Drugs, Health Technologies, Health Systems
Reimbursement Recommendation
Durvalumab (Imfinzi)
Indication: Treatment of patients with resectable muscle invasive bladder cancer in combination with gemcitabine and cisplatin as neoadjuvant treatment, followed by adjuvant Imfinzi monotherapy treatment after radical cystectomy
Sponsor: AstraZeneca Canada Inc.
Final recommendation: Reimburse with conditions
Summary
What Is the Reimbursement Recommendation for Imfinzi?
Canada’s Drug Agency (CDA-AMC) recommends that Imfinzi be reimbursed by public drug plans for the treatment of patients with muscle invasive bladder cancer (MIBC) in combination with gemcitabine-cisplatin as neoadjuvant treatment, followed by Imfinzi as adjuvant monotherapy after radical cystectomy if certain conditions are met.
Why Did CDA-AMC Recommend Reimbursement?
The pan-Canadian Oncology Drug Review Expert Review Committee (pERC) determined that Imfinzi in combination with gemcitabine-cisplatin as neoadjuvant treatment, followed by Imfinzi as adjuvant monotherapy after radical cystectomy demonstrates acceptable clinical value versus neoadjuvant gemcitabine-cisplatin in patients with MIBC. Given that Imfinzi is expected to be an additive treatment to gemcitabine-cisplatin, acceptable clinical value refers to added value versus gemcitabine-cisplatin alone.
Evidence from 1 clinical trial (NIAGARA) demonstrated that in patients with MIBC, treatment with Imfinzi in combination with gemcitabine-cisplatin as neoadjuvant treatment, followed by Imfinzi as adjuvant monotherapy after radical cystectomy resulted in a statistically significant improvement in pathologic complete response (pCR), event-free survival (EFS) at 24 months, and overall survival (OS) at 36 months compared to neoadjuvant gemcitabine-cisplatin and no adjuvant therapy after radical cystectomy.
Which Patients Are Eligible for Coverage?
Imfinzi should only be covered for adults with stage T2N0-1M0 to T4aN0-1M0 bladder cancer who are eligible for surgery and have not received prior systemic chemotherapy or immunotherapy for the treatment of MIBC. Patients should also have a good performance status.
What Are the Conditions for Reimbursement?
Imfinzi should only be reimbursed if treatment is discontinued upon the occurrence of disease progression or unacceptable toxicity, if treatment is prescribed by clinicians with expertise and experience in treating bladder cancer, and the cost of Imfinzi is reduced.
Review Background
Disease background: MIBC is a serious type of bladder cancer in which the tumour has grown into the muscle layer of the bladder. This makes it more likely to spread to other parts of the body. About 12,300 new cases of bladder cancer were expected in Canada in 2024 and roughly 25% of these were expected to be MIBC at the time of diagnosis.
Indication and reimbursement request: Durvalumab (Imfinzi) has been approved by Health Canada for the treatment of patients with resectable MIBC in combination with gemcitabine-cisplatin as neoadjuvant treatment, followed by adjuvant Imfinzi monotherapy after radical cystectomy. The sponsor is seeking reimbursement for this patient population.
Drug under review: Durvalumab is a PD-L1 inhibitor that is administered by IV infusion. The dosage recommended in the product monograph for neoadjuvant treatment is a fixed dose of 1,500 mg every 3 weeks (21 days) for a maximum of 4 cycles in combination with gemcitabine-cisplatin depending on renal function status. Following radical cystectomy, durvalumab monotherapy is given at a fixed dose of 1,500 mg every 4 weeks for a maximum of 8 cycles. Patients with a body weight of 30 kg or less must receive weight-based dosing of durvalumab at 20 mg/kg until weight increases to greater than 30 kg or for a maximum of 12 total cycles.
Treatment costs: At the submitted price of $938.67 per 2.4 mL vial, the per-patient 28-day cycle cost of durvalumab is expected to be $15,644 for the neoadjuvant portion of treatment and $11,733 for the adjuvant portion of treatment, based on the Health Canada–recommended dosages. Durvalumab is indicated for use in combination with neoadjuvant gemcitabine-cisplatin; the per-patient 28-day cycle cost of the regimen is expected to be $17,624 for the neoadjuvant portion and $11,733 for the adjuvant portion, based on a body surface area of 1.85 cm2.
