC base case | ||
1. Combining incidence and prevalence rates | Included the incidence of GPP (1.95 per million) in addition to the prevalent population | Applied a 1.8% population growth rate to the Canadian population and applied the prevalence rate to the total population each year |
2. Prevalence rate | 2.77 per million | 5.44 per million |
3. Flare treatment market uptake of spesolimab | ██%, ██%, and ██% in years 1, 2, and 3, respectively | 85%, 90%, and 95% in years 1, 2, and 3, respectively |
4. Mark-ups and dispensing fees | Included | Excluded |
CDA-AMC base case | Reanalysis 1 + 2 + 3 + 4 | |
CDA-AMC = Canada’s Drug Agency.
The results of the CDA-AMC stepwise reanalysis are presented in summary format in Table 18 and a more detailed breakdown is presented in Table 19.
The CDA-AMC reanalysis suggests that reimbursing spesolimab would be associated with an incremental cost of $560,297 in Year 1, $1,594,793 in year 2, and $2,620,204 in year 3, for a 3-year budgetary impact of $4,775,294. In the CDA-AMC reanalysis, there were a total of 64 patients eligible for treatment with spesolimab in year 3, compared with 97 patients in the sponsor’s analysis.
Table 18: Summary of the CDA-AMC Reanalyses of the Budget Impact Analysis
Stepped analysis | Three-year total ($) |
|---|---|
Submitted base case | 6,544,058 |
CDA-AMC reanalysis 1 | 2,497,450 |
CDA-AMC reanalysis 2 | 8,847,422 |
CDA-AMC reanalysis 3 | 6,598,259 |
CDA-AMC reanalysis 4 | 6,302,377 |
CDA-AMC base case | 4,775,294 |
CDA-AMC = Canada’s Drug Agency.
CDA-AMC conducted the following scenario analyses to address remaining uncertainty, using the CDA-AMC base case (results are provided in Table 19):
Assuming the public drug coverage rate is 64% for patients less than 65 years old to account for coverage under other public funding programs.
Assuming market uptake for spesolimab in the preventive setting is 20%, 50%, and 55% in years 1, 2, and 3, respectively.
Assuming treatment of flares with a GPPGA total score of 2 or greater, as per the reimbursement requested population.
Assuming that the price of spesolimab is reduced by 79% (CDA-AMC’s estimated price reduction from the cost-utility analysis).
Results of CDA-AMC’s scenario analysis demonstrate that the estimated budget impact is sensitive to changes in coverage rate for those under 65 years of age and preventive setting market share of spesolimab. The scenario analysis with increased coverage rate for those under 65 years of age estimated a 3-year budget impact of $8,292,185, a 74% increase from the CDA-AMC base case. The scenario that assumed a higher market uptake of spesolimab in the preventive setting estimated the 3-year budget impact of 6,501,317, a 36% increase from the CDA-AMC base case. This is likely because 98% of the incremental budget impact is associated with costs from the preventive setting; hence, a moderate increase in spesolimab market share in the preventive setting would have a significant impact on the estimated budget impact.
