Drugs, Health Technologies, Health Systems
Health Technology Review
Airway Management in Out-of-Hospital Emergencies
Key Messages
What Is the Issue?
Airway management is key to prehospital emergency care, impacting a patient’s survival and recovery. Endotracheal intubation has been considered the gold standard for airway management, yet its success in uncontrolled settings such as outside of the hospital can vary owing to the complexity of the procedure.
Extraglottic airway devices, which include supraglottic and retroglottic airway devices, are an easier-to-insert alternative to endotracheal intubation; however, their impact on patient outcomes needs review.
What Did We Do?
We compared the effectiveness of different types of extraglottic airway devices with endotracheal intubation, to inform decisions regarding the use of extraglottic airway devices in out-of-hospital emergencies. We focused on 2 types of supraglottic airway devices (i-gels and laryngeal mask airways) and 1 type of retroglottic airway device (King laryngeal tubes). We also sought to identify evidence-based guidelines regarding the use of extraglottic airway devices for this patient population.
We searched key resources, including journal citation databases and conducted a focused internet search for relevant evidence published since 2019. Identified literature was reviewed, appraised, and summarized.
What Did We Find?
Most identified studies were largely comprised of adults who had an out-of-hospital cardiac arrest (OHCA).
I-gels were associated with higher rates of successful device insertion compared with King laryngeal tubes and laryngeal mask airways. I-gels also had higher rates of survival and return of spontaneous circulation (ROSC), and similar rates of adverse events compared with King laryngeal tubes. Significant differences for first-pass success, survival, and ROSC between i-gels and laryngeal mask airways were not reported. We did not identify any studies that met the inclusion criteria for this review that compared laryngeal mask airways with King laryngeal tubes.
When compared with endotracheal intubation, i-gels and King laryngeal tubes were associated with higher rates of successful device insertion; however, King laryngeal tubes, laryngeal mask airways, and i-gels tended to have similar clinical outcomes or inconsistent findings. Exceptions included King laryngeal tubes having higher rates of ROSC, and laryngeal mask airways having lower survival to admission, though the difference on survival to discharge was not significant. A subgroup analysis by a randomized controlled trial (RCT) suggested i-gels may lead to better outcomes than endotracheal intubation for specific patients.
In a nonrandomized study (NRS) that included patients with noncardiac arrest emergencies as well as pediatric patients, extraglottic airway devices were associated with higher rates of first-pass success than endotracheal intubation, with a larger effect seen in pediatric patients.
One evidence-based guideline suggested supraglottic airway devices may be used for patients who had an OHCA. For pediatric patients who had an OHCA, experienced trauma, or experienced a medical emergency, the guideline recommended supraglottic airway devices compared with endotracheal intubation. The guideline noted missing sufficient evidence to make strong recommendations, yet reported that:
supraglottic airway devices were favoured compared with endotracheal intubation for pediatric patients owing to factors such as higher first-pass success rates, harms associated with failed endotracheal intubation attempts, and procedure rarity.
for adults who had an OHCA or medical emergency and determining between supraglottic airway devices and endotracheal intubation, the authors recommended considering documented success with endotracheal intubation. The authors recommended supraglottic airway devices for systems without documented success, and for systems with documented success, they suggested using either strategy.
What Does it Mean?
Additional high-quality randomized studies are needed to fully understand the impact of extraglottic airway devices on patient-important outcomes for OHCA as well as other indications and for pediatric patients.
Studies reported that i-gels were easier to insert and may also be associated with improved outcomes compared with King laryngeal tubes and similar outcomes compared with laryngeal mask airways. The use of the extraglottic airway devices was reported to result in similar patient outcomes as endotracheal intubation for patients who had out-of-hospital emergencies. Both i-gels and King laryngeal tubes were reported as easier to insert than endotracheal intubation.
As most identified studies focused on adults who had an OHCA, it is unclear if these findings are generalizable to other patient groups, including patients with other indications and pediatric patients. Few studies reported on adverse events, which may result in an overestimation of the benefits of extraglottic airway devices. The identified evidence-based guideline includes different recommendations for pediatric and adult patients, which indicates that other factors may influence which advanced airway management strategy is optimal.
Decisions regarding the use of extraglottic airway devices may depend on specific patient factors (e.g., adult versus pediatric, cause of emergency), local factors (e.g., if paramedics can maintain proficiency in endotracheal intubation), and each management strategy’s training needs.
Abbreviations
AHRQ
Agency for Healthcare Research and Quality
ETCO2
end-tidal carbon dioxide
GRADE
Grading of Recommendations Assessment, Development, and Evaluation
NRS
nonrandomized study
OHCA
out-of-hospital cardiac arrest
RCT
randomized controlled trial
ROSC
return of spontaneous circulation
SR
systematic review
Context and Policy Issues
What Is Airway Management?
Airway management are treatments and techniques to allow for oxygenation and ventilation, keeping the airway open.1 Airway management is a vital aspect of prehospital emergency care, and is key to a patient’s survival and their potential for recovery.2 They are used for a variety of critical conditions that require airway management, including cardiac arrest, trauma, medication or drug toxicity, pneumonia, and pulmonary edema.3 A variety of airway management techniques are available, including endotracheal intubation and extraglottic airway devices.2
What are Extraglottic Airway Devices?
Extraglottic airway devices are invasive devices that are inserted through the oropharynx but do not enter the larynx or trachea.3 They are important tools for airway management used to establish an airway for oxygenation and ventilation as well as to administer anesthetic gases without entering the trachea.4 They are used in a variety of health care settings including prehospital.
There are 2 categories of extraglottic airway devices:
Supraglottic airway devices sit in the hypopharynx facing the glottis, with the tip in the esophageal inlet.5 Types of supraglottic airway devices include:
Retroglottic devices are laryngeal tubes that are seated behind the glottic opening, or terminate in the upper esophagus.3,4 One example of a retroglottic device is the King laryngeal tube, which has a pharyngeal cuff and esophageal cuff, with a port between the cuffs at the laryngeal inlet to allow for gas exchange.
Why Is It Important To Do This Review?
Endotracheal intubation is considered the gold standard for airway management;2 however, it is a complex procedure, and its success rate in out-of-hospital emergencies varies.2,6 Extraglottic devices, particularly supraglottic devices, have been increasingly used in out-of-hospital emergency settings owing to their simpler insertion technique and thus can be taught more successfully compared with endotracheal intubation.6,7 Determining which device or procedure is most clinically effective in out-of-hospital and prehospital settings may help to improve patient outcomes.
Objective
The purpose of this report is to summarize and critically appraise the evidence regarding the clinical effectiveness between different types of extraglottic airway devices for out-of-hospital emergencies, as well as extraglottic devices compared with endotracheal intubation. This review focused on 2 types of supraglottic airway devices (i-gels and laryngeal mask airways) and 1 type of retroglottic airway device (King laryngeal tubes). This review also aimed to summarize and critically appraise evidence-based guidelines regarding extraglottic airway devices for out-of-hospital emergencies.
Research Questions
What is the comparative clinical effectiveness of different types of extraglottic airway devices in out-of-hospital emergencies?
What is the clinical effectiveness of extraglottic airway devices versus endotracheal intubation?
What are the evidence-based guidelines regarding extraglottic airway devices in out-of-hospital emergencies?
Methods
An information specialist conducted a customized literature search, balancing comprehensiveness with relevancy, of multiple sources and grey literature on August 8, 2024.
Two reviewers screened citations and selected studies based on the inclusion criteria presented in Table 1. Two reviewers critically appraised and included publications using established critical appraisal tools.
Appendix 1 presents a detailed description of methods.
Criteria | Description |
|---|---|
Population | Individuals in out-of-hospital emergencies (e.g., emergency medical services before the hospital, rural or remote health centres) |
Intervention | Q1,2: Extraglottic airway devices, specifically:
Q3: Extraglottic devices |
Comparator | Q1: Compared with each other, inclusive of the following comparisons:
Q2: Endotracheal intubation Q3: NA |
Outcomes | Q1,2: Clinical benefits and harms (e.g., trauma or throat injury, mortality, morbidity, airway complications) and user and provider satisfaction or preferences Q3: Recommendations regarding best-practices related to their use (e.g., appropriate indication, longevity of use, storage) |
Study designs | Health technology assessments, systematic reviews, randomized controlled trials, nonrandomized studies, evidence-based guidelines |
Publication date | Since January 1, 2019 |
LMA = laryngeal mask airway; NA = not applicable; vs. = versus.
Summary of Evidence
Quantity of Research Available
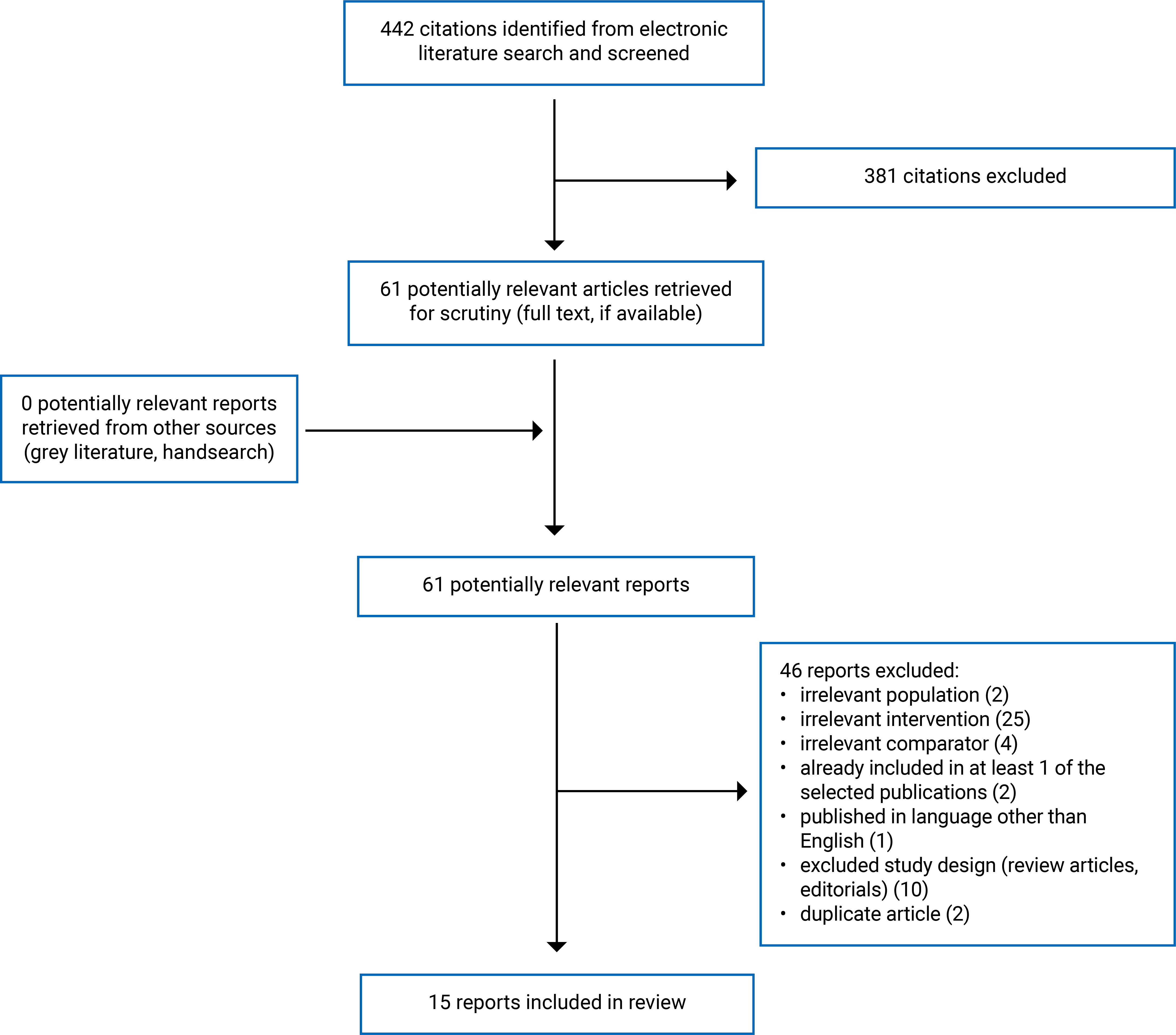
A total of 442 citations were identified. Following screening of titles and abstracts, 381 citations were excluded and 61 potentially relevant reports from the electronic search were retrieved for full-text review. No potentially relevant publications were retrieved from the grey literature search for full-text review. Of these potentially relevant articles, 46 publications were excluded for various reasons, and 15 publications met the inclusion criteria and were included in this report.
Publications included 3 systematic reviews (SRs),2,8,9 1 RCT,10 10 NRSs,11-20 and 1 evidence-based guideline.21 This review did not identify any health technology assessments that met the inclusion criteria. Appendix 2 presents the PRISMA22 flow chart of the study selection.
Additional references of potential interest are provided in Appendix 8.
Summary of Study Characteristics
This review included 15 publications, including 3 SRs,2,8,9 1 RCT,10 10 NRSs,11-20 and 1 evidence-based guideline.21 The 3 included SRs2,8,9 conducted meta-analyses; 1 SR2 did not conduct a meta-analysis for a comparison of interest for this report, subsequently that meta-analysis result (bag valve mask versus endotracheal intubation) was not included in this review. Two SRs2,9 had broader inclusion criteria than this report, including other types of airway interventions such as bag valve masks. This review reported on the characteristics and results from the subset of relevant studies.
Summaries of study characteristics are organized by research question and comparison. Additional details regarding the characteristics of the included publications are provided in Appendix 3.
Question 1: Clinical Effectiveness Between Extraglottic Airway Devices
This review included 1 SR2 and 4 observational studies11-14 that compared the clinical effectiveness of i-gel with alternative extraglottic airway devices (laryngeal mask airway and King laryngeal tube).
I-gel Compared With Laryngeal Mask Airway
One SR2 compared i-gel with laryngeal mask airways in patients who had an OHCA requiring airway management. Authors from the US conducted the SR without specifying countries where the relevant primary studies were performed. The SR did not report the types of health care providers inserting the devices.2
I-gel Compared With King Laryngeal Tube
Three retrospective12-14 and 1 prospective11 NRS compared the clinical effectiveness of i-gel and King laryngeal tubes in patients who had an OHCA with 2 studies,13,14 specifically on nontraumatic OHCA. These studies were conducted in Canada,12 US,13,14 and Norway.12 Reported patient mean or median age ranged from 63 to 71 years, with 25% to 36.9% being female.11-14 Airway device insertion was attempted by the emergency medical system personnel11,13,14 or paramedics.12
Clinical outcomes assessed across studies included:
survival (e.g., survival to hospital admission and hospital discharge)2,13,14
survival with a favourable neurologic outcome (defined as a Cerebral Performance Category score of 1 or 2 at discharge)14
end-tidal carbon dioxide (ETCO2) levels13
difficulty of insertion11
complications.11
Question 2: Clinical Effectiveness of Extraglottic Airway Devices Compared With Endotracheal Intubation
This review included 2 SRs with meta-analyses8,9 and 7 primary studies10,15-20 comparing the clinical effectiveness of supraglottic airway devices with endotracheal intubation.
I-gel Compared With Endotracheal Intubation
Six primary studies compared the effectiveness of i-gel with endotracheal intubation, including 1 open, parallel, multicentre, cluster RCT,10 2 prospective observational studies,19,20 and 3 retrospective studies.15,17,18 Four studies focused on patients who had an OHCA,10,15,17,19 while 2 other studies18,20 included more than 85% of patients experiencing OHCA. The mean or median age of patients ranged from 60.1 years to 73 years, with 54% to 71.3% being male.10,15,17-20 Airway device insertion was attempted by paramedics10,18,20 or personnel from the emergency medical system or ambulance team.15,17,19
Laryngeal Mask Airway Compared With Endotracheal Intubation
One SR with meta-analysis compared laryngeal mask airways with tracheal intubation in patients who had an OHCA. The relevant 7 observational studies and 2 RCTs included in the SR were conducted in Japan, England, Taiwan, South Korea, and the US. The mean or median age of patients ranged from 60.8 years to 75.8 years, with 55% to 76% being male. The SR did not specify the types of health care providers inserting the devices.9
Multiple Types of Supraglottic Airway Devices Compared With Endotracheal Intubation
One SR with meta-analysis8 and 1 retrospective observational study16 compared the clinical effectiveness of multiple supraglottic airway devices (i.e., i-gel, laryngeal mask airway, and King laryngeal tube) as a group to tracheal intubation. All 4 RCTs in the SR were relevant to this report, including 1 study on laryngeal mask airway supreme, 1 on King laryngeal tube, and 2 on i-gel. They were conducted in adults aged 64 to 75 years who had an OHCA in the Netherlands, UK, US, and Taiwan. The SR did not specify the type of health care provider(s) who inserted the devices.8 The retrospective study, conducted in the US, included patients with at least 1 advanced airway management attempt by personnel from the emergency medical system. The median age was 65.2 years for adults and 2.3 years for pediatric patients, and 41.4% of adults and 38.9% of pediatric patients were female.9
Clinical outcomes assessed across studies for research question 2 included:
survival outcomes: survival to admission9 and discharge,9,10,17 survival to longest follow-up,8 mortality at 28 days18
ventilator-free days10
hospital length of stay10
functional outcomes: good functional recovery (defined as a modified Rankin Scale score of 0 to 3 or as defined by study authors),8,10 favourable neurologic outcome (defined as Cerebral Performance Category score of 1 to 2),17 poor neurologic outcome (defined as a Cerebral Performance Category score of 3 to 5)15
quality of life (assessed by the EQ-5D-5L scale)10
time-related outcomes: time to advanced airway placement,8 time to successful first attempt,18 and time to successful airway placement18
Question 3: Guidelines Regarding the Use of Extraglottic Airway Devices
This report included 1 evidence-based guideline published in 2024 by the US Agency for Health Care Research and Quality (AHRQ) for emergency medical services clinicians on prehospital airway management.21 They conducted an SR of RCTs and observational studies and rated evidence by quality based on the AHRQ method guide; quality was rated on a scale that ranged from insufficient to high.23,24 Using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) method, an expert panel assessed the strengths of recommendations and reached consensus on final statements.21
Summary of Critical Appraisal
Summaries of critical appraisal are presented, organized by study design. Additional details regarding the strengths and limitations of the included publications are provided in Appendix 4.
Systematic Reviews
All 3 SRs2,8,9 included clear presentation of research questions, study eligibility criteria, as well as funding and conflicts of interest for authors. All SRs2,8,9 also assessed risk of bias in included studies, with 2 SRs2,8 also evaluating the strength or certainty of evidence.
