CADTH Reimbursement Review
Entrectinib (Rozlytrek)
Sponsor: Hoffmann-La Roche Limited
Indication: For the treatment of extracranial solid tumours with NTRK gene fusion.
This multi-part report includes:
Clinical Review
Pharmacoeconomic Review
Ethics Review
Stakeholder Input
Clinical Review
Abbreviations
AE
adverse event
ALK
anaplastic lymphoma kinase
BICR
blinded independent central review
BOR
best objective response
CBCN
Canadian Breast Cancer Network
CBR
clinical benefit rate
CCC
Colorectal Cancer Canada
CCOD
clinical cut-off date
CI
confidence interval
CMN
congenital mesoblastic nephroma
CNS
central nervous system
CR
complete response
CRC
colorectal cancer
DAC
Drug Advisory Committee
DLT
dose-limiting toxicity
DOR
duration of response
ECOG PS
Eastern Cooperative Oncology Group Performance Status
EGFR
epidermal growth factor receptor
EORTC
European Organisation for Research and Treatment of Cancer
FH
Flatiron Health
FISH
fluorescence in situ hybridization
FMI
Foundation Medicine Incorporated
GI
gastrointestinal
GHS
global health status
GIST
gastrointestinal stromal tumour
GMI
growth modulation index
HNT
head, neck, and thyroid
HR
hazard ratio
HRQoL
health-related quality of life
ICC
intraclass correlation coefficients
IC-ORR
intracranial objective response rate
IC-DOR
intracranial duration of response
IC-PFS
intracranial progression-free survival
IFS
infantile fibrosarcoma
KIT
KIT proto-oncogene
LCC
Lung Cancer Canada
LHF
Lung Health Foundation
MAIC
matching-adjusted indirect comparison
MASC
mammary analogue secretory carcinoma
mCRC
metastatic colorectal cancer
MID
minimal important difference
MSI-H
microsatellite instability high
NE
not estimable
NGS
next-generation sequencing
NOC/c
Notice of Compliance with conditions
NSCLC
non–small cell lung carcinoma
NTRK
neurotrophic tyrosine receptor kinase
OH-CCO
Ontario Health Cancer Care Ontario
OLA
Ontario Lung Association
ORR
objective response rate
OS
overall survival
PD
progressive disease
PFS
progression-free survival
PK
pharmacokinetic
PR
partial response
PRO
patient-reported outcome
PS
performance status
QLQ-C30
Quality of Life Questionnaire Core 30
QLQ-CR29
Quality of Life Questionnaire Colorectal Cancer 29
QLQ-LC13
Quality of Life Questionnaire Lung Cancer 13
RECIST 1.1
Response Evaluation Criteria in Solid Tumours Version 1.1
ROS1
ROS proto-oncogene 1
RP2D
recommended phase II dose
SD
standard deviation
STS
soft tissue sarcoma
TRAE
treatment-related adverse event
TRK
tropomyosin receptor kinase
TTD
time to discontinuation
TTP
time to progression
TTR
time to tumour response
Executive Summary
An overview of the submission details for the drug under review is provided in Table 1.
Item | Description |
|---|---|
Drug product | Entrectinib (Rozlytrek) capsules, 100 mg and 200 mg, oral |
Indication | For the treatment of adult patients who have unresectable, locally advanced or metastatic extracranial solid tumours (including brain metastases) with NTRK gene fusion without a known acquired resistance mutation and no satisfactory treatment options |
Reimbursement request | As per indication |
Health Canada approval status | NOC/c |
Health Canada review pathway | Standard |
NOC/c date | February 10, 2020 |
Sponsor | Hoffmann-La Roche Limited |
NOC/c = Notice of Compliance with conditions; NTRK = neurotrophic tyrosine receptor kinase.
Introduction
The neurotrophic tyrosine receptor kinase (NTRK) genes encode the neurotrophin family of receptors.1 The fusion of NTRK genes results from chromosomal rearrangements that — based on preclinical data — lead to constantly activated downstream signalling pathways without the need for ligands.2 Although reported to be prevalent in 0.28% of all solid cancers, NTRK oncogenic fusions are observed with variable frequency across a spectrum of pediatric and adult cancers. The frequency depends, in part, on the number of patients screened and on the NTRK fusion detection technique used.1 In adults in Canada, the 3 most common cancers are lung, colorectal, and breast cancer.3 NTRK fusions are generally less prevalent in common cancers (presenting in 0.1% to 1% of non–small cell lung cancers [NSCLCs]4-6 and in 2% to 3% of sporadic colorectal cancers [CRCs]4); however, NTRK fusion is more common in certain colorectal tumours with high levels of microsatellite instability (MSI-H)7,8 and in primary thyroid cancers (6%).9 In contrast, NTRK fusions are nearly ubiquitous among rare cancer types, such as mammary analogue secretory carcinoma (MASC) and infantile fibrosarcoma (IFS).4,10
Entrectinib is an inhibitor of tropomyosin receptor kinases (TRK) TRKA, TRKB, and TRKC (encoded by the NTRK1, NTRK2, and NTRK3 genes, respectively), ROS proto-oncogene 1 (ROS1) (encoded by the gene ROS1), and anaplastic lymphoma kinase (ALK) (encoded by the gene ALK). Entrectinib is indicated for the treatment of adult patients who have unresectable, locally advanced or metastatic extracranial solid tumours (including brain metastases) with NTRK gene fusion without a known acquired resistance mutation and no satisfactory treatment options. Entrectinib received a Notice of Compliance with conditions (NOC/c) for this indication on February 10, 2020, from Health Canada pending the results of new information to verify its clinical benefit. The sponsor’s reimbursement request is per the Health Canada–approved indication.
The product monograph states that a validated assay is required for the selection of patients with NTRK fusion–positive unresectable, locally advanced or metastatic extracranial solid tumours, including brain metastases. NTRK fusion–positive status should be established before initiating entrectinib therapy.
Entrectinib is available as 100 mg and 200 mg capsules. The recommended dose is 600 mg orally once daily. From the starting dose of 600 mg once daily, the dose can be reduced twice (i.e., to 400 mg once daily and then to 200 mg once daily) if there is a need to manage adverse events (AEs). It is recommended in the product monograph that patients are treated until disease progression or unacceptable toxicity. Health Canada has not authorized an indication for entrectinib for pediatric use; there are no recommendations in the product monograph regarding dosing in pediatric patients.
The objective of this systematic review is to assess the beneficial and harmful effects of entrectinib (600 mg orally once daily) for the treatment of adult patients who have unresectable, locally advanced or metastatic extracranial solid tumours (including brain metastases) with NTRK gene fusion without a known acquired resistance mutation and no satisfactory treatment options.
Stakeholder Perspectives
The information in this section is a summary of input provided by the patient groups who responded to CADTH’s call for patient input and from clinical experts consulted by CADTH for the purpose of this review.
Patient Input
Four patient groups provided input into CADTH’s review: Lung Health Foundation (LHF); Lung Cancer Canada (LCC); Colorectal Cancer Canada (CCC); and the Canadian Breast Cancer Network (CBCN). All of the groups obtained information through surveys to support their input. Patient groups expressed the need for treatments that could extend progression-free survival (PFS), delay disease progression, relieve cancer-related symptoms, improve quality of life, and minimize side effects from treatment. Also, patents wish to reduce the impact of cancer on their ability to care for children and dependents, continue working, spend time with loved ones, participate in social activities, travel, maintain friendships, and pursue personal interests. Similar to the clinicians who provided input to CADTH, the patient groups highlighted inconsistency across Canadian jurisdictions with access to NTRK fusion testing. Patients emphasized a desire for NTRK fusion testing to be available earlier with the hope of avoiding exposure to alternative treatments that may be less effective and associated with more AEs than a therapy targeting tropomyosin receptor kinase (TRK).
Clinician Input
Input From Clinical Experts Consulted by CADTH
A panel of 3 clinical oncologists from across Canada provided input for this review. Given that entrectinib is approved for use in a manner that is independent of tumour histology (with the exception of primary central nervous system [CNS] tumours), each of the clinicians on the review team has expertise in the diagnosis and management of different types of primary tumours. The clinicians consulted by CADTH felt that it was difficult to fully characterize the unmet need for patients who could be eligible for treatment with entrectinib. This is due to the breadth of potentially advanced solid tumours that may harbour NTRK fusion mutations and to variability in the availability and effectiveness of potential alternative therapies. However, the clinical oncologists agreed that, in the case of metastatic solid malignancies, virtually all patients eventually progress on currently available therapies, with the possible exception of select patients with select cancer types who are receiving immunotherapies.
The clinicians noted that the appropriateness of recommending that patients try other treatments before initiating treatment with entrectinib would depend on the cancer subtype and the efficacy of front-line therapy. As with the patient group input, the clinical experts consulted by CADTH agreed that TRK-targeting therapies, such as entrectinib, should be considered early in the course of treating cancers involving NTRK fusion. This agreement was based on the following rationale:
Entrectinib may be associated with higher response rates, have a better safety profile, and be more tolerable than existing alternatives.
Given its mechanism of action and the available evidence, the clinical experts believed that entrectinib would be efficacious in patients with NTRK gene fusions and advanced disease, regardless of the number of prior therapies.
Patients may no longer be fit for any systemic therapy after receiving alternative treatments (e.g., poor performance status).
The presence of NTRK fusion mutations is clinically actionable and the Canadian consensus guidelines recommend the use of TRK inhibitors as the preferred option for patients with NTRK fusion tumours.
Treatments targeting the tumour site (as opposed to the TRK) were perceived as likely to be less effective and potentially more toxic than a TRK-targeting therapy (i.e., entrectinib or larotrectinib), particularly for tumours where the alternative is chemotherapy. All clinicians noted that there is considerable variability in access to NTRK fusion testing across tumour sites and across Canadian jurisdictions. The determination of when to identify targeted therapies (such as entrectinib) as potential treatment options is influenced by whether or not tumours are routinely tested for NTRK fusion and the timing of such testing.
The clinicians noted that in adults, clinically meaningful outcomes could include objective response; non-progression; patient-reported improvements in ability to perform activities; improved survival; stabilization, improvement, or reduced severity of symptoms; or no deterioration in quality of life. Clinicians noted that treatment response is typically assessed every 3 months, and once response is established or remission achieved, the interval may be prolonged. The clinicians noted that treatment failure would be determined by disease progression, treatment intolerability, poor quality of life (e.g., poor PS), or patient request to discontinue treatment.
Clinician Group Input
Five clinician groups provided input into this review, including LCC and the Lung Cancer, Breast Cancer, Gastrointestinal (GI) Cancer, and Head, Neck, and Thyroid (HNT) Cancer Drug Advisory Committees (DACs) from Ontario Health Cancer Care Ontario (OH-CCO). The input from the clinician groups was similar to the input from the clinical experts consulted by CADTH with respect to the unmet medical needs of adult patients who have unresectable, locally advanced or metastatic extracranial solid tumours (including brain metastases) with NTRK gene fusion without a known acquired resistance mutation and no satisfactory treatment options. The clinician groups also noted that the place in therapy for entrectinib would vary depending on the tumour site, the availability of safe and effective alternative therapies, and the timing of access to NTRK fusion testing. Input regarding important end points, timing and criteria for evaluation, and likely discontinuation criteria was the same as that provided by the experts consulted by CADTH.
Clinical Evidence
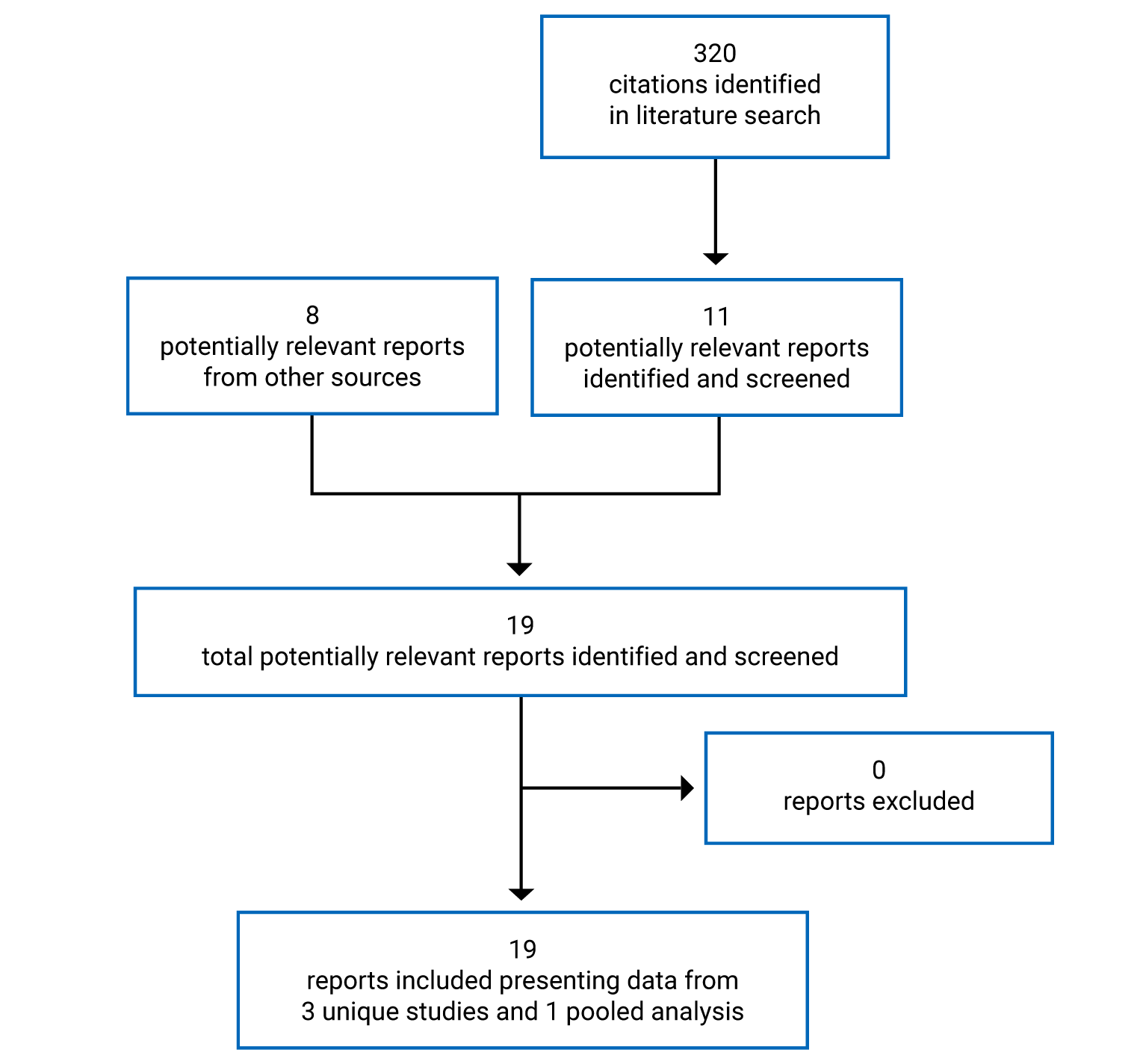
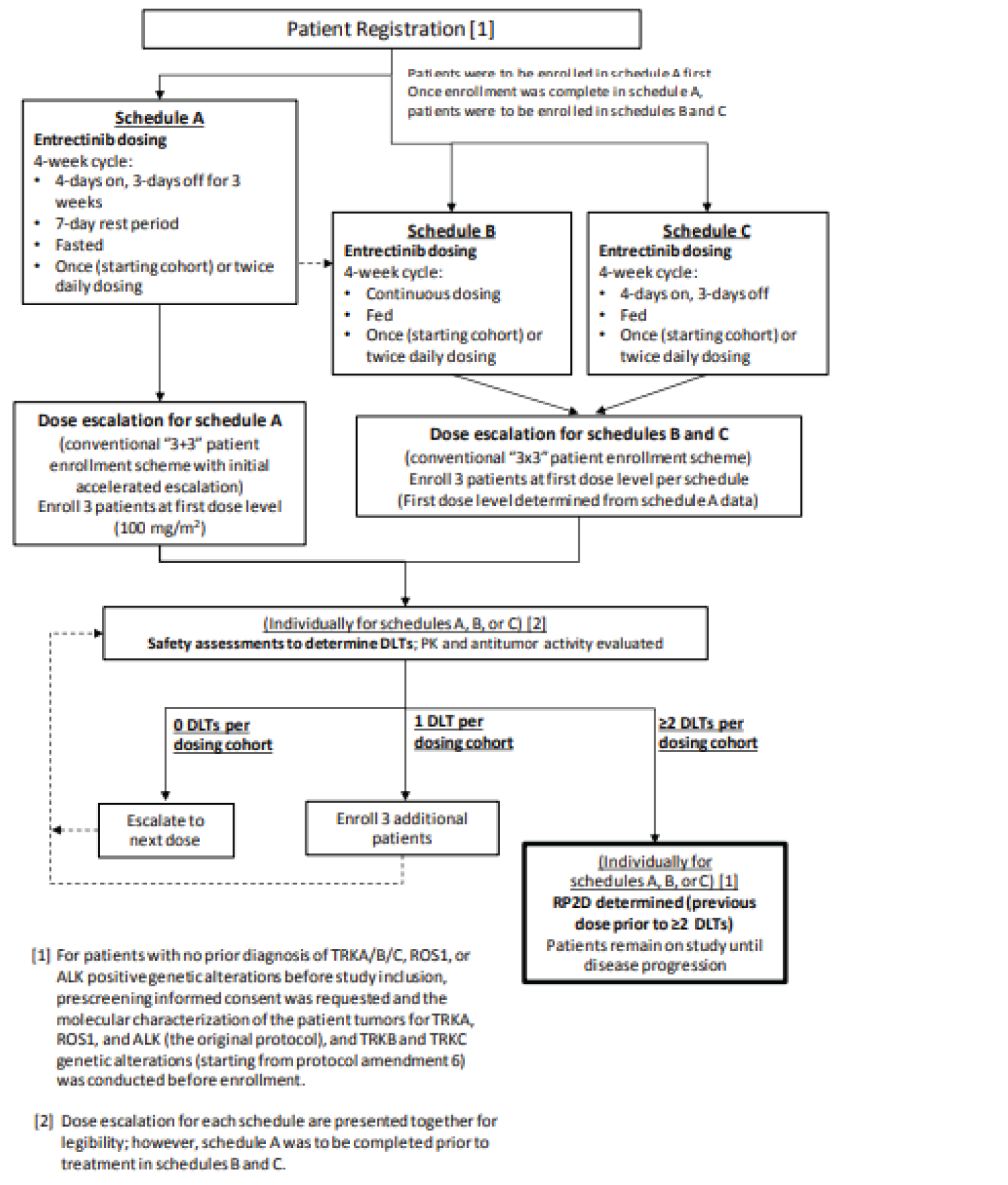
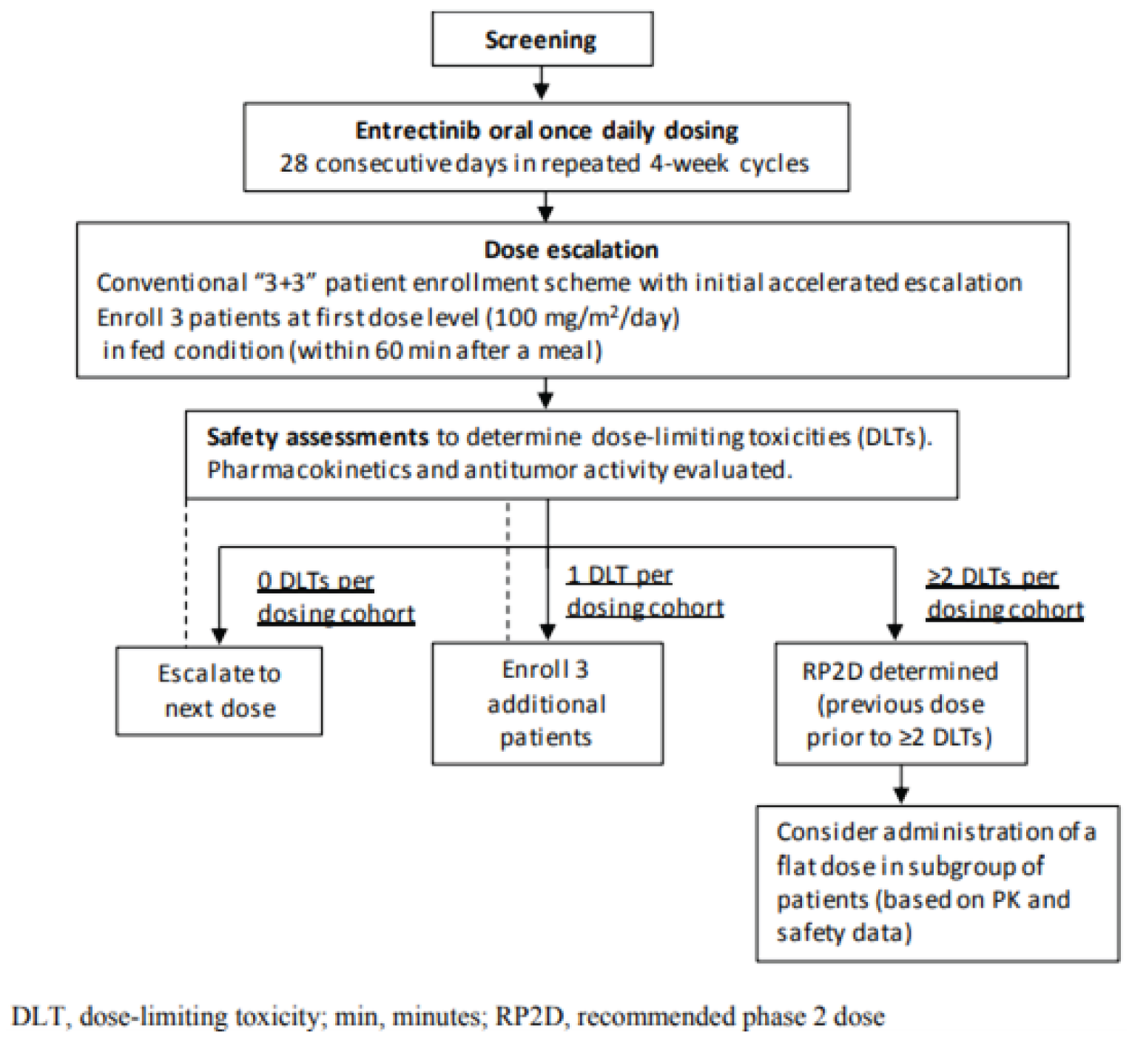
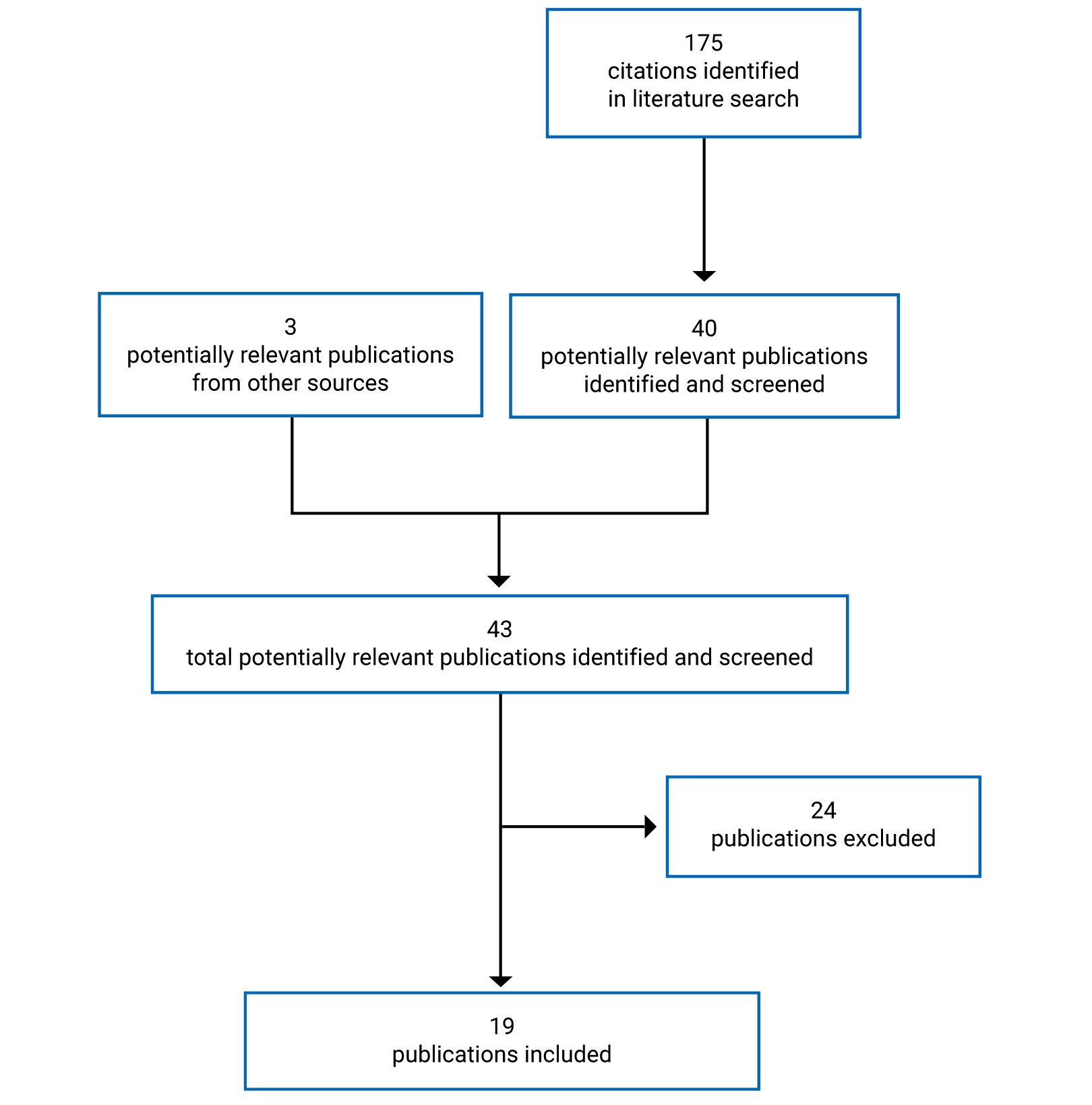
Pivotal Studies and Protocol Selected Studies
Description of Studies
The submission for entrectinib was based on a pooled analysis of 3 multi-centre, open-label, single-arm trials of entrectinib in adults with advanced or metastatic solid tumours: ALKA (phase I), STARTRK-1 (phase I), and STARTRK-2 (an ongoing phase II basket trial). The primary evaluation for the pooled analysis was based on a May 31, 2018, clinical cut-off date (CCOD), and the evaluation has subsequently been updated with larger sample sizes and longer follow-ups (October 31, 2018, and August 31, 2020). Whenever available, the CADTH report reflects the most recent analysis (CCOD of August 31, 2020).
The pooled analysis for the August 31, 2020 CCOD consisted of the following datasets:
NTRK safety-evaluable population (N = 193): all patients with an NTRK fusion–positive tumour who received at least 1 dose of entrectinib
NTRK efficacy-evaluable population (N = 121; 98% from STARTRK-2): all patients with NTRK fusion–positive extracranial primary tumours who received at least 1 dose of entrectinib, had measurable disease at baseline, and had at least 12 months of follow-up
NTRK efficacy-evaluable population with CNS metastases at baseline (N = 19, based on blinded independent central review [BICR] assessment): the subpopulation used for the evaluation of the “intracranial efficacy” end points.
Nearly all patients in the pooled analysis were from the STARTRK-2 trial, in which patients received entrectinib at the dosage recommended in the Canadian product monograph (i.e., a starting dosage of 600 mg once daily with up to 2 dose reductions permitted to manage AEs).
The primary outcomes in the pooled analysis were objective response rate (ORR), defined as the proportion of patients with a best overall response (BOR) of either complete response (CR) or partial response (PR), according to Response Evaluation Criteria in Solid Tumours Version 1.1 (RECIST 1.1) and as determined by BICR; duration of response (DOR); and best objective response (BOR). Secondary efficacy end points in the pooled analysis included time to tumour response (TTR); clinical benefit rate (CBR), defined as the proportion of patients with CR, PR, or stable disease for at least 6 months; PFS; and overall survival (OS). In addition, the sponsor pre-specified the following intracranial efficacy end points that were evaluated in the pooled subset of patients who had CNS metastases at baseline: intracranial ORR (IC-ORR), intracranial duration of response (IC-DOR), and intracranial progression-free survival (IC-PFS). Health-related quality of life (HRQoL) data were evaluated only in the STARTRK-2 trial and included change from baseline in the European Organisation for Research and Treatment of Cancer (EORTC) Quality of Life Questionnaire Core 30 (QLQ-C30); the EORTC Quality of Life Questionnaire Lung Cancer 13 (QLQ-LC13) for the subset of patients with NSCLC; and the EORTC Quality of Life Questionnaire Colorectal Cancer 29 (QLQ-29) for the subset of patients with metastatic colorectal cancer (mCRC).
The NTRK efficacy-evaluable analysis set (N = 121) was 51.2% female, with a mean age of 55.9 years (64.5% were less than 65 years of age). Baseline Eastern Cooperative Oncology Group Performance Status (ECOG PS) results were 0 (43.8%), 1 (47.1%), or 2 (9.1%). The majority of patients had previously received some form of anti-cancer therapy (n = 97; 80.2%); 74 patients (61.2%) had received prior radiotherapy and 103 patients (85.1%) had received previous cancer surgery. Approximately 30.6% of patients did not have prior systemic anti-cancer therapy. For those with a history of prior systemic therapy, 28.9% had 1 line, 21.5% had 2 lines, 9.9% had 3 lines, and 5.8% had 4 lines. The most frequent systemic prior anti-cancer therapy was chemotherapy (n = 88; 72.7%), followed by targeted therapy (n = 24; 19.8%), immunotherapy (n = 13; 10.7%), and hormonal therapy (n = 10; 8.3%).
The solid tumour types that were reported for at least 5% of the patients included sarcoma (n = 26; 21.5%); MASC (n = 24; 19.8%), NSCLC (n = 22; 18.2%); thyroid cancer (n = 13; 10.7%); colon cancer (n = 10; 8.3%); and breast cancer (n = 7; 5.8%). Nearly all patients had metastatic disease at baseline (96.7%). The most common metastatic sites were lung (61.2%) and lymph nodes (55.4%). There were 19 patients (17.2%) with CNS metastases at baseline, as assessed by BICR, with 17 patients (14.0%) reporting prior radiotherapy of the brain.
Efficacy Results
A summary of the results for key efficacy end points is provided in Table 2. Unless otherwise noted, the efficacy results reported are from the August 2020 CCOD.
ORR: In the NTRK efficacy-evaluable dataset, the ORR by BICR was 61.2% (95% confidence interval [CI], 51.87 to 69.88). A BOR of CR or PR was demonstrated by 15.7% and 45.5% of patients, respectively. The point estimates for ORR ranged widely across tumour types, and the CIs reflected a high degree of uncertainty for many tumour types. At least 1 patient demonstrated a response to treatment in each of the tumour types, with the exception of neuroblastoma (n = 1). The ORR for the larger subgroup populations were generally consistent with the results for the overall population; however, a higher proportion of tumour response was reported among the patients with salivary MASC tumours (20 out of 24; 83.3% [95% CI, 62.6 to 95.3]) and a lower proportion was reported for those with colorectal carcinoma (2 out of 10; 20% [95% CI, 2.5 to 55.6]).
IC-ORR: The BICR-assessed IC-ORRs were 52.6% (95% CI, 28.86 to 75.55) and 63.6% (95% CI, 30.8 to 89.1) for all patients with baseline CNS disease (10 out of 19 responded) and those with measurable disease at baseline (7 out of 11 responded), respectively. A subgroup analysis demonstrated similar results for those who had no prior brain radiotherapy or brain radiotherapy greater than or equal to 6 months before the initiation of treatment (55.6%; 95% CI, 21.2 to 86.3 [n = 9]) and those with prior brain radiotherapy within 6 months of initiating treatment with entrectinib (50.0%; 95% CI, 18.7 to 81.3 [n = 10]).
TTR: The median times to objective response were 1.0 month (95% CI, 0.9 to 1.0) for the overall population and 1.3 months (95% CI, 0.9 to 2.8) for patients with CNS metastases at baseline.
DOR: The DOR among responders was 20.0 months (95% CI, 13.0 to 38.2). For the patients who demonstrated a CR or PR with entrectinib, responses of at least 6 months, 12 months, 18 months, 24 months, 30 months, and 36 months were reported for 58 patients (78%), 46 patients (62%), 32 patients (43%), 20 patients (27%), 10 patients (14%), and 4 patients (5%), respectively. The event-free probabilities were 0.82 (95% CI, 0.73 to 0.91) at 6 months, 0.66 (95% CI, 0.55 to 0.77) at 12 months, 0.49 (95% CI, 0.37 to 0.61) at 24 months, and 0.39 (95% CI, 0.24 to 0.53) at 36 months.
IC-DOR: The IC-DORs among responders were 17.2 months (95% CI, 7.4 to not estimable [NE]) and 22.1 months (95% CI, 7.4 to NE) for all patients with baseline CNS disease and those with measurable disease at baseline, respectively.
CBR: The CBR was 63.6% (95% CI, 54.8 to 71.7).
PFS: The median PFS was 13.8 months (95% CI, 10.1 to 19.9), with a total of 72 patients (59.5%) experiencing progressive disease (PD) or death at the August 31, 2020 CCOD. Updated subgroup analyses for PFS were not reported for the August 2020 CCOD.
IC-PFS: The median PFS was 10.1 months (95% CI, 6.3 to 26.7), with a total of 13 patients (68.4%) experiencing CNS PD or death at the August 2020 CCOD (5 PD events and 8 deaths).
OS: The median OS was 33.8 months (95% CI, 23.4 to 46.4); a total of 49 patients (40.5%) had died at the time of the August 2020 CCOD. Among those with CNS metastases at baseline, the median OS was 19.9 months (95% CI, 7.9 to NE), with 52.6% having died at the time of the August 2020 CCOD.
Table 2: Summary of Key Results From Integrated Efficacy Analysis
Analysis | Total population (N = 121) | With CNS metastases at baseline (N = 19) |
|---|---|---|
Overall response rate | ||
Responders, n (%) | 74 (61.2) | 12 (63.2) |
ORR (95% CI) | 61.2 (51.87 to 69.88) | 63.2 (38.36 to 83.71) |
Best response, n (%) | ||
CR | 19 (15.7) | 1 (5.3) |
PR | 55 (45.5) | 11 (57.9) |
SD | 13 (10.7) | 4 (21.1) |
PD | 13 (10.7) | 2 (10.5) |
Non-CR or non-PD | 6 (5.0) | 0 |
Missing or unevaluable | 15 (12.4) | 1 (5.3) |
Clinical benefit rate | ||
Patients with event, n (%) | 77 (63.6) | NR |
Clinical benefit rate (95% CI) | 63.6% (54.8 to 71.7) | NR |
TTR (months) | ||
Median TTR (95% CI) | 1.0 (0.9 to 1.0) | 1.3 (0.9 to 2.8) |
DOR (months) | ||
Median DOR (95% CI) | 20.0 (13.0 to 38.2) | 15.2 (6.0 to 29.4) |
PFS | ||
Median PFS, (95% CI) | 13.8 (10.1 to 19.9) | 11.7 (5.1 to 30.3) |
OS | ||
Deaths, n (%) | 49 (40.5) | 10 (52.6) |
Median OS, (95% CI) | 33.8 (23.4 to 46.4) | 19.9 (7.9 to NE) |
CI = confidence interval; CR = complete response; DOR = duration of response; NE = not estimable; NR = not reported; ORR = overall response rate; OS = overall survival; PD = progressive disease; PFS = progression-free survival; PR = partial response; SD = stable disease; TTR = time to response.
Source: Supplemental Efficacy Results (clinical cut-off date: August 31, 2020).11
Harms Results
Table 3 provides a summary of AEs reported in the safety-evaluable population (N = 193). Nearly all patients who were NTRK fusion–positive experienced at least 1 AE (99.5%); 46.1% of patients experienced at least 1 serious adverse event; 69.4% experienced at least 1 AE greater than or equal to grade 3. The proportions of patients with AEs leading to dose interruption or dose reduction were 54.4% and 26.9%, respectively. The proportion of patients with an AE leading to discontinuation was 14.5%.
The product monograph for entrectinib provides detailed recommendations for the management of AEs that require temporary interruption, dose reduction, or discontinuation of treatment with entrectinib. The clinical experts consulted by CADTH indicated that those recommendations are a reasonable reflection of how patients would be managed in clinical practice. The product monograph also includes black box warnings that the drug may cause congestive heart failure and fetal harm when administered to a pregnant woman. The clinical experts consulted by CADTH noted that the patients would likely be screened and monitored for risk factors and symptoms related to heart failure before treatment and during follow-up visits while on treatment. Overall, the clinical experts consulted by CADTH agreed that the safety and tolerability of entrectinib was reasonable.
Table 3: Summary of AEs (Safety-Evaluable Population)
AEs, n (%) | Safety-evaluable patients (N = 193) |
|---|---|
Patients with AE | 192 (99.5) |
Patients with SAE | 89 (46.1) |
Patients with NCI CTCAE ≥ grade 3 AE | 134 (69.4) |
Patients with AE leading to dose interruption | 105 (54.4) |
Patients with AE leading to discontinuation | 28 (14.5) |
Patients with AE leading to dose reduction | 52 (26.9) |
Patients with AE leading to death | 14 (7.3) |
AE = adverse event; NCI CTCAE = National Cancer Institute Common Terminology Criteria for Adverse Events; SAE = serious adverse event.
Source: Supplemental Efficacy Results (clinical cut-off date: August 31, 2020).11
Critical Appraisal
Internal Validity
Due to the rarity of NTRK fusion cancers, the sponsor conducted pooled analyses of efficacy and safety as the basis for the regulatory and reimbursement review submissions. Although the pooled analyses included patients from 3 trials, nearly all of the patients were from the STARTRK-2 trial (98% and 97% of patients in the efficacy and safety analyses sets, respectively). This reduces the potential uncertainty that can arise from between-study heterogeneity (e.g., differences in study design, objectives, phases, outcome measures, and eligibility criteria across trials) that has been previously noted by CADTH for larotrectinib. Despite the use of pooled analyses, the sample sizes for each individual cancer type were too small, as would be expected due to the low prevalence of NTRK gene fusions (9 out of 14 tumour types contained fewer than 10 patients); the resulting 95% CI was too wide to evaluate the consistency of the effect of entrectinib on different tumour types.
The efficacy end points were evaluated using BICR-assessed outcomes for the primary analyses (with investigator-assessed outcomes provided as a sensitivity analysis). This is an important design feature because the trials were all open-label, single-arm studies. DCR, PFS, and OS are important end points for evaluating the efficacy of cancer treatments; however, these cannot be interpreted in the absence of a control group. In addition, a number of survival outcomes (PFS, OS, and DOR) were analyzed using the Kaplan–Meier method to pool data across the 3 trials, which could be problematic given that traditional survival analysis methods (such as Kaplan–Meier curves) rely on the assumption that a single survival distribution can be used to estimate the survival outcome for all patients included in the analysis. However, as noted previously, nearly all of the patients were derived from a single trial (STARTRK-2), limiting concerns about the pooled approach for survival analyses. HRQoL analyses were conducted for only 1 of the trials (STARTRK-2), and these were limited by the open-label administration of entrectinib, the lack of a comparator group, the absence of statistical testing, and the small sample size for disease-specific instruments for those with NSCLC (N = 12) and mCRC (N = 7).
External Validity
The patient population in the pooled analysis was considered a reasonable reflection of the target population in Canada (i.e., adults with NTRK fusion–positive, unresectable, locally advanced or metastatic extracranial solid tumours). Not all solid tumour types were represented in the pooled analysis, and the majority of tumour types were seen in fewer than 10 patients. This resulted in wide CIs within subgroup analyses, reducing confidence in the generalizability of the results. Patients included in the pooled analysis had ECOG PS scores of 0, 1, or 2. The clinical experts noted the relatively fast median TTR along with the high rate of response and favourable toxicity profile. These are key considerations for potentially making entrectinib available to patients with an ECOG PS of 3 (a population that was not studied in the clinical trials) in situations where the oncologist believes that tumour-related symptoms are driving the PS.
Nearly all patients in the pooled analysis received entrectinib at the dosage recommended in the Canadian product monograph (i.e., a starting dosage of 600 mg once daily with up to 2 dose reductions permitted to manage AEs).
There were no direct or indirect comparisons filed by the sponsor to evaluate the comparative efficacy and safety of entrectinib versus larotrectinib or other alternative therapies.
Indirect Comparisons
No studies have directly compared entrectinib versus larotrectinib for patients with NTRK-positive tumours. The sponsor did not include an indirect comparison in its application to CADTH because it believes meaningful comparisons are not feasible due to the following challenges: NTRK fusions are expressed in only 1% or less of all solid tumours; patient enrolment in trials is low; ongoing trials are single arm and open label, and the study population is heterogeneous with regard to baseline characteristics (e.g., age, ECOG status, tumour site, and presence of CNS metastases). In the absence of direct or indirect evidence comparing entrectinib and larotrectinib in the submission, CADTH conducted a literature search to identify any relevant published indirect comparisons. CADTH identified 1 matching-adjusted indirect comparison (MAIC) of entrectinib versus larotrectinib in adult patients with NTRK gene fusion–positive tumours.
Description of Studies
Garcia-Foncillas et al. (2022) conducted a MAIC to compare the efficacy and safety of entrectinib and larotrectinib in adults with NTRK fusion–positive tumours. The MAIC was funded by the manufacturer of larotrectinib; therefore, patient-level data were available for the larotrectinib-treated patients, but not for entrectinib-treated patients. The data used for entrectinib were derived from the earlier May 31, 2018 and October 31, 2018 CCODs (i.e., a smaller sample size than the August 31, 2020 CCOD data included in the current submission to CADTH). Adult patients were selected for inclusion in the MAIC if they were TRK inhibitor-naive and had NTRK fusion (as determined by an independent review committee) and an ECOG PS of 2 or less. Patients were matched according to the following baseline characteristics: sex, age, race, ECOG PS, tumour type, metastatic disease (versus locally advanced, unresectable disease), NTRK fusion type, prior lines of systemic therapy for metastatic disease, and CNS metastases.
Efficacy Results
Garcia-Foncillas et al. (2022) reported that, compared with entrectinib, larotrectinib was associated with a statistically significantly greater duration of OS (hazard ratio [HR] = 0.43; 95% CI, 0.23 to 0.83; P < 0.05) and DOR (HR = 0.49; 95% CI, 0.25 to 0.98; P < 0.05). The authors reported no statistically significant difference for PFS (HR = 0.66; 95% CI, 0.42 to 1.03; P = 0.07) or ORR (risk difference [RD] = 3.8; 95% CI, –11.7 to 19.3; P = 0.63). Results were similar in sensitivity analyses applying different specifications for the MAIC and using a simulated treatment comparison method.
Harms Results
There were no statistically significant differences reported between larotrectinib and entrectinib for serious treatment-related adverse events (TRAEs) or TRAEs leading to discontinuation.
Critical Appraisal
Several key details from the MAIC were not provided in the published study, limiting CADTH’s ability to appraise the reported study. However, the primary limitation of the results is due to the unanchored nature of the comparison, which would require the inclusion of all prognostic factors and effect modifiers to ensure unbiased results. Due to this limitation and others, drawing firm conclusions based on the results of this MAIC is not recommended.
Intra-Patient Growth Modulation Index Analysis
The sponsor provided the intra-patient comparison of efficacy in a single-arm trial of entrectinib in tumour-agnostic indications. The sponsor’s objective was to generate and analyze evidence for the comparative effectiveness of entrectinib by exploring the role of intra-patient comparison as an alternative to a traditional comparator arm.
Populations and Methods
Analyses were conducted on retrospectively collected data from the STARTRK-2 trial to generate intra-patient comparisons. There were 3 cohorts of patients based on their prior systemic therapy in the metastatic setting and the presence or absence of documented progression:
In the “documented progression on prior therapy” cohort, patients received at least 1 systemic therapy for metastatic disease before commencing entrectinib, and there is clear documentation of PD on the most recent prior therapy, as captured in electronic case report forms.
In the “no documented progression on prior therapy” cohort, patients received at least 1 systemic therapy for metastatic disease before commencing entrectinib and had no documentation of PD on the most recent prior therapy. This cohort includes patients who stopped prior therapy due to toxicity, completion of the course, or other reasons.
In the “no prior therapy” cohort, patients received no prior systemic therapy for metastatic disease before starting entrectinib. However, they may have received prior (neo) adjuvant therapy.
A total of 71 patients with efficacy-evaluable NTRK fusion–positive disease who were enrolled in STARTRK-2 up to April 30, 2018 (data cut-off date: October 31, 2018) were included in the analysis. Of 71 patients, 51 patients had received systemic therapy before commencing entrectinib (38 had documented PD and 13 had no documented PD on the most recent prior systemic therapy); and 20 patients had not received prior systemic therapy. Among those who had received prior systemic therapy, 21 patients (41.2%) received 1 line, 20 patients (39.2%) received 2 lines, and 10 patients (19.6%) received 3 or more lines. The treatment regimens varied greatly within and between tumour types. The most common tumour types were sarcoma (22.5%), NSCLC (16.9%), MASC (16.9%), and thyroid cancer (9.9%).
The key analysis used was growth modulation index (GMI), as defined by the ratio of PFS on entrectinib to time to discontinuation (TTD) on the most recent prior therapy. TTD was chosen instead of time to progression (TTP) to measure the efficacy of the prior therapy due to the limited data available to define a TTP outcome reliably. A GMI ratio of greater than or equal to 1.3 was selected as the threshold to indicate a clinically meaningful benefit. Additional analyses explored TTD and ORR for entrectinib and prior systemic therapy.
Results
For patients with PD on prior systemic therapy, median GMI was 2.53 (range = 0.09 to 61.5) with 25 patients (65.8%) having a GMI of greater than or equal to 1.3. For GMI thresholds of greater than or equal to 1.5, greater than or equal to 1.8, and greater than or equal to 2.0, 23 patients (60.5%), 23 patients (60.5%), and 22 patients (57.9%), respectively, met these thresholds. Of 7 patients with a GMI of less than 1.0, 4 patients (57.1%) were censored for PFS.
Kaplan–Meier survival analysis showed that curves for PFS and TTD on entrectinib were similar (HR of PFS to TTD = 1.08; 95% CI, 0.6 to 1.9), with a median PFS of 11.2 months (95% CI, 6.7 to NE) and a median TTD of 9.9 months (94% CI, 7.3 to 14.8) on entrectinib. Both PFS and TTD on entrectinib were longer than TTD on most recent prior therapy, which had a median of 2.9 months (95% CI, 2.0 to 4.9). The ORRs for entrectinib were 60.5% (all PR) in patients with documented progression on prior therapy, 46.2% (all PR) in patients with no documented progression on prior therapy, and 80% (5 CR and 11 PR) in patients with no prior therapy.
The ORRs for most recent prior systemic therapy were 15.8% (1 CR and 5 PR) in patients with documented progression on prior therapy and 7.7% (1 PR) in patients with no documented progression on prior therapy.
Critical Appraisal
The following important limitations were identified:
GMI (or PFS ratio) is not a validated efficacy end point and does not take into consideration the impact of treatment on patient symptoms and quality of life (as acknowledged by the sponsor).
The sponsor was unable to obtain the PFS data on prior systematic therapy and had to rely on TTD as a surrogate for PFS. This is particularly problematic if patients continue prior therapy beyond disease progression because PFS is overestimated.
The GMI analysis assumes that tumour growth follows linear kinetics over time (i.e., the same growth rate at the time of diagnosis, for prior therapies, and at time of entrectinib treatment). Evidence suggests that this may not be a valid assumption and that some tumour growth may occur according to exponential or logarithmic rates. In addition, PFS is expected to decrease with successive lines of therapy.
Two additional underlying assumptions of the GMI analysis are required to make this approach meaningful: participant characteristics regarding all sources of heterogeneity of interest are consistent over time; and measurements compared in the ratio are compatible — i.e., PFS on entrectinib is defined and assessed in the same manner as TTD (as a surrogate for PFS) on most recent prior therapy, and all other reasons for an event are the same. However, neither condition has been met. It is unclear how patients’ baseline characteristics, such as age and ECOG PS, which change over time, affect disease progression.
The timing of tumour assessment while on entrectinib was controlled through standardized clinical trial protocols, but not when patients were receiving prior therapies outside of the clinical trial setting; RECIST 1.1 was used to assess entrectinib response, but not for prior therapies; and PFS on entrectinib was analyzed by BICR, whereas TTD on prior therapy was based on investigator assessment.
Summary
The results show longer PFS with entrectinib relative to the TTD with the last prior treatment; however, this observation relies on many assumptions, including the key assumption (i.e., TTD as a surrogate for PFS), which appears to be invalid, based on the information provided about the calculation of the GMI. There is no formal investigation of differences in the GMI by tumour type or other patient characteristics, and the descriptive individual GMI results suggest large variations in the GMI. It is unclear how some of the presented results were obtained or if inferences made with them are valid, given the intra-patient nature of the analysis. However, if the GMI can be considered a reliable comparison tool, it appears to support the case that entrectinib may be beneficial in many of the tumour types when other treatments have failed, and that this is the case across many patient characteristics, which would mitigate many of the concerns about patient heterogeneity (other than tumour type). Without inference (the presented CI), large variation in GMI is evident across the tumour types and remains a main limitation.
Exploratory Efficacy Analyses Comparing Entrectinib Against Standard of Care
The sponsor provided a report comparing OS in patients with NTRK fusion–positive solid tumours who were treated with entrectinib from the sponsor’s clinical trials (pooled dataset) against patients treated using standard of care from the Flatiron Health (FH) and Foundation Medicine Incorporated (FMI) clinico-genomic database.
Populations and Methods
The FH database is a US longitudinal database with de-identified data originating from approximately 280 cancer clinics and representing 2.8 million patients with cancer (the majority from community oncology settings). The FH data platform aggregates and processes patient-level data.
Median crude and matched, weighted OS durations were estimated through the Kaplan–Meier survival curve. HRs were estimated using weighted univariate Cox proportional hazards models for entrectinib-treated patients compared to non–entrectinib-treated patients. The index date for the end point analyzed is the start of entrectinib treatment for trial patients and the NTRK-positive test report date for those who received standard of care.
The nearest neighbour propensity score-matching model with replacement was used to perform the matching, with each match first done within each tumour type (i.e., direct match by tumour). Characteristics included in this analysis were tumour type and histology, age of patient, stage of cancer, number and type of previous treatment, and type of centre where patient is treated.
Patient Characteristics
Before matching, the study population with the same tumour types consisted of |||||| patients who received standard of care and |||||| entrectinib-treated patients. Compared with the standard-of-care group, patients in the entrectinib-treated group were younger (median age: ||||||||||||||||||||||||). The entrectinib-treated group included a higher proportion of women (||||||||||||||||||), treated patients only in academic centres, and had a lower proportion of patients with a history of smoking (||||||||||||||). Standard-of-care patients were more heavily treated at the index date (e.g., only |||||| had not received treatment before the index date versus |||||| in entrectinib patients), and were more likely to have stage IV disease at the time of initial diagnosis (||||||||||||). When the 2 cohorts were matched, the main analyses included only |||||| trial patients matched to |||||| standard-of-care patients, and only a moderate balance of cohorts could be achieved for the 4 a priori selected variables (i.e., ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Results
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Critical Appraisal
The following important limitations prevent firm conclusions from being drawn based on the results of this analysis:
The sample size for the comparison was very small, with only |||||| patients included in the standard-of-care group. CADTH acknowledges that NTRK fusion positive cancer is a rare condition; however, this remains an important limitation.
There was heterogeneity across the entrectinib and standard-of-care groups even after propensity score matching.
The groups were matched based solely on |||||| characteristics, which is not sufficient to control for potential confounding factors.
There were missing values in relevant covariates, such as ECOG PS (||||||||||||||||||||||||||||||||||||||||||) and number of metastatic sites (under-reported in the real-world evidence data) that prevented their inclusion in the a priori matching.
The sample size was too small to allow for exploration by subgroup of tumour types or lines of prior therapy (both of which were identified as subgroups of interest for CADTH’s review).
Patients received entrectinib in a clinical trial setting. The timing of tumour assessment while on entrectinib was controlled through standardized clinical trial protocols, but not when patients were receiving prior therapies outside of the clinical trial setting; RECIST 1.1 was used to assess entrectinib response, but not for prior therapies; and PFS on entrectinib was analyzed by BICR, whereas TTD on prior therapy was based on investigator assessment.
Summary
The results show a longer median OS with entrectinib compared with standard of care (|||||||||||||||||||||||||||||||||||||||||||||||| versus ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||). Important limitations with this analysis, including the small sample size and heterogeneity across treatment groups, prevent firm conclusions from being drawn.
Conclusions
The clinical data supporting the efficacy of entrectinib in a histology-agnostic population of adult patients with NTRK fusion–positive cancer are derived from a pooled analysis of 3 open-label, single-arm trials, including 2 phase I trials (ALKA and STARTRK-1) and a phase II basket trial (STARTRK-2). In total, 121 and 193 adults with NTRK-positive cancer of different histologies were included in the most recent pooled analyses of efficacy and safety, respectively. Results showed that treatment with entrectinib treatment was associated with an ORR of 61.2% (95% CI, 51.87 to 69.88), with 15.7% and 45.5% of patients demonstrating CR or PR, respectively. The median time to response was 1 month (95% CI, 0.9 to 1.0), and the median DOR was 20 months (95% CI, 13.0 to 38.2). Across different tumour types, the ORR varied widely, with a similarly wide range of uncertainty. Combined with the differences in sample sizes across the different tumour types, the majority of which had been experienced by fewer than fewer than 10 patients, these factors limit the generalizability of the findings of the mixed cancer population. Among patients with CNS metastases at baseline (N = 19), the sponsor conducted analyses examining the intracranial efficacy of entrectinib with respect to CNS lesions. The IC-ORR was 52.6% (95% CI, 28.9 to 75.6) with a median IC-DOR of 17.2 months (95% CI, 7.4 to NE) and a median IC-PFS of 10.1 months (95% CI, 6.3 to 26.7).
While the rarity of NTRK fusion creates practical challenges in terms of conducting a randomized controlled trial, the methodological limitations of single-arm trials with small sample sizes mean the results should be interpreted based on clinical judgment. The clinical experts consulted by CADTH indicated that the response rates reported for entrectinib are clinically meaningful, particularly for those with CNS metastases at baseline, given that these patients typically have poor prognoses. The comparative evidence included in this review for entrectinib versus larotrectinib or standard therapies has important methodological limitations and is insufficient for drawing conclusions on comparative efficacy.
The clinical experts consulted by CADTH noted that patients would likely be screened and monitored for risk factors in accordance with the recommendations in the product monograph (e.g., risk of congestive heart failure) and that AEs would likely be managed in accordance with the dosage interruption and reduction scenarios reported in the product monograph. Overall, the clinical experts noted that the AE profile of entrectinib was acceptable and that entrectinib may be more tolerable than some alternatives (e.g., chemotherapy or radiation) for patients who have advanced diseases. Patient groups also noted a preference for targeted therapy and a desire to avoid systemic therapies that may be associated with greater toxicity. From a regulatory perspective, the sponsor is required to provide additional integrated safety analyses to Health Canada as part of the conditional market authorization (i.e., NOC/c); this will include further characterizing the off-target pharmacodynamics of entrectinib (given that it is not a selective inhibitor of TRK proteins), the cardiac risks, and the risks of fractures to identify risk factors and support labelling instructions for dose modification and monitoring.
Introduction
Disease Background
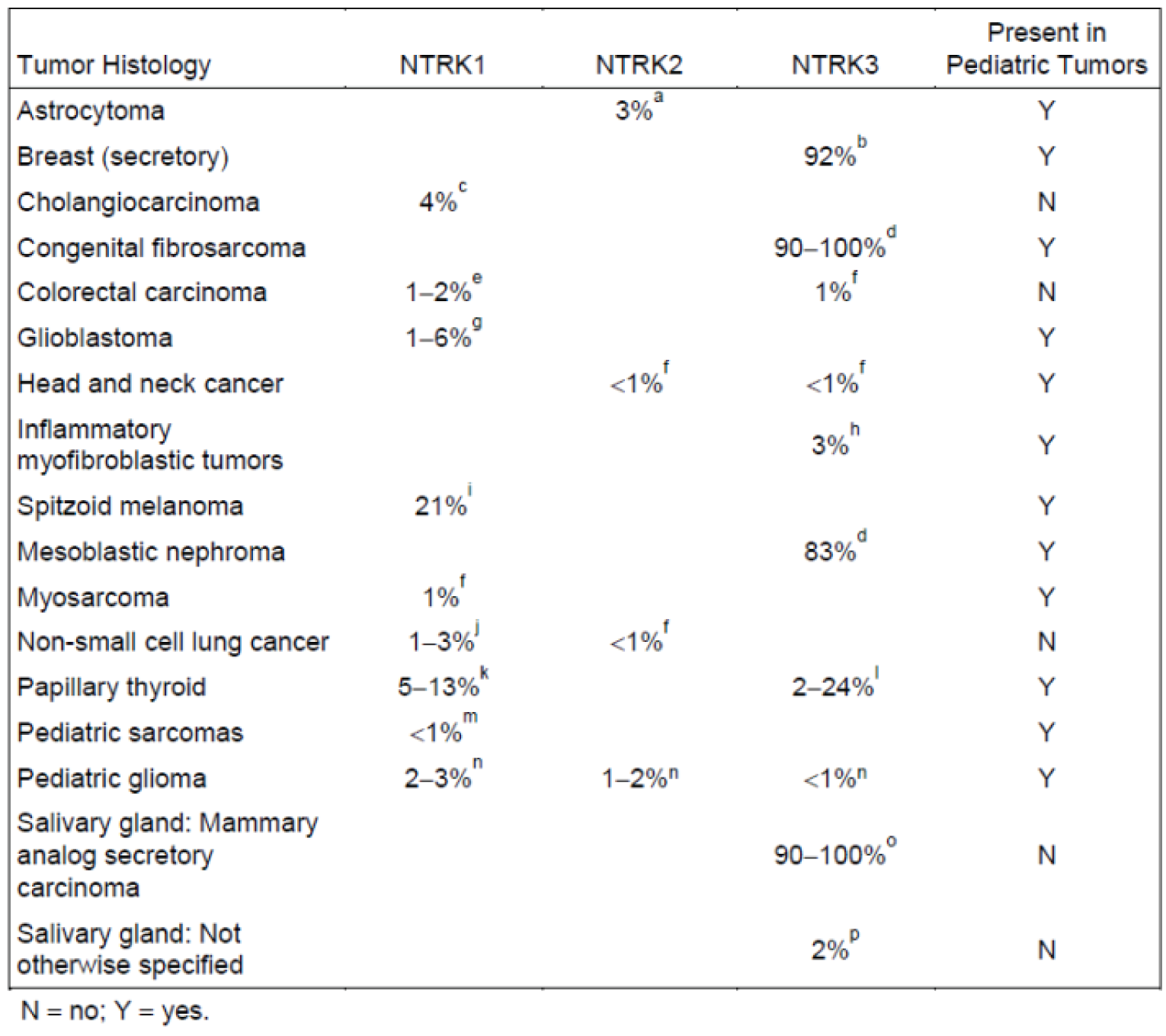
The NTRK genes encode the neurotrophin family of receptors. A recent study estimated the prevalence of NTRK gene fusion at 0.28% of all solid cancers. NTRK oncogenic fusions arise from exact intrachromosomal or interchromosomal rearrangements that juxtapose the kinase domain-containing 30 region of NTRK with the 50 region of NTRK gene partners. Preclinical data have demonstrated that chimeric oncogenic fusions may lead to the partial or complete deletion of the immunoglobulin-like domain of TRK, which has an inhibitory influence on downstream signalling pathways in the absence of activating ligands.2 Available literature demonstrates that NTRK gene fusions are oncogenic drivers in various cancers12 (refer to Appendix 3 for details).12-15 Although reported to be prevalent in 0.28% of all solid cancers,16 NTRK oncogenic fusions are observed in variable frequencies across a spectrum of pediatric and adult cancers, with some uncertainty regarding exact frequencies (Figure 1).1 Different studies have reported varying frequencies. The variation in frequency may be explained by the number of patients screened and the NTRK fusion detection techniques used.
Lung, colorectal, and breast cancer are the 3 most commonly diagnosed cancers in Canada (Table 4):
In NSCLC, NTRK fusions (occurring in approximately 0.1% to 1% of cases)4-6 are less common than other oncogenic gene rearrangements that involve the ALK, ROS1, and RET proto-oncogene and occur at frequencies of approximately 4% to 6%, 1% to 2%, and 1% to 2%, respectively.17-19
The NTRK mutation is also quite rare in breast cancer except in the rare subtype of secretory breast cancer, in which the prevalence of NTRK fusion has been reported to be 92%.
NTRK gene fusions are also rare in sporadic CRCs (occurring in 2% to 3% of cases).4 These appear to be common in colorectal tumours with MSI-H and exclusive of RAS and BRAF mutations (which represent about 55% of mCRC cases).8
The NTRK mutation is uncommon in adult sarcomas (1%); it is more frequently found in gastrointestinal stromal tumours (GISTs),20 particularly wild-type GISTs (lacking mutations in KIT proto-oncogene [KIT] and PDGFRA).
NTRK gene fusions are observed in 6% of adults with primary thyroid cancers.9
Although the frequency of NTRK fusions is low in common cancer types, NTRK3 fusions are nearly ubiquitous among rare cancer types, such as MASC and IFS.4,10 In pediatric oncology, NTRK fusions are pathognomonic in specific, rare cancers, including IFS (91% to 100%)21 and cellular congenital mesoblastic nephroma (CMN) (83%).4 NTRK fusions are also commonly observed in several other very rare pediatric cancers, including secretory breast cancer (92%)22 and MASC of the salivary gland (100%).23 In addition, there are significant numbers of NTRK fusion cancers among children with papillary thyroid carcinoma (9.4% to 25.9%),24,25 undifferentiated sarcomas (1%; frequency in adult versus pediatric not specified),12 high-grade gliomas (7.1%),4 inflammatory myofibroblastic tumours, and acute leukemia (rarely).26
Table 4: Incidence and Mortality Associated With Solid Tumours Among People in Canada in 2019
Affected organ | Projected incidence | Projected mortalitya | 5-year net survival (%) | ||||
|---|---|---|---|---|---|---|---|
Cases | ASIRb | Males | Females | ||||
Deaths | ASMRb | Deathsa | ASMRb | ||||
Lung and bronchus | 29,300 | 62.1 | 10,900 | 54.7 | 10,100 | 43.1 | 19 |
Breast | 27,200 | 66.8 | 55 | 0.3 | 5,000 | 22.4 | 88 |
Colorectal | 26,300 | 60.6 | 5,200 | 26.8 | 4,400 | 18.2 | 65 |
Prostate | 22,900 | 118.1 | 4,100 | 22.2 | — | — | 93 |
Bladder | 11,800 | 25.0 | 1,800 | 9.7 | 700 | 2.8 | 75 |
Thyroid | 8,200 | 21.8 | 100 | 0.5 | 130 | 0.5 | 98 |
Melanoma | 7,800 | 21.7 | 840 | 4.4 | 450 | 2.0 | 88 |
Uterus (body, NOS) | 7,200 | 34.5 | — | — | 1,250 | 5.3 | 83 |
Kidney and renal pelvis | 7,200 | 17.0 | 1,250 | 6.4 | 670 | 2.8 | 71 |
Pancreas | 5,800 | 12.9 | 2,700 | 13.5 | 2,500 | 10.7 | 8 |
Oral | 5,300 | 12.7 | 1,050 | 5.3 | 430 | 1.8 | 64 |
Stomach | 4,100 | 9.3 | 1,200 | 6.2 | 760 | 3.3 | 28 |
Brain or CNS | 3,000 | 7.1 | 1,400 | 7.1 | 1,050 | 4.7 | 23 |
Ovary | 3,000 | 14.2 | — | — | 1,900 | 8.4 | 45 |
Liver | 3,000 | 6.7 | 1,100 | 5.4 | 280 | 1.2 | 19 |
Esophagus | 2,300 | 5.6 | 1,700 | 8.6 | 500 | 2.1 | 15 |
Cervix | 1,350 | 7.2 | — | — | 410 | 2.0 | 72 |
Larynx | 1,150 | 2.4 | 330 | 1.7 | 75 | 0.3 | 62 |
Testis | 1,150 | 6.4 | 35 | 0.2 | — | — | 97 |
ASIR = age-standardized incidence rate; ASMR = age-standardized mortality rate; CNS = central nervous system; NOS = not otherwise specified.
aCanada totals include provincial and territorial estimates.
bRates are age-standardized to the 2011 Canadian population and are per 100,000.
Source: Canadian Cancer Statistics Advisory Committee (2019).3
Figure 1: Incidence of NTRK Gene Fusions Across Multiple Solid Tumour Histologies
NTRK = neurotrophic tyrosine receptor kinase.
Source: Committee for Medicinal Products for Human Use assessment report.27
Standards of Therapy
There is currently no reimbursed drug that targets the NTRK pathway. Among adult cancers, defining accepted clinical practice is difficult because NTRK gene fusions can be observed in a multitude of solid cancers. Patients with advanced solid tumours are largely treated with standard of care (i.e., chemotherapy, immunotherapy, and/or targeted therapy), as defined by their primary disease site.28 Ultimately, many of these cancers have a poor prognosis, and patients who progress on upfront therapies will have limited subsequent therapeutic options.
NTRK-Targeted Therapy
Larotrectinib (Vitrakvi) is approved for use in Canada to treat adult and pediatric patients with locally advanced or metastatic solid tumours who have an NTRK gene fusion without a known acquired resistance mutation or in cases where surgical resection is likely to result in severe morbidity and patients have no satisfactory treatment options. Following a resubmission, CADTH issued a recommendation that larotrectinib be reimbursed with conditions. At the time of this review of entrectinib, larotrectinib is currently listed as under consideration for negotiation by the pan-Canadian Pharmaceutical Alliance.
Selected Disease Site–Specific Burden and Need Considerations
Secretory Breast Cancer
NTRK gene fusions are quite rare in breast cancer.22,26 Currently, there a number of standard therapy options for patients with advanced breast cancer that have improved survival considerably,4 but many patients will ultimately go on to exhaust available therapies and be left with no suitable therapeutic options.28 Secretory breast carcinoma is a very rare histologic subtype of breast cancer that is seen in fewer than 1% of patients with invasive breast cancer; this subtype is seen in children and adults and is associated with a generally favourable prognosis and a low likelihood of metastases.22,26 However, for those patients with advanced, inoperable disease, treatments options are limited. Secretory breast carcinomas are also associated with a prevalence of NTRK gene fusions that is greater than 90%.4
Sarcoma and GIST
Sarcomas are a relatively rare tumour subtype representing more than 100 hundred subtypes. These are often categorized into soft tissue sarcomas (STSs) and bony sarcomas. STSs are associated with a less favourable prognosis, and in the adult population, these have a high risk of recurrence. Metastatic or unresectable STS is generally not curable. Limited effective cytotoxic therapies exist for STS, especially in the metastatic setting or upon relapse.29
In adults with sarcoma and NTRK fusions, standard therapies include radiation and surgery as well as cytotoxic therapy (i.e., doxorubicin) for those with advanced disease. However, in the advanced setting, traditional chemotherapy has limited effectiveness.28 The clinical exerts consulted for this review stated that there are examples of pediatric patients who were fusion-positive with locally advanced disease having enough response to the drug to facilitate curative surgical resection.
NTRK fusions are also seen in 3% to 4% of GIST tumours.20 For GIST tumours with cKIT and PDGFRA mutations, targeted therapies represent the current standard of care. For 10% to 15% of GIST tumours that are classified as wild type with no KIT or PDFGFRA mutations, there is a significant unmet need for effective therapies.30
Thyroid Cancer
For patients with advanced, inoperable thyroid cancer that has progressed on radioactive iodine therapy, current treatments include small-molecule tyrosine kinase inhibitors.28 NTRK gene fusions may be identified in 6% of thyroid cancers.9
Gastrointestinal Cancers
For patients with advanced CRC, there is an unmet need for better therapies in patients with chemorefractory disease (i.e., have progressed on 2 or more prior lines of therapy). NTRK gene fusions are uncommon in CRC.7,8 The clinical experts consulted by the review team noted that for patients with non-colorectal GI cancers, particularly pancreatic cancer and cholangiocarcinoma, there is a significant unmet need for better therapies.
Lung Cancer
Lung cancer remains the most common cancer in Canada.31 NTRK fusions are estimated in up to 1% of patients with NSCLC4 (compared to ALK fusions in 3% to 5%, ROS1 fusions in 1% to 2%, and epidermal growth factor receptor [EGFR] mutations in 20%).5 Systemic treatment options for advanced NSCLC include chemotherapy, immunotherapy, and combination and biomarker-directed targeted therapies, with response rates ranging from 45% to 60% in those without ALK, EGFR, ROS1, or BRAF-deranged lung cancer. While current therapies have improved outcomes for patients with NSCLC, patients will ultimately become refractory and/or intolerant; hence, there is a need for effective and tolerable therapies in pre-treated patients.31
Drug
Mechanism of Action
Entrectinib is an inhibitor of TRKA, TRKB, and TRKC (encoded by the NTRK1, NTRK2, and NTRK3 genes, respectively), ROS1 (encoded by the ROS1 gene), and ALK (encoded by the ALK gene). Fusion proteins that include TRK, ROS1, or ALK kinase domains drive tumorigenic potential by hyperactivating downstream signalling pathways, leading to unconstrained cell proliferation. Entrectinib potently inhibits TRK, ROS1, and ALK, leading to the inhibition of downstream signalling pathways and cell proliferation and the induction of tumour cell apoptosis.
Indication Under Review
Entrectinib is indicated for the treatment of adult patients who have unresectable, locally advanced or metastatic extracranial solid tumours (including brain metastases) with NTRK gene fusion without a known acquired resistance mutation and no satisfactory treatment options. The sponsor’s reimbursement request is per the indication. The product monograph states that a validated assay is required for the selection of patients with NTRK fusion–positive unresectable, locally advanced or metastatic extracranial solid tumours, including brain metastases. NTRK fusion–positive status should be established before initiation of entrectinib therapy.
Entrectinib received a NOC/c for this indication on February 10, 2020, pending the results of new information to verify its clinical benefit. Table 5 provides a summary of the status of the confirmatory studies required to address the conditions of the NOC/c.
Table 5: Update on Confirmatory Studies for Entrectinib NOC/c
Qualifying notice | Status update |
|---|---|
Confirmatory studies | |
Submit the final report as an SNDS-C, from the first 54 patients with NTRK fusion solid tumours enrolled across the ALKA, STARTRK-1, and STARTRK-2 studies, to verify and describe the clinical benefit and further characterize the duration of response in patients who achieved a CR or PR to entrectinib. All responding patients will be followed for ≥ 2 years from the onset of response or until disease progression, whichever comes first. DOR will be assessed by ICR. | Closed. Submitted to Health Canada on September 24, 2021. |
Submit the final report as an SNDS-C from ongoing and proposed trials conducted to verify and describe the clinical benefit of entrectinib, based on more precise estimation of the ORR and mature response duration per IRC assessment, in adult patients who have unresectable, locally advanced or metastatic extracranial solid tumours (including brain metastases) with NTRK gene fusion without a known acquired resistance mutation and no satisfactory treatment options. A sufficient number of patients will be evaluated to characterize response and durability of response more precisely for each of the following tumour types: colorectal cancers, gynecological cancers, and melanoma. A minimum of 40 patients with cancers other than pediatric solid tumours, colorectal cancers, central nervous system cancers, gynecological cancers, melanoma, soft tissue sarcoma, NSCLC, MASC, and secretory breast cancer will also be studied. ORR and DOR will be assessed by IRC and all responding patients will be followed for ≥ 12 months from the onset of response. | Ongoing. Due June 2027. |
Other studies | |
Determine the functional activation or inhibition of off-target receptors, transporters, and/or channels that, at concentrations of 10 µm, showed greater than 50% inhibition by entrectinib or M5 in the secondary pharmacology studies submitted in the NDS. As part of an integral safety assessment, include EC50 or IC50 data for target receptors, transporters, and channels that are still significantly affected at a concentration less than 1 µm, particularly those involved in suicidal intent and behaviour, as described in Muller et al. (2015). | Closed. Submitted to Health Canada on September 25, 2020. |
Submit integrated safety analyses and supporting data from an adequate number of patients enrolled in clinical trial(s) designed to characterize the cardiac risks and its sequelae in patients exposed to entrectinib with reasonable precision; identify risk factors for development of these sequelae; and support labelling instructions for dose modification and monitoring. The design of the trial should include sufficient cardiac monitoring to achieve these objectives. | Ongoing. Final report targeted for submission in Q2 2022. |
Submit integrated safety analyses and supporting data from an adequate number of patients enrolled in clinical trial(s) designed to characterize the risk of fractures and its sequelae in patients exposed to entrectinib with reasonable precision; identify risk factors for the development of these sequelae; and support labelling recommendations to mitigate the risk of skeletal fractures. The design of the trial should include sufficient bone monitoring to achieve these objectives, including but not limited to initial and serial assessment of BMD with DXA scans and markers of bone formation, bone resorption, and calcium metabolism. | Ongoing. Final report targeted for submission in Q2 2025. |
BMD = bone mineral density; CR = complete response; DOR = duration of response; DXA = dual X-ray absorptiometry; EC50 = concentration of the drug that provides half the maximal response; IC50 = half-maximal inhibitory concentration; ICR = independent central review; M5 = major active metabolite of entrectinib; MASC = mammary analogue secretory carcinoma; NDS = new drug submission; NOC/c = Notice of Compliance with conditions; NSCLC = non–small cell lung carcinoma; NTRK = neurotrophic tyrosine receptor kinase; ORR = objective response rate; PR = partial response; Q2 = second quarter; SNDS-C = supplement to a new drug submission – confirmatory.
Source: Sponsor-provided additional information.32
Recommended Dosage
The recommended dose of entrectinib is 600 mg orally once daily. It is recommended in the product monograph that patients are treated until disease progression or unacceptable toxicity. The product monograph provides recommendations for the management of AEs that require temporary interruption, dose reduction, or discontinuation of treatment. From the starting dose of 600 mg once daily, the dose can be reduced twice: first to 400 mg once daily and then to 200 mg once daily. Treatment with entrectinib should be permanently discontinued if patients are unable to tolerate a dose of 200 mg once daily. The product monograph recommends that the use of concomitant strong or moderate cytochrome P450 3A (CYP3A) inhibitors and entrectinib should be avoided or limited to 14 days or less. If concomitant use of strong or moderate CYP3A inhibitors cannot be avoided, the dose of entrectinib should be reduced to 100 mg once daily with strong CYP3A inhibitors or to 200 mg once daily with moderate CYP3A inhibitors.
Health Canada has not authorized an indication for entrectinib for pediatric use, and there are no recommendations in the product monograph regarding dosing in pediatrics.
Table 6: Key Characteristics of Entrectinib and Larotrectinib
Characteristic | Entrectinib | Larotrectinib |
|---|---|---|
Mechanism of action | Inhibitor of TRKA, TRKB, and TRKC (encoded by the genes NTRK1, NTRK2, and NTRK3, respectively), ROS1 (encoded by the ROS1 gene), and ALK (encoded by the ALK gene). | Selective TRK inhibitor. Larotrectinib targets the TRK family of proteins (inclusive of TRKA, TRKB, and TRKC) that are encoded by NTRK1, NTRK2, and NTRK3 genes, respectively. |
Indicationa | For the treatment of adult patients who have unresectable, locally advanced or metastatic extracranial solid tumours (including brain metastases) with NTRK gene fusion without a known acquired resistance mutation and no satisfactory treatment options. | For the treatment of adult and pediatric patients who have solid tumours with NTRK gene fusion without a known acquired resistance mutation; are metastatic or in whom surgical resection is likely to result in severe morbidity; and who have no satisfactory treatment options. |
Route of administration | Oral | Oral |
Dosage forms and strengths | 100 mg and 200 mg capsules |
|
Recommended dose | 600 mg once daily |
|
Serious adverse effects or safety issues | The product monograph includes black box warnings that entrectinib may cause congestive heart failure and, when administered to a pregnant person, fetal harm. | Notable harms include fatigue, nausea, dizziness, vomiting, anemia, ALT increase, AST increase, cough, constipation, diarrhea, sepsis, pyrexia, neutrophil count decrease, lymphocyte count decrease, hypokalemia, hyponatremia, hypoglycemia, hypophosphatemia, gait disturbance, paresthesia, myalgia, and/or weight increase. |
Other | Approved as NOC/c pending the results of trials to verify its clinical benefit. |
|
ALK = anaplastic lymphoma kinase; ALT = alanine aminotransferase; AST = aspartate aminotransferase; NOC/c = Notice of Compliance with condition; NTRK = neurotrophic tyrosine receptor kinase; ROS1 = ROS proto-oncogene 1; TRK = tropomyosin receptor kinase.
aHealth Canada–approved indications.
Source: Product monographs.34,35
Stakeholder Perspectives
Patient Group Input
This section was prepared by CADTH staff based on the input provided by patient groups.
A total of 4 patient groups submitted input. The LHF is a charity that provides education (on tobacco cessation and prevention of respiratory illness), programs, and services for patients and health care providers. It also invests in research and policy improvement for lung health. LCC, a national charity and member of the Global Lung Cancer Coalition, serves as a resource for lung cancer education, patient support, research, and advocacy. CCC is a patient association dedicated to raising awareness of colon cancer prevention and treatment. It provides education, support, and advocacy for patients with colon cancer and their families to improve their quality of life. The CBCN, a member of Canadian Cancer Action Network, is a national charity engaged in education and advocacy to ensure the best quality of care for all people in Canada affected by breast cancer.
Lung cancer: Two organizations submitted survey responses. The OLA/LHF collected responses from 14 patients with lung cancer (1 of whom had experience with entrectinib) and caregivers through an online survey on or before December 18, 2021, as well as from 3 patients with lung cancer through phone interviews from September 2021, to October 2021. In addition, a registered nurse and a certified respiratory educator participated in the OLA/LHF survey. Another organization, LCC, conducted interviews with patients from January to February 2022 to collect their thoughts and experiences with lung cancer. (The total number of patients is unspecified; 8 patients had experiences with entrectinib and 1 patient has NTRK fusion mutation.)
According to the OLA/LHF survey results, patients found symptoms of lung cancer challenging, including shortness of breath (64%), fatigue (57%), cough (21%), chest tightness (14%), pain, and the psychosocial effects of receiving a poor diagnosis, such as depression (25%), worry about metastasis, concern about maintaining relationships with families and friends, feelings of isolation, and withdrawal from social activities due to stigma. These effects had great impacts on patients’ activities of daily living (60%), work (38%), leisure activities, and hobbies (28%) as well as on their emotional well-being (20%). Family members and caregivers also experienced psychosocial burden, fatigue, and emotional exhaustion due to caring for patients and the resulting impacts on their ability to work as well as their relationships, emotional well-being, and independence to travel and socialize.
The LCC survey results showed that patients with lung cancer want to better manage their symptoms, minimize side effects from treatments, improve quality of life, live longer while maintaining independence and functionality, minimize burden on their caregivers and loved ones, delay disease progression (i.e., achieve long-term remission), and prolong survival.
In addition, OLA/LHF survey respondents expressed the need for a therapy to treat brain metastases. Both OLA/LHF and LCC surveys highlighted the importance of biomarker testing, access to which is not routinely available across the country during diagnosis. However, patients acknowledged that new funding was announced in 2021 to expand next-generation sequencing (NGS) tests in many provinces. A number of patients from both surveys said they wished they had been screened for a biomarker sooner.
CRC: CCC conducted an online survey that was disseminated by the Canadian Cancer Society across pan-tumour forums and panels from December 7, 2021, to February 8, 2022. A total of 6 patients from Canada, the US, and Indonesia who have NTRK fusion mutation responded (2 patients with thyroid cancer, 2 patients with sarcoma, 1 patient with neuroendocrine tumours, and 1 patient with CRC at primary sites). Two out of 6 patients had experience with entrectinib.
Three out of 6 patients experienced symptoms from their cancers, such as pain, fatigue, cough, shortness of breath, and mobility issues, and 5 patients reported that the symptoms affected their daily lives. Some patients felt that the symptoms affected their work, daily activities, and ability to exercise. Three out of 6 patients also expressed psychological impacts, such as depression, stress, and fear of dying early. Five out 6 patients said that new therapy should improve their physical condition and quality of life. Five out of 6 patients also said they would take therapy to improve their quality of life even if it did not extend their OS. However, 2 patients said they would tolerate significant side effects if therapy could extend their survival by 2 months to 12 months. Patients expressed the need to carry on social activities without the burden of side effects from treatments. Lastly, patients emphasized that access to therapy and the option to be able to decide (along with physicians) which drug to take are important.
In terms of biomarker testing, 5 out of 6 patients had their testing done after diagnosis. One patient said she wished she had had the testing done earlier so she could have avoided unnecessary harmful treatments. Patients who responded to the CCC survey highlighted the importance of equitable access to biomarker testing so they can benefit from targeted therapy for their tumour profile.
Breast cancer: CBCN collated survey results from 2012 Lived Experience of Metastatic Breast Cancer Patients and Caregivers Survey Report and the 2017 Lived Experience Breast Cancer Patient Survey, both of which were conducted online, as well as a review of current studies and grey literature. A total of 146 patients in Canada with breast cancer (most of whom had metastatic breast cancer) participated in the surveys. None had experience with entrectinib.
In a 2012 survey, patients reported that fatigue (54%), insomnia (39%), and pain (37%) had a significant or debilitating impact. The 2017 survey showed similar results. In the 2012 survey, patients reported that cancer had a significant impact on social aspects of their lives, such as their ability to work (71%), caregiving responsibilities (21%), exercise (49%), hobbies and personal interests (42%), participation in social events and activities (41%), and spending time with loved ones (22%). Other challenges reported by the 2012 survey respondents included guilt, feeling like a burden to caregivers, fear of death, poor body image, not knowing what will happen to their children, and marital stress. Patients cited extended PFS, delayed progression, relief from cancer-related symptoms, improved quality of life, and minimal side effects from treatment as desirable outcomes. Also, patients wish to reduce the impact of cancer on their ability to care for children and dependents, continue working, spend time with loved ones, participate in social activities, travel, maintain friendships, and pursue personal interests. Lastly, patients said NTRK (fusion) gene testing is essential for optimal health outcomes and should be readily available and reimbursed appropriately. This testing is not currently part of routine breast cancer care.
Clinician Input
Input From Clinical Experts Consulted by CADTH
All CADTH review teams include at least 1 clinical specialist with expertise regarding the diagnosis and management of the condition for which the drug is indicated. Clinical experts are a critical part of the review team and are involved in all phases of the review process (e.g., providing guidance on the development of the review protocol; assisting in the critical appraisal of clinical evidence; interpreting the clinical relevance of the results; and providing guidance on the potential place in therapy). In addition, as part of the entrectinib review, a panel of 3 clinical experts from across Canada was convened to characterize unmet therapeutic needs, assist in identifying and communicating situations where there were gaps in the evidence that could be addressed through the collection of additional data, promote the early identification of potential implementation challenges, gain further insight into the clinical management of patients living with a condition, and explore the potential place in therapy of the drug (e.g., potential reimbursement conditions). A summary of this panel discussion is presented in this section.
Unmet Needs
Similar to the input that was received during the CADTH review of larotrectinib, the clinicians felt that it was difficult to fully characterize the unmet need for patients who could be eligible for treatment with entrectinib. This is due to the breadth of potentially advanced solid tumours that may harbour NTRK fusion mutations and to variability in the availability and effectiveness of potential alternative therapies. However, they agreed that, in the case of metastatic solid malignancies, virtually all patients eventually progress on currently available therapies, with the possible exception of select patients receiving immunotherapies in select cancer types.
Overall, the clinicians felt that an ideal treatment would prolong survival, which was noted as the most important goal for adult patients with advanced, incurable disease. Additional important treatment goals included minimizing toxicity, decreasing cancer-related symptoms (i.e., pain and shortness of breath), maintaining or improving quality of life, delaying disease progression, improving PS, prolonging life, maintaining independence, and reducing burdens on caregivers.
Place in Therapy
The clinicians noted that the appropriateness of recommending that patients try other treatments before initiating treatment with entrectinib would depend on the cancer subtype and the efficacy of front-line therapy. However, it was agreed that entrectinib should be considered early in the course of NTRK fusion cancer treatment. This was based on the rationale that the NTRK fusion is the oncogenic driver in these tumours. Treatments targeting the tumour site as opposed to the TRK were perceived as likely to be less effective and having the potential to be more toxic than TRK-targeting therapies (i.e., entrectinib or larotrectinib), particularly for tumours where the alternative is chemotherapy. In addition, it was noted that therapies that do not target the TRK would be a poor use of patients’ time because patients endure side effects of treatments that are likely to be less efficacious than TRK-targeting treatments.
All clinicians noted that there is considerable variability in access to NTRK fusion testing across tumour sites and Canadian jurisdictions. Whether or not tumours are routinely tested for NTRK fusion, and the timing of such testing, would influence when targeted therapies such as entrectinib are identified as potential treatment options.
Patient Population
The clinicians indicated that entrectinib should be considered in adult patients with a good ECOG PS and advanced solid tumours that harbour an NTRK fusion. Entrectinib has not been approved by Health Canada for use in pediatric patients or patients with a primary CNS tumour.
The indication approved by Health Canada states that entrectinib should be limited to patients who have “no satisfactory treatment options.” The clinical experts consulted by CADTH noted that the definition of “no satisfactory treatment options” would depend on the tumour sites and reflect the range of alternative therapies available for those tumours (e.g., some have no alternatives, while others may have several). The clinical experts agreed that “no satisfactory treatment options” would be interpreted by clinicians to mean suboptimal treatment for the patient with respect to achieving treatment goals (e.g., improving survival and disease-free interval) or be associated with poor quality of life and/or significant toxicity.
The indication approved by Health Canada states that entrectinib should be limited to patients with unresectable or metastatic disease. The clinical experts noted that patients who have metastatic disease that is amenable to surgery (e.g., a patient with NTRK fusion–positive sarcoma who experiences a single lung metastasis at recurrence that is removed surgically with no other evidence of disease) should not be eligible for treatment with entrectinib. Rather, such patients would be eligible only at time of subsequent metastasis that is unresectable or if they develop multiple metastases that are not amenable to local control with stereotactic ablative radiotherapy or surgical resection.
Although other systemic therapies may be available, treatments like entrectinib are selected for patients based on molecular findings to target the oncogene that is driving their cancer. These treatments are more attractive and potentially more beneficial in the context of personalized or precision medicine. Unless the potential alternative treatment is known to be safe and highly effective, entrectinib would ideally be considered before exhausting other systemic therapy options because:
Compared to alternatives, entrectinib may be associated with higher response rates, have a better safety profile, and be more tolerable.
Given entrectinib’s mechanism of action and the available evidence for it, the clinical experts believed that it would be efficacious in patients with NTRK gene fusions and advanced disease regardless of the number of prior therapies.
Patients may no longer be fit for any systemic therapy after receiving alternative treatments (e.g., they may have poor ECOG PS).
The presence of NTRK fusion mutations is clinically actionable, and the Canadian consensus guidelines recommend the use of TRK inhibitors as the preferred option for patients with NTRK fusion tumours.
The clinicians noted that entrectinib or other similar drugs targeting an oncogene driving the cancer may change the existing treatment paradigm as precision medicine approaches are adopted in clinical practice.
When asked how to identify the patients best suited for treatment with entrectinib, the clinicians noted that NTRK fusions are relatively rare in common cancers; therefore, a diagnostic testing algorithm would be required. Given the rarity, tumour NTRK assessment in enriched populations may also be required. Specifically, routine clinical testing (i.e., fluorescence in situ hybridization [FISH]) establishes NTRK fusion status in classic histologies (e.g., IFS and CMN). Fusion panels in NGS are routinely used for other STS cases at diagnosis and include NTRK coverage. In patients with glioma who lack BRAF (V600E) or BRAF fusion, H3K27M mutations will require NGS testing that includes NTRK1, NTRK2, and NTRK3 gene fusions. Patients with differentiated thyroid cancer that is either unresectable or progressive and/or symptomatic after surgery and refractory to radioactive iodine therapy will receive NGS screening. Reflex testing should also be considered. The clinicians noted that patients with poor ECOG PS, who are unable to tolerate oral therapies, who lack NTRK gene fusions, and who have resectable disease would be least suitable for treatment with entrectinib. In adult patients, the clinicians were not aware of any additional predictive biomarkers of efficacy for entrectinib beyond the presence of NTRK gene fusion.
The clinicians also noted that a potential acquired resistance can be identified on a new biopsy or circulating tumour DNA analysis. It should not affect the testing strategy upfront because it happens after treatment initiation and affects subsequent therapy.
Assessing Response to Treatment
In terms of outcomes that are used to determine whether a patient is responding to treatment in clinical practice, the clinicians indicated that typical metrics of treatment efficacy include disease evaluation by cross-sectional imaging modalities (MRI, CT, PET/CT) to assess response by RECIST 1.1 (for solid tumours) or Response Assessment in Neuro-Oncology (for CNS tumours), symptom improvement, treatment tolerability, and TTP. In addition, for patients with differentiated thyroid cancer, thyroglobulin levels are monitored.
