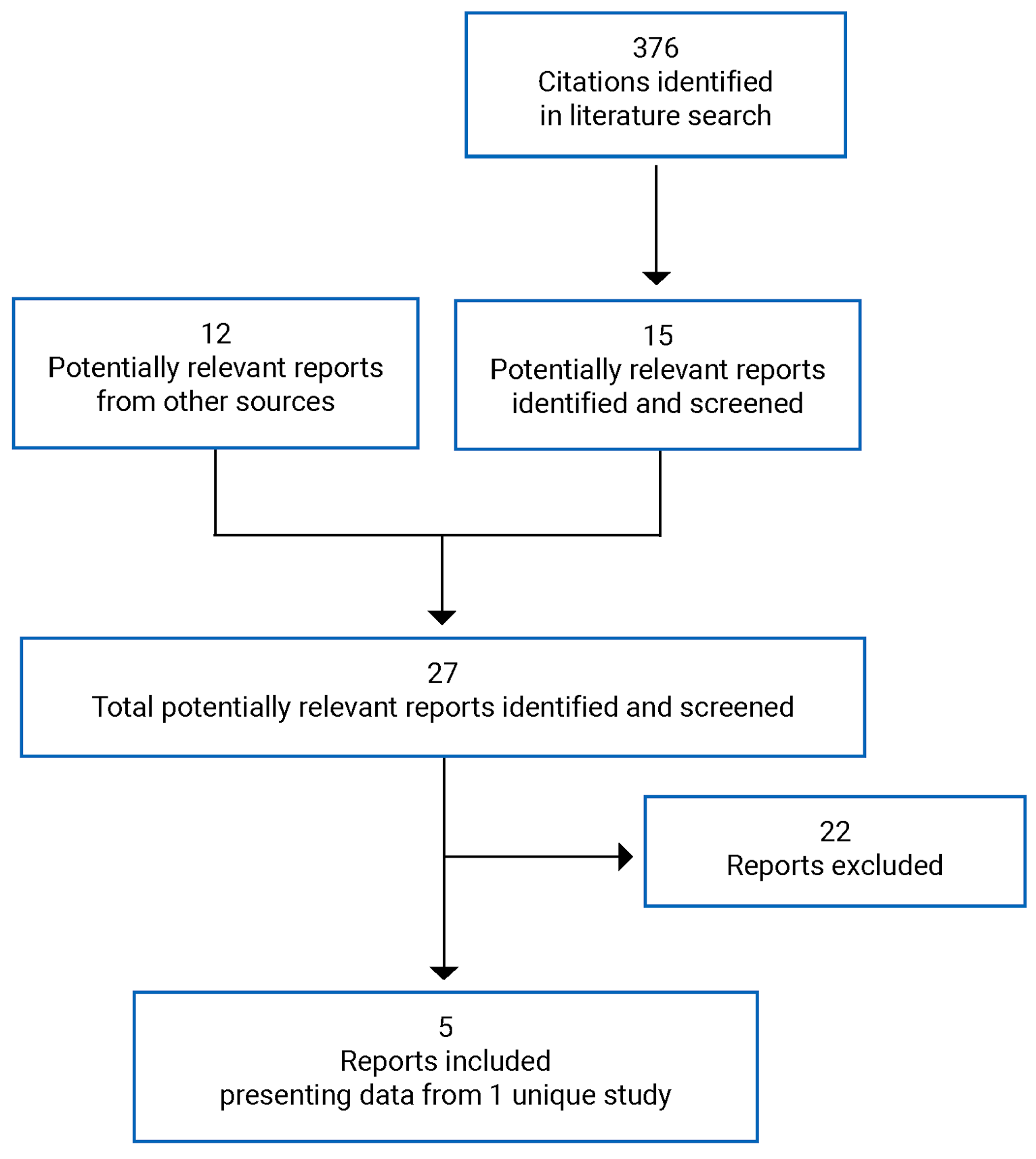
CADTH Reimbursement Review
Mogamulizumab (Poteligeo)
Sponsor: Kyowa Kirin Canada, Inc.
Therapeutic area: Mycosis fungoides, Sézary syndrome
This multi-part report includes:
Clinical Review
Pharmacoeconomic Review
Stakeholder Input
Clinical Review
Abbreviations
AE
adverse event
BOR
best overall response
CCR4
CC chemokine receptor 4
CI
confidence interval
CR
complete response
CTCL
cutaneous T-cell lymphoma
DOR
duration of response
ECOG
Eastern Cooperative Oncology Group
ECP
extracorporeal photopheresis
EORTC
European Organisation for Research and Treatment of Cancer
EQ-5D-3L
3-level EQ-5D
FACT-G
Functional Assessment of Cancer Therapy–General
HDAC
histone deacetylase
HR
hazard ratio
HRQoL
health-related quality of life
IPCW
inverse probability censoring weighting
IRR
infusion-related reaction
ISCL
International Society for Cutaneous Lymphomas
ITT
intention to treat
LCT
large-cell transformation
MAIC
matching-adjusted indirect comparison
MF
mycosis fungoides
mSWAT
modified Severity-Weighted Assessment Tool
NE
not estimable
NOC
Notice of Compliance
ORR
overall response rate
OS
overall survival
PD
progressive disease
PFS
progression-free survival
PR
partial response
PUVA
psoralen plus UVA light therapy
QoL
quality of life
RCT
randomized controlled trial
RPSFT
rank-preserving structural failure time
SAE
serious adverse event
SAP
statistical analysis plan
SD
standard deviation
SOC
system organ class
SS
Sézary syndrome
TEAE
treatment-emergent adverse event
TSEBT
total skin electron beam therapy
TTF
time-to-treatment failure
TTR
time to response
VAS
Visual Analogue Scale
Executive Summary
An overview of the submission details for the drug under review is provided in Table 1.
Item | Description |
|---|---|
Drug product | Mogamulizumab (Poteligeo), 20 mg/5 mL single-use vial IV infusion, 4 mg/mL |
Indication | For the treatment of adult patients with relapsed or refractory MF or SS after at least 1 prior systemic therapy |
Reimbursement request | As per indication |
Health Canada approval status | NOC |
Health Canada review pathway | Standard |
NOC date | June 2, 2022 |
Sponsor | Kyowa Kirin Canada, Inc. |
MF = mycosis fungoides; NOC = Notice of Compliance; SS = Sézary syndrome.
Introduction
Mycosis fungoides (MF) and Sézary syndrome (SS) are the 2 most common types of cutaneous T-cell lymphoma (CTCL), accounting for approximately one-half to two-thirds of CTCL cases (SS accounts for only 2% to 3% of all CTCLs).1-5 The incidence of MF increases with age and is approximately 5.6 cases per 1 million people.4-8 Clinically, SS affects mostly patients older than 60 years of age and is associated with a poor prognosis.7,9 From 1992 through 2010 in Canada, there were 2,510 documented cases of MF (representing 37.5% of CTCL cases), and 110 cases of SS (representing 1.6% of CTCL cases).10
Both MF and SS result from infiltration of malignant T-lymphocytes, preferentially to skin. MF is usually associated with an indolent clinical course and intermittent, stable, or slow progression, while SS is a rare leukemic subtype of CTCL characterized by a more aggressive course of disease and shorter survival.11 MF may be asymptomatic or manifest with symptoms of significant itch, pain, or burning of the skin. The clinical presentation of MF is highly variable, ranging from non-specific erythematous scaly patches to thin plaques or papulonodular eruptions, appearing mostly on non–sun-exposed areas.12 In approximately 30% of cases, the patches or plaques evolve into tumours.13 Patients with SS present with a triad of skin redness (erythroderma), enlarged lymph nodes (lymphadenopathy), and a large number of circulating atypical lymphocytes (Sézary cells) in the skin, lymph nodes, and peripheral blood.12,14 The psychological aspects of this incurable cancer affecting the skin present a significant burden to patients.15
As MF and SS can resemble other inflammatory dermatoses, diagnosis can be challenging, sometimes taking upward of 2 to 6 years.16-21 Diagnosis of MF and SS is confirmed by skin biopsy and blood tests, though characteristic histologic features of MF may be absent in early disease.22 Secondary infections are a common complication related to an impaired skin barrier23,24 and a dysregulated immune system due to the lymphoma itself and some immunosuppressive treatments.
Both MF and SS are incurable diseases; however, if discovered early, the prognosis for patients with early-stage MF can be very good.25 Clinical staging of patients with MF is essential, not only for assessment of prognosis but also to guide decisions in the management of the disease. For patients with early-stage disease with limited skin involvement (stage IA or IB to IIA), skin-directed therapies are used, including topicals, phototherapy, and radiation therapy. Systemic therapy is recommended for advanced-stage disease (stage ≥ IIB). In advanced MF and SS, first-line treatment consists of oral retinoid therapy, with or without interferon, or single-agent chemotherapy, including methotrexate, gemcitabine, or liposomal doxorubicin. Second- and third-line treatment for MF consist of previously unused single-agent chemotherapy (anti-cancer) therapy, histone deacetylase (HDAC) inhibitors, or immunotherapies, such as brentuximab vedotin (in patients with CD30-positive disease). In SS, or for patients with blood involvement, extracorporeal photopheresis (ECP), with or without retinoids and interferon, may be used in either the first- or second-line setting; otherwise, eligible treatment options beyond the first-line setting remain the same as in MF.
Mogamulizumab has received a Health Canada Notice of Compliance for the treatment of adult patients with relapsed or refractory MF or SS after at least 1 prior systemic therapy. Mogamulizumab is a defucosylated, humanized immunoglobulin G1 (IgG1) kappa monoclonal antibody that binds to CC chemokine receptor 4 (CCR4), a G protein–coupled receptor for CC chemokines that is involved in the trafficking of lymphocytes to various organs and is expressed on the surface of some T-cell malignancies and on regulatory T cells and a subset of T helper 2 T cells.26
The objective of the current review is to review the beneficial and harmful effects of mogamulizumab 1.0 mg/kg for the treatment of adults with relapsed or refractory MF or SS after at least 1 prior systemic therapy.
Stakeholder Perspectives
The information in this section is a summary of input provided by the patient and clinician groups that responded to CADTH’s call for patient input and from the clinical experts consulted by CADTH for the purpose of this review.
Patient Input
Between March 8, 2021, and September 21, 2021, Lymphoma Canada, in collaboration with the Canadian Skin Patient Alliance and Cutaneous Lymphoma Foundation, conducted an anonymous online survey of CTCL patients, primarily patients with MF or SS. A total of 449 patients who responded to the survey were diagnosed with either MF or SS; of these, 46 (14%) were living in Canada. The majority of respondents were aged 60 years or older (69%) and more than half were female (54%).
MF can be difficult to diagnose, with symptoms often occurring for many years before a diagnosis is made. It can masquerade as other more common skin conditions, such as eczema or psoriasis. The majority of patients participating in this survey were diagnosed between 1 and 5 years ago (41%), with a portion of patients diagnosed more than 10 years ago (26%). Only 23% of patients had their condition correctly diagnosed as MF or SS at presentation. Symptoms of MF or SS that most impacted patients’ quality of life (QoL) at diagnosis included visible patches or lesions (78%) and itchiness of skin or skin lesions (57%). Symptoms that most commonly affected respondents’ current QoL were similar to those at diagnosis, with the stress of diagnosis plus the fear of and worry about disease progression as well as anxiety impacting their current well-being.
A total of 327 patients provided information about their experience with CTCL treatments for MF and SS. Of the 211 patients who received systemic treatments, 27% received interferon, 26% received methotrexate, 24% received oral bexarotene, 23% received mogamulizumab, and 10% were treated with romidepsin. According to the patients who responded, the most common side effects they experienced during MF or SS CTCL treatments included fatigue (41%), skin pain or burning (34%), skin irritation or rash (33%), and moderate or severe itching (30%). A total of 27% of survey respondents indicated that the number of clinic visits had the most significant impact on their QoL, while 21% indicated that treatment-related fatigue had the most significant impact on their QoL, and cited additional negative impacts due to treatment on their ability to work (21%), travel (21%), and have intimate relationships (19%).
Access to treatment within the patient’s community is an important consideration, as certain treatments may be available only at specific tertiary cancer centres. A subanalysis of patients living in Canada revealed that 32% of them could not access treatment locally. Most patients responding to this survey were living in the US, where more treatment options are approved for use, including mogamulizumab; however, 81% of all respondents cited the importance of having an increased number of treatment options available. Patients indicated that longer survival (82%) and better QoL (76%) are extremely important outcomes for new therapies, followed by longer treatment-free periods (70%), easier treatment application (68%), and fewer side effects (64%).
Of all patients who responded to the survey, 52 (12%) had experience with mogamulizumab, accessed largely through private insurance (42%), public drug programs (29%), compassionate access programs (17%), or clinical trials (8%). At the time of the survey, 44% were still receiving treatment, 12% of patients had “completed their full course of mogamulizumab” [sic], 12% had discontinued due to side effects, and 10% did not have their symptoms controlled with mogamulizumab. Only 2 patients had their disease progress during treatment, while 25% were in remission. Of the 52 respondents, a total of 36% of patients had all of their symptoms managed by mogamulizumab, with major symptom management in skin itchiness (62%), red skin patches or rash (56%), and skin pain (25%). When asked to describe their experience with mogamulizumab, 69% of patients indicated they had a good to excellent experience with the therapy.
Clinician Input
Input From the Clinical Experts Consulted by CADTH
According to advice obtained from a clinical expert consulted by CADTH for this review, treatment selection in Canada is often guided by which drugs are funded and available rather than the one with the best chances of response and fewest side effects. They noted that most currently available treatment options demonstrate suboptimal response rates (< 30% to < 50%) or responses of limited duration (< 4 months), and acknowledged that these responses are primarily in the context of skin; however, when used in SS, they are in the context of blood involvement, as well. Per the clinical experts, in Canada, systemic therapy is generally limited to interferon, isotretinoin (off-label use for MF), or oral methotrexate. Bexarotene was noted by the experts as a retinoid that is superior to isotretinoin; however, it is not approved by Health Canada. Other options include local radiotherapy, ECP, chemotherapy (gemcitabine or liposomal doxorubicin), vorinostat, romidepsin, and pralatrexate, though many of these are not funded in Canada. Brentuximab was recently approved for use in CD30-expressing MF, limited or advanced stage, that has relapsed following one of these systemic approaches. The experts stated that some patients derive benefit from existing therapies before ultimately progressing and requiring an alternative, thus agreed that other systemic treatments, including interferon or retinoids with or without ECP (as available), should be attempted before mogamulizumab, based on availability.
The experts noted that diagnosis often takes upward of 3 years, with misdiagnosis occurring frequently early on. There are no pre-symptomatic tests to identify patients best suited for treatment with mogamulizumab. According to the experts, the patients most suitable for mogamulizumab are those with advanced-stage MF (stage IIIB, IVA, IVB) with or without blood involvement, or SS that has failed front-line systemic therapy. Conversely, the clinical experts indicated that patients with stage IA MF should not be treated with mogamulizumab, as they were not included in the pivotal clinical trial, and patients with stage IB, IIA, or IIB are less likely to derive benefit (as evidenced by the response outcomes demonstrated in the study), possibly due to the mechanism of action of mogamulizumab. Overall, the clinical experts felt that aside from patients with stage IA, who were not eligible for the MAVORIC trial, funding criteria should not include staging information, as the primary outcome of the MAVORIC trial demonstrated improved progression-free survival (PFS) across all included stages (IB to IV), but the study was not powered to detect differences by stage.
The experts expressed that the goals of treatment for MF and SS consist of prolonged survival, improved response rate, improvement in skin-related symptoms, and QoL. They noted that these outcomes are reflective of what is measured in clinical trials, as assessed using existing defined response criteria for MF and SS in any of each disease compartment (skin, lymph nodes, and blood). These also include criteria to define progressive disease (PD), and they noted that intolerable AEs, notably mogamulizumab-associated rash that does not respond to management algorithms or other AEs, would be reason to discontinue.
Clinician Group Input
The clinician group input was provided by 2 clinician groups: a network of Canadian cutaneous lymphoma providers and the Ontario Health (Cancer Care Ontario) Hematology Cancer Drug Advisory Committee. The clinician groups noted the individualized approach to treatment as well as the lack of a defined standard of care in MF or SS. Along with suboptimal response rates and limited duration with current treatments, access issues were cited as a major unmet need in this population. The clinician groups noted the lack of publicly funded treatments in Canada and that treatment selection is guided more by which drugs are funded and available rather than necessarily the one with the best chances of response and fewest side effects. The clinician groups expressed that patients should be offered mogamulizumab if they meet the eligibility criteria from the clinical trial, with 1 clinician group highlighting its use in advanced-stage MF, with or without blood involvement, and Sézary syndrome, and that mogamulizumab would likely be used in the second line following chemotherapy, brentuximab vedotin (if CD30-positive), or interferon. One clinician group considered the potential for mogamulizumab’s concurrent use with ECP, particularly for patients with SS. Important goals of treatment, as well as factors considered clinically meaningful to treatment outcomes, include disease control via a reduction in the frequency or severity of symptoms, improvement in symptoms, or stabilization of disease, prolonged survival, and improvement in QoL. Lastly, the clinician groups noted that mogamulizumab would be received in an outpatient setting, and highlighted that disease progression, AEs, and treatment-related toxicity should be considered when deciding to discontinue treatment.
Drug Program Input
The Provincial Advisory Group identified the following jurisdictional implementation issues: relevant comparators, considerations for initiation of therapy, considerations for prescribing of therapy, generalizability, funding algorithm, care provision issues, and system and economic issues. The clinical experts consulted by CADTH weighed evidence from the MAVORIC trial and used their clinical expertise to provide responses to the Provincial Advisory Group’s implementation questions. Refer to Table 5 for more details.
Clinical Evidence
Pivotal Studies and Protocol Selected Studies
Description of Studies
One study (MAVORIC) was included in the review. MAVORIC is an open-label, multi-centre, 1-way crossover, phase III randomized controlled trial (RCT) evaluating the effectiveness of mogamulizumab compared with vorinostat in patients with CTCL whose disease failed to respond to at least 1 prior course of systemic therapy. Patients in the MAVORIC trial were required to have a histologically confirmed diagnosis of MF or SS; stage IB, IIA, IIB, III, or IV disease; and an Eastern Cooperative Oncology Group (ECOG) Performance Status of 0 or 1. A total of 372 patients were randomized 1:1 to either mogamulizumab 1.0 mg/kg IV infusion on days 1, 8, 15, and 22 of the first cycle, and on days 1 and 15 of subsequent cycles (n = 186), or vorinostat 400 mg by mouth once daily on day 1 of each 28-day cycle (n = 186). The primary outcome of the MAVORIC trial was PFS, with key secondary end points of overall response rate (ORR) and health-related quality of life (HRQoL) as assessed by the Skindex-29, Functional Assessment of Cancer Therapy–General (FACT-G), and the 3-level EQ-5D questionnaire (EQ-5D-3L). Other secondary outcomes included best overall response (BOR) and duration of response (DOR). Overall survival (OS) was an exploratory outcome of the MAVORIC trial. MAVORIC was conducted in 11 countries; however, no Canadian investigative sites were included. The clinical data cut-off for the final analysis of the MAVORIC study was December 31, 2016. No interim analyses were conducted.
Demographic and baseline characteristics of the MAVORIC trial were well balanced: the median age was 64 years, 58.1% were male, and 69.9% were White. Most patients had an ECOG Performance Status of 0 (56.5%). Most patients had MF (54.8%), with the remaining patients diagnosed with SS (45.2%). The population in the MAVORIC trial was heavily pre-treated, with a median of 3 prior systemic therapies in each treatment arm.