Additional strengths included protocol registration by 2 SRs,2,8 which can improve transparency and may help to reduce selective reporting. One9 of the 2 SRs2,8 reported that while funnel plots to assess publication bias were planned, they were not conducted as fewer than 10 trials were included in the analysis. It is unclear whether Yang et al. developed a protocol in advance.9 Two SRs2,8 performed database and grey literature searches; 1 study8 reported the grey literature search strategy. Yang et al. searched databases but did not report a grey literature search; it is unclear whether the SR identified all relevant studies.9 Duplicate and independent screening of study titles, abstracts, full texts, and data extraction were performed in 2 SRs.8,9 The other SR2 screened full texts in duplicate. It did not consistently screen abstracts by 2 reviewers, and the data extraction method was unclear, increasing the risk of missing relevant studies or errors in data extraction. The 2 SRs8,9 described included studies in detail, while the other SR2 provided limited information.
The 3 SRs2,8,9 had common limitations including the lack of justification for choice of included study designs and sources of funding for included studies. None of the SRs explicitly accounted for risk of bias in the interpretation of findings, which can impact the interpretation of results. Forestell et al. reported no evidence of effect modification on outcomes of interest in subgroup analyses by risk of bias level,8 and Yang et al. acknowledged potential bias in some outcomes.9 Other limitations included the absence of a list of excluded studies8,9 and the missing results of discussion on heterogeneity in review.2,9 Two SRs8,9 conducted meta-analyses; 1 study9 explained choice of the analysis model and assessed publication bias.
Randomized Controlled Trial
The RCT10 clearly reported the study objective, the intervention, participant characteristics, main outcomes including P values, main findings, and funding sources. Paramedics were randomized as clusters to use the intervention or control to treat patients. The intervention and control groups were followed for the same period. Statistical tests were used appropriately, and the main outcome measures were accurate and reliable. The primary outcomes were powered to detect a clinically important difference. The study reported severe adverse events; however, it is unclear if all important adverse events were reported.10
Several factors affected the internal validity of this RCT. Allocation concealment was not conducted during patient enrolment. To reduce selection bias, all eligible patients were automatically enrolled. Blinding was performed in ambulance control room personnel, clinical staff caring for patients beyond the emergency department, researchers assessing the outcomes, and patients (who were unconscious during the intervention) to reduce performance and detection bias. The lack of blinding for paramedics and emergency department staff owing to the nature of the interventional procedure may lead to performance bias. Other limitations included crossover between groups (4 patients in the intervention group and 3 in the control group) and about one-half of the eligible patients not consenting to follow-up, thus reducing the power of the follow-up analyses. Sensitivity analyses (intention-to-treat analysis and as treated) were performed to estimate these effects.10
The study was conducted across 4 ambulance services, which were representative of the treatment most of the patients received. Patients were likely representative of the population from which they were recruited. Researchers acknowledged that participating paramedics were volunteers whose airway skills may not represent paramedics who did not participate in the study. Study findings may not be generalized to patients treated by paramedics with different skill levels. Characteristics of patients lost to follow-up were not reported. The large number of missing data in follow-up reduced the generalizability of findings to patients recovering after OHCA.10
Nonrandomized Studies
All 10 NRSs11-20 clearly described objectives, inclusion criteria, interventions, patient characteristics, main outcomes with random variability estimates, and findings. Eight studies11,12,15-20 reported actual P values for main outcomes; 4 studies11,17,18,20 reported some adverse events.
Methodological limitations in all included NRSs,11-20 such as the lack of randomization and blinding owing to the observational design, increased the risks of selection, performance, and detection bias. Some outcomes such as neurologic outcomes,14,17 successful airway management,12,16,20 and ease of insertion11 were subjectively evaluated and may be influenced by the lack of blinding. One study noted that successful airway management may be overreported.16 Seven12-18 retrospective studies relied on data not specifically collected for them, leading to missing data and low data quality owing to unstandardized collection. All studies followed patients in the intervention and control groups for the same amount of time and used appropriate statistical tests to analyze main outcomes, which were measured using accurate and valid methods.
Regarding internal validity impacted by confounders, 6 studies13,15-17,19,20 recruited patients from the same population during the same period, ensuring consistency in health care received between groups. One study11 recruited different populations, and 3 other studies12,14,18 recruited patients from different periods of time. Eight studies11,12,15-19 did not identify potential confounders, including 2 studies19,20 not describing patient baseline similarities, making it difficult to interpret study findings. Two studies13,14 did not describe characteristics of patients lost to follow-up. While 6 studies12-14,16,17,20 considered confounders in analysis, 412,16,17,20 only adjusted for some. It is unclear whether findings from the other 4 studies11,15,18,19 not assessing confounders may be affected by unmeasured factors (e.g., patient age).
For external validity, all11-14,16-20 but 1 study15 included patients representative of the entire eligible population and treated in settings representative of the treatment received by most patients. One study13 with substantial missing data indicated that the findings may not be generalizable to patients not being followed up.
Four studies13,15,18,20 indicated that they were likely underpowered to detect a significant effect for certain outcomes. Five studies11,12,14,17,19 did not report whether sample size calculations were performed. It is uncertain whether the nonsignificant differences in some outcomes could be owing to insufficient statistical power.
Evidence-Based Guideline
The AHRQ guideline21 clearly described its scope and purpose, including objectives, health questions, and the targeted population. Relevant professional groups and target users were involved in guideline development, though patient views and preferences were not included. It reported a systematic search for evidence, study selection criteria, evidence strengths and limitations, methods for formulating recommendations, and the link between recommendations and supporting evidence. It also considered health benefits, side effects, and risks in formulating the recommendations. The lack of external review and an updating procedure may reduce the rigour of guideline development.21
The guideline provided clear recommendations and options for management of the condition. It described facilitators and barriers to its application, with advice on implementation and resources required. The guideline did not report the monitoring or auditing criteria, which may limit its applicability.21
Regarding editorial independence, the guideline stated that views of the funding body had no influence on its content. It also recorded and addressed competing interests of guideline development members.21
Summary of Findings
Appendix 5 presents the main study findings.
Question 1: Clinical Effectiveness Between Extraglottic Airway Devices
I-gel Compared With King Laryngeal Tube
Overall, patients who had an OHCA and received i-gels tended to have better outcomes compared with King laryngeal tubes. Outcomes where i-gels were associated with statistically significantly better outcomes included:
higher rates of successful device insertion (e.g., higher proportion of patients who had first-pass success, fewer attempts required for successful insertion, fewer patients with failed insertion; 3 NRSs11-13). One NRS11 reported the proportion of patients who had a successful insertion after 3 attempts from the same personnel or attempts from 2 or more personnel did not differ statistically significantly between groups
higher survival to discharge at home (2 NRSs13,14), hospital admission, hospital discharge, and discharge with good neurologic outcomes (1 NRS;14 tools used to measure neurologic outcomes are presented as footnotes in Appendix 5)
fewer patients with low levels of ETCO2 (used as an indirect measure of ventilation success; low ETCO2 levels indicate ventilation failure) (1 NRS13)
reported ease of device insertion: i-gels were more likely to be reported as easy to insert, while King laryngeal tubes were more likely to be reported as medium-to-very difficult when inserting (1 NRS11).
Results were mixed for rearrest: 1 NRS13 reported statistically significantly lower odds of rearrest for patients treated with i-gels compared with King laryngeal tubes, while another NRS14 did not find a statistically significant difference.
One NRS11 reported that the number of reported complications generally did not differ statistically significantly between groups, excluding anatomic conditions, problematic insertion, and insertion taking longer than 30 seconds, all of which were higher in the King laryngeal tube group.
I-gels Compared With Laryngeal Mask Airways
The SR2 overall reported no significant differences for patients who had an OHCA and received i-gels compared with laryngeal mask airways on first-pass success, survival, and ROSC. They identified 1 RCT that reported i-gels were associated with higher proportions of overall successful insertion (90% versus 57%).
King Laryngeal Tubes Compared With Laryngeal Mask Airways
This review did not identify any studies that compared King laryngeal tubes with laryngeal mask airways that met the inclusion criteria for this report; therefore, no summary can be provided.
Question 2: Clinical Effectiveness of Extraglottic Airway Devices Compared With Endotracheal Intubation
I-gels Compared With Endotracheal Intubation
Most included studies were restricted to patients who experienced OHCA; 2 studies18,20 included patients with noncardiac emergencies, though for both studies, more than 85% of patients had cardiac arrest. Across these studies, i-gels were associated with better outcomes than endotracheal intubation on a few outcomes:
first-pass success (1 RCT10 and 1 NRS18); however, 1 NRS20 also reported similar rates between groups
overall success at device insertion (1 RCT10 and 2 NRSs18,20)
shorter time to device insertion (1 RCT from 1 SR8 and 1 NRS18) and shorter time to first attempt (1 NRS18).
Mixed results were found for:
ROSC: 2 RCTs8,10 (including 1 from the SR8) reported that patients in the i-gel group were more likely to experience ROSC, while 1 RCT (from the SR8) and 3 NRSs17-19 reported no statistically significant difference between groups.
favourable neurologic outcome: 1 RCT10 and 2 NRSs15,17 reported no statistically significant difference (tools used to measure neurologic outcomes are presented as footnotes in Appendix 5). The RCT10 reported that more patients in the i-gel group had better functional outcomes from 1 sensitivity analysis (excluded patients who did not receive i-gel or endotracheal intubation), and 2 subgroup analyses:
restricted to non-Utstein patients (Utstein patients are those whose OHCA was likely owing to a cardiac cause, is witnessed, and has an initial rhythm amenable to defibrillation)
restricted to patients who had ventilation success within first 2 attempts.
No statistically significant difference was found for:
survival (2 RCTs8,10 including 1 from the SR,8 and 2 NRSs17,18)
length of stay in the hospital (1 RCT10) or intensive care unit (1 RCT10 and 1 NRS18) or number of days that were ventilator-free (1 NRS18)
quality of life at 30 days or hospital discharge, 3 months, or 6 months (1 RCT;10 tools used to measure quality of life are presented as footnotes in Appendix 5)
Laryngeal Mask Airways Compared With Endotracheal Intubation
One SR with meta-analysis9 reported that based on 5 studies of patients who had an OHCA, survival to admission was statistically significantly higher for patients who received endotracheal intubation. Survival to discharge was also slightly higher in the endotracheal intubation group but this was not statistically significant, based on a pooled analysis of 8 studies.
Findings for ROSC were mixed: 1 RCT (identified by 1 SR8) reported no statistically significant difference on sustained ROSC, though it was slightly higher for the patients who received laryngeal mask airways. However, 1 SR with meta-analysis9 reported that based on their pooled analysis of 7 studies, endotracheal intubation was associated with statistically significantly higher ROSC rates.
King Laryngeal Tube Compared With Endotracheal Intubation
One RCT identified by 1 SR8 reported that patients who had an OHCA who received King laryngeal tubes tended to have higher rates of ROSC and shorter time to device insertion. Survival was also slightly higher, though this was not statistically significant, for patients in the King laryngeal tube group.
Multiple Types of Supraglottic Airway Devices Compared With Endotracheal Intubation
Publications that grouped together several types of supraglottic airway devices included 1 NRS16 and 1 SR with meta-analysis8 that pooled multiple studies. The individual studies from the meta-analysis8 have been reported individually by specific type of device previously, and their pooled results are not summarized here. Details about the pooled results from this meta-analysis are available in Appendix 5 (Table 19, Table 20, and Table 21).
The NRS16 included adult and pediatric patients who had an OHCA as well as other indications and reported better outcomes for patients who received supraglottic airway devices compared with endotracheal intubation. They also reported that patients who received supraglottic airway devices tended to have higher rates of first-pass success compared with patients who received endotracheal intubation. This difference was larger for pediatric patients compared with adult patients.
Question 3: Guidelines Regarding the Use of Extraglottic Airway Devices
The guideline by AHRQ21 provided several recommendations regarding the use of supraglottic airway devices for patients experiencing out-of-hospital emergencies. All recommendations were conditional, with the certainty of evidence reported as very low or low to moderate owing to limited data, particularly on patient outcomes. These recommendations are summarized in Table 2.
Table 2: Summary of Recommendations Regarding the Use of Extraglottic Airway Devices
Condition | Adult patient | Pediatric patient | ||
|---|---|---|---|---|
SGA or BVM | SGA or ETI | SGA or BVM | SGA or ETI | |
OHCA | SGA or BVM can be used | In systems with demonstrated high ETI proficiency, suggest either SGA or ETI. In systems without demonstrated high ETI proficiency, suggest SGA compared with ETI | SGA or BVM can be used | Recommend SGA compared with ETI |
Trauma | SGA or BVM can be used | SGA or ETI can be used | No recommendation owing to missing relevant studies | Recommend SGA compared with ETI |
Medical emergencies | No recommendation owing to missing relevant studies | In systems with demonstrated high ETI proficiency, suggest either SGA or ETI. In systems without demonstrated high ETI proficiency, suggest SGA compared with ETI | No recommendation owing to missing relevant studies | Recommend SGA compared with ETI |
BVM = bag valve mask; ETI = endotracheal intubation; OHCA = out-of-hospital cardiac arrest; SGA = supraglottic airway devices.
Notes:
High ETI proficiency appeared to be defined by the guideline as having documentation of high intubation first-pass success rates. The guideline authors noted that the literature did not have an established definition for high first-pass success rate (i.e., what percentage would be considered high).
SGA was favoured for pediatric patients owing to factors such as higher first-pass success rates, harms associated with failed ETI attempts, and procedure rarity. Additional details regarding factors considered for each recommendation are available in Appendix 5.
For patients who had an OHCA (adult or pediatric) or adults who have experienced trauma, the guideline recommended supraglottic airway devices or bag valve masks, based on little evidence to indicate if one was superior.21 The guideline also noted that bag valve mask ventilation often required more clinicians, indicating resource availability might influence treatment choice. The guideline did not provide recommendations regarding the use of bag valve mask or supraglottic airway devices for pediatric patients who have experienced trauma or patients of any age with medical emergencies owing to lack of evidence.
For adults who had an OHCA or medical emergency, the guideline suggested that choosing between supraglottic airway devices and endotracheal intubation should be influenced by documented success with endotracheal intubation:21
for systems with demonstrated high endotracheal intubation proficiency, use either supraglottic airway devices or endotracheal intubation
for systems without demonstrated high endotracheal intubation proficiency, use supraglottic airway devices compared with endotracheal intubation.
For pediatric patients who had an OHCA, experienced trauma, or experienced a medical emergency, they suggested using supraglottic airway devices compared with endotracheal intubation.21
A summary of recommendations from nonevidence-based guidelines is available in Appendix 7.
Limitations
Risk of Bias of Included Studies in Systematic Reviews
Of the included studies of interest in the 3 SRs, as assessed by the review authors, 8 studies from 3 SRs were at low risk of bias; however, the other 7 studies were either at high risk of bias or had concerns about the relevant selection, performance, and detection bias as well as comparability.2,8,9
External Validity
Most included studies were restricted to patients who had experienced an OHCA, including all studies that compared extraglottic airway devices. Some studies also had other restriction criteria that may limit their generalizability. For example, Smida et al.14 was restricted to patients with successful advanced airway management, and Edwards et al.15 only included patients who were transferred to a specialized heart attack centre. It is unclear if these findings are generalizable to a wider population, as well as to patients who required advanced airway management for a different reason. It is also unclear if these findings are generalizable to patients in Canada, as only 1 NRS12 comparing i-gel with King laryngeal tube was conducted in Canada.
Evidence Gaps
Most studies were restricted to adults only; while a few studies did not state they were restricted to adults, most tended to have a higher mean or median age, suggesting their patients were likely primarily adults or older adults. Only 1 study16 provided a subgroup analysis of pediatric patients. This study also grouped together multiple types of extraglottic devices compared with endotracheal intubation, and did not report results by specific type of device (e.g., i-gels compared with endotracheal intubation). This review did not identify any studies that compared different supraglottic airway devices in a pediatric population.
This review identified 1 evidence-based guideline21 that provided recommendations regarding the general use of supraglottic airway devices, including situations where they are preferred compared with endotracheal intubation. This guideline did not provide recommendations regarding the use of specific types of extraglottic airway devices. They also were unable to provide recommendations for specific populations owing to a lack of evidence.
This review identified few studies comparing i-gels with laryngeal mask airways (1 SR with 2 RCTs)2 and King laryngeal tubes with endotracheal intubation (1 SR with 1 RCT).8 Few studies reported on adverse events, which were limited to studies that compared i-gels with King laryngeal tubes,11 and studies that compared i-gels with endotracheal intubation.10,17,18,20 Without a clear understanding of the type and frequency of adverse events an intervention is associated with, decision-makers may overestimate the benefits and underestimate the risks of an intervention to patients. This review did not identify any publications that compared laryngeal mask airways with King laryngeal tubes.
Complexity of Assessing Airway Management in Out-of-Hospital Emergencies
It is a challenge to assess the effectiveness of different airway management devices outside of controlled settings, as outcomes are also affected by factors such as provider experience and training and cause of respiratory distress that can be hard to control for.6 Well-conducted RCTs can help to address the issue of confounders yet are also difficult to conduct well. Most studies included in this report were NRSs, and their findings may have been influenced by confounding variables.
Conclusions and Implications for Decision- or Policy-Making
Summary of Evidence
This rapid review evaluated the literature regarding the clinical effectiveness of different types of extraglottic airway devices, as well as extraglottic airway devices compared with endotracheal intubation, for patients experiencing out-of-hospital emergencies, and evidence-based guidelines regarding the use of extraglottic airway devices. This review identified:
1 SR2 and 4 NRSs11-14 that compared different types of extraglottic devices
2 SRs,8,9 1 RCT,10 and 6 NRSs15-20 that compared an extraglottic airway device with endotracheal intubation
1 evidence-based guideline21 regarding the use of extraglottic airway devices for this patient population.
All the studies that compared extraglottic airway devices were focused on patients who experienced OHCA. Studies that compared i-gels to King laryngeal tubes tended to report i-gels were associated with higher rates of successful device insertion11-13 and survival,13,14 including survival with good neurologic outcomes.14 Only 1 NRS11 reported on adverse events for this comparison and found similar rates of complications between groups, though a few complications occurred more frequently in the King laryngeal tube group. Two NRSs13,14 reported on rearrest and had mixed findings. One SR2 reported 2 RCTs that compared i-gels with laryngeal mask airways: they reported no significant differences between groups, except successful insertion, which was higher in the i-gel group.
Most studies that compared extraglottic airway devices with endotracheal intubation were restricted to patients who experienced OHCA or primarily included this patient group. I-gels were associated with higher or similar rates of successful insertion, when compared with endotracheal intubation;15,18,20 no statistically significant difference was found on survival,8,10,17,18 hospital length of stay,10,18 quality of life,10 or adverse events.10,17,18 Results were mixed for ROSC8,10,17-19 and neurologic outcomes.10,15,17 The RCT10 noted i-gels were more likely to be associated with favourable neurologic outcomes if the analysis was restricted to certain subgroups. This may indicate that i-gels are more clinically effective than endotracheal intubation for specific patient populations, but further studies are needed to assess this.
