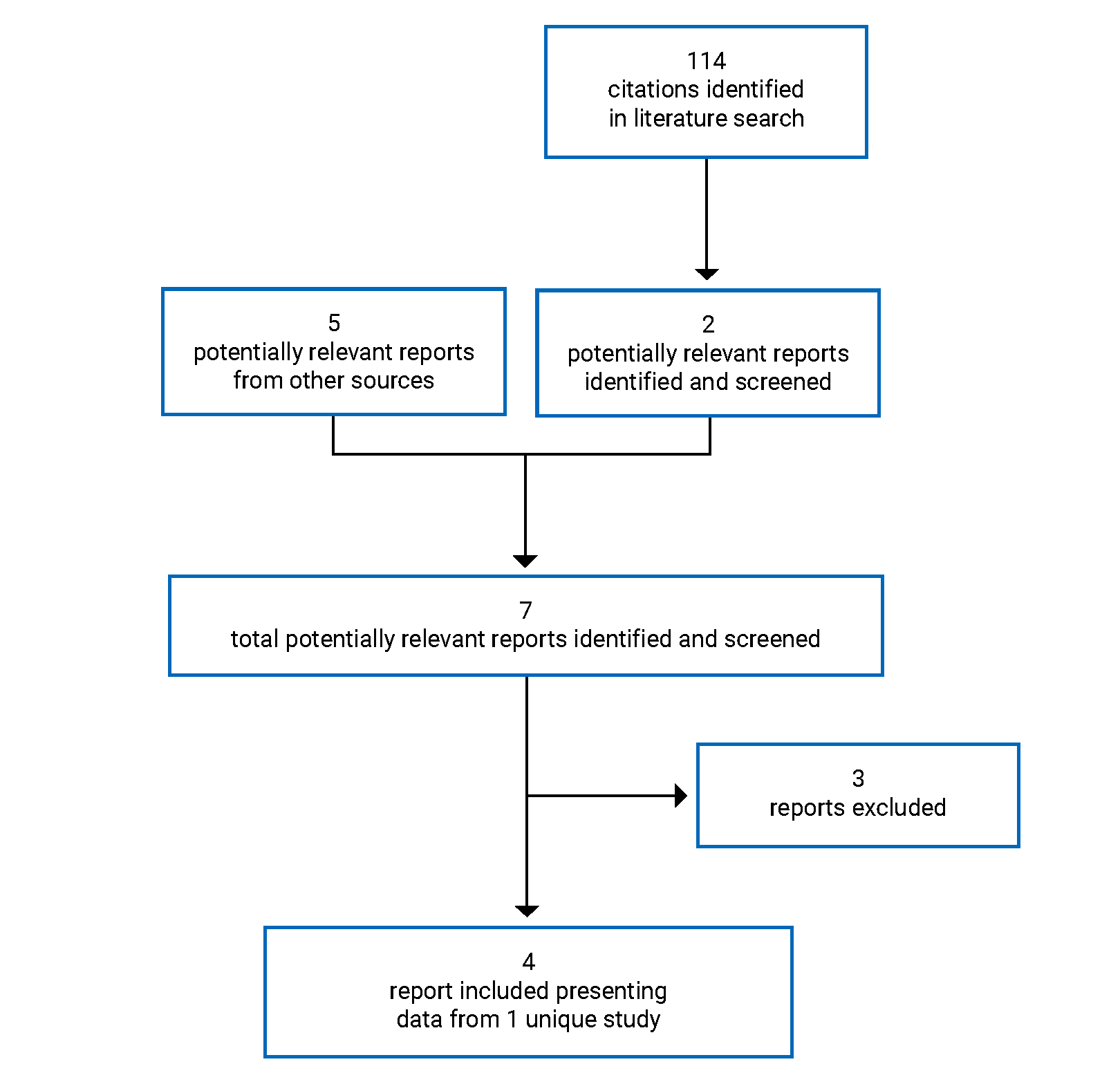
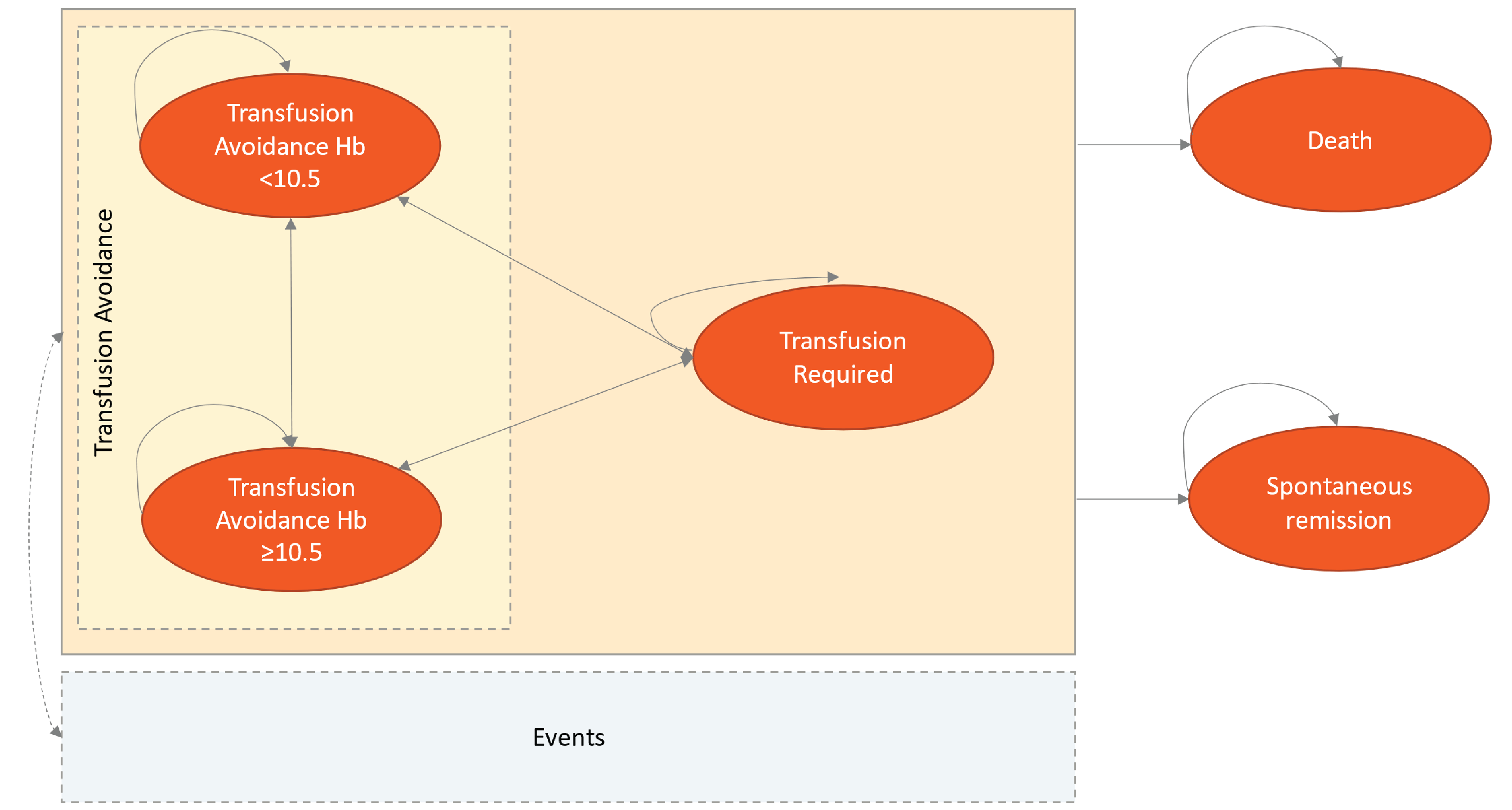
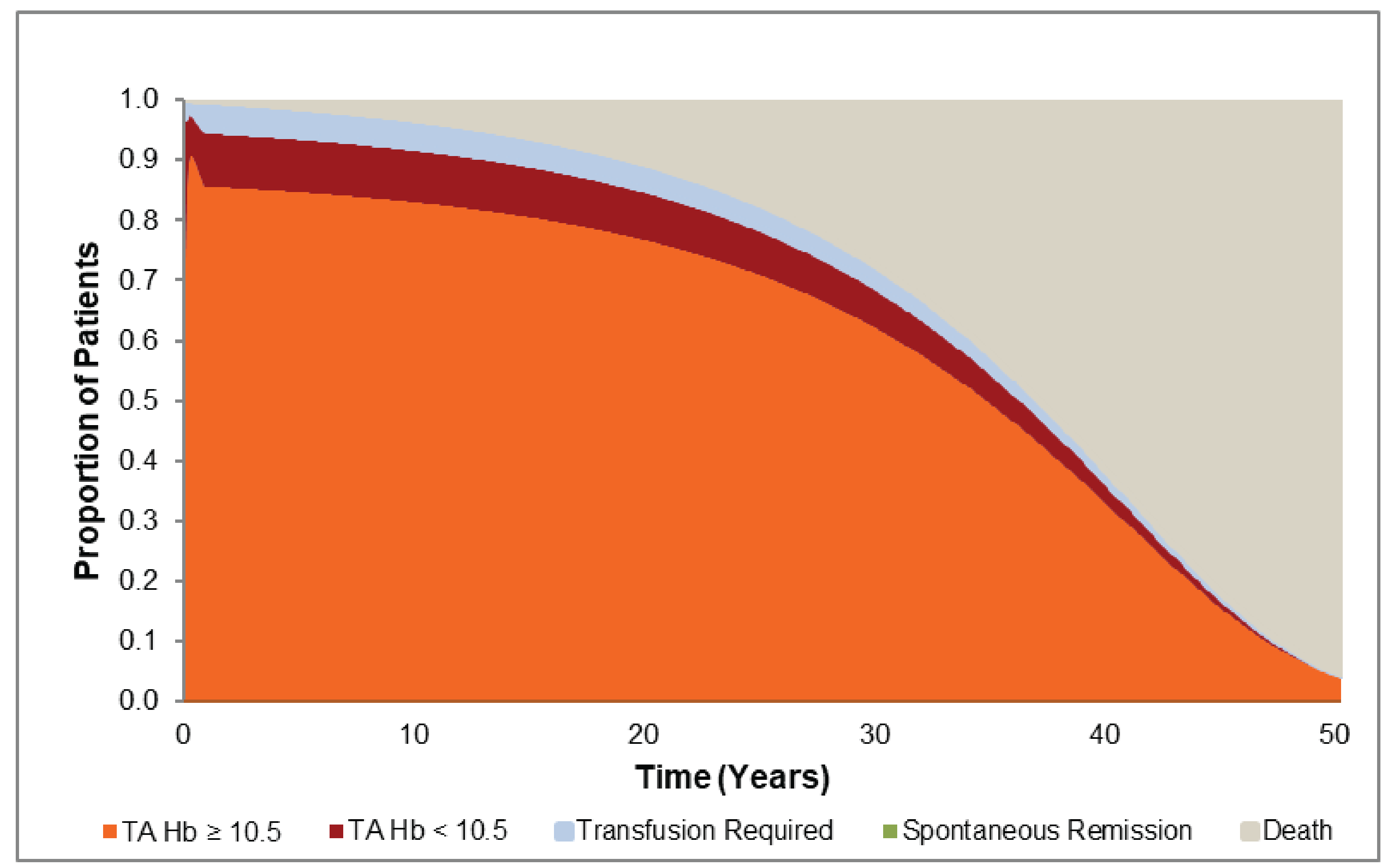
CADTH Reimbursement Review
Pegcetacoplan (Empaveli)
Sponsor: Sobi Canada Inc.
Therapeutic area: Paroxysmal nocturnal hemoglobinuria
This multi-part report includes:
Clinical Review
Pharmacoeconomic Review
Stakeholder Input
Clinical Review
Abbreviations
AE
adverse event
ARC
absolute reticulocyte count
C5
complement component 5
CI
confidence interval
EORTC QLQ-C30
European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30
ESS
effective sample size
EVH
extravascular hemolysis
FACIT
Functional Assessment of Chronic Illness Therapy
Hb
hemoglobin
HRQoL
health-related quality of life
ITC
indirect treatment comparison
ITT
intention to treat
IVH
intravascular hemolysis
LASA
Linear Analogue Scale Assessment
LDH
lactate dehydrogenase
LS
least square
MAIC
match-adjusted indirect comparison
MID
minimally important difference
mITT
modified intention to treat
MMRM
mixed model for repeated measures
NIM
noninferiority margin
PNH
paroxysmal nocturnal hemoglobinuria
PP
per protocol
QoL
quality of life
RBC
red blood cell
RCT
randomized controlled trial
SD
standard deviation
SE
standard error
TEAE
treatment-emergent adverse event
ULN
upper limit of normal
Executive Summary
An overview of the submission details for the drug under review is provided in Table 1.
Item | Description |
|---|---|
Drug product | Pegcetacoplan (Empaveli), 54 mg/mL, solution, subcutaneous infusion |
Indication | For the treatment of adult patients with paroxysmal nocturnal hemoglobinuria who have an inadequate response to, or are intolerant of, a C5 inhibitor |
Reimbursement request | Per indication |
Health Canada approval status | Notice of Compliance |
Health Canada review pathway | Priority review |
Notice of Compliance date | December 8, 2022 |
Sponsor | Sobi Canada Inc. |
Introduction
Paroxysmal nocturnal hemoglobinuria (PNH) is an extremely rare chronic disease characterized by intravascular hemolysis (IVH) and heterogenous signs and symptoms that include hemoglobinuria, anemia, abdominal pain, dyspnea, fatigue, dysphagia, and erectile dysfunction.1 Complications of PNH include thrombosis, chronic kidney disease, and pulmonary hypertension.2 Although the incidence of PNH has not been extensively characterized, 1 study in the UK estimated an annual incidence of clinical PNH of approximately 0.13 per 100,000 persons.3 PNH is a consequence of an acquired genetic mutation, leading to clonal expansion of hematopoietic stem cells that produce abnormal blood cells that are susceptible to complement-mediated IVH.2
Terminal complement component 5 (C5) inhibitors (e.g., eculizumab, ravulizumab) are the first-line standard of care for patients with hemolytic PNH that has high disease activity (i.e., symptomatic and lactate dehydrogenase [LDH] 1.5 times above the upper limit of normal [ULN], indicative of clinically significant IVH). Approximately 20% to 30% of patients have ongoing and clinically significant anemia, despite C5 inhibitor treatments, due to reasons such as breakthrough IVH, complement component 3 (C3)-mediated extravascular hemolysis (EVH), development of human-antihuman or eculizumab-neutralizing antibodies, and C5 genetic polymorphism.1 Prior to the approval of pegcetacoplan, there was no approved subsequent therapy for patients with an inadequate response to a C5 inhibitor due to C3-mediated EVH. The clinical experts consulted by CADTH noted that the current treatment approach for these patients is to provide best supportive care (e.g., red blood cell [RBC] transfusion, steroids, splenectomy, danazol, epoetin alfa) while continuing C5 inhibitor treatment, although many best supportive care therapies are associated with toxicities. Referral to clinical trials is an alternative option. Patients with concurrent bone marrow insufficiency may also receive immunosuppressive therapy.
Pegcetacoplan is a proximal C3 inhibitor that has an indication for the treatment of adult patients with PNH who have an inadequate response to, or are intolerant of, a C5 inhibitor. The product monograph recommends pegcetacoplan 1,080 mg subcutaneous infusion be given twice weekly with a syringe system infusion pump by a health care professional, the patient, or a caregiver.4 A dosage increase to 1,080 mg every third day may be considered if the LDH level is at least 2 times above the ULN on twice-weekly dosing.4 This is the first CADTH reimbursement review for pegcetacoplan.
The objective of this report is to perform a systematic review of the beneficial and harmful effects of pegcetacoplan (54 mg/mL) solution for subcutaneous infusion for the treatment of adult patients with PNH who have an inadequate response to, or are intolerant of, a C5 inhibitor.
Stakeholder Perspectives
The information in this section is a summary of input provided by the patient groups that responded to CADTH’s call for patient input and from clinical experts consulted by CADTH for the purpose of this review.
Patient Input
The Canadian Association of PNH Patients and the Aplastic Anemia & Myelodysplasia Association of Canada submitted a joint input for this review. Information was gathered through 1-on-1 interviews with 6 individuals diagnosed with PNH living in Canada and from the scientific literature. The patient group expressed the following negative impacts of PNH: persistent anemia (manifested as fatigue or extreme fatigue), hemolysis leading to thrombosis, employment absenteeism for patients and caregivers, dependence on frequent blood transfusions, and reduced quality of life (QoL). According to the input, patients with PNH, particularly those experiencing EVH, need alternative treatment options because of the inability of available treatments with eculizumab or ravulizumab to thoroughly control IVH and prevent EVH. The patient group also expressed the need for therapies to improve anemia, reduce or eliminate transfusion requirements, and improve fatigue and QoL. Among the 3 patients who had used pegcetacoplan, all noted an immediate normalization of hematological parameters, easier administration (self-administered subcutaneous infusion twice weekly at home) than eculizumab or ravulizumab (requiring visits for IV transfusions), reduced blood transfusions, and improved physical functioning and QoL. Some patients stated the importance for proper injection training.
Clinician Input
Input From Clinical Experts Consulted by CADTH
The clinical experts noted there is a need for treatments that are effective in patients who have insufficient control of IVH despite treatment with eculizumab, address the issue of C3-mediated EVH, and can be used in patients with intolerance to C5 inhibitors or poor venous access. The clinical experts noted that pegcetacoplan is a C3 inhibitor with a mechanism of action in addition to C5 inhibitors because it inhibits a more upstream effector than C5 in the complement activation pathway. They anticipated that the drug would serve as a second-line treatment for PNH.
The clinical experts noted that suitable candidates for pegcetacoplan treatment include patients with PNH who have persistent anemia (with or without a history of ongoing blood transfusion needs) and evidence of EVH despite an adequate trial of C5 inhibitor treatment, patients with intolerance to a C5 inhibitor, and patients with a rare C5 genetic polymorphism (which prevents eculizumab from binding to its target molecule and is mainly present in patients of Japanese descent). The clinical experts noted that pegcetacoplan could be considered in patients who are geographically isolated or who have poor venous access. Patients with a PNH clone size (i.e., the proportion of blood cells deficient in complement system regulatory protein) of less than 10% should not receive pegcetacoplan.
The clinical experts noted that a clinically meaningful treatment response would include improvements in hemolytic parameters (LDH, bilirubin), hemoglobin (Hb) level, and QoL, and a reduction in transfusion needs. In general, follow-up assessments are conducted every 3 months, and treatment response is determined, per clinical judgment, by the treating physician based on a global assessment of all patient parameters, chronology of symptoms, and laboratory results. The clinical experts noted that treatment discontinuation is not considered unless 1 of the following occurs: treatment failure (persistent anemia and ongoing transfusion needs) necessitating a switch to a more effective treatment, intolerance to pegcetacoplan, or resolution of disease after bone marrow transplant. The clinical experts noted that patients with PNH should be managed by hematologists in consultation with PNH specialists.
Clinician Group Input
One clinician group, the Canadian PNH Network, submitted input for this review based on contributions from 11 clinicians. The group noted that the current standard of care is C5 inhibitors (i.e., eculizumab and ravulizumab), which act via terminal complement blockade. They noted that although C5 inhibitors are not curative treatment, these treatments have been shown to be effective in controlling IVH, leading to significant improvements in fatigue, QoL, transfusion dependence, thrombosis, and overall survival. The only curative treatment for PNH is allogeneic hematopoietic stem cell transplant, which is available to patients with predominant or progressive bone marrow failure or eligible patients with evidence of clonal evolution. However, the group highlighted 3 unmet needs: some patients do not have access to standard care due to highly restrictive reimbursement criteria; eculizumab has a high treatment burden because it requires venous access for administration and nurse visits every 2 weeks; and, despite treatment, approximately one-third of patients remain anemic due to EVH and some are transfusion-dependent. The group expressed that drugs that exploit proximal complement blockade, such as C3 inhibitors, address the EVH risk, significantly increase Hb, and improve QoL. Regarding place in therapy, the group stated pegcetacoplan is the first C3 inhibitor that protects against EVH and noted that it would fit into the current treatment landscape as an alternative (i.e., switch) option for patients with no or inadequate response or intolerance to eculizumab or ravulizumab. These would include patients with persistent anemia (Hb less than 10.5 g/dL or perhaps higher, if symptomatic) despite stable doses of eculizumab or ravulizumab and those who have had other causes of ongoing anemia (e.g., breakthrough hemolysis or bone marrow failure) ruled out. The group indicated that a clinically meaningful response to treatment would be sustained control of LDH level (i.e., below 1.5 times the ULN), an increase in Hb (or possibly Hb stabilization without further need for transfusion), and improvement in anemia-related symptoms. Treatment discontinuation should be considered in patients who have adverse events (AEs) that preclude ongoing therapy (e.g., recurrent breakthrough hemolysis, issues with effective self-administration, intolerable pain from infusion). The group indicated that the treatment and monitoring of patients should ideally be done by clinicians who specialize in the area, although patients can self-administer pegcetacoplan at home or anywhere they prefer.
Drug Program Input
Input was obtained from the drug programs that participate in the CADTH reimbursement review process. The following were identified as key factors that could potentially affect the implementation of a CADTH recommendation for pegcetacoplan:
relevant comparators
consideration for initiation of therapy
consideration for continuation or renewal of therapy
consideration of discontinuation of therapy
consideration for prescribing of therapy
generalizability
care provision issues
system and economic issues.
The clinical experts consulted by CADTH provided advice on the potential implementation issues raised by the drug programs. Refer to Table 4 for more details.
Clinical Evidence
Pivotal Studies and Protocol-Selected Studies
Description of Studies
The CADTH systematic review identified 1 relevant study, PEGASUS,5 which was a pivotal phase III, open-label, randomized controlled trial (RCT) comparing pegcetacoplan (1,080 mg twice weekly via subcutaneous infusion) with eculizumab (at a patient’s established dosage regimen via IV infusion) in adult patients with PNH who continued to have Hb levels of less than 10.5 g/dL despite treatment with eculizumab at a stable dosage for at least 3 months (N = 80). After receiving both interventions concurrently in a 4-week run-in period, patients were randomized to either pegcetacoplan or eculizumab monotherapy in a 1:1 ratio for a 16-week randomized controlled period. The primary outcome was change from baseline (before the run-in period) at week 16 in Hb (primary end point), and the key secondary end points were transfusion avoidance, change in baseline at week 16 in absolute reticulocyte count (ARC), LDH, and Functional Assessment of Chronic Illness Therapy (FACIT)-Fatigue score.
At baseline, the mean age was 48.8 years (standard deviation [SD] = 16.0 years) and the majority of patients were female and white (both 61.3%). Patients had a mean time since diagnosis of 10.2 years (SD = 8.6 years), had received eculizumab for a mean duration of 1,808.7 days (SD = 1,447.6 days) before the study, and 30% of patients received eculizumab at a dose higher than the maintenance dose approved for PNH by Health Canada. The study population had a mean Hb level of 8.7 g/dL (SD = 1.0 g/dL), reasonable control of IVH (mean LDH = 282.4 U/L; SD = 211.0 U/L), elevated mean ARC (mean = 216.9 × 109 cells/mL; SD = 71.7 × 109 cells/mL) and indirect bilirubin (mean = 33.8 µmol/L; SD = 25.8 µmol/L) levels, and low haptoglobin (mean = 0.135 g/L; SD = 0.121 g/L), all of which were consistent with the signs of EVH.
Efficacy Results
The key efficacy results from the PEGASUS trial are summarized in Table 2.
Table 2: Summary of Key Results From the Pivotal Trial — Randomized Controlled Period
Key outcomes | Pegcetacoplan (N = 41) | Eculizumab (N = 39) |
|---|---|---|
Hb (ITT; censored for transfusion) | ||
Patients included in the baseline analysis,a n (%) | 41 (100) | 39 (100) |
Baseline Hb level (g/dL), mean (SD) | 8.69 (1.08) | 8.68 (0.89) |
Patients included in the analysis at 16 weeks, n (%) | 36 (87.8) | 6 (15.4) |
Change from baseline at week 16 in Hb levela,b (g/dL), LS mean (SE) | 2.37 (0.36) | –1.47 (0.67) |
Difference in LS meansb (g/dL), (95% CI) | 3.84 (2.33 to 5.34) | Reference |
P valuec | < 0.0001 | Reference |
Hb response in the absence of transfusion,d n (%) | 31 (75.6) | 0 |
Risk differencee (95% CI) | 0.67 (0.55 to 0.80) | Reference |
Hb normalization in the absence of transfusion,f n (%) | 14 (34.1) | 0 |
Risk differencee (95% CI) | 0.30 (0.15 to 0.46) | Reference |
Transfusion avoidance (ITT; censored for transfusion) | ||
Patients achieving transfusion avoidance,g n (%) | 35 (85.4) | 6 (15.4) |
Risk differencee (%), (95% CI) | 62.5 (48.3 to 76.8) | Reference |
P valueh,i | < 0.0001 | Reference |
FACIT-Fatigue Scale (ITT; censored for transfusion) | ||
Patients included in the baseline analysis,a n (%) | 41 (100) | 38 (97.4) |
FACIT-Fatigue score, mean (SD) | 32.16 (11.38) | 31.55 (12.51) |
Patients included in the analysis at 16 weeks, n (%) | 35 (85.4) | 6 (15.4) |
Change from baseline at week 16 in FACIT-Fatigue score,a LS mean (SE) | 9.22 (1.61) | –2.65 (2.82) |
Difference in LS means (points),b (95% CI) | 11.87 (5.49 to 18.25) | Reference |
P valuej | 0.0005 | Reference |
LDH (ITT; censored for transfusion) | ||
Patients included in the baseline LDH analysis,a n (%) | 41 (100) | 39 (100) |
LDH level (U/L), mean (SD) | 257.5 (97.6) | 308.6 (284.8) |
Patients included in the LDH analysis at 16 weeks, n (%) | 35 (85.4) | 6 (15.4) |
Change from baseline at week 16 in LDH level (U/L),a,b LS mean (SE) | –14.8 (42.7) | –10.1 (71.0) |
Difference in LS means (U/L),b (95% CI) | –4.6 (–181.3 to 172.0) | Reference |
P valueh | 0.96 | Reference |
ARC (ITT; censored for transfusion) | ||
Patients included in the baseline ARC analysis, n (%) | 41 (100) | 39 (100) |
ARC (109 cells/L), mean (SD) | 217.5 (75.0) | 216.2 (69.1) |
Patients included in the ARC analysis at 16 weeks, n (%) | 34 (82.9) | 6 (15.4) |
Change from baseline in ARC at week 16a,b (109 cells/L), LS mean (SE) | –135.8 (6.5) | 27.8 (11.9) |
Difference in LS meansb (109 cells/L), (95% CI) | –163.6 (–189.9 to –137.3) | Reference |
P valueh | < 0.0001 | Reference |
Harms (safety set) | ||
TEAEs, n (%) | 36 (87.8) | 34 (87.2) |
Serious TEAEs, n (%) | 7 (17.1) | 6 (15.4) |
Withdrawal from treatment due to TEAE, n (%) | 3 (7.3) | 0 |
Deaths, n (%) | 0 | 0 |
Notable harms (safety set) | ||
Serious bacterial infection–related TEAE, n (%) | 1 (2.4) | 0 |
Injection site–related TEAE, n (%) | 15 (36.6) | 1 (2.6) |
Antipegcetacoplan peptide antibody-positive, n (%) | 0 | 2 (5.1) |
Thrombosis, n (%) | 0 | 0 |
Hemolysis, n (%) | 4 (9.8) | 9 (23.1) |
ARC = absolute reticulocyte count; CI = confidence interval; FACIT = Functional Assessment of Chronic Illness Therapy; Hb = hemoglobin; ITT = intention to treat; LDH = lactate dehydrogenase; LS = least square; SD = standard deviation; SE = standard error; TEAE = treatment-emergent adverse event.
aBaseline was defined as the average of measurements recorded before the first dose of pegcetacoplan.
bThe analysis was conducted using a mixed model for repeated measures (MMRM), with change from baseline at week 16 in outcome measure as the dependent variable. The model was adjusted for treatment group, study visit, number of packed RBC transfusions in the previous 12 months (< 4 vs. ≥ 4), platelet count at screening (< 100,000/mm3 vs. ≥ 100,000/mm3), the study visit by treatment group interaction, and baseline measurement level (continuous).
cThe significance threshold was set at a 2-sided alpha of 0.05.
dHb response was defined as an increase of at least 1 g/dL in Hb from baseline, excluding data before the randomized controlled period.
eThe risk difference and 95% CI were constructed using the stratified Miettinen-Nurminen method.
fHb normalization was defined as a Hb level at or above the lower limit of the normal range.
gTransfusion avoidance was defined as the proportion of patients who did not require a transfusion during the randomized controlled period. Patients who did not have a transfusion but withdrew before week 16 were considered to have had a transfusion.
hIn accordance with the hierarchal testing procedure, noninferiority but not superiority testing was conducted.
iThe P value was based on Cochran-Mantel-Haenszel chi-square test stratified by the number of packed RBC transfusions in the previous 12 months (< 4 vs. ≥ 4) and the platelet count at screening (< 100,000/mm3 vs. ≥ 100,000/mm3).
jIn accordance with the hierarchal testing procedure, neither noninferiority nor superiority testing was conducted.
Source: PEGASUS Clinical Study Report (16 weeks).6
Survival
Survival was not assessed in the efficacy analysis.
Hb Outcomes
Change from baseline at week 16 in Hb level was the primary end point. The mean Hb level at baseline was 8.69 g/dL (SD = 1.08 g/dL) in the pegcetacoplan arm and 8.68 g/dL (SD = 0.89 g/dL) in the eculizumab arm. The least square (LS) mean change from baseline at week 16 in Hb level (censored for transfusion) was 2.37 g/dL (standard error [SE] = 0.36 g/dL) in the pegcetacoplan arm and –1.47 g/dL (SE = 0.67 g/dL) in the eculizumab arm, with a between-group difference of 3.84 g/dL (95% confidence interval [CI], 2.33 to 5.34 g/dL) in favour of pegcetacoplan (P < 0.0001). Results of the sensitivity analyses (controlled-based pattern imputation and tipping point analyses) and supportive analysis using all patient data (uncensored for transfusion) were consistent with the primary analysis.
Hb response (i.e., at least 1 g/dL increase) in the absence of transfusion (secondary end point) was achieved in 75.6% of patients in the pegcetacoplan arm and 0% of patients in the eculizumab arm, with an adjusted risk difference of 67.5% (95% CI, 54.5% to 80.4%). Hb normalization in the absence of transfusion at week 16 (secondary end point) was achieved in 34.1% of patients in the pegcetacoplan arm and 0% of patients in the eculizumab arm, with an adjusted risk difference of 30.4% (95% CI, 14.9% to 45.9%). The differences between treatments for both outcomes were not tested for statistical significance.
Transfusion
Transfusion avoidance was a key secondary end point and was tested for noninferiority according to the hierarchal testing procedure. Transfusion avoidance was achieved in 85.4% of patients in the pegcetacoplan arm and 15.4% of patients in the eculizumab arm, with an adjusted risk difference of 62.5% (95% CI, 48.3% to 76.8%) in the intention-to-treat (ITT) analysis. The lower bound of the 95% CI of risk difference was greater than the noninferiority margin (NIM) of –20% in both the ITT and per-protocol (PP) analysis sets, the supporting noninferiority of pegcetacoplan to eculizumab.
The mean number of packed RBC units transfused (secondary end point) was 0.6 units (SD = 2.03 units) in the pegcetacoplan arm and 5.1 units (SD = 5.6 units) in the eculizumab arm. The difference between treatments was not tested for statistical significance.
Thrombotic Events
Thrombotic events were not assessed in the efficacy analysis.
Symptoms of PNH
Change from baseline at week 16 in FACIT-Fatigue score was a key secondary end point but was not tested for inferiority or superiority because of prior failure in the testing hierarchy. The LS mean change from baseline at week 16 in the FACIT-Fatigue score in the ITT set (censored for transfusion) was 9.22 points (SE = 1.61 points) in the pegcetacoplan arm and –2.65 points (SE = 2.82 points) in the eculizumab arm, with a between-group difference in LS means of 11.87 points (95% CI, 5.49 to 18.25 points).
A responder analysis that assessed the proportion of patients with at least a 3-point increase in the FACIT-Fatigue score from baseline at week 16 (censored for transfusion) was conducted, and the proportion was 73.2% in the pegcetacoplan arm and 0% in the eculizumab arm. The differences between treatment arms were not tested for statistical significance.
Health-Related Quality of Life
Change from baseline at week 16 in Linear Analogue Scale Assessment (LASA) and European Organisation for Research and Treatment of (EORTC QLQ-C30) scores were secondary end points. The LS mean between-group difference in change from baseline at week 16 in LASA score (censored for transfusion) was 59.1 points (95% CI, 16.9 to 101.3 points). The LS mean between-group differences in LS mean change from baseline at week 16 in EORTC QLQ-C30 global health status, fatigue, pain, and dyspnea scores (censored for transfusion) were, respectively, 18.62 points (95% CI, 0.12 to 37.13 points) points, –20.74 points (95% CI, –35.29 to –6.19 points), –2.76 points (95% CI, –20.36 to 14.85 points), and –14.57 points (95% CI, –29.90 to 0.76 points). The differences between treatment arms for all health-related quality of life (HRQoL) outcomes were not adjusted for multiplicity.
Breakthrough Hemolysis
Breakthrough hemolysis was not assessed in the efficacy analysis.
Complications of PNH Other Than Thrombotic Events
Complications of PNH were not assessed in the efficacy analysis.
Hemolytic Parameters
Change from baseline at week 16 in LDH and ARC were key secondary end points and were tested for noninferiority, according to the hierarchal testing procedure.
The mean LDH level at baseline was 257.5 U/L (SD = 97.6 U/L) in the pegcetacoplan arm and 308.6 U/L (SD = 284.8 U/L) in the eculizumab arm. The LS mean change from baseline at week 16 in LDH level (censored for transfusion) was –14.8 U/L (SE = 42.7 U/L) in the pegcetacoplan arm and –10.1 U/L (SE = 71.0 U/L) in the eculizumab arm, with a between-group difference in LS means of –4.6 U/L (95% CI, –181.3 to 172.0 U/L). Noninferiority was not met because the upper bound of the 95% CI of the between-group difference was not less than the NIM of 20 U/L in both the ITT and PP sets. Results of a supportive analysis based on data uncensored for transfusion were consistent with the primary analysis.
The mean ARC at baseline was 217.5 × 109 cells/L (SD = 75.0 × 109 cells/L) in the pegcetacoplan arm and 216.2 × 109 cells/L (SD = 69.1 × 109 cells/L) in the eculizumab arm. The LS mean change from baseline in ARC at week 16 in the ITT set (censored for transfusion) was –135.8 × 109 cells/L (SE = 6.5 × 109 cells/L)in the pegcetacoplan arm and 27.9 × 109 cells/L (SE = 11.9 × 109 cells/L) in the eculizumab arm, with a between-group adjusted mean difference of –163.6 × 109 cells/L (95% CI, –189.9 to –137.3 × 109 cells/L). Noninferiority was met because the upper bound of the 95% CI of the adjusted mean difference was less than the prespecified NIM of 10 × 109 cells/L in the ITT set, and results were consistent in the PP set. Results of a supportive analysis based on data uncensored for transfusion were also consistent with the primary analysis.
LDH normalization in the absence of transfusion (secondary end point) was achieved in 70.7% of patients in the pegcetacoplan arm and 15.4% of patients in the eculizumab arm, with an adjusted risk difference of 48.8% (95% CI, 32.3% to 65.3%). LDH normalization (uncensored for transfusion) was achieved in 73.2% of patients in the pegcetacoplan arm and 59.0% of patients in the eculizumab arm, with a risk difference of 12.3% (95% CI, 7.0% to 31.5%). The difference between treatment arms was not tested for statistical significance.
Reticulocyte normalization in the absence of transfusion (secondary end point) was achieved in 78.0% of patients in the pegcetacoplan arm and 2.6% of patients in the eculizumab arm, with a risk difference of 66.4% (95% CI, 53.1% to 79.7%). Reticulocyte normalization (uncensored for transfusion) was 80.5% in the pegcetacoplan arm and 17.9% in the eculizumab arm, with a risk difference of 54.8% (95% CI, 38.8% to 70.7%). The difference between treatment arms was not tested for statistical significance.
Health Care Resource Utilization
Health care resource utilization was not assessed in the study.
Harms Results
In the run-in period, treatment-emergent adverse events (TEAEs) were reported in 69 patients (86.3%), but none led to death or discontinuation of the study treatment or the study. A serious TEAE was reported in 1 patient who developed sepsis, which resolved during the run-in period despite continued treatment with pegcetacoplan and eculizumab.
The key harms results from the randomized controlled period of the PEGASUS trial are summarized in Table 2.
TEAEs were reported in 87.8% of patients in the pegcetacoplan arm and 87.2% of patients in the eculizumab arm. The most common TEAEs related to pegcetacoplan (in at least 10% of patients) were diarrhea, injection-site erythema, injection site reaction, and abdominal pain. There was a similar incidence of serious TEAEs in both arms (pegcetacoplan: 17.1%; eculizumab 15.4%). Withdrawal from study treatment due to TEAEs occurred in 3 (7.3%) patients in the pegcetacoplan arm, all due to breakthrough hemolysis. No patients in the eculizumab withdrew from the study treatment due to a TEAE. No deaths were reported in either arm.
The pegcetacoplan arm had a notably higher incidence of injection site–related TEAEs (36.6%) than the eculizumab arm (2.6%). Breakthrough hemolysis was reported less frequently in the pegcetacoplan arm (9.8%) than in the eculizumab arm (23.1%). In the pegcetacoplan arm, there was no report of thrombosis or antipegcetacoplan peptide antibody response, and 1 patient-reported serious treatment-emergent bacterial infection but was unrelated to encapsulated organism. There were no reports of pulmonary hypertension or chronic kidney disease.
Critical Appraisal
Appropriate methods of randomization were used. Although imbalances in some baseline characteristics between treatment groups were noted, none was expected to cause confounding. The open-label design could introduce reporting bias for subjective efficacy end points (i.e., FACIT-Fatigue, LASA, EORTC QLQ-C30) in favour of pegcetacoplan. The high number of major protocol deviations related to study assessment or schedule noncompliance could compromise the completeness and reliability of study data, introducing uncertainties to the results, although the direction and extent of bias are unclear. The statistical analyses were generally well designed, with adequate sample size and appropriate multiplicity adjustments for all key secondary outcomes. Other secondary outcomes were either not tested for statistical significance or not adjusted for multiplicity. No justification was provided for the chosen NIMs, although the NIMs were considered reasonable by the clinical experts. Supportive PP analyses were conducted for end points tested for noninferiority, and results were consistent with the primary ITT analysis. There was a high amount of missing data due to censoring for transfusion. Nonetheless, with respect to the primary end point (change in Hb), results from the sensitivity analyses and supportive analysis using different imputation methods and censoring rules were consistent with the primary analysis, increasing certainty of the findings. It is unclear if Hb improvement is a predictor of long-term clinical outcomes, given that long-term studies are scarce for this rare disease. The reliability, validity, and responsiveness of the FACIT-Fatigue, LASA, and EORTC QLQ-C30 scales have not been previously characterized in patients with PNH, which limits conclusions that can be made on these outcomes.
There was no major concern about the generalizability of the study population, given that the inclusion and exclusion criteria and patient baseline characteristics were consistent with clinical practice. Patients with intolerance to eculizumab were not included; however, this represents a very small population of patients in clinical practice, per clinical expert input. Eculizumab was considered a representative comparator, and the distribution of eculizumab dosing aligns with clinical practice. The follow-up duration was adequate for assessing the efficacy outcomes included in the study, but inadequate for other clinically important outcomes, such as breakthrough hemolysis, survival, thrombosis, and other complications of PNH. The clinical relevance of the FACIT-Fatigue, EORTC QLQ-C30, and LASA instruments is uncertain because they are not used in clinical practice, although they did capture some common symptoms of PNH (e.g., fatigue, dyspnea, pain) reported by patients.
Indirect Comparisons
Description of Studies
The sponsor submitted an anchored matching-adjusted indirect comparison (MAIC) to evaluate the relative efficacy of pegcetacoplan, compared to ravulizumab, in adult patients with PNH previously treated with eculizumab.7 The MAIC did not report a systematic literature review to identify relevant studies for inclusion. Two studies were included in the analysis: patient-level data from the PEGASUS study, which compared pegcetacoplan with eculizumab; and aggregate patient data from the ALXN1210-PNH-302 study (referred to as the ALXN study in this report),8 which compared ravulizumab with eculizumab. Outcomes analyzed were transfusion avoidance, number of packed RBCs transfused, Hb level stabilization, change from baseline in LDH level, LDH level normalization, fatigue and fatigue symptoms, global health status, and physical functioning. A propensity score model using logistic regression was used to account for between-study differences in patient baseline characteristics. Effect modifiers were matched in the weighting process separate for clinical and hematological outcomes and fatigue and QoL outcomes. Compared with patients who received ravulizumab, a greater proportion of patients who received pegcetacoplan had a history of transfusions in the year before the study (72.2% versus 13.4%), and mean Hb was lower for patients who received pegcetacoplan (8.7 g/dL versus 11.1 g/dL). There was no adjustment for transfusion history or mean Hb in the analysis.
Efficacy Results
After matching and anchoring on eculizumab, treatment with pegcetacoplan compared with ravulizumab was associated with more transfusion avoidance (adjusted difference = 71.4%; 95% CI, 53.5% to 89.3%), more Hb level stabilization (adjusted difference = 75.5%; 95% CI, 56.4% to 94.6%), more LDH level normalization in the absence of transfusions (adjusted difference = 64.0%; 95% CI, 41.8% to 86.1%), and fewer mean units of packed RBCs transfused (adjusted difference = –5.7 units; 95% CI, –7.2 to –4.2 units). In addition, treatment with pegcetacoplan, compared with ravulizumab, was associated with improvements in adjusted difference in mean change from baseline in fatigue (8.2 points; 95% CI, 3.8 to 12.6 points), global health status (9.6 points; 95% CI, 0.1 to 19.0 points), physical functioning (11.5 points; 95% CI, 3.6 to 19.5 points), and fatigue symptoms (–13.3 points; 95% CI, –23.7 to –3.0 points). There was no difference in the mean change from baseline in LDH levels (adjusted mean difference = 0.3 U/L; 95% CI, –154.5 to 155.1 U/L).
Critical Appraisal
The anchored MAIC has several limitations, including the inability to adjust for 2 clinically important effect modifiers (Hb level and history of transfusions) — which differed substantially between the 2 studies at baseline, the heterogeneity between studies regarding duration of follow-up (i.e.,16 weeks for PEGASUS versus 26 weeks for ALXN), the treatment administration schedule, and the dosing regimen. These limitations may introduce unmeasurable confounding in the relative treatment effect estimates, although the direction and extent of bias are unclear. Overall, there is uncertainty in the relative treatment effect estimates favouring pegcetacoplan over ravulizumab.
Other Relevant Evidence
Description of Study
Safety and efficacy results from the 32-week, single-arm, extension period (after the randomized controlled period) for the PEGASUS trial (n = 77), during which all patients received pegcetacoplan, were also submitted by the sponsor and are presented in this report.
Efficacy Results
The results were reported as summary statistics and indicated that efficacy — assessed with Hb level, transfusion avoidance, ARC, LDH level, FACIT-Fatigue Scale score, LASA scores, EORTC QLQ-C30 scores, as well as the number of packed RBC units transfused — was generally maintained in the pegcetacoplan to pegcetacoplan group for another 32 weeks after the randomized controlled period. In the eculizumab to pegcetacoplan group, improvement was consistently observed across all outcomes after initiation of pegcetacoplan, and the patients in this group achieved benefits from pegcetacoplan similar to those seen in the pegcetacoplan to pegcetacoplan group.
Harms Results
The TEAE profile of pegcetacoplan was generally consistent with the randomized controlled period. Hemolysis was reported in 15 (19.5%) patients and led to treatment discontinuation in 2 patients. Thrombosis was reported in 2 (2.6%) patients, but neither was deemed by the sponsor to be related to pegcetacoplan. There were no reports of serious bacterial infection or renal failure. The incidence of pulmonary hypertension was not reported.
Critical Appraisal
The noncomparative design of the extension period precludes the drawing of conclusions about the comparative efficacy of pegcetacoplan and eculizumab beyond 16 weeks. It is difficult to ascertain if the observed effects can be attributed to pegcetacoplan alone because of the lack of control for confounding in a nonrandomized study. No formal statistical testing was conducted, and results were presented using descriptive statistics. The open-label design can influence the reporting of subjective outcomes (FACIT-Fatigue, LASA, EORTC QLQ-C30, harms), introducing uncertainties to the results. Although the study results seem to suggest that the efficacy and safety of pegcetacoplan can be maintained long-term, outcomes such as survival, thrombosis, and other complications of PNH require a much longer duration of follow-up to provide certainty in the results.
Conclusions
In the PEGASUS trial, pegcetacoplan demonstrated a clinically meaningful improvement in Hb level compared with eculizumab in a 16-week randomized controlled period in a study population that was representative of patients with PNH who had clinically significant anemia despite an adequate trial of eculizumab and who had signs of EVH. The evidence strongly suggested that Hb improvement translated into an improvement in transfusion avoidance with pegcetacoplan than with eculizumab. Results also suggested that pegcetacoplan could reduce fatigue, compared with eculizumab, but the magnitude of benefit and its clinical relevance is uncertain. Analyses of hemolytic markers lent support to the ability of pegcetacoplan to reduce EVH and maintain IVH control. The benefits of pegcetacoplan were sustained through week 48 in the extension period, but the single-arm design precludes conclusions about the comparative efficacy of pegcetacoplan and eculizumab beyond week 16. The sponsor-submitted MAIC assessing the comparative efficacy of pegcetacoplan and ravulizumab showed that transfusion avoidance, number of packed RBCs transfused, Hb level stabilization, LDH level normalization, fatigue and fatigue symptoms, global health status, and physical functioning all favoured pegcetacoplan; however, the findings should be interpreted with caution, given the lack of matching of some effect modifiers and the heterogeneity in study designs and populations. The safety profile of pegcetacoplan was generally similar to that of eculizumab, but with a higher incidence of nonserious injection site–related reactions and diarrhea. A longer duration of follow-up is required to provide certainty about the incidence of breakthrough hemolysis, thrombosis, and survival with pegcetacoplan treatment.
Introduction
Disease Background
PNH is an extremely rare chronic disease characterized by IVH and heterogenous signs and symptoms that include hemoglobinuria, anemia, abdominal pain, fatigue, dyspnea, dysphagia, and erectile dysfunction.1 Complications of PNH include thrombosis, chronic kidney disease, and pulmonary hypertension.2 In Canada, the median age of disease onset has been estimated at 43 years.1 Although the incidence of PNH has not been extensively characterized, 1 study in the UK estimated an annual incidence of clinical PNH of approximately 0.13 per 100,000 persons.3 PNH is a consequence of an acquired genetic mutation, leading to clonal expansion of hematopoietic stem cells that produce abnormal RBCs, leukocytes, and platelets deficient in glycophosphatidylinositol anchor proteins.2 These abnormal RBCs are susceptible to complement-mediated IVH. PNH may develop as subclinical PNH, with no substantial clinical findings and no bone marrow abnormalities;9 as hemolytic PNH, typically with a white blood cell clone size of greater than 10%; or in association with a bone marrow disorder, such as aplastic anemia or myelodysplastic syndrome, which is often accompanied by smaller clone sizes (i.e., the proportion of blood cells deficient in glycophosphatidylinositol anchor protein).1 Studies examining the survival of patients with PNH after diagnosis show a range of median survival, from 14.6 years to 32 years,10,11 whereas results from a study of patients with PNH treated with eculizumab suggested that their survival was similar to that of age-matched controls.12
It is estimated that 20% to 30% of patients have ongoing and clinically significant anemia, despite standard-of-care treatment with C5 inhibitors, per input from clinical experts consulted by CADTH. According to the Canadian PNH Network, there is no consensus on the method to assess response to anticomplement treatment, and the group suggested that response evaluation be based on symptoms, transfusion requirements, LDH level, and complete blood count parameters.1 Response categories were proposed by DeZern et al. (2013) as follows13:
complete response, defined as transfusion independence, normal Hb for age and sex, absence of symptoms, LDH less than 1.5 times the ULN
good partial response, defined as decreased transfusions from pretreatment status, LDH less than 1.5 times the ULN, no thromboembolism after the start of therapy, and Hb remaining low for age and sex
suboptimal response, defined as unchanged transfusion needs and persistent or new symptoms after the start of therapy.
An international working group proposed a response classification system based on similar parameters (Table 42 in Appendix 4).14
The Canadian PNH Network noted the following causes of eculizumab failure1:
breakthrough (intravascular) hemolysis, the most common cause of treatment failure; it can be due to situational events that amplify complement activity (e.g., surgery, infection) or insufficient dosing of eculizumab, which usually occurs near the end of each dosing cycle
C3-mediated extravascular hemolysis, an iatrogenic effect of C5 inhibitor treatment caused by increased C3 disposition on erythrocytes, resulting in erythrocytes being opsonized with C3 fragments and destroyed via extravascular destruction
the development of human-antihuman or eculizumab-neutralizing antibodies (low incidence)
C5 genetic polymorphism, which prevents eculizumab from binding to its target molecule and is mostly found in patients of Japanese descent (low incidence).
The clinical experts noted that ongoing anemia could also be caused by issues such as insufficient bone marrow function, renal insufficiency, and deficiency in iron, folate, and vitamin B12, and by gastrointestinal bleed, which should be thoroughly assessed before the underlying cause(s) of C5 inhibitor treatment failure and appropriate management strategies can be determined.
Standards of Therapy
For patients with subclinical PNH (i.e., asymptomatic and no bone marrow failure), no treatment intervention is usually required and they are managed with watchful waiting.15 For patients with hemolytic PNH and associated bone marrow disorders, the only curative therapy is allogeneic stem cell transplant; however, due to significant morbidities and mortality, hematopoietic cell transplant is generally reserved for patients with severe bone marrow failure due to bone marrow disorders or patients with PNH complications who are unresponsive to C5 inhibitors.15,16
According to the clinical experts consulted by CADTH, in patients diagnosed with hemolytic PNH who have high disease activity (i.e., symptomatic and LDH above 1.5 times the ULN [indicative of clinically significant IVH]), the current standard of care in the first-line setting are terminal C5 inhibitors (i.e., eculizumab, ravulizumab), which have been shown to effectively reduce uncontrolled complement activation and its complications. In patients who have an inadequate response to C5 inhibitors, clinical management can vary, depending on the underlying cause of treatment failure. According to the Canadian PNH Network1 and input from the clinical experts, current treatment approaches in Canada for these patients are as follows:
Breakthrough (intravascular) hemolysis: If due to situational events that amplify complement activity, address the underlying cause of the breakthrough event. If due to insufficient dosing of eculizumab, increase the dose of eculizumab (e.g., 1,200 mg every 14 days) or shorten infusion intervals (e.g., every 12 days). This should lead to appropriate complement inhibition in 98% of patients.17
C3-mediated EVH: According to the clinical experts, before the approval of pegcetacoplan, there was no approved subsequent therapy for patients with ongoing anemia due to EVH in Canada. The treatment approach is to provide best supportive care (e.g., RBC transfusion, corticosteroids, splenectomy, danazol, epoetin alfa) while continuing C5 inhibitor treatment (to prevent life-threatening IVH), although many best supportive care therapies are associated with toxicities. Alternatively, patients could be referred to clinical trials of proximal complement inhibitors (e.g., C3 or factor D inhibitor). In patients with concurrent bone marrow insufficiency, immunosuppressive therapy should be considered as an add-on therapy.
C5 genetic polymorphism: Referral to clinical trials of proximal complement inhibitors should be considered because patients with this genetic trait are not expected to respond to C5 inhibitor treatments.
Vaccination, with or without prophylactic antibiotics, for meningococcal infections is recommended for all patients receiving complement inhibitors.1
The clinical experts noted that the goals of treatment of PNH include increasing Hb level, avoiding transfusion, preventing hemolysis and chronic complications of PNH, and improving QoL and life expectancy.
Drug
Pegcetacoplan is a proximal complement inhibitor that binds to C3 and its activation fragment C3b, thereby regulating the cleavage of C3 and the generation of downstream effectors of complement activation and inhibiting complement-mediated extravascular and IVH.
This is the first review for pegcetacoplan by CADTH. Pegcetacoplan was granted a Health Canada Notice of Compliance for the indication of treatment of adult patients with PNH who have an inadequate response to, or are intolerant of, a C5 inhibitor. The sponsor is seeking reimbursement of pegcetacoplan for that indication.
Per the product monograph, the recommended dosage of pegcetacoplan is 1,080 mg twice weekly by subcutaneous infusion with a syringe system infusion pump administered by a health care professional, the patient, or a caregiver.4 It is recommended that pegcetacoplan be administered in addition to the patient’s current dose of C5 inhibitor treatment for the first 4 weeks to minimize the risk of hemolysis due to abrupt treatment discontinuation.4 A dosage increase to 1,080 mg every third day may be considered if the LDH level is greater than 2 times the ULN with twice-weekly dosing.4 It is recommended that treatment with pegcetacoplan continue for the patient’s lifetime, unless discontinuation is clinically indicated.4
The key characteristics of treatments for PNH are summarized in Table 3.