Efficacy Results
ORR was higher in the mogamulizumab group compared with the vorinostat group; 52 patients (28.0%) treated with mogamulizumab achieved an ORR compared with 9 (4.8%) with vorinostat.27 The results of subgroup and post hoc analyses also suggested that mogamulizumab provided superior response rates in patients with SS (37.0% versus 2.3%),27 advanced-stage disease (30.0% versus 2.9%),28 and B2 blood involvement (37.4% versus 3.2%).29 Additionally, 83 patients (66.9%) treated with mogamulizumab versus 23 patients (18.4%) treated with vorinostat achieved an ORR in the blood compartment, and 78 (41.9%) versus 29 (15.6%) patients achieved an ORR in the skin compartment.27
At the time of the data cut-off (December 31, 2016), the median efficacy follow-up was 17.0 months. The median PFS was 7.70 months (95% confidence interval [CI], 5.67 to 10.33) in the mogamulizumab group, compared with 3.10 months (95% CI, 2.87 to 4.07) in the vorinostat group (P < 0.0001). The HR for mogamulizumab versus vorinostat was 0.53 (95% CI, 0.41 to 0.69), in favour of mogamulizumab. Results of sensitivity analyses varying the definition of PFS were consistent with the primary analysis.27 Multiple subgroup and post hoc analyses of PFS were conducted, demonstrating improved survival with mogamulizumab in patients with SS (13.30 versus 3.13 months), advanced-stage disease (9.40 versus 3.07 months),28 stage III or IV disease (10.90 versus 3.00 months),27 and B1 (8.63 versus 2.53 months) and B2 (11.17 versus 3.30 months) blood involvement.29
HRQoL was assessed using the Skindex-29, FACT-G, and EQ-5D-3L. In general, the magnitude of improvement from baseline to cycle 5 was greater with mogamulizumab compared with vorinostat. Across all scale domains for the Skindex-29, mogamulizumab and vorinostat reduced scores from baseline to cycle 5, with mean symptom scale scores demonstrating the greatest improvement (−18.0 versus −8.2). Results were consistent across HRQoL measures, with both mogamulizumab and vorinostat demonstrating an increase in total FACT-G score at cycle 5. With the EQ-5D Visual Analogue Scale (VAS), mean baseline scores increased from 60.9 (SD = 22.10) to 69.0 (SD = 20.30) at cycle 5 for mogamulizumab compared with 60.8 (SD = 20.02) to 63.5 (SD = 20.08) for vorinostat.27,30
Other secondary outcomes evaluated also favoured mogamulizumab, with a BOR of 34.9% versus 6.5%, and a median DOR of 14.07 months with mogamulizumab compared with 9.13 months with vorinostat. At a median efficacy follow-up of 17.0 months, there was no difference in median OS between mogamulizumab (median OS was not estimable [NE]) and vorinostat (43.93 months).27
Harms Results
The overall incidence of treatment-emergent adverse events (TEAEs) was consistent between the mogamulizumab (97.3%) and vorinostat (99.5%) arms. AEs were reported for randomized treatment. The incidence of specific TEAEs in patients who crossed over was not reported. The most frequently reported TEAEs with mogamulizumab were infusion-related reaction (33.2%), drug eruption (23.9%), diarrhea (23.4%), and fatigue (23.4%). The most frequently reported TEAEs with vorinostat were diarrhea (61.8%), nausea (42.5%), fatigue (37.6%), and thrombocytopenia (30.6%). The incidence of serious adverse events (SAEs) was higher in the mogamulizumab group compared with vorinostat (37.5% versus 24.7%). The most frequently occurring SAEs by system organ class (SOC) were infections and infestations (16.3% versus 10.8%). Withdrawals due to AEs were similar between treatment groups during the randomized treatment period, with 19.0% of patients in the mogamulizumab group and 23.1% of patients in the vorinostat group discontinued treatment due to AEs, most frequently due to drug eruption for mogamulizumab (7.1%). A total of 5 (2.7%) and 9 (4.8%) patients, respectively, died due to AEs during the randomized treatment period.27
Notable harms, including infusion-related reactions (IRRs) and infections, were generally more frequent in the mogamulizumab group. In the randomized treatment phase, IRRs with mogamulizumab occurred in 33.2% of patients compared with 0.5% of patients receiving vorinostat, while infections and infestations occurred in 64.1% and 50% of patients, respectively. Though not expressly reported as a group in the MAVORIC trial, the most frequently occurring immune-related TEAEs with mogamulizumab were drug eruption (23.9% versus 0.5%) and fatigue (23.4% versus 37.6%).27
Table 2: Summary of Key Results From Pivotal and Protocol Selected Studies
Outcome | MAVORIC | |
|---|---|---|
Mogamulizumab (N = 186) | Vorinostat (N = 186) | |
ORR (ITT) | ||
ORR (confirmed CR + PR), n (%) | 52 (28.0) | 9 (4.8) |
Confirmed CR, n (%)a | 4 (2.2) | 0 |
Confirmed PR, n (%) | 47 (25.3) | 9 (4.8) |
Risk difference, mogamulizumab vs. vorinostat (95% CI)b | 23.1 (12.8 to 33.1) | |
Adjusted P valuec | < 0.0001 | |
PFS (ITT) | ||
PFS event, n (%) | 110 (59.1) | 131 (70.4) |
Censored, n (%) | 76 (40.9) | 55 (29.6) |
Median PFS, months (95% CI)d | 7.70 (5.67 to 10.33) | 3.10 (2.87 to 4.07) |
HR (95% CI)e | 0.53 (0.41 to 0.69) | |
Log-rank P valuee | < 0.0001 | |
OS (ITT) | ||
OS event, n (%) | 40 (21.5) | 47 (25.3) |
Censored, n (%) | 146 (78.5) | 139 (74.7) |
Median OS, months (95% CI) | NE (NE to NE) | 43.93 (43.57 to NE) |
HR (95% CI)f | 0.93 (0.61 to 1.43) | |
Log-rank P valuef | 0.9439 | |
Harms, n (%) (safety analysis set)g | ||
AEs | 179 (97.3) | 185 (99.5) |
SAEs | 69 (37.5) | 46 (24.7) |
WDAE (from study treatment) | 35 (19.0) | 43 (23.1) |
Deaths | 5 (2.7) | 9 (4.8) |
Notable harms | ||
IRR, n (%) | 61 (33.2) | 1 (0.5) |
Infections, n (%) | 118 (64.1) | 93 (50.0) |
AE = adverse event; BOR = best overall response; CI = confidence interval; CR = complete response; HR = hazard ratio; IRR = infusion-related reaction; ITT = intention-to-treat population; NE = not estimable; ORR = overall response rate; OS = overall survival; PFS = progression-free survival; PR = partial response; SAE = serious adverse event; WDAE = withdrawal due to adverse event.
aOne patient in the mogamulizumab group had a confirmed PR based on investigator’s assessment. Since this patient had a CR with relapse in the skin only, they should have been counted as having a confirmed CR, since a relapse in skin does not constitute progression if it does not meet the specified criteria for progression.
bThe 95% CIs for response rate are the exact 95% CIs. The 95% CI for the risk difference is the exact 95% unconditional CI for the risk difference (mogamulizumab minus vorinostat).
cThe P value was obtained from a Cochran-Mantel-Haenszel test adjusting for disease type, disease stage, and region. The adjusted P value was calculated using the Šídák method.
dThe 95% CIs were obtained from SAS PROC LIFETEST using log-log transformation.
eHRs and 95% CIs are based on Cox proportional hazards model with treatment, disease type, disease stage, and region as covariates. The P value (2-sided) was obtained from a stratified log-rank test (1-sided test at the 0.025 level or equivalent 2-sided test at the 0.05 level) with disease type, disease stage, and region as stratification factors.
fHR and 95% CI are based on Cox proportional hazards model with treatment, disease type, disease stage, and region as covariates. The P value (2-sided) was obtained from a stratified log-rank test with disease type, disease stage, and region as stratification factors.
gHarms were all considered treatment-emergent.
Source: MAVORIC (0761-010) Clinical Study Report.27
Critical Appraisal
The MAVORIC trial was an open-label, phase III RCT. In general, patients did not differ with regard to baseline characteristics, indicating that randomization was successful. There were no notable differences between mogamulizumab and vorinostat among patients discontinuing randomized treatment, with most patients in both groups discontinuing due to disease progression. The reviewers and the clinical experts consulted by CADTH agreed the open-label design used was appropriate. However, they noted this could potentially increase the risk of bias in the reporting of outcomes that are subjective in measurement and interpretation, such as response, HRQoL, and AEs; however, a blinded independent review was instituted to assess for any potential investigator bias. The MAVORIC trial also included a 1-way crossover design, where patients with disease that failed treatment with vorinostat were able to cross over to the mogamulizumab group, which may have confounded the results for OS. As an exploratory end point, results for OS are considered descriptive The end points considered in the study were clinically appropriate for this population per the clinical experts consulted by CADTH and have been recommended by the International Society for Cutaneous Lymphomas (ISCL), the United States Cutaneous Lymphoma Consortium, and the Cutaneous Lymphoma Task Force of the European Organisation for Research and Treatment of Cancer (EORTC).31 Acceptable methods to account for multiplicity were used in the MAVORIC trial to adjust for the overall studywise type I error rate for the key secondary end points. All subgroup analyses of the MAVORIC trial, including disease type, stage, blood involvement, region, age, gender, race, and lactate dehydrogenase levels, were pre-specified; however, they were not controlled for multiplicity.
In discussion with the clinical experts consulted by CADTH, the inclusion and exclusion criteria for the MAVORIC trial were considered generally appropriate for enrolling patients with a rare disease. There were, however, no Canadian sites in the MAVORIC trial. The MAVORIC trial enrolled patients with MF or SS with stage IB, IIA, IIB, III, or IV disease. The inclusion of so many stages of disease was considered by the CADTH review team and clinical experts to be quite broad, given the differences in disease behaviour and prognosis at each stage. The clinical experts also noted that the trial included a high proportion of patients with SS compared with the overall incidence of SS in Canada. The clinical experts also expected more patients with an ECOG Performance Status of 2 in Canadian clinical practice, who were not eligible for the MAVORIC trial. Though approved by Health Canada for the treatment of CTCL, vorinostat is not widely accessible in Canada; thus, the comparative efficacy of mogamulizumab relative to vorinostat in patients in Canada with MF or SS may not be generalizable.
Results for HRQoL demonstrated improvements in HRQoL domains at various time points in the analysis; however, per the definitions of these populations, HRQoL results were presented only as observed data and not for the entire intention-to-treat (ITT) population. As such, these patients can be considered responders to treatment, which may bias the results. Moreover, high attrition rates were observed for all HRQoL measures, ranging from 42.2% to 44.8% of mogamulizumab-treated patients and 66.3% to 67.03% of vorinostat-treated patients failing to complete the assessments at 6 months compared with baseline. This created uncertainty in the results and thus may impact the generalizability of the results.
Indirect Comparisons
Description of Studies
The sponsor submitted an indirect treatment comparison (ITC) that compared individual patient data from the MAVORIC trial with the populations of relevant trial reports for comparators of interest in the treatment of CTCL, including the subtypes MF and SS. The objective of the sponsor-submitted report was to assess the feasibility of performing ITCs to compare mogamulizumab with relevant comparators of interest for the outcomes of PFS, OS, time-to-treatment failure (TTF), ORR, and Skindex-29 score. Of specific focus was the comparison between mogamulizumab (MAVORIC trial) and the drugs in the 2 arms of the ALCANZA study (brentuximab vedotin and physician’s choice [methotrexate or bexarotene]), given that these comparators were expected to be the main comparators in the cost-effectiveness model.
An initial feasibility assessment was conducted based on the findings of a clinical systematic literature review. The review extracted evidence from 39 publications detailing 26 different studies: 14 parallel trials and 12 single-arm studies. The feasibility assessment was done to determine whether a network meta-analysis was possible by evaluating the network connectivity of included trials via comparators, the trial inclusion and exclusion criteria (i.e., population), the demographic and disease characteristics, and the study end points.
Based on the comparators of interest considered in the feasibility assessment, no connected networks could be formed with mogamulizumab. As such, an unanchored matching-adjusted indirect comparison (MAIC) was used as an alternative analytical method to compare treatment from the ALCANZA trial with mogamulizumab. Multiple MAICs were conducted, including the comparison of the mogamulizumab arm with the brentuximab vedotin arm for the entire (ITT) population of the MAVORIC and ALCANZA trials, as well as the MF-only population of the MAVORIC trial. Additional comparisons included the vorinostat arm of the MAVORIC trial with the physician’s choice (methotrexate or bexarotene) arm in the ITT and MF-only populations. The outcomes evaluated included PFS, OS, and response in skin.
Efficacy Results
Two studies were included in the sponsor-submitted MAIC: MAVORIC and ALCANZA. For PFS, the results of the MAIC comparing mogamulizumab with brentuximab after weighting demonstrated a greater probability of PFS events with mogamulizumab over brentuximab using both the ITT (HR = 2.21; 95% CI, 1.68 to 3.19) and MF-only (HR = 2.52; 95% CI, 1.78 to 3.75) populations, while there was no difference between vorinostat and physician’s choice. For OS, there was no difference between mogamulizumab and brentuximab after matching patients to the ALCANZA population in the MAVORIC ITT (HR = 0.90; 95% CI, 0.62 to 1.27) or MF-only populations (HR = 0.79; 95% CI, 0.45 to 1.18). The comparison between vorinostat and physician’s choice could not be conducted due to the crossover in the trials. | | | |||||| | | ||| | ||||||||||| | |||||||||| | ||| | |||||| | | || | || | ||||||||| | ||||| | | || | | | | | | | || | || | | | ||| | |||| | | | | || | ||||||||| | |||||| | || | | | ||| | ||||| | ||| | ||| | | | | |||||||| | ||||| | |||||||| | | | ||||||||| |||||||
Critical Appraisal
The choice to conduct an MAIC between MAVORIC and ALCANZA was justified, considering the lack of a common comparator. Moreover, no rationale or justification for using brentuximab and physician’s choice from the ALCANZA trial as the primary comparator in the MAIC was provided. The main comparator for the MAIC, brentuximab vedotin, is indicated for adult patients with primary cutaneous anaplastic large-cell lymphoma or CD30-expressing MF who have received prior systemic therapy. This population was different from that of the MAVORIC trial, as no SS patients were included, and CD30 positivity was not assessed in MAVORIC, increasing the uncertainty in the analyses. The MAIC also provided a naive comparison between vorinostat in the MAVORIC trial and physician’s choice (consisting of methotrexate or bexarotene) in the ALCANZA trial. These 3 drugs (vorinostat, methotrexate, and bexarotene) are rarely used in Canadian clinical practice, according to the clinical experts consulted by CADTH, thus further limiting the generalizability of these results.
There were many key differences between the MAVORIC and ALCANZA trials that impact the comparability of populations within these studies, particularly the inclusion criteria of the studies (i.e., the eligible population), the specific diagnosis, as well as differences in various baseline characteristics, such as CD30 status, disease stage, blood involvement, prior treatments, and treatments in the study. The heterogeneity in population was accounted for by conducting matched analyses with the ITT population of MAVORIC as well as the MF-only population of MAVORIC to the ALCANZA population, resulting in an effective sample size that was reduced by 50.5% for the ITT population, and 25.7% for the MF-only population. There were also considerable differences in baseline age, ECOG Performance Status, disease stage, and blood involvement; however, it was uncertain as to what direction this may impact results. A comprehensive list of prognostic factors and treatment-effect modifiers for weighting was provided (including these baseline characteristics); however, the method of identification, justification, and validation of prognostic factors and treatment-effect modifiers was unclear, and it was uncertain whether all key factors were included in weighting; thus, the risk of bias on the relative treatment effects for unanchored MAICs is considered substantial and must be considered.
Overall, given the differences between MAVORIC and ALCANZA in terms of inclusion and exclusion criteria, the diagnosed population and the populations included in the analyses, the baseline characteristics, the differences in study design, and the large reduction in effective sample size, the results of the MAIC are uncertain and may not be generalizable.
Conclusions
Based on the clinical evidence from the phase III, open-label RCT, MAVORIC, conducted in patients with MF or SS whose condition has failed to respond to at least 1 prior systemic therapy, mogamulizumab was associated with a clinically meaningful and statistically significant improvement in PFS and ORR compared with vorinostat; however, given the open-label design of the study, these results must be interpreted with caution due to the potential for reporting biases. Notably, these results were most pronounced in patients with advanced SS and blood involvement. Patients highlighted the need for accessible, alternative treatment options that improve survival, and HRQoL; however, due to high attrition rates, the short follow-up duration of the MAVORIC trial, and the high rate of crossover, there remains uncertainty in the effect of mogamulizumab on HRQoL and OS, as no difference between mogamulizumab and vorinostat was observed for OS. The sponsor-submitted MAIC was associated with significant clinical heterogeneity in the included studies, which resulted in multiple limitations and notable uncertainty in the comparative efficacy of mogamulizumab. Given the difficulty in accessing treatment for MF and SS, the lack of standard of care, and that vorinostat is not widely available in Canada, there is some uncertainty around the true significance of the comparative efficacy of mogamulizumab. Overall, the available evidence suggests that mogamulizumab provides an additional treatment option for patients with MF or SS whose disease has failed to respond to at least 1 systemic therapy, delaying disease progression, and providing a meaningful clinical response in the overall population of both MF and SS.
Introduction
Disease Background
CTCLs are a group of extra-nodal non-Hodgkin lymphomas in which cancerous lymphocytes affect the skin. MF and SS are the 2 most common types of CTCL. Both MF and SS result from infiltration of malignant T-lymphocytes preferentially to skin. MF is usually associated with an indolent clinical course and intermittent, stable, or slow progression, while SS is a rare leukemic subtype of CTCL characterized by a more aggressive course of disease and shorter survival.11 Together, MF and SS account for approximately one-half to two-thirds of CTCL cases (SS accounting for only 2% to 3% of all CTCLs).1-5
MF is a highly symptomatic disease, with variable clinical presentation. The clinical presentation of MF is highly variable, ranging from non-specific erythematous scaly patches to thin plaques or papulonodular eruptions appearing mostly on non–sun-exposed areas.12 In approximately 30% of cases, the patches or plaques evolve into tumours.13 In some instances, MF can spread to the lymph nodes, blood, or other organs, such as the spleen, liver, or lungs, though mucosal involvement is rare.9,12 Patients with SS present with a triad of skin redness (erythroderma), enlarged lymph nodes (lymphadenopathy), and a large number of circulating atypical lymphocytes (Sézary cells) in the skin, lymph nodes, and peripheral blood.12,14 The psychological aspects of this incurable cancer affecting the skin present a significant burden to patients.15
According to the National Cancer Institute, the incidence of MF has remained stable since 1995 with an incidence of 5.6 per million persons.5 In Canada, 2,510 cases of MF were documented from 1992 through 2010 representing 37.5% of CTCL cases, and 110 cases of SS representing 1.6% of CTCL cases.10 The incidence of MF increases with age, with most patients diagnosed in their 50s and 60s, with an increased incidence in patients greater than 70 years.4,6-8 MF is more common in Black patients than Caucasians, and males, with a male-to-female ratio of 1.6 to 2:1.2,4,22 Due to the indolent course of the disease, the median survival in early-stage disease ranges from 10 to 35 years, dropping to approximately 4 years with advanced disease.7,32,33 Clinically, SS mostly affects patients > 60 years of age and is associated with a poor prognosis (median survival of approximately 3 years).7,9
As MF and SS can resemble other inflammatory dermatoses, diagnosis can be challenging, sometimes taking upwards of 2 to 6 years.16-21 Diagnosis of MF or SS is confirmed by skin biopsy and blood tests, though characteristic histologic features of MF may be absent in early disease.22 Diagnosis is further confirmed with immunophenotyping and DNA analysis of the T-cell receptor gene rearrangement to define the clonal population (CD2, CD3, CD4, CD5, CD7, CD8, CD20, and CD30).5,12 Patients with MF and SS are typically characterized by the following immunophenotype: positive for CD2, CD3, CD5, and CD4; negative for CD8; and lack of certain T-cell markers (e.g., negative for CD7 and CD26 expression).12,34
MF and SS are classified using a CTCL-specific modification of the tumour, nodes, metastasis, and blood classification system.4,16 Both MF and SS are defined histologically and staged by the same criteria.2,4 Patients are classified based on the number and type of skin lesions they have (T), lymph node involvement (N), metastasis or visceral organ involvement (M), and peripheral blood involvement (B), resulting in a diagnosis of a disease stage from IA through to IVB (Table 3).4 These stages can be grouped as early (stages IA, IB, and IIA) and advanced (stages IIB to IVB) disease.4,18 Approximately two-thirds of MF patients have early-stage disease at the time of diagnosis.18-20
Table 3: ISCL Clinical Staging of MF and SS
Clinical stage | T (skin) | N (node) | M (visceral) | B (blood involvement) |
|---|---|---|---|---|
IA (limited skin involvement) | T1 (patches, papules, and/or plaques covering < 10% of BSA) | N0 | M0 | B0 or B1 |
IB (skin-only disease) | T2 (patches, papules, and/or plaques covering ≥ 10% of BSA) | N0 | M0 | B0 or B1 |
IIA | T1 to T2 | N1 to N2 | M0 | B0 or B1 |
IIB (tumour stage) | T3 (1 or more tumours ≥ 1 cm in diameter) | N0 to N2 | M0 | B0 or B1 |
IIIA (erythrodermic disease) | T4 (confluence of erythema ≥ 80% BSA) | N0 to N2 | M0 | B0 |
IIIB (erythrodermic disease) | T4 (confluence of erythema ≥ 80% BSA) | N0 to N2 | M0 | B1 |
IVA1 | T1 to T4 | N0 to N2 | M0 | B2 |
IVA2 | T1 to T4 | N3 | M0 | B0 or B1 or B2 |
IVB | T1 to T4 | N0 to N3 | M1 | B0 or B1 or B2 |
— | Large-cell transformation | — | — | — |
BSA = body surface area; ISCL = International Society for Cutaneous Lymphomas; MF = mycosis fungoides; SS = Sézary syndrome.