One SR with meta-analysis9 reported that compared with endotracheal intubation, laryngeal mask airways were associated with improved survival to admission, though it may not be associated with a statistically significantly improved survival to discharge. Results for ROSC were mixed.8,9 One RCT was identified by 1 SR8 and found higher rates of ROSC and shorter time to device insertion for patients who received King laryngeal tubes compared with patients who received endotracheal intubation; survival was also higher for this group, but it was not statistically significant.
One NRS16 included patients who required airway management for cardiac arrest as well as patients who had not had a cardiac arrest and included pediatric and adult patients. Their analyses indicated that supraglottic airway devices in general (which included i-gels, King laryngeal tubes, and laryngeal mask airways) were associated with higher rates of first-pass success than endotracheal intubation, with a larger benefit seen in pediatric patients.
In summary, the identified publications suggest that using i-gels may lead to improved outcomes when compared with King laryngeal tubes. I-gels were preferred than laryngeal mask airways and endotracheal intubation on successful device insertion; however, other outcomes were either not statistically significantly different or had mixed findings. Limited evidence suggests that laryngeal mask airways and King laryngeal tubes may lead to improved or similar outcomes when compared with endotracheal intubation. The identified studies were primarily in adults who had an OHCA, and their generalizability to other populations is unclear.
The identified guideline21 recommended using a supraglottic airway device for airway management in adults or pediatric patients who had an OHCA, and for adults who have experienced trauma. They suggested supraglottic airway devices or endotracheal intubation can be used for adults who have experienced trauma, but recommended supraglottic airway devices compared with endotracheal intubation if endotracheal intubation proficiency has not been demonstrated by adults who had an OHCA or medical emergency. They also recommended supraglottic airway devices compared with endotracheal intubation for pediatric patients who had an OHCA, have experienced trauma, or have had medical emergencies.21 The guideline also noted that when studies on patient-oriented outcomes were unavailable, they considered studies on outcomes like procedure success or first-pass success, owing to the harms associated with failed device insertion.21
Considerations for Future Research
Additional high-quality studies with large sample sizes that assess the effectiveness of extraglottic airway devices for patients with other types of emergencies (i.e., other than cardiac arrest), as well as for pediatric patients, may help to develop our understanding of what airway management strategies are appropriate for specific patient populations. Studies that assess patient-important outcomes like survival, quality of life, and adverse events would also be beneficial.
Considerations for Decision- or Policy-Making
In addition to considering clinical effectiveness, health care decision-makers may consider other factors related to the use of different airway management strategies. For example, endotracheal intubation is a complex procedure that requires extensive training and experience, as well as more time for application.19 Supraglottic airway devices are comparatively easier to use, require less training, and are safe for patients. In settings where it is difficult for emergency medical services to obtain the experience required to maintain their skills in endotracheal intubation in this population (e.g., rural areas where a paramedic may not need to provide advanced airway management for more than a year),12 supraglottic airway devices may be safer.21 However, there may be some patients who cannot experience effective ventilation with a supraglottic airway device, and require endotracheal intubation for ventilation.10 Considering specific patient factors when deciding which strategy to use may help to optimize patient outcomes. Decision-makers may also want to consider the barriers and facilitators for accessing training for different management strategies and devices.
References
1.Huitink JM, Bretschneider JH. Airway Management Academy: A global initiative to increase patient safety during airway management by medical education. Trends in Anaesthesia and Critical Care. 2015;5(1):42-47.
2.Carney N, Cheney T, Totten A, et al. Prehospital Airway Management: A Systematic Review. Comparative Effectiveness Review No. 243. (Prepared by the Pacific Northwest Evidence-based Practice Center under Contract No. 290-2015-00009-I.). Rockville (MD): Agency for Healthcare Research and Quality; 2021: https://effectivehealthcare.ahrq.gov/products/prehospital-airway-management/research. Accessed 2024 Aug 14.
3.Braude D, Steuerwald M, Wray T, Galgon R. Managing the Out-of-Hospital Extraglottic Airway Device. Ann Emerg Med. 2019;74(3):416-422. PubMed
4.Laurin EG, Wolfson B, Grayzel J. Extraglottic devices for emergency airway management in adults. In: Post TW, ed. UpToDate. Waltham (MA): UpToDate; 2024: https://www.uptodate.com.
5.Doyle DJ, Hagberg CA, Crowley M. Supraglottic airways (SGAs) for airway management for anesthesia in adults. In: Post TW, ed. UpToDate. Waltham (MA): UpToDate; 2024: https://www.uptodate.com.
6.Wang HE, Benger JR. Endotracheal intubation during out-of-hospital cardiac arrest: New insights from recent clinical trials. J Am Coll Emerg Physicians Open. 2020;1(1):24-29. PubMed
7.Stuby L, Jampen L, Sierro J, et al. Effect of Early Supraglottic Airway Device Insertion on Chest Compression Fraction during Simulated Out-of-Hospital Cardiac Arrest: Randomised Controlled Trial. J Clin Med. 2022;11(1):217. PubMed
8.Forestell B, Ramsden S, Sharif S, et al. Supraglottic Airway Versus Tracheal Intubation for Airway Management in Out-of-Hospital Cardiac Arrest: A Systematic Review, Meta-Analysis, and Trial Sequential Analysis of Randomized Controlled Trials. Crit Care Med. 2024;52(2):e89-e99. PubMed
9.Yang Z, Liang H, Li J, et al. Comparing the efficacy of bag-valve mask, endotracheal intubation, and laryngeal mask airway for subjects with out-of-hospital cardiac arrest: an indirect meta-analysis. Ann. 2019;7(12):257. PubMed
10.Benger JR, Kirby K, Black S, et al. Supraglottic airway device versus tracheal intubation in the initial airway management of out-of-hospital cardiac arrest: the AIRWAYS-2 cluster RCT. Health Technol Assess. 2022;26(21):1-158. PubMed
11.Lonvik MP, Elden OE, Lunde MJ, Nordseth T, Bakkelund KE, Uleberg O. A prospective observational study comparing two supraglottic airway devices in out-of-hospital cardiac arrest. BMC Emerg Med. 2021;21(1):51. PubMed
12.Price P, Laurie A, Plant E, Chandra K, Pishe T, Brunt K. Comparing the First-Pass Success Rate of the King LTS-D and the i-gel Airway Devices in Out-of-Hospital Cardiac Arrest. Cureus. 2022;14(11):e30987. PubMed
13.Smida T, Menegazzi J, Crowe R, Scheidler J, Salcido D, Bardes J. A Retrospective Nationwide Comparison of the iGel and King Laryngeal Tube Supraglottic Airways for Out-of-Hospital Cardiac Arrest Resuscitation. Prehosp Emerg Care. 2024;28(2):193-199. PubMed
14.Smida T, Menegazzi J, Scheidler J, Martin PS, Salcido D, Bardes J. A retrospective comparison of the King Laryngeal Tube and iGel supraglottic airway devices: A study for the CARES surveillance group. Resuscitation. 2023;188:109812. PubMed
15.Edwards T, Williams J, Cottee M. Influence of prehospital airway management on neurological outcome in patients transferred to a heart attack centre following out-of-hospital cardiac arrest. Emerg Med Australas. 2019;31(1):76-82. PubMed
16.Jarvis JL, Wampler D, Wang HE. Association of patient age with first pass success in out-of-hospital advanced airway management. Resuscitation. 2019;141:136-143. PubMed
17.Kim TY, Kim S, Han SI, et al. Gastric Inflation in Prehospital Cardiopulmonary Resuscitation: Aspiration Pneumonia and Resuscitation Outcomes. Rev Cardiovasc Med. 2023;24(7):198. PubMed
18.Levi D, Hoogendoorn J, Samuels S, et al. The i-gel((R)) supraglottic airway device compared to endotracheal intubation as the initial prehospital advanced airway device: A natural experiment during the COVID-19 pandemic. J Am Coll Emerg Physicians Open. 2024;5(2):e13150. PubMed
19.Matic I, Pajic Matic I, Marcikic M, Jurjevic M, Zanko B, Dosen I. Comparison of Different out-of-Hospital Airway Management Techniques in Patients with Cardiac Arrest in Slavonia Region. Acta Clin. 2021;60(4):590-594. PubMed
20.Nichols M, Fouche PF, McPherson T, Evens T, Bendall J. Lessons from the first two years of a new out-of-hospital airway registry in New South Wales. Paramedicine. 2023;20(5):152-160.
21.Jarvis JL, Panchal AR, Lyng JW, et al. Evidence-Based Guideline for Prehospital Airway Management. Prehosp Emerg Care. 2024;28(4):545-557. PubMed
22.Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol. 2009;62(10):e1-e34. PubMed
23.Berkman ND, Lohr KN, Ansari M, et al. AHRQ Methods for Effective Health Care Grading the Strength of a Body of Evidence When Assessing Health Care Interventions for the Effective Health Care Program of the Agency for Healthcare Research and Quality: An Update. Methods Guide for Effectiveness and Comparative Effectiveness Reviews. Rockville (MD): Agency for Healthcare Research and Quality (US); 2008.
24.Gage CB, Powell JR, Bosson N, et al. Evidence-Based Guidelines for Prehospital Airway Management: Methods and Resources Document. Prehosp Emerg Care. 2024;28(4):561-567. PubMed
25.Shea BJ, Reeves BC, Wells G, et al. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ. 2017;358:j4008. PubMed
26.Downs SH, Black N. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J Epidemiol Community Health. 1998;52(6):377-384. PubMed
27.Agree Next Steps Consortium. The AGREE II Instrument. Hamilton (ON): AGREE Enterprise; 2017: https://www.agreetrust.org/wp-content/uploads/2017/12/AGREE-II-Users-Manual-and-23-item-Instrument-2009-Update-2017.pdf. Accessed 2024 Aug 23.
28.Grossestreuer AV, Abella BS, Sheak KR, et al. Inter-rater reliability of post-arrest cerebral performance category (CPC) scores. Resuscitation. 2016;109:21-24. PubMed
29.Broderick JP, Adeoye O, Elm J. Evolution of the Modified Rankin Scale and Its Use in Future Stroke Trials. Stroke. 2017;48(7):2007-2012. PubMed
30.van Hout B, Janssen MF, Feng YS, et al. Interim scoring for the EQ-5D-5L: mapping the EQ-5D-5L to EQ-5D-3L value sets. Value Health. 2012;15(5):708-715. PubMed
31.Schauer SG, Tapia AD, Jeschke EA, et al. Expert Consensus Panel Recommendations for Selection of the Optimal Supraglottic Airway Device for Inclusion to the Medic's Aid Bag. Med J (Ft Sam Houst Tex). 2023(Per 23-1/2/3):97-102.
32.Australian and New Zealand Committee on Resuscitation. Guideline 11.6 – Equipment and Techniques in Adult Advanced Life Support. ANZCOR; 2024: https://www.anzcor.org/assets/anzcor-guidelines/guideline-11-6-equipment-and-techniques-in-adult-advanced-life-support-248.pdf. Accessed 2024 Aug 14.
33.Carlson JN, Colella MR, Daya MR, et al. Prehospital Cardiac Arrest Airway Management: An NAEMSP Position Statement and Resource Document. Prehosp Emerg Care. 2022;26:54-63. PubMed
34.Lyng JW, Baldino KT, Braude D, et al. Prehospital Supraglottic Airways: An NAEMSP Position Statement and Resource Document. Prehosp Emerg Care. 2022;26(sup1):32-41.
35.National Association of State Emergency Medical Services Officials. Model Clinical Guideline: Prehospital Airway Management. Falls Church (VA): NASEMO; 2023: https://nasemso.org/wp-content/uploads/Model-Clinical-Guideline-Prehospital-Airway-Management.pdf. Accessed 2024 Aug 14.
36.Callaway CW, Soar J, Aibiki M, et al. Part 4: Advanced Life Support: 2015 International Consensus on Cardiopulmonary Resuscitation and Emergency Cardiovascular Care Science With Treatment Recommendations. Circulation. 2015;132(16 Suppl 1):S84-145. PubMed
Appendix 1: Detailed Methods and Selection of Included Studies
Please note that this appendix has not been copy-edited.
Literature Search Methods
An information specialist conducted a literature search on key resources including MEDLINE, the Cochrane Database of Systematic Reviews, the International HTA Database, the websites of Canadian and major international health technology agencies, as well as a focused internet search. The search approach was customized to retrieve a limited set of results, balancing comprehensiveness with relevancy. The search strategy comprised both controlled vocabulary, such as the National Library of Medicine’s MeSH (Medical Subject Headings), and keywords. Search concepts were developed based on the elements of the research questions and selection criteria. The main search concepts were extraglottic airway devices and the prehospital setting. The search was completed on August 8, 2024, and limited to English-language documents published since January 1, 2019.
Selection Criteria and Methods
Two reviewers independently screened citations and selected studies, with 1 reviewer required to include or exclude a study. In the first level of screening, titles and abstracts were reviewed and potentially relevant articles were retrieved and assessed for inclusion. The final selection of full-text articles was based on the inclusion criteria presented in Table 1.
Exclusion Criteria
Articles were excluded if they met any of the following criteria:
did not meet the selection criteria outlined in Table 1
were duplicate publications
were published before 2019
were systematic reviews in which all relevant studies were captured in other more recent or more comprehensive systematic reviews
were primary studies captured in 1 or more included systematic reviews
were studies on airway devices during in-hospital, or nonemergency or scheduled surgeries and procedures
were studies on intubating laryngeal mask airways (e.g., laryngeal mask airway Fastrach, Air-Q).
Guidelines with unclear or alternative methodology were not included in the text, but their characteristics and findings are presented in Appendix 1.
Critical Appraisal of Individual Studies
Two reviewers conducted critical appraisal. The included publications were critically appraised by 1 reviewer using the following tools as a guide: A Measurement Tool to Assess Systematic Reviews 2 (AMSTAR 2)25 for systematic reviews, the Downs and Black checklist26 for randomized and nonrandomized studies, and the Appraisal of Guidelines for Research and Evaluation (AGREE) II instrument27 for guidelines. Summary scores were not calculated for the included studies; rather, the strengths and limitations of each included publication were described narratively.
Appendix 2: Selection of Included Studies
Please note that this appendix has not been copy-edited.
Appendix 3: Characteristics of Included Publications
Please note that this appendix has not been copy-edited.
Table 3: Characteristics of Included Systematic Reviews
Study citation, country, funding source | Study designs and numbers of primary studies included | Population characteristics | Intervention and comparator(s) | Clinical outcomes, length of follow-up |
|---|---|---|---|---|
Question 1: comparison between supraglottic airway devices | ||||
Carney et al. (2021)2 US Funding source: National Highway Traffic Safety Administration Office of Emergency Medical Services | Study designs included: RCTs, prospective and retrospective comparative studies, case-control studies. Number of studies included: 99 total (22 RCTs, 77 NRSs); 2 (both RCTs) relevant to present review | Eligible patients included any patients requiring prehospital ventilatory support or airway protection. In the 2 studies of interest to this report, both included adult patients who had a cardiac arrest (N = 454). | Eligible interventions and comparators: Comparisons between any airway intervention (bag valve mask, SGAs, ETI) Relevant intervention: i-gel Relevant comparator: LMA | Reported outcomes:
Follow-up: NR |
Question 2: supraglottic airway devices vs. endotracheal intubation | ||||
Forestell et al. (2024)8 Canada Funding source: Authors’ funding sources include the National Institutes of Health, the Centers for Disease Control and Prevention, Abiomed Inc., OLL Medical Corporation, Vapotherm Inc., ZOLL Circulation Inc., CPR Therapeutics Inc., Heartbeam Inc., Invero Health LLC, Kestra Medical Technologies Inc., and Orixha Inc. | Study designs included: RCTs. Number of studies included: 4 total, all relevant to present review. | Eligible patients included adult patients (18 years or older) who had an OHCA who were randomized to either SGA or tracheal intubation.
| Interventions: any SGA, including i-gel (2 RCTs), LMA supreme (1 RCT), and King LT (1 RCT) Comparator: Tracheal intubation | Outcomes:
Follow-up: used longest follow-up reported |
Yang et al. (2019)9 China Funding source: Science and Technology Foundation of Guangdong Province, China, the Research Program for Colleges and Universities in Guangzhou. | Study designs included: Comparative studies. Number of studies included: 13 total, 9 relevant to this report (2 RCTs, 7 NRSs). | Eligible patients included adult patients who had an OHCA. Across the studies relevant to this report, for the LMA or ETI groups, and where reported:
| Eligible interventions and comparators: Comparisons between BVM, ETI, and LMA Relevant comparison: LMA vs. ETI | Outcomes:
Follow-up: NR |
BVM = bag valve mask; ETI = endotracheal intubation; LMA = laryngeal mask airway; LT = laryngeal tube; NR = not reported; NRS = nonrandomized study; OHCA = out-of-hospital cardiac arrest; RCT = randomized controlled trial; ROSC = return of spontaneous circulation; SGA = supraglottic airway; vs. = versus.
