CADTH Reimbursement Review
Nivolumab (Opdivo)
Sponsor: Bristol Myers Squibb Canada
Therapeutic area: Urothelial carcinoma
This multi-part report includes:
Clinical Review
Pharmacoeconomic Review
Stakeholder Input
Clinical Review
Abbreviations
AE
adverse event
CI
confidence interval
CrI
credible interval
CSR
Clinical Study Report
CT
computed tomography
CTCAE
Common Terminology Criteria for Adverse Events
DBL
database lock
DFS
disease-free survival
DMFS
distant metastasis-free survival
DSS
disease-specific survival
ECOG
Eastern Cooperative Oncology Group
EMA
European Medicines Agency
EORTC QLQ-C30
European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30
EQ-5D-3L
EuroQol 5-Dimensions 3-Levels questionnaire
GC
gemcitabine + cisplatin
HR
hazard ratio
HRQoL
health-related quality of life
ICI
immune checkpoint inhibitor
IMAE
immune-mediated adverse event
ITC
indirect treatment comparison
IV
intravenous
LRC
locoregional control
LRDFS
locoregional disease-free survival
MIUC
muscle-invasive urothelial carcinoma
MVAC
methotrexate + vinblastine + doxorubicin + cisplatin
MVEC
methotrexate + vinblastine + epirubicin + cisplatin
NA
not available
NMA
network meta-analysis
NMIBC
non–muscle-invasive bladder cancer
NOC
Notice of Compliance
NUTRFS
non-urothelial tract recurrence-free survival
OR
odds ratio
OS
overall survival
PD-1
programmed cell death 1 receptor
PD-L1
programmed death-ligand 1
PD-L2
programmed death-ligand 2
PFS
progression-free survival
PFS2
progression-free survival after next line of subsequent therapy
PS
performance status
RCT
randomized controlled trial
SAE
serious adverse event
TTR
time to recurrence
UC
urothelial carcinoma
UTUC
upper tract urothelial carcinoma
VAS
Visual Analogue Scale
Executive Summary
An overview of the submission details for the drug under review is provided in Table 1.
Item | Description |
|---|---|
Drug product | Nivolumab (Opdivo), 240 mg every 2 weeks or 480 mg every 4 weeks, IV administration |
Indication | As monotherapy for the adjuvant treatment of adult patients with urothelial carcinoma who are at high risk of recurrence after undergoing radical resection of urothelial carcinoma |
Reimbursement request | As per indication |
Health Canada approval status | NOC with conditions |
Health Canada review pathway | Standard |
NOC date | June 27, 2022 |
Sponsor | Bristol Myers Squibb Canada |
NOC = Notice of Compliance.
Introduction
Bladder cancer is the fifth most common cancer in Canada resulting in an estimated 2,600 deaths in 2020, and an estimated 12,500 new cases of bladder cancer were projected in Canada in 2021.1 The most common histological type of bladder cancer is urothelial carcinoma (UC) which typically arises in the bladder but may develop in any location lined with urothelium including the renal pelvis, ureter, urethra, and prostatic urethra.1-4 Approximately 33% to 40% of patients with bladder cancer present with or progress to muscle-invasive disease, and the 5-year survival among patients with localized muscle-invasive urothelial carcinoma (MIUC) is approximately 40% to 50%.5,6
Radical surgery (e.g., cystectomy) with regional lymphadenectomy along with cisplatin-based combination chemotherapy is considered the therapeutic gold standard for MIUC.7 The Canadian Urological Association guideline recommends that eligible patients with muscle-invasive bladder cancer (cT2-T4a N0 M0) should be considered to receive neoadjuvant cisplatin-based combination chemotherapy. There is a lack of high-quality evidence in patients with upper tract UC (UTUC) due to their small number. However, because both share similar etiology, findings for bladder cancer are generalized to patients with UTUC.4 The Canadian Urological Association guideline recommends that adjuvant cisplatin-based chemotherapy should be offered to patients with high risk of recurrence (pT3-T4 and or N+)6 who are eligible for cisplatin-based chemotherapy and have not received neoadjuvant chemotherapy. The National Comprehensive Cancer Network guidelines suggest that adjuvant therapy in bladder cancer may be most suitable for patients who value a delay in recurrence and accept the risk of side effects, even though an increased chance for cure has not been firmly established in this area.8 The 5-year survival rate has been estimated to be 40% to 50% for patients with high-risk residual disease of pT3-pT4 pN- or any pT pN+ at radical cystectomy followed by cisplatin-based chemotherapy upon recurrence.9
The clinical experts and clinician groups consulted by CADTH agreed that there is an unmet need for effective treatment options that improve overall and disease-free survival (DFS) in patients at high risk of disease recurrence at cystectomy. Specifically, the clinical experts felt that there was an unmet need in patients who have not received neoadjuvant chemotherapy and are ineligible for adjuvant cisplatin-based chemotherapy, and in patients who present with significant high-risk features at cystectomy after treatment with neoadjuvant cisplatin-based chemotherapy.
The proposed Health Canada indication and reimbursement request submitted by the sponsor for review by CADTH was for nivolumab (240 mg every 2 weeks or 480 mg every 4 weeks, IV administration) as a monotherapy for the adjuvant treatment of patients with MIUC who are at high risk of recurrence after undergoing radical resection. Nivolumab underwent review by Health Canada through the standard review pathway and received a Notice of Compliance (NOC) with conditions on June 27, 2022; these conditions included the conduct of timely, well-designed studies to verify the clinical benefit of the drug. The Health Canada–approved indication and updated reimbursement request is for nivolumab (240 mg every 2 weeks or 480 mg every 4 weeks, IV administration) as a monotherapy for the adjuvant treatment of adult patients with UC who are at high risk of recurrence after undergoing radical resection of UC. The CADTH review team, in consultation with the clinical experts for this review, agreed with the sponsor’s assessment that this revision to the indication does not meaningfully impact the CADTH clinical review. Nivolumab has been previously reviewed by CADTH as a monotherapy for gastroesophageal junction or esophageal adenocarcinoma, gastroesophageal junction or esophageal adenocarcinoma, metastatic melanoma, and metastatic renal cell carcinoma and in combination with ipilimumab for malignant pleural mesothelioma and non–small cell lung cancer. According to the latest product monograph, nivolumab is also approved for several indications including melanoma, non–small cell lung cancer, malignant pleural mesothelioma, renal cell carcinoma, squamous cell cancer of the head and neck, classical Hodgkin lymphoma, and esophageal or gastroesophageal junction and gastric cancer.10
The objective of this report was to perform a systematic review of the beneficial and harmful effects of nivolumab (IV injection over 30 minutes of 240 mg every 2 weeks) for the adjuvant treatment of patients with MIUC who are at high risk of recurrence after undergoing radical resection of MIUC.
Stakeholder Perspectives
The information in this section is a summary of input provided by the patient groups who responded to CADTH’s call for patient input and from clinical experts consulted by CADTH for the purpose of this review.
Patient Input
One patient advocacy group, Bladder Cancer Canada, provided input for adjuvant treatment of patients with MIUC who are at high risk of recurrence after undergoing radical resection. The group gathered information through online surveys and 1-to-1 telephone interviews and responses from a total of 7 (6 patients from Canada and 1 patient from the US) patients were included in the patient input. All patient respondents (N = 7) reported having been diagnosed with MIUC and 2 patients reported receiving nivolumab (1 patient indicated receiving nivolumab for the adjuvant treatment following radical resection and the other patient reported having received nivolumab in combination with ipilimumab).
When Bladder Cancer Canada asked respondents to indicate their experience with treatments they have undergone since diagnosis, most patient respondents (n = 6) reported having received radical cystectomy. Additional treatments received by patient respondents included gemcitabine + cisplatin (GC) (received by 3 patients each), transurethral resection (received by 2 patients), and methotrexate + vinblastine + doxorubicin + cisplatin (MVAC) and antibody drug conjugates (received by 1 patient each). Patients reported fatigue to be the most common as well as “the most-difficult-to-tolerate” side effect of these treatments, followed by nausea and constipation. Two respondents indicated that they had to be hospitalized due to side effects from treatment.
According to the patient input received, respondents expected new treatments to improve the following key outcomes: preventing recurrence, controlling disease progression, reducing symptoms, maintaining quality of life, and managing side effects. Bladder Cancer Canada indicated that participants rated preventing recurrence as the most important outcome and managing side effects as the least important outcome. According to Bladder Cancer Canada, the patients’ responses were indicative of a willingness to tolerate side effects if treatment was effective. Furthermore, when Bladder Cancer Canada asked specifically about their willingness to tolerate new side effects from treatment that could control disease progression or prevent recurrence, most patient respondents were supportive of tolerating side effects if the treatment showed benefit.
Patient respondents (n = 2) who had direct experience with nivolumab indicated that, overall, nivolumab was an effective treatment, controlling disease progression and preventing recurrence. One patient also reported having improved cancer symptoms, side effects, and quality of life, while the other patient indicated having slightly worse side effects and quality of life. One patient indicated having experienced the following side effects with nivolumab: itchy skin (pruritus) and fatigue. The other patient reported the following side effects from treatment with nivolumab: diarrhea, joint swelling, colitis, and pneumonitis. In addition, this patient experienced immune checkpoint inhibitor (ICI)-related interstitial lung disease. However, since this patient received both nivolumab and ipilimumab, the patient reported that the patient’s treating respirologist did not indicate which drug caused the lung disease. Overall, 1 patient reported that the side effects of nivolumab were completely tolerable, while the other patient noted they were somewhat challenging. Overall, both patient respondents noted that they would recommend nivolumab to other patients with MIUC.
Clinician Input
Input From Clinical Experts Consulted by CADTH
The clinical experts consulted by CADTH agreed that there is an unmet need for effective treatment options that improve overall and DFS in patients at high risk of disease recurrence, who have not received neoadjuvant chemotherapy and are ineligible for adjuvant cisplatin-based chemotherapy for medical reasons; and in patients who present with residual disease at cystectomy after treatment with neoadjuvant cisplatin-based chemotherapy. The clinical experts noted that data on nivolumab compared to cisplatin-based chemotherapy in patients who have not received neoadjuvant chemotherapy and are eligible to received cisplatin-based chemotherapy were not available from the CheckMate 274 trial. Given the absence of robust comparative data between adjuvant nivolumab and adjuvant chemotherapy, the clinical experts consulted by CADTH were uncertain whether nivolumab addressed an unmet need in patients at high risk of recurrence who are eligible for adjuvant cisplatin-based chemotherapy. The clinical experts anticipated that adjuvant nivolumab would be the preferred treatment over adjuvant chemotherapy in select clinical circumstances only (e.g., gemcitabine allergy or strong patient preference against chemotherapy).
If public coverage were available, nivolumab could increase the number of patients who receive adjuvant systemic therapy, as some providers may underutilize perioperative systemic chemotherapy therapy, or do not refer their patients for consideration of treatment. In the expert’s view, the benefits of perioperative cisplatin-based chemotherapy are well established from randomized controlled trials (RCTs), and only patients who are not candidates for this treatment for specific medical reasons or patients at high risk of recurrence despite neoadjuvant chemotherapy should be considered for nivolumab.
In the opinion of the clinical experts, an assessment of effectiveness of treatment should primarily be based on overall survival (OS). DFS may be considered a reasonable surrogate in patients without other treatment options. However, for patients who are eligible for adjuvant chemotherapy, DFS on its own may not be an adequate outcome to guide treatment selection. Patients would be identified based on pathology results following surgery, and knowledge of prior systemic treatments for MIUC. The clinical experts also confirmed that nivolumab should be discontinued if there is disease recurrence or intractable severe adverse effects. As nivolumab is now commonly used and familiar to the oncology community, treatment and monitoring could be done by specialists in community settings.
The pivotal trial, CheckMate 274 also allowed entry of patients “who declined” adjuvant cisplatin-based chemotherapy. Nivolumab would usually have less adverse effects than chemotherapy. The clinical experts were of the opinion that an RCT comparing nivolumab to adjuvant chemotherapy (not placebo) should inform treatment of patients who are suitable for but “who declined” standard adjuvant cisplatin-based chemotherapy.
Clinician Group Input
The views of the clinician groups were consistent with the views of the clinical experts consulted by CADTH. Two clinician groups provided input: Bladder Cancer Canada (a registered national charity) surveyed 6 clinicians, and the Ontario Health (Cancer Care Ontario) Genitourinary Cancer Drug Advisory Committee included input from 3 clinicians. Clinicians from both groups commented that Opdivo would fill a gap in the standard of care for patients with a high risk of recurrence with or without neoadjuvant cisplatin-based chemotherapy, or for patients who are unfit or ineligible for adjuvant cisplatin-based chemotherapy. The clinicians from Bladder Cancer Canada highlighted that many patients recover poorly from surgery and are not fit for adjuvant chemotherapy. All patients with UC with ypT2 or higher or pT3 or higher or node positive would be the target population, which constitutes approximately two-thirds of patients with cystectomy or nephroureterectomy. These patients are often frail or have a solitary kidney and thus cannot receive the current standard of adjuvant chemotherapy. The clinicians from Bladder Cancer Canada noted the following important treatment goals in the adjuvant setting (in order of priority): increasing OS, preventing metastases, controlling disease progression, maintaining quality of life, minimizing adverse events (AEs), and reducing severity of symptoms. Clinicians from both groups agreed that there is some debate on the effectiveness of adjuvant chemotherapy and currently poor use of it in clinical practice. Both groups mentioned that nivolumab would change how MIUC would be treated and it may become the main drug used in the adjuvant setting for patients.
Drug Program Input
The Provincial Advisory Group identified the following jurisdictional implementation issues: eligible patient population, timeline for initiation of therapy, consideration for discontinuation of therapy, relevant comparators, and downstream sequencing of care. The clinical experts consulted by CADTH weighed evidence from the CheckMate 274 study and other clinical considerations to provide responses to the Provincial Advisory Group’s drug program implementation questions. Refer to Table 5 for more details.
Clinical Evidence
Pivotal Studies and Protocol-Selected Studies
Description of Studies
CheckMate 274 was a phase III, randomized, double-blind, placebo-controlled study (N = 709) funded by Bristol Myers Squibb. The primary objective was to compare the DFS for nivolumab versus placebo in all randomized patients and patients with tumours expressing programmed death-ligand 1 (PD-L1) (≥ 1% membranous staining in tumour cells). Secondary objectives included comparing the OS for nivolumab versus placebo in all randomized patient and in patients with tumours expressing PD-L1 (≥ 1% membranous staining in tumour cells), as well as evaluating non-urothelial tract recurrence-free survival (NUTRFS) and disease-specific survival (DSS) in each study group in patients with tumours expressing PD-L1 (≥ 1% membranous staining in tumour cells) and all randomized patients.
After screening, eligible subjects were randomized in a 1:1 ratio to the nivolumab or placebo treatment group and stratified by pathologic nodal status (N+ versus N0/x with < 10 nodes removed versus N0 with ≥ 10 nodes removed), tumour cell PD-L1 expression (≥ 1%, < 1%, or indeterminate), and use of cisplatin-neoadjuvant chemotherapy (yes versus no). All subjects were treated until recurrence of disease, unacceptable toxicity, or withdrawal of consent with a maximum of 1 year of treatment. Tumour imaging assessments were to be performed every 12 weeks from the date of first dose to week 96, then every 16 weeks from week 96 to week 160, then every 24 weeks until non-urothelial tract recurrence or treatment was discontinued (whichever occurred later) for a maximum of 5 years.
The mean ages of patients in the nivolumab and placebo arms were 65.3 years and 65.9 years, respectively, and the nivolumab group had a slightly larger proportion of patients younger than 65 years (155 [43.9%] in nivolumab group and 136 [38.2%] in placebo group). Approximately 75% of patients in both groups were White males; almost 48% were enrolled in Europe, and approximately 14% in the US and 15% in the rest of the world, including Canada. Approximately 79% of patients had a primary tumour in the urinary bladder, almost 74% had PT3 or PT4A at resection, and almost 59% had PD-L1 expression of less than 1%. Regarding prior cancer therapy, almost 43% had received prior neoadjuvant cisplatin therapy, and of those not treated with cisplatin, 123 (34.8%) in the nivolumab group and 108 (30.3%) in the placebo group were unwilling to take cisplatin, while the rest were deemed ineligible. Baseline demographic and disease characteristics were generally well balanced between study arms.
Efficacy Results
Key efficacy results of the CheckMate 274 trial for all randomized patients and all randomized patients with PD-L1 expression 1% or greater are summarized in Table 2. As of the final primary analysis data cut-off date (August 27, 2020), minimum follow-up time was 5.9 months, and median follow-up time among all randomized patients was 20.9 months and 19.5 months in the nivolumab and placebo groups, respectively. Median treatment durations were 8.77 months (range: 0 to 12.5) in the nivolumab group and 8.21 months (range, 0 to 12.6) in the placebo arm. In all randomized patients with tumour cell PD-L1 expression of 1% or greater, the minimum follow-up time was 6.3 months, and the median follow-up was 22.1 months and 18.7 months in the nivolumab and placebo groups, respectively.
OS was a key secondary end point in the CheckMate 274 trial and was not reported in the primary Clinical Study Report (CSR) and OS data were not available from the sponsor at the time of this review. Among all treated patients, there were 95 (27.1%) deaths reported in the nivolumab group and 107 (30.7%) deaths reported in the placebo group. The primary cause of death was disease recurrence (73 [20.8%] in the nivolumab group and 90 [25.9%] in the placebo group).
At the data cut-off date of August 27, 2020, the efficacy analyses of DFS in all randomized patients showed that patients in the nivolumab group had longer DFS than those in the placebo arm. The observed median DFS was longer in the nivolumab group (20.8 months [95% confidence interval {CI}, 16.5 to 27.6] versus 10.8 months [95% CI, 8.3 to 13.9]) compared with the placebo arm (hazard ratio [HR] = 0.70; 98.22% CI, 0.55 to 0.90; log-rank P = 0.0008). The observed median NUTRFS was 22.9 months (95% CI, 19.2 to 33.4) in the nivolumab group and 13.7 months (95% CI, 8.4 to 20.3) in the placebo group, with an HR of 0.72 (95% CI, 0.59 to 0.89). Results from the updated analysis at the February 1, 2021, data cut-off date were consistent with results from the primary analysis.
Among exploratory outcomes, median distant metastasis-free survival (DMFS) was 40.5 months (95% CI, 22.4 to NA [not available]) in the nivolumab group and 29.5 months (95% CI, 16.7 to NA) in the placebo group with an HR of 0.75 (95% CI, 0.59 to 0.94). Time to recurrence (TTR) was 27.0 months (95% CI, NA to 19.5) in the nivolumab group and 11.4 months (95% CI, 20.0 to 8.4) in the placebo group with an HR of 0.67 (95% CI, 0.54 to 0.83). Recurrence rates were higher in the placebo group (37.0%) than in the nivolumab group (23.0%) at 6 months. Results from the updated analysis at the February 1, 2021, data cut-off date were consistent with results from the primary analysis.
Results for patient-reported outcomes (assessed by the European Organization for Research and Treatment of Care Core Quality of Life questionnaire [EORTC QLQ-C30] and EuroQol 5-Dimensions 3-Levels questionnaire [EQ-5D-3L]) suggested similar overall health status in both study groups.
Table 2: Summary of Key Results From the CheckMate 274 Trial From August 27, 2020 (Data Cut-Off Date)
Outcome measure | All randomized patients | Patients with tumour PD-L1 ≥ 1% | ||
|---|---|---|---|---|
Nivolumab n = 353 | Placebo n = 356 | Nivolumab n = 353 | Placebo n = 356 | |
DFS primary definition | ||||
Events | 170 (48.2) | 204 (57.3) | 55 (39.3) | 81 (57.0) |
Median DFS, months (95% CI)a | 20.8 (16.5 to 27.6) | 10.8 (8.3 to 13.9) | Not reached. (21.19 to not reached) | 8.41 (5.59 to 21.19) |
HR (CI)b | 0.70 (98.22% CI, 0.55 to 0.90) | 0.55 (98.72% CI, 0.35 to 0.85) | ||
Stratified log-rank P valuec | 0.0008d | 0.0005e | ||
Rate at 6 months, % (95% CI)a | 74.9 (69.9 to 79.2) | 60.3 (54.9 to 65.3) | 74.5 (66.2 to 81.1) | 55.7 (46.8 to 63.6) |
NUTRFS | ||||
Events | 162 (45.9) | 190 (53.4) | 54 (38.6) | 78 (54.9) |
Median NUTRFS, months (95% CI)a | 22.9 (19.2 to 33.4) | 13.7 (8.4 to 20.3) | Not reached (24.57 to not reached) | 10.84 (5.65 to 22.14) |
HR (95% CI)b | 0.72 (0.59 to 0.89) | 0.55 (0.39 to 0.79) | ||
Rate at 6 months, % (95% CI)a | 77.0 (72.1 to 81.1) | 62.7 (57.3 to 67.6) | 75.3 (67.0 to 81.7) | 56.7 (47.8 to 64.6) |
DMFS | ||||
Events | 132 (37.4) | 152 (42.7) | 47 (33.6) | 61 (43.0) |
Median DMFS, months (95% CI)a | 40.5 (22.4 to not reached) | 29.5 (16.7 to not reached) | Not reached (25.8 to not reached) | 21.19 (10.6 to not reached) |
HR (95% CI)b | 0.75 (0.59 to 0.94) | 0.61 (0.42 to 0.90) | ||
Rate at 6 months, % (95% CI)a | 82.5 (78.0 to 86.2) | 69.8 (64.5 to 74.4) | 78.7 (70.7 to 84.8) | 65.7 (56.8 to 73.3) |
TTR | ||||
Events | 153 (43.3) | 193 (54.2) | 47 (33.6) | 75 (52.8) |
Median TTR, months (95% CI)a | 27.0 (not reached to 19.5) | 11.4 (20.0 to 8.4) | Not reached (not reached to 29.67) | 11.4 (29.6 to 6.5) |
HR (95% CI)b | 0.67 (0.54, 0.83) | 0.51 (0.35, 0.73) | ||
Rate at 6 months, % (95% CI)a | 23.0 (18.7 to 27.6) | 37.0 (31.9 to 42.2) | 23.3 (16.5 to 30.7) | 41.4 (32.9 to 49.6) |
CI = confidence interval; DFS = disease-free survival; DMFS = distant metastasis-free survival; HR = hazard ratio; NUTRFS = non-urothelial tract recurrence-free survival; PD-L1 = programmed death-ligand 1; TTR = time to recurrence.
Note: Values are n (%) unless otherwise indicated. The primary definition of DFS accounted for subsequent anticancer therapy and new non-urothelial carcinoma primary cancer. The secondary definition of DFS accounted for disease assessments occurring on or after initiation of subsequent anticancer therapy.
aBased on Kaplan-Meier estimates.
bStratified Cox proportional hazard model. HR is nivolumab over placebo.
cTwo-sided P values from stratified regular log-rank test.
dLog-rank test stratified by prior neoadjuvant cisplatin, pathological nodal status, PD-L1 status (≥ 1% vs. < 1% or indeterminate) as entered in the interactive response technology.
eLog-rank test stratified by prior neoadjuvant cisplatin, pathological nodal status, as entered in the interactive response technology.
fBased on cumulative incidence estimates.
Source: Primary Clinical Study Report and erratum.9,11
Harms Results
A summary of harms in the CheckMate 274 trial are presented in Table 3. A total of 347 patients (98.9%) in the nivolumab group and 332 patients (95.4%) in the placebo group experienced at least 1 AE, whereas 148 patients (42.2%) in the nivolumab group and 122 patients (35.1%) in the placebo group experienced a grade 3 or higher AE. A total of 29.6% of patients in the nivolumab group and 30.2% of patients in the placebo group experienced an all-causality serious adverse event (SAE). The most common SAEs (≥ 2% in either of the arms) in the nivolumab versus placebo arms were urinary tract infection (2.6% versus 6.0%, respectively) and malignant neoplasm progression (2.3% versus 5.5%, respectively).
All-causality AEs leading to study drug discontinuation occurred in 18.2% of patients in the nivolumab group versus 9.2% in the placebo arm. There were more deaths in the placebo group (107 [30.7%]) than in the nivolumab group (95 [27.1%]), most commonly due to disease progression (20.8% in the nivolumab group and 25.9% in the placebo arm). There were 3 treatment-related deaths: 2 due to pneumonitis and 1 due to bowel perforation.
Immune-mediated adverse events (IMAEs) were identified as notable harms by the clinical experts and were more frequently reported in patients in the nivolumab group than in the placebo arm. They include rash (11.4% versus 2.3%), pneumonitis (4.8% versus 0.6%), diarrhea or colitis (4.0% versus 0.9%), hepatitis (2.8% versus 0.3%), nephritis or renal dysfunction (2.0% versus 0.9%), and hypersensitivity or infusion reactions (0.6% versus 0.0%).
Table 3: Summary of Key Harms Results From the CheckMate 274 Study From August 27, 2020 (Data Cut-Off Date)
Harms | Nivolumab (n = 351) | Placebo (n = 348) |
|---|---|---|
Patients with ≥ 1 AE | 347 (98.9) | 332 (95.4) |
Grade 3 or 4 | 148 (42.2) | 122 (35.1) |
Drug-related AEs | 272 (77.5) | 193 (55.5) |
Patients with ≥ 1 SAE | 104 (29.6) | 105 (30.2) |
Grade 3 or 4 | 81 (23.1) | 73 (21.0) |
Patients who stopped treatment due to AEs | 64 (18.2) | 32 (9.2) |
Grade 3 or 4 | 39 (11.1) | 21 (6.0) |
Deaths | 95 (27.1) | 107 (30.7) |
Primary reason for death | ||
Disease | 73 (20.8) | 90 (25.9) |
Study drug toxicity | 2 (0.6) | 0.0 |
Unknown | 3 (0.9) | 3 (0.9) |
Other | 17 (4.8) | 14 (4.0) |
IMAEsa | ||
Rash | 40 (11.4) | 8 (2.3) |
Pneumonitis | 17 (4.8) | 2 (0.6) |
Diarrhea or colitis | 14 (4.0) | 3 (0.9) |
Hepatitis | 10 (2.8) | 1 (0.3) |
Nephritis or renal dysfunction | 7 (2.0) | 3 (0.9) |
Hypersensitivity or infusion reactions | 2 (0.6) | 0.0 |
AE = adverse event; IMAE = immune-mediated adverse event; SAE = serious adverse event.
Note: Values are n (%) unless otherwise indicated. AEs were defined and graded using MedDRA version 23.0 and Common Terminology Criteria for Adverse Events version 4.0. All events are within 30 days of the last dose of study drug unless otherwise indicated (any time for deaths, 100 days for IMAEs and other events of special interest). Results are from the July 10, 2020, database lock.
aAll-causality IMAEs within 100 days of last dose treated with immune-modulating medication.
Source: CheckMate 274 Clinical Study Report.9
Critical Appraisal
Internal Validity
Despite the trial’s blind design, it is possible that some AEs, such as IMAEs, allowed the possible detection of the intervention being received by some patients. If trial investigators or patients were aware of the intervention assignment, this may have affected behaviour (such as initiation of subsequent treatment given that DFS was investigator assessed or adherence to treatment), imaging assessments, or perceived health-related quality of life (HRQoL). OS was considered an outcome of primary importance by the clinical experts consulted by CADTH in guiding treatment selection in clinical practice. The first interim analysis for OS was planned with the February 1, 2021, data cut-off date at which point OS did not cross the pre-specified boundary for declaring statistical significance.12 No OS data were submitted by the sponsor. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. Updated results for DFS, NUTRFS, DMFS, and TTR from the May 19, 2021, database lock (DBL) (data cut-off February 1, 2021) were overall consistent with results from the final primary analysis. However, these updated results were only available in poster format (poster presentation at Society of Urologic Oncology Congress in December 202113) and no CSR was provided for this data cut-off, so the CADTH review team was unable to conduct a rigorous evaluation of the methods and reporting of these analyses. Maintaining quality of life was rated as an important outcome by patients, yet there was no formal statistical comparison and missing HRQoL data at later time points post-baseline. The interpretation of results for the HRQoL instruments (i.e., the ability to assess trends over time and to make comparisons across treatment groups) is limited by the significant decline in patients available to provide assessment over time.
External Validity
According to the clinical experts consulted by CADTH for this review, the demographic and disease characteristics of the CheckMate 274 study population were reflective of the Canadian population with MIUC. The study protocol was amended based on findings from the CA209275 study (46% of study patients were PD-L1-positive) to cap PD-L1-negative patients included in the study at 54%. The clinical experts consulted by CADTH noted that the PD-L1 biomarker is currently not used in Canadian clinical practice to guide treatment selection in the target population. The experts noted that research on this biomarker’s definitions, methods of measurement, and cut-off values are currently still evolving. The trial capped the proportion of patients with UTUC at 20% as supported by previous studies and confirmed by clinical experts consulted by CADTH. The experts felt that it was reasonable to generalize the results of the CheckMate 274 study to patients with UTUC because of the similar etiology between UTUC and bladder cancer, and because patients with UTUC were included in the pivotal trial and are similarly treated as patients with bladder cancer in Canadian clinical practice. Almost 98% patients under study were Eastern Cooperative Oncology Group (ECOG) Performance Status (PS) 0 or 1 yet the experts expected that, in clinical practice, a higher proportion of patients with ECOG PS 2 may also receive nivolumab because recurrence of the cancer is high and AEs are tolerable. Cisplatin ineligibility was defined using the Galsky criteria,14 which are commonly used in clinical trials and clinical practice. The clinical experts consulted by CADTH noted that experienced clinicians may apply some flexibility in terms of using adjuvant chemotherapy in patients with creatinine clearance of greater than 50 mL/min and those with hearing loss if patients choose to received adjuvant chemotherapy after a discussion of the clinical risks. The reimbursement request is for consideration of nivolumab 240 mg every 2 weeks or 480 mg every 4 weeks; however, the pivotal study only included dosing of 240 mg every 2 weeks. The clinical experts felt that the results of the CheckMate 274 trial could be generalized to a dosing of 480 mg every 4 weeks as this dosing regimen has been previously approved for nivolumab as a monotherapy with other indications.
The study included 3 groups of patients at high risk of disease recurrence: patients who received neoadjuvant cisplatin-based therapy and were therefore not eligible for adjuvant cisplatin-based therapy, patients who did not receive neoadjuvant cisplatin-based therapy and were either cisplatin ineligible (155 [21.9%]), or were cisplatin eligible but refused adjuvant chemotherapy (231 [32.6%]).The clinical experts noted that data on adjuvant nivolumab compared to adjuvant cisplatin-based chemotherapy in patients who have not received neoadjuvant chemotherapy and are eligible to receive cisplatin-based chemotherapy were not available from the CheckMate 274 trial. Given the absence of robust comparative data between adjuvant nivolumab and adjuvant chemotherapy, the clinical experts consulted by CADTH were uncertain whether nivolumab addressed an unmet need in patients at high risk of recurrence who are eligible for adjuvant cisplatin-based chemotherapy. The clinical experts noted that more robust direct evidence from a randomized trial (e.g., a previous RCT in this setting was conducted by Sternberg et al.15) is required to address the comparative effectiveness and safety of nivolumab compared with cisplatin-based chemotherapy in the adjuvant setting. The clinical experts anticipated that adjuvant nivolumab would be the preferred treatment over adjuvant chemotherapy in select clinical circumstances only (e.g., gemcitabine allergy or strong patient preference against chemotherapy). The clinicians from the Cancer Care Ontario Genitourinary Cancer Drug Advisory Committee providing input for this submission concurred with the clinical experts consulted by CADTH in that they noted that the comparative effectiveness between adjuvant nivolumab and chemotherapy is unknown at the moment, and it may be possible that patients eligible for cisplatin-based adjuvant chemotherapy may be better suited for chemotherapy than nivolumab. These clinicians noted that currently neither adjuvant nivolumab (long-term OS results are awaited from the CheckMate 274 trial) nor adjuvant chemotherapy have demonstrated an OS benefit versus surveillance. The CheckMate 274 trial was not designed to detect differences in treatment effects across subgroups of cisplatin-eligible versus cisplatin-ineligible patients and the clinical experts noted that any assumption about the extent to which the subgroup of cisplatin-eligible patients may have influenced the results seen in the overall trial population is speculative.
A review of studies assessing the appropriateness of DFS as a surrogate outcome is presented in Appendix 5. At the individual level, there was a moderate to substantial agreement between DFS and OS. However, in the absence of the trial-level association between DFS and OS in the present target population, it cannot be firmly concluded to what extent the improvements in DFS observed in patients in the nivolumab group of the CheckMate 274 trial would translate into OS benefits. The clinical experts consulted by CADTH anticipated that in the comparison of adjuvant nivolumab against an active comparator (e.g., adjuvant chemotherapy), primarily OS rather than DFS, would guide treatment selection in the adjuvant setting.
Indirect Comparisons
Indirect evidence from 2 network meta-analyses (NMAs) (1 sponsor-submitted NMA and 1 published NMA) evaluated the effectiveness of nivolumab compared to cisplatin-based chemotherapy in the treatment of UC. They address a gap in the pivotal clinical trial which includes a subgroup of patients that are cisplatin eligible but who were unwilling to take it.
Description of Studies
A total of 5 randomized trials comprising 854 patients were included in the sponsor-submitted indirect treatment comparison (ITC).16 The list of comparators included for the analysis included GC, MVAC, and methotrexate + vinblastine + epirubicin + cisplatin (MVEC). The sponsor-submitted ITC included a feasibility assessment using 3 steps: creating networks of evidence based on the systematic review results, conducting a heterogeneity assessment to explore whether there were systematic differences among the studies included in the network across treatment comparisons that might affect the validity of the NMA results, and testing of the proportional hazard assumption for the CheckMate 274 trial.
The published NMA included 9 studies comprising 2,444 patients: 2 studies involved an assessment of investigator’s choice immunotherapy (nivolumab [n = 353] and atezolizumab [n = 406]) including the pivotal study of this review,17 5 studies involved assessment of cisplatin-based chemotherapy (n = 468, regimens include: cisplatin; GC; cisplatin, vinblastine, and methotrexate; MVAC or MVEC; cisplatin, doxorubicin, and cyclophosphamide) in patients with bladder UC, and 2 studies involved assessment of cisplatin- or platin-based chemotherapy (gemcitabine with cisplatin or carboplatin) in patients with UTUC. The authors conducted an NMA using random and fixed-effect models with a Bayesian approach to compare treatments directly and indirectly with observation or placebo as the common comparator arm. Arm-based analyses were performed to estimate the odds ratio (OR) and 95% credible interval (Crl) to evaluate the disease progression rate in bladder UC and UTUC separately.
Efficacy Results
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
In the published NMA, in patients with bladder UC, chemotherapy (OR = 0.50; 95% CrI, 0.19 to 1.06), atezolizumab (OR = 1.01; 95% CrI, 0.19 to 5.46), and nivolumab (OR = 0.59; 95% CrI, 0.11 to 3.34) did not lower the likelihood of disease progression compared to observation or placebo. In patients with UTUC, chemotherapy (OR = 0.36; 95% CrI, 0.13 to 0.92) was significantly associated with a lower likelihood of disease progression compared to observation or placebo. On the other hand, atezolizumab (OR = 1.39; 95% Crl, 0.28 to 7.25) and nivolumab (OR = 1.21; 95% CrI, 0.29 to 4.95) were not associated with a lower likelihood of disease progression compared to observation or placebo.
Harms Results
Both ITCs did not assess harms outcomes due to heterogeneity in the reporting of AEs across individual studies.
Critical Appraisal
Both NMAs included a limited number of studies with heterogeneity across these studies. In the sponsor-submitted ITC, there was heterogeneity in the tumour staging of patients, definition of end points, treatment doses and regimens, and median follow-up times. Moreover, 3 studies were single-centre studies in Europe. There were wide CrIs observed for all the treatment options reflecting uncertainty in the evidence base, indicating that results should be interpretated with caution. In the published NMA, there was heterogeneity in the components of the chemotherapy regimen and the median follow-up time. Four trials were older chemotherapy trials with smaller sample sizes and inconsistent reporting of outcomes which may have led to confounding of the results. In both ITCs, the methodological concerns identified and the observed heterogeneity across study designs and populations precluded drawing definitive conclusions about the comparative effectiveness of adjuvant nivolumab versus adjuvant chemotherapy.
Conclusions
One sponsor-submitted, ongoing, phase III, multinational, double-blind, randomized, placebo-controlled trial provided evidence regarding the efficacy and safety of nivolumab compared with placebo in patients at high risk of recurrence after radical resection of MIUC (with primary site either in the bladder or upper urinary tract). Compared to placebo, adjuvant treatment with nivolumab (240 mg every 2 weeks IV infusion until disease recurrence or unacceptable toxicity for a total treatment duration of 1 year) showed a statistically significant DFS benefit in the treatment of patients (aged ≥ 18 years) with completely resected MIUC. The absolute difference in median DFS between treatment groups (approximately 10 months) was considered clinically meaningful by the clinical experts consulted by CADTH in patients at high risk of recurrence who are ineligible to receive adjuvant cisplatin-based chemotherapy. Results for OS were not available at the time of this review. While some evidence suggests individual-level associations between DFS and OS, trial-level associations between DFS and OS have not been assessed in the target population. Therefore, it cannot be firmly concluded to what extent the improvements in DFS observed in patients in the nivolumab group of the CheckMate 274 trial would translate into OS benefits. HRQoL analyses were descriptive only and limited by high rates of missing data; thus, changes over time could not be interpreted. Data on adjuvant nivolumab compared to adjuvant cisplatin-based chemotherapy in patients at high risk of recurrence who are eligible to received cisplatin-based chemotherapy were not available from the CheckMate 274 trial. ITCs of nivolumab with cisplatin-based chemotherapy favoured chemotherapy, but the methodological concerns identified and the observed heterogeneity across study designs and populations precluded drawing definitive conclusions about the comparative effectiveness of adjuvant nivolumab versus adjuvant chemotherapy. Given the lack of robust comparative data between adjuvant nivolumab and adjuvant chemotherapy, the clinical experts consulted by CADTH were unsure if adjuvant nivolumab addressed an unmet need in patients who are at high risk of recurrence and eligible for adjuvant chemotherapy.
The safety profile of nivolumab in this study was consistent with the known safety profile of nivolumab, and no additional safety signals were identified with adjuvant nivolumab therapy in this study.
Introduction
Disease Background
Bladder cancer is the fifth most common cancer in Canada resulting in an estimated 2,600 deaths in 2020. According to the Canadian Cancer Society, approximately 12,500 new cases of bladder cancer were projected in Canada in 2021.1 UC, also known as transitional cell carcinoma, is the most common histological type of bladder cancer, accounting for 90% to 95% of cases.1-4 UC typically arises in the bladder (90% of cases) but may also develop as UTUC in the renal pelvis (8% of cases) or the ureter and urethra (2% of cases).1,8 Age, tobacco use, chemical carcinogens, family history, arsenic exposure, and use of indwelling catheters are known risk factors for bladder cancer. Although the reason is unknown, bladder cancer is more common in males.2,5,18,19
UC can be characterized as non–muscle-invasive bladder cancer (NMIBC), muscle-invasive bladder cancer (MIBC) that starts at T2 staging, or metastatic, based on the extent of invasion into the wall of the bladder. Nearly 26% of bladder cancers cases are muscle-invasive at diagnosis and between 10% and 20% of NMIBC cases progress to muscle-invasive disease. This leads to approximately 33% to 40% of patients with bladder cancer that present with or develop muscle-invasive disease.5 Five-year survival among patients with localized MIBC is approximately 40% to 50%.6
MIBC is diagnosed initially with cystoscopy and the diagnosis is confirmed with a transurethral resection of bladder tumour with adequate sampling of muscularis propria and clearance of all macroscopic disease, and imaging of the upper urinary tract.6,7 Transurethral resection of bladder tumour pathology includes examination of the histological type of the tumour, depth of invasion, grade, presence of carcinoma in situ, and lymphovascular invasion.
Standards of Therapy
According to the clinical experts consulted by CADTH, the primary goal of treatment is cure. Radical surgery (e.g., cystectomy) with regional lymphadenectomy along with neoadjuvant cisplatin-based combination chemotherapy is considered the preferred treatment in the guidelines for patients with MIBC.7 For patients who are medically inoperable (e.g., elderly or with comorbidities), or who prefer bladder preservation, radiation therapy with concurrent radio-sensitizing chemotherapy is offered. The Canadian Urological Association guideline recommends that eligible patients with MIBC (cT2-T4a N0 M0) should be considered to receive neoadjuvant cisplatin-based combination chemotherapy.6 It is estimated that approximately 20% of patients receive neoadjuvant chemotherapy.20 Preference to bypass neoadjuvant chemotherapy may be based on concerns delaying surgery, risk of venous thromboembolism, treatment-related mortality, and the non-selective nature of neoadjuvant treatment.6 There is a lack of high-quality evidence in patients with UTUC due to their small number. However, because both share similar etiology, findings for bladder cancer are generalized to patients with UTUC.4
Although OS benefit has not been demonstrated with adjuvant chemotherapy based on phase III trial evidence,21 the clinical experts and clinician groups consulted by CADTH and National Comprehensive Cancer Network clinical guidelines8 agree there are benefits to offering adjuvant cisplatin-based chemotherapy to patients with residual disease (pT3-T4 and or N+)6 who are eligible for cisplatin-based chemotherapy and have not received neoadjuvant chemotherapy. Evidence on chemotherapy in the adjuvant setting often suffers from poor accrual, early termination, and lack of power.6 A recent phase III trial in patients with bladder cancer of pT3-pT4 or pN1 to 3 after radial cystectomy demonstrated a significant progression-free survival (PFS) benefit (HR = 0.54; 95% CI, 0.40 to 0.73) in patients treated with adjuvant cisplatin-based chemotherapy compared with deferred adjuvant treatment until relapse.15 Another phase III trial in patients with UTUC that compared adjuvant chemotherapy with surveillance, showed a DFS advantage over surveillance (HR = 0.45; 95% CI, 0.30 to 0.68).22 The National Comprehensive Cancer Network guidelines suggest that adjuvant therapy in bladder cancer may be most suitable for patients who value a delay in recurrence and accept the risk of side effects, even though an increased chance for cure has not been firmly established in this area.8 Most recurrences after radical cystectomy occur within 2 years with poor prognosis23; median post-recurrence OS has been estimated to be approximately 6 months.24,25 Advanced pathological stage, positive surgical margins, high lymph node density, and early recurrence may lead to poorer outcome.24 Patients with stage T3 or T4 disease were found to have a shorter mean TTR (mean = 12.9 months) than patients with stage T2 disease (mean = 22.7 months).26 The recurrence rates after surgery were 5%, 20%, and 40% for patients with stage T1, T2, and T3 bladder cancer, respectively.27 Seventy-five percent to 80% of patients and 20% to 45% of patients, respectively, survived for 5 years after neoadjuvant cisplatin-based chemotherapy with less than pT2N0 stage and pT2N0 or greater residual disease at radical cystectomy.9 The 5-year survival rate was 40% to 50% for patients with high-risk residual disease of pT3-pT4 pN- or any pT pN+ at radical cystectomy followed by cisplatin-based chemotherapy upon recurrence.9 Some patients may be cisplatin ineligible according to the Galsky criteria which include creatinine clearance less than 60 mL/min, Common Terminology Criteria for Adverse Events (CTCAE) version 4, grade 2 or above, audiometric hearing loss, grade 2 peripheral neuropathy, ECOG PS of 2, or New York Heart Association Class III or IV heart failure.14 Patients who are ineligible for neoadjuvant cisplatin chemotherapy proceed directly to surgery. The clinical experts consulted by CADTH agreed that currently the percentage of patients receiving neoadjuvant chemotherapy is likely less than 50% in clinical practice. While some patients are cisplatin ineligible, preference to bypass neoadjuvant chemotherapy may be based on concerns delaying surgery, risk of venous thromboembolism, treatment-related mortality, and the non-selective nature of neoadjuvant treatment.6
The clinical experts and clinician groups consulted by CADTH agreed that there is an unmet need for effective treatment options that improve overall and DFS in patients at high risk of disease recurrence at cystectomy. Specifically, the clinical experts felt there was an unmet need in patients, who have not received neoadjuvant chemotherapy and are ineligible for adjuvant cisplatin-based chemotherapy; and in patients who present with significant high-risk features at cystectomy after treatment with neoadjuvant cisplatin-based chemotherapy. The clinical experts noted that the latter group of patients has a poor prognosis and requires additional effective adjuvant therapy but does not benefit from additional cisplatin-based chemotherapy. While patients who are not eligible to receive neoadjuvant chemotherapy currently do not have any active adjuvant treatment options, patients eligible for cisplatin-based chemotherapy who have not received neoadjuvant chemotherapy and are at high risk of recurrence after surgery (pT3/T4 and/or N+) should be offered adjuvant cisplatin-based chemotherapy according to the Canadian Urological Association guidelines.6
Drug
The proposed Health Canada indication and reimbursement request initially submitted by the sponsor for review by CADTH was for nivolumab as a monotherapy for the adjuvant treatment of patients with MIUC who are at high risk of recurrence after undergoing radical resection of MIUC. Nivolumab underwent review by Health Canada through the standard review pathway and received an NOC on June 27, 2022; these conditions included the conduct of timely, well-designed studies to verify the clinical benefit of the drug.28 Subsequently, this reimbursement request was revised to align with the indication on the NOC: as a monotherapy for the adjuvant treatment of adult patients with UC who are at high risk of recurrence after undergoing radical resection of UC. The Health Canada rationale for this revision was that 7% (n = 50) of the patients in the pivotal trial presented with no muscle-invasive disease in the radical surgery specimen, and the revised indication would better include this patient population. All patients were diagnosed with MIUC before surgery, but 2 factors can cause absence of evidence of muscle-invasive disease after radical cystectomy: neoadjuvant chemotherapy and resection by transurethral resection of bladder tumour. The CADTH review team, in consultation with the clinical experts for this review, agreed with the sponsor’s assessment that this revision to the indication does not meaningfully impact the CADTH clinical review. The initial indication was approved by the FDA on August 19th, 2021, and is currently under review at the National Institute for Health and Care Excellence, European Medicines Agency (EMA), and Australian Therapeutic Goods Administration.
Nivolumab is a human IgG4 monoclonal antibody directed against programmed cell death protein 1 (PD-1). Binding of nivolumab to PD-1 blocks its interaction with its ligands PD-L1 and programmed death-ligand 2 (PD-L2). Upregulation of PD-L1 and PD-L2 occurs in some tumours and inhibits antitumour T-cell responses. Thus, nivolumab can release PD-1 pathway-mediated inhibition of antitumour immunity resulting in decreased tumour growth. Nivolumab has been previously reviewed by CADTH as a monotherapy for gastroesophageal junction or esophageal adenocarcinoma, gastroesophageal junction or esophageal adenocarcinoma, metastatic melanoma, and metastatic renal cell carcinoma, and in combination with ipilimumab for MPM and non–small cell lung cancer. According to the latest product monograph, nivolumab is also approved for several indications including melanoma, non–small cell lung cancer, malignant pleural mesothelioma, renal cell carcinoma, squamous cell cancer of the head and neck, classical Hodgkin lymphoma, and esophageal or gastroesophageal junction and gastric cancer.10
Key characteristics of nivolumab are shown in Table 4. Nivolumab is available as IV infusion (sterile solution of injection, 40 mg nivolumab per 4mL and 100 mg nivolumab per 10 mL). The recommended dose is 240 mg every 2 weeks (30-minute IV infusion) or 480 mg every 4 weeks (30-minute IV infusion).10
Table 4: Key Characteristics of Nivolumab and Cisplatin-Based Adjuvant Chemotherapy for MIUC
Detail | Nivolumab | Cisplatin-based chemotherapy |
|---|---|---|
Mechanism of action | Blockade of PD-1: PD-L1/PD-L2 interaction and release of antitumour T-cell responses | Interacts with DNA to form DNA adducts: regulates protein kinase and activates p53 leading to a series of signalling cascade and apoptosis in cancer cells |
Indication(s) | As a monotherapy for the adjuvant treatment of adult patients with urothelial carcinoma who are at high risk of recurrence after undergoing radical resection of urothelial carcinoma | As neoadjuvant, adjuvant, and first-line metastatic treatment for urothelial carcinoma |
Route(s) of administration | IV | IV |
Recommended dose | 240 mg every 2 weeks | 3 to 4 cycles (maximum 6 cycles) with methotrexate 30 mg/m2, vinblastine 3 mg/m2, doxorubicin 30 mg/m2, and cisplatin 70 mg/m2; or gemcitabine 1,000 mg/m2 and cisplatin 70 mg/m2 |
Serious adverse effects or safety issues | Severe and/or fatal immune-mediated adverse reactions | Myelotoxicity, neurologic toxicity, cardiotoxicity, renal dysfunction, nephrotoxicity, thrombotic microangiopathy, and hepatotoxicity |
MIUC = muscle-invasive urothelial carcinoma; PD-1 = programmed death 1 receptor; PD-L1 = programmed death-ligand 1; PD-L2 = programmed death-ligand 2.
