CADTH Health Technology Review
Treatment Options for VEXAS Syndrome
Rapid Review
Authors: Sarah Goring, Jennifer Horton
Abbreviations
ASCT
allogenic stem cell transplant
MDS
myelodysplastic syndrome
SAID
systemic autoinflammatory diseases
VEXAS
vacuoles, E1 enzyme, X-linked, autoinflammatory, somatic
Key Messages
VEXAS (vacuoles, E1 enzyme, X-linked, autoinflammatory, somatic) syndrome is a disorder caused by a genetic mutation, and is characterized by hematologic and autoinflammatory symptoms that are severe and progressive. It is typically seen in older men and is considered to be rare.
Proposed treatments for VEXAS syndrome include DNA hypomethylating agents such as azacitidine, anti-IL6 monoclonal antibodies such as tocilizumab, and Janus kinase inhibitors such as ruxolitinib.
We found 5 non-randomized studies on the effectiveness of the proposed treatments — 4 on azacitidine; 1 on ruxolitinib; and 1 on azacitidine, ruxolitinib, and tocilizumab.
There are several limitations to the research studies, most notably that they included a small number of patients who were retrospectively identified as having VEXAS syndrome. Therefore, the effectiveness of tocilizumab, ruxolitinib, and azacitidine for treating VEXAS syndrome is uncertain.
We did not find any evidence-based guidelines on the management of VEXAS syndrome.
Context and Policy Issues
VEXAS (vacuoles, E1 enzyme, X-linked, autoinflammatory, somatic) syndrome was first described in 2020, and is characterized by hematologic and autoinflammatory symptoms.1 Myeloid lineage-restricted mutations in the UBA1 gene were detected among men with a late-onset, treatment-refractory inflammatory syndrome with associated hematologic abnormalities, and VEXAS syndrome was established.
The UBA1 gene encodes the ubiquitin-like modifier activating enzyme 1, which is an enzyme that initiates ubiquitylation, and is involved in diverse aspects of intracellular signalling and protein degradation.1 The UBA1 gene is X-linked, and mutations are believed to be somatic.2 Consequently, VEXAS syndrome predominantly presents among males in late adulthood; however, VEXAS syndrome can also be present in women with X-acquired monosomy.3
The incidence and prevalence of VEXAS has not yet been established in Canada, or worldwide; however, VEXAS is considered to be rare.4,5 When VEXAS syndrome was first identified using a genotype-first approach, 3 out of a cohort of 2,560 patients with periodic fever, systemic inflammation, and/or other undiagnosed disease (0.001%) were identified as having a UBA1 gene mutation, whereas no UBA1 mutations were evident among healthy controls in the Genome Aggregation Database (gnOMAD) reference population database of nearly 200,000 individuals worldwide.1,2,6 More targeted testing among male patients with MDS or chronic myelomonocytic leukemia alongside systemic autoinflammatory diseases (SAID) has resulted in a prevalence estimate of 9% within this narrower group of symptomatic males.7
Clinical features of VEXAS syndrome include both hematologic abnormalities and autoinflammatory symptoms that are severe and progressive2; however, the clinical phenotype can be heterogeneous.8 Common hematologic abnormalities include macrocytic anemia and thromboembolic disease, and many patients have an associated hematological malignancy, such as myelodysplastic syndrome (MDS).9 These hematologic symptoms are often accompanied by difficult-to-control, multi-organ autoimmune manifestations, including ear and/or nose chondritis (inflammation of the cartilage), dermatological conditions, vasculitis, and recurrent fevers.9 Prior to discovering the genetic mutation that causes VEXAS syndrome, patients were treated for their hematologic and autoinflammatory symptoms, often as separate entities and by different specialists; however, the discovery of the UBA1 gene mutation highlights the common etiology of these disparate symptoms and can help inform therapeutic strategies.2
Two broad strategies for therapeutic management of VEXAS have been proposed, alongside supportive care.4 The first strategy is to eradicate the UBA1-mutated clone, which could potentially be curative. Allogenic stem cell transplant (ASCT) represents 1 such approach9; however, many patients with VEXAS present with high disease burden and may not be candidates for transplant.4 DNA hypomethylating agents such as azacitidine (which is a current standard of care for MDS10) also target mutated clones, and have been proposed as potential treatment for VEXAS. A second broad strategy involves blocking cytokines and other signalling pathways that mediate inflammatory response.4 Corticosteroids are a standard therapy for many types of inflammatory conditions; however, they can carry substantial risks of adverse events when used long-term at high doses.11 Other drugs that target cytokines and effector cell signalling pathways, and which have been considered for the treatment of VEXAS syndrome, include anti-IL6 monoclonal antibodies such as tocilizumab, and Janus kinase (JAK) inhibitors, such as ruxolitinib.4
The purpose of the current report is to summarize evidence regarding the clinical effectiveness of tocilizumab, ruxolitinib, and azacitidine among patients with VEXAS syndrome, relative to corticosteroids or no treatment, or as non-comparative estimates, and to summarize evidence-based treatment guidelines for VEXAS syndrome.
Research Questions
What is the clinical effectiveness of tocilizumab for patients diagnosed with VEXAS syndrome?
What is the clinical effectiveness of ruxolitinib for patients diagnosed with VEXAS syndrome?
What is the clinical effectiveness of azacitidine for patients diagnosed with VEXAS syndrome?
What are the evidence-based guidelines regarding the treatment options for the management of VEXAS syndrome?
Methods
Literature Search Methods
A limited literature search was conducted by an information specialist on key resources including MEDLINE, Embase, the Cochrane Database of Systematic Reviews, the International HTA Database, the websites of Canadian and major international health technology agencies, and clinical trials registries (US National Institutes of Health’s clinicaltrials.gov, WHO’s International Clinical Trials Registry, Health Canada Clinical Trials Database, and the European Clinical Trials Registry), as well as a focused internet search. The search strategy comprised both controlled vocabulary, such as the National Library of Medicine’s MeSH (Medical Subject Headings), and keywords. The main search concept was VEXAS syndrome. No filters were applied to limit the retrieval by study type. Where possible, retrieval was limited to the human population. The search was completed on September 20, 2022, and limited to English-language documents published since January 1, 2017.
Selection Criteria and Methods
One reviewer screened citations and selected studies. In the first level of screening, titles and abstracts were reviewed and potentially relevant articles were retrieved and assessed for inclusion. The final selection of full-text articles was based on the inclusion criteria presented in Table 1.
Criteria | Description |
|---|---|
Population | Patients diagnosed with VEXAS syndrome |
Intervention | Q1: Tocilizumab Q2: Ruxolitinib Q3: Azacitidine Q4: Any treatment options |
Comparator | Q1 to Q3: Corticosteroids, no comparator Q4: Not applicable |
Outcomes | Q1 to Q3: Clinical effectiveness (e.g., progression free survival, overall survival, response rate, duration of response, quality of life, safety [i.e., adverse events ≥ grade 3 and grade 4, serious adverse events, deaths]) Q4: Recommendations regarding the treatment options for the management of VEXAS syndrome (e.g., treatment regimens, dosages, appropriate patient populations) |
Study designs | Health technology assessments, systematic reviews, randomized controlled trials, non-randomized studies, evidence-based guidelines |
VEXAS = Vacuoles, E1 enzyme, X-linked, autoinflammatory, somatic; Q = question.
Exclusion Criteria
Articles were excluded if they did not meet the selection criteria outlined in Table 1, they were duplicate publications, or they were published before 2017. Systematic reviews in which all relevant studies were captured in other more recent or more comprehensive systematic reviews were excluded. Primary studies retrieved by the search were excluded if they were captured in 1 or more included systematic reviews. Guidelines with unclear methodology were also excluded.
Critical Appraisal of Individual Studies
The included publications were critically appraised by 1 reviewer using the Downs and Black checklist12 for randomized and non-randomized studies. Summary scores were not calculated for the included studies; rather, the strengths and limitations of each included publication were described narratively.