Table 4: Characteristics of Included Primary Clinical Studies
Study citation, country, funding source | Study design | Population characteristics | Intervention and comparator(s) | Clinical outcomes, length of follow-up |
|---|---|---|---|---|
Question 1: comparison between supraglottic airway devices | ||||
Smida et al. (2024)13 US Funding source: Authors reported receiving no financial support for this article | Retrospective observational study | Adults (older than 18 years) who had a nontraumatic OHCA between 2018 and 2021, and airway insertion was attempted by EMS (N = 93,866; i-gel, n = 54,189; King laryngeal tube: n = 39,677). Data were retrieved from the ESO Data Collaborative public use research datasets.
| Intervention: i-gel Comparator: King LT | Outcomes:
Follow-up: NR |
Smida et al. (2023)14 US Funding source: One author reported receiving salary from the West Virginia Clinical and Translational Science Institute. | Retrospective observational study | Patients who had nontraumatic OHCA between 2013 and 2021, and airway insertion was successfully attempted by EMS (N = 93,866). Data were retrieved from the Cardiac Arrest Registry to Enhance Survival public use research dataset. I-gel (n = 41,281)
King LT (n = 126,623)
| Intervention: i-gel Comparator: King LT | Outcomes:
Follow-up: NR |
Price et al. (2022)12 Canada Funding source: Authors reported receiving no financial support for this article | Retrospective comparative cross-sectional design; | Adults (older than 18 years) who had an OHCA between February 1, 2015, and September 30, 2020, and airway insertion was attempted by paramedics (N = 2,680; King laryngeal tube: n = 1,290; i-gel: n = 1,390).
| Intervention: King LT Comparator: i-gel | Outcomes:
Follow-up: NR |
Lønvik et al. (2021)11 Norway Funding source: Authors reported receiving no external funding | Prospective observational study | Adults (older than 18 years) who had an OHCA from March 2016, to February 2017, and had a supraglottic airway device inserted by EMS (N = 250; i-gel: n = 191; King LT: n = 59)
| Intervention: King LT Comparator: i-gel | Outcomes:
Follow-up: NR |
Question 2: supraglottic airway devices vs. endotracheal intubation | ||||
Levi et al. (2024)18 US Funding source: Authors reported receiving no specific funding for this work. | Retrospective cohort with historical control | Adults (older than 18 years) who had a nontraumatic OHCA or respiratory arrest or need for airway support who were treated by a paramedic (N = 199; i-gel: n = 116; ETI: n = 83)
| Intervention: i-gel Comparator: ETI | Outcomes:
Follow-up: NR |
Kim et al. (2023)17 South Korea Funding source: Authors reported receiving no external funding for this work. They acknowledged support from Institute of Convergence Science (ICONS), Yonsei University. | Retrospective, multicentre observational study involving 2 hospitals | Patients included adults (older than 19 years) who had a nontraumatic OHCA, had been admitted to the emergency department, and undergone a chest or abdominal radiography from December 2015, to December 2020 (N = 499; SGA, n = 334; ETI, n = 8). Advance airway management was performed by EMTs. Baseline characteristics were provided across all patients, divided by those who had GI (n = 284) and those who did not have GI (n = 215):
| Eligible interventions: BVM, SGA (i-gel), ETI Main comparator: Chest compression only Intervention of interest: i-gel Comparator of interest: ETI | Outcomes:
Follow-up: NR |
Nichols et al. (2023)20 Australia Funding source: Authors reported receiving no funding for this work. | Review of prospectively collected registry data | Patients who received an advanced airway attempt by an intensive care paramedic with ETI or i-gel from August 8, 2020, to August 8, 2022 (N = 872; i-gel, n = 222; ETI, n = 705). Baseline characteristics (note: some patients had missing data)
| Intervention: i-gel Comparator: ETI | Outcomes:
Follow-up: NR |
Benger et al. (2022)10 UK Funding source: funded by the NIHR Health Technology Assessment program with support from the NIHR Comprehensive Research Networks | Open, parallel, 2-group, multicentre, cluster randomized controlled trial. Paramedics were randomized to i-gel or ETI | Adult patients (18 years or older) with a nontraumatic OHCA and were attended by a participating paramedic (N = 9,296; i-gel n = 4,886; ETI n = 4,410).
| Intervention: i-gel Comparator: ETI | Outcomes:
Follow-up: 6 months |
Matić et al. (2021)19 Croatia Funding source: Emergency Medicine Institute of the Brod-Posavina County and Dr. Josip Benčević General Hospital | Prospective cohort study | Patients who had an OHCA from October 1, 2014, to August 1, 2016, and were attended by the emergency ambulance team within 20 minutes and received some form of CPR (N = 92; i-gel, n = 45; ETI, n = 47).
| Intervention: i-gel Comparator: ETI | Outcome: ROSC Follow-up: NR |
Edwards et al. (2019)15 UK Funding source: NR; authors acknowledge assistance from clinicians from participating Heart Attack Centres and staff of the London Ambulance Service NHS Trust | Retrospective cohort study based on ambulance records | Adult patients (18 years or older) who had an OHCA and experienced ROSC where the resuscitation attempt involved active airway management and ventilation, and the patient was transferred directly to a specialist heart attack centre. Originally 220 patients included, with complete data outcome for 95% baseline statistics based on N = 209 (i-gel, n = 152; ETI, n = 57)
| Intervention: i-gel Comparator: ETI | Outcome: poor neurologic outcome, defined as a CPC score of 3 to 5 Follow-up: NR |
Jarvis et al. (2019)16 US Funding source: None | Retrospective review of electronic health records and records of emergency calls | Patients (99 years or younger) who had at least 1 advanced airway management attempt (N = 29,368; ETI: n = 22,519; SGA: n = 6,849) Adult (aged 15 to 99 years) characteristics:
Pediatric (aged 14 years or younger) characteristics:
| Intervention: SGA (i-gel, King laryngeal tube, laryngeal mask airway) Comparator: ETI | Outcome: First-pass success Follow-up: NR |
BVM = bag valve mask; CPC = cerebral performance category; CPR = cardiopulmonary resuscitation; ED = emergency department; EMT = emergency medical technicians; EMS = emergency medical system; EQ-5D-5L = EuroQol 5-Dimension 5-level; ETCO2 = end-tidal carbon dioxide; ETI = endotracheal intubation; GI = gastric inflation; IQR = interquartile range; LT = laryngeal tube; NHS = National Health Service; NIHR = National Institute for Health and Care Research; NR = not reported; OHCA = out-of-hospital cardiac arrest; SD = standard deviation; SGA = supraglottic airway devices; ROSC = return of spontaneous circulation; vs. = versus.
Table 5: Characteristics of Included Guideline
Intended users, target population | Intervention and practice considered | Major outcomes considered | Evidence collection, selection, and synthesis | Evidence quality assessment | Recommendations development and evaluation | Guideline validation |
|---|---|---|---|---|---|---|
AHRQ (2024)21 | ||||||
Intended users: EMS clinicians Target population: Patients requiring airway management in the prehospital setting | Eligible interventions:
Practice considered: prehospital setting |
| A SR was conducted, with 2 reviewers independently screening (title abstract and full text). Data were synthesized separately for each question, with outcomes pooled where appropriate; if pooling was inappropriate, findings were summarized qualitatively. | The strength of evidence for each question was indicated as low, moderate, high, or insufficient based on the AHRQ Methods Guide. | Expert panel generated practice recommendations based on SR and GRADE methodology. Final recommendations were developed via facilitated panel discussions, where a consensus was defined as agreement of 85% of more of panel members. | NR |
AHRQ = Agency for Health Care Research and Quality; EMS = emergency medical services; GRADE = Grading of Recommendations Assessment, Development, and Evaluation; NR = not reported; SR = systematic review.
Appendix 4: Critical Appraisal of Included Publications
Please note that this appendix has not been copy-edited.
Table 6: Strengths and Limitations of Systematic Reviews Using AMSTAR 225
Strengths | Limitations |
|---|---|
Forestell et al. (2024)8 | |
Presented population, intervention, control, and outcomes of interest. Protocol was registered on PROSPERO. Used a comprehensive search strategy including grey literature sources. Screening (title and abstract, full text), data extraction, and risk of bias assessment were done by 2 reviewers independently and in duplicate. Included studies were described in adequate detail. Assessed risk of bias using the Cochrane Risk of Bias 2 tool. Assessed certainty of evidence using GRADE approach, with disagreements resolved by consensus. Stated methods intended to be used for investigating heterogeneity. Assessed potential impact of risk of bias on results of meta-analysis by subgroup analyses by level of risk of bias (high vs. low). Conducted subanalyses to investigate clinical heterogeneity. Stated funding and conflicts of interest for authors conducting the review. | Did not provide justification for choice of included study designs. Did not provide list of excluded studies, though noted they captured reasons for exclusion at full-text screening stage. Did not report sources of funding for included studies. Did not provide explanation for choice of meta-analysis model. Did not discuss risk of bias in individual studies when interpreting results of review, though noted that no evidence of effect modification on outcomes of interest was found in subgroup analyses by risk of bias level. Did not conduct funnel plots to assess publication bias as planned due to few included trials. |
Carney et al. (2021)2 | |
Research questions and inclusion criteria included description of population, intervention, control, and outcomes of interest. All methods were determined a priori and published on their website as well as PROSPERO. Used a comprehensive search strategy, describing publication date range, databases used, and search strategies. Reference lists of systematic reviews and included articles were also used to identify potential studies for inclusion. A portal was also set up to facilitate submission of published and unpublished studies. Abstracts needed to be dual reviewed by 2 reviewers to confirm exclusion, and full texts were independently reviewed by 2 team members. A list of all excluded studies with justification for exclusion was provided in an appendix. Assessed risk of bias of individual studies included in the review, considering study risk of bias, consistency, directness, precision, and reporting bias. Strength of evidence was reviewed by the full team of investigators before assigning a final grade. Authors reported sources of funding and that they did not have any conflicts of interest. | Did not provide justification for study designs included and excluded in the review, or justification for only including English-language articles. Grey literature search methods were unclear. Abstracts were included if they were deemed appropriate by at least 1 reviewed. Unclear if data extraction was done in duplicate. Limited description of included studies (e.g., did not report country of study). Did not report sources of funding for included studies. Risk of bias in individual studies were not accounted for when discussing results of the review. Did not discuss potential impact of heterogeneity in results of interest for this review. |
Yang (2019)9 | |
Presented population, intervention, control, and outcomes of interest. Used a comprehensive search strategy. Screening (title and abstract, full text) and data extraction were conducted by 2 reviewers independently. Included studies were described in adequate detail. Risk of bias was assessed using the Cochrane risk of bias assessment tool. Stated they preferred the fixed-effects model compared with the random-effects model when there was no statistically significant heterogeneity, and vice-versa if there was statistically significant heterogeneity. Assessed for publication bias using funnel plots and Egger’s and Begger’s tests, which indicated no significant publication bias. Stated funding and conflicts of interest for authors conducting the review. | Unclear if protocol was developed in advance. Did not provide justification for excluded study designs. Unclear if grey literature was searched for. Did not provide list of excluded studies. Did not report sources of funding for included studies. Did not discuss risk of bias in individual studies when interpreting results of review, though noted in limitations the potential for selection and reporting bias for some outcomes. Did not discuss heterogeneity in review results. |
AMSTAR 2 = A Measurement Tool to Assess Systematic Reviews 2; vs. = versus.
Table 7: Strengths and Limitations of Clinical Studies Using the Downs and Black Checklist26
Strengths | Limitations |
|---|---|
Randomized controlled trial | |
Benger et al. (2022)10 | |
Clearly reported study’s aim, main outcomes, characteristics of patients, and interventions of interest. Baseline characteristics were similar between groups. As this was a randomized trial with a relatively large sample size, distributions of confounders may be similar between groups. Clearly described main findings including simple outcome data and confidence intervals. Reported some adverse events. Reported actual P values. Patients were blinded at the time of the intervention and authors reported this was likely maintained throughout the trial. Ambulance control room personnel, clinical staff caring for the patients beyond the ED, and researchers assessing the outcomes at hospital discharge and during follow-ups were blinded. Patients appear to be representative of the population from which they were recruited. Staff and facilities where patients were treated were likely representative of the treatment most patients receive. Follow-up appears to be the same for all study patients. Statistical tests appear to be appropriate. There was crossover between intervention and control groups. Sensitivity analyses (intention-to-treat analysis and as treated) were performed to estimate the effect. Patients from intervention and control groups were likely recruited from the same population over the same period of time. Paramedics were randomized as clusters to either use the intervention or control to treat patients. Allocation was concealed until enough information to identify the paramedic had been entered into the system, but not during patient enrolment. To reduce selection bias, all eligible patients were automatically enrolled. Losses of patients to follow-up were considered in sensitivity analyses. Main outcome measures appear to be valid and reliable. The primary outcomes were adequately powered for detecting a clinically important target difference. | Unclear if all important adverse events were reported; authors reported no severe adverse events only. Only about one-half of patients survived to 30 days to hospital discharge and consented to active follow-up. There were no report characteristics of patients not being followed. Participating paramedics were volunteers, and their airway skills may not be representative of those who chose not to take part. Blinding of paramedics (who delivered intervention) and ED staff was not possible. The analysis of long-term outcomes was affected by missing data with limited power. |
Nonrandomized studies | |
Levi et al. (2024)18 | |
Clearly described objective, main outcomes, inclusion criteria for patients, patient characteristics, interventions, and main findings. Estimated random variability in data for main outcomes. Reported adverse events. Patients do not appear to have been lost to follow-up. Reported actual P values. All eligible patients were included and were likely representative of the population from which they were recruited. Staff and facilities where patients were treated were likely representative of the treatment most patients receive. Main outcomes were objective and unlikely to be impacted by lack of blinding. Patients in different intervention groups appear comparable on several clinical and demographic variables. Follow-up appeared to be the same for all study patients. Statistical tests appear to be appropriate. Compliance with the intervention was likely reliable. Main outcome measures appear to be valid and reliable. | List of confounders not provided; as this is not a randomized trial, differences may have impacted outcomes. Patients in the intervention and control group were recruited from different periods of time which may have influenced the findings. Due to the nature of the study design, patients and staff were not blinded, and patients were not randomized. No adjustments for confounders were made in the analyses, which may affect the results due to the nonrandomized study design. Authors acknowledged that the study was underpowered to make conclusion on outcomes including mortality, ventilator-free days, or ICU length of stay. |
Smida et al. (2024)13 | |
Clearly described objective, main outcomes, inclusion criteria for patients, patient characteristics, interventions, and main findings. Clearly described distributions of principal confounders in each group of subjects. Estimated random variability in data for main outcomes. Data were from patient care records and appears to include all patients who met inclusion criteria within the time period of analysis. Patients appear to be representative of the population from which they were recruited, and care received was likely representative of what most patients receive. Main outcomes were objective and unlikely to be impacted by lack of blinding. Length of follow-up likely the same for all patients. Statistical tests appear to be appropriate. Main outcome measures appear to be valid and reliable. Compliance with intervention likely reliable; patients who did not receive interventions of interest were excluded. Patients who received intervention or control were from the same population over the same period of time. Performed adequate adjustment for confounding in the analyses. | Did not report adverse events. Did not report characteristics of patients lost to follow-up. Only 19.25% of patients transported to an emergency department had available disposition data. Did not report actual P values for the main outcomes. Due to the retrospective study design, patients were not blinded or randomized. Did not consider losses of patients to follow-up when analyzing survival to discharge to home. Authors acknowledged that the substantial missing data may have limited the power to detect differences between the intervention and the control. |
Kim et al. (2023)17 | |
Clearly described objective, main outcomes, inclusion criteria for patients, patient characteristics, interventions, and main findings. Estimated random variability in data for main outcomes Patients do not appear to have been lost to follow-up. Reported actual P values. All eligible patients were included and were likely representative of the population from which they were recruited. Staff and facilities where patients were treated were likely representative of the treatment most patients receive. Follow-up appeared to be the same for all study patients. Statistical tests appear to be appropriate. Compliance with the intervention was likely reliable. Patients from different intervention groups appear to have been recruited from the same population. Main outcome measures appear to be valid and reliable. | List of confounders not provided; as this is not a randomized trial, differences may have impacted outcomes. Authors conducted adjusted analyses but acknowledged that controlling completely for confounders was challenging. Unclear if all important adverse events have been reported. Due to the nature of the study design, patients and staff were not blinded, and patients were not randomized. Most main outcomes were objective. However, the neurologic outcomes at discharge were assessed by clinicians and may be impacted by lack of blinding. Did not report sample size calculation. |
Nichols et al. (2023)20 | |
Clearly described objective, main outcomes, inclusion criteria for patients, patient characteristics, interventions, and main findings. Estimated random variability in data for main outcomes Patients do not appear to have been lost to follow-up. Reported some adverse events. Reported actual P values. Patients were likely recruited from the same population over the same period of time. All eligible patients were included and were likely representative of the population from which they were recruited. Staff and facilities where patients were treated were likely representative of the treatment most patients receive. Follow-up appeared to be the same for all study patients. Statistical tests appear to be appropriate. Compliance with the intervention was likely reliable. Patients from different intervention groups were likely recruited from the same population. Main outcome measures appear to be valid and reliable. | List of confounders not provided; as this is a nonrandomized trial, differences may have impacted outcomes. Authors conducted adjusted analyses but acknowledged that they could not adjust for some confounders like operator experience. Unclear if all important adverse events were reported. Differences in adverse events by intervention was unclear due to the lack of reporting adverse events by intervention group, Unclear if patients in the intervention and control groups differed at baseline. Due to the nature of the study design, patients and staff were not blinded, and patients were not randomized. Most main outcomes were objective. However, successful airway placement was subjectively reported by clinicians and may be impacted by lack of blinding. |
Smida et al. (2023)14 | |
Clearly described objective, main outcomes, inclusion criteria for patients, patient characteristics, interventions, and main findings. Clearly described distributions of principal confounders in each group of subjects. Estimated random variability in data for main outcomes. Data were from patient care records and appears to include all patients who met inclusion criteria within the time period of analysis. Patients appear to be representative of the population from which they were recruited, and care received was likely representative of what most patients receive. Length of follow-up likely the same for all patients. Statistical tests appear to be appropriate. Main outcome measures appear to be valid and reliable. Compliance with intervention likely reliable; patients who did not receive interventions of interest were excluded. Patients who received intervention or control were from the same population. The control was available during the whole study period, while intervention was available 3 years after patient recruitment began. Patients who received intervention or control were not recruited over the same period of time. However, point estimate of the odds ratio comparing the 2 groups found consistent results across all calendar years after the intervention was available. Performed adequate adjustment for confounding in the analysis. | Did not report adverse events. Did not describe characteristics of patients lost to follow-up. Did not report actual P values for the main outcomes. Most main outcomes were objective. However, the neurologic outcomes at discharge were assessed by clinicians and may be impacted by lack of blinding. Due to the retrospective study design, patients were not blinded or randomized. Did not consider losses of patients to follow-up. Did not report sample size, though the study had a relatively large sample size. |
Price et al. (2022)12 | |
Clearly described objective, main outcomes, inclusion criteria for patients, patient characteristics, interventions, and main findings. Estimated random variability in data for main outcomes. No loss to follow-up. Reported actual P values. Data were from patient care records and appears to include all patients who met inclusion criteria within the time period of analysis. Patients appear to be representative of the population from which they were recruited, and care received was likely representative of what most patients receive. Length of follow-up likely the same for all patients. Statistical tests appear to be appropriate. Main outcome measures appear to be valid and reliable. Compliance with intervention likely reliable; patients who did not receive interventions of interest were excluded. Patients who received intervention or control were from the same population. | List of confounders not provided; authors acknowledge they could not control for geographic location of cardiac arrest or frequency with which paramedics used SGAs. Some analyses controlled for patient age and sex, but other potential confounders appear to not have been incorporated in analyses. Did not report adverse events. Due to study design, patients were not blinded or randomized. Patients who received the intervention were recruited at a different time than patients who received the control, as this was a historical control study. Most main outcomes were objective. However, successful airway placement was subjectively reported by clinicians and may be impacted by lack of blinding. Did not report sample size calculation. |
Lønvik et al. (2021)11 | |
Clearly described objective, main outcomes, inclusion criteria for patients, interventions, and main findings. Estimated random variability in data for main outcomes. Reported adverse events. Patients do not appear to have been lost to follow-up due to short follow-up time. Reported actual P values. All eligible patients were included and were likely representative of the population from which they were recruited. Staff and facilities where patients were treated were likely representative of the treatment most patients receive. Follow-up appeared to be the same for all study patients. Statistical tests appear to be appropriate. Compliance with the intervention was likely reliable. Main outcome measures appear to be valid and reliable. Study subjects in different intervention groups were recruited over the same period of time. | List of confounders not provided; authors noted differences between populations and geography, but assumed these would not have impacted primary end points. Patients in different intervention groups were not from the same population, as they received different interventions provided from different ambulance services: the ambulance services of Møre-og Romsdal and St. Olav’s hospital used the i-gel, while the Nord-Trøndelag used the King laryngeal tube. These were separate administrative units, but all followed the same guidelines, protocols, training, and certification. Due to the nature of the study design, patients and staff were not blinded, and patients were not randomized. Most main outcomes were objective. However, the ease to insert of the intervention or control was assessed by clinicians and may be impacted by lack of blinding. No adjustments for confounders were made in the analyses, which may affect the results due to the nonrandomized study design. Did not report sample size calculation. |
Matić et al. (2021)19 | |
Clearly reported study’s aim, patient inclusion criteria, main outcomes, characteristics of patients, and interventions of interest. Clearly described main findings including simple outcome data. Patients do not appear to have been lost to follow-up. Reported actual P values. All eligible patients were included and were likely representative of the population from which they were recruited. Staff and facilities where patients were treated were likely representative of the treatment most patients receive. Follow-up was likely the same for all study patients. Statistical tests appear to be appropriate. Compliance with the intervention was likely reliable. Patients from different intervention groups were likely recruited from the same population over the same period of time. Main outcome measures appear to be valid and reliable. Main outcomes were objective and unlikely to be impacted by lack of blinding. | List of confounders not provided; as this is not a randomized trial, differences may have impacted outcomes. Did not report adverse events. Unclear if patients in the intervention and control groups differed at baseline. Due to the nature of the study design, patients and staff were not blinded, and patients were not randomized. No adjustments for confounders were made in the analyses, which may affect the results due to the nonrandomized study design. Did not report sample size calculation. |
Edwards et al. (2019)15 | |
Clearly reported study’s aim, main outcomes, patient inclusion criteria, characteristics of patients, and interventions of interest. Patients in different intervention groups appear comparable on several clinical and demographic variables. Clearly described main findings including simple outcome data. Patients do not appear to have been lost to follow-up. Reported actual P values. All eligible patients were included and were likely representative of the population from which they were recruited. Staff and facilities where patients were treated were likely representative of the treatment most patients receive. Follow-up was likely the same for all study patients. Statistical tests appear to be appropriate. Compliance with the intervention was likely reliable. Patients from different intervention groups were likely recruited from the same population over the same period of time. Main outcome measures appear to be valid and reliable. | List of confounders not provided; as this is not a randomized trial, differences may have impacted outcomes. Did not report adverse events. Due to the nature of the study design, patients and staff were not blinded, and patients were not randomized. Restricted to a specific patient population, including those fulfilling postreturn of spontaneous circulation criteria for transfer to a specialist cardiac centre; findings may not be generalizable to those not meeting all the inclusion criteria. One researcher assessed the main outcome which may have inserted bias due to interrater variability in scoring. No adjustments for confounders were made in the analyses, which may affect the results due to the nonrandomized study design. Authors acknowledged that the sample size may be underpowered to detect a clinically significant effect. |
Jarvis et al. (2019)16 | |
Clearly reported study’s aim, patient inclusion criteria, main outcomes, characteristics of patients, and interventions of interest. Patients in different intervention groups appear comparable on several clinical and demographic variables. Clearly described main findings including simple outcome data. Patients do not appear to have been lost to follow-up. Reported actual P values. All eligible patients were included and were likely representative of the population from which they were recruited. Staff and facilities where patients were treated were likely representative of the treatment most patients receive. Follow-up was likely the same for all study patients. Statistical tests appear to be appropriate. Main outcome measures appear to be valid and reliable. Compliance with the intervention was likely reliable. Patients from different intervention groups appear to have been recruited from the same population over the same period of time. | List of confounders not provided. While authors did adjust for some confounders, the findings may have been impacted by unmeasured confounders. Did not report adverse events. Due to the nature of the study design, patients and staff were not blinded, and patients were not randomized. Data used came from electronic health records not collected for the purpose of this study, which may impact data quality. Outcome was based on subjective reporting, which may have led to overreporting of success. Multiple types of supraglottic airway devices were grouped together, which may make have hidden important differences (e.g., if 1 type of device leads to better outcomes than others). Did not report sample size calculation. |
ED = emergency department; ICU = intensive care unit; SGA = supraglottic airway device.