Highlights of Input From Interested Parties
The patient group input (from Bladder Cancer Canada and Association Cancer Vessie France) noted the following regarding impacts of the disease, unmet needs, and important outcomes:
The most frequently reported cancer-related symptom was urinary problems, including increased frequency, pain, and/or blood during urination.
The side effects of the available treatments include fatigue, nausea, and sometimes hospitalization.
The most important treatment goals for patients include controlling disease progression, preventing recurrence, improving OS, maintain health-related quality of life, and improved treatment tolerability.
The clinician groups (Bladder Cancer Canada and the Ontario Health [Cancer Care Ontario] Genitourinary Cancer Drug Advisory Committee) and the clinical experts consulted by CDA-AMC noted the following regarding unmet needs arising from the disease and place in therapy for the drug under review:
There is a need for bladder-sparing and curative treatments that improve response rates and reduce relapses.
Durvalumab would fit well within a perioperative treatment framework as a first-line treatment, administered as neoadjuvant therapy alongside cisplatin-based chemotherapy, and continued as adjuvant treatment following cystectomy.
The participating public drug programs raised potential implementation issues related to considerations for initiation, discontinuation, and prescribing of therapy; generalizability of trial populations to broader populations; care provision issues; and system and economic issues.
Recommendation
With a vote of 15 in favour to 0 against, pERC recommends that durvalumab be reimbursed for the treatment of patients with MIBC in combination with gemcitabine-cisplatin as neoadjuvant treatment, followed by Imfinzi as adjuvant monotherapy after radical cystectomy only if the conditions listed in Table 1 are met.
Table 1: Reimbursement Conditions and Reasons
Reimbursement condition | Reason | Implementation guidance |
|---|---|---|
Initiation (neoadjuvant therapy) | ||
1. Treatment with durvalumab in combination with gemcitabine-cisplatin as neoadjuvant therapy, should be initiated in adult patients who are eligible for surgery and meet all of the following criteria: 1.1. have clinical stage T2N0-1M0 to T4aN0-1M0 bladder cancer 1.2. have not received prior systemic chemotherapy or immunotherapy for the treatment of MIBC. | Evidence from the NIAGARA trial demonstrated that treatment with the durvalumab regimen resulted in a clinical benefit compared with gemcitabine-cisplatin alone in patients with these characteristics. | pERC agreed with the clinical experts that durvalumab may be added for patients already receiving chemotherapy. Otherwise, efforts should be made to initiate all treatments concurrently. The experts noted that patients typically receive 3 to 4 neoadjuvant cycles with chemotherapy, although the exact number may be adjusted based on clinical judgment. |
2. Patients should have a good performance status. | Patients with an ECOG performance status of 0 to 1 were included in the NIAGARA trial. | Patients with an ECOG performance status of 2 may be treated at the discretion of the treating clinician. |
Renewal (continuation with adjuvant therapy) | ||
3. Continued reimbursement of durvalumab adjuvant therapy should be based on meeting all of the following criteria: 3.1. completion of neoadjuvant durvalumab in combination with gemcitabine-cisplatin 3.2. completion of a surgical procedure for the bladder 3.3. initiation of adjuvant therapy no more than 6 months after surgery with no evidence of disease progression. | In the NIAGARA trial, patients were required to undergo a radical cystectomy to continue with adjuvant durvalumab. In the NIAGARA trial, adjuvant therapy was recommended to begin within 120 days after, and no earlier than 42 days after, radical cystectomy. However, commencing treatment no more than 6 months after surgery is considered appropriate based on expert opinion. | pERC agreed with the experts that adjuvant durvalumab is not recommended for patients who have completed surgery without receiving neoadjuvant durvalumab due to a lack of supporting data. pERC agreed with the experts that a bladder-sparing alternative to radical cystectomy, such as trimodal therapy, should not limit eligibility for adjuvant treatment with durvalumab. pERC agreed with the experts, who indicated that immunotherapy-based re-treatment may be appropriate for patients with a disease-free interval of at least 6 months after completing adjuvant durvalumab. |
Discontinuation | ||
