CADTH Health Technology Review
Post–COVID-19 Condition Treatment and Management Rapid Scoping Review
Protocol Registration
Open Science Framework Registration
National Collaborating Centre for Methods and Tools
Scoping Review
Authorship Information
Authors
Yi-Sheng Chao, Sarah C. McGill, Michelle Gates, Thyna Vu, Angie Hamson
Contributors
Sinwan Basharat, Sarah Garland, David Kaunelis, Pierre Martinelli, Gino De Angelis
Acknowledgement
The authors would like to thank the 3 patients living with long COVID, including Beth Pardo and Amanda Porter, who contributed to the report by commenting on the findings and for generously sharing their time, perspectives, and expertise.
Abbreviations
COVID-19
coronavirus disease 2019
HTA
health technology assessment
ICU
intensive care unit
INESSS
Institut national d'excellence en santé et en services sociaux
NICE
National Institute for Health and Care Excellence
PRISMA
Preferred Reporting Items for Systematic reviews and Meta-Analyses
PRISMA-ScR
Preferred Reporting Items for Systematic reviews and Meta-Analyses extension for Scoping Reviews
SARS-CoV-2
severe acute respiratory syndrome coronavirus 2
Key Messages
This rapid living scoping review provides an up-to-date map of the latest published evidence, and identifies research gaps regarding the treatment and management for post–COVID-19 condition.
Most of the included studies were from a few countries, particularly the US, the UK, and Canada. Also, most of the studies were case reports or series, meaning they included few participants.
Most of the identified published research on treatments for post–COVID-19 condition focused on:
vaccines administered 3 months after initial infection
pharmacological interventions for cardiovascular, neurological, and pulmonary symptoms and diagnoses
non-pharmacological interventions for pulmonary symptoms.
There were fewer studies related to other types of symptoms and organ systems.
There were notable evidence gaps across all treatments for post–COVID-19 condition, as noted by most of the evidence being from case reports, in which physicians may more often report patients with uncommon symptoms or diagnoses. Notable evidence gaps included treatments for children and adolescents with post–COVID-19 condition, and those without symptoms during initial infection. We identified 1 qualitative study and no economic evaluations.
Most identified guidelines regarding the treatment and management of post–COVID-19 condition, including all Canadian guidelines, provided limited guidance specific to patients with symptoms lasting at least 3 months after initial infection.
Abstract
Background
According to WHO, post–COVID-19 condition is characterized by new or persisting symptoms occurring 12 or more weeks following an initial COVID-19 infection. People with post–COVID-19 condition have been reported to experience a range of heterogeneous symptoms, including fatigue, shortness of breath, muscle aches, and cognitive and mental health challenges. With the vast numbers of COVID-19 cases worldwide, there is the potential that a substantial number of individuals will develop long-term complications. Therefore, there is much interest in developing a clearer understanding of how to treat and manage this condition.
The objective of this rapid living scoping review was to characterize the current evidence landscape on the treatment and management of post–COVID-19 condition and to identify evidence gaps. Pharmacological interventions, non-pharmacological interventions, and evidence related to health systems for people of all ages in any context were of interest.
Methods
We undertook a rapid living scoping review of primary studies, evidence syntheses, economic evaluations, and guidelines that followed up with participants of any age for at least 12 weeks or 3 months after COVID-19 diagnosis or symptom onset, and assessed treatments and management, in any setting. We searched online databases (Ovid MEDLINE All [1946–], Ovid Embase [1974–], Cochrane Database of Systematic Reviews) and grey literature sources for studies published (commercially or as grey literature) between January 1, 2019, and October 21, 2022. A single reviewer screened studies and charted data following a pilot round. We charted details of the study design, population (country, age groups, sex or gender), treatment specifications (pharmacological, non-pharmacological, care model) and context (setting). We categorized post–COVID-19 condition by organ systems or types of indication/diagnosis and treatment and management strategies. We summarized the distribution of evidence by types of post–COVID-19 condition and treatment categories within tables and figures, and identified evidence gaps by categorizing the types of conditions that were least studied. Three adults who are currently living with post–COVID-19 condition were invited to attend a meeting to discuss the scoping review. At the meeting, CADTH staff presented the findings and facilitated a discussion about interventions or treatments, areas of controversy or uncertainty, and priorities from their perspectives. These views and comments were incorporated into the scoping review.
Results
In this modified update to a previously published CADTH scoping review, we included 113 reports: 94 primary studies (63 case reports/series), 12 evidence syntheses, and 7 guidelines. We identified no economic evaluations and only 2 qualitative studies. The majority of studies came from only a few countries, particularly the US (n = 32), the UK (n = 11), Italy (n = 9), Spain (n = 7), and Canada (n = 7). We identified 9 or more primary studies using pharmacological interventions for cardiovascular, neurological, and pulmonary conditions and non-pharmacological interventions for fatigue, neurological, and pulmonary conditions. Areas with 3 to 8 primary studies included pharmacological interventions for certain types of indication/diagnosis, such as fatigue, gastrointestinal, generalized or unspecified, otorhinolaryngological, and rheumatological conditions, and non-pharmacological interventions for cardiovascular, gastrointestinal, generalized or unspecified, mental and behavioural, and otorhinolaryngological conditions. We identified 3 rapid reviews and 2 systematic reviews assessing the effectiveness of vaccines administered 3 months after initial infection on post–COVID-19 condition, 2 rapid reviews on care models, 1 systematic review that included 6 pharmacological and 4 non-pharmacological interventions, 1 systematic review that identified non-pharmacological interventions, and 3 systematic reviews on care models or health service use for post–COVID-19 condition. We found fewer than 3 primary studies using pharmacological or non-pharmacological interventions for certain types of indication/diagnosis, such as renal and hematological conditions. Finally, there were several population groups for which relatively few studies were identified, including children younger than 18 years of age and those who were asymptomatic during initial infection.
Conclusions
The research on post–COVID-19 condition treatment and management is emerging, but to date is characterized predominantly by case reports/series. There are fewer studies of other designs, evidence syntheses, or qualitative studies; no economic analyses were identified. The majority of the identified evidence has looked at certain types of treatment or management for post–COVID-19 condition, such as vaccination. There appears to be limited evidence regarding certain types of indication/diagnosis, such as hematological conditions, and population groups, including children and those without symptoms during initial infection.
Introduction and Rationale
Post–COVID-19 condition, commonly referred to as long COVID (among many other terms being used), is a relatively new condition recognized by the Public Health Agency of Canada (PHAC) and WHO.1,2 On October 6, 2021, WHO published a case definition of post–COVID-19 condition, developed by a Delphi consensus process that included patients, clinicians, researchers, and others, representing all WHO regions.3 This definition indicates that post–COVID-19 condition is characterized by new or persistent symptoms, usually occurring 3 months from the initial infection, lasting for at least 2 months, which cannot be explained by another cause3; however, the US Centers for Disease Control and Prevention (CDC) considers that symptoms occurring 4 weeks after initial infection could be identified as post–COVID-19 condition.4,5 In 2022, Statistics Canada estimated that 32.0% of people living in Canada aged 18 years or older had reported previous lab-confirmed COVID-19 infection and 8.3% suspected previous infection.6 Of these, 1.4 million adults living in Canada were having symptoms 3 months after their initial infection.6 Considerable variations in prevalence have been observed between males and females and across different age groups.6
In May 2022, CADTH published a scoping review that mapped the available evidence and gaps up to February 2022 regarding post–COVID-19 condition across the following concepts: risk factors and prevention, classification, diagnostic tests, treatment and management, and health system issues (e.g., increased health care services use and policy impact).7 A relatively small proportion (n = 74 of 637; 11.6%) of the published reports evaluated the treatment or management of post–COVID-19 condition.7 Most of the evidence-based guidelines did not provide recommendations specific to post–COVID-19 condition according to the WHO definition (i.e., patients with symptoms at least 3 months after initial infection).7 However, there were 67 registrations or protocols of clinical trials that aimed to investigate interventions for the treatment of post–COVID-19 condition.7 Similarly, a recent systematic review identified 6 published clinical trials and 54 trial registrations on the drugs of post–COVID-19 condition.8 Other author groups have identified at least 26 ongoing clinical trials in which interventions for post–COVID-19 condition were evaluated.9 It remains uncertain what kind of information the protocols or clinical trials might provide for the treatment or management of post–COVID-19 condition. Herein, we have updated (with modification) the treatment and management concept within CADTH’s scoping review in response to stakeholder requests and to understand the direction of research about the treatment and management of post–COVID-19 condition.7
The rapidly changing evidence base warrants continuous efforts to characterize the literature and identify research gaps; therefore, we transitioned the scoping review into living mode.
Objective
The purpose of this rapid living scoping review is to provide an up-to-date map of the latest published evidence (including primary studies, evidence syntheses, and guidelines) available in peer-reviewed journals and grey literature, and to identify research gaps regarding the treatment and management of post–COVID-19 condition in any population and setting. In clinical practice, management can refer to a broad spectrum that includes specific treatments and interventions.10 To avoid duplication of efforts, the scope of this review focuses on the treatment and management of post–COVID-19 condition, but excludes rehabilitation because this is being covered in a Cochrane living review.11,12
Research Questions
The following research questions are addressed in the scoping review:
What is the current evidence landscape on the treatment and management of post–COVID-19 condition for people of any age in any setting?
What are the knowledge gaps on the treatment and management of post–COVID-19 condition for people of any age in any setting?
Methods
Protocol Development
The protocol for this rapid living scoping review13 was adapted from the protocol of the previously published CADTH scoping review.7 We adapted our previous methodology to enable the review to be performed rapidly and chose to focus on final reports of studies with results (rather than protocols, registrations, and pre-prints) because we planned to move the scoping review into living mode to keep pace with developments in the evidence base. The protocol and this scoping review report were externally reviewed by content experts. We registered the review topic at the National Collaborating Centre for Methods and Tools (NCCMT); to increase awareness and avoid duplication, and posted the protocol on the Open Science Framework (OSF).
Study Design
We conducted a rapid living scoping review of published primary studies, evidence syntheses, economic evaluations, and evidence-based guidelines. We have reported the scoping review according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses extension for Scoping Reviews (PRISMA-ScR)14 with considerations for updates provided in PRISMA 2020.15
Eligibility Criteria
The review’s eligibility criteria, including the specific populations, concepts, and contexts (PCC) of interest are shown in Table 1. The inclusion criteria were informed by CADTH Horizon Scanning16 and adapted from CADTH’s published scoping review on post–COVID-19 condition,7 with a focus on treatment and management. We included full reports with results of primary studies, evidence syntheses, economic evaluations, and evidence-based guidelines that provided results related to any treatment or management option (except rehabilitation) used for post–COVID-19 condition among people of all ages in any setting. A definition of “rehabilitation” was not reported in the Cochrane review17 and the corresponding authors could not be reached. We adopted the definition of rehabilitation provided by WHO, to include interventions designed to improve physical functioning and minimize disability.18 This included interventions related specifically to the concepts of rehabilitation, respiratory rehabilitation, exercise, physical therapy modalities, physical therapy, functional recovery, and occupational therapy.19
Table 1: Selection Criteria for Clinical Research Questions
Category | Selection criteria |
|---|---|
Population | People of all ages with post–COVID-19 condition, defined as any symptoms experienced 12 weeks or more after initial infection, diagnosis, or symptom onset that cannot be explained by another cause For Canadian guidelines, any timing of post–COVID-19 condition was accepted (i.e., could be < 12 weeks). |
Concept | Any health care options related to treatment and/or management of post–COVID-19 conditions including but not limited to:
Exclude: rehabilitationa |
Context | Any contextual setting (e.g., provided in person or virtually) |
Study designs | Full reports of:
Exclusions:
|
Time frame | 2019 to present (sources searched between 2019 and February 20227 rescreened as needed and merged with those searched in this update) |
Language of publication | English or French |
aRehabilitation includes interventions designed to improve physical functioning and minimize disability.25 Although the Cochrane Rehabilitation team did not publish a definition of rehabilitation,11 publications were identified by searching bibliographic databases using keywords and subject headings for rehabilitation, respiratory rehabilitation, exercise, physical therapy modalities, physical therapy, functional recovery, and occupational therapy.19 These interventions were considered rehabilitation interventions and were not eligible in this report. When rehabilitation was included as part of a multi-component intervention, the study was included.
bThese may be quantitative, qualitative, or mixed methods, and must include a research question; a list of the sources searched and a reproducible search strategy; clear inclusion and exclusion criteria; a description of methods for study selection; and information about how the data were synthesized. Although appraisal of the quality of the included studies is often recommended for systematic reviews,20 we did not consider this to be required for the purpose of the present scoping review.
cSystematic reviews using abbreviated methods.
dThe same criteria as systematic reviews, except the unit of analysis is systematic reviews rather than primary studies.
Literature Search Strategy
The literature search was performed by an information specialist using a peer-reviewed search strategy according to the PRESS (Peer Review of Electronic Search Strategies) checklist.21 The complete search strategy is presented in Appendix 1.
Published literature was identified by searching the following bibliographic databases: MEDLINE and Embase via the Ovid platform, and the Cochrane Database of Systematic Reviews. All Ovid searches were run simultaneously as a multi-file search. Duplicates were removed using Ovid deduplication for multi-file searches, followed by manual deduplication in Endnote. The literature search strategy used in this report is a modified continuation of one developed for the original CADTH post–COVID-19 condition scoping review.7 The search strategy comprised both controlled vocabulary, such as the National Library of Medicine’s MeSH (Medical Subject Headings), and keywords. The main search concept was post–COVID-19 condition or synonyms, plus a new concept for treatment and management. Parts of the strategy were adapted from CADTH’s COVID-19 search string.22
The search for the original scoping review7 was limited to English- or French-language documents published between January 1, 2019, and December 21, 2021, with search updates until February 4, 2022. For the current review, database searches were rerun on July 20, 2022, and updated on October 21, 2022, to capture any articles made available on post–COVID-19 treatment or management since the initial search date. Where possible, retrieval was limited to the human population. No filters were applied to limit the retrieval by study type. For this review, conference abstracts and pre-prints were excluded, as well as comments, newspaper articles, editorials, and letters.
