CADTH Health Technology Review
Case Carts in Surgical Settings
Rapid Review
Authors: Monica Emode, Melissa Severn
Key Messages
What Is the Issue?
Case carts are commonly used in perioperative practice to transport surgical instruments and supplies to and from surgical settings in a timely manner. Open case carts are easy to manoeuvre and clean, while closed case carts effectively contain both sterile and contaminated surgical supplies during transport. Appropriate selection of case carts for surgical settings may support effective workflow processes, infection prevention and control measures, and personnel safety.
What Did We Do?
To inform decisions about appropriate selection of case carts, CADTH sought to identify and summarize the literature comparing infection control and personnel safety between open and closed case carts. An information specialist conducted a search of peer-reviewed and grey literature sources. CADTH also attempted to identify guidelines about the appropriate use of case carts in surgical settings.
What Did We Find?
No studies directly evaluating the comparative clinical effectiveness of open versus closed case carts or guidelines concerning their use in surgical settings were identified that met the inclusion criteria for this review. Research focused on the management of infection control and prevention and personnel safety is needed to evaluate the clinical effectiveness of open and closed case carts and inform guidance concerning the selection of open versus closed case carts in surgical settings.
What Does It Mean?
Without any comparative effectiveness evidence, decision-makers may want to consider the organizational features (e.g., sterile processing workflow systems, transport distance to the operating room) to inform the choice of either open or closed case carts. Measures to promote personnel safety when using any case cart may include training for use as well as cart inspection and monitoring processes.
Abbreviations
OR
operating room
SPD
sterile processing department
Context and Policy Issues
What Are Case Carts?
Case carts can be used to transport surgical instruments and supplies to the operating room (OR) from the sterile processing department (SPD).1 The most common types of case carts used in surgical settings are open case carts and closed case carts.2 Open case carts are lightweight, easy to manoeuvre and clean, provide greater visibility of surgical tools and supplies, and can be contained with a disposable cover.1 Open case carts allow for easy removal of surgical trays and may be more accessible for decontamination than closed case carts. In contrast, closed case carts, which include walls and doors that surround the shelves, are advantageous for transporting contaminated supplies and maintaining the sterile integrity of cart contents from dust or debris and are versatile enough to be used as a workspace in the OR.1,3 Closed case carts are optimal for transporting unsterile surgical instruments to the SPD. These carts often have additional features (e.g., locks to prevent tampering with surgical tools) that may also help to maintain a sterile environment when transporting surgical supplies to the OR. In addition, biohazard indicators can be made visible on all sides of a closed case cart.3-6
Why Is It Important to Do This Review?
An efficient case cart system can improve workflow in health care facilities, enhance patient care, and reduce OR turnover time.1,5,7-9 Furthermore, case carts that are easy, quick, and convenient to clean may be useful in health care settings to support infection control priorities.1,8 Given their respective designs, open and closed case carts may offer distinct potential benefits related to infection prevention and control; however, their relative benefits are unclear. It is also unclear whether the greater manoeuvrability of open case carts translates to fewer workplace injuries for health care staff. Comparative clinical evidence and guidelines for the use of case carts may provide useful context to inform purchasing decisions and other considerations for managing OR workflows.
Objective
The purpose of this report is to summarize the evidence identified from medical databases and grey literature searching regarding the comparative clinical effectiveness of open versus closed case carts in managing infection control and prevention and personnel safety. We also aimed to identify evidence-based guidelines regarding the use of case carts in surgical settings.
Research Questions
What is the comparative clinical effectiveness of closed case carts versus open case carts in surgical settings?
What are the evidence-based guidelines regarding case carts in surgical settings?
Methods
Literature Search Methods
An information specialist conducted a database literature search on key resources including MEDLINE, Embase, CINAHL, and the Cochrane Database of Systematic Reviews. A limited grey literature search was conducted on key resources, such as UpToDate, CMA Infobase, NICE, Cochrane, TRIP Database, ECRI, CADTH, International HTA database, and AHRQ. A focused internet search was also conducted. The search approach was customized to retrieve a limited set of results, balancing comprehensiveness with relevancy. The search strategy comprised both controlled vocabulary, such as the National Library of Medicine’s MeSH (Medical Subject Headings), and relevant keywords. Search concepts were developed based on the elements of the research questions and selection criteria. The main search concepts were case carts, surgical instruments, and operating rooms. Study design search filters were not used; however, conference abstracts were excluded from the Embase results. The search was completed on September 6, 2023, and limited to English-language documents published since January 1, 2013.