Source: Olsen et al. (2007).4
Standards of Therapy
Both MF and SS are incurable diseases though, if discovered early, the prognosis for patients with early-stage MF can be very good.25 Most treatment options do not result in durable remissions and are often given in an ongoing or maintenance fashion to achieve disease control with as little impact on QoL as possible. Clinical staging of patients with MF is essential, not only for assessment of prognosis but also to guide decisions in management of the disease. Selection of systemic therapies with lower toxicity treatments versus other systemic regimens is dependent on clinical features such as the extent of patches or plaques, disease burden profile in the skin, lymph nodes, and blood; prior therapies; comorbidities; and pathologic features, including confirmation of large-cell transformation (LCT) or folliculotropic MF and immunohistochemistry data (i.e., CD30-positive). A disease that relapses (at the same stage) after discontinuation of therapy often responds well to re-treatment with previous therapy.25
For patients with early-stage disease with limited skin involvement (stage IA or IB to IIA), skin-directed therapies including topicals (topical nitrogen mustard, compounded carmustine, or imiquimod), phototherapy (psoralen plus UVA [PUVA] or narrow-band UVB light) or radiation, including external beam radiation therapy such as involved-site radiation therapy or total skin electron beam therapy (TSEBT) are used, which may provide disease control without major cumulative toxicities; however, its use is limited by the lack of availability of this technique in Canada. In cases of resistant or progressive skin disease, biologic response modifiers such as retinoids (isotretinoin, alitretinoin, bexarotene) or interferon may be combined with topical therapies.16
Systemic therapy is recommended for advanced-stage disease (stage ≥ IIB). In advanced MF and SS, first-line treatment consists of oral retinoid therapy (e.g., bexarotene, isotretinoin) with or without interferon, or single-drug chemotherapy or other anti-cancer therapies, which are generally preferred to combination chemotherapy (i.e., cyclophosphamide, doxorubicin, vincristine, and prednisone [CHOP] or cyclophosphamide, doxorubicin, vincristine, etoposide, and prednisone [CHEOP]). Single-drug chemotherapy for MF and SS includes methotrexate, gemcitabine, or liposomal doxorubicin, with pralatrexate or chlorambucil used in some cases. Second- and third-line treatment for MF consist of previously unused single-drug chemotherapy or other anti-cancer therapy, HDAC inhibitors such as vorinostat or romidepsin, or immunotherapies such as brentuximab vedotin (in patients with CD30-positive disease), or alemtuzumab. In SS, or in patients with blood involvement, ECP with or without retinoids and interferon may be used in either the first- or second-line setting; otherwise, eligible treatment options beyond the first-line setting remain the same as in MF. Multi-drug chemotherapy regimens are generally reserved for disease refractory to multiple prior therapies or for bulky lymph node or solid organ disease, and/or as a bridge to allogeneic hematopoietic stem-cell transplant, given the greater toxicity.16 In many cases, given the lack of reliable options, enrolment in a clinical trial may be pursued.16,25 Systemic therapy may often be combined with skin-directed therapy to maximize clinical responses in the skin compartment and to provide added efficacy without cumulative toxicities.25
Drug
Mogamulizumab is a defucosylated, humanized IgG1 kappa monoclonal antibody that binds to CCR4, a G protein–coupled receptor for CC chemokines that is involved in the trafficking of lymphocytes to various organs. Non-clinical in vitro studies demonstrate that mogamulizumab binding targets a cell for antibody-dependent cellular cytotoxicity, resulting in depletion of the target cells. CCR4 is expressed on the surface of some T-cell malignancies and is expressed on regulatory T cells and a subset of T helper 2 T cells.26
Mogamulizumab is provided as a 20 mg/5 mL vial for reconstitution at a dose of 1.0 mg/kg. Mogamulizumab is administered as an IV infusion over at least 60 minutes at a dose of 1 mg/kg on days 1, 8, 15, and 22 of the first 28-day cycle, then on days 1 and 15 of each subsequent 28-day cycle until disease progression or unacceptable toxicity.26
Mogamulizumab has received a Notice of Compliance (NOC) from Health Canada for the treatment of adult patients with relapsed or refractory MF or SS after at least 1 prior systemic therapy.26 The Health Canada NOC was granted on June 2, 2022.35 Mogamulizumab has not been previously reviewed by CADTH. The sponsor’s reimbursement request is the same as the proposed Health Canada indication.
Mogamulizumab was approved by the Ministry of Health, Labour, and Welfare in Japan for the treatment of primary and relapsed or refractory, CCR4-positive adult T cell leukemia/lymphoma (December 14, 2014, and March 30, 2012, respectively), relapsed or refractory, CCR4-positive peripheral T cell lymphoma (March 17, 2014); and relapsed or refractory CCR4-positive CTCL (March 17, 2014). Mogamulizumab has been granted an orphan designation for CTCL in the EU, Japan, and the US.27
Key characteristics of mogamulizumab and the main comparators as specified in the protocol for this review (Table 6) are summarized in Table 4.
Table 4: Key Characteristics of Treatment for CTCL, MF, and SS
Detail | Mogamulizumab | Biologic response modifiers | Retinoids | Single- or multi-drug chemotherapy | HDACi | Other mAbs |
|---|---|---|---|---|---|---|
Drug | Mogamulizumab | Interferon alfa | Isotretinoin | Gemcitabine | Vorinostat | Brentuximab vedotin |
Mechanism of action | Mogamulizumab is a defucosylated, humanized IgG1 kappa monoclonal antibody that binds to CCR4, a G protein–coupled receptor for CC chemokines that is involved in the trafficking of lymphocytes to various organs. Non-clinical in vitro studies demonstrate that mogamulizumab binding targets a cell for ADCC, resulting in depletion of the target cells. CCR4 is expressed on the surface of some T-cell malignancies and is expressed on regulatory T cells and a subset of T helper 2 T cells. | Interferons exert their cellular activities by binding to specific membrane receptors on the cell surface. The results of several studies suggest that, once bound to the cell membrane, interferon initiates a complex sequence of intracellular events that includes the induction of certain enzymes. | The mechanism of action of isotretinoin is unknown. Vitamin A is important for functional integrity of the skin and is known to affect the keratinization process. In acne patients, improvement occurs in association with a reduction in sebum secretion. The decrease in sebum secretion is temporary and is related to either the dose or duration of isotretinoin administration and reflects a reduction in sebaceous gland size and an inhibition of sebaceous gland differentiation. The clinical experts noted that the mechanism of action of isotretinoin in CTCL is not clearly established. It is postulated that isotretinoin’s effect is mediated through binding with the retinoic acid receptor, influencing downstream signalling pathways which regulate cell growth. Specifically, it is believed to activate P53, a tumour suppressor gene, along with influencing the tumour microenvironment. | Gemcitabine is a cell cycle–dependent oncolytic drug of the antimetabolite class. It is a deoxycytidine analogue (difluoro-deoxycytidine) that is metabolized intracellularly to the active diphosphate and triphosphate nucleosides. The cytotoxic effects of gemcitabine are exerted through incorporation of the triphosphate into DNA, resulting in inhibition of DNA synthesis and induction of apoptosis. | Vorinostat is a potent inhibitor of HDAC1, HDAC2 and HDAC3 and HDAC6 (IC50 < 86 nM). These enzymes catalyze the removal of acetyl groups from the lysine residues of proteins, including histones. The anti-neoplastic effect of vorinostat is attributed to the inhibition of HDAC activity and subsequent accumulation of acetylated proteins, including histones. Histone acetylation results in the transcriptional activation of genes, including tumour suppressor genes, whose expression leads to induction of differentiation, apoptosis, and/or inhibition of tumour growth. | Brentuximab vedotin is an IgG1 ADC directed against CD30. The small molecule, MMAE, is a potent microtubule-disrupting drug. MMAE is covalently attached to the antibody via a linker. Non-clinical data suggest that the anti-cancer activity of brentuximab is due to the binding of the ADC to CD30-expressing cells, followed by internalization of the ADC-CD30 complex, and the release of MMAE via proteolytic cleavage. Binding of MMAE to tubulin disrupts the microtubule network within the cell, subsequently inducing cell-cycle arrest and apoptotic death of the cells. |
Indicationa | The treatment of adult patients with relapsed or refractory MF or SS after at least 1 prior systemic therapy. | Multiple indications: Chronic hepatitis C, chronic active hepatitis B, CML, MM, NHL (FL), malignant melanoma, AIDS-related Kaposi sarcoma, hairy cell leukemia, BCC, condylomata acuminata. | Severe nodular and/or inflammatory acne, acne conglobata, recalcitrant acne. | Multiple indications: Locally advanced or metastatic pancreatic adenocarcinoma, locally advanced or metastatic NSCLC, stage IV TCC of the bladder (+ cisplatin), and for unresectable, locally recurrent, or metastatic breast cancer (+ paclitaxel). | The treatment of cutaneous manifestations in patients with advanced CTCL who have progressive, persistent, or recurrent disease subsequent to prior systemic therapies. | Multiple indications including the treatment of adult patients with pcALCL or CD30-expressing MF who have received prior systemic therapy. |
Route of administration | IV | Disease-dependent (SC, IM, IV, and IL) | Oral | IV | Oral | IV |
Recommended dose | The recommended dose is 1 mg/kg administered as an IV infusion over at least 60 minutes. | Indication-specific dosing. According to the clinical experts, therapeutic doses in CTCL typically range from 3 MU to 15 MU weekly. | 0.5 mg/kg daily for a period of 2 to 4 weeks. Maintenance dose of between 0.1 mg/kg and 1 mg/kg daily and, in exceptional instances, up to 2 mg/kg daily, depending on individual response and tolerance. | Indication-specific dosing. | 400 mg once daily. | For MF: 1.8 mg/kg up to a maximum of 180 mg in combination with CHP every 3 weeks for 6 or 8 cycles or until disease progression or unacceptable toxicity. |
Serious adverse effects or safety issues | Complications, including severe GVHD, have been reported in patients who received allogeneic HSCT after mogamulizumab. Serious skin reactions, including SJS and TEN, have been reported in patients treated with mogamulizumab; some of these cases were reported with fatal outcomes. Patients should be closely monitored for symptoms or signs that suggest serious skin reactions, including SJS or TEN. Serious infections and IRR have been reported in patients treated with mogamulizumab. | Alpha interferons cause or aggravate fatal or life-threatening neuropsychiatric, autoimmune, ischemic, and infectious disorders. Patients should be monitored closely with periodic clinical and laboratory evaluations. Patients with persistently severe or worsening signs or symptoms of these conditions should be withdrawn from therapy. In many cases, but not all cases, these disorders resolve after stopping interferon therapy. |
| Gemcitabine should be used with extreme caution in patients whose bone marrow reserve may have been compromised by prior irradiation or chemotherapy, or whose marrow function is recovering from previous chemotherapy. Gemcitabine can suppress bone marrow function manifested by leucopenia, thrombocytopenia, and anemia. Patients should be closely monitored before each dose for granulocyte and platelet counts. Periodic physical examination and checks of renal and hepatic function should be made to detect non-hematologic toxicity. Administration of gemcitabine in patients with concurrent liver metastases or a pre-existing medical history of hepatitis, alcoholism, or liver cirrhosis may lead to exacerbation of the underlying hepatic insufficiency. Acute shortness of breath with a temporal relationship to gemcitabine administration may occur. | The following are clinically important AEs: Thromboembolism, including fatal cases; thrombocytopenia; and anemia. | Clinically significant and/or life-threatening AEs include: JC virus infection resulting in PML and death, SJS and TEN, serious and opportunistic infections, acute pancreatitis, gastrointestinal complications, and pulmonary toxicity. In addition, combination therapy with AVD may result in clinically significant and/or life-threatening febrile neutropenia. |
Additional options in class | See “Other mAbs” | Interferon alfa-2a, interferon alfa-2b | Bexarotene, alitretinoin | Methotrexate, liposomal doxorubicin, etoposide, CHOP, CHEOP | Romidepsin | Alemtuzumab, pembrolizumab |
ADC = antibody-drug conjugate; ADCC = antibody-dependent cellular cytotoxicity; AE = adverse event; AVD = doxorubicin, vinblastine, and dacarbazine; BCC = basal cell carcinoma; CHEOP = cyclophosphamide, doxorubicin, vincristine, etoposide, and prednisone; CHOP = cyclophosphamide, doxorubicin, vincristine, and prednisone; CHP = cyclophosphamide, doxorubicin, and prednisone; CML = chronic myelogenous leukemia; CTCL = cutaneous T-cell lymphoma; FL = follicular lymphoma; GVHD = graft-versus-host disease; HDAC = histone deacetylase; HDACi = histone deacetylase inhibitor; HSCT = hematopoietic stem-cell transplant; IC50 = half-maximal inhibitory concentration; IgG1 = immunoglobulin G1; IL = intralesional; IM = intramuscular; IRR = infusion-related reactions; JC = John Cunningham; mAbs = monoclonal antibodies; MF = mycosis fungoides; MM = multiple myeloma; MMAE = monomethyl auristatin E; NHL = non-Hodgkin lymphoma; NSCLC = non–small cell lung cancer; pcALCL = primary cutaneous anaplastic large-cell lymphoma; PML = progressive multifocal leukoencephalopathy; SC = subcutaneous; SJS = Stevens-Johnson syndrome; SS = Sézary syndrome; TCC = transitional cell carcinoma; TEN = toxic epidermal necrolysis.
aHealth Canada–approved indication.
Source: Product monographs for mogamulizumab,26 interferon alfa-2b,36 isotretinoin,37 gemcitabine,38 vorinostat,39 and brentuximab vedotin.40
Stakeholder Perspectives
Patient Group Input
This section was prepared by CADTH staff based on the input provided by patient groups.
Lymphoma Canada, in collaboration with the Canadian Skin Patient Alliance and Cutaneous Lymphoma Foundation, conducted an anonymous online survey between March 8, 2021, and September 21, 2021. The respondents consisted of patients with CTCL, primarily MF or SS. A total of 449 patients who responded to the survey had been diagnosed with either MF or SS; 46 (14%) of these patients were living in Canada. The majority of respondents were aged 60 years or older (69%), and more than half were female (54%).
MF can be difficult to diagnose, with symptoms that can occur for many years before a diagnosis is made. It can masquerade as other more common skin conditions, such as eczema or psoriasis. The majority of patients participating in this survey were diagnosed 1 to 5 years ago (41%), with a portion of patients diagnosed more than 10 years ago (26%). Only 23% of patients had their condition correctly diagnosed as MF or SS at presentation. Symptoms of MF and SS that most impacted patients’ QoL at diagnosis included visual patches or lesions (78%), and itchiness of skin or skin lesions (57%). Symptoms that most commonly affected respondents’ current QoL were similar to those at diagnosis, with the stress of diagnosis plus the fear of and worry about disease progression as well as anxiety impacting their current well-being.
A total of 327 patients provided information about their experience with CTCL treatment for MF and SS. Of the 211 patients who received systemic treatments, 27% received interferon, 26% received methotrexate, 24% received oral bexarotene, 23% received mogamulizumab, and 10% were treated with romidepsin. The most common side effects respondents experienced by patients during their MF or SS CTCL treatments included fatigue (41%), skin pain or burning (34%), skin irritation or rash (33%), and moderate or severe itching (30%). A total of 27% of survey respondents indicated that the number of clinic visits had the most significant impact on their QoL, while 21% indicated that treatment-related fatigue had the most significant impact on their QoL, citing additional negative impacts due to treatment on their ability to work (21%), travel (21%), and have intimate relationships (19%).
Access to treatment within the patient’s community is an important consideration, as certain treatments may be available only at specific tertiary cancer centres. A subanalysis of patients living in Canada revealed that 32% of them could not access treatment locally. Most patients responding to this survey were living in the US, where more treatment options are approved for use, including mogamulizumab; however, 81% of all respondents cited the importance of having an increased number of treatment options available. Patients indicated that longer survival (82%) and better QoL (76%) are extremely important outcomes for new therapies, followed by longer treatment-free periods (70%), easier treatment application (68%), and fewer side effects (64%).
Of all patients who responded to the survey, 52 (12%) had experience with mogamulizumab, accessed largely through private insurance (42%), public drug programs (29%), compassionate access programs (17%), or clinical trials (8%). At the time of the survey, 44% were still receiving treatment, 12% of patients “completed their full course of mogamulizumab” [sic], and 12% discontinued due to side effects, and 10% did not have their symptoms controlled with mogamulizumab. Only 2 patients had their disease progress during treatment, while 25% were in remission. Of the 52 respondents, a total of 36% of patients had all of their symptoms managed by mogamulizumab, with major symptom management in skin itchiness (62%), red skin patches or rash (56%), and skin pain (25%). When asked to describe their experience with mogamulizumab, 69% of patients indicated they had a good to excellent experience with the therapy.
Clinician Input
Input From the Clinical Experts Consulted by CADTH
All CADTH review teams include at least 1 clinical specialist with expertise in the diagnosis and management of the condition for which the drug is indicated. Clinical experts are a critical part of the review team and are involved in all phases of the review process (e.g., providing guidance on the development of the review protocol, assisting in the critical appraisal of clinical evidence, interpreting the clinical relevance of the results, and providing guidance on the potential place in therapy). The following input was provided by 2 clinical specialists with expertise in the diagnosis and management of MF and SS.
Unmet Needs
The clinical experts highlighted that MF or SS is an incurable disease that is often characterized by extensive and disfiguring cutaneous involvement with intense skin-related symptoms that include itching, burning, and pain. They emphasized that treatment selection in Canada is often guided by which drugs are funded and available rather than the 1 with the best chances of response and fewest side effects. The clinical experts stated that many of the treatments (TSEBT, ECP, or UV light) must be delivered at specialized or urban centres and thus are not practically accessible to many patients living with cutaneous lymphoma.
Per the clinical experts, the treatment goals for MF and SS consist of prolonged survival, improved response rates, improvement in skin-related symptoms, and QoL. It was highlighted by the experts that many currently available treatment options have suboptimal response rates (< 30% to < 50%), or responses of limited duration (< 4 months), and they acknowledged that these responses are primarily in the context of skin; however, when used in SS, they are in the context of blood involvement as well. They noted that some treatments may improve objective parameters, but patients may continue to experience skin symptoms or impairment of QoL or function. Additional goals of treatment noted by the experts included improved independence and reducing the burden on caregivers.
Place in Therapy
According to the clinical experts consulted by CADTH, limited-stage MF is treated with skin-directed therapy, including topical corticosteroids, UV light therapy, which is either narrow-band UVB or PUVA, and less commonly topical carmustine or other topical drugs. In some patients who are refractory or intolerant to these approaches, systemic therapy may be required. In Canada, this generally is limited to interferon, isotretinoin (off-label use for MF), or oral methotrexate. Bexarotene was noted by the experts as a retinoid that is superior to isotretinoin; however, it is not approved by Health Canada and is not listed on provincial cancer formularies. Advanced-stage MF involving tumours may be treated with local radiotherapy and systemic therapies similar to those used for limited-stage disease. Advanced MF with extra-cutaneous nodal or visceral involvement requires systemic therapies such as chemotherapy (gemcitabine, or liposomal doxorubicin). The experts expressed that many novel drugs that are recommended by international guidelines, such as vorinostat, romidepsin, and pralatrexate, are not funded in Canada. Brentuximab was recently approved for use in CD30-expressing MF, limited or advanced stage, that has relapsed following 1 of these systemic approaches. In SS, a front-line approach includes twice-monthly ECP, which is only available in some specialized urban centres. Systemic therapies such as interferon and isotretinoin are typically added upfront, then further treatments are limited by available funded options. This may include romidepsin, alemtuzumab, or pembrolizumab.
