Drugs, Health Technologies, Health Systems
Reimbursement Review
Ferric Carboxymaltose (Ferinject)
Sponsor: CSL Vifor
Therapeutic area: Iron deficiency anemia
This multi-part report includes:
Clinical Review
Pharmacoeconomic Review
Clinical Review
Abbreviations
AE
adverse event
ANCOVA
analysis of covariance
BMI
body mass index
CDA-AMC
Canada’s Drug Agency
CHF
chronic heart failure
CI
confidence interval
CKD
chronic kidney disease
CrI
credible interval
DIC
deviance information criterion
ESA
erythropoiesis-stimulating agent
FACIT-F
Functional Assessment of Chronic Illness Therapy–Fatigue
FAS
full analysis set
FWG
Formulary Working Group
GI
gastrointestinal
GRADE
Grading of Recommendations Assessment, Development and Evaluation
HRQoL
health-related quality of life
HUB
heavy uterine bleeding
IBD
inflammatory bowel disease
IBDQ
Inflammatory Bowel Disease Questionnaire
IDA
iron deficiency anemia
ITT
intention to treat
LOCF
last observation carried forward
mITT
modified intention to treat
MMRM
mixed model for repeated measures
NMA
network meta-analysis
NR
not reported
OR
odds ratio
PCS
potentially clinically significant
RCT
randomized controlled trial
SAE
serious adverse event
SD
standard deviation
SE
standard error
SF-36
Short-Form (36) Health Survey
SOC
standard of care
TEAE
treatment-emergent adverse event
TOI
time of intervention
TSAT
transferrin saturation
Executive Summary
An overview of the submission details for the drug under review is provided in Table 1.
Table 1: Background Information of Application Submitted for Review
Item | Description |
|---|---|
Drug product | Ferric carboxymaltose (Ferinject), 50 mg elemental iron per mL, vial for injection (2 mL, 10 mL, 20 mL) |
Sponsor | CSL Vifor |
Indication | For the treatment of iron deficiency anemia in adult and pediatric patients 1 year of age and older when oral iron preparations are not tolerated or are ineffective. The diagnosis of iron deficiency must be based on laboratory tests. |
Reimbursement request | As per indication |
Health Canada approval status | NOC |
Health Canada review pathway | Standard |
NOC date | March 11, 2024 |
Recommended dose | The iron dose needed for repletion using ferric carboxymaltose is determined based on the patient’s body weight and hemoglobin level. The maximum recommended cumulative dose is 1,000 mg of iron (20 mL of ferric carboxymaltose) per week. If the total iron need is higher, then the administration of an additional dose should be a minimum of 7 days from the first dose. A single administration of ferric carboxymaltose should not exceed either 15 mg iron/kg body weight, or 1,000 mg iron (20 mL of ferric carboxymaltose). Reassessment should be performed by the clinician based on the individual patient’s condition. The hemoglobin level should be reassessed no earlier than 4 weeks post final ferric carboxymaltose administration to allow adequate time for erythropoiesis and iron utilization. In the event the patient requires further iron repletion, the need for iron should be recalculated. In adult patients with hemodialysis-dependent chronic kidney disease, a single maximum daily dose of 200 mg iron as ferric carboxymaltose should not be exceeded. |
NOC = Notice of Compliance.
Introduction
Anemia is a medical condition in which the blood has reduced capacity to carry oxygen because of low hemoglobin levels. Although anemia has a range of causes, iron deficiency is the most prevalent.1 Iron deficiency anemia (IDA) results when body iron levels are insufficient to sustain hemoglobin synthesis.2 Alberta clinical practice guidelines describe IDA as “a serious condition whereby low levels of iron are associated with anemia and the presence of microcytic hypochromic red cells.”3 British Columbia clinical guidelines define anemia based on hemoglobin levels relative to the population mean: “[a] low hemoglobin level, most frequently defined as a hemoglobin value over 2 standard deviations below the gender- and age-adjusted mean. A hemoglobin value below the local, lab-specific lower reference interval indicates anemia.”4
In a Canadian Health Measures Survey study, approximately 2% of the population in Canada was identified as having IDA, with the prevalence in females approximately 9 times higher than that of males. In a recent study by Cooper et al., IDA prevalence in the total population, corrected for inflammation ranged from 2% to 2.5%. Notably, less than 1% of males exhibited IDA regardless of the correction method, whereas this figure rose to approximately 4% for all females. For females of child-bearing age, the corrected IDA prevalence ranged between 5% and 6.2%.5 Iron deficiency and IDA during the prenatal period have been associated with adverse effects on neonatal growth, neurocognitive development, and perinatal morbidity and mortality. A 2022 study assessed iron status during early and late pregnancy among 60 pregnant women receiving 27 mg/day of elemental iron as part of a randomized trial.6 High rates of iron deficiency were observed: 28% of participants exhibited probable iron deficiency at baseline (mean gestational age = 16 weeks), with the proportion increasing to 81% by study end (mean gestational age = 32 weeks).6 This deterioration in iron status occurred despite widespread supplementation and highlights a potential need for additional iron beyond the recommended dose to support needs during later pregnancy. While the general Canadian population maintains a relatively low prevalence of anemia (approximately 3%), this is not the case among Canada's Indigenous population. In a 2017 study of the Inuit population in Nunavik (aged 16 years and older), the prevalence of anemia in women of child-bearing age was 20%, with 14% of women experiencing IDA and 23% exhibiting iron deficiency without anemia.7 Information for other groups of Indigenous Peoples were not available.
Anemia has a range of causes, so a diagnosis of IDA is based on identification of both anemia and iron deficiency. IDA is diagnosed based on blood tests showing low hemoglobin levels coupled with markers for iron deficiency, typically ferritin levels and transferrin saturation (TSAT).
The key goals in the treatment of iron deficiency and IDA are the correction of the hemoglobin deficit and repletion of iron stores (the correction phase), and maintenance of iron levels over time (the maintenance phase).2,8 Oral iron, often in the form of iron salts (e.g., ferrous gluconate, ferrous sulphate, or ferrous fumarate), is the first-line therapy for most cases of iron deficiency and IDA, and is relatively safe, effective, and inexpensive. Some patients may be unable to absorb ferrous sulphate adequately because of impaired intestinal uptake resulting from gastrointestinal (GI) disease or clinical conditions such as chronic inflammation, which may in turn lead to elevated levels of hepcidin.9-11 In other instances, the iron deficit becomes so profound that severe symptomatic anemia results and transfusion of red blood cells may be required.9,12 Oral iron supplements carry the additional burden of GI side effects, which can have a detrimental impact on patient adherence. There is also a need to deliver iron rapidly in certain clinical situations, including, for example, when a patient with iron deficiency requires an urgent surgery with risk of blood loss.13 IV iron treatment may also be preferred over oral iron in some situations, such as for chemotherapy-induced anemia14-16 and in patients with chronic kidney disease (CKD), with or without concomitant erythropoiesis-stimulating treatment.17-20 In children, IV iron substitution may be necessary when oral iron therapy is not possible, ineffective, or poorly tolerated. According to Mattiello et al.,21 a switch to IV iron is proposed for cases with severe anemia (hemoglobin < 70 g/L) to avoid transfusion, for patients with an underlying secondary disease fulfilling a formal indication for IV iron (e.g., IBD, chronic GI or genitourinary bleeding, or celiac disease), and/or for situations of nonadherence to oral treatment and symptomatic refractory IDA with a clinical impact.21
The objective of this Clinical Review is to review and critically appraise the clinical evidence submitted by the sponsor on the beneficial and harmful effects of ferric carboxymaltose 50 mg/mL by IV infusion in the treatment of IDA in adult and pediatric patients aged 1 year and older when oral iron preparations are not tolerated or are ineffective.
Perspectives of Patients, Clinicians, and Drug Programs
The information in this section is a summary of input provided by the patient and clinician groups who responded to a call for input and from a clinical expert consulted by Canada’s Drug Agency (CDA-AMC) for the purpose of this review.
Patient Input
One patient group, the Gastrointestinal Society, provided input to this submission. The group obtained input through discussions with health care professionals and researchers, and as well as surveys and interviews conducted on digestive and liver diseases, although the precise number of respondents was not reported.
The GI Society describes dizziness, extreme fatigue, headache, shortness of breath, tiredness, and muscle weakness as common symptoms of IDA. IDA can have significant impact on a patient, physically, mentally, financially, and socially, and they emphasized the extreme fatigue that can lead to social isolation.
The GI Society emphasized the tolerability issues associated with oral iron products, and the infusion-related side effects associated with parenteral iron as limitations of treatment. Other limitations include the practical issues associated with having to take time off work and obtain transport to attend infusions.
Clinician Input
Input From Clinical Experts Consulted by CDA-AMC
The clinical expert consulted for this review noted the key limitations of orally administered iron, namely tolerability, adherence, and delayed onset of effect. The expert noted the importance of having additional options for parenteral iron, as some patients have tolerability issues. The expert also noted that, in many cases, ferric carboxymaltose may be seen as simply another option among several formulations of IV iron.
The patients most suited for ferric carboxymaltose would be adults with iron deficiency or IDA, those unable to tolerate or respond to oral iron, and preoperative patients with IDA according to the clinical expert; those least suitable would be patients with a history of hypophosphatemia. The expert did not believe there were any issues with diagnosing IDA, with ferritin and TSAT being key tests of iron stores.
The clinical expert noted that the key outcomes for assessing response include a rise in hemoglobin, improvement in symptoms of iron deficiency and IDA, improved function, and reduced transfusion burden. The clinical expert believed that 4 weeks would be the minimum follow-up for assessing the efficacy of IV iron supplementation, and 12 weeks would be ideal.
Clinician Group Input
A group of 12 clinicians, including those with specialties in cardiology, obstetrics and gynecology, hematology, gastroenterology, and nephrology, as well as a primary care physician, provided input.
The clinician group was in general agreement with the input provided by the clinical expert.
The clinician group was in agreement with the clinical expert that important unmet needs with IV iron supplementation include the need to reduce visits to hospitals and infusion clinics and to have a product available that has evidence supporting its use during pregnancy.
The clinician group did not elaborate on its experience with ferric carboxymaltose, but did note that it has been available in Europe for 20 years.
Drug Program Input
The Formulary Working Group (FWG) asked whether there are specific lab thresholds that should be considered for initiation of oral iron, and the clinical expert responded that, in patients with non-CKD, non–chronic heart failure (CHF), non-oncology, the definition of IDA is hemoglobin less than 130 g/L (for males) or less than 120 g/L (for females) and ferritin less than 30 mcg/L. Values for hemoglobin also vary in pregnancy (first and third trimester: < 110 g/L; second trimester: < 105 g/L) and in children (< 24 months: < 105 g/L; 24 to 59 months: < 110 g/L; 5 to 11 years: < 115 g/L). The clinical expert also provided specific cutoffs for ferritin and TSAT based on various conditions such as inflammation (TSAT less than 20% and ferritin less than 100 mcg/L),CKD (TSAT less than 30% and ferritin less than 500 mcg/L), CHF(ferritin less than 100 mcg/L or ferritin less than 300 mcg/L with TSAT less than 20%); and dialysis (ferritin less than 200 mcg/L or TSAT less than 20%).
The FWG asked how an inadequate response to oral iron is defined and the appropriate length of follow-up before making that assessment. The clinical expert stated that failure to normalize iron stores within 3 months or failure to increase hemoglobin by 10 g/L within 4 weeks indicated an inadequate response. In response to whether there are populations in which oral iron is inappropriate, the clinical expert identified those with bariatric surgery, gastrectomy, IBD, CKD, prior small bowel resection, preoperative IDA (4 weeks or less to surgery), or profound IDA (e.g., symptomatic) and patients with a history of heart failure.
The FWG asked what the benefits of ferric carboxymaltose are over alternatives such as iron sucrose in patients receiving hemodialysis. The clinical expert responded that, other than a higher dose of iron infused at the session during which it is administered, there is no major benefit in patients coming regularly for dialysis. In response to an FWG question about how many patients would require more than 1 infusion, the clinical expert responded that, at any given time, a minority of patients will receive more than 1 g (1,000 mg), although patients frequently require longstanding treatment (i.e., they need IV iron every 3 months) if they have ongoing risk factors for iron deficiency (e.g., heavy menstrual bleeding or malabsorption).
Clinical Evidence
The sponsor conducted a systematic search-and-selection procedure to identify studies relevant to the efficacy and harms of ferric carboxymaltose relative to relevant comparators in the Canadian setting. The sponsor identified 60 reports of 40 clinical trials, including studies among patients with iron deficiency and heart failure. However, heart failure is outside the scope of this report. The current report focuses only on select studies of IDA identified in the sponsor’s systematic review, with detailed summaries and appraisals of these studies. High-level characteristics and results of the supportive studies submitted to Health Canada as well as studies of the use of ferric carboxymaltose during pregnancy are provided in Appendix 1.
A summary of the clinical evidence included by the sponsor in the review of ferric carboxymaltose is presented in 3 sections. To inform the broad indication under review, the first section, Pivotal Studies and Select Randomized Controlled Trials, includes the pivotal studies submitted to Health Canada for the indication under review and other RCTs from the sponsor’s systematic review that enrolled patients with IDA arising from various causes. The second section includes indirect evidence from the sponsor. The third section summarizes studies addressing gaps and includes 1 study of pediatric patients to inform the use of ferric carboxymaltose among children and adolescents, 1 study among adult patients with IDA arising from various causes to provide additional evidence for the broad indication, and 5 studies comparing the efficacy of ferric carboxymaltose and ferric derisomaltose.
Pivotal Studies and Select Randomized Controlled Trials
Description of Studies
The systematic review portion of this report focuses on 4 studies: the 2 pivotal phase III RCTs per the Health Canada review (VIT-IV-CL-015 and FERGIcor) and 2 phase III RCTs (1VIT05006 and VIT-IRON-2011-004) selected from the sponsor’s submitted systematic review. The pivotal trials focused on specific populations: patients with CKD (VIT-IV-CL-015, N = 240) and those with IBD (FERGIcor, N = 304), with each study randomizing patients 1:1 to either ferric carboxymaltose or iron sucrose. Study 1VIT05006 and Study VIT-IRON-2011-004 included a more heterogeneous IDA population (i.e., patients had IDA arising from various causes). Study 1VIT05006 (N = 559) was a placebo-controlled trial with a crossover design and Study VIT-IRON-2011-004 (N = 371) was a noninferiority study that randomized patients 1:1 to either ferric carboxymaltose or iron sucrose. The primary outcome in 3 of the trials was to assess hemoglobin response, as an increase from baseline. The 1VIT05006 trial was not designed to assess efficacy outcomes.
In the FERGIcor study, the mean age was 52.7 years (standard deviation [SD] = 13.8 years) in the ferric carboxymaltose group and 51.0 years (SD = 12.6 years) in the iron sucrose group, while in VIT-IV-CL-015 and the other studies the mean age was approximately 40 years. In Study VIT-IV-CL-015, approximately 42% of patients were female and in the FERGIcor study 59% were female. In the nonpivotal trials enrolling patients with IDA arising from various causes, approximately 90% of the patients were female. The overall mean hemoglobin level was 93.2 g/L in Study VIT-IV-CL-015, 102.2 g/dL in the FERGIcor study, █████ ███ in Study 1VIT05006, and less than 80 g/L in Study VIT-IRON-2011-004.
Efficacy Results
Hospitalizations (All-Cause and Anemia-Related)
This outcome was not assessed in any of the 4 trials that were the focus of this systematic review section.
Hemoglobin
In the VIT-IV-CL-015 study, the primary response rate was defined as the percentage of patients reaching an increase in hemoglobin of 10 g/L or greater at 4 weeks after baseline. The percentage of patients with a response in the ferric carboxymaltose group was 46.4% (45 of 97), and in the iron sucrose group it was 37.2% (32 of 86), with a reported P value for the between-group difference of 0.2101. The mean increase in hemoglobin levels in the ferric carboxymaltose group at week 4 was ███ ███████ ████ ███ ███ and in the iron sucrose group ███ ███████ ████ ███ ███ with a reported P value for the between-group difference of ████████. No between-group difference and confidence interval (CI) was reported for either end point.
In the FERGIcor study, the primary outcome was patients with a response of an increase in hemoglobin of at least 20 g/L at week 12. In the full analysis set (FAS), the percentage patients with IDA who responded to treatment was 65.79% (150 of 240) in the ferric carboxymaltose group and 53.64% (118 of 220) in the iron sucrose group (Table 17). The between-group difference was 12.15% higher (95% CI, 3.07% to 20.97%; P = 0.004) in the ferric carboxymaltose group compared with that in the iron sucrose group. The FAS set was also analyzed using the last observation carried forward (LOCF) and the worst-case method and the results were consistent with the primary analysis.
In the VIT-IRON-2011-004 study, 99.5% of patients (184 of 185) in each of the ferric carboxymaltose and 98.3% (177 of 180) in the iron sucrose group achieved an increase in hemoglobin of at least 20 g/L from baseline to week 8, for a between-group difference of 1.13 (95% CI, −2.02 to 4.68) (Table 17). The criteria for noninferiority was met. From mean baselines of 77.4 g/L (SD = 14.95 g/L) in the ferric carboxymaltose group and 80.5 g/L (SD = 14.45 g/L) in the iron sucrose group, the mean change from baseline to week 8 was ████ ███ ███████ in the ferric carboxymaltose group and ████ ███ ███████ in the iron sucrose group. The between-group difference was ████ ███ ████ ███ █████ ██ ██████ ███████.
Patient-Reported Health-Related Quality of Life
In the FERGIcor study, for the physical component of the SF-36, from a mean baseline of 44.17 (SD = 7.36) in the ferric carboxymaltose group and 44.98 (SD = 7.23) in the iron sucrose group, there were changes from baseline to week 12 of 3.88 (SD = 6.77) and 2.64 (SD = 7.14), respectively, for a reported between-group P value of 0.157. For the mental component of the SF-36, from mean baselines of 40.02 (SD = 11.04) in the ferric carboxymaltose group and 41.30 (SD = 11.70) in the iron sucrose group, there were changes from baseline to week 12 of 5.91 (SD = 10.74) and 5.56 (SD = 10.36), respectively, for a between-group P value of 0.583. For the Inflammatory Bowel Disease Questionnaire (IBDQ) total score, from mean baselines of 150.8 (SD = 35.2) in the ferric carboxymaltose group and 152.7 (SD = 34.4) in the iron sucrose group, there were changes from baseline to week 12 of 21.1 (SD = 32.3) and 19.7 (SD = 28.8), respectively, for a between-group P value of 0.872. Between-group differences with CIs were not reported for any of the health-related quality of life (HRQoL) measures.
HRQoL was not assessed in VIT-IV-CL-015 or VIT-IRON-2011-004 studies.
Serum Ferritin
In the VIT-IV-CL-015 study, the mean baseline for serum ferritin was ████ █████ ███████ ██ ████ in the ferric carboxymaltose group and ████ █████ ███████ ██ ████ in the iron sucrose group. After week 4, there were changes from baseline of █████ █████ ███████ █████ █████ in the ferric carboxymaltose group and █████ █████ ███████ ███ █████ in the iron sucrose group (Table 17), with a reported between-group P value of ██████. The between-group difference and CI were not reported.
In the FERGIcor study, the mean baseline was █████ █████ ████ in the ferric carboxymaltose group and █████ █████ ████ in the iron sucrose group. After week 4, there were changes from baseline of ████ █████ ████ in the ferric carboxymaltose group and █████ █████ ████ in the iron sucrose group (Table 17) for an estimated between-group difference (repeated measures analysis) of █████████ ███ █████ ██ ████████ ███████.
In the VIT-IRON-2011-004 study, the mean baseline was ████ █████ ███████ in the ferric carboxymaltose group and ████ ██████ ███████ in the iron sucrose group. After week 8, there were changes from baseline of ███ █████ █████████ in the ferric carboxymaltose group and ██████ █████ ████████ in the iron sucrose group, for a least square mean between groups of █████ █████ ████ ███ ██████ ███████ ███████ (Table 17).
Transferrin Saturation
In Study VIT-IV-CL-015, there were █████ ████████ ███████ in the ferric carboxymaltose group and █████ ████████ ███████ in the iron sucrose group who had a normal TSAT of between 20% and 50% by week 4, for a reported between-group P value of ████████. The between-group difference and CI were not reported.
In the FERGIcor study, 117 of 222 patients (52.7%) in the ferric carboxymaltose group and 76 of 209 patients (36.4%) in the iron sucrose group had a normal TSAT level (20% to 50%) at week 12. The odds ratio (OR) was 2.05 (95% CI, 1.37 to 3.06; P < 0.001) in favour of ferric carboxymaltose (Table 17). The absolute between-group difference and CI were not reported.
The TSAT response (achieving a normal TSAT) was not assessed in the VIT-IRON-2011-004 study.
Patients Without Anemia
For patients without anemia in the FERGIcor trial, (a hemoglobin level of ≥ 120 g/L for females or ≥ 130 g/L for males), the percentage of patients at week 12 was 72.8% (166 of 228) in the ferric carboxymaltose group and 61.8% (136 of 220) in the iron sucrose group. The OR was 1.65 (95% CI, 1.10 to 2.46; P = 0.015). The absolute between-group difference and CI were not reported.
Harms Results
Adverse Events
In the overall study population in the VIT-IV-CL-015 trial, ██ ███████ patients in the ferric carboxymaltose group and ██ ███████ patients in the iron sucrose group had at least 1 adverse event (AE). In the FERGIcor study, AEs were reported in ████████ patients in the ferric carboxymaltose group and in ███ ███████ patients in the iron sucrose group.
In the overall study population in the 1VIT05006 trial, ███ ███████ patients in the ferric carboxymaltose group and ███ ███████ patients in the placebo group reported at least 1 AE. In the VIT-IRON-2011-004 study, ███ ███████ patients in the ferric carboxymaltose group and ███ ███████ patients in the iron sucrose group had AEs.
Serious Adverse Events
In the VIT-IV-CL-015 study, serious adverse events (SAEs) were reported in | ██████ patients in the ferric carboxymaltose group and ██ ██████ patients in the iron sucrose group. In the FERGIcor study, SAEs were reported in ██ ██████ patients in the ferric carboxymaltose group and ██ ██████ patients in the iron sucrose group.
In the 1VIT05006 study, | ██████ patient in the ferric carboxymaltose group and | ██████ patients in the placebo group had an SAE. In Study VIT-IRON-2011-004 ██ patients ██████ in the ferric carboxymaltose group and | ██████ patients in the iron sucrose group had an SAE.
Withdrawals Due to Adverse Events
In the VIT-IV-CL-015 study, | ██████ patients in the ferric carboxymaltose group and ███████ patients in the iron sucrose group withdrew from study medication due to AEs. In the FERGIcor study, 7 patients (2.9%) in the ferric carboxymaltose group and 2 patients (0.8%) in the iron sucrose group withdrew from the study drug because of an AE.
In Study 1VIT05006, | ██████ patients in the ferric carboxymaltose group and | ██████ patients in the placebo group withdrew from study medication due to an AE. In the VIT-IRON-2011-004 study, | ██████ patients in the ferric carboxymaltose group and ██████ patients in the iron sucrose group withdrew from study medication due to an AE.
Mortality
Across the studies, ███ ███████ in the ferric carboxymaltose group died during Study VIT-IV-CL-015 because of an acute anterior myocardial infarction. ███ ███████ died more than a week after study medication was withdrawn due to a nonserious AE. In Study 1VIT05006, ███ ███████ in the ferric carboxymaltose group died due to pneumonia. There were no deaths reported in the other 2 studies.
Notable Harms
In the FERGIcor study, hypophosphatemia was observed in 6 patients (2.5%) in the ferric carboxymaltose group and none were observed in the iron sucrose group. Hypophosphatemia was not reported in the VIT-IV-CL-015 study.
In the VIT-IRON-2011-004 study, hypophosphatemia was observed in ██ patients in the ferric carboxymaltose group ███████ and in ██ patients ██████ in the iron sucrose group. The number of patients with decreased blood phosphorous or hypophosphatemia was not reported in Study 1VIT05006.
Critical Appraisal
None of the active-controlled trials were blinded, and this may bias assessment of patient-reported outcomes such as HRQoL as well as the assessment of subjective harms. One of the 3 studies did not plan for any formal comparisons between ferric carboxymaltose and iron sucrose, while the other 2 did not implement a multiple-testing procedure, increasing the risk that statistically significant results (beyond the primary outcome) were false positives. Fewer patients in the ferric carboxymaltose group compared to the iron sucrose group withdrew from Study VIT-IV-CL-015 (████ ██████ █████), and this may also bias assessment of efficacy and harms in this relatively short (4-week) study. Between-group differences with CIs were infrequently reported, limiting the usefulness of judgments about the precision of the effect estimates.
Few if any patients in the included studies identified as Indigenous, and this is an important gap because there is a disproportionate number of patients with IDA in Canada who are Indigenous.
One of the pivotal studies, FERGIcor, assessed HRQoL, but the other studies did not, and no studies formally assessed the impact of ferric carboxymaltose on fatigue and other important outcomes in this patient population.
GRADE Summary of Findings and Certainty of the Evidence
In the absence of a complete body of evidence for any patient population or comparison, no Grading of Recommendations Assessment, Development and Evaluation (GRADE) assessment was performed for this review.
Table 2: Results for Ferric Carboxymaltose Versus Iron Sucrose for Patients With Iron Deficiency Anemia
Outcome | Studies, N | Ferric carboxymaltose | Iron sucrose | Between-group difference (95% CI) P value (statistical test) |
|---|---|---|---|---|
Hemoglobin | ||||
Patients with ≥ 10 g/L increase in hemoglobin at week 4, n of N (%) | VIT-IV-CL-015 (patients with CKD) N = 240 | Week 4 (PPS): 45 of 97 (46.4) | Week 4 (PPS): 32 of 86 (37.2) | NR P = 0.2101a (chi-square) |
Patients with ≥ 20 g/L increase in hemoglobin at week 12, n of N (%) | FERGIcor (patients with IBD) N = 465 | Week 12 (FAS, observed): 150 of 240 (65.8) | Week 12 (FAS, observed): 118 of 220 (53.6) | 12.15 (3.07 to 20.97) P = 0.004 (ANCOVA) |
Patients with ≥ 20 g/L increase in hemoglobin at any time to week 8, n of N (%) | VIT-IRON-2011-004 (patients with IDA arising from various causes) N = 371 | Week 8 (FAS): 184 of 185 (99.5) | Week 8 (FAS): 177 of 180 (98.3) | 1.13 (−2.02 to 4.68) P value = NR |
Mean change from baseline in hemoglobin | VIT-IV-CL-015 (patients with CKD) N = 240 |
N = 97
|
| NR |██████a (ANCOVA) |
FERGIcor (patients with IBD) N = 465 |
|
| NR |█████b (Repeated measures) | |
VIT-IRON-2011-004 (patients with IDA arising from various causes) N = 371 |
|
| ████ ██████ ██ ███████ █ █████a (repeated measures) | |
Health-related quality of life | ||||
SF-36 physical component, mean (SD) Scale ranges from 0 to 100, higher scores indicate better HRQoL | FERGIcor (patients with IBD) N = 465 |
|
| NR P = 0.157b (ANCOVA) |
SF-36 mental component, mean (SD) Scale ranges from 0 to 100, higher scores indicate better HRQoL | FERGIcor (patients with IBD), N = 465 |
|
| NR P = 0.583b (ANCOVA) |
IBDQ total score, mean (SD) Total score can range from 32 (very poor HRQoL) to 224 (perfect HRQoL) | FERGIcor (patients with IBD) N = 465 |
|
| NR P = 0.872b (ANCOVA) |
Ferritin | ||||
Mean (95% CI) change from baseline in serum ferritin (mcg/L) | VIT-IV-CL-015 (patients with CKD) N = 240 |
|
| NR |████a (ANCOVA) |
FERGIcor (patients with IBD) N = 465 |
|
| █████ ██████ ██ ███████ (repeated measures) | ██████ | |
VIT-IRON-2011-004 (patients with IDA arising from various causes) N = 371 |
|
| █████ ██████ ██ ██████ ██ █ █████a (repeated measures) | |
Transferrin saturation | ||||
Patients with a normal TSAT level of 20% to 50%, n/N (%) | VIT-IV-CL-015 (patients with CKD) N = 240 | Week 4: █████ ██████ | Week 4: █████ ██████ | NR ████████ (chi-square) |
FERGIcor (patients with IBD), N = 465 | Week 12 (FAS, observed): 117 of 222 (52.7) | Week 12 (FAS, observed): 76/209 (36.4) | OR = 2.05 (1.37 to 3.06) P < 0.001b (logistic regression) | |
Patients without anemia | ||||
Patients who were nonanemic (hemoglobin ≥ 120 g/L for males or ≥ 130 g/L females) at week 12 | FERGIcor N = 465 | Week 12 (FAS, observed): 166 of 228 (72.8) | Week 12 (FAS, observed): 136 of 220 (61.8) | OR = 1.65 (95% CI, 1.10, 2.46) P = 0.015 (logistic regression) |
ANCOVA = analysis of covariance; CFB = change from baseline; CKD = chronic kidney disease; CI = confidence interval; FAS = full analysis set; HRQoL = health-related quality of life; IBD = inflammatory bowel disease; IBDQ = Inflammatory Bowel Disease Questionnaire; NR = not reported; OR = odds ratio; PPS = per-protocol set; SF-36 = Short-Form (36) Health Survey; TSAT = transferrin saturation.
aThe statistical analysis plan for this study did not call for any formal hypothesis testing; therefore, all reported P values should be considered supportive in nature.
bThese P values have not been adjusted for multiple comparisons.
Source: Sponsor Summary of Clinical Evidence and the CSR for FERGIcor, VIT-IV-CL-015, and VIT-IRON-2011-004
Long-Term Extension Studies
There were no long-term extension studies submitted by the sponsor.
Indirect Comparisons
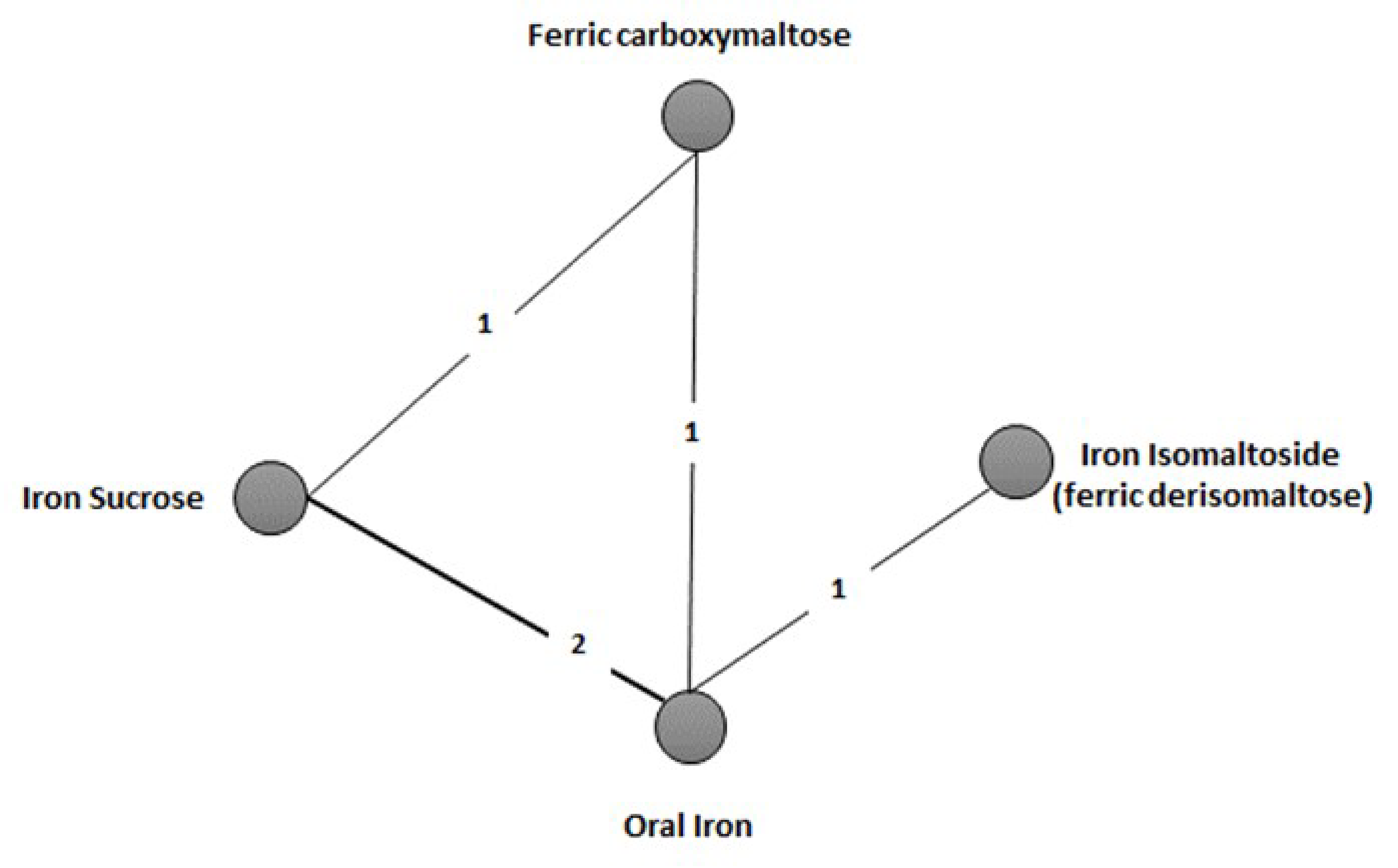
The sponsor submitted a summary of indirect evidence from a published network meta-analysis (NMA) that compared the effects of ferric carboxymaltose with those of other IV iron therapies in the context of adults with IDA associated with IBD.22
Description of Studies
The NMA submitted by the sponsor included 5 RCTs. All studies were performed in patients with IBD. The interventions in the studies included ferric carboxymaltose, iron sucrose, iron isomaltoside (also known as ferric derisomaltose), and oral iron.
Efficacy and Harms Results
Point estimates for the ORs comparing ferric carboxymaltose with ferric derisomaltose and iron sucrose favoured ferric carboxymaltose; however, the 95% credible intervals (CrIs) were wide, included effects favouring the comparator interventions, and showed no differences. Harms were not assessed in the NMA. Therefore, the summary of the published NMA submitted by the sponsor was insufficient to determine whether there is a difference in efficacy or harms for ferric carboxymaltose compared to other injectable forms of iron in patients with IDA and inflammatory bowel disease.
Critical Appraisal
The sponsor selected a published NMA that used a Bayesian approach with fixed-effects models for the analyses. Limited information is available regarding the methods used in the NMA model. The NMA was informed by a systematic review of relevant databases, although the search may not have been exhaustive. The date last searched was June 2016, more than 8 years ago at the time of writing this report. Relevant studies available since this time would have been excluded from the analyses. The authors performed an assessment of bias for the included studies but did not explain how the results of this assessment were incorporated into the analyses. It was not clear if the authors incorporated an assessment of clinical heterogeneity into their analyses.
Another limitation of the NMA is that it contained a small amount of data from 5 studies, resulting in a network with only 1 or 2 studies connecting the nodes, along with wide CrIs. Only 1 outcome was assessed in the NMA. There was no analysis of harms or other outcomes that are important for patients.
There was heterogeneity in the time point for end point evaluation, which could have biased the results. Few patient characteristics were reported across the trials, challenging a thorough assessment of the plausibility of the exchangeability assumption. Interpretation of the effect estimates was limited by imprecision. The 95% CrIs for the comparisons of ferric carboxymaltose and ferric derisomaltose and iron sucrose were wide and included the potential for no difference or that either treatment could be favoured.
Another significant limitation is related to the sponsor’s lack of clear, a priori criteria for selecting the NMA by Aksan et al.22 At least 2 other published NMAs have investigated the relative efficacy of ferric carboxymaltose in broader IDA populations. There was no protocol outlining a priori how 1 or more NMAs would be selected for presentation among multiple NMAs reporting on the same population, comparisons, and outcomes. As such, there is risk of bias in the selection of the NMA.
Summary of Indirect Treatment Comparison
A summary of the published NMA submitted by the sponsor was insufficient to determine whether there is a difference in efficacy for ferric carboxymaltose compared to other injectable forms of iron in patients with IDA and IBD. Important efficacy and harms outcomes were not investigated.
Studies Addressing Gaps in the Evidence from Pivotal Studies and Select RCTs
This section presents additional evidence from 6 studies that address gaps in the evidence from pivotal trials and select RCTs: 1 study in pediatric patients, 1 study in patients with IDA resulting from various causes, and 4 studies comparing ferric carboxymaltose with ferric derisomaltose in patients with IDA.
Study 1VIT17044
Study 1VIT17044 is a phase III, multicentre, randomized, active-controlled, open-label clinical trial conducted at 30 sites across 4 countries (the US, Ukraine, Poland, and Canada). The trial enrolled 79 patients who were assigned to receive ferric carboxymaltose (n = 40) or oral iron (n = 39). The trial aimed to investigate the efficacy and safety of ferric carboxymaltose versus oral iron in pediatric patients with IDA and a documented history of inadequate response to oral iron. In the 1VIT17044 trial, the least square mean change in hemoglobin from baseline to day 35 obtained through an analysis of covariance (ANCOVA) model was 2.22 g/dL (██ █ █████) in the ferric carboxymaltose group and 1.92 g/dL (██ █ █████) in the oral iron group. The treatment difference at day 35 was 0.30 g/dL (95% CI, −0.28 t 0.88; P = 0.3108). The results of the mixed model for repeated measures (MMRM) and the subgroup analyses align with the main analysis. The least square mean change in ferritin from baseline to day 35 obtained through the ANCOVA model were ██████ █████ ███ █ ███████ in the ferric carboxymaltose and █████ █████ ███ █ ███████ in the oral iron group. The treatment difference at day 35 was ██████ █████ ████ ███ ██████ ███████ ████████████████. The least square mean change in TSAT from baseline to day 35 obtained through the ANCOVA model was ██████ ███ █ ██████ in the ferric carboxymaltose and █████ ███ █ ██████ in the oral iron group. The treatment difference at day 35 was ██████ ████ ███ █████ ██████ ████████████████. In the 1VIT17044 trial, a larger percentage of participants in the ferric carboxymaltose group than in the oral iron group experienced at least 1 treatment-emergent adverse event (TEAE) ██ ███████ versus ██████████. Numerically more patients in the ferric carboxymaltose group than in the oral iron group experienced metabolism and nutrition disorders ████ ███ ███ and hypophosphatemia ████ ███ ███. Numerically fewer patients in the ferric carboxymaltose group than the oral iron group experienced GI disorders █████ ███ ██████ and constipation ███ ███ ██████. One patient in the ferric carboxymaltose group experienced a TEAE that led to treatment discontinuation. None of the patients experienced any SAE or any TEAE leading to death.
Critical Appraisal
Internal validity: Although the methods for randomization were likely appropriate, due to the small sample size there is an increased risk that prognostic balance was not achieved, as evidenced by imbalances in patients’ baseline disease and demographic characteristics. Notably, the baseline serum ferritin level was higher in the oral iron arm. There were also baseline imbalances by ethnicity and by body mass index (BMI). The effect of these differences on efficacy outcomes is unclear. The trial was open-label; however, the efficacy outcomes are objective, so it is unlikely that bias was introduced to the measurements. There is a risk of bias in the reporting of subjective harms (e.g., GI disorders and headaches) because patients knew which treatment they had been assigned (e.g., it is possible that known harms could be overestimated). The authors used the LOCF method to impute missing outcomes data and conducted sensitivity analyses using an MMRM under the missing-at-random assumption. Although neither method may be appropriate (the LOCF may not be reflective of the true trajectory of the outcome and the MMRM assumes data are missing at random, which is not possible to assess and may not be plausible), the attrition rate was low (5% or less) in each group. As such, it is unlikely that missing data would have introduced bias. The intention-to-treat (ITT) analysis was appropriate for estimating the effect of assignment to the interventions. Because there were no adjustments for multiple comparisons, there is an increased risk of type I error (false positives) for statistically significant results. Although the subgroup analyses were preplanned, these were unlikely sufficiently powered to detect subgroup differences.
External validity: The included patients were aged 1 to 17 years and results are not generalizable to other age groups. Further, given the small sample size, it is unlikely that the results would be broadly generalizable to all pediatric patients with IDA. As the comparator in the trial was oral iron, this study does not inform about the efficacy or harms of ferric carboxymaltose relative to other IV iron formulations in pediatric patients. Indigenous Peoples, who are disproportionately affected by IDA, are not represented in this trial. Although the outcomes measures were appropriate, some outcomes that may be important to patients (e.g., HRQoL) were not reported.
Study 1VIT09031
The 1VIT09031 trial is a phase III, multicentre, randomized, active-controlled, open-label study of the efficacy and safety of IV ferric carboxymaltose in adult patients with IDA who had an unsatisfactory response or intolerance to oral iron. Cohort assignment was based on results from a 14-day run-in period with oral iron. Patients with inadequate response to oral iron (hemoglobin increase < 1 g/dL) were assigned to cohort 1, and patients who were intolerant of oral iron were assigned to cohort 2. Oral iron was the comparator arm in cohort 1, and other IV iron standard of care (SOC) as chosen by the investigator was the comparator in cohort 2. In cohort 1, ███ patients received ferric carboxymaltose and ███ received oral iron. In cohort 2, ███ patients received ferric carboxymaltose and ███ received IV SC. Patients were followed up to day 35 for efficacy assessment, and up to day 120 for safety assessment. For the protocol-specified primary treatment group comparison (cohort 1), the mean increases in hemoglobin from baseline to the highest value between baseline and day 35 or time of intervention (TOI) were 1.57 g/dL ███ █ █████ in group A (ferric carboxymaltose) and 0.80 ███ █ █████ in group B (oral iron) (P = 0.001). No between-group difference and CI were reported. In a post hoc comparison of group C (ferric carboxymaltose) versus group D (IV SOC) (cohort 2), the mean increases in hemoglobin from baseline to the highest value between baseline and day 35 or TOI were 2.90 g/dL ███ █ █████ in group C (ferric carboxymaltose) and 2.16 ███ █ █████ group D (IV SOC). No between-group difference and CI were reported. Subgroup analysis revealed the mean increase in hemoglobin from baseline to the highest value between baseline and day 35 or TOI was greater for the ferric carboxymaltose group than the comparator group regardless of baseline hemoglobin value or etiology of IDA.
In cohort 1, the proportion of patients achieving hemoglobin above 12.0 g/dL was █████ in the ferric carboxymaltose group and █████ in the oral iron group (P < ████). The proportion of patients with a clinically meaningful increase in hemoglobin (as defined by the investigators) was █████ in the ferric carboxymaltose group and ████ in the oral iron group (P < ████). The proportion of patients with a hemoglobin level greater than 12 g/dL and a ferritin increase of 160 ng/mL or greater was █████ in the ferric carboxymaltose group and ████ in the oral iron group (P < ████). The mean change in hemoglobin was ███ g/dL (SD = ███) in the ferric carboxymaltose group and ███ g/dL (SD = ███) in the oral iron group (P < ████). The means of changes in ferritin were █████ ng/mL (SD = █████) in the ferric carboxymaltose group and ████ ng/mL (SD = ████) in the oral iron group (P < ████). The mean change in TSAT was █████ (SD = ████) in the ferric carboxymaltose group and █████ (SD = ████) in the oral iron group (P < ████). Between-group differences and CIs were not reported for any outcome.
In cohort 2, the proportion of patients achieving hemoglobin above 12.0 g/dL was █████ in the ferric carboxymaltose group and █████ in the IV iron group (P < ████). The proportion of patients with a clinically meaningful increase in hemoglobin (as defined by the investigators) was █████ in the ferric carboxymaltose group and █████ in the IV iron group (P < ████). The proportion of patients with a hemoglobin level greater than 12 g/dL and a ferritin increase of 160 ng/mL or greater was █████ in the ferric carboxymaltose group and ████ in the IV iron group (P < ████). The means of changes in hemoglobin were ███ g/dL (SD = ███) in the ferric carboxymaltose group and ███ g/dL (SD = ███) in the IV iron group (P < ████). The mean change in ferritin was █████ ng/mL (SD = █████) in the ferric carboxymaltose group and ████ ng/mL (SD = █████) in the IV iron group (P < ████). The mean change in TSAT was ██████ (SD = ████) in the ferric carboxymaltose group and ████ (SD = ████) in the oral iron group (P < ████). Between-group differences and CIs were not reported for any outcome.
There were numerically more TEAEs in group A ███████ compared with group B ███████. Overall rates of TEAEs were similar between group C (ferric carboxymaltose) and group D (IV SOC) (██ ██ ███ across groups). The TEAS most commonly experienced (≥ 5%) were nausea in group A ██████, hypophosphatemia in group C ██████, and dizziness in group D ██████. No TEAE was experienced in 5% or more of patients in group B. █████ ██████ patients in group A (ferric carboxymaltose), ██ ██████ patients in group B (oral iron), ██ ██████ patients in group C (ferric carboxymaltose), and ████████ patients in group D (IV iron) experienced at least 1 SAE during the treatment phase. █████ ██████ patients in group A (ferric carboxymaltose) and ████ in the group B (oral iron) experienced hypersensitivity reactions. ███ ██████ patients in group C (ferric carboxymaltose) and █████ in group D (IV iron) experienced hypersensitivity reactions. Hypersensitivity reactions in the patients receiving ferric carboxymaltose were either grade 2 or 3, and in the IV SOC group they were grades 1 to 3. ██████ ██████ patients in group A (ferric carboxymaltose) and ██████ in group B (oral iron) experienced skin and subcutaneous tissue disorders. ████████ ██████ patients in group C (ferric carboxymaltose) and ████████ in group D (IV iron) experienced skin and subcutaneous tissue disorders. All TEAEs associated with skin and subcutaneous tissue disorders (erythema, pruritus, rash, maculopapular rash, and urticaria) were either grade 1 or 2. In group A (ferric carboxymaltose), ████ of patients experienced hypophosphatemia and █████ experienced potentially clinically significant (PCS) low-phosphorus levels compared with ██ ███ ████ in group B (oral iron), respectively. In group C (ferric carboxymaltose), ████ of patients experienced hypophosphatemia and █████ experienced PCS low-phosphorus levels compared with ██ ███ ████ in group D (IV SOC), respectively. Hypophosphatemia was mainly grade 1 to 3 in severity, with a single grade 4 event. ████ PCS low-phosphorus events were grade 3, with ██ grade ██ events in patients in the ferric carboxymaltose group. ██ PCS low-phosphorus events were associated with serious or severe AEs. ███ patient had events of somnolence, fatigue, tingling finger, swollen hand, and an elevated white blood cell count on days when phosphorus levels were grade 4. In total, ██ deaths were reported in the study, ██ in group B (oral iron), ██ in group C (ferric carboxymaltose), and ███ in group D (IV iron). ████ were considered related to study drug by investigator.
Critical Appraisal
Internal validity: Although the methods for randomization and allocation concealment appeared to be adequate, imbalances were evident at baseline in some important clinical characteristics. Most notably, serum ferritin was higher in group C (ferric carboxymaltose) compared with group D (IV SOC). It is not certain if this imbalance may have biased the results. The trial was open-label; however, the efficacy outcomes are objective, so it is unlikely that bias was introduced in their measurement. There is a risk of bias in the reporting of subjective harms because patients knew which treatment they had been assigned (e.g., it is possible that known harms could be overestimated). Across groups, up to ███ of patients did not complete the study, and methods for handing missing data are not clear. As such, there is a risk of bias due to missing outcomes data, but the extent and direction of the bias cannot be predicted. The modified intention-to-treat (mITT) analysis was appropriate for estimating the effect of assignment to the interventions. Although the mITT population was a subset of the ITT population, few patients (< 4% across groups) were excluded from the mITT analysis set. Because there were no adjustments for multiple comparisons, there is an increased risk of type I error (false positives) for statistically significant results. Between-group differences and CIs were not reported for any outcome, precluding judgments about the precision of the effects.
External validity: In this study, all patients received 2 doses of 15 mg/kg, up to a maximum single dose of 750 mg and a maximum cumulative dose of 1,500 mg. These values were all lower than the recommended dosing in the product monograph, which recommends a maximum single relative dose of 20 mg/kg, a maximum single absolute dose of 1,000 mg, and a maximum total dose of 2,000 mg. In group D, patients could have been assigned to any of 5 IV iron formulations, only 2 of which were considered relevant comparators for this review. As such, the generalizability of the results may be limited. The comparison of group A (ferric carboxymaltose) to group B (oral iron) does not inform the efficacy and harms of ferric carboxymaltose relative to other IV oral formulations available in Canada, limiting the applicability of these results. The study was conducted at 84 centres in the US only, with no patients from Canada and no representation of Indigenous Peoples, who are disproportionately affected by IDA. As only adults were enrolled in the study, it is uncertain whether the results can be generalized to pediatric patients. For group A (ferric carboxymaltose) and group B (oral iron), the study selected patients based on adherence to oral iron following a run-in phase, and a large proportion of these patients █████ were not enrolled. As such, the enrolled patients may not be representative of patients seen in clinical practice (as patients with lower adherence were not enrolled). Although relevant outcomes were investigated in the study, other outcomes that may be important to patients (e.g., HRQoL) were not investigated.