Summary of Evidence
Quantity of Research Available
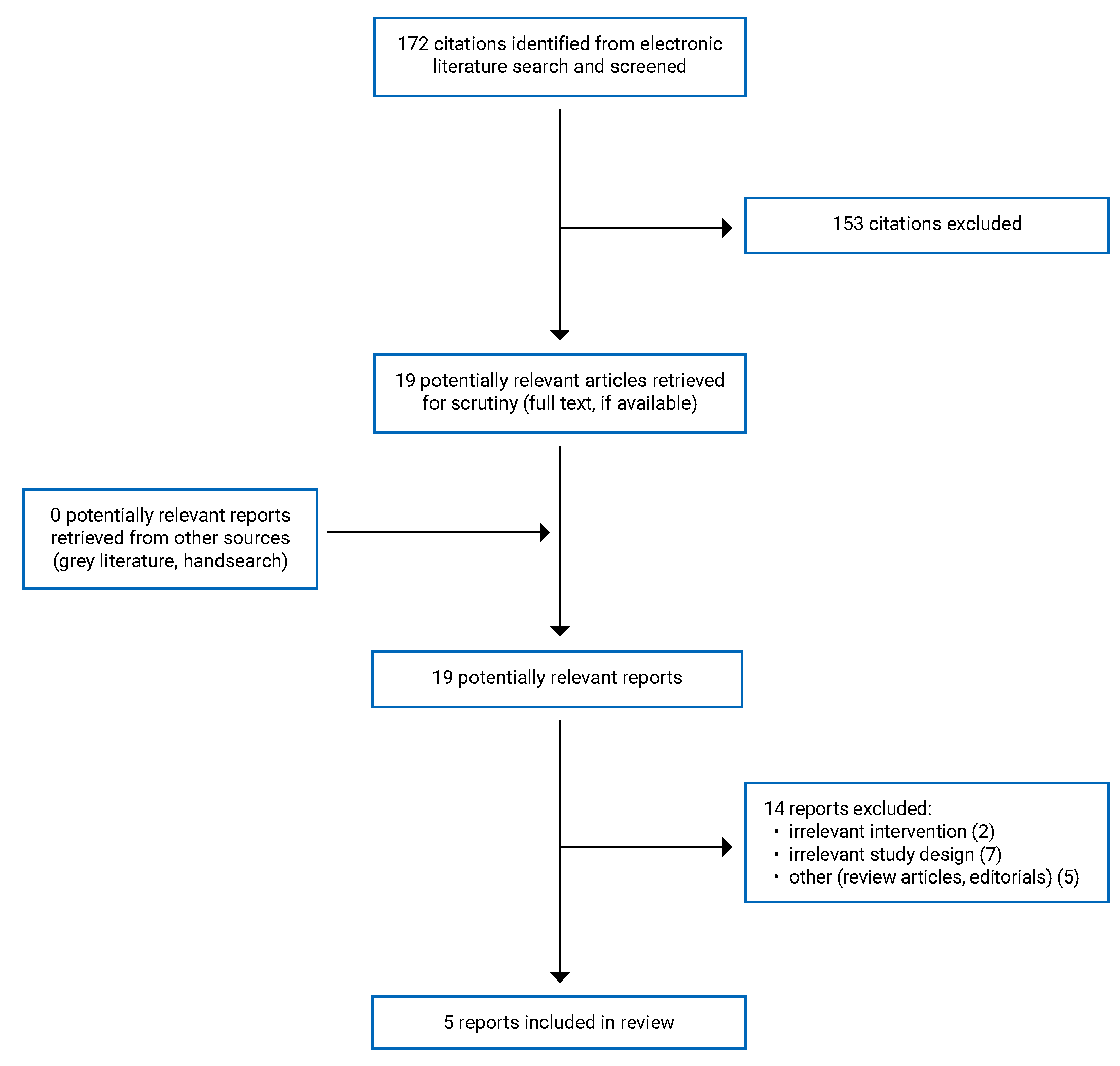
A total of 172 citations were identified in the literature search. Following screening of titles and abstracts, 153 citations were excluded and 19 potentially relevant reports from the electronic search were retrieved for full-text review. No potentially relevant publications were retrieved from the grey literature search for full-text review. Of the potentially relevant articles, 14 publications were excluded for various reasons, while 5 publications met the inclusion criteria and were included in this report. All were non-randomized studies. Appendix 1 presents the PRISMA13 flow chart of the study selection.
Additional references of potential interest are provided in Appendix 5.
Summary of Study Characteristics
The characteristics of the 5 primary studies14-18 are summarized in the subsequent sections, and additional details are provided in Appendix 2.
Study Design
All 5 primary studies14-18 were non-randomized studies. Four of the studies14-17 were retrospective cohort studies; 3 of these15-17 were uncontrolled (i.e., single-arm cohort studies), and the fourth14 included within-patient comparisons (i.e., the same patient may have received multiple lines of therapy) between outcomes associated with ruxolitinib, tocilizumab, and azacitidine, compared with corticosteroids. The fifth study18 was a single-arm prospective clinical trial, in which the diagnosis of VEXAS was made retrospectively because the UBA1 mutation associated with VEXAS was discovered after the trial had been started.
In 1 study,15 the study eligibility criteria aligned with the current report, while in the other 4 studies,14,16-18 the eligibility criteria were broader, yet the data were summarized for relevant subgroups.
All 5 studies14-18 were published in 202114 or 2022,15-18 with study periods ranging from July 201718 to February 202216 (where reported).
Country of Origin
All 5 primary studies14-18 included sites in France. Of these, 1 was a multi-country study,16 including patients treated in France, the US, and Portugal. The other 4 studies14,15,17,18 were exclusively conducted in France: 1 was conducted at the Lyon University Hospital,14 and the other 3 study populations were sourced from centres of the Groupe Francophone des Myélodysplasies (GFM) (3 studies),15,17,18 the Médecine interne, hématologie et oncologie (MINHEMON) group (2 studies),15,17 and/or the Filière de santé des maladies auto-immunes et auto-inflammatoires rares (FAI2R) group (1 study).17
Patient Population
In 3 of the 5 primary studies,15,17,18 study eligibility criteria restricted the patient population to those with specific hematologic or autoinflammatory conditions. One study15 was restricted to patients who had a documented diagnosis of MDS, 1 study17 was restricted to patients with relapsing polychondritis, and 1 study18 was restricted to patients with steroid-dependent or refractory SAID with MDS or chronic myelomonocytic leukemia (although no patients in the subgroup relevant to the current report had chronic myelomonocytic leukemia).
The median age of patients, either at symptom onset or at treatment initiation, was between 64 and 68 years in 4 of the studies14-17 and 76 years in 1 study.18 All included patients were male, with the possible exception of 2 female patients in 1 study17 who had acquired monosomy X (although in this study, patient characteristics were not available by treatment, and hence these female patients may not have been included in the subgroup relevant to the current report).
MDS was present in 100% of patients of 2 studies,15,18 by design (i.e., according to study eligibility criteria). In the other 3 studies,14,16,17 between 40%16 and 75%17 of patients were reported to have MDS. The Revised International Prognostic Scoring System (IPSS-R) for Myelodysplastic Syndromes Risk was reported among patients with MDS in 4 studies;14-16,18 the majority of those patients were categorized as having low or very low risk scores.
In the 4 retrospective cohort studies,14-17 fever was reported as being present in the majority of patients, ranging from 60%17 to 91%,15 while in the 1 single-arm clinical trial, 8% of patients were reported to have fever at baseline.18
Median baseline C-reactive protein levels were elevated in all studies, ranging from 30 mg/L to 117 mg/L, while hemoglobin and platelet levels tended to be low, with study-level medians ranging from 94 mg/L to 103 mg/L, and from 108 × 109/L to 211 × 109/L, respectively.14-18
Interventions and Comparators
None of the 5 primary studies14-18 was designed to directly compare outcomes between discrete groups of patients treated with the interventions or comparators of interest in this report. Four14-16,18 of the 5 studies did not include a control group; outcomes for 1 relevant intervention group were reported (either azacitidine14,15,18 or ruxolitinib).16 The fifth study14 was a retrospective cohort of 11 patients who were each treated with up to 6 lines of therapy, including all 3 interventions of interest in this report (azacitidine, ruxolitinib, and tocilizumab), as well as the comparator of interest (corticosteroids). The study authors compared outcomes between these interventions and corticosteroids; however, the comparisons did not involve mutually exclusive patient populations (i.e., for a patient who received both azacitidine and corticosteroids, that patient’s outcomes following azacitidine, and following corticosteroids, were included in the comparison).
Four studies14,15,17,18 evaluated the use of azacitidine. In 2 of the studies,15,18 azacitidine was the only treatment considered, and it was administered at 75 mg/m2 per day in both studies. In the clinical trial,18 the protocol-specified treatment was that azacitidine was to be delivered for 7 days, over six 4-week cycles, or until overt disease progression or a major adverse event. In the retrospective cohort study,15 azacitidine was given for 5 to 7 days, for a median of 11 cycles (range, 2 cycles to 35 cycles; cycle length not defined). In the other 2 studies,14,17 azacitidine-specific subgroups were reported within a broader patient population. Dosing details were not provided, although the duration of treatment was reported in 1 of the studies,14 and ranged from 4.3 months to 5.9 months.
Two studies14,16 involved ruxolitinib, with dosing described in 1study.16 In that study, patients received a mean starting dosage of 15 mg per day (range, 10 mg to 20 mg), and the mean dosage at the last follow-up was 25.4 mg per day (range, 10 mg to 50 mg). In the other study,14 dosing was not described; however, the duration of treatment was reported for the 2 patients who received ruxolitinib (1.8 months and 4.5 months).
The use of tocilizumab was described in 1 study,14 for 4 patients. Dosing details were not provided, but the duration of treatment was reported for those 4 patients as 0.3 months, 2.8 months, 8.5 months, and 29.8 months.
Although corticosteroids were used in all 5 studies, corticosteroid use was described as a baseline characteristic in 4 of these studies,15-18 rather than as a study comparator. In the 1 remaining study,14 treatment with corticosteroids and associated outcomes were described; however, corticosteroid use was concurrent with or in sequence with other interventions of interest, within the same patient, which did not enable comparison between independent groups of patients receiving different treatments, or receiving treatments over distinct time periods. In that 1 study,14 dosing details were not provided; however, the median duration of treatment was reported to be 14.8 months (range, 1.0 months to 71.8 months).
Outcomes
Response
Treatment response was reported as an outcome in 4 out of 5 studies,15-18 measured according to MDS-related hematological parameters and/or inflammatory markers of disease.
MDS-related response was reported in 4 studies.15-18 It was defined according to the 2006 international working group (IWG) criteria19 in 2 studies,15,18 and was not defined in the other 2.16,17 According to the 2006 IWG criteria, MDS-related response requires attainment of peripheral blood target levels for hemoglobin (≥ 11 g/dL), platelet (≥ 100 × 109/L), and neutrophil levels (≥ 1.0 × 109/L), and serum blood blasts of 0%. For complete remission, there must also be a maximum of 5% myeloblasts in the bone marrow, with normal maturation of all cell lines; while for partial remission, the bone marrow blasts must have decreased by 50% or more over pre-treatment, but may still be greater than 5%. Stable disease is defined as failure to achieve at least partial remission, but with no evidence of progression (defined as worsening of cytopenias, increase in percentage of bone marrow blasts, or progression to a worse prognostic category) for more than 8 weeks. In addition, the 2006 IWG criteria include a separate category for “hematologic improvement” in which the targets for complete or partial remission are not all reached, but improvements are seen. Hematologic improvements are defined separately for the 3 hematopoietic lineages: erythroid, platelet, and neutrophil. For erythroids, a hematologic improvement is defined as an increase by 1.5 g/dL (15 g/L) or more, or a reduction of at least 4 red blood cell (RBC) transfusions over 8 weeks. For platelets, a hematologic improvement is defined as an increase of 30 × 109/L or more (if baseline levels are < 20 × 109/L), or an increase from less than 20 × 109/L to greater than20 × 109/L, and by at least 100%. For neutrophils, hematologic improvement is defined as a minimum 100% increase and an absolute increase greater than 0.5 × 109/L.