Grey literature (literature that is not commercially published) was identified by searching sources listed in relevant sections of Grey Matters: A Practical Tool For Searching Health-Related Grey Literature checklist,23 and CADTH COVID-19 Grey Literature Resources,24 which includes the websites of regulatory agencies, health technology assessment (HTA) agencies, clinical guideline repositories, systematic review repositories, patient-related groups, and professional associations. Google was used to search for additional internet-based materials. These searches were supplemented by reviewing bibliographies of key papers and through contacts with experts (via email), as appropriate. Refer to Appendix 1 for more information on the grey literature search strategy.
Regular alerts will update the database literature searches and grey literature searches for this living review. The most recent search update incorporated into this report was conducted on October 21, 2022.
Study Selection
The systematic review management software DistillerSR (Evidence Partners, Ottawa, Canada)25 was used to facilitate study selection. Pilot testing was conducted for a random sample of 50 references identified in the literature search to ensure a thorough understanding of the eligibility criteria. Then, 1 reviewer screened titles and abstracts of all citations retrieved from the literature search. Full texts of records that were judged to be potentially relevant by the reviewer were retrieved based on the pre-determined selection outlined in Table 1. One reviewer examined all full-text articles for inclusion in the review. When the reviewer was uncertain about whether to include certain articles, these were discussed with the team, who came to consensus on inclusion or exclusion. Due to time and resources constraints, we did not contact article authors in the event of unclear or conflicting information in the literature. Instead, a team decision about inclusion or exclusion was made based on the available information. The reasons for exclusion of articles at the full-text level were documented.
Sources included in the previous CADTH review7 that were relevant to treatment and management were included in the current scoping review. These were rescreened by a single reviewer to exclude those that focused solely on rehabilitation.
Charting (Data Extraction)
Charting was performed by 1 reviewer, who authored the previously published CADTH scoping review.7 Before formal charting, 2 members of the review team tested the data extraction form on a random sample of 5 included studies and met to review discrepancies. The form was edited and further pilot exercises were run, until the single reviewer was ready to proceed with the full data charting. In the event of unclear or conflicting information, the reviewer consulted the team. Relevant information was charted, including the following:
study characteristics (e.g., first author’s name, publication year, country or countries of the participants) and methodology (e.g., study design, methods to confirm COVID-19 infection)
population (e.g., number of participants, age, sex and/or gender, race or ethnicity, vaccination status)
concept (e.g., treatment details, comparators, and follow-up times)
context in which the treatment was being offered (i.e., acute care hospital, intensive care unit, outpatient clinic, community, nursing home, and virtual care).
Data were charted for all relevant concepts and contexts at any duration of follow-up. Specific population characteristics (including race, ethnicity, culture, language, occupation, sex and/or gender, education, socioeconomic status, and other characteristics that might be associated with barriers) were charted to identify characteristics that stratified health opportunities and outcomes related to health equity and equity considerations.26 We recorded studies as being relevant to these characteristics when 65% or more (i.e., the majority) of the population fit into a specific category (e.g., 65% of the population was all of a single ethnicity). In a previous scoping review, most studies did not report population characteristics, except for sex/gender and age, or investigated populations with mixed characteristics.7 We adopted a 65% threshold to select references that focused on certain characteristics that might be associated with greater risks of infection or developing post–COVID-19 condition.
In June 2022, the Institut national d’excellence en santé et en services sociaux (INESSS) published a post–COVID-19 condition management support tool intended for front-line clinicians.27 The tool’s development included a systematic review of accepted clinical practice guidelines at the time it was drafted, and relied on the knowledge, experience, and contribution of stakeholders across Quebec.27 We adopted this categorization in our charting of the types of treatment and management, and tests to assist in management. The tests to assist in management included basic tests, specialized tests, and investigations.28 Basic tests to be considered included blood tests for whole blood, liver function, inflammation markers, thyroid function, diabetes, and existing comorbidities.28 Specialized tests included blood tests for coagulation disorder, myocardial injury, viral infection, and rheumatological conditions.28 The investigations included chest X-ray and echocardiography.28
Given the findings of our previous scoping review,7 we anticipated there would be a large volume of case reports and case series in the literature that included small numbers of patients. Because studies of larger numbers of patients were considered to be more informative, and to ensure the feasibility of data charting with a small review team, we did not perform full data charting, and instead only collected and reported the details of the publication, population, treatment or management, and testing to assist in management for case reports and case series. We used customized charting forms for guidelines and evidence syntheses. Characteristics were extracted directly as reported by the guideline and systematic review authors. We did not retrieve their included primary studies for further information. When applicable, population characteristics were described using available data, such as ranges of median ages and sample sizes reported in primary studies.
Attempts were not made to contact the corresponding authors of these studies to obtain missing information or to clarify conflicting information due to time and resource constraints. Data charting was an iterative process, whereby additional items were added as the research team learned about the research base and recognized new items of importance. The full list of data that were charted can be found in Appendix 2, Table 12.
Quality Appraisal
As it was not within the purpose of the scoping review to consider the quality of the included studies, we did not perform a quality appraisal.
Descriptive Synthesis
Descriptive mapping was performed, including the presentation of study characteristics within summary tables, visual displays, and in the main text. For example, we tabulated the categories of post–COVID-19 condition against population characteristics, diagnoses, and study design. Study characteristics that often were not mutually exclusive were presented in tables as suggested by the JBI manual.29 When possible, we presented the case series separately from other study designs to avoid overemphasizing these studies that include few participants. The treatment and management strategies were grouped in categories, such as clinical manifestations classified by the organ systems the interventions aim to treat.30,31 The characteristics of the included guidelines and systematic reviews are summarized in separate tables.7
The results are also presented in diagrams, such as bar charts or other visual displays, to demonstrate the distributions of the publications across different topics, disciplines, countries, and other categories. Moreover, additional figures or tables that demonstrate the details in the evidence base and gaps in evidence, as well reference lists that may be filtered by topic, are available online on the project page.
. The visualization helped to identify potential gaps in research related to the treatment of post–COVID-19 condition. The gaps were also identified through the examination of the distributions of studies across patient characteristics, clinical manifestations and organ systems, and other characteristics.
Patient Engagement
Prior to completion of the scoping review, we identified 3 adults (aged 18 years or older) who are currently living with post–COVID-19 condition through contact with support groups, clinical experts involved in the project, CADTH Liaison Officers, and other CADTH networks across Canada. These people with lived experience were invited to attend a meeting to discuss the scoping review. At the meeting, CADTH staff presented the findings of the scoping review, and a patient engagement officer facilitated a discussion about interventions or treatments, areas of controversy or uncertainty, and priorities from their perspectives. The patients were provided the opportunity to ask questions, and to provide context and considerations about the findings. These views and comments were incorporated into the scoping review.
Their contribution is acknowledged in the report, and their involvement is reported using the Guidance for Reporting Involvement of Patients and the Public 2 (GRIPP2) Framework.32 CADTH will continue to engage patient partners with the ongoing development of the findings and future updates.
Future Updates
The COVID-19 pandemic remains an ongoing issue and new SARS-CoV-2 variants are spreading quickly in Canada.33 With more new cases of infection, the burden of post–COVID-19 condition is expected to grow.34 The rapid changes in the pandemic and the increasing number of therapeutic interventions tested in clinical trials make it important to continue monitoring new and emerging strategies to manage and treat post–COVID-19 condition. Considering the need to keep pace with the evolving evidence base, we plan to initially update our dashboard found on our project page every 3 months. The team will meet and decide whether there is a need to continue to perform regular updates or to change the inclusion criteria to exclude certain study designs, and may modify the frequency of the updates or choose to take the review out of living mode. This decision will be based on the volume of accumulating literature or the significance of the finding in primary studies at each update; for example, if few studies are identified in previous updates, it may be reasonable to extend the time lag between updates without the scoping review becoming substantially out of date. We may choose to transition out of living mode when the accumulation of new information becomes slow enough that continual updates do not seem informative, or if there is a shift in priorities to other topics. At this time, we will publish a final version of the report. To accomplish the updates, we will continue using the same review methods until specified otherwise. The reasons to transition out of living or the deviations to the methods will be listed with reasons in the latest version of the report. In each update, the literature search results will be screened at once. We will synthesize the data extracted in previous reports and the update to draw conclusions. We will determine and report whether there are any changes in the conclusions compared to previous reports.
Results
Quantity of Research Available
A total of 1,131 unique citations were identified in the electronic literature search. Following screening of titles and abstracts, 104 potentially relevant reports were retrieved for full-text review, in addition to 34 from the grey literature search and 74 from the baseline review.7 Of these 212 potentially relevant articles, 113 reports were included in this scoping review. The study selection process is outlined in Appendix 2, Figure 3.
Study Characteristics
The published sources included 11 interventional primary studies (9.6%), 81 observational primary studies (70.4%), 2 qualitative primary studies (1.7%), 7 systematic reviews (7.0%), 5 rapid reviews (4.3%), 4 Canadian guidelines (3.5%), and 3 guidelines from other countries (3.5%). Among the 81 observational primary studies, 63 (77.8%) were case reports or series.
The included reports were published between November 16, 2020, and October 25, 2022 (including reports that were available online by October 21, 2022, but were officially published later). On average, there were 4.6 included studies that became available monthly (median = 3; interquartile range, 1 to 7). The number of studies published over time is presented in Figure 1.
The number of reports (including primary studies, rapid and systematic reviews) by country of the included participants or country in which the guidelines were applicable (in countries with more than 2 included reports) is presented in Figure 2. A large proportion of reports included participants or were applicable to populations from only a few countries, particularly the US (n = 31), the UK (n = 11), Italy (n = 9), Spain (n = 7), and Canada (n = 7). There was lesser representation (3 to 6 reports) of participants or populations the guidelines were applicable to from Japan, India, and certain European countries (primarily Turkey, Switzerland, and the Netherlands). Relatively few reports (1 or 2) included participants from Africa, South America, Asia, and other European countries.
Figure 1: Number of Included Studies by Month of Publication
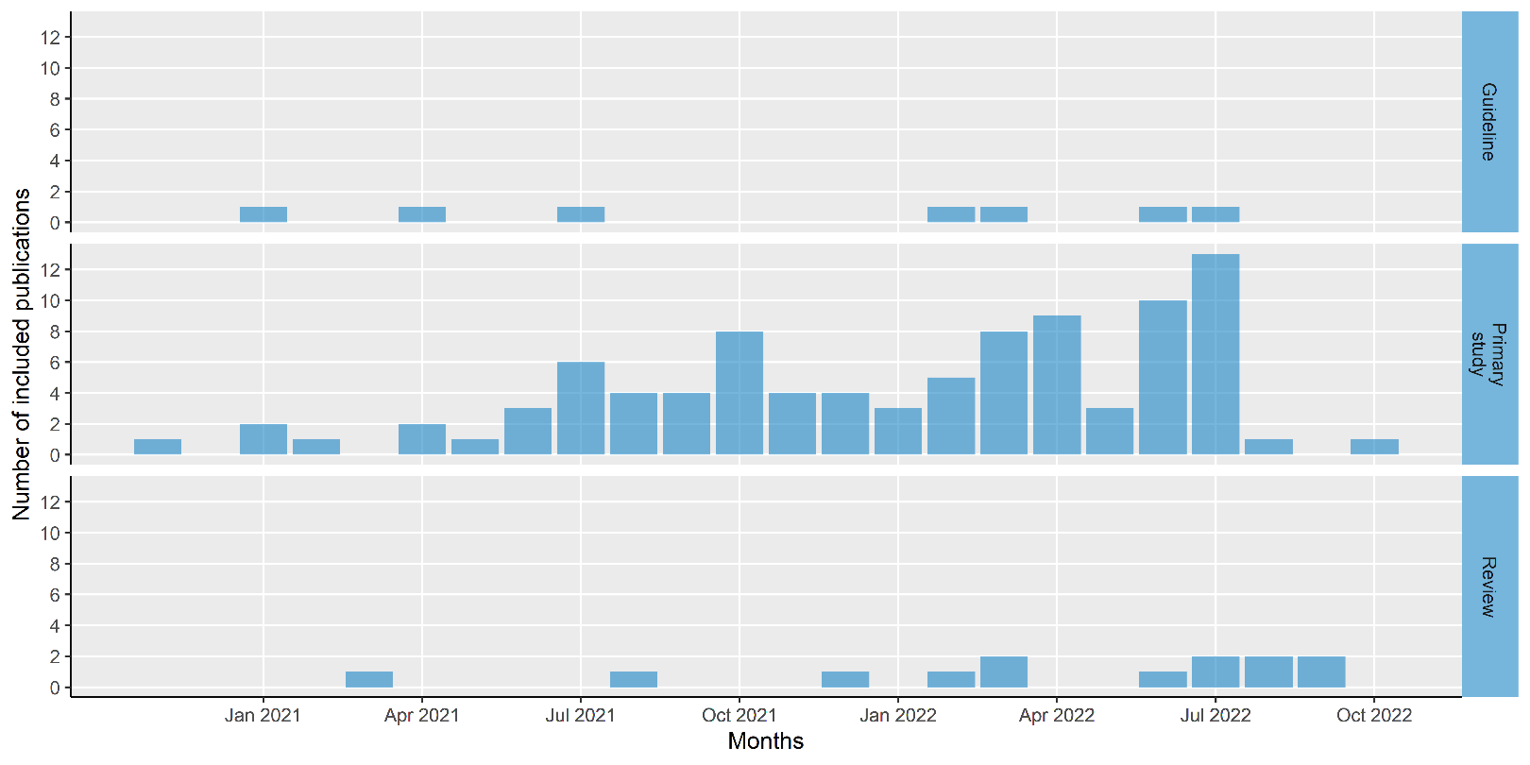
Guideline = Canadian guidelines and those from other countries; Primary study = interventional, observational, and qualitative primary studies; Review = both rapid and systematic reviews.