Zoller (2023)
Zoller (2023) is a multicentre, randomized, double-blind, active-controlled, clinical trial conducted at 20 outpatient hospital clinics in Austria, Denmark, Germany, Sweden, and the UK. The trial compared the incidence of hypophosphatemia after treatment with ferric carboxymaltose (N = 48 patients) versus ferric derisomaltose (N = 49 patients) in 97 patients with IDA and IBD. By day 70 (the end of the trial), the levels of ferritin and TSAT increased in both treatment groups. The hemoglobin increase by day 70 in the ferric derisomaltose group was 24.9 g/L (95% CI, 21.1 g/L to 28.8 g/L) and in the ferric carboxymaltose group was 25.2 g/L (95% CI, 21.3 g/L to 29.1 g/L). The between-group difference and CI were not reported for any efficacy outcome. Both ferric derisomaltose and ferric carboxymaltose resulted in improvement in fatigue symptoms and increased Functional Assessment of Chronic Illness Therapy–Fatigue (FACIT-F) scale scores (possible scores range from 0 to 52), which was statistically significantly greater for patients treated with ferric derisomaltose versus ferric carboxymaltose at days 35 and 49. The between-group difference and CI were not reported at any follow-up time point.
Numerically, more patients experienced hypophosphatemia and vitamin D deficiency in the ferric carboxymaltose group compared with those in the ferric derisomaltose group (hypophosphatemia: 28.6% versus 2.1% and vitamin D deficiency: 34.7% versus 22.9%, respectively). Numerically, fewer patients experienced headache and nausea in the ferric carboxymaltose group compared with those in the ferric derisomaltose group (headache: 10.2% versus 18.8%; and nausea: 2.0% versus 12.5%, respectively). Discontinuation due to AEs occurred among 6.3% of patients in the ferric derisomaltose group and 2.0% of patients in the ferric carboxymaltose group. There were no deaths in the trial. Hypophosphatemia in this trial was defined as a serum phosphate level of less than 2.0 mg/dL. The primary end point was the incidence of hypophosphatemia at any time after the first dose to day 35, which was reported as 8.3% (4 of 48) in the ferric derisomaltose group and 51.0% (25 of 49) in the ferric carboxymaltose group with an adjusted risk difference of −42.8% (95% CI, –57.1% to –24.6%; P < 0.0001) favouring ferric derisomaltose. The majority of patients recovered from hypophosphatemia by day 70. In an analysis by diagnosis of IBD in the ferric carboxymaltose group, the risk differences were 43.1% and 45.5% higher for patients with ulcerative colitis and Crohn disease, respectively (interaction P value = 0.1948). The highest incidence of hypophosphatemia occurred within 2 weeks of treatment in both arms. The secondary safety end point was the incidence of hypophosphatemia at any time from baseline to day 70, which occurred among 12.5% (6 of 48) of patients in the ferric derisomaltose group and 59.2% of patients (29 of 49) in the ferric carboxymaltose group, with an adjusted risk difference of −46.6% (95% CI, −60.9% to –28.1%; P < 0.0001) favouring ferric derisomaltose. Per the investigators, the mean decreases in phosphate concentration from baseline after the first and second doses were significantly greater after ferric carboxymaltose infusion compared to ferric derisomaltose infusion (between-group differences and CIs were not reported). For more than 1 month after the second infusion, 4.7% (2 of 43) of patients in the ferric carboxymaltose group remained hypophosphatemic. On day 70 the mean serum phosphate remained significantly lower in the ferric carboxymaltose group compared to the ferric derisomaltose group (between-group differences and CIs were not reported).
Critical Appraisal
Internal validity: Although the methods for randomization appeared appropriate, there is an increased risk that prognostic balance was not achieved due to the small sample size, as evidenced by imbalances in some baseline disease and demographic characteristics. Notably, there were imbalances at baseline in IBD diagnosis (Crohn disease or ulcerative colitis). The use of some concomitant medications (e.g., interleukin inhibitors and vitamin D supplements) was also imbalanced between groups. The effect of these differences on the efficacy and safety results is uncertain. Although the absolute risk differences for hypophosphatemia among patients with Crohn disease and ulcerative colitis who were treated with ferric carboxymaltose versus ferric derisomaltose were similar (and the interaction P value was not statistically significant), the subgroups were small, and the analysis was likely not sufficiently powered to detect subgroup differences. As the trial was double-blinded and methods to maintain the blinding appeared to be adequate, there is likely a low risk of bias in the measurement of the outcomes. Considering the importance of patient-reported outcomes, the use of the FACIT-F scale, which is a reliable and valid instrument for measuring fatigue in IBD, was appropriate.23 Because the trial was double-blinded, the risk of bias due to reporting subjective patient-reported outcomes is low. However, judgments about the precision of the effects are precluded because the between-group difference and CIs were not reported at any follow-up time point. For the analysis of hypophosphatemia, 2 patients in the ferric derisomaltose group and 1 patient in the ferric carboxymaltose group did not have a postbaseline observation and were imputed as having hypophosphatemia in the primary analysis. As the proportion of patients with missing data was low for this outcome, there is likely a low risk of bias due to missing outcomes data. Further, a post hoc sensitivity analysis in which these patients were imputed as either being free of hypophosphatemia or were excluded yielded similar results to the primary analysis. There was no imputation of missing values in this trial, except for the change from baseline in patients with no postbaseline measurements, which was set to zero at the first postbaseline visit. Furthermore, the attrition rates were 10% in the ferric derisomaltose group and 12.5% in the ferric carboxymaltose group; as a result, the risk of bias because of missing data is not high. Statistical analyses in this trial were not adjusted for multiple comparisons. As such, there is an increased risk of false-positive conclusions for statistically significant results. Particularly for efficacy outcomes (e.g., change in hemoglobin, ferritin, TSAT levels and fatigue), between-group differences with CIs were not reported, and no conclusions could be made about the magnitude (including clinical importance) of the estimated effects and their precision.
External validity: All patients enrolled in this trial had IBD; however, the results may not be broadly generalizable given the small sample size of the trial. The length of follow-up was relatively short, which limits the usefulness of conclusions about efficacy and safety over a longer period and among patients who require chronic treatment with IV iron. Although the efficacy outcomes measured were appropriate, conclusions about the magnitude of the estimated effects and their precision were limited due to insufficient reporting of between-group differences and CIs. Indigenous Peoples, who are disproportionately affected by IDA, were not represented in this study. In this trial, patients received a single IV infusion of 1,000 mg at baseline (day 0) and, depending on the a priori calculated iron dose, either 500 mg or 1,000 mg at day 35. According to the product monograph for ferric carboxymaltose, a single dose should not exceed 15 mg iron/kg of body weight or 1,000 mg of iron. Based on the product monograph for ferric derisomaltose24 the allowable iron dose per infusion is limited to 20 mg iron/kg of body weight. The dosage administered in this trial therefore aligns with those of the product monographs.
Emrich (2020)
Emrich (2020) is a prospective, single-centre, double-blind study. The study randomized 26 women with IDA to receive ferric carboxymaltose (n = 13) and or ferric derisomaltose (n = 13). This trial aimed to assess hypophosphatemia after high-dose iron repletion with ferric carboxymaltose and ferric derisomaltose. The trial assessed the quality of life using the SF-36, functional impairment by the Sheehan disability scale, and fatigue by the German version of the Multidimensional Fatigue Inventory, but the results have not been published. Changes in the levels of hemoglobin, ferritin, and TSAT were not trial end points and were not reported. The primary outcome was hypophosphatemia at any postinfusion study visit. This was reported for 75% (9 of 12) of the patients in the ferric carboxymaltose group and 8% (1 of 13) of the patients in the ferric derisomaltose group, as measured at study visit 4 (days 5 to 9) (P = 0.001). At study visit 5 (days 33 to 37), 25% (3 of 12) and 8% (1 of 13) of patients in the ferric carboxymaltose and ferric derisomaltose groups, respectively, had hypophosphatemia. The between-group difference with CI was not reported at any follow-up time point.
Critical Appraisal
Internal validity: The methods of randomization appeared to be appropriate; however, there are imbalances in some baseline characteristics that may be due to the small sample size. Furthermore, due to logistical reasons, after the inclusion of 26 patients (instead of the estimated 30 to achieve 80% power) the interim analysis was conducted, and the trial was completed after interim analysis. Therefore, there is the possibility that if more patients had been enrolled, then the effect size may have been differed. Due to lack of information in the publication, the adequacy of blinding is unclear. As only 1 patient was excluded from the analyses, the risk of bias because of missing data is low. There is an increased risk of false-positive conclusions for statistically significant results, as the statistical analyses were not adjusted for multiple comparisons. The between-group differences and CIs were not reported for the primary outcome of hypophosphatemia, precluding conclusions about the magnitude of the estimated effect and its precision.
External validity: Given the small sample size of the trial and considering that all patients enrolled in this trial were female and Caucasians (wording used in the source) the results may not be broadly generalizable. Further, the results also are not generalizable to the patients who were excluded from the study because of advanced CKD, pregnancy, ongoing lactation, untreated hyperparathyroidism, hemochromatosis, active malignancy, bronchial asthma, atopic dermatitis, active alcohol or drug abuse, or a history of a psychological illness or seizures. The length of follow-up was relatively short, which limits conclusions about the safety result (hypophosphatemia) over a longer duration. In this trial, only 1 infusion was performed, which limits detection of consequences of repeated infusions.
Wolf (2020)
Wolf (2020) reports on 2 identically designed, open-label, randomized clinical trials that assessed the effects of ferric carboxymaltose and ferric derisomaltose on hypophosphatemia. In trial A, 123 patients were randomized to receive ferric carboxymaltose (N = 61) or ferric derisomaltose (N = 62). In trial B, 122 patients were randomized to receive ferric carboxymaltose or ferric derisomaltose (N = 61 in each group). In total, 122 patients were randomized to receive ferric carboxymaltose and 123 to receive ferric derisomaltose. The results of hemoglobin, ferritin, and TSAT measurements from trials A and B are presented; however, changes from baseline and between-group differences with CIs were not reported for hemoglobin, ferritin, and TSAT outcomes at any time point.
Overall, in the ferric carboxymaltose versus ferric derisomaltose groups, 27 of 60 patients (45.0%) versus 7 of 63 patients (11.1%) patients in trial A and 28 of 57 (49.1%) versus 14 of 62 (22.6%) in trial B experienced AEs. Serious or severe hypersensitivity reactions occurred in 1 patient (0.8%) in the ferric derisomaltose group (swollen eyelid unilaterally) and in 2 patients (1.7%) in the ferric carboxymaltose group (dyspnea and swelling). Regarding the specific adverse drug reaction, in the ferric carboxymaltose versus ferric derisomaltose groups, 12 (20.0%) versus 0 patients in trial A and 7 (12.3%) versus 0 in trial B experienced decreased blood phosphorus. In the ferric carboxymaltose versus ferric derisomaltose groups, 12 (20.0%) versus 0 patients in trial A and 14 (24.6%) versus 2 (3.2%) in trial B experienced hypophosphatemia. The incidence of hypophosphatemia at any time from baseline to day 35 in the ferric derisomaltose group compared with the ferric carboxymaltose group in trial A was 7.9% versus 75.0% (adjusted rate difference = −67.0%; 95% CI, −77.4% to −51.5%; P < 0.001) favouring ferric derisomaltose, and in trial B it was 8.1% versus 73.7% (adjusted rate difference = −65.8%; 95% CI, −76.6% to −49.8%; P < 0.001) favouring ferric derisomaltose.
Critical Appraisal
Internal validity: Both trials A and B were open-label randomized clinical studies. Although the methods for randomization appeared to be appropriate, there were some imbalances in baseline characteristics, such as sex and race, and minor imbalances in ferritin and TSAT levels. These imbalances may have resulted from the small sample sizes of the included studies, which increases the risk that prognostic balance between groups may not have been achieved. The open-label nature of the study may increase the risk of bias in determining the magnitude of the subjective safety outcomes. The efficacy outcomes are objective and unlikely to be at risk of bias on account of the open-label design. However, the changes from baseline and between-group differences with CIs were not reported for some of the efficacy outcomes (hemoglobin, ferritin, and TSAT) which precludes making judgments about the precision of the effects. There was no adjustment for multiple comparisons, which increases the risk of type I error (false positives) for statistically significant results. There was no imputation of missing values in this trial, except for the change from baseline in patients with no postbaseline measurements, which was set to zero at the first postbaseline visit. The risk of bias due to missing data on outcomes is low, considering the low rate of attrition in both trials.
External validity: The included patients were mostly women with IDA due to gynecological bleeding. In both trials, most patients were white; therefore, results may not be generalizable to a broader population. Although the outcome measures were appropriate, some outcomes that may be important to patients (e.g., HRQoL) were not reported. Patients with conditions such as alcohol or drug abuse, pregnancy or lactation, untreated hyperparathyroidism, kidney transplant, a body weight less than 50 kg, or hemochromatosis or other iron-storage disorder were excluded, and the results are not generalizable to these groups of patients. In this study, all patients in the ferric carboxymaltose groups received 750 mg of ferric carboxymaltose on days 0 and 7, whereas those in the ferric derisomaltose group received 1,000 mg on day 0. According to the product monograph for ferric carboxymaltose, a single dose should not exceed 15 mg iron/kg of body weight or 1,000 mg of iron. Based on the product monograph for ferric derisomaltose, the allowable iron dose per infusion is limited to 20 mg iron/kg of body weight. While the dose administered aligned with the product monographs, patients in the ferric carboxymaltose arm received 500 mg more iron compared with those in the ferric derisomaltose arm. Because of the short follow-up time, this study does not inform the long-term clinical implications of these drugs. Additionally, the clinical outcomes associated with hypophosphatemia have not been reported.
Conclusions
Results from 2 phase III pivotal trials in patients with IDA secondary to IBD and to CKD, and another phase III RCT selected because it included patients with more generic IDA, suggest that ferric carboxymaltose is at least as effective at increasing hemoglobin as iron sucrose, after 4 to 12 weeks of follow-up. None of the included studies were designed to formally assess other efficacy outcomes, such as TSAT and ferritin levels, or important outcomes such as HRQoL; however, there was no indication from the included trials of a notable, clinically significant difference between ferric carboxymaltose and iron sucrose for any of these outcomes. There was no indication of a clear difference in risk of AEs or SAEs associated with ferric carboxymaltose versus iron sucrose, with the exception of hypophosphatemia, which may occur more frequently with ferric carboxymaltose. There is evidence that ferric carboxymaltose may be efficacious and safe for use in pregnancy and in children, and the clinical expert consulted for this review noted that there is a shortage of parenteral iron formulations with evidence in these populations. There is also evidence that the efficacy of ferric carboxymaltose is comparable to that of ferric derisomaltose; however, there is clearly an increased risk of hypophosphatemia with ferric carboxymaltose versus this other iron formulation. The clinical significance of this increased risk of hypophosphatemia is not known.
Introduction
The objective of this report is to review and critically appraise the evidence submitted by the sponsor on the beneficial and harmful effects of ferric carboxymaltose 50 mg/mL IV infusion in the treatment of IDA in adult and pediatric patients aged 1 year and older when oral iron preparations are not tolerated or are ineffective.
Disease Background
Contents within this section have been informed by materials submitted by the sponsor and clinical expert input. The following have been summarized and validated by the CDA-AMC review team.
Overview of the Condition
Iron deficiency is defined heterogeneously, but can be characterized as a condition in which iron availability is insufficient to meet the body’s needs.25 The British Columbia clinical guidelines on the diagnosis and management of iron deficiency define the condition as “insufficient total body iron stores, caused by increased requirements, decreased intake, increased loss, and/or decreased absorption.”4 Prolonged iron deficiency can lead to anemia.
Anemia is a medical condition in which the blood has reduced capacity to carry oxygen, either due to a reduced number of red blood cells or low hemoglobin levels. Anemia has a range of causes; however, iron deficiency is the most prevalent.1 IDA results when body iron levels are insufficient to sustain hemoglobin synthesis.2 Alberta clinical practice guidelines define IDA as “a serious condition whereby low levels of iron are associated with anemia and the presence of microcytic hypochromic red cells.”3
The British Columbia clinical guidelines define anemia based on hemoglobin levels relative to the population mean, as “[a] low hemoglobin level, most frequently defined as a hemoglobin value over 2 standard deviations below the gender- and age-adjusted mean. A hemoglobin value below the local, lab-specific lower reference interval indicates anemia.”4 Patients with IDA report dizziness, extreme fatigue, headache, shortness of breath, and muscle weakness as frequent symptoms of their condition.
Prevalence of IDA in the General Population
In a Canadian Health Measures Survey study, approximately 2% of the population in Canada were identified as having IDA, with females experiencing a prevalence approximately 9 times higher than males. Total population IDA prevalence, corrected for inflammation in a recent study by Cooper et al.,5 ranged from 2% to 2.5%. Notably, less than 1% of males exhibited IDA regardless of the correction method, whereas this figure rose to approximately 4% for all females. For females of child-bearing age, the corrected IDA prevalence ranged between 5% and 6.2%.5
Prevalence of Iron Deficiency and IDA in Pregnant Females
Maternal ID, anemia, and IDA are most prevalent during the third trimester of pregnancy and may manifest as fatigue, restless leg syndrome, pica, hair loss, and irritability, which are often dismissed as pregnancy-related symptoms. Iron deficiency and IDA during the prenatal period have been associated with adverse effects on neonatal growth, neurocognitive development, and perinatal morbidity and mortality. A 2022 study conducted in British Columbia assessed iron status during early and late pregnancy among 60 pregnant patients receiving 27 mg/day of elemental iron as part of a randomized trial.6 High rates of iron deficiency were observed: 28% of participants exhibited probable iron deficiency at baseline (mean gestational age = 16 weeks), with the proportion increasing to 81% by study end (mean gestational age = 32 weeks).6 This deterioration in iron status occurred despite widespread supplementation and highlights a potential need for additional iron beyond the recommended dosage to support the needs of later stages of pregnancy.
Prevalence of Iron Deficiency and IDA in Indigenous Adults and Children
While the general Canadian population maintains a relatively low prevalence of anemia (approximately 3%), this is not the case among the Indigenous population. In a 2017 study of the Inuit in Nunavik (16 years and older), women of child-bearing age exhibited a 20% prevalence of anemia, with 14% of them experiencing IDA and 23% demonstrating iron deficiency without anemia, while the remainder had another, unspecified type of anemia.7 Data were not available for other Indigenous Peoples.
Anemia has a range of causes, so a diagnosis of IDA is based on identification of both anemia and iron deficiency. IDA is diagnosed based on blood tests showing low hemoglobin levels coupled with markers for ID, typically ferritin levels and TSAT.
Standards of Therapy
Contents within this section have been informed by materials submitted by the sponsor and clinical expert input. The following have been summarized and validated by the review team.
The key goals in the treatment of iron deficiency and IDA are the correction of the hemoglobin deficit and repletion of iron stores (the correction phase) and maintenance of iron levels over time (the maintenance phase).2,8
Oral iron, often in the form of ferrous sulphate, is the first-line therapy for most cases of iron deficiency, and IDA and is relatively safe, effective, and inexpensive. Some patients may be unable to absorb ferrous sulphate adequately due to impaired intestinal uptake resulting from GI disease or clinical conditions such as chronic inflammation, which may in turn lead to elevated levels of hepcidin.9-11 In other instances, the rate of absorption of even high-dose oral ferrous sulphate is insufficient to correct the anemia, and blood transfusion may be indicated.9,12
Oral iron supplements also carry the additional burden of GI side effects, which can have a detrimental impact on patient adherence to treatment.2,8 A systematic review and meta-analysis of RCTs found a significantly increased risk of GI side effects, particularly constipation, nausea, and diarrhea, for ferrous sulphate compared to placebo (OR = 2.32; 95% CI = 1.74 to 3.08; P < 0.0001).26
There is also a need to deliver iron rapidly in certain clinical situations, including, for example, when a patient with iron deficiency requires an urgent surgery with risk of blood loss.13 While repletion of iron stores with oral iron may require administration over several months, the correction of iron stores following IV iron administration occurs within a few weeks.8,12
IV iron treatment may also be preferred over oral iron in other situations. Oncology guidelines suggest that IV iron is superior to oral iron in combination with erythropoiesis-stimulating agents (ESAs) for chemotherapy-induced anemia.14-16 There is also strong evidence from clinical trials supporting the preference of IV iron in patients with CKD with or without concomitant ESA treatment.17-20 In children, IV iron substitution may be necessary in situations where oral iron therapy is not possible, ineffective, or poorly tolerated. According to Mattiello et al.,21 a switch to IV iron is proposed for cases with severe anemia (hemoglobin < 70 g/L) to avoid transfusion, for cases with an underlying secondary disease fulfilling a formal indication for IV iron (e.g., IBD, chronic GI or genitourinary bleeding, or celiac disease), and/or for situations of nonadherence to oral treatment and symptomatic refractory IDA with clinical impact.21
In cases of severe IDA, blood transfusion may be needed to increase hemoglobin to secure sufficient oxygen transport to the tissues and simultaneously replete iron stores.27,28 Blood transfusions are the first-line treatment of iron deficiency only in cases of severe and critical anemia. Generally, blood transfusions are not indicated to treat iron deficiency due to the risk of potential disease transmission and transfusion-related complications.29-32
The earliest IV iron preparations were associated with acute toxicity resulting from the release of bioactive labile iron. Subsequent iron formulations were complexes with carbohydrates, which release iron more slowly. Newer IV iron products, such as ferric carboxymaltose can be given in larger doses over a shorter period.12
In addition to ferric carboxymaltose, there are currently 3 other IV iron products approved in Canada: Ferrlecit (sodium ferric gluconate complex in sucrose; 12.5 mg/mL), Monoferric (ferric derisomaltose; 100 mg/mL), Venofer (iron sucrose; 20 mg/mL, pms-Iron Sucrose (iron sucrose injection; 20 mg/mL). None of these products is indicated for pediatric populations. The product monograph for ferric derisomaltose cautions against its use in pregnancy based on data from animal studies, and the product monograph for iron sucrose states that it should only be used if the potential benefit outweighs the potential risk to the fetus. Most of these products must be administered over a long period of time and/or with a series of sessions to attain a cumulative dose of 1,000 mg.
Drug Under Review
Each millilitre of ferric carboxymaltose contains 50 mg of elemental iron, and the dose is determined by the iron need and followed up with assessments after iron repletion. Ferric carboxymaltose is indicated for the treatment of IDA in adult and pediatric patients aged 1 year and older when oral iron preparations are not tolerated or are ineffective and for the treatment of iron deficiency in adult patients with New York Heart Association Class II or III heart failure and to improve exercise capacity. The diagnosis of iron deficiency must be based on laboratory tests.
Ferric carboxymaltose contains iron in a stable ferric state as a non-dextran iron complex consisting of a polynuclear iron-hydroxide core with a carbohydrate ligand. The complex is designed to provide utilizable iron for the iron transport and storage proteins in the body (transferrin and ferritin, respectively).
The sponsor’s reimbursement request is consistent with the indication. However, this review focuses only on the IDA indication while another review examines iron deficiency in adult patients with heart failure. Ferric carboxymaltose was granted a Notice of Compliance on March 11, 2024, under the standard review process at Health Canada.
Key characteristics of ferric carboxymaltose are summarized in Table 3 with other IV treatments available for IDA. According to the clinical expert consulted for this review, Ferrlecit is not broadly used in Canada relative to the other IV iron formulations. It was also not considered a comparator in the sponsor’s systematic review. As such, it was not a relevant comparator for this review.
Table 3: Key Characteristics of Ferric Carboxymaltose, Ferric Derisomaltose, and Iron Sucrose
Characteristic | Ferric carboxymaltose | Ferric derisomaltose | Iron sucrose |
|---|---|---|---|
Mechanism of action | Replenishes iron | Replenishes iron | Replenishes iron |
Indicationa | For the treatment of IDA in adult and pediatric patients 1 year of age and older when oral iron preparations are not tolerated or are ineffective For the treatment of iron deficiency in adult patients with New York Heart Association Class II or III heart failure and to improve exercise capacity | Treatment of IDA in adult patients who have intolerance or unresponsiveness to oral iron therapy; diagnosis must be based on laboratory tests | Treatment of IDA in patients with:
|
Route of administration | IV | IV | IV |
Recommended dose | A single FCM administration should not exceed either:
The maximum recommended cumulative dose of is 1,000 mg of iron (20 mL) per week; if the total iron need is higher, then the administration of an additional dose should be a minimum of 7 days apart from the first dose | IV administration as drip infusion, bolus injection, or injection into venous limb of dialyzer Total dosage based on iron need determined either from simplified table, or Ganzoni formula; a single infusion should not exceed 20 mg of iron/kg of body weight or 1,500 mg. | Administration intravenously by slow injection or infusion. Administered as a total cumulative dose of 1,000 mg over a 14-day period as a 200 mg slow IV injection undiluted over 2 to 5 minutes on 5 different occasions within the 14-day period |
Serious adverse effects or safety issues | Hypersensitivity reactions including anaphylaxis | Hypersensitivity reactions including anaphylaxis Hypotension | Hypersensitivity reactions including anaphylaxis Hypotension |
CKD = chronic kidney disease; IDA = iron deficiency anemia.
aHealth Canada–approved indication.
Source: Product monographs for ferric carboxymaltose,33 ferric derisomatlose,24 and iron sucrose.34
Perspectives of Patients, Clinicians, and Drug Programs
Patient Group Input
This section was prepared by the review team based on the input provided by patient groups. The full original patient input(s) received by the team have been included in the Perspectives of Patients, Clinicians, and Drug Programs section of this report.
One patient group, the GI Society, provided input to this submission. The GI Society is a national charity formed in 2008 on the groundwork of its partner organization, the Canadian Society of Intestinal Research, and is committed to improving the lives of people with GI and liver conditions, supporting research, advocating for appropriate patient access to health care, and promoting GI and liver health. The GI Society stated that it obtained information for this submission from meetings and discussions with health care professionals and researchers, as well as surveys and interviews conducted on digestive and liver diseases. The number of survey respondents or interviewees was not mentioned; however, the patient group clarified that none of the patients had experience with ferric carboxymaltose.
According to the patient group input, dizziness, extreme fatigue, headaches, shortness of breath, tiredness, and muscle weakness are common and frequent symptoms of IDA. Other signs include extreme paleness, cold hands and feet, depression, and cravings for things that are not food (pica) or having a poor appetite, particularly in infants and children. The GI Society stated that IDA can have significant effects on a person physically, mentally, financially, and socially. Because IDA often arises from a pre-existing condition, such as celiac disease and/or IBD, many individuals must manage treating both conditions. IDA symptoms, especially extreme fatigue, can make it difficult to take part of daily activities, responsibilities, and social gatherings, leading many individuals to feel isolated.
The GI Society explained that common side effects of oral iron products are constipation, stomach upset, nausea, vomiting, dark stools, or teeth staining from liquid iron products. Some individuals are also unable to absorb or tolerate oral iron well for several reasons, such as time constraints from surgery, pregnancy, or childbirth; malabsorption; and intolerable side effects with oral iron. The GI Society added that, although many iron supplements are available over the counter, several have various marketing claims that may not necessarily be true, some may not have been subject to quality control regulations during manufacturing, and the efficacy claims of others may not have the clinical experience to support their efficacy claims. These products can also incur high out-of-pocket costs for patients. The GI Society believes that infusion-related side effects, such as staying an extra 30 minutes after an infusion to monitor for allergies or adverse reactions; staining of the skin or veins with iron (which can take 6 months or more to fade); Fishbane reaction, which is a pseudoallergy that includes flushing of the skin and can cause temporary (about 15 minutes) muscle contractions; taking time off school or work and arranging transportation to get to the clinic, are some of the disadvantages of IV iron administration. Moreover, some patients do not like the infusion process itself and the need for an IV insertion.
The GI Society stated that having alternative treatment options is crucial and that patients, their families, and health care professionals need access to treatments that have higher doses of iron, are better absorbed, and have fewer side effects.
Clinician Input
Input From the Clinical Expert Consulted by CDA-AMC
All CDA-AMC review teams include at least 1 clinical specialist with expertise regarding the diagnosis and management of the condition for which the drug is indicated. Clinical experts are a critical part of the review team and are involved in all phases of the review process (e.g., providing guidance on the development of the review protocol, assisting in the critical appraisal of clinical evidence, interpreting the clinical relevance of the results, and providing guidance on the potential place in therapy). The following input was provided by 1 clinical specialist with expertise in the diagnosis and management of IDA.
Unmet Needs
The clinical expert consulted for this review noted the key limitations of oral iron as tolerability (due to GI side effects) and adherence, as well as slow onset of effect (noteworthy for patients requiring rapid correction of their hemoglobin). The limitations of IV iron are its expense, and lack of coverage for those less than 65 years of age. These limitations are exacerbated when patients require urgent treatment (such as preoperatively or in pregnancy) as they do not have time to apply for additional coverage. The clinical expert noted that, because of these limitations, patients will access emergency departments for their IV iron. The clinical expert also noted that some patients may have a reaction to IV iron. Although they may try other IV formulations, their options are limited if the alternatives fail. The clinical expert noted that high-dose IV iron (1 g formulations instead of 300 mg with iron sucrose) is needed to reduce hospital visits for patients, and there is no approved therapy for IV iron replacement in pregnancy.
Place in Therapy
In the opinion of the clinical expert consulted for this review, ferric carboxymaltose would represent an alternative to ferric derisomaltose. The clinical expert believed that it would be used in the first line in situations where IV iron would be considered the first line, otherwise it would represent second-line therapy after failure to tolerate or respond to oral iron. The clinical expert did not describe ferric carboxymaltose as a drug that would shift the treatment paradigm for patients with IDA. The clinical expert noted that, for patients with iron deficiency or IDA, access to ferric carboxymaltose should be based on failure to tolerate or respond to oral iron after a 4-week trial, except in situations such as end-stage renal disease on dialysis, heart failure with reduced ejection fraction, moderate to severe anemia, and preoperatively in patients with iron deficiency and anemia, as these patients do not have time to respond to oral iron. The clinical expert agreed that patients with IBD should also have access to IV iron, per the European Crohn’s and Colitis Organization’s 2024 guidelines,35 when their disease is clinically active, there is previous intolerance to oral agents, or if ESAs are needed. The clinical expert also agreed that patients with chemotherapy-induced anemia are also among those most suited for treatment with ferric carboxymaltose, given that the National Comprehensive Cancer Network guidelines advocate the use of IV iron over oral iron in this patient population.16
Patient Population
According to the clinical expert consulted for this review, the patients most suited for treatment with ferric carboxymaltose would be those adults with iron deficiency or IDA (ferritin less than 30 mcg/L or TSAT less than 20%), with an alternate definition for those with CHF or end-stage renal disease, pediatric patients with IDA, those unable to tolerate or respond to oral iron and preoperative patients with IDA. According to the clinical expert, the least suitable patients would be those with a history of clinically significant hypophosphatemia (i.e., those patients who are symptomatic or require treatment for their hypophosphatemia). The clinical expert also noted that those most in need of intervention are those under the age of 65 who lack drug coverage, and pregnant patients.
The clinical expert noted there are no issues with diagnosing IDA, and that ferritin and TSAT are the 2 most informative tests to assess iron stores. The clinical expert went on to note that ferritin is the gold-standard laboratory test, with levels of less than 50 mcg/L representing iron insufficiency and less than 30 mcg/L highly suggestive (and thus considered diagnostic) of iron deficiency. Ferritin can be elevated in cases of inflammation; in those cases, a TSAT less than 20% is informative and used as a diagnosis.
Assessing the Response to Treatment
The most important outcomes for assessing response include a rise in hemoglobin, improvement in symptoms of iron deficiency and IDA, improved function, and reduced transfusion burden. The clinical expert believed that 4 weeks would be the minimum duration of follow-up (with 12 weeks as ideal) to assess the efficacy of iron supplementation. Follow-up in the included trials that reported efficacy data ranged between 4 and 12 weeks.
Discontinuing Treatment
According to the clinical expert, reasons for discontinuation include severe allergic reaction or anaphylaxis (once), and a different formulation should be considered in cases of minor allergic reactions. The clinical expert noted that in the population with non-CHF and non–end-stage renal disease a ferritin level greater than 50 mcg/L and a sustained TSAT of greater than 20% should be warrant discontinuation.
Prescribing Considerations
The clinical expert did not believe that a specialist would be required to order ferric carboxymaltose, adding that it should be administered in community, hospital, and private infusion clinics; emergency departments; and inpatient units by home care nurses.
Clinician Group Input
This section was prepared by the CDA-AMC review team based on the input provided by clinician groups. The full original clinician group input received by CDA-AMC is included in the Perspectives of Patients, Clinicians, and Drug Programs section of this report.
A group of 12 clinicians, including those with specialties in cardiology, obstetrics and gynecology, hematology, gastroenterology, and nephrology, as well as a primary care physician, were assembled to provide input.
The clinician group was in general agreement with the input provided by the clinical expert.
The clinician group agreed with the clinical expert that important unmet needs with IV iron supplementation include the ability to reduce visits to the hospital or infusion clinic and to have a product available that has evidence for use in pregnancy.
The clinician group did not elaborate on its experience with ferric carboxymaltose; however, it did note that the drug has been available in Europe for 20 years.
Drug Program Input
The drug programs provide input on each drug being reviewed through our Reimbursement Review processes by identifying issues that may affect their ability to implement a recommendation. The implementation questions and corresponding responses from the clinical experts consulted by the review team are summarized in Table 4.
Table 4: Summary of Drug Plan Input and Clinical Expert Response
Drug program implementation questions | Clinical expert response |
|---|---|
Relevant comparators | |
Ferric derisomaltose (Monoferric) and iron sucrose (Venofer) were considered comparators by the manufacturer. There were no direct-comparative studies in the submission comparing Ferinject to Monoferric. Monoferric was also compared to iron sucrose. Iron sodium ferric gluconate complex (Ferrliecit) is also a benefit in some jurisdictions. There were a number of clinical trials included in the submission materials. (54 completed interventional clinical studies.) | Comment from the drug programs to inform CDEC deliberations. |
Due to the need for access to IV infusion centres for administration of IV iron, funding for outpatients may vary between jurisdictions, or funding may be through special programs or provided through health authorities. | Comment from the drug programs to inform CDEC deliberations. |
Considerations for initiation of therapy | |
Definitions of IDA requiring IV iron vary. For example, in Saskatchewan, the definition of IDA in adult patients is hemoglobin < 130 g/L; and ferritin < 30 mcg/L or transferrin saturation < than 30% or blood loss > 1,000 mL within 7 days. The product monograph for Ferinject has dosing recommendations for patients with hemoglobin greater than 140 g/L. Question for clinical expert: Are there specific laboratory parameters thresholds that should be considered for initiation of IV iron? | In non-CKD, non-CHF, non-oncology populations (i.e., generic IDA) the definition of IDA is hemoglobin < 130 g/L (males) or < 120 g/L (females) and ferritin < 30 (although many clinicians will use ferritin < 50 mcg/L as there is evidence of normalized iron homeostasis when ferritin is > 50 mcg/L). Values for hemoglobin also vary in pregnancy (first and third trimester: < 110 g/L; second trimester: < 105 g/L) and in children (aged < 24 months: < 105 g/L; 24 to 59 months: < 110 g/L; 5 to 11 years: < 115 g/L). The clinical expert also added the following cutoffs for ferritin and TSAT, based on specific conditions, are:
If iron deficiency is confirmed with anemia, either oral or IV formulations are appropriate (it is a matter of tolerability, time to treatment, accessibility, or drug coverage). |
Ferinject is the only IV iron product indicated for pediatric patients. Question for CDEC: How are pediatric patients with IDA currently managed? If they cannot tolerate oral iron or need urgent iron replacement, what is the drug of choice for those patients? Ferinject also has an indication specific for Heart Failure (refer to alternate review). Question for CDEC: Are there patient populations who should not receive Ferinject? | According to the clinical expert, pediatric patients with IDA who cannot tolerate oral iron or need urgent iron replacement are currently treated using any of the existing IV iron formulations (FDI or iron sucrose), and there does not appear to be concerns using any of them. The clinical expert also stated that in their opinion there are no adult patient populations who should not receive Ferinject. |
Oral iron is a first-line treatment and inexpensive. Intolerance to oral iron can be difficult to determine or assess. Assessments of intolerance:
Question for CDEC: How do you define inadequate response to oral iron? What is an appropriate trial of oral iron? Are there populations in which oral iron is inappropriate (i.e., bariatric surgery, IBD, CKD, short bowel)? |
Populations in which oral iron is inappropriate: prior bariatric surgery, gastrectomy, IBD, CKD, prior small bowel resection, CHF, preoperative IDA (with 4 weeks or less to surgery), profound IDA (e.g., symptomatic anemia), and active malignancy. |
For patients on hemodialysis, Ferinject can be administered during a hemodialysis session. Question for CDEC: What are the benefits of Ferinject over alternatives such as iron sucrose in patients receiving hemodialysis? Note: The indication for Monoferric is for patients who have non–hemodialysis-dependent CKD, and in patients on hemodialysis the shorter infusion time is not seen as a benefit, as chair and nursing time are constant in this patient population. Note: The CDEC recommendation for Monoferric notes that it can be directly injected into the venous limb of a dialyzer. | There is a higher dose of iron infused at that session, but the clinical expert agreed there was no major benefit if patients are coming regularly for dialysis. |
Monoferric would occupy a similar therapeutic space; Ferinject is indicated in pediatric patients and HD patients. It also has a specific indication for iron deficiency associated with heart failure. | Comment from the drug programs to inform CDEC deliberations. |
Considerations for continuation or renewal of therapy | |
IDA should be assessed and diagnosed through laboratory tests. The dose of Ferinject is determined through a combination of weight and hemoglobin level. Hemoglobin level should be assessed no earlier than 4 weeks after IV iron infusion (to allow erythropoiesis and iron utilization). | Comment from the drug programs to inform CDEC deliberations. |
The reimbursement criteria of other injectable iron products (IDA indication) do not include renewal criteria. | Comment from the drug programs to inform CDEC deliberations. |
Considerations for prescribing of therapy | |
Doses range from 500 mg to 2,000 mg depending on body weight and hemoglobin level. The maximum dose is 1,000 mg per week; patients needing doses on the higher end (i.e., 2,000 mg) will require a second dose administered a minimum of 7 days from the first dose. Question for clinical expert: Approximately how many patients with IDA will require more than 1 infusion? | At any given time, a minority of patients will receive more than 1,000 mg, although it is frequent for individuals to require longstanding treatment (e.g., to need IV iron every 3 months). |
Ferinject is a high-dose IV iron preparation which can be administered in 15 minutes (compared to 30 minutes for Monoferric). Ferinject can be administered by injection or by IV infusion, or directly into the dialyzer. | Comment from the drug programs to inform CDEC deliberations. |
Care provision issues | |
The drug is administered intravenously, and patient monitoring is required for signs and symptoms of hypersensitivity reactions during administration and for at least 30 minutes for each administration. | Comment from the drug programs to inform CDEC deliberations. |
System and economic issues | |
Involvement of additional payers Proper management of IDA may lead to reduced requirements for blood products that are funded outside of the drug plan. | Comment from the drug programs to inform CDEC deliberations. |
Presence of confidential negotiated prices for comparators Monoferric was successfully negotiated through the pan-Canadian Pharmaceutical Alliance. | Comment from the drug programs to inform CDEC deliberations. |
Special programs or initiatives for the introduction and management of the drug(s) under review Anemia management programs may exist in certain jurisdictions. | Comment from the drug programs to inform CDEC deliberations. |
Other systems or economic issues: Savings from reduced chair time are likely not realized, but there is a potential for increased capacity (i.e., more patient access). | Comment from the drug programs to inform CDEC deliberations. |
CHF = chronic heart failure; CKD = chronic kidney disease; IBD = inflammatory bowel disease; IDA = iron deficiency anemia; pERC = pan-Canadian Oncology Drug Review Expert Review Committee; TSAT = transferrin saturation.
Clinical Evidence
The objective of the Clinical Review report is to review and critically appraise the clinical evidence submitted by the sponsor on the beneficial and harmful effects of ferric carboxymaltose 50 mg/mL IV infusion in the treatment of IDA in adult and pediatric patients aged 1 year and older when oral iron preparations are not tolerated or are ineffective. The focus is on comparing ferric carboxymaltose to relevant comparators and identifying gaps in the current evidence.
The sponsor conducted a systematic search and selection to identify studies relevant to the efficacy and harms of ferric carboxymaltose relative to relevant comparators in the Canadian setting. The sponsor identified 60 reports of 40 clinical trials, including studies among patients with iron deficiency and heart failure. However, heart failure is outside the scope of this report. The current report focuses only on select studies identified in the sponsor’s systematic review, with detailed summaries and appraisals of these studies. Appendix 1 contains additional supportive studies submitted to Health Canada (Table 34, Table 35, Table 36, Table 37 and Table 38), as well as studies of ferric carboxymaltose in pregnancy (Table 39).
A summary of the clinical evidence included by the sponsor in the review of ferric carboxymaltose is presented in 3 sections, with a critical appraisal of the evidence included at the end of each section. To inform the broad indication under review, the first section, Pivotal Studies and Select RCTs, includes the pivotal studies submitted to Health Canada for the indication under review and other RCTs from the sponsor’s systematic review that enrolled patients with IDA arising from various causes. The second section includes indirect evidence from the sponsor. The third section, studies addressing gaps, includes a study among pediatric patients to inform the use of ferric carboxymaltose among children and adolescents that was excluded from the sponsor’s systematic review due to an irrelevant comparator (oral iron); a study among adult patients with IDA arising from various causes to provide additional evidence for the broad indication that was excluded from the sponsor’s systematic review due to an irrelevant comparator (oral iron); and 5 studies comparing ferric carboxymaltose with ferric derisomaltose, as this was identified as a relevant comparator, but these 5 studies were not included in the Pivotal Studies and Select RCTs section.
Included Studies
Clinical evidence from the following sources are included in the CDA-AMC review and appraised in this document:
two pivotal studies and 2 other RCTs in patients with IDA arising from various causes
one indirect treatment comparison
seven additional studies addressing gaps in evidence.
Pivotal Trials and Selected RCTs
Contents within this section have been informed by materials submitted by the sponsor. The following have been summarized and validated by the CDA-AMC review team.
Description of Studies
Characteristics of the pivotal studies are summarized in Table 5. Characteristics of 2 additional studies identified in the sponsor’s systematic review are summarized in Table 6.
Table 5: Details of Pivotal Trials
Detail | VIT-IV-CL-015 | FER-IBD-07-COR (FERGIcor) | |
|---|---|---|---|
Designs and populations | |||
Study design | Phase III, open-label, multicentre, active-control RCT | Phase III, open-label, multicentre, active-control RCT | |
Locations | ██ █████ ██ █████ █████████ █████████ █████████ ███████ ████████ ███████ █████████ ████████ | 88 sites in 14 countries (Austria, Denmark, France, Germany, Norway, Romania, Spain, Sweden, Switzerland, UK, Estonia, Lithuania, Russia, Ukraine) | |
Patient enrolment dates | █████ █████ ████████ |███ █████ █████████ | Start date: October 28, 2008 End date: December 10, 2009 | |
Randomized (N) | 240 randomized FCM: N = 120 IV iron sucrose: N = 120 | 485 randomized FCM: N = 244 IV iron sucrose: N = 241 | |
Inclusion criteria | █████ ██ ██ ██ ███ ██ ███ ████████ ████████████████████████ ████ ████ ██████████ █████████ ██ ███████ ████████████ ███████ ██ ██ ████ ███ ███ ██ █████ ███ ██ ███ ███████████ ████ ██████ █████ ████████ ███ █████████████ ███████ ████ ███ ██ ████ ████████ ████ ████████ ███ ██ ███ █ ████ █████ ██ █████████ █ ███ ██████ ███ ███ ████ ███ ███ █████████████ █████ ███ █████ ██ ███ ████████ ██ ███ ████████████ ████████ ███ ████ ██ ███ ████ ██████████████████ ████████ ██████ ██████████ ███ ███████████████████████████████████ ██ ████████████ █████████ ████ ████████ █████ ████████ ████ ██ █████████ █████████ ██ ████████ ██ ██████████ ██████ ██ █████ ███████ █████ ███ █████ ███ ███ ██ ██ ███ █████ █████ ███ ████ ████ ██ ███ █████ ██████████ |
| |
Exclusion criteria | ████ ██████ ███ ██ █████ ███████████ ██ ████ ██ ██████████ ████ █████████ ██████ ██ ████ █████ ██ ██████████ ██ ███████████ ████ ███ █ █████ ███████████ ██████ ███ ████████████████ ████████████████ ██ ██████████ ██ ███ ██ ██ ████████ █████ ██ ██████ ████████████████ ██ █████ ████ ███████ ███████████████████ ███████ ███ ██ █████ ████ ███████████ ████████ ████ █████████ ███████ ███ ██████ ████ ████████ ██ ███ ██ ███ █████████ ████████ ███ ██████ ██ █████████ ██ ████████ ███████████ ██ ███████ ██ █████████ ██ █████ █████ ██ ███ ███████ ██ █████ ███████████ █████████ ██ ███████ ███ ██████████ ███ ███████ █████ ██ ███████████████████████ ████ ██ ███████████████ ████ ██████ ███ ██ ████ █████ ██ █████████████████ ██ █████████ ██ █████ ██ ███████ ███████ ███████████ ███ ██ ███ ██ █████ █████████████ ██ █████ ███████ ██ ████████████████████ ███████ ██ ████ ███ ██████████████ ██████ █████████ ██ ██████████ █████ ████ █████████ ██ ████ ██ ███ ██████ ███ ████████████ ████ █████████████ █████ ███████████████████ ██████████████ ████████████████ ███ ███████████ ██████████ █████████ █████ ██ ███████ ██████████████████████ ██ █████████ █████████ ████ ████ ███ ██████████ |
| |
Drugs | |||
Intervention | ███ █████ ████████ ███████ ████████ ██████████ ██ █████ ████ ██ ██ █████ █████████ ████████ ████ ███ ████████████ ██████ ████ █ ████ █████ ███ █████ ██ █ ████████ ████████ ████████████ ███ █████ ██████ | FCM, up to 3 infusions on days 1, 8, and 15 according to patient body weight (< 70 kg, ≥ 70 kg) and hemoglobin level (< 100 g/L, ≥ 100 g/L) (range = 1,000 mg to 2,000 mg total, with weekly doses not exceeding 1,000 mg); diluted in up to 250 mL of sterile 0.9% sodium chloride | |
Comparator | ██ ██ █████ ████████ ███████ ████████ ██████████ ██ █████ ████ ██ ██ █████████ ████ ██ ███████ ████████ ████ ███ ████████████ ██████ ████ █ ████ █████ ███ █████ ██ █ ████████ ████████ ████████████ ███ █████ ██████ | IV iron sucrose dosage individually calculated according to a modified Ganzoni formula; up to 11 infusions twice a week with a single dose not exceeding 200 mg given not more than 3 times a week; diluted in up to 200 mL of sterile 0.9% sodium chloride | |
Study duration | |||
Screening phase | | █████ | 2 weeks | |
Treatment phase | | █████████ | FCM: 2 weeks IV iron sucrose: 4 weeks | |
Follow-up phase | | █████████ | FCM: 10 weeks IV iron sucrose: 8 weeks | |
Outcomes | |||
Primary end point | ██████ ███ ██████████ ██ ████████ ████████ ██ ████████ ██ ██ ██ ███ ███ ██ █ █████ █████ ████████ | Number of patients responding as defined by an increase in hemoglobin of at least 20 g/L at week 12 as compared to baseline | |
Secondary and exploratory end points | ██████████████ ██████ ███ ██████████ ██ ████████ ████ ██ █████ ██ ██ ███ ██ ███ ███ █████████ ██████ ████████ ██████ ██████ █████ ███ ████ ███ ██ ████████ ████ █ ████████ ██ ████ ███ ██ ████ ███ ██ ████████ ████ █ ████████ ██ ████ ███ ██ ████ █████ █████ █████████ ███ ██ ███ ███████ █████ ███ ██ ████████████ ████████ ██ ███ ████████ ███ ████ ██████ █████ █████████████ ██████ ████ ████████ ██████ ██ ███ █████ █████████ ███ ████ ██ █████ ██ ██ ███ ██ ███ ███ █████████ █████████ ██████████ ███ ██ ██████ ████ ████████ ██████ ██ ███ █████ ████████ ███ █████ ████████████ ███ ███ ██ █████ █████████████████████ | Secondary: Proportion of patients who achieved either a hemoglobin increase ≥ 2 g/dL or hemoglobin gender specific normal range at week 12 Response was defined as follows:
Proportion of patients without anemia at week 12 Response was defined as follows:
Proportion of patients with a ferritin measurement > 100 mcg/L at week 12 Proportion of patients with a TSAT score of between 20% and 50% at week 12 Proportion of patients without anemia with a ferritin measurement > 100 mcg/L at week 12 Response defined as follows:
Hemoglobin, ferritin, and TSAT adjusted for baseline measurement at subsequent visits (week 1 to week 12) Health-related quality of life as measured by SF-36v2 adjusted for baseline score at week 12 IBDQ adjusted for baseline score at week 12 | |
Publication status | |||
Publications | No publications identified Vifor (International) Inc. VIT-IV-CL-015 - A Multi-Centre, Controlled Phase III Study to Compare the Efficacy And Safety Of VIT-45 And Venofer® in the Treatment of Iron Deficiency Anaemia Associated With Chronic Renal Failure in Patients On Haemodialysis. St. Gallen, Switzerland. 2005 | Evstatiev et al. (2011)36 EUCTR2008-002333-75-DE. A multi-centre, randomized, prospective, open-label study to investigate the efficacy and safety of a standardized correction dosage regimen of IV ferric carboxymaltose (FERINJECT®) versus iron sucrose (VENOFER®) for treatment of iron deficiency anaemia in patients with inflammatory bowel disease. 2008. | |
AUC = area under the curve; CVD = cardiovascular disease; EPO = erythropoietin; FCM = ferric carboxymaltose; IBD = inflammatory bowel disease; IBDQ = Inflammatory Bowel Disease Questionnaire; IDA = iron deficiency anemia; RCT = randomized control trial; SF-36v2 = Short-Form (36) Health Survey Version 2; TSAT = transferrin saturation.