Source: Product monograph for Opdivo; Lerner (2022).7
Stakeholder Perspectives
Patient Group Input
This section was prepared by CADTH staff based on the input provided by patient groups.
One patient advocacy group, Bladder Cancer Canada, provided input for adjuvant treatment of patients with MIUC who are at high risk of recurrence after undergoing radical resection. The group gathered information through online surveys and 1-to-1 telephone interviews and responses from a total of 7 (6 patients from Canada and 1 patient from the US) patients were included in the patient input. All patient respondents (N = 7) reported having been diagnosed with MIUC and 2 patients reported receiving nivolumab (1 patient indicated receiving nivolumab for the adjuvant treatment following radical resection and the other patient reported having received nivolumab in combination with ipilimumab).
When Bladder Cancer Canada asked respondents to indicate their experience with treatments they have undergone since diagnosis, most patient respondents (n = 6) reported having received radical cystectomy. Additional treatments received by patient respondents included GC (received by 3 patients each), transurethral resection (received by 2 patients), and MVAC and antibody drug conjugates (received by 1 patient each). Patients reported fatigue to be the most common as well as “the most-difficult-to-tolerate” side effect of these treatments, followed by nausea and constipation. Two respondents indicated that they had to be hospitalized due to side effects from treatment.
According to the patient input received, respondents expected new treatments to improve the following key outcomes: preventing recurrence, controlling disease progression, reducing symptoms, maintaining quality of life, and managing side effects. Bladder Cancer Canada indicated that participants rated preventing recurrence as the most important outcome and managing side effects as the least important outcome. According to Bladder Cancer Canada, the patients’ responses were indicative of a willingness to tolerate side effects if treatment was effective. Furthermore, when Bladder Cancer Canada asked specifically about their willingness to tolerate new side effects from treatment that could control disease progression or prevent recurrence, most patient respondents were supportive of tolerating side effects if the treatment showed benefit.
Patient respondents (n = 2), who had direct experience with nivolumab indicated that, overall, nivolumab was an effective treatment, controlling disease progression and preventing recurrent. One patient also reported having improved cancer symptoms, side effects, and quality of life, while the other patient indicated having slightly worse side effects and quality of life. One patient indicated having experienced the following side effects with nivolumab: itchy skin (pruritus) and fatigue. The other patient reported the following side effects from treatment with nivolumab: diarrhea, joint swelling, colitis, and pneumonitis. In addition, this patient experienced ICI-related interstitial lung disease. However, since this patient received both nivolumab and ipilimumab, the patient reported that the patient’s treating respirologist did not indicate which drug caused the lung disease. Overall, 1 patient reported that the side effects of nivolumab were completely tolerable, while the other patient noted they were somewhat challenging. Overall, both patient respondents noted that they would recommend nivolumab to other patients with MIUC.
Clinician Input
Input From Clinical Experts Consulted by CADTH
All CADTH review teams include at least 1 clinical specialist with expertise regarding the diagnosis and management of the condition for which the drug is indicated. Clinical experts are a critical part of the review team and are involved in all phases of the review process (e.g., providing guidance on the development of the review protocol, assisting in the critical appraisal of clinical evidence, interpreting the clinical relevance of the results, and providing guidance on the potential place in therapy). The following input was provided by 2 clinical specialists with expertise in the diagnosis and management of MIUC.
Unmet Needs
The clinical experts consulted by CADTH agreed that there is an unmet need for effective treatment options that improve overall and DFS in patients at high risk of disease recurrence, who have not received neoadjuvant chemotherapy and are ineligible for adjuvant cisplatin-based chemotherapy for medical reasons; and in patients who present with residual disease at cystectomy after treatment with neoadjuvant cisplatin-based chemotherapy. The clinical experts noted that data on nivolumab compared to cisplatin-based chemotherapy in patients who have not received neoadjuvant chemotherapy and are eligible to received cisplatin-based chemotherapy were not available from the CheckMate 274 trial. Given the absence of robust comparative data between adjuvant nivolumab and adjuvant chemotherapy, the clinical experts consulted by CADTH were uncertain whether nivolumab addressed an unmet need in patients at high risk of recurrence who are eligible for adjuvant cisplatin-based chemotherapy. The clinical experts anticipated that adjuvant nivolumab would be the preferred treatment over adjuvant chemotherapy in select clinical circumstances only (e.g., gemcitabine allergy or strong patient preference against chemotherapy).
Place in Therapy
The clinical experts noted that nivolumab would address the unmet needs mentioned previously and would be used as a component of first-line curative adjuvant treatment and as an alternative to cisplatin-based chemotherapy in selected patients only. If public coverage were available, nivolumab could increase the number of patients who receive adjuvant systemic therapy, as some providers may underutilize perioperative systemic chemotherapy therapy, or do not refer their patients for consideration of treatment. In the expert’s view, the benefits of perioperative cisplatin-based chemotherapy are well established from RCTs, and only patients who are not candidates for this treatment for specific medical reasons or patients at high risk of recurrence despite neoadjuvant chemotherapy should be considered for nivolumab. The clinical experts noted that adjuvant nivolumab has not been shown to be noninferior to standard therapy with adjuvant cisplatin-based chemotherapy.
Patient Population
According to the clinical experts, only patients who are not candidates for perioperative cisplatin-based chemotherapy for specific medical reasons or patients at high risk or recurrence despite neoadjuvant chemotherapy should be considered for nivolumab. These patients have the greatest need and potential for benefit. Patients would be identified based on pathology results from radical surgery and knowledge of prior systemic treatments for MIUC. The clinical experts anticipated no issues related to diagnosis, companion diagnostics, or misdiagnosis. Furthermore, the clinical experts noted that as nivolumab is an adjuvant treatment, one cannot directly assess whether patients respond to treatment because they are without evidence of disease. However, lack of effectiveness in terms of cancer relapse is identifiable.
Assessing Response to Treatment
In the opinion of the clinical experts, an assessment of effectiveness of treatment should primary be based on OS. DFS may be considered a reasonable surrogate in patients without other treatment options. However, for patients who are eligible for adjuvant chemotherapy, DFS on its own may not be an adequate outcome to guide treatment selection.
Discontinuing Treatment
The clinical experts note that nivolumab should be discontinued if there is disease recurrence or intractable severe adverse effects.
Prescribing Conditions
According to the clinical experts, a specialist should decide whether treatment with nivolumab is appropriate. As nivolumab is now commonly used and familiar to the oncology community, treatment and monitoring could be done by trained individuals in community settings.
Additional Considerations
The pivotal trial, CheckMate274 also allowed entry of patients “who declined” adjuvant cisplatin-based chemotherapy. Nivolumab would usually have less adverse effects than chemotherapy. The clinical experts were of the opinion that an RCT comparing nivolumab to adjuvant chemotherapy (not placebo) should inform treatment of patients who are suitable for but “who declined” standard adjuvant cisplatin-based chemotherapy.
Clinician Group Input
This section was prepared by CADTH staff based on the input provided by clinician groups.
The views of the clinician groups were consistent with the views of the clinical experts consulted by CADTH. Two clinician groups provided input: Bladder Cancer Canada (a registered national charity) surveyed 6 clinicians, and the Ontario Health (Cancer Care Ontario) Genitourinary Cancer Drug Advisory Committee included input from 3 clinicians. Clinicians from both groups commented that Opdivo would fill a gap in the standard of care for patients with a high risk of recurrence with or without neoadjuvant cisplatin-based chemotherapy, or for patients who are unfit or ineligible for adjuvant cisplatin-based chemotherapy. The clinicians from Bladder Cancer Canada highlighted that many patients recover poorly from surgery and are not fit for adjuvant chemotherapy. All UC patients with ypT2 or higher or pT3 or higher or node positive would be the target population, which constitutes approximately 2-thirds of patients with cystectomy or nephroureterectomy. These patients are often frail or have a solitary kidney and thus cannot receive the current standard of adjuvant chemotherapy. The clinicians from Bladder Cancer Canada noted the following important treatment goals in the adjuvant setting (in order of priority): increased OS, preventing metastases, controlling disease progression, maintaining quality of life, minimizing AEs, and reducing severity of symptoms. Clinicians from both inputs agreed that there is some debate on the effectiveness of adjuvant chemotherapy and currently poor use of it in clinical practice. Both groups mentioned that nivolumab would change how MIUC would be treated and it may become the main drug used in the adjuvant setting for patients.
Drug Program Input
The drug programs provide input on each drug being reviewed through CADTH’s reimbursement review processes by identifying issues that may impact their ability to implement a recommendation. The implementation questions and corresponding responses from the clinical experts consulted by CADTH are summarized in Table 5.
Table 5: Summary of Drug Plan Input and Clinical Expert Response
Drug program implementation questions | Clinical expert response |
|---|---|
Inclusion criteria for the CheckMate 274 trial included:
If recommended for reimbursement, will the trial criteria define the patient population eligible for treatment with nivolumab? | Only patients at high risk of recurrence after radical surgical resection who are ineligible for adjuvant cisplatin-based chemotherapy or patients at high risk of recurrence after radical surgical resection despite neoadjuvant chemotherapy should be considered for adjuvant nivolumab. These patients have the greatest need and potential to benefit from adjuvant nivolumab. |
Patients in the trial were stratified according to their tumour cell PD-L1 expression level (≥ 1%, < 1%, or indeterminant). Is PD-L1 status required in order to be eligible for treatment in this setting? | Evidence from the CheckMate 274 trial demonstrated that adjuvant nivolumab resulted in a statistically significant improvement in DFS in all randomized patients, the majority of which had PD-L1 expression status < 1% (59.5% and 58.7% of patients had PD-L1 expression status < 1% in the nivolumab and placebo groups, respectively). PD-L1 expression level is currently not used to guide treatment decisions in Canadian clinical practice in the present target setting. This area of biomarker analysis is currently still an evolving field of research. |
In the CheckMate 274 trial, subjects were deemed ineligible for adjuvant cisplatin due to any of the following criteria:
Are these criteria consistent with those used in clinical practice to determine if a patient is ineligible for cisplatin therapy? | In the CheckMate 274 trial, patients were deemed ineligible for adjuvant chemotherapy according to the Galsky criteria.14 The Galsky criteria are clinically established criteria used in clinical trials and clinical practice in the present target population. Experienced clinicians may apply some flexibility in terms of using adjuvant chemotherapy in patients with creatinine clearance > 50 mL/min and those with hearing loss if patients prefer to receive adjuvant chemotherapy after a discussion of the clinical risks. |
In the CheckMate 274 study, eligible patients must have had radical surgery withing 120 days before randomization. What is considered the maximum time frame from surgical resection to initiate nivolumab? | 120 days is a reasonable maximum time frame. |
The CheckMate 274 trial did not permit dose modifications due to toxicity; however, treatment with nivolumab could be interrupted or delayed for a maximum period of 6 weeks. If treatment interruptions occur, should the remainder of the doses be given even if it will take more than a year to deliver the treatments (provided there has been no disease progression in between)? | In clinical situations in which treatment with adjuvant nivolumab would be interrupted due to toxicity, treatment should be stopped, or remaining doses given, depending on whether the toxicity was resolved and the nature and severity of the toxicity. If the interruption of treatment with adjuvant nivolumab was due to reasons other than toxicity (e.g., logistic reasons), remaining treatment cycles should also be completed. Remaining doses could be given even if it would take more than a year to deliver the complete treatment with nivolumab. The clinical experts felt that delivering treatment with nivolumab beyond 2 years would likely not be reasonable. |
The nivolumab dose in the CheckMate 274 study was 240 mg IV every 14 days. If funded, in line with other indications for nivolumab, jurisdictions would implement a weight-based dose (3 mg/kg IV every 14 days, up to a maximum of 240 mg). Other indications for nivolumab use extended dosing intervals of every 4weeks (6 mg/kg, up to 480 mg). Is a every-4-week dosing interval of nivolumab appropriate for adjuvant treatment of MIUC? | The CheckMate 274 trial used a nivolumab dose of 240 mg IV every 14 days. Generalizing the trial results to an alternative nivolumab dosing schedule of 480 every 4 weeks (or weight-based dose of 6 mg/kg IV up to 480 mg) seems reasonable. |
Should patients with ECOG Performance Status of ≥ 2 be eligible for nivolumab in this indication? | The CheckMate 274 trial allowed patients to enter the trial if they had an ECOG Performance Status of 0 or 1; patients who did not received cisplatin-based neoadjuvant chemotherapy and were considered ineligible for cisplatin adjuvant chemotherapy, could enter the study with ECOG Performance Status of 2. Most patients in the trial had an ECOG Performance Status of 0 and 1 and only a few patients had an ECOG Performance Status of 2. Given the generally well-tolerated toxicity profile of nivolumab, it would be reasonable to offer adjuvant nivolumab to patients at high risk of recurrence after radical surgical resection up to and inclusive of an ECOG Performance Status of 2. |
Are patients who have undergone a partial cystectomy (or partial nephrectomy in the setting of a renal pelvis tumour) or bladder-preserving chemoradiation eligible for treatment with nivolumab in the adjuvant setting? | The CheckMate 274 trial included patients who had undergone radical survival resection within 120 days before randomization. Patients who had undergone partial cystectomy or partial nephrectomy were excluded. It would be reasonable to generalize the CheckMate 274 trial to patients who have undergone a partial cystectomy or partial nephrectomy if all other trial eligibility criteria are met. Adjuvant nivolumab was not studied in patients who received bladder-preserving chemoradiation in the CheckMate 274 trial. There are no data to generalize the trial results to patients who received bladder-preserving chemoradiation. |
Are patients with bladder cancer of histological subtype other than urothelial carcinoma or transitional cell carcinoma eligible for adjuvant nivolumab? | A minority of patients in the CheckMate 274 trial had a minor histological variant. Patients with any urothelial component in the histological subtype should be eligible for adjuvant nivolumab. |
Are patients with non–muscle-invasive bladder cancer eligible for treatment with adjuvant nivolumab? | The CheckMate 274 trial included patients with MIUC. Results should not be generalized to patients with non–muscle-invasive bladder cancer. |
The current standard of care after surgery is surveillance. For patients who are already on active surveillance, is there a maximum time frame since surgical resection to allow such patients to access nivolumab? | The CheckMate 274 trial allowed treatment with adjuvant nivolumab within 120 days after surgery, which is a reasonable time frame. |
Under what clinical circumstances would adjuvant nivolumab be preferred over adjuvant platinum chemotherapy for those patients who can tolerate platinum? | The CheckMate 274 trial did not assess the comparative efficacy of adjuvant nivolumab compared with adjuvant chemotherapy. Neoadjuvant cisplatin-based chemotherapy before radical surgery has demonstrated some overall survival benefit which may be generalizable to the adjuvant setting. Given the absence of a direct comparison, there is no robust evidence to ascertain which of the agents (i.e., adjuvant nivolumab or adjuvant chemotherapy) has superior efficacy. Given the lack of robust comparative evidence, adjuvant chemotherapy would be the preferred choice unless patients have gemcitabine allergy or there is a strong patient preference against chemotherapy. |
Can the downstream sequencing be clarified (e.g., re-treatment with downstream PD-1 or PD-L1 inhibitor provided the disease recurs more than 6 months from the last dose of adjuvant nivolumab; eligibility for downstream enfortumab vedotin)? | Patients who recur and have only received perioperative nivolumab would be offered gemcitabine plus carboplatin followed by avelumab maintenance therapy (if response or stable disease) or pembrolizumab if progressive disease despite chemotherapy. These patients would be offered enfortumab if their cancer progresses despite these immunotherapies. Patients who recur > 12 months after receiving neoadjuvant followed by nivolumab would receive similar treatment to this. However, those recurring < 12 months after neoadjuvant chemotherapy followed by nivolumab would be offered enfortumab. |
DFS = disease-free survival; ECOG = Eastern Cooperative Oncology Group; MIUC = muscle-invasive urothelial carcinoma; NYHA = New York Heart Association; PD-1 = programmed death 1 receptor; PD-L1 = programmed death-ligand 1; RECIST: Response Evaluation Criteria in Solid Tumors.
Clinical Evidence
The clinical evidence included in the review of nivolumab (Opdivo) is presented in 2 sections. The first section, the Systematic Review, includes pivotal studies provided in the sponsor’s submission to CADTH and Health Canada, as well as those studies that were selected according to an a priori protocol. The second section includes indirect evidence from the sponsor and indirect evidence selected from the literature that met the selection criteria specified in the review. No additional relevant studies addressing important gaps in the evidence were identified for the third section.
Systematic Review (Pivotal and Protocol-Selected Studies)
Objectives
To perform a systematic review of the beneficial and harmful effects of nivolumab (IV injection over 30 minutes of 240 mg every 2 weeks or 480 mg every 4 weeks) for the adjuvant treatment of patients with MIUC who are at high risk of recurrence after undergoing radical resection of MIUC.
Methods
Studies selected for inclusion in the systematic review included pivotal studies provided in the sponsor’s submission to CADTH and Health Canada, as well as those meeting the selection criteria presented in Table 6. Outcomes included in the CADTH review protocol reflect outcomes considered to be important to patients, clinicians, and drug plans.
Of note, the systematic review protocol presented in the following was established before the granting of an NOC from Health Canada.
Table 6: Inclusion Criteria for the Systematic Review
Criteria | Description |
|---|---|
Population | Patients with muscle-invasive urothelial carcinoma who are at high risk of recurrence following radical surgical resection. Subgroups:
|
Intervention | Nivolumab (240 mg every 2 weeks or 480 mg every 4 weeks, IV administration) |
Comparator |
|
Outcomes | Efficacy outcomes:
Harms outcomes:
|
Study designs | Published and unpublished phase III and IV RCTs |
AE = adverse event; HRQoL = health-related quality of life; PD-L1 = programmed death-ligand 1; RCT = randomized controlled trial; SAE = serious adverse event; WDAE = withdrawal due to adverse event.
aThese outcomes were identified as being of particular importance to patients in the input received by CADTH from patient groups.
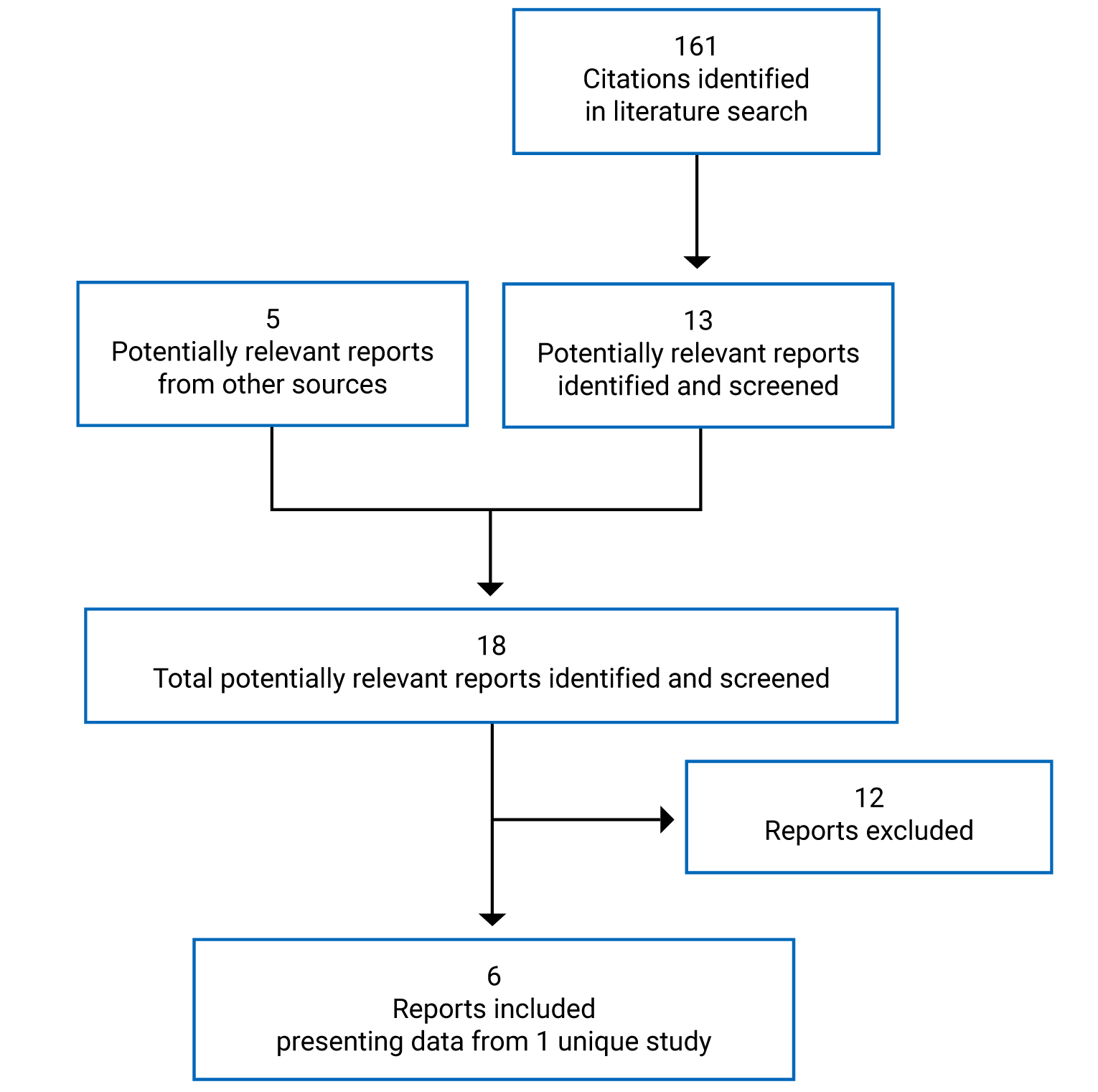
The literature search for clinical studies was performed by an information specialist using a peer-reviewed search strategy according to the PRESS Peer Review of Electronic Search Strategies checklist.29
Published literature was identified by searching the following bibliographic databases: MEDLINE All (1946‒) via Ovid and Embase (1974‒) via Ovid. All Ovid searches were run simultaneously as a multi-file search. Duplicates were removed using Ovid deduplication for multi-file searches, followed by manual deduplication in Endnote. The search strategy comprised both controlled vocabulary, such as the National Library of Medicine’s MeSH (Medical Subject Headings), and keywords. The main search concepts were Opdivo (nivolumab) and UC. Clinical trials registries were searched: the US National Institutes of Health’s clinicaltrials.gov, WHO’s International Clinical Trials Registry Platform search portal, Health Canada’s Clinical Trials Database, and the European Union Clinical Trials Register.
CADTH-developed search filters were applied to limit retrieval to RCTs or controlled clinical trials. Retrieval was not limited by publication date or by language. Conference abstracts were excluded from the search results. Refer to Appendix 1 for the detailed search strategies.
The initial search was completed on March 22, 2022. Regular alerts updated the search until the meeting of the CADTH pan-Canadian Oncology Drug Review Expert Committee on August 10, 2022.
Grey literature (literature that is not commercially published) was identified by searching relevant websites from the Grey Matters: A Practical Tool For Searching Health-Related Grey Literature checklist.30 Included in this search were the websites of regulatory agencies (FDA and EMA). Google was used to search for additional internet-based materials. Refer to Appendix 1 for more information on the grey literature search strategy.
These searches were supplemented by reviewing bibliographies of key papers and through contacts with appropriate experts. In addition, the sponsor of the drug was contacted for information regarding unpublished studies.
Two CADTH clinical reviewers independently selected studies for inclusion in the review based on titles and abstracts, according to the predetermined protocol. Full-text articles of all citations considered potentially relevant by at least 1 reviewer were acquired. Reviewers independently made the final selection of studies to be included in the review, and differences were resolved through discussion.
A focused literature search for NMAs dealing with Opdivo (nivolumab) and UC was run in MEDLINE All (1946–) on March 22, 2022. No limits were applied to the search.
Findings From the Literature
A total of 161 studies were identified from the literature for inclusion in the systematic review (Figure 1). One study was identified from the literature for inclusion in the systematic review. The included study is summarized in Table 7. A list of excluded studies is presented in Appendix 2.
Table 7: Details of Included Study (CheckMate 274)
Component | Description |
|---|---|
Designs and populations | |
Study design | Phase III, randomized, DB, placebo-controlled study |
Locations | 170 sites in 30 countries including Canada (n = 17 patients), US, and from across Europe, South America, and East Asia |
Patient enrolment dates | Initiated: March 23, 2016. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |
Data cut-off dates | Final DFS analysis: August 27, 2020 First interim analysis for OS: February 1, 2021 (OS data were not provided to CADTH by sponsor) ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |
Randomized (N) | 709 (n = 353 in the nivolumab group and n = 356 in the placebo group) |
Inclusion criteria | Men and women, aged ≥ 18 years. Post-radical surgical resection (R0) for IUC performed within 120 days before randomization. Pathologic evidence of urothelial carcinoma (originating in bladder, ureter, or renal pelvis) at high risk of recurrence based on pathologic staging of radical surgery tissue (i or ii):
Dominant component of histology needs to be urothelial carcinoma or transitional cell carcinoma. Tumour tissue provided for biomarker analyses: patient must have a PD-L1 expression level classification (> 1%, < 1%, indeterminate). Life expectancy > 6 months. ECOG PS 0 or 1 (patients who have not received cisplatin-based neoadjuvant chemotherapy and are considered ineligible for cisplatin adjuvant chemotherapy may enter the study with ECOG PS 2). Adequate hematologic, renal, and liver function. All patients must have disease-free status defined as no clinical or radiographic evidence of recurrence of disease documented by a complete physical examination and imaging studies within 4 weeks of randomization. Patients who are found to have high-risk NMIBC at the time of screening are not eligible for study entry. Patients with low-riskb papillary lesions may enter the study if rendered free of disease at cystoscopy. Patients with intermediate-risk NMIBC may enter the study if intravesical chemotherapy or Bacille Calmette-Guérin is not required. |
Exclusion criteria |
|
Drugs | |
Intervention | Nivolumab 240 mg as 30-minute IV infusion every 2 weeks |
Comparator(s) | Placebo (normal saline solution or a 5% dextrose solution) 30-minute IV infusion every 2 weeks |
Duration | |
Phase | |
Screening | 4 weeks |
Treatment | Until toxicity, disease recurrence, or discontinuation from study for maximum of 1 year |
Follow-up | After 1 year of treatment or when the decision is made to discontinue patient from study therapy (e.g., due to toxicity or recurrence). |
Outcomes | |
Primary end point | DFS |
Secondary and exploratory end points | Secondary:
Exploratory:
|
Notes | |
Publicationsa | |
CTCAE = Common Terminology Criteria for Adverse Events; DB = double blind; DFS = disease-free survival; DMFS = distant metastasis-free survival; DSS = disease-specific survival; ECOG PS = Eastern Cooperative Oncology Group Performance Status; EORTC QLQ-C30 = European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30; IUC = invasive urothelial carcinoma; LRDFS = locoregional disease-free survival; LRC = locoregional control; MDSC = myeloid-derived suppressor cells; NMIBC = non–muscle-invasive bladder cancer; NUTRFS = non-urothelial tract recurrence-free survival; OS = overall survival; PD-L1 = programmed death-ligand 1; PD-L1 CPS = programmed death-ligand 1 combined positive score; PFS2 = progression-free survival after next line of subsequent therapy; TTR = time to recurrence.
aOne additional report was included: the Clinical Study Report for the CheckMate 274 study from the submission to CADTH.9 In addition, 3 conference posters related to the pivotal trial were submitted by the sponsor.13,32,33
bLow-risk NMIBC is defined as low-grade lesions or papillary urothelial neoplasms of low malignant potential (papillary urothelial neoplasm of low malignant potential; WHO/International Society of Urological Pathology 2004 grading system), or TaG1 lesions (WHO 1973 grading system) that are less than 3 cm in diameter. High-risk NMIBC is defined as any T1 lesion, any lesion containing carcinoma in situ either alone or concomitantly with papillary disease (e.g., carcinoma in situ with Ta/T1 lesions), and any Ta high-grade (WHO/International Society of Urological Pathology 2004 grading system) or TaG3 (WHO 1973 grading system) lesion. Intermediate-risk NMIBC is defined as lesions not meeting the criteria of high risk or low risk.
Source: CheckMate 274 Clinical Study Report.9
Description of Studies
CheckMate 274 is an ongoing phase III, randomized, double-blind, randomized placebo-controlled study (N = 709) of adjuvant nivolumab compared with placebo in patients with high-risk invasive UC at radical resection. The primary objective was to compare the DFS for nivolumab versus placebo in all randomized patients and in patients with tumours expressing PD-L1 (≥ 1% membranous staining in tumour cells). Secondary objectives included comparing the OS for nivolumab versus placebo in all randomized patients and in patients with tumours expressing PD-L1 (≥ 1% membranous staining in tumour cells), as well as evaluating NUTRFS and DSS in each study group in patients with tumours expressing PD-L1 (≥ 1% membranous staining in tumour cells) and all randomized patients. The study was funded by Bristol Myers Squibb.
After screening, eligible patients were randomized in a 1:1 ratio to the nivolumab or placebo treatment arm and stratified by pathologic nodal status (N+ versus N0/x with < 10 nodes removed versus N0 with ≥ 10 nodes removed), tumour cell PD-L1 expression (≥ 1%, < 1%, or indeterminate), and use of cisplatin-neoadjuvant chemotherapy (yes versus no). All patients were treated until recurrence of disease, unacceptable toxicity, or withdrawal of consent with a maximum of 1 year of treatment.
Baseline disease assessments using CT (CT) or MRI were performed within 28 days before randomization. From the date of the first dose until week 96, tumour imaging was performed every 12 weeks; then from week 96 to week 160, it was performed every 16 weeks; after week 160 it was performed every 24 weeks until non-urothelial tract recurrence or treatment was discontinued for a maximum of 5 years. Patients also had 2 follow-up visits within 100 days from the last dose for safety assessments.
A summary of the design of the CheckMate 274 study is provided in Figure 2. Unless otherwise specified, the data extracted are from the primary CSR with data cut-off date of August 27, 2020.
Figure 2: CheckMate 274 Study Design Schematic
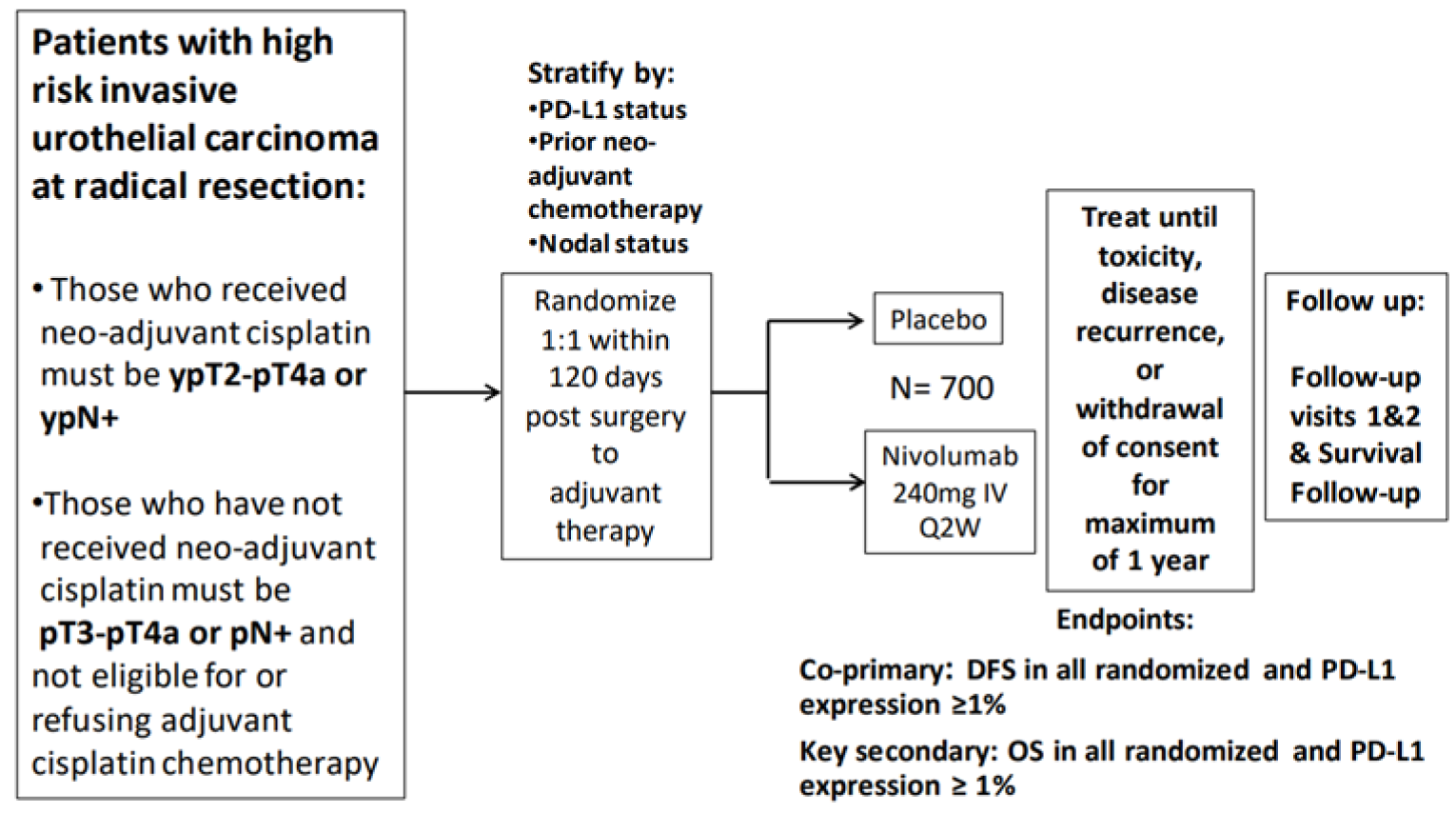
DFS = disease-free survival; OS = overall survival; PD-L1 = programmed death-ligand 1; Q2W = every 2 weeks.
Source: CheckMate 274 Clinical Study Report.9
Populations
Inclusion and Exclusion Criteria
Key inclusion and exclusion criteria for the CheckMate 274 study are shown in Table 7. Adult patients (aged ≥ 18 years) who had undergone radical surgical resection for MIUC were eligible within 120 days of surgery if they were deemed at high risk of recurrence: staged at ypT2-pT4a or ypN+ and had received neoadjuvant cisplatin chemotherapy, or staged at pT3-pT4a or pN+ and had not received neoadjuvant cisplatin chemotherapy because they were ineligible or refused treatment. Patients were eligible if they had ECOG PS 0 or 1, although cisplatin-ineligible patients were allowed to enter the study with ECOG PS 2. Patients who were eligible for adjuvant cisplatin-based chemotherapy but who refused it despite being informed by the investigator about the treatment options were included in the study (patient’s refusal had to be thoroughly documented and eligibility was discussed with the medical monitor for each patient according to the trial protocol).9 All patients were required to have disease-free status to be eligible, but patients with upper urinary tract primaries that had an intact bladder and were found to have low-risk papillary lesions or intermediate-risk NMIBC (with no history of intravesical chemotherapy or Bacille Calmette-Guérin) were allowed to enter the study if rendered free of disease at cystoscopy. All patients were required to have a PD-L1 expression level classification using tumour tissue from the most recently resected site of disease or from the transurethral resection that yielded the initial muscle-invasive diagnosis. Patients with autoimmune diseases or a condition requiring systemic treatment with either corticosteroids (> 10 mg daily prednisone equivalent) or other immunosuppressive medications within 14 days of treatment were excluded. Patients who had undergone partial cystectomy or nephrectomy or who had received adjuvant therapy were also excluded from the study.
Baseline Characteristics
The baseline demographic and disease characteristics of all randomized patients and patients with PD-L1 expression of 1% or greater are presented in Table 8. The mean ages of patients in the nivolumab and placebo arms were 65.3 years and 65.9 years, respectively, and the nivolumab group had a slightly larger proportion of patients younger than 65 years (155 [43.9%] in nivolumab group and 136 [38.2%] in placebo group). Approximately 75% of patients in both arms were White males, almost 48% were enrolled in Europe, and approximately 14% in the US and 15% in the rest of the world, including Canada. Approximately 52% of all randomized patients in both study groups had no nodal involvement (N0). Approximately 79% of all randomized patients in both study groups had a primary tumour in the urinary bladder, almost 74% had PT3 or PT4A at resection, and almost 59% had PD-L1 expression of less than 1%. Regarding prior cancer therapy, almost 43% had received prior neoadjuvant cisplatin therapy, and of those not treated with cisplatin, 123 (34.8%) in the nivolumab group and 108 (30.3%) in the placebo group were unwilling to take cisplatin-based chemotherapy (reasons for refusing cisplatin base chemotherapy were not provided in the sponsor’s submission), while the rest were deemed ineligible. Baseline demographic and disease characteristics were generally well balanced between study arms.
Table 8: Summary of Baseline Characteristics in CheckMate 274 Trial
Characteristic | All randomized patients | Patients with tumour PD-L1 ≥ 1% | ||
|---|---|---|---|---|
Nivolumab (n = 353) | Placebo (n = 356) | Nivolumab (n = 140) | Placebo (n = 142) | |
Age, years | ||||
Mean | 65.3 | 65.9 | 64.4 | 65.9 |
Median (range) | 66 (30 to 92) | 67 (42 to 88) | 66 (34 to 92) | 66 (45 to 84) |
Age category, n (%) | ||||
< 65 years | 155 (43.9) | 136 (38.2) | 67 (47.9) | 61 (43.0) |
≥ 65 years | 198 (56.1) | 220 (61.8) | 73 (52.1) | 81 (57.0) |
|||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| |
Sex, n (%) | ||||
Male | 265 (75.1) | 275 (77.2) | 101 (72.1) | 112 (78.9) |
Female | 88 (24.9) | 81 (22.8) | 39 (27.9) | 30 (21.1) |
Race, n (%) | ||||
White | 264 (74.8) | 272 (76.4) | 104 (74.3) | 109 (76.8) |
Black or African American | 2 (0.6) | 3 (0.8) | 0 | 2 (1.4) |
American Indian or Alaska Native | 1 (0.3) | 0 | 1 (0.7) | 0 |
Asian | 80 (22.7) | 75 (21.1) | 33 (23.6) | 28 (19.7) |
Other | 6 (1.7) | 5 (1.4) | 2 (1.4) | 2 (1.4) |
Not reported | 0 | 1 (0.3) | 0 | 1 (0.7) |
Region, n (%) | ||||
|||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| |
US | 49 (13.9) | 53 (14.9) | 17 (12.1) | 24 (16.9) |
Europe | 170 (48.2) | 171 (48.0) | 73 (52.1) | 70 (49.3) |
Asia | 80 (22.7) | 74 (20.8) | 33 (23.6) | 28 (19.7) |
Rest of the world | 43 (12.2) | 52 (14.6) | 12 (8.5) | 16 (11.3) |
Baseline ECOG performance status | ||||
0 | 224 (63.5) | 221 (62.1) | 86 (61.4) | 85 (59.9) |
1 | 122 (34.6) | 125 (35.1) | 51 (36.4) | 53 (37.3) |
2 | 7 (2.0) | 9 (2.5) | 3 (2.1) | 4 (2.8) |
NA | 0 | 1 (0.3) | 0 | 0 |
Tumour location | ||||
Urinary bladder | 279 (79.0) | 281 (78.9) | 113 (80.7) | 117 (82.4) |
Renal pelvis | 44 (12.5) | 52 (14.6) | 19 (13.6) | 14 (9.9) |
Ureter | 30 (8.5) | 23 (6.5) | 8 (5.7) | 11 (7.7) |
Time from initial disease diagnosis to randomization, years | ||||
< 1 year | 325 (92.1) | 324 (91.0) | 132 (94.3) | 129 (90.8) |
≥ 1 years | 28 (7.9) | 32 (9.0) | 8 (5.7) | 13 (9.2) |
Pathologic stage at resection | ||||
Tumour stage | ||||
PTX | 5 (1.4) | 0 | 4 (2.9) | 0 |
PT0 | 5 (1.4) | 7 (2.0) | 3 (2.1) | 3 (2.1) |
PTIS | 4 (1.1) | 3 (0.8) | 0 | 0 |
PT1 | 13 (3.7) | 14 (3.9) | 4 (2.9) | 2 (1.4) |
PT2 | 62 (17.6) | 65 (18.3) | 19 (13.6) | 26 (18.3) |
PT3 | 206 (58.4) | 204 (57.3) | 87 (62.1) | 83 (58.5) |
PT4A | 57 (16.1) | 62 (17.4) | 23 (16.4) | 27 (19.0) |
Not reported | 1 (0.3) | 1 (0.3) | 0 | 1 (0.7) |
Nodes stage with node density | ||||
N0/x with < 10 nodes removed | 94 (26.6) | 99 (27.8) | 38 (27.1) | 38 (26.8) |
N0 with ≥ 10 nodes removed | 91 (25.8) | 88 (24.7) | 42 (30.0) | 38 (26.8) |
N1 | 71 (20.1) | 72 (20.2) | 29 (20.7) | 33 (23.2) |
N2 | 84 (23.8) | 76 (21.3) | 28 (20.0) | 26 (18.3) |
N3 | 12 (3.4) | 20 (5.6) | 3 (2.1) | 7 (4.9) |
Not reported | 1 (0.3) | 1 (0.3) | 0 | 0 |
|||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| |
Baseline PD-L1 expression status | ||||
|||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| |
Prior cancer therapy summary | ||||
Neoadjuvant therapy | 160 (45.3) | 159 (44.7) | |||||||||||||||||||| | |||||||||||||||||||| |
Prior radiotherapy | 8 (2.3) | 11 (3.1) | |||||||||||||||||||| | |||||||||||||||||||| |
Prior cisplatin therapy | 153 (43.3) | 155 (43.5) | |||||||||||||||||||| | |||||||||||||||||||| |
Reason not treated with cisplatin | ||||
Unwilling to take | 123 (34.8) | 108 (30.3) | |||||||||||||||||||| | |||||||||||||||||||| |
Ineligible, renal function | 53 (15.0) | 53 (14.9) | |||||||||||||||||||| | |||||||||||||||||||| |
Ineligible, neuropathy | 2 (0.6) | 1 (0.3) | |||||||||||||||||||| | |||||||||||||||||||| |
Ineligible, audiometric loss | 4 (1.1) | 15 (4.2) | |||||||||||||||||||| | |||||||||||||||||||| |
Ineligible, performance status | 7 (2.0) | 12 (3.4) | |||||||||||||||||||| | |||||||||||||||||||| |
Ineligible, heart function | 4 (1.1) | 4 (1.1) | |||||||||||||||||||| | |||||||||||||||||||| |
Other | 6 (1.7) | 6 (1.7) | |||||||||||||||||||| | |||||||||||||||||||| |
NA | 1 (0.3) | 2 (0.6) | |||||||||||||||||||| | |||||||||||||||||||| |
Time from completion of prior adjuvant/neoadjuvant therapy to randomization | ||||
0 to 30 days | 1 (0.6) | 0 | |||||||||||||||||||| | |||||||||||||||||||| |
> 30 to 60 days | 0 | 1 (0.6) | |||||||||||||||||||| | |||||||||||||||||||| |
> 60 to 90 days | 18 (11.3) | 20 (12.6) | |||||||||||||||||||| | |||||||||||||||||||| |
> 90 to 120 days | 51 (31.9) | 54 (34.0) | |||||||||||||||||||| | |||||||||||||||||||| |
> 120 days | 90 (56.3) | 83 (52.2) | |||||||||||||||||||| | |||||||||||||||||||| |
NA | 0 | 1 (0.6) | |||||||||||||||||||| | |||||||||||||||||||| |
Type of surgerya | ||||
Radical cystectomy | 88 (24.9) | 93 (26.1) | |||||||||||||||||||| | |||||||||||||||||||| |
Radical cystoprostatectomy | 192 (54.4) | 186 (52.2) | |||||||||||||||||||| | |||||||||||||||||||| |
Radical nephroureterectomy | 71 (20.1) | 73 (20.5) | |||||||||||||||||||| | |||||||||||||||||||| |
Radical ureterectomy | 2 (0.6) | 2 (0.6) | |||||||||||||||||||| | |||||||||||||||||||| |
Other | 1 (0.3) | 3 (0.8) | |||||||||||||||||||| | |||||||||||||||||||| |
Time from surgery to randomization | ||||
0 to 30 days | 2 (0.6) | 3 (0.8) | |||||||||||||||||||| | |||||||||||||||||||| |
> 30 to 60 days | 79 (22.4) | 70 (19.7) | |||||||||||||||||||| | |||||||||||||||||||| |
> 60 to 90 days | 165 (46.7) | 177 (49.7) | |||||||||||||||||||| | |||||||||||||||||||| |
> 90 to 120 days | 103 (29.2) | 95 (26.7) | |||||||||||||||||||| | |||||||||||||||||||| |
> 120 days | 4 (1.1) | 11 (3.1) | |||||||||||||||||||| | |||||||||||||||||||| |
ECOG = Eastern Cooperative Oncology Group; NA = not applicable; PD-L1 = programmed death-ligand 1.
Note: Redacted rows have been deleted.
aSome patients may have been treated with more than 1 type of therapy.
Source: CheckMate 274 Clinical Study Report.9
Interventions
Patients were randomized in a blinded fashion 1:1 to receive either nivolumab 240 mg IV infusion over 30 minutes every 2 weeks for a maximum duration of 1 year (n = 353) or placebo IV infusion over 30 minutes with the same dosing schedule as nivolumab (n = 356). The sponsor, patients, investigator, and site staff were blinded, and an unblinded pharmacist and unblinded site monitor were assigned in each investigative site. Maximum treatment duration was 1 year. Discontinuation criteria included any grade 2 drug-related uveitis or eye pain or blurred vision not responding to topical therapy or requiring systemic treatment, any grade 3 non-skin, drug-related AE lasting more than 7 days (with some exceptions), any grade 4 drug-related AE or laboratory abnormality (with some exceptions), and any dosing delay lasting greater than 6 weeks from the last dose (except when delay is to allow for prolonged steroid tapers or is due to non-drug-related reasons).
Dose Modifications
Nivolumab or placebo dose reductions were not permitted for the management of toxicities of individual patients according to the protocol; however, dose reductions were reported in both arms in the study. Doses of nivolumab and placebo were allowed to be interrupted, delayed, or discontinued depending on how well the patient tolerated the treatment. Dose interruption refers to interruption of the actual IV infusion during administration. A dose given more than 3 days after the scheduled dosing date was considered a dose delay, and the maximum dose delay window allowed was 42 days between doses. Dose delay criteria included any grade 2 or greater non-skin, drug-related AE (excluding fatigue or laboratory abnormalities), any grade 2 skin, drug-related AE, and any grade 3 drug-related laboratory abnormality (with some exceptions such as lymphopenia).
Concomitant Medications
Treatments that were not permitted within 28 days of first dose included chemotherapy, radiation therapy, biologics for cancer, intravesical therapy (with the exception of a single dose of intravesical chemotherapy immediately after resection of low-risk NMIBC), or investigational therapy. Systemic treatment with corticosteroids or other immunosuppressive medications were also not permitted, except for inhaled or topical steroids, and adrenal replacement steroids in the absence of active autoimmune disease.
Outcomes
A list of efficacy end points identified in the CADTH review protocol that were assessed in the clinical trial included in this review is provided in Table 9. These end points are further summarized in the following. A detailed discussion on the validity of the outcome measures is provided in Appendix 4. A review of studies assessing the appropriateness of DFS as a surrogate outcome is also presented in Appendix 5.
