Drugs, Health Technologies, Health Systems
Health Technology Review
Cabozantinib for Second-Line Treatment of Advanced or Metastatic Renal Cell Carcinoma
Key Messages
What Is the Issue?
Immune checkpoint inhibitor–based therapies, especially dual immune checkpoint inhibitor regimens, are now frequently used as initial treatments for advanced or metastatic renal cell carcinoma (RCC).
Cabozantinib has shown promise in RCC; however, cabozantinib is currently funded in the second-line setting for adult patients with advanced or metastatic RCC who have previously received other vascular endothelial growth factor (VEGF)-targeted therapy, limiting its availability in some second-line scenarios.
There is a need to understand the clinical effectiveness of cabozantinib when used after the failure of first-line immune checkpoint inhibitor (ICI) regimens.
What Did We Do?
To inform decisions about the use of cabozantinib for adults with advanced or metastatic RCC who have previously received ICIs in the first line, we sought to identify and summarize related evidence regarding the clinical effectiveness of second-line treatment with cabozantinib compared with sunitinib, axitinib, and pazopanib.
We searched key resources, including journal citation databases, and conducted a focused internet search for relevant evidence published since 2020.
One reviewer screened articles for inclusion based on predefined criteria, critically appraised the included publication, and summarized the findings.
What Did We Find?
One systematic review that included 2 small retrospective observational studies provided evidence for the research question.
Limited retrospective evidence suggests that second-line treatment with cabozantinib may be associated with numerically longer progression-free survival (PFS) compared to sunitinib and axitinib, but shorter PFS compared to pazopanib.
Cabozantinib may also have a potentially higher objective response rate (ORR) compared to sunitinib, axitinib, and pazopanib.
One small retrospective study indicated that cabozantinib had the numerically lowest rate of treatment discontinuation due to toxicity compared to sunitinib, pazopanib, and axitinib.
No studies reporting adverse event rates, overall survival (OS), or quality of life were identified.
What Does It Mean?
Decision-makers should consider the limited quantity and quality of the current evidence available for this patient population. Due to very low confidence in the body of evidence (small sample size, retrospective design, lack of statistical analyses), decision-makers choosing among second-line interventions cabozantinib, sunitinib, axitinib, and pazopanib should consider factors such as clinician expertise and/or patients’ values and preferences.
Higher-quality research (with larger sample sizes, appropriate statistical tests, and transparent reporting) is needed to better inform decision-making, such as robust clinical trials or well-planned prospective observational studies focusing on second-line therapy with tyrosine kinase inhibitors (TKIs) such as cabozantinib.
Abbreviations
CI
confidence interval
ICI
immune checkpoint inhibitor
ORR
objective response rate
OS
overall survival
PFS
progression-free survival
RCC
renal cell carcinoma
TKI
tyrosine kinase inhibitor
VEGF
vascular endothelial growth factor
VEGFR
vascular endothelial growth factor receptors
Context and Policy Issues
What Is RCC and Its First-Line Treatment?
RCC, a malignancy originating in the renal cortex, accounts for 80% to 85% of primary renal neoplasms.1 In Canada, RCC is the 10th most common cancer, with an estimated 8,100 new diagnoses and 1,950 deaths from RCC each year.2 Due to its often asymptomatic nature, approximately 25% of individuals with RCC are diagnosed at an advanced or metastatic stage.3 ICI-based systemic therapies, such as the combination of TKIs and ICIs, or dual ICI regimens (e.g., ipilimumab plus nivolumab), have become the standard of care for advanced or metastatic RCC in the first-line setting.4 However, patients often experience disease progression despite initial benefits from these treatments, necessitating effective second-line strategies.4,5
What Is the Second-Line Treatment of Advanced or Metastatic RCC?
Second-line therapy for advanced or metastatic RCC requires careful consideration of the agents used, patient eligibility for specific therapy, and individual patient and disease characteristics.5,6 When advanced or metastatic RCC progresses after first-line therapy, the previous treatment received often guides the selection of a second-line therapy.5,6 For patients progressing after an ICI regimen (with or without prior antiangiogenic therapy), antiangiogenic therapy is often the second-line option.6,7
What Is Antiangiogenic Therapy?