The clinicians noted that in adults, objective response, non-progression, patient-reported improvements in ability to perform activities, improved survival, stabilization, improvement or reduced severity of symptoms, and improvement or no deterioration in quality of life would all be considered clinically meaningful outcomes. Clinicians noted that treatment response is typically assessed every 3 months and that once response is established or remission is achieved, this interval may be prolonged.
Discontinuing Treatment
The clinicians noted that treatment failure would be determined by disease progression, treatment intolerability, poor quality of life (e.g., poor ECOG PS), or patient request to discontinue treatment.
Prescribing Conditions
The clinicians reported that all treatment settings are appropriate for entrectinib administration in adults, given that it is an oral therapy. The clinicians also noted the requirement for the NTRK fusion status of the tumour to be documented. This can be done through FISH (which is routine in IFS or CMN) or NGS panels (for non-IFS, STS, glioma, and differentiated thyroid tumours). Immunohistochemistry can be used for screening in non-CNS histologies, but NTRK fusion must be confirmed using NGS or FISH.
Entrectinib has only be studied as monotherapy and should not be given in combination with other systemic anti-cancer therapies. There is insufficient evidence to evaluate the efficacy of sequential usage of entrectinib or larotrectinib for disease progression on 1 of the 2 drugs. The clinical experts noted that a patient who experiences intolerance to 1 of the TRK inhibitors could be given a trial with the other to determine if tolerability improves.
Clinician Group Input
This section was prepared by CADTH staff based on the input provided by clinician groups.
The following clinician groups have provided input:
OH-CCO Lung Cancer DAC: 5 clinician members held joint discussions through meetings and email.
LCC: 22 clinicians gathered information from publicly available sources, published manuscripts, conference presentations, and members’ experiences.
OH-CCO GI Cancer DAC: 3 clinicians held joint discussions at a committee meeting.
OH-CCO Breast Cancer DAC: 2 clinician members held joint discussions at a committee meeting.
OH-CCO HNT DAC: 2 clinician members held joint discussions through email.
The OH-CCO DACs provide clinical and health system guidance on drug-related issues in support of CCO’s mandate, including the Provincial Drug Reimbursement Programs and the Systemic Treatment Program. LCC is a national charity that increases awareness of lung cancer, supports and educates patients with lung cancer and their families, and engages in research and advocacy for patient access to best care. The LCC Medical Advisory Committee is made up of clinicians and key opinion leaders across Canada.
Unmet Needs
Lung cancer: The OH-CCO Lung Cancer DAC and LCC have identified improved survival (efficacy) and ease of administration or improved compliance as goals of therapy. In addition, OH-CCO Lung Cancer stated that improved quality of life and delayed time to chemotherapy are treatment goals; LCC mentioned improved response and tolerability as treatment goals. Both groups said that patients who are refractory to chemotherapy and/or immunotherapy, such as those with NTRK fusion–positive mutation without targeted therapy options, are a subset of patients with NSCLC who have unmet needs.
GI cancer: According to the OH-CCO GI Cancer DAC, treatment goals include prolonging life, delaying disease progression, improving quality of life, and decreasing cancer symptoms. This group stated that there is an unmet need for patients with all subtypes of GI cancer who are considered palliative.
Breast cancer: The OH-CCO Breast Cancer DAC identified improved OS with acceptable toxicity as the treatment goal. This group cited a prevalence rate of more than 90% for NTRK fusion mutation in a subset of patients with breast cancer (i.e., secretory breast cancer, which is the current unmet need) as well as any other patients with NTRK fusion–positive breast cancer.
HNT cancer: The OH-CCO HNT DAC listed cancer shrinkage, improvement in the severity of cancer-related symptoms, ability to maintain and/or improve quality of life, and delayed disease progression as treatment goals. The group stated that the unmet needs are in patients with salivary gland tumours (for which chemotherapy associated with a poor response rate and significant toxicity is available) and thyroid cancers (for which multi-tyrosine kinase inhibitors with toxicity and short DORs are available).
Place in Therapy
Lung cancer: The OH-CCO Lung Cancer DAC stated that entrectinib would displace first-line treatments (i.e., chemotherapy and immunotherapy combinations or pembrolizumab as a single drug) to second-line treatment, and second-line treatment (i.e., docetaxel) to third-line treatment. In contrast, LCC said that entrectinib would most likely be tried after chemotherapy and/or immunotherapy as the first-line option(s), and as the first-line option when these therapies are contraindicated (e.g., due to comorbidities or poor ECOG PS). LCC also added that while it is reasonable to try chemotherapy and/or immunotherapy before entrectinib, it should not be mandated. Lastly, LCC mentioned that entrectinib would be an additional option or an alternative to larotrectinib (e.g., when intolerant to larotrectinib).
GI cancer: The OH-CCO GI Cancer DAC stated that entrectinib would not affect the sequencing of current therapies because it is recommended that patients try other treatments first; therefore, entrectinib would be an additional line of treatment (i.e., the indication would fall between any line of therapies). The group added that NTRK mutation testing should be added to an NGS panel for patients with GI cancer.
Breast cancer: According to the OH-CCO Breast Cancer DAC, because very few patients with breast cancer have NTRK mutation and NTRK gene testing is not routine, it remains unclear where entrectinib would fit into the current treatment paradigm. (Note: the clinical experts consulted by CADTH identified secretory breast cancer as an area where entrectinib may be beneficial.) OH-CCO Breast Cancer said entrectinib would be an additional line of therapy where many treatments for metastatic breast cancer are palliative.
HNT cancer: The OH-CCO HNT DAC stated that ideally, entrectinib would be used as a first-line therapy if NTRK mutation was identified before treatment initiation or as a later line if a patient is already on a different systemic therapy at the time of disease progression or intolerance (thereby pushing back the currently available treatments to later lines). OH-CCO HNT does not believe it would be appropriate to recommend other treatments before entrectinib.
Patient Population
Lung cancer: The OH-CCO Lung Cancer DAC stated that patients with metastatic NSCLC containing an NTRK fusion mutation are a subset of patients for whom the new treatment is needed. The group pointed out that ideally, these patients should be identified by reflex gene testing on lung cancer biopsies; however, some patients are missed due to lack of testing and/or problems with the availability of testing. LCC agreed that all patients with NSCLC and NTRK fusion mutation identified by NGS (or 2-part process of immunohistochemistry confirmed by NGS) should be offered entrectinib if they have not already received larotrectinib and if it is not otherwise contraindicated. LCC added that if NGS panels are limited, NTRK gene testing should be reserved for those who have tested negative for other driver mutations, given that NTRK mutation is mutually exclusive with these. Both groups agreed that patients without NTRK fusion mutation should not be treated with entrectinib. OH-CCO Lung Cancer added that patients with extremely poor ECOG PS should not be treated with entrectinib. In addition, LCC said that patients with ROS1 and NSCLC respond well to entrectinib.
GI cancer: The OH-CCO GI Cancer DAC said that suitable patients should be identified by NTRK gene testing and the least suitable patients would be those without NTRK fusion mutation and/or with poor ECOG PS.
Breast cancer: The OH-CCO Breast Cancer DAC stated that patients with NTRK fusion–positive breast tumours are suitable for entrectinib. The group added that ideally, all patients with metastatic breast cancer should be tested for NTRK fusion mutation; however, given its low prevalence in breast cancer overall and its higher prevalence in secretory breast cancer, identifying this subset is helpful.
HNT cancer: The OH-CCO HNT DAC reported that patients with NTRK mutations (found most commonly in salivary gland tumours and thyroid cancers) are most suitable for entrectinib treatment, while those without NTRK mutations and/or who have early stage or resectable cancers are the least suitable. The group expressed that, as with other types of solid tumours, molecular testing should be routinely performed on advanced and/or metastatic salivary and thyroid cancers to identify NTRK mutation.
Assessing Response to Treatment
Lung cancer: The OH-CCO Lung Cancer DAC said that treatment responses (i.e., stabilization, no deterioration in symptoms, or reduction in the frequency or severity of symptoms such as cough, shortness of breath, fatigue, anorexia, or pain) should be assessed every 4 weeks to 8 weeks and CT imaging should be done every 3 months to 4 months. LCC stated that clinical assessment of tolerability, symptoms, and tumour response by imaging (CT or MRI) should be done every 6 weeks to 12 weeks, depending on patient factors, with frequency and specific measurement tool determined by local practice. LCC added that meaningful improvement would be indicated by reduced or improved symptoms, prolonged life, and good quality of life, such as independent living and the ability to achieve personal life goals.
GI cancer: The OH-CCO GI Cancer DAC stated that the outcomes used to determine response are improvement in cancer symptom control and objective response by tumour markers. Reduction in the frequency or severity of symptoms as well as response on imaging assessed as per standard GI cancer practice (i.e., every 2 months to 4 months) are considered meaningful.
Breast cancer: The OH-CCO Breast Cancer DAC stated that OS is the outcome used to determine response to treatment, and tumour response within the breast is considered a clinically meaningful response, with frequency of assessment as per the study protocol.
HNT cancer: The OH-CCO HNT DAC mentioned that cancer-related or clinical symptoms, physical exam findings related to the cancer (i.e., lymphadenopathy), and imaging assessed approximately every 3 months are the outcomes used to determine response. In addition, OH-CCO HNT listed improved cancer-related symptoms and shrinkage or stabilization of cancer as clinically meaningful responses.
Discontinuing Treatment
Lung cancer: Both the OH-CCO Lung Cancer DAC and LCC stated that intolerance or toxicity (especially ≥ grade 3), patient wish, and unequivocal disease progression (clinical or radiographic) not amenable to control with targeted radiation are criteria for discontinuing treatment.
GI, breast, and HNT cancer: According to the 3 clinician groups representing these cancer types, unequivocal disease progression and/or intolerance should guide decisions about treatment discontinuation.
Prescribing Conditions
Lung cancer: Both the OH-CCO Lung Cancer DAC and LCC said that home is an appropriate setting for the treatment. OH-CCO Lung Cancer added that it is appropriate to start and administer entrectinib in certain hospitalized patients. LCC added that entrectinib should be prescribed only by a specialist, most commonly a medical or pediatric oncologist; however, in some jurisdictions where respirologists oversee systemic therapy, it is appropriate for them to prescribe entrectinib.
GI cancer: The OH-CCO GI Cancer DAC mentioned that an outpatient cancer centre with a medical oncologist is the appropriate setting for treatment with entrectinib.
Breast cancer: The OH-CCO Breast Cancer DAC stated that a cancer centre with expertise in toxicity management is the appropriate setting for this treatment.
HNT cancer: The OH-CCO HNT DAC said entrectinib should be prescribed by a specialist (i.e., a medical oncologist) and can be taken at home as an oral therapy.
Additional Considerations
The OH-CCO GI Cancer DAC said that NTRK gene testing is currently funded only for patients with CRC. It recommends expanding testing to include all GI cancers and to expand access to provincial pathology lab reports with NTRK testing results, which are currently not available to clinicians.
The OH-CCO Breast Cancer DAC said that NTRK fusion mutation is rare in breast cancer, but that a third of patients in the clinical trial (N = 6) had a CR with entrectinib; therefore, a different paradigm (i.e., a disease-agnostic perspective) is needed for evaluating these treatments.
Drug Program Input
The drug programs provide input on each drug being reviewed through CADTH’s reimbursement review processes by identifying issues that may affect the programs’ abilities to implement a recommendation. The implementation questions and corresponding responses from the clinical experts consulted by CADTH are summarized in Table 7.
Table 7: Summary of Drug Plan Input and Clinical Expert Response
Drug program implementation questions | Clinical expert response |
|---|---|
Relevant comparators | |
The pivotal studies of entrectinib included patients with CNS metastases who were previously treated and/or asymptomatic. For patients with NTRK fusion–positive solid tumours and CNS metastases, is there a preferred TRK inhibitor (e.g., larotrectinib vs. entrectinib)? | Based on the limited available data, entrectinib may have more CNS penetration and promising CNS activity. However, additional data and longer-term follow-up would be required to establish any conclusions regarding the comparative effectiveness of entrectinib and larotrectinib for patients with CNS metastases. |
How do the efficacy and safety of entrectinib compare to best supportive care for patients with unresectable, locally advanced or metastatic solid tumours with confirmed NTRK fusion–positive disease who have exhausted all other therapies? | The clinical experts consulted by CADTH noted that entrectinib would be expected to act in a manner similar to other current histology-specific targeted drugs (such as ALK or EGFR). This is based on the evidence available — the data that demonstrate that patients with NTRK-positive cancers do not have better outcomes compared to patients without the mutation — as well as on the very high response rates observed with entrectinib and larotrectinib in the presence of an NTRK gene fusion and on clinical expert opinion and experience, recognizing that the ORRs reported for entrectinib and larotrectinib surpass the expected response rates with alternate systemic therapies in advanced diseases. |
How do the efficacy and safety of entrectinib compare to existing systemic therapies used for the treatment of unresectable, locally advanced or metastatic solid tumours with confirmed NTRK fusion–positive disease in any line of therapy? | |
Considerations for initiation of therapy | |
Patients with an ECOG PS of 2 or less were eligible for the pivotal trials. Should eligibility for treatment with entrectinib be limited to patients with an ECOG PS of 2 or less? | The clinical experts suggested that patients with an ECOG PS ≤ 3 could be eligible for treatment with entrectinib if the oncologist believes that tumour-related symptoms are driving the PS. The rationale is based on the high rate of response, duration of response, median time to response (i.e., approximately 1 month), and favourable toxicity profile. |
What is an appropriate definition for “no satisfactory treatment options” for unresectable, locally advanced or metastatic solid tumours with confirmed NTRK fusion–positive disease? | The clinical experts consulted by CADTH noted that the definition of “no satisfactory treatment options” would depend on the tumour sites and reflect the range of alternative therapies available for those tumours (e.g., some have no alternatives, while others may have several alternatives). The clinical experts agreed that “no satisfactory treatment options” would be interpreted by clinicians to mean suboptimal treatment for the patient with respect to achieving treatment goals (e.g., improving survival and disease-free interval), or be associated with poor quality of life and/or significant toxicity. |
The funding request for entrectinib is for use in adult patients with extracranial solid tumours only. Pediatric patients were not included in the funding request or the Health Canada approval. Patients with primary CNS solid tumours were not included in the funding request. Is there evidence to inform the use of entrectinib in:
| The sponsor initially sought regulatory approval for an indication that would include use in pediatric patients as well as those with primary CNS tumours. Health Canada did not approve usage in pediatric patients, citing the negative benefit vs. risk profile of entrectinib in the pediatric population. It also did not approve usage in patients with primary CNS tumours, citing the lack of sufficient efficacy data to support benefit in primary brain tumours. |
Considerations for continuation or renewal of therapy | |
The STARTRK-2, STARTRK-1, and ALKA trials performed on-treatment tumour assessments through CT and/or MRI at the end of cycle 1 (i.e., 4 weeks) and at the end of alternate cycles thereafter (i.e., every 8 weeks), or whenever a clinical deterioration was observed, and at end of treatment if not done in the previous 4 weeks. What are clinically appropriate modalities and frequencies to assess therapeutic response to entrectinib? | In terms of outcomes that are used to determine whether a patient is responding to treatment in clinical practice, the clinicians indicated that typical metrics of treatment efficacy include disease evaluation by cross-sectional imaging modalities (MRI, CT, and PET/CT) to assess response by RECIST 1.1 (for solid tumours) or RANO (for CNS tumours), symptom improvement, treatment tolerability, and time to progression. The clinicians noted that treatment response is typically assessed every 3 months, and that once response is established or remission is achieved, this interval may be prolonged. |
Considerations for discontinuation of therapy | |
The STARTRK-2, STARTRK-1, and ALKA trials permitted dose reductions due to toxicity (up to a maximum of 2) and treatment interruption of up to 28 days due to treatment-related adverse effects. Treatment was discontinued if symptoms did not resolve. Are the treatment interruption and/or discontinuation parameters used in STARKTR-2, STARKTR-1, and ALKA applicable to clinical practice? | The product monograph for entrectinib provides detailed recommendations for the management of adverse events that require temporary interruption, dose reduction, or discontinuation of treatment with entrectinib. The clinical experts consulted by CADTH indicated that this is a reasonable reflection of how patients would be managed in clinical practice. |
ALK = anaplastic lymphoma kinase; CNS = central nervous system; ECOG PS = Eastern Cooperative Oncology Group Performance Status; EGFR = epidermal growth factor receptor; NTRK = neurotrophic tyrosine receptor kinase; ORR = objective response rate; RANO = Response Assessment in Neuro-Oncology; RECIST 1.1 = Response Evaluation Criteria In Solid Tumours Version 1.1; TRK = tropomyosin receptor kinase; vs. = versus.
Clinical Evidence
Systematic Review (Pivotal and Protocol Selected Studies)
Objectives
To perform a systematic review of the beneficial and harmful effects of entrectinib (100 mg and 200 mg) for the treatment of adult patients who have unresectable, locally advanced or metastatic extracranial solid tumours (including brain metastases) with NTRK gene fusion without a known acquired resistance mutation and no satisfactory treatment options.
Methods
The studies selected for inclusion in the systematic review include pivotal studies provided in the sponsor’s submission to CADTH and Health Canada as well as those meeting the selection criteria presented in Table 8. Outcomes included in the CADTH review protocol reflect outcomes considered to be important to patients, clinicians, and drug plans.
Table 8: Inclusion Criteria for the Systematic Review
Criteria | Description |
|---|---|
Population | Adult patients who have unresectable, locally advanced or metastatic extracranial solid tumours (including brain metastases) with NTRK gene fusion without a known acquired resistance mutation and no satisfactory treatment options Subgroups:
|
Intervention | Entrectinib, 600 mg once daily, oral |
Comparator |
|
Outcomes | Efficacy outcomes:
Harms outcomes: AEs, SAEs, WDAEs, mortality, notable harms and/or harms of special interest (e.g., heart failure, QTc prolongation, neurologic events [e.g., cognitive impairment, mood disorders, dizziness, sleep disturbances, syncope], skeletal fractures, hepatotoxicity, hyperuricemia, and ophthalmologic events [e.g., vision disorders]) |
Study designs | Phase II, III, or IV clinical trials |
AE = adverse event; BSC = best supportive care; CNS = central nervous system; ECOG PS = Eastern Cooperative Oncology Group Performance Status; HRQoL = health-related quality of life; NTRK = neurotrophic tyrosine receptor kinase; OS = overall survival; PFS = progression-free survival; SAE = serious adverse event; WDAE = withdrawal due to adverse event.
The literature search for clinical studies was performed by an information specialist using a peer-reviewed search strategy according to the PRESS Peer Review of Electronic Search Strategies checklist.36
Published literature was identified by searching the following bibliographic databases: MEDLINE All (1946–) through Ovid and Embase (1974–) through Ovid. All Ovid searches were run simultaneously as a multi-file search. Duplicates were removed using Ovid deduplication for multi-file searches, followed by manual deduplication in Endnote. The search strategy comprised both controlled vocabulary, such as the National Library of Medicine’s MeSH (Medical Subject Headings), and keywords. The main search concept was Rozlytrek (entrectinib). Clinical trials registries were searched: the US National Institutes of Health’s clinicaltrials.gov, WHO’s International Clinical Trials Registry Platform search portal, Health Canada’s Clinical Trials Database, and the European Union Clinical Trials Register.
No filters were applied to limit the retrieval by study type. Retrieval was not limited by publication date or by language. Conference abstracts were excluded from the search results. Refer to Appendix 1 for the detailed search strategies.
The initial search was completed on February 23, 2022. Regular alerts updated the search until the meeting of the CADTH pan-Canadian Oncology Drug Review Expert Committee on July 13, 2022.
Grey literature (literature that is not commercially published) was identified by searching relevant websites from the CADTH Grey Matters: A Practical Tool For Searching Health-Related Grey Literature checklist.37 Included in this search were the websites of regulatory agencies (US FDA and European Medicines Agency). Google was used to search for additional internet-based materials. Refer to Appendix 1 for more information on the grey literature search strategy.
These searches were supplemented by reviewing bibliographies of key papers and through contacts with appropriate experts. In addition, the drug sponsor was contacted for information regarding unpublished studies.
Two CADTH clinical reviewers independently selected studies for inclusion in the review based on titles and abstracts, according to the predetermined protocol. Full-text articles of all citations considered potentially relevant by at least 1 reviewer were acquired. Reviewers independently made the final selection of studies to be included in the review, and differences were resolved through discussion.
Findings From the Literature
A total of 320 studies were identified from the literature for inclusion in the systematic review (Figure 2). The included studies are summarized in Table 9. There were no excluded studies.
Table 9: Details of Individual Included Studies
Detail | ALKA-372 to 001 study | STARTRK-1 study | STARTRK-2 study |
|---|---|---|---|
Designs and populations | |||
Study design | Multi-centre, non-randomized, open-label, phase I dose escalation study | Multi-centre, non-randomized, open-label, phase I dose escalation and expansion study | Multi-centre, non-randomized, open-label, phase II basket study |
Locations | 2 sites in Italy | 10 sites across 3 countries (US, Spain, South Korea) | > 150 sites across 15 countries (Australia, Belgium, France, Germany, Hong Kong, Italy, Japan, South Korea, Netherlands, Poland, Singapore, Spain, Taiwan, UK, US) |
Patient enrolment dates | October 2012 to March 2018 | August 2014 to May 2018 | Ongoing since November 2015 |
Enrolled (N) | N (total) = 58 N (integrated analysis) = 1 (efficacy analysis); 57 (safety analysis) | N [total] = 76 N [integrated analysis] = 2 (efficacy analysis); 76 (safety analysis) | N [integrated analysis] = 118 (efficacy analysis); 188 (safety analysis) |
Inclusion criteria |
|
|
|
Exclusion criteria |
|
|
|
Drugs | |||
Intervention | Entrectinib orally in 3 dosing schedules until phase II dose determined (dose range = 100 mg/m2 to 1,800 mg/m2): Schedule A:
Schedule B:
Schedule C:
| Dose escalation:
Dose expansion: 600 mg flat dose of entrectinib, orally, q.d. for 28 consecutive days in repeated 4-week cycles | Entrectinib orally 600 mg q.d. continuously for 28 days (4-week cycles) Note: For patients with CNS disease who have been on study for ≥ 2 cycles of treatment (i.e., 8 weeks) with a BR of SD as per RECIST 1.1 and without treatment-related grade ≥ 2 AEs, dose escalation to 800 mg q.d. was allowed |
Comparator(s) | NA (single-arm trial) | NA (single-arm trial) | NA (single-arm trial) |
Outcomes | |||
Primary end point | First-cycle DLTs and MTD administered in 3 different dosing schedules (A, B, and C) | Dose escalation: First-cycle DLTs, MTD, and RP2D Dose expansion: ORR, defined as the proportion of patients with complete or partial response | ORR by BICR assessment in each patient population basket (NTRK1, NTRK2, and NTRK3; ROS1; ALK) |
Secondary and exploratory end points |
| Dose escalation:
Dose expansion:
| Secondary:
Exploratory:
|
Notes | |||
Publications | De Braud et al. (2015)38 De Braud et al. (2014)39 | Patel et al. (2015)40 Drilon et al. (2016)41 Drilon et al. (2017)42 | Paz-Ares et al. (2021)43 Drilon et al. (2017)44 |
AE = adverse event; ALCL = anaplastic large-cell lymphoma; ALK = anaplastic lymphoma kinase; BICR = blinded independent central review; BM = brain metastases; BR = best response; CBR = clinical benefit rate; CHF = coronary heart failure; CNS = central nervous system; CTCAE = Common Terminology Criteria for Adverse Events; DLT = dose-limiting toxicity; DOR = duration of response; DVT = deep vein thrombosis; ECOG PS = Eastern Cooperative Oncology Group Performance Status; EIAED = enzyme-inducing anti-epileptic drug; EMA = European Medicines Agency; HRQoL = health-related quality of life; GI = gastrointestinal; MI = myocardial infarction; MTD = maximum tolerated dose; NA = not applicable; NCI = National Cancer Institute; NSCLC = non–small cell lung cancer; NTRK = neurotrophic tyrosine receptor kinase; ORR = objective response rate; OS = overall survival; PFS = progression-free survival; PD = progressive disease; PK = pharmacokinetic; q.d. = once a day; RANO = Response Assessment in Neuro-Oncology; RECIST = Response Evaluation Criteria in Solid Tumours; ROS1 = ROS proto-oncogene 1; RP2D = recommended phase II dose; SD = stable disease; TKI = tyrosine kinase inhibitor; TRK = tropomyosin receptor kinase; TTR = time to response.
Note: Eight additional reports were included: FDA Multidisciplinary Review45; Health Canada Pharmaceutical Safety and Efficacy Assessment46; EMA European Public Assessment Report27; Clinical Study Reports for ALKA, STARTRK-1, and STARTRK-247-49; and the Integrated Reports for Safety and Efficacy.50
Source: Drug reimbursement review sponsor submission.50
Table 10: Details of Integrated Analysis
Detail | Integrated analysis |
|---|---|
Designs and populations | |
Study design | Pooled analyses of efficacy and safety data from patients who were NTRK fusion–positive enrolled in the ALKA, STARTRK-1, and STARTRK-2 trials |
Enrolled (N) | Integrated efficacy-evaluable population: N = 121 (August 2020 CCOD):
Integrated safety-evaluable population: N = 193 (August 2020 CCOD):
|
Inclusion criteria | Integrated efficacy-evaluable population (N = 121):
Integrated NTRK safety-evaluable population (N = 193):
|
Drugs | |
Intervention |
|
Comparator(s) | NA (single-arm trial) |
Outcomes | |
Primary end points |
|
Secondary and exploratory end points |
|
Notes | |
Publications | |
BOR = best objective response; CBR = clinical benefit rate; CCOD = clinical cut-off date; CNS = central nervous system; DOR = duration of response; NA = not applicable; NTRK = neurotrophic tyrosine receptor kinase; ORR = objective response rate; OS = overall survival; PFS = progression-free survival; q.d. = once a day; TRK = tropomyosin receptor kinase.
Source: Demetri et al. (2022).51
Description of Studies
The CADTH systematic review included 3 open-label, single-group trials (ALKA, STARTRK-1, and STARTRK-2) and a pooled analysis of entrectinib in adult patients with advanced or metastatic solid tumours.
ALKA-372 to 001 Study
ALKA-372 to 001 was an open-label, multi-centre, non-randomized, single-group, phase I dose escalation trial in adult patients with advanced and/or metastatic solid tumours with NTRK1, NTRK2, NTRK3, ROS1, or ALK genetic alterations.45 Patients were enrolled from October 26, 2012 to March 27, 2018. The trial was conducted at 2 sites in Milan, Italy.54 Three dosing schedules were investigated using a conventional 3 plus 3 patient enrolment scheme. All patients had to be observed for 1 cycle before subsequent patients were enrolled at the next (higher) dose level.45
STARTRK-1 Study
STARTRK-1 was an open-label, multi-centre, phase I trial in adult patients with locally advanced or metastatic solid tumours.45 STARTRK-1 and ALKA-372 to 001 were conducted concurrently and were interdependent; i.e., dose escalation decisions in 1 study affected the conduct of the other.55 Patients were enrolled from August 7, 2014 to May 10, 2018. The trial was conducted at 10 sites in 3 countries (8 sites in the US, 1 in Spain, and 1 in South Korea).54 The trial included a dose escalation and a dose expansion phase; molecular alterations were required only for enrolment in the dose expansion phase (NTRK1, NTRK2, NTRK3, ROS1, or ALK by local testing). Dose escalation was investigated using a conventional 3 plus 3 enrolment scheme with an accelerated titration design. Eligible patients enrolled in the dose expansion phase were grouped into molecularly-defined cohorts under a Simon’s 2-stage (minmax) design determined by the type of molecular alteration harboured by the patient’s tumour.45
STARTRK-2 Study
STARTRK-2 is an open-label, multi-centre, phase II basket trial in adult patients with locally advanced or metastatic solid tumours with an NTRK1, NTRK2, NTRK3, ROS1, or ALK molecular alteration.45 STARTRK-2 is ongoing and is being conducted at more than 150 sites in 15 countries.54 Patients are assigned to different baskets according to tumour type and gene fusion (Figure 3). NTRK1, NTRK2, and NTRK3 are treated as a combined NTRK1, NTRK2, and NTRK3 gene rearrangement basket. Broadly, the baskets of patients are NTRK1, NTRK2, and NTRK3; ROS1; and ALK.45 Depending on the patient population sub-basket, prior systemic treatment is allowed; however, prior treatment with inhibitors of TRK, ROS1, or ALK are not allowed in patients with tumours harbouring those respective gene rearrangements except in patients with ALK or ROS1-rearranged NSCLC with CNS-only progression who were previously treated with crizotinib. Sub-baskets include patients who are or have: ALK-positive NSCLC with CNS progression post-crizotinib; ALK-positive non-NSCLC solid tumours; NTRK fusion–positive solid tumours; ROS1-positive NSCLC; ROS1-positive NSCLC CNS progression post-crizotinib; ROS1-positive non-NSCLC solid tumours; or who are enrolled in the Japan recommended phase II dose (RP2D) safety and tolerability substudy.56 Patients are enrolled in a non-evaluable basket if they are not assessable for the primary end point (i.e., did not have measurable disease or co-dual oncogenic drivers) but contribute to the assessment of safety and pharmacokinetics (PK).45 Treatment is continued until progression, patient withdrawal, or the occurrence of unacceptable toxicity.54
Integrated Analysis
The regulatory and reimbursement submissions for entrectinib are based on the pooled analyses of efficacy and safety data from patients enrolled in the 3 trials who were NTRK fusion–positive. The sponsor made the decision to pool efficacy and safety data from these trials based on early guidance from the FDA and European Medicines Agency. This decision was deemed justified due to the rarity of the tumours with NTRK gene fusion, the challenges involved in identifying and enrolling patients with rare diseases in clinical trials, and the lack of an established comparator in this disease population. Additionally, the eligibility criteria and trial conduct (including patient population, study end points, dosing schedules, and tumour assessment methodology and frequency) were similar across the 3 trials, further supporting the pooled efficacy and safety analyses.57
Figure 3: Schematic Showing the Design of the STARTRK-2 Basket Trial
ALK = anaplastic lymphoma kinase; CLIA = Clinical Laboratory Improvement Amendments; MD = medical doctor; NGS = next-generation sequencing; NTRK = neurotrophic tyrosine receptor kinase; ROS1 = ROS proto-oncogene 1; RT-PCR = reverse transcriptase polymerase chain reaction.
Populations
Inclusion and Exclusion Criteria
ALKA-372 to 001 Study
The ALKA-372 to 001 study included adult patients greater than or equal to 18 years of age with histologically or cytologically confirmed diagnoses of advanced and/or metastatic solid tumours with ALK-positive alterations and ALK-negative patients with TRKA or ROS1 alterations. The trial was expanded to include ALK-negative patients with TRKB or TRKC genetic alterations (as per amendment 6), with all molecular alterations confirmed by local testing only. Patients for whom no alternative effective standard therapy was available, for whom standard therapy was considered unsuitable, or who had been refused therapy (per protocol amendment 8) were eligible for the study.
Other main eligibility criteria included: an ECOG PS score of less than or equal to 2 and life expectancy of at least 3 months; controlled asymptomatic CNS involvement in the absence of therapy with an anticonvulsant (up to protocol amendment 7) or in the presence of therapy with non–enzyme-inducing anti-epileptic drugs (per protocol amendment 8) or requiring steroids at a stable dose (dexamethasone ≤ 4 mg/day or equivalent) for at least 2 weeks. The study had an accelerated dose escalation phase per cohort until a predetermined level of toxicity was observed. At that point, the dose escalation followed a modified Fibonacci scheme (50%, 40%, or 33% increments). Dose escalation was planned to continue until the RP2D was determined.45 Treatment was continued until progression, patient withdrawal, or the occurrence of unacceptable toxicity.54 There was no limit to how many cycles a patient could receive.58 Dose modifications were permitted for toxicities, and doses could be maintained or reduced after recovery from toxicities if patients recovered within a maximum of 2 weeks.45
STARTRK-1 Study
The STARTRK-1 trial included adult patients greater than or equal to 18 years of age with any locally advanced or metastatic tumours (dose escalation only) or histologically or cytologically confirmed diagnosis of locally advanced or metastatic solid tumours with an NTRK1, NTRK2, NTRK3, ROS1, or ALK molecular alteration (dose expansion). Patients must have had measurable disease according to RECIST 1.1, a life expectancy of at least 3 months, and an ECOG PS of less than or equal to 2. Prior cancer therapy was allowed, including crizotinib, ceritinib, investigational drugs, and radiotherapy. Patients with controlled asymptomatic CNS involvement were allowed. Resolution of any acute toxic effects (excluding alopecia) of any prior anti-cancer therapy (National Cancer Institute Common Terminology Criteria for Adverse Events version 4.03 grade ≤ 1) was required.54
STARTRK-2 Study
The STARTRK-2 trial includes adult patients greater than or equal to 18 years of age with locally advanced or metastatic solid tumours harbouring an NTRK1, NTRK2, NTRK3, ROS1, or ALK molecular alteration based on central testing, or based on local testing if a nucleic acid–based method is used and the local laboratory is Clinical Laboratory Improvement Amendments certified or equivalently accredited. (Patients enrolled based on local testing are required to submit a tissue sample for central confirmation.)54 Patients are required to have an ECOG PS of less than or equal to 2 and a minimum life expectancy of 4 weeks.49 Prior cancer therapy is allowed, excluding approved or investigational TRK, ROS1, or ALK inhibitors (non-NSCLC patients only) in patients with tumours that harbour the respective gene arrangements. Patients with CNS involvement — including leptomeningeal carcinomatosis that is asymptomatic or previously treated and controlled — are allowed.54
Integrated Analyses of NTRK Fusion–Positive Solid Tumours
The pooled analysis for the August 31, 2020 CCOD consisted of the following datasets:
NTRK safety-evaluable population (N = 193; 97% from STARTRK-2): all patients with an NTRK fusion–positive tumour who received at least 1 dose of entrectinib
NTRK efficacy-evaluable population (N = 121; 98% from STARTRK-2): all patients with NTRK fusion–positive extracranial primary tumours who received at least 1 dose of entrectinib and had measurable disease at baseline and at least 12 months of follow-up
NTRK efficacy-evaluable population with CNS metastases at baseline (N = 19 based on BICR assessment): subpopulation used for the evaluation of the “intracranial efficacy” end points.
The following procedures were employed to confirm that patients had solid tumours harbouring NTRK gene fusions:
confirmation that a Clinical Laboratory Improvement Amendments certified or equivalently accredited nucleic acid–based local test was used (several different assay methods were used in patients across the trials)
adequate nucleic acid specimen for producing a reliable test result
presence of an NTRK fusion known to result in oncogenic drive activity
gene fusions predicted to translate into a fusion protein with a functional kinase domain were considered positive gene fusions
other molecular alterations, such as rearrangements, deletions, or over-expression, were not considered positive for a gene fusion
lack of co-occurrence with other strong onco-driver mutations likely to confer resistance
The criteria to molecularly define eligibility for inclusion in the NTRK-positive, efficacy-evaluable population were applied consistently to all 3 study populations so that all patients who were NTRK-positive in the integrated dataset met the molecular criteria determined as per the STARTRK-2 protocol.59 A total of 193 adult patients were included in the updated, integrated safety analysis, and 121 adults were included in the updated efficacy analysis. Reasons for exclusion from the efficacy analysis population were not reported for the August 31, 2020 CCOD. As shown in Table 16, the NTRK efficacy-evaluable analysis population was further subdivided into those with CNS metastases (n = 26, based on investigator assessment; n = 19, based on BICR) and those without CNS metastases (N = 95, based on investigator assessment; N = 102, based on BICR).
Baseline Characteristics
Baseline characteristics for the ALKA, STARTRK-1, and STARTRK-2 trials and the integrated analysis are summarized in Table 11. The NTRK efficacy-evaluable analysis set (N = 121) included 59 male patients (48.8%) and 62 female patients (51.2%). The mean age was 55.9 years (standard deviation [SD] = 15.6), with a larger proportion of patients less than 65 years of age (64.5%) versus greater than or equal to 65 years of age (35.5%). The majority of patients were White (60.3%). Most patients had an ECOG PS of 0 (43.8%) or 1 (47.1%); a minority had an ECOG PS of 2 (9.1%). The mean body mass index of patients was 24.80 (SD = 5.15). Approximately 39% of patients reported a history of smoking.
The solid tumour types that were reported in at least 5% of the patients included sarcoma (n = 26; 21.5%), MASC (n = 24; 19.8%), NSCLC (n = 22; 18.2%), thyroid cancer (n = 13; 10.7%), colon cancer (n = 10; 8.3%), and breast cancer (n = 7; 5.8%). Nearly all patients (96.7%) had metastatic disease at baseline. The most common metastatic sites were lung (61.2%) and lymph nodes (55.4%). The majority of patients had NTRK3 fusions (n = 67; 55.4%), followed by NTRK1 (n = 48; 39.7%) and NTRK2 fusions (n = 6; 5.0%).
The majority of patients had received some form of prior anti-cancer therapy (n = 97; 80.2%): 74 patients (61.2%) had received any prior radiotherapy and 103 patients (85.1%) had previous cancer surgery. Overall, 30.6% of patients (n = 37) did not have prior systemic anti-cancer therapy, whereas 28.9% (n = 35) had 1 prior systemic therapy, 21.5% (n = 26) had 2 prior systemic therapies, 9.9% (n = 12) had 3 prior systemic therapies, 5.8% (n = 7) had 4 prior systemic therapies, and 3.3% (n = 4) had more than 4 lines of prior systemic therapy. The most frequent systemic prior anti-cancer therapy was chemotherapy (n = 88; 72.7%), followed by targeted therapy (n = 24; 19.8%), immunotherapy (n = 13; 10.7%), and hormonal therapy (n = 10; 8.3%).
There were 19 patients (17.2%) with CNS metastases at baseline as assessed by a BICR, with 17 patients (14.0%) reporting prior radiotherapy of the brain.
Figure 4: Safety and Efficacy Analysis Populations (CCOD: August 31, 2020)
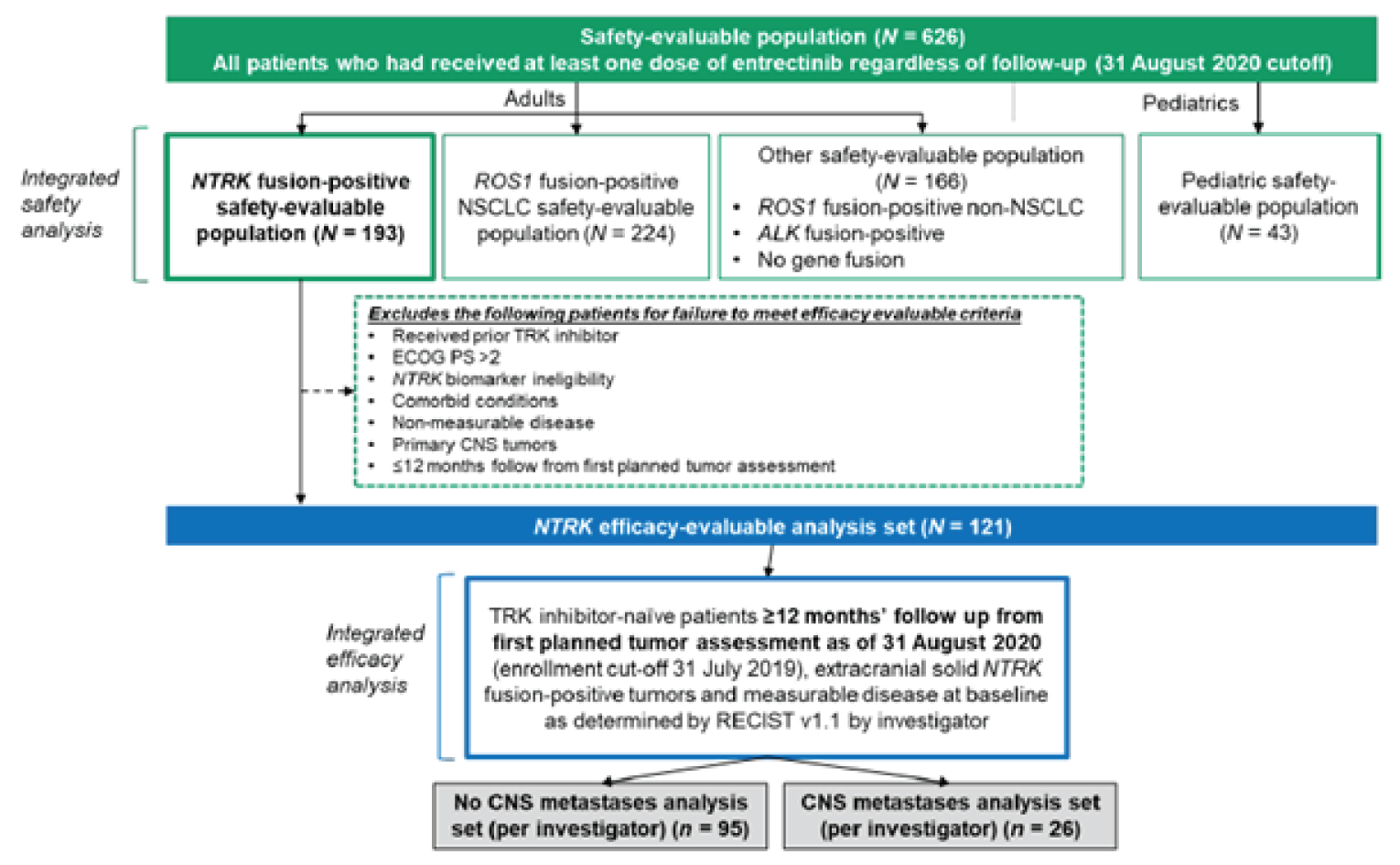
ALK = anaplastic lymphoma kinase; CNS = central nervous system; CCOD = clinical cut-off date; ECOG PS = Eastern Cooperative Oncology Group Performance Status; NSCLC = non–small cell lung carcinoma; NTRK = neurotrophic tyrosine receptor kinase; RECIST v1.1 = Response Evaluation Criteria in Solid Tumours Version 1.1; ROS1 = ROS proto-oncogene 1; TRK = tropomyosin receptor kinase.
Source: Drug reimbursement review sponsor submission.50
Table 11: Summary of Baseline Characteristics
Characteristic | ALKA study (N = 1) | STARTRK-1 study (N = 2) | STARTRK-2 study (N = 118) | Integrated analysis (N = 121) |
|---|---|---|---|---|
Sex, n (%) | ||||
Male | 0 | 1 (50.0) | 58 (49.2) | 59 (48.8) |
Female | 1 (100.0) | 1 (50.0) | 60 (50.8) | 62 (51.2) |
Age (years) | ||||
Mean (Std) | 75.0 (NE) | 44.0 (2.8) | 56.0 (15.6) | 55.9 (15.6) |
Median (IQR) | 75.0 (75.0 to 75.0) | 44.0 (42.0 to 46.0) | 57.0 (47.0 to 68.0) | 57.0 (46.0 to 68.0) |
< 65 years, n (%) | 0 | 2 (100.0) | 76 (64.4) | 78 (64.5) |
≥ 65 years, n (%) | 1 (100.0) | 0 | 42 (35.6) | 43 (35.5) |
Ethnicity, n (%) | ||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
Race, n (%) | ||||
Asian | 0 | 0 | 29 (24.6) | 29 (24.0) |
Black or African American | 0 | 0 | 3 (2.5) | 3 (2.5) |
White | 1 (100.0) | 2 (100.0) | 70 (59.3) | 73 (60.3) |
Other | 0 | 0 | 1 (0.8) | 1 (0.8) |
Not reported | 0 | 0 | 15 (12.7) | 15 (12.4) |
Country or region, n (%) | ||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
Weight (kg) | ||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
BSA (m2) | ||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
BMI (kg/m2) | ||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
ECOG PS, n (%) | ||||
0 | 0 | 0 | 53 (44.9) | 53 (43.8) |
1 | 1 (100.0) | 2 (100.0) | 54 (45.8) | 57 (47.1) |
2 | 0 | 0 | 11 (9.3) | 11 (9.1) |
History of smoking, n (%) | ||||
No | 0 | 0 | 72 (62.1) | 72 (61.0) |
Yes | 0 | 2 (100.0) | 44 (37.9) | 46 (39.0) |
Current | 0 | 0 | 10 (22.7) | 10 (21.7) |
Former | 0 | 2 (100.0) | 34 (77.3) | 36 (78.3) |
NTRK category, n (%) | ||||
NTRK1 | 1 (100.0) | 1 (50.0) | 46 (39.0) | 48 (39.7) |
NTRK2 | 0 | 0 | 6 (5.1) | 6 (5.0) |
NTRK3 | 0 | 1 (50.0) | 66 (55.9) | 67 (55.4) |
Primary diagnosis, n (%) | ||||
Breast | 0 | 0 | 7 (5.9) | 7 (5.8) |
Cholangiocarcinoma | 0 | 0 | 1 (0.8) | 1 (0.8) |
Colon cancer | 1 (100.0) | 0 | 9 (7.6) | 10 (8.3) |
CUP | 0 | 0 | 3 (2.5) | 3 (2.5) |
GI (other) | 0 | 0 | 1 (0.8) | 1 (0.8) |
Gynecological | 0 | 0 | 2 (1.7) | 2 (1.7) |
Head and neck | 0 | 0 | 2 (1.7) | 2 (1.7) |
Neuroblastoma | 0 | 0 | 1 (0.8) | 1 (0.8) |
Neuroendocrine | 0 | 0 | 5 (4.2) | 5 (4.1) |
NSCLC | 0 | 1 (50.0) | 21 (17.8) | 22 (18.2) |
Pancreatic | 0 | 0 | 4 (3.4) | 4 (3.3) |
Salivary (MASC) | 0 | 1 (50.0) | 23 (19.5) | 24 (19.8) |
Sarcoma | 0 | 0 | 26 (22.0) | 26 (21.5) |
Thyroid | 0 | 0 | 13 (11.0) | 13 (10.7) |
Histology, n (%) | ||||
Adenocarcinoma | 0 | 1 (50.0) | 17 (14.4) | 18 (14.9) |
Angiosarcoma | 0 | 0 | 1 (0.8) | 1 (0.8) |
Breast (non-secretory) | 0 | 0 | 2 (1.7) | 2 (1.7) |
Breast (secretory) | 0 | 0 | 4 (3.4) | 4 (3.3) |
Breast (NOS) | 0 | 0 | 1 (0.8) | 1 (0.8) |
Cervical adenosarcoma | 0 | 0 | 1 (0.8) | 1 (0.8) |
Cholangiocarcinoma | 0 | 0 | 1 (0.8) | 1 (0.8) |
Chondrosarcoma | 0 | 0 | 1 (0.8) | 1 (0.8) |
Colon | 0 | 0 | 1 (0.8) | 1 (0.8) |
CRC | 1 (100.0) | 0 | 9 (7.6) | 10 (8.3) |
CUP | 0 | 0 | 3 (2.5) | 3 (2.5) |
Endometrial carcinoma | 0 | 0 | 1 (0.8) | 1 (0.8) |
Endometrial stromal sarcoma | 0 | 0 | 1 (0.8) | 1 (0.8) |
Follicular dendritic cell sarcoma | 0 | 0 | 1 (0.8) | 1 (0.8) |
GIST | 0 | 0 | 2 (1.7) | 2 (1.7) |
Head and neck (NOS) | 0 | 0 | 2 (1.7) | 2 (1.7) |
Inflammatory myofibroblastic tumour | 0 | 0 | 1 (0.8) | 1 (0.8) |
Leiomyosarcoma | 0 | 0 | 2 (1.7) | 2 (1.7) |
MASC | 0 | 1 (50.0) | 23 (19.5) | 24 (19.8) |
MPNST | 0 | 0 | 1 (0.8) | 1 (0.8) |
Neuroblastoma | 0 | 0 | 1 (0.8) | 1 (0.8) |
Neuroendocrine | 0 | 0 | 3 (2.5) | 3 (2.5) |
Non-CRC GI (NOS) | 0 | 0 | 1 (0.8) | 1 (0.8) |
NSCLC (NOS) | 0 | 0 | 2 (1.7) | 2 (1.7) |
Ovarian | 0 | 0 | 1 (0.8) | 1 (0.8) |
Pancreatic | 0 | 0 | 4 (3.4) | 4 (3.3) |
Papillary thyroid | 0 | 0 | 10 (8.5) | 10 (8.3) |
Paraganglioma | 0 | 0 | 1 (0.8) | 1 (0.8) |
Sarcoma other | 0 | 0 | 10 (8.5) | 10 (8.3) |
Spindle cell | 0 | 0 | 5 (4.2) | 5 (4.1) |
Squamous cell carcinoma | 0 | 0 | 2 (1.7) | 2 (1.7) |
Thyroid (other) | 0 | 0 | 3 (2.5) | 3 (2.5) |
Time since diagnosis (months) | ||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
Stage at initial diagnosis, n (%) | ||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
Extent of disease, n (%) | ||||
Locally advanced | 0 | 0 | 4 (3.4) | 4 (3.3) |
Metastatic disease | 1 (100.0) | 2 (100.0) | 114 (96.6) | 117 (96.7) |
Metastatic sites, n (%) | ||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
Previous cancer treatments, n (%) | ||||
Any previous therapy | 1 (100.0) | 2 (100.0) | 94 (79.7) | 97 (80.2) |
Any chemotherapy | 1 (100.0) | 2 (100.0) | 85 (72.0) | 88 (72.7) |
Any immunotherapy | 0 | 1 (50.0) | 12 (10.2) | 13 (10.7) |
Any targeted therapy | 1 (100.0) | 1 (50.0) | 22 (18.6) | 24 (19.8) |
Any hormonal therapy | 0 | 0 | 10 (8.5) | 10 (8.3) |
Prior lines of therapy, n (%) | ||||
0 | 0 | 0 | 37 (31.4) | 37 (30.6) |
1 | 0 | 0 | 35 (29.7) | 35 (28.9) |
2 | 0 | 0 | 26 (22.0) | 26 (21.5) |
3 | 1 (100.0) | 0 | 11 (9.3) | 12 (9.9) |
4 | 0 | 2 (100.0) | 5 (4.2) | 7 (5.8) |
> 4 | 0 | 0 | 4 (3.4) | 4 (3.3) |
Any previous radiotherapy | 0 | 1 (50.0) | 73 (61.9) | 74 (61.2) |
Any previous surgeries | 0 | 2 (100.0) | 101 (85.6) | 103 (85.1) |
Baseline CNS lesions by investigator, n (%) | ||||
Measurable | 0 | 1 (50.0) | 5 (4.2) | 6 (5.0) |
Present | 0 | 0 | 20 (16.9) | 20 (16.5) |
Absent | 1 (100.0) | 1 (50.0) | 93 (78.8) | 95 (78.5) |
Baseline CNS lesions by BICR assessment, n (%) | ||||
Measurable | 0 | 1 (50.0) | 10 (8.5) | 11 (9.1) |
Present | 0 | 0 | 8 (6.8) | 8 (6.6) |
Absent | 1 (100.0) | 1 (50.0) | 100 (84.7) | 102 (84.3) |
Any prior radiotherapy of the brain, n (%) | ||||
Prior CNS disease treatment | 0 | 0 | 17 (14.4) | 17 (14.0) |
No | 1 (100.0) | 2 (100.0) | 101 (85.6) | 104 (86.0) |
Time from end of prior radiotherapy to first dose, n (%) | ||||
n | 0 | 0 | 17 | 17 |
< 2 months | 0 | 0 | 7 (41.2) | 7 (41.2) |
2 months to < 6 months | 0 | 0 | 5 (29.4) | 5 (29.4) |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
BICR = blinded independent central review; BMI = body mass index; BSA = body surface area; CNS = central nervous system; CRC = colorectal cancer; CUP = carcinoma of unknown primary; ECOG = Eastern Cooperative Oncology Group; GI = gastrointestinal; GIST = gastrointestinal stromal tumour; IQR = interquartile range; MASC = mammary analogue secretory carcinoma; MPNST = malignant peripheral nerve sheath tumour; NE = not estimable; NOS = not otherwise specified; NSCLC = non–small cell lung carcinoma; NTRK = neurotrophic tyrosine receptor kinase; Std = standard deviation.
Source: Supplemental Efficacy Results (clinical cut-off date: August 31, 2020).11
Interventions
ALKA-372 to 001 Study
In the ALKA-372 to 001 study, 3 entrectinib dosing schedules were investigated as follows:
schedule A: 4 days on, 3 days off for 3 weeks, followed by a 7-day rest period in a 4-week cycle; fasted condition; once or twice daily dosing
schedule B: continuous daily dosing in a 4-week cycle; fed condition; once-daily dosing
schedule C: 4 days on, 3 days off in a 4-week cycle; fed condition; once or twice daily dosing.
The dose escalation for all schedules was planned to continue until the RP2D was determined or until the study was terminated at the discretion of the sponsor. For all schedules, a conventional 3 plus 3 patient enrolment scheme was followed during dose escalation. The study had an initial 100% accelerated escalation phase until a predetermined level of toxicity was encountered. At that point, escalation was to follow a modified Fibonacci scheme. Dose escalation continued until a dose-limiting toxicity (DLT) was observed in 2 of 3 or 2 of 6 patients in cycle 1. If 2 of 3 patients experienced a DLT, then enrolment into the cohort ceased. If a dose level was deemed safe (i.e., 0 of 3 or 1 of 6 patients experienced a DLT), then additional patients (at least 15 total patients per cohort) could have been enrolled at the sponsor’s discretion to obtain additional safety, tolerability, and PK data. A DLT was defined as an AE occurring during the first treatment cycle that fulfilled pre-specified criteria and grading (generally grade ≥ 3 in severity) and for which a causal relationship to entrectinib could not be excluded. Failure to recover (excluding alopecia) after delaying the initiation of next treatment administration by a maximum of 14 days — and failure to complete the first-cycle treatment with at least 75% of the planned doses because of a drug-related toxicity — also met the criteria for a DLT.
STARTRK-1 Study
In the STARTRK-1 dose escalation phase, each cycle consisted of 28 days in repeated 4-week cycles (cycle 1 was 42 days until it was reduced to 28 days in amendment 5 for all patients). The starting dose was 100 mg/m2 once daily in the fed condition. (Doses of 200 mg/m2, 400 mg/m2, 600 mg, and 800 mg were also studied.) Dose escalation followed an accelerated phase in which the dose was doubled in successive cohorts until 1 patient experienced DLT in the first cycle or 2 patients experienced AEs at least possibly related to entrectinib that were grade 2 or greater severity (but not considered to be DLTs) and that occurred during the first cycle, whichever came first. If these situations occurred, dose escalation followed a modified Fibonacci scheme (i.e., 50%, 40%, or 33% increments). RP2D was to be determined based on available safety, tolerability, and PK and pharmacodynamic data from different dose levels and schedules tested; once determined, administration of a flat dose was to be considered for a subgroup of patients if data supported the decision. In dose expansion, patients received entrectinib orally in repeat 28-day cycles at a fixed dose of 600 mg once daily determined as the RP2D from dose escalation. Treatment was continued until progression, patient withdrawal, or the occurrence of unacceptable toxicity. Treatment could continue beyond progression after discussion with the sponsor.45 There was no limit to how many cycles during which a patient could receive treatment.60 Dose modifications were permitted for toxicities, and doses could be maintained or reduced (up to 2 dose levels) unless a patient was receiving the starting dose of 100 mg/m2/day (in which case, the patient would be instructed to stop treatment); dose interruptions were allowed for a maximum of 28 days for recovery.45,54
STARTRK-2 Study
In STARTRK-2, entrectinib is administered at a dose of 600 mg orally as capsules on a continuous daily dosing regimen in 28-day cycles. Doses can be reduced by 2 dose levels (400 mg and 200 mg) to manage AEs if these are considered related to entrectinib, or doses can be resumed at the initial dose if AEs are considered unrelated to entrectinib (and treatment is interrupted until the AE stabilizes). Dose escalation to 800 mg once daily is allowed for patients with CNS disease if they have been on treatment for at least 2 cycles with a best response of stable disease and without grade 2 or greater AEs.45 Treatment is continued until progression, patient withdrawal, or the occurrence of unacceptable toxicity.54
Outcomes
A list of efficacy end points identified in the CADTH review protocol that were assessed in the clinical trials included in this review is provided in Table 12. These end points are further summarized here. A detailed discussion and critical appraisal of the outcome measures are provided in Appendix 3.
Table 12: Summary of Outcomes of Interest Identified in the CADTH Review Protocol
Outcome measure | ALKA-372 to 001 study | STARTRK-1 study dose escalation | STARTRK-1 study dose expansion | STARTRK-2 study | Integrated analysis |
|---|---|---|---|---|---|
ORR | Secondary | Secondary | Primary | Primary | Primary |
PFS | NA | Secondary | Secondary | Secondary | Secondary |
OS | NA | Secondary | Secondary | Secondary | Secondary |
BOR | NA | Secondary | Secondary | Secondary | Secondary |
TTR | NA | NA | NA | Secondary | Secondary |
CBR | NA | NA | NA | Secondary | Secondary |
Intracranial ORR | NA | NA | Secondary | Secondary | Secondary |
Intracranial DOR | NA | NA | NA | NA | Secondary |
Intracranial PFS | NA | NA | NA | Secondary | Secondary |
QLQ-C30 | NA | NA | NA | Exploratory | NA |
QLQ-LC13 (NSCLC) | NA | NA | NA | Exploratory | NA |
QLQ-CR29 (mCRC) | NA | NA | NA | Exploratory | NA |
Safety profile | Secondary | Secondary | NA | Secondary | NA |
PK parameters | Secondary | Secondary | Secondary | Secondary | NA |
PD parameters | Secondary | Secondary | Secondary | NA | NA |
First-cycle DLTs, MTD | Primary | NA | NA | NA | NA |
First-cycle DLTs, MTD, RP2D | NA | Primary | NA | NA | NA |
BOR = best overall response; CBR = clinical benefit rate; DLT = dose-limiting toxicity; DOR = duration of response; mCRC = metastatic colorectal cancer; MTD = maximum tolerated dose; NA = not applicable; NSCLC = non–small cell lung carcinoma; ORR = overall response rate; OS = overall survival; PD = pharmacodynamic; PFS = progression-free survival; PK = pharmacokinetic; QLQ-C30 = Quality of Life Questionnaire Core 30; QLQ-CR29 = Quality of Life Questionnaire Colorectal Cancer 29; QLQ-LC13 = Quality of Life Questionnaire Lung Cancer 13; RP2D = recommended phase II dose; TTR = time to tumour response.
Source: Clinical Study Reports and Common Technical Document, Section 2.7.3.47-49,61
Tumour Response Assessment
Tumour responses were assessed using CT or MRI by investigator (local) assessment and by BICR and evaluated using RECIST 1.1. Screening assessments were performed within 30 days before the first administration of entrectinib, and on-treatment tumour assessments were performed at the end of every odd cycle (starting with cycle 1) or when clinical deterioration was observed, and at end of treatment (if not done in the previous 4 weeks). Tumour assessments could be performed outside of protocol-defined time points at the discretion of the investigator. For patients who experienced a CR or PR, response confirmation was performed no fewer than 4 weeks from when response criteria were first met. Tumour response was reassessed at the time of study drug continuation unless a tumour assessment had been performed within the previous 4 weeks. BICR assessment of tumour scans was conducted prospectively for patients enrolled in STARTRK-2 and retrospectively for patients in ALKA or STARTRK-1.61,62
Figure 5: Dose Modifications for Entrectinib-Related Adverse Events
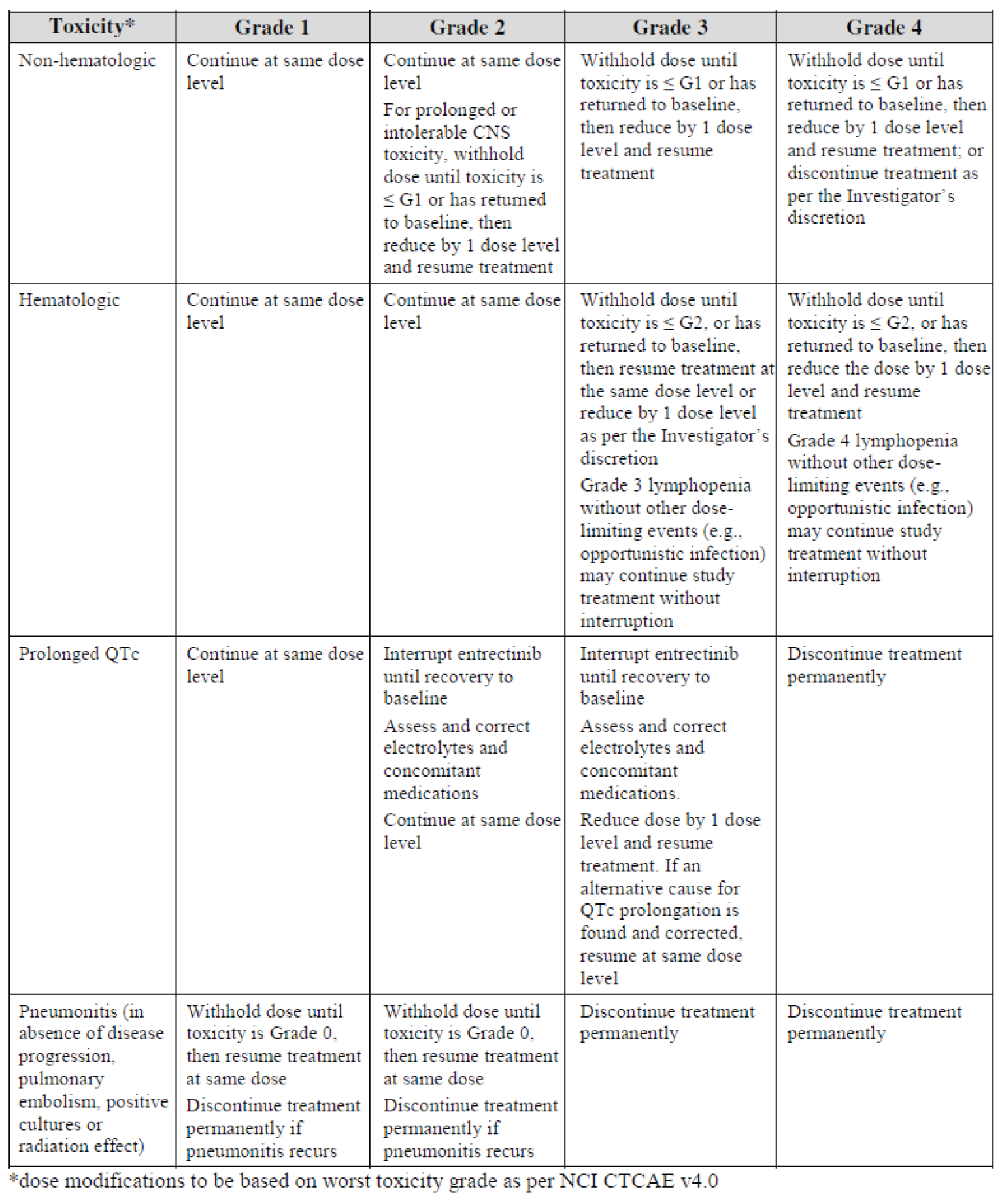
CNS = central nervous system; G = grade of adverse event based on NCI CTCAE v4.0; NCI CTCAE = National Cancer Institute Common Terminology Criteria for Adverse Events.
Source: Clinical Study Report.49
Objective Response Rate
ORR was defined as the proportion of patients with confirmed CR or PR. A confirmed response is a response that persists on repeat imaging greater than or equal to 4 weeks after the initial documentation of the response. Non-responders included:
patients without a confirmed objective response
patients without a post-baseline tumour assessment
patients who received at least 1 dose of entrectinib and discontinued for any reason before undergoing 1 post-baseline response evaluation.62
Duration of Response
DOR (in months) is calculated only for responders (as defined previously). It is measured from the date of first objective response (CR or PR) to the first documentation of radiographic disease progression or the date of death due to any cause, whichever is earlier. For patients without disease progression or death, DOR was censored at the last tumour assessment date.62
Best Overall Response
BOR was the best radiologic overall response recorded from the start of the study treatment until disease progression and was based on RECIST 1.1. A BOR status of CR or PR required confirmation no earlier than 4 weeks from the first response. Stable disease can be assigned only after a patient meets stable disease criteria for at least 5 weeks (i.e., ≥ 35 days) following the first dose of the study treatment. Otherwise, the best response will not be evaluable. Other cases of a best response that are not evaluable include no post-baseline scans available and missing subsets of scans at all time points. Patients with only non-target lesions can be assessed only as CR, non-CR/non-PD, PD, or not evaluable. All other determinations of BOR are specified in the RECIST 1.1 guidelines.62
Clinical Benefit Rate
CBR is the proportion of patients meeting 1 of the following criteria: confirmed CR or confirmed PR; stable disease for no fewer than 6 months following the start of entrectinib. Patients without a post-baseline tumour assessment or patients who received at least 1 dose of entrectinib and discontinue for any reason before undergoing 1 post-baseline response evaluation were counted as not achieving clinical benefit.62
Progression-Free Survival
PFS was defined as time (months) from first dose of entrectinib to first documentation of radiographic disease progression or death due to any cause. PFS data for patients without progression or death were censored on the date of the last tumour assessment (or, if no tumour assessment was performed after the baseline visit, at the date of first dose of entrectinib).62
Time to CNS Progression
Time to CNS progression was defined as time (months) from first dose of entrectinib to first documentation of radiographic CNS disease progression or death due to any cause. Radiographic CNS disease progression is defined as an occurrence of a new CNS lesion or progression in any CNS lesion per RECIST 1.1 criteria. Censoring rules similar to those defined previously were applied: patients without radiographic CNS progression or death were censored on the date of the last tumour assessment. (Note: patients without CNS lesions present at baseline, per investigator assessment, were not required to have scheduled brain scans every 8 weeks.)
Overall Survival
OS was defined as the time (months) from the first dose of entrectinib to the date of death due to any cause. Patients who were alive at the time of the analysis were censored on the last known date that they were alive. In addition, the following censoring rules apply:
Patients with no post-baseline information were censored on the date of first dose of entrectinib.
Patients who were lost to follow-up or withdrew consent for further follow-up were censored on the last known date on which they were alive.62
Intracranial Objective Response Rate
Selecting only CNS lesion(s) (target, non-target, or both, as determined by BICR) for each patient, the RECIST 1.1 algorithms for time point response and BOR assessment were used to determine intracranial response. Patients with confirmed CR or confirmed PR in the CNS lesion(s) were referred to as intracranial responders. A confirmed intracranial response is a CNS response that persists on repeat imaging greater than or equal to 4 weeks after initial documentation of CNS response. This analysis was performed for patients presenting with measurable CNS lesions at baseline as well as for patients with only non-measurable CNS lesions at baseline.62
Intracranial Duration of Response
IC-DOR was calculated only for intracranial responders and is measured from the date of first intracranial response to first documentation of radiographic CNS disease progression or date of death due to any cause, whichever is earlier. For patients without CNS disease progression who have not died within 30 days of the last dose of study treatment, IC-DOR was censored at the last tumour assessment date before any date of subsequent anti-cancer therapy, including surgery or radiotherapy to the brain.62
Intracranial Progression-Free Survival
IC-PFS was defined as the time (months) from the first dose of entrectinib to the first documentation of radiographic CNS disease progression or death due to any cause. Radiographic CNS disease progression is defined as an occurrence of a new CNS lesion or progression in any CNS lesion per RECIST 1.1 criteria. Similar censoring rules, as defined previously in this review, were applied: patients without radiographic CNS progression or death were censored on the date of the last tumour assessment.62
Safety Analyses
Safety analyses were performed on the safety population analysis sets, as previously described. These were performed separately for patients with and without CNS disease at baseline. Summary statistics on AEs, treatment-emergent AEs, serious AEs, dose modifications (reductions and interruptions), discontinuations due to AEs, and deaths were reported.63
Health-Related Quality of Life
Data on patient-reported HRQoL and health status were collected in the STARTRK-2 trial through self-administered questionnaires that included the EORTC QLQ-C30, the EORTC QLQ-LC13, and the EORTC QLQ-CR29.45,64 HRQoL assessments were administered before the first dose of entrectinib at cycle 1 (day 1) and for each subsequent cycle (day 1), as well as at the end of treatment. The EORTC QLQ-C30 is a validated questionnaire that comprises 30 questions to assess global health status (GHS), functioning, and symptoms using multi-item and single-item measures.45 Each domain and item of the QLQ-C30 was linear-transformed to standardize the raw score to a range from 0 to 100. A clinically meaningful change from baseline was defined as an improvement or worsening of greater than or equal to 10 points. A higher score on the functional scales represents a higher and/or healthy level of functioning; similarly, a higher score for GHS represents higher HRQoL. Conversely, a higher score on symptom scales represents higher severity of the symptom. The average scores and mean changes from baseline for each item in the QLQ-C30 were presented in tabular and graphical format.45
Statistical Analysis
Clinical Cut-Off Dates
As shown in Table 13, the sponsor conducted a series of analyses with longer-term follow-ups and increasing sample sizes (largely through the accrual of additional patients in the ongoing STARTRK-2 trial).
Table 13: Integrated Analyses for Entrectinib for Adults With NTRK Fusion Tumours
ECOD | CCOD | N (efficacy) | Application | Role in CADTH review |
|---|---|---|---|---|
July 31, 2019 | August 31, 2020 | N = 121 | Updated analysis used in the current submission to CADTH | Primary dataset used by CADTH |
April 30, 2018 | October 31, 2018 | N = 74 |
| Safety data, subgroup data, and health-related quality of life data that were not reported on, updated as of August 31, 2020 CCOD |
November 30, 2017 | May 31, 2018 | N = 54 |
| Not used (but noted that these are the data reflected in the Canadian product monograph) |
CCOD = clinical cut-off date; ECOD = enrolment cut-off date; EMA = European Medicines Agency; NTRK = neurotrophic tyrosine receptor kinase.
Source: Sponsor’s Clinical Summary.59
Power Calculation
For the integrated analysis, a proportion of responding patients that exceeded 30% was considered to be clinically meaningful; thus, assuming a true ORR of 60%, a sample size of 56 patients was required to yield a 95% CI with plus or minus 14% precision that would exclude a lower limit of 30%.61,62 Based on a summary of treatment options for common tumours that harbour NTRK fusions (including salivary gland cancer, secretory breast cancer, sarcoma, thyroid cancer, CRC, lung cancer, and glioblastoma), it was determined that expected rates of response to later lines of treatment in the refractory setting for metastatic and locally advanced unresectable solid tumours are typically less than 30%, and a median DOR is generally less than 10 months across available approved drugs.
Statistical Analysis
The primary efficacy outcomes were ORR and DOR, as per the BICR assessment. Secondary outcomes included CBR, PFS, time to CNS progression, and OS. Additional secondary outcomes focused on patients with CNS metastases at baseline and included IC-ORR, IC-DOR, and IC-PFS.
BICR-assessed ORR, CBR, and IC-ORR were presented as an efficacy summary (number and proportion of patients with confirmed CR or PR) and corresponding 2-sided 95% Clopper-Pearson exact CIs.45 For time-to-event end points, which included DOR, PFS, OS, IC-DOR, and IC-PFS, the median, 25th and 75th percentiles were presented using the Kaplan–Meier method, and 2-sided 95% CIs were calculated using the Brookmeyer and Crowley (1982) and Klein and Moeschberger (1997) methods. For DOR and PFS, landmark analyses at 6 months, 9 months, and 12 months with corresponding 95% CIs were also calculated using the Kalbfleisch and Prentice method (1980).
Formal statistical significance testing was not performed; thus, no P values were reported. Point estimates with 95% CIs were reported to estimate the magnitude of treatment effect. The sponsor reported that no statistical adjustments for multiplicity were made due to the rarity of the patient population and expectation of significant clinical benefit; and no statistical adjustments were made to account for subgroup effects associated with the pooling of data in the analysis.61,62 Pre-specified sensitivity analyses to evaluate the robustness of therapeutic efficacy included ORR and DOR for the enrolled population (i.e., the analyses were not limited to patients who were NTRK fusion-gene positive), as well as ORR and DOR, including the NTRK efficacy non-evaluable analysis set.
Subgroup Analyses
Subgroup analyses for BICR-assessed ORR by sex, age group, ECOG PS, any prior systemic therapy, number of prior systemic therapies, and tumour histology were pre-specified; graphic displays of efficacy end points (waterfall and swimmer plots) were provided to depict each patient’s best tumour response and time on study, including time to first objective response and DOR. Additional subgroups were identified for analyses, but were not reported.
Analysis Populations
The analysis sets used in the integrated evaluations efficacy and safety are summarized in Table 15.
Table 14: Statistical Analyses of Efficacy End Points
End point | Statistical model | Adjustment factors | Sensitivity analyses |
|---|---|---|---|
Pooled analysis | |||
ORR | 2-sided 95% exact binomial CIs for the proportion of patients with response were calculated using the Clopper-Pearson method. | None |
|
OS DOR PFS IC-PFS IC-DOR | The Kaplan–Meier method was used to estimate the median, 25th, and 75th percentiles. Two-sided 95% CIs were calculated using the Brookmeyer and Crowly (1982) and Klein and Moeschberger (1997) methods. | None | For DOR and PFS, landmark analyses at 6 months, 9 months, and 12 months were also provided, with the corresponding 2-sided 95% CIs calculated using the Kalbfleisch and Prentice (1980) method. |
CBR | Two-sided 95% exact binomial CIs for the proportion of patients with response were calculated using the Clopper-Pearson method. | None | None |
CBR = clinical benefit rate; CI = confidence interval; DOR = duration of response; ECOG PS = Eastern Cooperative Oncology Group Performance Status; IC = intracranial; NTRK = neurotrophic tyrosine receptor kinase; ORR = overall response rate; OS = overall survival; PFS = progression-free survival.
Source: Statistical Analysis Plan.62
Table 15: Analysis Sets in the Integrated Evaluations of Efficacy and Safety
Analysis set | Description |
|---|---|
NTRK efficacy-evaluable analysis set | Patients with the following characteristics
|
CNS disease analysis set | Patients in the NTRK efficacy-evaluable analysis set with CNS metastases at baseline (presented as both BICR-assessed CNS disease and investigator-assessed CNS disease) |
NTRK safety-evaluable population | Patients with NTRK-positive tumours who received at least 1 dose of entrectinib |
BIRC = blinded independent central review; CNS = central nervous system; NTRK = neurotrophic tyrosine receptor kinase; RECIST 1.1 = Response Evaluation Criteria in Solid Tumours Version 1.1; TRK = tropomyosin receptor kinase.
Source: Statistical Analysis Plan.62
Results
Patient Disposition
Table 16 provides a summary of the patient disposition for those included in the NTRK efficacy-evaluable population (N = 121). ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| Eighty-five patients (70.2%) discontinued treatment with entrectinib. Disease progression (56 out of 85; 65.9%) and AEs (17 out of 85; 20.0%) were the most frequently cited reasons.
Exposure to Study Treatments
Treatment with Entrectinib
Exposure to entrectinib was reported only for the NTRK safety-evaluable population (Table 17). The mean and median durations of therapy were 11.54 months (SD = 11.49) and 8.34 months (interquartile range = 2.73 to 17.25), respectively.11
Table 16: Patient Disposition (NTRK Efficacy-Evaluable Analysis Set; August 2020 CCOD)
Study status | ALKA (N = 1) study | STARTRK-1 study (N = 2) | STARTRK-2 study (N = 118) | Total (N = 121) |
|---|---|---|---|---|
Ongoing | 0 | 0 | 54 (45.8%) | 54 (44.6%) |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
Discontinued treatment | 1 (100.0%) | 2 (100.0%) | 82 (69.5%) | 85 (70.2%) |
Reason for discontinuation | ||||
Due to an AE | 0 | 0 | 17 (20.7%) | 17 (20.0%) |
Informed consent withdrawn | 0 | 0 | 7 (8.5%) | 7 (8.2%) |
Disease progression | 1 (100.0%) | 2 (100.0%) | 53 (64.6%) | 56 (65.9%) |
Other | 0 | 0 | 5 (6.1%) | 5 (5.9%) |
AE = adverse event; CCOD = clinical cut-off date; NTRK = neurotrophic tyrosine receptor kinase.
Source: Sponsor-provided additional information.65
Table 17: Summary of Exposure (NTRK Safety-Evaluable Population; August 2020 CCOD)
Analysis | ALKA-372 to 001 study (N = 1) | STARTRK-1 study (N = 4) | STARTRK-2 study (N = 188) | Integrated analysis (N = 193) |
|---|---|---|---|---|
Number of cycles received | ||||
Mean (Std) | 5.0 (NE) | 11.5 (9.0) | 14.1 (14.1) | 14.0 (13.9) |
Median (IQR) | 5.0 (5.0 to 5.0) | 11.0 (6.0 to 17.0) | 10.0 (4.0 to 20.0) | 10.0 (4.0 to 20.0) |
Total number of doses missed | ||||
Mean (Std) | 1.0 (NE) | 0.0 (0.0) | 4.7 (8.2) | 4.6 (8.1) |
Median (IQR) | 1.0 (1.0 to 1.0) | 0.0 (0.0 to 0.0) | 2.0 (1.0 to 5.0) | 2.0 (1.0 to 5.0) |
Treatment duration (months) | ||||
n | 1 | 4 | 186 | 191 |
Mean (Std) | 3.34 (NE) | 10.17 (7.86) | 11.61 (11.59) | 11.54 (11.49) |
Median (IQR) | 3.34 (3.34 to 3.34) | 9.86 (5.01 to 15.33) | 8.30 (2.73 to 17.25) | 8.34 (2.73 to 17.25) |
CCOD = clinical cut-off date; IQR = interquartile range; NE = not estimable; NTRK = neurotrophic tyrosine receptor kinase; Std = standard deviation.
Source: Supplemental Efficacy Results (clinical cut-off date: August 31, 2020).11
Concomitant Medications
Across all 3 trials included in the efficacy analysis, seizure prophylaxis and non–enzyme-inducing anti‐epileptic drugs were allowed during the study for patients with controlled asymptomatic CNS involvement. Prophylactic use of granulocyte colony-stimulating factor or erythropoietin could be initiated according to American Society of Clinical Oncology guidelines in patients who were having difficulty with severe neutropenia or anemia. Palliative radiotherapy was allowed to specific sites if medically necessary.60,66-68 In STARTRK-1 and STARTRK-2, it was recommended that moderate to strong inhibitors and inducers of cytochrome P450 CYP3A be used with caution, as well as cytochrome P450 substrates.60,68
Subsequent Medications
Following BICR assessment of PD, 6 patients (11.1%) received subsequent anti-cancer treatment, most frequently with systemic drugs (n = 5; 9.3%) and 1 patient received a radiopharmaceutical treatment (Yttrium). Systemic anti-cancer drugs included doxorubicin (n = 2), bevacizumab (n = 1), everolimus (n = 1), irinotecan (n = 1), nivolumab (n = 1), and others. One additional patient received doxorubicin after discontinuation of entrectinib but before PD.69 A total of 18 patients (33.3%) continued treatment with entrectinib beyond the BICR assessment of PD.70
Efficacy
Only those efficacy outcomes and analyses of subgroups identified in the review protocol are reported in this section.
Table 18: Summary of Efficacy End Points (NTRK Efficacy-Evaluable Analysis Set; August 2020 CCOD)
Analysis | Total population (N = 121) | With CNS metastases at baseline (N = 19) |
|---|---|---|
Objective response rate | ||
Responders, n (%) | 74 (61.2) | 12 (63.2) |
ORR (95% CI) | 61.2% (51.87 to 69.88) | 63.2% (38.36 to 83.71) |
Best response, n (%) | ||
CR | 19 (15.7) | 1 (5.3) |
PR | 55 (45.5) | 11 (57.9) |
SD | 13 (10.7) | 4 (21.1) |
PD | 13 (10.7) | 2 (10.5) |
Non-CR or PD | 6 (5.0) | 0 (0.0) |
Missing or unevaluable | 15 (12.4) | 1 (5.3) |
Clinical benefit rate | ||
Patients with event, n (%) | 77 (63.6) | NR |
Clinical benefit rate (95% CI) | 63.6% (54.8 to 71.7) | NR |
Time to response (months) | ||
Median TTR (95% CI) | 1.0 (0.9 to 1.0) | 1.3 (0.9 to 2.8) |
Duration of response (months) | ||
Patients with response, n (%) | 74 (61.2) | 12 (63.2) |
Median DOR (95% CI) | 20.0 (13.0 to 38.2) | 15.2 (6.0 to 29.4) |
Progression-free survival | ||
Patients with event, n (%) | 72 (59.5) | 13 (68.4) |
Median PFS, (95% CI) | 13.8 (10.1 to 19.9) | 11.7 (5.1 to 30.3) |
Overall survival | ||
Deaths, n (%) | 49 (40.5) | 10 (52.6) |
Median OS, (95% CI) | 33.8 (23.4 to 46.4) | 19.9 (7.9 to NE) |
CCOD = clinical cut-off date; CI = confidence interval; CNS = central nervous system; CR = complete response; DOR = duration of response; NE = not estimable; NR = not reported; NTRK = neurotrophic tyrosine receptor kinase; ORR = overall response rate; OS = overall survival; PD = progressive disease; PFS = progression-free survival; PR = partial response; SD = stable disease; TTR = time to response.
Source: Supplemental Efficacy Results (clinical cut-off date: August 31, 2020).11
Objective Response Rate
A summary of the primary and secondary efficacy analyses is shown in Table 18. The ORR by BICR was 61.2% (95% CI, 51.87 to 69.88), which met the sponsor’s predefined threshold for a clinically meaningful ORR, given that the lower limit of the 95% CI excluded 30% (in accordance with the sponsor’s statistical analysis plan).62 A total of 19 patients (17.7%) demonstrated a BOR of CR and 55 patients (45.5%) demonstrated a PR.50 There were 13 patients (10.7%) who had stable disease and 13 patients (10.7%) who had PD. There were 6 patients (5.0%) with non-CR or non-PD (i.e., patients with non-target lesions as assessed by BICR, but measurable disease at baseline as assessed by the investigator) and 15 patients (12.4%) who were missing or non-evaluable. The median times to objective response were 1.0 month (95% CI, 0.9 to 1.0) for the overall population and 1.3 months (95% CI, 0.9 to 2.8) for patients with CNS metastases at baseline.
As shown in Table 19, the point estimates for ORR ranged widely across tumour types, and the CIs reflected a high degree of uncertainty for many tumour types. In light of the small sample sizes and the wide CIs, no meaningful conclusions can be drawn about the efficacy of entrectinib in different types of solid tumours. As noted in CADTH’s review of larotrectinib for NTRK-positive tumours, it is not methodologically meaningful to evaluate the ORR (and by extension, the effectiveness) of TRK inhibitors on different cancer types separately. At least 1 patient demonstrated a response to treatment in each of the tumour types, with the exception of neuroblastoma (n = 1). The ORRs for the larger subgroup populations were generally consistent with the results for the overall population; however, a higher proportion of patients with salivary MASC tumours experienced tumour response (20 patients out of 24 patients [83.3%], 95% CI, 62.6 to 95.3), while a lower proportion of those with colorectal carcinoma experienced tumour response (2 patients out of 10 patients [20%], 95% CI, 2.5 to 55.6). A waterfall plot for the baseline change in tumour sum for all patients is illustrated in Figure 6.
As shown in Table 7, the drug programs that participate in the CADTH reimbursement review process noted that efficacy data based on prior lines of systemic cancer therapy are of particular interest from an implementation perspective. Upon request from CADTH, the sponsor provided subgroup analyses from the updated efficacy dataset (N = 121) based on prior exposure to systemic cancer therapies. The ORR for patients who had not received any prior systemic cancer therapies was 81.1% (95% CI, 65.8 to 90.5); for those who had received 1 prior systemic therapy, it was 57.1% (95% CI, 40.9, 72.0); for 2 prior systemic therapies, 57.7% (95% CI, 38.9, 74.5); for 3 prior systemic therapies, 50.0% (95% CI, 25.4, 74.6); and for 4 prior systemic therapies, it was 42.9% (95% CI, 5.8, 75.0) (summarized in Table 19).32 The sponsor provided a further breakdown, with ORRs separated by tumour type and number of prior systemic therapies (Table 38 on page 125).
Intracranial Objective Response Rate
The intracranial efficacy of entrectinib in patients with baseline CNS metastases is highlighted in Table 20. The BICR-assessed IC-ORRs were 52.6% (95% CI, 28.86 to 75.55) and 63.6% (95% CI, 30.8 to 89.1) for all patients with baseline CNS disease (10 patients out of 19 patients responded) and those with measurable disease at baseline (7 patients out of 11 patients responded), respectively. The sponsor reported the results of a subgroup analysis based on prior brain radiotherapy, and the ORRs were similar between those who had no prior brain radiotherapy or had brain radiotherapy greater than or equal to 6 months before the initiation of treatment (55.6%; 95% CI, 21.2 to 86.3 [n = 9]) versus those who had brain radiotherapy within 6 months of initiating treatment with entrectinib (50.0%; 95% CI, 18.7 to 81.3 [n = 10]).
Table 19: Subgroup Analyses (NTRK Efficacy-Evaluable Analysis Set)
Subgroup | n | Response n (%) | ORR (95% CI) | DOR median, months (95% CI) | PFS median (95% CI) | OS median (95% CI) | |
|---|---|---|---|---|---|---|---|
Subgroup analyses by baseline CNS metastases (August 31, 2020 CCOD; N = 121) | |||||||
With CNS metastases | 19 | 12 (63.2) | 63.2 (38.36 to 83.71) | 15.2 (6.0 to 29.4) | 11.7 (5.1 to 30.3) | 19.9 (7.9 to NE) | |
Without CNS metastases | 102 | 62 (60.8) | 60.8 (50.62 to 70.31) | 29.0 (12.9 to NE) | 13.8 (10.2 to 20.4) | 37.1 (23.9 to NE) | |
Subgroup analyses by tumour type (August 31, 2020 CCOD for ORR and DOR, N = 121; October 31, 2018 CCOD for PFS and OS, N = 74)a | |||||||
Sarcoma | 26 | 15 (57.7) | 57.7 (36.9 to 76.7) | 15.0 (4.6 to NE) | 10.1 (6.5 to 11.2) | 16.8 (10.6 to 20.9) | |
Salivary (MASC) | 24 | 20 (83.3) | 83.3 (62.6 to 95.3) | NE (NE to NE) | NE (7.7 to NE) | NE (NE to NE) | |
NSCLC | 22 | 14 (63.6) | 63.6 (40.7 to 82.8) | 19.9 (10.4 to 29.4) | 14.9 (4.7 to NE) | 14.9 (5.9 to NE) | |
Thyroid cancer | 13 | 7 (53.8) | 53.8 (25.1 to 80.8) | 13.2 (7.9 to NE) | 11.8 (6.5 to NE) | NE (8.7 to NE) | |
Colorectal carcinoma | 10 | 2 (20.0) | 20.0 (2.5 to 55.6) | 17.6 (15.1 to 20.0) | 2.4 (1.0 to 16.0) | 16.0 (2.4 to NE) | |
Breast cancer | 7 | 5 (71.4) | 71.4 (29.0 to 96.3) | 12.9 (4.2 to NE) | 10.1 (5.1 to NE) | 23.9 (5.1 to 23.9) | |
Neuroendocrine tumours | 5 | 2 (40.0) | 40.0 (5.3 to 85.3) | NE (11.1 to NE) | NE (0.9 to NE) | NE (NE to NE) | |
Pancreatic cancer | 4 | 3 (75.0) | 75.0 (19.4 to 99.4) | 12.9 (7.1 to 12.9) | 8.0 (6.2 to 17.5) | 13.4 (11.2 to NE) | |
CUP | 3 | 1 (33.3) | 33.3 (0.8 to 90.6) | 9.1 (NE to NE) | NA | NA | |
Gynecologic | 2 | 1 (50.0) | 50.0 (1.3 to 98.7) | 38.2 (NE to NE) | NE (13.7 to NE) | NE (NE to NE) | |
Head and neck | 2 | 2 (100.0) | 100.0 (15.8 to 100.0) | NE (16.9 to NE) | NA | NA | |
Cholangiocarcinoma | 1 | 1 (100.0) | 100.0 (2.5 to 100.0) | 9.3 (NE to NE) | 12.0 (NE to NE) | NE (NE to NE) | |
Upper GI adenocarcinoma | 1 | 1 (100.0) | 100.0 (2.5 to 100.0) | 29.0 (NE to NE) | NE (NE to NE) | NE (NE to NE) | |
Neuroblastoma | 1 | 0 (NA) | 0 (NA) | NA | 0.1 (NE to NE) | 0.1 (NE to NE) | |
Subgroup analyses by ECOG PS (October 31, 2018 CCOD; N = 74) | |||||||
ECOG PS | 0 | 30 | 20 (66.7) | 66.7 (47.19 to 82.71) | NR | NR | NR |
1 | 34 | 22 (64.7) | 64.7 (46.49 to 80.25) | NR | NR | NR | |
2 | 10 | 5 (50.0) | 50.0 (18.71 to 81.29) | NR | NR | NR | |
Subgroup analyses by prior systemic therapies (August 30, 2022 CCOD; N = 121) | |||||||
Number of prior systemic therapies | 0 | 37 | 30 | 81.1 (65.8 to 90.5) | NR | NR | NR |
1 | 35 | 20 | 57.1 (40.9 to 72.0) | NR | NR | NR | |
2 | 26 | 15 | 57.7 (38.9 to 74.5) | NR | NR | NR | |
3 | 12 | 6 | 50.0 (25.4 to 74.6) | NR | NR | NR | |
4 | 7 | 3 | 42.9 (15.8 to 75.0) | NR | NR | NR | |
5 | 3 | 0 | 0.0 (0.0 to 56.1) | NR | NR | NR | |
6 | 1 | 0 | 0.0 (0.0 to 79.3) | NR | NR | NR | |
CCOD = clinical cut-off date; CI = confidence interval; CNS = central nervous system; CUP = carcinoma of unknown primary; DOR = duration of response; ECOG PS = Eastern Cooperative Oncology Group Performance Status; GI = gastrointestinal; MASC = mammary analogue secretory carcinoma; NA = not applicable; NE = not estimable; NR = not reported; NSCLC = non–small cell lung carcinoma; NTRK = neurotrophic tyrosine receptor kinase; ORR = objective response rate; OS = overall survival; PFS = progression-free survival.
aThe subgroup analyses for the October 31, 2018 CCOD included the following: sarcoma (n = 16); NSCLC (n = 1); salivary (MASC) (n = 1); thyroid cancer (n = 7); colorectal carcinoma (n = 7); breast cancer (n = 6); neuroendocrine tumours (n = 4); pancreatic cancer (n = 3); gynecologic (n = 2); cholangiocarcinoma (n = 1); upper GI tract adenocarcinoma (n = 1); neuroblastoma (n = 1).