Inflammatory-related treatment response was reported in 3 studies,16-18 all of which incorporated both clinical (e.g., resolution of signs and symptoms) and laboratory (e.g., full or partial reduction in C-reactive protein levels) measures of response. In the single-arm trial by Mekinian et al., in which all patients had SAID and were steroid-dependent or steroid-refractory, autoinflammatory-related treatment response was the study’s primary outcome. Complete SAID response was defined as: absence of new signs and complete disappearance of previous signs; negativation of acute phase reactants (e.g., serum C-reactive protein); and normalization of the disease-specific immunological parameters (if appropriate).18 Partial SAID response was defined as: greater than 50% improvement of previous clinical signs, greater than 50% decrease of acute phase reactants, and greater than 50% decrease of immunological parameters (if appropriate).18 Heiblig et al.16 reported on clinical and biologic response separately, with clinical response defined as complete resolution of clinical symptoms, and biologic response defined according to C-reactive protein levels (complete normalization for complete response, and ≥ 50% reduction for partial response). Khitri et al.17 did not provide a definition for inflammatory-related treatment response, but the results included 2 categories of response: 1 group of patients was described as having complete clinical and laboratory response (not further defined), and the other group as having at least 50% improvement of clinical and laboratory C-reactive protein parameters.
In 1 study,15 the response outcome definition encompassed both MDS-related hematologic parameters and inflammatory-related parameters, and was labelled as VEXAS response. Major VEXAS response was defined as complete clinical remission (assumed to be defined according to MDS-related parameters), and a minimum 50% steroid dose reduction to less than 10 mg per day during 1 month or more. Minor VEXAS response was defined as a 50% steroid dose reduction but to more than 10 mg per day, during 1 month or more (unclear if the definition of minor VEXAS response also required complete or partial MDS remission).
Time to Next Treatment
Two studies14,15 reported the time to next treatment, with Bourbon et al.14 further specifying this as the time to the addition of a new steroid-sparing agent.
Corticosteroid Dose Reduction
Dose reductions and discontinuations of corticosteroids were identified as key outcomes of treatment, and were reported in 3 studies.15,16,18
Mortality
Deaths were reported in 4 of the 5 studies.14-16,18 In 118 study, overall survival was reported as a time-to-event outcome and presented using a Kaplan-Meier curve. In 3 other studies,14-16 the number of deaths was reported.
Adverse Events
Adverse events were reported in narrative format in 2 studies.15,16 In a third study (the single-arm trial),18 a tabular summary of all treatment-emergent grade 3 to 4 adverse events was provided. Although all patients in that trial were treated with azacitidine, the tabular summary captured a broader patient population (i.e., including VEXAS and non-VEXAS patients) than is relevant to the current report.
Summary of Critical Appraisal
In all 5 primary studies,14-18 the reporting was clear for the objectives, patient characteristics, main outcomes, and/or adverse events. However, 3 of the 5 studies14,16,18 were published as letters to the editor, and 1 study15 was published as a “short report”; the more limited word count associated with these publication types resulted in some study-related details being omitted. Specifically, details on the outcome definition were unclear in 2 studies,15,16 and dosing details for the intervention and control groups were lacking in 2 studies.14,17 A further limitation in reporting relates to the objectives of the current report. Four14,16-18 of the studies captured a broader patient population than is relevant to the current report. Patient characteristics were not reported for the subgroup of interest in 2 of these studies,16,17 although they were available in the third18 study, and were obtained from an individual patient-level supplemental table in the fourth14 study. Adverse events for the relevant patient population were reported in the 1 study15 for which the full patient population aligned with the relevant population for the current report. In 2 other studies,16,18 adverse event data were comprehensively reported for the overall population; however, for the subgroup relevant to the current report, adverse events were reported only anecdotally (in 1 study)16 or not at all (1 study).18 Adverse events were not reported at all in 2 other studies.14,17 Outcomes were well defined in most studies; however, certain details regarding outcome definitions were lacking in 2 of the studies,15,16 and in 1 study,17 the results reporting was unclear and may have involved an error in the reported denominator.
A main limitation in all 5 studies14-18 was the non-comparative (i.e., uncontrolled) nature of the study designs. The 1 clinical trial18 was designed as a single-arm trial, and 3 of the 4 retrospective cohort studies15-17 did not include a comparator group of patients receiving corticosteroids. Treatment effects in uncontrolled studies are susceptible to a high risk of bias, as observed outcomes may not be attributable to the intervention alone, as there may be uncontrolled factors (e.g., the patient population) that may have contributed to the findings of these studies. The fourth14 retrospective cohort study did involve within-study comparisons between relevant interventions (ruxolitinib, tocilizumab, azacitidine) and corticosteroids as a comparator group; however, these comparisons were not between mutually exclusive patient populations. Patients in this study received up to 6 lines of therapy, and the treatments of interest could be received sequentially or concurrently for the same patient. Studies involving within-patient comparisons of treatment over time (e.g., crossover trials) may also result in biased estimates of effect, especially when treatments have carry-over effects and/or are taken concurrently. Furthermore, such studies may be poorly suited to progressive diseases like VEXAS, as disease progression over time may confound the relationship between treatments and outcomes. These threats to the internal validity of comparisons between intervention and comparator groups indicate that comparisons between treatments in this study14 should be interpreted with caution. None of the 5 primary studies involved random allocation to treatment groups, and none involved blinding of patients, investigators, or outcome assessors. Based on these study design features, the findings from these studies provide absolute estimates of effect, but do not enable a robust comparison of the relative benefits and harms of different VEXAS treatments.
There were several other notable limitations that impacted the internal and external validity of the evidence base. One such limitation was the sample size, which ranged from 2 to 17 for any given treatment group; sample size calculations were not reported for any study. Additionally, the retrospective nature of the studies may have led to inaccuracies and missing data relating to treatment and outcomes, as the datasets were not designed specifically to evaluate VEXAS-related treatments or outcomes.
With regard to the application of findings to the Canadian setting, the external validity of all studies is somewhat limited, given that all 5 studies were conducted in France, with 1 study16 also including US- and Portugal-based study sites. The extent to which findings will generalize to expected outcomes in Canadian centres is unclear.
Additional details regarding the strengths and limitations of included publications are provided in Appendix 3.
Summary of Findings
Appendix 4 presents the main study findings.
Clinical Effectiveness of Tocilizumab for Patients Diagnosed With VEXAS
Response
No studies involving tocilizumab reported on treatment response.
Time to Next Treatment
The time to the addition of a new steroid-sparing agent after tocilizumab was reported in 1 study.14 Among the 4 patients who received a course of tocilizumab (for 0.3 months to 29.8 months), the median time to next treatment was 8 months (range, 0.4 months to 37 months). In a narrative comparison, Bourbon et al.14 compared this with the time to next treatment for the 10 patients in their study who received corticosteroids (4 of whom also received tocilizumab); the median time to next treatment from corticosteroid initiation was 3.9 months. The authors summarized that based on these median times to next treatment, both tocilizumab and corticosteroids were considered “transiently effective”14 treatments.
Steroid Dose Reduction
No studies involving tocilizumab reported on steroid dose reduction.
Mortality
Deaths were reported in 1 study14 involving tocilizumab treatment: 1 patient of the 4 patients who received a course of tocilizumab had died by the end of follow-up; however, timing of death with respect to having received tocilizumab was not specified. In the same study, in the comparator group of patients receiving corticosteroids (in which 4 patients also received a course of tocilizumab), 30% of patients died; however, deaths were not presented comparatively between treatment groups, given that 4 patients belonged to both groups and outcomes were not specific to a particular line of treatment.
Adverse Events
No studies involving tocilizumab reported on adverse events.
Clinical Effectiveness of Ruxolitinib for Patients Diagnosed With VEXAS
Response
Response was reported in 1 study16 involving a group of patients with VEXAS who were treated with ruxolitinib, with no control group relevant to this review. Response outcomes included both MDS-related and autoinflammatory-related treatment response outcomes.
In terms of MDS response, Heiblig et al.16 reported that 17% of the 12 ruxolitinib-treated patients in their study had MDS progression while on treatment, although they did not report how many patients had MDS complete or partial remission, or stable disease.
In terms of autoinflammatory-related treatment response, Heiblig et al.16 reported that at 1, 3, and 6 months follow-up, almost all (87% to 92%) of the patients who remained on treatment with ruxolitinib achieved a clinical response, with the majority of responses categorized as complete. Similarly, almost all (87% to 92%) of the patients had a biologic response at 1, 3, and 6 months follow-up, categorized as either complete (38% to 50%) or partial (42% to 50%).
Time to Next Treatment
Time to next treatment after ruxolitinib was reported in 2 studies,14,16 involving a total of 14 patients. In both studies, the majority of patients had not initiated a new therapy by the end of the study period; the median was not reached among 12 patients in 1 study,16 and there was no reported next treatment (and hence the median was not reached) in 2 patients in the other14 study. The former study16 did not include a relevant control group; however, in the latter study,14 the authors provided a narrative comparison of the median time to next treatment for ruxolitinib. These authors14 contrasted the “interesting result”14 that the median was not reached against the time to next treatment associated with corticosteroids (3.9 months; note that 2 patients also received ruxolitinib), which was described by the authors as reflecting the “transiently effective”14 nature of corticosteroids.