Table 9: Summary of Outcomes of Interest Identified in the CADTH Review Protocol
Outcome measure | CheckMate 274 trial |
|---|---|
DFSa in patients with ≥ 1% PD-L1 and in all randomized patients | Primary |
OSb in patients with ≥ 1% PD-L1 and in all randomized patients | Key secondary |
NUTRFSc in patients with ≥ 1% PD-L1 and in all randomized patients | Secondary |
DSSd in patients with ≥ 1% PD-L1 and in all randomized patients | Secondary |
DMFS in patients with ≥ 1% PD-L1 and in all randomized patients | Exploratory |
TTR in patients with ≥ 1% PD-L1 and in all randomized patients | Exploratory |
LRDFS in patients with ≥ 1% PD-L1 and in all randomized patients | Exploratory |
LRC in patients with ≥ 1% PD-L1 and in all randomized patients | Exploratory |
HRQoL using EORTC QLQ-C30 and EQ-5D-3L | Exploratory |
Symptom severity | Not reported |
PFS2 in patients with ≥ 1% PD-L1 and in all randomized patients | Exploratory |
Overall safety/tolerability (incidence of AEs, SAEs, select AEs, IMAEs in patients with ≥ 1% PD-L1 and in all randomized patients) | Exploratory |
AE = adverse event; CPS = combined positive score; DFS = disease-free survival; DMFS = distant metastasis-free survival; DSS = disease-specific survival; EORTC QLQ-C30 = European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30; EQ-5D-3L = EuroQol 5-Dimensions 3-Levels questionnaire; HRQoL = health-related quality of life; IMAE = immune-mediated adverse event; LRC = locoregional control; LRDFS = locoregional disease-free survival; NUTRFS = non-urothelial tract recurrence-free survival; OS = overall survival; PD-L1 = programmed death-ligand 1; PFS2 = progression-free survival after the next line of subsequent systemic anticancer therapy; SAE = serious adverse event; TTR = time to recurrence.
aDefined as the time between the date of randomization and the date of first documented recurrence (local urothelial tract, local non-urothelial tract, or distant), or death due to any cause, whichever occurs first.
bReported in a separate interim analysis report with a data cut-off date of February 1, 2021. Results were not available from the sponsor at the time of this CADTH review. Defined as the time from randomization to the date of death from any cause. For patients that are alive, their survival time will be censored at the date of last contact date (or “last known alive date”). OS will be censored at the date of randomization for patients who were randomized but had no follow-up.
cProgrammatically determined based on the disease recurrence date provided by the investigator.
dResults were not available from the sponsor at the time of this CADTH review. Defined as the time from randomization to the date of death due to disease (urothelial carcinoma). For patients who are alive, their survival time will be censored at the date of last contact date (or “last known alive date”). DSS will be censored at the date of randomization for patients who were randomized but had no follow-up.
Source: CheckMate 274 Clinical Study Report.9
The primary outcome of the CheckMate 274 study was DFS. DFS was defined as the time between the date of randomization and the date of first documented recurrence (local urothelial tract, local non-urothelial tract, or distant), or death due to any cause, whichever occurred first. Disease assessment imaging included chest CT, and CT or MRI of the abdomen, pelvis, upper urinary tract, as well as cystoscopy for patients with UTUC and intact bladders.
DFS was assessed by the investigator and derived by a statistical program (SAS) using an algorithm based on the disease recurrence date provided by the investigator. Disease recurrence of the local urothelial tract was defined as any high- and intermediate-risk NMIBC and any new invasive UC in the lower or upper urothelial tract (defined as T2 or greater), including lesions thought to be a second primary UC. As the primary definition, patients who started subsequent therapy (radiotherapy, surgery, or systemic therapy) or developed a new primary cancer without recurrence, were censored on the last evaluable disease assessment date before the start of subsequent therapy or development of new non-urothelial primary cancer. The secondary definition of DFS ignored subsequent anticancer therapy in the sensitivity analysis and only censored at the last evaluable disease assessment on or before the date of new non-UC primary cancer.
A key secondary outcome was OS, which was reported in a separate interim analysis report with a data cut-off date of February 1, 2021, as the number of deaths needed to trigger the first OS interim analysis had not been reached at the August 27, 2020, data cut-off date. Results for OS were not available from the sponsor at the time of this CADTH review. OS was defined as the time from randomization to the date of death from any cause. For patients that are alive, their survival time was censored at the date of last contact date (or “last known alive date”). OS was censored at the date of randomization for patients who were randomized but had no follow-up.
DSS was assessed as secondary end point, defined as the time from randomization to the date of death due to UC, which was followed continuously as part of OS follow-up and every 3 months after the study drug was discontinued. DSS was not reported in the sponsor-submitted materials.
NUTRFS is a secondary outcome and was defined as the time between the date of randomization and the date of first local non-urothelial tract or distant recurrence or death of any cause, whichever occurred first. NUTRFS accounted for subsequent anticancer therapy by censoring the date of last evaluable disease assessment for patients who remained alive and recurrence-free.
Exploratory outcomes included DMFS (time between the date of randomization and the date of first distant recurrence or date of death, whichever occurred first) and TTR (time between the date of randomization and the date of first recurrence [local urothelial tract, local non-urothelial tract, or distant] or death due to disease, whichever occurred first), locoregional DFS (LRDFS), and locoregional control (LRC). PFS2 (progression-free survival after the next line of subsequent therapy) was assessed as exploratory outcome as well.
HRQoL was assessed as an exploratory outcome using the EORTC QLQ-C30 and the EQ-5D-3L. Baseline assessments were completed before the first dose, then assessments were completed every 4 weeks during the first 6 months of treatment, every 6 weeks thereafter until treatment completion or discontinuation, and at the 2 follow-up visits 35 days and 115 days after the last dose.
The EORTC QLQ-C30 consists of 30 questions that are scored to create 5 multi-item functional scales (physical, role, cognitive, emotional, and social), 3 multi-item symptom scales (fatigue, pain, and nausea and vomiting), 6 single-item symptom scales (dyspnea, loss of appetite, insomnia, constipation, and diarrhea), a 2-item quality of life scale, and perceived financial impact of the disease.34 All the scales and single-item measures range in score from 0 to 100. Most questions have 4 response options (“not at all,” “a little,” “quite a bit,” and “very much”), with scores on these items ranging from 1 to 4. For the 2 items that form the global quality of life scale, the response format is a 7-point Likert-type scale with anchors at 1 (“very poor”) and 7 (“excellent”). Raw scores for each scale are computed as the average of the items that contribute to a particular scale. Scale sum scores are transformed so that a high score on the functional scales represents a high or healthy level of functioning, a high score on the symptom scales represents a high level of symptomatology, and a high score on the global health status or quality of life represents a high quality of life.35 The sponsor defined clinically meaningful change as a change of 10 points for the EORTC QLQ-C30 domains at 2 or more consecutive visits. A detailed discussion and critical appraisal of the HRQoL measure is provided in Appendix 4.
The EQ-5D-3L produces 3 types of data for each respondent: a profile indicating the extent of problems on each of 5 dimensions (mobility, self-care, usual activities, pain or discomfort, and anxiety or depression) represented by a 5-digit descriptor; a population preference-weighted health index score based on the descriptive system; and a self-reported current health status based on the EuroQol Visual Analogue Scale (VAS) that is used to assess the overall health of the respondent rather than selected dimensions of individuals’ health.36,37 The sponsor defined clinically meaningful change as a change of 7 points for the EuroQol VAS at 2 or more consecutive visits. A detailed discussion and critical appraisal of the HRQoL measure is provided in Appendix 4.
Symptom severity was not assessed in the CheckMate 274 trial.
Safety and tolerability were assessed using the incidence AEs, SAEs, select AEs, and IMAEs in patients, and deaths and laboratory abnormalities, and using the National Cancer Institute CTCAE version 4.0 guidelines. AEs, SAEs, and select AEs were assessed using the 30-day safety window. IMAEs are specific events occurring within 100 days of the last dose that included diarrhea or colitis, hepatitis, pneumonitis, nephritis and renal dysfunction, rash, and endocrine dysfunction (adrenal insufficiency, hypophysitis, hypothyroidism or thyroiditis, hyperthyroidism, and diabetes mellitus).
Statistical Analysis
Statistical analysis of all efficacy outcomes (specified in the protocol for the systematic literature review) in the CheckMate 274 study is summarized in Table 10.
Table 10: Statistical Analysis of End Points in the CheckMate 274 Trial
End point | Statistical model | Adjustment factors | Sensitivity analyses |
|---|---|---|---|
Primary outcome | |||
DFS in 2 co-primary populations: all randomized patients and patients with PD-L1 ≥ 1% |
| Stratification factors:
|
|
Secondary outcomes | |||
OS in 2 co-primary populations: all randomized patients and patients with PD-L1 ≥ 1% | As per DFS analysis | As per DFS analysis | NA |
NUTRFS | As per DFS analysis (without log-rank test) | As per DFS analysis | NA |
Exploratory outcomes | |||
DMFS |
| NA | NA |
PFS2 | As per DMFS analysis | NA | NA |
TTR |
| As per DFS analysis | NA |
LRDFS | As per TTR analysis | As per DFS analysis | NA |
LRC | As per TTR analysis | As per DFS analysis | NA |
EORTC QLQ-C30 | Descriptive statistics | NA | NA |
EuroQol EQ-5D-3L | Descriptive statistics | NA | NA |
CI = confidence interval; CRF = case report form; DFS = disease-free survival; DMFS = distant metastasis-free survival; EORTC QLQ-C30 = European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire Core 30; EQ-5D-3L = EuroQol 5-Dimensions 3-Levels questionnaire; HR = hazard ratio; IRT = interactive response technology; LRC = locoregional control; LRDFS = locoregional disease-free survival; NA = not applicable; NUTRFS = non-urothelial tract recurrence-free survival; OS = overall survival; PD-L1 = programmed death-ligand 1; PFS2 = progression-free survival after the next line of subsequent systemic anticancer therapy; TTR = time to recurrence.
Source: CheckMate 274 Clinical Study Report Protocol.9
Power Calculation
The sample size determination considered the comparison of the primary outcome of DFS and the secondary outcome of OS.
For DFS, curves may not follow an exponential decay and a flattening of the curves may be observed toward the end of the tail because a meaningful number of long-term disease-free survivors may be observed. Therefore, an exponential cure rate distribution was used to calculate the sample size. Approximately 410 DFS events in all randomized patients were to provide approximately 87% power to detect an average HR of 0.72, and approximately 162 DFS events in patients with a tumour cell PD-L1 expression level of 1% or greater were to provide approximately 80% power to detect an average HR of 0.61 with a 2-sided alpha of 0.05. It was expected that the required number of events in each population was to be reached when all patients had at least a minimum of 17 months of follow-up. An interim DFS analysis was to be performed when 85% of DFS events in each population were observed (i.e., 348 and 137 DFS events in all randomized and in patients with a tumour cell PD-L1 expression level ≥ 1%, respectively). At the time of the August 27, 2020, data cut-off, the actual number of DFS events was 374 among all randomized patients (91.2% of total DFS events) and 136 among patients with tumour cell PD-L1 expression level of 1% or greater (84.0% of the total DFS events). An interim analysis for DFS was performed and the boundary for statistical significance for DFS (observed P = 0.0008 and = 0.0005, respectively) was crossed (adjusted alpha = 0.01784 and 0.01282, respectively). The testing procedures for all randomized patients and all randomized patients with a PD-L1 expression level of 1% or greater are presented in Figure 3 and Figure 4 respectively. In a response from the sponsor dated April 20, 2022, the sponsors clarified that since the boundary for statistical significance for DFS was crossed on the interim analysis on August 27, 2020, the interim analysis became the final analysis for DFS. In the first interim analysis for OS with a data cut-off on February 1, 2021, the analysis did not cross the pre-specified boundary for declaring statistical significance and results were not provided to the CADTH review team and the study continues to remain blinded.
Statistical Test or Model
DFS distributions were compared between treatment groups using a 2-sided stratified log-rank test at the overall significance level of 2.5% (2-sided) each in all randomized patients and in patients with a PD-L1 expression level of 1% or greater.
For DFS, HR and corresponding 2-sided 100 × (1-adjusted alpha) % CIs were estimated in a Cox proportional hazards model using treatment as a single covariate, stratified by PD-L1 status (only in the randomized population comparison), prior neoadjuvant cisplatin chemotherapy, and positive lymph node status. NUTRFS HR and corresponding 2-sided 95% CIs were estimated using the same method.
Figure 3: Hierarchical Procedure With Group Sequential Testing in the Randomized Patients
DFS = disease-free survival; OS = overall survival.
Figure 4: Hierarchical Procedure With Group Sequential Testing in the Patients With PD-L1 Expression Level ≥ 1%
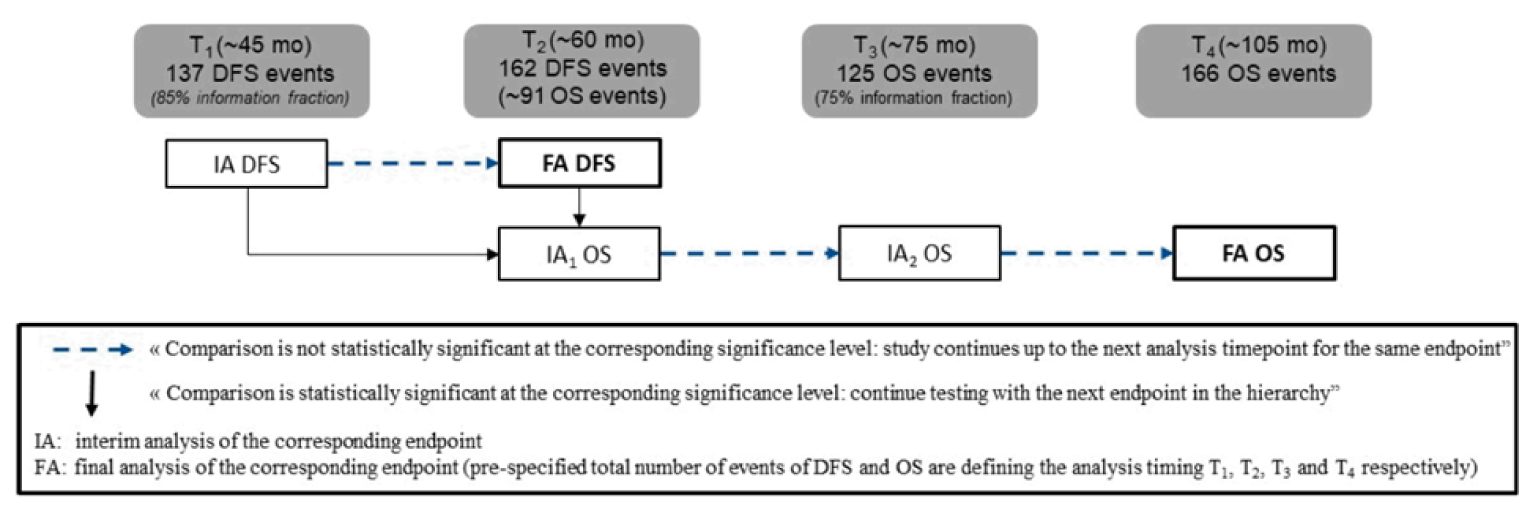
DFS = disease-free survival; OS = overall survival; PD-L1 = programmed death-ligand 1.
In addition, DFS curves were estimated using the Kaplan-Meier product-limit method. Median DFS and the corresponding 2-sided 95% CIs using the log-log transformation were computed, as well as DFS rates at 6 months, 1 year, and 2 years with the corresponding 2-sided 95% CIs using the log-log transformation. NUTRFS and OS distribution curves were estimated using Kaplan-Meier methodology by treatment arm. Median values and the corresponding 2-sided 95% CIs using the log-log transformation were also computed with rates at 6 months, 1 year, and 2 years with the corresponding 2-sided 95% CIs using the log-log transformation.
Exploratory outcomes DMFS, PFS2, TTR, LRDFS, and LRC were analyzed descriptively in all randomized patients and in patients with a PD-L1 expression level of 1% or greater.
Patient-reported and HRQoL outcomes using the EQ-5D-3L and EORTC QLQ-C30, were assessed as the exploratory outcomes. The mean scores and mean changes from baseline for these exploratory outcomes were summarized by treatment group at each assessment time using descriptive statistics. No statistical analysis was performed to test between-group differences.
Subgroup Analyses
Median DFS based on Kaplan-Meier product-limit method along with 2-sided 95% CIs produced the following subgroups: PD-L1 status, use of prior neoadjuvant cisplatin therapy, prior cisplatin status, use of any prior neoadjuvant systemic therapy, pathological lymph node status, pathological status, age, region, gender, race, baseline ECOG status, smoking status, baseline hemoglobin, baseline creatinine clearance, time from surgery for invasive urothelial carcinoma to randomization, initial tumour origin, and minor histological variants.
The following subgroups, planned a priori in the statistical analyses plan, aligned with the subgroups pre-specified in the protocol for this CADTH review: tumour PD-L1 expression of 1% or greater, primary location of tumour, use of neoadjuvant treatment, and nodal status. Only those subgroups identified in the CADTH review protocol are reported in the efficacy section.
Sensitivity Analyses
Sensitivity analyses for DFS was conducted using an unstratified log-rank test, using a multivariate Cox regression model to estimate the treatment effect after adjustment for potential prognostic factors, with patients with no relevant protocol deviation, while accounting for missing disease assessments before DFS event, using a stratified time-dependent Cox model with effects for treatment and period-by-treatment interaction, and by adding a time-dependent variable defined by treatment by time interaction.
Protocol Changes
The major changes to the study protocol are summarized as follows.
In Revised Protocol 2, the period of time between radical resection and randomization was extended from 90 days to 120 days.
In Revised Protocol 3, a more precise description of low- and high-risk NMIBC was added, patients with carcinoma in situ at urethral or ureteral surgical margins were excluded from the study, PD-L1 expression of 1% or greater was adjusted from 50% to 46% in accordance with recently published data, and a 20% cap on the number of patients with pelvis and ureter cancer randomized to the study was added.
In Revised Protocol 4, sample size was increased from 640 to 700, the cap on PD-L1 expression of 1% or greater was removed, and PFS2 was added as an exploratory end point.
In Revised Protocol 5, a formal interim OS analysis at the time of final DFS analysis for both study populations (PD-L1 ≥ 1% and all randomized patients) was added.
Analysis Populations
The efficacy population in the CheckMate 274 trial was all randomized patients and all randomized patients with tumour PD-L1 expression of 1% or greater, and this population was used for analysis of all efficacy outcomes. The population used for safety outcome analyses was all treated patients and all treated patients with tumour PD-L1 expression of 1% or greater. The HRQoL-evaluable population was all randomized patients who had an assessment at screening or baseline and at least 1 follow-up assessment.
Results
There were 3 DBL dates provided in the sponsor’s submission:
August 27, 2020 DBL: the final DFS DBL date with the analysis of all end points except for OS and DSS since the number of deaths to trigger the first OS interim analysis had not been reached per the statistical design
April 13, 2021 DBL (data cut-off August 27, 2020): an error in analysis of DFS and NUTRFS results was discovered, a new restricted DBL with the same data cut-off date (August 27, 2020) was performed, and an erratum was provided11
May 19, 2021 DBL (data cut-off February 1, 2021): first interim OS analysis (OS data were not provided to CADTH by the sponsor at the time of this review); efficacy end points including DFS, NUTRFS, DMFS, and TTR were included in a poster presentation at the Society of Urologic Oncology Congress in December 2021.13
Patient Disposition
Patient disposition for the CheckMate 274 study is presented in Table 11. Of the 1,075 patients screened, a total of 709 (66%) patients were randomized to receive nivolumab (n = 353) or placebo (n = 356). The most common causes for screening failure were that patients no longer met study criteria (273 patients [25.4%]) and patients withdrew consent (50 patients [4.7%]). Of the 709 patients randomized, 2 patients in the nivolumab group and 8 patients in the placebo group did not receive their assigned treatment.
At the time of the data cut-off date of August 27, 2020, a total of 383 (54%%) patients discontinued treatment (n = 187 [53.3%] in the nivolumab group and n = 196 [56.3%] in the placebo arm). The main reason for discontinuation of treatment was disease recurrence (25.6% and 42.2%, respectively) followed by study drug toxicity (14% versus 2.3%, respectively). A total of 93.2% patients in both arms were still continuing in the study at the time of data cut-off date for documentation of progression and death. The efficacy population (i.e., intention-to-treat population) included 709 patients, while the safety population included 699 patients.
Protocol Violations
At the time of the data cut-off date of August 27, 2020, a total of 12 (3.4%) patients had protocol deviations in the nivolumab group, and 18 (5.1%) patients in the placebo group. The most commonly reported type of violation was randomization more than 120 days after surgery (4 [1.1%] patients in the nivolumab group and 11 [3.1%] patients in the placebo group).
Exposure to Study Treatments
For all treated patients, the median duration on therapy was similar between the nivolumab and placebo arms. As of the August 27, 2020, data cut-off date, the median treatment durations were 8.77 months (range: 0 to 12.5) in the nivolumab group and 8.21 months (range: 0 to 12.6) in the placebo arm. A total of 7 (2%) patients in the nivolumab group and 13 (3.7%) patients in the placebo group received treatment for a duration greater than 12 months. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. Details on dose intensity are presented in Table 12.
Table 11: Patient Disposition on August 27, 2020 (Data Cut-Off Date)
Component | All randomized patients | Patients with tumour PD-L1 ≥ 1% | ||
|---|---|---|---|---|
Nivolumab n = 353 | Placebo n = 356 | Nivolumab n = 140 | Placebo n = 142 | |
Screened, N | 1,075 | NA | ||
Randomized, N (%) | 353 | 356 | 140 | 142 |
Treated, n (%) | 351 (99.4) | 348 (97.8) | 139 (99.3) | 139 (97.9) |
|||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| |
Discontinued study treatment, n (%)a | 187 (53.3) | 196 (56.3) | 71 (51.1) | 87 (62.6) |
Reason for treatment discontinuation, n (%)a | ||||
Disease recurrence | 90 (25.6) | 147 (42.2) | 33 (23.7) | 59 (42.4) |
Study drug toxicity | 49 (14.0) | 8 (2.3) | 24 (17.3) | 7 (5.0) |
Death | 0.0 | 1 (0.3) | 0.0 | 1 (0.7) |
AE unrelated to study drug | 16 (4.6) | 15 (4.3) | 4 (2.9) | 8 (5.8) |
Patient request to discontinue study treatment | 19 (5.4) | 4 (1.1) | 6 (4.3) | 2 (1.4) |
Patient withdrew consent | 5 (1.4) | 7 (2.0) | 3 (2.2) | 3 (2.2) |
Lost to follow-up | 1 (0.3) | 0.0 | 0.0 | 0.0 |
|||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| |
Follow-up length, months | ||||
Mean (SD) | 22.2 (12.2) | 20.7 (13.2) | 22.4 (12.8) | 20.3 (13.5) |
Median (range) | 20.9 (0.1 to 48.3) | 19.5 (0 to 50) | 22.1 (0.1 to 47.5) | 18.7 (0 to 49.1) |
AE = adverse event; NA = not applicable; PD-L1 = programmed death-ligand 1; SD = standard deviation.
Note: Redacted rows have been deleted.
aPercentages based on patients entering treatment period.
Source: CheckMate 274 Clinical Study Report.9
Table 12: Dose Intensity on August 27, 2020 (Data Cut-off Date)
Item | All treated patients | Treated patients with tumour PD-L1 ≥ 1% | ||
|---|---|---|---|---|
Nivolumab n = 351 | Placebo n = 348 | |||||||||||||||||||| | |||||||||||||||||||| | |
Doses received, mean (SD) | 16.7 (9.0) | 16.5 (8.9) | |||||||||||||||||||| | |||||||||||||||||||| |
Cumulative dose (mg), mean (SD) | 3,997.7 (2,156) | NA | |||||||||||||||||||| | |||||||||||||||||||| |
Relative dose intensity, n (%) | ||||
90% to < 110% | 297 (84.6) | NA | |||||||||||||||||||| | |||||||||||||||||||| |
70% to < 90% | 49 (14.0) | NA | |||||||||||||||||||| | |||||||||||||||||||| |
50% to < 70% | 3 (0.9) | NA | |||||||||||||||||||| | |||||||||||||||||||| |
< 50% | 2 (0.6) | NA | |||||||||||||||||||| | |||||||||||||||||||| |
NA = not applicable; PD-L1 = programmed death-ligand 1; SD = standard deviation.
Source: CheckMate 274 Clinical Study Report.9
Dose Modification
Among all treated patients in the study, 162 (46.2%) patients in the nivolumab group and 146 (42.0%) patients in the placebo group had at least 1 dose delay, with the majority requiring only 1 dose delay (25.9% in the nivolumab group and 24.1% in the placebo arm). Of all dose delay incidents, the most common reason was an adverse effect (47.4% in the nivolumab group and 41.0% in the placebo arm). Among all treated patients, 12 (3.4%) patients in the nivolumab group and 6 (1.7%) patients in the placebo group had at least 1 infusion interruption, with the majority requiring only 1 infusion interruption (2.3% in the nivolumab group and 1.4% in the placebo arm). Of all interrupted infusions, the most common reason was a hypersensitivity reaction (59.3% in the nivolumab group and 42.9% in the placebo arm). Dose reductions were reported in 6 (1.7%) patients in the nivolumab group and 2 (0.6%) patients in the placebo arm, and the most commonly reported reason was “other,” followed by hypersensitivity reaction and infusion administration issues.
Concomitant Medications
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Subsequent Anticancer Therapy
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 13: Subsequent Anticancer Therapy
|||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| | ||
|---|---|---|---|---|
|||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| | |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | ||||
|||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| |
|||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| |
|||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| |
|||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| | |||||||||||||||||||| |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||9
Note: Redacted rows have been deleted.
Efficacy
Only those efficacy outcomes and analyses of subgroups identified in the review protocol are reported in the following.
Overall Survival
OS was a key secondary end point in the CheckMate 274 trial and was not reported in the primary CSR (at the time of the data cut-off date of August 27, 2020, the number of deaths to trigger the first OS interim analysis had not been reached per the statistical design). Among all treated patients, there were 95 (27.1%) deaths reported in the nivolumab group and 107 (30.7%) deaths reported in the placebo group. The primary cause of death was disease recurrence (73 [20.8%] in the nivolumab group and 90 [25.9%] in the placebo group).
In the absence of OS data, it has been suggested by EMA that PFS2 may be 1 of the relevant outcomes in the adjuvant setting. As of the August 27, 2020, data cut-off, median PFS2 was 44.6 months in patients in the nivolumab group and 40.7 months in the placebo group (HR = 0.79; 95% CI, 0.61 to 1.02). More details about the exploratory analysis of PFS2 are presented in Appendix 3.
Disease-Free Survival
As of the August 27, 2020, data cut-off date, minimum follow-up time was 5.9 months, and median follow-up time among all randomized patients was 20.9 months and 19.5 months in the nivolumab and placebo groups, respectively. In all randomized patients with a tumour cell PD-L1 expression of 1% or greater, the minimum follow-up time was 6.3 months, and the median follow-up was 22.1 months and 18.7 months in the nivolumab and placebo groups, respectively.
In the DFS analysis, 183 (51.8%) patients and 152 (42.7%) patients were censored in the nivolumab group and in the placebo group, respectively. Among all randomized patients, median DFS was 20.8 months (95% CI, 16.5 to 27.6) in the nivolumab group compared to 10.8 months (95% CI, 8.3 to 13.9) in the placebo group, with an HR of 0.70 (98.22% CI, 0.55 to 0.90; P = 0.0008) in favour of the nivolumab group. DFS rate at 6 months was 74.9 (95% CI, 69.9 to 79.2) in the nivolumab group and 60.3 (95% CI, 54.9 to 65.3) in the placebo group. As shown in Figure 5, Kaplan-Meier curves for DFS separated after 3 months, favouring nivolumab. DFS results are presented in Table 14. Among all randomized patients with a tumour cell PD-L1 expression of 1% or greater, median DFS was not reached in the nivolumab group compared to 10.8 months (95% CI, 5.7 to 21.2) in the placebo group, with an HR of 0.55 (98.72% CI, 0.35 to 0.85; P = 0.0005). DFS rate at 6 months was 74.5 (95% CI, 66.2 to 81.1) in the nivolumab group and 55.7 (95% CI, 46.8 to 63.6) in the placebo group. As shown in Figure 6, Kaplan-Meier curves for DFS separated after 3 months, favouring nivolumab.
When DFS was analyzed using the secondary DFS definition, which accounted for disease assessments occurring on or after initiation of subsequent anticancer therapy, the results were consistent with the analysis for the primary DFS definition with an HR of 0.70 (98.22% CI, 0.55 to 0.90; P = 0.0006) in favour of nivolumab.
At the updated DFS analysis at the February 1, 2021, data cut-off date, results were consistent with results from the final primary analysis. The minimum follow-up time was 11 months, and median follow-up time among all randomized patients was 24.4 months and 22.5 months in the nivolumab and placebo groups, respectively. In all randomized patients with a tumour cell PD-L1 expression of 1% or greater, the minimum follow-up time was 11.4 months, and the median follow-up was 25.5 months and 22.4 months in the nivolumab and placebo groups, respectively. Among all treated patients, median DFS was 22.0 months in the nivolumab group and 10.9 months in the placebo group with an HR of 0.70 (95% CI, 0.57 to 0.85). Among all treated patients with a tumour cell PD-L1 expression of 1% or greater, median DFS was not reached in the nivolumab group and was 8.4 months in the placebo group with an HR of 0.53 (95% CI, 0.38 to 0.75).13
DFS Subgroup Analysis
The DFS results by subgroups of interest, as specified a priori in the protocol for this CADTH review, are summarized in Table 15. The treatment effect of DFS was consistent with the primary analysis across patient subgroups, except for the subgroups of upper urothelial tract primary tumour location categories; however, sample size and number of events were relatively lower in these patient categories. The HRs were 0.56 (95% CI, 0.40 to 0.80) and 0.82 (95% CI, 0.63 to 1.06) in patients with 1% or greater and less than 1% tumour PD-L1 expression, respectively; 0.52 (95% CI, 0.38 to 0.71) and 0.92 (95% CI, 0.69 to 1.21) in patients with and without history of neoadjuvant cisplatin therapy respectively; and 0.64 (95% CI, 0.48 to 0.85), 0.85 (95% CI, 0.57 to 1.28), and 0.67 (95% CI, 0.41 to 1.10) in patients with N+, N0 or NX with less than 10 nodes removed, and N0 with 10 nodes removed, respectively. HR was greater than 1 in upper urothelial tract primary tumour location categories, favouring placebo over nivolumab. The wide CIs in subgroups reflected uncertainty in the effect estimates.
Table 14: DFS Among All Randomized Patients and Patients With Tumour Cell PD-L1 Expression ≥ 1% in the CheckMate 274 Trial on August 27, 2020 (Data Cut-off)
Item | All randomized patients | Patients with tumour PD-L1 ≥ 1% | ||
|---|---|---|---|---|
Nivolumab n = 353 | Placebo n = 356 | Nivolumab n = 140 | Placebo n = 142 | |
DFS primary definition | ||||
Events, n (%) | 170 (48.2) | 204 (57.3) | 55 (39.3) | 81 (57.0) |
Median DFS, months (95% CI)a | 20.8 (16.5 to 27.6) | 10.8 (8.3 to 13.9) | Not reached (21.19 to NA) | 8.41 (5.59 to 21.19) |
HR (% CI)b | 0.70 (98.22% CI, 0.55 to 0.90) | 0.55 (98.72% CI, 0.35 to 0.85) | ||
Stratified log-rank P valuec | 0.0008d | 0.0005e | ||
Rate at 6 months, % (95% CI)a | 74.9 (69.9 to 79.2) | 60.3 (54.9 to 65.3) | 74.5 (66.2 to 81.1) | 55.7 (46.8 to 63.6) |
CI = confidence interval; DFS = disease-free survival; HR = hazard ratio; NA = not applicable; PD-L1 = programmed death-ligand 1.
Note: The primary definition of DFS accounted for subsequent anticancer therapy and new non-urothelial carcinoma primary cancer.
aBased on Kaplan-Meier estimates.
bStratified Cox proportional hazard model. HR is nivolumab over placebo.
cTwo-sided P values from stratified regular log-rank test.
dLog-rank test stratified by prior neoadjuvant cisplatin, pathological nodal status, as entered in the interactive response technology.
eLog-rank test stratified by prior neoadjuvant cisplatin, pathological nodal status, PD-L1 status (≥ 1% vs. < 1% or indeterminate) as entered in the interactive response technology.
Source: CheckMate 274 Clinical Study Report.9
Table 15: Disease-Free Survival Subgroups Analysis on August 27, 2020 (Data Cut-Off Date)
Subgroups | All randomized patients | ||
|---|---|---|---|
Nivolumab (n = 353) | Placebo (n = 356) | HR (95% CI)a | |
Tumour PD-L1 expression, n of N | |||
≥ 1% | 55 of 139 | 79 of 141 | 0.56 (0.40 to 0.80) |
< 1% | 114 of 210 | 120 of 209 | 0.82 (0.63 to 1.06) |
Primary location of tumoura | |||
Urinary bladder | 129 of 279 | 166 of 281 | 0.62 (0.49 to 0.78) |
Renal pelvis | 24 of 44 | 25 of 52 | 1.23 (0.67 to 2.23) |
Ureter | 17 of 30 | 13 of 23 | 1.56 (0.70 to 3.48) |
Use of neoadjuvant cisplatin treatment | |||
Yes | 70 of 153 | 10 of 155 | 0.52 (0.38 to 0.71) |
No | 100 of 200 | 104 of 201 | 0.92 (0.69 to 1.21) |
Nodal status | |||
N+ | 95 of 167 | 116 of 168 | 0.64 (0.48 to 0.85) |
N0 or NX with < 10 nodes removed | 46 of 94 | 50 of 99 | 0.85 (0.57 to 1.28) |
N0 with 10 nodes removed | 29 of 91 | 37 of 88 | 0.67 (0.41 to 1.10) |
CI = confidence interval; HR = hazard ratio; PD-L1 = programmed death-ligand 1.
aStratified Cox proportional hazard model. HR is not computed for subset (except age, region, and sex) category with less than 10 patients per treatment arm. PD-L1 status is based on clinical database.
Figure 5: Kaplan-Meier Plot of Disease-Free Survival (Primary Definition) — All Randomized Patients on the August 27, 2020, Data Cut-Off Date
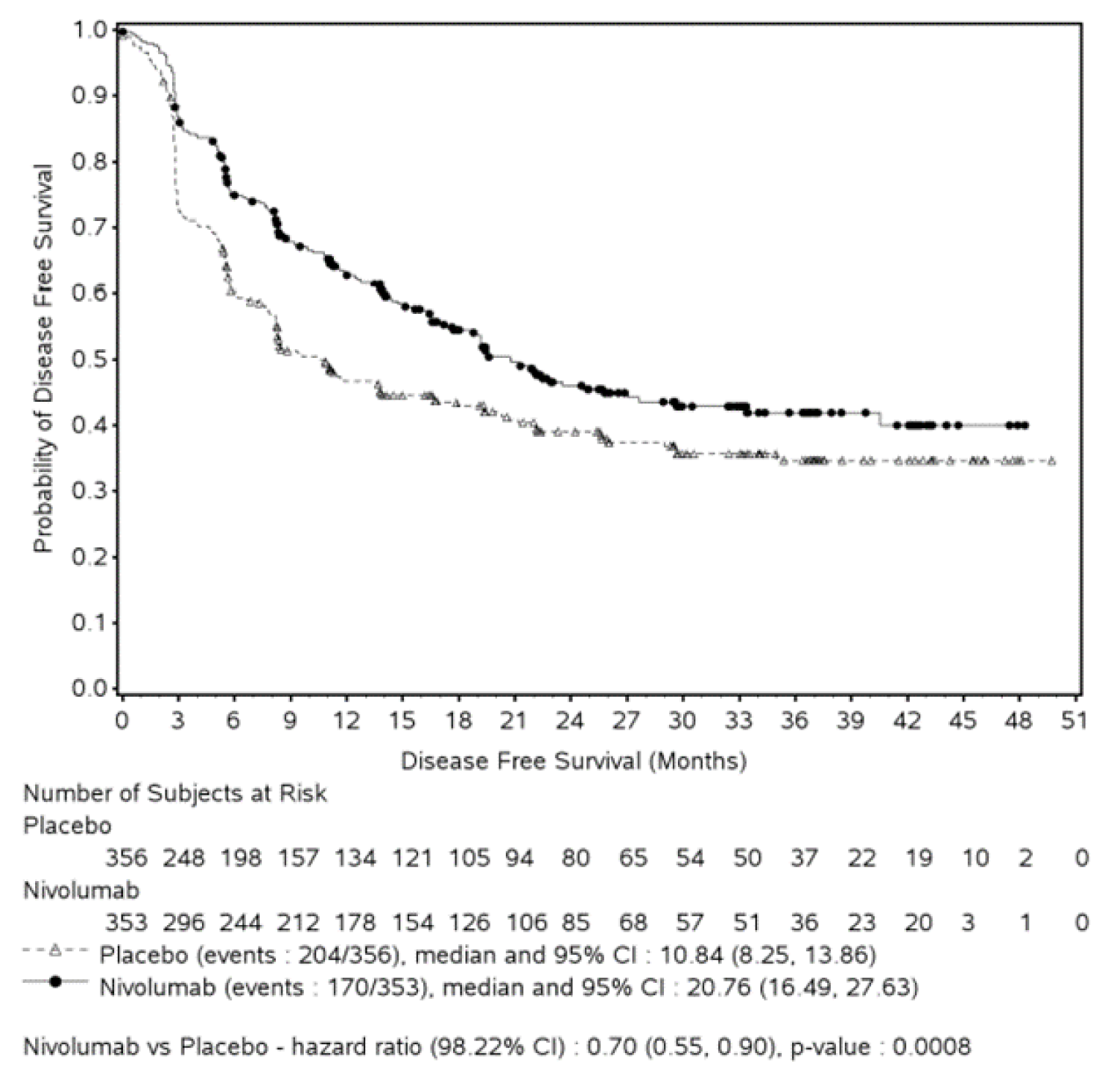
CI = confidence interval.
Note: Statistical model for hazard ratio and P value-stratified Cox proportional hazard and stratified log-rank test. Symbols represent censored observations.
Source: Figure 4.7-1 Primary Clinical Study Report erratum.11
Figure 6: Kaplan-Meier Plot of Disease-Free Survival (Primary Definition) — All Randomized Patients With Tumour PD-L1 Expression ≥ 1% on the August 27, 2020 (Data Cut-Off Date)
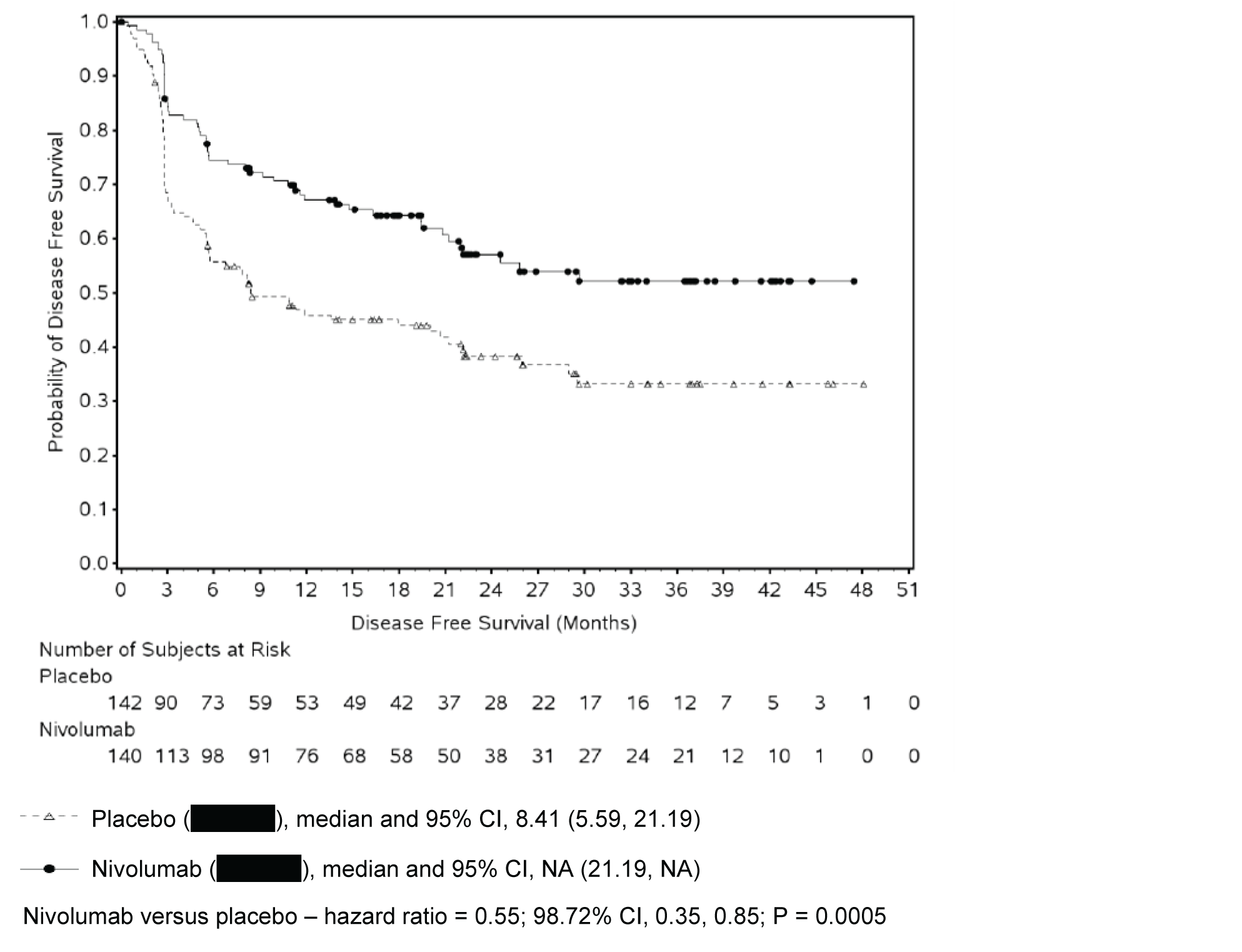
CI = confidence interval, NA = not applicable; PD-L1 = programmed death-ligand 1.
Note: Statistical model for hazard ratio and P value: stratified Cox proportional hazard and stratified log-rank test. Symbols represent censored observations.
Source: Figure 4.13-1, Primary Clinical Study Report erratum.11
Non-Urothelial Tract Recurrence-Free Survival
As of the August 27, 2020, data cut-off date, among all randomized patients in the NUTRFS analysis, 191 (54.1%) patients and 166 (46.6%) patients were censored in the nivolumab group and in the placebo group, respectively. Most patients were still in follow-up (134 [38.0%] in the nivolumab group and 113 [31.7%] in the placebo group). Among all randomized patients, median NUTRFS was 22.9 months (95% CI, 19.2 to 33.4) in the nivolumab group compared to 13.7 months (95% CI, 8.4 to 20.3) in the placebo group, with an HR of 0.72 (95% CI, 0.59 to 0.89). Among all randomized patients with tumour cell PD-L1 expression of 1% or greater, median NUTRFS was not reached in the nivolumab group and was 10.8 months (95% CI, 5.7 to 22.1) in the placebo group, with an HR of 0.55 (95% CI, 0.39 to 0.79). NUTRFS results are presented in Table 16 and Kaplan-Meier curves for NUTRFS are presented in Figure 7.
At the updated analysis with data cut-off date of February 1, 2021, median NUTRFS among all treated patients was 26 months (95% CI, 19.5 to 41.1) in the nivolumab group and compared to 13.7 months (95% CI, 8.4 to 20.0) in the placebo group, with an HR of 0.71 (95% CI, 0.58 to 0.88) favouring nivolumab.13
HRQoL
EORTC QLQ-C30
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
At baseline, mean EORTC QLQ-C30 summary scores in all domains and for all randomized patients were comparable between treatment arms.31 In all randomized patients at the August 27, 2020, data cut-off date, quality of life remained stable in both treatment arms, with no mean change scores from baseline reaching the minimal important difference (MID) for the patient (i.e., mean change ≥ 10 points)38 at any time point for either treatment group as shown in Figure 8. Results for patients with PD-L1 expression of 1% or greater were consistent with the overall trial population.
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. In a related publication,31 the authors suggested that impact on HRQoL does not differ substantially between drug toxicity and disease recurrence, an assumption that was deemed as reasonable by 1 of the clinical experts consulted by CADTH.
This outcome was not assessed at the updated analysis with a data cut-off date February 1, 2021.
Table 16: NUTRFS Among All Randomized Patients and Patients With Tumour Cell PD-L1 Expression ≥ 1% in the CheckMate 274 Trial on August 27, 2020 (Data Cut-Off Date)
Item | All randomized patients | Patients with tumour PD-L1 ≥ 1% | ||
|---|---|---|---|---|
Nivolumab n = 353 | Placebo n = 356 | Nivolumab n = 140 | Placebo n = 142 | |
Events, n (%) | 162 (45.9) | 190 (53.4) | 54 (38.6) | 78 (54.9) |
Median NUTRFS, months, (95% CI)a | 22.9 (19.2 to 33.4) | 13.7 (8.4 to 20.3) | Not reached. (24.57 to not reached) | 10.84 (5.65 to 22.14) |
HR (95% CI)b | 0.72 (0.59 to 0.89) | 0.55 (0.39 to 0.79) | ||
Rate at 6 months, % (95% CI)a | 77.0 (72.1 to 81.1) | 62.7 (57.3 to 67.6) | 75.3 (67.0 to 81.7) | 56.7 (47.8 to 64.6) |
CI = confidence interval; HR = hazard ratio; NUTRFS = non-urothelial tract recurrence-free survival; PD-L1 = programmed death-ligand 1.
aBased on Kaplan-Meier estimates.
bStratified Cox proportional hazard model. HR is nivolumab over placebo.