Note: Based on literature available by October 2022. The first date online was extracted where available, but this was not always available, in which case the publication date was used. Some studies may have been available by October 2022, but were not yet officially published; thus, some of the studies in the figure are presented as published in August.
Figure 2: Number of Included Reports by Country of Participants
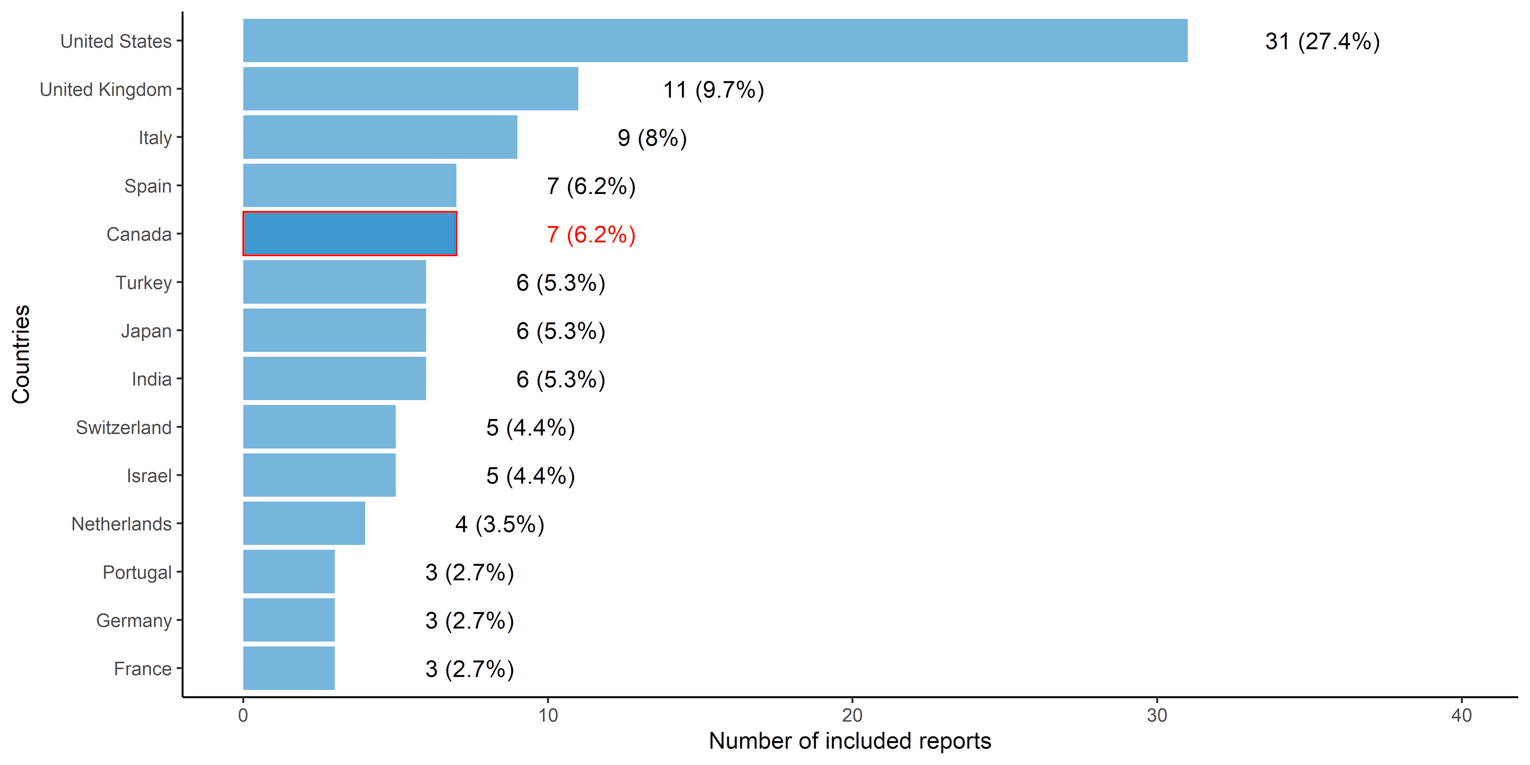
Note: Reports include primary studies, rapid or systematic reviews, and guidelines. Countries with 3 or more included reports are shown.
Study Characteristics
Table 2 presents the characteristics of participants in the primary studies, excluding case reports/series, and shows the study characteristics, with studies classified by indication/diagnosis. Among the 31 primary studies, the median sample size was 47 (range, 9 to 160,663). The following age groups were represented: children (younger than 18 years of age) (n = 4; 12.9%), adults (18 years of age or older) (n = 29; 93.5%), older adults (65 years of age or older) (n = 12; 38.7%), and not reported (n = 1; 3.2%). The sex or gender distribution was reported in 26 studies (83.9%). The median proportion of males was 34.0% (interquartile range = 17.4% to 41.1%).
We aimed to identify sources that focused on equity considerations by characterizing the research available for certain groups potentially facing disadvantages, or those with greater risk of infection based on the population characteristics (i.e., studies where these populations represented at least 65% of the sample). The groups that we identified were health care workers (n = 4; 13.8%) and people who were unemployed (n = 1; 3.4%), while the socioeconomic characteristics that we aimed to chart were not reported in 24 primary studies (82.8%). Three studies focused on certain races or ethnicities: Asian (n = 1; 3.4%) and White (n = 2; 6.8%).
Using the same 65% threshold, we identified several studies with populations of patients having comorbidities: HIV infection (n = 1; 3.2%), cancer (n = 2; 6.5%), cardiovascular or heart disease (n = 1; 3.2%), diabetes (n = 3; 9.7%), hypertension (n = 4; 12.9%), kidney disease (n = 2; 6.5%), mental illnesses (n = 3; 9.7%), and obesity (n = 2; 6.5%). Seventeen primary studies (54.8%) did not report any information on comorbidities.
We recorded vaccination status and identified studies that fit into the following groupings: all participants vaccinated (1 or 2 doses depending on the vaccines) (n = 1; 3.2%), mixed (defined as some participants vaccinated and others not) (n = 2; 6.5%), and participants not vaccinated (n = 1; 3.2%). Most studies did not report any information on vaccination status (n = 27; 87.1%).
Table 2: Basic Characteristics of Included Primary Studies (Excluding Case Reports/Series), by Classification of Indication/Diagnosis
Classification of indication/diagnosis | Cardiovascular | Dermatologic | Fatigue | Generalized or unspecified symptoms | Mental and behavioural | Neurologicalal | Otorhinolaryngologic | Pulmonary | Renal | Rheumatologic | Total |
|---|---|---|---|---|---|---|---|---|---|---|---|
Total, N | 1 | 1 | 6 | 11 | 4 | 6 | 6 | 5 | 1 | 1 | 31 |
Study design, n (%) | |||||||||||
Primary study-observational | 1 (100) | 1 (100) | 2 (33.3) | 9 (81.8) | 4 (100) | 5 (83.3) | 2 (33.3) | 3 (60) | 1 (100) | 1 (100) | 18 (58.1) |
Primary study-interventional | 0 | 0 | 3 (50) | 1 (9.1) | 0 | 1 (16.7) | 4 (66.7) | 2 (40) | 0 | 0 | 11 (35.5) |
Primary study-qualitative | 0 | 0 | 1 (16.7) | 1 (9.1) | 0 | 0 | 0 | 0 | 0 | 0 | 2 (6.5) |
Age category, n (%) | |||||||||||
Children/pediatric (0 to < 18 years) | 0 | 0 | 0 | 2 (18.2) | 0 | 0 | 1 (16.7) | 1 (20) | 0 | 0 | 4 (12.9) |
Adults (18 years and older) | 1 (100) | 1 (100) | 5 (83.3) | 10 (90.9) | 4 (100) | 6 (100) | 6 (100) | 5 (100) | 1 (100) | 1 (100) | 29 (93.5) |
Older adults (65 years and older) | 0 | 0 | 2 (33.3) | 5 (45.5) | 2 (50) | 1 (16.7) | 2 (33.3) | 2 (40) | 0 | 0 | 12 (38.7) |
Not reported | 0 | 0 | 1 (16.7) | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 (3.2) |
Sex | |||||||||||
Male (%), median (interquartile range) | 19.4 (19.4 to 19.4) | 19.4 (19.4 to 19.4) | 15 (0 to 30) | 40.9 (27.0 to 53.6) | 25.5 (19 to 32.5) | 25.5 (19.8 to 37.2) | 34.6 (33.3 to 36.2) | 18 (14.3 to 26.1) | 82.2 (82.2 to 82.2) | 21 (21 to 21) | 34 (17.4 to 41.1) |
NA = not available.
Note: Eligible references could be included under more than 1 age category. Classification of indication/diagnosis was based on the categories of manifestations that the Institut national d’excellence en santé et en services sociaux (INESSS) recommended clinicians to consider,28 except for fatigue, gastrointestinal, and generalized or unspecified symptoms.
Acute Infection in Primary Studies, Excluding Case Reports/Series
We collected information on the severity of acute infection (defined as asymptomatic, symptomatic but not hospitalized, hospitalized, or in intensive care unit [ICU]), with categories that were not mutually exclusive (i.e., a study could include patients with different severities). Severity was not reported in 17 (54.8%) of the primary studies. One study (3.2%) included participants who had been asymptomatic, 11 (35.5%) included participants who had been symptomatic but not hospitalized, 10 (32.3%) included participants who had been hospitalized, 7 (22.6%) included participants who had been admitted into ICUs, and 1 (3.2%) included participants who were classified using other severity categories.
There were multiple methods for diagnosing or identifying COVID-19 infection. Overall, 11 (35.5%) studies did not report the methods. There were 15 studies (48.4%) that used polymerase chain reaction tests, 4 (12.9%) that used antibody tests, 3 (9.7%) that used other or unspecified lab tests, 1 (3.2%) that used other tests (e.g., chest CT), 1 (3.2%) that was diagnosed by a clinician, and 2 (6.5%) that used self-report.
Participant Characteristics in Case Reports/Series
Among the 63 case reports/series, 55 reported single cases, 5 reported 2 cases, 1 reported 4 cases, 10 reported 10 cases, and 1 reported 20 cases. The case reports/series included participants in the following age groups: children (n = 5; 7.9%), adults (n = 52; 82.5%), and older adults (n = 6; 9.5%). The sex distribution was reported in 63 (100%) reports. Twenty-five (39.7%) and 34 (54.0%) included males or females only, respectively. In the other 4 case reports/series, 10%, 25%, 30%, and 50% of the participants were male.
Treatment and Management
Classification of Indications and Diagnoses in Primary Studies, Excluding Case Reports/Series
We categorized the indication or diagnosis that the management and treatment aimed to address into the following categories: generalized or unspecified (n = 11; 35.5%), fatigue (n = 6; 19.4%), neurological (n = 6; 19.4%), otorhinolaryngological (n = 6; 19.4%), pulmonary (n = 5; 16.1%), mental and behavioural (n = 4; 12.9%), cardiovascular (n = 1; 3.2%), dermatological (n = 1; 3.2%), renal (n = 1; 3.2%), rheumatological (n = 1; 3.2%), and other conditions (such as eye conditions; n = 1; 3.2%).
Treatment and Management in the Primary Studies, Excluding Case Reports/Series
We first defined the major groups of treatment and management for post–COVID-19 condition: pharmacological interventions (n = 20; 64.5%), non-pharmacological interventions (n = 14; 45.2%), care models (n = 2; 6.5%).13 We further stratified these interventions into 10 subgroups: drugs (n = 14; 45.2%), surgery/procedure (n = 9; 29%), vaccine (n = 5; 16.1%), rehabilitation (used in combination with other eligible interventions; n = 4; 12.9%), care models (n = 2; 6.5%), herbal/Chinese medicine (n = 2; 6.5%), psychological support (n = 2; 6.5%), oxygen (n = 1; 3.2%), physiotherapy (used in combination with other eligible interventions; n = 1; 3.2%), and supplements (n = 1; 3.2%).
We identified basic tests (n = 11; 35.5%), specialized tests (n = 13,; 41.9%), investigations (n = 4; 12.9%), and a “not reported” category (n = 14; 45.2%) used as guidance for post–COVID-19 care in the included studies.
We identified the following settings of treatment: outpatient clinic (n = 20; 64.5%), acute care hospital (n = 6; 19.4%), community (n = 5; 16.1%), or not reported (n = 1; 3.2%).
Details in Treatment and Management, Excluding Case Reports/Series
The interventions were either single-component (n = 21; 67.7%) or multi-component (n = 10; 32.3%).
Interventions were initiated during various periods of time after COVID-19 infection: 6 months (n = 12; 38.7%), 7 to 12 months (n = 12; 38.7%), 3 months (n = 11; 35.5%), 5 months (n = 10; 32.3%), 4 months (n = 9; 29%), 13 months or later (n = 3; 9.7%), 1 month (n = 2; 6.5%), 2 months (n = 2; 6.5%), or not reported (n = 2; 6.5%).
The follow-up time also varied across the studies: no follow-up (cross-sectional design) (n = 5; 16.1%), 1 month (n = 11; 35.5%), 3 months (n = 11; 35.5%), 2 months (n = 6; 19.4%), 4 months (n = 4; 12.9%), not reported (n = 3; 9.7%), 5 months (n = 2; 6.5%), 7 to 12 months (n = 2; 6.5%), and 6 months (n = 1; 3.2%).
We identified several types of comparators to the treatment and management in the primary studies that were not case reports or series: no comparator (n = 12; 38.7%), standard care (n = 6; 19.4%), drugs (n = 3; 9.7%), placebo (n = 3; 9.7%), rehabilitation (n = 3; 9.7%), matched controls (not healthy) (n = 2; 6.5%), patients (n = 2; 6.5%), healthy controls (n = 1; 3.2%), and non-drug interventions (n = 1; 3.2%).