Steroid Dose Reduction
Steroid dose reductions and discontinuations were reported in 1 study16 involving ruxolitinib treatment (with no relevant control group). In this study, an 83.6% reduction in daily steroid dose was reported for the subset of patients who remained on treatment after 6 months. Two patients (out of 12) in that study were able to discontinue corticosteroids altogether.
Mortality
Deaths were reported in 2 studies14,16 involving ruxolitinib treatment. In 1 study,16 which did not have a control group relevant to this report, 2 patients taking ruxolitinib died (17%): 1 patient died from colon cancer progression, and 1 died from an undetermined cause. In the other study,14 no deaths were reported among the 2 patients who received ruxolitinib. In the comparator group of that study,14 30% of patients who had received corticosteroids had died by the end of follow-up; however, deaths were not presented comparatively between treatment groups, given that 2 patients belonged to both groups and outcomes were not specific to a particular line of treatment.
Adverse Events
Adverse events were reported in 1 study16 involving ruxolitinib treatment, in which 2 patients (17%) had venous thromboembolism.
Clinical Effectiveness of Azacitidine for Patients Diagnosed With VEXAS
Response
Hematological MDS-related response or improvement was captured in 3 of the 4 studies involving azacitidine;15,17,18 none of the studies involved a relevant control group. The proportion of patients with complete remission or complete response was reported in 2 of these studies: 33% of patients had complete response in the single-arm trial by Mekinian et al.18 (according to IWG 2006 criteria), and 73% of patients had a complete response in the study by Khitri et al.17 (definition not reported). Hematologic improvement associated with azacitidine treatment was reported in 42%18 and 45%15 of patients in 2 studies,15,18 with improvements in erythroids (27%) and/or platelets (27%) in 1 of the studies.15
Inflammation-related treatment response was reported in 2 studies,17,18 among VEXAS populations having either relapsing polychondritis and steroid dependence (1 study)17 or SAID (1 study).18 Neither study included a relevant control group. Among these patients, 25%18 and 50%17 had a complete response to azacitidine, with a further 50%17,18 of patients in both studies having a partial response.
VEXAS response, reported in 1 single-group cohort study,15 was achieved by 46% of patients, categorized as either major response (18%), or minor response (27%). Among patients with VEXAS response, the time to response was after 4 cycles (n = 4) or after 6 cycles (n = 1), and the duration of VEXAS response ranged from 6 months to 27 months; 2 patients’ responses were ongoing at the end of the study period.
Time to Next Treatment
Time to next treatment after azacitidine was reported in 2 studies.14,15 Bourbon et al.14 reported the median time to next treatment after azacitidine as 21.9 months, which they noted as an “interesting finding”14 relative to the “transiently effective”14 findings associated with corticosteroids (median of 3.9 months). However, it should be noted that the median time to next treatment after azacitidine was based on 4 patients, with individual times of 0.8 months, 4.1 months, and 39.7 months, and with 1 patient having no next treatment captured by the end of the study period. The median time to next treatment following azacitidine in the second15 study was 7.5 months among the 73% who received a new treatment following azacitidine, with individual times ranging from 1 month to 25 months; the remaining 27% of patients who remained on treatment at the end of the study period had received 11 to 35 cycles of azacitidine. This latter study was a single-group cohort study (i.e., uncontrolled study).
Steroid Dose Reduction
Steroid dose reductions or discontinuations were reported in 2 studies15,18 involving azacitidine treatment; neither study included a control group relevant to this report. In these studies, 91% to 100% of patients were steroid dependent or steroid refractory at baseline, and in both studies, a trend toward reduced steroid dose after azacitidine initiation was observed. Furthermore, in 1 study,15 1 patient was reported as having discontinued corticosteroids altogether. In the second18 single-arm study, data were provided in a graph format for the subgroup relevant to the current report but summary statistics were not provided; however, it is noteworthy that in the overall population of that study, in which all patients were steroid dependent or refractory and all patients received azacitidine, a statistically significant reduction in steroid dose was reported, from a median of 50 mg per day at baseline to 10 mg per day after 6 cycles of treatment.
Mortality
Deaths were reported in 3 studies involving azacitidine treatment.14,15,18 In 1 of the 3 studies,14 no deaths were reported among the 4 patients who received azacitidine. In the other 2 studies,15,18 1 patient treated with azacitidine in each study died, representing 8%18 to 9%15 of the relevant study sample. In 1 study,15 this was attributed to an infection following ASCT, rather than during treatment with azacitidine. In the second18 study, the cause of death was not stated; in a visual presentation of patient-specific data (presented as a swimmer plot), the death was shown to occur approximately 3 months after a 4-month course of azacitidine.
Adverse Events
Two15,18 studies involving azacitidine reported on adverse events, although 1 study captured a broader patient population than is relevant to the current report. In 1 study,15 the reported serious adverse events were: pneumocystis infection, severe colitis, and bacterial pneumonia. In the study capturing adverse events among a broader patient population, the most common treatment-emergent grade 3 to 4 adverse event was neutropenia.18
Evidence-Based Guidelines Regarding Treatment Options for Patients Diagnosed With VEXAS
No relevant evidence-based guidelines regarding treatment options for patients diagnosed with VEXAS were identified; therefore, no summary can be provided.
Limitations
The present review captures the emerging evidence relating to treatment options for the newly described VEXAS syndrome; however, the evidence base has several notable limitations.
One limitation was the absence of prospective studies and randomized controlled trials within the evidence base; all 5 studies14-18 in the evidence base were non-randomized studies that lacked an independent control group, and included retrospectively identified patients with VEXAS.14-18 At the time this report was written, VEXAS syndrome had been established for slightly less than 2 years; thus, there may have been insufficient time for designing, implementing, and disseminating results from high-quality randomized trials or prospective observational studies that have a reasonable duration of follow-up (e.g., to the end of a 6-month course of treatment). As a result, all studies in this review included retrospectively identified patients, who were identified from a limited number of centres that conducted genetic testing to identify patients with VEXAS syndrome. A new international registry has recently been developed for VEXAS patients by the Autoinflammatory Diseases Alliance (AIDA), which will capture both retrospective and prospective observational data relating to VEXAS treatment, and may help to address this paucity of high-quality evidence.5 However, randomized clinical trials may be more challenging to design and implement, and it may be years before trial results can be expected.4 Heiblig et al. 16discussed many of the challenges associated with clinical trials for VEXAS syndrome, highlighting issues relating to the clinical heterogeneity of the disease (e.g., variability in hematologic abnormalities and presence of MDS; severity of the autoinflammatory syndrome), and the need to define valid and accurate criteria for treatment response upon which trials can be powered.4 This review did not identify any planned or ongoing randomized trials registered in clinical trial registries (refer to Appendix 5); however, 1 non-randomized phase II trial evaluating the efficacy of ASCT was initiated in October 2022 and expected to be complete in 2025.20
A second noteworthy limitation of the evidence was the sample size (with all relevant study groups having fewer than 18 patients) and limited geographic variability. Although statistical uncertainty was rarely reported within the evidence base, in general there is increased statistical uncertainty as sample sizes get smaller, and hence the estimated outcomes (e.g., response rates) are likely imprecise. Furthermore, the limited geographic variability may have limited the external validity of the findings when considering a Canadian setting. An additional concern regarding the limited geographic distribution is the shared source populations across studies. Many patients were identified from the MINHEMON and GFM centres in France, and as a result, there was overlap in the included patients across the studies. This was explicitly noted by Comont et al.,15 who noted that 2 of the 4 patients with MDS who were treated with azacitidine in the study by Bourbon et al.14 were also included in their cohort of 17 patients.15 However, certain characteristics of the studies limited the degree of overlap (e.g., different patient eligibility criteria and different interventions being evaluated), and hence it is not expected that there was full overlap of patients between any 2 studies. Nevertheless, consistency of findings across multiple studies may reflect similarities in the study samples, rather than corroborate findings across independent populations.
A third limitation was the lack of standardization of line of therapy or time since disease onset across patients and studies. Given the retrospective nature of the studies, many of the patients had undiagnosed VEXAS at the time of treatment, and were refractory to treatment for underlying conditions such as MDS, relapsing polychondritis, and/or autoinflammatory disease. As such, these patients had received multiple lines of therapy; however, they were not delivered in a standardized sequence. With a progressive disease such as VEXAS, patients receiving later lines of therapy may have more severe disease, and therefore, the timing of treatment with respect to disease onset may be related to prognosis. As this feature was not controlled for across the patients or studies, it may have introduced bias in the estimated outcomes, particularly if certain treatments are more likely to be given later in disease progression. Furthermore, some treatments may have been received concomitantly, and this was not always clear within the reported studies. For example, corticosteroids were provided concomitantly in the majority of studies, as a goal of therapy was in fact reducing the corticosteroid dose and/or steroid discontinuation.
Finally, no treatment guidelines were identified in this review. In a news article published by the American Society of Hematology (ASH) Clinical News magazine,21 it was stated that members of the French VEXAS registry group have been developing a draft treatment guideline for VEXAS syndrome, and also that they intend to initiate an international consortium with up-to-date treatment guidelines for VEXAS syndrome. A goal of the AIDA registry was also to create standardized treatment protocols for VEXAS.5
Conclusions and Implications for Decision- or Policy-Making
This report identified 1 single-arm clinical trial,18 3 uncontrolled retrospective cohort studies15-17 informing the clinical effectiveness and safety of ruxolitinib or azacitidine for patients with VEXAS (i.e., these studies did not include a comparator and were uncontrolled), and 1 retrospective study14 that included within-patient comparisons between ruxolitinib, tocilizumab, and azacitidine interventions versus a comparison group of corticosteroids. No relevant treatment guidelines were identified.
