CADTH Health Technology Review
Interferons, Glatiramer Acetate, and Minocycline for Relapsing-Remitting Multiple Sclerosis and Clinically Isolated Syndrome
Rapid Review
Key Messages
What Is the Issue?
Multiple sclerosis is a chronic autoimmune disorder that causes damage to central nervous system cells. Relapsing-remitting multiple sclerosis is characterized by relapses (episodes of new or worsening symptoms) followed by periods of partial or complete recovery (remission). A clinically isolated syndrome is the first episode of clinical symptoms and objective findings suggestive of multiple sclerosis. It lasts at least 24 hours and occurs without fever or infection.
Disease-modifying therapies for multiple sclerosis include interferon beta-1a and interferon beta-1b, and glatiramer acetate.
Minocycline is an antibiotic that may have a potential role in treating multiple sclerosis. Understanding its possible benefits and harms in clinically isolated syndrome and relapsing-remitting multiple sclerosis is important to clarify its potential role in these patients.
What Did We Do?
The purpose of this report is to update 2 previous reports we conducted in 2019. We summarize the evidence regarding the clinical effectiveness of glatiramer acetate, interferon beta-1a, and interferon beta-1b for clinically isolated syndrome, as well as minocycline for clinically isolated syndrome and relapsing-remitting multiple sclerosis. Evidence-based guidelines were also summarized.
We searched key resources, including journal citation databases, and conducted a focused internet search for relevant evidence published since 2019. One reviewer screened articles for inclusion based on predefined criteria, critically appraised the included studies, and narratively summarized the findings.
What Did We Find?
We found 1 systematic review that evaluated the safety of interferon beta-1a and glatiramer acetate in patients with clinically isolated syndrome.
Serious adverse events and withdrawals due to adverse events were similar in patients with clinically isolated syndrome who received interferon beta-1a and placebo.
Serious adverse events were similar in patients with clinically isolated syndrome who received glatiramer acetate and placebo. Withdrawals due to adverse events were more common in patients with clinically isolated syndrome who received glatiramer acetate than placebo.
We did not identify any studies that evaluated the clinical effectiveness of interferon beta-1a, interferon beta-1b, glatiramer acetate, or minocycline in patients with clinically isolated syndrome, or minocycline in patients with relapsing-remitting multiple sclerosis published since our previous reports. We did not identify any evidence-based guidelines published since our earlier reports.
What Does This Mean?
It is uncertain if interferon beta-1a, interferon beta-1b, glatiramer acetate, and minocycline are effective treatments for clinically isolated syndrome, or if minocycline is an effective treatment for relapsing-remitting multiple sclerosis due to the lack of evidence identified in this report.
Future studies are needed to understand the clinical effectiveness of interferon beta-1a, interferon beta-1b, glatiramer acetate, and minocycline in patients with clinically isolated syndrome, and minocycline in patients with relapsing-remitting multiple sclerosis.
Abbreviations
CDEC
Canadian Drug Expert Committee
RCT
randomized controlled trial
Context and Policy Issues
What Are Relapsing-Remitting Multiple Sclerosis and Clinically Isolated Syndrome?
Multiple sclerosis is a chronic autoimmune disorder of the central nervous system that causes demyelination (damage to the protective cover [myelin sheath] around nerve cells) and neurodegeneration (damage and/or death of nerve cells).1,2 Symptoms of multiple sclerosis can include constant fatigue, vision deficit, intense pain, sensory dysfunction, gait disturbances, cognitive impairment, urine incontinence, and spasticity.1,3 The diagnosis of multiple sclerosis is based on clinical symptoms, cerebrospinal fluid analysis, and MRI.3 It is estimated that more than 77,000 people older than 20 live with multiple sclerosis in Canada, and almost 75% of them are women.4 The cause of multiple sclerosis is unknown, and the age of onset is typically between 20 and 40 years.3
Clinically isolated syndrome is the first episode of clinical symptoms and objective findings suggestive of multiple sclerosis. It lasts at least 24 hours and occurs without fever or infection.3 Patients with clinically isolated syndrome may eventually convert to multiple sclerosis but do not yet fulfill the diagnostic criteria.5 Clinically isolated syndrome is the first demyelinating event in 85% of patients with multiple sclerosis.3 Relapsing-remitting multiple sclerosis, the most common form of the disease, is characterized by relapses (episodes of new or worsening symptoms) followed by periods of partial or complete recovery (remission).1,6
What Is the Current Practice?