Source: Demetri et al. (2022)51; drug reimbursement review sponsor submission50; sponsor-provided additional information.32
Figure 6: Waterfall Plot of Best Percentage Change From Baseline in Tumour Sum as per BICR Assessment (NTRK Efficacy-Evaluable Analysis Set; CCOD: August 2020; N = 121)
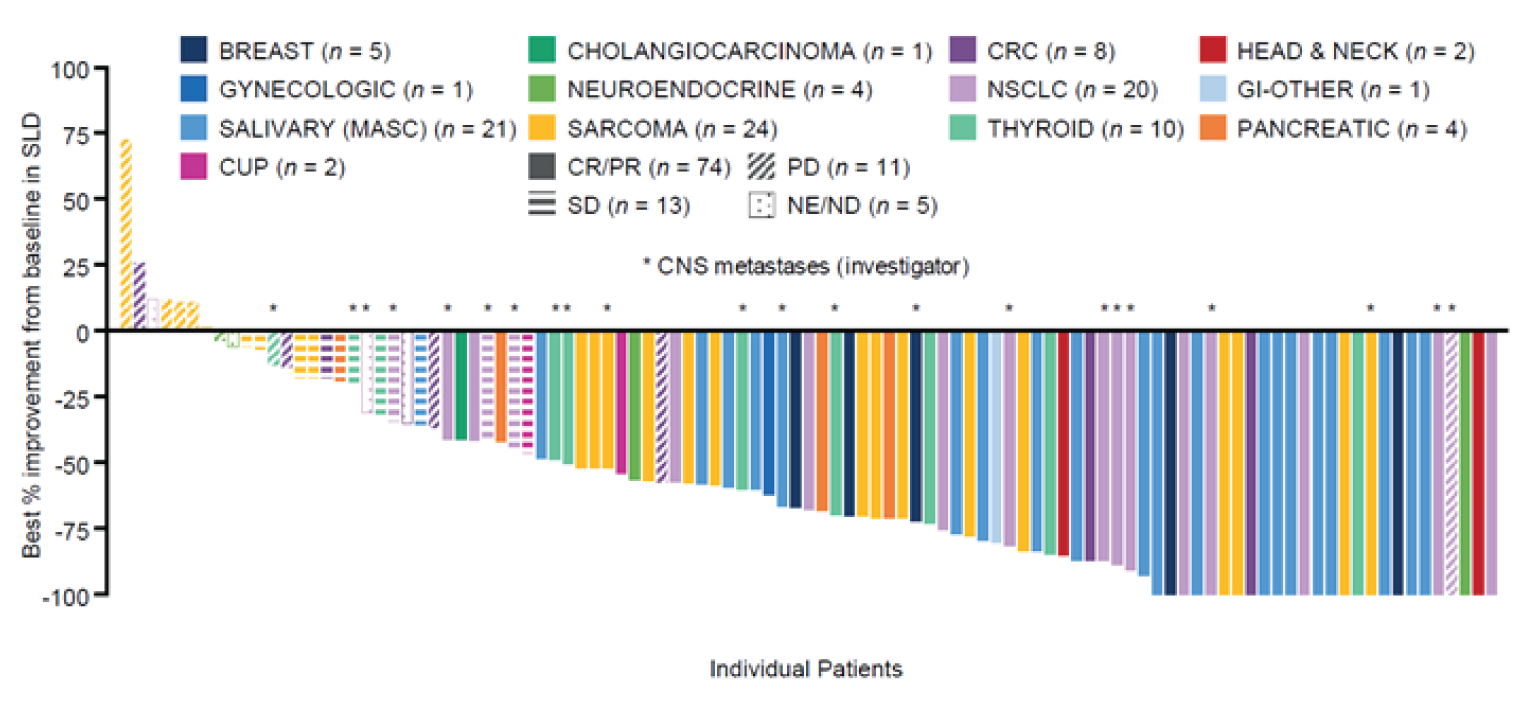
BICR = blinded independent central review; CCOD = clinical cut-off date; CNS = central nervous system; CR = complete response; CRC = colorectal cancer; CUP = carcinoma of unknown primary; GI = gastrointestinal; MASC = mammary analogue secretory carcinoma; ND = not determined; NE = not estimable; NSCLC = non–small cell lung cancer; NTRK = neurotrophic tyrosine receptor kinase; PD = progressive disease; PR = partial response; SD = stable disease; SLD = sum of the longest diameter.
Source: Drug reimbursement review sponsor submission.50
Table 20: Intracranial Efficacy (BICR-Assessed) in Patients With NTRK Fusion–Positive Solid Tumours and BICR-Assessed CNS Metastases at Baseline
Efficacy parameter | Patients with CNS metastases at baseline (BICR-assessed) | |
|---|---|---|
Measurable disease (n = 11) | All patients (N = 19) | |
Intracranial objective response rate | ||
Patients with response, n (%) | 7 (63.6) | 10 (52.6) |
(95% CI) | (30.8 to 89.1) | (28.9 to 75.6) |
Best response, n (%) | ||
CR | 3 (27.3) | 6 (31.6) |
PR | 4 (36.4) | 4 (21.1) |
SD | 2 (18.2) | 2 (10.5) |
PD | 1 (9.1) | 1 (5.3) |
Non-CR or non-PD | 0 (0.0) | 5 (26.3) |
Missing or unevaluable | 1 (9.1) | 1 (5.3) |
Duration of intracranial response | n = 7 | n = 10 |
Patients with event, n (%) | 4 (57.1) | 6 (60.0) |
Median, months (95% CI) | 22.1 (7.4 to NE) | 17.2 (7.4 to NE) |
Intracranial progression-free survival | ||
Patients with event, n (%) | 6 (54.5) | 13 (68.4) |
Median, months (95% CI) | 19.9 (5.9 to NE) | 10.1 (6.3 to 26.7) |
BICR = blinded independent central review; CI = confidence interval; CNS = central nervous system; CR = complete response; NE = not estimable; NTRK = neurotrophic tyrosine receptor kinase; PD = progressive disease; PR = partial response; SD = stable disease.
Source: Supplemental Efficacy Results (clinical cut-off date: August 31, 2020).11
Figure 7: Kaplan–Meier Plots for DOR and Intracranial DOR (CCOD: August 2020)
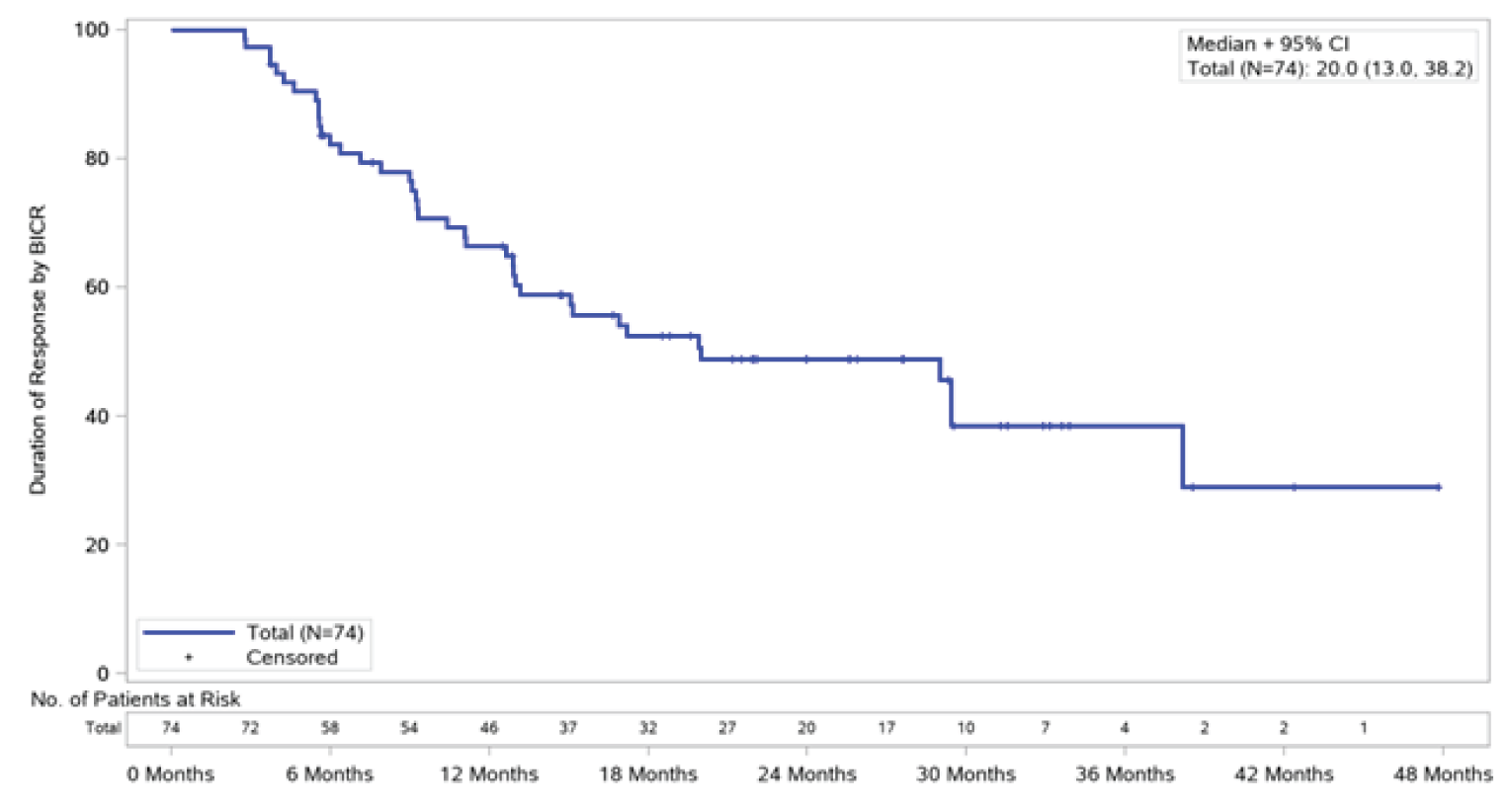
BICR = blinded independent central review; CCOD = clinical cut-off date; CI = confidence interval; DOR = duration of response; No. = number.
Source: Supplemental Efficacy Results (clinical cut-off date: August 31, 2020).11
Duration of Response
The DOR among responders was 20.0 months (95% CI, 13.0 to 38.2) at the August 31, 2020 data cut-off. For the patients who demonstrated a CR or PR with entrectinib, responses of at least 6 months, 12 months, 18 months, 24 months, 30 months, and 36 months were reported for 58 patients (78%), 46 patients (62%), 32 patients (43%), 20 patients (27%), 10 patients (14%), and 4 patients (5%). The event-free probabilities were 0.82 (95% CI, 0.73 to 0.91) at 6 months, 0.66 (95% CI, 0.55 to 0.77) at 12 months, 0.49 (95% CI, 0.37 to 0.61) at 24 months, and 0.39 (95% CI, 0.24 to 0.53) at 36 months.
Intracranial Duration of Response
The IC-DORs among responders were 17.2 months (95% CI, 7.4 to NE) and 22.1 months (95% CI, 7.4 to NE) for all patients with baseline CNS disease and those with measurable disease at baseline, respectively.
Figure 8: Kaplan–Meier Plots for DOR and Intracranial DOR (CCOD: August 2020)
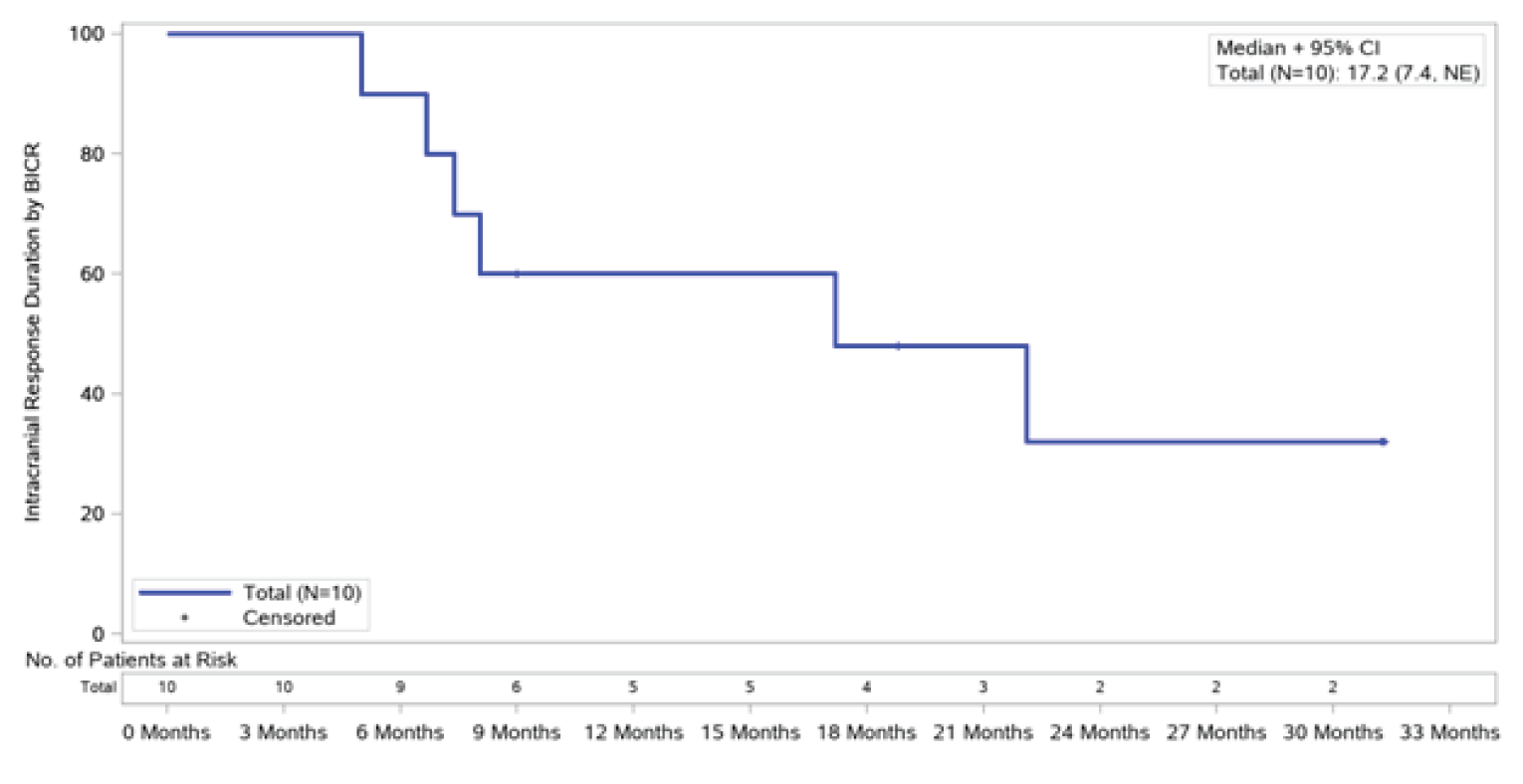
BICR = blinded independent central review; CCOD = clinical cut-off date; CI = confidence interval; DOR = duration of response; NE = not estimable; No. = number.
Source: Supplemental Efficacy Results (clinical cut-off date: August 31, 2020).11
Table 21: DOR, PFS, and OS for Overall Efficacy Population and Patients With Baseline CNS Metastases
Analyses | DOR | PFS | OS | ||||
|---|---|---|---|---|---|---|---|
Overall efficacy population (N = 74) | Baseline CNS metastases (N = 10) | Overall efficacy population (N = 121) | CNS metastases at baseline (N = 19) | Overall efficacy population (N = 121) | Baseline CNS metastases (N = 19) | ||
Time to event | Median (95% CI) | 20.0 (13.0 to 38.2) | 17.2 (7.4 to NE) | 13.8 (10.1 to 19.9) | 10.1 (6.3 to 26.7) | 33.8 (23.4 to 46.4) | 19.9 (7.9 to NE) |
6 months | Remaining at risk (n) | 58 | 9 | 81 | 12 | 99 | 13 |
Event-free probability | 0.82 (0.73 to 0.91) | 0.90 (0.71 to 1.00) | 0.74 (0.66 to 0.82) | 0.71 (0.50 to 0.93) | 0.88 (0.82 to 0.94) | 0.77 (0.57 to 0.97) | |
9 months | Remaining at risk (n) | 54 | 6 | 64 | 9 | 94 | 11 |
Event-free probability | 0.78 (0.68 to 0.87) | 0.60 (0.30 to 0.90) | 0.62 (0.52 to 0.71) | 0.53 (0.30 to 0.77) | 0.85 (0.79 to 0.92) | 0.65 (0.43 to 0.88) | |
12 months | Remaining at risk (n) | 46 | 5 | 55 | 7 | 86 | 11 |
Event-free probability | 0.66 (0.55 to 0.77) | 0.60 (0.30 to 0.90) | 0.53 (0.43 to 0.62) | 0.48 (0.24 to 0.71) | 0.81 (0.74 to 0.89) | 0.65 (0.43 to 0.88) | |
18 months | Remaining at risk (n) | 32 | 4 | 37 | 6 | 59 | 8 |
Event-free probability | 0.52 (0.41 to 0.64) | 0.48 (0.16 to 0.80) | 0.41 (0.32 to 0.51) | 0.41 (0.17 to 0.65) | 0.68 (0.59 to 0.76) | 0.52 (0.28 to 0.76) | |
24 months | Remaining at risk (n) | 20 | 2 | 27 | 4 | 37 | 6 |
Event-free probability | 0.49 (0.37 to 0.61) | 0.32 (0.00 to 0.65) | 0.38 (0.28 to 0.47) | 0.33 (0.09 to 0.56) | 0.58 (0.48 to 0.68) | 0.46 (0.21 to 0.70) | |
30 months | Remaining at risk (n) | 10 | 2 | 18 | 3 | 26 | 5 |
Event-free probability | 0.39 (0.24 to 0.53) | 0.32 (0.00 to 0.65) | 0.36 (0.26 to 0.46) | 0.24 (0.02 to 0.47) | 0.56 (0.46 to 0.67) | 0.46 (0.21 to 0.70) | |
36 months | Remaining at risk (n) | 4 | NA | 6 | NA | 16 | 3 |
Event-free probability | 0.39 (0.24 to 0.53) | NA | 0.27 (0.16 to 0.38) | NA | 0.49 (0.37 to 0.61) | 0.36 (0.11 to 0.62) | |
48 months | Remaining at risk (n) | NA | NA | 1 | NA | 1 | NA |
Event-free probability | NA | NA | 0.18 (0.02 to 0.34) | NA | 0.20 (0.00 to 0.48) | NA | |
CI = confidence interval; CNS = central nervous system; DOR = duration of response; NA = not applicable; NE = not estimable; OS = overall survival; PFS = progression-free survival.
Source: Supplemental Efficacy Results (clinical cut-off date: August 31, 2020).11
Figure 9: Kaplan–Meier Plots for Intracranial Progression-Free Survival (CCOD: August 2020)
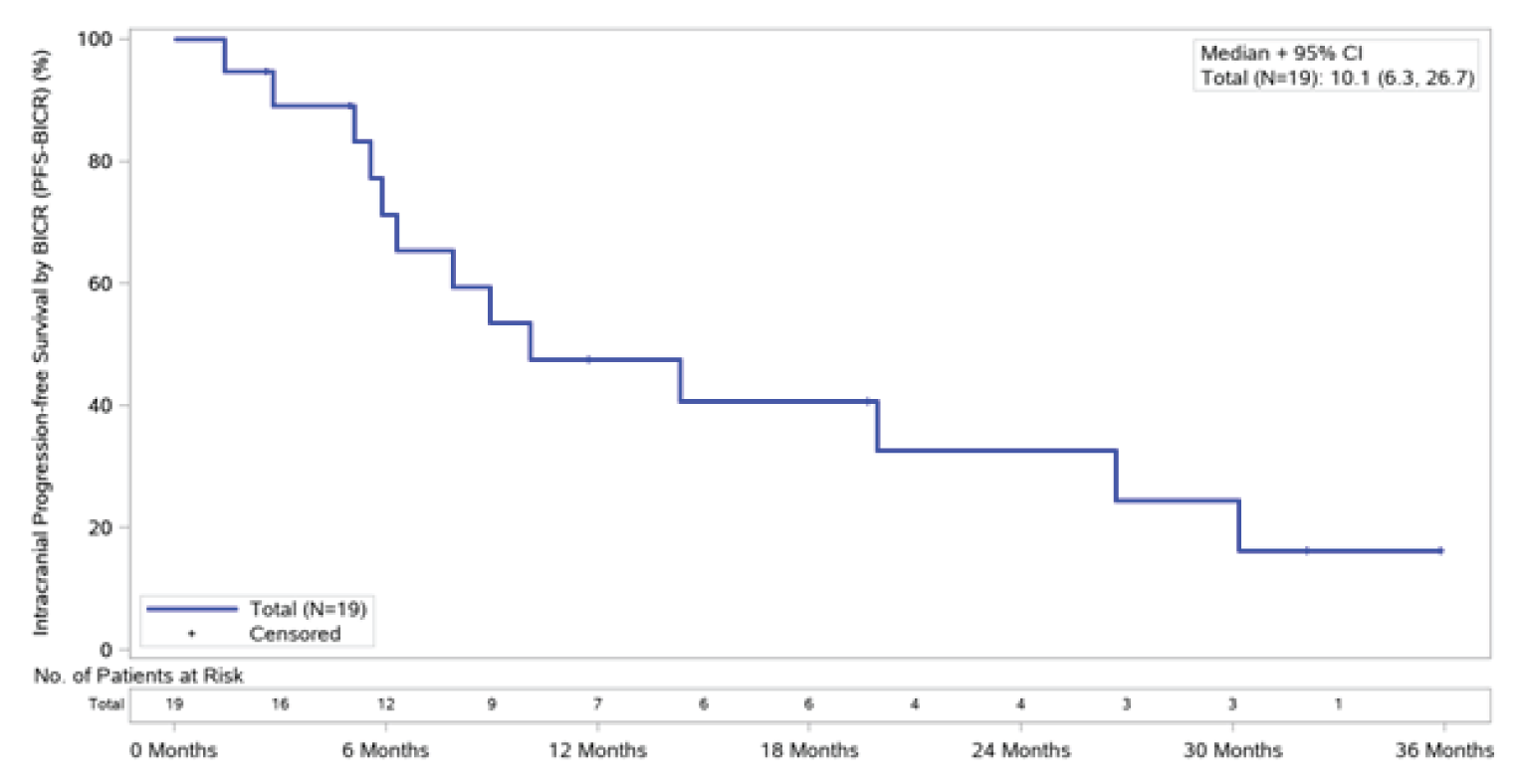
BICR = blinded independent central review; CCOD = clinical cut-off date; CI = confidence interval; No. = number.
Source: Supplemental Efficacy Results (clinical cut-off date: August 31, 2020).11
Overall Survival
A total of 49 patients (40.5%) had died by the time of the August 2020 CCOD. The median OS was 33.8 months (95% CI, 23.4 to 46.4). A total of 10 patients (50.2%) with baseline CNS metastases had died by the time of the August 2020 CCOD. The median OS was 19.9 months (95% CI, 7.9 to NE). The OS event-free rates from 6 months through 48 months are summarized in Table 21. Updated subgroup analyses for OS were not reported for the August 31, 2020 CCOD72; therefore, the results for the October 2018 CCOD are reported in Table 19.
The EORTC QLQ-C30
Results for the QLQ-C30 from August 2020 (N = 110) are summarized in Table 22. Study STARTRK-2 evaluated patient-reported outcomes (PROs) of the treatment impact on functioning and HRQoL based on the QLQ-C30. A change from baseline of greater than or equal to 10 points on a scale of 1 to 100 was considered clinically meaningful by the sponsor, with higher scores reflecting better functioning and HRQoL. The sponsor reported that cycle 10 was selected as the last time point providing representative information, noting that by then, less than 50% of the PRO evaluable population was still in the STARTRK-2 study. At baseline, patients reported moderate to high baseline values on HRQoL and functioning scales. Mean scores at baseline were 65.4 (SD = 23.3) for GHS; 66.8 (SD = 33.9) for role functioning; 75.9 (SD = 25.2) for physical functioning; and 82.9 (SD = 19.6) for cognitive functioning. Based on the October 31, 2018 CCOD, the product monograph reports that from cycle 2 to cycle 10, 20% to 31% of patients met the criteria for a clinically meaningful improvement in GHS, while 25% to 41% of patients met the criteria for a clinically meaningful worsening. Across the functioning scales, 19% to 48% of patients met the criteria for a clinically meaningful improvement in role and physical functioning, while 33 to 46% of patients met the criteria for a clinically meaningful worsening in cognitive functioning.
Figure 10: Kaplan–Meier Plots for OS for Overall Population and Patients with Baseline CNS Metastases (CCOD: August 2020)
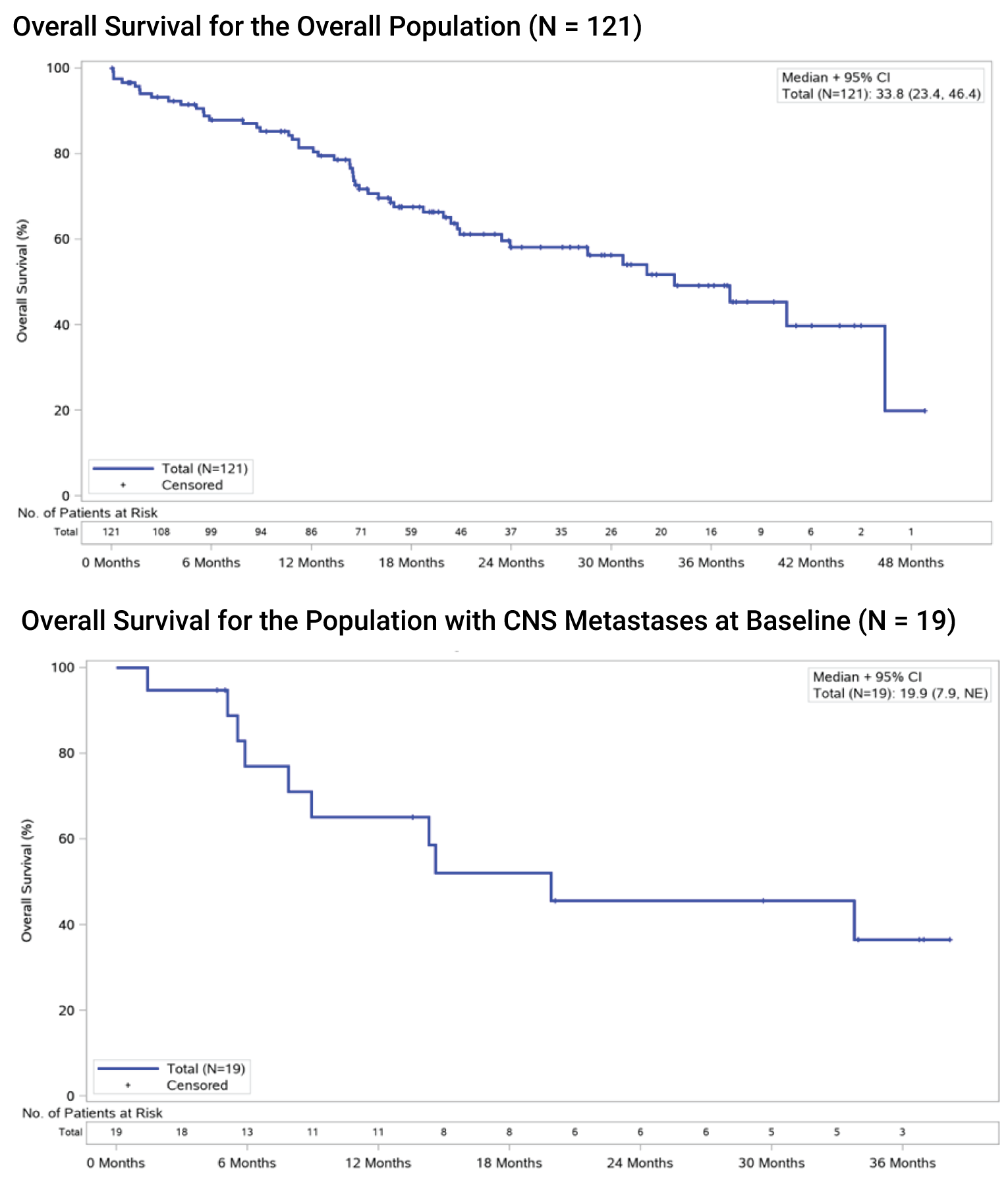
CCOD = clinical cut-off date; CI = confidence interval; CNS = central nervous system; NE = not estimable; No. = number; OS = overall survival.
Source: Supplemental Efficacy Results (clinical cut-off date: August 31, 2020).11
The EORTC QLQ-C29
Updated results for the EORTC QLQ-C29 were not available for the updated August 2020 CCOD; therefore, the results from the October 2018 CCOD are summarized in Table 22. The ability to interpret changes from baseline for these questionnaires is limited by the small number of patients who completed questionnaires initially and the rate of patient drop-off at later cycles. The sponsor reported that among the 7 mCRC patients, baseline results for the QLQ-CR29 showed low abdominal symptom burden (abdominal pain [mean = 27.8], bloating [mean = 33.3], and stool frequency [mean = 13.9]). The scores were variable throughout the study and followed no trend, but remained low overall at most study visits throughout the study.59
The EORTC QLQ-LC13
Updated results for the EORTC QLQ-LC13 were not available for the updated August 2020 CCOD; therefore, the results from the October 2018 CCOD are summarized in Table 22. The ability to interpret changes from baseline for these questionnaires is limited by the small number of patients who completed questionnaires initially and the rate of patient drop-off at later cycles. The sponsor reported that patients with NSCLC (n = 12) showed moderate lung symptom burden at baseline (chest pain [mean = 5.6], coughing [mean = 38.9], and dyspnea [mean score = 26.9]) and at most study visits throughout the study.59
Table 22: EORTC QLQ-C30, QLQ-C29, and QLQ-LC13 (STARTRK-2)
Scale element | Parameter | Baseline | End of treatment change from baseline |
|---|---|---|---|
EORTC QLQ-C30 (August 31, 2020 CCOD) | |||
Global health status (revised) | n | 110 | 44 |
Mean (Std) | 65.38 (23.25) | –6.63 (27.35) | |
Median (IQR) | 66.67 (50.00 to 83.33) | –4.17 (–16.67 to 16.67) | |
Cognitive functioning | n | 110 | 44 |
Mean (Std) | 82.88 (19.61) | –0.76 (17.22) | |
Median (IQR) | 83.33 (66.67 to 100.00) | 0.00 (0.00 to 0.00) | |
Emotional functioning | n | 110 | 44 |
Mean (Std) | 75.91 (22.53) | 0.38 (19.27) | |
Median (IQR) | 83.33 (66.67 to 91.67) | 0.00 (–8.33 to 12.50) | |
Physical functioning (revised) | n | 110 | 44 |
Mean (Std) | 75.94 (25.22) | –9.70 (27.49) | |
Median (IQR) | 80.00 (60.00 to 100.00) | –6.67 (–26.67 to 6.67) | |
Role functioning (revised) | n | 110 | 43 |
Mean (Std) | 66.82 (33.90) | –1.55 (30.61) | |
Median (IQR) | 66.67 (33.33 to 100.00) | 0.00 (–16.67 to 16.67) | |
Social functioning | n | 110 | 44 |
Mean (Std) | 73.79 (27.51) | –4.55 (29.72) | |
Median (IQR) | 83.33 (66.67 to 100.00) | 0.00 (–16.67 to 0.00) | |
Appetite loss | n | 110 | 44 |
Mean (Std) | 23.33 (30.14) | –6.82 (32.61) | |
Median (IQR) | 0.00 (0.00 to 33.33) | 0.00 (–33.33 to 0.00) | |
Constipation | n | 110 | 44 |
Mean (Std) | 13.94 (24.47) | 6.06 (29.00) | |
Median (IQR) | 0.00 (0.00 to 33.33) | 0.00 (0.00 to 16.67) | |
Diarrhea | n | 110 | 44 |
Mean (Std) | 9.09 (19.11) | 11.36 (33.68) | |
Median (IQR) | 0.00 (0.00 to 0.00) | 0.00 (0.00 to 33.33) | |
Dyspnea | n | 110 | 43 |
Mean (Std) | 23.03 (31.85) | 11.63 (34.03) | |
Median (IQR) | 0.00 (0.00 to 33.33) | 0.00 (0.00 to 33.33) | |
Fatigue | n | 110 | 43 |
Mean (Std) | 35.86 (28.70) | –3.88 (32.16) | |
Median (IQR) | 33.33 (11.11 to 55.56) | 0.00 (–22.22 to 22.22) | |
Financial difficulties | n | 110 | 44 |
Mean (Std) | 19.09 (26.89) | 2.27 (23.18) | |
Median (IQR) | 0.00 (0.00 to 33.33) | 0.00 (0.00 to 0.00) | |
Nausea and vomiting | n | 110 | 44 |
Mean (Std) | 8.48 (16.40) | 0.76 (25.15) | |
Median (IQR) | 0.00 (0.00 to 16.67) | 0.00 (–8.33 to 16.67) | |
Pain | n | 110 | 44 |
Mean (Std) | 29.55 (30.60) | –4.55 (29.50) | |
Median (IQR) | 16.67 (0.00 to 50.00) | 0.00 (–25.00 to 16.67) | |
Insomnia | n | 110 | 43 |
Mean (Std) | 29.70 (30.74) | –13.95 (30.20) | |
Median (IQR) | 33.33 (0.00 to 33.33) | 0.00 (–33.33 to 0.00) | |
EORTC QLQ-C29 (STARTRK-2; October 2018 CCOD; N = 7) | |||
Abdominal pain | n | 6 | 2 |
Mean (Std) | 27.78 (38.97) | –16.67 (23.57) | |
Median (IQR) | 16.67 (0.00 to 33.33) | –16.67 (–33.33 to 0.00) | |
Bloating | n | 6 | 2 |
Mean (Std) | 33.33 (29.81) | –33.33 (47.14) | |
Median (IQR) | 33.33 (0.00 to 66.67) | –33.33 (–66.67 to 0.00) | |
Stool frequency | n | 6 | 2 |
Mean (Std) | 13.89 (16.39) | 8.33 (35.36) | |
Median (IQR) | 8.33 (0.00 to 33.33) | 8.33 (–16.67 to 33.33) | |
EORTC QLQ-LC13 (STARTRK-2; October 2018 CCOD; N = 12) | |||
Coughing | n | 12 | 2 |
Mean (Std) | 38.89 (34.33) | 33.33 (47.14) | |
Median | 33.33 (16.67 to 50.00) | 33.33 (0.00 to 66.67) | |
Dyspnea | n | 12 | 2 |
Mean (Std) | 26.85 (31.59) | 44.44 (62.85) | |
Median (IQR) | 11.11 (5.56 to 44.44) | 44.44 (0.00 to 88.89) | |
Pain in arm or shoulder | n | 12 | 2 |
Mean (Std) | 27.78 (31.25) | 0.00 (0.00) | |
Median (IQR) | 33.33 (0.00 to 33.33) | 0.00 (0.00 to 0.00) | |
Pain in chest | n | 12 | 2 |
Mean (Std) | 5.56 (12.97) | 16.67 (23.57) | |
Median (IQR) | 0.00 (0.00 to 0.00) | 16.67 (0.00 to 33.33) | |
Pain in other parts | n | 12 | 2 |
Mean (Std) | 33.33 (28.43) | 0.00 (47.14) | |
Median (IQR) | 33.33 (16.67 to 33.33) | 0.00 (–33.33 to 33.33) | |
CCOD = clinical cut-off date; EORTC = European Organisation for Research and Treatment of Cancer; IQR = interquartile range; QLQ-C30 = Quality of Life Questionnaire Core 30; QLQ-CR29 = Quality of Life Questionnaire Colorectal Cancer 29; QLQ-LC13 = Quality of Life Questionnaire Lung Cancer 13; QoL = quality of life; Std = standard deviation.
Source: Supplementary Results Report (clinical cut-off date: 31 October 2018).50
Harms
Table 23 provides a summary of AEs reported in the safety-evaluable population (N = 193). Nearly all patients who were NTRK fusion–positive experienced at least 1 AE (99.5%); ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||11
Adverse events
Table 24 provides a summary of AEs reported in the safety-evaluable population (N = 193).
Serious Adverse Events
Table 25 provides a detailed summary of the ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.
Withdrawals Due to Adverse Events
Table 26 provides a detailed summary of withdrawals due to AEs that were reported for adult patients in the NTRK safety-evaluable population for the August 31, 2020 CCOD. The overall proportion of patients who withdrew from the study treatment as a result of AEs was |||||||.
Mortality
Deaths in the NTRK safety-evaluable population from the October 31, 2018 CCOD (N = 113) are summarized in Table 27. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||73||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||46
Adverse Events of Special Interest
AEs of special interest identified by the sponsor included adverse CNS events, weight changes, congestive heart failure, increases in blood creatinine, QTc prolongation, vision disorders, hematologic events, hepatotoxicity, and pneumonitis events. In addition to these, Health Canada noted that skeletal fractures and hyperuricemia were of special interest in its review of entrectinib. Table 28 provides a summary of the AEs of special interest based on a pooled dataset of 355 patients.
Table 23: Summary of Adverse Events (NTRK Safety-Evaluable Population; August 2020 CCOD; N = 193)
Adverse events, n (%) | NTRK safety-evaluable patients (N = 193) |
|---|---|
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
AE = adverse event; CCOD = clinical cut-off date; NCI CTCAE = National Cancer Institute Common Terminology Criteria for Adverse Events; NTRK = neurotrophic tyrosine receptor kinase; SAE = serious adverse event.
Source: Supplemental Efficacy Results (clinical cut-off date: August 31, 2020).11
Table 24: Treatment-Emergent Adverse Events Reported in ≥ 10% of Patients or With ≥ 1 Grade 3 or Grade 4 Event (NTRK Safety-Evaluable Population; August 2020 CCOD)
MedDRA system organ class preferred term, n (%)a | NTRK safety-evaluable population (N = 193) | |||
|---|---|---|---|---|
Grade 1 | Grade 2 | Grade 3 | Grade 4 | |
Nervous system disorders | ||||
Dysgeusia | 57 (29.5) | 14 (7.3) | 0 | 0 |
Dizziness | 45 (23.3) | 18 (9.3) | 6 (3.1) | 0 |
Headache | 20 (10.4) | 6 (3.1) | 3 (1.6) | 0 |
Paresthesia | 23 (11.9) | 5 (2.6) | 0 | 0 |
Peripheral sensory neuropathy | 11 (5.7) | 5 (2.6) | 1 (0.5) | 0 |
Cognitive disorder | 5 (2.6) | 4 (2.1) | 3 (1.6) | 0 |
Syncope | 2 (1.0) | 2 (1.0) | 7 (3.6) | 0 |
Somnolence | 4 (2.1) | 3 (1.6) | 1 (0.5) | 0 |
Tremor | 4 (2.1) | 0 | 1 (0.5) | 0 |
Seizure | 0 | 2 (1.0) | 1 (0.5) | 0 |
Cerebrovascular accident | 0 | 0 | 1 (0.5) | 0 |
Hydrocephalus | 1 (0.5) | 0 | 1 (0.5) | 0 |
Spinal cord compression | 0 | 0 | 1 (0.5) | 1 (0.5) |
Depressed Level of consciousness | 0 | 0 | 1 (0.5) | 0 |
Encephalopathy | 0 | 0 | 1 (0.5) | 0 |
Generalized tonic-clonic seizure | 0 | 0 | 1 (0.5) | 0 |
Ischemic stroke | 0 | 0 | 1 (0.5) | 0 |
Thalamic infarction | 0 | 0 | 1 (0.5) | 0 |
Gastrointestinal disorders | ||||
Diarrhea | 48 (24.9) | 26 (13.5) | 5 (2.6) | 0 |
Constipation | 53 (27.5) | 23 (11.9) | 0 | 0 |
Nausea | 43 (22.3) | 11 (5.7) | 2 (1.0) | 0 |
Vomiting | 24 (12.4) | 10 (5.2) | 2 (1.0) | 0 |
Abdominal pain | 21 (10.9) | 5 (2.6) | 0 | 0 |
Dysphagia | 15 (7.8) | 1 (0.5) | 2 (1.0) | 0 |
Colitis | 0 | 0 | 1 (0.5) | 0 |
Small intestinal obstruction | 0 | 0 | 1 (0.5) | 0 |
General disorders and administration-site conditions | ||||
Fatigue | 25 (13.0) | 29 (15.0) | 12 (6.2) | 0 |
Edema peripheral | 41 (21.2) | 16 (8.3) | 2 (1.0) | 0 |
Pyrexia | 23 (11.9) | 4 (2.1) | 0 | 0 |
Asthenia | 9 (4.7) | 4 (2.1) | 4 (2.1) | 0 |
Pain | 8 (4.1) | 2 (1.0) | 1 (0.5) | 0 |
Edema | 6 (3.1) | 2 (1.0) | 2 (1.0) | 0 |
Non-cardiac chest pain | 1 (0.5) | 2 (1.0) | 2 (1.0) | 0 |
Localized Edema | 1 (0.5) | 1 (0.5) | 1 (0.5) | 0 |
General physical health deterioration | 0 | 0 | 2 (1.0) | 0 |
Investigations | ||||
Weight increased | 29 (15.0) | 18 (9.3) | 20 (10.4) | 0 |
Blood creatinine increased | 39 (20.2) | 22 (11.4) | 3 (1.6) | 0 |
ALT increased | 31 (16.1) | 6 (3.1) | 6 (3.1) | 1 (0.5) |
AST increased | 26 (13.5) | 9 (4.7) | 5 (2.6) | 1 (0.5) |
Neutrophil count decreased | 5 (2.6) | 6 (3.1) | 5 (2.6) | 0 |
WBC count decreased | 9 (4.7) | 5 (2.6) | 2 (1.0) | 0 |
Blood alkaline phosphatase increased | 12 (6.2) | 2 (1.0) | 1 (0.5) | 0 |
Blood uric acid increased | 9 (4.7) | 0 | 0 | 1 (0.5) |
Lymphocyte count decreased | 2 (1.0) | 3 (1.6) | 1 (0.5) | 1 (0.5) |
Hemoglobin decreased | 3 (1.6) | 0 | 1 (0.5) | 0 |
Electrocardiogram QT prolonged | 2 (1.0) | 0 | 1 (0.5) | 0 |
Ejection fraction decreased | 0 | 0 | 1 (0.5) | 0 |
Musculoskeletal and connective tissue disorders | ||||
Myalgia | 24 (12.4) | 9 (4.7) | 2 (1.0) | 0 |
Arthralgia | 19 (9.8) | 9 (4.7) | 1 (0.5) | 0 |
Pain in extremity | 23 (11.9) | 6 (3.1) | 0 | 0 |
Muscular weakness | 7 (3.6) | 6 (3.1) | 2 (1.0) | 0 |
Osteoarthritis | 4 (2.1) | 0 | 2 (1.0) | 0 |
Bone pain | 2 (1.0) | 2 (1.0) | 1 (0.5) | 0 |
Arthritis | 2 (1.0) | 1 (0.5) | 1 (0.5) | 0 |
Osteonecrosis | 0 | 2 (1.0) | 2 (1.0) | 0 |
Intervertebral disc protrusion | 0 | 1 (0.5) | 1 (0.5) | 0 |
Pathological fracture | 0 | 1 (0.5) | 1 (0.5) | 0 |
Infections and infestations | ||||
Urinary tract infection | 7 (3.6) | 25 (13.0) | 6 (3.1) | 0 |
Pneumoniab | 1 (0.5) | 4 (2.1) | 11 (5.7) | 0 |
Upper respiratory tract infection | 6 (3.1) | 9 (4.7) | 2 (1.0) | 0 |
Sepsisb | 0 | 0 | 1 (0.5) | 5 (2.6) |
Bronchitis | 2 (1.0) | 2 (1.0) | 1 (0.5) | 0 |
Gastroenteritis | 0 | 1 (0.5) | 1 (0.5) | 0 |
Septic shock | 0 | 0 | 1 (0.5) | 1 (0.5) |
Abdominal abscess | 0 | 0 | 1 (0.5) | 0 |
Bacteremia | 0 | 0 | 1 (0.5) | 0 |
Cellulitis orbital | 0 | 0 | 1 (0.5) | 0 |
Device related infection | 0 | 0 | 1 (0.5) | 0 |
Endocarditis | 0 | 0 | 1 (0.5) | 0 |
Kidney infection | 0 | 0 | 1 (0.5) | 0 |
Meningitis viral | 0 | 0 | 1 (0.5) | 0 |
Osteomyelitis | 0 | 0 | 1 (0.5) | 0 |
Pertussis | 0 | 0 | 1 (0.5) | 0 |
Pneumonia viral | 0 | 0 | 1 (0.5) | 0 |
Wound infection | 0 | 0 | 1 (0.5) | 0 |
Respiratory, thoracic, and mediastinal disordersb | ||||
Dyspnea | 21 (10.9) | 12 (6.2) | 8 (4.1) | 0 |
Cough | 23 (11.9) | 10 (5.2) | 0 | 0 |
Pleural effusion | 2 (1.0) | 7 (3.6) | 4 (2.1) | 0 |
Hypoxiab | 0 | 5 (2.6) | 5 (2.6) | 1 (0.5) |
Pulmonary embolism | 2 (1.0) | 0 | 7 (3.6) | 2 (1.0) |
Pneumonia aspiration | 0 | 1 (0.5) | 1 (0.5) | 0 |
Aspiration | 0 | 0 | 1 (0.5) | 1 (0.5) |
Lung disorder | 0 | 0 | 2 (1.0) | 0 |
Pneumonitis | 0 | 1 (0.5) | 1 (0.5) | 0 |
Pneumothorax | 1 (0.5) | 0 | 1 (0.5) | 0 |
Asthma | 0 | 0 | 1 (0.5) | 0 |
Hemothorax | 0 | 0 | 1 (0.5) | 0 |
Respiratory distress | 0 | 0 | 1 (0.5) | 0 |
Respiratory failure | 0 | 0 | 1 (0.5) | 0 |
Upper airway obstruction | 0 | 0 | 1 (0.5) | 0 |
Metabolism and nutrition disorders | ||||
Hyperuricaemia | 15 (7.8) | 2 (1.0) | 2 (1.0) | 4 (2.1) |
Dehydration | 5 (2.6) | 8 (4.1) | 4 (2.1) | 0 |
Hypocalcemia | 9 (4.7) | 3 (1.6) | 4 (2.1) | 1 (0.5) |
Decreased appetite | 9 (4.7) | 4 (2.1) | 3 (1.6) | 0 |
Hyperglycemia | 10 (5.2) | 2 (1.0) | 2 (1.0) | 1 (0.5) |
Hypokalemia | 11 (5.7) | 2 (1.0) | 2 (1.0) | 0 |
Hypoalbuminemia | 7 (3.6) | 5 (2.6) | 1 (0.5) | 0 |
Hyperkalemia | 10 (5.2) | 1 (0.5) | 1 (0.5) | 0 |
Hyponatremia | 7 (3.6) | 0 | 3 (1.6) | 0 |
Hypophosphatemia | 3 (1.6) | 4 (2.1) | 3 (1.6) | 0 |
Hypernatremia | 7 (3.6) | 1 (0.5) | 1 (0.5) | 0 |
Increased appetite | 4 (2.1) | 0 | 1 (0.5) | 0 |
Fluid retention | 2 (1.0) | 0 | 1 (0.5) | 0 |
Hypoglycemia | 2 (1.0) | 0 | 1 (0.5) | 0 |
Hypercalcaemia | 1 (0.5) | 0 | 1 (0.5) | 0 |
Hypermagnesemia | 1 (0.5) | 0 | 1 (0.5) | 0 |
Lactic acidosis | 0 | 0 | 1 (0.5) | 0 |
Tumour lysis syndrome | 0 | 0 | 1 (0.5) | 0 |
Blood and lymphatic system disorders | ||||
Anemia | 20 (10.4) | 21 (10.9) | 20 (10.4) | 1 (0.5) |
Neutropenia | 7 (3.6) | 2 (1.0) | 4 (2.1) | 0 |
Lymphopenia | 2 (1.0) | 1 (0.5) | 1 (0.5) | 0 |
Bone marrow failure | 0 | 0 | 1 (0.5) | 0 |
Leukocytosis | 0 | 0 | 1 (0.5) | 0 |
Skin and subcutaneous tissue disorders | ||||
Rash maculo-papular | 4 (2.1) | 0 | 1 (0.5) | 0 |
Skin ulcer | 0 | 2 (1.0) | 2 (1.0) | 0 |
Eye disorders | ||||
Diplopia | 2 (1.0) | 1 (0.5) | 1 (0.5) | 0 |
Renal and urinary disorders | ||||
Acute kidney injury | 7 (3.6) | 5 (2.6) | 3 (1.6) | 0 |
Urinary retention | 3 (1.6) | 3 (1.6) | 1 (0.5) | 0 |
Bladder diverticulum | 0 | 0 | 1 (0.5) | 0 |
Perinephritis | 0 | 0 | 1 (0.5) | 0 |
Renal injury | 0 | 0 | 1 (0.5) | 0 |
Urinary tract obstruction | 0 | 0 | 1 (0.5) | 0 |
Vascular disorders | ||||
Hypotension | 12 (6.2) | 13 (6.7) | 4 (2.1) | 1 (0.5) |
Hypertension | 2 (1.0) | 7 (3.6) | 1 (0.5) | 0 |
Orthostatic hypotension | 5 (2.6) | 1 (0.5) | 2 (1.0) | 0 |
Embolism | 0 | 1 (0.5) | 2 (1.0) | 0 |
Injury, poisoning, and procedural complications | ||||
Fall | 11 (5.7) | 10 (5.2) | 3 (1.6) | 0 |
Fracture | 0 | 5 (2.6) | 2 (1.0) | 0 |
Procedural pain | 0 | 4 (2.1) | 1 (0.5) | 0 |
Ankle fracture | 0 | 1 (0.5) | 1 (0.5) | 0 |
Foot fracture | 0 | 1 (0.5) | 1 (0.5) | 0 |
Radiation pneumonitis | 1 (0.5) | 0 | 1 (0.5) | 0 |
Spinal fracture | 1 (0.5) | 0 | 1 (0.5) | 0 |
Femoral neck fracture | 0 | 0 | 1 (0.5) | 0 |
Hip fracture | 0 | 0 | 1 (0.5) | 0 |
Ligament rupture | 0 | 0 | 1 (0.5) | 0 |
Wrist fracture | 0 | 0 | 1 (0.5) | 0 |
Psychiatric disorders | ||||
Confusional state | 8 (4.1) | 3 (1.6) | 2 (1.0) | 0 |
Anxiety | 5 (2.6) | 5 (2.6) | 1 (0.5) | 0 |
Depression | 1 (0.5) | 1 (0.5) | 1 (0.5) | 0 |
Mental status changes | 1 (0.5) | 0 | 1 (0.5) | 0 |
Lack of spontaneous speech | 0 | 0 | 1 (0.5) | 0 |
Cardiac disordersb | ||||
Cardiac failure | 0 | 0 | 2 (1.0) | 0 |
Cardiac failure congestive | 0 | 0 | 2 (1.0) | 0 |
Left ventricular dysfunction | 0 | 1 (0.5) | 1 (0.5) | 0 |
Acute right ventricular failure | 0 | 0 | 1 (0.5) | 0 |
Atrial flutter | 0 | 0 | 1 (0.5) | 0 |
Brugada syndrome | 0 | 0 | 1 (0.5) | 0 |
Pericardial effusion | 0 | 0 | 1 (0.5) | 0 |
Ear and labyrinth disorders | ||||
Vertigo | 9 (4.7) | 2 (1.0) | 1 (0.5) | 0 |
Deafness neurosensory | 1 (0.5) | 0 | 1 (0.5) | 0 |
Reproductive system and breast disorders | ||||
Benign prostatic hyperplasia | 0 | 0 | 1 (0.5) | 0 |
Endocrine disorders | ||||
Adrenal insufficiency | 0 | 0 | 1 (0.5) | 0 |
Immune system disorders | ||||
Anaphylactic reaction | 0 | 0 | 1 (0.5) | 0 |
Hepatobiliary disorders | ||||
Biliary colic | 0 | 0 | 1 (0.5) | 0 |
Surgical and medical procedures | ||||
Shoulder arthroplasty | 0 | 0 | 1 (0.5) | 0 |
Other (no coding available) | ||||
Other | 0 | 0 | 1 (0.5) | 0 |
ALT = alanine transaminase; AST = aspartate aminotransferase; CCOD = clinical cut-off date; MedDRA = Medical Dictionary for Regulatory Activities; NTRK = neurotrophic tyrosine receptor kinase.
aFor preferred term rows, multiple occurrences of the same adverse event in 1 individual are counted at the highest grade for this patient; for the overall (first) row, a patient contributes only with the adverse event occurring with the highest grade.
bFourteen grade 5 adverse events were reported (n = 1: cerebrovascular accident, death, sudden death, pneumonia, sepsis, hypoxia, pneumonia aspiration, atrioventricular block, cardiac arrest, and ventricular fibrillation; n = 2: acute respiratory failure, cardio-respiratory arrest).
Source: Sponsor-provided additional information.65
Table 25: Treatment-Emergent SAEs (NTRK Safety-Evaluable Population; August 31, 2020 CCOD; N = 193)
SAEs, n (%) | NTRK safety-evaluable population (N = 193) |
|---|---|
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
CCOD = clinical cut-off date; NTRK = neurotrophic tyrosine receptor kinase; SAE = serious adverse event.
Note that some redacted cells have been removed.
Source: Sponsor-provided additional information.32
Table 26: WDAEs (NTRK Safety-Evaluable Population; August 31, 2020 CCOD; N = 193)
WDAEs, n (%) | NTRK safety-evaluable population (N = 193) |
|---|---|
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
CCOD = clinical cut-off date; NTRK = neurotrophic tyrosine receptor kinase; WDAE = withdrawal due to adverse event.
Note that some redacted cells have been removed.
Source: Sponsor-provided additional information.32
Table 27: Mortality (NTRK Safety-Evaluable Population; October 31, 2018 CCOD; N = 113)
Deaths, n (%) | NTRK safety population (N = 113) |
|---|---|
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
AE = adverse event; CCOD = clinical cut-off date; NTRK = neurotrophic tyrosine receptor kinase.
Source: Safety Update Report (CCOD: October 31, 2018).73
Table 28: Adverse Events of Special Interest (Safety Analysis Population; May 31, 2018 CCOD; N = 355)
Preferred terms | Safety analysis population (n = 355) | Description |
|---|---|---|
Cognitive impairment | ||
| Any grade: 96 (27%) Grade ≥ 3: 16 (4.5%) | 77% of patients who experienced cognitive impairment had symptoms that occurred within 3 months of starting entrectinib. Among the 96 patients with cognitive impairment, 13% required a dose reduction, 18% required a dose interruption, and 1% discontinued due to AEs. |
Mood disorders | ||
| Any grade: 36 (10%) Grade ≥ 3: 2 (0.6%) | One suicide was completed and reported 11 days after the last dose of entrectinib. Among the 36 patients with mood disorders, 6% required a dose reduction, 6% required a dose interruption, and none discontinued due to mood disorders. |
Dizziness | ||
| Any grade: 136 (38%) Grade ≥ 3: 3 (0.8%) | 10% of patients required a dose reduction, 7% required a dose interruption, and 0.7% required treatment discontinuation due to dizziness. Among those who experienced dizziness, almost 80% did not have an alternative cause (e.g., neurologic or cardiovascular); thus, the dizziness was likely attributable to entrectinib. |
Sleep disturbance | ||
| Any grade: 51 (14%) Grade ≥ 3: 2 (0.6%) | Among those who experienced sleep disturbance, 6% required a dose reduction; no patients discontinued due to sleep disturbance. |
Congestive heart failure | ||
| Any grade: 12 (3.4%) SAEs: 7 (2.0%) Grade ≥ 3: 8 (2.3%) | Treatment was interrupted in 3 patients, reduced in 1 patient, and discontinued in 3 patients who experienced CHF as an SAE. Five of the patients with CHF as an SAE recovered. Of the 12 patients with CHF, 7 had a past medical cardiac history at baseline that may have predisposed them to CHF events. |
Skeletal fractures | ||
| Any grade: 27 (5.4%) SAEs: 12 (2.4%) | 27 adults were identified as having skeletal fractures. Some fractures occurred in the setting of a fall or other trauma to the affected area. Most fractures occurred in the lower extremity (e.g., femoral or tibial shaft). 41% of patients interrupted entrectinib treatment and none discontinued. |
Vision disorders | ||
| Any grade: 75 (21%) Grade ≥ 3: 3 (0.8%) | 28.5% of patients experienced eye disorders (3 patients reported these as a grade 3 AE). The most frequently reported eye disorders were blurred vision (9%), photophobia (5.4%), dry eyes (3.4%), diplopia (3.1%), and eye pain (2.5%). Vitreous floaters and cataracts occurred in 1.1% of patients. Vitreous detachment occurred in 0.8% of patients, while vitreous adhesions, blindness, corneal erosion, keratitis, and retinal hemorrhage occurred in 0.3% of patients. |
AE = adverse event; CCOD = clinical cut-off date; CHF = congestive heart failure; SAE = serious adverse event.
Source: FDA Multidisciplinary Review45 and Health Canada Pharmaceutical Safety and Efficacy Assessment.46
Critical Appraisal
Internal Validity
Single-Arm Trials
The trials included in the pooled analyses of efficacy and safety were all single-arm studies with no comparator group. Health Canada and FDA noted that the time-to-event end points are not interpretable without a control group; therefore, the PFS, OS, and time to CNS progression cannot be considered in the definitive analysis to support efficacy.45,46
Heterogeneity of Included Trials
Due to the rarity of NTRK fusion cancers, the sponsor conducted pooled analyses of efficacy and safety as the basis for the regulatory and reimbursement review submissions. Although the pooled analyses included patients from 3 trials, nearly all of the patients were from the STARTRK-2 ongoing trial (98% and 97% in the efficacy and safety analyses, respectively). This reduces the potential uncertainty that can arise from between-study heterogeneity (e.g., differences in study design, objectives, phases, outcome measures, and eligibility criteria across trials) that has been previously noted by CADTH for larotrectinib. There were differences in NTRK gene fusion testing across and within trials to identify patients for inclusion in the NTRK efficacy and safety-evaluable populations.45 However, any failure to accurately screen patients for NTRK fusion–positive tumours could bias the efficacy results against entrectinib, given the mechanism of action and lack of a control in these studies (unless the patient’s tumour harboured a different mutation that was responsive to entrectinib, such as ROS1 with NSCLC).
Heterogeneity of Tumour types
The NTRK1, NTRK2, and NTRK3 genes encode TRKA, TRKB, and TRKC proteins, which have been established to be part of oncogenic pathways. Oncogenic NTRK gene fusions are found in a diverse range of tumour types in adult and pediatric patients, but the overall prevalence is rare; it is estimated at 0.2% to 0.4% across solid tumours. However, NTRK fusions have been found to be relatively common (≥ 75%) in select rare cancers, including MASC, secretory breast cancer, mesoblastic nephroma, and congenital fibrosarcoma.74 There are limited data on the natural history of NTRK fusion–positive solid tumours, with some evidence to suggest that patients with NTRK fusion–positive papillary carcinoma, colorectal carcinoma, or NSCLC may have poorer disease-free survival and/or OS compared to patients without these genetic alterations.8,75,76 Thus, the integrated efficacy analysis is limited by the absence of data on the prognosis and natural history of patients who have a NTRK fusion–positive tumour overall and on whether this is similar across all tumour types and disease stages.
This issue is compounded by the fact that some tumour types may be overrepresented in the overall NTRK efficacy-evaluable population, in whom the best available evidence may suggest that the NTRK fusion is less common (for example, sarcoma, MASC, and NSCLC make up ≥ 50% of the included patients; however, the prevalence of NTRK fusion is higher only in MASC patients, among these subgroups).74 There is still some degree of uncertainty as to whether the presence of an NTRK fusion is a stronger prognostic marker than histologic tumour type; for example, research has shown that V600E BRAF–mutant melanoma and hairy cell leukemia are responsive to BRAF inhibition, while colon tumours with the same BRAF mutation are not.77-81 This may affect the generalizability of the efficacy results.
The sample sizes for each individual tumour type were small, with the majority involving fewer than 10 patients in the updated pooled analyses (and substantially fewer in the earlier analyses used for the regulatory approvals). As such, the point estimates of the ORR across tumour types ranged widely and, despite the use of pooled analyses, the associated CIs for the subgroup analyses reflected a high degree of uncertainty around the point estimates of the ORR for many tumour types. As noted in CADTH’s review of larotrectinib for NTRK positive tumours, it is not methodologically meaningful to evaluate the ORR (and by extension, the effectiveness) of TRK inhibitors on different cancer types separately. The experts emphasized that the response rates observed across the tumour sites may be influenced by the number of prior therapies, which will vary considerably across the different tumours. The sponsor’s submission did not include a detailed breakdown of the number of prior therapies used by patients within the individual tumour type subgroups.
Blinding
All of the trials used an open-label design, which could have introduced bias in the reporting of subjective outcome measures, such as the HRQoL instruments in STARTRK-2. Open-label, single-group trials can also be subject to selection bias during patient recruitment if investigators are more likely to enrol patients who are more likely to benefit from the treatment and/or experience fewer AEs. The efficacy end points in the pooled analyses were evaluated through BICR assessment for the primary analyses (with investigator-assessed outcomes provided as a sensitivity analyses); this is an important design feature, given that the trials were all open-label, single-arm studies. BICR assessment in the phase I trials (ALKA and STARTRK-1) was conducted retrospectively, whereas patients in the STARTRK-2 trial had prospective BICR assessment.45 However, as noted earlier, nearly all patients in the pooled analyses were from STARTRK-2, limiting concerns about the appropriateness of pooling data from the 3 studies.
Given that STARTRK-2 was an open-label study, all patients were aware that they were receiving entrectinib. Such knowledge could result in biased assessments of HRQoL because patients could have over-reported treatment gains. The patients who were able to complete HRQoL assessments at later cycles are unlikely to be representative of the patients who discontinued entrectinib treatment earlier; thus, assessments of HRQoL at these later time points may overestimate the treatment effect in favour of entrectinib.
Outcomes
The primary outcomes (i.e., ORR and DOR) were considered appropriate by the clinical experts consulted by CADTH and by regulatory authorities for a basket trial design for a rare mutation.27,45,46 The following secondary end points are difficult to interpret in the absence of a control group: CBR, PFS, OS, time to CNS progression, and IC-PFS. STARTRK-2 is ongoing; therefore, the results may change as more patients are recruited.
Time windows for tumour scans varied by trial, and tumour scans outside of protocol-defined time points could be conducted at the investigator’s discretion. However, this was not limited to clinical worsening. Information about whether tumour scans conducted outside of protocol-defined time points were to assess tumour response or PD was not provided; therefore, it is unclear if these assessments may have biased the efficacy results, given that what prompted the off-schedule assessments is unknown. Additionally, differences in the objectives of the trials may have led to differential selection of patients (e.g., the types of patients recruited to phase I trials [versus phase II] may be different in terms of having more advanced disease or fewer alternative therapeutic options). These differences can contribute to selection bias for patients included in the efficacy populations.
NTRK Fusion Testing
A variety of different NTRK testing methodologies were used to identify patients with NTRK gene fusion, including ribonucleic acid (RNA)- and DNA-based NGS assay, polymerase chain reaction, and NanoString. Most patients (92%) were identified as positive by central RNA-based NGS testing; however, there were 4 patients identified as NTRK fusion–positive by local testing methods for whom this identification could not be confirmed through central RNA-based testing, and 5 patients for whom central testing was not conducted. In total, due to the variation of NTRK fusion testing methodologies, 9 patients (16.7%) may or may not have had a confirmed NTRK fusion–positive tumour.45 Given the small sample size of the integrated analyses, 9 patients (16.6%) represents a large proportion of patients who may not have had an NTRK gene fusion–positive solid tumour centrally confirmed by NGS testing. This uncertainty has the potential to confound the results in an unknown direction because entrectinib activity in non–NTRK gene fusion–positive solid tumours is unknown.
Statistical Analysis
The clinical experts consulted by CADTH generally agreed that an ORR that excluded 30% in the lower limit of the 95% CI was considered clinically meaningful for most patients who have exhausted standard therapies or for whom no suitable alternatives exist. This is consistent with FDA guidelines on statistical analysis considerations for basket trials when outlining a clinically important response rate.82 However, there are also issues of intra-tumour heterogeneity at the cellular level as well as tumour resistance, and passenger mutations can eventually become onco-drivers in different environmental contexts. Pooling data on survival outcomes (i.e., PFS and OS) could also be problematic. Given the mix of tumour types with varying prior lines of therapy and likely differing survival probabilities, unadjusted survival estimates may oversimplify results. Thus, a short-term end point, such as ORR, may not predict OS in the context of intra-tumour heterogeneity. Generally, the ability of ORR, or even PFS, to predict OS benefit is context-dependent (i.e., it varies by tumour type).83
External Validity
Patient Population
The clinical experts consulted by CADTH indicated that the population included in the pooled analysis was a reasonable reflection of the target population in Canada, noting that because NTRK fusion tumours are rare, the small sample size is expected. The entrectinib trials included patients with NTRK-positive solid tumours regardless of their tumour type. However, not all solid tumour types were represented in the pooled analysis. As part of the conditions for marketing authorization, the sponsor is mandated to provide additional evidence based on a sufficient number of patients to more precisely characterize response and durability of response for each of the following tumour types: CRC, gynecological cancers, and melanoma.84
The eligibility criteria for the entrectinib trials did not restrict the number of previous lines of systematic therapy. Both the clinical experts consulted by CADTH and the clinician groups who provided input noted that entrectinib should be considered early in NTRK fusion cancer treatment. Although it is challenging to interpret evidence from subgroups in the pooled analysis due to small sample sizes and heterogeneity across tumour types, the clinical experts noted that the ORRs observed across subgroups based on number of prior treatment regimens suggest that entrectinib may be more efficacious when used earlier in the course of treatment.
Patients included in the pooled analysis had ECOG PS scores of 0, 1, or 2. The clinical experts noted the relatively fast median time to response along with the high rate of response and favourable toxicity profile as key considerations for potentially making entrectinib available to patients with an ECOG PS of 3 (a population that was not studied in the clinical trials) in situations where the oncologist believes that tumour-related symptoms are driving the PS.
Intervention
Nearly all patients in the pooled analysis received entrectinib at the dosage recommended in the Canadian product monograph (i.e., 600 mg once daily to start, with up to 2 dose reductions permitted to manage AEs). There were no direct or indirect comparisons filed by the sponsor to evaluate the comparative efficacy and safety of entrectinib versus larotrectinib or other alternative therapies.
Comparative Efficacy
In the absence of comparative effectiveness data, it is unclear whether patients will have better or worse outcomes with entrectinib when compared to the most relevant current treatment options for their tumour types. Although a clinically meaningful ORRs were demonstrated in the integrated analysis (noting that these varied across tumour types), uncertainty remains regarding whether entrectinib improves PFS and OS as well as HRQoL, given that no statistical inferences can be made for these end points. The small sample size, paired with limited data on the natural history of NTRK fusion–positive tumours overall — or for individual tumour types — makes it difficult to interpret how the observed treatment-effect estimates obtained for entrectinib compared to standard of care. Because treatments exist for some tumour types (where biomarkers such as programmed death-ligand 1 and MSI-H are targetable), evidence gaps remain as to the ideal treatment choice for patients who harbour these coexisting biomarkers.
For the purpose of evaluating cost-effectiveness, the efficacy of standard therapies was estimated through modelling techniques; the estimated efficacy reflects a hypothetical average patient across multiple tumour types and disease stages with varying prior lines of therapy. Refer to the CADTH Pharmacoeconomic Report for more details.
Outcomes
The STARTRK-2, STARTRK-1, and ALKA trials performed on-treatment tumour assessments through CT and/or MRI at the end of cycle 1 (4 weeks) and at the end of alternate cycles thereafter (i.e., every 8 weeks), or whenever a clinical deterioration was observed, and at end of treatment, if not done in the previous 4 weeks. The clinical experts consulted by CADTH noted that the imaging methods used in Canadian practice to evaluate response to treatment and disease progression are similar (i.e., MRI, CT, and PET/CT to assess response by RECIST 1.1 for solid tumours or by Response Assessment in Neuro-Oncology for CNS tumours); however, the timing of the assessments can differ, given that while treatment response is typically assessed once every 3 months, the interval may be prolonged once response has been established or remission achieved.
Table 29 summarizes the generalizability of the evidence.
Table 29: Assessment of the Generalizability of Evidence for Entrectinib
Domain | Factor | Evidence | CADTH's assessment of generalizability |
|---|---|---|---|
Population | Histological tumour type | The entrectinib trials included patients with NTRK-positive solid tumours, regardless of tumour type. The following tumour types were recorded in the study:
| Although some variation was observed in response rates within the subgroup analyses by tumour type, from a histology-agnostic, biomarker-driven perspective, the overall pooled analysis results are generalizable to all patients in the pooled analysis. Nonetheless, some of these tumour types were under-represented in the study population, resulting in wide confidence intervals and reducing confidence in their generalizability. |
Co-mutations | The entrectinib trials included in the submitted pooled analysis focused on the presence of documented NTRK gene fusions (determined by local testing). | The results are generalizable to cancers with NTRK1, NTRK2, and NTRK3 gene fusions. | |
Line of therapy | The eligibility criteria for the 3 entrectinib trials in adults and the pooled analysis were not restricted based on previous lines of systematic therapy (0 to > 4). 0 = 37 (30.6%) 1 = 35 (28.9%) 2 = 26 (21.5%) 3 = 12 (9.9%) 4 = 7 (5.8%) > 4 = 4 (3.3%) | There is no clinical rationale to suggest that the trial results would not be applicable to Canadian practice. | |
CNS metastases | Entrectinib is not indicated for use in patients with primary CNS tumours; however, it is indicated for use in patients with CNS metastases. The sponsor conducted pre-specified subgroup analyses to examine the efficacy of entrectinib in patients with primary CNS tumours. | The clinical experts consulted by CADTH indicated that the intracranial efficacy results are clinically important, particularly given that these patients typically have a poor prognosis. | |
ECOG PS | The inclusion criteria for the pooled analysis required patients to have an ECOG PS ≤ 2. | The clinical experts suggested that patients with an ECOG PS ≤ 3 could be eligible for treatment with entrectinib if the oncologist believes that tumour-related symptoms are driving the PS. The rationale is based on the high rate of response, duration of response, median time to response (i.e., approximately 1 month), and favourable toxicity profile. | |
Intervention | Dosing schedule | 98% of patients in the pooled analysis received a dosage of 600 mg once daily. | This is reflective of recommendations in the product monograph for entrectinib. |
Dose reductions | To manage adverse events, the starting dose of 600 mg once daily could be reduced twice in the STARTRK-2 trial, first to 400 mg once daily and then to 200 mg once daily. | This is reflective of recommendations in the product monograph for entrectinib. | |
Comparator | Larotrectinib | There were no direct or indirect comparisons to evaluate the comparative safety and efficacy of entrectinib compared with larotrectinib. | The lack of comparative data for entrectinib and larotrectinib remains an important gap in the available evidence. Given the rarity of NTRK-positive tumours, adequately powered direct or indirect comparative studies are unlikely to be conducted. |
Standard non–NTRK-targeted therapies | There were no direct or indirect comparisons to evaluate the comparative safety and efficacy of entrectinib compared with alternative non–NTRK targeting therapies. | The following secondary end points cannot be interpreted in the absence of a control group: CBR, PFS, OS, time to CNS progression, and IC-PFS. | |
Subsequent therapy | Post-progression entrectinib therapy | In STARTRK-2, patients could continue entrectinib treatment beyond BICR-confirmed progression. In the October 2018 CCOD date set, a total of 18 patients (33.3%) were treated with entrectinib beyond BICR-confirmed progression. | The clinical experts consulted by CADTH noted that, in Canadian practice, there would be interest in continuing some patients on entrectinib therapy after disease progression. This would likely occur if patients were symptomatic, had few or no other treatment options, and the patient and provider believe the drug is continuing to be beneficial. |
Outcomes | Appropriateness of primary and secondary outcomes | The primary end points of the integrated analysis were ORR and DOR determined by BICR. Secondary end points included CBR, PFS, time to CNS progression, and OS. Intracranial efficacy end points included IC-ORR, IC-DOR, and IC-PFS. | The primary and secondary outcomes were appropriate for a basket trial design. |
HRQoL | HRQoL was evaluated only in the STARTRK-2 study. | The lack of a comparator group, open-label administration, and heterogenous study population make it challenging to evaluate the impact of entrectinib on HRQoL. In addition, it was noted that patients enrolled in confirmatory clinical trials may receive optimized supportive care and symptom management that may not be reflective of routine clinical practice, in which access to health care teams may be more limited. | |
Timing of assessment | The STARTRK-2, STARTRK-1, and ALKA trials performed on-treatment tumour assessments through CT and/or MRI at the end of cycle 1 (4 weeks) and at the end of alternate cycles thereafter (i.e., every 8 weeks), or whenever a clinical deterioration was observed, and at end of treatment, if not done in the previous 4 weeks. | The clinical experts consulted by CADTH noted that the imaging methods used in Canadian practice to evaluate response to treatment and disease progression are similar (i.e., MRI, CT, PET/CT to assess response by RECIST 1.1 for solid tumours or RANO for CNS tumours); however, the timing of assessments can differ. Treatment response is typically assessed once every 3 months, but once response has been established or remission achieved, the interval may be further prolonged. | |
Setting | Countries participating in the trial | STARTRK-2 was a multinational trial conducted in 15 countries (Australia, Belgium, France, Germany, Hong Kong, Italy, Japan, South Korea, Netherlands, Poland, Singapore, Spain, Taiwan, UK, US). There were no Canadian sites. | While there may be small differences in practice patterns, the clinical experts consulted for this review do not feel these differences would be sufficient to expect that the results would not be generalizable to the population of patients in Canada. |
BICR = blinded independent central review; CBR = clinical benefit rate; CCOD = clinical cut-off date; CNS = central nervous system; DOR = duration of response; ECOG PS = Eastern Cooperative Oncology Group Performance Status; HRQoL = health-related quality of life; IC-DOR = intracranial duration of response; IC-ORR = intracranial objective response rate; IC-PFS = intracranial progression-free survival; NSCLC = non–small cell lung carcinoma; NTRK = neurotrophic tyrosine receptor kinase; ORR = objective response rate; OS = overall survival; PFS = progression-free survival; RANO = Response Assessment in Neuro-Oncology; RECIST 1.1 = Response Evaluation Criteria in Solid Tumours Version 1.1.
Indirect Evidence
No studies have directly compared entrectinib versus larotrectinib for patients with NTRK-positive tumours. The sponsor did not include an indirect comparison in its application to CADTH. It provided the following reasons why it believes it is not feasible to compare entrectinib and larotrectinib:
The data from the entrectinib integrated analysis are based on an adult population, whereas the data within the larotrectinib program involve both adult and pediatric populations.
Subgroup analyses of the adult population in the larotrectinib studies demonstrated differences in the characteristics of the patient population (e.g., there was a higher proportion of patients with CNS metastases in the most recent entrectinib dataset [21.5% with entrectinib versus 14% for larotrectinib]), and there were differences in the distribution of patients based on prior lines of systemic anti-cancer therapy within the studies (e.g., 19% versus 31% of patients in the entrectinib and larotrectinib populations, respectively, had received ≥ 3 prior lines of therapy).
Differences in primary tumour type distribution, limited information regarding histology of the tumour types, and inability to assess adult tumour-specific response rates for larotrectinib add to the challenge of conducting a meaningful comparison across the study populations, specifically in instances with small sample sizes and sensitivity of response rates changing depending on individual patient responses.
There are differences in the reporting methods between the studies. The entrectinib studies primarily use BICR to report efficacy outcomes, whereas the recent larotrectinib adult sub-analysis used investigator-assessed response.
A UK National Institute for Health Research-sponsored study of modelling approaches for histology-independent cancer drugs conducted to inform National Institute for Health and Care Excellence appraisals concluded that, although it is theoretically possible, implementing a MAIC analysis in a tumour-agnostic setting is challenging because it would require making strong assumptions about the prognostic value of population characteristics across tumour types; in addition, the small sample sizes typically seen in phase II trials for tumour-agnostic treatments (such as NTRK inhibitors) would be able to account for only a small number of observed characteristics.
The sponsor further noted a formal feasibility assessment (reported by Chu et al., 2020; funded by the sponsor) concluded that an indirect comparison was not feasible due to differences in the study populations (mixed pediatric and adult population for larotrectinib and adult-only for entrectinib) and substantial differences in the follow-up periods between the analyses (15.5 months for entrectinib versus 12 months for larotrectinib). The authors reported that an unanchored MAIC was not feasible for specific tumour types due to the small number of patients and differences in the number of patients who had CNS metastases at baseline.85
Objectives and Methods for the Summary of Indirect Evidence
In the absence of direct or indirect evidence comparing entrectinib and larotrectinib in the submission, CADTH conducted a literature search to identify any relevant published indirect comparisons. A focused literature search for indirect comparisons dealing with entrectinib or NTRK gene fusion was run in MEDLINE All (1946–) on May 6, 2022. No limits were applied to the search. CADTH identified 1 MAIC that compared entrectinib and larotrectinib in adult patients with NTRK gene fusion–positive tumours.86
Description of MAIC by Garcia-Foncillas et al. (2022)
Objective
The objective of the MAIC was to compare the efficacy and safety of entrectinib and larotrectinib in adults with NTRK fusion–positive tumours.
Study Selection Methods
Garcia-Foncillas et al. (2022) extracted patient-level data from an integrated analysis of 3 larotrectinib clinical trials (a phase I trial [NCT02122913], SCOUT [NCT02637687], and NAVIGATE [NCT02576431]) based on a July 2020 CCOD. For entrectinib, the authors extracted aggregate results for the integrated patient population from 3 phase I/2 clinical trials — ALKA-372 to 001, STARTRK-1, and STARTRK-2 — based on the earlier May 31, 2018 and October 31, 2018 CCODs. The MAIC was funded by the manufacturer of larotrectinib; therefore, patient-level data were available for the larotrectinib-treated patients, but not for the entrectinib-treated patients.
Patient Selection Methods
The authors reported that the key eligibility criteria from the entrectinib clinical trials were applied to the individual patient data available for the larotrectinib clinical trials. Patients in the larotrectinib patient population who had primary CNS tumours were excluded because they had not been included in the entrectinib population. After applying the entrectinib eligibility criteria to the larotrectinib population, the following criteria were used to select patients for inclusion in the MAIC: NTRK fusion as determined by an independent review committee; at least 18 years of age; ECOG PS of 2 or less; and TRK inhibitor-naive.
Methods for Matching Patients
Patients were matched based on baseline characteristics that were available (as previously noted, patient-level data were limited to the larotrectinib dataset) and their perceived potential to be treatment-effect modifiers (based on clinical opinion). The following matching baseline characteristics were used in the analysis: sex; age (> 57 years [to match the median age range in the entrectinib population]); race (White, Black, Asian, other); ECOG PS (0, 1, or 2); tumour type (thyroid, sarcoma, salivary, or lung); metastatic disease (versus locally advanced, unresectable disease); NTRK fusion type (NTRK1, NTRK2, or NTRK3); prior lines of systemic therapy for metastatic disease (0, 1, 2, or more than 3); and previous CNS metastases. The authors assumed that the average baseline characteristics of the entrectinib safety population were the same as that of those of the efficacy population (separate reporting of characteristics was not available).
Patients treated with larotrectinib were assigned weights to generate a weighted average of selected baseline characteristics that were matched with those of the entrectinib population. The weights were obtained using a logistic regression model for the propensity of enrolment in the larotrectinib versus entrectinib clinical trials. The logistic regression model was estimated using the method of moments.
Table 30: Baseline Characteristics Before and After Matching of Larotrectinib Efficacy Population and Entrectinib
Variable | Entrectinib (N = 74) | Larotrectinib (N = 117) | ||
|---|---|---|---|---|
Before matching | After matchinga (%) | |||
N (%) | N (%) | P valueb | ||
Male | 35 (47.3%) | 54 (46.2%) | > 0.99 | 47.3% |
Age > 57 years | 37 (50.0%) | 52 (44.4%) | 0.55 | 50.0% |
Race | ||||
White | 52 (70.3%) | 86 (73.5%) | 0.75 | 70.3% |
Black | 2 (2.7%) | 5 (4.3%) | 0.71 | 2.7% |
Asian | 13 (17.6%) | 14 (12.0%) | 0.38 | 17.6% |
Other/not reported | 7 (9.5%) | 12 (10.3%) | > 0.99 | 9.4% |
ECOG PS score | ||||
0 | 30 (40.5%) | 41 (35.0%) | 0.54 | 40.5% |
1 | 34 (45.9%) | 61 (52.1%) | 0.49 | 45.9% |
2 | 10 (13.5%) | 15 (12.8%) | > 0.99 | 13.6% |
Primary tumour type | ||||
Thyroid | 7 (9.5) | 25 (21.4) | 0.05 | 9.5 |
Salivary | 13 (17.6) | 21 (17.9) | > 0.99 | 17.6 |
Sarcoma | 16 (21.6) | 25 (21.4) | > 0.99 | 21.6 |
Lung | 13 (17.6) | 13 (11.1) | 0.29 | 17.6 |
Other | 25 (33.8) | 33 (28.2) | 0.51 | 33.7 |
Metastatic disease (vs. locally advanced, unresectable disease) | 52 (96.3) | 106 (90.6) | 0.23 | 96.3 |
Central nervous system metastases (yes) | 16 (21.6) | 14 (12.0) | 0.11 | 21.6 |
NTRK gene fusion | ||||
NTRK1 | 30 (40.5) | 52 (44.4) | 0.70 | 40.5 |
NTRK2 | 2 (2.7) | 3 (2.6) | > 0.99 | 2.7 |
NTRK3 | 42 (56.8) | 62 (53.0) | 0.72 | 56.8 |
Prior lines of systemic therapy for metastatic disease | ||||
0 | 20 (27.0) | 30 (25.6) | 0.97 | 27.0 |
1 | 21 (28.4) | 29 (24.8) | 0.70 | 28.4 |
2 | 20 (27.0) | 23 (19.7) | 0.31 | 27.0 |
3+ | 13 (17.6) | 35 (29.9) | 0.08 | 17.6 |
Prior therapy (chemotherapy)c | 60 (81.1) | 68 (58.1%) | < 0.01d | 58.1 |
P < 0.01d | ||||
Prior therapy (hormonal therapy)c | 9 (12.2) | 6 (5.1%) | 0.14 | 2.0 |
P < 0.01d | ||||
Prior therapy (immunotherapy)c | 9 (12.2) | 14 (12.0%) | 1.00 | 15.9 |
P = 0.53 | ||||
Prior therapy (targeted therapy)c | 18 (24.3) | 31 (26.5%) | 0.87 | 18.5 |
P = 0.36 | ||||
ECOG PS = Eastern Cooperative Oncology Group Performance Status; NTRK = neurotrophic receptor tyrosine kinase; vs. = versus.
aMatching variables include the following: male; age above the median in entrectinib population in Rolfo (2020) (> 57 years); White, Black, Asian; ECOG PS score 0; ECOG PS score 1; tumour (thyroid); tumour (sarcoma); tumour (salivary); tumour (lung); metastatic disease (vs. locally advanced, unresectable disease); central nervous system metastases (yes); NTRK1; NTRK2; prior lines of systemic therapy for metastatic disease (0); prior lines of systemic therapy for metastatic disease (1); prior lines of systemic therapy for metastatic disease (2).
bP values for continuous variables and categorical variables were calculated using the Wald test.
cIn the primary analysis, prior therapy type was not adjusted for.
dDenotes statistical significance (alpha < 0.05).
Source: Garcia-Foncillas et al. (2022).86 © 2022 by the authors. Licensee MDPI, Basel, Switzerland. Reprinted in accordance with Attribution 4.0 International (CC BY 4.0) https://creativecommons.org/licenses/by/4.0/
Outcome Assessment
Garcia-Foncillas et al. (2022) conducted analyses for OS, PFS, DOR, ORR, CR rate, serious treatment-related AEs (defined as grade ≥ 3), and treatment-related AEs that led to treatment discontinuation. The following statistical approaches were used:
Time-to-event outcomes (OS, PFS, DOR): Survival curves were compared using weighted log-rank tests, and HRs were estimated from weighted, otherwise unadjusted Cox proportional hazards models.
Categorical end points (ORR, CR rate, AEs): Risk differences were calculated and weighted chi-square tests were used for statistical comparisons.
Sensitivity Analyses and Simulated Treatment Comparison
Sensitivity analyses were conducted by adding GI tumours and replacing the number of prior lines of systemic cancer therapy by the type of prior cancer therapies (i.e., hormonal therapy, chemotherapy, immunotherapy, or targeted therapy). An additional sensitivity analysis was conducted using a simulated treatment comparison method. The authors used the following approach for the simulated treatment comparison:
Regression models were applied using the same set of matching baseline characteristics in the primary analysis using the larotrectinib patient-level data.
Cox regression models were used for OS, PFS, and DOR, and logistic regression models were used for ORR, CR, and safety outcomes.
The simulated treatment comparison then combined the regression model estimates, the baseline characteristics for the entrectinib-treated population, and the correlations between covariates that were estimated from larotrectinib-treated patient-level data to simulate pseudo-patient-level outcome data for a hypothetical larotrectinib-treated population in the entrectinib clinical trial.
Results of MAIC
Efficacy Results
Figure 12 summarizes the results of the MAICs for the efficacy and safety end points. Garcia-Foncillas et al. (2022) reported that, compared with entrectinib, larotrectinib was associated with a statistically significantly greater duration of OS (hazard ratio [HR] = 0.43; 95% CI, 0.23 to 0.83; P < 0.05) and DOR (HR = 0.49; 95% CI, 0.25 to 0.98; P < 0.05). The authors reported no statistically significant difference for PFS (HR = 0.66; 95% CI, 0.42 to 1.03; P = 0.07) or ORR (risk difference [RD] = 3.8; 95% CI, –11.7 to 19.3; P = 0.63). Results were similar in sensitivity analyses applying different specifications for the MAIC and using a simulated treatment comparison method.
The results of the sensitivity analyses are summarized in Figure 13 and were similar to those reported for the primary analyses for the different specifications of the MAIC and the simulated treatment comparison.
Safety Results
As shown in Figure 12, there were no statistically significant differences reported between larotrectinib and entrectinib for serious TRAEs or TRAEs leading to discontinuation.
Critical Appraisal of MAIC
Garcia-Foncillas et al.86 conducted a MAIC of efficacy and safety end points for larotrectinib versus entrectinib for the treatment of TRK fusion–positive cancers. The study was funded by Bayer Health care Pharmaceuticals LLC and Loxo Oncology, Inc. Conflicts of interest were noted among the study investigators. Due to the lack of a head-to-head comparison between the comparators of interest and the fact that each comparator was investigated in single-arm trials, an unanchored MAIC was the required approach for the indirect comparison.
The comparison used individual patient-level data from 2 studies of larotrectinib (NAVIGATE and SCOUT) and aggregate-level data from 3 studies of entrectinib (ALKA-372 to 001, STARTRK-1, and STARTRK-2). For each comparator, studies were combined without accounting for between-study heterogeneity. As mentioned previously, the majority of patients on entrectinib came from the STARTRK-2 study; hence, results are unlikely to be biased by the naive pooling for this comparator. The distribution of patients on larotrectinib across the 2 studies was not described. Thus, the potential impact of the pooling of patients for this comparator is unknown. Study investigators provided minimal detail about the study design for the larotrectinib studies; therefore, it is unclear what potential sources of heterogeneity exist between the studies for the 2 comparators. The removal of patients from the larotrectinib studies to match the inclusion criteria for entrectinib was deemed appropriate and would not limit the generalizability of results to the patient population of interest.