Selection Criteria and Methods
One reviewer screened citations and selected studies. In the first level of screening, titles and abstracts were reviewed and potentially relevant articles were retrieved and assessed for inclusion. The final selection of full-text articles was based on the inclusion criteria presented in Table 1.
Criteria | Description |
|---|---|
Population | Individuals receiving care in surgical settings |
Intervention | Closed case carts |
Comparator | Open case carts |
Outcomes | Q1: Clinical benefits and harms (e.g., infection prevention and control, personnel safety) Q2: Recommendations regarding the use of case carts (e.g., closed vs. open) |
Study designs | Health technology assessments, systematic reviews, randomized controlled trials, nonrandomized studies, evidence-based guidelines |
Search time frame | Published after January 1, 2013 |
Exclusion Criteria
Articles were excluded if they did not meet the selection criteria outlined in Table 1, they were duplicate publications, or were published before 2013. Guidelines that were not focused explicitly on case carts and guidelines with unclear methodology were also excluded.
Summary of Evidence
Quantity of Research Available
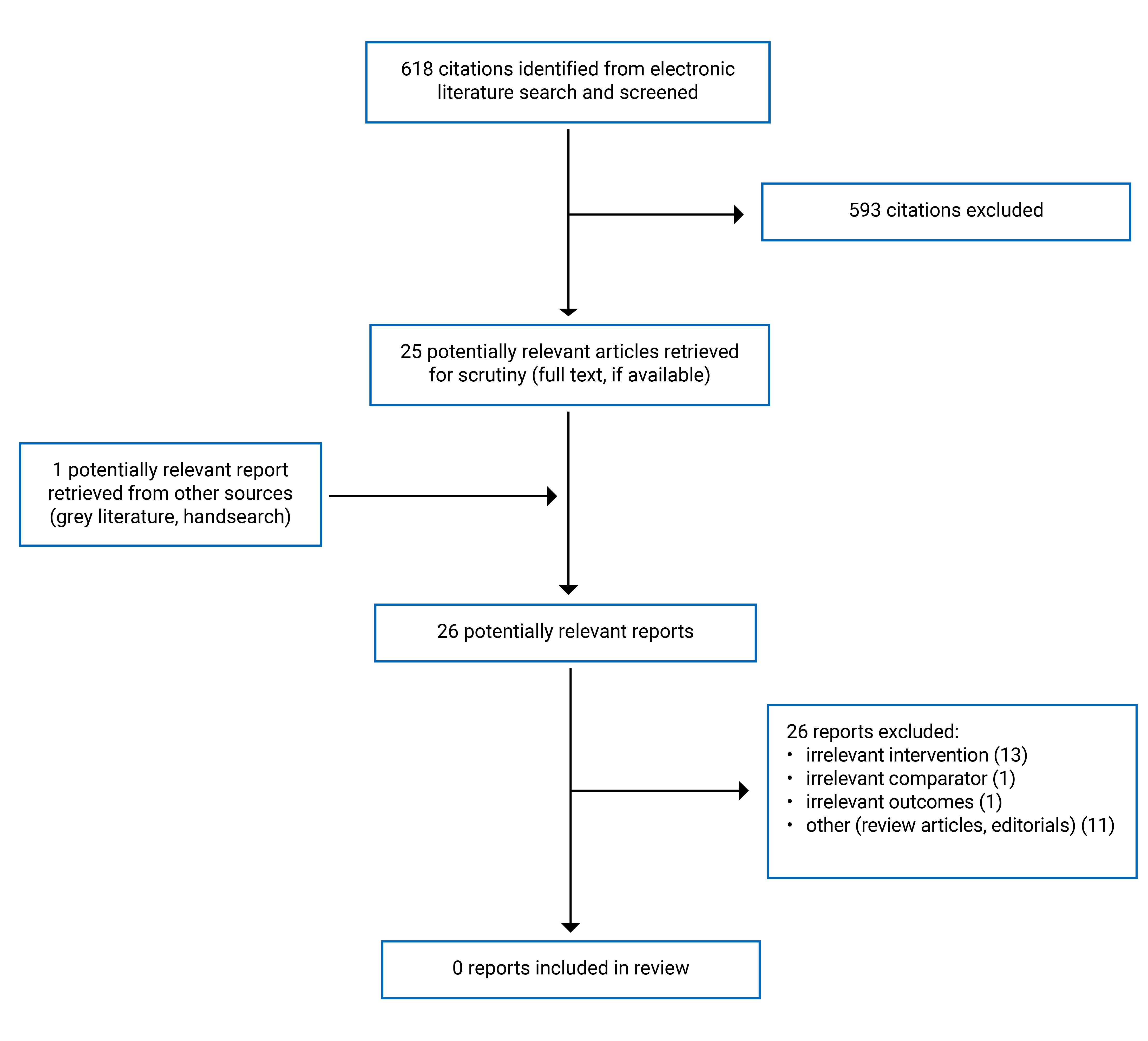
A total of 618 citations were identified in the literature search. Following screening of titles and abstracts, 593 citations were excluded and 25 potentially relevant reports from the electronic search and 1 potentially relevant report from handsearching were retrieved for full-text review. Of these potentially relevant articles, 26 publications were excluded for various reasons, and 0 publications met the inclusion criteria and were included in this report. Appendix 1 presents the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA)10 flow chart of the study selection. Additional references of potential interest that did not meet the inclusion criteria but may provide additional background on case carts are provided in Appendix 2.
Summary of Findings
We searched for documents published since January 1, 2013, and screened literature search results based on the research questions and inclusion criteria in Table 1. In this search, there were no studies or evidence-based guidelines that met those criteria. Therefore, we cannot provide a summary of the comparative clinical effectiveness of open versus closed case carts in surgical settings on infection rates or personnel safety outcomes or provide recommendations for their use.
Limitations
There were no eligible studies or evidence-based guidelines directly evaluating the clinical effectiveness of open case carts versus closed case carts in surgical settings identified through a search of medical databases and grey literature. We are unable to comment on the quality of evidence in this area because no studies met the inclusion criteria for this review. In line with Rapid Review methodology, which balances rigour with timeliness, the literature search conducted to inform this report was limited to 10 years (January 1, 2013, to September 6, 2023). It is possible that relevant literature regarding case carts in surgical settings exists but was published more than 10 years ago and therefore excluded by the date-limited search.