There is no known cure for multiple sclerosis; however, treatments can decrease inflammation and reduce symptoms and the accumulation of disability.7 Treatment of multiple sclerosis includes disease-modifying therapies, acute relapse treatment, comorbidity management, symptom control, psychological support, rehabilitative strategies, and lifestyle modifications.2 The goal of disease-modifying therapies in relapsing-remitting multiple sclerosis is to decrease the frequency of relapses and reduce short-term disability.2 Disease-modifying therapies for multiple sclerosis include injectable medications (e.g., interferons, glatiramer acetate), oral medications (e.g., siponimod, fumarates, teriflunomide), and monoclonal antibodies (e.g., natalizumab, ocrelizumab).2
The therapeutic approach in clinically isolated syndrome is similar to multiple sclerosis management; however, it does not include symptomatic treatment due to the lack of chronic and severe symptoms.3 The goal of disease-modifying therapies in clinically isolated syndrome is to reduce the risk of conversion to multiple sclerosis.3
What Are Interferon Beta-1a and Interferon Beta-1b?
Interferons are naturally occurring cytokines (signalling proteins that affect the immune system) with complex effects on the central nervous system that are not fully understood.8 The mechanism of action of interferon-beta includes the inhibition of T-cell (white blood cell that is part of the immune system) activation and proliferation as well as reduction of T-cells crossing the blood-brain barrier.9 Interferon beta-1a is indicated for the treatment of relapsing forms of multiple sclerosis, including relapsing progressive multiple sclerosis and people who have experienced a single demyelinating event accompanied by abnormal MRI scans with lesions typical of multiple sclerosis.10 The recommended dosage is 30 mcg injected intramuscularly once a week.10 Interferon beta-1b is indicated for treating patients with a single demyelinating event with at least 2 clinically silent lesions typical of multiple sclerosis on MRI, relapsing-remitting multiple sclerosis, and secondary-progressive multiple sclerosis.11 The recommended dosage is 0.25 mg injected subcutaneously every other day.11 In 2013, the Canadian Drug Expert Committee (CDEC) recommended that interferon beta-1a (Rebif) not be publicly reimbursed for clinically isolated syndrome.12
What Is Glatiramer Acetate?
Glatiramer acetate is a mixture of synthetic polypeptides (chains of amino acids) containing 4 naturally occurring amino acids (L-glutamic acid, L-alanine, L-tyrosine, and L-lysine).8 Glatiramer acetate inhibits the activation of T-cells and prevents the entry of these cells into the brain.8 Glatiramer acetate is indicated for the treatment of ambulatory patients with relapsing-remitting multiple sclerosis, including patients who have experienced a single demyelinating event and have lesions typical of multiple sclerosis on MRI.13 Glatiramer acetate is available in 2 strengths, and the recommended dosage is 20 mg/mL once per day or 40 mg/mL 3 times per week at least 48 hours apart.13 In 2017, CDEC recommended glatiramer acetate (Glatect) be reimbursed for its approved indication of ambulatory patients with relapsing-remitting multiple sclerosis, including patients who have experienced a single demyelinating event and have lesions typical of multiple sclerosis on MRI.14 In contrast, in 2009, CDEC recommended glatiramer acetate (Copaxone) not be publicly reimbursed for the clinically isolated syndrome.15
What Is Minocycline?
Minocycline is a semisynthetic tetracycline antibiotic.16 Minocycline is a potential option for chronic neurologic disorders such as multiple sclerosis due to its anti-inflammatory and neuroprotective effects.16 Minocycline is indicated for treating various infections, including gall bladder infections, urinary tract infections, skin and soft tissue infections, and respiratory tract infections.17 Minocycline is available in capsules intended for oral administration, and the recommended dosage for adults is 100 mg or 200 mg initially, followed by 100 mg every 12 hours.17
Why Is It Important to Do This Review?
The current report is an update to 2 of our previous reports published in 2019.18,19 The 2019 report evaluating the clinical effectiveness of minocycline in patients with clinically isolated syndrome identified 1 randomized controlled trial (RCT).19 Evidence from this RCT indicated that the risk of conversion from clinically isolated syndrome to multiple sclerosis at 6 months was statistically significantly lower in patients treated with minocycline versus placebo; however, this difference between groups was not sustained at 24 months.19 The 2019 report that evaluated glatiramer acetate and interferon betas for clinically isolated syndrome identified 1 systematic review, 1 RCT, and 1 evidence-based guideline.18 Evidence from these studies suggested that compared to placebo, glatiramer acetate and interferon betas slowed the conversion from clinically isolated syndrome to multiple sclerosis and reduced the development of new brain lesions.18 Both previous reports outlined important limitations, including the limited quantity of available evidence and the risk of bias in the included studies.18,19 To inform decision-making, a request was made to determine whether new evidence has emerged since our previous reports.