4. Neoadjuvant treatment with durvalumab in combination with gemcitabine-cisplatin, followed by adjuvant durvalumab, should be discontinued upon the occurrence of any of the following: 4.1.disease progression 4.2. unacceptable toxicity 4.3. maximum number of 12 cycles of durvalumab. | In the NIAGARA trial, treatment was discontinued upon disease progression or unacceptable toxicity, which is consistent with clinical practice. Patients in the NIAGARA trial received a maximum of 12 cycles of durvalumab — neoadjuvant durvalumab plus gemcitabine-cisplatin every 3 weeks for 4 cycles, followed by adjuvant durvalumab monotherapy every 4 weeks for 8 cycles. | pERC agreed with the clinical experts, who indicated that it would be appropriate to continue durvalumab if neoadjuvant chemotherapy had to be discontinued. At least 1 cycle of gemcitabine-cisplatin should be administered with durvalumab in the neoadjuvant setting to be eligible for continued durvalumab monotherapy. |
Prescribing | ||
5. Neoadjuvant treatment with durvalumab in combination with gemcitabine-cisplatin, followed by adjuvant durvalumab, should be prescribed by clinicians with expertise and experience in treating bladder cancer. | This is meant to ensure that durvalumab is prescribed for appropriate patients and that adverse effects are managed in an optimized and timely manner. | Clinical experts suggested using durvalumab with noncisplatin regimens but noted that survival benefits were established with cisplatin-based chemotherapy. pERC acknowledged this input but noted that there is no evidence included in the submission to support the use of durvalumab in combination with noncisplatin regimens; therefore, they could not recommend it. pERC noted that jurisdictions may consider implementing weight-based dosing up to a cap for durvalumab, as follows: 20 mg/kg up to 1,500 mg every 3 weeks (in combination with gemcitabine-cisplatin) for 4 cycles before surgery, followed by 20 mg/kg up to 1,500 mg every 4 weeks for 8 cycles after surgery. |
Pricing | ||
6. A reduction in price | Using the CDA-AMC base-case analysis, the ICER for neoadjuvant durvalumab in combination with gemcitabine-cisplatin, followed by adjuvant durvalumab monotherapy was $65,194 per QALY gained when compared with neoadjuvant gemcitabine-cisplatin in the indicated population. A band 1a price reduction would be required to achieve cost-effectiveness at a $50,000 per QALY gained threshold. No price reduction would be required to achieve cost-effectiveness at a $100,000 per QALY gained threshold. Price reductions for any given willingness-to-pay threshold are available in the Main Report and Supplemental Material document. In the absence of comparative clinical evidence against ddMVAC or adjuvant nivolumab, the drug program cost of neoadjuvant durvalumab in combination with gemcitabine-cisplatin, followed by adjuvant durvalumab monotherapy should also be no more than ddMVAC or adjuvant nivolumab. | The CDA-AMC analysis is based on public list prices for all treatments. Further price reductions may be required if there are price arrangements (discounts) currently in place for any treatment included in the economic analysis. Likewise, further price reductions may be required to address the economic feasibility of adoption. If perioperative durvalumab remains more costly than ddMVAC or adjuvant nivolumab after applying a band 1 price reduction, and there is no evidence showing superior clinical benefit, pERC considers that there is no basis for accepting a price premium. This issue is underscored by patient and clinician input identifying ddMVAC and adjuvant nivolumab as relevant treatment options, a view supported by clinical experts and reflected in the sponsor’s BIA. |
Feasibility of adoption | ||
7. The economic feasibility of adoption of neoadjuvant durvalumab in combination with gemcitabine-cisplatin, followed by adjuvant durvalumab monotherapy must be addressed. | At the submitted price, the incremental budget impact of neoadjuvant durvalumab in combination with gemcitabine-cisplatin, followed by adjuvant durvalumab monotherapy is expected to be greater than $40 million in year 3. | — |
BIA = budget impact analysis; CDA-AMC = Canada’s Drug Agency; ECOG = European Cooperative Oncology Group; ICER = incremental cost-effectiveness ratio; ddMVAC = dose-dense methotrexate, vinblastine, doxorubicin, and cisplatin; MIBC = muscle invasive bladder cancer; pERC = pan-Canadian Oncology Drug Review Expert Review Committee; QALY = quality-adjusted life-years.
aRegarding the size of the price reduction required, band 1 = 1% to 24%, band 2 = 25% to 49%, band 3 = 50% to 74%, and band 4 = 75% or greater.