Table 3: Key Characteristics of Pegcetacoplan, Eculizumab, and Ravulizumab
Characteristic | Pegcetacoplan | Eculizumab | Ravulizumab |
|---|---|---|---|
Mechanism of action | C3 inhibitor; proximal complement inhibition | C5 inhibitor; terminal complement inhibition | C5 inhibitor; terminal complement inhibition |
Indicationa | For the treatment of adult patients with PNH who have an inadequate response to, or are intolerant of, a C5 inhibitor | For the treatment of patients with PNH to reduce hemolysis | For the treatment of adult patients with PNH |
Route of administration | SC | IV | IV |
Recommended dose | 1,080 mg twice weekly Dose adjustment: 1,080 mg every third day if LDH > 2 × ULN Pegcetacoplan should be administered in addition to the patient’s current dose of C5 inhibitor treatment for the first 4 weeks of treatment to minimize the risk of hemolysis with abrupt treatment discontinuation | 600 mg every 7 days for the first 4 weeks, followed by 900 mg for the fifth dose 1 week later, then 900 mg every 2 weeks thereafterb | One loading dose, and 2 weeks later, start maintenance dose once every 8 weeks thereafter Weight-based dosing:
|
Serious adverse effects or safety issues | Meningococcal infections | Meningococcal infections | Meningococcal infections and sepsis |
Other |
|
|
|
C3 = complement component 3; C5 = complement component 5; LDH = lactate dehydrogenase; PNH = paroxysmal nocturnal hemoglobinuria; SC = subcutaneous; ULN = upper limit of normal.
aHealth Canada–approved indication.
bDose escalation of eculizumab to 1,200 mg every 14 weeks or reduction of dosing interval to 900 mg every 12 days is considered in patients with PNH experiencing breakthrough hemolysis in clinical practice, per the Canadian PNH Network.1
Sources: Product monographs of pegcetacoplan,4 eculizumab,18 and ravulizumab.19
Stakeholder Perspectives
Patient Group Input
This section was prepared by CADTH staff based on the input provided by patient groups. The full original patient input received by CADTH has been included in the stakeholder section at the end of this report.
The Canadian Association of PNH Patients and the Aplastic Anemia & Myelodysplasia Association of Canada submitted a joint input for this review. Information was gathered through 1-on-1 interviews with 6 individuals diagnosed with PNH living in Canada and from the scientific literature. The patient group expressed the following negative impacts of PNH: persistent anemia (manifested as fatigue or extreme fatigue), hemolysis leading to thrombosis, employment absenteeism for patients and caregivers, dependence on frequent blood transfusions, and reduced QoL. According to the input, patients with PNH, particularly those experiencing EVH, need alternative treatment options because of the inability of eculizumab or ravulizumab to thoroughly control IVH and prevent EVH. The patient group also expressed the need for therapies to improve anemia, reduce or eliminate transfusion requirements, and improve fatigue and QoL. Among 3 patients who have used pegcetacoplan, they noted an immediate normalization of hematological parameters, easier administration (self-administered easily through subcutaneous infusion twice weekly at home) than with eculizumab or ravulizumab (visits for IV transfusions), reduced blood transfusions, and improved physical functioning and QoL. Some patients stated the importance of proper injection training.
Clinician Input
Input From Clinical Experts Consulted by CADTH
All CADTH review teams include at least 1 clinical specialist with expertise in the diagnosis and management of the condition for which the drug is indicated. Clinical experts are a critical part of the review team and are involved in all phases of the review process (e.g., providing guidance on the development of the review protocol, assisting in the critical appraisal of clinical evidence, interpreting the clinical relevance of the results, and providing guidance on the potential place in therapy). The following input was provided by 2 clinical specialists with expertise in the diagnosis and management of PNH.
Unmet Needs
The clinical experts noted that C5 inhibitors are effective in controlling IVH; however, not all patients respond to treatment. While on C5 inhibitor treatment, some patients remain anemic, with chronic hemolysis and/or acute hemolytic crisis, requiring ongoing transfusions despite no evidence of bone marrow failure. The clinical experts also noted that none of the currently approved treatments addresses C3-mediated EVH, and clinical trial enrolment is the only alternative. Although best supportive care measures are available to patients who have an inadequate response to C5 inhibitors, they are not always effective and are associated with significant toxicities, according to the clinical experts. Transfusion, the mainstay supportive measure, is associated with complications such as risk of iron and fluid overload, infection, and poor QoL, and access is limited by blood shortages. Transfusion is only performed when a patient is severely anemic (i.e., Hb less than 7 g/dL to 8 g/dL). Corticosteroids are associated with a lot of side effects (e.g., osteoporosis, corticosteroid dependence, adrenal insufficiency, opportunistic infections, dyspepsia), and epoetin alfa is associated with a theoretical risk of thrombosis. Bone marrow transplant is a curative treatment for patients with PNH; however, the clinical experts noted that few patients are candidates and bone marrow transplant is associated with high morbidity and mortality.
The clinical experts noted there is a need for treatments that are effective in patients who have insufficient control of IVH despite treatment with eculizumab, that address the issue of C3-mediated EVH, and that can be used in patients with intolerance to C5 inhibitors (although rare in clinical practice) or difficult venous access.
Place in Therapy
The clinical experts noted that pegcetacoplan is a proximal C3 inhibitor and has a mechanism of action in addition to existing treatments because C3 is a more upstream effector than C5 in the complement activation pathway. However, the clinical experts anticipated that pegcetacoplan will mainly serve as a second-line treatment in patients who are intolerant to C5 inhibitors or who have an inadequate response to C5 inhibitors. The clinical experts noted that C5 inhibitors will likely remain the preferred treatment for PNH in a first-line setting because clinicians have much more experience with C5 inhibitors than with pegcetacoplan in treatment-naive patients.
The clinical experts did not expect pegcetacoplan to cause a major shift in the current treatment paradigm because it is reserved for second-line and will only be used in select patients who are expected to benefit from treatment, based on clinical judgment by clinicians and individual patient needs. The clinical experts noted the drug would instead allow for a greater proportion of patients with improved treatment outcomes.
Patient Population
The clinical experts noted that appropriate candidates for pegcetacoplan treatment include patients with PNH who have persistent anemia (with or without history of ongoing blood transfusion needs) and evidence of EVH, despite an adequate trial of C5 inhibitor treatment; patients with intolerance to a C5 inhibitor (uncommon in clinical practice); and patients with a rare C5 genetic polymorphism (mainly in patients of Japanese descent). The clinical experts noted that pegcetacoplan could potentially be considered in patients who are geographically isolated or who have poor venous access. Patient with a PNH clone size of less than 10% should not receive pegcetacoplan, according to the clinical experts.
The clinical experts noted the following laboratory tests that help inform eligibility assessment: complete blood count, LDH, Coombs test, and blood tests for end organ damage; complement testing is not readily available at the moment and treatment decisions are not based on it. The clinical experts noted that C3-loading is demonstrated in most patients receiving eculizumab, but only 10% to 20% of patients developed sufficient C3-loading to cause clinically significant anemia. According to the clinical experts, suspicion of C3-mediated EVH would come when there are ongoing low Hb levels, elevated reticulocyte counts and bilirubin levels (indicating presence of hemolysis), but normal or slightly elevated LDH levels (indicating reasonable control of IVH). Additional laboratory tests are required to rule out other causes of ongoing anemia, such as IVH, inadequate bone marrow function, iron or vitamin B12 deficiency, and gastrointestinal bleed. The clinical experts noted that because all patients with PNH are expected to be under the care of a PNH expert, all of the above factors would be taken into consideration when the treating physician selects suitable candidates for pegcetacoplan. The clinical experts considered ongoing anemia with Hb levels of less than 10 g/dL to be indicative of a suboptimal response to a C5 inhibitor, recognizing that there is variability in the cut-offs used by other clinicians.
The clinical experts noted that it is possible that clinicians may fail to identify patients who can benefit from a switch to pegcetacoplan treatment, although the chance is low because all patients are presumably treated by PNH experts.
Assessing Response to Treatment
The clinical experts noted that a clinically meaningful treatment response would include improvement in hemolytic parameters (LDH, bilirubin) and Hb level, reduced transfusion needs, and improved QoL. The clinical experts added that normalization of laboratory parameters may not be a realistic goal in every patient; treatment response is generally determined, per clinical judgment, by the treating physician, based on a global assessment of all patient parameters, chronology of symptoms, and laboratory results.
The clinical experts noted that follow-up assessments are usually conducted every 3 months, although more frequent follow-up visits are warranted for patients who have recently switched to pegcetacoplan for closer monitoring of symptoms and laboratory parameters until patients become stabilized. Patients are also monitored with PNH flow cytometry every 6 months to 12 months for changes in their clone size.
Discontinuing Treatment
The clinical experts noted that once a patient is switched to pegcetacoplan, it is unlikely that pegcetacoplan would be discontinued unless 1 of the following occurs: treatment failure (persistent anemia and ongoing transfusion needs) necessitating a switch to a more effective treatment, intolerance to pegcetacoplan, or resolution of disease after bone marrow transplant.
Prescribing Conditions
The clinical experts noted that the diagnosis, treatment, and monitoring of patients with PNH should be managed by hematologists. Given the rarity of the disease, consultation with hematology specialists who have expertise in the management of PNH is encouraged.
Clinician Group Input
This section was prepared by CADTH staff based on the input provided by clinician groups. The full original clinician group input received by CADTH has been included in the stakeholder section at the end of this report.
One clinician group, the Canadian PNH Network, submitted input for this review based on contributions from 11 clinicians. The group noted that the current standard of care comprises C5 inhibitors (i.e., eculizumab and ravulizumab), which act via terminal complement blockade. They noted that although C5 inhibitors are not curative treatment, these treatments have been shown to be effective in controlling IVH, leading to significant improvement in fatigue, QoL, transfusion dependence, thrombosis, and overall survival. The only curative treatment for PNH is allogeneic hematopoietic stem cell transplant and is available to patients with predominant or progressive bone marrow failure or to eligible patients with evidence of clonal evolution. However, the group highlighted 3 unmet needs: some patients do not have access to standard care due to highly restrictive reimbursement criteria; eculizumab has a high treatment burden because it requires venous access for administration every 2 weeks and nurse visits; and, despite treatment, approximately one-third of patients remain anemic due to EVH and some are transfusion-dependent. The group explained that drugs that exploit proximal complement blockade, such as C3 inhibitors, address EVH risk, significantly increase Hb, and improve QoL. Regarding its place in therapy, the group stated that pegcetacoplan is the first C3 inhibitor that protects against EVH, and it would fit into the current treatment landscape as an alternative (i.e., switch) option for patients with no or inadequate response or intolerance to eculizumab or ravulizumab. Eligible patients would include those with persistent anemia (Hb less than 10.5 g/dL or perhaps higher, if symptomatic) despite stable doses of eculizumab or ravulizumab who have had other causes of ongoing anemia (e.g., breakthrough hemolysis or bone marrow failure) ruled out. The group indicated that a clinically meaningful response to treatment would be sustained control of LDH level (i.e., below 1.5 times the ULN), an increase in Hb (or possibly Hb stabilization without further need of transfusion), and improvement in anemia-related symptoms. Treatment discontinuation should be considered in patients who have AEs that preclude ongoing therapy (e.g., recurrent breakthrough hemolysis, issues with effective self-administration, intolerable pain from injection). The group indicated that the treatment and monitoring of patients should ideally be done by clinicians who specialize in the area, although patients can self-administer pegcetacoplan at home or anywhere they prefer.
Drug Program Input
The drug programs provide input on each drug being reviewed through CADTH’s reimbursement review processes by identifying issues that may impact their ability to implement a recommendation. The implementation questions and corresponding responses from the clinical experts consulted by CADTH are summarized in Table 4.
Table 4: Summary of Drug Plan Input and Clinical Expert Response
Drug program implementation questions | Clinical expert response |
|---|---|
Relevant comparators | |
Eculizumab was used as the comparator in the pivotal trial (PEGASUS) and is an appropriate comparator. Although eculizumab is covered by most provincial and federal drug programs, the reimbursement information is not publicly available for most. Another relevant comparator is ravulizumab, which was not used in the PEGASUS trial. At the time of trial initiation, ravulizumab was not available. Likewise, it is still in negotiations and is not yet listed on any jurisdiction’s formulary. | For CDEC consideration. |
Consideration for initiation of therapy | |
As pegcetacoplan is indicated for the second-line treatment of PNH, it is presumed that patients have already met any diagnostic criteria that would have made them eligible for a C5 inhibitor, such as the comparator, eculizumab. 1.Question for clinical experts: Would there be any scenario in which a patient gained access to treatment with a C5 inhibitor without having met drug program criteria? 2.Question for CDEC: In the hypothetical scenario that a patient started C5 inhibitor treatment without going through a drug program’s criteria process, and the patient has intolerance or an inadequate response, would the patient be eligible for consideration of treatment with pegcetacoplan? | 1.The clinical experts noted that some patients who participated in clinical trials of C5 inhibitors continue to access a C5 inhibitor treatment via compassionate access program after completion of the clinical trials. The clinical experts noted the provincial funding criteria differ from the eligibility criteria for clinical trials; therefore, it is possible that these patients would not have met the provincial funding criteria for C5 inhibitors. 2.For CDEC consideration. |
The inclusion criteria for the PEGASUS trial required the patient to have been on a stable dose of eculizumab for at least 3 months before screening. In addition, patients were required to have a Hb < 10.5 g/dL at time of screening, showing ongoing anemia despite C5 inhibitor treatment. As noted in the submission, there is no universally accepted method of determining response to C5 inhibitor treatment. Assessment can include clinical improvement in signs and symptoms of PNH, biochemical evidence of reduced IVH (i.e., LDH < 1.5 × ULN), and improved blood parameters (e.g., Hb). Another inclusion criterion from the PEGASUS trial was that patients (or caregivers) be willing and able to self-administer pegcetacoplan. Questions for clinical experts: 1.Is it sufficient to look at a single test result from 1 point in time when assessing C5 inhibitor treatment response? How likely is it that an inadequate lab value is not accurately reflective of the treatment, but influenced by circumstantial factors at the time of testing? 2.Is 3 months a sufficient duration to see an adequate response to C5 inhibitor treatment if a patient only stabilized on the dose 3 months before enrolling in the study? 3.What other specific indicators of inadequate response would be appropriate to document to support a patient’s need for second-line treatment with pegcetacoplan? 4.In terms of intolerance to C5 inhibitor treatment, what would constitute intolerance that would require a switch to pegcetacoplan, and what documentation would be reasonable to expect in support of this scenario? Questions for CDEC: In the event of a positive recommendation with conditions, would it maintain consistency with the PEGASUS trial inclusion criteria for lab parameters, or would consideration be given to requiring evidence of inadequate lab parameters over time (i.e., a visible decline in Hb over the previous 3-month period)? | 1.The clinical experts noted that a switch from a C5 inhibitor to pegcetacoplan would not be undertaken easily and would not be considered by clinicians unless patients were unsatisfied with treatment or physicians believed that a change would be in favour of the patient’s health. According to the clinical experts, the decision to switch to pegcetacoplan would not be based on a single episode of anemia but on clinical judgment of all patient parameters, chronology of symptoms, and laboratory results to assess the cause of ongoing anemia. The clinical experts noted that hemolysis can be chronic and low grade, or acute in response to infection, stress, pregnancy, or other situations; as such, hemolytic parameters and Hb levels can vary widely. 2.The clinical experts noted although 3 months is a reasonable time frame in which to assess response to C5 inhibitor treatments in patients on a stable dose of C5 inhibitor, most clinicians would not be ready to implement a switch in therapy to pegcetacoplan at 3 months because additional investigations are generally required to rule out other contributors of treatment failure (e.g., bone marrow failure, polymorphism) and ascertain EVH as the cause of treatment failure. To account for the additional turnaround time for these investigational tests, a switch to pegcetacoplan would most likely take place after at least 6 months of C5 inhibitor treatment at a stable dose in clinical practice, as per the clinical experts. 3.The clinical experts commented that the indicators included in the inclusion criteria of the PEGASUS trial were adequate to support a patient’s need for second-line treatment with pegcetacoplan. 4.The clinical experts noted that intolerance to C5 inhibitors is rare in clinical practice, and it most likely manifests as an infusion-related reaction if it does occur. The clinical experts commented that the need for specific supporting documentation for intolerance is unnecessary because the decision to switch from a C5 inhibitor to pegcetacoplan upon intolerance is made at the discretion of the treating physician and will not be made lightly. The clinical experts added that some latitude should be given to allow patients to switch back to a C5 inhibitor in the event that pegcetacoplan is less tolerated by patients compared with the original C5 inhibitor treatment. |
Question for CDEC: Pegcetacoplan is indicated for second-line therapy, with the prior treatment being a C5 inhibitor. If granted a positive recommendation with conditions, would the required duration of C5 inhibitor treatment be consistent with the inclusion criteria from the PEGASUS trial (i.e., a duration greater than or equal to 3 months on a stable dose)? | For CDEC consideration. |
Considerations for continuation or renewal of therapy | |
With regards to assessment of response, the primary end point was change in Hb from baseline to the end of the 16-week randomized controlled period. LDH was monitored, too, and influenced dose modification in 2 patients. Secondary end points included transfusion avoidance, change from baseline to week 16 in ARC, LDH level, and FACIT-Fatigue score. Depending on individual circumstances and location, there may be challenges with frequent blood monitoring and other follow-up factors. | For CDEC consideration. |
To facilitate implementation of a recommendation to reimburse with conditions, consider whether renewal conditions can be aligned with the criteria for eculizumab and ravulizumab. | For CDEC consideration. |
Considerations for discontinuation of therapy | |
The submission noted that Canadian physicians were surveyed and, in patients with an inadequate response to pegcetacoplan, these patients would most likely return to their original C5 inhibitor treatment. Question for clinical experts: 1.Would you agree with this statement; if not, how would you approach this scenario? 2.What would constitute an inadequate response significant enough to discontinue therapy? | The clinical experts noted the decision to continue pegcetacoplan or to return to a patient’s original C5 inhibitor treatment should be determined on a case-by-case basis, and take factors such as treatment tolerance, ease of administration, side effects, and efficacy into consideration, with the goal of maintaining quality of life. The clinical experts noted that treatment discontinuation should be considered in patients with persistent anemia and ongoing transfusion needs despite an adequate trial of pegcetacoplan. |
To facilitate implementation of a recommendation to reimburse with conditions, consider whether discontinuation conditions can be aligned with the criteria for eculizumab and ravulizumab. | For CDEC consideration. |
Considerations for prescribing of therapy | |
Patients should be monitored closely for signs and symptoms of hemolysis, including LDH concentration. If the LDH rises to > 2 × ULN, the dosing regimen may be modified to 1,080 mg every third day (vs. standard dosage of twice weekly). Question for clinical experts: Would you expect the percentage of patients requiring an increase in dosing frequency to align with what was seen in the PEGASUS trial? If not, would it be more or less? Two of 41 patients in the trial (4.9%) required a dose increase to every third day. | The clinical experts noted an increased dosing frequency of pegcetacoplan will likely be required in some patients in the case of hemolysis; however, it is difficult to predict the proportion of patients requiring a dose increase, given the short duration of follow-up and the small sample size in the pivotal trial. |
Question for CDEC: Pegcetacoplan is administered by self-subcutaneous infusion. In the rare circumstance that a patient or caregiver is unwilling or unable to self-administer, would treatment with pegcetacoplan be initiated and, if yes, under what conditions (i.e., where would administration occur and who would cover the cost of this)? | For CDEC consideration. |
Question for CDEC: Given the PSP is expected to train patients on how to self-administer the medication, would there be any anticipated issues anticipated for training patients outside of metro areas? | For CDEC consideration. The clinical experts did not any anticipate issues related to self-administration training for such patients. |
Question for clinical experts: Although not analyzed in the PEGASUS trial, what would the likelihood be of a patient being treated with both a C5 inhibitor and pegcetacoplan due to inadequate response on pegcetacoplan alone? | The clinical experts noted that most hematologists are reluctant to prescribe C5 and C3 inhibitors concurrently and they have limited experience with this treatment approach. |
Question for CDEC: To facilitate implementation of a recommendation to reimburse with conditions, if the diagnostic criteria are to be included, consider whether these conditions can be aligned with the criteria for eculizumab and ravulizumab. | For CDEC consideration. |
Generalizability | |
Question for CDEC: Patients matching the indication but who had certain cardiovascular factors that would potentially confound cardiac safety outcomes were excluded from the trial. Would this patient population be eligible for treatment with pegcetacoplan? | For CDEC consideration. The clinical experts noted that pegcetacoplan treatment could be considered in patients with cardiovascular disease or in those receiving QT-prolonging medications. |
Care provision issues | |
Considerations for CDEC: As pegcetacoplan is self-administered by subcutaneous infusion, there is training involved, and specialized pump supplies. Per the submission, the FreedomEdge pump system will be available at no charge to patients through the PSP. The training will also be via the PSP. If a patient starts on treatment with pegcetacoplan and, over time, is unable or unwilling to continue to self-administer for any reason or have a caregiver do so, how would this be approached in terms of who will administer it, where will it be administered, and who would provide coverage of the administration should it require private services? The submission also noted that there were 14 treatment interruptions during the trial, mainly due to pump malfunction or user error. In similar situations, would the PSP be providing replacement doses? If the FreedomEdge pump system supplies become short-stocked, are there alternative pump systems available that can be used in its place, and would they be provided by the PSP? Switching from a C5 inhibitor to pegcetacoplan requires a 4-week overlap period when the patient receives both pegcetacoplan and their C5 inhibitor for the first 4 weeks. This is to minimize the risk of hemolysis after abrupt discontinuation of C5 inhibitor treatment. As this 4-week period would require the provision of 2 medications, it may have a significant impact on cost, depending on if drug plans reimburse this initial period. | For CDEC consideration. The CADTH review team noted that the costs of both pegcetacoplan and eculizumab in the first 4 weeks of pegcetacoplan initiation were accounted for in the budget impact analysis submitted by the sponsor, but costs related to administration of the drug when a patient or caregiver is unable to perform subcutaneous injection and replacement doses due to pump malfunction were not. It is unclear if alternative pump systems can be used and if they will be provided by the PSP based on the information submitted by the sponsor. |
Question for clinical experts: Although the submission noted most TEAEs were mild or moderate in severity, if a patient develops intolerance to pegcetacoplan, would it be expected that the patient would return to treatment with the previously discontinued C5 inhibitor and hence discontinue the pegcetacoplan? if they had also been intolerant to the C5 inhibitor, would they discontinue all therapy or continue with the pegcetacoplan? | The clinical experts noted that discontinuation of both C3 and C5 inhibitor treatments would be unlikely, and patients would continue with the better tolerated treatment. |
At times, prophylactic antibiotics may be clinically indicated, given the risk of serious infections with encapsulated bacteria. | For CDEC consideration. The CADTH review team noted that the cost of prophylactic antibiotics was not accounted for in the budget impact model, based on the assumption that all patients would be vaccinated against encapsulated bacteria at least 2 weeks before the initiation of pegcetacoplan and would not require treatment with prophylactic antibiotics. |
Although the indication is for inadequate response or intolerance to C5 inhibitors, it could be anticipated that jurisdictions might see requests indicating that infusion clinics and/or hospitals are not readily accessible to a patient, arguing for initiation of treatment with pegcetacoplan in the first line in this situation. Given that treatment with pegcetacoplan will require regular follow-up and blood work, and possibly transfusions, it would be a difficult argument to make against the accessibility to use C5 inhibitor infusions. However, Hypothetically, can CDEC address how this type of request might be approached? | For CDEC consideration. The clinical experts noted that in their experience, access to infusion support can be a challenge for patients receiving eculizumab treatment who live in rural areas (e.g., Northwest Territories). They would prefer prescribing ravulizumab as the first-line treatment for these patients. |
System and economic issues | |
The drug cost is significant and has the potential to create a high budget impact. | For CDEC consideration. |
It’s unclear whether the cost of the required vaccinations will be picked up by the PSP or fall on the drug programs. | For CDEC consideration. The CADTH team noted that vaccination costs were included in budget impact analysis, but not in the pharmacoeconomic model. In the budget impact analysis, the costs of vaccines against Streptococcus pneumoniae and Haemophilus influenzae were accounted for, but the costs of meningococcal vaccines were not, based on the assumption that patients would have received this vaccine before the initiation of eculizumab or ravulizumab treatment. The sponsor noted that the PSP will provide logistical support to assist physicians and patients with the coordination of pretreatment vaccination but will not provide coverage for the cost of pretreatment vaccination. |
For most drug plans, reimbursement information is not publicly available. Likewise, PLAs exist with confidential negotiated prices. Ravulizumab is currently undergoing pCPA negotiations, the outcome of which will impact price comparisons. Both concerns make it difficult to assess budget impact and any potential cost savings. | For CDEC consideration. |
ARC = absolute reticulocyte count; C3 = complement component 3; C5 = complement component 5; CDEC = CADTH Canadian Drug Expert Committee; EVH = extravascular hemolysis; FACIT = Functional Assessment of Chronic Illness Therapy; Hb = hemoglobin; IVH = intravascular hemolysis; LDH = lactate dehydrogenase; pCPA = pan-Canadian Pharmaceutical Alliance; PLA = product listing agreement; PNH = paroxysmal nocturnal hemoglobinuria; PSP = Patient Support Program; TEAE = treatment-emergent adverse event; ULN = upper limit of normal.
Clinical Evidence
The clinical evidence included in the review of pegcetacoplan is presented in 3 sections. The first section, the systematic review, includes the pivotal study provided in the sponsor’s submission to CADTH and Health Canada, as well as studies that were selected according to an a priori protocol. The second section includes indirect evidence from the sponsor and indirect evidence selected from the literature that met the selection criteria specified in the review. The third section includes sponsor-submitted long-term extension studies and additional relevant studies that were considered to address important gaps in the evidence included in the systematic review.
Systematic Review (Pivotal and Protocol-Selected Studies)
Objectives
To perform a systematic review of the beneficial and harmful effects of pegcetacoplan (54 mg/mL) solution for subcutaneous infusion for the treatment of adult patients with PNH who have an inadequate response to, or are intolerant of, a C5 inhibitor.
Methods
Studies selected for inclusion in the systematic review will include pivotal studies provided in the sponsor’s submission to CADTH and Health Canada, as well as those meeting the selection criteria presented in Table 5. Outcomes included in the CADTH review protocol reflect outcomes considered to be important to patients, clinicians, and drug plans.
Of note, the systematic review protocol presented below was established before the granting of a Notice of Compliance from Health Canada.
Table 5: Inclusion Criteria for the Systematic Review
Criteria | Description |
|---|---|
Patient population | Adult patients with PNH who have an inadequate response to, or are intolerant of, a C5 inhibitor Subgroups: disease severity (e.g., PNH clone size, serum LDH, history of thrombotic event) |
Intervention | Pegcetacoplan 1,080 mg subcutaneous infusion twice weekly |
Comparators |
|
Outcomes | Efficacy outcomes:
Harms outcomes:
|
Study design | Published and unpublished phase III and IV RCTs |
AE = adverse event; C5 = complement component 5; HRQoL = health-related quality of life; LDH = lactate dehydrogenase; PNH = paroxysmal nocturnal hemoglobinuria; RCT = randomized controlled trial; SAE = serious adverse event; WDAE = withdrawal due to adverse event.
The literature search for clinical studies was performed by an information specialist using a peer-reviewed search strategy according to the PRESS Peer Review of Electronic Search Strategies checklist.20
Published literature was identified by searching the following bibliographic databases: MEDLINE All (1946–) via Ovid and Embase (1974–) via Ovid. All Ovid searches were run simultaneously as a multifile search. Duplicates were removed using Ovid deduplication for multifile searches, followed by manual deduplication in Endnote. The search strategy comprised both controlled vocabulary, such as the National Library of Medicine’s MeSH (Medical Subject Headings), and keywords. The main search concept was pegcetacoplan. The clinical trials registries searched were the US National Institutes of Health’s clinicaltrials.gov, WHO’s International Clinical Trials Registry Platform (ICTRP) search portal, Health Canada’s Clinical Trials Database, and the European Union Clinical Trials Register.
No filters were applied to limit the retrieval by study type. Retrieval was not limited by publication date or by language. Conference abstracts were excluded from the search results. Refer to Appendix 1 for the detailed search strategies.
The initial search was completed on September 26, 2022. Regular alerts updated the search until the meeting of the CADTH Canadian Drug Expert Committee (CDEC) on January 25, 2023.
Grey literature (literature that is not commercially published) was identified by searching relevant websites from the Grey Matters: A Practical Tool For Searching Health-Related Grey Literature checklist.21 Included in this search were the websites of regulatory agencies (FDA and European Medicines Agency). Google was used to search for additional internet-based materials. Refer to Appendix 1 for more information on the grey literature search strategy. Two CADTH clinical reviewers independently selected studies for inclusion in the review based on titles and abstracts, according to the predetermined protocol. Full-text articles of all citations considered potentially relevant by at least 1 reviewer were acquired. Reviewers independently made the final selection of studies to be included in the review, and differences were resolved through discussion.
Findings From the Literature
One study (PEGASUS)5 was identified from the literature for inclusion in the systematic review (Figure 1). The included study is summarized in Table 6. A list of excluded studies is presented in Appendix 2.
Table 6: Details of the Included Study
Detail | PEGASUS |
|---|---|
Design and population | |
Study design | Phase III, open-label, randomized, active-controlled trial |
Locations | 44 sites in 11 countries: Australia (1), Asia (10), Europe (17), North America (16, including 2 sites in Canada) |
Patient enrolment dates | Start date: June 14, 2018 Completion date: August 13, 2020 |
Randomized (N) | 80 |
Inclusion criteria |
|
Exclusion criteria |
|
Drugs | |
Intervention | Pegcetacoplan 1,080 mg subcutaneous infusion twice weekly |
Comparator(s) | Eculizumab IV infusion (based on dosing established before study entry) |
Duration | |
Phase | |
Screening | 8 weeks |
Run-in | 4 weeks |
Randomized controlled | 16 weeks |
Open-label extension | 32 weeks |
Follow-up | 12 weeks |
Outcomes (randomized controlled phase) | |
Primary end point | Change from baseline at week 16 in Hb level |
Secondary and exploratory end points | Key secondary:
Other secondaryf:
Exploratory:
Safety: AE, SAE, WDAE, deaths, thromboembolic events, laboratory parameters, ECG |
Notes | |
Publications | Hillmen et al. (2021)5 |
AE = adverse event; ARC = absolute reticulocyte count; AV = atrioventricular; C3 = complement component 3; ECG = electrocardiogram; EORTC QLQ-C30 = European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30; FACIT = Functional Assessment of Chronic Illness Therapy; Hb = hemoglobin; LASA = Linear Analogue Scale Assessment; LDH = lactate dehydrogenase; NYHA = New York Heart Association; PNH = paroxysmal nocturnal hemoglobinuria; QTcF = QT interval corrected for heart rate using Fridericia’s formula; RBC = red blood cell; SAE = serious adverse event; ULN = upper limit of normal; WDAE = withdrawal due to adverse event.
Note: Three additional reports were included (FDA report,22 Cella et al. (2022),23 PEGASUS Clinical Study Report [16 weeks]6).
aDiagnosis confirmed with high-sensitivity flow cytometry.
bIncluding N. meningitidis types A, C, W, Y, and B.
cIncluding unexplained syncope or syncope from an uncorrected cardiac etiology.
dIncluding Mobitz type II second-degree AV block, 2:1 AV block, and high-grade AV block.
ePatients with an implanted pacemaker or implantable cardiac defibrillator with backup pacing capabilities were allowed.
fOnly secondary end points pertaining to the randomized controlled period were summarized. Additional secondary end points pertaining to the open-label period, which included change from baseline and change from week 17 to week 48 with respect to Hb level, ARC, LDH level, FACIT-Fatigue Scale score, LASA score, EORTC QLQ-C30 score, as well as the number of packed RBC units transfused during open-label pegcetacoplan.
Source: PEGASUS Clinical Study Report (16 weeks).6
Description of Studies
One study (PEGASUS)5 met the inclusion criteria for the CADTH systematic review. The PEAGSUS trial was a phase III, open-label, randomized, active-controlled trial that aimed to compare the efficacy and safety of pegcetacoplan with eculizumab in adults with PNH who continued to have Hb levels of less than 10.5 g/dL despite treatment with eculizumab (N = 80). The study was conducted at 44 sites in 11 countries (including 2 sites in Canada). It was initiated on June 14, 2018, and completed on August 13, 2020. The study consisted of the following 5 periods:
Screening period (4 weeks) — study eligibility assessment.
Run-in period (week –4 to day –1) — all patients received pegcetacoplan in addition to eculizumab at the established dose to avoid a period of insufficient complement inhibition.
Randomized controlled period (week 1 to week 16) — patients were randomized in a 1:1 ratio using interactive response technology to receive either pegcetacoplan or eculizumab monotherapy. Randomization was stratified by the number of packed RBC transfusions in the previous 12 months (less than 4 versus at least 4), and the platelet count at screening (less than 100,000/mm3 versus at least 100,000/mm3).
Open-label pegcetacoplan period (week 17 to week 48) — All patients received pegcetacoplan monotherapy. For patients who received eculizumab in the randomized controlled period, they first entered a 4-week run-in period during which pegcetacoplan was given in addition to eculizumab to ensure sufficient complement inhibition. They then received pegcetacoplan monotherapy for the remainder of the pegcetacoplan period.
Follow-up period (week 49 to week 60) — Patients continued to be monitored for an additional 12 weeks in the follow-up period unless they opted to transition to an open-label extension study.
Efficacy and safety were assessed in the 16-week randomized controlled period, and the results are summarized in this section. The primary efficacy end point was change from baseline at week 16 in Hb level, and the key secondary efficacy end points included transfusion avoidance, change from baseline at week 16 in ARC, LDH, and FACIT-Fatigue Scale score. Results of the open-label pegcetacoplan period and follow-up period are presented in the Other Relevant Evidence section of this report.
Figure 2: Study Design Schematic of the PEGASUS Trial
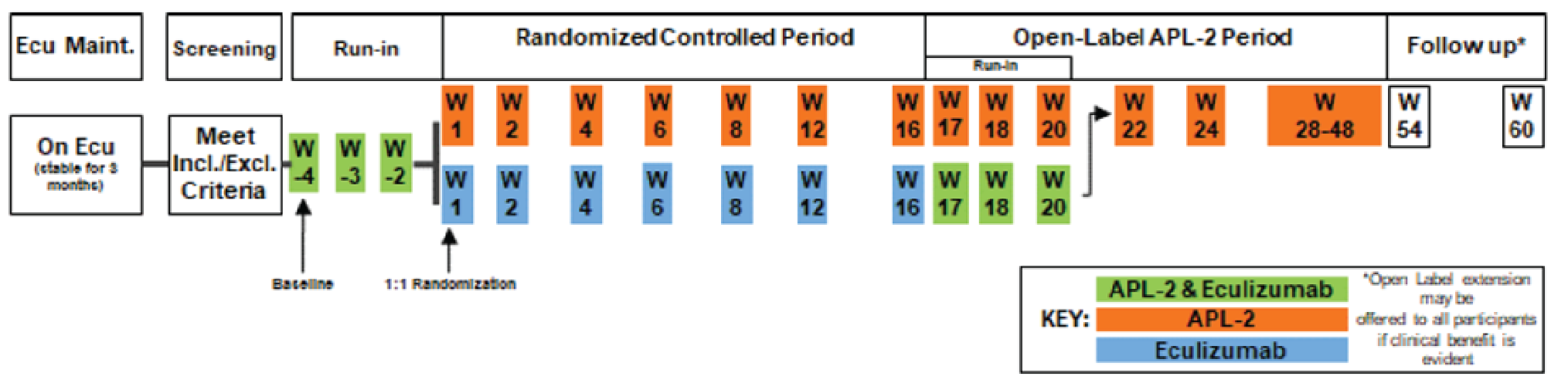
APL-2 = pegcetacoplan; ecu = eculizumab; Excl = exclusion; Incl = inclusion; Maint. = maintenance; W = week.
Source: PEGASUS Clinical Study Report (16 weeks).6
Populations
Inclusion and Exclusion Criteria
The key inclusion and exclusion criteria in the PEGASUS trial are summarized in Table 6. The study enrolled adults with PNH who were on a stable dose of eculizumab for at least 3 months with an Hb level of less than 10.5 g/dL, an ARC level above the ULN, a platelet count above 50,000/mm3, and an absolute neutrophil count above 500/mm3. Patients who had a history of bone marrow transplant, hereditary complement deficiency, or were treated with iron, folic acid, and vitamin B12 at a stable dose for less than 4 weeks were excluded. Patients with certain cardiovascular diseases or who were receiving QT-prolonging medications that, in the sponsor’s opinion, could potentially confound the study’s cardiac safety outcomes were also excluded from the study.
Baseline Characteristics
A summary of baseline patient demographics and disease characteristics is shown in Table 7. The study population had a mean age of 48.8 years (SD = 16.0 years). The majority of patients were female and white (both 61.3%). Patients had a mean time since diagnosis of 10.1 years (SD = 8.6 years) and had received eculizumab for a mean duration of 1,808.7 days (SD = 1,447.6 days). Most patients (70.0%) received eculizumab 900 mg every 2 weeks (i.e., Health Canada–approved maintenance dose), although higher doses (1,200 mg or 1,500 mg every 2 weeks) or more frequent dosing of eculizumab (900 mg every 11 days) were also used. Fifty-five percent of patients had received at least 4 transfusions in the previous 12 months. Patients had a mean Hb level of 8.7 g/dL (SD = 1.0 g/dL), mean LDH level of 282.42 U/L (SD = 211.0 U/L), mean ARC of 216.9 × 109 cells/mL (SD = 71.7 × 109 cells/mL), mean indirect bilirubin level of 33.8 µmol/L (SD = 25.8 µmol/L), and mean haptoglobin level of 0.135 g/L (SD = 0.1206 g/L).
Overall, the baseline demographics and disease characteristics were balanced between treatment arms, except the mean time since diagnosis was shorter in the pegcetacoplan arm (mean = 8.74 years; SD = 7.36 years) than the eculizumab arm (mean = 11.7 years; SD = 9.58 years). The proportion of patients on eculizumab at 900 mg every 2 weeks before the study (63.4%) and the mean LDH levels (257.5 U/L; SD = 97.6 U/L) were lower in the pegcetacoplan arm compared eculizumab arm (76.9% on eculizumab 900 mg every 2 weeks; mean LDH level = 308.6 U/L; SD = 284.8 U/L). A higher proportion of patients had a history of thrombosis in the pegcetacoplan arm (36.6%) than the eculizumab arm (25.6%).
Table 7: Summary of Baseline Characteristics in the PEGASUS Trial — Randomized Controlled Period (ITT)
Baseline characteristic | Pegcetacoplan (N = 41) | Eculizumab (N = 39) |
|---|---|---|
Demographics | ||
Age (years), mean (SD) | 50.2 (16.3) | 47.3 (15.8) |
Sex, n (%) | ||
Female | 27 (65.9) | 22 (56.4) |
Male | 14 (34.1) | 17 (43.6) |
Race, n (%) | ||
White | 24 (58.5) | 25 (64.1) |
Asian | 5 (12.2) | 7 (17.9) |
Black or African American | 2 (4.9) | 0 |
Other | 0 | 1 (2.6) |
Not reported | 10 (24.4) | 6 (15.4) |
Region, n (%) | ||
Europe | 25 (61.0) | 19 (48.7) |
Asia-Pacific | 6 (14.6) | 12 (30.8) |
North America | 10 (24.4) | 8 (20.5) |
Disease characteristicsa | ||
Time since diagnosis (years), mean (SD) | 8.7 (7.4) | 11.7 (9.6) |
Duration of eculizumab (days), mean (SD) | 1,868.3 (1,568.2) | 1,745.9 (1,326.7) |
Current eculizumab dosing, n (%) | ||
900 mg IV every 2 weeks | 26 (63.4) | 30 (76.9) |
1,200 mg IV every 2 weeks | 12 (29.3) | 9 (23.1) |
1,500 mg IV every 2 weeks | 2 (4.9) | 0 |
900 mg IV every 11 days | 1 (2.4) | 0 |
Number of transfusions in the previous 12 months | ||
Mean (SD) | 6.1 (7.3) | 6.9 (7.7) |
< 4 transfusions, n (%) | 20 (48.8) | 16 (41.0) |
0 | 10 (24.4) | 10 (25.6) |
1 | 4 (9.8) | 3 (7.7) |
2 | 3 (7.3) | 3 (7.7) |
3 | 3 (7.3) | 0 |
≥ 4 transfusions, n (%) | 21 (51.2) | 23 (59.0) |
Platelet count at screening (count/mm3), n (%) | ||
< 100,000 | 12 (29.3) | 9 (23.1) |
≥ 100,000 | 29 (70.7) | 30 (76.9) |
Hemoglobin level (g/dL), mean (SD) | 8.7 (1.1) | 8.7 (0.9) |
ARC level (109 cells/mL), mean (SD) | 217.5 (75.0) | 216.2 (69.1) |
LDH level (U/L), mean (SD) | 257.5 (97.6) | 308.6 (284.8) |
Haptoglobin level (g/L), mean (SD) | 0.144 (0.125) | 0.125 (0.116) |
Total bilirubin level (µmol/L), mean (SD) | 42.5 (31.5) | 40.5 (26.6) |
Indirect bilirubin level (µmol/L), mean (SD) | 34.7 (28.5) | 32.9 (23.0) |
Patients with history of ≥ 1 thrombosis, n (%) | 15 (36.6) | 10 (25.6) |
ARC = absolute reticulocyte count; ITT = intention to treat; LDH = lactate dehydrogenase; SD = standard deviation.
aAll baseline disease characteristics were recorded at day –28 (first dose of pegcetacoplan in the run-in period). The mean value for Hb included local and central laboratory values; all other laboratory parameters were based on central laboratory values.
Source: PEGASUS Clinical Study Report (16 weeks).6
Interventions
Pegcetacoplan was dosed at 1,080 mg twice weekly via subcutaneous infusion. A dose increase of pegcetacoplan to 1,080 mg every third day was allowed in patients on pegcetacoplan monotherapy if the LDH level was at least 2 times above the ULN. Eculizumab was administered based on the patient’s dose established before study entry (900 mg, 1,200 mg, or 1,500 mg every 2 weeks, or 900 mg every 11 days) via IV infusion; no dose change of eculizumab was allowed during the study.
No blinding of interventions occurred in the study. Pegcetacoplan was self-administered by patients using a syringe system infusion pump, and infusion training was provided in the run-in period. Eculizumab infusion could be performed at home, at a local infusion centre, or by a local hematologist.
Concomitant medications allowed in the study included antibiotics (prophylaxis and treatment), iron supplements (if initiated before study enrolment and dosage remained stable), iron chelation (if tolerance was previously established), and QT-prolongation drugs (if dosage was stable for at least 3 weeks before enrolment). Phlebotomy or venesection were allowed if Hb was within the normal range. These medications and procedures were prohibited if the predefined criteria were not met. Transfusions were to be given if Hb level was less than 7 g/dL without symptoms or less than 9 g/dL with symptoms. In the event that patients did not meet the criteria but a transfusion was deemed necessary by the investigators, a transfusion could be given but was considered a protocol deviation.
Outcomes
A list of efficacy end points identified in the CADTH review protocol that were assessed in the clinical trials included in this review is provided in Table 8 and summarized subsequently. A detailed discussion and critical appraisal of the outcome measures is provided in Appendix 4.
Table 8: Summary of Outcomes of Interest Identified in the CADTH Review Protocol
Outcome measure | PEGASUS | End points measured in PEGASUS (randomized controlled period) |
|---|---|---|
Survival | Not measured as an efficacy end point |
|
Hb level | Primary |
|
Secondary |
| |
Transfusions | Key secondary |
|
Secondary |
| |
Thrombotic events | Not measured as an efficacy end point |
|
Symptoms of PNH | Key secondary |
|
HRQoL | Secondary |
|
Secondary |
| |
Breakthrough hemolysis events | Not measured as an efficacy end point |
|
Complications of PNH other than thrombotic events | Not measured as an efficacy end point |
|
Intravascular hemolysis | Key secondary |
|
Extravascular hemolysis | Key secondary |
|
Secondary |
| |
Health care resource utilization | Not measured | Not measured |
ARC = absolute reticulocyte count; EORTC QLQ-C30 = European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30; FACIT = Functional Assessment of Chronic Illness Therapy; Hb = hemoglobin; HRQoL = health-related quality of life; LASA = Linear Analogue Scale Assessment; LDH = lactate dehydrogenase; PNH = paroxysmal nocturnal hemoglobinuria; RBC = red blood cell.
Source: PEGASUS Clinical Study Report (16 weeks).6
Efficacy Outcomes
Unless otherwise specified, baseline was defined as the average of measurements recorded before the first dose of the investigational product (i.e., before the start of the run-in period [week –4]) and assessments were conducted at all study visits during the run-in period (i.e., weeks –4, –3, and –2), and the randomized controlled period (i.e., weeks 1, 2, 4, 6, 8, 12, and 16).
Hb Level
Hb level was assessed at each study visit based on samples collected before study drug administration and analyzed in a central laboratory. The change from baseline at week 16 in Hb level was the primary efficacy end point. Secondary end points related to Hb level included Hb response in the absence of transfusions at week 16 (with response defined as an increase of at least 1 g/dL in Hb from baseline) and Hb normalization in the absence of transfusions at week 16 (with normalization defined as the Hb level being above the lower limit of the normal range [i.e., 12 g/dL in females and 13.6 g/dL in males]).
Hb level is used in the diagnosis and monitoring of disease activity of PNH,16 and low levels of Hb indicate anemia. The validity, reliability, responsiveness, or MID estimates of these parameters have not been studied in patients with PNH. It has been proposed that Hb level is a strong surrogate biomarker for clinical benefits in patients with sickle cell disease receiving stem cell or gene therapy.24,25 It has also been proposed that an increase in Hb of 1 g/dL from baseline indicates disease improvement in patients with sickle cell disease.24,25
Transfusions
Transfusion avoidance, a key secondary efficacy end point, was defined as the proportion of patients who did not require a transfusion during the randomized controlled period. Patients were to receive a transfusion if Hb level was less than 7 g/dL without symptoms or less than 9 g/dL with symptoms, if deemed necessary by the investigators. The number of packed RBC units transfused was assessed as a secondary efficacy end point.
Symptoms of PNH
The change from baseline at week 16 in FACIT-Fatigue Scale score was a key secondary efficacy end point. The instrument was administered at each study visit, except at week –3.
The FACIT-Fatigue Scale is a 13-item, patient-reported, fatigue-specific, QoL questionnaire that uses a 5-point Likert scale. It assesses tiredness, weakness, and difficulty conducting usual activities as a result of fatigue over the previous week. The total score ranges from 0 (extreme fatigue) to 52 (no fatigue). A study demonstrated content validity of this instrument in 29 treatment-naive patients with PNH receiving a C5 inhibitor in the UK, US, France, and Spain.26 However, evidence for other types of validity, reliability, and responsiveness have not been previously assessed in this patient population. A study established a clinically important difference of 5 points in treatment-naive patients with PNH receiving eculizumab.27
HRQoL or QoL
The change from baseline at week 16 in LASA score and EORTC QLQ C-30 were secondary efficacy end points. The instruments were administered at each visit, except at week –3.
The LASA used in the trial was a 3-item, self-administered questionnaire that assessed QoL; it appeared to be an adaptation of the LASA reported by Locke et al. (2007).28 More details on the original version of the LASA and its properties can be found in Table 41 in Appendix 4. The version used in this study included 3 visual analogue scales that assess energy level, activities of daily living, and overall QoL over the previous week. Each scale employed a 0 to 100 scale, and a combined score (range, 0 to 300) was computed. A higher score indicates higher functioning. The validity, reliability, responsiveness, and MID estimates of this instrument have not been previously studied in patients with PNH.
The EORTC QLQ-C30, a 30-item, patient-reported, cancer-specific questionnaire that evaluates HRQoL in patients by assessing 3 domains: function, symptom, and global health status/QoL. There are subscales in the function domain (physical, role, emotional, cognitive, and social functioning) and the symptom domain (dyspnea, insomnia, appetite loss, constipation, diarrhea, and financial impact), and outcome score for each scale is computed using a linear transformation of the raw score, such that scores range from 0 to 100. A higher score for the function and symptom scales represents a higher (better) level of functioning and a higher (worse) level of symptoms, respectively. A higher score for global health status/QoL represents a better level of global health status. A study demonstrated content validity of this instrument in 29 treatment-naive-patients with PNH who received a C5 inhibitor in the UK, US, France, and Spain;26 however, evidence for other validity measures, reliability, responsiveness, and MID estimates have not been established in this patient population.
Intravascular Hemolysis
Serum LDH was assessed at each study visit based on samples collected before study drug administration and analyzed in a central laboratory. The change from baseline at week 16 in LDH level was a key secondary efficacy end point.
LDH is a marker of IVH29 and is used in the diagnosis and monitoring of disease activity of PNH.16 It has been reported that an LDH level of at least 1.5 times above the ULN is associated with 4.8-fold increase in risk of mortality30 and an increased risk of thromboembolism (odds ratio = 7.0; P = 0.013)31 based on data from a national South Korean PNH registry that includes patients who are eculizumab-naive (N = 301).
Extravascular Hemolysis
The change from baseline at week 16 in ARC was a key secondary efficacy end point. Reticulocyte normalization in the absence of transfusions at week 16 was a secondary efficacy end point, with normalization defined as a reticulocyte count being below the ULN (i.e., 120 × 109 cells/L). The change from baseline at week 16 in indirect bilirubin and haptoglobin levels were secondary end points. Reticulocyte count and indirect bilirubin level were assessed at each study visit, whereas haptoglobin was assessed at weeks –4, 1, 4, 8, 12, and 16. The samples were collected before study drug administration and were analyzed in a central laboratory.
Reticulocyte, indirect bilirubin, and haptoglobin are hemolytic markers used in the diagnosis and monitoring PNH disease activity.16 Elevated reticulocyte count is an indicator of marrow compensatory response that produces RBCs in the presence of hemolysis.29 Bilirubin is produced from the breakdown of Hb and is usually elevated in hemolysis.29 Haptoglobin functions as a scavenger that binds free Hb from lysed RBCs, and reduction in haptoglobin level is a marker of hemolysis.32 The validity, reliability, responsiveness, and MID estimates of these measures have not been studied in patients with PNH.
Harms Outcomes
Safety assessments were conducted at each study visit to assess TEAEs, serious TEAEs, withdrawal due to AEs, laboratory parameters, and electrocardiogram. Survival (death events), thrombotic events, hemolysis, and renal failure are reported in the Harms section.
Breakthrough hemolysis was defined as at least 1 of the following in the presence of LDH elevation to at least 2 times the ULN after prior LDH reduction to below 1.5 times the ULN on therapy: a new or worsening symptom or sign of IVH (fatigue, hemoglobinuria, abdominal pain, dyspnea); anemia (Hb less than 10 g/dL); or major adverse vascular events, including thrombosis, dysphagia, or erectile dysfunction).
Statistical Analysis
A summary of the statistical analysis of efficacy outcomes in the PEGASUS trial is shown in Table 9.
Sample Size and Power Calculations
A sample size calculation determined that 64 patients (32 per treatment arm) was required to demonstrate a statistically significant difference between pegcetacoplan and eculizumab with respect to the change from baseline in Hb level at week 16 (primary end point) at a 2-sided significance level of 0.05 with a power of 90%, assuming a treatment difference between pegcetacoplan and eculizumab of 1 g/dL and a SD for the change from baseline of 1.2 g/dL. The study aimed to enrol at least 70 patients to account for loss of power due to discontinuations.
Table 9: Statistical Analysis of Efficacy End Points
End point | Statistical model | Adjustment factors | Additional analyses |
|---|---|---|---|
Change from baseline at week 16 in Hb level (ITT set) |
| Treatment group, study visit, stratification factors,a study visit by treatment group interaction, baseline Hb level (continuous) | Sensitivity analyses:
Supportive analyses:
|
Hb response in the absence of transfusions at week 16 (ITT) Hb normalization in the absence of transfusions at week 16 (ITT) Reticulocyte normalization in the absence of transfusions at week 16 (ITT) |
| Stratification factorsa | Supportive analysis (mITT) |
Transfusion avoidance (ITT) |
| Stratification factorsa | Supportive analyses (PP, mITT) |
Number of packed RBC units transfused (ITT) |
| Not applicable | Supportive analyses:
|
Change from baseline at week 16 in FACIT-Fatigue Scale score (ITT) Change from baseline at week 16 in LDH level (ITT) Change from baseline at week 16 in ARC (ITT) |
| Treatment group, study visit, stratification factors,a study visit by treatment group interaction, baseline value (continuous) | Supportive analyses (PP, mITT, uncensored for transfusion) |
Change from baseline at week 16 in LASA score (ITT) Change from baseline at week 16 in EORTC QLQ-C30 score (ITT) Change from baseline at week 16 in indirect bilirubin level (ITT) Change from baseline at week 16 in haptoglobin level (ITT) |
| Treatment group, study visit, stratification factors,a study visit by treatment group interaction, baseline value (continuous) | Supportive analysis (mITT) |
ANCOVA = analysis of covariance; ARC = absolute reticulocyte count; CI = confidence interval; CMH = Cochran-Mantel-Haenszel; EORTC QLQ-C30 = European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30; FACIT = Functional Assessment of Chronic Illness Therapy; Hb = hemoglobin; ITT = intention to treat; LASA = Linear Analogue Scale Assessment; LDH = lactate dehydrogenase; mITT = modified intention to treat; MMRM = mixed model for repeated measures; PP = per protocol; RBC = red blood cell.
aStratification factors: number of packed RBC transfusions in the previous 12 months (< 4 vs. ≥ 4), and platelet count at screening (< 100,000/mm3 vs. ≥ 100,000/mm3).
Source: PEGASUS Statistical Analysis Plan (16 weeks).33
Statistical Analysis for Primary Efficacy End Point
Statistical Model
In the primary analysis, the difference in the mean change from baseline in Hb level at week 16 between treatment arms in the ITT set was tested at a 2-sided significance level of 0.05 using a mixed model for repeated measures (MMRM). The model was stratified by fixed categorical effects, including treatment group, study visit, stratification factors (number of packed RBC transfusions in the previous 12 months [less than 4 versus at least 4], platelet count at screening [less than 100,000/mm3 versus at least 100,000/mm3]), study visit by treatment group interaction, and continuous fixed covariate of Hb level, assuming an unstructured covariance matrix.
Handling of Intercurrent Events and Missing Data
Transfusions, discontinuation of study treatment, and withdrawal from the study during the randomized controlled period were considered intercurrent events in the study. Transfusions or withdrawals from the study were handled using a while-on-treatment strategy in which all measurements after the intercurrent events were censored. Discontinuations of study treatment were handled using a treatment-policy strategy in which all observed values were used regardless of the occurrence of intercurrent events. Missing data were implicitly imputed by the MMRM based on the assumption that data were missing at random.