Objective
The purpose of this report is to summarize new evidence regarding the clinical effectiveness of minocycline, glatiramer acetate, interferon beta-1a, and interferon beta-1b for clinically isolated syndrome and minocycline for relapsing-remitting multiple sclerosis as well as evidence-based guidelines.
Research Questions
What is the clinical effectiveness of minocycline for relapsing-remitting multiple sclerosis?
What is the clinical effectiveness of minocycline for clinically isolated syndrome?
What is the clinical effectiveness of glatiramer acetate for clinically isolated syndrome?
What is the clinical effectiveness of interferon beta-1a for clinically isolated syndrome?
What is the clinical effectiveness of interferon beta-1b for clinically isolated syndrome?
What are the evidence-based guidelines regarding minocycline for relapsing-remitting multiple sclerosis or clinically isolated syndrome?
What are the evidence-based guidelines regarding the use of glatiramer acetate and interferon beta-1a and 1b for clinically isolated syndrome?
Methods
Literature Search Methods
The literature search strategy used in this report is an update of searches developed for 2 of our previous reports.18,19 For the current report, an information specialist conducted a literature search on key resources, including MEDLINE, Embase, the Cochrane Database of Systematic Reviews, the International HTA Database, Canadian and major international health technology agencies, as well as a focused internet search. The search approach was customized to retrieve a limited set of results, balancing comprehensiveness with relevancy. Search concepts were developed based on the elements of the research questions and selection criteria. The main search concepts were minocycline, glatiramer acetate, interferon beta-1a, interferon beta-1b, multiple sclerosis, and clinically isolated syndrome. Retrieval was limited to the human population. All searches were limited to English-language documents. For the initial search on glatiramer acetate, interferon beta-1a, or interferon beta-1b for clinically isolated syndrome, results were limited to documents published between January 1, 2014 and August 19, 2019. For the initial search on minocycline for clinically isolated syndrome or multiple sclerosis, results were limited to documents published between January 1, 2009 and August 16, 2019. For the current report, database searches were rerun on April 24, 2024 to capture any articles published or made available since the initial search dates. The search of major health technology agencies was also updated to include documents published since August 2019.
Selection Criteria and Methods
One reviewer screened citations and selected studies. In the first level of screening, titles and abstracts were reviewed, and potentially relevant articles were retrieved and assessed for inclusion. As an update to 2 of our previous reports, articles were included if they were made available since the previous search date and were not included in the 2019 reports. The final selection of full-text articles was based on the inclusion criteria presented in Table 1.
Criteria | Description |
|---|---|
Population | Q1 and Q6: Adult patients with relapsing-remitting multiple sclerosis Q2 to Q7: Adult patients with clinically isolated syndrome |
Intervention | Q1, Q2, and Q6: minocycline Q3 and Q7: glatiramer acetate Q4 and Q7: interferon beta-1a Q5 and Q7: interferon beta-1b |
Comparator | Q1: Multiple Sclerosis therapies (i.e., interferon beta-1a, interferon beta-1b, glatiramer acetate, dimethyl fumarate, ocrelizumab, teriflunomide, peginterferon beta), placebo Q2: Clinically Isolated Syndrome therapies (i.e., interferon beta-1a, interferon beta-1b, glatiramer acetate), placebo Q3: interferon beta-1a, interferon beta-1b, placebo Q4: glatiramer acetate, interferon beta-1b, placebo Q5: interferon beta-1a, glatiramer acetate, placebo Q6 and Q7: NA |
Outcomes | Q1 to Q5: Clinical effectiveness (e.g., progression to clinically definite multiple sclerosis, time to progression to clinically definite multiple sclerosis, disability, relapse, changes in number and volume of lesions, health-related quality of life) and harms (e.g., adverse events) Q6: Guidelines regarding the use of minocycline for relapsing-remitting multiple sclerosis or clinically isolated syndrome Q7: Guidelines regarding the use of glatiramer acetate and interferon beta-1a and 1b for clinically isolated syndrome |
Study designs | Health technology assessments, systematic reviews, randomized controlled trials, nonrandomized studies, evidence-based guidelines |
NA = not applicable.