Among the comparators reported, we identified single-component (n = 12; 38.7%) and multi-component comparators (n = 4; 12.9%), while some were not reported (n = 3; 9.7%).
Classification of Indications and Diagnoses in Case Reports/Series
We also categorized the indication or diagnosis in case reports/series: pulmonary (n = 15; 23.8%), neurological (n = 13; 20.6%), cardiovascular (n = 12; 19%), fatigue (n = 7; 11.1%), gastrointestinal (n = 7; 11.1%), otorhinolaryngological (n = 5; 7.9%), mental and behavioural (n = 4; 6.3%), generalized or unspecified (n = 3; 4.8%), rheumatological (n = 3; 4.8%), hematological (n = 2; 3.2%), dermatological (n = 1; 1.6%), and others (such as patients with metabolic disease or concurrent cancer treatment; n = 14; 22.2%).
Treatment and Management in Case Reports/Series
The major groups of treatment and management for post–COVID-19 condition in case reports/series were identified: pharmacological interventions (n = 41; 65.1%) and non-pharmacological interventions (n = 36; 57.1%). We further stratified these interventions into 9 subgroups: drugs (n = 38; 60.3%), surgery/procedure (n = 27; 42.9%), oxygen (n = 4; 6.3%), herbal/Chinese medicine (n = 3; 4.8%), supplements (n = 3; 4.8%), behavioural management (n = 2; 3.2%), psychological support (n = 2; 3.2%), physiotherapy (used in combination with other eligible interventions; n = 1; 1.6%), and vaccine (n = 1; 1.6%).
We identified the following settings of treatment: acute care hospital (n = 36; 57.1%), outpatient clinic (n = 25; 39.7%), not reported (n = 3; 4.8%), and community (n = 1; 1.6%).
The frequencies of the types of treatment and management in primary studies (excluding case reports/series) are listed in Table 3.
Table 3: Treatment or Management in Included Primary Studies (Excluding Case Reports/Series), by Classification of Indication/Diagnosis
Classification of indication | Cardiovascular | Dermatological | Fatigue | Generalized or unspecified | Mental and behavioural | Neurological | Otorhinolaryngological | Pulmonary | Renal | Rheumatological | Total |
|---|---|---|---|---|---|---|---|---|---|---|---|
Total, N | 1 | 1 | 6 | 11 | 4 | 6 | 6 | 5 | 1 | 1 | 31 |
Type of treatment, n (%) | |||||||||||
Pharmacological interventions | 1 (100) | 1 (100) | 3 (50) | 8 (72.7) | 2 (50) | 3 (50) | 4 (66.7) | 2 (40) | 1 (100) | 1 (100) | 20 (64.5) |
Non-pharmacological interventions | 0 | 0 | 4 (66.7) | 1 (9.1) | 3 (75) | 3 (50) | 5 (83.3) | 2 (40) | 0 | 0 | 14 (45.2) |
Care models | 0 | 0 | 0 | 2 (18.2) | 0 | 0 | 0 | 1 (20) | 0 | 0 | 2 (6.5) |
Treatment details, n (%) | |||||||||||
Drugs | 0 | 0 | 2 (33.3) | 3 (27.3) | 2 (50) | 2 (33.3) | 4 (66.7) | 2 (40) | 1 (100) | 1 (100) | 14 (45.2) |
Physiotherapy (used in combination with other eligible interventions) | 0 | 0 | 1 (16.7) | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 (3.2) |
Psychological support or treatment | 0 | 0 | 1 (16.7) | 0 | 1 (25) | 0 | 0 | 0 | 0 | 0 | 2 (6.5) |
Supplements | 0 | 0 | 0 | 0 | 0 | 0 | 1 (16.7) | 0 | 0 | 0 | 1 (3.2) |
Oxygen | 0 | 0 | 0 | 0 | 0 | 1 (16.7) | 0 | 0 | 0 | 0 | 1 (3.2) |
Surgery or procedure | 0 | 0 | 3 (50) | 1 (9.1) | 2 (50) | 3 (50) | 2 (33.3) | 2 (40) | 0 | 0 | 9 (29) |
Behavioural management | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 (0) |
Herbal/Chinese medicine | 0 | 0 | 1 (16.7) | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2 (6.5) |
Rehabilitation (used in combination with other eligible interventions) | 0 | 0 | 1 (16.7) | 0 | 0 | 0 | 3 (50) | 0 | 0 | 0 | 4 (12.9) |
Care models | 0 | 0 | 0 | 2 (18.2) | 0 | 0 | 0 | 1 (20) | 0 | 0 | 2 (6.5) |
Vaccine | 1 (100) | 1 (100) | 0 | 5 (45.5) | 0 | 1 (16.7) | 0 | 0 | 0 | 0 | 5 (16.1) |
Tests to assist in treatment and management, n (%) | |||||||||||
Basic tests | 0 | 0 | 2 (33.3) | 3 (27.3) | 2 (50) | 3 (50) | 3 (50) | 2 (40) | 1 (100) | 1 (100) | 11 (35.5) |
Specialized tests | 0 | 0 | 2 (33.3) | 3 (27.3) | 1 (25) | 2 (33.3) | 4 (66.7) | 2 (40) | 1 (100) | 0 | 13 (41.9) |
Investigations | 0 | 0 | 2 (33.3) | 0 | 1 (25) | 2 (33.3) | 0 | 1 (20) | 0 | 0 | 4 (12.9) |
Not reported | 1 (100) | 1 (100) | 4 (66.7) | 7 (63.6) | 2 (50) | 3 (50) | 0 | 2 (40) | 0 | 0 | 14 (45.2) |
Single or multi-component treatment, n (%) | |||||||||||
Multiple | 0 | 0 | 1 (16.7) | 3 (27.3) | 1 (25) | 0 | 3 (50) | 2 (40) | 1 (100) | 0 | 10 (32.3) |
Single | 1 (100) | 1 (100) | 5 (83.3) | 8 (72.7) | 3 (75) | 6 (100) | 3 (50) | 3 (60) | 0 | 1 (100) | 21 (67.7) |
Guidelines
We identified 4 Canadian guidelines27,35-37 and 3 international guidelines.38-40
In Canada, guidelines from Alberta,35 British Columbia,36 Ontario,37 and Quebec27 were identified; the methods and relevant recommendations (i.e., recommendations specific to 12 weeks or later) are summarized in Table 4 and Table 5, respectively. The Alberta guideline did not define post–COVID-19 condition.35 The Ontario guideline was applicable to those experiencing symptoms at least 4 weeks after initial infection.37 The British Columbia36 and Quebec27 guidelines focused on the patients with symptoms at least 3 months after initial infection. The Canadian guidelines varied in the target population, interventions of interest, emphasis on self-management, and level of detail in the recommended care models.27,35-37
Table 4: Methods Used by Canadian Guidelines for Post–COVID-19 Care
Method | Alberta35 | British Columbia36 | Ontario37 | Quebec27 |
|---|---|---|---|---|
Funding/sponsorship | NR | NR | NR | NR |
Did the guideline describe their methodology? | Yes | No | Yes | Yes |
Method(s) used | ||||
Systematic literature search (search strategy provided) a | No | NR | No | Yes |
Literature search, unclear if systematic | Yes | NR | No | No |
Expert feedback | No | NR | No | No |
Stakeholder feedback/consultation | No | NR | No | Yes |
Based on a specific guideline b | No | NR | Yes | No |
Authors’ clinical experience | No | NR | No | No |
Literature searched | ||||
CINAHL | No | NR | NR | No |
Cochrane library | No | NR | NR | Yes |
EMBASE | Yes | NR | NR | No |
Guidelines databases | No | NR | NR | Yes |
MEDLINE | Yes | NR | NR | No |
PubMed | Yes | NR | NR | Yes |
Other databases | No | NR | NR | Yes |
Other (e.g., reference list searching) | No | NR | NR | No |
End of literature search date | NR | NR | NR | NR |
Publication date | July 2021 | NR | December 2021 | June 2022 |
NA = not applicable; NR = not reported.
aIf the methods provided a detailed search strategy and/or specified their search was systematic.
bThe guideline based their recommendations on what a previously published guideline recommends.
Table 5: Summary of Recommendations From Canadian Guidelines for Post–COVID-19 Care
Recommendation | Alberta35 | British Columbia36 | Ontario37 | Quebec27 |
|---|---|---|---|---|
Population | Children, adultsa | NR | Adults | Children, adultsa |
Classification of indication/diagnosis | Mental and behavioural; others; renal; pulmonary | Mental and behavioural disorders; neurological; others; pulmonary | Generalized or unspecified | Cardiovascular; dermatological; hematological; mental and behavioural; neurological; otorhinolaryngological; renal; pulmonary; rheumatological |
Type of treatment and management | Non-pharmacological interventions ; pharmacological interventions | Care models | Care models ; non-pharmacological interventions ; pharmacological interventions | Care models ; non-pharmacological interventions ; pharmacological interventions |
Management | ||||
Self-management support | Yes | Yes | Yes | Yes |
Referral to specialist(s) | Yes | NR | NR | Yes |
Referral to post–COVID-19 clinic | Yes | Yes | NR | Yes |
NR = not reported.
aSummarizes literature on children and adults, but unclear if guidance specific to 12+ weeks applies to children.
Three guidelines for patients in Europe,38 Spain,38,40 and the UK40 focused on patients with post–COVID-19 condition according to the WHO definition (i.e., 12 or more weeks following initial infection). These guidelines are summarized in Table 6 and Table 7.
Table 6: Methods Used by International Guidelines for Post–COVID-19 Care
Method | Europe38 | Spain39 | UK40 |
|---|---|---|---|
Funding/sponsorship | None | None | NR |
Did the guideline describe their methodology? | Yes | Yes | Yes |
Method(s) used | |||
Systematic literature searcha | Yes | Yes | Yes |
Literature search, unclear if systematic | NA | NA | NA |
Expert feedback | No | Yes | Yes |
Stakeholder feedback/consultation | No | No | Yes |
Based on a specific methodological guidelineb | No | No | No |
Authors’ clinical experience | Yes | Yes | No |
Literature searched | |||
CINAHL | NR | NR | Yes |
Cochrane library | Yes | NR | Yes |
Embase | NR | NR | Yes |
Guidelines databases | NR | NR | NR |
MEDLINE | NR | NR | Yes |
PubMed | Yes | Yes | NR |
Other databases | Yes | NR | Yes |
Other sources (e.g., reference checking) | Yes | NR | Yes |
End of literature search date | December 31, 2021 | January 13, 2021 | NR |
Publication date | February 17, 2022 | April 2021 | January 3, 2022 |
NR = not reported; NA = not applicable.
aIf the methods provided a detailed search strategy and/or specified their search was systematic.
bThe guideline based their recommendations on what a previously published guideline recommends.
Table 7: Summary of Recommendations by International Guidelines for Post–COVID-19 Care
Recommendation | Europe38 | Spain39 | UK40 |
|---|---|---|---|
Population | Adults | NR | Children, adults, older adults |
Type of indication/diagnosis | Mental and behavioural; neurological; others; otorhinolaryngological; pulmonary | Cardiovascular; mental and behavioural; neurological; others; otorhinolaryngological; pulmonary | Generalized or unspecified |
Type of treatment and management | Non-pharmacological interventions ; Pharmacological interventions | Care models; Non-pharmacological interventions | Care models |
Management | |||
Self-management support | NR | Yes | Yes |
Referral to specialist(s) | Yes | Yes | Yes |
Referral to post-COVID clinic | NR | NR | NR |
ICU = intensive care unit; NR = not reported.
aSummarizes literature on children and adults, but unclear if guidance specific to 12+ weeks applies to children.
Systematic and Rapid Reviews
We included 5 rapid reviews5,41-44 and 7 systematic reviews.28,45-50 Three of the rapid reviews were produced by governmental agencies to study the impact of vaccination on post–COVID-19 condition.42-44 There was an overlap in the primary studies included in the 3 rapid reviews, particularly those from France, the UK, and the US.42-44 Another rapid review led by the Ontario COVID-19 Science Advisory Table reviewed the evidence related to post–COVID-19 condition and concluded that “there was no known treatments specifically for the post COVID-19 condition” (p. 20).5 This review also summarized the care models reviewed in a CADTH report16 and 2 clinical trials.5 The other rapid review focused on vulnerable populations and summarized the strategies recommended by guidelines or studied in trials.41 Self-management and post–COVID-19 care models were treatment and management strategies summarized in this review.41
Two of the systematic reviews were authored by governmental agencies28,49 that did not specify the types of indication/diagnosis in patients with post–COVID-19 condition. One reviewed pharmacological and non-pharmacological interventions for post–COVID-19 condition49 and 1 reviewed care models for post–COVID-19 condition.28 The other 2 were authored by researchers to review dental care48 and care models for post–COVID-19 condition.47 Two systematic reviews evaluated the effects of vaccination on existing post–COVID-19 condition and included different numbers of primary studies.46,50 The other systematic review screened the interventions on post–COVID-19 condition evaluated in trials and identified 2 trials, 1 of which studied olfactory training for anosmia/hyposmia and the other studied aromatherapy for fatigue.45
A summary of rapid reviews and systematic reviews are available in Table 8.