Figure 11: Summary of Efficacy and Safety Results From MAIC by Garcia-Foncillas et al. (2022)
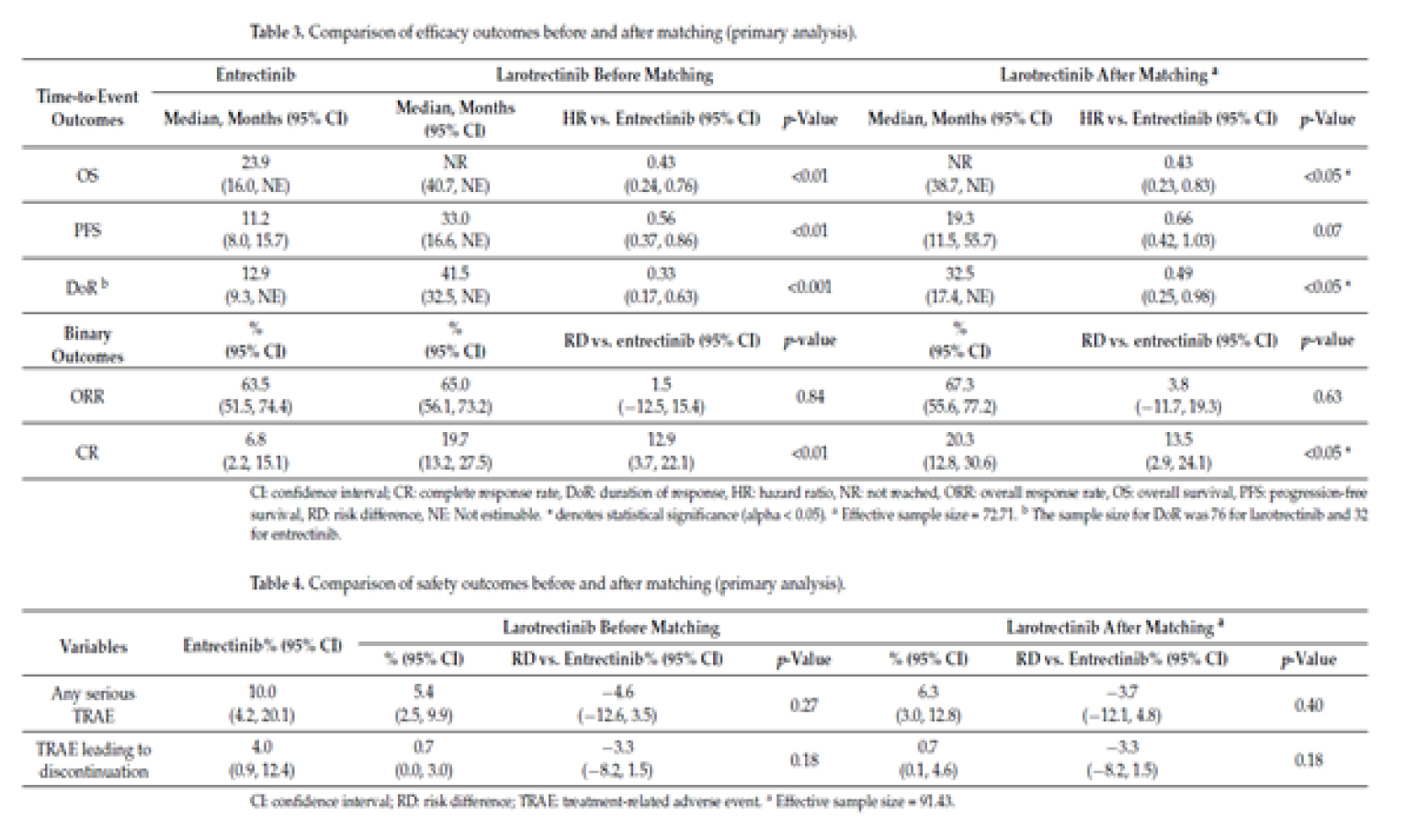
CI = confidence interval; CR = complete response rate; DoR = duration of response; HR = hazard ratio; NE = not estimable; NR = not reached; ORR = objective response rate; OS = overall survival; PFS = progression-free survival; RD = risk difference; TRAE = treatment-related adverse event; vs. = versus.
Source: Garcia-Foncillas et al. (2022).86 © 2022 by the authors. Licensee MDPI, Basel, Switzerland. Reprinted in accordance with Attribution 4.0 International (CC BY 4.0) https://creativecommons.org/licenses/by/4.0/
To account for between-trial differences in patient baseline characteristics, the following variables were included in the weighting process for the MAIC: sex; age (> 57 years [to match the median age range in the entrectinib population]); race (White, Black, Asian, other); ECOG PS score (0, 1, 2), tumour type (thyroid, sarcoma, salivary, lung); metastatic disease (versus locally advanced, unresectable disease); NTRK fusion type (NTRK1, NTRK2, or NTRK3); prior lines of systemic therapy for metastatic disease (0, 1, 2, or 3+); and previous CNS metastases. Investigators reported that the list of variables was identified based on the availability of data and clinical input on potential effect modifiers. Due to the unanchored nature of the comparison, an unbiased comparison would require the inclusion of all prognostic factors and all effect modifiers for the outcomes of interest in the weighting process. This assumption is generally considered to be difficult to meet,87 and is particularly unlikely for this study because the investigators did not conduct a systematic review to identify all such variables. Furthermore, the variables included in the weighting process may not adequately account for sources of heterogeneity for each variable, particularly tumour type, accounting for only 4 locations out of many across the tumour-agnostic studies. Investigators conducted additional sensitivity analyses by adding the variables for number and type of previous therapies and GI tumour indicator to the weighting process, and an alternative analysis using a simulated treatment comparison to examine the robustness of the results to the methodological approach. These sensitivity analyses were generally consistent with the primary results. However, the investigators did not examine the sensitivity of results to unbalanced, unmeasured prognostic factors or effect modifiers; the absence of this examination is the primary limitation of the study. Due to the limitations of the unanchored design for the MAIC, drawing firm conclusions regarding the comparison of end points between larotrectinib and entrectinib is not recommended.
Figure 12: Sensitivity Analyses From MAIC by Garcia-Foncillas et al. (2022)
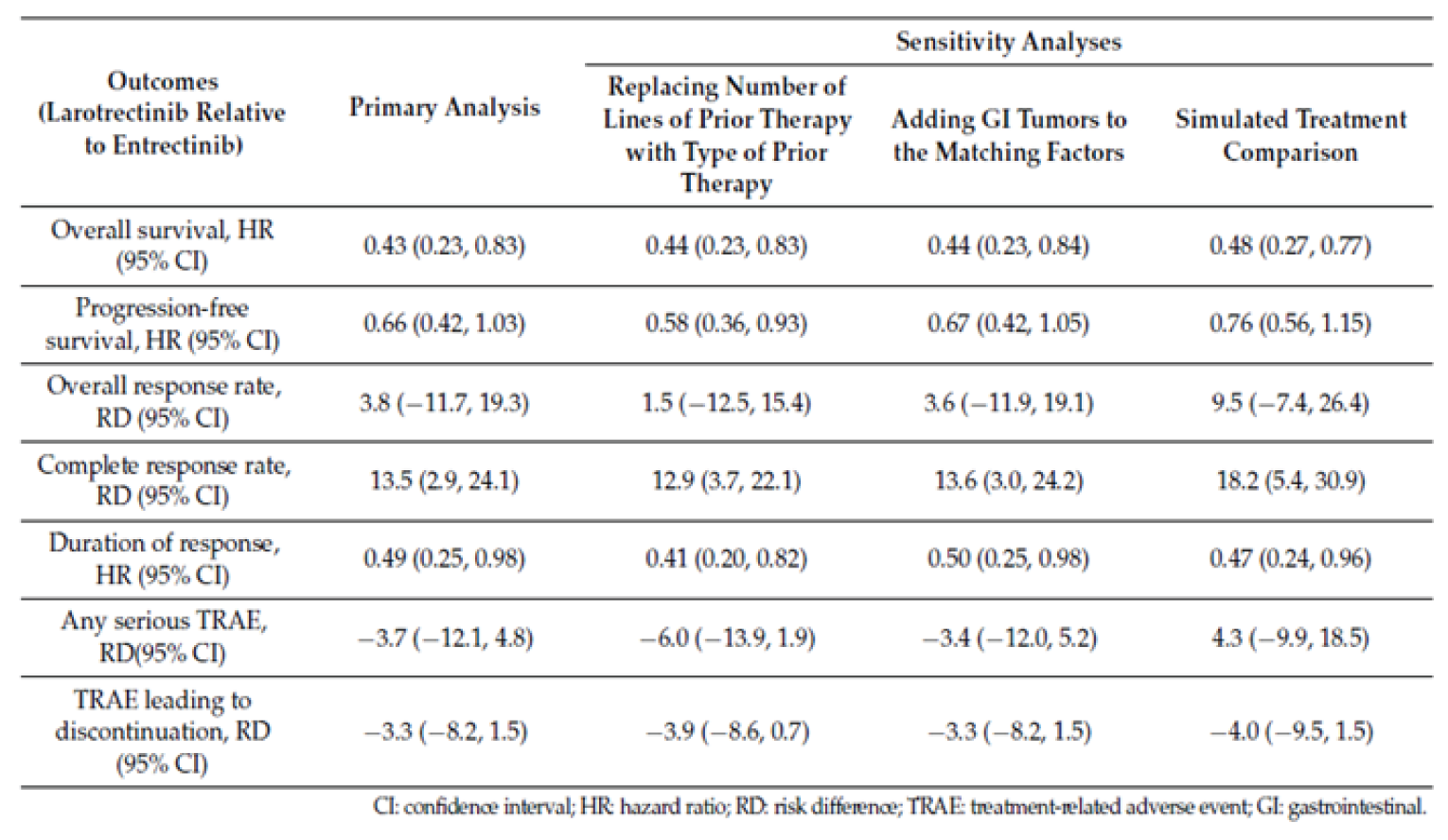
CI = confidence interval; GI = gastrointestinal; HR = hazard ratio; RD = risk difference; TRAE = treatment-related adverse event.
Source: Garcia-Foncillas et al., 2022.86 © 2022 by the authors. Licensee MDPI, Basel, Switzerland. Reprinted in accordance with Attribution 4.0 International (CC BY 4.0) https://creativecommons.org/licenses/by/4.0/
Summary
Garcia-Foncillas et al. (2022) conducted a MAIC to compare the efficacy and safety of entrectinib and larotrectinib in adults with NTRK fusion–positive tumours. The MAIC was funded by the manufacturer of larotrectinib; therefore, patient-level data were available for the larotrectinib-treated patients, but not for the entrectinib-treated patients. The data used for entrectinib were derived from the earlier CCODs (May 31, 2018 and October 31, 2018) (i.e., a smaller sample size than the August 31, 2020 CCOD data included in the current submission to CADTH). Adult patients were selected for inclusion in the MAIC if they had NTRK fusion (as determined by BICR); had an ECOG PS of 2 or less; and were TRK inhibitor-naive. Patients were matched according to the following baseline characteristics: sex, age, race, ECOG PS, tumour type, metastatic disease (versus locally advanced, unresectable disease), NTRK fusion type, prior lines of systemic therapy for metastatic disease, and CNS metastases.
Garcia-Foncillas et al. (2022) reported that compared with entrectinib, larotrectinib was associated with a statistically significantly greater duration of OS (HR = 0.43; 95% CI, 0.23 to 0.83; P < 0.05) and DOR (HR = 0.49; 95% CI, 0.25 to 0.98; P < 0.05). The authors reported no statistically significant difference for PFS (HR = 0.66; 95% CI, 0.42 to 1.03; P = 0.07) or ORR (RD = 3.8; 95% CI, –11.7 to 19.3; P = 0.63). Results were similar in sensitivity analyses applying different specifications for the MAIC and using a simulated treatment comparison method. There were no statistically significant differences reported between larotrectinib and entrectinib for serious TRAEs or TRAEs leading to discontinuation.
Several key details from the MAIC were not provided in the published study. This absence limits the ability to appraise the reported study. However, the primary limitation of the results stems from the unanchored nature of the comparison; to ensure unbiased results, the comparison requires the inclusion of all prognostic factors and all effect modifiers. Due to this limitation and others, firm conclusions based on the results of this MAIC are not recommended.
Other Relevant Evidence
This section includes an additional relevant study included in the sponsor’s submission to CADTH that was considered to address important gaps in the evidence included in the systematic review.|
Intra-Patient Growth Modulation Index Analysis
The sponsor provided the intra-patient comparison of efficacy in a single-arm trial of entrectinib in tumour-agnostic indications. The sponsor’s objective was to generate and analyze evidence for the comparative effectiveness of entrectinib by exploring the role of intra-patient comparison as an alternative to a traditional comparator arm.
Populations and Methods
Analyses were conducted on retrospectively collected data from the STARTRK-2 trial to generate intra-patient comparisons. There were 3 cohorts of patients. Patients were assigned to cohorts based on their prior systemic therapy in the metastatic setting and the presence or absence of documented progression:
Patients in the “documented progression on prior therapy” cohort had received at least 1 systemic therapy for metastatic disease before commencing entrectinib and clear documentation of PD on the most recent prior therapy, as captured in electronic case report forms.
Patients in the “no documented progression on prior therapy” cohort had received at least 1 systemic therapy for metastatic disease before commencing entrectinib and had no documentation of PD on the most recent prior therapy. This cohort includes patients who stopped prior therapy due to toxicity, completion of the course, or other reasons.
Patients in the “no prior therapy” cohort had received no prior systemic therapy for metastatic disease before starting entrectinib. However, they may have received prior (neo) adjuvant therapy.
A total of 71 patients with efficacy-evaluable NTRK fusion–positive disease who were enrolled in the STARTRK-2 trial up to April 30, 2018 (data cut-off: October 31, 2018) were included in the analysis. Among these 71 patients, 51 patients had received systemic therapy before commencing entrectinib (38 had documented PD and 13 had no documented PD on the most recent prior systemic therapy); 20 patients had not received prior systemic therapy. Among those who had received prior systemic therapy, 21 patients (41.2%) had received 1 line, 20 patients (39.2%) had received 2 lines, and 10 patients (19.6%) had received 3 or more lines of therapy. The treatment regimens varied greatly within and between tumour types. The most common tumour types were sarcoma (22.5%), NSCLC (16.9%), MASC (16.9%), and thyroid cancer (9.9%).
The primary analysis used was the GMI, defined by the ratio of PFS on entrectinib to TTD on the most recent prior therapy. TTD was chosen instead of TTP to measure the efficacy of the prior therapy due to the limited data available to reliably define a TTP outcome. A GMI ratio of greater than or equal to 1.3 was selected as the threshold to indicate a clinically meaningful benefit based on previous evidence, including von Hoff.88 This threshold was also used in GMI analyses conducted for larotrectinib that were previously reviewed by CADTH.89
Additional analyses explored TTD and ORR for entrectinib and prior systemic therapy.
Interventions and Comparators
The analysis involved entrectinib (continuous 600 mg once daily) and all previous systemic therapies received in the metastatic setting.
Outcomes
The GMI was calculated as the ratio of PFS on entrectinib and TTD on the most recent prior therapy in the same patient.
For entrectinib:
TTD for any reason was defined as the time from the start of entrectinib therapy until the end of entrectinib therapy.
PFS was defined as the time from the first dose of entrectinib to the first documentation of radiographic PD or death due to any cause, whichever occurred first.
Response and PFS were assessed by BICR using RECIST 1.1.
For prior therapies:
TTD was defined as the time from the start of the most recent prior therapy until the end of the most recent prior therapy.
Response was assessed by the treating physician and recorded on the electronic case report form.
ORR was defined as the proportion of patients achieving a CR or PR.
Statistical Analysis
Kaplan–Meier methodology was used to explore the median TTD on entrectinib or on the most recent prior systemic therapy as well as median PFS on entrectinib in the cohort with documented PD on prior therapy. A Kaplan–Meier analysis of GMI (taking censoring into account) was also performed. Patients receiving ongoing entrectinib therapy were censored for TTD; patients who had not progressed or died were censored for PFS. The time-to-event analysis used Kaplan–Meier methods implemented with R statistical software. Further details regarding the methodology of the Kaplan–Meier estimation and/or how the HRs were calculated were not described in the submission materials.
TTD and ORR were further investigated for individual patients in all 3 cohorts for entrectinib and for the most recent prior systemic therapy in the prior systemic therapy cohorts.
Missing start dates for prior therapy were imputed as the earliest possible date (i.e., January 1 or the first day of the month); missing end dates for prior therapy were imputed as the latest possible date (i.e., December 31, end of the month, or the start of entrectinib).
Results
For GMI in patients with PD on prior systemic therapy, the median GMI was 2.53 (range = 0.09 to 61.5), with 25 patients (65.8%) having a GMI greater than or equal to 1.3. For GMI thresholds of greater than or equal to 1.5, greater than or equal to 1.8, and greater than or equal to 2.0, 23 patients (60.5%), 23 patients (60.5%), and 22 patients (57.9%), respectively, met these thresholds. Of 7 patients with a GMI of less than 1.0, 4 patients (57.1%) were censored for PFS.
A Kaplan–Meier survival analysis showed that curves for PFS and TTD on entrectinib were similar (HR of PFS to TTD = 1.08; 95% CI, 0.6 to 1.9), with a median PFS of 11.2 months (95% CI, 6.7 to NE) and a median TTD of 9.9 months (94% CI, 7.3 to 14.8) on entrectinib. Both PFS and TTD on entrectinib were longer than TTD on most recent prior therapy, which had a median of 2.9 months (95% CI, 2.0 to 4.9).
The ORRs for entrectinib were 60.5% (all PR) in patients with documented progression on prior therapy, 46.2% (all PR) in patients with no documented progression on prior therapy, and 80% (5 CR and 11 PR) in patients with no prior therapy. The ORRs for most recent prior systemic therapy were 15.8% (1 CR and 5 PR) in patients with documented progression on prior therapy and 7.7% (1 PR) in patients with no documented progression on prior therapy.
Figure 13: GMI for Individual Patients With Progression on Prior Therapy (Krebs et al. [2021])
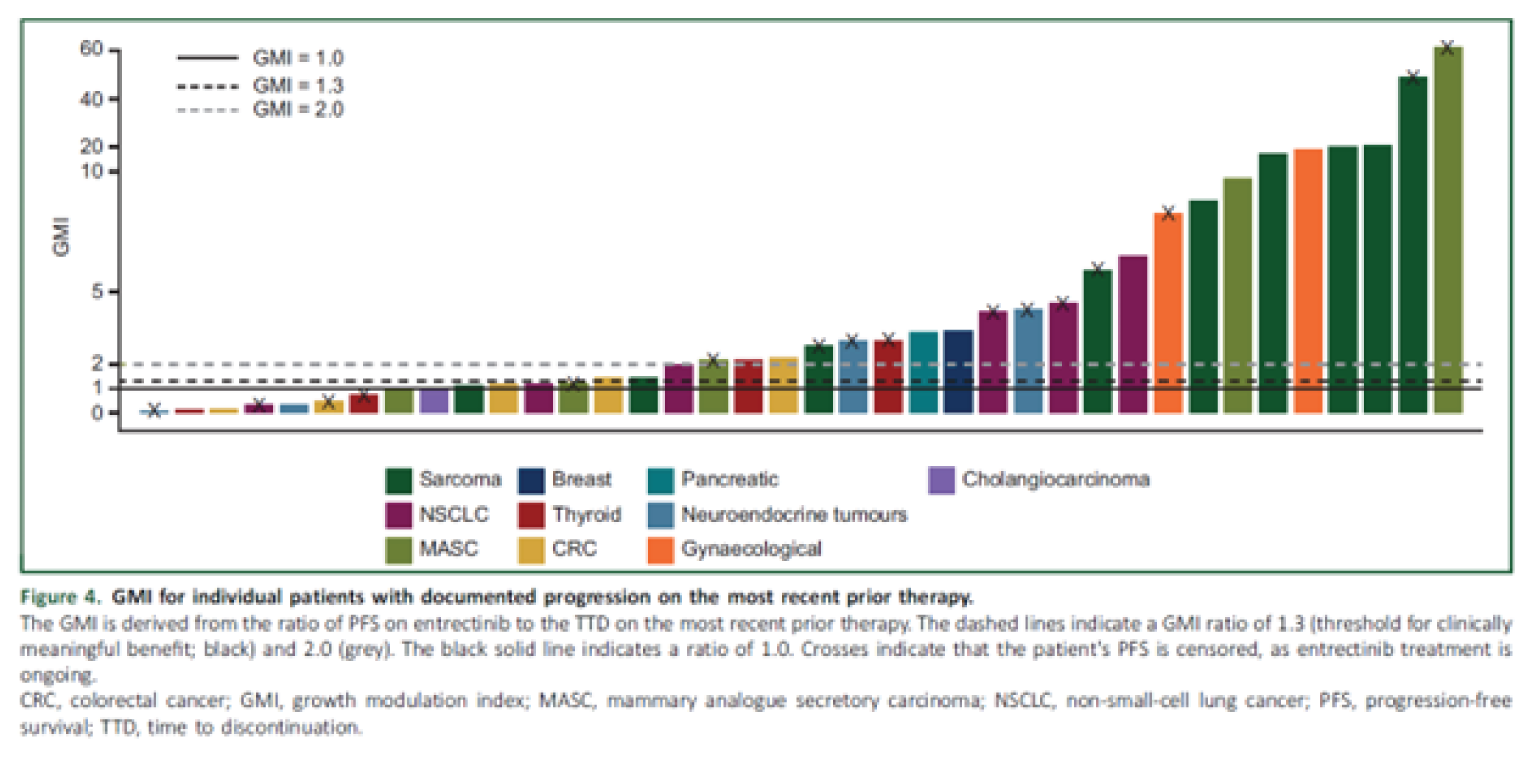
CRC = colorectal cancer; GMI = growth modulation index; MASC = mammary analogue secretory carcinoma; NSCLC = non–small cell lung cancer; PFS = progression-free survival; TTD = time to discontinuation.
Source: Krebs et al. (2021).90
2059 to 7029/© 2021 The Authors. Published by Elsevier Ltd. on behalf of European Society for Medical Oncology. This is an open access article under the CC BY licence (https://creativecommons.org/licenses/by/4.0/).
Figure 14: Kaplan–Meier Curves for TTD on Entrectinib Versus Most Recent Prior Systemic Therapy (Krebs et al. [2021])
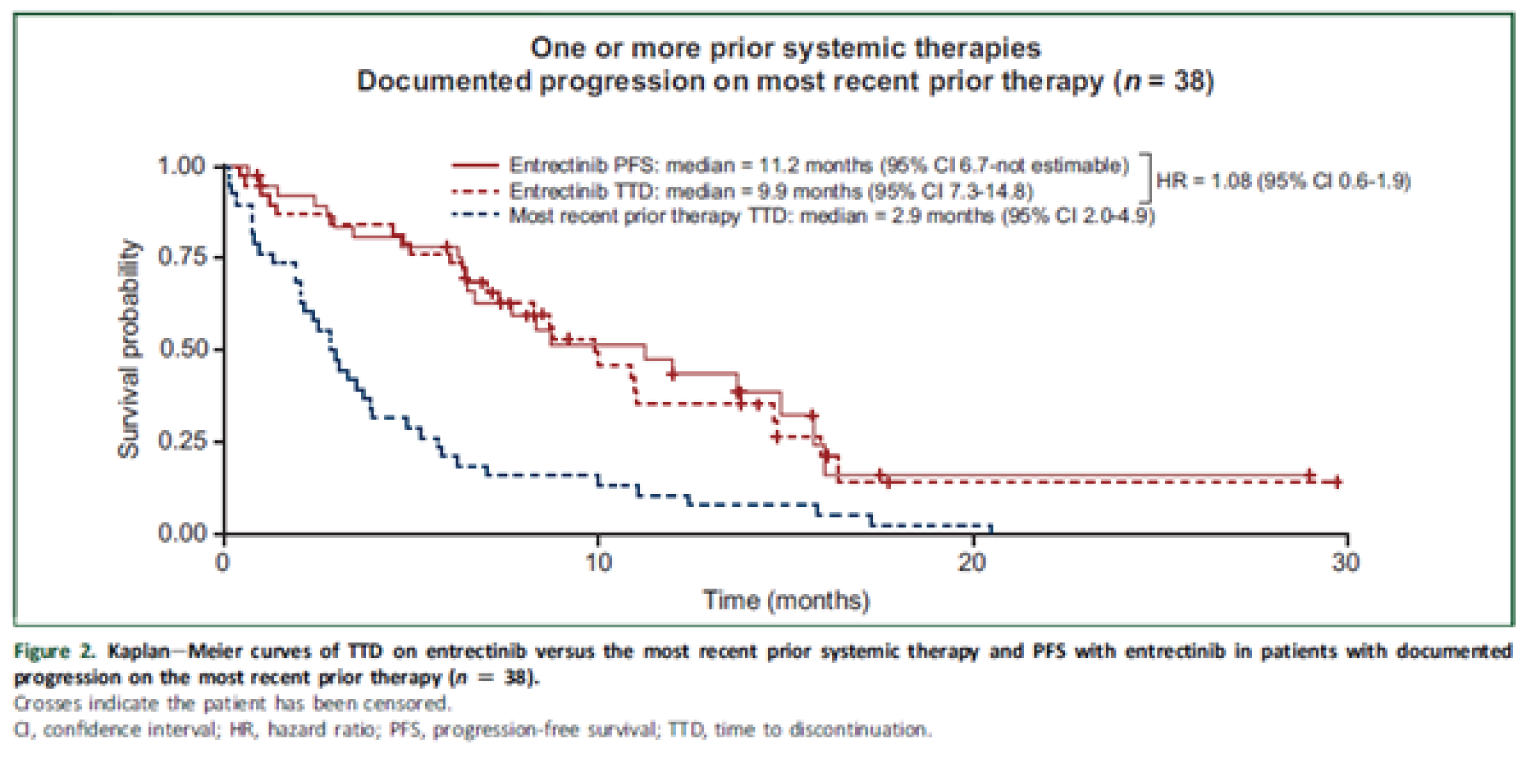
CI = confidence interval; HR = hazard ratio; PFS = progression-free survival; TTD = time to discontinuation.
Source: Krebs et al. (2021).90
2059 to 7029/© 2021 The Authors. Published by Elsevier Ltd. on behalf of European Society for Medical Oncology. This is an open access article under the CC BY licence (https://creativecommons.org/licenses/by/4.0/).91
Critical Appraisal
As the sponsor mentioned, the GMI (or PFS ratio) is not a validated efficacy end point and does not take into consideration the impact of treatment on a patient’s symptoms and quality of life. Moreover, in calculating the GMI, the sponsor could not obtain PFS data on prior systematic therapy and had to rely on TTD as a surrogate for PFS. This method becomes invalid if patients continue on prior therapy beyond progression, thereby overestimating PFS on prior therapy.
As the sponsor acknowledged, GMI analysis assumes that tumour growth follows linear kinetics over time (i.e., the same growth rate at the time of diagnosis, for prior therapies, and at time of entrectinib treatment). However, some evidence from tumour models suggests that tumour growth may be exponential or logarithmic.91 In addition, PFS is expected to decrease with successive lines of therapy.92 Taking all of these limitations together, the interpretation of the results from the GMI analysis becomes further complicated.
Two additional underlying assumptions of the GMI analysis are required to make this approach meaningful: participant characteristics with regard to all sources of heterogeneity of interest are consistent over time; and measurements compared in the ratio are compatible (i.e., PFS on entrectinib was defined and assessed in the same manner as TTD [as a surrogate for PFS] on most recent prior therapy) while all other reasons for an event are the same. However, neither condition has been met. It is unclear how patients’ baseline characteristics — such as age and ECOG PS, which change over time — affect disease progression. In addition, the timing of tumour assessment was controlled and standardized across patients while they were on entrectinib, but not for prior therapies; RECIST 1.1 was used to assess entrectinib response, but not for prior therapies; and PFS on entrectinib was analyzed by BICR whereas TTD on prior therapy was based on investigator assessment.
No actual statistical analysis was performed on individual GMI values. No CIs are provided for the medians (only minimum-maximum ranges), and it is unclear if the median is meaningfully greater than 1 or even 1.33. Due to censoring, the individual GMI values should not be used for rigorous statistical testing. Although the percentages of participants above certain cut-offs of GMI are presented, these are not accompanied by any statistical inference, measure of uncertainty, or CIs. Looking only at the estimated proportions that were reported among the groups with at least 7 patients (sarcoma, NSCLC, MASC, thyroid cancer), only the sarcoma group has at least 75% of patients with a GMI of 1.33 or greater. The other 3 groups did not, which suggests variation among the groups.
Summary
In summary, the results show a longer PFS on entrectinib relative to the TTD with the last prior treatment; however, this observation relies on many assumptions, including the key assumption (TTD as a surrogate for PFS), which appears to be invalid, based on the information provided about the calculation of the GMI. There is no formal investigation of differences in the GMI by tumour type or other patient characteristics, and the descriptive individual GMI results suggest large variations in the GMI. It is unclear how some of the presented results were obtained or if the inferences made with them are valid, given the intra-patient nature of the analysis. However, if the GMI can be considered a reliable comparison tool, then it appears to support the case that entrectinib may be beneficial in many of the tumour types when other treatments have failed, and that this is the case across many patient characteristics, mitigating many of the concerns about patient heterogeneity (other than tumour type). Without inference (the presented CI), large variation in GMI is evident across the tumour types and remains a main limitation.
Exploratory Efficacy Analyses Comparing Entrectinib Against Standard of Care
The sponsor provided a technical report comparing OS in patients with NTRK fusion–positive solid tumours who were treated with entrectinib from the sponsor’s clinical trials (pooled dataset) versus patients treated under standard of care from the FH and FMI clinico-genomic database.93
Populations and Methods
The FH database is a US longitudinal database with de-identified data originating from approximately 280 cancer clinics and representing 2.8 million patients with cancer (the majority from community oncology settings). The FH data platform aggregates and processes patient-level data. This sponsor-submitted study uses the FH pan-tumour clinico-genomic database, which includes all patients tested by FMI NGS-based comprehensive genomic profiling. The study populations are described in Table 31.
|||||||||||||||||||||||| | |||||||||||||||||||||||| |
|---|---|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |
CGDB = clinico-genomic database; FH = Flatiron Health; NGS = next-generation sequencing.
a|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Outcomes
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Statistical Analysis
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.
Patient Characteristics
The study population with the same tumour types consisted of |||||||| patients who received standard of care and |||||||| entrectinib-treated patients. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.
Table 32: Patient Characteristics in the Overall Population
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|---|---|---|---|---|
|||||||||||||||||||||||| | ||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | ||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | ||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | ||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | ||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | ||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | ||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | ||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | ||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | ||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | ||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | ||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | ||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | ||
CRC = colorectal cancer; CUP = carcinoma of unknown primary; ECOG PS = Eastern Cooperative Oncology Group Performance Status; MSI-H = microsatellite instability high; MSS = microsatellite stable; NGS = next-generation sequencing; NSCLC = non–small cell lung carcinoma; SMD = standardized mean difference; Std = standard deviation.
Source: Perez, L. and D. Hibar (2021). Update of the comparative efficacy analyses of NTRK fusion–positive solid tumours between entrectinib-treated patients from Roche trial (pooled dataset) and patients treated under standard of care from the Flatiron/FMI clinico-genomic database [sponsor-submitted report].93
Matching
Patient characteristics and standardized mean difference before and after matching are summarized in Table 33 and Table 34, respectively. When the 2 cohorts were matched, the main analyses included only |||||| trial patients matched to |||||| standard or care patients, and ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.
Table 33: Patient Characteristics and Standardized Mean Difference Before Matching
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|---|---|---|---|---|---|
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
SMD = standardized mean difference.
Source: Perez, L. and D. Hibar (2021). Update of the comparative efficacy analyses of NTRK fusion–positive solid tumours between entrectinib-treated patients from Roche trial (pooled dataset) and patients treated under standard of care from the Flatiron/FMI clinico-genomic database [sponsor-submitted report].93
Table 34: Patient Characteristics and Standardized Mean Difference After Matching
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |
|---|---|---|---|---|
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |
|||||||||||||||||||||||| | ||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | ||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | ||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | ||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | ||||
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
|||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| | |||||||||||||||||||||||| |
NSCLC = non–small cell lung carcinoma; SMD = standardized mean difference.
Source: Perez, L. and D. Hibar (2021). Update of the comparative efficacy analyses of NTRK fusion–positive solid tumours between entrectinib-treated patients from Roche trial (pooled dataset) and patients treated under standard of care from the Flatiron/FMI clinico-genomic database [sponsor-submitted report].93
Results
The median OS in entrectinib-treated patients was ||||||||||||||||||||||||||||||||||||||||||||||||, and the OS for the standard-of-care patients was ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. The HR for the matched population was |||||||||||||||||||||||||||||||||||||||||||||||| for the treatment of entrectinib versus standard of care, as shown in Figure 16.
In the sensitivity analysis that was adjusted for ECOG PS at the time of index, |||||||||||| standard-of-care patients could be matched to |||||| entrectinib-treated trial patients; the HR for entrectinib versus standard of care was ||||||||||||||||||||||||. In the subgroup analysis of patients receiving palliative care (after the NGS test and with tumours matching those in the trial), | standard-of-care patients could be matched to |||||| entrectinib-treated patients (||||||||||||||||||||||||||||||||||||||).

Confidential figure removed at sponsor’s request.
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Source: Perez, L. and D. Hibar (2021). Update of the comparative efficacy analyses of NTRK fusion–positive solid tumours between entrectinib-treated patients from Roche trial (pooled dataset) and patients treated under standard of care from the Flatiron/FMI clinico-genomic database [sponsor-submitted report].93
Critical Appraisal
The following important limitations prevent firm conclusions from being drawn based on the results of this analysis:
The sample size for the comparison was very small, with only |||||| patients included in the standard-of-care group. CADTH acknowledges that this is a rare condition; however, this remains an important limitation.
There was heterogeneity across the entrectinib and standard-of-care groups even after propensity score matching.
The groups were matched based solely on |||||| characteristics, which is not sufficient to control for potential confounding factors.
There were missing values in relevant covariates, such as ECOG PS (|||||| missing in the standard-of-care patients) and number of metastatic sites (under-reported in the real-world evidence data) that prevented their inclusion in the a priori matching.
The sample size was too small to allow for exploration by subgroup of tumour types or lines of prior therapy (both of which were identified as subgroups of interest for CADTH’s review).
Summary
The results show a longer median OS for patients treated with entrectinib compared with patients receiving standard of care (|||||||||||||||||||||||||||||||||||||||||||||||||||||| versus ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||). Important limitations with this analysis, including the small sample size and heterogeneity across treatment groups, prevent firm conclusions from being drawn.
Discussion
Summary of Available Evidence
The submission for entrectinib was based on a pooled analysis of 3 multi-centre, open-label, single-arm trials of entrectinib in adults with advanced or metastatic solid tumours: ALKA (phase I), STARTRK-1 (phase I), and STARTRK-2 (ongoing phase II basket trial). The pooled analysis has been updated several times to reflect larger sample sizes and longer-term follow-ups. The primary analysis submitted to Health Canada had a CCOD of May 31, 2020; these data are reflected in the current Canadian product monograph for entrectinib (N = 54 for efficacy). Subsequent updates were conducted based on CCODs of October 31, 2018 (N = 74 for efficacy) and August 31, 2020 (N = 121 for efficacy). Whenever available, the CADTH report reflects the August 31, 2020 CCOD. The relevant datasets in the pooled analysis for the August 31, 2020 CCOD consisted of: the NTRK safety-evaluable population (N = 193; 97% from STARTRK-2), which included all patients with an NTRK fusion–positive tumour who received at least 1 dose of entrectinib; the NTRK efficacy-evaluable population (N = 121; 98% from STARTRK-2), which included all patients with NTRK fusion–positive extracranial primary tumours who received at least 1 dose of entrectinib, had measurable disease at baseline, and had at least 12 months of follow-up; and the NTRK efficacy-evaluable population of patients with CNS metastases at baseline (N = 19 based on BICR assessment), which included the subpopulation of patients that was used for the evaluation of the intracranial efficacy end points.
The primary efficacy outcomes in the pooled analysis were ORR, DOR, and BOR. Secondary efficacy end points included TTR, CBR, PFS, and OS. In addition, the sponsor pre-specified the following intracranial efficacy end points, which were evaluated in the pooled subset of patients who had CNS metastases at baseline: IC-ORR, IC-DOR, and IC-PFS. HRQoL data were evaluated only in the STARTRK-2 trial and included change from baseline in the QLQ-C30; the QLQ-LC13 for the subset of patients with NSCLC; and the QLQ-CR29 for the subset of patients with mCRC.
The NTRK efficacy-evaluable analysis set (N = 121) was 51.2% female, with a mean age of 55.9 years (64.5% of patients were less than 65 years of age). Baseline ECOG PS scores were 0 (43.8%), 1 (47.1%), or 2 (9.1%). The majority of patients had received some form of prior anti-cancer therapy (n = 97; 80.2%), with 74 patients (61.2%) receiving any prior radiotherapy and 103 patients (85.1%) having previous cancer surgery. Just under a third of patients (30.6%) did not have prior systemic anti-cancer therapy. For those with prior systemic therapy, 28.9% had 1 line, 21.5% had 2 lines, 9.9% had 3 lines, and 5.8% had 4 lines of systemic therapy. The most frequent systemic prior anti-cancer therapy was chemotherapy (n = 88; 72.7%), followed by targeted therapy (n = 24; 19.8%), immunotherapy (n = 13; 10.7%), and hormonal therapy (n = 10; 8.3%). The solid tumour types that were reported for at least 5% of patients included sarcoma (n = 26; 21.5%), MASC (n = 24; 19.8%), NSCLC (n = 22; 18.2%), thyroid cancer (n = 13; 10.7%), colon cancer (n = 10; 8.3%), and breast cancer (n = 7; 5.8%). Nearly all patients had metastatic disease at baseline (96.7%); the most common metastatic sites were lung (61.2%) and lymph nodes (55.4%).
Nearly all patients in the pooled analysis were from the STARTRK-2 trial, in which patients received entrectinib at the dosage recommended in the Canadian product monograph (i.e., 600 mg once daily to start, with up to 2 dose reductions permitted to manage AEs). The clinical experts consulted by CADTH indicated that this is an accurate reflection of how entrectinib is likely to be used in clinical practice.
In addition to the pooled analysis, CADTH included additional studies to examine important gaps in the evidence. This included 1 indirect comparison of entrectinib versus larotrectinib, 1 intra-patient comparison of entrectinib versus traditional comparator treatments, and 1 comparison of entrectinib versus standard of care.
Interpretation of Results
Efficacy
The primary outcome in the pooled analysis was ORR assessed by a blinded independent review committee. This was considered a clinically relevant outcome and an appropriate assessment of response in the NTRK-positive patient population with heterogeneous primary tumours, according to the clinicians consulted by CADTH for this review. The regulatory approval of entrectinib by Health Canada was based on the May 31, 2018 CCOD (N = 54), with an ORR of 57% (95% CI, 43% to 71%) and a median DOR of 10.4 months. Health Canada concluded that the effect of entrectinib was clinically meaningful for patients with NTRK fusion–positive, unresectable or metastatic extracranial solid tumours who had experienced disease progression following systemic therapy for their disease.46 Results from the updated August 31, 2020 dataset demonstrated an ORR of 61.2% (95% CI, 51.87 to 69.88) for the overall population, with 15.7% and 45.5% of patients demonstrating a complete or a PR, respectively. The median DOR at the time of the updated analysis filed with CADTH was 20 months (95% CI, 13.0 to 38.2). The clinical experts consulted by CADTH indicated that the effective size for the overall trial population should be considered clinically meaningful.
In contrast to larotrectinib, entrectinib is not indicated for use in patients with primary CNS tumours; however, entrectinib is indicated for use in patients with NTRK-positive extracranial solid tumours who have brain metastases.34,35 CNS metastases are common in some metastatic tumour types, with a prevalence of NTRK gene fusions (most notably NSCLC, CRC, and breast cancer).59 Patients with CNS metastases have a worse prognosis and are affected by symptoms that result in a significant impairment of their quality of life. Patient group input noted the hope that entrectinib could be effective for those who have developed CNS metastases. There was no evidence identified comparing the efficacy of entrectinib for the treatment of CNS metastases versus alternative approaches currently used in clinical practice (e.g., neurosurgical resection, whole brain radiation therapy, stereotactic radiosurgery).
The pooled analyses included 19 patients who had CNS metastases at baseline (i.e., 15.7% of the total population); there were several pre-specified end points to evaluate the intracranial efficacy of entrectinib in these patients. Response rates were similar for those with CNS metastases at baseline compared with the overall population (ORR = 63.2%; 95% CI, 38.36 to 83.71), albeit with greater uncertainty in the estimates due to the small sample size. The median PFS and OS durations for patients with CNS metastases at baseline were 11.7 months (95% CI, 4.7 to 30.2) and 19.9 months (95% CI, 7.9 to NE), respectively. The clinical experts consulted by CADTH indicated that the intracranial efficacy results are clinically important, particularly given that these patients typically have poor prognoses.
The median time to response was 1 month (95% CI, 0.9 to 1.0). This would correspond with the first scheduled follow-up assessment at 4 weeks. The clinical experts noted this relatively fast median time to response, along with the high rate of response and favourable toxicity profile, as key considerations for potentially making entrectinib available to patients with an ECOG PS of 3 (a population that was not studied in the clinical trials) in situations where the oncologist believes that tumour-related symptoms are driving the PS.
At data cut-off, 72 out of 121 efficacy-evaluable patients (59.5%) had experienced disease progression or died. Median PFS and OS durations were 13.8 months (95% CI, 10.1 to 19.9) and 33.8 months (95% CI, 23.4 to 46.4), respectively. Health Canada and the FDA noted that the time-to-event end points are not interpretable without a control group; therefore, PFS, OS, and time to CNS progression cannot be considered in the definitive analysis to support efficacy.45,46 Recognizing the limitations of the data due to the small sample size, heterogenous patient population, and lack of a comparator group, caution and clinical judgment must be used in interpreting the PFS and OS results. The clinical experts considered the PFS and OS to be clinically meaningful in a population with advanced solid tumours. In addition, the clinical experts noted that, due to the rarity of NTRK fusion mutations, it would be challenging to conduct large clinical trials.
The clinical experts consulted by CADTH noted that entrectinib should be considered early during NTRK fusion cancer treatment. This was based on the rationale that NTRK fusion is the oncogenic driver in these tumours. Treatments targeting the tumour site as opposed to the TRK were perceived as likely to be less effective while being potentially more toxic than TRK-targeting therapies (i.e., entrectinib or larotrectinib), particularly for tumours where the alternative is chemotherapy. This perspective is also reflected in the 2021 Canadian Consensus for Biomarker Testing and Treatment of TRK Fusion Cancer in Adults, which generally recommends TRK-targeting therapy early in the course of treatment, when accessible.28 Similar commentary was provided by patient groups, which felt that earlier access to NTRK fusion testing may have allowed them to avoid treatments with more harmful side effects. Although interpreting the evidence from subgroups within the pooled analysis is challenging due to small sample sizes and heterogeneity across tumour types, the clinical experts noted that the ORR observed across subgroups based on the number of prior treatment regimens suggests that entrectinib may be more efficacious when used earlier in the course of treatment.
Across tumour types, the point estimates of the ORR ranged widely, and the CIs reflected a high degree of uncertainty around the point estimate of the ORR for many tumour types. At least 1 patient demonstrated a response to treatment in each of the tumour types, with the exception of neuroblastoma (n = 1), for which the sponsor reported that the sole patient included in the trial died 3 days after treatment initiation due to an unrelated AE.59 The sample sizes of the tumour subgroups ranged from single patients (cholangiocarcinoma, adenocarcinoma of the upper GI tract, and neuroblastoma) to 36 patients (sarcoma). In light of the small sample sizes and wide CIs, no meaningful conclusions can be drawn about the efficacy of entrectinib in different types of solid tumours. As noted in CADTH’s review of larotrectinib for NTRK-positive tumours, it is not methodologically meaningful to evaluate the ORR (and by extension, the effectiveness) of TRK inhibitors on different cancer types separately. The experts emphasized that the response rates observed across the tumour sites may be influenced by the number of prior therapies, which will vary considerably across the different tumours. For example, there are many treatments approved for mCRC, but comparatively fewer for sarcoma. This may be further influenced by differential timing with identification of the NTRK fusion in different tumour types (an important consideration in the Canadian context, given the variability in access to NTRK fusion testing across jurisdictions and by tumour types). Despite these limitations, the clinical experts agreed that the ORR observed with entrectinib across a wide range of tumours was consistent, and not previously seen with available therapies. This is particularly meaningful within the population of patients for whom there are no effective systemic treatment options available and/or for whom prognosis is poor.
As part of the conditions for marketing authorization, the sponsor is mandated to provide additional evidence with a sufficient number of patients to more precisely characterize response and durability of response for each of the following tumour types: CRC, gynecological cancers, and melanoma.84 Subgroup analyses showed a lower response rate among patients with mCRC (2 patients out of 10 patients; ORR = 20.0%; 95% CI, 2.5 to 55.6). There were only 2 patients with gynecological cancers and none with melanoma in the updated August 31, 2020 CCOD.11
There was no evidence identified in this review to evaluate the efficacy of sequential usage of larotrectinib and entrectinib following disease progression or lack of response to 1 of the drugs; however, some of the patients who participated in the call for patient group input noted exposure to sequential therapy.
Quality of Life
HRQoL was an exploratory objective in the STARTRK-2 study, measured by the QLQ-C30, QLQ-LC13, and QLQ-CR29. HRQoL analyses were conducted for only 1 of the trials (STARTRK-2) and were limited by the open-label administration of entrectinib, lack of comparator group, absence of statistical testing, high dropout rates at later assessment time points, and small sample size for the disease-specific instruments (e.g., NSCLC [N = 12] and mCRC [N = 7]).
Comparative Efficacy
No studies have directly compared entrectinib versus larotrectinib for patients with NTRK-positive tumours. The sponsor did not include an indirect comparison in its application to CADTH because it believes it is not feasible to conduct meaningful comparisons due to the following challenges: NTRK fusions are only expressed in up to 1% of all solid tumours; patient enrolment in trials is low; ongoing trials are single arm and open label, and the study population is heterogeneous with regard to baseline characteristics (e.g., age, ECOG PS, tumour site, and presence of CNS metastases). In the absence of direct or indirect evidence comparing entrectinib and larotrectinib in the submission, CADTH conducted a literature search to identify any relevant published indirect comparisons, and identified 1 MAIC that compared entrectinib and larotrectinib in adult patients with NTRK gene fusion–positive tumours.86 Garcia-Foncillas et al. (2022) reported that, compared with entrectinib, larotrectinib was associated with a statistically significantly greater duration of OS and DOR, with statistically significant differences for PFS or ORR.86 However, due to major limitations associated with this study, firm conclusions for the efficacy and safety end points comparing entrectinib and larotrectinib are not recommended.
The sponsor provided an intra-patient comparison to generate and analyze evidence for the comparative effectiveness of entrectinib versus the patient’s most recent prior systemic therapy. The results show a longer PFS with entrectinib relative to the TTD with the last prior treatment; however, this observation relies on many assumptions, including the key assumption (i.e., TTD as a surrogate for PFS), which appears to be invalid, based on the information provided about the calculation of the GMI. There is no formal investigation of differences in the GMI by tumour type or other patient characteristics, and the descriptive individual GMI results suggest large variations in the GMI. It is unclear how some of the presented results were obtained or if inferences made with them are valid, given the intra-patient nature of the analysis. However, if the GMI can be considered a reliable comparison tool, it appears to support the case that entrectinib may be beneficial in many of the tumour types when other treatments have failed, and that this is the case across many patient characteristics, mitigating many of the concerns about patient heterogeneity, other than tumour type. Without inference (the presented CI), a large variation in GMI is evident across the tumour types and remains a main limitation.
The sponsor provided a report comparing OS in patients with NTRK fusion–positive solid tumours who were treated with entrectinib from the sponsor’s clinical trials (pooled dataset) versus patients treated under standard of care from the FH and MI clinico-genomic database. The results show a longer median OS for patients treated with entrectinib compared with patients who received standard of care (20.9 months [95% CI, 16.8 to NE] versus 11.7 months [95% CI, 3.7 to NE]; HR = 0.46 [95% CI, 0.19 to 1.09; P = 0.0311]). Important limitations with this analysis, including the small sample size and heterogeneity across treatment groups, prevent firm conclusions from being drawn.
Harms
The product monograph for entrectinib provides detailed recommendations for the management of AEs that require temporary interruption, dose reduction, or discontinuation of treatment with entrectinib. The clinical experts consulted by CADTH indicated that this a reasonable reflection of how patients would be managed in clinical practice. From the starting dose of 600 mg once daily, the dose can be reduced twice to assist in the management of AEs: first to 400 mg once daily and then to 200 mg once daily. This approach is similar to that shown on the product monograph for larotrectinib; however, the dosage of larotrectinib can be reduced 3 times as a result of AEs (i.e., from a starting dose of 100 mg twice daily, patients can have sequential reductions to 75 mg twice daily, to 50 mg twice daily, and finally to 100 mg once daily). The clinical experts consulted by CADTH noted that this difference is unlikely to be a major consideration for physicians when deciding between these 2 TRK-targeting therapies; however, the approach could evolve as more experience is gained in clinical practice.
The product monograph for entrectinib includes black box warnings that the drug may cause congestive heart failure and, when administered to a pregnant woman, fetal harm. The product monograph for larotrectinib does not contain any black box warnings, although there is a statement regarding potential risk of fetal harm when larotrectinib is administered to a pregnant woman. The clinical experts consulted by CADTH noted that patients would likely be screened and monitored for risk factors and symptoms related to heart failure before treatment and during follow-up visits while on treatment. The influence of the safety profile differences between entrectinib and larotrectinib on prescriber choice could vary depending on individual physician preferences, but the experts consulted by CADTH did not feel that these differences were likely to influence their decision for the majority of patients. The clinical experts noted that, should a patient experience intolerance to larotrectinib and/or entrectinib, there could be interest in switching to the alternative TRK-targeting therapy. (This possibility could be of particular interest to patients who experience the known AEs with entrectinib that are not documented in the product monograph for larotrectinib.)
Health Canada concluded that the safety profile of entrectinib was considered acceptable in the setting of a life-threatening disease for which no satisfactory treatment options are available; however, given that entrectinib is not a selective inhibitor of the TRK proteins, its safety profile is a result of a number of off-target effects in addition to those known to be related to inhibition of the TRK proteins; thus, it is broader than that of larotrectinib, a selective inhibitor of the TRK proteins.46 As part of the conditional market authorization for entrectinib, the sponsor is required to provide additional integrated safety analyses to Health Canada. These are to include further characterization of off-target receptors, transporters, and/or channels that may be activated or inhibited by entrectinib, as well as further characterization of potential cardiac risks and risks of fractures and support labelling instructions for dose modification and monitoring.84
Garcia-Foncillas et al. (2022) reported that the MAIC demonstrated no statistically significant differences between larotrectinib and entrectinib for serious TRAEs or TRAEs leading to discontinuation.86 As previously noted, firm conclusions should not be drawn from this MAIC regarding the comparative safety of entrectinib and larotrectinib due to important limitations with the analysis.
The Krebs et al. (2021) intra-patient comparison did not include an evaluation of any safety end points.90
Other Considerations
The indication for entrectinib approved by Health Canada for use in the treatment of NTRK fusion tumours is more restrictive than the indications approved by the European Medicines Agency and FDA, neither of which exclude usage in patients with primary CNS tumours or in pediatric patients.94,95 The sponsor’s initial submission to Health Canada contained an extrapolation package to support a similar benefit-risk profile of entrectinib for use in pediatric patients. Based on the limited data submitted, the safety of entrectinib in pediatric patients has not been established; therefore, Health Canada has not authorized an indication for pediatric use at this time. In addition, there is no indication for primary CNS tumours, given that the efficacy of entrectinib in primary brain tumours has not been established.96
Conclusions
The clinical data supporting the efficacy of entrectinib in a histology-agnostic patient population of adults with NTRK fusion–positive cancer are derived from a pooled analysis of 3 open-label, single-arm trials, including 2 phase I trials (ALKA and STARTRK-1) and a phase II ongoing basket trial (STARTRK-2). In total, 121 and 193 adults with NTRK-positive cancer of different histologies were included in the most recent pooled analyses of efficacy and safety, respectively. Results showed that treatment with entrectinib was associated with an ORR of 61.2% (95% CI, 51.87 to 69.88), with 15.7% and 45.5% of patients demonstrating CR and PR, respectively. The median time to response was 1 month (95% CI, 0.9 to 1.0), and the median DOR was 20 months (95% CI, 13.0 to 38.2). Across different tumour types, the ORR varied widely, with a similarly wide range of uncertainty. Combined with the differences in sample sizes across the tumour types, the majority of which had been experienced by fewer than 10 patients, these factors limit the generalizability of the findings to the mixed cancer population. Among patients with CNS metastases at baseline (N = 19), the sponsor conducted analyses examining the intracranial efficacy of entrectinib with respect to CNS lesions. The IC-ORR was 52.6% (95% CI, 28.9 to 75.6), with a median IC-DOR of 17.2 months (95% CI, 7.4 to NE) and a median IC-PFS of 10.1 months (95% CI, 6.3 to 26.7).
While the rarity of NTRK fusion creates practical challenges in the conduct of a randomized controlled trial, the methodological limitations of single-arm trials with small sample sizes mean the results should be interpreted based on clinical judgment. The clinical experts consulted by CADTH indicated that the response rates reported for entrectinib are clinically meaningful, particularly for those with CNS metastases at baseline, given that those patients typically have a poor prognosis. The comparative evidence included in this review for entrectinib versus larotrectinib or standard therapies has important methodological limitations and is insufficient for drawing conclusions about comparative efficacy.
The clinical experts consulted by CADTH noted that patients would likely be screened and monitored for risk factors in accordance with the recommendations in the product monograph (e.g., risk of congestive heart failure) and that AEs would likely be managed in accordance with the dosage interruption and reduction scenarios reported in the product monograph. Overall, the clinical experts noted that the AE profile of entrectinib was acceptable and that for patients who have advanced disease, entrectinib may be more tolerable than some alternatives (e.g., chemotherapy or radiation). Patient groups similarly noted a preference for targeted therapy and a desire to avoid systemic therapies that may be associated with greater toxicity. From a regulatory perspective, the sponsor is required to provide additional integrated safety analyses to Health Canada as part of the conditional market authorization (i.e., NOC/c); this will include further characterization of the off-target pharmacodynamics of entrectinib (given that entrectinib is not a selective inhibitor of the TRK proteins) as well as further characterization of potential cardiac risks and risks of fractures and support labelling instructions for dose modification and monitoring.
References
1.Kheder ES, Hong DS. Emerging Targeted Therapy for Tumors with NTRK Fusion Proteins. Clinical Cancer Research. 2018;24(23):5807-5814. PubMed
2.Arevalo JC, Conde B, Hempstead BL, Chao MV, Martin-Zanca D, Perez P. TrkA immunoglobulin-like ligand binding domains inhibit spontaneous activation of the receptor. Mol Cell Biol. 2000;20(16):5908–5916. PubMed
3.Canadian Cancer Statistics Advisory Committee. Canadian cancer statistics 2019. In: Toronto (ON): Canadian Cancer Society; 2019. Accessed 2021 Feb 1.
4.Vaishnavi A, Le AT, Doebele RC. TRKing down an old oncogene in a new era of targeted therapy. Cancer Discov. 2015;5(1):25-34. PubMed
5.Vaishnavi A, Capelletti M, Le AT, et al. Oncogenic and drug-sensitive NTRK1 rearrangements in lung cancer. Nat Med. 2013;19(11):1469-1472. PubMed
6.Farago AF, Le LP, Zheng Z, et al. Durable Clinical Response to Entrectinib in NTRK1-Rearranged Non-Small Cell Lung Cancer. J Thorac Oncol. 2015;10(12):1670-1674. PubMed
7.Pagani F, Randon G, Guarini V, et al. The Landscape of Actionable Gene Fusions in Colorectal Cancer. Int J Mol Sci. 2019;20(21):5319. PubMed
8.Pietrantonio F, Di Nicolantonio F, Schrock AB, et al. ALK, ROS1, and NTRK rearrangements in metastatic colorectal cancer. J Natl Cancer Inst. 2017;109(12). PubMed
9.Gatalica Z, Xiu J, Swensen J, Vranic S. Molecular characterization of cancers with NTRK gene fusions. Mod Pathol. 2019;32(1):147-153. PubMed
10.Farago AF, Azzoli CG. Beyond ALK and ROS1: RET, NTRK, EGFR and BRAF gene rearrangements in non-small cell lung cancer. Transl Lung Cancer Res. 2017;6(5):550-559. PubMed
11.Hoffmann-La Roche Ltd. Supplemental Efficacy Results (Clinical cut-off date: August 31, 2020) [internal sponsor's report]. In: San Diego, CA: Hoffmann-La Roche Ltd; 2020.
12.Stransky N, Cerami E, S. S, Kim JL, Lengauer C. The landscape of kinase fusions in cancer. Nat Commun. 2014;5:4846. PubMed
13.Rosen EY, Schram AM, Young RJ, et al. Larotrectinib demonstrates CNS efficacy in TRK fusion-positive solid tumors. JCO Precis Oncol. 2019;3. PubMed
14.Hetchman JF, Benayed R, Hyman DM, et al. Pan-TRK immunohistochemistry is an efficient and reliable screen for the detection of NTRK fusions. Am J Surg Pathol. 2017;41(11):1547-1551. PubMed
15.Bazhenova L, Jiao X, Lokker A, et al. Cancers with NTRK gene fusions: molecularcharacteristics and prognosis. Clinical Cancer Research. 2020;26(12 Suppl 1).
16.Drilon A, Laetsch TW, Kummar S, et al. Efficacy of larotrectinib in TRK fusion-positive cancers in adults and children. N Engl J Med. 2018;378(8):731-739. PubMed
17.Shaw AT, Yeap BY, Mino-Kenudson M, et al. Clinical features and outcome of patients with non-small-cell lung cancer who harbor EML4-ALK. J Clin Oncol. 2009;27(26):4247-4253. PubMed
18.Wang R, Hu H, Pan Y, et al. RET fusions define a unique molecular and clinicopathologic subtype of non-small-cell lung cancer. J Clin Oncol. 2012;30(35):4352-4359. PubMed
19.Bergethon K, Shaw AT, Ou SH, et al. ROS1 rearrangements define a unique molecular class of lung cancers. J Clin Oncol. 2012;30(8):863-870. PubMed
20.Center for Drug Evaluation Research. Multi-disciplinary review(s). Vitrakvi (larotrectinib) capsules. Company: BAYER HLTHCARE. Application No.: 210861 & 211710. Approval date: 11/26/2018 (FDA drug approval package). In: Silver Spring (MD): U. S. Food and Drug Administration (FDA); 2018: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2018/210861Orig1s000_21171Orig1s000TOC.cfm. Accessed 2021 Mar 19.
21.Bourgeois JM, Knezevich SR, Mathers JA, Sorensen PH. Molecular detection of the ETV6-NTRK3 gene fusion differentiates congenital fibrosarcoma from other childhood spindle cell tumors. Am J Surg Pathol. 2000;24(7):937-946. PubMed
22.Tognon C, Knezevich SR, Huntsman D, et al. Expression of the ETV6-NTRK3 gene fusion as a primary event in human secretory breast carcinoma. Cancer cell. 2002;2(5):367-376. PubMed
23.Bishop JA, Yonescu R, Batista D, Begum S, Eisele DW, Westra WH. Utility of mammaglobin immunohistochemistry as a proxy marker for the ETV6-NTRK3 translocation in the diagnosis of salivary mammary analogue secretory carcinoma. Human pathology. 2013;44(10):1982-1988. PubMed
24.Ricarte-Filho JC, Li S, Garcia-Rendueles ME, et al. Identification of kinase fusion oncogenes in post-Chernobyl radiation-induced thyroid cancers. The Journal of clinical investigation. 2013;123(11):4935-4944. PubMed
25.Prasad ML, Vyas M, Horne MJ, et al. NTRK fusion oncogenes in pediatric papillary thyroid carcinoma in northeast United States. Cancer. 2016;122(7):1097-1107. PubMed
26.Solomon JP, Linkov I, Rosado A, et al. NTRK fusion detection across multiple assays and 33,997 cases: diagnostic implications and pitfalls. Mod Pathol. 2020;33(1):38-46. PubMed
27.European Medicines Agency. Committee for Medicinal Products for Human Use (CHMP) assessment report - Rozlytrek. In: Amsterdam (NL): European Medicines Agency; 28 May 2020: https://www.ema.europa.eu/en/documents/assessment-report/rozlytrek-epar-public-assessment-report_en.pdf. Accessed 2022 Mar 15.
28.Bebb DG, Banerji S, Blais N, et al. Canadian Consensus for Biomarker Testing and Treatment of TRK Fusion Cancer in Adults. Curr Oncol. 2021;28(1):523-548. PubMed
29.Clark MA, Fisher C, Judson I, Thomas JM. Soft-tissue sarcomas in adults. N Engl J Med. 2005;353(7):701-711. PubMed
30.Hemming M, Heinrich M, Bauer S, George S. Translational insights into gastrointestinal stromal tumor and current clinical advances. Annals of Oncology. 2018;29(10):2037-2045. PubMed
31.Wu Y-L, Planchard D, Lu S, et al. Pan-Asian adapted clinical practice guidelines for the management of patients with metastatic non-small-cell lung cancer: a CSCO–ESMO initiative endorsed by JSMO, KSMO, MOS, SSO and TOS. Annals of Oncology. 2019;30(2):171-210. PubMed
32.Hoffmann-La Roche Ltd. Hoffmann-La Roche Ltd response to May 9, 2022 request for additional information regarding Rozlytrek (entrectinib) reimbursement review [internal additional sponsor's information]. In: Mississauga (ON): Hoffmann-La Roche Ltd; 2022.
33.CADTH. CADTH Drug Reimbursement Expert Review Committee final recommendation: larotrectinib (Vitrakvi) (Bayer Inc.). In: Ottawa (ON): CADTH; 2022: https://www.cadth.ca/sites/default/files/DRR/2021/PC0221%20Vitrakvi%20-%20CADTH%20Final%20Rec%20KG_NA_Corrected-meta.pdf. Accessed 2022 Mar 23.
34.Hoffmann-La Roche Ltd. Rozlytrek (entrectinib): capsules, 100 mg and 200 mg, oral [product monograph]. In: Mississauga (ON): Hoffmann-La Roche Limited; 2021 Jan 7.
35.Bayer Inc. Vitrakvi (larotrectinib): oral capsules, 25 mg and 100 mg; oral solution 20 mg/mL [product monograph]. In: Mississauga (ON): Bayer Inc; January 24, 2022: https://pdf.hres.ca/dpd_pm/00064510.PDF.
36.McGowan J, Sampson M, Salzwedel DM, Cogo E, Foerster V, Lefebvre C. PRESS Peer Review of Electronic Search Strategies: 2015 guideline statement. Journal of clinical epidemiology. 2016;75:40-46. PubMed
37.Grey matters: a practical tool for searching health-related grey literature. In: Ottawa (ON): CADTH; 2019: https://www.cadth.ca/grey-matters. Accessed 2022 Feb 1.
38.Braud FGD, Niger M, Damian S, et al. Alka-372-001: First-in-human, phase I study of entrectinib – an oral pan-trk, ROS1, and ALK inhibitor – in patients with advanced solid tumors with relevant molecular alterations. J Clin Oncol. 2015;33(15_suppl):2517-2517.
39.Braud FGD, Pilla L, Niger M, et al. Phase 1 open label, dose escalation study of RXDX101, an oral pan-trk, ROS1, and ALK inhibitor, in patients with advanced solid tumors with relevant molecular alterations. J Clin Oncol. 2014;32(15_suppl):2502-2502.
40.Patel MR, Bauer TM, Liu SV, et al. STARTRK1: Phase 1/2a study of entrectinib, an oral PanTrk, ROS1, and ALK inhibitor, in patients with advanced solid tumors with relevant molecular alterations. Journal of Clinical Oncology Conference. 2015;33(15 SUPPL. 1).
41.Drilon A, De Braud FG, Siena S, et al. Entrectinib, an oral pan-Trk, ROS1, and ALK inhibitor in TKI-naive patients with advanced solid tumors harboring gene rearrangements: updated phase I results. Cancer research Conference: 107th annual meeting of the american association for cancer research, AACR. 2016;76(14 Supplement).
42.Drilon A, Siena S, Ou SI, et al. Safety and Antitumor Activity of the Multitargeted Pan-TRK, ROS1, and ALK Inhibitor Entrectinib: Combined Results from Two Phase I Trials (ALKA-372-001 and STARTRK-1). Cancer Discov. 2017;7(4):400-409. PubMed
43.Paz-Ares L, Barlesi F, Siena S, et al. Patient-reported outcomes from STARTRK-2: a global phase II basket study of entrectinib for ROS1 fusion-positive non-small-cell lung cancer and NTRK fusion-positive solid tumours. Esmo Open. 2021;6(3):100113. PubMed
44.Drilon A, Sankhala KK, Liu SV, et al. STARTRK-2: A global phase 2, open-label, basket study of entrectinib in patients with locally advanced or metastatic solid tumors harboring TRK, ROS1, or ALK gene fusions. Cancer Research Conference: American Association for Cancer Research Annual Meeting. 2017;77(13 Supplement 1).
45.Center for Drug Evaluation Research. NDA/BLA Multidisciplinary Review and Evaluation NDA 212725. Rozlytrek (entrectinib). In: Silver Spring (MD): U.S. Food and Drug Administration; 2019: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2019/212725Orig1s000,%20212726Orig1s000MultidisciplineR.pdf. Accessed 2022 Mar 15.
46.Health Canada reviewer's report: Rozlytrek (entrectinib) [internal sponsor's report]. In: Drug reimbursement review sponsor submission: Rozlytrek (entrectinib), capsules, 100 mg and 200 mg, oral [internal sponsor's package]. Mississauga (ON): Hoffman-La Roche Limited; 2022 Jan 25. In.
47.Hoffmann-La Roche Ltd. Clinical Study Report: ALKA-372-001. A Phase 1, Dose Escalation Study of Entrectinib (RXDX-101) In Adult Patients With Advanced/Metastatic Solid Tumors [internal sponsor's report]. In: San Diego, CA: Hoffmann-La Roche Ltd; 2018.
48.Hoffmann-La Roche Ltd. Clinical Study Report: STARTRK-1. A Phase 1, Multicenter, Open-Label Study of Oral Entrectinib (RXDX-101) in Adult Patients With Locally Advanced or Metastatic Cancer Confirmed to be Positive for NTRK1, NTRK2, NTRK3, ROS1, or ALK Molecular Alterations [internal sponsor's report]. In: San Diego, CA: Hoffmann-La Roche Ltd,; 2018.
49.Hoffmann-La Roche Ltd. Interim Clinical Study Report: STARTRK-2. An Open-Label, Multicenter, Global Phase II Basket Study of Entrectinib for the Treatment of Patients with Locally Advanced or Metastatic Solid Tumors that Harbor NTRK1/2/3, ROS1, or ALK Gene Rearrangements [internal sponsor's report]. In: San Diego, CA: Hoffmann-La Roche Ltd; 2018.
50.Hoffmann-La Roche Ltd. Drug reimbursement review sponsor submission: Rozlytrek (entrectinib), capsules, 100 mg and 200 mg, oral [internal sponsor's package]. In: Mississauga (ON): Hoffman-La Roche Limited; 2022 Jan 25.
51.Demetri GD, De Braud F, Drilon A, et al. Updated Integrated Analysis of the Efficacy and Safety of Entrectinib in Patients with NTRK Fusion-Positive Solid Tumors. Clinical cancer research: an official journal of the American Association for Cancer Research. 2022;10.
52.Doebele RC, Drilon A, Paz-Ares L, et al. Entrectinib in patients with advanced or metastatic NTRK fusion-positive solid tumours: integrated analysis of three phase 1-2 trials. Lancet Oncol. 2020;21(2):271-282. PubMed
53.Bazhenova L, Liu SV, Lin JJ, et al. Efficacy and safety of entrectinib in patients with locally advanced/metastatic NTRK fusion-positive (NTRK-fp) solid tumours. . ESMO Congress; 2021.
54.Doebele RC, Drilon A, Paz-Ares L, et al. Entrectinib in patients with advanced or metastatic NTRK fusion-positive solid tumours: integrated analysis of three phase 1-2 trials. Lancet Oncol. 2019;11:11. PubMed
55.Clinical Study Report RXDX-101-01 (STARTRK-1) A Phase 1, Multicenter, Open-label Study of Oral Entrectinib (RXDX-101) in Adult Patients with Locally Advanced or Metastatic Cancer Confirmed to be Positive for NTRK1, NTRK2, NTRK3, ROS1, or ALK Molecular Alterations [internal sponsor's report]. In: Mississauga (ON): F. Hoffman-La Roche Ltd.; 2018 Oct.
56.Interim Clinical Study Report - GO40782 (STARTRK-2): An Open-Label, Multicenter, Global Phase II Basket Study of Entrectinib for the Treatment of Patients with Locally Advanced or Metastatic Solid Tumors that Harbor NTRK1/2/3, ROS1, or ALK Gene Rearrangements [internal sponsor's report]. In: Mississauga (ON): Hoffman La-Roche Ltd.; 2018 Nov.
57.Hoffman-La Roche Ltd. Section 2.5 - Clinical Overview In: Drug reimbursement review sponsor submission: Rozlytrek (entrectinib), capsules, 100 mg and 200 mg, oral [internal sponsor's package]. Mississauga (ON): Hoffman-La Roche Limited; 2022 Jan 25. In.
58.Clinical Study Report GO40784 (ALKA-372-001) A Phase 1, Dose Escalation Study of Entrectinib (RXDX-101) In Adult Patients With Advanced/ Metastatic Solid Tumors [internal sponsor's report]. In: Mississauga (ON): Hoffman-La Roche Ltd.; 2018 Oct.
59.Hoffmann-La Roche Ltd. ROZLYTREK® (entrectinib) Clinical Summary for the Treatment of Patients with NTRK fusion-positive solid tumours [internal sponsor's package]. In: Mississauga (ON): Hoffman-La Roche Limited; December 2021.
60.Protocol GO40784 (RXDX-101-01) A Phase 1, Multicenter open-label study of oral ebtrectinib (RXDX-101) in adult patients with locally advanced or metastatic cancer confirmed to be positive for NTRK1, NTRK2, NTRK3, ROS1, or ALK molecular alterations [internal sponsor's report]. In: Mississauga (ON): Hoffman-La Roche Ltd.; 2019 Feb.
61.Hoffmann-La Roche Ltd. Common Technical Document Section 2.7.3 In: Drug reimbursement review sponsor submission: Rozlytrek (entrectinib), capsules, 100 mg and 200 mg, oral [internal sponsor's package]. Mississauga (ON): Hoffman-La Roche Limited; 2022 Jan 25. In.
62.Hoffmann-La Roche Ltd. Statistical Analysis Plan: Evaluation of the Efficacy and Safety of Oral Entrectinib (RXDX-101) for the Treatment of Patients With Locally Advanced or Metastatic Solid Tumors That Harbor NTRK Gene Fusions [internal sponsor's report]. In: San Diego, CA: Hoffmann-La Roche Ltd; 2018.
63.Ignyta. Statistical Analysis Plan: Evaluation of the Efficacy and Safety of Oral Entrectinib (RXDX-101) for the Treatment of Patients With Locally Advanced or Metastatic Solid Tumors That Harbor NTRK Gene Fusions August 10 2018. In:2018.
64.Hoffman La-Roche Ltd. Rozlytrek (Entrectinib): Economic Evaluation for the Treatment of NTRK Fusion-Positive, Locally Advanced or Metastatic Solid Tumours. In:2019.
65.Hoffmann-La Roche Ltd. Hoffmann-La Roche Ltd response to March 17, 2022 request for additional information regarding Rozlytrek (entrectinib) reimbursement review [internal additional sponsor's information]. In: Mississauga (ON): Hoffmann-La Roche Ltd; 2022.
66.FDA. Master Protocols: Efficient Clinical Trial Design Strategies to Expedite Development of Oncology Drugs and Biologics Guidance for Industry DRAFT GUIDANCE https://www.fda.gov/media/120721/download. In:2018.
67.Protocol GO40783 (ALKA-372-001) A phase I dose-escalation study of entrectinib (RXDX-101) in adult patients with advanced/metastatic solid tumors [internal sponsor's report]. In: Mississauga (ON): Hoffman-La Roche Ltd.; 2019 Mar.
68.Protocol GO40782 (RXDX-101-02) An open-label, multicenter, global phase 2 basket study of entrectinib for the treatment of patients with locally advanced or metastatic solid tumors that harbor NTRK1/2/3, ROS1, OR ALK gene rearrangements [internal sponsor's report]. In: Mississauga (ON): Hoffman La-Roche Ltd.; 2019 May.
69.Hoffmann-La Roche response to pCODR checkpoint meeting questions on Rozlytrek (entrectinib) for ROS-1-positive NSCLC. Mississauga (ON): Hoffmann-La Roche; 2019. In.
70.Hoffmann-La Roche response to pCODR checkpoint meeting questions on Rozlytrek (entrectinib) for ROS-1-positive NSCLC. Mississauga (ON): Hoffmann-La Roche; 2020.
71.Hoffmann-La Roche Ltd. Hoffmann-La Roche Ltd response to March 29, 2022 request for additional information regarding Rozlytrek (entrectinib) reimbursement review [internal additional sponsor's information]. In: Mississauga (ON): Hoffmann-La Roche Ltd; 2022.
72.Hoffmann-La Roche Ltd. Hoffmann-La Roche Ltd response to April 4, 2022 request for additional information regarding Rozlytrek (entrectinib) reimbursement review [internal additional sponsor's information]. In: Mississauga (ON): Hoffmann-La Roche Ltd; 2022.
73.Hoffmann-La Roche Ltd. Safety Update Report for Entrectinib November 2019 (Clinical cutoff date: 31 October 2018) [internal sponsor's report]. In: San Diego, CA: Hoffmann-La Roche Ltd; 2019.
74.Cocco E, Scaltriti M, Drilon A. NTRK fusion-positive cancers and TRK inhibitor therapy. Nat Rev Clin Oncol. 2018;15(12):731-747. PubMed
75.Okamura K, Harada T, Wang S, et al. Expression of TrkB and BDNF is associated with poor prognosis in non-small cell lung cancer. Lung Cancer. 2012;78(1):100-106. PubMed
76.Musholt TJ, Musholt PB, Khaladj N, Schulz D, Scheumann GFW, Klempnauer J. Prognostic significance of RET and NTRK1 rearrangements in sporadic papillary thyroid carcinoma. Surgery. 2000;128(6):984-993. PubMed
77.Tiacci E, Trifonov V, Schiavoni G, et al. BRAF mutations in hairy-cell leukemia. The New England journal of medicine. 2011;364(24):2305-2315. PubMed
78.Flaherty KT, Puzanov I, Kim KB, et al. Inhibition of mutated, activated BRAF in metastatic melanoma. The New England journal of medicine. 2010;363(9):809-819. PubMed
79.Renfro LA, Sargent DJ. Statistical controversies in clinical research: basket trials, umbrella trials, and other master protocols: a review and examples. Annals of oncology: official journal of the European Society for Medical Oncology. 2017;28(1):34-43. PubMed
80.Prahallad A, Sun C, Huang S, et al. Unresponsiveness of colon cancer to BRAF(V600E) inhibition through feedback activation of EGFR. Nature. 2012;483(7387):100-103. PubMed
81.Hyman DM, Puzanov I, Subbiah V, et al. Vemurafenib in Multiple Nonmelanoma Cancers with BRAF V600 Mutations. The New England journal of medicine. 2015;373(8):726-736. PubMed
82.U.S. Food and Drug Administration. Master Protocols: Efficient Clinical Trial Design Strategies to Expedite Development of Oncology Drugs and Biologics Guidance for Industry In:2022: https://www.fda.gov/media/120721/download. Accessed 2022 Mar 3.
83.Beckman R, Antonijevic Z, Kalamegham R, Chen C. Adaptive Design for a Confirmatory Basket Trial in Multiple Tumor Types Based on a Putative Predictive Biomarker. Clin Pharmacol Ther. 2016;100(6):617-625. PubMed
84.Health Canada. Rozlytrek - Notice of Compliance with Conditions - Qualifying Notice. https://www.canada.ca/en/health-canada/services/drugs-health-products/drug-products/notice-compliance/conditions/rozlytrek-qualifying-notice.html. Published 2020. Accessed April 22, 2022.
85.Chu P, Batson S, Hodgson M, Mitchell CR, Steenrod A. Systematic review of neurotrophic tropomyosin-related kinase inhibition as a tumor-agnostic management strategy. Future oncology (London, England). 2020;16(4):61-74. PubMed
86.Garcia-Foncillas J, Bokemeyer C, Italiano A, et al. Indirect Treatment Comparison of Larotrectinib versus Entrectinib in Treating Patients with TRK Gene Fusion Cancers. Cancers. 2022;14(7):31. PubMed
87.Phillippo D, Ades T, Dias S, Palmer S, Abrams KR, Welton N. NICE DSU Technical Support Document 18: Methods for population-adjusted indirect comparisons in submissions to NICE. In: University of Bristol; 2016: https://research-information.bris.ac.uk/ws/portalfiles/portal/94868463/Population_adjustment_TSD_FINAL.pdf. Accessed 2022 May 26.
88.Von Hoff DD. There are no bad anticancer agents, only bad clinical trial designs- twenty-first Richard and Hinda Rosenthal foundation award lecture. Clinical Cancer Research. 1998;4:1079-1086. https://www.scopus.com/inward/record.uri?eid=2-s2.0-0031896592&partnerID=40&md5=8004bcaa1728811c657a543ee8f8afbf. Published 5. Accessed 2022 Jun 9.
89.CADTH. CADTH Clinical and Pharmacoeconomic Review: larotrectinib (Vitrakvi) (Bayer Inc.). In: Ottawa (ON): CADTH; 2022: https://www.cadth.ca/sites/default/files/DRR/2021/PC0221-Vitrakvi-combined-report-meta.pdf. Accessed March 23, 2022.
90.Krebs MG, Blay JY, Le Tourneau C, et al. Intrapatient comparisons of efficacy in a single-arm trial of entrectinib in tumour-agnostic indications. ESMO Open. 2021;6(2):100072. PubMed
91.Le Tourneau C, Paoletti X, Coquan E, Sablin MP, Zoubir M, Tannock IF. Critical evaluation of disease stabilization as a measure of activity of systemic therapy: Lessons from trials with arms in which patients do not receive active treatment. J Clin Oncol. 2014;32(3):260-263. PubMed
92.Bailey CH, Jameson G, Sima C, et al. Progression-free Survival Decreases with Each Subsequent Therapy in Patients Presenting for Phase I Clinical Trials. J. 2012;3:7-13. PubMed
93.Perez L, Hibar D. Update of the comparative efficacy analyses of NTRK fusion-positive solid tumors between entrectinib treated patients from Roche trial (pooled dataset) and patients treated under standard of care from the Flatiron/FMI clinico-genomic database. In: Drug reimbursement review sponsor submission: Rozlytrek (entrectinib), capsules, 100 mg and 200 mg, oral [internal sponsor's package]. Mississauga (ON): Hoffman-La Roche Limited; 2022 Jul 25. In:2021.
94.European Medicines Agency. Summary of Product Characteristics - Rozlytrek. In: Amsterdam (The Netherlands): European Medicines Agency; 2020: https://www.ema.europa.eu/en/documents/product-information/rozlytrek-epar-product-information_en.pdf. Accessed April 22, 2022.
95.Center for Drug Evaluation Research. Highlights of Prescribing Information - Rozlytrek. In:2019: https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/212726s000lbl.pdf. Accessed April 22, 2022.
96.Health Canada. Summary Basis of Decision - Rozlytrek - Health Canada. In:2020: https://hpr-rps.hres.ca/reg-content/summary-basis-decision-detailTwo.php?linkID=SBD00485.
97.Nicklasson M, Bergman B. Validity, reliability and clinical relevance of EORTC QLQ-C30 and LC13 in patients with chest malignancies in a palliative setting. Qual Life Res. 2007;16(6):1019-1028. PubMed
98.Aaronson NK, Ahmedzai S, Bergman B, et al. The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst. 1993;85(5):365-376. PubMed
99.Wong CK, Chen J, Yu CL, Sham M, Lam CL. Systematic review recommends the European Organization for Research and Treatment of Cancer colorectal cancer-specific module for measuring quality of life in colorectal cancer patients. Journal of clinical epidemiology. 2015;68(3):266-278. PubMed
100.Cocks K, King MT, Velikova G, et al. Evidence-based guidelines for interpreting change scores for the European Organisation for the Research and Treatment of Cancer Quality of Life Questionnaire Core 30. Eur J Cancer. 2012;48(11):1713-1721. PubMed
101.Maringwa JT, Quinten C, King M, et al. Minimal important differences for interpreting health-related quality of life scores from the EORTC QLQ-C30 in lung cancer patients participating in randomized controlled trials. Support Care Cancer. 2011;19(11):1753-1760. PubMed
102.Musoro JZ, Sodergren SC, Coens C, et al. Minimally important differences for interpreting the EORTC QLQ-C30 in patients with advanced colorectal cancer treated with chemotherapy. Colorectal Dis. 2020;22(12):2278-2287. PubMed
103.Bergman B, Aaronson NK, Ahmedzai S, Kaasa S, Sullivan M. The EORTC QLQ-LC13: a modular supplement to the EORTC Core Quality of Life Questionnaire (QLQ-C30) for use in lung cancer clinical trials. EORTC Study Group on Quality of Life. Eur J Cancer. 1994;30a(5):635-642. PubMed
104.van der Hout A, Neijenhuijs KI, Jansen F, et al. Measuring health-related quality of life in colorectal cancer patients: systematic review of measurement properties of the EORTC QLQ-CR29. Support Care Cancer. 2019;27(7):2395-2412. PubMed
105.Whistance RN, Conroy T, Chie W, et al. Clinical and psychometric validation of the EORTC QLQ-CR29 questionnaire module to assess health-related quality of life in patients with colorectal cancer. Eur J Cancer. 2009;45(17):3017-3026. PubMed
106.Giesinger JM, Kieffer JM, Fayers PM, et al. Replication and validation of higher order models demonstrated that a summary score for the EORTC QLQ-C30 is robust. Journal of clinical epidemiology. 2016;69:79-88. PubMed
107.Quality of Life FAQ. Brussels (BE): EORTC; 2021. In: https://qol.eortc.org/faq/. Accessed March 14, 2022.
108.EORTC QLQ-C30 scoring manual. Belgium (BE): EORTC; 2001. https://www.eortc.org/app/uploads/sites/2/2018/02/SCmanual.pdf. Accessed March 14, 2022.
109.Revicki D, Hays RD, Cella D, Sloan J. Recommended methods for determining responsiveness and minimally important differences for patient-reported outcomes. Journal of clinical epidemiology. 2008;61(2):102-109. PubMed
110.Lung Cancer (update of QLQ-LC13). Brussels (BE): EORTC. https://qol.eortc.org/questionnaire/qlq-lc29/. Accessed March 14, 2022.
111.Reeve BB, Wyrwich KW, Wu AW, et al. ISOQOL recommends minimum standards for patient-reported outcome measures used in patient-centered outcomes and comparative effectiveness research. Qual Life Res. 2013;22(8):1889-1905. PubMed
112.Koller M, Hjermstad MJ, Tomaszewski KA, et al. An international study to revise the EORTC questionnaire for assessing quality of life in lung cancer patients. Ann Oncol. 2017;28(11):2874-2881. PubMed
113.Ganesh V, Agarwal A, Popovic M, et al. Comparison of the FACT-C, EORTC QLQ-CR38, and QLQ-CR29 quality of life questionnaires for patients with colorectal cancer: a literature review. Support Care Cancer. 2016;24(8):3661-3668. PubMed
114.Gujral S, Conroy T, Fleissner C, et al. Assessing quality of life in patients with colorectal cancer: an update of the EORTC quality of life questionnaire. Eur J Cancer. 2007;43(10):1564-1573. PubMed
Appendix 1: Literature Search Strategy
Note that this appendix has not been copy-edited.
Clinical Literature Search
Overview
Interface: Ovid
Databases: MEDLINE All (1946-present) and Embase (1974-present). Note: Subject headings and search fields have been customized for each database. Duplicates between databases were removed in Ovid.
Date of search: February 23, 2022
Alerts: Bi-weekly search updates until project completion
Search filters applied: None
Limits: Conference abstracts: excluded
Syntax | Description |
|---|---|
/ | At the end of a phrase, searches the phrase as a subject heading |
MeSH | Medical Subject Heading |
.fs | Floating subheading |
exp | Explode a subject heading |
* | Before a word, indicates that the marked subject heading is a primary topic; or, after a word, a truncation symbol (wildcard) to retrieve plurals or varying endings |
# | Truncation symbol for one character |
? | Truncation symbol for one or no characters only |
adj# | Requires terms to be adjacent to each other within # number of words (in any order) |
.ti | Title |
.ot | Original title |
.ab | Abstract |
.hw | Heading word; usually includes subject headings and controlled vocabulary |
.kf | Keyword heading word |
.dq | Candidate term word (Embase) |
.pt | Publication type |
.mp | Mapped term |
.rn | Registry number |
.nm | Name of substance word (MEDLINE) |
.yr | Publication year |
.jw | Journal title word (MEDLINE) |
.jx | Journal title word (Embase) |
freq=# | Requires terms to occur # number of times in the specified fields |
medall | Ovid database code: MEDLINE All, 1946 to present, updated daily |
oemezd | Ovid database code; Embase, 1974 to present, updated daily |
Multi-Database Strategy
# Searches
(Rozlytrek* or entrectinib* or RXDX101 or RXDX-101 or NMSE628 or NMS E628 or NMS-E-628 or L5ORF0AN1I).ti,ab,kf,ot,hw,rn,nm.
1 use medall
*entrectinib/
(Rozlytrek* or entrectinib* or RXDX101 or RXDX-101 or NMSE628 or NMS E628 or NMS-E-628).ti,ab,kf,dq.
3 or 4
5 use oemezd
6 not (conference abstract or conference review).pt.
2 or 7
remove duplicates from 8
Clinical Trials Registries
ClinicalTrials.gov
Produced by the US National Library of Medicine. Targeted search used to capture registered clinical trials.
[Search terms -- Rozlytrek OR entrectinib OR RXDX101 OR RXDX-101 OR NMSE628 OR NMS E628 OR NMS-E-628]
WHO International Clinical Trials Registry Platform
International Clinical Trials Registry Platform, produced by the World Health Organization. Targeted search used to capture registered clinical trials.
[Search terms -- Rozlytrek OR entrectinib OR RXDX101 OR RXDX-101 OR NMSE628 OR NMS E628 OR NMS-E-628]
Health Canada’s Clinical Trials Database
Produced by Health Canada. Targeted search used to capture registered clinical trials.
[Search terms -- Rozlytrek OR entrectinib OR RXDX101 OR RXDX-101 OR NMSE628 OR NMS E628 OR NMS-E-628]
EU Clinical Trials Register
European Union Clinical Trials Register, produced by the European Union. Targeted search used to capture registered clinical trials.
[Search terms -- Rozlytrek OR entrectinib OR RXDX101 OR RXDX-101 OR NMSE628 OR NMS E628 OR NMS-E-628]
Grey Literature
Search dates: February 8 – 11, 2022
Keywords: Rozlytrek or entrectinib or RXDX101 or RXDX-101 or NMSE628 or NMS E628 or NMS-E-628 solid tumours, NTRK gene fusion, TRK fusion-positive cancers, Tissue-agnostic cancers
Limits: None
Updated: Search updated prior to the completion of stakeholder feedback period
Relevant websites from the following sections of the CADTH grey literature checklist Grey Matters: A Practical Tool for Searching Health-Related Grey Literature were searched:
Health Technology Assessment Agencies
Health Economics
Clinical Practice Guidelines
Drug and Device Regulatory Approvals
Advisories and Warnings
Drug Class Reviews
Clinical Trials Registries
Databases (free)
Health Statistics
Internet Search
Appendix 2: Description and Appraisal of Outcome Measures
Note that this appendix has not been copy-edited.
Aim
To describe the following outcome measures and review their measurement properties (validity, reliability, responsiveness to change, and MID):
EORTC QLQ-C30, EORTC QLQ-LC13 (lung cancer supplemental module), and EORTC QLQ-CR29 (CRC supplemental module) were PRO measures in the STARTRK-2 study.