Exclusion Criteria
Articles were excluded if they did not meet the selection criteria outlined in Table 1 or were duplicate publications. Systematic reviews in which all relevant studies were captured in other more recent or more comprehensive systematic reviews were excluded. Primary studies retrieved by the search were excluded if they were captured in 1 or more included systematic reviews. Guidelines with unclear methodology were also excluded.
Critical Appraisal of Individual Studies
The included publication was critically appraised by 1 reviewer using A MeaSurement Tool to Assess systematic Reviews 2 (AMSTAR 2)20 as a guide. Summary scores were not calculated for the included study; instead, the strengths and limitations were described narratively.
Summary of Evidence
Quantity of Research Available
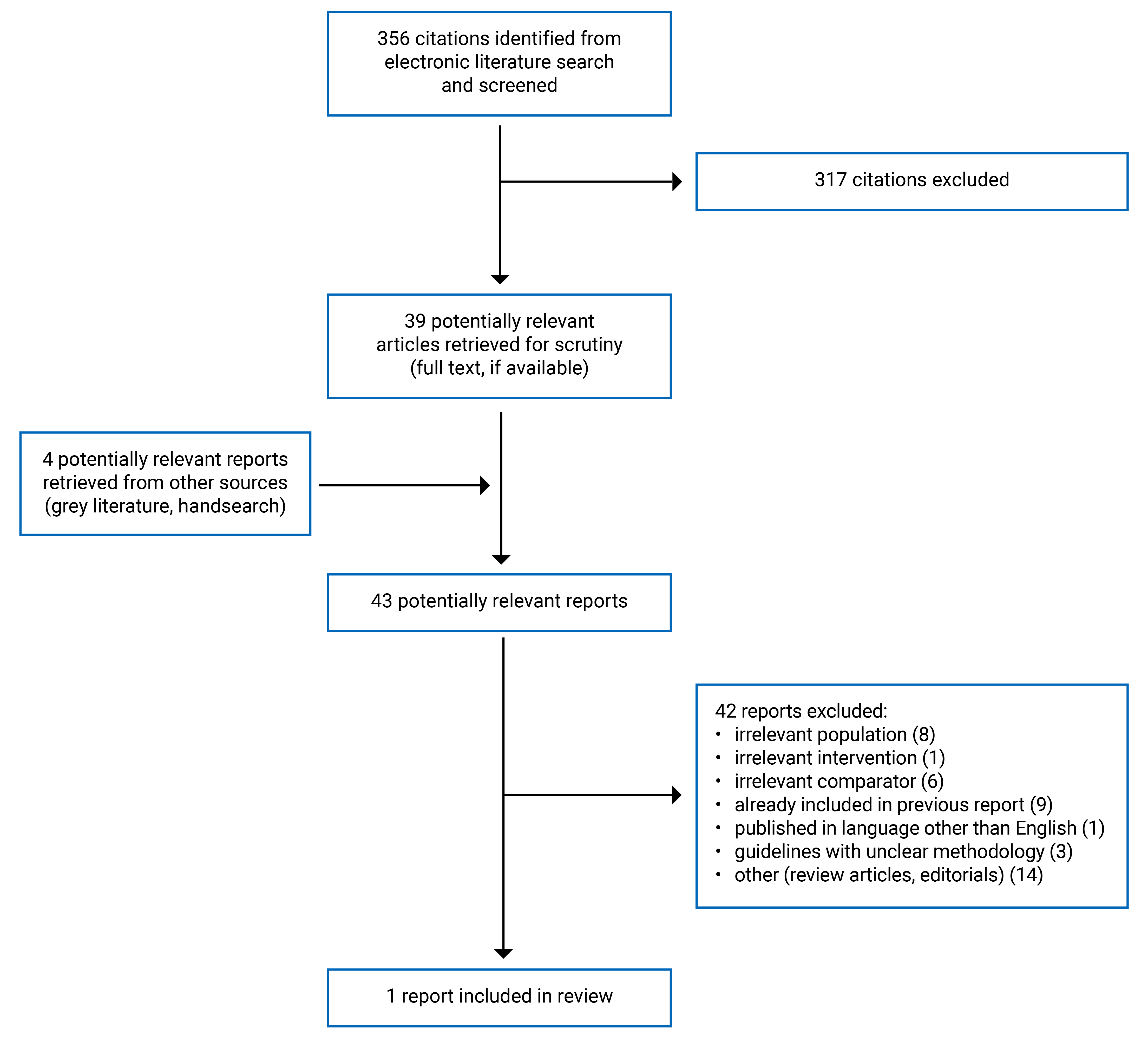
A total of 356 citations were identified in the literature search. Following the screening of titles and abstracts, 317 citations were excluded, and 39 potentially relevant reports from the electronic search were retrieved for full-text review. Four potentially relevant publications were retrieved from the grey literature search for a full-text review. Of these potentially relevant articles, 42 publications were excluded for various reasons, and 1 systematic review met the inclusion criteria and was included in this report. Appendix 1 presents the PRISMA21 flow chart of the study selection. Additional references of potential interest are provided in Appendix 2.