The resulting evidence base informing the effectiveness and safety of azacitidine, ruxolitinib, and tocilizumab was of limited quality due to the studies’ size, lack of independent control group, and retrospective nature. However, some findings were promising, albeit inconsistent.
The hematologic improvement among 42% to 45% of patients treated with azacitidine in 2 studies,15,18 and complete hematologic response among 73% of patients in a third17 study, shows promise that the benefits of azacitidine that have been seen with MDS may extend to VEXAS syndrome. Additionally, the partial and complete inflammation-related treatment response (observed in 75% to 100% of patients in 2 studies)17,18 following treatment with azacitidine was considered novel, and suggests that more research is needed to understand how the mechanism of action of azacitidine affects inflammation pathways.9,11 However, based on the initial findings, azacitidine does not appear promising as a curative option for VEXAS syndrome, although some researchers believe that it may provide an important bridge to ASCT, which may in turn provide a cure.11
The initial findings on the effectiveness of ruxolitinib showed promise,14,16 primarily for controlling autoinflammatory symptoms based on the observed 83.6% reduction in steroid dose and 17% discontinuation rate reported by Heiblig et al.,16 and based on the time to next treatment data from 2 patients in the study by Bourbon et al.14 However, data are still lacking with respect to its effect on hematologic parameters. In commentary that accompanied the publication by Heiblig et al.,16 it was stated that “more data are needed before we can have confidence in these provisional findings.”22
Tocilizumab is currently indicated for other inflammatory conditions;23 however, evidence on its effectiveness in treating VEXAS was limited (based on a subgroup of 4 patients in 1 study),14 and proved insufficient for drawing robust conclusions.
Despite the limited conclusions regarding the effectiveness of tocilizumab, ruxolitinib, and azacitidine that can be drawn from the evidence base, these initial findings provide insights that can help inform future study design, which can in turn inform future guidance on optimal treatment of VEXAS syndrome. As more patients are identified through genetic sequencing, and as large-scale international registries and consortia are established, the evidence base is expected to grow. Future exploration of novel treatments, including the possibility of curing VEXAS via gene editing, may be explored in the future.
References
1.Beck DB, Ferrada MA, Sikora KA, et al. Somatic Mutations in UBA1 and Severe Adult-Onset Autoinflammatory Disease. N Engl J Med. 2020;383(27):2628-2638. PubMed
2.Grayson PC, Patel BA, Young NS. VEXAS syndrome. Blood. 2021;137(26):3591-3594. PubMed
3.Barba T, Jamilloux Y, Durel CA, et al. VEXAS syndrome in a woman. Rheumatology (Oxford). 2021;60(11):e402-e403. PubMed
4.Heiblig M, Patel BA, Groarke EM, Bourbon E, Sujobert P. Toward a pathophysiology inspired treatment of VEXAS syndrome. Semin Hematol. 2021;58(4):239-246. PubMed
5.Vitale A, Caggiano V, Della Casa F, et al. Development and Implementation of the AIDA International Registry for Patients With VEXAS Syndrome. Front Med (Lausanne). 2022;9:926500. PubMed
6.Gudmundsson S, Singer-Berk M, Watts NA, et al. Variant interpretation using population databases: Lessons from gnomAD. Hum Mutat. 2022;43(8):1012-1030. PubMed
7.Zhao LP, Schell B, Sébert M, et al. Prevalence of UBA1 mutations in MDS/CMML patients with systemic inflammatory and auto-immune disease. Leukemia. 2021;35(9):2731-2733. PubMed
8.Koster MJ, Kourelis T, Reichard KK, et al. Clinical Heterogeneity of the VEXAS Syndrome: A Case Series. Mayo Clin Proc. 2021;96(10):2653-2659. PubMed
9.Gurnari C, McLornan DP. Update on VEXAS and role of allogeneic bone marrow transplant: Considerations on behalf of the Chronic Malignancies Working Party of the EBMT. Bone Marrow Transplant. 2022. PubMed
10.Khan C, Pathe N, Fazal S, Lister J, Rossetti JM. Azacitidine in the management of patients with myelodysplastic syndromes. Ther Adv Hematol. 2012;3(6):355-373. PubMed
11.Patel BA, Young NS. Towards treatments for VEXAS. Br J Haematol. 2022;196(4):804-805. PubMed
12.Downs SH, Black N. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J Epidemiol Community Health. 1998;52(6):377-384. PubMed
13.Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol. 2009;62(10):e1-e34. PubMed
14.Bourbon E, Heiblig M, Gerfaud Valentin M, et al. Therapeutic options in VEXAS syndrome: insights from a retrospective series. Blood. 2021;137(26):3682-3684. PubMed
15.Comont T, Heiblig M, Rivière E, et al. Azacitidine for patients with Vacuoles, E1 Enzyme, X-linked, Autoinflammatory, Somatic syndrome (VEXAS) and myelodysplastic syndrome: data from the French VEXAS registry. Br J Haematol. 2022;196(4):969-974. PubMed
16.Heiblig M, Ferrada MA, Koster MJ, et al. Ruxolitinib is more effective than other JAK inhibitors to treat VEXAS syndrome: a retrospective multicenter study. Blood. 2022;140(8):927-931. PubMed
17.Khitri MY, Guedon AF, Georgin-Lavialle S, et al. Comparison between idiopathic and VEXAS-relapsing polychondritis: analysis of a French case series of 95 patients. RMD Open. 2022;8(2). PubMed
18.Mekinian A, Zhao LP, Chevret S, et al. A Phase II prospective trial of azacitidine in steroid-dependent or refractory systemic autoimmune/inflammatory disorders and VEXAS syndrome associated with MDS and CMML. Leukemia. 2022. PubMed
19.Cheson BD, Greenberg PL, Bennett JM, et al. Clinical application and proposal for modification of the International Working Group (IWG) response criteria in myelodysplasia. Blood. 2006;108(2):419-425. PubMed
20.National Cancer Institute. NCT05027945: A Phase II Study of Allogeneic Hematopoietic Stem Cell Transplant for Subjects With VEXAS (Vacuoles, E1 Enzyme, X-linked, Autoinflammatory, Somatic) Syndrome. Bethesda (MD): ClinicalTrials.gov: https://clinicaltrials.gov/ct2/show/NCT05027945. Accessed 2022 Sep 23.
21.Azvolinksy A. The vexing VEXAS syndrome. 2022; https://ashpublications.org/ashclinicalnews/news/5960/The-Vexing-VEXAS-Syndrome. Accessed 2022 Sep 14.
22.Conway R. Ruxolitinib takes center stage for VEXAS syndrome. Blood. 2022;140(8):807-808. PubMed
23.Goyal A, Narayanan D, Wong W, et al. Tocilizumab for treatment of cutaneous and systemic manifestations of vacuoles, E1 enzyme, X-linked, autoinflammatory, somatic (VEXAS) syndrome without myelodysplastic syndrome. JAAD Case Rep. 2022;23:15-19. PubMed
Appendix 1: Selection of Included Studies
Appendix 2: Characteristics of Included Publications
Note that this appendix has not been copy-edited.
Table 2: Characteristics of Included Primary Clinical Studies
Study citation, country, funding source | Study design | Population characteristics | Intervention and comparator(s) | Clinical outcomes, length of follow-up |
|---|---|---|---|---|
Comont et al. (2022)15 France (centres of the MINHEMON group and GFM centres) Funding source: none stated | Study design: Uncontrolled retrospective cohort study of VEXAS Study period: Unclear start date of the study period. However, VEXAS was diagnosed retrospectively through genomic testing among patients with compatible symptoms (between December 2020 and May 2021). | Number patients: 11 Key eligibility criteria:
Key baseline characteristics:
| Intervention: Azacitidine at 75 mg/m2/day for 5 to 7 days (cycle duration not reported) Median number of cycles of azacitidine: 11 (range: 2 to 35). Time from MDS diagnosis to azacitidine onset
Comparator: none | Outcomes:
Follow-up: Median follow-up from azacitidine initiation was 32 months (range: 12 to 75). |
Heiblig et al. (2022)16 France, US, Portugal (total of 10 hospitals) Funding source: None | Study design: Uncontrolled retrospective cohort study Study period: April 2018 to February 2022 | Number patients: 12 (subgroup of ruxolitinib-treated patients with VEXAS, among 30 patients with VEXAS treated with any Janus kinase inhibitor) Key eligibility criteria:
Key baseline characteristicsb:
| Intervention: Ruxolitinib
Comparator: none | Outcomes:
Follow-up: Median duration of follow-up in the ruxolitinib-treated subgroup was 6.9 months (range: 1 to 41). |
Khitri et al. (2022)17 France (centres of the FAI2R group, the MINHEMON group, and GFM centres) Funding source: None | Study design: Uncontrolled retrospective cohort study Study period: December 2019 to June 2021 | Number patients: 17 (subgroup of azacitidine-treated patients with VEXAS, among 55 patients with VEXAS and 40 patients without VEXAS) Key eligibility criteria:
Key baseline characteristics (n = 55 patients with VEXAS)b:
| Intervention: Azacitidine Comparator: none | Outcomes:
Follow-up: Median duration of follow-up in the VEXAS group (55 patients) was 37 months (IQR: 15 to 76)b |
Mekinian et al. (2002)18 NCT02985190 France (18 centres) Funding source: Groupe Francophone des Myélodysplasies (GFM, French MDS group). Celgene Corporation supplied the study drug. | Study design: Prospective single-arm (uncontrolled) clinical trial, with retrospective identification of VEXAS Study period: July 2017 to June 2020 | Number patients: 12 (subgroup of patients with VEXAS, among total sample of 29 patients) Key eligibility criteria:
Key baseline characteristics: (VEXAS subgroup n = 12)
| Intervention: Azacitidine at 75 mg/m2 for 7 days, every 4 weeks for 6 cycles (or until overt disease progression or major adverse event) Concomitant treatment with prednisone, administered at 1 mg/kg for 1 month, followed by a gradual decrease over 6 months at the physician’s discretion Median number of cycles of azacitidine = 11 (range: 2 to 35). Comparator: none | Primary end point: response of SAID after 6 cycles of azacitidine.