Rationale for the Recommendation
Clinical Value
Based on the totality of the clinical evidence, pERC concluded that durvalumab in combination with gemcitabine-cisplatin as neoadjuvant treatment, followed by durvalumab as adjuvant monotherapy after radical cystectomy demonstrates acceptable clinical value compared with neoadjuvant gemcitabine-cisplatin in patients with MIBC. Given that durvalumab is expected to be an additive treatment to gemcitabine-cisplatin, acceptable clinical value refers to added value versus gemcitabine-cisplatin alone.
Evidence from 1 randomized controlled trial (NIAGARA, N = 1063) demonstrated that treatment with durvalumab plus gemcitabine-cisplatin before surgery, followed by durvalumab alone after surgery likely results in added clinical benefit for patients with MIBC compared with gemcitabine-cisplatin alone before surgery in pCR, EFS at 24 months, and OS at 36 months. A post-hoc reanalysis demonstrated a pCR rate of 37.3% in the durvalumab group versus 27.5% in the gemcitabine-cisplatin group (odds ratio = 1.60; 95% confidence interval [CI], 1.22 to 2.08) in favour of the durvalumab group. The hazard ratio for EFS was 0.68 (95% CI, 0.558 to 0.817; P < 0.0001) in favour of the durvalumab group, indicating a 32% overall reduction in the risk of experiencing an EFS event. Finally, the interim analysis of OS resulted in a hazard ratio of 0.75 (95% CI, 0.59 to 0.93; P = 0.0106) in favour of the durvalumab group.
Based on the NIAGARA study, harms were consistent with the known safety profile of cisplatin-based chemotherapy and PD-L1 inhibitors, with no new safety signals identified.
The committee concluded that durvalumab plus cisplatin-based chemotherapy addresses some, but not all, of the unmet needs for patients living with MIBC. The results from the phase III NIAGARA trial showed clinically meaningful improvements in EFS, pCR, and OS, with moderate certainty evidence, which meets patients’ goals to delay recurrence, improve health-related quality of life, and extend survival. The committee agreed that harms were consistent with the known safety profile of cisplatin-based chemotherapies and PD-L1 inhibitors, with no new safety signals identified. Overall, the drug meets some important unmet needs identified by patients (improvements in EFS, pCR, and OS).
Further information on the committee’s discussion around clinical value is provided in the Summary of Deliberation section.
Developing the Recommendation
The determination of acceptable clinical value was sufficient for pERC to recommend reimbursement of durvalumab. As part of the deliberation on whether to recommend reimbursement, the committee also considered unmet clinical need, unmet nonclinical need, and health inequity. Information on this discussion is provided in the Unmet Clinical Need and Distinct Social and Ethical Considerations domains in the Summary of Deliberation section.
Because pERC recommended that durvalumab be reimbursed, the committee also deliberated on whether reimbursement conditions should be added to address important economic considerations, health system impacts, or social and ethical considerations, or to ensure clinical value is realized. The resulting reimbursement conditions, with accompanying reasons and implementation guidance, are stated in Table 1.
Summary of Deliberation
pERC considered all domains of value of the deliberative framework before developing its recommendation: clinical value, unmet clinical need, distinct social and ethical considerations, economic considerations, and impacts on health systems. For further information on the domains of value, refer to Expert Committee Deliberation at Canada’s Drug Agency.
The committee considered the following key discussion points, organized by the 5 domains of value.
Clinical Value
Appropriate comparators: The committee considered neoadjuvant gemcitabine-cisplatin, perioperative dose-dense methotrexate, vinblastine, doxorubicin, and cisplatin (ddMVAC), and adjuvant nivolumab to be appropriate comparators for perioperative durvalumab in patients with resectable MIBC.