The experts noted that mogamulizumab was not studied in the upfront setting and that it would be used, as in the clinical trial, in the second line and beyond in patients who were intolerant or whose condition was refractory to first-line systemic treatments. The experts stated that some patients derive benefit from existing therapies before ultimately progressing and requiring an alternative treatment; thus, they agreed that other systemic treatments should be attempted before mogamulizumab, based on availability. Currently, mogamulizumab is only available to patients via special access programs. The experts agreed it would likely cause a shift in current therapy and would be considered after chemotherapy, brentuximab (in patients with CD30-positive disease), or interferon for advanced MF, or following interferon and/or a retinoid (isotretinoin) for SS.
The clinical experts highlighted the mechanism of action of mogamulizumab, considering its modification of the underlying disease mechanism, and the potential impact of its concurrent use with ECP.
Patient Population
The approach for managing MF and SS is individualized based on the extent of disease or stage, the goals of therapy, access to treatment, patient tolerance, and practical considerations related to administration. The experts noted that diagnosis can be challenging, often taking approximately 3 years, with misdiagnosis occurring frequently early on. As such, the experts agreed there was no pre-symptomatic testing that would identify the patients best suited for treatment with mogamulizumab.
The experts highlighted that the group of patients most suitable for mogamulizumab are those with advanced-stage MF (stage IIIB, IVA, IVB) with or without blood involvement, or SS that has failed to respond to front-line systemic therapy. These patients are also at the highest need of intervention, as they typically have the greatest symptom burden and a worse prognosis. Overall, the clinical experts felt that aside from patients with stage IA, who were not eligible for the MAVORIC trial, funding criteria should not include staging information, as the primary outcome of the MAVORIC trial demonstrated improved PFS across all included stages (IB to IV) and was not powered to detect differences by stage.
Experts expressed that currently available treatment options have suboptimal response rates and duration. Also, these patients sometimes cannot access TSEBT or ECP, so systemic therapy would be a better option. They are a subpopulation of the broader group of patients with limited-stage MF, who predicably progress to advanced stages over many years or decades.
According to the clinical experts, the patients not suitable for treatment with mogamulizumab are those who are currently benefiting from first-line treatment and those who are scheduled to have an allogenic transplant within 3 months of receiving the drug. It was also noted by the clinical experts that patients with stage IA MF were not included in the pivotal clinical trial, so should not be treated with mogamulizumab, as these patients, and those with stage IB, IIA, or IIB (as evidenced by the response outcomes demonstrated on study), are less likely to derive benefit due to the mechanism of action of mogamulizumab.
Assessing Response to Treatment
In general, the outcomes assessed in clinical practice reflect those typically measured in clinical trials using the defined response criteria for MF and SS found in the literature.31 One expert noted that response in clinical practice should be assessed monthly or every 15 days initially. Clinically meaningful response is generally measured as compartmentalized and composite responses, with a reduction in the severity of symptoms and an increase in PFS considered most important. An objective improvement of skin typically corresponds with improvement of other measures of disease burden in blood, lymph nodes, and viscera. Skin examination is assessed by an individual with expertise, generally using the modified Severity-Weighted Assessment Tool (mSWAT) score (a validated assessment tool).31 Subjective improvement in QoL assessments (e.g., Skindex-29) can be considered a clinically meaningful end point, with improvement in specific symptom parameters such as pruritis (itch) considered very crucial for some patients living with MF or SS. Additional serial diagnostic imaging, in the case of nodal and other extra-cutaneous disease compartments, and complete blood count (with or without flow cytometry for the count of Sézary cells) were noted as tools by the experts to determine response.
Discontinuing Treatment
According to the clinical experts, major factors to be considered when discontinuing treatment include disease progression in any of the 4 disease compartments (skin, lymph nodes, viscera, and blood). The experts noted there are published criteria for PD, in which case, discontinuation would be advised. Additionally, the experts noted that intolerable AEs, notably mogamulizumab-associated rash that does not respond to management algorithms, or other unacceptable AEs would be a reason to discontinue. It was proposed by the experts that one may consider continuing therapy with stable disease if the treatment is tolerated.
Prescribing Conditions
The clinical experts stated that the diagnosis of CTCL (MF and SS) is complex and requires clinicopathological correlation by experts in the field. Patients are most likely identified and under the care of a multidisciplinary group of doctors that may include but is not limited to hematologists, medical oncologists, dermatologists, radiation oncologists, hematopathologists, and dermatopathologists. Experts believe that mogamulizumab would typically be administered in an outpatient, ambulatory setting at an oncology or infusion centre under the supervision of trained personnel.
Clinician Group Input
This section was prepared by CADTH staff based on the input provided by clinician groups.
The clinician group input was provided by 2 clinician groups: a network of Canadian cutaneous lymphoma providers and the Ontario Health (Cancer Care Ontario) Hematology Cancer Drug Advisory Committee. Clinician groups noted the individualized approach to treatment as well as the lack of a defined standard of care for MF or SS. Along with suboptimal response rates and the limited duration with current treatments, access issues were cited as a major unmet need in this population. The clinician groups highlighted the lack of publicly funded treatments in Canada and noted that treatment selection is guided more by which drugs are funded and available rather than necessarily the one with the best chances of response and fewest side effects. The clinician groups stated that patients should be offered mogamulizumab if they meet the eligibility criteria from the clinical trial, with 1 clinician group highlighting its use in advanced-stage MF (with or without blood involvement) or SS, and that mogamulizumab would likely be used in the second line following chemotherapy, brentuximab vedotin (if CD30-positive), or interferon. One clinician group considered the potential for mogamulizumab’s concurrent use with ECP, particularly for patients with SS. Important goals of treatment, as well as outcomes of treatment considered clinically meaningful, include disease control via a reduction in the frequency or severity of symptoms, an improvement in symptoms or stabilization of disease, prolonged survival, and an improvement in QoL. Lastly, the clinician groups noted that mogamulizumab would be received in an outpatient setting and highlighted that disease progression, AEs, and treatment-related toxicity should be considered when deciding to discontinue treatment.
Drug Program Input
The drug programs provide input on each drug being reviewed through CADTH’s reimbursement review processes by identifying issues that may impact their ability to implement a recommendation. The implementation questions and corresponding responses from the clinical experts consulted by CADTH are summarized in Table 5.
The Provincial Advisory Group’s implementation questions and corresponding responses from the clinical experts consulted by CADTH are summarized in Table 5.
Table 5: Summary of Drug Plan Input and Clinical Expert Response
Implementation issues and questions from the drug programs | Clinical experts’ response |
|---|---|
Relevant comparators | |
The comparator in the pivotal trial, MAVORIC, is vorinostat, which is not publicly funded in Canada. It is noted the pERC conditionally recommended brentuximab vedotin for the treatment of adult patients with CD30-positive MF who have had 1 prior systemic therapy in December 2020. This treatment is funded in some provinces. There is no standard of care for the treatment of MF or SS in patients who progress after 1 prior systemic therapy. Public funding of treatments varies across provinces. | No response required. For consideration by pERC. |
How does mogamulizumab compare with brentuximab vedotin for patients with CD30-positive MF? Should these drugs be sequenced and, if so, is there a preferred order? | CD30 status was not assessed in the MAVORIC study. Patients are required to have CD30-positive immunohistochemical expression for treatment with brentuximab vedotin; however, this is not broadly expressed in patients with MF or SS. For CD30-positive MF, brentuximab vedotin would be sequenced ahead of mogamulizumab; otherwise, mogamulizumab would be given ahead of brentuximab. CD30 is seldomly expressed in SS, and patients with SS are not eligible for brentuximab under Canadian funding models. |
Considerations for initiation of therapy | |
The MAVORIC trial enrolled patients with stages IB to IV previously treated with systemic therapy. Should funding criteria include staging information? | Aside from patients with stage IA, who were not eligible for the MAVORIC trial, funding criteria should not include staging information, as the primary outcome of the MAVORIC trial demonstrated improved PFS across all included stages (IB to IV) and was not powered to detect differences by stage. However, the clinical experts noted that there was a clearer benefit observed in the MAVORIC trial for advanced stages (III to IV). |
MAVORIC enrolled patients with an ECOG PS of 0 to 1. Is it reasonable to treat patients with an ECOG PS of 2 or greater with mogamulizumab? | Patients with an ECOG PS of 2 were not eligible for the MAVORIC trial, which clinical experts noted may not be reflective of Canadian clinical practice for MF and SS, particularly for those with advanced-stage and multiply relapsed disease. One expert clarified that higher ECOG PS scores could be a result of extensive disease or infections. Thus, the experts stated that patients with an ECOG PS of 2 should be treated with mogamulizumab only at the discretion of the treating physician. |
MAVORIC excluded patients with CNS metastasis, significant cardiac disease (class III or IV NYHA) and large-cell transformation. Should these patients be eligible for mogamulizumab? | It is currently unclear whether patients with CNS metastases or significant cardiac disease should be excluded from using mogamulizumab. Due to safety concerns, prescribing for advanced heart failure is a concern, and mogamulizumab would not be prescribed in the absence of future or additional data supporting use in patients with CNS metastases. Physicians should use discretion in these populations. Large-cell transformation is a clinical challenge in the management of MF, with no standard of care and a generally poor prognosis. Patients with large-cell transformation should be considered for mogamulizumab, provided they meet other eligibility. |
The majority of patients in the MAVORIC trial had disease that failed to respond to more than 1 prior systemic therapy. Should patients be required to have failed more than 1 prior systemic therapy to be eligible for mogamulizumab? | In line with the MAVORIC trial, patients in Canada are likely to have received multiple prior systemic therapies. There is no concern surrounding the use of mogamulizumab in patients whose disease failed multiple systemic therapies. |
In MAVORIC, patients with a global complete response could continue treatment for up to 12 months or until progression, whichever came first. Upon relapse, would these patients be eligible for re-treatment and if so, if there a reasonable time frame (i.e., patients must have been off therapy for a minimum time frame to be eligible for re-treatment)? | There is currently no evidence to suggest re-treatment with mogamulizumab in patients who relapse. However, patients with cutaneous lymphoma can be re-treated with prior therapies and still achieve a response. In MF and SS, this is an unlikely scenario to pursue for someone relapsing after mogamulizumab due to prognosis, practicality, and cost considerations. If mogamulizumab treatment is simply paused (< 3 months) due to intercurrent illness or other complications, it would be reasonable to restart once these issues are resolved. |
Considerations for prescribing of therapy | |
Mogamulizumab is supplied as 20 mg vials with potential for wastage. | No response required. For consideration by pERC. |
Generalizability | |
Should patients currently receiving a second-line systemic therapy be eligible to switch to mogamulizumab? | If current treatment is effective and well tolerated, switching to mogamulizumab is not required. In line with the clinical trial, patients would be eligible to switch following the failure of systemic therapy. |
Funding algorithm | |
Drug may change place in therapy of comparator drugs. Brentuximab vedotin for the treatment of adult patients with CD30-positive MF who have had 1 prior systemic therapy. | No response required. For consideration by pERC. |
Care provision issues | |
Potential for infusion reactions (grade 1 or 2 incidence rate of 32%; grade 3 incidence rate of 2%). Drug rashes are common and must be monitored for. | No response required. For consideration by the pERC. |
System and economic issues | |
Brentuximab vedotin for previously treated CD30+ MF has confidential pricing (pCPA). | No response required. For consideration by pERC. |
CNS = central nervous system; ECOG PS = Eastern Cooperative Oncology Group Performance Status; HC = Health Canada; MF = mycosis fungoides; NYHA = New York Heart Association; pCPA = pan-Canadian Pharmaceutical Alliance; pERC = CADTH pan-Canadian Oncology Drug Review Expert Review Committee; PFS = progression-free survival; SS = Sézary syndrome.
Clinical Evidence
The clinical evidence included in the review of Poteligeo (mogamulizumab) is presented in 3 sections. The first section, the systematic review, includes pivotal studies provided in the sponsor’s submission to CADTH and Health Canada, as well as those studies that were selected according to an a priori protocol. The second section includes indirect evidence from the sponsor and indirect evidence selected from the literature that met the selection criteria specified in the review. The third section includes sponsor-submitted long-term extension studies and additional relevant studies that were considered to address important gaps in the evidence included in the systematic review.
Systematic Review (Pivotal and Protocol Selected Studies)
Objectives
To perform a systematic review of the beneficial and harmful effects of mogamulizumab 1.0 mg/kg for the treatment of adults with MF or SS who have received at least 1 prior systemic therapy.
Methods
Studies selected for inclusion in the systematic review included pivotal studies provided in the sponsor’s submission to CADTH and Health Canada, as well as those meeting the selection criteria presented in Table 6. Outcomes included in the CADTH review protocol reflect outcomes considered to be important to patients, clinicians, and drug plans.
Of note, the systematic review protocol presented in Table 6 was established before the granting of an NOC from Health Canada.
Table 6: Inclusion Criteria for the Systematic Review
Criteria | Description |
|---|---|
Population | Adult patients with MF or SS who have received at least 1 prior systemic therapy. Subgroups:
|
Intervention | Mogamulizumab 1.0 mg/kg IV infusion |
Comparatora | Retinoids:
Single- or multi-drug chemotherapy:
HDACi:
Other targeted therapies:
Other:
|
Outcomes | Efficacy outcomes:
Harms Outcomes:
|
Study designs | Published and unpublished phase III and IV RCTs |
AE = adverse event; BOR = best overall response; CHEOP = cyclophosphamide, doxorubicin, vincristine, prednisone, etoposide; CHOP = cyclophosphamide, doxorubicin, vincristine, prednisone; CR = complete response; DOR = duration of response; ECP = extracorporeal photopheresis; HDACi = histone deacetylase inhibitor; HRQoL = health-related quality of life; IRR = infusion-related reaction; MF = mycosis fungoides; ORR = overall response rate; OS = overall survival; PFS = progression-free survival; PR = partial response; RCT = randomized controlled trial; SAE = serious adverse event; SCT = stem-cell therapy; SS = Sézary syndrome; TSEBT = total skin electron beam therapy; TTF = time-to-treatment failure; TTR = time to response; vs. = versus; WDAE = withdrawal due to adverse event.
aOf note, CADTH acknowledges differences in reimbursement status across jurisdictions as well as off-label use.
bHighlighted in the patient group input as important outcomes.
The literature search was performed by an information specialist using a peer-reviewed search strategy according to the PRESS Peer Review of Electronic Search Strategies checklist.41
Published literature was identified by searching the following bibliographic databases: MEDLINE All (1946—) through Ovid and Embase (1974—) through Ovid. All Ovid searches were run simultaneously as a multi-file search. Duplicates were removed using Ovid deduplication for multi-file searches, followed by manual deduplication in Endnote. The search strategy comprised both controlled vocabulary, such as the National Library of Medicine’s MeSH (Medical Subject Headings), and keywords. The main search concept was mogamulizumab. Clinical trial registries were searched: the US National Institutes of Health’s clinicaltrials.gov, Health Canada’s Clinical Trials Database, and the European Union Clinical Trials Register.
No filters were applied to limit the retrieval by study type. Retrieval was not limited by publication date or by language. Conference abstracts were excluded from the search results. See Appendix 1 for the detailed search strategies.
The initial search was completed on October 12, 2021. Regular alerts updated the search until the meeting of the CADTH pan-Canadian Oncology Drug Review Expert Committee (pERC) on February 9, 2022.
Grey literature (literature that is not commercially published) was identified by searching relevant websites from the Grey Matters: A Practical Tool For Searching Health-Related Grey Literature tool.42 Included in this search were the websites of regulatory agencies (US FDA and European Medicines Agency). Google was used to search for additional internet-based materials. See Appendix 1 for more information on the grey literature search strategy.
Two CADTH clinical reviewers independently selected studies for inclusion in the review based on titles and abstracts, according to the predetermined protocol. Full-text articles of all citations considered potentially relevant by at least 1 reviewer were acquired. Reviewers independently made the final selection of studies to be included in the review, and differences were resolved through discussion.
Findings From the Literature
A total of 376 studies were identified from the literature for inclusion in the systematic review (Figure 1). The included studies are summarized in Table 7. A list of excluded studies is presented in Appendix 2.
Table 7: Details of Included Studies
Detail | MAVORIC (Study 0761-010) |
|---|---|
Designs and populations | |
Study design | Phase III, open-label RCT |
Locations | Australia, Denmark, France, Germany, Italy, Japan, the Netherlands, Spain, Switzerland, UK, and US |
Patient enrolment dates | December 12, 2012, to January 29, 2016 |
Randomized (N) | 372 |
Inclusion criteria |
|
Exclusion criteria |
|
Drugs | |
Intervention | Mogamulizumab 1.0 mg/kg administered as an IV infusion over at least 1 hour on days 1, 8, 15, and 22 of the first cycle and on days 1 and 15 of subsequent cycles of each 28-day cycle |
Comparator(s) | Vorinostat 400 mg once daily orally beginning on day 1 of each 28-day cycle |
Duration | |
Phase | |
Screening | 30 days before the start of study treatment |
Treatment | 28-day cycles for mogamulizumab and vorinostat regimens |
Follow-up | Until disease progression or treatment discontinuation |
Outcomes | |
Primary end point | PFS defined as the time from the day of randomization to a treatment arm until documented progression or death due to any cause |
Secondary and exploratory end points | Key secondary end points:
Other secondary end points:
Exploratory end points:
|
Notes | |
Publications | Kim et al. (2018)43 |
ALT = alanine aminotransferase; ANC = absolute neutrophil count; AST = aspartate aminotransferase; CNS = central nervous system; CTCL = cutaneous T-cell lymphoma; DBP = diastolic blood pressure; DOR = duration of response; ECOG PS = Eastern Cooperative Oncology Group Performance Status; EQ-5D-3L = 3-level EQ-5D; FACT-G = Functional Assessment of Cancer Therapy–General; HRQoL = health-related quality of life; HTLV-1 = human T-cell lymphotropic virus; IL-2 = interleukin-2; LCT = large-cell transformation; MF = mycosis fungoides; NYHA = New York Heart Association; ORR = objective response rate; OS = overall survival; PFS = progression-free survival; PUVA = psoralen plus UVA light therapy; RCT = randomized controlled trial; SBP = systolic blood pressure; SS = Sézary syndrome; TTF = time-to-treatment failure; TTR = time to response; ULN = upper limit of normal.
Source: MAVORIC (0761-010) Clinical Study Report.27
Description of Studies
One study was included in the review. The MAVORIC study (Study 0761-010) is an open-label, multi-centre, phase III RCT evaluating the effectiveness of mogamulizumab compared with vorinostat in patients with CTCL whose disease has failed to respond to at least 1 prior course of systemic therapy. The primary objective of MAVORIC was to compare the PFS of mogamulizumab versus vorinostat for patients with relapsed or refractory CTCL.27
Using the ClinTrak interactive voice or web response system (CTIVRS), a total of 372 patients were randomized 1:1 via screening numbers assigned by the investigator (or designee) to either mogamulizumab 1.0 mg/kg IV infusion on days 1, 8, 15, and 22 of the first cycle and on days 1 and 15 of subsequent cycles (n = 186), or vorinostat 400 mg by mouth once daily on day 1 of each 28-day cycle (n = 186). Randomization was stratified by disease type (MF or SS) and disease stage (IB or II versus stage III or IV). One-way crossover from vorinostat to mogamulizumab was permitted in the case of disease progression or intolerable toxicity. Screening assessments were performed within 30 days before the start of study treatment. The clinical data cut-off for the final analysis of the MAVORIC study was December 31, 2016. No interim analyses were conducted.27 MAVORIC was conducted in 11 countries; however, no Canadian investigative sites were included.
