CADTH Reimbursement Review
Pembrolizumab in Combination With Lenvatinib (Keytruda and Lenvima)
Sponsor: Merck Canada Inc.
Therapeutic area: Advanced endometrial cancer
This multi-part report includes:
Clinical Review
Pharmacoeconomic Review
Stakeholder Input
Clinical Review
Abbreviations
AE
adverse event
CCRAN
Colorectal Cancer Resource & Action Network
CCS
Canadian Cancer Society
CCSN
Canadian Cancer Survivor Network
CI
confidence interval
cLDA
constrained longitudinal data analysis
CNS
central nervous system
CR
complete response
dMMR
mismatch repair deficient
DOR
duration of response
ECOG PS
Eastern Cooperative Oncology Group Performance Status
EORTC QLQ-C30
European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30
EORTC QLQ-EN24
European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Endometrial Cancer Module
EQ-5D-5L
5-Level EQ-5D
FAS
full analysis set
FIGO
International Federation of Gynecology and Obstetrics
GHS
Global Health Status
HRQoL
health-related quality of life
HR
hazard ratio
IA1
first interim analysis
IA2
second interim analysis
IHC
immunohistochemistry
IIR
independent imaging review
ITC
indirect treatment comparison
ITT
intention to treat
LEN
lenvatinib
MID
minimal important difference
MMR
mismatch repair
MSI
microsatellite instability
MSI-H
microsatellite instability-high
MSI-L
microsatellite instability-low
MSS
microsatellite stable
NE
not estimable
ORR
objective response rate
OS
overall survival
PD-1
programmed cell death receptor 1
PD-L1
programmed death ligand 1
PEM
pembrolizumab
PFS
progression-free survival
pMMR
mismatch repair proficient
PRO
patient-reported outcome
QoL
quality of life
SAE
serious adverse event
SD
standard deviation
TEAE
treatment-emergent adverse event
TPC
treatment of physician’s choice
VAS
Visual Analogue Scale
VEGF
vascular endothelial growth factor
Executive Summary
An overview of the submission details for the drug under review is provided in Table 1.
Item | Description |
|---|---|
Drug product | Pembrolizumab in combination with lenvatinib (Pembrolizumab: powder for solution for infusion 50 mg, or solution for infusion 100 mg/4 mL vial IV infusion over 30 minutes; lenvatinib: 4 mg and 10 mg capsules [as lenvatinib mesylate], oral) |
Indication | Adult patients with advanced endometrial carcinoma that is not MSI-H or dMMR who have disease progression following prior platinum-based systemic therapy and are not candidates for curative surgery or radiation |
Reimbursement request | As per indication |
Health Canada approval status | NOC/c |
Health Canada review pathway | Project Orbis |
NOC date | September 20, 2019 |
Sponsor | Merck Canada Inc. |
dMMR = mismatch repair deficient; MSI-H = microsatellite instability-high; NOC = Notice of Compliance; NOC/c = Notice of Compliance with Conditions.
Introduction
Endometrial cancer is the most common gynecological cancer in Canada.1 It usually occurs in patients over the age of 50 years, with an average age at diagnosis of 60 years.2 Diagnosis of endometrial cancer commonly occurs at an early stage for approximately 80% of patients.1,3,4 The prognosis depends primarily on tumour stage, tumour histology, and grade.
In terms of histology, there are 2 subtypes. Type I endometrial cancers are low-grade (1 or 2) endometrioid tumours and represent 80% of patients.1,3,5 Type II endometrial cancers account for 10% to 20% of patients and include grade 3 endometroid tumours as well as tumours of non-endometroid cancers.1,3 The 5-year survival rates are approximately 80% to 90% and 20% for type I and type II endometrial cancer, respectively. The prognosis for patients with recurrent endometrial cancer is poor, with a median survival of about 12 months.6 Molecular testing of cancer biomarkers during endometrial biopsy assists in identifying treatment options and stratifying risk.1 Two molecular cancer biomarkers commonly assessed are microsatellite instability (MSI) and mismatch repair (MMR) protein expression.7 Based on the variability of DNA microsatellite lengths, endometrial cancer can be classified into microsatellite instability-high (MSI-H), microsatellite instability-low (MSI-L), and microsatellite stable (MSS).7,8 Based on the DNA MMR status, endometrial cancer can be classified into mismatch repair deficient (dMMR) and mismatch repair proficient (pMMR). In clinical practice and in clinical trials, the terms dMMR and MSI-H are often used interchangeably, while non-MSI-H and pMMR are also interchangeable.7-10
Treatment options for endometrial cancer are dependent on stage and pathologic factors after initial surgery and based on estimated risk of disease recurrence.11-13 Individuals diagnosed with advanced or recurrent endometrial cancer may require adjuvant radiotherapy and/or chemotherapy, depending on the extent and location of spread and/or pathologic risk factors. The current standard of care for patients with advanced or recurrent disease is platinum-based chemotherapy as a doublet or single drug.14,15 The most common platinum-based therapies are carboplatin and paclitaxel.11,14-16 However, for patients with advanced or recurrent endometrial cancer who have progressed on or after platinum-based chemotherapy, there is currently no established, standard effective or curative second-line therapy.11,15 In clinical practice, these patients are typically re-treated with platinum-based chemotherapy, but the response is poor. Response rates ranging from 10% to 15% have been observed among all available treatment options.15 The clinical experts consulted by CADTH indicated that there is a great unmet need for effective therapies with acceptable toxicity profiles for patients with dMMR or MSI-H recurrent or advanced endometrial cancer who have progressed on or after treatment with a platinum-containing regimen.
Pembrolizumab (PEM) is an inhibitor of programmed cell death receptor 1 (PD-1). The recommended dose is 200 mg every 3 weeks or 400 mg every 6 weeks administered as an IV infusion for up to 24 months or until disease progression or unacceptable toxicity. Health Canada has issued market authorization for PEM in various indications, such as classical Hodgkin lymphoma, primary mediastinal B-cell lymphoma, urothelial carcinoma, melanoma, non–small cell lung carcinoma, renal cell carcinoma, head and neck squamous cell carcinoma, and colorectal cancer.8 The Health Canada–approved indication of interest for this review is PEM in combination with lenvatinib (LEN) for the treatment of adult patients with advanced endometrial carcinoma that is not MSI-H or dMMR who have disease progression following prior platinum-based systemic therapy and are not candidates for curative surgery or radiation.8 The CADTH reimbursement request aligns with this Health Canada indication (refer to Table 3).
The objective of this clinical review is to review the beneficial and harmful effects of PEM in combination with LEN (PEM + LEN) for the treatment of adult patients with advanced endometrial carcinoma that is not MSI-H or dMMR who have disease progression following prior platinum-based systemic therapy and are not candidates for curative surgery or radiation.
Stakeholder Perspectives
The information in this section is a summary of the input provided by the patient groups who responded to CADTH’s call for patient input and from clinical expert(s) consulted by CADTH for the purpose of this review.
Patient Input
This section was prepared by CADTH staff based on the input provided by patient groups.
The input from patient advocacy groups for PEM + LEN for the treatment of advanced endometrial cancer was provided by Colorectal Cancer Resource & Action Network (CCRAN) in collaboration with the Canadian Cancer Society (CCS) and the Canadian Cancer Survivor Network (CCSN). CCRAN is a Canadian not-for-profit patient advocacy group focusing on patients with colorectal cancer, with an extended mandate to support other cancer populations that lack capacity or representative patient groups.
The information provided by CCS was collected through an online survey conducted from October 22, 2021, to November 3, 2021, with 22 responders from Canada (20 patients and 2 caregivers). CCSN conducted an outreach survey on December 5, 2021, and provided feedback from 1 patient in Canada with endometrial cancer. CCRAN provided additional feedback from 1 caregiver and 3 patients with advanced endometrial cancer through telephone interviews that took place from December 1, 2021, to December 14, 2021 in Canada.
The 3 patient groups reported that individuals with endometrial cancer experience physical symptoms (e.g., vaginal bleeding, pelvic pain, diarrhea, nausea, and fatigue) and psychological symptoms (feeling isolated and lonely). Some of the patients expressed substantial frustration related to their long diagnostic journeys, noting that the lengthy time periods may have contributed to disease progression and an advanced stage at diagnosis. Endometrial cancer negatively affects quality of life (QoL) for patients and their families. Many patients report issues with work, daily chores, and socialization. Caregivers and family members have to take on additional responsibilities and deal with stress and anxiety.
Regarding current treatment, patients reported experience with a variety of options, including surgery, chemotherapy, and hormonal therapy. The CCSN survey and CCRAN interviews captured a general lack of efficacy and debilitating side effects of the standard of care treatments indicated for the management of advanced endometrial cancer.
Three patients in Canada had experience with PEM + LEN combination therapy, whether through a clinical trial or private pay plan. Two of these 3 patients reported significant amelioration of cancer-induced symptoms, with disease regression and superior QoL, after 26 months of therapy in 1 case and after 4 months of therapy in the other. They reported being able to function at an almost normal level and resume their daily activities. Treatment-induced side effects were reported by 2 of the patients and included diarrhea, fatigue, and urinary tract infection. One patient experienced a dose reduction of LEN (to 10 mg/day from 14 mg/day) due to a headache at the beginning of the treatment. Patients appreciated having access to an oral treatment (LEN) as well as the short infusion time for PEM (30 minutes to 45 minutes every 3 weeks).
Key outcomes identified by patient advocacy groups as important to patients with endometrial cancer include improved symptoms, cancer control, fewer side effects, good QoL, and extension of survival.
Overall, the CCRAN patient group indicated that there is an urgent, unmet need for the treatment of patients with advanced endometrial cancer. The group emphasized that patients need access to treatments with fewer side effects that would extend and improve the quality of their lives. CCRAN strongly supports the use of PEM + LEN combination therapy as a second-line treatment option for MSS patients or pMMR tumours patients that are inoperable, metastatic, and/or recurrent.
Clinician Input
Input From the Clinical Experts Consulted by CADTH
The clinical experts consulted for this review indicated that currently, there is a lack of treatment options and no standard second-line therapy for individuals with metastatic or recurrent endometrial carcinoma. Both clinical experts noted that most patients undergoing current therapies show low response rates, short durations of response, and progression. This represents a critical unmet need in this patient population.
The clinical experts indicated that patients with endometrial carcinoma who have progressed on platinum chemotherapy currently receive cytotoxic treatments, such as carboplatin and paclitaxel, doxorubicin, or pegylated liposomal doxorubicin. Additional chemotherapeutic drugs that can be taken occasionally include topotecan, gemcitabine, pemetrexed ifosfamide, and hormonal treatments (megestrol acetate, tamoxifen). The described treatments are not considered curative and have low expected response rates and short durations.
Both clinical experts indicated that PEM + LEN combination therapy would become standard second-line therapy for patients with endometrial cancer after recurrence or failure of typical platinum-based regimens. This treatment combination would address the underlying disease process. The clinical experts were of the view that it would be preferable to initiate treatment with the drug under review before other therapies.
Clinical experts indicated that currently, there is no evidence to support re-treatment with the same drugs in the event of relapse.
Clinical experts agreed that patients with endometrial carcinoma who experience recurrent or progressive disease following platinum-containing chemotherapy and have a good Eastern Cooperative Oncology Group Performance Status (ECOG PS) (i.e., 0 or 1) would benefit most from PEM + LEN combination therapy. The experts also indicated that, while not supported by clinical trial evidence, the treatment might be extended to patients with an ECOG PS of 2 if they were appropriately informed and motivated. The experts noted that there is currently a lack of data on treatment response among patients with other histologic endometrial cancer types of (e.g., carcinosarcoma, endometrial leiomyosarcoma, and endometrial stromal sarcomas). One expert indicated that unstable central nervous system (CNS) metastases should be treated with neurosurgical resection and post-operative cranial irradiation before considering treatment with PEM + LEN combination therapy.
Regarding the identification of patients, 1 clinical expert mentioned that standard practice includes a clinical examination by an oncologist, diagnostic imaging, and biopsies. The other expert noted that biomarker testing for MMR status through immunohistochemistry (IHC) staining is used in many centres in Canada.
The clinical experts reported that treatment with PEM + LEN combination therapy would be least suitable in patients with a poor ECOG PS (3 or 4). In addition, 1 expert noted that patients with a history of multiple lines of chemotherapy or an intolerance of or contraindications to PEM would be least suited to receive the drug under review.
According to the clinical experts, treatment response in clinical practice is evaluated through imaging (e.g., CT, MRI), assessment of clinical symptoms, and physical exam findings. One expert noted that treatment benefit for most biologics would include the absence of progression and good tolerance of the treatment.
Both experts agreed that improved progression-free survival (PFS) and overall survival (OS), maintained or improved QoL, and control of symptoms can be considered clinically meaningful responses to the treatment under review. Treatment response should be assessed every 12 weeks to 16 weeks (3 months to 4 months).
According to the clinical experts, treatment with PEM + LEN combination therapy should be discontinued in the case of disease progression (confirmed clinically or on imaging) or the appearance of serious immune adverse events (AEs) or intolerable treatment toxicities.
Both experts noted that, if toxicity or tolerability issues are related to LEN, it would be reasonable to continue treatment with PEM alone, if the patient is benefiting from it.
Clinical experts consulted by CADTH indicated that treatment administration and monitoring of patients with endometrial cancer should be undertaken by a specialist, namely a gynecologic oncologist or a medical oncologist. Treatment monitoring can potentially be conducted by a general practitioner in oncology with supervision by 1 of the specialists.
The experts recommended administering PEM + LEN in an infusion setting with appropriate monitoring capabilities, such as a hospital or oncology clinic. In terms of companion diagnostics, 1 expert noted that detection of MMR status through IHC staining would be required.
In reference to dosing, clinical experts consulted by CADTH noted that fixed dosing would be applied for PEM and anticipated that dose modifications of LEN would be common in clinical practice. One clinical expert indicated that less frequent administrations (i.e., over 6-week periods) would be better for patients, clinicians, and health centres.
One clinical expert expressed concerns about the high cost of the treatment under review and indicated that the costs might improve with increased availability of other PD-L1 inhibitors on the market.
Clinician Group Input
One joint clinician submission was provided by 7 physicians on behalf of the Ontario Health (Cancer Care Ontario) Gynecology Cancer Drug Advisory Committee. The clinician group noted the absence of currently available therapies for patients with recurrent or progressive endometrial cancer. The group recognized the unmet needs of this patient population, indicating that most patients remain unresponsive to available treatments and highlighting a need for better-tolerated treatment options. The clinician group stated that the LEN + PEM combination could be used second-line as a preferred option for all patients with endometrial cancer who experienced a recurrence or progression after platinum-based chemotherapy. Prolonged life, delayed disease progression, symptomatic relief, partial response, full response, and improved health-related quality of life (HRQoL) were identified as the most important treatment goals. In terms of assessing response to treatment, the clinician group stated that imaging, clinical exam, and symptomatic improvement should be assessed in clinical practice. The clinician group also advised that LEN in combination with PEM should be discontinued if a patient experiences disease progression or intolerable side effects related to the treatment. Lastly, outpatient hospital settings were noted as appropriate treatment settings for these patients.
Of note, 5 out of 7 physicians provided CADTH with a conflict of interest declaration in the clinician group input.
Drug Program Input
The drug programs provide input on each drug being reviewed through CADTH’s reimbursement review processes by identifying issues that may have an impact on their ability to implement a recommendation. The implementation questions and corresponding responses from the clinical experts consulted by CADTH are summarized in Table 4.
Clinical Evidence
Pivotal Studies and Protocol Selected Studies
Description of Studies
KEYNOTE-775 is an ongoing, phase III, multi-centre, randomized, open-label, active-controlled superiority study comparing PEM + LEN to treatment of physician’s choice (TPC) for the treatment of adult patients 18 years of age or older with advanced endometrial carcinoma who have disease progression following prior platinum-based systemic therapy and are not candidates for curative surgery or radiation. The KEYNOTE-775 trial included a total of 827 patients: 697 with pMMR disease and 130 with dMMR disease. This review focuses on patients with pMMR disease only. A total of 697 patients with pMMR disease were randomized in a 1-to-1 ratio to receive PEM + LEN (n = 346) or TPC (n = 351). The primary outcomes were PFS and OS. The secondary outcomes included objective response rate (ORR) and HRQoL (measured using the European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30 [EORTC QLQ-C30] Global Health Status [GHS] scale). The exploratory outcomes included duration of response (DOR) and other HRQoL measurements (i.e., the EORTC QLQ-30, the EORTC QLQ Endometrial Cancer Module [EORTC QLQ-EN24] Urological Symptoms Score, and the 5-Level EQ-5D [EQ-5D-5L]).
The findings of this study were based on the first interim analysis (IA1) with a data cut-off date of October 26, 2020. However, it was indicated that the success criteria for the study hypotheses of PFS, OS, and ORR were all met at IA1; consequently, the second interim analysis (IA2) was not required.17,18
Efficacy Results
The key efficacy findings of the KEYNOTE-775 trial are summarized in Table 2. Based on an interim analysis, PEM + LEN combination therapy demonstrated a statistically significant and clinical meaningful improvement in OS compared to TPC (hazard ratio [HR] = 0.68; 95% confidence interval [CI], 0.56 to 0.84; P = 0.0001). Such improvements were also reported in key subgroup analyses of patients with an ECOG PS of 0 (HR = 0.56; 95% CI, 0.42 to 0.75), patients with non-endometrioid cancer (HR = 0.56; 95% CI, 0.42 to 0.74), and patients with 1 prior line of systemic therapy (HR = 0.61; 95% CI, 0.47 to 0.78).
Similarly, PEM + LEN combination therapy showed a statistically significant and clinical meaningful improvement in PFS compared to TPC (HR = 0.60; 95% CI, 0.50 to 0.72; P = 0.0001). Key subgroup analyses of PFS were consistent with the primary analysis (i.e., HR < 1) in patients with an ECOG PS of 0 (HR = 0.57; 95% CI, 0.45 to 0.72) and patients with an ECOG PS of 1 (HR = 0.65; 95% CI, 0.49 to 0.86), patients with endometrioid cancer (HR = 0.59; 95% CI, 0.46 to 0.76), patients with non-endometrioid cancer (HR = 0.56; 95% CI, 0.43 to 0.73), and patients with 1 prior line of systemic therapy (HR = 0.52; 95% CI, 0.42 to 0.65).
Overall, no obvious between-group differences in change from baseline were observed in the patient-reported or HRQoL outcomes.
In addition, ORR was statistically significantly higher in patients receiving PEM + LEN combination therapy than in those receiving TPC. The between-group difference (PEM + LEN versus TPC) was 15.2% (95% CI, 9.1% to 21.4%; P < 0.0001). The results for ORR are in line with the survival benefit seen for OS and PFS.
Harms Results
The key harms findings of the KEYNOTE-775 trial are shown in Table 2 in this section and in tables 49 to 54 in Appendix 3. The proportion of patients with at least 1 treatment-emergent adverse event (TEAE) appeared similar in the PEM + LEN and TPC groups (99.7% in each). The frequency of serious adverse events (SAEs) was higher in the PEM + LEN arm than in the TPC arm. However, when adjusted for exposure, the incidence of SAEs appeared to be similar between the 2 treatment groups. That is, the number of SAEs per 100 person-months was 9.83 versus 9.40 in the PEM + LEN and TPC groups, respectively (Table 49). More patients discontinued the study medication due to AEs with PEM + LEN than with TPC (PEM + LEN versus TPC: 31.0% versus 8.3%). The notable AEs (i.e., the AEs of special interest for this review) were higher in the PEM + LEN group versus the TPC group. The higher incidence of notable harms in the PEM + LEN group was driven primarily by hypothyroidism, hyperthyroidism, and hypertension. Overall, the clinical experts consulted by CADTH for this review agreed that the safety profile of PEM + LEN observed in this study appeared consistent with the known safety profile of each individual drug (PEM or LEN). No additional safety signals were identified. Additionally, the clinical experts indicated that the AEs observed in the study were generally manageable through dose interruption or discontinuation of PEM, LEN, or both, or with LEN dose reduction (with or without concomitant steroid therapy).
Table 2: Summary of Key Results From Pivotal and Protocol Selected Studies
Outcomes | KEYNOTE-775 (pMMR) | |
|---|---|---|
PEM + LEN (N = 346) | TPC (N = 351) | |
Efficacy (ITT population) | N = 346 | N = 351 |
OS (ITT) | ||
Events, n (%) | 165 (47.7) | 203 (57.8) |
Median OS (95% CI), moa | 17.4 (14.2 to 19.9) | 12.0 (10.8 to 13.3) |
HRb (95% CI) | 0.68 (0.56 to 0.84) | |
P valuec | 0.0001c | |
12-month OS rates (95% CI), %a | 61. 6 (56.1 to 66.6) | 49.5 (43.8 to 55.0) |
PFS (ITT) | ||
Events, n (%) | 247 (71.4) | 238 (67.8) |
Median PFS (95% CI), mo.a | 6.6 (5.6 to 7.4) | 3.8 (3.6 to 5.0) |
HRb (95% CI) | 0.60 (0.50 to 0.72) | |
P valuec | < 0.0001c | |
6-month PFS rates (95% CI), %a | 52.1 (46.5 to 57.3) | 36.2 (30.5 to 41.9) |
EORTC QLQ-C30 GHS (FAS) | ||
Baseline | ||
N (% of ITT) | 316 (91.3) | 298 (84.9) |
Mean (SD) | 66.56 (21.44) | 66.64 (22.43) |
At week 12 | ||
N (% of ITT) | 256 (74.0) | 192 (54.7) |
Mean (SD) | 60.94 (21.35) | 62.80 (21.67) |
Change from baseline to week 12 | ||
N (% of ITT) | 327 (94.5) | 310 (98.4) |
LSM (95% CI) | –6.80 (–9.43 to –4.17) | –7.96 (–10.86 to –5.05) |
LSM difference (PEM + LEN vs TPC) (95% CI)d | 1.16 (–2.49 to 4.81) | |
P valued | 0. 5316 | |
ORR (ITT) | ||
N | 346 | 351 |
Events, n (%) | 105 (30.3) | 53 (15.1) |
Difference (PEM + LEN vs. TPC)e in % (95% CI) vs. TPC | 15.2 (9.1 to 21.4) | |
P valuef | < 0.0001 | |
DOR (ITT) | ||
Median (range), monthsg | 9.2 (1.6+ to 23.7+) | 5.7 (0.0+ to 24.2+) |
Harms (safety analysis population) | N = 406 | N = 388 |
Patients with at least 1 TEAE,a n (%) | 341 (99.7) | 324 (99.7) |
Patients with at least 1 SAE, n (%) | 170 (49.7) | 94 (28.9) |
Patients with dose modificationb due to | 316 (92.4) | 137 (42.2) |
Patients with dose interruptionc due to | 235 (68.7) | 91 (28.0) |
Interruption of PEM | 165 (48.2) | 0 (0.0) |
Interruption of LEN | 199 (58.2) | 0 (0.0) |
Interruption of both PEM and LEN | 100 (29.2) | 0 (0.0) |
Patients with dose reductiond due to an adverse event | 229 (67.0) | 42 (12.9) |
Patients with a AEs leading to DC from | 106 (31.0) | 27 (8.3) |
Discontinued PEM | 60 (17.5) | 0 (0.0) |
Discontinued LEN | 97 (28.4) | 0 (0.0) |
Discontinued both PEM and LEN | 43 (12.6) | 0 (0.0) |
Deaths due to adverse eventsb | 16 (4.7) | 15 (4.6) |
Notable harms, n (%) | ||
Adrenal insufficiency | 3 (0.9) | 0 (0.0) |
Colitis | 18 (5.3) | 1 (0.3) |
Hepatitis | 6 (1.8) | 0 (0.0) |
Hyperthyroidism | 37 (10.8) | 4 (1.2) |
Hypophysitis | 2 (0.6) | 0 (0.0) |
Hypothyroidism | 189 (55.3) | 3 (0.9) |
Nephritis | 2 (0.6) | 0 (0.0) |
Pneumonitis | 4 (1.2) | 1 (0.3) |
Type 1 diabetes mellitus | 3 (0.9) | 0 (0.0) |
Hypertension | 224 (65.5) | 17 (5.2) |
AE = adverse event; CI = confidence interval; DC = discontinuation; DOR = duration of response; ECOG PS = Eastern Cooperative Oncology Group Performance Status; EORTC QLQ-C30 GHS = European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30 Global Health Status; FAS = full analysis set; HR = hazard ratio; ITT = intention to treat; LEN = lenvatinib; LSM = least squares mean; MMR = mismatch repair; ORR = objective response rate; OS = overall survival; PEM = pembrolizumab; PEM + LEN = pembrolizumab plus lenvatinib combination therapy; PFS = progression-free survival; pMMR = mismatch repair proficient; PRO = patient-reported outcome; SAE = serious adverse event; SD = standard deviation; TEAE = treatment-emergent adverse event; TPC = treatment of physician’s choice; vs. = versus.
aFrom the product-limit (Kaplan-Meier) method for censored data.
bBased on Cox regression model with the Efron method of tie handling with treatment as a covariate stratified by ECOG PS, geographic region, and prior history of pelvic radiation.
cOne-sided P value based on log-rank test stratified by ECOG PS, geographic region, and prior history of pelvic radiation. The P value has been adjusted for multiple testing.
dBased on a constrained longitudinal data analysis model with PRO scores as the response variable with covariates for treatment by study visit interaction, stratification factors, MMR status, ECOG PS, geographic region, and prior history of pelvic radiation. Note: For baseline and week 12, N is the number of patients in each treatment group with non-missing assessments at the specific time point; for change from baseline, N is the number of patients in the analysis population in each treatment group with at least 1 measurement. The P value has not been adjusted for multiple testing.
eBased on the Miettinen and Nurminen method stratified by ECOG PS, geographic region, and prior history of pelvic radiation.
fOne-sided P value for testing. H0 means the difference in percentage equals 0, whereas H1 means the difference in percentage is greater than 0. The P value has been adjusted for multiple testing.
gFor DOR, the + symbol indicates there is no progressive disease at the time of last disease assessment.
Source: Clinical Study Report.19
Critical Appraisal
The included pivotal study (KEYNOTE-775) was relatively well-designed. This section discusses its methodological limitations.
The KEYNOTE-775 trial was an open-label trial. Study investigators and patients were aware of their treatment status, which increases the risk of detection and performance biases that have the potential to influence subjective outcome reporting (i.e., safety and HRQoL). The direction of anticipated bias related to these outcomes is unclear.
In the pMMR population, it was noted that 47 patients (13.7%) in the PEM + LEN group and 37 patients (11.4%) in the TPC group received antineoplastic drugs as concomitant medications (Table 19). The impact of those concomitant anticancer drugs on the comparative efficacy assessment between the 2 treatment groups remains unknown. Nevertheless, due to the very small number of patients using these individual drugs (e.g., carboplatin, cisplatin, doxorubicin, paclitaxel, LEN, and PEM), the clinical experts consulted for this review considered the unknown potential impact on the comparative efficacy assessment (PEM + LEN versus TPC) to be negligible.
The patient-reported and HRQoL outcome — the EORTC QLQ-C30 GHS — was assessed as a secondary outcome. However, it was not controlled for type I error. The other patient-reported and HRQoL outcomes (the EORTC QLQ-C30 Physical Functioning scale, the EORTC QLQ-EN24 Urological Symptoms Score, and EQ-5D Visual Analogue Score [VAS] score) were assessed as exploratory outcomes. There is a potential risk of bias because of the large number of patients who did not have complete measures, substantial missing data on all of these outcomes, and the open-label nature of the study design. As well, there may have been differential recall bias. Overall, the magnitude and direction of the impact of these missing data and recall bias on the patient-reported and HRQoL outcomes are unknown. The HRQoL findings should be viewed as supportive evidence only.
The primary analyses of PFS, OS, and ORR were based on intention-to-treat (ITT) analysis. In the pMMR population, important protocol deviations were reported for 17 patients: 9 (2.6%) in the LEN + PEM group and 8 (2.3%) in the TPC group. No per-protocol analysis was conducted to assess whether the per-protocol analysis was consistent with the ITT analysis. However, given that the proportion of patients with important deviations was relatively low and balanced across both groups, the impact on comparative efficacy findings (PEM + LEN versus TPC) is expected to be negligible.
Furthermore, the median follow-up duration for pMMR was 12.2 months, which is relatively short; this may mean that survival data (e.g., OS) are evolving. Although the protocol-specified criteria were met for the efficacy analyses, monitoring for safety and efficacy is ongoing. The sponsor provided a final descriptive analysis (cut-off date: March 1, 2022 ||||||||||||||||||||||||||||||||||||||||||||, the results of the final analysis appeared consistent with that observed in IA1 (cut-off date: October 26, 2020). (Refer to Appendix 5.)
This study was a multinational, multi-centre trial with 67 sites in 21 countries. A total of 58 patients in Canada participated in the trial at 11 sites in Canada. According to the clinical experts consulted for this review, the KEYNOTE-775 study population (i.e., adult patients with advanced pMMR endometrial carcinoma who have disease progression following prior platinum-based systemic therapy in any setting and are not candidates for curative surgery or radiation) is considered reflective of the requested target population. There is no concern about generalizing the findings from the pivotal study to Canadian clinical settings.
Indirect Comparisons
No indirect comparison evidence was identified.
Other Relevant Evidence
One additional relevant study (KEYNOTE-146) included in the sponsor’s submission to CADTH was considered to provide additional longer-term evidence for this review.
Description of Study
KEYNOTE-146 is an ongoing, multinational, open-label, single-arm phase Ib and II study of PEM + LEN in patients with selected solid tumours, including endometrial carcinoma. This review reports only on the cohort of patients with advanced non-MSI-H or pMMR endometrial cancer.
Included patients were adults (≥ 18 years old) with histologically and/or cytologically confirmed advanced pMMR endometrial carcinoma, with up to 2 prior lines of systemic therapy, an ECOG PS of 0 or 1, and life expectancy of ≥ 12 weeks.
Patients (N = 94) received PEM + LEN combination therapy, with PEM 200 mg IV once every 3 weeks (to a maximum of 35 PEM treatments) and LEN 20 mg once daily orally. The primary efficacy outcome was ORR at week 24. Key secondary outcomes were ORR at the data cut-off date, DOR, PFS, and OS.
At the data cut-off date (January 10, 2019), the median duration of treatment with PEM + LEN was 7.38 months (range = 0.03 months to 37.78 months). Overall, the median follow-up time for OS was 18.7 months (95% CI, 14.1 months to 20.9 months).
Efficacy Results
Overall Survival
The median OS was 16.4 months (95% CI, 13.5 to 25.9). The survival probabilities of patients at 12 months, 18 months, and 24 months were 69.5% (95% CI, 58.6 to 78.1%), 43.8% (95% CI, 31.2, 55.7%), and 39.2% (95% CI, 26.7 to 51.5%), respectively.
Progression-Free Survival
The median PFS was 5.4 months (95% CI, 4.4 to 7.6). PFS rates at 6 months, 12 months, and 18 months were 49.4%, 33.2%, and 33.2.0%, respectively.
Objective Response Rate
At the data cut-off date (January 10, 2019), among patients who had been enrolled for at least 26 weeks before the cut-off, 36 patients out of 94 patients achieved an objective response, resulting in an ORR of 38.3% (95% CI, 28.5 to 48.9).
Duration of Response
Based on the product-limit (Kaplan-Meier) method for censored data, the median DOR was not reached (95% CI, 6.3 to not reached; range = 1.2 months to 33.1 months).
Harms Results
At the data cut-off date, all patients had experienced at least 1 TEAE (N = 94, 100%). The most common TEAEs (occurring in ≥ 50% patients) were hypertension (63.8%), diarrhea (62.8%), fatigue (54.3%), and decreased appetite (51.1%). The percentage of patients reporting an SAE was 52.1%. The most frequent SAEs (> 5%) were hypertension and abdominal pain, reported by 7.4% and 5.3% of patients, respectively. The proportion of patients with an AE leading to discontinuation from the treatment was 25.5%. The most common events leading to discontinuation from the treatment were muscular weakness and pancreatitis, both reported in 2.1% patients. Three patients (3.2%) died due to an AE. Overall, the safety profile of PEM + LEN was generally consistent with the known safety profiles of each drug when used as monotherapy, with no new safety signals identified for the combination.
Critical Appraisal
The main limitation of the KEYNOTE-146 study was its single-arm design, with no comparator arm. Such a design, in addition to a lack of consideration of confounding variables, precludes causal inferences (i.e., the outcomes cannot be directly attributed to PEM). Without an active comparator or any statistical hypothesis testing, it is not possible to assess the relative therapeutic benefit or safety of PEM against other available treatments (such as chemotherapy) in this population. As indicated in the Health Canada reviewer’s report,20 due to the nature of the limitation of the single-arm design, the findings observed in the KEYNOTE-146 trial needed to be confirmed in a phase III study (i.e., KEYNOTE-775).
Overall, no apparent generalizability issue was identified.
Conclusions
One sponsor-submitted, phase III, multinational, open-label, randomized, active-controlled trial (KEYNOTE-775) was included in this review. Compared with TPC (doxorubicin or paclitaxel), PEM + LEN combination therapy showed a statistically significant and clinically meaningful benefit in terms of OS, PFS, and ORR in the treatment of adult patients with advanced pMMR (i.e., non-MSI-H or dMMR) endometrial carcinoma who have disease progression following prior platinum-based systemic therapy and are not candidates for curative surgery or radiation. The clinical experts consulted for this review indicated that the safety profile of PEM + LEN observed in this study appeared consistent with the known safety profile of each individual drug (PEM or LEN); no additional safety signals were identified. The AEs observed in the study were generally manageable through dose interruption, dose discontinuation, or LEN dose reduction, with or without concomitant steroid therapy.
Introduction
Disease Background
Endometrial cancer arises from the cells of the uterine lining. It is the most common gynecological cancer in Canada.1 More than 95% of all uterine cancers are endometrial.3 Uterine cancer is ranked as the 17th leading cause of cancer death in Canada.3,21 CCS estimated that 8,000 women would be diagnosed with uterine cancer in 2021 and that 1,400 women would die of the disease.21,22 Endometrial cancer most often occurs in patients over the age of 50 years, with an average age at diagnosis at 60 years.2 Diagnosis occurs at an early stage for approximately 80% of patients because uterine bleeding is an early presenting symptom.1,3,4 The most common method of diagnosis is endometrial biopsy, followed by endometrial curettage and hysterectomy specimen.1 The International Federation of Gynecology and Obstetrics (FIGO) criteria are commonly used to determine the stage of endometrial cancer, which is based on the size of the tumour and the extent to which the tumour has spread to lymph nodes or distant sites (i.e., metastasis).23 Generally, the higher the stage number, the more the cancer has spread.24 Tumour stage is fixed regardless of tumour type.1 The prognosis of endometrial cancer depends primarily on the tumour stage, tumour histology, and grade. Overall survival by FIGO stage is 80% to 90% for stage I, 70% to 80% for stage II, and 20% to 60% for stages III and IV.1 In terms of histology, there are 2 subtypes. Type I endometrial cancers are low-grade (1 or 2) endometrioid tumours, and represent 80% of patients.1,3,5 Type II endometrial cancers account for 10% to 20% of cases and include grade 3 endometroid tumours as well as tumours of non-endometroid histology, such as serous clear-cell, mucinous, squamous, transitional cell, mesonephric carcinosarcoma, and undifferentiated.1,3 The 5-year survival rate for type I endometrial cancer is around 80% to 90%, while the 5-year survival rate for type II endometrial cancer is as low as 20%. Other notable factors that may affect the prognosis for endometrial cancer include race, age, uterine tumour location, peritoneal cytology results, and lymph vascular space invasion.1
Molecular testing for cancer biomarkers during endometrial biopsy assists in decision-making about treatment options and risk stratification.1 Two molecular cancer biomarkers are commonly assessed in patients with endometrial cancer. One is MSI, which evaluates DNA hypermutability. The other is MMR protein expression, which evaluates the functional status of the MMR DNA repair pathway.7 Tumours can be classified as MSI-H (exhibiting ≥ 30% to 40% microsatellite marker instability), MSI-L (exhibiting instability at < 30% to 40% of loci), and MSS (exhibiting no unstable markers).25 Based on MMR status, endometrial cancer can be classified into dMMR and pMMR. IHC is used to test for dMMR, in which the cells’ ability to repair mistakes during the division process is impaired. The tumour is immunohistochemically assessed for the loss of at least 1 of the following MMR proteins: MLH1, MSH2, MSH6, and/or PMS2.26-28 MSI statuses are determined by analyzing 5 tumour microsatellite loci using polymerase chain reaction–based assays (either the 5 mononucleotide loci [BAT25, BAT26, NR21, NR24, and Mono27] or the 5 mixed mononucleotide and dinucleotide loci [BAT25, BAT26, Di 5S346, Di 2S123, and Di 17S250]).29 If MMR status cannot be determined from tumour samples, the sample may undergo genetic testing using next-generation sequencing to identify MSI status.
MMR status testing is already widely available and performed routinely in local laboratories. Tumours can be screened by IHC or polymerase chain reaction, and there is a high concordance between the 2 techniques.30-32 Although MSI and MMR are 2 distinct biomarkers, these are both signs of an altered MMR pathway. In clinical trials, the terms dMMR and MSI-H are often used interchangeably (i.e., termed MSI-H or dMMR or MSI-H/dMMR). Non-MSI-H (including MSS and MSI-L) and pMMR are also interchangeable terms.7-10 The clinical experts CADTH consulted for this review agreed that dMMR and MSI-H are commonly used interchangeably in clinical practice. The defect or absence of MMR protein causes DNA hypermutation and high levels of MSI-H in sections of the DNA.28 Compared with MSS endometrial cancers, MSI-H or dMMR endometrial cancer is associated with a higher neoantigen load and increased PD-1–expressing, tumour-infiltrating lymphocytes, and programmed death ligand 1 (PD-L1)–expressing intraepithelial and peritumoral immune cells.33 Thus, MSI-H or dMMR tumour status is predictive of clinical benefit from PD-1 inhibitor monotherapy. For patients with pMMR gynecologic tumours, the combination of a PD-1 inhibitor and another targeted drug may be necessary to achieve the same level of response.34 pMMR represents approximately 69% to 75% of primary endometrial cancers and 70% to 87% of recurrent endometrial cancers.35-39
Recurrence occurs in approximately 13% to 20% of patients with endometrial cancer, with rates varying greatly by FIGO stage at diagnosis; these are highest (> 65%) among patients with stage IV endometrial cancer.6,11,40 The prognosis for patients with recurrent endometrial cancer is poor, with a median survival of about 12 months.6
Standards of Therapy
Treatment options for endometrial cancer depend on stage and pathologic factors after initial surgery and are based on an estimated risk of disease recurrence. Early-stage endometrial cancer and/or type I tumour endometrial cancers can be cured through surgery alone.11-13 Individuals diagnosed with advanced or recurrent endometrial cancer may require adjuvant radiotherapy and/or chemotherapy, depending on the extent and location of spread and/or pathologic risk factors. Cancer Care Ontario and Alberta Health Services recommendations favour combination chemotherapy over single-drug chemotherapy for individuals with advanced or recurrent endometrial cancer because it elicits a higher response rate with less toxicity.11,14 The current standard of care for patients with advanced or recurrent disease is platinum-based chemotherapy as a doublet or single drug,14 a standard echoed by the European Society of Gynecological Oncology, the European Society of Radiotherapy and Oncology, and the European Society of Pathology.15 The most common platinum-based therapy is carboplatin plus paclitaxel.11,14-16 For a subset of patients with low-grade recurrent or metastatic endometrial cancer who are estrogen and/or progesterone receptor–positive, or for patients with poor tolerance to systemic therapy, hormonal therapy — such as megestrol, letrozole, and medroxyprogesterone — may be used.14
For patients with newly diagnosed advanced or recurrent endometrial cancer, response rates for standard first-line treatment range from 40% to 62%.41-44 However, for patients with advanced or recurrent endometrial cancer who have progressed on or after platinum-based chemotherapy, there is currently no established, standard effective or curative second-line therapy.11,15 Patients with recurrent endometrial cancer are typically re-treated with either platinum-based chemotherapy or doxorubicin, but the response is poor. The response rates range from 10% to 15% among all available treatment options.15 The clinical experts consulted by CADTH noted that median survival rates range from 12 months to 15 months after re-treatment. Various single-drug chemotherapies may be administered to patients who are resistant or refractory to platinum-based chemotherapy, with response rates typically below 15% and no known, clear survival benefit.45 Hormonal treatments may also be used for disease control, but are not considered curative.
The clinical experts consulted by CADTH for this review indicated that there is a great unmet need for effective therapies with acceptable toxicity profiles that achieve disease control, reduce disease-related symptoms, improve HRQoL, prevent disease progression, and prolong survival among patients with recurrent or advanced endometrial cancer that has progressed on or following prior treatment with a platinum-containing regimen. There is currently no standard, effective second-line therapy for recurrent or refractory disease, and commonly used therapies are non-curative.
Drug
PEM is a selective, humanized monoclonal antibody that enhances the immune system’s detection of tumours and facilitates tumour regression through the PD-1 pathway. The Health Canada–recommended dose is 200 mg every 3 weeks or 400 mg every 6 weeks administered as an IV infusion for up to 24 months or until disease progression or unacceptable toxicity.
LEN is a targeted antineoplastic drug that belongs to the family of receptor tyrosine kinase inhibitors that selectively inhibit the kinase activities of vascular endothelial growth factor (VEGF) receptors. The Health Canada–recommended dose is 20 mg orally once daily until unacceptable toxicity or disease progression.
The Health Canada–approved indication of interest for this review is PEM + LEN combination therapy for the treatment of adult patients with advanced endometrial carcinoma that is not MSI-H or dMMR who have disease progression following prior platinum-based systemic therapy and are not candidates for curative surgery or radiation.8 The CADTH reimbursement request aligns with this Health Canada indication (refer to Table 3).
Table 3: Key Characteristics of Pembrolizumab, Lenvatinib, Doxorubicin, and Paclitaxel
Characteristic | Pembrolizumab8 | Lenvatinib46 | Doxorubicin47 | Paclitaxel48 |
|---|---|---|---|---|
Mechanism of action | Exerts dual ligand blockade of the PD-1 pathway on antigen or tumour cells and reactivates tumour-specific cytotoxic T lymphocytes in the tumour microenvironment | A targeted antineoplastic drug that belongs to the family of RTK inhibitors that selectively inhibit the kinase activities of VEGF receptors VEGFR1 (FLT1), VEGFR2 (KDR), and VEGFR3 (FLT4) in addition to other proangiogenic and oncogenic pathway-related RTKs, including FGFRs 1, 2, 3, and 4; PDGFRA; KIT; and RET. In addition, lenvatinib has selective, direct, antiproliferative activity in hepatocellular cell lines dependent on activated FGFR signalling, which is attributed to the inhibition of FGFR signalling by lenvatinib. Enhanced antiangiogenic leads to antitumour activity were observed. The antitumour activity of the combination of lenvatinib and an anti-PD-1 monoclonal antibody was greater than that of either as monotherapy.46 | Antineoplastic drug (DNA binding activity and inhibition of nucleic acid synthesis) | Antineoplastic drug (antimicrotubule activity; disrupts dynamic equilibrium within the microtubule system and blocks cells in the late G2 and M phases, inhibiting cell replication and impairing the function of nervous tissue) |
Indicationa | Used in combination with lenvatinib for the treatment of adult patients with advanced endometrial carcinoma that is not MSI-H or dMMR who have disease progression following prior platinum-based systemic therapy and are not candidates for curative surgery or radiation | Used in combination with pembrolizumab for the treatment of adult patients with advanced endometrial carcinoma that is not MSI-H or dMMR who have disease progression following prior platinum-based systemic therapy and are not candidates for curative surgery or radiation | Used alone or in combination for the treatment of neoplastic conditions, such as gynecologic carcinomas and testicular carcinomasb | Used alone or in combination for the treatment of ovarian, breast, or lung cancerc |
Route of administration | IV | Oral | IV and intravesical | IV |
Recommended dose | Used in combination with pembrolizumab and given as 200 mg every 3 weeks or 400 mg every 6 weeks for up to 24 months (i.e., 35 doses of 200 mg or 18 doses of 400 mg) or until unacceptable toxicity or disease progression, whichever is longer, in combination with lenvatinib | Used in combination with pembrolizumab and administered as 20 mg orally once daily until unacceptable toxicity or disease progression | The most commonly used dosage schedule is 60 mg/m2 to administered at 21-day intervals. It is recommended not to exceed a maximum cumulative dose of 550 mg/m2. | The recommended regimen is 175 mg/m2 IV injection over 3 hours every 3 weeks (for ovarian cancer)a |
Serious adverse effects or safety issues | Hepatic impairment Immune-mediated adverse reactions, including immune-mediated pneumonitis, colitis, hepatitis, nephritis and renal dysfunction, endocrinopathies, adrenal insufficiency, hypophysitis, type 1 diabetes mellitus, thyroid disorders, and severe skin reactions Infusion-related reactions Renal impairment Teratogenic risk | Serious reactions and/or life-threatening events include: Hypertension Cardiac failure Arterial thromboembolism Gastrointestinal perforation and fistula formation Hepatotoxicity and/or hepatic failure Renal failure and impairment Hemorrhage, including posterior reversible encephalopathy syndrome | Acute life-threatening arrhythmias Cardiomyopathy Secondary acute AML and MDS Extravasation and tissue necrosis Severe myelosuppression (clinical consequences of which may include fever, infections, septic shock, hemorrhage, tissue hypoxia, or death) | Bone marrow suppression Hypotension Should be administered as diluted infusion Patients should be pre-treated with corticosteroids, antihistamines, and H2 antagonist Should not be administered to patients with baseline neutrophil counts of less than 1,500 cells/mm3 |
AML = acute myelogenous leukemia; dMMR = mismatch repair deficient; FGFR = fibroblast growth factor receptor; M = mitotic; MDS = myelodysplastic syndrome; MSI-H = microsatellite instability-high; PD-1 = programmed cell death receptor 1; PDGFRA = platelet derived growth factor receptor alpha; RTK = receptor tyrosine kinase; VEGF = vascular endothelial growth factor; VEGFR1 = vascular endothelial growth factor receptor 1; VEGFR2 = vascular endothelial growth factor receptor 2; VEGFR3 = vascular endothelial growth factor receptor 3.
aHealth Canada–approved indication.
bDoxorubicin is indicated for gynecologic carcinomas and testicular carcinomas, but it is not described specifically for endometrial carcinomas.
cPaclitaxel is indicated for ovarian, breast, or lung cancer, but it is not described specifically for endometrial carcinomas.
Stakeholder Perspectives
Patient Group Input
This section was prepared by CADTH staff based on the input provided by patient groups.
The input from patient advocacy groups for PEM + LEN for the treatment of advanced endometrial cancer was provided by CCRAN in collaboration with CCSN and CCSN. CCRAN is a Canadian not-for-profit patient advocacy group focusing on patients with colorectal cancer, but its mandate extends to support other cancer populations who lack capacity or representative patient groups.
The information provided by CCS was collected through an online survey conducted from October 22 to November 3, 2021, with 22 responders from Canada (20 patients and 2 caregivers). CCSN conducted an outreach survey on December 5, 2021, and provided feedback from 1 patient in Canada with endometrial cancer. CCRAN provided additional feedback from 1 caregiver and 3 patients with advanced endometrial cancer through telephone interviews that took place from December 1 to December 14, 2021, in Canada.
The 3 patient groups reported that individuals with endometrial cancer experience physical symptoms (e.g., vaginal bleeding, pelvic pain, diarrhea, nausea, and fatigue) and psychological symptoms (feeling isolated and lonely). Some of the patients expressed substantial frustration related to their long diagnostic journeys, noting that these might have contributed to their disease progression and advanced stage at diagnosis. Endometrial cancer negatively affects the QoL of patients and their families. Many patients report issues with work, daily chores, and socialization. Caregivers and family members have to take on additional responsibilities and deal with stress and anxiety.
Regarding current treatment, patients reported a variety of options, including surgery, chemotherapy, and hormonal therapy. The CCSN survey and CCRAN interviews captured a general lack of efficacy and debilitating side effects of the standard of care treatments indicated for the management of advanced endometrial cancer.
Three patients in Canada had experience with PEM + LEN combination therapy through a clinical trial or private pay plan. Two of the 3 patients reported significant amelioration of cancer-induced symptoms, disease regression, and superior QoL after 26 months of therapy in 1 case and 4 months of therapy in the other. They reported being able to function at an almost normal level and resume their daily activities. Treatment-induced side effects were reported by 2 of the patients and included diarrhea, fatigue, and urinary tract infection. One patient experienced a dose reduction of LEN (to 10 mg/day from 14 mg/day) due to a headache at the beginning of the treatment. Patients appreciated having access to an oral treatment (LEN) as well as the short infusion time of PEM (30 minutes to 45 minutes every 3 weeks).
Key outcomes identified by the patient advocacy groups as important to patients with endometrial cancer include improved symptoms, cancer control, fewer side effects, good QoL, and extension of survival.
Overall, the CCRAN patient group indicated that there is an urgent, unmet need for the treatment of patients with advanced endometrial cancer. The group emphasized that patients need access to treatments with fewer side effects that would extend and improve their QoL. CCRAN strongly supports the use of PEM + LEN combination therapy as a second-line treatment option for patients who are MSS or pMMR and whose tumours are inoperable, metastatic, and/or recurrent.
Clinician Input
Input From the Clinical Experts Consulted by CADTH
All CADTH review teams include at least 1 clinical specialist with expertise regarding the diagnosis and management of the condition for which the drug is indicated. Clinical experts are a critical part of the review team and are involved in all phases of the review process (e.g., providing guidance on the development of the review protocol; assisting in the critical appraisal of clinical evidence; interpreting the clinical relevance of the results; and providing guidance on the potential place in therapy). The following input was provided by 2 clinical specialists with expertise in the diagnosis and management of endometrial cancer.
Unmet Needs
Currently, there is a lack of treatment options and no standard second-line therapy for individuals with metastatic or recurrent EC. Both clinical experts noted that most patients undergoing current therapies show low response rates, short DOR, and progression. This represents a critical unmet need in this patient population.
Place in Therapy
Patients with endometrial cancer who have progressed on platinum chemotherapy currently receive cytotoxic treatments, such as carboplatin and paclitaxel, doxorubicin, or pegylated liposomal doxorubicin. Additional chemotherapeutic drugs that can be taken occasionally include topotecan, gemcitabine, pemetrexed ifosfamide, and hormonal treatments (megestrol acetate, tamoxifen). These treatments are not considered curative and have low expected response rates and short durations.
Both clinical experts indicated that PEM + LEN would become standard second-line therapy for patients with endometrial cancer after recurrence or failure of typical platinum-based regimens. This treatment combination would address the underlying disease process. The clinical experts felt it would be preferable to initiate treatment with the drug under review before other therapies.
Clinical experts also indicated that currently, there is no evidence to support re-treatment with the same drugs in the event of relapse.
Patient Population
Clinical experts agreed that patients with endometrial carcinoma who experience recurrent or progressive disease following platinum-containing chemotherapy and have a good ECOG PS (e.g., 0 or 1) would benefit most from PEM + LEN combination therapy. Although not supported by clinical trial evidence, the experts also indicated that the treatment might be extended to patients with an ECOG PS of 2 if the patient is appropriately informed and motivated. The experts noted that there is currently a lack of data on treatment response among patients with other histologic types of endometrial cancer (e.g., carcinosarcoma, endometrial leiomyosarcoma, and endometrial stromal sarcomas). One expert indicated that unstable CNS metastases should be treated with neurosurgical resection and post-operative cranial irradiation before considering treatment with PEM + LEN combination therapy.
Regarding the identification of patients, 1 clinical expert mentioned that standard practice includes a clinical examination by an oncologist, diagnostic imaging, and biopsies. The other expert noted that biomarker testing for MMR status through IHC staining is used in many centres in Canada.
The clinical experts reported that treatment with PEM + LEN combination therapy would be least suitable in patients with poor a ECOG PS (3 or 4). One expert added that patients with multiple lines of chemotherapy, and those with intolerance of or contraindications to PEM, would be least suited to receive the drug under review.
Assessing Response to Treatment
According to the clinical experts, treatment response in clinical practice is evaluated through imaging (e.g., CT, MRI), assessment of clinical symptoms, and physical exam findings. One expert noted that treatment benefit for most biologics would include the absence of progression and good tolerance of the treatment.
Both experts agreed that improved PFS and OS, maintained or improved QoL, and control of symptoms can be considered clinically meaningful responses to the treatment under review. Treatment response should be assessed every 12 weeks to 16 weeks (3 months to 4 months).
Discontinuing Treatment
According to the clinical experts, treatment with PEM + LEN combination therapy should be discontinued in the case of disease progression (confirmed clinically or on imaging), or the appearance of serious immune AEs or intolerable treatment toxicities.
Both experts noted that, if toxicity or tolerability issues are related to LEN, it would be reasonable to continue treatment with PEM alone if the patient is benefiting from it.
Prescribing Conditions
Clinical experts consulted by CADTH indicated that treatment administration and monitoring of patients with endometrial cancer should be undertaken by a specialist, namely a gynecologic oncologist or a medical oncologist. Treatment monitoring can potentially be conducted by a general practitioner in oncology with supervision by 1 of the specialists.
The experts recommended administering PEM + LEN in an infusion setting with appropriate monitoring capabilities, such as a hospital or oncology clinic. In terms of companion diagnostics, 1 expert noted that detection of MMR status through IHC staining would be required.
In reference to dosing, clinical experts consulted by CADTH noted that fixed dosing would be applied for PEM and anticipated that dose modifications of LEN would be common in clinical practice. One clinical expert indicated that less frequent administrations (i.e., over 6-week periods) would be better for patients, clinicians, and health centres.
Additional Considerations
One clinical expert expressed concerns about the high cost of the treatment under review and indicated that the costs might improve with increased availability of other PD-L1 inhibitors on the market.
Clinician Group Input
This section was prepared by CADTH staff based on the input provided by clinician groups.
One joint clinician submission was provided by 7 physicians on behalf of the Ontario Health (Cancer Care Ontario) Gynecology Cancer Drug Advisory Committee. The clinician group noted the absence of currently available therapies for patients with recurrent or progressive endometrial cancer. The group recognized the unmet needs of this patient population, indicating that most patients remain unresponsive to available treatments and highlighting a need for better-tolerated treatment options. The clinician group stated that the LEN + PEM combination could be used second-line as a preferred option for all patients with endometrial cancer who experienced a recurrence or progression after platinum-based chemotherapy. Prolonged life, delayed disease progression, symptomatic relief, partial response, full response, and improved HRQoL were identified as the most important treatment goals. In terms of assessing response to treatment, the clinician group stated that imaging, clinical exam, and symptomatic improvement should be assessed in clinical practice. The clinician group also advised that LEN in combination with PEM should be discontinued if a patient experiences disease progression or intolerable side effects related to the treatment. Lastly, outpatient hospital settings were noted as appropriate treatment settings for these patients.
Of note, 5 out of 7 physicians provided CADTH with a conflict of interest declaration within the clinician group input.
Drug Program Input
The drug programs provide input on each drug being reviewed through CADTH’s reimbursement review processes by identifying issues that may have an impact on their ability to implement a recommendation. The implementation questions and corresponding responses from the clinical experts consulted by CADTH are summarized in Table 4.
Table 4: Summary of Drug Plan Input and Clinical Expert Response
Drug program implementation questions | Clinical expert response |
|---|---|
Considerations for initiation of therapy | |
What is the guidance on the maximum number of prior lines of platinum therapy for patients to be eligible for PEM + LEN combination therapy? | Clinical experts expressed uncertainty regarding the number of previous platinum-based treatments that should be tried before PEM + LEN combination therapy. While patients are unlikely to have had more than 3 previous treatments, such patients should not be disqualified if they have good fitness and expectation of benefit. Clinical experts suggested that the PEM + LEN combination might be preferable to other treatments after platinum therapy because of the toxicity of alternative chemotherapy options (such as doxorubicin). |
What is the guidance on re-treatment? | The clinical experts indicated that re-treatment with the same regimen is a valid question. However, there are no data to show what the response would be. In general, use of the same medications in a relapse setting could be considered, but relapses usually take place in short intervals, and responses are poor. The longer the interval between treatment termination and reintroduction, the more likely it is that a response would be seen. |
Considerations for prescribing of therapy | |
Jurisdictions may implement weight-based dosing up to a maximum dose for pembrolizumab (i.e., 2 mg/kg to a maximum of 200 mg every 3 weeks). Should pembrolizumab 4 mg/kg (to a maximum of 400 mg) IV every 6 weeks be an option? | The clinical experts indicated that the weight-based dosing is seen only in pediatric patients. All adult regimens have had fixed doses, as indicated. The experts mentioned that for very underweight patients, weight-based dosing might be appropriate. A clinical expert noted that less frequent administrations (i.e., 400 mg every 6 weeks) would be better for patients, clinicians, and health centres. |
For patients on PEM + LEN, if 1 of the drugs has to be discontinued due to toxicity, can the other drug be continued? Lenvatinib is not publicly funded for endometrial cancer. | The clinical experts indicated that, in the event of disease progression, both medications should be stopped. If toxicity is related to lenvatinib, stopping lenvatinib and continuing pembrolizumab would be reasonable if the patient is benefiting. If the toxicity is related to pembrolizumab, it is not certain whether there is value in stopping pembrolizumab and continuing lenvatinib. Also, it might be hard to determine which drug is causing which toxicity. Therefore, one might consider discontinuing both drugs. The toxicity profiles of the 2 drugs are different, so 1 clinical expert thinks that most of the time, clinicians have a pretty clear idea which drug is responsible. |
Due to the high frequency of dose modifications of lenvatinib reported in the KEYNOTE-775 trial (in which 66.5% of patients required modifications), are “dose modifications for lenvatinib” in clinical practice anticipated to be common? | The clinical experts indicated that modifying the dosage of lenvatinib is common in Canadian clinical practice settings. The frequency of modification would be the same or higher than the 66.5% reported in the study |
Generalizability | |
Can PEM + LEN combination therapy be extended to patients with an ECOG PS of > 1? | The clinical experts believe that the PEM + LEN combination treatments could be extended to patients with an ECOG PS of 2 if the patients are appropriately informed and motivated. These drugs should likely not be used in patients with an ECOG PS of 3 or 4. |
The KEYNOTE-775 trial excluded patients with carcinosarcoma and sarcoma (i.e., leiomyosarcoma and stromal sarcomas). Can PEM + LEN combination therapy be extended to patients with endometrial carcinomas or sarcomas? | The clinical experts indicated that the evidence to date is in carcinomas; as a result, they are not aware of benefit in pure sarcomas. PEM + LEN combination therapy may be extended to patients with carcinosarcomas, but it should be noted that there is no supporting research evidence for this at the moment. |
The KEYNOTE-775 trial excluded patients with unstable CNS metastases. Can PEM + LEN combination therapy be extended to patients with unstable CNS metastases? | The clinical experts indicated that unstable CNS metastasis should be treated with typical methods. Currently, this means neurosurgical resection and/or cranial irradiation. PEM + LEN combination treatment may be considered subsequently. |
Can pERC clarify the instances wherein time-limited funding would be applicable? | One expert indicated that patients who had started next-line therapy after platinum-based chemotherapy should be given the choice to switch to PEM + LEN on a time-limited basis. However, the preference would be for them to continue with their current regimens and switch to PEM + LEN when progression occurs, particularly if they are responding to their current treatment. |
Care provision issues | |
MSI/MMR testing is needed to confirm eligibility for PEM + LEN. Is there a standardized test to determine non-MSI-H or MMR status to guide the implementation of eligibility criteria? | The clinical experts indicated that MMR testing is based on IHC staining of the tumour as a screening test. MSI status is determined based on PCR testing. These are probably the standard tests across Canada. |
When should testing for MSI or MMR status take place in patients with endometrial cancer? | MSI or MMR status (i.e., non-MSI-H or pMMR cancer) needs to be determined before considering PEM + LEN. |
CNS = central nervous system; ECOG PS = Eastern Cooperative Oncology Group Performance Status; IHC = immunohistochemistry; LEN = lenvatinib; MMR = mismatch repair; MSI = microsatellite instability; MSI-H = microsatellite instability-high; PCR = polymerase chain reaction; PEM = pembrolizumab; PEM + LEN = pembrolizumab plus lenvatinib combination therapy; pERC = CADTH pan-Canadian Oncology Drug Review Expert Review Committee; pMMR = mismatch repair proficient.