Note: Three additional reports were included (Health Canada reviewers report and Clinical Study Reports for VIT-IV-CL-015 and FERGIcor).
Source: Sponsor’s Summary of Clinical Evidence.37
Table 6: Studies in Patients With IDA Arising From Various Causes
Detail | 1VIT05006 | VIT-IRON-2011-004 |
|---|---|---|
Designs and populations | ||
Study design | Phase III, double-blind, open-label, multicentre, active-control RCT | Phase III, open-label, multicentre, active-control RCT |
Locations | ██ ██████ ██████ ███████ ███████ █████████ | 19 sites: China |
Patient enrolment dates | █████ █████ █████████ |███ █████ █████████ | Start date: June 6, 2017 End date: February 25, 2019 |
Randomized (N) | ██████████ █████ ████████████████ ████████ ███ █████ | Randomized: N = 371 FCM: n = 188 IV iron sucrose: n = 183 |
Inclusion criteria | ████████ ████ ██ ██ █████ ███ █████ ██ ███ ██████ ██ ████ ████████ █████████████ ██ ████ ████████ █ █████ █████ ██ ███ █████████ ████████████ █████ ██ ████ ███████████ ████ ███ ██ ██████████████████ █████████ ███ ████████ ██ ████████ ██ ███ ███ █ ███ █████ █████████████ ██████████ ████ ██████████ ████████ ████ ██ █████████ ██ █████ █████████████ ███ ██████████ ███████ ███ ███ █████████ ██ ████████████████ ████ █████ |
|
Exclusion criteria | █████ ████████████ ████████ ███████████████ ████████ ███████████ ████ ██ ███ █ ████ █████ ██ █████████████ ██ ███████ ████ ███████████████████████ ██████████ ██████████ ████████████ ████ ████████ ██ ███ █████ █████████████ ███████ ██████ █████████ ████████████ █████ ██████████ ██████ ██ ███ █████ ██ ███ ██████████ ████ █████████████ █████████████████ ██ ███ ███████ ████ █████ █████ ██ ████████████████ █████ ██ ███ ████████ ████ ████████ ██ █████ ██████████████ ████████ ██████████ █████████ ███████████████████ █████ ███████ ███ ███████ ████ ██████████ ████████████████████ ████ ███ ███████ ██ ████████ ██ ████████ ██████ ███ ████████████ ███████ ██ ████ █████ ███ ███ ████ ██ ███████████████████ ██ ███████████████ ████████ ███ ██████████ ██ ███████████████ ████████████ ███ ████ ██ █████████ █████████ ███ ███ ████ ███ █████ █████ ██ ███████ ███████ ██ █████ █████ ██████ ████ ██ ██████████ ███████████████ █████████ ████ ███████████ █████████ |
|
Drugs | ||
Intervention | ███ ██ █████ ██ ██ █ ███████ ██ ████ ██ ██ ██████ ███████ █████ ██ ███ ██ █████ ██ ████ ████████████ ██ ███ ██ ██████ ██████ ███ █████ ██ ███ ██ ███ ██ ████ ████████████ ██ ███ ██ ██████ ███████ | FCM: dosing was determined based on the patient’s hemoglobin and body weight at screening and was administered as undiluted injections or diluted infusions; for the 500 mg dose, FCM was diluted in 100 mL saline and administered over at least 5 minutes, For the 1,000 mg dose, FCM was diluted in 250 mL saline and administered over at least 15 minutes |
Comparator | ███████ ██ ██ ███ ██ ██ ██████ ██████ ████ ██ ████████ ███ ██████ ███ ███ ███ ██ ██ ██████ ██████ ███ ███ ██████ ███ ███ ███ ██ ██ ██████ ███████ █ ███ ███ ██ ████████ ████ ███████ ████ ██ ███████ ██████ ███████ ██ ███ ███████████ ████ ██████ ██ ████ | Iron sucrose: dosing regimen was determined based on the individual iron deficit calculated using the Ganzoni formula; administered as single 200 mg iron doses, with a maximum of 3 doses per week and up to a total of 11 injections; administered either via slow IV push injection at a rate of 1 mL undiluted solution per minute (not exceeding 60 minutes) or as a drip infusion of diluted product (with a maximum of 10 mL per 200 mL saline) for at least 30 minutes (not exceeding 60 minutes) N = 183 |
Study duration | ||
Screening phase | ██ ██ █ ████ | Up to 1 week |
Treatment phase | | █████ | 8 weeks |
Follow-up phase | ██ | Within 4 days of week 8 |
Outcomes | ||
Primary end point | ███████████████████████ | Percentage of patients who achieved an increase in hemoglobin of ≥ 2 g/dL from baseline at any visit up to week 8: |
Secondary and exploratory end points | ████ |
|
Publication status | ||
Publications | No publications identified Luitpold Pharmaceuticals I. 1VIT05006 - A Multicenter, Randomized, Blinded, Placebo-Controlled, Cross-Over Study to Investigate the Safety and Tolerability of Intravenous VIT-45 in Patients with Iron Deficiency Anemia. Norristown, PA, USA2006. | Jin, et al. (2023)38 NCT03591406. To Assess the Impact of Ferric Carboxymaltose Compared with Iron Sucrose in Chinese Subjects on Correcting Iron Deficiency Anaemia. 2018 |
ALT = alanine transaminase; AST = aspartate transaminase; CO = crossover; CVD = cardiovascular disease; FCM = ferric carboxymaltose; HbAg = hepatitis B surface antigen; HBV = hepatitis B virus; HCV = hepatitis C virus; hsCRP = high-sensitivity C-reactive protein; IBDQ = Inflammatory Bowel Disease Questionnaire; IDA = iron deficiency anemia; NDD-CKD = non-dialysis dependent chronic kidney disease; NR = not reported; QoL = quality of life; RCT = randomized control trial; SF-36 = Short-Form (36) Health Survey; TSAT = transferrin saturation.
Note: Three additional reports were included (Health Canada reviewers report and Clinical Study Reports for 1VIT05006 and VIT-IRON-2011-004).
Source: Details included in the table are from the Clinical Study Reports for 1VIT0500639 and VIT-IRON-2011-004.40
Study VIT-IV-CL-015 is a pivotal phase III, open-label, active-controlled, multicentre randomized trial that enrolled 240 patients with IDA in hemodialysis or hemodiafiltration with IDA secondary to chronic renal failure. ████████ ████ ████████ ███████ ████ ██ █████ ███ ███ ██ ████ ████ ██ █████ ██ █████ ████████ ██████████ ████████ ████ ████████ ███ ███████████ ████ █ ██████ ███████ ███ ████████ ████ ██████████ ██ ██████ ██████ ██████████████ █ ███████ ██ ██ ████ ███████ █████ ████████ ████████ ████ ████████ ███ ███████ ████ ████████ ████ ███████ ██ █████ ████████ ██ █████ ██ ██ ███ ██ ███ ████ ████ ████████ ██ █████ █ █████ █████ ███ ███ ██ █████ ██████████ ████████ ████ ████████ ████████ ██ ███████ ██████ ██████████████ ██ ██ ████ ███████ ██ █ ███ ██████ ██████████ █████████ ██ ███████ ███ ████████ ██ ██████ ██ █████████ █████ ███ ██ ████ ███ ██ ████ █████ █████████████ ███ ███████ ███ █████ ██ ███████████ █████ ███████████ ███████ ███ ███████ █████████ ██ ████ █████ ███ ██ ███████ ███ ████████ ██ ██████ ██████████████ ██████████ ███ ████ ███████ ██████████ ██ ████████ ██ ████████████ ████ ███ ██████████ ████ ███████ █████ ████████ ███ ███ █████████ █████████ ███ ██ ███████ ███ ██████ ██ ██████ ██████████████ ██████████ ███ ████ ███████ ██████████ ██ ████ ███████ ███████████
Study FER-IBD-07-COR (FERGIcor) is a pivotal phase III, open-label, active-controlled, multicentre randomized trial of 485 patients with IDA and IBD. Patients were enrolled in this trial between October 28, 2008, and December 10, 2009, from 88 centres in 14 European countries. Randomization was based on a 1:1 ratio and was conducted using a predefined, computer-generated randomization list without stratification. After a 14 day-screening period during which study eligibility was assessed, patients were randomly assigned to either ferric carboxymaltose (n = 244) or IV iron sucrose (n = 241). Patients returned for safety and efficacy assessments at weeks 4, 8 and 12. Patients who were not anemic at week 12 were invited to continue to participate in the maintenance study. The primary objective of this study was to evaluate the noninferiority in efficacy of the standardized dosage regimen of ferric carboxymaltose compared to individually calculated dosage regimens of iron sucrose in the correction of IDA in patients with IBD in remission. The secondary objective was to evaluate the safety and tolerability of the standardized correction dose regimen of ferric carboxymaltose.
Study 1VIT05006 was a phase III, randomized, blinded, placebo-controlled crossover study that compared the safety and tolerability of ferric carboxymaltose versus placebo in patients who have IDA. ████████ ████ ████████ ██ ████ █████ ████ ██ █████ ██ ███ ██████ ███████ ██████ ███ ██████████ █████████ ████ ███ █████ ███ ███ ████ ███████ █████████ ██ ████████ ███ █████ █████ █ █████████ ██████ ██ ██ ██ █ █████ █████████████ ██ ███ ███ ████████ ███ ███████ ███ █████ ██ ███████████ █████ ████████ ██████ █████ ██ █ ███ █████ ████ ██████████████ ██ ███████ █████ ████ █ ███████ ██ ███ ██████ ██████ █ ███ ███ █████████ ██ █████ ███████ ██████ ██ ███ ███ █████████ ██ ███ ███ ██████ ███ ███ ███ █████████ ██ █████ ███████████ ████████ ████████ █ ██████ ███████ ████ ██ ██████ ██████ ███████████████ ██ █████ ████████ ████ ███ ██ ███████ █████████████ ████ ██ ███████ ██ ███ ██ ███ ██ ███ █ ████ ███████ ████ ██ ███████ ███ █████ █████████████ ███ ███ █████ ████ ██ █████ █████ ██ ████ ████████ ████ ███ █ ██ ███████████ █████ ██ ███ ██████ ████ ██ █████ ████ ██ ███ ██ ███ ███ ███ ██████ █████ ██ ████ ████████ ████ ███ █ ██████ ████ ██ █████ ████ ██ ███ ███ █████████ ██ ███ ████████ ███ █████ █████████ ██████ ███ █████████ ██ ███ ███████████████ ███████ ██ ███ ████ ██ █████ █████ ████████████ ████ ███ ████████ ██ ██████ ██████████████ ████ ███████ ██ ████████ ██ ████████ ██ ██ █████ ██████████
Study VIT-IRON-2011-004 was a phase III, randomized, open-label active-controlled trial that tested the noninferiority of ferric carboxymaltose versus iron sucrose in Chinese patients with various conditions that result in IDA. A total of 371 patients were randomized at 19 sites in China beginning June 6, 2017, to either ferric carboxymaltose (n = 188) or iron sucrose (n = 183). Randomization was conducted using a predefined, computer-generated randomization list, using interactive response technology and was stratified by hemoglobin level (< 100 g/L versus ≥ 100 g/L). This was a 10-week study, with a screening period of up to 1 week, and a 9-week treatment period, which included follow-up. Ferric carboxymaltose was administered on day 1 and if needed, days 8 and 15, up to a single dose of 1,000 mg iron, while the iron sucrose dose was calculated individually using the Ganzoni formula, up to a maximum single dose of 200 mg iron 3 times a week.
Populations
Inclusion and Exclusion Criteria
The 2 pivotal studies included adult patients with IDA secondary to specific conditions, chronic renal failure (Study VIT-IV-CL-015) and IBD (the FERGIcor study). The other 2 studies were chosen for this section because they included patients with IDA due to a variety of causes and as such could inform the broad indication under review. Study 1VIT05006 there were 3 cohorts, a CKD, 2 with IBD, and cohort 3 included patients with IDA secondary to other conditions. Patients in Study VIT-IRON-2011-004 simply had to have microcytic hypochromic anemia due to any cause. Each study used different laboratory parameter cutoffs for hemoglobin from 70 g/L to 120 g/L (females) or 130 g/L (males) in the FERGIcor study, to 115 g/L or less in Study VIT-IV-CL-015, 120 g/L or less in Study 1VIT05006 or less than 110 g/L (females) or 120 g/L (males) in Study VIT-IRON-2011-004.
Interventions
VIT-IV-CL-015
The administration of ferric carboxymaltose and iron sucrose was conducted during patients’ scheduled hemodialysis sessions by medical personnel during the trial period.
███ ██████ ████ ██ ███ ████████████ ███ ██ █████████████ █████ ████ ██████████ ██ ██ ████ ███ ██ ██ ███████ ████████ ██ ██████ ██████████████ ██ █████ ███ ██████████ ██████ ██████████████ ███ ████████ ██ █ ██ ████████ ██████████ ███ ██ ██ ████ ███ ████████ ██ ███ █████████ ██ ██ █████ █████████ ████████ ████ ███ ████████████ ██████ ████ █ ████ █████ ███ █████ ██ █ ████████ ████████
███ ██████ ████ ██ ███ ██████████ ███ ██ ███ ████ ██████████ ██ ██ ████ ███ ███ ██ ███████ ████████ ██ ████ ███████ ██ █████ ███ ██████████ ████ ███████ ███ ████████ ██ █ ██ ████████ ██████████ ███ ██ ██ ████ ███ ████████ ██ ███ █████████ ██ ██ █████████ ████ ██ ███████ ████████ ████ ███ ████████████ ██████ ████ █ ████ █████ ███ █████ ██ █ ████████ ████████ ███ █████ ████ ███ ████████████ ████ █ ███████ ██ ██ ███ ███ ██ ████████████ ███████ ██ ███ ██ ████ █ ███ ████ █████████ ████ ███ █████████ ███ ██ █████ █████ ██████ ██ ████████ █████ ███ ██████████ ██████████ ██████████ ████ ██████ ██ ███ ███████ ████████ ███ ████████ ██ ████ ██████ ████████ ████████ █ ████ ██ ███ ██ ██ ████ ██ ██████ ██████████████ ██ ████ ████████ ██████ ███ ███ █████ ████ █████ ███ ████ ████ ███ ██ ████ ██ ██████ ██████████████ ██ ████ ███████ █████████ ██ ███ ██████████ ████ ████████ ██████████████ ███████████ ██████ ██████ ████ ███████ ██ ██████████████
Study FERGIcor
In FERGIcor, ferric carboxymaltose and iron sucrose were administered intravenously to patients by study personnel during study follow-up visits.
The dosage form of the intervention was 5% weight by volume iron (50 mg/L), as a sterile solution of ferric carboxymaltose in water for injection. Drip infusion ferric carboxymaltose was diluted in sterile 0.9% sodium chloride. The intervention was packaged in 10 mL vials containing 500 mg of iron per vial. The ferric carboxymaltose dose was selected in accordance with the results of a randomized, controlled study in patients with IBD41 and was calculated using a simplified dosing matrix based on baseline hemoglobin (below or above 10 g/dL) and patients’ weight (below or above 70 kg). Using this dosing matrix, patients received a total of 1,000 mg, 1,500 mg, or 2,000 mg IV iron, split into up to 3 infusions (on days 1, 8 and 15). The total weekly dose was not to exceed 1,000 mg for any patient, regardless of their body weight.
The form of the iron sucrose was similar to that described previously. The dosage of iron sucrose was selected for each patient based on the individually calculated iron dose following the Ganzoni formula. Patients received iron sucrose infusions twice a week for a maximum of 11 infusions, depending on their calculated iron deficit. A single dose was not to exceed 200 mg of iron.
Study 1VIT05006
██ ███ ██ ████████ ████████ ██ ██████ ██████████████ ████████ █ ███████ ████ ██ █████ ██ ██ ████ ██ ██████ ███ █████ ███ ██████ ██ ██ ███ ████ ██ ███████ ██████████████ █████ ██ ███ ████ ██████ ████ ███ █████████ ██████ █████ ██ ███ ██ █████ ██ ████ ████████████ ██ ███ ██ ██████ ██████ ████ ██ ███████ ███ █████ ██ ███ ██ ███ ██ ████ ████████████ ██ ███ ██ ██████ ██████ ████ ██ ████████ ████████ ████████ ██ ███████ ████ ████ █████ █████ ██ ██████ ████ ██████ ███ ███ ███ ██ ██ ██████ ██████ ███ ███ ██████ ██ ██ ██ █████ ███ ██ ██ ██████ ███████ ████ ██ ███████ ██████████████ ██ ███ ██ ████████ ███████ ████ ██ ███ █████ █████ ███ ████████ ██████ ██████████████ ██ ███████ ██ ██████ ████ ████ ███████ ██ ██████████
Study VIT-IRON-2011-004
In the ferric carboxymaltose group, study drug administration occurred on day 1 and, if needed, on days 8 and 15. Patients randomized to iron sucrose received injections and/or infusions of a maximum of 200 mg of iron 3 times a week until the calculated total iron dose was administered. For both injection and infusion, ferric carboxymaltose was to be administered up to a maximum single dose of 1,000 mg iron (up to a maximum of 20 mg/kg every 2 weeks) over at least 5 minutes for the 500 mg dose and over at least 15 minutes for the 1,000 mg dose. Administration time was not to exceed 30 minutes for either the 500 or the 1,000 mg dose. Iron sucrose was administered by slow IV push injection at a rate of 1 mL of undiluted solution per minute (10 mL of the product in at least 10 minutes; administration time should not exceed 60 minutes) or by drip infusion in a dilution of 1 mL of iron sucrose in maximum 20 mL (0.9% weight by volume) physiological saline (10 mL of the product in maximum 200 mL saline). The drip infusion rate was 200 mL solution in at least 30 minutes and should not exceed 60 minutes.
Other forms of oral or IV iron were prohibited, as were ESAs and blood transfusions. If patients required ESAs or a transfusion for symptomatic anemia, they were withdrawn from the study.
Outcomes
A list of efficacy end points assessed in this Clinical Review is provided in Table 7, followed by descriptions of the outcome measures. Summarized end points are based on outcomes included in the sponsor’s Summary of Clinical Evidence as well as any outcomes identified as important to this review by the clinical expert consulted by CDA-AMC and input from patient and clinician groups and public drug plans. Using the same considerations, the CDA-AMC review team selected end points considered to be most relevant to inform expert committee deliberations and finalized this list of end points in consultation with members of the expert committee. HRQoL was prioritized because of the impact that IDA has on patient quality of life, and all instruments used were reported. Of the various laboratory markers reported, hemoglobin, ferritin, and TSAT were identified as most relevant by the clinical expert. Of these 3 parameters, hemoglobin was considered to be most important by the clinical expert, and it was therefore reported as both a binary outcome (patients who responded) and change from baseline, while ferritin was reported as change from baseline and TSAT was reported as a binary outcome, based on input from the clinical expert. Regarding time points, the clinical expert agreed that it was best to report the latest where available, noting that some laboratory parameters such as ferritin peak immediately after infusion and then decline gradually over time.
In the absence of a complete body of evidence for any patient population or comparison, the certainty of evidence was not assessed by the CDA-AMC review team for any outcome.
Table 7: Outcomes Summarized From Pivotal Studies and Select RCTs
Outcome measure | Time point | VIT-IV-CL-015 | FERGIcor | VIT-IRON-2011-004 | 1VIT05006 |
|---|---|---|---|---|---|
Hospitalizations for anemia | Any | Not assessed | Not assessed | Not assessed | Not assessed |
HRQoL-SF36 mental component summary — mean change from baseline | 12 weeks | Not assessed | Secondary | Not assessed | Not assessed |
HRQoL-SF36 physical component summary — mean change from baseline | 12 weeks | Not assessed | Secondary | Not assessed | Not assessed |
HRQoL-IBDQ — mean change from baseline | 12 weeks | Not assessed | Secondary | Not assessed | Not assessed |
Number and percentage of patients reaching an increase in hemoglobin of ≥ 10 g/L at 4 weeks after baseline | 4 weeks | Primary | Not assessed | Not assessed | Not assessed |
Number of patients responding, as defined by an increase in hemoglobin of at least 2 g/dL at week 12 compared to baseline | 12 weeks | Not assessed | Primary | Not assessed | Not assessed |
Patients who achieved an increase in hemoglobin of ≥ 2 g/dL from baseline at any visit up to week 8 | 8 weeks | Not assessed | Not assessed | Primary | Not assessed |
Proportion of patients without anemia at week 12 | 12 weeks | Not assessed | Secondary | Not assessed | Not assessed |
Serum ferritin — mean change from baseline | 4 weeks | Secondary | Not assessed | Not assessed | Not assessed |
12 weeks | Not assessed | Secondary | Not assessed | Not assessed | |
8 weeks | Not assessed | Not assessed | Secondary | Not assessed | |
Patients with a normal TSAT level (20% to 50%) | 4 weeks | Secondary | Not assessed | Not assessed | Not assessed |
12 weeks | Not assessed | Secondary | Not assessed | Not assessed | |
Notable harms —hypophosphatemia | End of treatment | Safety and harms | Safety and harms | Safety and harms | Safety and harms |
Notable harms — infection | End of treatment | Safety and harms | Safety and harms | Safety and harms | Safety and harms |
Notable harms — allergic reactions | End of treatment | Safety and harms | Safety and harms | Safety and harms | Safety and harms |
Notable harms — rash | End of treatment | Safety and harms | Safety and harms | Safety and harms | Safety and harms |
Notable harms — changes in blood pressure | End of treatment | Safety and harms | Safety and harms | Safety and harms | Safety and harms |
HRQoL = health-related quality of life; IBDQ = Intestinal Bowel Disease Questionnaire; SF-36 = Short-Form (36) Health Survey; TSAT = transferrin saturation.
Sources: Sponsor’s Summary of Clinical Evidence37 and Clinical Study Reports for VIT-IV-CL-015,42 FERGIcor,43 1VIT05006,39 and VIT-IRON-2011-004.40
Hemoglobin
Hemoglobin is a protein in red blood cells that delivers oxygen to tissues, and low levels of hemoglobin indicate anemia. In IDA, therapy consists of the of the iron deficit and treatment of the underlying disease; the aim of treatment is to return both hemoglobin and iron stores to target levels. The WHO criteria for anemia are hemoglobin levels of less than 110 g/L in children aged 6 to 59 months and in pregnant patients, less than 115 g/L in children aged 5 to 11 years, less than 105 g/L for patients in the second trimester of a pregnancy, less than 120 g/L in children aged 12 to14 years and in patients who are not pregnant, and less than 130 g/L in men.44 Differences in hemoglobin of 20 g/L or greater are considered clinically important in various conditions, although differences as small as 1 g/dL have been reported to have significant clinical impacts in patients with CKD and heart failure.45-47 In Study VIT-IV-CL-015, the primary end point was an assessment of the percentage of patients to reach the target increase (≥ 10 g/L or ≥ 1 g/dL) in hemoglobin in 4 weeks. In the FERGIcor study, the primary end point was the number of patients responding with a minimum increase of 20 g/L at week 12 compared with baseline. Also in the FERGIcor study, a hemoglobin level greater than 160 g/L was indicative of iron overload. In Study VIT-IRON-2011-004, the primary outcome was the percentage of patients achieving an increase in hemoglobin of at least 20 g/L from baseline.
In the FERGIcor study, the number of patients who were nonanemic at week 12 was also reported as a secondary outcome. Nonanemic was defined as achieving a hemoglobin level of at least 120 g/L in females and at least 130 g/L in males by week 12.
Inflammatory Bowel Disease Questionnaire
The IBDQ was a secondary outcome measure in the FERGIcor study. The IBDQ is a validated and reliable disease-specific tool to measure HRQoL in adult patients with IBD, UC, or Crohn disease.48 It consists of 32 questions scored in 4 domains: bowel symptoms (e.g., loose stools, abdominal pain), emotional health (e.g., anger, depression, irritability), systemic function (e.g., fatigue, sleep pattern), and social function (e.g., ability to attend work and social events). The response for each question ranges from 1 to 7, with 1 corresponding to significant impairment and 7 corresponding to no impairment. The overall IBDQ score is the sum of the responses to each of the questions. A total IBDQ score can range from 32 (very poor HRQoL) to 224 (perfect HRQoL). The questionnaire can be interviewer-administered or self-administered and involves a recall period of the last 2 weeks. Table 8 provides more details about the validity, reliability, and responsiveness of this scale.
Short-Form (36) Health Survey
Version 2 of the SF-36 was used as a secondary outcome measure in the FERGIcor study. This generic quality-of-life instrument is considered a standard instrument for assessing patient-based health care outcomes and was used because of the positive relationship reported between hemoglobin and all but 2 of the survey’s domains.49 The SF-36 was developed to measure generic health concepts relevant across age, disease, and treatment groups.50 The SF-36 measures 8 multi-item dimensions of health: physical functioning (10 items), social functioning (2 items), role limitations due to physical problems (4 items), role limitations due to emotional problems (3 items), mental health (5 items), energy and/or vitality (4 items), pain (2 items) and general health perception (5 items). There is a further unscaled single item asking respondents about health changes over the past year. The score of the SF-36 ranges from 0 to 100, with higher scores indicating better health. Table 8 provides for more details about the validity, reliability, and responsiveness of this scale.
Serum Ferritin
Ferritin is an iron-storage protein whereas serum ferritin is an indicator of iron status.51 In the presence of inflammation (i.e., elevated C-reactive protein), serum ferritin levels of less than 100 mcg/L are indicative of IDA52 and, according to the clinical expert, less than 30 mcg/L in the noninflamed population. Target levels of serum ferritin in the VIT-IV-CL-015 study were 200 mcg/L to 800 mcg/L. In both the VIT-IV-CL-015 and FERGIcor studies, serum ferritin levels greater than 800 mcg/L were also indicative of iron overload. No minimal important difference for serum ferritin was identified in the literature. The change in serum ferritin from baseline at weeks 2, 4, 6, and 8 was assessed as a secondary outcome in the VIT-IRON-2011-004 study, and change from baseline at weeks 1, 2, and 4 was assessed as a secondary outcome in the VIT-IRON-2011-004 study. The change from baseline in ferritin was also assessed as a secondary outcome in the FERGIcor study, adjusted for baseline measurement at subsequent visits (week 1 to 12).
Transferrin Saturation
TSAT is the ratio of serum iron concentration to the total iron-binding capacity expressed as a percentage, and is an indicator of iron deficiency. TSAT levels of less than 20% are a diagnostic criterion for IDA.52 In the VIT-IV-CL-015 and FERGIcor studies, TSAT outcome target levels were between 20% and 50%, assessed as a secondary outcome. In the VIT-IV-CL-015 and FERGIcor studies, TSAT values greater than 50% were indicative of iron overload. In the VIT-IRON-2011-004 study, only the change from baseline in TSAT, not patients with a TSAT responds, was assessed. No minimal importance difference for TSAT was identified in the literature.
Table 8: Summary of Outcome Measures and Their Measurement Properties
Outcome measure | Type | Conclusions about measurement properties | Minimal important difference |
|---|---|---|---|
IBDQ | The 32-item interviewer- or self-administered IBDQ is the most frequently used instrument to capture disease-specific quality of life in randomized clinical trials for IBD. It consists of 32 items classified into 4 domains of functioning and well-being: bowel symptoms (10 items), systemic symptoms (5 items), emotional function (12 items), and social function (5 items).48,53 Total scores range from 32 to 224, with higher scores representing a better quality of life. | The IBDQ has been validated in a variety of settings, countries, and languages.54-57 Validity
Reliability
Responsiveness
| A point change in actual score of ≥ 30, or an improvement of ≥ 15 points above the placebo score is associated with clinical benefits in IBD patients, including those with UC. |
SF-36 physical component summary and mental component summary | The SF-36 PCS is based on the weighted T scores of the 4 physical SF-36 scales: physical functioning, role limitations due to physical health problems, pain, and general health perceptions. It is measured on a scale of 0 to 100, with an increase in score indicating improvement in health status.59 | Validity
A study in patients with IBD assessing psychometric properties of the SF-36 showed the following results:61 Reliability
Measures of improvement or deterioration were generally acceptable for most groups except for role-physical in patients with UC and physical functioning and role-physical in patients with Crohn disease. Yarlas et al. (2018) conducted a systematic literature review of the reliability, construct validity, and responsiveness of the SF-36 in patients with UC and came to the conclusion that the SF-36 is reliable, valid, and responsive when used with patients with UC, supporting the inclusion of the SF-36 as an end point in clinical trials for this patient population.61 | An absolute score increases of 3 to 5 points in the norm-based scores (mean score of 50 ± 10 in the general population) are considered clinically important.62 |
CDAI = Crohn Disease Activity Index; CKD = chronic kidney disease; FACT-An = Functional Assessment of Cancer Therapy–Anemia; IBD = inflammatory bowel disease; IBDQ = Inflammatory Bowel Disease Questionnaire; SF-36 = Short-Form (36) Health Survey; UC = ulcerative colitis.
Statistical Analysis
Sample Size and Power Calculation
Study VIT-IV-CL-015
The sample size was chosen by the sponsor based on feasibility rather than statistical arguments. The trial was not aimed to prove a specific statistical hypothesis. Results were analyzed primarily to provide estimates and descriptive statistics. The investigators decided that a total of 240 patients would be randomized (120 patients in the ferric carboxymaltose group and 120 patients in the iron sucrose group).
Study FERGIcor
Sample sizes of 183 in both the ferric carboxymaltose and iron sucrose groups provided the basis for achieving 90% power (beta = 0.10) at a 2.5% significance level by using a 1-sided equivalence test of proportions when the proportion for the ferric carboxymaltose group with standardized correction dosage regimen was 0.82 and the proportion in the iron sucrose group being tested for equivalence was 0.75. The maximum allowable difference between these proportions that would indicate they were equivalent (the range of equivalence) was 0.07. No justification was provided for this noninferiority margin. Assuming a dropout rate of 15%, a total of 420 patients were needed to be randomized to ensure 366 patients completed the study.
Study 1VIT5006
No formal sample size calculations were performed. The sponsor noted that “the statistical precision of estimated parameters (as measured by CI width) will be adequate with the planned sample size”; however, no further justification was provided.
Study VIT-IRON-2011-004
The planned sample size per study protocol was determined to be a total of 368 patients (184 per treatment group), to ensure that at least 147 patients are available for the primary analysis. The sample size was based on showing noninferiority in the difference in the proportion of patients achieving an increase in hemoglobin of 2 g/dL or more at any time up to week 8 between ferric carboxymaltose and iron sucrose. Using a 1-sided alpha level of 2.5%, and a −15% noninferiority margin, it was estimated that 147 patients per group would have an 80% power to detect whether ferric carboxymaltose is noninferior to iron sucrose with an expected proportion of patients in the iron sucrose group with a response of 70%. No justification was provided for the noninferiority margin. The total planned sample size was 368, which allowed for an estimated dropout rate of 20% of the randomized patients.
Statistical Testing and Multiple-Testing Procedure
The methods for statistical testing of relevant efficacy end points, including methods for imputing missing data and for sensitivity analyses in the included trials are listed in Table 9. There was no mention of multiple testing procedures in any of the trials.
Subgroup Analyses
In the FERGIcor study, a subgroup analysis was performed on patients with a hemoglobin response (patients with an increase from baseline of at least 20 g/L), looking at those who had a baseline hemoglobin of at least 70 g/L and less than 100 g/L, or patients with a baseline hemoglobin of at least 100 g/L and less than 120 g/L. This was a descriptive analysis with no formal comparisons planned.
In the 1VIT05006 study, no formal efficacy analyses were planned; however, for safety analyses, subgroups were summarized for any subgroup that included at least 30 patients, including different types of CKD (hemodialysis-dependent, peritoneal dialysis–dependent, and non–dialysis-dependent) and postpartum anemia and CKD.
No subgroup analyses were performed in Study VIV-IV-CL-015 study or in Study VIT-IRON-2011-004.
Secondary Outcomes of the Studies
Table 9 describes the methods used in the statistical analysis of the efficacy end points in the VIT-IV-CL-015, FERGIcor, and VIT-IRON-2011-004 studies.
Table 9: Statistical Analysis of Efficacy End Points
End point | Statistical model | Adjustment factors | Handling of missing data | Sensitivity analyses |
|---|---|---|---|---|
VIT-IV-CL-015 | ||||
Percentage of patients to reach an increase in hemoglobin of ≥ 10 g/L increase at week 4 | Tabulated per treatment group together with its 95% CI, compared with 2-sided chi-square test | NA | Missing values were not replaced | Exploratory analyses to compare the 2 treatment groups were performed by means of the Cochran-Mantel-Haenszel test allowing for severity of anemia (≤ 100 g/L vs. > 100 g/L to ≤ 115 g/L) and country |
Change from baseline in hemoglobin, ferritin, and TSAT at weeks 1, 2, 4, and follow-up | ANCOVA | NR | Missing values were not replaced | NA |
FERGIcor | ||||
Number of patients with a response as defined by an increase in hemoglobin of ≥ 2 g/dL at week 12 as compared to baseline | One-sided 97.5% CI evaluated by a noninferiority margin of 7%. | NA | The LOCF algorithm was used as appropriate, although only where at least 1 postbaseline score was present (if all assessment postbaseline visits were missing, the LOCF algorithm was not applied) If a patient has a missing response at week 12 then the response is taken to be nonresponse | NA |
Proportion of patients with a TSAT between 20% and 50% at week 12 | Logistic regression analysis | Treatment, sex, and disease status at baseline as explanatory variables | NR | NR |
Proportion of patients without anemia at week 12 | Logistic regression analysis | Treatment, sex, and disease status at baseline as explanatory variables | NR | NR |
Hemoglobin adjusted for baseline measurement at subsequent visits (week 1 to week 12) | ANCOVA | Treatment, sex, country or region, baseline score | NR | NR |
Serum ferritin adjusted for baseline measurement at subsequent visits (week 1 to week 12) | Repeated measures analysis of covariance | Sex, baseline hemoglobin, and baseline disease status as explanatory variables | NR | NR |
HRQoL measured by the SF-36 adjusted for baseline score at week 12 | ANCOVA | Treatment, sex, and disease status at baseline | NR | NR |
Disease-specific HRQoL measured by the IBDQ adjusted for baseline score at week 12 | ANCOVA | Treatment, sex, and disease status at baseline | NR | NR |
VIT-IRON-2011-004 | ||||
Patients with ≥ 2 g/dL increase in hemoglobin at any time to week 8, | 2-sided chi-square test Cochran-Mantel-Haenszel test | Cochran-Mantel-Haenszel: severity of anemia (≤ 100 g/L vs. > 100 g/L to ≤ 115 g/L) and country | NR | NR |
Mean change from baseline in hemoglobin | Descriptive statistics | NA | NA | NA |
Mean (95% CI) change from baseline in serum ferritin (mcg/L) | Descriptive statistics | NA | NA | NA |
Patients with a normal TSAT level of 20% to 50%, | 2-sided chi-square test Cochran-Mantel-Haenszel test | Cochran-Mantel-Haenszel: severity of anemia (≤ 100 g/L vs. > 100 g/L to ≤ 115 g/L) and country | NA | NA |
ANCOVA = analysis of covariance; HRQoL = health-related quality of life; IBDQ = Inflammatory Bowel Disease Questionnaire; LOCF = last observation carried forward; NA = not applicable; NR = not reported; TSAT = transferrin saturation.
Source: Sponsor’s Summary of Clinical Evidence.37
Analysis Populations
Relevant analysis populations for the included trials are described in Table 10.
Table 10: Analysis Populations of VIT-IV-CL-015, FERGIcor, 1VIT5006, and VIT-IRON-2011-004 Studies
Population | Definition | Application |
|---|---|---|
VIT-IV-CL-015 | ||
FAS | All patients from the safety set who have any postdose efficacy data | Supportive analysis |
PPS | All patients from the FAS who completed the study as planned with no major deviations from the study protocol | Efficacy analysis |
Safety set | All randomized patients who received at least 1 dose of study drug | Safety analysis |
FERGIcor | ||
FAS | All randomized patients who received at least 1 dose of study drug and who attended at least 1 postbaseline visit, following the principle of ITT; patients were included in the analysis according to the treatment to which they were randomized | Primary and secondary efficacy analyses |
PPS | All randomized patients who were compliant with the study protocol, i.e., who did not experience any major protocol deviations | Supportive analysis |
Safety set | All randomized patients who received at least 1 dose of study treatment; patients were included according to the treatment received | Safety analysis |
1VIT05006 | ||
Completer | All patients who completed study drug in both periods of the crossover | All statistical tests |
Safety set | All randomized patients who received at least 1 dose of study drug | Safety analysis |
VIT-IRON-2011-004 | ||
FAS | All patients who were randomized to treatment, received at least 1 dose of study treatment, and had at least 1 baseline and postbaseline value (hemoglobin, ferritin, TSAT); the FAS was created in accordance with the intention-to-treat principles; patients were analyzed in the group to which they were randomized | Supportive efficacy analysis for the primary outcome, efficacy analysis for secondary outcomes |
PPS | All patients who, in addition to the FAS criteria, had a study drug compliance rate of between 80% and 120% and had no major protocol violations | Primary efficacy analysis for the primary outcome, efficacy analysis for secondary outcomes |
Safety set | All randomized patients who have received at least 1 dose of study drug; patients were analyzed based on the treatment they received | Safety analysis |
FAS = full analysis set; PPS = per-protocol set; TSAT = transferrin saturation.
Sources: Sponsor’s Summary of Clinical Evidence37 and Clinical Study Reports for 1VIT0500639 and VIT-IRON-2011-004.40
Results
Patient Disposition
A summary of patient disposition from the pivotal trials is in Table 11. A summary of patient disposition in the 2 RCTs in patients with IDA arising from various causes is provided in Table 12.
Table 11: Summary of Patient Disposition From Pivotal Studies
Patient disposition | VIT-IV-CL-015 | FERGIcor | ||
|---|---|---|---|---|
FCM | Iron sucrose | FCM | Iron sucrose | |
Screened, N | 446 | 880 | ||
Reason for screening failure, n (%) | ███ ██████ | 395 (44.9) | ||
Failed inclusion or exclusion criteria | NR | 361 (91.4) | ||
No reason | NR | 7 (1.8) | ||
Number of iron sucrose injections | NR | 2 (0.5) | ||
Other | NR | 25 (6.3) | ||
Randomized, N (%) | ███ | ███ | 244 | 241 |
Discontinued from study, n (%) | | █████ | ██ ██████ | 22 (9.0) | 26 (10.8) |
Reason for discontinuation, n (%) | ██ | ██ | — | — |
Adverse event | | █████ | | █████ | 8 (3.3) | 4 (1.7) |
Illness | | █████ | | █████ | 0 (0.0) | 1 (0.4) |
Significant blood loss | | █████ | | █████ | 0 (0.0) | 2 (0.8) |
Patient required blood transfusion | | █████ | | █████ | 0 (0.0) | 0 (0.0) |
Incompatible concomitant medication | | █████ | | █████ | 0 (0.0) | 2 (0.8) |
Death | | █████ | | █████ | 0 (0.0) | 0 (0.0) |
Lost to follow-up | | █████ | | █████ | 0 (0.0) | 0 (0.0) |
Physician decision | | █████ | | █████ | 1 (0.4) | 0 (0.0) |
Withdrawal by patient | | █████ | | █████ | 5 (2.0) | 12 (5.0) |
Noncompliance | | █████ | | █████ | 0 (0.0) | 5 (2.1) |
Other | | █████ | | █████ | 10 (4.1) | 6 (2.5) |
Analysis populations | ||||
FAS, N | ███ █████ | ███ █████ | 240 (98.4) | 235 (97.5) |
PPS, N | ██ ██████ | ██ ██████ | 227 (93.0) | 189 (78.4) |
Safety, N | 119 (99.2) | 118 (98.3) | 244 (100.0) | 239 (99.2) |
FAS = full analysis set; FCM = ferric carboxymaltose; NR = not reported; PP = per-protocol set.
Source: Sponsor’s Summary of Clinical Evidence.37
Table 12: Disposition for Trials In Patients With IDA Arising From Various Causes
Patient disposition | 1VIT05006 | VIT-IRON-2011-004 | ||
|---|---|---|---|---|
FCM | Placebo | FCM | Iron sucrose | |
Screened, N | NR | ███ | ||
Reason for screening failure, n | NR | ███ ██████| | ||
Failed inclusion or exclusion | NR | ███ | ||
Withdrawal by patient | NR | ██ | ||
Physician decision | NR | ██ | ||
Rescreened and randomized, n | NR | 10 | ||
Enrolled, n | ███ | ███ | ||
Randomized and not dosed | ██ | | █████ | | █████ | |
Received blood transfusion | ██ | ██ | ██ | |
Selection criteria/study compliance | ██ | ██ | ██ | |
Lost to follow-up | ██ | ██ | ██ | |
Patient request | ██ | ██ | ██ | |
Other | ██ | ██ | ██ | |
Received both doses | ███ ██████ | ██ | ██ | |
Randomized, N (%) | ███ | ███ | ███ | ███ |
Discontinued from study, n (%) | ██ █████ | ██ █████ | | █████ | | █████ |
Reason for discontinuation, n (%) | ||||
Adverse event | | █████ | | █████ | | █████ | | █████ |
Required blood transfusion | | █████ | | █████ | | █████ | | █████ |
Lost to follow-up | | █████ | | █████ | | █████ | | █████ |
Withdrawal by patient | | █████ | | █████ | | █████ | | █████ |
Selection criteria or study compliance | | █████ | | █████ | | █████ | | █████ |
Other | | █████ | | █████ | | █████ | | █████ |
Protocol violation | | █████ | | █████ | | █████ | | █████ |
Analysis populations | ||||
Completed study, N | ███ █████ | ███ █████ | ███ █████ | ███ █████ |
FAS, N | ██ | ██ | ███ █████ | ███ █████ |
PPS, N | ██ | ██ | ███ █████ | ███ █████ |
Safety, N | ███ █████ | ███ █████ | ███ ████ | ███ ████ |
FAS = full analysis set; FCM = ferric carboxymaltose; IDA = iron deficiency anemia; PPS = per-protocol set.
aPatients may have had more than 1 reason for screening failure.
Sources: Clinical Study Report for 1VIT0500639 and VIT-IRON-2011-004.40
Baseline Characteristics
The baseline characteristics of patients enrolled in the VIT-IV-CL-015 and FERGIcor pivotal trials are provided in Table 13, and those from the trials among patients with IDA arising from various causes are in Table 14. The mean ages in the VIT-IV-CL-015 study were ████ ██████ years in the ferric carboxymaltose group and ████ ██████ in the iron sucrose group. In the other studies the mean age was around 40 years. Nearly all patients in the pivotal trials (at least 97% across treatment groups) were white. In the VIT-IV-CL-015 study approximately ███ of patients were female and in the FERGIcor study 59% were female. In the trials involving patients with IDA arising from various causes, almost all (approximately 90%) of the patients were female. In the 1VIT05006 study, ███ were Caucasian (as worded in the source), and in the VIT-IRON-2011 to 04 study all the patients were Chinese. Overall mean hemoglobin levels were ████ ███ in the VIT-IV-CL-015 study, █████ ████ in the FERGIcor study, █████ ███ in Study 1VIT05006, and less than ██ ███ in Study VIT-IRON-2011-004. There were differences in serum ferritin levels between studies, with a baseline of ████ █████ in Study VIV-IV-CL-015 and ████ █████ in the FERGIcor study, and also differences in the studies with patients with IDA arising from other conditions: ████ █████ in Study 1VIT05006 and ███ █████ in Study VIT-IRON-2011-004. Similarly, there were differences between studies in TSAT in the pivotal trials, with a mean baseline of █████ in Study VIT-IV-CL-015, and ████ in the FERGIcor study, while the mean baselines were ████ in Study 1VIT05006 and ████ in Study VIT-IRON-2011-004.
The baseline characteristics outlined in Table 13 and Table 14 are limited to those most relevant to this review or those expected to affect the outcomes or interpretation of the study results.
Table 13: Summary of Baseline Characteristics of Patients From Pivotal Studies
Characteristic | VIT-IV-CL-015 | FERGIcor | ||
|---|---|---|---|---|
FCM (N = 97) | Iron sucrose (N = 86) | FCM (N = 244) | Iron sucrose (N = 239) | |
Age (years), mean (SD) | ████ ████ | ████ ██ | █████ ███ | █████ █ |
Female, n (%) | ██ ██████ | ██ █████ | 146 (59.8) | 138 (57.7) |
White, n (%) | ██ ██████ | ██ ██ | ███ █ | ███ █ |
Body mass (kg), mean (SD) | ████ ████ | ████ ██ | █████ | █████ ██ |
Body mass index (kg/m2), mean (SD) | █████ | █████ | NR | NR |
Conditions at baseline, n (%) | ||||
Cardiovascular disorders | ██ ██████ | ██ █████ | NR | NR |
Cardiac disorders | ██ | ██ | ██ █████ | ████████ |
Vascular disorders | ██ | ██ | ██ █████ | ████████ |
Hypertension | ██ | ██ | ██ █████ | ██ █████ |
Any surgical procedures | ██ ██████ | ███████ | ████████ | ████████ |
Genitourinary | ██ ██████ | ███████ | ██ | ██ |
Hepatobiliary disorders | ██ | ██ | ██ ████ | ███████ |
Endocrine and metabolic | ██ ██████ | ██ █████ | ██ | ██ |
Metabolism and nutrition disorders | ██ | ██ | ██ █████ | ██ █████ |
Abdominal and gastrointestinal | ██ ██████ | ███████ | ████████ | ████████ |
Musculoskeletal and connective tissue disorder | ██ | ██ | ████████ | ████████ |
Infections and infestations | ██ | ██ | ████████ | ██ █████ |
Respiratory, thoracic, mediastinal | ██ | ██ | ██ █████ | ██ █████ |
Renal and urinary disorders | ██ | ██ | ██ █████ | ██ █████ |
Nervous system disorders | ██ | ██ | ██ █████ | ██ █████ |
Glomerulopathy | ██ ██████ | ██ █████ | NR | NR |
Vascular nephropathy | | █████ | | █████ | NR | NR |
Interstitial nephropathy | ██ ██████ | ███████ | NR | NR |
Other | ██ ██████ | ███████ | NR | NR |
Hemoglobin (g/L), mean (SD) | ████ ██ | █████ | 101.1 (15.1) | 103.3 (15.2) |
Ferritin (ng/mL), mean (SD) | ████ ███ | ████ █ | 14.94 (24.83) | 17.9 (27.81) |
TSAT (%), mean (SD) | ██████ █ | ██████ | 8.95 (9.12) | 9.51 (9.27) |
Dialysis sessions per week, n (%) | ||||
2 | | █████ | ██ █████ | NA | NA |
3 | ██ ██████ | ██ █████ | NA | NA |
Patient on erythropoietin treatment, n (%) | ██ ██████ | ██ █████ | Na | NA |
IBD diagnosis, n (%) | ||||
Crohn disease | ██ | ██ | 86 (35.2) | 74 (31.0) |
Ulcerative colitis | ██ | ██ | ███████ | ███████ |
Anemia | ██ | ██ | 244 (100.0) | 239 (100.0) |
Duration of anemia (months), mean (SD) | ██ | ██ | █████ | █████ |
FCM = ferric carboxymaltose; IBD = inflammatory bowel disease; SD = standard deviation; TSAT = transferrin saturation.