Conclusions and Implications for Decision- or Policy-Making
The literature search conducted for this review did not identify evidence directly evaluating the comparative clinical effectiveness of open and closed case carts or any evidence-based guidelines regarding case carts in surgical settings that met our inclusion criteria. Although potential benefits of open and closed case carts related to infection prevention and control have been discussed in health care commentary (e.g., ease of cleaning, containment of biohazardous materials, features used to maintain the sterile environment),1 without comparative evidence, we are unable to conclude whether open and closed case carts are more beneficial in maintaining infection control and prevention. Additional research using randomized studies with patient-level outcomes is needed to evaluate whether infection control and prevention can be enhanced with either open or closed case carts, although it is unclear if any research is forthcoming. We also did not identify any comparative evidence that evaluated the relative safety of open and closed case carts for the health care personnel who use them. More research is also needed to understand how personnel safety can be improved when using open and closed case carts. However, suggested measures to promote personnel safety while using any type of case cart include personnel training on the manufacturer’s instructions for use11 and implementing case cart inspection and monitoring processes after each use to ensure case carts remain free of debris and in good working condition.12
Decision-makers may wish to also consider organizational features (e.g., workflow systems, types of surgeries performed when selecting case carts). In some contexts, case cart choice may depend on the distance from the SPD to the OR. Collaboration between OR and SPD team members may help to determine the appropriate case cart to select based on their organization’s sterile processing workflow system. Closed case carts are advantageous in keeping dust and debris away from sterile surgical supplies and may be better than open case carts if the SPD is located farther away from the OR.4-6,8 In addition, the types of surgical procedures performed in the ORmay influence the selection of case carts. For example, orthopedic surgery may require fewer surgical tools, and therefore a smaller case cart. Furthermore, the use of key metrics (e.g., number of surgeries performed, number of instruments being reprocessed, average turnaround time for case cart preparation and sterilization) may inform the number and type of case carts required by the health care organization.4,7,13 These metrics may be considered in addition to the open or closed features.