Clinical Evidence
The clinical evidence included in the review of PEM in combination with LEN is presented in 3 sections. The first section, the Systematic Review, includes pivotal studies provided in the sponsor’s submission to CADTH and Health Canada, as well as those studies that were selected according to an a priori protocol. The second section includes indirect evidence from the sponsor (if submitted) and indirect evidence selected from the literature that met the selection criteria specified in the review. No indirect treatment comparison (ITC) was submitted by the sponsor or identified in the CADTH literature search. The third section includes sponsor-submitted long-term extension studies and additional relevant studies that were considered to address important gaps in the evidence included in the systematic review (if available). No additional relevant study was identified.
Systematic Review (Pivotal and Protocol Selected Studies)
Objectives
To perform a systematic review of the beneficial and harmful effects of PEM in combination with LEN for the treatment of adult patients with advanced endometrial carcinoma that is not MSI-H or dMMR who have disease progression following prior platinum-based systemic therapy and are not candidates for curative surgery or radiation.
Methods
Studies selected for inclusion in the systematic review included pivotal studies provided in the sponsor’s submission to CADTH and Health Canada as well as those meeting the selection criteria (presented in Table 5). Outcomes included in the CADTH review protocol reflect those considered to be important to patients, clinicians, and drug plans.
Table 5: Inclusion Criteria for the Systematic Review
Criteria | Description |
|---|---|
Population | Adult patients with advanced endometrial carcinoma that is not MSI-H or dMMR who have disease progression following prior platinum-based systemic therapy and are not candidates for curative surgery or radiation Subgroups:
|
Intervention | PEM for IV infusion over 30 minutes in combination with LEN administered orally:
|
Comparators |
|
Outcomes | Efficacy outcomes:
Harms outcomes:
|
Study designs | Published and unpublished phase III and IV RCTs |
AE = adverse event; dMMR = mismatch repair deficient; DOR = duration of response; ECOG PS = Eastern Cooperative Oncology Group Performance Status; HRQoL = health-related quality of life; FIGO = International Federation of Gynecology and Obstetrics; ICU = intensive care unit; LEN = lenvatinib; MSI-H = microsatellite instability-high; ORR = objective response rate; OS = overall survival; PEM = pembrolizumab; PFS = progression-free survival; RCT = randomized controlled trial; SAE = serious adverse event; WDAE = withdrawal due to adverse event.
aPatient-important outcome identified in patient input.
The literature search for clinical studies was performed by an information specialist using a peer-reviewed search strategy according to the PRESS Peer Review of Electronic Search Strategies checklist.49
Published literature was identified by searching the following bibliographic databases: MEDLINE All (1946–) through Ovid and Embase (1974–) through Ovid. The search strategy comprised both controlled vocabulary, such as the National Library of Medicine’s MeSH (Medical Subject Headings), and keywords. The main search concepts were Keytruda/pembrolizumab and endometrial cancer. Clinical trial registries were searched: the US National Institutes of Health’s clinicaltrials.gov, WHO’s International Clinical Trials Registry Platform (ICTRP) search portal, Health Canada’s Clinical Trials Database, and the European Union Clinical Trials Register.
No filters were applied to limit the retrieval by study type. Retrieval was not limited by publication date or by language. Conference abstracts were excluded from the search results. Refer to Appendix 1 for the detailed search strategies.
The initial search was completed on January 21, 2022. Regular alerts updated the search until the meeting of the CADTH pan-Canadian Oncology Drug Review Expert Review Committee on July 13, 2022.
Grey literature (literature that is not commercially published) was identified by searching relevant websites from the CADTH Grey Matters: A Practical Tool For Searching Health-Related Grey Literature tool.50 Included in this search were the websites of regulatory agencies (US FDA and European Medicines Agency). Google was used to search for additional internet-based materials. Refer to Appendix 1 for more information on the grey literature search strategy.
Two CADTH clinical reviewers independently selected studies for inclusion in the review based on titles and abstracts, according to the predetermined protocol. Full-text articles of all citations considered potentially relevant by at least 1 reviewer were acquired. Reviewers independently made the final selection of studies to be included in the review, and differences were resolved through discussion.
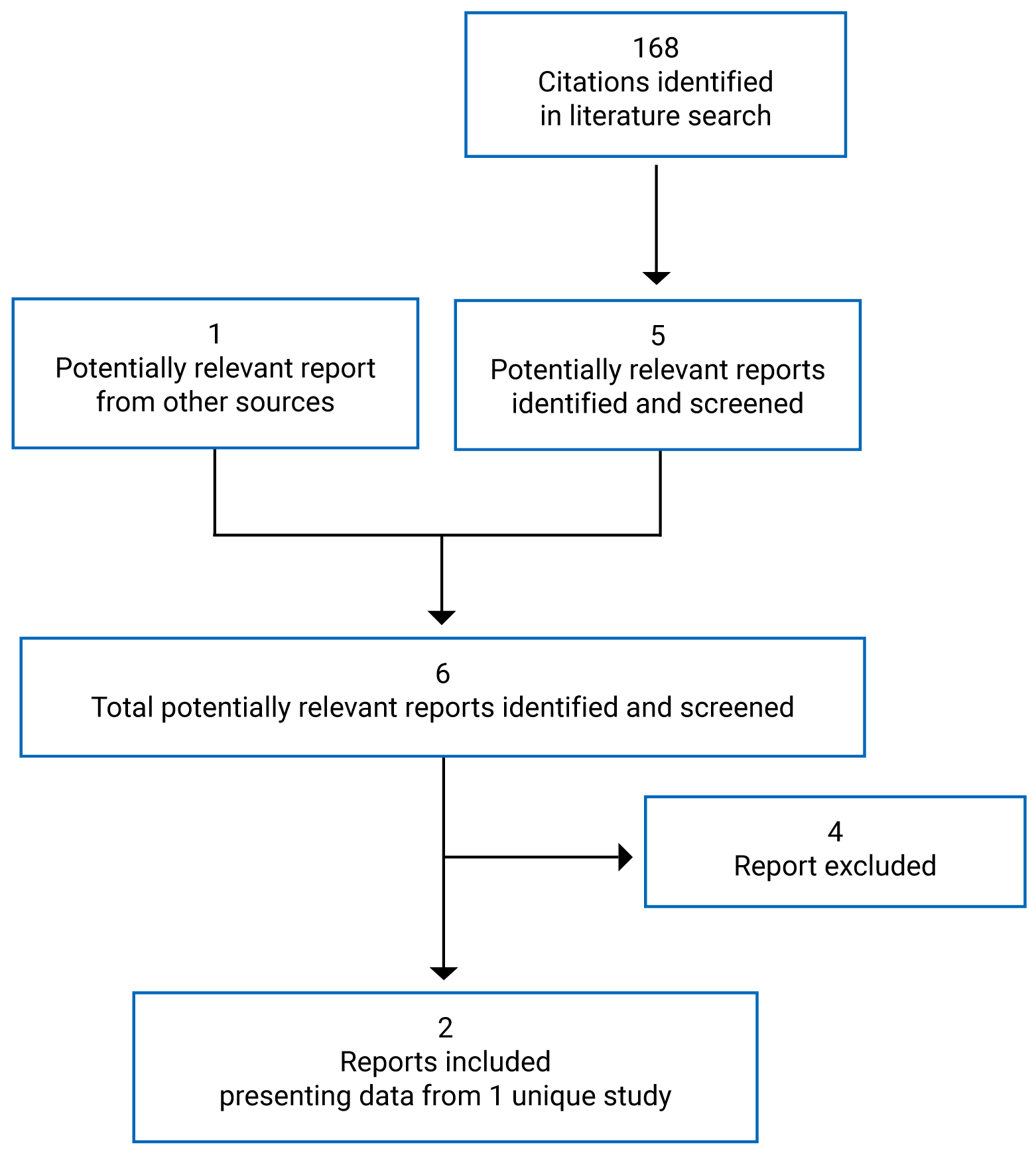
Findings From the Literature
A total of 1 study (KEYNOTE-775)19 was identified from the literature for inclusion in the systematic review (Figure 1). The included study, presented in 2 documents,19,51 is summarized in Table 6. A list of excluded studies is presented in Appendix 2.
Table 6: Details of Included Studies
Study design | KEYNOTE-775 (all comers included pMMR and dMMR)a |
|---|---|
Designs and populations | |
Study design | Open-label, multi-centre, phase III, parallel, active-controlled RCT |
Locations | 167 global sites in 21 countries Region 1: Europe, US, Canada (11 sites; N = 58 patients), Australia, New Zealand, Israel Region 2: Rest of world (e.g., South American, Central American, and Asian countries) |
Patient enrolment dates | June 11, 2018 to February 03, 2020 Cut-off date for interim analysis 1: October 26, 2020 |
Randomized (N)b | 697 for pMMR |
Inclusion criteria |
|
Exclusion criteria |
|
Drugs | |
Intervention | Pembrolizumab IV infusion over 30 minutes in combination with oral lenvatinib as follows:
Maximum doses of study drugs:
|
Comparators | TPC consisting of either:
Maximum doses of study drugs:
|
Duration | |
Phase | Phase III |
Screen | 4 weeks |
Open label | Up to 24 months or 35 doses of 200 mg of pembrolizumab |
Follow-up |
|
Outcomes | |
Primary end points | PFS OS |
Secondary and exploratory end points | Secondary: ORR Exploratory:
PROs and HRQoL:
|
Notes | |
Publications | Makker et al. (2022)51 |
BICR = blinded independent central review; CNS = central nervous system; DCR = disease control rate; dMMR = mismatch repair deficient; DOR = duration of response; ECOG PS = Eastern Cooperative Oncology Group Performance Status; EORTC QLQ-C30 = European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30; EORTC QLQ-EN24 = European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Endometrial Cancer Module; EQ-5D-5L VAS = 5-Level EQ-5D Visual Analogue Scale; HRQoL = health-related quality of life; GHS = Global Health Status; MMR = mismatch repair; ORR = objective response rate; OS = overall survival; PD = progressive disease; PD-1 = programmed cell death 1; PD-L1 = programmed cell death ligand 1; PD-L2 = programmed cell death ligand 2; PFS = progression-free survival; PFS2 = progression-free survival on next-line therapy; pMMR = mismatch repair proficient; PRO = patient-reported outcome; QoL = quality of life; QTc = heart-rate corrected QT; RCT = randomized controlled trial; RECIST = Response Evaluation Criteria in Solid Tumours; q.3.w. = every 3 weeks; q.8.w. = every 8 weeks; q.12.w. = every 12 weeks; TPC = treatment of physician’s choice; VEGF = vascular endothelial growth factor.
Note: Only information related to pMMR is reported in the rest of this report, unless otherwise specified. There is no major impact of the protocol amendments on the pMMR population (Table 26 in Appendix 3).
aThe information reported in Table 6 is for all comers, including pMMR and dMMR. There is no information only for pMMR.
bThe number of randomized patients is for pMMR only, which is the population of interest in this review. Please note that overall, the KEYNOTE-775 study was conducted in patients with pMMR or dMMR cancer. A total of 827 patients were included (all comers: N = 827; pMMR = 697; dMMR: N = 130). Also note that pMMR was not considered a subgroup in the original study design. The sample size and power calculations were based on pMMR populations. dMMR was designed as a subgroup. In this review, only the pMMR population is of interest, which aligns with Health Canada’s indication and the sponsor’s reimbursement request.8,19,32
Source: Clinical Study Report19 and Makker et al. (2022).51
Description of Studies
The KEYNOTE-775 trial is an ongoing, phase III, randomized, open-label, active-controlled, multi-centre superiority study comparing PEM + LEN to TPC (doxorubicin or paclitaxel) for the treatment of adult patients with advanced endometrial carcinoma who have disease progression following prior platinum-based systemic therapy in any setting and are not candidates for curative surgery or radiation.
The trial was conducted at 167 global sites in 21 countries (Canada, US, and 19 other countries in Europe, South America, Central America, and Asia). Fifty-eight patients in Canada at 11 Canadian research sites participated. The key characteristics of the study design are summarized in Table 6 and Figure 2.
The primary objective of this study was to assess whether PEM + LEN prolongs PFS and OS when compared to TPC. The secondary objectives were to compare the ORR, EORTC QLQ-C30 GHS, and the safety and tolerability of PEM + LEN combination therapy versus TPC. Other objectives (as exploratory) were to assess the DOR and EORTC QLQ-C30 Physical Function scale score, the EORTC QLQ-EN24 Urological Symptoms Score, and the 5-Level EQ-5D Visual Analogue Scale (EQ-5D-5L VAS) score in patients treated with PEM + LEN combination therapy versus TPC.
Figure 2: Study Design Schematic
![The KEYNOTE-775 trial (pMMR population) included an informed consent period, a screening period (28 days), and a treatment period (i.e., PEM + LEN combination therapy or TPC [doxorubicin or paclitaxel]). During the treatment period, tumours were assessed every 8 weeks. This was also done in the post-treatment period, which included a 30-day safety follow-up period as well as follow-up for disease assessment and survival status.](https://canjhealthtechnol.ca/index.php/cjht/article/download/PC0288r/version/521/1082/3569/PC0288CL-fig02.png)
BICR = blinded independent central review; PD = progressive disease; Q8W = every 8 weeks; Q12W = every 12 weeks; RECIST 1.1 = Response Evaluation Criteria in Solid Tumours version 1.1.
Source: Clinical Study Report.19
A total of 1,178 patients were screened. In the pMMR population, 697 patients were randomized in a 1:1 ratio to receive PEM + LEN (n = 346) or TPC (n = 351). A total of 346 patients received PEM + LEN and 351 patients received TPC (doxorubicin or paclitaxel). Treatment allocation and randomization were done centrally using an interactive response technology system. Treatment allocation and randomization were stratified according to MMR status; ECOG PS status (0 or 1); geographic region (region 1 [Europe, US, Canada, Australia, New Zealand, and Israel] or region 2 [rest of the world]); and prior history of pelvic radiation (yes or no). The first patient was randomized on June 11, 2018, and the last patient was enrolled on February 03, 2020. The cut-off date for IA1 was October 26, 2020. A total of 667 patients received at least 1 dose of PEM + LEN (N = 342 [98.8%]) or TPC (N = 325 [92.6%]) as assigned. Four patients (1.2%) in the PEM + LEN arm and 26 patients (7.4%) in the TPC arm did not receive treatment. No reasons were provided for this.
Pre-specified interim analysis results (cut-off date of October 26, 2020) for the primary outcomes (PFS and OS), secondary outcomes (ORR and EORTC QLQ-30 GHS), ORR, and other exploratory outcomes (e.g., DOR, HRQoL outcomes, and safety outcomes) were provided in the submission. The sponsor indicated that the PFS and OS results in IA1 were considered final because both the PFS and OS analyses met the pre-specified statistical significance threshold.18
There is no major impact of protocol amendments on the pMMR population (Table 26 in Appendix 3).
Populations
Inclusion and Exclusion Criteria
Eligible patients were those (≥ 18 years old) with confirmed advanced, recurrent, or metastatic endometrial cancer of any histologic subtype except for carcinosarcoma and sarcoma. Patients had disease progression after receiving 1 previous platinum-based chemotherapy regimen, with no history of exposure to regimens targeting VEGF or PD-1. Patients may have received 2 lines of platinum-based chemotherapy if 1 was given as neoadjuvant or adjuvant therapy. There was no restriction regarding the previous receipt of hormonal therapy. Other criteria included patients who had an ECOG PS score of 0 or 1. The main exclusion criteria were carcinosarcoma, endometrial leiomyosarcoma, and endometrial stromal sarcomas; patients with CNS metastases were also excluded unless they had completed local therapy and discontinued the use of corticosteroids for this indication at least 4 weeks before starting treatment in this study. Patients who had received more than 1 prior systemic chemotherapy regimen (other than adjuvant or neoadjuvant therapy) for endometrial cancer were excluded. Patients with prior treatment with any drug targeting VEGF-directed angiogenesis, or any anti-PD-1, anti-PD-L1, or anti-PD-L2 drug, were excluded. Refer to Table 6.
Baseline Characteristics
The main baseline demographic and disease characteristics of all randomized patients with pMMR tumours are summarized in Table 7, and appeared balanced between the PEM + LEN and TPC arms.
The median age was 65 years (range = 30 years to 86 years). Patients had a baseline ECOG PS of either 0 (60.1%) or 1 (39.7%). One patient (0.3%) in the PEN+LEN arm had an ECOG PS of 3. A total of 431 patients (61.8%) were White and 154 patients (21.4%) were Asian. A total of 19.2% patients had endometrioid carcinoma, while 41.6% of patients reported a history of prior pelvic radiation (refer to Table 7).
Details about prior therapies for endometrial cancer were reported for the pMMR population. Treatment with 1 previous platinum-based therapy was reported for 77.7% of the patients in the PEM + LEN group and for 73.2% of those in the chemotherapy group. Treatment with 2 prior platinum-based therapies (including neoadjuvant or adjuvant therapies) was reported in 21.7% patients in the PEM + LEN arm and in 26.8% of patients in the TPC arm; 1 patient (0.3%) in the PEM + LEN arm had received 3 or more platinum-based therapies. It was reported that 56.9% of patients in the PEM + LEN arm and 62.4% of patients in the TPC arm had previously received systemic treatment as neoadjuvant or adjuvant therapy: 36.1% of patients in the PEM + LEN arm and 37.9% of patients in the TPC arm had previously received only neoadjuvant or adjuvant therapy; 8.7% and 10.0% had received palliative hormonal therapy previously, respectively; and 44.8% and 45.3% had received external beam radiotherapy previously, respectively. History of hysterectomy was reported in 72.8% patients in the PEM + LEN arm and 79.5% of patients in the TPC arm. (Refer to Table 7 in this section and Table 29 in Appendix 3.)
Table 7: Participant Characteristics in Patients With pMMR Tumours (ITT Population)
Characteristic | KEYNOTE-775 | |
|---|---|---|
PEM + LEN (N = 346) | TPC (N = 351) | |
Total # of patients, N | 346 | 351 |
Sex | ||
Female, n (%) | 346 (100.0) | 351 (100.0) |
Age (years), n (%) | ||
< 65 | 171 (49.4) | 165 (47.0) |
≥ 65 | 175 (50.6) | 186 (53.0) |
Median (range) | 65.0 (30 to 82) | 66.0 (35 to 86) |
Race, n (%) | ||
Asian | 74 (21.4) | 80 (22.8) |
White | 220 (63.6) | 211 (60.1) |
Other | 23 (6.6) | 24 (6.8) |
Missing | 29 (8.4) | 36 (10.3) |
Region,a n (%) | ||
Region 1 | 1,202 (58.4) | 204 (58.1) |
Region 2 | 2,144 (41.6) | 147 (41.9) |
ECOG Performance Status, n (%) | ||
0 | 212 (61.3) | 207 (59.0) |
1 | 133 (38.4) | 144 (41.0) |
3 | 1 (0.3) | 0 (0.0) |
Prior history of pelvic radiation, n (%) | ||
Yes | 142 (41.0) | 148 (42.2) |
No | 204 (59.0) | 203 (57.8) |
Elapsed time (years) from initial diagnosis, n (%) | ||
Patients with data | 346 | 351 |
Median (range) | 1.7 (0 to 21) | 2.1 (0 to 26) |
Histology of initial diagnosis, n (%) | ||
Clear-cell carcinoma | 29 (8.4) | 17 (4.8) |
Endometrioid carcinoma | 60 (17.3) | 74 (21.1) |
Endometrioid carcinoma with squamous differentiation | 5 (1.4) | 6 (1.7) |
High-grade endometrioid carcinoma | 73 (21.1) | 77 (21.9) |
High-grade mucinous carcinoma | 0 (0.0) | 1 (0.3) |
High-grade serous | 62 (17.9) | 64 (18.2) |
Low-grade endometrioid carcinoma | 50 (14.5) | 41 (11.7) |
Low-grade mucinous carcinoma | 1 (0.3) | 0 (0.0) |
Mixed | 18 (5.2) | 13 (3.7) |
Neuroendocrine | 2 (0.6) | 0 (0.0) |
Serous carcinoma | 37 (10.7) | 48 (13.7) |
Unclassified | 0 (0.0) | 2 (0.6) |
Undifferentiated histology | 4 (1.2) | 2 (0.6) |
Other | 5 (1.4) | 6 (1.7) |
FIGO stage at initial diagnosis, n (%) | ||
I | 9 (2.6) | 10 (2.8) |
IA | 41 (11.8) | 53 (15.1) |
IB | 40 (11.6) | 51 (14.5) |
II | 30 (8.7) | 22 (6.3) |
III | 5 (1.4) | 6 (1.7) |
IIIA | 23 (6.6) | 29 (8.3) |
IIIB | 11 (3.2) | 8 (2.3) |
IIIC | 22 (6.4) | 20 (5.7) |
IIIC1 | 14 (4.0) | 20 (5.7) |
IIIC2 | 22 (6.4) | 20 (5.7) |
IV | 25 (7.2) | 23 (6.6) |
IVA | 4 (1.2) | 7 (2.0) |
IVB | 100 (28.9) | 82 (23.4) |
# of patients with primary lesion or metastasis at initial diagnosis,c n (%) | ||
Brain | 1 (0.3) | 2 (0.6) |
Bone | 34 (9.8) | 31 (8.8) |
Liver | 93 (26.9) | 92 (26.2) |
Lung | 142 (41.0) | 135 (38.5) |
Intra-abdominal b | 171 (49.4) | 160 (45.6) |
Lymph node | 204 (59.0) | 203 (57.8) |
# of patients with primary lesion or metastasis at initial diagnosis,c n (%) | ||
Brain | 1 (0.3) | 0 (0.0) |
Bone | 12 (3.5) | 7 (2.0) |
Liver | 19 (5.5) | 13 (3.7) |
Lung | 26 (7.5) | 13 (3.7) |
Intra-abdominal b | 94 (27.2) | 85 (24.2) |
Lymph node | 115 (33.2) | 108 (30.8) |
ECOG = Eastern Cooperative Oncology Group; FIGO = International Federation of Gynecology and Obstetrics; ITT = intention to treat; PEM + LEN = pembrolizumab plus lenvatinib combination therapy; pMMR = mismatch repair proficient; TPC = treatment of physician’s choice (doxorubicin or paclitaxel).
aRegion 1 = Europe, US, Canada, Australia, New Zealand, Israel; region 2 = rest of world.
bIncludes reported locations of colon, abdominal cavity, omentum, small intestine, peritoneal cavity, and peritoneum. Does not include lymph nodes or other organs.
cLesion location as determined by investigator review.
Source: Clinical Study Report.19
Interventions
The study intervention(s) and comparators are detailed in Table 28 in Appendix 3.
Intervention and comparators: Patients received treatment with either PEM + LEN (N = 342) or TPC (N = 325) (doxorubicin [N = 239] or paclitaxel [N = 86]). Patients in the PEM + LEN arm received IV infusion of PEM 200 mg over 30 minutes every 3 weeks for up to 35 cycles or a total duration of 24 months in combination with 20 mg of LEN once daily, orally. Patients in the TPC arm received either doxorubicin (N = 239) (60 mg/m2 by IV every 3 weeks) or paclitaxel (N = 86) (80 mg/m2 by IV once a week). Paclitaxel was administered in 28-day cycles (i.e., with weekly administration, 3 weeks on and 1 week off).
Treatment cycles were counted continuously regardless of dose interruptions or missed doses for both treatment arms.
Patients continued to receive the study treatment until disease progression, development of unacceptable toxicity, withdrawal of consent, receipt of 35 administrations of PEM (approximately 2 years), or a cumulative dose of 500 mg/m2 of doxorubicin. Discontinuation of PEM treatment was considered for patients who had attained a confirmed complete response (CR), been treated with PEM for at least 8 cycles (at least 24 weeks), and had at least 2 treatments with PEM beyond the date when the initial CR was declared. Patients who stopped the study treatment after receiving 35 administrations of PEM for reasons other than disease progression or intolerability, or who attained a CR and stopped PEM, may be eligible for up to an additional year of treatment with PEM (17 cycles), with or without LEN, upon experiencing disease progression (i.e., second course phase).19 In the presence of clinical benefit, patients who completed 35 cycles of treatment with PEM (approximately 2 years) could continue on LEN alone beyond this time point.
Patients were permitted to continue the study treatment beyond disease progression if the maximum dosage of the study drugs had not been reached (e.g., 35 administrations of PEM or a cumulative dose of 500 mg/m2 of doxorubicin), as long as the treating investigator considered that the participant could experience a clinical benefit from continued treatment and was tolerating the treatment.
Patients who completed PEM continued to receive LEN until disease progression, development of unacceptable toxicity, or withdrawal of consent. Following the demonstration of a survival benefit in the interim analysis, eligible patients in the TPC group who experienced disease progression were allowed to cross over to receive LEN + PEM. PEM dose reductions were not permitted for the management of toxicities of individual patients. However, doses of PEM were allowed to be interrupted, delayed, or discontinued, depending on how well the patient tolerated the treatment.
Lenvatinib
The starting dose of LEN was 20 mg/day for patients enrolled. Dose reductions of LEN occurred in succession based on the previous dose level (14 mg/day, 10 mg/day, and 8 mg/day).
LEN dose reduction and interruption were allowed for patients experiencing toxicity related to PEM + LEN combination therapy. The dose reduction on LEN is presented in Table 13 and Table 39 in Appendix 3. A total of 229 patients (67.0%) treated with PEM + LEN experienced a dose reduction of LEN, and 42 patients (12.9%) treated with TPC (doxorubicin or paclitaxel) experienced a dose reduction (refer to Table 39).
Information about dose interruption and/or delay in pMMR is presented in Table 13 in this section and Table 38 in Appendix 3. A total of 235 patients (68.7%) in the PEM + LEN group and 91 patients (28%) in the TPC group experienced a dose interruption. In the PEM + LEN group, a total of 100 patients (29.2%), 199 patients (58.2%), and 165 patients (48.2%) experienced PEM + LEN, LEN, and PEM dose interruptions, respectively (Table 13).
Information about the administration of PEM, LEN, doxorubicin, and paclitaxel is summarized in Table 41 in Appendix 3.
Concomitant Treatments
Patients were prohibited from receiving the following therapies during the screening and treatment phases of the study: concurrent anticancer therapies (e.g., chemotherapy), targeted therapies (e.g., tyrosine kinase inhibitors), hormonal therapy directed at endometrial carcinoma, radiotherapy (with the exception of palliative radiotherapy, as specified), antitumour interventions (e.g., surgical resection, surgical debulking of tumour), or cancer immunotherapy. Patients were permitted to use the following concomitant medications: hormone replacement therapy; thyroid hormone suppressive therapy; adjuvant hormonal therapy for history of definitely treated breast cancer; anticoagulants, including low molecular weight heparin, warfarin, and anti-Xa drugs; anti-inflammatory drugs; bisphosphonates or denosumab; antihypertensive therapies (including additional antihypertensive treatment as appropriate if blood pressure increased once the participant was enrolled); palliative radiotherapy for up to 2 painful, pre-existing, non-target bone metastases. The details of the potential relevant concomitant medications used in the study are presented in Table 30.
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Subsequent Cancer Therapy During the Trial
Of the 346 patients in the PEM + LEN arm, 109 patients (31.5%) received any subsequent systemic anticancer therapy. Of the 351 patients in the TPC arm, 176 patients (50.1%) received subsequent therapy (Table 31). The most common form of subsequent anticancer therapy was chemotherapy, which was received by 92 patients (26.6%) in the PEM + LEN group and 119 patients (33.9%) in the TPC group; 4 patients (1.2%) in the PEM + LEN group and 42 patients (12.0%) in the TPC group received any subsequent PD-1 or PD-L1 inhibitors. Twenty-four patients (6.9%) in the PEM + LEN group and 51 patients (14.5%) in the TPC group received hormonal therapy.
A total of 6 patients (1.7%) in the PEM + LEN group and 11 patients (3.1%) in the TPC group received 1 line of subsequent systemic therapy; 81 patients (23.4%) in the PEM + LEN group and 134 patients (38.2%) in the TPC group received 2 lines of subsequent systemic therapy; 55 patients (15.9%) in the PEM + LEN group and 78 patients (22.0%) in the TPC group received 3 or more subsequent lines of systemic therapy (Table 31).
Outcomes
A list of efficacy end points identified in the CADTH review protocol that were assessed in the clinical trials included in this review is provided in Table 8. These end points are further summarized in this section. A detailed discussion and critical appraisal of the outcome measures is provided in Appendix 4.
The primary outcomes of the KEYNOTE-775 trial were OS and PFS. OS was defined as the time from the date of randomization to the date of death from any cause. PFS was defined as the time from the date of randomization to the date of the first documentation of disease progression, or death from any cause (whichever occurred first). The secondary outcome was ORR, defined as the proportion of patients who achieved either CR or partial response.
DOR was an exploratory outcome. It was defined as the time from the date a response was first documented until the date of the first documentation of disease progression or date of death, whichever occurred first.
Patient-reported and HRQoL outcomes included the EORTC QLQ-C30 GHS scale, the EORTC QLQ-C30 Physical Functioning scale, the EORTC QLQ-EN24 Urological Symptoms Score, and the EQ-5D-5L VAS score (refer to Table 8).
Table 8: Summary of Outcomes of Interest Identified in the CADTH Review Protocol
Outcome measure | KEYNOTE-775 | Description |
|---|---|---|
OS | Primary | Defined as the time from date of randomization to date of death from any cause |
PFS | Primary | Defined as the time from date of randomization to the date of the first documentation of disease progression, as determined by BICR per RECIST 1.1, or death from any cause (whichever occurred first) |
EORTC QLQ-C30 GHS | Secondary | A self-reported instrument designed to measure the physical, psychological, and social functions of patients with cancers |
ORR | Secondary | ORR, defined as the proportion of patients who have the best overall response of either complete or partial response, as determined by BICR per RECIST 1.1 |
DOR | Exploratory | Defined as the time from the date a response was first documented until the date of the first documentation of disease progression by BICR and investigator assessment of objective radiographic disease assessment per RECIST 1.1, or date of death, whichever occurred first |
Patient-reported and HRQoL outcomes | ||
EORTC QLQ-C30 GHS | Secondary | A self-reported instrument designed to measure the physical, psychological, and social functions of patients with cancers The EORTC QLQ-C30 incorporates 5 functional scales (Physical, Role, Cognitive, Emotional, Social); 3 symptom scales (Fatigue; Pain; Nausea and Vomiting); a Global Health Status scale; and single items that assess additional symptoms commonly reported by patients with cancer (e.g., dyspnea, appetite loss, sleep disturbance, constipation, and diarrhea). It also incorporates the perceived financial impacts of the disease and treatment.52-54 |
EORTC QLQ-30 Physical Functioning scale | Exploratory | A self-reported instrument designed to measure the physical, psychological, and social functions of patients with cancers The EORTC QLQ-C30 incorporates 5 functional scales (Physical, Role, Cognitive, Emotional, Social); 3 symptom scales (Fatigue; Pain; Nausea and Vomiting); a Global Health Status scale; and single items that assess additional symptoms commonly reported by patients with cancer (e.g., dyspnea, appetite loss, sleep disturbance, constipation, and diarrhea). It also incorporates the perceived financial impact of the disease and treatment.52-54 |
EORTC QLQ-EN24 Urological Symptoms Score | Exploratory | A 24-item, self-reported questionnaire for patients at all stages of endometrial cancer. The EORTC QLQ-EN24 consists of 10 symptom scales (lymphedema, urological symptoms, gastrointestinal symptoms, poor body image, sexual/vaginal problems, pain in the back and pelvis, tingling/numbness, muscular pain, hair loss, and taste change) and 3 functional scales (sexual interest, activity, and enjoyment). |
EQ-5D VAS | Exploratory | A generic, self-reported HRQoL tool that assesses 5 domains: mobility, self-care, usual activities, pain/discomfort, and anxiety/depression. Each domain has 5 levels: no problems; slight problems; moderate problems; severe problems; and extreme problems. The EQ-5D-5L also includes an EQ VAS, a vertical visual analogue scale, a global assessment of patients’ health with values that range from 100 (best imaginable health) to 0 (worst imaginable health).55-58 |
BICR = blinded independent central review; DOR = duration of response; EORTC QLQ-C30 = European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30; EQ VAS: EQ visual analogue scale; EORTC QLQ-EN24 = European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Endometrial Cancer Module; EQ-5D VAS = EQ-5D Visual Analogue Scale; HRQoL = health-related quality of life; ORR = objective response rate; OS = overall survival; PFS = progression-free survival; RECIST 1.1 = Response Evaluation Criteria in Solid Tumours version 1.1.
Statistical Analysis
Analysis strategies are summarized in Table 32 in Appendix 3.
Primary Outcomes: PFS and OS
Power Calculation
Note that the sample size and power calculations were based on the pMMR population.
With approximately 368, 463, and 526 OS events in the pMMR population at the planned IA1 (at 27 months follow-up), IA2 (at 35 months’ follow-up), and final OS analysis (at 43 months’ follow-up), respectively, the study would have 90% power to detect an HR of 0.75 at the 1-sided 0.0245 significance level.
The study was designed to have 90% power to detect a statistically significant difference in OS at a 1-sided alpha equal to 0.0245. As a result, the study had at least 99% power to detect a statistically significant difference in PFS at a 1-sided alpha equal to 0.0005. Assuming an accrual period of 19 months and a follow-up period of 24 months, a total of 660 patients were required to observe 526 death events by 43 months after the first participant was randomized (i.e., 19 months’ enrolment plus 24 months’ follow-up).
For OS, a total of 526 OS events were required to detect a statistically significant difference at a 0.0245 level with 90% power, assuming that the HR was 0.75 (median OS = 16.4 months in the PEM + LEN arm and 12.3 months in the TPC arm); the IA1 was performed when approximately 368 OS events were observed (i.e., 70% of the total target death events); the IA2 analysis was planned when approximately 463 OS events were observed (i.e., 88% of the total target death events); and the Lan-DeMets spending function with O’Brien-Fleming boundary was used.
The final PFS analysis was planned to be performed at the time of IA1, 27 months after the first participant was randomized. A total of 564 PFS events were estimated to be observed to detect a statistically significant difference at a 0.0005 level with greater than 99% power under the assumption that the HR was 0.55 (median PFS was 7.3 months in the PEM + LEN arm and 4 months in the TPC arm).
It was indicated that the success criteria for the study hypotheses of PFS, OS, and ORR were all met at IA1; consequently, the IA2 was not required.17,18
Statistical Test or Model
Median PFS and OS for each treatment arm were estimated and plotted using the Kaplan-Meier product-limit method (refer to Table 9). The primary hypotheses were evaluated by comparing PFS and OS using a stratified log-rank test. The HRs were estimated using a stratified Cox regression model. Event rates over time were estimated within each treatment group using the Kaplan-Meier method. The HRs for PFS and OS were assessed based on the Cox regression model with the Efron method of tie handling with treatment as a covariate stratified by ECOG PS, geographic region, and prior history of pelvic radiation in the pMMR population (refer to Table 9).
Subgroup Analyses
The influences of baseline demographics and disease characteristics on the treatment effect among all randomized patients were examined by OS and PFS HRs (along with the 95% Cis). The key subgroups included ECOG PS (0 or 1), histology (endometrioid or non-endometrioid), and the number of lines of prior systemic anticancer therapy (1, 2, and ≥ 3). Only those subgroups identified in the CADTH review protocol are reported in the Efficacy section.
Sensitivity Analyses
Two sensitivity analyses with different sets of censoring rules for PFS were performed. The first sensitivity analysis follows the ITT principle. That is, progressive disease and deaths were counted as events regardless of missed study visits or initiation of new anticancer therapies The second sensitivity analysis considered the initiation of new anticancer treatment or the discontinuation of treatment due to reasons other than CR. If a participant met multiple criteria for censoring, the censoring criterion that occurred earliest was applied (Table 9).
Table 9: Statistical Analysis of Efficacy End Points in the KEYNOTE-775 Trial (pMMR)
End point | Statistical model for | Adjustment factors | Sensitivity analyses |
|---|---|---|---|
PFS | The primary hypotheses was evaluated by comparing PFS using a stratified log-rank test. The HR was estimated using a stratified Cox regression model. Event rates over time were estimated within each treatment group using the Kaplan-Meier method. | Based on a Cox regression model with the Efron method of tie handling with treatment as a covariate stratified by MMR status, ECOG Performance Status, geographic region, and prior history of pelvic radiation | Two sensitivity analyses with different sets of censoring rules were performed. The first followed the intention-to-treat principle: that is, disease progressions and deaths were counted as events regardless of missed study visits or initiation of new anticancer therapy. The second sensitivity analysis considered the initiation of new anticancer treatment or the discontinuation of treatment due to reasons other than complete response — whichever occurs later — to be a PD event for patients without documented PD or death. If a participant met multiple criteria for censoring, the censoring criterion that occurred earliest was applied. The censoring rules for the primary and sensitivity analyses are summarized in Table 32. |
OS | The primary hypotheses was evaluated by comparing OS using a stratified log-rank test. The HR was estimated using a stratified Cox regression model. Event rates over time were estimated within each treatment group using the Kaplan-Meier method. | Based on a Cox regression model with the Efron method of tie handling with treatment as a covariate stratified by MMR status, ECOG Performance Status, geographic region, and prior history of pelvic radiation | Not conducted |
ORR | The primary hypotheses were evaluated by comparing ORR using a stratified log-rank test. The HR was estimated using a stratified Cox regression model. Event rates over time were estimated within each treatment group using the Kaplan-Meier method. | Based on a Cox regression model with the Efron method of tie handling with treatment as a covariate stratified by MMR status, ECOG Performance Status, geographic region, and prior history of pelvic radiation | Not conducted |
ECOG = Eastern Cooperative Oncology Group; HR = hazard ratio; MMR = mismatch repair; ORR = objective response rate; OS = overall survival; PD = progressive disease; PFS = progression-free survival; pMMR = mismatch repair proficient.
Source: Clinical Study Report.19
Secondary Outcomes: ORR and HRQoL
The stratified Miettinen and Nurminen method was used to compare ORR between 2 treatment groups. The difference in ORR and its 95% CI from the stratified Miettinen and Nurminen method with strata weighting by sample size was reported. The stratification factors used for randomization were applied to the analysis. The point estimate of ORR was provided by treatment group, together with 95% CI, using the exact binomial method proposed by Clopper and Pearson.59 Cis were not adjusted for multiplicity.
In addition, the EORTC QLQ-C30 GHS was used as a secondary outcome. The HRQoL analyses were based on the HRQoL and patient-reported outcome (PRO) full analysis set (FAS) population. The FAS was defined as all randomized patients who had at least 1 HRQoL assessment available for the specific end point and had received at least 1 dose of the study intervention. Patients were analyzed in the treatment group to which they were randomized. The Clinical Study Report did not report on how the missing data were handled (i.e., last observation carried forward, multiple imputation, or other).
Multiplicity
The total family-wise error rate (type I error) among the dual-primary PFS and OS and the secondary ORR end points was controlled at a 1-sided 0.025 level. A type I error rate of 0.0005 was initially allocated to test PFS, and a type I error rate of 0.0245 was initially allocated to test OS between 2 treatment arms in patients with pMMR tumours. Details about the alpha allocation strategy among the hypotheses for PFS, OS, and ORR are provided in Figure 6, Appendix 3. The secondary outcome, EORTC QLQ-C30 GHS, was not controlled for multiplicity.
Other Exploratory Outcomes
Patient-reported and HRQoL outcomes, including the EORTC QLQ-C30 Physical Functioning scale, the EORTC QLQ-EN24 Urological Symptoms Score, and the EQ-5D-5L VAS score, were assessed using a constrained longitudinal data analysis (cLDA) model, with PRO scores as the response variable with covariates for treatment by study visit interaction, MMR status, ECOG PS, geographic region, and prior history of pelvic radiation. The mean scores and mean changes from baseline to week 12 for these exploratory outcomes were summarized by treatment arm at the week 12 assessment.
Safety Outcomes
Only descriptive statistics of safety were presented.
Analysis Populations
The efficacy populations in the KEYNOTE-775 trial for the analysis of the primary outcomes of OS and PFS and the secondary outcome of DOR were the ITT populations (i.e., all randomized patients; refer to Table 10). The DOR was assessed based on the number of responders. The HRQoL analyses were based on the HRQoL PRO FAS population, defined as all randomized patients with at least 1 HRQoL assessment available for the specific end point who received at least 1 dose of the study intervention. Patients were analyzed in the treatment groups to which they were randomized. The safety analysis was assessed based on all patients as treated.
Results
Patient Disposition
Patient disposition in the KEYNOTE-775 study (for the pMMR population) is presented in Table 10. Of the 1,178 patients with pMMR tumours screened, a total of 697 were randomized to receive PEM + LEN (n = 346) or TPC placebo (n = 351). Of the 697 randomized patients, 4 (1.2%) in the PEM + LEN arm and 26 (7.4%) in the TPC arm did not receive their assigned treatment. No reason was provided for this19 (Table 10).
At the time of the data cut-off date of October 26, 2020, a total of 485 patients (72.7%) discontinued treatment (n = 247 [72.2%] in the PEM + LEN arm and n = 238 [73.2%] in the TPC arm). The main reasons for discontinuation of treatment were disease progression (N = 156 [45.6%] in the PEM + LEN arm versus N = 144 [44.3%] in TPC arm); AEs (N = 56 [16.4%] in the PEM + LEN arm versus N = 29 [8.9%] in the TPC arm); withdrawal by patient (N = 17 [5.0%] in the PEM + LEN arm versus N = 23 [7.1%] in the TPC arm), clinical progression (N = 13 [3.8%] in the PEM + LEN arm versus N = 19 [5.8%] in the TPC arm); physician decision (N = 3 [0.9%] in the PEM + LEN arm versus N = 17 [5.2%] in the TPC arm); and CR (N = 2 [0.6%] in the PEM + LEN arm versus N = 3 [0.9%] in the TPC arm). A total of 95 patients (27.8%) in the PEM + LEN arm and 9 patients (2.8%) in the TPC arm were still receiving the treatment at the time of data cut-off.
The efficacy population (i.e., ITT population) included 697 patients, while the safety population included 668 patients.
Table 10: Disposition of Patients With pMMR Tumours (ITT Population)
Patient disposition | PEM + LEN (N = 346) | TPC (N = 351) |
|---|---|---|
Screened, N | 1,178 | — |
Randomized | 346 | 351 |
Patients treated, n (%) | 342 | 325 |
Patients not treated, n (%) | 4 | 26 |
Reason for not being treated, n (%) | NR | NR |
Status for trial, n (%) | ||
Discontinued | 168 (48.6) | 219 (62.4) |
Death | 161 (46.5) | 196 (55.8) |
Lost to follow-up | 0 (0.0) | 2 (0.6) |
Withdrawal by patient | 7 (2.0) | 21 (6.0) |
Patients ongoing | 178 (51.4) | 132 (37.6) |
Status for study medication in trial, n (%) | ||
Started | 342 | 325 |
Completed | 0 (0.0) | 78 (24.0) |
Discontinued | 247 (72.2) | 238 (73.2) |
Adverse event | 56 (16.4) | 29 (8.9) |
Clinical progression | 13 (3.8) | 19 (5.8) |
Complete response | 2 (0.6) | 3 (0.9) |
Non-compliance with study drug | 0 (0.0) | 1 (0.3) |
Non-study anticancer therapy | 0 (0.0) | 2 (0.6) |
Physician decision | 3 (0.9) | 17 (5.2) |
Progressive disease | 156 (45.6) | 144 (44.3) |
Withdrawal by patient | 17 (5.0) | 23 (7.1) |
Patients ongoing | 95 (27.8) | 9 (2.8) |
ITT, N | 346 | 351 |
PP, N | Not performed | Not performed |
Safety (APaT), N | 343 | 325 |
APaT = all patients as treated; ITT = intention to treat; PEM + LEN = pembrolizumab plus lenvatinib combination therapy; NR = not reported; pMMR = mismatch repair proficient; PP = per protocol; TPC = treatment of physician’s choice (doxorubicin or paclitaxel).
Notes: If the overall count of patients is calculated and displayed in a cell in the first row, then it is used as the denominator for the percentage calculation. Otherwise, the numbers of patients in population is used as the denominator for the percentage calculation.
For PEM + LEN, completion of study medication means 35 infusions of pembrolizumab. For TPC of doxorubicin, it indicates having received a lifetime maximum cumulative dose of doxorubicin. For TPC of paclitaxel, it indicates that per the investigator, a maximum tolerable dose was reached.
Source: Clinical Study Report.19
Exposure to Study Treatments
Information about drug exposures for the pMMR population is presented in Table 36.
The median duration on treatment was longer in the PEM + LEN arm than in the TPC arm. As of the data cut-off date, the median treatment durations were 7.22 months (range = < 0.03 to 26.9, reported as 219.5 days; range = 1 day to 817 days in the Clinical Study Report) and 3.49 months (range = < 0.03 to 25.8, reported as 106.0 days; range = 1 day to 785 days in the Clinical Study Report) in the PEM + LEN arm and TPC arm, respectively (refer to Table 36).
The median follow-up durations for pMMR were 12.2 months (range = 0.3 to 26.9) in the PEM + LEN arm and 10.8 months (range = 0.3 to 26.3) in the TPC arm (refer to Table 43 in Appendix 3).
Efficacy
Only those efficacy outcomes and analyses of subgroups identified in the review protocol are reported here.
Overall Survival
At the preplanned IA1, a total of 368 death events (165 [47.7%] in the PEM + LEN arm and 203 [57.8%] in the TPC arm) had occurred. The median OS was longer in the PEM + LEN arm (17.4 months; 95% Cl, 14.2 to 19.9) than in the TPC arm (12.0 months; 95% Cl, 10.8 to 13.3). The HR for PEM + LEN versus TPC was 0.68 (95% CI, 0.56 to 0.84; P < 0.0001) (Figure 3 and Table 11).
Figure 3: Kaplan-Meier Estimates of Overall Survival in Patients With pMMR Tumours (ITT Population)
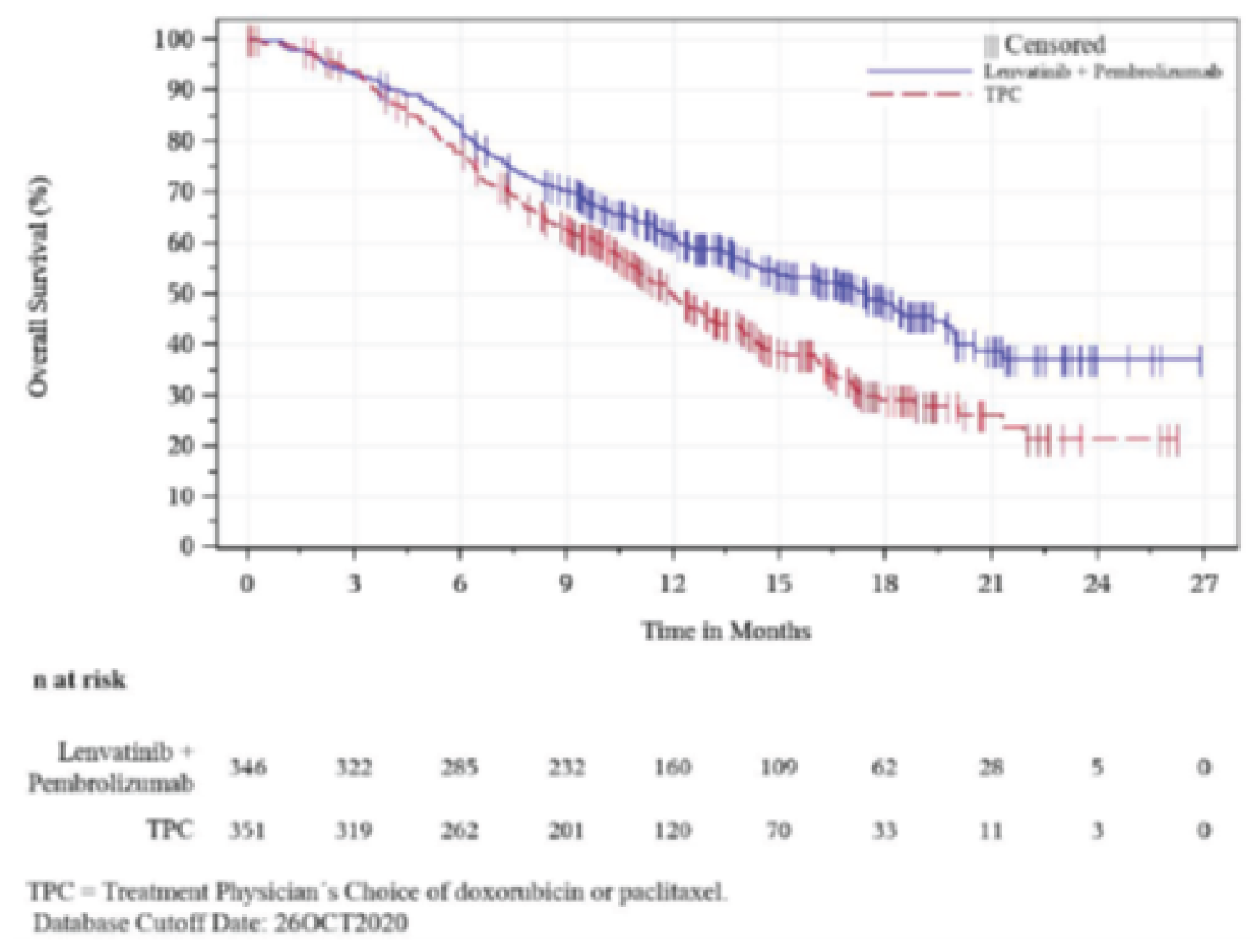
ITT = intention to treat; OS = overall survival; PEM + LEN = pembrolizumab plus lenvatinib combination therapy; pMMR = mismatch repair proficient.
Source: Clinical Study Report.19
Table 11: Key Outcomes for the KEYNOTE-775 Trial
Outcome | PEM + LEN (N = 346) | TPC (N = 351) |
|---|---|---|
OS (ITT) | ||
Events, n (%) | 165 (47.7) | 203 (57.8) |
Median OS (95% CI), monthsa | 17.4 (14. 2 to 1 9.9) | 12.0 (10.8 to 13.3) |
HRb (95% CI) | 0.68 (0.56 to 0.84) | |
P valuec | 0.0001c | |
12-month OS rates (95% CI), %a | 61.6 (56.1 to 66.6) | 49.5 (43.8 to 55.0) |
PFS (ITT) | ||
Events, n (%) | 247 (71.4) | 238 (67.8) |
Median PFS (95% CI), mo.a | 6.6 (5.6 to 7.4) | 3.8 (3.6 to 5.0) |
HRb (95% CI) | 0.60 (0.50 to 0.72) | |
P valuec | < 0.0001c | |
6-month PFS rates (95% CI), %a | 52.1 (46.5 to 57.3) | 36.2 (30.5 to 41.9) |
EORTC QLQ-C30 GHS (FAS) | ||
Baseline | ||
N (% of ITT) | 316 (91.3) | 298 (84.9) |
Mean (SD) | 66. 56 (21.44) | 66.64 (22.43) |
At week 12 | ||
N (% of ITT) | 256 (74.0) | 192 (54.7) |
Mean (SD) | 60.94 (21.35) | 62.80 (21.67) |
Change from baseline to week 12 | ||
N (% of ITT) | 327 (94.5) | 310 (98.4) |
LS mean (95% CI) | –6.80 (–9.43 to –4.17) | –7.96 (–10.86 to –5.05) |
LSM difference (95% CI)d | 1.16 (–2.49 to 4.81) | |
P valued | 0.5316 | |
ORR (ITT) | ||
N | 346 | 351 |
Events, n (%) | 105 (30.3) | 53 (15.1) |
Difference in % (95% CI)e vs. TPC | 15.2 (9.1 to 21.4) | |
P valuef | < 0.0001 | |
DOR (months) | ||
Median (range)g | 9.2 (1.6+ to 23.7+) | 5.7 (0.0+ to 24.2+) |
CI = confidence interval; cLDA = constrained longitudinal data analysis; DOR = duration of response; ECOG = Eastern Cooperative Oncology Group; EORTC QLQ-C30 GHS = European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30 Global Health Status; FAS = full analysis set; ITT = intention to treat; HR = hazard ratio; LSM = least squares mean; ORR = objective response rate; OS = overall survival; PEM + LEN = pembrolizumab plus lenvatinib combination therapy; PFS = progression-free survival; PRO = patient-reported outcome; SD = standard deviation; TPC = treatment of physician’s choice; vs. = versus.
aFrom the product-limit (Kaplan–Meier) method for censored data.
bBased on Cox regression model with the Efron method of tie handling with treatment as a covariate stratified by ECOG Performance Status, geographic region, and prior history of pelvic radiation.
cOne-sided P value based on log-rank test stratified by ECOG Performance Status, geographic region, and prior history of pelvic radiation. The P value has been adjusted for multiple testing.
dBased on a cLDA model with the PRO scores as the response variable with covariates for treatment by study visit interaction, MMR status, ECOG Performance Status, geographic region, and prior history of pelvic radiation. Note: For baseline and week 12, N is the number of patients in each treatment group with non-missing assessments at the specific time point; for change from baseline, N is the number of patients in the analysis population in each treatment group. The P value has not been adjusted for multiple testing.
eBased on the Miettinen and Nurminen method stratified by ECOG Performance Status, geographic region, and prior history of pelvic radiation.
fOne-sided P value for testing. H0 means the difference in percentage equals 0, whereas H1 means the difference in percentage is greater than 0. The P value has been adjusted for multiple testing.
gFor DOR, “+” indicates there is no progressive disease by the time of the last disease assessment.
Source: Clinical Study Report.19
The OS rates at 12 months were 61.6% (95% Cl, 56.1% to 66.6%) in the PEM + LEN arm and 49.5% (95% Cl, 43.8% to 55.0%) in the TPC arm, respectively. The OS rates over the time are presented in Table 44 and Table 45. OS is summarized by Kaplan-Meier plot in Figure 3.
The sponsor indicated that the IA1 analysis for OS met the success criteria for the hypothesis of the primary efficacy end point of OS; i.e., the combination of LEN + PEM was superior to TPC with respect to OS in patients with pMMR tumours. Therefore, IA2 and a final analysis were not required.17,18,60
OS Subgroup Analysis
Pre-specified subgroup analyses for OS are presented in Table 46 in Appendix 3.
For patients with an ECOG PS of 0 and an ECOG PS of 1, the HRs (PEM + LEN versus TPC) were 0.56 (95% CI, 0.42 to 0.75) and 0.87 (95% CI, 0.64 to 1.18), respectively. For patients with endometrioid cancer and non-endometrioid cancer, the HRs (PEM + LEN versus TPC) were 0.78 (95% CI, 0.57 to 1.05) and 0.56 (95% CI, 0.42 to 0.74), respectively. For patients with 1, 2, and greater than or equal to 3 prior lines of systemic therapy, the HRs (PEM + LEN versus TPC) were 0.61 (95% CI, 0.47 to 0.78), 0.88 (95% CI, 0.59 to 1.30), and 0.75 (95% CI, 0.24 to 2.37), respectively.
It should be noted that the prior lines of therapy (1, 2, and ≥ 3) used for the subgroup analysis in pMMR included prior lines of neoadjuvant and/or adjuvant systemic therapy (refer to Table 29).
OS Sensitivity Analysis
No sensitivity analysis for OS was done in this study.
Progression-Free Survival
At the preplanned IA1, a total of 485 events (247 [71.4%] in the PEM + LEN arm and 238 [67.8%] in the TPC arm) had occurred. The median PFS was longer in the PEM + LEN arm (6.6 months; 95% Cl, 5.6 to 7.4) than in the TPC arm (3.8 months; 95% Cl, 3.6 to 5.0). The HR for PEM + LEN versus TPC was 0.60 (95% Cl, 0.50 to 0.72; P < 0.0001) (Table 11 and Figure 4).
The PFS rates at 6 months were 53.5% (95% Cl, 48.4% to 58.3%) in the PEM + LEN arm and 34.4% (95% Cl, 29.2% to 39.4%) in the TPC arm, respectively. The PFS rates over time are presented in Table 47 and Table 48 in Appendix 3. PFS is summarized using a Kaplan-Meier plot in Figure 4.
Figure 4: Kaplan-Meier Estimates of Progression-Free Survival (Primary Censoring Rule) in Patients With pMMR Tumours (ITT Population)
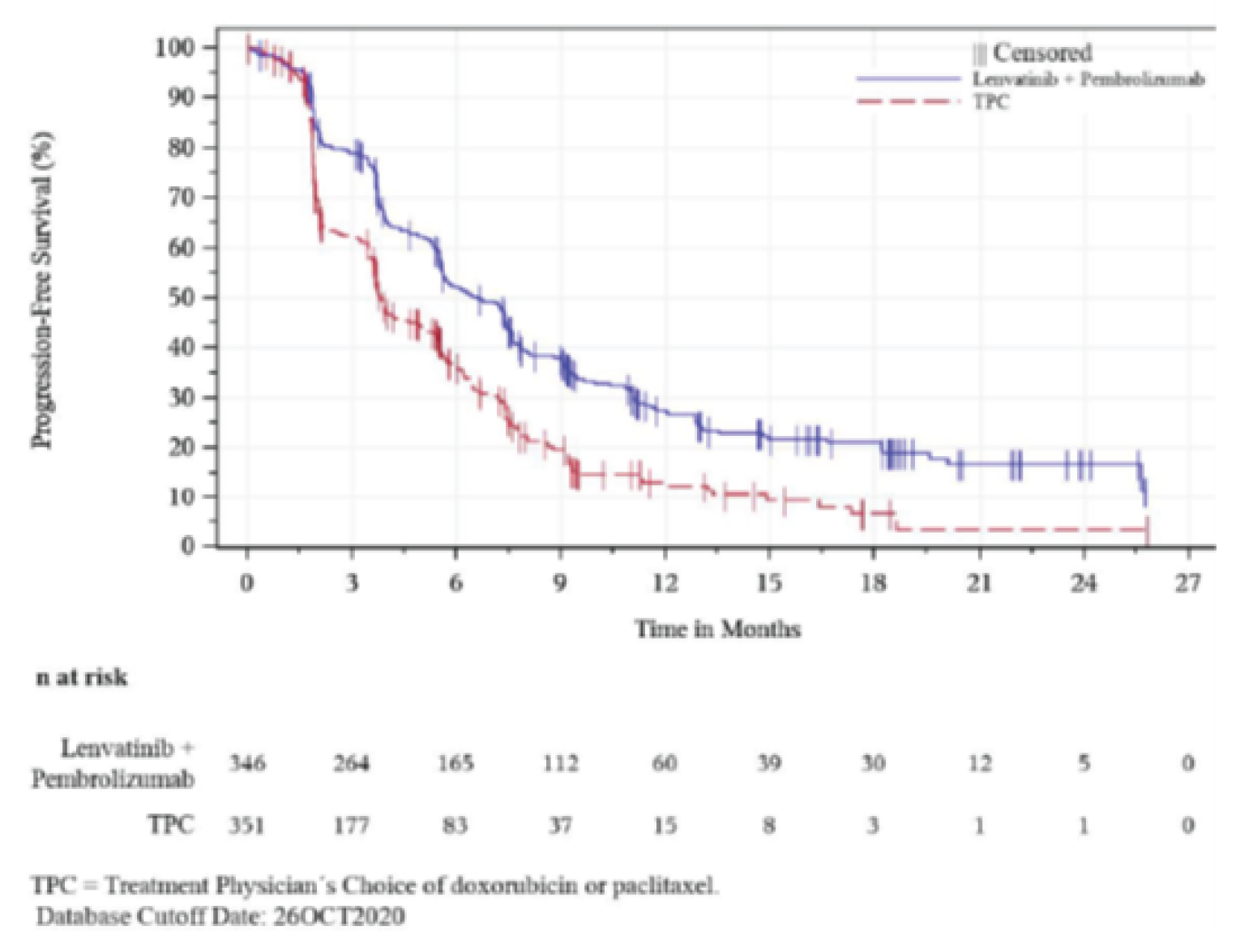
ITT = intention to treat; PEM + LEN = pembrolizumab plus lenvatinib combination therapy; PFS = progression-free survival; pMMR = mismatch repair proficient.