Populations
Inclusion and Exclusion Criteria
The inclusion and exclusion criteria for the MAVORIC trial are summarized in Table 7. Patients were required to have a histologically confirmed diagnosis of MF or SS; stage IB, IIA, IIB, III, or IV disease that had failed to respond to at least 1 prior systemic therapy; and an ECOG Performance Status of 0 or 1.27
Patients with stage IA were not included in the MAVORIC trial. Due to the potential increased risk of post-transplant complications (i.e., graft-versus-host disease), patients were excluded from entry into the MAVORIC trial if they had already received an allogenic hematopoietic stem-cell transplant. Patients who received a transplant after treatment with mogamulizumab are being followed for details and outcome for at least 180 days post transplant.27
Baseline Characteristics
Baseline characteristics for the MAVORIC trial are summarized in Table 8. Demographic and baseline characteristics of the ITT population were well balanced between arms: the median patient age was 64 years, 58.1% of patients were male and 69.9% were White. There was a higher representation of patients who were Black or African American in the mogamulizumab group compared with the vorinostat group (12.9% versus 7.0%). The majority of patients in each group had an ECOG Performance Status of 0 (57.0% and 55.9% in the mogamulizumab and vorinostat arms, respectively).27
In the mogamulizumab group, 105 (56.5%) and 81 (43.5%) patients had MF or SS, respectively, while 99 (53.2%) and 87 (46.8%) patients in the vorinostat group had MF or SS. The proportion of patients with various clinical stages was well balanced between arms. There were 140 patients (37.7%) with stage IB to IIB, while 232 (62.4%) had stage IIIA to IV. The majority of patients had IVA1: 82 (44.1%) versus 73 (39.2%) in the mogamulizumab and vorinostat arms, respectively. All patients in both arms had skin involvement, while 124 (66.7%) and 122 (65.6%) in the mogamulizumab group had nodal and blood involvement at baseline compared with 122 patients (65.6%) for each category in the vorinostat group. Visceral involvement was rare, occurring in 6 patients overall.27
The population in the MAVORIC trial in each treatment arm had received a median of 3 prior systemic therapies. In total, 68 patients (18.3%) received 1 prior systemic therapy, 155 (41.7%) had received 2 to 3 prior systemic therapies, and 148 (39.9%) had received 4 or more prior systemic therapies.27
Table 8: Summary of Baseline Characteristics (ITT)
Characteristic | MAVORIC | |
|---|---|---|
Mogamulizumab (N = 186) | Vorinostat (N = 186) | |
Age (years) | ||
Mean (SD) | 62.8 (13.34) | 63.3 (12.58) |
Median (range) | 63.5 (25 to 101) | 65.0 (25 to 89) |
< 65 years | 99 (53.2) | 89 (47.8) |
≥ 65 years | 87 (46.8) | 97 (52.2) |
Gender, n (%) | ||
Male | 109 (58.6) | 107 (57.5) |
Female | 77 (41.4) | 79 (42.5) |
Race, n (%) | ||
White | 125 (67.2) | 135 (72.6) |
Asian | 12 (6.5) | 7 (3.8) |
Black or African American | 24 (12.9) | 13 (7.0) |
Native American or Alaska Native | 0 | 1 (0.5) |
Native Hawaiian or other Pacific Islander | 1 (0.5) | 0 |
Other | 0 | 5 (2.7) |
Not applicablea | 24 (12.9) | 25 (13.4) |
ECOG PS, n (%) | ||
0 | 106 (57.0) | 104 (55.9) |
1 | 78 (41.9) | 82 (44.1) |
2 | 2 (1.1)b | 0 |
Disease type, n (%) | ||
MF | 105 (56.5) | 99 (53.2) |
SS | 81 (43.5) | 87 (46.8) |
Clinical stage, n (%) | ||
IB | 15 (8.1) | 27 (14.5) |
IIA | 21 (11.3) | 22 (11.8) |
IIB | 32 (17.2) | 23 (12.4) |
IIIA | 9 (4.8) | 9 (4.8) |
IIIB | 13 (7.0) | 7 (3.8) |
IVA1 | 73 (39.2) | 82 (44.1) |
IVA2 | 19 (10.2) | 12 (6.5) |
IVB | 4 (2.2) | 4 (2.2) |
Disease site, n (%) | ||
Skin | 186 (100.0) | 186 (100.0) |
Nodes | 124 (66.7) | 122 (65.6) |
Viscera | 3 (1.6) | 3 (1.6) |
Blood | 122 (65.6) | 122 (65.6) |
Other (includes bone marrow) | 13 (7.0) | 7 (3.8) |
CCR4 expression status, n (%) | ||
≥ 10% CCR4 expression | 134 (72.0) | 146 (78.5) |
< 10% CCR4 expression | 6 (3.2) | 4 (2.2) |
Missing (no sample or test failure) | 46 (24.7) | 36 (19.4) |
LDH (u/L) | ||
Mean (SD) | 341.2 (250.0) | 302.2 (187.32) |
Median (range) | 255.0 (136 to 1986) | 245.0 (121 to 1,432) |
Number of prior systemic regimens received, n (%) | ||
Mean (SD) | 4.1 (3.17) | 3.4 (2.34) |
Median (range) | 3.0 (1 to 18) | 3.0 (0 to 14) |
0 | 0 | 1 (0.5) |
1 | 28 (15.1) | 40 (21.5) |
2 | 40 (21.5) | 38 (20.4) |
3 | 40 (21.5) | 37 (19.9) |
4 | 22 (11.8) | 18 (9.7) |
5 | 12 (6.5) | 21 (11.3) |
≥ 6 | 44 (23.7) | 31 (16.7) |
Best response to last systemic CTCL therapy before study entry, n (%) | ||
Complete response or partial response | 62 (33.3) | 69 (37.1) |
Stable disease | 46 (24.7) | 32 (17.2) |
Progressive disease | 59 (31.7) | 67 (36.0) |
Not applicable | 2 (1.1) | 3 (1.6) |
Unknown | 17 (9.1) | 15 (8.1) |
CCR4 = CC chemokine receptor 4; CTCL = cutaneous T-cell lymphoma; ECOG PS = Eastern Cooperative Oncology Group Performance Status; ITT = intention-to-treat population; LDH = lactate dehydrogenase; MF = mycosis fungoides; SD = standard deviation; SS = Sézary syndrome.
aNot reported for those countries that do not allow race or ethnicity to be collected.
bTwo patients had an ECOG PS of 1 at pre-treatment but an ECOG PS of 2 on cycle 1, day 1.
Source: MAVORIC (0761 to 010) Clinical Study Report.27
Interventions
Mogamulizumab
Mogamulizumab was administered via IV infusion on an outpatient basis at a dose of 1.0 mg/kg over at least 1 hour on days 1, 8, 15, and 22 of the first cycle and on days 1 and 15 of subsequent cycles until disease progression according to a global composite scoring system or unacceptable toxicity. Each treatment cycle was 28 days. Patients were observed for 1 hour following administration.27
The start of a new cycle could be delayed for up to 2 weeks after completion of the prior cycle to allow resolution of treatment-related toxicities or for non-medical reasons. A delay of the start of a new cycle of greater than 2 weeks was to be discussed with the medical monitor or designee. No dose modifications of mogamulizumab were permitted in this study.27
Mogamulizumab was given under the direct supervision of study personnel. A weight-based volume of mogamulizumab was diluted into 250 mL of saline (0.9% sodium chloride) and then filtered through a 0.22 μm protein-sparing (low protein binding) in-line filter.27
It was recommended that patients be premedicated with acetaminophen or paracetamol orally and diphenhydramine 50 mg IV (or equivalent antihistamine) before the first mogamulizumab infusion.27
Vorinostat
Vorinostat was administered on an outpatient basis at a dose of 400 mg orally once daily with food (4 × 100 mg capsules) until disease progression or unacceptable toxicity. Each treatment cycle was 28 days. The prescribing information used in the US was followed for recommendations regarding dose modification of vorinostat, including dose omission, reduction, or interruption for management of severe or intolerable adverse reactions.27
Patients randomized to the vorinostat arm who had received 2 full treatment cycles and demonstrated progression of disease at the 8-week (cycle 2, day 26 to 28) assessment, or anytime thereafter, were allowed to cross over to treatment with mogamulizumab after discussion with the medical monitor or designee and receipt of approval from the sponsor. All summaries for the crossover portion were purely descriptive. No statistical comparisons were made for data collected from patients who crossed over from vorinostat to mogamulizumab.27
Patients were required to return pill bottles at each study visit. Any unused capsules were counted.27
Prior and Concomitant Therapy
While on study treatment, patients were not permitted to receive any experimental therapy or anti-cancer therapy other than the study medication. Any patient requiring other anti-cancer therapy was to be removed from study treatment.27
Permitted treatments during the study included:27
hematopoietic growth factors and erythropoiesis-stimulating drugs
patients taking low-dose systemic corticosteroid (≤ 20 mg prednisone equivalent) for at least 4 weeks before the pre-treatment visit were allowed to continue use; patients were allowed to receive intra-articular corticosteroid injections; intraocular, inhaled, or nasal corticosteroids; and replacement doses of systemic corticosteroids, as needed
patients on a stable dose of medium or low-potency topical corticosteroids for at least 4 weeks before the pre-treatment visit could continue use at the same dose, although the investigator was to attempt to taper the medication to the lowest dosage tolerable while on study
prophylactic treatment for infectious complications of T-cell lymphoma was permitted at the discretion of the investigator
patients receiving vorinostat and concomitant therapy with coumarin-derivative anticoagulants were to be monitored for prolongation of prothrombin time and international normalized ratio results
influenza vaccination consisting of killed virus or viral particles was allowed to be administered to the patients in the study.
Initiation of treatment with systemic steroids or an increase in the dose of steroids while on study was not permitted except to treat an infusion reaction. Patients who required systemic steroid therapy to treat a severe skin rash were to be discontinued from the study. Initiation of treatment with topical corticosteroids while on study was not permitted, except to treat an acute rash.27
Other prohibited treatments during the study included:27
other concurrent HDAC inhibitors including valproic acid
immunomodulatory drugs such as methotrexate, azathioprine, IV immunoglobulin, cyclophosphamide, cyclosporine, mycophenolate, infliximab, etanercept, leflunomide, adalimumab, lenalidomide, abatacept, rituximab, anakinra, interferon alpha, interferon beta, interleukin-2, and natalizumab
any experimental therapy or anti-cancer therapy, including radiation and phototherapy, other than the study medications
any live or live attenuated vaccine
alternative medicines, particularly St. John’s wort.
The prior CTCL medications used by patients at baseline are summarized in Table 9.
Table 9: Prior CTCL Therapy (ITT)
Characteristic | MAVORIC | |
|---|---|---|
Mogamulizumab (N = 186) | Vorinostat (N = 186) | |
Type of prior therapy received, n (%) | ||
Skin-directed therapies | ||
PUVA | 80 (43.0) | 63 (33.9) |
Topical steroid | 67 (36.0) | 65 (34.9) |
Bexarotene, topical | 11 (5.9) | 6 (3.2) |
Systemic therapies | ||
Bexarotene | 107 (57.5) | 110 (59.1) |
Interferon alpha | 81 (43.5) | 94 (50.5) |
Methotrexate | 69 (37.1) | 73 (39.2) |
Extracorporeal photopheresis | 71 (38.2) | 65 (34.9) |
Romidepsin | 45 (24.2) | 32 (17.2) |
Nitrogen mustard | 28 (15.1) | 40 (21.5) |
Doxorubicin HCL liposome | 23 (12.4) | 19 (10.2) |
Pralatrexate | 14 (7.5) | 13 (7.0) |
Carmustine | 13 (7.0) | 13 (7.0) |
Brentuximab vedotin | 16 (8.6) | 4 (2.2) |
Denileukin diftitox | 5 (2.7) | 3 (1.6) |
Chlorambucil | 3 (1.6) | 4 (2.2) |
Etoposide | 3 (1.6) | 4 (2.2) |
IL-12 | 0 | 1 (0.5) |
Other (skin-directed and systemic) | 131 (70.4) | 121 (65.1) |
Prior radiotherapy, n (%) | ||
No | 131 (70.4) | 134 (72.0) |
Yes | 55 (29.6) | 52 (28.0) |
CTCL = cutaneous T-cell lymphoma; HCL = hydrochloride; IL-12 = interleukin-12; ITT = intention-to-treat population; PUVA = psoralen plus UVA light therapy.
Source: MAVORIC (0761 to 010) Clinical Study Report.27
Outcomes
A list of efficacy end points identified in the CADTH review protocol that were assessed in the clinical trials included in this review is provided in Table 10. These end points are further summarized after the table. A detailed discussion and critical appraisal of the outcome measures is provided in Appendix 4.
Table 10: Summary of Outcomes of Interest in MAVORIC
Outcome measure | MAVORIC |
|---|---|
PFS | Primary end point |
ORR | Key secondary end point |
HRQoL (Skindex-29, FACT-G, EQ-5D) | Key secondary end point |
BOR | Secondary end point |
DOR | Secondary end point |
TTR | Secondary end point |
Safety | Secondary end point |
OS | Exploratory end point |
TTF | Exploratory end point |
BOR = best overall response; DOR = duration of response; EQ-5D-3L = 3-Level EQ-5D; FACT-G = Functional Assessment of Cancer Therapy–General; HRQoL = health-related quality of life; ORR = objective response rate; OS = overall survival; PFS = progression-free survival; TTF = time-to-treatment failure; TTR = time to response.
Primary Efficacy Outcome
The primary outcome of the MAVORIC trial was investigator-assessed PFS, defined as the time from randomization until documented PD in any compartment per the global composite CTCL response criteria (Table 11 and Table 12) or death due to any cause. The data cut-off date for the MAVORIC study was December 31, 2016. The primary efficacy analysis of the MAVORIC trial was conducted on randomized patients and does not include patients who crossed over from vorinostat to receive mogamulizumab.27
Patients who discontinued study treatment for reasons other than disease progression were contacted every 3 months (± 14 days) until documented disease progression or death, or initiation of alternative therapy. Patients who discontinued treatment after achieving a complete response (CR) were assessed every 8 weeks (± 14 days) for the first 6 months (if a CR was achieved before 1 year on study) and then every 16 weeks (± 14 days) thereafter until progression. All patients or their referring physician were contacted every 3 months (± 14 days) to ascertain OS status.27
The following censoring rules were applied for PFS:27
In the event that a randomized patient withdrew from the study for any reason before documented progression, the time from the day of randomization to the last post-baseline tumour assessment from any compartment (skin, blood, bone marrow, lymph nodes, or viscera) was used as a censored time point.
For patients who were randomized to a treatment arm but had an unknown baseline assessment for a compartment, the PFS time was censored at the randomization date if there was no post-baseline tumour assessment for that compartment or if there was any evidence of lymphoma in that compartment at the post-baseline evaluation.
For patients randomized to a treatment arm who withdrew from treatment before the first post-baseline tumour assessment for any reason other than disease progression, the PFS time was censored at the last documented visit.
For patients who initiated a new anti-cancer therapy (including crossover to mogamulizumab) in the absence of a PFS event, the PFS time was censored at the last tumour assessment (for any compartment) before the start of the new anti-cancer therapy.
For patients not known to have died or have documented progression as of the data cut-off date for the primary analysis, the PFS time was censored at the date of the last tumour assessment (for any compartment) before data cut-off.
Secondary Efficacy Outcomes
The key secondary outcomes of the MAVORIC trial included investigator-assessed ORR (i.e., a confirmed CR or partial response [PR]) and change from baseline in HRQoL.27
Clinical Response (Overall Response Rate)
Prior to the start of treatment, skin disease (lesions and erythema) and blood were evaluated by the investigator using the mSWAT, and flow cytometry. The mSWAT is an objective, quantitative, severity-weighted method to assess the extent of skin lesions. Assessment of disease in skin and blood was evaluated at the end of each cycle. Further details on the mSWAT are reported in Appendix 4. Assessment of disease in lymph nodes and viscera was conducted by CT scan) and was performed at the end of cycle 1 (4 weeks), and every 8 weeks thereafter during the first year of treatment, and every 16 weeks thereafter. ORR was defined as the proportion of patients who were responders (confirmed CR or PR) based on investigator’s assessment per a global composite response score (Table 11) that was based on a response in each compartment (skin, blood, lymph nodes, and viscera) that was subsequently confirmed by 2 or more consecutive observations for a minimum of 4 weeks. The global composite response was assessed at 4 weeks (end of cycle 1) and every 8 weeks thereafter (cycle 3, 5, and so forth). After the first year, global composite response was assessed every 16 weeks (cycle 17, 21, and so forth). Individual criteria for measurement of response in skin, as determined by changes in mSWAT score, blood, viscera, and lymph nodes are summarized in Table 12. All individual response outcomes were investigator-assessed. Node, viscera, and blood response contributed toward global response only if disease was present at baseline unless there was global PD, which was defined as new disease in a previously uninvolved compartment.27
Patients who met the criteria for PD in any compartment, or had a relapse in the blood, lymph nodes, or viscera confirmed at 2 consecutive visits, were to be discontinued from study treatment due to disease progression or relapse, and the overall global composite score was determined at that time, even if not all compartments had been assessed. If clinical progression was noted at any time before the scheduled assessments for efficacy, the assessments (CT, mSWAT, skin photographs, and flow cytometry) were done at that time to fully document disease progression.27
As per the study’s published global composite response criteria, in cases where the definition of PD or relapse was met but the clinical impression was questionable, the patient could remain on study for at least 4 weeks to avoid them being removed prematurely from the study. Clinical progression was considered for patients with disease progression that did not meet the criteria for PD based on CTCL response criteria. If PD was confirmed at the subsequent evaluation, the patient was to be discontinued from treatment. If PD was not confirmed at the subsequent evaluation, the patient could remain on study. If patients experienced an overall CR, treatment could continue for up to 12 months or until PD, whichever came first.27
Additionally, a blinded independent review of data for each patient was performed by an independent review committee to determine progression and date of progression. The independent reviewer was supplied with the following: results of the independent radiology review (assessment of lymph nodes and viscera), mSWAT score as assessed by the investigator, results of central flow cytometry, results of biopsies if appropriate for assessment of disease, and date of death for patients who died before progression. The independent reviewer also had access to CT scans and skin photographs. The reviewer also reviewed the date of any objective responses (CR or PR).27
Table 11: Global Composite Scoring System
Global score | Definition | Skin | Nodes | Viscera | Blood |
|---|---|---|---|---|---|
CR | Complete disappearance of all clinical evidence of disease | CR | All categories have CR/NI | ||
PR | Regression of measurable disease | CR | All categories do not have a CR/NI and no category has a PD | ||
PR | No category has a PD and if any category involved at baseline, at least 1 of the 3 categories has a CR or PR | ||||
SD | Failure to attain CR, PR, or PD | PR | No category has a PD and if any other category involved at baseline, no CR or PR in any | ||
SD | CR/NI, PR, SD in any category and no category has a PD | ||||
PD | Progressive disease | PD in any category | |||
Relapse | Recurrence of disease in prior CR | Relapse in any category | |||
CR = complete response; NI = non-involved; PD = progressive disease; PR = partial response; SD = stable disease.
Source: MAVORIC (0761 to 010) Clinical Study Report.27
Table 12: Response Criteria for Skin, Blood, Viscera, and Lymph Nodes
Response | Skina | Bloodb | Viscera | Lymph nodes |
|---|---|---|---|---|
CR | 100% clearance of skin lesionsc | B0d |
| All lymph nodes ≤ 15 mm in GTD by the method used to assess lymph nodes at baseline; in addition, lymph nodes that were ≤ 15 mm in GTD and > 10 mm in the short axis at baseline now ≤ 10 mm in the short axis |
PR | 50% to 99% clearance of skin disease from baseline without new tumours (T3) in patients with only T1, T2, or T4 skin disease | > 50% decrease in quantitative measurements of blood tumour burden from baseline in those with high tumour burden at baseline (B2)e |
| Cumulative reduction ≥ 50% of the SPD as compared with baseline and no new lymph node (> 15 mm in the GTD or > 10 mm in the short axis if the GTD is > 10 to ≤ 15 mm) |
PD | Meets any of the following:
| Meets any of the following:
| Meets any of the following:
| Meets any of the following:
|
SD | < 25% increase to < 50% clearance in skin disease from baseline without new tumours (T3) in patients with T1, T2, or T4-only skin disease | Fails to attain criteria for CR, PR, or PD | Fails to attain criteria for CR, PR, or PD | Fails to attain the criteria for CR, PR, or PD |
Relapse | Any disease recurrence in those with CR | Increase of neoplastic blood lymphocytes to ≥ B1 in those with CR |
|
|
Unable to evaluate | — | — | An area of visceral disease present at baseline that was not measured or subsequently became unevaluable, leading to an inability to determine the status of that particular area for the time point in question | A lymph node present at baseline that was not measured or subsequently became unevaluable, leading to an inability to determine the status of that particular node for the time point in question |
CR = complete response; GTD = greatest transverse diameter; MF = mycosis fungoides; mSWAT = modified Severity-Weighted Assessment Tool; PD = progression of disease; PR = partial response; SD = stable disease; SPD = sum of the products of the diameters; SS = Sézary syndrome.
aAs assessed by mSWAT scores.
bAs determined by absolute numbers of neoplastic cells/uL.
cA biopsy of normal-appearing skin was unnecessary to assign a CR. However, a skin biopsy of a representative area of the skin was to be performed if there was any question of residual disease where otherwise a CR would exist. If histologic features were suspicious or suggestive of MF or SS (see histologic criteria for early MF), the response was to be considered a PR only.
dIf a bone marrow biopsy was performed at baseline and determined to unequivocally be indicative of lymphomatous involvement then, to confirm a global CR where blood assessment met criteria for B0, a repeat bone marrow biopsy must have shown no residual disease, or the response was to be considered a PR only.
eThere is no PR in those with B1 disease at baseline, as the difference within the range of neoplastic cells that define B1 is not considered significant and should not affect the determination of global objective response.