Efficacy versus gemcitabine-cisplatin: One phase III, open-label, multicentre randomized controlled trial (NIAGARA; N = 1,083) evaluated perioperative durvalumab plus neoadjuvant gemcitabine-cisplatin, followed by adjuvant durvalumab versus neoadjuvant gemcitabine-cisplatin alone in adults with clinical stage T2N0-1M0 to T4aN0-1M0 MIBC. At the second interim analysis, perioperative durvalumab improved the probability of achieving pCR, EFS at 24 months, and OS at 36 months compared with neoadjuvant gemcitabine-cisplatin alone. Median OS was not reached in either group. These results were statistically significant and considered clinically meaningful by the clinical experts. However, the relative contribution of the neoadjuvant versus adjuvant components remains uncertain, and the efficacy of durvalumab in patients managed with bladder-sparing approaches is unknown. pERC also acknowledged that in the trial, 6 patients (1.1%) in the durvalumab treatment group had a complete clinical response and chose not to undergo cystectomy. These patients were able to enter a noncystectomy extension phase in which they received adjuvant durvalumab monotherapy.
Clinical importance of treatment effects: The committee considered the improvements in pCR, EFS, and OS to be clinically meaningful based on patient and clinical expert input. Patients prioritized disease control, prolonged survival, and tolerability, while experts viewed at least 5% to 10% improvements in OS and 8% to 10% in pCR and EFS as clinically important. Perioperative durvalumab likely meets these thresholds.
Certainty of the evidence: The certainty of evidence for pCR, EFS at 24 months, and OS at 36 months was moderate, with each outcome downgraded 1 level for imprecision. In each case, the point estimate met the clinically important threshold suggested by the experts, but the lower bound of the CI crossed this threshold. For EFS, the inclusion of patients who did not undergo surgery could have introduced bias into this open-label study; however, the results from a post-hoc analysis that censored these patients were consistent, which reduces concern. OS data were immature at the time of the analysis but had already reached statistical significance at an interim analysis. Overall, the evidence suggests a meaningful clinical benefit with perioperative durvalumab, though some uncertainty remains regarding the magnitude of effect.
ITC and gaps in the evidence: The sponsor-submitted indirect treatment comparison (ITC) feasibility assessment found that valid ITCs between perioperative durvalumab and ddMVAC or adjuvant nivolumab could not be conducted due to major methodological and clinical differences across the trials, including immortal time bias, baseline heterogeneity, and unaligned outcomes reporting. Nonetheless, comparisons to ddMVAC and adjuvant nivolumab remain a notable gap in the evidence.
Harms: Durvalumab was generally considered tolerable in the NIAGARA study. The addition of durvalumab did not delay surgery or reduce completion of neoadjuvant chemotherapy cycles. Serious adverse events were comparable between groups, though slightly more frequent in the durvalumab group during the adjuvant phase, given that patients in the gemcitabine-cisplatin group did not receive postsurgery treatment. pERC noted that the product monograph states that serious embolic adverse events were reported at a frequency of 3% or higher in patients in the durvalumab treatment group (4.7%) compared to the gemcitabine-cisplatin group (1%). Overall, the committee indicated that no new safety signals were observed.
Downstream immunotherapy: The participating drug plans sought guidance about eligibility for downstream immunotherapy. pERC noted that in the absence of evidence about downstream immunotherapy in the submission, a disease-free interval of 6 months before immunotherapy-based re-treatment was felt to be reasonable. pERC also noted that guidance for downstream therapies should be considered in an updated algorithm for MIBC.
Discontinuation of adjuvant therapy: The clinical experts consulted by CDA-AMC indicated that it may take longer than 1 cycle of adjuvant therapy to observe its impact on disease; therefore, a decision to discontinue durvalumab might not be made immediately if progression is observed after 1 cycle. pERC acknowledged the experts’ input but noted that there is no evidence to support continuation of therapy upon the occurrence of disease progression.
Clinical value: Based on the totality of evidence, the committee determined that neoadjuvant durvalumab plus gemcitabine-cisplatin, followed by durvalumab after surgery provides added clinical value compared with neoadjuvant gemcitabine-cisplatin in resectable MIBC.
Unmet Clinical Need
Input on unmet clinical need: Patients and clinicians identified a continued need for curative treatments that improve response rates and reduce relapses. Relapse after standard therapy remains common, and current treatments often result in substantial physical and emotional burden due to loss of bladder function and treatment-related toxicity.
Severity of the disease: The committee considered MIBC to be a serious and life-threatening condition that is associated with a high risk of progression and mortality.