Source: CheckMate 274 Clinical Study Report.9
Figure 7: Kaplan-Meier Plot of NUTRFS — All Randomized Patients on August 27, 2020 (Data Cut-Off Date)
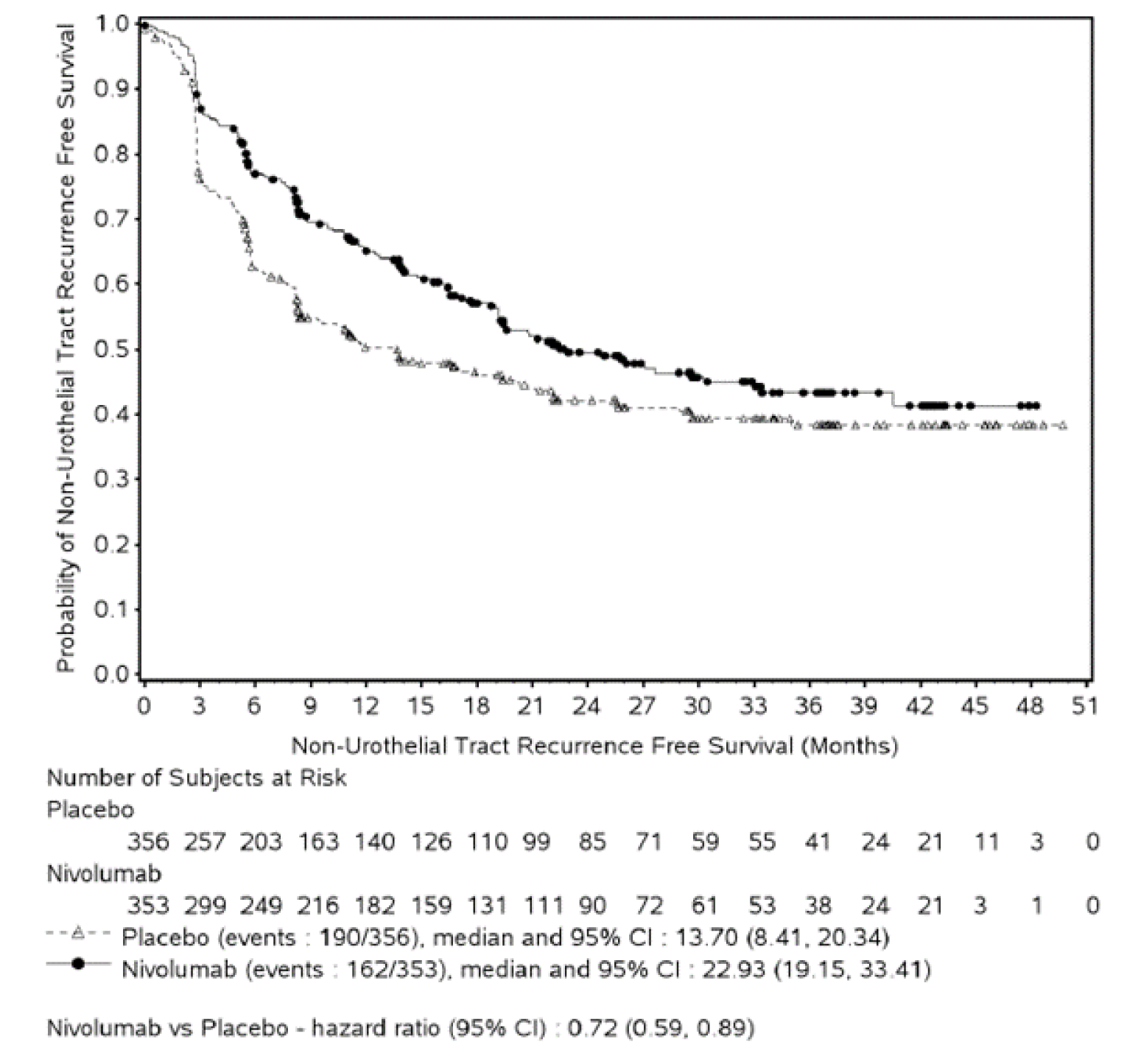
CI = confidence interval; NUTRFS = non-urothelial tract recurrence-free survival.
Note: Statistical model for hazard ratio is the stratified Cox proportional hazard. Symbols represent censored observations.
Source: CheckMate 274 Clinical Study Report erratum.11
Disease-Specific Survival
DSS was not included in the CSR or supplemental reports provided by the sponsor.
Figure 8: Mean Changes in EORTC QLQ-C30 Global Health Status Score From Baseline — All Randomized Patients on August 27, 2020 (Data Cut-Off Date) [Redacted]

The figure contained confidential information and was redacted.
Patient-reported Health Utilities and General Health Status (EQ-5D-3L)
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
At baseline, mean EQ-5D-3L utility index and EuroQol VAS scores for all randomized patients were comparable between treatment groups.
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Figure 9: Mean Changes in EQ-5D-3L Utility Index Score From Baseline — All Randomized Patients on August 27, 2020 (Data Cut-Off Date) [Redacted]

The figure contained confidential information and was redacted.
Figure 10: Mean Changes in Overall Self-Related Health Status EuroQol VAS on August 27, 2020 (Data Cut-Off Date) [Redacted]

The figure contained confidential information and was redacted.
Symptom Severity
Patients rated reduced symptoms as an important outcome of treatment; however, this was not assessed in the clinical trial. According to the clinical expert consulted for this review, patients are cancer-free, and thus free from cancer symptoms, at enrolment in the clinical trial post-surgery. Nivolumab may delay cancer recurrence and thus delay recurrence of cancer symptoms, but it would not influence post-surgery symptoms.
Distant Metastasis-Free Survival
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. Most patients were still in follow-up (142 [40.2%] in the nivolumab group and 119 [33.4%] in the placebo group). Among all randomized patients, median DMFS was 40.5 months (95% CI, 22.4 to NA) in the nivolumab group and 29.5 months (95% CI, 16.7 to NA) in the placebo group with an HR of 0.75 (95% CI, 0.59 to 0.94). At 6 months, DMFS rates were 82.5% (95% CI, 78.0 to 86.2) in the nivolumab group and 69.8% (95% CI, 64.5 to 74.4) in the placebo group. Results are presented in Table 17.
At the updated analysis with a February 1, 2021 data cut-off date, median DMFS among all treated patients was 41.1 months in the nivolumab group and 29.2 months in the placebo arm, with an HR of 0.73 (95% CI, 0.58 to 0.92).13
Time to Recurrence
At the August 27, 2020, data cut-off date, among all randomized patients, median TTR was 27.0 months (95% CI, NA to 19.5) in the nivolumab group and 11.4 months (95% CI, 20.0 to 8.4) in the placebo group with an HR of 0.67 (95% CI, 0.54 to 0.83). At 6 months recurrence rates were 37.0% (95% CI, 31.9 to 42.2) in the placebo group and 23% (95% CI, 18.7 to 27.6) in the nivolumab group. Results are presented in Table 18.
At the updated analysis (data cut-off date of February 1, 2021), median TTR among all treated patients was 25.8 (95% CI, 19.6 to NE) months in the nivolumab group and 11.1 (95% CI, 8.3 to 19.4) months in the placebo group.13
Locoregional DFS
At the August 27, 2020, the data cut-off date among all randomized patients, median LRDFS was not reached in either treatment group.
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. Results are presented in Table 19.
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Locoregional Control
Results are presented in Table 20. At the analysis as of the August 27, 2020, data cut-off date median time to LRC was not reached in either treatment group among all randomized patients. At 6 months, LRC rates were 9.7% (95% CI, 6.9 to 13.2) in the nivolumab group and 17.0% (95% CI, 13.2 to 21.2) in the placebo group. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.
Table 17: DMFS Among All Randomized Patients and Patients with Tumour Cell PD-L1 Expression ≥ 1% in the CheckMate 274 Trial on August 27, 2020 (Data Cut-Off Date)
Item | All randomized patients | Patients with tumour PD-L1 ≥ 1% | ||
|---|---|---|---|---|
Nivolumab n = 353 | Placebo n = 356 | Nivolumab n = 140 | Placebo n = 142 | |
Events, n (%) | 132 (37.4) | 152 (42.7) | 47 (33.6) | 61 (43.0) |
Median DMFS, months (95% CI)a | 40.5 (22.4 to not reached) | 29.5 (16.7 to not reached) | Not reached (25.8 to not reached) | 21.19 (10.6 to not reached) |
HR (95% CI)b | 0.75 (0.59 to 0.94) | 0.61 (0.42 to 0.90) | ||
Rate at 6 months, % (95% CI)a | 82.5 (78.0 to 86.2) | 69.8 (64.5 to 74.4) | 78.7 (70.7 to 84.8) | 65.7 (56.8 to 73.3) |
CI = confidence interval; DMFS = distant metastasis-free survival; HR = hazard ratio; PD-L1 = programmed death-ligand 1.
aBased on Kaplan-Meier estimates.
bStratified Cox proportional hazard model. HR is nivolumab over placebo.
Source: CheckMate 274 Clinical Study Report.9
Table 18: TTR Among All Randomized Patients and Patients With Tumour Cell PD-L1 Expression ≥ 1% in the CheckMate 274 Trial on August 27, 2020 (Data Cut-Off Date)
Item | All randomized patients | Patients with tumour PD-L1 ≥ 1% | ||
|---|---|---|---|---|
Nivolumab n = 353 | Placebo n = 356 | Nivolumab n = 140 | Placebo n = 142 | |
Events, n (%) | 153 (43.3) | 193 (54.2) | 47 (33.6) | 75 (52.8) |
Median TTR, months (95% CI)a | 27.0 (Not reached to 19.5) | 11.4 (20.0 to 8.4) | Not reached (Not reached to 29.67) | 11.4 (29.6 to 6.5) |
HR (95% CI)b | 0.67 (0.54 to 0.83) | 0.51 (0.3 to 0.73) | ||
Rate at 6 months (95% CI), %a | 23.0 (18.7 to 27.6) | 37.0 (31.9 to 42.2) | 23.3 (16.5 to 30.7) | 41.4 (32.9 to 49.6) |
CI = confidence interval; HR = hazard ratio; PD-L1 = programmed death-ligand 1; TTR = time to recurrence.
aBased on cumulative incidence estimates.
bStratified cause-specific hazard model.
Source: CheckMate 274 Clinical Study Report.9
Table 19: LRDFS Among All Randomized Patients and Patients With Tumour Cell PD-L1 Expression ≥ 1% in the CheckMate 274 Trial on August 27, 2020 (Data Cut-Off Date)
Item | All randomized patients | Patients with tumour PD-L1 ≥ 1% | ||
|---|---|---|---|---|
Nivolumab (n = 353) | Placebo (n = 356) | Nivolumab (n = 140) | Placebo (n = 142) | |
Events, n (%) | 76 (21.5) | 98 (27.5) | 19 (13.6) | 40 (28.2) |
Median LRDFS, months (95% CI)a | Not reached | Not reached | Not reached | Not reached |
HR (95% CI)b | 0.68 (0.50 to 0.91) | 0.41 (0.24 to 0.70) | ||
Rate at 6 months, % (95% CI)a | 11.2 (8.1 to 14.8) | 18.8 (14.8 to 23.1) | 7.5 (3.8 to 12.8) | 21.1 (14.6 to 28.4) |
CI = confidence interval; HR = hazard ratio; LRDFS = locoregional disease-free survival; PD-L1 = programmed death-ligand 1.
aBased on cumulative incidence estimates.
bStratified cause-specific hazard model.
Source: CheckMate 274 Clinical Study Report.9
Table 20: LRC Among All Randomized Patients and Patients With Tumour Cell PD-L1 Expression ≥ 1% in the CheckMate 274 Trial on August 27, 2020 (Data Cut-Off Date)
Item | All randomized patients | Patients with tumour PD-L1 ≥ 1% | ||
|---|---|---|---|---|
Nivolumab (n = 353) | Placebo (n = 356) | Nivolumab (n = 140) | Placebo (n = 142) | |
Events, n (%) | 63 (17.8) | 88 (24.7) | 14 (10.0) | 35 (24.6) |
Median LRC, months (95% CI)a | Not reached | Not reached | Not reached | Not reached |
HR (95% CI)b | 0.61 (0.44 to 0.85) | 0.33 (0.18 to 0.62) | ||
Rate at 6 months, % (95% CI)a | 9.7 (6.9 to 13.2) | 17.0 (13.2 to 21.2) | 5.3 (2.3 to 10.0) | 18.8 (12.7 to 25.9) |
CI = confidence interval; HR = hazard ratio; LRC = locoregional control; PD-L1 = programmed death-ligand 1.
aBased on cumulative incidence estimates.
bStratified cause-specific hazard model.
Source: CheckMate 274 Clinical Study Report.9
Harms
Only those harms identified in the review protocol are reported in the following. Refer to Table 21 for detailed harms data. Safety was analyzed in all treated patients.
Adverse Events
A total of 347 (98.9%) of patients in the nivolumab group and 332 (95.4%) of patients in the placebo group experienced at least 1 AE. The most common all-causality AEs (≥ 15% in either of the arms) in the nivolumab and placebo groups were pruritis (30.2% versus 16.1%), diarrhea (29.1% versus 26.1%), fatigue (27.4% versus 24.4%), urinary tract infection (19.9% versus 19.0%), rash (18.8% versus 9.8%), nausea (16.2% versus 12.6%), and constipation (13.4% versus 15.2%).
Grade 3 and 4 AEs occurred in a total of 148 (42.2%) of patients in the nivolumab group and 122 (35.1%) of patients in the placebo group. The most common all-causality grade 3 and 4 AEs in the nivolumab and placebo groups were urinary tract infection (5.4% versus 6.3%) and diarrhea (1.7% versus 1.1%).
Serious Adverse Events
A total of 104 (29.6%) of patients in the nivolumab group and 105 (30.2%) of patients in the placebo group experienced an all-causality SAE. The most common SAEs (≥ 2% in either of the arms) in the nivolumab versus the placebo group were urinary tract infection (2.6% versus 6.0%) and malignant neoplasm progression (2.3% versus 5.5%).
Withdrawals Due to AEs
All-causality AEs leading to study drug discontinuation occurred in 64 (18.2%) of patients in the nivolumab group versus 32 (9.2%) in the placebo group; the most commonly reported were pneumonitis (1.7% versus 0.3%) and malignant neoplasm progression (1.4% versus 2.6%).
Mortality
There were 95 (27.1%) deaths in the nivolumab group and 107 (30.7%) deaths in the placebo group, most commonly due to disease progression (73 [20.8%] in the nivolumab group and 90 [25.9%] in the placebo arm). There were 4 (1.1%) deaths in the nivolumab group and 9 (2.6%) deaths in the placebo arm within 30 days of the last dose, and 16 (4.6%) deaths in the nivolumab group and 35 (10.1%) deaths in the placebo group within 100 days of the last dose. There were 3 treatment-related deaths in the nivolumab group: 2 due to pneumonitis and 1 due to bowel perforation.17,39
Notable Harms
IMAEs were more frequently reported in patients in the nivolumab group than in the placebo arm, and included rash (11.4% versus 2.3%), pneumonitis (4.8% versus 0.6%), diarrhea or colitis (4.0% versus 0.9%), hepatitis (2.8% versus 0.3%), nephritis or renal dysfunction (2.0% versus 0.9%), and hypersensitivity/infusion reactions (0.6% versus 0.0).
Table 21: Summary of Harms in the CheckMate 274 Trial in All Treated Patients on August 27, 2020 (Data Cut-Off Date)
Harms | Nivolumab (n = 351 | Placebo (n = 348) |
|---|---|---|
Patients with ≥ 1 AE | 347 (98.9) | 332 (95.4) |
Grade 3 or 4 | 148 (42.2) | 122 (35.1) |
Most common eventsa | ||
Pruritus | 106 (30.2) | 56 (16.1) |
Diarrhea | 102 (29.1) | 91 (26.1) |
Fatigue | 96 (27.4) | 85 (24.4) |
Urinary tract infection | 70 (19.9) | 66 (19.0) |
Rash | 66 (18.8) | 34 (9.8) |
Nausea | 57 (16.2) | 44 (12.6) |
Constipation | 47 (13.4) | 53 (15.2) |
Patients with ≥ 1 SAE | 104 (29.6) | 105 (30.2) |
Grade 3 or 4 | 81 (23.1) | 73 (21.0) |
Most common eventsb | ||
Urinary tract infection | 9 (2.6) | 21 (6.0) |
Malignant neoplasm progression | 8 (2.3) | 19 (5.5) |
Patients who stopped treatment due to AEs | 64 (18.2) | 32 (9.2) |
Grade 3 or 4 | 39 (11.1) | 21 (6.0) |
Most common eventsc | ||
Pneumonitis | 6 (1.7) | 1 (0.3) |
Malignant neoplasm progression | 5 (1.4) | 9 (2.6) |
Rash | 4 (1.1) | 0.0 |
Deaths | 95 (27.1) | 107 (30.7) |
Primary reason for death | ||
Disease | 73 (20.8) | 90 (25.9) |
Study drug toxicity | 3 (0.9) | 0.0 |
Unknown | 3 (0.9) | 3 (0.9) |
Other | 17 (4.8) | 14 (4.0) |
Notable harms | ||
IMAEsd | ||
Rash | 40 (11.4) | 8 (2.3) |
Pneumonitis | 17 (4.8) | 2 (0.6) |
Diarrhea/colitis | 14 (4.0) | 3 (0.9) |
Hepatitis | 10 (2.8) | 1 (0.3) |
Nephritis/renal dysfunction | 7 (2.0) | 3 (0.9) |
Hypersensitivity/infusion reactions | 2 (0.6) | 0.0 |
AE = adverse event; IMAE = immune-mediated adverse event; SAE = serious adverse event.
Note: Values are n (%). AEs were defined and graded using MedDRA version 23.0 and Common Terminology Criteria for Adverse Events version 4.0. All events are within 30 days of the last dose of study drug unless otherwise indicated (any time for deaths, 100 days for IMAEs and other events of special interest).
Note: Results are from the July 10, 2020 database lock.
aAEs of 15% or greater in either study group are shown.
bSAEs with frequency of 2% or greater in either study group are shown.
cAEs leading to discontinuation with frequency in greater than 1% in either study group are shown.
dAll-causality IMAEs within 100 days of last dose treated with immune-modulating medication.
Source: CheckMate 274 Clinical Study Report.9
Critical Appraisal
Internal Validity
The included pivotal study, CheckMate 274, was a phase III, randomized, double-blind, placebo-controlled study of nivolumab in adult patients who had undergone radical resection of MIUC originating in the bladder or upper urinary tract (renal pelvis or ureter) and were at high risk of recurrence (N = 709). Despite the trial’s blind design, it is possible that some AEs, such as IMAEs, allowed the possible detection of the intervention being received by some patients. If trial investigators or patients were aware of the intervention assignment, this may have affected behaviour (such as initiation of subsequent treatment given that DFS was investigator assessed or adherence to treatment) or perceived HRQoL. The study used an appropriate randomization method (1:1 using an interactive voice response system) but allocation concealment procedures were not adequately described. Randomization appeared adequate in balancing baseline demographic and disease characteristics between the nivolumab and placebo arms.
A memo to the Data Monitoring Committee describing an unintentional unmasking issue following the August 27, 2020, data cut-off date was submitted by the sponsor. The efficacy and safety analyses at the August 27, 2020, data cut-off date were not influenced due to the timing of this event. The investigation into this event concluded that, given the short time during which the data were unmasked and the absence of active data cleaning following the DBL, the chances of any unauthorized person seeing the data were extremely low.
According to the protocol, the time between complete resection and randomization should be 120 days; however, there were protocol deviations where approximately 4% of patients were randomized 120 or more days after resection. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||. Nevertheless, the proportions of patients that experienced a dose delay or infusion interruption were comparable (< 5%) in both treatment arms. Due to the limited number of patients who were affected by protocol deviations or were treated with nivolumab for more than 12 months, no significant impact on the comparative clinical efficacy of nivolumab versus placebo would be expected.
Maintaining quality of life was rated as an important outcome by patients, yet there was no formal statistical comparison and there were missing HRQoL data at later time points post-baseline. The interpretation of results for the HRQoL instruments (i.e., the ability to assess trends over time and to make comparisons across treatment groups) is limited by the significant decline in patients available to provide assessment over time. The CADTH review team conducted an assessment on the validity of outcome measures and did not find evidence of validity or an MID of the EORTC QLQ-C30 or EQ-5D-3L in patients with UC. The clinical experts consulted by CADTH pointed out that since patients are disease-free at the start of the study, they are likely to experience stable quality of life scores, except for IMAEs or the effects of clinical visits for assessment or infusions for patients who would have otherwise been under surveillance.
Symptom reduction was identified as an outcome in the CADTH review protocol; however, it was not assessed as a separate outcome in the pivotal study as it was not considered applicable in a disease-free patient population.
Updated results for DFS, NUTRFS, DMFS, and TTR from the May 19, 2021, DBL (data cut-off February 1, 2021) were consistent overall with results from the final primary analysis. Results were only available in poster format (poster presentation at the Society of Urologic Oncology Congress in December 202113) and no CSR was provided for this data cut-off, so the CADTH review team was unable to conduct a rigorous evaluation of the methods and reporting of these analyses.
OS was considered an outcome of primary importance by the clinical experts consulted by CADTH in guiding treatment selection in clinical practice. The first interim analysis for OS was planned with the February 1, 2021, data cut-off date at which point OS did not cross the pre-specified boundary for declaring statistical significance.12 No OS data were submitted by the sponsor. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.
External Validity
According to the clinical experts CADTH consulted for this review, the CheckMate 274 study population is considered reflective of the requested reimbursement population. The following considerations are of importance regarding the external validity of the study.
Population: According to the clinical experts consulted by CADTH for this review, the demographic and disease characteristics of the CheckMate 274 study9 population were reflective of the Canadian population with MIUC. More males than females and more patients older than 65 years were enrolled in both arms, which is consistent with the population of patients in clinical practice according to the clinical experts. Most participants were White, male, and staged at pT3 or lower; however, the clinical experts did not think this would limit generalizability to patients in Canadian clinical practice. The study protocol was amended based on findings from the CA209275 study40 (46% of study patients were PD-L1 positive) to cap PD-L1-negative patients included in the study at 54%. The clinical experts consulted by CADTH noted that the PD-L1 biomarker is currently not used in Canadian clinical practice to guide treatment selection in the target population. The experts noted that research on this biomarker’s definitions, methods of measurement, and cut-off values are currently still evolving. The trial capped the proportion of patients with UTUC at 20% as supported by previous studies and confirmed by clinical experts consulted by CADTH. The experts felt that it was reasonable to generalize the CheckMate 274 study results to patients with UTUC because of the similar etiology between UTUC and bladder cancer, and because patients with UTUC were included in the pivotal trial and are treated similarly to patients with bladder cancer in Canadian clinical practice. Almost 98% of patients in the CheckMate 274 trial had ECOG PS 0 or 1. The experts anticipated that, in clinical practice, a higher proportion of patients with ECOG PS 2 may receive nivolumab because recurrence of the cancer is high and AEs are tolerable. Cisplatin ineligibility was defined using the Galsky criteria14 which are commonly used in clinical trials and clinical practice. The clinical experts consulted by CADTH noted that experienced clinicians may apply some flexibility in terms of using adjuvant chemotherapy in patients with a creatinine clearance of greater than 50 mL/min and those with hearing loss if patients prefer to receive adjuvant chemotherapy after a discussion of the clinical risks.
The study included 3 groups of patients at high risk of disease recurrence: patients who received neoadjuvant cisplatin-based therapy and were therefore not eligible for adjuvant cisplatin-based therapy, and patients who did not receive neoadjuvant cisplatin-based therapy and were either cisplatin ineligible (155 [21.9%]) or were cisplatin eligible but refused adjuvant chemotherapy (231 [32.6%]). The clinical experts noted that data on adjuvant nivolumab compared to adjuvant cisplatin-based chemotherapy in patients who have not received neoadjuvant chemotherapy and were eligible to receive cisplatin-based chemotherapy was not available from the CheckMate 274 trial. Given the absence of robust comparative data between adjuvant nivolumab and adjuvant chemotherapy, the clinical experts consulted by CADTH were uncertain whether nivolumab addressed an unmet need in patients at high risk of recurrence who are eligible for adjuvant cisplatin-based chemotherapy. The clinical experts noted that more robust direct evidence from a randomized trial (e.g., a previous RCT in this setting was conducted by Sternberg et al.15) is required to address the comparative effectiveness and safety of nivolumab compared with cisplatin-based chemotherapy in the adjuvant setting. In the absence of robust comparative data, the clinical experts anticipated that adjuvant nivolumab would be the preferred treatment over adjuvant chemotherapy in select clinical circumstances only (e.g., gemcitabine allergy or strong patient preference against chemotherapy). The clinicians from the Cancer Care Ontario Genitourinary Cancer Drug Advisory Committee providing input for this submission concurred with the clinical experts consulted by CADTH in that they noted that the comparative effectiveness between adjuvant nivolumab and chemotherapy is unknown at the moment, and it may be possible that patients eligible for cisplatin-based adjuvant chemotherapy may be better suited for chemotherapy than nivolumab. These clinicians noted that currently neither adjuvant nivolumab (long-term OS results are awaited from the CheckMate 274 trial) nor adjuvant chemotherapy have demonstrated an OS benefit versus surveillance. The CheckMate 274 trial was not designed to detect differences in treatment effects across subgroups of cisplatin-eligible versus cisplatin-ineligible patients and the clinical experts noted that any assumption about the extent to which the subgroup of cisplatin-eligible patients may have influenced the results seen in the overall trial population is speculative.
Appropriateness of comparator: The clinical experts consulted by CADTH agreed that placebo was an appropriate comparator in Canadian clinical practice for patients at high risk of recurrence after radical resection of MIUC who are not eligible to receive adjuvant chemotherapy. The clinical experts agreed that, currently, there are no active alternative treatment options in the adjuvant setting for these patients. However, the clinical experts noted that for patients who are at high risk of recurrence and are eligible for adjuvant chemotherapy (regardless of whether they decline such treatment) the appropriate comparator would be adjuvant chemotherapy and not placebo.
Relevance of DFS as primary end point: The clinical experts consulted by CADTH and clinician groups providing input agreed that delaying recurrence in the adjuvant setting of UC is a clinically meaningful end point to clinicians and their patients. Once UC recurs, the prognosis of patients is poor (median post-recurrence OS in patients with UC of the bladder has been estimated to be approximately 6 months). The clinical experts consulted by CADTH and the clinician groups agreed that an absolute improvement of 10 months in median DFS with nivolumab compared with placebo as observed in the CheckMate 274 trial is clinically meaningful in patients at high risk of recurrence who are currently not eligible for adjuvant chemotherapy. Remaining disease-free for a longer period of time is of relevance to the patients as recurrence is associated with disease symptoms and likely decreased quality of life. However, the clinical experts consulted by CADTH anticipated that in the comparison of adjuvant nivolumab against an active comparator (e.g., adjuvant chemotherapy), primarily OS rather than DFS would guide treatment selection in the adjuvant setting.
DFS is an established end point in the adjuvant setting, and benefits have been noted in guidance from the FDA and EMA.41,42 A review of studies assessing the appropriateness of DFS as a surrogate outcome is presented in Appendix 5. At the individual level, there was a moderate to substantial agreement between DFS and OS. However, in the absence of the trial-level association between DFS and OS in the present target population, it cannot be firmly concluded to what extent the improvements in DFS observed in patients in the nivolumab group of the CheckMate 274 trial would translate into OS benefits.
Intervention: The reimbursement request is for consideration of nivolumab 240 mg every 2 weeks or 480 mg every 4 weeks; however, the pivotal study only included dosing of 240 mg every 2 weeks. The clinical experts felt that the results of the CheckMate 274 trial could be generalized to a dosing of 480 mg every 4 weeks as this dosing regimen has been previously approved for nivolumab as a monotherapy with other indications.
Setting: This study was a multinational, multi-centre trial. The study population was drawn from a wide variety of different sites across the globe, with 14.4% of patients in the US ||||||||||||||||||||. The clinical experts indicated that there is no concern generalizing the findings from the pivotal study to the Canadian clinical setting.
Indirect Evidence
Objectives and Methods for the Summary of Indirect Evidence
The CheckMate 274 trial compared nivolumab to placebo in adult patients who have undergone radical resection of MIUC originating in the bladder or upper urinary tract and are at high risk of recurrence. The objective of this section is to summarize and critically appraise available indirect evidence comparing nivolumab to other relevant comparators for adjuvant treatment of MIUC. One sponsor-submitted ITC, prepared by OPEN Health for the sponsor,16 is summarized and critically appraised in the following.
In addition, a supplemental search of the medical literature for publicly available ITCs was conducted by the CADTH review team. A focused literature search for NMAs dealing with Opdivo (nivolumab) and UC was run in MEDLINE All (1946–) on March 22, 2022. No limits were applied to the search. Five potentially relevant articles were retrieved, and only 1 published NMA by Laukhtina et al.43 was included.
Therefore, the sponsor-submitted NMA and 1 published NMA were appraised and summarized for this review.
Description of Indirect Comparisons
The sponsor submitted a separate report with details on the systematic review conducted before the ITC; details on study selection criteria and methods are presented in Table 22.
Table 22: Study Selection Criteria and Methods for ITCs
Criteria | Sponsor-submitted ITC | Laukhtina et a. (2022) |
|---|---|---|
Population | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | Non-organ confined bladder UC or high-risk patients with UTUC |
Intervention | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | Treated with adjuvant systemic therapy after radical surgery (radical cystectomy or radical nephroureterectomy) |
Comparator | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | Observation or placebo after radical surgery |
Outcome | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | Differential effects on disease progression rate and AEs |
Study design | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | Phase III randomized studies |
Publication characteristics | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | English |
Exclusion criteria | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | Phase I and II clinical trials, observational studies, reviews, letters, editorials, replies from authors, case reports, and articles not published in English |
Databases searched | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | PubMed and Web of Science |
Selection process | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | 2 reviewers screened, independently, the titles and abstracts then full texts of eligible articles; discrepancies were resolved by referring to the co-authors in a Delphi consensus |
Data extraction process | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| | Data from each study were independently extracted by 2 reviewers |
Quality assessment | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||44 ||||||||||||||||||||||||||||||||||||||||||||45 | The risk-of-bias evaluation of each study was assessed |
AE = adverse event; ITC = indirect treatment comparison; UC = urothelial carcinoma; UTUC = upper tract urothelial carcinoma.
Source: Sponsor-submitted ITC and systematic literature review16 and Laukhtina et al. (2022).43
Methods of Sponsor-Submitted NMA
Objectives
Indirect evidence from 1 sponsor-submitted NMA evaluated the effectiveness of nivolumab compared to cisplatin-based chemotherapy in the treatment of UC. The NMA addressed a gap in the pivotal clinical trial, which included a subgroup of patients who were cisplatin eligible but declined to take it. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.
Study Selection Methods
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||47||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
ITC Analysis Methods
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||47||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||47||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Results of Sponsor-Submitted NMA
Summary of Included Studies
The systematic literature review identified 73 studies as potentially relevant for the ITCs. A total of 5 randomized trials were included in the NMA: CheckMate 274 Group C, Cognetti et al.,48 Lehmann et al.,49 Sternberg et al.,15 and Zhegalik et al.50
A heterogeneity analysis provided a comparison on the similarity of the included studies according to the PICOS framework: population, intervention, comparator, outcome, and study design. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Results
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Figure 11: Network of Evidence [Redacted]

This figure contained confidential information and was redacted.
Table 23: Assessment of Homogeneity for Sponsor-Submitted NMA
||||||||||||||||||||||||||||||| | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |
|---|---|
||||||||||||||||||||||||||||||| | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |
||||||||||||||||||||||||||||||| | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |
||||||||||||||||||||||||||||||| | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |
||||||||||||||||||||||||||||||| | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||| |
NMA = network meta-analysis.
Note: Redacted rows have been deleted.
||||||||||||||||||||| | ||||||||||||||||||||| | ||||
|---|---|---|---|---|---|
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| |
||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| | ||||||||||||||||||||| |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||16
Critical Appraisal of Sponsor-Submitted NMA
In the sponsor-submitted ITC, there was heterogeneity in the tumour staging of patients, definition of end points, treatment doses and regimens, and median follow-up times. ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||.Overall, the methodological concerns identified and the observed heterogeneity across study designs and populations precluded drawing definitive conclusions about the comparative effectiveness of adjuvant nivolumab versus adjuvant chemotherapy.
Methods of Laukhtina et al.43
Objectives
The main objective of this NMA is to determine oncologic and toxicity outcomes of adjuvant immunotherapy with ICIs (including nivolumab) compared to adjuvant chemotherapy.
Study Selection Methods
The searches were conducted in PubMed and Web of Science in June 2021 to capture phase III randomized studies. Inclusion criteria included non-organ confined bladder UC or high-risk patients with UTUC, treated with adjuvant systemic therapy after radical surgery compared with those who underwent observation or received placebo after radical surgery, to assess the differential effects on disease progression rate and AEs. Two reviewers screened abstracts and full text, conducted data extraction, and assessed the risk of bias of each study. Full details on the study selection methods are presented in Table 22.
ITC Analysis Methods
The authors conducted an NMA using random and fixed-effect models with a Bayesian approach to compare treatments directly and indirectly with observation or placebo as the common comparator arm. Arm-based analyses were performed to estimate OR and 95% Crl to evaluate the disease progression rate in bladder UC and UTUC separately. A 2-sided P of less than 0.05 or 95% CrI that did not include a value of 1 established statistical significance.
Results of Laukhtina et al.43
Summary of Included Studies
The authors screened 356 records and the NMA included 9 studies comprising 2,444 patients. Two studies involved an assessment of ICI therapy (nivolumab [n = 353] and atezolizumab51 [n = 406]) including the pivotal study of this review,17 5 studies involved assessment of cisplatin-based chemotherapy (n = 468, regimens including: cisplatin; GC; cisplatin, vinblastine, and methotrexate; MVAC or MVEC; cisplatin, doxorubicin, and cyclophosphamide) in patients with bladder UC,48,49,52-54 and 2 studies involved assessment of cisplatin- or platin-based chemotherapy (gemcitabine with cisplatin or carboplatin) in patients with UTUC.22,55 In the 9 RCTs, patients were either receiving placebo (n = 356) or under observation (n = 871). According to risk of bias, trials were of moderate quality, with risks of bias due to lack of blinding and bias in the selection of reported results.
There was heterogeneity in the sample sizes across the included trials (range: 49 to 809 patients); 4 older chemotherapy studies had a sample size of less than 100 patients. The included patients differed in cancer staging across the studies and were not specific to patients with MIUC: 1 study even included all stages of UC (T1 to T4, and/or pN+, M0). There was also heterogeneity in the outcomes reported across studies; 3 older studies did not report any outcome measure, 4 studies reported DFS, and 2 studies reported PFS. Follow-up time differed across the trials, with the median follow-up time ranging from 20.9 months to 160 months. Finally, there was high heterogeneity in the chemotherapy regimens among the included trials, but all were cisplatin-based. The clinical expert consulted by CADTH agreed that these regimens were comparable except for the single-agent cisplatin RCT included in this analysis.
Results
Treatment approaches were categorized into 4 groups: anti-PD-L1 therapy (atezolizumab), anti-PD-1 therapy (nivolumab), chemotherapy, and observation or placebo.
In patients with bladder UC, chemotherapy (OR = 0.50; 95% CrI, 0.19 to 1.06), atezolizumab (OR = 1.01; 95% CrI, 0.19 to 5.46), and nivolumab (OR = 0.59; 95% CrI, 0.11 to 3.34) were not significantly associated with a lower likelihood of the disease progression rate compared to observation or placebo. A network plot of RCTs and a summary of the Bayesian NMA of disease progression rate is presented in Figure 12.
In patients with UTUC chemotherapy (OR = 0.36; 95% CrI, 0.13 to 0.92) was significantly associated with a lower likelihood of disease progression, while atezolizumab (OR = 1.39; 95% Crl, 0.28 to 7.25) and nivolumab (OR = 1.21; 95% CrI, 0.29 to 4.95) were not compared to observation or placebo. A network plot of RCTs and a summary of the Bayesian NMA of disease progression rate are presented in Figure 13.
The trials that assessed cisplatin-based chemotherapy did not report comparative AEs, therefore the authors only conducted an NMA between ICI agents. Atezolizumab (OR = 2.80; 95% CrI, 0.47 to 16.4) and nivolumab (OR = 4.37; 95% CrI, 0.65 to 30.7) were not associated with a higher likelihood of all AEs compared to observation or placebo.
Critical Appraisal of Laukhtina et al.43
This NMA included a limited number of studies with some heterogeneity across these studies, including components of the chemotherapy regimen and the median follow-up time. Four trials were older chemotherapy trials with smaller sample sizes and inconsistent reporting of outcomes which may have led to confounding of the results. The clinical expert consulted by CADTH confirmed that the chemotherapy regimens are part of the adjuvant chemotherapy treatment paradigm except for the RCT with single-agent cisplatin therapy which is not part of the current clinical paradigm according to the clinical experts consulted by CADTH. The 2 trials involving the assessment of ICIs including the pivotal trial for nivolumab, provided immature follow-up with no OS data. Moreover, the AEs were not reported or comparable in the chemotherapy RCTs so the authors could not include them in the toxicity NMA.
Figure 12: Summary of the Bayesian Network Meta-Analysis of Disease Progression Rate in Patients Treated With Adjuvant Therapy for Bladder Cancer
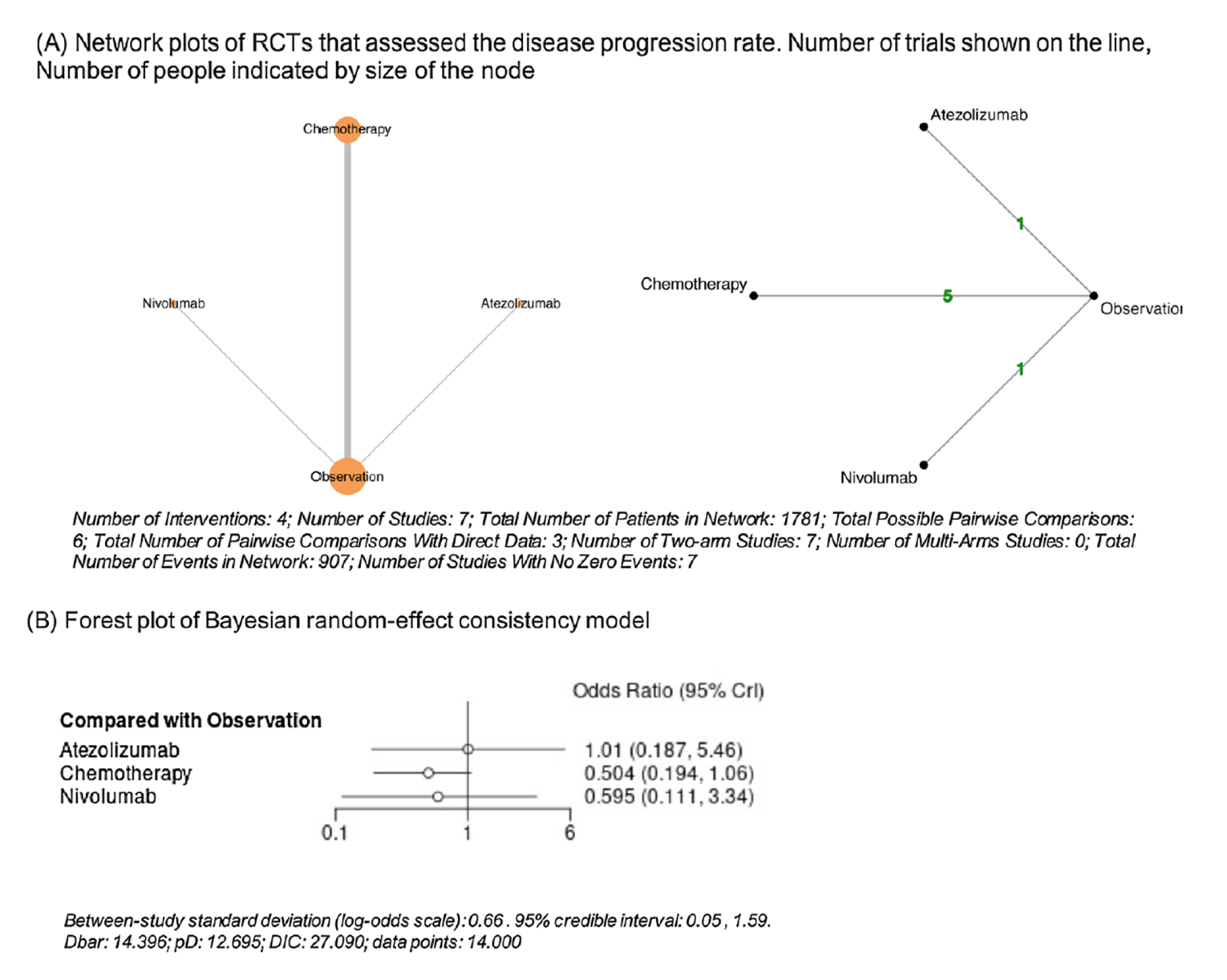
CrI = credible interval; Dbar = posterior mean of the deviance; DIC = deviance information criterion; RCT = randomized controlled trial.
Source: Laukhtina, et al. (2022).43 Reprinted in accordance with Attribution 4.0 International (CC BY 4.0) https://creativecommons.org/licenses/by/4.0/.
The CADTH review team was unable to rigorously assess the methods in this article because insufficient details on the methods were provided, and there was no discussion on possible adjustments for heterogeneity, potential effect modifiers, or feasibility assessment. The authors discussed the use of DFS as a surrogate for OS in the ICI trials and concluded that “further analyses of mature OS data should be performed.”43 This article also included all patients from the CheckMate 274 trial and not only the patients who were eligible for cisplatin-based chemotherapy but refused cisplatin-based chemotherapy, similar to the sponsor-submitted ITC. They also used data from the August 27, 2020, data cut-off as per the published article,17 rather than data from the later data cut-off date used in the sponsor-submitted ITC.
Figure 13: Summary of the Bayesian Network Meta-Analysis of Disease Progression Rate in Patients Treated With Adjuvant Therapy for Upper Tract Urothelial Carcinoma
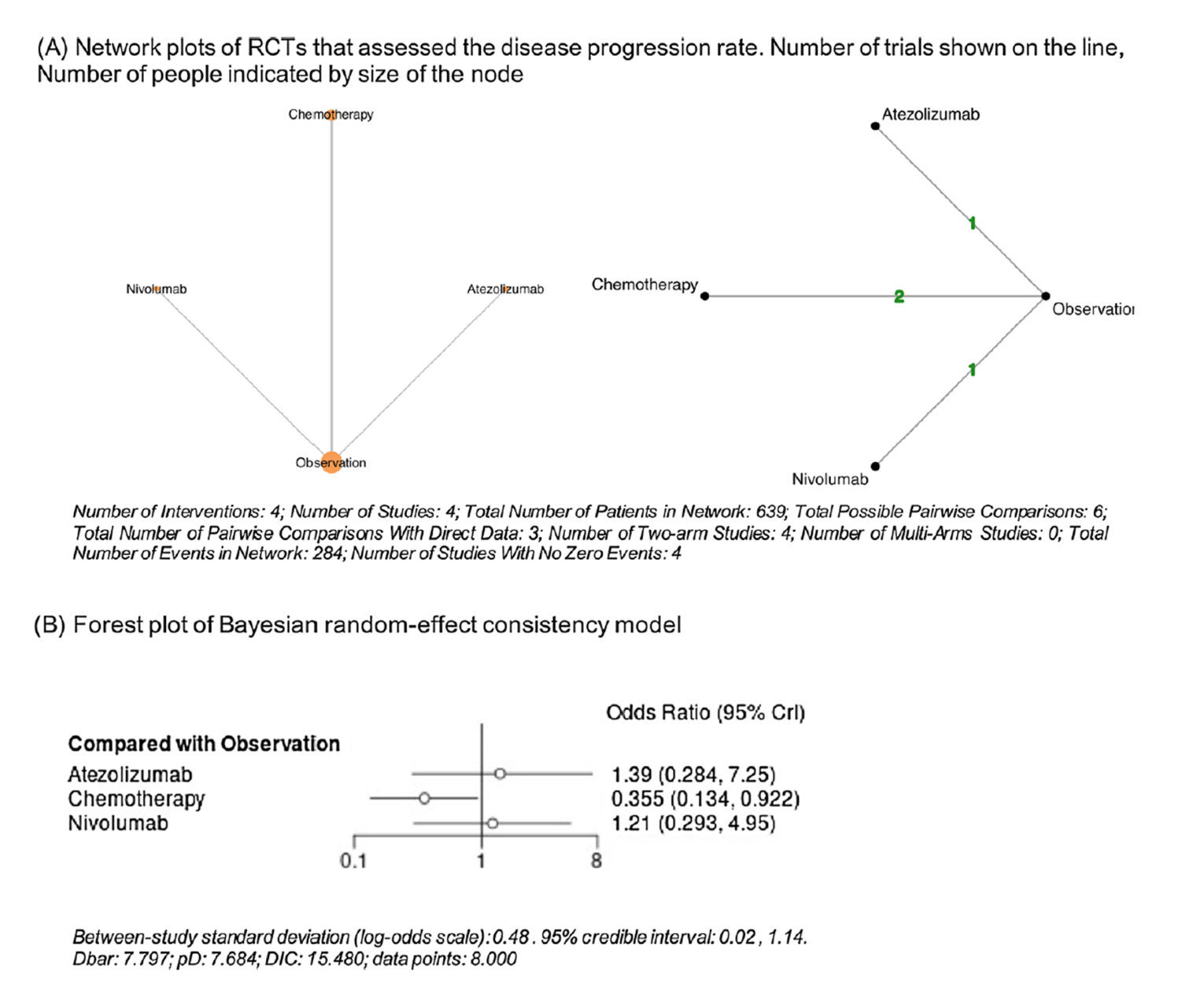
CrI = credible interval; Dbar = posterior mean of the deviance; DIC = deviance information criterion; RCT = randomized controlled trial.
Source: Laukhtina, et al. (2022).43 Reprinted in accordance with Attribution 4.0 International (CC BY 4.0) https://creativecommons.org/licenses/by/4.0/.
Summary
Indirect evidence from 2 NMAs (1 sponsor-submitted NMA and 1 published NMA) evaluated the effectiveness of nivolumab compared to cisplatin-based chemotherapy in the treatment of UC. They addressed a gap in the evidence in that comparative data between adjuvant nivolumab and adjuvant chemotherapy in the subgroup of patients who are cisplatin eligible but who were unwilling to take it, were not available from the pivotal trial.
Description of Studies
A total of 5 randomized trials (N = 857) were included in the sponsor-submitted ITC.16 The list of comparators included for the analysis included GC, MVAC, and MVEC. The NMA was conducted in a Bayesian framework using non-informative priors. Convergence was assessed by examining the Gelman-Rubin statistics, the size of the Monte Carlo error, auto-correlation function, trace plots, and Kernel density plots. The sponsor-submitted ITC included a feasibility assessment using 3 steps: creating networks of evidence based on the systematic review results, conducting a heterogeneity assessment to explore whether there were systematic differences among the studies included in the network across treatment comparisons that might affect the validity of the NMA results, and testing of the proportional assumption for the CheckMate 274 trial.
The published NMA included 9 studies comprising 2,444 patients: 2 studies involved an assessment of ICI therapy (nivolumab [n = 353] and atezolizumab [n = 406]) including the pivotal study of this review,17 5 studies involved assessment of cisplatin-based chemotherapy (n = 468, regimens include: cisplatin; GC; cisplatin, vinblastine, and methotrexate; MVAC or MVEC; cisplatin, doxorubicin, and cyclophosphamide) in patients with bladder UC, and 2 studies involved assessment of cisplatin- or platin-based chemotherapy (gemcitabine with cisplatin or carboplatin) in patients with UTUC. The authors conducted an NMA using random and fixed-effect models with a Bayesian approach to compare treatments directly and indirectly with observation or placebo as the common comparator arm. Arm-based analyses were performed to estimate OR and 95% Crl to evaluate the disease progression rate in bladder UC and UTUC separately.
Efficacy Results
In both NMAs, cisplatin-based chemotherapy was superior to nivolumab with regards to outcome measure.
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
In the published NMA, in patients with bladder UC, chemotherapy (OR = 0.50; 95% CrI, 0.19 to 1.06), atezolizumab (OR = 1.01; 95% CrI, 0.19 to 5.46), and nivolumab (OR = 0.59; 95% CrI, 0.11 to 3.34) did not lower the likelihood of disease progression rate compared to observation or placebo. In patients with UTUC, chemotherapy (OR = 0.36; 95% CrI, 0.13 to 0.92) was significantly associated with a lower likelihood of disease progression compared to observation or placebo. On the other hand, ICI therapies atezolizumab (OR = 1.39; 95% Crl, 0.28 to 7.25) and nivolumab (OR = 1.21; 95% CrI, 0.29 to 4.95) were not favourable to observation or placebo.
Harms Results
Both ITCs did not assess harms outcomes due to heterogeneity in the reporting of AEs across individual studies.
Critical Appraisal
Both NMAs included a limited number of studies with heterogeneity across these studies. In the sponsor-submitted ITC, there was heterogeneity in the tumour staging of patients, definition of end points, treatment doses and regimens, and median follow-up times. Moreover, 3 studies were single-centre studies in Europe. In the published NMA, there was heterogeneity in the components of the chemotherapy regimen and the median follow-up time. Four trials were older chemotherapy trials with smaller sample sizes and inconsistent reporting of outcomes which may have led to confounding of the results. The methodological concerns regarding heterogeneity across study designs and populations in the ITCs precluded drawing definitive conclusions about the comparative effectiveness of adjuvant nivolumab versus adjuvant chemotherapy.
Other Relevant Evidence
No long-term extension studies and additional relevant studies were included in the sponsor’s submission to CADTH.
Discussion
Summary of Available Evidence
The CADTH systematic review included 1 pivotal phase III RCT (CheckMate 274).