Secondary end points:
|
Bourbon et al. (2021)14 France (Lyon University Hospital) Funding source: none | Study design: Retrospective cohort study Study period: Not reported | Number patients: 10 (subgroup of patients with VEXAS treated with tocilizumab (n = 4); azacitidine (n = 4); ruxolitinib (n = 2) and/or steroids (n = 10), among 11 patients with VEXAS treated with any intervention)d Key eligibility criteria:
Key baseline characteristics (n = 10):
| Intervention: Tocilizumab (n = 4), azacitidine (n = 4), and/or ruxolitinib (n = 2)d Duration of treatment, in months: median (range)
Comparator: 10 out of the cohort of 11 patients received corticosteroidsd Median duration of treatment, in months: 14.8 (range: 10.1, 71.8) | Outcomes: Time to next steroid-sparing treatment Follow-up: Median duration of follow-up was 25.1 months (IQR: 14.4 to 86.0)b |
CRP = C-reactive protein; FAI2R = Filière de santé des maladies auto-immunes et auto-inflammatoires rares; GFM = Groupe Francophone des Myélodysplasies; IPSS-R = Revised International Prognostic Scoring System; IQR = interquartile range; IWG = international working group; JAKi = Janus kinase inhibitor; MDS = myelodysplastic syndrome; MINHEMON = Médecine interne, hématologie et oncologie; NA = not applicable; NR = not reported; SAID = systemic autoinflammatory diseases; VEXAS = vacuoles, E1 enzyme, X-linked, autoinflammatory, somatic.
aFor the relevant subgroup captured in the current report.
bApplies to the broader study population; characteristics were not available for the study subgroup relevant to the report.
cNote: in the VEXAS cohort, no patients had chronic myelomonocytic leukemia.
dPatients received multiple treatments.
Appendix 3: Critical Appraisal of Included Publications
Note that this appendix has not been copy-edited.
Table 3: Strengths and Limitations of Clinical Studies Using the Downs and Black Checklist12
Strengths | Limitations |
|---|---|
Comont (2022)15 | |
|
|
Heiblig (2022)16 | |
|
|
Khitri (2022)17 | |
|
|
Mekinian (2022)18 | |
|
|
Bourbon (2021)14 | |
|
|
IPSS-R = Revised International Prognostic Scoring System; MDS = myelodysplastic syndrome; MINHEMON = Médecine interne, hématologie et oncologie; SAID = systemic autoinflammatory diseases; VEXAS = vacuoles, E1 enzyme, X-linked, autoinflammatory, somatic.
Appendix 4: Main Study Findings
Note that this appendix has not been copy-edited.
Table 4: Summary of Findings by Outcome — MDS Response
Study citation | Time point | Definition | Sample size | Response criteria for altering natural history n (%) | Criteria for HI n (%) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
CR | PR | SD | PD | HI | HI-E | HI-P | ||||
Azacitidine | ||||||||||
Comont (2022)15 | NR | IWG 2006 criteria | 11 | NRa | NRa | 6 (55)a | NRa | 5 (45)a | 3 (27) | 3 (27) |
Khitri (2022)17 | NR | MDS hematologic response | 17 | NR (73)b | NR (13)b | NR (13)b | NR | NR | NR | |
Mekinian (2022)18 | 6 cycles | IWG 2006 criteria | 12 | 4 (33)c | NR | 2 (17)c | 1 (8)c | 5 (42)c | NR | NR |
Ruxolitinib | ||||||||||
Heiblig (2022)16 | NR | MDS progression | 12 | NR | NR | NR | 2 (17) | NR | NR | NR |
CR = complete remission; HI = hematologic improvement; HI-E = hematologic improvement, erythroid; HI-P = hematologic improvement, platelet; IWG = international working group; MDS = myelodysplastic syndrome; NR = not reported; PD = progressive disease; PR = partial remission; SD = stable disease; VEXAS-RP = VEXAS (vacuoles, E1 enzyme, X-linked, autoinflammatory, somatic) with relapsing polychondritis.
aThe primary end point of this study was VEXAS response and there was some lack of clarity regarding MDS response, which was a secondary end point. Individual patient-level data were provided for all 11 patients, with a row for ‘Hematological response’ which was assumed to be MDS response. In total, 3 patients had responses listed as “not applicable”, 6 had responses categorized as SD with or without HI-E or HI-P, and one had HI alone (both HI-E and HI-P). None of the patients were listed as having CR or PR, which may contrast with the finding that 2 patients were reported as having a major VEXAS response, defined as requiring complete clinical remission and steroid dose reduction. However, it was unclear how complete clinical remission was defined for that end point, and hence these findings may not be contradictory.
bReported denominator was 17; however, no numerators were provided and the reported percentages do not align with any choice of numerator divided by 17. Result quoted from the text: “Seventeen of patients with VEXAS-RP (31%) had been treated by azacitidine. All of them had underlying MDS and experienced MDS hematological complete response (n=; 73%), partial response (n=; 13%) and none in 13%.”17
cMDS response was reported for each patient in a supplemental appendix; each VEXAS patient was listed as having one of 4 outcomes: CR, SD, PD, or HI.
Table 5: Summary of Findings by Outcome — VEXAS Response
Study citation | Time point | Definitions | Sample size | Response N (%) | ||
|---|---|---|---|---|---|---|
Major | Minor | None or NR | ||||
Azacitidine | ||||||
Comont (2022)15 | NR | Major: Complete clinical remission and ≥ 50% steroid reduction to < 10 mg/day during ≥ 1 montha Minor: ≥ 50% steroid reduction but not to < 10 mg/day, during ≥ 1 montha | 11 | 2 (18)b | 3 (27)b | 6 (55)a |
NR = not reported; VEXAS = vacuoles, E1 enzyme, X-linked, autoinflammatory, somatic.
aIt was unclear from the reporting whether minor response also required complete or partial remission, and whether complete clinical remission was based on the myelodysplastic syndrome international working group 2006 criteria for complete remission.
bDuration of response reported for the 5 patients as: 6, 8+, 12, 21, 27+ months (where “+” indicates the response was ongoing at the end of the study period).
Table 6: Summary of Findings by Outcome — Inflammation-Related Treatment Response
Study citation | Time point | Definitions | Sample size | Response N (%) | ||
|---|---|---|---|---|---|---|
Complete | Partial | None | ||||
Azacitidine | ||||||
Khitri (2022)17 | NR | Clinical and laboratory responsea | 10a | 5 (50) | 5 (50) | 0 (0) |
Mekinian (2022)18 | 6 cycles | SAID responseb | 12 | 3 (25) | 6 (50) | 3 (25) |
Ruxolitinib | ||||||
Heiblig (2022)16 | 1 month | Clinical | 12 | 8 (67) | 3 (25)c | 1 (8)c |
3 months | 12 | 10 (83) | 1 (8)c | 1 (8)c | ||
6 months | 8 | 6 (75)d | 1 (13)c | 1 (13)c | ||
1 month | Biologic (> 50% CRP reduction) | 12 | 6 (50)c | 5 (42)c | 1 (8)c | |
3 months | 12 | 6 (50)c | 5 (42)c | 1 (8)c | ||
6 months | 8 | 3 (38)c | 4 (50)c | 1 (13)c | ||
CRP = C-reactive protein; SAID = systemic autoinflammatory disease.
aIncluding CPR parameters; reported for a subgroup of steroid-dependent patients.
bComplete SAID response defined as: absence of new signs and complete disappearance of previous signs; negativation of acute phase reactants (e.g., serum C-reactive protein); and normalization of the disease-specific immunological parameters (if appropriate). Partial SAID response defined as: > 50% improvement of previous clinical signs; acute phase reactants (e.g., serum C-reactive protein) decrease of > 50%; and decrease of immunological parameters of > 50% (if appropriate).
cEstimates were digitized from a figure.
dThe value reported for complete clinical response in the text is 87%, whereas the figure shows that 87% represents the proportion of patients with either partial or complete response (which are shown as separate components of a stacked bar chart). Given the explicit breakdown in the figure, it was assumed that the value in the text refers to any response, rather than to complete response only.