Table 8: Summary of Included Rapid and Systematic Reviews
First author’s surname, country | Year published | Countries of primary studies | Population | Severity of acute illness | Type of indication or diagnosis | Treatment and management | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
Asymptomatic | Symptomatic, not hospitalized | Hospitalized | ICU | Pharmacological interventions | Non-pharmacological interventions | Care model | |||||
Rapid reviews | |||||||||||
PHAC Emerging Science Group,42 Canada | 2022 | France, Germany, Hungary, Indonesia, Italy, Switzerland, UK, and US | Adults | NR | NR | NR | NR | Generalized or unspecified symptoms | Yes (vaccine) | NR | NR |
Public Health Ontario,43 Canada | 2022 | France, Switzerland, UK, and US | Children, adults | NR | NR | NR | NR | Generalized or unspecified symptoms | Yes (vaccine) | NR | NR |
Quinn,5 Canada | 2022 | UK and not reported in 1 included review | Adults, and unspecified in 1 trial | NR | NR | NR | NR | Generalized or unspecified symptoms | “no known treatments specifically for the post COVID-19 condition” (p. 20)5 | “no known treatment specifically for the post COVID-19 condition” (p. 20)5 | Primary care provider model, specialized post–COVID-19 condition clinic model, and hybrid care model in 1 review; usual care pathway in 1 trial; post–COVID-19 condition clinics in 1 trial |
UK Health Security Agency,44 UK | 2022 | France, UK, and US | Adults | Yes | Yes | Yes | NR | Generalized or unspecified symptoms | Yes (vaccine) | Yes | NR |
Warkentin,41 Canada | 2022 | UK | NR | NR | NR | NR | NR | Generalized or unspecified symptoms | NR | Yes | Yes |
Systematic reviews | |||||||||||
Gao,50 China | 2022 | Italy, Morocco, the Netherlands, Spain, Switzerland, UK, and US | Children, adults, and older adults | NR | NR | NR | NR | Generalized or unspecified | Yes (vaccine) | NR | NR |
Institut national d’excellence en santé et en services sociaux (INESSS),28 Canada | 2022 | Australia, Belgium, Canada, Ireland, Italy, the Netherlands, Spain, Switzerland, UK, US, other countries unspecified | Children, adults | Yes | Yes | Yes | NR | Generalized or unspecified symptoms | NR | NR | Yes |
Ngeow,48 Malaysia | 2022 | NR | Children, adults | NR | NR | NR | NR | Patients with dental condition | NR | NR | Yes |
Notarte,46 US | 2022 | France, India, Israel, Italy, Japan, Russia, South Africa, Switzerland, UK, US | Adults, older adults | NR | NR | NR | NR | Cardiovascular; dermatological; fatigue; generalized or unspecified; gastrointestinal; hematological; mental and behavioural; neurological; others; otorhinolaryngological; renal; pulmonary; rheumatological | Yes (vaccine) | NR | NR |
Veronese,45 Italy | 2022 | Italy and US | Adults | NR | NR | NR | NR | Fatigue, anosmia/hyposmia | NR | Yes | NR |
Agency for Health Technology Assessment and Assessment of Social Services (SBU),49 Sweden | 2021 | NR | NR | NR | NR | NR | NR | Generalized or unspecified symptoms | Yes | Yes | NR |
Décary,47 Canada | 2021 | Canada, China, France, Hungry, Ireland, UK, and US | Adults | NR | NR | NR | NR | Generalized or unspecified symptoms | NR | NR | Yes |
ICU = intensive care unit; NR = not reported; PHAC = Public Health Agency of Canada.
aCADTH reported the country/countries of included primary studies if provided in the review. If CADTH were unable to find these data, CADTH reported the country of the first author.
Evidence Gap Mapping
A summary of areas where reports were identified, along with areas where reports were not identified, is presented in Table 9. Thus far, much of the published literature on post–COVID-19 condition treatment and management is limited to case reports or series and has focused on certain types of indication/diagnosis, with some studies having a long follow-up time — for example, a year after infection or hospital discharge.51,52 Socioeconomic status and characteristics that could help identify populations facing barriers have not been widely reported, and few studies focused on those who might be at greater risk of infection or experiencing post–COVID-19 condition. We included 3 rapid reviews on vaccination for post–COVID-19 symptoms,42-44 which may be a potential area for systematic reviews and meta-analyses. We found 9 or more primary studies evaluating pharmacological interventions for cardiovascular, neurological, and pulmonary conditions or non-pharmacological interventions for pulmonary condition. We identified fewer than 9 published primary studies on most types of indication/diagnosis, such as fatigue, generalized symptoms, or mental and behavioural disorders. Although fatigue and mental symptoms were the symptoms that patients with post–COVID-19 condition most often reported,53 these symptoms were not investigated as much as pulmonary or cardiovascular conditions. Table 10 shows that certain populations, including children and those requiring treatment in the communities, were reported less often in the included studies. In the primary studies that were not case reports/series, being asymptomatic during initial infection, vaccination against COVID-19 before infection, and long-term follow-up were reported less often. In terms of study design, we found only 1 qualitative study and no economic evaluations. When compared to the symptoms that patients with post–COVID-19 condition often reported,53 we noticed that the symptoms that were frequently reported are not the conditions that were commonly studied in the included studies.
Table 9: Summary of Evidence by Treatment and Management, Indication or Diagnosis, and Characteristics
State of evidence map | Pharmacological interventions | Non-pharmacological interventions (e.g., surgery or procedures) | Care models |
|---|---|---|---|
Areas where rapid or systematic reviews were identified | |||
Areas where several published primary studies (≥ 9) were identified, a majority of which were case reports/series |
|
|
|
Areas where some (3 to 8) primary studies were identified, a majority of which were case reports or series |
|
|
|
Areas with gaps in evidence (< 3 published primary studies, no systematic reviews) |
|
|
|
Table 10: Summary of Evidence Gaps by Characteristics, Study Design, and Prevalent Symptoms
Population and study characteristics | Specific areas with notable gaps |
|---|---|
Population characteristics |
|
Study design |
|
Conditions most often reported by patients with post–COVID-19 condition53 |
|
Patient Engagement
We spoke with 3 patient contributors in a 1-hour call. The patient contributors discussed several themes, some of which were directly related to the treatment and management of post–COVID-19 condition. These themes include accessibility of diagnostics and treatments (i.e., limited specialists, long wait times, access in rural and remote communities), the variability of symptoms and diagnoses (i.e., postural orthostatic tachycardia syndrome, post-exertion malaise, cardiac and lung damage), treatments of interest, and the need for knowledge dissemination and clinician education. Patients also highlighted the need to distinguish between the subtypes of post–COVID-19 condition (i.e., persistent symptoms like a cough or fatigue that eventually resolve), and chronic health issues that arise from post–COVID-19 condition (such as cardiac and lung damage, postural orthostatic tachycardia syndrome, cognitive symptoms, ongoing fevers, gastrointestinal symptoms).
Discussion
In this review we identified evidence related to treatment and management of post–COVID-19 condition, described their characteristics, and categorized these based on the classification of indication/diagnosis recommended by an HTA agency, INESSS.27 We summarized the evidence gaps using this classification, population characteristics, study characteristics, and study design.
Compared to the symptoms and conditions that were most often reported, such as fatigue, sleep disorder, anxiety, depression, and shortness of breath,53 there might be gaps between the conditions that were most studied and those most experienced by patients. We located only 1 qualitative study and no economic evaluations. Similar to the previous review,7 there were some notable evidence gaps and other areas with relatively more studies. There were few studies on certain types of indications or diagnoses developed after COVID-19 infection, such as gastrointestinal, renal, rheumatological, and mental and behavioural conditions. In addition, the majority of identified primary studies were case studies or case series, with relatively few (N = 11) primary interventional studies, approximately half of which (N = 5) were randomized controlled trials (RCTs). Thus, there is a lack of high-quality evidence regarding what treatments and management methods are effective for treating post–COVID-19 condition. There were also few guidelines providing recommendations for individuals living with post–COVID-19 condition according to the WHO definition (12 or more weeks after initial infection). Some of the identified guidelines provide guidance for people who still have symptoms at 4 or fewer weeks after initial infection that might be applicable to those with symptoms 12 months after initial infection,39,54 and it is unclear whether these recommendations will be revised for people who are experiencing symptoms at 12 or more weeks.
We identified few published studies for certain population groups. Relatively few studies included children, and there may be important differences between adults and children (e.g., while many studies report shortness of breath as a common post–COVID-19 symptom, the National Institute for Health and Care Excellence (NICE) guidelines suggest that it is less common in children and older adults).55 Thus, even if trials indicate treatments to be effective in adults, it is not known if these treatments can be used to treat children or adolescents with similar effects. We also did not identify many studies that included people who had been asymptomatic during the acute illness phase. This may be due in part to lack of COVID-19 testing, particularly earlier in the pandemic when access may have been restricted or otherwise difficult to access for people who were asymptomatic.
It is unclear whether the clinical responses of post–COVID-19 condition to treatments may differ between people who had milder acute illness (i.e., were not hospitalized) and people who had more severe acute illness and were hospitalized. Thus, the lack of published evidence focused on people who had milder acute illness, particularly people who had been asymptomatic, is an important evidence gap. There are also limited studies assessing post–COVID-19 condition in breakthrough infections (i.e., for individuals who had been vaccinated, then became infected with COVID-19). This may be an important consideration with the spread of the Omicron variant, which has become dominant in Canada and many other countries, as vaccines are less effective at preventing infection from this variant, and breakthrough infections have become increasingly common.56 There was also a lack of information regarding the SARS-CoV-2 variant being assessed in studies.57,58 Determining whether different variants have different long-term effects may also be important to plan for diagnosis, treatment, or management. Finally, future guidelines or updates to existing guidelines may be needed to ensure clear and consistent recommendations for post–COVID-19 condition that incorporate the findings of emerging evidence, so that health care providers can provide adequate care.
We did not retrieve data on whether participants were located in rural or remote areas because this often was not reported in most studies. There were also few studies that provided insight into treatments for post–COVID-19 condition in populations that might be at high risk (e.g., health care workers) or who might experience disadvantage. It would be useful for future studies to report on these characteristics to inform considerations about equity in distribution of post–COVID-19 condition, access to treatment, and/or treatment outcomes.
We engaged with 3 patients with post–COVID-19 condition and discussed the key results with them. The patients described their experiences and major themes related to their condition. The patient collaborators were highly engaged in the consultation, sharing their perspectives, priorities, and lived experiences. The patients expressed interest in seeing more treatment and management options for post–COVID-19 condition.
Limitations
This review has the strength of having a wider scope than other reviews.8,59 This enabled us to produce a comprehensive evidence map for the treatment and management of post–COVID-19 condition. However, there are several limitations to this review. We explicitly excluded studies focused solely on rehabilitation to avoid overlap with an existing living Cochrane review on the topic. This review was limited to studies available in English or French; as this limitation was applied at the level of the search, it is not clear how many studies may have been excluded for this reason, and findings could differ slightly had other languages been included. Due to the wide breadth of topics covered and time constraints, we did not search reference lists of included studies or contact experts to supplement the search; as a result, there is a possibility that some relevant studies were missed, but it is unlikely that this would have a large impact on the overall high-level findings. Not all primary studies adopted the definition of post–COVID-19 condition by WHO, and studies adopting broader definitions (such as that of the CDC) would not have been included. Additionally, studies initiated treatment or management strategies for post–COVID-19 condition at different time points after COVID-19 infection. These studies were excluded from our scoping review, in attempt to include the most relevant information related to post–COVID-19 condition. This also made it difficult to compare treatment and management strategies, because patients had different recovery time after initial infection. In fact, the severity classification recommended by WHO was rarely reported.
Conclusions
In conclusion, as of October 2022, although a large volume of research has been conducted, this is limited primarily to case reports/series. The interventions and classifications represented by the largest number of primary studies were pharmacological interventions for cardiovascular symptoms or conditions, and non-pharmacological interventions for pulmonary conditions. We located 5 rapid reviews and 7 systematic reviews on pharmacological and non-pharmacological interventions, and care models. We found 4 Canadian guidelines and 3 guidelines from other countries.
Some areas where gaps (little published evidence) were identified include treatment and management for certain types of symptoms (e.g., dermatological, hematological, renal, and rheumatological), pediatric populations, and people who were asymptomatic during initial infection. We did not identify any economic evaluations, and identified only 2 qualitative studies. More research in these areas may help to close some of the current evidence gaps.
References
1.Statement from the Chief Public Health Officer of Canada on July 7, 2021. Ottawa (ON): Public Health Agency of Canada; 2021: https://www.canada.ca/en/public-health/news/2021/07/statement-from-the-chief-public-health-officer-of-canada-on-july-7-2021.html. Accessed 2021 Sep 1.
2.Rajan S, Khunti K, Alwan N, et al. In the wake of the pandemic: preparing for long COVID. (European Observatory Policy Brief 39). Copenhagen (Denmark): European Observatory on Health Systems and Policies; WHO; 2021: https://www.who.int/europe/publications/i/item/in-the-wake-of-the-pandemic-preparing-for-long-covid. Accessed 2021 Sep 1.
3.World Health Organization. A clinical case definition of post COVID-19 condition by a Delphi consensus. Geneva (Switzerland): World Health Organization; 2021: https://apps.who.int/iris/rest/bitstreams/1376291/retrieve. Accessed 2022 Dec 9.
4.Long COVID or post-COVID conditions. Atlanta (GA): Centers for Disease Control and Prevention (CDC); 2022: https://www.cdc.gov/coronavirus/2019-ncov/long-term-effects/index.html. Accessed 2022 Nov 29.
5.Quinn KL, Katz GM, Bobos P, et al. Understanding the post COVID-19 condition (long COVID) in adults and the expected burden for Ontario. Science Briefs of the Ontario COVID-19 Science Advisory Table. 2022;3(65). 10.47326/ocsat.2022.03.65.1.0. Accessed 2022 Dec 9.
6.Long-term symptoms in Canadian adults who tested positive for COVID-19 or suspected an infection, January 2020 to August 2022. Ottawa (ON): Statistics Canada; 2022: https://www150.statcan.gc.ca/n1/daily-quotidien/221017/dq221017b-eng.htm. Accessed 2022 Oct 21.