Source: Clinical Study Report.19
The sponsor indicated that the IA1 analysis for PFS met the success criteria for the hypothesis of the primary efficacy end point of PFS (i.e., the combination of LEN + PEM was superior to TPC with respect to PFS in both pMMR groups). Therefore, no IA2 or final analysis were required.17,18,60
PFS Subgroup Analysis
Pre-specified subgroup analyses for PFS are presented in Table 49 in Appendix 3. The HRs (PEM + LEN versus TPC) for patients with an ECOG PS of 0 and an ECOG PS of 1 were 0.57 (95% CI, 0.45 to 0.72) and 0.65 (95% CI, 0.49 to 0.86), respectively. The HRs (PEM + LEN versus TPC) for patients with endometrioid cancer and those with non-endometrioid cancer were 0.59 (95% CI, 0.46 to 0.76) and 0.56 (95% CI, 0.43, 0.73), respectively. The HRs (PEM + LEN versus TPC) for patients with 1, 2, and greater than or equal to 3 prior lines of systemic therapy were 0.52 (95% CI, 0.42 to 0.65), 0.74 (95% CI, 0.53 to 1.04), and 0.60 (95% CI, 0.12 to 3.07), respectively.
PFS Sensitivity Analysis
Two sensitivity analyses with different sets of censoring rules were performed to assess PFS in the pMMR population to support the robustness of the primary analysis for PFS. The first sensitivity analysis followed the ITT principle: that is, disease progressions and deaths were counted as events regardless of missed study visits or initiation of new anticancer therapy. The second sensitivity analysis considered the initiation of new anticancer treatments or the discontinuation of treatment for reasons other than CR (whichever occurred later) to be a progressive disease event for patients without documented progressive disease or death. If a participant met multiple criteria for censoring, the censoring criterion that occurred earliest was applied (refer to Table 9).
Both sensitivity analyses for PFS showed results that were consistent with the PFS primary analysis in the pMMR primary analysis population (Table 50 and Table 51 in Appendix 3).
Health-Related Quality of Life
The findings of patient-reported and HRQoL outcomes (i.e., the EORTC QLQ-C30 GHS, the EORTC QLQ-C30 Physical Functioning scale, the EORTC QLQ-EN24 Urological Symptoms Score, and the EQ-5D VAS score) are presented in Table 12. Among patients completing the HRQoL measures, patients in both arms appeared to experience deterioration in HRQoL when assessed at week 12 (Table 12). No obvious between-group differences in change from baseline were observed. These findings suggest that there appeared to be no apparent between-group differences (for PEM + LEN versus TPC) observed in terms of HRQoL during the treatment period at week 12 (refer to Table 52, Table 53, Figure 8, Figure 9, Figure 10, and Figure 11 in Appendix 3).
No subgroup or sensitivity analysis was done for HRQoL outcomes.
Symptom Reduction
Symptom severity reduction was not assessed as a separate outcome in the KEYNOTE-775 trial. However, urological symptoms were assessed using the EORTC QLQ-EN24 Urological Symptoms Score (Table 12).
Objective Response Rate
At IA1, the KEYNOTE-775 trial met the success criteria for the hypothesis of the secondary efficacy end point on ORR; i.e., the combination of LEN + PEM was superior to TPC with respect to ORR in patients with pMMR tumours. The confirmed ORR was higher in the PEM + LEN arm compared to the TPC arm (30.3% versus 15.1%, respectively). The between-group difference in ORR (PEM + LEN versus TPC) was 15.2% (95% Cl, 9.1% to 21.4%; P < 0.0001) (refer to Table 54 in Appendix 3).
ORR subgroup analysis results are presented in Table 55 in Appendix 3. The between-group differences in ORR (PEM + LEN versus TPC) were 14.7% (95% CI, 6.5% to 22.7%) and 16.1% (95% CI, 6.7 to 25.6) for patients with an ECOG PS of 0 and an ECOG PS of 1, respectively. The between-group difference in ORR (PEM + LEN versus TPC) was 11.6% (95% CI, 3.1% to 20.1%) versus 19.9% (95% CI, 11.1 to 228.7) in patients with endometrioid cancer and patients with non-endometrioid cancer, respectively. The between-group differences in ORR (PEM + LEN versus TPC) were 17.9% (95% CI, 10.5% to 25.27%), 10.9% (95% CI, –0.7 to 22.8), and 1.8% (95% CI, 377.7 to 38.2) for patients with 1, 2, and greater than or equal to 3 lines of systemic therapy, respectively (Table 55).
No sensitivity analysis was done for ORR.
Duration of Response
The median DOR was 9.2 months (range = 1.6 to 23.7) in the PEM + LEN arm and 5.7 months (range = 0.0 to 24.2) in the TPC arm (Table 11, Table 56, and Figure 12 in Appendix 3).
No subgroup or sensitivity analysis was done for DOR.
Table 12: Analysis of Change From Baseline to Week 12 in EORTC QLQs and EQ-5D VAS (pMMR, FAS)
Treatment | Baseline | Week 12 | CFB to week 12 | Difference of CFB (PEM + LEN vs. TPC)a | ||||
|---|---|---|---|---|---|---|---|---|
N (%) | Mean (SD) | N (%) | Mean (SD) | N (%) | LS mean (95% CI)a | LS mean CFB (95% CI) | ||
EORTC QLQ-C30 GHS | ||||||||
PEM + LEN | 316 (91) | 66.56 (21.44) | 256 (74) | 60.94 (21.35) | 327 (95) | –6.80 (–9.43 to –4.17) | 1.16 (–2.49 to 4.81) P = 0.5316 | |
TPC | 298 (85) | 66.64 (22.43) | 192 (55) | 62.80 (21.67) | 310 (88) | –7.96 (–10.86 to –5.05) | ||
EORTC QLQ-C30 Physical Functioning | ||||||||
PEM + LEN | 316 (91) | 79.56 (19.21) | 256 (74) | 71.46 (21.64) | 327 (95) | –10.42 (–12.65 to –8.19) | –1.74 (–4.99 to 1.51) P = 0.2931 | |
TPC | 298 (85) | 76.58 (20.85) | 192 (55) | 72.81 (21.13) | 310 (88) | –8.68 (–11.13 to –6.23) | ||
EORTC QLQ-EN24 Urological Symptoms Score | ||||||||
PEM + LEN | 268 (77) | 14.89 (18.09) | 217 (63) | 12.37 (18.29) | 282 (82) | –2.20 (–4.28 to –0.12) | –2.98 (–5.96 to –0.00) P = 0.0496 | |
TPC | 249 (71) | 16.13 (19.79) | 157 (45) | 16.56 (19.29) | 266 (76) | 0.78 (–1.55 to 3.11) | ||
EQ-5D VAS score | ||||||||
PEM + LEN | 319 (92) | 74.08 (18.33) | 256 (74) | 70.23 (18.63) | 327 (95) | –5.35 (–7.59 to –3.11) | 2.06 (–1.09 to 5.20) P = 0.1992 | |
TPC | 303 (86) | 74.13 (18.61) | 193 (55) | 70.90 (19.77) | 310 (88) | –7.41 (9.85 to –4.96) | ||
CFB = change from baseline; cLDA = constrained longitudinal data analysis; ECOG = Eastern Cooperative Oncology Group; EORTC QLQ-C30 GHS = European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30 Global Health Status; EORTC QLQ-EN24 = European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Endometrial Cancer Module; FAS = full analysis set; LS = least squares; PEM + LEN = pembrolizumab plus lenvatinib combination therapy; pMMR = mismatch repair proficient; PRO = patient-reported outcome; SD = standard deviation; TPC = treatment of physician’s choice; vs. = versus.
Note: For baseline and week 12, N is the number of patients in each treatment group with non-missing assessments at the specific time point; for change from baseline, N is the number of patients in the analysis population in each treatment group with at least 1 measurement.
aBased on a cLDA model with PRO scores as the response variable with covariates for treatment by study visit interaction, MMR status, ECOG Performance Status, geographic region, and prior history of pelvic radiation.
Source: Clinical Study Report.19
Harms
Only those harms identified in the review protocol are reported. Refer to Table 13 for detailed harms data.
Adverse Events
A total of 99.7% of patients in both the PEM + LEN arm and TPC arm experienced at least 1 AE (Table 13).
The most common AEs (≥ 40% in either of arm) were hypertension (65.5% for PEM + LEN versus 5.2% for TPC), hypothyroidism (55.3% for PEM + LEN versus 0.9% for TPC), diarrhea (54.7% for PEM + LEN versus 19.7% for TPC), nausea (49.4% for PEM + LEN versus 47.4% for TPC), decreased appetite (4,428% for PEM + LEN versus 20.6% for TPC), and anemia (24.3% for PEM + LEN versus 48.9% for TPC) (refer to Table 57 in Appendix 3). The exposure-adjusted AE rates per 100 person-months are presented in Table 58 in Appendix 3.
Serious Adverse Events
A total of 49.7% of patients in PEM + LEN arm and 28.9% of patients in the TPC arm experienced an SAE (Table 13). The most common SAEs (≥ 2% in either of the arms) were hypertension (4.4% for PEM + LEN versus 0% for TPC), urinary tract infection (3.2% for PEM + LEN versus 0.6% for TPC), acute kidney injury (2.3% for PEM + LEN versus 0.9% for TPC), colitis (2.0% for PEM + LEN versus 0.3% for TPC), pyrexia (2.0% for PEM + LEN versus 0.9% for TPC), decreased appetite (2.0% for PEM + LEN versus 0% for TPC), febrile neutropenia (0.6% for PEM + LEN versus 4.0% for TPC), and neutropenia (0.3% for PEM + LEN versus 2.2% for TPC) (Table 59 in Appendix 3).
Mortality
At the time of the pre-specified IA1 a total of 4.7% of patients in the PEM + LEN arm and 4.6% of patients in the TPC arm had died due to AEs (refer to Table 13 in this section and Table 60 in Appendix 3). The most common AEs (i.e., affecting system organ classes) resulting in death were cardiac disorders (0.6% for PEM + LEN versus 0.9% for TPC), gastrointestinal disorders (1.2% for PEM + LEN versus 0% for TPC), general disorders and administration site conditions (0.9% for PEM + LEN versus 1.2% for TPC), and infections and infestations (0.6% for PEM + LEN versus 1.5% for TPC) (refer to Table 60 in Appendix 3).
Withdrawals Due to Adverse Events
All-cause AEs leading to study drug discontinuation occurred in 31.0% of patients in the PEM + LEN arm and in 8.3% of patients in the TPC arm (Table 13). Among patients in the PEN+LEN arm, 17.5%, 18.4%, and 12.6.0% discontinued PEM, LEN, or both due to AEs, respectively. Although 8.3% of patients in the TPC arm discontinued treatment due to AEs, no specific information was reported for the number of patients who discontinued from doxorubicin versus paclitaxel.
Notable Harms
Hyperthyroidism occurred in 10.8% of patients treated with PEM + LEN and in 1.2% patients treated with TPC. The following notable harms were reported in the study (Table 13): hypothyroidism (55.3% for PEM + LEN versus 0.9% for TPC), pneumonitis (1.2% for PEM + LEN versus 0.3% for TPC), colitis (5.3% for PEM + LEN versus 0.3% for TPC), adrenal insufficiency (0.9% for PEM + LEN versus 0% for TPC), hepatitis (1.8% for PEM + LEN versus 0% for TPC), hypophysitis (0.6% for PEM + LEN versus 0% for TPC), nephritis (0.6% for PEM + LEN versus 0% for TPC), type 1 diabetes mellitus (0.9% for PEM + LEN versus 0% for TPC), and hypertension (65.5 for PEM + LEN versus 5.2% for TPC).
Table 13: Summary of Harms in the KEYNOTE-775 Trial
Adverse events | PEM + LEN (N = 342) | TPC (N = 325) |
|---|---|---|
Patients with at least 1 TEAE, n (%) | 341 (99.7) | 324 (99.7) |
Patients with at least 1 SAE, n (%) | 170 (49.7) | 94 (28.9) |
Patients with dose modificationa due to an AE | 316 (92.4) | 137 (42.2) |
Patients with dose interruptionb due to an AE | 235 (68.7) | 91 (28.0) |
Interruption of PEM | 165 (48.2) | 0 (0.0) |
Interruption of LEN | 199 (58.2) | 0 (0.0) |
Interruption of both PEM and LEN | 100 (29.2) | 0 (0.0) |
Patients with dose reductionc due to an adverse event | 229 (67.0) | 42 (12.9) |
Patients with a AEs leading to DC from the treatment, n (%) d | 106 (31.0) | 27 (8.3) |
Discontinued PEM | 60 (17.5) | 0 (0.0) |
Discontinued LEN | 97 (28.4) | 0 (0.0) |
Discontinued both PEM and LEN | 43 (12.6) | 0 (0.0) |
Deaths due to AEs e | 16 (4.7) | 15 (4.6) |
Notable harms, n (%) | ||
Adrenal insufficiency | 3 (0.9) | 0 (0.0) |
Colitis | 18 (5.3) | 1 (0.3) |
Hepatitis | 6 (1.8) | 0 (0.0) |
Hyperthyroidism | 37 (10.8) | 4 (1.2) |
Hypophysitis | 2 (0.6) | 0 (0.0) |
Hypothyroidism | 189 (55.3) | 3 (0.9) |
Nephritis | 2 (0.6) | 0 (0.0) |
Pneumonitis | 4 (1.2) | 1 (0.3) |
Type 1 diabetes mellitus | 3 (0.9) | 0 (0.0) |
Hypertension | 224 (65.5) | 17 (5.2) |
AE = adverse event; DC = discontinuation; PEM + LEN = pembrolizumab plus lenvatinib combination therapy; pMMR = mismatch repair proficient; SAE = serious adverse event; TEAE = treatment-emergent adverse event; TPC = treatment of physician’s choice (doxorubicin or paclitaxel); WDAE = withdrawal due to adverse event.
Note: Non-serious AEs were followed for up to 30 days after the last dose. SAEs were followed for up to 90 days after the last dose.
aDefined as an action taken to reduce the dose or interrupt or withdraw the drug.
bFor PEM + LEN, the dose interruption of either pembrolizumab or lenvatinib.
cFor PEM + LEN, the dose reduction for only lenvatinib.
dFor PEM + LEN, the discontinuation of either pembrolizumab or lenvatinib.
eDeath due to AE.
Source: Clinical Study Report.19
Critical Appraisal
Internal Validity
The included pivotal study (KEYNOTE-775) was a relatively well-designed, prospective, multi-centre, open-label, randomized, parallel, active-controlled trial. The study used an appropriate randomization and allocation method (i.e., an interactive response technology system). It should be noted that the pMMR population was not considered a subgroup in the original study design. The sample size and power calculations were based on pMMR populations.19
Overall, most of the demographic and baseline characteristics were well balanced between arms (Table 7). The key prognostic factors, such as ECOG PS (0 or 1), histology of tumour type (e.g., endometrioid, non-endometrioid), and prior history of pelvic radiation, were well balanced between arms. Minor imbalances between the 2 study arms were observed in the proportion of patients with number of prior systemic therapies, type of prior therapy (such as neoadjuvant or adjuvant), and history of prior hysterectomy (Table 29 in Appendix 3). However, the clinical experts consulted for this review stated that these minor imbalances would have been unlikely to affect the comparative study results between the PEM + LEN and TPC groups.
A total of 4 patients (1.2%) in the PEM + LEN arm and 26 patients (7.4%) in the TPC arm did not receive their assigned treatment. The relatively small percentage of patients who did not received the treatment medication is unlikely to have had a significant impact on the comparative assessment of the efficacy of PFS and OS in the ITT analysis.
A total of 229 patients (67%) experienced a dose reduction of LEN in the PEM + LEN arm and a total of 42 patients (12.9%) experienced a dose reduction in the TPC group (either doxorubicin or paclitaxel) due to AEs (Table 13). The proposed dose regimen of LEN was 20 mg. The actual median dose of LEN was 13.8 mg, ranging from 3 mg to 20 mg in the pMMR population (Table 41); however, detailed dose reduction information was not reported for the pMMR population. The clinical experts consulted for this review indicated that the dose modifications (reductions or interruptions) of LEN, doxorubicin, or paclitaxel reflects clinical practice in Canada; therefore, it was not expected to have an impact on the comparative clinical efficacy of PEM + LEN versus the TPC.
Based on the study design, during the treatment phase, patients were prohibited from receiving concurrent anticancer therapies, such as chemotherapy or targeted therapies (e.g., tyrosine kinase inhibitors). ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. Based on the concomitant medication information reported, while the reasons for prohibiting these additional concomitant anticancer drugs were not clearly described in the Clinical Study Report, the clinical experts consulted for this review considered that the potential impact of this restriction on the comparative efficacy assessment (of PEM + LEN versus TPC) would be negligible because of the very small numbers of patients using them (e.g., carboplatin, cisplatin, doxorubicin, paclitaxel, LEN, and PEM).
KEYNOTE-775 was an open-label trial. As a result, the study investigators and patients were aware of their treatment status, which increases the risk of detection and performance biases that have the potential to influence outcome reporting. However, OS, PFS, ORR, and DOR are all objective response measurements. These were assessed using the Response Evaluation Criteria in Solid Tumours (version 1.1) through independent central radiologic review. The open-label design is less of a concern for objective responses such as OS, PFS, ORR, and DOR; however, it remains a concern for subjective end points, such as safety and HRQoL. The direction of anticipated bias related to these outcomes is unclear.
The primary outcomes were PFS and OS. PFS was defined as the time between the randomization date and the first date of disease progression (assessed by imaging) or death. Therefore, the efficacy measurement was objective and not likely biased by the subsequent systemic therapy. However, subsequent systemic anticancer treatment (e.g., chemotherapy or PD-1 or PD-L1 inhibitors) may contribute to patients’ OS benefit in both arms. The impact of a subsequent systemic anticancer treatment on comparative OS may introduce potential bias. However, more patients in the TPC group than in PEN + LEN group received subsequent systemic anticancer treatments (Table 31), which would make the between-group differences less pronounced; the direction of bias would likely go against PEM + LEN treatment. Furthermore, the reported OS results are considered final based on IA1, according to pre-specified stopping criteria. A limitation of this study is the relatively short duration of follow-up, which may mean that survival data (e.g., OS) are evolving. Therefore, whether more mature OS efficacy results would be consistent with the interim results is unknown.
EORTC QLQ-C30 GHS scores were assessed as a secondary outcome but not controlled for type I error. The other patient-reported and HRQoL outcomes (the EORTC QLQ-C30 Physical Functioning scale, EORTC QLQ-EN24 Urological Symptoms Score, and EQ-5D VAS score) were assessed as exploratory outcomes using a cLDA model. The cLDA model is most appropriate when data are missing at random or missing completely at random. It is expected to be biased when the missing data are not missing at random, which is likely the case with these data. There is a potential risk of bias because a large number of patients did not have complete measures (there are substantial missing data on these outcomes) as well as due to the open-label nature of the design. How the missing data were handled or imputed was not clearly described in the Clinical Study Report. As well, there may have been differential recall bias. Overall, the magnitude and direction of the impact of these missing data and recall bias on the patient-reported and HRQoL outcomes is unknown. No minimal important difference (MID) was identified for HRQoL measures in the population. Overall, the findings for HRQoL should be viewed as supportive evidence only.
In subgroups that were not part of the randomization scheme, imbalances in characteristics may bias the results observed between the groups.
Symptom reduction was identified an outcome in the CADTH review protocol. Symptom severity was not assessed as a separate outcome in KEYNOTE-775. However, the severity of urological symptoms was assessed using the EORTC QLQ-EN24 Urological Symptoms Score (Table 12).
DOR was assessed as an exploratory outcome. No formal statistical analysis was performed.
Finally, the primary analyses of PFS, OS, and ORR were based on the ITT analysis. In the pMMR population, important protocol deviations were reported for 17 patients: 9 patients (2.7%) in the LEN + PEM arm and 8 patients (2.3%) in the TPC arm. There was no protocol deviation information reported for the pMMR population. No per-protocol analysis was conducted to assess whether the per-protocol analysis was consistent with the ITT analysis. Therefore, it is unknown if the protocol deviation may have had an impact on the findings. However, given that the number of patients with important deviations was relatively low and balanced across both groups, its impact on the comparative efficacy findings (PEM + LEN versus TPC) are expected to be negligible.
External Validity
This study was a multinational, multi-centre trial, with 67 sites in 21 countries. A total of 58 patients living in Canada participated at 11 sites across Canada. According to the clinical experts consulted for this review, the KEYNOTE-775 trial population (i.e., adult patients with advanced pMMR endometrial carcinoma who have disease progression following prior platinum-based systemic therapy in any setting and are not candidates for curative surgery or radiation) is considered reflective of the requested target population. There is no concern about the generalization of findings from the pivotal study to Canadian clinical settings.
The following considerations are of importance regarding the external validity of KEYNOTE-775 trial.
It is uncertain whether the findings can be generalized to patients with an ECOG PS of 2 or greater because no such patients were included in the study. The clinical experts believe that the PEM + LEN combination treatment could be extended to patients with an ECOG PS of 2 who are appropriately informed and motivated. These drugs should likely not be used in patients with an ECOG PS of 3 or 4.
Patients with carcinosarcoma were excluded from the trial. The clinical experts indicated that the evidence to date is in carcinomas; as a result, they are not aware of benefit in pure sarcomas. However, given that carcinosarcomas are a combination, theoretically, PEM + LEN combination therapy could be extended to patients with carcinosarcomas. Nevertheless, it was a reasonable exclusion criterion in the clinical trial, and there is no supporting research evidence at the moment.
The Health Canada–recommended dose of LEN in the PEM + LEN combination treatment is 20 mg orally once daily until unacceptable toxicity or disease progression. However, the median dosage of LEN reported in the trial was 13.8 mg (range = 3 mg to 20 mg). The clinical experts consulted for this review indicated that dose modification is very common in clinical practice. Therefore, there is no concern about the dose reduction of LEN in the routine clinical setting. In the TPC group, 239 patients out of 351 patients (68.1%) received doxorubicin, and 86 patients (24.51%) patients received paclitaxel (Table 35). The clinical experts indicated that the choice would vary based on clinicians’ preference. It is likely irrelevant to the efficacy assessment because there does not seem to be scientific evidence about efficacy differences between doxorubicin and paclitaxel. The clinical experts indicated that the dose of paclitaxel used in the trial is aligned with the clinical standard dosage for this treatment; that is, 60 mg or 80 mg administered through IV once a week, with a cycle of 3 weeks on and 1 week off.
In the KEYNOTE-775 trial, the only dosage of PEM was 200 mg every 3 weeks. The clinical experts indicated that clinically, physicians and patients may prefer dosing 400 mg every 6 weeks. Either dose can be continued in combination with LEN for up to 24 months (i.e., 18 doses of 400 mg or 35 doses of 200 mg) or until unacceptable toxicity disease progression, whichever is longer.
Indirect Evidence
No ITC was identified in this review.
Other Relevant Evidence
This section includes an additional relevant study, KEYNOTE-146,61 that was included in the sponsor’s submission to CADTH and was considered to provide additional longer-term evidence for this review.
KEYNOTE-14661,62 is an ongoing, multinational, open-label, single-arm, phase Ib and II study of PEM + LEN in patients with selected solid tumours, including endometrial carcinomas. This section focuses on only the cohort of patients with advanced non-MSI-H or pMMR endometrial cancer.
Table 14: Details of the KEYNOTE-146 Trial
Detail | KEYNOTE-146a |
|---|---|
Designs and populations | |
Study design | Non-randomized, multi-centre, single-arm, open-label, ongoing phase Ib and II trial |
Locationsb | Conducted in US and non-US countries (e.g., Spain) that enrolled 94 patients with advanced non- MSI-H and/or pMMR endometrial carcinoma |
Patient enrolment dates | The first patient’s first visit was on July 22, 2015 to provide a signature indicating informed consent. The study is ongoing. The data cut-off date for the current analysis was January 10, 2019. |
Number of patients (N)a | 94 |
Inclusion criteriac | Key inclusion criteria (for all cohorts):
Patients in the non-MSI-H or pMMR cohort were required to have:a
|
Exclusion criteriac |
|
Drugs | |
Intervention | Lenvatinib 20 mg once daily orally and PEM 200 mg IV once every 3 weeks in 3-week cycles (to a maximum of 35 PEM treatments). |
Comparator(s) | None |
Duration | |
Phase | |
Screening | 25 days |
Baseline period | 3 days The purpose of the baseline period was to confirm protocol eligibility as specified by the inclusion and exclusion criteria. |
Treatment | Eight cycles of treatment (3 weeks each) or after early discontinuation of study drug. Patients who completed 8 cycles of the study drug continued to receive it during the treatment period of the extension phase. Treatment period of the extension phase: Patients who were still receiving treatment at the end of the treatment period in phase Ib or II of the trial continued to receive the same study drug during the treatment period of the extension phase. Patients could receive the study drug until progressive disease, development of unacceptable toxicity, withdrawal of consent, or termination of the study by the sponsor. Patients could receive up to 35 treatments (over approximately 2 years) with PEM. Lenvatinib treatment could be continued as monotherapy thereafter. At the time of data cut-off (January 10, 2019), the median duration of treatment with PEM + LEN was 7.38 months (range = 0.03 months to 37.78 months) in the non-MSI-H pMMR cohort (N = 94). |
Extension | The extension phase consisted of 2 periods:
|
Follow-up | The follow-up period consisted of an off-treatment visit and follow-up visits. The off-treatment visit occurred within 30 days after the last dose of the study drug. After the off-treatment visit, patients entered the follow-up period of the extension phase unless they had withdrawn their consent to continue in the study. During the follow-up period, patients were treated by the investigator according to the prevailing local standard of care. Patients were followed every 12 weeks (± 1 week) for survival. Tumour assessments were performed as detailed in the Schedules of Procedures/Assessments for phase Ib and phase II, and subsequent anticancer treatments were recorded. Follow-up visits continued as long as study patients were alive unless they withdrew consent, or until the sponsor terminated the study. Overall median follow-up for OS = 18.7 months (95% CI, 14.1 months to 20.9 months) |
Outcomes | |
Primary end point | ORR at week 24 based on investigator assessment using irRECIST |
Secondary and exploratory end points | Secondary
|
Notes | |
Publications | Makker (2020)63 Makker (2021)64 |
CI = confidence interval; ECOG PS = Eastern Cooperative Oncology Group Performance Status; DOR = duration of response; irRECIST = immune-related Response Evaluation Criteria in Solid Tumours; MSI-H = microsatellite instability-high; ORR = objective response rate; OS = overall survival; PFS = progression-free survival; PD-1 = programmed cell death 1; PD-L1 = programmed cell death ligand 1; PD-L2 = programmed cell death ligand 2; PEM = pembrolizumab; PEM + LEN = pembrolizumab plus lenvatinib combination therapy; pMMR = mismatch repair proficient.
Note: The sponsor provided an updated analysis for pMMR (cut-off date = August 18, 2020; median follow-up duration = 35.8 months; 95% CI, 31.2 to 41.2). The sponsor indicated that the findings of the updated analysis were largely aligned with those reported in the analysis on January 10, 2019. (Refer to Appendix 5.)
aThe extracted information focused on patients with endometrial cancer with pMMR or non-MSI-H status in the KEYNOTE-146 trial only where available. Overall, KEYNOTE-146 included 283 patients. Among the 283 patients, 159 patients had non-endometrial carcinoma. A total of 124 patients had endometrial carcinoma. Among the 124 patients with endometrial carcinoma, 16 patients had no prior anticancer treatment; therefore, they did not meet the inclusion criteria for this review. One hundred and eight patients were previously treated with at least 1 systemic anticancer therapy. Among the 108 patients, 94 were non-MSI-H or pMMR, 11 were MSI-H or dMMR, and 3 had unknown MSI and/or MMR status. Therefore, in this report, only information about patients with non-MSI-H or pMMR tumours (N = 94) was extracted, unless otherwise specified. That is, the KEYNOTE-146 trial indicates the endometrial patients with pMMR/non-MSI-H status (N = 94) included unless otherwise specified. Patients with pMMR endometrial cancer are those with histologically confirmed endometrial carcinoma who were previously treated with at least 1 systemic anticancer therapy and had sufficient follow-up to provide a median follow-up of at least 12 months. In addition, for all responders, there was an opportunity for follow-up after initial objective response (as assessed by the investigator) of at least 6 months.
bPatients with endometrial cancer were enrolled across 20 sites, including 15 sites in the US (N = 101 patients) and 5 sites in Spain (N = 23 patients).61
cOverall inclusion and exclusion criteria for all cohorts. No inclusion or exclusion criteria specifically for patients with pMMR endometrial carcinoma were provided in the Clinical Study Report.
Source: Clinical Study Report.61
Methods
Inclusion and exclusion criteria for the KEYNOTE-146 trial are summarized in Table 14.
Briefly, patients were deemed eligible if they met the following criteria: greater than or equal to 18 years old; histologically and/or cytologically confirmed, advanced pMMR endometrial carcinoma, with up to 2 prior lines of systemic therapy (unless discussed with the sponsor). Patients must have had progressive disease after previous treatment. They also had to have measurable disease (at least 1 lesion of ≥ 10 mm in the longest diameter for a non-lymph node or ≥ 15 mm in the short-axis diameter for a lymph node), an ECOG PS of 0 or 1, and life expectancy of greater than or equal to 12 weeks.
Eligible patients (N = 94) were included to receive PEM + LEN combination treatment, consisting of PEM 200 mg IV once every 3 weeks (to a maximum of 35 PEM treatments) and LEN 20 mg once daily orally.
The primary efficacy measure was ORR at week 24. Key secondary measures were ORR at the data cut-off date, DOR, PFS, and OS.
Populations
Detailed patient baseline disease and demographic characteristics for the KEYNOTE-146 trial are summarized in Table 15 and Table 16. The median age was 66.0 years (range = 40 years to 80 years). Patients had a baseline ECOG PS of either 0 (52.1%) or 1 (47.9%). A total of 81 patients (86.2%) were White; 6 patients (6.4%) were Black or African American. All patients (N = 94, 100%) had received a prior platinum therapy regimen. A total of 48 patients (51.1%) had received 1 line of prior therapy; 36 patients (38.32%) had received 2 lines of prior therapies. Ten patients (10.6%) had received 3 or more lines of prior therapies. The most common histologic subtypes of disease were endometrioid adenocarcinoma (N = 46, 48.9%) and serous adenocarcinoma (N = 33, 35.1%).
Table 15: Participant Characteristics in Patients With pMMR Tumours (FAS Population, KEYNOTE-146 Trial)
Characteristic | PEM + LEN (N = 94) |
|---|---|
Total number of patients, N | 94 |
Sex | |
Female, n (%) | 94 (100.0) |
Age (years), n (%) | |
< 65 years | 36 (38.3) |
≥ 65 years | 58 (61.7) |
Median (range) | 66 (40 to 80) |
Race, n (%) | |
Black or African American | 6 (6.4) |
White | 81 (86.2) |
Other | 7 (7.4) |
Missing | 0 |
Region,a n (%) | |
US | 81 (86.2) |
Non-US | 13 (13.8) |
ECOG PS, n (%) | |
0 | 49 (52.1) |
1 | 45 (47.9) |
Time since original diagnosis (month) | |
Median (range) | 23.5 (3.88 to 250.84) |
Time since last disease progression to first dose (month) | |
Median (range) | 1.5 (0.16 to 15.8) |
Histology, n (%) | |
Endometrioid carcinoma | 46 (48.9) |
Non-endometrioid adenocarcinoma | 48 (51.1) |
Serous adenocarcinoma | 33 (35.1) |
Clear-cell adenocarcinoma | 5 (5.3) |
Adenocarcinoma, not otherwise specified | 1 (1.1) |
Other | 9 (9.6) |
FIGO stage at initial diagnosis, n (%) | |
I | 31 (33.0) |
II | 5 (5.3) |
III | 21 (22.3) |
IV | 33 (35.1) |
Not assigned | 4 (4.3) |
Patients with metastasis (sites of metastasis by independent image review), n (%) | |
Brain | 2 (2.1) |
Bone | 8 (8.5) |
Liver | 27 (28.7) |
Lung | 47 (50.0) |
Peritoneum | 40 (42.6) |
Lymph node | 46 (48.9) |
Pelvis | 8 (8.5) |
Other | 29 (30.9) |
ECOG PS = Eastern Cooperative Oncology Group Performance Status; FAS = full analysis set; FIGO = International Federation of Gynecology and Obstetrics; PEM + LEN = pembrolizumab plus lenvatinib combination therapy; pMMR = mismatch repair proficient.
Source: Clinical Study Report.61
Table 16: Prior Medications and Therapeutic Setting Treatment (pMMR, KEYNOTE-146 Trial)
Characteristic | PEM + LEN (N = 94) |
|---|---|
Number of previous medication regimens, n (%) | |
1 | 48 (51.1) |
2 | 36 (38.3) |
≥ 3 | 10 (10.6) |
Prior platinum therapy regimen | 94 (100.0) |
Prior anthracycline therapy regimen | 16 (17.0) |
Duration of most recent medication (months) | |
Median (range) | 4.14 (0.03 to 42.05) |
Therapeutic setting, n (%)a | |
Adjuvant | 68 (72.3) |
Neoadjuvant | 8 (8.5) |
Metastatic | 34 (36.2) |
Locally advanced | 0 |
Unknown | 2 (2.1) |
PEM + LEN = pembrolizumab plus lenvatinib combination therapy; pMMR = mismatch repair proficient.
aPatients may be counted in multiple categories.
Source: Clinical Study Report.61
Interventions
Patients received PEM + LEN combination therapy, consisting of LEN 20 mg once daily orally and PEM 200 mg IV once every 3 weeks in 3-week cycles (to a maximum of 35 PEM treatments).
PEM dose reductions were not permitted, per protocol; PEM dose interruptions due to TEAEs were allowed.
LEN reductions and/or interruptions due to TEAEs were allowed.
Concomitant Medications
One patient (1.1%) received antineoplastic and immunomodulating drugs (i.e., methotrexate). A total of 28 patients (29.8%) received corticosteroids for systemic use (Table 61, Appendix 3).
Subsequent Cancer Therapy
Subsequent anticancer treatments were recorded during the follow-up phase.61
Outcomes
The primary efficacy measure was ORR at week 24. Key secondary measures were ORR at the data cut-off date (January 10, 2019), PFS, OS, and DOR.
Statistical Analysis
There was no statistical analysis information for the non-MSI-H or pMMR cohort.
The efficacy analysis was based on the FAS and included all patients who entered the study treatment period. The safety outcomes analysis included all patients who received any PEM + LEN treatment (i.e., PEM 200 mg plus LEN 20 mg).
Power Calculation
No power calculation was performed. It was indicated that the initial size of the cohort with endometrial cancer could be expanded to approximately 120 evaluable patients. Expansion decisions were based on the results of 2 separate interim analyses that were to occur when 21 patients and 60 patients, respectively, had sufficient follow-up to be evaluated for response. At the first analysis, if there were more than 3 responses, then approximately 40 additional patients could be enrolled. If there were more than 12 responses at the second analysis, then approximately 60 additional patients could be enrolled.
Primary Outcome: ORR at Week 24
The rate (%) and 95% CI for ORR were presented.
Secondary Outcomes: ORR at the Cut-Off Date, DOR, PFS, and OS
The median (95% CI) PFS and OS were estimated. The median (95% CI) DOR was plotted using the Kaplan-Meier method. Event rates over time were also estimated.
Subgroup and Sensitivity Analyses
A subgroup analysis was conducted for ORR. The subgroup analysis of interest defined in the review protocol was based on histologic subtype. No subgroup analyses were conducted for OS and PFS.
No sensitivity analyses were conducted for any outcomes.
Patient Disposition
A total of 94 patients started the PEM + LEN combination therapy. Among them, 47 were still ongoing. Forty-seven patients (50%) discontinued from the trial. The main reason for discontinuation was death (N = 44, 46.8%). (Refer to Table 17.)
Table 17: Disposition of Patients With pMMR Tumours (FAS, KEYNOTE-146 Trial)
Disposition | PEM + LEN (N = 94) |
|---|---|
Status in study, n (%) | |
Started | 94 |
Discontinued (off study) | 47 (50%) |
Death | 44 (46.8) |
Withdrawal of consent | 3 (3.2) |
Patients ongoing at data cut-off date | 47 (50) |
FAS,a n (%) | 94 (100%) |
PP, n (%) | Not performed |
Safety set, n (%) | 94 (100%) |
FAS = full analysis set; ITT = intention to treat; PEM + LEN = pembrolizumab plus lenvatinib combination therapy; pMMR = mismatch repair proficient; PP = per protocol.
Note: The FAS was the same as the ITT analysis set in this study. It included all patients who entered the study treatment period. The efficacy analyses were based primarily on the full analysis set.
Source: Clinical Study Report.61
Exposure to Study Treatments
At the data cut-off date (January 10, 2019), the median duration of treatment with PEM + LEN was 7.38 months (range = 0.03 months to 37.78 months) in the non-MSI-H or pMMR cohort (N = 94). The overall treatment duration was defined as the duration between the earliest first-dose start date of either medication and the latest end date of either medication. The duration of the treatment was 12 months or longer in 16 patients (17%), 18 months or longer in 8 patients (8.7%), and 24 months or longer in 5 patients (5.3%). A total of 65 patients (69.1%) experienced a LEN dose reduction. A total of 67 patients (71.3%) experienced a LEN dose interruption. The median number of PEM doses in the pMMR cohort was 9.5 cycles (range = 1 cycle to 35 cycles).
Overall, the median follow-up time for OS was 18.7 months (95% CI, 14.1 months to 20.9 months).
Efficacy
Overall Survival
The results for OS are summarized in Table 18 and Figure 5. There were 44 deaths (46.8%), and the median OS was 16.4 months (95% CI, 13.5 to 25.9). The survival probabilities of patients at 12 months, 18 months, and 24 months were 69.5% (95% CI, 58.6 to 78.1%), 43.8% (95% CI, 31.2, 55.7%), and 39.2% (95% CI, 26.7 to 51.5%), respectively.
Table 18: Summary of Overall Survival in the pMMR Population (FAS, KEYNOTE-146 Trial)
OS | PEM + LEN (N = 94) |
|---|---|
Deaths, n (%) | 44 (46.8) |
Censored patients, n (%) | 50 (53.2) |
Patients censored before data cut-off, n (%) | 3 (3.2) |
Lost to follow-up | 0 (0.0) |
Withdrawal of consent | 3 (3.2) |
Study terminated by sponsor | 0 (0.0) |
Other | 0 (0.0) |
Patients censored at data cut-off, n (%) | 47 (50.0) |
OS (months)a | |
Median (95% CI) | 16.4 (13.5 to 25.9) |
OS rate (95% CI)b | |
At 12 months | 69.5 (58.6 to 78.1) |
At 18 months | 43.8 (31.2 to 55.7) |
At 24 months | 39.2 (26.7 to 51.5) |
Follow-up time for OS (months)a | |
Median (95% CI) | 18.7 (14.1 to 20.9) |
CI = confidence interval; FAS = full analysis set; OS = overall survival; PEM + LEN = pembrolizumab plus lenvatinib combination therapy; pMMR = mismatch repair proficient.
aThe median was estimated using Kaplan-Meier methodology; 95% CIs were constructed with a generalized Brookmeyer and Crowley method.
bPoint estimates for OS were based on Kaplan-Meier methodology; 95% CIs were based on the Greenwood formula using log-log transformation.
Source: Clinical Study Report.61
Figure 5: Kaplan-Meier Plot of OS (pMMR, KEYNOTE-146 Trial, FAS)
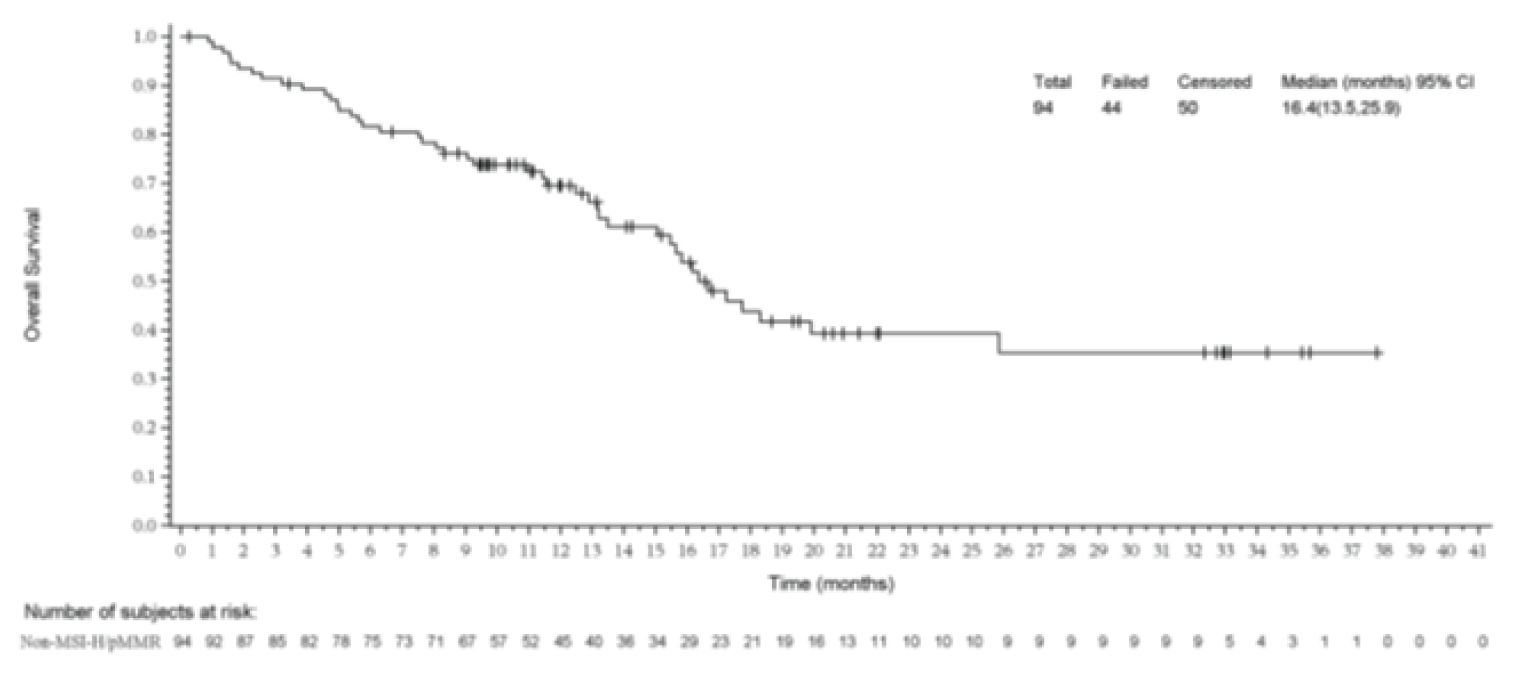
CI = confidence interval; MSI-H = microsatellite instability-high; pMMR = mismatch repair proficient.
Note: The median was estimated using the Kaplan-Meier method; the 95% CI was constructed with a generalized Brookmeyer and Crowley method.
Source: Clinical Study Report.61
Progression-Free Survival
The results for PFS per independent imaging review (IIR) are summarized in Table 19 and Figure 6. At the data cut-off date, 58 patients (61.7%) had experienced a PFS event. The median PFS was 5.4 months (95% CI, 4.4 months to 7.6 months). PFS rates at 6 months, 12 months, and 18 months were 49.4%, 33.2%, and 33.2.0%, respectively.
Table 19: Summary of Progression-Free Survival by IIR in pMMR (FAS, KEYNOTE-146 Trial)
PFS | PEM + LEN (N = 94) |
|---|---|
Patients with events, n (%) | 58 (61.7) |
Progressive disease | 49 (52.1) |
Deaths, n (%) | 9 (9.6) |
Censored patients, n (%) | 36 (38.3) |
No post-baseline tumour assessment | 1 (1.1) |
Death or progression after more than one missing assessment | 4 (4.3) |
New anticancer treatment starteda | 7 (7.4) |
No progression and no death at the time of data cut-off | 24 (25.5) |
PFS (months)b | |
Median (95% CI) | 5.4 (4.4 to 7.6) |
PFS rate (95% CI)c | |
At 3 months | 71.0 (60.3 to 79.2) |
At 6 months | 49.4 (38.4 to 59.4) |
At 9 months | 34.9 (24.8 to 45.3) |
At 12 months | 33.2 (23.1 to 43.6) |
At 18 months | 27.2 (16.6 to 38.9) |
At 24 months | NR |
Follow-up time for PFS (months)b | |
Median (95% CI) | 9.9 (9.6 to 15.9) |
CI = confidence interval; FAS = full analysis set; IIR = independent imaging review; mTOR = mammalian target of rapamycin; NR = not reported; PEM + LEN = pembrolizumab plus lenvatinib combination therapy; PFS = progression-free survival; PK = protein kinase; pMMR = mismatch repair proficient.
aThe new anticancer treatments initiated before progressive disease by IIR were doxorubicin, carboplatin, temsirolimus, gemcitabine, leuprorelin, pembrolizumab, LY3023414 (mTOR and DNA-PK inhibitor), letrozole, and paclitaxel-bevacizumab.
bThe median was estimated using Kaplan-Meier methodology; 95% CIs were constructed with a generalized Brookmeyer and Crowley method.
cPoint estimates were based on Kaplan-Meier methodology; 95% CIs were based on the Greenwood formula using log-log transformation.
Source: Clinical Study Report.61
Figure 6: Kaplan-Meier Plot of PFS (pMMR, KEYNOTE-146 Trial, FAS)
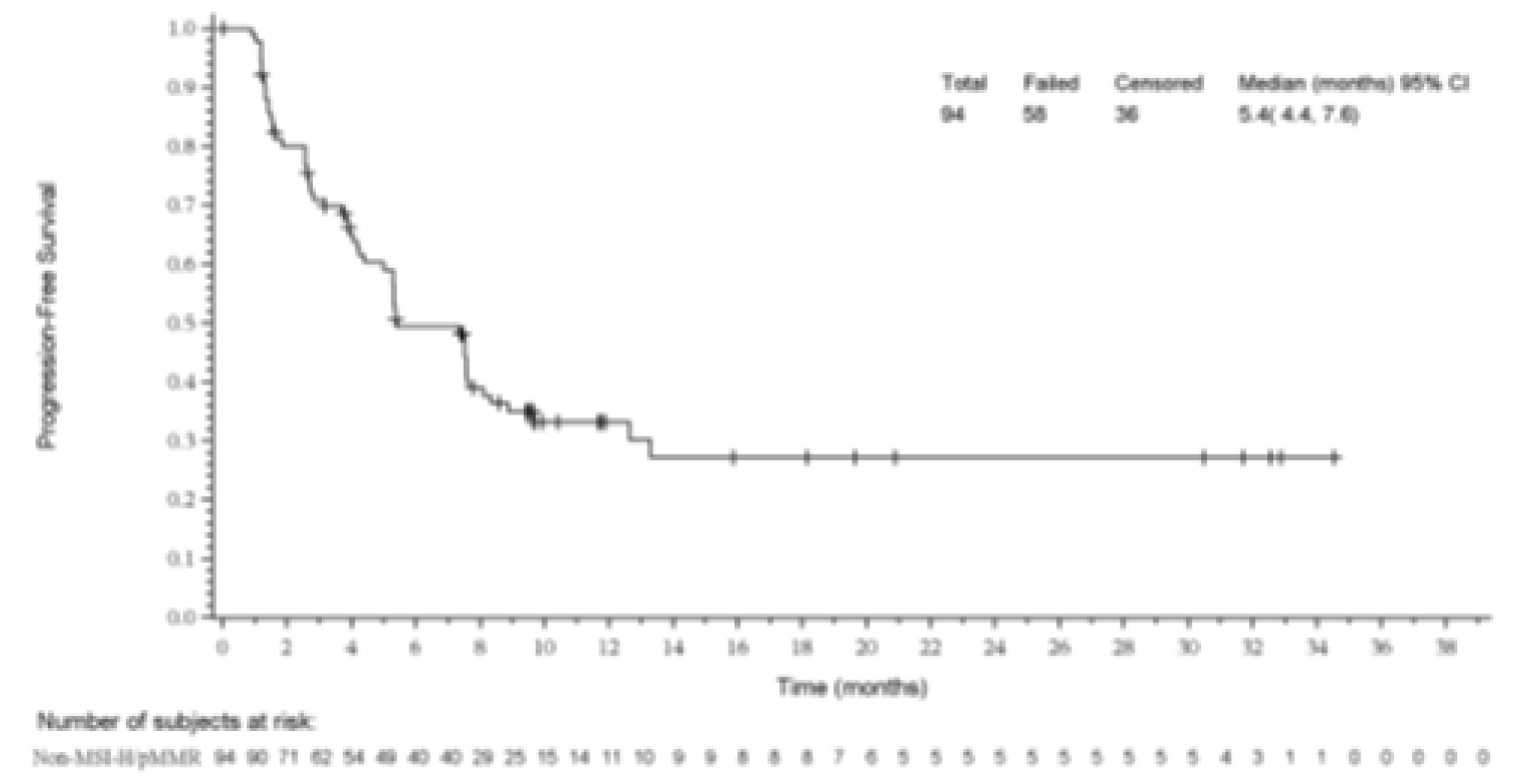
CI = confidence interval; FAS = full analysis set; MSI-H = microsatellite instability-high; pMMR = mismatch repair proficient.
Note: The median was estimated using the Kaplan-Meier method, and the 95% CI was constructed with a generalized Brookmeyer and Crowley method. All tumour assessments were considered as long as patients did not start a new anticancer therapy.
Source: Clinical Study Report.61
Health-Related Quality of Life
HRQoL was not reported.
Symptom Reduction
Symptom reduction was not reported.
Objective Response Rate
At week 24 (the primary outcome), 37 patients (39.4%; 95% CI, 29.4% to 50.0%) had achieved the ORR.
The results for ORR (per IIR) as of the data cut-off date are presented in Table 20. Among patients who had been enrolled for at least 26 weeks before the data cut-off date, 36 patients out of 94 patients achieved an objective response, resulting in an ORR of 38.3% (95% CI, 28.5 to 48.9). Ten patients (10.6%) achieved CR, and 26 patients (27.7%) achieved a partial response.
The subgroup analysis showed that the ORRs were 26.1% (95% CI, 14.3% to 41.1%), 50.0% (95% CI, 35.2% to 64.8%), 42.4% (95% CI, 25.5% to 60.8%), and 80.0% (95% CI, 28.4 to 99.5) in patients with endometrioid adenocarcinoma, non-endometrioid adenocarcinoma, serous adenocarcinoma, and clear-cell adenocarcinoma, respectively (Table 21).
Duration of Response
The results for DOR are presented in Table 22 and Figure 13 in Appendix 3.
Among responders (N = 36), 21 patients (58.3%) were censored, primarily due to lack of progression and no death at the data cut-off date (N = 18, 50%). The probabilities of DOR for 6 months or longer and 12 months or longer were 76% and 51%, respectively (Table 22).
Based on the product-limit (Kaplan–Meier) method for censored data, the median DOR was not estimable (NE) (95% CI, 6.3 to NE; range = 1.2 months and ongoing to 33.1 months and ongoing). The maximum DOR among the 36 responders was 33.1 months and ongoing (refer to Figure 13).
The subgroup analysis showed that the median DORs were NE (95% CI, 4.8 to NE), 11.2 months (95% CI, 6.2 months to NE), NE (95% CI, 5.1 months to NE), and NE (95% CI, 6.3 months to NE) in patients with endometrioid adenocarcinoma, non-endometrioid adenocarcinoma, serous adenocarcinoma, and clear-cell adenocarcinoma, respectively.
Harms
The AEs are presented in Table 23 in this section and Table 62 in Appendix 3. By the data cut-off date, all patients had experienced at least 1 TEAE (N = 94, 100%). The most common TEAEs (occurring in ≥ 50% patients) were hypertension (63.8%), diarrhea (62.8%), fatigue (54.3%), and decreased appetite (51.1%). The proportion of patients reporting an SAE was 52.1%. The most frequent SAEs (> 5%) were hypertension and abdominal pain, reported in 7.4% and 5.3% patients, respectively. The proportion of patients with an AE leading to discontinuation from the treatment was 25.5%. The most common events leading to discontinuation from the treatment were muscular weakness and pancreatitis, each reported in 2.1% patients. Three patients (3.2%) died due to an AE. Overall, the safety profile of PEM + LEN was generally consistent with the known safety profiles of each drug when used as monotherapy, with no new safety signals identified for the combination.61
Table 20: Summary of Tumour Response in pMMR (FAS, KEYNOTE-146 Trial)
Tumour response | PEM + LEN (N = 94) |
|---|---|
Number of patients with ORR (CR + PR), n (%) | 36 (38.3) |
ORR rate, % (95% CI) | 38.3 (28.5 to 48.9) |
Best overall response, n (%) | |
Complete response | 10 (10.6) |
Partial response | 26 (27.7) |
Stable disease | 38 (40.4) |
Progressive disease | 12 (12.8) |
Not evaluable | 8 (8.5) |
CR = complete response; FAS = full analysis set; ORR = objective response rate; PEM + LEN = pembrolizumab plus lenvatinib combination therapy; pMMR = mismatch repair proficient; PR = partial response.
Source: Clinical Study Report.61
Table 21: Objective Response by Histologic Subtype in pMMR (FAS, KEYNOTE-146 Trial)
Subgroup | PEM + LEN (N = 94) |
|---|---|
Histologic subtype, n (%) (95% CI) | |
Endometrioid adenocarcinoma, n (%) | 46 (48.9) |
Patients with response, n (%) | 12 (26.1) |
ORR % (95% CI) | 26.1 (14.3 to 41.1) |
Non-endometrioid adenocarcinoma, n (%) | 48 (51.1) |
Patients with response, n (%) | 24 (50) |
ORR % (95% CI) | 50.0 (35.2 to 64.8) |
Serous adenocarcinoma | 33 (35.1) |
Patients with response, n | 14 (42.4) |
ORR % (95% CI) | 42.4 (25.5 to 60.8) |
Clear-cell adenocarcinoma n (%) | 5 (5.3) |
Patients with response, n (%) | 4 |
ORR % (95% CI) | 80.0 (28.4 to 99.5) |
CI = confidence interval; FAS = full analysis set; ORR = objective response rate; PEM + LEN = pembrolizumab plus lenvatinib combination therapy; pMMR = mismatch repair proficient.
Source: Clinical Study Report.61
Table 22: Summary of Duration of Response in pMMR (FAS, KEYNOTE-146 Trial)
DOR | PEM + LEN (N = 94) |
|---|---|
Number of patients with a response (CR + PR), N | 36 |
Duration of objective response among responders (months)a | |
Median (95% CI) | NE (6.3 to NE) |
Range (min, max) | (1.2+ to 33.1+)b |
Censored duration of response, n (%) | 21/36 (58.3) |
No progression and no death at time of data cut-off | 18/36 (50.0) |
Death or progression after more than 1 missing assessment | 1/36 (2.8) |
New anticancer treatment started | 2/36 (5.6) |
Patients with duration of response among responders: | |
≥ 6 months | |
N | 25 |
Probability (95% CI)b | 0.76 (0.58 to 0.87) |
≥ 12 months | |
N | 8 |
Probability (95% CI)b | 0.51 (0.30 to 0.68) |
Time to response (months) among responders | |
Median (range) | 1.4 (1.1 to 8.0) |
CI = confidence interval; CR = complete response; DOR = duration of response; FAS = full analysis set; NE = not estimable because not reached; PEM + LEN = pembrolizumab plus lenvatinib combination therapy; pMMR = mismatch repair proficient; PR = partial response.
aThe median duration was estimated using the Kaplan-Meier method; the 95% CI was constructed with a generalized Brookmeyer and Crowley method.
b+ indicates the time is censored.
cProbability and 95% CI were calculated using the Kaplan-Meier product-limit method and Greenwood formula.
Source: Clinical Study Report.61
Table 23: Summary of Harms (Safety Set, pMMR, KEYNOTE-146 Trial)
AEs | PEM + LEN (N = 94) |
|---|---|
Patients with at least 1 TEAE, n (%) | 94 (100.0) |
Patients with at least 1 SAE, n (%) | 49 (52.1) |
Patients with dose interruptionc due to an adverse event, n (%) | 74 (78.7) |
Patients with LEN dose reduction due to an adverse event, n (%) | 63 (67.0) |
Patients with a AEs leading to DC from the treatment, n (%) | 24 (25.5) |
Both PEM and LEN discontinuation | 15 (16.0) |
PEM discontinuation | 19 (20.2) |
LEN discontinuation | 21 (22.3) |
Death due to AEs, n (%) | 3 (3.2) |
Notable harms, n (%) | |
Adrenal insufficiency | 3 (3.2) |
Colitis | 2 (2.1) |
Hepatitis | 1 (1.1) |
Hyperthyroidism | 5 (5.3) |
Hypophysitis | 1 (1.1) |
Hypothyroidism | 46 (48.9) |
Nephritis | 2 (2.1) |
Pneumonitis | 1 (1.1) |
Type 1 diabetes mellitus | NR |
Hypertension | 60 (63.8) |
AE = adverse event; DC = discontinuation; LEN = lenvatinib; NR = not reported; PEM = pembrolizumab; PEM + LEN = pembrolizumab plus lenvatinib combination therapy; pMMR = mismatch repair proficient; SAE = serious adverse event; TEAE = treatment-emergent adverse event.
Source: Clinical Study Report.61
Critical Appraisal
Internal Validity
The main limitation of the KEYNOTE-146 trial was its single-arm study design (no comparator arm). Such a design, in addition to a lack of consideration for confounding variables, precludes causal inferences (i.e., the outcomes cannot be directly attributed to PEM). Without an active comparator or any statistical hypothesis testing, it is not possible to assess the relative therapeutic benefit or safety of PEM versus other available treatments (such as chemotherapy in this population). Although inclusion and exclusion criteria are stated, selection procedures are not described; therefore, the potential for selection bias cannot be excluded.