Table 8: Strengths and Limitations of Guideline Using AGREE II27
Item | AHRQ (2024)21 |
|---|---|
Domain 1: scope and purpose | |
1. The overall objective(s) of the guideline is (are) specifically described. | Yes |
2. The health question(s) covered by the guideline is (are) specifically described. | Yes |
3. The population (patients, public, and so on) to whom the guideline is meant to apply is specifically described. | Yes |
Domain 2: stakeholder involvement | |
4. The guideline development group includes individuals from all relevant professional groups. | Yes |
5. The views and preferences of the target population (patients, public, and so on) have been sought. | No |
6. The target users of the guideline are clearly defined. | Yes |
Domain 3: rigour of development | |
7. Systematic methods were used to search for evidence. | Yes |
8. The criteria for selecting the evidence are clearly described. | Yes |
9. The strengths and limitations of the body of evidence are clearly described. | Yes |
10. The methods for formulating the recommendations are clearly described. | Yes |
11. The health benefits, side effects, and risks have been considered in formulating the recommendations. | Yes |
12. There is an explicit link between the recommendations and the supporting evidence. | Yes |
13. The guideline has been externally reviewed by experts before its publication. | No |
14. A procedure for updating the guideline is provided. | No |
Domain 4: clarity of presentation | |
15. The recommendations are specific and unambiguous. | Yes |
16. The different options for management of the condition or health issue are clearly presented. | Yes |
17. Key recommendations are easily identifiable. | Yes |
Domain 5: applicability | |
18. The guideline describes facilitators and barriers to its application. | Yes |
19. The guideline provides advice or tools on how the recommendations can be put into practice. | Yes |
20. The potential resource implications of applying the recommendations have been considered. | Yes |
21. The guideline presents monitoring or auditing criteria. | No |
Domain 6: editorial independence | |
22. The views of the funding body have not influenced the content of the guideline. | Yes |
23. Competing interests of guideline development group members have been recorded and addressed. | Yes |
AGREE II = Appraisal of Guidelines for Research and Evaluation II; AHRQ = Agency for Health Care Research and Quality.
Appendix 5: Main Study Findings
Please note that this appendix has not been copy-edited.
Table 9: Summary of First-Pass Success Outcomes Between Supraglottic Airway Devices
Study citation, study design | Proportion of patients with successful FPS | Main findings | Notes | |
|---|---|---|---|---|
I-gel | Comparator | |||
I-gel compared with King laryngeal tube | ||||
Smida et al. (2024)13 NRS | ||||
All patients | 94.8% | 90.2% | aOR (95% CI): 1.94 (1.79 to 2.09) a | — |
Excluding patients with prehospital endotracheal intubation attempts before or after supraglottic airway device insertion | NR | NR | aOR (95% CI): 2.09 (1.87 to 2.33) a | — |
Price et al. (2022)12 NRS | 90.6% | 76.7% | Χ2(1) = 96.01, P < 0.001 OR (95% CI): 2.94 (2.35 to 3.67), P < 0.001 | — |
Lønvik et al. (2021)11 NRS | 157 of 191 (82%) | 41 of 59 (69%) | RR (95% CI): 0.86 (0.71 to 1.01) P = 0.07 | Direction of RR compares King laryngeal tube with i-gel. |
I-Gel compared with laryngeal mask airway | ||||
Carney et al. (2021)2 SR (1 relevant RCT, Benger et al., 2016) | NR | NR | NR | SR authors reported no difference. |
aOR = adjusted odds ratio; CI = confidence interval; CPR = cardiopulmonary resuscitation; ECG = electrocardiogram; FPS = first-pass success; NR = not reported; NRS = nonrandomized study; OHCA = out-of-hospital cardiac arrest; OR = odds ratio; RCT = randomized controlled trial; RR = risk ratio; SR = systematic review.
aModels adjusted for age, sex, initial ECG rhythm, witnessed status, bystander CPR, response interval, OHCA location, and whether patients received supraglottic airway devices after failed intubation attempts (rescue supraglottic airway devices).
Table 10: Summary of Other Success Rates Between Supraglottic Airway Devices
Study citation and study design | Outcome | Method of measurement | Result | Notes |
|---|---|---|---|---|
I-gel compared with King laryngeal tube | ||||
Price et al. (2022)12 NRS | Overall success | Number of attempts, mean (SD) | I-gel: 1.07 (0.29) King LT: 1.14 (0.44) | — |
Correlation, adjusting for age and sex | −0.093 | P < 0.001 | ||
Mann–Whitney U test | U = 714,918.00, z = −4.357, P < 0.001, d = 0.15 | Statistically significant difference in mean rank number of attempts for successful insertion, favouring a lower mean rank for the i-gel (median = 1, mean rank = 1,204.75) than for the King LT (median = 1; mean rank = 1,265.97). | ||
Lønvik et al. (2021)11 NRS | Success after 3 attempts from same personnel, or attempts from 2 or more personnel | Number of attempts, n (%) | I-gel: 7 (4%) King LT: 3 (5%) | — |
RR (95% CI) | 1.39 (0.37 to 5.20) | — | ||
P value | 0.63 | — | ||
Unsuccessful insertion | N (%) | I-gel: 27 (14%) King LT: 15 (25%) | — | |
RR (95% CI) | 1.80 (1.03 to 3.15) | — | ||
P value | 0.04 | — | ||
I-gel compared with laryngeal mask airway | ||||
Carney et al. (2021)2 SR (1 relevant RCT, Middleton et al., 2014) | Overall success | Proportion of patients who had successful insertion | I-gel: 90% LMA: 57% | — |
RR (95% CI) | 1.58 (1.11 to 2.24) | — | ||
P value | 0.023 | — | ||
CI = confidence interval; LMA = laryngeal mask airway; LT = laryngeal tube; NRS = nonrandomized study; RCT = randomized controlled trial; RR = risk ratio; SD = standard deviation; SR = systematic review.
Table 11: Summary of Survival Outcomes Between Supraglottic Airway Devices
Study citation, study design | Outcome | Adjusted odds ratio (95% CI) |
|---|---|---|
I-gel compared with King laryngeal tube | ||
Smida et al. (2024)13, a NRS (N = 93,866) | ||
All patients (n = 9,456) | Survival to discharge at home | 1.36 (1.06 to 1.76) |
Excluding patients with prehospital endotracheal intubation attempts before or after supraglottic airway device insertion (n = 6,893) | Survival to discharge at home | 1.26 (0.95 to 1.68) |
Smida et al. (2023)14, b | Survival to hospital admission | 1.07 (1.02 to 1.12) |
Survival to hospital discharge | 1.35 (1.26 to 1.46) | |
Survival with a CPC score of 1 or 2 at dischargec | 1.45 (1.33 to 1.58) | |
Initial ECG rhythm: shockable rhythm | 1.44 (1.28 to1.61) | |
Initial ECG rhythm: nonshockable rhythm | 1.50 (1.33 to 1.69) | |
Witnessed status: EMS witnessed | 1.37 (1.14 to 1.65) | |
Witnessed status: bystander witnessed | 1.42 (1.28 to 1.58) | |
Witnessed status: unwitnessed | 1.60 (1.36 to 1.88) | |
Sex: male | 1.42 (1.28 to 1.57) | |
Sex: female | 1.53 (1.34 to 1.76) | |
Location type: home or residence | 1.38 (1.25 to 1.53) | |
Location type: public | 1.76 (1.51 to 2.04) | |
Location type: nursing home or health care facility | 1.15 (0.85 to 1.56) | |
Age (years): < 18 | 1.76 (0.91 to 3.41) | |
Age (years): 18 to 65 | 1.47 (1.32 to 1.63) | |
Age (years): > 65 | 1.47 (1.28 to 1.67) | |
Transported to a hospital (excluding field terminations) | 1.45 (1.33 to 1.59) | |
Year of OHCA: 2016 (i-gel, n = 61; King LT, n = 14,914) | 1.98 (0.69 to 5.66) | |
Year of OHCA: 2017 (i-gel, n = 2,513; King LT, n = 14,763) | 1.19 (0.90 to 1.58) | |
Year of OHCA: 2018 (i-gel, n = 4,142; King LT, n = 14,695) | 1.24 (0.97 to 1.60) | |
Year of OHCA: 2019 (i-gel, n = 6,674; King LT, n = 15,102) | 1.49 (1.22 to 1.81) | |
Year of OHCA: 2020 (i-gel, n = 12,224; King LT, n = 18,441) | 1.66 (1.41 to 1.96) | |
Year of OHCA: 2021 (i-gel, n = 15,667; King LT, n = 17,875) | 1.55 (1.33 to 1.82) | |
I-gel compared with laryngeal mask airway | ||
Carney et al. (2021)2 SR (1 relevant RCT, Benger et al., 2016) | Survival to hospital discharge | NR; SR authors reported no difference |
CI = confidence interval; CPC = Cerebral Performance Category; CPR = cardiopulmonary resuscitation; ECG = electrocardiogram; EMS = emergency medical service; LT = laryngeal tube; NR = not reported; NRS = nonrandomized study; OHCA = out-of-hospital cardiac arrest; RCT = randomized controlled trial; SR = systematic review.
aModels adjusted for age, sex, initial ECG rhythm, witnessed status, bystander CPR, response interval, OHCA location, and whether patients received supraglottic airway devices after failed intubation attempts (rescue supraglottic airway devices). (p.194)13
bAdjusted for age, sex, calendar year of OHCA, initial ECG rhythm (shockable, nonshockable), witnessed status (unwitnessed, witnessed by bystander, witnessed by 9 to 1-1 responder), bystander CPR, response interval, and OHCA location (home or residence, public, nursing home or health care facility) (p.2)14
cCPC is a 5-point scale assessing neurologic functions after a resuscitation attempt. A CPC score of 1 or 2 is generally defined as a good outcome: 1 = good cerebral performance (conscious, alert, able to work, might have mild neurologic or psychologic deficit); 2 = mild cerebral disability (conscious, sufficient cerebral function for independent activities of daily life, able to work in sheltered environment). Scores of 3 to 5 mean poor outcomes: 3 = severe neurologic disability (conscious, dependent on others for daily support because of impaired brain function); 4 = comma or vegetative state (any degree of coma without the presence of all brain death criteria); 5 = brain death.14,28
Table 12: Summary of Return of Spontaneous Circulation Outcomes Between Supraglottic Airway Devices
Study citation and study design | Adjusted odds ratio (95% CI) | Notes |
|---|---|---|
I-gel compared with King laryngeal tube | ||
Smida et al. (2024)13 NRS | ||
All patients | 1.19 (1.13 to 1.26) | — |
Excluding patients with prehospital endotracheal intubation attempts before or after supraglottic airway device insertion | 1.17 (1.11 to 1.25) | — |
Smida et al. (2023)14 NRS | 1.03 (0.99 to 1.08) | — |
I-gel compared with laryngeal mask tube | ||
Carney et al. (2021)2 SR (2 relevant RCTs) | NR | SR authors reported no difference. |
CI = confidence interval; NR = not reported; NRS = nonrandomized study; RCT = randomized controlled trial; SR = systematic review.
Table 13: Summary of Rearrest Outcomes – I-Gels Compared With King Laryngeal Tubes
Study citation and study design | Adjusted odds ratio (95% CI) |
|---|---|
Smida et al. (2024)13 NRS | |
All patients | 0.73 (0.67 to 0.79) |
Excluding patients with prehospital endotracheal intubation attempts before or after supraglottic airway device insertion | 0.73 (0.66 to 0.81) |
Smida et al. (2023)14 NRS | 1.07 (0.97 to 1.19) |
CI = confidence interval; CPR = cardiopulmonary resuscitation; NRS = nonrandomized study.
Note: Rearrest was defined as having had “both documented prehospital ROSC (return of spontaneous circulation) and met one or more of the following conditions: (1) documented pulseless rhythms (PEA, asystole, ventricular fibrillation/tachycardia, torsades de points, unknown AED shockable/nonshockable rhythm) at emergency department (ED) arrival. (2) Documented defibrillation attempts, resumption of manual/- mechanical CPR, or 1 milligram bolus dose epinephrine administration after documented ROSC. (3) Greater than one documented CPR discontinuation” (p.194).13
Table 14: Summary of End-Tidal Carbon Dioxide Levels – I-Gels Compared With King Laryngeal Tubes
Study citation and study design | Adjusted odds ratio (95% CI) | |
|---|---|---|
ETCO2 < 5 mm Hg | ETCO2 < 10 mm Hg | |
Smida et al. (2024)13 NRS | ||
Excluding patients with prehospital endotracheal intubation attempts before or after supraglottic airway device insertion (n = 53,632) | 0.81 (0.70 to 0.95) | 0.84 (0.76 to 0.94) |
CI = confidence interval; ETCO2 = end-tidal carbon dioxide; NRS = non = randomized study.
Table 15: Summary of Complications in Supraglottic Airway Devices – I-Gels Compared With King Laryngeal Tubes
Complication | Lønvik et al. (2021)11 NRS | ||
|---|---|---|---|
I-gel n = 191 n (%) | King laryngeal tube n = 59 (%) n (%) | P value | |
Any reported complications | 92 (48) | 32 (54) | 0.435 |
Air leakage | 37 (19) | 5 (8) | 0.050 |
Aspiration | 24 (13) | 8 (14) | 0.842 |
Anatomic conditions | 22 (12) | 19 (32) | < 0.001 |
Problematic insertion | 13 (7) | 10 (17) | 0.018 |
Foreign object | 5 (3) | 2 (3) | 0.753 |
Hard to ventilate | 11 (6) | 6 (10) | 0.240 |
Insertion > 30 s | 3 (2) | 4 (7) | 0.034 |
Bleeding | 14 (7) | 7 (12) | 0.272 |
Dislocation | 11 (6) | 2 (3) | 0.474 |
Problems with bag valve mask | 27 (14) | 8 (14) | 0.911 |
Other | 14 (7) | 4 (7) | 0.886 |
NRS = nonrandomized study.