Source: Sponsor’s Summary of Clinical Evidence.37
Table 14: Baseline Characteristics of Patients With IDA From Various Causes in 1VIT05006 and VIT-IRON-2011-004
Characteristic | 1VIT05006 | VIT-IRON-2011-004 | ||
|---|---|---|---|---|
FCM/placebo (N = 559) | Placebo (N = 559) | FCM (N = 187) | Iron sucrose (N = 180) | |
Mean (SD) age, years | ████ ███████ | ███ ████ | ███ ████ | |
Female, n (%) | ███ ██████ | ██ █████ | ██ █████ | |
Race, n (%) | ||||
Caucasian | ███ ██████ | ██ | ██ | |
African American | ███ ██████ | ██ | ██ | |
Hispanic | ███ ██████ | ██ | ██ | |
Other | ██ █████ | ██ | ██ | |
Asian | | █████ | ██ | ██ | |
Chinese | ██ | ███ ██ | ███ ██ | |
Mean hemoglobin, g/L (SD) | █████ ██████ | ████ ██ | ████ █ | |
Mean ferritin, mcg/L (SD) | █████ ████████ | ████ █ | ████ ██ | |
Mean TSAT, % (SD) | ████ ███████ | ████ ██ | ████ ██ | |
Etiology, n (%) | ||||
Menorrhagia | NR | NR | ███ ████ | ██ ██████ |
IDA | NR | NR | ██ ██████| | ██ ██████ |
Inadequate diet | NR | NR | | █████ | | █████ |
Gastrointestinal disorders | NR | NR | ██ ██████ | ██ ██████ |
Crohn disease | NR | NR | | █████ | | █████ |
Hemorrhoids | NR | NR | | █████ | | █████ |
Malabsorption | NR | NR | | █████ | | █████ |
Hemorrhoidal hemorrhage | NR | NR | | █████ | | █████ |
Uterine leiomyoma | NR | NR | | █████ | | █████ |
Metabolism and nutritional disorders | NR | NR | | █████ | | █████ |
FCM = ferric carboxymaltose; IDA = iron deficiency anemia; NR = not reported; SD = standard deviation; TSAT = transferrin saturation.
aThe cause of IDA is unknown.
Sources: Clinical Study Reports for 1VIT0500639 and VIT-IRON-2011-004.40
Exposure to Study Treatments
Exposure to study treatments is summarized in Table 15 for the pivotal studies and in Table 16 for the studies of patients with IDA arising from various causes.
Table 15: Summary of Patient Exposure From Pivotal Studies
Exposure | VIT-IV-CL-015 | FERGIcor | ||
|---|---|---|---|---|
FCM (N = 119) | Iron sucrose (N = 118) | FCM (N = 244) | Iron sucrose (N = 239) | |
Total amount received (mg of iron in the form of FCM or equivalent as iron sucrose) | ||||
Mean (SD) | ██████ ██ | ██████ | ██████ ██ | ██████ ██ |
Median | ██████ | ██████ | ██████ | ██████ |
Minimum to maximum | ████████ | █████████ | █████████ | █████████ |
True adherence categories, n (%) | ||||
< 90% | NR | NR | ██ █████ | ██ ██████ |
90 to < 100% | NR | NR | | █████ | | █████ |
100% | NR | NR | ███ ██████ | ███ ██████ |
> 100% to 110% | NR | NR | | █████ | | █████ |
> 110% | NR | NR | | █████ | ██ █████ |
FCM = ferric carboxymaltose; NR = not reported; SD = standard deviation.
Source: Sponsor’s Summary of Clinical Evidence.37
Table 16: Exposure of Patients With IDA From Various Causes in 1VIT05006 and VIT-IRON-2011-004
Exposure | 1VIT05006 | VIT-IRON-2011-004 | |||
|---|---|---|---|---|---|
FCM/placebo (N = 559) | Placebo | FCM (N = 187) | Iron sucrose (N = 180) | ||
Total amount received (mg iron FCM or equivalent as iron sucrose) | |||||
Mean (SD) | █████ ████████ | ██████ ██ | ██████ ██ | ||
Median | █████ | ██████ | ██████ | ||
Minimum to maximum | ██ █████ | █████ ████ | ████ ████ | ||
Adherence | |||||
Mean (SD) | NR | NR | ████ █████ | █████ ███ | |
< 80%, n (%) | NR | NR | | █████ | | █████ | |
80 to 120%, n (%) | NR | NR | ███ ██████ | ███ ██████ | |
FCM = ferric carboxymaltose; IDA = iron deficiency anemia; NR = not reported; SD = standard deviation.
Sources: Clinical Study Reports for 1VIT0500639 and VIT-IRON-2011-004.40
Efficacy
Key results of the pivotal trials and other RCTs among patients with IDA arising from various causes are summarized in Table 17.
Table 17: Summary of Efficacy Results (Pivotal Trials and Select RCTs)
Outcome | Studies, N | FCM | Iron sucrose | Between-group difference (95% CI) and P value (statistical test) |
|---|---|---|---|---|
Hemoglobin | ||||
Patients with ≥ 10 g/L increase in hemoglobin at week 4, n of N (%) | VIT-IV-CL-015 (patients with CKD) N = 240 | Week 4 (PPS): 45 of 97 (46.4) | Week 4 (PPS): 32 of 86 (37.2) | NR P = 0.210a (chi-square) |
Patients with ≥ 20 g/L increase in hemoglobin at week 12, n of N (%) | FERGIcor (patients with IBD) N = 465 | Week 12 (FAS, observed): 150 of 240 (65.8) | Week 12 (FAS, observed): 118 of 220 (53.6) | 12.15 (3.07 to 20.97) P = 0.004 (ANCOVA) |
Patients with ≥ 20 g/L increase in hemoglobin at any time to week 8, n of N (%) | VIT-IRON-2011-004 (patients with IDA arising from various causes) N = 371 | Week 8 (FAS): 184 of 185 (99.5) | Week 8 (FAS): 177 of 180 (98.3) | 1.13 (−2.02 to 4.68) P = NR |
Mean change from baseline in hemoglobin | VIT-IV-CL-015 (patients with CKD) N = 240 |
|
| NR |██████a (ANCOVA) |
FERGIcor (patients with IBD) N = 465 |
|
| NR |█████b (repeated measures) | |
VIT-IRON-2011-004 (patients with IDA arising from various causes) N = 371 |
|
| ████ ██████ ██ ███████ █ █████b (repeated measures) | |
HRQoL | ||||
SF-36 physical component, mean (SD) Scale ranges from 0 to 100, higher scores indicate better HRQoL | FERGIcor (patients with IBD) N = 465 |
|
| NR P = 0.157b (ANCOVA) |
SF-36 mental component, mean (SD) Scale ranges from 0 to 100, higher scores indicate better HRQoL | FERGIcor (patients with IBD) N = 465 |
|
| NR P = 0.583b (ANCOVA) |
IBDQ total score, mean (SD) Total score can range from 32 (very poor HRQoL) to 224 (perfect HRQoL) | FERGIcor (patients with IBD) N = 465 |
|
| NR P = 0.872b (ANCOVA) |
Ferritin | ||||
Mean (95% CI) change from baseline in serum ferritin (mcg/L) | VIT-IV-CL-015 (patients with CKD) N = 240 |
|
| NR |████a (ANCOVA) |
FERGIcor (patients with IBD) N = 465 |
|
| █████ ██████ ██ (repeated measures) | ██████ | |
VIT-IRON-2011-004 (patients with IDA arising from various causes) N = 371 |
|
| █████ ██████b (repeated measures) | |
TSAT | ||||
Patients with a normal TSAT level of 20% to 50%, n of N (%) | VIT-IV-CL-015 (patients with CKD) N = 240 | Week 4: █████ ███ | Week 4: █████ ██ | NR █████ (chi-square) |
FERGIcor (patients with IBD) N = 465 | Week 12 (FAS, observed): 117 of 222 (52.7) | Week 12 (FAS, observed): 76 of 209 (36.4) | OR = 2.05 (1.37 to 3.06) P < 0.001b (logistic regression) | |
Patients without anemia | ||||
Patients who were nonanemic (Hb ≥ 120 g/L for males or ≥ 130 g/L for females) at week 12 | FERGIcor N = 465 | Week 12 (FAS, observed): 166 of 228 (72.8) | Week 12 (FAS, observed): 136 of 220 (61.8) | OR = 1.65 (95% CI, 1.10 to 2.46) P = 0.015 (logistic regression) |
ANCOVA = analysis of covariance; CFB = change from baseline; CI = confidence interval; CKD = chronic kidney disease; FAS = full analysis set; FCM = ferric carboxymaltose; HRQoL = health-related quality of life; IBD = inflammatory bowel disease; IBDQ = Inflammatory Bowel Disease Questionnaire; IDA = iron-deficient anemia; NR = not reported; OR = odds ratio; PPS = per-protocol set; RCT = randomized controlled trial; SD = standard deviation; SF-36 = Short-Form (36) Health Survey; TSAT = transferrin saturation.
aThe Statistical Analysis Plan for this study did not call for any formal hypothesis testing, therefore, all reported P values should be considered supportive in nature
bP values have not been adjusted for multiple comparisons.
Sources: Sponsor’s Summary of Clinical Evidence37 and Clinical Study Reports for 1VIT0500639 and VIT-IRON-2011-004.40
Hospitalizations (All-Cause and Anemia-Related)
This outcome was not assessed in any of the 4 trials that were the focus of the systematic review section.
Hemoglobin
In Study VIT-IV-CL-015, the primary response rate was defined as the percentage of patients reaching an increase in hemoglobin of ≥ 10 g/L at 4 weeks after baseline. The percentages of patients with a response (per-protocol set) were 45 of 97 (46.4%) in the ferric carboxymaltose group and 32 of 86 (37.2%) in the iron sucrose group, with a reported P value for the between-group difference of 0.2101. Similar responses were reported for the ITT population: ██████ ███████ in the ferric carboxymaltose group and ██████ ███████ in the iron sucrose group. The mean (range) increase of hemoglobin in the ferric carboxymaltose group at week 4 was ███ █████ ███ ███ and in the iron sucrose group ███ █████ ███ ███ with a reported P value for the between-group difference of ██████. No between-group difference and CI were reported for either end point.
In the FERGIcor study, the primary outcome was patients who exhibited a response, defined as an increase in hemoglobin of at least 20 g/L at week 12. In the FAS, the percentage of patients with a response was 65.79% (150 of 240) in the ferric carboxymaltose group and 53.64% (118 of 220) in the iron sucrose group (Table 17). The between-group difference was 12.15% higher (95% CI, 3.07 to 20.97; P = 0.004) in the ferric carboxymaltose group compared with the iron sucrose group. The FAS set was also analyzed using the LOCF and the worst-case methods; the results were consistent with the primary analysis.
In Study VIT-IRON-2011-004, 99.5% of patients in the ferric carboxymaltose group and 98.3% of patients in the iron sucrose group achieved an increase in hemoglobin of at least 20 g/L from baseline to week 8, for a between-group difference of 1.13 (95% CI, −2.02 to 4.68) (Table 17). The criteria for noninferiority were met. The results for the FAS population were identical to those of the PPS. From a mean baseline of ████ ███ ███████ in the ferric carboxymaltose group and ████ ███ ███████ in the iron sucrose group, the mean change from baseline to week 8 was ████ ███ ███████ in the ferric carboxymaltose group and ████ ███ ███████ in the iron sucrose group. The between-group difference was ████ ███ ████ ███ █████ ██ ██████ ███████.
Patient-Reported HRQoL
In the FERGIcor study, for the physical component of the SF-36, from mean baselines of 44.17 (SD = 7.36) in the ferric carboxymaltose group and 44.98 (SD = 7.23) in the iron sucrose group, there were changes from baseline to week 12 of 3.88 (SD = 6.77) and 2.64 (SD = 7.14) (Table 17) for a reported between-group P value of 0.157. For the mental component of the SF-36, from a mean baseline of 40.02 (SD = 11.04) in the ferric carboxymaltose group and 41.30 (SD = 11.70) in the iron sucrose group, there was a change from baseline to week 12 of ████ ███████ ███ ████ ███████, for a between-group P value of ███████. For the IBDQ total score, from mean baselines of 150.8 (SD = 35.2) in the ferric carboxymaltose group and 152.7 (SD = 34.4) in the iron sucrose group, there was a change from baseline to week 12 of ████ ██████ ███ ████ ██████, for a between-group P value of ███████. Between-group differences and CIs were not reported for any of the HRQoL measures.
HRQoL was not assessed in either Study VIT-IV-CL-015 or Study VIT-IRON-2011-004.
Serum Ferritin
In Study VIT-IV-CL-015, from mean baselines of ████ mcg/L (range = ██ ███) in the ferric carboxymaltose group and ████ mcg/L (range = ██ ████ in the iron sucrose group, after week 4 there were changes from baseline of █████ mcg/L (range = █████ ████) in the ferric carboxymaltose group and █████ █████ (range = ███ ████) in the iron sucrose group (Table 17), with a reported between-group P value of less than ████. The between-group difference and CI were not reported.
In the FERGIcor study, from mean baselines of █████ mcg/L (95% CI, not reported [NR]) in the ferric carboxymaltose group and █████ mcg/L (95% CI, NR) in the iron sucrose group, after week 4 there were changes from baseline of ████ mcg/L (95% CI, NR) in the ferric carboxymaltose group and █████ mcg/L (95% CI, NR) in the iron sucrose group (Table 17). for an estimated difference between groups (repeated measures analysis) of █████ (95% CI, █████ ██ ██████; P < █████).
In Study VIT-IRON-2011-004, from mean baselines of ████ mcg/L (█████) in the ferric carboxymaltose group and ████ mcg/L (█████) in the iron sucrose group, after week 8 there were changes from baseline of 200 mcg/L (███████) in the ferric carboxymaltose group and ██████ mcg/L (██████) in the iron sucrose group, for a least square mean between groups of 52.65 mcg/L (95% CI, ██████ █████), P < █████ (Table 17).
Transferrin Saturation
In VIT-IV-CL-015, there were █████ patients (█████) in the ferric carboxymaltose group and █████ patients (██████ in the iron sucrose group who had a normal TSAT of between 20% and 50% by week 4, for a reported between-group P value ██████. The between-group difference and CI were not reported.
In the FERGIcor trial, there were 117 or 222 patients (52.7%) in the ferric carboxymaltose group and 76 of 209 patients (36.4%) in the iron sucrose group with a normal TSAT level (20% to 50%) at week 12. The OR was ████ ████ ██ █████ ██████ ███████ in favour of ferric carboxymaltose (Table 17). The absolute between-group difference and CI were not reported.
The TSAT response (achieving a normal TSAT) was not assessed in VIT-IRON-2011-004.|
Patients Without Anemia
In the FERGIcor trial, for the proportion of patients without anemia (a hemoglobin level of ≥ 120 g/L for females or ≥ 130 g/L for males) at week 12, the percentages of patients with a response were 72.8% (166 of 228) in the ferric carboxymaltose group and 61.8% (136 of 220) in the iron sucrose group. The OR was ████ ████ ██ █████ █████ ███████. The absolute between-group difference and CI were not reported.
Harms
Refer to Table 18 and Table 19 for harms data.
Adverse Events
In the overall population in Study VIT-IV-CL-015, ██████ ███████ patients in the ferric carboxymaltose group and ██████ ███████ patients in the iron sucrose group had at least 1 AE. In the FERGIcor study, there were AEs in ███████ █████ patients in the ferric carboxymaltose group and in ███████ ███████ patients in the iron sucrose group. The most common AEs, defined as occurring in at least 2% of patients in either treatment group of the overall population, are presented in Table 18.
In the overall population in Study 1VIT05006, ███████ ███████ patients in the ferric carboxymaltose group and ███████ ███████ patients in the placebo group reported at least 1 AE. In Study VIT-IRON-2011-004, ███████ ███████ patients in the ferric carboxymaltose group and ███████ ███████ patients in the iron sucrose group had an AE. The most common AEs are reported in Table 19.
Serious Adverse Events
In Study VIT-IV-CL-015, SAEs were reported in █████ ██████ patients in the ferric carboxymaltose group and █████ ██████ patients in the iron sucrose group. In the FERGIcor study SAEs were reported in ██████ ██████ patients in the ferric carboxymaltose group and ██████ ██████ patients in the iron sucrose group. The most frequently occurring SAEs are reported in Table 18.
In Study 1VIT05006, █████ ██████ patients in the ferric carboxymaltose group and █████ ██████ patients in the placebo group had an SAE. In VIT-IRON-2011-004 ██████ ██████ patients in the ferric carboxymaltose group and █████ ██████ patients in the iron sucrose group had an SAE (Table 19).
Withdrawals Due to Adverse Events
In Study VIT-IV-CL-015, █████ ██████ patients in the ferric carboxymaltose group and █████ ██████ patients in the iron sucrose group withdrew from study medication due to AEs. In the FERGIcor study, 7 of 224 patients (2.9%) in the ferric carboxymaltose group and 2 of 239 patients (0.8%) in the iron sucrose group withdrew from the study drug due to an AE. Reasons for withdrawal from study drug are reported in Table 18.
In Study 1VIT05006, █████ ██████ patients in the ferric carboxymaltose group and █████ ██████ patients in the placebo group withdrew from study medication due to an AE. In Study VIT-IRON-2011-004 █████ ██████ patients in the ferric carboxymaltose group and █████ ██████ patients in the iron sucrose group withdrew from study medication due to an AE (Table 19).
Mortality
███ patient in the ferric carboxymaltose group died during Study VIT-IV-CL-015 because of acute anterior myocardial infarction, which was serious, severe in intensity and unlikely to be related to study medication (Table 18). ███ ███████ died more than a week after study medication was withdrawn due to another nonserious AE. In 1VIT05006, ███ ███████ in the ferric carboxymaltose group died due to pneumonia. These were the only deaths across the 4 studies (Table 19).
Notable Harms
In the FERGIcor study, hypophosphatemia was observed in 6 patients (2.5%) in the ferric carboxymaltose group, and none were observed in the iron sucrose group. Hypophosphatemia was not reported in Study VIT-IV-CL-015.
In Study VIT-IRON-2011, hypophosphatemia was observed in ██ patients receiving ferric carboxymaltose ███████ and in ██ patients ██████ in the iron sucrose group. The number of patients with decreased blood phosphorous or hypophosphatemia was not reported in 1VIT05006.
Table 18: Summary of Harms Results From Pivotal Studies
Adverse events | VIT-IV-CL-015 | FERGIcor | ||
|---|---|---|---|---|
FCM (N = 119) | Iron sucrose (N = 118) | FCM (N = 244) | Iron sucrose (N = 239) | |
Most common AEs, n (%) | ||||
≥ 1 AE | ██ █████ | ██ █████ | ███ ████ | ███ ████ |
Most common TEAEs (≥ 2% in any group) | ||||
Cardiac disorders | █████ | █████ | ██ | ██ |
Gastrointestinal disorders | █████ | █████ | ██ █████ | ██ █████ |
Vomiting | █████ | █████ | ██ | ██ |
Colitis ulcerative | ██ | ██ | ██ █████ | █████ |
Nausea | ██ | ██ | █████ | ██ █████ |
Abdominal pain | ██ | ██ | █████ | █████ |
Diarrhea | ██ | ██ | █████ | █████ |
General disorders and administration site conditions | █████ | █████ | ██ █████ | ██ █████ |
Fatigue | ██ | ██ | █████ | █████ |
Pyrexia | ██ | ██ | █████ | █████ |
Infections and infestations | ██ █████ | ██ █████ | ██ | ██ |
Nasopharyngitis | ██ | ██ | ██ █████ | █████ |
Influenza | ██ | ██ | █████ | █████ |
Respiratory tract infection viral | ██ | ██ | █████ | █████ |
Injury, poisoning, and procedural complications | █████ | █████ | ██ █████ | ██ █████ |
Increased alanine aminotransferase | ██ | ██ | █████ | █████ |
Increased C-reactive protein | ██ | ██ | █████ | █████ |
Increased serum ferritin | ██ | ██ | █████ | █████ |
Decreased blood phosphorus | ██ | ██ | █████ | █████ |
Investigations | █████ | █████ | ██ | ██ |
Increased blood pressure | █████ | █████ | ██ | ██ |
Metabolism and nutritional disorders | █████ | █████ | █████ | █████ |
Musculoskeletal and connective tissue disorders | █████ | ██ █████ | ██ █████ | ██ █████ |
Arthralgia | █████ | █████ | █████ | █████ |
Muscle cramp | █████ | █████ | ██ | ██ |
Nervous system disorders | █████ | █████ | ██ █████ | ██ █████ |
Headache | █████ | █████ | ██ █████ | █████ |
Dizziness | ██ | ██ | █████ | █████ |
Psychiatric disorders | █████ | █████ | ██ | ██ |
Skin and subcutaneous tissue disorders | █████ | █████ | ██ █████ | ██ █████ |
Rash | ██ | ██ | █████ | █████ |
Surgical and medical procedures | █████ | █████ | ██ | ██ |
Vascular disorders | ██ █████ | ██ █████ | █████ | █████ |
Hemorrhage | █████ | █████ | ██ | ██ |
Hypertension | █████ | █████ | ██ | ██ |
Hypotension | ██ █████ | ██ █████ | ██ | ██ |
Serious adverse events, n (%) | ||||
Patients with ≥ 1 serious adverse event | █████ | █████ | ██ █████ | ██ █████ |
Most common (occurring in > 1 patient in any group) | ||||
Cardiac disorders | █████ | █████ | █████ | █████ |
Myocardial infarction | █████ | █████ | █████ | █████ |
Gastrointestinal disorders | █████ | █████ | █████ | █████ |
Gastrointestinal hemorrhage | █████ | █████ | █████ | █████ |
Melaena | █████ | █████ | █████ | █████ |
Ulcerative colitis | █████ | █████ | █████ | █████ |
Infections and infestations | █████ | █████ | █████ | █████ |
Pneumonia | █████ | █████ | █████ | █████ |
Gastroenteritis | █████ | █████ | █████ | █████ |
Patients who stopped treatment due to AEs, n (%) | ||||
Patients with ≥ 1 AE leading to study drug withdrawal | █████ | █████ | 7 (2.9) | 2 (0.8) |
Most common (occurring in > 1 patient in any group) | — | — | — | — |
Gastrointestinal disorders | █████ | █████ | █████ | █████ |
Gastrointestinal hemorrhage | █████ | █████ | █████ | █████ |
Melaena | █████ | █████ | █████ | █████ |
Investigations | █████ | █████ | █████ | █████ |
Increased serum ferritin | █████ | █████ | █████ | █████ |
Skin and subcutaneous tissue disorders | █████ | █████ | █████ | █████ |
Pruritus | █████ | █████ | █████ | █████ |
Rash | █████ | █████ | █████ | █████ |
Generalized rash | █████ | █████ | █████ | █████ |
Deaths, n (%) | ||||
Patients who died | █████ | █████ | █████ | █████ |
Deaths due to a cardiovascular event | █████ | █████ | █████ | █████ |
AEs of special interest, n (%) | ||||
Hypophosphatemia | ██ | ██ | 6 (2.5) | 0 (0.0) |
Infections and infestations | ██ █████ | ██ █████ | NR | NR |
Nasopharyngitis | ██ | ██ | ██ █████ | █████ |
Influenza | ██ | ██ | █████ | █████ |
Respiratory tract infection viral | ██ | ██ | █████ | █████ |
Hypertension | █████ | █████ | ██ | ██ |
Hypotension | ██ █████ | ██ █████ | ██ | ██ |
AE = adverse event; FCM = ferric carboxymaltose; NA = not applicable; NR = not reported; TEAE = treatment-emergent adverse event.
Source: Sponsor’s Summary of Clinical Evidence.37
Table 19: Summary of Harms Results for Trials in Patients With IDA From Various Causes
Adverse events | 1VIT05006 | VIT-IRON-2011-004 | ||
|---|---|---|---|---|
FCM (N = 559) | Placebo (N = 559) | FCM (N = 187) | Iron sucrose (N = 180) | |
Most common AEs, n (%) | ||||
≥ 1 AE | ███ ████ | ███ █████ | ███ ████ | ███ ████ |
Most common TEAEs (≥ 2% in any group) | ||||
Investigations | ██ █████ | ██ █████ | ██ █████ | ██ █████ |
Urine phosphorus decreased: | ██ | ██ | ██ █████ | ██ █████ |
Alanine aminotransferase increased: | █████ | █████ | ██ █████ | █████ |
White blood cells urine-positive | ██ | ██ | █████ | █████ |
Aspartate aminotransferase increased: | █████ | █████ | █████ | █████ |
Gamma-glutamyl transferase increased: | ██ | ██ | █████ | █████ |
Hepatic enzyme increased | ██ | ██ | █████ | █████ |
Red blood cells-positive | ██ | ██ | █████ | █████ |
Metabolism and nutrition disorders | ██ | ██ | ██ █████ | █████ |
Hypophosphatemia | ██ | ██ | ██ █████ | █████ |
Gastrointestinal disorders | ██ █████ | ██ █████ | ██ █████ | ██ █████ |
Nausea | ██ █████ | ██ █████ | █████ | █████ |
Upper abdominal pain | ██ | ██ | █████ | █████ |
Crohn disease | ██ | ██ | █████ | █████ |
Diarrhea | █████ | █████ | ██ | ██ |
General disorders and administration site conditions | ██ █████ | ██ █████ | ██ █████ | ██ █████ |
Pyrexia | █████ | █████ | ██ █████ | █████ |
Fatigue | █████ | █████ | ██ | ██ |
Hepatobiliary disorders | ██ | ██ | ██ █████ | ██ █████ |
Abnormal hepatic function | ██ | ██ | ██ █████ | █████ |
Skin and subcutaneous tissue disorders | ██ █████ | ██ █████ | ██ █████ | █████ |
Rash | █████ | █████ | █████ | █████ |
Urticaria | ██ | ██ | █████ | █████ |
Reproductive system and breast disorders | ██ | ██ | █████ | █████ |
Menorrhagia | ██ | ██ | █████ | █████ |
Nervous system disorders | ██ █████ | ██ █████ | █████ | █████ |
Headache | ██ █████ | ██ █████ | █████ | █████ |
Dizziness | ██ █████ | █████ | ██ | ██ |
Respiratory, thoracic, and mediastinal disorders | █████ | █████ | █████ | █████ |
Blood and lymphatic system disorders | ██ | ██ | █████ | █████ |
Musculoskeletal and connective tissue disorders | ██ █████ | █████ | ██ | ██ |
Serious adverse events, n (%) | ||||
Patients with ≥ 1 SAE | █████ | █████ | ██ █████ | █████ |
Most common SAE (occurring in > 1 patient in any group) | ||||
Crohn disease | NA | NA | █████ | █████ |
Menorrhagia | NA | NA | █████ | █████ |
Patients who stopped treatment due to AEs, n (%) | ||||
Patients with ≥ 1 AE leading to study drug withdrawal | █████ | █████ | █████ | █████ | |
Deaths, n (%) | ||||
Patients who died | ██████ | █████ | █████ | █████ |
AEs of special interest, n (%) | ||||
Blood phosphorus decreased | ██ | ██ | ██ █████ | █████ |
Infections and infestation | ██ █████ | ██ █████ | ██ █████ | ██ █████ |
Upper respiratory tract infection | ██ | ██ | ██ █████ | ██ █████ |
Urinary tract infection | ██ | ██ | █████ | █████ |
AE = adverse event; FCM = ferric carboxymaltose; IDA = iron deficiency anemia; NA = not applicable; NR = not reported; SAE = serious adverse events; TEAE = treatment-emergent adverse event.
Sources: Clinical Study Reports for 1VIT0500639 and VIT-IRON-2011-004.40
Critical Appraisal
Internal Validity
The pivotal trials and the other trials with IDA caused by various conditions appeared to be reasonably well-designed with measures taken to maintain allocation concealment during the randomization process and to achieve reasonable similarity in baseline characteristics between groups. All of the active-controlled trials lacked blinding. It is unlikely that laboratory values such as hemoglobin, ferritin, and TSAT would be influenced by patient knowledge of their treatment assignment. However, it is possible that patient-reported outcomes such HRQoL and subjective harms could be influenced by patients knowing which group they were assigned to. This bias may be mitigated by the fact that both groups received active treatment. However, there still may be a bias if patients assume that a newer therapy is more effective, or the reverse may be true if patients assume that a more established therapy is more effective. As Study 1VIT05006 was double-blinded, there is a low risk of bias in the measurement of the outcomes. As there were no substantial differences in reported harms in this trial, it is unlikely that patients would have become unblinded due to known effects of the intervention or comparator drugs.
There were fewer patients who withdrew from Study VIT-IV-CL-015 in the ferric carboxymaltose group than from the iron sucrose group █████ ██████ ██████, with the largest single reason being an AE █████ ██████ █████. There was not only a numerical difference in withdrawals between the 2 groups, but the percentage of withdrawals in the iron sucrose group was also high, at greater than 10%. This difference in withdrawals may bias assessments of efficacy outcomes if patients are withdrawing due to a lack of efficacy, and, particularly in this case, where there is a notable difference in withdrawals due to AE, assessment of harms. The sponsor did not plan for any formal statistical analyses in this study, no sensitivity analyses were performed, and no attempt was made to replace missing values. The direction of the potential bias is uncertain. There were no clear differences in withdrawals in the FERGIcor, VIT-IRON-2011 to 04, and 1VIT05006 studies.
The VIT-IV-CL-015 study was not planned to perform formal comparisons between ferric carboxymaltose and iron sucrose, and the other 2 studies that assessed efficacy outcomes did not appear to have a multiple-testing procedure in place. This limits the strength of conclusions that can be drawn about the efficacy of ferric carboxymaltose versus iron sucrose, outside of the primary outcome of patients with a hemoglobin response, as any statistically significant results reported after the primary outcome are at risk of type I error (a false-positive result).
The 2 active-controlled studies, FERGIcor and VIT-IRON-2011-004, that planned for formal between-group comparisons both used noninferiority designs; however, neither provided a justification for the noninferiority margin they chose. For example, in Study VIT-IRON-2011-004, the investigators used a noninferiority margin of 15% for the primary outcome and assumed a proportion patients who responded of 70% in the iron sucrose group. However, it is unclear where either of these estimates were obtained. The impact of this omission is likely minimal, given that there was a higher proportion of patients who responded in the ferric carboxymaltose group compared with the iron sucrose group. Both of these noninferiority trials planned to conduct their primary analysis of efficacy in the per-protocol population rather than in the FAS or ITT population. Because the results for the per-protocol population and FAS or ITT population were consistent with each other in each trial, there are no concerns with this approach.
There was a crossover design used in 1VIT05006, which was the only blinded study and the only study with a placebo control. Patients crossed over only 7 days after receiving their initial treatment assignment, and it is unclear if that was adequate for washout to prevent additive effects from first-line treatment.
The scales used to assess HRQoL, and the laboratory markers used to assess efficacy are all well-established and well validated instruments or assays. However, it is unclear how useful the IBDQ instrument would be for assessing the specific impact of IDA in patients with IBD. A generic instrument, the SF-36, was also used to assess HRQoL in this study, and it is unclear whether this instrument can separate changes in HRQoL due to the underlying condition (IBD) from those due to improvements in IDA.
Between-group differences, with a 95% CI, were not always reported in the included studies because formal comparisons were not planned. However, where provided, there appeared to be a reasonable degree of precision around the mean difference for most outcomes. Where absolute between-group differences with CIs were not reported (e.g., HRQoL, change from baseline hemoglobin, and patients with a TSAT response in the FERGIcor study, and patients with a hemoglobin response, change from baseline hemoglobin, change from baseline ferritin, and patients with a TSAT response in Study VIT-IV-CL-015), the precision of the effect estimates (i.e., whether the 95% CIs included values that would suggest a conclusion different from that suggested by the point estimate) could not be judged. There tended to be greater variability in within-group changes in outcomes such as ferritin; for example, in VIT-IV-CL-015, the change from baseline in ferritin to week 4 ranged from −205 mcg/L to 1,786 mcg/L. A formal GRADE assessment was not performed in this review given that a complete body of evidence was not included for any patient population or comparison.
External Validity
The only study conducted among patients with IDA arising from various causes that reported efficacy results was conducted solely in China, and all participants were Chinese. The clinical expert did not believe this to be a significant issue with respect to generalizability, as hemoglobinopathies were excluded from the trial (conditions such as thalassemia that are seen more commonly in patients who are Chinese). There were few patients who identified as Indigenous in any of the included studies, even though there are a disproportionate number of Indigenous people who have IDA in Canada. The clinical expert consulted on this review believed that the pivotal trial that included patients with IBD would likely be generalizable to any patient with IDA, whereas the pivotal trial that included patients with CKD would be less generalizable, given the nature of CKD, in which the pathophysiology of anemia is much more complicated. The relatively short follow-up in the pivotal trials and in Study 1-VIT-IRON-2011-004 (4 to 12 weeks) is likely not adequate for assessing the effects of ferric carboxymaltose on hard clinical outcomes, such as hospitalizations for anemia; however, the clinical expert believed that a minimum of 4 weeks would likely be adequate, but not ideal, for assessing laboratory response to an iron infusion in Canada.
The clinical expert noted that IV iron was administered more frequently in the trial among patients with CKD (2 to 3 times weekly) than it would be in Canadian practice, where it would be administered weekly (at most). The clinical expert also noted that dosing of IV iron replenishment in Canada is based on the Ganzoni formula, and this differs slightly from the approach taken in the clinical trials. The clinical expert believed iron sucrose as an appropriate comparator, one that is commonly used in Canada. However, there was no pivotal trial or trial among patients with IDA arising due to various conditions with a comparison to ferric derisomaltose.
The clinical expert found that, across the trials, the inclusion criteria for some lab parameters were different than would be seen in clinical practice, noting that, in Canada, a ferritin level of less than 50 mcg/L would warrant treatment, while the clinical trials used a ferritin level of less than 100 mcg/L or less than 200 mcg/L for inclusion. It is not clear what the implications are with respect to generalizability.
The clinical expert consulted for this review believed that hemoglobin is likely the most important laboratory value when assessing response to ferric carboxymaltose or other iron replacement products, and agreed with positioning hemoglobin as a primary outcome. The clinical expert also believed that the cutoffs used to define a patient who responded in the pivotal trials and other select trials as appropriate. However, the clinical expert also agreed that clinical outcomes such as fatigue and HRQoL are important measures of treatment success, and these outcomes were absent from 3 of the included trials. Although HRQoL was measured in the FERGIcor trial, it is uncertain whether the measurement tools used were appropriate to estimate the HRQoL impacts of IV iron therapy.
GRADE Summary of Findings and Certainty of the Evidence
Results
In the absence of a complete body of evidence for any patient population or comparison, a formal GRADE assessment was not performed for this review.
Indirect Evidence
Contents within this section were informed by materials submitted by the sponsor. The following have been summarized and validated by the CDA-AMC review team.
Objectives for the Summary of Indirect Evidence
The objective of this section is to summarize and critically appraise the methods and findings of the indirect evidence submitted by the sponsor. The sponsor submitted a summary of indirect evidence from a published NMA that compared the effects of ferric carboxymaltose with those of other IV iron therapies in the context of adults with IDA associated with IBD.22
The sponsor noted that at least 2 other published NMAs compare the effects of ferric carboxymaltose with those of other iron therapies.63,64 These NMAs, by Pollock and Muduma (2019) and Han et al. (2023), compared the effects of ferric carboxymaltose and iron derisomaltose among patients with IDA secondary to various conditions (excluding pregnancy), and during pregnancy, respectively. The reason given by the sponsor for selecting the NMA by Aksan et al. was the relatively low levels of statistical, clinical, and methodological heterogeneity compared to the other 2 NMAs. The methods for identifying the NMA by Aksan et al. and other candidate NMAs were not reported. There was no protocol outlining a priori how 1 or more NMAs would be selected for presentation among multiple NMAs reporting on the same population, comparisons, and outcomes.
Description of Indirect Comparison
Aksan et al. published a systematic literature review and NMA in 2017 that used a literature search updated to June 2016. The authors screened 322 records that were potentially of interest, from which 15 studies were selected for inclusion in the systematic review. Of these, 5 studies were included in the NMA based on the systematic review methods and selection criteria described in Table 20.
Table 20: Study Selection Criteria and Methods for Systematic Review Contributing to the NMA Submitted by the Sponsor
Characteristics | Indirect comparison |
|---|---|
Population | Patients with iron deficiency anemia associated with inflammatory bowel disease |
Intervention and comparators | The meta-analysis included iron sucrose, oral iron, ferric derisomaltose, and ferric carboxymaltose; no restrictions were imposed on the treatment dose; the indirect treatment comparison included only studies investigating iron treatment as monotherapy |
Outcome | Treatment response rate, defined as the proportion of patients showing hemoglobin normalization or an increase ≥ 2 g/dL, compared to baseline |
Study designs | RCTs with minimum of 4 weeks of observation time, published in English |
Exclusion criteria | RCTs not reporting data on hematopoietic response, not published in English, carried out exclusively in children and/or adolescents or pregnant and/or lactating females, and including iron in combination with other treatments were excluded |
Databases searched | PubMed, SCOPUS, Web of Science, Cochrane Database of Systematic Reviews, Cochrane Central, Cochrane Methodology Register |
Selection process | Two independent reviewers assessed the studies identified via the database search for relevance; in case of uncertainty, a third independent reviewer was consulted, until consensus was reached |
Data extraction process | Two authors reviewed and extracted the data independently, with a third reviewer participating to resolve differences in opinion between the first 2; information extracted included the number of participants, hematological inclusion criteria, participants’ baseline characteristics, iron dosage, response rate, hemoglobin increase after treatment, and safety outcomes |
Quality assessment | The risk of bias of the eligible studies was assessed using the Cochrane Collaboration’s Risk of Bias tool (v. 2011); the number of reviewers involved and whether they worked independently was not reported |
NMA = network meta-analysis; RCT = randomized controlled trial.
Source: Sponsor’s Summary of Indirect Comparisons.22
Network Meta-Analysis
Objectives
The objective of the NMA was to compare the efficacy and tolerability of different IV iron formulations used to treat IDA in IBD patients in a systematic review and Bayesian network meta-analysis.
Study Selection Methods
A literature search was performed in PubMed, Scopus, the Web of Science, and the Cochrane Library to identify original studies analyzing the efficacy and safety of IV iron as monotherapy for IDA in patients with IBD. The last search was completed in June 2016. Only publications in the English language were included.
Network Meta-Analysis Methods
The NMA was performed within a Bayesian framework using Markov chain Monte Carlo methods. The authors did not provide any information on the prior distributions used for the modelling parameters. Similarly, no information was provided on the convergence diagnostic, the burn-in period, the number of iterations, or the number of chains used.
A fixed-effect model and a random-effect model were fitted on the data. The deviance information criterion (DIC) was used to assess the model fit and to select the model for the main analysis.
Nodes in the network were built considering different types of IV iron compounds. However, different doses of the same drug were pooled in the same nodes.
Statistical heterogeneity was tested using I2 statistics. Clinical and methodological heterogeneity was informally assessed by reviewing the included studies. However, more formal analyses and methods, such as meta-regression analyses, were missing. Network consistency was checked using the node-split approach. No subgroup analyses were performed.
The ITC results were presented as ORs related to response rate. Only 1 outcome was assessed in the NMA. Harms were not analyzed.
Results of the Network Meta-Analysis
Summary of Included Studies
The authors included 5 RCTs in the NMA. All studies were performed in patients with inflammatory bowel disease. The interventions in the studies included ferric carboxymaltose, iron sucrose, iron isomaltoside (also known as ferric derisomaltose), and oral iron. The evidence network is depicted in Figure 1. A summary of the characteristics of the included studies is presented in the following section. Sources of clinical and methodological heterogeneity across the included studies is summarized in Table 21.
Summary of Design and Population Characteristics of the RCTs in the NMA (Aksan et al.)22
Evstatiev et al. (2011), efficacy outcomes measured at 12 weeks
N = 483 (ferric carboxymaltose subgroup: n = 244; iron sucrose subgroup: n = 239)
Median age: 39.5 years (ferric carboxymaltose group); 38 years (iron sucrose group)
Interventions:
IV ferric carboxymaltose, 500 or 1,000 mg; up to 3 infusions (mean total dose = 1,377 mg ± 381 mg)
IV iron sucrose, 200 mg; up to 11 infusions, twice weekly (mean total dose = 1,160 mg ± 316 mg)
Kulnigg et al. (2008), efficacy outcomes measured at 12 weeks
N = 196 (ferric carboxymaltose group: n = 136, oral iron group: n = 60)
Median age: 40 years (ferric carboxymaltose group); 45 years (ferrous sulphate group)
Interventions:
IV ferric carboxymaltose: iron requirements calculated according to Ganzoni formula; maximum permitted dose of 1,000 mg, or 15 mg/kg body weight in patients weighing less than 66 kg; a maximum of 3 infusions was permitted (per treatment cycle)
Oral iron sulphate: 100 mg ferrous iron, administered twice daily for 12 weeks
Lindgren et al. (2009), efficacy outcomes measured at 20 weeks
N = 91 (iron sucrose group: n = 45; oral iron group: n = 46)
Median age: 42 years
Interventions:
IV iron sucrose: total dose individually determined according to SPC, based on the Ganzoni formula; drug given either in a single weekly dose of 200 mg, or every second week until cumulative dose reached; 1,000 mg was given to replenish iron stores; mean dose = 1,708 mg ± 331 mg
Oral iron sulphate: 100 mg, 2 tablets twice daily; mean dose = 38,387 mg ± 19,955 mg
Reinisch et al. (2013), efficacy outcomes measured at 8 weeks
N = 327 (219 in the iron isomaltoside group and 108 in the oral iron sulphate group)
Median age: 36 years
Interventions
IV iron isomaltoside (also called ferric derisomaltose): Ganzoni formula used; patients randomized to either a single once-weekly infusion of up to 1,000 mg of iron isomaltoside over 15 minutes until reaching cumulative dosage or to a single once-weekly 500 mg bolus injection over 2 minutes until reaching cumulative dosage Mean dose: 885 mg ± 238 mg (infusion) and 883 mg ± 296 mg (bolus)
Oral iron sulphate: 200 mg oral iron daily for 8 weeks; total dose: 11,200 mg elemental iron
Schröder et al. (2005), efficacy outcomes measured at 6 weeks
N = 46 (iron sucrose group: n = 22; oral iron group: n = 24)
Median age): 35 years (males); 33 years (females)
Interventions:
Iron sucrose group received a loading dose of iron sucrose of the maximal recommended concentration of 7 mg/kg body weight administered as a drip infusion over 3.5 hours, at visit 1; thereafter, 200 mg of iron sucrose was infused over 30 minutes once or twice weekly over 5 weeks in a fixed schedule; median total dose = 1,418 mg (1,375 mg to 1,452 mg); total mean dose = 1,418 mg)
Oral iron sulphate; 100 mg to 200 mg per day for 6 weeks; median total dose = 4,200 mg; total mean dose = 5,600 mg.
Risk-of-Bias Assessment of Included RCTs
All trials were considered at low risk of selection bias by the NMA authors (i.e., randomization and allocation concealment were considered adequate). Participants and personnel were not blinded to the interventions in 4 of 5 studies; these were considered at unclear risk of performance bias by the NMA authors. There was incomplete outcome data for 2 studies that lacked clear numerical presentations of hematological results and associated statistics. It is unclear from the manuscripts of 3 studies whether the authors had full access to collected data. Both of the latter cases were considered at unclear risk of selective reporting bias by the NMA authors.22
Table 21: Assessment of Clinical and Methodological Heterogeneity of Trials in the NMA
Characteristic | Description |
|---|---|
Disease severity | Patients in the 2 RCTs investigating FCM had an average hemoglobin level at baseline of 9.5 g/dL. Patients in the RCT investigating FD had an average baseline hemoglobin level of 9.6 g/dL. The 2 studies investigating iron sucrose had similar hemoglobin levels at baseline (10.35 g/dL and 9.7 g/dL). |
Study population | All studies included only patients with IBD. |
Treatment and comparator administration schedule | The 2 RCTs investigating FCM show some heterogeneity in the planned administration schedule:
Despite the differences in the planned administration schedule, the average exposure to FCM in the 2 trials was comparable and amounted to 1,377 mg in Evstatiev et al. (2011) and 1,405.5 mg in Kulnigg et al. (2008). In the RCT investigating FD, 7 patients received weekly FD infusions of up to 1,000 mg until reaching the total required iron dose, calculated using an adapted Ganzoni formula. The oral iron group received 200 mg iron per day for 8 weeks. In the trials comparing FCM or FD vs. oral iron, the 2 treatments showed similar levels of relative exposure vs. the common comparator. FCM and FD patients received, respectively, 91.6% and 92.1% iron less than did the oral iron arm. The 2 trials investigating iron sucrose vs. oral iron showed a more heterogenous relative treatment exposure: the iron sucrose group received 95.6% and 66.2% less iron than did the oral iron arm in the studies from Lindgren et al. (2009) and Schröder et al. (2005) respectively. |
Definition of end points | All RCTs adopted the same definition of treatment response, defined as an increase in hemoglobin from baseline of ≥ 2 mg/dL. |
Timing of end point Evaluation | One of the 2 RCTs investigating FCM measured the outcomes at 12 weeks. The other RCT investigating FCM reported the proportion of patients who responded at multiple time points up to 12 weeks. The RCT investigating FD measured the outcomes at 8 weeks. The 2 RCTs investigating iron sucrose measured outcomes at 20 weeks and 6 weeks. |
Withdrawal frequency | The average withdrawal rate in the 2 RCTs investigating FCM (10.7%) was close to the withdrawal rate in the RCT investigating FD (12.4%). The withdrawal rates in the study investigating iron sucrose ranged from 14.3% to 23.9%. |
Study design | Four RCTs were open-label. One RCT was evaluator-blind. |
FCM = ferric carboxymaltose; FD = ferric derisomaltose; IDA = iron deficiency anemia; NMA = network meta-analysis; RCT = randomized controlled trial.
Source: Sponsor’s Summary of Indirect Comparisons.22
Results
Efficacy
Point estimates for the ORs for hemoglobin response comparing ferric carboxymaltose with ferric derisomaltose and iron sucrose favoured ferric carboxymaltose. However, the 95% CrIs were wide and included effects favouring the comparator interventions, suggesting uncertainty with and imprecision indifferences in the ORs between ferric carboxymaltose versus iron sucrose (OR = 0.70; 95% CrI = 0.48 to 1.0) and ferric carboxymaltose versus iron isomaltoside (OR = 0.69; 95% CrI = 0.34 to 1.4).
Results of the NMA found that treatment with ferric carboxymaltose had a superior response compared to treatment with oral iron (OR = 1.9; 95% CrI, 1.1 to 3.2).
The node-splitting approach showed consistency in the analysis, with all P values without statistical significance. In line with the assessment of consistency, the low I2 statistics (10%) showed limited statistical heterogeneity. The fixed-effect model offered a better fit (DIC = 18.215) compared with the random-effect model (DIC = 15.585).
Harms
Harms were not assessed in the NMA.
Critical Appraisal of the Network Meta-Analysis
The sponsor selected a published NMA that used a Bayesian approach with fixed-effects models for the analyses. Limited information is available regarding the methods used in the NMA model. The NMA was informed by a systematic review of relevant databases. However, there was no search of trial registries, reference lists of included studies, or grey literature, and no evidence of consultation with content experts, limiting the comprehensiveness of the search.65 The date last searched was June 2016, more than 8 years before this report. Relevant studies available since this time would have been excluded from the analyses. The authors assessed the bias of the included studies but did not explain how the results of this assessment were incorporated into the analyses. All included trials were at unclear overall risk of bias, according to the authors. It was not reported how many reviewers were involved in the risk-of-bias appraisals and whether they worked independently. As such, the risk of errors and/or biases in the appraisals themselves is uncertain. It was not clear if the authors incorporated an assessment of clinical heterogeneity into their analyses. For example, the included study by Kulnigg et al.41 allowed for a second treatment cycle in case of relapse.