In addition, KEYNOTE-146 was an open-label trial. The study investigators and patients were aware of their treatment status — a situation that increases the risk of detection and performance biases that have the potential to influence outcome reporting. The open-label design is a concern for subjective end points, such as safety, but is unlikely to affect objective outcomes, such as OS. The direction of anticipated bias related to these outcomes is unclear.
Based on the study design, during the follow-up phase, other subsequent anticancer treatments appeared to be allowed. Therefore, it is possible that OS data were confounded by the use of subsequent anticancer therapies potentially received by some patients after disease progression; this may result in bias of OS results.
External Validity
Overall, according to the clinical experts consulted by CADTH, the inclusion and exclusion criteria of the KEYNOTE-146 trial were reasonable, and the baseline patient characteristics, concomitant medications, and prohibited medications were reflective of patients seen in clinical practice for the indication under review. Of the 94 patients included, the majority were White (86.2%) and in the US (86.2%). There were no barriers to identifying patients who would most benefit from the treatment, given that testing for MMR and/or MSI status is standard practice in Canada.
Patients with an ECOG PS of greater than 1 were excluded from this study. Therefore, there is uncertainty in whether the findings may be generalized to these populations. The clinical experts consulted by CADTH indicated that it may be reasonable to expect patients with CNS metastases controlled by radiation, those with carcinosarcomas, and patients with higher ECOG PS scores could benefit from treatment with PEM + LEN.
Overall, as indicated in the Health Canada reviewer’s report,20 due to the limitations inherent in the single-arm design, the findings observed in the KEYNOTE-146 trial need to be confirmed in a phase III study (i.e., KEYNOTE-775).
Discussion
Summary of Available Evidence
This CADTH clinical review report included input from patient groups, clinician groups, clinical experts, drug programs, and a single pivotal phase III randomized controlled trial.
Overall, the KEYNOTE-775 trial was conducted in patients who had either pMMR or dMMR endometrial cancer. A total of 827 patients were included (N = 827); 697 patients were pMMR and 130 patients were dMMR. It is important to emphasize that the pMMR population was not considered a subgroup in the original study design. The sample size and power calculations were based on pMMR populations. The dMMR population was designed as a subgroup. In this review, the pMMR population is the only population of interest, which aligns with Health Canada’s indication and the sponsor’s reimbursement request.8,19,32
The KEYNOTE-775 trial (pMMR population) is a relatively well-designed, ongoing, phase III, multi-centre, randomized, open-label, active-controlled superiority study comparing PEM + LEN to TPC for the treatment of adult patients with advanced endometrial carcinoma that is pMMR (i.e., not MSI-H or dMMR) who have disease progression following prior platinum-based systemic therapy and are not candidates for curative surgery or radiation. A total of 697 patients were randomized in a 1:1 ratio to receive PEM + LEN (n = 346) or TPC (n = 351).
The primary outcomes were PFS and OS. Secondary outcomes included ORR and HRQoL (measured using the EORTC QLQ-C30 GHS scale). Exploratory outcomes included DOR and other HRQoL measurements (i.e., based on the EORTC QLQ-30, EORTC QLQ-EN24 Urological Symptoms Score, and EQ-5D-5L).
This study’s findings were based on IA1 with a data cut-off date of October 26, 2020. It was indicated that the success criteria for the study hypotheses of PFS, OS, and ORR were all met at IA1; consequently, IA2 was not required.17,18
No ITC or other relevant study was identified for this review.
An additional relevant study (KEYNOTE-146) that was included in the sponsor’s submission to CADTH was considered to provide additional longer-term follow-up evidence for this review.
Interpretation of Results
Efficacy
Based on an interim analysis, the median OS was statistically significantly longer in PEM + LEN (17.4 months) compared with TPC (12.0 months; HR = 0.68; 95% CI, 0.56 to 0.84; P = 0.0001). The subgroup analyses of OS were consistent with the primary analysis.
PFS findings were in line with OS. The median PFS was 6.6 months (95% Cl, 5.6 to 7.4) in the PEM + LEN arm, which was statistically significantly longer compared to TPC (HR = 0.60; 95% CI, 0.50 to 0.72; P = 0.0001). The subgroup analyses of PFS were consistent with the primary analysis.
Patient-reported and HRQoL outcomes were identified as important for patients and assessed as secondary or exploratory outcomes. Although there is no expectation of significant improvement in QoL with treatment for this population, overall, there appeared to be no between-group difference observed for PEM + LEN versus TPC in terms of HRQoL.
The clinical experts indicated that ORR is an important outcome in this patient population because, in their experience, improved ORR (based on radiographic evaluation) is usually correlated with improvement in other important clinical outcomes, such as PFS, OS, slower decline in ECOG PS, and delays in the presentation of clinical symptoms, worsening, or HRQoL deterioration; however, these are not always absolutely proportionally correlated. ORR was assessed as the secondary outcome in this study. It was reported that the response rates ranged between 10% and 15% among all available existing treatment options,15 which is consistent with the ORR reported in the TPC arm (15.1%) in the KEYNOTE-775 trial. ORR in KEYNOTE-775 was statistically significant greater in favour of PEM + LEN combination therapy over TPC. The results for ORR are in line with the survival benefit seen for OS and PFS. However, it remains uncertain what impact the clinically meaningful ORR has on improvement in cancer symptoms. Given the known toxicities associated with PEM + LEN, improvement in symptoms may be difficult to determine. Regardless, halting the progression of the disease, improving survival, and improving symptoms are important outcomes for patients.
The median follow-up duration was 12.2 months, which is relatively short and may mean that responses (e.g., OS) are evolving; although the protocol-specified criteria were met for the efficacy analyses, monitoring of safety and efficacy are ongoing. The sponsor provided a final descriptive analysis (cut-off date: March 1, 2022. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. The results of the final analysis appeared to be consistent with those observed in IA1 (cut-off date = October 26, 2020). (Refer to Appendix 5.) In the KEYNOTE-146 trial, the median follow-up time for OS was 18.7 months (95% CI, 14.1 months to 20.9 months), which was longer than that in KEYNOTE-775 trial. However, the KEYNOTE-146 trial was limited due to its single-arm design. It is noted that the results observed in this trial were consistent with those reported in the KEYNOTE-775 trial.
Overall, the KEYNOTE-775 trial met the predefined criteria for statistical significance for both of its primary end points (OS and PFS) and for its key secondary end point (ORR). The clinical experts consulted by CADTH for this review indicated that the findings for OS, PFS, and ORR reported in the KEYNOTE-775 trial are considered clinically meaningful in this population.
According to the clinical experts consulted by CADTH, patients recruited for the KEYNOTE-775 trial were considered representative of patients in Canadian clinical practice. There were no major concerns about the generalizability of the findings to Canadian practice. The clinical experts anticipated that because of the mechanism of action and acceptable safety profile of PEM + LEN combination therapy, they would expect to experience a benefit of treatment with it for all patients with pMMR endometrial cancer who have received at least 1 previous line of systemic therapy.
Harms
The safety profile of PEM + LEN for the treatment of various cancers, including endometrial cancer, has been well established in previous clinical trials.8,32,46,65 The proportions of patients experiencing at least 1 TEAE were similar in the PEM + LEN group and the TPC group (i.e., 99.7% in both). The frequency of SAEs was higher than in the TPC arm in the KEYNOTE-775 trial. However, when adjusted for exposure, the incidence of SAEs appeared similar between the 2 treatment groups (i.e., the number of SAEs per 100 person-months were 9.83 versus 9.4 in the PEM + LEN versus TPC groups, respectively) (Table 58). More patients discontinued the study medication due to AEs than did those on TPC (PEM + LEN = 31.0%; TPC = 8.3%). Notable AEs (i.e., the AEs of special interest for this review) were higher in the PEM + LEN group than in the TPC group. The higher incidence of notable harms in the PEM + LEN group was driven primarily by hypothyroidism, hyperthyroidism, and hypertension. Overall, the clinical experts consulted by CADTH for this review agreed that the safety profile of PEM + LEN observed in this study appeared consistent with the known safety profile of each individual drug (PEM or LEN), with no additional safety signals identified. Additionally, the clinical experts indicated that the AEs observed in the study were generally manageable with dose interruption or discontinuation of PEM, LEN, or both, or with LEN dose reduction, with or without concomitant steroid therapy.
Conclusions
One sponsor-submitted, phase III, multinational, open-label, randomized, active-controlled trial (KEYNOTE-775) was included in this review. Compared with TPC (doxorubicin or paclitaxel), PEM + LEN combination therapy showed a statistically significant and clinically meaningful benefit in terms of OS, PFS, and ORR in the treatment of adult patients with advanced pMMR (i.e., non-MSI-H or dMMR) endometrial carcinoma who have disease progression following prior platinum-based systemic therapy and are not candidates for curative surgery or radiation. The clinical experts consulted for this review indicated that the safety profile of PEM + LEN observed in this study appeared consistent with the known safety profile of each individual drug (PEM or LEN), with no additional safety signals identified. The AEs observed in the study were generally manageable with dose interruption, dose discontinuation, or LEN dose reduction, with or without concomitant steroid therapy.
References
1.Plaxe SC, Mundt AJ. Overview of endometrial carcinoma. In: Post TW, ed. UpToDate. Waltham (MA): UpToDate; 2022 Feb 28: https://www.uptodate.com. Accessed 2022 Feb 28.
2.Uterine cancer: risk factors and prevention. Alexandria (VA): American Society of Clinical Oncology; 2020: https://www.cancer.net/cancer-types/uterine-cancer/risk-factors-and-prevention. Accessed 2022 Feb 28.
3.Cancerous tumours of the uterus. Toronto (ON): Canadian Cancer Society; 2021: https://cancer.ca/en/cancer-information/cancer-types/uterine/what-is-uterine-cancer/cancerous-tumours. Accessed 2022 Feb 28.
4.Ott PA, Bang YJ, Berton-Rigaud D, et al. Safety and antitumor activity of pembrolizumab in advanced programmed death ligand 1-positive endometrial cancer: results from the KEYNOTE-028 study. Obstet Gynecol Surv. 2018;73(1):26-27.
5.Endometrial cancer: detection, diagnosis, staging, survival rates. Atlanta (GA): American Cancer Society; 2021: https://www.cancer.org/cancer/endometrial-cancer/detection-diagnosis-staging/survival-rates.html1. Accessed 2022 Feb 28.
6.Fung-Kee-Fung M, Dodge J, Elit L, et al. Follow-up after primary therapy for endometrial cancer: a systematic review. Gynecol Oncol. 2006;101(3):520-529. PubMed
7.Kurnit KC, Westin SN, Coleman RL. Microsatellite instability in endometrial cancer: new purpose for an old test. Cancer. 2019;125(13):2154-2163. PubMed
8.PrKeytruda® (pembrolizumab): powder for solution for infusion 50 mg, solution for infusion 100 mg/4 mL vial [draft product monograph]. Kirckland (QC): Merck Canada; 2021 Jun 2.
9.O'Malley DM, Bariani GM, Cassier PA, et al. Pembrolizumab in patients with microsatellite instability-high advanced endometrial cancer: results from the KEYNOTE-158 study. J Clin Oncol. 2022;40(7):752-761. PubMed
10.Lorusso D, Colombo N, Casado Herraez A, et al. Health-related quality of life in advanced endometrial cancer patients treated with lenvatinib + pembrolizumab or treatment of physician’s choice [poster]. Presented at the American Society of Clinical Oncology Annual Meeting; Jun 4-8, 2021; Virtual format.
11.Endometrial cancer. (Clinical practice guideline GYNE-002. Version 5). Edmonton (AB): Alberta Health Services; 2015: https://www.albertahealthservices.ca/assets/info/hp/cancer/if-hp-cancer-guide-gyne002-endometrial.pdf. Accessed 2022 Feb 28.
12.Genital tract cancers in females: endometrial cancer. BC guidelines. Victoria (BC): Province of British Columbia; 2014: https://www2.gov.bc.ca/gov/content/health/practitioner-professional-resources/bc-guidelines/endometrial-cancer. Accessed 2022 Feb 28.
13.Treatment option overview for endometrial cancer. Bethesda (MD): National Cancer Institute; 2021: https://www.cancer.gov/types/uterine/hp/endometrial-treatment-pdq#_36. Accessed 2022 Mar 2.
14.Systemic therapy for advanced or recurrent endometrial cancer and advanced or recurrent uterine papillary serous carcinoma. Toronto (ON): Cancer Care Ontario; 2019: https://www.cancercareontario.ca/en/guidelines-advice/types-of-cancer/501. Accessed 2022 Feb 28.
15.Concin N, Matias-Guiu X, Vergote I, et al. ESGO/ESTRO/ESP guidelines for the management of patients with endometrial carcinoma. Int J Gynecol Cancer. 2021;31(1):12-39. PubMed
16.Campos SM, Cohn DE. Treatment of metastatic endometrial cancer. In: Post TW, ed. UpToDate. Waltham (MA): UpToDate; 2021 Nov 8: https://www.uptodate.com. Accessed 2022 Feb 28.
17.Study protocol number: 775-00/E7080-G000-309 Protocol Title: A Multicenter, Open-label, Randomized, Phase 3 Trial to Compare the Efficacy and Safety of Lenvatinib in Combination with Pembrolizumab Versus Treatment of Physician’s Choice in Participants with Advanced Endometrial Cancer [internal sponsor's report]. Woodcliff Lake (NJ): Eisai Inc.; 2018 Feb 13.
18.Pharmacoeconomic evaluation [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Keytruda (pembrolizumab), powder for solution for infusion 50 mg, solution for infusion 100 mg/4 mL vial with levantinib. Kirkland (QC): Merck Canada; 2022 Jan 6.
19.Clinical Study Report: P775V01MK3475. A multicenter, open-label, randomized, phase 3 trial to compare the efficacy and safety of lenvatinib in combination with pembrolizumab versus treatment of physician’s choice in participants with advanced endometrial cancer [internal sponsor's report]. Woodcliff Lake (NJ): Eisai; 2021.
20.Health Canada's reviewer report: Keytruda (pembrolizumab) [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Keytruda (pembrolizumab), powder for solution for infusion 50 mg, solution for infusion 100 mg/4 mL vial with levantinib. Kirkland (QC): Merck Canada; 2022 Jan 6.
21.Cancer-specific stats 2021. Toronto (ON): Canadian Cancer Society; 2021: https://cdn.cancer.ca/-/media/files/research/cancer-statistics/2021-statistics/2021_cancer-specific-stats.pdf?rev=bbadfd869b66415e97be9655a842858c&hash=3E552CE702E1BD7657E4E91FB7BA624B&_ga=2.72656595.1540445839.1639772303-397212376.1636582496. Accessed 2022 Feb 28.
22.Uterine cancer statistics. Toronto (ON): Canadian Cancer Society; 2021: https://cancer.ca/en/cancer-information/cancer-types/uterine/statistics. Accessed 2022 Feb 28.
23.Endometrial cancer stages. Atlanta (GA): American Cancer Society; 2019: https://www.cancer.org/cancer/endometrial-cancer/detection-diagnosis-staging/staging.html. Accessed 2022 Feb 28.
24.Stages of uterine cancer. Toronto (ON): Canadian Cancer Society; 2021: https://cancer.ca/en/cancer-information/cancer-types/uterine/staging. Accessed 2022 Feb 28.
25.Boland CR, Thibodeau SN, Hamilton SR, et al. A National Cancer Institute Workshop on Microsatellite Instability for cancer detection and familial predisposition: development of international criteria for the determination of microsatellite instability in colorectal cancer. Cancer Res. 1998 Nov 15;59(22). PubMed
26.McEachron J, Zhou N, Spencer C, et al. Adjuvant chemoradiation associated with improved outcomes in patients with microsatellite instability-high advanced endometrial carcinoma. Int J Gynecol Cancer. 2021;31(2):203-208. PubMed
27.Pakish JB, Zhang Q, Chen Z, et al. Immune microenvironment in microsatellite-instable endometrial cancers: hereditary or sporadic origin matters. Clin Cancer Res. 2017;23(15):4473-4481. PubMed
28.Meyer LA, Broaddus RR, Lu KH. Endometrial cancer and Lynch syndrome: clinical and pathologic considerations. Cancer Control. 2009;16(1):14-22. PubMed
29.Marabelle A, Le DT, Ascierto PA, et al. Efficacy of pembrolizumab in patients with noncolorectal high microsatellite instability/mismatch repair-deficient cancer: results from the phase II KEYNOTE-158 study. J Clin Oncol. 2020;38(1):1-10. PubMed
30.Smithgall M.C., Remotti H, Hisiao SJ. (2021) Investigation of discrepant mismatch repair immunohistochemistry and microsatellite instability polymerase chain reaction test results for gynecologic cancers using next-generation sequencing. Human Pathology. 119: 41-50. . PubMed
31.Malapelle U, Parente P, Pepe F et al. Evaluation of micro satellite instability and mismatch repair status in different solid tumors: a multicenter analysis in a real world setting. Cell. 2021 10(8):1878. PubMed
32.Drug Reimbursement Review sponsor submission: Keytruda (pembrolizumab), powder for solution for infusion 50 mg, solution for infusion 100 mg/4 mL vial with levantinib [internal sponsor's package]. Kirkland (QC): Merck Canada; 2022 Mar 4.
33.Howitt BE, Shukla SA, Sholl LM, et al. Association of polymerase e-mutated and microsatellite-instable endometrial cancers with neoantigen load, number of tumor-infiltrating lymphocytes, and expression of PD-1 and PD-L1. JAMA Oncol. 2015;1(9):1319-1323. PubMed
34.Rubinstein MM, Makker V. Optimizing immunotherapy for gynecologic cancers. Curr Opin Obstet Gynecol. 2020;32(1):1-8. PubMed
35.Green AK, Feinberg J, Makker V. A review of immune checkpoint blockade therapy in endometrial cancer. Am Soc Clin Oncol Educ Book. 2020;40:1-7. PubMed
36.Lorenzi M, Amonkar M, Zhang J, Mehta S, Liaw K-L. Epidemiology of microsatellite instability high (MSI-H) and deficient mismatch repair (dMMR) in solid tumors: a structured literature review. J Oncol. 2020:1807929.
37.Zhao P, Li L, Jiang X, Li Q. Mismatch repair deficiency/microsatellite instability-high as a predictor for anti-PD-1/PD-L1 immunotherapy efficacy. J Hematol Oncol. 2019;12(1):54. PubMed
38.Ryan NAJ, Glaire MA, Blake D, Cabrera-Dandy M, Evans DG, Crosbie EJ. The proportion of endometrial cancers associated with Lynch syndrome: a systematic review of the literature and meta-analysis. Genet Med. 2019;21(10):2167-2180. PubMed
39.Bonneville R, Krook MA, Kautto EA, et al. Landscape of microsatellite instability across 39 cancer types. JCO Precis Oncol. 2017;2017.
40.Sorbe B, Juresta C, Ahlin C. Natural history of recurrences in endometrial carcinoma. Oncol Lett. 2014;8(4):1800-1806. PubMed
41.Sovak MA, Dupont J, Hensley ML, et al. Paclitaxel and carboplatin in the treatment of advanced or recurrent endometrial cancer: a large retrospective study. Int J Gynecol Cancer. 2007;17(1):197-203. PubMed
42.Sorbe B, Andersson H, Boman K, Rosenberg P, Kalling M. Treatment of primary advanced and recurrent endometrial carcinoma with a combination of carboplatin and paclitaxel-long-term follow-up. Int J Gynecol Cancer. 2008;18(4):803-808. PubMed
43.Makker V, Hensley ML, Zhou Q, Iasonos A, Aghajanian CA. Treatment of advanced or recurrent endometrial carcinoma with doxorubicin in patients progressing after paclitaxel/carboplatin: Memorial Sloan-Kettering Cancer Center experience from 1995 to 2009. Int J Gynecol Cancer. 2013;23(5):929-934. PubMed
44.Miller DS, Filiaci VL, Mannel RS, et al. Carboplatin and paclitaxel for advanced endometrial cancer: final overall survival and adverse event analysis of a phase III trial (NRG Oncology/GOG0209). J Clin Oncol. 2020;38(33):3841-3850. PubMed
45.Brooks RA, Fleming GF, Lastra RR, et al. Current recommendations and recent progress in endometrial cancer. CA Cancer J Clin. 2019;69(4):258-279. PubMed
46.PrLenvima® (lenvatinib): 4 mg and 10 mg capsules [product monograph]. Mississauga (ON): Eisai; 2021 Jul 22.
47.PrDoxorubinicin (doxorubicin hydrochloride): 2 mg/mL, 10 mg (5 mL0 and 50 mg (25 mL) solution for injection [product monograph]. Kirkland (QC): Pfizer 2021 Sep 30.
48.PrPaclitaxel Injection USP (paclitaxel): 6 mg/mL solution for injection [product monograph]. Kirkland (QC): Accord Healthcare; 2020 Sep 14: https://pdf.hres.ca/dpd_pm/00057982.PDF. Accessed 2022 Feb 11.
49.McGowan J, Sampson M, Salzwedel DM, Cogo E, Foerster V, Lefebvre C. PRESS Peer Review of Electronic Search Strategies: 2015 guideline statement. J Clin Epidemiol. 2016;75:40-46. PubMed
50.Grey matters: a practical tool for searching health-related grey literature. Ottawa (ON): CADTH; 2019: https://www.cadth.ca/grey-matters. Accessed 2022 Jan 22.
51.Makker V, Colombo N, Casado Herraez A, et al. Lenvatinib plus pembrolizumab for advanced endometrial cancer. N Engl J Med. 2022;386(5):437-448. PubMed
52.Aaronson NK, Ahmedzai S, Bergman B, et al. The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst. 1993;85(5):365-376. PubMed
53.Davda J, Kibet H, Achieng E, Atundo L, Komen T. Assessing the acceptability, reliability, and validity of the EORTC Quality of Life Questionnaire (QLQ-C30) in Kenyan cancer patients: a cross-sectional study. J Patient Rep Outcomes. 2021;5(1):4. PubMed
54.EORTC quality of life. https://qol.eortc.org/. Accessed 2022 Mar 9.
55.Pickard AS, Neary MP, Cella D. Estimation of minimally important differences in EQ-5D utility and VAS scores in cancer. Health Qual Life Outcomes. 2007;5:70. PubMed
56.EuroQol Research Foundation. EQ-5D-5L user guide: basic information on how to use the EQ-5D-5L instrument. 2021; https://euroqol.org/wp-content/uploads/2021/01/EQ-5D-5LUserguide-08-0421.pdf. Accessed 2022 Jun 14.
57.Health Quality Council of Alberta. 2014 Alberta population norms for EQ-5D-5L. 2014; https://d10k7k7mywg42z.cloudfront.net/assets/542f01f2edb2f37083002e54/2014_EQ_5D_5L_report_FINALFINAL.pdf. Accessed 2022 Jun 14.
58.Herdman M, Gudex C, Lloyd A, et al. Development and preliminary testing of the new five-level version of EQ-5D (EQ-5D-5L). Qual Life Res. 2011;20(10):1727-1736. PubMed
59.Clopper CJ, Pearson ES. The use of confidence or fiducial limits illustrated in the case of the binomial. Biometrika 1934;26(4):404-13.
60.Keytruda (MK-3475) MSI-H endometrial carcinoma Indirect Treatment Comparison KN158 vs. KN775 [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Keytruda (pembrolizumab), powder for solution for infusion 50 mg, solution for infusion 100 mg/4 mL vial. Kirkland (QC): Merck Canada; 2022 Jan 6.
61.Clinical Study Report E7080-A001-111. A multicenter, open-label phase 1b/2 trial of lenvatinib (E7080) plus pembrolizumab in subjects with selected solid tumors [internal sponsor's report]. Woodcliff Lake (NJ): Eisai; 2019.
62.Eisai Inc. A Trial of Lenvatinib (E7080) Plus Pembrolizumab in Participants With Selected Solid Tumors. Bethesda (MD) ClinicalTrials.gov; 2022 Mar 7: https://www.clinicaltrials.gov/ct2/show/NCT02501096. Accessed 2022 Jun 14.
63.Makker V, Taylor MH, Aghajanian C, et al. Lenvatinib plus pembrolizumab in patients with advanced endometrial cancer. J Clin Oncol. 2020;38(26):2981-2992. PubMed
64.Makker V, Taylor MH, Oaknin A, et al. Characterization and management of adverse reactions in patients with advanced endometrial carcinoma treated with lenvatinib plus pembrolizumab. Oncologist. 2021;26(9):e1599-e1608. PubMed
65.Clinical study report: Study E7080-A001-111/KEYNOTE 146 Endometrial Carcinoma, A Multicenter, Open-Label Phase 1b/2 Trial of Lenvatinib (E7080) Plus Pembrolizumab in Subjects With Selected Solid Tumors [internal sponsor's report]. Woodcliff Lake (NJ): Eisai Inc. 2018 Jul 31.
66.Ackroyd SA, Huang ES, Kurnit KC, Lee NK. Pembrolizumab and lenvatinib versus carboplatin and paclitaxel as first-line therapy for advanced or recurrent endometrial cancer: A Markov analysis. Gynecol Oncol. 2021;162(2):249-255. PubMed
67.Makker V, Rasco D, Vogelzang NJ, et al. Lenvatinib plus pembrolizumab in patients with advanced endometrial cancer: an interim analysis of a multicentre, open-label, single-arm, phase 2 trial. Lancet Oncol. 2019;20(5):711-718. PubMed
68.Marth C, Tarnawski R, Tyulyandina A, et al. Phase 3, randomized, open-label study of pembrolizumab plus lenvatinib versus chemotherapy for first-line treatment of advanced or recurrent endometrial cancer: ENGOT-en9/LEAP-001. Int J Gynecol Cancer. 2022;32(1):93-100. PubMed
69.Taylor MH, Lee CH, Makker V, et al. Phase IB/II Trial of Lenvatinib Plus Pembrolizumab in Patients With Advanced Renal Cell Carcinoma, Endometrial Cancer, and Other Selected Advanced Solid Tumors. J Clin Oncol. 2020;38(11):1154-1163. PubMed
70.Osoba D, Zee B, Pater J, Warr D, Kaizer L, Latreille J. Psychometric properties and responsiveness of the EORTC quality of Life Questionnaire (QLQ-C30) in patients with breast, ovarian and lung cancer. Qual Life Res. 1994;3(5):353-364. PubMed
71.Zhao H, Kanda K. Translation and validation of the standard Chinese version of the EORTC QLQ-C30. Qual Life Res. 2000;9(2):129-137. PubMed
72.Butler L, Bacon M, Carey M, Zee B, Tu D, Bezjak A. Determining the relationship between toxicity and quality of life in an ovarian cancer chemotherapy clinical trial. J Clin Oncol. 2004;22(12):2461-2468. PubMed
73.Bedard G, Zeng L, Zhang L, et al. Minimal important differences in the EORTC QLQ-C30 in patients with advanced cancer. Asia Pac J Clin Oncol. 2014;10(2):109-117. PubMed
74.Osoba D, Rodrigues G, Myles J, Zee B, Pater J. Interpreting the significance of changes in health-related quality-of-life scores. J Clin Oncol. 1998;16(1):139-144. PubMed
75.Gallardo-Rincon D, Toledo-Leyva A, Bahena-Gonzalez A, et al. Validation of the QLQ-EN24 instrument for the assessment of health-related quality of life for women with endometrial cancer in Mexico. Arch Gynecol Obstet. 2021;304(3):773-782. PubMed
76.Greimel E, Nordin A, Lanceley A, et al. Psychometric validation of the European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire-Endometrial Cancer Module (EORTC QLQ-EN24). Eur J Cancer. 2011;47(2):183-190. PubMed
77.Stukan M, Zalewski K, Mardas M, et al. Independent psychometric validation of European Organization for Research and Treatment of Cancer Quality of Life Questionnaire-Endometrial Cancer Module (EORTC QLQ-EN24). European Journal of Cancer Care. 2018;27(1). PubMed
78.Bjordal K, de Graeff A, Fayers PM, et al. A 12 country field study of the EORTC QLQ-C30 (version 3.0) and the head and neck cancer specific module (EORTC QLQ-H&N35) in head and neck patients. EORTC Quality of Life Group. Eur J Cancer. 2000;36(14):1796-1807. PubMed
79.Bergman B, Sullivan M, Sörenson S. Quality of life during chemotherapy for small cell lung cancer. II. A longitudinal study of the EORTC Core Quality of Life Questionnaire and comparison with the Sickness Impact Profile. Acta Oncol. 1992;31(1):19-28. PubMed
80.Bjordal K, Kaasa S. Psychometric validation of the EORTC Core Quality of Life Questionnaire, 30-item version and a diagnosis-specific module for head and neck cancer patients. Acta Oncol. 1992;31(3):311-321. PubMed
81.EORTC QLC-C30 scoring manual. European Organisation for Research and Treatment of Cancer (EORTC) 2001; https://www.eortc.org/app/uploads/sites/2/2018/02/SCmanual.pdf. Accessed April 26 2021.
82.Study 309/KEYNOTE-775: Final Analysis Results In: Drug Reimbursement Review sponsor submission: Keytruda (pembrolizumab), powder for solution for infusion 50 mg, solution for infusion 100 mg/4 mL vial with levantinib [internal sponsor's package]. Kirkland (QC): Merck Canada; 2022 Mar 4.
83.Lenvatinib and pembrolizumab in advanced endometrial carcinoma: long-term efficacy and safety update from a phase 1b/2 study [internal sponsor's report]. Drug Reimbursement Review sponsor submission: Keytruda (pembrolizumab), powder for solution for infusion 50 mg, solution for infusion 100 mg/4 mL vial with levantinib. Kirkland (QC): Merck Canada; 2022 Mar 4.
Appendix 1: Literature Search Strategy
Note that this appendix has not been copy-edited.
Clinical Literature Search
Overview
Interface: Ovid
Databases:
MEDLINE All (1946-present)
Embase (1974-present)
Note: Subject headings and search fields have been customized for each database. Duplicates between databases were removed in Ovid.
Date of search: January 21, 2022
Alerts: Bi-weekly search updates until project completion
Search filters applied: No filters were applied to limit retrieval by study type.
Limits:
Conference abstracts: excluded
Syntax | Description |
|---|---|
/ | At the end of a phrase, searches the phrase as a subject heading |
exp | Explode a subject heading |
* | Before a word, indicates that the marked subject heading is a primary topic; or, after a word, a truncation symbol (wildcard) to retrieve plurals or varying endings |
? | Truncation symbol for one or no characters only |
adj# | Requires terms to be adjacent to each other within # number of words (in any order) |
.ti | Title |
.ot | Original title |
.ab | Abstract |
.hw | Heading word; usually includes subject headings and controlled vocabulary |
.kf | Author keyword heading word (MEDLINE) |
.dq | Candidate term word (Embase) |
.pt | Publication type |
.mp | Mapped term |
.rn | Registry number |
.nm | Name of substance word (MEDLINE) |
medall | Ovid database code: MEDLINE All, 1946 to present, updated daily |
oemezd | Ovid database code; Embase, 1974 to present, updated daily |
Multi-Database Strategy
(pembrolizumab* or Keytruda* or SCH-900475 or SCH900475 or MK-3475 or MK3475 or lambrolizumab* or HSDB8257 or HSDB 8257 or Merck 3475 or DPT0O3T46P).ti,ab,ot,kf,hw,nm,rn.
exp Endometrial neoplasms/
((endometri* or uterus or uterine) adj3 (cancer* or carcinoma* or neoplas* or sarcoma* or tumo?r* or adenocarcinoma* or carcinosarcoma*)).ti,ab,kf.
1 and (2 or 3)
4 use edal
*pembrolizumab/
(pembrolizumab* or Keytruda* or SCH-900475 or SCH900475 or MK-3475 or MK3475 or lambrolizumab* or HSDB8257 or HSDB 8257 or Merck 3475).ti,ab,kf,dq.
6 or 7
exp Endometrium tumor/
((endometri* or uterus or uterine) adj3 (cancer* or carcinoma* or neoplas* or sarcoma* or tumo?r* or adenocarcinoma* or carcinosarcoma*)).ti,ab,kf,dq.
9 or 10
8 and 11
12 use oemezd
12 not (conference abstract or conference review).pt.
5 or 14
remove duplicates from 15
Clinical Trials Registries
ClinicalTrials.gov
Produced by the US National Library of Medicine. Targeted search used to capture registered clinical trials.
Search – pembrolizumab | “Endometrial Neoplasms”
WHO ICTRP
International Clinical Trials Registry Platform, produced by the World Health Organization. Targeted search used to capture registered clinical trials.
Search terms – pembrolizumab AND endometrial
Health Canada’s Clinical Trials Database
Produced by Health Canada. Targeted search used to capture registered clinical trials.
Search terms – pembrolizumab AND endometrial
EU Clinical Trials Register
European Union Clinical Trials Register, produced by the European Union. Targeted search used to capture registered clinical trials.
Search terms – pembrolizumab AND endometrial
Grey Literature
Search dates: January 13-18, 2022
Keywords: Keytruda/pembrolizumab, endometrial cancer
Limits: Conference abstracts excluded
Updated: Search updated prior to the completion of stakeholder feedback period
Relevant websites from the following sections of the CADTH grey literature checklist Grey Matters: A Practical Tool for Searching Health-Related Grey Literature were searched:
Health Technology Assessment Agencies
Health Economics
Clinical Practice Guidelines
Drug and Device Regulatory Approvals
Advisories and Warnings
Drug Class Reviews
Clinical Trials Registries
Databases (free)
Appendix 2: Excluded Studies
Note that this appendix has not been copy-edited.
Reference | Reason for Exclusion |
|---|---|
Ackroyd SA, Huang ES, Kurnit KC, Lee NK. Pembrolizumab and lenvatinib versus carboplatin and paclitaxel as first-line therapy for advanced or recurrent endometrial cancer: A Markov analysis. Gynecol Oncol. 2021;162(2):249-255.66 | First-line treatment, Population not of interest |
Makker V, Rasco D, Vogelzang NJ, et al. Lenvatinib plus pembrolizumab in patients with advanced endometrial cancer: an interim analysis of a multi-centre, open-label, single-arm, phase 2 trial. Lancet Oncol. 2019;20(5):711-718.67 | Study design not of interest (single-arm phase II) |
Marth C, Tarnawski R, Tyulyandina A, et al. Phase 3, randomized, open-label study of pembrolizumab plus lenvatinib versus chemotherapy for first-line treatment of advanced or recurrent endometrial cancer: ENGOT-en9/LEAP-001. Int J Gynecol Cancer. 2022;32(1):93-100.68 | First-line treatment, Population not of interest |
Taylor MH, Lee CH, Makker V, et al. Phase IB/II Trial of Lenvatinib Plus Pembrolizumab in Patients With Advanced Renal Cell Carcinoma, Endometrial Cancer, and Other Selected Advanced Solid Tumors. J Clin Oncol. 2020;38(11):1154-1163.69 | Study design not of interest (Phase Ib/II) |
Appendix 3: Detailed Outcome Data
Note that this appendix has not been copy-edited.
Document | Date of Issue | Overall Rationale |
|---|---|---|
Original protocol | February 13, 2018 | Not applicable. |
Amendment 01 | March 21, 2018 | Germany-specific amendment to address country-specific request for HIV/HBV/HCV testing and pregnancy testing at screening. |
Amendment 02 | June 06, 2018 | UK-specific amendment to address country-specific requests for HIV/HBV/HCV testing at screening and contraception use. |
Amendment 03 | August 31, 2018 | Global protocol amendment to provide clarity with respect to the number of prior lines of treatment in order to be eligible for the study. |
Amendment 04 | October 01, 2018 | Germany-specific amendment to address country-specific requests for HIV/HBV/HCV testing and pregnancy testing and to incorporate changes implemented in Amendment 03 to provide clarity with respect to the number of prior lines of treatment in order to be eligible for the study. |
Amendment 05 | October 02, 2018 | UK-specific amendment to address country-specific requests for HIV/HBV/HCV testing and to incorporate changes implemented in Amendment 03 to provide clarity with respect to the number of prior lines of treatment in order to be eligible for the study. |
Amendment 06 | February 18, 2020 | Revision to the statistical analysis plan to add an interim efficacy analysis to evaluate the superiority of PFS and OS. |
Amendment 07 | June 12, 2020 | Revision to clarify that, based on multiplicity testing strategy, the study would be considered positive if testing of either PFS or OS is significant in patients with pMMR tumours. PFS and OS would then be tested in the all-comer population. Revision to the statistical analysis plan to revise the timing of interim efficacy analysis following communications with health authorities. |
PFS= progression-free survival; pMMR= mismatch repair proficient; OS = overall survival.
Source: Clinical Study Report.19
Table 27: Dose Modification Guidelines for Lenvatinib-Related Adverse Events (PEM + LEN Combination Arm)
Treatment-Related Toxicitya,b | Management | Dose Adjustment |
|---|---|---|
Grade 1 or Tolerable Grade 2 | Continue treatment | No change |
Intolerable Grade 2c,d or Grade 3e | ||
First occurrence | Interrupt lenvatinib until resolved to Grade 0-1, or tolerable Grade 2 | Reduce lenvatinib dose to 14mg once a day (1-level reduction) |
Second occurrence (same toxicity or new toxicity) | Interrupt lenvatinib until resolved to Grade 0-1, or tolerable Grade 2 | Reduce lenvatinib dose to 10 mg once a day (1-level reduction) |
Third occurrence (same toxicity or new toxicity) | Interrupt lenvatinib until resolved to Grade 0-1, or tolerable Grade 2 | Reduce lenvatinib dose to 8 mg orally once a day (1-level reduction) |
Fourth occurrence (same toxicity or new toxicity) | Interrupt lenvatinib | Discuss with MSD |
Grade 4f | Discontinue Study Treatment | Discontinue Study Treatment |
AE = adverse event; BMI = body mass index; CTCAE = Common Terminology Criteria for Adverse Events; MSD = Merck Sharp & Dohme Corporation; PEM + LEN = pembrolizumab + lenvatinib combination.
aAn interruption of study treatment for more than 28 days will require MSD’s approval before treatment can be resumed.
bInitiate optimal medical management for nausea, vomiting, hypertension, hypothyroidism and/or diarrhea prior to any lenvatinib interruption or dose reduction.
cApplicable only to Grade 2 toxicities judged by the participant and/or physician to be intolerable.
dObese patients with weight loss do not need to return to the baseline weight or 10% of baseline weight (i.e., Grade 1weight loss). These patients will restart the study drug(s) at a lower dose once their weight remains stable for at least 1 week and they reached the normal BMI (if the weight loss occurred but it is still above normal BMI, they can restart the study treatment at a lower dose once the weight has been stable for at least 1 week). Normal BMI should be used as the new baseline for further dose reductions.
eFor asymptomatic laboratory abnormalities, such as Grade ≥3 elevations of amylase and lipase that are not considered clinically relevant by the investigator, continuation of treatment should be discussed with MSD.
fExcluding laboratory abnormalities judged to be non-life-threatening, in which case manage as Grade 3.
Note: For grading, refer to CTCAE version 4.0. Collect all AE grades (i.e., decreasing and increasing CTCAE grade).
Source: Clinical Study Report.19
Study Drug | Dose Formulation | Unit Dose Strength(s) | Dosage Level(s) | Route of Administration |
|---|---|---|---|---|
Lenvatinib | Capsule | 10 mg,4 mga | 20 mg | Orally, q.d. |
Pembrolizumab | Solution for infusion | 25 mg/mL | 200 mg | IV, q.3.w. |
Doxorubicin | Solution for infusion | Variable | 60 mg/m2 | IV, q.3.w. |
Paclitaxel | Solution for infusion | Variable | 80 mg/m2 | IV, q.w.b |
q.3.w. = every 3 weeks; q.d. = once daily; q.w. = every week.
a4 mg capsules provided for successive dose reduction of lenvatinib.
b28-day cycle with weekly administration; 3 weeks on and 1 week off.
Note: Maximum doses of study drugs: pembrolizumab (35 cycles); doxorubicin (cumulative lifetime dosage of 500 mg/m2 or lower as consistent with site’s standard of care); paclitaxel (per site standard of care); no maximum number of doses for lenvatinib.
Source: Clinical Study Report.19
Table 29: Prior Therapies for Endometrial Cancer (ITT Population)
Characteristic | KEYNOTE-775 | |
|---|---|---|
PEM + LEN (N=346) | TPC (N=351) | |
Patients in population, N | 346 | 351 |
Prior lines of systemic therapy, n (%) | ||
1 | 244 (70.5) | 226 (64.4) |
2 | 92 (26.6) | 114 (32.5) |
≥3 | 10 (2.9) | 11 (3.1) |
Prior lines of platinum-based therapy | ||
0 | 1 (0.3) | 0 (0.0) |
1 | 269 (77.7) | 257 (73.2) |
2 | 75 (21.7) | 94 (26.8) |
≥3 | 1 (0.3) | 0 (0.0) |
With neoadjuvant/adjuvant, n (%) | 197 (56.9) | 219 (62.4) |
Primary Therapy, n (%) | 60 (17.3) | 40 (11.4) |
Progressive Disease/Relapse, n (%) | 165 (47.7) | 183 (52.1) |
Palliative Hormonal Therapy, n (%) | 30 (8.7) | 35 (10.0) |
Prior Systemic Therapies Received by Setting a n (%) | ||
Neoadjuvant/adjuvant only | 125 (36.1) | 133 (37.9) |
Primary therapy | 55 (15.9) | 35 (10.0) |
Progressive disease/relapse only | 90 (26.0) | 92 (26.0) |
Treatment in both neoadjuvant/adjuvant and PD/relapse setting | 71 (20.5) | 86 (24.5) |
Not Applicable | 5 (1.4) | 5 (1.4) |
Interval from End of Most Recent Therapy to First Dose (months) | ||
Patients with data, n | 342 | 325 |
Median (range) | 4.8 (0 to 74) | 5.5 (0 to 100) |
History of Prior Hysterectomy, n (%) | 252 (72.8) | 279 (79.5) |
History of Prior External Beam Radiotherapy, n (%) | 155 (44.8) | 159 (45.3) |
History of Prior Brachytherapy | 88 (25.4) | 97 (27.6) |
ITT = intention to treat; n = number of patients with event; N = total number of patients; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
aDoes not include the therapeutic setting of palliative hormonal therapy.
Source: Clinical Study Report.19
Table 30: Patients With Specific Concomitant Medications (Incidence > 0% in 1 or More Treatment Groups) (APaT Population)
Characteristics | KEYNOTE-775 | |
|---|---|---|
PEM + LEN (N=342) | TPC (N=325) | |
Patients in population, N | 342 | 325 |
With 1 or more concomitant medications, n (%) | 341 (99.7) | 322 (99.1) |
Antineoplastic And Immunomodulating Drugs, n (%) | ||
|||||||||||||||||||||||||||||||||| | ||||||||||||||||| | ||||||||||||||||| |
Antineoplastic Drugs | 1 (0.3) | 0 (0.0) |
Arsenic Trioxide | 1 (0.3) | 0 (0.0) |
Bevacizumab | 0 (0.0) | 1 (0.3) |
Calcium Folinate; fluorouracil; oxaliplatin | 0 (0.0) | 1 (0.3) |
Capecitabine | 1 (0.3) | 1 (0.3) |
Capecitabine; oxaliplatin | 0 (0.0) | 1 (0.3) |
Carboplatin | 4 (1.2) | 2 (0.6) |
Celecoxib | 13 (3.8) | 8 (2.5) |
Cisplatin | 4 (1.2) | 1 (0.3) |
Clarithromycin | 6 (1.8) | 11 (3.4) |
Clarithromycin lactobionate | 2 (0.6) | 0 (0.0) |
Cyclophosphamide | 13 (3.8) | 10 (3.1) |
Cyclophosphamide; fluorouracil; methotrexate | 1 (0.3) | 0 (0.0) |
Docetaxel | 5 (1.5) | 3 (0.9) |
Doxorubicin | 9 (2.6) | 7 (2.2) |
Doxorubicin Hydrochloride | 3 (0.9) | 0 (0.0) |
Epirubicin | 1 (0.3) | 4 (1.2) |
Epirubicin Hydrochloride | 1 (0.3) | 0 (0.0) |
Everolimus | 0 (0.0) | 1 (0.3) |
Fluorouracil | 5 (1.5) | 5 (1.5) |
Gemcitabine | 1 (0.3) | 1 (0.3) |
Lapatinib | 1 (0.3) | 0 (0.0) |
Lenvatinib | 0 (0.0) | 1 (0.3) |
Methotrexate | 2 (0.6) | 0 (0.0) |
Mitomycin | 1 (0.3) | 0 (0.0) |
Oxaliplatin | 1 (0.3) | 0 (0.0) |
Paclitaxel | 5 (1.5) | 3 (0.9) |
Paclitaxel albumin | 1 (0.3) | 0 (0. 0) |
Pegylated liposomal doxorubicin hydrochloride | 1 (0.3) | 0 (0.0) |
Pembrolizumab | 1 (0.3) | 1 (0.3) |
Rituximab | 1 (0.3) | 0 (0.0) |
Tegafur; uracil | 1 (0.3) | 0 (0.0) |
Trastuzumab | 2 (0.6) | 1 (0.3) |
Tretinoin | 1 (0.3) | 0 (0.0) |
|||||||||||||||||||||||||||||||||| | ||||||||||||||||| | ||||||||||||||||| |
Immunostimulants | 10 (2.9) | 113 (34.8) |
Immunosuppressants | 7 (2.0) | 1 (0.3) |
APaT = all patients as treated; n = number of patients with event; N = total number of patients; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
Source: Clinical Study Report.19
Table 31: Summary of Subsequent Systemic Anticancer Treatment Patients (ITT Population)
Characteristic | KEYNOTE-775 | |
|---|---|---|
PEM + LEN (N=346) | TPC (N=351) | |
Started Study Treatment, n (%) | 342 (98.8) | 325 (92.6) |
Discontinued Study Treatment, n (%) | 247 (71.4) | 238 (67.8) |
Received Any Subsequent Systemic Anticancer Therapy, n (%) | 109 (31.5) | 176 (50.1) |
Subsequent systemic therapy by type, n (%) | ||
Chemotherapy | 92 (26.6) | 119 (33.9) |
bortezomib | 1 (0.3) | 0 (0.0) |
capecitabine | 2 (0.6) | 0 (0.0) |
carboplatin | 30 (8.7) | 47 (13.4) |
cisplatin | 8 (2.3) | 23 (6.6) |
cyclophosphamide | 6 (1.7) | 10 (2.8) |
docetaxel | 3 (0.9) | 10 (2.8) |
doxorubicin | 55 (15.9) | 16 (4.6) |
epirubicin | 2 (0.6) | 1 (0.3) |
etoposide | 0 (0.0) | 1 (0.3) |
fluorouracil | 1 (0.3) | 1 (0.3) |
gemcitabine | 14 (4.0) | 34 (9.7) |
ifosfamide | 0 (0.0) | 1 (0.3) |
melphalan | 0 (0.0) | 1 (0.3) |
mitoxantrone | 0 (0.0) | 1 (0.3) |
oxaliplatin | 3 (0.9) | 2 (0.6) |
paclitaxel | 33 (9.5) | 0 (14.2) |
tegafur | 0 (0.0) | 1 (0.3) |
topotecan | 3 (0.9) | 3 (0.9) |
vinorelbine | 0 (0.0) | 1 (0.3) |
Hormonal therapy | 24 (6.9) | 51 (14.5) |
Any PD-1 or PD-L1 checkpoint | 4 (1.2) | 42 (12.0) |
durvalumab | 0 (0.0) | 1 (0.3) |
nivolumab | 0 (0.0) | 3 (0.9) |
pembrolizumab | 4 (1.2) | 38 (10.8) |
Targeted therapy | 8 (2.3) | 12 (3.4) |
abemaciclib | 1 (0.3) | 0 (0.0) |
adavosertib | 0 (0.0) | 1 (0.3) |
afatinib | 0 (0.0) | 1 (0.3) |
everolimus | 4 (1.2) | 5 (1.4) |
MAK 683 | 1 (0.3) | 0 (0.0) |
olaparib | 0 (0.0) | 4 (1.1) |
palbociclib | 1 (0.3) | 0 (0.0) |
temsirolimus | 2 (0.6) | 1 (0.3) |
Any VEGF/VEGFR inhibitor | 10 (2.9) | 43 (12.3) |
Bevacizumab | 7 (2.0) | 14 (4.0) |
lenvatinib | 3 (0.9) | 32 (9.1) |
Subsequent lenvatinib and pembrolizumab | 3 (0.9) | 32 (9.1) |
Subsequent systemic therapy by lines | ||
1 subsequent line | 6 (1.7) | 11 (3.1) |
2 subsequent lines | 81 (23.4) | 134 (38.2) |
>=3 subsequent lines | 55 (15.9) | 78 (22.2) |
ITT = intention to treat; n = number of patients with event; N = total number of patients; PEM + LEN = pembrolizumab + lenvatinib combination; pMMR = mismatch repair proficient; TPC = treatment of physician’s choice (doxorubicin or paclitaxel).
Source: Clinical Study Report.19
Table 32: Summary of Interim and Final Analysis Strategy
Analyses | Key outcomes | Timing | Estimated Time after First Patient Randomized | Primary Purpose of Analysis |
|---|---|---|---|---|
IA1 | PFS OS | Both approximately 368 OS events and at least 6 months after last participant randomized | Approximately 27 months | Final PFS analysis Interim OS analysis |
IA2 | OS | Both approximately 463 OS events and at least 12 months after last participant randomized | Approximately 35 months | Interim OS analysis |
FA | OS | Both approximately 526 OS events and at least 18 months after last participant randomizeda | Approximately 43 months a | Final OS analysis |
FA = final analysis; IA1 = interim analysis 1; IA2 = interim analysis 2; OS = overall survival; PFS = progression-free survival; pMMR = mismatch repair proficient.
aNote that if events accrue slower than expected for the FA, the sponsor may conduct the analysis up to 3 months after the estimated timing of the FA (i.e., approximately 46 months after first participant randomized).
Source: Clinical Study Report.19
Figure 7: Multiplicity Graph for Type I Error Control of Study Hypotheses
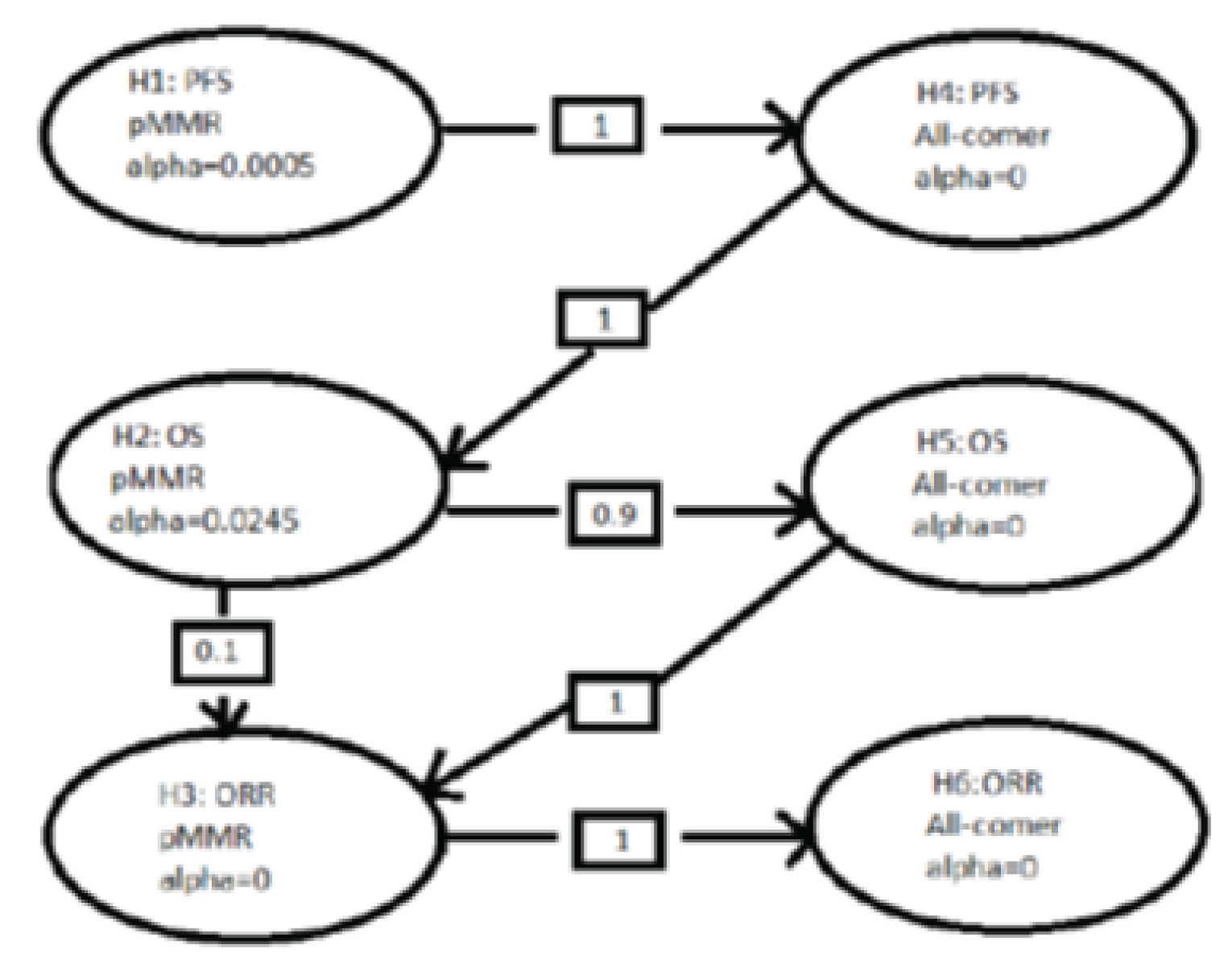
ORR = objective response rate; OS = overall survival; PFS = progression-free survival; pMMR = mismatch repair proficient.
Source: Clinical Study Report.19
Table 33: Censoring Rules for Primary Analysis of Progression-Free Survival Based on RECIST 1.1
Situation | Primary Analysis | Sensitivity Analysis 1 | Sensitivity Analysis 2 |
|---|---|---|---|
PD or death documented after ≤ 1 missed disease assessment, and before new anticancer therapy, if any | Progressed at date of documented PD or death | Progressed at date of documented PD or death | Progressed at date of documented PD or death |
Death or progression immediately after ≥ 2 consecutive missed disease assessments, or after new anticancer therapy | Censored at last disease assessment prior to the earlier date of ≥ 2 consecutive missed disease assessment and new anti- cancer therapy, if any | Progressed at date of documented PD or death | Progressed at date of documented PD or death |
No PD and no death; and new anticancer treatment is not initiated | Censored at last disease assessment | Censored at last disease assessment | Progressed at treatment discontinuation due to reasons other than complete response; otherwise censored at last disease assessment if still on study treatment or completed study treatment |
No PD and no death; new anticancer treatment is initiated | Censored at last disease assessment before new anticancer treatment | Censored at last disease assessment | Progressed at date of new anticancer treatment if new anticancer treatment is initiated; otherwise progressed at treatment discontinuation if treatment is discontinued due to reasons other than complete response; otherwise censored at last disease assessment if still on study therapy or completed the study therapy |
PD = progressive disease; RECIST = Response Evaluation Criteria in Solid Tumours.
Source: Clinical Study Report.19
Table 34: Summary of Important Protocol Deviations (ITT Population)
Category | PEM + LEN | TPC | Total |
|---|---|---|---|
Patients in population, N | 411 | 351 | 697 |
With 1 or more important protocol deviations, n (%) | 9 (2.6) | 8 (2.3) | 17 (2.4) |
Discontinuation Criteria, n (%) | 3 (0.9) | 3 (0.9) | 6 (0.9) |
Participant developed study intervention discontinuation criteria but was not discontinued from study intervention. | 2 (0.6) | 3 (0.9) | 6 (0.9) |
Participant developed trial specific discontinuation criteria but was not discontinued from the trial. | 1 (0.3) | 0 (0.0) | 1 (0.1) |
Prohibited Medications, n (%) | 0 (0.0) | 1 (0.3) | 1 (0.1) |
Concurrent anticancer therapies such as chemotherapy, targeted therapies (e.g., tyrosine kinase inhibitors), hormonal therapy directed at EC, radiotherapy (with the exception of palliative radiotherapy as specified in Section 6.5.1), antitumour interventions (surgical resection, surgical debulking of tumour, and so on), live vaccines (within 30 days) or concurrent investigational therapies, while on treatment or before study entry during screening unless allowed per protocol. | 0 (0.0) | 1 (0.3) | 1 (0.1) |
Study Intervention, n (%) | 7 (2.0) | 4 (1.1) | 11 (1.6) |
Participant was administered improperly stored study intervention that was deemed unacceptable for use. | 6 (1.7) | 3 (0.9) | 9 (1.3) |
Participant was dispensed study intervention other than what was assigned in the allocation schedule, i.e., incorrect medication or potential cross treatment. | 1 (0.3) | 1 (0.3) | 2 (0.3) |
ITT = intention to treat; n = number of patients with event; N = total number of patients; PEM + LEN = = pembrolizumab + lenvatinib combination; pMMR= mismatch repair proficient; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
Source: Clinical Study Report.19
Table 35: Study Population for Lenvatinib Plus Pembrolizumab, Doxorubicin, and Paclitaxel
Category | KEYNOTE-775 | |||
|---|---|---|---|---|
PEM + LEN | Doxorubicin | Paclitaxel | Total | |
Number of Patients Screened, N | 1,178 | |||
Number of Patients Randomized (ITT), N | 346 | 254 | 97 | 697 |
Number of Patients Received Treatment (Actual Treatment) (APaT)a n (%) | 342 (98.9) | 239 (94.0) | 86 (88.7) | 667 |
Number of Patients Randomized and Did not Receive Treatment, n (%) | 4 (1.1) | 16 (6.0) | 10 (10.3) | 30 |
APaT = all patients as treated; ITT = intention to treat; PEM + LEN = pembrolizumab + lenvatinib combination; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
aIncludes 1 participant in the doxorubicin column for whom the investigator site selected paclitaxel prior to randomization, but was actually treated with doxorubicin.
Source: Clinical Study Report.19
Table 36: Summary of Drug Exposure (APaT Population)
Category | KN=775 | |
|---|---|---|
PEM + LEN (N=342) | TPC (N=325) | |
Duration On Therapy (days)a | ||
Median (Range) | 219.5 (1.0 to 817.0) | 106.0 (1.0 to 785.0) |
Duration on Both Lenvatinib and Pembrolizumab (days)b | ||
Median (Range) | 187.0 (1.0 to 784.0) | N A |
Duration on Lenvatinib (days)c | ||
Median (Range) | 202.5 (1.0 to 817.0) | N A |
Duration on Pembrolizumab (days)d | ||
Median (Range) | 206.0 (1.0 to 784.0) | N A |
APaT = all patients as treated; NA = Not applicable; PEM + LEN = pembrolizumab + lenvatinib combination; pMMR = mismatch repair proficient; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
aDuration on Therapy is calculated as the days between first dose date and last dose date in each treatment arm.
bFor lenvatinib + pembrolizumab, defined as from the first date when both drugs were taken until the date when 1 of the 2 drugs was first discontinued.
cFor lenvatinib defined as from the first date when lenvatinib was taken until the date when lenvatinib was discontinued.
dFor pembrolizumab, defined as from the first date when pembrolizumab was taken until the date when pembrolizumab was discontinued.