Subgroup, Sensitivity, and Supportive Analyses
The following prespecified subgroup analyses were conducted for the primary end point: the number of packed RBC transfusions received in the previous 12 months, platelet count at screening, sex, and race; however, none of these analyses was of interest to this review, according to the systematic review protocol.
Two prespecified sensitivity analyses were conducted to examine departures from assumptions regarding missing data, including the control-based pattern imputation method and a tipping point analysis, both of which assume missingness not at random.
Five prespecified supportive analyses were conducted: PP set; modified ITT (mITT) set; completer set; ITT set using all available data (regardless of whether the Hb level was measured after a transfusion); and nonparametric randomization-based analysis of covariance in the ITT set. Refer to the Analysis Populations section for population definitions.
Statistical Analysis for Secondary Efficacy End Points
Multiplicity Adjustment
If the primary end point was met, the key secondary end points were tested for noninferiority using a closed testing procedure in the following hierarchical order: transfusion avoidance, change from baseline at week 16 in ARC, change from baseline at week 16 in LDH level, and change from baseline at week 16 in FACIT-Fatigue score. It was preplanned that if all key secondary end points demonstrated noninferiority, superiority was then assessed for all key secondary end points, in the same hierarchical order, using a closed testing procedure at a 2-sided significance level of 0.05. No multiplicity adjustment was performed for any other secondary end points.
Noninferiority Margin
Pegcetacoplan was considered noninferior to eculizumab in a key secondary end point when the following criteria were met:
Transfusion avoidance — the lower bound of the 95% CI was greater than the NIM of −0.2 (i.e., –20%)
ARC — the upper bound of the 95% CI for the treatment difference was less than the NIM of 10 × 109 cells
LDH — the upper bound of the 95% CI for the treatment difference was less than the NIM of 20 U/L
FACIT-Fatigue score — the lower bound of the 95% CI for the treatment difference was greater than the NIM of –3 points.
Statistical Model and Test
Continuous secondary end points (i.e., ARC, LDH, FACIT-Fatigue score, EORTC QLQ-C30 score, LASA score, indirect bilirubin, and haptoglobin) were analyzed in the ITT set using the same statistical approach as in the primary analysis of the primary end point, except using their own baseline value as a covariate. For categorical secondary end points (i.e., transfusion avoidance, Hb response, Hb normalization, and reticulocyte normalization), the primary analysis was performed in the ITT population. The risk difference between treatments and the respective 95% CI were constructed using the stratified Miettinen-Nurminen method.
The methods of handling intercurrent events and missing data were the same as in the primary analysis of the primary end point. Of note, in the transfusion avoidance analysis, patients who did not have a transfusion but withdrew from the study were considered to have had a transfusion.
For all key secondary end points, supportive analyses (PP, mITT sets) and an additional analysis based on data uncensored for transfusion were conducted. For other secondary end points, a supportive analysis in the mITT set was performed.
The number of packed RBC units transfused in the ITT set was summarized using descriptive statistics, and 2 supporting analyses were performed to account for subjects who withdrew during the randomized controlled period before week 16. The first supportive analysis assessed the end point using log-incidence density ratios (nonparametric) adjusted for treatment. The second supportive analysis estimated the number of units of packed RBCs based on the time the patients were in the randomized controlled period (i.e., number per week multiplied by duration of end point).
Analysis Populations
ITT set: included all randomized patients and was analyzed by treatment assignment.
mITT set: included all randomized patients who received at least 1 dose of monotherapy study drug beyond week 4 of the randomized controlled period and was analyzed by treatment assignment.
PP set: included all randomized patients who did not violate any inclusion or exclusion criteria and/or deviate from the protocol in a way that could influence efficacy assessment. Protocol deviations and their classifications were subject to an independent blinded review before database lock. The analysis was based on the treatment assignment.
Completer set: included all randomized patients who had completed the week-16 efficacy assessment and was analyzed by treatment assignment.
Safety set: included all randomized patients who received at least 1 dose of monotherapy study drug and the analysis was based upon the actual treatment received.
Results
Patient Disposition
A summary of patient disposition in the randomized controlled period of the PEGASUS trial is shown in Table 10. Of 102 screened patients, 21 (19.6%) did not meet the eligibility criteria and did not move past the screening stage and 1 patient met the eligibility criteria but did not enter any additional study periods. It is unclear which eligibility criteria contributed to the screen failure. The ITT set, mITT set, and safety set included 80 patients who were randomized to the pegcetacoplan arm (n = 41) and the eculizumab arm (n = 39) in the randomized controlled period. In the pegcetacoplan arm, 3 (7.3%) patients were discontinued from study treatment due to an AE, 1 of whom (2.4%) was also discontinued from the study. No patient was discontinued from study treatment or the study in the eculizumab arm.
Nine patients were excluded from the PP set (5 in the pegcetacoplan arm and 4 in the eculizumab arm) due to major protocol deviations (1 in the pegcetacoplan arm due to a violation of the inclusion criterion related to ARC, 1 in the pegcetacoplan arm due to missing week-16 visit, and 2 in the eculizumab arm due to receiving a transfusion despite Hb being above 9 g/dL), laboratory discrepancies between the local and central labs (2 in the eculizumab arm), or study drug discontinuation before week 16 (3 in the pegcetacoplan arm).
Table 10: Patient Disposition in the PEGASUS Trial — Randomized Controlled Period
Patient disposition | Pegcetacoplan | Eculizumab |
|---|---|---|
Screened, n | 102 | |
Randomized, n | 41 | 39 |
Discontinued from study, n (%) | 1 (2.4) | 0 |
Adverse event | 1 (2.4) | 0 |
Discontinued from study treatment, n (%) | 3 (7.3) | 0 |
Adverse event | 3 (7.3) | 0 |
ITT, n (%) | 41 (100) | 39 (100) |
mITT, n (%) | 41 (100) | 39 (100) |
PP, n (%) | 36 (87.8) | 35 (89.7) |
Completer set, n (%) | 37 (90.2) | 38 (97.4) |
Safety, n (%) | 41 (100) | 39 (100) |
ITT = intention to treat; mITT = modified intention to treat; PP = per protocol.
Source: PEGASUS Clinical Study Report (16 weeks).6
Major Protocol Deviations
Major protocol deviations are summarized in Table 11. Major protocol deviation was reported in 73.8% of patients in the pegcetacoplan arm and 76.9% of patients in the eculizumab arm. Most major protocol deviations were related to study assessment or schedule noncompliance in both treatment arms (pegcetacoplan: 48.8%; eculizumab: 64.1%). Reasons for these deviations included missed timing for laboratory or vital signs collection, missed visits, missed vaccinations, and assessments not completed by the patient.
Major protocol deviations related to informed consent (16.3%), eligibility criteria (7.5%), prohibited concomitant medication (6.3%), study drug noncompliance (6.4%), and safety reporting (2.5%) were also reported, although no notable imbalance was identified between the treatment arms for any of these categories. The 3 patients (7.7%) with major protocol deviations due to nonadherence to protocol-specified criteria for packed RBC transfusion were all from the eculizumab arm.
One study site was closed due to noncompliance to good clinical practice standards, and patients from this site (n = 2) were not excluded in neither the ITT nor the PP set.
Exposure to Study Treatments
The mean duration of treatment in the randomized controlled period was 104.9 days (SD = 17.96 days) in the pegcetacoplan arm and 98.4 days (SD = 4.89 days) in the eculizumab arm. In the pegcetacoplan arm, 5 (12.2%) patients had an interruption in at least 1 infusion, and the mean number of infusions completed per patient was 30.4 (SD = 5.13). Dose escalation of pegcetacoplan (to 1,080 mg every third day) was required in 2 patients (4.9%). The mean treatment compliance rate was 99.9% (SD = 1.8%) in the pegcetacoplan arm and 100% (SD = 0.0) in the eculizumab arm. Treatment exposure in the randomized controlled period is summarized in Table 12.
Table 11: Major Protocol Deviations — Randomized Controlled Period (ITT)
Major protocol deviation category | Pegcetacoplan (N = 41) | Eculizumab (N = 39) |
|---|---|---|
Patient with at least 1 major protocol deviation, n (%) | 29 (70.7) | 30 (76.9) |
Study assessment or schedule noncompliance | 20 (48.8) | 25 (64.1) |
Informed consent | 7 (17.1) | 6 (15.4) |
Enrolment and inclusion or exclusion criteria | 4 (9.8) | 2 (5.1) |
Prohibited concomitant medication | 2 (4.9) | 3 (7.7) |
Study drug noncompliance | 3 (7.3) | 2 (5.1) |
Safety reporting | 0 | 2 (5.1) |
ITT = intention to treat.
Source: PEGASUS Clinical Study Report (16 weeks).6
Table 12: Treatment Exposure in the PEGASUS Trial — Randomized Controlled Period (Safety Set)
Category or analysis set | Pegcetacoplan (N = 41) | Eculizumab (N = 39) |
|---|---|---|
Total dose administered (mg), mean (SD) | 32,874.1 (5,596.49) | 7,792.3 (1,123.52) |
Duration of treatment in the randomized controlled period (days), mean (SD) | 104.9 (17.96) | 98.4 (4.89) |
Patients with interruption in ≥ 1 pegcetacoplan infusion, n (%) | 5 (12.2) | NA |
Number of infusions completed per patient, mean (SD) | 30.4 (5.13) | NA |
Dose escalation required, n (%) | 2 (4.9) | NA |
Treatment compliancea (%) | ||
Mean (SD) | 99.9 (1.8) | 100.0 (0.0) |
> 90% to ≤ 100%, n (%) | 40 (97.6) | 39 (100.0) |
> 100%, n (%) | 1 (2.4) | 0 |
NA = not applicable (infusion for eculizumab was not evaluated as to whether it was completed or not); SD = standard deviation.
aCompliance rate = total number of study infusions taken from day 1 to end of randomized controlled period (week 16) / total number of expected infusions to end of the randomized controlled period, where total numbers of expected infusions are calculated as treatment duration (days) / 7 × 2.5 for pegcetacoplan; treatment duration (days) / 7 × 2 for eculizumab.
Source: PEGASUS Clinical Study Report (16 weeks).6
Efficacy
Only those efficacy outcomes identified in the review protocol are reported here. Refer to Appendix 3 for detailed efficacy data.
Survival
Survival was not measured as an efficacy end point in this study. Refer to the Harms section for analysis on death events.
Hb Level
Hb outcomes in the ITT set (primary analysis) are summarized in Table 13.
Change From Baseline in Hb Level
A plot of LS mean change from baseline in Hb over time (censored for transfusion) in the randomized controlled period (ITT set) is shown in Figure 3. In the primary analysis, the LS mean change from baseline at week 16 in Hb level (primary end point) was 2.37 g/dL (SE = 0.36 g/dL) in the pegcetacoplan arm and –1.47 g/dL (SE = 0.67 g/dL) in the eculizumab arm, with a between-group difference in LS means of 3.84 g/dL (95% CI, 2.33 to 5.34 g/dL) in favour of pegcetacoplan (P < 0.0001). Results of the sensitivity analyses (controlled-based pattern imputation and tipping point analyses) and supportive analysis using all patient data (uncensored for transfusion; Table 39 in Appendix 3) were also consistent with the primary analysis.
Hb Response and Normalization in the Absence of Transfusion
Hb response (i.e., an increase of at least 1 g/dL from baseline) and Hb normalization in the absence of transfusion at week 16 were secondary end points. Hb response in the absence of transfusion was achieved in 75.6% of patients in the pegcetacoplan arm and by no patients in the eculizumab arm, with an adjusted risk difference of 67.5% (95% CI, 54.5% to 80.4%). Hb normalization in the absence of transfusion was achieved in 34.1% of patients in the pegcetacoplan arm and 0% of patients in the eculizumab arm, with an adjusted risk difference of 30.4% (95% CI, 14.9% to 45.9%). Results based on data uncensored for transfusion were the same (Table 39).
Table 13: Hb Outcomes in the PEGASUS Trial — Randomized Controlled Period (ITT; Censored for Transfusion)
Hb outcomes | Pegcetacoplan (N = 41) | Eculizumab (N = 39) |
|---|---|---|
Baselinea | ||
n (%) | 41 (100) | 39 (100) |
Hb level (g/dL), mean (SD) | 8.69 (1.08) | 8.68 (0.89) |
Week 16 | ||
n (%) | 36 (87.8) | 6 (15.4) |
Hb level (g/dL), mean (SD) | 11.65 (1.89) | 9.27 (0.84) |
Change from baseline at week 16 in Hb levela,b (g/dL), LS mean (SE) | 2.37 (0.36) | –1.47 (0.67) |
Difference in LS meansb (g/dL), (95% CI) | 3.84 (2.33 to 5.34) | Reference |
P valuec | < 0.0001 | Reference |
Patients achieving Hb response in the absence of transfusions at week 16,d n (%) | 31 (75.6) | 0 |
Risk differencee (%), (95% CI) | 67.5 (54.5 to 80.4) | Reference |
Patients achieving Hb normalization in the absence of transfusions at week 16,f n (%) | 14 (34.1) | 0 |
Risk difference (%)e (95% CI) | 30.4 (14.9 to 45.9) | Reference |
CI = confidence interval; Hb = hemoglobin; ITT = intention to treat; LS = least squares; SD = standard deviation; SE = standard error.
aBaseline was defined as the average of measurements recorded before the first dose of pegcetacoplan.
bThe analysis was conducted using a MMRM, with change from baseline at week 16 in Hb level as the dependent variable. The model was adjusted for treatment group, study visit, number of packed RBC transfusions in the previous 12 months (< 4 vs. ≥ 4), platelet count at screening (< 100,000/mm3 vs. ≥ 100,000/mm3), the study visit by treatment group interaction, and baseline Hb level (continuous).
cThe significance threshold was set at a 2-sided alpha of 0.05.
dHb response was defined as an increase of at least 1 g/dL in Hb from baseline, excluding data before the Randomized Controlled Period.
eThe risk difference and 95% CI were constructed using the stratified Miettinen-Nurminen method. This end point was not included in the statistical testing hierarchy.
fHb normalization was defined as a Hb level at or above the lower limit of the normal range.
Source: PEGASUS Clinical Study Report (16 weeks).6
Figure 3: LS Mean Change From Baseline in Hb Over Time in the PEGASUS Trial — Randomized Controlled Period (ITT; Censored for Transfusion)
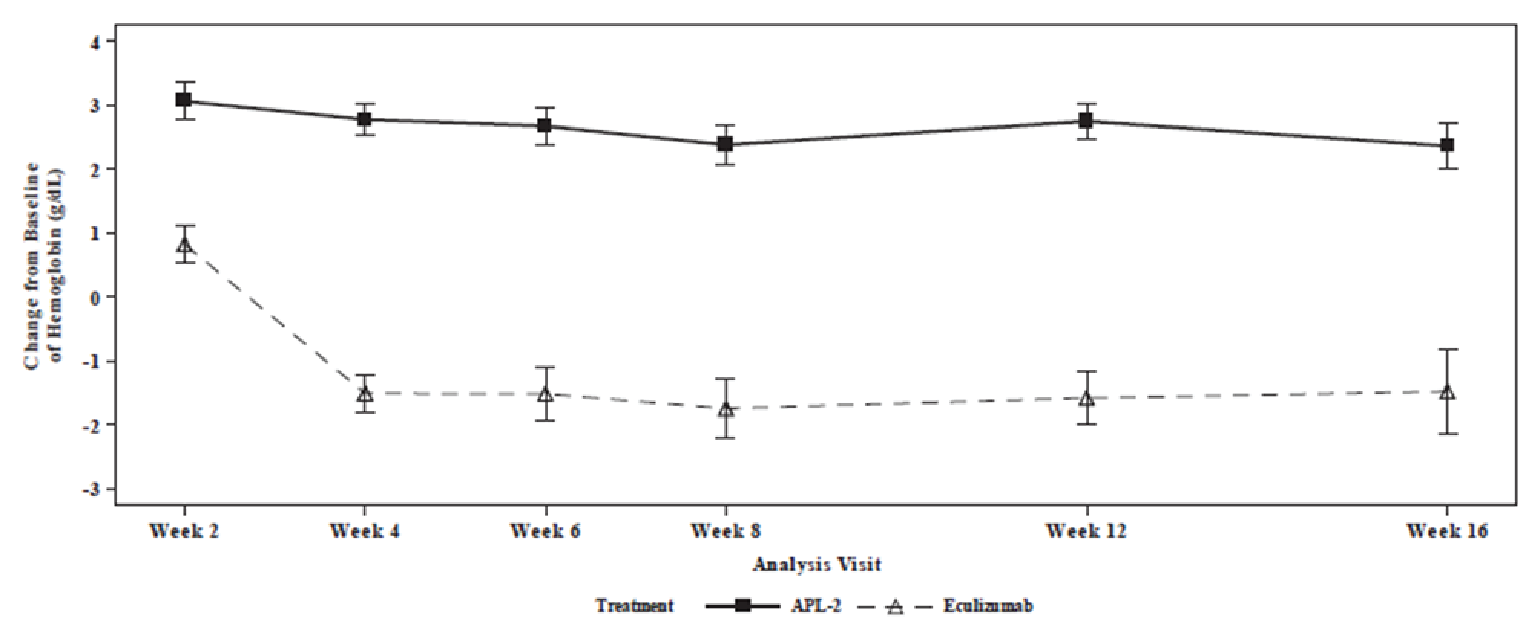
APL-2 = pegcetacoplan; Hb = hemoglobin; ITT = intention to treat; LS = least squares.
Note: Baseline was defined as the average of measurements recorded before the first dose of pegcetacoplan.
Source: PEGASUS Clinical Study Report (16 weeks).6
Transfusion
Transfusion avoidance and the number of packed RBC units transfused analyses in the ITT set (primary analysis) are summarized in Table 14.
Transfusion Avoidance
Transfusion avoidance (i.e., the proportion of patients who did not require a transfusion) during the randomized controlled period was a key secondary end point and is summarized in Table 14. The end point was tested for noninferiority according to the hierarchical testing procedure. In the primary analysis (ITT), the adjusted risk difference in transfusion avoidance between the pegcetacoplan and eculizumab arms was 62.5% (95% CI, 48.3% to 76.8%). Results of the supportive analyses were consistent with the primary analysis (PP analysis shown in Table 38 in Appendix 3). The lower bound of the 95% CI of risk difference was greater than the NIM of –20% in both ITT and PP analysis sets, supporting noninferiority between treatment groups in transfusion avoidance.
Number of Packed RBC Units Transfused
As summarized in Table 14, the mean number of packed RBC units transfused during the randomized controlled period (secondary end point) was 0.6 units (SD = 2.03 units) in the pegcetacoplan arm and 5.1 units (SD = 5.6 units) in the eculizumab arm in both the primary ITT analysis and the supportive mITT analysis.
Table 14: Tranfusion Outcomes in the PEGASUS Trial — Randomized Controlled Period (ITT)
Transfusion outcomes | Pegcetacoplan (N = 41) | Eculizumab (N = 39) |
|---|---|---|
Patients achieving transfusion avoidance,a,b n (%) | 35 (85.4) | 6 (15.4) |
Risk differencec (%), (95% CI) | 62.5 (48.3 to 76.8) | Reference |
P valued | < 0.0001 | Reference |
Patients receiving transfusion,b n (%) | 6 (14.6) | 33 (84.6) |
Received at least 1 transfusion | 5 (12.2) | 33 (84.6) |
Withdrew from the study without having had a transfusion | 1 (2.4) | 0 |
Number of packed RBC units transfused (units), mean (SD) | 0.6 (2.03) | 5.1 (5.60) |
CI = confidence interval; ITT = intention to treat; RBC = red blood cell; SD = standard deviation.
aTransfusion avoidance was defined as the proportion of patients who did not require a transfusion during the randomized controlled period.
bPatients who did not have a transfusion but withdrew before week 16 were considered to have had a transfusion.
cThe risk difference and 95% CI were constructed using the stratified Miettinen-Nurminen method.
dThe P value was based on Cochran-Mantel-Haenszel chi-square test stratified by the number of packed RBC transfusions in the previous 12 months (< 4 vs. ≥ 4) and the platelet count at screening (< 100,000/mm3 vs. ≥ 100,000/mm3). In accordance with the hierarchal testing procedure, noninferiority but not superiority testing was conducted.
Source: PEGASUS Clinical Study Report (16 weeks).6
Thrombotic Event
Thrombotic event was not measured as an efficacy end point in this study. Refer to the Harms section for analysis on thrombotic events.
Symptoms of PNH
FACIT-Fatigue Scale
Change from baseline at week 16 in FACIT-Fatigue score was a key secondary end point. A higher FACIT-Fatigue score indicates less fatigue. As shown in Table 15, the LS mean change from baseline at week 16 in FACIT-Fatigue score in the ITT set (censored for transfusion) was 9.22 points (SE = 1.61 points) in the pegcetacoplan arm and –2.65 points (SE = 2.82 points) in the eculizumab arm, with a between-group difference in LS mean of 11.87 points (95% CI, 5.49 to 18.25 points). Results of the supportive analyses were consistent with the primary ITT analysis (PP analysis shown in Table 38; analysis based on data uncensored for transfusion in Table 39 in Appendix 3).
An additional responder analysis was performed to assess the proportion of patients with at least a 3-point increase in FACIT-Fatigue score from baseline at week 16 (censored for transfusion), and the proportion was 73.2% in the pegcetacoplan arm and 0% in the eculizumab arm.
Table 15: FACIT-Fatigue Score Outcomes in the PEGASUS Trial — Randomized Controlled Period (ITT; Censored for Transfusion)
FACIT-Fatigue Scale | Pegcetacoplan (N = 41) | Eculizumab (N = 39) |
|---|---|---|
Baselinea | ||
n (%) | 41 (100.0) | 38 (97.4) |
FACIT-Fatigue score, mean (SD) | 32.16 (11.38) | 31.55 (12.51) |
Week 16 | ||
n (%) | 35 (85.4) | 6 (15.4) |
FACIT-Fatigue score, mean (SD) | 42.49 (8.83) | 34.67 (16.35) |
Change from baseline at week 16 in FACIT-Fatigue score,a LS mean (SE) | 9.22 (1.61) | –2.65 (2.82) |
Difference in LS means (points),b (95% CI) | 11.87 (5.49 to 18.25) | Reference |
P valuec | 0.0005 | Reference |
Patient with ≥ 3-point increase in FACIT-Fatigue score from baseline at week 16, n (%) | 30 (73.2) | 0 |
CI = confidence interval; FACIT = Functional Assessment of Chronic Illness Therapy; ITT = intention to treat; LS = least square; SD = standard deviation; SE = standard error.
aBaseline was defined as the last available nonmissing observation before the first dose of pegcetacoplan.
bThe analysis was conducted using a MMRM, with change from baseline at week 16 in FACIT-Fatigue score as the dependent variable. The model was adjusted for treatment group, study visit, number of packed RBC transfusions in the previous 12 months (< 4 vs. ≥ 4), platelet count at screening (< 100,000/mm3 vs. ≥ 100,000/mm3), the study visit by treatment group interaction, and baseline FACIT-Fatigue score (continuous).
cIn accordance with the hierarchal testing procedure, noninferiority and superiority testing was not conducted.
Source: PEGASUS Clinical Study Report (16 weeks).6
HRQoL or QoL
LASA
As shown in Table 16, the LS mean change from baseline at week 16 in LASA score (secondary end point, censored for transfusion) was 49.4 points (SE = 10.2 points) in the pegcetacoplan arm and –9.7 points (SE = 19.0 points) in the eculizumab arm, with a between-group difference of 59.1 points (95% CI, 16.9 to 101.3 points). Results of the supportive mITT analysis (censored for transfusion) were consistent with the primary analysis.
Table 16: Change From Baseline at Week 16 in LASA Score in the PEGASUS Trial — Randomized Controlled Period (ITT; Censored for Transfusion)
LASA score | Pegcetacoplan (N = 41) | Eculizumab (N = 39) |
|---|---|---|
Baselinea | ||
n (%) | 40 (97.6) | 38 (97.4) |
LASA score, mean (SD) | 161.0 (68.0) | 156.7 (61.2) |
Week 16 | ||
n (%) | 34 (82.9) | 6 (15.4) |
LASA score, mean (SD) | 217.6 (65.7) | 175.7 (88.9) |
Change from baseline at week 16 in LASA score,a LS mean (SE) | 49.4 (10.2) | –9.7 (19.0) |
Difference in LS means (points),b (95% CI) | 59.1 (16.9 to 101.3) | Reference |
P valuec | 0.0069 | Reference |
CI = confidence interval; ITT = intention to treat; LASA = Linear Analogue Scale Assessment; LS = least squares; SD = standard deviation; SE = standard error.
aBaseline was defined as the average of measurements recorded before the first dose of pegcetacoplan.
bThe analysis was conducted using a MMRM, with the change from baseline to 16 weeks in LASA score as the dependent variable. The model was adjusted for treatment group, study visit, number of packed RBC transfusions in the previous 12 months (< 4 vs. ≥ 4), platelet count at screening (< 100,000/mm3 vs. ≥ 100,000/mm3), the study visit by treatment group interaction, and baseline LASA score (continuous).
cThis end point was not adjusted for multiplicity.
Source: PEGASUS Clinical Study Report (16 weeks).6
EORTC QLQ-C30
Change from baseline at week 16 in the global health status/QoL scale score, and select symptom subscales relevant to PNH (i.e., fatigue, pain, and dyspnea) are summarized in Table 17. The results for other subscales are summarized in Table 40 in Appendix 3.
The LS mean difference in change from baseline at week 16 in global health status score between pegcetacoplan and eculizumab was 18.62 (95%CI, 0.12 to 37.13) points. The difference in LS mean change in score from baseline at week 16 between pegcetacoplan and eculizumab with respect to fatigue, pain, and dyspnea was –20.74 (95% CI, –35.29 to –6.19) points, –2.76 (95% CI, –20.36 to 14.85) points, and –14.57 (95% CI, –29.90 to 0.76) points, respectively.
Table 17: Change From Baseline at Week 16 in Global Health Status/QoL, Fatigue, Pain, and Dyspnea Symptom Subscale Scores of EORTC QLQ C-30 in the PEGASUS Trial — Randomized Controlled Period (ITT; Censored for Transfusion)
Interventions | Total N | Baseline scale scorea | Week 16 scale score | Change from baseline at week 16 in score,a LS mean (SE) | Treatment group difference vs. eculizumab, LS mean differenceb (95% CI) | P valuec | ||
|---|---|---|---|---|---|---|---|---|
n (%) | Mean (SD) | n (%) | Mean (SD) | |||||
Global health status/quality of life | ||||||||
Pegcetacoplan | 41 | 41 (100.0) | 56.30 (20.39) | 35 (89.7) | 71.67 (23.50) | 15.91 (3.64) | 18.62 (0.12 to 37.13) | 0.0486 |
Eculizumab | 39 | 37 (90.2) | 56.53 (20.24) | 5 (12.8) | 51.67 (23.86) | –2.71 (8.52) | Reference | Reference |
Symptom scale: fatigue | ||||||||
Pegcetacoplan | 41 | 41 (100.0) | 49.59 (29.09) | 35 (89.7) | 26.67 (20.38) | –22.93 (3.32) | –20.74 (–35.29 to –6.19) | 0.0062 |
Eculizumab | 39 | 38 (92.7) | 50.29 (24.74) | 6 (15.4) | 38.89 (34.96) | –2.18 (6.64) | Reference | Reference |
Symptom scale: pain | ||||||||
Pegcetacoplan | 41 | 41 (100.0) | 19.51 (26.85) | 35 (89.7) | 18.10 (24.71) | –0.74 (4.32) | −2.76 (–20.36 to 14.85) | 0.7554 |
Eculizumab | 39 | 38 (92.7) | 15.79 (25.10) | 6 (15.4) | 16.67 (25.82) | 2.01 (7.84) | Reference | Reference |
Symptom scale: dyspnea | ||||||||
Pegcetacoplan | 41 | 41 (100.0) | 33.33 (27.89) | 35 (89.7) | 14.29 (23.27) | –20.12 (3.49) | –14.57 (–29.90 to 0.76) | 0.0620 |
Eculizumab | 39 | 38 (92.7) | 43.86 (32.05) | 6 (15.4) | 22.22 (40.37) | –5.55 (7.02) | Reference | Reference |
CI = confidence interval; EORTC QLQ-C30 = European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30; ITT = intention to treat; LS = least squares; SD = standard deviation; SE = standard error.
aBaseline was the last available non-missing observation before the first dose of pegcetacoplan.
bThe analysis was conducted using a MMRM, with the change from baseline to 16 weeks in EORTC QLQ-C30 score as the dependent variable. The model was adjusted for treatment group, study visit, number of packed RBC transfusions in the previous 12 months (< 4 vs. ≥ 4), platelet count at screening (< 100,000/mm3 vs. ≥ 100,000/mm3), the study visit by treatment group interaction, and baseline EORTC QLQ-C30 score (continuous).
cThis end point was not adjusted for multiplicity.
Source: PEGASUS Clinical Study Report (16 weeks).6
Breakthrough Hemolysis
Breakthrough hemolysis was not assessed in the efficacy analysis. Refer to the Harms section for data on hemolysis.
Complications of PNH
Complications of PNH were not assessed in the efficacy analysis. Refer to the Harms section for data on complications of PNH.
Intravascular Hemolysis
Change From Baseline in LDH Level
As summarized in Table 18, the LS mean change from baseline in LDH level at week 16 (key secondary end point) in the ITT set (censored for transfusion) was –14.8 U/L (SE = 42.7 U/L) in the pegcetacoplan arm and –10.1 U/L (SE = 71.0 U/L) in the eculizumab arm. The between-group difference in LS means was –4.6 U/L (95% CI, –181.3 to 172.0 U/L). Noninferiority was not met because the upper bound of the 95% CI of the between-group difference was not less than the NIM of 20 U/L in both the ITT and PP sets (PP analysis in Table 38 in Appendix 3). Results of a supportive analysis based on data uncensored for transfusion (Table 39 in Appendix 4) were consistent with the primary analysis. Change from baseline in LDH level was the third secondary outcome in the statistical testing hierarchy for noninferiority, and no further testing was conducted.
An additional LDH normalization analysis was conducted to assess the proportion of patients who achieved LDH normalization (i.e., LDH level at or below ULN at week 16) in the absence of transfusion (during the randomized controlled period). The proportion was 70.7% in the pegcetacoplan arm and 15.4% in the eculizumab arm, with a risk difference of 48.8% (95% CI, 32.3% to 65.3%). In the analysis based on data uncensored for transfusion (Table 39), LDH normalization was achieved in 73.2% of patients in the pegcetacoplan arm and 59.0% of patients in the eculizumab arm, with a risk difference of 12.3% (95% CI, –7.0% to 31.5%).
Table 18: LDH Outcomes in the PEGASUS Trial — Randomized Controlled Period (ITT; Censored for Transfusion)
LDH level | Pegcetacoplan (N = 41) | Eculizumab (N = 39) |
|---|---|---|
Baselinea | ||
n (%) | 41 (100) | 39 (100) |
LDH level (U/L), mean (SD) | 257.5 (97.6) | 308.6 (284.8) |
Week 16 | ||
n (%) | 35 (85.4) | 6 (15.4) |
LDH level (U/L), mean (SD) | 188.8 (79.1) | 183.3 (28.8) |
Change from baseline at week 16 in LDH level (U/L),a,b LS mean (SE) | –14.8 (42.7) | –10.1 (71.0) |
Difference in LS means (U/L),b (95% CI) | –4.6 (–181.3 to 172.0) | Reference |
P valuec | 0.96 | Reference |
Patient achieving LDH normalization in the absence of transfusion,d n (%) | 29 (70.7) | 6 (15.4) |
Risk differencee (%), (95% CI) | 48.8 (32.3 to 65.3) | Reference |
CI = confidence interval; ITT = intention to treat; LDH = lactate dehydrogenase; LS = least squares; SD = standard deviation; SE = standard error.
aBaseline was defined as the average of measurements recorded before the first dose of pegcetacoplan.
bThe analysis was conducted using a MMRM, with change from baseline at week 16 in LDH level as the dependent variable. The model was adjusted for treatment group, study visit, number of packed RBC transfusions in the previous 12 months (< 4 vs. ≥ 4), platelet count at screening (< 100,000/mm3 vs. ≥ 100,000/mm3), the study visit by treatment group interaction, and baseline LDH level (continuous).
cIn accordance with the hierarchal testing procedure, noninferiority but not superiority testing was conducted.
dThe normal range of LDH is 113 to 226 U/L.
eThe risk difference and 95% CI were constructed using the stratified Miettinen-Nurminen method.
Source: PEGASUS Clinical Study Report (16 weeks).6
Extravascular Hemolysis
A summary of reticulocyte, indirect bilirubin, and haptoglobin outcomes is provided in Table 19.
Reticulocyte Outcomes
The change from baseline in ARC at week 16 (censored for transfusion) was a key secondary end point and was tested for noninferiority according to the hierarchical testing procedure. The LS mean change from baseline in ARC at week 16 in the ITT set (censored for transfusion) was –135.8 × 109 cells/L (SE = 6.5 × 109 cells/L) in the pegcetacoplan arm and 27.9 × 109 cells/L (SE = 11.9 × 109 cells/L) in the eculizumab arm, with a between-group adjusted mean difference of –163.6 × 109 cells/L (95% CI, –189.9 to –137.3 × 109 cells/L). Noninferiority was met because the upper bound of the 95% CI of the adjusted mean difference was less than the prespecified the NIM of 10 × 109 cells/L in the ITT and PP sets (PP analysis in Table 38 in Appendix 3). Results of a supportive analysis based on data uncensored for transfusion (Table 39 in Appendix 3) were also consistent with the primary analysis.
Reticulocyte normalization in the absence of transfusions at week 16, a secondary end point, was achieved in 78.0% of patients in the pegcetacoplan arm and 2.6% of patients in the eculizumab arm, with a risk difference of 66.4% (95% CI, 53.1% to 79.7%). In the analysis based on data uncensored for transfusion (Table 39), ARC normalization was achieved in 80.5% of patients in the pegcetacoplan arm and 17.9% of patients in the eculizumab arm, with a risk difference of 54.8% (95% CI, 38.8% to 70.7%).
Indirect Bilirubin and Haptoglobin
The LS mean change from baseline in indirect bilirubin level at week 16 (secondary end point) in the ITT set (censored for transfusion) was –17.8 µmol/L (SE = 2.3 µmol/L) in the pegcetacoplan arm and 4.15 µmol/L (SE = 4.48 µmol/L) in the eculizumab arm, with a between-group difference of –21.9 µmol/L (95% CI, –32.5 to –11.4 µmol/L).
The LS mean change from baseline in haptoglobin level at week 16 (secondary end point) in the ITT set (censored for transfusion) was –0.02 g/L (SE = 0.03 g/L) in the pegcetacoplan arm and 0.12 g/L (SE = 0.06 g/L) in the eculizumab arm, with a between-group difference of –0.14 g/L (–0.28 to –0.01 g/L).
Table 19: Reticulocyte, Indirect Bilirubin, and Haptoglobin Outcomes in the PEGASUS Trial — Randomized Controlled Period (ITT; Censored for Transfusion)
Parameters | Pegcetacoplan (N = 41) | Eculizumab (N = 39) |
|---|---|---|
Reticulocyte | ||
Baselinea | ||
n (%) | 41 (100) | 39 (100) |
ARC (109 cells/L), mean (SD) | 217.5 (75.0) | 216.2 (69.1) |
Week 16 | ||
n (%) | 34 (82.9) | 6 (15.4) |
ARC (109 cells/L), mean (SD) | 77.7 (26.9) | 220.0 (92.3) |
Change from baseline in ARC at week 16a,b (109 cells/L), LS mean (SE) | –135.8 (6.5) | 27.8 (11.9) |
Difference in LS meansb (109 cells/L), (95% CI) | –163.6 (–189.9 to –137.3) | Reference |
P valuec | < 0.0001 | Reference |
Patient achieving reticulocyte normalization in the absence of transfusion,d n (%) | 32 (78.0) | 1 (2.6) |
Risk differencee,f (%), (95%CI) | 66.4 (53.1 to 79.7) | Reference |
Indirect bilirubin | ||
Baselinea | ||
n (%) | 41 (100) | 39 (100) |
Indirect bilirubin level (µmol/L), mean (SD) | 34.7 (28.5) | 32.9 (23.0) |
Week 16 | ||
n (%) | 34 (82.9) | 6 (15.4) |
Indirect bilirubin level (µmol/L), mean (SD) | 13.7 (16.3) | 25.4 (20.6) |
Change from baseline at week 16 in indirect bilirubin levela,b (µmol/L), LS mean (SE) | –17.8 (2.7) | 4.2 (4.5) |
Difference in LS meansb (µmol/L), (95% CI) | –21.9 (–32.5 to –11.4) | Reference |
P valuef | 0.0002 | Reference |
Haptoglobin | ||
Baselinea | ||
n (%) | 41 (100) | 39 (100) |
Haptoglobin level (g/L), mean (SD) | 0.14 (0.13) | 0.13 (0.12) |
Week 16 | ||
n (%) | 34 (82.9) | 6 (15.4) |
Haptoglobin level (g/L), mean (SD) | 0.14 (0.16) | 0.26 (0.32) |
Change from baseline at week 16 in haptoglobin levela,b (g/L), LS mean (SE) | –0.02 (0.03) | 0.12 (0.06) |
Difference in LS meansb (g/L), (95% CI) | –0.14 (–0.28 to –0.01) | Reference |
P valuef | 0.0369 | Reference |
ARC = absolute reticulocyte count; CI = confidence interval; ITT = intention to treat; LS = least squares; SD = standard deviation; SE = standard error.
aBaseline was defined as the average of measurements recorded before the first dose of pegcetacoplan.
bThe analysis was conducted using a MMRM. The model was adjusted for treatment group, study visit, number of packed RBC transfusions in the previous 12 months (< 4 vs. ≥ 4), platelet count at screening (< 100,000/mm3 vs. ≥ 100,000/mm3), the study visit by treatment group interaction, and their own baseline level (continuous).
cIn accordance with the hierarchal testing procedure, noninferiority but not superiority testing was conducted.
dReticulocyte normalization was defined as a reticulocyte count below the ULN (i.e., 120 × 109 cells/L).
eThe risk difference and 95% CI were constructed using the stratified Miettinen-Nurminen method.
fThis end point was not included in the statistical testing hierarchy.
Source: PEGASUS Clinical Study Report (16 weeks).6
Health Care Resource Utilization
Health care resource utilization was not measured in this study.
Harms
Only those harms identified in the review protocol are reported here. Refer to Table 20 for detailed harms data in the randomized controlled period.
Table 20: Summary of Harms in the PEGASUS Trial — Randomized Controlled Period (Safety Set)
Harms | Pegcetacoplan (N = 41) | Eculizumab (N = 39) |
|---|---|---|
Patients with ≥ 1 TEAE | ||
n (%) | 36 (87.8) | 34 (87.2) |
Most common events,a n (%) | ||
Hemolysis | 4 (9.8) | 9 (23.1) |
Headache | 3 (7.3) | 9 (23.1) |
Diarrhea | 9 (22.0) | 1 (2.6) |
Injection site erythema | 7 (17.1) | 0 |
Fatigue | 2 (4.9) | 6 (15.4) |
Anemia | 0 | 5 (12.8) |
Injection site reaction | 5 (12.2) | 0 |
Abdominal pain | 5 (12.2) | 4 (10.3) |
Dizziness | 1 (2.4) | 4 (10.3) |
Back pain | 3 (7.3) | 4 (10.3) |
Patients with ≥ 1 serious TEAE | ||
n (%) | 7 (17.1) | 6 (15.4) |
Most common events,b n (%) | ||
Hemolysis | 2 (4.9) | 1 (2.6) |
Anemia | 0 | 2 (5.1) |
Patients who discontinued treatment due to a TEAE | ||
n (%) | 3 (7.3) | 0 |
Hemolysis | 3 (7.3) | 0 |
Deaths | ||
n (%) | 0 | 0 |
Notable harms | ||
Serious bacterial infection–related TEAE, n (%) | 1 (2.4) | 0 |
Injection site–related TEAE, n (%) | 15 (36.6) | 1 (2.6) |
Most common events,c n (%) | ||
Injection site erythema | 7 (17.1) | 0 |
Injection site reaction | 5 (12.2) | 0 |
Injection site swelling | 4 (9.8) | 0 |
Injection site induration | 3 (7.3) | 0 |
Anti-pegcetacoplan peptide antibody-positive, n (%) | 0 | 2 (5.1) |
Thrombosis, n (%) | 0 | 0 |
Pulmonary hypertension | 0 | 0 |
Chronic kidney disease | 0 | 0 |
Hemolysis, n (%) | 4 (9.8) | 9 (23.1) |
TEAE = treatment-emergent adverse event.
Note: A TEAE was defined as an AE that commenced on or after the first study drug administration or an AE that increased in severity after pre-treatment.
aFrequency > 10%.
bOccurred in ≥ 2 patients.
cOccurred in ≥ 3 patients.
Source: PEGASUS Clinical Study Report (16 weeks).6
Run-In Period
During the run-in period (when both pegcetacoplan and eculizumab were used concurrently for 4 weeks), TEAEs were reported in 69 patients (86.3%), but none led to death, discontinuation of the study treatment or the study. The most common TEAEs were injection site erythema (38.8%), injection site pruritus (13.8%), and headache (12.5%), none of which was serious. A serious TEAE was reported in 1 patient due to sepsis and was considered related to both pegcetacoplan and eculizumab. This serious TEAE was resolved during the run-in period despite continued treatment with both pegcetacoplan and eculizumab. This patient was later randomly assigned to pegcetacoplan and had no subsequent TEAEs related to infection.
Randomized Controlled Period
Adverse Events
During the randomized controlled period, TEAEs were reported in 87.8% and 87.2% of patients in the pegcetacoplan arm and the eculizumab arm, respectively. The most common TEAEs (in at least 10% of patients) with pegcetacoplan treatment included diarrhea, injection site erythema, injection site reaction, and abdominal pain, all of which were more commonly reported in the pegcetacoplan arm than the eculizumab arm. The incidence of breakthrough hemolysis (9.8%), headache (7.3%), fatigue (4.9%), dizziness (2.4%), backpain (7.3%), and anemia (0% were numerically lower in the pegcetacoplan arm than the eculizumab arm (hemolysis and headache: 23.1%; fatigue: 15.4%; dizziness and backpain: 10.3%; anemia: 12.8%).
Serious Adverse Events
Serious TEAEs were reported in 7 (17.1%) patients in the pegcetacoplan arm, with the most common event being hemolysis, reported in 2 patients. Serious TEAEs were reported in 6 (15.4%) patients in the eculizumab arm, with the most common event being anemia (reported in 2 patients).
Withdrawals Due to TEAE
Three (7.3%) patients in the pegcetacoplan arm discontinued treatment due to a TEAE, all as a result of breakthrough hemolysis. No patient in the eculizumab arm discontinued treatment due to a TEAE.
Mortality
No death was reported in either treatment arm of the study.
Notable Harms
Serious bacterial infections: Serious treatment-emergent bacterial infection was reported in 1 (2.4%) patient in the pegcetacoplan and none of the patients in the eculizumab arm. No serious infections in the study were known to be caused by an encapsulated organism.
Infusion-related reaction: A notably higher incidence of injection site–related TEAEs was reported in the pegcetacoplan arm (36.6%) than the eculizumab arm (2.6%), with the most common events (≥ 5%) in the pegcetacoplan arm being injection site erythema (17.1%), reaction (12.2%), swelling (9.8%), and induration (7.3%).
Immunogenicity: No patient in the pegcetacoplan arm and 2 patients in the eculizumab arm had a positive antipegcetacoplan peptide antibody response.
Thrombosis: There was no report of thrombosis in either treatment arm.
Pulmonary hypertension: There was no report of pulmonary hypertension in either treatment arm.
Chronic kidney disease: There was no report of chronic kidney disease in either treatment arm.
Hemolysis: Breakthrough hemolysis was reported in 4 (9.8%) patients in the pegcetacoplan arm and in 9 (23.1%) patients in the eculizumab arm.
Critical Appraisal
Internal Validity
The PEGASUS trial was a phase III, randomized, open-label, active-controlled trial. The methods of randomization, which involved stratification by the number of RBC transfusions in the previous 12 months and platelet count and concealment of the randomized assignment with interactive response technology, were appropriate. There was no notable difference between treatment arms for most baseline characteristics. The pegcetacoplan arm had a shorter mean time since diagnosis, a lower mean LDH level, and a higher proportion of patients with a history of thrombosis and a higher proportion of patients receiving eculizumab at an increased dose at baseline than in the eculizumab arm, although the clinical experts consulted by CADTH commented that these differences were unlikely to have confounded the study results.
The open-label study design was reasonable, given the distinct routes and frequency of administration of the 2 interventions, which could allow investigators and patients to make inferences about treatment assignment, regardless of blinding. However, reporting of FACIT-Fatigue, LASA, and EORTC QLQ-C30 outcomes was prone to bias in favour of pegcetacoplan, given the subjective nature of these outcomes, although the extent of bias is unclear. Reporting of AEs could also have been biased due to the open-label study design, with potential for bias in favour of eculizumab. In addition, a high number of major protocol deviations related to study assessment or schedule noncompliance (54.3%), (e.g., missed timing for laboratory or vital signs collection, missed visits, assessment incomplete by patient) was reported; these were more common in the eculizumab arm (64.1%) than the pegcetacoplan arm (48.8%). Nonetheless, all major protocol deviations that were judged to have influenced efficacy assessment were excluded from the PP set.
Transfusion was allowed in patients with Hb levels lower than 9 g/dL if symptoms were present. Because symptom evaluation is subjective, transfusion decisions could be influenced by knowledge of treatment assignment, leading to potential bias in favour of pegcetacoplan. Transfusion was allowed at the discretion of the investigators in 2 patients in the eculizumab arm who did not meet the predefined clinical criteria. Considering that an independent blinded review of these protocol deviations took place before the database lock, the incidence of deviations was small, and such deviations were excluded from the PP set, the concern for bias is small.
Prior to the randomized controlled period, patients received pegcetacoplan and eculizumab concurrently to avoid insufficient complement inhibition in the 4-week run-in period. The clinical experts commented that the interventions administered during the run-in period would not influence the primary efficacy assessment at week 16, given than the drugs require frequent dosing (every 2 weeks for eculizumab, twice weekly for pegcetacoplan) and would have been completely washed out by week 16.
The statistical analyses were generally well designed. The study was powered to detect a treatment difference in the primary end point between treatment arms in the ITT population and the enrolled sample size was adequate. A hierarchical testing procedure was appropriately used to account for multiplicity in all key secondary efficacy end points. No formal hypothesis testing was conducted for categorical secondary efficacy outcomes (Hb normalization, Hb response, reticulocyte normalization) or for number of packed RBCs transfused. The statistical testing for HRQoL outcomes (LASA, EORTC QLQ-C30 scores), indirect bilirubin, and haptoglobin were associated with an increased risk of type I error due to lack of control for multiplicity. No conclusion can be made about subgroup effects because the subgroups of interest related to disease activity were not addressed in the submission.
The sponsor assumed a treatment difference of 1 g/dL between pegcetacoplan and eculizumab in the sample size calculation, as informed by PNH experts. The clinical experts consulted by CADTH considered an improvement of 2 g/dL to be highly favourable; however, an improvement of at least 1 g/dL was considered reasonable and is expected to result in a clinically meaningful improvement in most patients with PNH, especially those with severe anemia at baseline. It has been proposed that Hb level is a strong surrogate biomarker for clinical benefits in patients with sickle cell disease receiving stem cell or gene therapy;24,25 however, evidence in patients with PNH is scarce due to the rarity of the disease. Justification for NIMs of the FACIT-Fatigue Scale, ARC, LDH level, and transfusion avoidance were not available from the sponsor due to the scarcity of information on comparable patient populations at the start of the trial. A recent study using registry data suggested the use of 5 points as a clinically important difference for the FACIT-Fatigue Scale for in PNH patients;27 however, the study examined a treatment-naive population that does not fully align with the patient population of interest to this review (treatment-experienced), and it is also unclear if the identified value was a MID. Nonetheless, the chosen NIM of 3 points would be considered conservative for the FACIT-Fatigue Scale. The clinical experts consulted by CADTH noted that the NIMs for transfusion avoidance and LDH level were reasonable.
For a conservative approach, it is generally preferred that the claim of noninferiority be based on agreement between the ITT and PP populations. PP analyses were conducted as supportive analyses in the trial. For the key secondary outcomes that were tested for noninferiority (i.e., transfusion avoidance, change from baseline at week 16 in ARC, change from baseline at week 16 in LDH level), results in the supportive PP analyses were consistent with those in the primary ITT analyses.
Bias due to study drop-outs is unlikely, given the low incidence of study discontinuation in both treatment arms (pegcetacoplan: 2.4%; eculizumab: 0%) at week 16. Missing data were found to be at least 10% in the pegcetacoplan arm and at least 85% in the eculizumab arm for all efficacy outcomes, which was largely due to data censoring after a transfusion event. Missing data for continuous outcomes were implicitly imputed by MMRM, which relies on the assumption that data are missing at random. This assumption is unlikely to hold for Hb outcomes because missingness was related to censoring for transfusion and transfusion need was dependent on Hb level. The study conducted sensitivity analyses (the control-based pattern imputation method and a tipping point analysis) that used imputation methods based on the missingness-not-at-random assumption for the primary end point, as well as a supportive analysis that assesses all patient data (i.e., no censoring for transfusion) for the primary and all key secondary efficacy end points. Results of these supportive and sensitivity analyses align with the primary analysis, increasing the certainty of the findings. However, the clinical experts indicated that LDH level is independent of transfusion. An agreement between the primary LDH analysis (censored for transfusion) and the additional analysis uncensored for transfusion was observed, providing reassurance for this outcome.
The validity, reliability, and responsiveness of the patient-reported outcome instruments (EORTC QLQ-C30, FACIT-Fatigue, LASA) have not been characterized in patients with PNH previously, aside from 1 study showing evidence of content validity for the EORTC QLQ-C30 and FACIT-Fatigue Scale in this patient population.26 In addition, MID estimates for PNH have not been established for these instruments.
External Validity
According to the clinical experts consulted by CADTH, the inclusion and exclusion criteria, in general, align with selection criteria that would be adopted by most clinicians when identifying suitable candidates for pegcetacoplan. The clinical experts commented that the inclusion of patients with an Hb level of 10.5 g/dL was consistent with the criteria used by most clinicians in clinical practice, although they recognized that there may be variability in the cut-offs used by other clinicians. The clinical experts noted most patients would not be switched to pegcetacoplan immediately after receiving a stable dose of eculizumab for 3 months, as it would generally take at least 6 months to ascertain the underlying cause of treatment failure; however, the concern is likely minimal because most patients appeared to have a long history of eculizumab treatment at baseline. In addition, patients with certain cardiovascular diseases and those receiving QT-prolonging medications were excluded from the study, which the clinical experts noted was reasonable and could be due to safety concerns, although there are rarely QT-related issues with complement inhibitors in their experience. Patients with intolerance to C5 inhibitors were also not included, although the impact on generalizability is expected to be small, as such patients are uncommon, per the clinical experts. The baseline characteristics of the study population were generally reflective of the population with PNH in Canada, according to the clinical experts.
The dosing, criteria for dose escalation, and administration of pegcetacoplan in the study were consistent with the product monograph. The clinical experts considered eculizumab to be an appropriate comparator, as it is a standard of care and is prescribed for most patients with PNH in clinical practice. Patients in the eculizumab arm received treatment based on their dose established before the study. The clinical experts commented that the distribution of eculizumab dosing in the study population aligns with clinical practice, with approximately 20% to 30% of patients receiving higher or more frequent doses than the standard recommended dosage. In the study, transfusion was provided according to prespecified criteria based on Hb levels and the presence of symptoms. The clinical experts noted the transfusion criteria were consistent with the criteria used in clinical practice.
The efficacy outcomes measured in the study were of clinical importance to both patients and clinicians. The clinical experts and patient group input both noted that improvements in Hb level and QoL and reductions in hemolysis, symptoms, and transfusion needs were the key goals of treatment for PNH, and all were measured in the trial. The clinical experts noted that the duration of follow-up (16 weeks) was adequate for assessing these outcomes; however, for other important outcomes, such as breakthrough hemolysis, survival, thrombosis, and other complications of PNH, a longer follow-up, of at least 2 years, would be required to gain certainty in the results. The clinical relevance of patient-reported outcome instruments (FACIT-Fatigue, EORTC QLQ-C30, LASA) is uncertain because they are not routinely administered in clinical practice, per the clinical experts. Nevertheless, these scales have captured some of the common symptoms of PNH (e.g., fatigue, pain, dyspnea) reported by patients and would be considered relevant in the assessment of pegcetacoplan.
It should be noted that this trial is the only phase III study to provide direct comparative evidence between pegcetacoplan and a C5 inhibitor. The absence of head-to-head evidence comparing pegcetacoplan and ravulizumab in patients with an inadequate response to ravulizumab represents an evidence gap in the management of PNH.
Indirect Evidence
Objectives and Methods for the Summary of Indirect Evidence
In the absence of a head-to-head comparison between pegcetacoplan and ravulizumab, CADTH conducted a literature review to identify indirect treatment comparisons (ITCs) that could supplement the available direct evidence from the PEGASUS trial in addition to the sponsor’s submitted ITC. A focused literature search for ITCs mentioning PNH, pegcetacoplan, ravulizumab, or eculizumab was conducted in MEDLINE All, with no limits applied to search results. Titles, abstracts, and full-text articles were screened for inclusion by 2 reviewers based on the review criteria outlined in Table 5. Of the 2 potentially relevant ITCs identified by the search, 1 was a duplicate of the sponsor-submitted ITC and the other did not include pegcetacoplan. In total, 1 sponsor-submitted ITC,7 which was used to inform the pharmacoeconomic model, was summarized and critically appraised.
Description of Indirect Comparison
The sponsor submitted an anchored MAIC to evaluate the relative efficacy of pegcetacoplan to ravulizumab in adult patients with PNH previously treated with eculizumab. The MAIC did not report a systematic literature review to identify relevant studies for inclusion. Two studies were included in the analysis: patient-level data from the PEGASUS study, comparing pegcetacoplan and eculizumab, and aggregate patient data from the ALXN study8 comparing ravulizumab and eculizumab. Outcomes analyzed were transfusion avoidance, number of packed RBCs transfused, Hb level stabilization, change from baseline in LDH level, LDH level normalization, fatigue and fatigue symptoms, global health status, and physical functioning.