Availability of treatment options: The clinical experts noted that patients with MIBC who are eligible should receive neoadjuvant cisplatin-based chemotherapy (gemcitabine-cisplatin or ddMVAC) before radical cystectomy. Patients who are not eligible for cisplatin should proceed directly to radical cystectomy or trimodal therapy for bladder preservation. Adjuvant nivolumab is recommended for patients with residual or high-risk disease after surgery.
Distinct Social and Ethical Considerations
Input on unmet nonclinical need: The committee noted an unmet nonclinical need for standardized access to neoadjuvant chemotherapy and bladder-sparing approaches across Canada. In their input for this submission, patients and clinicians highlighted the need for bladder-sparing approaches.
Equity considerations: Current care varies by region and centre, resulting in inconsistent treatment opportunities. Equity concerns were identified for patients in marginalized, remote, and/or under-resourced settings. Specifically, the committee acknowledged that patients in these settings are less likely to receive trimodal therapy or multidisciplinary care, limiting their treatment options. This creates inequities in access to guideline-recommended therapies.
Ethical implications: Radical cystectomy is associated with high morbidity and can cause permanent urinary changes, sexual dysfunction, and have a psychological impact on patients. pERC acknowledged the clinical experts’ input that bladder-sparing treatment options are an alternative to cystectomy that can reduce this burden. Although it was not studied in the NIAGARA trial, pERC referred to input from the clinical experts that indicated that patients who proceed with an alternative to radical cystectomy may also be eligible for adjuvant durvalumab in consultation with the treating clinician.
Economic Considerations
Health impacts versus relevant comparators: Neoadjuvant durvalumab in combination with gemcitabine-cisplatin, followed by adjuvant durvalumab monotherapy is predicted to be associated with a gain of 1.83 life-years compared to neoadjuvant gemcitabine-cisplatin and may result in a gain of 1.51 quality-adjusted life-years compared to gemcitabine-cisplatin alone.
Cost versus relevant comparators: Neoadjuvant durvalumab in combination with gemcitabine-cisplatin, followed by adjuvant durvalumab monotherapy is predicted to be associated with higher costs to the health care system than neoadjuvant gemcitabine-cisplatin (incremental costs = $98,565), primarily driven by the increased costs associated with drug acquisition.
Key findings of the economic evaluation: Based on the submitted evidence, and using the sponsor’s cost-utility analysis, the CDA-AMC base-case analysis estimated that the incremental cost-effectiveness ratio for neoadjuvant durvalumab in combination with gemcitabine-cisplatin, followed by adjuvant durvalumab monotherapy in MIBC was $65,194 per quality-adjusted life-year gained when compared with neoadjuvant gemcitabine-cisplatin (Figure 1).
Figure 1: Estimate of the ICER Used by pERC to Inform the Price Condition
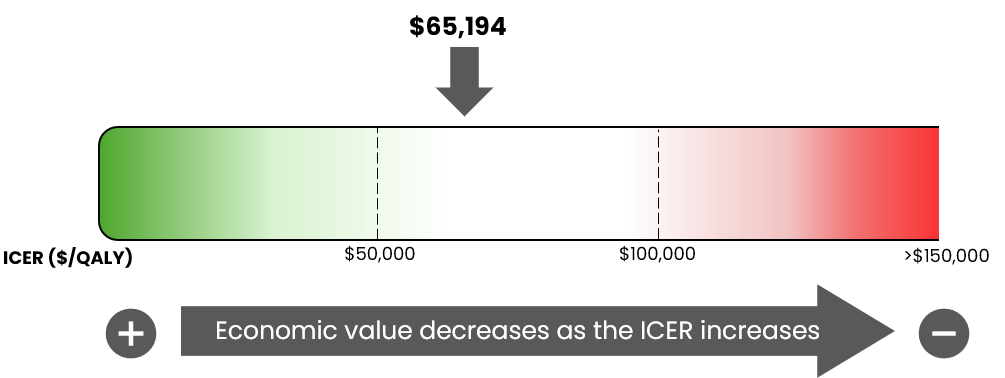
ICER = incremental cost-effectiveness ratio; pERC = pan-Canadian Oncology Drug Review Expert Review Committee; QALY = quality-adjusted life-year.