Source: Clinical Study Report.19
Table 37: Patients With Adverse Events Resulting in Treatment Discontinuation (Incidence > 1% in 1 or More Treatment Groups) (APaT Population)
AEs | KEYNOTE-775 | ||||
|---|---|---|---|---|---|
PEM + LEN | TPC | ||||
PEM or LEN DC | PEM and LEN DCa | LEN DCb | PEM DCc | ||
Patients in population, N | 342 | 342 | 342 | 342 | 325 |
With 1 or more AEs, n (%) | 106 (31.0) | 43 (12.6) | 97 (28.4) | 60 (17.5) | 27 (8.3) |
Hypertension | 7 (2.0) | 0 (0.0) | 7 (2.0) | 0 (0.0) | 0 (0.0) |
Asthenia | 6 (1.8) | 1 (0.3) | 6 (1.8) | 1 (0.3) | 1 (0.3) |
Diarrhea | 6 (1.8) | 1 (0.3) | 4 (1.2) | 3 (0.9) | 1 (0.3) |
Decreased appetite | 5 (1.5) | 3 (0.9) | 5 (1.5) | 3 (0.9) | 0 (0.0) |
Alanine aminotransferase increased | 4 (1.2) | 1 (0.3) | 1 (0.3) | 4 (1.2) | 0 (0.0) |
Proteinuria | 4 (1.2) | 0 (0.0) | 4 (1.2) | 0 (0.0) | 0 (0.0) |
Vomiting | 4 (1.2) | 0 (0.0) | 4 (1.2) | 0 (0.0) | 0 (0.0) |
APaT = all patients as treated; DC = discontinuation; LEN = lenvatinib; n = number of patients with event; N = total number of patients included in the analysis; PEM + LEN = pembrolizumab + lenvatinib combination; PEM= pembrolizumab; pMMR = mismatch repair proficient; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
aDiscontinuation of lenvatinib and pembrolizumab due to the same adverse event.
bDiscontinuation of lenvatinib, regardless of action taken for pembrolizumab.
cDiscontinuation of pembrolizumab, regardless of action taken for lenvatinib.
Source: Clinical Study Report.19
Table 38: Patients With Adverse Events Resulting in Treatment Interruption (Incidence > 3% in 1 or More Treatment Groups) (APaT Population)
AEs | PEM + LEN | TPC | |||
|---|---|---|---|---|---|
LEN or PEM Interruption | LEN and PEM Interruptiona | LEN Interruptionb | PEM Interruptionc | ||
Patients in population, N | 342 | 342 | 342 | 342 | 325 |
With 1 or more adverse events, n (%) | 235 (68.7) | 100 (29.2) | 199 (58.2) | 165 (48.2) | 91 (28.0) |
Diarrhea | 46 (13.5) | 17 (5.0) | 37 (10.8) | 28 (8.2) | 0 (0.0) |
Hypertension | 44 (12.9) | 6 (1.8) | 39 (11.4) | 12 (3.5) | 0 (0.0) |
Proteinuria | 28 (8.2) | 2 (0.6) | 22 (6.4) | 8 (2.3) | 0 (0.0) |
Decreased appetite | 23 (6.7) | 2 (0.6) | 17 (5.0) | 8 (2.3) | 0 (0.0) |
Vomiting | 19 (5.6) | 4 (1.2) | 17 (5.0) | 6 (1.8) | 2 (0.6) |
Alanine aminotransferase increased | 18 (5.3) | 9 (2.6) | 12 (3.5) | 15 (4.4) | 1 (0.3) |
Fatigue | 16 (4.7) | 3 (0.9) | 12 (3.5) | 8 (2.3) | 2 (0.6) |
Nausea | 16 (4.7) | 1 (0.3) | 12 (3.5) | 5 (1.5) | 0 (0.0) |
Aspartate aminotransferase increased | 13 (3.8) | 8 (2.3) | 8 (2.3) | 13 (3.8) | 0 (0.0) |
Asthenia | 12 (3.5) | 4 (1.2) | 8 (2.3) | 8 (2.3) | 2 (0.6) |
Palmar-plantar erythrodysesthesia syndrome | 12 (3.5) | 1 (0.3) | 7 (2.0) | 7 (2.0) | 0 (0.0) |
Abdominal pain | 11 (3.2) | 3 (0.9) | 10 (2.9) | 4 (1.2) | 1 (0.3) |
Weight decreased | 11 (3.2) | 3 (0.9) | 9 (2.6) | 5 (1.5) | 1 (0.3) |
APaT = all patients as treated; LEN = lenvatinib; n = number of patients with event; N = total number of patients included in the analysis; PEM + LEN = pembrolizumab + lenvatinib combination; PEM= pembrolizumab; pMMR = mismatch repair proficient; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
aInterruption of lenvatinib and pembrolizumab due to the same adverse event.
bInterruption of lenvatinib, regardless of action taken for pembrolizumab.
cInterruption of pembrolizumab, regardless of action taken for lenvatinib.
Source: Clinical Study Report.19
Table 39: Patients With Adverse Events Resulting in Dose Reduction (Incidence > 2% in 1 or More Treatment Groups) (APaT Population)
AEs | KEYNOTE-775 | |
|---|---|---|
PEM + LEN (N=342) | TPC(N=325) | |
Patients in population, N | 342 | 3 25 |
With 1 or more adverse events, n (%) | 229 (67.0) | 42 (12.9) |
Hypertension | 60 (17.5) | 0 (0.0) |
Diarrhea | 39 (11.4) | 1 (0.3) |
Palmar-plantar erythrodysesthesia syndrome | 30 (8.8) | 0 (0.0) |
Proteinuria | 24 (7.0) | 0 (0.0) |
Fatigue | 23 (6.7) | 3 (0.9) |
Decreased appetite | 21 (6.1) | 1 (0.3) |
Asthenia | 18 (5.3) | 2 (0.6) |
Weight decreased | 17 (5.0) | 0 (0.0) |
Arthralgia | 15 (4.4) | 0 (0.0) |
Nausea | 15 (4.4) | 0 (0.0) |
Platelet count decreased | 11 (3.2) | 0 (0.0) |
Stomatitis | 9 (2.6) | 2 (0.6) |
Vomiting | 8 (2.3) | 1 (0.3) |
Thrombocytopenia | 7 (2.0) | 2 (0.6) |
APaT = all patients as treated; n = number of patients with event; N = total number of patients included in the analysis; PEM + LEN = pembrolizumab + lenvatinib combination; pMMR = mismatch repair proficient; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
Source: Clinical Study Report.19
Table 40: Summary of Dose Reduction on Lenvatinib (APaT Population)
Category | KEYNOTE-775 |
|---|---|
PEM + LEN (N=342) | |
Patients in population, N | 342 |
Number of Patients with a Dose Reduction, N (%) | 245 (71.6) |
Number of Dose Reductions, n (%) | |
0 | 97 (28.4) |
1 | 93 (27.2) |
2 | 88 (25.7) |
3 | 43 (12.6) |
4 | 21 (6.1) |
Time to First Dose Reduction (Months) | |
N | 245 |
Median (Range) | 1.9 (0.1 to 22.8) |
APaT = all patients as treated; n = number of patients with event; N = total number of patients included in the analysis; PEM + LEN = pembrolizumab + lenvatinib combination; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
Source: Clinical Study Report.19
Table 41: Administration of Lenvatinib (APaT Population)
Category | KEYNOTE-775 |
|---|---|
PEM + LEN | |
Patients in population | 342 |
Dose Intensity (mg/day)a | |
N | 342 |
Median (Range), mg/day | 13.8 (3 to 20) |
Percentage of planned dose received (%)b | |
N | 342 |
Median (Range), % | 68.9 (16 to 100) |
APaT = all patients as treated; PEM + LEN = pembrolizumab + lenvatinib combination.
aDose Intensity (mg/day) = total dose received during the study/ (end date of last dose of study drug – start date of first dose of study drug + 1).
bReceived dose as percentage of planned starting dose (%) = dose intensity (mg/day)/planned daily dose(mg/day) x 100.
Source: Clinical Study Report.19
Table 42: Administration of Pembrolizumab, Doxorubicin, and Paclitaxel (APaT Population)
Dose cycles | KEYNOTE-775 | ||
|---|---|---|---|
PEM + LEN | TPC | ||
Pembrolizumab | Doxorubicin | Paclitaxel | |
Patients in population | 342 | 239 | 86 |
Number of Cycles Received | |||
Median (Range) | 10.0 (1 to 35) | 5.0 (1 to 10) | 6.0 (1 to 27) |
ITT = intention to treat; PEM + LEN = pembrolizumab + lenvatinib combination; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
Source: Clinical Study Report.19
Table 43: Summary of Follow-Up Duration (ITT Population)
Range | PEM + LEN (N=346) | TPC (N=325) | Total (N=697) |
|---|---|---|---|
Median (Range) | 12.0 (0.3, 26.9) | 10.8 (0.3, 26.3) | 11.4 (0.3, 26.9) |
ITT = intention to treat; PEM + LEN = pembrolizumab + lenvatinib combination; pMMR = mismatch repair proficient; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
aFollow-up duration is defined as the time from randomization to the date of death or the database cut-off date if the participant is still alive.
Source: Clinical Study Report.19
Table 44: Analysis of Overall Survival (ITT Population)
Outcomes | KEYNOTE-775 | |
|---|---|---|
PEM + LEN (N= 346) | TPC (N= 351) | |
OS (ITT) | ||
Events, n (%) | 165 (47.7) | 203 (57.8) |
Person-month | 4,128.6 | 3,564.8 |
Event Rate 100 Person- months | 4.0 | 5.7 |
Median OS (95% CI), mo.a | 17.4 (14. 2 to 1 9.9) | 12.0 (10.8 to 13.3) |
HRb (95% CI)b | 0.68 (0.56 to 0.84) | |
P valuec | 0.0001c | |
12-month OS Rates (95% CI), %a | 61.6 (56.1 to 66.6) | 49.5 (43.8 to 55.0) |
CI= confidence interval; HR = hazard ratio; ITT = intention to treat; OS=overall survival; PEM + LEN = pembrolizumab + lenvatinib combination; pMMR = mismatch repair proficient; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
aFrom product-limit (Kaplan-Meier) method for censored data.
bBased on Cox regression model with the Efron method of tie handling with treatment as a covariate stratified by ECOG Performance Status, geographic region, and prior history of pelvic radiation.
cOne-sided P value based on log-rank test stratified by ECOG Performance Status, geographic region, and prior history of pelvic radiation.
Note: Analysis includes 1 participant who was stratified with a dMMR status, but actually had a pMMR status; stratification factors for this participant are derived from actual ECOG Performance Status, geographic region, and prior history of pelvic radiation.
Source: Clinical Study Report.19
Table 45: Summary of Overall Survival Rate Over Time (ITT Population)
OS | KEYNOTE-775 | |
|---|---|---|
PEM + LEN (N=346) | TPC (N=351) | |
Summary of OS rate at time point, % (95% CI)a | ||
6 months | 82.9 (78.5 to 86.5) | 77.9 (73.1 to 81.9) |
12 months | 61.6 (56.1 to 66.6) | 49.5 (43.8 to 55.0) |
18 months | 48.2 (41.9 to 54.3) | 29.2 (23.1 to 35.5) |
24 months | 37.2 (29.5 to 45.0) | 21.5 (13.9 to 30.1) |
CI= confidence interval; ITT = intention to treat; OS=overall survival; PEM + LEN = pembrolizumab + lenvatinib combination; pMMR = mismatch repair proficient; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
aFrom product-limit (Kaplan-Meier) method for censored data.
Source: Clinical Study Report.19
Table 46: OS by Subgroup Factors Point Estimate and Nominal 95% CI (ITT Population)
Subgroups | PEM + LEN(N=346) | TPC(N=351) | PEM + LEN vs. TPC | ||
|---|---|---|---|---|---|
N | Number of Events (%) | N | Number of Events (%) | HR (95% CI)a | |
Overall primary analysis (ITT) | 346 | 165 (47.7) | 351 | 203 (57.8) | 0.68 (0.56 to 0.84) |
Subgroup analysis | |||||
ECOG Performance Status | |||||
0 | 212 | 82 (38.7) | 207 | 114 (55.1) | 0.56 (0.42 to 0.75) |
1 | 133 | 82 (61.7) | 144 | 89 (61.8) | 0.87 (0.64 to 1.18) |
Histology | |||||
Endometrioid | 188 | 76 (40.4) | 198 | 91 (46.0) | 0.78 (0.57 to 1.05) |
Non-endometrioid | 158 | 89 (56.3) | 153 | 112 (73.2) | 0.56 (0.42 to 0.74) |
Prior Lines of Therapyb | |||||
1 | 244 | 114 (46.7) | 226 | 140 (61.9) | 0.61 (0.47 to 0.78) |
2 | 92 | 46 (50.0) | 114 | 56 (49.1) | 0.88 (0.59 to 1.30) |
≥3 | 10 | 5 (50.0) | 11 | 7 (63.6) | 0.75 (0.24 to 2.37) |
CI = confidence interval; HR = hazard ratio; ITT = intention to treat; OS = overall survival; PEM + LEN = pembrolizumab + lenvatinib combination; pMMR = mismatch repair proficient; TPC = treatment physician´s choice of doxorubicin or paclitaxel; vs. = versus.
aBased on Cox regression model with the Efron method of tie handling with treatment as a covariate. Subgroup analyses were conducted using an unstratified Cox model.
bThe # of prior lines in this table included the prior lines of neoadjuvant and adjuvant adjunct anticancer therapy (Refer to Table 18). One of the inclusion criteria was radiographic evidence of disease progression after 1 prior systemic, platinum-based chemotherapy regimen. Note: there is no restriction regarding prior hormonal therapy. One of the exclusion criteria was patient had received greater than 1 prior systemic chemotherapy regimen (other than adjuvant or neoadjuvant) for endometrial carcinoma. Patients may have received up to 2 regimens of platinum-based chemotherapy in total, as long as 1 was given in the neoadjuvant or adjuvant treatment setting. (Refer to Table 6)
Source: Clinical Study Report.19
Table 47: Analysis of Progression-Free Survival (Primary Censoring Rule, ITT Population)
Outcomes | KEYNOTE-775 | |
|---|---|---|
PEM + LEN (N= 346) | TPC (N= 351) | |
Events, n (%) | 247 (71.4) | 238 (67.8) |
Person-month | 2,538.0 | 1,458.8 |
Event Rate 100 Person- months | 9.7 | 16.3 |
Median PFS (95% CI), mo.a | 6.6 (5.6 to 7.4) | 3.8 (3.6 to 5.0) |
HRb (95% CI)b | 0.60 (0.50 to 0.72) | |
P valuec | < 0.0001c | |
6-month PFS rates (95% CI), %a | 52.1 (46.5 to 57.3) | 36.2 (30.5 to 41.9) |
CI= confidence interval; HR = hazard ratio; ITT = intention to treat; PEM + LEN = pembrolizumab + lenvatinib combination; PFS=progression-free survival; pMMR = mismatch repair proficient; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
aFrom product-limit (Kaplan-Meier) method for censored data.
bBased on Cox regression model with the Efron method of tie handling with treatment as a covariate stratified by ECOG Performance Status, geographic region, and prior history of pelvic radiation.
cOne-sided P value based on log-rank test stratified by ECOG Performance Status, geographic region, and prior history of pelvic radiation.
Note: Analysis includes 1 participant who was stratified with a dMMR status, but actually had a pMMR status; stratification factors for this participant are derived from actual ECOG Performance Status, geographic region, and prior history of pelvic radiation.
Source: Clinical Study Report.19
Table 48: Summary of Progression-Free Survival Rate Over Time (Primary Censoring Rule, ITT Population)
PFS | KEYNOTE-775 | |
|---|---|---|
PEM + LEN (N=346) | TPC(N=351) | |
Summary of Progression-Free Survival rate at time point, % (95% CI)a | ||
6 months | 52.1 (46.5 to 57.3) | 36.2 (30.5 to 41.9) |
12 months | 27.6 (22.5 to 32.8) | 13.1 (8.9 to 18.3) |
18 months | 21.1 (16.3 to 26.3) | 6.6 (3.0 to 12.1) |
24 months | 16.8 (11.8 to 22.4) | 3.3 (0.5 to 11.4) |
CI= confidence interval; ITT = intention to treat; PEM + LEN = pembrolizumab + lenvatinib combination; PFS=progression-free survival; pMMR = mismatch repair proficient; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
aFrom product-limit (Kaplan-Meier) method for censored data.
Source: Clinical Study Report.19
Table 49: Progression-Free Survival by Subgroup
Analysis | KEYNOTE-775 | ||||
|---|---|---|---|---|---|
PEM + LEN(N=346) | TPC(N=351) | PEM + LEN vs. TPC | |||
N | Number of Events (%) | N | Number of Events (%) | HR (95% CI)a | |
Overall primary analysis (ITT) | 346 | 247 (71.4) | 351 | 238 (67.8) | 0.60 (0.50 to 0.72) |
Subgroup analysis | |||||
ECOG Performance Status | |||||
0 | 212 | 149 (70.3) | 207 | 137 (66.2) | 0.57 (0.45 to 0.72) |
1 | 133 | 98 (73.7) | 144 | 101 (70.1) | 0.65 (0.49 to 0.86) |
Histology | |||||
Endometrioid | 188 | 122 (64.9) | 198 | 131 (66.2) | 0.59 (0.46 to 0.76) |
Non-endometrioid | 158 | 125 (79.1) | 153 | 107 (69.9) | 0.56 (0.43 to0.73) |
Prior Lines of Therapyb | |||||
1 | 244 | 177 (72.5) | 226 | 163 (72.1) | 0.52 (0.42 to 0.65) |
2 | 92 | 67 (72.8) | 114 | 72 (63.2) | 0.74 (0.53 to 1.04) |
≥3 | 10 | 3 (30.0) | 11 | 3 (27.3) | 0.60 (0.12 to 3.07) |
CI= confidence interval; HR = hazard ratio; ITT = intention to treat; PEM + LEN = pembrolizumab + lenvatinib combination; PFS=progression-free survival; pMMR = mismatch repair proficient; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
aBased on Cox regression model with the Efron method of tie handling with treatment as a covariate. Subgroup analyses were conducted using an unstratified Cox model.
bThe # of prior lines in this table included the prior lines of neoadjuvant and adjuvant adjunct anticancer therapy (refer to Table 18). One of the inclusion criteria was radiographic evidence of disease progression after 1 prior systemic, platinum-based chemotherapy regimen. Note: there was no restriction regarding prior hormonal therapy. One of the exclusion criteria was patient who had received greater than 1 prior systemic chemotherapy regimen (other than adjuvant or neoadjuvant) for endometrial carcinoma. Patients may have received up to 2 regimens of platinum-based chemotherapy in total, as long as 1 was given in the neoadjuvant or adjuvant treatment setting. (Refer to Table 6)
Source: Clinical Study Report.19
Table 50: Sensitivity Analysis of Progression-Free Survival (Sensitivity Censoring Rule 1, ITT Population)
Outcomes | KEYNOTE-775 | |
|---|---|---|
PEM + LEN (N= 346) | TPC (N= 351) | |
PFS (ITT) | ||
Events, n (%) | 264 (76.3) | 284 (80.9) |
Person-month | 2,651.4 | 1,807.6 |
Event Rate 100 Person- months | 10.0 | 15.7 |
Median PFS (95% CI), mo.a | 6.6 (5.6 to 7.4) | 4.1 (3.7 to 5.6) |
HRb (95% CI)b | 0.62 (0.53 to 0.74) | |
P valuec | < 0.0001c | |
6-month PFS rates (95% CI), %a | 52.3 (46.8 to 57.5) | 40.2 (34.8 to 45.5) |
CI= confidence interval; HR = hazard ratio; ITT = intention to treat; PEM + LEN = pembrolizumab + lenvatinib combination; PFS=progression-free survival; pMMR = mismatch repair proficient; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
aFrom product-limit (Kaplan-Meier) method for censored data.
bBased on Cox regression model with the Efron method of tie handling with treatment as a covariate stratified by ECOG Performance Status, geographic region, and prior history of pelvic radiation.
cOne-sided P value based on log-rank test stratified by ECOG Performance Status, geographic region, and prior history of pelvic radiation.
Note: Analysis includes 1 participant who was stratified with a dMMR status, but actually had a pMMR status; stratification factors for this participant are derived from actual ECOG Performance Status, geographic region, and prior history of pelvic radiation.
Source: Clinical Study Report.19
Table 51: Sensitivity Analysis of Progression-Free Survival (Sensitivity Censoring Rule 2, ITT Population)
Outcomes | KEYNOTE-775 | |
|---|---|---|
PEM + LEN (N= 346) | TPC (N= 351) | |
PFS (ITT) | ||
Events, n (%) | 285 (82.4) | 330 (94.0) |
Person-month | 2,653.8 | 1,784.6 |
Event Rate 100 Person- months | 10.7 | 18.5 |
Median PFS (95% CI), mo.a | 6.1 (5.6, 7.4) | 3.8 (3.6, 5.0) |
HRb (95% CI)b | 0.56 (0.48, 0.66) | |
P valuec | < 0.0001c | |
6-month PFS rates (95% CI), %a | 50.9 (45.5, 56.0) | 36.2 (31.1, 41.3) |
CI= confidence interval; HR = hazard ratio; ITT = intention to treat; PEM + LEN = pembrolizumab + lenvatinib combination; PFS=progression-free survival; pMMR = mismatch repair proficient; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
aFrom product-limit (Kaplan-Meier) method for censored data.
bBased on Cox regression model with the Efron method of tie handling with treatment as a covariate stratified by ECOG Performance Status, geographic region, and prior history of pelvic radiation.
cOne-sided P value based on log-rank test stratified by ECOG Performance Status, geographic region, and prior history of pelvic radiation.
Note: Analysis includes 1 participant who was stratified with a dMMR status, but actually had a pMMR status; stratification factors for this participant are derived from actual ECOG Performance Status, geographic region, and prior history of pelvic radiation.
Source: Clinical Study Report.19
Figure 8: Empirical Mean Change From Baseline and 95% CI for the EORTC QLQ-C30 GHS/QoL Over Time by Treatment Group (FAS)
CI= confidence interval; FAS = full analysis set; ITT = intention to treat; PEM + LEN= pembrolizumab + lenvatinib combination; PFS=progression-free survival; pMMR = mismatch repair proficient; PRO = patient-reported outcome; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
Source: Clinical Study Report.19
Figure 9: Empirical Mean Change From Baseline and 95% CI for the EORTC QLQ-C30 Physical Functioning Scale Score Over Time by Treatment Group (FAS)
CI= confidence interval; FAS = full analysis set; PEM + LEN= pembrolizumab + lenvatinib combination; pMMR = mismatch repair proficient; PRO = patient-reported outcome; SD = Standard deviation; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
Source: Clinical Study Report.19
Table 52: Analysis of Change From Baseline in EORTC QLQ-C30 Physical Functioning Scale Score to Week 12 (FAS)
Outcomes | KEYNOTE-775 | |
|---|---|---|
PEM + LEN (N= 346) | TPC (N= 351) | |
EORTC QLQ-C30 Physical Functioning scale score (FAS) | ||
Baseline | ||
N | 316 | 298 |
Mean (SD) | 79.56 (19.21) | 76.58 (20.85) |
At week 12 | ||
N | 256 | 192 |
Mean (SD) | 71.46 (21.64) | 72.81 (21.13) |
Change from Baseline to Week 12 | ||
N | 327 | 310 |
LS mean (95% CI) | –10.42 (–12.65 to –8.19) | –10.42 (–12.65 to –8.19) |
LSM Difference (95% CI)a | –1.74 (–4.99 to 1.51) | |
P value | P = 0.2931 | |
CI= confidence interval; FAS = full analysis set; LSM = least squares mean; PEM + LEN= pembrolizumab + lenvatinib combination; pMMR = mismatch repair proficient; PRO = patient-reported outcome; SD = Standard deviation; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
aBased on a cLDA model with the PRO scores as the response variable with covariates for treatment by study visit interaction, stratification factors MMR status, ECOG Performance Status, geographic region, and prior history of pelvic radiation.
Note: For baseline and week 12, N is the number of patients in each treatment group with non-missing assessments at the specific time point; for change from baseline, N is the number of patients in the analysis population in each treatment group.
Source: Clinical Study Report.19
Table 53: Analysis of Change From Baseline in EORTC QLQ-EN24 Urological Symptoms Score to Week 12 (pMMR, FAS)
Outcomes | KEYNOTE-775 | |
|---|---|---|
PEM + LEN (N= 346) | TPC (N= 351) | |
Baseline | ||
N | 268 | 249 |
Mean (SD) | 14.89 (18.09) | 16.13 (19.79) |
At week 12 | ||
N | 217 | 157 |
Mean (SD) | 12.37 (18.29) | 16.56 (19.29) |
Change from Baseline to Week 12 | ||
N | 282 | 266 |
LS mean (95% CI) | –2.20 (–4.28 to –0.12) | 0.78 (–1.55 to 3.11) |
LSM Difference (95% CI)a | –2.98 (–5.96 to -0.00) | |
P value | P = 0.0496 | |
CI= confidence interval; FAS = full analysis set; LSM = least squares mean; PEM + LEN= pembrolizumab + lenvatinib combination; pMMR = mismatch repair proficient; PRO = patient-reported outcome; SD = Standard deviation; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
aBased on a cLDA model with the PRO scores as the response variable with covariates for treatment by study visit interaction, stratification factors MMR status, ECOG Performance Status, geographic region, and prior history of pelvic radiation.
Note: For baseline and week 12, N is the number of patients in each treatment group with non-missing assessments at the specific time point; for change from baseline, N is the number of patients in the analysis population in each treatment group.
Source: Clinical Study Report.19
Table 54: Summary of Best Overall Response (ITT Population)
Response Evaluation | KEYNOTE-775 | |||
|---|---|---|---|---|
PEM + LEN (N = 346) | TPC (N = 351) | |||
N | % (95% CI)a | N | % (95% CI)a | |
CR | 18 | 5.2 (3.1 to 8.1) | 9 | 2.6 (1.2 to 4.8) |
PR | 87 | 25.1 (20.7 to 30.1) | 44 | 12.5 (9.3 to 16.5) |
Objective response (CR+PR) | 105 | 30.3 (25.5 to 35.5) | 53 | 15.1 (11.5 to 19.3) |
Stable Disease (SD) | 168 | 48.6 (43.2 to 54.0) | 139 | 39.6 (34.4 to 44.9) |
Disease Control [CR+PR+(SD ≥ 7 Weeks)] | 248 | 71.7 (66.6 to 76.4) | 163 | 46.4 (41.1 to 51.8) |
Clinical Benefit [CR+PR+(SD ≥ 23 Weeks)] | 165 | 47.7 (42.3 to 53.1) | 85 | 24.2 (19.8 to 29.0) |
Progressive Disease | 54 | 15.6 (11.9 to 19.9) | 108 | 30.8 (26.0 to 35.9) |
Not Evaluable (NE) | 2 | 0.6 (0.1 to 2.1) | 7 | 2.0 (0.8 to 4.1) |
No Assessment (NA) | 17 | 4.9 (2.9 to 7.8) | 44 | 12.5 (9.3 to 16.5) |
CI= confidence interval; CR = complete response; ITT = intention to treat; PEM + LEN= pembrolizumab + lenvatinib combination; pMMR = mismatch repair proficient; PR = partial response; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
aBased on binomial exact CI method.
Note: NE: Post-baseline assessment(s) available, but not evaluable. No Assessment: No post-baseline assessment available for response evaluation. For best overall response of CR and PR, only confirmed responses are included.
Source: Clinical Study Report.19
Figure 10: Empirical Mean Change From Baseline and 95% CI for the EORTC QLQ-EN24 Urological Symptoms Score Over Time by Treatment Group (FAS)
CI= confidence interval; FAS = full analysis set; PEM + LEN= pembrolizumab + lenvatinib combination; pMMR = mismatch repair proficient; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
Source: Clinical Study Report.19
Figure 11: Empirical Mean Change From Baseline and 95% CI for the EQ-5D VAS Score Over Time by Treatment Group (FAS)
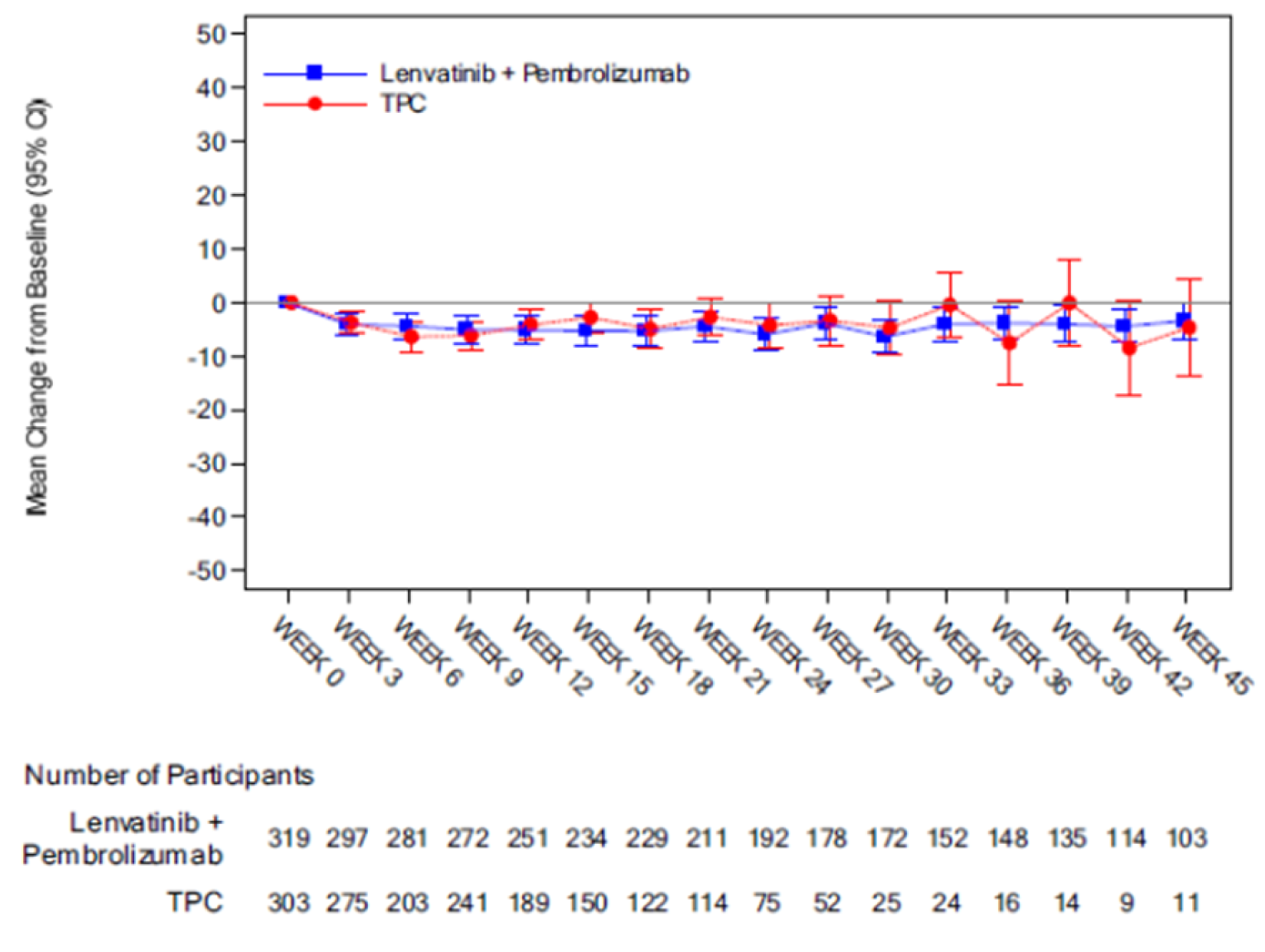
CI= confidence interval; FAS = full analysis set; PEM + LEN= pembrolizumab + lenvatinib combination; pMMR = mismatch repair proficient; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
Source: Clinical Study Report.19
Table 55: Objective Response Rate (Confirmed) by Subgroups Point Estimate and Nominal 95% CI (ITT Population)
Subgroup | PEM + LEN (N=346) | TPC (N=351) | Difference in % | ||
|---|---|---|---|---|---|
N | Number of Responses (ORR %) | N | Number of Responses (ORR %) | ||
Overall primary analysis (ITT) | 346 | 105 (30.3) | 351 | 53 (15.1) | 15.2 (9.1 to 21.4) |
Subgroup analysis, n (%) | |||||
ECOG Status | |||||
0 | 212 | 67 (31.6) | 207 | 35 (16.9) | 14.7 (6.5 to 22.7) |
1 | 133 | 38 (28.6) | 144 | 18 (12.5) | 16.1 (6.7 to 25.6) |
Histology | |||||
Endometrioid | 188 | 56 (29.8) | 198 | 36 (18.2) | 11.6 (3.1 to 20.1) |
Non-endometrioid | 158 | 49 (31.0) | 153 | 17 (11.1) | 19.9 (11.1 to 28.7) |
Prior Lines of Therapy | |||||
1 | 244 | 76 (31.1) | 226 | 30 (13.3) | 17.9 (10.5 to 25.2) |
2 | 92 | 27 (29.3) | 114 | 21 (18.4) | 10.9 (–0.7 to 22.8) |
≥3 | 10 | 2 (20.0) | 11 | 2 (18.2) | 1.8 (–33.7 to 38.2) |
CI= confidence interval; ECOG = Eastern Cooperative Oncology Group; ITT = intention to treat; n = number of patients with event; ORR=objective response rate; PEM + LEN= pembrolizumab + lenvatinib combination; pMMR = mismatch repair proficient; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
aORR difference and 95% CI is based on the Miettinen & Nurminen method. Subgroup analyses were conducted using the unstratified M & N method.
bThe number of prior lines in this table included the prior lines of neoadjuvant and adjuvant adjunct anticancer therapy (refer to Table 18). One of the inclusion criteria was radiographic evidence of disease progression after 1 prior systemic, platinum-based chemotherapy regimen. Note: there is no restriction regarding prior hormonal therapy. One of the exclusion criteria was patient had received greater than 1 prior systemic chemotherapy regimen (other than adjuvant or neoadjuvant) for endometrial carcinoma. Patients may have received up to 2 regimens of platinum-based chemotherapy in total, as long as 1 was given in the neoadjuvant or adjuvant treatment setting. (Refer to Table 6)
Note: Response is based on best overall response using BICR assessment per RECIST 1.1 with confirmation.
Source: Clinical Study Report.19
Table 56: Summary of Time to Response and Duration of Response in Patients With Confirmed Response (ITT Population)
Category | KEYNOTE-775 | |
|---|---|---|
PEM + LEN(N=346) | TPC (N=351) | |
Number of patients with responsea | 105 | 53 |
Time to Response (months) | ||
Median (Range) | 2.1 (1.5 to 9.4) | 3.5 (1.0 to 7.4) |
Response Durationb (months) | ||
Median (Range) | 9.2 (1.6+ to 23.7+) | 5.7 (0.0+ to 24.2+) |
Number (%b) of Patients with Extended Response Duration: | ||
≥6 months | 55 (65.6) | 14 (42.1) |
≥12 months | 27 (42.3) | 5 (32.8) |
≥18 months | 8 (35.5) | 1 (16.4) |
≥24 months | 0 (NR) | 1 (16.4) |
ITT = intention to treat; n = number of patients with event; NR = not reported; ORR=objective response rate; PEM + LEN= pembrolizumab + lenvatinib combination; pMMR = mismatch repair proficient; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
aIncludes patients with complete response or partial response
bFrom product-limit (Kaplan-Meier) method for censored data.
Note: "+" indicates there is no progressive disease by the time of last disease assessment.
Source: Clinical Study Report.19
Figure 12: Kaplan-Meier Estimates of Duration of Response in Patients With Confirmed Response (ITT Population)
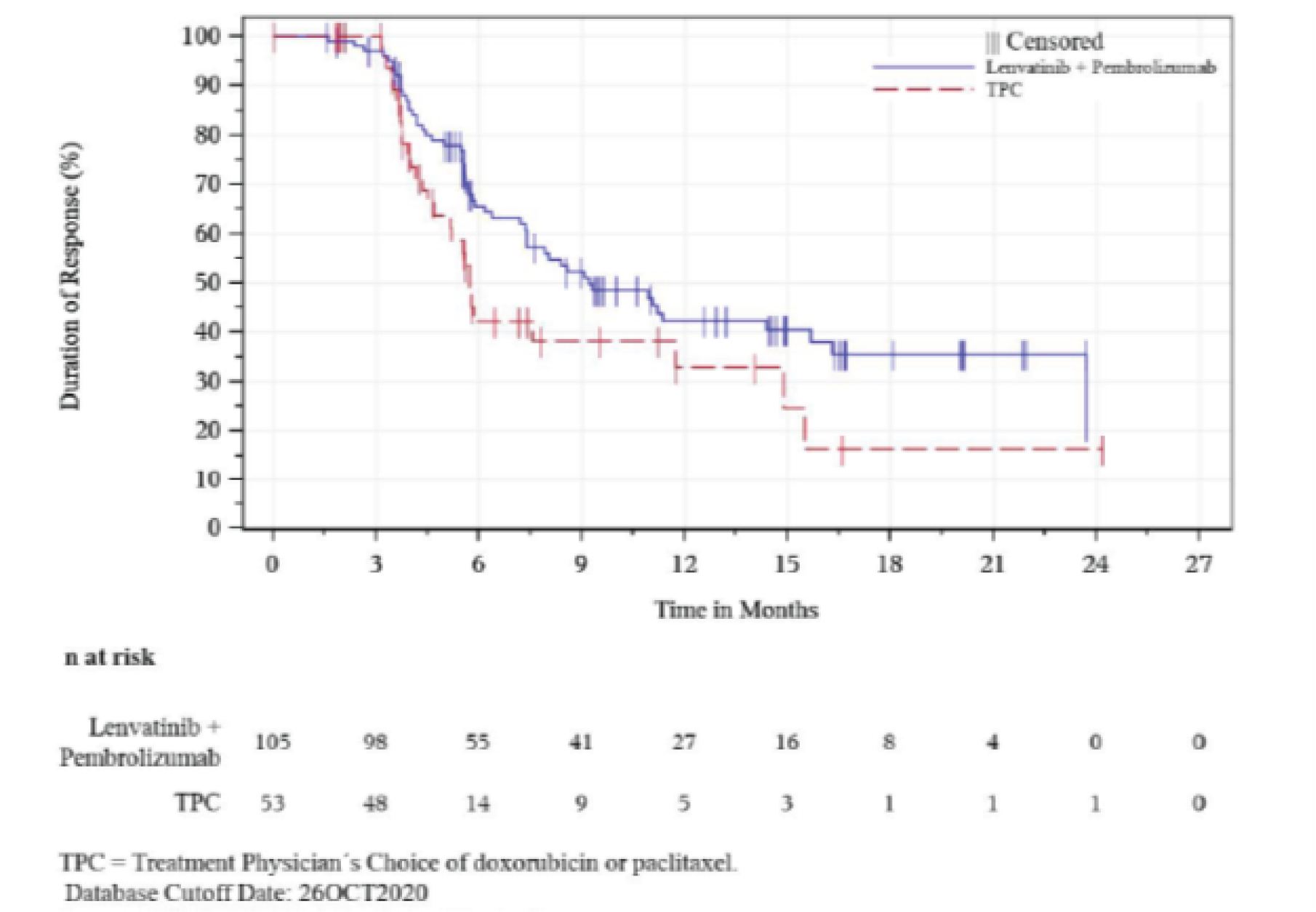
BICR = Blinded Independent Central Review; CI= confidence interval; ITT = intention to treat; PEM + LEN= pembrolizumab + lenvatinib combination; = TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
Source: Clinical Study Report.19
Table 57: Patients With Adverse Events (Incidence ≥ 30% in 1 or More Treatment Groups, APaT Population)
AEs | KEYNOTE-775 | |
|---|---|---|
PEM + LEN (N=342) | TPC (N=325) | |
With 1 or more adverse events, n (%) | 341 (99.7) | 324 (99.7) |
Hypertension | 224 (65.5) | 17 (5.2) |
Hypothyroidism | 189 (55.3) | 3 (0.9) |
Diarrhea | 187 (54.7) | 64 (19.7) |
Nausea | 169 (49.4) | 154 (47.4) |
Decreased appetite | 151 (44.2) | 67 (20.6) |
Vomiting | 125 (36.5) | 67 (20.6) |
Weight decreased | 117 (34.2) | 18 (5.5) |
Fatigue | 113 (33.0) | 92 (28.3) |
Arthralgia | 108 (31.6) | 28 (8.6) |
Anemia | 83 (24.3) | 159 (48.9) |
APaT = All participants as treated; AE = adverse event; PEM + LEN= pembrolizumab + lenvatinib combination; pMMR = mismatch repair proficient; SAE=serious adverse event; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
Note: Every participant is counted a single time for each applicable row and column. A specific AE appears on this report only if its incidence in 1 or more of the columns meets the incidence criterion in the report title, after rounding. The AEs are ordered decreasingly by the incidence in the first column. MedDRA preferred terms "Neoplasm progression," "Malignant neoplasm progression" and "Disease progression" not related to the drug are excluded. Non-serious AEs up to 30 days of last dose and SAEs up to 120 days of last dose are included.
Source: Clinical Study Report.19
Table 58: Exposure-Adjusted Adverse Event Summary (Including Multiple Occurrences of Events) (APaT Population)
AEs | KEYNOTE-775 | |
|---|---|---|
# of AEs (Rate -Events/100 person-months)a | ||
PEM + LEN (N=342) | TPC(N=325) | |
Number of Patients exposed, N | 406 | 388 |
Total exposureb in person-months | 3,174.26 | 1,510.54 |
Total events (rate) | ||
with 1 or more AEs | 7,534 (237.35) | 3,853 (255.07) |
with SAEs | 312 (9.83) | 142 (9.40) |
with dose modificationc due to an AE | 1,249 (39.35) | 279 (18.47) |
with dose interruptiond due to an AE | 702 (22.12) | 171 (11.32) |
interruption of pembrolizumab | 372 (11.72) | 0 (0.00) |
interruption of lenvatinib | 523 (16.48) | 0 (0.00) |
interruption of both pembrolizumab and lenvatinib | 193 (6.08) | 0 (0.00) |
with dose reductione due to an AE | 506 (15.94) | 72 (4.77) |
Death due to AEs | 16 (0.50) | 15 (0.99) |
discontinuede due to an AE | 158 (4.98) | 36 (2.36) |
discontinued Pembrolizumab | 81 (2.55) | 0 (0.00) |
discontinued Lenvatinib | 128 (4.03) | 0 (0.00) |
discontinued both Pembrolizumab and Lenvatinib | 51 (1.61) | 0 (0.00) |
discontinued due to a SAE | 76 (2.39) | 11 (0.73) |
discontinued Pembrolizumab | 48 (1.51) | 0 (0.00) |
discontinued Lenvatinib | 67 (2.11) | 0 (0.00) |
discontinued both Pembrolizumab and Lenvatinib | 39 (1.23) | 0 (0.00) |
APaT = All participants as treated; AE = adverse event; PEM + LEN= pembrolizumab + lenvatinib combination; pMMR = mismatched repair proficient; SAE=serious adverse event; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
aEvent rate per 100 person-months of exposure = event count *100/person-months of exposure.
bDrug exposure is defined as the between the first-dose date + 1 day and the earlier of the last dose date + 30 or the database cut-off date.
cDefined as an action taken of dose reduced, drug interrupted, or drug withdrawn.
dFor lenvatinib + pembrolizumab, the dose interruption of either pembrolizumab or lenvatinib.
eFor lenvatinib + pembrolizumab, the dose reduction for only lenvatinib.
Source: Clinical Study Report.19
Table 59: Patients With Serious Adverse Events by Decreasing Incidence (Incidence ≥ 1% in 1 or More Treatment Groups, APaT Population)
AEs | KEYNOTE-775 (pMMR) | |
|---|---|---|
PEM + LEN (N=342) | TPC(N=325) | |
With 1 or more SAE, n (%) | 170 (49.7) | 94 (28.9) |
Hypertension | 15 (4.4) | 0 (0.0) |
Urinary tract infection | 11 (3.2) | 2 (0.6) |
Acute kidney injury | 8 (2.3) | 3 (0.9) |
Colitis | 7 (2.0) | 1 (0.3) |
Decreased appetite | 7 (2.0) | 0 (0.0) |
Pyrexia | 7 (2.0) | 3 (0.9) |
Diarrhea | 6 (1.8) | 3 (0.9) |
Vomiting | 6 (1.8) | 2 (0.6) |
Dehydration | 4 (1.2) | 1 (0.3) |
Ileus | 4 (1.2) | 0 (0.0) |
Intestinal obstruction | 4 (1.2) | 2 (0.6) |
Pulmonary embolism | 4 (1.2) | 5 (1.5) |
Febrile neutropenia | 2 (0.6) | 13 (4.0) |
Anemia | 1 (0.3) | 6 (1.8) |
Neutropenia | 1 (0.3) | 7 (2.2) |
APaT = All participants as treated; AE = adverse event; PEM + LEN= pembrolizumab + lenvatinib combination; pMMR = mismatch repair proficient; SAE=serious adverse event; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
Every participant is counted a single time for each applicable row and column. A specific AE appears on this report only if its incidence in 1 or more of the columns meets the incidence criterion in the report title, after rounding. The AEs are ordered decreasingly by the incidence in the first column. MedDRA preferred terms "Neoplasm progression," "Malignant neoplasm progression" and "Disease progression" not related to the drug are excluded. SAEs up to 120 days of last dose are included.
Source: Clinical Study Report.19
Table 60: Patients With Adverse Events Resulting in Death (Incidence > 0% in 1 or More Treatment Groups, APaT Population)
AEs | KEYNOTE-775 | |
|---|---|---|
PEM + LEN (N=342) | TPC (N=325) | |
Patients in population, N | 342 | 325 |
With 1 or more adverse events, n (%) | 16 (4.7) | 15 (4.6) |
Cardiac disorders | 2 (0.6) | 3 (0.9) |
Acute myocardial infarction | 1 (0.3) | 0 (0.0) |
Cardiac failure congestive | 0 (0.0) | 1 (0.3) |
Cardiogenic shock | 0 (0.0) | 1 (0.3) |
Right ventricular dysfunction | 1 (0.3) | 0 (0.0) |
Toxic cardiomyopathy | 0 (0.0) | 1 (0.3) |
Gastrointestinal disorders | 4 (1.2) | 0 (0.0) |
Colitis | 1 (0.3) | 0 (0.0) |
Intestinal perforation | 1 (0.3) | 0 (0.0) |
Lower gastrointestinal hemorrhage | 1 (0.3) | 0 (0.0) |
Malignant gastrointestinal obstruction | 1 (0.3) | 0 (0.0) |
General disorders and administration site conditions | 3 (0.9) | 4 (1.2) |
Death | 2 (0.6) | 3 (0.9) |
Multiple organ dysfunction syndrome | 1 (0.3) | 1 (0.3) |
Infections and infestations | 2 (0.6) | 5 (1.5) |
Influenza | 0 (0.0) | 1 (0.3) |
Pneumonia | 2 (0.6) | 2 (0.6) |
Sepsis | 0 (0.0) | 2 (0.6) |
Injury, poisoning, and procedural complications | 0 (0.0) | 1 (0.3) |
Subdural hematoma | 0 (0.0) | 1 (0.3) |
Metabolism and nutrition disorders | 1 (0.3) | 0 (0.0) |
Decreased appetite | 1 (0.3) | 0 (0.0) |
Neoplasms benign, malignant, and unspecified (incl cysts and polyps) | 1 (0.3) | 0 (0.0) |
Myelodysplastic syndrome | 1 (0.3) | 0 (0.0) |
Psychiatric disorders | 1 (0.3) | 0 (0.0) |
Assisted suicide | 1 (0.3) | 0 (0.0) |
Renal and urinary disorders | 1 (0.3) | 0 (0.0) |
Acute kidney injury | 1 (0.3) | 0 (0.0) |
Respiratory, thoracic, and mediastinal disorders | 1 (0.3) | 2 (0.6) |
Aspiration | 0 (0.0) | 1 (0.3) |
Pulmonary embolism | 1 (0.3) | 1 (0.3) |
APaT = All participants as treated; AE = adverse event; PEM + LEN= pembrolizumab + lenvatinib combination; pMMR = mismatch repair proficient; SAE=serious adverse event; TPC = Treatment Physician´s Choice of doxorubicin or paclitaxel.
Note: Every participant is counted a single time for each applicable row and column. SAEs up to 120 days of last dose are included. MedDRA preferred terms "Neoplasm progression," "Malignant neoplasm progression" and "Disease progression" not related to the drug are excluded.
Source: Clinical Study Report.19
Table 61: Concomitant Medications Used in pMMR (KEYNOTE-146 Trial)
Characteristics | PEM + LEN (N=94) |
|---|---|
Antineoplastic and immunomodulating drugs (i.e., methotrexate) n, (%) | 1 (1.1) |
Immunosuppressants, n, (%) | 1 (1.1) |
Corticosteroids, for systemic use, Plain, n, (%) | 28 (29.8) |
n = # of patients with events; N = total # of patients included in the analysis; PEM + LEN= pembrolizumab + lenvatinib combination; pMMR = mismatch repair proficient.
Source: Clinical Study Report.61
Figure 13: Kaplan-Meier Plot of Duration of Response (pMMR, KEYNOTE-146 Trial)
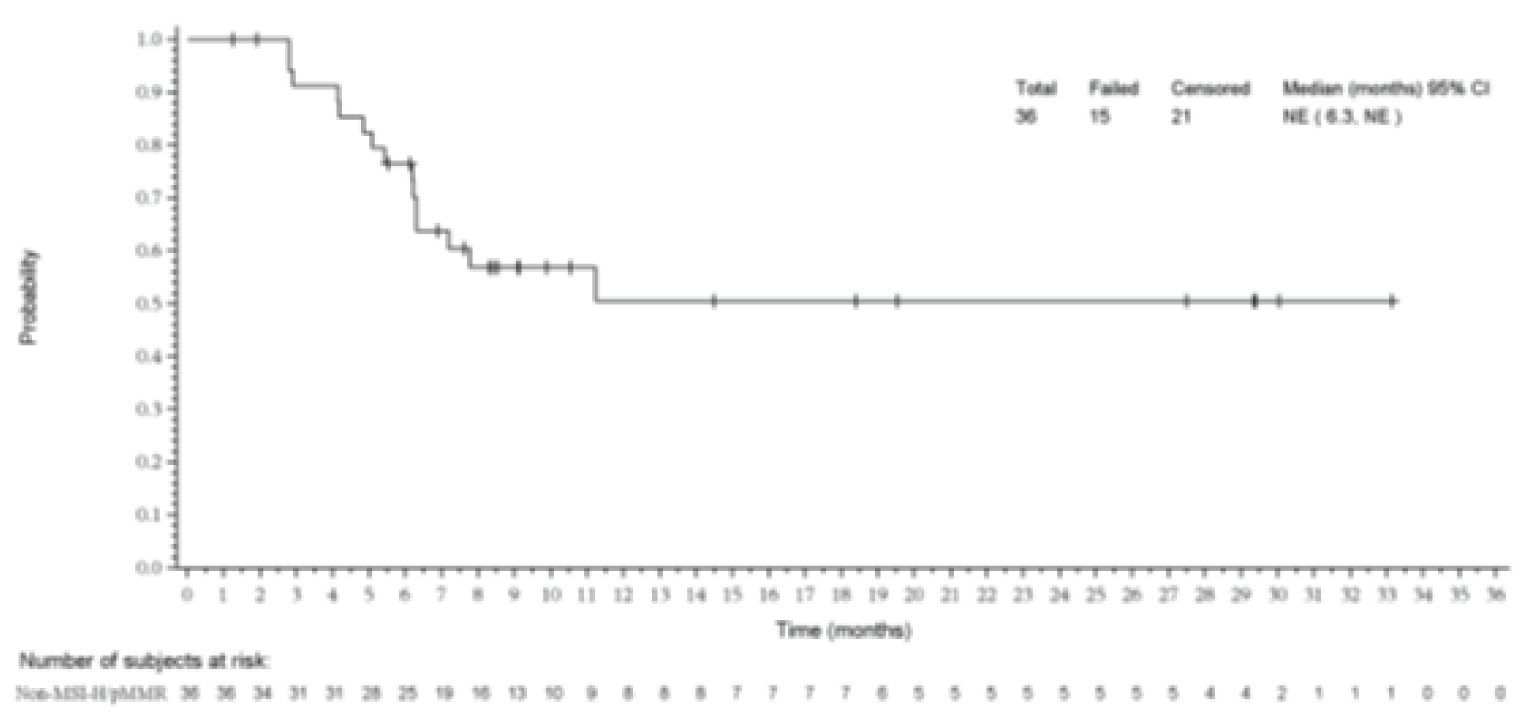
NE = not estimable, + =censored.
Note: Duration of response among responders is defined as the time from the date that a confirmed response was first documented as the evidence of CR or PR until the date of the first documentation of disease progression or date of death from any cause, whichever occurs first. The median is estimated using Kaplan-Meier method, and the 95% confidence interval is constructed with a generalized Brookmeyer and Crowley method. All tumour assessments are considered as long as the patients did not start a new anticancer therapy.
Source: Clinical Study Report.61
Table 62: Adverse Events in Patients With pMMR Tumours in the KEYNOTE-146 Trial (Safety Analysis Set)
AEs | PEM + LEN (N=94) |
|---|---|
TEAE (occurred in >20% patients), n (%) | |
Hypertension | 60 (63.8) |
Diarrhea | 59 (62.8) |
Fatigue | 51 (54.3) |
Decreased appetite | 48 (51.1) |
Hypothyroidism | 46 (48.9) |
Nausea | 45 (47.9) |
Stomatitis | 34 (36.2) |
Vomiting | 37 (39.4) |
Weight loss | 34 (36.2) |
Arthralgia | 31 (33.0) |
Headache | 31 (33.0) |
Constipation | 30 (31.9) |
Urinary tract infection | 27 (28.7) |
Dysphonia | 27 (28.7) |
Hypomagnesemia | 25 (26.6) |
Abdominal pain | 25 (26.6) |
Palmar-plantar erythrodysesthesia syndrome | 24 (25.5) |
Dyspnea | 23 (24.5) |
Cough | 20 (21.3) |
Myalgia | 20 (21.3) |
SAEs (>5%) n, % | |
Hypertension | 7 (7.4) |
abdominal pain | 5 (5.3) |
WDAE (>2%) n, % | |
Muscular weakness | 2 (2.1) |
Pancreatitis | 2 (2.1) |
AE = adverse event; n = number of patients with event; N = total number of patients included in the analysis; PEM + LEN= pembrolizumab + lenvatinib combination; pMMR = mismatch repair proficient; SAE = serious adverse event; TEAE = treatment-emergent adverse event; WDAE = withdrawals due to adverse event.
Source: Clinical Study Report.61
Appendix 4: Description and Appraisal of Outcome Measures
Note that this appendix has not been copy-edited.
Aim
To describe the following outcome measures and review their measurement properties (validity, reliability, responsiveness to change, and MID):
EORTC QLQ-C30 version 3.0
EQ-5D-5L version 2.0
EORTC QLQ-EN24
Findings
Table 63: Summary of Outcome Measures and Their Measurement Properties
Outcome Measure | Type | Conclusions about Measurement Properties | MID |
|---|---|---|---|
EORTC QLQ-C30 | A self-reported instrument designed to measure the physical, psychological, and social functions of patients with cancers. The EORTC QLQ-C30 incorporates 5 functional scales (Physical, Role, Cognitive, Emotional and Social); 3 symptom scales (Fatigue; Pain and Nausea and vomiting), a global health and quality of life scale; and single items that assess additional symptoms commonly reported by patients with cancer (dyspnea, appetite loss, sleep disturbance, constipation, and diarrhea), as well as the perceived financial impact of the disease and treatment.52-54 | Validity: Construct, criterion, and discriminate validity were demonstrated in patients with ovarian and gestational trophoblastic disease cancers.70,71 The EORTC QLQ-30 was found to adequately assess the effect of expected toxicities on patients’ HRQoL during and following treatment.72 No reported validation studies were found for patients with EC. Reliability: Minimum reliability with Cronbach alpha > 0.70 was met in 7 of 9 subscales in patients with gestational trophoblastic disease, ovarian cancer, and other types of gynecological cancers.71 Responsiveness: There were no data available for responsiveness. | Among patients with various advanced cancers73,74:
No reported MID was found for patients with EC. |
EQ-5D-5L | EQ-5D-5L is a generic, preference-based HRQoL questionnaire consisting of an index score and VAS score. The index score is based on 5 dimensions: mobility, self-care, usual activities, pain/discomfort, and anxiety/depression. Score ranges from 0 (“dead”) to 1 (“perfect health”) where negative scores represent “worse than dead.” The EQ VAS ranges from 0 (worst health imaginable) to 100 (best health imaginable). | Measurement properties have not been assessed in patients with endometrial cancer. | VAS: MID estimates for patients with various cancers (not including endometrial cancer) are 8 – 11 based on ECOG PS (0.5 SD = 9), 7 - 10 based on FACT-G (0.5 SD = 9).55 |
EORTC QLQ-EN24 | A 24-item self-reported questionnaire for patients at all stages of endometrial cancer. The EORTC QLQ-EN24 consists of 10 symptom scales (lymphedema, urological symptoms, gastrointestinal symptoms, poor body image, sexual/vaginal problems, pain in the back and pelvis, tingling/numbness, muscular pain, hair loss, and taste change), and 3 functional scales (sexual interest, activity, and enjoyment). | Validity: Among patients with endometrial cancer, EORTC QLQ-EN24 demonstrated good convergent and discriminant validity, compared with the EORTC QLQ 30.75-77 Reliability: Test-retest reliability was observed for all multi-item scales (range from 0.81 to 0.92) and the single-item questions (range from 0.66 to 0.97).76 For urological symptoms scale, good test-retest reliability was shown (Cronbach alpha 0.92).76 Good internal consistency was demonstrated for 5 multi-item scales (Cronbach alpha range from 0.74 to 0.86)75-77 For urological symptoms scale, internal consistency ranged from 0.75 to 0.86.75-77 Responsiveness: There were no data available for responsiveness. | Not identified |
CIN = cervical intraepithelial neoplasia; EC = endometrial cancer; EORTC QLQ-C30 = European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30; EQ VAS: EQ visual analogue scale; EORTC QLQ-EN24: European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Endometrial Cancer Module; HPV: human papillomavirus; HRQoL: health-related quality of life ICC: inter-class correlation; MID = minimal important difference
European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30
Description
The EORTC QLQ-C30 is a self-reported instrument designed to measure the physical, psychological, and social functions of patients with cancers.54 The EORTC QLQ-C30 consists of 30 items that are scored to create 5 multi-item functional scales, 3 multi-item symptom scales, 6 single-item symptom scales, and 2 global QoL scales (Table 55) Version 3.0 of the questionnaire is the most current version and has been in use since December of 1997.54,78 It is intended for use in the adult population only.54
Table 64: EORTC QLQ-C30 Scales
Functional Scales (15 Questions) | Symptom Scales (7 Questions) | Single-Item Symptom Scales (6 Questions) | Global Quality of Life (2 Questions) |
|---|---|---|---|
Physical function (5) | Fatigue (3) | Dyspnea (1) | Global Quality of Life (2) |
Role function (2) | Pain (2) | Insomnia (1) | — |
Cognitive function (2) | Nausea and vomiting (2) | Appetite loss (1) | — |
Emotional function (4) | — | Constipation (1) | |
Social function (2) | — | Diarrhea (1) | — |
— | — | Financial impact (1) | — |
Scoring
The EORTC QLQ-C30 uses a 1-week recall period to assess function and symptoms.54 Twenty-eight questions are scored on a 4-point Likert scale (1: not at all; 2: a little; 3: quite a bit; 4: very much). The 2 questions that make up the global HRQoL scale are scored on a 7-point Likert scale with anchors at 1 (“very poor”) and 7 (“excellent”).