There were several limitations to the NMA. First, it contained a very small amount of data from 5 studies, resulting in a network with only 1 or 2 studies connecting the nodes and wide CrIs. Only 1 outcome was assessed in the NMA. There was no analysis of harms or other outcomes important to patients (e.g., symptoms or HRQoL). Furthermore, because studies of children and adolescents with IBD were excluded, it is uncertain whether the results can be generalized to this population. Generalizability is also limited because the NMA included patients taking oral iron, and therefore patients analyzed in the included trials may differ from those eligible for ferric carboxymaltose according to the Health Canada indication, which specifies that it is for the treatment of IDA “when oral iron preparations are not tolerated or are ineffective.” The inclusion criteria for the NMA did not place restrictions on the treatment dose, and doses could have been used that do not reflect those of clinical practice in Canada.
There was heterogeneity in the time point for end point evaluation, which could have biased the results in favour of 1 of the included treatment groups. Few patient characteristics were reported across the trials, challenging a thorough assessment of the plausibility of the exchangeability assumption. Interpretation of the effect estimates was limited by imprecision. The 95% CrIs for the comparisons of ferric carboxymaltose and ferric derisomaltose and iron sucrose were wide and included the potential for no difference, or that either treatment could be favoured.
Another significant limitation is related to the sponsor’s lack of clear, a priori criteria for selecting the NMA by Aksan et al. At least 2 other published NMAs investigate the relative efficacy of ferric carboxymaltose (in broader IDA populations). The methods for identifying the NMA by Aksan et al. and other candidate NMAs were not reported. There was no protocol outlining a priori how 1 or more NMAs would be selected for presentation among multiple NMAs reporting on the same population, comparisons, and outcomes. As such, there is a risk of bias in the selection of the NMA, as it is possible that it was selected based on its results.
Summary
The summary of a published NMA submitted by the sponsor was insufficient to determine whether there is a difference in efficacy for ferric carboxymaltose compared with other injectable forms of iron in patients with IDA and IBD. Important efficacy and harms outcomes were not investigated.
Studies Addressing Gaps in the Evidence From Pivotal Trials and Select RCTs
Contents within this section were informed by materials submitted by the sponsor. The following summary was validated by the review team.
This section presents additional evidence from 6 studies that address gaps in the evidence from pivotal trials and select RCTs: 1 study in pediatric patients, 1 study in patients with IDA resulting from various causes, and 4 studies comparing ferric carboxymaltose with ferric derisomaltose in patients with IDA.
The sponsor’s systematic review identified 2 trials, both in pediatric patients, that were excluded due to the comparators. Study 1VIT17044 compared ferric carboxymaltose with oral iron, while Study 1VIT18045 was a single-arm trial with no comparator. As ferric carboxymaltose is indicated in the pediatric population, Study 1VIT17044 is summarized here to provide evidence for pediatric patients. The 1VIT18045 trial was not summarized, as it lacks a randomized comparator, limiting causal conclusions.
Additionally, Study 1VIT09031, which investigated the efficacy and harms of ferric carboxymaltose compared with oral iron among adult patients with IDA secondary to various causes with inadequate response or intolerance to oral iron, was excluded from the sponsor’s systematic review because an oral iron formulation is not considered an appropriate comparator. It is summarized here to provide additional information to inform the broad indication for ferric carboxymaltose.
Moreover, an additional 4 studies are summarized here as they provide a comparison of ferric carboxymaltose and ferric derisomaltose, which is considered an appropriate comparator but not evaluated in the sponsor’s pivotal trials and select RCTs. The 4 studies were published as Zoller (2023), Emrich (2020), and Wolf (2020), the latter involving 2 trials.
Description of Studies
Characteristics of the 6 studies summarized in this section are detailed in Table 22.
The 1VIT17044 trial66 is a phase III, multicentre, randomized, active-controlled, open-label, clinical trial conducted in 30 sites across 4 countries (the US, Ukraine, Poland, and Canada). The trial enrolled 79 patients who were assigned to receive ferric carboxymaltose (n = 40) or oral iron (n = 39). The trial aimed to investigate the efficacy and safety of ferric carboxymaltose versus oral iron in pediatric patients with IDA and a documented history of inadequate response to oral iron.
The 1VIT09031 trial67 is a phase III, multicentre, randomized, active-controlled, open-label study to investigate the efficacy and safety of IV ferric carboxymaltose in adult patients with IDA who had an unsatisfactory response or intolerance to oral iron. Cohort assignment was based on results from a 14-day run-in period with oral iron. Patients with an inadequate response to oral iron (hemoglobin increase < 1 g/dL) were assigned to cohort 1, and patients who were intolerant of oral iron were assigned to cohort 2. Oral iron was the comparator arm in cohort 1, and other IV iron SOC per investigator’s choice was the comparator in cohort 2. In cohort 1, a total of 246 patients received ferric carboxymaltose and 253 received oral iron. In cohort 2, a total of 253 patients received ferric carboxymaltose and 245 received IV SOC. Patients were followed up to day 35 for efficacy assessment, and up to day 120 for safety assessment.
Zoller (2023)68 is a multicentre, randomized, double-blind, active-controlled, clinical trial conducted at 20 outpatient hospital clinics in Austria, Denmark, Germany, Sweden, and the UK. The trial compared the incidence of hypophosphatemia after treatment with ferric carboxymaltose (N = 48 patients) versus ferric derisomaltose (N = 49 patients) in 97 patients with IDA and IBD.
Emrich (2020)69 is a prospective, single-centre, double-blind study. The study randomized 26 women with IDA to receive ferric carboxymaltose (N = 13) or ferric derisomaltose (n = 13). This trial aimed to assess hypophosphatemia after high-dose iron repletion with ferric carboxymaltose and ferric derisomaltose.
Wolf (2020)70 reports on 2 identically designed, open-label, randomized clinical trials that aimed to assess the effects of ferric carboxymaltose and ferric derisomaltose on hypophosphatemia. In trial A, 123 patients were randomized to receive ferric carboxymaltose (N = 61 patients) or ferric derisomaltose (N = 62 patients). In trial B, 122 patients were randomized to receive ferric carboxymaltose or ferric derisomaltose (N = 61 patients in each group). In total, 122 patients were randomized to receive ferric carboxymaltose and 123 to receive ferric derisomaltose.
Table 22: Details of Studies Addressing Gaps in the Evidence From Pivotal Trials and Select RCTs
Detail | 1VIT17044 | 1VIT09031 | Zoller | Emrich | Wolf |
|---|---|---|---|---|---|
Study design | Phase III, multicentre, randomized, active-controlled, open-label trial | Phase III, multicentre, randomized, active-controlled, open-label trial | Randomized, multicentre, active-controlled, double-blind, clinical trial | Prospective, single-centre, active-controlled, double-blind trial | 2 identically designed, active-controlled, open-label, randomized clinical trials |
Enrolled, N | 79 (40 in the FCM group and 39 in the oral iron group) | A total of 1,497 patients received oral iron for a run-in, and of these, 699 were enrolled into the study; a further 312 patients did not undergo the run-in and were enrolled in cohort 2 directly | 97 patients randomized into 2 groups: 49 in FD and 48 in FCM | 26 randomized into 2 groups: 13 in FCM and 13 in FD | In trial A, 123 were randomized (62 in FD and 61 in FCM) In trial B, 122 were randomized (61 to FD and 61 to FCM) In total, 123 patients were randomized to receive FD and 122 to receive FCM |
Key inclusion criteria | Aged between 1 and 17 years with a hemoglobin level < 11 g/dL, ferritin < 300 ng/mL, and TSAT < 30%; only patients with inadequate response to oral iron therapy for at least 8 weeks before randomization were eligible | Aged ≥ 18 years with IDA of any etiology, hemoglobin ≤ 11 g/dL, serum ferritin ≤ 100 ng/mL or ≤ 300 ng/mL when TSAT ≤ 30%, and a demonstrated unsatisfactory response or intolerance to oral iron | Aged ≥ 18 years with IBD and IDA (hemoglobin < 130 g/L and serum ferritin ≤ 100 ng/mL), intolerant of or unresponsive to oral iron, or a clinical need to administer iron rapidly, body weight ≥ 50 kg, glomerular filtration rate ≥ 65 mL/min/1.73 m2, serum phosphate > 2.5 mg/dL | Adult women with iron deficiency anemia (hemoglobin < 12 g/dL, serum ferritin ≤ 100 ng/mL or a serum ferritin ≤ 300 ng/mL and (TSAT) ≤ 30%) because of uterine bleeding in whom oral iron repletion was not tolerated or efficient | In both trial A and trial B: adults aged 18 and older with IDA, hemoglobin level of 11 g/dL or less and serum ferritin level of 100 ng/mL or less with a history of intolerance to 1 month or more of oral iron |
Key exclusion criteria | Pregnant or lactating with anemia due to reasons other than iron deficiency | Required dialysis for CKD; had ALT or AST > 1.5 times the upper limit of normal; or who were pregnant | Anemia due to factors other than IDA, hemochromatosis or other iron-storage disorder, or IV iron use within 30 days before screening, hemoglobin levels ≥ 100 g/L with a body weight < 70 kg, hypersensitivity to any component in FD or FCM | Advanced CKD, hypophosphatemia (plasma phosphorus < 2.5 mg/dL), pregnant, lactating, hemochromatosis, untreated hyperparathyroidism, active malignancy, known bronchial asthma or atopic dermatitis, alcohol or drug abuse, history of a psychological illness or seizures, previous treatment with IV iron substitution within the previous 30 days, or a known hypersensitivity to any IV iron preparations | In both trial A and trial B: a body weight less than 50 kg, estimated glomerular filtration rate < 65 mL/min/1.73 m2, serum phosphate < 2.5 mg/dL, acute bleeding > 500 mL within 72 hours before study inclusion, hemochromatosis or other iron-storage disorder, alcohol or drug abuse, or IV iron use within 30 days before screening |
Intervention | FCM administered as either an undiluted IV push at a rate of 100 mg (2 mL per minute or in ≤ 250 mL of normal saline) and infused over 15 minutes; a dose of FCM at 15 mg/kg to a maximum single dose of 750 mg (whichever was smaller) on days 0 and 7 for a maximum total dose of 1,500 mg | In both cohorts: FCM 15 mg/kg to a maximum of 750 mg per dose on day 0 and 7 (maximum total dose 1,500 mg) | FCM, single 20-minute IV infusion of 1,000 mg at baseline (day 0) and, depending on the a priori calculated iron dose, either 500 mg or 1,000 mg at day 35 | FCM 20 mg/kg up to a maximum of 1,000 mg | In both trial A and trial B: FCM 750 mg, infused on days 0 and 7 |
Comparator(s) | Oral ferrous sulphate daily for 28 days to a maximum daily dose of 130 mg of elemental iron < 12 years: 6 mg (elemental iron)/kg/day divided into 2 daily doses, drops or elixir Infants and children (ages 1 to < 4 years): oral ferrous sulphate drops Children (ages ≥ 4 to < 12 years): oral ferrous sulphate elixir or oral ferrous sulphate tablets ≥ 12 to 17 years (adolescents): ferrous sulphate tablet (65 mg of elemental iron per tablet per dose) twice a day | In cohort 1: oral ferrous sulphate tablets, 325 mg, 3 times daily In cohort 2: IV SOC iron at the investigator’s discretion | FD, single 20-minute IV infusion of 1,000 mg at baseline (day 0) and, depending on the a priori calculated iron dose, either 500 mg or 1,000 mg at day 35 | FD 20 mg/kg up to a maximum of 1,000 mg | In both trial A and trial B: in FD groups, 1,000 mg, infused on day 0 |
Primary end point | Change in hemoglobin from baseline to day 35 | Change in cohort 1 from baseline to the highest observed hemoglobin any time between baseline and day 35 or time of intervention The primary composite safety end point was the proportion of patients reporting any of the following: all-cause mortality, nonfatal myocardial infarction, nonfatal stroke, unstable angina requiring hospitalization, congestive heart failure, arrhythmias, protocol-defined hypotensive events, or protocol-defined hypertensive events | Incidence of hypophosphatemia (serum phosphate < 2.0 mg/dL) at any time from baseline to day 35, after all patients had received 1,000 mg of either iron formulation | Incidence of hypophosphatemia (plasma phosphorus < 2.0 mg/dL) | Incidence of hypophosphatemia (serum phosphate < 2.0 mg/dL) at any time from baseline to day 35 |
Secondary end points | Change in serum ferritin and TSAT from baseline to day 35 Safety outcomes included frequency and severity of TEAEs | Supportive efficacy end points in cohort 1 included:
All these measures in cohort 2, including mean change from baseline to highest observed hemoglobin, were supportive efficacy end points. | Incidence of hypophosphatemia at any time through day 70, changes from baseline to each post randomization visit in hemoglobin, ferritin, transferrin saturation, various laboratory measures, and FACIT-F score adverse events | Changes in plasma phosphorus and some other laboratory measures, measures of cardiac function, quality of life, functional impairment, and fatigue level | Secondary efficacy end points:
Secondary safety end points:
Post hoc analyses included study site–adjusted analyses of the primary end point, and the prevalence of hypophosphatemia at each postrandomization visit |
Publications | None | Onken (2014) | Zoller (2023) | Emrich (2020) | Wolf (2020) |
ALT = alanine transaminase AST = aspartate transaminase; CKD = chronic kidney disease; FACIT-F = Functional Assessment of Chronic Illness Therapy–Fatigue; FCM = ferric carboxymaltose; FD = ferric derisomaltose; HUB = heavy uterine bleeding; IBD = inflammatory bowel disease, IDA = iron deficiency anemia; SOC = standard of care; TEAE = treatment-emergent adverse event; TOI = time of intervention; TSAT = transferrin saturation.
Sources: Clinical Study Reports for 1VIT1704466 and 1VIT09031,67 Onken (2014),71 Zoller (2023),68 Emrich (2020),69 Wolf (2020),70 and Sponsor’s Summary of Clinical Evidence.37
Study 1VIT17044
Statistical Analysis
All efficacy analyses were based on the ITT population, which included all patients who received at least 1 dose of the study drug, had a baseline hemoglobin measurement, and at least 1 postbaseline measurement. Patients were analyzed in the group to which they were randomized. The treatment group difference for change in hemoglobin, change in ferritin, and change in TSAT was assessed with an ANCOVA, using treatment and randomization strata (hemoglobin and age categories) as fixed factors and the baseline value of hemoglobin, ferritin, and TSAT as a covariate.
Missing values were imputed using the LOCF. An MMRM was conducted as a sensitivity analysis, assuming data were missing at random. There was a single primary comparison. Supportive and/or secondary analyses were not adjusted for multiple comparisons.
For the primary outcome (change in hemoglobin from baseline), subgroup analyses were conducted for the following subgroups:
baseline hemoglobin (less than 10 g/dL; 10 g/dL or greater)
age (1 year to less than 12 years; 12 years or greater to 17 years).
Results
Patient Disposition
In the 1VIT17044 trial, 39 patients (98%) in the ferric carboxymaltose group and 37 (95%) in the oral iron group completed the study. ████ ███ ████ in the ferric carboxymaltose group and ████ ███ ████ in the oral iron group discontinued the study.
Baseline Characteristics
The baseline characteristics of 1VIT17044 trial are summarized in Table 23. Notable imbalances were evident in patient characteristics at baseline. There were fewer Hispanic or Latino patients in the ferric carboxymaltose group compared with the oral iron group ████ ███ ████. There were also fewer patients with an obese BMI (██ ███ ███) and more patients in the normal-weight BMI category ████ ███ ████ in the ferric carboxymaltose group compared with the oral iron group. Mean baseline ferritin was lower in the ferric carboxymaltose group █████ ng/mL) compared with the oral iron group (█████ ng/mL).
Table 23: Summary of Baseline Characteristics of Patients in the 1VIT17044 Trial
Characteristic | 1VIT17044 trial | |
|---|---|---|
FCM (n = 40) | Oral iron (n = 39) | |
Age (years), mean (SD) | ████ ██████ | ████ ██████ |
Age group, n (%) | ||
≥ 12 to ≤ 17 | ██ ██████ | ██ ██████ |
≥ 1 to < 12 | ██ ██████ | ██████ |
Female, n (%) | ██ ██████ | ██ ██████ |
Hispanic or Latino, n (%) | ██ ██████ | ██ ██████ |
Weight (kg), mean (SD) | ████ ██████ | ████ ██████ |
BMI (kg/m2), mean (SD) | ████ █████ | ████ █████ |
Normal-weight BMI | ██ ██████ | ██ ██████ |
Obese BMI | █████ | ██████ |
Hemoglobin (g/dL), mean (SD) | ████ ██████ | ████ ██████ |
Ferritin (ng/mL), mean (SD) | ████ ███████ | █████ ███████ |
TSAT, mean (SD), % | ███ ██████ | ███ ██████ |
BMI = body mass index; FCM = ferric carboxymaltose; SD = standard deviation; TSAT = transferrin saturation.
Note: Data are presented as mean (SD) or n (%)
Sources: Clinical Study Report for 1VIT1704466 and sponsor’s Summary of Clinical Evidence.37
Exposure to Study Treatments
In the 1VIT17044 trial, ██ █████ patients were exposed to at least 1 treatment dose. The mean amount of iron administered in the ferric carboxymaltose group was ██████ ██ ███████, with █████ of the patients receiving 2 doses. In the oral iron group, the mean amount of iron administered was ██████ ██ ██████). No additional iron preparations were allowed in the 1VIT17044 study. However, patients were allowed to receive ESAs, provided they were already on stable ESA therapy for at least 8 weeks before screening and that no ESA dosing or product changes were anticipated for the length of the trial. All the patients ██████ in the ferric carboxymaltose group and █████ in the oral iron group reported prior use of medications, most commonly ferrous sulphate (█████ in the ferric carboxymaltose group and █████ in the oral iron group). █████ of patients in the ferric carboxymaltose group and █████ in the oral iron group reported concomitant medications. Salbutamol sulphate was the most common concomitant medication and was used in ████ of patients in the ferric carboxymaltose group and ████ in the oral iron group.
Efficacy
In the 1VIT17044 trial, the least square mean change in hemoglobin from baseline to day 35 obtained through the ANCOVA model were ████ g/dL (standard error [SE] = █████) in the ferric carboxymaltose and ████ g/dL (SE = █████) in the oral iron group. The treatment difference at day 35 was ████ g/dL (95% CI, ██████ ████; P = ██████). The results of the MMRM model and the subgroup analyses align with those of the main analysis. The least square mean change in ferritin from baseline to day 35 obtained through the ANCOVA model were ██████ ng/mL (SE = ██████) in the ferric carboxymaltose and █████ ng/mL (SE = ██████) in the oral iron group. The treatment difference at day 35 was ██████ ng/mL (95% CI, ██████ ██████; P < ██████). The least square mean change in TSAT from baseline to day 35 obtained through the ANCOVA model were ██████ (SE = █████) in the ferric carboxymaltose and █████ (SE = █████) in the oral iron group. The treatment difference at day 35 was ██████ (95% CI, █████ █████; P < ██████).
Harms
In the 1VIT17044 trial, a larger percentage of participants in the ferric carboxymaltose group than in the oral iron group experienced at least 1 TEAE ██ ███████ versus ██ ██████, respectively). Numerically more patients in the ferric carboxymaltose group than the oral iron group experienced metabolism and nutrition disorders ████ ███ ███ and hypophosphatemia ████ ███ ████ ███████████ █████ ████████ ██ the ferric carboxymaltose group than the oral iron group experienced gastrointestinal disorders █████ ███ ██████ and constipation ███ ███ ██████. ███ ███████ in the ferric carboxymaltose group experienced a TEAE that led to treatment discontinuation. ████ ██ ███ ████████ experienced any SAE or any TEAE leading to death. Details are listed in Table 24.
Table 24: Summary of AEs in 1VIT17044 Occurring in 5% or More of Patients in Any Treatment Group (Safety Population)
TEAE | FCM (n = 40) | Oral iron (n = 38) |
|---|---|---|
Any TEAE | ██ ██████ | ██ ██████ |
Metabolism and nutrition disorders | ██████ | ██ |
Hypophosphatemia | ██████ | ██ |
Infections and infestations | █████ | ██████ |
Nasopharyngitis | █████ | █████ |
General disorders and administration site conditions | █████ | ██ |
Investigations | █████ | █████ |
Gastrointestinal disorders | █████ | ██████ |
Vomiting | █████ | █████ |
Constipation | ██ | ██████ |
Nervous system disorders | █████ | █████ |
Headache | █████ | █████ |
Skin and subcutaneous tissue disorders | █████ | █████ |
Urticaria | █████ | ██ |
AE = adverse event; FCM = ferric carboxymaltose, TEAE = treatment-emergent adverse event.
Sources: Clinical Study Report for 1VIT1704466 and sponsor’s Summary of Clinical Evidence.37
Critical Appraisal
Internal Validity
Although the methods for randomization were likely appropriate, there is an increased risk that prognostic balance was not achieved due to the small sample size, as evidenced by imbalances in patients’ baseline disease and demographic characteristics. Notably, the baseline serum ferritin level was higher in oral iron arm. There were also baseline imbalances by ethnicity and by BMI. The effect of these differences on efficacy outcomes is unclear. Although the trial was open-label, the efficacy outcomes are objective, making it unlikely that their measurements are biased. There is a risk of bias in the reporting of subjective harms (e.g., gastrointestinal disorders and headache) because patients knew which treatment they had been assigned (e.g., it is possible that known harms could be overestimated). The authors used the LOCF method to impute missing outcomes data and conducted sensitivity analyses using MMRM under the missing-at-random assumption. Although neither method may be appropriate (LOCF may not be reflective of the true trajectory of the outcome and MMRM assumes data are missing at random, which is not possible to assess and may not be plausible), the attrition rate was low (5% or less) in each group. As such, it is unlikely that missing data would have introduced bias. The ITT analysis was appropriate for estimating the effect of assignment on the interventions. Because there were no adjustments for multiple comparisons, there is an increased risk of type I error (false positives) for statistically significant results. Although the subgroup analyses were preplanned, these were unlikely to be sufficiently powered to detect subgroup differences.
External Validity
The included patients are from age 1 to 17 years, and results are not generalizable to other age groups. Given the small sample size, it is also unlikely that the results would be broadly generalizable to all pediatric patients with IDA. As the comparator in the trial was oral iron, this study does not provide information about the efficacy or harms of ferric carboxymaltose relative to other IV iron formulations in pediatric patients. Indigenous Peoples, who are disproportionately affected by IDA, are not represented in this trial. Although the outcomes measures were appropriate, some outcomes that may be important to patients (e.g., HRQoL) were not reported.
Study 1VIT09031
Populations
A 14-day run-in period of 325-mg oral ferrous sulphate tablets (65 mg elemental iron) 3 times a day was required before cohort assignment. Patients who had an inadequate response to oral iron (hemoglobin increase < 1 g/dL) were assigned to cohort 1. Patients who were intolerant of oral iron (e.g., severe gastrointestinal AEs) or deemed unsuitable by investigator for the oral iron run-in (e.g., low hemoglobin requiring rapid correction) were assigned to cohort 2. Patients who tolerated oral iron and had a hemoglobin increase ≥ 1 g/dL, or who were inadequately (< 67%) adherent, were not randomized.
Interventions
In cohort 1, patients were stratified by IDA etiology (heavy uterine bleeding [HUB], GI disorders, and other), baseline hemoglobin, and baseline cardiovascular risk (Framingham model). Patients were randomized 1:1 to receive the following:
group A — ferric carboxymaltose 15 mg/kg to a maximum of 750 mg per dose on day 0 and 7 (maximum total dose of 1,500 mg), as an undiluted IV push at 100 mg/min
group B — oral ferrous sulphate 325 mg 3 times a day for an additional 14 days.
In cohort 2, patients were randomized to the following:
group C — ferric carboxymaltose 15 mg/kg to a maximum of 750 mg per dose on day 0 and 7 (maximum total dose 1,500 mg), as an undiluted IV push at 100 mg/min
group D — IV standard care (other IV iron: Venofer, Ferrlecit, Feraheme, INFeD, or Dexferrum).
Outcomes
The primary efficacy end point was the mean change in cohort 1 from baseline to the highest observed hemoglobin observed any time between baseline and day 35 or TOI. Supportive efficacy end points in cohort 1 included:
the proportion of patients achieving hemoglobin > 12 g/dL any time between baseline and day 35 or TOI
the mean change from baseline to highest observed ferritin any time between baseline and day 35 or TOI
the proportion of patients achieving hemoglobin > 12 g/dL and ferritin increase ≥ 160 ng/mL any time between baseline and day 35 or TOI
the proportion of patients achieving a clinically meaningful increase in hemoglobin between baseline and day 35 or TOI (i.e., ≥ 1 g/dL for CKD, ≥ 2 g/dL for HUB or GI disorders, ≥ 3 g/dL for postpartum, and ≥ 2 g/dL for others).
All these measures in cohort 2, including mean change from baseline to highest observed hemoglobin, were supportive efficacy end points.
The primary composite safety end point was the proportion of patients reporting any of the following: all-cause mortality, nonfatal myocardial infarction, nonfatal stroke, unstable angina requiring hospitalization, congestive heart failure, arrhythmias, protocol-defined hypotensive events, or protocol-defined hypertensive events. These events were adjudicated in a blinded fashion by the Clinical Events Classification Committee of the Duke Clinical Research Institute.
Patients were to return for efficacy and safety evaluations on days 7, 14, and 35. Patients were contacted by telephone on day 90 and returned on day 120 for an AE assessment.
Statistical Analysis
In the 1VIT09031 trial, the safety population consisted of patients who received a dose of randomized treatment and was used for safety analyses. No formal statistical analyses for treatment group differences in safety were performed. The primary population for evaluating all efficacy end points was the mITT population, defined as patients from the safety population who had at least 1 postbaseline hemoglobin assessment.
The superiority of group A (ferric carboxymaltose) versus group B (oral iron) for change from baseline to highest hemoglobin anytime between baseline and day 35 or TOI was assessed by an ANCOVA, using treatment and etiology of IDA (HUB, GI disorders, and other) as fixed factors and baseline hemoglobin as a continuous covariate. All statistical tests were at the 2-tailed 0.05 alpha level, unless stated otherwise. No adjustment for multiple testing was made.
Results
Patient Disposition
In the 1VIT09031 trial, 1,497 patients received oral iron as part of the run-in, and of these, ███ █████ were enrolled into the study. A further ███ patients did not undergo the run-in and were enrolled in cohort 2 directly. A total of ████ patients were randomized. Across treatment groups, ██ ██ ███ of patients did not complete the treatment phase and ██ ██ ███ did not complete the study as scheduled. The sponsor stated that the failure of the majority █████ of patients to complete the treatment phase could be due to selection criteria or adherence reasons. For the majority █████ of patients who did not complete the study as scheduled, the reason was study adherence. Details are provided in Table 25.
Table 25: Patient Disposition in the 1VIT09031 Trial
Disposition | Cohort 1 | Cohort 2 | ||
|---|---|---|---|---|
FCM | Oral iron | FCM | IV SOC | |
Randomized | ███ | ███ | ███ | ███ |
Treated (safety population) | ███ | ███ | ███ | ███ |
mITT population,a n (%) | ███ ██ | ███ ███ | ███ ███ | ███ ███ |
Completed treatment phase (screening to day 35), n (%) | ███ ███ | ███ █ | ███ █ | ███ █ |
Did not complete treatment phase, n (%) | ██ ████ | ██ ████ | ██ ████ | ██ ████ |
Patients with interventions during days 0 to 35b, n (%) | ██████ | ██████ | ██████ | ███████ |
Completed study as scheduled (screening to day 120), n (%) | ██ █████ | ██ █████ | ██ ████ | ██ ████ |
Did not complete study as scheduled, n (%) | ██ █████ | ██ █████ | ██ ████ | ██ █ ███ |
FCM = ferric carboxymaltose; mITT = modified intention-to-treat; SOC = standard care.
aAll patients in the safety population had at least 1 postbaseline hemoglobin assessment.
bPatients could have had multiple interventions.
Sources: Health Canada Review Report72 and Clinical Study Report for 1VIT09031.67
Baseline Characteristics
The baseline characteristics of 1VIT09031 trial are summarized in Table 26. Most patient characteristics appeared balanced at baseline; however, patients in the ferric carboxymaltose group of cohort 2 had a higher mean ferritin (25.9 ng/mL) compared with patients in the IV iron group (14.9 ng/mL).
Approximately 67% and 69% of patients assigned to group C and group D of cohort 2, respectively, were deemed inappropriate for oral iron (low hemoglobin requiring rapid correction, defined as less than 8 g/dL or less than 9 g/dL, respectively, with evidence of cardiovascular or pulmonary dysfunction).
Exposure to Study Treatments
The majority ███████ of patients in group D received Venofer as the IV SOC. The next most frequently given IV SOC was iron dextran ██████. Details are provided in Table 27.
Table 26: Summary of Baseline Characteristics in the 1VIT09031 Trial
Characteristic | Cohort 1 | Cohort 2 | ||
|---|---|---|---|---|
FCM (N = 246) | Oral iron (N = 253) | FCM (N = 253) | IV SOC (N = 245) | |
Age (years), mean (SD) | ███ ████ | ███ ████ | ███ █████ | ████ ████ |
Sex, n (%) | ||||
Female | ███ █████ | ███ █████ | ███ █████ | ███ █████ |
Male | ██ ██████ | ██ █████ | ██ █████ | ███████ |
Weight (kg), mean (SD) | ██████ ██ | ██████ ██ | ██████ █ | ██████ █ |
IDA etiology, n (%) | ||||
Heavy uterine bleeding | ████ ████ | ████ ██ | ███████ | ████ ███ |
Gastrointestinal disorder | ██ ██████ | ██ ██████ | ██ ██████ | ██ ██████ |
Other | ██ ██████ | ███ ███ | ██ ██████ | ██ ██████ |
Baseline hemoglobin (g/dL), mean (SD) | ████ ████ | ████ ████ | ███ ████ | ███ ████ |
Baseline TSAT (%), mean (SD) | ████ ████ | ████ █ | ████ ██ | ███ ██ |
Baseline ferritin (ng/mL), mean (SD) | ████ ████ | ████ █ | ████ █ | ████ ████ |
Erythropoietin use, n (%) | ||||
No | ███ █████ | ███ █████ | ███ █████ | ███ █████ |
Yes | ██ | ██ | ██████ | ██████ |
Prior iron therapy, n (%) | ||||
No | ████████ | ████████ | ████████ | ████████ |
Yes | ███ ███ | ███ ███ | ███ ███ | ███ ███ |
History of iron intolerance, n (%) | ||||
No | ████ ███ | ████ ███ | ████ ███ | ████ ███ |
Yes | ██████ | ██████ | ██ █████ | ██ █████ |
Cardiovascular risk category, n (%) | ||||
0 or 1 | ███ ███ | ███ ███ | ███ ██ | ███ ███ |
2 or 3 | ██ █████ | ██ █████ | ██ █████ | ██ █████ |
FCM = ferric carboxymaltose; IDA = iron deficiency anemia; SD = standard deviation; SOC = standard care; TSAT = transferrin saturation.
Sources: Health Canada Review Report72 and Clinical Study Report for 1VIT09031.67
Table 27: Summary of Exposure to Treatment in 1VIT09031
Extent of exposure | Group A: FCM | Group B: oral | Group C: FCM | Group D: IV SOC |
|---|---|---|---|---|
Total dose of iron (mg) | ||||
Mean (SD) | ███████ | ███████ | ███████ | ███████ |
Median (range) | ███████ | ███████ | ███████ | ███████ |
Minimum to maximum | ███████ | ███████ | ███████ | ███████ |
Max single injection (mg) | ||||
Mean (SD) | ███████ | ███████ | ███████ | ███████ |
Median (range) | ███████ | ███████ | ███████ | ███████ |
Minimum to maximum | ███████ | ███████ | ███████ | ███████ |
Total number of injections, n (%) | ||||
1 | ███████ | ███████ | ███████ | ███████ |
2 | ███████ | ███████ | ███████ | ███████ |
≥ 3 | ███████ | ███████ | ███████ | ███████ |
FCM = ferric carboxymaltose; SOC = standard care; SD = standard deviation
Sources: Health Canada Review Report72 and Clinical Study Report for 1VIT09031.67
Efficacy
For the protocol-specified primary treatment group comparison (cohort 1), the mean increases in hemoglobin from baseline to the highest value between baseline and day 35 or TOI were 1.57 g/dL (SD = ████) in group A (ferric carboxymaltose) and 0.80 (SD = ████) in group B (oral iron) (P = 0.001). No between-group difference and CI were reported. In a post hoc comparison of group C (ferric carboxymaltose) versus group D (IV SOC) (cohort 2), the mean increases in hemoglobin from baseline to the highest value between baseline and day 35 or TOI were 2.90 g/dL (SD = ████) in group C (ferric carboxymaltose) and 2.16 (SD = ████) in group D (IV SOC). No between-group difference and CI were reported. Details are provided in Table 28.
Table 28: Summary of Primary End Points in the 1VIT09031 Trial (mITT Population)
Hemoglobin (g/dL) | Cohort 1 | Cohort 2 | ||
|---|---|---|---|---|
FCM (N = 244) | Oral iron (N = 251) | FCM (N = 245) | IV SOC (N = 237) | |
Baseline mean (SD) | ████ █████ | ████ █████ | ███ ██████ | ███ ██████ |
Highest value mean (SD) | ████ █████ | ████ █████ | ████ █████ | ████ █████ |
Change to highest value mean (SD) | 1.57 (████) | 0.80 (████) | 2.90 (████) | 2.16 (████) |
P value | 0.001 | 0.001a | ||
FCM = ferric carboxymaltose; mITT = modified intention-to-treat; SOC = standard care; SD = standard deviation.
aPost hoc comparison; P values were not adjusted for multiple comparisons.
Sources: Health Canada Review Report72 and Clinical Study Report for 1VIT09031.67
Subgroup analysis revealed the mean increase in hemoglobin from baseline to the highest value between baseline and day 35 or TOI was greater for the ferric carboxymaltose group than the comparator group regardless of baseline hemoglobin value or etiology of IDA.
In cohort 1, the proportion of patients achieving a hemoglobin level above 12.0 g/dL was █████ in the ferric carboxymaltose group and █████ in the oral iron group (P < ████). The proportions of patients with a clinically meaningful increase in hemoglobin (as defined by the investigators) were █████ in the ferric carboxymaltose group and ████ in the oral iron group (P < ████). The proportions of patients with a hemoglobin level above 12 g/dL and ferritin increase of 160 ng/mL or greater were █████ in the ferric carboxymaltose group and ████ in the oral iron group (████). The means of changes in hemoglobin were ███ g/dL (SD = ███) in the ferric carboxymaltose group and ███ g/dL (SD = ███) in the oral iron group (████). The means of changes in ferritin were █████ ng/mL (SD = █████) in the ferric carboxymaltose group and ████ ng/mL (SD = ████) in the oral iron group (████). The means of changes in TSAT were █████ (SD = ████) in the ferric carboxymaltose group and █████ (SD = ████) in the oral iron group (████). Between-group differences and CIs were not reported for any outcome.
In cohort 2, the proportions of patients achieving a hemoglobin level greater than 12.0 g/dL were █████ in the ferric carboxymaltose group and █████ in the IV iron group (P < ████). The proportions of patients with a clinically meaningful increase in hemoglobin (as defined by the investigators) were █████ in the ferric carboxymaltose group and █████ in the IV iron group (████). The proportions of patients with a hemoglobin level above 12 g/dL and a ferritin increase of 160 ng/mL or greater were █████ in the ferric carboxymaltose group and ████ in the IV iron group (████). The means of changes in hemoglobin were ███ g/dL (SD = ███) in the ferric carboxymaltose group and ███ g/dL (SD = ███) in the IV iron group (████). The means of changes in ferritin were █████ ng/mL (SD = █████) in the ferric carboxymaltose group and ████ ng/mL (SD = █████) in the IV iron group (████). The means of changes in TSAT were ██████ (SD = ████) in the ferric carboxymaltose group and ████ (SD = ████) in the oral iron group (████). Between-group differences and CIs were not reported for any outcome. Table 29 provides more details.
Table 29: Summary of Secondary End Points in 1VIT09031 (mITT Population)
Secondary efficacy end point | Cohort 1 | Cohort 2 | ||
|---|---|---|---|---|
FCM (N = 244) | Oral iron (N = 251) | FCM (N = 245) | IV SOC (N = 237) | |
Hemoglobin > 12.0 g/dL, n (%) | ██████ | ██████ | ██████ | ██████ |
Clinically meaningful increase in hemoglobin, n (%)§ | ██████ | ██████ | ██████ | ██████ |
Hemoglobin > 12 g/dL and a ferritin increase ≥ 160 ng/mL, n (%) | ██████ | ██████ | ██████ | ██████ |
Change in hemoglobin (g/dL), mean (SD) | ██████ | ██████ | ██████ | ██████ |
Change in ferritin (ng/mL), mean (SD) | ██████ | ██████ | ██████ | ██████ |
Change in TSAT (%), mean (SD) | ██████ | ██████ | ██████ | ██████ |
FCM = ferric carboxymaltose; mITT = modified intention-to-treat; SOC = standard care; SD = standard deviation; TSAT = transferrin saturation.
aDefined as 1 g/dL or greater for CKD, 2 g/dL or greater for heavy uterine bleeding or gastrointestinal disorders, 3 g/dL of greater for postpartum, and 2 g/dL or greater for others; P < 0.05; P values were not adjusted for multiple comparisons.
Source: Health Canada Review Report.72
Harms
There were numerically more TEAEs in group A (█████) compared with group B (█████). Overall rates of TEAEs were similar between groups C (ferric carboxymaltose) and D (IV SOC) (██ ██ ███ across groups). The TEAEs most commonly experienced (by ≥ 5%) were nausea (████) in group A, hypophosphatemia (████) in group C, and dizziness (████) in group D. No TEAE was experienced in 5% or more of patients in group B.
█████ ██████ patients in group A (ferric carboxymaltose), ██ ██████ patients in group B (oral iron), ██ ██████ patients in group C (ferric carboxymaltose), and ██ ██████ patients in group D (IV iron) experienced at least 1 SAE during the treatment phase.
█████ ██████ patients in group A (ferric carboxymaltose) and ████ in the group B (oral iron) experienced hypersensitivity reactions. ███ ██████ patients in group C (ferric carboxymaltose) and██████ in group D (IV iron) experienced hypersensitivity reactions. Hypersensitivity reactions were either grade 2 or 3 in the ferric carboxymaltose patients, and grades 1 to 3 in the IV iron arm.
██████ ██████ patients in group A (ferric carboxymaltose) and ██████ in group B (oral iron) experienced skin and subcutaneous tissue disorders. ████████ ██████ patients in group C (ferric carboxymaltose) and ██ ██████ in group D (IV iron) experienced skin and subcutaneous tissue disorders. All TEAEs associated with skin and subcutaneous tissue disorders (erythema, pruritus, rash, maculopapular rash, and urticaria) were either grade 1 or 2.
In group A (ferric carboxymaltose), ████ of patients experienced hypophosphatemia and █████ experienced PCS low-phosphorus levels compared with ██ ███ ████ in group B (oral iron), respectively. In group C (ferric carboxymaltose), ████ of patients experienced hypophosphatemia and █████ experienced PCS low-phosphorus levels compared with ██ ███ ████ in group D (IV SOC), respectively. Hypophosphatemia was mainly grade 1 to 3 in severity, ████ █ ██████ grade 4 event. Most PCS low-phosphorus events were grade 3, with 6 grade ██ events in patients in the ferric carboxymaltose group. ██ PCS low-phosphorus events were associated with serious or severe AEs. ███ patient had events of somnolence, fatigue, tingling finger, swollen hand, and an elevated white blood cell count on days when phosphorus levels reached grade 4.
In total ██████ reported in the study, ██ group B (oral iron), ██ group C (ferric carboxymaltose), and ███ in group D (IV iron). ████ ████ were considered related to the study drug by investigator. Details are provided in Table 30.
Primary Composite Safety End Point
█████ ████████ ███████ in group A (ferric carboxymaltose), ████████ ███████ in group B (oral iron), ██ ████████ ███████ in group C (ferric carboxymaltose), and ██ ████████ ███████ in group D (IV SOC) met the primary composite safety end point. The most common component of the primary composite safety end point was protocol-defined hypertensive events in group A (ferric carboxymaltose; █████), death due to any cause in group B (oral iron; █████), protocol-defined hypertensive events in group C (ferric carboxymaltose; █████), and protocol-defined hypertensive events in group D (IV SOC; █████).
Table 30: Summary of AEs in 1VIT09031 (Safety Population)
Adverse event | Cohort 1 | Cohort 2 | ||
|---|---|---|---|---|
FCM (N = 246) | Oral iron (N = 253) | FCM (N = 253) | IV SOC (N = 245) | |
Any TEAE, n (%) | ████████ | ████████ | ████████ | ████████ |
TEAE, grade 3 to 5, n (%) | ||||
Grade 3 | █████████ | ██ █████ | ███ ██ | ███ ███ |
Grade 4 | ██████ | ██████ █ | ██████ | ██████ |
Grade 5 | ██████ | ██████ | ██████ | ██████ |
Most common (≥ 5.0%) adverse events, n (%) | ||||
Nausea | ██ ██████ | ██████ | ██ ██████ | ██ ██████ |
Hypophosphatemia | ██ ██████ | ██ | ██ ██████ | ██ |
Dizziness | ██████ | ██████ | ██████ | ██ ██████ |
Serious adverse events, n (%) | ||||
Any SAE, n (%) | ██████ | ██ ██████ | ██ ██████ | ██ ██████ |
Discontinuation due to adverse events, n (%) | ||||
Premature discontinuation from study drug due to AE, n (%) | ██████ | ██ | ██ ██████ | ██ ██████ |
Discontinuation from study due to AE, n (%) | ██████ | ██████ | ██████ | ██████ |
Deaths, n (%) | ||||
Patients who died | ██ | █████ | █████ | █████ |
Adverse events of special interest, n (%) | ||||
Hypersensitivity, n (%) | ██████ | ██ | ██████ | ██████ |
Skin and subcutaneous tissue disorders, n (%) | ██ ██████ | ██████ | ██ ██████ | ██ ██████ |
Hypophosphatemia, n (%) | ██ ██████ | ██ | ██ ██████ | ██ |
PCSa low phosphorus, n (%) | ███████ ██ | ██████████ | ██████ ██ | █████████ |
AE = adverse event; FCM = ferric carboxymaltose; PCS = potentially clinically significant; SAE = serious adverse event; TEAE = treatment-emergent adverse event; SOC = standard of care.
aPCS is defined as having baseline value within normal limits (2.2 mg/dL to 5.2 mg/dL) and decreased to a value defined as grade 3 (< 2.0 mg/dL to 1.0 mg/dL) or grade 4 (< 1.0 mg/dL).
Sources: Health Canada Review Report72 and Clinical Study Report for 1vit09031.67
Critical Appraisal
Internal Validity
Although the methods for randomization and allocation concealment appeared adequate, there were imbalances at baseline in some important clinical characteristics. Most notably, serum ferritin was higher in group C (ferric carboxymaltose) compared with group D (IV SOC). It is not certain if this imbalance may have biased the results. Although the trial was open-label, the efficacy outcomes are objective, and it is unlikely that bias was introduced in their measurement. There is a risk of bias in the reporting of subjective harms because patients knew which treatment they had been assigned (e.g., it is possible that known harms could be overestimated). Across groups, up to ███ of patients did not complete the study, and the methods used to handle missing data are not clear. As such, there is a risk of bias due to missing outcomes data, but the extent and direction of the bias cannot be predicted. The mITT analysis was appropriate for estimating the effect of assignment to the interventions. Although the mITT population was a subset of the ITT population, few patients (less than 4% across groups) were excluded from the mITT analysis set. Because there were no adjustments for multiple comparisons, there is an increased risk of type I error (false positives) for statistically significant results. Between-group differences and CIs were not reported for any outcome, precluding judgments about the precision of the effects.
External Validity
In this study, all patients received 2 doses of 15 mg/kg, up to a maximum single dose of 750 mg and a maximum cumulative dose of 1,500 mg. These values were all lower than the dosing recommended in the product monograph, which is a maximum single relative dose of 20 mg/kg, a maximum single absolute dose of 1,000 mg, and a maximum total dose of 2,000 mg. In group D, patients could have been assigned to any of 5 IV iron formulations, only 2 of which were considered relevant comparators for this review. As such, the generalizability of the results may be limited. Further, the comparison of group A (ferric carboxymaltose) to group B (oral iron) does not inform about the efficacy and harms of ferric carboxymaltose relative to other IV oral formulations available in Canada, limiting the applicability of these results. The study was conducted in 84 centres in the US only, with no patients from Canada and no representation of Indigenous Peoples, who are disproportionately affected by IDA. As only adults were enrolled in the study, it is uncertain whether the results could be generalized to pediatric patients. Further, for group A (ferric carboxymaltose) and group B (oral iron) the study selected patients based on adherence to oral iron following a run-in phase and a large proportion of these patients (███) were not enrolled. As such, the enrolled patients may not be representative of patients seen in clinical practice (as patients with lower adherence were not enrolled). Although relevant outcomes were investigated in the study, other outcomes that may be important to patients (e.g., HRQoL) were not investigated.
Zoller (2023)
Statistical Analysis
A sample size of 120 patients (60 per treatment group) was estimated to be adequate to detect a statistically significant difference between groups with a 5% alpha and 80% power, based on an assumed incidence of hypophosphatemia of 15% in the ferric derisomaltose group and 40% in the ferric carboxymaltose group. Fewer than 120 patients were ultimately enrolled, as recruitment ended early because of the COVID-19 pandemic.
The safety analysis set included all patients who received at least 1 dose of the study drug. The ITT analysis set included all randomized patients. The primary end point was analyzed using the safety analysis set, and the difference between the incidence of hypophosphatemia in the 2 groups was calculated using the Cochran-Mantel-Haenszel method with 95% CIs. The secondary safety end points, and bone-specific alkaline phosphatase were also analyzed using the safety set, while the secondary efficacy end points, and FACIT-F score were analyzed using the ITT set. A test of interaction was carried out using logistic regression to examine if a diagnosis of Crohn disease versus ulcerative colitis modified the effect of the ferric derisomaltose and ferric carboxymaltose on the incidence of hypophosphatemia. Additionally, a sensitivity analysis was included in the MMRM analysis of change in FACIT-F score for Crohn disease, ulcerative colitis, and their interactions with treatment. The longitudinal changes in FACIT-F score, biomarkers of bone and mineral homeostasis, anemia, and iron parameters were analyzed with the MMRM. No imputation of missing values was performed, except for patients with no postbaseline measurements, and the change from baseline was set to zero at the first postbaseline visit. For efficacy end points, there were no reported adjustments for multiple comparisons. AE data were presented descriptively.
Results
Patient Disposition
Out of 156 patients who were screened in this trial, 97 (62%) were enrolled. Of these, 48 patients received ferric derisomaltose (5 [10%] discontinued) and 49 patients received ferric carboxymaltose (6 [12%] discontinued). In the ferric carboxymaltose group, 49 patients were included in the safety analysis set and 48 in the ITT set. In the ferric derisomaltose group, 44 patients were included in the safety analysis set and 49 in the ITT set.
Baseline Characteristics
Baseline characteristics are summarized in Table 31. Patients’ characteristics were generally well balanced between treatment groups. The proportion of patients with ulcerative colitis was lower in the ferric carboxymaltose group compared with the ferric derisomaltose group (55% versus 67%, respectively), whereas the proportion of patients with Crohn disease was higher (45% versus 33%).