Table 19: Detailed Breakdown of the CDA-AMC Reanalyses of the BIA
Stepped analysis | Scenario | Year 0 (current situation) ($) | Year 1 ($) | Year 2 ($) | Year 3 ($) | Three-year total ($) |
|---|---|---|---|---|---|---|
Submitted base case | Reference | 621,610 | 1,059,206 | 1,496,802 | 1,934,397 | 4,490,405 |
New drug | 621,610 | 1,534,087 | 3,450,353 | 6,050,023 | 11,034,463 | |
Budget impact | 0 | 474,881 | 1,953,552 | 4,115,625 | 6,544,058 | |
CDA-AMC base case | Reference | 1,152,154 | 1,172,892 | 1,194,004 | 1,215,496 | 3,582,393 |
New drug | 1,152,154 | 1,733,190 | 2,788,797 | 3,835,700 | 8,357,687 | |
Budget impact | 0 | 560,297 | 1,594,793 | 2,620,204 | 4,775,294 | |
CDA-AMC scenario analysis 1: Public drug coverage | Reference | 1,972,220 | 2,007,720 | 2,043,858 | 2,080,648 | 6,132,226 |
New drug | 1,972,220 | 2,981,359 | 4,813,110 | 6,629,942 | 14,424,411 | |
Budget impact | 0 | 973,640 | 2,769,251 | 4,549,294 | 8,292,185 | |
CDA-AMC scenario analysis 2: Preventive setting market uptake | Reference | 1,152,154 | 1,172,892 | 1,194,004 | 1,215,496 | 3,582,393 |
New drug | 1,152,154 | 2,257,545 | 3,811,055 | 4,015,111 | 10,083,710 | |
Budget impact | 0 | 1,084,653 | 2,617,051 | 2,799,614 | 6,501,317 | |
CDA-AMC scenario analysis 3: Reimbursement- requested population | Reference | 1,139,023 | 1,159,525 | 1,180,396 | 1,201,644 | 3,541,565 |
New drug | 1,139,023 | 1,712,993 | 2,767,828 | 3,813,937 | 8,294,759 | |
Budget impact | 0 | 553,468 | 1,587,432 | 2,612,294 | 4,753,194 | |
CDA-AMC scenario analysis 4: 79% price reduction | Reference | 1,152,154 | 1,172,892 | 1,194,004 | 1,215,496 | 3,582,393 |
New drug | 1,152,154 | 1,231,849 | 1,293,512 | 1,347,305 | 3,872,666 | |
Budget impact | 0 | 58,956 | 99,507 | 131,809 | 290,272 |
CDA-AMC = Canada’s Drug Agency; BAC = best available care; BIA = budget impact analysis.
Ethics Review
Abbreviations
GPP
generalized pustular psoriasis
IL
interleukin
SC
subcutaneous
Ethical Considerations
Generalized pustular psoriasis (GPP) is a rare, severe, chronic skin disease. It is characterized by recurrent episodes (GPP flares) involving the widespread eruption of sterile, macroscopically visible pustules, which may occur with or without systemic inflammation.1 If left untreated, GPP flares can be life-threatening due to the risk of severe complications such as renal failure, heart failure, and sepsis.2 While GPP can present with skin manifestations similar to psoriasis vulgaris (e.g., painful erythema, scaling), it is distinguished by a unique pathophysiology that involves immune dysregulation leading to the activation of immune cells around abnormalities in the interleukin-36 (IL-36) pathway.1
This brief report is informed by the sponsor’s submission and patient group, clinician group, and drug plan input received by CDA-AMC for this review; direct consultation with 3 clinical experts (dermatologists) who have experience treating patients with GPP in Canada (2 of these experts have direct experience using spesolimab); and engagement with the clinical and economic reviewers.
This brief report highlights ethical considerations regarding the use of spesolimab for the treatment of GPP in adults and pediatric patients aged 12 years and older. Spesolimab is a monoclonal antibody administered as a single-dose IV infusion for the treatment of acute GPP flares and as a subcutaneous (SC) injection every 4 weeks for the prevention of flares. Spesolimab targets abnormalities in the IL-36 pathway that contribute to GPP’s distinct pathophysiology and, as such, is the first treatment option indicated specifically for GPP. This report outlines considerations relevant for decision-making regarding the public reimbursement and implementation of spesolimab in Canada; however, it does not necessarily present an exhaustive list of all ethical considerations associated with GPP and its treatment.
Diagnosis, Treatment, and Experiences of People Living With GPP
Living with GPP presents significant physical and psychosocial burdens for patients and their caregivers. During a severe GPP flare, for instance, patients are at an elevated risk of mortality (2% to 16%)2 due to the systemic impacts on cardiac, lung, and renal function. Beyond this, the clinical experts and the clinician group and patient group input all highlighted how recurrent GPP flares can involve the spontaneous, rapid onset of painful inflammatory pustules, diffuse erythema, and pruritus. GPP flares can also be highly disruptive to people’s lives and daily activities. Patients may need to be hospitalized or may become bedridden and be unable to work, participate in social activities, or maintain physical intimacy. The patient group input and the clinical experts described long-term impacts of GPP on mental health and well-being, including experiences of diminished self-esteem and depression and anxiety between flares.