Findings
Table 36: Summary of outcome measures and their measurement properties
Outcome measure | Type | Conclusions about measurement properties | MID |
|---|---|---|---|
EORTC QLQ-C30 | 30-item, patient-reported, cancer-specific, quality of life questionnaire using 4-point Likert scales and a 7-Likert scale. 4-point Likert scale
7-point Likert scale
Raw scores from each scale are converted to a 0-100 scale using a linear transformation, with a higher score reflecting better function on the function scale, higher symptom burden on the symptom scales, and better quality of life on global health status/QoL scale. | NSLCL Validity: A strong construct validity for the physical, role, social functions, fatigue symptom, and global quality of life has been demonstrated with WHO PS and a standard 6-minute walk test; between emotional function and HADS anxiety scale; between pain scale and BPI intensity and interference subscales.97 Also, construct validity with ECOG PS, weight loss, and WHO Acute and Subacute Toxicity scale with (some of) functional, symptom, and global QoL scale has been established.98 Reliability: Most of functional and some symptom scales, global QoL scale showed Cronbach alpha > 0.7.97 Internal consistency for pre-treatment and on-treatment periods ranged from alpha = 0.52 – 0.89.98 Responsiveness: Over 28-day period of pre- and on treatment, some functional and symptom scales, global QoL scale showed responsiveness as stratified by ECOG PS.98 CRC Limited evidence was found in patients with CRC.99 | Various cancers100: (improvement, deterioration)
NSCLC101:a
CRC102:a
|
EORTC QLQ-LC13 | 13-item, patient-reported, lung cancer-specific, quality of life questionnaire using a 4-point Likert scale (except for pain medication item, which is dichotomous – yes or no) to be administered with EORTC QLQ-C30. Symptoms: a. cough (1) b. hemoptysis (1) c. dyspnea (3) d. pain (3) e. pain medication (1) Treatment-related side effects: a. sore mouth or tongue (1) b. dysphagia (1) c. neuropathy (1) d. alopecia (1) All scale and item scores are linearly transformed to a 0-100 scale, with higher scores representing increased symptom burden. | Validity: Construct validity has been established between disease type and pain score (P < 0.001) as well as ECOG PS and dyspnea, coughing, pain scores (P < 0.001).103 Correlation between spirometry and dyspnea score was found to be weak (r = 0.24).97 BPI intensity score and pain score were found to be modest (r > 0.4).97 Reliability: Reliability coefficient (Cronbach alpha) range for dyspnea scores was 0.81-0.83,103 which was confirmed by another study with alpha = 0.76.97 Internal consistency for pain scale was found to be unacceptable (alpha = 0.53 – 0.54) when QLQ-LC13 was used alone but acceptable (alpha = 0.8) when administered with QLQ-C30.103 Responsiveness: Dyspnea, coughing, and pain scores improved over time between pre-treatment and on-treatment period (all P < 0.05). Responsiveness of chest pain, dyspnea and coughing to change in ECOG PS was also noted.103 | Not identified in patients with NSCLC. |
EORTC QLQ-CR29 | 29-item, patient-reported, colorectal cancer-specific, quality of life questionnaire using a 4-point Likert scale to be administered with EORTC QLQ-C30. Raw scores are linearly transformed to a score from 0 to 100 where higher functioning scores and lower symptom scores indicate better QoL. | Validity, reliability, responsiveness to change99,104,105: Evidence was of indeterminate quality in patients with CRC. | Not identified in patients with CRC. |
BPI = Brief Pain Inventory; CR29 = colorectal cancer – 29; CRC = colorectal cancer; ECOG PS = Eastern Cooperative Oncology Group Performance Status; EORTC QLQ-C30 = European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire – Core 30; HADS = Hospital Anxiety and Depression Scale; LC13 = lung cancer – 13; MID = minimal important difference; NSCLC = non–small cell lung cancer; QoL = quality of life; WHO = World Health Organization.
aFor select item, refer to the description for MIDs for all scales.
European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30
Description
The EORTC QLQ-C30 is among the most commonly used PRO measures in oncology clinical trials.106 It is a multi-dimensional, cancer-specific, evaluative measure of HRQoL. It was designed specifically for the purpose of assessing changes in participants’ HRQoL in clinical trials, in response to treatment.107 The questionnaire consists of 30 questions that are scored to create 5 multi-item functional scales (physical, role, cognitive, emotional, social), 3 multi-item symptom scales (fatigue, pain, nausea and vomiting), 6 single-item symptom scales (dyspnea, insomnia, appetite loss, constipation, diarrhea, financial impact), and a 2-item GHS/QoL scale. It is available in 90 different languages and is intended for use in adult populations only. Version 3.0 of the questionnaire is the most current version and has been in use since December of 1997.107
Table 37: Scales of EORTC QLQ-C30
Functional Scales (15 Questions) | Symptom Scales (7 Questions) | Single-Item Symptom Scales (6 Questions) | Global Quality of Life (2 Questions) |
|---|---|---|---|
Physical function (5) | Fatigue (3) | Dyspnea (1) | Global Quality of Life (2) |
Role function (2) | Pain (2) | Insomnia (1) | — |
Cognitive function (2) | Nausea and vomiting (2) | Appetite loss (1) | — |
Emotional function (4) | — | Constipation (1) | — |
Social function (2) | — | Diarrhea (1) | — |
— | — | Financial impact (1) | — |
Scoring
The EORTC QLQ-C30 uses a 1-week recall period in assessing function and symptoms. Most questions have 4 response options (“not at all,” “a little,” “quite a bit,” “very much”), with scores on these items ranging from 1 to 4. For the 2 items from the GHS/quality of life scale; however, the response format is a 7-point Likert-type scale, with anchors between 1 (very poor) and 7 (excellent).
Raw scores for each scale are computed as the average of the items that contribute to a particular scale. This scaling approach is based upon the assumption that it is appropriate to provide equal weighting to each item that comprises a scale. There is also an assumption that, for each item, the interval between response options is equal (for example, the difference in score between “not at all” and “a little” is the same as “a little” and “quite a bit,” at a value of 1 unit). Each raw scale score is converted to a standardized score that ranges from 0 to 100 using a linear transformation. Higher scores for the functioning scales and GHS/quality of life denote a better level of functioning/ quality of life (i.e., a better state of the patient), while higher scores on the symptom scales indicate a higher level of symptom burden (i.e., a worse state of the patient). According to the EORTC QLQ-C30’s scoring algorithm, if there are missing items for a scale, the score for the scale can still be computed if there are responses for at least half of the items. In calculating the scale score, the missing items are simply ignored — an approach that assumes that the missing items have values equal to the average of those items for what the respondent completed.108
Minimal Important Difference
A study by Cocks, et al.100 used a systematic review of the literature and experts’ opinion to evaluate meaningful differences and magnitude of changes, i.e., minimal important difference (MID), as defined by subtle but nevertheless clinically meaningful changes, in the QLQ-C30 scores. In a meta-analysis of 118 relevant papers (13.6% from US/Canada; 17.8% of lung cancer; 10.2% of CRC; other types of cancers include breast, head and neck, multiple, prostate, hematological, GI, brain, urology/kidney, testicular, gynecological) with timescales ranging from 4 days to 5 years, expert panel estimated trivial, small, medium, and large size classes for meaningful change in the scales. Given that medium and large changes could not be estimated for all scales due to insufficient data and response shift, i.e., psychological adaption of patients to their changing health status, small differences have been taken to represent the MIDs.
Estimated MIDs (improvement, deterioration) for EORTC QLQ-C30 in patients with various cancers100
Functional scales:
Physical function (2 to 7, -10 to -5)
Role function (6 to 12, -14 to -7)
Cognitive function (3 to 7, -7 to -1)
Emotional function (6 to 9, -12 to -3)
Social function (3 to 8, -11 to -6)
Symptom scales:
Fatigue (4 to 9, -10 to -5)
Pain (5 to 9, -11 to -3)
Nausea and vomiting (3 to 9, -11 to -5)
Single-item symptom scales:
Dyspnea (2 to 9, -11 to -5)
Insomnia (5 to 9, -9 to -2)
Appetite loss (7 to 13, -14 to -2)
Diarrhea (3 to 11, - 15 to -5)
Financial impact (> 3, -10 to -2)
Global quality of life: (5 to 8, -10 to -5)
Validity
Nicklasson et al.97 conducted a construct validity test with 112 Swedish patients diagnosed with lung cancer or pleural mesothelioma, including 85 (76%) patients with NSCLC, not amenable to curative or life prolonging treatment. The results were based on a known groups approach with WHO PS and a standard 6-minute walk test, and significant interaction effects were observed for physical, role, social function, global quality of life (P < 0.0001) and several symptom scales, the strongest seen with fatigue (P < 0.0001). When the known group was walk test, the strongest interaction effect was seen with physical function, followed by fatigue, role functioning, global quality of life and social functioning. In a correlation analysis employing walking distance (> 200m, n = 58) as a continuous variable, a strong correlation (r = 0.77) with physical functioning; substantial correlations (r > 0.4) with fatigue, role functioning and global quality of life; a modest correlation (r = 0.21) was seen with dyspnea. With the spirometry as a comparator, correlations with the single-item dyspnea were modest (r = 0.33).
According to Nicklasson, et al.,97 a strong correlation was seen between emotional functioning and the HADS (Hospital Anxiety and Depression Scale) anxiety scale (r = -0.75, P < 0.0001). The HADS depression scale correlated moderately (r > 0.4) with all functioning scales, fatigue (r = 0.52, P < 0.0001) and appetite loss (r = 0.48, P < 0.0001). Both BPI (Brief Pain Inventory) intensity and interference subscales correlated strongly with the pain scale (r = 0.72, P < 0.0001; r = 0.68, P < 0.0001, respectively).
Another group, Aaronson et al.,98 tested construct validity in 305 patients with nonresectable lung cancer (63.1% of NSCLC) undergoing either radiotherapy or chemotherapy from 13 countries including Canada. Based on a known groups approach, patients with better ECOG PS scores at the pre-treatment stage reported significantly higher QLQ-C30 scores for physical, role, cognitive function, overall quality of life and significantly lower symptom levels for 5 of 7 symptom scales (diarrhea and financial impact were excluded from analyses) (ANOVA n = 295, P < 0.001 to P < 0.05). While patients were on treatment, all functional scores and all symptom scores showed statistically significant group differences as expected according to their ECOG PS (ANOVA n = 265, P < 0.001 to P < 0.05). Similarly, statistically significant group differences were observed in pre-treatment physical, role function, overall quality of life, fatigue, dyspnea, appetite loss, and constipation, with patients having less weight loss as a known group reporting better functioning and lower symptom levels in expected direction (ANOVA n = 295, P < 0.001 to P < 0.05). Lastly, statistically significant group differences were observed in 5 of 6 functional scales and 5 of 7 symptom measures with WHO Acute and Subacute Toxicity Scales as known group variables while patients were being treated (ANOVA n = 244, P < 0.001 to P < 0.05).
Reliability
Nicklasson et al.97 performed reliability testing in the same population as described in the validity section. All functional scales except for cognitive function scale (alpha = 0.57), select symptom scales (fatigue, nausea and vomiting), and global quality of life scale showed an internal consistency of 0.70 or higher, which is accepted threshold for group comparisons.
Aaronson, et al.98 tested reliability in the same population as described in the validity section before and during treatment. The pre-treatment reliability coefficients for physical (alpha = 0.68 versus 0.71), role (alpha = 0.54 versus 0.52), cognitive (alpha = 0.56 versus 0.73), emotional (alpha = 0.73 versus 0.80) and social (alpha = 0.68 versus 0.77) functions were generally lower compared to those during treatment (pre- versus during treatment). The Cronbach alpha coefficients before treatment in symptom scales for fatigue (alpha = 0.80 versus 0.85), pain (alpha = 0.82 versus 0.76), nausea and vomiting (alpha = 0.65 versus 0.73) were also generally lower than those of during treatment (pre-treatment versus during treatment). The GHS showed alpha of 0.86 before treatment and 0.89 after treatment.
Responsiveness to Change
According to the Aaronson et al.98 ANOVA with divided patient samples based on ECOG PS (improved / decrease in score of at least 1 = 13%, unchanged = 57%, deteriorated / increase in score of at least 1 = 30% of patients), between-group differences over time (averaged 28 days, SD = 19 days) were statistically significant in physical (P < 0.001), role (P < 0.001), fatigue (P < 0.01), nausea and vomiting (P < 0.05), and global quality of life (P < 0.01) scales. Diarrhea and financial impact were excluded from analyses. No changes were noted in QLQ-C30 scores among those patients whose PS had remained unchanged.
Minimal Important Difference
Maringwa et al.101 estimated MIDs based on anchor- and distribution-based methods by pooling data from 2 RCTs on EORTC. Total 812 patients with palliative, locally advanced, and/or metastatic NSCLC that are undergoing treatment were enrolled. As for anchors chosen, physician-rated WHO PS and weight change were used based on their relevance to patients with NSCLC. For anchor-based MID estimates, improvement was considered to be 1 category change in PS, 5 - <20% weight gain and deterioration was considered to be 1 category change in PS, 5 - <20% weight loss. Effect size of 0.2 (SD), 0.5 SD, and threshold of 1 standard error of mean (SEM) of HRQoL scores have been reported as distribution-based MIDs to compare with the anchor-based MIDs.
Estimated anchor-based (improvement with PS, weight gain; deterioration with PS, weight loss) and distribution-based (0.2 SD, 0.5 SD, SEM) MIDs for EORTC QLQ-C30 in patients with NSCLC101
Physical function (due to differences in QLQ-C30 versions used in pooled data from 2 EORTC RCTs, analysis was restricted to trial 1 (version 3) data only): (9, 5; 4, 6) (5, 12, 7)
Role function:a (14, 7; 5, 5) (6, 17, 14)
Social function: (5, 7; 7, 9) (6, 14, 10)
Fatigue: (14, 5; 6, 11) (5, 13, 11)
Pain: (16, 2; 3,7), (6, 16, 12)
Global health scale: (9, 4; 4, 4) (4, 11, 9)
Other Considerations and Limitations
The limitation of MID estimation performed by Maringwa et al.101 is poor correlations between changes in either anchor (WHO PS or weight) and QLQ-C30. For example, for changes in GHS scores and changes in both anchors, the correlations coefficients range from 0.10 to 0.14 in absolute values. The Spearman rank correlation of at least 0.30 is suggested to be acceptable association.109
EORTC QLQ-C30 for patients with CRC
Validity, Reliability, and Responsiveness to Change
Wong et al.99 conducted a systematic review of HRQoL instruments used to assess patients with CRC, which examined the psychometric properties of the instruments. The level of evidence supporting the measurements was ranked on a 4-point Likert scale of “poor,” “fair,” “good,” or “excellent” and each instrument was given an overall rating of “unknown,” “limited,” “moderate,” or “strong.” In general, very few instruments demonstrated moderate or excellent evidence supporting the psychometric properties that were investigated. However, there was evidence identified for internal consistency, reliability, structural validity, a priori hypothesis testing, and responsiveness for the EORTC QLQ-C30.104
Minimal Important Difference
Musoro et al.102 compared EORTC QLQ-C30 data from 3 clinical trials (N = 1,491) to estimate MIDs of patients with advanced CRC treated with chemotherapy. For their analyses, the financial impact scale was omitted. Clinical anchors were used and those with a correlation of |0.3| or greater were given priority in their analyses. To estimate the MID for within-group changes, an effect size was calculated from the mean score divided by the SD of the change scores for all time points. For between-group changes, linear regression was used. An effect size between 0.2 and 0.8 was deemed acceptable because a value of less than 0.2 was considered not clinically important, while a value greater than 0.8 was more than minimally important.
The estimated MIDs ranged from around 7 to 18 points and 5 to 10 points for improvement and deterioration within-group changes, respectively. The MIDs for between-group changes were estimated to be from 5 to 14 points and 4 to 10 points for improvement and deterioration, respectively. Scales that are missing (pain, cognitive function, social function, dyspnea, and insomnia) either did not have an anchor or had an effect size outside of the 0.2 to 0.8 range.
EORTC QLQ-C30 anchor-based MIDs for within- (W: improvement, deterioration) and between-group (B: improvement, deterioration) changes in patients with CRC102
Physical function: (W: 7.31 to 8.52, -8.43 to – 6.09), (B: 6.05 to 10.04, -7.23 to -4.16)
Role function: (W: 10.43 to 18.06, -10.66), (B: 7.95 to 14.17, -9.96)
Social function: (W: 8.11 to 10.26, -6.18), (B: 6.73 to 7.79, - 6.03)
Fatigue (symptom score directions were reversed to align with function scores (0 represents the worst possible scores and 100 represents the best): (W: 7.65 to 13.82, -7.73 to -7.05), (B: 5.43 to 12.01, -6.98 to -6.76)
Nausea and vomiting ((symptom score directions were reversed to align with function scores (0 represents the worst possible scores and 100 represents the best): (W: 7.75, -7.95 to -5.30), (B: 7.34, -7.33 to – 5.17)
Appetite loss (symptom score directions were reversed to align with function scores (0 represents the worst possible scores and 100 represents the best): (W: 12.28, - 9.78), (B: 10.0, -7.11)
Diarrhea (symptom score directions were reversed to align with function scores (0 represents the worst possible scores and 100 represents the best): (W: 6.35, - 7.96), (B: 8.25, -5.46)
Constipation (symptom score directions were reversed to align with function scores (0 represents the worst possible scores and 100 represents the best): (W:12.75, not estimated), (B: 14.56, not estimated) (not estimated either due to no suitable anchor available or the effect size outside of the 0.2 to 0.8 range).
Global quality of life: (W: 7.14 to 10.34, -7.97 to -4.83), (B: 5.53 to 6.36, -9.12 to -6.81)
EORTC Quality of Life Questionnaire Lung Cancer 13
Description
The EORTC QLQ-LC13 is a self-reported, lung cancer-specific questionnaire with 13 items addressing symptoms associated with lung cancer and its standard treatment. This module supplements the QLQ-C30 questionnaire. While the QLQ-C30 questionnaire covers basic components of HRQoL relevant to a wide range of patients with cancer, the QLQ-LC13 measures specific symptoms and side effects experienced by patients with lung cancer receiving non-surgical treatment. When administered together with the QLQ-C30 questionnaire, the QLQ-LC13 assesses disease- and treatment-specific symptoms of lung cancer in clinical trials.110
The QLQ-LC13 supplementary module comprises both multi-item and single-item measures of lung cancer-associated symptoms (coughing, hemoptysis, dyspnea, and pain) and side effects from conventional chemotherapy and radiotherapy (hair loss, neuropathy, sore mouth and dysphagia). Only dyspnea and pain domains are comprised of multi-items and the rest are single items. All items employ a 1-week time frame. Except for the 1 item on pain medication, which asks for dichotomous response (no or yes), all items are scored on a 4-point Likert scale ranging from 1 (not at all) to 4 (very much). All scale and item scores are linearly transformed to a 0 to 100 scale, with higher scores representing increased symptom levels. Missing items are ignored if at least half of the items are filled per dimension.110
Validity
Bergman et al.103 tested construct validity with 883 patients (NSCLC: 62%) with non-resectable lung cancer who were receiving either chemotherapy or radiotherapy from 17 countries (Canadian, n = 115). Two known groups, namely disease stage (local, locoregional, metastatic) and ECOG PS, were selected. Patients with metastatic disease reported higher levels of pain and more frequent use of pain medication (P < 0.001) compared to patients with local disease. However, stage of disease was not significantly related to coughing or dyspnea. In contrast, ECOG PS divided into 2 levels (0-1 versus 2-4), was related significantly to pre-treatment dyspnea and coughing with elevated symptom score levels found primarily in patients with a poorer PS (P < 0.001). Statistically significant differences in pain scores were also observed as a function of ECOG PS (P < 0.001).
Nicklasson et al.97 conducted another validity study with 112 Swedish patients (NSCLC: 76%) diagnosed with lung cancer or pleural mesothelioma not amenable to curative or life prolonging treatment. The finding showed that correlation between spirometry results and QLQ-LC13 3-item dyspnea scale was weak, although statistically significant (n = 96; r = 0.24; P < 0.05). Furthermore, the convergent validity testing indicated that the BPI subscale correlated modestly (r > 0.4) with the QLQ-LC13 pain items.
Reliability
Based on the study results from Bergman et al.,103 the QLQ-LC13 dyspnea items formed a 3-item scale with internal consistency estimates of 0.81-0.83. When combined with the 1 QLQ-C30 questionnaire item on dyspnea, the reliability of the combined 4-item scale was improved further, with Cronbach alpha coefficients of 0.85-0.86. The finding for dyspnea scale has also been confirmed by Nicklasson et al.97 study (n = 112), which showed reliability estimate for dyspnea of alpha = 0.76.
In contrast, the QLQ-LC13 pain items did not form a reliable scale with alpha estimates of 0.53-0.54. When combined with the 2 QLQ-C30 questionnaire items on pain, the alpha coefficient improved considerably to 0.80. However, the reliability of the combined (QLQ-LC13 and QLQ-C30) 5-item pain scale was still lower than that of the 2-item QLQ-C30 pain scale (alpha = 0.83), even though both were in acceptable range (alpha > 0.70).111
Responsiveness to Change
In the Bergman et al.103 study, patients completed the QLQ-LC13 questionnaire once prior to the start of treatment (“pre-treatment”) and following the first course of radiotherapy or second course of chemotherapy (“on treatment”). The results showed that dyspnea, coughing, and pain scores changed significantly over time in the expected direction (i.e., declined or improved; P < 0.001 for all 3 domains except for extra thoracic pain item which showed P < 0.05). When compared between groups divided by ECOG PS (‘improved at least 1 level,’ ‘unchanged,’ ‘deteriorated at least 1 level’), ANOVA revealed significant interaction effects (group differences over time) for dyspnea (P < 0.001), chest pain (P < 0.01) and coughing (P < 0.01).
Minimal Important Difference
No information on an MID for the QLQ-LC13 was found in the literature.
Other Considerations and Limitations
The study conducted by Bergman et al.103 did not specify the time interval between “pre-treatment” or ‘”on treatment” when testing responsiveness to change. Also, results for responsiveness to change would have been more robust had Bergman et al. administered the questionnaire either multiple times throughout the study or after few cycles of therapies to capture accumulated effects of treatments. Furthermore, 1 item on perceived medication effectiveness in QLQ-LC13 questionnaire was excluded for further analyses because it caused confusion among patients, potentially due to its positive wording in contrast to the rest of questions. This exclusion reduces the reliability estimate of the multi-item pain scales of the QLQ-LC13. Lastly, an updated lung cancer module, QLQ-LC29, was published in 2017.106 The QLQ-LC29 better reflects the quality of life impact by major treatment advances, such as immune-oncology, compared to QLQ-LC13, which was published in 1994 and its psychometric properties have been assessed.112
European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire – Colorectal Cancer 29
Description
In 2017, the EORTC QLQ-CR29 was developed as a revised version of its 38-item predecessor, EORTC QLQ-CR38, also specific to CRC.113 The 29-item version was updated based on evidence from the literature (n = 20), interviews with patients who had a confirmed CRC diagnosis (n = 199), and consultations with health care professionals (n = 11).114 It was designed to reflect newer treatments and different side effects of such treatments.113,114 The EORTC QLQ-CR29 is a supplemental, CRC-specific module, which is intended to be assessed alongside the generic QLQ-C30.113,114
The EORTC QLQ-CR29 uses a 4-point Likert scale to assess the following items: urinary frequency, blood/mucus in stools, stool frequency, body image, urinary incontinence, dysuria, abdominal pain, buttock pain, bloating, dry mouth, hair loss, taste, anxiety, weight, flatulence, fecal incontinence, sore skin, embarrassment, stoma care problems, sexual interest for men and/or women, impotence, and dyspareunia.113 The questionnaire has a recall period of 1 week. Raw scores are linearly transformed to a score from 0 to 100 where higher functioning scores and lower symptom scores indicate better quality of life.
Validity
van der Hout et al. conducted a systematic literature review to assess the psychometric properties of the EORTC QLQ-CR29.104 The review included 11 studies, though not all studies evaluated every property. In summary, structural and construct validity were both deemed indeterminate for methodological reasons.
Whistance et al. conducted an international study of 351 patients with CRC in which patients completed both the EORTC QLQ-CR29 and QLQ-C30.105 Treatments included surgery, chemotherapy, radiotherapy, and palliative chemotherapy and patients were spread across the different tumour stages. The group calculated a correlation < 0.40 between the EORTC QLQ-CR29 and QLQ-C30 indicating the 2 questionnaires do not have overlapping topics.
Reliability
Evidence of subscale and single-item test-retest reliability was considered insufficient due to unacceptable intraclass correlation coefficients (ICCs) (< 0.7) as well as missing values. Based on the 6 studies that were evaluated, urinary frequency and urinary incontinence had the lowest ICCs ranging from 0.33 to 0.68 and 0.11 to 1.0, respectively. There were no subscales or items that had ICCs > 0.7 for all 6 studies, though stool frequency, body image, buttock pain, and hair loss had ICCs > 0.7 for 4 of 6 studies. Internal consistency was rated sufficient though the quality of evidence was low. The authors concluded that further investigation of the properties of the EORTC QLQ-CR29 is needed.104 On the other hand, Whistance et al. found that EORTC QLQ-CR29 questionnaire scales had ICCs > 0.68 while individual items had ICCs > 0.55.105
Wong et al.99 conducted systematic review of HRQoL instruments used to assess patients with CRC. The investigators concluded that their literature search found evidence of moderate strength supporting reliability though the evidence supporting validity and responsiveness was of uncertain strength for the EORTC QLQ-CR29.
Responsiveness to Change
van der Hout found responsiveness to be indeterminate because there were inconsistencies with sensitivity to detect score changes.104
Minimal Important Difference
No MID information was identified in patients with CRC.
Appendix 3: Detailed Outcome Data
Note that this appendix has not been copy-edited.
Table 38: Subgroup Analyses by Tumour Type and Prior Systemic Cancer Therapy (NTRK Efficacy-Evaluable Analysis Set)
Tumour type | Lines of therapy | Number of patients | Responders | ORR (95% CI) |
|---|---|---|---|---|
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| |
CI = confidence interval; CRC = colorectal cancer; CUP = carcinoma of unknown primary; GI = gastrointestinal; MASC = mammary analogue secretory carcinoma; NSCLC = non–small cell lung carcinoma; ORR = objective response rate.
Source: Sponsor-provided additional information32
Pharmacoeconomic Review
Abbreviations
AE
adverse event
BIA
budget impact analysis
CI
confidence interval
CNS
central nervous system
CRC
colorectal cancer
DAC
Drug Advisory Committee
GI
gastrointestinal
ICER
incremental cost-effectiveness ratio
IHC
immunohistochemistry
LCC
Lung Cancer Canada
MASC
mammary analogue secretory carcinoma
NGS
next-generation sequencing
NSCLC
non–small cell lung cancer
NTRK
neurotrophic tyrosine receptor kinase
OH-CCO
Ontario Health Cancer Care Ontario
OS
overall survival
PFS
progression-free survival
QALY
quality-adjusted life-year
SOC
standard of care
STS
soft tissue sarcoma
Executive Summary
The executive summary comprises 2 tables (Table 1 and Table 2) and a conclusion.
Item | Description |
|---|---|
Drug product | Entrectinib (Rozlytrek), 100 mg and 200 mg capsule |
Submitted price | Entrectinib, 200 mg capsule, $95.33 Entrectinib, 100 mg capsule, $47.67 |
Indication | For the treatment of adult patients who have unresectable, locally advanced or metastatic extracranial solid tumours (including brain metastases) with NTRK gene fusion without a known acquired resistance mutation and no satisfactory treatment options |
Health Canada approval status | NOC/c |
Health Canada review pathway | Standard |
NOC/c date | February 10, 2020 |
Reimbursement request | As per indication |
Sponsor | Roche |
Submission history | Previously reviewed: Yes Indication: For the treatment of NTRK fusion–positive, locally advanced or metastatic solid tumours in adult and pediatric patients Recommendation date: NA; withdrawn by sponsor |
NA = not applicable; NOC/c = Notice of Compliance with conditions; NTRK = neurotrophic tyrosine receptor kinase.
Table 2: Summary of Economic Evaluation
Component | Description |
|---|---|
Type of economic evaluation | Cost-utility analysis Partition survival model |
Target population | Adult patients who have unresectable, locally advanced or metastatic extracranial solid tumours (including brain metastases) with NTRK gene fusion without a known acquired resistance mutation and no satisfactory treatment options |
Treatment | Entrectinib (600 mg daily) |
Comparators | Comparators for each tumour site analyzed (representing best supportive care, including therapies used in first- and second-line):
|
Perspective | Canadian publicly funded health care payer |
Outcome | QALYs |
Time horizon | Lifetime (10 years) |
Key data source | Single-arm entrectinib trials: ALKA (phase I), STARTRK-1 (phase I), and STARTRK-2 (ongoing phase II basket trial) Naive comparison based on literature estimates for comparator PFS and OS; 1 trial selected per comparator. Comparator populations were not selected for NTRK fusion status. |
Submitted results |
|
Key limitations |
|
CADTH reanalysis results |
|
BSC = best supportive care; CRC = colorectal cancer; ICER = incremental cost-effectiveness ratio; IHC = immunohistochemistry; MASC = mammary analogue secretory carcinoma; MSI-H = microsatellite instability high; NGS = next-generation sequencing; NSCLC = non–small cell lung cancer; NTRK = neurotrophic tyrosine receptor kinase; OS = overall survival; PFS = progression-free survival; QALY = quality-adjusted life-year.
Conclusions
The CADTH clinical review noted that, due to the lack of any comparative evidence, the clinical effectiveness of entrectinib is highly uncertain. It was noted that response rates across different tumour types varied widely, with a similarly wide range of uncertainty. Combined with differences in sample sizes across the different tumour types (most subtypes had fewer than 10 patients), the generalizability of the findings to the mixed cancer population is limited.
CADTH undertook reanalyses to address several key limitations of the sponsor’s model. CADTH’s reanalyses included a more plausible extrapolation for long-term progression-free survival (PFS) and overall survival (OS), greater uncertainty in PFS and OS (consistent with the small sample sizes for each patient subtype), an assumption of no expected treatment benefit in patient subtypes with observational data representing 5 or fewer patients, and inclusion of the costs of using next-generation sequencing (NGS) to identify eligible patients. The CADTH reanalysis was also stratified by tumour types so that statistical and model-based analyses were appropriately performed on homogeneously defined populations and then subsequently combined into weighted averages to estimate aggregated cost-effectiveness. CADTH was unable to adjust for all the levels of uncertainty introduced by the small sample sizes for individual tumour sites, the lack of direct comparative trial data, and the fact that there is no clinical evidence provided for many potential clinical indications of solid tumours with neurotrophic tyrosine receptor kinase (NTRK) fusion mutations. CADTH reanalysis did not combine comparators within tumour types because the comparators varied so substantially in PFS and OS that it was assumed the patients considering these treatment alternatives may be different in clinically meaningful ways; however, how those clinical differences may affect the effectiveness of entrectinib is not known. It is possible that limitations beyond those identified could exist and could result in an underestimate of the true incremental cost-effectiveness ratio (ICER) for entrectinib.
Based on the available evidence and analysis performed by CADTH, the ICER of entrectinib compared to second-line treatment options, in the pooled analysis, exceeds $550,000 per quality-adjusted life-year (QALY) gained in patients with known NTRK fusion mutations and exceeds $10,000,000 per QALY gained when incorporating the costs of patient identification. In a scenario analysis that compared entrectinib to first-line comparators, the pooled analysis exceeds $750,000 per QALY gained in patients with known NTRK fusion mutations; incorporating the costs of identifying patients increases the ICER to more than $8,000,000 per QALY gained. In all analyses, there was substantial heterogeneity across tumour (sub)types.
With or without accounting for the cost of patient identification, for the pooled analysis, there is no price reduction that would achieve an overall ICER of $50,000 per QALY gained relative to therapies used in second line. However, there are numerous specific indications for which price reduction may achieve cost-effectiveness at a threshold of $50,000 per QALY gained in patients with known NTRK mutations. The tumour sites where cost-effectiveness is most achievable have high NTRK prevalence (which lowers the cost of identifying a single eligible patient) and higher QALY gains.
CADTH notes that for many tumour sites, there is substantial uncertainty regarding whether entrectinib provides net clinical benefit compared to the treatment alternatives identified by the sponsor. For numerous indications, there was a greater than 25% probability that entrectinib was clinically dominated (i.e., providing fewer life-years and fewer QALYs) by the alternative treatment. The CADTH analysis likely underestimated the magnitude of this uncertainty, given that the analysis was based on naive comparators with no adjustment for confounding, such as NTRK fusion status.
Finally, although a direct comparison to larotrectinib has not been conducted, CADTH notes that the sponsor presented no evidence to suggest entrectinib will lead to better health outcomes. A naive comparison, as the sponsor conducted against all other comparators, would suggest that entrectinib may be associated with worse outcomes than larotrectinib. This is shown in the unadjusted results from Garcia-Foncillas et al.1 There is a substantial amount of confounding when comparing data in this manner, limiting any conclusions that can be drawn. However, this highlights the uncertainty raised by CADTH concerning the sponsor’s approach to its submitted analysis.
Stakeholder Input Relevant to the Economic Review
This section is a summary of the feedback received from the patient groups, registered clinicians, and drug plans that participated in the CADTH review process.
Four patient groups provided input for the review of entrectinib for adults with solid tumours with NTRK gene fusions: the Lung Health Foundation (formerly the Ontario Lung Association), Lung Cancer Canada (LCC), Colorectal Cancer Canada, and the Canadian Breast Cancer Network. The Lung Health Foundation collected survey responses without geographical location from 14 patients with lung cancer, 1 of whom had experience with entrectinib, and 1 caregiver, as well as phone interview responses from 3 patients in Canada. LCC conducted an unspecified number of phone interviews. Among those interviewed, 8 patients (5 in the US, 2 in Canada, and 1 in the UK) had experience with entrectinib, including 1 patient with NTRK fusion who has used entrectinib for more than 5.5 years.
Colorectal Cancer Canada conducted an online survey to which a total of 6 patients with NTRK fusion mutation who had used an NTRK inhibitor responded: 2 patients with thyroid cancer (Canada, US), 2 with sarcoma (Canada, US), 1 with neuroendocrine tumours (US), and 1 with colorectal cancer (CRC) (Indonesia). Two of these patients (1 Canada, 1 US) had experience with entrectinib. The Canadian Breast Cancer Network collated results from 2012 and 2017 surveys to which 146 patients in Canada with metastatic breast cancer had responded, none of whom had experience with entrectinib. Cancer type varied among responding patients, but most were diagnosed at an advanced stage or with a rare form of cancer. Most patients had suboptimal success with conventional therapies (surgeries, radiation, radioactive iodine, chemotherapies) in terms of halting tumour growth or achieving lasting remission, and many reported significant side effects, such as fatigue, vomiting, weight loss, hair loss, anemia, neuropathy, and brain fog, as well as psychosocial effects, such as anxiety, stress, depression, and isolation. Patients noted that key outcomes included delayed progression, improved survival, symptoms, and quality of life, greater independence and functionality, and reduced side effects. Some expressed the need for treatments for brain metastases. For the most part, patients with experience using entrectinib reported improved quality of life and tumour reduction as well as reduced side effects, with several patients reporting 4 or more years of entrectinib usage, although some patients did report side effects, such as fluid on the lungs, bone deterioration, fatigue, neuropathy, weight gain, constipation, and vomiting. A number of patients wished they had been screened for biomarkers sooner, and all 4 submissions emphasized the importance of biomarker testing for more patients, preferably at diagnosis.
Five joint clinician group inputs were received for this review. These groups comprised 22 clinicians from LCC and 4 Ontario Health Cancer Care Ontario (OH-CCO) Drug Advisory Committees (DACs). There were 5 clinician members in the OH-CCO Lung Cancer DAC, 3 in the Gastrointestinal (GI) Cancer DAC, 2 in the Breast Cancer DAC, and 2 in the Head, Neck, and Thyroid Cancer DAC. The groups identified improving OS, improving response, delaying progression, improving quality of life, improving symptoms, delaying time to chemotherapy, improving toxicity, improving adherence, and ease of administration as unmet needs. Patients with NTRK gene fusions were identified as refractory to chemotherapy and/or immunotherapy and having high unmet needs in the absence of targeted therapy. The OH-CCO Lung Cancer DAC stated that entrectinib would displace first-line treatments, such as chemo- and immunotherapy combinations or pembrolizumab, to second-line, and second-line treatments to third-line. However, LCC said entrectinib would most likely be tried after chemotherapy and/or immunology as the first-line option or as first-line where other therapies are contraindicated. The OH-CCO GI Cancer DAC cancer stated that entrectinib would not affect the current sequencing of therapies, but would be an additional line of treatment, and stated that NTRK mutation testing should be added to the NGS panel for patients with GI cancer and that clinicians should be able to access provincial pathology lab reports with NTRK results (which are currently not available to them). According to the OH-CCO Breast Cancer DAC, entrectinib’s place in the current treatment paradigm remains uncertain, but it would be an additional line of therapy where many treatments for metastatic breast cancer are palliative. The OH-CCO Head, Neck, and Thyroid Cancer DAC stated that entrectinib would ideally be used as first-line therapy if NTRK mutation was identified before therapy, or as a later line if a patient progressed on or was intolerant to a different systemic therapy, thereby pushing back other currently available treatments. The group also expressed that molecular testing should be routinely performed on advanced and/or metastatic salivary and thyroid cancers.
Drug plan input noted that while the number of eligible patients for entrectinib identified through NTRK testing may be small, the number of patients eligible for NTRK testing is large and could represent a significant resource and/or budget impact. The input noted that a validated immunohistochemistry (IHC) assay is available in Canada; however, uncertainties exist around who should be tested, what the optimal testing strategy is, the optimal time to test, and how potential acquired resistance mutations might affect NTRK testing strategy.
Several of these concerns were addressed in the sponsor’s model:
The sponsor included PFS and OS as outcomes in the analysis.
The sponsor included both first- and second-line comparators.
Adverse effects of therapy were included.
In addition, CADTH addressed some of these concerns as follows:
CADTH included testing costs, accounting for the size of the eligible population that would require testing and using costs associated with NGS, considered the most appropriate testing strategy.
CADTH analyzed all tumour sites separately, accounting for different comparators and place in therapy.
Economic Review
Economic Evaluation
Summary of the Sponsor’s Economic Evaluation
Overview
The current review is for entrectinib (Rozlytrek) as first- or second-line therapy for adults with unresectable, locally advanced or metastatic cancer who have extracranial solid tumours with NTRK gene fusion. Entrectinib is taken orally, with an adult dosage of 600 mg once per day until disease progression. The cost per 28 days, based on the recommended dosing, is $8,007.72 for adult patients.
The clinical trial evidence presents data on 121 patients with NTRK fusion cancers, with primary solid tumours at 14 primary sites (including cancers of unknown primary). Model-based analyses were presented for a “pooled analysis” aggregating data across all tumour sites included in the trial data. For 6 adult primary tumour sites (with n > 5), a stratified analysis was also presented using the pooled analysis costs and efficacy for the entrectinib arm and tumour site-specific comparators. Comparators selected for the pooled and stratified analysis included both first- and second-line (best supportive care) options.
Despite the availability of naive comparison data for larotrectinib, which is another Health Canada–approved treatment for locally advanced or metastatic NTRK fusion cancer, the sponsor did not include larotrectinib in its set of comparators. Larotrectinib is also administered orally and has a 28-day cost of $11,724. The sponsor indicated that differences in outcomes between the trials may be due to differences in the patient population, specifically that the patients in the larotrectinib trial may have had better prognoses. However, this same risk of difference in the comparator population prognosis is present in all of the included naive comparator analyses for all the included lines of therapy.
The sponsor’s base-case analysis was performed from the public payer perspective. Costs and benefits were discounted at 1.5% per year. The sponsor’s analysis was performed using a 10-year time horizon, representing a lifetime analysis for patients with locally advanced or metastatic solid tumours aged 57 years at treatment initiation. The sponsor’s base-case analysis included incremental testing costs for tumour sites where NGS testing is not already provided through public funding for other mutations, using IHC screening.
Model Structure
The sponsor’s analysis used a 3-health-state partitioned survival model (schematic presented in Figure 1). This type of model does not require the explicit calculation of transition probabilities, but can otherwise be considered conceptually similar to a Markov model with time-varying transition probabilities. The probability of death within the model is determined solely by the survival curve for OS; therefore, it is not influenced by changes to the PFS curve. The PFS and OS curves for entrectinib are based on data from the entrectinib clinical trials. The PFS and OS curves for the comparator arms were derived using estimates from the literature. The fractions of patients in each health state at any point in time were informed directly by the survival curve analysis. Aligned with the dosing schedule for entrectinib, the model’s cycle length was 7 days. In the sponsor’s analysis, patients in the entrectinib arm received treatment based on the observed duration of treatment in the entrectinib clinical trials. In the comparator arm, patients received standard of care (SOC) until progression.
Comparators in the analysis represented a selection of first- and second-line options available to patients with locally advanced or metastatic cancer, developed with insight from clinical experts in Canada. Further, the sponsor indicated that if NTRK gene fusion is detected before the initiation of any systemic therapy for metastatic cancer, “[entrectinib] should be considered as first-line therapy, especially for instances where there is no satisfactory SOC for that tumour type.”
Model Inputs
Two analyses were conducted: a pooled analysis compared entrectinib to SOC averaging across all tumour sites and a tumour-specific analysis compared entrectinib to SOC in each individual tumour site.
For the pooled analysis, parametric survival curves were fit to data from the entrectinib trials, which aggregated PFS and OS across all tumour types. These parametric curves allowed the sponsor to extrapolate PFS and OS beyond what was seen in the trial. Because there was no comparator arm in the trial, the sponsor had to rely on naive comparisons to estimate PFS and OS for those who received SOC. In the pooled analysis, the sponsor calculated a weighted average of the median PFS and median OS across 17 cancer sites (including subtypes) based on the distribution of patients across tumour sites in the entrectinib clinical trial: breast cancer, secretory; breast cancer, non-secretory; CRC; mammary analogue secretory carcinoma (MASC); non–small cell lung cancer (NSCLC), squamous; NSCLC, non-squamous; neuroendocrine, pancreatic, and soft tissue sarcoma (STS); thyroid cancer, papillary; thyroid cancer, anaplastic; neuroblastoma; head and neck cancer; endometrial cancer; ovarian cancer; cholangiocarcinoma; and cancer of unknown primary. Estimates of median PFS and OS for each individual tumour site were derived from the literature except for neuroblastoma, head and neck cancer, endometrial cancer, ovarian cancer, cholangiocarcinoma, and cancer of unknown primary. For these tumour sites, the sponsor estimated PFS and OS using a simple average of the median PFS and OS of the other sites. The sponsor then fit an exponential distribution to the pooled PFS and OS median estimate to derive a PFS and OS survival curve for those who received SOC.
For the tumour-specific analyses, the sponsor used the same PFS and OS survival curves from the pooled analysis for those who received entrectinib. Therefore, regardless of tumour site, the sponsor assumed the same OS and PFS for those who received entrectinib. For SOC, the sponsor used tumour-specific estimates of PFS and OS, as described previously, and extrapolated long-term outcomes assuming an exponential parametric fit.
In the entrectinib arm, the sponsor assumed that treatment continued until the overall aggregated analysis median time on treatment, as described in the trial. In the comparator arms, treatment was continued only in the pre-progression state.
The rates of grade 3 and 4 adverse events (AEs) for patients in the entrectinib arms were estimated from the entrectinib trial. For comparator arms, AE rates were specific to each tumour type and treatment received, based on rates observed in the literature. For both the entrectinib and comparator arms, only AEs occurring in more than 5% of the population were included in the analysis. For the entrectinib arm, prevalence over 5% was determined only for the aggregate population, not stratified by tumour type.
The entrectinib and comparator arms used the same health utility values for the pre-progression and post-progression health states. Health utilities for the pre-progression health states (0.788) were estimated based on responses collected in the STARTRK-2 trial. Health utilities for the post-progression health states (0.642) were estimated based on the literature, with values identified for each tumour (sub)type included in the analysis (including site-specific values for pancreatic cancer and neuroendocrine tumours), then combined using the prevalence of each tumour (sub)type in the entrectinib trial at treatment initiation. Utilities were further reduced over time, accounting for average health loss associated with increasing age, based on the Brazier multiplier.2 Disutilities for AEs were estimated by type of AE using literature values aggregated into tumour (sub)type–specific values based on the frequency of occurrence, then aggregated into a weighted average using the frequency of each tumour (sub)type in the entrectinib clinical trial.
There is no single specific diagnostic test that can detect NTRK fusion cancers. Detection methods include NGS, fluorescence in situ hybridization, IHC, and reverse transcriptase polymerase chain reaction. In the sponsor’s analysis, patients with advanced stage or metastatic disease who are potentially eligible for entrectinib therapy and can supply a tumour sample are tested using NGS and/or IHC. NGS can test for the presence of multiple genomic alterations in a single tumour tissue sample. IHC screens for a single genetic abnormality at a time, and patients who test positive for NTRK gene fusions with IHC require a confirmatory NGS test before initiating treatment. Table 8 summarizes the sponsor’s assumptions regarding the status quo and incremental testing protocol for each tumour site.
The sponsor’s analysis estimated the weekly cost of cancer care based on the annual net cost of cancer-related health care expenses in patients with cancer minus the total cost of inpatient chemotherapy and outpatient drugs. The sponsor then divided this result by the total number of patients with cancer who were diagnosed over 4 years.3 Entrectinib drug costs and dosing information were provided by the sponsor and product monograph. Dosing and unit costs of the therapies used in the comparator arm were sourced from the literature and product monograph. Administration costs were also included for infusion-based treatments. The aggregate weekly cost of treatment was calculated as a simple average of individual comparators (first- and second-line) for each tumour (sub)type, followed by a weighted average of tumour (sub)types using the frequency of each tumour (sub)type in the entrectinib clinical trial. No adjustment to this weekly cost was made because the distribution of cancer types in the pre-progression and post-progression health states changed over time in the model. In the base-case analysis, the sponsor did not assume wastage for infusion-based treatments or for entrectinib. In the analyses in which entrectinib was the first-line therapy, the sponsor’s analysis did not include any costs associated with active second-line therapies. The sponsor estimated the cost of AEs using the Ontario Analysis Tool.4 Finally, the sponsor’s analysis assumes that IHC costs $80 per test and that NGS costs $1,400 per test.
The sponsor’s model adjusted costs to 2021 Canadian dollars using the medical care component of the Consumer Price Index.
Summary of Sponsor’s Economic Evaluation Results
The sponsor presented probabilistic analyses for the pooled and tumour-specific analyses using 5,000 iterations.
Base-Case Results
The sponsor’s pooled analysis indicates that entrectinib increases survival, quality-adjusted survival, and costs (primarily due to drug costs). For patients with known NTRK fusion cancer, the sponsor’s pooled analysis indicates incremental costs of $102,608 per patient, an increase in life expectancy of 1.16 years, and an increase in quality-adjusted life expectancy of 0.88 QALYs. The pooled ICER of entrectinib compared to the weighted average combination of first- and second-line therapies across all tumour types in the sponsor’s clinical trial efficacy analysis is $116,868 per QALY gained. Incorporating the additional case detection cost of $30,730 per patient identified with NTRK fusion cancer, the sponsor calculated an ICER of $151,852 per QALY gained. The sponsor’s analysis indicates that the probability that entrectinib is cost-effective at a willingness-to-pay threshold of $50,000 per QALY gained is less than 1%. The sponsor did not report the proportion of the health benefit accrued in the observed versus the extrapolated period.
The sponsor’s tumour-stratified analysis assumed the same PFS and OS for each tumour type as the pooled analysis for those receiving entrectinib. Only the costs of case detection and model parameters for the comparator arm were varied by tumour site, despite substantial variations in tumour-specific responses and durations of response for these tumour types. As such, these results are not reported.
Table 3: Summary of the Sponsor’s Economic Evaluation Results
Drug | Total costs ($) | Incremental costs ($) | Total QALYs | Incremental QALYs | ICER vs. reference ($/QALY) |
|---|---|---|---|---|---|
Pooled comparator | 95,524 | Reference | 1.612 | Reference | Reference |
Entrectinib (pooled analysis) | 228,862 | 133,338 | 2.49 | 0.878 | 151,852 |
ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year vs. = versus.
Source: Sponsor’s pharmacoeconomic submission.5
Sensitivity and Scenario Analysis Results
The sponsor’s sensitivity analysis indicated that its pooled analysis results are sensitive to the parameters describing the PFS and OS curves of patients on entrectinib, although the analysis did not use empirically justified uncertainty. Varying the progression rates by plus or minus 10% of the base case led to a range of ICERs, from $138,889 to $168,209 per QALY gained.
The tumour-stratified analysis functions as a sensitivity analysis on the properties of the comparator. Compared to a mix of first- and second-line treatments for NSCLC, including several combination therapies with pembrolizumab with a weekly cost of $5,736, the sponsor’s analysis finds entrectinib to be cost-saving. In contrast, compared to a mix of first- and second-line treatments for breast cancer (including carboplatin, paclitaxel, docetaxel, and eribulin) with a weekly cost of $794, the sponsor’s analysis finds entrectinib to have an ICER of $307,190 per QALY gained.
CADTH Appraisal of the Sponsor’s Economic Evaluation
CADTH identified several key limitations to the sponsor’s analysis that have notable implications on the economic analysis:
Appropriateness of pooling across tumour sites: Pooled analysis is not generally appropriate for modelling clinical effectiveness or cost-effectiveness and is discouraged in CADTH’s guidelines for economic evaluation and more specific guidelines for tumour-agnostic products.6,7
First, entrectinib’s PFS and OS curves are affected by the tumour-type distribution in the clinical trial, which is not representative of the patient population eligible for entrectinib treatment. The sponsor’s pharmacoeconomic report acknowledges this when advising against comparison to larotrectinib, another tumour-agnostic product, claiming that “tumour-agnostic” effectiveness is contingent on the distribution of cancer subtypes, the number of prior lines of therapy (19% of patients in the entrectinib trial have received ≥ 3 prior lines of therapy versus 31% of patients in the larotrectinib trials), and frequency of central nervous system (CNS) brain metastasis across the trial populations (experienced by 21.5% of patients in the entrectinib trial versus 14% of patients in the larotrectinib trials). However, in the sponsor’s analysis, the results are aggregated across tumour types with very different baseline prognoses; the analysis combines patients with and without CNS brain metastasis and with different numbers of prior lines of therapy. When considering the efficacy of comparator treatments to entrectinib, no consideration is made to matching the comparator trial population on these features or to perform any analysis to try to control for these confounders. In acknowledging the limitations associated with comparing larotrectinib to entrectinib, the sponsor must also acknowledge the limitations associated with a pooled analysis.
Second, it is inappropriate to perform statistical survival analysis on highly heterogeneous populations while relying on a core assumption that the sample is taken from a common population. In the pooled analysis presented by the sponsor, individuals with poorer-prognosis tumour types leave the at-risk population earlier due to faster rates of progression or death; the effect is that the remaining population disproportionately represents patients with better initial prognosis. A pooled analysis does not represent the heterogeneity in response, PFS, or OS observed in the stratified efficacy analysis of the entrectinib trial results. The pooled analysis also affects the accuracy of modelling the comparator arm. Over time, the definition of each health state would vary based on its composition, affecting the costs and QALYs accrued over a patient’s lifetime. For example, if patients receiving relatively lower-cost comparator treatments have a poorer average prognosis, the average cost of treatment in the pre-progression state for the comparator arm would increase over time. The limitations of modelling heterogenous populations are well discussed in the literature.8
Further, averaging across comparators that vary in their own costs and effectiveness can mask the patient populations, settings, or conditions under which the new treatment may or may not be cost-effective. The decision-relevant information is the population and indication-specific ICER compared to the appropriate comparators for that population and indication. Averaging in a situation in which the average identifies that a novel therapy is “not cost-effective” may harm (through restricted access) the individuals for whom the therapy is cost-effective.
Where information was available to do so, CADTH reported its analyses stratified by tumour type. CADTH also presents a pooled analysis which aggregates the results from the analyses for each separate tumour type.
The stratified analysis provided in the sponsor’s report for 6 cancer types does not use tumour type–specific entrectinib effectiveness data: The sponsor’s selected tumour types for tumour-specific analyses represent cancer types for which the sample size exceeded 5 patients in the entrectinib trials. The sponsor’s stratified analysis assumes that all the indications for which it provided stratified analysis — that is, NSCLC (combining squamous and non-squamous), CRC, breast cancer (combining secretory and non-secretory), MASC, STS, and thyroid cancer (combining papillary and anaplastic) — have the same PFS and OS, despite different response rates and durations of response by tumour type. The entrectinib clinical report indicates an overall aggregated cohort response rate (complete or partial) of 61.2%, with wide variation across tumour types: 83.3% for MASC (95% confidence interval [CI], 62.6 to 95.3%; n = 24) compared to 20% for CRC (95% CI, 2.5 to 55.6%; n = 10). Similarly, the aggregated median duration of response is reported to be 20 months, with substantial variation across tumour types: not yet estimable in numerous tumour types, but already observed to be a median of only 12.9 months in patients with breast cancer and 13.2 months in patients with thyroid cancer.
CADTH requested median PFS, median OS, and Kaplan–Meier survival curves for the PFS and OS for each tumour (sub)type. The sponsor refused to meet this request and provided only Kaplan–Meier curves based on the August 2020 data cuts for NSCLC (n = 22), MASC (n = 24), and sarcoma (n = 26). For the remaining tumour sites, CADTH used estimates of tumour-specific outcomes data, where available. Table 10 provides information about what data were used to derive the CADTH estimates.
A health economic evaluation was not provided for 8 clinical indications for which there were patients included in the clinical trial and for which reimbursement is sought: Entrectinib clinical trial data include data for neuroendocrine cancers (n = 5), pancreatic cancer (n = 4), cancer of unknown primary (n = 3), cholangiocarcinoma and/or non-CRC GI cancer (not otherwise specified) (n = 2), ovarian cancer (n = 1), endometrial cancer (n = 1), neuroblastoma (n = 1), and head and neck cancer (not otherwise specified) (n = 1). For 2 of these cancer subtypes, specific tumour site-relevant comparators were included in the weighted average estimates for PFS, OS, cost, and utilities used for the comparator arm. Without clinical justification, the sponsor assumed PFS, OS, costs, and benefits based on a simple average of other cancer types for which it did collect data. In addition, the broader clinical literature indicates a large number of cancer sites affected with NTRK fusion, as shown in Figure 2.9 Substantial uncertainty exists about the clinical and cost-effectiveness of entrectinib in tumour types with small sample sizes and for which health economic analysis was not performed.
For tumour types with 5 or fewer patients with entrectinib clinical trial data, and for which the sponsor did not provide tumour-specific analyses, CADTH assumes no incremental QALY loss or gain, but full incremental cost of treatment, due to the absence of any clinical evidence in the pooled analysis to suggest otherwise. In the CADTH reanalysis, the incremental cost of treatment relied on the sponsor’s own analysis using median duration of treatment with no adjustments.
CADTH notes that many tumour sites that may be eligible for treatment with entrectinib were not included in the trial. These include appendix, bone, CNS, hepatic, melanoma, and prostate cancers.
Aggregation of disparate tumour types with very different prevalence of NTRK fusion cancer: In the tumour-stratified analysis, the sponsor combined cancer subtypes NSCLC (squamous and non-squamous), breast cancer (secretory and non-secretory), and thyroid cancer (papillary and anaplastic) with different rates of NTRK fusion positivity. While the difference is generally small for NSCLC (NTRK prevalence for squamous = 0.17%; adenocarcinoma = 0.26%; and large cell = 0.47%) and for thyroid cancer (NTRK prevalence for papillary = 1.83% and for anaplastic = 0.99%),9 the difference is much larger when comparing non-secretory and secretory breast cancers. NTRK fusion is characteristic of secretory breast cancer, occurring in approximately 95% of cases, in contrast to its low prevalence in non-secretory breast cancer (0.13% in the sponsor’s report). As a result, the testing strategy and testing costs are very different per identified case.
The CADTH reanalysis applied the appropriate testing costs separately to secretory and non-secretory breast cancers, based on tumour-specific prevalence of NTRK fusion mutations. Other aggregations of tumour subtype were left consistent with the sponsor’s analysis.
Aggregation of first- and second-line treatment options: In both the pooled and tumour-stratified analysis performed by the sponsor, the sponsor used a simple average across first- and second-line comparators, with limited clinical information or justification presented. The sponsor states that patients were “matched” based on tumour type and prior lines of therapy but does not provide more detailed information. The number of patients making first- versus second-line treatment decisions in the clinical trial population was not available on a tumour-specific basis. The only aggregate information provided is that 19% of patients in the entrectinib trial have received greater than or equal to 3 prior lines of therapy. Across tumour types, there is substantial variation in the representation of first-line therapies, with 100% of treatment alternatives for thyroid cancer, neuroendocrine cancer, and MASC versus 50% of treatment alternatives in breast cancer and squamous NSCLC and 33% of treatment alternatives for non-squamous NSCLC. The sponsor did provide a sensitivity analysis in which only the first-line comparators were selected for each tumour type.
Subsequent therapy costs were also not considered by the sponsor. The feedback from clinical experts consulted for this review indicated that if entrectinib was used as a first-line therapy, patients would likely receive current SOC after progressing on entrectinib. The exclusion of these relevant costs biases the sponsor’s comparison of entrectinib against first-line therapies in favour of entrectinib.
CADTH stratified the pooled analysis by separating first- and second-line comparators.
Second-line comparisons were maintained as a base-case analysis. First-line comparisons were made as a scenario analysis, with additional consideration for potential additional costs associated with subsequent therapy use.
Representation of uncertainty in PFS and OS: The parametric survival models selected by the sponsor underestimated the uncertainty in the data’s observed period by relying on the assumption that the sample is taken from a common population. In fact, because the population is a mixture of heterogeneous subgroups, the overall PFS and OS survival curves are a mixture of the component distributions. The variance in the mixture of distributions would be larger than either the variance of a weighted average of distributions (represented by the [weighted] sum of random variables) or the variance calculated, assuming all the survival data come from a common population.
The sponsor extrapolated to the end of the 10-year analysis horizon using the parametric survival curves selected to fit the data in the observed period. The sponsor did not incorporate any treatment-waning effect or increased uncertainty in the extrapolation period. Likewise, the sponsor fit curves only to the pooled PFS and OS data across all tumour sites and did not fit data to individual tumour sites.
In the CADTH reanalysis, uncertainty for each of the tumour types was estimated using the tumour type–specific data, accounting for the small sample size, as represented in the 95% Cis around the median PFS or OS, when provided, and using a binomial model around the point in the survival curve at which less than 50% of the original population remains at risk. A table of inputs used to derive these estimates is provided (Table10).
CADTH treated the observed period differently from the unobserved (extrapolated) period post-trial data cut-off for each tumour site. Using a threshold of less than 50% of the original population at risk to define the beginning of the unobserved period, CADTH assumed that PFS and OS followed the rates predicted by the comparator arm. This threshold was selected because of the lack of number-at-risk data available for several of the tumour subtypes. In several cases, median PFS and median OS were the only data provided.
Diagnostic testing strategy for the identification of patients with NTRK fusion cancers: According to the CADTH clinical experts consulted for this review, IHC for the detection of NTRK fusions is in development, and the technology is not yet available in all provinces. Limitations of IHC for NTRK screening include the inability to discriminate between the expression of wild-type and fusion protein; subjective interpretation due to heterogeneity in normal tissue expression, requiring strict controls; and the fact that standardized antibodies are not yet widely available.10 The demonstrated sensitivity of IHC varies significantly across studies.11-13 Further, a study of 4,108 NTRK gene fusion-negative tumours identified a specificity of 96% that varied substantially across tumour types,14,15 presumably due to variation in the rates of wild-type protein expression. A small recent study evaluating IHC in 66 fusion-positive cases and 317 fusion-negative cases identified an overall sensitivity of 87.9% (79% in NTRK3 fusions) and specificity ranging from 20.8% in gliomas to 52% in salivary gland tumours to 74% in sarcomas and 100% in colon, lung, thyroid, and appendix cancers and cholangiocarcinoma.16 Until IHC testing has been reliably validated for clinical use, it is not appropriate to consider IHC screening a “base-case” technology. Given currently available technologies, NTRK gene fusion testing would occur through NGS.
Further, clinical experts consulted for this review indicated a preference for using NGS because of its potential ability to screen for multiple mutations using a single sample from the patient. Concerns were raised about IHC “using up” patients’ available samples, thereby preventing the exploration of other treatment options, given the low probability, for most patients, of an NTRK fusion mutation being identified.
CADTH used NGS as the base-case testing strategy for identifying patients with NTRK gene fusion cancers.
Completeness and accuracy of non-treatment health care costs: The sponsor’s analysis does not include age-specific non-cancer health care costs. Because entrectinib is predicted to increase life expectancy, this omission biases the health economic analysis toward the intervention by reducing the incremental cost.
The sponsor’s analysis estimated the weekly cost of cancer care based on the annual net cost of cancer-related health care expenses in patients with cancer in 2012 less the total cost of inpatient chemotherapy and outpatient drugs, which the sponsor then divided by the total number of patients with cancer diagnosed over 4 years.3 Further, the sponsor’s calculation of cancer-related costs used the total annual costs of patients with any cancer diagnosis in Ontario in 2012 without justification for the representativeness of the specific cancer types present in the entrectinib trials.
CADTH included non-cancer health care costs consistent with the average age of patients with each cancer type using Canadian national average health care expenditures. CADTH also estimated the cancer-related costs using the continuing care costs of patients for each tumour type from an Ontario-based study.17 These costs are outlined in Table 9.
Inappropriate weights used to derive the distribution of eligible NTRK fusion cancers across tumour sites: The sponsor’s analysis used weights informed by the distribution of tumour types in the entrectinib trial in some places. These weights differ substantially from the relative incidence of the population that would be expected to receive treatment if entrectinib were funded, based on the incidence of each cancer type and the prevalence of NTRK fusion cancers within that tumour type.
CADTH assumed more appropriate Canadian weights, where possible. A comparison of the weights used in the sponsor’s analysis and the CADTH reanalysis is presented in Table 10.
Appropriateness or representativeness of the comparator survival curves: The single arm of the clinical trials informing the median PFS and OS for each of the comparator arms may not be representative data for those treatment alternatives. Further, no trial selected to represent a comparator arm had any information about NTRK status. It is possible that the subset of patients affected by NTRK fusion have different PFS or OS rates compared with the other patients in these trials. Likewise, no attempt was made to observe or control for any confounding factors that would influence PFS and OS between trials.
CADTH was unable to address this limitation and highlights the significant uncertainty this adds to the analysis.
The sponsor’s model makes several computational errors: In cases where simple calculations are required, a significant amount of complexity impeded the CADTH review. The sponsor’s model used 25,110 MATCH functions, 5,278 ISERROR and IFERROR functions, and 9,427 CHOOSE functions. Many of these functions limit CADTH’s ability to robustly validate the model because these override cells and can hide when an error is occurring. Further, some inputs are difficult to change for a sensitivity analysis because the sponsor hardcoded calculations into input cells (e.g., cancer-related health care costs). Important information within the analysis is repeated several times across multiple sheets, making it unclear which data are being used to generate results. In several cases, inputs are transformed from daily to weekly to monthly (or yearly) values and then back to weekly, across numerous sheets, but using different ratios in different steps. This leads to inflation of values over their 7-day cost (for example, the 7-day cost of entrectinib was inappropriately inflated from $2002 to $2009). The sponsor’s model also discounted costs and benefits differently in the entrectinib arm than in the comparator arm. The total dose calculations for pembrolizumab and bevacizumab referenced height in centimetres instead of body weight in kilograms. This resulted in overestimating the weekly cost of pembrolizumab to $4,923 (rather than $2,099) and the weekly cost of bevacizumab to $3,268 (rather than $1,370).
The CADTH reanalysis was able to correct several of these small computational errors. It also corrected the weekly costs of pembrolizumab and bevacizumab and aligned the approach to discounting across the 2 arms of the model analysis. However, the lack of transparency and inflexibility of the modelling approach impeded CADTH’s ability to validate the model robustly.
No comparison to larotrectinib: In September 2021, larotrectinib was recommended by the CADTH pan-Canadian Oncology Drug Review Expert Review Committee for use in adult and pediatric patients with locally advanced or metastatic solid tumours harbouring an NTRK gene fusion. For adult patients, entrectinib covers the same Health Canada indication. The sponsor notes that larotrectinib could not be compared against entrectinib due to differences in the trial populations, such as tumour type distribution. However, this rationale would preclude a naive comparison against any treatment because the trial was single-armed. CADTH identified a matched adjusted indirect comparison of larotrectinib to entrectinib; however, due to the unanchored nature of the approach, firm conclusions could not be adopted. Given that the analysis was conducted on an aggregate level, CADTH was unable to use the results in the analysis.
CADTH was unable to assess the cost-effectiveness of entrectinib versus larotrectinib. No evidence was presented that would indicate entrectinib should be priced higher than larotrectinib.
The inclusion criteria for AEs resulted in an underestimation of the impact of AEs in the tumour-specific analysis: The sponsor included AEs in the entrectinib arm only if these occurred in more than 5% of the population in the pooled analysis. AE rates were not presented by their frequency of occurrence within each tumour (sub)type.
CADTH was unable to fix this error.
CADTH Reanalyses of the Economic Evaluation
The CADTH reanalysis addresses many of the limitations of the submitted model and report, as outlined previously. The CADTH reanalysis was derived by making changes in model parameter values and assumptions, in consultation with clinical experts.
Table 4: CADTH Revisions to the Submitted Economic Evaluation
Stepped analysis | Sponsor’s value or assumption | CADTH value or assumption |
|---|---|---|
Corrections to sponsor’s base case | ||
Corrected weekly drug costs | The total dose calculations for pembrolizumab and bevacizumab referenced height in cm instead of body weight in kg. This resulted in overestimating the weekly cost of pembrolizumab to $4,923 and the weekly cost of bevacizumab to $3,268. Through a series of multiplication of daily costs to monthly and annual costs and then division back to weekly costs using different multipliers, the sponsor’s analysis inflated the 7-day cost of treatments (entrectinib and all comparators). | The CADTH reanalysis calculated the weekly cost of pembrolizumab and bevacizumab using body weight in kg. This resulted in a weekly cost of $2,099 for pembrolizumab and a weekly cost of $1,370 for bevacizumab. CADTH used the daily cost multiplied by 7 to calculate the 7-day cost of all drug treatments for the 7-day cycle length of the model. |
Corrected differential approach to discounting | The sponsor’s analysis discounted the entrectinib arm in every cycle, but only discounted the comparator arm annually. | The CADTH reanalysis used the same discounting approach (the one the sponsor used in the entrectinib arm) for both arms of the analysis. |
Changes to derive the CADTH base case | ||
1. Used tumour-specific PFS and OS data from entrectinib trial | The sponsor’s stratified analysis assumed that all the indications for which it provided stratified analysis — that is, NSCLC (combining squamous and non-squamous), colorectal cancer, breast cancer (combining secretory and non-secretory), MASC, soft tissue sarcomas, and thyroid cancer (combining papillary and anaplastic) — have the same PFS and OS, despite the different response rates and durations of response by tumour type presented in its clinical report. | CADTH requested median PFS, median OS, and KM survival curves for the PFS and OS for each tumour (sub)type. In response to this request, the sponsor provided KM curves based on the August 2020 data cut for NSCLC (n = 22), MASC (n = 24), and sarcoma (n = 26). From the October 2018 data cut-off, median PFS and median OS were also available for thyroid cancer (n = 7), colorectal cancer (n = 7), and breast cancer (n = 6). The CADTH reanalysis used tumour-specific outcomes data where available. |
2. Applied appropriate testing costs | The sponsor’s analysis used IHC to screen for NTRK fusion mutations. However, clinical experts engaged for this review indicated that IHC is still in development, is not clinically validated, and is not widely available. These experts indicated a strong preference for NGS because it would not “use up” a patient’s sample for a low-probability test. (NGS can screen for several mutations simultaneously.) Further, the sponsor’s analysis combined secretory and non-secretory breast cancer. NTRK fusion is characteristic of secretory breast cancer, occurring in approximately 95% of cases, but it has a low prevalence in non-secretory breast cancer (occurring in 0.13% of cases, according to the sponsor’s report). As a result, the testing strategy and testing costs are very different per identified case. | CADTH used NGS as the base-case testing strategy for identifying patients with NTRK gene fusion cancers. The CADTH reanalysis stratified the analysis by secretory and non-secretory cancer to the extent possible. The costs and benefits, given known mutation, were assumed to be the same because tumour subtype–specific prognosis and effect information were not available. Analyses, including the cost of identifying NTRK fusion cancers, were separated to account for the relatively low incremental cost of identifying patients with secretory breast cancer who have NTRK fusion and the relatively high cost of identifying patients with non-secretory breast cancer who have NTRK fusion. |
3. Used more appropriate prevalence estimates | The sponsor’s analysis used weights informed by the distribution of tumour types in the entrectinib trial in some places. These weights differ substantially from the relative incidence in the population that could be expected to receive treatment if entrectinib were funded, based on the incidence of each cancer type and the prevalence of NTRK fusion cancers within that tumour type. | CADTH assumed more appropriate Canadian weights, where possible. The number of incident patients with each tumour type was estimated using the sponsor’s BIA for indications included in the BIA. For indications excluded from the BIA, CADTH estimated the number of incident cases from publicly available CADTH reviews of treatments targeting populations with NTRK fusion, estimates calculated from the total incidence of the cancer type in Canada (excluding Quebec), the proportion of patients who are in stage III or IV, and the prevalence of NTRK mutation. A comparison of the weights used in the sponsor’s analysis and the CADTH reanalysis is presented in Table 12. |
4. Updated health care costs | The sponsor’s analysis does not include age-specific, non-cancer health care costs. The sponsor’s analysis estimated the weekly cost of cancer care based on the annual net cost of cancer-related health care expenses in patients with cancer in 2012 less the total cost of inpatient chemotherapy and outpatient drugs, which it then divided by the total number of patients diagnosed with cancer over 4 years. | CADTH included non-cancer health care costs consistent with the average age of patients with each cancer type using Canadian national average health care expenditures. CADTH estimated the cancer-related costs using the continuing care costs of patients for each tumour type using an Ontario-based study. Further details regarding sources and costs used are provided in Appendix 4. |
5. Updated uncertainty estimates around PFS and OS | The parametric survival models selected by the sponsor underestimated the uncertainty in the data’s observed period by relying on the assumption that the sample is taken from a common population. | Uncertainty for each of the tumour types was estimated using tumour type–specific data, accounting for the small sample size as represented in the 95% confidence intervals around the median PFS or OS, when provided, and using a binomial model around the point in the survival curve at which less than 50% of the original population remains at risk. Further details on the values used by CADTH are provided in Table 10. |
6. Separated out the analysis by first- and second-line comparators | The sponsor’s analysis combined comparisons across tumour types; but within tumour types, the analysis also combined comparators used as first- and second-line therapies with very different prognoses, representing a high level of within-group heterogeneity. | CADTH analyzed the cost-effectiveness of entrectinib against first- and second-line comparators separately. |
7. Added subsequent therapy costs | The sponsor’s analysis comparing entrectinib to first-line therapies did not account for the likely possibility that the current first-line therapy would become an active second-line therapy. | CADTH performed reanalysis using the sponsor’s framework, assuming that the current first-line therapy would be displaced and that no additional lines of therapy would be added to the treatment sequence of patients. In addition, CADTH included an analysis in which the additional cost of the comparator is added to the entrectinib arm, representing the additional costs associated with the current first-line therapy becoming a second-line of therapy. |
CADTH base case (vs. subsequent lines) | 1 + 2 + 3 + 4 + 5 | |
CADTH scenario analysis vs. 1L (excluding subsequent therapy costs) | 1 + 2 + 3 + 4 + 5 + 6 | |
CADTH scenario analysis vs. 1L (including subsequent therapy costs) | 1 + 2 + 3 + 4 + 5 + 6 + 7 | |
1L = first line; BIA = budget impact analysis; IHC = immunohistochemistry; KM = Kaplan–Meier; MASC = mammary analogue secretory carcinoma; NGS = next-generation sequencing; NSCLC = non–small cell lung cancer; NTRK = neurotrophic tyrosine receptor kinase; OS = overall survival; PFS = progression-free survival.
Base-Case Results
The CADTH reanalysis evaluated the cost-effectiveness of entrectinib compared to first- and second-line comparators for patients with each NTRK fusion cancer stratified by primary solid tumour site. Specifically, detailed analyses were performed for breast cancer (secretory and non-secretory), CRC, MASC, NSCLC, STS, and thyroid cancer; the sponsor had provided some tumour-specific outcomes data for each of these. The CADTH reanalysis was also performed for neuroendocrine and pancreatic cancers. For these sites, tumour-specific outcomes data were not provided; therefore, the CADTH reanalysis assumed entrectinib to have the same PFS and OS (i.e., no incremental QALY gain) as the treatment alternatives suggested by the sponsor. CADTH could not perform reanalysis for cancer of unknown primary, cholangiocarcinoma, ovarian cancer, endometrial cancer, neuroblastoma, or head and neck cancer because tumour-specific outcomes data and specific treatment comparators were not provided. For these indications, CADTH used the entrectinib treatment cost to represent the incremental cost of entrectinib and assumed no net QALY gain. CADTH performed stratified reanalysis on all tumour types for which the sponsor provided a stratified analysis.
Detailed results for each tumour subtype, including detailed cost and QALY breakdown, are presented in Appendix 4.
Tumour-Specific Analysis Summary
Compared to second-line treatment alternatives, entrectinib was dominated in several cases, providing less clinical benefit at a higher cost in patients with CRC (versus FOLIFOX and FOLFIRI) and patients with thyroid cancer (versus lenvatinib and sorafenib). Among cases in which entrectinib treatment increased costs and provided positive clinical benefit, in patients known to have an NTRK fusion mutation, ICERs ranged from $149,881 per QALY gained (NSCLC, versus pemetrexed and cisplatin) to $288,673 per QALY gained (breast cancer, versus eribulin). When NTRK fusion status needs to be established before entrectinib treatment, ICERs remained stable for patients with secretory breast cancer, in whom NTRK fusion mutation is characteristic, at $290,655 per QALY gained (versus carboplatin) and $294,420 per QALY gained (versus eribulin). In tumour sites where NTRK fusion mutation is rare, the ICER for testing and treatment with entrectinib exceeded $1,000,000 per QALY gained.
Pooled Analysis
For the pooled analysis, overall incremental costs and overall incremental QALYs were calculated using a weighted average across tumour sites, with the weights determined by the relative incidence of patients with NTRK fusion cancers each year (Table 10).
Two analyses were performed. One included only indications for which the sponsor provided at least comparator PFS and OS curves. The other included all indications in the clinical trial and Health Canada approval for which no appropriate comparators had been identified and no clinical data were provided relating to either the PFS or OS for entrectinib or comparators. For indications for which patients were present in the clinical trial, but for whom no clinical data were provided, the expected health gain compared to the alternative is equal to 0, but the average cost of entrectinib treatment, as calculated by the sponsor, is still incurred.
The pooled analysis indicates that for all indications included in the sponsor’s modelled analysis, for patients with a known NTRK fusion mutation, the ICER is $586,995 per QALY gained compared to second-line alternatives. Including all clinical indications present in the clinical trial population, for patients with a known NTRK fusion mutation, the ICER increases to $2,146,211 per QALY gained compared to second-line alternatives. Incorporating the costs of testing increases the ICER to more than $8,000,000 per QALY gained.
Scenario Analysis
CADTH conducted a scenario analysis that compared entrectinib to comparators used in the first-line setting. In the sponsor’s model, it was not structurally possible to consider the costs and benefits of a second-line active therapy option after progression with first-line therapy. However, patients considering these first-line alternatives may still be able to use these active lines of therapy after progressing on entrectinib. CADTH conducted an analysis without subsequent therapy costs, as per the sponsor’s base case, and ananalysis that considered the possibility that the costs of the comparator would still be incurred in the entrectinib arm when the comparator was subsequently provided.
The identified incremental QALYs from entrectinib treatment compared to first-line treatment alternatives ranged from −0.44 QALYs (CRC: entrectinib versus pembrolizumab) to 1.1 QALYs (MASC: entrectinib versus sunitinib) (Table 12). The CADTH reanalysis identified high uncertainty concerning whether entrectinib was clinically superior to first-line alternatives in many tumour types. The proportion of simulations in which the first-line alternative resulted in greater health gains than entrectinib exceeded 25% for most tumour types, including breast cancer (29% versus paclitaxel and 55% versus docetaxel), CRC (99.5% versus pembrolizumab, 85.2% versus FOLFOX, and 83.1% versus FOLFIRI), NSCLC (25.3% versus pembrolizumab + pemetrexed and cisplatin), and thyroid cancer (93.5% versus lenvatinib and 53% versus sorafenib). This uncertainty is driven, in part, by the reliance on naive comparisons to clinical trial data in patients who may not (and in many cases, are unlikely to) have NTRK fusion mutations. Substantial uncertainty remains regarding PFS and OS after entrectinib treatment for these tumour types in the absence of randomized comparative data and due to the relatively small sample sizes for each tumour type. Among tumour types for which the average health gain was positive and entrectinib increased average costs, the ICER of entrectinib compared to first-line alternatives in patients with known NTRK mutation ranged from $94,645 per QALY gained (MASC, versus sunitinib) to $1,584,600 (breast cancer, versus paclitaxel).
Incorporating the costs of NTRK fusion testing, the cost-effectiveness of entrectinib testing and treatment remained lowest for patients with MASC ($96,021 per QALY gained versus sunitinib and $178,876 per QALY gained versus gefitinib). In all other tumour types, testing and treatment with entrectinib had an ICER exceeding $1,000,000 per QALY gained.
When subsequent therapy costs are included, ICERs for patients with a known NTRK mutation range from $195,302 (MASC, versus gefitinib) to $2,590,977 (breast cancer, versus paclitaxel). The impact of including subsequent therapy costs for each tumour site is outlined in Table 29.
Price Reduction Analysis
For each clinical indication comparator pair, CADTH identified the threshold price at which the ICER would be $50,000 per QALY gained. The results are outlined in Table 27 (versus subsequent lines of therapy). An additional price reduction analysis was performed against first-line therapies (Table 28). There is substantial heterogeneity in the price reductions required across tumour sites to achieve cost-effectiveness at a threshold of $50,000 per QALY. When the cost of testing to identify eligible patients is included, price reductions can only make entrectinib cost-effective at a willingness-to-pay threshold of $50,000 per QALY gained for clinical indications in which NTRK fusion is characteristic (i.e., secretory breast cancer and MASC).
Table 5: Summary of the CADTH Reanalysis for Second-Line Comparators: Incremental Costs, QALYs, and Cost-Effectiveness of Entrectinib in Patients Known and Not Known to Have an NTRK Fusion Cancer
Tumour site (vs. drug comparator) | Incremental cost (95% CI) | Incremental cost of testing (95% CI) | Incremental QALY (95% CI) | Probability that entrectinib is clinically dominated | ICER (95% CI) | ICER, including testing costs (95% CI) |
|---|---|---|---|---|---|---|
Breast, secretory (vs. carboplatin) | $69,847 ($2,454 to $96,244) | $1,492 ($1,400 to $2,156) | 0.21 (–0.37 to –0.42) | 9.9% | $285,289 ($46,688 to dominated) | $290,655 ($60,934 to dominated) |
Breast, secretory (vs. eribulin) | $67,290 (–$2,338 to $95,185) | $1,501 ($1,400 to $2,230) | 0.19 (–0.39 to –0.41) | 10.0% | $288,673 ($45,750 to dominated) | $294,420 ($53,783 to dominated) |
Breast, non-secretory (vs. carboplatin) | $69,811 ($3,003 to $97,083) | $8,450,567 ($290,276 to $45,567,192) | 0.21 (–0.36 to –0.42) | 9.6% | $285,984 ($60,629 to dominated) | $8,381,912 ($1,240,340 to dominated) |
Breast, non-secretory (vs. eribulin) | $67,301 (–$2,256 to $95,185) | $5,655,132 ($1,400 to $27,831,282) | 0.19 (–0.39 to –0.41) | 10.0% | $288,680 ($46,017 to dominated) | $3,831,977 ($159,208 to dominated) |
Colorectal (vs. pembrolizumab) | –$17,534 (–$62,128 to $15,487) | $2,262,316 ($127,489 to $13,275,956) | –0.08 (–0.49 to –0.07) | 68.2% | ICER of comparator vs. entrectinib: $229,650 (cost-saving to dominated) | Dominated ($4,609,896 to dominated) |
Colorectal (vs. bevacizumab + FOLFOX) | –$41,289 (–$91,457 to $9,644) | $2,974,016 ($129,143 to $16,460,883) | –0.07 (–0.38 to –0.08) | 62.5% | ICER of comparator vs. entrectinib: $629,100 (cost-saving to dominated) | Dominated ($3,681,180 to dominated) |
Colorectal (vs. FOLFOX) | $21,331 (–$34,950 to $82,912) | $3,335,467 ($126,978 to $17,174,338) | –0.15 (–0.70 to –0.04) | 85.2% | Dominated (dominated to ICER of comparator vs. entrectinib: $5,122 per QALY gained) | Dominated ($12,650,148 to dominated) |
Colorectal (vs. FOLFIRI) | $25,841 (–$21,817 to $77,881) | $3,025,599 ($129,760 to $15,932,605) | –0.13 (–0.66 to –0.05) | 83.1% | Dominated (dominated to ICER of comparator vs. entrectinib: $8,921 per QALY gained) | Dominated ($11,813,787 to dominated) |
NSCLC (vs. pemetrexed and cisplatin) | $76,516 ($10,699 to $108,267) | $6,855,052 ($179,393 to $23,313,833) | 0.52 (0.16 to –0.78) | 0.2% | $149,881 ($29,890 to $405,246) | $2,054,979 ($453,034 to $60,135,130) |
NSCLC (vs. pemetrexed) | $91,353 ($33,351 to $116,958) | $4,449,406 ($173,724 to $21,140,865) | 0.61 (0.31 to –0.84) | 0.0% | $150,093 ($61,258 to $280,273) | $1,639,061 ($409,698 to $39,004,996) |
NSCLC (vs. cisplatin) | $120,374 ($56,654 to $153,165) | $5,139,639 ($174,101 to $25,576,677) | 0.53 (0.14 to –0.81) | 0.6% | $221,058 ($116,942 to $749,070) | $2,095,530 ($485,880 to $71,741,758) |
NSCLC (vs. docetaxel) | $99,079 ($44,800 to $125,001) | $4,819,904 ($168,230 to $25,723,166) | 0.62 (0.32 to –0.85) | 0.0% | $159,441 ($81,945 to $290,603) | $1,686,586 ($415,344 to $45,091,142) |
Pancreatic (vs. gemcitabine + nab-paclitaxel) | $29,943 ($21,903 to $38,093) | $2,189,416 ($98,828 to $9,742,173) | 0.00 (–0.09 to –0.09) | 49.2% | Dominated ($380,269 to dominated) | Dominated ($3,161,075 to dominated) |
Soft tissue sarcoma (vs. eribulin) | $69,180 ($54,841 to $79,597) | $2,203,529 ($186,620 to $9,827,651) | 0.18 (–0.03 to –0.35) | 3.6% | $369,135 ($207,914 to dominated) | $5,317,866 ($1,145,467 to dominated) |
Thyroid (vs. lenvatinib) | $112,928 ($48,493 to $154,724) | $2,377,651 ($15,209 to $6,034,653) | –0.30 (–0.99 to –0.06) | 93.5% | Dominated ($2,158,604 to dominated) | Dominated ($4,755,807 to dominated) |
Thyroid (vs. sorafenib) | $47,014 ($146 to $73,859) | $1,670,278 ($15,094 to $5,727,073) | –0.05 (–0.60 to –0.22) | 53.0% | Dominated ($167,780 to dominated) | Dominated ($470,328 to dominated) |
Othera | 183,444 | 562,085 | 0 | 50% | Dominated | Dominated |
Pooled, excluding other | $66,388 ($49,306 to $79,281) | $3,248,555 ($416,828 to $13,993,730) | 0.11 (–0.06 –to 0.21) | 7.2% | $586,995 ($314,387 to dominated) | $13,529,393 ($3,308,598 to dominated) |
Pooled, including other | $101,377 ($89,401 to $110,416) | $2,445,546 ($460,246 to $9,978,897) | 0.07 (–0.04 to –0.15) | NR | $1,272,991 ($692,763 to dominated) | $16,746,589 ($5,049,732 to dominated) |
CI = confidence interval; ICER = incremental cost-effectiveness ratio; NSCLC = non–small cell lung cancer; NR = not reported; NTRK = neurotrophic tyrosine receptor kinase; QALY = quality-adjusted life-year; vs. = versus.
Note: ICERs with testing costs were calculated by CADTH by adding average testing costs to the incremental costs and recalculating the ICER.
aOther includes neuroblastoma, head and neck cancer, endometrial cancer, ovarian cancer, cholangiocarcinoma, and cancer of unknown primary. For each of these cancer types, the incremental cost was assumed to be the drug cost estimated in the sponsor’s analysis; the incremental QALY benefit was assumed to be 0 because no clinical evidence was provided for these indications. The weight of these in the pooled analysis was determined by their relative prevalence. CADTH notes that there are other cancer types not identified in the trial that are not included here. Therefore, the ICER in the pooled results including “other” may underestimate the ICER.
Issues for Consideration
Larotrectinib is currently undergoing negotiation with the pan-Canadian Pharmaceutical Alliance. If a letter of intent is issued, it may make larotrectinib a commonly used comparator to entrectinib in this patient population.
Overall Conclusions
The CADTH clinical review noted that, due to the lack of any comparative evidence, the clinical effectiveness of entrectinib is highly uncertain. It was noted that response rates varied widely across different tumour types, with a similarly wide range of uncertainty. Combined with the differences in sample sizes across the different tumour types (most subtypes had fewer than 10 patients), the generalizability of the findings to the mixed cancer population is limited.
CADTH undertook reanalyses to address several key limitations of the sponsor’s model. CADTH’s reanalyses included a more plausible extrapolation for long-term PFS and OS; increased the uncertainty in PFS and OS (consistent with the small sample sizes for each patient subtype); assumed no expected treatment benefit in patient subtypes with observational data representing 5 or fewer patients; and included the costs of NGS testing to identify eligible patients. The CADTH reanalysis also stratified tumour types so that statistical and model-based analyses were appropriately performed on homogeneously defined populations, then subsequently combined these into weighted averages to estimate aggregated cost-effectiveness. CADTH was unable to adjust for all levels of uncertainty introduced by the small sample sizes for individual tumour sites, the lack of direct comparative trial data, and the fact that there was no clinical evidence provided for many potential clinical indications of solid tumours with NTRK fusion mutations. The CADTH reanalysis did not combine comparators within tumour types because the comparators varied so substantially in PFS and OS that it was assumed the patients considering these treatment alternatives may be different in clinically meaningful ways; however, how these clinical differences may affect the effectiveness of entrectinib is not known. It is possible that limitations beyond those identified could exist and could result in an underestimate of the true ICER for entrectinib.
Based on the available evidence and analysis performed by CADTH, the ICER of entrectinib compared to second-line treatment options, in the pooled analysis, exceeds $550,000 per QALY gained in patients with known NTRK fusion mutations and exceeds $10,000,000 per QALY gained when incorporating the costs of patient identification. In a scenario analysis that compared entrectinib to first-line comparators, the pooled analysis exceeds $750,000 per QALY gained in patients with known NTRK fusion mutations; incorporating the costs of identifying patients increases the ICER to more than $8,000,000 per QALY gained. In all analyses, there was substantial heterogeneity across tumour (sub)types.
With or without accounting for the cost of patient identification, for the pooled analysis, there is no price reduction that would achieve an overall ICER of $50,000 per QALY gained. However, there are numerous specific indications for which price reduction does make achieving cost-effectiveness at a threshold of $50,000 per QALY gained possible in patients with known NTRK mutations. The tumour sites where cost-effectiveness is most achievable, such as MASC, have high NTRK prevalence and higher QALY gains.
CADTH notes that for many tumour sites, there is substantial uncertainty regarding whether entrectinib provides net clinical benefit compared to the treatment alternatives identified by the sponsor. For numerous indications, there was a greater than 25% probability that entrectinib was clinically dominated (i.e., providing fewer life-years and fewer QALYs) by the alternative treatment. The CADTH analysis likely underestimated the magnitude of this uncertainty, given that the analysis is based on naive comparators with no adjustment for confounding, such as NTRK fusion status.
Finally, although a direct comparison to larotrectinib has not been conducted, CADTH notes that the sponsor presented no evidence to suggest entrectinib will lead to better health outcomes. A naive comparison, as the sponsor conducted against all other comparators, would suggest that entrectinib may have worse outcomes relative to larotrectinib. This is shown in the unadjusted results by Garcia-Foncillas et al.1 There is a substantial amount of confounding when comparing data in this manner, limiting any conclusions that can be drawn. However, this highlights the uncertainty raised by CADTH concerning the sponsor’s approach to its submitted analysis.
References
1.Garcia-Foncillas J, Bokemeyer C, Italiano A, et al. Indirect Treatment Comparison of Larotrectinib versus Entrectinib in Treating Patients with TRK Gene Fusion Cancers. Cancers (Basel). 2022;14(7). PubMed
2.Ara R, Brazier JE. Populating an economic model with health state utility values: moving toward better practice. Value Health. 2010;13(5):509-518. PubMed
3.de Oliveira C, Weir S, Rangrej J, et al. The economic burden of cancer care in Canada: a population-based cost study. CMAJ Open. 2018;6(1):E1-E10. PubMed
4.Ontario Case Costing Initiative (OCCI). Toronto (ON): Ontario Health and Long-Term Care; 2017: https://data.ontario.ca/dataset/ontario-case-costing-initiative-occi. Accessed 2022 Apr.
5.Pharmacoeconomic evaluation [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Rozlytrek (entrectinib), capsules, 100 mg and 200 mg, oral [internal sponsor's package]. Mississauga (ON): Hoffmann-La Roche Limited; 2022 Jan 25.
6.Guidelines for the economic evaluation of health technologies: Canada. 4th ed. Ottawa (ON): CADTH; 2017: https://www.cadth.ca/guidelines-economic-evaluation-health-technologies-canada-4th-edition. Accessed 2022 May 2.
7.Guidelines for Economic Evaluations of Tumour-Agnostic Products. Ottawa (ON): CADTH; 2021: https://www.cadth.ca/sites/default/files/pdf/mh0016-cadth-economic-guidance-for-tumor-agnostic-products-rev-june22.pdf. Accessed 2022 May 2.