The CheckMate 274 study is an ongoing phase III, randomized, double-blind, placebo-controlled, multi-centre, superiority study comparing nivolumab to placebo in patients at high risk of recurrence after radical resection of MIUC (with primary site either in the bladder or upper urinary tract). A total of 709 patients were randomized in a 1:1 ratio to receive nivolumab (n = 353) or placebo (n = 356), stratified by PD-L1 expression, pathologic nodal status, and use of cisplatin-neoadjuvant chemotherapy. No crossover between the treatment groups was permitted.
The primary outcome was DFS, which was defined as the time between randomization date and first date of recurrence or death from all causes, whichever occurred first. Recurrence was defined as the appearance of 1 or more new lesions, which can be local, regional, or distant in location from the primary resected site (by imaging or pathology). OS was assessed as key secondary outcome, and NUTRFS and DSS were assessed as secondary outcomes. The results of OS and DSS were not available at the time of this review. Exploratory outcomes were patient-reported HRQoL, DMFS, TTR, and LRC.
The CheckMate 274 trial enrolled adult patients who were ineligible for, or eligible for but refused, adjuvant therapy with staging criteria of pT3-pT4a or pN+ and patients who received neoadjuvant cisplatin-based chemotherapy with staging criteria of ypT2-pT4a or ypN+. Patients were eligible to enter the trial after radical surgical resection for invasive urothelial carcinoma, with evidence of UC (originating in bladder, ureter, or renal pelvis) at high risk of recurrence based on pathological staging, neoadjuvant cisplatin chemotherapy ineligible criteria based on Galsky criteria, PD-L1 status (PD-L1 > 1%, PD-L1 < 1%, or indeterminate), and all patients had to have disease-free status defined as no clinical or radiograph evidence or recurrence of disease.
Indirect evidence from 2 NMAs (1 sponsor-submitted NMA and 1 published NMA) evaluated the effectiveness of nivolumab compared to cisplatin-based chemotherapy in the treatment of UC. The indirect evidence addressed a gap in the evidence in that the placebo-controlled pivotal clinical trial did not provide a comparison of adjuvant nivolumab to adjuvant chemotherapy for the subgroup of patients that were cisplatin eligible but unwilling to take adjuvant chemotherapy. In both NMAs, cisplatin-based chemotherapy was superior to nivolumab with regards to DFS and disease progression rate. However, both NMAs included a limited number of heterogenous studies, and no firm conclusion can be drawn about the comparative effectiveness of adjuvant nivolumab versus adjuvant chemotherapy.
Interpretation of Results
Efficacy
Administration of nivolumab in the CheckMate 274 study9 resulted in statistically significant prolongation of DFS compared with placebo in all randomized patients (median DFS of 20.8 months versus 10.8 months), a difference deemed as meaningful according to the clinical experts. In the subgroups identified as important by the clinical experts, DFS HRs favoured nivolumab over placebo regardless of tumour PD-L1 expression, use of neoadjuvant cisplatin treatment, or nodal status. The trial was not designed to detect differences in treatment effects across subgroups, so no conclusions can be made on the basis of subgroup results. At the individual patient level, there has been moderate to substantial agreement between the DFS and OS found in studies examining the association between DFS and OS in MIUC. However, in the absence of the trial-level association between DFS and OS in the present target population, it cannot be firmly concluded to what extent the improvements in DFS observed in patients in the nivolumab group of the CheckMate 274 trial would translate into OS benefits. The clinical experts consulted by CADTH and the input from clinician groups noted that OS is of primary importance in guiding treatment selection in the adjuvant setting. Given the lack of OS data from the pivotal trial, it remained uncertain if adjuvant nivolumab increased the cure rate compared to surveillance. Although not formally tested, NUTRFS results were also consistent with DFS results among patients who received nivolumab compared to placebo (median NUTRFS of 22.9 months versus 13.7 months).
Clinical experts consulted by CADTH agreed that placebo was a relevant comparator for patients who were ineligible for cisplatin-based chemotherapy, and that adjuvant nivolumab addressed a gap in the current treatment paradigm. Specifically, nivolumab would act as an effective alternative to surveillance for cisplatin-ineligible patients at high risk of recurrence, and as effective adjuvant treatment for patients at high risk of recurrence after cystectomy subsequent to treatment with neoadjuvant chemotherapy. However, the clinical experts felt that in patients who were eligible for cisplatin-based chemotherapy but refused to take it, adjuvant chemotherapy rather than placebo would be the relevant comparator. Comparative data of adjuvant nivolumab versus adjuvant chemotherapy were not available from the pivotal trial. The ITC evidence presented in this review suggested that adjuvant cisplatin-based chemotherapy was superior to adjuvant nivolumab in terms of DFS and disease progression rate outcomes; however, the included NMAs had limitations associated with clinical and methodological heterogeneity which precluded drawing definitive conclusions about the comparative effectiveness of adjuvant nivolumab versus adjuvant chemotherapy.
The clinical experts consulted by CADTH were uncertain if nivolumab addressed an unmet need in patients who are eligible for cisplatin-based adjuvant chemotherapy given the absence of comparative OS data between adjuvant nivolumab and adjuvant chemotherapy.
Patient input to CADTH rated “preventing recurrence” as the most important outcome, and that they would be willing to tolerate significant side effects from drugs that can prevent recurrence. One patient stated that “side effects are easier to live with than not living with loved ones or having to go through chemo treatment.” HRQoL analyses were descriptive only and limited by high rates of missing data, thus changes over time could not be interpreted. Patients rated reduced symptoms as an important outcome of treatment; however, this was not assessed in the clinical trial. According to the clinical expert consulted for this review, patients were cancer-free, and thus free from cancer symptoms at enrolment in the clinical trial post-surgery. Nivolumab may delay cancer recurrence and thus delay recurrence of cancer symptoms, but it would not influence post-surgery symptoms.
Harms
A total of 347 (98.9%) of patients in the nivolumab group and 332 (95.4%) of patients in the placebo group experienced at least 1 AE, and grade 3 and 4 AEs occurred in a total of 148 (42.2%) patients in the nivolumab group and 122 (35.1%) patients in the placebo group. The proportion of patients discontinuing study treatment due to toxicity was higher in the nivolumab group (64 [18.2%]) than in the placebo group (32 [9.2%]). SAEs were similar in patients treated with nivolumab versus placebo (29.6% versus 30.2%). IMAEs were more frequently reported in patients in the nivolumab group than in the placebo arm, and include rash (11.4% versus 2.3%), pneumonitis (4.8% versus 0.6%), and diarrhea or colitis (4.0% versus 0.9%). There were 95 (27.1%) deaths in the nivolumab group and 107 (30.7%) deaths in the placebo group, most commonly due to disease progression (73 [20.8%] in the nivolumab group and 90 [25.9%] in the placebo arm). There were 3 treatment-related deaths in the nivolumab group, 2 due to pneumonitis and 1 due to bowel perforation. No new safety signals were observed with nivolumab, and the clinical experts anticipated that overall nivolumab would have a more favourable safety profile than adjuvant chemotherapy.
Conclusions
One sponsor-submitted, ongoing, phase III, multinational, double-blind, randomized placebo-controlled trial provided evidence regarding the efficacy and safety of nivolumab compared with placebo in patients at high risk of recurrence after radical resection of MIUC (with primary site either in the bladder or upper urinary tract). Compared to placebo, adjuvant treatment with nivolumab (240 mg every 2 weeks IV infusion until disease recurrence or unacceptable toxicity for a total treatment duration of 1 year) showed a statistically significant DFS benefit in the treatment of patients (≥ 18 years old) with completely resected MIUC. The absolute difference in median DFS between treatment groups (approximately 10 months) was considered clinically meaningful by the clinical experts consulted by CADTH in patients at high risk of recurrence who are ineligible to receive adjuvant cisplatin-based chemotherapy. Results for OS were not available at the time of this review. While some evidence suggests individual-level associations between DFS and OS, trial-level associations between DFS and OS have not been assessed in the target population. Therefore, it cannot be firmly concluded to what extent the improvements in DFS observed in patients in the nivolumab group of the CheckMate 274 trial would translate into OS benefits. HRQoL analyses were descriptive only and limited by high rates of missing data, thus changes over time could not be interpreted. Data on adjuvant nivolumab compared to adjuvant cisplatin-based chemotherapy in patients at high risk of recurrence who are eligible to received cisplatin-based chemotherapy were not available from the CheckMate 274 trial. ITCs of nivolumab with cisplatin-based chemotherapy favoured chemotherapy, but the methodological concerns identified and the observed heterogeneity across study designs and populations precluded drawing definitive conclusions about the comparative effectiveness of adjuvant nivolumab versus adjuvant chemotherapy. Given the lack of robust comparative data between adjuvant nivolumab and adjuvant chemotherapy, the clinical experts consulted by CADTH were unsure if adjuvant nivolumab addressed an unmet need in patients who are at high risk of recurrence and eligible for adjuvant chemotherapy.
The safety profile of nivolumab in this study was consistent with the known safety profile of nivolumab, and no additional safety signals were identified with adjuvant nivolumab therapy in this study.
References
1.Canadian Cancer Society. Bladder cancer. 2021; https://cancer.ca/en/cancer-information/cancer-types/bladder. Accessed 2021 Oct 14.
2.American Cancer Society. Cancer facts & figures 2021. Atlanta (GA): American Cancer Society; 2021: https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2021/cancer-facts-and-figures-2021.pdf?bcs-agent-scanner=6d745fe2-1baa-7a44-a2b6-2f677a8a646c. Accessed 2021 Oct 14.
3.Cancer Treatment Centers of America. Bladder cancer types. 2021; https://www.cancercenter.com/cancer-types/bladder-cancer/types. Accessed 2021 Oct 14.
4.Alberta Health Services. Muscle-Invasive Bladder Cancer 2021: https://www.albertahealthservices.ca/assets/info/hp/cancer/if-hp-cancer-guide-gu014-ambc.pdf. Accessed 2022 Apr 25.
5.Cancer stat facts: bladder cancer. Bethesda (MD): National Cancer Institute; 2021: https://seer.cancer.gov/statfacts/html/urinb.html. Accessed 2021 Oct 14.
6.Kulkarni GS, Black PC, Sridhar SS, et al. Canadian Urological Association guideline: Muscle-invasive bladder cancer. Canadian Urological Association journal. 2019;13(8):230-238. PubMed
7.Lerner S. Overview of the initial approach and management of urothelial bladder cancer. In: T.W. P, ed. UpToDate. Waltham (MA): UpToDate; 2022: https://www.uptodate.com/contents/overview-of-the-initial-approach-and-management-of-urothelial-bladder-cancer#! Accessed 2022 Apr 25.
8.Flaig TW, Spiess PE, Agarwal N, et al. Bladder Cancer, Version 3.2020, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2020;18(3):329-354. PubMed
9.Clinical Study Report: CA209274. A Phase 3 Randomized, Double-blind, Multi-center Study of Adjuvant Nivolumab versus Placebo in Subjects with High Risk Invasive Urothelial Carcinoma (CheckMate 274: CHECKpoint pathway and nivoluMAb clinical Trial Evaluation 274) [internal sponsor's report]. Princeton (NJ): Bristol-Myers Squibb; 2020 Dec 4.
10.Opdivo nivolumab for injection Intravenous Infusion, 10 mg nivolumab /mL 40 mg and 100 mg single-use vials [product monograph]. Montreal (QC): Bristol-Myers Squibb Canada; 2022 Jun 27.
11.Erratum to Primary Clinical Study Report for Study CA209274 A Phase 3 Randomized, Double-blind, Multi-center Study of Adjuvant Nivolumab versus Placebo in Subjects with High Risk Invasive Urothelial Carcinoma. Princeton, NJ: Bristol-Myers Squibb; 2021 May 5.
12.BMS response to April 20th 2022 DRR request for additional information regarding Opdivo DRR review [internal additional sponsor's information]. Bristol Myers Squibb; 2022.
13.Galsky M, Witjes JA, Gschwend JE, et al. Disease-free survival with longer follow-up from the phase 3 CheckMate 274 trial of adjuvant nivolumab in patients who underwent surgery for high-risk muscle-invasive urothelial carcinoma [conference abstract]. J Urol. 2022;207(5S).
14.Galsky MD, Hahn NM, Rosenberg J, et al. A consensus definition of patients with metastatic urothelial carcinoma who are unfit for cisplatin-based chemotherapy. The lancet oncology. 2011;12(3):211-214. PubMed
15.Sternberg CN, Skoneczna I, Kerst JM, et al. Immediate versus deferred chemotherapy after radical cystectomy in patients with pT3–pT4 or N+ M0 urothelial carcinoma of the bladder (EORTC 30994): an intergroup, open-label, randomised phase 3 trial. The lancet oncology. 2015;16(1):76-86. PubMed
16.Indirect treatment comparisons for Nivolumab in the adjuvant setting of Muscle Invasive Urothelial Carcinoma [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Opdivo (nivolumab). Saint-Laurent (QC): Bristol-Myers Squibb; 2022 Feb 22.
17.Bajorin DF, Witjes JA, Gschwend JE, et al. Adjuvant Nivolumab versus Placebo in Muscle-Invasive Urothelial Carcinoma. New England Journal of Medicine. 2021;384(22):2102-2114. PubMed
18.Daneshmand S. Epidemiology and risk factors of urothelial (transitional cell) carcinoma of the bladder. In: Post TW, ed. UpToDate. Waltham (MA): UpToDate; 2021: www.uptodate.com. Accessed 2021 Jul 13.
19.Malats N, Real FX. Epidemiology of bladder cancer. Hematol Oncol Clin North Am. 2015;29(2):177-189, vii. PubMed
20.Drug Reimbursement Review sponsor submission: Opdivo (nivolumab) [internal sponsor's package]. Saint-Laurent (QC): Bristol-Myers Squibb; 2022 Feb 22.
21.Powles T, Bellmunt J, Comperat E, et al. Bladder cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Annals of Oncology. 2022;33(3):244-258. PubMed
22.Birtle A, Johnson M, Chester J, et al. Adjuvant chemotherapy in upper tract urothelial carcinoma (the POUT trial): a phase 3, open-label, randomised controlled trial. The Lancet. 2020;395(10232):1268-1277. PubMed
23.Dason S, Cha EK, Falavolti C, et al. Late Recurrences Following Radical Cystectomy Have Distinct Prognostic and Management Considerations. The Journal of urology. 2020;204(3):460-465. PubMed
24.Mitra AP, Quinn DI, Dorff TB, et al. Factors influencing post-recurrence survival in bladder cancer following radical cystectomy. BJU Int. 2012;109(6):846-854. PubMed
25.Kim HS, Jeong CW, Kwak C, Kim HH, Ku JH. Disease-free survival at 2 and 3 years is a significant early surrogate marker predicting the 5-year overall survival in patients treated with radical cystectomy for urothelial carcinoma of the bladder: external evaluation and validation in a cohort of Korean patients. Frontiers in Oncology. 2015;5:246. PubMed
26.Koh DM, Husband JE. Patterns of recurrence of bladder carcinoma following radical cystectomy. Cancer Imaging. 2003;3(2):96-100.
27.Slaton JW, Swanson DA, Grossman HB, Dinney CP. A stage specific approach to tumor surveillance after radical cystectomy for transitional cell carcinoma of the bladder. J Urol. 1999;162(3 Pt 1):710-714. PubMed
28.Health Canada. Notice of Compliance for nivolumab (OPDIVO). 2022 Jun 27: https://health-products.canada.ca/noc-ac/info.do?lang=en&no=28464. Accessed 2022 Jul 25.
29.McGowan J, Sampson M, Salzwedel DM, Cogo E, Foerster V, Lefebvre C. PRESS Peer Review of Electronic Search Strategies: 2015 guideline statement. Journal of clinical epidemiology. 2016;75:40-46. PubMed
30.Grey matters: a practical tool for searching health-related grey literature. Ottawa (ON): CADTH; 2019: https://www.cadth.ca/grey-matters. Accessed 2022 Jun 17.
31.Witjes JA, Galsky MD, Gschwend JE, et al. Health-related Quality of Life with Adjuvant Nivolumab After Radical Resection for High-risk Muscle-invasive Urothelial Carcinoma: Results from the Phase 3 CheckMate 274 Trial. European Urology Oncology. 2022;11:11. PubMed
32.Galsky MD, Witjes JA, Gschwend JE, et al. Impact of recurrence on health-related quality of life in patients at high risk of recurrence after radical surgery for muscle-invasive urothelial carcinoma: results from the phase 3 CheckMate 274 trial. American Society of Clinical Oncology (ASCO) Annual Meeting; June 4-8, 2021.
33.Gschwend JE, Bajorin D, Galsky MD, et al. Health-related quality of life with adjuvant nivolumab versus placebo after radical resection for high -risk muscle -invasive urothelial carcinoma: results from the phase 3 CheckMate 274 trial. the European Association of Urology (EAU) Virtual Congress; July 8-12, 2021.
34.Aaronson NK, Ahmedzai S, Bergman B, et al. The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst. 1993;85(5):365-376. PubMed
35.Fayers PM. Interpreting quality of life data: population-based reference data for the EORTC QLQ-C30. Eur J Cancer. 2001;37(11):1331-1334. PubMed
36.Brooks R. EuroQol: the current state of play. Health Policy. 1996;37(1):53-72. PubMed
37.EuroQol Group. EuroQol--a new facility for the measurement of health-related quality of life. Health Policy. 1990;16(3):199-208. PubMed
38.Osoba D, Aaronson NK, Muller M, et al. Effect of neurological dysfunction on health-related quality of life in patients with high-grade glioma. J Neurooncol. 1997;34(3):263-278. PubMed
39.Bajorin DF, Witjes JA, Gschwend JE, et al. Erratum: Adjuvant Nivolumab versus Placebo in Muscle-Invasive Urothelial Carcinoma (N Engl J Med 2021 Jun3; 384(22):2102-2114) New England Journal of Medicine. 2021;385(9):864. PubMed
40.Sharma P, Retz M, Siefker-Radtke A, et al. Nivolumab in metastatic urothelial carcinoma after platinum therapy (CheckMate 275): a multicentre, single-arm, phase 2 trial. The lancet oncology. 2017;18(3):312-322. PubMed
41.U.S. Food and Drug Administration. Clinical Trial Endpoints for the Approval of Cancer Drugs and Biologics Guidance for Industry. Food and Drug Administration; 2018: https://www.fda.gov/media/71195/download. Accessed 2022 Jun 17.
42.European Medicines Agency. Guideline on the clinical evaluation of anticancer medicinal products 2019: https://www.ema.europa.eu/en/documents/scientific-guideline/draft-guideline-evaluation-anticancer-medicinal-products-man-revision-6_en.pdf. Accessed 2022 Jun 17.
43.Laukhtina E, Sari Motlagh R, Mori K, et al. Chemotherapy is superior to checkpoint inhibitors after radical surgery for urothelial carcinoma: a systematic review and network meta-analysis of oncologic and toxicity outcomes. Crit Rev Oncol Hematol. 2022;169:103570. PubMed
44.Centre for Reviews and Dissemination. Systematic Reviews. CRD's guidance for for undertaking reviews in health care. 2009.
45.Haute Autorite de Sante HAS). Evaluating Health Technology - Methodological Guidance: Choices in methods for economic evaluation - HAS. Validated by the CEESP on 6 April 2020 ed2020.
46.Agence Nationale d'Accreditation et d'Evaluation en Sante (ANAES). Guide d'analyse de la literature et gradation des recommandations. 2000.
47.Dias S, Welton NJ, Sutton AJ, Ades A. NICE DSU technical support document 2: a generalised linear modelling framework for pairwise and network meta-analysis of randomised controlled trials. 2011: https://www.ncbi.nlm.nih.gov/books/NBK310366/pdf/Bookshelf_NBK310366.pdf. Accessed 2022 Jun 17.
48.Cognetti F, Ruggeri E, Felici A, et al. Adjuvant chemotherapy with cisplatin and gemcitabine versus chemotherapy at relapse in patients with muscle-invasive bladder cancer submitted to radical cystectomy: an Italian, multicenter, randomized phase III trial. Annals of oncology. 2012;23(3):695-700. PubMed
49.Lehmann J, Franzaring L, Thüroff J, Wellek S, Stöckle M. Complete long-term survival data from a trial of adjuvant chemotherapy vs control after radical cystectomy for locally advanced bladder cancer. BJU International. 2006;97(1):42-47. PubMed
50.Zhegalik AG, Polyakov SL, Rolevich AI, et al. Long-term results of a single-center prospective randomized trial assessing efficacy of a shortened course of adjuvant chemotherapy after radical cystectomy in patients with locally advanced bladder cancer. Central European Journal of Urology. 2020;73(1):26. PubMed
51.Bellmunt J. Treatment of metastatic urothelial cancer of the bladder and urinary tract. In: Post TW, ed. UpToDate. Waltham (MA): UpToDate; 2021: www.uptodate.com. Accessed 2021 Jul 13.
52.Freiha F, Reese J, Torti FM. A Randomized Trial of Radical Cystectomy Versus Radical Cystectomy Plus Cisplatin, Vinblastine and Methotrexate Chemotherapy for Muscle Invasive Bladder Cancer. The Journal of Urology. 1996;155(2):495-500. PubMed
53.Skinner Donald G, Daniels John R, Russell Christy A, et al. The Role of Adjuvant Chemotherapy Following Cystectomy for Invasive Bladder Cancer: A Prospective Comparative Trial. J Urol. 1991;145(3):459-464. PubMed
54.Studer Urs E, Bacchi M, Biedermann C, et al. Adjuvant Cisplatin Chemotherapy Following Cystectomy for Bladder Cancer: Results of a Prospective Randomized Trial. J Urol. 1994;152(1):81-84. PubMed
55.Luo Y, Feng B, Wei D, et al. Adjuvant chemotherapy after radical nephroureterectomy improves the survival outcome of high-risk upper tract urothelial carcinoma patients with cardiovascular comorbidity. Scientific Reports. 2020;10(1):17674. PubMed
56.Alevizakos M, Bellmunt J. Adjuvant immunotherapy for muscle-invasive urothelial carcinoma of the bladder. Expert Review of Anticancer Therapy. 2022:1-9. PubMed
57.Fenner A. Adjuvant nivolumab improves survival after radical cystectomy. Nature Reviews Urology. 2021;18(8):444. PubMed
58.Galsky MD, Saci A, Szabo PM, et al. Nivolumab in Patients with Advanced Platinum-resistant Urothelial Carcinoma: Efficacy, Safety, and Biomarker Analyses with Extended Follow-up from CheckMate 275. Clinical Cancer Research. 2020;26(19):5120-5128. PubMed
59.Hakenberg OW. Nivolumab for the treatment of bladder cancer. Expert Opinion on Biological Therapy. 2017;17(10):1309-1315. PubMed
60.Hayakawa N, Kikuchi E. Editorial Comment to Effect of optimal neoadjuvant chemotherapy on oncological outcomes of locally advanced bladder cancer with laparoscopic radical cystectomy: A matched-pair analysis in a multicenter cohort. International Journal of Urology. 2021;28(6):664. PubMed
61.Kumar N. Checkmate 274 trial: Is Nivolumab the new standard in adjuvant setting for high-risk muscle invasive urothelial carcinoma? Indian Journal of Urology. 2021;37(4):369-371. PubMed
62.Mancini M, Righetto M, Noessner E. Checkpoint Inhibition in Bladder Cancer: Clinical Expectations, Current Evidence, and Proposal of Future Strategies Based on a Tumor-Specific Immunobiological Approach. Cancers. 2021;13(23):29. PubMed
63.Petrelli F, Perego G, Vavassori I, Luciani A. Neoadjuvant or adjuvant immunotherapy in bladder cancer: biological opportunity or clinical utility? Tumori. 2021:3008916211061604. PubMed
64.Swami U, Haaland B, Kessel A, et al. Comparative Effectiveness of Immune Checkpoint Inhibitors in Patients with Platinum Refractory Advanced Urothelial Carcinoma. J Urol. 2021;205(3):709-717. PubMed
65.Teo MY, Rosenberg JE. Nivolumab for the treatment of urothelial cancers. Expert Review of Anticancer Therapy. 2018;18(3):215-221. PubMed
66.Doherty MK, Leung Y, Su J, et al. Health utility scores from EQ-5D and health-related quality of life in patients with esophageal cancer: a real-world cross-sectional study. Dis Esophagus. 2018;31(12). PubMed
67.Pickard AS, Neary MP, Cella D. Estimation of minimally important differences in EQ-5D utility and VAS scores in cancer. Health Qual Life Outcomes. 2007;5:70. PubMed
68.Osoba D, Aaronson N, Zee B, Sprangers M, te Velde A. Modification of the EORTC QLQ-C30 (version 2.0) based on content validity and reliability testing in large samples of patients with cancer. The Study Group on Quality of Life of the EORTC and the Symptom Control and Quality of Life Committees of the NCI of Canada Clinical Trials Group. Qual Life Res. 1997;6(2):103-108. PubMed
69.Bjordal K, de Graeff A, Fayers PM, et al. A 12 country field study of the EORTC QLQ-C30 (version 3.0) and the head and neck cancer specific module (EORTC QLQ-H&N35) in head and neck patients. EORTC Quality of Life Group. Eur J Cancer. 2000;36(14):1796-1807. PubMed
70.Westhofen T, Eismann L, Buchner A, et al. Baseline Health-related Quality of Life Predicts Bladder Cancer-specific Survival Following Radical Cystectomy. European Urology Focus. 2022;17:17. PubMed
71.Zahran MH, El-Hefnawy AS, Zidan EM, El-Bilsha MA, Taha DE, Ali-El-Dein B. Health-related quality of life after radical cystectomy and neobladder reconstruction in women: impact of voiding and continence status. International Journal of Urology. 2014;21(9):887-892. PubMed
72.Zhang Y, Wang Y, Song B, Li H. Patients' self-report anxiety, depression and quality of life and their predictive factors in muscle invasive bladder cancer patients receiving adjuvant chemotherapy. Psychol Health Med. 2020;25(2):190-200. PubMed
73.Anderson CB, Rapkin B, Reaves BC, et al. Idiographic quality of life assessment before radical cystectomy. Psychooncology. 2017;26(2):206-213. PubMed
74.Desgrandchamps F, Leroux S, Ravery V, et al. Subcutaneous pyelovesical bypass as replacement for standard percutaneous nephrostomy for palliative urinary diversion: prospective evaluation of patient's quality of life. J Endourol. 2007;21(2):173-176. PubMed
75.Kostakopoulos N, Protogerou V, Skolarikos A, et al. VIP neobladder (Padovana) reconstruction following radical cystectomy for bladder cancer Complications, functional outcome and quality of life evaluation in 95 cases. Ann Ital Chir. 2015;86(4):362-367. PubMed
76.Licht T, Nickels A, Rumpold G, Holzner B, Riedl D. Evaluation by electronic patient-reported outcomes of cancer survivors' needs and the efficacy of inpatient cancer rehabilitation in different tumor entities. Support Care Cancer. 2021;29(10):5853-5864. PubMed
77.Mansson A, Henningsohn L, Steineck G, Mansson W. Neutral third party versus treating institution for evaluating quality of life after radical cystectomy. Eur Urol. 2004;46(2):195-199. PubMed
78.Morganstern BA, Bochner B, Dalbagni G, Shabsigh A, Rapkin B. The psychological context of quality of life: a psychometric analysis of a novel idiographic measure of bladder cancer patients' personal goals and concerns prior to surgery. Health Qual Life Outcomes. 2011;9:10. PubMed
79.Pazooki D, Edlund C, Dahlstrand C, Lindholm E, Tornqvist H, Jonsson O. Continent cutaneous urinary diversion is still a valid alternative after cystectomy for bladder carcinoma. Scand J Urol Nephrol. 2005;39(6):468-473. PubMed
80.Shi H, Yu H, Bellmunt J, et al. Comparison of health-related quality of life (HRQoL) between ileal conduit diversion and orthotopic neobladder based on validated questionnaires: a systematic review and meta-analysis. Qual Life Res. 2018;27(11):2759-2775. PubMed
81.Terret C, Perol D, Albrand G, Droz JP. Quality of life in geriatric oncology--An evaluation of standard questionnaires in elderly men with urological malignancies. Crit Rev Oncol Hematol. 2011;77(3):201-209. PubMed
82.Volz Y, Grimm T, Ormanns S, et al. Radical cystectomy for locally advanced urothelial carcinoma of the urinary bladder: Health-related quality of life, oncological outcomes and predictors for survival. Urol. 2021;39(5):299.e215-299.e221. PubMed
83.Osoba D, Burchmore M. Health-related quality of life in women with metastatic breast cancer treated with trastuzumab (Herceptin). Semin Oncol. 1999;26(4 Suppl 12):84-88. PubMed
84.Snyder CF, Blackford AL, Sussman J, et al. Identifying changes in scores on the EORTC-QLQ-C30 representing a change in patients' supportive care needs. Qual Life Res. 2015;24(5):1207-1216. PubMed
85.Teckle P, Peacock S, McTaggart-Cowan H, et al. The ability of cancer-specific and generic preference-based instruments to discriminate across clinical and self-reported measures of cancer severities. Health Qual Life Outcomes. 2011;9:106. PubMed
86.U.S. Food and Drug Administration. Table of Surrogate Endpoints That Were the Basis of Drug Approval or Licensure. 2022; https://www.fda.gov/drugs/development-resources/table-surrogate-endpoints-were-basis-drug-approval-or-licensure, 2022 May 4.
87.U.S. Food and Drug Administration. FDA Facts: Biomarkers and Surrogate Endpoints. 2017; https://www.fda.gov/about-fda/innovation-fda/fda-facts-biomarkers-and-surrogate-endpoints, 2022 May 4.
88.U.S. Food and Drug Administration. Bladder Cancer: Developing Drugs and Biologics for Adjuvant Treatment. Guidance Document. 2020: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/bladder-cancer-developing-drugs-and-biologics-adjuvant-treatment. Accessed 2022 Jun 17.
89.Executive summary of rapid report A10-05: Validity of surrogate endpoints in oncology. Cologne (DE): Institute for Quality and Efficiency in Health Care; 2011: https://www.iqwig.de/download/a10-05_executive_summary_v1-1_surrogate_endpoints_in_oncology.pdf. Accessed 2022 Jun 17.
90.Gyawali B, Hey SP, Kesselheim AS. Evaluating the evidence behind the surrogate measures included in the FDA's table of surrogate endpoints as supporting approval of cancer drugs. EClinicalMedicine. 2020;21:100332. PubMed
91.Haslam A, Hey SP, Gill J, Prasad V. A systematic review of trial-level meta-analyses measuring the strength of association between surrogate end-points and overall survival in oncology. Eur J Cancer. 2019;106:196-211. PubMed
92.Buyse M. Statistical validation of surrogate outcome measures. Trials. 2011;12(Suppl 1):A93-A93.
93.Buyse M, Molenberghs G, Burzykowski T, Renard D, Geys H. The validation of surrogate endpoints in meta-analyses of randomized experiments. Biostatistics. 2000;1(1):49-67. PubMed
94.Sonpavde G, Khan MM, Lerner SP, et al. Disease-free survival at 2 or 3 years correlates with 5-year overall survival of patients undergoing radical cystectomy for muscle invasive bladder cancer. The Journal of urology. 2011;185(2):456-461. PubMed
95.Fajkovic H, Cha EK, Xylinas E, et al. Disease-free survival as a surrogate for overall survival in upper tract urothelial carcinoma. World J Urol. 2013;31(1):5-11. PubMed
96.Nuhn P, May M, Fritsche HM, et al. External validation of disease-free survival at 2 or 3 years as a surrogate and new primary endpoint for patients undergoing radical cystectomy for urothelial carcinoma of the bladder. Eur J Surg Oncol. 2012;38(7):637-642. PubMed
Appendix 1: Literature Search Strategy
Note that this appendix has not been copy-edited.
Clinical Literature Search
Overview
Interface: Ovid
Databases:
MEDLINE All (1946-present)
Embase (1974-present)
Note: Subject headings and search fields have been customized for each database. Duplicates between databases were removed in Ovid.
Date of search: March 22, 2022
Alerts: Bi-weekly search updates until project completion
Search filters applied: randomized controlled trials; controlled clinical trials
Limits:
Conference abstracts: excluded
Syntax | Description |
|---|---|
/ | At the end of a phrase, searches the phrase as a subject heading |
MeSH | Medical Subject Heading |
.fs | Floating subheading |
exp | Explode a subject heading |
* | Before a word, indicates that the marked subject heading is a primary topic; or, after a word, a truncation symbol (wildcard) to retrieve plurals or varying endings |
# | Truncation symbol for one character |
? | Truncation symbol for one or no characters only |
adj# | Requires terms to be adjacent to each other within # number of words (in any order) |
.ti | Title |
.ot | Original title |
.ab | Abstract |
.hw | Heading word; usually includes subject headings and controlled vocabulary |
.kf | Keyword heading word |
.dq | Candidate term word (Embase) |
.pt | Publication type |
.mp | Mapped term |
.rn | Registry number |
.nm | Name of substance word (MEDLINE) |
.yr | Publication year |
.jw | Journal title word (MEDLINE) |
.jx | Journal title word (Embase) |
freq=# | Requires terms to occur # number of times in the specified fields |
medall | Ovid database code: MEDLINE All, 1946 to present, updated daily |
oemezd | Ovid database code; Embase, 1974 to present, updated daily |
Multi-Database Strategy
# Searches
Nivolumab/
(opdivo* or nivolumab* or nivo or bms 936558 or bms936558 or cmab 819 or cmab819 or mdx 1106 or mdx1106 or ono 4538 or ono4538 or HSDB 8256 or HSDB8256 or GTPL 7335 or GTPL7335 or 31YO63LBSN).ti,ab,kf,ot,hw,rn,nm.
1 or 2
Carcinoma, Transitional Cell/ or Urinary bladder neoplasms/ or Ureteral neoplasms/ or Urethral neoplasms/ or Urologic Neoplasms/
((urologic* or urothel* or urinary tract or bladder or uretra* or urethra* or ureter* or (transitional adj3 cell*) or transitional epithel* or renal pelvis or uroepitheli* or uro-epitheli* or urogenital* or uro-genital* or vesical* or uretal*) and (tumor* or tumour* or cancer* or carcinoma* or malignan* or metasta* or adenocarcinoma* or adeno-carcinoma* or neoplas*)).ti,ab,kf.
4 or 5
3 and 6
7 use medall
*nivolumab/
(opdivo* or nivolumab* or nivo or bms 936558 or bms936558 or cmab 819 or cmab819 or mdx 1106 or mdx1106 or ono 4538 or ono4538 or HSDB 8256 or HSDB8256 or GTPL 7335 or GTPL7335).ti,ab,kf,dq.
9 or 10
Transitional Cell Carcinoma/ or Urinary tract carcinoma/ or exp bladder cancer/ or exp ureter cancer/ or exp urethra cancer/ or urinary tract cancer/
((urologic* or urothel* or urinary tract or bladder or uretra* or urethra* or ureter* or (transitional adj3 cell*) or transitional epithel* or renal pelvis or uroepitheli* or uro-epitheli* or urogenital* or uro-genital* or vesical* or uretal*) and (tumor* or tumour* or cancer* or carcinoma* or malignan* or metasta* or adenocarcinoma* or adeno-carcinoma* or neoplas*)).ti,ab,kf,dq.
12 or 13
11 and 14
15 not (conference abstract or conference review).pt.
16 use oemezd
8 or 17
(Randomized Controlled Trial or Controlled Clinical Trial or Pragmatic Clinical Trial or Equivalence Trial or Clinical Trial, Phase III).pt.
Randomized Controlled Trial/
exp Randomized Controlled Trials as Topic/
"Randomized Controlled Trial (topic)"/
Controlled Clinical Trial/
exp Controlled Clinical Trials as Topic/
"Controlled Clinical Trial (topic)"/
Randomization/
Random Allocation/
Double-Blind Method/
Double Blind Procedure/
Double-Blind Studies/
Single-Blind Method/
Single Blind Procedure/
Single-Blind Studies/
Placebos/
Placebo/
Control Groups/
Control Group/
(random* or sham or placebo*).ti,ab,hw,kf.
((singl* or doubl*) adj (blind* or dumm* or mask*)).ti,ab,hw,kf.
((tripl* or trebl*) adj (blind* or dumm* or mask*)).ti,ab,hw,kf.
(control* adj3 (study or studies or trial* or group*)).ti,ab,kf.
(Nonrandom* or non random* or non-random* or quasi-random* or quasirandom*).ti,ab,hw,kf.
allocated.ti,ab,hw.
((open label or open-label) adj5 (study or studies or trial*)).ti,ab,hw,kf.
((equivalence or superiority or non-inferiority or noninferiority) adj3 (study or studies or trial*)).ti,ab,hw,kf.
(pragmatic study or pragmatic studies).ti,ab,hw,kf.
((pragmatic or practical) adj3 trial*).ti,ab,hw,kf.
((quasiexperimental or quasi-experimental) adj3 (study or studies or trial*)).ti,ab,hw,kf.
(phase adj3 (III or "3") adj3 (study or studies or trial*)).ti,hw,kf.
or/19-49
18 and 50
remove duplicates from 51
Clinical Trials Registries
ClinicalTrials.gov
Produced by the US National Library of Medicine. Targeted search used to capture registered clinical trials.
[Search -- (opdivo OR nivolumab) AND (urologic OR urothelial OR urinary OR bladder OR uretra OR urethra OR ureter OR transitional)]
WHO ICTRP
International Clinical Trials Registry Platform, produced by the World Health Organization. Targeted search used to capture registered clinical trials.
[Search terms -- (opdivo OR nivolumab) AND (urologic OR urothelial OR urinary OR bladder OR uretra OR urethra OR ureter OR transitional)]
Health Canada’s Clinical Trials Database
Produced by Health Canada. Targeted search used to capture registered clinical trials.
[Search terms -- (opdivo OR nivolumab) AND (urologic OR urothelial OR urinary OR bladder OR uretra OR urethra OR ureter OR transitional)]
EU Clinical Trials Register
European Union Clinical Trials Register, produced by the European Union. Targeted search used to capture registered clinical trials.
[Search terms -- (opdivo OR nivolumab) AND (urologic OR urothelial OR urinary OR bladder OR uretra OR urethra OR ureter OR transitional)]
Grey Literature
Search dates: March 9 – March 11, 2022
Keywords: (opdivo OR nivolumab) AND (urologic OR urothelial OR urinary OR bladder OR uretra OR urethra OR ureter OR transitional OR renal pelvis or uroepithelial or uro-epithelial or urogenital or uro-genital or vesical or uretal)
Limits: None
Updated: Search updated prior to the completion of stakeholder feedback period
Relevant websites from the following sections of the CADTH grey literature checklist Grey Matters: A Practical Tool for Searching Health-Related Grey Literature were searched:
Health Technology Assessment Agencies
Health Economics
Clinical Practice Guidelines
Drug and Device Regulatory Approvals
Advisories and Warnings
Drug Class Reviews
Clinical Trials Registries
Databases (free)
Internet Search
Appendix 2: Excluded Studies
Note that this appendix has not been copy-edited.
Reference | Reason for exclusion |
|---|---|
Alevizakos, M. and J. Bellmunt (2022). "Adjuvant immunotherapy for muscle-invasive urothelial carcinoma of the bladder." Expert Review of Anticancer Therapy: 1-9.56 | Review article |
Bajorin, D. F., et al. (2021). "Adjuvant Nivolumab versus Placebo in Muscle-Invasive Urothelial Carcinoma." New England Journal of Medicine 384(22): 2102-2114. 17 | Duplicate (provided by sponsor) |
Fenner, A. (2021). "Adjuvant nivolumab improves survival after radical cystectomy." Nature Reviews Urology 18(8): 444.57 | Research brief |
Galsky, M. D., et al. (2020). "Nivolumab in Patients with Advanced Platinum-resistant Urothelial Carcinoma: Efficacy, Safety, and Biomarker Analyses with Extended Follow-up from CheckMate 275." Clinical Cancer Research 26(19): 5120-5128.58 | Study population (metastatic UC) |
Hakenberg, O. W. (2017). "Nivolumab for the treatment of bladder cancer." Expert Opinion on Biological Therapy 17(10): 1309-1315.59 | Study population (metastatic UC) |
Hayakawa, N. and E. Kikuchi (2021). "Editorial Comment to Effect of optimal neoadjuvant chemotherapy on oncological outcomes of locally advanced bladder cancer with laparoscopic radical cystectomy: A matched-pair analysis in a multicenter cohort." International Journal of Urology 28(6): 664.60 | Intervention (neoadjuvant)/Editorial |
Kumar, N. (2021). "Checkmate 274 trial: Is Nivolumab the new standard in adjuvant setting for high-risk muscle-invasive urothelial carcinoma?" Indian Journal of Urology 37(4): 369-371.61 | Editorial |
Laukhtina, E., et al. (2022). "Chemotherapy is superior to checkpoint inhibitors after radical surgery for urothelial carcinoma: a systematic review and network meta-analysis of oncologic and toxicity outcomes." Critical Reviews in Oncology-Hematology 169: 103570.43 | Systematic Review |
Mancini, M., et al. (2021). "Checkpoint Inhibition in Bladder Cancer: Clinical Expectations, Current Evidence, and Proposal of Future Strategies Based on a Tumor-Specific Immunobiological Approach." Cancers 13(23): 29.62 | Study population (metastatic UC) |
Petrelli, F., et al. (2021). "Neoadjuvant or adjuvant immunotherapy in bladder cancer: biological opportunity or clinical utility?" Tumori: 3008916211061604.63 | Editorial |
Swami, U., et al. (2021). "Comparative Effectiveness of Immune Checkpoint Inhibitors in Patients with Platinum Refractory Advanced Urothelial Carcinoma." Journal of Urology 205(3): 709-717.64 | Study population (metastatic UC) |
Teo, M. Y. and J. E. Rosenberg (2018). "Nivolumab for the treatment of urothelial cancers." Expert Review of Anticancer Therapy 18(3): 215-221.65 | Review article |
UC =urothelial carcinoma
Appendix 3: Detailed Outcome Data
Note that this appendix has not been copy-edited.
PFS After the Next Line of Subsequent Systemic Anticancer Therapy (PFS2)
Results are presented in Table 27. As of the August 27, 2020, data cut-off date, in PFS2 analysis among all randomized patients, 245 (69.4%) patients in the nivolumab group and 231 (64.9%) patients in the placebo group were censored. Median PFS2 was 44.6 months in patients in the nivolumab group and 40.7 months in the placebo group (HR = 0.79 [95% CI, 0.61 to 1.02]).
Table 27: PFS2 Among All Randomized Patients and Patients With Tumour Cell PD-L1 Expression ≥ 1% in the CheckMate 274 Trial
Item | All randomized patients | Patients with tumour PD-L1 ≥ 1% | ||
|---|---|---|---|---|
Nivolumab N= 353 | Placebo N= 356 | Nivolumab N= 140 | Placebo N= 142 | |
Events, n (%) | 108 (30.6) | 125 (35.1) | 36 (25.7) | 54 (38.0) |
Median PFS2 (95% CI), monthsa | 44.0 (38.0, NA) | 40.7 (29.6, NA) | NA. (37.1, NA) | 39.4 (25.2, NA) |
HR (95% CI)b | 0.79 (0.61, 1.02) | 0.60 (0.39, 0.91) | ||
Rate at 6 months (95% CI), %a | 95.1 (92.2, 96.9) | 88.7 (84.8, 91.6) | 94.1 (88.5, 97.0) | 86.5 (79.4, 91.3) |
CI = confidence interval; PFS2 = progression-free survival after the next line of subsequent systemic anticancer therapy.
aBased on Kaplan-Meier Estimates
bStratified Cox proportional hazard model. Hazard ratio is nivolumab over placebo.
Source: CheckMate 274 Clinical Study Report9
Appendix 4: Description and Appraisal of Outcome Measures
Note that this appendix has not been copy-edited.
Aim
To describe the following outcome measures and review their measurement properties (validity, reliability, responsiveness to change, and MID):
EORTC QLQ-C30
EQ-5D-3L
Findings
Table 28: Summary of Outcome Measures and Their Measurement Properties
Outcome measure | Type | Conclusions about measurement properties | MID |
|---|---|---|---|
EORTC QLQ-C30 | 30-item, patient-reported, cancer-specific, quality of life questionnaire using 4- and 7-point Likert scales. | No evidence of validity, reliability, or responsiveness in patients with urothelial cancer | No MID identified in patients with urothelial cancer Patients with cancer38:
Patient with breast and colorectal cancer: |
EQ-5D-3L | A generic preference-based HRQoL instrument that has been applied to a wide range of health conditions and treatments. The EQ-5D-3L consists of 2 components: the descriptive system (from which the index score is generated) and the VAS. The index score consists of 5 domains (mobility, self-care, usual activities, pain/discomfort, and anxiety/depression) and ranks on a 3-point ordinal scale. A higher score indicates better HRQoL. The VAS tool assesses patient’s self-rated health on a vertical visual analogue scale. | No evidence of validity, reliability, or responsiveness of the EQ-5D-3L for patients with urothelial cancer was identified. The validation of EQ-5D-3L is available across countries around the world and in various conditions.36,37 Validity: Strong correlation was observed between EQ-5D-3L and FACT-E total score (R = 0.73), as well as FACT-E subclass domains to varying degrees. Also, moderately strong correlation with ECOG scale (Spearman r = -0.65) and EQ-5D-3L HUS.66 Responsiveness: EQ-5D-3L index score displayed a similar pattern as mean FACT-E and its subscale scores (P < 0.05 for time points studied). However, a ceiling effect was observed in the EQ-5D-3L index score.66 | Unknown for patients with urothelial cancer. Assessed among patients with the following cancers: stage III or IV cancers of the bladder, brain, breast, colon/rectum, head/neck, liver/pancreas, kidney, lung, lymphoma, ovary, and prostate (not including esophageal cancer)67 MID (UK): 0.10 to 0.12 based on ECOG PS range and 0.09 to 0.10 based on FACT-G score MID (US): 0.07 to 0.09 grouped by ECOG PS and 0.06 to 0.07 grouped by FACT-G score MIDs for VAS: 8 to 12 using ECOG PS and 7 to 10 using FACT-G quintiles The sponsor defined a MID as a mean change score from baseline of 0.08 for the EQ-5D utility score and of 7 for the EQ-5D VAS based on Pickard et al. (2007)9,67 |
ECOG PS = Eastern Cooperative Oncology Group performance status; EORTC QLQ-C30 = European Organization for Research and Treatment of Cancer Quality of Life Questionnaire Core 30; EQ-5D-3L = EuroQoL-5 Dimensions-3 levels; Five Dimensions; FACT-E = Functional Assessment of Cancer Therapy – Esophageal; FACT-G = Functional Assessment of Cancer Therapy – General; HRQoL = health-related quality of life; MID = minimal important difference; VAS = visual analogue scale.