Table 16: Summary of Reported Difficulty of Insertion – I-Gel Compared With King Laryngeal Tube
Study citation and study design | Reported difficulty of insertion n (%) | RR (95% CI) | P value | |
|---|---|---|---|---|
I-gel | King laryngeal tube | |||
Lønvik et al. (2021)11 NRS | ||||
Easy | 152 (80%) | 30 (51%) | 0.64 (0.49 to 0.82) | < 0.001 |
Medium | 24 (13%) | 13 (22%) | 1.75 (0.95 to 3.22) | 0.07 |
Hard | 15 (8%) | 16 (27%) | 3.45 (1.82 to 6.56) | < 0.001 |
CI = confidence interval; NRS = nonrandomized study; RR = risk ratio.
Table 17: Summary of First-Pass Success Outcomes – Supraglottic Airway Devices Compared With Endotracheal Intubation
Study citation and study design | Outcome | Results | Notes | |
|---|---|---|---|---|
Intervention | ETI | |||
I-gel compared with endotracheal intubation | ||||
Levi et al. (2024)18 | Proportion of patients who had FPS, % (95% CI) | 96% (92% to 99%) | 68% (57% to 78%) | P < 0.001 |
Nichols et al. (2023)20 | Proportion of patients who had FPS, n of N (%) | |||
All patients | 183 of 222 (82.4%) | 575 of 705 (81.3%) | — | |
Direct laryngoscopy | NR | 83 of 122 (68.0%) | — | |
Video laryngoscopy | NR | 489 of 583 (83.9%) | — | |
Benger et al. (2022)10 | Proportion of patients with initial ventilation success (up to 2 attempts at AAM), % | 4,255 of 4,868 (87.4%) | 3,473 of 4,397 (79.0%) | OR (95% CI): 1.92 (1.66 to 2.22); P < 0.001 ADP, % (95% CI): 8.3 (6.3 to 10.2); P < 0.001 RR (95% CI): 1.11 (1.08 to 1.13); P < 0.001 Both OR and ADP adjusted for stratification factors fitted as fixed effects. |
Multiple types of supraglottic airway devices compared with endotracheal intubation | ||||
Jarvis et al. (2019)16 | Proportion of patients who had FPS, % | |||
All patients | 89.8% | 72.4% | — | |
Adult patients | 89.8% | 72.7% | — | |
Pediatric patients | 84.6% | 58.5% | — | |
aOR for adult patients compared with pediatric patients (95% CI)a | 1.63 (0.7 to 3.3) | 1.80 (1.49 to 2.17) | Factors independently associated with increased ETI FPS included use of RSI or SAI, female sex, and those identifying as white. The only factor independently associated with increased SGA FPS was SAI use. | |
Sensitivity analysis: for each additional year in age (age input as a continuous variable in years instead of a categorical variable), aOR (95% CI) a | 0.999 (0.995 to 1.004) | 1.007 (1.006 to 1.009) | When using 16 years of age as the cut-off, the odds of SGA FPS was higher in adults than pediatric patients, but not significantly different at any other age break point. Authors noted these findings align with previous reports showing increased difficulty with EMS intubation for younger patients. | |
AAM = advanced airway management; ADP = adjusted difference in proportions of patients ; aOR = adjusted odds ratio; CI = confidence interval; EMS = emergency medical services; ETI = endotracheal intubation; FPS = first-pass success; NR = not reported ; OR = odds ratio; RR = risk ratio; RSI = rapid sequence intubation; SAI = sedation assisted intubation; SGA = supraglottic airway device.
Note: Jarvis et al. (2019) included multiple types of SGAs; refer to the baseline characteristics table for more details.
aModel adjusted for indication, use of medications (rapid sequence intubation and sedation assisted intubation), sex, and patient identity.
Table 18: Summary of Other Success Rates and Placement Attempts – I-Gel Compared With Endotracheal Intubation
Study citation and study design | Outcome | Statistical analyses | ||
|---|---|---|---|---|
Measurement | I-gel | ETI | ||
Levi et al. (2024)18 | Reason for first-pass failure, n (%) | P = 0.445 | ||
Inability to visualize airway | 1 (20%) | 14 (52%) | — | |
Inability to open mouth | 0 | 1 (4%) | — | |
Inability to secure tube | 0 | 1 (4%) | — | |
Not specified | 4 (80%) | 11 (41%) | — | |
Second-pass success, n (%) | P = 0.057 | |||
No | 1 (20%) | 4 (15%) | — | |
Yes | 1 (20%) | 19 (70%) | — | |
Not specified | 3 (60%) | 4 (15%) | — | |
Number of airway placement attempts, mean (95% CI) | 1.0 (0.99 to 1.03) | 1.3 (1.18 to 1.40) | P < 0.001 | |
Total number of airway placement attempts, n (%) | 1: 115 (99%) 2: 1 (1%) 3: 0 | 1: 61 (74%) 2: 20 (24%) 3: 2 (2%) | P < 0.001 | |
Nichols et al. (2023)20 NRS | Success rates, n of N (%) | 193 of 222 (86.9%) | 655 of 705 (92.9%) | NR |
Patients who had direct laryngoscopy | NR | 101 of 122 (82.8%) | NR | |
Patients who had video laryngoscopy | NR | 552 of 583 (94.7%) | NR | |
Benger et al. (2022)10 RCT | Any ventilation success, n of N (%) | 573 of 753 (76.1) | 2,163 of 3,050 (70.9) | Not formally tested. |
CI = confidence interval; ETI = endotracheal intubation; NR = not reported; NRS = nonrandomized study; RCT = randomized controlled trial.
Table 19: Summary of Return of Spontaneous Circulation Outcomes – Supraglottic Airway Devices Compared With Endotracheal Intubation
Study citation and study design | Outcome | Statistical analyses | Notes | ||
|---|---|---|---|---|---|
Measurement | Intervention | ETI | |||
I-gel compared with endotracheal intubation | |||||
Levi (2024) NRS | Patients with ROSC before or at ED arrival, % (95% CI) | 37% (27 to 46) | 27% (17 to 37) | P = 0.203 | ROSC before or at ED arrival |
Forestell et al. (2024)8 SR (2 relevant RCTs) | Patients with ROSC, n of N | 1,175 of 3,838 | 1,009 of 3,558 | RR (95% CI): 1.08 (1.01 to 1.16) | Result from Benger et al., 2018. |
Patients with ROSC, n of N | 74 of 286 | 103 of 385 | RR (95% CI): 0.97 (0.75 to 1.25) | Result from Lee et al., 2022. | |
Kim et al. (2023)17 NRS | Patients with ROSC, n (%) | 109 (32.6%) | 2 (25.0%) | P = 0.89 | This test also included comparisons to other interventions that are not reported here. |
Benger et al. (2022)10 RCT | Patients with any ROSC during or after AAM by trial paramedic, n of N (%) | 1,295 of 4,155 (31.2%) | 992 of 3,416 (29.0%) | OR: 1.13 (95% CI, 1.01 to 1.27); P = 0.03 | ADP, % (95% CI): 2.5 (0.1 to 4.8); P = 0.04 |
ROSC of patients admitted to ED or hospital | 2,263 of 4,886 (46.3) | 1,922 of 4,410 (43.6) | Not formally tested | — | |
ROSC of patients on ED or hospital arrival, n of N (%) | 1,495 of 4,880 (30.6%) | 1,249 of 4,404 (28.4%) | OR 1.12 (95% CI 1.02 to 1.23); P = 0.02 | ADP, % (95% CI): 2.2 (0.3 to 4.2); P = 0.03 RR (95% CI): 1.08 (1.01 to 1.16); P = 0.02 | |
ROSC of patients who survived to ED discharge, n of N (%) | 1,033 of 2,259 (45.7) | 861 of 1,919 (44.9) | Not formally tested | — | |
Matić et al. (2021)19 NRS | Patients with ROSC, n (%) | 11 (24%) | 13 (28%) | P = 0.725 | No statistically significant differences on ROSC by initial rhythm (ventricular fibrillation, ventricular tachycardia, asystole, pulseless electrical activity) |
Laryngeal mask airways compared with endotracheal intubation | |||||
Forestell et al. (2024)8 SR (1 relevant RCT) | Patients with sustained ROSC, n of N | 40 of 87 | 36 of 101 | RR (95% CI): 1.29 (0.91 to 1.83) | Result from Mulder et al., 2013. Specifies intervention as LMA-S. |
Yang et al. (2019)9 SR with MA (includes 7 studies) | Patients with ROSC, % | 23% | 48% | Pooled RR (95% CI): 0.72 (0.65 to 0.80) I2 = 64.2%, P = 0.010 | Weights are based on random-effects analyses. Includes 2 RCTs and 5 NRSs. The 2 RCTs did not report significant differences, while the NRSs tended to report ETI as having more patients experience ROSC. |
King laryngeal tube compared with endotracheal intubation | |||||
Forestell et al. (2024)8 SR (1 relevant RCT) | Patients with ROSC, n of N | 195 of 700 | 171 of 700 | RR (95% CI): 1.14 (0.96 to 1.36) | Result from Wang et al., 2018. |
Multiple types of supraglottic airway devices compared with endotracheal intubation | |||||
Forestell et al. (2024)8 SR (4 RCTs) | Patients with ROSC, n of N | 1,484 of 4,911 | 1,319 of 4,744 | Pooled RR (95% CI): 1.09 (1.02 to 1.16) | This is the pooled RR for the 4 studies reported above (Mulder et al., 2013; Benger et al., 2018; Wang et al., 2018; and Lee et al., 2022). |
AAM = advanced airway management; ADP = adjusted difference in proportions of patients; CI = confidence interval; ED = emergency department; ETI = endotracheal intubation; LMA-S = laryngeal mask airway supreme; ROSC = return of spontaneous circulation; MA = meta-analysis; NR = not reported; NRS = nonrandomized study; OR = odds ratio; RCT = randomized controlled trial; RR = risk ratio; SR = systematic review.
Note: The results from Forestell et al. (2024) have been reported as individual studies as well as the pooled findings. This is to allow for reviewing the results of studies by specific types of supraglottic airway devices as well as combined.
Table 20: Summary of Time-Related Outcomes – Supraglottic Airway Devices Compared With Endotracheal Intubation
Study citation and study design | Outcome | Result | Statistical analyses | Notes | |
|---|---|---|---|---|---|
Intervention | ETI | ||||
I-gel compared with endotracheal intubation | |||||
Forestell et al. (2024)8 SR: 1 relevant RCT (i-gel: n = 286; ETI: n = 385). | Time to advanced airway placement (minutes), mean (SD) | 13.9 (4.5) | 15.9 (5.4) | MD (95% CI): −2.00 (−2.75 to −1.25 | Results from Lee et al., 2022. |
Levi et al. (2024)18 NRS | Time to first attempt (minutes), mean (95% CI) | 5.9 (5.1 to 6.7) | 8.3 (6.9 to 9.6) | P = 0.002 | (ETI: n = 80, i-gel: n = 110) |
Time to successful placement (minutes), mean (95% CI) | 6 (5.1 to 6.8) | 9.6 (8.2 to 11.1) | P < 0.001 | (ETI: n = 74, i-gel: n = 110) | |
King laryngeal tube compared with endotracheal intubation | |||||
Forestell et al. (2024)8 SR: 1 relevant RCT (King LT: n = 700; ETI: n = 700). | Time to advanced airway placement (minutes), mean (SD) | 10.8 (4.7) | 13.7 (5.9) | MD (95% CI): −2.90 (−3.46 to −2.34) | Results from Wang et al., 2018. |
Multiple types of supraglottic airway devices compared with endotracheal intubation | |||||
Forestell et al. (2024)8 SR: 2 RCTs (SGA: n = 986; ETI: n = 1084). | Time to advanced airway placement (minutes), mean (SD) | — | — | Pooled MD (95% CI): −2.49 (−3.37 to −1.61) | This is the pooled RR for the 2 studies reported above (Wang et al., 2018; Lee et al., 2022). |
CI = confidence interval; ETI = endotracheal intubation; MD = mean difference; NRS = nonrandomized study; RCT = randomized controlled trial; SD = standard deviation; SGA = supraglottic airway device; SR = systematic review.
Note: The results from Forestell et al. (2024) have been reported as individual studies as well as the pooled findings. This is to allow for reviewing the results of studies by specific type of supraglottic airway devices as well as combined.
Table 21: Summary of Survival Outcomes – Supraglottic Airway Devices Compared With Endotracheal Intubation
Study citation and study design | Outcome | Statistical analyses | Notes | ||
|---|---|---|---|---|---|
Measure | Intervention | ETI | |||
I-gel compared with endotracheal intubation | |||||
Forestell et al. (2024)8 SR includes 2 relevant RCTs. | Survival at longest follow-up, n of N | 308 of 3,838 | 300 of 3,558 | RR (95% CI): 0.95 (0.82 to 1.11) | Result from Benger et al., 2018. |
Survival at longest follow-up, n of N | 24 of 286 | 33 of 385 | RR (95% CI): 0.98 (0.59 to 1.62) | Result from Lee et al., 2022. | |
Levi et al. (2024)18 NRS | Mortality at 28 days, % (95% CI) | 14 (7 to 20) | 11% (4 to 18) | P = 0.656 | — |
Kim et al. (2023)17 NRS | Survival at discharge, n (%) | 50 (15.0% | 2 (25.0%) | P = 0.22 | This test included comparisons to other interventions that are not reported here. |
Benger et al. (2022)10 RCT | Survival to ED discharge | NR | NR | Not formally tested | Authors reported patients surviving to ED discharge appeared higher in the i-gel group than ETI group. |
Survival to hospital discharge (active consent), n of N (%) | NR | NR | NR | Authors reported no clear differences between treatment groups on survival. Similar results identified for patients without active consent. The SR by Forestell et al. (2024)8 reported above also presents survival numbers from the same trial (AIRWAYS-2), though a different publication (Benger et al., 2018). It was unclear if this finding is the same result reported by Forestell et al.8 | |
Died within 3 months of OHCA | 2 of 4,886 (0.0) | 2 of 4,410 (0.1) | NR | — | |
Died between 3 to 6 months of OHCA | 3 of 4,886 (0.1) | 3 of 4,410 (0.1) | NR | — | |
Survived to 6-month follow-up | 198 of 4,886 (4.1) | 190 of 4,410 (4.3) | NR | — | |
Survived to 3 months post OHCA, unknown survival status at 6 months post OHCA | 1 of 4,410 (0.0) | 0 of 4,886 (0.0) | NR | — | |
Time to death or last follow-up, up to 6 months (minutes), median (IQR) | 67 (41 to 216) | 63 (41 to 267) | HR (95% CI): 0.97 (0.93 to 1.02) P = 0.22 | — | |
Time to death, up to 72 hours (minutes), median (IQR) | 67 (41 to 205) | 63 (41 to 246) | HR (95% CI): 0.96 (0.92 to 1.00) P = 0.07 | — | |
Survival up to 72 hours, % | 13.6% | 13.1% | OR (95% CI): 1.04 (0.92 to 1.18) P = 0.54 | ADP, % (95% CI): 0.4 (1.0 to 1.9); P = 0.54 RR (95% CI): 1.04 (0.93 to 1.15); P = 0.53 | |
Laryngeal mask airways compared with endotracheal intubation | |||||
Yang et al. (2019)9 SR with MA | Survival rate to admission, % (5 included studies) | 19% | 27% | Pooled RR (95% CI): 0.85 (0.5 to 0.97) I2 = 66.3%, P = 0.018 | Weights are based on random-effects analyses. |
Survival to discharge, % (8 included studies) | 4% | 12% | Pooled RR (95% CI): 0.90 (0.80 to 1.02) I2 = 38.1%, P = 0.126 | Weights are based on random-effects analyses. | |
King laryngeal tube compared with endotracheal intubation | |||||
Forestell et al. (2024)8 SR: 1 relevant RCT | Survival at longest follow-up, n of N | 76 of 700 | 56 of 699 | RR (95% CI): 1.36 (0.98 to 1.88) | Result from Wang et al., 2018. |
Multiple types of supraglottic airway devices compared with endotracheal intubation | |||||
Forestell et al. (2024)8 SR: 3 RCTs | Survival at longest follow-up, n of N | 408 of 4,824 | 389 of 4,642 | Pooled RR (95% CI): 1.06 (0.84 to 1.34) | This is the pooled RR for the 3 studies reported above (Benger et al., 2018; Wang et al., 2018; and Lee et al., 2022). |
ADP = adjusted difference in proportions of patients; CI = confidence interval; ED = emergency department; ETI = endotracheal intubation; HR = hazard ratio; IQR = interquartile range; MA = meta analysis ; NR= not reported ; NRS = nonrandomized study; OHCA = out-of-hospital cardiac arrest; OR = odds ratio; RCT = randomized controlled trial; RR = risk ratio; SD = standard deviation; SGA = supraglottic airway device; SR = systematic review.
Note: The results from Forestell et al. (2024) have been reported as individual studies as well as the pooled findings. This is to allow for reviewing the results of studies by specific type of supraglottic airway devices as well as combined.