7.Chao Y-S, McGill SC, Gates M. Clinical classification and interventions for post-COVID-19 condition: a scoping review. Can J Health Technol. 2022 May;2(5). https://www.cadth.ca/post-covid-19-condition-scoping-review. Accessed 2022 Dec 9.
8.Chee YJ, Fan BE, Young BE, Dalan R, Lye DC. Clinical trials on the pharmacological treatment of long COVID: a systematic review. J Med Virol. 2022. PubMed
9.Ledford H. Long-COVID treatments: why the world is still waiting. Nature. 2022;608(7922):258-260. PubMed
10.Ghany MG, Strader DB, Thomas DL, Seeff LB. Diagnosis, management, and treatment of hepatitis C: an update. Hepatology (Baltimore, Md). 2009;49(4):1335. PubMed
11.Ceravolo MG, De Sire A, Andrenelli E, Negrini F, Negrini S. Systematic rapid” living” review on rehabilitation needs due to COVID-19: update to March 31st, 2020. Eur J Phys Rehabil Med. 2020;56(3):347-353. PubMed
12.REH-COVER - Rapid living Systematic Reviews. London (UK): Cochrane Rehabilitation; 2022: https://rehabilitation.cochrane.org/covid-19/reh-cover-rapid-living-systematic-reviews. Accessed 2022 Nov 11.
13.Post–COVID-19 condition treatment and management: protocol for a rapid living scoping review. (CADTH health technology review). Ottawa (ON): CADTH 2022: https://www.cadth.ca/sites/default/files/pdf/htis/2022/RE0040-001-Protocol.pdf. Accessed 2022 Dec 9.
14.Tricco AC, Lillie E, Zarin W, et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): checklist and explanation. Ann Intern Med. 2018;169(7):467-473. PubMed
15.Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. PubMed
16.Vu T, McGill SC. CADTH horizon scan: an overview of post-COVID-19 condition (long COVID). Can J Health Technol. 2021 Sep;1(9). https://www.cadth.ca/overview-post-covid-19-condition-long-covid. Accessed 2022 Dec 9.
17.Andrenelli E, Negrini F, De Sire A, et al. Rehabilitation and COVID-19: a rapid living systematic review 2020 by Cochrane Rehabilitation Field. Update as of September 30th, 2020. Eur J Phys Rehabil Med. 2020;56(6):846-852. PubMed
18.World Health Organization. Rehabilitation. 2021; https://www.who.int/news-room/fact-sheets/detail/rehabilitation. Accessed 2022 Dec 9.
19.Ceravolo MG, Sire Ad, Andrenelli E, Negrini F, Negrini S. Supplementary digital material 1. Eur J Phys Rehabil Med. 2020;56(3). PubMed
20.Krnic Martinic M, Pieper D, Glatt A, Puljak L. Definition of a systematic review used in overviews of systematic reviews, meta-epidemiological studies and textbooks. BMC Med Res Methodol. 2019;19(1):203. PubMed
21.McGowan J, Sampson M, Salzwedel DM, Cogo E, Foerster V, Lefebvre C. PRESS Peer Review of Electronic Search Strategies: 2015 guideline statement. J Clin Epidemiol. 2016;75:40-46. PubMed
22.CADTH COVID-19 search strings. Ottawa (ON): CADTH; 2019: https://covid.cadth.ca/literature-searching-tools/cadth-covid-19-search-strings/. Accessed 2022 Dec 9.
23.Grey matters: a tool for searching health-related grey literature. Ottawa (ON): CADTH; 2022: https://greymatters.cadth.ca/. Accessed 2022 Dec 9.
24.CADTH COVID-19 grey literature resources. Ottawa (ON): CADTH; 2019: https://covid.cadth.ca/literature-searching-tools/cadth-covid-19-grey-literature-resources/. Accessed 2022 Dec 9.
25.DistillerSR. Version 2.35. Ottawa (ON): DistillerSR Inc.; 2022: https://www.evidencepartners.com. Accessed 2022 Dec 9.
26.O'Neill J, Tabish H, Welch V, et al. Applying an equity lens to interventions: using PROGRESS ensures consideration of socially stratifying factors to illuminate inequities in health. J Clin Epidemiol. 2014;67(1):56-64. PubMed
27.Management support tool - post-COVID-19 conditions. Montreal (QC): Institut National d'Excellence En Santé et Services Sociaux (INESSS); 2022 Jun: https://www.inesss.qc.ca/fileadmin/doc/INESSS/COVID-19/2022_Outil_Covid_Affections_postCov19_AN-VF_01.pdf. Accessed 2022 Dec 9.
28.Organisation des soins et des services pour la prévention et la prise en charge des affections post-COVID-19. État des connaissances rédigé par Geneviève Morrow. Québec (QC): Institut national d’excellence en santé et en services sociaux (INESSS); 2022: https://www.inesss.qc.ca/fileadmin/doc/INESSS/COVID-19/INESSS_Affections_soins_post_COVID_EC.pdf. Accessed 2022 Aug 12.
29.Aromataris E, Munn Z, editors. JBI manual for evidence synthesis. Adelaide (Australia): JBI; 2020: 10.46658/JBIMES-20-01. Accessed 2022 Dec 9.
30.Yan Z, Yang M, Lai C-L. Long COVID-19 syndrome: a comprehensive review of its effect on various organ systems and recommendation on rehabilitation plans. Biomedicines. 2021;9(8):966. PubMed
31.Crook H, Raza S, Nowell J, Young M, Edison P. Long COVID—mechanisms, risk factors, and management. BMJ. 2021;374:n1648. PubMed
32.Staniszewska S, Brett J, Simera I, et al. GRIPP2 reporting checklists: tools to improve reporting of patient and public involvement in research. BMJ. 2017;358:j3453. PubMed
33.Government of Canada. COVID-19 daily epidemiology update. 2022 Sep 16; https://health-infobase.canada.ca/covid-19/epidemiological-summary-covid-19-cases.html. Accessed 2022 Sep 21.
34.Rubin R. As their numbers grow, COVID-19 “Long Haulers” stump experts. JAMA. 2020;324(14):1381-1383. PubMed
35.What is known about the optimal management for patients with post-COVID conditions or complications occurring after acute COVID-19 infection? (COVID-19 Scientific Advisory Group Rapid Evidence Report). Edmonton: Alberta Health Services; 2021: https://www.albertahealthservices.ca/assets/info/ppih/if-ppih-covid-19-sag-rapid-review-management-post-covid-conditions.pdf. Accessed 2021 Dec 1.
36.BC Provincial Health Services Authority. Provincial post-COVID-19 recovery care pathway. Vancouver (BC): PHSA; [2022]: http://www.phsa.ca/health-info-site/Documents/post_covid-19_Provincial_Recovery_Pathway.pdf. Accessed 2022 Apr 21.
37.Post COVID-19 condition: guidance for primary care. Assessment and management of the post-COVID-19 condition. Toronto: Ontario Health; 2021: https://www.ontariohealth.ca/sites/ontariohealth/files/2021-12/PostCovidConditionsClinicalGuidance_EN.pdf. Accessed 2022 Apr 21.
38.Yelin D, Moschopoulos CD, Margalit I, et al. ESCMID rapid guidelines for assessment and management of long COVID. Clin Microbiol Infect. 2022;28(7):955-972. PubMed
39.Siso-Almirall A, Brito-Zeron P, Conangla Ferrin L, et al. Long COVID-19: Proposed primary care clinical guidelines for diagnosis and disease management. Int J Environ Res Public Health. 2021;18(8):20. PubMed
40.National Institute for Health and Care Excellence (NICE), Scottish Intercollegiate Guidelines Network (SIGN), Royal College of General Practitioners (RCGP). COVID-19 rapid guideline: managing the longterm effects of COVID-19. London (UK): NICE; 2022: https://www.nice.org.uk/guidance/ng188/resources/covid19-rapid-guideline-managing-the-longterm-effects-of-covid19-pdf-51035515742. Accessed 2022 Mar 22.
41.Warkentin L, Guo B, Tjosvold L. Long COVID and vulnerable populations. Edmonton (AB): Institute of Health Economics; 2021 Mar: https://www.ihe.ca/advanced-search/long-covid-and-vulnerable-populations. Accessed 2022 Nov 14.
42.PHAC Emerging Science Group. Evidence Brief on the associations and safety of COVID-19 vaccination and post COVID-19 condition: update 1. Ottawa (ON): Public Health Agency of Canada; 2022: https://www.nccmt.ca/covid-19/covid-19-evidence-reviews/528 (available upon request). Accessed 2022 Dec 9.
43.Impact of vaccination on post-acute COVID-19 syndrome (PACS) – what we know so far. Synthesis. Toronto (ON): Public Health Ontario,; 2022: https://www.publichealthontario.ca/-/media/Documents/nCoV/COVID-WWKSF/2022/04/impact-vaccination-post-acute-covid-19-syndrome.pdf?sc_lang=en. Accessed 2022 Aug 12.
44.Harrison S, Walters B, Simmons Z, Cook M, Clark R. The effectiveness of vaccination against long COVID: a rapid evidence briefing. London (UK): UK Health Security Agency; 2022: https://www.icpcovid.com/sites/default/files/2022-02/Ep%20241-9%20UK%20Health%20Security%20Agency%20The%20effectiveness%20of%20vaccination%20against%20long%20COVID%20Feb%202022.pdf. Accessed 2022 Aug 12.
45.Veronese N, Bonica R, Cotugno S, et al. Interventions for improving long COVID-19 symptomatology: a systematic review. Viruses. 2022;14(9). PubMed
46.Notarte KI, Catahay JA, Velasco JV, et al. Impact of COVID-19 vaccination on the risk of developing long-COVID and on existing long-COVID symptoms: A systematic review. EClinicalMedicine. 2022;53:101624. PubMed
47.Décary S, Dugas M, Stefan T, et al. Care models for Long COVID: a living systematic review. First update – December 2021. Toronto (ON): SPOR Evidence Alliance, COVID-END Network; 2021: https://sporevidencealliance.ca/wp-content/uploads/2021/12/Care-Models-for-Long-COVID_Update_2021.12.04.pdf. Accessed 2022 Mar 22.
48.Ngeow WC, Tang L, Ho JY, et al. The provision of dental care to COVID-19 survivors: a concise review. Int Dent J. 2022;72(4):421-435. PubMed
49.Agency for Health Technology Assessment and Assessment of Social Services (SBU). Post COVID-19 – effective treatment and rehabilitation, living review. Post COVID-19 – effective treatment and rehabilitation. 2021. Last updated 2022 Jun 15; https://www.sbu.se/en/publications/sbu-bereder/post-covid-19--effective-treatment-and-rehabilitation/. Accessed 2022 Aug 12.
50.Gao P, Liu J, Liu M. Effect of COVID-19 vaccines on reducing the risk of long COVID in the real world: A systematic review and meta-analysis. Int J Environ Res Public Health. 2022;19(19):12422. PubMed
51.Wynberg E, Han AX, Boyd A, et al. The effect of SARS-CoV-2 vaccination on post-acute sequelae of COVID-19 (PASC): A prospective cohort study. Vaccine. 2022;40(32):4424-4431. PubMed
52.Kute VB, Ray DS, Aziz F, et al. Management strategies and outcomes in renal transplant recipients recovering from COVID-19: A retrospective, multicentre, cohort study. EClinicalMedicine. 2022;46:101359. PubMed
53.Razak F, Katz GM, Cheung AM, et al. Understanding the Post COVID-19 Condition (long COVID) and the expected burden for Ontario. Science Briefs of the Ontario COVID-19 Science Advisory Table. 2021;2(44). https://covid19-sciencetable.ca/wp-content/uploads/2021/09/Understanding-the-Post-COVID-19-Condition-Long-COVID-and-the-Expected-Burden-for-Ontario_published_20210914-3.pdf. Accessed 2022 Dec 9.
54.Whiteson JH, Azola A, Barry JT, et al. Multi-disciplinary collaborative consensus guidance statement on the assessment and treatment of cardiovascular complications in patients with post-acute sequelae of SARS-CoV-2 infection (PASC). Pm R. 2022;14(7):855-878. PubMed
55.National Institute for Health and Care Excellence. COVID-19 rapid guideline: managing the long-term effects of COVID-19. (NICE guideline NG188). 2020; https://www.nice.org.uk/guidance/ng188. Accessed 2022 Apr 21.
56.Government of Canada. COVID-19 daily epidemiology update. 2022 Mar 9; https://health-infobase.canada.ca/covid-19/epidemiological-summary-covid-19-cases.html. Accessed 2022 Mar 1.
57.Penetra SLS, da Silva MFB, Resende P, et al. Post-acute COVID-19 syndrome after reinfection and vaccine breakthrough by the SARS-CoV-2 Gamma variant in Brazil. Int J Infect Dis. 2022;114:58-61. PubMed
58.Guimaraes RRL, Aranda CS, Schmid LS, et al. PASC, reinfections and variants: COVID-19 in patients with inborn errors of immunity in Sao Paulo-Brazil [conference abstract]. Allergy. 2021;76(SUPPL 110):494.
59.Carson E, Hemenway AN. A scoping review of pharmacological management of postacute sequelae of severe acute respiratory syndrome coronavirus 2 infection in 2021. Am J Ther. 2022;29(3):e305-e321. PubMed
Appendix 1: Literature Search Methods
Note that this appendix has not been copy-edited.
Note: For the literature search strategy for the previously published scoping review, please refer to the appendices of that review:
Chao Y-S et al. Clinical Classification and Interventions for Post–COVID-19 Condition: A Scoping Review. Canadian Journal of Health Technologies. 2022;2(5).7
Overview
Interface: Ovid
Databases:
MEDLINE All (1946-present) via Ovid
Embase (1974-present) via Ovid
Note: Subject headings and search fields have been customized for each database. Duplicates were removed using Ovid deduplication for multi-file searches, followed by manual deduplication in Endnote.
Date of search current report: July 21, 2022.
Date of last alert incorporated: October 21, 2022.