Antiangiogenic therapy disrupts tumour growth by targeting the VEGF pathway, thereby inhibiting the formation of new blood vessels (angiogenesis).6,7 This approach often employs TKI, a class of drugs that block tyrosine kinase enzymes involved in signal transduction pathways associated with cell division and survival, including those related to angiogenesis. Examples of TKIs include cabozantinib, sunitinib, pazopanib, axitinib, sorafenib, lenvatinib, and tivozanib.7 While all these agents target the VEGF pathway, their specific tyrosine kinase profiles, indications, efficacy, and safety profiles differ.7 For instance, cabozantinib inhibits VEGF receptors (VEGFR) as well as MET and AXL genes, whereas sunitinib primarily targets VEGFR but also inhibits tyrosine kinases linked to the PDGF receptor and the c-KIT oncogene.7 Pazopanib targets TKIs associated with VEGFR, platelet-derived growth factor receptor, and KIT receptor, whereas axitinib selectively inhibits VEGFR1, VEGFR2, and VEGFR3.7
A consensus document from the Kidney Cancer Research Network of Canada on the management of advanced kidney cancer notes that cabozantinib, axitinib, sunitinib, and pazopanib with or without ICI regimens could be used in both first-line and later settings.8 Moreover, cabozantinib has demonstrated advantages in specific patient populations, such as those with bone metastases, and is often considered after the failure of other treatments.7,8
Why Is It Important to Do This Review?
Although cabozantinib has shown promise in advanced or metastatic RCC, there are limited data that specifically address its efficacy and safety in the second-line setting, after initial ICI treatment.8,9 In Canada, cabozantinib is currently funded for adult patients with advanced or metastatic RCC who have previously received other VEGF-targeted therapy, with or without ICI therapy, for the second-line setting.10 Given ICI-based therapies are commonly employed in the first-line setting (particularly dual ICI),4,5,8 there is a need to understand the clinical effectiveness of cabozantinib when used as a second-line treatment following progression on initial ICI regimens.
Objectives
This rapid review aims to evaluate the evidence for cabozantinib as a second-line treatment for adults with advanced or metastatic RCC whose disease has progressed after receiving prior ICI therapy in the first line. The review will compare cabozantinib to other commonly used antiangiogenic agents, including sunitinib, pazopanib, and axitinib in this patient population.
Research Question
What is the clinical effectiveness of cabozantinib in patients with advanced or metastatic renal cell carcinoma who have previously received immune checkpoint inhibitors in the first line?
Methods
Literature Search Methods
An information specialist conducted a literature search on key resources including MEDLINE, Embase, the Cochrane Database of Systematic Reviews, the International HTA Database, the websites of Canadian and major international health technology agencies, as well as a focused internet search. The search approach was customized to retrieve a limited set of results, balancing comprehensiveness with relevancy. The search strategy comprised both controlled vocabulary, such as the National Library of Medicine’s MeSH (Medical Subject Headings), and keywords. Search concepts were developed based on the elements of the research questions and selection criteria. The main search concepts were cabozantinib and RCC. The search was completed on February 7, 2025, and limited to English-language documents published since January 1, 2020.
Selection Criteria and Methods
One reviewer screened citations and selected studies. In the first level of screening, titles and abstracts were reviewed and potentially relevant articles were retrieved and assessed for inclusion. The final selection of full-text articles was based on the inclusion criteria presented in Table 1.
Criteria | Description |
|---|---|
Population | People aged 18 years and older with advanced or metastatic renal cell carcinoma whose disease has progressed after receiving prior systemic therapy for metastatic disease with an immune checkpoint inhibitor in the first line |
Intervention | Cabozantinib |
Comparator | Sunitinib, pazopanib, axitinib |
Outcomes | Overall survival, progression-free survival, objective response rate, quality of life, adverse events, treatment discontinuation |
Study designs | Randomized controlled trials, health technology assessments, systematic reviews including published network meta-analysis or meta-analysis |
Exclusion Criteria
We excluded studies if they did not meet the selection criteria outlined in Table 1, were duplicate publications, or were published before 2020. We excluded systematic reviews if more recent or comprehensive reviews had already captured all relevant studies. Additionally, we excluded primary studies if they were included in 1 or more of the selected systematic reviews. We also excluded single-arm studies and those with unclear, undefined, or combined comparators.
Critical Appraisal of the Individual Study
One reviewer critically appraised the included systematic review using A MeaSurement Tool to Assess systematic Reviews 2 (AMSTAR 2)11 as a guide. Summary scores were not calculated for the included studies; rather, the strengths and limitations of each included publication were described narratively.