The clinical experts indicated that people experiencing their first GPP flare would likely present to their local emergency department for assessment and diagnosis by a medical dermatologist (if available). However, the patient group input noted that some individuals may experience delays in receiving an accurate diagnosis, at times requiring multiple hospital visits and self-advocacy with their family doctors to obtain a referral to a specialist familiar with GPP. While delays in diagnosis are uncommon, GPP is a very rare skin condition with little public awareness. As a result, some people may experience a delayed diagnosis and thereby delayed access to appropriate care and treatment.
The clinical experts described how the timely diagnosis and treatment of GPP (whether for an acute flare or long-term flare prevention) could be further hampered by growing gaps in access to publicly funded dermatology services across Canada. This may be exacerbated for people with GPP living in rural or remote areas where specialized dermatology services are more limited. While telehealth services could help bridge some of these gaps by connecting family doctors and emergency departments with specialists, the limited geographic availability of hospitals with dermatology specialists and the infusion centres needed to treat acute flares and/or provide preventive therapy with current treatment options remains a challenge and leads to ongoing disparities in access to appropriate dermatological care. The patient group input indicated that, for this reason, having a treatment option that limited the need to travel would be ideal.
There is currently no targeted treatment option indicated for the treatment of acute GPP flares or the long-term management and prevention of flares. Instead, in cases of severe GPP flares, providers use a variety of off-label, fast-acting biologics (i.e., IL-17 and tumour necrosis factor alpha inhibitors) indicated for plaque psoriasis. Similarly, other off-label psoriasis biologics (e.g., IL-23 and IL-12 or IL-23 inhibitors) and nonbiologic systemic therapies are used to support long-term management and flare prevention. The absence of targeted therapy is further complicated by the lack of consensus guidelines regarding the treatment of GPP flares or long-term management of GPP in Canada. As such, the clinical experts and clinician group input indicated that current practice is inadequate due to the inconsistent efficacy of off-label treatment options in GPP, their contraindication in some patients (e.g., individuals who are pregnant), and variability in access to these therapies across jurisdictions. Further, given the absence of treatment options indicated and reimbursed specifically for GPP, the clinical experts highlighted experiences of moral distress in having to misrepresent patients’ diagnoses to gain access to off-label treatments indicated for psoriasis.
The clinical experts and the patient group and clinician group input all indicated that the treatment goals for GPP include the rapid control of acute flares that not only alleviates symptoms quickly but also mitigates the potential for long-term systemic damage or mortality due to prolonged flares. Additionally, all expressed an interest in preventive treatment options that could reduce the frequency and severity of GPP flares and improve the overall quality of life for people with GPP.
Clinical Evidence Used in the Evaluation of Spesolimab
Spesolimab was evaluated in the 2 randomized, placebo-controlled, double-blind, phase II Effisayil 1 (N = 53) and phase IIb Effisayil 2 (N = 123) trials. The Effisayil 1 trial evaluated the safety, efficacy, and tolerability of a single-dose IV infusion of spesolimab for acute GPP flares of moderate to severe intensity in adults aged 18 to 75. The trial results suggest that patients receiving spesolimab likely experienced better outcomes for the primary end point (resolution of acute GPP flares with no visible pustules after 1 week of treatment) when compared with placebo. The Effisayil 2 trial evaluated the safety and efficacy of spesolimab SC injections for the prevention of GPP flares in patients aged 12 to 75 with a history of GPP. As with the Effisayil 1 study, the Effisayil 2 trial results suggest that people treated with spesolimab SC likely experienced better outcomes for the primary end point (time to first GPP flare up to week 48) when compared with placebo. Experiences of adverse events (e.g., pustular psoriasis, fever, infection) and serious adverse events (e.g., pustular psoriasis) were high across both the active treatment and placebo arms of the Effisayil 1 and Effisayil 2 trials. However, the clinical experts indicated that no new safety concerns were identified. Also, they considered the safety profile manageable, given the potentially life-threatening nature of severe GPP flares.