Certainty of the evidence: The sponsor’s cost-utility analysis included neoadjuvant gemcitabine-cisplatin as the sole comparator. As a result, the relative cost-effectiveness of neoadjuvant durvalumab in combination with gemcitabine-cisplatin followed by adjuvant durvalumab monotherapy versus other relevant regimens, such as ddMVAC or adjuvant nivolumab, remains unknown. No indirect or head-to-head comparative evidence was provided to inform clinical differences relative to these treatment options, despite their established use in practice in Canada. Given the absence of comparative clinical evidence, pERC concluded that there is insufficient justification for a price premium for durvalumab relative to ddMVAC or adjuvant nivolumab. Accordingly, any pricing condition should assume cost parity with these relevant comparators until robust comparative evidence becomes available.
Impacts on Health Systems
Anticipated budget impact: CDA-AMC estimated 2,384 patients would be eligible for treatment with neoadjuvant durvalumab in combination with gemcitabine-cisplatin, followed by adjuvant durvalumab monotherapy over a 3-year period (year 1 = 788; year 2 = 793; year 3 = 802), of whom 1,269 are expected to receive durvalumab (year 1 = 331; year 2 = 420; year 3 = 518). The estimated incremental budget impact of reimbursing neoadjuvant durvalumab in combination with gemcitabine-cisplatin, followed by adjuvant durvalumab monotherapy is predicted to be approximately $111 million over the first 3 years, with an expected expenditure of $170 million on durvalumab.
Sources of Information Used by the Committee
To make its recommendation, the committee considered the following information (links to the full documents for the review can be found on the project webpage):
the CDA-AMC review of the clinical and pharmacoeconomic evidence submitted by the sponsor, as well as relevant ethical issues for durvalumab (refer the Main Report and Supplemental Material document)
the sponsor’s comments on the draft report and the CDA-AMC responses
patients' perspectives gathered by 1 patient group, Bladder Cancer Canada (refer to the Patient and Clinician Group Input document)
input from 2 clinician groups, Bladder Cancer Canada and the Ontario Health (Cancer Care Ontario) Health Cancer Care Ontario Genitourinary Cancer Drug Advisory Committee (refer to the Patient and Clinician Group Input document)
input from public drug programs that participate in the reimbursement review process (refer to the Supplemental Material document)
input from 2 clinical experts with expertise in the management of MIBC consulted by CDA-AMC.
pERC Information
Members of the committee
Dr. Catherine Moltzan (Chair), Dr. Kelvin Chan (Vice-Chair), Paul Agbulu, Dr. Phillip Blanchette, Dr. Matthew Cheung, Dr. Michael Crump, Annette Cyr, Dr. Jennifer Fishman, Dr. Jason Hart, Terry Hawrysh, Dr. Yoo-Joung Ko, Dr. Aly-Khan Lalani, Amy Peasgood, Dr. Anca Prica, Dr. Michael Raphael, Dr. Adam Raymakers, Dr. Patricia Tang, Dr. Pierre Villeneuve, and Danica Wasney
Meeting date: November 12, 2025
Regrets: Three expert committee members did not attend.
Conflicts of interest: None
ISSN: 2563-6596
Canada’s Drug Agency (CDA-AMC) is a pan-Canadian health organization. Created and funded by Canada’s federal, provincial, and territorial governments, we’re responsible for driving better coordination, alignment, and public value within Canada’s drug and health technology landscape. We provide Canada’s health system leaders with independent evidence and advice so they can make informed drug, health technology, and health system decisions, and we collaborate with national and international partners to enhance our collective impact.
Disclaimer: CDA-AMC has taken care to ensure that the information in this document was accurate, complete, and up to date when it was published, but does not make any guarantee to that effect. Your use of this information is subject to this disclaimer and the Terms of Use at cda-amc.ca.
The information in this document is made available for informational and educational purposes only and should not be used as a substitute for professional medical advice, the application of clinical judgment in respect of the care of a particular patient, or other professional judgments in any decision-making process. You assume full responsibility for the use of the information and rely on it at your own risk.
CDA-AMC does not endorse any information, drugs, therapies, treatments, products, processes, or services. The views and opinions of third parties published in this document do not necessarily reflect those of CDA-AMC. The copyright and other intellectual property rights in this document are owned by the Canadian Agency for Drugs and Technologies in Health (operating as CDA-AMC) and its licensors.
Questions or requests for information about this report can be directed to Requests@CDA-AMC.ca