Methods of MAIC
Objectives
The objective of the sponsor’s MAIC was to assess the comparative efficacy of pegcetacoplan and ravulizumab in adult patients with PNH previously treated with eculizumab.
Study Selection Methods
The authors did not report a literature search or describe their methods for data extraction or quality assessment of the 2 included studies. The outcomes and their corresponding time of assessment from the 2 studies are summarized in Table 13. Definitions for clinical, hematological, fatigue, and QoL outcomes were similar in the PEGASUS and ALXN studies.
Table 21: Comparison of Study Outcomes in the PEGASUS and ALXN Studies
Outcome | PEGASUS | ALXN |
|---|---|---|
Clinical and hematological | ||
Transfusion avoidance | Proportion of patients with transfusion avoidance through week 16 | Proportion of participants who remained transfusion-free and did not require a transfusion, per protocol-specified guidelines, through week 26 |
Transfusion requirements | Total number of units of packed RBCs transfused from baseline to week 16 | Total number of units of packed RBCs transfused from baseline to week 26 |
Hb level stabilization | Proportion of patients with avoidance of a ≥ 2 g/dL decrease in Hb level in the absence of transfusion from baseline through week 16 | Proportion of patients with avoidance of a ≥ 2 g/dL decrease in Hb level in the absence of transfusion from baseline through week 26 |
LDH level | Week 16 change from baseline in LDH level | Week 16 change from baseline in LDH level |
LDH level normalization | Proportion of patients with LDH level ≤ 1 × ULN (226 U/L) at week 16 in the absence of transfusions from baseline through week 16a | Proportion of patients with a LDH level ≤ 1 × ULN (246 U/L) from baseline at week 16b |
Fatigue and quality of life | ||
Fatigue | Week 16 change from baseline in FACIT-Fatigue score | Week 26 change from baseline in FACIT-Fatigue score |
Global health status | Week 16 change from baseline in global health status EORTC QLQ-C30 score | Week 26 change from baseline in global health status EORTC QLQ-C30 score |
Physical functioning | Week 16 change from baseline in physical functioning EORTC QLQ-C30 score | Week 26 change from baseline in physical functioning EORTC QLQ-C30 score |
Fatigue symptoms | Week 16 change from baseline in fatigue symptoms EORTC QLQ-C30 score | Week 26 change from baseline in fatigue symptoms EORTC QLQ-C30 score |
EORTC QLQ-C30 = European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30; FACIT = Functional Assessment of Chronic Illness Therapy; Hb = hemoglobin; LDH = lactate dehydrogenase; RBC = red blood cell; ULN = upper limit of normal.
aLDH level normalization is defined as the proportion of patients who achieved an LDH level ≤ 1 × ULN (226 U/L) in the absence of transfusions from baseline through the end of follow-up.
bLDH level normalization is defined as the proportion of patients who achieved an LDH level ≤ 1 × ULN (246 U/L), with or without transfusions (i.e., patients were not excluded if they received transfusions during follow-up).
Source: Sponsor-submitted ITC.7
MAIC Analysis Methods
The authors used an anchored MAIC by combining individual patient data from the PEGASUS trial and aggregate data from the ALXN trial to compare pegcetacoplan to ravulizumab anchored to the common comparator eculizumab. Baseline demographic and clinical characteristics were summarized using frequencies and percentages for categorical variables and means and SDs for continuous variables before and after matching procedures. Baseline characteristics were classified as an effect modifier, a purely prognostic variable, or neither, based on clinical input regarding biological plausibility. Classification of variables by type of outcome are provided in Table 22.
Table 22: Classification of Variables by Type of Outcome
Characteristic | Outcomes related to hemoglobina | Outcomes related to intravascular hemolysisb | Outcomes related to fatigue and quality of lifec |
|---|---|---|---|
Age | Effect modifier | Effect modifier | Effect modifier |
Sex | Effect modifier | Effect modifier | Neither |
Race | Effect modifier | Effect modifier | Neither |
Weight | Neither | Neither | Effect modifier |
Height | Neither | Neither | Neither |
Transfusion history | Effect modifier | Effect modifier | Effect modifier |
History of aplastic anemia | Effect modifier | Effect modifier | Effect modifier |
LDH value (U/L) | Effect modifier | Effect modifier | Effect modifier |
Hemoglobin (g/dL) | Effect modifier | Effect modifier | Effect modifier |
Number of years on eculizumab before first study infusion | Purely prognostic | Purely prognostic | Purely prognostic |
Number of years from diagnosis to consent | Purely prognostic | Purely prognostic | Purely prognostic |
LDH = lactate dehydrogenase.
aOutcomes include transfusion avoidance, transfusion requirements, Hb level, and hemoglobin stabilization.
bOutcomes include LDH level (percent change and change from baseline), LDH normalization, and breakthrough hemolysis.
cOutcomes include fatigue (FACIT-Fatigue Scale), general health status (EORTC QLQ-C30), physical functioning (EORTC QLQ-C30), and fatigue symptoms (EORTC QLQ-C30).
Source: Sponsor-submitted ITC.7
A propensity score model using logistic regression was used to estimate the likelihood of enrolment in the ALXN study versus the PEGASUS study. The logistic regression model was estimated using the method of moments. Weights were assigned such that the weighted means and proportions of baseline characteristics in the PEGASUS study exactly matched those reported in the ALXN study. The weight of each patient enrolled in the PEGASUS study was equal to the inverse odds of their enrolment in the ALXN study versus the PEGASUS study. Model adequacy was assessed with visual inspection of histograms of patient weights and effective sample size (ESS). Models that retained at least 50% of the initial PEGASUS population while adjusting for effect modifiers were selected. The distribution of baseline demographic and clinical characteristics was compared across patient groups before and after matching. Comparisons of clinical and hematological outcomes with fatigue and QoL outcomes were adjusted for separate baseline characteristics. Before matching, Wald tests and 95% CIs were used to compare categorical and continuous outcomes (i.e., chi square distribution and z score, respectively). After matching, outcomes were compared between balanced treatment groups using weighted Wald tests with 95% CIs that incorporated weights generated during matching.
Anchored comparisons based on the unweighted sample before matching and unanchored comparisons (i.e., excluded data for patients randomized to eculizumab) were conducted as sensitivity analyses. All effect modifiers, whether balanced or imbalanced at baseline between the PEGASUS and ALXN studies and all prognostic variables were included as matching variables. In addition, a sensitivity analysis was conducted by revising the definition of LDH level normalization in the PEGASUS study to match that in the ALXN study, which meant that patients who received a transfusion during follow-up were not excluded from the measurement of LDH level normalization.
Results of MAIC
Summary of Included Studies
Study characteristics of the PEGASUS and ALXN studies are summarized in the Table 23.
The patients included in the MAIC were 18 years or older, previously treated with eculizumab (for at least 3 months in the PEGASUS study and for at least 6 months in the ALXN study), received meningococcal vaccination, had an absolute neutrophil count greater than 500/mm3 at screening, had an adequate platelet count at screening (greater than 50,000/mm3 in the PEGASUS study and greater than 30,000/mm3 in the ALXN study), and did not have a history of bone marrow transplant. The following criteria were not included in the ALXN study: Hb level less than 10.5 g/dL and ARC greater than 1 times the ULN at screening. The individual patient data from the PEGASUS study were reanalyzed, and patients with an LDH level of 1.5 times the ULN or less at screening and without major adverse vascular events in the 6 months before treatment were selected to align more closely with the patients enrolled in the ALXN study. After implementing these 2 criteria, the ESSs for the pegcetacoplan and eculizumab groups were 36 (87.8% of the original sample size) and 32 patients (82.1% of the original sample size), respectively. Five patients in the pegcetacoplan group and 7 patients in the eculizumab group were excluded in the analysis because their LDH was greater than 1.5 times the ULN at screening. No patients in the PEGASUS study had a major adverse vascular event in the 6-month period before treatment. In the ALXN study, 97 patients in the ravulizumab group and 98 patients in the eculizumab group were included in the analysis.
The baseline characteristics of patients before and after matching are summarized in Table 24. Prior to matching, the distribution of effect modifiers, including patient age, race (white, African American, and other or multiple races), weight, history of aplastic anemia, and LDH level, was similar for patients randomized to receive pegcetacoplan in the PEGASUS study and those randomized to receive ravulizumab in the ALXN study. There were fewer patients of Asian race in the pegcetacoplan group than in the ravulizumab group (13.9% versus 23.7%). Compared with patients who received ravulizumab, a greater proportion of pegcetacoplan patients were female (69.4% versus 48.5%) and had a history of transfusions in the year before the study (72.2% versus 13.4%). Mean Hb was lower for patients who received pegcetacoplan than for those who received ravulizumab (8.7 g/dL versus 11.1 g/dL).
Comparisons of clinical and hematological outcomes were adjusted for patient age at first study infusion, female sex, white race, Asian race, history of aplastic anemia, and LDH level at baseline, such that the means and proportions of these effect modifiers matched exactly for patients receiving pegcetacoplan and ravulizumab. After matching procedures, the ESS was 24 patients and 12 patients for pegcetacoplan and eculizumab, respectively. Comparisons for fatigue and QoL outcomes were adjusted for patient age at first study infusion, body weight, history of aplastic anemia, and LDH level at baseline. The authors noted that they did not match the effect modifiers of Hb level and history of transfusions due to reduced ESS and the presence of extreme patient inverse probability weights. No description was provided for the handling of different follow-up times reported in the studies (16 weeks for PEGASUS and 26 weeks for ALXN). After matching procedures, the ESS was 34 patients and 9 patients for pegcetacoplan and eculizumab, respectively.
Table 23: Summary of Study Characteristics
Study characteristic | PEGASUS | ALXN |
|---|---|---|
Key inclusion criteria |
|
|
Key exclusion criteria |
|
|
Treatment Description | The treatment period consisted of 3 parts: 1.4-week run-in period during which all patients received both pegcetacoplan and eculizumab 2.16-week randomized controlled period during which patients were randomized to receive either monotherapy pegcetacoplan or monotherapy eculizumab 3.32-week open-label period during which all patients received pegcetacoplan | The treatment period consisted of 3 parts: 1.4-week enrolment period during which all patients received monotherapy eculizumab 2.26-week randomized treatment period during which patients were randomized to receive either monotherapy ravulizumab or monotherapy eculizumab 3.open-label extension period during which all patients received ravulizumab for up to 2 years |
Sample size | 80 patients
| 195 patients
|
Location | Australia, Belgium, Canada, France, Germany, Japan, Republic of Korea, Russia, Spain, UK, US | Australia, Canada, France, Germany, Italy, Japan, Republic of Korea, Netherlands, Spain, UK, US |
Duration of randomized treatment period | 16 weeks | 26 weeks |
Treatment dose |
|
|
CABG = coronary artery bypass graft; ESS = effective sample size; LDH = lactate dehydrogenase; MAVE = major adverse vascular event; NYHA = New York Heart Association; PNH = paroxysmal nocturnal hemoglobinuria; ULN = upper limit of normal.
Source: Sponsor-submitted ITC.7
Table 24: Baseline Characteristics of the PEGASUS and ALXN Studies Before and After Matching — Clinical and Hematological Outcomes
Characteristic | PEGASUS (before matching) | PEGASUS (after matchinga) | ALXN (as reported) | |||
|---|---|---|---|---|---|---|
Pegcetacoplan | Eculizumab | Pegcetacoplan | Eculizumab | Ravulizumab | Eculizumab | |
Sex | ||||||
Male, % | 30.6 | 40.6 | 51.5 | 49.0 | 51.5 | 49.0 |
Female,b % | 69.4 | 59.4 | 48.5 | 51.0 | 48.5 | 51.0 |
Age at first infusion of study drug,b mean (SD) years | 49.0 (16.8) | 48.8 (14.0) | 46.4 (15.5) | 48.8 (12.0) | 46.4 (14.4) | 48.8 (14.0) |
Race | ||||||
White,b % | 58.3 | 65.6 | 51.5 | 62.2 | 51.5 | 62.2 |
Asian,b % | 13.9 | 15.6 | 23.7 | 19.4 | 23.7 | 19.4 |
Black, % | 5.6 | 0.0 | 11.1 | 0.0 | 5.2 | 3.1 |
Other or multiple races, % | 0.0 | 3.1 | 0.0 | 9.2 | 3.1 | 1.0 |
Not reported, % | 22.2 | 15.6 | 13.7 | 9.2 | 16.5 | 14.3 |
Weight, mean (SD) kg | 75.2 (19.6) | 73.2 (14.2) | 81.8 (27.1) | 79.1 (10.8) | 72.4 (16.8) | 73.4 (14.6) |
Height, mean (SD) cm | 167.1 (9.7) | 168.8 (7.4) | 170.3 (12.6) | 169.6 (7.9) | 168.3 (10.1) | 168.8 (9.9) |
Time on eculizumab before first study infusion, mean (SD) years | 5.4 (4.4) | 5.1 (3.8) | 5.1 (4.5) | 5.8 (3.9) | 6.0 (3.5) | 5.6 (3.5) |
History of transfusions in the 1 year before first dose, % | 72.2 | 71.9 | 66.7 | 72.9 | 13.4 | 12.2 |
Age at PNH diagnosis, mean (SD) years | 40.5 (17.0) | 35.7 (13.4) | 38.6 (15.6) | 36.0 (12.0) | 34.1 (14.4) | 36.8 (14.1) |
Time from PNH diagnosis to consent, mean (SD) years | 8.5 (7.1) | 13.0 (9.8) | 7.8 (7.4) | 12.8 (9.8) | 12.4 (8.4) | 11.9 (9.4) |
LDH,b mean (SD) U/L | 229.0 (57.2) | 203.5 (35.5) | 228.0 (50.9) | 235.2 (30.7) | 228.0 (48.7) | 235.2 (49.7) |
Hemoglobin, mean (SD) g/dL | 8.7 (1.1) | 8.7 (0.8) | 8.7 (1.1) | 8.6 (0.7) | 11.1 (1.8) | 10.9 (1.8) |
History of major adverse vascular events, % | 25.0 | 18.8 | 19.9 | 9.6 | 28.9 | 22.4 |
History of aplastic anemia,b % | 27.8 | 18.8 | 35.1 | 39.8 | 35.1 | 39.8 |
ESS = effective sample size; LDH = lactate dehydrogenase; PNH = paroxysmal nocturnal hemoglobinuria; SD = standard deviation.
aAfter matching procedures, the ESS was 24 patients and 12 patients for pegcetacoplan and eculizumab, respectively.
bIndicates variable included in matching procedures. These included age at first infusion of study drug, female, white, Asian, LDH, and history of aplastic anemia.
Source: Sponsor-submitted ITC.7
Table 25: Baseline Characteristics of the PEGASUS and ALXN Studies Before and After Matching — Fatigue and QoL Outcomes
Characteristic | PEGASUS (before matching) | PEGASUS (after matchinga) | ALXN (as reported) | |||
|---|---|---|---|---|---|---|
Pegcetacoplan | Eculizumab | Pegcetacoplan | Eculizumab | Ravulizumab | Eculizumab | |
Male, % | 30.6 | 40.6 | 25.0 | 57.7 | 51.5 | 49.0 |
Female, % | 69.4 | 59.4 | 75.0 | 42.3 | 48.5 | 51.0 |
Age at first infusion of study drug,b mean (SD) years | 49.0 (16.8) | 48.8 (14.0) | 46.4 (16.6) | 48.8 (12.1) | 46.4 (14.4) | 48.8 (14.0) |
White, % | 58.3 | 65.6 | 56.7 | 53.8 | 51.5 | 62.2 |
Asian, % | 13.9 | 15.6 | 14.3 | 34.4 | 23.7 | 19.4 |
African American, % | 5.6 | 0.0 | 5.0 | 0.0 | 5.2 | 3.1 |
Other or multiple races, % | 0.0 | 3.1 | 0.0 | 6.1 | 3.1 | 1.0 |
Not reported, % | 22.2 | 15.6 | 23.9 | 5.7 | 16.5 | 14.3 |
Weight,b mean (SD) kg | 75.2 (19.6) | 73.2 (14.2) | 72.4 (17.4) | 73.4 (11.8) | 72.4 (16.8) | 73.4 (14.6) |
Height, mean (SD) cm | 167.1 (9.7) | 168.8 (7.4) | 166.0 (8.8) | 169.1 (7.5) | 168.3 (10.1) | 168.8 (9.9) |
Time on eculizumab before first study infusion, mean (SD) years | 5.4 (4.4) | 5.1 (3.8) | 5.7 (4.6) | 6.2 (3.7) | 6.0 (3.5) | 5.6 (3.5) |
History of transfusions within 1 year before first dose, % | 72.2 | 71.9 | 74.8 | 71.1 | 13.4 | 12.2 |
Age at PNH diagnosis, mean (SD) years | 40.5 (17.0) | 35.7 (13.4) | 37.7 (16.5) | 36.0 (11.5) | 34.1 (14.4) | 36.8 (14.1) |
Time from PNH diagnosis to consent, mean (SD) years | 8.5 (7.1) | 13.0 (9.8) | 8.6 (7.2) | 12.8 (9.4) | 12.4 (8.4) | 11.9 (9.4) |
LDH,b mean (SD) U/L | 229.0 (57.2) | 203.5 (35.5) | 228.0 (61.6) | 235.2 (35.8) | 228.0 (48.7) | 235.2 (49.7) |
Hemoglobin, mean (SD) g/dL | 8.7 (1.1) | 8.7 (0.8) | 8.8 (1.1) | 8.7 (0.8) | 11.1 (1.8) | 10.9 (1.8) |
History of major adverse vascular events, % | 25.0 | 18.8 | 23.8 | 11.9 | 28.9 | 22.4 |
History of aplastic anemia,b % | 27.8 | 18.8 | 35.1 | 39.8 | 35.1 | 39.8 |
ESS = effective sample size; LDH = lactate dehydrogenase; PNH = paroxysmal nocturnal hemoglobinuria; QoL = quality of life; SD = standard deviation.
aAfter matching procedures, the ESS was 34 patients and 9 patients for pegcetacoplan and eculizumab, respectively.
bIndicates variable included in matching procedures. These included age at first infusion of study drug, weight, LDH, and history of aplastic anemia.
Source: Sponsor-submitted ITC.7
Results
Clinical and Hematological Outcomes
The results for clinical and hematological outcomes are presented in Figure 4. After matching and anchoring on eculizumab, treatment with pegcetacoplan compared with ravulizumab was associated with more transfusion avoidance (adjusted difference = 71.4%; 95% CI, 53.5% to 89.3%), more Hb level stabilization (adjusted difference = 75.5%; 95% CI, 56.4% to 94.6%), more LDH level normalization in the absence of transfusions (adjusted difference = 64.0%; 95% CI, 41.8% to 86.1%), and fewer mean number of units of packed RBCs transfused (adjusted difference = –5.7 units; 95% CI, –7.2 to –4.2 units). There was no difference found in the mean change from baseline in LDH levels (adjusted mean difference = 0.3 U/L; 95% CI, –154.5 U/L to 155.1 U/L), although the CI was relatively wide.
Fatigue and QoL Outcomes
The results for fatigue and QoL outcomes are presented in Figure 5. All reported fatigue and QoL outcomes favoured pegcetacoplan compared with ravulizumab. Treatment with pegcetacoplan was associated with improvements in the adjusted difference in mean change from baseline in fatigue (8.2 points; 95% CI, 3.8 to 12.6 points), global health status (9.6 points; 95% CI, 0.1 to 19.0 points), physical functioning (11.5 points; 95% CI, 3.6 to 19.5 points), and fatigue symptoms (–13.3 points; 95% CI, –23.7 to –3.0 points).
Sensitivity Analyses
The authors noted anchored comparisons based on the unweighted sample before matching and unanchored comparisons that excluded patients randomized to receive eculizumab in both studies were both consistent with the primary analysis, although the data were not reported. The authors also noted the sensitivity analysis that revised the PEGASUS definition of LDH level normalization to match the ALXN definition supported the primary analysis, such that pegcetacoplan was associated with more LDH level normalization irrespective of transfusion status (adjusted difference = 57.4%; 95% CI, 35.3% to 79.5%). The latter data were not reported in detail.
Figure 4: Anchored Comparisons of Clinical and Hematological Outcomes After Matching — PEGASUS Versus ALXN Studies
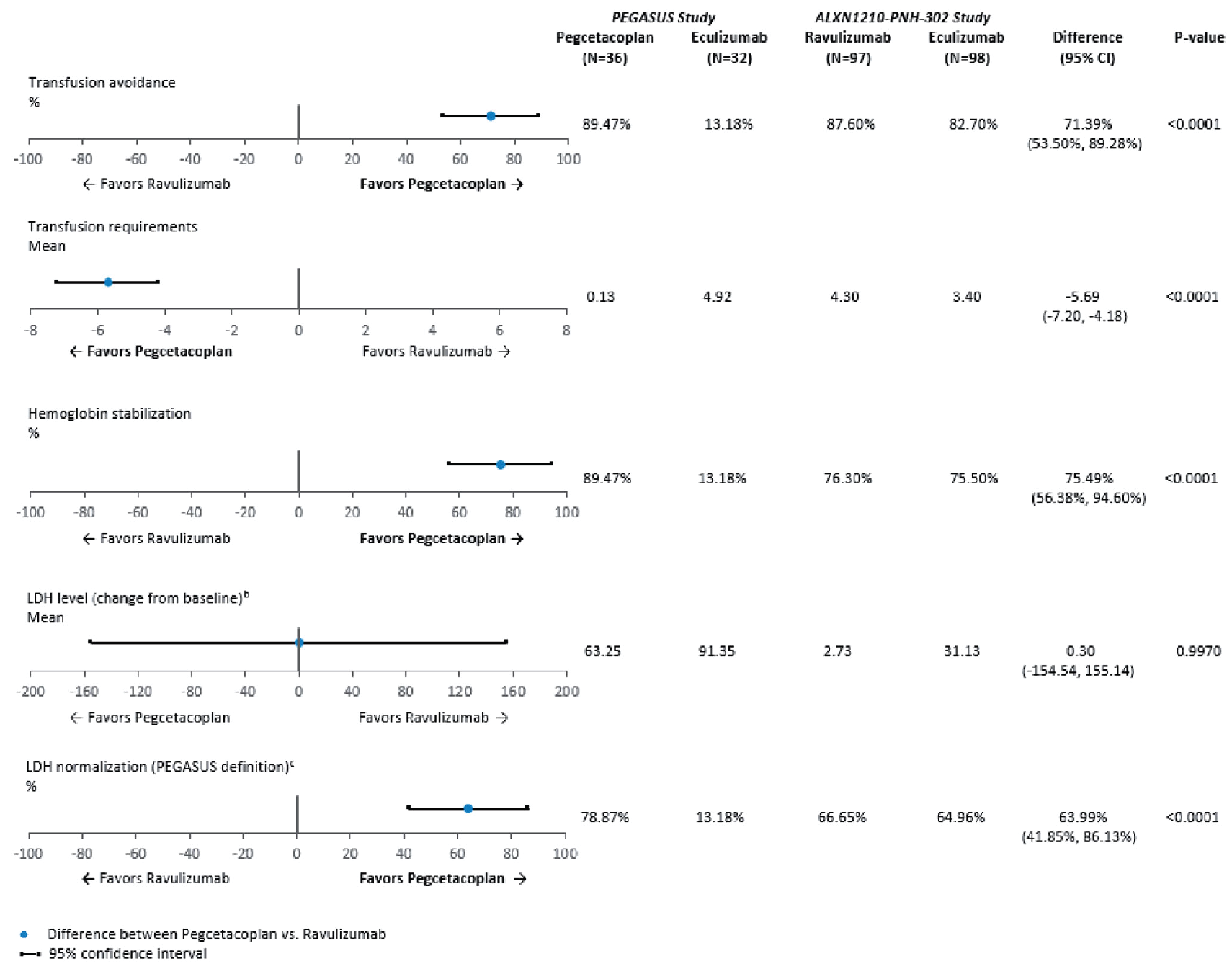
CI = confidence interval; LDH = lactate dehydrogenase.
a Baseline characteristics were matched on age at first infusion of study drug, female, white, Asian, history of aplastic anemia, and LDH.
b Change from baseline in LDH level was examined for week 16 for the ALXN study. During follow-up, LDH level was available at week 16 (day 113) for 94 patients who received ravulizumab and 96 patients who received eculizumab in the ALXN study.
c In the PEGASUS study, LDH level normalization is defined as the proportion of patients who achieved an LDH level ≤ 1 × ULN (226 U/L) in the absence of transfusions from baseline through the end of follow-up. In the ALXN study, LDH level normalization is defined as the proportion of patients who achieved an LDH level ≤ 1 × ULN (246 U/L), with or without transfusions.
Source: Bhak et al. (2021).7 Copyright 2021 Informa UK Limited, Taylor & Francis Group. Reprinted in accordance with Creative Commons Attribution Licence CC BY-NC-ND 4.0.
Figure 5: Anchored Comparisons of Fatigue and QoL Outcomes After Matching — PEGASUS Versus ALXN Studies
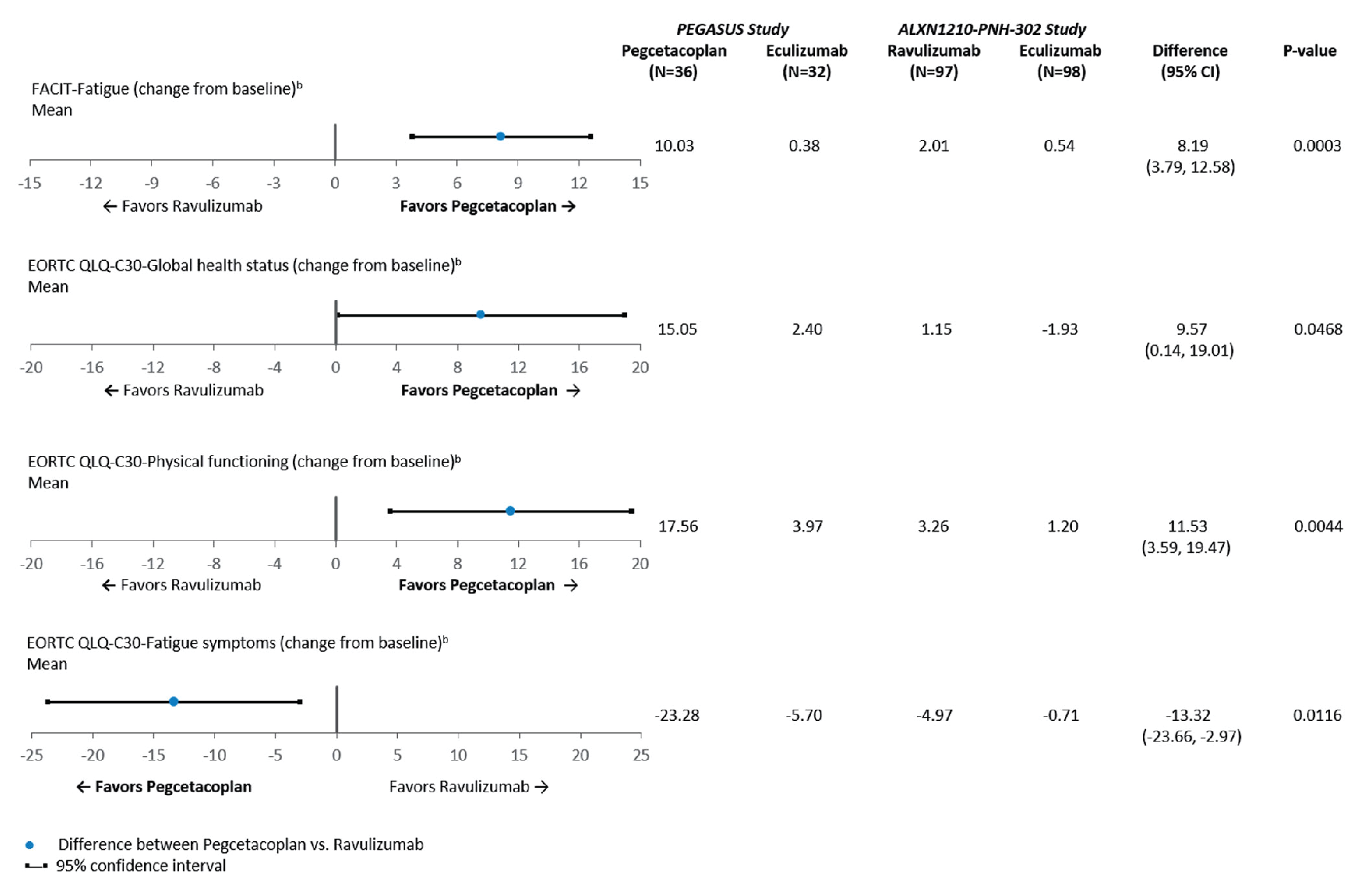
EORTC QLQ-C30 = European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30; FACIT = Functional Assessment of Chronic Illness Therapy; LDH = lactate dehydrogenase; QoL = quality of life.
a Baseline characteristics were matched on age at first infusion of study drug, weight, history of aplastic anemia, and LDH.
b Data were available for 67 of 68 patients in the PEGASUS study.
Source: Bhak et al. (2021).7 Copyright 2021 Informa UK Limited, Taylor & Francis Group. Reprinted in accordance with Creative Commons Attribution Licence CC BY-NC-ND 4.0.
Critical Appraisal of MAIC
The rationale for conducting the MAIC and its objectives were clearly reported. The authors did not report a systematic literature search, describe their methods for data extraction, or conduct quality assessment of the PEGASUS or ALXN studies. Based on CADTH’s systematic literature reviews, it’s unlikely any relevant studies were missed. The MAIC included relevant outcomes identified in the CADTH systematic review protocol, although it’s not clear if the analysis for LDH level normalization in the absence of transfusions was accurately compared across studies (in PEGASUS, LDH level normalization was assessed in the absence of transfusions, whereas in ALXN, it was assessed with or without transfusions), potentially introducing uncertainly into the estimate. Some important outcomes, such as Hb and bilirubin levels and safety, were not available for both studies in the comparison. As such, their comparative assessment remains unknown.
There were several similarities in study design, including the inclusion and exclusion criteria, across the 2 studies, which made the comparison appropriate. However, there were also several differences. The duration of follow-up for the 2 studies were different (i.e.,16 weeks for PEGASUS and 26 weeks for ALXN), which may result in the overestimation or underestimation of outcomes. The following inclusion criteria were not applied to the ALXN study: Hb level less than 10.5 g/dL and ARC greater than ULN at screening. The mean baseline Hb level was 8.7 g/dL the PEGASUS study, compared with approximately 11.0 g/dL the in ALXN study, and the difference in absolute reticulocyte counts is unknown, given that it was not reported in the ALXN study. To improve the similarity between patient samples across the 2 studies, the analysis selectively included patients from the PEGASUS study with an LDH level of 1.5 times the ULN or less at screening and without major adverse vascular events in the 6 months before treatment, reducing patient numbers. There were also differences in the treatment administration schedule, dosing regimen, and treatment modifications, and the 4-week run-in period in the PEGASUS study was not present in the ALXN study. The authors noted that these differences in study design could not be adjusted for in the analysis, which could have introduced residual confounding due unreported or unobserved cross-study differences, although the direction and extent of bias are unclear.
To account for between-study differences in patient baseline characteristics, several relevant effect modifiers were matched in the weighting process, with separate sets of variables used for clinical and hematological outcomes and fatigue and QoL outcomes. These variables were selected based on clinical input regarding biological plausibility. The clinical experts consulted by CADTH agreed that the effect modifiers used in the MAIC were reasonable, although they considered Hb level and history of transfusions as key effect modifiers. Baseline Hb levels were substantially lower in the PEGASUS study than in the ALXN study, and the proportion of patients with recent transfusion was higher in the PEGASUS study (approximately 72.0% versus approximately 12.5%). These 2 effect modifiers were not matched because they would have reduced the ESS below a preset threshold of 50% of eligible patients and because of the presence of extreme patient weights, which might have introduced unmeasurable confounding in the relative treatment effect estimates, although the direction and extent of bias are unclear. To mitigate for some of these limitations, the authors noted that sensitivity analyses using anchored comparisons based on the unweighted sample before matching and unanchored comparisons that included both effect modifiers and prognostic variables were consistent with the primary analysis.
In consideration of the strengths and limitations of the anchored MAIC, there is uncertainty about the relative treatment effect estimates favouring pegcetacoplan over ravulizumab.
Other Relevant Evidence
This section presents results from the long-term extension period of the PEGASUS trial34 included in the sponsor’s submission to CADTH that were considered to address important gaps in the evidence in the systematic review.
The PEGASUS Study: Open-Label Pegcetacoplan Period
The pivotal PEGASUS trial included a 16-week primary evaluation period in which patients received randomized assignment to either pegcetacoplan or eculizumab. Presented in this section are the efficacy and safety outcomes for patients in the open-label pegcetacoplan period, up to 48 weeks after baseline. All patients, regardless of the drug received in the primary evaluation period, transitioned to pegcetacoplan for the extension period.
Methods
After the 16-week randomized controlled period, all patients who continued in the open-label pegcetacoplan period received pegcetacoplan for an additional 32 weeks. Patients who received pegcetacoplan in the randomized controlled period continued to receive pegcetacoplan monotherapy for the whole open-label pegcetacoplan period, whereas patients who received eculizumab in the randomized controlled period first entered a run-in period in which they received pegcetacoplan in addition to eculizumab for 4 weeks, and then transitioned to pegcetacoplan monotherapy for the remainder of the open-label period. All patients then had the option to continue in an ongoing long-term extension study35 or a 12-week follow-up period.
Populations
All patients who entered the extension period were originally enrolled in the randomized controlled period of the PEGASUS study. As such, the inclusion and exclusion criteria are consistent with what is reported in Systematic Review section. Of the 80 patients who received pegcetacoplan or eculizumab in the randomized controlled period, 77 (96.3%) patients entered the open-label pegcetacoplan period. No notable differences in patient baseline demographics or disease characteristics were identified between the open-label period and the randomized controlled period.
Interventions
Patients either maintained their treatment with pegcetacoplan or transitioned from eculizumab to pegcetacoplan. Use of the study drug and concomitant medications was consistent with that in the randomized controlled period.
Outcomes
The change from baseline and change from week 17 to week 48 with respect to Hb level, ARC, LDH level, FACIT-Fatigue Scale score, LASA scores, EORTC QLQ-C30 scores, as well as the number of packed RBC units transfused during the open-label period (week 17 to week 48 and week 20 to week 48) were secondary end points of the PEGASUS trial. A post hoc analysis of transfusion avoidance during the 48-week study period was of interest to this review and was also summarized. Outcome definitions were consistent with those reported in the Systematic Review section.
Harms outcomes were consistent with the primary evaluation period. Briefly, TEAEs, serious TEAEs, withdrawal due to AEs, deaths, thrombotic events, laboratory parameters, and electrocardiograms were assessed.
In the open-label period, all efficacy and harms outcomes were assessed at each study visit (i.e., weeks 17, 18, 20, 22, and 24, and every 4 weeks thereafter until week 48).
Statistical Analysis
There was no comparator arm in the extension period because all patients either maintained or transitioned to treatment with pegcetacoplan. As such, there was no formal statistical testing conducted on the results. All efficacy results presented are for the ITT set of patients who entered the extension period, and descriptive statistics were used to summarize the results rather than the statistical models used in the primary evaluation period. Missing data in the extension period were not imputed. All harms results presented are for the safety set of patients who entered the extension period.
Patient Disposition
A total of 80 patients were treated with either pegcetacoplan (n = 41) or eculizumab (n = 39) in the 16-week randomized controlled period. Of these, 95.1% of patients in the pegcetacoplan arm and all patients in the eculizumab arm entered the extension period. Three patients in the pegcetacoplan to pegcetacoplan group discontinued treatment during the extension period (2 due to AEs, 1 due to physician decision) and 7 patients in the eculizumab to pegcetacoplan group discontinued treatment due to AEs.
Table 26: Patient Disposition in the PEGASUS Study — Open-Label Pegcetacoplan Period
Category or analysis set | PEG-PEG | ECU-PEG |
|---|---|---|
Enrolled in the 16-week randomized controlled period, n | 41 | 39 |
Completed 16-week randomized controlled period, n (%) | 38 (92.7) | 39 (100) |
Entered into 32-week open-label pegcetacoplan period, n (%) | 38 (92.7) | 39 (100) |
Discontinued from study treatment during the 32-week open-label pegcetacoplan period, n (%) | 3 (7.3) | 7 (17.9) |
Adverse events | 2 (4.9) | 7 (17.9) |
Physician’s decision | 1 (2.4) | 0 |
ITT, n (%) | 38 (92.7) | 39 (100) |
Safety, n (%) | 38 (92.7) | 39 (100) |
ECU = eculizumab; ITT = intention to treat; PEG = pegcetacoplan.
Source: PEGASUS Clinical Study Report (48 weeks).36
Exposure to Study Treatments
In the open-label pegcetacoplan period, the mean duration of pegcetacoplan treatment was 209.8 days (SD = 39.2 days). Seven (9.1%) patients had an interruption in at least 1 pegcetacoplan infusion. The mean number of infusions completed per patient was 61.0 (SD = 12.1). Dose escalation of pegcetacoplan (to 1,080 mg every third day) was required in 15 patients (19.5%), including 2 patients who dose-escalated during the randomized controlled period. The mean treatment compliance rate was 100.8% (SD = 6.2%). Treatment exposure in the open-label pegcetacoplan period is summarized in Table 27.
Table 27: Treatment Exposure in the PEGASUS Study — Open-Label Pegcetacoplan Period (Safety Set)
Category or analysis set | PEG-PEG and ECU-PEG combined (N = 77) |
|---|---|
Duration of pegcetacoplan treatment in the open-label period (days), mean (SD) | 209.8 (39.2) |
Patients with interruption in ≥ 1 pegcetacoplan infusion, n (%) | 7 (9.1) |
Number of infusions completed per patient, mean (SD) | 61.0 (12.1) |
Dose escalation required,a n (%) | 15 (19.5) |
Treatment complianceb (%) | |
Mean (SD) | 100.8 (6.2) |
< 80% | 1 (1.3) |
> 80% to ≤ 90% | 1 (1.3) |
> 90% to ≤ 100% | 45 (58.4) |
> 100% to ≤ 120% | 30 (39.0) |
ECU = eculizumab; PEG = pegcetacoplan; SD = standard deviation.
aDose escalation of pegcetacoplan to 1,080 mg every third day.
bCompliance rate = total number of study infusions taken / total number of expected infusions, where the total number of expected infusions was calculated as: (last dose – first dose + 3.5 days) / 7 days) × 2.
Source: PEGASUS Clinical Study Report (48 weeks).36
Efficacy
Survival
Survival was not measured as an efficacy end point in this study. Refer to the Harms section for analysis on death events.
Hb Level
Change in Hb Level
As summarized in Table 28, the mean change from week 17 in Hb level at week 48 (secondary end point) was –0.16 g/dL (SD = 1.15 g/dL) in the pegcetacoplan to pegcetacoplan group and 2.89 g/dL (SD = 2.08 g/dL) g/dL in the eculizumab to pegcetacoplan group. The mean change from baseline in Hb level at week 48 (secondary end point) was 2.47 g/dL (SD = 1.72 g/dL) in the pegcetacoplan to pegcetacoplan group and 2.93 g/dL (SD = 2.09 g/dL) in the eculizumab to pegcetacoplan group. The mean Hb level at each study visit over time (whole study) is shown in Figure 6.
Table 28: Change in Hb Level in the PEGASUS Study — Open-Label Pegcetacoplan Period (ITT; Uncensored for Transfusion)
Hemoglobin | PEG-PEG | ECU-PEG |
|---|---|---|
Open-label pegcetacoplan period (week 17 to week 48) | ||
ITT set in the open-label period analysis, N | 38 | 39 |
Week 17 | ||
n (%) | 37 (97.4) | 38 (97.4) |
Hb level (g/dL), mean (SD) | 11.56 (1.70) | 8.63 (1.18) |
Week 48 | ||
n (%) | 33 (86.6) | 29 (74.4) |
Hb level (g/dL), mean (SD) | 11.30 (1.77) | 11.55 (2.24) |
Change from week 17 in Hb level at week 48 (g/dL), mean (SD) | –0.16 (1.15) | 2.89 (2.08) |
Whole study (baseline to week 48) | ||
ITT set in the whole study analysis, N | 41 | 39 |
Baseline | ||
n (%) | 41 (100.0) | 39 (100.0) |
Hb level (g/dL), mean (SD) | 8.69 (1.08) | 8.68 (0.89) |
Week 48 | ||
n (%) | 33 (80.5) | 30 (76.9) |
Hb level (g/dL), mean (SD) | 11.30 (1.77) | 11.57 (2.21) |
Change from baseline in Hb level at week 48 (g/dL), mean (SD) | 2.47 (1.72) | 2.93 (2.09) |
ECU = eculizumab; Hb = hemoglobin; ITT = intention to treat; PEG = pegcetacoplan; SD = standard deviation.
Note: Baseline was defined as the average of measurements recorded before the first dose of pegcetacoplan.
Source: PEGASUS Clinical Study Report (48 weeks).36
Figure 6: Hb Level Over Time in the PEGASUS Study — Whole Study Analysis (ITT)
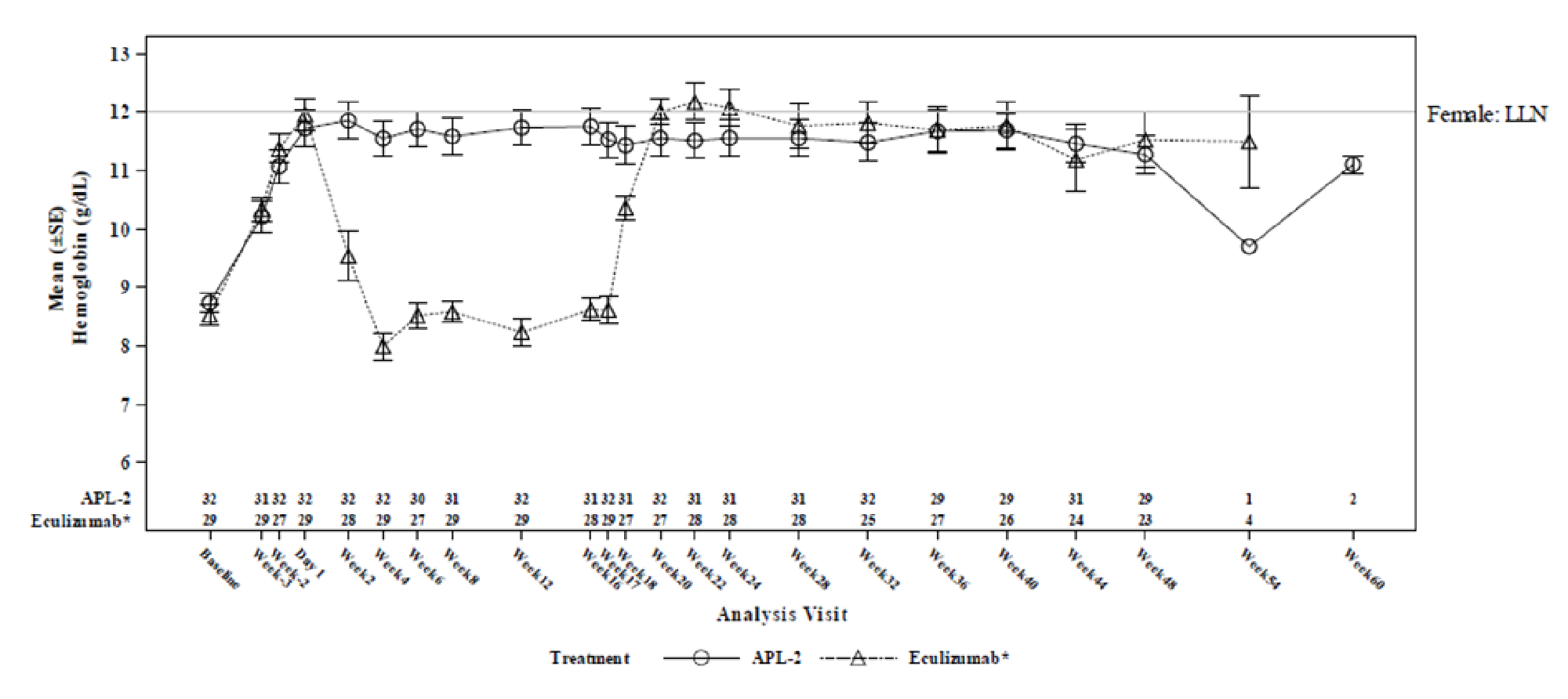
ALP-2 = pegcetacoplan; Hb = hemoglobin; ITT = intention to treat; LLN = lower limit of normal; SE = standard error.
Note: Baseline was defined as the average of measurements recorded before the first dose of pegcetacoplan. Study periods were as follows: run-in period (baseline to day –1); randomized controlled period (day 1 to week 16); open-label pegcetacoplan period (week 17 to week 48); and follow-up period (week 49 to week 60). The eculizumab arm refers to patients who received eculizumab in the randomized controlled period and subsequently completed a 4-week run-in period (week 17 to week 20) with concurrent treatment with eculizumab and pegcetacoplan, followed by pegcetacoplan monotherapy until week 48.
Source: PEGASUS Clinical Study Report (48 weeks).36
Transfusion
Transfusion avoidance (post hoc) and number of packed RBC units transfused analyses are summarized in Table 29.
Transfusion Avoidance (Post Hoc Analysis)
In a post hoc analysis of transfusion avoidance, transfusion avoidance was achieved in 73.2% of patients on pegcetacoplan monotherapy (i.e., baseline to week 48) in the pegcetacoplan to pegcetacoplan group, and in 71.8% of patients on pegcetacoplan monotherapy (i.e., week 17 to week 48) in the eculizumab to pegcetacoplan group from baseline to week 48.
Number of Packed RBC Units Transfused
In the pegcetacoplan to pegcetacoplan group, the mean number of packed RBC units transfused in the open-label pegcetacoplan period (week 17 to week 48; secondary end point) was 1.79 (SD = 4.67) units. In the eculizumab to pegcetacoplan group, the mean number of packed RBC units transfused was 2.82 (SD = 8.30) units in the pegcetacoplan-only period (week 20 to 48).
Table 29: Transfusion Outcomes in the PEGASUS Study — Open-Label Pegcetacoplan Period (ITT)
Transfusion outcomes | PEG-PEG | ECU-PEG |
|---|---|---|
Transfusion avoidance | ||
ITT set in the whole study analysis, n | 41 | 39 |
Patients achieved transfusion avoidance while on pegcetacoplan monotherapy,a n (%) | 30 (73.2) | 28 (71.8) |
Patients receiving transfusion while on pegcetacoplan monotherapy, n (%) | 11 (26.8) | 11 (28.2) |
Received at least 1 transfusion | 7 (17.1) | 10 (25.6) |
Withdrew from the study without having had a transfusion | 4 (9.8) | 1 (2.6) |
Open-label pegcetacoplan period (week 17 to week 48) | ||
ITT set in the open-label period analysis, n | 38 | 39 |
Number of packed RBC units transfused, mean (SD) | ||
Pegcetacoplan and eculizumab run-in period (week 17 to week 20) | NA | 0.36 (0.81) |
Pegcetacoplan monotherapy periodb | 1.79 (4.67) | 2.82 (8.30) |
ECU = eculizumab; ITT = intention to treat; NA = not applicable; PEG = pegcetacoplan; RBC = red blood cell; SD = standard deviation.
aPatients who did not have a transfusion but withdrew before week 48 were considered to have had a transfusion. Pegcetacoplan monotherapy was given from baseline to week 48 in the pegcetacoplan to pegcetacoplan arm, and from week 17 to week 48 in the eculizumab to pegcetacoplan arm.
bThe pegcetacoplan monotherapy period refers to week 17 to week 48 in the pegcetacoplan to pegcetacoplan arm, and week 20 to week 48 in the eculizumab to pegcetacoplan arm.
Source: PEGASUS Clinical Study Report (48 weeks).36
Thrombotic Event
Thrombotic events were not measured as an efficacy end point in this study. Refer to the Harms section for analysis on thrombotic events.
Symptoms of PNH
FACIT-Fatigue Scale
As summarized in Table 30, the mean change from week 17 in FACIT-Fatigue score at week 48 (secondary end point) was 1.3 points (SD = 7.8 points) in the pegcetacoplan to pegcetacoplan group and 10.2 points (SD = 11.0 points) in the eculizumab to pegcetacoplan group. The mean change from baseline in FACIT-Fatigue score at week 48 (secondary end point) was 10.1 points (SD = 9.1 points) points in the pegcetacoplan to pegcetacoplan group and 9.62 points (SD = 10.3 points) in the eculizumab to pegcetacoplan group. The mean FACIT-Fatigue score at each study visit over time is shown Figure 7.
Table 30: Change in FACIT-Fatigue Score in the PEGASUS Trial — Open-Label Pegcetacoplan Period and Whole Study Analyses (ITT; Uncensored for Transfusion)
FACIT-Fatigue Scale | PEG-PEG | ECU-PEG |
|---|---|---|
Open-label pegcetacoplan period (week 17 to week 48) | ||
ITT set in the open-label period analysis, n | 38 | 39 |
Week 17 | ||
n (%) | 38 (100) | 36 (92.3) |
FACIT-Fatigue score, mean (SD) | 39.9 (10.0) | 31.6 (12.3) |
Week 48 | ||
n (%) | 30 (78.9) | 26 (66.7) |
FACIT-Fatigue score, mean (SD) | 40.6 (10.1) | 43.3 (8.4) |
Change from week 17 in FACIT-Fatigue score at week 48, mean (SD) | 1.3 (7.8) | 10.2 (11.0) |
Whole study (baseline to week 48) | ||
ITT set in the whole study analysis, N | 41 | 39 |
Baseline | ||
n (%) | 41 (100) | 38 (97.4) |
FACIT-Fatigue score, mean (SD) | 32.2 (11.4) | 31.6 (12.5) |
Week 48 | ||
n (%) | 30 (73.2) | 29 (74.4) |
FACIT-Fatigue score, mean (SD) | 40.6 (10.1) | 42.52 (8.7) |
Change from baseline in FACIT-Fatigue score at week 48, mean (SD) | 10.1 (9.1) | 9.62 (10.3) |
ECU = eculizumab; FACIT = Functional Assessment of Chronic Illness Therapy; ITT = intention to treat; PEG = pegcetacoplan; SD = standard deviation.
Note: Baseline was defined as the average of measurements recorded before the first dose of pegcetacoplan.
Source: PEGASUS Clinical Study Report (48 weeks).36
Figure 7: FACIT-Fatigue Scale Score Over Time in the PEGASUS Trial — Whole Study Analysis (ITT)
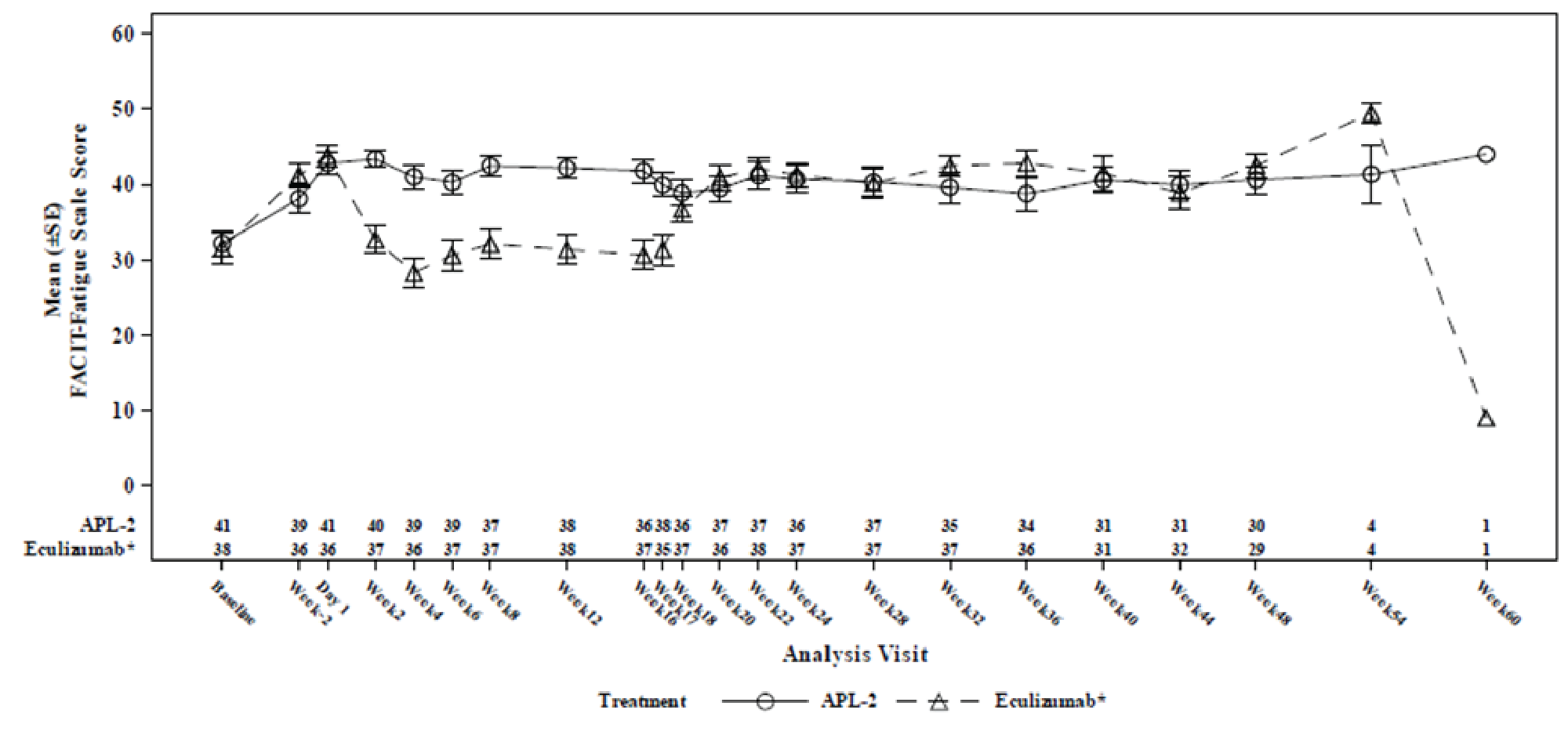
ALP-2 = pegcetacoplan; FACIT = Functional Assessment of Chronic Illness Therapy; ITT = intention to treat; SE = standard error.