8.Zaric GS. The impact of ignoring population heterogeneity when Markov models are used in cost-effectiveness analysis. Med Decis Making. 2003;23(5):379-396. PubMed
9.Westphalen CB, Krebs MG, Le Tourneau C, et al. Genomic context of NTRK1/2/3 fusion-positive tumours from a large real-world population. NPJ Precis Oncol. 2021;5(1):69. PubMed
10.Wong D, Yip S, Sorensen PH. Methods for Identifying Patients with Tropomyosin Receptor Kinase (TRK) Fusion Cancer. Pathol Oncol Res. 2020;26(3):1385-1399. PubMed
11.Gatalica Z, Xiu J, Swensen J, Vranic S. Molecular characterization of cancers with NTRK gene fusions. Mod Pathol. 2019;32(1):147-153. PubMed
12.Hechtman JF, Benayed R, Hyman DM, et al. Pan-Trk Immunohistochemistry Is an Efficient and Reliable Screen for the Detection of NTRK Fusions. Am J Surg Pathol. 2017;41(11):1547-1551. PubMed
13.Rudzinski ER, Lockwood CM, Stohr BA, et al. Pan-Trk Immunohistochemistry Identifies NTRK Rearrangements in Pediatric Mesenchymal Tumors. Am J Surg Pathol. 2018;42(7):927-935. PubMed
14.Feng J, Ebata K, Hansen F, et al. TRK wild-type and fusion protein expression in solid tumors: Characterization by immunohistochemistry and in situ hybridization. Annals of Oncology. 2018;29:vi27.
15.Hsiao SJ, Zehir A, Sireci AN, Aisner DL. Detection of Tumor NTRK Gene Fusions to Identify Patients Who May Benefit from Tyrosine Kinase (TRK) Inhibitor Therapy. J Mol Diagn. 2019;21(4):553-571. PubMed
16.Solomon JP, Linkov I, Rosado A, et al. NTRK fusion detection across multiple assays and 33,997 cases: diagnostic implications and pitfalls. Mod Pathol. 2020;33(1):38-46. PubMed
17.de Oliveira C, Pataky R, Bremner KE, et al. Phase-specific and lifetime costs of cancer care in Ontario, Canada. BMC Cancer. 2016;16(1):809. PubMed
18.DeltaPA. [Ottawa (ON)]: IQVIA; 2022: https://www.iqvia.com/. Accessed 2022 Mar 25.
19.Cancer Care Ontario: funded evidence-informed regimens. 2022; https://www.cancercareontario.ca/en/drugformulary/regimens. Accessed 2022 Mar 04.
20.Hoffmann-La Roche Ltd. Rozlytrek (entrectinib): capsules, 100 mg and 200 mg, oral [product monograph]. Mississauga (ON): Hoffmann-La Roche Limited; 2021 Jan 7.
21.Bayer Inc. Vitrakvi (larotrectinib): capsules, 25 mg and 100 mg, oral [product monograph]. Mississauga (ON): Bayer Inc.; 2022 Jan 24: https://pdf.hres.ca/dpd_pm/00064510.PDF.
22.CADTH Reimbursement Review Clinical and Pharmacoeconomic Report: Larotrectinib (Vitrakvi). Ottawa (ON): CADTH; 2021: https://www.cadth.ca/sites/default/files/DRR/2021/PC0221-Vitrakvi-combined-report-meta.pdf. Accessed 2022 Mar 29.
23.CADTH Reimbursement Review Clinical and Pharmacoeconomic Report: Pembrolizumab (Keytruda). Ottawa (ON): CADTH; 2021: https://www.cadth.ca/sites/default/files/DRR/2022/PC0250CL-Keytruda.pdf. Accessed 2022 Mar 29.
24.Exceptional Access Program (EAP). Toronto (ON): Ontario Ministry of Health; Ontario Ministry of Long-Term Care; 2021: https://www.health.gov.on.ca/en/pro/programs/drugs/odbf/odbf_except_access.aspx. Accessed 2022 Mar 16.
25.Ontario Ministry of Health, Ontario Ministry of Long-Term Care. Ontario drug benefit formulary/comparative drug index. 2021; https://www.formulary.health.gov.on.ca/formulary/. Accessed 2022 Mar 16.
26.Man D, Wu J, Shen Z, Zhu X. Prognosis of patients with neuroendocrine tumor: a SEER database analysis. Cancer Manag Res. 2018;10:5629-5638. PubMed
27.Canadian Cancer Statistics Advisory Committee. Canadian Cancer Statistics 2021. Toronto (ON): Canadian Cancer Society; 2021: https://cdn.cancer.ca/-/media/files/research/cancer-statistics/2021-statistics/2021-pdf-en-final.pdf. Accessed 2021 April 27.
28.Canadian Institute for Health Information. National Health Expenditure Trends, 2021 2021; https://www.cihi.ca/en/national-health-expenditure-trends. Accessed 30 Apr 2022.
29.Canadian Cancer Statistics Advisory Committee in collaboration with the Canadian Cancer Society, Statistics Canada and the Public Health Agency of Canada. Canadian Cancer Statistics: A 2018 special report on cancer incidence by stage. Toronto (ON): Canadian Cancer Society; 2018: https://cdn.cancer.ca/-/media/files/research/cancer-statistics/2018-statistics/canadian-cancer-statistics-2018-en.pdf. Accessed 2022 Mar 11.
30.Canadian Cancer Statistics Advisory Committee in collaboration with the Canadian Cancer Society, Statistics Canada and the Public Health Agency of Canada. Canadian Cancer Statistics: A 2018 special report on cancer incidence by stage. Supplementary Data. Toronto (ON): Canadian Cancer Society; 2018: https://cdn.cancer.ca/-/media/files/research/cancer-statistics/2018-statistics/2018-past-editions/online-supplementary-data-2018.xlsx?rev=7da8b2e62cd54efea0ead28fb35add24&hash=AE40353DFDDE06D243F6CB6C3BCAA074&_ga=2.230204382.370804896.1647001632-911802007.1632235443. Accessed 2022 Mar 11.
31.Bebb DG, Banerji S, Blais N, et al. Canadian Consensus for Biomarker Testing and Treatment of TRK Fusion Cancer in Adults. Curr Oncol. 2021;28(1):523-548. PubMed
32.Brenner DR, Weir HK, Demers AA, et al. Projected estimates of cancer in Canada in 2020. CMAJ. 2020;192(9):E199-E205. PubMed
33.Canadian Cancer Society. Soft tissue sarcoma statistics. https://cancer.ca/en/cancer-information/cancer-types/soft-tissue-sarcoma/statistics. Accessed 2022 Mar 11.
34.Anderson JL, Haidar YM, Armstrong WB, Tjoa T. Analysis of Clinical Features of Mammary Analog Secretory Carcinoma Using the Surveillance, Epidemiology, and End Results Database. JAMA Otolaryngol Head Neck Surg. 2019;145(1):91-93. PubMed
35.Brule SY, Al-Baimani K, Jonker H, et al. Palliative systemic therapy for advanced non-small cell lung cancer: Investigating disparities between patients who are treated versus those who are not. Lung Cancer. 2016;97:15-21. PubMed
36.Bylsma LC, Gillezeau C, Garawin TA, et al. Prevalence of RAS and BRAF mutations in metastatic colorectal cancer patients by tumor sidedness: A systematic review and meta-analysis. Cancer Med. 2020;9(3):1044-1057. PubMed
37.Gainor JF, Shaw AT. Novel targets in non-small cell lung cancer: ROS1 and RET fusions. Oncologist. 2013;18(7):865-875. PubMed
38.Rosen EY, Goldman DA, Hechtman JF, et al. TRK Fusions Are Enriched in Cancers with Uncommon Histologies and the Absence of Canonical Driver Mutations. Clin Cancer Res. 2020;26(7):1624-1632. PubMed
39.Skalova A, Vanecek T, Simpson RH, et al. Mammary Analogue Secretory Carcinoma of Salivary Glands: Molecular Analysis of 25 ETV6 Gene Rearranged Tumors With Lack of Detection of Classical ETV6-NTRK3 Fusion Transcript by Standard RT-PCR: Report of 4 Cases Harboring ETV6-X Gene Fusion. Am J Surg Pathol. 2016;40(1):3-13. PubMed
40.Zehir A, Benayed R, Shah RH, et al. Mutational landscape of metastatic cancer revealed from prospective clinical sequencing of 10,000 patients. Nature Medicine. 2017;23(6):703-713. PubMed
41.Farago AF, Taylor MS, Doebele RC, et al. Clinicopathologic Features of Non-Small-Cell Lung Cancer Harboring an NTRK Gene Fusion. JCO Precis Oncol. 2018;2018.
42.Canadian Institute for Health Information. National Health Expenditure Trends, 1975 to 2018. Ottawa, ON: CIHI; 2018: https://www.cihi.ca/sites/default/files/document/nhex-trends-narrative-report-2018-en.pdf.
43.Budget Impact Analysis [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Rozlytrek (entrectinib), capsules, 100 mg and 200 mg, oral [internal sponsor's package]. Mississauga (ON): Hoffman-La Roche Limited; 2022 Jan 25.
44.CADTH Reimbursement Recommendation: Larotrectinib (Vitrakvi). Ottawa (ON): CADTH; 2021: https://www.cadth.ca/sites/default/files/DRR/2021/PC0221%20Vitrakvi%20-%20CADTH%20Final%20Rec%20KG_NA_Corrected-meta.pdf. Accessed 2022 Apr 27.
45.pan-Canadian Pharmaceutical Alliance. Vitrakvi (larotrectinib). 2022; https://www.pcpacanada.ca/negotiation/21598. Accessed 2022 Apr 29.
46.Canadian Institute for Health Information. Who is paying for these services? 2021; https://www.cihi.ca/en/who-is-paying-for-these-services. Accessed 2022 Apr 27.
47.Table: 17-10-0057-01. Projected population, by projection scenario, age and sex, as of July 1 (x 1,000). Ottawa (ON): Statistics Canada; 2022: https://www150.statcan.gc.ca/t1/tbl1/en/tv.action?pid=1710005701. Accessed 2022 May 02.
48.Uterine Cancer: Statistics. 2022; https://www.cancer.net/cancer-types/uterine-cancer/statistics. Accessed 2022 May 02.
49.Kassirian S, Dzioba A, Hamel S, et al. Delay in diagnosis of patients with head-and-neck cancer in Canada: impact of patient and provider delay. Curr Oncol. 2020;27(5):e467-e477. PubMed
50.Neuroendocrine Tumour (NET) Incidence Significantly Increases in Canada. 2016; https://cnets.ca/2015/02/neuroendocrine-tumour-net-incidence-significantly-increases-in-canada/. Accessed 2022 May 5.
Appendix 1: Cost Comparison Table
Note that this appendix has not been copy-edited.
The comparators presented in the following table have been deemed to be appropriate based on feedback from clinical experts. Comparators may be recommended (appropriate) practice or actual practice. Existing Product Listing Agreements are not reflected in the table and as such, the table may not represent the actual costs to public drug plans.
Table 6: CADTH Cost Comparison Table for the Treatment of Solid Tumours With NTRK Gene Fusion
Treatment | Strength / concentration | Form | Price | Recommended dosage | Daily cost | Cost per 28 days |
|---|---|---|---|---|---|---|
Entrectinib (Rozlytrek) | 100 mg 200 mg | Capsule | $47.6667a $95.3333a | 600 mg daily, may be reduced to 400 mg or 200 mg based on tolerabilityb | $286.00 | $8,008 |
Larotrectinib (Vitrakvi) | 25 mg 100 mg | Capsule | $52.3375c $209.3500c | 100 mg twice daily, may be reduced to 75 mg or 50 mg twice daily, or 100 mg daily based on tolerabilityb | $418.70 | $11,724 |
Non–small cell lung cancer - Squamous | ||||||
Carboplatin (Generics) | 50 mg 150 mg 450 mg 600 mg | 10 mg/mL solution for injection | $70.0000 $210.0000 $600.0000 $775.000 | Target AUC 6 on Day 1 every 21 days | $56.90 | $1,593 |
Paclitaxel (generics) | 30 mg | 6 mg/mL solution for injection | $300.0000 | 175 to 200 mg/m2 on Day 1 every 21 days | $157.14 to 171.43 | $4,400 to 4,800 |
Pembrolizumab (Keytruda) | 100 mg | 25 mg/mL solution for infusion | $4,400.0000d | 2 mg/kg on Day 1 every 21 days, 200 mg max | $419.05 | $11,733 |
CRBPPACL+PEMB | $633.10 to 647.38 | $17,727 to 18,127 | ||||
CRBPPACL | $214.05 to 228.33 | $5,993 to 6,393 | ||||
Docetaxel (generics) | 20 mg 80 mg 160 mg | 20 mg/mL solution for injection | $249.0000 $497.0000 $990.0000 | 75 mg/m2 on Day 1 every 21 days | $59.24 | $1,659 |
Pembrolizumab (Keytruda) | 100 mg | 25 mg/mL solution for infusion | $4,400.0000d | 2 mg/kg on Day 1 every 21 days, 200 mg max | $419.05 | $11,733 |
Non–small cell lung cancer – Non-Squamous | ||||||
Cisplatin (generics) | 50 mg 100 mg | 1 mg/mL solution for injection | $135.0000 $270.0000 | 75 mg/m2 on Day 1 every 21 days | $19.29 | $540 |
Pemetrexed (generics) | 100 mg 500 mg 1,000 mg | 10 mg/mL lyophilized powder | $429.0000 $2,145.0000 $4,290.0000 | 500 mg/m2 Day 1 every 21 days | $183.86 | $5,148 |
Pembrolizumab (Keytruda) | 100 mg | 25 mg/mL solution for infusion | $4,400.0000d | 2 mg/kg on Day 1 every 21 days, 200 mg max | $419.05 | $11,733 |
CISPPEME+PEMB | $622.19 | $17,421 | ||||
CISPPEME | $203.14 | $5,688 | ||||
Cisplatin (generics) | 50 mg 100 mg | 1 mg/mL solution for injection | $135.0000 $270.0000 | 75 mg/m2 on Day 1 every 21 days | $19.29 | $540 |
Docetaxel (generics) | 20 mg 80 mg 160 mg | 20 mg/mL solution for injection | $249.0000 $497.0000 $990.0000 | 75 mg/m2 on Day 1 every 21 days | $59.24 | $1,669 |
Pembrolizumab (Keytruda) | 100 mg | 25 mg/mL solution for infusion | $4,400.0000d | 2 mg/kg on Day 1 every 21 days, 200 mg max | $419.05 | $11,733 |
Pemetrexed (generics) | 100 mg 500 mg 1,000 mg | 10 mg/mL lyophilized powder | $429.0000 $2,145.0000 $4,290.0000 | 500 mg/m2 Day 1 every 21 days | $183.86 | $5,148 |
Colorectal cancer | ||||||
Oxaliplatin (generics) | 100 mg 200 mg | 5 mg/mL solution for injection | $72.5400 $145.0800 | 85 mg/m2 Day 1 every 14 days | $10.36 | $290 |
Leucovorin (generics) | 50 mg 500 mg | 10 mg/mL solution for injection | $68.9400 $689.0000 | 400 mg/m2 Day 1 every 14 days | $73.84 | $2,067 |
Fluorouracil (generics) | 5,000 mge | 50 mg/mL solution for injection | $160.9000 | 400 mg/m2 on Day 1, then 2,400 mg/m2 continuous infusion over 46 hours starting Day 1 every 14 days | $11.58e | $324 |
Bevacizumab (SEBs) | 100 mg 400 mg | 25 mg/mL solution for injection | $347.0000 $1,388.0000 | 5 mg/kg on Day 1 every 14 days | $99.14 | $2,226 |
MFOLFOX6+BEVA | $194.93 | $5,458 | ||||
MFOLFOX6 | $95.78 | $2,682 | ||||
Irinotecan (generics) | 40 mg 100 mg 500 mg | 20 mg/mL | $208.3500 $520.8500 $2,604.2500 | 180 mg/m2 on Day 1 every 14 days | $126.49 | $3,542 |
Leucovorin (generics) | 50 mg 500 mg | 10 mg/mL solution for injection | $68.9400 $689.0000 | 400 mg/m2 Day 1 every 14 days | $73.84 | $2,067 |
Fluorouracil (generics) | 5,000 mge | 50 mg/mL solution for injection | $160.9000 | 400 mg/m2 on Day 1, then 2,400 mg/m2 continuous infusion over 46 hours starting Day 1 every 14 days | $11.58e | $324 |
FOLFIRI | $211.91 | $5,934 | ||||
Breast Cancer | ||||||
Carboplatin (Generics) | 50 mg 150 mg 450 mg 600 mg | 10 mg/mL solution for injection | $70.0000 $210.0000 $600.0000 $775.000 | Target AUC 6 on Day 1 every 21 days | $56.90 | $1,593 |
Docetaxel (generics) | 20 mg 80 mg 160 mg | 20 mg/mL solution for injection | $249.0000 $497.0000 $990.0000 | 75 mg/m2 on Day 1 every 21 days | $59.24 | $1,669 |
Eribulin (Halaven) | 1 mg | 0.5 mg/mL solution for injection | $422.0000 | 1.4 mg/m2 on Days 1 and 8 every 21 days | $120.57 | $3,376 |
Mammary analogue secretory carcinoma | ||||||
Cisplatin (generics) | 50 mg 100 mg | 1 mg/mL solution for injection | $135.0000 $270.0000 | 70 mg/m2 on Day 1 every 21 days | $19.29 | $540 |
Gemcitabine (generics) | 1,000 mg 2,000 mg | 40 mg/mL lyophilized powder | $270.0000 $540.0000 | 1,000 mg/m2 on Days 1 and 8 every 21 days | $51.43 | $1,440 |
CISPGEMC | $70.71 | $1,980 | ||||
Gefitinib (generics) | 250 mg | Tablets | $62.3050 | 250 mg daily | $62.31 | $1,745 |
Sunitinib (Sutent) | 12.5 mg 25 mg 50 mg | Capsules | $64.4157f $128.8308f $257.6610f | 37.5 mg daily | $193.25 | $5,411 |
Soft tissue sarcoma | ||||||
Doxorubicin (generics) | 10 mg 50 mg 200 mg | 2 mg/mL lyophilized powder | $50.0000 $255.0000 $770.0000 | 50 to 75 mg/m2 on Day 1 every 21 days | $21.67 to 33.81 | $607 to 947 |
Eribulin (Halaven) | 1 mg | 0.5 mg/mL solution for injection | $422.0000 | 1.4 mg/m2 on Days 1 and 8 every 21 days | $120.57 | $3,376 |
Imatinib (generics) | 400 mg | Tablets | $20.8314f | 400 mg twice daily | $41.66 | $1,167 |
Pancreatic cancer | ||||||
Oxaliplatin (generics) | 100 mg 200 mg | 5 mg/mL solution for injection | $72.5400 $145.0800 | 85 mg/m2 Day 1 every 14 days | $10.36 | $290 |
Leucovorin (generics) | 50 mg 500 mg | 10 mg/mL solution for injection | $68.9400 $689.0000 | 400 mg/m2 Day 1 every 14 days | $73.84 | $2,067 |
Irinotecan (generics) | 40 mg 100 mg 500 mg | 20 mg/mL | $208.3500 $520.8500 $2,604.2500 | 180 mg/m2 on Day 1 every 14 days | $126.49 | $3,542 |
Fluorouracil (generics) | 5,000 mge | 50 mg/mL solution for injection | $160.9000 | 400 mg/m2 on Day 1, then 2,400 mg/m2 continuous infusion over 46 hours starting Day 1 every 14 days | $11.58e | $324 |
FOLFIRINOX | $228.28 | $6,224 | ||||
Gemcitabine (generics) | 1,000 mg 2,000 mg | 40 mg/mL lyophilized powder | $270.0000 $540.0000 | 1,000 mg/m2 on Days 1, 8, and 15 every 28 days | $57.86 | $1,620 |
Nab-paclitaxel (Abraxane) | 100 mg | 2 mg/mL lyophilized powder | $971.0000 | 125 mg/m2 on Days 1, 8, and 15 every 28 days | $208.07 | $5,826 |
GEMCNPAC | $265.93 | $7,446 | ||||
Neuroendocrine cancer | ||||||
Octreotide (generics) | 10 mg 20 mg 30 mg | Vial, powder for injectable suspension | $990.6975 $1,279.9350 $1,642.1400 | 10 to 30 mg IM on Day 1 every 28 days | $35.38 to $58.65 | $991 to $1,642 |
Thyroid cancer - papillary | ||||||
Lenvatinib (Lenvima) | 24 mg | Capsules | $233.8697f | 24 mg daily | $233.87 | $6,548 |
Sorafenib (Nexavar) | 200 mg | Tablets | $46.4689f | 400 mg twice daily | $185.88 | $5,205 |
Thyroid cancer - anaplastic | ||||||
Doxorubicin (generics) | 10 mg 50 mg 200 mg | 2 mg/mL lyophilized powder | $50.0000 $255.0000 $770.0000 | 50 to 75 mg/m2 on Day 1 every 21 days | $21.67 to 33.81 | $607 to 947 |
Paclitaxel (generics) | 30 mg | 6 mg/mL solution for injection | $300.0000 | 175 to 200 mg/m2 on Day 1 every 21 days | $157.14 to 171.43 | $4,400 to 4,800 |
CARB = carboplatin; CISP = cisplatin; IM = intramuscular; NSCLC = non–small cell lung cancer; PACL = paclitaxel; PEMB = pembrolizumab; PEME = pemetrexed; SEB = subsequent entry biologic.
All prices are IQVIA Delta PA wholesale list prices (March 2022),18 unless otherwise indicated, and do not include dispensing fees or administration. Costs assume a body weight of 75 kg or a body surface area of 1.8 m2 and include wastage of unused medication in vials. Regimen dosing is from the Cancer Care Ontario Regimen database unless otherwise indicated.19
aSponsor’s submitted price.5
cAs submitted to CADTH for the review of Vitrakvi.22
dAs submitted to CADTH for the review of Keytruda.23
eBulk pharmacy vial, no wastage was assumed.
fList price from Ontario Drug Benefit Formulary or Exceptional Access Program.24,25
Appendix 2: Submission Quality
Note that this appendix has not been copy-edited.
Description | Yes/No | Comments |
|---|---|---|
Population is relevant, with no critical intervention missing, and no relevant outcome missing | No | Model provided by sponsor aggregated heterogeneous populations together into a single analysis. |
Model has been adequately programmed and has sufficient face validity | No | Model organization was poor with many unnecessary intermediate calculations through which the sponsor introduced computational errors. Model extensively used IFERROR and CHOOSE functions masking model parameters driving the analysis. |
Model structure is adequate for decision problem | No | Partitioned survival model assumed independence of the PFS and OS that has not been established empirically. Sponsor included comparison against first-line treatment alternatives without considering the possibility that current first-line alternatives would become active second-line alternatives. Model structure did not allow for this possibility to be explored effectively and fulsomely. |
Data incorporation into the model has been done adequately (e.g., parameters for probabilistic analysis) | No | Sponsor performed survival analysis on heterogeneous population using methods that rely on the assumption of a common population. Sponsor used this aggregate PFS and OS analysis to inform the PFS and OS of tumour-stratified analyses instead of using the outcomes of patients with those specific tumour sites, despite high observed heterogeneity in overall response rate and duration of response across tumour types. |
Parameter and structural uncertainty were adequately assessed; analyses were adequate to inform the decision problem | No | Sponsor’s analysis assumed no waning of treatment effect. Sponsor used distribution of patients in their clinical trials, instead of prevalence based weights, to create a pooled analysis; Sponsor only used weights on the comparator arms and assumed that patients receiving entrectinib, regardless of tumour type, have the same PFS and OS despite high observed heterogeneity in overall response rate and duration of response across tumour types. |
The submission was well organized and complete; the information was easy to locate (clear and transparent reporting; technical documentation available in enough details) | No | Model organization was poor with many unnecessary intermediate calculations through which the sponsor introduced computational errors. Model extensively used IFERROR and CHOOSE functions masking model parameters driving the analysis. Some inputs were physically blacked-out. Some sections of input sheets were hidden, greyed out, and some (unused) model columns were in error. Not all data were provided to CADTH, such as PFS and OS for individual tumour sites, despite numerous requests. |
Appendix 3: Additional Information on the Submitted Economic Evaluation
Note that this appendix has not been copy-edited.
Table 8: Sponsor’s Assumptions Regarding Testing by Tumour Site
Tumour site | Status quo testing assumptions | Incremental testing assumptions | Incremental cost per case detected |
|---|---|---|---|
Breast—secretory | None | 100% NGS | $1,467 |
Breast—non-secretory | None | 100% IHC 9% of IHC followed up with NGS | $158,462 |
Colorectal cancer | None | 100% IHC 9% of IHC followed up with NGS | $68,667 |
MASC | None | 100% NGS | $1,556 |
NSCLC | 100% with NGS | No additional testing required | $0 |
Neuroendocrine | None | 100% IHC 9% of IHC followed up with NGS | $1,075 |
Pancreatic | None | 100% IHC 9% of IHC followed up with NGS | $50,244 |
Soft tissue sarcoma | None | 100% IHC 9% of IHC followed up with NGS | $89,565 |
Thyroid cancer | None | 100% IHC 9% of IHC followed up with NGS | $8,957 |
“Other” | None | 100% IHC 9% of IHC followed up with NGS | $1,075 |
Appendix 4: Additional Details on the CADTH Reanalyses and Sensitivity Analyses of the Economic Evaluation
Note that this appendix has not been copy-edited.
Figure 2: Prevalence of NTRK Fusion–Positive Tumours in Adult Patients
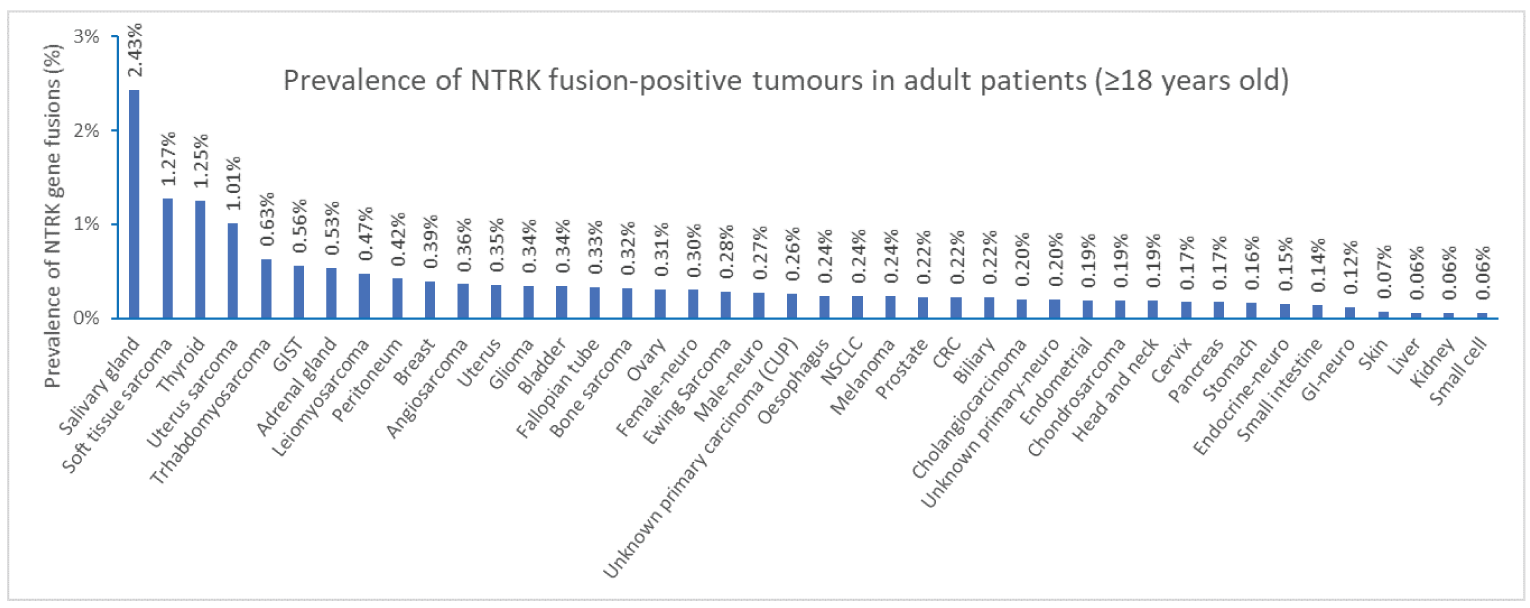
CRC = colorectal cancer; GI = gastrointestinal; GIST = gastrointestinal stromal tumour; NSCLC = non–small cell lung cancer; NTRK = neurotrophic tyrosine receptor kinase.
Source: Westphalen, C.B., Krebs, M.G., Le Tourneau, C. et al. Genomic context of NTRK1/2/3 fusion–positive tumours from a large real-world population. npj Precis. Onc. 5, 69 (2021). Copyright Springer Nature 2021. Reprinted in accordance with CC BY 4.0. https://creativecommons.org/licenses/by/4.0/9
Table 9: CADTH Reanalysis: NTRK Testing Costs by Tumour Site
Tumour site | NTRK fusion prevalence (95% CI) | Number needed to treat using universal NGS to find 1 NTRK fusion cancer (95% CI)b | Cost per case detected, universal NGS testing (95% CI) |
|---|---|---|---|
Breast—secretory | 95.45% (75.00% - 98.00%) | 1.1 (1 - 2) | $1,501 ($1,400 - $2,221) |
Breast—non-secretory | 0.13% (0.08% - 0.22%) | 6,036 (207 - 32,548) | $8,450,567 ($290,276 - $45,567,192) |
Colorectal cancer | 0.30% (0.10% - 0.50%) | 2,382 (91 - 12,267) | $3,335,467 ($126,978 - $17,174,338) |
MASC | 90.00% (75.00% - 95.00%) | 1.2 (1 - 2) | $1,643 ($1,400 - $3,054) |
NSCLC | 0.22% (0.10% - 0.34%) | 3,671 (124 - 18,269) | $5,139,639 ($174,101 - $25,576,677) |
Neuroendocrine | 0.15% (0.10% - 0.3%) | 739 (374, 1232) | $1,034,081 ($523,607 - $1,724,260) |
Pancreatic | 0.41% (0.10% - 1.00%) | 1,564 (71 - 6,959) | $2,189,416 ($98,828 - $9,742,173) |
Soft tissue sarcoma | 0.23% (0.10% - 1.00%) | 1,336 (138 - 6,741) | $1,870,088 ($193,368 - $9,436,777) |
Thyroid cancer | 2.30% (1.00% - 7.70%) | 1,698 (11 - 4,310) | $2,377,651 ($15,209 - $6,034,653) |
“Other”* | |||
Cancer of unknown primary | 0.26%a | 385 | $538,461 |
Cholangiocarcinoma | 0.20%a | 500 | $700,000 |
Endometrial | 0.19%a | 526 | $736,842 |
Head and Neck | 0.19%a | 526 | $736,842 |
Neuroblastoma | 0.52%a | 192 | $269,231 |
Ovarian | 0.31%a | 323 | $451,612 |
aWestphalen et al. 2021.9
bThe number needed to test in order to identify 1 NTRK fusion cancer patient were generated probabilistically using beta distributions fit to the NTRK fusion prevalence
Table 10: Inputs Used to Derive PFS and OS Estimates CADTH Reanalysis
Tumour site | Median PFS (SE)c | Exponential rate for the observed period Mean (95% CI)a | Observed periodb | Exponential rate for the extrapolation period Mean (95% CI)a |
|---|---|---|---|---|
Progression-free survival | ||||
Breast cancer | 10.1 (4.7) | 0.0270 (0.0083 - 0.1448) | 10 | 0.0195 (0.0162 - 0.0240) |
Colorectal cancer | 2.4 (6.8) | 0.1503 (0.0102 - 0.3199) | 2.5 | 0.0220 (0.0200 - 0.0243) |
MASC | 70% at 21 months (represented as beta(9.1, 3.9)) | 0.0042 (0.0011 - 0.0092) | 21 | 0.0392 (0.0266 - 0.0620) |
NSCLC | 14 (5.1) | 0.0153 (0.0067 - 0.0397) | 15 | 0.0303 (0.0275 - 0.0335) |
Neuroendocrine | Assumed same PFS distribution as comparator | 0.0089 (0.0081 - 0.0098) | NA | 0.0089 (0.0081 - 0.0098) |
Pancreatic | Assumed same PFS distribution as comparator | 0.0252 (0.0210 - 0.0309) | NA | 0.0252 (0.0210 - 0.0309) |
Soft tissue sarcoma | 10 (1.8) | 0.0166 (0.0118 - 0.0249) | 10 | 0.0641 (0.0440 - 0.1008) |
Thyroid cancer | 11.8 (2.7) | 0.0144 (0.0094 - 0.0245) | 12 | 0.0088 (0.0073 - 0.0108) |
Overall survival | ||||
Breast cancer | 23.9 (9.4) | 0.0106 (0.0038 - 0.0266) | 10 | 0.0063 (0.0052 - 0.0078) |
Colorectal cancer | 16 (6.8) | 0.0163 (0.0054 - 0.0516) | 2.5 | 0.0124 (0.0113 - 0.0137) |
MASC | 92% at 27 months (represented as beta(11.96, 1.04)) | 0.0007 (0.0000 - 0.0027) | 21 | 0.0065 (0.0044 - 0.0102) |
NSCLC | 75% at 15 months (represented as beta) (9.75 to 3.25) | 0.0046 (0.0010 - 0.0109) | 15 | 0.0136 (0.0123 - 0.0150) |
Neuroendocrine | Assumed same OS distribution as comparator | 0.0025 (0.0023 - 0.0027) | NA | 0.0025 (0.0023 - 0.0027) |
Pancreatic | Assumed same OS distribution as comparator | 0.0145 (0.0121 - 0.0179) | NA | 0.0145 (0.0121 - 0.0179) |
Soft tissue sarcoma | 19 (3.1) | 0.0087 (0.0064 - 0.0126) | 10 | 0.0124 (0.0085 - 0.0193) |
Thyroid cancer | 22.0 (6.6) | 0.0083 (0.0046 - 0.0175) | 12 | 0.0054 (0.0045 - 0.0066) |
aMean and empiric 95% CI of 6,000 simulations
bEnd of observed period estimated to be the point at which 50% of the population was no longer at risk for progression or death as determined by median PFS if number-at-risk information was not available
cProbabilistic analysis used a truncated normal distribution (with minimum value of 0.5 months) around the median survival using the standard error (SE) noted when median PFS (OS) were observed. Exponential rates where then determined directly from median PFS and OS. For cases where median PFS or OS have not yet been reached or reported, another critical point was established on Kaplan-Meier curves based on when 50% of the at-risk population remained. Probabilistic analysis used a beta distribution around that critical point to establish the exponential rate.
Table 11: CADTH Reanalysis: NTRK Testing Costs by Tumour Site
Tumour site | Agea | Annual age-specific health care costs unrelated to cancerb | Annual incremental cost of non-treatment cancer carec | Total non-treatment health care costs (Annual) | 7-day cycle cost |
|---|---|---|---|---|---|
Breast cancer | 60 | 8,344 | 5,450 | 13,794 | 264.36 |
Colorectal cancer | 68 | 6,686 | 7,209 | 13,895 | 266.29 |
MASC | 63 | 7,225 | 5,450 | 12,675 | 242.92 |
NSCLC | 66 | 7,241 | 7,209 | 14,450 | 276.93 |
Neuroendocrine | 62 | 7,218 | 5,450 | 12,668 | 242.78 |
Pancreatic | 66 | 9,247 | 7,209 | 16,456 | 315.38 |
Soft tissue sarcoma | 61 | 7,230 | 5,450 | 12,680 | 243.02 |
Thyroid cancer | 47 | 4,200 | 3,255 | 7,455 | 142.88 |
aSEER database (2018)26 and Canadian Cancer Statistics (2021).27
bNational Health Expenditure Trends, 2021.28
cde Oliveira, 2016.
Table 12: Weights Used to Derive Pooled Analysis Results: Comparison of Values Used in CADTH Reanalysis to Those Used in the Sponsor’s Pharmacoeconomic and Budget Impact Analyses
Tumour site | CADTH reanalysis (% used to derive weight)c | Sponsor Prevalence based estimatea (% of patients) | Sponsor clinical trial-based estimatesb (% of patients) |
|---|---|---|---|
Breast—secretory | 0.001% | 0.5 (1.2%) | 5.0 (4.1%) |
Breast—non-secretory | 0.4% | 0.5 (1.2%) | 2.0 (1.7%) |
Colorectal cancer | 8.3% | 5.0 (11.6%) | 10.0 (8.3%) |
MASC | 9.5% | 6.0 (14.%) | 24.0 (19.8%) |
NSCLC—squamous | 20.2% | 5.5 (12.8%) | 2.0 (1.7%) |
NSCLC—non-squamous | 5.5 (12.8%) | 20.0 (16.5%) | |
Neuroendocrine | 0.25% | 3.0 (7.0%) | 5.0 (4.1%) |
Pancreatic | 10% | 4.0 (9.3%) | 4.0 (3.3%) |
Soft tissue sarcoma | 0.6% | 1.0 (2.3%) | 26.0 (21.5%) |
Thyroid—papillary | 23.8% | 6.0 (14.0%) | 10.0 (8.3%) |
Thyroid—other | 6.0 (14.0%) | 4.0 (3.3%) | |
“Other” | |||
Cancer of unknown primary | 11.8% | 0.0 (0%) | 9.0 (7.4%) |
Cholangiocarcinoma | 0.4% | ||
Endometrial | 2.8% | ||
Head and Neck | 6.2% | ||
Neuroblastoma | 0.3% | ||
Ovarian | 5.6% | ||
Total (n) | 100% | 43 | 121 |
aSponsor used “prevalence based” estimates were used in the budget impact analysis
bSponsor used clinical trial-based estimates in the cost-effectiveness analysis
cCADTH incidence-based estimates relied on the incidence estimates provided by the sponsor in their budget impact analysis wherever possible. For cancer of unknown primary, cholangiocarcinoma, and pancreatic cancer, previously published CADTH estimates were used. For other cancer types, estimates were generated using the annual number of cancers by type in Canada outside of Quebec, the proportion diagnosed at stage III or IV, and the prevalence of NTRK fusion cancer. Proportion of population differs in the first-line and second-line analysis because some tumour types (e.g., MASC) are only included in one analysis.
Detailed Results of CADTH Base Case: Second-Line Comparators
Table 13: CADTH Reference Case Analysis: Breast Cancer (Versus Carboplatin)
Output | Entrectinib | Comparator | Incremental (95% CI) |
|---|---|---|---|
Life-years | |||
Total life-years | 1.75 (0.88 - 2.17) | 1.51 (1.23 - 1.79) | 0.25 (-0.64 - 0.53) |
QALYs | |||
In PFS | 0.57 (0.06 - 0.81) | 0.30 (0.21 - 0.38) | 0.27 (-0.22 - 0.48) |
In progression | 0.65 (0.17 - 1.09) | 0.72 (0.52 - 0.93) | -0.07 (-0.53 - 0.36) |
Total QALYs | 1.22 (0.64 - 1.54) | 1.02 (0.81 - 1.23) | 0.21 (-0.37 - 0.42) |
Costs | |||
Drug costs | $75,848 ($7,307 - $101,045) | $5,865 ($4,764 - $7,002) | $69,984 ($1,933 - $94,837) |
AE costs | $1,617 ($1,297 - $1,937) | $5,148 ($4,121 - $6,155) | -$3,531 (-$4,586 - -$2,475) |
Other health care costs | $24,196 ($12,079 - $30,659) | $20,802 ($16,517 - $25,343) | $3,394 (-$8,716 - $7,463) |
Total costs | $101,662 ($34,055 - $129,126) | $31,815 ($27,302 - $36,488) | $69,847 ($2,454 - $96,244) |
Results | |||
Percent of simulations entrectinib is clinically dominated | 9.9% | ||
ICER ($ per QALY gained) | $285,289 ($46,688 - Dominated) | ||
NGS Testing costs (Secretory) | $1,492 ($1,400 - $2,156) | $0 | $1,492 ($1,400 - $2,156) |
ICER including secretory testing costs ($ per QALY gained) | $290,655 ($60,934 - Dominated) | ||
NGS Testing costs (Non-Secretory) | $8,450,567 ($290,276 - $45,567,192) | $0 | $8,450,567 ($290,276 - $45,567,192) |
ICER including non-secretory testing costs ($ per QALY gained) | $8,381,912 ($1,240,340 - Dominated) | ||
Table 14: CADTH Reference Case Analysis: Breast cancer (Versus Eribulin)
Output | Entrectinib | Comparator | Incremental (95% CI) |
|---|---|---|---|
Life-years | |||
Total life-years | 1.79 (0.90 - 2.19) | 1.55 (1.25 - 1.84) | 0.24 (-0.65 - 0.53) |
QALYs | |||
In PFS | 0.60 (0.06 - 0.86) | 0.35 (0.26 - 0.45) | 0.24 (-0.26 - 0.46) |
In progression | 0.66 (0.16 - 1.12) | 0.70 (0.49 - 0.92) | -0.05 (-0.54 - 0.39) |
Total QALYs | 1.25 (0.64 - 1.56) | 1.06 (0.84 - 1.28) | 0.19 (-0.39 - 0.41) |
Costs | |||
Drug costs | $79,238 ($7,386 - $105,981) | $16,824 ($13,676 - $20,045) | $62,413 (-$7,489 - $88,464) |
AE costs | $1,613 ($1,295 - $1,928) | $ ($ - $) | $1,613 ($1,295 - $1,928) |
Other health care costs | $24,695 ($12,175 - $31,028) | $21,431 ($16,875 - $26,117) | $3,264 (-$9,110 - $7,431) |
Total costs | $105,546 ($35,567 - $134,620) | $38,256 ($32,782 - $43,830) | $67,290 (-$2,338 - $95,185) |
Results | |||
Percent of simulations entrectinib is clinically dominated | 10.0% | ||
ICER ($ per QALY gained) | $288,673 ($45,750 - Dominated) | ||
NGS Testing costs (Secretory) | $1,501 ($1,400 - $2,230) | $0 | $1,501 ($1,400 - $2,230) |
ICER ($ per QALY gained) | $294,420 ($53,783 - Dominated) | ||
NGS Testing costs (Non-Secretory) | $5,655,132 ($1,400 - $27,831,282) | $0 | $5,655,132 ($1,400 - $27,831,282) |
ICER ($ per QALY gained) | $3,831,977 ($159,208 - Dominated) | ||
Table 15: CADTH Reference Case Analysis: CRC (Versus Pembrolizumab)
Output | Entrectinib | Comparator | Incremental |
|---|---|---|---|
Life-years | |||
Total life-years | 1.94 (1.07 - 2.70) | 2.03 (1.26 - 2.80) | -0.08 (-0.75 - 0.10) |
QALYs | |||
In PFS | 0.27 (0.05 - 0.60) | 0.39 (0.23 - 0.56) | -0.12 (-0.45 - 0.12) |
In progression | 1.01 (0.38 - 1.61) | 0.97 (0.46 - 1.48) | 0.05 (-0.43 - 0.37) |
Total QALYs | 1.28 (0.70 - 1.79) | 1.36 (0.85 - 1.88) | -0.08 (-0.49 - 0.07) |
Costs | |||
Drug costs | $35,784 ($8,188 - $75,705) | $53,759 ($33,342 - $74,503) | -$17,975 (-$60,470 - $13,820) |
AE costs | $1,615 ($1,309 - $1,926) | $ ($ - $) | $1,615 ($1,309 - $1,926) |
Other health care costs | $26,978 ($14,585 - $38,175) | $28,152 ($17,200 - $39,409) | -$1,174 (-$10,587 - $1,453) |
Total costs | $64,377 ($27,269 - $107,239) | $81,911 ($58,724 - $105,645) | -$17,534 (-$62,128 - $15,487) |
Results | |||
Percent of simulations entrectinib is clinically dominated | 68.2% | ||
ICER ($ per QALY gained) | ICER of Comparator vs. Entrectinib: $229,650 (Cost-saving - Dominated) | ||
NGS Testing cost | $2,262,316 ($127,489 - $13,275,956) | $0 | $2,262,316 ($127,489 - $13,275,956) |
ICER ($ per QALY gained) | Dominated ($4,609,896 - Dominated) | ||
Table 16: CADTH Reference Case Analysis: CRC (Versus BEVA/FOLFOX)
Output | Entrectinib | Comparator | Incremental |
|---|---|---|---|
Life-years | |||
Total life-years | 1.51 (1.02 - 1.71) | 1.52 (1.38 - 1.66) | -0.01 (-0.50 - 0.11) |
QALYs | |||
In PFS | 0.39 (0.06 - 0.82) | 0.69 (0.52 - 0.82) | -0.30 (-0.71 - 0.08) |
In progression | 0.64 (0.21 - 1.04) | 0.41 (0.30 - 0.53) | 0.23 (-0.20 - 0.60) |
Total QALYs | 1.03 (0.69 - 1.26) | 1.10 (0.91 - 1.26) | -0.07 (-0.38 - 0.08) |
Costs | |||
Drug costs | $51,659 ($9,587 - $103,136) | $91,804 ($83,034 - $100,519) | -$40,145 (-$88,554 - $9,954) |
AE costs | $1,613 ($1,296 - $1,931) | $2,547 ($2,044 - $3,041) | -$934 (-$1,518 - -$346) |
Other health care costs | $20,925 ($14,038 - $24,648) | $21,135 ($18,335 - $24,018) | -$210 (-$7,070 - $1,587) |
Total costs | $74,197 ($28,241 - $126,538) | $115,486 ($106,352 - $124,923) | -$41,289 (-$91,457 - $9,644) |
Results | |||
Percent of simulations entrectinib is clinically dominated | 62.5% | ||
ICER ($ per QALY gained) | ICER of Comparator vs. Entrectinib: $629,100 (Cost-saving - Dominated) | ||
Testing cost | $2,974,016 ($129,143 - $16,460,883) | $0 | $2,974,016 ($129,143 - $16,460,883) |
ICER ($ per QALY gained) | Dominated ($3,681,180 - Dominated) | ||
Table 17: CADTH Reference Case Analysis: CRC (Versus FOLFOX)
Output | Entrectinib | Comparator | Incremental |
|---|---|---|---|
Life-years | |||
Total life-years | 2.14 (1.34 - 2.63) | 2.27 (1.83 - 2.71) | -0.13 (-0.94 - 0.07) |
QALYs | |||
In PFS | 0.44 (0.06 - 0.96) | 0.79 (0.57 - 1.01) | -0.36 (-0.88 - 0.07) |
In progression | 1.00 (0.36 - 1.58) | 0.79 (0.48 - 1.12) | 0.21 (-0.39 - 0.68) |
Total QALYs | 1.44 (0.89 - 1.82) | 1.59 (1.25 - 1.92) | -0.15 (-0.70 - 0.04) |
Costs | |||
Drug costs | $58,196 ($9,734 - $122,011) | $31,357 ($25,288 - $37,333) | $26,839 (-$25,424 - $87,153) |
AE costs | $1,611 ($1,280 - $1,925) | $5,374 ($4,309 - $6,424) | -$3,763 (-$4,865 - -$2,666) |
Other health care costs | $29,775 ($18,276 - $37,337) | $31,520 ($24,914 - $38,381) | -$1,745 (-$12,874 - $984) |
Total costs | $89,582 ($33,821 - $155,527) | $68,251 ($59,212 - $77,407) | $21,331 (-$34,950 - $82,912) |
Results | |||
Percent of simulations entrectinib is clinically dominated | |||
ICER ($ per QALY gained) | Entrectinib is Dominated (Dominated - ICER of comparator vs. Entrectinib: $5,122/QALY gained) | ||
Testing cost | $3,335,467 ($126,978 - $17,174,338) | $0 | $3,335,467 ($126,978 - $17,174,338) |
ICER ($ per QALY gained) | Dominated ($12,650,148 - Dominated) | ||
Table 18: CADTH Reference Case Analysis: CRC (Versus FOLFIRI)
Output | Entrectinib | Comparator | Incremental |
|---|---|---|---|
Life-years | |||
Total life-years | 2.14 (1.32 - 2.63) | 2.27 (1.84 - 2.70) | -0.13 (-0.94 - 0.07) |
QALYs | |||
In PFS | 0.37 (0.06 - 0.81) | 0.65 (0.47 - 0.83) | -0.28 (-0.71 - 0.09) |
In progression | 1.06 (0.42 - 1.59) | 0.91 (0.61 - 1.23) | 0.15 (-0.43 - 0.55) |
Total QALYs | 1.43 (0.86 - 1.80) | 1.56 (1.24 - 1.88) | -0.13 (-0.66 - 0.05) |
Costs | |||
Drug costs | $49,050 ($9,243 - $102,155) | $19,593 ($15,828 - $23,427) | $29,457 (-$12,466 - $80,675) |
AE costs | $1,613 ($1,293 - $1,936) | $3,450 ($2,774 - $4,132) | -$1,836 (-$2,567 - -$1,101) |
Other health care costs | $29,745 ($17,614 - $37,414) | $31,524 ($24,958 - $38,402) | -$1,780 (-$12,835 - $990) |
Total costs | $80,408 ($32,705 - $135,441) | $54,567 ($46,834 - $62,323) | $25,841 (-$21,817 - $77,881) |
Results | |||
Percent of simulations entrectinib is clinically dominated | |||
ICER ($ per QALY gained) | Entrectinib is Dominated (Dominated - ICER of comparator vs. Entrectinib: $8,921/QALY gained) | ||
Testing cost | $3,025,599 ($129,760 - $15,932,605) | $0 | $3,025,599 ($129,760 - $15,932,605) |
ICER ($ per QALY gained) | Dominated ($11,813,787 - Dominated) | ||
Table 19: CADTH Reference Case Analysis: NSCLC Versus (Pemetrexed Plus Cisplatin)
Output | Entrectinib | Comparator | Incremental |
|---|---|---|---|
Life-years | |||
Total life-years | 2.10 (1.56 - 2.50) | 1.40 (1.26 - 1.53) | 0.70 (0.16 - 1.08) |
QALYs | |||
In PFS | 0.88 (0.38 - 1.18) | 0.50 (0.38 - 0.60) | 0.38 (-0.11 - 0.63) |
In progression | 0.62 (0.23 - 1.09) | 0.48 (0.37 - 0.59) | 0.14 (-0.24 - 0.61) |
Total QALYs | 1.50 (1.11 - 1.83) | 0.98 (0.83 - 1.12) | 0.52 (0.16 - 0.78) |
Costs | |||
Drug costs | $116,757 ($52,509 - $147,281) | $48,607 ($43,929 - $53,167) | $68,150 ($4,307 - $98,074) |
AE costs | $1,614 ($1,294 - $1,939) | $3,425 ($2,747 - $4,090) | -$1,812 (-$2,546 - -$1,056) |
Other health care costs | $30,363 ($22,162 - $37,243) | $20,185 ($17,483 - $22,970) | $10,178 ($2,407 - $15,834) |
Total costs | $148,733 ($83,120 - $181,012) | $72,217 ($66,804 - $77,747) | $76,516 ($10,699 - $108,267) |
Results | |||
Percent of simulations entrectinib is clinically dominated | |||
ICER ($ per QALY gained) | $149,881 ($29,890 - $405,246) | ||
Testing cost | $6,855,052 ($179,393 - $23,313,833) | $0 | $6,855,052 ($179,393 - $23,313,833) |
ICER ($ per QALY gained) | $2,054,979 ($453,034 - $60,135,130) | ||
Table 20: CADTH Reference Case Analysis: NSCLC Versus (Pemetrexed)
Output | Entrectinib | Comparator | Incremental |
|---|---|---|---|
Life-years | |||
Total life-years | 1.80 (1.36 - 2.16) | 0.99 (0.80 - 1.18) | 0.81 (0.39 - 1.12) |
QALYs | |||
In PFS | 0.78 (0.34 - 1.04) | 0.28 (0.20 - 0.36) | 0.50 (0.06 - 0.73) |
In progression | 0.51 (0.20 - 0.92) | 0.40 (0.27 - 0.54) | 0.11 (-0.20 - 0.51) |
Total QALYs | 1.29 (0.96 - 1.59) | 0.68 (0.54 - 0.83) | 0.61 (0.31 - 0.84) |
Costs | |||
Drug costs | $103,692 ($45,817 - $128,842) | $24,772 ($20,051 - $29,499) | $78,920 ($20,710 - $103,491) |
AE costs | $1,611 ($1,299 - $1,928) | $922 ($740 - $1,102) | $688 ($322 - $1,049) |
Other health care costs | $26,027 ($19,437 - $31,965) | $14,282 ($11,361 - $17,479) | $11,745 ($5,521 - $16,455) |
Total costs | $131,329 ($73,478 - $158,016) | $39,976 ($34,375 - $45,746) | $91,353 ($33,351 - $116,958) |
Results | |||
Percent of simulations entrectinib is clinically dominated | 0.0% | ||
ICER ($ per QALY gained) | $150,093 ($61,258 - $280,273) | ||
Testing cost | $4,449,406 ($173,724 - $21,140,865) | $0 | $4,449,406 ($173,724 - $21,140,865) |
ICER ($ per QALY gained) | $1,639,061 ($409,698 - $39,004,996) | ||
Table 21: CADTH Reference Case Analysis: NSCLC (Versus Cisplatin)
Output | Entrectinib | Comparator | Incremental |
|---|---|---|---|
Life-years | |||
Total life-years | 2.07 (1.47 - 2.67) | 1.36 (0.83 - 1.87) | 0.71 (0.14 - 1.09) |
QALYs | |||
In PFS | 0.84 (0.35 - 1.15) | 0.42 (0.24 - 0.61) | 0.42 (-0.07 - 0.68) |
In progression | 0.64 (0.20 - 1.15) | 0.53 (0.18 - 0.89) | 0.11 (-0.31 - 0.56) |
Total QALYs | 1.48 (1.05 - 1.90) | 0.95 (0.59 - 1.29) | 0.53 (0.14 - 0.81) |
Costs | |||
Drug costs | $111,846 ($48,239 - $143,847) | $3,419 ($2,093 - $4,743) | $108,428 ($44,828 - $139,964) |
AE costs | $1,614 ($1,296 - $1,920) | $ ($ - $) | $1,614 ($1,296 - $1,920) |
Other health care costs | $30,011 ($21,139 - $39,313) | $19,679 ($11,938 - $27,385) | $10,332 ($1,986 - $16,077) |
Total costs | $143,471 ($80,048 - $177,817) | $23,097 ($15,041 - $30,872) | $120,374 ($56,654 - $153,165) |
Results | |||
Percent of simulations entrectinib is clinically dominated | 0.6% | ||
ICER ($ per QALY gained) | $221,058 ($116,942 - $749,070) | ||
Testing cost | $5,139,639 ($174,101 - $25,576,677) | $0 | $5,139,639 ($174,101 - $25,576,677) |
ICER ($ per QALY gained) | $2,095,530 ($485,880 - $71,741,758) | ||
Table 22: CADTH Reference Case Analysis: NSCLC (Versus Docetaxel)
Output | Entrectinib | Comparator | Incremental |
|---|---|---|---|
Life-years | |||
Total life-years | 1.77 (1.31 - 2.21) | 0.94 (0.58 - 1.31) | 0.83 (0.39 - 1.12) |
QALYs | |||
In PFS | 0.78 (0.36 - 1.04) | 0.27 (0.16 - 0.39) | 0.51 (0.09 - 0.74) |
In progression | 0.49 (0.17 - 0.91) | 0.38 (0.14 - 0.63) | 0.11 (-0.20 - 0.48) |
Total QALYs | 1.27 (0.93 - 1.61) | 0.65 (0.42 - 0.90) | 0.62 (0.32 - 0.85) |
Costs | |||
Drug costs | $103,335 ($48,842 - $129,209) | $10,049 ($6,266 - $13,868) | $93,286 ($39,189 - $117,801) |
AE costs | $1,613 ($1,297 - $1,931) | $7,783 ($6,291 - $9,314) | -$6,170 (-$7,727 - -$4,654) |
Other health care costs | $25,548 ($18,694 - $32,630) | $13,586 ($8,320 - $19,046) | $11,963 ($5,658 - $16,538) |
Total costs | $130,497 ($76,241 - $157,986) | $31,418 ($24,709 - $38,160) | $99,079 ($44,800 - $125,001) |
Results | |||
Percent of simulations entrectinib is clinically dominated | 0.0% | ||
ICER ($ per QALY gained) | $159,441 ($81,945 - $290,603) | ||
Testing cost | $4,819,904 ($168,230 - $25,723,166) | $0 | $4,819,904 ($168,230 - $25,723,166) |
ICER ($ per QALY gained) | $1,686,586 ($415,344 - $45,091,142) | ||
Table 23: CADTH Reference Case Analysis: Pancreatic (Versus Gemcitabine + Nab-paclitaxel)
Output | Entrectinib | Comparator | Incremental |
|---|---|---|---|
Life-years | |||
Total life-years | 1.01 (0.91 - 1.11) | 1.01 (0.92 - 1.11) | 0.00 (-0.14 - 0.13) |
QALYs | |||
In PFS | 0.52 (0.39 - 0.62) | 0.52 (0.39 - 0.62) | 0.00 (-0.07 - 0.07) |
In progression | 0.22 (0.15 - 0.30) | 0.22 (0.15 - 0.30) | 0.00 (-0.10 - 0.10) |
Total QALYs | 0.74 (0.61 - 0.85) | 0.74 (0.61 - 0.86) | 0.00 (-0.09 - 0.09) |
Costs | |||
Drug costs | $69,134 ($62,619 - $75,694) | $40,821 ($36,972 - $44,771) | $28,314 ($20,591 - $35,996) |
AE costs | $1,613 ($1,298 - $1,925) | $ ($ - $) | $1,613 ($1,298 - $1,925) |
Other health care costs | $16,661 ($14,379 - $18,948) | $16,644 ($14,429 - $18,884) | $17 (-$2,241 - $2,190) |
Total costs | $87,408 ($80,505 - $94,218) | $57,465 ($53,038 - $61,970) | $29,943 ($21,903 - $38,093) |
Results | |||
Percent of simulations entrectinib is clinically dominated | 49.2% | ||
ICER ($ per QALY gained) | Dominated ($380,269 - Dominated) | ||
Testing cost | $2,189,416 ($98,828 - $9,742,173) | $0 | $2,189,416 ($98,828 - $9,742,173) |
ICER ($ per QALY gained) | Dominated ($3,161,075 - Dominated) | ||
Table 24: CADTH Reference Case Analysis: Soft Tissue Sarcoma (Versus Eribulin)
Output | Entrectinib | Comparator | Incremental |
|---|---|---|---|
Life-years | |||
Total life-years | 1.77 (1.34 - 2.22) | 1.59 (0.99 - 2.18) | 0.19 (-0.12 - 0.42) |
QALYs | |||
In PFS | 0.59 (0.43 - 0.74) | 0.25 (0.15 - 0.37) | 0.34 (0.21 - 0.45) |
In progression | 0.64 (0.35 - 0.95) | 0.80 (0.41 - 1.21) | -0.16 (-0.36 - 0.02) |
Total QALYs | 1.24 (0.93 - 1.56) | 1.06 (0.67 - 1.45) | 0.18 (-0.03 - 0.35) |
Costs | |||
Drug costs | $79,027 ($64,387 - $91,240) | $12,014 ($7,455 - $16,556) | $67,013 ($53,163 - $76,730) |
AE costs | $1,614 ($1,305 - $1,931) | $1,801 ($1,449 - $2,150) | -$186 (-$645 - $295) |
Other health care costs | $22,458 ($16,661 - $28,964) | $20,105 ($12,387 - $28,179) | $2,353 (-$1,463 - $5,346) |
Total costs | $103,099 ($87,272 - $116,967) | $33,919 ($25,005 - $43,187) | $69,180 ($54,841 - $79,597) |
Results | |||
Percent of simulations entrectinib is clinically dominated | 3.6% | ||
ICER ($ per QALY gained) | $369,135 ($207,914 - Dominated) | ||
Testing cost | $2,203,529 ($186,620 - $9,827,651) | $0 | $2,203,529 ($186,620 - $9,827,651) |
ICER ($ per QALY gained) | $5,317,866 ($1,145,467 - Dominated) | ||
Table 25: CADTH Reference Case Analysis: Thyroid (Versus Lenvatinib)
Output | Entrectinib | Comparator | Incremental |
|---|---|---|---|
Life-years | |||
Total life-years | 3.05 (1.98 - 3.76) | 3.42 (2.80 - 4.04) | -0.37 (-1.43 - 0.15) |
QALYs | |||
In PFS | 1.33 (0.79 - 1.77) | 1.66 (1.18 - 2.10) | -0.33 (-0.83 - -0.01) |
In progression | 0.84 (0.18 - 1.42) | 0.81 (0.33 - 1.29) | 0.04 (-0.60 - 0.56) |
Total QALYs | 2.17 (1.42 - 2.71) | 2.47 (1.93 - 2.99) | -0.30 (-0.99 - 0.06) |
Costs | |||
Drug costs | $178,012 ($114,069 - $225,154) | $57,266 ($46,720 - $67,882) | $120,746 ($58,746 - $161,854) |
AE costs | $1,616 ($1,305 - $1,930) | $6,660 ($5,318 - $8,002) | -$5,044 (-$6,413 - -$3,685) |
Other health care costs | $22,750 ($14,552 - $28,678) | $25,523 ($20,248 - $30,920) | -$2,774 (-$10,670 - $1,101) |
Total costs | $202,378 ($137,547 - $250,360) | $89,450 ($77,353 - $101,921) | $112,928 ($48,493 - $154,724) |
Results | |||
Percent of simulations entrectinib is clinically dominated | 93.5% | ||
ICER ($ per QALY gained) | Dominated ($2,158,604 - Dominated) | ||
Testing cost | $2,377,651 ($15,209 - $6,034,653) | $0 | $2,377,651 ($15,209 - $6,034,653) |
ICER ($ per QALY gained) | Dominated ($4,755,807 - Dominated) | ||
Table 26: CADTH Reference Case Analysis: Thyroid (Versus Sorafenib)
Output | Entrectinib | Comparator | Incremental |
|---|---|---|---|
Life-years | |||
Total life-years | 2.51 (1.66 - 3.08) | 2.60 (2.11 - 3.08) | -0.09 (-0.93 - 0.33) |
QALYs | |||
In PFS | 1.02 (0.64 - 1.34) | 1.00 (0.71 - 1.28) | 0.02 (-0.34 - 0.24) |
In progression | 0.76 (0.24 - 1.20) | 0.83 (0.48 - 1.19) | -0.07 (-0.57 - 0.32) |
Total QALYs | 1.78 (1.20 - 2.22) | 1.84 (1.46 - 2.21) | -0.05 (-0.60 - 0.22) |
Costs | |||
Drug costs | $136,251 ($91,371 - $166,949) | $86,137 ($69,689 - $102,318) | $50,114 ($4,239 - $76,379) |
AE costs | $1,615 ($1,295 - $1,935) | $4,051 ($3,280 - $4,843) | -$2,437 (-$3,282 - -$1,599) |
Other health care costs | $18,721 ($12,246 - $23,401) | $19,384 ($15,405 - $23,445) | -$663 (-$6,892 - $2,469) |
Total costs | $156,587 ($111,189 - $188,177) | $109,573 ($92,306 - $126,420) | $47,014 ($146 - $73,859) |
Results | |||
Percent of simulations entrectinib is clinically dominated | 53.0% | ||
ICER ($ per QALY gained) | Dominated ($167,780 - Dominated) | ||
Testing cost | $1,670,278 ($15,094 - $5,727,073) | $0 | $1,670,278 ($15,094 - $5,727,073) |
ICER ($ per QALY gained) | Dominated ($470,328 - Dominated) | ||
Scenario Analyses
Table 27: CADTH Price Reduction Analyses: Second-Line Comparators
Tumour site | Weekly cost required to achieve ICER < $50,000 per QALY gained | |
|---|---|---|
Second-line comparators | Patients with known mutation (% price reduction) | Including testing to identify eligible patients (% price reduction) |
Breast—secretory (vs. carboplatin) | $432 (78%) | $ 392 (80%) |
Breast—secretory (vs. eribulin) | $548 (73%) | $ 510 (75%) |
Breast—non-secretory (vs. carboplatin) | $431 (78%) | Cannot be achieved |
Breast—non-secretory (vs. eribulin) | $548 (73%) | Cannot be achieved |
Colorectal cancer (vs. pembrolizumab) | No price reduction required | Cannot be achieved |
Colorectal cancer (vs. bevacizumab + FOLFOX) | No price reduction required | Cannot be achieved |
Colorectal cancer (vs. FOLFOX) | $1,015 (49%) | Cannot be achieved |
Colorectal cancer (vs. FOLFIRI) | $674 (66%) | Cannot be achieved |
NSCLC (vs. pemetrexed and cisplatin) | $1,133 (43%) | Cannot be achieved |
NSCLC (vs. pemetrexed) | $828 (59%) | Cannot be achieved |
NSCLC (vs. cisplatin) | $323 (84%) | Cannot be achieved |
NSCLC (vs. docetaxel) | $683 (66%) | Cannot be achieved |
Pancreatic (vs. gemcitabine + nab-paclitaxel) | $1,135 (43%) | Cannot be achieved |
Soft tissue sarcoma (vs. eribulin) | $479 (76%) | Cannot be achieved |
Thyroid cancer (vs. lenvatinib) | $565 (72%) | Cannot be achieved |
Thyroid cancer (vs. sorafenib) | $1,271 (37%) | Cannot be achieved |
Pooled excluding “Other” | $913 (54%) | Cannot be achieved |
Pooled including “Other” | Cannot be achieved | Cannot be achieved |
ICER = incremental cost-effectiveness ratio; NSCLC = non–small cell lung cancer vs. = versus.
It was discussed that entrectinib may replace first-line alternatives if a patient’s NTRK status is known at this point in time. Given this may contradict the Health Canada indication, which specifies no satisfactory treatment options remaining, as well as the sponsor’s inappropriate model structure assuming no subsequent therapies given after treatment failure, CADTH reserved the analysis of entrectinib versus first-line comparators as a scenario analysis. Two analyses are presented. One which maintains the sponsor’s assumptions of no subsequent therapy costs and one which attempts to include these.
Table 28: Summary of the CADTH Scenario Reanalysis for First-Line Comparators in Patients Known and Not Known to Have an NTRK Fusion Cancer
Tumour site (vs. comparator) | Incremental cost (95% CI) | Incremental cost of testing (95% CI) | Incremental QALY (95% CI) | Probability that entrectinib is clinically dominated | ICER (95% CI) | ICER including testing (95% CI) |
|---|---|---|---|---|---|---|
Breast—secretory (vs. paclitaxel) | $54,624 (-$23,546 - $87,212) | $1,501 ($1,400 - $2,221) | 0.03 (-0.72 - 0.31) | 29.3% | $1,584,600 ($16,271 - Dominated) | $1,628,150 ($36,530 - Dominated) |
Breast—secretory (vs. docetaxel) | $71,752 (-$23,617 - $115,165) | $1,494 ($1,400 - $2,238) | -0.11 (-1.02 - 0.22) | 55.4% | Dominated ($239,206 - Dominated) | Dominated ($351,369 - Dominated) |
Breast—non-secretory (vs. paclitaxel) | $54,624 (-$23,546 - $87,212) | $8,450,567 ($290,276 - $45,567,192) | 0.03 (-0.72 - 0.31) | 29.3% | $1,584,600 ($16,271 - Dominated) | $246,730,690 ($2,063,627 - Dominated) |
Breast—non-secretory (vs. docetaxel) | $71,752 (-$23,617 - $115,165) | $8,450,567 ($290,276 - $45,567,192) | -0.11 (-1.02 - 0.22) | 55.4% | Dominated ($239,206 - Dominated) | Dominated ($3,644,916 - Dominated) |
Colorectal cancer (vs. pembrolizumab) | -$116,039 (-$235,482 – -$10,394) | $3,279,420 ($127,563 - $14,885,755) | -0.44 (-1.64 - -0.07) | 99.5% | ICER of Comparator vs. Entrectinib: $268,337 ($44,347 - $1,174,425) | Dominated (Dominated - ICER of Comparator vs. Entrectinib: $4,884) |
Colorectal cancer (vs. FOLFOX) | $21,331 (-$34,950 - $82,912) | $3,335,467 ($126,978 - $17,174,338) | -0.15 (-0.70 - 0.04) | 85.2% | Dominated (Dominated - ICER of comparator vs. Entrectinib: $5,122/QALY gained) | Dominated ($12,650,148 - Dominated) |
Colorectal cancer (vs. FOLFIRI) | $25,841 (-$21,817 - $77,881) | $3,025,599 ($129,760 - $15,932,605) | -0.13 (-0.66 - 0.05) | 83.1% | Dominated (Dominated - ICER of comparator vs. Entrectinib: $8,921/QALY gained) | Dominated ($11,813,787 - Dominated) |
MASC (vs. sunitinib) | $102,898 ($46,920 - $144,031) | $1,643 ($1,400 - $3,054) | 1.10 (0.74 - 1.35) | 0.0% | $94,645 ($45,062 - $146,709) | $96,021 ($46,633 - $149,256) |
MASC (vs. gefitinib) | $193,153 ($149,970 - $227,646) | $1,631 ($1,400 - $2,991) | 1.07 (0.65 - 1.34) | 0.0% | $177,435 ($135,447 - $293,531) | $178,867 ($136,630 - $296,134) |
NSCLC (vs. pembrolizumab + pemetrexed and cisplatin) | -$57,514 (-$141,016 – -$12,507) | $4,359,188 ($171,663 - $23,085,702) | 0.18 (-0.48 - 0.64) | 25.3% | Cost-saving (Cost-saving - ICER of comparator vs. Entrectinib: $106,331/QALY gained) | $7,084,215 ($362,949 - Dominated) |
NSCLC (vs. Pembrolizumab) | $68,604 ($6,847 - $97,231) | $4,913,786 ($171,789 - $24,929,813) | 0.51 (0.13 - 0.80) | 0.5% | $134,126 ($15,108 - $413,302) | $2,054,815 ($419,695 - $82,964,750) |
Neuroendocrine (vs. octreotide) | $171,293 ($148,315 - $194,189) | $1,034,081 ($523,607 - $1,724,260) | 0.00 (-0.48 - 0.47) | 49.0% | Dominated ($388,412 - Dominated) | Dominated ($1,444,389 - Dominated) |
Pancreatic (vs. FOLFIRINOX) | $47,546 ($30,580 - $64,324) | $1,781,641 ($95,801 - $9,224,945) | 0.00 (-0.22 - 0.23) | 50.3% | Dominated ($229,064 - Dominated) | Dominated ($1,328,932 - Dominated) |
Soft tissue sarcoma (vs. doxorubicin) | $85,397 ($67,694 - $98,083) | $1,870,088 ($193,368 - $9,436,777) | 0.19 (0.04 - 0.29) | 0.7% | $439,191 ($294,334 - $1,944,047) | $5,049,652 ($1,312,475 - $98,263,742) |
Thyroid cancer (vs. lenvatinib) | $112,928 ($48,493 - $154,724) | $2,377,651 ($15,209 - $6,034,653) | -0.30 (-0.99 - 0.06) | 93.5% | Dominated ($2,158,604 - Dominated) | Dominated ($4,755,807 - Dominated) |
Thyroid cancer (vs. sorafenib) | $47,014 ($146 - $73,859) | $1,670,278 ($15,094 - $5,727,073) | -0.05 (-0.60 - 0.22) | 53.0% | Dominated ($167,780 - Dominated) | Dominated ($470,328 - Dominated) |
Other* | 183,444 | 562,085 | 0 | 50% | Dominated | Dominated |
Pooled excluding “Other” | $52,340 ($32,265 - $67,474) | $2,609,383 ($298,217 - $12,124,609) | 0.15 (-0.03 - 0.29) | 4.1% | $328,657 ($179,312 - Dominated) | $7,214,686 ($1,788,941 - Dominated) |
Pooled including “Other” | $87,702 ($73,042 - $98,754) | $2,057,174 ($369,389 - $9,005,900) | 0.11 (-0.02 - 0.21) | NR | $742,888 ($433,236 - Dominated) | $9,209,215 ($2,943,511 - Dominated) |
ICER = incremental cost-effectiveness ratio; MASC = mammary analogue secretory carcinoma; NR = not reported; NTRK = neurotrophic tyrosine receptor kinase; NSCLC = non–small cell lung cancer; QALY = quality-adjusted life-year; vs. = versus.
Note: ICERs with testing costs were calculated by CADTH by adding average testing costs to the incremental costs and recalculating the ICER.
*Other includes neuroblastoma, head and neck cancer, endometrial cancer, ovarian cancer, cholangiocarcinoma, and cancer of unknown primary. For each of these cancer types, incremental cost was assumed to be the drug cost estimated in the sponsor’s analysis, the incremental QALY benefit was assumed to be 0 because no clinical evidence was provided for these indications. Their weight in the pooled analysis was determined by their relative prevalence. CADTH notes there are other cancer types not identified in the trial which are not included above. Therefore, the ICER in the pooled results including other may underestimate the ICER.
Table 29: including Second-Line Therapy Costs
Tumour site (vs. comparator) | Incremental costs including second-line therapy | ICER for known NTRK fusion cancer ($/ QALY) for entrectinib vs. comparator |
|---|---|---|
Breast—secretory (vs. paclitaxel) | $89,315 | 2,590,977 |
Breast—secretory (vs. docetaxel) | $98,927 | Dominated |
Breast—non-secretory (vs. paclitaxel) | $89,315 | 2,590,977 |
Breast—non-secretory (vs. docetaxel) | $98,927 | Dominated |
Colorectal cancer (vs. pembrolizumab) | $91,390 | Dominated |
Colorectal cancer (vs. FOLFOX) | $52,688 | Dominated |
Colorectal cancer (vs. FOLFIRI) | $45,434 | Dominated |
MASC (vs. sunitinib) | $214,963 | 195,302 |
MASC (vs. gefitinib) | $207,035 | 193,752 |
NSCLC (vs. pembrolizumab + pemetrexed and cisplatin) | $135,139 | 760,833 |
NSCLC (vs. Pembrolizumab) | $121,747 | 236,655 |
Neuroendocrine (vs. octreotide) | $220,942 | Dominated |
Pancreatic (vs. FOLFIRINOX) | $67,952 | Dominated |
Soft tissue sarcoma (vs. doxorubicin) | $96,379 | 513,364 |
Thyroid cancer (vs. lenvatinib) | $170,194 | Dominated |
Thyroid cancer (vs. sorafenib) | $133,151 | Dominated |
ICER = incremental cost-effectiveness ratio; NSCLC = non–small cell lung cancer vs. = versus
Table 30: CADTH Price Reduction Analyses
Tumour site | Weekly cost required to achieve ICER < $50,000 per QALY gained | ||
|---|---|---|---|
First-line comparators | Patients with known mutation (% price reduction) | Patients with known mutation including subsequent therapy costsb (% price reduction) | Including testing costs to identify eligible patients (% price reduction)a |
Breast—secretory (vs. paclitaxel) | $793 (60%) | < $2 (>99%) | $ 759 (62%) |
Breast—secretory (vs. docetaxel) | $523 (74%) | < $2 (>99%) | $ 494 (75%) |
Breast—non-secretory (vs. paclitaxel) | $793 (60%) | < $2 (>99%) | Cannot be achieved |
Breast—non-secretory (vs. docetaxel) | $523 (74%) | < $2 (>99%) | Cannot be achieved |
Colorectal cancer (vs. pembrolizumab) | No price reduction required | Cannot be achieved | Cannot be achieved |
Colorectal cancer (vs. FOLFOX) | $1,015 (49%) | Cannot be achieved | Cannot be achieved |
Colorectal cancer (vs. FOLFIRI) | $674 (66%) | Cannot be achieved | Cannot be achieved |
MASC (vs. sunitinib) | $1,552 (22%) | $497 (75%) | $1,536 (23%) |
MASC (vs. gefitinib) | $511 (74%) | $363 (82%) | $ 493 (75%) |
NSCLC (vs. pembrolizumab + pemetrexed and cisplatin) | No price reduction required | $136 (93%) | Cannot be achieved |
NSCLC (vs. pembrolizumab) | $1,224 (39%) | $259 (87%) | Cannot be achieved |
Neuroendocrine (vs. octreotide) | $439 (78%) | Cannot be achieved | Cannot be achieved |
Pancreatic (vs. FOLFIRINOX) | $815 59%) | $305 (85%) | Cannot be achieved |
Soft tissue sarcoma (vs. doxorubicin) | $346 (83%) | $106 (95%) | Cannot be achieved |
Thyroid cancer (vs. lenvatinib) | $565 (72%) | Cannot be achieved | Cannot be achieved |
Thyroid cancer (vs. sorafenib) | $1,271 (37%) | $6 (>99%) | Cannot be achieved |
Pooled excluding “Other” | $954 (52%) | Cannot be achieved | Cannot be achieved |
Pooled including “Other” | $356 (82%) | Cannot be achieved | Cannot be achieved |
ICER = incremental cost-effectiveness ratio; NSCLC = non–small cell lung cancer vs. = versus
aif subsequent therapy costs are included then there is no price reduction which could achieve an ICER below $50,000 per QALY for any tumour site
binclusion of subsequent therapy costs as estimated in Table 29
Appendix 5: Submitted Budget Impact Analysis and CADTH Appraisal
Note that this appendix has not been copy-edited.
Table 31: Summary of Key Take-Aways
Key Take-aways of the Budget Impact Analysis |
|---|
|
Summary of Sponsor’s Budget Impact Analysis
In the submitted budget impact analysis (BIA), the sponsor assessed the introduction of entrectinib for the treatment of adult patients with unresectable locally advanced or metastatic extracranial solid tumours, including brain metastases, that have an NTRK gene fusion without a known acquired resistance mutation and with no satisfactory treatment options. The BIA was undertaken from the perspective of a Canadian public payer over a 3-year time horizon (2022 to 2024) using an epidemiological approach. The sponsor included drug acquisition costs and NTRK gene fusion testing costs. Data from the model were obtained from: the Canadian Cancer Society,29,30 Key inputs to the BIA are documented in Table 32.
Key assumptions include:
NSCLC patients are already being tested for the NTRK gene fusion and will not accrue additional testing costs due to entrectinib.
Costs for entrectinib are fully additive; the model does not consider displacement of a particular set of therapies.
Only patients with CRC, MASC, thyroid cancer, NSCLC, STS, and breast cancer will be treated with entrectinib.
Clinical practice in Canada will involve IHC screening followed by confirmatory NGS testing.
Patients will receive entrectinib for an average of 13.8 months.
Table 32: Sponsor’s Estimation of the Size of the Eligible Population
Epidemiology Inputs | CRC | MASC | Thyroid | NSCLC | STS | Breast |
|---|---|---|---|---|---|---|
Incident patients | ||||||
Incident patients in Year 1 / Year 2 / Year 3 | 22,507 / 23,565 / 24,113a | 396 / 428 / 444b | 7,987 / 9,283 / 10,007a | 24,843 / 25,917 / 26,917c | 1,138 / 1,248 / 1,306d | 24,258 / 26,584 / 27,829a |
Percentage of patients with stage III cancer at diagnosis | 29.1%a | 10.9%e | 14.0%a | 16.0%a | 20.0%d | 12.0%a |
Percentage of patients with stage III locally advanced | 85.0%a | 5.0%e | 12.5%a | 30.0%a | 19.0%d | 0%a |
Percentage of patients with stage IV cancer at diagnosis | 20.0%a | 1.8%e | 9.0%a | 49.0%a | 15.0%d | 5.0%a |
Percentage of patients receiving systemic treatment | 75.0%b | 90.0%f | 90.0%f | 55.0%g | 90.0%f | 90.0%f |
Total stage III locally advanced/stage IV receiving systemic treatment | 7,551 / 7,906 / 8,090 | 8 / 9 / 9 | 773 / 898 / 968 | 7,351 / 7,669 / 7,833 | 193 / 211 / 221 | 1,092 / 1,196 / 1,252 |
Plus, prevalent patients (year 1 only) | ||||||
Incident patients stage III locally advance and Stage IV in base year receiving SOC therapy | 7,380 | 8 | 717 | 7,197 | 184 | 1,043 |
Percentage of SOC patients progressing to another therapy | 75%f | |||||
Total patients stage III locally advanced/Stage IV patients progressing to another therapy from base year and considered for NTRK therapy | 5,535 | 6 | 538 | 5,398 | 138 | 782 |
Incident and prevalent patients | ||||||
Percentage of patients considered for NTRK therapy | 30.0%h | 100%f | 100%f | 100%i | 100%f | 18.0%j |
Percentage of patients already NTRK tested via public funding ($0) | 0% | 0% | 0% | 100% | 0% | 0% |
Percentage of patients NTRK tested (IHC screening test, $80) | 100% | 100% | 100% | 0% | 100% | 100% |
Percentage of patients NTRK tested (NGS confirmation due to positive IHC, $1,400) | 9.0%k | 90.0%k | 9.0%k | 0% | 9.0%k | 9.0%k |
Percentage of patients NTRK true positive rate | 0.3%l | 90.0%m | 2.3%n | 0.2%o | 0.2%p | 0.1%n |
Percentage of patients publicly reimbursed | 69%q | |||||
NTRK positive patients eligible for publicly funded entrectinib (Year 1/Year 2/Year 3) | 8 / 5 / 5 | 9 / 6 / 6 | 21 / 14 / 14 | 19 / 12 / 12 | 1 / 0 / 0 | 0 / 0 / 0 |
Cost of treatment (per patient per month) | ||||||
Entrectinib (Rozlytrek) | $8,705 | |||||
CRC = colorectal cancer; IHC = immunohistochemistry; MASC = mammary analogue secretory carcinoma; NGS = next-generation sequencing; NSCLC = non–small cell lung cancer; NTRK = neurotrophic tyrosine receptor kinase; STS = soft tissue sarcoma.
aDerived from Canadian Cancer Statistics, 2018, including supplementary data.29,30
bCanadian Consensus for Biomarker Testing and Treatment of TRK Fusion Cancer in Adults (2021).31
cDerived from Brenner et al. 2020.32
dDerived from Canadian Cancer Society 2016 incidence.33
eAnderson 2019.34
fSponsor’s clinician-informed assumption.
gBrule 2016.35
hCited as Bylsma 2020,36 input derivation unclear.
iGainor 2013.37
jCited as SEER US 2010 to 2014, data not provided.
kCited as Feng 2018,14 input derivation unclear.
lRosen 2020.38
mCited as Skalova 2016,39 input derivation unclear.
nCited as Zehir 2017,40 input derivation unclear.
oFarago 2018.41
pSponsor’s unpublished epidemiology research, not provided.
qFrom Canadian Institute for Health Information National Health Expenditure Trends 2018, overall proportion of health spending which is publicly funded.42
Summary of the Sponsor’s BIA Results
Results of the sponsor’s base case suggested that the reimbursement of entrectinib for adult patients with locally advanced or metastatic solid tumours harbouring an NTRK gene fusion would be $7,141,152 in Year 1, $5,356,313 in Year 2, and $5,220,609 in Year 3, for a 3-year budget impact of $17,718,075. This total included $2,270,320 in NTRK testing costs.
CADTH Appraisal of the Sponsor’s BIA
CADTH identified several key limitations to the sponsor’s analysis that have notable implications on the results of the BIA:
Population estimate does not include the entire indicated population: The sponsor’s model includes prevalence and incidence estimates for specific tumour types including CRC, MASC, thyroid cancers, NSCLC, STSs, and breast cancer. However, entrectinib is a tumour-agnostic treatment indicated for adult patients with unresectable, locally advance, or metastatic extracranial solid tumours including brain metastases that have an NTRK gene fusion without a known acquired resistance mutation and with no satisfactory treatment options. As such, treatment with entrectinib is unlikely to be limited to the 6 included cancer types. In addition to the tumour types included in the sponsor’s BIA, the clinical trial also included neuroendocrine cancer (n=5), pancreatic cancer (n=4), cancer of unknown primary (n=3), cholangiocarcinoma / non-CRC GI cancer not otherwise specified (n=2), ovarian cancer (n=1), endometrial cancer (n=1), neuroblastoma (n=1) and head and neck (n=1). The broader clinical literature indicates a large number of cancer sites affected with NTRK gene fusions.9
No comparator treatments are displaced: The sponsor assumed, based on clinician input, that entrectinib would not replace a specific line of therapy but would instead be an extra line of therapy, and therefore the sponsor considered the cost of entrectinib to be fully additive (i.e., no other therapies would be displaced).43 This is a conservative approach and may not represent clinical reality for all tumour types (see Stakeholder Input section) nor the displacement of some comparator costs past the 3-year time horizon, however the magnitude to which the resulting budget impact is overestimated is unknown. Additionally, larotrectinib (Vitrakvi) received a positive funding recommendation from the CADTH pan-Canadian Oncology Drug Review Expert Review Committee for an overlapping but not identical patient population: the treatment of adult and pediatric patient with locally advanced or metastatic solid tumours who have a NTRK gene fusion without known acquired resistance mutation or where surgical resection is likely to result in severe morbidity and have no satisfactory treatment options, with conditions.44 At the time of this review, larotrectinib was undergoing funding negotiations with pCPA.45 Should larotrectinib be funded for such patients, the budgetary impact of entrectinib would be smaller than estimated as it would gain less market share within its indication and is less expensive than larotrectinib at their submitted prices when both are used at their unreduced recommended dosing.20,21 As the testing costs associated with the establishment of eligibility for entrectinib are also required to establish eligibility for larotrectinib, incremental costs associated with testing prior to entrectinib treatment would also be reduced if larotrectinib is already funded.
Due to the variety of treatments that might be displaced within the 3-year time horizon, uncertainty in potential market share between larotrectinib and entrectinib, and uncertainty in incremental testing costs should NTRK testing become standard practice prior to entrectinib funding over the time horizon, CADTH was unable to account for this limitation. Should larotrectinib be funded for a similar patient population to entrectinib and should NTRK testing already be funded in many or all tumour types, the budgetary impact of reimbursing entrectinib will be substantially less than estimated.
Diagnostic testing strategy does not represent Canadian clinical practice: According to the clinical experts consulted by CADTH, IHC for the detection of NTRK fusions is in development and not yet available in all provinces. Until IHC testing has been reliably validated for clinical use, it is not appropriate to consider IHC screening in the base case. Given currently available technologies, NTRK fusion testing would occur via NGS. The clinical experts consulted by CADTH also preferred the use of NGS because of the potential ability to screen for multiple mutations using a single sample from the patient; as the NTRK fusion has a low probability of being identified for most patients, concerns were raised about IHC “using up” patients’ available sample preventing the exploration of other treatment options. See CADTH’s Appraisal of the Sponsor’s Economic Analysis for further detail.
CADTH used NGS testing as the base-case testing strategy for identifying patients with NTRK gene fusion cancers. As in the sponsor’s submission, it was assumed the NSCLC testing for NTRK fusions is already funded and thus was not included.
Proportion of patients eligible for public funding underestimated: The sponsor assumed that 69% of patients eligible for entrectinib would be funded by a public plan based on the overall proportion of health care expenditures paid by public plans as reported by the Canadian Institute for Health Information (CIHI) in 2018.42 However, not only was this estimate updated by CIHI to 75% in 2021,46 but the overall proportion of all health care expenditure that is publicly funded is not representative of the proportion of cancer therapies which are publicly funded. Several provinces fund all cancer treatments, while others fund oral cancer therapies as they do other oral medications. Previous CADTH reviews, including that of larotrectinib,22 have estimated that 80% of patients in Canada excluding Quebec are eligible for public funding of oral cancer therapies.
CADTH assumed that 80% of patients would be reimbursed by a public plan in reanalyses.
CADTH Reanalyses of the BIA
CADTH undertook a series of reanalyses, including a complete reanalysis of the estimated budgetary impact using inputs from the CADTH review of larotrectinib where available, updated with 2021 incidence data from the Canadian Cancer Society27 and with 2021 NTRK fusion prevalence data from Westphalen et al. 2021,9 see Table 34. The sponsor’s staging, testing eligibility and NTRK fusion prevalence estimates were used for tumour types included in the submitted analysis (i.e., breast, colorectal, MASC, NSCLC, STS, and thyroid).
The results of the CADTH step-wise reanalysis are presented in summary format in Table 35 where reanalyses 2 and 3 were conducted within the sponsor’s submitted model to show their individual impacts, while reanalyses 1 and the CADTH base case were conducted within CADTH’s reworked model. A more detailed breakdown of the sponsor’s and CADTH’s base-case results is available in Table 36.
Based on CADTH reanalysis, the estimated budgetary impact of funding entrectinib, assuming no displacement of other treatment options, is $69,746,533 in Year 1, $42,266,837 in Year 2, and $42,005,060 in Year 3, for a 3-year budget impact of $154,018,431. When including only drug costs (i.e., a drug plan perspective), the budgetary impact of reimbursing entrectinib would be $13,444,089 in Year 1, $9,080,920 in Year 2, and $8,435,525 in Year 3, for a 3-year budget impact of $30,960,534. Results of the CADTH base case by tumour type are reported in Table 37.
As assumed by the sponsor, the CADTH base case assumed that 75% of patients who would have been eligible for entrectinib in the base year would be eligible as the prevalent population in Year 1. A scenario analysis was conducted excluding these patients.
Table 33: CADTH Revisions to the Submitted Budget Impact Analysis
Stepped analysis | Sponsor’s value or assumption | CADTH value or assumption |
|---|---|---|
Corrections to sponsor’s base case | ||
None | — | — |
Changes to derive the CADTH base case | ||
1. Inclusion of other NTRK-relevant tumour types | Breast, colorectal, MASC, NSCLC, STS, Thyroid | Appendix, bone sarcoma, breast, cancer of unknown primary, cholangiocarcinoma, CNS/glioma, colorectal, endometrial, head and neck, hepatic, MASC, melanoma, neuroendocrine, NSCLC, ovarian, pancreas, prostate, STS, thyroid |
2. Testing to determine NTRK eligibility | IHC screening followed by confirmatory NGS | NGS alone |
3. Proportion of patients who are publicly reimbursed | 69% | 80% |
CADTH base case | 1 + 2 + 3 | |
CNS = central nervous system; IHC = immunohistochemistry; MASC = mammary analogue secretory carcinoma; NGS = next-generation sequencing; NSCLC = non–small cell lung cancer; NTRK = neurotrophic tyrosine receptor kinase; STS = soft tissue sarcoma.