Raw scores for each scale are computed as the average of the items that contribute to a particular scale.54 This scaling approach is based on the assumption that it is appropriate to provide equal weighting to each item that comprises a scale. There is also an assumption that, for each item, the interval between response options is equal (for example, the difference in score between “not at all” and “a little” is the same as “a little” and “quite a bit,” at a value of 1 unit). Each raw scale score is converted to a standardized score that ranges from 0 to 100 using a linear transformation, with a higher score reflecting better function on the function scales, higher symptoms on the symptom scales, and better HRQoL (i.e., higher scores simply reflect higher levels of response on that scale). Thus, a decline in score on the symptom scale would reflect an improvement, whereas an increase in score on the function and HRQoL scales would reflect an improvement. According to the EORTC QLQ-C30 scoring algorithm, if there are missing items for a scale (i.e., the participant did not provide a response), the score for the scale can still be computed if there are responses for at least half of the items. In calculating the scale score, the missing items are simply ignored — an approach that assumes that the missing items have values equal to the average of those items for what the respondent completed.
Psychometric Properties
The EORTC QLQ-C30 was originally validated in patients with lung cancer and head and neck cancer from various European and North American countries, as well as from Australia.52,79,80 The scales of the EORTC QLQ-C30 have been found to assess distinct components of HRQoL; distinguishing between patients with different PS and degrees of weight loss, and responsive to change over time.52,79,80
A literature search was conducted to identify validation information of the EORTC QLQ-C30 in patients with endometrial cancer and none were identified. Of note, there is a validated version of the EORTC QLQ that was developed specific for endometrial cancer: EORTC QLQ-EN24.76 A validation study of the Mexican-Spanish version of the EORTC QLQ-EN24 in 189 patients with endometrial cancer included a brief validation of the QLQ-C30.75 The study confirmed the internal consistency and reliability of the QLQ-C30 and found that its convergent and discriminant validity (Cronbach alpha range = 0.77 to 0.89) was consistent with its original report.75
In a validation study of the EORTC QLQ-C30, patients with breast cancer (n = 121), ovarian cancer (n = 111) lung cancer (n = 160) and a heterogenous group of other cancers (n = 121) completed the questionnaire before and on day 8 of chemotherapy.70 The item-domain correlations of the EORTC QLQ-C30 were not different across the primary tumour sites (i.e., breast, ovary, or lung). The correlations for all items, except for item 5 (whether the responders needed help with eating, dressing, washing, or using the toilet) and the physical function domain (r = -0.3), were highly correlated within their own domain than with any other domains (r = -0.65 to 0.95). At day 8 of chemotherapy, the item-domain for item 5 and the physical function domain was 0.49 for the entire group and ranged from -0.38 for ovarian cancer to -0.55 for breast cancer. These higher values at day 8 suggest that item 5 was more relevant in the week after chemotherapy than before chemotherapy.70 Similarly, items asking about vomiting showed a higher correlation with domains for nausea/vomiting on day 8 after chemotherapy (r = 0.89) than before chemotherapy (r = 0.74). The questionnaire also demonstrated good internal consistency for most domains at baseline and at day 8 (Cronbach alpha > 0.70). However, values were <0.70 at baseline and day 8 for role function (0.66 and 0.53, respectively) and cognitive function (0.63 and 0.58, respectively). Examination of the discrimination of the domain scores according to metastatic disease (i.e., individuals with distant metastases or individuals with disease confined to the primary site and regional lymph nodes) found No significant differences in mean scores of emotional function, cognitive function, and nausea/vomiting at baseline and day 8. However, the mean scores for each of the other domains differed between the groups at baseline, with patients with localized disease tending to have better physical function, role function, social function, less fatigue and pain, and better global HRQoL. At day 8, only social function and fatigue were shown to be no longer significant. Patients with lung and ovarian cancer reported lower scores for all these domains, with patients with ovarian cancer reporting the lowest scores for all domains. After chemotherapy, many of the differences seen at baseline between the groups were no longer evident. At day 8, patients with ovarian cancer had the smallest magnitude of change, with no change in role function, social function, or global HRQoL, while being the only cancer group reporting a significant improvement in pain.70
An analysis of data from a Canadian randomized controlled trial of paclitaxel and cisplatin versus cyclophosphamide and cisplatin in the treatment of 153 patients with epithelial ovarian cancer found the EORTC QLQ-C30 adequately assessed the effect of expected toxicities on patients HRQoL during and after treatment.72 At baseline, prior to the initiation of treatment, there was close agreement in the “mild or none” category between the symptoms recorded on case report forms and paired EORTC QLQ-C30 questions, calculated by dividing 2 times the number of patients classified in the mild or none category with both toxicity and QoL assessments, with the sum of patients classified as mild or none based on toxicity and QoL assessment only. The greatest degree of agreement ranged between 0.80 (95% CI: 0.75 to 0.86) to 0.98 (95% CI: 0.92 to 0.99). The pairing of lethargy with Question 18, and mood with Question 22 were slightly weaker in agreement compared to the other pairs at 0.72 and 0.73, respectively. The weakest pairs were constipation with Question 16, and lethargy with Question 18 at 0.44 and 0.44, respectively. During treatment and at the end of cycles 3 and 6, all but 1 symptom and HRQoL pairs demonstrated marked agreement ranging from 0.71 to 0.93. The 1 exception was the pair assessing symptom hair loss and Question 42 with a degree of agreement of 0.50 and 0.37 at cycles 3 and 6, respectively. A regression model predicting global HRQoL scores based on baseline grades of the most frequently observed toxicities and scores corresponding to HRQoL question found that the questions related to motor weakness (question 12), anorexia (question 13), mood (question 24), gastrointestinal pain (question 40) and vomiting (question 15) explained 60% of the variance in baseline global HRQoL on the EROTC QLQ-C30. When patients were off chemotherapy, 78% of symptoms and HRQoL pairs had high levels of agreement (> 0.80).
Minimal Important Difference
A literature search was conducted to identify the MID of the EORTC QLQ-C30 in patients with endometrial cancer and none were identified. Following is a summary of the MID of the EORTC QLQ-C30 in patients with cancer in general.
Change in the EORTC QLQ-C30 may be interpreted in terms of small, moderate, or large changes in HRQoL.74 A study of patients with breast cancer and small cell lung cancer estimated that a clinically relevant change in score on any of the EORTC QLQ-C30 scales to be 10 points.74 Using an anchor-based approach to estimate the MID in which patients who reported “a little” change (for better or worse) on the subjective significance questionnaire had corresponding changes on a function or symptom scale of the EORTC QLQ-C30 of approximately 5 to 10 points. Participants who reported a “moderate” change had corresponding changes in the EORTC QLQ-C30 of about 10 to 20 points, and those who reported being “very much” changed had corresponding changes of more than 20 points.
A Canadian study estimated the MID for the EORTC QLQ-C30 among 369 patients with advanced cancer, the most common cancer being breast cancer, followed by lung, prostate, gastrointestinal, renal cell, and other cancers.73 Patients completed the questionnaire at baseline and 1 month post-radiation. Using both an anchor- and distribution-based methods for improvement and deterioration, 2 anchors of overall health and overall HRQoL were used, both taken directly from the EORTC QLQ-C30 (questions 29 and 30) where patients rated their overall health and HRQoL themselves. Improvement and deterioration were categorized as an increase or decrease by 2 units to account for the natural fluctuation of patient scoring. With these 2 anchors, the estimated MIDs across all EORTC QLQ-C30 scales ranged from 9.1 units to 23.5 units for improvement, and from 7.2 units to 13.5 units for deterioration. Distribution-based estimates were closest to 0.5 standard deviation (SD).
EQ-5D-5L
The EQ-5D-5L is a generic QoL instrument developed by the EuroQol Group. It may be applied to a wide range of health conditions and treatments.56 The EQ-5D-5L was developed by the EuroQol Group as an improvement to the 3-Level EQ-5D (EQ-5D-3L) instrument, to measure small and medium health changes and reduce ceiling effects. As a generic measure of HRQoL that can capture the net effect of treatment benefits and harms, the EQ-5D-5L provides valuable information from a patient perspective. In addition, the EQ-5D-5L is used in clinical trials to obtain utility weights for economic models.57 The EQ-5D-5L consists of the EQ-5D descriptive system and the EQ VAS.
The EQ VAS records the respondent’s self-rated health on a vertical VAS58 where the end points are labelled 0 (“the worst health you can imagine”) and 100 (“the best health you can imagine”). The respondents are asked to mark an X on the point of the VAS that best represents their health on that day. The VAS scores can be summarized and analyzed as continuous data.56,57
The EQ-5D-5L has been extensively validated across countries around the world and in various conditions. However, the psychometric properties of the EQ-5D-5L have not been assessed in patients with non–small cell lung cancer; therefore, its validity, reliability, and responsiveness to change have not been discussed further in this report.
Minimal Important Difference
Pickard et al.55 conducted a retrospective analysis on 534 patients with cancer (not including EC) to estimate an MID for the EQ-5D-5L VAS based on anchor-based (ECOG PS and the Functional Assessment of Cancer Therapy - General (FACT-G) total score-based quintiles) and distribution-based (0.5 SD and standard error of the measure) methods. Based on ECOG PS grade as an anchor, the estimated MID in patients with all cancers ranged from 8 to 11 (0.5 SD = 10). The FACT-G quintile-based MID for patients with all cancers ranged from 7 to 10 (0.5 SD = 9).
Other Considerations and Limitations
Because the EQ-5D-5L is intended to measure HRQoL in the general population, there may be a mismatch between its domains or dimensions and HRQoL in patients with endometrial cancer that are affected by treatments and/or the disease. Also, Pickard et al. used an older version of EQ-5D-3L VAS in their study.55 In discussion with the CADTH review team, however, it was determined to be only a slight difference between the old EQ-5D-3L VAS, which has the numerical scale overlapped on the VAS, and the most recent version of EQ-5D-3L or 5L VAS, where the VAS has been harmonized between the 3L and 5L and the scale has been placed to the right side of the VAS. Therefore, the MID estimate using the old version of EQ-5D-3L VAS was reported as no major discrepancies in interpretation are expected.
EORTC QLQ-EN24
Description
EORTC QLQ-EN24 represents a validated version of the EORTC QLQ that was developed specific for endometrial cancer, designed to be used in conjunction with the core questionnaire (QLQ-C30).76 The EORTC QLQ-EN24 assesses disease and treatment specific aspects of the HRQoL of patients with EC. It uses a 7-day recall period and includes 3 functional, single-item scales (sexual interest, activity and enjoyment) and 10 symptom scales, which incorporate both multi-item scales and single-item measures (lymphedema, urological symptoms, gastrointestinal symptoms, poor body image, sexual/vaginal problems, pain in the back and pelvis, tingling/numbness, muscular pain, hair loss, and taste change).76
Scoring
EORTC QLQ-EN24 is a self-reported instrument that contains 24 questions, scored on a 4-point Likert scale (1: not at all; 2: a little; 3: quite a bit; 4: very much). Scoring algorithm for the QLQ-EN24 includes linear transformations to a 0–100 scale, equivalent to the process described for function and symptom scales / single items of the core, QLQ-C30, questionnaire.81 Higher scores on the symptom scales indicate higher level of symptoms or problems, while higher scores on sexuality items (sexual interest, activity and enjoyment) suggest better sexual functioning.76
Psychometric Properties
In the original validation study EORTC QLQ-EN24, 268 patients with histologically confirmed endometrial cancer who had pelvic surgery without adjuvant treatment, were undergoing adjuvant treatment or had completed treatment more than 3 months ago across Europe, Australia, and Asia.76 Multi-trait scaling analyses was applied to test the psychometric properties of the QLQ-EN24. Internal consistency was confirmed with acceptable Cronbach alpha coefficient range (0.74 to 0.86) for 5 multi-item scales (lymphedema 0.80, urological symptoms 0.75, gastrointestinal symptoms 0.74, poor body image 0.86, sexual/vaginal problems 0.86). Sexual functioning items were kept as single-item scales (sexual interest, activity, and enjoyment) due to poor psychometric properties observed when combining these items with vaginal symptoms. Evidence of convergent validity for the scales was supported by higher item-scale correlations (0.40 to 0.84 values for item correlation with their own scale), while the evidence of discriminant validity was supported by lower correlations (0.08 to 0.39 values for item correlation with the other scales).76
Test-retest reliability was assessed in the group of patients who had completed treatment using Pearson’s correlations. Questionnaires were completed twice by the patients (3 months after treatment and 3-7 days later), and high correlations were observed for all multi-item scales (ranging from 0.81 to 0.92) as well as single-item questions (ranging from 0.72 to 0.97).76
Pearson’s product moment correlations between the various scales of the EORTC QLQ-C30 and the QLQ-EN24 module were employed to test convergent and divergent validity. Most QLQ-EN24 scales were weakly correlated with the QLQ-C30 scales (r < 0.40). Higher correlations were observed between back/pelvic pain (QLQ-EN24) and pain scale (QLQ-C30) (r = 0.57), between gastrointestinal symptom scale (QLQ-EN24) and diarrhea item (QLQ-C30) (r = 0.51), and between taste change item (QLQ-EN24) and appetite loss item (QLQ-C30) (r = 0.62).76
Construct validity was assessed using a known groups approach and was specifically based on clinical parameters (KPS) and treatment status. Patients with lower PS (≤ 80) showed significantly higher scores on 4 multi-item (lymphoedema, urological symptoms, gastrointestinal symptoms, and body image problems) and 3 single-item scales (back/pelvic pain, muscular/joint pain, and taste change). Moreover, significantly lower scores on the sexual interest and activity scales were observed when comparing individuals with lower to those with better performance status. When assessing patients with different treatment status, patients on adjuvant treatment reported significantly more issues with tingling and numbness, hair loss, and taste change, compared to patients after surgery or who had completed treatment. The post-treatment group had significantly higher muscle and joint pain and lower body image in comparison to adjuvant treatment and after surgery groups. Individuals on adjuvant treatment showed lowest levels of sexual interest, activity, and enjoyment.76
Another validation study of the Mexican-Spanish version of the QLQ-EN24 was performed in 189 patients with EC.75 The study confirmed the scale structure, the internal consistency and reliability of the QLQ-EN24 (Cronbach alpha for symptoms scales: lymphedema=0.66, urological symptoms=0.76, gastrointestinal symptoms=0.75, poor body image=0.89, sexual/vaginal problems=0.86), which was aligned with its original assessment.76 Clinical validity was assessed through a known-group comparisons approach. Six symptom scores (lymphoedema, urological symptoms, gastrointestinal symptoms, body image problems, tingling/numbness, and taste change) of the QLQ-EN24 were strongly associated with the basal serum albumin level of patients. Presence of menopause was associated with higher scores on the lymphoedema and tingling/numbness scales, and higher sexual functioning scores (sexual interest, activity, and enjoyment).75
Analysis of a Polish version of the QLQ-EN24 was conducted among 208 women with endometrial cancer before surgery, during adjuvant treatment and follow-up.77 Reliability and internal consistency of the QLQ-EN24 multi-item scales were of acceptable levels (Cronbach alpha range from 0.77 to 0.97). Evidence od convergent and divergent validity was assessed through Pearson’s product moment correlation between QLQ-EN24 and QLQ-C30 sales. Several QLQ-EN24 correlated moderately to highly with QLQ-C30 (r =0.66 for back/pelvis pain (EN24) and pain (C30); r= 0.64 for gastrointestinal symptoms (EN24) and pain (C30); r=0.63 for body image (EN24) and emotional functioning (C30); r=0.59 for tingling/numbness (EN24) and physical functioning (C30); r=0.57 for body image (EN24) and fatigue (C30), lymphoedema (EN24) and physical functioning (C30); r=0.56 for tingling/numbness (EN24) and fatigue (C30) (r =.56)). Known-group comparisons were employed to test the clinical validity of QLQ-EN24 according to different clinical parameters of patients (histological grade, BMI, comorbidity, treatment status, disease stage). Patients undergoing adjuvant treatment described significantly higher levels of symptoms such as hair loss and taste change, when compared to those before treatment or during observation. Patients with high-grade histology reported significantly more severe symptoms (tingling/ numbness, muscular pain, hair loss, and taste change) than patients with lower grade tumours (G1 and G2). Comorbidity was associated with significantly lower scores on sexual functioning items (interest, activity, and enjoyment), and higher scores on the lymphoedema scale. There were no differences in any of the QLQ-EN24 scores when analyzing patients with diverse BMI levels and disease stage.77
Minimal Important Difference
A literature search was conducted to identify MID of the EORTC- QLQ-EN24 In patients with endometrial cancer and none were identified.
Appendix 5: Updated Data Provided by the Sponsor
Note that this appendix has not been copy-edited.
Final Analysis of KEYNOTE-775 Trial (Cut-Off Date: March 1, 2022)
The sponsor provided a final descriptive analysis (Cut-off date: March 1, 2022. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||).82 The results of the final analysis appeared consistent to that observed in the interim analysis 1 (Cut-off date: October 26, 2020) summarized in this review report.(Refer to Table 65, Figure 14, Figure 15, and Figure 16)
No QoL outcome (e.g., EORTC QLQ-C30 GHS) was reported in the final analysis. No AEs results for the pMMR population was reported in the final analysis.
It was indicated that the final analysis is descriptive only with no inferential testing because the success criteria for all the primary hypotheses of PFS and OS were met at IA1. Reported p values are nominal.82
Table 65: Key Results of KEYNOTE-775 Trial Reported in IA1 and Final Analysis (pMMR)
Outcomes | Interim analysis 1 (October 26, 2020) | Final analysis (March 1, 2022) | ||
|---|---|---|---|---|
PEM + LEN (N= 346) | TPC (N= 351) | PEM + LEN (N= 346) | TPC (N= 351) | |
OS (ITT) | ||||
Events, n (%) | 165 (47.7) | 203 (57.8) | |||||||||||||||||||| | |||||||||||||||||||| |
Median OS (95% CI), moa | 17.4 (14. 2 to 1 9.9) | 12.0 (10.8 to 13.3) | 18.0 (14.9-20.5) | 12.2 (11.0-14.1) |
HRb (95% CI) | 0.68 (0.56 to 0.84) | 0.70 (0.58-0.83) | ||
P valuec | 0.0001c | <0.0001 | ||
12-month OS Rates (95% CI), %a | 61.6 (56.1 to 66.6) | 49.5 (43.8 to 55.0) | |||||||||||||||||||| | |||||||||||||||||||| |
PFS (ITT) | ||||
Events, n (%) | 247 (71.4) | 238 (67.8) | |||||||||||||||||||| | |||||||||||||||||||| |
Median PFS (95% CI), mo.a | 6.6 (5.6 to 7.4) | 3.8 (3.6 to 5.0) | 6.7 (5.6-7.4) | 3.8 (3.6-5.0) |
HRb (95% CI) | 0.60 (0.50 to 0.72) | 0.60 (0.50–0.72) | ||
P valuec | < 0.0001c | <0.0001 | ||
6-month PFS rates (95% CI), %a | 52.1 (46.5 to 57.3) | 36.2 (30.5 to 41.9) | |||||||||||||||||||| | |||||||||||||||||||| |
EORTC QLQ-C30 GHS (FAS) | ||||
Baseline | NR | NR | ||
N (% of ITT)) | 316 (91.3) | 298 (84.9) | NR | NR |
Mean (SD) | 66. 56 (21.44) | 66.64 (22.43) | NR | NR |
At week 12 | NR | NR | ||
N (% of ITT) | 256 (74.0) | 192 (54.7) | NR | NR |
Mean (SD) | 60.94 (21.35) | 62.80 (21.67) | NR | NR |
Change from Baseline to Week 12 | NR | NR | ||
N (% of ITT) | 327 (94.5) | 310 (98.4) | NR | NR |
LS mean (95% CI) | –6.80 (–9.43 to –4.17) | –7.96 (–10.86 to –5.05) | NR | NR |
LSM Difference (95% CI)d | 1.16 (–2.49 to 4.81) | NR | NR | |
P valued | 0.5316 | NR | NR | |
ORR (ITT) | ||||
N | 346 | 351 | 346 | 351 |
Events, n (%) | 105 (30.3) | 53 (15.1) | 112 (32.4) | 53 (15.1) |
Difference in % (95% CI)e vs. TPC | 15.2 (9.1 to 21.4) | 17.2 (11.0 to 23.5) | ||
P valuef | <0.0001 | <0.0001 | ||
DOR (months) | ||||
Median (Range)g | 9.2 (1.6+ to 23.7+) | 5.7 (0.0+ to 24.2+) | 9.3 (1.6 to 39.5) | 5.7 (0.0 to 37.1) |
DOR = duration of response; EORTC QLQ-C30 GHS = ORTC QLQ-C30 Global Health Status; FAS = full analysis set; ITT = intention to treat; LSM = least squares mean; n = number of patients with event; N = total number of patients included in the analysis; NR= not reached ORR=objective response rate; OS=overall survival; PEM + LEN = pembrolizumab + lenvatinib combination; PFS=progression-free survival; pMMR = mismatch repair proficient; SD = Standard deviation; TPC = treatment of physician’s choice.
aFrom product-limit (Kaplan-Meier) method for censored data.
bBased on Cox regression model with the Efron method of tie handling with treatment as a covariate stratified by ECOG Performance Status, geographic region, and prior history of pelvic radiation.
cOne-sided P value based on log-rank test stratified by, ECOG Performance Status, geographic region, and prior history of pelvic radiation. The P value has been adjusted for multiple testing.
dBased on a cLDA model with the PRO scores as the response variable with covariates for treatment by study visit interaction, stratification factors MMR status, ECOG Performance Status, geographic region, and prior history of pelvic radiation. Note: For baseline and week 12, N is the number of patients in each treatment group with non-missing assessments at the specific time point; for change from baseline, N is the number of patients in the analysis population in each treatment group. The P value has not been adjusted for multiple testing.
eBased on Miettinen & Nurminen method stratified by ECOG Performance Status, geographic region, and prior history of pelvic radiation.
fOne-sided P value for testing. H0: difference in % = 0 versus H1: difference in % > 0. The P value has been adjusted for multiple testing.
gFor DOR, "+" indicates there is no progressive disease by the time of last disease assessment. No progressive disease reported at the last assessment (i.e., final analysis)
Source: Clinical Study Report19 and the sponsor provided additional final analysis.82
Figure 14: Kaplan-Meier Estimates of OS in Patients With pMMR Tumours (KEYNOTE-775 Trial, Cut-Off Date: March 1, 2022, ITT)
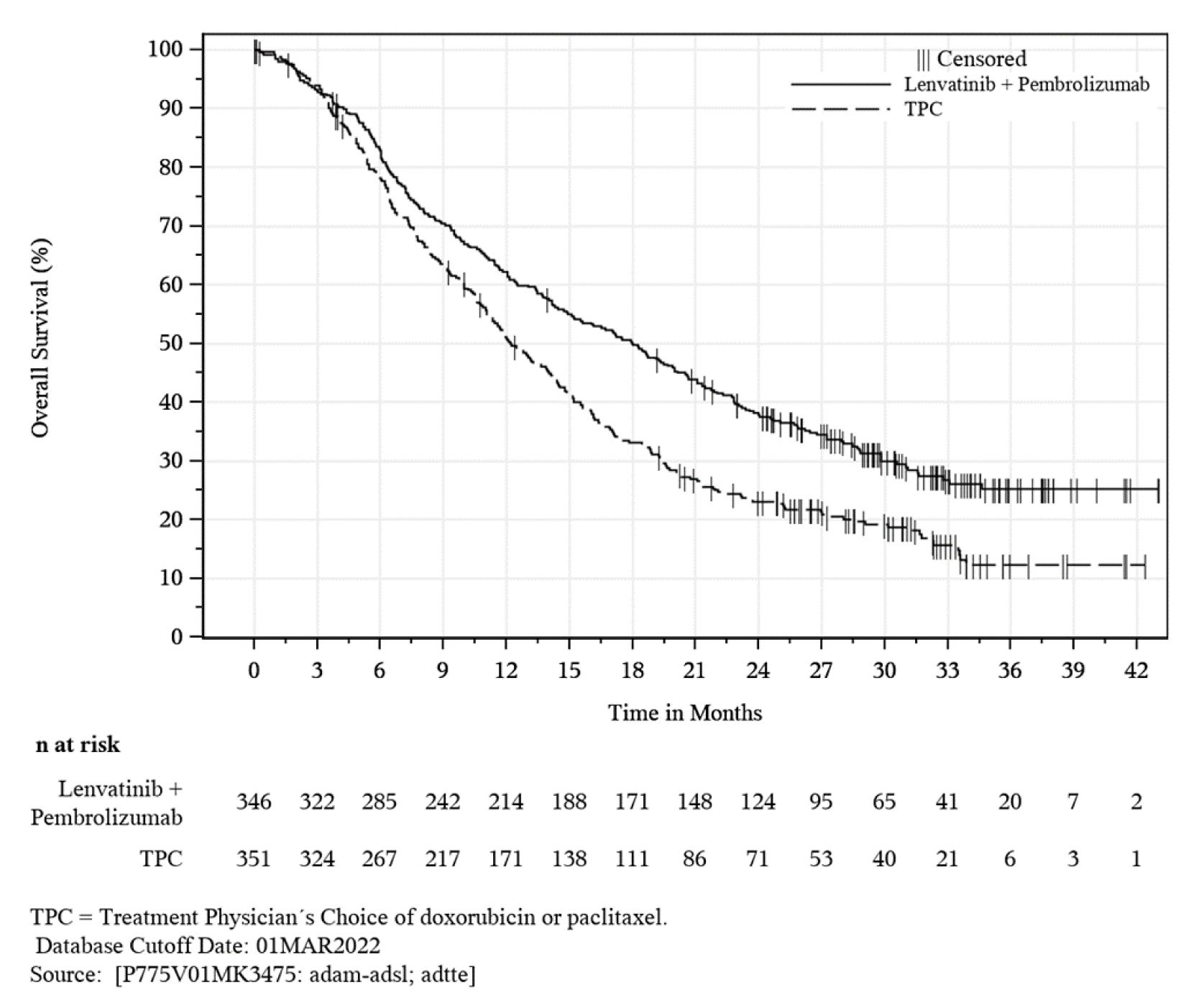
Source: Sponsor’s submission.32
Figure 15: Kaplan-Meier Estimates of PFS (Primary Censoring Rule) in Patients With pMMR Tumours (KEYNOTE-775 Trial, Cut-Off Date: March 1, 2022, ITT)
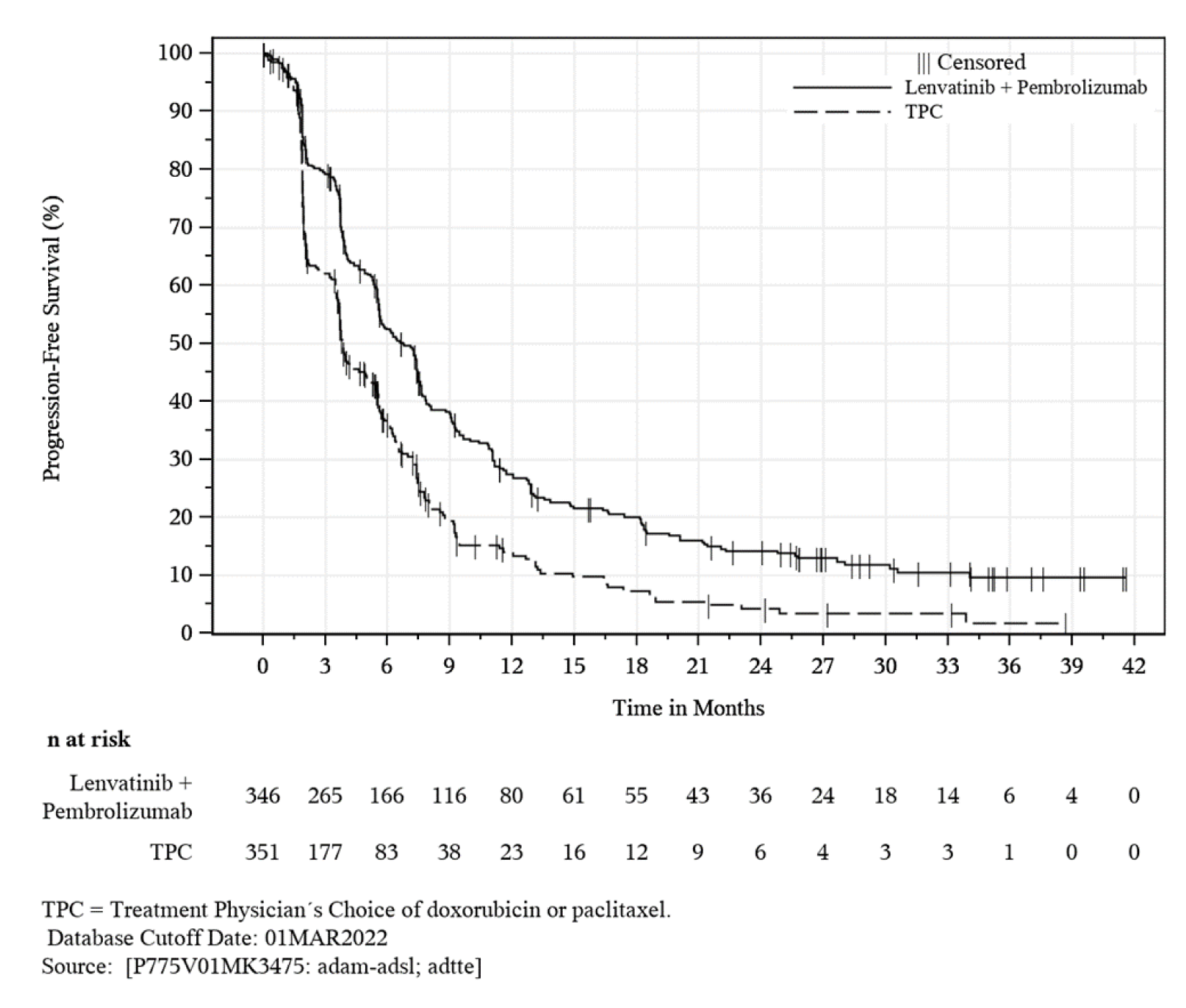
Source: Sponsor’s submission.32
Figure 16: Kaplan-Meier Estimates of Duration of Response in pMMR With Confirmed Response (KEYNOTE-775 Trial, Cut-Off Date: March 1, 2022, ITT)
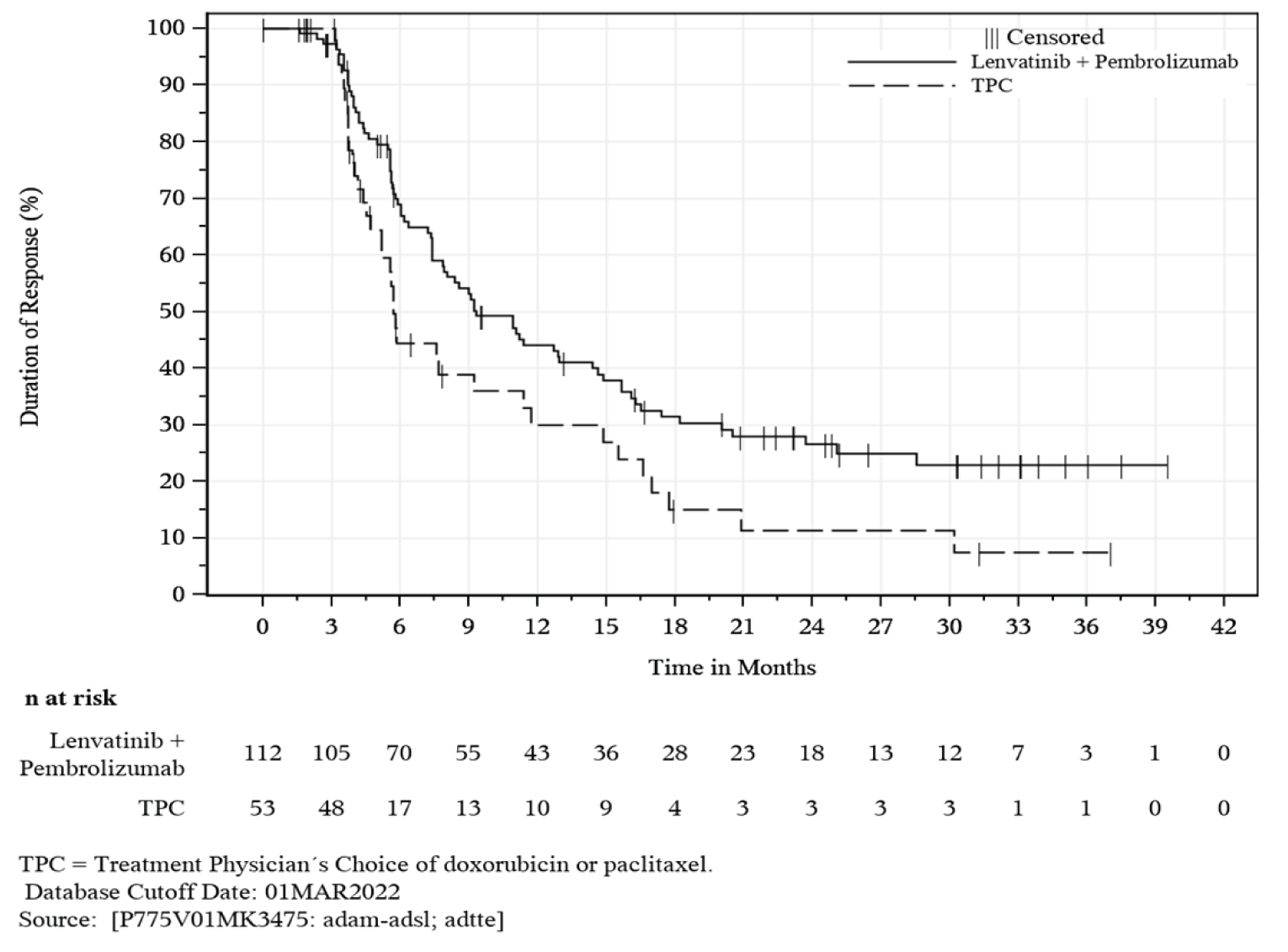
Source: Sponsor’s submission.32
KEYNOTE-146: Updated Results on August 18, 2020
The sponsor provided an update analysis for pMMR (cut-off date: August 18, 2020. Median follow-up duration, 35.8 month; 95% CI, 31.2 to 41.2).83 The sponsor indicated that the findings of the updated analysis were largely aligned with that reported in the analysis on January 10, 2019. (Refer to Table 66, Figure 17, Figure 18, and Figure 19). No AE data for pMMR group was reported in this updated analysis.
Table 66: Key Results of KEYNOTE-146 Trial Reported on January 10, 2019, and Updated on August 18, 2020 (pMMR)
Outcomes | PEM + LEN (N=94) | PEM + LEN (N=94) |
|---|---|---|
January 10, 2019 | August 18, 2020 | |
OS | ||
Deaths, n (%) | 44 (46.8) | 63 |
Censored Patients, n (%) | 50 (53.2) | 31 |
OS (months)a | ||
Median (95% CI) | 16.4 (13.5, 25.9) | 17.2 (15.0 to 25.8) |
Follow-up time for OS (months)a | ||
Median (95% CI) | 18.7 (14.1, 20.9) | 35.8 (31.2 to 41.2) |
PFS | ||
Patients with Events, n (%) | 58 (61.7) | 68 |
Censored Patients, n (%) | 36 (38.3) | 26 |
PFS (months)b | ||
Median (95% CI) | 5.4 (4.4 to 7.6) | 7.4 (4.4 to 7.6) |
Follow-up time for PFS (months)b | ||
Median (95% CI) | 9.9 (9.6, 15.9) | 35.8 (31.2 to 41.2) |
Tumour Response | ||
# of patients with ORR (CR + PR), n (%) | 36 (38.3) | 36 (38.3) |
ORR rate, %, (95% CI) | 38.3 (28.5 to 48.9) | 38.3 (28.5 to 48.9) |
Best Overall Response, n (%) | ||
Complete response | 10 (10.6) | 8 (8.5) |
Partial response | 26 (27.7) | 28 (29.8) |
Stable disease | 38 (40.4) | NR |
Progressive disease | 12 (12.8) | NR |
Not evaluable | 8 (8.5) | NR |
DOR | ||
Number of Patients With a Response (CR+PR), N | 36 | 18 |
Duration of Objective Response Among Responders (months)a | ||
Median (95% CI) | NE (6.3, NE) | 23.0 (8.5 to NE) |
Range (min, max) | (1.2+, 33.1+) | |
Censored Duration of Response, n/N (%) | 21/36 (58.3) | 18 |
Patients with Duration of Response Among Responders: | ||
≥6 months | ||
N | 25 | 29 |
Probability (95% CI)b | 0.76 (0.58,0.87) | 0.85 (0.68 to 0.94) |
≥12 months | ||
N | 8 | 21 |
Probability (95% CI)b | 0.51 (0.30 to 0.68) | 0.64 (0.45 to 0.78) |
Time to Response (months) Among Responders | Median (Range) 1.4 (1.1, 8.0) | Mean (SD) 3.2 (3.65) |
CI= confidence interval; CR = complete response; FAS = full analysis set; NE = not estimable because not reached; OS=overall survival; ORR = objective response rate; PEM + LEN = pembrolizumab plus lenvatinib combination therapy; PFS = progression-free survival; pMMR = mismatch repair proficient; PR = partial response, SD = standard deviation.
aThe median is estimated using Kaplan-Meier methodology, and their 95% CIs were constructed with a generalized Brookmeyer and Crowley method.
bPoint estimates for OS were based on Kaplan-Meier methodology and 95% CIs were based on the Greenwood formula using log-log transformation.
Source: Clinical Study Report61 and the sponsor provided updated analysis data (August 18, 2020)83
Figure 17: Kaplan-Meier Plot of OS in pMMR (KEYNOTE-146 Trial, Cut-Off Date: August 18, 2020)
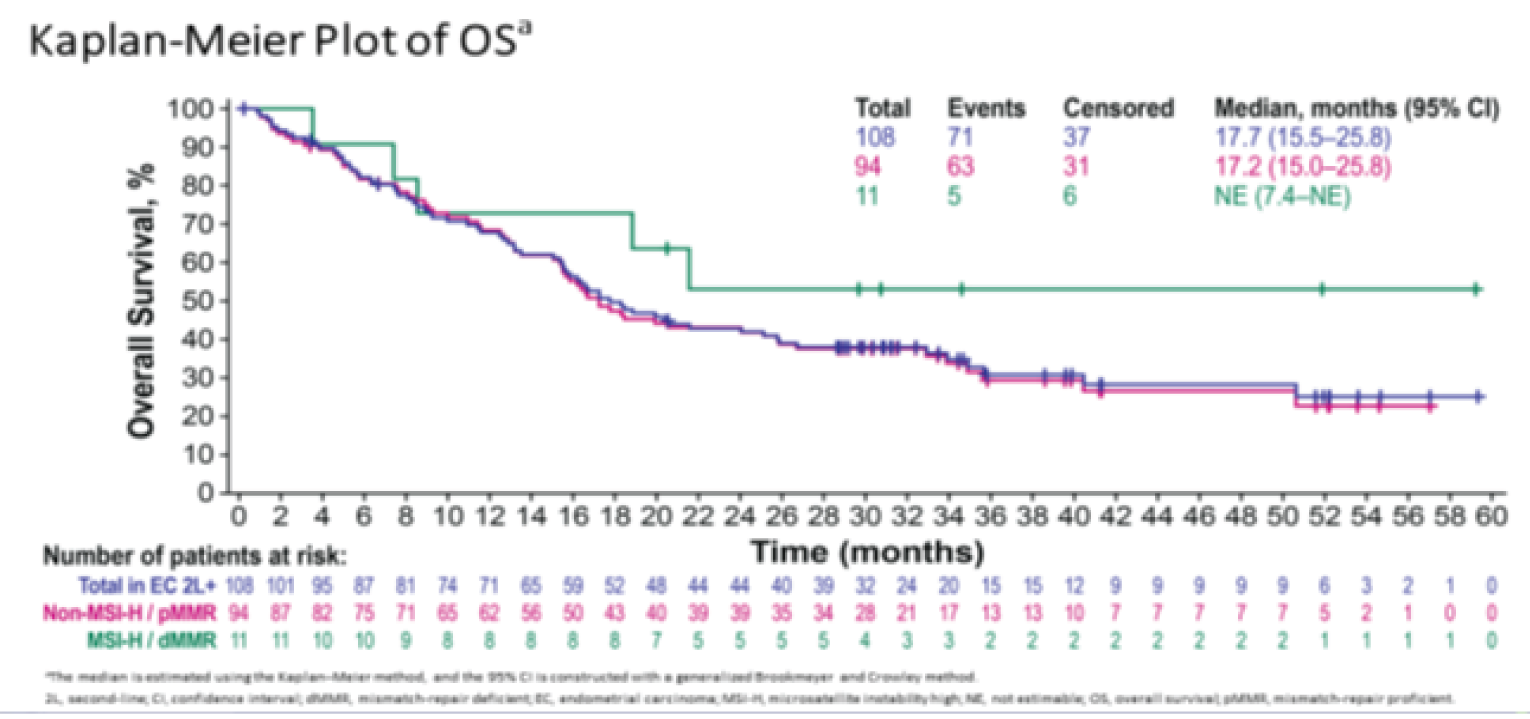
Source: Sponsor’s submission.32
Figure 18: Kaplan-Meier Plot of PFS in pMMR (KEYNOTE-146 Trial, Cut-Off Date: August 18, 2020)
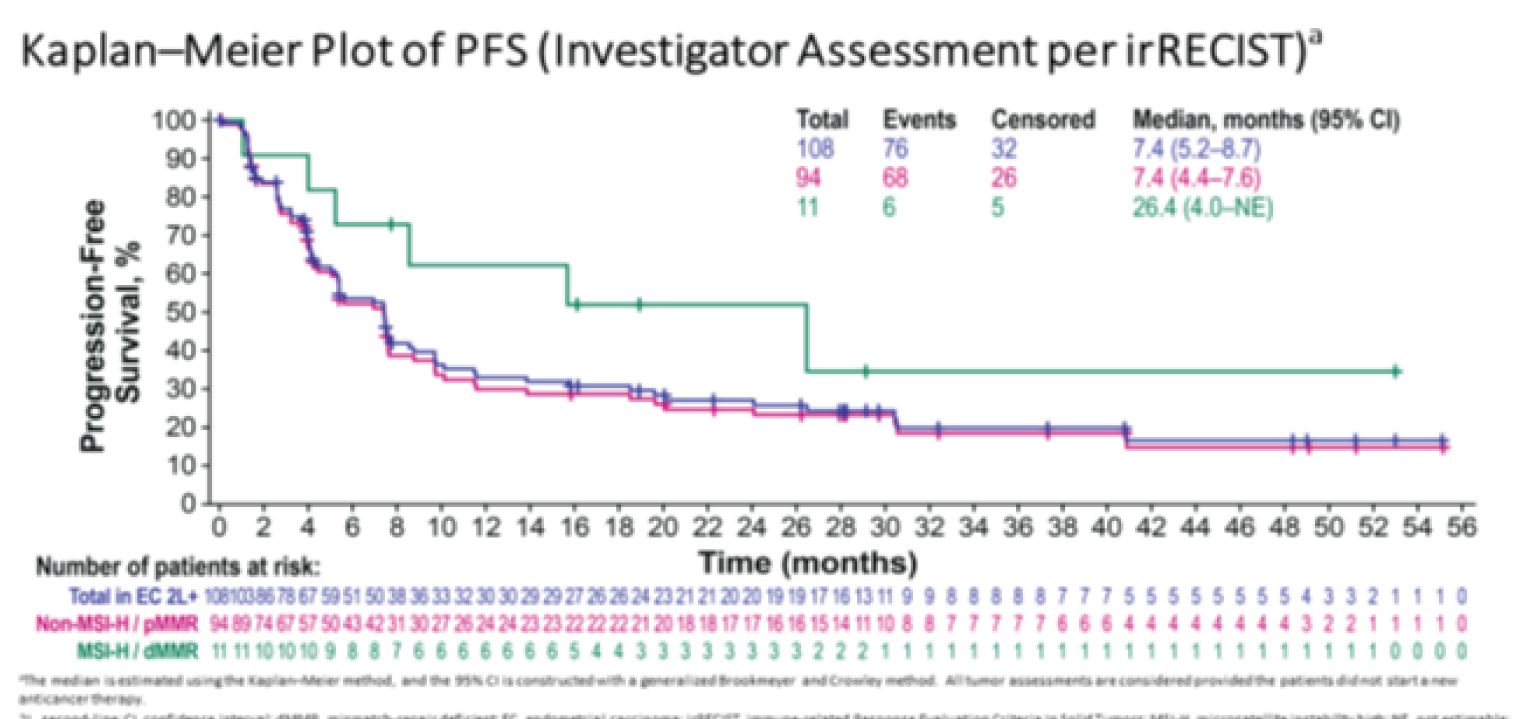
Source: Sponsor’s submission.32
Figure 19: Kaplan-Meier Estimates of DOR in pMMR With Confirmed Response (KEYNOTE-146 Trial, Cut-Off Date: August 18, 2020)
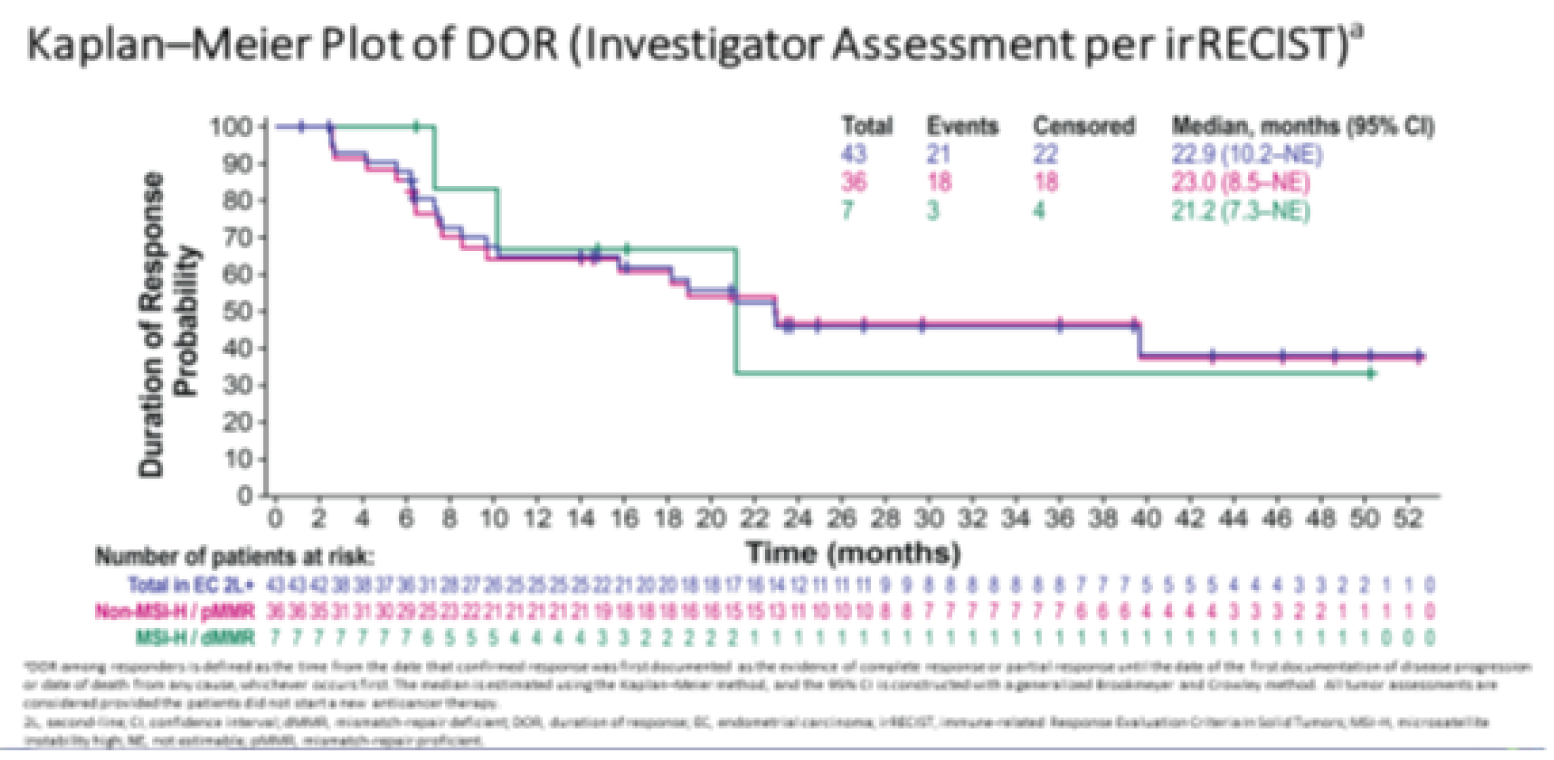
Source: Sponsor’s submission.32
Pharmacoeconomic Review
Abbreviations
AE
adverse event
BIA
budget impact analysis
dMMR
mismatch repair deficient
ICER
incremental cost-effectiveness ratio
KM
Kaplan-Meier
LEN
lenvatinib
MSI-H
microsatellite instability-high
OS
overall survival
PCC
physician’s choice of chemotherapy
PEM
pembrolizumab
PEM + LEN
pembrolizumab plus lenvatinib combination therapy
PFS
progression-free survival
pMMR
mismatch repair proficient
PSM
partitioned survival model
QALY
quality-adjusted life-year
TOT
time on treatment
Executive Summary
The executive summary comprises 2 tables (Table 1 and Table 2) and a conclusion.
Item | Description |
|---|---|
Drug product | Pembrolizumab (Keytruda), 200 mg administered IV in combination with 20 mg lenvatinib taken orally |
Submitted price | Pembrolizumab 100 mg, solution: $4,400.00 per 100 mg/4 mL vial |
Indication | In combination with lenvatinib for the treatment of adult patients with advanced endometrial carcinoma that is not MSI-H or dMMR who have disease progression following prior platinum-based systemic therapy and are not candidates for curative surgery or radiation |
Health Canada approval status | NOC/c |
Health Canada review pathway | Project Orbis |
NOC date | September 20, 2019 |
Reimbursement request | As per indication |
Sponsor | Merck Canada Inc. |
Submission history | Previously reviewed: Yes Pembrolizumab (Keytruda) has been reviewed for multiple indications at CADTH. The following indications were reviewed in 2020 and 2021: Indication: MSI-H or dMMR endometrial cancer Recommendation date: In progress Recommendation: TBD Indication: Esophageal carcinoma, gastroesophageal junction adenocarcinoma Recommendation date: December 20, 2021 Recommendation: Reimburse with clinical criteria and/or conditions Indication: Classical Hodgkin lymphoma Recommendation date: November 1, 2021 Recommendation: Reimburse with clinical criteria and/or conditions Indication: Metastatic or unresectable recurrent head and neck squamous cell carcinoma Recommendation date: December 22, 2020 Recommendation: Recommended on the condition of cost-effectiveness being improved to an acceptable level Indication: Advanced renal cell carcinoma Recommendation date: April 2, 2020 Recommendation: Recommended on the condition of cost-effectiveness being improved to an acceptable level |
dMMR = mismatch repair deficient; MSI-H = microsatellite instability-high; NOC = Notice of Compliance; NOC/c = Notice of Compliance with Conditions; TBD = to be determined.
Table 2: Summary of Economic Evaluation
Component | Description |
|---|---|
Type of economic evaluation | Cost-utility analysis PSM |
Target population | Second-line treatment of adult patients with advanced endometrial carcinoma that is not MSI-H or dMMR who have disease progression following prior platinum-based systemic therapy and are not candidates for curative surgery or radiation |
Treatment | Pembrolizumab in combination with lenvatinib |
Comparator | PCC (doxorubicin or paclitaxel) |
Perspective | Canadian publicly funded health care payer |
Outcomes | QALYs, LYs |
Time horizon | Lifetime (20 years) |
Key data source | KEYNOTE-775, a multi-centre, open-label, randomized, phase III trial |
Submitted results | The ICER for pembrolizumab + lenvatinib was $125,808 per QALY (incremental costs = $158,046; incremental LYs = 1.84; incremental QALYs = 1.26). |
Key limitations |
|
CADTH reanalysis results |
|
dMMR = mismatch repair deficient; ICER = incremental cost-effectiveness ratio; LY = life-year; MSI-H = microsatellite instability-high; OS = overall survival; PCC = physician’s choice of chemotherapy; PFS = progression-free survival; PSM = partitioned survival model; QALY = quality-adjusted life-year.
Conclusions
The CADTH clinical review noted that the results of the KEYNOTE-775 trial showed that, compared to physician’s choice of chemotherapy (PCC), pembrolizumab plus lenvatinib (PEM + LEN) combination therapy showed a statistically significant and clinically meaningful benefit in terms of overall survival (OS) and progression-free survival (PFS) in the treatment of adult patients with advanced mismatch repair proficient (pMMR) (i.e., not microsatellite instability-high [MSI-H] or mismatch repair deficient [dMMR]) endometrial carcinoma who have disease progression following prior platinum-based systemic therapy and are not candidates for curative surgery or radiation. The clinical experts CADTH consulted for this review indicated that the safety profile of PEM + LEN observed in this study appeared consistent with the known safety profile of each individual drug (i.e., PEM or LEN) and that no additional safety signals were identified. The clinical review noted that longer duration of follow-up may be needed to further confirm the long-term benefit of PEM + LEN combination therapy over PCC chemotherapy in this population.
CADTH identified several limitations in the sponsor’s pharmacoeconomic analysis that have notable implications for the cost-effectiveness results. First, the sponsor’s chosen survival extrapolations overestimated the survival of patients in the model on either therapy compared to clinical expectations. The partitioned survival model (PSM) also produced an apparent post-progression survival benefit that may be the result of a structural bias and favours PEM + LEN. CADTH updated the costs of concomitant and comparator medications (LEN, doxorubicin, and paclitaxel) according to publicly available sources. CADTH also noted limitations in the comparator choice, health state utility value, and drug wastage calculations. CADTH made various changes to the base case to address these limitations. The CADTH reanalysis resulted in an incremental cost-effectiveness ratio (ICER) for PEM + LEN of $366,399 per quality-adjusted life-year (QALY) compared to PCC (incremental costs = $150,222; incremental QALYs = 0.41; incremental life-years = 0.54), with a 0% probability of cost-effectiveness at a $50,000 per QALY threshold. Even with a 100% price reduction for PEM, PEM + LEN would not be cost-effective at this threshold.
Despite the benefits noted in the CADTH clinical review pertaining to OS and PFS, the extrapolation of these data beyond the period for which Kaplan–Meier (KM) data are available is uncertain, with approximately 85% of the incremental benefit of PEM + LEN coming from the extrapolated period (compared to 52% in the CADTH base case). In addition, uncertainty remains in the analysis regarding the choice of comparators and the impact of re-treatment with platinum therapy, which CADTH could not address. CADTH performed scenario analyses on the OS extrapolation, using a Weibull or log-logistic distribution for both treatments. These analyses indicated that results are sensitive to assumptions about the long-term OS benefit of PEM + LEN compared to PCC, which was noted as a gap in the available trial evidence. The cost-effectiveness results were robust to other parameters varied in the CADTH reanalysis and scenario analyses.
Stakeholder Input Relevant to the Economic Review
This section is a summary of the feedback received from the patient groups, registered clinicians, and drug plans that participated in the CADTH review process (specifically, information that pertains to the economic submission).
One patient group, the Colorectal Cancer Resource and Action Network, a collaboration between the Canadian Cancer Society and the Canadian Cancer Survivor Network, provided input for the current review. Data were collected from 20 Canadian patients with endometrial cancer and 2 caregivers through an online survey. Patient input emphasized that there are limited treatment options for patients with advanced endometrial cancer. These include surgery, radiation therapy (in addition to hormonal therapy), and standard chemotherapy treatments indicated for endometrial cancer. Significant treatment-related toxicities or adverse effects of treatment were noted, including fatigue, constipation, peripheral neuropathy (numbness, tingling, and pain in the nerves), and changes in libido or sexual function. Importantly, patient input noted the overall lack of efficacy associated with standard of care chemotherapies that compromised their everyday quality of life in addition to causing toxic side effects. Additionally, patients reported that the activities or tasks of daily living that were moderately to significantly affected included exercise, work, travel, and household chores, which affected their quality of life. Patients indicated that drug affordability, improved efficacy (e.g., OS), disease regression, a reduction in side effects and toxicities (to none or minimal), and improved quality of life were the most clinically meaningful outcomes and important treatment goals. Three patients with experience on PEM + LEN achieved disease regression and improved quality of life on the therapy. Patients appreciated the oral formulation of LEN and the short infusion time of PEM compared to standard therapies.
One clinician group provided input for this review: the Ontario Health (Cancer Care Ontario) Gynecology Cancer Drug Advisory Committee. The group reiterated the same treatment goals that were expressed by patients as being most important: prolonged life, delayed disease progression, symptomatic relief, partial response, full response, and improved health-related quality of life. Importantly, registered clinicians indicated that there is currently no standard of care for the indicated population, and most patients do not respond to the limited number of available treatments. Clinicians noted that PEM + LEN combination therapy would replace potential treatment with doxorubicin or carboplatin plus paclitaxel and would become the preferred treatment option post-platinum therapy.
Drug plan input was received for this review. Drug plans requested guidance on the number of prior lines of platinum-based therapy required for a patient to meet the eligibility criteria for the KEYNOTE-775 trial. Drug plans also requested guidance on re-treatment for the indicated population and on treatment discontinuation, particularly with respect to the continued use of 1 of the drugs if the other drug within the doublet is discontinued. Drug plans anticipated the potential for indication creep for patients who may be eligible, such as for patients with carcinosarcoma and sarcoma (i.e., leiomyosarcoma and stromal sarcomas) or unstable central nervous system metastases (who were excluded from the pivotal trial). The plans also require clarity about whether patients with an Eastern Cooperative Oncology Group Performance Status of 1 or greater would be eligible for PEM + LEN. Drug plans also anticipate that drug wastage is likely for PEM and raised concerns about whether weight-based dosing of PEM would be applicable for the given indication in Canadian clinical practice. Importantly, the drug plans indicated that mismatch repair (MMR) or MSI-H testing in endometrial cancer is not standard across jurisdictions.
Several of these concerns were addressed in the sponsor’s model:
PFS and OS were modelled for the overall population. Health-related quality of life was incorporated into the model by progression status.
CADTH was unable to address the following concerns raised in stakeholder input:
Additional relevant comparators could not be incorporated into the analysis due to model constraints. These comparators included single-drug chemotherapies (i.e., carboplatin, paclitaxel, pegylated liposomal doxorubicin), combination chemotherapies (i.e., carboplatin plus paclitaxel, carboplatin plus pegylated liposomal doxorubicin), and a mix of treatments comprising various chemotherapies and hormonal therapies.