Note: Baseline was defined as the average of measurements recorded before the first dose of pegcetacoplan. Study periods were as follows: run-in period (baseline to day –1); randomized controlled period (day 1 to week 16); open-label pegcetacoplan period (week 17 to week 48); and follow-up period (week 49 to week 60). The eculizumab arm refers to patients who received eculizumab in the randomized controlled period and subsequently completed a 4-week run-in period (week 17 to week 20) with concurrent treatment with eculizumab and pegcetacoplan, followed by pegcetacoplan monotherapy until week 48.
Source: PEGASUS Clinical Study Report (48 weeks).36
Health-Related Quality of Life
Linear Analogue Scale Assessment
As summarized in Table 31, the mean change from week 17 in LASA score at week 48 (secondary end point) was 13.1 points (SD = 46.3 points) in the pegcetacoplan to pegcetacoplan group and 62.9 points (SD = 60.1 points) in the eculizumab to pegcetacoplan group. The mean change from baseline in LASA score at week 48 (secondary end point) was 58.7 points (SD = 51.2 points) in the pegcetacoplan to pegcetacoplan group and 56.5 points (SD = 65.6 points) in the eculizumab to pegcetacoplan group. The mean change in LASA score at each study visit over time (whole study) is shown in Figure 10 in Appendix 3.
Table 31: Change in LASA Score in the PEGASUS Trial — Open-Label Pegcetacoplan Period and Whole Study Analyses (ITT; Uncensored for Transfusion)
LASA score | PEG-PEG | ECU-PEG |
|---|---|---|
Open-label pegcetacoplan period (week 17 to week 48) | ||
ITT set in the open-label period analysis, N | 38 | 39 |
Week 17 | ||
n (%) | 38 (100) | 36 (92.3) |
LASA score, mean (SD) | 202.6 (69.7) | 151.1 (60.5) |
Week 48 | ||
n (%) | 30 (78.9) | 26 (66.7) |
LASA score, mean (SD) | 214.0 (66.8) | 221.0 (62.5) |
Change from week 17 in LASA score at week 48, mean (SD) | 13.1 (46.3) | 62.9 (60.1) |
Whole study (baseline to week 48) | ||
ITT set in the whole study analysis, N | 41 | 39 |
Baseline | ||
n (%) | 40 (97.6) | 38 (97.4) |
LASA score, mean (SD) | 161.0 (68.0) | 156.7 (61.3) |
Week 48 | ||
n (%) | 29 (70.7) | 29 (74.4) |
LASA score, mean (SD) | 216.0 (67.1) | 218.4 (60.1) |
Change from baseline in LASA score at week 48, mean (SD) | 58.7 (51.2) | 56.5 (65.6) |
ECU = eculizumab; ITT = intention to treat; LASA = Linear Analogue Scale Assessment; PEG = pegcetacoplan; SD = standard deviation.
Note: Baseline was defined as the average of measurements recorded before the first dose of pegcetacoplan.
Source: PEGASUS Clinical Study Report (48 weeks).36
EORTC QLQ-C30
Change in EORTC QLQ-C30 (week 14 to week 48 and baseline to week 48) in the global health status/QoL score and select symptom subscales (i.e., fatigue, pain, and dyspnea) are summarized in Table 32. The mean score at each visit over time (whole study) for these scales are shown in Figure 11 (global health status/QoL), Figure 12 (fatigue), Figure 13 (pain), and Figure 14 (dyspnea).
In the pegcetacoplan to pegcetacoplan group, mean scores in the open-label period were consistent with those in the randomized controlled period for the global health status/QoL scale and all functional and symptom subscales.
In the eculizumab to pegcetacoplan group at week 48, an improvement (i.e., rise in global in health status and functioning subscale scores; decrease in symptom subscale scores) in mean score of at least 10 points from week 17 was observed for the global health status/QoL scale, as well as for the dyspnea, fatigue, physical functioning, role functioning, and social functioning subscales. For pain and for all other subscales, mean scores in the open-label period were consistent with those in the randomized controlled period.
Overall, there was no notable difference in the change from baseline mean scores at week 48 between the pegcetacoplan to pegcetacoplan group and the eculizumab to pegcetacoplan group for any EORTC QLQ-C30 subscales.
Table 32: Change in EORTC QLQ-C30 Scores in the PEGASUS Trial — Open-Label Pegcetacoplan Period and Whole Study Analyses (ITT; Uncensored for Transfusion)
Interventions | Total N | Baseline scale score | Week 17 scale score | Week 48 scale score | Change from week 17 in score at week 48, LS mean (SD) | Change from baseline in score at week 48, LS mean (SD) | |||
|---|---|---|---|---|---|---|---|---|---|
n (%) | Mean (SD) | n (%) | Mean (SD) | n (%) | Mean (SD) | ||||
Open-label pegcetacoplan period (week 17 to week 48) | |||||||||
Global health status/quality of life | |||||||||
PEG-PEG | 38 | NA | NA | 36 (94.7) | 67.8 (21.8) | 30 (78.9) | 73.1 (20.4) | 7.2 (19.7) | NA |
ECU-PEG | 39 | NA | NA | 38 (97.4) | 50.9 (19.8) | 26 (66.7) | 75.0 (20.5) | 23.1 (22.1) | NA |
Symptom scale: fatigue | |||||||||
PEG-PEG | 38 | NA | NA | 36 (94.7) | 31.3 (26.1) | 30 (78.9) | 31.5 (26.3) | –3.0 (20.8) | NA |
ECU-PEG | 39 | NA | NA | 38 (97.4) | 47.2 (28.0) | 26 (66.7) | 23.9 (25.9) | –23.08 (28.8) | NA |
Symptom scale: pain | |||||||||
PEG-PEG | 38 | NA | NA | 36 (94.7) | 19.7 (28.2) | 30 (78.9) | 20.6 (31.8) | –2.8 (23.2) | NA |
ECU-PEG | 39 | NA | NA | 38 (97.4) | 22.7 (29.0) | 26 (66.7) | 15.4 (20.0) | –5.77 (21.1) | NA |
Symptom scale: dyspnea | |||||||||
PEG-PEG | 38 | NA | NA | 36 (94.7) | 14.9 (21.5) | 30 (78.9) | 18.9 (20.9) | 3.3 (25.3) | NA |
ECU-PEG | 39 | NA | NA | 38 (97.4) | 38.9 (29.3) | 26 (66.7) | 16.7 (21.6) | –19.2 (28.6) | NA |
Whole study (baseline to week 48) | |||||||||
Global health status/quality of life | |||||||||
PEG-PEG | 41 | 41 (100) | 56.3 (20.4) | NA | NA | 30 (73.2) | 73.1 (20.4) | NA | 18.9 (17.6) |
ECU-PEG | 39 | 37 (94.9) | 56.5 (20.2) | NA | NA | 28 (71.8) | 73.5 (19.8) | NA | 14.0 (22.9) |
Symptom scale: fatigue | |||||||||
PEG-PEG | 41 | 41 (100) | 49.6 (29.1) | NA | NA | 30 (73.2) | 31.5 (26.3) | NA | –21.5 (26.7) |
ECU-PEG | 39 | 38 (97.4) | 50.3 (24.7) | NA | NA | 29 (74.4) | 24.5 (24.6) | NA | –23.8 (29.5) |
Symptom scale: pain | |||||||||
PEG-PEG | 41 | 41 (100) | 19.5 (26.8) | NA | NA | 30 (73.2) | 20.6 (31.8) | NA | 0.6 (27.8) |
ECU-PEG | 39 | 38 (97.4) | 15.8 (25.1) | NA | NA | 29 (74.4) | 16.7 (19.4) | NA | 3.5 (20.6) |
Symptom scale: dyspnea | |||||||||
PEG-PEG | 41 | 41 (100) | 33.3 (27.9) | NA | NA | 30 (73.2) | 18.9 (20.9) | NA | –17.8 (30.0) |
ECU-PEG | 39 | 38 (97.4) | 43.9 (32.1) | NA | NA | 29 (74.4) | 17.2 (21.1) | NA | –27.6 (33.4) |
ECU = eculizumab; EORTC QLQ-C30 = European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30; ITT = intention to treat; LS = least squares; NA = not applicable’ PEG = pegcetacoplan; SD = standard deviation.
Note: Baseline was defined as the average of measurements recorded before the first dose of pegcetacoplan.
Source: PEGASUS Clinical Study Report (48 weeks).36
Complications of PNH
Complications of PNH were not assessed in this study.
Intravascular Hemolysis
Change From Baseline in LDH Level
As summarized in Table 33, the change from week 17 in LDH level at week 48 (secondary end point) was 8.0 U/L (SD = 129.28 U/L) in the pegcetacoplan to pegcetacoplan group and –46.84 U/L (SD = 292.61 U/L) in the eculizumab to pegcetacoplan group. The mean change from baseline in LDH level at week 48 (secondary end point) was –41.5 U/L (SD = 153.7 U/L) in the pegcetacoplan to pegcetacoplan group and –105.3 U/L (SD = 315.6 U/L) in the eculizumab to pegcetacoplan group. The change in LDH level over time (whole study) is shown in Figure 8.
Table 33: Change in LDH Level in the PEGASUS Trial — Open-Label Pegcetacoplan Period and Whole Study Analyses (ITT; Uncensored for Transfusion)
LDH level | PEG-PEG | ECU-PEG |
|---|---|---|
Open-label pegcetacoplan period (week 17 to week 48) | ||
ITT set in the open-label period analysis, N | 38 | 39 |
Week 17 | ||
n (%) | 38 (100) | 37 (94.9) |
LDH level (U/L), mean (SD) | 208.5 (121.2) | 258.0 (234.9) |
Week 48 | ||
n (%) | 33 (86.8) | 28 (71.8) |
LDH level (U/L), mean (SD) | 222.67 (141.09) | 230.20 (136.24) |
Change from week 17 in LDH level at week 48 (U/L), mean (SD) | 8.0 (129.28) | –46.8 (292.6) |
Whole study (baseline to week 48) | ||
ITT set in the whole study analysis, N | 41 | 39 |
Baseline | ||
n (%) | 41 (100) | 39 (100) |
LDH level (U/L), mean (SD) | 257.5 (97.7) | 308.6 (284.8) |
Week 48 | ||
n (%) | 33 (80.5) | 30 (76.9) |
LDH level (U/L), mean (SD) | 222.7 (141.1) | 224.1 (133.5) |
Change from baseline in LDH level at week 48 (U/L), mean (SD) | –41.5 (153.7) | –105.3 (315.6) |
ECU = eculizumab; ITT = intention to treat; LDH = lactate dehydrogenase; PEG = pegcetacoplan; SD = standard deviation.
Note: Baseline was defined as the average of measurements recorded before the first dose of pegcetacoplan.
Source: PEGASUS Clinical Study Report (48 weeks).36
Figure 8: LDH Level Over Time in the PEGASUS Trial — Whole Study Analysis (ITT)
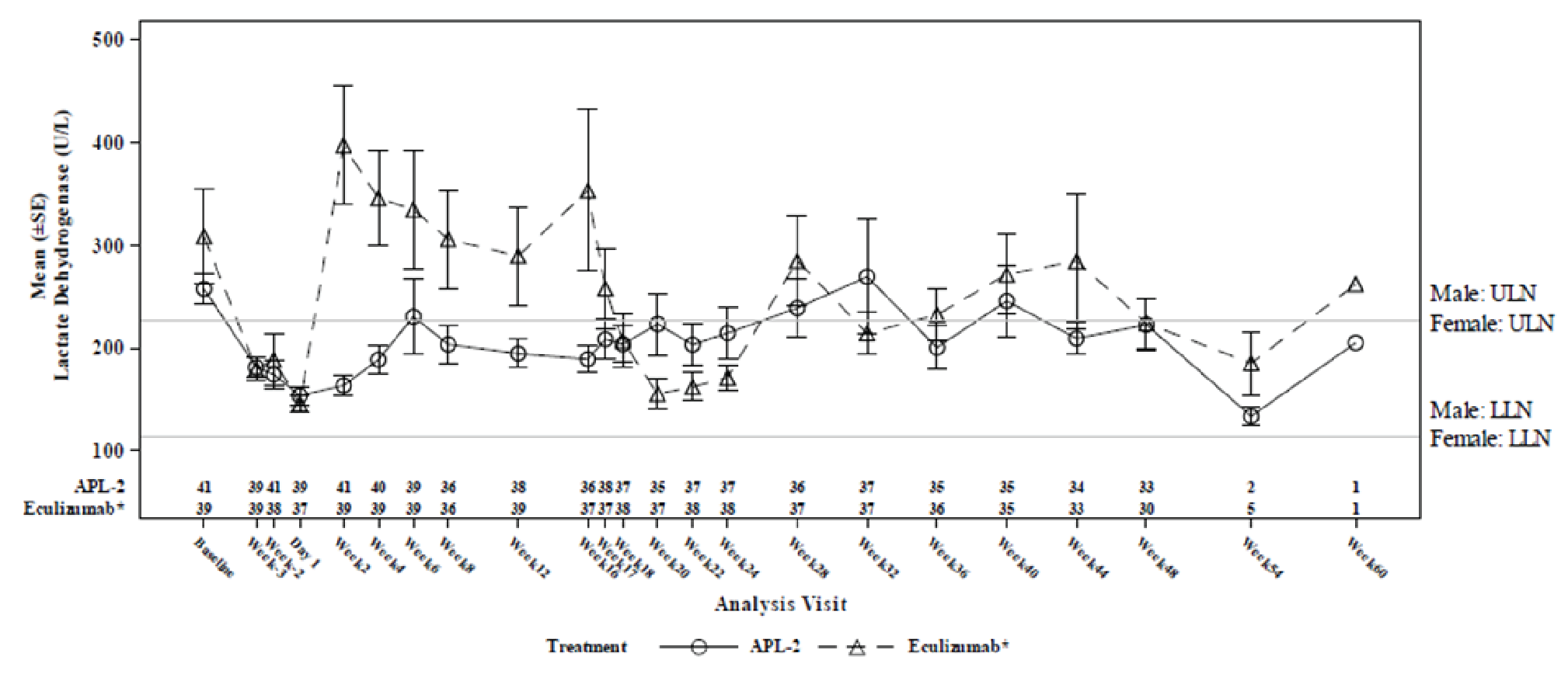
ALP-2 = pegcetacoplan; ITT = intention to treat; LDH = lactate dehydrogenase; LLN = lower limit of normal; SE = standard error; ULN = upper limit of normal.
Note: Baseline was defined as the average of measurements recorded before the first dose of pegcetacoplan. Study periods were as follows: run-in period (baseline to day –1); randomized controlled period (day 1 to week 16); open-label pegcetacoplan period (week 17 to week 48); and follow-up period (week 49 to week 60). The eculizumab arm refers to patients who received eculizumab in the randomized controlled period and subsequently completed a 4-week run-in period (week 17 to week 20) with concurrent treatment with eculizumab and pegcetacoplan, followed by pegcetacoplan monotherapy until week 48.
Source: PEGASUS Clinical Study Report (48 weeks).36
Extravascular Hemolysis
Absolute Reticulocyte Count
As summarized in Table 34, the mean change in ARC level from week 17 to week 48 (secondary end point) was –6.5 × 109 cells/L (SD = 26.5 × 109 cells/L) in the pegcetacoplan to pegcetacoplan group and –121.2 × 109 cells/L (SD = 70.8 × 109 cells/L) in the eculizumab to pegcetacoplan group. The mean change from baseline in ARC at week 48 (secondary end point) was –135.6 × 109 cells/L (SD = 67.9 × 109 cells/L) in the pegcetacoplan to pegcetacoplan group and –128.2 × 109 cells/L (SD = 59.6 × 109 cells/L) in the eculizumab to pegcetacoplan group. The change in ARC level over time (whole study) is shown in Figure 9.
Table 34: Change From Baseline in ARC in the PEGASUS Trial — Open-Label Pegcetacoplan Period and Whole Study Analyses (ITT; Uncensored for Transfusion)
ARC | PEG-PEG | ECU-PEG |
|---|---|---|
Open-label pegcetacoplan period (week 17 to week 48) | ||
ITT set in the open-label period analysis, N | 38 | 39 |
Week 17 | ||
n (%) | 36 (94.7) | 38 (97.4) |
ARC (109 cells/L), mean (SD) | 86.1 (36.5) | 207.6 (84.3) |
Week 48 | ||
n (%) | 31 (81.6) | 29 (74.4) |
ARC (109 cells/L), mean (SD) | 80.0 (26.8) | 94.0 (50.1) |
Change from week 17 in ARC at week 48 (109 cells/L), mean (SD) | –6.5 (26.5) | –121.2 (71.0) |
Whole study (baseline to week 48) | ||
ITT set in the whole study analysis, N | 41 | 39 |
Baseline | ||
n (%) | 41 (100.0) | 39 (100.0) |
ARC (109 cells/L), mean (SD) | 217.5 (75.0) | 216.15 (69.1) |
Week 48 | ||
n (%) | 31 (75.6) | 29 (74.4) |
ARC (109 cells/L), mean (SD) | 80.0 (26.8) | 94.0 (50.1) |
Change from baseline in ARC at week 48 (109 cells/L), mean (SD) | –135.6 (67.9) | –128.2 (59.6) |
ARC = absolute reticulocyte count; ECU = eculizumab; ITT = intention to treat; PEG = pegcetacoplan; SD = standard deviation.
Note: Baseline was defined as the average of measurements recorded before the first dose of pegcetacoplan.
Source: PEGASUS Clinical Study Report (48 weeks).36
Health Care Resource Utilization
This end point was not measured in the study.
Harms
Only those harms identified in the open-label pegcetacoplan period are reported below. Refer to Table 35 for detailed harms data.
Adverse Events
TEAEs were reported in 92.2% of patients, with the most common TEAEs (≥ 10%) being hemolysis, nasopharyngitis, diarrhea, cough, injection site erythema, fatigue, headache, and upper respiratory tract infection.
Serious Adverse Events
Serious TEAEs were reported in 23.4% of patients, with the most common TEAEs being hemolysis, reported in 5 (6.5%) patients, and gastroenteritis, reported in 2 (2.6%) patients.
Figure 9: ARC Over Time in the PEGASUS Trial — Whole Study Analysis (ITT)
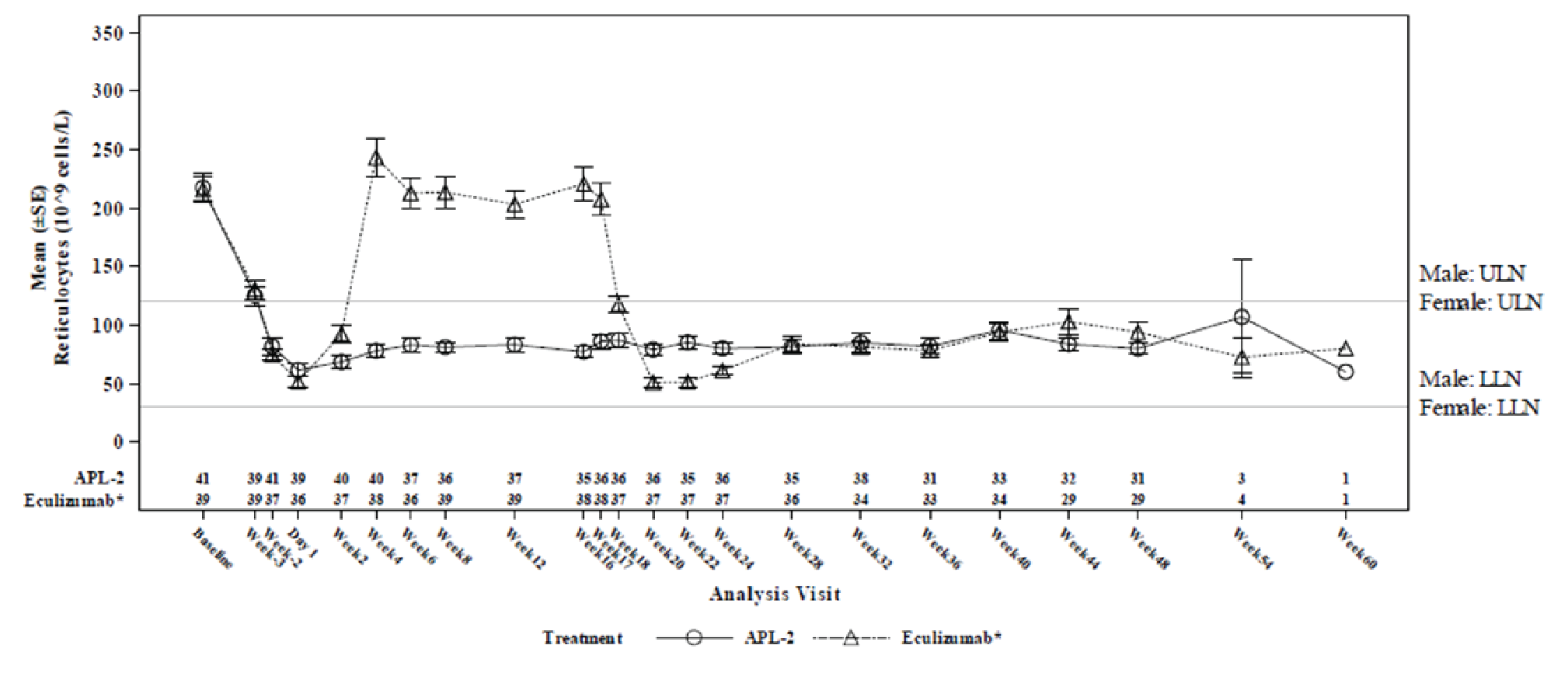
ALP-2 = pegcetacoplan; ARC = absolute reticulocyte count; ITT = intention to treat; LLN = lower limit of normal; SE = standard error; ULN = upper limit of normal.
Note: Baseline was defined as the average of measurements recorded before the first dose of pegcetacoplan. Study periods were as follows: run-in period (baseline to day –1); randomized controlled period (day 1 to week 16); open-label pegcetacoplan period (week 17 to week 48); and follow-up period (week 49 to week 60). The eculizumab arm refers to patients who received eculizumab in the randomized controlled period and subsequently completed a 4-week run-in period (week 17 to week 20) with concurrent treatment with eculizumab and pegcetacoplan, followed by pegcetacoplan monotherapy until week 48.
Source: PEGASUS Clinical Study Report (48 weeks).36
Withdrawals Due to Adverse Events
Nine (11.7%) patients discontinued treatment due to a TEAE of hemolysis (2 patients) and bone marrow failure, hemolytic anemia, acute myeloid leukemia, diffuse large B-cell lymphoma, intestinal ischemia, COVID-19, and hypersensitivity pneumonitis (1 patient each).
Mortality
One death (1.3%) due to COVID-19 infection was reported.
Notable Harms
There were no serious infections known to be caused by an encapsulated organism. Injection site–related TEAE was reported in 20 (26.0%) patients. Injection site erythema (11.7%), induration (6.5%),and pruritis (6.5%) were the most common injection site–related TEAEs. No patient had a positive antipegcetacoplan antibody response. Two (2.6%) patients reported a thrombotic event, neither of which was deemed by the sponsor to be related to pegcetacoplan. One event occurred in the setting of diffuse large B-cell lymphoma, sepsis, and breakthrough hemolysis, and the other occurred in the setting of pneumonia infection and renal failure. There was no report of renal failure and pulmonary hypertension. Hemolysis was reported in 15 (19.5%) patients.
Table 35: Summary of Harms in the PEGASUS Trial — Open-Label Pegcetacoplan Period (Safety Set)
Harms | PEG-PEG and ECU-PEG combined (N = 77) |
|---|---|
Patients with ≥ 1 TEAE | |
n (%) | 71 (92.2) |
Most common events,a n (%) | |
Hemolysis | 15 (19.5) |
Nasopharyngitis | 12 (15.6) |
Diarrhea | 11 (14.3) |
Cough | 9 (11.7) |
Injection site erythema | 9 (11.7) |
Fatigue | 8 (10.4) |
Headache | 8 (10.4) |
Upper respiratory tract infection | 8 (10.4) |
Patients with ≥ 1 serious TEAE | |
n (%) | 18 (23.4) |
Most common events,b n (%) | |
Hemolysis | 5 (6.5) |
Gastroenteritis | 2 (2.6) |
Patients who discontinued treatment due to TEAE | |
n (%) | 9 (11.7) |
Most common events,b n (%) | |
Hemolysis | 2 (2.6) |
Deaths | |
n (%) | 1 (1.3) |
COVID-19 infection | 1 (1.3) |
Notable harms | |
Serious bacterial infection–related TEAE, n (%) | 0 |
Injection site–related TEAE, n (%) | 20 (26.0) |
Most common events,b n (%) | |
Injection site erythema | 9 (11.7) |
Injection site induration | 5 (6.5) |
Injection site pruritis | 5 (6.5) |
Antipegcetacoplan peptide antibody-positive, n (%) | 0 |
Thrombosis, n (%) | 2 (2.6) |
Deep vein thrombosis | 1 (1.3) |
Jugular vein thrombosis | 1 (1.3) |
Pulmonary hypertension, n (%) | NR |
Renal failure, n (%) | 0 |
Hemolysis, n (%) | 15 (19.5) |
ECU = eculizumab; NR = not reported; PEG = pegcetacoplan; TEAE = Treatment-emergent adverse event.
Note: A TEAE was defined as an AE that commenced on or after the first study drug administration or an AE that increased in severity after pretreatment.
aFrequency > 10%.
bOccurred in ≥ 2 patients.
Critical Appraisal
Internal Validity
The single-arm extension period of the PEGASUS trial provided evidence of the efficacy and safety of pegcetacoplan in patients with PNH who had inadequate response to eculizumab administered for up to 48 weeks. The noncomparative study design is a key limitation of the analysis. The lack of comparators precludes conclusions about the comparative efficacy of pegcetacoplan and eculizumab beyond the 16-week randomized controlled period. There is no control for confounding factors in a nonrandomized study, so it is difficult to determine whether the observed effects can be attributed to pegcetacoplan alone. There was no formal statistical testing, and results were presented using descriptive statistics. The open-label design can also introduce uncertainties about subjective outcomes (FACIT-Fatigue, LASA, EORTC QLQ-C30, harms), as reporting of outcomes could be influenced by knowledge of treatment assignment.
External Validity
Much of the appraisal of the RCT with respect to population generalizability also applies to the extension period. In addition, although the study results seem to suggest that the efficacy and safety of pegcetacoplan can be maintained over the long-term, outcomes such as breakthrough hemolysis, survival, thrombosis, and other complications of PNH require a much longer duration of follow-up before firm conclusions can be drawn, given that PNH is a chronic condition.
Discussion
Summary of Available Evidence
This report summarizes the evidence for pegcetacoplan in the treatment of PNH based on 1 phase III RCT with a single-arm extension period and 1 ITC.
One study, PEGASUS, met the inclusion criteria for the Systematic Review section. PEGASUS was a phase III, open-label, RCT that aimed to demonstrate the superiority of pegcetacoplan to eculizumab in adult patients with PNH who continued to have Hb levels less than 10.5 g/dL, despite treatment with eculizumab (N = 80) at a stable dose for at least 3 months. In the 16-week randomized controlled period, the change from baseline at week 16 in Hb (primary end point), transfusion avoidance, and change in baseline at week 16 in ARC, LDH, and FACIT-Fatigue score (key secondary end points) were assessed. All patients had adequate bone marrow function and no history of bone marrow transplant or hereditary complement deficiency. At baseline, the mean age was 48.8 years (SD = 16.0 years), and the majority of patients were female and white. Patients had received eculizumab for a mean duration of approximately 5 years before the study, and 30% of them received eculizumab at a dose higher than the maintenance dose approved for PNH by Health Canada. The study population had a mean Hb level of 8.7 g/dL (SD = 1.0 g/dL), reasonable control of IVH (mean LDH less than 1.5 × ULN), elevated mean ARC and bilirubin levels, and low haptoglobin levels, consistent with the signs of EVH.
Safety and efficacy results from the 32-week single-arm extension period for the PEGASUS trial (N = 77), during which all patients received pegcetacoplan, were also submitted by the sponsor and are presented in this report.
One sponsor-submitted ITC was presented in this report. Given the lack of direct comparative evidence between pegcetacoplan and ravulizumab, the sponsor performed an anchored MAIC to evaluate the relative efficacy of pegcetacoplan to ravulizumab in adult patients with PNH previously treated with eculizumab. The analysis was informed by patient-level data from the PEGASUS study, comparing pegcetacoplan and eculizumab, and aggregate patient data from the ALXN study, comparing ravulizumab and eculizumab. Outcomes analyzed were transfusion avoidance, number of packed RBCs transfused, Hb level stabilization, change from baseline in LDH level, LDH level normalization, fatigue and fatigue symptoms, global health status, and physical functioning. After matching, 68 patients from the PEGASUS study and 195 patients from the ALXN study were included.
Interpretation of Results
Efficacy
Evidence from the pivotal, phase III PEGASUS trial supported the superiority of pegcetacoplan to eculizumab with respect to the primary end point of change from baseline in Hb level at week 16 in adult patients with PNH who had an inadequate response to eculizumab, addressing a key treatment outcome noted by both patients and clinicians. The clinical experts consulted by CADTH considered the benefits of pegcetacoplan on Hb to be clinically meaningful. It has been proposed that Hb level is a strong surrogate biomarker for clinical benefits in patients with sickle cell disease receiving stem cell or gene therapy,24,25 but evidence for PNH is scarce due to rarity of the disease. The sponsor noted that thromboembolic events have been linked to hemolysis in this patient population, potentially through the buildup of cell-free plasma Hb.14,37-41 Nonetheless, it is challenging to draw conclusions about long-term outcomes (e.g., thrombosis, pulmonary hypertension, chronic kidney disease) based on Hb findings because data relating the 2 are limited.
Transfusion dependence poses a significant treatment burden to patients and is associated with AEs and poor QoL, per clinician input. Transfusion avoidance was much more frequently observed in the pegcetacoplan arm than in the eculizumab arm; however, only noninferiority between treatments was concluded because superiority was not tested, in accordance with the hierarchal testing procedure. The clinical experts considered the results to be supportive of the benefit of pegcetacoplan in improving anemia, given that transfusion needs are dependent on Hb level.
Fatigue, the most commonly reported anemia-related symptom of PNH, was assessed using the key secondary end point of change from baseline at week 16 in FACIT-Fatigue score. The analysis showed that patients receiving pegcetacoplan had more improvement in mean fatigue scores from baseline at week 16 than patients receiving eculizumab; however, no conclusions on noninferiority or superiority could be drawn because such statistical tests were not conducted, per the hierarchal testing procedure. Results were in favour of pegcetacoplan with respect to the LASA scale and the EORTC QLQ-C30 global health status/QoL and fatigue subscales, but not with respect to other symptom scales relevant to PNH, such as pain and dyspnea. However, it should be noted that these HRQoL outcomes are subject to a risk of type I error due to the lack of adjustment for multiplicity and a risk of reporting bias due to the open-label design and a large amount of missing data due to censoring for transfusion. Because these patient-reported outcome instruments are not used routinely in clinical practice and have not been validated in patients with PNH, it is impossible to draw any definitive conclusions regarding the effect of pegcetacoplan on fatigue and HRQoL. Nonetheless, based on their clinical experience, the clinical experts expected that the mean change in Hb observed in the pegcetacoplan arm would result in noticeable improvements in fatigue, activities of daily living, and HRQoL in patients.
Hemolytic markers, used by clinicians to assess disease activity, were measured in the study as supportive evidence for the control of intravascular and extravascular hemolysis. The study could not demonstrate noninferiority between the interventions with respect to the key secondary end point of change from baseline at week 16 in LDH level, a biomarker of IVH. A goal of treatment for PNH is to achieve sustained LDH control, per clinician input. Given that IVH was reasonably controlled at baseline, as expected with prior eculizumab treatment, and remained so at week 16 in both groups, the clinical experts were not concerned about the inability to conclude noninferiority regarding change in LDH. Noninferiority with respect to the key secondary end point of change from baseline at week 16 in ARC, an indicator of EVH, was established between the interventions. Despite a more notable reduction in ARC from baseline in the pegcetacoplan arm than in the eculizumab arm, no conclusion of superiority could be drawn because such test was not conducted, per the hierarchal testing procedure. Results of the indirect bilirubin analysis were also in favour of pegcetacoplan. Although results of the haptoglobin analysis did not favour pegcetacoplan, the clinical experts expressed no concerns because haptoglobin is a poor measure of treatment response. In addition, results of the indirect bilirubin and haptoglobin analyses should be interpreted with caution because of the increased risk of type I error due to the lack of control for multiplicity. In consultation with the clinical experts, the CADTH review team considered that the overall evidence related to hemolytic markers supports pegcetacoplan as an effective treatment for controlling both intravascular and extravascular hemolysis.
Responder analyses were conducted for the primary and key secondary outcomes, including Hb response, Hb normalization, LDH normalization, and ARC normalization in the absence of transfusion, as well as for FACIT-Fatigue score. Results were in favour of pegcetacoplan across all analyses, with numerically higher proportions of patients achieving response or normalization with pegcetacoplan than with eculizumab. However, the difference between treatment arms has not been tested for statistical significance and should be interpreted with caution. Further, the FACIT-Fatigue responder analysis was subject to some uncertainties because an MID estimate has not been established in the study population of patients with C5 inhibitor experience.
No definitive conclusion can be drawn with respect to long-term outcomes such as survival, breakthrough hemolysis, thrombosis, and other complications of PNH, or with respect to health care utilization because these outcomes were not evaluated in the efficacy analysis. Although they were included in the harms analysis (except health care utilization), results should be considered exploratory in the absence of formal hypothesis testing. The use of an Hb level of less than 10.5 g/dL as the cut-off for study eligibility, the exclusion of patients with certain cardiovascular diseases or receiving QT-prolonging medications, and the absence of evidence in patients with intolerance to eculizumab introduced some uncertainties to the generalizability of the study findings, although unlikely to be significant.
Results of the open-label extension period of the PEGASUS trial suggested that the benefits of pegcetacoplan with respect to Hb, transfusion avoidance, fatigue, HRQoL, and hemolytic parameters (LDH and ARC) were sustained through week 48. However, the noncomparative design of the extension period meant that the comparative efficacy of pegcetacoplan and eculizumab beyond the 16-week randomized controlled period could not be established, which is a limitation given the chronic nature of the disease.
In the sponsor-submitted anchored MAIC, after matching and anchoring on eculizumab, treatment with pegcetacoplan, compared with ravulizumab, was associated with more transfusion avoidance, more Hb level stabilization, more LDH level normalization in the absence of transfusions, and fewer units of packed RBCs transfused. In addition, treatment with pegcetacoplan, compared with ravulizumab, was associated with improvements in fatigue, global health status, physical functioning, and fatigue symptoms. There was no difference in mean change from baseline in LDH levels.
The anchored MAIC had several strengths and limitations. Strengths included anchored comparisons, which used a common comparator, eculizumab; relevant effect modifiers, which were matched in the weighting process separately for clinical and hematological outcomes and fatigue and QoL outcomes; and consistency in results, which were noted in sensitivity analyses that used anchored comparisons based on the unweighted sample before matching and unanchored comparisons that included both effect modifiers and prognostic variables. Limitations included the lack of matching of 2 clinically important effect modifiers (Hb level and history of transfusions) and the heterogeneity between studies regarding duration of follow-up (i.e.,16 weeks for the PEGASUS study versus 26 weeks for the ALXN study), treatment administration schedule, and dosing regimen. Given the strengths and limitations of the anchored MAIC, there is uncertainty in the relative treatment effect estimates that favour pegcetacoplan over ravulizumab.
Harms
In the randomized controlled period of the PEGASUS trial, the most common TEAEs associated with pegcetacoplan (reported in at least 20% of patients) were injection site–related reactions and diarrhea, both of which were more commonly reported in the pegcetacoplan arm than the eculizumab arm, but none were serious or led to treatment discontinuation. Compared with eculizumab, pegcetacoplan was associated with a notably lower incidence of headache and breakthrough hemolysis. Breakthrough hemolysis resulted in dose escalation and discontinuation of pegcetacoplan in 2 (4.9%) and 3 (7.3%) patients, respectively. The occurrence of AEs and serious AEs during the extension period was similar to that during the randomized controlled period. The study, overall, had no reports of serious infection caused by encapsulated bacteria. Two patients in the pegcetacoplan arm experienced thrombosis during the open-label period, but neither case was deemed by the sponsor to be related to pegcetacoplan.
The clinical experts noted that most AEs associated with pegcetacoplan were observed in clinical practice were mild, with the exception of hemolysis, which has the potential to become serious. Nonetheless, the experts considered the lower incidence of breakthrough hemolysis with pegcetacoplan compared with eculizumab to be favourable. To gain certainty in the incidence of breakthrough hemolysis and dose escalation, a longer duration of follow-up, of at least 2 years, and a larger sample size would be required to adequately account for breakthrough hemolysis due to situational events (e.g., infection, surgery, pregnancy, vaccination), according to the clinical experts. The same also applies to long-term outcomes such as thrombosis and survival. A long-term extension study of patients who previously completed a pegcetacoplan study that aims to provide safety data for up to 2 years is ongoing and results will be forthcoming.35
Conclusions
In the PEGASUS trial, pegcetacoplan demonstrated a clinically meaningful improvement in Hb level compared with eculizumab in a 16-week randomized controlled period in a study population that was representative of patients with PNH who had clinically significant anemia, despite an adequate trial of eculizumab, and had signs of EVH. The evidence strongly suggested that Hb improvement translated into an improvement in transfusion avoidance with pegcetacoplan over eculizumab. Results also suggested that pegcetacoplan could reduce fatigue compared with eculizumab, but the magnitude of benefit and its clinical relevance is uncertain. Analyses of hemolytic markers lent support to the ability of pegcetacoplan to reduce EVH and maintain IVH control. The benefits of pegcetacoplan were sustained through week 48 in the extension period, but the single-arm design precludes conclusions on the comparative efficacy between pegcetacoplan and eculizumab beyond week 16. The sponsor-submitted MAIC assessing the comparative efficacy of pegcetacoplan with ravulizumab showed transfusion avoidance, number of packed RBCs transfused, Hb level stabilization, LDH level normalization, fatigue and fatigue symptoms, global health status, and physical functioning in favour of pegcetacoplan; however, the findings should be interpreted with caution, given the lack of matching of some effect modifiers and the heterogeneity in study designs and populations. The safety profile of pegcetacoplan was generally similar to that of eculizumab, although there was a higher incidence of nonserious injection site–related reactions and diarrhea. A longer duration of follow-up is required to provide certainty on the incidence of breakthrough hemolysis, thrombosis, and survival with pegcetacoplan treatment.
References
1.Patriquin CJ, Kiss T, Caplan S, et al. How we treat paroxysmal nocturnal hemoglobinuria: a consensus statement of the Canadian PNH Network and review of the national registry. Eur J Haematol. 2019;102(1):36-52. PubMed
2.Brodsky RA. Paroxysmal nocturnal hemoglobinuria. Blood. 2014;124(18):2804-2811. PubMed
3.Hill A, Platts P, Smith A, Richards SJ, Cullen M. The incidence and prevalence of paroxysmal nocturnal hemoglobinuria (PNH) and survival of patients in Yorkshire [abstract]. Presented at: 64th ASH Annual Meeting; New Orleans, LA; Dec 10-13, 2022. Blood. 2006;108(11):985. https://www.sciencedirect.com/science/article/pii/S0006497118424251. Accessed 2022 Nov 8.
4.PrEmpaveliTM (pegcetacoplan): solution for infusion: 54 mg/mL solution for infusion for subcutaneous use [draft product monograph]. Stockholm (SE): Swedish Orphan Biovitrum AB; 2022.
5.Hillmen P, Szer J, Weitz I, et al. Pegcetacoplan versus eculizumab in paroxysmal nocturnal hemoglobinuria. N Engl J Med. 2021;384(11):1028-1037. PubMed
6.Clinical Study Report: APL2-302. A phase 3, randomized, multicenter, open-label, active-comparator controlled study to evaluate the efficacy and safety of pegcetacoplan in patients with paroxysmal nocturnal hemoglobinuria (PNH) week 16 analysis [internal sponsor's report]. Waltham (MA): Apellis Pharmaceuticals; 2020.
7.Bhak RH, Mody-Patel N, Baver SB, et al. Comparative effectiveness of pegcetacoplan versus ravulizumab in patients with paroxysmal nocturnal hemoglobinuria previously treated with eculizumab: a matching-adjusted indirect comparison. Curr Med Res Opin. 2021;37(11):1913-1923. PubMed
8.Kulasekararaj AG, Hill A, Rottinghaus ST, et al. Ravulizumab (ALXN1210) vs eculizumab in C5-inhibitor-experienced adult patients with PNH: the 302 study. Blood. 2019;133(6):540-549. PubMed
9.Brodsky RA. Clinical manifestations and diagnosis of paroxysmalnocturnal hemoglobinuria. In: Post TW, ed. UpToDate. Waltham (MA): UpToDate; 2022: https://www.uptodate.com. Accessed 2022 Sep 14.
10.Nishimura JI, Kanakura Y, Ware RE, et al. Clinical course and flow cytometric analysis of paroxysmal nocturnal hemoglobinuria in the United States and Japan. Medicine (Baltimore). 2004;83(3):193-207. PubMed
11.Socie G, Mary JY, de Gramont A, et al. Paroxysmal nocturnal haemoglobinuria: long-term follow-up and prognostic factors. French Society of Haematology. Lancet. 1996;348(9027):573-577. PubMed
12.Kelly RJ, Hill A, Arnold LM, et al. Long-term treatment with eculizumab in paroxysmal nocturnal hemoglobinuria: sustained efficacy and improved survival. Blood. 2011;117(25):6786-6792. PubMed
13.DeZern AE, Dorr D, Brodsky RA. Predictors of hemoglobin response to eculizumab therapy in paroxysmal nocturnal hemoglobinuria. Eur J Haematol. 2013;90(1):16-24. PubMed
14.Risitano AM, Marotta S, Ricci P, et al. Anti-complement treatment for paroxysmal nocturnal hemoglobinuria: time for proximal complement inhibition? A position paper from the SAAWP of the EBMT. Front Immunol. 2019;10:1157. PubMed
15.Brodsky RA. Treatment and prognosis of paroxysmal nocturnalhemoglobinuria. In: Post TW, ed. UpToDate. Waltham (MA): UpToDate; 2022: https://www.uptodate.com. Accessed 2022 Nov 08.
16.Cancado RD, Araujo ADS, Sandes AF, et al. Consensus statement for diagnosis and treatment of paroxysmal nocturnal haemoglobinuria. Hematol Transfus Cell Ther. 2021;43(3):341-348. PubMed
17.Hillmen P, Muus P, Roth A, et al. Long-term safety and efficacy of sustained eculizumab treatment in patients with paroxysmal nocturnal haemoglobinuria. Br J Haematol. 2013;162(1):62-73. PubMed
18.PrSoliris® (eculizumab): 30 mL parenteral solution (10 mg/mL) [product monograph]. Zurich (CH): Alexion Pharma GmbH; 2021 Mar 25: https://pdf.hres.ca/dpd_pm/00060546.PDF. Accessed 2022 Nov 08.
19.PrUltomiris™ (ravulizumab): 10 mg/mL concentrate for solution for infusion [product monograph]. Zurich (CH): Alexion Pharma GmbH; 2021 Sep 22: https://pdf.hres.ca/dpd_pm/00063038.PDF. Accessed 2022 Nov 08.
20.McGowan J, Sampson M, Salzwedel DM, Cogo E, Foerster V, Lefebvre C. PRESS Peer Review of Electronic Search Strategies: 2015 guideline statement. J Clin Epidemiol. 2016;75:40-46. PubMed
21.Grey matters: a practical tool for searching health-related grey literature. Ottawa (ON): CADTH; 2019: https://www.cadth.ca/grey-matters. Accessed 2022 Sep 20.
22.Center for Drug Evaluation Research. Integrated review: Empaveli (pegcetacoplan) for subcutaneous infusion. Company: Apellis Pharamceuticals, Inc. Application No.:NDA 215014. Approval date: 05/14/2021 (FDA approval package). Pegcetacoplan (Empaveli) Subcutaneous infusion. . Silver Spring (MD): U.S. Food and Drug Administration (FDA); 2021: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2021/215014Orig1s000IntegratedR.pdf. Accessed 2022 Oct 12.
23.Cella D, Sarda SP, Hsieh R, et al. Changes in hemoglobin and clinical outcomes drive improvements in fatigue, quality of life, and physical function in patients with paroxysmal nocturnal hemoglobinuria: post hoc analyses from the phase III PEGASUS study. Ann Hematol. 2022;101(9):1905-1914. PubMed
24.Farrell AT, Panepinto J, Desai AA, et al. End points for sickle cell disease clinical trials: renal and cardiopulmonary, cure, and low-resource settings. Blood Adv. 2019;3(23):4002-4020. PubMed
25.Lehrer-Graiwer J, Yokoshima L, Tong B, Love TW. Accelerated approval of Oxbryta® (voxelotor): a case study on novel endpoint selection in sickle cell disease. Contemp Clin Trials. 2020;98:106161. PubMed
26.Weitz I, Meyers G, Lamy T, et al. Cross-sectional validation study of patient-reported outcomes in patients with paroxysmal nocturnal haemoglobinuria. Intern Med J. 2013;43(3):298-307. PubMed
27.Cella D, Johansson P, Ueda Y, et al. Clinically important difference for the FACIT-fatigue scale in paroxysmal nocturnal hemoglobinuria: a derivation from international PNH registry patient data [poster]. Presented at: BSH 2022 ASM; Manchester, UK; Apr 3-5, 2022. 2022: https://www.postersessiononline.eu/173580348_eu/congresos/BSH2022/aula/-PO_96_BSH2022.pdf. Accessed 2022 Oct 16.
28.Locke DE, Decker PA, Sloan JA, et al. Validation of single-item linear analog scale assessment of quality of life in neuro-oncology patients. J Pain Symptom Manage. 2007;34(6):628-638. PubMed
29.Barcellini W, Fattizzo B. Clinical applications of hemolytic markers in the differential diagnosis and management of hemolytic anemia. Dis Markers. 2015;2015:635670. PubMed
30.Jang JH, Kim JS, Yoon SS, et al. Predictive factors of mortality in population of patients with paroxysmal nocturnal hemoglobinuria (PNH): results from a Korean PNH registry. J Korean Med Sci. 2016;31(2):214-221. PubMed
31.Lee JW, Jang JH, Kim JS, et al. Clinical signs and symptoms associated with increased risk for thrombosis in patients with paroxysmal nocturnal hemoglobinuria from a Korean Registry. Int J Hematol. 2013;97(6):749-757. PubMed
32.Shih AW, McFarlane A, Verhovsek M. Haptoglobin testing in hemolysis: measurement and interpretation. Am J Hematol. 2014;89(4):443-447. PubMed
33.Statistical Analysis Plan: Study 302. A phase 3, randomized, multicenter, open-label, active-comparator controlled study to evaluate the efficacy and safety of ALP-2 in patients with paroxysmal nocturnal hemoglobinuria (PNH) week 16 analysis [internal sponsor's report]. Waltham (MA): Apellis Pharmaceuticals; 2019.
34.de Latour RP, Szer J, Weitz IC, et al. Pegcetacoplan versus eculizumab in patients with paroxysmal nocturnal haemoglobinuria (PEGASUS): 48-week follow-up of a randomised, open-label, phase 3, active-comparator, controlled trial. Lancet Haematol. 2022;9(9):e648-e659. PubMed
35.Hoffman K, Machaidze Z, Yeh M, Weitz IC. Evaluation of the long-term safety and efficacy of pegcetacoplan treatment for paroxysmal nocturnal hemoglobinuria patients: an extension study [abstract]. Presented at: 63rd ASH Annual Meeting; Atlanta, GA; Dec 11-14, 2021. Blood. 2021;138(Suppl 1):2175. 10.1182/blood-2021-148042. Accessed 2022 Nov 8.
36.Clinical Study Report: APL2-302. A phase 3, randomized, multicenter, open-label, active-comparator controlled study to evaluate the efficacy and safety of pegcetacoplan in patients with paroxysmal nocturnal hemoglobinuria (PNH) week 48 analysis [internal sponsor's report]. Waltham (MA): Apellis Pharmaceuticals; 2021.
37.Audebert HJ, Planck J, Eisenburg M, Schrezenmeier H, Haberl R. Cerebral ischemic infarction in paroxysmal nocturnal hemoglobinuria report of 2 cases and updated review of 7 previously published patients. J Neurol. 2005;252(11):1379-1386. PubMed
38.Gralnick HR, Vail M, McKeown LP, et al. Activated platelets in paroxysmal nocturnal haemoglobinuria. Br J Haematol. 1995;91(3):697-702. PubMed
39.Hall C, Richards S, Hillmen P. Primary prophylaxis with warfarin prevents thrombosis in paroxysmal nocturnal hemoglobinuria (PNH). Blood. 2003;102(10):3587-3591. PubMed
40.Rosse WF, Nishimura J. Clinical manifestations of paroxysmal nocturnal hemoglobinuria: present state and future problems. Int J Hematol. 2003;77(2):113-120. PubMed
41.Rother RP, Bell L, Hillmen P, Gladwin MT. The clinical sequelae of intravascular hemolysis and extracellular plasma hemoglobin: a novel mechanism of human disease. JAMA. 2005;293(13):1653-1662. PubMed
42.Aaronson NK, Ahmedzai S, Bergman B, et al. The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst. 1993;85(5):365-376. PubMed
43.Georgakopoulos A, Kontodimopoulos N, Chatziioannou S, Niakas D. EORTC QLQ-C30 and FACT-Lym for the assessment of health-related quality of life of newly diagnosed lymphoma patients undergoing chemotherapy. Eur J Oncol Nurs. 2013;17(6):849-855. PubMed
44.Osoba D, Rodrigues G, Myles J, Zee B, Pater J. Interpreting the significance of changes in health-related quality-of-life scores. J Clin Oncol. 1998;16(1):139-144. PubMed
45.Cella D. The Functional Assessment of Cancer Therapy-Anemia (FACT-An) scale: a new tool for the assessment of outcomes in cancer anemia and fatigue. Semin Hematol. 1997;34(3 Suppl 2):13-19. PubMed
46.Chandran V, Bhella S, Schentag C, Gladman DD. Functional assessment of chronic illness therapy-fatigue scale is valid in patients with psoriatic arthritis. Ann Rheum Dis. 2007;66(7):936-939. PubMed
47.Cançado RD, Araújo ADS, Sandes AF, et al. CONSensus statement for diagnosis and treatment of paroxysmal nocturnal haemoglobinuria. Hematol Transfus Cell Ther. 2021;43(3):341-348. PubMed
48.Marchand A, Galen RS, Van Lente F. The predictive value of serum haptoglobin in hemolytic disease. JAMA. 1980;243(19):1909-1911. PubMed
Appendix 1: Literature Search Strategy
Note that this appendix has not been copy-edited.
Clinical Literature Search
Overview
Interface: Ovid
Databases:
MEDLINE All (1946 to present)
Embase (1974 to present)
Note: Subject headings and search fields have been customized for each database. Duplicates between databases were removed in Ovid.
Date of search: September 26, 2022
Alerts: Biweekly search updates until project completion
Search filters applied: No filters were applied to limit the retrieval by study type.
Limits:
No date or language limits were used
Conference abstracts: excluded
Syntax | Description |
|---|---|
/ | At the end of a phrase, searches the phrase as a subject heading |
MeSH | Medical Subject Heading |
.fs | Floating subheading |
exp | Explode a subject heading |
* | Before a word, indicates that the marked subject heading is a primary topic; or, after a word, a truncation symbol (wildcard) to retrieve plurals or varying endings |
.ti | Title |
.ot | Original title |
.ab | Abstract |
.hw | Heading word; usually includes subject headings and controlled vocabulary |
.kf | Author keyword heading word (MEDLINE) |
.dq | Candidate term word (Embase) |
.rn | Registry number |
.nm | Name of substance word (MEDLINE) |
medall | Ovid database code: MEDLINE All, 1946 to present, updated daily |
oemezd | Ovid database code; Embase, 1974 to present, updated daily |
Multidatabase Strategy
(pegcetacoplan* or Empaveli* or Aspaveli* or apl 2 or apl2 or TO3JYR3BOU).ti,ab,kf,ot,hw,rn,nm.
use medall
*pegcetacoplan/ or (pegcetacoplan* or Empaveli* or Aspaveli* or apl 2 or apl2).ti,ab,kf,dq.
3 use oemezd
4 not (conference review or conference abstract).pt.
2 or 5
remove duplicates from 6
Clinical Trials Registries
ClinicalTrials.gov
Produced by the US National Library of Medicine. Targeted search used to capture registered clinical trials.
[Search -- Studies with results | pegcetacoplan or Empaveli or Aspaveli]
WHO ICTRP
International Clinical Trials Registry Platform, produced by the WHO. Targeted search used to capture registered clinical trials.
[Search terms -- pegcetacoplan or Empaveli or Aspaveli]
Health Canada’s Clinical Trials Database
Produced by Health Canada. Targeted search used to capture registered clinical trials.
[Search terms -- pegcetacoplan or Empaveli or Aspaveli]
EU Clinical Trials Register
European Union Clinical Trials Register, produced by the European Union. Targeted search used to capture registered clinical trials.
[Search terms -- pegcetacoplan or Empaveli or Aspaveli]
Grey Literature
Search dates: September 13, 2022 to September 20, 2022
Keywords: pegcetacoplan, Empaveli, Aspaveli, paroxysmal nocturnal hemoglobinuria
Limits: None
Relevant websites from the following sections of the CADTH grey literature checklist Grey Matters: A Practical Tool for Searching Health-Related Grey Literature were searched:
Health Technology Assessment Agencies
Health Economics
Clinical Practice Guidelines
Drug and Device Regulatory Approvals
Advisories and Warnings
Drug Class Reviews
Clinical Trials Registries
Databases (free)
Health Statistics
Internet Search
Appendix 2: Excluded Studies
Note that this appendix has not been copy-edited.