EORTC QLQ-C30
Description and Scoring
The EORTC QLQ-C30 is one of the most used patient-reported outcome measures in oncology clinical trials. It is a multidimensional, cancer-specific, measure of HRQoL.34
The core questionnaire of the EORTC QLQ-C30 consists of 30 questions that are scored to create 5 multi-item functional scales (physical, role, cognitive, emotional, and social), 3 multi-item symptom scales (fatigue, pain, and nausea and vomiting), 6 single-item symptom scales (dyspnea, loss of appetite, insomnia, constipation and diarrhea), 2-item quality of life scale, and perceived financial impact of the disease.34
The EORTC QLQ-C30 uses a 1-week recall period to assess function and symptoms. All the scales and single-item measures range in score from 0 to 100. Most questions have 4 response options (“not at all,” “a little,” “quite a bit,” “very much”), with scores on these items ranging from one to 4. For the 2 items that form the global QoL scale, the response format is a 7-point Likert-type scale with anchors at 1 = “very poor” and 7 = “excellent.” Raw scores for each scale are computed as the average of the items that contribute to a particular scale. Scale sum scores are transformed so that a high score on the functional scales represents a high/healthy level of functioning, a high score on the symptom scales represents a high level of symptomatology, and a high score on the global health status/QoL represents a high QoL.35
According to the EORTC QLQ-C30 scoring algorithm, if there are missing items for a scale (i.e., the participant did not provide a response), the score for the scale can still be computed if there are responses for at least half of the items. It is assumed that the missing items have values equal to the average of those items for what the respondent completed.35
Assessment of Validity and Reliability
In its initial development, the EORTC QLQ-C30 underwent an evaluation of its psychometric properties and demonstrated reliability and validity in cancer patients in multicultural clinical research settings.34 A revision of the EORTC QLQ-C30 was undertaken to improve low internal consistency estimates and content validity for the role functioning scale and emphasis on physical functioning in the global QoL scale.68 The original and new versions were assessed in a total of 1,181 patients with cancer in Canada and the Netherlands. Internal consistency improved in role functioning scale in the new version (Cronbach alpha ranging from 0.78-0.88), and substitution of the new item for the previous did not alter internal consistency (Cronbach alpha ranging from 0.81-0.92).68
The EORTC QLQ-C30 (version 3.0) is the version currently in use, which differed from the previous version 2.0 in that the number of response options for the first 5 items of the questionnaire that comprise the Physical Function scale were increased from 2 response options (yes/no in version 2.0) to 4 (not at all, a little, quite a bit, very much). Internal consistency reliability, construct validity, criterion validity, and responsiveness of the EORTC QLQ-C30 Version 3.0 was assessed in 622 head and neck cancer patients from 12 countries which demonstrated that version 3.0 was more reliable than previous versions.69 Internal consistency of the multi-item scales was assessed using Cronbach alpha, with a value of 0.70 being considered adequate. The internal consistency of the new Physical Function scale of the EORTC QLQ-C30 Version 3.0 was 0.84, compared with 0.66 in version 1.0. The EORTC QLQ-C30 Version 3.0 was able to discriminate between head and neck cancer patients who were disease-free, who were newly diagnosed, and those with recurrent disease. As well, differences were noted between stages and according to Karnofsky PS, as the new scale had a stronger association with Karnofsky PS. Further, there was a high correlation observed between scores on the EORTC QLQ-C30 Version 3.0 and symptom/toxicity scores. Responsiveness to change was assessed using the standardized response mean (SRM), with an SRM of 0.20 being considered small, 0.50 being considered medium, and 0.80 being considered large. The changes in the scores of QLQ-C30 demonstrated a small to medium SRM in response to treatment over time with scores mostly deteriorating between 5 and 10 points.69 Though the EORTC QLQ-C30 has been used to assess HRQoL among bladder cancer/tumor patients including MIUC,70-82 it has not been specifically designed for this indication.
Evidence of validity, reliability, and responsiveness of the EORTC QLQ-C30 was not identified in the literature for patients with urothelial cancer.
Minimal Important Difference
One study by Osoba et al. (1999)83 conducted in patients with breast cancer and small cell lung cancer estimated a clinically relevant change in score on any scale of the EORTC QLQ-C30 to be 10 points. The estimate was based on a study that used an anchor-based approach to estimate the MID in which patients who reported “a little” change (for better or worse) on the subjective significance questionnaire had corresponding changes on a function or symptom scale of the EORTC QLQ-C30 of approximately 5 to 10 points. Participants who reported a “moderate” change had corresponding changes in the EORTC QLQ-C30 of about 10 to 20 points, and those who reported being “very much” changed had corresponding changes of more than 20 points.83
More recently in 2015, a Canadian study by Snyder et al. (2015)84 estimated the MIDs of EORTC QLQ-C30 scales using data from 193 newly diagnosed breast and colorectal cancer patients. The Supportive Care Needs Survey-Short Form-34 (SCNS-SF34) was used as an anchor; mean changes in EORTC QLQ-C30 scales associated with improvement, worsening, and no-change in supportive care based on the SCNS-SF34 was then calculated. MIDs were assessed for the following scales: Physical function, role function, emotional function, global health/QoL (i.e., GHS), pain, and fatigue. For improvement, MIDs associated with a statistically significantly improved supportive care needs ranged from 10 to 32 points. For worsening, MIDs associated with a statistically significantly worsening of supportive care needs ranged from nine to 21 points. The range for unchanged supportive care needs was from 1-point worsening to 16-point improvement in EORTC QLQ-C30 score. Based on this, the authors suggested a 10-point change in EORTC QLQ- C30 score represented changes in supportive care needs, and therefore should be considered for clinical use.
No MID in patients with UC has been identified.
EQ-5D-3L
Description and Scoring
The EQ-5D-3L is a generic preference-based HRQoL instrument that has been applied to a wide range of health conditions and treatments.36,37 The first of 2 parts of the EQ-5D-3L is a descriptive system that classifies respondents (aged ≥ 12 years) into one of 243 distinct health states. The descriptive system consists of the following 5 dimensions: mobility, self-care, usual activities, pain/discomfort, and anxiety/depression. Each dimension has 3 possible levels (1, 2, or 3) representing ‘no problems,’ ‘some problems,’ and ‘extreme problems,’ respectively. Respondents are asked to choose one level that reflects their own health state for each of the 5 dimensions. A scoring function can be used to assign a value (EQ- 5D-3L index score) to self-reported health states from a set of population-based preference weights.36,37 The second part is a vertical, calibrated 20 cm VAS (EQ-VAS) that has end points labelled 0 and 100, with respective anchors of ‘worst imaginable health state’ and ‘best imaginable health state,’ respectively. Respondents are asked to rate their own health by drawing a line from an anchor box to the point on the EQ-VAS which best represents their own health on that day. Hence, the EQ-5D-3L produces 3 types of data for each respondent:
A profile indicating the extent of problems on each of the 5 dimensions represented by a 5-digit descriptor, such as 11121, 33211
A population preference-weighted health index score based on the descriptive system
A self-reported current health status based on the EQ-VAS that is used to assess the overall health of the respondent rather than selected dimensions of individuals’ health
The EQ-5D-3L index score is generated by applying a multi-attribute utility function to the descriptive system. Different utility functions are available that reflect the preferences of specific populations (e.g., US or UK). The lowest possible overall score (corresponding to severe problems on all 5 attributes) varies depending on the utility function that is applied to the descriptive system (e.g., −0.59 for the UK algorithm and −0.109 for the US algorithm). Scores less than 0 represent health states that are valued by society as being worse than dead, while scores of 0 and 1.00 are assigned to the health states ‘dead’ and ‘perfect health,’ respectively. The sponsor’s submission reported that in the CheckMate 274 trial the utility index was computed using the scoring algorithm based on the UK trade-trade-off value set.9
Assessment of Validity and Reliability
Teckle et al. (2011) conducted a study of patients with cancer (N = 184) at the Vancouver Cancer Clinic.85 There was evidence of moderate concurrent validity between the EQ-5D-3L and EORTC QLQ-C30 as well as between the EQ-5D-3L and EQ-VAS (Pearson’s correlation coefficient = 0.43 for both) and strong concurrent validity between the EQ-VAS and EORTC QLQ-C30 (Pearson’s correlation coefficient = 0.73). The EQ-5D-3L demonstrated acceptable internal consistency for all 5 functioning scales along with global health status (Cronbach alpha ranged from 0.77 to 0.82).
Doherty et al. (2018)66 analyzed 119 patients with histologically confirmed esophageal cancer and gastroesophageal junction cancer (Siewert I/II) of all disease stages at ambulatory clinics in a cross-sectional survey study and a real-world setting. Bivariate Spearman correlation and multivariable linear regression analyses characterized associations between the various esophageal cancer subscales (Functional Assessment of Cancer Therapy – Esophageal [FACT-E]), scales, and the FACT-E derived symptom complexes with EQ-5D-3L derived index scores. A strong correlation was observed between EQ-5D-3L and FACT-E (R = 0.73): the strongest correlation observed with the physical well-being subscale, followed in descending order by Functional Well-Being, esophageal-specific symptoms, emotional well-being, and social well-being subscales. In addition, the association between FACT-E and EQ-5D-3L was maintained in a multivariable model (β of 0.0044, P < 0.001). Lastly, moderately strong correlation was also seen between a self-reported PS based on the ECOG scale (Spearman r = -0.65) and EQ-5D-3L index score.
No evidence of validity and reliability of the EQ-5D-3L for patients with UC was identified.
Responsiveness to Change
Doherty et al. (2018)66 analyzed the same sample in longitudinal follow-up surveys at baseline and predetermined subsequent visits, i.e., pre-treatment, during chemoradiotherapy, and post-treatment (within the first 6 months following definitive chemoradiotherapy or surgery), surveillance (more than 6 months from definitive treatment without relapse or progression), progression, and palliative chemotherapy. EQ-5D-3L index score displayed a similar pattern as mean FACT-E and subscale scores, which dropped from baseline through treatment and recovered during post-treatment surveillance (P < 0.001), but with smaller differences (P = 0.07). In addition, the ceiling effect was observed in the EQ-5D-3L index score. Briefly, among patients with stage II/III esophageal cancer, mean EQ-5D-3L index score varied across disease states (P < 0.001), along with FACT-E and subscales (P < 0.001). Among patients with advanced disease, there was no significant difference between baseline and on-treatment total scores of EQ-5D-3L but improved EC subscale scores were noted (P = 0.003). Overall, the observed differences in mean EQ-5D-3L index score across cancer stages and disease states suggests some sensitivity to change in clinical circumstances.
No evidence of responsiveness to change of the EQ-5D-3L for patients with UC was identified.
Minimal Important Difference
Pickard et al. (2007)67 conducted a retrospective analysis on cross-sectional data collected from 534 cancer patients with 11 types of cancer, including stage III or IV cancers of the bladder, brain, breast, colon/rectum, head/neck, liver/pancreas, kidney, lung, lymphoma, ovary, and prostate. A range of MIDs in EQ-5D index-based utility (UK and US) scores and VAS scores were estimated using both anchor-based (ECOG PS and Functional Assessment of Cancer Therapy – General (FACT-G) total score-based quintiles) and distribution-based (0.5 standard deviation and standard error of the measure) approaches. Important differences in EQ-5D utility and VAS scores were similar for all cancers (not including EC). For UK-utility scores, MID estimates based on ECOG PS ranged from 0.10 to 0.12 and from 0.09 to 0.10 using FACT-G quintiles for all cancers (not including esophageal cancer). For US-utility scores, MIDs ranged from 0.07 to 0.09 grouped by ECOG PS and when based on FACT-G quintiles, MIDs were 0.06 to 0.07 in all cancers (not including esophageal cancers). MIDs for VAS scores ranged from 8 to 12 (PS) and 7 to 10 (FACT-G quintiles).
No information on the MID of the EQ-5D-3L in patients with urothelial cancer was found.
Other Considerations and Limitations
One limitation of the EQ-5D-3L was a significant ceiling effect, and left skew of the data; for example, EQ-5D-3L may be more useful in a population with worse overall health status.66 Further, though the EQ-5D-3L may be an adequate tool to summarize HRQoL, responsiveness to change may still be better captured using a more disease-specific tool such as EORTC QLQ-BLM30.
Appendix 5: DFS as a Surrogate Outcome
Note that this appendix has not been copy-edited.
This section describes the DFS outcome measure and summarizes evidence that examines the validity of DFS as surrogate for OS in patients with MIUC. Studies discussed in this section were identified by reviewing bibliographies of key papers and through contacts with appropriate clinical experts consulted by CADTH. In addition, the sponsor was contacted for information regarding relevant studies.
In the CheckMate274 trial DFS was the primary outcome used to evaluate efficacy of nivolumab compared with placebo in all randomized patients and in patients with tumours expressing PD-L1. DFS was defined as time from randomization until death from any cause or recurrence of tumour. Patients were evaluated for recurrence every 12 weeks until week 96, every 16 weeks until week 160, and every 24 weeks until recurrence or treatment was discontinued for a maximum of 5 years.9
In a guidance document assessing end points used in clinical trials for cancer drugs and biologics, the FDA noted the benefits and drawbacks to using DFS.41 Advantages of the DFS outcome include being an objective measure based on quantitative assessment, being evaluable sooner, and needing a smaller sample size compared with studies using OS as an end point. Using DFS as an end point has its disadvantages such as variable definitions across studies, the potential for assessment bias especially in open-label studies, including noncancer deaths, and achieving a balanced timing of assessments across treatment groups is essential. The FDA guidelines recommend clearly defining the end point, outlining the schedule for assessments, including an estimation of the treatment effect size and ensuring blinding of treatment assignments to help reduce bias. According to the FDA guidance, the Oncology Drug Advisory Committee agreed in December 2003, that DFS prolongation represented clinical benefit if the magnitude of this benefit outweighed the toxicity of the adjuvant treatment.41 According to the FDA, DFS has served as the key outcome for FDA’s Accelerated or Traditional Approvals for adjuvant breast cancer hormonal therapy, adjuvant colon cancer, adjuvant cytotoxic breast cancer, adjuvant therapy for stage III non–small cell lung cancer, melanoma, renal cell cancer, and gastrointestinal stromal tumour.86 The FDA guidance noted that while reasonable likely surrogate end points, that have not been validated (i.e., not shown to reliably predict, or correlate with, clinical benefit) may be used under the FDA’s Accelerated Approval program to shorten the time to patient access, sponsors must verify the predicted clinical benefit with post-approval clinical trials.41,87 The FDA’s guidance on the trial design for adjuvant treatments of MIBC states that, if DFS is the primary end point, the FDA approval does not require demonstration of an OS benefit.88 However, the FDA recommends that the trial should include a plan for formal interim analysi of OS at the time of final DFS analysis and continued follow-up should be planned for to allow conduct of the final OS analysis.88 The FDA does not discuss the validity of DFS as surrogate end point for OS in their guidance.88 The EMA guidance notes that while a benefit in OS is the most convincing outcome, other possible primary end points include PFS, DFS, and patient-reported outcomes.42 The EMA guidance states that while DFS is considered a benefit to a patient regardless of cure, OS data should be reported in the adjuvant setting, as adjuvant therapy may limit treatment options upon disease recurrence; in established areas of adjuvant therapy (e.g., breast or colorectal cancer) and if a favourable benefit-risk ratio exists based on DFS, safety and survival data, and PFS results for next-line therapy are available, the reporting of mature survival data may be delayed until after licensing.42
Currently there are no universally accepted methods to assess validity of surrogate outcomes, however, correlation-based approaches are primarily used.89 In order to examine the validity of surrogate end points for OS, it has been suggested to evaluate the strength of correlation between OS and the surrogate outcome.90,91 For the purpose of assessing the validity of surrogates for regulatory approval, focus has been placed on trial-level surrogacy, that is, the correlation of treatment effect on the surrogate outcome with the treatment effect on OS91 (e.g., correlations between the HR of the surrogate outcome and the HR of OS or between the differences in the median of the surrogate outcome [i.e., median of surrogate outcome in treatment group minus median of surrogate in control group] and the difference in median OS duration [i.e., median OS in treatment group minus median OS in control group]). In contrast, correlations that assess the prognostic validity of a surrogate in individual patients (e.g., correlations of median DFS with median OS), cannot confirm the validity of a surrogate for predicting treatment effects on OS.90 Buyse, M (2011) similarly suggested that a good surrogate must have individual-level and trial-level association.92 The Institute of Quality and Efficacy in Health Care89 suggested that an observed correlation between the surrogate outcome and the clinical end point on its own, cannot adequately demonstrate validity of a surrogate. Instead, comprehensive data are required, preferable a meta-analysis of several randomized trials showing robust results. A requirement of large datasets was also suggested by Buyse et al. (2000)93 noting that multiple randomized studies are required to obtain precise estimates and to distinguish individual-level from trial-level associations between the end points and effects of interest.93 In the guidance of the Institute of Quality and Efficacy in Health Care89 it is cautioned that the validity of a surrogate is likely both disease specific and intervention specific and that conclusions about the validity of a surrogate cannot be easily applied across different indications or interventions.89 The guidance further suggests that a correlation between the surrogate and a clinical end point could be characterized as strong if the correlation coefficient is at least 0.85, as low, if the correlation coefficient is 0.7 or less, and as medium if the correlation coefficient is between 0.85 and 0.7.89 However, the guidance notes that currently no universally accepted threshold exists to determine validity.
Sonpavde, et al. (2011)94 conducted a retrospective review of 2,724 patients (844 [31%] patients had received adjuvant chemotherapy) from 10 centres worldwide to evaluate correlation between DFS at year 2 (DFS2) and 3 (DFS3) with OS at year 5 (OS5) using Cox proportional hazards modelling and the kappa statistic. Patients underwent radical cystectomy with curative intent for MIBC between 1980 and 2008. None of the patients included in the study received neoadjuvant therapy. DFS was defined as the duration from RC to disease recurrence or death from any cause. Survival rates at DFS2, DFS3, and OS5 were 0.63, 0.57, and 0.47, respectively. Of all recurrences 80.9% were within 2 years of RC and 90.6% within 3 years. Median time from disease recurrence to death was 5 months, indicating aggressive disease. Following RC, survival curves for DFS and OS overlapped for the first 50 months and separated afterwards. The overall agreement of DFS2 and DFS3 with OS5 was 79% (interquartile range [IQR] 77% to 80%) and 81% (IQR 80-82%), respectively. The strength of the correlation was constant regardless of pathological stage, adjuvant chemotherapy, year of RC, and centre of RC. The kappa statistics were 0.57 (95% CI 0.53-0.60) for DFS2/OS5 and 0.61 (95% CI 0.58-0.64) for DFS3/OS5, indicating moderate to substantial agreement. The HR for DFS as a time-dependent variant was 12.7 (95% CI 11.60-13.90), indicating a strong relationship. Lastly, authors hypothesized that a relative increase in DFS2 by 22% or more may represent a clinically relevant improvement. The study supports 2- and 3-year DFS as a potential surrogate for 5-year OS. However, there were a number of limitations including the retrospective design, selection bias from excluding patients with incomplete data, not representing some of current practices, undefined chemotherapy regimen (in 31% of patients), no differentiation between locoregional and distant metastasis, and noncancer mortality in the elderly. Sonpavde, et al. noted that while it has not been defined what a relevant level of association of DFS to OS is, the authors suggest that the level of association found in their study was similar to that observed in colon cancer. The study also noted that improved systemic therapy in the future may extend survival after recurrence and reduce association between DFS and OS.94
Kim, et al. (2015)25 performed a retrospective, external validation study to confirm the Sonpavde et al. results for correlations between DFS2, DFS3, and OS5. The group collected clinical data of 422 patients (adjuvant chemotherapy was administered in 102 [24.2%] patients) treated with RC for UC of the bladder cancer between 1991 and 2012 from 1 centre in South Korea. None of the included patients received neoadjuvant therapy. DFS was defined as the interval between RC and disease recurrence or death from any cause. Based on their results, DFS2, DFS3, and OS5 rates were 0.76, 0.72, and 0.67, respectively. Among all recurrences, 91 cases (68.4%) occurred within the first 2 years and 107 cases (80.4%) within the first 3 years following RC. Kaplan-Meier analysis with the log-rank test showed significant differences in OS rates according to DFS2 and DFS3 status following RC (P < 0.001). Kappa statistics for DFS2/OS5 was 0.59 (standard error [SE] = 0.045) and DFS3/OS5 was 0.62 (SE = 0.043) (P < 0.05). Similarly, Kendall tau-b test for DFS2/OS5 was 0.60 (SE = 0.044) and DFS3/OS5 was 0.63 (SE = 0.043) (P < 0.05). These results indicate moderate to substantial agreement between DFS2, DFS3, and OS5. The HR between OS in patients with disease recurrence and those without recurrence declined according to the extended disease-free interval and reached the lowest level at 36 months. Authors estimated that OS5 rates for patients who were recurrence-free exceeded 90% at 24 months after RC. Subgroup analysis according to adjuvant chemotherapy status showed correlation was maintained. Kim et al. noted that their study confirmed significant correlations of DFS2 and DFS3 with OS5 und suggested that DFS2 and DFS3 could be used as potential early surrogates for OS. However, the authors identified a number of limitations including the retrospective, non-randomized design, small sample size from a single institution, guideline changes in surgical technique and pathological report content over time, inter-physician variation in surgical expertise, determination or type of adjuvant chemotherapy regimen, and post-operative follow-up strategies.
Fajkovic et al. (2013)95 evaluated if DSF with 2- to 3- year median follow-up is an appropriate surrogate end point for OS in patients with UTUC. None of the patients included in the study had received neoadjuvant chemotherapy. Using retrospective data from 2,492 patients treated with radical nephroureterectomy for UTUC at 23 international centre, kappa statistics were calculated to determine agreement between DSF and OS. In addition, the HR was used for predicting OS from DFS. Fajkovic et al. found that the overall agreement between 2- and 3-year DFS with 5-year OS were 85% and 87%, respectively. The kappa statistics indicated moderate reliability for 2-year DFS and 5-year OS (kappa, 0.59; 95% CI, 0.55 to 0.63) and 3-year DFS and 5-year OS (kappa, 0.61; 95% CI, 0.61 to 0.68). The hazard ratio for DFS as a time-dependent variable for predicting OS indicated a strong relationship between DFS and OS (HR, 11.5; 95% CI, 9.1 to 14.4). Limitations noted by the authors included the retrospective study design and the acknowledgement that adequate time is required to assess the delay versus prevention of recurrence hypothesis. The study authors acknowledged that external validation is required before accepting DFS as an appropriate surrogate end point.
Nuhn et al. (2012)96 externally validated the correlation between DFS2 or DFS3 and OS5 for patients with pT2-4a MIUC that underwent RC. The study retrospectively reviewed the records of 2,483 patients which underwent RC for their UC of the bladder from 8 European between 1989 and 2008. Furthermore, the authors included records of 745 patients with positive soft tissue surgical margins and patients with -T0-T1, pT4b, which were excluded in the previous study. The kappa statistics were 0.73 (SE: 0.016) for DFS2/OS5 and 0.80 (SE: 0.014) for DFS3/OS5. The results for the kappa statistics for those previously excluded patients were 0.67 (SE: 0.033) for DFS2/OS5 and 0.78 (SE: 0.027) for DFS3/OS5. The overall agreement of DFS2 and DFS3 with OS5 was 86.5% and 90.1%, respectively, in those patients previously excluded. The authors concluded that the findings suggest that DFS2 and DFS3 are surrogate markers for survival with RC.
Conclusion
Four articles were summarized, which assessed the appropriateness of DFS as a surrogate outcome for OS. The studies suggested that, at the individual level, there was a moderate to substantial agreement between DFS2, DFS3, and OS5. Key limitations identified by the CADTH review team included the retrospective, non-randomized study designs of all 4 studies, which could not control for the presence of unidentified confounding factors. Furthermore, it has been noted that the validity of a surrogate is likely both disease specific and intervention specific.89 It is unclear how many patients in the 4 studies discussed previously aligned with the inclusion criteria of being at high risk of recurrence as defined in the CheckMate274 trial. As well, the CheckMate274 study included 319 (45%) patients who had received neoadjuvant therapy, while none of the patients included in the analyses of the 4 studies discussed received neoadjuvant therapy; a proportion of patients in the 3 studies by Sonpavde et al., Kim et al., and Fajkovic et al. received adjuvant chemotherapy, while none of the patients in the CheckMate274 received adjuvant chemotherapy. It is unclear if the results of the retrospective analyses discussed previously can be fully generalized to the patient population of the CheckMate274 trial. Additionally, it has been noted that a valid surrogate outcome should demonstrate an individual-level and a trial-level association92; especially if the surrogate end point is expected to predict a treatment effect on the true clinical end point, given an observed treatment effect on the surrogate end point. The 4 studies discussed previously suggested a modest to substantial individual-level association, which may indicate that DFS and OS are potentially causally linked to each other. However, no data on trial-level correlation was identified and therefor it is not known if a proportion (and if yes, what size of proportion) of a treatment effect would be captured by DFS. No study was identified that investigated patient-level and or trial-level association with nivolumab in the present target population; therefore, it cannot be firmly concluded to what extend the improvements in DFS observed in patients in the nivolumab group of the CheckMate274 trial would translate into OS benefits.
Pharmacoeconomic Review
Abbreviations
BIA
budget impact analysis
DFS
disease-free survival
EORTC
European Organisation for Research and Treatment of Cancer
HR
hazard ratio
ICER
incremental cost-effectiveness ratio
ITC
indirect treatment comparison
ITT
intention to treat
KM
Kaplan-Meier
MIUC
muscle-invasive urothelial carcinoma
OS
overall survival
PFS
progression-free survival
QALY
quality-adjusted life-year
WTP
willingness to pay
Executive Summary
The executive summary comprises 2 tables (Table 1 and Table 2) and a conclusion.
Item | Description |
|---|---|
Drug product | Nivolumab (Opdivo), IV infusion, 10 mg/mL |
Submitted price | Nivolumab 40 mg and 100 mg single-use vials Price per unit: $1,955.56 per 100 mg Price per unit: $788.22 per 40 mg |
Indication | As monotherapy for the adjuvant treatment of adult patients with urothelial carcinoma who are at high risk of recurrence after undergoing radical resection of urothelial carcinoma |
Health Canada approval status | NOC |
Health Canada review pathway | Standard |
NOC date | June 27, 2022 |
Reimbursement request | As per indication |
Sponsor | As per application overview |
Submission history | Previously reviewed: yes Indication: adjuvant treatment of completely resected esophageal or gastroesophageal junction cancer in patients who have residual pathologic disease following prior neoadjuvant chemoradiotherapy Recommendation date: January 26, 2022 Recommendation: Reimburse with conditions Indication: treatment of adult patients with advanced (not amenable to curative therapy or local therapeutic measures) or metastatic hepatocellular carcinoma who are intolerant to or have progressed on sorafenib therapy Recommendation date: October 4, 2018 Recommendation: Do not reimburse Indication: treatment of adult patients with classical Hodgkin lymphoma that has relapsed or progressed after ASCT and brentuximab vedotin, or ≥ 3 lines of systemic therapy including ASCT Recommendation date: March 2, 2018 Recommendation: Reimburse with clinical criteria and/or conditions Indication: advanced or metastatic renal cell carcinoma in patients who have received prior systemic therapy Recommendation date: June 30, 2016 Recommendation: Reimburse with clinical criteria and/or conditions |
ASCT = autologous stem cell transplantation; NOC = notice of compliance.
Table 2: Summary of Economic Evaluation
Component | Description |
|---|---|
Type of economic evaluation | Cost-utility analysis Markov model with 3 health states (disease-free, recurred disease [including regional and distant recurrence], and death) |
Target population | Patients with MIUC who are at high risk of recurrence following radical resectiona |
Treatment | Nivolumab |
Comparators | Observation (i.e., no active treatment) Adjuvant chemotherapy |
Perspective | Canadian publicly funded health care payer |
Outcomes | QALYs, LYs |
Time horizon | 30 years |
Key data sources |
|
Submitted results |
|
Key limitations |
|
CADTH reanalysis results |
|
DFS = disease-free survival; EORTC = European Organisation for Research and Treatment of Cancer; ICER = incremental cost-effectiveness ratio; inc. = incremental; ITT = intention to treat; LY = life-year; MIUC = muscle-invasive urothelial carcinoma; OS = overall survival; QALY = quality-adjusted life-year; WTP = willingness to pay.
aThe sponsor indicated that the modelled population is functionally the same as the Health Canada–indicated population, as tumours on the urethra, which are part of urothelial carcinoma but not MIUC, are exceedingly rare, and it is expected that patients with urethra primary tumours represent less than 1% of all patients with urothelial carcinoma.
Conclusions
Evidence from the CheckMate 274 trial indicated that adjuvant treatment with nivolumab demonstrated a clinically meaningful and statistically significant improvement in disease-free survival (DFS) in patients at high-risk of recurrence after radical resection of muscle-invasive urothelial carcinoma (MIUC) when compared to placebo, which was considered a proxy for observation or surveillance (i.e., no active treatment). However, as no overall survival (OS) data from the CheckMate 274 trial are available and no study was identified that investigated patient-level and or trial-level associations with nivolumab in the present target population, it cannot be firmly concluded to what extent the improvements in DFS observed in patients in the nivolumab group of the CheckMate 274 trial would translate into OS benefits. While nivolumab appeared to have more adverse events than placebo, the clinical experts consulted by CADTH anticipated that the toxicity profile observed with nivolumab would be more favourable than adjuvant chemotherapy. The relative efficacy of nivolumab and adjuvant chemotherapy is highly uncertain because there is no head-to-head comparison of the 2 treatment options. The sponsor submitted an indirect treatment comparison (ITC), but it is associated with uncertainty due to the limited number of included studies, and heterogeneity across the included studies in terms of trial design and eligibility as well as outcome definitions. An additional ITC was identified in the literature, but was associated with similar limitations. The sponsor’s economic evaluation did not use ITC evidence to inform the comparison of nivolumab and chemotherapy, but was based on a naive comparison of efficacy data obtained from the CheckMate 274 trial and the European Organisation for Research and Treatment of Cancer (EORTC) 30994 study which suggested that nivolumab was not as effective as chemotherapy (or at best, similarly effective). The naive comparison findings were aligned ||||||||||||||||||||||||||||||, and another published ITC identified in the literature.
CADTH was unable to address all identified limitations, but made corrections and several revisions to derive the CADTH base case. In patients who had received neoadjuvant chemotherapy, or were not able to receive adjuvant chemotherapy, in the CADTH base case nivolumab is associated with an incremental cost-effectiveness ratio (ICER) of $112,826 per quality-adjusted life-year (QALY) compared with observation (incremental costs: $78,222; incremental QALYs: 0.70). A price reduction of at least 56% is required for nivolumab to be cost-effective compared to observation at the willingness-to-pay (WTP) threshold of $50,000 per QALY. Scenario analyses highlighted that the key driver included assumptions on DFS data prediction, cure time points, and health utility values. In patients who had not received neoadjuvant chemotherapy and were able to receive adjuvant chemotherapy, nivolumab was dominated by adjuvant chemotherapy (i.e., nivolumab was more costly and associated with fewer QALYs). This finding was robust to changes in model’s assumptions and input parameters. Given that nivolumab is less effective and incurred higher non-drug costs than adjuvant chemotherapy based on the available evidence, even with a 100% price reduction for nivolumab, adjuvant chemotherapy remained the optimal treatment at a $50,000 per QALY WTP threshold.
CADTH was unable to fully assess the uncertainty associated with external data used to derive DFS and the transitions from recurrence to death, or the implications of the sponsor’s 3-state model compared with a more appropriate 4-state model which separated locoregional and distant recurrence. Given the uncertainty associated with whether nivolumab will lead to an improvement in OS, alongside the overestimation of relative DFS due to the application of the sponsor’s cure assumption, the predicted benefit associated with nivolumab may have been overestimated. As a result, an incremental gain of 0.81 life-years compared with observation shown in the CADTH base case should be interpreted with caution. Additional information is required to validate the long-term survival outcomes predicted by the analysis. If OS benefits associated with adjuvant nivolumab are not realized, the ICER of nivolumab relative to observation will increase.
The economic results align with the available clinical evidence in suggesting that there is some clinical benefit with respect to DFS for nivolumab when compared with observation, and that in patients eligible for treatment, nivolumab may be less effective than adjuvant chemotherapy, although CADTH could not make conclusions regarding the relative efficacy of nivolumab and adjuvant chemotherapy given substantial limitations with the comparative evidence. The submitted clinical evidence is not sufficient to draw conclusions on the long-term clinical benefits of adjuvant nivolumab in patients with MIUC. As such, whether treatment will outweigh the potential harms associated with overtreatment, particularly in patients that would not otherwise be treated, is unknown.
Stakeholder Input Relevant to the Economic Review
This section is a summary of the feedback received from the patient groups, registered clinicians, and drug plans that participated in the CADTH review process (specifically, information that pertains to the economic submission).
Patient input received from the Bladder Cancer Canada was collected through online surveys and 1-to-1 interviews between February 16 and March 15, 2022 with patients with MIUC residing in Canada (n = 6) and the US (n = 1). Patients noted fatigue as the commonly reported side effect with currently available treatments, followed by nausea, constipation, and hospitalization. Patients ranked the bladder cancer treatment outcomes from most to least important as preventing recurrence, controlling disease progression, reducing symptoms, maintaining quality of life, and managing side effects. Two patients who had experience with nivolumab noted nivolumab was more effective at controlling disease progression and preventing recurrence compared to other therapies. Regarding drug side effects and quality of life, patient input did not concur; 1 patient reported improvement and the other reported deterioration in these outcomes. Generally, patients with MIUC are willing to tolerate treatment side effects given treatment improves patient-preferred treatment outcomes.
Clinician input received from the Bladder Cancer Canada and Ontario Health (Cancer Care Ontario) Genitourinary Cancer Drug Advisory Committee noted current standard of care is different for subgroups within the MIUC population. Patients who did not receive neoadjuvant chemotherapy and undergo radical resection are currently treated with cisplatin and gemcitabine-based chemotherapy in the adjuvant setting. Patients who received neoadjuvant chemotherapy or have poor performance status after surgery are currently surveilled. Clinicians noted that current treatment goals include increasing OS, preventing metastases, controlling disease progression, maintaining quality of life, minimizing adverse events, and reducing the severity of symptoms. The clinicians indicated that progression-free survival (PFS) is a good surrogate of OS in urothelial carcinoma. The clinicians noted that nivolumab would be used in the adjuvant setting following radical resection for patients at high risk of recurrence, with or without neoadjuvant cisplatin-based chemotherapy, or for patients who are unfit or ineligible to receive adjuvant cisplatin-based chemotherapy and did not receive neoadjuvant chemotherapy. The clinicians noted nivolumab may be given to patients with lower kidney function and higher performance status than platinum-based chemotherapy. Further, nivolumab would be an additional option for patients and fill a gap in the current standard of care. Patients would be identified based on post-operative pathology reports of cystectomy specimens. Clinicians indicated that a clinically meaningful response to treatment would include delay of recurrence, delay of metastases, and prolonged survival.
CADTH-participating drug plans noted considerations related to the relevant comparators, therapy initiation, and potential implementation factors. The plans also noted different comparators for subgroups within the MIUC population, which mostly aligned with the clinician input. Public drug plans noted patients with muscle-invasive bladder cancer are currently observed in the adjuvant setting if they had received neoadjuvant chemotherapy with cisplatin and gemcitabine before surgery. On the other hand, patients with upper tract urothelial carcinoma receive chemotherapy with platinum (either cisplatin or carboplatin) and gemcitabine in the adjuvant setting. However, the current standard of care after surgery is surveillance. The plans noted eligibility for treatment should be aligned with the CheckMate 274 trial. The plans indicated jurisdictions would be implementing a weight-based dose of nivolumab (3 mg/kg every 14 days, up to a maximum of 240 mg per dose). The drug plans also anticipate higher market uptake if nivolumab becomes the preferred standard of care in the adjuvant setting.
Several of these concerns were addressed in the sponsor’s model.
Treatment side effects and its impact on disease recurrence, survival, and quality of life were considered.
Cost-effectiveness of nivolumab compared to adjuvant chemotherapy among patients who are eligible for cisplatin but do not receive neoadjuvant chemotherapy was included in a separate base-case analysis.
In addition, CADTH addressed some of these concerns as follows.
A weight-based dosing of nivolumab was considered in CADTH’s scenario analysis.
CADTH was unable to address the following concerns raised from stakeholder input.
Cost-effectiveness of nivolumab compared to adjuvant chemotherapy among patients with upper tract urothelial carcinoma or those who do not receive neoadjuvant therapy (including cisplatin-eligible and -ineligible patients) was unknown due to the lack of data.
Economic Review
The current review is for nivolumab (Opdivo) for patients with MIUC who are at high risk of recurrence following radical resection.
Economic Evaluation
Summary of Sponsor’s Economic Evaluation
Overview
The sponsor submitted a cost-utility analysis evaluating the cost-effectiveness of nivolumab for the adjuvant treatment of patients with MIUC who are at high risk of recurrence following radical resection.1 The sponsor presented 2 analyses; in 1 analysis, the sponsor estimated the cost-effectiveness of nivolumab compared with observation (i.e., no active treatment) in patients who received neoadjuvant chemotherapy, and patients who are ineligible for cisplatin. In the other analysis, the sponsor estimated the cost-effectiveness of nivolumab compared with adjuvant chemotherapy in cisplatin-eligible patients who have not received prior neoadjuvant chemotherapy. The modelled population (as captured within the 2 analyses) was in line with the trial population. The sponsor’s Health Canada indication was revised upon final Notice of Compliance to “the adjuvant treatment of adult patients with urothelial carcinoma who are at high risk of recurrence after undergoing radical resection of urothelial carcinoma;” the sponsor indicated that the modelled population is functionally the same as the Health Canada–indicated population.1
Nivolumab is available as 40 mg and 100 mg single-use vials (10 mg/mL). According to the product monograph, the recommended dose is 240 mg every 2 weeks (30-minute IV infusion) or 480 mg every 4 weeks (30-minute IV infusion) as long as clinical benefit is observed or until treatment is no longer tolerated up to a total treatment duration of 1 year.2 The cost of nivolumab is $782.22 for a 40 mg vial and $1,955.56 for a 100 mg vial, equating to a per 7-day cost of $2,347. The sponsor assumed no vial sharing in the drug cost calculation. The sponsor’s analysis assumed that there was no treatment cost for those in the observation option, and a total 7-day adjuvant chemotherapy cost of $527.1
The clinical outcomes were QALYs and life-years. The economic analysis was undertaken over a time horizon of 30 years from the perspective of a Canadian publicly funded health care system.1 Costs and QALYs were discounted at a rate of 1.5% per annum.3
Model Structure
The sponsor submitted a Markov model with a cycle length of 7 days and the following mutually exclusive health states: disease-free, recurred disease (comprising who have either had a local recurrence or a distant recurrence), and death (Appendix 3, Figure 1).1 All patients begin in the disease-free health state where they can either remain, transition to death, or transition to the recurred disease health state. Patients in the recurred disease health state can remain or transition to death. All patients in the disease-free state are assumed to receive nivolumab for up to 12 months (mean number of doses = 16.9). Adjuvant chemotherapy was estimated for a duration of between 3.2 months and 3.75 months. Disease recurrence included local urothelial tract, local non-urothelial tract, or distant. The model incorporated a curative assumption, where the risk of recurrence decreased to 0% after 5 years.1
Model Inputs
The modelled population reflected the baseline patient characteristics of the enrolment population in the CheckMate 274 trial, a randomized, double-blind, placebo-controlled phase III trial, which evaluates nivolumab as an adjuvant therapy in adults (aged ≥ 18 years) with MIUC who are at high risk of recurrence following radical resection.1,4 The sponsor’s model assumed a mean age of 65.6 years, a median weight of |||||| kilograms, and a mean body surface area of |||||| m2.
The transition from disease-free to recurred disease from year 0 to year 3 was informed by Kaplan–Meier (KM) curves for DFS obtained from the CheckMate 274 trial (data cut-off date of May 19, 2021).4 DFS data based on the intention-to-treat (ITT) population (n = 709) were used to inform a comparison of nivolumab versus observation, while pooled DFS data for a subgroup of trial participants who were cisplatin-eligible but did not receive neoadjuvant chemotherapy (n = 230) were used to inform the nivolumab inputs for the comparison of nivolumab versus adjuvant chemotherapy, which was undertaken as a naive comparison. CADTH noted that the sponsor undertook an ITC comparing nivolumab with adjuvant chemotherapy though this comparison was not used to inform the economic analysis. The sponsor adjusted the 4- and 5-year DFS data for the nivolumab and observation arms using hazard rates derived from PFS data observed in the deferred chemotherapy arm of the EORTC 30994 study (Sternberg et al.).5 This trial was an open-label, randomized, phase III trial that recruited patients with histologically proven urothelial carcinoma of the bladder, Pt3–Pt4 disease or node positive (Pn1-3) M0 disease, or both, after radical cystectomy and bilateral lymphadenectomy from European countries and Canada between 2002 and 2008. The sponsor also used PFS data from the immediate chemotherapy arm of this trial to represent DFS among cisplatin-eligible patients who did not receive prior neoadjuvant chemotherapy. After 5 years, the sponsor assumed that patients in the disease-free health state who did not progress to the recurred disease health state would not have a recurrence; therefore, they would only transition from the disease-free health state to death based on the age-specific background mortality rates observed in the general Canadian population.
Transition from recurred disease state to death was assumed to be independent of treatment but dependent on whether patients were eligible to receive cisplatin. For cisplatin-eligible patients, the sponsor derived survival data from the phase III randomized clinical trial EORTC 30987, investigating paclitaxel in combination with cisplatin and gemcitabine compared with gemcitabine + cisplatin for the treatment of patients with locally advanced or metastatic urothelial carcinoma without prior systemic therapy.6 For cisplatin-ineligible patients, survival data were obtained from the phase II/III randomized multi-centre clinical trial EORTC study 30986 that focused on patients with advanced urothelial carcinoma, who were cisplatin ineligible as assessed by a WHO performance score of 2 or more (n = 42, 17.6%), or impaired renal function assessed by a glomerular filtration rate of greater than 30 and less than 60 mL/min (n = 131, 55.0%) or both (n = 65, 27.3%).7 An exponential distribution was fit to the data from both studies to estimate the transition over the model time horizon.
The sponsor assumed that cisplatin-ineligible patients who received nivolumab, adjuvant chemotherapy, or were under observation may receive pembrolizumab as a subsequent line of therapy. The sponsor performed an ITC and derived the OS benefits of pembrolizumab compared to carboplatin-based regimens in cisplatin-ineligible patients and patients expressing ≥ 1% programmed death-ligand 1.1 The ITC pooled OS data from the KEYNOTE-361 and IMvigor130 trials. A resulting hazard ratio (HR) of 0.83 (95% CI, 0.57 to 1.18) was applied to the OS curve reported in the EORTC 30986 study to reflect the survival benefit of pembrolizumab among cisplatin-ineligible patients.8,9
Health state utility values were derived from the EuroQol 5-Dimensions 3-Levels questionnaire data collected as part of the CheckMate 274 trial, using the Canadian value set. The sponsor used linear mixed-effects repeated measures models with random intercepts to account for repeated EuroQol 5-Dimensions 3-Levels questionnaire measurements per subject within a health state. Utility scores were further adjusted to reflect declining utility due to grade 3 or 4 adverse events that affected at least 2% of all study subjects enrolled in the CheckMate 274 trial. Disutility due to adverse events were obtained from the published literature.10-12
Costs included drug (acquisition, administration, monitoring, subsequent treatments), disease management, adverse events, and terminal care. Drug acquisition costs for each treatment were sourced from IQVIA Delta PA and the publicly available sources. Drug administration costs including costs associated with vial administration for IV therapies were obtained from the Ontario Schedule of Benefits. Treatment dosing of subsequent treatments was based on previous pCODR submissions.13-15 The proportion of patients receiving pembrolizumab was informed by the CheckMate 274 trial. For those receiving other treatments, the sponsor assumed that patients might receive gemcitabine + cisplatin or gemcitabine + carboplatin; patient distribution was informed by the Canadian sources.
The sponsor assigned disease management costs for disease-free and recurred disease health states. Resource use per cycle was sourced from the previous National Institute for Health and Care Excellence Health Technology Assessment report and verified by clinical experts,1,16 while unit costs were sourced from the Ontario Schedule of Benefits. The model further assumed that disease management costs for the disease-free health state were only incurred for the first 5 years and following those 5 years it was assumed patients were no longer followed up if they maintained DFS. The disease management costs for the recurred disease health state were applied throughout the model time horizon. A terminal care cost was applied to all patients who transitioned to the death health state over the time horizon. The cost estimate was based on hospital costs incurred during the 30 days of cancer treatment in Canada cited in a published cost-utility analysis of atezolizumab in patients with metastatic bladder cancer.17
Summary of Sponsor’s Economic Evaluation Results
All analyses were run probabilistically with 1,000 iterations. The deterministic and probabilistic results were similar, and the probabilistic findings are presented in the paragraphs that follow.
Base-Case Results
In the sponsor’s base case, nivolumab was associated with an ICER of $64,046 per QALY when compared to observation in patients who received neoadjuvant chemotherapy and those patients who were ineligible for cisplatin, and nivolumab was dominated (i.e., higher costs but fewer QALYs) when compared to adjuvant chemotherapy in cisplatin-eligible patients who had not received neoadjuvant chemotherapy (Table 3). At a WTP of $50,000 per QALY, the probabilities of nivolumab being cost-effective was 0% compared to observation and 0% compared to adjuvant chemotherapy.
At the end of the model time horizon (i.e., 30 years), 3.2% of cisplatin-ineligible patients or those who received neoadjuvant chemotherapy and 4.1% of cisplatin-eligible patients who did not receive neoadjuvant chemotherapy were still alive in the model.
A breakdown of the sponsor-submitted results for the base-case population by trial duration and extrapolated period shows that 77% of the expected QALY gains come from the time beyond the trial period.
Table 3: Summary of the Sponsor’s Economic Evaluation Results
Drug | Total costs ($) | Incremental costs ($) | Total QALYs | Incremental QALYs | ICER vs. reference ($/QALY) |
|---|---|---|---|---|---|
Patients who were not eligible to receive cisplatin or those who received neoadjuvant therapy | |||||
Observation | 25,097 | Reference | 5.46 | Reference | Reference |
Nivolumab | 100,458 | 75,361 | 6.64 | 1.18 | 64,046 |
Patients who were eligible to receive cisplatin but did not receive neoadjuvant therapy | |||||
Adjuvant chemotherapy | 28,067 | Reference | 7.74 | Reference | Reference |
Nivolumab | 100,409 | 72,342 | 6.68 | –1.06 | Dominated |
ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year.
Source: Sponsor’s pharmacoeconomic submission.1
Sensitivity and Scenario Analysis Results
The sponsor’s deterministic 1-way sensitivity analyses on the base case revealed minimal variations in the cost-effectiveness results, showing that the main drivers were health utility associated with disease-free health state and a discount rate for QALYs.
The sponsor performed scenario analyses by varying a time horizon, using alternative assumptions on transition probability from disease-free health state, using alternative parametric models for DFS curves, varying cure points, and varying survival rates for recurred disease health state. Key drivers of the cost-effectiveness results in patients who were cisplatin ineligible or those who received neoadjuvant chemotherapy included a shorter time horizon of 15 years ($84,794 per QALY gained) and using a 4-year curative assumption ($61,936 per QALY gained). For patients who were eligible to receive cisplatin but who did not receive neoadjuvant therapy, nivolumab remained a dominated option in all scenario analyses.
CADTH Appraisal of the Sponsor’s Economic Evaluation
CADTH identified several key limitations to the sponsor’s analysis that have notable implications on the economic analysis:
Long-term survival benefits of nivolumab are highly uncertain. The sponsor’s model assumed that patients received nivolumab for a maximum of 12 months (16.9 doses), but the DFS extrapolations assumed that the trend of DFS benefit of nivolumab was sustained after treatment discontinuation until year 5. After this time point, it was assumed patients were functionally cured, and age-specific mortality rates of the general Canadian population were applied over the remainder of the time horizon. The same approach was applied to both analyses (nivolumab versus observation and nivolumab versus adjuvant chemotherapy). The clinical experts consulted by CADTH indicated that it is uncertain whether the treatment benefits associated with nivolumab would be maintained after treatment discontinuation to the extent predicted in the sponsor’s model. As the sponsor’s model implies a positive association between DFS and OS, extrapolations for these end points reflect within-trial trends in the rates of progression and death. The long-term survival benefits of nivolumab are therefore highly uncertain given that the OS data from the CheckMate 274 trial are currently unavailable.
The sponsor attempted to minimize the level of uncertainty by limiting DFS extrapolations to 5 years and applying a 5-year cure assumption. Applying this 5-year cure rate to both the nivolumab and comparator arm introduced uncertainty, as it implies a shorter post-treatment period for the nivolumab arm given the duration of treatment of up to 1 year, as opposed to the duration of up to 4 cycles of chemotherapy or no active treatment with regard to monotherapy. A difference in time to cure rate from treatment discontinuation should have been applied between the different treatments; however, the sponsor’s model structure precluded CADTH from exploring the impact of this limitation. As such, this overestimates the benefit of nivolumab relative to the comparators. Furthermore, CADTH was concerned about the use of an external data source to inform DFS data as it increases the number of required assumptions in the model and associated uncertainty. Combining data from the CheckMate 274 and EORTC 30994 trials assumed that both trials had comparable patient and study characteristics. This assumption is challenging to fulfill given that the sponsor generated pseudo-patient-level data and hazard rates from the KM curves reported in the EORTC 30994 study. Given this identified limitation, it was unclear whether the survival benefits estimated in the model were due to the efficacy of nivolumab, or due to the parameter and structural uncertainty within the model. CADTH noted that DFS was based on investigator assessment. While investigator assessment may be considered to have greater external validity, the internal validity of investigator assessment is limited compared with the central review.