Summary of Evidence
Quantity of Research Available
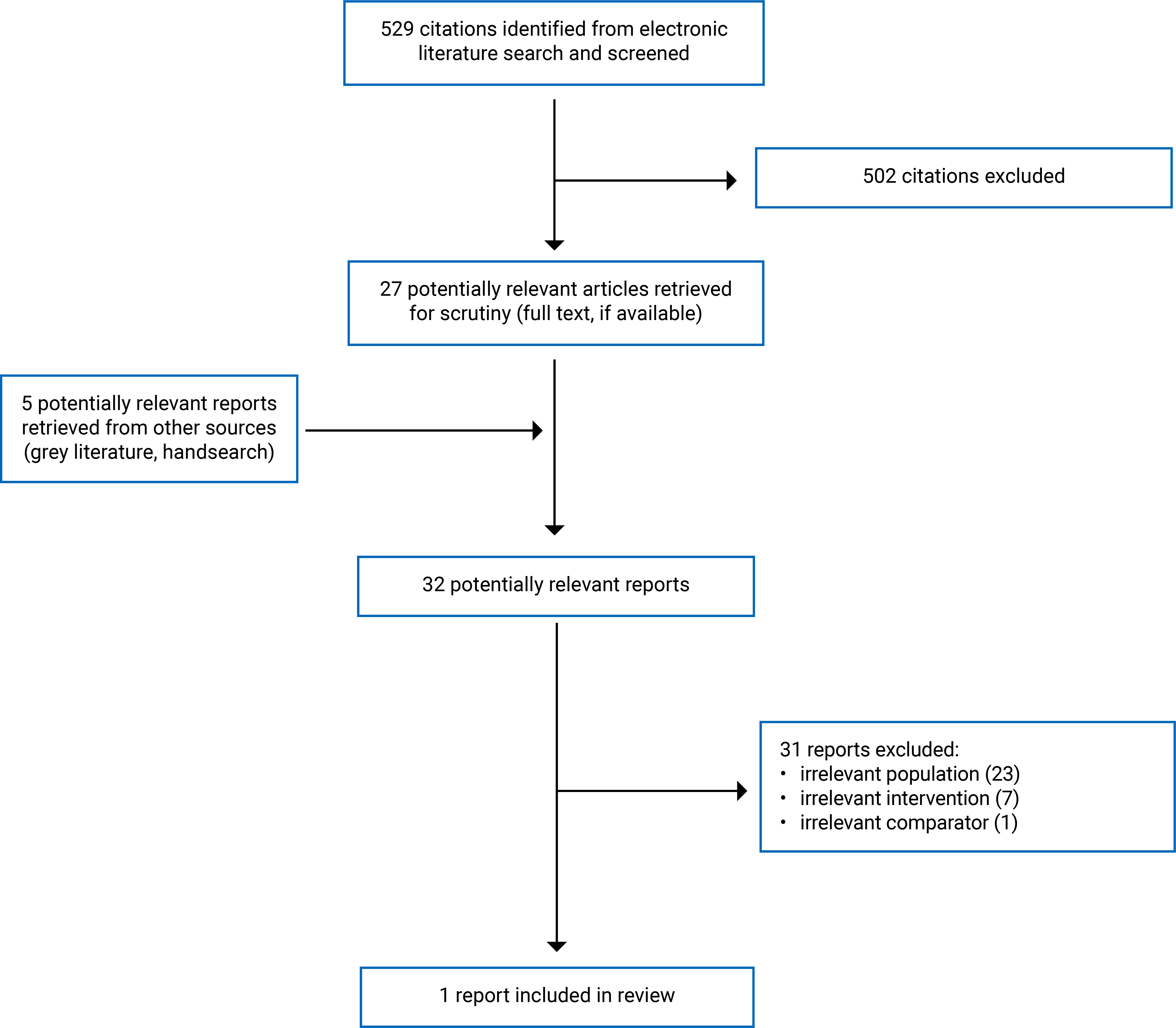
We identified a total of 529 citations in the literature search. After screening titles and abstracts, we excluded 502 citations and retrieved 27 potentially relevant reports from the electronic search for full-text review. Additionally, 5 potentially relevant publications from the grey literature search were retrieved for full-text review. Of these 32 potentially relevant articles, we excluded 31 publications based on the inclusion criteria and included 1 systematic review12 that met the inclusion criteria. Figure 1 (Appendix 1) presents the PRISMA13 flow chart of the study selection. Appendix 2 presents additional references of potential interest that were excluded. We did not find any randomized controlled trials or health technology assessments that met our inclusion criteria.