Alerts: Database search updates every 3 months until completion of the living review.
Search filters applied: No filters were applied to limit the retrieval by study type. Comments, newspaper articles, editorials, and letters were removed.
Limits:
Publication date limit: articles published or added to the databases from January 30, 2022 – present
Humans
Languages: English or French
Syntax | Description | |
|---|---|---|
/ | At the end of a phrase, searches the phrase as a subject heading | |
MeSH | Medical Subject Heading | |
.fs | Floating subheading | |
exp | Explode a subject heading | |
* | Before a word, indicates that the marked subject heading is a primary topic; or, after a word, a truncation symbol (wildcard) to retrieve plurals or varying endings | |
# | Truncation symbol for one character | |
? | Truncation symbol for one or no characters only | |
adj# | Requires terms to be adjacent to each other within # number of words (in any order) | |
.ti | Title | |
.ot | Original title | |
.ab | Abstract | |
.hw | Heading word; usually includes subject headings and controlled vocabulary | |
.kf | Author keyword heading word (MEDLINE, Embase) | |
.nm | Name of substance, including CAS and other registry numbers and chemical names (MEDLINE) | |
.ox | Organism supplementary concept word for emerging virus and other organism terms (MEDLINE) | |
.rx | Rare disease supplementary concept for emerging rare disease terms (MEDLINE) | |
.px | Protocol supplementary concept (MEDLINE) | |
freq = # | requires that the term(s) occur # number of times or more within the field(s) searched | |
.pt | Publication type | |
.ez | Medline Entrez date (YYYYMMDD with * for any truncation) | |
.da | Medline entry date (YYYYMMDD with * for any truncation) | |
.dt | Medline creation date (YYYYMMDD with * for any truncation) | |
Limit to dc | Embase date created. Format as: dc = YYYYMMDD-YYYYMMDD | |
medall | Ovid database code: MEDLINE All, 1946 to present, updated daily | |
oemezd | Ovid database code; Embase, 1974 to present, updated daily | |
Ovid Multi-Database Strategy – MEDLINE and Embase
MEDLINE post–COVID-19 treatment or management terms by themselves
1. ((post covid* or post coronavirus* or long covid* or long coronavirus*) adj2 (treatment* or management)).ab.
MEDLINE main post–COVID-19 terms
2. (long COVID* or long coronavirus* or longCOVID* or longcoronavirus*).ti,ab,kf.
3. (sequela* adj5 (COVID* or coronavirus* or corona virus* or SARS-COV-2 or SARS-COV2 or SARSCOV-2 or SARSCOV2)).ti,ab,kf.
4. ((post or chronic or long term or longterm) adj3 (COVID* or coronavirus* or corona virus* or SARS-COV-2 or SARS-COV2 or SARSCOV-2 or SARSCOV2) adj4 (condition* or sequela* or syndrome* or subsyndrome* or clinical syndrome* or disorder* or symptom* or outcome* or clinical outcome* or function* or followup or follow-up or subtyp* or sub-typ* or phenotyp* or complication* or survivor*)).ti,ab,kf.
5. ((post acute or postacute or late complication*) adj3 (COVID* or coronavirus* or corona virus* or SARS-COV-2 or SARS-COV2 or SARSCOV-2 or SARSCOV2)).ti,ab,kf.
6. PASC.ti,kf.
7. (post-COVID* adj fatigue).ti,ab,kf.
8. or/2-7
MEDLINE COVID-19 set
9. COVID-19/ or SARS-CoV-2/
10. (COVID* or coronavirus* or corona virus* or 2019nCoV or 19nCoV or COVID19* or COVID or SARS-COV-2 or SARSCOV-2 or SARS-COV2 or SARSCOV2 or SARS coronavirus 2 or Severe Acute Respiratory Syndrome Coronavirus 2 or Severe Acute Respiratory Syndrome Corona Virus 2).ti,ab,kf,nm,ot,ox,rx,px.
11. 9 or 10
MEDLINE additional post–COVID-19 terms and subtypes, to combine with COVID-19 set
12. (((post acute or postacute or sub-acute or subacute or chronic or long or longterm or late) adj sequela*) or PASC).ti,ab,kf.
13. (long haul* or longhaul*).ti,ab,kf.
14. ((persist* or long* or residual or prolonged) adj8 ((olfactory or chemosensor*) adj (disorder* or dysfunction*))).ti,ab,kf.
15. (post* or chronic* or long or longterm or sequela*).ti,ab,kf.
16. (PFS or (pulmonary adj3 fibro*) or (lung adj3 fibro*) or fatigue syndrome? or myalgic encephalomyelitis or ME-CFS or ME?CFS or (postural adj3 tachycardia*) or POTS or MIS-C or MIS-A or PIMS or PIMSTS or PIMS-TS).ti,ab,kf.
17. ((multisystem* or multi-system*) adj3 (inflamm* or hyperinflamm*)).ti,ab,kf.
18. 15 and (16 or 17)
19. or/12-14,18
20. 11 and 19
MEDLINE treatment or management set
21. Therapeutics/
22. (dh or dt or pd or su).fs. [diet therapy, drug therapy, pharmacology superheading, surgery]
23. (treatment* or therap* or management or managing or drug? or medication? or pharmacolog* or pharmaceutic* or non-drug? or nonpharmacolog* or non-pharmacolog* or biologic or biologics or diet* or supplement* or vitamin* or treat or treated or treating or placebo* or off-label*).ti,kf.
24. (treatment* or therap* or management or managing or drug? or medication? or pharmacolog* or pharmaceutic* or non-drug? or nonpharmacolog* or non-pharmacolog* or biologic or biologics or diet* or supplement* or vitamin* or treat or treated or treating or placebo* or off-label*).ab. /freq=3
25. ((care or treatment or healthcare) adj2 (model* or path* or plan*)).ti,ab,kf.
26. ((interdisciplin* or inter-disciplin* or cross-disciplin* or multidiscipline* or multi-disciplin* or inter-disciplin*) adj3 (team* or clinic? or centre? or center? or model* or program*)).ti,ab,kf.
27. (recovery adj2 (team* or clinic? or centre? or center? or model* or program*)).ti,ab,kf.
28. (care adj2 (coordinat* or co-ordinat*)).ti,ab,kf.
29. (patient-centred care or patient-centered care or case manag*).ti,ab,kf.
30. exp Drug Therapy/ or exp Biological Therapy/ or exp “Nebulizers and Vaporizers”/
31. (infusion* or inhaler* or nasal spray* or nebulize*).ti,ab,kf.
32. vaccin*.ti,kf.
33. vaccin*.ab. /freq=3
34. (anticoagulants or (anticoagulant* adj2 (therap* or oral* or intravenous* or IV)) or apixaban* or clopidogrel* or heparin*).ti,ab,kf.
35. (aceclofenac* or antibiotic* or antidepressant* or antihistamine* or ((antiviral* or anti-viral*) adj2 (agent* or oral*)) or atenolol* or atorvastatin*).ti,ab,kf.
36. (bamlanivimab* or beta blocker* or casirivimab* or cilgavimab* or convalescent plasma* or corticosteroid* or dexamethasone* or enol-oxaloacetate* or enoxaparin* or etanercept* or Evusheld* or fludrocortisone* or hydroxychloroquine* or intravenous immunoglobulin* or immune-modulator* or immunomodulator* or imdevimab* or meglucocorticoid* or glucocorticosteroid* or indomethacin* or ivermectin*).ti,ab,kf.
37. (levocetirizine* or megestrol* or methylnicotinamide* or methylprednisolone* or monoclonal antibod* or montelukast* or N115 or N-115 or Nacetylcysteine* or sodium pyruvate* or naltrexone* or nicotinamide* or nintedanib* or nirmatrelvir* or Paxlovid* or perflubron* or pirfenidone* or prednisolone* propranolol* or remdesivir* or ritonavir* or rituximab*).ti,ab,kf.
38. (S1226 or S-1226 or selective serotonin reuptake inhibitor* or SSRI* or sildenafil* or sotrovimab* or steroid* or sulodexide* or tiazotic acid* or tixagevimab* or thiotriazolin* or vasodilator* or vasopressin* or vortioxetine* or vinpocetine*).ti,ab,kf.
39. (cellular therap* or cell-based therap* or stem cell* or actovegin* or amniotic epithelial cell* or hAEC? or Ampion* or Cerebrolysin* or COVI-MSC* or cSVF or Leronlimab* or MON002 or RSLV-132 or Ruconest*).ti,ab,kf.
40. exp Dietary Supplements/ or exp Nutrition Therapy/ or Medical Marijuana/ or exp Cannabinoids/
41. (dietician? or nutritionist?).ti,ab,kf.
42. (sublingual or oral drops).ti,ab,kf.
43. (antioxidant* or anti-oxidant* or Bioarginina C or L-arginine or CBD or cannabis or Myoquinon or coenzyme Q10 or Niagen or nicotinamide riboside or probiotic* or pro-biotic* or prebiotic* or pre-biotic* or lactobacill* or inulin* or bacteriotherap*).ti,ab,kf.
44. exp Surgical Procedures, Operative/ or Blood Component Removal/ or exp Compression Bandages/ or exp Electric Stimulation Therapy/ or exp Organ Transplantation/ or Neuromuscular Blocking Agents/ or exp Radiofrequency Therapy/
45. (device? or procedure? or technique?).ti,kf.
46. (surgery or surgeries or surgical* or operation).ti,ab,kf.
47. (apheresis or blood washing or blood component removal or plasmapheresis).ti,ab,kf.
48. ((compression or pressure) adj3 (bandage* or garment* or clothing* or socks or stocking? or hose)).ti,ab,kf.
49. ((lung? or organ?) adj2 transplantation).ti,ab,kf.
50. ((electrical or bioelectric or nerve or transcranial or trans-cranial) adj3 (stimulator or stimulation*)).ti,ab,kf.
51. ((monopolar or intervention*) adj3 (radiofrequenc* or radio frequenc*)).ti,ab,kf.
52. (neurophysiological facilitation or neuromodulation or Parasym? or Dolphin* or tDCS or INDIBA).ti,ab,kf.
53. (nerve block* or neuromuscular block* or ganglion block).ti,ab,kf.
54. (hyperbaric oxygen therap* or HBOT or supplemental oxygen or hydrogen therap* or (hydrogen adj3 inhalation) or molecular hydrogen).ti,ab,kf.
55. exp Phototherapy/ or exp Psychiatric Somatic Therapies/ or exp Psychotherapy/ or exp Therapy, Computer-Assisted/ or exp Virtual Reality/
56. (cognitive restructuring or ICBT or CBT or mind-body or meditation* or mentalPlus or hypnotherapy* or hypnosis or phototherapy or psychotherap* or progressive muscle relaxation or breathing retraining or pranayama* or olfactory training).ti,ab,kf.
57. ((cognitive or singing or breathing or breath or breathe or choral or choir or opera or ENO) adj2 (class or classes or instruction* or intervention* or training or program*)).ti,ab,kf.
58. (virtual reality or virtual environment? or virtual technolog* or ((computer* or digital or online or smartphone or virtual or web or webbased) adj3 (game? or gaming or app)) or gamification).ti,ab,kf.
59. exp Alternative Medicine/ or exp Biological Products/ or exp Complementary therapies/ or Osteopathic Medicine/
60. (complementary medicine? or alternative medicine? or traditional medicine?).ti,ab,kf.
61. (acupuncture or acupressure or auriculotherap* or acupoint? or chiropract* or osteopath* or reflexology*).ti,ab,kf.
62. (biofeedback or bio-feedback or sensory feedback).ti,ab,kf.
63. (Ayurved* or Agastya Haritaki or Ashwagandha or CIM-Meg19 or Pippali).ti,ab,kf.
64. (homoeopath* or naturopath*).ti,ab,kf.
65. (ozone or autohemotherapy*).ti,ab,kf.
66. or/21-65
MEDLINE results before limits: post–COVID-19 combined with treatment or management
67. (8 or 20) and 66
68. 1 or 67
69. 68 use medall
Embase post–COVID-19 treatment or management terms by themselves
70. long COVID/dm, dt, th [Disease Management, Drug Therapy, Therapy]
71. ((post covid* or post coronavirus* or long covid* or long coronavirus*) adj2 (treatment* or management)).ab.
72. 70 or 71
Embase main post–COVID-19 terms
73. Long covid/
74. (long COVID* or long coronavirus* or longCOVID* or longcoronavirus*).ti,ab,kf.
75. (sequela* adj5 (COVID* or coronavirus* or corona virus* or SARS-COV-2 or SARS-COV2 or SARSCOV-2 or SARSCOV2)).ti,ab,kf.
76. ((post or chronic or long term or longterm) adj3 (COVID* or coronavirus* or corona virus* or SARS-COV-2 or SARS-COV2 or SARSCOV-2 or SARSCOV2) adj4 (condition* or sequela* or syndrome* or subsyndrome* or clinical syndrome* or disorder* or symptom* or outcome* or clinical outcome* or function* or followup or follow-up or subtyp* or sub-typ* or phenotyp* or complication* or survivor*)).ti,ab,kf.
77. ((postCOVID* or postcoronavirus* or postcorona virus* or postSARS-COV-2 or postSARS-COV2 or postSARSCOV-2 or postSARSCOV2) adj4 (condition* or sequela* or syndrome* or disorder* or symptom* or outcome* or clinical outcome* or function* or followup or follow-up or subtyp* or sub-typ* or phenotyp* or complication* or survivor*)).ti,ab,kf.
78. ((post acute or postacute or late complication*) adj3 (COVID* or coronavirus* or corona virus* or SARS-COV-2 or SARS-COV2 or SARSCOV-2 or SARSCOV2)).ti,ab,kf.
79. PASC.ti,kf.
80. (post-COVID* adj fatigue).ti,ab,kf.