References
1.Nadeau K. The case for conformity, collaboration in managing case carts. Healthcare Purchasing News. 2022;46(8):20-26.
2.Lakeside Manufacturing. Open or Closed Case Carts: What is Best for your Facility? 2022; https://www.elakeside.com/medical/open-or-closed-case-carts-what-is-best-for-your-facility/. Accessed 2023 Oct 11.
3.Richmond LI. A closed case cart system. AORN Journal. 1974;19(1):101-104. PubMed
4.Strzalka C, Zabriskie K. Physician Based Practices and Instrument Sterilization- How do you do it?...46th Annual Conference, APIC 2019, Philadelphia, PA. American Journal of Infection Control. 2019;47:S21-S21.
5.Alfred M, Catchpole K, Huffer E, Taafe K, Fredendall L. A Work Systems Analysis of Sterile Processing: Sterilization and Case Cart Preparation. Advances in Health Care Management. 2019;18:24. PubMed
6.Fast O, Fast C, Fast D, Veltjens S, Salami Z, White MC. Limited sterile processing capabilities for safe surgery in low-income and middle-income countries: experience in the Republic of Congo, Madagascar and Benin. BMJ glob. 2017;2(Suppl 4):e000428.
7.Nadeau K. Navigating case carts, kits and trays in the surgical services setting. Healthcare Purchasing News. 2023;47(8):22-25.
8.Dekonenko C, Oyetunji TA, Rentea RM. Surgical tray reduction for cost saving in pediatric surgical cases: A qualitative systematic review. J Pediatr Surg. 2020;55(11):2435-2441. PubMed
9.Bransby D. The case cart conundrum. Healthcare Purchasing News. 2016;40(9):62-65.
10.Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol. 2009;62(10):e1-e34. PubMed
11.Kyle E. Clinical Issues-May 2020. AORN Journal. 2020;111(5):558-566. PubMed
12.Williamson JE. Maintaining case cart & containers critical for safety, success. Healthcare Purchasing News. 2018;42(3):54-54.
13.Nadeau K. Off-site need not be off-putting. Healthcare Purchasing News. 2018;42(6):52-58.
Appendix 1: Selection of Included Studies
Appendix 2: References of Potential Interest
Note that this appendix has not been copy-edited.
Guidelines and Recommendations
Association of Surgical Technologists. AST standards of practice for creating the sterile field; 2011. https://static1.squarespace.com/static/579770cd197aea84455d6908/t/57d85f79e6f2e109efb83b21/1473798009594/Standard_Creating_Sterile_Field.pdf
Additional References
Basch JW. Implementing a case cart system in an existing hospital: a team approach. Hosp Mater Manage Q. 1984 Nov;6(2):49-54. PubMed
Bennett CW. A Surgical Case Cart System. Hospital Topics. 1982;60(4), 27-29. PubMed
Kapsar PP. Case cart systems—yea or nay?. AORN Journal. 1979;30(1), 58-63. PubMed
Mathias JM. What makes a case cart system work? OR Manager. 1992 Jun;8(6):16-18. PubMed
Schutta AM, Daly DS. The Surgical Case Cart System does it Belong in your Hospital? Hospital Topics. 1978;56(1), 10-17. PubMed
Welch TC. A Case Cart System: Planning, Implementing the System. AORN journal. 1990;52(5), 993-998. PubMed
Contributors: Camille Santos, Elizabeth Carson
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up-to-date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third-party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.
Questions or requests for information about this report can be directed to Requests@CADTH.ca