Reference | Reason for exclusion |
|---|---|
Hoffman K, Machaidze Z, Yeh M, Weitz IC. Evaluation of the long-term safety and efficacy of pegcetacoplan treatment for paroxysmal nocturnal hemoglobinuria patients: an extension study [poster abstract]. Blood. 2021;138(1): 2175-2176. | Study design (open-label extension study) |
Clinical Study Report: APL2-302. A phase 3, randomized, multicenter, open-label, active-comparator controlled study to evaluate the efficacy and safety of pegcetacoplan in patients with paroxysmal nocturnal hemoglobinuria (PNH) week 48 analysis (internal sponsor's report). Waltham (MA); Apellis Pharmaceuticals; 2021. | Study design (open-label extension period of PEGASUS; summarized under Other Relevant Evidence) |
de Latour RP, Szer J, Weitz IC, et al. Pegcetacoplan versus eculizumab in patients with paroxysmal nocturnal haemoglobinuria (PEGASUS): 48-week follow-up of a randomised, open-label, phase 3, active-comparator, controlled trial. Lancet Haematol. 2022;9(9):e648-e659. | Study design (open-label extension period of PEGASUS; summarized under Other Relevant Evidence) |
Appendix 3: Detailed Outcomes Data
Note that this appendix has not been copy-edited.
Table 38: Primary and Key Secondary End Points in the PEGASUS Trial — Randomized Controlled Period (PP; Censored for Transfusion)
End points | Pegcetacoplan (N = 36) | Eculizumab (N = 35) |
|---|---|---|
Hemoglobin | ||
Change from baseline at week 16 in hemoglobin levela,b (g/dL), LS mean (SE) | 2.94 (0.29) | –1.31 (0.49) |
Difference in LS meansb (g/dL), (95% CI) | 4.25 (3.17 to 5.34) | Reference |
P valuec | < 0.0001 | Reference |
Transfusion avoidance | ||
Patients achieving transfusion avoidanced, n (%) | 35 (97.2) | 6 (17.1) |
Risk differencee (95% CI) | 0.68 (0.55 to 0.81) | Reference |
P valuef | < 0.0001 | Reference |
ARC | ||
Change from baseline in ARC at week 16a,b (109 cells/L), LS mean (SE) | –142.81 (6.74) | 17.88 (11.80) |
Difference in LS meansb (109 cells/L), (95% CI) | –160.68 (–186.68 to –134.68) | Reference |
P valuef | < 0.0001 | Reference |
LDH level | ||
Change from baseline at week 16 in LDH level (U/L)a,b, LS mean (SE) | –69.83 (25.64) | –27.15 (44.17) |
Difference in LS meansb (U/L), (95% CI) | –42.68 (–146.30 to 60.93) | Reference |
P valuef | 0.397 | Reference |
FACIT-Fatigue Scale | ||
Change from baseline at week 16 in FACIT-Fatigue scorea,b, LS mean (SE) | 10.61 (1.57) | –1.81 (2.81) |
Difference in LS meansb (points), (95% CI) | 12.42 (6.19 to 18.66) | Reference |
P valueg | 0.0002 | Reference |
ARC = absolute reticulocyte count; CI = confidence interval; FACIT = Functional Assessment of Chronic Illness Therapy; LDH = lactate dehydrogenase; LS = least square; PP = per-protocol; SE = standard error.
aBaseline was defined as the average of measurements recorded before taking the first dose of pegcetacoplan.
bThe analysis was conducted using a mixed model for repeated measures (MMRM). The model was adjusted for treatment group, study visit, number of packed RBC transfusions within the past 12 months (< 4 vs. ≥ 4), platelet count at screening (< 100,000/mm3 vs. ≥ 100,000/mm3), the study visit by treatment group interaction, and the baseline level of the measurement (i.e., hemoglobin, LDH, ARC level, or FACIT-Fatigue score).
cThe significance threshold was set at a 2-sided alpha of 0.05.
dTransfusion avoidance was defined as the proportion of patients who did not require a transfusion during the randomized controlled period. Patients who did not have a transfusion but withdrew before week 16 were considered as having a transfusion.
eThe risk difference and 95% CI were constructed using the stratified Miettinen-Nurminen method.
fThe p value was based on Cochran-Mantel-Haenszel chi-square test stratified by the number of packed RBC transfusions within the past 12 months (< 4 vs. ≥ 4) and the platelet count at screening (< 100,000/mm3 vs. ≥ 100,000/mm3). In accordance with the hierarchal testing procedure, noninferiority but not superiority testing was conducted.
gIn accordance with the hierarchal testing procedure, noninferiority and superiority testings were not conducted.
Source: PEGASUS Clinical Study Report (16 weeks)6
Table 39: Primary and Key Secondary End Points in the PEGASUS Trial — Randomized Controlled Period (ITT; Uncensored for Transfusion)
End points | PEGASUS (randomized controlled period) | |
|---|---|---|
Pegcetacoplan (N = 41) | Eculizumab (N = 39) | |
Hemoglobin outcomes | ||
Change from baseline at week 16 in hemoglobin levela,b (g/dL), LS mean (SE) | 2.66 (0.25) | –0.03 (0.26) |
Difference in LS meansb (g/dL), (95% CI) | 2.69 (1.99 to 3.38) | Reference |
P valuec | < 0.0001 | Reference |
Hemoglobin normalizationd, n (%) | 14 (34.1) | 0 |
Risk difference (%), (95% CI) | 30.4 (14.9 to 45.9) | Reference |
ARC outcomes | ||
Change from baseline in ARC at week 16a,b (109 cells/L), LS mean (SE) | –132.54 (9.02) | 4.42 (9.15) |
Difference in LS meansb (109 cells/L), (95% CI) | –136.96 (–161.87 to –112.05) | Reference |
P valuec | < 0.0001 | Reference |
Reticulocyte normalizationd, n (%) | 33 (80.5) | 7 (17.9) |
Risk difference (%), (95% CI) | 54.8 (38.8 to 70.7) | Reference |
LDH outcomes | ||
Change from baseline at week 16 in LDH level (U/L)a,b, LS mean (SE) | −43.44 (40.55) | 41.73 (39.61) |
Difference in LS meansb (U/L), (95% CI) | –85.17 (–192.91 to 22.57) | Reference |
P valuec | 0.1207 | Reference |
LDH normalizationd, n (%) | 30 (73.2) | 23 (59.0) |
Risk difference (%), (95% CI) | 12.3 (–7.0 to 31.5) | Reference |
FACIT-Fatigue | ||
Change from baseline at week 16 in FACIT-Fatigue scorea,b, LS mean (SE) | 9.65 (1.410) | –1.69 (1.47) |
Difference in LS meansb (points), (95% CI) | 11.34 (7.47 to 15.22) | Reference |
P valuee | < 0.0001 | Reference |
ARC = absolute reticulocyte count; CI = confidence interval; FACIT = Functional Assessment of Chronic Illness Therapy; ITT = intention to treat; LDH = lactate dehydrogenase; LS = least square; SE = standard error.
aBaseline was defined as the average of measurements recorded before taking the first dose of pegcetacoplan.
bThe analysis was conducted using a mixed model for repeated measures (MMRM). The model was adjusted for treatment group, study visit, number of packed RBC transfusions within the past 12 months (< 4 vs. ≥ 4), platelet count at screening (< 100,000/mm3 vs. ≥ 100,000/mm3), the study visit by treatment group interaction, and the baseline level of the measurement (i.e., hemoglobin, LDH, ARC level, or FACIT-Fatigue score).
cThe significance threshold was set at a 2-sided alpha of 0.05.
dThe risk difference and 95% CI were constructed using the stratified Miettinen-Nurminen method.
eIn accordance with the hierarchal testing procedure, noninferiority and superiority testings were not conducted.
Source: PEGASUS Clinical Study Report (16 weeks)6
Table 40: Change From Baseline at Week 16 in Functional and Symptom Subscale Scores of EORTC QLQ C-30 in the PEGASUS Trial — Randomized Controlled Period (ITT; Censored for Transfusion)
Interventions | Total N | Baseline scale scorea | Week 16 scale score | Change from baseline at week 16 in scorea, LS mean (SE) | Treatment group difference vs. eculizumab, LS Mean Differenceb (95% CI) | P valuec | ||
|---|---|---|---|---|---|---|---|---|
n (%) | Mean (SD) | n (%) | Mean (SD) | |||||
Functional Scale – Physical Functioning | ||||||||
Pegcetacoplan | 41 | 41 (100) | 71.38 (20.23) | 35 (89.7) | 89.52 (11.80) | 16.92 (2.08) | 12.86 (4.86 to 20.86) | 0.0023 |
Eculizumab | 39 | 38 (92.7) | 72.11 (20.14) | 6 (15.4) | 87.78 (18.58) | 4.06 (3.61) | Reference | Reference |
Functional Scale – Role Functioning | ||||||||
Pegcetacoplan | 41 | 41 (100) | 63.82 (29.56) | 35 (89.7) | 80.48 (24.08) | 15.39 (3.93) | 24.43 (8.84 to 40.01) | 0.0027 |
Eculizumab | 39 | 38 (92.7) | 59.65 (33.92) | 6 (15.4) | 66.67 (36.52) | –9.04 (6.95) | Reference | Reference |
Functional Scale – Emotional Functioning | ||||||||
Pegcetacoplan | 41 | 41 (100) | 72.36 (25.38) | 35 (89.7) | 81.67 (22.85) | 7.98 (3.37) | 4.11 (–11.58 to 19.80) | 0.6013 |
Eculizumab | 39 | 37 (90.2) | 69.59 (22.67) | 5 (12.8) | 78.33 (36.13) | 3.86 (7.24) | Reference | Reference |
Functional Scale – Cognitive Functioning | ||||||||
Pegcetacoplan | 41 | 41 (100) | 76.02 (24.45) | 35 (89.7) | 85.24 (20.12) | 5.76 (3.26) | 9.56 (–4.52 to 23.64) | 0.1792 |
Eculizumab | 39 | 37 (90.2) | 75.23 (25.95) | 5 (12.8) | 76.67 (43.46) | –3.80 (6.42) | Reference | Reference |
Functional Scale – Social Functioning | ||||||||
Pegcetacoplan | 41 | 41 (100) | 69.51 (28.84) | 35 (89.7) | 83.33 (24.25) | 15.08 (2.95) | 11.27 (–2.38 to 24.92) | 0.1039 |
Eculizumab | 39 | 37 (90.2) | 64.86 (32.82) | 5 (12.8) | 80.00 (44.72) | 3.82 (6.35) | Reference | Reference |
Symptom Scale – Nausea and Vomiting | ||||||||
Pegcetacoplan | 41 | 41 (100) | 3.66 (8.75) | 35 (89.7) | 3.33 (7.88) | –0.34 (1.63) | –0.01 (–8.38 to 8.35) | 0.9975 |
Eculizumab | 39 | 38 (92.7) | 5.26 (11.69) | 6 (15.4) | 11.11 (27.22) | –0.33 (3.88) | Reference | Reference |
Symptom Scale – Insomnia | ||||||||
Pegcetacoplan | 41 | 41 (100) | 32.52 (34.55) | 35 (89.7) | 23.81 (35.77) | –9.18 (3.96) | 0.32 (–15.67 to 16.30) | 0.9686 |
Eculizumab | 39 | 38 (92.7) | 29.82 (29.80) | 6 (15.4) | 16.67 (40.83) | –9.50 (7.09) | Reference | Reference |
Symptom Scale – Appetite Loss | ||||||||
Pegcetacoplan | 41 | 41 (100) | 12.20 (17.88) | 35 (89.7) | 5.71 (15.09) | –3.76 (3.36) | –7.95 (–23.23 to 7.33) | 0.3002 |
Eculizumab | 39 | 38 (92.7) | 13.16 (23.94) | 6 (15.4) | 16.67 (40.83) | 4.19 (7.01) | Reference | Reference |
Symptom Scale – Constipation | ||||||||
Pegcetacoplan | 41 | 41 (100) | 11.38 (20.561) | 35 (89.7) | 16.19 (24.75) | 2.98 (3.25) | 1.79 (–15.70 to 19.29) | 0.8374 |
Eculizumab | 39 | 37 (90.2) | 10.81 (22.30) | 5 (12.8) | 20.00 (44.72) | 1.19 (8.13) | Reference | Reference |
Symptom Scale – Diarrhea | ||||||||
Pegcetacoplan | 41 | 41 (100) | 11.38 (23.11) | 35 (89.7) | 13.33 (21.69) | 0.31 (3.71) | –1.38 (–19.28 to 16.52) | 0.8775 |
Eculizumab | 39 | 37 (90.2) | 11.71 (21.11) | 5 (12.8) | 6.67 (14.91) | 1.68 (8.20) | Reference | Reference |
Financial Difficulties | ||||||||
Pegcetacoplan | 41 | 41 (100) | 18.70 (26.93) | 35 (89.7) | 8.57 (16.85) | –6.82 (3.85) | –7.40 (−21.76 to 6.95) | 0.3066 |
Eculizumab | 39 | 37 (90.2) | 24.32 (37.39) | 5 (12.8) | 20.00 (44.721) | 0.58 (6.30) | Reference | Reference |
CI = confidence interval; EORTC QLQ-C30 = European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30; ITT = intention to treat; LS = least square; SD = standard deviation; SE = standard error.
aBaseline was the last available nonmissing observation before taking the first dose of pegcetacoplan.
bThe analysis was conducted using a mixed model for repeated measures (MMRM), with the change from baseline to 16 weeks in EORTC QLQ-C30 score as the dependent variable. The model was adjusted for treatment group, study visit, number of packed RBC transfusions within the past 12 months (< 4 vs. ≥ 4), platelet count at screening (< 100,000/mm3 vs. ≥ 100,000/mm3), the study visit by treatment group interaction, and baseline EORTC QLQ-C30 score (continuous).
cThis end point was not adjusted for multiplicity.
Note: Data collected after transfusion was excluded from the analysis.
Source: PEGASUS Clinical Study Report (16 weeks)6
Figure 10: LASA Score Over Time in the PEGASUS Trial — Whole Study Analysis (ITT)
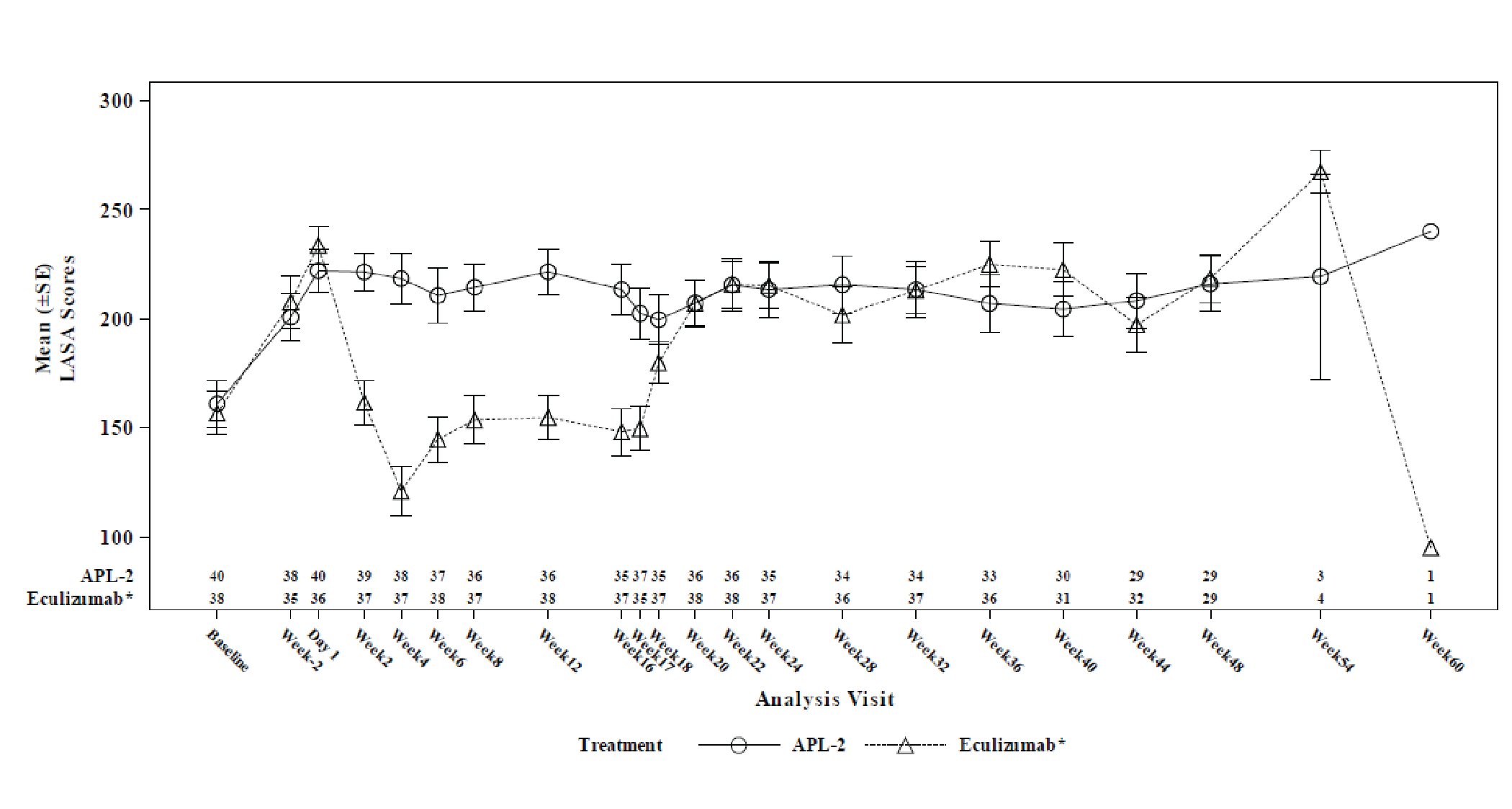
ALP-2 = pegcetacoplan; ITT = intention to treat; SE = standard error.
Note: Baseline was defined as the average of measurements recorded before taking the first dose of pegcetacoplan. Study periods were as follows: run-in period (baseline to day −1); randomized controlled period (day 1 to week 16); open-label pegcetacoplan period (week 17 to week 48); and follow-up period (week 49 to week 60). The eculizumab arm refers to patients who received eculizumab in the randomized controlled phase and subsequently completed a 4-week run-in period (week 17 to week 20) with concurrent treatment with eculizumab and pegcetacoplan, followed by pegcetacoplan monotherapy until week 48.
Source: PEGASUS Clinical Study Report (48 weeks)36
Figure 11: EORTC QLQ-30 Global Health Status/QoL Score Over Time in the PEGASUS Trial — Whole Study Analysis (ITT)
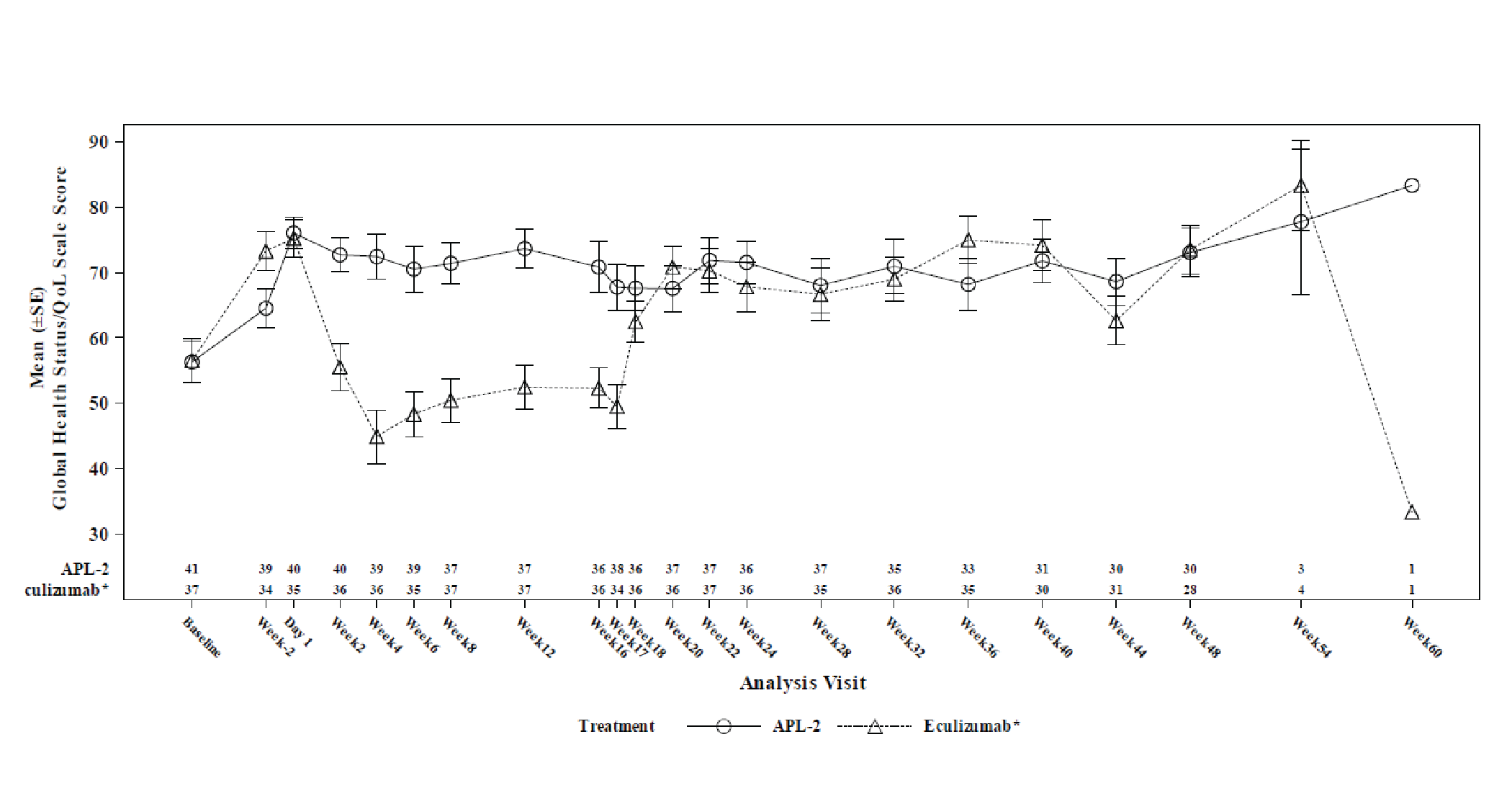
ALP-2 = pegcetacoplan; ITT = intention to treat; QoL = quality of life; SE = standard error.
Note: Baseline was defined as the average of measurements recorded before taking the first dose of pegcetacoplan. Study periods were as follows: run-in period (baseline to day −1); randomized controlled period (day 1 to week 16); open-label pegcetacoplan period (week 17 to week 48); and follow-up period (week 49 to week 60). The eculizumab arm refers to patients who received eculizumab in the randomized controlled phase and subsequently completed a 4-week run-in period (week 17 to week 20) with concurrent treatment with eculizumab and pegcetacoplan, followed by pegcetacoplan monotherapy until week 48.
Source: PEGASUS Clinical Study Report (48 weeks)36
Figure 12: EORTC QLQ-30 Fatigue Subscale Score Over Time in the PEGASUS Trial — Whole Study Analysis (ITT)
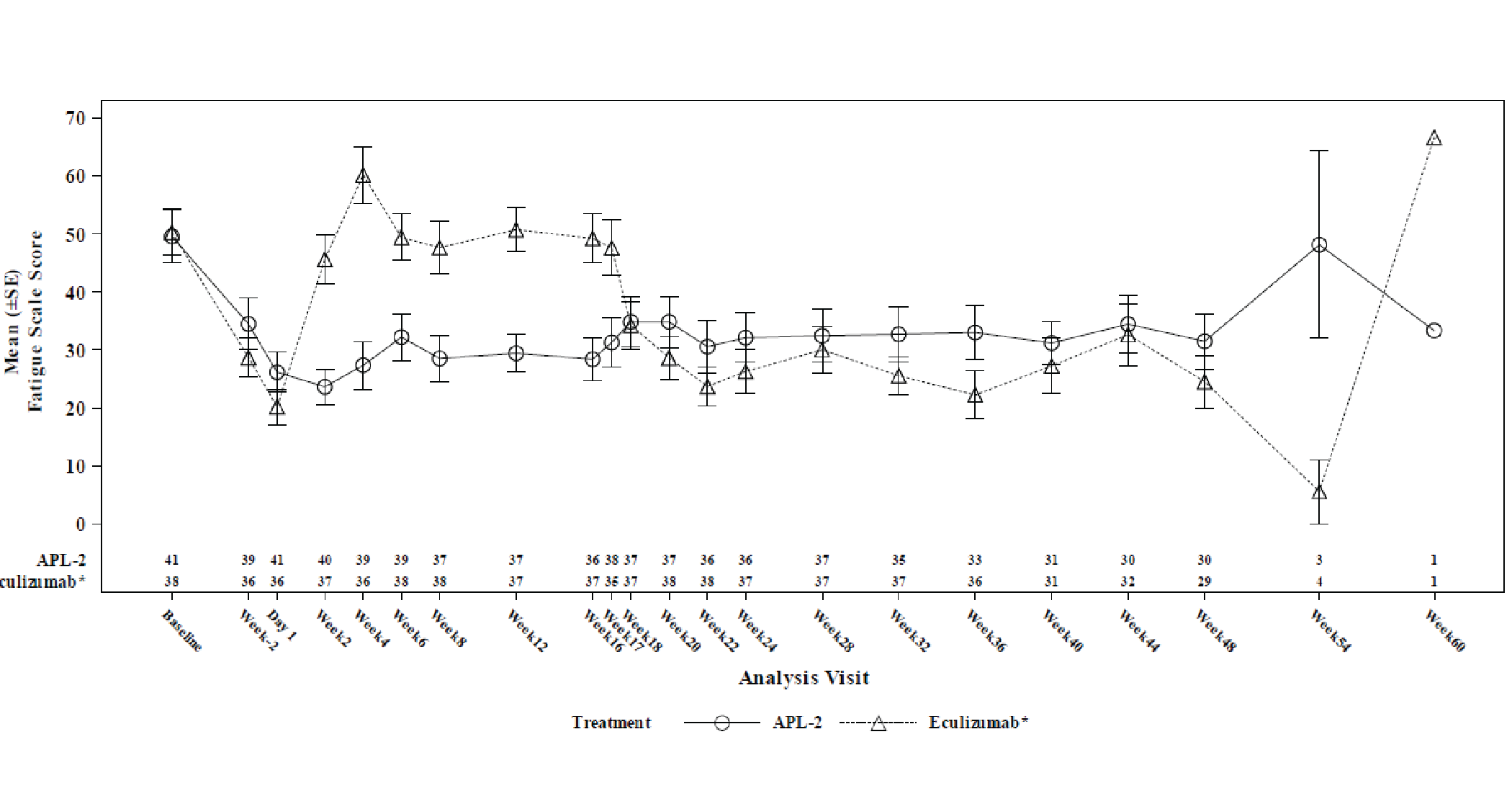
ALP-2 = pegcetacoplan; EORTC QLQ-C30 = European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30; ITT = intention to treat; SE = standard error.
Note: Baseline was defined as the average of measurements recorded before taking the first dose of pegcetacoplan. Study periods were as follows: run-in period (baseline to day −1); randomized controlled period (day 1 to week 16); open-label pegcetacoplan period (week 17 to week 48); and follow-up period (week 49 to week 60). The eculizumab arm refers to patients who received eculizumab in the randomized controlled phase and subsequently completed a 4-week run-in period (week 17 to week 20) with concurrent treatment with eculizumab and pegcetacoplan, followed by pegcetacoplan monotherapy until week 48.
Source: PEGASUS Clinical Study Report (48 weeks)36
Figure 13: EORTC QLQ-C30 Pain Subscale Score Over Time in the PEGASUS Study — Whole Study Analysis (ITT)
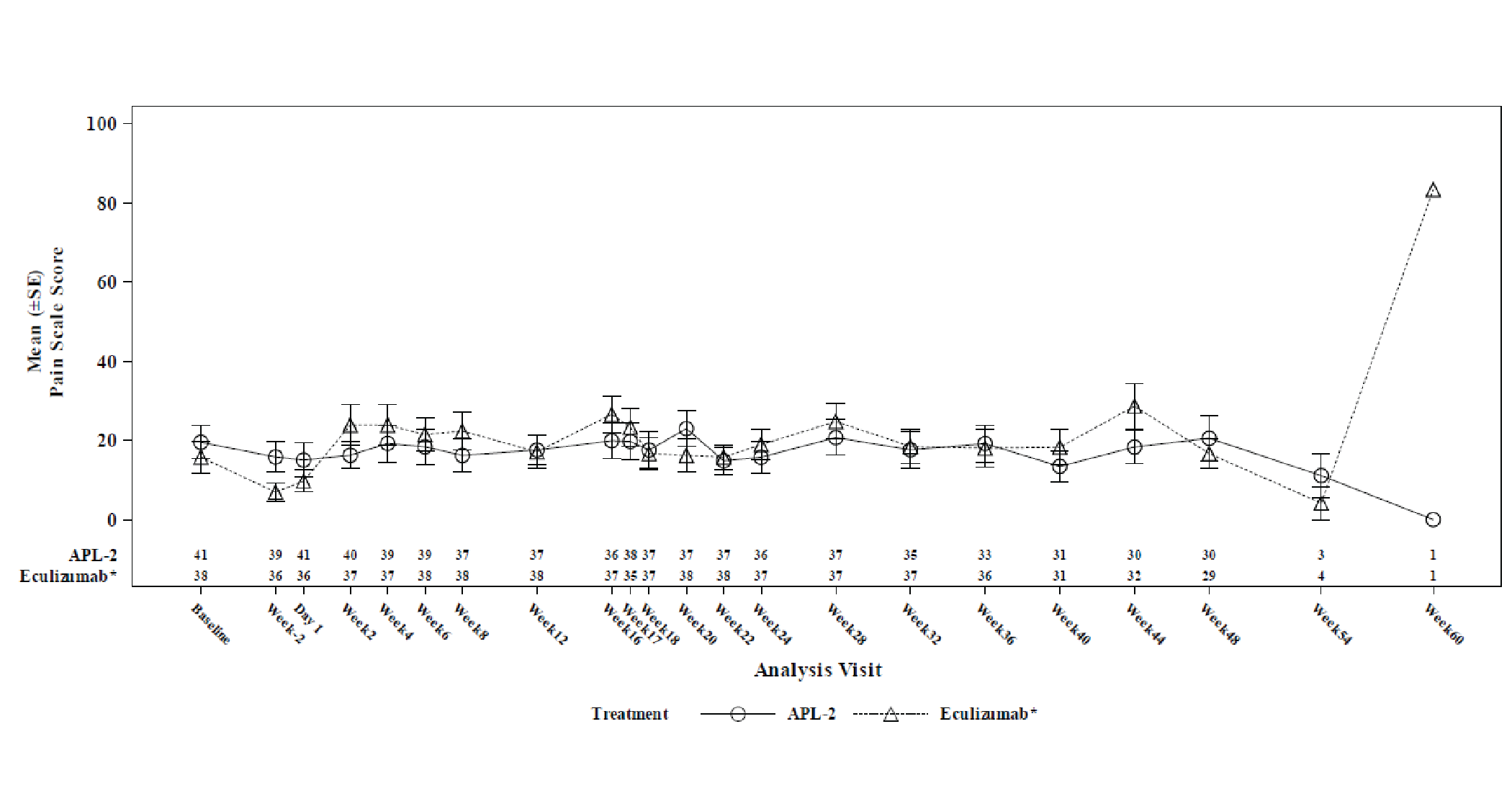
ALP-2 = pegcetacoplan; EORTC QLQ-C30 = European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30; ITT = intention to treat; SE = standard error.
Note: Baseline was defined as the average of measurements recorded before taking the first dose of pegcetacoplan. Study periods were as follows: run-in period (baseline to day −1); randomized controlled period (day 1 to week 16); open-label pegcetacoplan period (week 17 to week 48); and follow-up period (week 49 to week 60). The eculizumab arm refers to patients who received eculizumab in the randomized controlled phase and subsequently completed a 4-week run-in period (week 17 to week 20) with concurrent treatment with eculizumab and pegcetacoplan, followed by pegcetacoplan monotherapy until week 48.
Source: PEGASUS Clinical Study Report (48 weeks)36
Figure 14: EORTC QLQ-30 Dyspnea Subscale Score Over Time in the PEGASUS Trial — Whole Study Analysis (ITT)
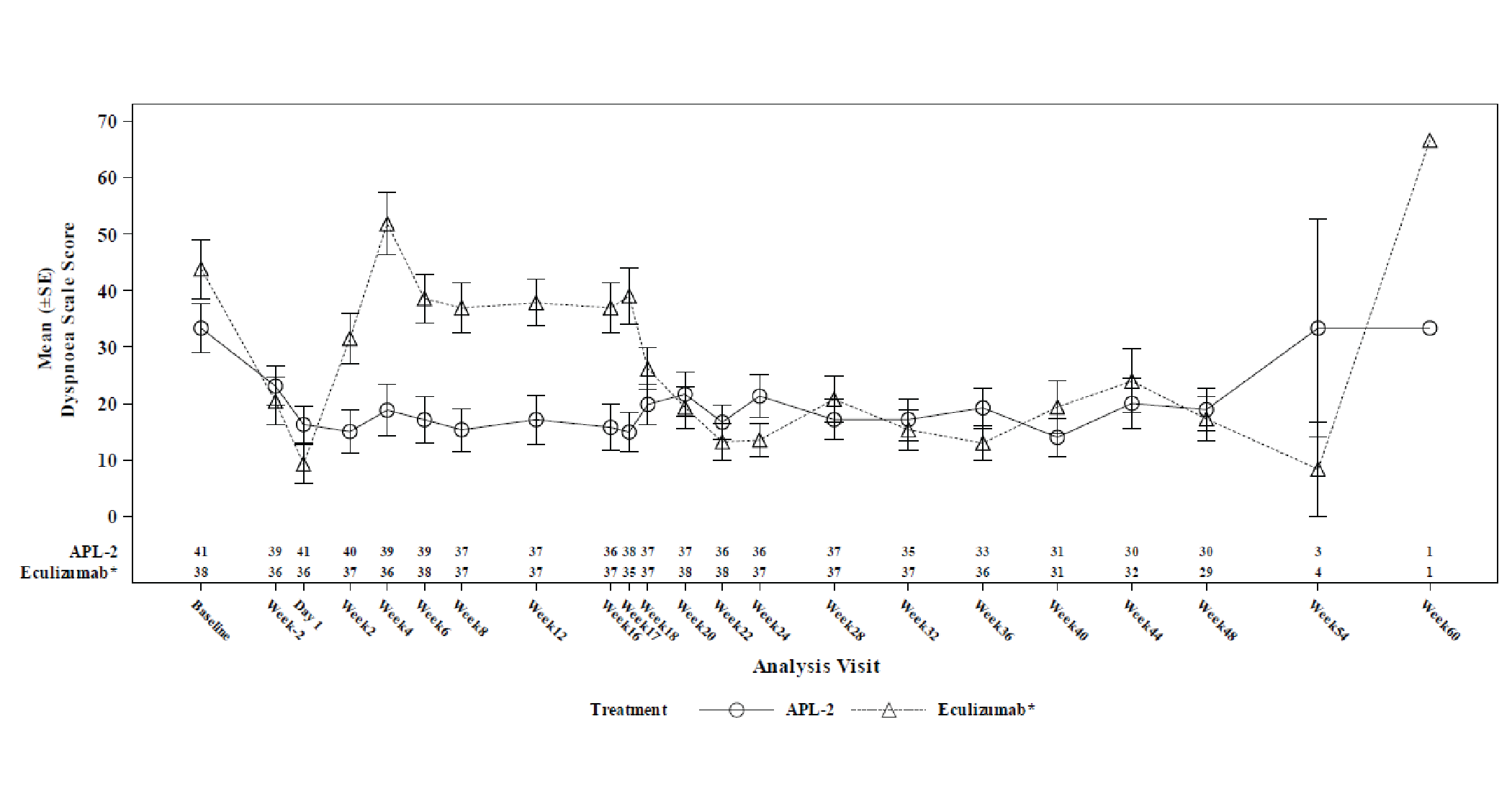
ALP-2 = pegcetacoplan; EORTC QLQ-C30 = European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30; ITT = intention to treat; SE = standard error.
Note: Baseline was defined as the average of measurements recorded before taking the first dose of pegcetacoplan. Study periods were as follows: run-in period (baseline to day −1); randomized controlled period (day 1 to week 16); open-label pegcetacoplan period (week 17 to week 48); and follow-up period (week 49 to week 60). The eculizumab arm refers to patients who received eculizumab in the randomized controlled phase and subsequently completed a 4-week run-in period (week 17 to week 20) with concurrent treatment with eculizumab and pegcetacoplan, followed by pegcetacoplan monotherapy until week 48.
Source: PEGASUS Clinical Study Report (48 weeks)36
Appendix 4: Description and Appraisal of Outcome Measures
Note that this appendix has not been copy-edited.
Aim
To describe the following outcome measures and review their measurement properties (validity, reliability, responsiveness to change, and minimal important difference [MID]):
EORTC QLQ-C30
FACIT-Fatigue
LASA
Hemoglobin
LDH ≥ 1.5 x ULN
Reticulocyte count
Bilirubin level
Haptoglobin level
Findings
Table 41: Summary of Outcome Measures and Their Measurement Properties
Outcome measure | Type | Conclusions about measurement properties | MID |
|---|---|---|---|
EORTC QLQ-C30 | 30-item, patient-reported, cancer-specific, quality of life questionnaire using 4- and 7-point Likert scales. It consists of 5 multiitem functional scales (physical, role, emotional, cognitive, and social), 3 multiitem symptom scales (fatigue, nausea-vomiting, and pain), 6 single-item symptom scales (dyspnea, insomnia, appetite loss, constipation, diarrhea, and financial impact), and a 2-item GHS/QoL scale. A 1-week recall period is used to assess the items.42 Each raw scale score is converted to a standardized score that ranges from 0 to 100 using a linear transformation. A higher score on the functional scales represents better functioning, a higher score on the symptom scales represents a higher level of symptomatology, and a higher score on the global health status/HRQoL scale represents a higher HRQoL.42 | Patients with PNH: The content validity was confirmed in patients with PNH.26 Convergent validity between EORTC QLQ-C30 scales and Hb, ARC, and indirect bilirubin (post hoc analysis using PEGASUS data) were:
Responsiveness (based on a post hoc analysis using PEGASUS data): Patients with improvements in hemoglobin, indirect bilirubin, and ARC showed improvements in physical functioning (P = 0.0103, P = 0.0050 and P = 0.0072, respectively) and fatigue scores (P = 0.0093, P = 0.0073 and P = 0.0162, respectively)23 Patients with cancer: Reliability of the EORTC QLQ-C30 in HL and DLBCL patients undergoing chemotherapy measured by Cronbach alpha was 0.79 for GHS/QoL, 0.51 to 0.85 for functional scales, and 0.82 to 0.86 for symptom scales/items.43 No evidence was identified for reliability in patients with PNH. | Patients with cancer44:
No MID was identified in patients with PNH. |
FACIT-Fatigue | 13-item, patient-reported, fatigue-specific, quality of life questionnaire using a 5-point Likert scale. It assesses tiredness, weakness, and difficulty conducting usual activities as a result of fatigue over the past week.45 The 13-item scale ranges from zero (extreme fatigue) to 52 (no fatigue). Higher scores indicate less fatigue.45 | Patients with PNH: The content validity was confirmed as an appropriate tool to be used in patients with PNH.26 Convergent validity between FACIT-Fatigue and Hb, ARC, and indirect bilirubin (post hoc analysis using PEGASUS data) were r = 0.47, r = −0.37 and r = −0.25, respectively23 Responsiveness (based on a post hoc analysis using PEGASUS data): Patients with improvements in hemoglobin, indirect bilirubin, and ARC showed improvements in FACIT-Fatigue scores (P < 0.0001, P = 0.0002 and P = 0.0002, respectively)23 Patients with cancer5 or psoriatic arthritis6: Internal consistency by Cronbach alpha was 0.9545 and test-retest by intraclass correlation coefficient was 0.95.46 No evidence was identified for reliability in patients with PNH. | Patients with PNH: Clinically important difference: 5 points.
Two approaches were used to determine clinically important differences:
|
LASA | Five-item, patient-reported, quality of life questionnaire using a 10-point Likert scale. It assesses physical, emotional, spiritual, and intellectual well-being, and an item for overall quality of life over the past week.28 The scales range from 0 (as bad as it can be) to 10 (as good as it can be). Higher scores indicate better QoL.28 Note: in the sponsor’s CSR, an abbreviated version of LASA was used to measure physical well-being and overall quality of life over the past week.6 | Patients with cancer: The validity of LASA scales in patients with cancer were confirmed with the following corresponding scales: Symptom Distress Scale, Profile of Mood States, and Functional Assessment for Cancer Therapy-Brain (r = 0.44 to 0.65; P < 0.001).28 No evidence was identified for validity, reliability, or responsiveness in patients with PNH. | No MID was identified in patients with PNH. |
Hemoglobin | Laboratory test used in diagnosis and monitoring of disease activity.47 | Patients with PNH: Refer to Table 42 for proposed response categories in patients with PNH. No evidence was identified for validity or reliability in patients with PNH. Patients with sickle cell disease: Proposed as a strong surrogate biomarker for clinical benefit in patients with sickle cell disease receiving stem cell or gene therapy.24,25 | Patients with sickle cell disease: Disease improvement: Hemoglobin > 1 g/dL from baseline.24,25 No MID was identified in patients with PNH. |
LDH ≥ 1.5 x ULN | Laboratory test used in diagnosis and monitoring of disease activity.47 | Patients with PNH: Associated with 4.8-fold increase in risk of mortality30 and increased risk of thromboembolism (OR 7.0; P = 0.013) in patients with PNH.31 Refer to Table 42 for proposed response categories in patients with PNH. No evidence was identified for reliability in patients with PNH. | No MID was identified in patients with PNH. |
Reticulocyte count | Laboratory test used in diagnosis and monitoring of disease activity.47 | Patients with PNH: Refer to Table 42 for proposed response categories in patients with PNH. No evidence was identified for validity or reliability in patients with PNH. | No MID was identified in patients with PNH. |
Bilirubin level | Laboratory test used in diagnosis and monitoring of disease activity.47 | No evidence was identified for validity, reliability, or responsiveness. | No MID was identified. |
Haptoglobin level | Laboratory test used in diagnosis and monitoring of disease activity.47 | Patients with hemolytic disease: The sensitivity and specificity were 83% and 96%, respectively, providing 87% probability of predicting hemolytic disease when the serum haptoglobin level is ≤ 25 mg/dL.48 No evidence was identified for validity, reliability, or responsiveness in patients with PNH. | No MID was identified in patients with PNH. |
DLBCL = diffuse large B-cell lymphoma; EORTC QLQ-C30 = European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30; LASA = Linear Analogue Scale Assessment; GHS = global health status; HL = Hodgkin’s lymphoma; LDH = lactate dehydrogenase; MID = minimal important difference; OR = odds ratio; PNH = paroxysmal nocturnal hemoglobinuria; QoL = quality of life; ULN = upper limit of normal; Hb = hemoglobin; ARC = absolute reticulocyte count; r = correlation coefficient;
Table 42: Proposed Classification of Hematological Response to Anticomplement Drugs in Patients With PNH — Severe Aplastic Anemia Working Party of the European Group for Bone Marrow Transplantation14
Response category | Red blood cell transfusions | Hemoglobin level | LDH levela,b | Absolute reticulocyte count |
|---|---|---|---|---|
Complete response | None | ≥ 12 g/dL | ≤ 1.5x ULN | and ≤ 150,000/μLc |
Major response | None | ≥ 12 g/dL | > 1.5x ULN | or > 150,000/μLc |
Good response | None | ≥ 10 and < 12 g/dL | A. ≤ 1.5x ULN B. > 1.5x ULN | Rule out bone marrow failured |
Partial response | None or occasional (≤ 2 every 6 months) | ≥ 8 and < 10 g/dL | A. ≤ 1.5x ULN B. > 1.5x ULN | Rule out bone marrow failured |
Minor responsef | None or occasional (≤ 2 every 6 months) Regular (3 to 6 every 6 months) Reduction by ≥ 50%e | < 8 g/dL < 10 g/dL < 10 g/dL | A. ≤ 1.5x ULN B. > 1.5x ULN | Rule out bone marrow failured |
No responsef | Regular (> 6 every 6 months) | < 10 g/dL | A. ≤ 1.5x ULN B. > 1.5x ULN | Rule out bone marrow failured |
LDH = lactate dehydrogenase; ULN = upper limit of the normal; ARC = absolute reticulocyte count.
aResponse categories are mostly based on red blood cell transfusion and hemoglobin level, but LDH and ARC serve as ancillary indicators to discriminate between complete and major response, as well as within suboptimal response categories.
bA. and B. indicate subcategories without or with residual significant intravascular hemolysis, respectively.
cTo rule out increased erythropoietic response to compensate ongoing hemolysis; the value of 150,000/μL is a tentative index based on 1.5x ULN (which in most laboratories is set at 100,000/μL).
dTo assess the relative contribution of the degree of bone marrow failure to any response less than complete: a value of ARC below 60,000/μL could be a tentative index to establish such a contribution; bone marrow investigation may be appropriate.
eFor patients with previous transfusion history (with a pre-treatment follow-up of at least 6 months).
fFor patients who do not accept red blood cell transfusions, minor response can be defined based on hemoglobin level ≥ 6 and < 8 g/dL, and no response based on hemoglobin < 6 g/dL. All hemoglobin, LDH and ARC values should be assessed based on the median value over a period of 6 months.
Source: Risitano et al. (2019)14
Pharmacoeconomic Review
Abbreviations
AE
adverse event
BIA
budget impact analysis
BTH
breakthrough hemolysis
C5
complement component 5
EORTC QLQ-C30
European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30
Hb
hemoglobin
ICER
incremental cost-effectiveness ratio
LDH
lactate dehydrogenase
PNH
paroxysmal nocturnal hemoglobinuria
PSP
Patient Support Program
QALY
quality-adjusted life-year
TA
transfusion avoidance
WTP
willingness to pay
Executive Summary
The executive summary comprises 2 tables (Table 1 and Table 2) and a conclusion.
Item | Description |
|---|---|
Drug product | Pegcetacoplan (Empaveli), subcutaneous solution for infusion |
Submitted price | Pegcetacoplan, 1,080 mg (54 mg/mL) solution for infusion: $4,970.00 per single-dose vial |
Indication | For the treatment of adult patients with PNH who have an inadequate response to, or are intolerant of, a C5 inhibitor |
Health Canada approval status | NOC |
Health Canada review pathway | Priority review |
NOC date | December 8, 2022 |
Reimbursement request | Per indication |
Sponsor | Sobi Canada Inc. |
Submission history | Previously reviewed: No |
C5 = complement component 5; NOC = Notice of Compliance; PNH = paroxysmal nocturnal hemoglobinuria.
Table 2: Summary of Economic Evaluation
Component | Description |
|---|---|
Type of economic evaluation | Cost-utility analysis Markov cohort model |
Target population | Adult patients with PNH who have an inadequate response to C5 inhibitors The population does not align with the full anticipated Health Canada indication, which also includes patients who are intolerant to complement component 5 inhibitors |
Treatment | Pegcetacoplan in addition to the patient’s current C5 inhibitor treatment during the first 4 weeks of treatment |
Comparators |
|
Perspective | Canadian publicly funded health care payer |
Outcomes | QALYs, LYs |
Time horizon | Lifetime (51.2 years) |
Key data source |
|
Submitted results |
|
Key limitations |
|
CADTH reanalysis results |
|
C5 = complement component 5; Hb = hemoglobin; ICER = incremental cost-effectiveness ratio; LY = life-year; PNH = paroxysmal nocturnal hemoglobinuria; QALY = quality-adjusted life-year.
Conclusions
The CADTH clinical review concluded that pegcetacoplan demonstrated clinically meaningful improvements in mean hemoglobin (Hb) level compared with eculizumab over 16 weeks of treatment in adult patients with paroxysmal nocturnal hemoglobinuria (PNH) who had an inadequate response to eculizumab. Transfusion avoidance was more frequently observed in the pegcetacoplan arm than in the eculizumab arm and, although only noninferiority could be concluded between treatments, the clinical experts considered the results to be supportive of the benefit of pegcetacoplan in improving anemia.
CADTH undertook reanalyses to address the key limitations in the sponsor’s analysis, which included adopting the same transition probability matrix for the first and subsequent cycles of eculizumab and ravulizumab and selecting a discontinuation rate for pegcetacoplan that was based on more patient observations. Based on the CADTH reanalysis, the results were largely consistent with the sponsors. In adult patients with PNH who have an inadequate response to complement component 5 (C5) inhibitors, pegcetacoplan remained less costly and more effective compared with continued use of eculizumab. The incremental cost-effectiveness ratio (ICER) of pegcetacoplan compared with continued use of ravulizumab was $62,144 per quality-adjusted life-year (QALY) gained. A price reduction of 0.9% would be needed for pegcetacoplan to be cost-effective at a willingness-to-pay (WTP) threshold of $50,000 per QALY.
These results were largely driven by the drug acquisition costs of the comparators, which were based on publicly available list prices, as drug costs accounted for more than 99% of total costs across all 3 treatments. CADTH conducted a series of 2-way price-reduction analyses to highlight the point at which pegcetacoplan would no longer be considered cost-effective compared with eculizumab. If the confidential price of eculizumab is discounted by only 11.5% from its current list price, pegcetacoplan would no longer be cost-effective at a WTP threshold of $50,000 per QALY. Further uncertainties could not be addressed by CADTH, including up-dosing of eculizumab and pegcetacoplan, which, if reimbursed by public drug plans, would be expected to increase drug costs. In a scenario analysis in which patients with breakthrough hemolysis are managed with pegcetacoplan up-dosing rather than a switch to eculizumab, the ICER of pegcetacoplan increased to $416,365 per QALY.
With respect to clinical effectiveness, the model was heavily driven by the difference in the utility values between 2 health states: Hb levels of 10.5 g/dL or greater; and Hb levels of less than 10.5 g/dL. There is limited predictive validity on the meaningfulness of this cut-off value and, given the uncertainty associated with the method to derive utility estimates, it is unclear whether the estimated incremental QALYs produced by the economic model would be realized. A scenario analysis demonstrated that if the difference in utility values between these health states is reduced, the ICER of pegcetacoplan would increase and a larger price reduction would be warranted. Furthermore, the current analyses do not model waning in any of the treatments, but the impact would be expected to be larger in the pegcetacoplan arm, given that patients would transition to health states associated with lower utilities. As such, if treatment waning had been incorporated, the incremental QALY gains would be smaller, leading to a higher ICER estimate for pegcetacoplan.
Stakeholder Input Relevant to the Economic Review
This section is a summary of the feedback received from the patient groups, registered clinicians, and drug plans that participated in the CADTH review process.
Patient input was jointly received for this review from the Canadian Association of PNH Patients and the Aplastic Anemia & Myelodysplasia Association of Canada. Patient input to describe the impact of pegcetacoplan on quality of life was gathered through interviews with 6 patients living in Canada who have PNH and from qualitative scientific literature. Three patients were treated with pegcetacoplan in clinical trial programs, whereas the remaining patients had extravascular hemolysis manifestations and were treated with eculizumab. Thrombosis was reported as the most devastating consequence of hemolysis in patients with PNH, as it can damage organs and cause premature death. Two patients interviewed had experienced thrombosis, which resulted in strokes. Patients who had experience with eculizumab or ravulizumab reported a burden associated with IV infusions and, despite treatment, some remained severely anemic, were transfusion-dependent, and experienced substantial fatigue. Patients who had experience with pegcetacoplan reported a better quality of life, considering that, before pegcetacoplan, they were dependent on blood transfusion. Possible benefits associated with pegcetacoplan included an increase in Hb level and a decrease in dependence on blood transfusions. Another benefit is that pegcetacoplan can be self-administered at home.
Clinician group input was received from the Canadian PNH Network. Clinician input noted that eculizumab remains the only standard-of-care option for patients with PNH, given that ravulizumab is not yet listed on public drug plans and access is only available to patients who participated in the initial clinical trials or who have private insurance plans. Existing treatments based on terminal complement inhibition with C5 blockade can cause extravascular hemolysis. The clinician group recognized that pegcetacoplan is the only available drug to treat extravascular hemolysis, which is expected to fill an unmet need in patients who have not responded adequately to C5 inhibitors. Patients with C3 detected on the surface of their red blood cells, particularly, would benefit from pegcetacoplan, although current clinical assays are not very sensitive for identifying extravascular hemolysis. The clinician group agreed with inclusion criteria used in the PEGASUS study to identify patients most likely to benefit from pegcetacoplan, which comprised patients with an insufficient response to C5 inhibitor treatment (Hb < 10.5 g/L). An expected clinically meaningful response to any new treatment would be sustained control of lactate dehydrogenase (LDH) (LDH ratio of < 1.5 times the upper limit of normal), with a further increase in Hb, decreased transfusion requirements, reduced risk of thrombosis, decreased anemia-related symptoms such as fatigue, and improved quality of life.
Drug plan input noted that eculizumab is listed on most public drug plans, whereas ravulizumab is currently involved in pan-Canadian Pharmaceutical Alliance negotiations. Concerns were raised regarding the assessment and monitoring of therapeutic response in the absence of universally accepted methods to determine response to C5 inhibitor treatment. The drug plans also noted the potential for up-dosing with pegcetacoplan and questioned whether patients with an inadequate response to pegcetacoplan alone could be treated with both a C5i and pegcetacoplan. The drug plans noted the uncertainty around what the Patient Support Program (PSP) covers, such as whether costs associated with required vaccinations and alternative pumps are covered by the PSP or drug plan. Furthermore, there is concern about the availability of alternative pump systems if the FreedomEdge syringe infusion system is out of stock.