The long-term extension study for the Effisayil 1 and Effisayil 2 trials (Effisayil ON) is ongoing, with limited information available during this review. Although the clinical experts indicated that the placebo comparator was warranted due to the absence of other treatment options specific to GPP, the long-term efficacy and harms of spesolimab relative to any comparator (including commonly used off-label therapies) are presently unknown. The lack of long-term evidence for safety and efficacy, as well as comparative effectiveness, highlights the importance of robust consent conversations and presents challenges for clinical and health system decision-making, including consideration of opportunity costs.
The clinical experts indicated that the trial populations were broadly generalizable to those seen in practice. However, they also suggested that the Effisayil 1 trial excluded patients who may have been more likely to experience drug-induced side effects, particularly patients living with hepatic disease. Though acknowledging the importance of narrow inclusion and exclusion criteria in trial settings, 1 clinical expert suggested that in the event of a severe, threatening GPP flare, providers would consider whether to prescribe spesolimab on a case-by-case risk-benefit assessment, especially as other off-label biologics may be similarly hepatotoxic. Similarly, while the product monograph notes that spesolimab IV and spesolimab SC are indicated for the treatment of GPP in patients aged 12 years and older, the Effisayil 1 trial did not include patients aged 12 to 17 years; thus, there is no clinical evidence regarding the efficacy and safety of spesolimab IV in this population. However, the clinical experts were comfortable with using it despite this absence and noted that existing evidence on the use of biologics for plaque psoriasis in pediatric patients suggests there is no clinically meaningful difference in safety between adolescents and adults. Regardless, they added that it is important to collect more real-world data in the populations excluded or absent from the trials to support future clinical decision-making. Registry data on people who were pregnant were mentioned as of particular interest. In the absence of evidence regarding the efficacy and safety of spesolimab IV in these populations, it will be important for clinical providers to facilitate consent conversations that transparently recognize the absence of data.
Clinical Use of Spesolimab
The clinical experts considered spesolimab to be a potentially paradigm-shifting treatment in the care of people with GPP due to its unique mechanism of action focused on the IL-36 signalling pathway involved in GPP pathogenesis. As a targeted therapy that may alleviate some challenges associated with current off-label treatment options (e.g., their varying efficacy, inconsistent jurisdictional availability, and contraindication in some patients), the clinical experts uniformly expressed a willingness to prescribe spesolimab IV as a first-line treatment for patients experiencing acute GPP flares and spesolimab SC as a first-line option for the long-term prevention of GPP flares. They described their personal experiences observing rapid resolution of GPP flares with spesolimab in their own patients as supporting this decision and highlighted their satisfaction with the efficacy and safety results of the Effisayil 1 and Effisayil 2 studies. The clinical experts believed it would be inappropriate to require patients to be unsuccessful in other, off-label options before accessing spesolimab for these reasons and because it would unnecessarily expose patients to an elevated risk of mortality. However, for patients who were already well managed with off-label biologics, the clinical experts indicated they would only consider shifting to spesolimab SC for long-term maintenance following treatment of an acute flare with spesolimab IV.
As pregnancy is a known trigger for GPP flares, some people with GPP require treatment for acute flares during pregnancy. While the product monograph has indicated that the use of spesolimab (IV or SC) should be avoided in individuals who are pregnant, the clinical experts indicated that this guidance may not be followed in practice. Instead, they suggested that decisions to use spesolimab in individuals who are pregnant would be contextual and assessed on a case-by-case basis according to a patient’s specific risk benefit, especially given the potentially life-threatening nature of acute flares. This raises an ethical consideration in that there is currently no evidence for the use of spesolimab in people who are pregnant. Additionally, there is some risk that spesolimab could cross the placental barrier as a monoclonal antibody and affect the unborn fetus. However, the clinical experts noted that this risk was not unique to spesolimab. For example, other biologics currently used off-label to control GPP flares were likely to have similar safety profiles in people who are pregnant, and some systemic therapies are contraindicated in individuals who are pregnant. As the sole treatment option specifically targeting the causal pathway of GPP, the clinical experts consulted assumed that spesolimab would have the best risk benefit for people with GPP. Regardless, the experts highlighted the importance of having clear conversations with patients who are pregnant that could help them weigh the potential risks and benefits of proceeding with spesolimab in the event of an acute flare.