Summary of Study Characteristics
In total, 1 systematic review that evaluated safety was included in this report.22 The systematic review had broader inclusion criteria than those identified in our research protocol. It included studies of any immunotherapy used as monotherapy. It included studies of the relevant interventions (interferon beta-1b, interferon beta-1a, glatiramer acetate) in patients with other types of multiple sclerosis (e.g., relapsing-remitting multiple sclerosis, progressive multiple sclerosis). Additional details regarding the characteristics of the included publication are provided in Appendix 3.
We did not identify any studies that evaluated the clinical effectiveness of interferon beta-1a, interferon beta-1b, glatiramer acetate, or minocycline in patients with clinically isolated syndrome, or minocycline in patients with relapsing-remitting multiple sclerosis published since our previous reports. We did not identify any evidence-based guidelines published since our earlier reports.
Study Design
The search for the systematic review by Tracamere et al. (2023)22 included RCTs published up to March 4, 2022. The systematic review identified 123 RCTs, including 5 relevant to the present report.22
Country of Origin
Authors in Italy published the systematic review.22
Patient Population
The relevant population included in the systematic review was adults (aged 18 to 50 years) with clinically isolated syndrome.22 The time since the patients’ neurologic event ranged from 19 days to 3 months.22 The mean baseline Expanded Disability Status Scale in the relevant population ranged from 1.0 to 1.5.22. The Expanded Disability Status Scale measures how much someone is impacted by multiple sclerosis, and ranges from 0 to 10, with greater levels of disability corresponding to higher scores.23
Interventions and Comparators
In the relevant RCTs included in the systematic review, the interventions were interferon beta-1a and glatiramer acetate.22 The dosages for interferon beta-1a ranged from 22 to 44 mcg.22 In 3 of the RCTs, interferon beta-1a was administered once weekly, and in 1 RCT, it was administered either once weekly or 3 times weekly.22 The dosage for glatiramer acetate was 20 mg daily.22 The comparator in all the relevant RCTs was placebo.22
Outcomes
The systematic review by Tramacere et al. (2023) assessed serious adverse events and withdrawal due to adverse events.22 The follow-up in the relevant RCTs ranged from 24 to 36 months.22
Summary of Critical Appraisal
An overview of the strengths and limitations of the included systematic review is provided in Table 2. Overall, the systematic review was well conducted with multiple strengths and no limitations based on the critical appraisal using AMSTAR 2.
Table 2: Strengths and Limitations of Systematic Review Using AMSTAR 220
Strengths | Limitations |
|---|---|
Tramacere et al. (2023)22 | |
| None |
AMSTAR 2 = A MeaSurement Tool to Assess systematic Reviews 2; RCT = randomized controlled trial; SR = systematic review.
Summary of Findings
Appendix 4 presents the main study findings.
Safety of Interferon Beta-1a
Serious Adverse Events
Serious adverse events were more common in patients with clinically isolated syndrome who received interferon beta-1a than in those who received placebo in 2 of the RCTs identified in the Tramacere et al. (2023)22 systematic review. In 2 other RCTs identified in the systematic review, serious adverse events were more common in patients with clinically isolated syndrome who received a placebo than in patients who received interferon beta-1a.22 None of the differences between groups were statistically significant.22
Withdrawal Due to Adverse Events
Withdrawal due to adverse events was more common in patients with clinically isolated syndrome who received a placebo than interferon beta-1a (2 RCTs). However, the differences between groups were not statistically significant.22
Safety of Glatiramer Acetate
Serious Adverse Events
Serious adverse events were more common in patients with clinically isolated syndrome who received a placebo than patients who received glatiramer acetate in 1 RCT identified in the Tramacere et al. (2023)22 systematic review. The difference in serious adverse events between treatment groups was not statistically significant.22
Withdrawal Due to Adverse Events
Withdrawal due to adverse events was statistically significantly more common in patients with clinically isolated syndrome who received glatiramer acetate than in those who received placebo (1 RCT).22
Limitations
This report is limited by the quantity of evidence identified. One systematic review was identified that evaluated the safety of interferon beta-1a and glatiramer acetate in patients with clinically isolated syndrome. We did not identify any studies that assessed the clinical effectiveness of interferon beta-1a, interferon beta-1b, glatiramer acetate, or minocycline in patients with clinically isolated syndrome. We did not identify any studies that evaluated the clinical effectiveness of minocycline in patients with relapsing-remitting multiple sclerosis. Additionally, we did not identify any evidence-based guidelines.