Summary of Study Characteristics
The systematic review12 had broader inclusion criteria than the present review. Specifically, the systematic review included studies on patients with solid tumours, such as RCC, hepatocellular carcinoma, metastatic urothelial carcinoma, and non–small cell lung cancer.12 This rapid review will describe only the characteristics and results of the subset of relevant studies on RCC.
This rapid review includes 1 systematic review that compared the efficacy or effectiveness of cabozantinib with sunitinib, pazopanib, or axitinib after prior ICI therapy among solid tumours.12 The systematic review, conducted in Canada, included data from 4 clinical trials and 14 retrospective observational studies on patients with RCC.12 Among these, only 2 retrospective observational studies provided the relevant drug comparators.12 The clinical trials and other observational studies examined cabozantinib as a second or later line of treatment (i.e., in a mixed population or as a third or later line of treatment), using unclear comparators (e.g., any monotherapy; or any combination, which likely included cabozantinib), or single-arm studies.12 The systematic review authors did not report the total sample size but provided the sample size for each arm.12 The clinical outcomes assessed included OS, PFS, ORR, and treatment discontinuation due to toxicity.12
Appendix 3 presents additional details regarding the characteristics of included publications.
Summary of Critical Appraisal
The systematic review clearly described its objectives.12 The preregistered protocol in PROSPERO and the comprehensive search across multiple databases (MEDLINE, Embase, and Cochrane Library) with documented search terms support the rigour of the identification process.12 The systematic review authors also conducted manual searches of bibliographies and abstract books, provided a study selection flow chart, performed dual data extraction, and assessed the risk of bias for eligible individual studies.12
The absence of a list of excluded studies limited understanding of the study selection process.12 Only 1 reviewer performed the selection of studies,12 which may have resulted in missing some eligible studies. The lack of clear reporting on participant, intervention, and control details hindered the ability to assess the applicability of the findings.12 The systematic review authors used the Grading of Recommendations Assessment, Development and Evaluation framework to assess the risk of bias for individual clinical trials,12 although Grading of Recommendations Assessment, Development and Evaluation was designed to assess the overall certainty of evidence.14 This likely resulted in inaccurate conclusions regarding the risk of bias. The failure to analyze the impact of risk of bias on outcome interpretations and the absence of statistical testing for group comparisons make the interpretation of evidence challenging. The funding source from Ipsen introduces a potential conflict of interest that needs to be considered when interpreting the review's findings.12
Appendix 4 presents additional details regarding the strengths and limitations of included publications.
Summary of Findings
Based on the systematic review included in this review,12 we have summarized the effectiveness and safety of cabozantinib for advanced or metastatic RCC compared with sunitinib, pazopanib, or axitinib. Appendix 5 presents the summary of findings by different outcomes.
Clinical Effectiveness of Cabozantinib Compared With Sunitinib, Pazopanib, or Axitinib
Based on retrospective observational data, the systematic review authors concluded that cabozantinib is at least as effective as other TKIs (i.e., sunitinib, pazopanib, or axitinib) for treating patients with advanced or metastatic RCC after ICI therapy.12
Progression-Free Survival
Two retrospective observational studies in the systematic review12 reported on PFS. One retrospective observational study found that cabozantinib (n = 20; 15.2 months) had a numerically longer median PFS than sunitinib (n = 6; 3.6 months) and axitinib (n = 25; 13.2 months) but a numerically shorter median PFS than pazopanib (n = 19; 24.4 months).12 However, all the confidence intervals (CIs) were extremely wide with significant overlap, indicating that any of the drugs compared could be favoured, though no statistical tests were conducted among these groups.12 The other retrospective observational study in this systematic review could not estimate the PFS for the cabozantinib group (n = 3).12
Objective Response Rate
Two retrospective observational studies in the systematic review reported on the ORR.12 One study found that cabozantinib (n = 3; 33%) had a numerically higher ORR than sunitinib (n = 4; 0%) and axitinib (n = 14; 29%) but a numerically lower ORR than pazopanib (n = 7; 43%).12 The other retrospective observational study found that cabozantinib (n = 19; 47.4%) had the numerically highest ORR compared to sunitinib (n = 6; 16.7%), pazopanib (n = 19; 42.1%), or axitinib (n = 24; 41.7%).12 The systematic review did not report CIs for these estimates and did not conduct any statistical tests among these groups.12
OS and Quality of Life
No relevant studies reported the effectiveness of cabozantinib compared with sunitinib, pazopanib, or axitinib as a second-line treatment on OS and quality of life.12
Safety of Cabozantinib Compared With Sunitinib, Pazopanib, or Axitinib
Treatment Discontinuation (Due to Toxicity)
One retrospective observational study in the systematic review reported on treatment discontinuation due to toxicity.12 The retrospective observational study found that cabozantinib (n = 19; 0%) had the numerically lowest rate of treatment discontinuation due to toxicity compared to sunitinib (n = 6; 17%), pazopanib (n = 19; 42%), or axitinib (n = 24; 12%).12 The systematic review did not report CIs for these estimates and did not conduct any statistical tests among these groups.12
Adverse Events
No relevant studies reported the adverse event rates of cabozantinib compared with sunitinib, pazopanib, or axitinib in the second-line setting.12
Limitations
In this rapid review, we identified 1 systematic review12 to answer the research questions on the effectiveness of cabozantinib compared to sunitinib, pazopanib, or axitinib. Although the included systematic review encompassed 4 clinical trials and 14 retrospective observational studies, only 2 retrospective observational studies involving a small number of patients provided data that met our research question criteria.12 Due to the nature of the observational studies, the body of evidence may be subject to selection bias or performance bias, potentially skewing findings in favour of the interventions. We are concerned about these biases, as they may lead to inaccurate conclusions about intervention effectiveness.