CADTH further noted that the sponsor derived probabilities of transition from a disease-free health state to recurred disease and death health states from DFS data. This approach introduces structurally dependent assumptions between the 2 transition probabilities and implies that improved DFS would translate to survival benefits. This assumption is deemed speculative given the unknown OS data from the CheckMate 274 trial. Furthermore, CADTH noted that the DFS data appraised as part of the clinical evidence were based on the full Clinical Study Report submitted to CADTH, while the DFS data used in the economic model were based on updated DFS data from a more recent data cut. These updated data were provided to CADTH, although only in abstract form. In this abstract, the KM curves appeared to close between the nivolumab and observation arms toward the tail compared with the KM curves in the earlier data cut.18 However, since the DFS data used to inform the economic model were based on an abstract, and limited information on methods was provided, CADTH was therefore unable to appraise the quality of such data.
CADTH attempted to address this concern, but the reanalysis was confined to the available data.
Clinical experts consulted by CADTH advised that predicted DFS curves from the Gompertz model were plausible based on the survival estimates at years 1, 2, and 3. As such, CADTH selected the Gompertz model to predict DFS data in years 4 and 5 in the base-case analysis of nivolumab versus observation and tested alternative parametric curves in scenario analyses. The sponsor’s 5-year curative assumption was retained in CADTH’s base case. Due to data limitations, CADTH did not change the DFS extrapolation for adjuvant chemotherapy in the sponsor’s base case. A scenario analysis using the alternative data source was performed to assess the uncertainty in the cost-effectiveness findings.
The submitted 3-health state Markov model is insufficient to capture care pathway, costs, and outcomes of patients with MIUC in the adjuvant setting. The sponsor’s 3-state Markov model included a disease-free state, a recurred disease state, and death. The sponsor’s decision to combine local and distant recurrence within a single recurred disease health state to estimate the costs and QALYs associated with each treatment option over the model time horizon was inappropriate, as fails to reflect disease trajectory and the potential differences in care pathways, costs, risk of death, and quality of life between patients with local recurrence and those with distant recurrence. The implication of the sponsor’s decision to group recurrence implies that patients who experience distant recurrence have the same prognosis, experience the same quality of life, and receive the same types of treatments and overall care costs as patients who experience a local recurrence. According to clinical experts consulted by CADTH, patients experiencing local and distant recurrence are likely to require different types of monitoring and treatment options, and patients with local recurrence would expect to have better health outcomes than patients who experience a distant recurrence. Using a 3-state Markov model also precluded the model’s ability to account for the sequence and the type of subsequent lines of chemotherapy. Clinical expert consulted by CADTH suggested that patients with MIUC may receive up to 3 lines of subsequent therapy and that the number of lines of therapy would influence patient survival.
CADTH was unable to assess the uncertainty associated with the model structure due to data limitations and the lack of flexibility with the submitted model.
Comparative efficacy of nivolumab and comparators in the modelled population is uncertain. For the comparison of nivolumab and observation, the sponsor derived DFS data from the ITT population of CheckMate 274 trial, which included both patients who received and did not receive prior neoadjuvant cisplatin-based chemotherapy. It is uncertain whether the efficacy of nivolumab versus observation observed in the ITT population is generalizable to patients with MIUC who are likely to be observed (i.e., not eligible to receive adjuvant chemotherapy) in Canada. According to the stakeholders and clinical experts consulted by CADTH, patients who receive prior neoadjuvant chemotherapy or have poor performance status after surgery are typically put under observation, while patients who have not received neoadjuvant chemotherapy before undergoing radical resection and are eligible to receive adjuvant chemotherapy, are typically treated with cisplatin and gemcitabine-based chemotherapy. As a result, DFS derived from the ITT population may not be generalizable to represent the treatment benefits of nivolumab in patients who received neoadjuvant chemotherapy or who are ineligible for cisplatin. There may be patients who are eligible for adjuvant chemotherapy that decline currently available treatments; this population has not been adequately addressed in the sponsor’s economic evaluation.
For the comparison of nivolumab and adjuvant chemotherapy, although the modelled population was based on a subset of cisplatin-eligible patients who did not receive neoadjuvant chemotherapy, the relative efficacy of nivolumab and adjuvant chemotherapy was associated with substantial uncertainty due to the use of a naive comparison. The sponsor derived the efficacy of nivolumab by pooling DFS data of cisplatin-eligible patients who did not receive neoadjuvant chemotherapy from the nivolumab and observation arms but obtained the efficacy of adjuvant chemotherapy from the active immediate chemotherapy arm of the published trial.5 The sponsor’s approach is subject to bias as unmeasured patient characteristics may confound the effect of nivolumab on DFS.19 ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
CADTH was unable to address this limitation due the lack of data.
Subsequent treatments for recurrent disease did not reflect clinical practice in Canada. The sponsor assumed that 100% of patients experiencing disease recurrence would receive subsequent chemotherapy, applied constantly over time. According to the clinical experts consulted by CADTH, fewer patients are likely to be eligible for subsequent chemotherapy. This expert opinion is supported by the CheckMate 274 trial, which reported that 55% to 60% of patients who experience disease recurrence received subsequent chemotherapy. A higher percentage of patients receiving subsequent chemotherapy would overestimate the incremental costs associated with nivolumab and observation as more patients undergoing observation experience disease recurrence and require subsequent therapies. Clinical experts consulted by CADTH indicated that the percentage is likely to range between 40% to 70%.
CADTH set an equal percentage of patients receiving subsequent chemotherapy (50%) for nivolumab and observation.
CADTH further noted that subsequent therapies used in the sponsor’s model did not represent currently available treatments in Canada. The sponsor considered cisplatin plus gemcitabine, carboplatin plus gemcitabine, and pembrolizumab. Based on feedback from clinical experts consulted by CADTH, the list of these treatments was outdated as patients may also be eligible for avelumab or enfortumab after disease recurrence. In addition, the distribution of subsequent chemotherapy was inappropriate. The clinical experts anticipated that the proportions of patients receiving carboplatin plus gemcitabine would be larger than those receiving cisplatin plus gemcitabine. This expectation applies to patients who receive adjuvant nivolumab or are under observation. For those receiving adjuvant chemotherapy, about one-third are likely to be resistant to chemotherapy, thereby eligible for pembrolizumab, and 25% may still be eligible for cisplatin during disease recurrence.
CADTH was unable to address the limitation regarding the omission of new subsequent treatments due to the lack of data. However, as part of CADTH’s base case comparing nivolumab to observation, the proportions of patients receiving cisplatin plus gemcitabine, or carboplatin plus gemcitabine, were switched to reflect current practice. For the comparison with adjuvant chemotherapy, the proportions of patients receiving cisplatin plus gemcitabine, carboplatin plus gemcitabine, and pembrolizumab were changed to 25%, 42%, and 33%, respectively.
Additional limitations were identified, but they were not considered to be a key limitation.
The sponsor obtained the terminal care cost from a published cost-utility analysis of atezolizumab compared with chemotherapy as second-line treatment for metastatic bladder cancer. The cost was presented in 2018 Canadian dollars, but the sponsor did not adjust for inflation to the 2021 currency year as it did for other cost estimates. CADTH also noted that the cost was not specific to bladder cancers and only focused on hospital costs. It is more appropriate to apply a terminal care cost for patients with bladder cancers because the estimate would better reflect specific services and palliative care required for this patient population before death.
CADTH inflated the terminal care cost to 2021 using the Consumer Price Index reported by Statistics Canada in the sponsor’s corrected base case. However, this cost was replaced with a terminal care cost associated with bladder cancers reported by de Oliveira et al. in CADTH’s base case.20 The terminal care costs reported in this study were weighted by the proportions of male and female participants in the CheckMate 274 trial, and the weighted cost was inflated to the 2021 currency year.
Additionally, the following key assumptions were made by the sponsor and have been appraised by CADTH (refer to Table 4).
CADTH Reanalyses of the Economic Evaluation
Base Case Results
CADTH corrected the sponsor’s models by inflating the terminal care cost to the 2021 currency year, removing gemcitabine 200 mg, and using corrected publicly listed prices for cisplatin, carboplatin, and gemcitabine. For both analyses, CADTH's base-case analyses assumed that 50% of patients experiencing disease recurrence required subsequent systemic chemotherapy, changed the distribution of subsequent systemic chemotherapy to align with clinical practice, and used bladder cancer-specific terminal care costs. For the model comparing nivolumab and observation, CADTH’s base case used an alternate survival model (Gompertz) to predict DFS data between years 4 and 5 for nivolumab and observation. Table 5 details the changes made to derive the CADTH's base case, and the summary results of the CADTH base case are presented in Table 6. Additional results are shown in Appendix 4.
Table 4: Key Assumptions of the Submitted Economic Evaluation (Not Noted as Limitations to the Submission)
Sponsor’s key assumption | CADTH comment |
|---|---|
The modelled population is aligned with the Health Canada–approved indication. | Likely acceptable. CADTH obtained clinical expert feedback which agreed with the sponsor that only a very small proportion of patients with urethral primary tumours would not be captured in the modelled population. |
Patient characteristics (i.e., age, sex, weight, body surface area) from the CheckMate 274 trial were representative of a Canadian population. | Acceptable. But the cost-effectiveness results estimated from the efficacy of nivolumab compared to observations derived from the ITT population of the CheckMate 274 trial might not be generalizable to the target patients with MIUC in Canada as the ITT population combined patients who received and did not receive prior neoadjuvant chemotherapy. |
The distribution of first recurrence or death event was assumed to be constant for up to 5 years. | This is an acceptable assumption given that exact timing of death events separated from recurrence events was not available in latest data cut-off of CheckMate 274 due to lack of OS data. However, the implication of this assumption on the cost-effectiveness results is unknown as the OS data are currently unavailable from the CheckMate 274 trial. |
The sponsor assumed the 5-year curative assumption and set the risk of death among patients experiencing recurrent disease to be equal to the age-specific all-cause mortality observed in the Canadian population after year 5. The sponsor’s model did not include disease management costs incurred on patients remaining in the disease-free health state after 5 years. | Clinical experts consulted by CADTH considered this assumption acceptable. |
The sponsor used mean number of doses reported in CheckMate 274 to reflect the duration of nivolumab in the adjuvant setting. The sponsor indicated that mean number of doses is expected to reflect the use of adjuvant nivolumab more accurately than time-to-discontinuation data as some patients were allowed to delay doses. | Acceptable. The clinical experts consulted by CADTH advised that patients with MIUC are expected to receive nivolumab for a maximum of 12 months, but the treatment duration may be prolonged due to treatment delays or interruptions as a result of toxicity. It is therefore reasonable to use a mean number of doses, although the CheckMate 274 trial also reported TTD data. CADTH used TTD data in scenario analysis. |
The exponential distribution was used to predict the transition from recurrent disease to death for both the cisplatin-eligible and ineligible populations. | The sponsor’s model was not flexible enough to test the impact of alternative survival models on the cost-effectiveness findings. However, the choice of survival model was unlikely to have a large impact on an ICER, as the model assumed that the transition from post-recurrence to death is independent of treatment. |
ICER = incremental cost-effectiveness ratio; ITT = intention to treat; MIUC = muscle-invasive urothelial carcinoma; OS = overall survival; TTD = time to discontinuation.
Table 5: CADTH Revisions to the Submitted Economic Evaluation
Stepped analysis | Sponsor’s value or assumption | CADTH value or assumption |
|---|---|---|
Corrections to sponsor’s base case | ||
1. Inflate terminal care cost to a 2021 currency year | $11,691.56 | $12,378.75 |
2. Updated cost per chemotherapya (vial) Cis 50 mg Cis 100 mg Carbo 150 mg Carbo 450 mg Gem 200 mg Gem 1,000 mg Gem 2,000 mg | $9.50 $19.00 $18.80 $56.39 $12.40 $62.00 $124.00 | $135.00 $270.00 $210.00 $599.99 NA a $270.30 $540.60 |
Changes to derive the CADTH base case | ||
1. DFS extrapolation for years 4 and 5 | Nivolumab vs. observation: DFS data for years 4 and 5 for nivolumab and observation arms were derived from the external data source (the deferred chemotherapy arm of Sternberg et al. [2015]). Nivolumab vs. adj. chemo: DFS data for years 4 and 5 for nivolumab and observation arms were derived from the external data source (the immediate chemotherapy arm of the Sternberg et al. [2015]). | Nivolumab vs. observation: A parametric model (Gompertz) was fitted to the DFS data from the CheckMate 274 trial to predict DFS data in years 4 and 5 for nivolumab and observation arms. Nivolumab vs. adj. chemo: No change to the sponsor’s model. |
2. % of patients receiving subsequent systematic chemotherapy | 100% | 50% |
3. Distribution of subsequent therapies | Nivolumab vs. observation Nivolumab: Cis + gem = 65.69% Carbo + gem = 30.91% Pembro = 3.40% Observation: Cis + gem = 59.98% Carbo + gem = 28.22% Pembro = 11.80% Nivolumab vs. adj. chemo Nivolumab: Cis + gem = 65.69% Carbo + gem = 30.91% Pembro = 3.40% Adj. chemo: Cis + gem = 59.98% Carbo + gem = 28.22% Pembro = 11.80% | Nivolumab vs. observation Nivolumab: Cis + gem = 30.91% Carbo + gem = 65.69% Pembro = 3.40% Observation: Cis + gem = 28.22% Carbo + gem = 59.98% Pembro = 11.80% Nivolumab vs. adj. chemo Nivolumab: Cis + gem = 65.69% Carbo + gem = 30.91% Pembro = 3.40% Adj. chemo: Cis + gem = 25.00% Carbo + gem = 42.00% Pembro = 33.00% |
4. End-of-life costs | $11,691.56 | $42,888.41 |
CADTH base case for patients who were ineligible to cisplatin or those who received neoadjuvant therapy | ― | 1 + 2 + 3 + 4 |
CADTH base case for patients who were eligible to cisplatin but did not receive neoadjuvant therapy | ― | 2 + 3 + 4 |
adj. chemo = adjuvant chemotherapy; cis = cisplatin; carbo = carboplatin; DFS = disease-free survival; gem = gemcitabine; NA = not available; pembro = pembrolizumab.
aGem 200 mg does not have a published price in the IQVIA Delta PA database.
Table 6: Summary of the CADTH Reanalysis Results
Drug | Total costs ($) | Total QALYs | ICER vs. reference ($/QALY) | Sequential ICER ($/QALY) |
|---|---|---|---|---|
Patients who were not eligible to receive cisplatin or those who received neoadjuvant therapy | ||||
Sponsor-corrected base case | ||||
Observation | 29,143 | 5.46 | Reference | Reference |
Nivolumab | 104,307 | 6.64 | 63,931 | 63,931 |
CADTH base case | ||||
Observation | 41,550 | 4.95 | Reference | Reference |
Nivolumab | 120,272 | 5.65 | 112,826 | 112,826 |
Patients who were eligible to receive cisplatin but did not receive neoadjuvant therapy | ||||
Sponsor-corrected base case | ||||
Adjuvant chemotherapy | 33,814 | 7.75 | Reference | Reference |
Nivolumab | 104,326 | 6.70 | Dominated | Dominated |
CADTH base case | ||||
Adjuvant chemotherapy | 51,979 | 7.69 | Reference | Reference |
Nivolumab | 118,996 | 6.60 | Dominated | Dominated |
ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year.
Table 7: CADTH Price-Reduction Analyses
Analysis | ICER for nivolumab vs. observation ($/QALY) | |
|---|---|---|
Price reduction | Sponsor-corrected base case | CADTH reanalysis |
No price reduction | 63,931 | 112,826 |
10% | 57,285 | 98,205 |
20% | 50,551 | 89,173 |
21% | 49,848 | 87,284 |
30% | 43,909 | 78,600 |
40% | 37,049 | 66,687 |
50% | 30,241 | 55,343 |
56% | 26,282 | 48,768 |
ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year.
Results from the CADTH base case suggest that, compared to observation, nivolumab was associated with higher costs ($78,722) and improved QALYs (0.70 QALYs), resulting in an ICER of $112,826 per QALY. The estimated ICER was higher than the sponsor’s base case due to a different approach (Gompertz model versus adjustment using PFS data reported by Sternberg et al.)5 for the DFS prediction for years 4 and 5. CADTH notes that due to the model structure, even after the CADTH analysis, nivolumab was associated with an incremental gain of 0.81 life-years compared with observation. This benefit should be interpreted with caution, given the lack of OS data to confirm this modelled benefit. The probability that nivolumab is cost-effective was 0% at the WTP threshold of $50,000 per QALY. Compared to adjuvant chemotherapy, results from the CADTH base case were consistent with the sponsor’s submission, suggesting that nivolumab was dominated by adjuvant chemotherapy because nivolumab was more costly ($67,017) and associated with fewer QALYs (–1.09 QALYs) compared with adjuvant chemotherapy.
Scenario Analysis Results
A series of scenario analysis were conducted based on the CADTH base-case analysis of nivolumab compared to observation. These analyses explored the impact of the following model parameters and assumptions: parametric survival models for DFS predictions, cure time points, alternative assumptions on disease recurrence to death, treatment duration, and dosing assumption of nivolumab. CADTH also explored the impact of health utility values by applying treatment-specific health utility values derived from the CheckMate 274 trial.
Results from scenario analyses (Appendix 4, Table 13) demonstrated that the assumption of the DFS predictions had the largest impact on the cost-effectiveness results, followed by the assumptions of a cure time point and health utility values. ICERs ranged from $90,712 per QALY (Scenario 1, assuming a 4-year cure time point) to $138,516 per QALY (Scenario 4, using KM curves from year 1 to 2 and a Gompertz model to predict DFS of nivolumab and observation from year 4 to 5). The ICERs were also influenced by nivolumab’s dosing assumption. If a weight-based dosing of nivolumab was assumed (Scenario 11), the ICER decreased to $107,395 per QALY. Cost-effectiveness findings were found to be robust to the assumptions and changes in input parameters used to inform the recurred disease health state.
A price-reduction analysis based on the sponsor-corrected and CADTH base case (Table 8) indicated that, at a WTP threshold of $50,000 per QALY, a price reduction of up to 56% is required for adjuvant nivolumab to be considered cost-effective compared to observation. A price-reduction analysis on the sponsor’s base-case analysis of nivolumab versus adjuvant chemotherapy was not reported, given that the nivolumab is less effective and incurred higher non-drug costs than adjuvant chemotherapy based on the available evidence. Even with a 100% price reduction for nivolumab, adjuvant chemotherapy remained the optimal treatment at a $50,000 per QALY WTP threshold.
Issues for Consideration
Drug plans suggested that eligibility for nivolumab should be aligned with inclusion criteria of the CheckMate 274 trial, including the consideration of performance status based on the programmed death-ligand 1 expression level. However, programmed death-ligand 1 testing is not universally available in Canada. Cost-effectiveness results of nivolumab in a programmed death-ligand 1 expression level of 1% or more reported in CADTH’s scenario analysis were exploratory because they were based on results of a subgroup of the CheckMate 274 trial and did not include the cost of programmed death-ligand 1 testing.
Clinical experts consulted by CADTH indicated that patients who experienced disease recurrence after adjuvant nivolumab may receive avelumab or enfortumab. The impact of these targeted therapies on the cost-effectiveness of adjuvant nivolumab remains unknown as they were not considered in the sponsor’s and CADTH’s base-case analyses.
CADTH identified published literature that raises concerns as to whether adjuvant treatment might lead to overtreatment of patients, and whether it will actually lead to improvements in OS when used in all patients when compared with use after the first relapse (i.e., only received by the group of patients that ultimately need treatment).21-23
Overall Conclusions
Evidence from the CheckMate 274 trial indicated that adjuvant treatment with nivolumab demonstrated a clinically meaningful and statistically significant improvement in DFS in patients at high risk of recurrence after radical resection of MIUC when compared to placebo, which was considered a proxy for observation or surveillance (i.e., no active treatment). However, as no OS data from CheckMate 274 are available and no study was identified that investigated a patient-level and or trial-level association with nivolumab in the present target population, it cannot be firmly concluded to what extent the improvements in DFS observed in patients in the nivolumab group of the CheckMate274 trial would translate into OS benefits. While nivolumab appeared to have more adverse events than placebo, the clinical experts consulted by CADTH anticipated that the toxicity profile observed with nivolumab would be more favourable than adjuvant chemotherapy. The relative efficacy of nivolumab and adjuvant chemotherapy is highly uncertain because there is no head-to-head comparison of the 2 treatment options. The sponsor submitted an ITC, but it is associated with uncertainty due to the limited number of included studies, and heterogeneity across the included studies in terms of trial design and eligibility as well as outcome definitions. An additional ITC was identified in the literature, but was associated with similar limitations. The sponsor’s economic evaluation did not use ITC evidence to inform the comparison of nivolumab and chemotherapy, but was based on a naive comparison of efficacy data obtained from the CheckMate 274 trial and the EORTC 30994 study which suggested that nivolumab was not as effective as chemotherapy (or at best, similarly effective). The naive comparison findings were ||||||||||||||||||||||||||||||||||||||||||||||||||, and another published ITC identified in the literature.
CADTH identified several key limitations within the sponsor's economic analysis, specifically the uncertainty associated with long-term survival benefits of nivolumab, the inappropriateness of the sponsor’s 3-health state Markov model to capture treatment pathways and health outcomes of patients with MIUC in the adjuvant setting, the high uncertainty in the comparative efficacy of nivolumab and comparators, and the lack of face validity of the distribution of subsequent treatments in patients with recurrent disease.
CADTH was unable to address all identified limitations but made corrections and several revisions to the sponsor's base case to derive the CADTH base case. CADTH corrected the terminal care cost and used publicly listed prices for cisplatin, carboplatin, and gemcitabine. CADTH's base case assumed 50% of patients experiencing disease recurrence required subsequent systemic chemotherapy, changed the distribution of subsequent systemic chemotherapy to align with clinical practice, and used bladder cancer-specific terminal care costs. For the model comparing nivolumab and observation, CADTH’s base case used an alternate survival model (Gompertz) to predict DFS data for years 4 and 5 for nivolumab and observation. CADTH undertook further scenario analyses to explore the impact of differing parametric approaches for DFS, alternative cure time points, assumptions regarding the treatment duration, and alternative health utility values.
In patients who had received neoadjuvant chemotherapy, or were not able to receive adjuvant chemotherapy, CADTH's base case reported that nivolumab was associated with an ICER of $112,826 per QALY compared with observation; this was higher than the sponsor's base case of $63,931 per QALY. A price reduction of 56% was required to make nivolumab cost-effective compared to observation at the WTP of $50,000 per QALY. Scenario analyses highlighted that the key drivers included assumptions on DFS data prediction, cure time points, and health utility values. In patients who had not received neoadjuvant chemotherapy, and were able to receive adjuvant chemotherapy, results from CADTH’s and the sponsor’s base case were consistent, suggesting that nivolumab was dominated by adjuvant chemotherapy (i.e., nivolumab was more costly and associated with fewer QALYs). This finding was robust to changes in the model’s assumptions and input parameters. Given that nivolumab is less effective and incurred higher non-drug costs than adjuvant chemotherapy based on the available evidence, even with a 100% price reduction for nivolumab, adjuvant chemotherapy remained the optimal treatment at a $50,000 per QALY WTP threshold.
CADTH was unable to fully assess the uncertainty associated with external data used to derive DFS and the transitions from recurrence to death, or the implications of the sponsor’s 3-state model compared with a more appropriate 4-state model which separated locoregional and distant recurrence. CADTH notes that due to the model structure, even with the CADTH reanalysis, nivolumab was associated with an incremental gain of 0.81 life-years compared with observation. Given the uncertainty associated with whether nivolumab will lead to an improvement in OS, alongside the overestimation of relative DFS due to the application of the sponsor's cure assumption, the modelled benefit associated with nivolumab may have been overestimated and should be interpreted with caution. Additional information is required to validate the long-term survival outcomes predicted by the analysis. If OS benefits associated with nivolumab are not realized then the ICER of nivolumab relative to observation will increase.
The economic results align with the available clinical evidence in suggesting that there is some clinical benefit with respect to DFS for nivolumab when compared with observation, and that in patients eligible for treatment, nivolumab may be less effective than adjuvant chemotherapy, although CADTH could not make conclusions regarding the relative efficacy of nivolumab and adjuvant chemotherapy given substantial limitations with the comparative evidence. The submitted clinical evidence is not sufficient to draw conclusions on the long-term clinical benefits of adjuvant nivolumab in patients with MIUC. As such, whether treatment will outweigh the potential harms associated with overtreatment, particularly in patients who would not otherwise be treated, is unknown.
References
1.Pharmacoeconomic evaluation [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Opdivo (nivolumab) [internal sponsor's package]. Saint-Laurent (QC): Bristol-Myers Squibb; 2022 Jul 18.
2.Opdivo nivolumab for injection Intravenous Infusion, 10 mg nivolumab /mL 40 mg and 100 mg single-use vials [product monograph]. Montreal (QC): Bristol-Myers Squibb Canada; 2022 Jun 27.
3.Guidelines for the economic evaluation of health technologies: Canada. 4th ed. Ottawa (ON): CADTH; 2017: https://www.cadth.ca/guidelines-economic-evaluation-health-technologies-canada-4th-edition. Accessed 2022 Jun 20.
4.Bajorin DF, Witjes JA, Gschwend JE, et al. Adjuvant Nivolumab versus Placebo in Muscle-Invasive Urothelial Carcinoma. N Engl J Med. 2021;384(22):2102-2114. PubMed
5.Sternberg CN, Skoneczna I, Kerst JM, et al. Immediate versus deferred chemotherapy after radical cystectomy in patients with pT3-pT4 or N+ M0 urothelial carcinoma of the bladder (EORTC 30994): an intergroup, open-label, randomised phase 3 trial. Lancet Oncol. 2015;16(1):76-86. PubMed
6.Bellmunt J, von der Maase H, Mead GM, et al. Randomized phase III study comparing paclitaxel/cisplatin/gemcitabine and gemcitabine/cisplatin in patients with locally advanced or metastatic urothelial cancer without prior systemic therapy: EORTC Intergroup Study 30987. J Clin Oncol. 2012;30(10):1107-1113. PubMed
7.De Santis M, Bellmunt J, Mead G, et al. Randomized phase II/III trial assessing gemcitabine/carboplatin and methotrexate/carboplatin/vinblastine in patients with advanced urothelial cancer who are unfit for cisplatin-based chemotherapy: EORTC study 30986. J Clin Oncol. 2012;30(2):191-199. PubMed
8.Powles T, Gschwend JE, Loriot Y, et al. Phase 3 KEYNOTE-361 trial: Pembrolizumab (pembro) with or without chemotherapy versus chemotherapy alone in advanced urothelial cancer [meeting abstract]. J Clin Oncol. 2017;35(15_suppl).
9.Galsky MD, Arija JAA, Bamias A, et al. Atezolizumab with or without chemotherapy in metastatic urothelial cancer (IMvigor130): a multicentre, randomised, placebo-controlled phase 3 trial. Lancet. 2020;395(10236):1547-1557. PubMed
10.Nafees B, Stafford M, Gavriel S, Bhalla S, Watkins J. Health state utilities for non small cell lung cancer. Health Qual Life Outcomes. 2008;6:84. PubMed
11.Gaither TW, Selekman R, Kazi DS, Copp HL. Cost-Effectiveness of Screening Ultrasound after a First, Febrile Urinary Tract Infection in Children Age 2-24 Months. J Pediatr. 2020;216:73-81 e71. PubMed
12.Beauchemin C, Letarte N, Mathurin K, Yelle L, Lachaine J. A global economic model to assess the cost-effectiveness of new treatments for advanced breast cancer in Canada. J Med Econ. 2016;19(6):619-629. PubMed
13.CADTH. pan-Canadian Oncology Drug Review. Initial Economic Guidance Report: Nivolumab (Opdivo) for Adjuvant Melanoma. 2019; https://www.cadth.ca/sites/default/files/pcodr/Reviews2019/10147NivolumabMelanoma%28Adjuvant%29_InEGR_NOREDACT-ABBREV_Post_04Jan2019_final.pdf. Accessed 2022 Jun 20.
14.CADTH. pan-Canadian Oncology Drug Review. Final Economic Guidance Report: Nab-paclitaxel (Abraxane) for Pancreatic Cancer. 2014; https://www.cadth.ca/sites/default/files/pcodr/pcodr-abraxane-mpc-fn-egr.pdf. Accessed 2022 Jun 20.
15.CADTH. pan-Canadian Oncology Drug Review. Final Economic Guidance Report: Pembrolizumab (Keytruda) for Nonsquamous Non Small Cell Lung Cancer. 2019; https://www.cadth.ca/sites/default/files/pcodr/Reviews2019/10153PembroNSQ-NSCLC_fnEGR_NOREDACT-ABBREV_Post_31May2019_final.pdf. Accessed 2022 Jun 20.
16.NICE. Bladder cancer: diagnosis and management. 2015; https://www.nice.org.uk/guidance/ng2. Accessed 2022 Jun 20.
17.Parmar A, Richardson M, Coyte PC, Cheng S, Sander B, Chan KKW. A cost-utility analysis of atezolizumab in the second-line treatment of patients with metastatic bladder cancer. Curr Oncol. 2020;27(4):e386-e394. PubMed
18.Galsky MD, Witjes JA, Gschwend JE, et al. Disease-free survival with longer follow-up from the phase 3 CheckMate 274 trial of adjuvant nivolumab in patients who underwent surgery for high-risk muscle-invasive urothelial carcinoma [poster]. J Urol. 2021;207(5S):e183.
19.Song F, Altman DG, Glenny AM, Deeks JJ. Validity of indirect comparison for estimating efficacy of competing interventions: empirical evidence from published meta-analyses. BMJ. 2003;326(7387):472. PubMed
20.de Oliveira C, Pataky R, Bremner KE, Rangrej J, et al. Phase-specific and lifetime costs of cancer care in Ontario, Canada. BMC Cancer. 2016;16, 809. https://bmccancer.biomedcentral.com/articles/10.1186/s12885-016-2835-7#Abs1. Accessed 2022 Apr 9. PubMed
21.Ragusi MAA, van der Velden BHM, van Maaren MC, et al. Population-based estimates of overtreatment with adjuvant systemic therapy in early breast cancer patients with data from the Netherlands and the USA. Breast Cancer Res Treat. 2022;193(1):161-173. PubMed
22.Gyawali B, Prasad V. Making adjuvant therapy decisions with uncertain data. Ann Oncol. 2019;30(3):361-364. PubMed
23.Gyawali B, West HJ. Lessons From ADAURA on Adjuvant Cancer Drug Trials: Evidence, Ethics, and Economics. J Clin Oncol. 2021;39(3):175-177. PubMed
24.Budget Impact Analysis [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Opdivo (nivolumab) [internal sponsor's package]. Saint-Laurent (QC): Bristol-Myers Squibb; 2022 Jul 18.
25.DeltaPA. [Ottawa (ON)]: IQVIA; 2021: https://www.iqvia.com/. Accessed 2022 Mar 30.
26.Cancer Care Ontario. Drug formulary: CRBPGEMC regimen. Gemcitabine-CARBOplatin. 2022; https://www.cancercareontario.ca/en/drugformulary/regimens/monograph/45316#DrugRegimen. Accessed 2022 Mar 30.
27.Cancer Care Ontario. CISPGEMC regimen: Gemcitabine-CISplatin. 2022; https://www.cancercareontario.ca/en/drugformulary/regimens/monograph/54311#DrugRegimen. Accessed 2022 Mar 30.
28.Canadian Cancer Society. Snapshot of incidence, mortality and survival estimates by cancer type. 2020; https://cdn.cancer.ca/-/media/files/research/cancer-statistics/2020-statistics/canadian-cancer-statistics/2020-resources/res-cancerstatistics-canadiancancerstatistics-2020_cancer-specific-stats.pdf?rev=a672053690e9493d921fe73453a749b4&hash=4F194657DA83EFC15ADB92A8E5ACBC5E&_ga=2.225167066.1105687320.1630428214-1396629587.1630428214. Accessed 2022 Jun 20.
29.Statistics Canada. Population estimates, quarterly. 2021.
30.Hutterer GC. Special Issue on Molecular Research Efforts in Urothelial Carcinoma: Summary of Included Topics. Int J Mol Sci. 2019;20(15). PubMed
31.Cancer Care Alberta. Upper tract urothelial tumours. Clinical practice guideline GU-008, version 2. 2021; https://www.albertahealthservices.ca/assets/info/hp/cancer/if-hp-cancer-guide-gu008-upper-tract.pdf. Accessed 2022 Jun 20.
32.Roupret M, Babjuk M, Burger M, et al. European Association of Urology Guidelines on Upper Urinary Tract Urothelial Carcinoma: 2020 Update. Eur Urol. 2021;79(1):62-79. PubMed
33.Birtle A, Johnson M, Chester J, et al. Adjuvant chemotherapy in upper tract urothelial carcinoma (the POUT trial): a phase 3, open-label, randomised controlled trial. Lancet. 2020;395(10232):1268-1277. PubMed
34.Bladder Cancer Canada. Upper tract urothelial carcinoma. 2022; https://bladdercancercanada.org/en/upper-tract/. Accessed 2022 Apr 18.
35.pan-Canadian Oncology Drug Review. Nivolumab (Opdivo) for adjuvant melanoma. 2019; https://www.cadth.ca/opdivo-melanoma-adjuvant-therapy-details. Accessed 2022 Jul 25.
36.CancerCare Manitoba. Regimen reference order – GENU – gemcitabine + CISplatin. 2020 Jun 9.
37.CancerCare Manitoba. Regimen reference order – GENU – gemcitabine + CARBOplatin. 2021; https://www.cancercare.mb.ca/export/sites/default/For-Health-Professionals/.galleries/files/treatment-guidelines-rro-files/regimen-reference-orders/genitourinary/GENU-gemcitabine-CARBOplatin.pdf. Accessed 2022 Jun 20.
38.Schedule of benefits for laboratory services: effective July 1, 2020. Toronto (ON): Ontario Ministry of Health; 2020: https://www.health.gov.on.ca/en/pro/programs/ohip/sob/lab/lab_mn2020.pdf. Accessed 2022 Jun 20.
39.Schedule of benefits for physician services under the Health Insurance Act: effective October 1, 2021. Toronto (ON): Ontario Ministry of Health; 2021: https://www.health.gov.on.ca/en/pro/programs/ohip/sob/physserv/sob_master.pdf. Accessed 2022 Jun 20.
40.Wehler E, Zhao Z, Pinar Bilir S, Munakata J, Barber B. Economic burden of toxicities associated with treating metastatic melanoma in eight countries. Eur J Health Econ. 2017;18(1):49-58. PubMed
41.Canadian Canceer Society. Canadian Cancer Statistics. 2022; https://cancer.ca/en/research/cancer-statistics/canadian-cancer-statistics. Accessed 2022 Apr 11.
42.Krishna SR, Konety BR. Current Concepts in the Management of Muscle Invasive Bladder Cancer. Indian J Surg Oncol. 2017;8(1):74-81. PubMed
43.National Collaborating Centre for Cancer (UK). Bladder Cancer: Diagnosis and Management. 5. Managing muscle-invasive bladder cancer. 2015; https://www.ncbi.nlm.nih.gov/books/NBK356289/. Accessed 2022 Apr 10.
Appendix 1: Cost Comparison Table
Note that this appendix has not been copy-edited.
The comparators presented in the following table have been deemed to be appropriate based on feedback from clinical expert(s) and drug plan. Comparators may be recommended (appropriate) practice or actual practice. Existing Product Listing Agreements are not reflected in the table and as such, the table may not represent the actual costs to public drug plans.
Table 8: CADTH Cost Comparison Table for Muscle-Invasive Urothelial Cancer
Treatment | Strength / concentration | Form | Price per vial ($) | Recommended dosage | Average daily cost ($) | Average 28-day cost ($) | |
|---|---|---|---|---|---|---|---|
Nivolumab (Opdivo) | 10 mg/mL | 4 mL 10 mL Vial IV infusion | 782.2200a 1,955.5600a | 240 mg Q2W 480 mg Q4W | 335.24 | 9,387 | |
Chemotherapy | |||||||
Gemcitabine | 40 mg/mL | 1,000 mg 2,000 mg Vial IV infusion | 270.3000 540.6000 | 1,000 mg/m2 on day 1 and 8 Q3W | 51.49 | 1,442 | |
Carboplatin | 10 mg/mL | 50 mg 150 mg 450 mg 600 mg | 70.0000 210.0000 599.9985 775.0020 | AUC 5 to 6 (600 to 900 mg) on day 1 Q3W | 46.90 | 1,313 | |
Gemcitabine + carboplatin | 98.33 | 2,753 | |||||
Gemcitabine | 40 mg/mL | 1,000 mg 2,000 mg | 270.3000 540.6000 | 1,000 mg/m2 on day 1, 8, and 15 Q4W Alternative dosing: 1,000 mg/m2 on day 1 and 8 Q3W | 57.92 Alternative dosing: 51.49 | 1,622 Alternative dosing: 1,442 | |
Cisplatin | 1 mg/mL | 50 mg 100 mg | 135.0000 270.0000 | 70 mg/m2 on day 1 Q4W Alternative dosing: 70 mg/m2 on day 1 Q3W | 14.46 Alternative dosing: 19.29 | 405 Alternative dosing: 540 | |
Gemcitabine + cisplatin | 72.32 | 2,070 | |||||
Gemcitabine + cisplatin (Alternative dosing) | 70.78 | 1,982 | |||||
AUC = area under the curve; Q3W = every 3 weeks; Q4W = every 4 weeks; Q6W = every 6 weeks.
aSponsor-submitted price.24
Note: Prices of chemotherapies were obtained from the DeltaPA IQVIA database25 (accessed, March 30, 2022), unless otherwise indicated. Cost calculations assume a body surface area of 1.80 m2 where applicable. Wastage of excess medication in vials is included in costs. Recommended dosage is based on Cancer Care Ontario monographs,26,27 unless otherwise indicated. For all target AUC calculations, dose calculations followed guidance from the Cancer Care Ontario product monograph for Carboplatin: Target AUC is 4 to 6. Carboplatin is dosed according to the following formula: Maximum carboplatin dose (mg) = target AUC (mg/mL per min) X (125 + 25); maximum dose is based on a capped GFR estimate at 125 mL/min for patients with normal renal function. Carboplatin is costed for a dose of 750 mg.
Appendix 2: Submission Quality
Note that this appendix has not been copy-edited.
Description | Yes/No | Comments |
|---|---|---|
Population is relevant, with no critical intervention missing, and no relevant outcome missing | No | Refer to CADTH appraisal section. |
Model has been adequately programmed and has sufficient face validity | No | CADTH identified minor errors in the drug cost calculations. The sponsor’s model was not flexible to modify because some calculations, such as transition probabilities and drug costs, were hardcoded and not transparent. See CADTH appraisal section. |
Model structure is adequate for decision problem | No | Refer to CADTH appraisal section. |
Data incorporation into the model has been done adequately (e.g., parameters for probabilistic analysis) | No | Refer to CADTH appraisal section. |
Parameter and structural uncertainty were adequately assessed; analyses were adequate to inform the decision problem | Yes | No comment. |
The submission was well organized and complete; the information was easy to locate (clear and transparent reporting; technical documentation available in enough details) | No | Key information was missing from the report. For example, the sponsor did not justify the reason for using a naive comparison as opposed to a sponsor-commissioned ITC study to represent the comparative efficacy of nivolumab and adjuvant chemotherapy. |
ITC = indirect treatment comparison.
Appendix 3: Additional Information on the Submitted Economic Evaluation
Note that this appendix has not been copy-edited.
Detailed Results of the Sponsor’s Base Case
Figure 2: Predicted DFS Data From Parametric Survival Models (ITT Population) — Nivolumab
Source: Sponsor’s pharmacoeconomic submission.1
Figure 3: Predicted DFS Data From Parametric Survival Models (ITT Population) — Observation
Source: Sponsor’s pharmacoeconomic submission.1
Appendix 4: Additional Details on the CADTH Reanalyses and Sensitivity Analyses of the Economic Evaluation
Note that this appendix has not been copy-edited.
Detailed Results of CADTH Base Case
Table 10: Disaggregated Summary of CADTH’s Economic Evaluation Results (Nivolumab Versus Observation)
Parameter | Nivolumab | Observation | Incremental |
|---|---|---|---|
Discounted LYs | |||
Total | 6.69 | 5.88 | 0.81 |
By health state | |||
Disease-free | 5.91 | 5.07 | 0.84 |
Recurred disease | 0.78 | 0.80 | –0.03 |
Discounted QALYs | |||
Total | 5.65 | 4.95 | 0.70 |
By health state | |||
Disease-free | 5.03 | 4.32 | 0.72 |
Recurred disease | 0.62 | 0.64 | –0.02 |
Discounted costs ($) | |||
Total | 120,272 | 41,550 | 78,722 |
Acquisition | 79,282 | 0 | 79,282 |
Administration | 1,261 | 0 | 1,261 |
Disease management | 7,594 | 7,458 | 136 |
Subsequent treatments | 3,614 | 6,503 | –2,889 |
Adverse events | 466 | 422 | 44 |
Monitoring | 968 | 0 | 968 |
Terminal care | 27,086 | 27,166 | –80 |
ICER ($/QALY) | 112,826 | ||
ICER = incremental cost-effectiveness ratio; LY = life-year; QALY = quality-adjusted life-year.
Table 11: Disaggregated Summary of CADTH’s Economic Evaluation Results (Nivolumab Versus Adjuvant Chemotherapy)
Parameter | Nivolumab | Adjuvant chemotherapy | Incremental |
|---|---|---|---|
Discounted LYs | |||
Total | 7.00 | 8.45 | –1.45 |
By health state | |||
Disease-free | 6.33 | 7.88 | –1.55 |
Recurred disease | 0.67 | 0.57 | 0.10 |
Discounted QALYs | |||
Total | 6.60 | 7.69 | –1.09 |
By health state | |||
Disease-free | 6.07 | 7.33 | –1.26 |
Recurred disease | 0.53 | 0.46 | 0.07 |
Adverse events | –0.00 | –0.10 | 0.10 |
Discounted costs ($) | |||
Total | 118,996 | 51,979 | 61,556 |
Acquisition | 79,361 | 3,428 | 75,933 |
Administration | 1,273 | 299 | 974 |
Disease management | 6,760 | 6,392 | 368 |
Subsequent treatments | 3,126 | 9,295 | –6,169 |
Adverse events | 464 | 5,825 | –5,361 |
Monitoring | 967 | 229 | 738 |
Terminal care | 27,015 | 26,497 | 518 |
ICER ($/QALY) | Nivolumab was dominated (more costly and fewer QALYs). | ||
ICER = incremental cost-effectiveness ratio; LY = life-year; QALY = quality-adjusted life-year.
Table 12: Summary of the Stepped Analysis of the CADTH Reanalysis Results
Stepped analysis | Drug | Total costs ($) | Total QALYs | ICER ($/QALYs) |
|---|---|---|---|---|
Patients who were not eligible to receive cisplatin or those who received neoadjuvant therapy | ||||
Sponsor’s base case | Observation | 25,097 | 5.46 | Ref. |
Nivolumab | 100,458 | 6.64 | 64,046 | |
Sponsor’s corrected base casea | Observation | 29,143 | 5.46 | Ref. |
Nivolumab | 104,307 | 6.64 | 63,931 | |
CADTH reanalysis 1 | Observation | 30,178 | 5.06 | Ref. |
Nivolumab | 106,150 | 5.76 | 108,287 | |
CADTH reanalysis 2 | Observation | 28,746 | 5.46 | Ref. |
Nivolumab | 104,040 | 6.64 | 63,862 | |
CADTH reanalysis 3 | Observation | 24,066 | 5.36 | Ref. |
Nivolumab | 99,441 | 6.55 | 63,525 | |
CADTH reanalysis 4 | Observation | 44,834 | 5.46 | Ref. |
Nivolumab | 119,997 | 6.64 | 63,820 | |
CADTH base case (1+2+3+4) | Observation | 41,550 | 4.95 | Ref. |
Nivolumab | 120,272 | 5.65 | 112,826 | |
Patients who were eligible to receive cisplatin but did not receive neoadjuvant therapy | ||||
Sponsor’s base case | Adjuvant chemotherapy | 28,067 | 7.74 | Ref. |
Nivolumab | 100,409 | 6.68 | Dominated | |
Sponsor’s corrected base casea | Adjuvant chemotherapy | 33,814 | 7.75 | Ref. |
Nivolumab | 104,326 | 6.70 | Dominated | |
CADTH reanalysis 2 | Adjuvant chemotherapy | 28,751 | 7.75 | Ref. |
Nivolumab | 100,387 | 6.69 | Dominated | |
CADTH reanalysis 3 | Adjuvant chemotherapy | 41,976 | 7.68 | Ref. |
Nivolumab | 102,245 | 6.60 | Dominated | |
CADTH reanalysis 4 | Adjuvant chemotherapy | 52,539 | 7.75 | Ref. |
Nivolumab | 123,378 | 6.69 | Dominated | |
CADTH base case (2+3+4) | Adjuvant chemotherapy | 51,979 | 7.69 | Ref. |
Nivolumab | 118,996 | 6.60 | Dominated | |
ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year; Ref. = reference.
agemcitabine 200 mg was removed because this dose does not have a published price in the IQVIA Delta PA database.
Scenario Analyses
CADTH conducted several additional scenario analyses to assess the uncertainty surrounding the model structure and parameters used in the sponsor’s model for nivolumab versus observation. CADTH found that increased ICERs were associated with the longer duration used for a curative assumption, the longer time horizon to use predicted DFS data from parametric survival curves, the longer duration of nivolumab use, and the lower health utility values for disease-free and recurred disease health states (as represented by the application of treatment-specific health utility). By contrast, smaller ICERs were associated with a shorter duration used for a curative assumption, the use of a generalized gamma model to predict DFS data for years 4 and 5 and the use of a weight-based dosing for nivolumab. The ICER reduced substantially if the model focused on patients with PD-L1 ≥1%.
Table 13: Summary of CADTH Scenario Analyses
Drug | Total costs ($) | Total QALYs | ICER ($/QALY) |
|---|---|---|---|
Sponsor's corrected base case | |||
Observation | 29,143 | 5.46 | Reference |
Nivolumab | 104,307 | 6.64 | 63,931 |
CADTH's base case | |||
Observation | 41,550 | 4.95 | Reference |
Nivolumab | 120,272 | 5.65 | 112,826 |
CADTH's scenario analysis 1: Assuming a 4-year cure time point | |||
Observation | 40,839 | 5.20 | Reference |
Nivolumab | 119,420 | 6.07 | 90,712 |
CADTH's scenario analysis 2: Assuming a 6-year cure time point | |||
Observation | 41,940 | 4.82 | Reference |
Nivolumab | 120,773 | 5.45 | 124,417 |
CADTH's scenario analysis 3: Using a generalized gamma model to predict DFS of nivolumab and observation for years 4 and 5 | |||
Observation | 41,950 | 4.82 | Reference |
Nivolumab | 120,503 | 5.69 | 90,781 |
CADTH's scenario analysis 4: Using KM curves from year 1 to 2 and Gompertz model to predict DFS of nivolumab and observation from year 3 to 5 | |||
Observation | 41,210 | 5.05 | Reference |
Nivolumab | 120,065 | 5.62 | 138,516 |
CADTH's scenario analysis 5: Using KM curves from year 1 to 4 and Gompertz model to predict DFS of nivolumab and observation in year 5 | |||
Observation | 41,134 | 5.17 | Reference |
Nivolumab | 120,063 | 5.85 | 115,499 |
CADTH's scenario analysis 6: Applying treatment-specific utility values | |||
Observation | 41,573 | 5.00 | Reference |
Nivolumab | 120,375 | 5.63 | 125,123 |
CADTH's scenario analysis 7: Using a lower bound of treatment effect of subsequent pembrolizumab (HR = 0.57) | |||
Observation | 42,083 | 4.01 | Reference |
Nivolumab | 120,590 | 5.67 | 118,942 |
CADTH's scenario analysis 8: Using a lower bound of treatment effect of subsequent pembrolizumab (HR = 1.18) | |||
Observation | 41,389 | 4.94 | Reference |
Nivolumab | 120,241 | 5.65 | 111,750 |
CADTH's scenario analysis 9: Using TTD to reflect duration of nivolumab | |||
Observation | 42,501 | 4.96 | Reference |
Nivolumab | 123,418 | 5.66 | 115,188 |
CADTH's scenario analysis 10: Applying a weight-based dosing for nivolumab | |||
Observation | 41,719 | 4.95 | Reference |
Nivolumab | 117,137 | 5.65 | 107,395 |
CADTH's scenario analysis 11: Assuming 30% of patients with disease recurrence received subsequent chemotherapy | |||
Observation | 38,801 | 4.95 | Reference |
Nivolumab | 118,692 | 5.66 | 113,947 |
CADTH's scenario analysis 12: Assuming 70% of patients with disease recurrence received subsequent chemotherapy | |||
Observation | 44,103 | 4.95 | Reference |
Nivolumab | 121,708 | 5.64 | 110,889 |
CADTH's scenario analysis 13: Focusing on PDL1 ≥ 1% | |||
Observation | 40,092 | 4.72 | Reference |
Nivolumab | 39,387 | 7.16 | 32,533 |
Appendix 5: Submitted Budget Impact Analysis and CADTH Appraisal
Note that this appendix has not been copy-edited.