Within the economic model, the sponsor incorporated a fixed dosing approach (i.e., 200 mg every 3 weeks or 400 mg every 6 weeks) for PEM, as per the KEYNOTE-775 trial. The sponsor did not include an option to assess the effects of weight-based dosing for PEM; as such, CADTH was unable to explore this option further. However, drug wastage of comparator treatments was incorporated, as appropriate. The cost-effectiveness of weight-based dosing for PEM remains unknown.
Economic Review
The current review is for PEM (Keytruda) in combination with LEN (Lenvima) for adult patients with advanced endometrial carcinoma.
Economic Evaluation
Summary of Sponsor’s Economic Evaluation
Overview
PEM is indicated as combination therapy with LEN for the treatment of adult patients with advanced endometrial cancer that is not MSI-H or dMMR who have disease progression following prior platinum-based systemic therapy in any setting and are not candidates for curative surgery or radiation.
The sponsor submitted a cost-utility analysis to assess the cost-effectiveness of PEM + LEN compared with a mixed basket comparator (PCC of doxorubicin or paclitaxel). The modelled population is consistent with the reimbursement request and is aligned with the trial population of KEYNOTE-775, a multi-centre, open-label, randomized, phase III trial involving patients with advanced endometrial cancer.1
PEM is supplied in single-use vials at a submitted price of $4,400 per injectable 4 mL vial (100 mg/mL). The recommended dose is 200 mg every 3 weeks or 400 mg every 6 weeks for up to 24 months (i.e., 35 doses of 200 mg or 18 doses of 400 mg) or until unacceptable toxicity or disease progression, whichever is longer, in combination with LEN 20 mg orally once daily until unacceptable toxicity or disease progression.2 The dosages of all comparator treatments were based on the doses administered in the KEYNOTE-775 trial (i.e., doxorubicin = 60 mg/m2 every 21 days; paclitaxel = 80 mg/m2 weekly for 3 out of every 4 weeks).3
The sponsor’s calculated cost of PEM was $8,800 every 3 weeks, resulting in a weekly per-cycle cost of $2,933. For LEN, the sponsor applied a dose reduction schedule based on a range of doses applied in the KEYNOTE-775 trial and calculated a weighted average drug cost of $864 per week in week 1, eventually decreasing to $378 weekly after consistent dose reductions throughout the model horizon. The cost of paclitaxel was $110 every 4 weeks, resulting in a weekly per-cycle cost of $27. For doxorubicin, the cost was $781 every 3 weeks, resulting in a weekly per-cycle cost of $260. An administration cost of $216 was added to each administration of PEM, doxorubicin, and paclitaxel.4
The clinical outcomes of interest were QALYs and LYs. The economic analysis was undertaken over a lifetime horizon (20 years) from the perspective of a publicly funded health care payer. A weekly cycle length was used with no half-cycle correction. Costs and outcomes were discounted at a rate of 1.5% annually.
Model Structure
A PSM was submitted to capture the long-term costs and effects associated with the natural history of advanced endometrial cancer that is not MSI-H or dMMR over the model time horizon. The model consisted of 3 primary health states (PFS, progressive disease, and death). The proportion of patients who were progression-free, experienced progressive disease, or died at any time over the model time horizon was derived from non–mutually exclusive KM survival curves. OS and PFS curves were derived from the KEYNOTE-775 trial for PEM + LEN and PCC (doxorubicin or paclitaxel) and were used to determine the proportion of patients in each health state (refer to Appendix 3, Figure 1). These survival curves were used to inform the proportion of patients who were in PFS, progressive disease, or death states at any time over the model’s time horizon. Specifically, all patients entered the model in the progression-free state; the proportion of progression-free patients was derived as the area under the PFS curve, while the proportion of patients with progressed disease was derived by the difference in the area under the curve between the OS and PFS curves. Progression in the KEYNOTE-775 trial was defined as the time from the date of randomization to the date of the first documentation of disease progression (according to Response Evaluation Criteria in Solid Tumours version 1.1) or death. OS was defined as the time from the date of randomization to the date of death from any cause. Time on treatment (TOT) was calculated for PEM and LEN separately, using the KEYNOTE-775 trial data to identify the proportion of patients who were alive and remained on treatment. Patients were assumed to remain on PEM + LEN for as long as they remained on LEN.
Model Inputs
The patient cohort comprised adult patients with advanced endometrial cancer (i.e., pMMR) whose baseline characteristics mainly reflected the pivotal trial, KEYNOTE-775.1 The median age of patients in the model was 63.7 years, with a mean weight of ||||||| kg and a body surface area of |||||||||| m2.5
Key clinical efficacy inputs (i.e., OS and PFS) and TOT for PEM + LEN were based on the results of the KEYNOTE-775 trial (i.e., data cut-off date of October 26, 2020). KM estimates of PFS, OS, and time to treatment discontinuation for PEM + LEN and PCC from the trial period were used to fit parametric survival curves to extrapolate the treatment effect beyond the observed trial data (maximum duration of follow-up = 26.9 months) at 26-week and 10-week cut points over the entire model time horizon (i.e., 20 years). To determine the best fitting distribution, diagnostic plots, goodness-of-fit statistics, visual inspection, and clinical expectations were assessed regarding long-term progression risk and survival. The combined KM and parametric log logistic function was chosen parametric survival distribution for the OS and PFS of both PEM + LEN and PCC. TOT data for PEM + LEN were obtained from the KEYNOTE-775 trial, and the parametric survival distribution chosen to extrapolate TOT for PEM + LEN over the lifetime was the generalized gamma distribution.4
Health state utility values were estimated using a regression model that adjusted for baseline utility and progression status using patients’ health-related quality of life data collected in the KEYNOTE-775 trial through the EuroQol 5-Dimensions 5-Levels questionnaire and EuroQol 5-Dimensions 5-Levels values based on the Canadian population. Utility data were fit to a series of linear mixed-effects regression models, and in the economic base case, the sponsor used the regression model that accounted for the effect of adverse events (AEs) and progression status (PFS or progressive disease). The sponsor incorporated utility values in the base case that differed by health state and whether patients were on or off treatment. The utility weight assigned to the PFS health state (0.794) for patients on or off treatment was greater than the utility weight assigned to the progressed-disease health state (0.752) for patients who had progressed and were on or off subsequent treatment.4
The model included drug acquisition and administration costs, costs associated with drug infusion time, subsequent treatment costs, dMMR or SI-H endometrial cancer screening costs, AEs, resource use for each health state, and terminal care. Drug acquisition costs for PCC and subsequent therapies were obtained from the IQVIA database and weighted by the distribution of patients on these therapies in the KEYNOTE-775 trial. Drug administration costs for therapies administered by IV infusion (PEM, paclitaxel, and doxorubicin) were assumed to be the same and estimated based on a Canadian cost-effectiveness study of systemic therapies for metastatic pancreatic cancer.6,7 The majority of AE costs were informed by the Ontario Case Costing Initiative.8 Others were derived from published literature.9,10
Summary of Sponsor’s Economic Evaluation Results
All analyses were run probabilistically (1,000 iterations for the base case and scenario analyses). The deterministic and probabilistic results were similar. The probabilistic findings are presented in this section.
Base-Case Results
In the sponsor’s base case, PEM + LEN was associated with an incremental cost of $158,046 and 1.26 QALYs over a 20-year time horizon (Table 3), which resulted in an ICER of $125,808 per QALY. In the sponsor’s base case, 7% of PEM + LEN patients and 2% of PCC patients were alive at the 20-year time horizon.
Results were driven by the OS projections, which predicted substantial differences in total life-years and the increased drug acquisition costs associated with the combination therapy. Because the sponsor did not present the proportion of incremental benefit (QALYs) derived within the trial compared with the extrapolation based on the submitted model, CADTH undertook proxy analyses and determined that approximately 85% of the incremental benefit associated with PEM + LEN was derived from the extrapolated period.
Table 3: Summary of the Sponsor’s Economic Evaluation Results
Drug | Total costs ($) | Incremental costs ($) | Total LYs | Incremental LYs | Total QALYs | Incremental QALYs | ICER vs. PCC ($/QALY) |
|---|---|---|---|---|---|---|---|
PCC | 40,428 | Reference | 2.10 | Reference | 1.53 | Reference | Reference |
PEM + LEN | 198,474 | 158,046 | 3.94 | 1.84 | 2.78 | 1.26 | 125,808 |
ICER = incremental cost-effectiveness ratio; LY = life-year; PEM + LEN = pembrolizumab plus lenvatinib combination therapy; PCC = physician’s choice of chemotherapy; QALY = quality-adjusted life-year; vs. = versus.
Source: Sponsor’s pharmacoeconomic submission.4
Sensitivity and Scenario Analysis Results
The sponsor conducted several sensitivity and scenario analyses. These included varying the discount rate, time horizon, and market share of doxorubicin and paclitaxel in PCC as well as excluding MSI-H testing costs, excluding AE costs, excluding subsequent treatment costs, excluding treatment stopping rules, including treatment waning, including AE disutilities, and modifying assumptions around subsequent treatment shares and survival extrapolations. The ICER was most sensitive to the scenario that explored the OS of PEM + LEN with a KM plus generalized gamma distribution fit at 26 weeks (ICER = $149,295 per QALY) and the OS of PEM + LEN with a KM plus log-logistic distribution fit at 52 weeks (ICER = $161,046 per QALY). The ICER was robust to other changes.
CADTH Appraisal of the Sponsor’s Economic Evaluation
CADTH identified several key limitations to the sponsor’s analysis that have notable implications for the economic analysis.
Long-term extrapolations of the OS and PFS data are likely overestimated. The sponsor fit several parametric survival curves to extrapolate OS and PFS for patients who received PEM + LEN and PCC over the model’s time horizon (20 years) based on KM data at a cut point of 26 weeks. The log-logistic function was chosen in all cases based on statistical fit, the fit to the observed hazard function, clinical plausibility, and external validation; however, these extrapolations led to survival results that the clinical experts consulted by CADTH considered clinically implausible. The clinical experts consulted by CADTH indicated that it is unlikely for 22% and 13% of patients to remain alive at year 5 and year 10, respectively, as suggested by the sponsor’s curves for the OS of PEM + LEN and PCC extrapolated beyond the observed period of the trial. Incremental QALYs for PEM + LEN and PCC are likely overestimated as a result of long-term OS extrapolations. CADTH selected alternate parametric distributions for the OS and PFS of PEM + LEN and PCC that were more clinically plausible based on expert feedback.
As part of the base case, CADTH chose gamma distributions for the OS data for both treatments and Weibull distributions for the PFS data. This was done in consultation with clinical experts. CADTH conducted a scenario analysis in which Weibull distributions were chosen for both OS and PFS.
The PSM structure applied in the economic analysis suggests a post-progression survival benefit, the magnitude of which is uncertain. The sponsor’s base-case results suggest that PEM + LEN is associated with longer survival after disease progression. Specifically, the sponsor’s results imply a post-progression survival benefit for patients receiving PEM + LEN relative to PCC such that roughly 70% of the incremental survival (1.28 life-years) would be experienced after patients have experienced disease progression and have discontinued PEM + LEN (Table 10). The sponsor’s use of a PSM introduces structural assumptions about the relationship between PFS and OS that likely do not accurately reflect causal relationships within the disease pathway. These assumptions may produce a post-progression survival bias that favours PEM + LEN. Due to the structural independence between the OS and PFS end points assumed in a PSM, extrapolations for each end point may reflect within-trial trends in the rates of progression and death. Given that the mechanism by which PEM + LEN would lead to post-progression survival benefits is uncertain, CADTH reached out to the sponsor for clarification. In response, the sponsor provided median OS and PFS data from the KEYNOTE-775 trial that suggested there may be evidence of post-progression survival gains; however, the trial was not powered to detect such a difference. While this finding suggests the possibility of a post-progression survival benefit, the evidence was highly uncertain, and the magnitude of the difference observed between median OS and PFS for PEM + LEN compared to PCC was not proportionate to the quantity of benefit produced in the sponsor’s base case.
CADTH was unable to address this limitation because the extent to which the implied post-progression benefit was due to the effect of treatment with PEM + LEN, structural bias within the PSM, or limitations within the comparator efficacy evidence could not be determined.
The sponsor’s choice of comparators does not reflect Canadian clinical practice for the indicated population. In the economic model, the sponsor could not include several single-drug and combination chemotherapy regimens (i.e., carboplatin, cisplatin, carboplatin plus docetaxel, carboplatin plus doxorubicin, and cisplatin plus doxorubicin) due to lack of evidence. Nor could it include hormone therapies (i.e., medroxyprogesterone, megestrol, tamoxifen, and aromatase inhibitors [exemestane, anastrozole, letrozole]) that were identified as relevant comparators for the indicated population by the clinical experts consulted by CADTH. Importantly, there are no specific drugs that are approved as second-line therapies, as noted by the clinician groups that provided input for the current review. However, this feedback notes that platinum-containing therapies may be used. The clinical experts consulted by CADTH noted that 20% to 60% of patients would likely be re-treated with a platinum-containing therapy if they had previously responded well and had a sufficiently long disease-free period. Otherwise, paclitaxel and liposomal doxorubicin were the most frequently used approaches.
CADTH was unable to address this limitation. The cost-effectiveness of PEM + LEN compared to other chemotherapy regimens, including platinum re-treatment, is unknown.
Pricing and dosing-related inputs for LEN were applied incorrectly, which underestimated the total costs for LEN. In the economic model, several dosing-related inputs were required (publicly available list prices, number of capsules per package for each formulation, and costs per treatment cycle). First, the sponsor selected drug costs for LEN that were sourced from the Institut national d’excellence en santé et en services sociaux in Quebec;11 however, these are not reflective of pan-Canadian pricing. Second, the sponsor inappropriately assumed 30 capsules per daily-dose carton containing 6 blister cards for each packaging configuration, even though the number of capsules per package varies for each formulation, as indicated in the product monograph.12 Lastly, the sponsor’s calculation of costs per dose of LEN was incorrect because the sponsor multiplied the number of milligrams per capsule by the proportion of the pack used per dose without considering the correct number of capsules per pack, as noted. Altogether, these pricing- and dosing-related inputs underestimated the total costs of LEN over the time horizon.
CADTH addressed this limitation by revising LEN pricing (for 4 mg, 8 mg, 10 mg, 14 mg, and 20 mg) to reflect pricing from the Ontario Exceptional Access Program and revising the sponsor’s formulas to correctly calculate LEN treatment costs.
Health state utility values lacked face validity: In the sponsor’s base case, utility values for patients in the progressed-disease health state (0.752) were broadly similar to the values for those who were in the progression-free health state (0.794). The clinical experts consulted by CADTH indicated that patient quality of life typically worsens with disease progression. As such, health state utility values for the progressed-disease health state lack face validity and likely overestimate patients’ quality of life post-progression in favour of PEM. The magnitude of bias in favour of PEM remains unknown, resulting in additional uncertainty about the impact of health state utility values on the ICER.
CADTH was unable to address this limitation in reanalysis. In a scenario analysis, CADTH explored the impact of alternate utility values for the progression-free and progressed-disease health states using the sponsor-provided option to do so.
Drug pricing for PCC (i.e., paclitaxel and doxorubicin) did not reflect the lowest publicly available list prices, and treatment costs were calculated by summing the costs for more than 1 formulation in consideration of partial (50%) drug wastage: In the economic model, the sponsor selected drug costs for paclitaxel that were sourced from the Institut national d’excellence en santé et en services sociaux (INESSS) in Quebec,11 while the price for doxorubicin was not based on the lowest publicly available pricing in the IQVIA DeltaPA database.13 Additionally, the sponsor applied a drug wastage setting that accounted only for partial drug wastage or vial sharing (i.e., 50% of the remaining vial quantity would be shared between patients and the remainder would result in product loss) for comparator treatments administered by IV injection. In consideration of partial wastage, the sponsor further calculated drug costs for paclitaxel and doxorubicin by summing the costs of the 2 modelled formulations of each treatment rather than selecting the treatment that was least costly. These issues overestimate the drug costs for the PCC. However, the clinical experts consulted by CADTH indicated that policies around vial sharing may vary across Canadian jurisdictions, and that partial or full drug wastage might be an area of uncertainty.
CADTH addressed this limitation by revising the pricing for comparators based on the lowest publicly available pricing listed in the DeltaPA; accounting for full (100%) drug wastage for drug prices in a scenario analysis; and revising cost formulas to correctly incorporate full drug wastage.
Additionally, the following key assumptions were made by the sponsor and have been appraised by CADTH (Table 4).
CADTH Reanalyses of the Economic Evaluation
Base-Case Results
The CADTH base case was derived by making changes to model parameter values and assumptions in consultation with clinical experts. These changes, summarized in Table 5, included alternate survival extrapolations for both OS and PFS and updated drug costs for LEN, paclitaxel, and doxorubicin, according to appropriate sources.
In the CADTH base case, PEM + LEN was associated with estimated total costs and QALYs of $200,838 and 1.35, respectively, compared with total costs and QALYs of $50,616 and 0.94, respectively, for patients receiving PCC. The ICER for PEM + LEN compared to PCC was $366,399 per QALY, and the probability of cost-effectiveness at a $50,000 per QALY willingness-to-pay threshold was 0%. Results of the stepped reanalysis are available in Table 6, with full disaggregated results available in Appendix 4, Table 11. In the CADTH base case, approximately 52% of the incremental benefit associated with PEM + LEN was derived from the extrapolated period.
Table 4: Key Assumptions of the Submitted Economic Evaluation (Not Noted as Limitations to the Submission)
Sponsor’s key assumption | CADTH comment |
|---|---|
The baseline characteristics of KEYNOTE-775 trial participants was representative of the Canadian patient population. | Appropriate. The clinical experts consulted by CADTH indicated that the baseline patient characteristics of patients in the KEYNOTE-775 trial were representative of patients with pMMR endometrial cancer in Canadian clinical practice. They expressed no concerns about the generalizability of the study findings. |
The sponsor assumed a lifetime time horizon of 20 years. | Appropriate. This time horizon is adequate to capture all lifetime associated costs and outcomes for the indicated population. |
The sponsor’s choice of chemotherapy treatments as part of the PCC group included doxorubicin and paclitaxel. The sponsor further assumed that 25% of patients would receive paclitaxel and 75% would be on doxorubicin. | Appropriate. The clinical experts consulted by CADTH indicated that the distribution of treatment use for patients who received paclitaxel and doxorubicin in the economic model aligned with the distribution of treatment use in Canadian clinical practice. Importantly, there is no true standard of care for second-line treatment in the indicated population. |
The distribution of subsequent therapies in the model was assumed to be based on the distribution of subsequent treatment use in the KEYNOTE-775 trial, a multi-centre, open-label, randomized, phase III trial of pembrolizumab plus lenvatinib.4 | Appropriate, according to the clinical experts consulted by CADTH. |
The sponsor assumed that 28% and 48% of patients who progressed on PEM + LEN and PCC, respectively, would likely receive subsequent therapies, based on the KEYNOTE-775 trial. | Inappropriate. The clinical experts consulted by CADTH indicated that a higher proportion of patients (approximately 60%) would be eligible to receive subsequent therapy following treatment with PEM + LEN. While the sponsor’s assumption likely underestimates the proportion of patients who receive subsequent therapy following initial treatment with pembrolizumab, this parameter has minimal to no impact on model results. |
In the economic model, the sponsor accounted for lenvatinib dose reductions, which affect total drug costs. The dose reduction schedule for lenvatinib was based on the KEYNOTE-775 trial, This schedule was used to govern treatment-related toxicities in each treatment cycle. Over the course of the trial, patients were administered lenvatinib at a range of doses (4 mg, 8 mg, 10 mg, 14 mg, 20 mg, and 40 mg), starting with 20 mg. The number of patients who received each dose of lenvatinib in each week was used to calculate the cost of administered lenvatinib. | Likely appropriate. However, the dose reduction schedule could not be validated by CADTH. |
The probabilities of patients experiencing grade 3+ AEs were derived from the KEYNOTE-775 trial for PEM + LEN and reflected those that occurred in greater than 2% of patients.4 | Appropriate. The CADTH clinical review indicated that the most common serious AEs reported in the KEYNOTE-775 trial (≥ 2% in either arm) were hypertension, urinary tract infection, acute kidney injury, colitis, pyrexia, decreased appetite, febrile neutropenia, and neutropenia. These AEs were included in the submitted model. |
In the economic model, the sponsor did not apply treatment waning effects for pembrolizumab over the lifetime time horizon.4 | Appropriate. The sponsor provided justification in its submission that pembrolizumab has previously been shown to sustain a long-term effect on overall survival in patients with endometrial cancer who discontinue pembrolizumab at 2 years. The clinical experts consulted by CADTH affirmed this based on their clinical knowledge and on literature demonstrating the longer-term immunotherapeutic effects of pembrolizumab after treatment discontinuation. |
The sponsor included dMMR or MSI-H screening costs as 1-time costs. | Appropriate. |
AE = adverse event; dMMR = mismatch repair deficient; MSI-H = microsatellite instability-high; PCC = physician’s choice of chemotherapy; PEM + LEN = pembrolizumab plus lenvatinib combination therapy; pMMR = mismatch repair proficient.
Table 5: CADTH Revisions to the Submitted Economic Evaluation
Stepped analysis | Sponsor’s value or assumption | CADTH value or assumption |
|---|---|---|
Changes to derive the CADTH base case | ||
1. Extrapolation of OS curves for both treatments | Log-logistic | Gamma |
2. Extrapolation of PFS curves for both treatments | Log-logistic | Weibull |
3. Lenvatinib pricing | Lenvatinib (20 mg): $165.64 Lenvatinib (14 mg): $110.42 Lenvatinib (10 mg): $71.64 Lenvatinib (8 mg): $65.14 Lenvatinib (4 mg): $32.57 | Lenvatinib (20 mg): $175.41 Lenvatinib (14 mg): $116.93 Lenvatinib (10 mg): $75.28 Lenvatinib (8 mg): $68.64 Lenvatinib (4 mg): $33.97 |
4. Comparator pricing | Paclitaxel: $5.27 per 30 mg; $17.56 per 100 mg Doxorubicin: $255.00 per 50 mg; $770.00 per 200 mg | Paclitaxel: $300.00 per 30 mg; $1,196.80 per 100 mg Doxorubicin: $252.25 per 50 mg; $770.00 per 200 mg |
CADTH base case | Reanalyses 1 + 2 + 3 + 4 | |
OS = overall survival; PFS = progression-free survival.
Table 6: Summary of the Stepped Analysis of the CADTH Reanalysis Results (Deterministic)
Stepped analysis | Drug | Total costs ($) | Total LYs | Total QALYs | Sequential ICER ($/QALY) |
|---|---|---|---|---|---|
Sponsor’s base case | PCC | 40,414 | 2.08 | 1.52 | Reference |
PEM + LEN | 194,086 | 3.93 | 2.78 | 121,439 | |
CADTH reanalysis 1: OS extrapolations | PCC | 40,503 | 1.24 | 0.95 | Reference |
PEM + LEN | 194,362 | 1.78 | 1.36 | 371,997 | |
CADTH reanalysis 2: PFS extrapolations | PCC | 40,258 | 2.08 | 1.51 | Reference |
PEM + LEN | 195,564 | 3.93 | 2.77 | 123,409 | |
CADTH reanalysis 3: lenvatinib pricing | PCC | 40,440 | 2.08 | 1.52 | Reference |
PEM + LEN | 195,785 | 3.93 | 2.78 | 122,760 | |
CADTH reanalysis 4: paclitaxel and doxorubicin pricing | PCC | 50,479 | 2.08 | 1.52 | Reference |
PEM + LEN | 195,306 | 3.93 | 2.78 | 114,449 | |
CADTH base case: | PCC | 50,505 | 1.24 | 0.94 | Reference |
PEM + LEN | 198,915 | 1.78 | 1.35 | 361,916 | |
CADTH base case: | PCC | 50,616 | 1.24 | 0.94 | Reference |
PEM + LEN | 200,838 | 1.78 | 1.35 | 366,399 |
ICER = incremental cost-effectiveness ratio; LY = life-year; OS = overall survival; PCC = physician’s choice of chemotherapy; PEM + LEN = pembrolizumab plus lenvatinib combination therapy; PFS = progression-free survival; QALY = quality-adjusted life-year.
Scenario Analysis Results
CADTH undertook price reduction analyses based on the sponsor’s and CADTH’s base case. The CADTH base case suggested that even with a 100% reduction in the price of PEM, PEM + LEN would not be cost-effective compared to PCC.
Table 7: CADTH Price Reduction Analyses
Analysis | ICERs for PEM + LEN vs. PCC ($/QALY) | |
|---|---|---|
Price reduction | Sponsor base case | CADTH reanalysis |
No price reduction | 125,808 | 366,399 |
10% | 114,312 | 340,750 |
20% | 104,849 | 308,088 |
30% | 94,438 | 277,594 |
40% | 85,690 | 247,679 |
50% | 75,898 | 215,643 |
60% | 64,603 | 185,093 |
70% | 55,850 | 155,224 |
76% | 49,505 | 136,982 |
80% | 44,499 | 124,984 |
90% | 34,642 | 94,670 |
99% | 26,388 | 66,542 |
ICER = incremental cost-effectiveness ratio; PCC = physician’s choice of chemotherapy; PEM + LEN = pembrolizumab plus lenvatinib combination therapy; QALY = quality-adjusted life-year; vs. = versus.
CADTH undertook several scenario analyses to determine the impact of alternative assumptions on the cost-effectiveness of PEM + LEN in the base case, which are outlined as follows:
used Weibull extrapolation for OS in the PEM + LEN and PCC treatments
used the sponsor’s original log-logistic extrapolation for OS in the PEM + LEN and PCC treatments
derived alternative health state utility values from the sponsor’s linear utility regression, which incorporated AEs and time to death
derived alternative health state utility values from the sponsor’s linear utility regression, which incorporated AEs, progression status, and on-treatment effect
assumed full (100%) drug wastage for unused product (doxorubicin and paclitaxel).
The results of these analyses are presented in Appendix 4, Table 12. The scenario in which a log-logistic distribution was assumed for OS had the largest impact, resulting in an ICER of $117,818 per QALY. Other tested scenarios did not have a meaningful impact on results.
Issues for Consideration
The submitted economic model did not include an option to assess a weight-based dosing regimen for PEM, but rather 1 fixed-dose regimen (i.e., 200 mg every 3 weeks) based on the Health Canada indication and the KEYNOTE-775 trial’s study protocol. The clinical experts consulted by CADTH for the CADTH review of dostarlimab noted that PEM and dostarlimab may be administered as either a flat-rate dose or through weight-based dosing based on the maximum capped dose, according to evidence from several pharmacokinetic studies in support of weight-based dosing of immunotherapies. Given that dostarlimab is an immunotherapy with monoclonal antibodies similar to PEM, weight-based dosing for both products is likely to be similarly implemented in Canadian practice — despite the absence of this weight-based dosing information in its product monograph — and will likely affect associated drug costs. However, the cost-effectiveness of PEM’s weight-based dosing regimen is unknown.
Overall Conclusions
The CADTH clinical review noted that results from the KEYNOTE-775 trial showed that compared to PCC, PEM + LEN combination therapy showed a statistically significant and clinically meaningful benefit in terms of OS and PFS in the treatment of adult patients with advanced pMMR (i.e., not MSI-H or dMMR) endometrial carcinoma who have disease progression following prior platinum-based systemic therapy and are not candidates for curative surgery or radiation. The clinical experts consulted for this review indicated that the safety profile of PEM + LEN observed in this study appeared consistent with the known safety profile of each individual drug (PEM or LEN) and that no additional safety signals were identified. The clinical review noted that a longer duration of follow-up may be needed to further confirm the long-term benefit of PEM + LEN combination therapy over PCC chemotherapy in this population.
CADTH identified several limitations in the sponsor’s pharmacoeconomic analysis that have notable implications on the cost-effectiveness results. First, the sponsor’s chosen survival extrapolations overestimated the survival of patients in the model on either therapy compared to clinical expectations. The PSM also produced an apparent post-progression survival benefit that may be the result of a structural bias and favours PEM + LEN. CADTH updated the costs of concomitant and comparator medications (LEN, doxorubicin, and paclitaxel) according to publicly available sources. CADTH also noted limitations in the comparator choice, health state utility value, and drug wastage calculations. CADTH made various changes to the base case to address these limitations. The CADTH reanalysis resulted in an ICER for PEM + LEN of $366,399 per QALY compared to PCC (incremental costs = $150,222; incremental QALYs = 0.41; incremental life-years = 0.54), with a 0% probability of cost-effectiveness at a $50,000 per QALY threshold. Even with a 100% price reduction for PEM, PEM + LEN would not be cost-effective at this threshold.
Despite the benefits noted in the CADTH clinical review pertaining to OS and PFS, the extrapolation of these data beyond the period for which KM data are available is uncertain, with approximately 85% of the incremental benefit with PEM + LEN coming from the extrapolated period (compared to 52% in the CADTH base case). In addition, uncertainty remains in the analysis regarding the choice of comparators and the impact of re-treatment with platinum therapy; CADTH could not address these. CADTH performed scenario analyses of the OS extrapolation, using a Weibull or log-logistic distribution for both treatments. These analyses indicate that results are sensitive to assumptions about the long-term OS benefit of PEM + LEN compared to PCC, which was noted as a gap in the available trial evidence. The cost-effectiveness results were robust to other parameters varied in the CADTH reanalysis and scenario analyses.
The sponsor did not include platinum-based therapies as comparators within its model. The clinical experts consulted by CADTH noted that 20% to 60% of patients would be eligible to be re-treated with platinum-based drugs if they experienced a sufficient disease-free period following their initial discontinuation of platinum. The cost-effectiveness of PEM + LEN compared to platinum rechallenge is unknown.
References
1.Clinical Study Report: P775V01MK3475. A multicenter, open-label, randomized, phase 3 trial to compare the efficacy and safety of lenvatinib in combination with pembrolizumab versus treatment of physician’s choice in participants with advanced endometrial cancer [internal sponsor's report]. Woodcliff Lake (NJ): Eisai; 2021.
2.PrKeytruda® (pembrolizumab): solution for infusion 100 mg/4 mL vial [product monograph]. Kirkland (QC): Merck Canada; 2021 Nov 24.
3.Makker V, Colombo N, Casado Herraez A, et al. Lenvatinib plus Pembrolizumab for Advanced Endometrial Cancer. N Engl J Med. 2022;386(5):437-448. PubMed
4.Pharmacoeconomic evaluation [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Keytruda (pembrolizumab), powder for solution for infusion 50 mg, solution for infusion 100 mg/4 mL vial with levantinib. Kirkland (QC): Merck Canada; 2022 Mar 4.
5.Pharmacoeconomic evaluation [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Keytruda (pembrolizumab), powder for solution for infusion 50 mg, solution for infusion 100 mg/4 mL vial[internal sponsor's package]. Toronto (ON): Merck; 2022 Jan 6.
6.Tam VC, Ko YJ, Mittmann N, et al. Cost-effectiveness of systemic therapies for metastatic pancreatic cancer. Curr Oncol. 2013;20(2):e90-e106. PubMed
7.Bank of Canada. Consumer Price Index, 2000 to present. 2021: https://www.bankofcanada.ca/rates/price-indexes/cpi/. Accessed 2022 Mar 23.
8.Ontario Case Costing Initiative (OCCI). Toronto (ON): Ontario Health and Long-Term Care; 2017: https://data.ontario.ca/dataset/ontario-case-costing-initiative-occi. Accessed 2022 Feb 28.
9.Dranitsaris G, Maroun J, Shah A. Severe chemotherapy-induced diarrhea in patients with colorectal cancer: a cost of illness analysis. Support Care Cancer. 2005;13(5):318-324. PubMed
10.Lathia N, Mittmann N, DeAngelis C, et al. Evaluation of direct medical costs of hospitalization for febrile neutropenia. Cancer. 2010;116(3):742-748. PubMed
11.AvastinMC - cancer de l'ovaire. Avis au ministre. Québec (QC): INESSS; 2016: https://www.inesss.qc.ca/fileadmin/doc/INESSS/Inscription_medicaments/Avis_au_ministre/Aout_2016/Avastin_2016_06.pdf. Accessed 2022 Mar 23.
12.Lenvima lenvatinib capsules 4 mg and 10 mg Lenvatinib (as lenvatinib mesylate) [product monograph]. Mississauga (ON): Eisai Limited; 2021: https://pdf.hres.ca/dpd_pm/00062284.PDF. Accessed 2022 Apr 1.
13.DeltaPA. [Ottawa (ON)]: IQVIA; 2021: https://www.iqvia.com/. Accessed 2022 Feb 28.
14.Ontario Ministry of Health, Ontario Ministry of Long-Term Care. Ontario drug benefit formulary/comparative drug index. 2021; https://www.formulary.health.gov.on.ca/formulary/. Accessed 2022 Feb 28.
15.Cancer Care Ontario: funded evidence-informed regimens. 2022; https://www.cancercareontario.ca/en/drugformulary/regimens. Accessed 2022 Feb 28.
16.CRBP: carboplatin for palliative therapy [drug monograph]. Drug formulary regimens. Toronto (ON): Cancer Care Ontario; 2022: https://www.cancercareontario.ca/en/drugformulary/regimens/monograph/45551. Accessed 2022 Mar 1.
17.Budget Impact Analysis [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Keytruda (pembrolizumab), powder for solution for infusion 50 mg, solution for infusion 100 mg/4 mL vial[internal sponsor's package]. Toronto (ON): Merck; 2022 Jan 6.
18.Merck. PrKeytruda® (pembrolizumab): solution for infusion 100 mg/4 mL vial [product monograph]. Kirkland (QC): Merck Canada; 2021 (2022 Apr 12).
19.CRBP: carboplatin for adjuvant therapy [product monograph]. Drug formulary regimens. Toronto (ON): Cancer Care Ontario; 2022: https://www.cancercareontario.ca/en/drugformulary/regimens/monograph/48166#DrugRegimen. Accessed 2022 Feb 9.
20.CISP: cisplatin for adjuvant, palliative therapy [product monograph]. Drug formulary regimens. Toronto (ON): Cancer Care Ontario; 2022: https://www.cancercareontario.ca/en/drugformulary/regimens/monograph/46931#DrugRegimen. Accessed 2022 Feb 10.
21.PACL(W): paclitaxel weekly for palliative therapy [product monograph]. Drug formulary regimens. Toronto (ON): Cancer Care Ontario; 2022: https://www.cancercareontario.ca/en/drugformulary/regimens/monograph/50491#DrugRegimen. Accessed 2022 Feb 9.
22.DOXO [product monograph]. Drug formulary regimens. Toronto (ON): Cancer Care Ontario; 2020: https://www.cancercareontario.ca/en/drugformulary/drugs/DOXOrubicin. Accessed 2022 Feb 10.
23.CRBPPACL: paclitaxel-carboplatin for adjuvant, palliative therapy [product monograph]. Drug formulary regimens. Toronto (ON): Cancer Care Ontario; 2022: https://www.cancercareontario.ca/en/drugformulary/regimens/monograph/45981#DrugRegimen. Accessed 2022 Feb 9.
24.CRBPDOCE: docetaxel-carboplatin for adjuvant therapy [product monograph]. Drug formulary regimens. Toronto (ON): Cancer Care Ontario; 2022: https://www.cancercareontario.ca/en/drugformulary/regimens/monograph/48181. Accessed 2022 Feb 9.
25.CISPDOXO: cisplatin-doxorubicin for adjuvant, palliative therapy [product monograph]. Drug formulary regimens. Toronto (ON): Cancer Care Ontario; 2022: https://www.cancercareontario.ca/en/drugformulary/regimens/monograph/45761#DrugRegimen. Accessed 2022 Feb 10.
26.Prpms-Medroxyprogesterone (medroxyprogesterone acetate): 2.5 mg, 5 mg and 10 mg tablets [product monograph]. Montreal (QC): Pharmascience; 2004 May 10: https://pdf.hres.ca/dpd_pm/00000786.PDF. Accessed 2022 Feb 10.
27.MEDR: medroxyprogesterone for adjuvant, curative therapy [product monograph]. Drug formulary regimens. Toronto (ON): Cancer Care Ontario; 2022: https://www.cancercareontario.ca/en/drugformulary/regimens/monograph/48176#DrugRegimen. Accessed 2022 Feb 10.
28.MEGE: megestrol for palliative therapy [product monograph]. Drug formulary regimens. Toronto (ON): Cancer Care Ontario; 2022: https://www.cancercareontario.ca/en/drugformulary/regimens/monograph/46956#DrugRegimen. Accessed 2022 Feb 10.
29.TMXF: tamoxifen for palliative therapy [product monograph]. Drug formulary regimens. Toronto (ON): Cancer Care Ontario; 2022: https://www.cancercareontario.ca/en/drugformulary/regimens/monograph/46961#DrugRegimen. Accessed 2022 Feb 10.
30.PrLetrozole (letrozole): 2.5 mg tablets [product monograph]. Toronto (ON): Teva Canada; 2010 Feb 10: https://pdf.hres.ca/dpd_pm/00010490.PDF. Accessed 2022 Feb 10.
31.EXEM: exemestane for palliative therapy [product monograph]. Drug formulary regimens. Toronto (ON): Cancer Care Ontario; 2022: https://www.cancercareontario.ca/en/drugformulary/regimens/monograph/44731#DrugRegimen. Accessed 2022 Feb 10.
32.Canadian Cancer Statistics Advisory Committee. Canadian cancer statistics: a 2018 special report on cancer incidence by stage. Toronto (ON): Canadian Cancer Society; 2018: https://cdn.cancer.ca/-/media/files/research/cancer-statistics/2018-statistics/canadian-cancer-statistics-2018-en.pdf. Accessed 2022 Feb 9.
33.Canadian Cancer Society. Canadian Cancer Statistics 2019. 2019; https://cdn.cancer.ca/-/media/files/research/cancer-statistics/2019-statistics/canadian-cancer-statistics-2019-en.pdf. Accessed 2022 Feb 9.
34.Uterine cancer: statistics. Cancer.Net. Alexandria (VA): American Society of Clincial Oncology; 2021: https://www.cancer.net/cancer-types/uterine-cancer/statistics. Accessed 2022 Feb 9.
35.Key statistics for endometrial cancer. Atlanta (GA): American Cancer Society; 2021: https://www.cancer.org/cancer/endometrial-cancer/about/key-statistics.html. Accessed 2022 Feb 9.
36.Brenner DR, Weir HK, Demers AA, et al. Projected estimates of cancer in Canada in 2020. CMAJ. 2020;192(9):E199-E205. PubMed
37.Endometrium. Vancouver (BC): Provincial Health Services Authority; 2018: http://www.bccancer.bc.ca/health-professionals/clinical-resources/cancer-management-manual/gynecology/endometrium. Accessed 2022 Mar 13.
38.Canadian Cancer Statistics Advisory Committee. Canadian Cancer Statistics 2021. Toronto (ON): Canadian Cancer Society; 2021: https://cdn.cancer.ca/-/media/files/research/cancer-statistics/2021-statistics/2021-pdf-en-final.pdf. Accessed 2022 Feb 9.
39.GiotrifMC – cancer du poumon non à petites cellules. Québec (QC): INESSS; 2014: https://www.inesss.qc.ca/fileadmin/doc/INESSS/Inscription_medicaments/Avis_au_ministre/Juin_2014/Giotrif_2014_06_CAV.pdf. Accessed 2022 Feb 9.
40.Extraits d'avis dont la publication avait été reportée. Québec (QC): INESSS; 2016: https://www.inesss.qc.ca/fileadmin/doc/INESSS/Inscription_medicaments/Avis_au_ministre/Aout_2016/Avis_extraits_publication_reportee.pdf. Accessed 2022 Feb 9.
41.Reaume MN, Leighl NB, Mittmann N, et al. Economic analysis of a randomized phase III trial of gemcitabine plus vinorelbine compared with cisplatin plus vinorelbine or cisplatin plus gemcitabine for advanced non-small-cell lung cancer (Italian GEMVIN3/NCIC CTG BR14 trial). Lung Cancer. 2013;82(1):115-120. PubMed
42.IQVIA. Delta PA. 2021; https://www.iqvia.com/. Accessed date unknown.
43.Treating advanced cancer of the uterus. Woolloomooloo (AU): Cancer Council NSW; 2021: https://www.cancercouncil.com.au/uterine-cancer/treatment/advanced-uterine-cancer/. Accessed 2022 Mar 11.
44.Endometrial cancer. (Clinical practice guideline GYNE-002. Version 5). Edmonton (AB): Alberta Health Services; 2015: https://www.albertahealthservices.ca/assets/info/hp/cancer/if-hp-cancer-guide-gyne002-endometrial.pdf. Accessed 2022 Mar 15.
Appendix 1: Cost Comparison Table
Note that this appendix has not been copy-edited.
The comparators presented in the following table have been deemed to be appropriate based on feedback from clinical expert(s) and drug plan. Comparators may be recommended (appropriate) practice or actual practice. Existing Product Listing Agreements are not reflected in the table and as such, the table may not represent the actual costs to public drug plans.
Table 8: CADTH Cost Comparison Table for pMMR Endometrial Cancer
Treatment | Strength / concentration | Form | Price ($) | Recommended dosage | Daily cost ($) | 28-day cost ($) |
|---|---|---|---|---|---|---|
Lenvatinib (Lenvima) | 4 mg/dose 8 mg/dose 10 mg/dose 12 mg/dose 14 mg/dose 18 mg/dose 20 mg/dose 24 mg/dose | Capsule | 33.9720a 68.6407a 75.2783a 101.916 a 116.9347a NA 175.4127a 233.8697a | 20 mg onceb daily | 150.56 | 4,216 |
Pembrolizumab (Keytruda) | 100 mg/4mL | Vial IV Infusion | 4,400.0000b | 200 mg Q3Wb | 419.05 | 11,733 |
Lenvatinib + Pembrolizumab | 569.60 | 15,949 | ||||
Single-drug chemotherapies | ||||||
Carboplatin | 50 mg 150 mg 450 mg 600 mg | Vial | 70.0000 210.0000 599.9985 775.00 | AUC 4 to 6 (600 to 900 mg) on day 1 Q3Wc | 36.90 to 56.90 | 1,033 to 1,593 |
Cisplatin | 1 mg/mL | 50 mL 100 mL Vial | 323.0000 270.0000 | 50-75 mg /m2 on day 1 Q3Wd | 12.86 to 19.29 | 360 to 540 |
Paclitaxel | 6 mg/mL | 5 16 25 50 Vial | 300.0000 1,196.8000 1,870.0000 3,740.0000 | 80 mg/m2 on days 1, 8, 15, and 22 Q4We | 214.29 | 6,000 |
Doxorubicin | 2 mg/mL | 5 mL 10 mL 25 mL 50 mL 100 mL Vial | 50.0000 588.6800 252.2500 1,304.2000 770.0000 | 50-60 mg /m2 on day 1 Q3Wf | 107.14 | 3,000 |
Platinum-based chemotherapies | ||||||
Carboplatin + Paclitaxelg | ||||||
Carboplatin | 50 mg 150 mg 450 mg 600 mg | Vial | 70.0000 210.0000 599.9985 840.0000 | AUC 4 to 6 (600 to 900 mg) on day 1 Q3W | 36.90 to 56.90 | 1,033 to 1,593 |
Paclitaxel | 6 mg/mL | 5 16 25 50 Vial | 300.0000 1,196.8000 1,870.0000 3,740.0000 | 175 mg /m2 on day 1 Q3W | 157.14 | 4,400 |
Carboplatin + Paclitaxel | 194.04 to 214.04 | 5,433 to 5,993 | ||||
Carboplatin + Docetaxelh | ||||||
Carboplatin | 50 mg 150 mg 450 mg 600 mg | Vial | 70.0000 210.0000 599.9985 840.0000 | AUC 5 to 6 (750 to 900 mg) Q3W | 46.90 to 56.90 | 1,313 to 1,593 |
Docetaxel | 20 mg 80 mg 160 mg | Vial | 249.0000 497.0000 925.000 990.0000 1,940.4000 1,850.0000 | 75 mg/m2 Q3W | 59.24 | 1,659 |
Carboplatin + Docetaxel | 106.14 to 116.14 | 2,972 to 3,252 | ||||
Cisplatin + Doxorubicini | ||||||
Cisplatin | 1 mg/mL (50 mL) [50 mg] 1 mg/mL (100 mL) [100 mg] | Vial | 323.0000 (6.4600 per mL) 270.0000 (2.7000 per mL) | 50 mg/m2 on day 1 Q3W | 12.86 | 360 |
Doxorubicin | 2 mg/mL (10 mL) [20 mg] 2 mg/mL (25 mL) [50 mg] 10 mg/mL (5mL) [50 mg] 100 mg 150 mg 200 mg | Vial | 665.4700 782.9100 252.2500 255.0000 285.0000 360.37000 70.0000 106.1200 1,304.2000 1,081.000 770.0000 973.0000 | 50 to 60 mg/m2 on day 1 Q3W | 6.67 | 187 |
Cisplatin + Doxorubicin | 19.53 | 547 | ||||
Hormone therapies | ||||||
Medroxyprogesterone | 2.5 mg 5 mg 10 mg 100 mg 150 mg/mL | Tablet Vial | 0.1183 0.2365 0.1670 1.2057 31.6900 | 200 to 400 mg once dailyj | 2.41 to 4.82 | 68 to 135 |
Megestrol | 40 mg 160 mg | Tablet | 1.3340 5.8151 | 160-200 mg once dailyk | 5.34 to 6.67 | 149 to 187 |
Tamoxifen | 10 mg 20 mg | Tablet | 0.1750 0.3500 | 20 mg twice dailyl | 0.35 | 10 |
Letrozole (Generic) | 2.5 mg | Tablet | 1.3780 | 2.5 mg dailym | 1.38 | 39 |
Exemestane (Generic) | 25 mg | Tablet | 1.3263 5.7533 | 25 mg dailyn | 1.33 | 37 |
Q3W = every 3 weeks; Q4W = every 4 weeks; Q6W = every 6 weeks; pMMR = mismatch repair proficient.
Note: Prices of chemotherapies were obtained from the DeltaPA IQVIA database13 (accessed February 28, 2022), and the prices of hormone therapies were obtained from the Ontario Drug Benefit Formulary14 (accessed February 28, 2022), unless otherwise indicated, and do not include dispensing fees. Cost calculations assume a body surface area of 1.80 m2 where applicable. Wastage of excess medication in vials is included in costs. Recommended dosage is based on Cancer Care Ontario monographs,15 unless otherwise indicated. For all target AUC calculations, dose calculations followed guidance from the Cancer Care Ontario product monograph for Carboplatin: Target AUC is 4 to 6. Carboplatin is dosed according to the following formula: Maximum carboplatin dose (mg) = target AUC (mg/mL per min) X (125 + 25); maximum dose is based on a capped GFR estimate at 125 mL/min for patients with normal renal function.16
aPrices is obtained from Ontario Exceptional Access Program drug list (accessed March 31, 2022).
bPembrolizumab price is based on the sponsor’s submission17; dosage is based on the draft product monograph. Pembrolizumab treatment is capped at 24 months (35 doses of 200 mg or 18 doses of 400 mg).18
cCarboplatin product monograph.19
dCisplatin product monograph.20
ePaclitaxel product monograph.21
fDoxorubicin product monograph.22
gCarboplatin + Paclitaxel product monograph.23
hCarboplatin + Docetaxel product monograph24
iCarboplatin + Doxorubicin product monograph.25
jMedroxyprogesterone product monograph.26,27
kMegestrol product monograph.28
lTamoxifen product monograph.29
mLetrozole product monograph.30
nExemestane product monograph.31
Appendix 2: Submission Quality
Note that this appendix has not been copy-edited.
Description | Yes/No | Comments |
|---|---|---|
Population is relevant, with no critical intervention missing, and no relevant outcome missing | Yes | No comment. |
Model has been adequately programmed and has sufficient face validity | Yes | No comment. |
Model structure is adequate for decision problem | No | See key limitations for the sponsor’s inappropriate use of partitioned survival modelling approach. |
Data incorporation into the model has been done adequately (e.g., parameters for probabilistic analysis) | Yes | No comment. |
Parameter and structural uncertainty were adequately assessed; analyses were adequate to inform the decision problem | Yes | No comment. |
The submission was well organized and complete; the information was easy to locate (clear and transparent reporting; technical documentation available in enough details) | No | Some information in the pharmacoeconomic model was unclear and difficult to locate. The approach used to estimate the impact of AEs on patient utility was insufficiently described. |
AE = adverse event.
Appendix 3: Additional Information on the Submitted Economic Evaluation
Note that this appendix has not been copy-edited.
Detailed Results of the Sponsor’s Base Case
Table 10: Disaggregated Results of the Sponsor’s Base-Case Analysis
Parameter | PEM + LEN | PCC | Incremental |
|---|---|---|---|
Discounted LYs | |||
Total LYs | 3.94 | 2.10 | 1.84 |
Progression-free survival | 1.12 | 0.57 | 0.56 |
Progressive disease | 2.82 | 1.53 | 1.28 |
Discounted QALYs | |||
Total QALYs | 2.78 | 1.53 | 1.26 |
Progression-free survival | 0.87 | 0.45 | 0.42 |
Progressive disease | 1.92 | 1.08 | 0.84 |
Discounted costs ($) | |||
Total costs | 198,474 | 40,428 | 158,046 |
Drug acquisition | 160,396 | 3,063 | 157,332 |
Administration | 3,162 | 1,525 | 1,637 |
Adverse events | 7,094 | 5,238 | 1,856 |
Disease management | 4,753 | 2,517 | 2,236 |
One-off progression | 190 | 222 | –31 |
Subsequent treatment | 1,194 | 4,660 | –3,466 |
End-of-life costs | 21,685 | 23,203 | –1,518 |
ICER ($/QALY) | 125,808 | ||
ICER = incremental cost-effectiveness ratio; LEN = lenvatinib; LY = life-year; PCC = physician’s choice chemotherapy; PEM = pembrolizumab; QALY = quality-adjusted life-year.
Appendix 4: Additional Details on the CADTH Reanalyses and Sensitivity Analyses of the Economic Evaluation
Note that this appendix has not been copy-edited.
Detailed Results of CADTH Base Case
Table 11: Disaggregated Results of the CADTH Base Case
Parameter | PEM + LEN | PCC | Incremental |
|---|---|---|---|
Discounted LYs | |||
Total LYs | 1.78 | 1.24 | 0.54 |
Progression-free survival | 0.86 | 0.49 | 0.37 |
Progressive disease | 0.92 | 0.74 | 0.17 |
Discounted QALYs | |||
Total QALYs | 1.35 | 0.94 | 0.41 |
Progression-free survival | 0.68 | 0.39 | 0.29 |
Progressive disease | 0.67 | 0.55 | 0.12 |
Discounted costs ($) | |||
Total costs | 200,838 | 50,616 | 150,222 |
Drug acquisition | 161,846 | 10,384 | 151,462 |
Administration | 3,120 | 1,534 | 1,585 |
Adverse events | 7,036 | 5,258 | 1,777 |
Disease management | 3,115 | 1,918 | 1,197 |
One-off progression | 185 | 227 | –42 |
Subsequent treatment | 1,990 | 7,511 | –5,521 |
End-of-life costs | 23,546 | 23,783 | –236 |
ICER ($/QALY) | 366,399 | ||
ICER = incremental cost-effectiveness ratio; LEN = lenvatinib; LY = life-year; PCC = physician’s choice chemotherapy; PEM = pembrolizumab; QALY = quality-adjusted life-year.
Scenario Analyses
Table 12: Summary of Scenario Analyses Conducted on the CADTH Base Case
Scenario | Drug | Total costs ($) | Total LYs | Total QALYs | ICER ($/QALY) |
|---|---|---|---|---|---|
CADTH base case | PCC | 50,616 | 1.24 | 0.94 | Ref. |
PEM + LEN | 200,838 | 1.78 | 1.35 | 366,399 | |
1. Weibull extrapolation used for OS | PCC | 50,590 | 1.33 | 1.01 | Ref. |
PEM + LEN | 203,092 | 2.41 | 1.79 | 197,619 | |
2. Log-logistic extrapolation used for OS | PCC | 50,350 | 2.08 | 1.51 | Ref. |
PEM + LEN | 198,619 | 3.93 | 2.77 | 117,818 | |
3. Alternate utility value regression model (time to death) | PCC | 50,505 | 1.24 | 0.94 | Ref. |
PEM + LEN | 198,915 | 1.78 | 1.36 | 347,666 | |
4. Alternate utility value regression model (progression and treatment status) | PCC | 50,647 | 1.24 | 0.89 | Ref. |
PEM + LEN | 203,325 | 1.78 | 1.32 | 356,313 | |
5. Full (100%) drug wastage for unused product | PCC | 54,781 | 1.24 | 0.94 | Ref. |
PEM + LEN | 203,901 | 1.78 | 1.35 | 363,217 |
ICER = incremental cost-effectiveness ratio; LY = life-year; OS = overall survival; QALY = quality-adjusted life-year.
Note: It should be noted that the sponsor provided an updated analysis for pMMR (cut-off date: August 18, 2020. Median follow-up duration, 35.8 months, 95% CI, 31.2 to 41.2). The sponsor indicated that the findings of the updated analysis were largely aligned with that reported in the analysis on January 10, 2019. The results of this additional data suggest that long-term OS is higher than was observed in the original submission. However, CADTH was not able to validate this additional information submitted late within the review period. Accordingly, the CADTH scenario analyses reflect the originally submitted data
Appendix 5: Submitted Budget Impact Analysis and CADTH Appraisal
Note that this appendix has not been copy-edited.
Table 13: Summary of Key Take-Aways
Key Take-aways of the Budget Impact Analysis |
|---|
|
Summary of Sponsor’s Budget Impact Analysis
The sponsor submitted a budget impact analysis (BIA)17 estimating the incremental budget impact of reimbursing PEM in combination with LEN for use by patients aged 18 years and older with advanced endometrial cancer that is not microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR), who have disease progression following prior platinum-based systemic therapy in any setting and are not candidates for curative surgery or radiation. The BIA was undertaken from the perspective of the Canadian public drug plans over a 3-year time horizon, and the sponsor’s pan-Canadian estimates reflect the aggregated results from provincial budgets (excluding Quebec). Key inputs to the BIA are documented in Table 14.
The sponsor estimated the number of eligible patients using an epidemiologic approach with data obtained from various sources17,32-37 including: Canadian Cancer Society statistics38 and a sponsor-conducted opinion survey of Canadian medical oncologists (N = 10).17 The sponsor adopted an average annual growth rate of 5.44% in estimating the total number of uterine cases over the time horizon.32,33,36 The sponsor estimated that 90% of these uterine cancers occur in the endometrium34 and 8% of endometrium carcinoma are advanced or metastatic cases.37 The sponsor assumed 84% of patients are referred to a medical oncologist or gynecologic oncologist, 87% receive first-line treatment and 63% move on to receive second-line treatment.17 The sponsor assumed 67% of patients who had received second-line treatment relapse and progress to advanced stages.17 The sponsor restricted the population to those who either do not undergo dMMR or MSI-H testing or are found dMMR or MSI-H negative after RET testing.
The BIA outcomes were estimated using the number of patients expected to be treated each week, which was modelled to increase over time. The treatment costs accrued under PEM + LEN combination therapy and comparators were based on KM time-on-treatment data in the KN775 trials. The market share of LEN+PEM increased from 0% to |||||||% over a |||||||||||||||||||| period based on a linear model. Drug costs were obtained from publicly available drug reviews39-41 and IQVIA DeltaPA database.42 No drug wastage was assumed.
Table 14: Summary of Key Model Parameters
Parameter | Sponsor’s estimate (reported as Year 1 / Year 2 / Year 3 if appropriate) |
|---|---|
Target population | |
Number of patients eligible for drug under review | 7,040 / 7,930 / 8,933 |
Proportion of endometrial cancer | 90% |
Proportion of advanced or metastatic disease | 8% |
Referral rate to medical oncologists or gynecologic oncologists | 84% |
First-line treatment rate by medical oncologists and gynecologic oncologists | 87% |
Second-line treatment rate | 63% |
Diagnosed earlier and progressed to advanced disease (includes recurrent disease) | 67% |
dMMR or MSI-H testing rate (from baseline to peak) | 70% to 90%17 |
Proportion of patients with dMMR or MSI-H EC | 15.7%17 |
pMMR/MSS/Untested EC patients | 629 / 664 / 700 |
Market Uptake (3 years) | |
Uptake (reference scenario) Pembrolizumab-Lenvatinib Paclitaxel Doxorubicin Docetaxel Ifosfamide Gemcitabine Clinical Trials | 0% / 0% / 0% 19% / 19% / 19% 56% / 56% / 56% 7% / 7% / 7% 0% / 0% / 0% 10% / 10% / 10% 8% / 8% / 8% |
Uptake (new drug scenario) Pembrolizumab-Lenvatinib Paclitaxel Doxorubicin Docetaxel Ifosfamide Gemcitabine Clinical Trials | ||||||% / ||||||% / ||||||% ||||||% / ||||||% / ||||||% ||||||% / ||||||% / ||||||% ||||||% / ||||||% / ||||||% ||||||% / ||||||% / ||||||% ||||||% / ||||||% / ||||||% ||||||% / ||||||% / ||||||% |
Cost of 1L treatment (per patient)a | |
Cost of treatment over cycle Pembrolizumab Q3W Pembrolizumab Q3W Lenvatinib (20 mg) Lenvatinib (14 mg) Lenvatinib (10 mg) Lenvatinib (8 mg) Lenvatinib (4 mg) Paclitaxel Doxorubicin Docetaxel Ifosfamide Gemcitabine Clinical Trials | $8,800.00 $17,600.00 $1,159.48 $772.94 $501.48 $456.00 $228.00 $24.86 $408.87 $128.60 $126.96 $4.51 $0.00 |
Cost of 2L treatment (per patient)a | |
Carboplatin Paclitaxel Doxorubicin | $66.28 $24.86 $408.87 |
dMMR = Mismatch repair deficient; MSI-H = Microsatellite instability-high; MSS = microsatellite stable; pMMR = mismatch repair proficient; Q3W = every 3 weeks
aTreatment duration is 35 maximum cycles for pembrolizumab (200 mg Q3W) and 8 maximum cycles for doxorubicin. Paclitaxel accrues costs over the time horizon.
Note: Cost estimation assumed average weight of 70.5 kg, body surface area of 1.77 m2 and no drug wastage.
Summary of the Sponsor’s BIA Results
The sponsor estimated the net 3-year budget impact of introducing LEN+PEM for treatment of patients aged 18 years and older with advanced endometrial cancer that is not MSI-H/dMMR, whose tumours have progressed following prior therapy to be $113,136,041 (Year 1: $10,313,968; Year 2: $42,840,169; Year 3: $59,981,904).
CADTH Appraisal of the Sponsor’s BIA
CADTH identified several key limitations to the sponsor’s analysis that have notable implications on the results of the BIA:
Unit costs of LEN, paclitaxel and doxorubicin were inaccurate: The sponsor submitted BIA17 obtained drug costs of paclitaxel and doxorubicin using published literature40 and IQVIA DeltaPA database,42 respectively. However, these estimates do not match the most recent cost estimates within the IQVIA DeltaPA database.13
In CADTH reanalysis, the price per 5-day blister card of LEN were updated to Extended Access Program costs and the prices per milligram of paclitaxel and doxorubicin was updated to the most recent costs in the IQVIA DeltaPA database.13
The sponsor’s choice of comparators did not reflect standard practice: In the submitted BIA,17 the sponsor assumed treatments that accrue market share include paclitaxel, doxorubicin, docetaxel, gemcitabine, and clinical trials. The clinical experts consulted for this review by CADTH suggested docetaxel is rarely used to treat patients with pMMR/MSS endometrial cancer. According to expert opinion, gemcitabine may have a market share up to 3% in the second-line setting and the introduction of LEN + PEM would decrease the market share of gemcitabine. The sponsor also assumed clinical trials capture 8% of market share and accrue no drug costs. The sponsor’s approach to include clinical trials in the market mix artificially decreases the estimated market size, disregarding the treatment costs incurred by those in clinical trials and underestimating the budget impact.