We found several methodological limitations in the body of evidence presented, as in the summary of the critical appraisal section. It is worth emphasizing that the systematic review covered a broader topic than our research question.12 The systematic review included several solid tumours and examined cabozantinib as a treatment beyond the second line,12 while we only focused on evidence for RCC and cabozantinib as a second-line treatment. The systematic review12 only presented outcome numbers without statistical tests, making the interpretation of between-group differences challenging.
In this rapid review, we also identified some evidence gaps. We found no high-quality evidence addressing our research question. There was no direct evidence regarding OS and quality of life. The 2 observational studies in the systematic review12 did not report comparisons between these agents on adverse event rates, and only 1 study reported treatment discontinuation due to drug toxicity. Although the systematic review was conducted in Canada, the country of the patients included in the 2 relevant studies was not reported,12 making the generalizability of the findings to settings in Canada unclear.
Conclusions and Implications for Decision- or Policy-Making
In this rapid review, we identified 1 systematic review12 to answer the research question. Two retrospective observational studies in the systematic review12 reported on the effectiveness of cabozantinib compared to sunitinib, pazopanib, or axitinib.
Based on 2 retrospective studies in the systematic review that had significant limitations,12 second-line cabozantinib may be associated with numerically longer PFS compared to second-line sunitinib and axitinib but shorter PFS compared to second-line pazopanib. The systematic review also indicates a potentially higher ORR with second-line cabozantinib compared to sunitinib, axitinib, and pazopanib.12 However, 1 observational study in the systematic review, which included a small cabozantinib cohort (n = 3), reported a numerically lower ORR compared to pazopanib.12 Based on 1 retrospective study with a small sample size, cabozantinib had the numerically lowest rate of treatment discontinuation due to toxicity compared to sunitinib, pazopanib, and axitinib.12 No studies reporting on adverse event rates, OS, or quality of life were identified.
Due to the small sample size, retrospective design, and lack of statistical tests, it is challenging to draw definitive conclusions. The limited evidence suggests that second-line cabozantinib may or may not be as effective as second-line sunitinib, axitinib, or pazopanib in terms of PFS and ORR and that it may have the lowest treatment discontinuation rate.12
Decision-makers should consider the limited quantity and quality of the current evidence available for this patient population. Due to very low confidence in the body of evidence, decisions regarding the choice of second-line interventions among cabozantinib, sunitinib, axitinib, or pazopanib should likely be based on other factors outside of the evidence summarized in this rapid review, such as clinician expertise and/or patients’ values and preferences.
Higher-quality research (larger sample sizes, appropriate statistical tests, and transparent reporting) is needed to better inform decision-making, such as robust clinical trials or well-planned prospective observational studies focusing on second-line therapy with TKIs such as cabozantinib.