Several of these concerns were addressed in the sponsor’s model:
the sponsor’s model included relevant comparators (i.e., eculizumab and ravulizumab)
the model structure captured Hb levels and the transfusion status of patients with PNH
patients in the transfusion avoidance (TA) health state had higher utility values than those in the transfusion required state.
CADTH was unable to address the following concerns raised from stakeholder input:
Uncertainties regarding the up-dosing of eculizumab and pegcetacoplan could not be addressed in CADTH’s base-case analysis because of the lack of evidence on how up-dosing could impact clinical outcomes.
The sponsor’s model structure could not be modified to explicitly incorporate some relevant outcomes, such as hemolysis.
The possible use of pegcetacoplan in combination with a C5 inhibitor for patients who had an inadequate response to pegcetacoplan treatment could not be assessed because this has not been clinically studied. The clinical experts consulted by CADTH noted that most hematologists would be reluctant to prescribe a C5 inhibitor and pegcetacoplan concurrently and that they have limited experience with this treatment approach.
Economic Review
The current review is for pegcetacoplan (Empaveli) for adult patients with PNH who have an inadequate response to C5 inhibitors (eculizumab and ravulizumab).
Economic Evaluation
Summary of Sponsor’s Economic Evaluation
Overview
Pegcetacoplan is indicated for the treatment of adult patients with PNH who have an inadequate response to, or are intolerant of, C5 inhibitor treatment (i.e., eculizumab or ravulizumab).1 The sponsor submitted a cost-utility analysis comparing pegcetacoplan with eculizumab and ravulizumab in the treatment of adult patients with PNH who have an inadequate response to C5 inhibitors.2 The target population does not align with the anticipated Health Canada indication.
Pegcetacoplan is available as 54 mg/mL in a 20 mL single-dose vial (total of 1,080 mg per vial) for subcutaneous infusion. The recommended dose of pegcetacoplan is 1,080 mg twice weekly,1 but dosage can be increased to 1,080 mg every third day if a patient’s LDH level rises to greater than 2 times the upper limit of normal (i.e., a sign of breakthrough hemolysis [BTH]).3,4 Pegcetacoplan is given to patients for the first 4 weeks in addition to their current dose of C5 inhibitor treatment.1 At the submitted price of $4,970 per vial, the annual cost of pegcetacoplan is $522,514. When the run-in period cost of C5 inhibitor treatment is included, the annual cost of the pegcetacoplan regimen estimated by the sponsor was between $522,514 and $566,874 in the first year, depending on the C5 inhibitor patients are switching from. The sponsor calculated the annual costs of eculizumab and ravulizumab to be $578,270 and $523,145, respectively, assuming a distribution of up-dosing regimens based on the PEGASUS trial for eculizumab and the product monograph dosing for ravulizumab.5,6
Outcomes of the model included QALYs and life-years over a lifetime time horizon of 51.2 years (with a cycle length of 4 weeks) from the Canadian public health care payer perspective. Both costs and outcomes were discounted at 1.5% per annum.2
Model Structure
The sponsor submitted a Markov cohort model based on the TA and transfusion required health states. In the sponsor’s base-case analysis, the model consists of 4 health states: TA and an Hb level less than 10.5 g/dL (“TA Hb < 10.5”), TA and a Hb level of at least or equal to 10.5 g/dL (“TA Hb ≥ 10.5”), transfusion required, and death (Figure 1).2 The Hb level of less than 10.5 g/dL was used as the cut-off to define an insufficient response to PNH treatment as per the inclusion criteria for the pivotal trial.7 All patients entered the model in the TA Hb < 10.5 health state. Patients remain in 1 of the TA health states based on their Hb level (i.e., TA Hb < 10.5 or TA Hb ≥ 10.5) unless a transfusion is required, in which case they would transition to the transfusion required health state. Patients in the transfusion required health state can transition to another health state in the next cycle or stay in the same health state if additional blood transfusions are required.2 All patients alive, regardless of health state, were at risk of developing PNH-related complications, including BTH, thrombosis, acute kidney damage, chronic kidney disease, pulmonary hypertension, and iron overload. Patients could die from PNH-related complications or other causes at any time.2
Model Inputs
The model’s baseline population characteristics used to inform the model were characterized according to the PEGASUS trial (NCT03500549), with a mean starting age of 48.8 years (SE = 1.79 years), average weight of 75.25 kg (SE = 1.97 kg), and 61.3% were female (49 of 80).3,4
The clinical efficacy parameters in the model were informed by the PEGASUS trial, which was a multinational, randomized, open-label trial that compared pegcetacoplan to eculizumab in 80 adults with PNH who had an inadequate response (defined as Hb < 10.5 g/dL), despite eculizumab treatment for at least or equal to3 months.3 Because there was no direct evidence comparing pegcetacoplan to ravulizumab, the sponsor assumed equivalent efficacy between ravulizumab and eculizumab.2,6 Patient-level data were used to derive transition probabilities for pegcetacoplan and eculizumab. The transition probabilities between health states were assumed to differ at 2 distinct time points: the treatment run-in period (i.e., first 4 weeks of treatment) and the period thereafter (i.e., > 4 weeks of treatment).3 As such, the PEGASUS trial’s week 4 data were used to calculate the transition probabilities for the first 4 weeks (i.e., cycle 1), and the trial’s week 8 to week 16 data were used to calculate the transition probabilities for all subsequent cycles.3 The likelihood of developing PNH-related complications was treatment-independent and varied by health state, with the exception of iron overload, for which the rates were treatment-dependent.
The sponsor assumed that treatment-related discontinuation due to adverse events (AEs) would only occur in the first year, regardless of the patient’s health state. Patients who did not discontinue treatments in the first year would continue to receive the same treatment for the rest of their lives.3,4 For patients in the transfusion required health state, treatment discontinuation due to lack of efficacy was further incorporated but was similarly assumed to only occur in the first year of treatment. Patients with an inadequate response to pegcetacoplan would be switched to eculizumab, and the eculizumab transition probabilities were used to inform disease progression for this subset of the patient population. Patients with an inadequate response to eculizumab were assumed to remain on eculizumab at a higher or more frequent dose.3
Background mortality across all health states was assumed to be equal to age- and sex-adjusted general population mortality rates.8
Health state utility values were estimated based on European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30 (EORTC QLQ-C30) data from the PEGASUS trial. EORTC QLQ-C30 data were mapped to 3 Level EQ-5D utilities using a mapping algorithm derived from patients with cancer.9 The sponsor’s base-case model did not include disutility weights associated with AEs or PNH-related complications, as it was assumed that their impact would already be accounted for in the mapped utility values.2
The dosing used to inform pegcetacoplan and eculizumab were obtained from the PEGASUS trial,7 and ravulizumab dosing was informed from the product monograph.1 Drug acquisition costs of pegcetacoplan were based on the sponsor’s submitted price, and other drug costs came from published sources.5,6 The dosing and frequency of supportive treatments were derived from the PEGASUS trial and validated with Canadian PNH experts.7 There were no administration costs because those were assumed to be covered by the respective drug manufacturers. Costs related to vaccination and prophylactic antibiotics were not included in the base-case model. In addition to treatment-acquisition costs, the following were included in the sponsor’s submitted model: the cost of managing PNH-related complications, the cost of blood transfusions, the cost of managing transfusion-related acute reactions, health care resources use costs, and AE management costs. The number of transfusions in each cycle depended on treatment and health state, as estimated from the PEGASUS trial, whereas the frequency of other health care resource use was estimated based on expert opinion. Unit costs were sourced from a variety of sources, including the Ontario Case Costing Initiative10 and the Ontario Schedule of Benefits.11
Summary of Sponsor’s Economic Evaluation Results
The sponsor presented a probabilistic base-case analysis based on 1,000 iterations, the results of which are presented here. Deterministic results align with the probabilistic results when pegcetacoplan was compared to eculizumab. However, deterministic results were different from the probabilistic results when pegcetacoplan was compared to ravulizumab. This was due to a large incremental difference in costs, arising from the wide uncertainty in the probabilistic distribution of patient weights, which have an impact on the calculation of treatment costs for ravulizumab. In the deterministic analysis, the cost difference between ravulizumab and pegcetacoplan was $133,954, whereas in the probabilistic analysis, the cost difference between ravulizumab and pegcetacoplan was $48,227. The probabilistic findings are presented here.
Base-Case Results
The results of the sponsor’s probabilistic base-case analysis demonstrated that pegcetacoplan dominated eculizumab (i.e., less costly, more effective). Compared to ravulizumab, the use of pegcetacoplan was associated with an incremental cost of $48,227 and 1.96 incremental QALYs, resulting in an ICER of $24,636 per QALY (Table 3).2
Drug costs accounted for more than 99% of total costs across all 3 treatments. There was no life-year gain associated with any of the treatments. All the QALY gains for pegcetacoplan compared with eculizumab and ravulizumab were accrued in the TA Hb ≥ 10.5 health state because the model predicted that pegcetacoplan, compared to other treatments, would have a higher proportion of patients transitioning to the TA Hb ≥ 10.5 health state, which was associated with a higher utility value.
Table 3: Summary of the Sponsor’s Economic Evaluation Results
Drug | Total costs ($) | Total QALYs | Sequential ICER ($/QALY) |
|---|---|---|---|
Ravulizumab | $14,212,985 | 18.152 | Reference |
Pegcetacoplan | $14,261,211 | 20.110 | $24,636 |
Eculizumab | $15,746,059 | 17.041 | Dominateda |
ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year.
aDominated refers to a treatment having higher total costs and lower total QALYs than the previous less costly treatment.
Source: Sponsor’s pharmacoeconomic submission.2
Sensitivity and Scenario Analyses Results
The sponsor conducted several scenario analyses, including adopting a broader perspective that included transportation and productivity costs; varying health state utility values; testing a different approach to modelling mortality, in which there would be differences in life-year gains between treatments; selecting alternative data sources to inform the treatment discontinuation rate; varying management of BTH in patients receiving pegcetacoplan; and excluding the 4-week run-in treatment cost of pegcetacoplan. In the scenario analyses, pegcetacoplan consistently dominated eculizumab. Pegcetacoplan was generally more costly and always more effective than ravulizumab. In the scenarios in which run-in-period pegcetacoplan costs were excluded (covered by the sponsor) and discontinuation data for pegcetacoplan were derived from the randomized controlled period of the PEGASUS trial only, pegcetacoplan dominated ravulizumab. In contrast, the scenario analysis that considered pegcetacoplan dose escalation for patients with BTH had the largest impact, with an ICER of $317,896 per QALY.
CADTH Appraisal of the Sponsor’s Economic Evaluation
CADTH identified several key limitations to the sponsor’s analysis that have notable implications on the economic analysis:
The model structure does not appropriately capture all aspects of the condition: An appropriate economic evaluation model structure should capture relevant and meaningful underlying clinical and/or biological processes. To capture costs and health-related quality of life associated with PNH disease progression and the impacts of treatment, the sponsor’s submitted model structure was based on patient Hb levels and transfusion status. This limited the attribution of costs and benefits to transfusion status and Hb levels without adequately accounting for other disease-related symptoms or the need for medical interventions, apart from the treatment of iron overload and BTH. Input from the clinician experts consulted by CADTH and patients recognized thrombosis as the most devastating consequence of PNH; nevertheless, this condition was not explicitly modelled. Rather, it was assumed to be a complication that only occurred in the transfusion required health state, with the parameter value informed from the PEGASUS trial. Similarly, other important aspects of the disease (i.e., pulmonary hypertension and chronic or acute kidney disease) were informed by data from the 16-week randomized controlled period of the PEGASUS trial in which no events were reported. The sponsor’s approach was to set the frequency of these events to 0 over the modelled time horizon, which implicitly assumes that there is no difference between pegcetacoplan and C5 inhibitors over a lifetime. There is no evidence to support this assumption. As noted from clinical expert feedback sought by CADTH and the CADTH clinical report, the PEGASUS trial duration was insufficient to provide certainty on the incidence of these events.
In the TA health states, this was further dichotomized based on an Hb level cut-off of 10.5 g/dL. Hb less than 10.5 g/dL alone was used as the main indicator of inadequate treatment response, without consideration of any other clinical symptoms. There is no universally accepted method to assess response to C5 inhibitor treatment. The clinical experts consulted by CADTH considered an improvement of at least 1 g/dL to be a clinically meaningful improvement in most patients with PNH, especially those with severe anemia at baseline. The clinical experts noted that a relative change in baseline Hb level would be considered a more meaningful measure of PNH treatment response than an arbitrary Hb cut-off value as the clinically meaningful Hb threshold may differ by patient based on baseline Hb levels. By dichotomizing TA, the sponsor selected different utilities values associated with each health state. This is important to note, given the Markov trace demonstrates rapid transitions in the first 2 model cycles; thereafter, the proportion of alive patients in each health state appeared stable over time. The majority of patients in the pegcetacoplan arm remained in the TA Hb ≥ 10.5 health state, fewer were in the TA Hb < 10.5 health state, and very few were in the transfusion required health state (Figure 1). In contrast, the majority of patients in the comparator treatment arms remained in the TA Hb < 10.5 and transfusion required health states, with nearly zero patients in the TA Hb ≥ 10.5 health state (Figure 2). Clinical expert feedback obtained by CADTH noted that these long-term predictions were optimistic. The majority of the incremental QALY gains for the pegcetacoplan group, as predicted by the model, arose from time spent in the TA Hb ≥ 10.5 health state.
CADTH was unable to address the limitation regarding the model being unable to capture all aspect of the disease.
Due to the uncertainty regarding the Hb threshold, CADTH conducted 2 scenario analyses in which equal utility values were assumed for both TA health states (TA Hb < 10.5 and TA Hb ≥ 10.5), and lower (0.738) and higher (0.809) utility values were used for both the TA Hb < 10.5 and TA Hb ≥ 10.5 health states.
Health state utility value estimation was inappropriate. The 3 Level EQ-5D utility weights used in this analysis were estimated by mapping the EORTC QLQ-C30 data from the PEGASUS trial. Because there is no mapping algorithm available for patients with PNH, a mapping algorithm developed for patients with cancer was employed.9 CADTH does not recommend the sponsor’s method of estimating health state utilities through mapping because utility values can vary greatly depending on the instruments being mapped, the algorithm used for mapping, and the severity of the included health states.12 Because patients with PNH do not share similar characteristics with patients with cancer, the use of a mapping algorithm developed for patients with cancer to generate utility values for patients with PNH is expected to lead to increased uncertainty.
CADTH was unable to address the uncertainty associated with utility estimates derived using mapping, given the paucity of alternate utility values. This calls into question the magnitude of the estimated incremental QALY gains associated with pegcetacoplan.
Eculizumab and pegcetacoplan dosing escalation is highly uncertain and may affect total treatment-related costs. The sponsor’s model considered that a proportion of patients on eculizumab or pegcetacoplan would be receiving a higher-dose regimen, based on data from the PEGASUS trial. With respect to eculizumab, the product monograph contains no approved dose escalation; the recommended maintenance dose regimen is 900 mg every 2 weeks.13 Yet, more than 30% of patients on eculizumab were assumed to be receiving an off-label up-dosing in the sponsor's model (i.e., 26.3%, 2.5% and 1.3% of patients received 1,200 mg every 14 days, 1,500 mg every 14 days, and 900 mg every 11 days, respectively).2 Based on the randomized controlled period of the PEGASUS trial, 4.9% of patients on pegcetacoplan were assumed to have had their dose adjusted to 1,080 mg every 3 days, as permitted in the product monograph.1 According to clinical expert feedback sought by CADTH for this review, there is uncertainty about the real-world distribution of patients on the different dose regimens. Some of the trial’s dose regimens are not commonly prescribed in Canada (i.e., 1,500 mg every 2 weeks for eculizumab). It was further noted that some public drug plans do not provide coverage for off-label dosing of eculizumab. Of note, use of the randomized controlled period to estimate the dosing frequency of pegcetacoplan is likely to result in an underestimation of pegcetacoplan drug costs, given that data from the open-label pegcetacoplan period suggest that more patients received escalated doses over time. Dose escalation increases drug acquisition costs and, given that the model’s main cost drivers are these parameters, alternative assumptions on dose escalation could significantly impact the expected cost difference and the cost-effectiveness of pegcetacoplan compared to a C5 inhibitor.
CADTH could not address this limitation in its base case due to the uncertainties regarding the proportion of patients who would require 1-off or continuous up-dosing, how dosage escalation would occur (e.g., higher dose given at the same administration intervals or a shortened interval between administrations), and whether up-dosing would be publicly reimbursed.
To explore uncertainty surrounding the reimbursement of up-dosing, CADTH performed scenario analyses in which up-dosing was assumed not to be reimbursed.
In addition, CADTH conducted a scenario analysis in which pegcetacoplan up-dosing from the PEGASUS trial for patients with BTH was assumed instead of a switch to eculizumab.
Limited validity in the transition probabilities of the comparators. The model assumed that patients on eculizumab would have different transition probabilities (i.e., clinical response) in the first 4 weeks of the model than beyond the first 4 weeks. According to clinical experts consulted by CADTH, given that this patient population had prior exposure to a C5 inhibitor, there is no rationale for patients to exhibit such a dramatic difference in clinical response to C5 inhibitors during these distinct time periods in the model.
CADTH conducted a base-case reanalysis that assumed identical transition probability matrices for the first and subsequent cycles of eculizumab and ravulizumab.
Optimistic assumptions regarding treatment discontinuation for pegcetacoplan. The model assumed that any treatment discontinuation due to AEs or lack of efficacy would happen only in the first year of treatment, based on the rates reported in the PEGASUS trial in the pegcetacoplan arm for week 1 to week 48 (randomized controlled period plus open-label pegcetacoplan period). According to the clinical experts consulted by CADTH, treatment discontinuation is not expected to be limited to only the first year of treatment. Indeed, treatment waning would be anticipated throughout the course of patient’s lifetime treatment, but was likely not observed in the trial given its limited study duration of 48 weeks.
To comprehensively consider all evidence available on pegcetacoplan, the CADTH base case selected discontinuation rates informed from week 1 to week 48 in the pegcetacoplan arm of the PEGASUS trial and from week 20 to week 48 in the eculizumab arm, when patients received open-label pegcetacoplan. CADTH was unable to adequately address treatment waning. If expected, the clinical efficacy of pegcetacoplan would be lower than predicted in the model and the incremental QALY gains would be smaller.
Additionally, the following key assumptions were made by the sponsor and have been appraised by CADTH (refer to Table 4).
Table 4: Key Assumptions of the Submitted Economic Evaluation
Sponsor’s key assumption | CADTH comment |
|---|---|
The efficacy of ravulizumab was considered to be equivalent to eculizumab. | Potentially reasonable. |
Background mortality was assumed to be equal to that of the general population, and there was no excess mortality risk associated with health states or complications. | Inappropriate. In the sponsor’s base case, background mortality was assumed to be equal to that of the general population, and the sponsor assumed that there was no excess mortality risk associated with health states or complications. The clinical experts consulted by CADTH noted that there is a mortality risk associated with PNH-related complications (such as BTH, thrombosis events), although the magnitude of the risk is uncertain. In addition, the clinical experts noted that the mortality rate of patients treated with complement inhibitors would not be expected to follow that of the general population, and there is limited validity in the average life expectancy predicted by the model for these patients (i.e.,76 years). |
The model assumed that patients with PNH were not at risk of developing PNH-related complications (acute kidney damage, chronic kidney disease, or pulmonary hypertension) if treated with either a C5 inhibitor or pegcetacoplan. | Inappropriate. The clinical experts consulted by CADTH noted that patients with PNH are always at risk of developing these complications, despite being on treatment with pegcetacoplan or a C5 inhibitor. However, the duration of the clinical trial that informed these input data is insufficient to adequately estimate the rates of these long-term complications. |
Costs of vaccines modelled were not included. | Inappropriate. The sponsor’s base case assumed that the cost of vaccines would be covered by federal programs and, therefore, set these costs as 0 in the model. As the model perspective is based on a publicly funded health care system, all costs borne by public systems should be captured, including vaccination costs. However, given that drug acquisition cost is the major driver of the results, the impact of omitting these costs is expected to be minimal. |
Costs of prophylactic antibiotics were not included. | Inappropriate. The sponsor’s base case did not model costs related to prophylactic antibiotics. This was inappropriate, as prophylactic antibiotics could be clinically indicated to manage serious infections. However, given that drug acquisition cost is the major driver of the results, the impact of omitting prophylactic antibiotic costs is expected to be minimal. |
BTH = breakthrough hemolysis; C5 = complement component 5; PNH = paroxysmal nocturnal hemoglobinuria.
CADTH Reanalyses of the Economic Evaluation
Base-Case Results
The CADTH base case was derived by making changes to parameter values, in consultation with clinical experts. CADTH reanalyses addressed several limitations in the economic model, as summarized in Table 5 and Table 6, which included assuming identical transition probability matrices for the first and subsequent cycles of eculizumab and ravulizumab and selecting discontinuation rate for pegcetacoplan based on more patient observations. CADTH was unable to address limitations regarding model structure and the method used to derive utility values.
Table 5: CADTH Revisions to the Submitted Economic Evaluation
Stepped analysis | Sponsor’s value or assumption | CADTH value or assumption |
|---|---|---|
Correctionsa to sponsor’s base case | ||
1.Utility values with limited face validity | The probabilistic distribution for the utility values overlapped, and there were instances in which sampling from the distribution produced illogical results with limited face validity (e.g., utility of TA Hb ≥ 10.5 g/dL less than transfusion required health state). | Corrected. CADTH added logic statements to the model. The probabilistically drawn utility value of the TA Hb ≥ 10.5 health state can only be greater or equal to the utility value for the TA Hb < 10.5 health state. Similarly, the probabilistically drawn utility value of the TA Hb < 10.5 health state can only be greater or equal to the utility value for the transfusion required health state. |
Changes to derive the CADTH base case | ||
1.Transition probabilities for the first cycle and subsequent cycles (eculizumab and ravulizumab) | Two different transitional probabilities were used: one for the first 4-weeks (based on the week 4 data from the PEGASUS trial); and the other for all cycles after the first 4 weeks (based on week 8 to week 16 of the trial) | Transition probabilities from week 8 to week 16 of the trial were used for all cycles (first and subsequent cycles) |
2.Source of pegcetacoplan discontinuation information | Randomized controlled period + open-label pegcetacoplan period | Randomized controlled period + open-label pegcetacoplan period (including patients from the eculizumab arm) |
CADTH base case | — | 1 + 2 |
TA Hb < 10.5 = transfusion avoidance and a hemoglobin level below 10.5 g/dL; TA Hb ≥ 10.5 = transfusion avoidance and a hemoglobin level of at least 10.5 g/dL.
aCorrections are minor programming errors that are not identified as limitations.
The results of CADTH’s stepped analysis are presented in Table 6. CADTH’s base-case reanalysis demonstrates that pegcetacoplan and ravulizumab dominated eculizumab (i.e., less costly, more effective). Specifically, pegcetacoplan resulted an incremental 2.91 QALY gain and a $1,427,177 cost savings compared with eculizumab. Compared with ravulizumab, the ICER of pegcetacoplan was $62,144 per QALY, and the probability that pegcetacoplan was cost-effective at a $50,000 per QALY WTP threshold was 46% of the probabilistic iterations.
Consistent with the sponsor’s submission, the majority (84%) of the QALYs for pegcetacoplan was accrued in the TA Hb ≥ 10.5 health state, and more than 99% of QALYs for both comparators were accrued from the transfusion required and TA Hb < 10.5 health states (Table 10). As in the sponsor’s base case, nearly all (> 99%) the total costs for pegcetacoplan and the comparators were drug costs (Table 10).
Table 6: Summary of the Stepped Analysis of the CADTH Reanalysis Results
Stepped analysis | Drug | Total costs ($) | Total QALYs | ICER ($/QALY) |
|---|---|---|---|---|
Sponsor’s base case (probabilistic) | Ravulizumab | 14,212,985 | 18.15 | Reference |
Pegcetacoplan | 14,261,211 | 20.11 | 24,636 | |
Eculizumab | 15,746,059 | 17.04 | Dominateda | |
Sponsor’s corrected base case (probabilistic) | Ravulizumab | 14,246,177 | 18.23 | Reference |
Pegcetacoplan | 14,289,935 | 20.15 | 22,816 | |
Eculizumab | 15,754,155 | 17.11 | Dominateda | |
CADTH reanalysis 1 (deterministic) | Ravulizumab | 14,156,057 | 18.08 | Reference |
Pegcetacoplan | 14,290,453 | 19.94 | 72,205 | |
Eculizumab | 15,747,137 | 16.89 | Dominateda | |
CADTH reanalysis 2 (deterministic) | Ravulizumab | 14,156,502 | 18.07 | Reference |
Pegcetacoplan | 14,349,415 | 19.82 | 110,675 | |
Eculizumab | 15,747,583 | 16.89 | Dominateda | |
CADTH base case (1 + 2) (deterministic) | Ravulizumab | 14,156,057 | 18.08 | Reference |
Pegcetacoplan | 14,349,410 | 19.82 | 111,038 | |
Eculizumab | 15,747,137 | 16.89 | Dominateda | |
CADTH base case (1 + 2) (probabilistic) | Ravulizumab | 14,246,172 | 18.24 | Reference |
Pegcetacoplan | 14,356,979 | 20.03 | 62,144 | |
Eculizumab | 15,784,157 | 17.12 | Dominateda |
ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year.
aDominated refers to a treatment having higher total costs and lower total QALYs compared to the previous less costly treatment.
Scenario Analysis Results
To address remaining uncertainty regarding parameterization of the model, CADTH conducted several scenario analyses. Full results are presented in Table 11. In all scenarios, eculizumab remained dominated (i.e., more costly, less effective). When an equal utility value was used for both TA Hb < 10.5 and TA Hb ≥ 10.5 health states, the ICER associated with pegcetacoplan increased from $103,441 to $313,336 per QALY, given that this reduced the magnitude of incremental QALY gains. The ICER increased to $416,365 per QALY when pegcetacoplan up-dosing for patients with BTH was allowed, instead of assuming that these patients would switch to eculizumab. This indicates the sensitivity of the model to mapped health utility values and the management of patients with BTH.
CADTH conducted a series of 2-way price-reduction analyses using the CADTH base case to examine the price changes at which pegcetacoplan would be considered cost-effective compared to ravulizumab and eculizumab. Under the CADTH base case, a 0.9% price reduction would be required for pegcetacoplan to be considered cost-effective at a WTP threshold of $50,000 per QALY if the price of the comparators remain at their publicly list price. However, the 2-way price-reduction analyses demonstrate how sensitive the model is to alternate drug acquisition costs (Table 7). This is noteworthy as the CADTH base-case analysis is based on the list price of eculizumab and ravulizumab and the confidentially negotiated price remains unknown.
Table 7: Two-Way Price-Reduction Analysis on the CADTH Base Case (Deterministic)
Price of pegcetacoplan | Price of eculizumab and ravulizumab | |||
|---|---|---|---|---|
List price | 20% reduction | 40% reduction | 60% reduction | |
Submitted price | At WTP threshold < $111,038 ravulizumab At WTP threshold ≥ $111,038 pegcetacoplan | At WTP threshold < $1,476,144 ravulizumab At WTP threshold ≥ $1,476,144 pegcetacoplan | At WTP threshold < $2,841,250 ravulizumab At WTP threshold ≥ $2,841,250 pegcetacoplan | At WTP threshold < $4,206,356 ravulizumab At WTP threshold ≥ $4,206,356 pegcetacoplan |
20% reduction | Pegcetacoplan dominant | At WTP threshold < $73,781 ravulizumab At WTP threshold ≥ $73,781 pegcetacoplan | At WTP threshold < $1,438,887 ravulizumab At WTP threshold ≥ $1,438,887 pegcetacoplan | At WTP threshold < $2,803,993 ravulizumab At WTP threshold ≥ $2,803,993 pegcetacoplan |
40% reduction | Pegcetacoplan dominant | Pegcetacoplan dominant | At WTP threshold < $36,525 ravulizumab At WTP threshold ≥ $36,525 pegcetacoplan | At WTP threshold < $1,401,631 ravulizumab At WTP threshold ≥ $1,401,631 pegcetacoplan |
60% reduction | Pegcetacoplan dominant | Pegcetacoplan dominant | Pegcetacoplan dominant | Pegcetacoplan dominant |
WTP = willingness to pay.
Issues for Consideration
Anticipated patent expiration of eculizumab: The patent for eculizumab is expected to expire on March 15, 2027.14 If eculizumab biosimilars become available and are considered clinically equivalent to eculizumab, pegcetacoplan is unlikely to remain less costly than eculizumab biosimilars. Consequently, the cost of pegcetacoplan at the submitted price would be less attractive to drug plans.
Comparator pricing based on publicly available prices: The modelled prices of eculizumab and ravulizumab are based on publicly accessible list prices and do not reflect any confidential pricing that may have been negotiated by public plans. As cost is the primary driver of this economic study, the cost savings of pegcetacoplan is, therefore, likely less than estimated if there are existing confidential discounts negotiated for the comparator(s).
Features of the PSP: CADTH submitted an additional information request to the sponsor to clarify the features of the purported PSP.15 The sponsor confirmed that some costs associated with pegcetacoplan would be covered (i.e., cost of subcutaneous infusion pump, needles, and patient and/or caregiver training).16 During the period for sponsor’s review and comment on the CADTH pharmacoeconomic report, the sponsor detailed that the PSP program will provide nurse-guided administration education and training, as well as logistical support to assist physicians and patients with the coordination of pretreatment vaccination.17 Costs associated replacement doses due to pump malfunction or user error were not addressed in the sponsor’s response.
Availability of infusion pumps: Pegcetacoplan requires specialized infusion pump systems (i.e., FreedomEdge). It remains unclear what options would be available if the FreedomEdge syringe infusion system is out of stock.
Up-dosing of pegcetacoplan increases LDH monitoring costs: The dosage of pegcetacoplan should be increased to 1,080 mg every 3 days if the patient has a significantly elevated LDH level. In the event of such a dose increase, it is recommended that LDH monitoring increases to twice weekly for at least 4 weeks.1 This may increase the overall health care costs associated with pegcetacoplan, which has not been captured in the sponsor’s model.
Intolerance to C5 inhibitors: The sponsor’s indication and requested reimbursement population for pegcetacoplan is patients with PNH who have an inadequate response to, or are intolerant of, C5 inhibitors. The sponsor’s economic model was instead based on the PEGASUS trial population, which only recruited patients who had an inadequate response to a C5 inhibitor. Although the cost-effectiveness of pegcetacoplan in the C5 inhibitor–intolerant population is unknown, clinical expert feedback consulted by CADTH noted that intolerance to C5 inhibitors is expected to be rare in clinical practice.
Overall Conclusions
The CADTH clinical review concluded that pegcetacoplan demonstrated clinically meaningful improvements in mean Hb level, compared with eculizumab, over 16 weeks of treatment in adult patients with PNH who had an inadequate response to eculizumab. TA was more frequently observed in the pegcetacoplan arm than in the eculizumab arm and, although only noninferiority could be concluded between treatments, the clinical experts considered the results to be supportive of the benefit of pegcetacoplan in improving anemia.
CADTH undertook reanalyses to address the key limitations in the sponsor’s analysis, which included adopting the same transition probability matrix for the first and subsequent cycles of eculizumab and ravulizumab and selecting a discontinuation rate for pegcetacoplan that was based on more patient observations. Based on the CADTH reanalysis, the results were largely consistent with the sponsors. In adult patients with PNH who have an inadequate response to C5 inhibitors, pegcetacoplan remained less costly and more effective compared with continued use of eculizumab. The ICER of pegcetacoplan compared to continued use of ravulizumab was $62,144 per QALY gained. A price reduction of 0.9% would be needed for pegcetacoplan to be cost-effective at a WTP threshold of $50,000 per QALY.
These results were largely driven by the drug acquisition costs of the comparators, which were based on publicly available list price as drug costs accounted for more than 99% of total costs across all 3 treatments. CADTH conducted a series of 2-way price-reduction analyses to highlight the point at which pegcetacoplan would no longer be considered cost-effective compared with eculizumab. If the confidential price of eculizumab is discounted by only 11.5% from its current list price, pegcetacoplan would no longer be cost-effective at a WTP threshold of $50,000 per QALY. Further uncertainties could not be addressed by CADTH, including up-dosing of eculizumab and pegcetacoplan, which, if reimbursed by public drug plans, would be expected to increase drug costs. In a scenario analysis in which patients with BTH are managed with pegcetacoplan up-dosing rather than a switch to eculizumab, the ICER of pegcetacoplan increased to $416,365 per QALY. Clinical efficacy was based on up-dosing rates reported in the PEGASUS trial, but the impact on costs and clinical outcomes if alternative up-dosing schedules are observed in the real-world clinical setting could not be addressed in the submitted model, which introduces uncertainty about this result.
With respect to clinical effectiveness, the model was heavily driven by the difference in the utility values between 2 health states: Hb levels of 10.5 g/dL or greater; and Hb levels of less than 10.5 g/dL. There is limited predictive validity on the meaningfulness of this cut-off value and, given the uncertainty associated with the method to derive utility estimates, it is unclear whether the estimated incremental QALYs produced by the economic model would be realized. A scenario analysis demonstrated that if the difference in utility values between these health states is reduced, there would be an increase in the ICER of pegcetacoplan and a larger price reduction would be warranted. Furthermore, the current analyses do not model waning in any of the treatments, but this is expected to be larger in the pegcetacoplan arm given patients would transition to health states associated with lower utilities. As such, if treatment waning had been incorporated, the incremental QALY gains would be smaller, leading to a higher ICER estimate for pegcetacoplan.
Other limitations in the sponsor’s model that could not be adequately addressed were related to the duration of the clinical evidence available and the model conceptualization of the condition. For instance, discontinuation of pegcetacoplan was assumed to only occur in the first year of the treatment. This does not align with the feedback CADTH got from clinical experts, which noted that treatment waning is anticipated over time and would continue beyond 1 year of treatment. Furthermore, the sponsor model did not capture all aspects of the disease conditions (such as thrombosis), given the trial duration was too limited to identify these events.
References
1.PrEmpaveliTM (pegcetacoplan): solution for infusion: 54 mg/mL solution for infusion for subcutaneous use [draft product monograph]. Stockholm (SE): Swedish Orphan Biovitrum AB; 2022.
2.Pharmacoeconomic evaluation [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Pegcetacoplan (brand name TBC) 54 mg/mL solution for subcutaneous infusion Oakville (ON): Sobi Canada Inc; 2022 Aug 26.
3.Clinical Study Report: APL2-302. A phase 3, randomized, multicenter, open-label, active-comparator controlled study to evaluate the efficacy and safety of pegcetacoplan in patients with paroxysmal nocturnal hemoglobinuria (PNH) week 16 analysis [internal sponsor's report]. Waltham (MA): Apellis Pharmaceuticals; 2020.
4.Clinical Study Report: APL2-302. A phase 3, randomized, multicenter, open-label, active-comparator controlled study to evaluate the efficacy and safety of pegcetacoplan in patients with paroxysmal nocturnal hemoglobinuria (PNH) week 48 analysis [internal sponsor's report]. Waltham (MA): Apellis Pharmaceuticals; 2021.
5.Canada PMPRB. Board order - Soliris 2022 Jul 21; https://www.canada.ca/en/patented-medicine-prices-review/services/hearings/decisions-and-orders/board-order-soliris.html. Accessed 2022 Jul 25.
6.CADTH reimbursement recommendation: Ravulizumab (ultomiris) Can J Health Technol. 2022;2(3). https://www.cadth.ca/sites/default/files/DRR/2022/SR0700%20Ultomiris%20-%20Final%20CADTH%20Rec.pdf. Accessed 2022 Nov 25.
7.Hillmen P, Szer J, Weitz I, et al. Pegcetacoplan versus eculizumab in paroxysmal nocturnal hemoglobinuria. N Engl J Med. 2021;384(11):1028-1037. PubMed
8.Kelly RJ, Hill A, Arnold LM, et al. Long-term treatment with eculizumab in paroxysmal nocturnal hemoglobinuria: sustained efficacy and improved survival. Blood. 2011;117(25):6786-6792. PubMed
9.Longworth L, Yang Y, Young T, et al. Use of generic and condition-specific measures of health-related quality of life in NICE decision-making: a systematic review, statistical modelling and survey. Health Technol Assess. 2014;18(9):1-224. PubMed
10.Ontario Case Costing. OCC costing analysis tool. https://hsimi.ca/occp/occpreports/Expire.aspx. Accessed 2022 Sep 24.
11.Schedule of benefits for physician services under the Health Insurance Act: effective October 26, 2015. Toronto (ON): Ontario Ministry of Health. Accessed 2022 Sep 24.
12.Guidelines for the economic evaluation of health technologies Ottawa (ON): CADTH; 2017: https://www.cadth.ca/guidelines-economic-evaluation-health-technologies-canada-4th-edition. . Accessed 2022 Nov 18.
13.PrSoliris® (eculizumab): 30 mL parenteral solution (10 mg/mL) [product monograph]. Zurich (CH): Alexion Pharma GmbH; 2021 Mar 25.
14.Health Canada. Patent register. https://pr-rdb.hc-sc.gc.ca/pr-rdb/patent_result-resultat_brevet.do?action=search_recherche&formId=10092&din=02322285&drugId=2548&lang=fr&patentNumber_numeroBrevet=2645810. Accessed 2022 Nov 22.
15.CADTH request for additional information regarding pegcetacoplan (brand name TBC) DRR review Ottawa (ON): CADTH; 2022 Sep 27.
16.Sobi Canada Inc. response to October 31 2021 DRR request for additional information regarding pegcetacoplan (brand name TBC) DRR review [internal sponsor's report] Oakville (ON): Sobi Canada Inc;; 2022 Nov 10.
17.Sobi Canada Inc. Comments to Pharmacoeconomic review report regarding pegcetacoplan (brand name TBC) [internal sponsor's report]. Oakville (ON): Sobi Canada Inc; 2022 Dec 8.
18.Government of Alberta. Interactive drug benefit list. https://idbl.ab.bluecross.ca/idbl/load.do. . Accessed 2022 Oct 23.
Appendix 1: Cost Comparison Table
Note this appendix has not been copy-edited.
The comparators presented in the following table have been deemed to be appropriate based on feedback from clinical experts and drug plan. Comparators may be recommended (appropriate) practice or actual practice. Existing Product Listing Agreements are not reflected in the table and as such, the table may not represent the actual costs to public drug plans.
Table 8: CADTH Cost Comparison Table for PNH
Treatment | Strength / concentration | Form | Price | Recommended dosage | Daily cost | Annual costa |
|---|---|---|---|---|---|---|
Pegcetacoplan | 54 mg/mL | 20 mL single-dose vial for subcutaneous infusion | $4,970.0000 | First 4 weeks: 1,080 mg twice weekly in addition to patient’s current dose of C5i Subsequent weeks: 1,080 mg twice weekly | $1,416.11a | $516,880 |
Pegcetacoplan + first 4 weeks of C5i (refer to maintenance costs below) | First 4 weeksb: $1,416.11 to $2,660.39 Subsequent weeksa: $1,416.00 | First year costsb: $516,880 to $556,932 Subsequent year costa: $516,880 | ||||
Compliment Component 5 inhibitors (C5i) | ||||||
Eculizumab (Soliris) | 10 mg/mL | 300 mg single-use vial for IV infusion | $6,675.3000c | Loading: 600 mg every 7 days for the first 4 weeks, then 900 mg for the fifth dose 1 week later Maintenance: 900 mg every 2 weeks thereafter | Year 1d: $1,517.94 Subsequent yearse: $1,426.50 | Year 1d: $554,050 Subsequent yearse: $520,673 |
Ravulizumab (Ultomiris) | 10 mg/mL | 30 mL single-dose vial for IV infusion | $7,296.6700 | Loading dose, with maintenance doses given starting 2 weeks after, then administered every 8 weeks thereafter, based on weight as followsf: ≥ 40 kg to < 60 kg: Loading: 2,400 mg; Maintenance: 3,000 mg | ≥ 40 kg to < 60 kg: Year 1g: $1,559.29 Subsequent years h: $1,299.41 | ≥ 40 kg to < 60 kg: Year 1g: $569,140 Subsequent yearsh: $474,284 |
≥ 60 kg to < 100 kg Loading: 2,700 mg; Maintenance: 3,300 mg | ≥ 60 kg to 100 kg: Year 1 g: $1,719.22 Subsequent years h: $1,429.35 | ≥ 60 kg to < 100 kg: Year 1g: $627,514 Subsequent yearsh: $521,712 | ||||
≥ 100 kg Loading: 3,000 mg; Maintenance: 3,600 mg | ≥ 100 kg: Year 1g: $1,879.14 Subsequent yearsh: $1,559.29 | ≥ 100 kg: Year 1g: $685,887 Subsequent yearsh: $569,140 | ||||
aCosts assume 104 1,080 mg doses.
bYear 1 assume 6 300 mg doses of eculizumab and 104 1,080mg doses of pegcetacoplan; or 104 1,080 mg doses of pegcetacoplan only (assumed patient would receive pegcetacoplan treatment during the last 4 weeks of the 8-week ravulizumab treatment cycle).
cAlberta drug formulary (accessed September 2022).18
dYear 1 costs assume four 600 mg doses and twenty-five 900 mg doses.
eSubsequent year costs assume 26 administrations per year.
fFor patients switching from eculizumab, the loading dose of ravulizumab is given 2 weeks after the last eculizumab infusion. Maintenance doses are then given every 8 weeks, starting 2 weeks after the loading dose.
gYear 1 costs assume 1 loading dose and 7 maintenance doses.
hSubsequent year dosing are based on an average of 6.5 administrations (52/8) per year.
Appendix 2: Submission Quality
Note this appendix has not copy-edited.
Description | Yes/No | Comments |
|---|---|---|
Population is relevant, with no critical intervention missing, and no relevant outcome missing | No | Thrombosis has been identified as an important outcome by clinical experts consulted by CADTH and inputs from clinicians, however this was not explicitly modelled (refer to CADTH key limitations). |
Model has been adequately programmed and has sufficient face validity | No | CADTH identified and corrected a programming error (refer to corrected base-case results). The probabilistic distribution for the utility values overlapped and there were instances where sampling from the distribution produced illogical results with limited face validity (e.g., utility of TA Hb ≥ 10.5 less than transfusion required state). |
Model structure is adequate for decision problem | No | Refer to CADTH key limitations. |
Data incorporation into the model has been done adequately (e.g., parameters for probabilistic analysis) | Yes | No comment. |
Parameter and structural uncertainty were adequately assessed; analyses were adequate to inform the decision problem | No | Clinical experts consulted by CADTH noted that switching from pegcetacoplan back to a C5i may depends on multiple factors. Some patients may remain on pegcetacoplan despite inadequate response; however, the submitted model does not provide options to explore the impact of when patients continue to remain on pegcetacoplan. |
The submission was well organized and complete; the information was easy to locate (clear and transparent reporting; technical documentation available in enough details) | Yes | No comment. |
Appendix 3: Additional Information on the Submitted Economic Evaluation
Note this appendix has not been copy-edited.
Figure 3: Markov Trace for Eculizumab and Ravulizumab
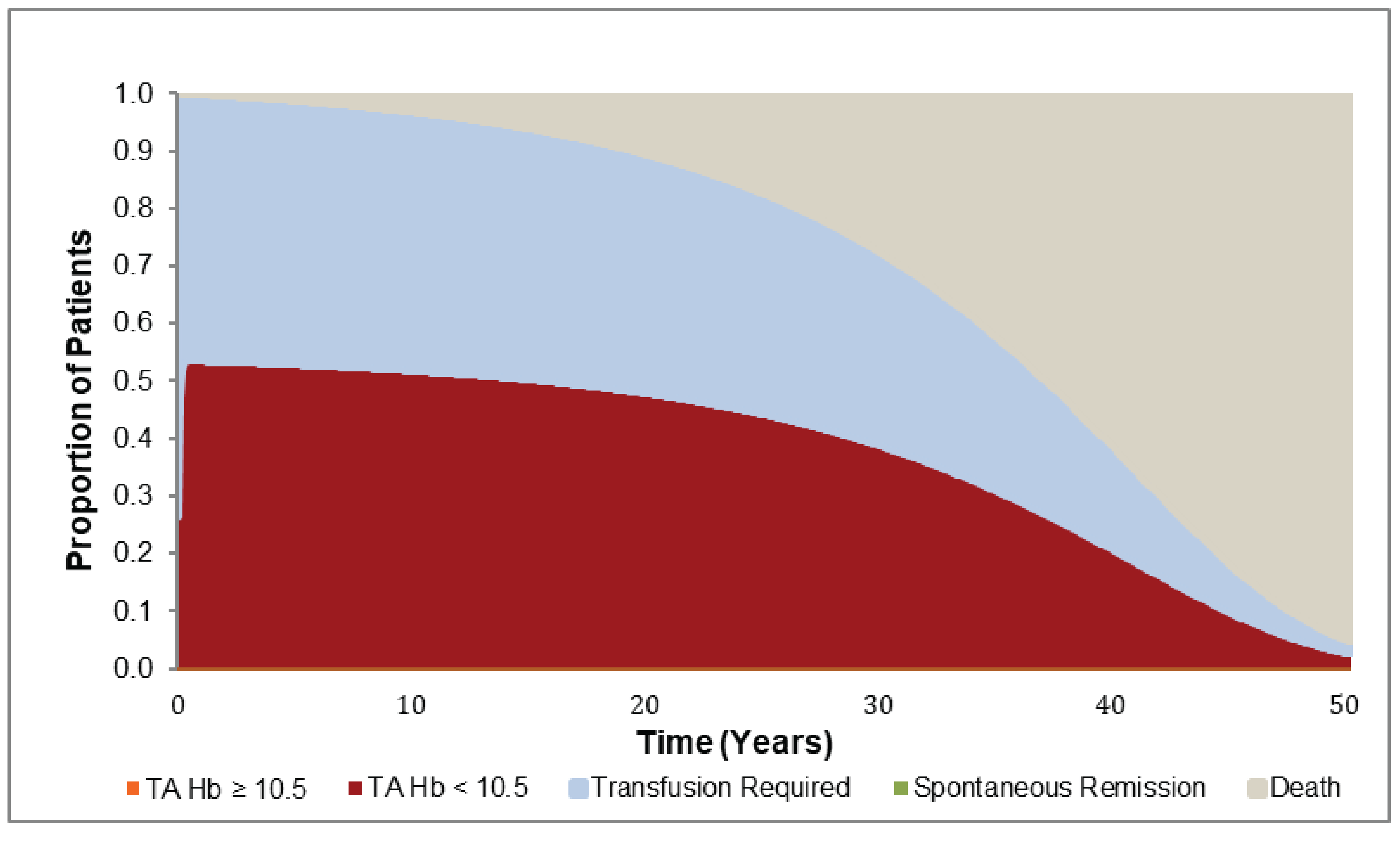
Source: Sponsor’s pharmacoeconomic submission.2
Appendix 4: Additional Details on the CADTH Reanalyses and Sensitivity Analyses of the Economic Evaluation
Note this appendix has not been copy-edited.
Table 10: Disaggregated Summary of CADTH’s Economic Evaluation Results
Treatment | Component | Value | Incremental (vs. ravulizumab) | Incremental (sequential) |
|---|---|---|---|---|
Discounted LYs | ||||
Ravulizumab | TA Hb < 10.5 | 14.466 | 0.000 | Reference |
TA Hb ≥ 10.5 | 0.024 | 0.000 | Reference | |
Transfusion required | 12.482 | 0.000 | Reference | |
Total | 26.972 | 0.000 | Reference | |
Pegcetacoplan | TA Hb < 10.5 | 3.075 | −11.390 | −11.390 |
TA Hb ≥ 10.5 | 22.145 | 22.122 | 22.122 | |
Transfusion required | 1.751 | −10.732 | −10.732 | |
Total | 26.971 | 0.000 | 0.000 | |
Eculizumab | TA Hb < 10.5 | 14.466 | 0.000 | 11.390 |
TA Hb ≥ 10.5 | 0.024 | 0.000 | −22.122 | |
Transfusion required | 12.482 | 0.000 | 10.732 | |
Total | 26.972 | 0.000 | 0.000 | |
Discounted QALYs | ||||
Ravulizumab | TA Hb < 10.5 | 10.096 | 0.000 | Reference |
TA Hb ≥ 10.5 | 0.018 | 0.000 | Reference | |
Transfusion required | 8.203 | 0.000 | Reference | |
Disutility associated with IV infusion | −0.072 | 0.000 | Reference | |
Total | 18.244 | 0.000 | Reference | |
Pegcetacoplan | TA Hb < 10.5 | 2.147 | −7.949 | −7.949 |
TA Hb ≥ 10.5 | 16.895 | 16.877 | 16.877 | |
Transfusion required | 1.147 | −7.056 | −7.056 | |
Disutility associated with IV infusion | −0.162 | −0.09 | −0.09 | |
Total | 20.028 | 1.784 | 1.784 | |
Eculizumab | TA Hb < 10.5 | 10.096 | 0 | 7.949 |
TA Hb ≥ 10.5 | 0.018 | 0 | −16.877 | |
Transfusion required | 8.203 | 0 | 7.056 | |
Disutility associated with IV infusion | −1.194 | −1.122 | −1.032 | |
Total | 17.123 | −1.121 | −2.905 | |
Discounted costs ($) | ||||
Ravulizumab | Drug acquisition | 14,066,348 | 0.000 | Reference |
Supportive treatment | 35,580 | 0.000 | Reference | |
Vaccination | 0 | 0.000 | Reference | |
Administration | 0 | 0.000 | Reference | |
Adverse events | 925 | 0.000 | Reference | |
Health state cost | 143,319 | 0.000 | Reference | |
Total | 14,246,172 | 0.000 | Reference | |
Pegcetacoplan | Drug acquisition | 14,308,346 | 241,998 | 241,998 |
Supportive treatment | 8,917 | −26,663 | −26,663 | |
Vaccination | 0 | 0 | 0 | |
Administration | 0 | 0 | 0 | |
Adverse events | 5,418 | 4,493 | 4,493 | |
Health state cost | 34,298 | −109,021 | −109,021 | |
Total | 14,356,979 | 110,807 | 110,807 | |
Eculizumab | Drug acquisition | 15,598,050 | 1,531,702 | 1,289,704 |
Supportive treatment | 35,580 | 0 | 26,663 | |
Vaccination | 0 | 0 | 0 | |
Administration | 0 | 0 | 0 | |
Adverse events | 8,152 | 7,227 | 2,734 | |
Health state cost | 142,374 | −945 | 108,076 | |
Total | 15,784,157 | 1,537,985 | 1,427,178 | |
ICER = incremental cost-effectiveness ratio; LY = life-year; QALY = quality-adjusted life-year; vs. = versus.
aDominated refers to a treatment having higher total costs and lower total QALYs when compared to the previous less costly treatment.
aDominated means eculizumab treatment resulted in higher total costs and lower total QALYs compared to ravulizumab.
Scenario Analyses
Table 11: CADTH Scenario Analyses
Stepped analysis | Drug | Total costs ($) | Total QALYs | ICER ($/QALYs) |
|---|---|---|---|---|
CADTH base case | Ravulizumab | 14,246,172 | 18.244 | Reference |
Pegcetacoplan | 14,356,979 | 20.028 | 62,144 | |
Eculizumab | 15,784,157 | 17.123 | Dominateda | |
1. Run-in period cost for pegcetacoplan is covered by the sponsor | Ravulizumab | 14,232,934 | 18.235 | Reference |
Pegcetacoplan | 14,248,865 | 20.138 | 8,373 | |
Eculizumab | 15,737,263 | 17.119 | Dominateda | |
2. Lower utility value for both TA Hb < 10.5 and TA Hb ≥ 10.5 (0.738) | Ravulizumab | 14,248,147 | 18.13609 | Reference |
Pegcetacoplan | 14,357,157 | 18.48399 | 313,336 | |
Eculizumab | 15,768,131 | 17.01503 | Dominateda | |
3. Higher utility value for both TA Hb < 10.5 and TA Hb ≥ 10.5 (0.809) | Ravulizumab | 14,268,511 | 19.105 | Reference |
Pegcetacoplan | 14,376,380 | 20.148 | 103,441 | |
Eculizumab | 15,814,927 | 17.976 | Dominateda | |
4. No up-dosing of eculizumab funded by public drug plans | Ravulizumab | 14,232,852 | 18.234 | Reference |
Pegcetacoplan | 14,284,549 | 20.149 | 27,009 | |
Eculizumab | 15,765,727 | 17.115 | Dominateda | |
5. No up-dosing of eculizumab and pegcetacoplan funded by public drug plans | Pegcetacoplan | 14,195,597 | 20.147 | Reference |
Ravulizumab | 14,250,913 | 18.240 | Dominateda | |
Eculizumab | 15,758,020 | 17.126 | Dominateda | |
6. Pegcetacoplan up-dosing for patients with BTH instead of switching to eculizumab | Ravulizumab | 14,156,502 | 18.073 | Reference |
Pegcetacoplan | 14,929,983 | 19.930 | 416,365 | |
Eculizumab | 15,747,583 | 16.886 | Dominateda |
BTH = breakthrough hemolysis; ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year.
aDominated refers to a treatment having higher total costs and lower total QALYs when compared to the previous less costly treatment.
Appendix 5: Submitted BIA and CADTH Appraisal
Note this appendix has not been copy-edited.
Table 12: Summary of Key Take-Aways
Key take-aways of the BIA |
|---|
|
Summary of Sponsor’s BIA
The sponsor submitted a budget impact analysis (BIA) estimating the budget impact of introducing pegcetacoplan for the treatment of adult patients with PNH who have an inadequate response to, or are intolerant of, a C5i treatment (eculizumab and ravulizumab). The base case of the BIA reflects the full Health Canada indication and the reimbursement requested population. The analysis was undertaken from a Canadian public drug plan payer perspective over a 3-year time horizon using an epidemiological approach. The sponsor’s base-case analysis included drug acquisition costs, as well as administrative costs of pegcetacoplan. Data inputs informing the BIA were obtained from the literature and assumptions. Key inputs to the BIA are documented in Table 13.
Key assumptions include:
The BIA model considered continuous up-dosing of pegcetacoplan and eculizumab in more than 4.9% and 30% of patients, respectively, based on data from the PEGASUS trial.3,4,7
The annual cost for ravulizumab was calculated assuming an average weight of 75.25 kg based on the average weight of patients enrolled in the PEGASUS trial.