Table 34: Summary of Key Model Parameters to Derive Population Size in CADTH Reanalysis
Tumour site | Canadian patients in base yeara | Stage III/IV | % Eligible for testing | NTRK fusion prevalence | Eligibleb and NTRK positive in base year |
|---|---|---|---|---|---|
Appendix | 213c | 100%c | 90%c | 0.52%c | 1.0 |
Bone sarcoma | 188c | 100%c | 90%c | 0.32%d | 0.5 |
Breast | 23,173e | 4.5%e | 18%e | 0.13%e | 0.2 |
Cancer of unknown primary | 4,276c | 100%c | 90%c | 0.26%c | 10.0 |
Cholangiocarcinoma | 104c | 100%c | 90%c | 0.20%d | 0.2 |
CNS / Glioma | 2,408fg | 56.0%c | 90%c | 0.34%d | 4.1 |
Colorectal | 21,996e | 33.6%e | 30%e | 0.30%e | 6.6 |
Endometrial | 5,593fg | 17.7%f | 90%h | 0.19%d | 1.7 |
Head and Neck | 5,748f | 50.0%i | 90%h | 0.19%d | 4.9 |
Hepatic | 2,563f | 37.7%c | 90%c | 0.06%d | 0.5 |
MASC | 382e | 2.1%e | 100%e | 90%e | 7.3 |
Melanoma | 6,758f | 14.6%c | 90%c | 0.24%d | 2.1 |
Neuroendocrine | 1,743j | 13.0%j | 90%h | 0.15%d | 0.3 |
NSCLC | 24,323e | 29.6%e | 100%e | 0.22%e | 15.8 |
Ovarian | 2,330f | 62.8%f | 90%h | 0.31%d | 4.1 |
Pancreas | 5,205f | 68.4%c | 90%c | 0.17%d | 5.4 |
Prostate | 18,643f | 22.4%c | 90%c | 0.22%d | 8.3 |
STS | 1,088e | 16.9%e | 100%e | 0.23%e | 0.4 |
Thyroid | 7,409e | 9.7%e | 100%e | 2.30%e | 16.5 |
CNS = central nervous system; MASC = mammary analogue secretory carcinoma; NSCLC = non–small cell lung cancer; NTRK = neurotrophic tyrosine receptor kinase; STS = soft tissue sarcoma.
a2021, excluding Quebec.
bEligibility implies eligible for entrectinib and does not imply public reimbursement.
cAs in CADTH Vitrakvi reanalyses; number of patients inflated to 2021 estimate by assuming the same growth as the general population outside of Quebec (M1 projection).22,47
dWestphalen 2021.9
eSponsor’s submitted estimate.43
fCanadian Cancer Statistics; 2021 for incidence,27 2018 supplementary material for staging.30
gDerived by considering a Cancer.net estimate that 90% of uterine cancers occur in the endometrium.48
hAssumption.
iKassirian 2020.49
jCanadian Neuroendocrine Tumour Society, 2016.50
Table 35: Summary of the CADTH Reanalyses of the BIA
Stepped analysis | Three-year total |
|---|---|
Submitted base case | $17,718,075 |
Submitted base case (drug costs only) | $15,447,755 |
CADTH reanalysis 1: Other NTRK-relevant tumour types included | $26,703,460 |
CADTH reanalysis 2: Only NGS testing used | $30,618,271 |
CADTH reanalysis 3: 80% of patients publicly funded | $20,180,760 |
CADTH base case (1 + 2 + 3) | $154,018,431 |
CADTH base case (1 + 2 + 3, drug costs only) | $30,960,534 |
BIA = budget impact analysis; NGS = next-generation sequencing; NTRK = neurotrophic tyrosine receptor kinase.
Table 36: Detailed Breakdown of the CADTH Reanalyses of the BIA
Stepped analysis | Scenario | Year 0 (current situation) | Year 1 | Year 2 | Year 3 | Three-year total |
|---|---|---|---|---|---|---|
Submitted base case | Drug Costs | $0 | $6,063,864 | $4,767,763 | $4,616,128 | $15,447,755 |
Testing Costs | $0 | $1,077,289 | $588,550 | $604,481 | $2,270,320 | |
Budget impact | $0 | $7,141,152 | $5,356,313 | $5,220,609 | $17,718,075 | |
CADTH base case | Drug Costs | $0 | $13,444,089 | $9,080,920 | $8,435,525 | $30,960,534 |
Testing Costs | $0 | $56,302,445 | $33,185,917 | $33,569,536 | $123,057,897 | |
Budget impact | $0 | $69,746,533 | $42,266,837 | $42,005,060 | $154,018,431 | |
CADTH Scenario A: Prevalent patients excluded | Drug Costs | $0 | $7,796,368 | $8,203,829 | $8,435,525 | $24,435,721 |
Testing Costs | $0 | $32,627,367 | $33,185,917 | $33,569,536 | $99,382,820 | |
Budget impact | $0 | $40,423,735 | $41,389,746 | $42,005,060 | $123,818,541 |
BIA = budget impact analysis.
Table 37: CADTH Base-Case Results by Tumour Type
Stepped analysis | Scenario | Year 0 (current situation) | Year 1 | Year 2 | Year 3 | Three-year total |
|---|---|---|---|---|---|---|
Appendix | Drug Costs | $0 | $146,496 | $94,616 | $86,152 | $327,264 |
Testing Costs | $0 | $471,974 | $274,327 | $277,558 | $1,023,859 | |
Budget impact | $0 | $618,470 | $368,943 | $363,710 | $1,351,123 | |
Bone sarcoma | Drug Costs | $0 | $79,922 | $51,618 | $47,001 | $178,541 |
Testing Costs | $0 | $418,419 | $243,199 | $246,063 | $907,680 | |
Budget impact | $0 | $498,340 | $294,817 | $293,064 | $1,086,221 | |
Breast | Drug Costs | $0 | $36,639 | $25,794 | $24,489 | $86,922 |
Testing Costs | $0 | $472,172 | $301,463 | $315,581 | $1,089,216 | |
Budget impact | $0 | $508,811 | $327,257 | $340,069 | $1,176,138 | |
Cancer of unknown primary | Drug Costs | $0 | $1,464,152 | $932,317 | $839,159 | $3,235,627 |
Testing Costs | $0 | $9,434,300 | $5,400,503 | $5,407,048 | $20,241,851 | |
Budget impact | $0 | $10,898,452 | $6,332,820 | $6,246,206 | $23,477,478 | |
Cholangiocarcinoma | Drug Costs | $0 | $27,589 | $17,819 | $16,225 | $61,632 |
Testing Costs | $0 | $231,101 | $134,323 | $135,905 | $501,329 | |
Budget impact | $0 | $258,690 | $152,142 | $152,130 | $562,961 | |
CNS/Glioma | Drug Costs | $0 | $596,192 | $376,052 | $338,476 | $1,310,720 |
Testing Costs | $0 | $2,937,677 | $1,665,762 | $1,667,781 | $6,271,220 | |
Budget impact | $0 | $3,533,868 | $2,041,814 | $2,006,257 | $7,581,940 | |
Colorectal | Drug Costs | $0 | $984,218 | $658,537 | $608,472 | $2,251,227 |
Testing Costs | $0 | $5,496,258 | $3,320,668 | $3,397,889 | $12,214,816 | |
Budget impact | $0 | $6,480,477 | $3,979,204 | $4,006,362 | $14,466,043 | |
Endometrial | Drug Costs | $0 | $249,271 | $160,994 | $146,593 | $556,858 |
Testing Costs | $0 | $2,197,935 | $1,277,512 | $1,292,557 | $4,768,003 | |
Budget impact | $0 | $2,447,205 | $1,438,506 | $1,439,150 | $5,324,861 | |
Head and neck | Drug Costs | $0 | $723,715 | $467,418 | $425,608 | $1,616,741 |
Testing Costs | $0 | $6,381,323 | $3,709,035 | $3,752,715 | $13,843,073 | |
Budget impact | $0 | $2,447,205 | $1,438,506 | $1,439,150 | $5,324,861 | |
Hepatic | Drug Costs | $0 | $76,329 | $48,613 | $43,763 | $168,705 |
Testing Costs | $0 | $2,131,257 | $1,220,264 | $1,221,931 | $4,573,452 | |
Budget impact | $0 | $2,207,586 | $1,268,877 | $1,265,695 | $4,742,157 | |
MASC | Drug Costs | $0 | $1,083,332 | $750,089 | $704,773 | $2,538,194 |
Testing Costs | $0 | $20,166 | $12,646 | $13,119 | $45,931 | |
Budget impact | $0 | $1,103,498 | $762,735 | $717,892 | $2,584,125 | |
Melanoma | Drug Costs | $0 | $314,506 | $203,127 | $184,957 | $702,590 |
Testing Costs | $0 | $2,195,404 | $1,276,041 | $1,291,068 | $4,762,514 | |
Budget impact | $0 | $2,509,910 | $1,479,168 | $1,476,026 | $5,465,103 | |
Neuroendocrine | Drug Costs | $0 | $45,037 | $29,088 | $26,486 | $100,610 |
Testing Costs | $0 | $503,007 | $292,364 | $295,807 | $1,091,179 | |
Budget impact | $0 | $548,044 | $321,452 | $322,293 | $1,191,789 | |
NSCLC | Drug Costs | $0 | $2,343,837 | $1,561,947 | $1,440,046 | $5,345,829 |
Testing Costs | $0 | $0 | $0 | $0 | $0 | |
Budget impact | $0 | $2,343,837 | $1,561,947 | $1,440,046 | $5,345,829 | |
Ovarian | Drug Costs | $0 | $601,249 | $388,323 | $353,587 | $1,343,160 |
Testing Costs | $0 | $3,249,301 | $1,888,601 | $1,910,842 | $7,048,743 | |
Budget impact | $0 | $3,850,550 | $2,276,923 | $2,264,429 | $8,391,903 | |
Pancreas | Drug Costs | $0 | $871,944 | $590,094 | $530,332 | $1,992,371 |
Testing Costs | $0 | $8,592,843 | $5,227,041 | $5,226,233 | $19,046,117 | |
Budget impact | $0 | $9,464,787 | $5,817,135 | $5,756,565 | $21,038,487 | |
Prostate | Drug Costs | $0 | $1,217,577 | $786,384 | $716,042 | $2,720,004 |
Testing Costs | $0 | $9,271,945 | $5,389,160 | $5,452,625 | $20,113,729 | |
Budget impact | $0 | $10,489,522 | $6,175,544 | $6,168,668 | $22,833,733 | |
STS | Drug Costs | $0 | $63,545 | $44,750 | $42,473 | $150,768 |
Testing Costs | $0 | $462,864 | $295,626 | $309,365 | $1,067,855 | |
Budget impact | $0 | $526,409 | $340,376 | $351,838 | $1,218,623 | |
Thyroid | Drug Costs | $0 | $2,518,540 | $1,893,342 | $1,860,890 | $6,272,772 |
Testing Costs | $0 | $1,834,501 | $1,257,382 | $1,355,448 | $4,447,331 | |
Budget impact | $0 | $4,353,041 | $3,150,725 | $3,216,338 | $10,720,103 |
Ethics Review
Abbreviations
HTA
health technology assessment
NTRK
neurotrophic tyrosine receptor kinase
TRK
tropomyosin receptor kinase
Summary
The literature on ethical issues related to tumour-agnostic therapies and evaluations of their effectiveness through basket trials — along with the use of genetic testing (companion diagnostic or otherwise) to identify people living with neurotrophic tyrosine receptor kinase (NTRK) fusion-positive solid tumours — were reviewed to identify ethical considerations related to the use of entrectinib.
Ethical issues identified in the context of genetic testing for NTRK fusion-positive solid tumours included questions regarding the validity and utility of the genetic tests used to identify NTRK gene fusions; the accessibility or availability of these tests; and considerations regarding resource allocation and costs of genetic testing.
Ethical issues identified regarding the application of basket trials using master protocols to evaluate the effectiveness of tumour-agnostic therapies included challenges to their scientific validity and the potential for undue risks to clinical trial participants. The literature identified that scientific validity could be affected by assumptions around: the use of a single treatment for a single biomarker in tumours that may have heterogenous molecular aberrations; the absence of comparative data; issues related to publication bias, given master protocols open-ended inclusion and exclusion criteria; and insufficient trial participation or diversity. Challenges identified in relation to the potential for undue risks to research participants in relevant clinical trials included those around the balance between risks and benefits and the ability to achieve valid informed consent from patients.
Objective
To identify and describe ethical considerations raised in the literature associated with the use of entrectinib, a tumour-agnostic therapy, for the treatment of adult patients who have unresectable, locally advanced or metastatic extracranial solid tumours (including brain metastases) with NTRK gene fusion without a known acquired resistance mutation and no satisfactory treatment options.
Research Question
This report addresses the following research question:
What are the ethical considerations raised in the published literature relevant to the use of entrectinib as a proposed tumour-agnostic therapy for adults living with NTRK fusion-positive solid tumours?
To address this question, a literature search strategy was developed to examine ethical issues related to tumour-agnostic therapies and the trial designs used to evaluate their effectiveness — specifically, basket trials using master protocols— and to examine the allocation and use of genetic testing (companion diagnostic or otherwise) to identify people with the relevant biomarkers (i.e., NTRK fusion-positive solid tumours). Rather than seek out literature specific to entrectinib alone, this broadened strategy allows for the identification of ethical considerations associated with the tools used to assess the effectiveness of entrectinib (e.g., basket trials) and the tests to identify the NTRK+ gene fusions at which entrectinib is aimed.
Methods
Data Collection: Review of Empirical and Normative Ethics Literature
A review of the empirical literature (i.e., that focused on explaining “what is” through observation) and normative literature (i.e., that focused on explaining “what ought to be” through argumentation) relevant to ethical considerations in the context of the use of entrectinib was conducted. This included a review of the literature relevant to the use of entrectinib as a tumour-agnostic therapy, including that related to the use of genetic testing (as a companion test or broadly), as well as the types of clinical trials that support these applications (i.e., basket trials using master protocols).
Literature Search Methods
A literature search was conducted by an information specialist on key resources including MEDLINE All (1946–) through Ovid and Philosopher’s Index through Ovid. Duplicates were removed by manual deduplication in Endnote. The search strategy comprised both controlled vocabulary, such as the National Library of Medicine’s MeSH (Medical Subject Headings), and keywords. The main search concepts were Rozlytrek (entrectinib), NTRK gene fusions, tumour-agnostic therapies, basket trials, and companion diagnostics.
CADTH-developed search filters were applied to limit retrieval to citations related to empirical and normative ethical considerations. The initial search was completed on February 16, 2022, and limited to English-language documents published since January 1, 2012. Due to the iterative nature of the literature review, additional searching was required to capture literature on companion diagnostics. This additional search was completed on March 11, 2022, and limited to English-language documents published since January 1, 2012.
Grey literature (literature that is not commercially published) was identified by searching sources listed in the ethics section of the CADTH Grey Matters: A Practical Tool For Searching Health-Related Grey Literature checklist. The grey literature searches for ethical considerations were completed on February 16, 2022, and March 11, 2022. The main search concepts were entrectinib, NTRK gene fusions, tumour-agnostic therapies, basket trials, and companion diagnostics. The search was limited to English-language documents published since January 1, 2012. Google was used to search for additional internet-based materials. These searches were supplemented by reviewing bibliographies of key papers and through contacts with experts, as appropriate.
Literature Screening and Selection
Eligible publications were those published in English that identified normative or empirical ethical considerations related to the use of entrectinib as a tumour-agnostic therapy for adults living with NTRK fusion-positive solid tumours. Given the novelty of tumour-agnostic therapies and the trial designs used to evaluate the effectiveness of these therapies, eligibility for inclusion was extended to publications that identified normative or empirical ethical considerations related to tumour-agnostic therapies or basket trials using master protocols. Similarly, considering the underlying importance of identifying NTRK gene fusions for the use of entrectinib, publications that identified ethical issues related to genetic testing – companion diagnostic or generally – were eligible for inclusion. The selection criteria can be found in Table 1.
Criteria | Description |
|---|---|
Population | Adult patients who have unresectable, locally advanced or metastatic extracranial solid tumours, (including brain metastases) with a neurotrophic tyrosine receptor kinase gene fusion without a known acquired resistance mutation and no satisfactory treatment options |
Interventions | Entrectinib, precision oncology applications |
Context | Any health system |
Outcomes | Normative literature: provides normative analysis of an ethical consideration Empirical literature: provides empirical research directly addressing an ethical issue |
Publication types | Primary or secondary research, normative analysis, opinion, commentary, book, or book chapter |
The selection of relevant literature proceeded in 2 stages. In the first stage, the titles and abstracts of citations were screened for relevance by a single reviewer. Citations were categorized as “retrieve, “do not retrieve,” or “unsure,” according to the selection criteria outlined in Table 1 and the following criteria:
The citation explicitly provides a normative analysis (i.e., focused on “what ought to be” through argumentation) of ethical considerations arising in the use of entrectinib (or tumour-agnostic therapies more broadly) or related to the identification, incidence, treatment, or outcomes of adults with NTRK gene fusions.
The citation presents empirical research (i.e., focused on explaining “what is” through observation) addressing ethical considerations arising in the use of use of entrectinib (or tumour-agnostic therapies more broadly) or related to the identification, incidence, treatment, or outcomes of adults with NTRK gene fusions.
The citation explicitly identifies, but does not investigate empirically, ethical considerations arising from the use entrectinib (or tumour-agnostic therapies more broadly) or related to the identification, incidence, treatment, and outcomes of adults with NTRK gene fusions.
For citations that were identified as “unsure,” a second reviewer screened and discussed these abstracts with the original reviewer. Once both reviewers came to an agreement on whether these abstracts were “retrieve” or “do not retrieve,” the primary reviewer proceeded to the second stage of the review.
In the second stage, the full-text reports categorized as “retrieve” were reviewed by the same reviewer. Reports meeting the criteria discussed earlier were included in the review, and reports that did not meet these criteria were excluded. Members of the CADTH review team were consulted to resolve uncertainties related to the eligibility of full-text reports. As a parallel process, grey literature was reviewed, and relevant sources were retrieved and reviewed following the selection criteria listed previously.
Data Extraction
One reviewer extracted basic details on publications using a data extraction form. The following publication details were recorded: first author, article title, publication objectives, publication type, date of publication, funding source, and key findings identified related to ethical considerations.
Data Summary
The same reviewer conducted 2 cycles of coding to abstract, identify, and synthesize relevant ethical issues in the literature. In the initial coding phase, the publications were reviewed for ethical content, and ethically relevant claims made in the literature were noted and grouped according to theme. The EUnetHTA Core Model 3.0 (Ethical Analysis Domain)1 questions deemed “critically important” were used as a guide to identify and categorize ethical considerations related to the use of entrectinib and ethical issues related to tumour-agnostic drug development, basket trials, and companion diagnostics for targeted therapies. The Core Model was chosen because it is a wide-ranging framework; the assessment questions in the domain are intended especially for identifying ethically relevant issues and conflicts.1 This guiding framework highlights the context of a technology and focuses on the following topics: benefit-harm balance, autonomy, respect for persons, justice and equity, legislation, and ethical consequences of the health technology assessment (HTA).
Once identified, passages or claims related to ethical content were coded using methods of qualitative description.2 Initial descriptive coding of the reports focused broadly on categories concerning what ethical considerations were described. Major themes and sub-codes were identified through repeated readings of the data.2 Once sub-codes emerged, these were deductively applied to all reports in the set, and ethical content was summarized into the thematic categories. This review focused on ethical considerations relating specifically to the use of entrectinib and related ethical issues deriving from tumour-agnostic drug development, basket trials, and companion diagnostics for targeted therapies. Other ethical considerations raised in the literature, but not related to the use of entrectinib as a tumour-agnostic treatment of NTRK fusion-positive tumours, were outside of the scope of the current review and are not included in the summary.
Results
Description of Included Publications
A total of 175 citations were identified in the literature search. Following the screening of titles and abstracts, 135 citations were excluded, and 40 potentially relevant publications from the electronic search were retrieved for full-text review. Three potentially relevant reports were retrieved from other sources, including the grey literature search. Of the potentially relevant publications, 24 publications were excluded for various reasons (e.g., ethical claims not specific to use of entrectinib [n = 9]; reviews of regulatory processes [n = 7]; no ethical claims made [n = 6]; focused on pediatric population [n = 2]). Nineteen publications met the inclusion criteria and are included in this report. Figure 1 presents the flow diagram for the inclusion and exclusion of publications of the study selection process.
Details regarding the characteristics of the included publications are reported in Table 2. None of the included publications reported directly on ethical considerations in the use of entrectinib for the treatment of adults with unresectable, locally advanced or metastatic extracranial solid tumours (including brain metastases) that have an NTRK gene fusion without a known acquired resistance mutation and satisfactory treatment options. A total of 11 publications examined ethical issues related to genetic testing for NTRK fusion-positive solid tumours, and 8 were related to the use of basket trials for tumour-agnostic therapies or precision oncology trials generally.
Key Ethics Considerations From the Literature
Ethical Issues in Genetic Testing for NTRK Fusion-Positive Solid Tumours
Given that NTRK gene fusions are present in only 1% or so of solid tumours broadly,3-5 knowing how to test, who to test, and when to test can be challenging.3,4,6-8 Given this, the validity, accessibility, and costs of genetic testing were all reported as primary ethical issues surrounding the use of tropomyosin receptor kinase (TRK) inhibitors (i.e., entrectinib) as a treatment for people living with NTRK fusion-positive solid tumours.
Validity and Utility of Testing
To appropriately treat people living with NTRK fusion-positive solid tumours, not only it is important to have diagnostic tests with a high degree of sensitivity (analytical validity), but — as an HTA from the Norwegian Institute of Public Health suggests4 — these tests should also be able to identify fusions that are more likely to respond to treatment and improve patient outcomes (clinical utility). This can reduce the likelihood of identifying false positives and affect the cost-effectiveness of TRK inhibitors (e.g., entrectinib) by limiting the number of people receiving a targeted therapy that will not work for them or that could potentially cause further harm.4 Similarly, some authors noted that, while not specifically focused on NTRK gene fusions, molecular testing would benefit from the ability to test for multiple biomarkers that are reflective of intra-tumoural heterogeneity, the possible presence of other driver mutations, and how these might be affected by geographic, ethnic, and other clinical differences.9-11 The Norwegian HTA4 and other Canadian3 or international6,7 consensus documents all demonstrate that questions remain regarding which diagnostic tests should be used to identify people living with NTRK gene fusions.
Relatedly, authors noted the importance of considering the prognostic and predictive value of the biomarkers in question for molecular testing to be useful.11,12 While a prognostic biomarker provides information about cancer outcomes regardless of treatment, predictive biomarkers offer information about the likely effects of targeted treatment.11 In a systematic review meant to inform National Institute for Health and Care Excellence appraisals of tumour-agnostic cancer drugs, the authors questioned whether the prognostic and/or predictive value of NTRK gene fusions was consistent across all tumour types.13 This calls into question whether TRK inhibitors (e.g., entrectinib) can truly be tumour-agnostic, given the uncertainty around the prognostic and predictive value of NTRK gene fusions.13
Access to Testing
According to 1 Canadian consensus document,3 access to diagnostic testing for NTRK gene fusions is limited in Canada, and available only sporadically through industry-sponsored programming, private pay, or some select government-funded institutions or programs. This variability in availability may lead to inequitable access to molecular testing.3,5 Other authors, focusing on precision oncology broadly, noted that better access to precision testing is necessary to maximize the benefits of precision oncology.10,12,14 Keeling et al.14 indicated a particular concern that diagnostic testing either happens too late or not at all due to the inefficient dispersal of diagnostic technologies.
Considerations for Resource Allocation and Costs
Improving access to testing for NTRK gene fusions could require screening a large number of patients who do not carry NTRK gene fusions; this could be costly.5,15 Given that NTRK gene fusions can happen across tumour histology, 1 author suggested that payers will need to consider whether it is the best use of resources to implement a broad strategy focused on testing all tumour types that might carry NTRK gene fusions, or if such a strategy should focus on rarer tumour types that have a greater chance of carrying these fusions.5 In either case, several authors indicated the importance of defining a consistent diagnostic strategy for identifying NTRK gene fusions.3,5,7 While this may include heavy upfront costs, the availability of molecular tests capable of consistently identifying people living with NTRK gene fusions likely to respond to therapy across tumour types also has the capacity to improve the efficient use of health resources.4
Canadian3 and international6 consensus documents indicate that there are still unsettled questions around where, among the variety of treatment pathways associated with heterogenous tumour types potentially carrying NTRK gene fusions, diagnostic testing for NTRK gene fusions (and subsequent treatment with TRK inhibitors like entrectinib) might belong. Given this heterogeneity, the implementation of TRK inhibitors (e.g., entrectinib) will variably affect how clinical care is organized and provided across tumour specializations. Therefore, it needs to be contended with.5
Ethical Issues in Clinical Trial Design for Tumour-Agnostic Therapies
As proposed in the context of tumour-agnostic therapies, evaluations of the effectiveness of TRK inhibitors like entrectinib (or larotrectinib) have required the implementation of trial designs that can focus on a single molecular change across a wide range of tumour types simultaneously. Basket trials with master protocols have increasingly be been used as a means of getting at the complexity of tumour-agnostic therapies.13,16,17 According to Murphy et al.,13 rather than developing a new protocol for each cancer type or drug under investigation, master protocols use a single protocol to evaluate multiple cancer populations or multiple drugs at the same time. As a type of trial that uses a master protocol, basket trials assume that molecular subtype (as opposed to tumour histology or location) is predictive of treatment response; therefore, these trials focus on evaluating the effectiveness of a single drug across a number of tumour types harbouring the molecular aberration in focus.13,16
While none of the included studies focused specifically on the use of basket trials for entrectinib, ethical issues reported across studies investigating tumour-agnostic therapies and/or basket trials are relevant to the consideration of entrectinib. These ethical considerations included a range of issues regarding scientific validity, considerations around the risk versus benefit ratio for clinical trial participants, and implications for informed consent.
Challenges Around the Scientific Validity of Basket Trials
Given that the purpose of tumour-agnostic basket trials is to identify the clinical effect of a single therapy on a single biomarker across a variety of tumour types, authors indicated the potential to neglect the complexity and heterogeneity of tumour histologies as an ethical issue.13,16-18 The assumption that the biomarker being worked on throughout the basket trial acts homogenously across all tumour types needs to be proven by providing any heterogenous treatment effects by cohort.16 If wide heterogeneity is observed, the hypothesis of agnostic effect is violated and cannot be supported.16
Another ethical issue for consideration was related to the rarity of biomarkers being investigated in basket trials, which makes it possible that patient accrual is insufficient to develop statistically significant findings.16-18 With this in mind, authors noted that there are questions as to whether the results of basket trials are robust enough to provide reimbursement recommendations.8,13 However, there is some consensus among clinicians in Canada that evidence from basket trials for the use of TRK inhibitors (e.g., entrectinib) to treat NTRK fusion-positive solid tumours is sufficient.3,8
Another ethical issue noted regarding the scientific validity of basket trials was situated around the challenges of collecting and analyzing comparative data.13,16,19 The breadth of tumour types with the potential to carry NTRK gene fusions, and uncertainties around where in the treatment algorithm TRK inhibitors might belong, make it challenging to develop the appropriate historical control data for comparative analysis against other standards of care.13 While authors of documents meant to support national HTA bodies' evaluations of tumour-agnostic therapies (e.g., entrectinib) considered it important to find ways of implementing comparative analyses for tumour-agnostic therapies,13,16 some authors challenged the use of randomized comparative trials as unethical for tumour-agnostic therapies (e.g., entrectinib), given the limitations around clinical equipoise in comparator arms.19 As well, given the open-ended adaptability of basket trials in which arms can be closed if proving ineffective, authors noted the possibility for trial results that do not demonstrate efficacy to go unpublished (and the attendant increased risk of publication bias) as other ethical issues affecting scientific validity.17
Surrogate end points rate that are common in basket trials, such as progression-free survival or objective response rate, were also challenged by authors for their potentially uncertain ability to describe actual clinical benefit to patients. While progression-free survival or objective response rate may be justifiable end points throughout the early research phases, authors were concerned that these end points may not necessarily translate into outcomes that patients living with the biomarkers under review consider valuable.
While not directly focused on basket trials for tumour-agnostic therapies, some authors also reported disparities regarding access to precision oncology clinical trials generally for breast, lung, and prostate cancers in the US, specifically where Black patients tend to be poorly represented in these trials, according to incidence.20,21 Authors noted how the unchecked under-representation of Black patients in precision oncology trials risks further entrenching existing health inequities by failing to determine whether trial results are applicable and safe for a diverse cancer population and leading to poor treatment decisions for Black patients.20,21
Risks for Clinical Trial Participants
In addition to the challenges related to the scientific validity of basket trials that use master protocols, some authors articulated as ethical considerations concerns about the risks of participating in basket trials for tumour-agnostic therapies and the possibility of informed consent not being achieved.
As has already been noted as a concern about the scientific validity of basket trials, testing a single therapy on a single biomarker was described as posing challenges to the balance of risks and benefits for trial participants.10,15,17 Given the possibility that tumour molecular profiles can evolve and become resistant to targeted therapy or develop new molecular alterations that drive disease progression,15 ignoring intra-tumoural molecular heterogeneity may inappropriately include or exclude prospective trial participants.17
Another issue raised in the published literature was that, depending on the location of a person’s tumour or how many lines of therapy they have already undergone, there is some risk that they could be harmed during the collection of a biopsy for genetic testing.17 Similarly, depending on testing strategy, it may be impossible or challenging to collect enough tumour tissue to use for testing, or the tissue sample taken may be of low quality or fail to “capture the complete genomic landscape,” which may result in false positives or negatives that send the patient to an inappropriate study arm and pose the potential for harm.15,17 The challenge of keeping genetic information collected throughout the trial safe and private was also identified as an ethical issue.17
Considering that participants in basket trials are living with life-threatening conditions, authors were concerned that therapeutic misunderstanding may be exacerbated in the context of precision oncology, given the use of language such as “personalized,” “individualized,” or “tailored,” and that this may interfere with patients’ ability to provide valid informed consent.17 These misunderstandings may include: patient misunderstanding of trial purpose for population level versus personal benefit; an overestimated expectation that the precision therapy has been adjusted to their individual needs; and a generally overestimated expectation of benefit from precision therapy.17 While many of these misunderstandings may be present for precision oncology clinical trials generally, authors noted that this does not lessen the importance of attending to them in the context of basket trials specifically.17
Limitations
This review is limited by the lack of published literature examining ethical considerations directly relevant to the use of entrectinib for the treatment of adults with unresectable, locally advanced or metastatic extracranial solid tumours (including brain metastases) that have an NTRK gene fusion without a known acquired resistance mutation and no satisfactory treatment options. The absence of directly applied, published, ethical analyses does not indicate that ethical considerations are not present. Many of the ethical issues associated with the testing and treatment of NTRK fusion-positive tumours, or those noted regarding basket trials or precision oncology broadly, are likely of relevance to entrectinib as well.
Finally, this review is limited to the ethical considerations explicitly discussed in the published literature and lacks ethical insights that might be derived from primary research, stakeholder engagement, or primary normative analysis. Some of the results and insights raised relating to clinical effectiveness, safety, and costs might be discussed more comprehensively in the clinical and pharmacoeconomic review sections, which may hold implicit ethical implications.
Conclusions
The literature on ethical issues related to tumour-agnostic therapies and evaluations of their effectiveness through basket trials — and the use of genetic testing (companion diagnostic or otherwise) to identify people living with biomarkers in question — were reviewed to identify ethical considerations related to the use of entrectinib as a tumour-agnostic therapy for adults living with NTRK fusion-positive solid tumours. Ethical considerations raised in the context of genetic testing for NTRK gene fusions indicate concerns around the validity and utility of various genetic tests that might be used to identify tumours harbouring these fusions, the accessibility of these tests, and challenges with resource allocation. Ethical issues identified regarding the application of basket trials using master protocols to evaluate the effectiveness of tumour-agnostic therapies included challenges to their scientific validity and the potential for undue risks to research participants. Scientific validity could be affected by assumptions around: the use of a single treatment for a single biomarker in tumours that may have heterogenous molecular aberrations; the absence of comparative data; publication bias, given s’ open-ended inclusion and exclusion criteria; and insufficient trial participation or diversity. The potential for undue risks to research participants in associated clinical trials included those related to the certainty of balance between risks and benefits and the ability to achieve valid informed consent from patients.
References
1.EUnetHTA Joint Action 2 Work Package 8, HTA Core Model version 3.0. 2016: https://www.htacoremodel.info/BrowseModel.aspx. Accessed 2022 Apr 25.
2.Sandelowski M. Whatever happened to qualitative description? Research in nursing & health. 2000;23(4):334-340. PubMed
3.Bebb DG, Banerji S, Blais N, et al. Canadian Consensus for Biomarker Testing and Treatment of TRK Fusion Cancer in Adults. Curr Oncol. 2021;28(1):523-548. PubMed
4.Norwegian Institute of Public Health. Health technology assessment: Tests for the detection of NTRK gene fusions in patients with locally advanced or metastatic solid tumours 2022; https://www.fhi.no/globalassets/dokumenterfiler/rapporter/2022/tests-for-the-detection-of-ntrk-gene-fusions-in-patients-with-locally-advanced-or-metastatic-solid-tumours-offentlig-rapport-2022.pdf Accessed 2022 Jun 22.
5.Walker A. Neurotrophic tyrosine kinase inhibitors: A review of implications for patients, clinicians and healthcare services. J Oncol Pharm Pract. 2020;26(8):2015-2019. PubMed
6.Yoshino T, Pentheroudakis G, Mishima S, et al. JSCO-ESMO-ASCO-JSMO-TOS: international expert consensus recommendations for tumour-agnostic treatments in patients with solid tumours with microsatellite instability or NTRK fusions. Ann Oncol. 2020;31(7):861-872. PubMed
7.Garrido P, Hladun R, de Álava E, et al. Multidisciplinary consensus on optimising the detection of NTRK gene alterations in tumours. Clinical and Translational Oncology. 2021;23(8):1529-1541. PubMed
8.Simmons C, Deyell RJ, MacNeill AJ, et al. Canadian consensus on TRK-inhibitor therapy for NTRK fusion-positive sarcoma. Int J Cancer. 2021;149(9):1691-1704. PubMed
9.Jorgensen JT. Companion diagnostics in oncology - current status and future aspects. Oncology. 2013;85(1):59-68. PubMed
10.Murciano-Goroff YR, Drilon A, Stadler ZK. The NCI-MATCH: A National, Collaborative Precision Oncology Trial for Diverse Tumor Histologies. Cancer Cell. 2021;39(1):22-24. PubMed
11.O'Brien CP, Taylor SE, O'Leary JJ, Finn SP. Molecular testing in oncology: problems, pitfalls and progress. Lung Cancer. 2014;83(3):309-315. PubMed
12.Govaerts L, Simoens S, Van Dyck W, Huys I. Shedding Light on Reimbursement Policies of Companion Diagnostics in European Countries. Value Health. 2020;23(5):606-615. PubMed
13.Murphy P, Glynn D, Dias S, et al. Modelling approaches for histology-independent cancer drugs to inform NICE appraisals: a systematic review and decision-framework. Health Technol Assess. 2021;25(76):1-228. PubMed
14.Keeling P, Clark J, Finucane S. Challenges in the clinical implementation of precision medicine companion diagnostics. Expert Rev Mol Diagn. 2020;20(6):593-599. PubMed
15.Tsimberidou AM, Fountzilas E, Nikanjam M, Kurzrock R. Review of precision cancer medicine: Evolution of the treatment paradigm. Cancer Treat Rev. 2020;86:102019. PubMed
16.Lengline E, Peron J, Vanier A, et al. Basket clinical trial design for targeted therapies for cancer: a French National Authority for Health statement for health technology assessment. Lancet Oncol. 2021;22(10):e430-e434. PubMed
17.Strzebonska K, Waligora M. Umbrella and basket trials in oncology: ethical challenges. BMC Med Ethics. 2019;20(1):58. PubMed
18.Horgan D, Ciliberto G, Conte P, et al. Bringing Onco-Innovation to Europe's Healthcare Systems: The Potential of Biomarker Testing, Real World Evidence, Tumour Agnostic Therapies to Empower Personalised Medicine. Cancers (Basel). 2021;13(3):02.
19.Bayle A, Italiano A, Massard C, Blay JY, Marabelle A. Basket trial health technology assessment requirements and limited access to innovations in oncology: The French paradox. Eur J Cancer. 2022;162:128-129. PubMed
20.Aldrighetti CM, Niemierko A, Van Allen E, Willers H, Kamran SC. Racial and Ethnic Disparities Among Participants in Precision Oncology Clinical Studies. JAMA netw. 2021;4(11):e2133205.
21.Esdaille AR, Ibilibor C, Holmes A, 2nd, Palmer NR, Murphy AB. Access and Representation: A Narrative Review of the Disparities in Access to Clinical Trials and Precision Oncology in Black men with Prostate Cancer. Urology. 2021;25:25. PubMed
Appendix 1: Selection of Included Studies
Appendix 2: Details of Included Publications
Note that this appendix has not been copy-edited.
Table 2: Details of Included Publications
First author, year | Publication type | Objective | Key ethical considerations | Funding source |
|---|---|---|---|---|
Bayle, 202219 | Commentary | To respond to Lengliné 2021 on criteria around HTA assessment of basket trial data | Randomized comparative trials for tumour-agnostic therapies can be challenging or untenable given limited “clinical equipoise of treatment success in comparator arms.” | None declared |
Norwegian Institute of Public Health, 20224 | Rapid HTA | “To assess molecular tests for the identification of NTRK gene fusions in locally advanced or metastatic solid tumours” | The availability of good companion diagnostics for targeted therapies (like TRK inhibitors) can improve the efficient use of resources. Biomarker tests not only need to have a high degree of accuracy for identifying the biomarker, but should also be able to identify biomarkers that can be worked on and can improve patient outcomes (clinical utility). Access to testing modalities with a high degree of specificity regarding the biomarker in question reduces likelihood of false positives which can have an impact on the cost-effectiveness of the therapy by limiting the amount of people receiving a targeted therapy that will not work for them or could potentially cause further harm. | None declared |
Aldrighetti, 202120 | Cross-sectional study | To understand how well precision oncology studies are representative of the diverse US cancer population regarding race and ethnicity | Racial and ethnic minorities are under-represented in precision oncology studies for breast, lung, and prostate cancers according to rates of incidence in the US. Need to increase diversity of precision oncology trials to understand whether trial results are broadly applicable and safe for a diverse cancer population. Lack of diversity in precision oncology trial populations may reinforce existing health inequalities. In addition to diversifying trial populations, there is a need attend to pre-existing and systemic barriers around access to cancer care for underserved populations. | None declared |
Bebb, 20213 | Guidelines | To “provide Canadian consensus on how to identify and treat patients with TRK fusion cancer” | There are questions around when and how to identify patients in Canada living with NTRK gene fusions. Limited access to NTRK fusion testing across Canada relies on industry-sponsored programs, private pay programs, or sporadically available public payer programs. Challenge identifying NTRK fusions in a methodologically consistent way that is economically feasible in public payer system. | Editorial support provided by MEDUCOM Health Inc. and sponsored by Bayer Canada Inc. |
Esdaille 202121 | Narrative Review | “To provide commentary on the disparities in access to clinical trials and precision oncology specific to Black men with Prostate Cancer (PCa) in the US and lend a general framework to aid in closing these gaps” | Need to increase inclusion of Black men in precision oncology trials for prostate cancer or risk increasing racial disparities in prostate cancer. Under-representation of Black men in precision oncology trials could heighten chance of developing poorly validated risk calculators which can lead to poor treatment decisions. | Bristol-Myers-Squibb University of Texas Health Science Center at San Antonio |
Garrido 20217 | Consensus document | To provide guidelines on the diagnostic, clinical and therapeutic aspects of “NTRK-rearranged tumours” and to discuss challenges associated with the detection of these tumours in public payer system | Need to define a diagnostic strategy for NTRK gene fusions that can help to ensure equitable access to molecular testing. | Financial support received in the form of unrestricted collaboration in the logistics of expert meeting from Bayer and Roche |
Horgan 202118 | Guidelines | To report on primary concerns around oncogenetic testing and propose actions to resolve them | Need high-quality and timely biomarker testing to be widely available. Issues around assessment of value in molecular diagnostics regarding which biomarkers are predictive of response to therapy, prognostic of progression of disease regardless of treatment or both. Need for clarity around where and when to initiate molecular testing for genetic alterations. Challenges for tumour-agnostic basket trials include developing comparative evidence and having statistically significant proof of effect across tumour types given low patient numbers; lack of clarity in diagnostic pathway across tumour histologies; clinical utility of surrogate end points like PFS or ORR; heterogeneity of previous lines of therapy across tumour types; complexity of health care sectors and reimbursement models. | Novartis, AstraZeneca, Eli Lilly and Company, Myriad Genetics |
Lengliné, 202116 | Recommendation | To articulate a series of recommendations for sponsor submissions that involve basket trials | Basket trials for tumour-agnostic therapies need to find ways to incorporate comparative analysis. Trial end points need to demonstrate an evaluation of clinical utility; end points like ORR need to be analyzed alongside duration of this response; common basket trial end points like ORR and PFS have not yet been validated as adequate surrogates for overall survival. Companion diagnostics should be evaluated for validity, reliability, and clinical utility at the same time as the targeted therapies. | None declared |
Murphy 202113 | Systematic Review and Decision Framework | To explore the “extent to which the National Institute for Health and Care Excellence’s existing approaches for assessing clinical and economic value can be applied to histology-independent indications, and any changes that might be required” | Is it appropriate to make “one size fits all” reimbursement recommendations on tumour-agnostic drugs, given the heterogeneity of tumour histology, small sample sizes, and variable response rates across tumours? Challenges around developing appropriate historical control data for tumour-agnostic therapies studied through basket trials. Challenges around understanding whether the prognostic and predictive value of NTRK fusion is consistent across tumour types given limited inclusion of tumour types with potential to carry NTRK fusions in clinical trials. Challenges associated with the generalizability of tumour-agnostic basket trials given differences in treatment histories across tumour types; presence of some, but not other tumour types that may carry biomarker in question; and whether distribution of tumour types in trails are what would be expected in clinical practice. Challenges regarding the distribution or availability of molecular testing across various tumour types. | NIHR |
Simmons 20218 | Consensus Document | To communicate the clinical importance of TRK inhibitors in NTRK fusion-positive sarcomas, based on a modified Delphi consensus, for the sarcoma community in Canada | There is a “need to consider new approaches to clinical development and reimbursement that are more in line with the molecularly diverse nature of cancer.” There is an unmet clinical need for systemic therapy options for people living with relapsed or refractory soft-tissue sarcoma or bone sarcoma – “especially among frail and heavily pre-treated patients.” Challenges around trial recruitment for sarcomas, which are rare/heterogeneous and whether basket trials can provide sufficient evidence to make reimbursement decisions regarding use of TRK inhibitors for NTRK fusion-positive relapsed or refractory sarcomas. | Bayer and Hoffman-La Roche Canada |
Govaerts 202012 | Review | To “systematically map in vitro diagnostic reimbursement procedures and identify policies for aligning these procedures with the pharmaceutical reimbursement procedures” | Desynchronized reimbursement review procedures between companion diagnostics and precision medicines can hamper the value of precision medicines and lead to potentially suboptimal clinical decisions. Questions around what sort of evidence would justify a “robust assumption” that a chosen biomarker has predictive clinical utility in the absence of randomized trial designs (that are replaced by basket trial designs) in which marker-positive and marker-negative populations receive the targeted therapy. | Research Foundation Flanders |
Keeling 202014 | Review | To describe “implementation challenges facing companion diagnostic tests and delaying clinical uptake” and to “assess the ‘siloed thinking’ perpetuating these challenges and propose steps toward resolution” | Need to improve availability of diagnostic testing in precision medicine as it currently happens “too late to ensure optimal outcomes” or not at all due to inefficient testing pathways or limited “deployment of testing technologies.” Need to understand that “better precision testing will enable equal or greater health outcomes for patients than new precision medicine treatments alone…” | Diaceutics |
Murciano-Goroff 202010 | Trial summary | Describe results of NCI-MATCH trial focused on broad-based tumour sequencing to identify (currently) actionable genetic alterations (genetic alterations that have known therapies that have either received approval, are in ongoing trials, or have robust pre-clinical data) | Need for improved access to broad-based tumour sequencing across diverse populations of cancer patients in order to maximize benefits of precision oncology. Need testing strategies that can identify potential presence of co-alterations or acquired resistance to targeted therapies early on in order to maximize responses to targeted therapies. | None declared |
Tsimberidou 202015 | Review | To “review the rapid evolution of precision medicine in oncology and, in particular, the challenge and opportunity that genomic science has revealed vis-à-vis the need for ‘N-of-1’ treatments” | Potential differences in response to targeted therapies based on histology or presence of genomic co-alterations. Heterogeneity between primary tumour and metastatic sites within one person may make it challenging to know whether the genomic information gathered from one biopsy is representative of larger, systemic disease. Tumour molecular profiles can evolve and become resistant to targeted therapy or develop new molecular alterations driving disease progression. Identifying rare and specific genomic alterations like NTRK gene fusions requires screening large amounts of patients who do not carry such fusions. Incomplete biologic/molecular profiles (e.g., histology/presence of co-alterations) can make it challenging to select appropriate targeted therapy. Potential differences in metabolism or adverse effects in populations not included in study population. Variability of specificity/sensitivity across molecular tests for specific alterations. Restrictive eligibility criteria across clinical trials for targeted therapies limits real-world applicability of therapeutics under review. | NIH/NCI |
Walker, 20205 | Commentary | To identify some potential challenges associated the approval of Larotrectinib (or other, like entrectinib, tumour-agnostic drugs) | Rarity of NTRK gene fusions across solid tumours broadly has made it difficult to enrol enough patients in trials to support a robust statistical analysis. Tumour-agnostic therapies (like those focused on NTRK gene fusions) have the potential to provide treatment access to a wider patient cohort than therapies focused on single histologies. Regulatory approval of tumour-agnostic TRK inhibitors might affect how care is provided in clinical settings. Regulatory approval of tumour-agnostic TRK inhibitors has provided a challenge of how to arrange diagnostic testing for NTRK gene fusions across a wide range of tumour histologies. Payers will need to decide how to provide this testing and whether it should focus broadly on any tumour type that might carry NTRK gene fusions or those, even more rare, tumour types that have a greater chance of carrying the fusions. To fully realize any potential benefit of TRK inhibitors, significant investment in diagnostic resources is likely to be required. | None declared |
Yoshino 20206 | Consensus Document | “To provide guidance for the use and management of the currently approved tumour-agnostic therapies in patients with solid tumours” | Questions around who should be tested for NTRK gene fusions Questions around when testing for NTRK gene fusions should be considered Questions around which biospecimen and diagnostic tests are most appropriate for identifying NTRK gene fusions Questions around which appropriate treatment options for people living with NTRK gene fusion-positive solid tumours Questions around where in the treatment algorithm treatment with TRK inhibitors be used | Japan Society of Clinical Oncology |
Stzrebonska, 201917 | Ethical analysis | To discuss 3 ethical requirements of clinical trials which may be challenged in basket and umbrella trial designs | Master protocols present questionable scientific validity (failure to account for intra-tumoural heterogeneity; high risk of trial result publication bias; insufficient participant enrolment). Questionable risk versus benefit ratio (unclear value of surrogate end points like ORR or PFS to patients; physical risks of biopsy – particularly for people who have undergone extensive systemic treatments; risk of low-quality tissue sample that may misidentify presence of genetic alteration in question and lead to inappropriate treatment; challenges of keeping molecular data private and safe). Challenges around gathering informed consent (e.g., patient misunderstanding of trial purpose for population level versus personal benefit; overestimated expectations that precision therapy adjusted just to them; overestimated expectations of benefit from precision therapy; language of ‘personalized’ or ‘individualized’ medicine can be misleading and falsely indicate the patient’s personal interest at the centre of trial). | National Science Centre, Poland |
O’Brien 201411 | Review | To discuss sources of uncertainty or confusion in molecular testing for oncology and draw out the advantages or challenges of fast paced development in the field of molecular testing | For molecular testing to be useful, important to consider the difference (and potential overlap) between prognostic and predictive biomarkers. Prognostic biomarkers describe tumour development regardless of treatment. Predictive markers describe how likely tumour is to respond to treatment in question. Molecular testing assays need to be able to account for diversity target population along lines of intra/inter-tumoural heterogeneity and how these might be affected by geographic, ethnic, and clinical differences. | None declared |
Jorgensen 20139 | Review | To “discuss issues related to the development and use of tissue-based companion diagnostics within oncology, including the regulatory aspects” | Companion diagnostic assays need to be able to test for multiple biomarkers that are reflective of intra-tumoural heterogeneity and possible presence of other driver mutations in the cancer being tested. | None declared |
abb = abbreviation; ORR = objective response rate; PFS = progression-free survival.
Stakeholder Input
Patient Input
Canadian Breast Cancer Network
About the Canadian Breast Cancer Network
The Canadian Breast Cancer Network (CBCN) is a leading, patient-directed, national health charity committed to ensuring the best quality of care for all Canadians affected by breast cancer through the promotion of information, education and advocacy activities. www.cbcn.ca. As a member of the Canadian Cancer Action Network, the Canadian Breast Cancer Network is committed to adhering to the Code of Conduct Governing Corporate Funding.
Information Gathering
Information for this submission was collected via: CBCN’s 2017 Lived Experience Breast Cancer Patient Survey: An online survey was distributed in English and French to patients living with breast cancer. No patients surveyed had direct experience with the treatment under review. Survey questions comprised of a combination of scoring options and free form commentary. Patients were contacted through the membership databases of CBCN and other patient organizations.
Patient Respondents Profile: 146 Canadian metastatic patients participated in the survey. The majority of respondents were from Ontario (53), Alberta (16), and British Columbia (15). The rest of the respondents were from Manitoba (10), Saskatchewan (9), Quebec (8), New Brunswick (3), Nova Scotia (3), Newfoundland and Labrador (2), and Prince Edward Island (2). Of those who responded about their gender, all identified as female. 74% of all participants’ first language was English and 9 identified French as their first language. Most of the respondents were first diagnosed with metastatic breast cancer when they were between the ages of 40-49 (48) and 50-59 (46). 22 were diagnosed between 60-69 years old, 20 between 30-39 years old, 5 between 70-79 and 4 between 20-29. One respondent was first diagnosed with metastatic breast cancer at 80 years or older. 71% of all respondents had children, with the majority (66) having children 20 years or older. 26 had children that were between the ages of 6 and 12, 19 had children between 13 and 19 years old, 11 had children between 2 to 5 years old and 1 respondent had a child less than one. The majority of participants were in a relationship (100) and 77% of all participants identified as heterosexual.
CBCN’s 2012 Lived Experience of Metastatic Breast Cancer Patients and Caregivers Survey Report: An online survey, conducted in collaboration with ReThink Breast Cancer, was distributed to patients living with metastatic breast cancer (mBC) and their caregivers. No patients surveyed had experience with the treatment under review. Survey questions comprised of a combination of scoring options and free form commentary. Patients were contacted through the membership databases of CBCN and other patient organizations.
Printed sources: A review was conducted of current studies and grey literature to identify issues and experiences that are commonly shared among many women living with breast cancer.
Disease Experience
Metastatic breast cancer (mBC) is the spread of cancerous cell growth to areas of the body other than where the cancer first formed, and is often more severe. It is most commonly spread to the bones, but can include the lungs, liver, brain and skin. Current treatment options for metastatic breast cancer are only effective at prolonging progression-free disease as most cases of advanced disease will progress and symptoms will worsen. Patients with a diagnosis of metastatic breast cancer understand the limitations of current treatment options and seek to live their remaining months and years with the best possible quality of life that they can achieve.
Some patients who have been diagnosed with metastatic breast cancer have been found to have neurotrophic tyrosine receptor kinase (NTRK) positive gene fusions. NTRK fusions are oncogenic drivers which have been found in over 25 tumour types, including those with limited treatment options such as secretory breast cancer (Bazhenova, L. et al. TRK fusion cancer: Patient characteristics and survival analysis in the real-world setting. Targ Oncol. 16, 389–399 (2021). https://doi.org/10.1007/s11523-021-00815-4). These gene fusions occur when the 3′ region of the NTRK gene is joined with the 5′ end of a fusion partner gene, leading to the expression of a chimeric protein with constitutively active or overexpressed kinase function that drives downstream signaling to promote tumour growth and survival (Ibid). Cancers that have metastasized to the central nervous system (CNS) are frequently associated with tumour types with NTRK+ fusions, including breast cancer. CNS metastases can be undetectable when small groups of tumour cells, known as micrometastases, cross the blood-brain barrier into the CNS. As these micrometastases grow in the CNS, they can reach a clinically significant size. Since standard scanning techniques only detect tumours and lesions when they get to a certain size, micrometastases can go undetected (Deeken J.F. & Löscher W. The blood-brain barrier and cancer: transporters, treatment, and Trojan horses. Clin Cancer Res. 13(6), 1663-74 (2007). https://doi.org/10.1158/1078-0432.ccr-06-2854). CNS disease can therefore be classified as measurable (≥ 1 dimension with a minimum size of 10mm by CT scan) or non-measurable (all other lesions, including those with unclear margins).
In our 2017 Survey (Lived Experience Breast Cancer Patient Survey), the majority of respondents experienced metastases to their bones (69%), liver (32%), and lungs (32%). 28 individuals reported having their tumour metastasize to parts other than their bones, liver, lungs and brain, while 16 had it metastasize to their brain.
The Physical Impact of Metastatic Breast Cancer
How the disease presents itself through symptoms, how it progresses, and how it is experienced varies by patient, but many of the effects of metastatic breast cancer represent a significant or debilitating impact on their quality of life. In our 2012 Lived Experience of Metastatic Breast Cancer Patients and Caregivers Survey Report (2012 Survey), patients were asked what impact cancer-related symptoms had on their quality of life:
54% of patients reported that fatigue resulted in a significant or debilitating impact, and 40% reported some or moderate impact;
39% of patients reported that insomnia resulted in a significant or debilitating impact, and 46% reported some or moderate impact;
37% of patients reported that pain resulted in a significant or debilitating impact, and 44% reported some or moderate impact.
These results were further reinforced in our 2017 Survey.
The Social Impact of Metastatic Breast Cancer
The impact of this disease spreads across all aspects of a patient’s life, restricting an individual’s employment and career, ability to care for children and dependents, and their ability to be social and meaningfully participate in their community. When asked in the 2012 Survey what kind of impact living with metastatic breast cancer has had on their quality of life:
Among those who were employed, 71% of patients identified significant restrictions to their ability to work;
Among those with children or dependents, 21% identified significant restrictions to their caregiving responsibilities and 53% reported some or moderate restrictions;
49% of patients identified significant restrictions to their ability to exercise and 38% identified some or moderate restrictions;
42% of patients identified significant restrictions to their ability to pursue hobbies and personal interests and 42% identified some or moderate restrictions;
41% of patients identified significant restrictions to their ability to participate in social events and activities and 41% identified some or moderate restrictions;
22% of patients identified significant to their ability to spend time with loved ones restrictions and 52% identified some or moderate restrictions.
Other experiences identified by patients included: guilt, the feeling of being a burden on caregivers, fear of death, poor body image, not knowing what functionality will be lost, fear of the impact of cancer and the loss of a parent on children, not knowing what will happen to children, the loss of support of loved ones, as well as marital stress/loss of fidelity and affection from husband.
Many of these sentiments were repeated by participants of our 2017 Survey anecdotally:
“Because my diagnosis happened with multiple spinal fractures and lesions in the hips and femurs[,] exercise is limited to walking and very low impact activities.”
“In dealing with lymphedema and bone [metastasis][,] some movement and exercising has become difficult.”
Experiences With Currently Available Treatments
The Goals of Current Therapy
The goal of treatment for metastatic breast cancer is to control disease progression (extending life) and to manage cancer-related symptoms (extending or stabilizing quality of life). Treatment options for mBC and their effectiveness vary among type of cancer, location of cancer, and how symptoms are experienced.
Patients diagnosed with metastatic breast cancer have very limited treatment options and patients with NTRK+ gene fusions are usually treated with TRK inhibitors. A research study (Demetri, G.D. et al. Characteristics and outcomes of patients (pts) with NTRK fusion-positive (NTRK+) metastatic/locally advanced (LA) solid tumours receiving non-TRK inhibitor (TRKi) standard of care (SoC), and prognostic value of NTRK fusions in clinical practice. Ann of Onc. 32(5), S399 (2021) was conducted to explore and compare the outcomes of NTRK fusion-positive individuals with metastatic or locally advanced solid tumours being treated with non-TRK inhibitor standard of care versus those receiving NTRK fusions in a real-world setting. Data for the study came from the Flatiron Health/Foundation Medicine clinicogenomic data, a US-wide longitudinal database of standard healthcare practice data. In the study, NTRK positivity was met when patients had a fusion or rearrangement involving NTRK 1/2/3 with predicted study known or likely functional study status. Patients were 18 years old and had one or more test by next-generation sequencing; had one or more NTRK+ test result; were diagnosed with a locally advanced or metastatic solid tumour between January 1, 2011, and December 31, 2019; and had no prior treatment with either entrectinib or larotrectinib. NTRK- patients were matched at a 10:1 ration to NTRK+ patients based on both tumour type and by using the nearest neighbour propensity score model. In total, 280 NTRK- patients were matched with 28 NTRK+ patients with locally advanced or metastatic solid tumours, one of which was a breast cancer patient. 4.6% of the NTRK- patients had brain metastases while 7.9% of the NTRK+ patients had brain metastases.
When diagnosis was used as the index date, the study found that the median overall survival (OS) was 10.2 months (with a 95% confidence interval of 7.2 to 14.1) among NTRK+ patients and 10.4 months (with a 95% confidence interval of 6.7 to 14.3) among the matched NTRK- patients. The hazard ration (HZ) of 1.6, with a 95% confidence interval (95% CI) of 1.0 to 2.5, was non-significant. When the last line of therapy before next-generation sequencing test report was used as the index date, the median OS among NTRK+ patients was 10.1, 95% CI [7.1, 13.1] and 10.5 months, 95% CI [8.6, 13.9] among the matched NTRK- group. In the instance, the hazard ration of 1.6, 95% CI [1.0, 2.5] was also not significant.
These results show that when treated with non-TRK inhibitors (non-TRKi) standard of care, NTRK+ patients had poor overall survival at a rate comparable to matched patients who were NTRK-. Such findings suggest that standard of care treatments lead to poor overall survival outcomes for both NTRK+ and NTRK- patients and that NTRK+ therefore require treatments and therapies with better efficacy and outcomes. Results from TRKi clinical trials on NTRK+ solid tumours indicate that TRKis, such as entrectinib, will likely led to better treatment outcomes among NTRK+ than non-TRKi standard of care therapies.
Participants in our 2017 Survey had been or at the time of the survey, were being treated with chemotherapy (102), surgery (96), radiation therapy (96), and hormone replacement therapy (91).
Key Factors for Decision-Making Around Treatment
Respondents in both our 2012 and 2017 surveys indicated that the following key factors influenced their decision-making around treatments:
Effectiveness of the treatment – how well the treatment stabilized their disease and delayed progression of their disease.
Prolonging life without sacrificing quality of life – being able to maintain productive, active lives with minimal disruption to daily routines.
Side effect management – minimizing risk while stabilizing their disease.
Ease and accessibility of treatments – affordability and ease of accessing treatments.
Treatment Efficacy
When asked how important progression-free survival (PFS) was in considering treatments, the majority of participants in our 2017 Survey revealed that efficacy of the treatment is an important or very important consideration to their decision-making. Of the patients who responded to the prompts, 74% indicated that a PFS of less than 3 months was important or very important, 86% indicated the same for a PFS of 3-5 months, and 98% indicated the same for a PFS of 6 months or longer. When asked about overall survival, 99% of those who responded to the question indicated that overall survival was important or very important when considering treatment options.
Metastatic patients in our 2017 Study also spoke on the importance of treatment effectiveness in their decision-making anecdotally:
“Effectiveness is most important and then all other things being equal - least side effects[.]”
“Effectiveness in reducing the size of the tumours, halting growth of potential bone involvement.”
“How well treatments will work[,] need to be around for my kids and family[.]”
“Anything to prolong my survival and maintain quality of life.”
“Survival is of upmost importance to me.”
Quality of Life
Quality of life was routinely cited by patients as a key factor in making treatment decisions. In our 2017 Survey, 92% of those who responded revealed that quality of life was important or very important to them when considering treatment options. More specifically, 91% and 67% of patients who replied indicated that mobility and productivity, respectively, were important or very important considerations when making decisions regarding treatment options.
This concern was reiterated anecdotally:
“Making sure I have some quality of life so I can [spend] as much time with my kids and family I don't want them to watch me suffer[.]”
“Trying to balance the most effective treatment regime with the least impact on my day to day living/quality of life. Maintaining a certain level of independence is important to me.”
[Q]uality of life is more important to me than quantity. I want what time I have left to be somewhat of a life. I don't want to spend the whole time being so sick that I am incapacitated[.]”
“Always quality of life. If I am to suffer greatly then no that is not what I want.”
“[…] If I can't have quality of life in the way that allows me to actively participate in my grandchildren's lives, then it's not worth the suffering. For what? I can only watch and look pathetic which makes them sad. I have quality of life at present, so I can tell you with certainty (in knowing the difference) that I would stop or limit my treatment.”
“That [I] can still enjoy my life[.]”
Patient willingness to tolerate treatment side effects: In our 2012 Metastatic Patient and Caregiver Survey, the responses to what level of side effects and how much impact on one’s quality of life would be worth extending progression-free disease by six months was shown to be determined at the personal level. When asked to rate how much impact different symptoms of cancer and cancer treatment would be considered tolerable:
Almost two-thirds of patients indicated that when it comes to fatigue, nausea, depression, problems with concentration, memory loss, diarrhea and insomnia, some or a moderate impact on one’s quality of life would be considered acceptable, and approximately one quarter of patients indicated that a strong or debilitating impact would be considered acceptable.
70% of patients indicated that when it comes to pain, some or a moderate impact on one’s quality of life would be considered acceptable, and 27% of patients indicated that a strong or debilitating impact would be considered acceptable.
Similar responses were also found in our 2017 Survey. Of those who responded in relation to what side effects would be acceptable in exchange for 6 months or less of benefits from breast cancer treatment:
The majority of patients indicated that nausea, depression, memory loss, lack of concentration, diarrhea, pain, and insomnia would be somewhat acceptable side effects.
When it comes to fatigue and hair loss, the majority (48% for both) indicated that they would be very acceptable side effects.
35% considered vomiting as a side effects side effects would be somewhat acceptable and 16% considered it to be very acceptable.
The financial burden of treating and managing breast cancer: The financial burden associated with living with advanced breast cancer extends far beyond any loss of income during a temporary or permanent absence from employment. In addition to the loss of income during illness, metastatic breast cancer patients can incur substantial costs associated with treatment and disease management. Research on the financial impact of breast cancer on patients identified the following (Janet Dunbrack, Breast Cancer: Economic Impact and Labour Force Re-entry. Canadian Breast Cancer Network, 2010):
80% of breast cancer patients report a financial impact due to their illness.
44% of patients have used their savings, and 27% have taken on debt to cover costs.
These findings were consistent with the responses in our 2012 Survey:
Nearly one-third of patients indicated that the cost of medication, the cost of alternative treatments (i.e. massage, physiotherapy, etc.) to manage symptoms and side effects, and the time required to travel to treatment had a significant or debilitating impact on their quality of life.
24% of patients indicated that the costs associated with travel had a significant or debilitating impact on their quality of life, and 41% of patients indicated that it had some or moderate impact on their quality of life.
In our 2017 Survey, the majority of metastatic breast cancer patients reported that their diagnosis had some (50) or a very large (53) impact on their finances. More specifically, of those who responded:
38% indicated that the costs of prescription medications had some impact on their quality of life, and 13% indicated that it had a significant impact on their quality of life.
The majority (41%) reported that the cost of other treatment had some impact on their quality of life, and 23% indicated that it had a significant time on their quality of life.
The cost of travel to treatment had some impact on the quality of life of 39% of patients and had a significant impact on the quality of life of 16% of patients.
9% indicated that the cost of cancer medications has stopped them from taking their medications and another 9% indicated the same for support medications.
Patients in the 2017 Survey also reported on the difficulties and unease of accessing cancer and support medications. Of the 101 individuals who indicated that they had private insurance at the time of diagnosis, 24% had challenges access their private insurance and another 24% had a private insurance claim denied. Concerns with the financial impacts of a metastatic breast cancer diagnosis as well as about accessing treatment was discussed more anecdotally:
“Many of the next step treatments are very expensive [and not covered by government programs] and it is a HUGE struggle to get [coverage]. […] When dealing with an incurable disease the last thing you want to have to do is spend time on a letter writing campaign to argue about whether or not you should receive the drugs [recommended by your physician]. At about $1500.00 a week, I don't know many who can afford that.”
“I have concerns that the next best line of treatment won't be available here.”
“Just because I am not in the lowest income bracket does not mean I don't need assistance. I am excluded from all programs I have tried to access.”
“Always a concern as you never know if the next drug will be covered or how long it takes to get approval from private coverage. Many times it delays treatment and this weighs on one's mind.”
“When I turn 65 I will no longer have private insurance. I will not be able to afford the medication I currently take never mind any future medication that I may require[.]”
“When medications aren't covered it is a tremendous financial burden especially when I wasn't working[.]”
Other financial and accessibility barriers that metastatic breast cancer patients mentioned include: not qualifying for insurance at work, not having access to certain treatments because of where they live, running out of treatment options in the future if their cancer progresses, inability to change employers due to loss of insurance, the prohibitive cost of new treatment options, and much more.
Patient Access to Local Resources and Supports During Treatment
When living with cancer, many patients experience significant barriers and challenges around availability of health care services and quality childcare in their community. In response to the 2012 Survey questions about the availability of supports such as childcare, transportation and alternative treatments in their community:
Among patients with children or other dependents, 53% indicated that there is minimal or no access to appropriate care for their loved ones when they are experiencing debilitating symptoms related to their cancer, and 40% identified barriers to accessing quality care during cancer treatment.
Similar results were found in our 2017 Survey among mBC patients with children at the time of their diagnosis:
25% reported that finding appropriate care for their children/dependents when experiencing side effects of cancer treatments was not accessible.
28% patients indicated that finding appropriate care for their children/dependents during cancer treatment was not accessible.
Among all metastatic breast cancer patients in our 2017 Survey who responded to the prompt, 15% indicated that accessing symptom management options in or around their community was not accessible and 19% reported the same regarding accessing transportation to appointments and treatment.
Patient Willingness to Tolerate Risk
When asked in the 2012 Survey about their willingness to tolerate risk with a new treatment:
34% of respondents were willing to accept serious risk with treatment if it would control the disease
45% of respondents were willing to accept some risk with treatment
21% of respondents were very concerned and felt less comfortable with serious risks with treatment
Need for Personal Choice
The open-ended questions and the key informant interviews showed that it is imperative that women with metastatic breast cancer have access to, and the option of what drugs they take. Most patients are well aware of the adverse effects of treatment up front and they want to make a personal choice that works for them. 32% percent of metastatic breast cancer patients in our 2017 Survey expressed being somewhat comfortable and 68% expressed being very comfortable with participating in treatment decisions. Metastatic breast cancer patients expressed the need for personal choice, autonomy, and being part of their healthcare team in our 2012 Survey as well as in the 2017 Survey anecdotally:
“I think patients (ESPECIALLY young patients) should be given more decision-making power in terms of access to radical treatments to control disease. […] With two small [children] I am determined to access any treatment that can extend my life and I hate struggling with doctors for this access.” – 2012 Survey
“I believe that I would prefer to tolerate severe restrictions in the quality of my life, if it meant that I would be able to have a longer period without progression.” – 2012 Survey
“It would be nice to have more choices and more information about them. I was lucky to get on a clinical trial perhaps because my oncologist was a research oncologist and involved in many. While I knew friend and acquaintances that had Stage IV BC and never informed of clinical trials, and sadly several did not survive the disease.” – 2017 Survey
“I am frustrated that ALL the treatment choices aren't given to me...I am told what I am taking next with no option or discussion on other options. My oncologist has assured me there are many treatments available, but have never shared which, so I have to turn to Facebook groups for guidance.” – 2017 Survey
“All the information about treatment options comes from your oncologist, how do patients know about other options if they are not told about them? Would I get the same treatment or access to immunotherapy if I am treated at a larger cancer centre?”
Improved Outcomes
For patients with metastatic breast cancer that are NTRK+, extension of progression-free survival is of critical concern. Like any other treatment for metastatic breast cancer, patients have an expectation that entrectinib (Rozlytrek) will extend their PFS with good quality of life. Entrectinib is a therapy for NTRK+ fusions that inhibits tyrosine kinase activity such as cancer cell transformation, proliferation, migration and invasiveness. It is also able to penetrate and stay in the CNS (Bazhenova, L. et al. TRK fusion cancer: Patient characteristics and survival analysis in the real-world setting. Targ Oncol. 16, 389–399 (2021)). Studies have shown that entrectinib has the ability to achieve target inhibition and tumour shrinkage with substantial exposure in the CNS (Rangaraju, S. et al. Preclinical and clinical efficacy of entrectinib in primary and metastatic brain tumours harboring NTRK, ROS1, or ALK gene fusions. Neuro-Onc. 19(3), iii106 (2017)). An analysis of three phase I and II clinical studies (ALKA, STARTRK-1, and STARTRK-2) showed that entrectinib had both systemic and intracranial efficacy in patients with NTRK+ fusions with solid tumours (Bazhenova, L. et al. TRK fusion cancer: Patient characteristics and survival analysis in the real-world setting. Targ Oncol. 16, 389–399 (2021)). Data from these studies showed an ORR (objective response rate) of 57.4%, a median duration of response (DoR) of 10.4 months and a median PFS of 11.2 months and intracranial responses were shown in 6 out of 11 patients with baseline CNS metastases (Ibid).
Amongst adult patients with NTRK+, TRKi-naïve solid tumours, entrectinib was associated with deep and durable response and well as long survival (Ibid). Within this study, an efficacy population of 121 patients with 14 different tumour types, including 7 with breast cancer, who were treated with entrectinib had an ORR of 61.2%, 95% CI [51.9, 69.9]. Among the 7 breast cancer patients, the ORR was 71.4%, 95% [29.0, 96.3]. In the larger population, the median DoR was 20 months, 95% CI [13.0, 38.2] and for just the breast cancer patients, the median DoR was 12.9 months, 95% CI [4.2, NE]. The intracranial response rates for patients treated with entrectinib was also promising. For baseline CNS metastases among 19 patients, an intracranial ORR of 52.6%, 95% CI [28.9, 75.6] was found; the median intracranial DoR was 17.2 months, 95% CI [7.4, NE]; and the median intracranial PFS was 10.1 months, 95% CI [6.3, 26.7]. For measurable baseline CNS metastases among 11 patients, an intracranial ORR of 63.6%, 95% CI [30.8, 89.1] was found; the median intracranial DoR was 22.1 months, 95% CI [7.4, NE]; and the median intracranial PFS was 19.9 months, 95% CI [5.9, NE].
Standard of Care and Improved Outcomes
An intrapatient analysis (Krebs, M.G et al. Intrapatient comparisons of efficacy in a single-arm trial of entrectinib in tumour-agnostic indications. ESMO Open. 6(2), 1-9 (2021)) was conducted to investigate the efficacy of entrectinib versus standard of case in rare NTRK+ solid tumours. Within this study, patients with NTRK+ solid tumour from the STARTRK-2 study were grouped according to prior systemic therapy and response. Patients with NTRK+ solid tumours (n=71) belonged to one of three groups: those with documented progression on prior therapy (n=38), those with no documented progression on prior therapy (n=13), and those with no prior systemic therapy (n=20). Prior therapy type included the following: chemotherapy, chemotherapy plus chemotherapy maintenance, chemotherapy plus monoclonal antibody, chemotherapy plus targeted therapy, hormone therapy plus targeted therapy, immunotherapy plus targeted therapy, immunotherapy, and targeted therapy. The main analyses of this study included comparing the PFS of patients on entrectinib versus the time-to discontinuation (TTD) on prior therapy, as well as the Growth Modulation Index (GMI), which is the ration of PFS on entrectinib to TTD on recent prior therapy. A GMI ratio is 1.3 or more is said to indicate clinically significant efficacy.
Results from this study demonstrated that when treated with entrectinib, the PFS (median PFS of 11.2 months) and TTD (median TTD of 9.9) was longer than recent prior therapy TTD (median TTD of 2.9 months, 95% CI [2.0, 4.9]. 25 patients (65.8% of patients) with documented progression on the most recent prior therapy had a GMI of ≥1.3 which indicates that entrectinib has clinically meaningful benefits. Data from this intrapatient analysis also show that there in comparable effectiveness of entrectinib compared to the standard of care among patients with rare NTRK+ solid tumours.
Patient Reported Outcomes
STARTRK-2 (Paz-Ares, L. et al. Patient-reported outcomes from STARTRK-2: A global phase II basket study of entrectinib for ROS1 fusion-positive non-small-cell lung cancer and NTRK fusion-positive solid tumours. ESMO Open. 6(3), 100113 (2021)) is an open-label, multicentre basket study investigating treatment outcomes on patients with NTRK1/2/3, ROS1 or ALK fusion-positive solid tumours on entrectinib. Patients with NTRK+ solid tumours in the STARTRK-2 study were administered quality of life questionnaires in order to analyse patient reported outcomes while being treated with entrectinib. Results from the various questionnaires showed that cognitive functioning was maintained at or just below a baseline level of 83.9 while on entrectinib (mean change from baseline of +0.8 to -7.6). Physical functioning was reported to be maintained at a baseline of 74.8 (mean change from baseline: +7.7 to -0.4). Overall, patient related outcomes indicate that entrectinib has a positive benefit to risk. While on entrectinib, moderate-to-high baseline functioning and health-related quality of life scores were either improved or maintained. Relative to baseline scores, low-to-moderate tumour-related symptom burden remained stable or trended towards clinical improvement. Among all patients, treatment-related symptoms remained stable or improved, with common treatment-related symptoms being absent or of low severity.
Adverse Effects
88 patients in the STARTRK-2 study also reported on treatment related adverse effects (TRAEs), none of which were experienced as “very much” by cycle 12. Some of the reported symptoms include the following:
Diarrhea
At baseline, it was reported as “not at all” by 65 patients, “a little” by 13 patients, “quite a bit” by 3 patients and “very much” by 1 patient
At cycle 12, it was reported as “not at all” by 8 patients, “a little” by 12 patients, “quite a bit” by 2 patients and “very much” by no patient
Nausea
At baseline, it was reported as “not at all” by 65 patients, “a little” by 19 patients, “quite a bit” by 4 patients and “very much” by 1 patient
At cycle 12, it was reported as “not at all” by 19 patients, “a little” by 1 patients, “quite a bit” by 2 patients and “very much” by no patient
Vomiting
At baseline, it was reported as “not at all” by 73 patients, “a little” by 8 patients, “quite a bit” by no patient and “very much” by 1 patient
At cycle 12, it was reported as “not at all” by 18 patients, “a little” by 4 patients, “quite a bit” and “very much” by no patient
Appetite loss
At baseline, it was reported as “not at all” by 46 patients, “a little” by 24 patients, “quite a bit” by 8 patients and “very much” by 4 patients
At cycle 12, it was reported as “not at all” by 18 patients, “a little” by 4 patients, “quite a bit” and “very much” by no patient
In summary, all patients had at least one adverse effect, 75% had an adverse effect that was grade 3 or higher, 39.8% had a TRAE at a grade 3 or higher, and 52.3% of patients had a serious adverse effect. TRAEs leading to dose reduction occurring in 31.8% of patients and in 4.5% of patients, TRAEs led to discontinuation. Adverse effects of any grade that were associated with an impairment on patients’ quality of life included constipation (45.5% of patients), diarrhea (38.6% of patients), dyspnoea and nausea (25% of patients), vomiting (19.3% of patients), decreased appetite (10.2% of patients) and dyspnoea exertional which occurred in 2 patients (2.3%).
When entrectinib was administered at a median dose intensity of 91.3% in a NTRK+ safety population which included 193 patients, adverse events reported in 10% or more of the population included dysgeusia (35.2% of NTRK+ patients), diarrhea (31.1% of NTRK+ patients), fatigue and weight increase (27.5%), constipation and blood creatine increase (25.9%), and dizziness (24.9%) (Bazhenova, L. et al. TRK fusion cancer: Patient characteristics and survival analysis in the real-world setting. Targ Oncol. 16, 389–399 (2021)). This shows that entrectinib demonstrates durable overall and intracranial responses regardless of CNS status at baseline and suggests that entrectinib can address many of the unmet needs of CNS-active treatment among patients with NTRK+ solid tumours.
Impact of Treatment Options to Patients
By delaying the progression of the disease, this treatment can relieve cancer-related symptoms, and improve a patient’s quality of life. When living with no or with minimal cancer-related symptoms, and with minimal side effects from treatment, patients are able to reduce the impact of cancer on their ability to care for children and dependents, continue with their employment and earn income, spend time with loved ones and participate in their life in a meaningful way by engaging in social activities, travelling, maintaining friendships, and pursuing personal interests.
Value to Patients
The value to patients of extending the time that their cancer is progression-free cannot be overestimated. Patients living with metastatic breast cancer are aware that their advanced disease will progress with worsening symptoms until death, and embrace opportunities to try new treatments, even if benefits may be as little as a six-month extension of progression-free disease. It is also very important for patients to have good quality of life when receiving treatment for metastatic disease. Patients that we speak to on a regular basis acknowledge the importance to have the energy to attend their children’s activities and to spend time with family and friends.
Experience With Drug Under Review
Given the rarity of this particular genetic mutation and that this treatment is not widely accessible in Canada, CBCN was unfortunately unable to connect with, and interview, breast cancer patients with experience on the treatment.
Companion Diagnostic Test
At this time, NTRK gene fusion testing is not implemented routinely in breast cancer care in Canada. While NTRK gene fusions are rare overall in cancer, and particularly so in breast cancer, accessing testing and treatment is of great importance for NTRK positive breast cancer patients. It is essential for optimal patient health outcomes that access to testing is facilitated and reimbursed appropriately, so as to prevent additional barriers for patients to access necessary and beneficial therapies.
Anything Else?
Not applicable.
Conflict of Interest Declaration — Canadian Breast Cancer Network
To maintain the objectivity and credibility of the CADTH CDR and pCODR programs, all participants in the drug review processes must disclose any real, potential, or perceived conflicts of interest. This Patient Group Conflict of Interest Declaration is required for participation. Declarations made do not negate or preclude the use of the patient group input. CADTH may contact your group with further questions, as needed.
Did you receive help from outside your patient group to complete this submission? If yes, please detail the help and who provided it.
CBCN did connect with the manufacturer, Hoffmann-La Roche Ltd., to identify clinicians that could connect us with patients with experience on the treatment.
All other research, interviews and outreach to patients was conducted independently by the Canadian Breast Cancer Network, as was the compilation of information and data for the writing of this submission.
As a member of the Canadian Cancer Action Network, the Canadian Breast Cancer Network is committed to adhering to the Code of Conduct Governing Corporate Funding.
Did you receive help from outside your patient group to collect or analyze data used in this submission? If yes, please detail the help and who provided it.
No. The Canadian Breast Cancer Network compiled and wrote this submission independently.
List any companies or organizations that have provided your group with financial payment over the past two years AND who may have direct or indirect interest in the drug under review.
Table 1: Conflict of Interest Declaration for Canadian Breast Cancer Network
Company | $0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 |
|---|---|---|---|---|
Hoffman-LaRoche | — | — | X | — |
Colorectal Cancer Canada
About Colorectal Cancer Canada
Colorectal Cancer Canada is registered with CADTH. www.colorectalcancercanada.com
Information Gathering
To help capture the patient perspective on the drug under review, Colorectal Cancer Canada launched an online patient/caregiver survey from December 7, 2021 to February 8, 2022 of which 6 patients responded. The survey was targeted to any adult TRK gene fusion positive cancer patient (stage I through IV) and/or their caregiver. Because of the small population of patients on Entrectinib (Rozlytrek), CCC was open to receiving patient input from patients/caregivers who are under the treatment of Entrectinib (Rozlytrek) or Larotrectinib (Vitrakvi), both NTRK inhibitors. Data was gathered from patients across Canada, the United States, and Indonesia. The survey was posted on the social media platforms of Colorectal Cancer Canada and disseminated by the Canadian Cancer Society across pan-tumour forums and panels. As a result of this outreach, six patients provided detailed and high quality responses to our questions:
The data on patient demographics is summarized and represented in Table 2 and will serve as the basis for this submission.
Table 2: Surveyed Patients – Information Gathering
Criteria | Patient 1 | Patient 2 | Patient 3 | Patient 4 | Patient 5 | Patient 6 |
|---|---|---|---|---|---|---|
Connection to Cancer | Patient undergoing treatment | Patient undergoing treatment | Patient undergoing treatment | Patient undergoing treatment | Patient undergoing treatment | Patient undergoing treatment |
Country and Region | Canada, British Columbia | USA, Illinois | USA, Pennsylvania | Canada, Manitoba | USA, Utah | Indonesia, Central Java |
A. Gender B. Age at Dx | A. Female B. 60-69 years | A. Female B. 40-49 years | A. Female B. 50-59 years | A. Female B. 40-49 years | A. Male B. 30-39 years | A. Female B. 30-39 years |
Date of Dx | 05/2011 | 03/03/2020 | 2004 | 01/2016 | 11/2020 | 10/10/2021 |
A. Stage at Dx B. Current Stage C. Type of Cancer D. Metastases | A. Stage II B. Stage IV C. Thryoid D. Brain/Central Nervous System, lung, thyroid, bones | A. Stage IV B. Stage IV C. Neuroendocrine Tumours D. Brain/Central Nervous System, vertebra | A. Stage I B. Stage IV C. Thyroid | A. I don’t know B. Stage IV C. Sarcoma D. Lung, Tricep, Pectoral, Thigh, Pancreatic tail | A. Stage III B. Stage IV C. Undifferentiated pleomorphic sarcoma D. Lung | A. Stage III B. Stage III C. Colorectal |
Disease Experience
Patients were asked if any cancer-induced symptoms were experienced prior to diagnosis. Three out of six patients (50%) had experienced symptoms. Patients reported pain, fatigue, cough, shortness of breath, and mobility as the symptoms which were more important to control than others. Aside from Patient 3, all patients felt that their symptoms affect their daily life. Patients felt symptoms affected their work-life, daily activities and the ability to exercise:
“I work 15 hr a week and get lots of help in shopping, cleaning, cooking…” (Patient 1)
“Extreme, rapid weight gain from NTRK inhibitors has affected physical, mental, emotional health, and social life.” (Patient 2)
“Only working 0.4FTE now, not active re: exercise and sports anymore, need at least 8 hours sleep to function properly” (Patient 4)
“I wasn’t able to work initially but when I switched to Vitrakvi I was able to go back. Made it easier to deal with cancer knowing I was still providing for my family.” (Patient 5)
“Unable to work, but I can take care of myself” (Patient 6)
Additionally, symptoms had a psychological impact on all patients including, 50% of patients expressing the fear of dying early, and being depressed.
“Being burden to my children, not able to work to provide for myself, dying early.” (Patient 1)
“Weight gain has alienated me from friends (85% of which I know through volleyball). Depression makes it hard to work daily.” (Patient 2).
“Stress” (Patient 3).
“Fear of not being able to watch my kids grow up, worry about finances, fatigue causes overreactions in situations” (Patient 4). “Knowing I might not be around in a few years has definitely depressed a few in my family.” (Patient 5).
“Sad, depressed” (Patient 6).
Aside from Patient 6, all patients rated access to new effective treatments for cancer as very important (10), on a scale of 1-10.
Experiences With Currently Available Treatments
All patients accessed previous therapies for the treatment of their cancer prior to current targeted therapy (Entrectinib or Larotrectinib). Therapies included chemotherapy, radiation therapy, radioactive iodine, and surgery. Patients 2 and 4 also received targeted therapy as previous therapy. Exceptionally, Patient 2 received the NTRK inhibitor Larotrectinib (Vitrakvi) prior to current therapy Entrectinib (Rozlytrek).
For two out of six patients (33%), their previous therapies were not able to control their symptoms, and for one out of six patients (16%), the therapies were able to only partially control them. Patients were questioned what side effects were most difficult to tolerate from their previous therapies and patients reported common side effects such as diarrhea, nausea, hair loss, vomiting, and fatigue.
Three out of six patients (50%) had to pay out of pocket for their previous treatments. Those patients did not receive financial assistance from a pharmaceutical/biotech company assistance program or any other assistance program. Patient 2 and Patient 4 received financial assistance, with 100% of the total cost of the treatment being provided. In addition to treatment cost, Patients 3 and Patient 6 noted travel as an additional expense incurred by accessing their treatments.
Paying out of pocket to access new drug therapies is dependent on the cost for all patients. For Patient 4, this wouldn’t be an option: “While loving is important, leaving my family with excessive debts would be a burden to me”.
Improved Outcomes
Aside from Patient 5, all patients highlighted the increased importance for new therapies to bring about improvement in their physical condition and quality of life. Trade-offs considered when choosing therapy include both extended overall survival and quality of life. For instance, all patients and except Patient 5 would take a drug that has been proven to provide better QoL during their lifetime even if it does not extend overall survival. They feel the need to be able to carry on social activities without the burden of the therapies side effects that aggravate their quality of life. Patient 1 would be willing to tolerate significant side effects in order to extend survival by just 2 months, and Patients 1 and 6 would be willing to do so to extend survival by 1 year.
Patient 1 from Canada and Patients 3 and 5 from the U.S. report that access to drug therapies in their respective countries is very appropriate/fair. Whereas, Patient 4 from Canada and Patient 6 from Indonesia rated access to drug therapies as limited/restrictive. Lastly, all patients find it very important to have a choice along with their physicians in deciding which drug to take.
Experience With Drug Under Review
Entrectinib (Rozlytrek): Two out of six patients (33.3%) are currently under treatment with the drug under review (Entrectinib). Patient 1 received the drug as second line therapy, and Patient 2 received the drug as third line therapy, after receiving the NTRK inhibitor Larotrectinib for four months. While Patient 1 was informed of the drug by her oncologist as a potential treatment option from the start of their discussion, Patient 2 was not and had first heard about Entrectinib (Rozlytrek) from the internet. Both Patients 1 and 2 had access to the drug under review via clinical trials. They had no financial restrictions when accessing the drug and did not experience issues accessing therapy.
Common side effects experienced while on the drug include cough, constipation, diarrhea, swelling of ankles/feet/hands, weight gain, and vomiting. When questioned about the most difficult aspects of the drug, Patients 1 and 2 both mentioned the management of side effects. However, according to Patient 1: “The side effects are more manageable. Easier to use than other treatments”. Patient 2 also mentioned difficult aspects such as the “hours spent in medical appointments”, “lifestyle changes”, the “inability to plan ahead”, “feeling isolated (difficulty connecting with friends, geographical remoteness)”, “impact on career”, “emotional drain”, “anxiety/worrying”, and the “feeling of helplessness”.
Patient 2 believes that an unmet patient need with current therapies that Entrectinib (Rozlytrek) can help alleviate include “[getting] through blood brain barrier”. Patients were also questioned on what they expect, or hope, that Entrectinib will have on the cancer and their prognoses, both patients expect it can “maintain or improve quality of life”, and “increase overall survival”. Patient 1 also expects it can “delay onset of symptoms”, “reduce side effects from current medications or treatments” and appreciates its “ease of use”. Patient 2 hopes it can “delay need for chemotherapy”. Additionally, Patient 1 who is presently on the drug rates her quality of life as an 8 on a scale of 1-10 with 1 being “low/severely impacted” and 10 being “high or normal living”. Patients were asked if the drug under review allowed them to fulfill or accomplish anything that they would not have otherwise been able to, had they not accessed the therapy. Patient 1 expressed that the drug allowed them to resume all their daily activities: “Mobility is much better, and my life is more active!”. Patient 1 also mentioned: “majority of symptoms were easy to manage and gone in three weeks”. Both patients appreciate the easily administered oral therapy and find it simple to integrate in their daily routine:
“It is easy and simple to take, not many side effects, I am not taking it long enough, but I feel good and have big hopes in helping with my cancer” (Patient 1).
Compared to other treatments, patients rated their overall experience with the drug under review as 6 (Patient 2) and 9 (Patient 1) on a scale of 1- 10, with 1 being “much worse” and 10 being “much better”.
Larotrectinib (Vitrakvi): Five out of six patients (83.3%) have undergone treatment with the NTRK inhibitor Larotrectinib (Vitrakvi). This includes Patient 2 who is currently on Entrectinib (Rozlytrek) but has taken Larotrectinib as previous therapy. While we recognize that these patients have not taken the drug under review (Entrectinib), they have responded to the survey in order to best describe their experience with an NTRK inhibitor. Patient experiences with drugs of the same biomarker can help highlight the promising results for other NTRK inhibitors such as the drug under review.
Patients 2, 3, 4, 5 and 6 received the Larotrectinib (Vitrakvi) drug as second line, first line, third line, second line and third line therapy, respectively. Patient 4 currently receives this drug in combination with radiation therapy. Only Patient 3 received this therapy under clinical trial in the U.S.