Conclusions and Implications for Decision- or Policy-Making
Due to the limited evidence identified since our last reports, it is difficult to draw conclusions on the clinical effectiveness and safety of interferon beta-1a, interferon beta-1b, glatiramer acetate, and minocycline for the treatment of patients with clinically isolated syndrome as well as minocycline for the treatment of patients with relapsing-remitting multiple sclerosis. The safety of interferon beta-1a and glatiramer acetate was assessed in 1 systematic review, which reported no significant differences in serious adverse events or withdrawals due to adverse events between patients with clinically isolated syndrome treated with interferon beta-1a or placebo. Additionally, the systematic review reported no significant difference in serious adverse events in patients with clinically isolated syndrome treated with glatiramer acetate versus placebo. Withdrawals due to adverse events were more common in patients with clinically isolated syndrome who received glatiramer acetate than placebo.
Our 2 previous reports on interferon betas, glatiramer acetate, and minocycline identified 1 systematic review, 2 RCTs, and 1 evidence-based guideline.18,19 Results from 1 RCT indicated that the risk of conversion from clinically isolated syndrome to multiple sclerosis at 6 months was statistically significantly lower in patients treated with minocycline versus placebo; however, this difference between groups was not sustained at 24 months.19 Evidence from 1 systematic review and 1 RCT suggested that compared to placebo, glatiramer acetate and interferon betas slowed down the conversion from clinically isolated syndrome to multiple sclerosis and reduced the development of new brain lesions.18 Both previous reports outlined important limitations, including the limited quantity of available evidence and the risk of bias in the included studies.18,19
Future studies and evidence-based guidelines on the use of interferon beta-1a, interferon beta-1b, glatiramer acetate, and minocycline in patients with clinically isolated syndrome, and minocycline in patients with relapsing-remitting multiple sclerosis would aid in decision-making around the use of these drugs.
References
1.Aguilar ALJ, Solano FDC. Advances in Therapeutic Approaches for the Treatment of Multiple Sclerosis: Current and Future Perspectives. J Popul Ther Clin Pharmacol. 2023;30(17):1107-1126.
2.McGinley MP, Goldschmidt CH, Rae-Grant AD. Diagnosis and Treatment of Multiple Sclerosis: A Review. JAMA. 2021;325(8):765-779. PubMed
3.Grzegorski T, Losy J. What do we currently know about the clinically isolated syndrome suggestive of multiple sclerosis? An update. Rev Neurosci. 2020;31(3):335-349. PubMed
4.Public Health Agency of Canada. Multiple Sclerosis. 2019: https://www.canada.ca/en/public-health/services/chronic-diseases/multiple-sclerosis.html. Accessed 2024 May 14.
5.Hartung HP, Graf J, Kremer D. Long-term follow-up of multiple sclerosis studies and outcomes from early treatment of clinically isolated syndrome in the BENEFIT 11 study. J Neurol. 2020;267(2):308-316. PubMed
6.Cleveland Clinic. Relapsing-Remitting Multiple Sclerosis (RRMS). 2023: https://my.clevelandclinic.org/health/diseases/14905-rrms-relapsing-remitting-multiple-sclerosis. Accessed 2024 May 14.
7.Metz LM. Clinically Isolated Syndrome and Early Relapsing Multiple Sclerosis. Continuum (Minneap Minn). 2019;25(3):670-688. PubMed
8.De Kleijn KMA, Martens GJM. Molecular Effects of FDA-Approved Multiple Sclerosis Drugs on Glial Cells and Neurons of the Central Nervous System. Int J Mol Sci. 2020;21(12):13. PubMed
9.Sorensen PS, Magyari M, Sellebjerg F. An update on combination therapies for multiple sclerosis: where are we now? Expert Rev Neurother. 2023;23(12):1173-1187. PubMed
10.Avonex (interferon beta-1a): liquid for injection, 30 µg, intramuscular [product monograph]. Toronto (ON): Biogen Canada Inc.; 2022: https://pdf.hres.ca/dpd_pm/00066986.PDF. Accessed 2024 May 15.