81. or/73-80
Embase COVID-19 set
82. exp Coronavirus disease 2019/
83. exp Severe acute respiratory syndrome coronavirus 2/
84. (COVID* or coronavirus* or corona virus* or 2019nCoV or 19nCoV or COVID19* or COVID or SARS-COV-2 or SARSCOV-2 or SARS-COV2 or SARSCOV2 or SARS coronavirus 2 or Severe Acute Respiratory Syndrome Coronavirus 2 or Severe Acute Respiratory Syndrome Corona Virus 2).ti,ab,kf,nm,ot.
85. or/82-84
Embase additional post–COVID-19 terms and subtypes, to combine with COVID-19 set
86. (((post acute or postacute or sub-acute or subacute or chronic or long or longterm or late) adj sequela*) or PASC).ti,ab,kf.
87. (long haul* or longhaul*).ti,ab,kf.
88. ((persist* or long* or residual or prolonged) adj8 ((olfactory or chemosensor*) adj (disorder* or dysfunction*))).ti,ab,kf.
89. (post* or chronic* or long or longterm or sequela*).ti,ab,kf.
90. (PFS or (pulmonary adj3 fibro*) or (lung adj3 fibro*) or fatigue syndrome? or myalgic encephalomyelitis or ME-CFS or ME?CFS or (postural adj3 tachycardia*) or POTS or MIS-C or MIS-A or PIMS or PIMSTS or PIMS-TS).ti,ab,kf.
91. ((multisystem* or multi-system*) adj3 (inflamm* or hyperinflamm*)).ti,ab,kf.
92. 89 and (90 or 91)
93. or/86-88,92
94. 85 and 93
Embase treatment or management set
95. therapy/
96. (pr or su or th).fs. [Embase pharmaceutics, surgery, therapy]
97. (treatment* or therap* or management or managing or drug? or medication? or pharmacolog* or pharmaceutic* or non-drug? or nonpharmacolog* or non-pharmacolog* or biologic or biologics or diet* or supplement* or vitamin* or treat or treated or treating or placebo* or off-label*).ti,kf.
98. (treatment* or therap* or management or managing or drug? or medication? or pharmacolog* or pharmaceutic* or non-drug? or nonpharmacolog* or non-pharmacolog* or biologic or biologics or diet* or supplement* or vitamin* or treat or treated or treating or placebo* or off-label*).ab. /freq=3
99. ((care or treatment or healthcare) adj2 (model* or path* or plan*)).ti,ab,kf.
100. ((interdisciplin* or inter-disciplin* or cross-disciplin* or multidiscipline* or multi-disciplin* or inter-disciplin*) adj3 (team* or clinic? or centre? or center? or model* or program*)).ti,ab,kf.
101. (recovery adj2 (team* or clinic? or centre? or center? or model* or program*)).ti,ab,kf.
102. (care adj2 (coordinat* or co-ordinat*)).ti,ab,kf.
103. (patient-centred care or patient-centered care or case manag*).ti,ab,kf.
104. exp *biological therapy/ or exp *drug therapy/ or exp *biological product/ or exp inhaler/ or exp nebulizer/
105. (infusion* or inhaler* or nasal spray* or nebulize*).ti,ab,kf.
106. vaccin*.ti,kf.
107. vaccin*.ab. /freq=3
108. (anticoagulants or (anticoagulant* adj2 (therap* or oral* or intravenous* or IV)) or apixaban* or clopidogrel* or heparin*).ti,ab,kf.
109. (aceclofenac* or antibiotic* or antidepressant* or antihistamine* or ((antiviral* or anti-viral*) adj2 (agent* or oral*)) or atenolol* or atorvastatin*).ti,ab,kf.
110. (bamlanivimab* or beta blocker* or casirivimab* or cilgavimab* or convalescent plasma* or corticosteroid* or dexamethasone* or enol-oxaloacetate* or enoxaparin* or etanercept* or Evusheld* or fludrocortisone* or hydroxychloroquine* or intravenous immunoglobulin* or immune-modulator* or immunomodulator* or imdevimab* or meglucocorticoid* or glucocorticosteroid* or indomethacin* or ivermectin*).ti,ab,kf.
111. (levocetirizine* or megestrol* or methylnicotinamide* or methylprednisolone* or monoclonal antibod* or montelukast* or N115 or N-115 or Nacetylcysteine* or sodium pyruvate* or naltrexone* or nicotinamide* or nintedanib* or nirmatrelvir* or Paxlovid* or perflubron* or pirfenidone* or prednisolone* propranolol* or remdesivir* or ritonavir* or rituximab*).ti,ab,kf.
112. (S1226 or S-1226 or selective serotonin reuptake inhibitor* or SSRI* or sildenafil* or sotrovimab* or steroid* or sulodexide* or tiazotic acid* or tixagevimab* or thiotriazolin* or vasodilator* or vasopressin* or vortioxetine* or vinpocetine*).ti,ab,kf.
113. (cellular therap* or cell-based therap* or stem cell* or actovegin* or amniotic epithelial cell* or hAEC? or Ampion* or Cerebrolysin* or COVI-MSC* or cSVF or Leronlimab* or MON002 or RSLV-132 or Ruconest*).ti,ab,kf.
114. exp dietary supplement/ or exp supplementation/ or exp cannabinoid/ or exp probiotic agent/
115. (dietician? or nutritionist?).ti,ab,kf.
116. (sublingual or oral drops).ti,ab,kf.
117. (antioxidant* or anti-oxidant* or Bioarginina C or L-arginine or CBD or cannabis or Myoquinon or coenzyme Q10 or Niagen or nicotinamide riboside or probiotic* or pro-biotic* or prebiotic* or pre-biotic* or lactobacill* or inulin* or bacteriotherap*).ti,ab,kf.
118. medical procedures/ or exp device therapy/ or exp surgery/ or exp compression bandage/ or exp electrotherapy/ or exp neuromuscular blocking agent/ or exp radiofrequency therapy/ or exp phototherapy/ or exp extracorporeal therapy/
119. (device? or procedure? or technique?).ti,kf.
120. (surgery or surgeries or surgical* or operation).ti,ab,kf.
121. (apheresis or blood washing or blood component removal or plasmapheresis).ti,ab,kf.
122. ((compression or pressure) adj3 (bandage* or garment* or clothing* or socks or stocking? or hose)).ti,ab,kf.
123. ((lung? or organ?) adj2 transplantation).ti,ab,kf.
124. ((electrical or bioelectric or nerve or transcranial or trans-cranial) adj3 (stimulator or stimulation*)).ti,ab,kf.
125. ((monopolar or intervention*) adj3 (radiofrequenc* or radio frequenc*)).ti,ab,kf.
126. (neurophysiological facilitation or neuromodulation or Parasym? or Dolphin* or tDCS or INDIBA).ti,ab,kf.
127. (nerve block* or neuromuscular block* or ganglion block).ti,ab,kf.
128. (hyperbaric oxygen therap* or HBOT or supplemental oxygen or hydrogen therap* or (hydrogen adj3 inhalation) or molecular hydrogen).ti,ab,kf.
129. exp mental health care/ or exp psychiatric treatment/ or virtual reality/ or exp computer assisted therapy/ or exp experimental therapy/
130. (cognitive restructuring or ICBT or CBT or mind-body or meditation* or mentalPlus or hypnotherapy* or hypnosis or phototherapy or psychotherap* or progressive muscle relaxation or breathing retraining or pranayama* or olfactory training).ti,ab,kf.
131. ((cognitive or singing or breathing or breath or breathe or choral or choir or opera or ENO) adj2 (class or classes or instruction* or intervention* or training or program*)).ti,ab,kf.
132. (virtual reality or virtual environment? or virtual technolog* or ((computer* or digital or online or smartphone or virtual or web or webbased) adj3 (game? or gaming or app)) or gamification).ti,ab,kf.
133. exp alternative medicine/ or chiropractic/ or exp osteopathic medicine/ or exp acupuncture/ or homeopathy/
134. (complementary medicine? or alternative medicine? or traditional medicine?).ti,ab,kf.
135. (acupuncture or acupressure or auriculotherap* or acupoint? or chiropract* or osteopath* or reflexology*).ti,ab,kf.
136. (biofeedback or bio-feedback or sensory feedback).ti,ab,kf.
137. (Ayurved* or Agastya Haritaki or Ashwagandha or CIM-Meg19 or Pippali).ti,ab,kf.
138. (homoeopath* or naturopath*).ti,ab,kf.
139. (ozone or autohemotherapy*).ti,ab,kf.
140. or/95-139
Embase results before limits: post–COVID-19 combined with treatment or management
141. (81 or 94) and 140
142. 72 or 141
143. 142 use oemezd
MEDLINE and Embase results combined before limits
144. 69 or 143
Human filter
145. exp animals/
146. exp animal experimentation/ or exp animal experiment/
147. exp models animal/
148. nonhuman/
149. exp vertebrate/ or exp vertebrates/
150. or/145-149
151. exp humans/
152. exp human experimentation/ or exp human experiment/
153. or/151-152
154. 150 not 153
155. 144 not 154
Limits for publication types, language, and date (date limits are edited for search updates)
156. (comment or newspaper article or editorial or letter or note).pt.
157. 155 not 156
158. 157 not (conference abstract or conference review or preprint).pt.
159. limit 158 to (english or french)
160. 159 use medall
161. 160 and (2022013* or 202202* or 202203* or 202204* or 202205* or 202206* or 202207*).dt,ez,da. [Create Date, Entrez date, Entry date]
162. 159 use oemezd
163. limit 162 to dc=20220130-20220720 [Embase date created]
Final results, deduplicated in Ovid
164. 161 or 163
165. remove duplicates from 164
Grey Literature
Dates of search for current report: July 19 to 21, 2022.
Alerts: Grey literature search updates every 6 months until completion of the living review.
Keywords: post–COVID-19, long COVID, post acute sequelae of COVID, chronic COVID condition, and synonyms; along with treatments (drug, device, or other non-drug) or care models. Publications solely on rehabilitation or exercise were excluded.
Limits: Publication years: 2019 to present. As this grey literature search is an update of the original grey literature searches for the first scoping review, particular attention was paid to new publications since January 2022.
Relevant websites from the following sections of the CADTH grey literature checklist Grey Matters: A Practical Tool for Searching Health-Related Grey Literature were searched:
Health Technology Assessment Agencies
Clinical Practice Guidelines
Clinical Trials Registries
Databases (free)
Internet Search
Appendix 2: Supplemental Tables and Figures
Note that this appendix has not been copy-edited.
Category | Data items – Categorical (options) | Data extracted – Open text |
|---|---|---|
Report information |
|
|
Population |
|
|
Treatment and managementa |
|
|
Context |
| NA |
ICU = intensive care unit; NA = not applicable; PCR = polymerase chain reaction; SARS-CoV-2 = severe acute respiratory syndrome coronavirus 2.
aIndicates questions where multiple categories could be selected.
Appendix 3: Patient Involvement in the Treatment and Management of Post–COVID-19 Condition
Note that this appendix has not been copy-edited.
Table 13: Patient Involvement in the Treatment and Management of Post–COVID-19 Condition
Section | Item number | Item | Section heading |
|---|---|---|---|
Aim | |||
Aim | 1 | Three patient contributors were involved in a consultation, commenting on priorities of patients with experience of post–COVID-19 condition (long COVID). | Methods Patient Engagement |
Methods | |||
Methods | 2 | After giving informed consent, 3 patient contributors discussed their perspectives on the scoping review themes and their priorities for post–COVID-19 condition research and treatment via a 1-hour zoom call and follow-up email correspondence. | Methods Patient Engagement |
Results | |||
Results of engagement | 3 | The researchers were made aware of the importance of several themes, in particular the accessibility of diagnostics and treatments (i.e., limited specialists, long wait times, access in rural and remote communities); the variability of symptoms and diagnoses (i.e., postural orthostatic tachycardia syndrome, post-exertion malaise, cardiac and lung damage); treatments of interest; and the need for knowledge dissemination and clinician education. Patients described a lack of empathy from their doctors, not being believed, biases in the health care system, and medical gaslighting. Patients highlighted the need to distinguish between the subtypes of post–COVID-19 condition (i.e., persistent symptoms like a cough or fatigue that eventually resolve), and chronic health issues that arise from post–COVID-19 condition (such as cardiac and lung damage, postural orthostatic tachycardia syndrome, cognitive symptoms, ongoing fevers, and gastrointestinal symptoms). | Results Patient Engagement |
Discussion and conclusions | |||
Discussion and conclusions | 4 | Success of patient engagement in the scoping review is related to several factors. The patient contributors were briefed on the objectives of the project in an introductory call and supported in their role in the engagement process by a Patient Engagement Officer. The research team was receptive to this involvement and incorporated it in their approach to the clinical evidence. Compensation was offered for patient collaborators’ time and expertise. However, there were limitations. The scoping review themes were already determined before engaging the patient collaborators. | Discussion |
Reflections and critical perspective | 6 | The patient collaborators were highly engaged in the consultation, sharing their perspectives, priorities, and lived experiences. One limitation of our approach is that people need reliable internet access to contribute to CADTH’s work, which may exclude some voices. | Discussion Limitations |
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up-to-date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third-party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.
![This figure presents a flow chart of the studies that were selected in this review. A total of 1,131 records were identified from electronic databases, and of these 148 were removed before screening. 983 records were then screened, and of these, 879 were excluded. Following initial screening, 104 records were sought for retrieval, an additional 34 records were identified from the grey literature, and another 74 records were identified from the baseline review. 212 records were then assessed for eligibility, with 99 reports being excluded for various reasons (e.g., duplicates [n = 2], follow-up < 12 weeks [n = 53], ineligible intervention [n = 34], ineligible study design [n = 12]). A total of 113 reports were included in this review (5 rapid reviews, 7 systematic reviews, 7 guidelines, and 94 primary studies, of which 63 were case reports/series.](https://canjhealthtechnol.ca/index.php/cjht/article/download/RE0040sr/version/531/1111/3759/RE0040-fig03.png)