Source: MAVORIC (0761 to 010) Clinical Study Report.27
Health-Related Quality of Life
The change from baseline through 6 months for the Skindex-29 score, FACT-G total score, and EQ-5D index score was also a key secondary end point of the MAVORIC trial.27 The Skindex-29, FACT-G, and EQ-5D-3L were evaluated at baseline (cycle 1, day 1) and at the end of cycle 1, then every 8 weeks through cycle 25 (i.e., end of cycle 3, 5, and up to cycle 25) and at the end-of-treatment visit.30
A detailed discussion and critical appraisal of the HRQoL outcome measures is provided in Appendix 4:
The Skindex-29 is a validated instrument to measure the effect of skin disease on HRQoL composed of 29 items assessing 3 domains: emotions, symptoms, and functioning on a 5-point Likert scale. Higher scores indicate a higher impact of skin disease. Responses to each item are transformed to a linear scale of 100 (never = 0, rarely = 25, sometimes = 50, often = 75, all the time = 100) for the purpose of calculating a scale score. A scale score is the mean of a patient’s responses to the items in each scale, and the composite Skindex-29 score is calculated as the average of the 3 scale scores to measure the overall impact on QoL. Higher scores indicate a higher impact of skin disease. Missing data for Skindex-29 were handled according to the questionnaire’s scoring guidelines. If responses to more than 25% of items were missing overall, the Skindex-29 score was treated as missing. If any scale had more than 25% of the responses missing, the scale score was missing. The scale scores were the average of the non-missing items in a given scale.27 The recall period of the Skindex-29 was 1 week.30
The FACT-G is a validated instrument for assessing HRQoL in patients with cancer consisting of 27 items in 4 domains: physical well-being, social/family well-being, emotional well-being, and functional well-being. The total FACT-G score is obtained by summing individual subscale scores. Response scores on negatively phrased questions are reversed before summing. Higher scores for the scales and subscales indicate better QoL. Missing data for FACT-G were handled according to the questionnaire’s scoring guidelines. If more than 50% of the items for a subscale had responses, the subscale scores were prorated by multiplying the sum of the subscale by the number of items in the subscale and then dividing by the number of items actually responded to. The total FACT-G score was scored only if the overall item response rate was greater than 80%.27 The recall period of the FACT-G was 7 days.30
The EQ-5D is a standardized, reliable, and validated instrument to measure HRQoL. The EQ-5D self-reported questionnaire includes the EQ-5D descriptive system and VAS. The 3-level version (EQ-5D-3L) descriptive system comprises the following dimensions: mobility, self-care, usual activities, pain/discomfort, and anxiety/depression. The EQ-5D index score is calculated based on the descriptive system using a set of item weights (value sets) to derive a single score ranging from −0.109 to 1, with 1 representing full health. The VAS records the patient’s self-rated health status on a scale of 0 to 100, with higher scores indicating better health. For EQ-5D-3L, the EQ-5D index score was treated as missing if not all 5 descriptors were responded.27 The recall period of the EQ-5D-3L was the current period.30
Other Secondary Efficacy Outcomes
Other secondary efficacy outcomes included PFS and ORR by independent review, BOR, DOR, changes in HRQoL measures at other time points, and change in pruritus evaluation. Secondary outcomes were assessed by both the investigator and independent review. Secondary outcomes in the MAVORIC trial were defined as follows:27
BOR: Defined as the best response recorded across all time points from the start of treatment until disease progression/recurrence or end of treatment.
DOR: Defined as the time from the date that criteria for CR or PR (whichever was recorded first) were met until the first date that PD or death was objectively documented.
Change in pruritus evaluation: Change in pruritus was evaluated by the Pruritus Likert Scale and the ItchyQoL questionnaire. The Pruritus Likert Scale measures the level of itching for pruritus on a scale of 0 to 10, with higher scores indicating greater itch. The ItchyQoL is a validated pruritus-specific QoL instrument consisting of 22 pruritus-specific questions in 3 major domains: symptoms, functioning, and emotions. Higher ItchyQoL scores indicate worse QoL. The ItchyQoL and the Pruritus Likert Scale were evaluated every 4 weeks at each cycle.30 The recall period for the ItchyQoL was not specified.
Exploratory Efficacy Outcomes
Exploratory efficacy end points included OS, defined as the time from the date of randomization until the date of death due to any cause, and TTF, defined as the time from the day of randomization until discontinuation of randomized treatment due to any reason, except for those who discontinued randomized treatment due to 1 year on treatment with a CR. Patients who were still alive at the end of the survival follow-up period or were lost to follow-up were censored on the last date the patient was known to be alive.27
An additional exploratory analysis was performed for comparison of PFS between patients with any exposure to mogamulizumab versus vorinostat. For this analysis, the PFS for patients who cross over from the vorinostat arm to the mogamulizumab arm was calculated from the first dose of mogamulizumab.35
Safety end points included AEs defined as any untoward medical occurrence in a patient in the clinical trial who had been administered a study drug (i.e., investigational product) or a comparator product. The intensity of AEs was graded using the National Cancer Institute’s Common Terminology Criteria for Adverse Events v.4.0. In the randomized treatment period, TEAEs were defined as AEs that occurred from the first dose of the randomized study drug through 90 days after the last dose of randomized study drug or the start of alternative CTCL therapy, whichever occurred first. The study protocol specified that disease progression was not to be considered an AE. Lymphopenia, the pharmacologic effect of the drug, was also not to be considered an AE. However, in cases where the investigators did record disease progression or lymphopenia as AEs, these events were included in the reporting and analysis of AEs.27
For patients who crossed over from vorinostat to mogamulizumab, TEAEs during the randomized treatment period were defined as AEs that occurred after the first dose of vorinostat and before the first dose of mogamulizumab. AEs that occurred after the start of mogamulizumab but were considered related to vorinostat were also counted as TEAEs for the randomized treatment period. The incidence of specific TEAEs in patients who crossed over was not reported.27
Statistical Analysis
Sample Size and Power Calculation
The sample size calculation for the MAVORIC study was based on the primary end point of PFS in the ITT population. Assuming a median PFS for vorinostat of 169 days for reference, a 50% improvement in the median PFS for mogamulizumab was predicted, resulting in a predicted median PFS for mogamulizumab of 254 days. Based on a 24-month accrual and 12-month follow-up, and a 10% rate for dropouts and sample size inflation, the estimated sample size was 317 patients. The sample size of 317 patients provided the 90% power necessary to observe 255 PFS events at a 1-sided 0.025 significance level.27
The final primary analysis comparing PFS between treatment groups was to be conducted when a total of 255 PFS events had occurred or a maximum of 24 months after the last randomized patient’s first dose, whichever came first. In the event the study was stopped before 255 PFS events being observed, the primary test was to be performed at less than 90% power under the current assumptions, according to the protocol.27
The planned enrolment and 255-event threshold for the primary analysis assumed that approximately 20% of the 317 randomized patients would be censored for the primary end point at the time of the primary analysis. Additionally, the 24-month time threshold for the primary analysis assumed that meaningful numbers of PFS events would accrue during the 24-month period after the enrolment of the last patient.27
Interim and Final Analyses
No formal interim analyses were planned or conducted for the MAVORIC study. The final primary analysis was conducted after 255 PFS events or a maximum of 24 months after the last randomized patient’s first dose.27
Analysis Populations
The following analysis populations were defined in the MAVORIC study:27
ITT set: Includes all patients randomized to a therapy (mogamulizumab or vorinostat) and assigned a study number.
Safety analysis set: Includes all patients who received at least 1 dose (even a partial dose) of the assigned study drug (mogamulizumab or vorinostat; does not include patients who crossed over from vorinostat to mogamulizumab).
Efficacy evaluable set: Includes all patients who received the first cycle of treatment (at least 1 dose) and who had a baseline tumour assessment and at least 1 post-baseline assessment for response. If a patient had a baseline tumour assessment but progressed (either died before progression or had documented progression in the follow-up period) during the study without any post-baseline tumour assessment, that patient was still considered as part of the efficacy evaluable set.
Efficacy Analyses
Summary statistics were presented by treatment arm. For continuous variables, the number of available observations (n), mean, standard deviation (SD), median, minimum, and maximum were provided. For categorical variables, the frequency and percentage in each category were displayed. Additionally, the point estimates were accompanied by the corresponding 2-sided 95% CIs.27
Primary Efficacy End Point
The primary efficacy end point of the MAVORIC trial was PFS per investigator assessment, performed on the ITT population using a stratified log-rank test at the 1-sided 2.5% significance level. Stratification was by disease type (MF or SS), disease stage (IB or II versus III or IV), and region (US, Japan, and rest of world). A Cox proportional hazard model with treatment, disease type, disease stage, and region (US, Japan, and rest of world) as covariates was used to assess the magnitude of the treatment difference in PFS. The hazard ratio (HR) along with the 95% CI obtained from the Cox proportional hazard model were presented. The median PFS and the 2-sided 95% CI for each treatment were estimated using the Kaplan-Meier method. The Kaplan-Meier estimate of PFS rates and the corresponding 95% CIs were also provided for each treatment arm by 6-month interval. Plots of the Kaplan-Meier estimate of the survival distribution function for PFS over time were presented by treatment arm.27
An additional analysis of the treatment effect on PFS when adjusted for potential prognostic factors was carried out using a multivariate Cox proportional hazard model. Analyses were performed based on PFS as assessed by the investigator for the first assigned therapy and the ITT set. Potential prognostic factors including disease type (MF or SS), disease stage (IB or II versus III or IV), compartment involvement (blood involvement or no blood involvement), region (US, Japan, rest of world), age group (< 65 or ≥ 65 years), gender (males or females), and race category (Black or African American, White, Other) were included as covariates in the multivariate Cox proportional hazard model for PFS. Backward selection was used to identify the final set of prognostic factors (exit P value was set to 0.1). The final model was then used to assess treatment effect when adjusted for these important prognostic factors.27
Sensitivity Analyses
A total of 4 sensitivity analyses using different definitions of PFS were conducted for the primary end point in the ITT population based on investigator assessment:27
This analysis defined PFS as the time from the day of randomization until documented progression in any compartment, based on the investigator’s assessment per CTCL response criteria or death due to any cause, provided that death was not more than 8 weeks after the last post-baseline tumour assessment or the last dose of the study drug (if the patient did not have any post-baseline tumour assessments). Using this definition, the progression reported during the follow-up period would not be considered an event in the sensitivity analysis but would be a censored value on the day of the last tumour assessment.
This analysis defined PFS as the time from the day of randomization until the earliest date of documented progression in any compartment based on the investigator’s assessment per CTCL response criteria, or clinical progression at the end of the randomized treatment period, or death due to any cause. Using this definition, clinical progression noted at the end of the randomized treatment would be considered an event in the sensitivity analyses.
This analysis defined PFS as the time from the day of randomization until documented progression or death due to any cause. The date of documented disease progression was the earliest date of disease progression in any compartment based on the investigator’s assessment per CTCL response criteria or disease progression reported during the follow-up period. For assessments where progression was reported by the investigator but was not confirmed by the independent review, the progression date was set to the last tumour assessment plus 1 day in the mogamulizumab treatment arm and was censored in the vorinostat treatment arm.
This analysis defined PFS as the time from the day of randomization until documented progression in any compartment per CTCL response criteria or death due to any cause, provided that death occurred:
within 90 days after the last dose of the study drug for patients who had no post-baseline tumour assessments
within 56 days of the last post-baseline tumour assessment if that date was more than 90 days after the last dose of study drug
within 90 days of the last dose of the study drug if the date corresponding to 56 days after the last post-baseline tumour assessment was 90 days or more after the last dose of the study drug.
Subgroup Analyses
Pre-specified subgroup analyses for the primary end point of PFS in the MAVORIC trial were conducted on the ITT population and efficacy evaluable set and included disease type (MF versus SS), disease stage (IB or II versus III or IV), blood involvement (yes versus no), region (US versus Japan versus rest of world), age (< 65 years versus ≥ 65 years), gender (male versus female), race (Black or African American versus White versus other), and lactate dehydrogenase (normal versus elevated). Results for subgroup analyses were assessed by the investigator in the ITT population and presented as forest plots of HRs.27
Key Secondary Efficacy End Points
Objective Response Rate
ORR was a key secondary end point of the MAVORIC study. A confirmed CR or PR was defined as a documented CR or PR based on the investigator’s assessment of overall response per a global composite response score that was subsequently confirmed by 2 or more consecutive observations for a minimum of 4 weeks. Mogamulizumab and vorinostat were compared using a Cochran-Mantel-Haenszel test adjusted for disease type, disease stage, and region). The exact 95% CIs for ORR were calculated for each treatment arm along with the difference in response rates between the 2 treatment arms. The number and percentage of patients achieving objective response (CR or PR) were presented for each treatment arm.27
Additional analysis of the treatment effect on ORR was conducted as previously described for PFS; however, a multivariate logistic regression model was used for ORR. Potential prognostic factors were the same for the PFS and ORR end points.27
Crossover from the vorinostat arm to the mogamulizumab arm was permitted in patients who had received 2 full treatment cycles and demonstrated progression of disease at the 8-week (cycle 2, day 26 to 28) assessment, or anytime thereafter. For patients who crossed over from the vorinostat arm to the mogamulizumab arm, ORR was estimated in a similar fashion as described previously. All summaries for the crossover portion were purely descriptive and no statistical comparisons were made.27
Health-Related Quality of Life
HRQoL, as measured by the Skindex-29, FACT-G, and EQ-5D, were key secondary end points of the MAVORIC study and were evaluated in the ITT and efficacy evaluable populations.27 An additional analysis of the HRQoL data was submitted that evaluated HRQoL as measured by the Skindex-29, FACT-G, and EQ-5D (and ItchyQoL and Pruritis Likert scale) in the cross-sectional analysis population that was derived from the MAVORIC ITT, which was defined at each QoL collection time point as any patient who had survived and maintained in the randomized period of the study into day 1 of each cycle and had available QoL data for that cycle. The longitudinal-period population was defined as all patients with an outcome collection period reported from baseline to each cycle for analysis. Any patient who had survived and maintained in the study from baseline to day 1 of cycle 1 to day 1 of a specified cumulative cycle (e.g., cycle 2), or who had discontinued treatment and had available QoL data for that time point, were included.30
For all HRQoL measures, the mean change from baseline in the Skindex-29 score, FACT-G total score, and EQ-5D-3L index score was evaluated. Additionally, for the Skindex-29, the mean between-group difference was assessed. Assessments of HRQoL were conducted at the end of cycle 1, cycle 3, and cycle 5, up to 6 months, or cycle 11. (Data beyond cycle 11 were summarized descriptively). A restricted maximum likelihood method was used as well as a repeated measures mixed-effects model with time, region, disease type, disease stage, treatment, time-by-treatment interaction as factors, and baseline score was a covariate. An unstructured covariance matrix was employed to model the correlation among repeated measurements. Appropriate contrasts were used to determine the difference between treatment groups across the end of cycles 1, 3, and 5. In the event of convergence problems, another variance-covariance structure was to be considered. The selection of any of these structures was determined after exploration of the observed correlation structure. Least squares mean, corresponding standard errors, and 2-sided 95% CIs were presented for the within-group change. For the between–treatment group comparison, the difference in least squares mean, corresponding standard errors, 2-sided 95% CIs, and 2-sided P value were also derived from this analysis of covariance (ANCOVA) model. This mixed-effects model was also used to compare the treatment differences in the mean changes in Skindex-29 score from baseline to each scheduled time point at the end of cycle 1, cycle 3, and cycle 5. If residuals of the ANCOVA model were not normally distributed, then the analysis was carried out using a Wilcoxon rank sum test instead. In addition to the analysis described previously, summary statistics, including n, mean, SD, median, minimum, and maximum, were provided for the Skindex-29, FACT-G, and EQ-5D scores at baseline and each scheduled time point, and the change in the total score at each of these scheduled assessments for each treatment arm.27
Multiplicity Adjustment
For the 4 key secondary end points (i.e., ORR, and change in the Skindex-29 score, FACT-G total score, and EQ-5D-3L index score from baseline through the 6-month assessment), comparisons between the 2 treatment arms were conducted using P values that were adjusted to control the overall studywise type I error rate to be less than 0.05. Since these tests were not independent, the 4 key secondary end point analyses were conducted using the Šídák adjusted P value method:27
Adjusted P value = 1 − (1 − P),4 where P is the original P value of the individual test.
These adjusted P values were then compared to 0.05 for each test.27
Other Secondary Efficacy End Points
Independent Review Assessment of PFS and ORR
Progression-free survival and ORR were also assessed by independent review using the same methods described previously for PFS and ORR.27
Best Overall Response
BOR was a secondary outcome of the MAVORIC trial. BOR was assessed by the investigator and by the independent reviewer during the randomized treatment period for all patients by treatment arm. BOR was summarized using frequency tables, and concordance between the investigator’s assessment and the independent review assessment of BOR was summarized.27
Duration of Response
DOR was a secondary end point of the MAVORIC trial. No comparisons between mogamulizumab and vorinostat were made, as DOR is conditional on patients achieving a confirmed response. The DOR was analyzed using survival analysis methods. The Kaplan-Meier estimate of the median DOR and the associated 95% CIs were estimated and presented separately for each treatment arm.27
Time to Response
Time to response was a secondary outcome of the MAVORIC trial. For patients who achieved a BOR of CR or PR during the randomized treatment period, the time to response was summarized descriptively. Time to response (TTR) was defined as the time from the date of randomization to the date that criteria for CR/PR (whichever was first recorded) were first met. Subjects who did not respond over the course of the study had a missing value for TTR.27
Changes in Pruritus Evaluation
The Likert scale for pruritus evaluation uses a numbered scale from 0 to 10 to measure the level of itching for pruritus with 10 indicating the worst itch imaginable and 0 indicating no itch. For the Pruritus Likert Scale data, the changes in the Likert scale score from baseline at different time points up to cycle 11 assessments during the randomized treatment period were analyzed using the Wilcoxon rank sum test. The treatment difference in the median change of the Likert scale score from baseline was estimated using the Hodges-Lehmann estimate, and a 2-sided 95% CI for the treatment difference was obtained using the Moses method.27
The ItchyQoL includes 22 pruritus-specific questions covering 3 major domains: symptoms, functioning, and emotions. The subscale scores consist of the average of the responses to the items in a given subscale. The overall score is the average of the responses to all items. Higher ItchyQoL scores indicate worse QoL. Missing data in the ItchyQoL questionnaire were handled according to the following rule: if responses to more than 25% of items were missing overall, the overall score was treated as missing; if any scale had more than 25% of the responses missing, the scale score was missing.27
Exploratory Efficacy End Points
Overall Survival
The OS rates for the mogamulizumab and vorinostat arms were compared as described for PFS based on the ITT population; however, this analysis was considered exploratory. Patients who were still alive at the end of the survival follow-up period or were lost to follow-up at the time of the analysis were censored on the last date the patient was known to be alive.27
Additional exploratory analyses of OS adjusting for the 1-way crossover were performed, which included censoring patients at the time of crossover, as well as the rank-preserving structural failure time (RPSFT) model, and the inverse probability censoring weighting (IPCW) method. The RPSFT model assumes that the experimental treatment is acting by multiplying survival time by a given factor once the patient starts receiving the experimental treatment. The RPSFT model reconstructs the survival time for the control arm as if no crossover were allowed and analyzes data based on reconstructed data. The IPCW method censors the patients who crossed over at the time of crossover, but patients were weighted according to their probability of crossover. The IPCW method artificially increases weights for patients with a low probability of crossover and decreases weights for patients with a high probability of crossover.35
Time-to-Treatment Failure
TTF for the mogamulizumab and vorinostat arms was compared as described for PFS based on the ITT population; however, it was considered exploratory. Patients who experienced a CR and discontinued randomized treatment after 1 year of treatment were censored at the last dose date of the randomized treatment. Patients who were randomized but did not take any study drug were censored at the last documented visit date.27
Safety Analyses
All safety summaries were based on the safety analysis set. TEAEs during the randomized treatment period were summarized by the number and percent of patients in each treatment arm and in total by SOC and preferred term for the safety population, and subgroups of gender (male versus female) and age (< 65 years versus ≥ 65 years).27
If a patient who had received mogamulizumab underwent a stem-cell transplant at any time during or after the 90-day follow-up period, AEs and SAEs were to be documented from the transplant procedure through 180 days post transplant.27
The safety data for patients in the vorinostat arm who crossed over to mogamulizumab were summarized separately in the same manner as those in the randomized treatment groups and presented separately. No safety results for patients who crossed over were provided.27
Protocol Amendments and Deviations
The sponsor-submitted Clinical Study Report included a total of 8 amendments that were made to the original protocol (issued June 19, 2012) of the MAVORIC study, which are summarized in Table 13. The submitted protocol corresponded to amendment 10, occurring on May 31, 2018. The first protocol amendment was conducted in July 2012, following requests from the FDA. The order of secondary objectives was modified at amendment 2 to reflect the importance of QoL data and to specify the exploratory objectives of OS and mogamulizumab exposure–response relationships.27 Most of the major protocol changes affected the inclusion and exclusion criteria of the patients eligible for the study. Amendment 5, which occurred 2 years after the original protocol, clarified that patients with either relapsed or refractory disease were eligible to participate, potentially impacting the type of patients included.27 Other protocol amendments were generally related to changes in or clarification of study procedures.