Table 7: Summary of Findings by Outcome — Time to Next Treatment
Study citation | Sample size | Median (range), months | Patient-specific times to event, in months | Notes |
|---|---|---|---|---|
Azacitidine | ||||
Comont (2022)15 | 11 | 7.5 (1, 25)a | 1, 4, 4, 6, 9, 11, 16, 25, NA, NA, NA | NA indicates no next treatment |
Bourbon (2021)14 | 4b | 21.9 (0.8, 39.7)c | 0.8, 4.1, 39.7, NA | NA indicates no next treatment |
Ruxolitinib | ||||
Heiblig (2022)16 | 12 | Not reached | Kaplan-Meier curve provided in publication | |
Bourbon (2021)14 | 2b | NA, NA | No next treatment recorded | |
Tocilizumab | ||||
Bourbon (2021)14 | 4b | 8 (0.4, 37)c | 0.4, 3.5, 12.5, 37 | 1 additional patient was given 1 dose of tocilizumab but was not included in the outcome summaryd |
Corticosteroids | ||||
Bourbon (2021)14 | 10b | 3.9 (1.6, 11.8)c | 1.6, 2, 2.1, 2.4, 2.7, 5.3, 6.4, 9, 11.8, NA | NA indicates no next treatment |
NA = not applicable.
aObtained from individual patient-level data; the calculation of the median time to next treatment excludes patients whose treatment with azacitidine was ongoing (listed as NA) and may be higher if included. The 3 patients whose azacitidine treatment was ongoing had received 11, 19, and 35 cycles of azacitidine by the end of the study period.
bPatients are not unique (i.e., 1 patient may have received multiple treatments); however, the time to next treatment (defined as next steroid-sparing treatment) is specific to the time after each distinct line of therapy.
cMedian was reported by the author; range was acquired from the individual patient-level data. Time to next treatment data were also presented visually, as Kaplan-Meier curves that compared different therapeutic strategies against steroids.
dTime to next treatment was listed as “not informative (neutropenia)”14 for this patient.
Table 8: Summary of Findings by Outcome — Corticosteroid Dose Reduction
Study citation | Sample size | Outcome definition | Result | Notes |
|---|---|---|---|---|
Azacitidine | ||||
Comont (2022)15 | 5 | Median steroid dose (range), as prednisone equivalent, mg/day: final | 30 (20 to 70) | Among responders to azacitidine |
5 | Median steroid dose (range), as prednisone equivalent, mg/day: final | 10 (0 to 20) | Among responders to azacitidine | |
11 | Steroid discontinuation, n (%) | 1 (9) | None | |
Mekinian (2022)18 | 12 | Daily prednisone equivalent dose, by patient, at baseline after cycles 3, 6, 12, 18, and 24 | NR | A figure is provided in an appendix of the publication; however, quantitative summary statistics are not provided. The visual representation depicts a downward trend in the total corticosteroid dose.a |
Ruxolitinib | ||||
Heiblig (2022)16 | 9 | Median steroid dose reduction, among patients still on treatment at 6 months | 83.6% | A figure displaying the prednisone dose at months 1, 3, and 6, was provided in the publication, relative to baseline; however, quantitative estimates were not provided.b |
12 | Steroid discontinuation, n (%) | 2 (17) | None | |
NR = not reported.
aIn the overall study population (i.e., not restricted to the VEXAS subgroup), the prednisone equivalent steroid dose decreased from a median of 50 mg/day (95% Confidence Interval [CI], 40 to 71) at initiation of azacitidine to 15 mg/day (95% CI, 13 to 31) (P < 0.0001) and 10 mg/day (95% CI, 5 to 10) (P < 0.0001) after 3 and 6 cycles, respectively.
bIn the overall study population (i.e., not restricted to the ruxolitinib subgroup), the authors reported the mean corticosteroid dose/day at baseline to be 30 mg (range, 5 to 60).
Table 9: Summary of Findings by Outcome — Mortality
Study citation | Sample size | Outcome definition | Result | Notes |
|---|---|---|---|---|
Azacitidine | ||||
Mekinian (2022)18 | 12 | Median overall survival, months | Not reached | Presented with a Kaplan-Meier curve |
12 | 1 year overall survival, n (%) | 11 (92) | Presented with a Kaplan-Meier curve | |
12 | Total deaths, n (%) | 1 (8) | None | |
Comont (2022)15 | 11 | Total deaths, n (%) | 1 (9) | Patient died from infection after allogenic stem cell transplantation |
Ruxolitinib | ||||
Heiblig (2022)16 | 12 | Total deaths, n (%) | 2 (17) | 1 patient died from colon cancer progression, and 1 patient died from an undetermined cause |
Mixed / multiple treatments | ||||
Bourbon (2021)14 | 10 | Total deaths, n (%) | 3 (30) | Deaths were reported by patient, but it was unclear what therapy had been received before death. Among the 3 patients who died, all 3 had received corticosteroids, and one had received tocilizumab. |
Table 10: Summary of Findings by Outcome — Safety
Adverse Event | Comont (2022)15 N = 11 Azacitidine | Mekinian (2022)18 N = 12 Azacitidine | Heiblig (2022)16 N = 12 Ruxolitinib |
|---|---|---|---|
Serious adverse events | |||
Pneumocystis infection | 1 (9) | NRb | NR |
Severe colitis | 1 (9)a | NRb | NR |
Bacterial pneumonia | 1 (9)a | NRb | NR |
Adverse events | |||
Venous thromboembolism | NR | NRb | 2 (17) |
NR = not reported.
aSevere colitis and bacterial pneumonia were experienced by the same patient.
bGrade 3 to 4 treatment-emergent adverse events were reported in the full study population of patients with autoimmune/autoinflammatory disorders and myelodysplastic syndrome and who were treated with azacitidine (n = 29); the most frequent events were: neutropenia (30% of 81 events), sepsis (17% of 81 events), thrombocytopenia (14% of 81 events), and bacterial infection (10% of 81 events).
Appendix 5: References of Potential Interest
Note that this appendix has not been copy-edited.
This section captures additional evidence that may be of interest, which was not eligible for the summary in the main report, including ongoing trials for the treatment of VEXAS.
This additional evidence was identified using the same search as in the main report; however, the population, intervention, comparator, outcomes, and study design (PICOS) criteria were relaxed so as not to restrict studies according to interventions or comparators, and furthermore, clinical trials were not required to report results (i.e., ongoing trials identified in clinical trial registries or via published trial protocols were included). Additionally, case reports, case series, review articles, guidelines with unclear methodology, and general grey literature were considered for inclusion.
Previous CADTH Reports
None.
Systematic Reviews
None.
Randomized Controlled Trials
None.
Non-Randomized Studies
Uncontrolled Clinical Trials (Any Treatment)
NCT05027945, A Phase II Study of Allogeneic Hematopoietic Stem Cell Transplant for Subjects With VEXAS (Vacuoles, E1 Enzyme, X-linked, Autoinflammatory, Somatic) Syndrome. https://clinicaltrials.gov/ct2/show/NCT05027945. Accessed 2022 Sep 23.
Note: This is a non-randomized phase II trial sponsored by the National Cancer Institute. The planned start date is October 2022, with an estimated primary completion date of July 2025. The treatment of interest is allogeneic hematopoietic stem cell transplant, which will be administered to treatment-refractory adults with symptomatic VEXAS, using one of 2 reduced intensity regimens.
Case Studies and Case Series Involving Treatment With Tocilizumab, Ruxolitinib, and/or Azacitidine
US
Goyal A, Narayanan D, Wong W, et al. Tocilizumab for treatment of cutaneous and systemic manifestations of vacuoles, E1 enzyme, X-linked, autoinflammatory, somatic (VEXAS) syndrome without myelodysplastic syndrome. JAAD Case Rep. 2022;23:15-19. PubMed
Note: This was a case report (N = 1); the patient’s treatment included tocilizumab, and the study was conducted in the US.
Kao RL, Jacobsen AA, Billington CJ, et al. A case of VEXAS syndrome associated with EBV-associated hemophagocytic lymphohistiocytosis. Blood Cells Mol Dis. 2022;93:102636. PubMed
Note: This was a case report (N = 1); the patient’s treatment included ruxolitinib, and the study was conducted in the US.
Italy
Manzoni M, Bosi A, Fabris S, et al. Clinical, Morphological and Clonal Progression of VEXAS Syndrome in the Context of Myelodysplasia Treated with Azacytidine. Clin Hematol Int. 2022;4(1-2):52-55. PubMed
Note: This was a case report (N = 1); the patient’s treatment included azacitidine, and the study was conducted in Italy.
The Netherlands
van der Made CI, Potjewijd J, Hoogstins A, et al. Adult-onset autoinflammation caused by somatic mutations in UBA1: A Dutch case series of patients with VEXAS. J Allergy Clin Immunol. 2022;149(1):432-439.e4. PubMed
Note: This was a case series (N = 12) reported narratively; the patients received multiple types of treatment, including tocilizumab ( n = 6), azacitidine (n = 1), and/or corticosteroids (n = 12); some outcome data are provided in the narrative patient-specific summaries; the study was conducted in the Netherlands.
Raaijmakers MHGP, Hermans M, Aalbers A, et al. Azacytidine Treatment for VEXAS Syndrome. Hemasphere. 2021;5(12):e661. PubMed
Note: This was a case series (N = 3); the patients’ treatment included azacitidine, and the study was based in the Netherlands.
France
Delplanque M, Aouba A, Hirsch P, et al. USAID Associated with Myeloid Neoplasm and VEXAS Syndrome: Two Differential Diagnoses of Suspected Adult Onset Still's Disease in Elderly Patients. J Clin Med. 2021;10(23). PubMed
Note: This could be categorized as a retrospective cohort study or as a case series (N = 6); 3 patients’ treatment included azacitidine (n = 3); the study was France-based; the authors noted that all 3 patients taking azacitidine “were identified and included in French VEXAS group cohort and could have been used in other published studies”; hence, given the earlier publication date of this study, and inclusion of 4 other France-based studies involving azacitidine use, it was assumed that these 3 patients were captured in other publications and hence this study was excluded from the main report.
Germany
Cordts I, Hecker JS, Gauck D, et al. Successful treatment with azacitidine in VEXAS syndrome with prominent myofasciitis. Rheumatology (Oxford). 2022;61(5):e117-e119. PubMed
Note: This was a case report (N = 1); the patient was treated with azacitidine; the study was conducted in Germany.
Japan
Kirino Y, Takase-Minegishi K, Tsuchida N, et al. Tocilizumab in VEXAS relapsing polychondritis: a single-center pilot study in Japan. Ann Rheum Dis. 2021;80(11):1501-1502. PubMed
Note: This was a case series (N = 3); the patients’ treatment included tocilizumab, and the study was conducted in Japan.