Table 14: Summary of Key Take-Aways
Key take-aways of the BIA |
|---|
|
BIA = budget impact analysis; MIUC = muscle-invasive urothelial carcinoma.
Summary of Sponsor’s Budget Impact Analysis
The sponsor submitted a budget impact analysis (BIA),24 assessing the expected budgetary impact of the introduction of nivolumab for the adjuvant therapy in patients with MIUC who are at high risk of recurrence after undergoing radical resection of MIUC. The sponsor’s Health Canada indication was revised upon final NOC; the sponsor indicated that the modelled population is functionally the same as the Health Canada–indicated population.24 The analysis was done from the perspective of a Canadian public drug payer over a 3-year time horizon; the base year was assumed to be 2022 and the 3-year time horizon ran from 2023 to 2025. Key inputs to the BIA are documented in Table 15.
The sponsor estimated population size using an epidemiology-based approach, with data obtained from published literature and Canadian Cancer Society statistics to estimate the number of MIUC patients among bladder cancer and upper tract urothelial cancer patients who would be eligible for nivolumab treatment.28-33 The incidence rate of upper tract urothelial cancer was derived from the incidence rate of bladder cancer based on the assumption that 90% to 95% of urothelial cancer cases arise in the bladder.34 Current standard of care includes observation and platinum-based chemotherapy in combination with gemcitabine. Patients accrued drug acquisition costs in their incident year only, which were obtained from published literature.14,15,35 Subsequent treatments included pembrolizumab, cisplatin-based chemotherapy, and carboplatin-based chemotherapy. Total costs were calculated by multiplying drug cost per administration with the mean number of doses. Mean treatment duration was based on the CheckMate 274 trial for nivolumab and on a maximum of 4 cycles for each chemotherapy regimen, as recommended in the respective product monographs.24,36,37 Dosing regimens of chemotherapies were obtained from Cancer Care Ontario Formulary.36,37 No vial sharing was assumed.
In scenario analyses, the sponsor also included monitoring costs, drug administration costs, and costs of grade 3-4 adverse events. Monitoring costs and drug administration costs were obtained from the Ontario Schedule of Benefits.38,39 The proportion of adverse events (AEs) (rash, constipation, leucocytes decrease, neutrophils, or granulocytes decrease, platelets decrease and hemoglobin decrease) and costs were obtained from CheckMate 274 trial and published literature.5,24,40
Table 15: Summary of Key Model Parameters
Parameter | Sponsor’s estimate, reported as Year 1 / Year 2 / Year 3 if appropriate |
|---|---|
Target population | |
Overall number of bladder cancer patients per 100,000 (overall) | 28.6 (8,700) |
Proportion of patients with muscle-invasive bladder cancer (stage II-IV) | 17.4% |
Proportion of patients receiving radical resection (or surgery) | 60.0% |
Proportion of patients at high risk of recurrence | 75.0% |
Proportion of high-risk patients that can receive adjuvant therapy | 70.0% |
Incidence of urothelial cancer per 100,000 (overall) | 30.9 (9,292) |
Patients with upper tract urothelial cancer | 7.5% |
Patients with muscle-invasive disease (stage II-IV) | 56.0% |
Patients receiving radical resection (or surgery) | 95.0% |
Patients at high risk of recurrence | 100% |
Patients eligible for adjuvant chemotherapy | 70.0% |
Patients receiving neoadjuvant carboplatin-based chemotherapy | 39.7% |
Number of patients eligible for drug under review (Year 1 / Year 2 / Year 3) | 581 / 588 / 595 |
Number of bladder cancer patients eligible for drug under review (Year 1 / Year 2 / Year 3) | 477 / 483 / 489 |
Number of urothelial cancer patients eligible for drug under review (Year 1 / Year 2 / Year 3) | 104 / 106 / 107 |
Market uptake (3 years) | |
Uptake (reference scenario): Observation Cisplatin-based chemotherapy Carboplatin-based chemotherapy | 80.0% / 85.0% / 90.0% 16.0% / 12.0% / 8.0% 4.0% / 3.0% / 2.0% |
Uptake (new drug scenario): Nivolumab Observation Cisplatin-based chemotherapy Carboplatin-based chemotherapy | 35.0% / 55.0% / 55.0% 55.0% / 37.5% / 40.0% 8.0% / 6.0% / 4.0% 2.0% / 1.5% / 1.0% |
Cost of treatment (per patient) | |
Cost of treatment over cycle: Nivolumab Observation Cisplatin-based chemotherapy Carboplatin-based chemotherapy | $4,693 $0 $350 $572 |
Nivolumab Observation Cisplatin-based chemotherapy Carboplatin-based chemotherapy | $114 $0 $119 $117 |
Cost of administration39 | |
Drug administration by intravenous infusion: Nivolumab Observation Cisplatin-based chemotherapy Carboplatin-based chemotherapy | $75 $0 $75 $75 |
Cost per adverse events40 | |
Diarrhea Fatigue Pruritus Urinary tract infection Nausea Rash Constipation Leucocytes decrease Neutrophils or granulocytes decrease Platelets decrease Hemoglobin decrease | $4,889 $6,055 $3,697 $5,273 $4,126 $3,500 $4,887 $8,730 $8,730 $6,055 $6,055 |
Summary of the Sponsor’s BIA Results
From the Canadian drug plan perspective, the sponsor estimated the net 3-year budget impact of introducing nivolumab for adjuvant treatment of muscle-invasive urothelial carcinoma patients who are at high risk of recurrence after undergoing radical resection of MIUC to be $66,718,914 (Year 1: $15,873,877; Year 2: $25,265,503; Year 3: $25,579,534). Similar 3-year budget impact was estimated from the Canadian health care payer perspective including the costs of monitoring and AEs.
CADTH Appraisal of the Sponsor’s BIA
CADTH identified several key limitations to the sponsor’s analysis that have notable implications on the results of the BIA:
Correct treatment costs of gemcitabine, cisplatin, and carboplatin: The sponsor leveraged published literature to obtain unit prices of gemcitabine, cisplatin, and carboplatin.14,15,35 CADTH obtained the unit prices of these components using IQVIA Delta PA database and updated outdated costs to reflect changes in prices.25 The sponsor based treatment cost of gemcitabine on 200 mg vial. However, the prices for 200 mg vial have ended and there are no current prices for this vial size in the IQVIA Delta PA database. CADTH based treatment costs on the combination of vials that have a current price and that lead to the least costly treatment costs.
CADTH corrected the unit prices of gemcitabine, cisplatin, and carboplatin to the most recent prices and adopted least costly cost per treatment.
The number of eligible patients is underestimated: The sponsor used outdated data to derive the incidence rate of bladder cancer. The most recent report by the Canadian Cancer Statistics estimates a higher number of bladder cancer cases than the sponsor’s estimates for base year.41 The clinical experts consulted for this review by CADTH also noted the number of eligible patients is underestimated in the sponsor’s submission. The sponsor estimated the number of patients eligible for nivolumab treatment using an epidemiologic approach, with inputs based on assumptions and data from published literature, which are associated with uncertainty. The sponsor estimated that 17.4% of patients with bladder cancer patients have muscle-invasive disease based on internal data. Based published literature, the proportion of bladder cancer cases progressing to muscle-invasive disease may range from 20% to 25%.42,43 The sponsor also assumed 70% of bladder cancer patients at high risk of recurrence can receive adjuvant therapy. However, the clinical experts noted nivolumab has a theoretical advantage that it may be given to patients with lower performance status than required for chemotherapy. As such, the assumed proportion of patients that can receive adjuvant therapy and the number of patients who would be eligible to receive nivolumab is likely underestimated.
The sponsor restricted the number of eligible patients derived from upper tract urothelial cancer population to those who received prior neoadjuvant carboplatin-based chemotherapy. CADTH was unable to verify the proportion of patients receiving prior neoadjuvant carboplatin-based chemotherapy in the sponsor’s original source, which considers carboplatin-based chemotherapy in the adjuvant setting (i.e., within 90 days after nephroureterectomy) in patients with locally advanced upper tract urothelial carcinoma.33 The clinical experts noted that immunotherapy is very attractive to both patients and physicians as an alternative to chemotherapy, and if reimbursed, would replace adjuvant chemotherapy. The sponsor underestimated the number of eligible patients by restricting the upper tract urothelial cancer population to a subgroup of patients who received prior neoadjuvant carboplatin-based chemotherapy.
The sponsor’s decision to estimating the budget impact based on disease area limits an appropriate analysis of patients who are eligible or ineligible for adjuvant chemotherapy. The BIA model derived the total eligible population based on estimated number of patients with bladder cancer and upper tract urothelial cancer who develop muscle-invasive disease, and included an estimate of prior neoadjuvant chemotherapy for solely the upper tract urothelial cancer population. As such, the model structure limits the feasibility of estimating the budget impact in a subgroup of MIUC population who are ineligible for adjuvant chemotherapy and have no current treatment option other than surveillance. A more appropriate approach would have been to sum the budget impact estimated in subgroups, and this added flexibility would have allowed CADTH to validate input paraments that are relevant to each subgroup. In its current form, the estimated budget impact is associated with substantial uncertainty.
CADTH attempted to derive the number of patients who may be ineligible for adjuvant chemotherapy using the randomized population in the CheckMate 274 trial, which found |||||||% of patients to be ineligible for adjuvant chemotherapy because they had received neo-adjuvant therapy (|||||||%) or were ineligible due to comorbidities (|||||||%). The budget impact is, then, explored in an exploratory analysis for this subpopulation that is ineligible for adjuvant chemotherapy who would otherwise be under surveillance.
As noted earlier, the sponsor assumed that the submitted population (adjuvant treatment of patients with MIUC who are at high risk of recurrence after undergoing radical resection of MIUC) represents the Health Canada–approved indication (adjuvant treatment of adult patients with urothelial carcinoma [UC] who are at high risk of recurrence after undergoing radical resection of UC). The sponsor indicated that UCs can be located in the lower (bladder and urethra) or the upper (renal pelvis and ureter) urinary tract, and that bladder tumours account for 90 to 95% of UCs. The sponsor noted that upper tract urothelial carcinomas are uncommon and account for only 5 to 10% of UCs, and that tumours on the urethra, part of UC but not MIUC, are exceedingly rare, and estimated that patients with urethra primary tumours represent <1% of all UC patients. This aligns with clinical expert feedback, but may suggest the estimated population is slightly underestimated.
In CADTH reanalysis, the number of bladder cancer cases in base year (2022) was based on the 2022 Canadian Cancer Statistics report.41 Further, the proportion of high-risk bladder cancer patients who can receive adjuvant therapy was increased to 90% and 100% of upper tract urothelial cancer patients eligible for adjuvant chemotherapy were included in estimated eligible population based on feedback from clinical experts.
CADTH explored the impact of assuming 25% of bladder cancer patients have muscle-invasive disease in a scenario analysis.
In an exploratory analysis, CADTH explored the budget impact in the subgroup ineligible for adjuvant chemotherapy, assuming |||||% of MIUC population is ineligible for adjuvant chemotherapy. All patients in this subgroup are assumed to be on surveillance (market share of observation in the reference scenario is 100%).
Market share of nivolumab is underestimated: The sponsor assumed nivolumab has a market share of 55% by year 3. The clinical experts consulted for this review by CADTH anticipate a higher market share of nivolumab that may be in the range of 70% by year 1 and 90% by year 3. The clinical experts noted nivolumab uptake would be higher and faster than the sponsor’s estimation because both patients and clinicians are very attuned to immunotherapy; many would prefer immunotherapy over adjuvant chemotherapy and nivolumab is expected to add a treatment option where current standard of care is surveillance. Should nivolumab be reimbursed by public drug plans, the market share of nivolumab, and therefore budget, impact would be higher than estimated by the sponsor.
In CADTH reanalysis, nivolumab has a market share of 70% in year 1, 80% in year 2 and 90% in year 3 based on feedback from clinical experts. The reimbursement of nivolumab displaced the market share of comparators proportionally to sponsor’s assumptions on market share displacement.
CADTH Reanalyses of the BIA
CADTH corrected the sponsor’s base case by updating unit prices of gemcitabine, cisplatin, and carboplatin, and adopting least costly treatment cost per cycle. CADTH revised the sponsor’s base case by updating number of bladder cancer cases based on the 2022 Canadian Cancer Statistics report, assuming 90% of bladder cancer patients at high risk of recurrence can receive adjuvant therapy, assuming 100% of upper tract urothelial cancer patients eligible for adjuvant therapy are included in estimated population size and increasing the market share of nivolumab based on feedback from clinical experts.
Table 16: CADTH Revisions to the Submitted BIA
Stepped analysis | Sponsor’s value or assumption | CADTH value or assumption |
|---|---|---|
Corrections to sponsor’s base case | ||
1. Update unit prices | Gemcitabine: 200 mg: $12.40 1,000 mg: $62.00 2,000 mg: $124.00 Carboplatin: 50 mg: $0.00 150 mg: $18.80 450 mg: $56.39 600 mg: $0.00 Cisplatin: 50 mg: $9.50 100 mg: $19.00 | Gemcitabine: 200 mg: NAa 1,000 mg: $270.30 2,000 mg: $540.60 Carboplatin: 50 mg: $70.00 150 mg: $210.00 450 mg: $600.00 600 mg: $775.00 Cisplatin: 50 mg: $135.000 100 mg: $270.00 |
2. Number of vials | Gemcitabine: 200 mg: 4 1,000 mg: 1 Carboplatin: 150 mg: 2 450 mg: 1 600 mg: 0 | Gemcitabine: 200 mg: 0 1,000 mg: 2 Carboplatin: 150 mg: 1 450 mg: 0 600 mg: 1 |
Changes to derive the CADTH base case | ||
1. Incidence of MIUC bladder cancer | Incidence rate of bladder cancer is based on outdated data number of patients with urothelial cancer is derived from | Incidence rate of bladder cancer is based on data from 2022 Canadian Cancer Statistics report. |
2. Proportion of high-risk patients that can receive adjuvant therapy (bladder cancer patients) | 70% | 90% |
3. Proportion of patients eligible for adjuvant chemotherapy who received neoadjuvant carboplatin-based chemotherapy (urothelial cancer patients) | 39.7% | 100% |
4. Market share of nivolumab and comparators (year 1 / year 2 / year 3) | Nivolumab: 35.0% / 55.0% / 55.0% Observation: 55.0% / 37.5% / 40.0% Cisplatin-based Chemotherapy: 8.00% / 6.00% / 4.00% Carboplatin-based Chemotherapy: 2.00% / 1.50% / 1.00% | Nivolumab: 70.0% / 80.0% / 90.0% Observation: 25.4% / 16.7% / 8.89% Cisplatin-based Chemotherapy: 3.69% / 2.67% / 0.89% Carboplatin-based Chemotherapy: 0.92% / 0.67% / 0.22% |
CADTH base case | Reanalysis 1 + 2 + 3 + 4 | |
BIA = budget impact analysis.
aGemcitabine 200 mg does not have a published price in the IQVIA Delta PA database.
In the CADTH reanalysis, the 3-year budget impact of reimbursing nivolumab from the public drug plan perspective for the adjuvant therapy in patients with MIUC who are at high risk of recurrence after undergoing radical resection of MIUC increased to $180,672,898 (Year 1: $51,773,325; Year 2: $60,173,636; Year 3: $68,725,937).
The results of the CADTH step-wise reanalysis are presented in summary format in Table 17 and a more detailed breakdown is presented in Table 18.
Table 17: Summary of the CADTH Reanalyses of the BIA
Stepped analysis | Three-year total ($) |
|---|---|
Submitted base case | 66,718,914 |
CADTH correction 1 | 66,339,436 |
CADTH correction 2 | 66,726,938 |
Submitted base case, corrected | 66,206,984 |
CADTH reanalysis 1 | 72,456,672 |
CADTH reanalysis 2 | 81,728,600 |
CADTH reanalysis 3 | 84,253,430 |
CADTH reanalysis 4 | 109,547,264 |
CADTH base case | 180,672,898 |
BIA = budget impact analysis.
CADTH also conducted additional scenario analyses to address remaining uncertainty, using the CADTH base case. Results are provided in Table 18. The scenario analysis involved:
Assuming 25% of bladder cancer patients have muscle-invasive disease.
Price reduction of 56% for nivolumab, the price reduction at which nivolumab would be considered cost-effective at a WTP of $50,000 per QALY in the CADTH base-case reanalysis of the cost-utility analysis (Table 7).
CADTH conducted an exploratory analysis to estimate the budget impact in the subgroup of MIUC patients who are ineligible for adjuvant chemotherapy, assuming |||||||% of MIUC population is ineligible for adjuvant chemotherapy. All patients are assumed to be on surveillance (market share of observation in the refence scenario is 100% for year 1, 2, and 3).
Table 18: Detailed Breakdown of the CADTH Reanalyses of the BIA
Stepped analysis | Scenario | Year 0 ($) (current situation) | Year 1 ($) | Year 2 ($) | Year 3 ($) | Three-year total ($) |
|---|---|---|---|---|---|---|
Submitted base case | Reference | 2,322,928 | 2,351,262 | 2,370,161 | 2,389,211 | 7,110,634 |
New drug | 2,322,928 | 18,225,139 | 27,635,663 | 27,968,745 | 73,829,548 | |
Budget impact | 0 | 15,873,877 | 25,265,503 | 25,579,534 | 66,718,914 | |
CADTH base case | Reference | 5,602,162 | 5,670,791 | 5,506,881 | 5,338,188 | 16,515,860 |
New drug | 5,602,162 | 57,444,116 | 65,680,517 | 74,064,125 | 197,188,758 | |
Budget impact | 0 | 51,773,325 | 60,173,636 | 68,725,937 | 180,672,898 | |
CADTH scenario analysis: 25% of bladder cancer patients have muscle-invasive disease | Reference | 7,315,123 | 7,200,433 | 6,927,586 | 6,653,305 | 20,781,324 |
New drug | 7,315,123 | 72,939,117 | 82,625,251 | 92,310,573 | 247,874,941 | |
Budget impact | 0 | 65,738,684 | 75,697,665 | 85,657,268 | 227,093,617 | |
CADTH scenario analysis: nivolumab price reduction of 56% | Reference | 5,602,162 | 5,670,791 | 5,506,881 | 5,338,188 | 16,515,860 |
New drug | 5,602,162 | 27,644,154 | 31,205,603 | 34,803,363 | 93,653,120 | |
Budget impact | 0 | 21,973,362 | 25,698,722 | 29,465,175 | 77,137,259 | |
CADTH exploratory analysis: Subgroup of MIUC population who are ineligible for adjuvant chemotherapy | Reference | 3,048,973 | 3,086,325 | 3,124,188 | 3,162,570 | 9,373,082 |
New drug | 3,048,973 | 37,200,863 | 42,590,538 | 48,107,686 | 127,899,087 | |
Budget impact | 0 | 34,114,538 | 39,466,350 | 44,945,116 | 118,526,005 |
BIA = budget impact analysis.
Stakeholder Input
Patient Input
Bladder Cancer Canada
About Bladder Cancer Canada
Bladder Cancer Canada (BCC) was formed in 2009 by two bladder cancer survivors who found that there was no one to talk to about their treatments, experiences and fears. Today, BCC is a registered national charity and the only organization in Canada serving those facing a bladder cancer diagnosis. Our objectives are to help bladder cancer patients and their support teams address the day-to-day issues of this disease; to increase awareness of bladder cancer among the general public and medical community; and to fund research which pursues the diagnosis, treatment and elimination of bladder cancer. www.bladdercancercanada.org
Information Gathering
Bladder Cancer Canada collected the information for this submission from online surveys and one-to-one interviews with patients.
Online surveys were conducted between February 16 and March 15, 2022. The survey asked questions about the impact of muscle-invasive urothelial carcinoma (MIUC) on the lives of patients, the effect of current treatments and the patient experience with Opdivo. BCC found it very difficult to identify patients with MIUC and Opdivo treatment experience. Potential respondents were identified through messages to the BCC mailing list. Messages were posted on Facebook and Twitter as well as the Cancer Survivors Network online discussion boards. Investigators from the CheckMate 274 clinical trial were also asked to relay the survey to patients with the appropriate experience.
A total of 7 patients completed the survey: 5 in English and 2 in French. 6 of these respondents were from Canada (representing Alberta, British Columbia, Ontario & Quebec) and 1 was from the United States. All of the respondents have been diagnosed with muscle-invasive urothelial carcinoma and 2 have treatment experience with Opdivo, 1 as adjuvant treatment following radical resection.
The online patient survey asked respondents if they would be willing to participate in an interview to elaborate on their responses. 2 patients were interviewed by telephone. Both have treatment experience with Opdivo.
Disease Experience
One respondent was diagnosed in 2021 and 2 were diagnosed in 2020; the other four respondents were diagnosed in 2017 or earlier.
Two respondents are currently receiving neoadjuvant treatment, 3 have had a pathological complete response, 1 indicated that they were receiving immunotherapy and 1 indicated that no other treatment was possible following surgery; the latter two respondents did not elaborate on these replies.
Experiences With Currently Available Treatments
Six respondents provided information about the treatments they have undergone since diagnosis.
Table 1: Treatments Received For Bladder Cancer
Treatments received | Number of treatments |
|---|---|
Radical cystectomy | 6 |
Cisplatin | 3 |
Gemcitabine | 3 |
Transurethal resection of bladder cancer | 2 |
MVAC (Methotrexate + Vinblastine + Adriamycin + Cisplatin) | 1 |
Experimental antibody drug conjugates (unspecified) | 1 |
Fatigue was the most commonly reported side effect of these treatments, followed by nausea and constipation. Fatigue was also identified as the most-difficult-to-tolerate side effect of treatment.
Two respondents required hospitalization due to the side effects associated with treatment.
Improved Outcomes
Bladder Cancer Canada asked patients to evaluate the importance of different outcomes for bladder cancer treatment on a scale of 1 (not important) to 5 (very important). All of these outcomes were rated as important with preventing recurrence receiving the highest score. However, it should be noted that managing side effects received the lowest score by an important margin, suggesting a willingness by patients to tolerate side effects for treatments that can support the other outcomes on this list.
Table 2: Importance of Outcome For Bladder Cancer Treatment
Importance of outcome | Average (n=6) |
|---|---|
Preventing recurrence | 5.00 |
Controlling disease progression | 4.67 |
Reducing symptoms | 4.67 |
Maintaining quality of life | 4.67 |
Managing side effects | 4.00 |
Respondents were also asked if they would be willing to tolerate new side effects from drugs that can control disease progression or prevent recurrence. On a scale of 1 (will not tolerate side effects) to 10 (will tolerate significant side effects), respondents gave an average score of 8.14 supporting the conclusion that patient values will tolerate side effects for progress towards other desired outcomes.
Comments include:
The cure is the number one thing or failing that – control disease.
Les effets secondaires sont plus faciles à vivre que de ne plus vivre avec ses proches ou d’avoir à passer à travers un traitement de chimio.
Experience With Drug Under Review
Two respondents had MIUC treatment experience with Opdivo.
Respondent A was treated with Opdivo for 6-12 months following a radical resection. He had a stage 4 tumour prior to his operation. He also received neoadjuvant cisplatin-based chemotherapy which did not work. His MIUC was diagnosed in 2016. He completed his course of treatment in 2017. He currently has no cancer cells.
Respondent B was treated with Opdivo for 1-2 years. Opdivo was prescribed off-label in combination with ipilimumab. He was previously treated with a radical resection and studer neobladder, followed by cisplatin-based adjuvant therapy, pembrolizumab, unspecified antibody drug conjugates accessed through clinical trials, and radiotherapy. His MIUC was diagnosed before 2015. He completed his course of treatment in 2019. He is currently in remission, but was subsequently treated with radiotherapy for abdominal lymph nodes. He also suffers from ICI-related interstitial lung disease that may be related to his cancer treatment.
Treatment
Patients were asked to rate how their life had changed on Opdivo compared to other therapies they had received on a scale of 1 (much worse) to 5 (much better).
Table 3: Changes Experienced When Taking Opdivo Compared to Other Therapies
Change on Opdivo | Respondent A | Respondent B |
|---|---|---|
Controlling disease progression | 5 | 5 |
Preventing recurrence | 5 | 4 |
Cancer symptoms | 5 | n/a |
Drug side effects | 5 | 2 |
Quality of life | 5 | 2 |
Comments include:
C’était le jour et la nuit avec la chimio. La chimio enlève presque le goût de vivre, c’est vraiment invasif, tandis qu’avec ce traitement, tu n’es pas immobilisé comme avec la chimio. Là, tu peux continuer à vivre presque normalement, malgré la fatigue. – Respondent A
Mon traitement hémîunotherapie à très bien été. - Respondent A
Side Effects
Respondent A experienced itchy skin (pruritus) and fatigue on Opdivo. These were treated with antihistamines and cortisone. Respondent B experienced diarrhea, joint swelling, colitis and pneumonitis. Respondent B’s respirologist believes that his ICI-related interstitial lung disease was caused by cancer immunotherapy. However, he did not indicate whether he believes this was the result of ipilimumab, Opdivo or both.
When asked to rate how much they could tolerate the side effects associated with Opdivo on a scale of 1 (completely intolerable) to 10 (completely tolerable), Respondent A gave a 10 and Respondent B gave an 8.
Comments include:
Les effets secondaires se contrôlaient mieux [avec Opdivo] et étaient plus faciles à vivre. – Respondent A
Helped with cancer, but side effects are challenging – Respondent B
Je n’avais pas de symptômes, j’ai juste eu des démangeaisons sur la peau quelques mois après les traitements. – Respondent A
Companion Diagnostic Test
Not applicable
Anything Else?
Respondents A and B both said that they would recommend Opdivo to other patients with MIUC.
Comments include:
Si le traitement peut guérir comme il l’a guéri sans chimio, c’est parfait. – Respondent A
Selon moi, c’est un très bon médicament. – Respondent A
For me, it was effective. – Respondent B
Patient Group Conflict of Interest Declaration
To maintain the objectivity and credibility of the CADTH reimbursement review process, all participants in the drug review processes must disclose any real, potential, or perceived conflicts of interest. This Patient Group Conflict of Interest Declaration is required for participation. Declarations made do not negate or preclude the use of the patient group input. CADTH may contact your group with further questions, as needed.
Did you receive help from outside your patient group to complete this submission? If yes, please detail the help and who provided it.
||||||||||||||||||||||||||||||, an independent consultant, prepared this submission with the assistance and oversight of Bladder Cancer Canada staff.
Did you receive help from outside your patient group to collect or analyze data used in this submission? If yes, please detail the help and who provided it.
||||||||||||||||||||||||||||||, an independent consultant, created the clinician surveys, oversaw survey distribution and collection, and analyzed the data for this submission with the assistance and oversight of Bladder Cancer Canada staff.
List any companies or organizations that have provided your group with financial payment over the past 2 years AND who may have direct or indirect interest in the drug under review.
Table 4: Financial Disclosures for Bladder Cancer Canada
Company | $0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 |
|---|---|---|---|---|
No COI | — | — | — | — |
Clinician Input
Bladder Cancer Canada
About Bladder Cancer Canada
Bladder Cancer Canada (BCC) was formed in 2009 by two bladder cancer survivors who found that there was no one to talk to about their treatments, experiences and fears. Today, BCC is a registered national charity and the only organization in Canada serving those facing a bladder cancer diagnosis. Our objectives are to help bladder cancer patients and their support teams address the day-to-day issues of this disease; to increase awareness of bladder cancer among the general public and medical community; and to fund research which pursues the diagnosis, treatment and elimination of bladder cancer. www.bladdercancercanada.org
Information Gathering
Bladder Cancer Canada collected the information for this submission from online surveys conducted between February 26 and March 11, 2022. The survey asked clinicians about their experience treating muscle-invasive urothelial carcinoma (MIUC) patients with nivolumab and other therapies. It also asked questions about their goals for MIUC treatment, current unmet needs and the potential role of nivolumab within the MIUC treatment paradigm. Potential respondents were identified from Caretaker 274 clinical trial investigators and the members of the BCC medical advisory board.
A total of 6 clinicians completed the survey. All 6 respondents were from Canada (representing Alberta, British Columbia, Ontario & Quebec). All respondents had experience treating patients with MIUC and 3 had experience treating MIUC patients with nivolumab following radical resection.
Current Treatments
The current standard of care for MIUC following radical resection is adjuvant cisplatin/gemcitabine-based chemotherapy for patients that were eligible but did not receive neoadjuvant chemotherapy (approximately 15% of patients). There is no alternative treatment for patients who received neoadjuvant chemotherapy, or patients that are unfit or ineligible for adjuvant chemotherapy. No off-label treatments were identified, and special access programs were limited to nivolumab. Several respondents identified nivolumab as the standard of care when it was available.
When asked for their impression of current MIUC treatments, one clinician gave this response:
Adjuvant gem/cis delays progress. Have observed some surprisingly favourable outcomes. But many patients recover poorly from surgery and are not fit for adjuvant chemo.
Treatment Goals
Bladder Cancer Canada asked clinicians to evaluate the importance of different outcomes for bladder cancer treatment on a scale of 1 (not important) to 5 (very important). Medical outcomes like preventing metastases, increasing overall survival and controlling disease progression were rated more highly than quality of life outcomes like reducing severity of symptoms and minimizing adverse events.
Table 5: Importance of Outcomes for Bladder Treatment
Importance of outcome | Average (n=6) |
|---|---|
Increasing overall survival | 4.83 |
Preventing metastases | 4.83 |
Controlling disease progression | 4.50 |
Maintaining quality of life | 4.00 |
Minimizing adverse events | 3.33 |
Reducing severity of symptoms | 3.33 |
Treatment Gaps (Unmet Needs)
Considering the treatment goals, please describe goals (needs) that are not being met by currently available treatments.
There is an unmet need for patients who receive neoadjuvant chemotherapy, but still have significant residual disease at the time of radical resection. These patients are at very high risk of disease recurrence, metastatic disease and death. A substantial percentage of patients will also relapse under the current standard of care, especially those with lymph node involvement at resection. For patients who are ineligible for adjuvant chemotherapy, there is no alternative treatment.
Some respondents also indicated that they lacked robust evidence for the use of adjuvant chemotherapy following radical resection.
Which patients have the greatest unmet need for an intervention such as the drug under review?
Clinicians identified patients who are cisplatin-ineligible and patients with significant residual disease following resection as having the greatest unmet need; more specifically, patients who have pT2+ or N+ at the time of resection.
Place in Therapy
How would the drug under review fit into the current treatment paradigm?
Nivolumab would be used as adjuvant therapy following radical resection for patients with a high risk of recurrence with or without neoadjuvant cisplatin-based chemotherapy, or for patients who are unfit or ineligible for adjuvant cisplatin-based chemotherapy and did not receive neoadjuvant chemotherapy. This would be an additional option for patients. There are currently no recommended treatments for high-risk patients after neoadjuvant chemotherapy.
Please indicate whether or not it would be appropriate to recommend that patients try other treatments before initiating treatment with the drug under review. Please provide a rationale from your perspective.
No other treatment would be recommended for patients who fit the indications identified in 6.1. There are currently no recommended treatments for high-risk patients after neoadjuvant chemotherapy or patients who are ineligible for adjuvant chemotherapy.
How would this drug affect the sequencing of therapies for the target condition?
Nivolumab would fill a gap in the current standard of care. As such, it would not affect the sequencing of therapies for MIUC.
Which patients would be best suited for treatment with the drug under review?
The patients best suited for treatment with nivolumab are all those eligible for the CheckMate 274 clinical trial (multiple clinicians presented their response in these terms).
How would patients best suited for treatment with the drug under review be identified?
Patients would be identified based on post-operative pathology reports of cystectomy specimens.
Which patients would be least suitable for treatment with the drug under review?
Patients who responded well to neoadjuvant chemotherapy and patients with contraindications for immunotherapy would be least suitable for treatment with nivolumab.
Is it possible to identify those patients who are most likely to exhibit a response to treatment with the drug under review?
PDL1+ patients may have enhanced outcomes, but this is not adequate for making treatment decisions and is not done in practice. Upper tract urothelial carcinoma would be prioritized for adjuvant chemotherapy. Patients with prior neoadjuvant chemotherapy appear to respond best.
What outcomes are used to determine whether a patient is responding to treatment in clinical practice?
Survival time and time to recurrence/metastatic disease would be the outcome used to determine whether patients are responding to treatment with nivolumab.
What would be considered a clinically meaningful response to treatment?
All 6 respondents identified delay of recurrence, delay of metastases and prolonged survival as clinically meaningful treatment responses. 5 respondents also included prevention of recurrence and prevention of survival as clinically meaningful responses.
How often should treatment response be assessed?
Five respondents, including the 3 respondents with nivolumab treatment experience, said that treatment response should be assessed every three months. The sixth respondent said that treatment should be assessed every six months.
What factors should be considered when deciding to discontinue treatment?
All six respondents said that adverse events should be factor when deciding to discontinue treatment. However, it should also be noted that the adverse effects of nivolumab in the adjuvant setting were in line with the profile of nivolumab in other cancer settings. 5 respondents said recurrence of disease should be a factor; the lone dissenter did not indicate why he disagreed. Two respondents said burden of treatment should also be a factor.
What settings are appropriate for treatment with the drug under review?
A majority of respondents said that hospital outpatient clinics and private infusion clinics were the appropriate settings for treatment with nivolumab. A smaller number of respondents also said that medical clinics and CLSCs would be an appropriate.
For non-oncology drugs, is a specialist required to diagnose, treat, and monitor patients who might receive the drug under review?
Not applicable.
Additional Information
When asked if they would recommend use of nivolumab for patients with MIUC following radical resection based on clinical evidence of disease-free survival benefits in the absence of overall survival data, all 6 respondents said that they would.
Comments include:
Progression means a lot to patients - this is a clinically relevant endpoint; and the treatment is well tolerated
A helpful option for patient who have received neoadjuvant chemo and an alternative for patients unfit for adjuvant chemotherapy
Conflict of Interest Declarations
To maintain the objectivity and credibility of the CADTH drug review programs, all participants in the drug review processes must disclose any real, potential, or perceived conflicts of interest. This conflict of interest declaration is required for participation. Declarations made do not negate or preclude the use of the clinician group input. CADTH may contact your group with further questions, as needed. Please see the Procedures for CADTH Drug Reimbursement Reviews (section 6.3) for further details.
Did you receive help from outside your clinician group to complete this submission? If yes, please detail the help and who provided it.
||||||||||||||||||||||||||||||, an independent consultant, prepared this submission with the assistance and oversight of Bladder Cancer Canada staff.
Did you receive help from outside your clinician group to collect or analyze any information used in this submission? If yes, please detail the help and who provided it.
||||||||||||||||||||||||||||||, an independent consultant, created the clinician surveys, oversaw survey distribution and collection, and analyzed the data for this submission with the assistance and oversight of Bladder Cancer Canada staff.
List any companies or organizations that have provided your group with financial payment over the past two years AND who may have direct or indirect interest in the drug under review. Please note that this is required for each clinician who contributed to the input — please add more tables as needed (copy and paste). It is preferred for all declarations to be included in a single document.
Declaration for Clinician 1
Name: Michel Pavic
Position: Oncologue médical
Date: 26-02-2022
Table 6: Conflict of Interest Declaration for Bladder Cancer Canada Clinician 1
Company | Check appropriate dollar range | |||
|---|---|---|---|---|
$0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 | |
BMS | X | — | — | — |
EMD Serono | X | — | — | — |
Merck | X | — | — | — |
Declaration for Clinician 2
Name: Wassim Kassouf
Position: Professor of Urology
Date: 04-03-2022
Table 7: Conflict of Interest Declaration for Bladder Cancer Canada Clinician 2
Company | Check appropriate dollar range | |||
|---|---|---|---|---|
$0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 | |
BMS – ad board | X | — | — | — |
Declaration for Clinician 3
Name: Peter Black
Position: Urologic Oncologist
Date: 08-03-2022
Table 8: Conflict of Interest Declaration for Bladder Cancer Canada Clinician 3
Company | Check appropriate dollar range | |||
|---|---|---|---|---|
$0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 | |
BMS | X | — | — | — |
GNE | X | — | — | — |
EMD-Serono | X | — | — | — |
Merck | X | — | — | — |
Pfizer | X | — | — | — |
Janssen | X | — | — | — |
Declaration for Clinician 4
Name: Aly-Khan Lalani
Position: Medical Oncologist
Date: 08-03-2022
Table 9: Conflict of Interest Declaration for Bladder Cancer Canada Clinician 4
Company | Check appropriate dollar range | |||
|---|---|---|---|---|
$0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 | |
AbbVie | X | — | — | — |
Astellas | X | — | — | — |
BMS | X | — | — | — |
Eisai | X | — | — | — |
Ipsen | X | — | — | — |
Janssen | X | — | — | — |
Merck | X | — | — | — |
Novartis | X | — | — | — |
Pfizer | X | — | — | — |
Roche | X | — | — | — |
TerSera | X | — | — | — |
Declaration for Clinician 5
Name: Ramy Saleh
Position: Med Onc staff
Date: 08-03-2022
Table 10: Conflict of Interest Declaration for Bladder Cancer Canada Clinician 5
Company | Check appropriate dollar range | |||
|---|---|---|---|---|
$0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 | |
No COI | — | — | — | — |
Declaration for Clinician 6
Name: Nimira Alimohamed
Position: Medical Oncology
Date: 11-03-2022
Table 11: Conflict of Interest Declaration for Bladder Cancer Canada Clinician 6
Company | Check appropriate dollar range | |||
|---|---|---|---|---|
$0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 | |
Pfizer | X | — | — | — |
EMD Serono | X | — | — | — |
Seagen | X | — | — | — |
Ontario Health Cancer Care Ontario GU Cancer Drug Advisory Committee
About Ontario Health Cancer Care Ontario GU Cancer Drug Advisory Committee
OH-CCO’s Drug Advisory Committees provide timely evidence-based clinical and health system guidance on drug-related issues in support of CCO’s mandate, including the Provincial Drug Reimbursement Programs (PDRP) and the Systemic Treatment Program.
Information Gathering
Discussed jointly via email
Current Treatments
Current treatment is adjuvant (post-operative) or neo-adjuvant (pre-operative) platinum-based chemotherapy but this treatment is often not given as patients have trouble tolerating it after surgery (too toxic or renal function impaired). Nivolumab can be given to patients with a much lower eGFR than platinum-based chemotherapy and patients don’t need as a high a performance status as that needed for chemo. Furthermore, many randomized trials of adjuvant platinum-based chemotherapy have been negative and there is still debate about the value and effectiveness of this approach. Thus, the vast majority of eligible patients are simply observed postoperatively. Lastly, if a patient receives neoadjuvant chemotherapy, they are not given adjuvant chemotherapy. Nivolumab would provide a different mechanism of action in the adjuvant setting to complement neoadjuvant chemotherapy.
Treatment Goals
Improved overall survival is the main goal. PFS is a good surrogate of OS in urothelial carcinoma (Referencing this paper- https://pubmed.ncbi.nlm.nih.gov/30196910/)
Treatment Gaps (Unmet Needs)
Considering the treatment goals in Section 4, please describe goals (needs) that are not being met by currently available treatments.
Poor use and eligibility of adjuvant chemotherapies in this space
Which patients have the greatest unmet need for an intervention such as the drug under review?
All urothelial carcinoma patients with ypT2 or higher or pT3 or higher or node positive would be the affected population, which constitutes about 2/3 of cystectomy/nephroureterectomy patients. These patients are often frail or have a solitary kidney and thus cannot receive the current standard of adjuvant chemotherapy. Nivolumab would address a significant unmet need in this population of patients.
Place in Therapy
How would the drug under review fit into the current treatment paradigm?
Nivolumab will change the paradigm for how muscle invasive urothelial carcinoma will be treated as many patients will now be eligible for a therapy that is more tolerable than platinum-based chemotherapy. We suspect that this will be the main drug used in the adjuvant setting for patients as it targets a different mechanism of action compared to neoadjuvant chemotherapy. The fact that many patients in the study received neoadjuvant chemotherapy followed by surgery and then adjuvant nivolumab with benefit points to importance of harnessing a different mechanism of action.
Please indicate whether or not it would be appropriate to recommend that patients try other treatments before initiating treatment with the drug under review. Please provide a rationale from your perspective.
If a patient received neoadjuvant chemotherapy or is platinum-ineligible in the adjuvant setting, there is no other therapy in the adjuvant setting. They cannot receive another therapy.
How would this drug affect the sequencing of therapies for the target condition?
If nivolumab fails early, patients may then move on to enfortumab vedotin. A late failure may result in platinum-based chemotherapy or immune-checkpoint rechallenge depending on how long it has been since the patient received their adjuvant nivolumab. This treatment has the potential to move up the sequencing of treatments in advanced urothelial carcinoma.
Which patients would be best suited for treatment with the drug under review?
Patients with ypT2 or higher or pT3/T4 or N+ who received neoadjuvant chemo or who were unfit for platinum-based chemotherapy would be ideally suited for adjuvant nivolumab.
How would patients best suited for treatment with the drug under review be identified?
Patients would be identified based on their postoperative pathology. This would provide definitive pathology and easily identify candidates for treatment.
Which patients would be least suitable for treatment with the drug under review?
Non-muscle invasive resected urothelial carcinoma. It is possible that those eligible for cisplatin-based adjuvant chemotherapy may be better suited to chemotherapy. We do not know whether nivolumab is better than cisplatin-based chemotherapy as Checkmate 274 did not compare the two. Furthermore, the adjuvant chemotherapry RCTs (e.g. Sternberg et al) only demonstrated a PFS benefit as well so we can’t currently say that one treatment impacts OS more than the other. Long term data may show that nivolumab has an OS benefit but we await these results.
Is it possible to identify those patients who are most likely to exhibit a response to treatment with the drug under review?
The primary endpoint of CM274 was PFS improvement in all comers, independent of PDL1 positivity, and the primary endpoint was met. Although subgroup analyses suggest that patients with higher PDL1 positivity may benefit more strongly to nivolumab, this should not be used as a decision to use/fund given that improvements were observe in the whole population.
What outcomes are used to determine whether a patient is responding to treatment in clinical practice?
Yes, in an adjuvant trial, PFS is a reasonable outcome given the high correlation with OS in urothelial carcinoma. Almost all other trials in this space have used PFS as the main outcome measure.
What would be considered a clinically meaningful response to treatment?
A clinically meaningful response would be an improvement in PFS. In CM274, a 30% improvement in the likelihood of recurrence was found, which is statistically significant and clinically meaningful. Furthermore, an absolute improvement of 16% in patients who were free of recurrence at 1-year was seen, translating to a number needed to treat (NNT) of 6 to prevent one recurrence at a year, which we feel is clinically very important.
How often should treatment response be assessed?
Patients are typically followed every 3 to 6 months with axial imaging and bloodwork after cystectomy or nephroureterectomy
What factors should be considered when deciding to discontinue treatment?
Severity and recurrence of immune-related side effects.
What settings are appropriate for treatment with the drug under review?
Any centre capable of providing IV immune checkpoint inhibitors and with experience managing immune-related side effects of nivolumab.
For non-oncology drugs, is a specialist required to diagnose, treat, and monitor patients who might receive the drug under review?
N/A
Additional Information
N/A
Conflict of Interest Declarations
To maintain the objectivity and credibility of the CADTH drug review programs, all participants in the drug review processes must disclose any real, potential, or perceived conflicts of interest. This conflict of interest declaration is required for participation. Declarations made do not negate or preclude the use of the clinician group input. CADTH may contact your group with further questions, as needed. Please see the Procedures for CADTH Drug Reimbursement Reviews (section 6.3) for further details.
Did you receive help from outside your clinician group to complete this submission? If yes, please detail the help and who provided it.
OH-CCO provided secretariat support to the DAC in completing this input.
Did you receive help from outside your clinician group to collect or analyze any information used in this submission? If yes, please detail the help and who provided it.
No
List any companies or organizations that have provided your group with financial payment over the past two years AND who may have direct or indirect interest in the drug under review. Please note that this is required for each clinician who contributed to the input — please add more tables as needed (copy and paste). It is preferred for all declarations to be included in a single document.
Declaration for Clinician 1
Name: Dr. Girish Kulkarni
Position: Genitourinary Drug Advisory Committee Lead
Date: 09/12/2021
Table 12: Conflict of Interest Declaration for Ontario Health Cancer Care Ontario GU Cancer Drug Advisory Committee Clinician 1
Company | Check appropriate dollar range | |||
|---|---|---|---|---|
$0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 | |
No COI | — | — | — | — |
Declaration for Clinician 2
Name: Dr. Aly-Khan Lalani
Position: Genitourinary Drug Advisory Committee Member
Date: 09/12/2021
Table 13: Conflict of Interest Declaration for Ontario Health Cancer Care Ontario GU Cancer Drug Advisory Committee Clinician 2
Company | Check appropriate dollar range | |||
|---|---|---|---|---|
$0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 | |
Merck | X | — | — | — |
Eisai | X | — | — | — |
Declaration for Clinician 3
Name: Dr. Sebastien Hotte
Position: Genitourinary Drug Advisory Committee Member
Date: 09/12/2021
Table 14: Conflict of Interest Declaration for Ontario Health Cancer Care Ontario GU Cancer Drug Advisory Committee Clinician 3
Company | Check appropriate dollar range | |||
|---|---|---|---|---|
$0 to $5,000 | $5,001 to $10,000 | $10,001 to $50,000 | In excess of $50,000 | |
Merck | X | — | — | — |
Eisai | — | X | — | — |
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up-to-date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
Redactions: Confidential information in this document may be redacted at the request of the sponsor in accordance with the CADTH Drug Reimbursement Review Confidentiality Guidelines
Stakeholder Input: The views expressed in each submission are those of the submitting organization or individual; not necessarily the views of CADTH or of other organizations. As such, they are independent of CADTH and do not necessarily represent or reflect the view of CADTH. No endorsement by CADTH is intended or should be inferred. By filing with CADTH, the submitting organization or individual agrees to the full disclosure of the information. CADTH does not edit the content of the submissions.
CADTH does use reasonable care to prevent disclosure of personal information in posted material; however, it is ultimately the submitter’s responsibility to ensure no identifying personal information or personal health information is included in the submission. The name of the submitting organization or individual and all conflict of interest information are included in the submission; however, the name of the author, including the name of an individual patient or caregiver submitting the patient input, are not posted.
Accessibility: CADTH is committed to treating people with disabilities in a way that respects their dignity and independence, supports them in accessing material in a timely manner, and provides a robust feedback process to support continuous improvement. All materials prepared by CADTH are available in an accessible format. Where materials provided to CADTH by a submitting organization or individual are not available in an accessible format, CADTH will provide a summary document upon request. More details on CADTH’s accessibility policies can be found here.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.