The last patient was randomized on January 29, 2016; however, it is unclear how many patients were enrolled before and after each amendment; therefore, it is difficult to determine any potential impacts the protocol changes may have had on the overall population. However, the changes to the protocol were not believed to impact the conduct or integrity of the study, nor did they affect the patients’ safety.
Table 13: Summary of Amendments and Key Changes to the MAVORIC Protocol
Amendment | Key changes to protocol |
|---|---|
Amendment 1 July 9, 2012 |
|
Amendment 2 February 19, 2013 |
|
Amendment 3 April 3, 2013 |
|
Amendment 4 November 14, 2013 |
|
Amendment 5 March 5, 2014 |
|
Amendment 6 March 6, 2015 |
|
Amendment 7 March 9, 2016 |
|
Amendment 8 January 20, 2017 |
|
Amendment 9 April 20, 2018 |
|
Amendment 10 May 31, 2018 |
|
AE = adverse event; ANCOVA = analysis of covariance; CR = complete response; CT = CT; CTCL = cutaneous T-cell lymphoma; EORTC = European Organisation for Research and Treatment of Cancer; EQ-5D-3L = 3-Level EQ-5D; FACT-G = Functional Assessment of Cancer Therapy–General; HSCT = hematopoietic stem-cell transplant; IRR = infusion-related reaction; ISCL = International Society for Cutaneous Lymphomas; KW-0761 = mogamulizumab; LCT = large-cell transformation; MF = mycosis fungoides; mSWAT = modified Severity-Weighted Assessment Tool; OS = overall survival; PD = progressive disease; PUVA = psoralen plus UVA light therapy; QoL = quality of life; SAE = serious adverse event; SJS = Stevens-Johnson syndrome; SS = Sézary syndrome; TEN = toxic epidermal necrolysis; VAS = Visual Analogue Scale.
Source: MAVORIC (0761 to 010) Clinical Study Report27 and sponsor submission.35
There was a total of 99 patients (53.2%) in the mogamulizumab group and 111 patients (59.7%) in the vorinostat group with major protocol deviations in the MAVORIC trial (Table 14).27
Table 14: Summary of Major Protocol Deviations (Intention-to-Treat Population)
Detail | Mogamulizumab (N = 186) | Vorinostat (N = 186) |
|---|---|---|
Patients with any major protocol deviations, n (%) | 99 (53.2) | 111 (59.7) |
Inclusion or exclusion criteria | 47 (25.3) | 54 (29.0) |
Investigational product | 8 (4.3) | 7 (3.8) |
Prohibited therapies and/or medications | 43 (23.1) | 37 (19.9) |
Study procedures | 36 (19.4) | 41 (22.0) |
Source: MAVORIC (0761 to 010) Clinical Study Report.27
Changes to Planned Analysis
The original statistical analysis plan was signed on February 24, 2014, and revised on November 22, 2016. Key changes to analyses that were not specified in the statistical analysis plan before database lock include the following:27
clarified that for PFS, documented progression includes disease progression in any compartment based on investigator’s assessment per CTCL response criteria, or documented disease progression reported during the follow-up period
clarified that confirmation of a CR and PR was required for the ORR and that confirmed and unconfirmed ORRs would be summarized; added summaries by disease type
added TTF as an exploratory efficacy variable
removed statement that each patient would be followed in the study for a minimum of 2 years to generate information on OS
added a sensitivity analysis for PFS that included clinical progression as a PFS event
added exploratory analysis of PFS for patients with any exposure to mogamulizumab versus patients who received vorinostat only
removed the CCR4-related analysis (a separate analysis plan was prepared to detail the CCR4 analysis)
added testing for QoL assessments beyond the 6-month assessment period
added exploratory analyses of OS adjusting for crossover; these analyses included the RPSFT model and the IPCW model.
Minor changes to the planned analyses defined in the final revised statistical analysis plan were provided in a supplement dated March 21, 2017 (before database lock):27
added a fourth sensitivity analysis of PFS to consider death during safety follow-up
clarified that exploratory analyses of OS accounting for crossover effect (RPSFT and IPCW analysis models) would not be presented in the Clinical Study Report but would be included elsewhere in the Common Technical Document (module 2.7.3)
specified that AEs that occurred more than 90 days after the last dose of the study drug but were related to the study drug would be considered as TEAEs.
Subsequent to database lock and availability of topline results, the following additional analyses were performed:27
summary of predefined protocol deviations
analysis and summary of dose intensity for vorinostat
analyses of response by compartment (blood, lymph nodes, skin, viscera):
confirmed response rate based on investigator’s assessment and independent review
confirmed plus unconfirmed response rate based on investigator’s assessment and independent review
DOR based on investigator’s assessment and independent review
BOR based on investigator’s assessment and independent review
TTR based on investigator’s assessment and independent review
confirmed ORR by number of prior systemic therapies received based on investigator’s assessment and independent review
summary of PFS based on investigator’s assessment, excluding patients who had specific protocol deviations.
Results
Patient Disposition
Table 15 summarizes the disposition of patients enrolled in the MAVORIC trial. The MAVORIC trial was a randomized, open-label, phase III clinical trial. A total of 464 patients were screened for eligibility, and 372 were randomized to receive mogamulizumab (n = 186), or vorinostat (n = 186), making up the ITT set, for which analyses did not include patients who crossed over from vorinostat to mogamulizumab. Two patients randomized to the mogamulizumab arm did not receive the study treatment (n = 184), comprising the safety analysis set, which also did not include patients who had crossed over. As of the data cut-off date (December 31, 2016), 27 (14.5%) and 10 (5.4%) randomized patients remained on mogamulizumab and vorinostat, respectively. The most common reason for discontinuation of randomized treatment in the mogamulizumab group was PD per CTCL response criteria in any category of skin, nodes, blood, and viscera (n = 76 out of 157; 48.4%).27
At the time of data cut-off, 109 patients (58.6%) randomized to mogamulizumab were continuing the study (treatment or follow-up), and 77 (41.4%) had discontinued the study, primarily due to death (n = 40; 21.5%) or withdrawal of consent (n = 30; 16.1%). Of the 186 patients randomized to vorinostat, 70 (37.6%) had discontinued from the study at the time of data cut-off, including 41 who had crossed over to mogamulizumab treatment, primarily due to death (n = 47; 25.3%) or withdrawal of consent (n = 19; 10.2%).27
A total of 136 patients (73.1%) randomized to the vorinostat group crossed over to receive treatment with mogamulizumab, while 40 patients randomized to vorinostat did not cross over but discontinued vorinostat, and 10 patients remained on randomized treatment. The most common reason for crossover was due to disease progression (n = 109; 80.1%), followed by drug intolerance (n = 27; 19.9%). At the time of data cut-off, 105 patients (77.2%) who crossed over from vorinostat had discontinued study treatment. The primary reasons for discontinuation following crossover were disease progression per CTCL response criteria or clinical disease progression (n = 61; 44.9%) and AEs (n = 22; 16.2%).27 Of the 61 patients who crossed over to mogamulizumab and discontinued due to progression, 46 experienced at least stable disease before progressing on mogamulizumab, 13 continued to progress, and 2 patients approved for crossover did not receive mogamulizumab treatment before progression.44 Thirty-one patients (22.7%) who crossed over from vorinostat were continuing to receive mogamulizumab at data cut-off.27
The last patient exited the MAVORIC trial on February 17, 2021.44
Table 15: Patient Disposition in the MAVORIC Trial
Disposition | Mogamulizumab | Vorinostat | |
|---|---|---|---|
Screened, N | 464 | ||
Randomized treatment phase (not including crossover) | |||
Randomized, N | 186 | 186 | |
Discontinued randomized treatment, N (%) | 157 (84.4) | 40 (21.5)a | |
Progressive disease, CTCL criteria | 76 (48.4) | 10 (25.0) | |
Progressive disease, clinicalb | 22 (14.0) | 8 (20.0) | |
Withdrew consent | 13 (8.3) | 5 (12.5) | |
Protocol non-compliance | 1 (0.6) | 0 | |
Investigator decision | 9 (5.7) | 0 | |
Adverse event | 28 (17.8) | 5 (12.5) | |
Death | 2 (1.3) | 2 (5.0) | |
Other | 6 (3.8) | 10 (25.0) | |
Randomized treatment ongoing at data cut-off, N (%) | 27 (14.5) | 10 (5.4) | |
Study follow-up (ITT) | |||
Patients randomized, N | 186 | 186 | |
Patients discontinued from the study, N (%) | 77 (41.4) | 70 (37.6) | |
Withdrawal of consent | 30 (16.1) | 19 (10.2) | |
Death | 40 (21.5) | 47 (25.3) | |
Lost to follow-up | 6 (3.2) | 4 (2.2) | |
Other | 1 (0.5) | 0 | |
Patients ongoing (treatment or follow-up), N | 109 | 116c | |
Patients who crossed over to mogamulizumab | |||
Randomized to vorinostat and crossed over to mogamulizumab, N (%) | NA | 136 (73.1) | |
Reason for crossover: Progressive diseased | NA | 109 (80.1) | |
Reason for crossover: Intolerance | NA | 27 (19.9) | |
Patients crossed over to mogamulizumab and subsequently discontinued mogamulizumab | 105 (77.2)e | NA | |
Progressive disease per CTCL response criteria | 42 (30.9) | NA | |
Progressive disease, clinical | 19 (14.0) | NA | |
Withdrawal of consent | 3 (2.2) | NA | |
Patient requires prohibited concomitant medication | 1 (0.7) | NA | |
Investigator decision | 7 (5.1) | NA | |
Adverse event | 22 (16.2) | NA | |
Death | 3 (2.2) | NA | |
Other | 8 (5.9) | NA | |
Patients ongoing with mogamulizumab treatment at data cut-off | 31 (22.7) | NA | |
Patients crossed over to mogamulizumab and discontinued from the study | 41 (30.1) | NA | |
Withdrawal of consent | 10 (7.4) | NA | |
Death | 28 (20.6) | NA | |
Lost to follow-up | 3 (2.2) | NA | |
Analysis sets | |||
ITT set, N | 186 | 186 | |
Efficacy evaluable set, N | 180 | 181 | |
Safety analysis set, N | 184 | 186 | |
CTCL = cutaneous T-cell lymphoma; ITT = intention-to-treat population; NA = not applicable; PD = progressive disease.
aPatients in the vorinostat arm who discontinued randomized treatment and did not cross over.
bPatients with disease progression who did not meet criteria for PD based on CTCL response criteria.
cIncludes patients who crossed over to mogamulizumab treatment.
dIncludes 6 patients with clinical progression.
eIncludes 3 patients who did not receive mogamulizumab after crossover.
Source: MAVORIC (0761 to 010) Clinical Study Report.27
Exposure to Study Treatments
Exposure to study treatments is summarized in Table 16. The median follow-up of the randomized phase of the MAVORIC trial was 17.0 months for efficacy and 34.5 months for safety.43 The mean and median duration of exposure to study treatments was higher in the randomized mogamulizumab arm and in the patients who crossed over to mogamulizumab compared with vorinostat. The median duration of treatment with mogamulizumab was 170 days compared with 84 days with vorinostat in the randomized treatment period. The median dose intensity was 97.49% and 95.12% for the mogamulizumab and vorinostat groups, respectively.
Results were similar in patients who crossed over to mogamulizumab from vorinostat with a similar mean number of cycles of mogamulizumab administered between randomized and crossover patients; the median duration of treatment was 169 days and the median dose intensity was 94.92%. Additionally, the proportion of patients initiating each cycle was similar between the patients randomized to mogamulizumab and those who crossed over to mogamulizumab.27
Table 16: Extent of Exposure During Randomized Treatment Period (Safety Analysis Set) and Crossover
Exposure | Randomized treatment | Crossover | |
|---|---|---|---|
Mogamulizumab (N = 184) | Vorinostat (N = 186) | Mogamulizumab (N = 136)a | |
Extent of exposure (days)b | |||
Mean (SD) | 245.2 (234.48) | 144.3 (172.48) | 255.6 (276.24) |
Median (range) | 170.0 (1 to 1,379) | 84.0 (4 to 1,058) | 169.0 (1 to 1,328) |
Total number of cycles initiatedb | |||
Mean (SD) | 9.1 (7.95) | 5.4 (6.05) | 9.5 (9.38) |
Median (range) | 6.0 (1 to 45) | 3.0 (1 to 36) | 7.0 (1 to 46) |
Patients, n (%), who initiated treatment for at least:b,c | |||
1 cycle | 184 (100.0) | 186 (100.0) | 133 (97.8) |
2 cycles | 170 (92.4) | 162 (87.1) | 114 (83.8) |
3 cycles | 150 (81.5) | 110 (59.1) | 103 (75.7) |
4 cycles | 129 (70.1) | 83 (44.6) | 88 (64.7) |
5 cycles | 113 (61.4) | 63 (33.9) | 83 (61.0) |
6 cycles | 103 (56.0) | 53 (28.5) | 76 (55.9) |
7 cycles | 91 (49.5) | 43 (23.1) | 69 (50.7) |
8 cycles | 84 (45.7) | 36 (19.4) | 59 (43.4) |
9 cycles | 77 (41.8) | 32 (17.2) | 52 (38.2) |
10 cycles | 73 (39.7) | 26 (14.0) | 49 (36.0) |
11 cycles | 62 (33.7) | 26 (14.0) | 42 (30.9) |
12 cycles | 50 (27.2) | 21 (11.3) | 35 (25.7) |
13 cycles | 46 (25.0) | 20 (10.8) | 31 (22.8) |
Number of mogamulizumab infusions administered | |||
Mean (SD) | 19.1 (15.64) | — | 19.8 (18.45) |
Median (range) | 14.0 (1 to 90) | — | 15.0 (1 to 94) |
Dose intensity, %d | |||
Mean (SD) | 94.41 (8.037) | 88.96 (14.44) | 94.07 (11.574) |
Median (range) | 97.49 (54.1 to 104.5) | 95.12 (35.4 to 100) | 97.92 (11.2 to 103.7) |
SD = standard deviation.
aExposure results are based on 133 patients who crossed over to mogamulizumab and were treated.
bTen patients randomized to vorinostat were ongoing at data cut-off and had missed the last dose date for vorinostat during the randomized treatment period. The last dose date has been imputed using the patient’s last visit date during the randomized treatment period.
cA patient is considered to have initiated treatment for a cycle if they received any assigned study drug for that cycle.
dPercentage dose intensity of mogamulizumab was calculated as 100 × (total actual dose ÷ total duration of treatment ÷ 7) ÷ (total planned dose ÷ total planned weeks). Percentage dose intensity of vorinostat was calculated as 100 × (sum of patient’s actual dosage per dosing interval × actual days exposed per dosing interval) ÷ (400 × expected dose days), where expected dose days is the last dose date minus the first dose date plus 1.
Source: MAVORIC (0761 to 010) Clinical Study Report.27
Dose Interruptions and Modifications
During the randomized treatment period, 65 patients (35.3%) in the mogamulizumab group had at least 1 dose withheld, while 70 (38.0%) did not receive the total planned dose of mogamulizumab. Mogamulizumab administration was temporarily interrupted in 17 patients (9.2%), primarily due to infusion reaction. Conversely, 101 patients (54.3%) in the vorinostat group had at least 1 dose modification during the randomized treatment period, and 34 patients (18.3%) missed at least 1 dose.27
In the crossover portion of the trial, 38 patients (27.9%) who crossed over to receive mogamulizumab had a dose withheld, 43 (31.6%) did not have their total planned dose administered, and 17 (12.5%) had their infusion temporarily interrupted.27
Efficacy
Only those efficacy outcomes and analyses of subgroups identified in the review protocol are reported subsequently. See Appendix 3 for detailed efficacy data.
Overall Response Rate
The ORR in the ITT population as assessed by the investigator was a key secondary efficacy end point of the MAVORIC trial. Results for ORR are summarized in Table 17. A total of 28% (95% CI, 21.6 to 35.0) of patients in the mogamulizumab group and 4.8% (95% CI, 2.2 to 9.0) of patients in the vorinostat group achieved a confirmed ORR. The risk difference between groups was 23.1%; P < 0.0001). Including both confirmed and unconfirmed responses, ORR based on investigator assessment was 34.9% for mogamulizumab compared with 6.5% for vorinostat (P < 0.0001).27
Table 17: Summary of Confirmed ORR Based on Investigator’s Assessment During Randomized Treatment Period (ITT)a
Outcome | Mogamulizumab (N = 186) | Vorinostat (N = 186) |
|---|---|---|
ORR (confirmed CR + PR), n (%) | 52 (28.0) | 9 (4.8) |
95% CIa | (21.6 to 35.0) | (2.2 to 9.0) |
Risk difference, mogamulizumab vs. vorinostat (95% CI)a | 23.1 (12.8 to 33.1) | |
P valueb | < 0.0001 | |
Adjusted P valueb | < 0.0001 | |
CI = confidence interval; CR = complete response; ITT = intention-to-treat population; ORR = overall response rate; PR = partial response.
aDoes not include crossover.
bThe 95% CI for the response rate is the exact 95% CI. The 95% CI for the difference is the exact 95% unconditional CI for the risk difference (mogamulizumab minus vorinostat).
cP value was obtained from Cochran-Mantel-Haenszel test, adjusting for disease type, disease stage, and region. The adjusted P value was calculated using the Šídák method.