Table 22: Summary of Functional Outcomes – I-Gel Compared With Endotracheal Intubation
Study citation and study design | Outcome | Results | Statistical analysis | Notes | |
|---|---|---|---|---|---|
I-gel | ETI | ||||
Kim et al. (2023)17 NRS | Number of patients with favourable neurologic outcome (CPC scorea of 1 or 2), n (%) | 17 (5.1%) | 1 (12.5% | P = 0.37 | This test also included comparisons to other interventions that are not reported here. |
Benger et al. (2022)10 RCT | Good functional recovery at 30 days or hospital discharge (mRS scoreb of 0 to 3), n of N (%) | ||||
Main analysis: patients attended by a trial paramedic, who had resuscitation attempted, and met eligibility criteria (ITT) | 311 of 4,882 (6.4%) | 300 of 4,407 (6.8%) | OR (95% CI): 0.92 (0.77 to 1.09); P = 0.33 | ADP, % (95% CI): −0.62 (−1.65 to 0.41); P = 0.24. RR (95% CI): 0.92 (0.79 to 1.08); P = 0.32. Time from OHCA to mRS assessment was slightly lower in the ETI group (25 days) than the i-gel group (28 days). | |
Sensitivity analysis: including nonresuscitated patients (ITT) | 311 of 11,462 (2.7%) | 300 of 10,741 (2.8%) | OR (95% CI): 0.96 (0.81 to 1.14); P = 0.63 | — | |
Sensitivity analysis: excluded patients who did not receive i-gel or ETI (ITT) | 163 of 4,158 (3.9%) | 88 of 3,418 (2.6%) | OR (95% CI): 1.57 (1.18 to 2.07); P = 0.002 | — | |
Sensitivity analysis: excluded patients who did not receive i-gel or ETI (as treated) | 193 of 4,630 (4.2%) | 58 of 2,838 (2.0%) | OR (95% CI): 2.06 (1.51 to 2.81); P < 0.001 | — | |
Additional analysis: as treated | 193 of 4,630 | 53 of 2,838 | ADP (95% CI): 2.06 (1.24 to 2.89) P < 0.001 | Additional analyses to assess the causal effect of the treatment received on the primary outcome. Used 2-stage least squares with 2 instruments: (1) randomization, and (2) whether 1 or 2 paramedics initially attended the OHCA. First stage: treatment received was regressed on the 2 instruments and their interaction, obtaining predicted probabilities. | |
Additional analysis: causal analysis | 31 of 4,882 | 300 of 4,407 | ADP (95% CI): –0.49 (–5.38 to 4.40) P = 0.81 | Results from second stage of analysis: mRS score was regressed on predicted probabilities and stratification factors used in randomization. | |
Subgroup analysis: Utstein group (ITT) | NR | NR | OR (95% CI): 1.04 (0.80 to 1.35) | Utstein group: “includes patients with an OHCA with a likely cardiac cause that is witnessed and has an initial rhythm amenable to defibrillation” (p.42) | |
Subgroup analysis: Non-Utstein (ITT) | NR | NR | OR (95% CI): 0.73 (0.54 to 0.98) | Interaction between Utstein vs. non-Utstein: P = 0.07 | |
Subgroup analysis: witnessed by ambulance staff (ITT) | NR | NR | OR (95% CI): 0.78 (0.55 to 1.09) | — | |
Subgroup analysis: not witnessed by ambulance staff (ITT) | NR | NR | OR (95% CI): 0.93 (0.75 to 1.16) | Interaction between witnessed and nonwitnessed OHCA: P = 0.37 | |
Subgroup analysis: initial ventilation success (first 2 attempts) (as treated) | NR | NR | OR (95% CI): 2.65 (1.81 to 3.87) | — | |
Subgroup analysis: no initial ventilation success (first 2 attempts) (as treated) | NR | NR | OR (95% CI): 0.75 (0.38 to 1.49) | Interaction between patients who had initial ventilation success and those who did not: P = 0.001 | |
Good functional recovery at 3 months (mRS score of 0 to 3), n of N (%) | 121 of 4,636 (2.6%) | 123 of 4,199 (2.9%) | OR (95% CI): 0.89 (0.69 to 1.14); P = 0.35 | ADP, % (95% CI): −0.51 (−1.18 to 0.16); P = 0.14. Sensitivity analyses based on worse-case and imputed-case analyses reported similar results (no statistically significant difference) | |
Good functional recovery at 6 months (mRS score of 0 to 3), n of N (%) | 136 of 4,661 (2.9%) | 134 of 4,212 (3.2%) | OR (95% CI): 0.91 (0.71 to 1.16); P = 0.43 | ADP, % (95% CI): −0.39 (−1.08 to 0.30); P = 0.27. Sensitivity analyses based on worse-case and imputed-case analyses reported similar results (no statistically significant difference) | |
Edwards et al.(2019)15 NRS | Odds of poorer outcome (defined as CPC scorea of 3 to 5), ETI compared with i-gel | ||||
Unadjusted: including all cases, OR (95% CI) | — | — | 1.07 (0.93 to 1.32) | — | |
Unadjusted: nonshockable cases, OR (95% CI) | — | — | 1.11 (1.01 to 1.28) | — | |
Unadjusted: shockable cases, OR (95% CI) | — | — | 1.01 (0.74 to 1.40) | — | |
Adjusted for bystander CPR, witnessed arrest, shockable rhythm, aOR (95% CI) | — | — | 0.84 (0.34 to 3.13) | — | |
Adjusted for time to ROSC, age, shockable rhythm, aOR (95% CI) | — | — | 0.90 (0.37 to 2.19) | — | |
ADP = adjusted difference in proportions of patients; aOR = adjusted odds ratio; CI = confidence interval; CPC = Cerebral Performance Category; CPR = cardiopulmonary resuscitation; ETI = endotracheal intubation; ITT = intention-to-treat; mRS = modified Rankin Scale; NR = not reported ; NRS = nonrandomized study; OHCA = out-of-hospital cardiac arrest; OR = odds ratio; RCT = randomized controlled trial; ROSC = return of spontaneous circulation ; RR = risk ratio; SR = systematic review.
aCPC is a 5-point scale assessing neurologic functions after a resuscitation attempt. A CPC score of 1 or 2 is generally defined as a good outcome: 1 = good cerebral performance (conscious, alert, able to work, might have mild neurologic or psychologic deficit); 2 = mild cerebral disability (conscious, sufficient cerebral function for independent activities of daily life, able to work in sheltered environment). Scores of 3 to 5 mean poor outcomes: 3 = severe neurologic disability (conscious, dependent on others for daily support because of impaired brain function); 4 = comma or vegetative state (any degree of coma without the presence of all brain death criteria); 5 = brain death.14,28
bThe mRS is a 6-point scale measuring disability with scores ranging from 0 to 5: 0 = no symptoms; 1 = no significant disability (able to carry out all usual activities, despite some symptoms); 2 = slight disability (able to look after own affairs without assistance, but unable to carry out all previous activities); 3 = moderate disability (requires some help, but able to walk unassisted); 4 = moderately severe disability (unable to attend to own bodily needs without assistance, and unable to walk unassisted); 5 = Severe disability (requires constant nursing care and attention, bedridden, incontinent); 6 = dead.29
Table 23: Summary of Ventilator-Free Days and Length of Stay – I-Gel Compared With Endotracheal Intubation
Study citation and study design | Outcome | Results | Statistical analysis | |
|---|---|---|---|---|
I-gel | ETI | |||
Levi et al. (2024)18 NRS | Ventilator-free days a (number of days), mean (95% CI) | 2.1 (0.9 to 3.4) | 1.7 (0.4 to 3.0) | P = 0.665 |
ICU length of stay (number of days), mean (95% CI) | 2.8 (1.6 to 3.9) | 2.1 (0.8 to 3.4) | P = 0.469 | |
Benger et al. (2022)10 RCT | ICU length of stay for patients who survived to ICU discharge (hours), median | 100.5 | 98.6 | No formal comparisons done |
Hospital length of stay among patients who survived to hospital discharge (days), median | 14.0 | 12.3 | No formal comparisons done | |
CI = confidence interval; ETI = endotracheal intubation; ICU = intensive care unit; NRS = nonrandomized study; RCT = randomized controlled trial.
a“Ventilator-free days were calculated as “0” if the patient died within 28 days of mechanical ventilation or remained ventilated for more than 28 days. Ventilator-free days were calculated as “28 – X” if a patient was successfully liberated from mechanical ventilation X days from initiation” (p.4)18
Table 24: Summary of Quality of Life – I-Gel Compared With Endotracheal Intubation
Outcome | Benger et al. (2022)10 RCT | |||
|---|---|---|---|---|
I-gel, median (IQR) | ETI, median (IQR) | OR (95% CI) | P value | |
EQ-5D index scoresa by patients who survived to time points and consented to active follow-up (complete case) | ||||
30 days or hospital discharge | 0.71 (0.40 to 0.84) | 0.76 (0.50 to 0.84) | 0.92 (0.72 to 1.18) | 0.53 |
3 months | 0.81 (0.68 to 1.0) | 0.80 (0.67 to 0.91) | 1.07 (0.83 to 1.38) | 0.63 |
6 months | 0.84 (0.67 to 1.0) | 0.84 (0.70 to 1.0) | 0.92 (0.74 to 1.15) | 0.47 |
EQ VAS scoresb by patients who survived to time points and consented to active follow-up (complete case) | ||||
30 days or hospital discharge | 65 (45 to 80) | 70 (50 to 80) | 0.81 (0.64 to 1.03) | 0.08 |
3 months | 80 (65 to 90) | 80 (60 to 90) | 1.08 (0.86 to 1.35) | 0.53 |
6 months | 80 (65 to 90) | 80 (65 to 90) | 1.01 (0.80 to 1.27) | 0.94 |
CI = confidence interval; ETI = endotracheal intubation; IQR = interquartile range; NRS = nonrandomized study; OR = odds ratio; RCT = randomized controlled trial; SR = systematic review; VAS = Visual Analogue Scale.
Note: Berger et al. (2022) also reported comparisons between survivors and nonsurvivors, as well as analyses using worst-case and inputted-case scenarios. All were not statistically significant and are not presented here.
aThe EQ-5D system measures health-related quality of life across 5 dimensions, including mobility, self-care, usual activities, pain and discomfort, and anxiety and depression. Responses to can be converted to a single index value, with 1 being the healthiest state.10,30
bThe EQ VAS is a self-rated scale measuring patients’ overall current health on a vertical visual analogue scale from 0 (worst imaginable health) to 100 (best imaginable health).10,30
Table 25: Summary of Adverse Events – I-Gel Compared With Endotracheal Intubation
Study citation and study design | Outcome | Results | Statistical analysis | Notes | |
|---|---|---|---|---|---|
Intervention | ETI | ||||
Levi et al. (2024)18 NRS | Number of complications, n (%; 95% CI) | ||||
Any | 1 (1%; 0 to 3) | 3 (4%; 0 to 8%) | P = 0.310 | — | |
Aspiration | 1 (1%; 0 to 3) | 0 | P = 0.999 | — | |
Esophageal intubation | 0 | 2 (2%; 0 to 6) | P = 0.173 | — | |
Hypotension and hypoxia | 0 | 1 (1%; 0 to 5) | P = 0.417 | — | |
Kim et al. (2023)17 NRS | Aspiration pneumonia, n (%) | 60 (18.0%) | 1 (12.5%) | P = 0.082 | This test included comparisons to other interventions that are not reported here. |
Multivariable logistic regression, comparing to chest compression only, aOR (95% CI) | 2.27 (0.71 to 7.26) | 6.25 (0.24 to 158.68) | NR | — | |
Nichols et al. (2023)20 NRS | Total postairway attempt adverse events | NR | NR | — | Authors only reported results overall, not by intervention group. 82.3% of attempts had no postairway attempts |
Adverse responses during or after airway management | NR | NR | — | Authors only reported results overall, not by intervention group. 91.9% had no adverse responses during or after airway management | |
Adverse responses during ventilation or transport | NR | NR | — | Authors only reported results overall, not by intervention group. 85.3% had no adverse events during transport | |
Benger et al. (2022)10, a | Number of unexpected serious adverse events, n | 0 | 0 | — | — |
Regurgitation at any time, n of N (%) | 1268 of 4,865 (26.1) | 1072 of 4,372 (24.5) | OR (95% CI): 1.08 (0.96 to 1.20), P = 0.21 | ADP, % (95% CI): 1.4 (−0.6 to 3.4), P = 0.17 | |
Regurgitation during or after initial i-gel or ETI attempt | 875 of 4,857 (18.0) | 543 of 4,361 (12.5) | NR | — | |
Aspiration at any time, n of N (%) | 729 of 4,824 (15.1) | 647 of 4,337 (14.9) | OR (95% CI): 1.01 (0.88 to 1.16), P = 0.84 | ADP, % (95% CI): 0.1 (−1.5 to 1.8) 0.86 | |
Aspiration during or after initial i-gel or ETI attempt | 473 of 4,829 (9.8) | 304 of 4,344 (7.0) | NR | — | |
Loss of previously established airway, n (%)b | 412 of 3,900 (10.6) | 153 of 3,081 (5.0) | OR (95% CI): 2.29 (1.86 to 2.82), P < 0.001 | ADP, % (95% CI): 5.9 (4.6 to 7.2), P < 0.001 | |
ADP = adjusted difference in proportions of patients; aOR = adjusted odds ratio; CI = confidence interval; ETI = endotracheal intubation; NR = not reported; NRS = nonrandomized study; OR = odds ratio; RCT = randomized controlled trial; SR = systematic review.
aAuthors reported regurgitation and aspiration before the i-gel or ETI attempt; they have not been reported here to focus on adverse events that may have been due to the intervention. Authors noted that the proportion of patients experiencing either event before the advanced airway management attempt was larger in the tracheal intubation group than ETI group, which was the reverse of the outcomes during or after the advanced airway management attempt; however, these differences were not formally tested.
bAuthors noted that there are some cardiac patients who cannot experience effective ventilation with a supraglottic airway device, and ETI may be the only way to experience effective ventilation.
Table 26: Summary of Recommendations in Included Guideline
Recommendations | Supporting evidence | Quality of evidence and strength of recommendations |
|---|---|---|
AHRQ (2024)21 | ||
Suggests using BVM ventilation or SGA for airway management of adult patients with OHCA. | Authors reported insufficient evidence to indicate if BVM ventilation alone or SGA would provide better results in this population. Three RCTs reported equivalent survival, multiple NRSs report no difference in rates of ROSC. One trial reported improved neurologic function with BVM, but this was a subgroup analysis that may be affected by bias. The panel noted that BVM ventilation often requires more clinicians than SGA, so resource availability may influence the choice to use SGA compared with BVM ventilation alone. | Strength of recommendation: Conditional Certainty of evidence: Very low |
For adults with OHCA: in systems with demonstrated high ETI proficiency, suggest either SGA or ETI. In systems without demonstrated high ETI proficiency, suggest SGA versus ETI. | A SR identified 3 RCTs that compared ETI with different SGAs; 2 found no difference in survival, while 1 found SGA had a slight survival advantage. Pooled results showed no difference. Four NRSs had similar findings. The panel recognized the ease of use of SGA devices justify the conditional recommendation in settings without demonstrated high ETI proficiency. | Strength of recommendation: Conditional Certainty of evidence: Low-moderate. |
Suggests using BVM ventilation or SGA for airway management of pediatric patients who had an OHCA. | Authors state there was no convincing evidence to indicate BVM ventilation alone or SGA would provide better results in this population. An SR identified 2 NRSs: no difference was reported on survival or ROSC, while BVM ventilation was associated with improved neurologic function. However, this result may have been due to bias as this was not a RCT. The panel also noted that BVM ventilation often requires more clinicians than SGA, so resource availability may influence the choice to use SGA versus BVM ventilation alone. | Strength of recommendation: Conditional Certainty of evidence: Very low. |
Suggest SGA versus ETI in airway management of pediatric patients who had an OHCA. | Pooled analysis of 3 NRSs showed no difference between SGA and ETI in ROSC, neurologic outcomes, and survival rates. Two NRSs had higher first-pass success with SGA than ETI; no RCTs were found for this. The panel favoured SGA versus ETI due to higher first-pass success and lower potential training burden. | Strength of recommendation: Conditional Certainty of evidence: Very low. |
Suggests BVM alone or SGA can be used for adults who have experienced trauma. | Limited evidence; 1 NRS from a combat setting found no difference in mortality. Panel noted “using an SGA in a patient who is not unconscious and has an intact gag reflex likely requires medication-assisted placement, which adds complexity and risk” (p.549), and “concluded that the decision of SGA or effective BVM ventilation should be driven by the goal of optimizing oxygenation and ventilation, and avoiding hypoxia, hypotension, and hyperventilation” (p.549) | Strength of recommendation: Conditional Certainty of evidence: Very low. |
No recommendations regarding using BVM alone or SGA for pediatric patients who have experienced trauma. | No relevant studies available, so panel could not provide recommendation. | Strength of recommendation: NA Certainty of evidence: NA |
Suggests SGA or ETI can be used for adults who have experienced trauma. | One NRS found “ETI was associated with lower mortality when compared with SGA” (p.549), though this was an older study (2005) using an older SGA, so it is unclear if newer SGAs may be more effective. Due to limited data, panel concluded they could not support ETI versus SGA or vice-versa. | Strength of recommendation: Conditional Certainty of evidence: Very low. |
Suggests SGA versus ETI in airway management for pediatric patients who have experienced trauma. | No studies identified on patient-oriented outcomes; 1 study reported lower first-pass success rates with ETI than SGA. Considering this as well as harm associated with failed ETI attempts, lack of evidence of benefit, and procedure rarity, panel recommends SGA versus ETI. | Strength of recommendation: Conditional Certainty of evidence: Very low. |
No recommendations concerning SGA versus BVM alone for adult or pediatric patients experiencing medical emergencies due to lack of evidence. | No relevant studies available, so panel could not provide recommendation. | Strength of recommendation: NA Certainty of evidence: NA |
Suggests SGA or ETI can be used for adults experiencing medical emergencies in agencies with documented high ETI success; in agencies without documented high ETI success, recommend SGA versus ETI. | No studies identified on patient-oriented outcomes. Three studies identified related to procedural success; 1 found similar rates, 1 found it was higher with SGA than ETI, and 1 found it varied depending on medication assistance. Overall, the panel noted higher first-pass success with SGA, provided recommendations based on if agencies have documented high ETI success. | Strength of recommendation: Conditional Certainty of evidence: Very low. |
Suggest SGA versus ETI for airway management of pediatric patients experiencing medical emergencies. | Based on studies that noted higher first-pass success with SGA than ETI. This procedure is also rare for pediatric patients, so clinicians have limited experience with it. Some agencies may be able to safely support pediatric intubation programs (e.g., high-volume critical care transport programs) but the panel recommends most agencies should focus on SGA versus ETI. | Strength of recommendation: Conditional Certainty of evidence: Very low. |
AHRQ = Agency for Health Care Research and Quality; BVM = bag valve mask; ETI = endotracheal intubation; NA = not applicable; NRS = nonrandomized study; OHCA = out-of-hospital cardiac arrest; RCT = randomized controlled trial; ROSC = return of spontaneous circulation; SGA = supraglottic airway device; SR = systematic review.
Appendix 6: Overlap Between Included Systematic Reviews
Please note that this appendix has not been copy-edited.
Table 27: Overlap in Relevant Primary Studies Between Included Systematic Reviews
Primary study citation | RQ1 | RQ2 | |
|---|---|---|---|
Carney et al. (2021)2 | Forestell et al. (2024)8 | Yang et al. (2018)9 | |
Benger J et al. Br J Anaesth. 2016 Feb;116(2):262 to 8 | Yes | — | Yes |
Benger JR et al. JAMA 2018; 320:779 to 791 a | — | Yes | — |
Fan YJ et al. J Formos Med Assoc 2017;116:134 to 5. | — | — | Yes |
Kajino K et al. Crit Care 2011;15:R236. | — | — | Yes |
Lee A-F et al. JAMA Netw Open 2022; 5:e2148871 | — | Yes | — |
McMullan J et al. Resuscitation 2014;85:617 to 22. | — | — | Yes |
Middleton PM et al. Resuscitation. 2014 Jul;85(7):893 to 7 | Yes | — | — |
Mulder PJ et al. Resuscitation 2013; 84:S17 | — | Yes | — |
Ono Y et al. Am J Emerg Med 2015;33:1360 to 3. | — | — | Yes |
Shin SD et al. Resuscitation 2012;83:313 to 9. | — | — | Yes |
Takei Y et al. J Anesth 2010;24:716 to 25. | — | — | Yes |
Tanabe S et al. J Emerg Med 2013;44:389 to 97. | — | — | Yes |
Wang HE et al. JAMA 2018; 320:769 to 778 | — | Yes | — |
Yeung J et al. Resuscitation 2014;85:898 to 904. | — | — | Yes |
RQ = research question.