References
1.Atkins MB, El Bakouny Z, Choueiri TK, UpToDate. Post TW, ed. Epidemiology, pathology, and pathogenesis of renal cell carcinoma 2025. Accessed February 6, 2025. http://www.uptodate.com
2.Brenner DR, Poirier A, Woods RR, et al. Projected estimates of cancer in Canada in 2022. CMAJ. 2022;194(17):E601-E607. doi:10.1503/cmaj.212097 PubMed
3.Atkins MB, UpToDate. Clinical presentation, diagnosis, and staging of renal cell carcinoma. Accessed February 6, 2025. http://www.uptodate.com
4.Cardenas LM, Sigurdson S, Wallis CJD, Lalani AK, Swaminath A. Advances in the management of renal cell carcinoma. CMAJ. 2024;196(7):E235-E240. doi:10.1503/cmaj.230356 PubMed
5.George D, Jonasch E, UpToDate. Post TW, ed. Systemic therapy for advanced and metastatic clear cell renal carcinoma. Accessed February 6, 2025. http://www.uptodate.com
6.Atkins MB, UpToDate. Post TW, ed. Overview of the treatment of renal cell carcinoma. Accessed February 6, 2025. http://www.uptodate.com
7.Atkins MB, UpToDate. Post TW, ed. Antiangiogenic and molecularly targeted therapy for advanced or metastatic clear cell renal carcinoma Accessed February 6, 2025. http://www.uptodate.com
8.Canil C, Kapoor A, Basappa NS, et al. Management of advanced kidney cancer: Kidney Cancer Research Network of Canada (KCRNC) consensus update 2021. Can Urol Assoc J. 2021;15(4):84-97. doi:10.5489/cuaj.7245 PubMed
9.Navani V, Wells JC, Boyne DJ, et al. CABOSEQ: the effectiveness of cabozantinib in patients with treatment refractory advanced renal cell carcinoma: results from the International Metastatic Renal Cell Carcinoma Database Consortium (IMDC). Clin Genitourin Cancer. 2023;21(1):106.e1-106.e8. doi:10.1016/j.clgc.2022.07.008 PubMed
10.Canada’s Drug Agency. Renal Cell Carcinoma (RCC). Provisional funding algorithm. 2022. Accessed February 6, 2025. https://www.cda-amc.ca/renal-cell-carcinoma-rcc
11.Shea BJ, Reeves BC, Wells G, et al. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ. 2017;358:j4008. doi:10.1136/bmj.j4008 PubMed
12.Graham J, Vogel A, Cheng A-L, Bjarnason GA, Neal JW. Cabozantinib after prior checkpoint inhibitor therapy in patients with solid tumors: a systematic literature review. Cancer Treat Rev. 2022;110:102453. doi:10.1016/j.ctrv.2022.102453 PubMed
13.Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol. 2009;62(10):e1-e34. PubMed
14.Guyatt GH, Oxman AD, Vist GE, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 2008;336(7650):924-6. doi:10.1136/bmj.39489.470347.AD PubMed
Appendix 1: Selection of Included Studies
Please note that this appendix has not been copy-edited.
Figure 1: PRISMA13 Flow Chart of Study Selection
Appendix 2: References of Potential Interest
Please note that this appendix has not been copy-edited.
Systematic Reviews
Wenzel M, Deuker M, Nocera L, et al. Median time to progression with TKI-based therapy after failure of immuno-oncology therapy in metastatic kidney cancer: a systematic review and meta-analysis. Eur J Cancer. 2021;155:245-255. doi:10.1016/j.ejca.2021.07.014. PubMed: PM34392067.
Liao Y, Hou H, Han Z, Liu Y. Systemic therapies for metastatic renal cell carcinoma in the second-line setting: a systematic review and network meta-analysis. Medicine (Baltimore). 2022;101(37):e30333. doi:10.1097/MD.0000000000030333. PubMed: PM36123840.
Heo JH, Park C, Ghosh S, Park S-K, Zivkovic M, Rascati KL. A network meta-analysis of efficacy and safety of first-line and second-line therapies for the management of metastatic renal cell carcinoma. J Clin Pharm Ther. 2021;46(1):35-49. doi:10.1111/jcpt.13282. PubMed: PM33112003.
Phase II Study
Albiges L, Schmidinger M, Taguieva-Pioger N, Perol D, Grunwald V, Guemas E. CaboPoint: a phase II study of cabozantinib as second-line treatment in patients with metastatic renal cell carcinoma. Future Oncol. 2022;18(8):915-926. PubMed: PM37980876.
Nonrandomized Studies
Santoni M, Aurilio G, Massari F, et al. Nivolumab VERSUS cabozantinib as second-line therapy in patients with advanced renal cell carcinoma: a real-world comparison. Clin Genitourin Cancer. 2022;20(3):285-295. doi:10.1016/j.clgc.2022.02.003. PubMed: PM35305916.