Health Systems Impact
The clinical experts and clinician group input both suggested that the public reimbursement of spesolimab may alter or limit the utilization of some of the health care resources associated with long-term flare prevention and treatment of acute GPP flares. However, there is presently no evidence demonstrating these impacts. This raises ethical considerations for health care planning and resource allocation, including how to fairly distribute or share the potential risks and benefits associated with reimbursing a therapy where the long-term value is currently unknown. Nonetheless, the clinical experts suggested that the reimbursement of spesolimab SC for long-term flare prevention may lead to a decreased reliance on the use of off-label biologics and systemic medications. Similarly, they expected that the preventive use of spesolimab SC may lessen hospital admissions and reduce the use of associated health care resources allocated to treating GPP flares. In the event of an acute flare, the clinical experts suggested that spesolimab IV for treatment of acute flares delivered in outpatient settings or emergency departments may limit the need for admission to a hospital or intensive care unit. This could have an overall benefit for health care resource utilization.
The fragmentation of the health care system can present challenges for reimbursement and equitable access to spesolimab. For example, spesolimab may be funded through different budget streams: hospital budgets for the treatment of acute flares with spesolimab IV in hospital, and provincial formularies for both the treatment of acute flares with spesolimab IV in outpatient infusion centres and for long-term prevention using spesolimab SC for self-administration. The clinical experts suggested this could lead to logistical challenges and potential gaps in coverage and inequities in financial support for patients. For example, if spesolimab IV is only covered through hospital budgets, people living in rural or remote locations with limited access to hospital services may not be able to access spesolimab IV or SC if they do not have private insurance due to a lack of public coverage.
The clinical experts indicated that some people with GPP may be living in locations with limited to no access to infusion centres, which are necessary for outpatient delivery of spesolimab IV for acute flares. As such, the option to self-administer spesolimab at home as an SC injection may be considered equity-enhancing for patients living in rural or remote locations with limited access to infusion.
The sponsor has indicated that the presence of a patient support program to aid in the implementation of spesolimab in Canada (e.g., outpatient administration at infusion centres and support for navigating reimbursement opportunities). While the sponsor has indicated providing compassionate and “free goods (i.e., medication at no charge to the patient)” to patients unable to afford spesolimab IV, it is unclear whether these supports include travel supports for patients residing far from infusion centres, a noted challenge to the equitable provision of timely care for people with GPP. The drug program input indicated the need to further clarify the parameters of this program with the sponsor should spesolimab be recommended for reimbursement.
References
1.Navarini AA, Burden AD, Capon F, et al. European consensus statement on phenotypes of pustular psoriasis. J Eur Acad Dermatol Venereol. 2017;31(11):1792-1799. PubMed
2.Choon SE, Navarini AA, Pinter A. Clinical Course and Characteristics of Generalized Pustular Psoriasis. Am J Clin Dermatol. 2022;23(Suppl 1):21-29. PubMed
ISSN: 2563-6596
Canada’s Drug Agency (CDA-AMC) is a pan-Canadian health organization. Created and funded by Canada’s federal, provincial, and territorial governments, we’re responsible for driving better coordination, alignment, and public value within Canada’s drug and health technology landscape. We provide Canada’s health system leaders with independent evidence and advice so they can make informed drug, health technology, and health system decisions, and we collaborate with national and international partners to enhance our collective impact.
Disclaimer: CDA-AMC has taken care to ensure that the information in this document was accurate, complete, and up to date when it was published, but does not make any guarantee to that effect. Your use of this information is subject to this disclaimer and the Terms of Use at cda-amc.ca.
The information in this document is made available for informational and educational purposes only and should not be used as a substitute for professional medical advice, the application of clinical judgment in respect of the care of a particular patient, or other professional judgments in any decision-making process. You assume full responsibility for the use of the information and rely on it at your own risk.
CDA-AMC does not endorse any information, drugs, therapies, treatments, products, processes, or services. The views and opinions of third parties published in this document do not necessarily reflect those of CDA-AMC. The copyright and other intellectual property rights in this document are owned by the Canadian Agency for Drugs and Technologies in Health (operating as CDA-AMC) and its licensors.
Questions or requests for information about this report can be directed to Requests@CDA-AMC.ca.