Note: Although both Carney et al. (2021) and Yang et al. (2018) included Benger et al. (2016), they reported on results from different comparisons. Carney et al. (2021) focused on comparisons between 2 types of supraglottic airway devices (i-gels and laryngeal mask airways), while Yang et al. (2018) reported on the comparisons between laryngeal mask airways and endotracheal intubation.
aThis publication is based on the AIRWAYS-2 trial, which was also reported on in Benger et al. (2022).10
Appendix 7: Summary of Nonevidence-Based Guidelines
Please note that this appendix has not been copy-edited.
Below are compiled recommendations from 5 guidelines identified from the literature search that were considered to not be evidence-based due to unclear or nonsystematic methodologies. The guideline from the Committee on Tactical Combat Casualty Care31 suggested adding gel-cuffed supraglottic airway device and the laryngeal tube supraglottic airway device to the medics’ aid bag in the prehospital combat setting, while other guidelines32-35 provided recommendations on general steps to use supraglottic airway devices. Characteristics of these guidelines were presented below in Table 28, and relevant recommendations were summarized in Table 29. No critical appraisal of these guidelines was conducted.
Table 28: Characteristics of Guidelines With Unclear or Alternative Methodology
Intended users, target population | Intervention and practice considered | Major outcomes considered | Evidence collection, selection, and synthesis | Evidence quality assessment | Recommendations development and evaluation | Guideline validation |
|---|---|---|---|---|---|---|
ANZCOR (2024)32 | ||||||
Intended users: Health professionals and those who provide health care in environments where equipment and drugs are available Target population: Adults who require ALS | Relevant intervention: SGAs | Insertion time, ventilation success rates | NR | The quality assessment of evidence supporting the recommendation relevant to this report was likely not assessed by the ANZCOR but retrieved from a 2015 CoSTR guideline.36 | Relevant recommendation development: NR. Recommendation evaluation: The evaluation of the recommendation relevant to this report was likely not conducted by the ANZCOR but retrieved from a 2015 CoSTR guideline.36 | NR |
NASEMSO (2023)35 | ||||||
Intended users: EMS medical directors and clinicians Target population: Patients requiring airway management to establish patency, protect the airway in an actual or impending impaired airway, and support oxygenation or ventilation in the prehospital setting | Relevant intervention: SGAs | Ventilatory rates/volumes, end-tidal CO2, and pulse oximetry | NR | NR | Recommendation development: the guideline reflects an evidence-informed review focused on out-of-hospital airway management and the Prehospital Airway Management Evidence Based Guideline Technical Expert Panel’s consensus recommendations. Recommendation evaluation: NR | NR |
NAEMSP (2022)33 | ||||||
Intended users: EMS clinicians Target population: Patients with OHCA | Relevant intervention: SGAs | Initial airway success, survival, ROSC | NR | NR | NR | NR |
NAEMSP (2022)34 | ||||||
Intended users: EMS clinicians Target population: Patients requiring the use of SGA in the prehospital setting | Relevant intervention: SGAs | Airway success rate, aspiration rate, survival, SGA placement | NR | NR | NR | NR |
CoTCCC (2023)31 | ||||||
Intended users: US military combat medics Target population: Patients requiring the use of airway management in the prehospital, combat environment | Relevant intervention: SGAs | Ease of manipulation, grip comfort, ease of insertion, required training, tolerance for the military environment, ability to secure the devices during transport | Studies conducted by the guideline development team were reviewed, including an analysis of end-user after action reviews, a market analysis, engineering testing, and prospective feedback from combat medics. | NR | Recommendation development: the expert consensus panel included 8 prehospital specialists, emergency medicine experts, and experienced combat medics. The panel members discussed the evidence in a virtual meeting. They made recommendations regarding their top 3 choices of devices including the options of military custom design. Recommendation evaluation: NR | NR |
ALS = advanced life support; ANZCOR = Australian and New Zealand Committee on Resuscitation; CoSTR = International Consensus on Cardiopulmonary Resuscitation and Emergency Cardiovascular Care Science With Treatment Recommendations; CoTCCC = Committee on Tactical Combat Casualty Care; EMS = Emergency Medical Services; NAEMSP = National Association of Emergency Medical Services Physicians; NASEMSO = National Association of State Emergency Medical Services Officials; NR = not reported; OHCA = out-of-hospital cardiac arrest; ROSC = return of spontaneous circulation; SGA = supraglottic airway device.
Table 29: Recommendations from Guidelines With Unclear or Alternative Methodology
Recommendations and supporting evidence | Quality of evidence and strength of recommendations |
|---|---|
ANZCOR (2024)32 | |
“ANZCOR suggests using either a supraglottic airway or tracheal tube as the initial advanced airway during CPR for cardiac arrest in any setting. Supraglottic airways are also a backup or rescue airway in a difficult or failed tracheal intubation.” (p. 6) | Quality of evidence: very lowa Strength of recommendations: weaka |
NASEMSO (2023)35 | |
Disease-specific good practice statements for airway management Adult trauma and medical emergencies “BVM ventilation is the starting point of positive pressure ventilation for adults with injuries and medical emergencies requiring airway management and may progress to SGA or ETI as needed.” (p. 4) “SGA should be considered as the primary invasive modality.” | Quality of evidence: NR Strength of recommendations: NR |
Disease-specific good practice statements for airway management Adult OHCA “SGA should be considered as the primary invasive modality.” (p. 4) | Quality of evidence: NR Strength of recommendations: NR |
Disease-specific good practice statements for airway management Pediatric trauma, medical emergencies, and OHCA “BVM is the starting point of positive pressure ventilation and may progress to SGA.” (p. 4) | Quality of evidence: NR Strength of recommendations: NR |
General steps in airway management Consider SGA for any unconscious patient without gag reflex “a. Consider using an appropriately sized SGA if BVM with OPA/NPA alone is not effective or inappropriate. i. Ventilation via SGA is often easier than via BVM. b. When escalating airway management strategies, use of an SGA is especially important in children as prehospital endotracheal intubation is an infrequently performed skill in this age group and has not been shown to improve outcomes over prehospital BVM or SGA.” (p. 5) | Quality of evidence: NR Strength of recommendations: NR |
General steps in airway management Invasive airway (SGA and ETI) placement “a. Optimize patient for first-pass success with pre-procedure resuscitation, preoxygenation, positioning, sedatives, and paralytics as indicated by patient presentation. i. In certain settings, sedatives and paralytics may improve first-pass success and overall success of airway management. However, such medications have not demonstrated improvement in patient survival. Because of the potential risks associated with using these medications, they should be reserved exclusively for EMS clinicians working within EMS systems that have established education, competency maintenance, quality improvement, and Medical Direction programs that demonstrate proficiency with airway management as a whole and medication facilitated airway management specifically. ii. Adequate preoxygenation (typically considered SpO2 ≥ 93% for at least 3 minutes) and the use of high flow apneic oxygenation with nasal cannula can decrease odds of hypoxia during airway attempts. b. Monitor clinical signs, pulse oximetry, cardiac rhythm, and blood pressure during invasive airway placement. c. Establish threshold values for safe initiation of SGA placement and ETI, as well as values requiring cessation of the attempt. These thresholds should be physiologically based and include oxygen desaturation, bradycardia, or hypotension.” (p. 6) | Quality of evidence: NR Strength of recommendations: NR |
General steps in airway management Postinvasive airway management “a. Inflate endotracheal tube cuff or SGA cuff (if applicable) with minimum air to seal airway and eliminate air leaks. If available, an ETT cuff manometer can be used to measure and adjust the ETT cuff pressure to the recommended 20 cmH2O pressure. b. Confirm invasive airway placement (ETT, SGA) with visual confirmation, absent gastric sounds, and bilateral breath sounds. Waveform capnography for confirmation is the standard of care and should be mandatory. c. Secure device manually. Once proper position is confirmed, secure the ETT or SGA with tape, twill, or appropriate commercial device. i. Note location of ETT or SGA at incisors or gum line and assess frequently for tube movement/displacement using continuous waveform capnography and visual inspection. ii. Head movement in children has been demonstrated to cause airway device displacement, therefore, consider cervical motion restriction to limit movement of the head and neck. d. Continuously monitor correct airway placement with repeat physical assessment and continuous waveform capnography during treatment and transport, paying particular attention to reassessing after each patient movement. e. Manual ventilation i. Tidal volume:
ii. Initial Rate (target to EtCO2):
iii. Continuously monitor EtCO2 to guide tidal volume and minute ventilation. f. Mechanical ventilation should be considered following invasive airway placement if available. See Mechanical Ventilation (Invasive) Guideline. g. Patients with an invasive airway should be provided and maintained with appropriate analgesia and sedation titrated to an appropriate target level using the Richmond Agitation-Sedation Scale (RASS) score or similar scale. h. Consider PEEP adjustment to achieve oxygenation goals (see above).” (p. 6 to 7) | Quality of evidence: NR Strength of recommendations: NR |
NAEMSP (2022)33 | |
“Based on the skill of the clinician and available resources, BVM, SGA, or ETI may be considered as airway management strategies in OHCA.” (p. 1) | Quality of evidence: NR Strength of recommendations: NR |
NAEMSP (2022)34 | |
Utility and selection of SGAs “SGAs have utility as a primary or secondary EMS airway intervention. EMS agencies should select SGA strategies that best suit available resources and local clinician skillset, as well as the nature of their clinical practice setting.” (p. 1) | Quality of evidence: NR Strength of recommendations: NR |
Availability of SGA devices “EMS agencies that perform endotracheal intubation must also equip their clinicians with SGA devices and ensure adequate training and competence.” (p. 2) | Quality of evidence: NR Strength of recommendations: NR |
Drug assisted SGA insertion “In select situations, drug assisted airway management may be used by properly credentialed EMS clinicians to facilitate SGA insertion.” (p. 3) | Quality of evidence: NR Strength of recommendations: NR |
Confirmation of SGA placement “Confirmation of initial and continuous SGA placement using waveform capnography is strongly encouraged as a best practice.” (p. 3) | Quality of evidence: NR Strength of recommendations: NR |
Conversion of an SGA to an ETT “When it is functioning properly, EMS clinicians should refrain from converting an SGA to an endotracheal tube. The decision to convert an SGA to an ET tube must consider the patient’s condition, the effectiveness of SGA ventilations, and the clinical context and course of initial SGA insertion.” (p. 4) | Quality of evidence: NR Strength of recommendations: NR |
SGA training and clinical use “SGA training, competency, and clinical use must be continuously evaluated by EMS agencies using focused quality management programs.” (p. 5) | Quality of evidence: NR Strength of recommendations: NR |
CoTCCC (2023)31 | |
Recommended SGAs for inclusion to the medics’ aid bag “The preponderance (7/8, 88%) of panel members recommended the gel-cuffed SGA, followed by the self-inflating-cuff SGA (5/8, 62%) and laryngeal tube SGA (5/8, 62%).” “The panel numbers concluded the gel-cuffed SGA and the laryngeal tube SGA devices were optimal for use.” | Quality of evidence: NR Strength of recommendations: NR |
ANZCOR = Australian and New Zealand Committee on Resuscitation; BVM = bag valve mask; CoTCCC = Committee on Tactical Combat Casualty Care; CPR = cardiopulmonary resuscitation; ETCO2 = end-tidal carbon dioxide; ETI = endotracheal intubation; ETT = endotracheal tube; NAEMSP = National Association of Emergency Medical Services Physicians; NASEMSO = National Association of State Emergency Medical Services Officials; NPA = nasopharyngeal airways; NR = not reported; OHCA = out-of-hospital cardiac arrest; OPA = oropharyngeal airways; PEEP = positive end expiratory pressure; SGA = supraglottic airway device.
aThe evidence quality assessment and recommendation evaluation was likely not conducted by the ANZCOR but retrieved from a 2015 guideline from the International Consensus on Cardiopulmonary Resuscitation and Emergency Cardiovascular Care Science With Treatment Recommendations.36
Appendix 8: References of Potential Interest
Please note that this appendix has not been copy-edited.
Systematic Reviews
Unclear Intervention
Amagasa S, Utsumi S, Moriwaki T, et al. Advanced airway management for pediatric out-of-hospital cardiac arrest: A systematic review and network meta-analysis. Am J Emerg Med. 2023;68():161-169.
Weihing VK, Crowe EH, Wang HE, Ugalde IT. Prehospital airway management in the pediatric patient: A systematic review. Acad Emerg Med. 2022;29(6):765-771. PubMed
Lavonas EJ, Ohshimo S, Nation K, et al. Advanced airway interventions for paediatric cardiac arrest: A systematic review and meta-analysis. Resuscitation. 2019;138():114-128.
Mixed or Alternative Intervention – Alternative Supraglottic Airway Devices
Wang CH, Lee AF, Chang WT, et al. Comparing Effectiveness of Initial Airway Interventions for Out-of-Hospital Cardiac Arrest: A Systematic Review and Network Meta-analysis of Clinical Controlled Trials. Ann Emerg Med. 2020;75(5):627-636. PubMed
Nonrandomized Studies
Unclear Intervention
Weigeldt M, Schulz-Drost S, Stengel D, Lefering R, Treskatsch S, Berger C. In-hospital mortality after prehospital endotracheal intubation versus alternative methods of airway management in trauma patients. A cohort study from the TraumaRegister DGU R. Eur. 2024;20():20.
Yoshimura S, Kiguchi T, Nishioka N, et al. Association of pre-hospital tracheal intubation with outcomes after out-of-hospital cardiac arrest by drowning comparing to supraglottic airway device: A nationwide propensity score-matched cohort study. Resuscitation. 2024;197():110129.
Nakayama R, Bunya N, Uemura S, Sawamoto K, Narimatsu E. Prehospital Advanced Airway Management and Ventilation for Out-of-Hospital Cardiac Arrest with Prehospital Return of Spontaneous Circulation: A Prospective Observational Cohort Study in Japan. Prehosp Emerg Care. 2024;28(3):470-477. PubMed
Song SR, Kim KH, Park JH, Song KJ, Shin SD. Association between prehospital airway type and oxygenation and ventilation in out-of-hospital cardiac arrest. Am J Emerg Med. 2023;65():24-30.
Tjerkaski J, Hermansson T, Dillenbeck E, et al. Strategies of Advanced Airway Management in Out-of-Hospital Cardiac Arrest during Intra-Arrest Hypothermia: Insights from the PRINCESS Trial. J. 2022;11(21):28.
Hongo T, Yumoto T, Naito H, Mikane T, Nakao A. Impact of different medical direction policies on prehospital advanced airway management for out-of hospital cardiac arrest patients: A retrospective cohort study. Resusc Plus. 2022;9():100210.
Jung E, Ro YS, Ryu HH, Shin SD. Association of prehospital airway management technique with survival outcomes of out-of-hospital cardiac arrest patients. PLoS One. 2022;17(6):e0269599. PubMed
Wang HE, Jaureguibeitia X, Aramendi E, et al. Airway strategy and ventilation rates in the pragmatic airway resuscitation trial. Resuscitation. 2022;176():80-87.
Chan JJ, Goh ZX, Koh ZX, et al. Clinical evaluation of the use of laryngeal tube versus laryngeal mask airway for out-of-hospital cardiac arrest by paramedics in Singapore. Singapore Med J. 2022;63(3):157-161. PubMed
Le Bastard Q, Rouzioux J, Montassier E, et al. Endotracheal intubation versus supraglottic procedure in paediatric out-of-hospital cardiac arrest: a registry-based study. Resuscitation. 2021;168():191-198.
Kim S, Lee DE, Moon S, et al. Comparing the neurologic outcomes of patients with out-of-hospital cardiac arrest according to prehospital advanced airway management method and transport time interval. Clin. 2020;7(1):21-29. PubMed
van Tulder R, Schriefl C, Roth D, et al. Laryngeal Tube Practice in a Metropolitan Ambulance Service: A Five-year Retrospective Observational Study (2009-2013). Prehosp Emerg Care. 2020;24(3):434-440. PubMed
Shekhar A, Blumen I, Narula J. ENDOTRACHEAL INTUBATION VERSUS SUPRAGLOTTIC AIRWAY INSERTION IN CARDIAC ARREST: THE PREHOSPITAL EXPERIENCE. Journal of the American College of Cardiology (JACC). 2021;77(18):3250-3250.
Mixed or Alternative Intervention – Alternative Supraglottic Airway Devices
Bartos JA, Clare Agdamag A, Kalra R, et al. Supraglottic airway devices are associated with asphyxial physiology after prolonged CPR in patients with refractory Out-of-Hospital cardiac arrest presenting for extracorporeal cardiopulmonary resuscitation. Resuscitation. 2023;186():109769.
Tham LP, Fook-Chong S, Binte Ahmad NS, et al. Pre-hospital airway management and survival outcomes after paediatric out-of-hospital cardiac arrests. Resuscitation. 2022;176():9-18.
Ryan KM, Bui MD, Dugas JN, Zvonar I, Tobin JM. Impact of prehospital airway interventions on outcome in cardiac arrest following drowning: A study from the CARES Surveillance Group. Resuscitation. 2021;163():130-135.
Nwanne T, Jarvis J, Barton D, Donnelly JP, Wang HE. Advanced airway management success rates in a national cohort of emergency medical services agencies. Resuscitation. 2020;146():43-49.
Ostermayer DG, Camp EA, Langabeer JR, et al. Impact of an Extraglottic Device on Pediatric Airway Management in an Urban Prehospital System. West J Emerg Med. 2019;20(6):962-969. PubMed
ISSN: 2563-6596
Canada’s Drug Agency (CDA-AMC) is a pan-Canadian health organization. Created and funded by Canada’s federal, provincial, and territorial governments, we’re responsible for driving better coordination, alignment, and public value within Canada’s drug and health technology landscape. We provide Canada’s health system leaders with independent evidence and advice so they can make informed drug, health technology, and health system decisions, and we collaborate with national and international partners to enhance our collective impact.
Disclaimer: CDA-AMC has taken care to ensure that the information in this document was accurate, complete, and up to date when it was published, but does not make any guarantee to that effect. Your use of this information is subject to this disclaimer and the Terms of Use at cda-amc.ca.
The information in this document is made available for informational and educational purposes only and should not be used as a substitute for professional medical advice, the application of clinical judgment in respect of the care of a particular patient, or other professional judgments in any decision-making process. You assume full responsibility for the use of the information and rely on it at your own risk.
CDA-AMC does not endorse any information, drugs, therapies, treatments, products, processes, or services. The views and opinions of third parties published in this document do not necessarily reflect those of CDA-AMC. The copyright and other intellectual property rights in this document are owned by the Canadian Agency for Drugs and Technologies in Health (operating as CDA-AMC) and its licensors.
Questions or requests for information about this report can be directed to Requests@CDA-AMC.ca.