Fujita K, Matsushita Y, Toyoda S, et al. The efficacy of second-line tyrosine kinase inhibitor for patients with metastatic non-clear cell renal cell carcinoma following first-line immune-oncology combination therapy. World J Urol. 2024;42(1):536. doi:10.1007/s00345-024-05252-2. PubMed
Narang A, Gebrael G, Jo Y, et al. Effectiveness of second-line cabozantinib in metastatic clear cell renal cell carcinoma patients after first-line treatment with immune checkpoint inhibitor-based combinations. Kidney Cancer. 2024;8(1):135-142. doi:10.3233/KCA-240016 PubMed
Tomida R, Takahashi M, Matsushita Y, et al. Comparison of cabozantinib and axitinib as second-line therapy after nivolumab plus ipilimumab in patients with metastatic clear cell renal cell carcinoma: a comparative analysis of retrospective real-world data. Clin Genitourin Cancer. 2024;22(3):102094. doi:10.1016/j.clgc.2024.102094. PubMed
Sazuka T, Matsushita Y, Sato H, et al. Efficacy and safety of second-line cabozantinib after immuno-oncology combination therapy for advanced renal cell carcinoma: Japanese multicenter retrospective study. Sci Rep. 2023;13(1):20629. doi:10.1038/s41598-023-48087-4. PubMed
Kang DH, Lee JY, Lee Y, Ha US. Optimal sequencing of the first- and second-line target therapies in metastatic renal cell carcinoma: based on nationally representative data analysis from the Korean National Health Insurance System. BMC Cancer. 2023;23(1):483. doi:10.1186/s12885-023-10991-3. PubMed
Reviews
Shaw T, Lee H, Figlin R. Second-line therapies in the changing landscape of first-line therapies for metastatic clear cell renal cell cancer. Oncology (Williston Park). 2021;35(6):306-310. doi:10.46883/ONC.2021.3506.0306. PubMed: PM34139792.
Deuker M, Chun FKH, Karakiewicz PI. Second-line tyrosine kinase inhibitor-therapy after immunotherapy-failure. Curr Opin Support Palliat Care. 2020;14(3):276-285. doi:10.1097/SPC.0000000000000519. PubMed: PM32769619.
Additional References
Guadalupi V, Carteni G, Iacovelli R, et al. Second-line treatment in renal cell carcinoma: clinical experience and decision making. Ther Adv Urol. 2021;13:17562872211022870. doi:10.1177/17562872211022870. PubMed: PM34211586.
Appendix 3: Characteristics of Included Publications
Please note that this appendix has not been copy-edited.
Table 2: Characteristics of Included Systematic Reviews
Study citation, country, funding source | Study designs and numbers of primary studies included | Population characteristics | Intervention and comparator(s) | Clinical outcomes, length of follow-up |
|---|---|---|---|---|
Graham et al. (2022)12 Canada Funding source: Ipsen | Study design: systematic review of clinical trials and observational studies Number of included publications: 21 (18 for RCC) Search: on May 19, 2021 | Patients with RCC, HCC, metastatic urothelial carcinoma, and non–small cell lung cancer who received ICIs therapy Number of participants: Total: NR; Barata et al.: n = 28; Shah et al.: n = 70 Median age (range): Total: NR; Barata et al.: 57 (37 to 77) years; Shah et al.: 59 (44 to 75) years Sex: female: Total: NR; Barata et al.: 15%; Shah et al.: 29% Disease duration: NR | Intervention: cabozantinib Comparator: sunitinib, pazopanib, axitinib, any monotherapy, any combination therapy. | Relevant outcomes:
Follow-up: NR |
AE = adverse event; CPIs = immune checkpoint inhibitors; HCC = hepatocellular carcinoma; NR = Not reported; ORR = objective response rate; OS = overall survival; PFS = progression-free survival; RCC = renal cell carcinoma.
Appendix 4: Critical Appraisal of Included Publications
Please note that this appendix has not been copy-edited.