Kunishita Y, Kirino Y, Tsuchida N, et al. Case Report: Tocilizumab Treatment for VEXAS Syndrome With Relapsing Polychondritis: A Single-Center, 1-Year Longitudinal Observational Study In Japan. Front Immunol. 2022;13:901063. doi:10.3389/fimmu.2022.901063 PubMed
Note: This was a case series (N = 3); the patients’ treatment included tocilizumab and the study was conducted in Japan and captures the same 3 patients as the 2021 publication by Kirino et al., but over a longer (1-year) period.
Case Studies and Case Series not Involving Treatment With Tocilizumab, Ruxolitinib, and/or Azacitidine
Australia
Islam S, Cullen T, Sumpton D, et al. VEXAS syndrome: lessons learnt from an early Australian case series. Intern Med J. 2022;52(4):658-662. PubMed
Note: This was a case series (N = 3); treatment was unclear and the study was conducted in Australia.
Pathmanathan K, Taylor E, Balendra J, Lim A, Carroll G. VEXAS syndrome: favourable clinical and partial haematological responses to subcutaneous abatacept therapy with 30-month follow-up. Rheumatology (Oxford). 2022;61(7):e174-e177. PubMed
Note: This was a case report (N = 1); the patient’s treatment included abatacept, and the study was conducted in Australia.
Belgium
Staels F, Betrains A, Woei-A-Jin FJSH, et al. Case Report: VEXAS Syndrome: From Mild Symptoms to Life-Threatening Macrophage Activation Syndrome. Front Immunol. 2021;12:678927. PubMed
Note: This was a case series (N = 2); the patients’ treatment was unclear (n = 1) or not relevant to the current report (siltuximab; n = 1) and the study was conducted in Belgium.
Canada
Dehghan N, Marcon KM, Sedlic T, Beck DB, Dutz JP, Chen LYC. Vacuoles, E1 enzyme, X-linked, autoinflammatory, somatic (VEXAS) syndrome: fevers, myalgia, arthralgia, auricular chondritis, and erythema nodosum. Lancet. 2021;398(10300):621. PubMed
Note: This was a case report (N = 1); the patient was treated with many therapies, including tocilizumab; the study was conducted in Canada.
China
Huang H, Zhang W, Cai W, et al. VEXAS syndrome in myelodysplastic syndrome with autoimmune disorder. Exp Hematol Oncol. 2021;10(1):23. PubMed
Note: This was a case report (N = 1); the patient was treated with immunosuppressants and the study was conducted in China.
Czech Republic
Ciferska H, Gregová M, Klein M, et al. VEXAS syndrome: a report of three cases. Clin Exp Rheumatol. 2022;40(7):1449. PubMed
Note: This was a case series (N = 3); the patients’ treatment was unclear and the study was conducted in the Czech Republic.
France
Diarra A, Duployez N, Fournier E, et al. Successful allogeneic hematopoietic stem cell transplantation in patients with VEXAS syndrome: a 2-center experience. Blood Adv. 2022;6(3):998-1003. PubMed
Note: This was a case series (N = 4) or cohort study; the patients’ treatment was ASCT and the study was conducted in France.
Loschi M, Roux C, Sudaka I, et al. Allogeneic stem cell transplantation as a curative therapeutic approach for VEXAS syndrome: a case report. Bone Marrow Transplant. 2022;57(2):315-318. PubMed
Note: This was a case report (N = 1); the patient’s treatment was ASCT and the study was conducted in France.
Magnol M, Couvaras L, Degboé Y, et al. VEXAS syndrome in a patient with previous spondyloarthritis with a favourable response to intravenous immunoglobulin and anti-IL17 therapy. Rheumatology (Oxford). 2021;60(9):e314-e315. PubMed
Note: This was a case report (N = 1); the patient was treated with immunoglobulin and anti-IL17 therapy, and the study was conducted in France.
Italy
Campochiaro C, Tomelleri A, Cavalli G, et al. Successful use of cyclosporin A and interleukin-1 blocker combination therapy in VEXAS syndrome: a single-center case series. Arthritis Rheumatol. 2022;74(7):1302-1303. PubMed
Note: This was a case series (N = not reported); the patient was treated with an unspecified IL-blocker and the study was conducted in Italy.
Japan
Matsubara A, Tsuchida N, Sakurai M, et al. A case of VEXAS syndrome with Sweet's disease and pulmonary involvement. J Dermatol. 2022;49(5):e177-e178. PubMed
Note: This was a case report (N = 1); the patient’s treatment was unclear and the study was conducted in Japan.
Matsuki Y, Kawai R, Suyama T, Katagiri K, Kanazawa N, Inaba Y. A case of VEXAS syndrome with myositis possibly associated with macrophage activation syndrome. J Dermatol. 2022;doi:10.1111/1346-8138.16535 PubMed
Note: This was a case report (N = 1); the patient’s treatment was unclear and the study was conducted in Japan.
Matsumoto H, Fujita Y, Fukatsu M, et al. Case Report: Coexistence of Multiple Myeloma and Auricular Chondritis in VEXAS Syndrome. Front Immunol. 2022;13:897722. PubMed
Note: This was a case report (N = 1); the patient was treated with lenalidomide hydrate and the study was conducted in Japan.
Uchino K, Kanasugi J, Enomoto M, et al. VEXAS syndrome. Int J Hematol. 2022;116(4):463-464. PubMed
Note: This was a case report (N = 1); the patient was treated with azathioprine and the study was conducted in Japan.
Sakuma M, Tanimura A, Yasui S, et al. A Case of polychondritis-onset refractory organizing pneumonia with cytopaenia diagnosed as VEXAS syndrome: the disease course of 7 years. Rheumatology (Oxford). 2021;60(10):e356-e359. PubMed
Note: This was a case report (N = 1); the patient’s treatment was unclear and the study was conducted in Japan.
Norway
Midtvedt Ø, Stray-Pedersen A, Andersson H, et al. A man in his sixties with chondritis and bone marrow failure. Tidsskr Nor Laegeforen. 2022;142(4). PubMed
Note: This was a case report (N = 1); the patient’s treatment was unclear and the study was conducted in Norway.
The Netherlands
van Leeuwen-Kerkhoff N, de Witte MA, Heijstek MW, Leavis HL. Case report: Up-front allogeneic stem cell transplantation in a patient with the VEXAS syndrome. Br J Haematol. 2022;doi:10.1111/bjh.18424 PubMed
Note: This was a case report (N = 1); the patient was treated with upfront allogeneic stem cell transplantation and the study was conducted in Norway.
US
Shaukat F, Hart M, Burns T, Bansal P. UBA1 and DNMT3A mutations in VEXAS syndrome. A case report and literature review. Mod Rheumatol Case Rep. 2022;6(1):134-139. PubMed
Note: This was a case report (N = 1); the patient’s treatment was unclear; the study was conducted in the US; a literature review accompanied the case report.
Koster MJ, Kourelis T, Reichard KK, et al. Clinical Heterogeneity of the VEXAS Syndrome: A Case Series. Mayo Clin Proc. 2021;96(10):2653-2659. PubMed
Note: This was a case series (N = 9); patients’ treatment included corticosteroids and immunosuppressants; the study was conducted in the US.
Guidelines and Recommendations
None
Review Articles
Gurnari C, McLornan DP. Update on VEXAS and role of allogeneic bone marrow transplant: Considerations on behalf of the Chronic Malignancies Working Party of the EBMT. Bone Marrow Transplant. 2022;doi:10.1038/s41409-022-01774-8 PubMed
Note: This is a review article that includes a section on therapeutic management.
Hernández-Rodríguez J, Mensa-Vilaró A, Aróstegui JI. Paradigm shift in monogenic autoinflammatory diseases and systemic vasculitis: The VEXAS syndrome. Med Clin (Barc). 2022;doi:10.1016/j.medcli.2022.06.018 PubMed
Note: This is a review article that includes a section on therapeutic management.
Watanabe R, Kiji M, Hashimoto M. Vasculitis associated with VEXAS syndrome: A literature review. Front Med (Lausanne). 2022;9:983939. PubMed
Note: This is a review article on vasculitis associated with VEXAS, including a section on current therapeutic approaches for VEXAS syndrome.
Grayson PC, Patel BA, Young NS. VEXAS syndrome. Blood. 2021;137(26):3591-3594. PubMed
Note: This is a review article VEXAS, published about 7 months after its first description. It includes a section on treatment of VEXAS based on early reports.
Heiblig M, Patel BA, Groarke EM, Bourbon E, Sujobert P. Toward a pathophysiology inspired treatment of VEXAS syndrome. Semin Hematol. 2021;58(4):239-246. PubMed
Note: This is a review article focusing on treatment of VEXAS based on disease pathophysiology, categorized according to whether the treatment targets the clone, blocks cytokines and pathways in the inflammatory response, or provides supportive care.
Additional References
Beck DB, Ferrada MA, Sikora KA, et al. Somatic Mutations in. N Engl J Med. 2020;383(27):2628-2638. PubMed
Note: This is the seminal paper describing VEXAS.
Patel BA, Young NS. Towards treatments for VEXAS. Br J Haematol. 2022;196(4):804-805. PubMed
Note: This is a commentary on the study of azacitidine by Comont et al. (included in main report).
Vitale A, Caggiano V, Della Casa F, et al. Development and Implementation of the AIDA International Registry for Patients With VEXAS Syndrome. Front Med (Lausanne). 2022;9:926500. PubMed
Note: This is a protocol for the newly developed AIDA International Registry for Patients With VEXAS.
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up to date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third-party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.
Questions or requests for information about this report can be directed to Requests@CADTH.ca