11.Betaseron (interferon beta-1b): lyophilized powder for subcutaneous injection, 0.3 mg/vial [product monograph]. Mississauga (ON): Bayer Inc.; 2023: https://pdf.hres.ca/dpd_pm/00072199.PDF. Accessed 2024 May 15.
12.CADTH. CADTH Reimbursement Review: Rebif (Interferon beta-1a) for clinically isolated syndrome. 2013: https://www.cadth.ca/interferon-beta-1a. Accessed 2024 May 15.
13.Copaxone (glatiramer acetate): 20 mg / mL and 40 mg / mL single-use pre-filled syringes for subcutaneous injection; 40 mg / mL single-use pre-filled pens for subcutaneous injection [product monograph]. Toronto (ON): Teva Canada Limited; 2021: https://pdf.hres.ca/dpd_pm/00059892.PDF. Accessed 2024 May 15.
14.CADTH. CADTH Reimbursement Review: Glatect (glatiramer acetate) for relapsing remitting multiple sclerosis (RRMS). 2017: https://www.cadth.ca/glatiramer-acetate-0. Accessed 2024 May 15.
15.CADTH. CADTH Reimbursement Review: Copaxone (glatiramer acetate) for clinically isolated syndrome (CIS), suggestive of multiple sclerosis. 2009: https://www.cadth.ca/glatiramer-acetate. Accessed 2024 May 15.
16.Rezaei A, Moqadami A, Khalaj-Kondori M. Minocycline as a prospective therapeutic agent for cancer and non-cancer diseases: a scoping review. Naunyn Schmiedebergs Arch Pharmacol. 2023;22:22. PubMed
17.Minocycline (minocycline hydrochloride): 50 mg and 100 mg capsules [prescribing information]. Vaughan (ON): AA Pharma Inc.; 2019: https://pdf.hres.ca/dpd_pm/00053334.PDF. Accessed 2024 May 15.
18.Glatiramer Acetate and Interferon Beta 1a and 1b for Clinically Isolated Syndrome: A Review of Clinical Effectiveness and Guidelines. (CADTH Rapid response report: summary with critical appraisal). Ottawa (ON): CADTH; 2019: https://www.cadth.ca/glatiramer-acetate-and-interferon-beta-1a-and-1b-clinically-isolated-syndrome-review-clinical. Accessed 2024 May 10.
19.Minocycline for Relapsing-Remitting Multiple Sclerosis and Clinically Isolated Syndrome: A Review of Clinical Effectiveness and Guidelines. (CADTH Rapid response report: summary with critical appraisal). Ottawa (ON): CADTH; 2019: https://www.cadth.ca/minocycline-relapsing-remitting-multiple-sclerosis-and-clinically-isolated-syndrome-review-clinical. Accessed 2024 May 10.
20.Shea BJ, Reeves BC, Wells G, et al. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ. 2017;358:j4008. PubMed
21.Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol. 2009;62(10):e1-e34. PubMed
22.Tramacere I, Virgili G, Perduca V, et al. Adverse effects of immunotherapies for multiple sclerosis: a network meta-analysis. Cochrane Database Syst Rev. 2023;11:CD012186. PubMed
23.Multiple Sclerosis Society (MS Society UK). What is the Expanded Disability Status Scale (EDSS)? [2024]: https://www.mssociety.org.uk/living-with-ms/treatments-and-therapies/getting-treatment-for-ms/expanded-disability-status-scale. Accessed 2024 May 10.
Appendix 1: Selection of Included Studies
Appendix 2: References of Potential Interest
Note that this appendix has not been copy-edited.
Guidelines With Unclear Methodology
Yamout B, Al-Jumah M, Sahraian MA, et al. Consensus recommendations for diagnosis and treatment of Multiple Sclerosis: 2023 revision of the MENACTRIMS guidelines. Mult Scler Relat Disord. 2024 Mar;83:105435. PubMed
Al Malik YM, Al Thubaiti IA, AlAmmari MA, et al. Saudi Consensus Recommendations on the Management of Multiple Sclerosis: Disease-Modifying Therapies and Management of Relapses. Clin Transl Neurosci. 2022; 6(4):27.