Table 31: Summary of Baseline Characteristics in Zoller (2023), Safety Analysis Set
Characteristic | Zoller (2023) | |
|---|---|---|
FD (N = 48) | FCM (N = 49) | |
Age (years), mean (SD) | 42.3 (14.1) | 41.9 (14.7) |
Sex | ||
Female, n (%) | 26 (54.2%) | 25 (51.0%) |
Male, n (%) | 22 (45.8%) | 24 (49.0%) |
Race | ||
Asian, n (%) | 6 (12.5%) | 4 (8.2%) |
White, n (%) | 39 (81.3%) | 44 (89.8%) |
Other, n (%) | 3 (6.3%) | 1 (2.0%) |
Physical characteristics | ||
Weight (kg), mean (SD) | 79.8 (15.4) | 80.6 (16.6) |
BMI (kg/m2), mean (SD) | 27.7 (5.5) | 27.5 (5.2) |
Disease characteristics and comorbidities | ||
IBD diagnosis, n (%) | ||
Crohn disease | 16 (33.3%) | 22 (44.9%) |
Ulcerative colitis | 32 (66.7%) | 27 (55.1%) |
Disease activity status | ||
Harvey-Bradshaw Index (Crohn disease), mean (SD) | 3.6 (2.4) | 3.8 (3.1) |
Partial Mayo score (ulcerative colitis), mean (SD) | 2.9 (2.4) | 2.6 (2.3) |
Fatigue | ||
FACIT-F score, mean (SD) | 22.1 (10.2) | 27.4 (12.8) |
Laboratory parameters | ||
Hemoglobin (g/L), mean (SD) | 105 (15) | 104 (14) |
Ferritin (ng/mL), mean (SD) | 9.5 (9.6) | 14.6 (28.7) |
TSAT (%), mean (SD) | 9.3 (8.4) | 7.1 (4.4) |
Phosphate (mg/dL), mean (SD) | 3.5 (0.6) | 3.5 (0.6) |
Urinary fractional excretion of phosphate (%), mean (SD) | 7.6 (3.1) | 9.4 (6.1) |
iFGF23 (pg/mL), mean (SD) | 46.6 (23.3) | 48.5 (22.9) |
Ionized calcium (mg/dL), mean (SD) | 5.1 (0.2) | 5.1 (0.2) |
Intact parathyroid hormone (pg/mL), mean (SD) | 54.9 (28.3) | 61.4 (46.6) |
25-hydroxyvitamin D (ng/mL), mean (SD) | 22.9 (10.2) | 23.7 (11.8) |
1,25-dihydroxyvitamin D (pg/mL), mean (SD) | 46.6 (14.8) | 51.8 (19.9) |
24,25-dihydroxyvitamin D (ng/mL), mean (SD) | 2.3 (1.7) | 2.3 (1.8) |
ALP (units/L), mean (SD) | 75.9 (34.8) | 73.1 (29.4) |
Bone-specific ALP (mcg/L), mean (SD) | 10.8 (3.6) | 12.4 (6.4) |
N-terminal P1NP (ng/mL), mean (SD) | 50.6 (19.8) | 74.9 (66.4) |
CTx (ng/mL), mean (SD) | 0.3 (0.2) | 0.4 (0.3) |
CRP (mg/L), mean (SD) | 9.5 (13.5) | 13.3 (30.8) |
Concomitant medication | ||
Biologic therapy, n (%)a | 20 (41.7%) | 21 (42.9%) |
Interleukin inhibitors | 2 (4.2%) | 7 (14.3%) |
TNF-alpha inhibitors | 16 (33.3%) | 16 (32.7%) |
Vedolizumab | 6 (12.5%) | 5 (10.2%) |
Glucocorticoids, n (%) | 6 (12.5%) | 3 (6.1%) |
Antiresorptive therapy, n (%)b | 4 (8.3%) | 1 (2.0%) |
Vitamin D supplements, n (%) | 6 (12.5%) | 10 (20.4%) |
Vitamin D, calcium supplements, n (%) | 21 (43.8%) | 19 (38.8%) |
ALP = alkaline phosphatase; BMI = body mass index; CRP = C-reactive protein; CTx = C-terminal collagen crosslinks; FACIT-F = Functional Assessment of Chronic Illness Therapy–Fatigue; FCM = ferric carboxymaltose; FD = ferric derisomaltose; IBD = inflammatory bowel disease; iFGF23 = intact fibroblast growth factor 23; P1NP = procollagen type 1 N-terminal propeptide; SD = standard deviation; TNF = tumour necrosis factor; TSAT = transferrin saturation.
aInterleukin inhibitors, TNF-alpha inhibitors, vedolizumab.
bBisphosphonates.
Source: Zoller et al. (2023).68
Exposure to Study Treatments
The first infusion of 1,000 mg of either ferric carboxymaltose or ferric derisomaltose was administered to all patients on day 0, except for 2 patients in the ferric derisomaltose group. To correct for the total iron deficits, patients received the second infusion on day 35. Among the patients in ferric derisomaltose group, 18.8% (9 of 48) received 1,000 mg, 66.7% (32 of 48) received 500 mg as a second infusion, and 14.6% (7 of 48) did not receive a second dose. Among the patients in ferric carboxymaltose group, 22.4% (11 of 49) received 1,000 mg, 69.4% (34 of 49) received 500 mg as the second infusion, and 8.2% (4 of 49) did not receive a second dose.
Efficacy
By day 70 (the end of the trial), levels of ferritin and TSAT increased in both treatment groups. The hemoglobin increase by day 70 was 24.9 g/L (95% CI, 21.1 to 28.8) in the ferric derisomaltose group and 25.2 g/L (95% CI, 21.3 to 29.1) in the ferric carboxymaltose group. The between-group difference and CIs were not reported for any efficacy outcome.
Both ferric derisomaltose and ferric carboxymaltose resulted in improvement in fatigue symptoms and increased FACIT-F scores (possible scores ranging from 0 to 52), and the improvements were statistically significantly greater for patients treated with ferric derisomaltose versus ferric carboxymaltose at days 35 and 49. The between-group difference and CI was not reported at any follow-up time point.
Harms
Harms results are summarized in Table 32. Numerically, more patients experienced hypophosphatemia and vitamin D deficiency in the ferric carboxymaltose group compared with those in ferric derisomaltose group (hypophosphatemia: 28.6% versus 2.1%; vitamin D deficiency: 34.7% versus 22.9%, respectively). Numerically, fewer patients experienced headaches and nausea in the ferric carboxymaltose group compared with the ferric derisomaltose group (headache: 10.2% versus 18.8%; nausea: 2.0% versus 12.5%, respectively). Discontinuation due to AEs occurred in 6.3% of patients in ferric derisomaltose group and 2.0% of patients in the ferric carboxymaltose group. There were no deaths in the trial.
Hypophosphatemia in this trial was defined as a serum phosphate level of less than 2.0 mg/dL. The primary end point was the incidence of hypophosphatemia at any time after the first dose to day 35, which was reported as 8.3% (4 of 48) in the ferric derisomaltose group and 51.0% (25 of 49) in the ferric carboxymaltose group, with an adjusted risk difference of −42.8% (95% CI, –57.1% to –24.6%; P < 0.0001) favouring ferric derisomaltose. In an analysis by diagnosis of IBD, the risk differences were 43.1% and 45.5% higher in the ferric carboxymaltose group for patients with ulcerative colitis and Crohn disease, respectively (interaction P value = 0.1948). The highest incidence of hypophosphatemia occurred within 2 weeks of treatment in both arms.
The secondary safety end point of the incidence of hypophosphatemia at any time from baseline to day 70 occurred in 12.5% of patients (6 of 48) in the ferric derisomaltose group and 59.2% of patients (29 of 49) in the ferric carboxymaltose group, with an adjusted risk difference of −46.6% (95% CI, −60.9% to –28.1%; P < 0.0001) favouring ferric derisomaltose.
According to the study’s investigators, the mean decreases in phosphate concentration from baseline after the first and second doses were significantly greater after ferric carboxymaltose infusion compared to ferric derisomaltose infusion (between-group differences and CIs were not reported). For more than 1 month after the second infusion, 4.7% (N = 2 of 43) of patients in the ferric carboxymaltose group remained hypophosphatemic. On day 70 the mean serum phosphate remained significantly lower in the ferric carboxymaltose group compared to the ferric derisomaltose group (between-group differences and CIs were not reported).
Table 32: Summary of Harms Results From Zoller (2023) Safety Analysis Set
Adverse events | Ferric carboxymaltose (N = 48) | Ferric derisomaltose (N = 49) |
|---|---|---|
≥ 1 adverse event | 44 (91.7%) | 44 (89.8%) |
Most common adverse events (≥ 10% in at least 1 treatment group), n (%) | ||
Vitamin D deficiency | 11 (22.9%) | 17 (34.7%) |
Hypophosphatemia | 1 (2.1%) | 14 (28.6%) |
Nasopharyngitis | 8 (16.7%) | 10 (20.4%) |
Headache | 9 (18.8%) | 5 (10.2%) |
Arthralgia | 7 (14.6%) | 6 (12.2%) |
Nausea | 6 (12.5%) | 1 (2.0%) |
Fatigue | 5 (10.4%) | 4 (8.2%) |
Serious adverse events, n (%) | ||
Patients with ≥ 1 serious adverse event | 5 (10.4%) | 6 (12.2%) |
Abscess neck | 1 (2.1%) | 0 (0.0%) |
Colitis ulcerative | 1 (2.1%) | 1 (2.0%) |
Deep-vein thrombosis | 1 (2.1%) | 0 (0.0%) |
Hypophosphatemia | 1 (2.1%) | 0 (0.0%) |
Intervertebral disc protrusion | 1 (2.1%) | 0 (0.0%) |
Rectal ulcer hemorrhage | 1 (2.1%) | 0 (0.0%) |
Hypersensitivity | 0 (0.0%) | 1 (2.0%) |
Intermittent claudication | 0 (0.0%) | 1 (2.0%) |
Migraine | 0 (0.0%) | 1 (2.0%) |
Non-Hodgkin lymphoma | 0 (0.0%) | 1 (2.0%) |
Subileus | 0 (0.0%) | 1 (2.0%) |
Patients who stopped treatment due to adverse events, n (%) | ||
Patients who stopped | 3 (6.3%) | 1 (2.0%) |
Deaths, n (%) | ||
Patients who died | 0 | 0 |
Source: Zoller et al. (2023).68
Critical Appraisal
Internal Validity
Although the methods for randomization appeared to be appropriate, the small sample size increased the risk that prognostic balance was not achieved, as shown by imbalances in some baseline disease and demographic characteristics. Notably, there were imbalances at baseline in IBD diagnosis (Crohn disease or ulcerative colitis). The use of some concomitant medications (e.g., interleukin inhibitors and vitamin D supplements) were also imbalanced between groups. The effect of these differences on the efficacy and safety results is uncertain. Although the absolute risk differences for hypophosphatemia among patients with Crohn disease and ulcerative colitis who were treated with ferric carboxymaltose versus ferric derisomaltose were similar (and the interaction P value was not statistically significant), the subgroups were small, and the analysis was likely not sufficiently powered to detect subgroup differences. As the trial was double-blinded and methods to maintain the blinding appeared adequate, there is likely a low risk of bias in the measurement of the outcomes.
Considering the importance of patient-reported outcomes, the use of the FACIT-F scale, which is a reliable and valid instrument for measuring fatigue in IBD, was appropriate.23 Because the trial was double-blinded, the risk of bias due to reporting subjective patient-reported outcomes was low. However, because the between-group difference with CI was not reported at any follow-up time point, it precludes judgments about the precision of the effects.
For the analysis of hypophosphatemia, 2 patients in the ferric derisomaltose group and 1 patient in the ferric carboxymaltose group did not have a postbaseline observation and were imputed as having hypophosphatemia in the primary analysis. As the proportion of patients with missing data was low for this outcome, there is likely a low risk of bias due to missing outcomes data. A post hoc sensitivity analysis, in which these patients were imputed as either being free of hypophosphatemia or were excluded, yielded results similar to those of the primary analysis.
There was no imputation of missing values in this trial except for the change from baseline in patients with no postbaseline measurements, which was set to zero at the first postbaseline visit. The attrition rates were 10% in the ferric derisomaltose group and 12.5% in the ferric carboxymaltose group; as a result, the risk of bias due to missing data is not high.
Statistical analyses in this trial were not adjusted for multiple comparisons, creating an increased risk of false-positive conclusions for statistically significant results. Particularly for efficacy outcomes (e.g., change in hemoglobin, ferritin, TSAT, and fatigue), between-group differences and CIs were not reported, making it impossible to draw meaningful conclusions about the magnitude (including clinical importance) of the estimated effects and their precision.
External Validity
All patients enrolled in this trial had IBD, and, according to the clinical expert consulted for this review, results among patients with IBD may be generalizable to the broader population of patients with IDA resulting from other causes. However, given the small sample size of the trial, the results may not be broadly generalizable.
The length of follow-up was relatively short, which limits our ability to draw conclusions about efficacy and safety over a longer period and among patients who require chronic treatment with IV iron. Although the efficacy outcomes measured were appropriate, because of insufficient reporting of between-group differences and CIs, the utility of conclusions about the magnitude of the estimated effects and their precision was limited. Indigenous Peoples, who are disproportionately affected by IDA, were not represented in this study.
In this trial, patients received a single IV infusion of 1,000 mg at baseline (day 0) and, depending on the a priori calculated iron dose, either 500 mg or 1,000 mg at day 35. According to the product monograph for ferric carboxymaltose33 a single dose should not exceed 15 mg iron/kg body weight, or 1,000 mg of iron. Based on the product monograph for ferric derisomaltose24 the allowable iron dose per infusion is limited to 20 mg iron/kg body weight. The dosage administered in this trial appears to align with those in the product monographs.
Emrich (2020)
Statistical Analysis
The 2 groups were compared using t tests for independent samples for continuous data and Fisher exact tests for categorical data.
A sample size of 30 patients in each group was estimated to achieve at least 80% power to detect a difference between groups of 40% for the primary end point (hypophosphatemia) with a 2-sided Fisher exact test and a significance level of 0.05. The proportion of patients receiving ferric carboxymaltose who would experience hypophosphatemia was estimated based on result of an earlier trial (60%).73
The initial plan was to randomize 60 patients, with an a priori interim analysis planned after 30 patients were randomized, with the option to stop the trial early if the primary end point was met. Because recruitment was slower than anticipated, the interim analysis was conducted after the inclusion of 26 patients. The trial was completed after the interim analysis.
Results
Patient Disposition
Out of 32 patients who were screened, 26 were randomized into 2 groups of ferric carboxymaltose (N = 13) and ferric derisomaltose (N = 13). One patient in the ferric carboxymaltose group who withdrew consent after receiving the study drug but before the final study visit was excluded from the analyses.
Baseline Characteristics
The baseline characteristics summarized in Table 33. There were numerical differences in baseline characteristics between the ferric carboxymaltose and ferric derisomaltose groups at baseline: the mean age was higher, the level of serum 25-hydroxyvitamin D3 was lower, and systolic blood pressure was higher. All of the patients in both groups were of European ethnicities. The proportions of patients who had previous IV iron therapy were 46.2% in the ferric carboxymaltose group and 38.5% in the ferric derisomaltose group.
Table 33: Summary of Baseline Characteristics in Emrich et al. (2020)
Characteristic | FCM (n = 13) | FD (n = 13) |
|---|---|---|
Demographics | ||
Age (years), mean (SD) | 40 (10) | 34 (11) |
Caucasian, n (%) | 13 (100) | 13 (100) |
eGFR (CKD-EPI) (mL/min/1.73 m2), mean (SD) | 102 (11) | 101 (21) |
BMI (kg/m2), mean (SD) | 26 (6) | 23 (2) |
Diabetes mellitus, n (%) | 1 (7.7) | 0 (0) |
Vital signs | ||
Systolic blood pressure (mm Hg), mean (SD) | 124 (10) | 114 (10) |
Diastolic blood pressure (mm Hg), mean (SD) | 79 (8) | 74 (10) |
Heart rate (beats per minute), mean (SD) | 71 (12) | 69 (12) |
Iron deficiency parameters | ||
Previous IV iron therapy, n (%) | 6 (46.2) | 5 (38.5) |
Hemoglobin (g/dL), mean (SD) | 10.1 (1.4) | 10.7 (1.2) |
Transferrin (mg/dL), mean (SD) | 344 (37) | 325 (34) |
Transferrin saturation (%), median (interquartile range) | 4 (3 to 7) | 5 (4 to 7) |
Ferritin (ng/mL), median (interquartile range) | 6 (4 to 8) | 8 (6 to 12) |
Hepcidin (ng/mL), median (interquartile range) | 0.9 (0.4 to 1.4) | 0.6 (0.1 to 1.1) |
Mineral metabolism parameters | ||
Plasma phosphorus (mg/dL), mean (SD) | 3.2 (0.4) | 3.6 (0.6) |
Urinary fractional phosphorus excretion (%), mean (SD) | 12 ( 6) | 14 (9) |
Plasma intact FGF23 (pg/mL), median (interquartile range) | 49 (42 to 60) | 47 (40 to 58) |
Plasma c-terminal FGF23 (RU/mL), median (interquartile range) | 385 (199 to 998) | 451 (190 to 840) |
Serum calcium (mmol/L), mean (SD) | 2.3 (0.1) | 2.3 (0.1) |
Serum parathormone (pg/mL), median (interquartile range) | 40 (37 to 55) | 40 (33 to 52) |
Serum 1.25-dihydroxyvitamin D (ng/L), mean (SD) | 55 (15) | 51 (15) |
Serum 25-hydroxyvitamin D3 (ng/mL), median (interquartile range) | 19 (10 to 22) | 24 (21 to 29) |
Alkaline phosphatase (U/L), median (interquartile range) | 74 (52 to 82) | 48 (41 to 91) |
Serum procollagen type 1 N propeptide (mcg/L), median (interquartile range) | 51 (32 to 63) | 57 (40 to 74) |
Urinary pyridinoline (mcg/g creatinine), median (interquartile range) | 176 (138 to 187) | 176 (153 to 236) |
Urinary desoxypyridinoline (mcg/g creatinine), median (interquartile range) | 36 (31 to 42) | 46 (27 to 58) |
Clinical safety parameters | ||
Left ventricular mass index (g/m2), mean (SD) | 69 (13) | 67 (10) |
Left atrial volume index (mL/m2), mean (SD) | 42 (10) | 38 (6) |
E/eʹ, mean (SD) | 6.9 (1.5) | 6.0 (1.4) |
Left ventricular ejection fraction (%), mean (SD) | 62 (8) | 61 (5) |
Heart rate (beats per minute), mean (SD) | 77 (9) | 79 (11) |
Supraventricular extrasystole (n) per 24 hours, median (interquartile range) | 2 (0 to 6) | 4 (0 to 52) |
Ventricular extrasystole (n) per 24 hours, median (interquartile range) | 8 (0 to 37) | 2 (0 to 8) |
Corrected QT interval (ms), mean (SD) | 406 (20) | 403 (22) |
QT dispersion (ms), mean (SD) | 45 (18) | 44 (17) |
CKD-EPI = Chronic Kidney Disease Epidemiology Collaboration; FCM = ferric carboxymaltose; FD = ferric derisomaltose, eGFR = estimated glomerular filtration rate; BMI = body mass index; FGF23 = fibroblast growth factor 23; E/eʹ = diastolic left ventricular function; RU = relative unit; SD = standard deviation.
Note: Continuous variables are presented as mean ± SD or median (interquartile range) as appropriate; categorial variables are presented as absolute numbers and percentage.
Source: Emrich IE, Lizzi F, Siegel JD, et al. Hypophosphatemia after high-dose iron repletion with ferric carboxymaltose and ferric derisomaltose-the randomized controlled HOMe aFers study. BMC Med. 2020;18(1):178. Available from: https://bmcmedicine.biomedcentral.com/articles/10.1186/s12916-020-01643-5 Reprinted in accordance with Creative Commons Attribution 4.0 International License (CC BY 4.0): https://creativecommons.org/licenses/by/4.0/deed.en69
Efficacy
The trial assessed quality of life using the SF-36 functional impairment by Sheehan disability scale, and fatigue by the German version of the Multidimensional Fatigue Inventory, but the results have not been published. Change in the level of hemoglobin, ferritin and TSAT were not among the trial end points and were not reported.
Harms
The primary outcome was hypophosphatemia at any postinfusion study visit. It was reported in 75% of the patients (9 of 12) in the ferric carboxymaltose group and 8% of the patients (1 of 13) in the ferric derisomaltose group, as measured at study visit 4 (days 5 to 9) (P = 0.001). At study visit 5 (days 33 to 37), 25% (3 of 12) and 8% (1 of 13) of patients in the ferric carboxymaltose and ferric derisomaltose groups, respectively, had hypophosphatemia. The between-group difference with CI was not reported at any follow-up time point.
Critical Appraisal
Internal Validity
The methods of randomization appeared to be appropriate; however, there are imbalances in some baseline characteristics that may be a result of the small sample size. Because of logistical reasons after the inclusion of 26 patients (instead of the estimated 30 to achieve 80% power), the interim analysis was conducted, and the trial was completed after interim analysis. There is therefore the possibility that the effect size may have differed if more patients had been enrolled. The lack of information in the publication makes the adequacy of the blinding unclear. Only 1 patient was excluded from the analyses, and the risk of bias from missing data is therefore low. There is an increased risk of false-positive conclusions for statistically significant results, as the statistical analyses were not adjusted for multiple comparisons. The between-group differences and CIs were not reported for the primary outcome of hypophosphatemia, which precludes making useful conclusions about the magnitude of the estimated effect and its precision.
External Validity
Given the small sample size of the trial and considering that all patients enrolled in this trial were females of European ethnicities, the results may not be broadly generalizable. The results also are not generalizable to patients with conditions that were excluded from the study, such as advanced CKD, pregnancy, ongoing lactation, untreated hyperparathyroidism, hemochromatosis, active malignancy, bronchial asthma, atopic dermatitis, active alcohol or drug abuse, or a history of a psychological illness or seizures. The length of follow-up was relatively short, which limits the usefulness of any conclusions about the safety result (hypophosphatemia) over a longer period of time. In this trial only 1 infusion was performed, which limits detection of consequences of repeated infusions.
Wolf (2020)
Outcomes
Statistical Analysis
The investigators estimated that 49 patients in each treatment group were required to detect a significant difference between groups with 80% power and alpha of 5%, assuming the incidences of hypophosphatemia were 15% for iron isomaltose and 40% for ferric carboxymaltose. To account for the uncertainty, and to acquire more safety information, 60 patients were randomized to each treatment group in both trials.
The safety datasets in both trials included all patients who received at least 1 dose of the study drug. Trial A included 63 patients in the ferric derisomaltose group and 60 patients in the ferric carboxymaltose group. Trial B included 62 patients in the ferric derisomaltose group and 57 patients in the ferric carboxymaltose group. The primary end point and all secondary safety end points were analyzed using the safety dataset. The secondary efficacy end points were analyzed based on the randomization group (ITT), which in trial A included 62 patients in the ferric derisomaltose group and 61 in the ferric carboxymaltose group, including 1 patient who erroneously received ferric derisomaltose. Trial B included 61 patients in the ferric derisomaltose group and 61 in the ferric carboxymaltose group, including 1 patient who erroneously received ferric derisomaltose.
The between-group difference for hypophosphatemia incidence was calculated using the Cochran-Mantel-Haenszel method and 95% Newcombe CIs, adjusting for randomized strata and trial in the pooled analyses. Strata were for type of underlying disease (women with IDA due to gynecological blood losses: yes or no).
In the post hoc analysis, the primary end point was analyzed by the Cochran-Mantel- Haenszel method with 95% Newcombe CIs, adjusting for individual study sites. For the primary analysis, the serum phosphate level was imputed as less than 2.0 mg/dL for the 3 patients with no postbaseline data across the 2 trials.
Longitudinal changes in anemia and iron parameters were analyzed using an MMRM with a restricted maximum likelihood–based approach. Imputation of missing values for patients without postbaseline values was applied in the mixed-model analyses, in which the change from baseline was set to zero at the first postbaseline visit. There was no adjustment for multiple comparisons.
Results
Patient Disposition
In total, 554 patients were screened for both trials, with 123 patients randomized to receive ferric derisomaltose and 122 to receive ferric carboxymaltose. Overall, 231 of 245 enrolled patients (94%) completed the trials.
Demographic and Baseline Characteristics
The mean age among both trials ranged from 42.2 years (SD = 12.9 years) to 46.3 years (SD = 11.6 years). In both trials, most patients enrolled were women with IDA due to gynecological bleeding. Most of the patients in both trials were white (range, 28 [45.2%] to 38 [63.3%]). In trial A, mean weight was 80.6 kg (SD = 16.6 kg) in the ferric derisomaltose group and 77.4 kg (SD = 20.2 kg) in the ferric carboxymaltose group. In trial B, mean weight was 90.1 kg (SD = 29.2 kg) in the ferric derisomaltose group and 84.2 kg (SD = 20.1 kg) in the ferric carboxymaltose group.
In trial A, 37 patients (58.7%) in the ferric derisomaltose group and 36 (60.0%) in the ferric carboxymaltose group were Hispanic. In trial B, 23 patients (37.1%) in the ferric derisomaltose group and 23 (40.4%) in the ferric carboxymaltose group were Hispanic.
In trial A, the mean BMI was 30.6 kg/m2 (SD = 6.1 kg/m2) in the ferric derisomaltose group and 29.6 kg/m2 (SD = 7.0) in the ferric carboxymaltose group. In trial B, the mean weight was 32.3 kg (SD = 8.6 kg) in the ferric derisomaltose group and 31.7 kg (SD = 7.9 kg) in the ferric carboxymaltose group.
In trial A, there was a gynecological cause of IDA in 41 patients (65.1%) in the ferric derisomaltose group and 42 (70.0%) in the ferric carboxymaltose group. In trial B, a gynecological cause of IDA was in 44 patients (71.0%) in the ferric derisomaltose group and 39 (68.4%) in the ferric carboxymaltose group.
In trial A, the mean hemoglobin level was 9.8 g/dL (SD = 1.3 g/dL) in the ferric derisomaltose group and 9.6 g/dL (SD = 1.3 g/dL) in the ferric carboxymaltose group. In trial B, the mean hemoglobin level was 9.6 g/dL (SD = 1.2 g/dL) in the ferric derisomaltose group and 9.3 g/dL (SD = 1.4 g/dL) in the ferric carboxymaltose group. (Data are presented for the as-randomized analysis set; the reference ranges were 11.6 g/dL to 16.4 g/dL for women aged 18 to 59 years and 12.7 g/dL to 18.1 g/dL for men aged 18 to 59 years.)
In trial A, the median ferritin level was 6.1 ng/mL (interquartile range [IQR], 2.9 ng/mL to 12.9 ng/mL) in the ferric derisomaltose group and 4.8 ng/mL (IQR, 3.1 ng/mL to 7.5 ng/mL) in the ferric carboxymaltose group. In trial B, the median ferritin level was 4.8 ng/mL (IQR, 2.8 ng/mL to 8.7 ng/mL) in the ferric derisomaltose group and 5.1 ng/mL (IQR, 2.7 ng/mL to 8.8 ng/mL) in the ferric carboxymaltose group. (Data are presented for the as-randomized analysis set; the reference ranges were 11.0 ng/mL to 306.8 ng/mL for women and 23.9 ng/mL to 336.2 ng/mL for men).
In trial A, the median TSAT was 5.6% (IQR, 3.5% to 9.7%) in the ferric derisomaltose group and 4.7% (IQR, 3.6% to 7.7%) in the ferric carboxymaltose group. In trial B, the median TSAT was 5.2% (IQR, 3.5% to 8.8%) in the ferric derisomaltose group and 4.8% (IQR, 3.2% to 9.2%) in the ferric carboxymaltose group. Data are presented for the as-randomized analysis set, calculated as (total serum iron [μmol/L] × 5.586)/(transferrin [g/L] × 100) × 70.9.
In trial A, the mean serum phosphate level was 3.3 mg/dL (SD = 0.6 mg/dL) in the ferric derisomaltose group and 3.3 mg/dL (SD = 0.5 mg/dL) in the ferric carboxymaltose group. In trial B, the mean serum phosphate level was 3.4 mg/dL (SD = 0.5 mg/dL) in the ferric derisomaltose group and 3.3 mg/dL (SD = 0.5 mg/dL) in the ferric carboxymaltose group (reference range = 2.2 mg/dL to 5.1 mg/dL).
Efficacy
The hemoglobin, ferritin, and TSAT results from trial A and B are presented in the following section.
In trial A, the hemoglobin levels in the ferric derisomaltose group versus the ferric carboxymaltose group were 9.8 g/dL (SD = 1.3 g/dL) versus 9.6 g/dL (SD = 1.3 g/dL) on day 0 and 12.1 g/dL (SD = 1.0 g/dL) versus 12.5 g/dL (SD = 1.1 g/dL) on day 35. The ferritin levels in the ferric derisomaltose group versus the ferric carboxymaltose group were 15.7 ng/mL (SD = 31.7 ng/mL) versus 11.7 ng/mL (SD = 29.4 ng/mL) on day 0 and 63.4 ng/mL (SD = 54.0 ng/mL) versus 120.6 ng/mL (SD = 103.3 ng/mL) on day 35. The TSATs in the ferric derisomaltose group versus the ferric carboxymaltose group were 16.6% (SD = 31.2%) versus 7.0% (SD = 6.7%) on day 0 and 20.8% (SD = 10.2%) versus 21.7% (SD = 9.3%) on day 35.
In trial B, the hemoglobin levels in the ferric derisomaltose group versus the ferric carboxymaltose group were 9.6 g/dL (SD = 1.2 g/dL) versus 9.3 g/dL (SD = 1.4 g/dL) on day 0 and 11.8 g/dL (SD = 1.4 g/dL) versus 12.3 g/dL (SD = 1.2 g/dL) on day 35. The ferritin levels in the ferric derisomaltose group versus the ferric carboxymaltose group were 10.5 ng/mL (SD = 13.4 ng/mL) versus 17.9 ng/mL (SD = 40.5 ng/mL) on day 0 and 66.7 ng/mL (SD = 74.2 ng/mL) versus 144.9 ng/mL (SD = 164.0 ng/mL) on day 35. The TSATs in the ferric derisomaltose group versus the ferric carboxymaltose group were 8.4% (SD = 8.1%) versus 9.2% (SD = 10.0%) on day 0 and 18.3% (SD = 10.5%) versus 23.9% (SD = 11.3%) on day 35.
No changes from baseline and between-group differences and CIs for hemoglobin, ferritin, and TSAT outcomes were reported at any time point.
Harms
A summary of AEs is presented in Table 37.
Overall, in the ferric carboxymaltose versus ferric derisomaltose groups, 27 of 60 patients (45.0%) versus 7 of 63 patients (11.1%) in trial A and 28 of 57 patients (49.1%) 14 of 62 patients (22.6%) in trial B experienced AEs. Hypophosphatemia in the ferric carboxymaltose versus ferric derisomaltose groups was reported by 12 of 60 patients (20.0%) versus 0 of 63 patients in trial A and 14 of 57 (24.6%) versus 2 of 62 (3.2%) in trial B. Decreased blood phosphorus was reported in 12 of 60 patients (20.0%) in the ferric carboxymaltose group versus 0 of 63 patients in the ferric derisomaltose group in trial A and in 7 of 57 patients (12.3%) versus 0 of 62 patients, respectively, in trial B. Parathyroid hormone increased in 1 of 60 patients (1.7%) in the ferric carboxymaltose group versus 0 of 63 patients in the ferric derisomaltose groups in trial A and in 5 of 57 patients (8.8%) versus 4 of 62 (6.5%), respectively, in trial B. Headaches were reported by 1 of 60 patients (1.7%) in the ferric carboxymaltose versus 1 of 63 patients (1.6%) in the ferric derisomaltose group in trial A and in 4 of 57 (7.0%) versus 3 of 62 (4.8%), respectively, in trial B. Nausea was reported by 4 of 60 (6.7%) patients in the ferric carboxymaltose versus 0 of 63 patients in the ferric derisomaltose group in trial A and in 4 of 57 (7.0%) versus 1 of 62 (1.6%), respectively, in trial B. Serum ferritin increased in 0 of 60 patients in the ferric carboxymaltose group versus 0 of 63 patients in the ferric derisomaltose group in trial A and 3 of 57 patients (5.3%) versus 0 of 62, respectively, in trial B.
Serious or severe hypersensitivity reactions occurred in 1 patient (0.8%) in the ferric derisomaltose group (swollen eyelid unilaterally) and in 2 patients (1.7%) in the ferric carboxymaltose group (dyspnea and swelling).
Regarding the specific adverse drug reaction, in the ferric carboxymaltose versus the ferric derisomaltose groups, 12 (20.0%) versus 0 patients, respectively, in trial A, and 7 (12.3%) versus 0, respectively, in trial B experienced decreased blood phosphorus. In the ferric carboxymaltose versus ferric derisomaltose groups, 12 (20.0%) versus 0 patients, respectively, in trial A, and 14 (24.6%) versus 2 (3.2%), respectively, in trial B experienced hypophosphatemia.
The incidence of hypophosphatemia at any time from baseline to day 35 was 7.9% in the ferric derisomaltose group compared with 75.0% in the ferric carboxymaltose group in trial A (adjusted rate difference = −67.0%; 95% CI, −77.4% to −51.5%; P < 0.001 favouring ferric derisomaltose) and in 8.1% versus 73.7%, respectively (adjusted rate difference = −65.8% (95% CI, −76.6% to −49.8%; P < 0.001 favouring ferric derisomaltose), in trial B.
Critical Appraisal
Internal Validity
Both trials A and B were open-label randomized clinical trials. Although the methods for randomization appeared appropriate, there were some imbalances in baseline characteristics, such as sex and race, and minor imbalances in ferritin and TSAT levels. These imbalances may have resulted from the small sample sizes of the included studies, which increases the risk that prognostic balance between groups may not have been achieved. The open-label nature of the study may increase the risk of bias in determining the magnitude of subjective safety outcomes. The efficacy outcomes are objective and unlikely to be at risk of bias on account of the open-label design. However, the changes from baseline and between-group differences and CIs were not reported for some of the efficacy outcomes (hemoglobin, ferritin, and TSAT), which precludes judgments about the precision of the effects. There was no adjustment for multiple comparisons, which increases the risk of type I error (false positives) for statistically significant results. There was no imputation of missing values in this trial except for the change from baseline in patients with no postbaseline measurements, which was set to zero at the first postbaseline visit. The risk of bias due to missing outcomes data is low given the low rate of attrition in both trials.
External Validity
The included patients were mostly women with IDA due to gynecological bleeding. In both trials, most of the patients were white; therefore, results may not be generalizable to a broader population. Although the outcome measures were appropriate, some outcomes that may be important to patients (e.g., HRQoL) were not reported. Patients with conditions such as alcohol or drug abuse, pregnancy or lactation, untreated hyperparathyroidism, kidney transplant, a body weight less than 50 kg, hemochromatosis or another iron-storage disorder were excluded, and the results are not generalizable to these group of patients. In this study, all patients in ferric carboxymaltose groups received 750 mg of ferric carboxymaltose on days 0 and 7, whereas those in the ferric derisomaltose group received 1,000 mg on day 0. According to the product monograph for ferric carboxymaltose, a single dose should not exceed 15 mg iron per kilogram of body weight or 1,000 mg of iron. Based on the product monograph for ferric derisomaltose, the allowable iron dose per infusion is limited to 20 mg iron per kilogram of body weight. While the dosage administered aligned with that of the product monographs, patients in the ferric carboxymaltose arm received 500 mg more iron versus those in the ferric derisomaltose arm. Additionally, the clinical outcomes associated with hypophosphatemia have not been reported. The length of follow-up was relatively short, which limits conclusions about the safety result (hypophosphatemia) over a longer period of time.
Discussion
Summary of Available Evidence
The sponsor’s systematic review identified 60 reports of 40 clinical trials. A selected sample of these trials were summarized and appraised in this report. The Pivotal Trials and Select RCTs section of this report focused on 4 studies: the 2 pivotal trials in the Health Canada review (VIT-IV-CL-015 and FERGIcor) and 2 RCTs selected in the sponsor’s submitted systematic review that were thought to represent a more heterogeneous population of patients with IDA (i.e., IDA arising from various causes): 1VIT05006 (N = 559 in a crossover design versus placebo) and VIT-IRON-2011-004 (N = 371, randomized 1:1 to either ferric carboxymaltose or iron sucrose). The pivotal trials focused on specific populations, either patients with CKD (VIT-IV-CL-015, N = 240) or with IBD (FERGIcor: N = 304), with each study randomizing patients 1:1 to either ferric carboxymaltose or iron sucrose. The 1VIT05006 trial was not designed to assess efficacy outcomes. The primary outcome in the other trials was to assess hemoglobin response, either an increase from baseline of 20 g/L (VIT-IRON-2011-004: any visit up to week 8; FERGIcor: after 12 weeks) or 10 g/L (VIT-IV-CL-015: after 4 weeks), although the latter study only performed descriptive analyses. Additionally, 6 RCTs were summarized to address gaps identified in the evidence from pivotal trials and select RCTs. One study enrolled a pediatric population (1VIT17044: N = 79 pediatric patients randomized 1:1 to ferric carboxymaltose or oral iron), 1 enrolled patients with IDA who had an unsatisfactory response or intolerance to oral iron that helped inform the Health Canada review of ferric carboxymaltose (1VIT09031, N = 1,497 patients, randomized 1:1 to either ferric carboxymaltose or oral or IV iron), and 4 studies featured ferric derisomaltose as a comparator.
With respect to the 4 RCTs included in the Pivotal Trials and Select RCTs section of this report, in the FERGIcor study, the mean age of patients was ████ ██████ years in the ferric carboxymaltose group and ████ ██████ years in the iron sucrose group, while in VIT-IV-CL-015 and in the other studies the mean age was around 40 years. Most patients in the pivotal trial and in the VIT-IV-CL-015 study were white and approximately ███ of patients were female. and in the FERGIcor study 59% were female. In the nonpivotal trials in patients with IDA arising from various causes, almost all the patients were female (approximately 90%). In the 1VIT05006 study ███ of patients were Caucasian (wording used in the source) and in the VIT-IRON-2011 to 04 study all patients were Chinese. Overall mean hemoglobin levels were ████ ███ in the VIT-IV-CL-015 study, █████ ████ in the FERGIcor study, █████ ███ in Study 1VIT05006, and less than ██ ███ in VIT-IRON-2011-004.
The sponsor submitted a summary of indirect evidence from a published NMA that compared the effects of ferric carboxymaltose with those of other IV iron therapies in adults with IDA associated with IBD.22 The authors included 5 RCTs in the NMA. The interventions in the studies included ferric carboxymaltose, iron sucrose, ferric derisomaltose, and oral iron. No clear conclusions could be drawn from this NMA because of methodological limitations and imprecision, as well as the potential for bias in its selection among multiple NMAs available in the published literature.
Interpretation of Results
Efficacy
Results from the 2 pivotal trials suggest that, in a population of patients with IDA secondary to CKD and a population of patients with IDA secondary to IBD, there were no clear, clinically meaningful differences between ferric carboxymaltose and iron sucrose for various key lab parameters such as hemoglobin and TSAT. There was a statically significant improvement in the number of patients with a hemoglobin response for ferric carboxymaltose over iron sucrose in 1 of the pivotal studies but not in the other, and this was also true with respect to patients with a TSAT response, although there was no multiplicity control for this outcome in these studies. Because between-group differences with CIs were infrequently reported, it was not possible to judge the precision of the between-group effects (whether the CIs included clinically important effects favouring either of the IV iron formulations compared is unknown). The clinical expert consulted for this review believed the IBD population would be more likely to be generalizable to populations with IDA secondary to diverse etiologies. The clinical expert also noted that, in many cases, ferric carboxymaltose could be seen as simply another option among several formulations of IV iron; however, there were some subpopulations for which ferric carboxymaltose may be able to address an unmet need, such as pediatric and pregnant patients. In an additional trial, conducted in a more generalizable IDA population (i.e., patients with IDA due to any cause), the results suggested that there was no clinically meaningful difference between ferric carboxymaltose and iron sucrose with respect to hemoglobin response. The studies reviewed provided only limited evidence on the effect of ferric carboxymaltose compared with iron sucrose on HRQoL, with the study in patients with IBD finding no important differences between the groups. However, it is unclear whether the measurement scales used in the study were appropriate for estimating changes in HRQoL associated with IV iron therapy, and no between-group differences and CIs were reported to inform the precision of the effects. Hospitalizations, an important clinical outcome in IDA, were not assessed in any of the trials that were focused on in this review.
The clinical expert consulted for this review believed that 1 of the greatest unmet needs when it comes to iron supplementation is in pregnancy. The clinical expert noted that there is often a need for a parenteral form of iron supplementation in pregnancy, particularly late in pregnancy, because oral supplements are often not tolerated, or iron stores must be replenished on a more urgent basis. The clinical expert noted that existing parenteral iron therapies have limited evidence for use in pregnancy, and therefore clinicians face a dilemma of having a patient who needs parenteral iron but limited options that are proven safe and effective. The product monograph for ferric derisomaltose states that it should not be used during pregnancy, whereas in the absence of well-controlled clinical studies, the product monograph for iron sucrose notes that it should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. With respect to ferric carboxymaltose, 5 active-controlled RCTs that compare ferric carboxymaltose to iron sucrose in pregnancy have been submitted to CDA-AMC as part of the sponsor’s systematic review (Appendix 1, Table 43).74-78 The sponsor only reported these studies in a very cursory manner; however, it does appear that ferric carboxymaltose may be efficacious in managing IDA in this population. The product monograph for ferric carboxymaltose notes that a careful assessment of benefits and risks should be considered before using it during a pregnancy, and that its use should be limited to week 16 and beyond, with careful monitoring of the fetus when ferric carboxymaltose is being administered.
Similarly, the clinical expert identified iron supplementation in pediatrics as another unmet need that could potentially be filled by ferric carboxymaltose. The clinical expect noted that clinicians are reluctant to administer an IV treatment to a child. Nevertheless, there are instances in which rapid replenishment of iron stores is needed, or cases involving a child who cannot tolerate an oral iron preparation. In the 1VIT17044 trial, which compared ferric carboxymaltose to oral iron in a population of patients aged between 1 and 17 years, there was no indication of a clear and clinically meaningful difference between ferric carboxymaltose and oral iron when it came to hemoglobin; however, there was some indication of an increased improvement in ferritin and TSAT for ferric carboxymaltose compared with oral iron after 35 days. There were more patients in the ferric carboxymaltose group than in the oral iron group who experienced at least 1 AE (███ ██████ ███), however there were no SAEs or deaths.
Although iron sucrose was used as the comparator in the pivotal trials and is, overall, the most common comparator used when assessing ferric carboxymaltose, there are other comparators of potential interest, including ferric derisomaltose and sodium ferric gluconate. With respect to ferric derisomaltose, 5 RCTs were identified in the sponsor’s systematic review, featuring patients with IDA secondary to uterine bleeding, IBD, postpartum IDA, and 2 studies in patients with IDA arising from various causes who were unresponsive to or intolerant of oral iron. Four of the 5 studies focused on the risk of hypophosphatemia as a primary outcome, and the results from these 4 studies are described in the discussion of harms in the next section, and in more detail in the Summary of Studies Addressing Gaps section of this report. Only 1 of the 4 studies assessed lab values, as a secondary outcome, and found no difference between ferric carboxymaltose and ferric derisomaltose in impact on hemoglobin, ferritin, or TSAT. Sodium ferric gluconate was not included as a comparator in the sponsor’s systematic review and the clinical expert consulted on this review noted that it only plays a niche role in management of IDA.
Harms
As noted, 4 RCTs compared ferric carboxymaltose to ferric derisomaltose, using the incidence of hypophosphatemia as their primary outcome. The results from all the RCTs suggest that the risk of hypophosphatemia is statistically higher in patients treated with ferric carboxymaltose than in those treated with ferric derisomaltose. For example, in a pair of studies by Wolf et al.,70 in 245 patients with IDA arising from various causes, approximately 74% of patients treated with ferric carboxymaltose developed hypophosphatemia versus 8% treated with ferric derisomaltose.70 In the RCTs that were the focus of this review, where hypophosphatemia and/or low blood phosphorous were reported, numerically more patients treated with ferric carboxymaltose experienced this event compared to those treated with iron sucrose. The loss of phosphorous appears to be due to enhanced urinary excretion, mediated by a hormone called iFGF23 that promotes this excretion.69 The clinical consequences of the hypophosphatemia are less clear. While there is evidence that very low phosphorous can lead to clear detrimental effects in severely ill patients, the impact of moderately low phosphorous on otherwise healthy patients is uncertain.79 There have been long-term cohort studies that suggest prolonged hypophosphatemia may have a negative impact on bone health, although these findings need to be confirmed in a prospective, controlled study.68 The product monograph notes the risk of hypophosphatemia and hypophosphatemic osteomalacia and fractures, the latter reported in the post-marketing period. The fact that some of these fractures required surgery is also noted in the product monograph, and patients are encouraged to seek medical advice if they experience arthralgia or bone pain. The product monograph identifies potential risk factors for hypophosphatemia, such as vitamin D deficiency, calcium and phosphate malabsorption, secondary hyperparathyroidism, hereditary hemorrhagic telangiectasia, IBD, and osteoporosis. Monitoring for hypophosphatemic osteomalacia is recommended for patients who receive multiple doses of ferric carboxymaltose for long-term treatment and who have any of these underlying risk factors, and it is recommended that serum phosphate levels be checked in patients at risk of low serum phosphate who require a repeat course of treatment within 3 months.
As part of the economic submission, the sponsor leveraged the indirect treatment comparison by Han et al. (2023)64 to support an assumption that ferric carboxymaltose is noninferior to ferric derisomaltose with respect to harms, citing a relative risk of 0.45 (95% CI, 0.14 to 1.45). This indirect comparison was not summarized in the sponsor’s Summary of Clinical Evidence because of the higher levels of statistical, clinical, and methodological heterogeneity compared to the comparison by Aksan (which did not report on harms). Notwithstanding the potential for other methodological limitations, the 95% CI for the effect estimate is imprecise (i.e., it includes the potential that either treatment could be favoured), precluding a conclusion of noninferiority or similarity. Further, the analysis of a summary-level harm does not inform on the specific risk of hypophosphatemia with ferric carboxymaltose compared with ferric derisomaltose. As such, the clinical review team relied on the available direct evidence to inform about this risk.
Other notable AE that tends to be associated with IV iron supplementation, according to the clinical expert, includes infection and various hypersensitivity reactions. There was no indication in the included trials of any clear increase in risk of these AEs for ferric carboxymaltose when compared to iron sucrose. Therefore, hypophosphatemia appears to be the main adverse effect associated with the use of ferric carboxymaltose.
Conclusion
Results from 2 phase III pivotal trials in patients with IDA secondary to IBD and to CKD, and another phase III RCT selected because it included patients with more cases of IDA arising from various causes, suggest that ferric carboxymaltose is at least as effective at increasing hemoglobin as iron sucrose after 4 to 12 weeks of follow-up. None of the included studies were designed to formally assess other efficacy outcomes, such as TSAT and ferritin, or important outcomes, such as HRQoL; however, there was no indication in the included trials of a notable of clinically significant difference between ferric carboxymaltose and iron sucrose for any of these outcomes. There was no indication of a clear difference in risk of AE or SAE associated with ferric carboxymaltose versus iron sucrose, with the exception of hypophosphatemia, which may occur more frequently with ferric carboxymaltose. There is evidence that ferric carboxymaltose may be efficacious and safe for use in pregnancy and in children, and the clinical expert noted that there is a shortage of parenteral iron formulations with such evidence in these populations. While the evidence indicates that the efficacy of ferric carboxymaltose is comparable to that of ferric derisomaltose, there is clearly an increased risk of hypophosphatemia with ferric carboxymaltose versus the other iron formulation. The clinical significance of this increased risk of hypophosphatemia is not known.
References
1.Numan S, Kaluza K. Systematic review of guidelines for the diagnosis and treatment of iron-deficiency anemia using intravenous iron across multiple indications. Curr Med Res Opin. 2020;36(11):1769-1782. PubMed
2.Barragán-Ibañez G, Santoyo-Sánchez A, Ramos-Peñafiel CO. Iron deficiency anaemia. Revista Médica del Hospital General de México. 2016;79(2):88-97.
3.Alberta Medical Association. Iron-deficiency anemia: clinical practice guideline [sponsor-supplied reference]. 2018.
4.Guidelines & Protocols Advisory Committee. Iron deficiency - diagnosis and management [sponsor-supplied reference]. Victoria (BC): BCGuidelines.ca; 2019: https://www2.gov.bc.ca/gov/content/health/practitioner-professional-resources/bc-guidelines/iron-deficiency.