The BIA model assumed that the first 4 weeks run-in period cost of pegcetacoplan would be reimbursed by public drug plans.
Patients switching from eculizumab or ravulizumab to pegcetacoplan were assumed to require 2 additional vaccines before beginning treatment (i.e., vaccines for S. pneumoniae and H. influenzae). This was assumed to be covered by federal plans and such costs were not included in the sponsor’s base-case analysis.
Table 13: Summary of Key Model Parameters
Parameter | Sponsor’s estimate (reported as year 1 / year 2 / year 3) |
|---|---|
Target population | |
Prevalence of PNH | 13.95 per million |
Proportion of patients with PNH treated with C5i | 39.3% |
Proportion of patients with PNH with inadequate response to C5i treatment | 43.4% |
Proportion of patients with PNH who are intolerant of a C5i treatment | 5% |
Percent of patients with PNH with inadequate response to a C5i treatment that require public drug plan coverage | 50% |
Number of patients eligible for drug under review | 42.2 / 42.7 / 43.2 |
Market uptake (3 years) | |
Uptake (reference scenario) | |
Eculizumab | 72% / 61% / 51% |
Ravulizumab | 28% / 39% / 49% |
Uptake (new drug scenario) | |
Pegcetacoplan | 80% / 90% / 95% |
Eculizumab | 14.4% / 6.1% / 2.6% |
Ravulizumab | 5.6% / 3.9% / 2.5% |
Cost of treatment (per patient) | |
Cost of treatment over a year | |
Pegcetacoplana | $522,514 |
Pegcetacoplan + first 4 weeks of C5i (Maintenance dose) | $562,646b to $566,874c |
Eculizumab (Maintenance dose)d | $578,270 |
Ravulizumab (Maintenance dose) | $523,145 |
PNH = Paroxysmal nocturnal hemoglobinuria; C5i = Complement 5 inhibitor
aDose-weighted annual cost of pegcetacoplan (4.9% of patients required up-dosing from 1,080 mg twice a week to 1,080 mg every 3 days).
bDose-weighted annual cost of pegcetacoplan plus half of the 8-week ravulizumab treatment cycle during the switch period (assuming pegcetacoplan treatment would start during the last 4 weeks of the ravulizumab treatment cycle).
cDose-weighted annual cost of pegcetacoplan plus weighted cost of eculizumab maintenance dose calculated based on a weighted average of the Health Canada–approved label and off-label dosing regimen.
dWeighted cost of eculizumab maintenance dose calculated based on a weighted average of the Health Canada–approved label and off-label dosing regimen.
Summary of the Sponsor’s BIA Results
The sponsor estimated that the reimbursement of pegcetacoplan for the treatment of adult patients with PNH who have an inadequate response to, or intolerance of, a C5i treatment will be associated with an incremental cost of $96,345 in year 1, and incremental cost savings of $1,116,395 in year 2, and $1,068,707 in year 3, for a 3-year total budget saving of $2,088,756.
CADTH Appraisal of the Sponsor’s BIA
CADTH identified several key limitations to the sponsor’s analysis that have notable implications on the results of the BIA:
The number of PNH patients eligible for C5i treatment and intolerance may be overestimated. The sponsor estimated that approximately 172 PNH patients would be treated with C5i in the baseline year and, among these patients, approximately 75 would have inadequate response. This was based on the estimate that, among patients with PNH, 39.3% would be treated with a C5i of whom 43.4% of patients would have inadequate response while 5% of patients would be intolerant. However, clinical experts consulted by CADTH for this review estimated that 20 to 30% of PNH patients had inadequate response to C5i and few, if any, would be intolerant.
In CADTH reanalysis, the proportion of new patients with inadequate response to, or intolerance to C5i therapy was assumed to be 30% and 0%, respectively; based on the feedback by the clinical experts sought by CADTH.
The anticipated uptake of pegcetacoplan and ravulizumab is not aligned with clinical expert expectations. The sponsor estimated that pegcetacoplan would have a market share of 80% / 90% / 95% in year 1, 2, and 3, respectively. However, clinical experts consulted by CADTH for this review felt that the sponsor’s market share for pegcetacoplan was too aggressive. If ravulizumab is publicly funded, the clinical experts expected the market share in 3 years would be split between pegcetacoplan and ravulizumab.
In CADTH reanalysis, the proportion of eligible patients who will use pegcetacoplan in year 1, 2, and 3 were changed to reflect two-thirds of the population not on eculizumab. Recognizing the uncertainties in the anticipated market shares, CADTH further conducted a scenario analysis based on the sponsor’s market share estimates.
Eculizumab and pegcetacoplan dosing escalation. The BIA assumed that more than 30% of patients on eculizumab and 4.9% on pegcetacoplan would be receiving a higher-dose regimen, based on data from the PEGASUS trial.3,7 The up-dosing of eculizumab was not aligned with its product monograph.13 According to clinical expert feedback sought by CADTH for this review, there is uncertainty to the real-world distribution of patients on the different dose regimens. It was further noted that some public drug plans do not provide coverage for off-label dosing of eculizumab. Dose escalation increases the drug acquisition costs and, given the model main cost driver is this parameter, alternative assumptions on dose escalation could significantly impact the BIA results.
CADTH performed 2 scenario analyses assuming up-dosing would not to be publicly reimbursed: i) no up-dosing of eculizumab, however up-dosing of pegcetacoplan was permitted; ii) no up-dosing of both pegcetacoplan and eculizumab.
The price of drugs paid by public drug plans is uncertain: Both the sponsor’s and CADTH’s analyses are based on publicly available list prices for all comparators. Actual costs paid by public drug plans are unknown. Confidential negotiated prices for comparators may lead to budgetary savings being limited or eliminated. Further, there is uncertainty on who will cover the first 4 weeks run-in period cost for pegcetacoplan, despite the sponsor’s assumption that it would be covered by the public drug plans.
CADTH conducted a scenario analysis assuming the run-in period cost for pegcetacoplan would be covered by the sponsor.
CADTH Reanalyses of the BIA
CADTH revised the sponsor’s base case by decreasing the proportion of patients with an inadequate response to or intolerance of C5i treatment and changing the market uptake in the new drug scenario. Table 14 notes the assumptions used by the sponsor in comparison to those used by CADTH in the reanalysis.
Applying these changes decreased the magnitude of the 3-year budget savings to $863,569. The results of the CADTH step-wise reanalyses are presented in summary format in Table 15 and a more detailed breakdown is presented in Table 15.
To address remaining uncertainty, CADTH conducted several scenario analyses and full results are presented in Table 16. In 3 years, the CADTH assumption of no up-dosing of pegcetacoplan and eculizumab resulted in an incremental cost of $1,167,005. The assumption that the sponsor will cover the first 4 weeks of treatment with pegcetacoplan was associated with a 3-year total budget savings of $1,537,829. When uptake of pegcetacoplan was anticipated to be aligned with the sponsor’s original estimates (e.g., 95% in 3 years), it resulted a 3-year total budget saving of $1,295,353.
Table 14: CADTH Revisions to the Submitted BIA
Stepped analysis | Sponsor’s value or assumption | CADTH value or assumption |
|---|---|---|
Changes to derive the CADTH base case | ||
1.a. PNH patients with an inadequate response to a C5i treatment | 43.4% | 30% |
1.b. PNH patients who are intolerant of C5i treatment | 5% | 0% |
2. Uptake of: Ravulizumab Pegcetacoplana | 5.6% / 3.9% / 2.5% 80% / 90% / 95% | 13.1% / 16.6% / 18% 53.3% / 60% / 63.3% |
CADTH base case | 1a + 1b + 2 | |
PNH = paroxysmal nocturnal hemoglobinuria; C5i = Complement 5 inhibitor.
aCADTH market share for pegcetacoplan was assumed to be 2/3 of the sponsor’s market share, based on clinical experts’ feedback that the market share would be split between ravulizumab and pegcetacoplan, if ravulizumab is publicly funded.
Table 15: Summary of the CADTH Reanalyses of the BIA
Stepped analysis | Three-year total |
|---|---|
Submitted base case | ($2,088,756) |
CADTH reanalysis 1a – inadequate response to C5i treatment | ($1,511,245) |
CADTH reanalysis 1b – intolerance to C5i treatment | ($1,872,864) |
CADTH reanalysis 2 – uptake of pegcetacoplan and ravulizumab | ($1,392,504) |
CADTH base case | ($863,569) |
BIA = budget impact analysis; C5i = Complement 5 inhibitor.
Note: The costs in bracket represent incremental cost savings.
Table 16: Detailed Breakdown of the CADTH Reanalyses of the BIAa
Stepped analysis | Scenario | Year 0 (current situation) | Year 1 | Year 2 | Year 3 | Three-year total |
|---|---|---|---|---|---|---|
Submitted base case | Reference | $23,754,924 | $23,737,950 | $23,760,854 | $23,801,489 | $71,300,293 |
New drug | $23,754,924 | $23,834,295 | $22,644,459 | $22,732,782 | $69,211,537 | |
Budget impact | $0 | $96,345 | ($1,116,395) | ($1,068,707) | ($2,088,756) | |
CADTH base case | Reference | $14,731,736 | $14,721,209 | $14,735,413 | $14,760,613 | $44,217,236 |
New drug | $14,731,736 | $14,761,042 | $14,273,855 | $14,318,771 | $43,353,668 | |
Budget impact | $0 | $39,833 | ($461,559) | ($441,843) | ($863,569) | |
CADTH scenario 1a: No up-dosing of eculizumab | Reference | $13,497,868 | $13,663,500 | $13,828,665 | $13,993,622 | $41,485,787 |
New drug | $13,497,868 | $14,224,170 | $13,906,083 | $14,035,170 | $42,165,423 | |
Budget impact | $0 | $560,670 | $77,418 | $41,548 | $679,636 | |
CADTH scenario 1b: No up-dosing of pegcetacoplan and eculizumab | Reference | $13,497,868 | $13,663,500 | $13,828,665 | $13,993,622 | $41,485,787 |
New drug | $13,497,868 | $14,165,389 | $13,839,170 | $13,963,711 | $41,968,270 | |
Budget impact | $0 | $501,889 | $10,505 | ($29,911) | $482,483 | |
CADTH scenario 2: Sponsor covers run-in period cost for pegcetacoplan | Reference | $14,731,736 | $14,721,209 | $14,735,413 | $14,760,613 | $44,217,236 |
New drug | $14,731,736 | $14,206,407 | $14,197,121 | $14,275,879 | $42,679,407 | |
Budget impact | $0 | ($514,803) | ($538,292) | ($484,734) | ($1,537,829) | |
CADTH Scenario 3: Uptake of pegcetacoplan as per sponsor’s original estimates | Reference | $14,731,736 | $14,721,209 | $14,735,413 | $14,760,613 | $44,217,236 |
New drug | $14,731,736 | $14,780,958 | $14,043,075 | $14,097,849 | $42,921,883 | |
Budget impact | $0 | $59,749 | ($692,338) | ($662,764) | ($1,295,353) |
BIA = budget impact analysis
aThe costs in bracket represent incremental cost savings.
Stakeholder Input
Patient Input
Canadian Association of PNH Patients and Aplastic Anemia & Myelodysplasia Association of Canada
About the Canadian Association of PNH Patients and Aplastic Anemia & Myelodysplasia Association of Canada
The Canadian Association of PNH Patients
This patient advocacy group is a not-for-profit Canadian organization formed in 2009. The mission of the organization is to connect Canadians affected by Paroxysmal Nocturnal Haemoglobinuria (PNH) and advocate for the best possible care for patients and ensure they are equipped with the most current tools and information to help them live well with the condition. The organization also provides support to caregivers and works to increase awareness and understanding of PNH.
The Canadian Association of PNH Patients was founded by Barry Katsof who is a PNH patient himself. He founded the patient group because he realized little to no support was granted to patients who require access to life sustaining medications. While his journey of gaining access to the first biologic was incredibly successful through self-advocacy, he now applies his learnings to supporting every PNH Canadians patients who may be in a similar situation that he found himself in back in 2007. The website is: http://www.pnhca.org
Aplastic Anemia & Myelodysplasia Association of Canada (AAMAC)
AAMAC was founded in 1987 by a concerned parent of a child diagnosed with aplastic anemia. One of the primary goals of the association was to advocate for the formation of a national bone marrow donor registry in Canada. Today, AAMAC is a federally incorporated and a registered national charity with the ambitious goal of providing a seamless support network for every Canadian patient, family member, friend and concerned healthcare provider dealing with aplastic anemia, myelodysplasia or PNH. The website: https://aamac.ca/
About PNH
Paroxysmal Nocturnal Haemoglobinuria is a very rare and debilitating disease of the bone marrow that affects the blood and major organs. It is a chronic, life-threatening illness and can have devastating effects upon a patient physically, mentally and emotionally. PNH has been estimated to affect as many as sixteen individuals per million worldwide although its global prevalence is understudied1. Most patients living with PNH suffer from impaired health-related quality of life2.
Information Gathering
The following submission reflects data from a total of six individuals impacted by PNH. Only three patients interviewed are being treated with pegcetacoplan through clinical trials, and the remaining patients are currently being treated with eculizumab despite the fact their PNH is manifested as extravascular hemolysis (EVH). We sought the opinion on the value of having pegcetacoplan approved for wide use in Canada. A qualitative descriptive approach, employing the technique of constant comparison, was used to produce a thematic analysis. We are citing scientific literature and peer-reviewed journals on pegcetacoplan and its impacts on quality of life (blood-transfusion dependence), and head-to-head comparison to eculizumab (because this molecule is currently the standard approach for all PNH patients).
Disease Experience
Paroxysmal nocturnal hemoglobinuria (PNH) is a rare, acquired, clonal hematopoietic stem cell disorder characterized by complement-mediated hemolysis (with or without hemoglobinuria), and some degree of bone marrow dysfunction3. This life-threatening disease can appear at any age and in any race or gender and is most often diagnosed in people in their early 30s3. Hemolysis occurs in PNH through two mechanisms (pathways): intravascular hemolysis (IVH), which occurs inside blood vessels, and extravascular hemolysis (EVH), which occurs in the liver and spleen. Thrombosis is of the most devastating consequence of hemolysis in PNH, as it can damage organs and cause premature death4. Thrombosis can occur in blood vessels throughout the body, and the first thrombotic event can be fatal4. As a matter of fact, most PNH-related deaths are due to thrombotic events5.
Two out of the five patients we have interviewed have experienced thrombosis, which led to having a stroke in both cases.
C5 inhibitors reduce IVH by inhibiting membrane attack complex formation and are the current standard of treatment for PNH. This explains why patients currently on a C5 (i.e., eculizumab) and whom, are known to manifest symptoms indicating their PNH is EVH will most likely not respond to this treatment. One important indication is blood transfusion dependant. We have interviewed Canadian patients who are currently on pegcetacoplan through clinical trial. All have described this treatment as a game changer- the utmost therapy that changed their lives for the best quality of life considering that in 2019 and earlier, they were dependant on blood transfusion and their lives were defined by PNH. Thanks for the results of ongoing efforts to better understand PNH, we now know this occurs because EVH persists in the presence of C5 inhibition. These C5 inhibitors have improved patient survival by addressing IVH, yet despite this, many people with PNH continue to experience ongoing hemolysis and persistently low hemoglobin6. According to a retrospective and a cross-sectional study of patients treated with C5 inhibitors, at least 72% had persistently low hemoglobin and at least 36% required one or more transfusions a year7.
PNH patients’ experience complement dysregulation which leads to chronic hemolysis and thrombosis characteristic of PNH8. Patients will manifest symptoms of hemolysis, which are a direct cause of the increased sensitivity of PNH cells to complement9.In PNH, blood cells lack complement regulatory proteins, so the body recognizes these healthy red blood cells as damaged2. This leads to uncontrolled activation of the complement cascade, initiated at C3, and results in the destruction of oxygen-carrying red blood cells10.
Available Treatments
Until recently, C5 inhibition with eculizumab or ravulizumab represented the only therapies approved by Health Canada for patients with PNH. Although C5-inhibitors reduce PNH-related signs and symptoms, many patients continue to exhibit persistent anemia (manifested as fatigue/extreme fatigue) and require frequent blood transfusions. Approximately a third of patients with paroxysmal nocturnal hemoglobinuria (PNH) remain transfusion dependent or have symptomatic anemia despite treatment with a C5 inhibitor11.
Pegcetacoplan may soon become the third Health Canada-approved treatment for adults with PNH, and the first treatment to target C3, a complement component upstream of C5. Inhibiting proximal complement activity with pegcetacoplan, described as the novel strategy in PNH, controls C5-mediated intravascular hemolysis and prevents C3-mediated extravascular hemolysis.
Pegcetacoplan is a C3 inhibitor targeted to control both IVH and EVH. The PADDOCK and PALOMINO studies assessed the safety and preliminary efficacy of pegcetacoplan in complement inhibitor-naïve patients. The PADDOCK [phase Ib], PALOMINO [phase IIa], PRINCE (phase III) found that pegcetacoplan contributed to superior improvements in primary and secondary endpoints related to hemoglobin levels, and other hematologic parameters and provided effective management of anemia and anemia-related complications such as fatigue & transfusion12. The PEGASUS study confirmed the long-term safety and durable efficacy of pegcetacoplan as demonstrated by sustained improvements in clinical and hematologic outcomes in pegcetacoplan treated patients.
Improved Outcomes
“Having access and being on this medication has given me the miracle of living a full life whereby I can work, pursue my passions, and give back to the community. I have been given back control over my life”. – Canadian Patient PNH interviewed for this submission
“After only 3 weeks starting on this new therapy, my hemoglobin went from 7.9 (and by the way when I was on Soliris all these years it was never above 9) to 12.2. I don’t remember feeling like this in a very long time- just getting up in the morning and not feeling this burden of fatigue on me. I feel great- I feel alive. My other CBC levels are all up, and don’t need to go to the clinic for continuous blood-transfusions. It is easy to inject myself. I have set reminders in my calendar and that’s it.” – Canadian Patient PNH interviewed for this submission
“In July 2019 I started this new therapy through the clinical trial. Right-a-way I noticed the changes: no more blood-transfusion and a better quality of life. Before, even on Soliris, I could not go back to dancing. I had been a dancer my whole life and with PNH I just did not have the energy to dance, even when on Soliris I could not. But because of this new drug I now dance. While it may seem benign to some, a flight of stairs for me was a big-feat to go up the stairs. Now this is not an issue. On Soliris I was not living at a normal level. On this new drug, I noticed a huge improvement on just being able to catch a breath. Before 2019 when I was on Soliris, it was hard on my family, because of constant blood transfusions appointments so my parents had to take a day off and drive me, it was emotionally stressful. Sometimes my mother had to take weeks off work because I was not well. Today, I can say my family notices improvements such as my better mood and the fact that we don’t worry about blood counts every week. If you don’t live with PNH, you just don’t know the feeling of worrying about your hemoglobin levels all the time”. – Canadian Patient PNH interviewed for this submission
As this miracle therapy, as described by patients, is being reviewed by CADTH, we wish to convey the importance of leaving the treatment decision between a physician and their patient, which is typically based on many factors (private/public insurance, blood-transfusion dependence etc.). There are currently two other biologics that are approved for use in Canada; however, both are C5. Through all cited scientific reviews elevated throughout this submission, it is clear EVH patients will still be blood-transfusion dependant and therefore C5 should not be the first-line therapy or gold standard of therapy for every PNH patients due to this very reason. EVH patients need alternative options because of the inability of C5 treatment to thoroughly control intravascular hemolysis and prevent extravascular hemolysis. Clinical studies have demonstrated patients with PNH has driven the development of pharmacotherapies that target alternative molecules in the complement cascade, such as C3 with pegcetacoplan13.
As pegcetacoplan targets the complement cascade upstream of C5 (targeted by ravulizumab and eculizumab), it provides more complete hemolysis protection by reducing terminal complement-mediated intravascular hemolysis and preventing C3b-associated extravascular hemolysis13. Comprehensive control of complement-mediated hemolysis with C3 will enable substantial improvements in key PNH outcomes including improvements in hematologic parameters, transfusion requirements, fatigue, and quality of life.
Well documented findings have shown that despite treatment with eculizumab and ravulizumab for a period of up to 5 years, some patients remained severely anemic, were transfusion-dependent, and reported substantial fatigue14. These clinical manifestations with C5 cannot be shadowed, as they have massive impacts on patients’ quality of life and their entire family ecosystem.
Already cited, improved quality of life and fatigue were measured among pegcetacoplan users determined by the FACIT-Fatigue questionnaires15. The results of the PEGASUS study suggests that the use of inhibitors against proximal mediators within the complement pathway may contribute to improved outcomes in patients with PNH by preventing both EVH and IVH.
According to the patients we interviewed, they individually described the true value of generalized fatigue and its impact on their quality of life. Fatigue is one of the most common complaints related by patients. It usually refers to nonspecific sense of a low energy level, or the feeling that near exhaustion is reached after relatively little exertion. This symptom led most of patients, before accessing pegcetacoplan, the challenge of getting out bed in the morning, not being able to do small activities during the day (i.e., grocery shopping), not being able to work etc. While not cited in the trials, there is a great impact of being blood-transfusion independent. Frequent blood-transfusion can cause hemochromatosis (iron overload). It has been well documented that patients can get too much iron in their blood if having multiple blood transfusions, which is the case for EVH patients. It can damage heart and liver. Transfusion iron overload is directly associated with the number of blood transfusions. One unit of transfused blood contains about 200-250 mg of iron16. In general, patients who receive more than 10 to 20 units of blood are at a significant risk of iron overload16. The prognosis of patients with iron overload depends significantly on early detection and adherence to preventive measures. For example, it takes about 1.5 months to reduce 50% of liver iron concentration, whereas cardiac iron concentration takes about 13 months16. Enhancement of quality of life and survival in transfusion iron overload patients has been steadily improved since the introduction of preventive therapies; however, mortality in transfusion iron overload is three times that of the general population16. Cirrhosis of the liver, hepatic failure, cardiomyopathy, conduction defects, heart failure, diabetes mellitus, hypogonadism, hypothyroidism, and arthropathy are long term complications of transfusion iron overload16. The most common cause of early death is dilated cardiomyopathy16. Other complications with frequent blood transfusions are allergic reactions, fever & acute immune hemolytic reaction. The latter is caused by a patient’s body attacking the transfused red blood cells. The attack triggers a release of a substance that damages the kidneys. This is often the case when the donor blood is not a proper match with the patient’s blood type16. Symptoms include nausea, fever, chills, chest and lower back pain, and dark urine.
Experience With Drug Under Review
According to the clinical study results of PEGASUS, there are important correlations between fatigue measured by FACIT-F and hemoglobin levels in the PNH patients participating in the trial. We, as the only patient groups who represent the Canadian PNH community, firmly believe that based on these results it will be very crucial for the specialized & treating physicians to consider innovative treatments based on which ones can increase hemoglobin ranges (i.e., even among >10 g/dL). Based on these results, PNH patients may experience improvements in fatigue and other symptoms from pegcetacoplan at various hemoglobin levels. This is a result of the trial demonstrating treatment to lead to a reduction of transfusion requirements, and higher hemoglobin levels.
We inquired about the value of subcutaneous injection vs intravenous infusion; the majority of patients revealed it is easy to manage at their comfort of their home the treatment. They revealed this form of treatment is easier compared to making appointments for the infusion through the patient-support-program and waiting for a nurse to confirm the time as it is another burden on life management- they can decide the two dates they will do injections without making multiple phone calls. Some patients elevated the importance for proper training for the injections, but overall, the feeling was overwhelmingly positive in terms of alternating injection sites and storing the medication.
“I have been a PNH patient since 2001. First no treatment for years then Soliris was approved and as a result there was hope! But after several years of acceptable lifestyle anemia set back in and I was in need of blood transfusions every 8 weeks +/- to maintain an acceptable hemoglobin. But along with the transfusions came increased iron levels. Iron reducing meds lowered the iron levels somewhat, but they played havoc with my neutrophils to the point that they were completely wiped out! Needless to say, panic set in with having to live with a very compromised immune system. Luckily there was a new drug in development, and I was fortunate to be accepted into the clinical trials here in Canada. Results were almost immediate. Blood labs returned to normal levels. Anemia was gone and so was the need for transfusions! After 3 years of being on pegcetacoplan life is pretty much back to normal in that I am able to cycle 30+ km at a time, hike up a ski hill and more importantly chase after my two delightful grandkids. Modern medicine for me has made life beautiful again.” – Canadian Patient PNH interviewed for this submission
“Having pegcetacoplan has been a huge improvement in my life and I believe it should be available to all PNH patients who could benefit from it. It changes PNH to a manageable disorder. Such a huge difference from managing prednisone to a dramatic improvement in my life. Soliris was an absolute breather when no other treatment was available and I thought Soliris had set the bar high, and pegcetacoplan is definitely better for me” – Canadian Patient PNH interviewed for this submission.
Companion Diagnostic Test
Not applicable.
Anything Else?
“What we went through in terms of advocacy to add the first biologic to public listing, no Canadians should have to go through what we have been through. We wish that this drug is available right away to all who need it. Please approve.” – Canadian Patient PNH interviewed for this submission
Pegcetacoplan therapy for patients with PNH has been shown to have several benefits compared to treatment with Health Canada approved therapies. In contrast to the requirement for intravenous dosing seen in eculizumab and ravulizumab, pegcetacoplan can be self-administered at home by the patient and or caregiver at their convenience and as prescribed (twice weekly). This convenience eliminates the continuous & strained need for IV drug administration and regular infusion clinic visits15. In addition to improved hematologic parameters, patients receiving pegcetacoplan have reported increased quality of life measures (such as decreased fatigue levels) in various clinical trials of the drug15. While it is not specific Canadian data, we are raising a recent cost analysis demonstrated $1.7 million in cost savings for one US health insurance payer over a 3-year period due to reduced transfusion requirements and fewer breakthrough hemolysis events when patients were treated with pegcetacoplan17.
The subcutaneous dosing alternative presents a number of advantages over intravenous dosing, including hospital and clinical cost savings, reduced time and resource use, increased flexibility in appointment scheduling, and reduced capacity bottlenecks and nursing overtime. These are all benefits towards subcutaneous that should be accounted for when looking at health care cost savings.
People with rare diseases frequently wait years for a proper diagnosis, have to travel long distances for specialty care, face high out-of-pocket health care expenses as well as delays of accessing the right therapy18. This combination of challenges in accessing appropriate medical care leads to poor health-related quality of life, low patient satisfaction and high levels of anxiety, depression and stigma18. A large-scale 2013 survey, which remains relevant today, reported that 90% of US participants and 91% of UK participants were anxious about the prognosis for their disease, which was exacerbated by the dearth of information available on their condition (83% in the US, 81% in the UK)18. This had a domino effect on other areas of their lives, resulting in social isolation and further deterioration in mental well-being. We are all aware mental health has been exacerbated because COVID-19 has caused disparities in diagnosis and treatment, and if there potentially a new drug that is accessible for PNH patients with EVH pathways, the question is: why should we not do everything that we can (i.e. Health Canada approval, CADTH approval without any restrictions/limitations in eligibility criteria, fair pCPA negotiations) so our patients can avoid massive health consequences just as mental health, allergic reaction, iron overload? This should be our social responsibility to help these patients to get access to the only therapy that could treat EVH pathway. This access would not only help the healthcare to reduce the burden, but it brings them back to the society and brings the family back together.
All PNH patients who could potentially contribute to the society, either by going to school to study for a career or by going back to the workforce, they need to be supported by having choice of treatment, so they have high enough hemoglobin levels and are no longer blood transfusion dependant. The latter is a burden of patients’ lives, especially it was in the context of the pandemic, because patients had to go to the clinic 1-4 times a month, paying for a parking ticket and being in an environment where the virus is highly transmissible. This scenario has significant impact on the healthcare system and economy. Patients from all walks of life should have the right and option to feel like any other individuals and not feel like a patient, which is reminded when they have to get to the clinic for an infusion or blood transfusion appointment.
We believe the drug manufacturer & all relevant stakeholders should negotiate in good faith to ensure pegcetacoplan is timely accessible for all eligible PNH patients. Time is of the essence. PNH does not wait, and it should not be acceptable by any of the reviewers that Canadian patients remain on blood transfusion while there is a therapy approved by Health Canada that treats the EVH pathway and keeps patients’ blood transfusion independent.
References
1. Shah N, Bhatt H. Paroxysmal nocturnal hemoglobinuria. Treasure Island, FL: StatPearls Publishing, 2021. [PubMed] [Google Scholar]
2. Brodsky RA. Paroxysmal nocturnal hemoglobinuria. Blood. 2014;124(18):2804–2811. doi: 10.1182/blood-2014-02-522128.
3. Hill A, DeZern AE, Kinoshita T, Brodsky RA (2017) Paroxysmal nocturnal haemoglobinuria. Nat Rev Dis Primers 3:17028
4. Hill A, Kelly RJ, Hillmen P (2013) Thrombosis in paroxysmal nocturnal hemoglobinuria. Blood 121(25):4985–4996 quiz 5105
5. Hillmen P, Lewis SM, Bessler M, et al. Natural history of paroxysmal nocturnal hemoglobinuria. N
6. Wong RSM. Safety and efficacy of pegcetacoplan in paroxysmal nocturnal hemoglobinuria. Ther Adv Hematol. 2022 Jul 28;13:20406207221114673. doi: 10.1177/20406207221114673. PMID: 35923770; PMCID: PMC9340389.
7. Paroxysmal nocturnal hemoglobinuria (PNH). The Sidney Kimmel Comprehensive Cancer Center. Website: https://www.hopkinsmedicine.org/kimmel_cancer_center/types_cancer/paroxysmal_nocturnal_hemoglobinuria_PNH.html. Accessed August 29, 2022.
8. Kulasekararaj AG, Hill A, Rottinghaus ST, et al. Ravulizumab (ALXN1210) vs eculizumab in C5-inhibitor-experienced adult patients with PNH: the 302 study. Blood 2019; 133: 540–549.
9. Hillmen P, Szer J, Weitz I, et al. Pegcetacoplan versus eculizumab in paroxysmal nocturnal hemoglobinuria. N Engl J Med 2021; 384: 1028–1037.
10. Raymond S. M. Wong, Humphrey W. H. Pullon, Ismail Amine, Andrija Bogdanovic, Pascal Deschatelets, Cedric G. Francois, Kalina Ignatova, Surapol Issaragrisil, Pimjai Niparuck, Tontanai Numbenjapon, Eloy Roman, Jameela Sathar, Raymond Xu, Mohammed Al-Adhami, Lisa Tan, Eric Tse, Federico V. Grossi. (2022) Inhibition of C3 with pegcetacoplan results in normalization of hemolysis markers in paroxysmal nocturnal hemoglobinuria. Annals of Hematology 101:9, 1971-1986.
11. Risitano AM, Marotta S, Ricci P, et al. Anti-complement treatment for paroxysmal nocturnal hemoglobinuria: time for proximal complement inhibition? A position paper from the SAAWP of the EBMT. Front Immunol 2019; 10: 1157
12. Dixon BP, Greenbaum LA, Huang L, et al. C3 inhibition with pegcetacoplan targets the underlying disease process of C3 glomerulopathy. J Am Soc Nephrol 2020; 31: 577.
13. Apellis Pharmaceuticals. EMPAVELI™ (pegcetacoplan) injection, for subcutaneous use. U.S. Prescribing Information, 2021, https://pi.apellis.com/files/PI_Empaveli.pdf
14. Dingli D, Matos JE, Lehrhaupt K, Krishnan S, Yeh M, Fishman J, Sarda SP, Baver SB (2022) The burden of illness in patients with paroxysmal nocturnal hemoglobinuria receiving treatment with the C5-inhibitors eculizumab or ravulizumab: results from a US patient survey. Ann Hematol 101(2):251–263
15. Wong RSM. Safety and efficacy of pegcetacoplan in paroxysmal nocturnal hemoglobinuria. Ther Adv Hematol. 2022 Jul 28;13:20406207221114673. doi: 10.1177/20406207221114673. PMID: 35923770; PMCID: PMC9340389.
16. Remacha A, Sanz C, Contreras E, De Heredia CD, Grifols JR, Lozano M, Nuñez GM, Salinas R, Corral M, Villegas A., Spanish Society of Blood Transfusion. Spanish Society of Haematology and Haemotherapy. Guidelines on haemovigilance of post-transfusional iron overload. Blood Transfus. 2013 Jan;11(1):128-39.
17. Talbird SE, Anderson S, Fishman J, et al. Budget impact of pegcetacoplan, a complement C3 inhibitor, for the treatment of paroxysmal nocturnal hemoglobinuria in us adults. ISPOR EU 2021, https://www.valueinhealthjournal.com/article/S1098-3015(21)02213-0/fulltext
18. Shire. Rare Disease Impact Report: Insights from patients and the medical community. 2013. https://globalgenes.org/wp-content/uploads/2013/04/ ShireReport-1.pdf. Accessed 10 Nov 2016.
Conflict of Interest Declaration — Canadian Association of PNH Patients & Aplastic Anemia & Myelodysplasia Association of Canada
To maintain the objectivity and credibility of the CADTH reimbursement review process, all participants in the drug review processes must disclose any real, potential, or perceived conflicts of interest. This Patient Group Conflict of Interest Declaration is required for participation. Declarations made do not negate or preclude the use of the patient group input. CADTH may contact your group with further questions, as needed.
Did you receive help from outside your patient group to complete this submission? If yes, please detail the help and who provided it.
Yes, industry consultant (Hamzo Pharma & Biotech Consultant).
Did you receive help from outside your patient group to collect or analyze data used in this submission? If yes, please detail the help and who provided it.
Yes, industry consultant (Hamzo Pharma & Biotech Consultant).
List any companies or organizations that have provided your group with financial payment over the past 2 years AND who may have direct or indirect interest in the drug under review.
Table 1: Financial Disclosures for the Canadian Association of PNH Patients
Company | $0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 |
|---|---|---|---|---|
Alexion Pharma Canada | — | — | X | — |
Novartis | — | — | X | — |
Roche | — | X | — | — |
Sobi | — | — | X | — |
Table 2: Financial Disclosures for the Aplastic Anemia & Myelodysplasia Association of Canada
Company | $0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 |
|---|---|---|---|---|
BMS | — | — | X | — |
Alexion Pharma Canada | — | — | X | — |
Sobi | — | — | X | — |
TAIHO | — | X | — | — |
Takeda | — | X | — | — |
Novartis | — | X | — | — |
Clinician Input
Canadian PNH Network
About the Canadian PNH Network
The Canadian PNH Network is a group of Canadian hematologists with a special interest and expertise in the care of patients with paroxysmal nocturnal hemoglobinuria (PNH). Members represent centres of excellence from Newfoundland, Nova Scotia, Quebec, Ontario, Alberta, and British Columbia. The Canadian PNH Network sites follow the vast majority of PNH patients in Canada, either directly or as part of shared-care relationships with community physicians. We also set consensus for diagnosis and management of PNH in the country (Patriquin CJ et al. [2019] Eur J Haematol) and serve as sites for ongoing observational and interventional research activities both nationally and internationally.
Information Gathering
Information for this submission was obtained via publicly available documents, congress abstracts, and the published literature (primarily the PEGASUS trial – Hillmen P et al. [2021] NEJM). Standard of care data were similarly obtained, and the members of the Canadian PNH Network were invited to contribute to the various segments.
Current Treatments and Treatment Goals
The current standard of care (SOC) for patients with hemolytic PNH is terminal complement inhibition with C5 blockade. Eculizumab, approved in Canada in 2009, remains the only option available across the country. There is a second-generation C5 inhibitor, ravulizumab, which is approved by Health Canada and has recently received favourable recommendation by CADTH; currently, only patients who were part of the initial clinical trials and patients whose private insurance companies cover ravulizumab have the opportunity to switch from eculizumab. To be approved for eculizumab in Canada, patients must have evidence of a PNH clone ≥ 10%, lactate dehydrogenase (LDH) > 1.5 x the upper limit of normal (ULN), and at least one significant clinical manifestation such as thrombosis, anemia, transfusion-dependence, renal or respiratory failure without other explanation, and smooth muscle dystonic symptoms requiring either hospitalization or opioid analgesia. Though there are some slight differences, the full details of approval criteria can be found here: (https://health.gov.on.ca/en/pro/programs/drugs/docs/frequently_requested_drugs.pdf, page 50).
The only curative treatment for PNH is allogeneic hematopoietic stem cell transplant. It should be noted, however, that this is reserved for patients with predominant or progressive bone marrow failure (which can coincide with, precede, or follow a diagnosis of PNH) or in those eligible patients with evidence of clonal evolution (e.g., AML, high-risk MDS). Transplant is not recommended for all patients given the increased risk of complications and transplant-related mortality compared to C5 inhibition. Though complement inhibition does not address the underlying marrow mutations which cause PNH, blockade of terminal complement activity and associated control of intravascular hemolysis (IVH) leads to significant improvement in quality of life, fatigue, transfusion-dependence, thrombosis, and overall survival. Supportive therapies for PNH patients, if needed, include hematinic support (folate, iron), analgesia, and anticoagulation either to treat or protect against thrombosis. It should be noted, however, that anticoagulation alone does not protect against thrombosis in PNH, which is the leading cause of death in untreated patients (40-67%).
Treatment with C5 inhibition, such as with eculizumab, is highly effective at controlling intravascular hemolysis. This is measured by targeting an LDH <1.5 x ULN. Associated with this, we would watch for improvement in hemoglobin, reduced transfusion needs, and absence of other end-organ complications like thrombosis, renal failure, and pulmonary hypertension. With C5 inhibition, PNH red cells are now able to survive and circulate where previously they would have been exquisitely sensitive to terminal complement-mediated IVH. Now that red cells survive, they can have more and more C3 split products bind to their membrane. As cell-bound complement inhibitors are missing, the dense C3 deposits drive extravascular hemolysis, mostly via receptors in the liver. Because of this, about a third of PNH patients remain symptomatically anemic and possibly still transfusion-dependent (Debureaux P et al. [2021] Bone Marrow Transplant – https://pubmed.ncbi.nlm.nih.gov/34226670/), with increasing rates of extravascular hemolysis coinciding with reduced levels of hematologic response. Due to the underlying disease phenotype, any C5 inhibitor can drive the extravascular hemolysis. In contrast, blocking complement at a proximal level, such as C3 by pegcetacoplan, this extravascular hemolysis is also blocked, allowing for increased hemoglobin.
General therapeutic approaches with a Canadian focus can be found in our consensus guidelines (Patriquin CJ et al. [2019] Eur J Haematol – https://onlinelibrary.wiley.com/doi/10.1111/ejh.13176).
Treatment Gaps (Unmet Needs)
Considering the treatment goals, please describe goals (needs) that are not being met by currently available treatments.
The major treatment gaps that remain for PNH management in Canada are threefold. First, even with SOC, the highly restrictive approval criteria via Exceptional Access Programs means that some patients with highly active, hemolytic PNH do not get access. Second, though eculizumab has changed the face of PNH, treatment burden remains high with fortnightly treatment necessitating venous access and either visits to infusion centres or visits from nurses to patients’ homes. Lastly, though terminal complement blockade with C5 inhibitors has led to significant improvements in care and survival, a large minority of patients remain symptomatically anemic without options for improving outcomes with current therapies. These unmet needs are expanded upon in the following paragraphs.
The EAP criteria for the provinces are highly restrictive. There are patients across the country with large-clone PNH with significant hemolysis who do not have approval because they are not anemic enough (usually because they are younger and have active, robust bone marrow compensatory activity) or perhaps have significant smooth muscle dystonia but have not accepted hospital admission or narcotics to treat. This creates a subset of our patients at exceptionally high risk of complications who can only be managed with supportive care, hoping that they do not in the interim develop devastating complications such as thrombosis. This should be compared to most other jurisdictions with approved complement inhibitors, in which high disease activity (HDA) criteria allow PNH patients to be treated as long as they have evidence of hemolysis (LDH > 1.5x ULN) and more appropriate clinical criteria, such as otherwise unexplained fatigue and abdominal pain (not mandating first that they are hospitalized or accept narcotics). These HDA criteria, outlined in a recent registry analysis (https://link.springer.com/article/10.1007/s00277-020-04052-z), also much more closely resemble the inclusion criteria for recent clinical trials in PNH naïve to targeted therapy (e.g., https://clinicaltrials.gov/ct2/show/NCT02946463).
Treatment burden is an important focus for PNH patients receiving eculizumab as SOC, particularly now that some of the most important needs are met, including improved survival. Novel therapeutics now can focus on maintaining similar control of the disease whilst reducing frequency of treatment (e.g., ravulizumab), providing self-administration options (e.g. pegcetacoplan), and even further exploring alternate formulations such as oral inhibitors (refer to Patriquin CJ & Kuo KHM [2019] Transfusion Medicine Reviews for details – https://pubmed.ncbi.nlm.nih.gov/31703946/).
PNH patients treated with eculizumab or ravulizumab will have increased survival of their red blood cells in circulation, which subsequently sets up the opportunity for extravascular hemolysis to occur. Though this likely happens in the vast majority of C5 inhibitor-treated patients, the clinical implications vary; approximately a third of patients will remain anemic (and possibly transfusion-dependent), which has significant impact on reduced quality of life and energy. Proximal complement blockade, such as with C3 as a target, addresses the extravascular hemolytic risk and can allow for profound increases in hemoglobin concentration for patients.
Place in Therapy
How would the drug under review fit into the current treatment paradigm?
Pegcetacoplan is the first proximal complement/C3 inhibitor to be clinically tested in PNH patients and is approved in some jurisdictions already. Unlike eculizumab and ravulizumab, it is the only drug currently that addresses and protects against extravascular hemolysis. With the data available to date, and as was the design of the PEGASUS trial (Hillmen P et al. [2021] NEJM), pegcetacoplan most appropriately would fit into the current treatment landscape as an alternative (i.e., switch) option for eculizumab (or ravulizumab)-treated patients with ongoing anemia or who otherwise just do not respond to eculizumab. It could also be used for those patients with intolerance to SOC, but that is quite uncommon. The overall treatment paradigm is not anticipated to change. Until we have more mature data on upfront use of pegcetacoplan in treatment-naïve patients (as was reported in the PRINCE trial – https://clinicaltrials.gov/ct2/show/NCT04085601), first-line treatment would be either eculizumab or ravulizumab; however, availability of pegcetacoplan would very effectively extend our current treatment paradigm. Whereas right now we have no effective options to treat extravascular hemolysis (prednisone and splenectomy have been tried and are ineffective and poorly tolerated), pegcetacoplan would be the first choice to give patients with persistent anemia.
Which patients would be best suited for treatment with the drug under review? Which patients would be least suitable for treatment with the drug under review?
As described above, and as seen in PEGASUS, the patients most likely to benefit from pegcetacoplan are those who have persistent anemia (Hb <105 g/L or perhaps higher if symptomatic) despite stable doses of eculizumab or ravulizumab. These patients with extravascular hemolysis currently have no effective options to improve their hemoglobin concentration save for transfusions and hematinic support if indicated. It is of course important that clinicians rule out other causes of ongoing anemia as well (e.g., breakthrough hemolysis or bone marrow failure), but this is easily done with standard testing. Pharmacokinetic breakthrough can be identified in patients with cyclical symptoms leading up to their next C5 inhibitor infusions who may also show increased LDH and CH50 values. Patients with bone marrow failure would likely show evidence of decreasing reticulocyte and platelet counts, and this could be confirmed with bone marrow biopsy/aspiration. Extravascular hemolysis is typically identified when patients have C3 loading detected on the surface of their red blood cells, which can be done by sensitive direct antiglobulin test (DAT) assays available at some but not all centres. It should be noted however that patients who otherwise met entry criteria for the PEGASUS trial improved, even without clear C3 loading, suggesting currently available clinical assays are not sensitive enough. As such, we would certainly expect patients with C3d+ DAT assays to benefit, but this would miss a group who would likely benefit just as much, so we would rely again on comparing our patients to the inclusion criteria for PEGASUS to identify potential responders.
What outcomes are used to determine whether a patient is responding to treatment in clinical practice? How often should treatment response be assessed?
Response to complement blockade in PNH patients first and foremost focuses on reduction in LDH, which is a consistent surrogate used to identify intravascular hemolysis activity. The goal is to have patients consistently fall below an LDH ratio of 1.5x the ULN. This not only reduces hemolysis and may improve hemoglobin and transfusion-dependence, but it also reduces the risk of thrombosis in PNH. Clinical outcomes related to this, as seen in the landmark eculizumab and ravulizumab trials, are decreased fatigue, transfusion requirements, improved QoL and, given the maturity of eculizumab data available, also improved overall survival. An important outcome of clinical (and clinical trial) interest is an increase in hemoglobin, particularly now that there are proximal inhibitors such as pegcetacoplan that can effectively achieve this.
A clinically meaningful response to treatment would be sustained control of LDH but with further hemoglobin increases (or possibly hemoglobin stabilization without further needs of transfusion for those who are transfusion-dependent) and improvement in anemia-related symptoms, such as but not limited to fatigue and dyspnea. The increase in hemoglobin in C3 inhibitor-treated patients is quite objective and not expected to vary across physician treaters. In fact, rather similar increments around 20-30 g/L have been seen not just in PEGASUS but in preliminary phase trials of other proximal inhibitors targeting complement factors B and D.
What factors should be considered when deciding to discontinue treatment with the drug under review?
Pegcetacoplan discontinuation should be considered in patients who have adverse events that preclude ongoing therapy. This may include issues with effective self-administration or intolerable pain from injections (this is not common based on the trial data). Due to the PEG formulation of the drug, diarrhea may occur, but is typically mild and manageable. The most important feature to watch for is evidence of breakthrough hemolysis. It is possible that some patients who take pegcetacoplan will have significant expansion of their circulating red blood cells and, in situations of severe complement-mediated stress, could have increased hemolytic events. As is seen in the 48-week data, hemolysis was reported as a reason to discontinue drug for a minority of patients. The supplementary PEGASUS data also suggest that pegcetacoplan-treated patients may have higher LDH values with breakthrough hemolytic events. How best to manage this is still a scientific question, with a substudy underway to test additional doses of pegcetacoplan. An alternative strategy is to provide a “rescue” dose of eculizumab to directly block terminal complement during the acute event. However, the international PNH community need to further establish standard management strategies for this potential. If this were to become a recurrent event for a patient on pegcetacoplan, it would be important to discuss returning to C5 inhibition, understanding that they would likely be giving up the higher hemoglobin concentration for possible reduced breakthrough risk.
What settings are appropriate for treatment with [drug under review]? Is a specialist required to diagnose, treat, and monitor patients who might receive [drug under review]?
PNH is an ultrarare disease with nuances to diagnosis, treatment, and overall management. Patients likely benefit being followed by clinicians who specialize in the area. Members of the Canadian PNH Network would certainly be included in this categorization. Monitoring of patients can be done with standard laboratory investigations and clinical visits. However, specifically regarding treatment with pegcetacoplan, this is done at the patient home (or wherever they prefer really), as it is self-administered. Patients can even travel with their drug, no longer tied to fortnightly infusion schedules with eculizumab.
Additional Information
Not applicable.
Conflict of Interest Declarations — Canadian PNH Network
To maintain the objectivity and credibility of the CADTH drug review programs, all participants in the drug review processes must disclose any real, potential, or perceived conflicts of interest. This conflict of interest declaration is required for participation. Declarations made do not negate or preclude the use of the clinician group input. CADTH may contact your group with further questions, as needed. Please refer to the Procedures for CADTH Drug Reimbursement Reviews (section 6.3) for further details.
Did you receive help from outside your clinician group to complete this submission?
No. This was completed independently by myself, as representing the Canadian PNH Network members, and approved by the co-signatories.
Did you receive help from outside your clinician group to collect or analyze any information used in this submission.
No. All information used to support this submission was publicly available.
List any companies or organizations that have provided your group with financial payment over the past two years AND who may have direct or indirect interest in the drug under review. Please note that this is required for each clinician who contributed to the input.
In the past, Canadian PNH Network meetings have been supported by Alexion (makers of eculizumab and ravulizumab, as well as proximal factor D inhibitors under trial). Some members of the Canadian PNH Network, including myself, have participated in advisory boards with the company but this was done independently, not as the Network as a whole. Similarly, some members of the Network have participated in advisory boards with Sobi, who are bringing this pegcetacoplan filing forward.
Personal disclosures of the Canadian PNH Network member so-signatories are listed below. This document was written on behalf of the Canadian PNH Network.
Declaration for Clinician 1
Name: Dr. Christopher Patriquin
Position: Consultant, Hematology & Apheresis (University Health Network) / Chair, Canadian PNH Network
Date: 15-08-2022
Table 3: COI Declaration for Canadian PNH Network — Clinician 1
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In Excess of $50,000 |
|---|---|---|---|---|
Alexion | — | — | X | — |
Sobi | — | X | — | — |
BioCryst | — | X | — | — |
Novartis | X | — | — | — |
Amgen | — | X | — | — |
Declaration for Clinician 2
Name: Dr. Monika Oliver
Position: Consultant, Hematology & Apheresis (University of Alberta Hospital) / Member, Canadian PNH Network
Date: 16-08-2022
Table 4: COI Declaration for Canadian PNH Network — Clinician 2
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In Excess of $50,000 |
|---|---|---|---|---|
Alexion | X | — | — | — |
Sobi | X | — | — | — |
Declaration for Clinician 3
Name: Dr. Thomas L Kiss
Position: Staff Physician, Hematology, Maisonneuve Rosemont Hospital, Montreal, Quebec/Member, Canadian PNH Network
Date: 17-08-2022
Table 5: COI Declaration for Canadian PNH Network — Clinician 3
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In Excess of $50,000 |
|---|---|---|---|---|
Alexion | — | X | — | — |
Sobi | — | X | — | — |
Novartis | X | — | — | — |
Declaration for Clinician 4
Name: Dr. Ian Chin-Yee
Position: Head, Laboratory Medicine, London Health Sciences, Schulich School of Medicine, Western University / Member, Canadian PNH Network
Date: 18-08-2022
Table 6: COI Declaration for Canadian PNH Network — Clinician 4
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In Excess of $50,000 |
|---|---|---|---|---|
No COI | — | — | — | — |
Declaration for Clinician 5
Name: Dr. Kuljit Grewal
Position: Hematologist, Associate Professor of Medicine, Memorial University of Newfoundland / Member, Canadian PNH Network
Date: 18-08-2022
Table 7: COI Declaration for Canadian PNH Network — Clinician 5
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In Excess of $50,000 |
|---|---|---|---|---|
Alexion | — | X | — | — |
Sobi | — | X | — | — |
Declaration for Clinician 6
Name: Dr. Loree Larratt
Position: Hematologist, Professor Emeritus, University of Alberta / Member, Canadian PNH Network
Date: 18-08-2022
Table 8: COI Declaration for Canadian PNH Network — Clinician 6
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In Excess of $50,000 |
|---|---|---|---|---|
Alexion | — | X | — | — |
Sobi | X | — | — | — |
Declaration for Clinician 7
Name: Dr. Jennifer Grossman
Position: Hematologist, Foothills Medical Centre, Alberta Health Services / Member, Canadian PNH Network
Date: 18-08-2022
Table 9: COI Declaration for Canadian PNH Network — Clinician 7
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In Excess of $50,000 |
|---|---|---|---|---|
No COI | — | — | — | — |
Declaration for Clinician 8
Name: Dr. Brian Leber
Position: Hematologist, Professor of Medicine, McMaster University / Member, Canadian PNH Network
Date: 18-08-2022
Table 10: COI Declaration for Canadian PNH Network — Clinician 8
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In Excess of $50,000 |
|---|---|---|---|---|
Alexion | — | X | — | — |
Declaration for Clinician 9
Name: Dr. Catherine Sperlich
Position: Hematologist,CISSS-Montérégie-Centre /Hôpital Charles-Lemoyne, associate clinical professor, Université de Sherbrooke/Member, Canadian PNH Network
Date: 22-08-2022
Table 11: COI Declaration for Canadian PNH Network — Clinician 9
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In Excess of $50,000 |
|---|---|---|---|---|
Alexion | X | — | — | — |
Sobi | X | — | — | — |
Declaration for Clinician 10
Name: Dr. Danièle Marceau
Position: Centre de Santé et de Services Sociaux Alphonse-Desjardins (Centre Hospitalier affilié Universitaire de Lévis), Lévis, QC, Canada/ Member, Canadian PNH Network
Date: 23-08-2022
Table 12: COI Declaration for Canadian PNH Network — Clinician 10
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In Excess of $50,000 |
|---|---|---|---|---|
No COI | — | — | — | — |
Declaration for Clinician 11
Name: Dr. Sue Robinson
Position: Hematologist, Professor of Medicine, Division of Hematology, Department of Medicine, Dalhousie University / Member, Canadian PNH Network
Date: 23-08-2022
Table 13: COI Declaration for Canadian PNH Network — Clinician 11
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In Excess of $50,000 |
|---|---|---|---|---|
Alexion | X | — | — | — |
Sobi | X | — | — | — |
Takeda | X | — | — | — |
Sanofi | X | — | — | — |
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up-to-date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
Redactions: Confidential information in this document may be redacted at the request of the sponsor in accordance with the CADTH Drug Reimbursement Review Confidentiality Guidelines
Stakeholder Input: The views expressed in each submission are those of the submitting organization or individual; not necessarily the views of CADTH or of other organizations. As such, they are independent of CADTH and do not necessarily represent or reflect the view of CADTH. No endorsement by CADTH is intended or should be inferred. By filing with CADTH, the submitting organization or individual agrees to the full disclosure of the information. CADTH does not edit the content of the submissions.
CADTH does use reasonable care to prevent disclosure of personal information in posted material; however, it is ultimately the submitter’s responsibility to ensure no identifying personal information or personal health information is included in the submission. The name of the submitting organization or individual and all conflict of interest information are included in the submission; however, the name of the author, including the name of an individual patient or caregiver submitting the patient input, are not posted.
Accessibility: CADTH is committed to treating people with disabilities in a way that respects their dignity and independence, supports them in accessing material in a timely manner, and provides a robust feedback process to support continuous improvement. All materials prepared by CADTH are available in an accessible format. Where materials provided to CADTH by a submitting organization or individual are not available in an accessible format, CADTH will provide a summary document upon request. More details on CADTH’s accessibility policies can be found here.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.