Common side effects experienced include constipation, joint pain, muscle pain, weight gain, and trouble breathing. When questioned about the most difficult aspects of the drug, most patients voiced a common opinion which included: management of side effects, impact on career, pain and fatigue. Patient 2 also reported social issues, mental issues, lifestyle changes and inability to plan ahead as difficult aspects:
“Being isolated from my social network from not being able to play volleyball anymore from the weight gain” (Patient 2).
When asked if there is a particular gap or unmet patient need with current therapies that Larotrectinib can help alleviate, patients expressed that it:
“Avoid unnecessary harmful treatments” (Patient 3)
“Side effects are minimal compared to chemo or radiation. There needs to be more genomic testing of turnouts so that targeted gene therapies are offered to patients earlier or so that they can start the drugs sooner after metastasis has occurred.” (Patient 4).
Patients 3, 4, 5 reported that the Larotrectinib drug was able to fully shrink/control their cancer and/or spread of the disease to other organs. Patient 2 reported that the drug was able to partially do so. Patients were also questioned on what they expect, or hope, that Larotrectinib will have on the cancer and their prognoses, all patients expect it can: "maintain or improve quality of life” and "increase overall survival". Patient 4 also expects it can “prevent further growth or spread of cancer”.
All patients expressed that the drug allowed them to resume all their daily activities. While patients 3 and 4 rate their quality of life as “high or normal living”, 9 (on a scale of 1-10), patients 2 and 5 rate it as low as 4 and 5 respectively. Patients were asked if the drug under review allowed them to fulfill or accomplish anything that they would not have otherwise been able to, had they not accessed the therapy. They expressed that:
“I lived long enough to be moved to a better treatment” (Patient 2). Patient 2 is currently on Entrectinib after Larotrectinib stopped working.
“Life! Who knows where I would be without it!” (Patient 3)
“Yes. Extended my health for 1.5 years before cancer mutated. Not in clinical trial for Selitrectinib after 2 years on Larotrectinib” (Patient 4)
“Able to live normally and not worry as much that I’ll die in months” (Patient 5)
Similar to Entrectinib, all patients appreciate the easily administered oral therapy and find it simple to integrate in their daily routine. When asked if they believe Larotrectinib (Vitrakvi) will change their long-term health and well-being for the better, Patients 3, 4, and 5 responded with a positive outlook:
“I have been stable for 3+ years” (Patient 3).
“Extending life with minimal side effects.” (Patient 4).
“Maybe not cure me but able to prolong my life while letting me live normally” (Patient 5).
Aside from Patient 2, who changed regimens after Larotrectinib stopped working, all four patients rated their overall experience with the drug under review as 10 “much better” on a scale of 1-10, with 1 being “much worse” and 10 being “much better”.
Patient 2 however still expresses that Larotrectinib allowed her to “[live] long enough to be moved to a better treatment”, that treatment being Entrectinib.
Companion Diagnostic Test
Patients were asked if they were aware that biomarkers can help determine a specific treatment option prior to their diagnoses, and only Patient 3 was aware. Similarly, patients were asked whether their oncologist or any other member of their medical team explained biomarker testing before treatment started, in which only Patient 2 responded “yes”.
Four out of six (66.7%) patients had one or more biopsies to further investigate the make-up of their tumour. Three out of six (83.3%) patients confirmed they tested positive for the unique biomarker, NTRK, and five out of six (83.3%) patients had their biomarker testing done after diagnosis which emphasizes the need to adopt biomarker testing as a standard practice at diagnosis. Following biomarker testing, Larotrectinib (Vitrakvi) was the first treatment that oncologists selected for Patients 2 and 4.
“Having genomic testing is key. Having choice of drugs is important” (Patient 4).
Anything Else?
The six patients provide evidence that NTRK inhibitors such as Entrectinib and Larotrectinib allow patients to have an improved quality of life and overall survival compared to previous standard therapies. Patients’ positive experiences with NTRK drugs propels oncology toward the goal of precision medicine that can greatly enhance cancer patients’ lives and provides evidence of the promising future of targeted therapies.
Specifically, according to two patients (Patient 1 and 2), Entrectinib can maintain quality of life, increase overall survival, and delay onset of symptoms with tolerable and manageable side effects. The drug under review therefore serves as an effective example for treatment based on tumour biomarkers (specifically NTRK) rather than tissue-specific status.
When asked about why access to Entrectinib (Rozlytrek) is so important, Patient 1 expressed that the drug enabled her “mobility and [her] life is more active” compared to previous therapy:
“I was two and a half years using Lenvatinib.I had to stop the medication after 3 weeks (for a week) to heal the sores on my feet, that prevented me from walking for a week to 19 days”
Patients also provided compelling comments on why targeted therapies should be accessible to patients with the NTRK biomarker:
“[Entrectinib] is easy and simple to take, not many side effects, I am not taking it long enough, but I feel good and have big hopes in helping with my cancer” (Patient 1).
“Larotrectinib is a miracle drug” (Patient 3).
Patients also highlighted the importance of equitable access to biomarker testing, which would allow patients to directly benefit from precision medicine by receiving the specific treatment targeted to their tumour profile.
“Larotrectinib is a game change in the course of my cancer journey; only wish I would have had biomarker testing earlier so I could have avoided unnecessary harmful treatments” (Patient 3).
“Genomic testing needs to become a standard in Canada for all new tumours, especially when rare like mine” (Patient 4).
CCC also had the privilege to receive input from Patient 4, a young woman who received Larotrectinib after testing positive for NTRK. She shares her story here: http://sarcomacancer.ca/story/monique
“Within a week of starting the new drug, my pain was significantly reduced, my energy was improved, and I was able to attend a Winnipeg Jets game with my family. After one month, my CT scans had showed shrinkage, I was completely weaned off my pain meds and by January my CT scan showed further shrinking. I went snowboarding with my kids for New Years and I was able to return to work on part time basis mid-January. Having my independence back and feeling well enough to return to work part time is something that is very important to me. I have worked hard for my career, my returning to work allows me to feel like things are more normal.”
Based on the objective research carried out as represented herein, Colorectal Cancer Canada strongly urges that a positive funding recommendation be issued for Entrectinib for the treatment of patients with the NTRK biomarker. We believe it is essential to provide these patients equitable access of such an effective drug that improves their quality of life and outcomes as well as the impact on their families, unaccompanied by any financial restrictions. Providing molecularly targeted therapies that are easily administered with minimal side effects, and permit patients to carry on normal lives is fundamental for basic and high-quality care in Canada.
“We need more drugs to be subsidized by the Health Canada and our provincial drug plans in order to make these kinds of drugs more accessible for cancer patients” (Patient 4).
Conflict of Interest Declaration — Colorectal Cancer Canada
To maintain the objectivity and credibility of the CADTH CDR and pCODR programs, all participants in the drug review processes must disclose any real, potential, or perceived conflicts of interest. This Patient Group Conflict of Interest Declaration is required for participation. Declarations made do not negate or preclude the use of the patient group input. CADTH may contact your group with further questions, as needed.
Did you receive help from outside your patient group to complete this submission? If yes, please detail the help and who provided it.
No
Did you receive help from outside your patient group to collect or analyze data used in this submission? If yes, please detail the help and who provided it.
No
List any companies or organizations that have provided your group with financial payment over the past two years AND who may have direct or indirect interest in the drug under review.
Table 3: Conflict of Interest Declaration for Colorectal Cancer Canada
Company | $0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 |
|---|---|---|---|---|
Abbvie Corp | — | X | — | — |
Amgen Canada | — | — | — | X |
AstraZeneca Canada | — | — | — | X |
Bayer Inc | — | — | X | — |
Boehringer Ingelheim Ltd | — | — | X | — |
Bristol Myers Squibb Canada | — | — | — | X |
Eli Lilly Canada | — | — | X | — |
GlaxoSmithKline | — | — | X | X |
Hoffman-La Roche | — | — | — | X |
Janssen Inc | — | — | — | X |
Merck Canada Inc. | — | — | — | X |
Novartis Pharma Canada | — | — | X | — |
Pfizer Canada | — | — | — | X |
Elevation Oncology | — | — | X | — |
Pendopharm | — | — | X | — |
Taiho Pharma Canada | — | — | — | X |
Lung Cancer Canada
About Lung Cancer Canada
Lung Cancer Canada is a registered national charitable organization that serves as Canada’s leading resource for lung cancer education, patient support, research, and advocacy. Lung Cancer Canada is a member of the Global Lung Cancer Coalition and is the only organization in Canada focused exclusively on lung cancer. https://www.lungcancercanada.ca/. Lung Cancer Canada is registered with CADTH.
Information Gathering
Data Collection: The information discussed throughout this submission consists of the thoughts and experiences of patients as collected through interviews Lung Cancer Canada had with them. All interviews were conducted in January to February 2022.
Demographic Data: NTRK gene fusions represent a very small minority of lung cancer patients with non-small cell lung cancer (NSCLC), representing only about 0.23% of the patient population. As such, it was difficult to identify and source patients with this specific biomarker with lung cancer as per the indication of this submission. Entrectinib is also approved for ROS1 indications, which represent a greater minority (about 1-2%) of lung cancer patients; thus, many of the patients interviewed for this submission are ROS1 patients. Nonetheless, all of the patients discussed in this submission have non-small cell lung cancer and have experience with entrectinib.
|||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| |
|---|---|---|---|---|---|---|---|
|||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| |
|||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| |
|||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| |
|||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| |
|||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| |
|||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| |
|||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| |
|||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| | |||||||||| |
Disease Experience
DR had retired 11 years ago back in 2011 just a few months before her first grandchild was born and lived a fairly active lifestyle that allowed her to often travel on family trips to Disney World in Orlando, go on cruises, and vacations around the world with her husband, family, and grandkids. DR has a family history of heart disease, and so when she felt a heart flutter in her chest in 2014, she immediately acted upon it to get it checked, and after the cardiologist noticed a small blockage in her stress test, DR underwent more testing that ultimately revealed she had stage 2 non-small cell lung cancer with a small nodule spread to one of her lymph nodes. Only a year after diagnosis was when DR underwent biomarker testing, which later found that she was positive for NTRK gene fusion, an incredibly rare mutation found in less than 1% of lung cancer cases. It was only through this discovery that DR was able to access entrectinib, which is one of only 2 targeted therapies currently available for those with NTRK in the USA. Once she started treatment with entrectinib in mid 2016, she has seen a lot of success with it and her cancer is almost completely gone. When DR was first diagnosed, her ultimate goal was to be able to live long enough so her grandkids would be able to remember her, and now almost 8 years later, she is doing very well, living a full worthwhile life, and her cancer is essentially resolved now with no symptoms. Entrectinib has the potential to give many other patients in Canada the same impact as it did with DR.
In August 2020, MR was just about to start her 20th year of being an elementary school teacher in the US while also being kept busy with her three young kids, ages 6, 11, and 12 also in elementary and middle school. She had always been an active individual, especially when in the classroom, and was often found outdoors biking, walking, hiking, and running with her husband and kids, so being diagnosed with lung cancer was never something she thought she would ever hear. Although she also had some risk factors for breast cancer, she did not experience any symptoms of feeling unwell apart from a cough earlier in the year. However during one bike ride, her hip suddenly started to bother her and over the course of the next few days, the pain got to the point where she had a hard time falling asleep. After several tests and scans, doctors found a large tumour in her hip along with several abnormal nodes in her chest, later confirming she had stage 4 NSCLC, positive for the ROS1 mutation. After a rocky year and a half with numerous treatments, she fortunately has shown no evidence of disease (NED) since August 2021 to this day, and continues to maintain her independence, keeping busy with her family, staying very active outdoors, and is even planning to return to work in the classroom as of February 2022. Both DR and MR’s cancer experiences are further detailed in Section 6, but the livelihood and quality of life they both have been able to maintain thanks to entrectinib allows patients like them to return to the activities and hobbies they love, regardless of their cancer diagnosis.
NTRK fusions are extremely rare in lung cancer, representing approximately 0.23% of cases, and therefore there is very limited data on the efficacy of current standard treatments in this population. Although NTRK fusion cancers impact a broad range of malignancies across the population, non-small cell lung cancer patients face additional barriers to accessing treatment for their disease, including stigma and inconsistent access to screening and biomarker testing across the country. With lung cancer being the most commonly diagnosed cancer in Canada in 2021, and as next generation sequence testing is becoming more and more widespread, these NTRK cases are now being identified regularly. There is a clear unmet need to provide therapeutic options for these NTRK NSCLC patients when no other satisfactory options exist, as current standard of care only focuses on systemic treatments that carry a multitude of additional burdens. Entrectinib has the potential to provide an additional treatment option for patients with these advanced tumours met with no satisfactory treatment options left.
Entrectinib is an oral targeted therapy that has been approved in the non-small cell lung cancer setting for both NTRK gene fusions as well as ROS1. In the ALKA-372, STARTRK-1 and ongoing STARTRK-2 phase 1 and 2 clinical trials, there have been very promising results showing clinically meaningful benefit and durability for patients in the NTRK patient population. 7% of participants showed complete response and 50% with partial responses, with a median duration of response at 10 months (Doebele et al., 2020). Patient-reported outcomes from the STARTRK-2 study of entrectinib expressed improvements in physical functioning, disease stability, and symptom burden (Doebele et al., 2020). The day-to-day qualitative impacts that oral targeted therapies like entrectinib have on patients are extremely promising, as will be discussed further in this document. With the limited number of treatment options available for this niche population, adding entrectinib as an additional treatment option will be able to provide patients like DR and MR an opportunity at better survival and quality of life. With these oral targeted treatments, patients are able to live longer, manage their symptoms, be independent, have a good quality of life, and return to enjoying hobbies they love. Patients already have a huge burden coping with their lung cancer diagnosis; the battle to survive this disease should be made easier by ensuring the availability of treatments that work beyond what is already the standard in Canada. Since there is already a comparable oral targeted therapy approved in Canada, Larotrectinib, the approval of entrectinib will ensure there are additional options available in the market for patients who may otherwise be unable to access larotrectinib, and may also increase competition and thus, lowering costs for the drugs. Larotrectinib is currently the only approved targeted therapy for NTRK fusion cancer patients, and this cannot remain the case in the future; additional options are necessary to diversity the market and improve access and cost for cancer patients. We hope that CADTH takes this into consideration.
Experiences With Currently Available Treatments
The current standard of care for patients with NTRK fusion non-small cell lung cancer are primarily systemic treatment options, such as chemotherapy and immunotherapy, both of which have been long-standing options for cancer patients. However, both come with many drawbacks with limited clinical benefit. Entrectinib would serve as an additional option for patients without access to NTRK drugs, or as an alternative to larotrectinib, which has also been approved by CADTH for the treatment of NTRK fusion cancers. LCC foresees that an NTRK inhibitor (entrectinib or larotrectinib) will soon become a standard of care option for these patients soon, as its efficacy, durability, and safety have all pointed towards this positive step forward.
Chemotherapy
Chemotherapy is typically presented as the first line of treatment for patients without known biomarker mutations, and has been a long-standing and well-documented standard of care for cancer patients. It does see some benefits and has been found to work. However, it is limited in its use as a viable long-term treatment option due to its harsh side effects, impact on the individual’s functionality, and increases dependence on caregivers in their daily activities. These have been well documented in previous LCC submissions.
AN was diagnosed in 2014 with stage 3 NSCLC when targeted therapy treatments were still new and very limited in number offered for the treatment of lung cancer. She had spread in both of her lungs, which the physicians classified as locally advanced, but did not have any metastases anywhere else in her body. She started with 6 rounds of cisplatin chemotherapy, which was the standard of care at the time, during which she had no side effects at all and continued to live her life as normal. After chemotherapy, she had a lobectomy to remove her lower right lobe, then followed up with 30 rounds of radiation. These initial treatments had slowly began to shrink her tumours, and AN was NED for several months. However, her cancer came back the following February in 2015, and she was placed on chemotherapy again (pemetrexed/bevacizumab) for nearly 2 years. Just like her first chemotherapy experience, she had virtually no side effects again, and was able to continue physical activity, walking around the city meeting with friends, driving herself to run errands. In late 2016, she started having problems with her insurance coverage of chemotherapy, which is when her oncologist suggested they start her on the entrectinib clinical trial. Fortunately, entrectinib was covered and she remained on it for 4 years.
Throughout the summer of 2021, AP had been battling a cold and towards the first week of September, she had a worsening cough that doctors brushed off as symptoms of pneumonia. By Labour Day weekend, her cough became much worse, the antibiotics were not working, and she woke up with a swollen leg from the knee down, which a trip to the ER confirmed it was blood clot. While she was admitted to the hospital for the next two days, chest scans revealed tumours in her lungs and nodules in her lymph nodes, and was later confirmed to be stage 4 non-small cell lung cancer. She completed one round of chemotherapy before her biomarker testing came back positive for the ROS1 rearrangement. She was then immediately started on entrectinib, which she is still currently on in February 2022.
Within 1.5 weeks after DR was diagnosed in 2014 with stage 2 NSCLC, she had surgery to remove the middle lobe of her right lung right away, and then started chemotherapy 6 weeks later for about 3 months. She was met with a lot of nausea, fatigue and tiredness during chemotherapy, and was too tired to even drive herself, so her husband drove her to hospital appointments most of the time. Her initial tumour was completely gone by the time she finished up chemotherapy, so she continued to live her life as normally as she could and remained disease-free for several months. However, about 6-7 months later in mid-2015, another new nodule had popped up in her upper right lung, so she underwent a 2nd surgery with a successful complete resection. Unfortunately, in 2016, 7 months after her 2nd surgery, a 3rd nodule appeared in the other lung, which had now progressed her to stage 4 lung cancer, so DR underwent biomarker testing at this point, which revealed she had the rare NTRK gene fusion mutation. This had now opened up the door to accessing TKI targeted therapies, so her oncologist started her on entrectinib right away, which she has been on for about 5.5 years ever since.
Radiation
After AN’s initial lobectomy surgery in 2014, she had 30 rounds of radiation done, 5 days a week for 6 weeks. Just like with chemotherapy, she had virtually no side effects from the radiation at all, no pain, fatigue, nausea, or anything, which she considers very lucky. She was able to continue living her life as she did before diagnosis, driving herself to run errands, spending time with her husband, friends, and family, and her physical activity was steady throughout. After these initial chemotherapy, surgery, and radiation treatments, AN was very lucky to have no evidence of disease (NED) for about a year until the next winter in February 2015, when her cancer came back. She was placed on chemotherapy again for nearly 2 years, which worked very well as it did the first time. In November 2016, she started the entrectinib clinical trial, which slowed her disease stability for 5 years while she was on it.
Targeted Therapies
Targeted therapies have recently emerged as a rapidly changing area of cancer treatment, with new developments for NSCLC happening frequently. Larotrectinib is currently the only other form of targeted therapy treatment for NTRK-positive NSCLC and thus, is pinned as the comparator against entrectinib. It has already been approved by CADTH for use in Canada, with similar efficacy rates as entrectinib and mild side effects. As sourced from LCC’s previous larotrectinib submission, patients found success with the treatment, so the approval of entrectinib will allow for additional options on the market for patients who may be unable to access larotrectinib, while also increasing competition and therefore lowering the prices of the drugs.
At diagnosis, NM’s tumour occupied 90 percent of her lungs, she was on oxygen and in a wheelchair. After genomic testing confirmed she had NTRK fusion, she was placed on larotrectinib. Four weeks after beginning treatment, she was able to take her kids the movies and even celebrate her birthday. When NM progressed on chemotherapy, she felt hopeless and was not optimistic she’d find another effective treatment. With the success of larotrectinib in her case, it gave her hope again and allowed NM to continue celebrating milestones with her family.
When initially diagnosed with NTRK-positive NSCLC, KT was told he had 3-4 weeks to live due to the advanced stage of his cancer. Four days after starting treatment with larotrectinib, he was already feeling better and his symptoms including pain, fatigue, weight loss, and incontinence improved, and had virtually no side effects from the drug apart from slight ankle swelling. His first scan after four weeks of treatment showed all the metastases in his body were gone and 65% of the tumour in his lungs had disappeared. As of 2019, KT was stable and had been on larotrectinib for 2 years. His family is absolutely thrilled that he had access to a targeted treatment that not only saved his life, but allowed him a quality of life that does not keep him bedridden, instead allowing him to even return to work, walk around the city, and even go to the gym every day.
Improved Outcomes
There have been many incredible advancements in non-small cell lung cancer research in recent years that have changed the treatment paradigm for patients in Canada. With NTRK fusions being a rare and relatively recent discovery in NSCLC, there has not been many opportunities for the development of new targeted therapy treatments for this population, until now. It is evident that NTRK-targeted therapies, including entrectinib, have been met with incredible success that gives patients their livelihoods back and allows them to plan further down the line for a possible future that previous therapies could not give them. When choosing a therapy, some of the most crucial outcomes that patients want to have include:
Improved management of their symptoms of non-small cell lung cancer
Allowing patients to have a full and worthwhile quality of life
Having manageable side effects
Allowing patients to live longer and maintain their independence and functionality so minimize the burden on their caregivers and loved ones
Delaying disease progression and settling patients into long-term remission for improved survivorship
In the case with entrectinib, since there is a comparable NTRK inhibitor approved in Canada, larotrectinib, entrectinib will serve as an alternative option for patients who have no satisfactory treatment options
Experience With Drug Under Review
Lung Cancer Canada was only able to source one patient with the specific NTRK mutation on entrectinib in Canada due to the narrow scope that NTRK mutations impact amongst lung cancer patients. However, LCC was able to speak to patients with ROS1+ NSCLC on the same treatment, in which entrectinib is also indicated and approved for, and LCC believes there would not be any major differences in experiences between these subgroups of patients with different biomarkers. The details and experiences of these patients are outlined below.
Table 5: Details and Experiences of Patients With Drug Under Review
Patient | Date diagnosed | Biomarker | Drug access method | Period on entrectinib | Duration on entrectinib | Currently on entrectinib? |
|---|---|---|---|---|---|---|
|||||||||| | 2014 | NTRK | Pharmaceutical patient access program | Mid 2016 - present | Over 5.5 years | Yes |
|||||||||| | July 2012 | ROS1 + | Pharmaceutical patient access program, insurance | Sept 2020 - present | 1.5 years | Yes |
|||||||||| | September 2021 | ROS1 + | Insurance | Sept 2021 - present | 6 months | Yes |
|||||||||| | October 2020 | ROS1 + | Insurance | Nov. 2020 – present | Almost 1.5 years | Yes |
|||||||||| | June 2019 | ROS1 + | Insurance | Dec 2019 - present | Over 2 years | Yes |
|||||||||| | August 2020 | ROS1 + | Pharmaceutical patient access program, out-of-pocket | Sept 2020 - present | 1.5 years | Yes |
|||||||||| | August 2020 | ROS1 + | Pharmaceutical patient access program | Aug – Sept 2021 | 2 months | No |
|||||||||| | February 2014 | ROS1 + | Clinical trial | Nov 2016 – Nov 2020 | 4 years | No |
Entrectinib is very effective in treating the cancer and maintaining stable disease
For AP, entrectinib was her first line of treatment right after she was diagnosed and found to have the ROS1 biomarker. She started the treatment in late September 2021 and her first scan in early December, 9 weeks after her 1st visit to the ER that resulted in her diagnosis, revealed an astonishing discovery – her cancer was 90% gone, which was virtually unheard of to her doctors. As of February, she has virtually no evidence of cancer and is feeling like her pre-cancer self.
DR also has a similar story in that she started entrectinib after a tumour popped up for a third time in 2016 and continues to get CAT scans every 8 weeks. For the past 5.5 years she has been on the drug, each scan consistently showed the tumour shrinking more and more every 2 months while on entrectinib until it was no longer visible. As of now, she described her latest scan with a “glass-like opaqueness”, with virtually no nodules or tumours visible in her scans and her cancer was pretty much gone as of February 2022.
CA started entrectinib in December 2019, and within 2-3 months, her doctors saw astonishing reductions in her tumours and her cancer was nearly gone. As of February 2022, over 2 years later, she has been NED since the first couple of months on entrectinib and has been stable since.
At diagnosis in October 2020, AY had no symptoms whatsoever, so she was shocked to learn she had mets all around her body in addition to the main tumour in her lung, including 22 lesions in her brain, thyroid, lymph nodes around her chest and lungs, and bones including her femur and clavicle. She started entrectinib has her first line of treatment, and her first 3-month scan showed excellent response in her lymph nodes and brain metastases were already shrinking. In her most recent scan in late 2021 showed almost all the metastases in her chest and abdomen were completely gone, 1 or 2 lymph node mets left, and her primary lung tumour is slightly smaller.
Compared to NW’s experience and lifestyle while on chemotherapy, she has come a very long way in her quality of life with entrectinib. Being on first-line chemotherapy for 8 years between 2012 and 2020 was very tough for her, and her quality of life due to side effects was a constant cycle of good and bad weeks between each treatment cycle. When she started entrectinib in September 2020, she felt an immediate difference in her quality of life, functionality, and physical symptoms, which are more consistent than it was with other treatments. It was incredibly effective and completely resolved her metastases and secondary tumours, only leaving her primary lung tumour, which has shrunk slightly but otherwise currently stable.
After maintaining NED status for several months after diagnosis until February 2015, AN had scans every 3 months for the 4 years she was entrectinib, which, by the end of her treatment in late 2020, had shrunk her tumour by about 40%.
Entrectinib is a very durable form of treatment
As outlined in the chart above, 6 of 8 of the patients interviewed by Lung Cancer Canada were on entrectinib for over a year, most of whom are still on the treatment to this day as of February 2022, and two patients who have been on entrectinib for 4 years or more, which far exceeds the expectations as outlined in the phase 3 trials. The median duration of response as reported in the trials was about 10.4 months, and median progression-free survival was 11.2 months (Doebele et al., 2020).
The need for a lasting and effective treatment for the NTRK patient population is high, and with this submission’s indication being for patients with advanced/metastatic disease, these patients need a treatment that can maintain disease stability at the least and be effective in shrinking their tumours and metastases. For such patients in late stages of their cancer journey, many of them have rapidly growing mets with worsening symptoms, and with the niche population that NTRK fusion mutations serves in NSCLC, there needs to be treatments available that are effective and durable. Entrectinib evidently meets these needs.
Side effects with entrectinib
CA has been on entrectinib since late 2019 for just over 2 years, and currently has very minimal and manageable side effects, even while on the full dosage. The first couple of months CA was on entrectinib, she had a lot of trouble with fluid in her lungs and had to have a pleurocentesis to remove the fluid buildup. She also had edema, brain fog, and weight gain. However, she was doing very well by October 2020, 10 months after starting treatment, in that she was able to walk several miles per day comfortably, but in November, she started having trouble bending and walking, and by the following spring, she could barely walk. It was discovered that entrectinib had deteriorated her bones very quickly, particularly her hips, thus leading to 2 hip replacements in August and October 2021 respectively. As of February 2022, CA is still currently on the full dosage and notes that these side effects are very manageable and has not been a major impact on her lifestyle and independence ever since.
AP started her treatment at the full 600mg dosage, but she had a number of tough side effects from it, especially during the first 2 months. She was extremely tired during the first 1.5 months and was in bed most of the day, was also nauseous, and experienced neuropathy in her hands, especially when going to sleep. It also affected her taste in a way that she still currently does not taste food normally to this day and continues to struggle occasionally with fatigue. Her oncologist lowered her dose from 600 to 400mg in December 2021 because they were worried about its side effects and did not have other patients on the drug, and also because AP started having fluid build-up in her chest cavity that required a thoracentesis, draining 5L on each side. AP has been feeling much better since her dose was lowered and as of February 2022, she’s been feeling almost like her normal, pre-cancer self, attributing it to the significant difference the lowered dose had on her physically.
DR experienced increased creatinine levels in her kidneys, constipation and diarrhea, leg cramps especially at night, and fatigue, which makes it hard for her to walk very far. In December 2021, she had to temporarily pause treatment due to severe dehydration, but it was resolved after 3 days with saline hydration and is now much better. Ever since, DR has experienced minimal side effects that are easily manageable.
For AY, skin issues have been her main side effect, particularly at the beginning of treatment. She had a lot of rashes, psoriasis on her scalp, and intense dryness where she would often wake up with cuts and bleeds on her fingers and toes, making it hard to walk for the first few months. However, she has been treated with steroids which have helped immensely and is almost back to normal now. She had to reduce her dosage to 400 mg recently after Christmas in 2021 due to severe vertigo, headaches, vomiting, and numbness in left arm. AY is doing much better now as of February 2022, and although she still has some lingering side effects like dizziness when getting up, she leads almost a normal life as pre-diagnosis.
Similarly, BL also struggled with severe rashes when first onboarded with the treatment in August 2021. She recalls it was like her skin was “on fire”; she could not sleep and frequently had to take cold showers and sleep with icepacks and cooling lotion during the night to manage it. She also reported cognitive impacts from entrectinib, making her extremely forgetful, she would forget mid-sentence what she was talking about, didn’t sound like herself when speaking, brain fogginess, and mild memory loss. Her oncologist discussed lowering her dose from the full dosage, but BL was adamant to push through because ultimately, she felt as if she had no other choice and did not want to die. Ultimately, she had to terminate treatment with entrectinib less than 2 months after starting because new biomarker testing revealed BL had the HER2 amplification, thus making her resistant to ROS1 inhibitors. She started chemotherapy in October 2021 and has been on it ever since.
AN also experienced significant neurological side effects with entrectinib when she started on the full dose in 2016. She found it extremely difficult to walk straight and keep her balance due to vertigo, almost as if she was drunk and someone was pulling her. Unfortunately, she fainted a few times because of the severe vertigo and low blood pressure, which was very scary for her although she continued to go out alone to run errands. Ultimately, she had her dosage lowered a few months in to 400mg, which helped immensely and was able to tolerate the drug very well. She still had numerous side effects including edema, weight gain, hypotension, mild neuropathy, constipation, and fatigue, yet it did not impact her independence and continued to go about her daily life for years while on treatment. After she developed the G2032R resistance mutation, she terminated treatment 4 years later in late 2020, and switched to a different targeted therapy, repotrectinib, which she’s still on to this day.
Entrectinib has improved patients’ quality of life in numerous ways
Their current livelihoods while on entrectinib are comparable to what they had prior to diagnosis.
Prior to diagnosis, DR had always been active with her family and grandkids, often going on family trips vacationing across the USA and going on cruises, so when she was initially diagnosed in 2014 with stage 2 NSCLC when her first grandchild was 3 years old and a second one on the way, her ultimate goal was to live long enough to see her grandkids go to school and grow old enough to remember DR. She was always around the kids, picking them up from school, watching them, and going to playgrounds together. After she developed arthritis and underwent back surgery and 2 knee replacements while on entrectinib, it was tough for her to continue to be active and recover. Although she continues to use a walker or scooter most of the time when running errands or going to Disney, she attributes her quality of life to be almost the same as before diagnosis, and has had no symptoms of the cancer other than dealing with the lingering functionality effects. Entrectinib has allowed her to start living a more “normal” life compared to when she was on chemotherapy, and it has not stopped her from continuing to pick up the grandkids, who are now 11, 8 and 7, from school or caring for them.
Early on NW’s diagnosis, she was very active and took dance and Zumba classes once a week, but chemotherapy took this energy away from her as she’d be very sick right after treatment, then got better overtime, then back to being bedridden once she went in for another round of treatment. Since she started entrectinib, however, she has been able to return to a normal level of functionality and has learned to manage and live with the side effects, which are much more consistent. NW has been able to return to exercising and as she states, “entrectinib has allowed me to function normally enough so you’re not just a cancer patient, but rather able to return to my old sense of self”. Recently, NW has even felt good enough on the drug that she actually went back to work recently as a high school teacher.
Being very active and as an avid horse rider prior to diagnosis, CA was in very good and active shape, walking and riding horses almost every day, singing in choirs, swimming, and was often found outdoors enjoying her many hobbies. 2 months after she started entrectinib in December 2019, there were already significant improvements in her disease status and has been NED since early 2020. By October 2020, she felt like her pre-cancer self again and was comfortably walking 3-10 miles every day, maintained her independence throughout her cancer journey, driving herself, walking with friends, and was completely independent since she lives alone. She has found an incredible support system in her friends and online with the ROS1ders patient group, which has helped her maintain her optimism and positivity throughout her cancer journey. CA’s day-to-day life now is virtually the same as she had prior to diagnosis, aside from some lingering side effects from the drug, and is able to continue enjoying a great quality of life thanks to entrectinib.
Thanks to entrectinib, AY also leads an almost-normal lifestyle that has allowed her to travel with her husband overseas while he was stationed at a British Embassy, return to her home country to visit her family, and is eager to continue travelling around the world when COVID protocols allow her to do so. She is able to take the train into the city and socialize with friends, meet up for coffee, walking and shopping around everywhere, and attributes her positive attitude to the success that entrectinib has given her in her disease status. Aside from some minor side effects from the drug, like dizziness when getting up, AY has continued to live just as she did prior to diagnosis as a retired pediatrician, and continues to visit her daughter often, who lives about 2 hours away by train, and has a great support system in her family and friends.
For some patients, entrectinib’s success have allowed them to even return to work.
While on other treatments, NW was in a constantly brutal cycle of being bedridden for a week directly after treatment, then slowly regained her energy and functionality for a few weeks afterwards, then being bedridden again when she went in for her next round of chemotherapy treatment. The inconsistency in her symptoms and functionality made it impossible for her to make any long-term plans and enjoy her life, thus being forced to take more than a year off from work in 2019 and 2020. However, once she started entrectinib in late 2020, the side effects have been much more manageable and consistent, to the point where she felt well enough to go back to work a year into treatment in September 2021. As of February 2022, she has returned to working part-time as a grade 12 English teacher at her local high school without any complications and is excited for what the future holds for her.
Though 63-year-old CA had already retired before she was diagnosed, she loved her career as a high school teacher so much that she was eager to get back into the classroom as soon as her cancer journey was stable. While she is still currently on entrectinib, she recently returned to work at her retirement job that she genuinely loves, teaching in adjunct at different universities across the United States hosting seminar classes every 2 weeks. She is required to be on her feet while teaching for 5-6 hours per day and is completely fine doing so with no issues at all. She also teaches online classes once a week and foresees herself continuing to do so as long as possible, thanks to entrectinib.
Similar to CA and NW’s stories, MR was also an elementary school teacher who was just starting her 20th year of teaching when she was diagnosed with lung cancer. She was always very active with her three kids and husband, was outdoors biking, hiking, and running prior to diagnosis, so when entrectinib was noticeably able to bring this energy back to her about 3 months into treatment, she was very happy and optimistic about being able to get back to the outdoors with her family. A year into treatment with entrectinib in August 2021, her scans showed no evidence of active disease, which has still carried forward until today in February 2022. She had to leave the classroom as soon as she was diagnosed in 2020 because of COVID-19, the side effects of her treatments, and her job nature was too much for her at the time. However while LCC was interviewing her, she was eager to get back into the classroom and was in the process of getting medically cleared to return to work, a thought that was not in her mind when she was diagnosed. Entrectinib has given patients like MR, NW, and CA the opportunity to return to work in the jobs that they are passionate in, making a drastic difference in their quality of life and thus allowing them to set goals and wishes for the future.
The oral dosage route of entrectinib has numerous benefits for patients over standard IV treatments.
As described in section 4, the current standard of care for many cancer diagnoses, including NSCLC, include chemotherapy and immunotherapy, which are given via the intravenous (IV) route. This form of dosage requires patients to receive treatment in hospital, and may result in long stays at the hospital and also put a burden on their loved ones or caregivers who may have to stay and care for them during their treatment. For patients interviewed for this submission, many found much greater success with entrectinib in terms of efficacy on their disease, while also citing the ease of treatment that an oral dosage provides. Having to simply take a maximum of 3 pills per day, depending on dosage, allows patients to continue focusing on recovery at home without the need to travel to hospitals or clinics for treatment, unlike the requirement of IV treatments. It also allows them to spend more time with their family, friends, loved ones, and continue enjoying the activities they love. The oral nature of entrectinib has allowed some to even go back to work, without needing to take time off to attend treatments at hospitals. It places much less burden and stress on caregivers and family, and patients are able to maintain their independence while on the treatment, a value that is unmatched with IV treatments.
Companion Diagnostic Test
NTRK gene fusions can be identified through next-generation sequencing, which is offered at many cancer centers across the country. With new funding announcements in 2021 made across multiple provinces, there have been recent expansion in next-generation sequencing testing that will support testing for NTRK fusions for lung cancer patients.
Anything Else?
In recent years, with the development and availability of targeted therapies to treat molecular mutations, lung cancer patients’ lives have changed for the better. For non-small-cell lung cancer (NSCLC) patients in particular, these treatments have resulted in remarkable improvements in the overall survival and response rates compared to the standard of care, chemotherapy, allowing patients to live longer, return to work and spend time with loved ones and enjoy their favorite activities. One of the mutations found in this lung cancer subtype is the NTRK gene fusion. This form of lung cancer until recently, with the approval of larotrectinib had been treated with the standard of care, chemotherapy, an IV form of therapy which has been shown to work but comes with well-known toxic side effects. There is also a high rate of progression post treatment. With eventual progression after treatment with chemotherapy or if patients are not able to receive chemotherapy due to reasons such as co morbidities or even the side effects, it is important to provide patients with other viable treatment options such as larotrectinib and now, entrectinib, oral targeted therapies.
A positive approval for entrectinib provides a wider range of treatment options, giving patients who may not have effective treatment options a chance at survival with an alternative treatment option and more options for their physicians to choose from. Entrectinib is a durable form of treatment that provides a clinically meaningful response with manageable side effects while allowing patients to still have meaningful lives. The response rates to this treatment are comparable to the currently approved targeted treatment, as such there is no reason this treatment should not also be given a positive approval. This approval will also allow for the collection of real-world data which would further demonstrate the efficacy of this form of treatment. With more options on the market, there is not only choice, but it would also foster market competition which could help lower the drug costs.
Entrectinib is able to change the course of the disease for many NTRK positive NSCLC patients and should be considered as there is an unmet need to ensure viable treatment options for patients. LCC hopes CADTH considers this form of treatment for a positive approval.
Reference: Doebele, R. C., Drilon, A., Paz-Ares, L., Siena, S., Shaw, A. T., Farago, A. F., Blakely, C. M., Seto, T., Cho, B. C., Tosi, D., Besse, B., Chawla, S. P., Bazhenova, L., Krauss, J. C., Chae, Y. K., Barve, M., Garrido-Laguna, I., Liu, S. V., Conkling, P., John, T., … trial investigators (2020). Entrectinib in patients with advanced or metastatic NTRK fusion-positive solid tumours: integrated analysis of three phase 1-2 trials. The Lancet. Oncology, 21(2), 271–282. DOI: 10.1016/S1470-2045(19)30691-6
Conflict of Interest Declaration — Lung Cancer Canada
To maintain the objectivity and credibility of the CADTH reimbursement review process, all participants in the drug review processes must disclose any real, potential, or perceived conflicts of interest. This Patient Group Conflict of Interest Declaration is required for participation. Declarations made do not negate or preclude the use of the patient group input. CADTH may contact your group with further questions, as needed.
Did you receive help from outside your patient group to complete this submission? If yes, please detail the help and who provided it.
No
Did you receive help from outside your patient group to collect or analyze data used in this submission? If yes, please detail the help and who provided it.
No
List any companies or organizations that have provided your group with financial payment over the past 2 years AND who may have direct or indirect interest in the drug under review.
Table 6: Conflict of Interest Declaration for Lung Cancer Canada
Company | $0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 |
|---|---|---|---|---|
Roche | X | — | — | — |
Clinician Input
Ontario Health (Cancer Care Ontario) Breast Cancer Drug Advisory Committee
About Ontario Health (Cancer Care Ontario) Breast Cancer Drug Advisory Committee
OH-CCO’s Drug Advisory Committees provide timely evidence-based clinical and health system guidance on drug-related issues in support of CCO’s mandate, including the Provincial Drug Reimbursement Programs (PDRP) and the Systemic Treatment Program.
Information Gathering
Discussed jointly at a DAC meeting.
Current Treatments
Currently there is a 0.39% prevalence of breast cancer patients with an NTRK gene fusion. The NTRK gene fusion is not very common in BC. In breast cancer, we do not routinely test for NTRK gene fusion.
Treatment Goals
The most important goals that an ideal treatment would address would be improve overall survival with acceptable toxicity.
Treatment Gaps (Unmet Needs)
Considering the treatment goals, please describe goals (needs) that are not being met by currently available treatments.
There is a very small percentage of patients with NTRK gene fusion in breast cancer patients. The prevalence in secretory breast carcinoma is over 90%. Therefore, if clinicians are able to locate a secretory breast cancer, then there is a high probability that the patient would have an NTRK gene fusion and would benefit from this indication. Ideally, testing for the gene fusion would be available for this population.
Which patients have the greatest unmet need for an intervention such as the drug under review?
Patients with breast cancer that have the NTRK gene fusion and secretory breast carcinoma patients.
Place in Therapy
How would the drug under review fit into the current treatment paradigm?
Unsure how this would fit into the current treatment paradigm as there are very few patients with breast cancer that have NTRK and testing for it is not routine.
Please indicate whether or not it would be appropriate to recommend that patients try other treatments before initiating treatment with the drug under review. Please provide a rationale from your perspective.
There are many treatments for mBC but all have palliative intent.
How would this drug affect the sequencing of therapies for the target condition?
In the study, Entrectinib was used in any line of therapy. Therefore, Entrectinib could be an additional line of therapy.
Which patients would be best suited for treatment with the drug under review?
Patients with NTRK gene fusion in breast tumours.
How would patients best suited for treatment with the drug under review be identified?
Ideally, all metastatic breast cancer patients would be tested. However, given the low prevalence of mutation in breast cancer overall, identifying subsets such as secretory breast cancer that have a higher prevalence of mutations would be helpful.
Which patients would be least suitable for treatment with the drug under review?
Not applicable.
Is it possible to identify those patients who are most likely to exhibit a response to treatment with the drug under review?
Secretory breast cancer patients seem to have a higher prevalence of mutations.
What outcomes are used to determine whether a patient is responding to treatment in clinical practice?
Overall survival.
What would be considered a clinically meaningful response to treatment?
A clinically meaningful response would be tumour response within the breast.
How often should treatment response be assessed?
As per the study.
What factors should be considered when deciding to discontinue treatment?
The usual factors (disease progression or toxicity).
What settings are appropriate for treatment with the drug under review?
Cancer center with expertise in toxicity management.
For non-oncology drugs, is a specialist required to diagnose, treat, and monitor patients who might receive the drug under review?
Not applicable.
Additional Information
Access to this drug for patients who are eligible would be ideal, but it is difficult to identify eligible patients in a cost effective manner since the prevalence of the target mutation is rare in breast cancer. A third of the breast cancer patients on this setting had a CR but it’s not easy to identify them and the evidence is only based on 6 patients. We need a different paradigm for evaluating these treatments from a disease agnostic perspective.
Conflict of Interest Declarations — Ontario Health (Cancer Care Ontario) Breast Cancer Drug Advisory Committee
To maintain the objectivity and credibility of the CADTH drug review programs, all participants in the drug review processes must disclose any real, potential, or perceived conflicts of interest. This conflict of interest declaration is required for participation. Declarations made do not negate or preclude the use of the clinician group input. CADTH may contact your group with further questions, as needed. Please see the Procedures for CADTH Drug Reimbursement Reviews (section 6.3) for further details.
Did you receive help from outside your clinician group to complete this submission? If yes, please detail the help and who provided it.
OH-CCO provided secretariat support to the DAC in completing this input.
Did you receive help from outside your clinician group to collect or analyze any information used in this submission? If yes, please detail the help and who provided it.
No.
List any companies or organizations that have provided your group with financial payment over the past two years AND who may have direct or indirect interest in the drug under review. Please note that this is required for each clinician who contributed to the input — please add more tables as needed (copy and paste). It is preferred for all declarations to be included in a single document.
Declaration for Clinician 1
Name: Dr. Andrea Eisen
Position: OH-CCO Breast Cancer Drug Advisory Committee Lead
Date: 11/02/2022
Table 7: Conflict of Interest Declaration for Ontario Health (Cancer Care Ontario) Breast Cancer Drug Advisory Committee — Clinician 1
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 |
|---|---|---|---|---|
No COI | — | — | — | — |
Declaration for Clinician 2
Name: Dr. Phillip Blanchette
Position: OH-CCO Breast Cancer Drug Advisory Committee Member
Date: 11/02/2022
Table 8: Conflict of Interest Declaration for Ontario Health (Cancer Care Ontario) Breast Cancer Drug Advisory Committee — Clinician 2
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 |
|---|---|---|---|---|
No COI | — | — | — | — |
Lung Cancer Canada
About Lung Cancer Canada
Lung Cancer Canada (LCC) is a national charity with the purpose of increasing awareness about lung cancer, providing support and education to lung cancer patients and their families, to support research and to advocate for access to the best care for all lung cancer patients in all provinces and territories. Through the LCC Medical Advisory Committee (MAC), we have been providing clinician input for submissions of new lung cancer drugs to the HTA process for many years. The LCC MAC is made up of clinicians and key opinion leaders in the field of lung cancer across the country. www.lungcancercanada.ca
Information Gathering
Information is from publicly available sources, primarily published manuscripts and conference presentations, together with experience of the members of the clinician group. This submission is entirely independent of the manufacturer (Roche).
Current Treatments
NTRK cancers are identified across a broad range of malignancies and represent a new paradigm in oncology. NTRK fusion cancers have just emerged in the past 3-4 years. NTRK fusions are a ‘tumour agnostic’ driver mutation, identified in a broad range of pediatric and adult cancers, including lung cancer.
Therefore, the current treatment is diverse depending on the standard of care treatment options in each known cancer.
That being said, there are no approved therapies for NTRK cancer until the last 12 months, with the emergence of larotrectinib and entrectinib.
NTRK fusions are extremely rare in lung cancer (approximately 0.23% of cases), and therefore there is little data on efficacy of current standard treatments. In lung cancer, treatment options include chemotherapy (platinum doublet chemotherapy) and immunotherapy (either monotherapy or in combination with chemotherapy), and then either docetaxel chemotherapy or best supportive care. Larotrectinib has been approved by CADTH in late 2021 (https://www.cadth.ca/larotrectinib), for treatment of NTRK fusion cancers when there are no satisfactory treatment options. This new application with entrectinib would provide an alternative to larotrectinib.
Treatment Goals
Efficacy / survival. Does the new treatment significantly improve survival compared to the standard of care, or does it provide at least equivalent efficacy in this population? The data presented for entrectinib identifies a prolonged disease control at a time when there are no other satisfactory treatment options. Due to the rarity of the condition, there are no randomized studies, therefore there is no statistically proven overall survival (OS) advantage. However, given the response rates and duration of response described by Doebele et al, in the context of patients having no other reasonable therapy options, it is unconceivable that entrectinib does not improve OS.
Efficacy / response. An improvement in cancer symptoms is most commonly achieved with significant tumour shrinkage, so an ideal treatment has a high tumour response rate and disease control rate. Entrectinib is associated with high tumour response rates (57%), commensurate with this being an active drug for a driver mutation, and now seen in multiple oncogenes driven lung cancer with approved therapies (e.g. osimertinib in EGFR+ NSCLC, alectinib in ALK+ NSCLC, larotrectinib in NTRK fusion positive cancers, and indeed entrectinib in ROS1 NSCLC).
Tolerability. An ideal therapy has low rates of treatment related adverse events, which means that it is accessible to a broader population and allows patients to remain on therapy with maximum benefit. Therefore, low rates of high-grade toxicity (grade 3-4) is important. In the published entrectinib studies, extremely low rates of grade 3 or 4 adverse events were reported (<10%).
Ease of administration. As an oral medication, entrectinib can be taken at home. It is easy and convenient for patients, reduces the burden on cancer centre infusion clinics, and indeed during the COVID-19 pandemic means there are fewer required trips to hospital.
Treatment Gaps (Unmet Needs)
Considering the treatment goals, please describe goals (needs) that are not being met by currently available treatments.
NTRK fusion cancers occur across a broad range of adult and pediatric malignancies, as previously described. On behalf of this submission with Lung Cancer Canada, we will largely restrict comments to the non-small cell lung cancer (NSCLC) population. In general (but not exclusively) NTRK NSCLC patients have no or little tobacco exposure, and this is associated with a reduced likelihood of response to immunotherapy agents. Specific data on efficacy to standard treatments is unclear due to the rarity of the disease (the larotrectinib positive approval was based on a trial with only 12 NSCLC patients, and the entrectinib study for this submission contains 10 NSCLC patients). However, it is clear that advanced NSCLC patients with NTRK fusions will, in their natural disease course, become resistant to chemotherapy and immunotherapy at which point there are no standard therapies available. Entrectinib is able to change the course of the disease, is well tolerated, easy to administer, and is demonstrated to deliver high response rates (57% response rate overall, and 70% response rate in NSCLC) and durable responses (duration of response 10 months, PFS 11 months) in line with targeted therapy efficacy.
As an oral therapy with few side effects, convenience and compliance are two assets to this drug.
Which patients have the greatest unmet need for an intervention such as the drug under review?
Certainly, this represents a niche population, with NTRK fusions only reported in 0.23% of non-small cell lung cancer cases (Farago et al. JCO Precision Oncology 2018). However, as lung cancer is the most commonly diagnosed cancer in Canada (29,600 cases in 2021: Canadian Cancer Society statistics), and as NGS testing is becoming more and more widespread, these cases are now being identified. There is a clear unmet need to provide therapeutic options for these patients when no other satisfactory options exist. It was this acceptance that led to the approval of larotrectinib, and now entrectinib is demonstrating similar efficacy.
Place in Therapy
How would the drug under review fit into the current treatment paradigm?
This would be an additional option for patients without access to NTRK drugs, or an alternative to larotrectinib. We foresee that an NTRK inhibitor (entrectinib or larotrectinib) will automatically become a standard of care option based on factors discussed (high response rate, including CNS response; long duration of response; low adverse event rate). Depending on availability, preference or tolerability, lung cancer patients with NTRK fusion cancers could receive either larotrectinib or entrectinib. We would also advocate the option of patients switching from one to the other drug in situations where the initial TRK inhibitor is not tolerated.
The application is for entrectinib to be used when there are no satisfactory treatment options. So for the lung cancer paradigm that may be after chemotherapy and/or immunotherapy, or as first-line treatment in patients with contraindications to either chemotherapy or immunotherapy (due to comorbidities or performance status).
Please indicate whether or not it would be appropriate to recommend that patients try other treatments before initiating treatment with the drug under review. Please provide a rationale from your perspective.
The current non-small cell lung cancer (NSCLC) algorithm would include first line platinum doublet chemotherapy, immunotherapy, or the combination, depending on patient factors such as PDL1 status, comorbidities and performance status. NTRK fusions are only reported as solitary driver mutations, i.e. they have not been reported to co-exist with other driver mutations such as EGFR or ALK.
It would be reasonable to try chemotherapy/immunotherapy options if appropriate, however biologically and clinically the most effective intervention is likely to be the NTRK drug (entrectinib), so first-line therapy should be available if other options are deemed clinically inappropriate. I.e Mandating prior therapy with chemotherapy or immunotherapy before receiving entrectinib would be wrong, as it would deny access to effective therapy to many patients.
How would this drug affect the sequencing of therapies for the target condition?
As described previously, the most likely sequencing of entrectinib in NSCLC would be after chemotherapy or immunotherapy (or the combination) but should be available as a first line option when those other therapies are contraindicated. As per the application and product monograph, as entrectinib is recommended when there are no other satisfactory treatment options, there are no standard of care therapies after entrectinib, and patients would likely have the option of clinical trials or best supportive care.
Which patients would be best suited for treatment with the drug under review?
All patients with an NTRK fusion identified should have access to a TRK inhibitor such as entrectinib. Due to its high efficacy and excellent tolerability, all patients should be offered this therapy if they have not otherwise received larotrectinib or have a specific contra-indication
How would patients best suited for treatment with the drug under review be identified?
NTRK gene fusions can be detected either through next generation sequencing (NGS), optimally using DNA and RNA platforms. It can also be detected as a stand-alone test, which is a 2 part process of immunohistochemistry (IHC), which if positive then leads onto a confirmatory NGS panel.
For lung cancer, currently many centres do perform NGS panels, and with new funding announcements in 2021 in multiple provinces, there has been a significant expansion of NGS testing that will allow identification of NTRK fusion patients, in addition to other subtypes such as EGFR, ALK, ROS1, BRAF, KRAS G12C, Her2, c-Met exon14 and Ret.
For centres who offer a more limited panel at present, NTRK testing can be reserved for those who are already pan-negative from the initial molecular panel, as NTRK is not reported to co-exist with other driver mutations.
Which patients would be least suitable for treatment with the drug under review?
Clearly patients without identifiable NTRK fusions will be least suitable, although ROS1 NSCLC patients do also respond well to entrectinib, as supported by a prior CADTH recommendation (https://cadth.ca/entrectinib-rozlytrek-ros1-positive-non-small-cell-lung-cancer). NTRK positive patients who have previously been treated with larotrectinib would not be suitable for entrectinib, unless they have had to stop the larotrectinib due to adverse events rather than progression, in which case a trial of entrectinib would be indicated.
Is it possible to identify those patients who are most likely to exhibit a response to treatment with the drug under review?
The response rates to entrectinib are impressive, with published disease control rates of >70%, the overall response rate of 57% rising to 70% in the NSCLC cohort. There are different subtypes of NTRK fusion (NTRK1, NTRK2 and NTRK3), but the authors of the submitted landmark study (Doebele et al. Lancet Oncology 2020) report similar response rates whether patients had NTRK1 or NTRK3 fusions (only 1 patient had an NTRK2 fusion), and responses were observed in all tumour types. Further, patients who had CNS metastases when starting entrectinib also had a 55% intracranial response rate. No patterns emerged of particular patient sub-populations where entrectinib was clearly ineffective, therefore concluding that all patients with an identified NTRK fusion should be able to receive entrectinib.
What outcomes are used to determine whether a patient is responding to treatment in clinical practice?
Standard of care follow up of these patients involves clinical assessment of tolerability and symptom assessment, and body imaging to assess tumour response (computed tomography [CT], magnetic resonance imaging [MRI]). The frequency of follow up and choice of response measurement tool will be dictated by local practice and the individual patient clinical scenario.
What would be considered a clinically meaningful response to treatment?
Dr. Paz Ares (ESMO Open 2021) published the Patient-reported outcomes from the STARTRK-2 study of entrectinib, including 88 NTRK positive patients, showing improvements in role and physical functioning and stability or improvements in symptom burden and treatment associated symptoms, concluding that the PRO findings were ‘consistent with the favourable safety profile of entrectinib, and further reinforce the positive benefit-risk profile of this treatment’.
Taking these finding with the trial results of durable response, intracranial response and overall high response rates, the clinically meaningful benefits are evident.
These treatments reduce or improve symptoms, prolong life and good quality of life, allowing patients to continue with independent living and achieving their life goals.
The entrectinib data also describes a larger than usual complete response rate, with many patients experiencing deep responses that can lead to significant improvements in symptom burden, therefore increasing quality of life and independence.
Anecdotally, a patient under my care with NTRK fusion cancer has responded well and been able to travel to his country of birth to see family he would otherwise have not been able to visit.
How often should treatment response be assessed?
Every 6-12 weeks, depending on patient factors
What factors should be considered when deciding to discontinue treatment?
In clinical practice, entrectinib will continue until one or more of the following conditions is/are fulfilled:
Toxicity requiring permanent discontinuation of therapy (although in the published entrectinib studies, this was uncommon at only 4%.)
Patient wishes
Unequivocal radiological disease progression
What settings are appropriate for treatment with the drug under review?
Entrectinib should only be prescribed by a specialist managing the condition. Given the tumour agnostic nature of NTRK fusions cancers, this will most commonly be a medical oncologist or pediatric oncologist. For the lung cancer patients, in some jurisdictions respirologists are also overseeing systemic therapy and would be appropriate to prescribe this. As this is a well-tolerated oral drug, there are no specific additional implications for treatment, as this can be taken at home.
For non-oncology drugs, is a specialist required to diagnose, treat, and monitor patients who might receive the drug under review?
Not applicable
Additional Information
As discussed earlier in this document, larotrectinib has already been approved by CADTH for the same indication as this submission for entrectinib (https://www.cadth.ca/larotrectinib). Both entrectinib and larotrectinib are targeted therapies that are well tolerated and have demonstrated efficacy in NTRK fusion cancers, and we would ask that entrectinib be approved to provide an option for patients and clinicians with this rare disease
Conflict of Interest Declarations — Lung Cancer Canada
To maintain the objectivity and credibility of the CADTH drug review programs, all participants in the drug review processes must disclose any real, potential, or perceived conflicts of interest. This conflict of interest declaration is required for participation. Declarations made do not negate or preclude the use of the clinician group input. CADTH may contact your group with further questions, as needed. Please see the Procedures for CADTH Drug Reimbursement Reviews (section 6.3) for further details.
Did you receive help from outside your clinician group to complete this submission? If yes, please detail the help and who provided it.
No.
Did you receive help from outside your clinician group to collect or analyze any information used in this submission? If yes, please detail the help and who provided it.
No.
List any companies or organizations that have provided your group with financial payment over the past two years AND who may have direct or indirect interest in the drug under review. Please note that this is required for each clinician who contributed to the input — please add more tables as needed (copy and paste). It is preferred for all declarations to be included in a single document.
Declaration for Clinician 1
Name: Dr. Paul Wheatley-Price
Position: Medical Oncologist, The Ottawa Hospital; Associate Professor, Department of Medicine, University of Ottawa
Date: 09-02-2022
Table 9: Conflict of Interest Declaration for Lung Cancer Canada — Clinician 1
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 |
|---|---|---|---|---|
Sanofi | X | — | — | — |
Astra Zeneca | X | — | — | — |
Jazz Pharmaceuticals | X | — | — | — |
Amgen | X | — | — | — |
Janssen | X | — | — | — |
Novartis | X | — | — | — |
Merck | X | — | — | — |
BMS | X | — | — | — |
Roche | X | — | — | — |
EMD Serono | X | — | — | — |
Pfizer | X | — | — | — |
Bayer | X | — | — | — |
Novartis | X | — | — | — |
Declaration for Clinician 2
Name: Dr. Rosalyn Juergens
Position: Chair, LCC Medical Advisory Committee; Medical Oncologist, Juravinski Cancer Center
Date: Feb 17, 2022
Table 10: Conflict of Interest Declaration for Lung Cancer Canada — Clinician 2
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 |
|---|---|---|---|---|
Bristol Myers Squibb | X | — | — | — |
Astra Zeneca | — | X | — | — |
Merck Sharp and Dohme | X | — | — | — |
Roche | X | — | — | — |
Declaration for Clinician 3
Name: Dr. Geoffrey Liu
Position: Medical Oncologist, Princess Margaret Cancer Centre
Date: Feb 17, 2022
Table 11: Conflict of Interest Declaration for Lung Cancer Canada — Clinician 3
Company | Nature or description of activities or interests | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 |
|---|---|---|---|---|---|
Takeda Canada | Advisory Board, Health Technology Assessment Submission Advice, Speaker’s Bureau, past 10 years | — | — | X | — |
Takeda Canada | (To institution, not individual) Observational Study funding, past 10 years | — | — | — | X |
Hoffman La Roche | Advisory Board, Health Technology Assessment Submission Advice, past 10 years | — | — | X | — |
Pfizer | Advisory Board, Health Technology Assessment Submission Advice, part 10 years | — | — | X | — |
AstraZeneca | Advisory Board, Health Technology Assessment Submission Advice, Speaker’s Bureau, past 10 years, | — | — | X | — |
AstraZeneca | (To institution, not individual) Observational Study funding, past 10 years | — | — | — | X |
Bristol Myers Squibb | Advisory Board | X | — | — | — |
Boehringer Ingerheim | (To institution, not individual) Observational Study funding, past 10 years | — | — | X | — |
Abbvie | Advisory Board, past 10 years | — | X | — | — |
Merck | Advisory Board, Health Technology Assessment Submission Advice, past 10 years | — | X | — | — |
EMD Serono | Speaker’s Bureau, past 10 years | X | — | — | — |
Novartis | Advisory Board,past 10 years | — | — | X | — |
Glaxo Smith Kline | Advisory Board, past 10 years | — | X | — | — |
Declaration for Clinician 4
Name: Dr Jeffrey Rothenstein
Position: Medical Oncologist, Lakeridge Health
Date: Feb 17, 2022
Table 12: Conflict of Interest Declaration for Lung Cancer Canada — Clinician 4
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 |
|---|---|---|---|---|
Roche | X | — | — | — |
Declaration for Clinician 5
Name: Dr Nicole Bouchard
Position: Respirologist, Sherbrooke University Hospital
Date: Feb 17, 2022
Table 13: Conflict of Interest Declaration for Lung Cancer Canada — Clinician 5
Company | Nature or description of activities or interests | $0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 |
|---|---|---|---|---|---|
Astra Zeneca | Advisory Role/Conference | X | — | — | — |
Bristol-Myers Squibb | Advisory Role/Research | X | — | — | — |
Merck | Advisory Role/Research/Conference | X | — | — | — |
Bayer | Advisory Role | X | — | — | — |
Pfizer | Conference/Research | X | — | — | — |
Roche | Advisory Role | X | — | — | — |
Declaration for Clinician 6
Name: Dr Normand Blais
Position: Medical Oncologist, Hôpital Notre Dame du CHUM
Date: Feb 17, 2022
Table 14: Conflict of Interest Declaration for Lung Cancer Canada — Clinician 6
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 |
|---|---|---|---|---|
Novartis | X | — | — | — |
Declaration for Clinician 7
Name: Dr. David Dawe
Position: Medical Oncologist, CancerCare Manitoba
Date: Feb 17, 2022
Table 15: Conflict of Interest Declaration for Lung Cancer Canada — Clinician 7
Company | Nature or description of activities or interests | $0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 |
|---|---|---|---|---|---|
AstraZeneca | Advisory boards | X | — | — | — |
Merck | Advisory Boards | X | — | — | — |
AstraZeneca | Research Grant | — | — | X | — |
Boehringer-Ingelheim | Honoraria | X | — | — | — |
Declaration for Clinician 8
Name: Dr Randeep Sangha
Position: Associate Professor, University of Alberta; Medical Oncologist, Cross Cancer Institute
Date: Feb 17, 2022
Table 16: Conflict of Interest Declaration for Lung Cancer Canada — Clinician 8
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 |
|---|---|---|---|---|
No COI | — | — | — | — |
Declaration for Clinician 9
Name: Dr Catherine Labbé
Position: Head of Respiratory Medicine Service, Université de Laval
Date: Feb 17, 2022
Table 17: Conflict of Interest Declaration for Lung Cancer Canada — Clinician 9
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 |
|---|---|---|---|---|
Amgen | X | — | — | — |
Astra Zeneca | — | X | — | — |
Brystol-Myers Squibb | X | — | — | — |
Jazz Pharmaceuticals | X | — | — | — |
LEO Pharma | X | — | — | — |
Merck | X | — | — | — |
Pfizer | X | — | — | — |
Roche | X | — | — | — |
Sanofi Genzyme | X | — | — | — |
Declaration for Clinician 10
Name: Dr. Donna Maziak
Position: Thoracic Surgeon, The Ottawa Hospital
Date: Feb 17, 2022
Table 18: Conflict of Interest Declaration for Lung Cancer Canada — Clinician 10
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 |
|---|---|---|---|---|
No COI | — | — | — | — |
Declaration for Clinician 11
Name: Dr Sunil Yadav
Position: Medical Oncologist, Saskatoon Cancer Centre
Date: Feb 17, 2022
Table 19: Conflict of Interest Declaration for Lung Cancer Canada — Clinician 11
Company | Nature or description of activities or interests | $0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 |
|---|---|---|---|---|---|
Bristol-Myers Squibb | Advisory Board | X | — | — | — |
Astra Zeneca | Advisory Board and Speaking | X | — | — | — |
Merck | Advisory Board and Speaking | — | — | X | — |
Roche | Advisory Board and Speaking | — | X | — | — |
Takeda | Advisory Board and Speaking | X | — | — | — |
Declaration for Clinician 12
Name: Dr. Quincy Chu
Position: Medical Oncologist, Cross Cancer Institute
Date: Feb 17, 2022
Table 20: Conflict of Interest Declaration for Lung Cancer Canada — Clinician 12
Company | Nature or description of activities or interests | $0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 |
|---|---|---|---|---|---|
Abbvie | Advisory Board and Honoraria | X | — | — | — |
Amgen | Advisory Board and Honoraria | X | — | — | — |
Astra Zeneca | Advisory Board and Honoraria | — | — | X | — |
Boehringer Ingeiheim | Advisory Board and Honoraria | — | X | — | — |
Bristol-Myers Squibb | Advisory Board and Honoraria | — | X | — | — |
Eisai | Advisory Board and Honoraria | X | — | — | — |
Merck | Advisory Board and Honoraria | — | — | X | — |
Novartis | Advisory Board and Honoraria | — | X | — | — |
Pfizer | Advisory Board and Honoraria | — | X | — | — |
Roche | Advisory Board and Honoraria | — | X | — | — |
Astra Zeneca | Research Funding | — | — | — | X |
Bristol-Myers Squibb | Educational Grant | X | — | — | — |
Declaration for Clinician 13
Name: Dr. Ronald Burkes
Position: Medical oncologist, Mount Sinai Health
Date: Feb 17, 2022
Table 21: Conflict of Interest Declaration for Lung Cancer Canada — Clinician 13
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 |
|---|---|---|---|---|
No COI | — | — | — | — |
Declaration for Clinician 14
Name: Dr. Zhaolin Xu
Position: Pathologist, QEII Health Sciences Centre
Date: Feb 17, 2022
Table 22: Conflict of Interest Declaration for Lung Cancer Canada — Clinician 14
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 |
|---|---|---|---|---|
AstraZeneca | X | — | — | — |
Declaration for Clinician 15
Name: Dr. Shaqil Kassam
Position: Medical Oncologist, Southlake Regional Hospital
Date: Feb 17, 2022
Table 23: Conflict of Interest Declaration for Lung Cancer Canada — Clinician 15
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 |
|---|---|---|---|---|
Roche | X | — | — | — |
Merck | X | — | — | — |
BMS | X | — | — | — |
Takeda | X | — | — | — |
Novartis | X | — | — | — |
Ipsen | X | — | — | — |
Sanofi | X | — | — | — |
Pfizer | X | — | — | — |
Declaration for Clinician 16
Name: Dr. Silvana Spadafora
Position: Medical Oncologist, Sault Area Hospital
Date: Feb 17, 2022
Table 24: Conflict of Interest Declaration for Lung Cancer Canada — Clinician 16
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 |
|---|---|---|---|---|
No COI | — | — | — | — |
Declaration for Clinician 17
Name: Dr. Stephanie Snow
Position: President, Lung Cancer Canada; Medical Oncologist, The QEII Health Sciences Center
Date: Dec 22, 2021
Table 25: Conflict of Interest Declaration for Lung Cancer Canada — Clinician 17
Company | Nature or description of activities or interests | $0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 |
|---|---|---|---|---|---|
Amgen | Advisory Role | X | — | — | — |
Astra Zeneca | Advisory Role | — | — | X | — |
Bayer | Advisory Role | — | X | — | — |
Boehringer Ingeiheim | Advisory Role | X | — | — | — |
Bristol-Myers Squibb | Advisory Role | — | — | X | — |
Eisai | Advisory Role | X | — | — | — |
Merck | Advisory Role | — | — | X | — |
Novartis | Advisory Role | X | — | — | — |
Pfizer | Advisory Role | X | — | — | — |
Purdue | Advisory Role | X | — | — | — |
Roche | Advisory Role | — | — | X | — |
Taiho | Advisory Role | X | — | — | — |
Takeda | Advisory Role | — | X | — | — |
Declaration for Clinician 18
Name: Dr. Kevin Jao
Position: Medical Oncologist, Hôpital Sacré-Cœur, Montreal
Date: Feb 17, 2022
Table 26: Conflict of Interest Declaration for Lung Cancer Canada — Clinician 18
Company | Nature or description of activities or interests | $0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 |
|---|---|---|---|---|---|
Bristol-Myers Squibb | Advisory Role | X | — | — | — |
Declaration for Clinician 19
Name: Dr. Callista Phillips
Position: Medical Oncologist, Hamilton Health Sciences Center
Date: Feb 17, 2022
Table 27: Conflict of Interest Declaration for Lung Cancer Canada — Clinician 19
Company | Nature or description of activities or interests | $0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 |
|---|---|---|---|---|---|
Astra Zeneca | Advisory Board Stage 3 NSCLC | X | — | — | — |
Bayer | National Consultancy meeting and Train the Trainer- Larotrectenib in NTRK fusion positive cancers | X | — | — | — |
Roche | Lung regional Consultancy meeting | X | — | — | — |
Declaration for Clinician 20
Name: Dr. Barb Melosky
Position: Medical Oncologist, BC Cancer
Date: Feb 17, 2022
Table 28: Conflict of Interest Declaration for Lung Cancer Canada — Clinician 20
Company | Nature or description of activities or interests | $0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 |
|---|---|---|---|---|---|
Novartis | Advisory Board | X | — | — | — |
Roche | Advisory Board | X | — | — | — |
Merck | Advisory Board | X | — | — | — |
Declaration for Clinician 21
Name: Dr. David Stewart
Position: Medical Oncologist, The Ottawa Hospital
Date: Feb 17, 2022
Table 29: Conflict of Interest Declaration for Lung Cancer Canada — Clinician 21
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 |
|---|---|---|---|---|
No COI | — | — | — | — |
Declaration for Clinician 22
Name: Dr. Cheryl Ho
Position: Medical Oncologist, BC Cancer
Date: Feb 17, 2022
Table 30: Conflict of Interest Declaration for Lung Cancer Canada — Clinician 22
Company | Nature or description of activities or interests | $0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 |
|---|---|---|---|---|---|
Bayer | Advisory role | X | — | — | — |
Roche | Advisory role, travel, research grants | — | — | — | X |
Ontario Health (Cancer Care Ontario) Lung Cancer Drug Advisory Committee
About Ontario Health (Cancer Care Ontario) Lung Cancer Drug Advisory Committee
OH-CCO’s Drug Advisory Committees provide timely evidence-based clinical and health system guidance on drug-related issues in support of CCO’s mandate, including the Provincial Drug Reimbursement Programs (PDRP) and the Systemic Treatment Program.
Information Gathering
This input was jointly discussed via Drug Advisory Committee meeting and email.
Current Treatments
Current standard treatment for NTRK+ NSCLC is the same as for all non-driver mutated NSCLC - combination platinum doublet chemotherapy + immunotherapy or immunotherapy in the first line. Patients receiving immunotherapy alone as first line will receive a platinum doublet as second line therapy. Some patients who progress on these treatments will go on to receive docetaxel.
NCCN and ASCO guidelines recommend entrectinib or larotrectinib as 1st line therapy of NTRK fusion+ NSCLC, or as 2nd line or later therapy if the NTRK fusion is discovered after initiating first line therapy.
Treatment Goals
Improve survival, improve quality of life, delay time to chemo, improve compliance.
Treatment Gaps (Unmet Needs)
Considering the treatment goals in Section 4, please describe goals (needs) that are not being met by currently available treatments.
The overwhelming majority of patients become refractory to chemotherapy and/or immunotherapy and die of their disease.
Chemotherapy has significant toxicities and can negatively impact quality of life.
Entrectinib is an oral therapy and therefore easier and more convenient to administer than current standards of chemo/immunotherapy which are administered in a hospital setting intravenously.
Which patients have the greatest unmet need for an intervention such as the drug under review?
These patients represent a very small subpopulation of NSCLC patients who have NTRK fusion (about 1% of NSCLC pts). Treatment of NTRK fusion pos NSCLC patients is an unmet need as currently there is no targeted therapy for this subset.
Place in Therapy
How would the drug under review fit into the current treatment paradigm?
Entrectinib would be used in some patients as 1st line displacing chemo/immunotherapy combinations or pembrolizumab as single agent to second line. It would also be used 2nd line or later in NTRK+ patients who did not receive it in first line.
Please indicate whether or not it would be appropriate to recommend that patients try other treatments before initiating treatment with the drug under review. Please provide a rationale from your perspective.
In general, targeted therapies have higher response rates with longer durations of response. RR of 57% in this trial of pre-treated patients is higher than RR for current 1st line standard of care. As an oral therapy, there is likely to be better compliance. Side effect profile is also favourable compared with chemotherapy resulting in better quality of life, therefore it would be appropriate to use entrectinib in the 1st line. However, there is no data comparing entrectinib to current 1st line therapy options for NTRK+ patients so it is also reasonable to use it in later lines of therapy.
How would this drug affect the sequencing of therapies for the target condition?
Entrectenib would displace 1st line immunotherapy/chemo combinations or 1st line immunotherapy single agent to 2nd line in patients without a driver mutation in many patients, 2nd line rx (docetaxel) to third line. If used in 2nd line or later, it would displace whatever would be used in 2nd line therapy to 3rd line. None of the current therapy options would be replaced by entrectinib.
Which patients would be best suited for treatment with the drug under review?
Patients with metastatic NSCLC containing an NTRK fusion.
How would patients best suited for treatment with the drug under review be identified?
By testing of presence of NTRK fusion which ideally would be done reflexively on all lung cancer biopsies. Some patients who are suitable for this treatment are missed because of lack of testing/availability of testing.
Which patients would be least suitable for treatment with the drug under review?
Patients without an NTRK fusion or extremely poor performance status.
Is it possible to identify those patients who are most likely to exhibit a response to treatment with the drug under review?
Yes – patients with NTRK fusion.
What outcomes are used to determine whether a patient is responding to treatment in clinical practice?
Refer to next answer.
What would be considered a clinically meaningful response to treatment?
Reduction in the frequency or severity of symptoms such as cough, sob, fatigue, anorexia, pain. Stabilization (no deterioration) of symptoms.
How often should treatment response be assessed?
Clinical assessment every 4-8 weeks, CT every 3-4 months.
What factors should be considered when deciding to discontinue treatment?
Disease progression (clinical or radiographic) not amenable to control with targeted radiation. Intolerance. Patient wishes. Toxicities especially if grade 3 or higher.
What settings are appropriate for treatment with the drug under review?
Home for most patients, but appropriate to start/administer in certain hospitalized patients.
For non-oncology drugs, is a specialist required to diagnose, treat, and monitor patients who might receive the drug under review?
Not applicable.
Additional Information
No.
Conflict of Interest Declarations — Ontario Health (Cancer Care Ontario) Lung Cancer Drug Advisory Committee
To maintain the objectivity and credibility of the CADTH drug review programs, all participants in the drug review processes must disclose any real, potential, or perceived conflicts of interest. This conflict of interest declaration is required for participation. Declarations made do not negate or preclude the use of the clinician group input. CADTH may contact your group with further questions, as needed. Please see the Procedures for CADTH Drug Reimbursement Reviews (section 6.3) for further details.
Did you receive help from outside your clinician group to complete this submission? If yes, please detail the help and who provided it.
OH-CCO provided secretariat support to the DAC in completing this input.
Did you receive help from outside your clinician group to collect or analyze any information used in this submission? If yes, please detail the help and who provided it.
No.
List any companies or organizations that have provided your group with financial payment over the past two years AND who may have direct or indirect interest in the drug under review. Please note that this is required for each clinician who contributed to the input — please add more tables as needed (copy and paste). It is preferred for all declarations to be included in a single document.
Declaration for Clinician 1
Name: Dr. Stacey Hubay
Position: OH-CCO Lung DAC Member
Date: 16-02-2022
Table 31: Conflict of Interest Declaration for Ontario Health (Cancer Care Ontario) Lung Cancer Drug Advisory Committee — Clinician 1
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 |
|---|---|---|---|---|
No COI | — | — | — | — |
Declaration for Clinician 2
Name: Dr. Stephanie Brule
Position: OH-CCO Lung DAC Member
Date: 16-02-2022
Table 32: Conflict of Interest Declaration for Ontario Health (Cancer Care Ontario) Lung Cancer Drug Advisory Committee — Clinician 2
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 |
|---|---|---|---|---|
No COI | — | — | — | — |
Declaration for Clinician 3
Name: Dr. Natasha Leighl
Position: OH-CCO Lung DAC Member
Date: 16-02-2022
Table 33: Conflict of Interest Declaration for Ontario Health (Cancer Care Ontario) Lung Cancer Drug Advisory Committee — Clinician 3
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 |
|---|---|---|---|---|
No COI | — | — | — | — |
Declaration for Clinician 4
Name: Dr. Mohammad Rassouli
Position: OH-CCO Lung DAC Member
Date: 16-02-2022
Table 34: Conflict of Interest Declaration for Ontario Health (Cancer Care Ontario) Lung Cancer Drug Advisory Committee — Clinician 4
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 |
|---|---|---|---|---|
No COI | — | — | — | — |
Declaration for Clinician 5
Name: Dr. Sara Kuruvilla
Position: OH-CCO Lung DAC Member
Date: 16-02-2022
Table 35: Conflict of Interest Declaration for Ontario Health (Cancer Care Ontario) Lung Cancer Drug Advisory Committee — Clinician 5
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 |
|---|---|---|---|---|
No COI | — | — | — | — |
Ontario Health (Cancer Care Ontario) Head, Neck and Thyroid Cancer Drug Advisory Committee
About Ontario Health (Cancer Care Ontario) Head, Neck and Thyroid Cancer Drug Advisory Committee
OH-CCO’s Drug Advisory Committees provide timely evidence-based clinical and health system guidance on drug-related issues in support of CCO’s mandate, including the Provincial Drug Reimbursement Programs (PDRP) and the Systemic Treatment Program.
Information Gathering
Discussed jointly via email.
Current Treatments
Current treatments depend upon the type of solid tumour.
Salivary gland tumours are a rare subset of cancers that do not have robust evidence for efficacious treatment. If possible, a salivary gland tumour undergoes molecular testing, and therapy is directed towards any actionable mutation (HER2, AR, NTRK). Otherwise, chemotherapy (combination or single agent) or best supportive care represent the standard therapy for salivary gland cancers.
Metastatic iodine refractory thyroid cancers are typically monitored or treated with local therapy until systemic therapy is started for rapidly progressive or widespread disease. Standard therapy is with multi-TKIs.
Treatment Goals
Cancer shrinkage, improvement in severity of cancer-related symptoms, maintain/improve quality of life, delay disease progression
Treatment Gaps (Unmet Needs)
Considering the treatment goals, please describe goals (needs) that are not being met by currently available treatments.
Currently available chemotherapy for salivary gland tumours has a poor response rate and significant toxicity. Treatments are needed that are better tolerated and more efficacious.
Currently available multi-TKI treatments for thyroid cancers are toxic, do not help every patient who tries them, and only help for a limited duration of time. They may significantly impair quality of life and cause serious morbidity due to side effects.
Which patients have the greatest unmet need for an intervention such as the drug under review?
Patients with salivary gland and thyroid cancers with NTRK mutations.
Place in Therapy
How would the drug under review fit into the current treatment paradigm?
This drug would ideally be used as a first line therapy if an NTRK mutation was identified prior to treatment initiation. However, it could also be used as a later line of therapy for those who are already on a different systemic therapy at the time of disease progression or intolerance to that therapy.
Please indicate whether or not it would be appropriate to recommend that patients try other treatments before initiating treatment with the drug under review. Please provide a rationale from your perspective.
Given its targeted nature, high likelihood of response, and favourable toxicity profile, we do not believe that it would be appropriate to recommend other treatments first.
How would this drug affect the sequencing of therapies for the target condition?
This would push back the currently available treatments to later lines of therapy.
Which patients would be best suited for treatment with the drug under review?
Patients with NTRK mutations. Within head and neck cancers, this is most commonly found in salivary gland tumours and thyroid cancers.
How would patients best suited for treatment with the drug under review be identified?
Molecular testing should be routinely performed on advanced/metastatic salivary and thyroid cancers. Cancer centres already have this technology in place to perform this test for other types of solid tumours.
Which patients would be least suitable for treatment with the drug under review?
Patients without NTRK mutations. Patients with early stage/resectable cancers.
Is it possible to identify those patients who are most likely to exhibit a response to treatment with the drug under review?
Yes, by identifying an NTRK mutation.
What outcomes are used to determine whether a patient is responding to treatment in clinical practice?
Monitoring of clinical symptoms and improvement/progression of cancer-related symptoms.
Monitoring of physical exam findings related to the cancer (ie. lymphadenopathy).
Monitoring of disease on imaging.
What would be considered a clinically meaningful response to treatment?
Improvement in cancer-related symptoms. Shrinkage or stabilization of cancer.
How often should treatment response be assessed?
Approximately every 3 months.
What factors should be considered when deciding to discontinue treatment?
Unequivocal disease progression. Treatment intolerance.
What settings are appropriate for treatment with the drug under review?
Oral therapy is taken at home. Prescribed by specialist (medical oncologist).
For non-oncology drugs, is a specialist required to diagnose, treat, and monitor patients who might receive the drug under review?
Not applicable.
Additional Information
Not applicable.
Conflict of Interest Declarations — Ontario Health (Cancer Care Ontario) Head, Neck and Thyroid Cancer Drug Advisory Committee
To maintain the objectivity and credibility of the CADTH drug review programs, all participants in the drug review processes must disclose any real, potential, or perceived conflicts of interest. This conflict of interest declaration is required for participation. Declarations made do not negate or preclude the use of the clinician group input. CADTH may contact your group with further questions, as needed. Please see the Procedures for CADTH Drug Reimbursement Reviews (section 6.3) for further details.
Did you receive help from outside your clinician group to complete this submission? If yes, please detail the help and who provided it.
OH-CCO provided secretariat support to the DAC in completing this input.
Did you receive help from outside your clinician group to collect or analyze any information used in this submission? If yes, please detail the help and who provided it.
No.
List any companies or organizations that have provided your group with financial payment over the past two years AND who may have direct or indirect interest in the drug under review. Please note that this is required for each clinician who contributed to the input — please add more tables as needed (copy and paste). It is preferred for all declarations to be included in a single document.
Declaration for Clinician 1
Name: Dr. Stephanie Brule
Position: Ontario Health Cancer Care Ontario Head, Neck and Thyroid Drug Advisory Member
Date: 18-02-2022
Table 36: Conflict of Interest Declaration for Ontario Health (Cancer Care Ontario) Head, Neck and Thyroid Cancer Drug Advisory Committee — Clinician 1
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 |
|---|---|---|---|---|
No COI | — | — | — | — |
Declaration for Clinician 2
Name: Dr. Michael Odell
Position: Ontario Health Cancer Care Ontario Head, Neck and Thyroid Drug Advisory Lead
Date: 18-02-2022
Table 37: Conflict of Interest Declaration for Ontario Health (Cancer Care Ontario) Head, Neck and Thyroid Cancer Drug Advisory Committee — Clinician 2
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 |
|---|---|---|---|---|
No COI | — | — | — | — |
Ontario Health (Cancer Care Ontario) Gastrointestinal Cancer Drug Advisory Committee
About Ontario Health (Cancer Care Ontario) Gastrointestinal Cancer Drug Advisory Committee
OH-CCO’s Drug Advisory Committees provide timely evidence-based clinical and health system guidance on drug-related issues in support of CCO’s mandate, including the Provincial Drug Reimbursement Programs (PDRP) and the Systemic Treatment Program.
Information Gathering
Discussed jointly at a DAC meeting.
Current Treatments
For patients with advanced GI cancers included in this study (colorectal, pancreas, cholangio, NE), treatments are palliative in nature, and proven sequential lines of therapy are offered to suitable patients (FOLFIRI-FOLFOX-biologics, gemAbraxane-FFX, cisgem-FOLFOX, various, respectively). By nature of being palliative intent, these currently available treatment options provide only limited survival.
Treatment Goals
Prolong life and delay disease progression, improve quality of life and decrease cancer symptoms.
Treatment Gaps (Unmet Needs)
Considering the treatment goals, please describe goals (needs) that are not being met by currently available treatments.
In all the subtypes, the patients are considered palliative. There is an unmet need in GI cancer palliative patients
Which patients have the greatest unmet need for an intervention such as the drug under review?
Palliative GI cancer patients.
Place in Therapy
How would the drug under review fit into the current treatment paradigm?
NTRK mutation reporting would need to be added to NGS clinical panel for GI patients. The DAC acknowledges that this indication would be useful between any of the current lines of therapies. This indication would be an additional line of treatment.
Please indicate whether or not it would be appropriate to recommend that patients try other treatments before initiating treatment with the drug under review. Please provide a rationale from your perspective.
Yes, it would be appropriate to recommend that patients try other treatments before initiating treatment. Depending on the GI cancer, the patient would go through their normal drug sequencing.
How would this drug affect the sequencing of therapies for the target condition?
This would not affect the sequencing of therapies.
Which patients would be best suited for treatment with the drug under review?
Patients with NTRK mutations.
How would patients best suited for treatment with the drug under review be identified?
Patients best suited for treatment with Entrectinib would be identified through NTRK testing.
Which patients would be least suitable for treatment with the drug under review?
Patients with non-NTRK mutation and poor performance status.
Is it possible to identify those patients who are most likely to exhibit a response to treatment with the drug under review?
Yes. Patients would be identified through NTRK testing.
What outcomes are used to determine whether a patient is responding to treatment in clinical practice?
Outcomes used to determine whether a patient is responding would be improvement in cancer symptom control, and objective response by tumour markers and imaging.
What would be considered a clinically meaningful response to treatment?
Reduction in the frequency or severity of symptoms. Response on imaging.
How often should treatment response be assessed?
As per standard GI practices. (2-4 months) Similar response assessments as the current lines of therapy.
What factors should be considered when deciding to discontinue treatment?
Disease progression or toxicity.
What settings are appropriate for treatment with the drug under review?
Cancer center (outpatient) under the management of medical oncologist.
For non-oncology drugs, is a specialist required to diagnose, treat, and monitor patients who might receive the drug under review?
Not applicable.
Additional Information
NTRK testing is included in the typical NGS panels used at most centers but are currently only used clinically for colorectal patients. NGS testing is not funded for other GI cancers. Despite being one of the mutations tested as part of a large panel of mutations for CRC, the results are not provided to clinicians. We would need provincial pathology laps to report the NTRK mutation status. The DAC advocated for companion diagnostic for NTRK testing for all GI cancers included in this drug submission.
Conflict of Interest Declarations — Ontario Health (Cancer Care Ontario) Gastrointestinal Cancer Drug Advisory Committee
To maintain the objectivity and credibility of the CADTH drug review programs, all participants in the drug review processes must disclose any real, potential, or perceived conflicts of interest. This conflict of interest declaration is required for participation. Declarations made do not negate or preclude the use of the clinician group input. CADTH may contact your group with further questions, as needed. Please see the Procedures for CADTH Drug Reimbursement Reviews (section 6.3) for further details.
Did you receive help from outside your clinician group to complete this submission? If yes, please detail the help and who provided it.
OH-CCO provided secretariat support to the DAC in completing this input.
Did you receive help from outside your clinician group to collect or analyze any information used in this submission? If yes, please detail the help and who provided it.
No.
List any companies or organizations that have provided your group with financial payment over the past two years AND who may have direct or indirect interest in the drug under review. Please note that this is required for each clinician who contributed to the input — please add more tables as needed (copy and paste). It is preferred for all declarations to be included in a single document.
Declaration for Clinician 1
Name: Dr. Erin Kennedy
Position: Ontario Health Cancer Care Ontario Gastrointestinal Cancer Drug Advisory Committee Lead
Date: 18-02-2022
Table 38: Conflict of Interest Declaration for Ontario Health (Cancer Care Ontario) Gastrointestinal Cancer Drug Advisory Committee — Clinician 1
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 |
|---|---|---|---|---|
No COI | — | — | — | — |
Declaration for Clinician 2
Name: Dr. Jim Biagi
Position: Ontario Health Cancer Care Ontario Gastrointestinal Cancer Drug Advisory Committee Member
Date: 18-02-2022
Table 39: Conflict of Interest Declaration for Ontario Health (Cancer Care Ontario) Gastrointestinal Cancer Drug Advisory Committee — Clinician 2
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 |
|---|---|---|---|---|
No COI | — | — | — | — |
Declaration for Clinician 3
Name: Dr. Tim Asmis
Position: Ontario Health Cancer Care Ontario Gastrointestinal Cancer Drug Advisory Committee Member
Date: 18-02-2022
Table 40: Conflict of Interest Declaration for Ontario Health (Cancer Care Ontario) Gastrointestinal Cancer Drug Advisory Committee — Clinician 3
Company | $0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 |
|---|---|---|---|---|
Bayer Advisory Board | X | — | — | — |
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up-to-date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
Redactions: Confidential information in this document may be redacted at the request of the sponsor in accordance with the CADTH Drug Reimbursement Review Confidentiality Guidelines.
Stakeholder Input: The views expressed in each submission are those of the submitting organization or individual; not necessarily the views of CADTH or of other organizations. As such, they are independent of CADTH and do not necessarily represent or reflect the view of CADTH. No endorsement by CADTH is intended or should be inferred. By filing with CADTH, the submitting organization or individual agrees to the full disclosure of the information. CADTH does not edit the content of the submissions.
CADTH does use reasonable care to prevent disclosure of personal information in posted material; however, it is ultimately the submitter’s responsibility to ensure no identifying personal information or personal health information is included in the submission. The name of the submitting organization or individual and all conflict of interest information are included in the submission; however, the name of the author, including the name of an individual patient or caregiver submitting the patient input, are not posted.
Accessibility: CADTH is committed to treating people with disabilities in a way that respects their dignity and independence, supports them in accessing material in a timely manner, and provides a robust feedback process to support continuous improvement. All materials prepared by CADTH are available in an accessible format. Where materials provided to CADTH by a submitting organization or individual are not available in an accessible format, CADTH will provide a summary document upon request. More details on CADTH’s accessibility policies can be found here.