Table 3: Strengths and Limitations of Systematic Reviews Using AMSTAR 211
Strengths | Limitations |
|---|---|
Graham et al. (2022)12 | |
The purpose of the study was clearly described. The protocol of this review was registered in the PROSPERO (CRD42021259873). Multiple databases were searched (MEDLINE, Embase, and Cochrane Library). The search terms in the 3 databases were provided. The authors manually searched bibliographies of relevant references and abstract books for several congresses to identify additional publications. A flow chart of study selection was provided. Data extraction was conducted by 2 researchers. The review authors assessed the studies’ RoB. The study designs of the individual study for inclusion were clearly described. | The list of excluded studies was not provided. The study selection was conducted by 1 reviewer. The details of participants, intervention, and control were not clearly reported. The authors used GRADE to rate the risk of bias. However, GRADE was designed to assess the overall certainty of evidence. The follow-up of some outcome measures (e.g., progressive disease) was unclear. The review authors stated that they included all eligible publications regarding their risk of bias and did not conduct further analyses on the impact of risk of bias on the outcome interpretations. There are no statistical tests conducted between groups for most of the comparisons. The review was founded by Ipsen, which is the manufacturer of the intervention (i.e., cabozantinib). |
AMSTAR 2 = A MeaSurement Tool to Assess systematic Reviews 2; GRADE = Grading of Recommendations Assessment, Development, and Evaluation; RoB = risk of bias.
Appendix 5: Main Study Findings
Please note that this appendix has not been copy-edited.
Table 4: Summary of Findings by Outcome — Progression-Free Survival
Study | Group | Sample size (n) | Months (95% CI) | Notes |
|---|---|---|---|---|
Graham et al. (2022)12 Systematic review | cabozantinib | 3 | NE | Barata et al. |
axitinib | 14 | 6.4 (4.4 to 8.4) | Barata et al. | |
pazopanib | 7 | 5.6 (1.2 to 10) | Barata et al. | |
sunitinib | 4 | 2.9 (0 to 7.6) | Barata et al. | |
Graham et al. (2022)12 Systematic review | cabozantinib | 20 | 15.2 (7.9 to NE) | Shah et al. |
pazopanib | 19 | 24.4 (6.1 to NE) | Shah et al. | |
sunitinib | 6 | 3.6 (0.9 to NE) | Shah et al. | |
axitinib | 25 | 13.2 (8.6 to NE) | Shah et al. |
CI = confidence interval; NE = not estimable.
Table 5: Summary of Findings by Outcome — Objective Response Rate
Study | Group | Sample size (n) | Percentage (%) | Notes |
|---|---|---|---|---|
Graham et al. (2022)12 Systematic review | cabozantinib | 3 | 33 | Barata et al. |
axitinib | 14 | 29 | Barata et al. | |
pazopanib | 7 | 43 | Barata et al. | |
sunitinib | 4 | 0 | Barata et al. | |
Graham et al. (2022)12 Systematic review | cabozantinib | 19 | 47.4 | Shah et al. |
pazopanib | 19 | 42.1 | Shah et al. | |
sunitinib | 6 | 16.7 | Shah et al. | |
axitinib | 24 | 41.7 | Shah et al. |
Table 6: Summary of Findings by Outcome — Treatment Discontinuation Due to Toxicity
Study | Group | Sample size (n) | Percentage (%) | Notes |
|---|---|---|---|---|
Graham et al. (2022)12 Systematic review | cabozantinib | 19 | 0 | Shah et al. |
pazopanib | 19 | 42 | Shah et al. | |
sunitinib | 6 | 17 | Shah et al. | |
axitinib | 24 | 12 | Shah et al. |
ISSN: 2563-6596
Canada’s Drug Agency (CDA-AMC) is a pan-Canadian health organization. Created and funded by Canada’s federal, provincial, and territorial governments, we’re responsible for driving better coordination, alignment, and public value within Canada’s drug and health technology landscape. We provide Canada’s health system leaders with independent evidence and advice so they can make informed drug, health technology, and health system decisions, and we collaborate with national and international partners to enhance our collective impact.
Disclaimer: CDA-AMC has taken care to ensure that the information in this document was accurate, complete, and up to date when it was published, but does not make any guarantee to that effect. Your use of this information is subject to this disclaimer and the Terms of Use at cda-amc.ca.
The information in this document is made available for informational and educational purposes only and should not be used as a substitute for professional medical advice, the application of clinical judgment in respect of the care of a particular patient, or other professional judgments in any decision-making process. You assume full responsibility for the use of the information and rely on it at your own risk.
CDA-AMC does not endorse any information, drugs, therapies, treatments, products, processes, or services. The views and opinions of third parties published in this document do not necessarily reflect those of CDA-AMC. The copyright and other intellectual property rights in this document are owned by the Canadian Agency for Drugs and Technologies in Health (operating as CDA-AMC) and its licensors.
Questions or requests for information about this report can be directed to Requests@CDA-AMC.ca.