5.Cooper M, Bertinato J, Ennis JK, Sadeghpour A, Weiler HA, Dorais V. Population Iron Status in Canada: Results from the Canadian Health Measures Survey 2012-2019. J Nutr. 2023;153(5):1534-1543. PubMed
6.Cochrane KM, Hutcheon JA, Karakochuk CD. Iron-Deficiency Prevalence and Supplementation Practices Among Pregnant Women: A Secondary Data Analysis From a Clinical Trial in Vancouver, Canada. J Nutr. 2022;152(10):2238-2244. PubMed
7.Lavoie A, Lemire M, Levesque B, Ayotte P. Determinants of iron deficiency and anemia among Nunavimmiut: results from the Qanuilirpitaa? 2017 Nunavik Health Survey. Can J Public Health. 2024;115(Suppl 1):152-167. PubMed
8.Pasricha SR, Tye-Din J, Muckenthaler MU, Swinkels DW. Iron deficiency. Lancet. 2021;397(10270):233-248. PubMed
9.Clark SF. Iron Deficiency Anemia. Nutr Clin Pract. 2008;23(2):128-141. PubMed
10.Ganz T. Hepcidin and iron regulation, 10 years later. Blood. 2011;117(17):4425-4433. PubMed
11.Karaskova E, Pospisilova D, Velganova-Veghova M, et al. Importance of Hepcidin in the Etiopathogenesis of Anemia in Inflammatory Bowel Disease. Dig Dis Sci. 2021;66(10):3263-3269. PubMed
12.Cançado RD, Munoz M. Intravenous iron therapy: how far have we come? Rev Bras Hematol Hemoter. 2011;33(6):461-469. PubMed
13.Munoz M, Gomez-Ramirez S, Campos A, Ruiz J, Liumbruno GM. Pre-operative anaemia: prevalence, consequences and approaches to management. Blood Transfus. 2015;13(3):370-379. PubMed
14.Dirix L, Awada A, Bron D, et al. Practice guidelines on the use of erythropoiesis-stimulating agents in the treatment of chemotherapy-induced anemia. Belg J Med Oncol. 2010;4:57-64.
15.Bokemeyer C, Aapro MS, Courdi A, et al. EORTC guidelines for the use of erythropoietic proteins in anaemic patients with cancer: 2006 update. Eur J Cancer. 2007;43(2):258-270. PubMed
16.Clinical practice guidelines in oncology. Cancer and chemotherapy induced anaemia. Version 2. 2018 [sponsor-supplied reference]. National Comprehensive Cancer Network; 2017.
17.Fishbane S, Frei GL, Maesaka J. Reduction in recombinant human erythropoietin doses by the use of chronic intravenous iron supplementation. Am J Kidney Dis. 1995;26(1):41-46. PubMed
18.Macdougall IC, Tucker B, Thompson J, Tomson CR, Baker LR, Raine AE. A randomized controlled study of iron supplementation in patients treated with erythropoietin. Kidney Int. 1996;50(5):1694-1699. PubMed
19.Shepshelovich D, Rozen-Zvi B, Avni T, Gafter U, Gafter-Gvili A. Intravenous versus oral iron supplementation for the treatment of anemia in CKD: an updated systematic review and meta-analysis. Am J Kidney Dis. 2016;68(5):677-690. PubMed
20.Macdougall IC, White C, Anker SD, et al. Intravenous Iron in Patients Undergoing Maintenance Hemodialysis. N Engl J Med. 2019;380(5):447-458. PubMed
21.Mattiello V, Schmugge M, Hengartner H, von der Weid N, Renella R, on behalf of the SPHWG. Diagnosis and management of iron deficiency in children with or without anemia: consensus recommendations of the SPOG Pediatric Hematology Working Group. Eur J Pediatr. 2020;179(4):527-545. PubMed
22.Aksan A, Işık H, Radeke HH, Dignass A, Stein J. Systematic review with network meta-analysis: comparative efficacy and tolerability of different intravenous iron formulations for the treatment of iron deficiency anaemia in patients with inflammatory bowel disease. Aliment Pharmacol Ther. 2017;45(10):1303-1318. PubMed
23.Tinsley A, Macklin EA, Korzenik JR, Sands BE. Validation of the functional assessment of chronic illness therapy-fatigue (FACIT-F) in patients with inflammatory bowel disease. Aliment Pharmacol Ther. 2011;34(11-12):1328-1336. PubMed
24.Monoferric - Ferric Derisomaltose for Injection [product monograph; sponsor-supplied reference]. Pharmacosmos, A. S.; 2022.
25.Cappellini MD, Comin-Colet J, de Francisco A, et al. Iron deficiency across chronic inflammatory conditions: International expert opinion on definition, diagnosis, and management. Am J Hematol. 2017;92(10):1068-1078. PubMed
26.Tolkien Z, Stecher L, Mander AP, Pereira DI, Powell JJ. Ferrous sulfate supplementation causes significant gastrointestinal side-effects in adults: a systematic review and meta-analysis. PLoS One. 2015;10(2):e0117383. PubMed
27.Dignass AU, Gasche C, Bettenworth D, et al. European consensus on the diagnosis and management of iron deficiency and anaemia in inflammatory bowel diseases. J Crohns Colitis. 2015;9(3):211-222. PubMed
28.Cotter J, Baldaia C, Ferreira M, Macedo G, Pedroto I. Diagnosis and treatment of iron-deficiency anemia in gastrointestinal bleeding: A systematic review. World J Gastroenterol. 2020;26(45):7242-7257. PubMed
29.Pang QY, An R, Liu HL. Perioperative transfusion and the prognosis of colorectal cancer surgery: a systematic review and meta-analysis. World J Surg Oncol. 2019;17(1):7. PubMed
30.Hofmann A, Farmer S, Towler SC. Strategies to preempt and reduce the use of blood products: an Australian perspective. Curr Opin Anaesthesiol. 2012;25(1):66-73. PubMed
31.Mehta N, Murphy MF, Kaplan L, Levinson W. Reducing unnecessary red blood cell transfusion in hospitalised patients. BMJ. 2021;373:n830. PubMed
32.Nash CM, Allen VM. The use of parenteral iron therapy for the treatment of postpartum anemia. J Obstet Gynaecol Can. 2015;37(5):439-442. PubMed
33.FERINJECT - Ferric Carboxymaltose Injection [product monograph; sponsor-supplied reference]. C. S. L. Vifor; 2024.
34.Venofer - Iron Sucrose Injection [product monograph; sponsor-supplied reference]. American Regent, I. N. C.; 2023.
35.Gordon H, Burisch J, Ellul P, et al. ECCO Guidelines on Extraintestinal Manifestations in Inflammatory Bowel Disease. J Crohns Colitis. 2024;18(1):1-37. PubMed
36.Evstatiev R, Marteau P, Iqbal T, et al. FERGIcor, a randomized controlled trial on ferric carboxymaltose for iron-deficiency anemia in inflammatory bowel disease. Gastroenterology. 2011;141(3):846-853.e841-842.
37.Ferric carboxymaltose injection Sponsor Summary of Clinical Evidence [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: FERINJECT® (Ferric Carboxymaltose Injection), Dispersion, 100 mg/2 mL, 500 mg/10 mL, and 1000 mg/20 mL (each corresponding to 50 mg elemental iron/mL [as ferric carboxymaltose]), intravenous. Glattbrugg (CH): CSL Vifor; 2024 May 10.
38.Jin J, Ran Z, Noseda E, et al. A randomized, controlled, open label non-inferiority trial of intravenous ferric carboxymaltose versus iron sucrose in patients with iron-deficiency anemia in China. Front Med. 2024;18(1):98-108. PubMed
39.Clinical Study Report: 1VIT05006. A Multicenter, Randomized, Blinded, Placebo-Controlled, Cross-Over Study to Investigate the Safety and Tolerability of Intravenous VIT-45 in Patients with Iron Deficiency Anemia [internal sponsor's report]. Norristown (PA): Luitpold Pharmaceuticals, Inc.; 2006 May 16.
40.VIT-IRON-2011-004 - An Open-label, Randomised Controlled Multicentre Study to Assess the Impact of Ferric Carboxymaltose in Correcting Iron Deficiency Anaemia Compared to Venofer® (Iron Sucrose) in Chinese Subjects [sponsor-supplied reference]. St. Gallen (CH): Vifor International, Inc; 2019.
41.Kulnigg S, Stoinov S, Simanenkov V, et al. A novel intravenous iron formulation for treatment of anemia in inflammatory bowel disease: the ferric carboxymaltose (FERINJECT) randomized controlled trial. Am J Gastroenterol. 2008;103(5):1182-1192. PubMed
42.VIT-IV-CL-015 - A Multi-Centre, Controlled Phase III Study to Compare the Efficacy And Safety Of VIT-45 And Venofer® in the Treatment of Iron Deficiency Anaemia Associated With Chronic Renal Failure in Patients On Haemodialysis [sponsor-supplied reference]. St. Gallen (CH): Vifor, Inc; 2005.
43.FER-IBD-07-COR - A multi-centre, randomised, prospective, open-label study to investigate the efficacy and safety of a standardised correction dosage regimen of intravenous ferric carboxymaltose (FERINJECT®) versus iron sucrose (VENOFER®) for treatment of iron deficiency anaemia in patients with inflammatory bowel disease [sponsor-supplied reference]. St. Gallen (CH): Vifor, A. G.; 2010.
44.Haemoglobin concentrations for the diagnosis of anaemia and assessment of severity [sponsor-supplied reference]. Vitamin and Mineral Nutrition Information System. Geneva (CH): World Health Organization; 2011.
45.Harrow BS, Eaton CB, Roberts MB, Assaf AR, Luo X, Chen Z. Health utilities associated with hemoglobin levels and blood loss in postmenopausal women: the Women's Health Initiative. Value Health. 2011;14(4):555-563. PubMed
46.Go AS, Yang J, Ackerson LM, et al. Hemoglobin Level, Chronic Kidney Disease, and the Risks of Death and Hospitalization in Adults With Chronic Heart Failure. Circulation. 2006;113(23):2713-2723. PubMed
47.Strand V, Cryer B, Luo X, et al. Effect of Blood Loss on Physical Function in Arthritis Patients: A Pooled Analysis. Health Outcomes Res Med. 2011;2(1):e27-e38.
48.Guyatt G, Mitchell A, Irvine EJ, et al. A new measure of health status for clinical trials in inflammatory bowel disease. Gastroenterology. 1989;96(3):804-810. PubMed
49.Lind M, Vernon C, Cruickshank D, et al. The level of haemoglobin in anaemic cancer patients correlates positively with quality of life. Br J Cancer. 2002;86(8):1243-1249. PubMed
50.Ware JE, Jr., Sherbourne CD. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care. 1992;30(6):473-483. PubMed
51.Kell DB, Pretorius E. Serum ferritin is an important inflammatory disease marker, as it is mainly a leakage product from damaged cells. Metallomics. 2014;6(4):748-773. PubMed
52.Kumar A, Sharma E, Marley A, Samaan MA, Brookes MJ. Iron deficiency anaemia: pathophysiology, assessment, practical management. BMJ Open Gastroenterol. 2022;9(1). PubMed
53.Irvine EJ. Development and subsequent refinement of the inflammatory bowel disease questionnaire: a quality-of-life instrument for adult patients with inflammatory bowel disease. J Pediatr Gastroenterol Nutr. 1999;28(4):S23-27. PubMed
54.Pallis AG, Mouzas IA, Vlachonikolis IG. The inflammatory bowel disease questionnaire: a review of its national validation studies. Inflamm Bowel Dis. 2004;10(3):261-269. PubMed
55.Chen XL, Zhong LH, Wen Y, et al. Inflammatory bowel disease-specific health-related quality of life instruments: a systematic review of measurement properties. Health Qual Life Outcomes. 2017;15(1):177. PubMed
56.Alrubaiy L, Rikaby I, Dodds P, Hutchings HA, Williams JG. Systematic review of health-related quality of life measures for inflammatory bowel disease. J Crohns Colitis. 2015;9(3):284-292. PubMed
57.Irvine EJ, Feagan B, Rochon J, et al. Quality of life: a valid and reliable measure of therapeutic efficacy in the treatment of inflammatory bowel disease. Canadian Crohn's Relapse Prevention Trial Study Group. Gastroenterology. 1994;106(2):287-296. PubMed
58.Yarlas A, Maher S, Bayliss M, et al. The Inflammatory Bowel Disease Questionnaire in Randomized Controlled Trials of Treatment for Ulcerative Colitis: Systematic Review and Meta-Analysis. J Patient Cent Res Rev. 2020;7(2):189-205. PubMed
59.Brazier JE, Harper R, Jones NM, et al. Validating the SF-36 health survey questionnaire: new outcome measure for primary care. BMJ. 1992;305(6846):160-164. PubMed
60.Finkelstein FO, van Nooten F, Wiklund I, Trundell D, Cella D. Measurement properties of the Short Form-36 (SF-36) and the Functional Assessment of Cancer Therapy - Anemia (FACT-An) in patients with anemia associated with chronic kidney disease. Health Qual Life Outcomes. 2018;16(1):111. PubMed
61.Bernklev T, Jahnsen J, Lygren I, Henriksen M, Vatn M, Moum B. Health-related quality of life in patients with inflammatory bowel disease measured with the short form-36: psychometric assessments and a comparison with general population norms. Inflamm Bowel Dis. 2005;11(10):909-918. PubMed
62.Samsa G, Edelman D, Rothman ML, Williams GR, Lipscomb J, Matchar D. Determining clinically important differences in health status measures: a general approach with illustration to the Health Utilities Index Mark II. Pharmacoeconomics. 1999;15(2):141-155. PubMed
63.Pollock RF, Muduma G. A systematic literature review and indirect comparison of iron isomaltoside and ferric carboxymaltose in iron-deficiency anemia after failure or intolerance of oral iron treatment. Expert Rev Hematol. 2019;12(2):129-136. PubMed
64.Han B, Sun W, Zhuang L. Comparison of the Efficacy and Safety of Ferric Carboxymaltose versus Ferric Derisomaltose in Correcting Iron Deficiency Anemia. Clinical Medication Journal. 2023;21(12):31-36.
65.Shea BJ, Reeves BC, Wells G, et al. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ. 2017;358:j4008. PubMed
66.Clinical Study Report: 1VIT17044. A multicenter, multinational, randomized, active-controlled study to investigate the efficacy and safety of intravenous ferric carboxymaltose in pediatric patients with iron-deficiency anemia [internal sponsor's report]. Norristown (PA): American Regent, Inc.; 2021 Jan 25.
67.Clinical Study Report: 1VIT09031. A Multi-center, Randomized, Active Controlled Study to Investigate the Efficacy and Safety of Intravenous Ferric Carboxymaltose (FCM) in Patients with Iron Deficiency Anemia (IDA) [internal sponsor's report]. Norristown (PA): Luitpold Pharmaceuticals, Inc.; 2011 Aug 22.
68.Zoller H, Wolf M, Blumenstein I, et al. Hypophosphataemia following ferric derisomaltose and ferric carboxymaltose in patients with iron deficiency anaemia due to inflammatory bowel disease (PHOSPHARE-IBD): a randomised clinical trial. Gut. 2023;72(4):644-653. PubMed
69.Emrich IE, Lizzi F, Siegel JD, et al. Hypophosphatemia after high-dose iron repletion with ferric carboxymaltose and ferric derisomaltose-the randomized controlled HOMe aFers study. BMC Med. 2020;18(1):178. PubMed
70.Wolf M, Rubin J, Achebe M, et al. Effects of iron isomaltoside vs ferric carboxymaltose on hypophosphatemia in iron-deficiency anemia: Two randomized clinical trials. JAMA. 2020;323(5):432-443. PubMed
71.Onken JE, Bregman DB, Harrington RA, et al. A multicenter, randomized, active-controlled study to investigate the efficacy and safety of intravenous ferric carboxymaltose in patients with iron-deficiency anemia. Transfusion. 2014;54(2):306-315. PubMed
72.Health Canada reviewer's report: Ferinject (ferric carboxymaltose) [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: FERINJECT® (Ferric Carboxymaltose Injection), Dispersion, 100 mg/2 mL, 500 mg/10 mL, and 1000 mg/20 mL (each corresponding to 50 mg elemental iron/mL [as ferric carboxymaltose]), intravenous. Glattbrugg (CH): CSL Vifor; 2024 May 10.
73.Wolf M, Koch TA, Bregman DB. Effects of iron-deficiency anemia and its treatment on fibroblast growth factor 23 and phosphate homeostasis in women. J Bone Miner Res. 2013;28(8):1793-1803. PubMed
74.Jain G, Bansal H. Comparative study of ferric carboxymaltose v/s iron-sucrose in iron-deficiency anemia during pregnancy in a tertiary care hospital of Kumaon region. International Journal of Academic Medicine and Pharmacy. 2023;5(3):823-827.
75.Jose A, Mahey R, Sharma JB, et al. Comparison of ferric carboxymaltose and iron sucrose complex for treatment of iron-deficiency anemia in pregnancy- randomised controlled trial. BMC Pregnancy Childbirth. 2019;19(1):54. PubMed
76.Rajwani S, Kshirsagar N, Patil SK. Randomized clinical trial of iv iron sucrose and iv ferric carboxymaltose in the treatment of moderate iron deficiency anaemia in pregnancy. International Journal of Research in Pharmaceutical Sciences. 2020;11(3):4937-4943.
77.Ranjan R, Seema. A comparative study of effectiveness and safety of ferric carboxymaltose and iron sucrose in treating iron-deficiency anemia in pregnant women. International Journal of Pharmaceutical and Clinical Research. 2023;15(3):621-628.
78.Sehrawat I, Jain D, Sharma AK, Saran M. Comparison Of ferric carboxymaltose and iron sucrose complex For treatment of iron-deficiency anemia in pregnancy: randomized controlled trial. Eur Chem Bull. 2023;121(Special issue 6):7453 –7458.
79.Amanzadeh J, Reilly RF, Jr. Hypophosphatemia: an evidence-based approach to its clinical consequences and management. Nat Clin Pract Nephrol. 2006;2(3):136-148. PubMed
80.1VIT14039 - IRON CLAD: Can Iron Lessen Anemia Due to Cancer And Chemotherapy: A Multi-Center, Randomized, Double-Blinded, Controlled Study to Investigate the Efficacy and Safety of Injectafer® (Ferric Carboxymaltose Injection) in Adults [sponsor-supplied reference]. Norristown (PA): Luitpold Pharmaceuticals, Inc; 2018.
81.American Regent Inc. NCT02453334: Can Iron Lessen Anemia Due to Cancer and Chemotherapy: A Study to Investigate the Efficacy and Safety of Injectafer® [sponsor-supplied reference]. ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2015: https://clinicaltrials.gov/show/NCT02453334.
82.Makharadze T, Boccia R, Krupa A, Blackman N, Henry DH, Gilreath JA. Efficacy and safety of ferric carboxymaltose infusion in reducing anemia in patients receiving chemotherapy for nonmyeloid malignancies: A randomized, placebo-controlled study (IRON-CLAD). Am J Hematol. 2021;96(12):1639-1646. PubMed
83.1VIT09030 - Randomized Evaluation of Efficacy and Safety of Ferric Carboxymaltose in Patients with Iron Deficiency Anemia and Impaired Renal Function [sponsor-supplied reference]. Norristown (PA): Luitpold Pharmaceuticals, Inc; 2011.
84.American Regent Inc. NCT00981045: Evaluation of Efficacy and Safety of Ferric Carboxymaltose (FCM) in Patients With Iron Deficiency Anemia and Impaired Renal Function [sponsor-supplied reference]. ClinicalTrials.gov. Bethesda (MD): U.S. National Library of Medicine; 2009: https://clinicaltrials.gov/show/NCT00981045.
85.Onken JE, Bregman DB, Harrington RA, et al. Ferric carboxymaltose in patients with iron-deficiency anemia and impaired renal function: the REPAIR-IDA trial. Nephrol Dial Transplant. 2014;29(4):833-842. PubMed
86.FER-CARS-01 - A pilot randomised double-blind controlled phase III study to compare the efficacy and safety of Ferinject® and Venofer® versus standard therapy in patients with chronic heart failure, renal failure and iron deficiency [sponsor-supplied reference]. St. Gallen (CH): Vifor, Inc; 2008.
87.Sharma A, Pehal Y, Saxena N, Agrawal S. A comparative study to evaluate safety and efficacy of intravenous iron sucrose versus ferric carboxymaltose in the treatment of iron-deficiency anemia in pregnancy. Journal of Obstetrics, Gynecology and Cancer Research. 2024.
88.Sneh R, Nitin K, Sanjay Kumar P. Randomized clinical trial of IV iron sucrose and IV ferric carboxymaltose in the treatment of moderate iron deficiency anaemia in pregnancy. International Journal of Research in Pharmaceutical Sciences. 2020;11(3):4937-4943.
89.Breymann C, Milman N, Mezzacasa A, Bernard R, Dudenhausen J, Fer-Asap investigators. Ferric carboxymaltose vs. oral iron in the treatment of pregnant women with iron-deficiency anemia: an international, open-label, randomized controlled trial (FER-ASAP). J Perinat Med. 2017;45(4):443-453. PubMed
Appendix 1: Detailed Outcome Data
Please note that this appendix has not been copy-edited.
Table 34: Health Canada Submission Studies — Study Characteristics
Study name or number | Clinical trials registry number | First author | Geography | Phase | Study design | Population | Subgroup |
|---|---|---|---|---|---|---|---|
1VIT1403980 IRON CLAD | NCT0245333481 | Makharadze et al. (2021)82 | US | 4 | DB PC RCT MC | IDA | Cancer chemotherapy |
1VIT0903083 REPAIR-IDA | NCT0098104584 | Onken et al. (2014)85 | US | 3 | OL AC MC RCT | IDA | NDD-CKD |
FER-CARS-0186 | — | — | Russia | 3 | DB PC AC MC RCT | ID | CHF, Renal failure |
AC = active-controlled; ALT = alanine transaminase; AST = aspartate transaminase; CHF = chronic heart failure; CKD = chronic kidney disease; CO = crossover; DB = double-blind; ECOG PS = Eastern Cooperative Oncology Group Performance Status; ESA = erythropoiesis-stimulating agents; FCM = ferric carboxymaltose; GFR = glomerular filtration rate; hbAg = hepatitis B antigen; HBV = hepatitis B virus; HCV = hepatitis C virus; hsCRP = high sensitivity C-reactive protein; IBD = inflammatory bowel disease; IDA = iron deficiency anemia; IS = iron sucrose; LVEF = left ventricular ejection fraction; MC = multicentre; MCH = mean corpuscular hemoglobin; NDD-CKD = non–dialysis-dependent chronic kidney disease; NYHA = New York Heart Association; OL = open-label; PC = placebo-controlled; RBC = red blood cell; RCT = randomized control trial; TSAT = transferrin saturation.
Table 35: Health Canada Submission Studies — Inclusion Criteria
Study name or number | Enrolment criteria | Intervention, comparator, | Duration of follow-up |
|---|---|---|---|
1VIT14039 | Inclusions:
Exclusions:
| FCM in two 15-minute infusions 7 days apart, each at a dose of 15 mg/kg (maximum permitted single and total doses of 750 mg and ≤ 1,500 mg, respectively) diluted in ≤ 250 mL saline. N = 122 Placebo ≤ 250 mL of normal saline administered in two 15-minute infusions. N = 122 | 18 weeks |
1VIT09030 | Inclusions: Male or female patients ≥ 18 years of age and able to give informed consent; Chronically impaired renal function as defined by these criteria:
OR
And either 1 or both of the following: 1. Kidney damage as indicated by abnormalities in composition of urine (as documented in the patient’s medical history). 2. Elevated risk of cardiovascular disease (Category 2 or 3) based on the Framingham model. 3. Screening Visit central laboratory hemoglobin ≤ 11.5 g/dL. 4. Latest Screening Visit ferritin ≤ 100 ng/mL or ≤ 300 when TSAT was ≤ 30%. 5. If on an ESA, a stable dose (+/− 20%) for 4 weeks before randomization. Exclusions:
| FCM: 15 mg iron/kg as an undiluted IV push at a rate of 100 mg/min (maximum cumulative dose 750 mg) on days 0 and 7, for a maximum total dose of 1,500 mg. N = 1,290 IS: 200 mg of iron administered as an undiluted IV push over 2 to 5 minutes on days 0, 7 and 14, with 2 additional doses given between days 0 and 7 and between days 7 and 14, for a total of 5 doses (1,000 mg). N = 1,294 | 56 days |
FER-CARS-01 | Inclusions:
Exclusions:
| FCM: VIT-45 solution containing 50 mg/mL iron was administered without dilution, by IV bolus injections. Patients received weekly IV injections of 200 mg (or 100 mg for the last dose, if necessary) until the dose, as rounded down, was reached. Afterwards the patients were scheduled to receive 200 mg of iron every 4 weeks until week 12. N = 30 IS solution containing 20 mg/mL iron was administered without dilution, by IV injection over 10 minutes, with the same treatment regimen as FCM. N = 27 Placebo: 5 mL of normal saline solution per injection as IV bolus injections, where the treatment regimen was based on the patients’ iron deficit in analogy to the treatment regimen for FCM. N = 15 | 12 weeks plus 2 weeks follow-up |
AC = active-controlled; ALT = alanine transaminase; AST = aspartate transaminase; CHF = chronic heart failure; CKD = chronic kidney disease; CO = crossover; DB = double-blind; ECOG PS = Eastern Cooperative Oncology Group Performance Status; ESA = erythropoiesis-stimulating agents; FCM = ferric carboxymaltose; GFR = glomerular filtration rate; hbAg = hepatitis B antigen; HBV = hepatitis B virus; HCV = hepatitis C virus; hsCRP = high sensitivity C-reactive protein; IBD = inflammatory bowel disease; IDA = iron deficiency anemia; IS = iron sucrose; LVEF = left ventricular ejection fraction; MC = multicentre; MCH = mean corpuscular hemoglobin; NDD-CKD = non–dialysis-dependent chronic kidney disease; NYHA = New York Heart Association; OL = open-label; PC = placebo-controlled; RBC = red blood cell; RCT = randomized control trial; TSAT = transferrin saturation.
Table 36: Health Canada Submission Studies — Patient Baseline Characteristics
Study name or number | Age | Female sex | Race | Hb | Ferritin | TSAT % | Health status | Other baseline characteristics |
|---|---|---|---|---|---|---|---|---|
1VIT14039 | ███ ██ ███ ██ ██ ████ | ███████ ██████ █ ██ ██████ | ██████████████████████████████████ | ███████ ██████ █████ █ ████ ███████ | ████ ████ ████ ███████ | ██████ ████ ██████ | ████ ██ ██ ████ █ ██ █ ████ █ ████ ██████████ ██████ █ ████ ███ ████ █ ██ ██████████ ██ ██ ███ ██ ██████████ ██ █████ █ ██ ████ | ██████████ ██████ █████ ████ ███████ █████ █ ███ ████ █████ ██ ████ ███████ █ ███████ ███ ██████ █ ███ █████████ ██████████ |
1VIT09030 | ███ █ ███████ ██████ █ ████ ██████ | ███ █ ██████ ███████ █ ███ ███████ | ██████████████████████████████████████████████████ | ███ ██████ █████ █ █████ ███████ | ███████ █████ █ █████ ███████ | ███ █ ███████ ██████ █ █████ ███████ | ███ █ ███████ ███████ ██ ██████ █ ██ ██████████ ███ ███████ █ ████ ████████ ██ ██████ █ ███ ██████ | ██ ██████ ███ █████████ ██ ███ █████ ████ █████ ███ ███ ████ ███████ ██████ ████ ███ ████ ███ ███████ ███ ███ ███ ████ |
FER-CARS-01 | ██ █ ██ ███████ █████ █ ███ █████ █ ████ ██████ | ██ █ ██ █ ███████ ██ ██████ █ ██ ███████████ | ██████ ██ ███ █ ██ ███████ █ ██ ██████ | ██ █ ███████ ███████ ████ █ █ █████ █ ██ █████ | ██ █ ██ ██████ ███████ █████ █ ███ ████ ████ ██████ | ████ ███████ ████ █ ██████ █ ███ ████ | ████ █████████ ███████ ████ █████ █████ █ ██████ ██████████ | ██ █████████ ████ █ ███ ███████ ███ ██████ ███████ ███ ██ ████ ████ ██████ █████████ ████ ███ ███████ ████████ █ ████ ███████ ███████ ██ ██ █ ████████ ████████ █████ ███ █████ █ ███ █████ █ ██ ██████ |
BMI = body mass index; CHF = chronic heart failure; CKD = chronic kidney disease; DBP = diastolic blood pressure; dL = decilitre; ECOG PS = Eastern Cooperative Oncology Group Performance Status; ESA = erythropoiesis-stimulating agents; FCM = ferric carboxymaltose; g = grams; GFR = glomerular filtration rate; HUB = heavy uterine bleeding; IS = iron sucrose; L = litre; MI = myocardial infarction; mL = millilitres; ng = nanograms; NYHA = New York Heart Association; SBP = systolic blood pressure; SD = standard deviation; ug = micrograms.
Note that 2 of these studies submitted to Health Canada, 1VIT05006 and VIT-IRON 2011-004 are reviewed in the systematic review section of the report and are not repeated here.
Table 37: Health Canada Submission Studies — Primary and Secondary Outcomes
Study name or number | Primary outcome(s) | Secondary outcome(s) |
|---|---|---|
1VIT14039 | ██████████ ██ ████████ ████ █ ████████ ██ ██ ██ █ ███ ████ ████ █████ █ ██ ████████ ██████████████ █████████████████ ██████ ████ ██████████ ██████ ███████████ ████ ███████ █████ █████ ███████ | ██████ ██ ██ ████ ████████ ██ ████ ██ ██ ██ █████████ █████████████ ████ █████████ ████ ████████████████ ████ ██████████ ████ ██████████ █████ ████ ████████ ████████████████████ ██ ████████ ████ ██ ████████ ██ ██████ ████ ████████ ███ ███ ████ █████ ██ █████ ████ ████████ ████ ████████ ██ ████ ███████ ██████████████ ██████ ████████████████ ████ ███████████ ███████████ ███ ██████████ ████████ ██ ██ █ ███ ████ ████ ████████ ██ ████ █████ █████ ███ ███ ████ █████ ██ █████ ████ █████████ ██████ █████████ ████████████ ███ ███████████████████ ██ ████████ ███ ████████ █ █████████ ████████████ █████ ██████████████ ██████ ████████████████ ██ ████████ ███ ████████ █ █████ ████████████████ ██████████████ ██████ ████████████████ ██ ████████ ███ ███████ ██ █ ██ ████ ████ ████████ ██ ████ █████ ██████████ ██████████████ ██████ ██ ███████████████ ██ ██████ ██ ██ ████ ████████ ████████ ██████ ██████ ███████ ████████████████ ███████ ██████████████ ████ ████████ ██ ████ ██ █████████████ █████ █████ ██████ ████ █████████ ████ ██████████████ █ ██████ ██ ██ |
1VIT09030 | ███ █ ███████████ ████████ █████████ ████ ████ ████ ████████ ███ ███████ ████████ ██ ████ ██ ███ ████ ███████ █████████ ███ ███ ███ ██ █████████ ██████ ████ ███ ██ ███ ████ ██ ██████████████ ███████ █ ████ ████████ ██ ███ ███████████ ████ ██ █████████████ ██████ █████████ ██████████ ██ █████ ████████████ ████████████ ██ █████ ███ ████ ████████ ██ █ ███████ █████████ ██████ █████████████ ██ ████ ████████ █ ███ ██ ████ ██████ ███████ ███████████ ██████ ███ ██ ████████ █████ ███████████ █████████ ██████ ████████ ███████████ ███ █ ███ █ ██ █ ███ █ ██████████ ████████ ██████████ ██████ ██ ███████ █ ██ ███████ █ ██████ ████████ ██████████████ ███ █ ███████ █ █████████ █ ██████ ████████ ███████████████ ███████ █ ██████████████ █ ████ ████████ █████████ █████████ ████████████████ ██ ██████████████ █ █████ ████████ ██████████ █████████ ██████████████████████ █████████████ ██ ███████ █ ██ ███████ █ █████ ████████ ██████████████████ ██ ███████ █ ██ ███████ █ █████ ████████ ██████████████████████ ████████████ ███████ ██ ███████ █ ██ ██████ █ █████ ███████ ██████ ████████████████ ███████████ ██████ ██ ███████ █ ██ ███████ █ ██████ ████████ ███████████████ ████████ █████████ ████████████████████████ ██████ ██ ███████ █ ██ ███████ █ █████ ████████ █████████████ ███ ██ ███████ ██ ███████ █ ██ ███████ █ ██████ ████████ ██████ | ███ █ ██████████████ ██ ████████ █████████ ██ ████████ █████████████ ██ ██ ████ ███ ████ ███████ ████████ ███ ███ ███████ ██ ████ ████████ █ ███ ██ ████ ████████ ███████████ ████████ █████ ███████ █████████████ ███████ ████ ████████ ██ ███████ ████████ ████████ ███████ ███████ ████████ ███ ███ ███ ███████ █████████ █ ██████ ████████████████████ ████ ████ ██████ ████████ █████████████ ███████ ████ ████████ ██ ███████ ████████ ████ ███ ███████ ████████ ███ ███ ███ ██████ ████████ █ █████ ███████████████████ ████ ████ █████ ███████ ██████ |
FER-CARS-01 | ███ █ ██ █ ████████ █ ███████████ ██ ███ ████ ████████ ██ ████████████████ ██ ████████ ████ ███████████ ██ ███████ █ ██ ███████ █ ██ ████████████ ██████ ██████████ █ ██████████████████████████ ██████ █ █████████████████ ██████ ██████ █ █████████████████████████ ██ ███████ ███ ███ █ ███████ ███ ██ █████ ██ ██ █████ ███ ███ ██ █████████ █ ████████████████████ ██████ █████████ ██ █████████████████████████████ █████████ █ ██████ █ ██ █████████████████ █████████ █ █████████████████████████ ███ █████████████ ████████████████████ ███ ██████ ████████████████ ██ ██ ██████ ████████████████ ███ ██████ ██ ███████████ ██████ ██████ ███████████████ ██ ████ ████ ████████ ██ ████████████████ ██ ████████ ████ ███████████ ██ ███████ █ ██ ███████ █ ██ ████████████████ ██ █ ██████ █ █████████████████████████████ ██ ██████ █ ██ ██████ ███ ██████████████ ████ █████ ███ █ ███████ ███ ██ █████ ██ ██ █████ ███ ███ ██ █████████ █ ████████████████████ ██████ ██████████ █ █████████████████████████ ███ █████████████ ████████████████████ ███ ██████ ████████████████ ██ ██ ██████ ████████████████ ███ ██████ ██ ███████████ ██████ ██████ ███████ | ███ █ ██ █ ███████████████ ██ ██████████████████ ██ ████ █████ ████████ ██ ███████████ ██ ███ ██ █████ ███████ ███ █████████ ██ ████ █████ ██ ██ █████ █ █████ ██ ███ ███ ██ █████████ ████████ ██ ██████████ █ █████ ███████████████████████████ ███ █████████████ ████████████████████ ███ ██████ ████████████████ ██ ██ ██████ ████████████████ ███ ██████ ██ ███████████ ██████ ██████ ███████████████ ██ ███████ ████████ ███ ██████████ █████ ██████████ ██ ████ ████████ ████ ██████ █ ███ ████ ██████ █ ███ ████ ███████████████ ███ █████████████ ███████████ █████████ ███ ██████ ████████████████ ██ ██ ██████ ████████████████ ███ ██████ ██ ███████████ ██████ ██████ ███████████████ ██ ████ ███ ████████ ██████████ █████ ██████████ ██ ████ ████████ ████ ███████ █ ███ █████ ███████ █ ██ ██████ ████████████████ ███ █████████████ ███████████ █████████ ███ ██████ ████████████████ ██ ██████ ████████████████ ███ ██████ ███████████ ██████ ██████ ███████████████ ██ ██ ██████ ██████████ █████ ██████████ ██ ████ ████████ ███ █████ █ ███ ███ █████ ████ ████ ██████████████ ███ ███████ █████ ███████████ █████████ ███ ██████ ████████████████ ██ ██████ ████████████████ ███ ██████ ███████████ ██████ ██████ ███████████████ ██ █████ ████ ████████ ██████████ █████ ██████████ ██ ████ ████████ ███ █████ █ ███ ███ █████ █ ███ ████ ██████████████ ███ ███████ █████ ███████████ █████████ ███ ██████ ████████████████ ██ ██████ ████████████████ ███ ██████ ███████████ ██████ ██████ ███████████████ ██ █████ ████████ ██████ ██████████ █████ ██████████ ██ ████ ████████ █████ ███████ █ ███ █████ ███████ █ ███ █████ ███████████████ ███ ███████ █████ ███████████ █████████ ███ ██████ ████████████████ ██ ██████ ████████████████ ███ ██████ ███████████ ██████ ██████ ███████████████ ██ █████ ███████████ █████ ██████████ █████ ██████████ ██ ████ ████████ ████ █████ █ ███ ████ █████ █ ███ ███ ██████████████ ███ ███████ █████ ███████████ █████████ ███ ██████ ██████████████████ ██ ██████ ████████████████ ███ ██████ ███████████ ██████ ██████ ███████████████ ██ ████ ███ ██████████ █████ ██████████ ██ ████ ████████ ███ ██████ █ ███ ███ █████ █ ███ ████ ███████████████ ███ ███████ █████ ███████████ █████████ ███ ██████ █████████████████ ██ ██████ ████████████████ ███ ██████ ████████████ ██████ ██████ ██████████████ █████ █████████ ████ ████████████ ████████ ███ █████████ █████ █████████ ███ ████████████████████ █████████████████ ███████████████ ███████████████████ ██ ███ ██ █████████ ████ ████████ ██ ███████████████ █████ ███████ ████████████████████████████████ █████ |
CI = confidence interval; dL = decilitre; ECG = electrocardiograph; FACIT = Functional Assessment of Chronic Illness Therapy; FCM = ferric carboxymaltose; g = grams; IS = iron sucrose; LS = least squares; NS = not statistically significant; NYHA = New York Heart Association; OR = odds ratio; PGA = Patient Global Assessment; SD = standard deviation; TEAE = treatment-emergent adverse event; TSAT = transferrin saturation; VO2 = maximal oxygen consumption.
Note: Two of these studies were submitted to Health Canada; 1VIT05006 and VIT-IRON 2011-004 are reviewed in the systematic review section of the report and are not repeated here.
Table 38: Health Canada Submission Studies — Subgroup and Other Analyses
Study name or number | Subgroup analyses | Other analyses |
|---|---|---|
1VIT14039 | ██████ ██ ██ ██ ████████ ████ ████████ ██ ██████ ███ █████ ██ ████ █████████ ████ ███████████████ ████ ██████████████████ ████ ███████ ██████ | ████████ ████████ █████████ ███ ████████ ████████ ███████ ███████ ███████ ████ ████████ ██ ████ ██ ██ ████████ ████ ████ ███ █████ ██████████████ ████ ███████ ██████ ██████ |
1VIT09030 | ███ █ ██ █ ███ ██ ███ ██████████████████ ████████ ██ ██████ ██████ ███████ ████ ██████ ████ ████████ ██ ██ ███████ ████████ ██ ██████ ██ ███ ████ ██████ ███ ████████ ███ ███ ███ ██ █████████ ██████ ████ ███ ██ ███ ████ ██ █████████████████████ ███ ████ █████████ █████ ████ ███████ █ █████████ █ ██████ █████████████ █████ ████ ███████ █ ████ ███████ ██████ ██████████ █████ ████ ███████ █ ████ ███████ █ ████ █████████ ████ ████ ██████ ████ ███████ █ ████ ███████ █ █████ █████████ ████ ███████ █ ████ █████ █████ █████████ ██████ ████ ███████ ████ ███████ █ ████ ███████ █ ██████ █████████ ████ ███████ █ ████ ███████ █ █████ ███████ ████ ███████ █ ████ ███████ █ ██████ ████ | NR |
FER-CARS-01 | NR | NR |
CI = confidence interval; CKD = chronic kidney disease; ESA = erythropoiesis-stimulating agents; FCM = ferric carboxymaltose; IS = iron sucrose; LS = least squares; NR = not reported; SD = standard deviation; SE = standard error
Note: Two of these studies were submitted to Health Canada; 1VIT05006 and VIT-IRON 2011-004 are reviewed in the systematic review section of the report and are not repeated here.
Table 39: Health Canada Submission Studies — Discontinuations and Adverse Events
Study name or number | Discontinuations | Adverse events |
|---|---|---|
1VIT14039 | ███ █ ████████ ███████████ ████████████ ████ ██████ ██ █ ████████ ██ █ ████████████ ██ ██████ ██████████ ██ █████████ ██ █ ████████████ ████████████████ ████████ | █████████████ ██████ █████ ████████ █████ ███ ████████ ███ ███████ █ ███████ █████████ █████ ██ ██████ █ ██ ███████ ██ ███████ ████████ █████ ██████ █ ██ ██████ ████████ ██████ █ ██ ██████ ███████ ██ ██████ █ ██ ███████████ █████████ █ ██ █████████████ ██ ██████ █ ██ ██████████ ██████ ██████████ ████ ███████ ██ ██ ███████████ ██ ██████ █ ██ ██████ ███ █████ ██ █████ ████ █████████████████████ ███████ ██████ ██ █████ █████ ████████ ████ ████████ █ ██ ████████ █ ███ ██████ ██████████ ██ ██ ████ ██ ███████ ██ ██████████ ████████ ██ ██████ █ ██ ███████ ██████████ █████████ █ ███ ██ ████ ███ ███████████ █████ █ ██████████ █████████ ██████████████████ ██████ ███████████ ████ ███████████ ██ ██████ ███████ ██████ █████ █ ██ ███████ ██████████ ████ ███ █████ ████████ █ ██ █ ███████████████ ██ ██████ █ ██ ███████ ██████████ █████ ███████ ███████ ███████ █████ ██████ ████████ ██████ ███████████ ██████████ █ ███████████████████████████████████ █████ █ ████ ████████ ██████████████ ██████ ██ ███ █ ██ ███████ ████████ ██ ███ █ ██ ███████ ██████████ ██ ██████ ██ ███████ ██████████ ██ █████ █ ██ █ ████████████ ███ █████████ ████████ █ ██████ █ ██ ███████ █████████████ ██ ██████ █████ █████ ████████ █ █████████████ ██████ █████████ ██ █████ █ ██ ███████ ██████████ ██████████ █ █████ █ ██ ██████████████ ███ █████████ ██ █████ █ ██ ██████████████ ███ █████████ ████ ███████ ██ ████ █ ██ ███ ████ ██ ████████ █████ █████████████ ████████ ███ ████████ ██████ ██ █████ █ ██ ██████████ ███ ██████████ ██████ ███████ ██ ██████ █████████ █ █████ ████████████ ████████ ██ █████ ████████████ █████████ ███ ███████ ███ █████ ███ ██████ ██ ████ █ ██ ███████ ████ ███████ ███████ ███████████ ██████ █████████ █ █████ ██████████ ██████████ █ ██████████ |
1VIT09030 | ███ █ ███ ██████████ ███ ███ ███████ ██████████ ███ █ ███████████ ██████ ███████████████████████ ███ █ ███████ ██ ██████████ ██ █ ██████████ ████████ ████████████ ████████████████ ███████ | ██████████████████ ███████ ██████ ███████████ ██ ███ ██ ████████ ██ █████████ ██████ ██████ ███████████ █ ███████ ████████ █ ██ █████████ ██████████ █████ █ █████ ███ ██████ █ ███ ███████ █████████████ ██████████ ██ █████ █ ██ ██████ ██████ ███████ ███████ ███████████ ██ █████ █ ██ ██████ ██████████████████████ ██████████ ███ ██████ █ ███ ███████ ███████ █████████████ ██ █████ █ ██ ██████ ██████ ██████████ ██ █████ █ ██ ██████ ██████ ███████ ███ ██████ █ ██ ██████ ███████ █████████ ██ █████ █ ██ ██████ █████████████ █████████ ███ ██████████████ ████ ███████████ ███ ██████ █ ███ ███████ ██████ ████████ ██ █████ █ ██ ██████ ██████ █████████ ████ █████ █ █████ █ ██ ██████ ███████ ██████ ███████████ ██ █████ █ ██ ██████ ████████████████ ███ █████████████ ███ ██████ █ ███ ███████ ██████ ████████████████ ██ █████ █ ██ ██████ ██████ █████ ███████████ █████ ██████████ ██ █████ █ ██ ██████ ██████ ███████ █████ ██████████ ██ █████ █ ██ ██████ ████████████████ ███ █████████ ██████████ ███ ██████ █ ███ ██████ ███████ █████████████ ██ █████ █ ██ ██████ █████████████████████ ███ ██████████ ██████ ██████████ ███ ██████ █ ███ ██████ ██████ ████ █████ ██ █████ █ ██ ██████ █████████████ ██████ ██████████ ███ ██████ █ ███ ███████ ██████ ██████████ ██ █████ █ ██ ██████ ██████ ██████████ ██ █████ █ ██ ██████ ██████ █████████ ██ █████ █ ██ ██████ ██████████████████ ████████ ███ ███████████ ██████████ ███ ██████ █ ███ ██████ ██████ █████████ ██ █████ █ ██ ██████ ██████████████ ██████████ ███ ██████ █ ███ ███████ ███████ █████████ ██ █████ ██████ ███████ █████████████ ███ ██████ █ ██ ██████ ██████ ████████████ ██ █████ █ ██ ██████ █████ |
FER-CARS-01 | ███ █ ██ ███████████████████████ ███████ ████████ ████████ ████████ | █████ ██ █████████ ██████ ██████ ███████████ █ ███████ ██████ █ ██ ██████ █ ███████ ████████████ ██ ██████ █ ██ ██████ █ ██ ███████████████████ █████ █ ███████████████████████ █████ █ ████████████████████ ███████ █████████ ███████████████ ██ █████ █████ █ ██████████ ██████████ ███████ ██████████ ████ ██████ █ ███████████████████████ █████ █ ███████████████████████ ████ ███████ ███████ █ █████ █████████████████ █████████ ██ ██ ███████ ██ ███ █████████ ██████ █ ███████████ ██████████ █ ██████████████████ ███████ ███████ ███ █ ██████████████████████████████████ ██████████ █ ██████████████████████████ ███ █████████████ █ ████████████████████████████████ █ ██████████████████████████ ███ █████████ ██████████ █ ██████████████████ █████████████ █ █████████████████████████ ██████ ██████████ █ ███████████████████████ ███ ███████ ██████████ █ █████████████████████████ ██████████ █ ██████████████████ ████████████ ████ █ ████████████████ |
AE = adverse event; ALT = alanine transaminase; AST = aspartate transaminase; FCM = ferric carboxymaltose; GGT = gamma-glutamyl transferase; IS = iron sucrose; NOS = not otherwise specified; STEAE = serious treatment-emergent adverse event; TEAE = treatment-emergent adverse event.
Note: Two of these studies were submitted to Health Canada; 1VIT05006 and VIT-IRON 2011-004 are reviewed in the systematic review section of the report and are not repeated here.
The SLR pulled in 5 studies in pregnancy; however, they are reported in Section 5 because of the agreed upon reporting structure with CDA-AMC for Section 2 and because FCM fills an important unmet need in Canada for IDA treatment in pregnancy.
The pivotal studies included in the main body of Section 2 did not include studies on pregnant women. Additional studies are presented in this Section that investigated the efficacy and safety of FCM to treat IDA during pregnancy.
Table 40: Studies of Ferric Carboxymaltose in IDA in Pregnancy
Study description | Summary of key results |
|---|---|
Jose et al. (2019);75 Open-label, randomized trial, 100 patients, 12-week follow-up. FCM vs. IS | FCM is more effective than IS in improving hemoglobin and serum ferritin levels, and in reducing fatigue. Neither FCM nor IS are associated with serious AEs. |
Ranjan and Seema (2023);87 Randomized, prospective study, 120 patients, 3-week follow-up. FCM vs. IS | FCM is associated with a more pronounced hemoglobin increase from baseline compared to IS. Neither FCM nor IS are associated with serious AEs. |
Jain and Bansal (2023);74 Randomized trial, 100 patients, 4-week follow-up. FCM vs. IS | Both FCM and IS are associated with significant increase in hemoglobin and serum iron from baseline. Neither FCM nor IS are associated with serious AEs. |
Rajwani et al. (2020);88 Open-label trial, 160 patients, 4-week follow-up. FCM vs. IS | FCM is associated with a more pronounced hemoglobin increase from baseline compared to IS. Neither FCM nor IS are associated with serious AEs, and FCM is associated with a lower incidence of AE. |
Sehrawat et al. (2023);78 Open-label, randomized trial, 200 patients, 4-week follow-up. FCM vs. IS | FCM is more effective than IS in increasing hemoglobin and ferritin levels from baseline. FCM is associated with a lower incidence of AE. |
Breymann et al., 2016;89 Open-label, randomized trial, 252 patients, 12-week follow-up. FCM vs. oral FS | FCM is associated with a more pronounced increase in hemoglobin from baseline than oral FS. Neither FCM nor oral FS are associated with serious AEs. |
AE = adverse event; FCM = ferric carboxymaltose; FS = ferrous sulphate; IS = iron sucrose; NOS = not otherwise specified; RCT = randomized controlled trial; STEAE = serious treatment-emergent adverse event; TEAE = treatment-emergent adverse event.
Pharmacoeconomic Review
Abbreviations
BIA
budget impact analysis
CDA-AMC
Canada’s Drug Agency
CKD
chronic kidney disease
IBD
inflammatory bowel disease
IDA
iron deficiency anemia
Executive Summary
The executive summary comprises 2 tables (Table 1 and Table 2) and a conclusion.