The sponsor’s analysis excluded platinum-based chemotherapy from the market mix. According to the clinical experts, pMMR/MSS patients would be rechallenged with platinum-based chemotherapies if they had a satisfactory response initially. In fact, a longer duration from treatment cessation to relapse would predict a better response to the previously administered treatment. If patients were not on treatment for shorter than 6 months, an estimated 20% to 30% of patients would be rechallenged with the platinum-based chemotherapy to which they initially responded. Should drug-free interval be greater than 12 months, the estimated proportion of patients being rechallenged would increase to 40% to 60%. As such, the sponsor’s assumptions on market share lacked external validity.
The sponsor’s submitted budget analysis is estimated for patient population who would not be eligible for re-treatment with platinum-based chemotherapy. According to the clinical experts, some patients may not be rechallenged; in cases of an allergic reaction or undesirable side effects such as myelosuppression or grade 3-4 neuropathy.
In CADTH reanalysis, the market share at baseline were revised based on feedback from clinical experts consulted for this review and excluded clinical trials. The reimbursement of LEN+PEM displaced the market share of paclitaxel, doxorubicin, and gemcitabine proportionally to sponsor’s assumptions on market share displacement.
CADTH could not address the limitation on the exclusion of platinum-based chemotherapy from the market mix.
The proportion of advanced or metastatic endometrial cancer is uncertain: In estimating the number of eligible patients, the sponsor estimated that 8% of all endometrial cancers are advanced or metastatic (which included stage IIIc, IVa, and IVb tumours).43 Clinical experts consulted by CADTH for this review suggested the proportion of advanced or metastatic endometrial cancer may be in the range of 25% to 30%. Advanced or metastatic endometrial cancer refer to cancers that have spread into the surrounding tissues and organs.43 CADTH noted uncertainty in the proportion of endometrial cancer classified as advanced or have metastasized in published literature. In another report by Alberta Health Services,44 approximately 30% of patients with endometrial cancer were reported to present with stage III and IV tumours. As such, there is uncertainty in the proportion of advanced or metastatic endometrial cancer, which may have been underestimated.
In a scenario analysis, the impact of uncertainty in the proportion of advanced or metastatic endometrial cancers was explored by adopting a value of 25% based on the feedback received from clinical experts.
There is uncertainty in market uptake of PEM + LEN combination chemotherapy: The sponsor assumed that PEM + LEN combination chemotherapy uptake is linear. The clinical experts consulted by CADTH considered the speed of LEN+PEM uptake to be rapid if it were publicly listed. The budget impact is sensitive to rate of market uptake, which is based on the sponsor’s assumptions.
CADTH explored the impact of market uptake of LEN+PEM in scenario analyses:
Assuming rapid uptake of LEN+PEM initially and plateauing off at 52 weeks (uptake curve: logarithmic)
Assuming rapid uptake of LEN+PEM after a delay and plateauing off at 52 weeks (uptake curve: exponential)
Inclusion of dMMR or MSI-H testing costs: The sponsor restricted the eligible population to those found dMMR or MSI-H negative and included the cost of dMMR or MSI-H testing. Given that the perspective of the BIA is restricted to costs borne by participating drug plans, the cost of dMMR or MSI-H testing should be excluded. The dMMR or MSI-H testing costs are, however, relevant to the health care payer perspective.
In CADTH reanalysis, the cost of dMMR or MSI-H testing was excluded. CADTH explored the impact of including the costs of dMMR or MSI-H testing under the health care payer perspective in a scenario analysis.
The budget impact model has limited transparency and flexibility: The sponsor’s submitted BIA model was unnecessarily complex, using circular referencing and hard coding. These modelling practices increase validity issues when making changes to the model and makes it difficult to ensure consistency throughout the budget impact model. Furthermore, the model lacked transparency and had limited flexibility to allow the reviewers to assess the impact of changing the sponsor’s base assumptions on estimated budgetary impact.
CADTH could not address this limitation. CADTH notes that the results presented should be treated with a degree of caution as the validity of the model calculations could not be thoroughly appraised.
CADTH Reanalyses of the BIA
CADTH revised the sponsor’s base case by updating LEN, paclitaxel, and doxorubicin costs, revising market share of comparators based on clinical experts, excluding clinical trial from the market mix, and excluding dMMR or MSI-H testing costs.
Table 15: CADTH Revisions to the Submitted BIA
Stepped analysis | Sponsor’s value or assumption | CADTH value or assumption |
|---|---|---|
Corrections to sponsor’s base case | ||
None | — | — |
Changes to derive the CADTH base case | ||
1. Update costs | Lenvatinib (20 mg): $828.2000 Lenvatinib (14 mg): $552.1000 Lenvatinib (10 mg): $358.2000 Lenvatinib (8 mg): $325.7150 Lenvatinib (4 mg): $162.8575 Paclitaxel: $0.1756 / mg Doxorubicin: $3.8500 / mg | Lenvatinib (20 mg): $877.0635 Lenvatinib (14 mg): $584.6735 Lenvatinib (10 mg): $376.3915 Lenvatinib (8 mg): $343.2035 Lenvatinib (4 mg): $169.8600 Paclitaxel: $10.0000 / mg Doxorubicin: $5.0000 / mg |
2. Market share assumptions (Base year / year 1 / year 2 / year 3) | Pembrolizumab – Lenvatinib: ||||||% / ||||||% / ||||||% / ||||||% Paclitaxel: ||||||% / ||||||% / ||||||% / ||||||% Doxorubicin: ||||||% / ||||||% / ||||||% / ||||||% Docetaxel: ||||||% / ||||||% / ||||||% / ||||||% Ifosfamide: ||||||% / ||||||% / ||||||% / ||||||% Gemcitabine: ||||||% / ||||||% / ||||||% / ||||||% Clinical Trials: ||||||% / ||||||% / ||||||% / ||||||% | Pembrolizumab – Lenvatinib: 0% / 32% / 63% / 63% Paclitaxel: 25% / 17% / 9% / 9% Doxorubicin: 72% / 49% / 27% / 27% Docetaxel: 0% / 0% / 0% / 0% Ifosfamide: 0% / 0% / 0% / 0% Gemcitabine: 3% / 2% / 1% / 1% Clinical Trials: 0% / 0% / 0% / 0% |
3. dMMR or MSI-H testing costs | Include | Exclude |
CADTH base case | Reanalysis 1 + 2 + 3 | |
dMMR = mismatch repair deficient; MSI-H = microsatellite instability-high
The results of the CADTH stepwise reanalysis are presented in summary format in Table 16 and a more detailed breakdown is presented in Table 17.
Based on the CADTH base case, the budget impact of the reimbursement of introducing LEN+PEM for treatment of patients with pMMR/MSS endometrial cancer in second- or subsequent-line of therapy is expected to be $9,469,160 in year 1, $40,112,025 in year 2, and $56,962,069 in year 3, with a 3-year total of $106,543,254. The budget impact is highly sensitive to uncertainty in the estimated proportion of patients whose disease is considered advanced or metastatic.
Table 16: Summary of the CADTH Reanalyses of the BIA
Stepped analysis | Three-year total |
|---|---|
Submitted base case | $113,136,041 |
CADTH reanalysis 1 | $107,846,779 |
CADTH reanalysis 2 | $112,947,201 |
CADTH reanalysis 3 | $112,994,294 |
CADTH base case | $106,543,254 |
BIA = budget impact analysis
CADTH also conducted additional scenario analyses to address remaining uncertainty, using the CADTH base case. Results are provided in Table 17.
Assuming 25% of patients with endometrial cancers are advanced or metastatic.
Assuming rapid uptake of LEN +PEM and plateauing off at 52 weeks (uptake curve is logarithmic based).
Assuming rapid uptake of PEM + LEN combination therapy after a delay and plateauing off at 52 weeks (uptake curve is exponential based).
Including dMMR or MSI-H testing costs.
Table 17: Detailed Breakdown of the CADTH Reanalyses of the BIA
Stepped analysis | Scenario | Year 0 (current situation) | Year 1 | Year 2 | Year 3 | Three-year total |
|---|---|---|---|---|---|---|
Submitted base case | Reference | $827,240 | $1,038,227 | $1,101,632 | $1,162,265 | $3,302,124 |
New drug | $827,240 | $11,352,195 | $43,941,801 | $61,144,169 | $116,438,165 | |
Budget impact | $0 | $10,313,968 | $42,840,169 | $59,981,904 | $113,136,041 | |
CADTH base case | Reference | $4,224,617 | $6,421,952 | $6,894,546 | $7,278,990 | $20,595,487 |
New drug | $4,224,617 | $15,891,111 | $47,006,571 | $64,241,058 | $127,138,741 | |
Budget impact | $0 | $9,469,160 | $40,112,025 | $56,962,069 | $106,543,254 | |
CADTH scenario analysis: 25% advanced or metastatic rate | Reference | $13,201,928 | $20,068,599 | $21,545,456 | $22,746,842 | $64,360,897 |
New drug | $13,201,928 | $49,659,723 | $146,895,536 | $200,753,307 | $397,308,566 | |
Budget impact | $0 | $29,591,124 | $125,350,079 | $178,006,465 | $332,947,668 | |
CADTH scenario analysis: Logarithmic market uptake of pembrolizumab + lenvatinib | Reference | $4,224,617 | $6,421,952 | $6,894,546 | $7,278,990 | $20,595,487 |
New drug | $4,224,617 | $15,753,717 | $46,901,821 | $64,215,626 | $126,871,164 | |
Budget impact | $0 | $9,331,765 | $40,007,275 | $56,936,636 | $106,275,677 | |
CADTH scenario analysis: Exponential market uptake of pembrolizumab + lenvatinib | Reference | $4,224,617 | $6,421,952 | $6,894,546 | $7,278,990 | $20,595,487 |
New drug | $4,224,617 | $16,016,479 | $47,142,881 | $64,277,407 | $127,436,767 | |
Budget impact | $0 | $9,594,527 | $40,248,335 | $56,998,417 | $106,841,280 | |
CADTH scenario analysis: Including dMMR or MSI-H testing costs | Reference | $4,400,160 | $6,607,050 | $7,089,719 | $7,484,785 | $21,181,554 |
New drug | $4,400,160 | $16,103,394 | $47,257,508 | $64,505,653 | $127,866,555 | |
Budget impact | $0 | $9,496,344 | $40,167,789 | $57,020,868 | $106,685,001 |
BIA = budget impact analysis; dMMR = mismatch repair deficient; MSI-H = microsatellite instability-high.
Stakeholder Input
Patient Input
Colorectal Cancer Resource & Action Network, Canadian Cancer Society, and Canadian Cancer Survivor Network
About Colorectal Cancer Resource & Action Network, Canadian Cancer Society, and Canadian Cancer Survivor Network
The Colorectal Cancer Resource & Action Network (herein ‘CCRAN’) led a collective patient input submission on Pembrolizumab monotherapy and Pembrolizumab in combination with Lenvatinib for the treatment of advanced endometrial cancer. The following patient advocacy groups thoughtfully collaborated with CCRAN to provide meaningful and compelling patient input:
Canadian Cancer Society (CCS)
Canadian Cancer Survivor Network (CCSN)
All patient groups are registered with CADTH.
Please note: CCRAN is a national not for profit patient advocacy group championing the health and wellbeing of Canadians touched by colorectal cancer and those at risk of developing the disease. CCRAN has recently expanded its patient-focused mandate to serve a population of patients with cancer outside of the colorectal cancer space by providing HTA patient and clinician input submissions within the oncology space for:
patient groups who do not have the capacity to perform these submissions and/or
a drug therapy wherein there currently is no representative patient group (such as the drug therapy currently under review).
Information Gathering
To ensure the advanced endometrial cancer patient perspective was captured for this critically important therapeutic under review, CCRAN employed a multi-faceted outreach approach. On November 11, 2021, we reached out to 12 Canadian clinicians via email who treat advanced endometrial cancer patients requesting assistance identifying patients who had/have experience with Pembrolizumab + Lenvatinib who would be willing to participate in a telephone interview to share that experience for an HTA patient input submission being made to two expert committees in Canada. That same email was followed up on November 28th, 2021. On December 6th , 2021, the President (Dr. Helen MacKay) of The Society of Gynecologic Oncology of Canada (‘GOC’) was respectfully contacted with the same request. The GOC kindly sent a mass email authored by the GOC President on CCRAN’s behalf to the members of the GOC on December 9th, 2021 requesting assistance with patient recruitment.
Additionally, a connection was made between CCRAN and the Mission Coordinator from the Canadian Cancer Society (CCS) on November 29, 2021, ensuring CCS support the inclusion of the endometrial cancer patient’s values and preferences in this submission through a recent online survey they conducted regarding the endometrial cancer patient’s:
Experience with respect to the diagnosis of their endometrial cancer
Experience with respect to their endometrial cancer journey
Experience with respect to the drug therapies administered prior to the therapy under review
The online survey was administered from October 22nd – November 3rd, 2021 wherein 22 survey respondents provided input and the results are herein attached and labelled as APPENDIX 2.
Figure 1: Descriptions of Survey Respondents
CCS survey respondents consisted of 20 endometrial cancer patients and 2 caregivers representing the perspective, voice and values of the endometrial cancer patient.
Figure 2: Location of Survey Respondents
Survey respondents resided in Quebec, British Columbia, Ontario, Alberta, Saskatchewan and Manitoba.
Figure 3: Age of Survey Respondents
The majority of survey respondents were between 50 – 59 and 70 – 79 years
The CCS survey findings will be referenced throughout this submission for they reflect the perspectives of the endometrial cancer patient population. CCS also sent out mass emails on behalf of CCRAN requesting advanced endometrial cancer patients who have experience with the therapy under review contact CCRAN for a telephone interview to kindly share that experience. Emails were also shared by the CCS with Cancer Connection Forum and with McGill University Health Center’s Gynecological Unit. Additional outreach efforts were made on behalf of CCRAN to the following American and European based organizations with similar requests:
Force: www.facingourrisk.org Cancer Care: www.cancercare.org ESGO https://engage.esgo.org
One additional outreach effort was made to the Canadian Cancer Survivor Network (CCSN) on December 5, 2021 who employed an online survey as well, resulting in one endometrial cancer patient having experience with the therapy under review. CCSN was kind enough to provide the summary of the patient’s insights which is herein attached and labelled APPENDIX 3.
It was the clinician and GOC outreach efforts that resulted in 1 caregiver and 3 advanced endometrial cancer patient telephone interviews. Telephone interviews were conducted by CCRAN between December 1st and December 14th 2021 inclusive, with each patient providing first hand compelling, relevant and high quality input regarding their:
experience with respect to the diagnosis of their cancer,
disease experience,
experience with respect to previous therapies administered prior to Pembrolizumab + Lenvatinib and
experience with respect to Pembrolizumab + Lenvatinib.
The mean age of the patients was 62 years and the median age was 64 years at the time of their diagnosis. The qualitative data from the interviews is summarized and represented entirely in APPENDIX 1 which is attached and will serve, for the most part, as the basis for this qualitative submission, in addition to the survey findings furnished by CCS, and the one survey result supplied by CCSN.
Disease Experience
Endometrial cancer is the most common gynecological malignancy in Canada and our interviewed patients were surprised to learn from their treating oncologists that its incidence is increasing. Sadly, no specific screening is recommended in the general population which may account, in part, for the advanced disease case counts, which are associated with a poor prognosis, primarily because patients with advanced endometrial carcinoma have limited treatment options.
Most women with endometrial cancer will present with abnormal bleeding, irregular cycles and/or excessive bleeding in the premenopausal female, or any bleeding in the post menopausal setting. In the advanced disease setting, women may complain of pelvic and abdominal discomfort, bloating, or presence of a mass, gastrointestinal or genitourinary symptoms, or constitutional symptoms. According to the qualitative data captured in Appendix 1, all four patients had been experiencing incessant vaginal bleeding for a period of at least 6 months to >2 years. Patient B reported having experienced a mucus-type vaginal discharge accompanied by significant abdominal discomfort in addition to her vaginal bleeding. Three of the four patients (Patients A, B, and D) expressed significant frustration and profound disappointment over their ongoing symptoms whose cause could not be identified for quite some time, despite repeated diagnostics, which they believe contributed to their disease progression and diagnosis of advanced stage disease.
“I had been symptomatic for years….whose cause couldn’t be understood or identified. I just kept bleeding and bleeding. The ultrasounds couldn’t pick anything up and then I had a CT scan that sorta picked up something so I went on to have a D&C which picked up my cancer…I was then scheduled for my surgery. And I have to tell you that 10 minutes before I went into my surgery, I found out that I had metastatic disease to my lungs. It was so shocking and disappointing. How awful for me.” Patient D
The Canadian Cancer Society (CCS) Survey findings (n=22) clearly demonstrated the impact of the disease on the patient’s day to day activities and ultimately their quality of life. Q3 of the survey asked “How much of an impact do symptoms associated with endometrial cancer have on your day-to-day activities and quality of life?” Patients diagnosed with endometrial cancer were asked to select all that apply from a list of tasks. Those tasks scoring highest in the moderate to significant impact range included the ability to:
conduct household chores
exercise
travel and
work
The CCSN survey respondent also expressed concerns regarding her day-to-day living. She identified “living with uncertainty, feeling isolated or lonely” and that she experienced “diarrhea, occasional interstitial cystitis, and herpes outbreaks” which compromised her quality of life.
Interviewed Patients B and D (APPENDIX 1) both shared that they had cancer induced symptoms with which they had struggled. Patient B had endured pelvic pain and constipation; while Patient D was fraught with fatigue, nausea, and a lack of appetite (Q17).
The four interviewed patients articulately and vividly described the toll a diagnosis of advanced endometrial cancer plays on the caregiver. Th diagnosis can be equally frightening for the caregiver, who is typically the spouse, but can be anyone – a family member, friend or loved one. The caregiver may take on many roles in the course of the patient’s journey. They will assume the role of medical translator, information specialist, housekeeper/meal preparation, financial advisor, psychosocial expert, and so much more. Quite often the caregiver’s function is overshadowed or under-valued; in large part because the focus is typically on the patient, thus discounting the essential role played and meaningful contribution delivered by the caregiver. Our interviewed patients painfully described the stress, and debilitating anxiety a caregiver undergoes once a diagnosis of advanced endometrial cancer is delivered to the family. They explain (Q22):
“Yes, it was life-altering. We have been through a major trauma, even to this day, it still affects us. The impact is lifelong. Both my sister and I were quite young. She and I would take turns taking my mom to her appointments. We would do research ….We struggled with the side effects of the treatments, learning how to give her injections every day for her thrombosis. …dreadful disease.” Caregiver A
“My husband has always been there but emotionally, it has been very draining and stressful for him. He had to become Dr. Google overnight. He came to all my appointments and to the 6 cycles of chemo and had to deal with all the chemo side effects which were brutal. Then covid kicked in and he couldn’t come with me so that was so very stressful for him. So he sat in his car and waited for me the whole time. He was there the whole time for me, including during surgery. And he has his own health issues…” Patient B
“My husband was there taking care of me, doing housework, cooking, I guess just taking on the bulk of responsibilities because I couldn’t do it. But then in 2019, he had a stroke and now he is different and compromised, it is difficult for him. He had to do rehabilitation because of his stroke. He really cannot help me. I had to make arrangements for him and for me. For example, who will go first, him or me? It is a terrible situation. His stroke may not have been caused by my diagnosis but my diagnosis may have impacted his health to the point where it could have contributed to his poor health….Cancer does atrocious things to not only the patient but to their family members.” Patient D
Experiences With Currently Available Treatments
Patients with advanced endometrial carcinoma have limited treatment options. If diagnosed with an early stage of the disease, patients will undergo surgery to remove the uterus (and perhaps the cervix), fallopian tubes and regional lymph nodes. Radiation therapy may also be indicated to kill cancer cells in addition to hormonal therapy to block cancer growth. Carboplatin in combination with paclitaxel are standard chemotherapy treatments indicated for endometrial cancer in both the adjuvant setting and first line treatment of metastatic endometrial cancer. These therapies, particularly the latter, are associated with treatment induced toxicities that compromise patients’ quality of life and fail to extend patients’ longevity in a meaningful way.
The CCS survey findings (Q8) captured the impact of treatment-induced toxicities as reported below in the table. Previously administered treatments included surgery, chemotherapy (Caelyx, Carbotaxol) and hormonal therapy (Anastrozole). Surveyed patients selected “changes in libido or sexual function” as the most significant side effect resulting from their previous treatment(s) with 10 responses appearing in the moderate to significant impact range. Fatigue (9), constipation (6),and peripheral neuropathy (5) followed, as highlighted in the table.
CCS captured patient input through first hand testimonials as well which described the harsh and debilitating side effects these patients endured as they underwent standard of care treatments indicated for the management of their advanced endometrial cancer. These testimonials also highlight the overall lack of efficacy associated with these standard of care therapies that compromised their everyday quality of life. Appearing below is one of the testimonials CCS secured outside of the Survey:
“The first line of chemotherapy gave me very bad side effects, pain and complications. They gave me first and second line chemotherapy sessions, both had very bad side effects and were not effective.….At that point, they gave me radiation to shrink the tumour, and yet that was only for one small tumour that I had….My abdominal pain became worse and worse. The pain medications were not effective.” CCS Patient Testimonial
Interviewed patients provided thoughtful input regarding their treatment journeys. Caregiver A summarized her mother’s experience as quite ghastly. Her mother underwent surgical resection and then received radiation therapy which she tolerated quite well and it controlled her cancer for approximately 3.5 years but spinal and bone metastases eventually ensued. She was prescribed Nexavar to which there was no response. Her mother was then prescribed second line Carbotaxol but could only endure 2 cycles due to “horrible side effects” that were debilitating and unmanageable. Her mother then succumbed to the disease.
Patient B underwent surgical resection for her cancer but shortly after her surgery (3 months) she was diagnosed with a mass that was strangulating her left ureter as well as metastases to her spleen, liver and peritoneum. She was prescribed 6 cycles of Carbotaxol to which there was little response. Her left kidney failed due to hydronephrosis. She was then recommended the therapy under review (Pembrolizumab + Lenvatinib).
After undergoing surgical resection for her cancer, Patient C endured 6 toxic cycles of Carbotaxol and 3 treatments of brachytherapy for any remnant microscopic disease. Sadly, she was diagnosed with a recurrence in her peritoneum and spine for which she was prescribed more Carbotaxol (6 cycles). She initially experienced a partial response to the Carbotaxol and eventually progression. She was then prescribed hormonal therapy, Letrazole in combination with Vitamin D but that too showed progressive disease. She then started Doxorubicin (6 cycles) but had to stop due to diminished quality of life and eventually progressive disease developed and was recommended Pembrolizumab + Lenvatinib).
Patient D was diagnosed with metastatic disease (lungs) as she was being wheeled into surgery. She was prescribed Carbotaxol. She could only endure one cycle of the carboplatin due to extreme neuropathy. She then continued on with the paclitaxel only, which regressed the lung metastases but was ineffective against the abdominal disease which was discovered post op. She was recommended Pembrolizumab monotherapy.
Carbotaxol was accessed by all four of our interviewed patients (Patients A, B, C and D) as a treatment option for women with advanced endometrial cancer. All four patients reported debilitating side effects while being on Carbotaxol despite having spent a relatively short period of time on the drug therapy (<2 months, 4 months, 6 months, 5 months respectively), and sadly none of the patients reported having benefited from the treatment. Patient reported side effects from Carbotaxol therapy included: neuropathy, fatigue, nausea, lack of energy, headaches, hypothermia, cramping in extremities, abdominal pain, hair loss and diarrhea. Patients were disheartened to learn of the lack of response and their quality of life was significantly impacted to the point where they were unable to function because they were physically unwell and debilitated. They were quite emphatic about their experience with Carbotaxol, despite the fact that it was short lived. In their words (Q8A-C);
“…her life started to deteriorate. So, when she started Nexavar, her quality of life got worse. She could no longer socialize or travel. And Carbotaxol was the worst of them all. Horrible side effects. Her outlook even became negative. She even stopped working. And it was so difficult to watch from a caregiver’s perspective.” Caregiver A
“I had typical side effects while I was on the Carbotaxol. I had hair loss….I had absolutely no energy. The coldness and chills were horrible. I couldn’t do any housework or shopping. I had to have everything delivered because that was very challenging for me due to fatigue and exhaustion. My energy level was so low and I was so weak. I tried to stock my house with products and food. My husband did the cooking and cleaning. Thank goodness. How do single women get through this? I can’t imagine. “ Patient B
“When I was on the Carbotaxol, my quality of life was really not so good, actually, it was pretty terrible. It was because of the pain in my hands. They were like electroshock waves were going through them. They were also numb at the same time. It was hard to explain. I hated it so much. I still have the pain to this day. The doxorubicin, was not painful but my heart is not the same anymore. I am at a lower level today and as a result I have to be careful with activity like running, walking etc…. My heart is 70% at its pumping capacity now because of doxorubicin. Isn’t that awful?” Patient C
“I was totally dependent on my husband while on Carbotaxol. He would have to give me showers, dry my hair, I couldn’t dry myself after a shower, he had to help me dress myself, I couldn’t cook, I couldn’t clean the house, I was a total clean freak and I had to abandon that. I couldn’t do anything around the house. I couldn’t leave the house for weeks and weeks because I had no energy due to the chemo. I couldn’t do grocery shopping or any type of shopping. I wasn’t engaged in life at all. There was no socializing for me at all. I couldn’t even go to church or see my kids and grandkids who I cherished so much. The side effects of chemo were so brutal. I had nausea, fatigue, no energy at all, I had headaches which bound me to bed, I was useless. I slept a lot. All these were so severe 80% of the time. They had to take me off the carboplatin because of the neuropathy. What sort of life is this to lead?” Patient D
Improved Outcomes
Q10 of the CCS survey asked “What improvements would you like to see in new treatments that are not achieved in currently available treatments?” Open ended survey replies included: fewer side effects such as skin issues, fatigue, bladder control, stamina, vaginal bleeding after intercourse, vaginal dryness, hair loss, pain, concentration problems (chemotherapy fog) and arthritis. Respondents also indicated they would like to see more drug affordability as the cancer journey can become quite expensive while undergoing treatment, as usual everyday activities may become interrupted, requiring additional expenditures to help cover or assume those activities.
All interviewed patients provided their perspective on the improvements (Q25) they would wish to see associated with a new drug therapy – improvements they believe are currently not available with standard of care therapies for the management of advanced endometrial cancer. They passionately expressed the following: a desire to access a therapy that would promote good quality of life while effectively regressing their disease.
“I would like to see an extension in life and of course a reduction in side effects that will permit a patient from living their life with some degree of normalcy or a good degree of quality of life.” Caregiver A
“Treatments should have better side effects for everyone.” Patient C
These interviewed patients focused heavily on being able to access a drug therapy that could be free of debilitating side effects, allowing them the ability to live their lives with some degree of normalcy. One patient (Patient B) also emphasized the need to access therapies that can be easily administered, in the comfort of their own home: an oral therapy that would minimize visits to the cancer centre. In patients’ own words:
“I would say that all therapies should come in a pill form, have no side effects or minimal side effects, and it should shrink the disease big time!” Patient B
“I would probably like to see a better quality of life – so I guess no side effects, because of what I went through. A better drug on the market because of the damage I underwent. It was horrible what I went through. Cancer is not so easy so it would be helpful to go through it with a drug that provides a good quality of life.” Patient D
Furthermore, the three interviewed patients (Patients B, C and D) maintain the therapy under review currently possesses these desired improvements and were extremely grateful to have been able to access this therapy (Q26). According to their explicit and detailed input, it has prolonged their life significantly with minimal to no side effects, promoting excellent quality of life. Their lives have been truly ameliorated, such that they have been able to resume normal daily activities, spend quality time with friends and family (pandemic notwithstanding), permitting them the freedom to “living life again”. In their own words, patients provided the following input regarding the current therapy (Pembrolizumab monotherapy and Pembrolizumab + Lenvatinib) possessing the desired improvements:
“Yes, I do! Look at my results! My disease was so stubborn but this therapy has reduced my disease considerably and the side effects have been so great and so very minimal for me. My quality of life has been excellent.” Patient B
“Yes! it’s fantastic, I do not feel any side effects at all. I would say it has been constructive being on this immunotherapy vs having been on previous therapies like chemo which I would consider as destructive.” Patient C
“Yes, I certainly do. It has given me all of that and so much more because I am myself again. I have my life back. I believe that says it all.” Patient D
Experience With Drug Under Review
APPENDIX 1 (Qs#12-27) captured the treatment-related experiences for three interviewed Canadian patients who are currently undergoing the therapy under review. One additional patient provided input regarding her experience with the therapy through the CCSN online survey. In total, four patients provided compelling input (Patient B and C provided input regarding Pembrolizumab + Lenvatinib; Patient D provided input regarding Pembrolizumab monotherapy; and the CCSN patient provided input regarding Pembrolizumab + Lenvatinib), all of whom underscored the dire need to improve therapeutics for the management of advanced endometrial cancer because patients diagnosed with recurrent or metastatic endometrial cancer have limited treatment options. And the options that are available to them are not only short lived but riddled with debilitating side effects. The following four patients were diagnosed with metastatic, chemotherapy resistant endometrial cancers treated with pembrolizumab + Lenvatinib who, having achieved long term durable responses, wished to share their experiences for the purposes of informing your deliberative processes:
Patient B was a 51 year old female who underwent surgical resection of her endometrial cancer. Three months post op, she was diagnosed with a mass that was strangulating her left ureter as well as metastases to her spleen, liver and peritoneum. She was prescribed 6 cycles of Carbotaxol to which there was little response. Her left kidney failed due to hydronephrosis. She was then recommended Pembrolizumab in combination with Lenvatinib in October 2019 as part of a clinical study in second line therapy.
Patient C was a 64 year old female who underwent surgical resection, endured 6 toxic cycles of Carbotaxol and 3 treatments of brachytherapy. She was diagnosed with recurrent disease in her peritoneum and spine for which she was prescribed first line Carbotaxol (6 cycles). Progression ensued. Hormonal therapy (Letrazole) + Vitamin D was then prescribed as part of second line therapy to which there was no response. Third line therapy followed consisting of Doxorubicin (6 cycles) to which she responded poorly and experienced diminished quality of life. In August 2021, the patient accessed Pembrolizumab + Lenvatinib through a private pay plan as part of fourth line therapy.
Patient D is a 69 year old female who learned of her metastatic disease to her lungs as she was being wheeled into surgery. She was prescribed first line Carbotaxol post op. She could only endure one cycle of the carboplatin due to significant neuropathy but did continue with the paclitaxel portion, which regressed the lung metastases but was ineffective against the abdominal disease, which appeared while on the paclitaxel. She was then recommended Pembrolizumab monotherapy as part of second line and began treatment in June 2021, accessed through her husband’s private insurance plan.
The CCSN patient accessed standard of care therapies such as Carbotaxol as well as complementary therapies and alternative medicines that were somewhat effective but, due to progressive disease, did go on to access Pembrolizumab + Lenvatinib through a clinical trial. The Pembrolizumab + Lenvatinib provided tumour shrinkage and slowed progression of disease according to the patient input. It did, however, cause undesirable treatment induced side effects such as diarrhea and herpes outbreaks.
The three interviewed patients accessed the therapy under review with great anticipation and hope because they had either exhausted standard of care therapies for the management of their cancer (Patient C) or had been recommended to enroll in a clinical trial that could prove helpful in the treatment of their cancer (Patient B) or could molecularly target their particular cancer: MSI-High Cancer (Patient D). Two of the three interviewed patients (Patients C,D) experienced no side effects whatsoever from their Pembrolizumab + Lenvatinib and Pembrolizumab monotherapy treatments respectively (Q15A).
“No, Not at all. It has been amazing. I am a little tired because of the previous therapies I was on, but no, no side effects at all, whatsoever.” Patient C
“Nope, no side effects whatsoever.” Patient D
Patient B experienced two treatment-induced side effects: diarrhea and mild fatigue, both of which are well managed and easily controlled. She was prescribed loperamide to control the diarrhea and Synthroid for the fatigue, both of which are no longer issues.
“The main side effect I have experienced is irritable bowel syndrome (diarrhea) for which I was prescribed loperamide. And I had some fatigue at the end of the treatment, in August so they put me on Synthroid. The diarrhea was mild because if I watch what I eat, it is ok. I get to manage it very well. And the fatigue is mild as well. That’s pretty much it. “ Patient B
And the CCSN patient reported the following treatment-induced side effects: fatigue, diarrhea, and urinary tract infection. She does report there has been tumour shrinkage and that the therapy has “slowed progression of disease”. Interviewed patients were asked to rate their quality of life while on the therapy under review (Q16). All three patients provided high scores: 9, 8, and 10, generating an average score of 9, and two patients provided comments with their scores:
“My life has definitely been better since being on this therapy both from a quality of life perspective and from a survival perspective.” Patient B
“I feel so good while on this therapy. I am as I am right now, all the time.” Patient D
All three interviewed patients maintain that Pembrolizumab + Lenvatinib or Pembrolizumab monotherapy has delivered a remarkable response wherein their disease has regressed significantly in addition to having provided them with an excellent quality of life (Q19). The objective findings from diagnostics such as imaging (CT – except for Patient C) and labs support their improved overall well being. Clinically, they feel better! In the patients’ own words:
“First, I felt better. I had no more cancer symptoms. I could go to the bathroom. I had no more constipation, and I had no more pain in my pelvis. And the tingling resolved. And the CT scan showed that I was responding to the therapy. They were taking measurements every time I had a CT scan. My cancer shrank by 70%! and now there is so much scar tissue. It is pretty much dead in there. I am so lucky to have been on this therapy. It has been so wonderful for me.” Patient B
“I had 2 CT scans so far that confirmed response. The first CT scan showed lots of improvement in all tumours including my belly. The second one showed more improvement in all tumours too. I also feel so well. That’s a sign that the treatment is working. I have no more cancer symptoms. I could go on taking this therapy with no side effects forever.” Patient D
All three interviewed patients cited how much easier the therapy under review was to use when compared to previously administered therapies, resulting in a superior quality of life (Q21). For patients who accessed Pembrolizumab + Lenvatinib: patients appreciated the opportunity to access an oral therapy (Lenvatinib) which is easily administered in the comfort of their own homes, thereby avoiding a long and grueling stay and infusion time at the cancer centre. Patients also appreciated the short infusion time associated with Pembrolizumab (30-45 minutes), administered every 3 weeks, which is unlike the infusion times associated with previously administered standard of care therapies for advanced endometrial cancer (Carbotaxol and Doxorubicin).
“Yes, it has. When I was on the Carbotaxol, I used to have to sit in an infusion chair for 8 hours a day at the hospital and I have no more veins because of it. I now require a portacath because of that now. What a horrible therapy. But now, because of this immunotherapy, it is done in a 45 minute infusion and I have no side effects. And the other half of the therapy I get to take at home. And the other beauty of it is, there is no Decadron anymore, woohoo! So no more discomfort. I am so happy about that. I love this treatment. It is wonderful and I have no pain. It is fantastic to take. And I get an oral therapy at home. What more can you ask for as a cancer patient?”
Two of the three interviewed patients (B and D) struggled with cancer induced symptoms prior to starting the therapy under review and in each case the therapy provided significant resolution of those symptoms. In Patient B’s case, her pelvic and abdominal disease regressed after starting Pembrolizumab + Lenvatinib which in turn resolved her pelvic pain and constipation. Patient D had been experiencing fatigue, nausea, lethargy and lack of appetite, due entirely to the disease in her abdomen but these symptoms completely resolved after commencing Pembrolizumab monotherapy. The therapy has regressed the disease in both these patients to the point where symptoms have completely ameliorated (Q17). Patients expressed how grateful they were to be rid of those cancer induced symptoms:
“….I don’t have any of those symptoms anymore. The therapy shrank my cancer and took all those symptoms away.” Patient B
“Pembrolizumab helped to resolve all of them. It was a miracle. I don’t have any of them anymore. Apparently, a lot of those symptoms were because of the disease in my belly. But the disease in my belly has shrunk so much, it is not giving me problems anymore or symptoms. They are completely gone.” Patient D
Interviewed patients have been undergoing the therapy under review for, what is in some cases, an extended period of time, reflective of sustained and durable responses: 26 months, 4 months, 5 months. Efficacy was radiographically and clinically confirmed in each patient’s case (save Patient C who was scheduled to undergo imaging within a few weeks of their telephone interview but the patient was quite emphatic that based on how they were feeling clinically and their labs, response was imminent and would correlate with CT scan findings (Q19).
Patients B, C and D were quite expressive and became emotional when describing what impact the therapy has had on their lives and what they have managed to accomplish or fulfill. They expressed profound disappointment with having accessed previous therapies that failed to successfully treat their cancer and caused indescribable pain, suffering and anguish, enduring unimaginable and debilitating side effects. But the therapy under review has been and continues to be their lifeline, a “miracle” drug (Patient D), a “God-sent” (Patient C), without which they would not be alive today. They credit their longevity and ability to function at an almost normal level entirely to Pembrolizumab + Lenvatinib or Pembrolizumab monotherapy. Patients were able to resume what most would consider to be a normal lifestyle while undergoing therapy, but wished to note that this was quite extraordinary for a cancer patient based on their experience with previously administered therapies. Interviewed patients reported significant improvements in health status while undergoing the therapy under review (both Pembrolizumab + Lenvatinib and Pembrolizumab monotherapy) with respect to physical function and overall quality of life. They were able to resume responsibilities such as grocery shopping, housework, cooking, baking, caring for their loved ones – responsibilities most healthy Canadians take for granted (Q24). But Patient C no longer takes these simple chores for granted in the setting of a cancer diagnosis. She is grateful to be able to perform them while actively undergoing cancer therapy. Patients were also able to fulfill and accomplish a great deal while on the therapy. They mention being able to travel, resume their studies, spend quality time with family and cherished friends, complete their sailing certification, rejoin the YMCA to rebuild muscle mass, engage in social media, and so much more. Patients can not only function, they can thrive.
This life altering therapy has been repeatedly referred to as a “gift” and a “miracle” or that which has provided patients with great “hope” because it has offered them significant life extending properties while improving the patient’s quality of life. Additionally, it is worth noting that adverse events resulting in permanent discontinuation of the drug occurred in none of the patients; dosage interruptions due to an adverse reaction occurred in none of the patients; one patient ( C ) experienced a dose reduction (14mg to 10mg/day) in the Lenvatinib in the first few days of starting therapy due to headaches but has never experienced any issues since (Q20). Patients were overcome with gratitude and emotion throughout the interviews when speaking of their experience with the therapy under review:
“Do you really want an answer? (volume increased 5x). 1000% of course, I am so grateful for this therapy. Of course, of course, of course. I couldn’t afford to pay for this on my own, so I am so terribly appreciative of this. This therapy is my hope for a great extension in life. It is my way of continuing to live. Is it going to change for me? Will I be able to accept it for free in the future? I am so scared I might run out of luck in that respect and be forced to pay for it which is why I am participating today. I pray that I will be able to accept this therapy for free and that others who qualify will be able to accept it for free as well so they can benefit like me.” Patient C
“If I had not accessed this therapy, my life would have spiraled out of control and inevitably, to death. I have been able to travel, take an interest in my studies and my loving family again. Nothing is more important than family. I have nephews who have moved into the city who are spending Christmas eve with us and I am looking forward to spending time with them which is so special to me and important to me. I have 2 nieces and 2 nephews in Ontario who I am seeing that I get to see because this therapy is making it possible to do so. These are special times because I am well enough to do it – all because of the therapy. These are not remarkable events but they are nevertheless remarkable to me.” Patient B
Companion Diagnostic Test
Both mismatch repair deficiency (dMMR) and high microsatellite instability (MSI-H) have been proposed as predictive biomarkers of response to the immune checkpoint inhibitor Pembrolizumab. Approximately 16% of patients diagnosed with recurrent endometrial cancer have tumours with high levels of MSI-H and dMMR (jco.19.02627 (1).pdf ) and these patients could benefit from Pembrolizumab monotherapy. Patients need to provide a tissue sample for biomarker analysis (that can be done through IHC or PCR) to help guide treatment. Our interviewed patients were not aware of any specific testing requirements to help determine their biomarker status that would identify them from potentially benefiting from Pembrolizumab monotherapy (Q11A). They were, however, aware of a myriad of testing they had to undergo, to which they happily subjected themselves because they had either exhausted therapeutic options or progressed on standard of care therapies.
“I don’t really know and I didn’t really ask. All I know is that my tissue went to a tissue bank for testing and my oncologist spoke to me about it.” Patient B
None of our interviewed patients incurred any out of pocket expenses for any diagnostic testing they required to qualify for their therapy (Q11D). Based on previous submissions made by CCRAN, patients’ MSI/MMR status could be determined through IHC testing at their local treatment centre and patients are typically grateful to access the test.
For those who qualify for the immunotherapy based on the identification of a unique biomarker, patients may be able to experience a life prolonging therapy with minimal to no side effects, allowing them to resume their day to day activities. For patients whose endometrial tumours are identified to be microsatellite stable (MSS) and mismatch repair proficient (pMMR), they too are able to avail themselves of a life-prolonging therapy that includes pembrolizumab in combination with lenvatinib, based on patient input in APPENDIX 1. The combination therapy, according to our interviewed patients (B and C, and CCSN patient), has demonstrated great efficacy in the treatment of their recurrent/metastatic disease. Upfront testing will identify the patients who qualify for monotherapy vs combination therapy and will ultimately change the treatment paradigm and guide treatment decisions. The result, according to patient input, will be improved quality of life due to fewer treatment induced toxicities, disease regression, an oral therapy permitting at home use, and reduced infusion time. For patients who are identified with the unique biomarker (MSI-H/dMMR), the therapy delivers on the promise of precision medicine guiding treatment decisions for advanced endometrial cancer.
Anything Else?
The standard of care for patients with advanced or recurrent endometrial cancer is multiagent systemic chemotherapy, which includes Carbotaxol in the first line setting. In addition to being quite toxic, this combination therapy has, according to our patient input, low response rates which creates an urgent, unmet need to provide treatment options that yield better outcomes for this patient population: outcomes that include fewer side effects contributing to an improved quality of life, an extension in progression free survival and overall survival.
The patients who received Pembrolizumab monotherapy or Pembrolizumab + Lenvatinib reported significant improvements in health status with respect to physical function and overall quality of life. According to patients, the therapy has fewer side effects compared to previously administered therapies. Patients expressed their profound gratitude for having been able to access the therapy under review because for the most part, it has delivered a robust, durable, safe and effective response compared to previously accessed therapies with a substantially favorable toxicity profile. This was repeatedly stressed throughout the captured data. They also appreciated the shorter infusion time of the therapy and the convenient use of an oral therapy, easily administered at home. Patients stated they wish to continue to avail themselves of the therapy and wish same for others who qualify.
The use of Pembrolizumab monotherapy in the MSI-H/dMMR patient and Pembrolizumab + Lenvatinib in the MSS/pMMR patients demonstrated a level of benefit unlike any other previously accessed treatment, as per the captured data in APPENDIX 1. These patients were permitted to resume a “normal, active lifestyle” which patients credit entirely to the therapy. They were once again engaged in life in a meaningful way – contributing members of society, their families and their communities. The use of the therapy under review helps to address the urgent, unmet need that currently exists in the management of metastatic/recurrent endometrial cancer.
If publicly funded:
Pembrolizumab would be an extremely important therapeutic option for patients whose progressive disease test positive for MSI-H/dMMR and whose disease has been deemed to be inoperable or metastatic/recurrent, and
Pembrolizumab + Lenvatinib would be an equally important therapeutic option for the MSS/pMMR patient population whose progressive disease has been deemed to be inoperable or metastatic/recurrent
Funding these therapies in the appropriate settings (based on the identification of a unique biomarker), aligns well with the patient perspectives captured within this submission. Interviewed patients and their caregivers strongly supported the need for a positive funding recommendation be issued for Pembrolizumab monotherapy and Pembrolizumab + Lenvatinib for the treatment of MSI-H/dMMR and MSS/pMMR advanced endometrial cancer. The interviewed advanced endometrial patient population whose voice was captured and submitted herein underscores that the therapy under review aligns well with the identified need for a new, effective, quickly and easily administered, less toxic treatment option that is capable of maintaining a high quality of life. Survey data provided by CCS for this submission further underscores that patients and caregivers are asking for treatments that are more effective, but with non-debilitating side effects so they can lead a better quality of life, with the longest remission possible and fewer visits to the clinic.
Patient Group Conflict of Interest Declaration — Colorectal Cancer Resource & Action Network, Canadian Cancer Society, and Canadian Cancer Survivor Network
To maintain the objectivity and credibility of the CADTH CDR and pCODR programs, all patients in the drug review processes must disclose any real, potential, or perceived conflicts of interest. This Patient Group Conflict of Interest Declaration is required for participation. Declarations made do not negate or preclude the use of the patient group input. CADTH may contact your group with further questions, as needed.
Did you receive help from outside your patient group to complete this submission? If yes, please detail the help and who provided it.
No.
Did you receive help from outside your patient group to collect or analyze data used in this submission? If yes, please detail the help and who provided it.
No
List any companies or organizations that have provided your group with financial payment over the past two years AND who may have direct or indirect interest in the drug under review.
Table 1: Conflict of Interest Declaration for the Colorectal Cancer Resource & Action Network
Company | Check Appropriate Dollar Range | |||
|---|---|---|---|---|
$0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 | |
Merck | — | — | X | — |
Did you receive help from outside your patient group to complete this submission? If yes, please detail the help and who provided it.
No.
Did you receive help from outside your patient group to collect or analyze data used in this submission? If yes, please detail the help and who provided it.
The network of the Division of Gynecologic Oncology at McGill University Health Center provided the testimonials which were written by patients. One nurse assisted a caregiver by writing down his verbal testimonial as indicated in the testimonial itself. They also shared our survey directly with patients.
List any companies or organizations that have provided your group with financial payment over the past two years AND who may have direct or indirect interest in the drug under review.
Table 2: Conflict of Interest Declaration for the Canadian Cancer Society
Company | Check Appropriate Dollar Range | |||
|---|---|---|---|---|
$0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 | |
Merck | — | — | — | X |
Did you receive help from outside your patient group to complete this submission? If yes, please detail the help and who provided it.
No.
Did you receive help from outside your patient group to collect or analyze data used in this submission? If yes, please detail the help and who provided it.
No.
List any companies or organizations that have provided your group with financial payment over the past two years AND who may have direct or indirect interest in the drug under review.
Table 3: Conflict of Interest Declaration for the Canadian Cancer Survivor Network
Company | Check Appropriate Dollar Range | |||
|---|---|---|---|---|
$0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 | |
Merck | — | — | X | — |
Clinician Input
Ontario Health (Cancer Care Ontario) Gynecology Cancer Drug Advisory Committee
About the Ontario Health (Cancer Care Ontario) Gynecology Cancer Drug Advisory Committee
OH-CCO’s Drug Advisory Committees provide timely evidence-based clinical and health system guidance on drug-related issues in support of CCO’s mandate, including the Provincial Drug Reimbursement Programs (PDRP) and the Systemic Treatment Program.
Information Gathering
This input was jointly discussed with the listed DAC members.
Current Treatments
Pembrolizumab monotherapy: This therapy would be for individuals with mismatch repair deficiency (dMMR)/MSI-H endometrial cancer (EC) that has progressed after chemotherapy (after adjuvant chemotherapy in advanced stage or after chemotherapy for recurrent disease). Currently there are no agents that have specific approval for treatment of recurrent/progressive EC. At time of recurrence there are limited chemotherapy options and trial of carboplatin/taxol retreatment or Adriamycin at progression is current standard of care. The response to these limited chemotherapy options is poor. There are limited options with clinical trial enrolment for patients with advanced EC. There is currently no evidence for another line of therapy and therefore best supportive care would be an option. There are no special access programs for this population of patients. There are a limited subset of patients that are ER/PR positive that would be eligible for hormonal therapy (ex. Letrozole). The hormonal therapy available has limited low-level data and not Health Canada approved indication.
Lenvatinib in combination with Pembrolizumab: Currently there are no agents that have specific approval for treatment of recurrent/progressive EC. At time of recurrence there are limited chemotherapy options and trial of carboplatin/taxol retreatment or Adriamycin at progression is current standard of care. The response to these limited chemotherapy options is poor. There are limited options with clinical trial enrolment for patients with advanced EC. There is currently no evidence for another line of therapy and therefore best supportive care would be an option. There are no special access programs for this population of patients. There are a limited subset of patients that are ER/PR positive that would be eligible for hormonal therapy (ex. Letrozole). The hormonal therapy available has limited low-level data and not Health Canada approved indication.
Treatment Goals
Pembrolizumab monotherapy: The most important goals would be prolonged life, delay disease progression, symptomatic relief, partial response, full response, improved health-related quality or life, and decreased treatment toxicity.
Lenvatinib in combination with Pembrolizumab: The most important goals would be prolonged life, delay disease progression, symptomatic relief, partial response, full response, and improved health-related quality or life.
Treatment Gaps (Unmet needs)
Considering the treatment goals, please describe goals (needs) that are not being met by currently available treatments.
Pembrolizumab monotherapy: Currently there are no agents that have specific approval for treatment of recurrent/progressive dMMR/MSI-H endometrial cancer. At time of recurrence there are limited chemotherapy options and trial of carboplatin/taxol followed by Adriamycin at progression. There is currently no standard of care for these patients. Most patients do not response to available treatments and become refractory to current treatment options. Treatments are needed that are better tolerated.
Lenvatinib in combination with Pembrolizumab: Currently there are no agents that have specific approval for treatment of recurrent/progressive endometrial cancer. At time of recurrence there are limited chemotherapy options and trial of carboplatin/taxol followed by Adriamycin at progression. There is currently no standard of care for these patients. Most patients do not response to available treatments and become refractory to current treatment options. Treatments are needed that are better tolerated.
Which patients have the greatest unmet need for an intervention such as the drug under review?
Pembrolizumab monotherapy: Patients with dMMR/MSI-H EC would have the greatest unmet need. There are current no options for dMMR/MSI-H endometrial cancer in the recurrent setting after platinum-based therapy.
Lenvatinib in combination with Pembrolizumab: There are current no options for endometrial cancer in the recurrent setting after platinum-based therapy. All patients with recurrent/progressive endometrial cancer post-platinum therapy would have the greatest unmet need.
Place in Therapy
How would the drug under review fit into the current treatment paradigm?
Pembrolizumab monotherapy: There is no established second line treatment paradigm for these patients. It would replace potential retreatment with carboplatin/taxol or adriamycin.
Lenvatinib in combination with Pembrolizumab: There is no established second line treatment paradigm for these patients. It would replace potential retreatment with carboplatin/taxol or adriamycin.
Please indicate whether or not it would be appropriate to recommend that patients try other treatments before initiating treatment with the drug under review. Please provide a rationale from your perspective.
Pembrolizumab monotherapy: No we would not recommend other treatments since there are no other approved second line therapies for patient population. This would be the preferred option post platinum therapy at time of recurrence after failing chemotherapy.
Lenvatinib in combination with Pembrolizumab: No we would not recommend other treatments since there are no other approved second line therapies for patient population. This would be the preferred option post platinum therapy at time of recurrence after failing chemotherapy.
How would this drug affect the sequencing of therapies for the target condition?
Pembrolizumab monotherapy: There is no current second line option and this would provide patients a second-line option. After failing platinum-based therapy, proceed to pembrolizumab monotherapy.
Lenvatinib in combination with Pembrolizumab: There is no current second line option and this would provide patients a second-line options. After failing platinum-based therapy, proceed to pembrolizumab in combo with lenvatinib.
Which patients would be best suited for treatment with the drug under review?
Pembrolizumab monotherapy: Individuals with dMMR)/MSI-H endometrial cancer (EC) that have progressed after platinum-based chemotherapy (after adjuvant chemotherapy in advanced stage or after chemotherapy for recurrent disease).
Lenvatinib in combination with Pembrolizumab: All EC patients that have recurred or progressed after platinum-based chemotherapy.
How would patients best suited for treatment with the drug under review be identified?
Pembrolizumab monotherapy: dMMR/MSI-H and evidence of recurrence on imaging (CT, MRI, PET Scan), biopsy, and examination. Immunohistochemisty for MMR is performed reflexively on all newly diagnosed cases of EC in Ontario and therefore dMMR patients are easily identified at time of recurrence/progression.
Lenvatinib in combination with Pembrolizumab: Evidence of recurrence on imaging (CT, MRI, PET Scan), biopsy, and examination.
Which patients would be least suitable for treatment with the drug under review?
Pembrolizumab monotherapy: Patients that are contraindication to immunotherapy, chemo-naïve, and MMR intact.
Lenvatinib in combination with Pembrolizumab: Patients that are contraindication to immunotherapy or Lenvatinib and chemo-naïve.
Is it possible to identify those patients who are most likely to exhibit a response to treatment with the drug under review?
Pembrolizumab monotherapy: Yes. Reflex MMR testing.
Lenvatinib in combination with Pembrolizumab: While a greater benefit from treatment was seen in dMMR, all patients regardless of MMR status should achieve benefit.
What outcomes are used to determine whether a patient is responding to treatment in clinical practice?
Pembrolizumab monotherapy: Imaging, clinical exam, and symptomatic improvement.
Lenvatinib in combination with Pembrolizumab: Imaging, clinical exam, and symptomatic improvement.
What would be considered a clinically meaningful response to treatment?
Pembrolizumab monotherapy: Reduction in the frequency or severity of symptoms, improvement of symptoms, disease response or stability.
Lenvatinib in combination with Pembrolizumab: Reduction in the frequency or severity of symptoms, improvement of symptoms, disease response or stability.
How often should treatment response be assessed?
Pembrolizumab monotherapy: As per standard of care.
Lenvatinib in combination with Pembrolizumab: As per standard of care.
What factors should be considered when deciding to discontinue treatment?
Pembrolizumab monotherapy: Disease progression or intolerable side effects.
Lenvatinib in combination with Pembrolizumab: Disease progression or intolerable side effects.
What settings are appropriate for treatment with the drug under review?
Pembrolizumab monotherapy: Hospital (outpatient clinic)
Lenvatinib in combination with Pembrolizumab: Hospital (outpatient clinic)
For non-oncology drugs, is a specialist required to diagnose, treat, and monitor patients who might receive the drug under review?
N/A
Additional Information
Pembrolizumab monotherapy: This therapy provides high response rate in long term responders. The current chemotherapy does not provide a durable response. More tolerable toxicity profile compared to alternatives.
Lenvatinib in combination with Pembrolizumab: This therapy provides high response rate in long term responders. The current chemotherapy does not provide a durable response. More tolerable toxicity profile compared to alternatives.
Conflict of Interest Declarations
To maintain the objectivity and credibility of the CADTH drug review programs, all patients in the drug review processes must disclose any real, potential, or perceived conflicts of interest. This conflict of interest declaration is required for participation. Declarations made do not negate or preclude the use of the clinician group input. CADTH may contact your group with further questions, as needed. Please see the Procedures for CADTH Drug Reimbursement Reviews (section 6.3) for further details.
Did you receive help from outside your clinician group to complete this submission? If yes, please detail the help and who provided it.
The DAC received secretariat support from Ontario Health-Cancer Care Ontario provincial drug reimbursement specialist.
Did you receive help from outside your clinician group to collect or analyze any information used in this submission? If yes, please detail the help and who provided it.
No
List any companies or organizations that have provided your group with financial payment over the past two years AND who may have direct or indirect interest in the drug under review. Please note that this is required for each clinician who contributed to the input — please add more tables as needed (copy and paste). It is preferred for all declarations to be included in a single document.
Declaration for Clinician 1
Name: Dr. Sarah Ferguson
Position: OH-CCO Gynecological Drug Advisory Committee Lead
Date: 08/12/2021
Table 4: Conflict of Interest Declaration for Ontario Health (Cancer Care Ontario) Gynecology Cancer Drug Advisory Committee Clinician 1
Company | Check Appropriate Dollar Range | |||
|---|---|---|---|---|
$0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 | |
N/A | — | — | — | — |
Declaration for Clinician 2
Name: Dr. Stephen Welch
Position: OH-CCO Gynecological Drug Advisory Committee Member
Date: 08/12/2021
Table 5: Conflict of Interest Declaration for Ontario Health (Cancer Care Ontario) Gynecology Cancer Drug Advisory Committee Clinician 2
Company | Check Appropriate Dollar Range | |||
|---|---|---|---|---|
$0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 | |
Merck (Advisory Board) | — | — | — | — |
Eisai Limited (Advisory Board) | — | — | — | — |
Declaration for Clinician 3
Name: Dr. Josee-Lyne Ethier
Position: OH-CCO Gynecological Drug Advisory Committee Lead
Date: 08/12/2021
Table 6: Conflict of Interest Declaration for Ontario Health (Cancer Care Ontario) Gynecology Cancer Drug Advisory Committee Clinician 3
Company | Check Appropriate Dollar Range | |||
|---|---|---|---|---|
$0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 | |
Merck (Advisory Board) | — | — | — | — |
Merck (Speaker) | — | — | — | — |
Declaration for Clinician 4
Name: Dr. Julie Ann Francis
Position: OH-CCO Gynecological Drug Advisory Committee Lead
Date: 08/12/2021
Table 7: Conflict of Interest Declaration for Ontario Health (Cancer Care Ontario) Gynecology Cancer Drug Advisory Committee Clinician 4
Company | Check Appropriate Dollar Range | |||
|---|---|---|---|---|
$0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 | |
N/A | — | — | — | — |
Declaration for Clinician 5
Name: Dr. Leah Jutzi
Position: OH-CCO Gynecological Drug Advisory Committee Lead
Date: 08/12/2021
Table 8: Conflict of Interest Declaration for Ontario Health (Cancer Care Ontario) Gynecology Cancer Drug Advisory Committee Clinician 5
Company | Check Appropriate Dollar Range | |||
|---|---|---|---|---|
$0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 | |
N/A | — | — | — | — |
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up-to-date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
Redactions: Confidential information in this document may be redacted at the request of the sponsor in accordance with the CADTH Drug Reimbursement Review Confidentiality Guidelines
Stakeholder Input: The views expressed in each submission are those of the submitting organization or individual; not necessarily the views of CADTH or of other organizations. As such, they are independent of CADTH and do not necessarily represent or reflect the view of CADTH. No endorsement by CADTH is intended or should be inferred. By filing with CADTH, the submitting organization or individual agrees to the full disclosure of the information. CADTH does not edit the content of the submissions.
CADTH does use reasonable care to prevent disclosure of personal information in posted material; however, it is ultimately the submitter’s responsibility to ensure no identifying personal information or personal health information is included in the submission. The name of the submitting organization or individual and all conflict of interest information are included in the submission; however, the name of the author, including the name of an individual patient or caregiver submitting the patient input, are not posted.
Accessibility: CADTH is committed to treating people with disabilities in a way that respects their dignity and independence, supports them in accessing material in a timely manner, and provides a robust feedback process to support continuous improvement. All materials prepared by CADTH are available in an accessible format. Where materials provided to CADTH by a submitting organization or individual are not available in an accessible format, CADTH will provide a summary document upon request. More details on CADTH’s accessibility policies can be found here.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.