Wiendl H, Gold R, Berger T, et al. Multiple Sclerosis Therapy Consensus Group (MSTCG): position statement on disease-modifying therapies for multiple sclerosis (white paper). Ther Adv Neurol Disord. 2021 Aug 18;14:17562864211039648. PubMed
Freedman MS, Devonshire V, Duquette P, et al. Treatment Optimization in Multiple Sclerosis: Canadian MS Working Group Recommendations. Can J Neurol Sci. 2020 Jul;47(4):437-455. PubMed
Review Articles
Collongues N, Becker G, Jolivel V, et al. A Narrative Review on Axonal Neuroprotection in Multiple Sclerosis. Neurol Ther. 2022 Sep;11(3):981-1042. PubMed
Piehl F. Current and emerging disease-modulatory therapies and treatment targets for multiple sclerosis. J Intern Med. 2021 Jun;289(6):771-791. PubMed
Additional References
Best Practices in MS Therapies Roundtable Group. Best Practices in Multiple Sclerosis Therapies. Hackensack (NJ): The Consortium of Multiple Sclerosis Centers; 2022: https://mscare.sharefile.com/share/view/s9f217488525840428fa35a051065d153.
Appendix 3: Characteristics of Included Publications
Table 3: Characteristics of Included Systematic Review
Study citation, country, funding source | Study design, outcomes | Intervention and comparators | Included studies | Population characteristics |
|---|---|---|---|---|
Tramacere et al. (2023)22 Italy Funding source: Fondazione I.R.C.C.S. Istituto Neurologico Carlo Besta, Milan, Italy and the Neurologic Institute Carlo Besta | Systematic review and network meta-analysis of RCTs Outcomes: serious adverse events, withdrawals due to adverse events | Eligible interventions: immunotherapies used as monotherapies Eligible comparators: placebo or another active drug Relevant interventions and comparators:
| 123 RCTs in total, 5 RCTs relevant to the present report | Eligible population: adults with multiple sclerosis or CIS Relevant population: adults with CIS Age range:
Time since the neurologic event:
Mean baseline EDSS:
|
CIS = clinically isolated syndrome; EDSS = Expanded Disability Status Scale; NR = not reported; RCT = randomized controlled trial.
Note that this appendix has not been copy-edited.
Appendix 4: Main Study Findings
Note that this appendix has not been copy-edited.
Table 4: Summary of Findings by Outcome — Serious Adverse Events
Citation | Primary study | Follow-up | Intervention | Control | Risk ratio (95% CI) | ||
|---|---|---|---|---|---|---|---|
Events | Total | Events | Total | ||||
Interferon beta-1a vs placebo | |||||||
Tramacere et al. (2023)22 (SR) | CHAMPS 2000 (RCT) | 36 months | 12 | 193 | 19 | 190 | 0.62 (0.31 to 1.25) |
ETOMS 2001 (RCT) | 24 months | 6 | 154 | 5 | 155 | 1.21 (0.38 to 3.87) | |
REFLEX 2012 (RCT) | 24 months | 14 | 344 | 12 | 171 | 0.58 (0.27 to 1.23) | |
Pakdaman 2007 (RCT) | 36 months | 9 | 104 | 7 | 98 | 1.21 (0.47 to 3.13) | |
Glatiramer acetate vs placebo | |||||||
Tramacere et al. (2023)22 (SR) | PreCISe 2009 (RCT) | 36 months | 11 | 243 | 19 | 238 | 0.57 (0.28 to 1.17) |
CI = confidence interval; RCT = randomized controlled trial; SR = systematic review.
Table 5: Summary of Findings by Outcome — Withdrawal Due to Adverse Events
Citation | Primary study | Follow-up | Intervention | Control | Risk ratio (95% CI) | ||
|---|---|---|---|---|---|---|---|
Events | Total | Events | Total | ||||
Interferon beta-1a vs placebo | |||||||
Tramacere et al. (2023)22 (SR) | CHAMPS 2000 (RCT) | 36 months | 2 | 193 | 7 | 190 | 0.28 (0.06 to 1.34) |
REFLEX 2012 (RCT) | 24 months | 9 | 344 | 8 | 171 | 0.56 (0.22 to 1.42) | |
Glatiramer acetate vs placebo | |||||||
Tramacere et al. (2023)22 (SR) | PreCISe 2009 (RCT) | 36 months | 15 | 243 | 4 | 238 | 3.67 (1.24 to 10.91) |
CI = confidence interval; RCT = randomized controlled trial; SR = systematic review.
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up-to-date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third-party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for noncommercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.
Questions or requests for information about this report can be directed to Requests@CADTH.ca.