Item | Description |
|---|---|
Drug product | Ferric carboxymaltose injection (Ferinject), 50 mg elemental iron/mL, vial for injection (2 mL, 10 mL, 20 mL) |
Indication | For the treatment of IDA in adult and pediatric patients 1 year of age and older when oral iron preparations are not tolerated or are ineffective. The diagnosis of iron deficiency must be based on laboratory tests. |
Health Canada approval status | NOC |
Health Canada review pathway | Standard |
NOC date | March 11, 2024 |
Reimbursement request | As per indication |
Sponsor | CSL Vifor |
Submission history | Previously reviewed: No |
IDA = iron deficiency anemia; NOC = Notice of Compliance.
Table 2: Summary of Economic Information
Component | Description |
|---|---|
Type of economic evaluation | Cost-minimization analysis |
Target population | Adults (aged 18 years and older), adolescents (aged 14 to 17 years), and pediatric patients (aged 1 to 13 years) with iron deficiency anemia when oral iron preparations are not tolerated or are ineffective. |
Treatment | Ferric carboxymaltose |
Dose regimen | The sponsor estimated an average cumulative iron dose (i.e., a treatment course) of 1,500 mg per adult, 1,119 mg per adolescent, and 600 mg per pediatric patient (weight-based and dependent on hemoglobin levels). A single ferric carboxymaltose administration should not exceed either 15 mg iron/kg body weight or 1,000 mg iron for adults, and should not exceed either 15 mg/kg or 750 mg iron for children and adolescents. If the total iron need (i.e., cumulative iron dose) is higher, then the administration of an additional dose should be a minimum of 7 days apart after the first dose. |
Submitted price | Ferric carboxymaltose, 50 mg elemental iron per mL, IV
|
Submitted treatment costs |
|
Comparator(s) |
|
Perspective | Canadian publicly funded health care payer |
Time horizon | Single treatment course (i.e., 1 cumulative iron dose) |
Key data source | The FERGlcor and REPAIR-IDA open-labelled, randomized control trials comparing ferric carboxymaltose to iron sucrose, a 2017 published meta-analysis (comparing ferric carboxymaltose iron sucrose, ferric derisomaltose, and oral iron in patients with inflammatory bowel disease), and 2 published indirect treatment comparisons (Pollock and Muduma [2019];1 Han et al. [2023]2) |
Costs considered | Drug acquisition costs, administration costs |
Submitted results | Ferric carboxymaltose was associated with cost-savings compared to ferric derisomaltose and iron sucrose (incremental savings of $16 and $362, respectively, per patient, per treatment course) Subgroup results (per patient, per treatment course)
|
Key limitations |
|
CDA-AMC reanalysis results | CDA-AMC did not undertake a base-case reanalysis. Given the higher rates of hypophosphatemia observed with ferric carboxymaltose, the extent of savings that will be realized with the use of ferric carboxymaltose compared to iron sucrose or ferric derisomaltose is highly uncertain. A scenario analysis including costs associated with monitoring and treating patients with nonsevere hypophosphatemia estimated that cost-savings would be reduced. Reimbursement of ferric carboxymaltose may lead to additional costs to the health care system that have not been fully considered within this analysis. |
CDA-AMC = Canada’s Drug Agency; CKD = chronic kidney disease; IBD = inflammatory bowel disease; IDA = iron deficiency anemia.
aTreatment costs include costs of drug acquisition, nurse time, infusion chair time, and infusion devices.
Conclusions
The Canada’s Drug Agency (CDA-AMC) Clinical Review included patients with iron deficiency anemia (IDA) arising from various causes and suggests that ferric carboxymaltose demonstrated equivalent efficacy when compared to ferric derisomaltose or iron sucrose. However, the assumption of equivalent safety remains highly uncertain and may be inappropriate because there may be an increased risk of hypophosphatemia with ferric carboxymaltose that may require additional monitoring and treatment. The clinical significance of this increased risk of hypophosphatemia is not known. If hypophosphatemia has a non-negligible quality-of-life impact, a cost-utility analysis would have been the preferred approach for the pharmacoeconomic analysis.
The sponsor submitted a cost-minimization analysis assuming equivalent clinical efficacy and safety among ferric carboxymaltose, iron sucrose, and ferric derisomaltose. In the submission, ferric carboxymaltose was associated with cost-savings of $16 and $362 per patient per treatment course when compared to ferric derisomaltose and iron sucrose, respectively, at public list price. However, the costs of monitoring and treating hypophosphatemia were not included by the sponsor. CDA-AMC did not undertake a base-case reanalysis as the limitations identified by CDA-AMC could not be addressed because they were related to the underlying assumption of equivalent clinical efficacy and safety between ferric carboxymaltose and its comparators and because of the variability in the administration of the total iron dose per treatment course. A scenario analysis was conducted that included monitoring and treatment costs based on assumptions and expert opinion for nonsevere hypophosphatemia. This analysis estimated that cost-savings would be reduced (incremental savings: $4 compared to ferric derisomaltose and $349 compared to iron sucrose); however, if rates of hypophosphatemia requiring treatment and monitoring are higher than assumed (10% based on CDA-AMC clinical expert opinion) or if patients develop more severe adverse events (e.g., severe hypophosphatemia, hypophosphatemia osteomalacia, and/or fractures), savings associated with the reimbursement of ferric carboxymaltose may be further reduced or eliminated. Due to the remaining uncertainty concerning the comparative safety across iron products and its associated costs, the magnitude of cost-savings associated with ferric carboxymaltose remains uncertain. All costs and incremental savings are further based on publicly available list prices and may not reflect actual prices paid by Canadian public drug plans.
Economic Review
The current review is for ferric carboxymaltose (Ferinject) for adult and pediatric patients aged 1 year and older with IDA when oral iron preparations are not tolerated or are ineffective.
Economic Information
Summary of Sponsor’s Economic Information
The sponsor submitted a cost-minimization analysis for ferric carboxymaltose compared with ferric derisomaltose and iron sucrose for the treatment of adults (aged 18 years and older), adolescents (aged 14 to 17 years), and pediatric patients (aged 1 to 13 years) with IDA when oral iron preparations are not tolerated or are ineffective.1 The reimbursement population aligns with the Health Canada–indicated population.
The sponsor assumed ferric carboxymaltose was associated with clinical efficacy and safety equivalent to those of ferric derisomaltose and iron sucrose, based on the following published literature: randomized control trials comparing ferric carboxymaltose with iron sucrose in patients with inflammatory bowel disease (IBD) (FERGlcor study)2 and chronic kidney disease (CKD) (REPAIR-IDA study),3 a 2017 published meta-analysis assessing the efficacy and tolerability of iron supplements in patients with IBD (ferric carboxymaltose, iron sucrose, ferric derisomaltose, and oral iron),4 an indirect treatment comparison by Pollock and Muduma (2019)5 comparing either ferric carboxymaltose or ferric derisomaltose to iron sucrose in patients with IBD, non–dialysis dependent CKD and heavy uterine bleeding, and an indirect treatment comparison by Han et al. (2023)6 in patients with IBD, heavy uterine bleeding, IDA during pregnancy, and patients with IDA. The analysis was conducted from the perspective of the publicly funded health payer over a time horizon corresponding with treatment duration (i.e., 1 treatment course). Discounting was not applied because a treatment course is less than 1 year.
Ferric carboxymaltose is available as a single-use vial, administered intravenously (by infusion [diluted] or injection [undiluted]).7 The recommended dosage of ferric carboxymaltose depends on the hemoglobin level and weight of each patient, and can range from 150 mg to 1,500 mg for patients aged 1 to 17 years and from 500 mg to 2,000 mg for patients aged 18 years and older. In the submitted economic model, the sponsor assumed a mean weight of 76.0 kg for adults, 53.9 kg for adolescents, and 27.7 kg for the pediatric population based on the demographics of patients with IDA in published literature.8 The sponsor also assumed a mean hemoglobin level of 10.22 g/dL for all 3 age groups, derived from the average pretreatment hemoglobin level for patients with IDA treated with IV iron in 1 Albertan hospital. As a result, the assumed average cumulative iron doses were 1,500 mg per adult, 1,119 mg per adolescent, and 600 mg per child per treatment course. At the submitted price of $45.00 per 100-mg vial, the sponsor estimated that the drug costs associated with ferric carboxymaltose per treatment course to be $675 per adult, $540 per adolescent, and $270 per child. At the same cumulative iron dosages, the drug cost per treatment course of ferric derisomaltose was the same as ferric carboxymaltose: $413 per adult, $330 per adolescent, and $165 per child for each course of treatment with iron sucrose.
The sponsor also included costs related to treatment administration comprising time with the nurse, infusion chair time, and infusion devices. These costs are influenced by 3 factors: the approach used to calculate the cumulative iron dose corresponding to total iron need, the maximum iron dose per single infusion of each drug, and the minimum infusion time of each drug. To calculate the cumulative iron dose for all treatment arms in the model, the sponsor used simplified tables from the ferric carboxymaltose product monograph for adults and the Ganzoni formula for adolescents and children.7 To calculate the maximum iron dose per single infusion for adults, the sponsor assumed that a single infusion would not exceed either 15 mg/kg or 1,000 mg for ferric carboxymaltose, 20 mg/kg or 1,500 mg for ferric derisomaltose (informed by their respective its product monographs), and 300 mg for iron sucrose (to align with the CADTH review of Monoferric).7,9,10 To calculate the maximum iron dose per single infusion for children and adolescents, the sponsor assumed that a single infusion would not exceed either 15 mg/kg or 750 mg for ferric carboxymaltose (as per product monograph), 20 mg/kg or 1,000 mg for ferric derisomaltose (informed by sponsor-sought expert opinion), and either 7 mg/kg or 300 mg for iron sucrose (to align with the CADTH review of Monoferric).7,9 The minimum infusion time adopted by the sponsor was based on their respective product monographs: 6 to 15 minutes for ferric carboxymaltose (≤ 500 mg), 20 to 30 minutes for ferric derisomaltose (≤ 1,000 mg), and 90 minutes for iron sucrose irrespective of the dose (per monograph).7,9,11 The IV preparation time per infusion and monitoring time after infusion were assumed to be identical for all iron products (6 minutes and 30 minutes, respectively).12 The sponsor assumed 100% adherence and no vial sharing. When including administration costs, a treatment course with ferric carboxymaltose was estimated by the sponsor to be $800 for adults, $665 for adolescents, and $381 for children.
Base-Case Results
The sponsor presented probabilistic (based on 5,000 iterations) and deterministic results for the individual subgroups (pediatrics, adolescents, and adults) and weighted probabilistic results combining all ages to reflect the overall indicated population. Although the deterministic and probabilistic results were similar when ferric carboxymaltose was compared to iron sucrose, when compared to ferric derisomaltose, the results did not align (i.e., ferric carboxymaltose was cost-saving in the probabilistic analysis and more costly in the deterministic analysis). This is because, across each probabilistic run, changes to the mean patient weight and hemoglobin level results in more variability in the number of infusions needed to deliver ferric derisomaltose as it has a higher threshold for maximum iron dose per single infusion.
For the overall indicated population, the sponsor’s submitted base case estimated a total cost per treatment course with ferric carboxymaltose of $755 per patient;, whereas, the total cost per treatment course with ferric derisomaltose and iron sucrose was estimated to be $771 and $1,117 per patient, respectively (Table 3). Based on the sponsor’s results, treatment with ferric carboxymaltose resulted in estimated cost-savings of $16 and $362 per patient per treatment course compared with ferric derisomaltose and iron sucrose, respectively. Results were similar across patient subgroups (compared to ferric derisomaltose: incremental savings of $15, $11, and $29 for adults, adolescents, and children, respectively; compared to iron sucrose: incremental savings of $393, $275, and $296 for adults, adolescents, and children, respectively). Drug acquisition costs accounted for the majority (83%) of total costs for ferric carboxymaltose ($630). The cost-savings estimated by the sponsor were entirely driven by reduced administration costs (nurse time, infusion chair time, and infusion devices) as ferric carboxymaltose had the same drug acquisition costs as ferric derisomaltose (incremental drug cost = $0) and higher drug acquisition costs (incremental drug cost = $245) compared with iron sucrose.
Sensitivity and Scenario Analysis Results
The sponsor conducted several scenario analyses (for adult patients only) to explore the impact of alternative approaches to calculating the total iron dose (i.e., a treatment course) based on different methods: Ganzoni formula, simplified tables from the ferric derisomaltose product monograph, a fixed-dose approach (1,000 mg), cumulative iron dose based on their respective product monographs, and alternative assumptions for the maximum dose per single infusion (1,000 mg for ferric carboxymaltose and ferric derisomaltose and 300 mg for iron sucrose as per Monoferric PE Review Report reanalysis10). Treatment with ferric carboxymaltose was still less costly than with either comparator across all scenarios. This was entirely driven by reduced administration costs. However, the magnitude of the cost-savings differed by more than 15% across most of the comparisons, highlighting the uncertainty in the cost-savings, which depends on how the total iron doses are calculated and the number of infusions required in clinical practice.
The sponsor also considered a scenario exploring the impact of adopting a societal perspective, which included additional costs associated with lost patient income, which was estimated based on time missed at work to travel and receive IV infusions, employment rate, and hourly wage. The cost-savings associated with ferric carboxymaltose treatment were reduced when it was compared with ferric derisomaltose but increased when compared with iron sucrose.
Table 3: Summary of the Sponsor’s Economic Evaluation Results
Drug | Total drug costs ($) | Incremental drug costs ($) | Total costs ($) | Incremental costs ($) |
|---|---|---|---|---|
All ages (aged 1 year and older), probabilistic | ||||
Ferric carboxymaltose | 630 | Reference | 755 | Reference |
Ferric derisomaltose | 630 | 0 | 771 | –16 |
Iron sucrose | 385 | 245 | 1,117 | –362 |
Note: The sponsor calculated the weighted all ages probabilistic results based on the following distribution of patients: 71% adults (aged 18 years and older), 13% adolescents (aged 14 to 17 years), and 15% pediatric (aged 1 to 13 years). Negative numbers indicate cost-savings.
Source: Sponsor’s economic submission.1
CDA-AMC Appraisal of the Sponsor’s Economic Information
CDA-AMC identified several key limitations to the sponsor’s analysis that have notable implications on the economic analysis:
The assumption of equivalent clinical efficacy and safety between ferric carboxymaltose and its comparators is uncertain: The CDA-AMC Clinical Review identified concerns with the generalizability of the sponsor-selected direct comparative evidence and several limitations of the sponsor-selected indirect clinical comparative evidence.2-6 The CDA-AMC Clinical Review of studies that included patients with IDA arising from various causes suggests that, for adults, ferric carboxymaltose is at least as effective as iron sucrose, and there is evidence that the efficacy of ferric carboxymaltose is comparable to ferric derisomaltose. However, comparative clinical evidence to other IV iron therapies in pediatric and adolescent patients is lacking. Therefore, there is insufficient evidence to support the assumption of equal efficacy and safety in these subgroups.
The sponsor also assumed equivalent overall safety between ferric carboxymaltose and other IV iron therapies based on the same published evidence.2-6 However, the CDA-AMC Clinical Review found an increased risk of hypophosphatemia with ferric carboxymaltose compared with ferric derisomaltose; it may occur more frequently with ferric carboxymaltose compared with iron sucrose. However, it is difficult to quantify the magnitude of these differences, and the clinical significance of this increased risk of hypophosphatemia is not known. Until the true prevalence and clinical impact of IV iron-induced hypophosphatemia is more fully characterized and quantified within the published literature, findings from a systematic review suggest that physicians and researchers should actively consider the possibility of hypophosphatemia in all patients receiving IV iron, particularly in those receiving ferric carboxymaltose. Therefore, the sponsor’s assumption of equivalent safety remains highly uncertain. CDA-AMC requested additional information from the sponsor;13 the sponsor replied that the clinical importance of the higher incidence of hypophosphatemia compared with ferric carboxymaltose has not been established. The sponsor highlighted that most hypophosphatemia occurrences are biochemically moderate, asymptomatic, and resolved without intervention. However, the ferric carboxymaltose product monograph notes the increased risk of hypophosphatemia, hypophosphatemic osteomalacia, and fractures (reported in the postmarketing period) that have required surgery.7 Monitoring for hypophosphatemic osteomalacia is recommended for patients who receive multiple doses of ferric carboxymaltose for long-term treatment and who have any underlying risk factors (vitamin D deficiency, calcium and phosphate malabsorption, secondary hyperparathyroidism, hereditary hemorrhagic telangiectasia, IBD, and osteoporosis).7 It is recommended that serum phosphate levels be checked in patients at risk of low serum phosphate who require a repeat course of treatment within 3 months. This is aligned with clinical expert input obtained by CDA-AMC. However, the cost-minimization analysis did not include costs related to monitoring or treating hypophosphatemia after treatment with ferric carboxymaltose. As a result, the validity of the sponsor’s cost-minimization analysis is uncertain, and it is also uncertain if the estimated cost-savings would be realized.
CDA-AMC was unable to address the uncertainty of comparative clinical efficacy and safety in reanalysis.
CDA-AMC included a scenario analysis with the addition of monitoring and treatment costs. This scenario assumed 10% of patients would require testing and treatment based on clinical expert input obtained by CDA-AMC. This would entail 2 laboratory tests, 1 general consultation visit and the cost of 1 bottle of K-phos (sodium phosphate) tablets, reflecting the management expected for nonsevere hypophosphatemia. No costs were applied to the routine management of hypophosphatemia for the comparators as the clinical expert input noted hypophosphatemia is more of a concern associated with treatment with ferric carboxymaltose.
Variability in clinical practice regarding the total iron dose per treatment course and other infusion planning aspects: The sponsor adopted a simplified table from the product monograph of ferric carboxymaltose to determine the cumulative iron dose per treatment course (i.e., total iron need) for any iron product for adult patients.7 For pediatric and adolescent patients, the sponsor used the Ganzoni formula targeting a hemoglobin level of 13 g/dL and 15 g/dL for those weighing less than 35 kg and those weighing more than 35 kg, respectively. Both ferric derisomaltose and iron sucrose are used off-label in pediatric, adolescent, and pregnant patient populations. Iron sucrose is also used off-label for the broader population of patients with IDA without CKD. According to the clinical input obtained by CDA-AMC for this review, clinical practices vary on how the cumulative iron dose to treat patients with IDA is determined. While some clinicians may adopt a 1,000 mg fixed dose per treatment course (for adults, adolescents, and pregnant patients) and reassess the iron needs of the patients, others will use the Ganzoni formula regardless of age. Furthermore, the clinical expert feedback noted that clinicians would adopt a lower target hemoglobin level for pediatric and adolescent populations (10 g/dL to 12.05 g/dL depending on age, based on CALIPER, a Canadian pediatric reference database).14 Additionally, according to the experts, the minimum infusion times adopted in Canadian clinical practice for the different iron products are age-dependent and longer than what was generally adopted by the sponsor.
The expected savings associated with ferric carboxymaltose would depend on the total iron dose per treatment course and the expected duration required to complete each infusion.
Finally, there is further variability in the maximum dose per single infusion for ferric derisomaltose. In clinical practice, a lower maximum dose (1,000 mg) may be provided than what is allowed in its product monograph, and this maximum dose per infusion would be identical to ferric carboxymaltose for adults. Altogether, these elements of dosing calculation and infusion planning will affect the costs related to nurse time, chair time, and infusion devices consumed. Therefore, the estimated cost-savings remain uncertain given the potential variabilities in clinical practice.
CDA-AMC could not address these limitations in reanalysis because any results could be misleading while there is remaining uncertainty concerning the comparative safety of ferric carboxymaltose, iron sucrose, and ferric carboxymaltose.
Confidential pricing agreements: The sponsor’s analyses uses pricing available for the comparator IV iron products from the Ontario Drug Benefit Formulary.15 However, confidential pricing agreements exist for ferric derisomaltose for this indication16 and iron sucrose has a generic price. Therefore, the submitted price of ferric carboxymaltose may require a price reduction to avoid incurring additional costs relative to ferric derisomaltose.
CDA-AMC was unable to address this limitation in reanalysis because the negotiated prices of other IV iron products are unknown.
CDA-AMC Reanalyses of the Economic Information
CDA-AMC did not undertake a reanalysis of the sponsor’s submission. The limitations identified by CDA-AMC could not be addressed and were related to the underlying assumption of equivalent clinical efficacy and safety between ferric carboxymaltose and comparators, and the lack of good supporting data to assert the costs of monitoring and treating hypophosphatemia.
A scenario analysis that explored the potential costs associated with monitoring and treating patients with nonsevere hypophosphatemia estimated lowered cost-savings associated with ferric carboxymaltose (Table 5). This scenario remains highly uncertain. If more than 10% of patients on ferric carboxymaltose require monitoring and treatment relative to the comparator therapies, or if patients develop severe hypophosphatemia, hypophosphatemic osteomalacia, and/or fractures, the cost-savings associated with ferric carboxymaltose may not be realized.
Issues for Consideration
Ferric carboxymaltose is also currently being reviewed by CDA-AMC for another distinct indication: the treatment of iron deficiency in adult patients with New York Heart Association class II and III heart failure to improve exercise capacity.17
Sodium ferric gluconate complex (Ferrliecit) is reimbursed in some jurisdictions; however, the clinical expert feedback obtained for this review was that it is predominantly used in a very specific population consisting of patients with IDA undergoing chronic hemodialysis who are receiving supplemental erythropoietin therapy. Given its selected use, the sodium ferric gluconate complex was not considered to be a relevant comparator.
Ferric carboxymaltose may be given as an IV bolus in which case the cost associated with infusion time would be reduced. The cost difference between ferric carboxymaltose IV bolus and other IV iron product infusions was not evaluated within this submission.
For IV iron products, treatment must be administered within a specific time frame after IV preparation (or it would otherwise be wasted) and patients need monitoring after treatment administration (accounted for in the cost analysis).
Although proper management of IDA may lead to reduced requirements for blood products and the use of ferric carboxymaltose may be associated with overall cost-savings compared to other IV iron products from a Canadian publicly funded health care payer perspective (due to decreased infusion time and thus reduced administration costs), from a public drug plan perspective, ferric carboxymaltose is associated with higher drug acquisition costs than iron sucrose and similar drug acquisition costs as ferric derisomaltose. Therefore, transitioning from these other IV iron products to ferric carboxymaltose may be complicated by increased costs for some budget holders (i.e., public drug plans), whereas the associated cost-savings are seen by others (e.g., hospital budget holders). Drug plans noted that there may be a potential for increased patient access within the existing system capacity because of the shorter infusion time associated with ferric carboxymaltose and that predicted savings from drug administration may not be realized. When considering drug costs alone, at the publicly available prices, the cost of ferric carboxymaltose is the same as for ferric derisomaltose and it would need to be reduced by 39% to equal that of iron sucrose when considering equivalent cumulative iron doses per treatment course and the age-dependent doses assumed by the sponsor.
Conclusions
The CDA-AMC Clinical Review including patients with IDA arising from various causes suggests that ferric carboxymaltose demonstrates equivalent efficacy when compared to ferric derisomaltose or iron sucrose. However, the assumption of equivalent safety remains highly uncertain and may be inappropriate because there may be an increased risk of hypophosphatemia with ferric carboxymaltose that may require additional monitoring and treatment. The clinical significance of this increased risk of hypophosphatemia is not known. If hypophosphatemia has a non-negligible quality-of-life impact, a cost-utility analysis would have been the preferred approach for the pharmacoeconomic analysis.
The sponsor submitted a cost-minimization analysis assuming equivalent clinical efficacy and safety among ferric carboxymaltose, iron sucrose, and ferric derisomaltose. In the submission, ferric carboxymaltose was associated with cost-savings of $16 and $362 per patient per treatment course when compared to ferric derisomaltose and iron sucrose, respectively, at public list prices. However, the costs of monitoring and treating hypophosphatemia were not included by the sponsor. CDA-AMC did not undertake a base-case reanalysis because the limitations identified by CDA-AMC could not be addressed given that they were related to the underlying assumption of equivalent clinical efficacy and safety between ferric carboxymaltose and its comparators and the variability in the administration of the total iron dose per treatment course. A scenario analysis was conducted that included monitoring and treatment costs based on assumptions and expert opinion for nonsevere hypophosphatemia. This analysis estimated that cost-savings would be reduced (incremental savings: $4 compared to ferric derisomaltose; $349 compared to iron sucrose); however, if rates of hypophosphatemia requiring treatment and monitoring are higher than assumed (10% based on expert opinion) or if patients develop more severe adverse events (e.g., severe hypophosphatemia, hypophosphatemic osteomalacia, and/or fractures), savings associated with the reimbursement of ferric carboxymaltose for the reimbursement population may be further reduced or eliminated. Due to the remaining uncertainty concerning the comparative safety across iron products and its associated costs, the magnitude of cost-savings associated with ferric carboxymaltose remains uncertain. All costs and incremental savings are also based on publicly available list prices and may not reflect actual prices paid by Canadian public drug plans.
Any potential cost-savings are entirely driven by reduced administration costs (nurse time, infusion chair time, and infusion devices); therefore, they would not be achieved from a public drug plan perspective because they may pertain to a different budget holder.
References
1.Pharmacoeconomic evaluation [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Ferinject® (ferric carboxymaltose injection), dispersion, 100 mg/2 mL, 500 mg/10 mL, and 1000 mg/20 mL (each corresponding to 50 mg elemental iron/mL [as ferric carboxymaltose]), intravenous. Glattbrugg (CH): CSL Vifor; 2024 May 10.
2.Evstatiev R, Marteau P, Iqbal T, et al. FERGIcor, a randomized controlled trial on ferric carboxymaltose for iron deficiency anemia in inflammatory bowel disease. Gastroenterology. 2011;141(3):846-853.e2. doi:10.1053/j.gastro.2011.06.005 PubMed
3.Onken JE, Bregman DB, Harrington RA, et al. Ferric carboxymaltose in patients with iron-deficiency anemia and impaired renal function: the REPAIR-IDA trial. Nephrol Dial Transplant. Apr 2014;29(4):833-42. doi:10.1093/ndt/gft251 PubMed
4.Aksan A, Işık H, Radeke HH, Dignass A, Stein J. Systematic review with network meta-analysis: comparative efficacy and tolerability of different intravenous iron formulations for the treatment of iron deficiency anaemia in patients with inflammatory bowel disease. Aliment Pharmacol Ther. 2017/05/01 2017;45(10):1303-1318. doi:https://doi.org/10.1111/apt.14043
5.Pollock RF, Muduma G. A systematic literature review and indirect comparison of iron isomaltoside and ferric carboxymaltose in iron deficiency anemia after failure or intolerance of oral iron treatment. Expert Rev Hematol. Feb 2019;12(2):129-136. doi:10.1080/17474086.2019.1575202 PubMed
6.Han B, Sun W, Zhuang L. Comparison of the efficacy and safety of ferric carboxymaltose versus ferric derisomaltose in correcting iron deficiency anemia. Clinical Medication Journal. 2023;21(12):31-36.
7.FERINJECT - Ferric Carboxymaltose Injection [product monograph; sponsor-submitted reference]. C. S. L. Vifor; 2024.
8.Dieticians of Canada. WHO growth charts for Canada [sponsor-submitted reference]. Accessed 2024 Jan 18, https://www.dietitians.ca/Secondary-Pages/Public/Who-Growth-Charts.aspx
9.Monoferric - Ferric Derisomaltose for Injection [product monograph; sponsor-submitted reference]. Pharmacosmos, A. S.; 2022.
10.Drug Reimbursement Review Pharmacoeconomic Report: Iron Isomaltoside 1000 (Monoferric) [sponsor-submitted reference]. CADTH; 2020.
11.Venofer - Iron Sucrose Injection [product monograph; sponsor-supplied reference]. American Regent, Inc.; 2023.
12.Bhandari S. Update of a comparative analysis of cost minimization following the introduction of newly available intravenous iron therapies in hospital practice. Ther Clin Risk Manag. 2011;7:501-9. doi:10.2147/TCRM.S25882 PubMed
13.CSL Behring response to July 30, 2024 CADTH Review Update Letter regarding Ferinject (ferric carboxymaltose) for iron deficiency anemia CADTH review: Studies Comparing Ferric Carboxymaltose Versus Ferric Derisomaltose, and Potential Implications for Pharmacoeconomic Submission [internal sponsor's report]. Ottawa (ON): CSL Behring; 2024 Aug 9.
14.SickKids. CALIPER Pediatric Reference Interval Database. Accessed 2024 Aug 19, https://caliperproject.ca/caliper/database/
15.Ontario Ministry of Health, Ontario Ministry of Long-Term Care. Ontario drug benefit formulary/comparative drug index. Accessed 2024 Jun 21, https://www.formulary.health.gov.on.ca/formulary/
16.pan-Canadian Pharmaceutical Alliance. Monoferric (iron (iii) isomaltoside 1000). Accessed 2024 Aug 26, https://www.pcpacanada.ca/negotiation/21194
17.CADTH. Drug Reimbursement Review: ferric carboxymaltose (Ferinject) for iron deficiency in adult patients with heart failure. Accessed 2024 Aug 22, https://www.cda-amc.ca/ferric-carboxymaltose-0
18.FERRLECIT - sodium ferric gluconate complex in sucrose injection [product monograph; sponsor-submitted reference]. sanofi-aventis Canada Inc; 2019.
19.Saskatchewan Drug Plan: search formulary. Accessed 2024 Aug 8, https://formulary.drugplan.ehealthsask.ca/SearchFormulary
20.Schedule of benefits for laboratory services. Ontario Ministry of Health; 2023. Accessed 2024 Aug 19. https://www.health.gov.on.ca/en/pro/programs/ohip/sob/lab/sob_lab_2023.pdf
21.Schedule of benefits for physician services under the Health Insurance Act: (June 29, 2023 (effective July 24, 2023)). Ontario Ministry of Health; 2023. Accessed 2024 Aug 19. https://www.health.gov.on.ca/en/pro/programs/ohip/sob/physserv/sob_master.pdf
22.Government of Alberta. Interactive drug benefit list. Accessed 2024 Aug 19, https://idbl.ab.bluecross.ca/idbl/load.do
23.Bourguignon S, Faller M, Champs FO, et al. Budget impact of intravenous ferric carboxymaltose in patients with chronic heart failure and iron deficiency in France. Research Support, Non-U.S. Gov't. ESC Heart Fail. Jun 2019;6(3):559-569. doi:10.1002/ehf2.12432 PubMed
24.PharmaStat [sponsor-supplied reference]. IQVIA; 2023. Accessed 2023 Dec. https://www.iqvia.com/
25.Iqvia. Canadian Drugstore and Hospital Purchases Audit (CDH) [sponsor-submitted reference]. Accessed 2023 Dec, https://www.iqvia.com/
26.C. S. L. Vifor. Data on file [sponsor-submitted reference].
Appendix 1: Additional Economic Information
Table 4: CDA-AMC Cost Comparison Table for Parenteral Iron Products for IDA
Treatment | Strength | Form | Price | Recommended dosagea | Average drug cost per treatment course |
|---|---|---|---|---|---|
Recommended use | |||||
Ferric Carboxymaltose (Ferinject) | 50 mg/mL | 100 mg / 2 mL 500 mg / 10 mL 1,000 mg / 20 mL Single-use vial for IV infusion | $45.0000b $225.0000b $450.0000b | Simplified cumulative iron dose:
| For patients (1 to 17 years): $90 to $675 |
| For adults (> = 18 years): $225 to $900 | ||||
| For pregnant patients: $450 to $675 | ||||
Actual practice (off-label use) – Except for ferric derisomaltose in adults which has recommended dosage for adults only | |||||
Ferric derisomaltose (Monoferric) | 100 mg/mL | 100 mg / 1 mL 500 mg / 5 mL 1,000 mg / 10 mL Single-use vial for IV infusion | 45.0000 225.0000e 450.0000e | For patients aged 1 to 17:
| For patients (1 to 17 years):
|
Simplified cumulative iron dose:
Pregnant: Off-label use; pregnant patients are likely to be treated same as other adults | For adults (> = 18 years): $225 to $900 | ||||
Iron sucrose (generic) | 20 mg/mL | 100 mg / 5 mL Single-use vial for IV infusion | 27.5000 | For patients aged 1 to 17:
| For patients (1 to 17 years):
|
For adults (≥ 18 years):
| For adults without CKD:
| ||||
| For adults with CKD (NDD, HDD, PDD): $275 | ||||
Pregnant: Off-label use. Pregnant patients are likely to be treated same as other adults | — | ||||
Sodium Ferric Gluconate Complex (Ferrlecit)l | 12.5 mg/mL | 62.5 mg / 5 mL vial for IV infusion | 28.3000m | For patients aged 1 to 17: Health Canada has not authorized use for pediatric | — |
Adults with HDD-CKD: Most patients will require a minimum total iron need of 1,000 mg, 125 mg at a time per consecutive hemodialysis session administered over 1 hour. A single infusion should not exceed 12.5 mg/minute. | $453 or more | ||||
CKD = chronic kidney disease; HDD = hemodialysis dependent; NDD = non-dialysis-dependent; PDD = peritoneal dialysis dependent; FCM = ferric carboxymaltose
Notes: The comparators presented in the above table have been deemed to be appropriate based on feedback from clinical expert(s) and drug plans. Comparators may be recommended (appropriate) practice or actual practice. Existing Product Listing Agreements are not reflected in the table and as such, the table may not represent the actual costs to public drug plans.
All prices are from the Ontario Drug Benefit Formulary (accessed June 21, 2024),15 unless otherwise indicated, and do not include dispensing fees. Drug wastage is included.
This table has not been copy-edited.
aRecommended doses vary depending on the combination of weight and hemoglobin levels, supporting tables available in the respective product monographs (when available).7,9,11,18
bSponsor’s submitted price.1
cThe lower range is the maximum recommended cumulative dose for a patient with 10 kg and hemoglobin ≥ 15 g/dL and the upper range is the maximum recommended cumulative dose for a patient with 60 kg and hemoglobin = 7 g/dL.
dThe lower range is the recommended dose for a patient with 35 kg and hemoglobin ≥ 14 /dL and the upper range is the recommended dose for a patient with 70 kg and above and hemoglobin < 10 g/dL.
eCDA-AMC review of Monoferric (Iron (III) Isomaltoside 1,000).10
fGanzoni formula for pediatric patients assuming a target hemoglobin of 10.6 g/dL for pediatric patients and 12.05 g/dL for adolescents; and storage iron of 15 mg/kg for pediatric patients and 500 mg for adolescents, based on clinical input obtained by CDA-AMC for this review.
gBased on clinical input obtained by CDA-AMC for this review.
hThe lower range is the recommended dose for a patient with < 50 kg regardless of hemoglobin levels and the upper range is the recommended dose for a patient with 70 kg and above and hemoglobin < 10 g/dL.
iIf used off-label at the same range of cumulative iron dose as per ferric carboxymaltose simplified tables for pediatrics (150 mg to 1,500 mg).
jThe recommended cumulative dose for adult patients is calculated assuming a target hemoglobin of 15 g/dL and storage iron of 500 mg based on clinical input obtained by CDA-AMC for this review.
kIf used off-label at the same range of cumulative iron dose as per ferric carboxymaltose simplified tables for adults (500 mg to 2,000 mg).
lIndicated for the treatment of iron deficiency anemia in patients undergoing chronic hemodialysis who are receiving supplemental erythropoietin therapy. The expert consulted by CDA-AMC did not consider Ferrlecit a key comparator.
mSaskatchewan formulary, accessed August 8, 2024.19
Additional Details on the CDA-AMC Reanalyses and Additional Analyses
Table 5: Scenario Analysis Conducted on the Sponsor’s Base Case, Probabilistic
Drug | Total drug costs ($) | Incremental drug costs ($) | Total costs ($) | Incremental costs ($) |
|---|---|---|---|---|
Sponsor’s base case | ||||
All ages (aged 1 year and older) | ||||
Ferric carboxymaltose | 630 | Reference | 755 | Reference |
Ferric derisomaltose | 630 | 0 | 771 | −16 |
Iron sucrose | 385 | 245 | 1,117 | −362 |
CDA-AMC exploratory analysis – including monitoring and treatment costs of hypophosphatemia | ||||
All ages (aged 1 year and older) | ||||
Ferric carboxymaltosea | 640 | Reference | 764 | Reference |
Ferric derisomaltose | 628 | 12 | 768 | −4 |
Iron sucrose | 384 | 256 | 1,113 | −349 |
aAssuming 10% of patients were tested and treated (symptomatic). Components added to the total drug cost: 2 laboratory tests (L194) from the Ontario Schedule of Benefits for Laboratory Services,20 1 general consultation visit (A005) from the Ontario Schedule of Benefits: Physician Services,21 price of sodium phosphate from the Alberta formulary assuming a full bottle (20 tablets) is dispensed per patient.22
Appendix 2: Submitted BIA and CDA-AMC Appraisal
Please note that this appendix has not been copy-edited.
Table 6: Summary of Key Take-Aways
Key take-aways of the BIA |
|---|
|
Summary of Sponsor’s BIA
The sponsor submitted a budget impact analysis (BIA)23 assessing the expected budgetary impact of the introduction of ferric carboxymaltose for the treatment of adult and pediatric patients 1 year of age and older IDA when oral iron preparations are not tolerated or are ineffective. The BIA was undertaken from the perspective of the pan-Canadian public drug plans over a 3-year time horizon (2025/26 to 2027/28).
A claims-based approach was used to estimate the eligible market size. Number of claims in the outpatient setting and sales volume (in mg) in inpatient setting were obtained from IQVIA PHARMASTAT (Q1 2019 to Q3 2023) and IQVIA Canadian Drugstore and Hospital Purchases Audit databases (January 2019 to September 2023), respectively, for ferric derisomaltose and iron sucrose.24,25 Both inpatient and outpatient future market size were based on the extrapolation of the historical data using a linear regression. The sponsor estimated that the proportion of claims and sales volume specific to IDA was 98.3% based on internal market calculations.26 In the outpatient setting, the sponsor assumed that each claim was representative of 1 cumulative iron dose (i.e., 1 treatment course). In the inpatient setting, the total number of mg of iron dispensed was converted to the number of 100 mg vials. Market shares in the reference scenario across the comparators, ferric derisomaltose and iron sucrose, were calculate based on their proportional number of claims in outpatient settings and on their proportional number of 100 mg vials in the inpatient setting. In the new drug scenario, the sponsor assumed claim-to-claim and vial-to-vial displacement between ferric carboxymaltose and comparators, and capture rates proportional to the shares of the 2 comparators in the reference scenario. The uptake of ferric carboxymaltose was based on internal market calculations for the ferric carboxymaltose sales in the European Union (1%, 3% and 9% in years 1 to 3), and assumed to be the same in inpatient and outpatient settings, across all jurisdictions. The BIA included costs related to drug acquisition, dispensing fees, and pharmacy mark-ups. The sponsor estimated the cost per claim by multiplying the total number of 100 mg vials needed to administer a cumulative iron dose of 1,383 mg (14 vials), based on the average body weight and hemoglobin level of the patient population considered in the cost-minimization analysis. In the inpatient setting, the number of 100 mg vials were multiplied by their respective costs for each iron product. Key inputs are presented in Table 7.
Table 7: Summary of Key Model Parameters
Parameter | Sponsor’s estimate (reported as year 1 / year 2 / year 3 if appropriate) |
|---|---|
Target population | |
Total number of claims, outpatient | 37,486 / 44,092 / 50,699 |
Total sale volume (in 100 mg vials), inpatient | 2,952,739 / 2,995,000 / 3,037,261 |
Market Shares and Uptake (3 years) | |
Reference scenario, outpatient Ferric derisomaltose Iron sucrose | 100.00% / 100.00% / 100.00% 0.00% / 0.00% / 0.00% |
Reference scenario, inpatient Ferric derisomaltose Iron sucrose | 2.96% / 3.39% / 3.81% 97.04% / 96.61% / 96.19% |
New drug scenario, outpatient Ferric carboxymaltose Ferric derisomaltose Iron sucrose | 1.00% / 3.00% / 9.00% 99.00% / 97.00% / 91.00% 0.00% / 0.00% / 0.00% |
New drug scenario, inpatient Ferric carboxymaltose Ferric derisomaltose Iron sucrose | 1.00% / 3.00% / 9.00% 2.93% / 3.29% / 3.47% 96.07% / 93.71% / 87.53% |
Cost of treatment | |
Cost per 100 mg vial, inpatient Ferric carboxymaltose Ferric derisomaltose Iron sucrose | $45.00 $45.00 $27.50 |
Cost per claim, outpatient Ferric carboxymaltose Ferric derisomaltose Iron sucrose | $630.00 $630.00 $385.00 |
Note: These are the parameters for Ontario. The sponsor estimated different market share for other jurisdictions, based on their number of claims and 100mg vials sales volume.
Summary of the Sponsor’s BIA Results
The sponsor estimated that the budget impact of reimbursing ferric carboxymaltose for the treatment of IDA to be $1,698,711 in year 1, $5,301,695 in year 2, $16,521,765 in year 3, for a 3-year total incremental budget impact of $23,522,171 ($364,157 in the outpatient settings and $23,158,014 in inpatient settings).
CDA-AMC Appraisal of the Sponsor’s BIA
CDA-AMC identified several key limitations to the sponsor’s analysis that have notable implications on the results of the BIA:
Use of a claims-based approach to estimate market size and market shares introduces uncertainty with the anticipated budget impact of ferric carboxymaltose: The sponsor estimated the current market size based on the number of outpatient claims and inpatient sales volume (of 100 mg vials) using historic public claims data for ferric derisomaltose and iron sucrose.24,25 The sponsor extrapolated the historical claims data from both settings using linear regression to project the future market size in both settings without any consideration of system capacity (i.e., resource constraints to the volume of patients the outpatient clinics and inpatient settings may have), changes in the number of patients with coverage by the drug plans, whether the inpatient use of IV iron is reimbursed by the drug plans or potential shifts from inpatient utilization to outpatient utilization. As the claims data reflects only patients who had access to coverage for the comparators, a significant portion of the population who may have IDA but did not have access to IV iron products in the past may have been excluded from future projections.
Additionally, the sponsor assumed that all claims for inpatient administration of iron therapy are covered by all jurisdictions under the drug plan perspective. However, differences between jurisdictional funding approaches exist, where some jurisdictions would cover inpatient administration of iron therapy and others may only fund outpatient patients with specific indications (e.g., CKD) and some others do not reimburse inpatient iron therapy. As we are unable to isolate these subpopulations from the claims, the inclusion of inpatient claims in the pan-Canadian estimates from a drug plan perspective is uncertain, as they may pertain to a different budget holder.
Finally, the sponsor assumed claim-to-claim and vial-to-vial displacement between ferric carboxymaltose and comparators in outpatient and inpatient settings, respectively. The sponsor also assumed that a single claim represented 1 full treatment course. However, there are differences in the maximum dose per single infusion between ferric carboxymaltose and comparators and it is uncertain whether a single claim truly includes all the infusions required to deliver a treatment course (i.e., 1 cumulative iron dose) or if it represents a maximum iron dose allowed per single infusion of each drug. The sponsor did not obtain the number of patients from claims and sales data which would have been more appropriate to estimate the number of patients eligible for the drug under review, given the differences in dosing, frequencies, and durations between treatments. For example, assuming an adult patient with IDA requires chronic treatment with iron sucrose, this patient can only access a maximum of approximately 4.7 treatment courses throughout the year (5 weekly infusions and reassessment after 4 weeks post final administration to allow adequate time for erythropoiesis and iron utilization); while if being treated with ferric derisomaltose, the same patient can access a maximum of approximately 8.6 treatment courses throughout the year (1 single infusion and reassessment after 4 weeks). Therefore, it is uncertain if treatment with ferric carboxymaltose will displace the same number of claims or treatment courses for the same average patient over time.
CDA-AMC was unable to address the limitations associated with the claims-based approach.
Additional minor limitations were identified but were not addressed by CDA-AMC as the limitation pertaining to the claims-based approach (as previously described) cannot be addressed:
Market uptake is uncertain: The sponsor’s assumption that ferric carboxymaltose has a market share of 9% in both inpatient and outpatient settings is likely underestimated. Clinical expert feedback obtained for this review noted the potential for higher uptake of ferric carboxymaltose in pediatric and pregnant populations as all other iron products are off-label for use in these subpopulations. However, as the sponsor’s budget impact (and claims data) were not estimated separately for these subgroups, the sponsor’s estimates of market displacement are uncertain. Additionally, the sponsor’s estimates that the future market shares of iron sucrose are going to be 0% in some jurisdictions (based on linear regression of the claims data discussed previously) lacks face validity. According to clinical input obtained by CDA-AMC for this review, clinicians will continue to prescribe iron sucrose for the proportion of patients who are hypersensitive to the other iron products.
Dispensing and mark-up fees are included for individuals receiving outpatient treatment: The sponsor included a dispensing fee and pharmacy mark-up in the estimation of cost per claim for all treatments. Dispensing fees and mark-ups vary across jurisdictions; however, the sponsor did not incorporate jurisdiction-specific fees for Ontario, New Brunswick, Nova Scotia, and Prince Edward Island. Consequently, the estimated budget impact may not accurately reflect the actual costs incurred by public health care payers.
The sponsor’s submitted model for the budget impact model is not user-friendly and unnecessarily complicated: Several of the model inputs and assumptions in the sponsor’s submitted budget impact model were difficult to test or modify with alternate inputs or assumptions due to unnecessary complexity (i.e., inconsistency between sheets; recalculation of jurisdictional budget impact does not feed into the pan-Canadian results sheet).
CDA-AMC Reanalyses of the BIA
In the absence of more reliable estimates to inform the key parameters of the BIA, the sponsor’s submitted base case was maintained (Table 8). CDA-AMC expects that the budget impact of reimbursing ferric carboxymaltose for the treatment of IDA will be sensitive to more reliable inputs which may affect the market size calculation, uptake and displacement of comparators by ferric carboxymaltose, and the prices of treatments for IDA paid for by the public drug plans.
Table 8: Detailed Breakdown of the CDA-AMC Reanalyses of the BIA
Stepped analysis | Scenario | Year 0 (current situation) | Year 1 | Year 2 | Year 3 | Three-year total |
|---|---|---|---|---|---|---|
Submitted base case | Reference | NA | 333,047,365 | 355,143,247 | 377,239,316 | 1,065,429,928 |
New drug | NA | 334,746,076 | 360,444,942 | 393,761,081 | 1,088,952,100 | |
Budget impact | NA | 1,698,711 | 5,301,695 | 16,521,765 | 23,522,171 | |
Submitted base case, outpatient | Reference | NA | 32,344,247 | 37,813,769 | 43,283,290 | 113,441,306 |
New drug | NA | 32,369,087 | 37,894,180 | 43,542,196 | 113,805,463 | |
Budget impact | NA | 24,840 | 80,412 | 258,906 | 364,157 | |
Submitted base case, inpatient | Reference | NA | 295,271,375 | 310,940,485 | 326,609,783 | 932,821,643 |
New drug | NA | 296,945,246 | 316,161,769 | 342,872,642 | 955,979,656 | |
Budget impact | NA | 1,673,871 | 5,221,284 | 16,262,859 | 23,158,014 |
BIA = budget impact analysis; NA = estimates for base year were not available within the sponsors submitted model
Note: Reference year costs are not available in the submitted model. The sponsor included reference year number of claims and vials, but did not calculate the budget for reference year.
ISSN: 2563-6596
Canada’s Drug Agency (CDA-AMC) is a pan-Canadian health organization. Created and funded by Canada’s federal, provincial, and territorial governments, we’re responsible for driving better coordination, alignment, and public value within Canada’s drug and health technology landscape. We provide Canada’s health system leaders with independent evidence and advice so they can make informed drug, health technology, and health system decisions, and we collaborate with national and international partners to enhance our collective impact.
Disclaimer: CDA-AMC has taken care to ensure that the information in this document was accurate, complete, and up to date when it was published, but does not make any guarantee to that effect. Your use of this information is subject to this disclaimer and the Terms of Use at cda-amc.ca.
The information in this document is made available for informational and educational purposes only and should not be used as a substitute for professional medical advice, the application of clinical judgment in respect of the care of a particular patient, or other professional judgments in any decision-making process. You assume full responsibility for the use of the information and rely on it at your own risk.
CDA-AMC does not endorse any information, drugs, therapies, treatments, products, processes, or services. The views and opinions of third parties published in this document do not necessarily reflect those of CDA-AMC. The copyright and other intellectual property rights in this document are owned by the Canadian Agency for Drugs and Technologies in Health (operating as CDA-AMC) and its licensors.
Questions or requests for information about this report can be directed to Requests@CDA-AMC.ca.