Source: MAVORIC (0761 to 010) Clinical Study Report.27
ORR by Independent Review
The ORR assessed by independent review was a secondary end point of the MAVORIC study. Results for confirmed ORR based on independent review were consistent with ORR based on investigator assessment. The confirmed ORR by independent review was 23.1% for mogamulizumab compared with 3.8% for vorinostat (P < 0.0001). The risk difference between mogamulizumab and vorinostat as assessed by independent review was 19.4% (95% CI, 9.0 to 29.4).27
Additional (Subgroup) Analysis of ORR
Multiple unplanned subgroup and post hoc analyses of ORR were identified for the MAVORIC trial, including analyses by disease type (MF versus SS),27 disease stage,28,45 disease compartment,27,45 blood involvement classification at baseline,29 number of prior systemic therapies,27 and type of prior systemic therapy.46
Though not an official pre-specified subgroup analysis in MAVORIC, ORR was estimated by disease type for patients with MF or SS at baseline. The results by disease type were consistent with the primary ORR analysis, where the proportion of MF patients with ORR was 21.0% in the mogamulizumab group compared with 7.1% in the vorinostat group, and 37.0% versus 2.3% in the mogamulizumab and vorinostat groups for patients with SS.27
A post hoc analysis conducted on patients from the MAVORIC trial with less advanced (stage IB and IIA) MF (n = 36 for mogamulizumab; n = 49 for vorinostat) demonstrated consistent ORR results with the primary analysis, with an ORR of 19.5% for mogamulizumab (20% in patients with stage IB and 19% in patients with IIA), compared with 10.2% for vorinostat (18.5% in IB and 0% in IIA). For patients with stage IB and IIA disease, compartment response rates for blood, skin, and lymph node compartments were 75%, 38.1%, and 15.0% for mogamulizumab, and 12.5%, 22.7%, and 0% for vorinostat, respectively.45 Conversely, in another post hoc analysis of patients with advanced (stage IIB to IVB) MF or SS (mogamulizumab, n = 150; vorinostat, n = 137), the investigator-assessed ORR was 30.0% for mogamulizumab and 2.9% for vorinostat.28
A post hoc analysis assessing response rate by disease compartment was performed to assist in the interpretation of the key secondary end point of ORR (Table 18). Patients were included in the analysis for a specific compartment if disease was present in the compartment at baseline or the patient had a post-baseline assessment indicating the presence or progression of disease in a compartment not involved at baseline. The investigator-assessed ORR in skin was 41.9% for mogamulizumab and 15.6% for vorinostat. The response rate in blood was also higher for mogamulizumab, with an investigator-assessed ORR of 66.9% for mogamulizumab and 18.4% for vorinostat. The ORR in lymph nodes was 15.4% for mogamulizumab compared with 3.8% for vorinostat. There were no confirmed responses achieved in visceral involvement in either arm as assessed by the investigator. Similar results were observed for ORR by compartment based on independent review (Table 40), though the proportion of patients with ORR in blood and lymph nodes was more than 5% higher, as assessed by the investigator, compared with independent review.27
Table 18: Summary of Confirmed Response Rate by Disease Compartment (ITT) by Investigator Assessment
Compartment | Mogamulizumab (N = 186) | Vorinostat (N = 186) |
|---|---|---|
Blood, n | 124 | 125 |
ORR (confirmed CR + PR), n (%) | 83 (66.9) | 23 (18.4) |
95% CIa | (57.9 to 75.1) | (12.0 to 26.3) |
Risk difference, mogamulizumab vs. vorinostat (95% CI)a | 48.5 (36.8 to 58.8) | |
P valueb | < 0.0001 | |
Skin, n | 186 | 186 |
ORR (confirmed CR + PR), n (%) | 78 (41.9) | 29 (15.6) |
95% CIa | (34.8 to 49.4) | (10.7 to 21.6) |
Risk Difference, mogamulizumab vs. vorinostat (95% CI)a | 26.3 (16.1 to 36.2) | |
P valueb | < 0.0001 | |
Lymph nodes, n | 136 | 133 |
ORR (confirmed CR + PR), n (%) | 21 (15.4) | 5 (3.8) |
95% CIa | (9.8 to 22.6) | (1.2 to 8.6) |
Risk difference, mogamulizumab vs. vorinostat (95% CI)a | 11.7 (−0.2 to 23.6) | |
P valueb | 0.0008 | |
Viscera, n | 6 | 4 |
ORR (confirmed CR + PR), n (%) | 0 | 0 |
95% CIa | (0.0 to 45.9) | (0.0 to 60.2) |
Risk difference, mogamulizumab vs. vorinostat (95% CI)a | — | |
P valueb | — | |
CI = confidence interval; CR = complete response; ITT = intention-to-treat population; ORR = overall response rate; PR = partial response; vs. = versus.
aThe 95% CIs for response rate are the exact 95% CIs. The 95% CI for difference is the exact 95% unconditional CI for the risk difference (mogamulizumab minus vorinostat).
bP value was obtained from a Cochran-Mantel-Haenszel test adjusting for disease type, disease stage, and region. The adjusted P value (for ORR) was calculated using the Šídák method.
Source: MAVORIC (0761 to 010) Clinical Study Report.27
The results for ORR by blood classification in a post hoc analysis are summarized in Table 45 of Appendix 3. When stratified by baseline blood classification, the ORR appeared higher with mogamulizumab than with vorinostat in all B0 (15.6% versus 6.5%), B1 (25.8% versus 6.5%), and B2 (37.4% versus 3.2%) assessments.29
An additional analysis subsequent to the main ORR results was the assessment of ORR by number of prior systemic therapies. The response rate for mogamulizumab was greater than that for vorinostat, regardless of the number of prior systemic regimens received, as assessed by the investigator (Table 19) and independent review (Table 41).27
Table 19: Confirmed ORR by Number of Prior Systemic Therapies (ITT) per Investigator Assessment
Number of prior systemic therapies | Mogamulizumab | Vorinostat | ||
|---|---|---|---|---|
n | ORR (95% CI)a | n | ORR (95% CI)a | |
0 | 0 | 0 | 1 | 100 (2.5 to 100.0) |
1 | 28 | 25.0 (10.7 to 44.9) | 40 | 5.0 (0.6 to 16.9) |
2 | 40 | 17.5 (7.3 to 32.8) | 38 | 5.3 (0.6 to 17.7) |
3 | 40 | 35.0 (20.6 to 51.7) | 37 | 5.4 (0.7 to 18.2) |
4 | 22 | 40.9 (20.7 to 63.6) | 18 | 0 (0.0 to 18.5) |
5 | 12 | 16.7 (2.1 to 48.4) | 21 | 4.8 (0.1 to 23.8) |
≥ 6 | 44 | 29.5 (16.8 to 45.2) | 31 | 3.2 (0.1 to 16.7) |
CI = confidence interval; ITT = intention-to-treat population; ORR = overall response rate.
aThe 95% CI for response rate is the exact 95% CI.
Source: MAVORIC (0761 to 010) Clinical Study Report.27
ORR by prior type of therapy received in patients treated with mogamulizumab is summarized in Table 47 and Figure 11 of Appendix 3. The ORR was relatively consistent, with the primary analysis ranging from 20.4% for immunoinhibitory treatments to 38.5% for HDAC inhibitors, and it was also consistent by specific systemic therapy, ranging from 19.6% for oral bexarotene to 37.5% for romidepsin.46
Best Overall Response
BOR was a secondary efficacy end point of the MAVORIC study. It was defined as the best response recorded across all time points from the start of treatment until disease progression or recurrence or end of treatment. Results for BOR as assessed by the investigator and independent review were consistent and are summarized in Table 20.27
Overall, there were more responders in the mogamulizumab group compared with the vorinostat group, as assessed by both the investigator (34.9% versus 6.5%) and independent review (29.0% versus 7.0%). As reported by the investigator, there were 4 confirmed CRs in the mogamulizumab group and none in the vorinostat group. The rate of confirmed PR as assessed by the investigator was higher in the mogamulizumab group compared with the vorinostat group (25.3% versus 4.8%); however, the proportion of patients with stable disease was higher in the vorinostat group compared with mogamulizumab (61.8% versus 43.0%). Results by independent review were consistent with the investigator’s assessment.27
Table 20: Summary of BOR During Randomized Treatment Period (ITT) by Investigator Assessment
Category | Mogamulizumab (N = 186) | Vorinostat (N = 186) |
|---|---|---|
Responders (CR + PR) | 65 (34.9) | 12 (6.5) |
CR | 5 (2.7)a | 0 |
Confirmed CR | 4 (2.2) | 0 |
PR | 60 (32.3) | 12 (6.5) |
Confirmed PR | 47 (25.3) | 9 (4.8) |
Stable disease | 80 (43.0) | 115 (61.8) |
Progressive disease | 1 (0.5) | 6 (3.2) |
Not assessableb | 40 (21.5) | 53 (28.5) |
BOR = best overall response; CR = complete response; ITT = intention-to-treat population; PR = partial response.
aOne patient in the mogamulizumab group had a confirmed PR based on investigator’s assessment, but the BOR was an unconfirmed CR at time of data cut. Since this patient had a CR with relapse in the skin only, they should have been counted as confirmed CR, since a relapse in the skin does not constitute progression if it does not meet the specified criteria for progression.
bIf there was no post-baseline tumour assessment or the response for all post-baseline tumour assessments was not assessable or could not be evaluated, the BOR was classified as not assessable.
Source: MAVORIC (0761 to 010) Clinical Study Report.27
Of the 136 patients who were randomized to vorinostat and crossed over to mogamulizumab, responses to mogamulizumab were observed in 41 patients (30.1%) during the crossover portion of the study. There were 7 (5.1%) confirmed CRs and 34 (25.0%) confirmed PRs that occurred during crossover.27
Duration of Response
DOR for patients with a confirmed CR or PR is summarized in Table 21. The median DOR as assessed by the investigator for confirmed responders was 14.07 months (95% CI, 9.43 to 19.17) for mogamulizumab and 9.13 months (95% CI, 4.67 to NE) for vorinostat. Results for DOR were consistent when assessed by independent review (Table 43); however, the median DOR could not be assessed for vorinostat, as 6 of the 7 observations were censored.27
Table 21: Summary of Investigator-Assessed DOR During Randomized Treatment Period (ITT)
Category | Mogamulizumab (N = 186) | Vorinostat (N = 186) |
|---|---|---|
Number of patients with confirmed CR or PR | 52 | 9 |
Patients who had PD or died, n (%) | 26 (50.0) | 4 (44.4) |
Patients censored, n (%) | 26 (50.0) | 5 (55.6) |
Median (95% CI) DOR (months)a | 14.07 (9.43 to 19.17) | 9.13 (4.67 to NE) |
CI = confidence interval; CR = complete response; DOR = duration of response; ITT = intention-to-treat population; NE = not estimable; PD = progressive disease; PR = partial response.
aKaplan-Meier estimate.
Source: MAVORIC (0761 to 010) Clinical Study Report.27
DOR by compartment was assessed as a post hoc analysis. The median duration of confirmed responses by investigator assessment in skin was 20.57 months (95% CI, 17.77 to NE) for mogamulizumab and 10.67 months (95% CI, 4.77 to NE) for vorinostat. The median duration of confirmed responses in blood and nodal responses for mogamulizumab was 25.47 months (95% CI, 16.10 to NE), and 15.47 months (95% CI, NE to NE). The median duration of confirmed responses for vorinostat in these compartments was not evaluable. The duration of confirmed responses was not evaluated in the visceral compartment, as no responses were observed.27
Additional (Subgroup) Analysis of DOR
In a post hoc analysis, the DOR was measured by number and type of prior treatment for patients in the MAVORIC trial. The median DOR by therapeutic class is summarized in Table 47, while the median DOR by specific prior therapy is summarized Figure 11 of Appendix 3. Median DOR in patients treated with mogamulizumab in the MAVORIC trial was consistent with the primary analysis, regardless of the number of prior therapies received, ranging from 9.4 months with 6 or more prior therapies to 19.9 months with 4 prior therapies (Table 46). Additionally, the DOR was consistent regardless of therapeutic class (Table 47), or specific therapy received (Figure 11).46
Progression-Free Survival
PFS per investigator assessment was the primary end point of the MAVORIC trial. Results for PFS are summarized in Table 22 and by Kaplan-Meier plot in Figure 2. At the time of the data cut-off (December 31, 2016), a total of 241 PFS events had occurred; 110 (59.1%) in the mogamulizumab group and 131 (70.4%) in the vorinostat group.27 The HR for mogamulizumab versus vorinostat was 0.53 (95% CI, 0.41 to 0.69), in favour of mogamulizumab. The median PFS was 7.70 months (95% CI, 5.67 to 10.33) in the mogamulizumab group, and 3.10 months (95% CI, 2.87 to 4.07) in the vorinostat group (P < 0.0001). Patients without progression who were continuing to receive randomized treatment were censored as of the cut-off date. The PFS rate at 6, 12, 18, and 24 months was 55.3% (95% CI, 47.1 to 62.6), 38.3% (95% CI, 30.2 to 46.4), 28.0% (95% CI, 19.8 to 36.8), and 14.1% (95% CI, 6.4 to 24.8) in the mogamulizumab group, and 28.8% (95% CI, 21.6 to 36.3), 15.3% (95% CI, 9.5 to 22.3), 7.2% (95% CI, 2.7 to 14.5), and 7.2% (95% CI, 2.7 to 14.5) for the vorinostat groups, respectively.27
A total of 133 patients crossed over from vorinostat to receive treatment with mogamulizumab due to either disease progression or intolerance. In the pre-specified additional exploratory analysis of PFS for these patients (i.e., PFS2), the median PFS2 calculated from the first dose of mogamulizumab was 8.87 months (95% CI, 5.37 to 14.77). Across all patients who were either randomized to mogamulizumab or received mogamulizumab during crossover (n = 319), the median PFS was 8.43 months (95% CI, 6.10 to 10.30).27
Table 22: Summary of PFS by Investigator’s Assessment (ITT)
Category | Mogamulizumab (N = 186) | Vorinostat (N = 186) |
|---|---|---|
PFS event, n (%) | 110 (59.1) | 131 (70.4) |
Progressive disease | 104 (55.9) | 128 (68.8) |
Death | 6 (3.2) | 3 (1.6) |
Censored, n (%) | 76 (40.9) | 55 (29.6) |
Clinical PDa | 16 (21.1) | 10 (18.2) |
Ongoing randomized treatment | 16 (21.1) | 7 (12.7) |
AE or intolerance | 14 (18.4) | 25 (45.5) |
Withdrew consent | 9 (11.8) | 3 (5.5) |
Investigator decision | 7 (9.2) | 0 |
Other | 5 (6.6) | 7 (12.7) |
Withdrawal before first post-baseline assessment | 3 (3.9) | 2 (3.6) |
New anti-cancer therapy | 3 (3.9) | 0 |
No baseline assessment and no post-baseline assessmentb | 2 (2.6) | 0 |
Protocol non-compliance | 1 (1.3) | 0 |
Lost to follow-up | 0 | 1 (1.8) |
Median PFS, months (95% CI)c | 7.70 (5.67 to 10.33) | 3.10 (2.87 to 4.07) |
HR (95% CI)d | 0.53 (0.41 to 0.69) | |
Log-rank P valued | < 0.0001 | |
PFS rate, % (95% CI)e | ||
6 months | 55.3 (47.1 to 62.6) | 28.8 (21.6 to 36.3) |
12 months | 38.3 (30.2 to 46.4) | 15.3 (9.5 to 22.3) |
18 months | 28.0 (19.8 to 36.8) | 7.2 (2.7 to 14.5) |
24 months | 14.1 (6.4 to 24.8) | 7.2 (2.7 to 14.5) |
30 months | 4.7 (0.5 to 17.7) | 7.2 (2.7 to 14.5) |
AE = adverse event; CI = confidence interval; CTCL = cutaneous T-cell lymphoma; HR = hazard ratio; ITT = intention-to-treat population; PD = progressive disease; PFS = progression-free survival.
aPatients with disease progression who did not meet the criteria for PD based on CTCL response criteria.
bPatients randomized but not treated.
c95% CIs obtained from SAS PROC LIFETEST using log-log transformation.
dThe HR and 95% CI are based on a Cox proportional hazards model with treatment, disease type, disease stage, and region as covariates. P value (2-sided) was obtained from a stratified log-rank test (1-sided test at 0.025 level or equivalent 2-sided test at 0.05 level) with disease type, disease stage, and region as stratification factors.
eKaplan–Meier estimate.
Source: MAVORIC (0761 to 010) Clinical Study Report.27
Figure 2: Kaplan-Meier Curve of PFS by Investigator’s Assessment (ITT)
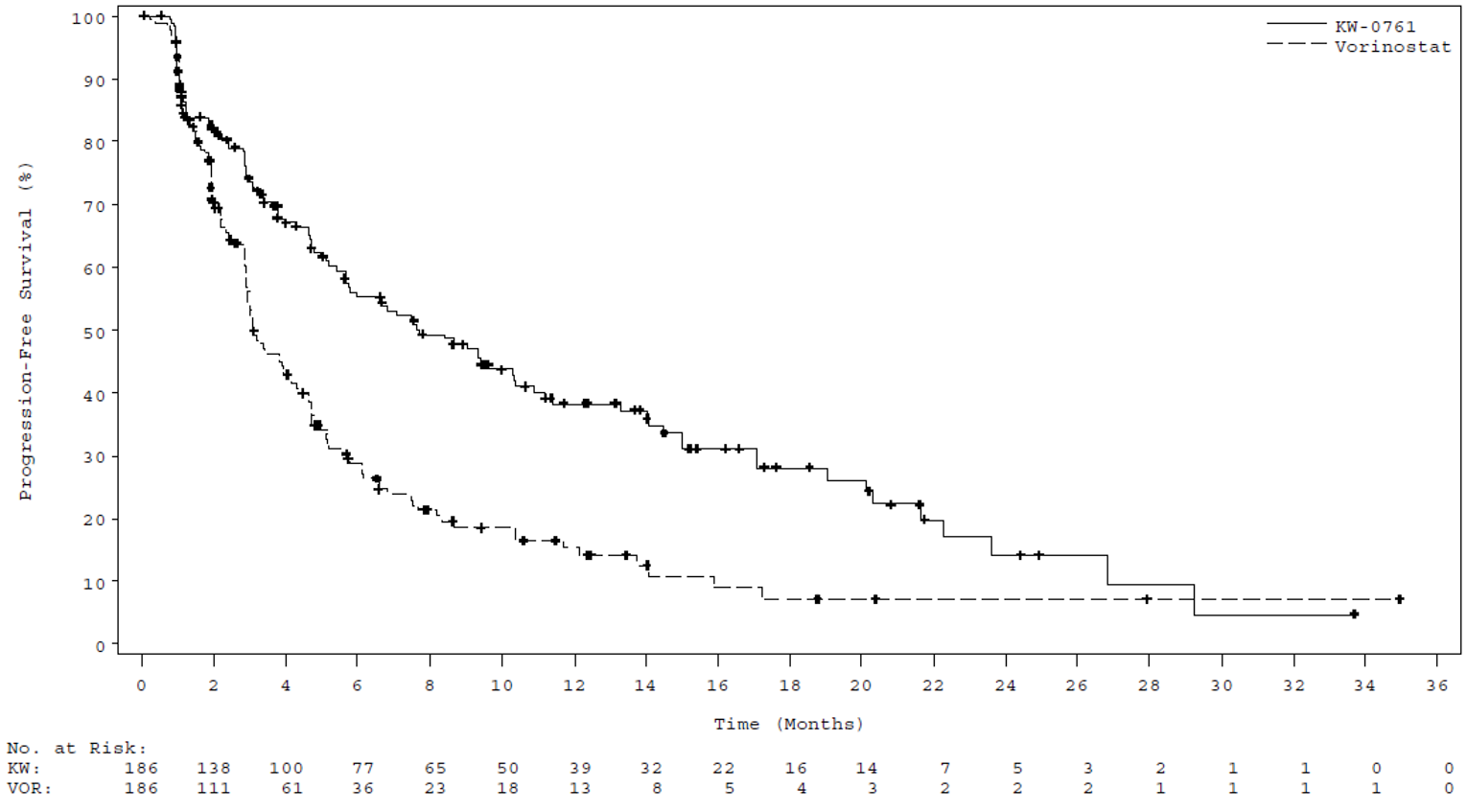
ITT = intention-to-treat population; KW = KW-0761 (mogamulizumab); PFS = progression-free survival; VOR = vorinostat.
Source: MAVORIC (0761 to 010) Clinical Study Report.27
Sensitivity Analysis of PFS
Results of sensitivity analyses for PFS were consistent with the primary analysis and are summarized in Table 44 of Appendix 3. In all cases, the PFS was favoured for the mogamulizumab group, with HRs of 0.52 (95% CI, 0.40 to 0.68), 0.61 (95% CI, 0.47 to 0.78), 0.72 (95% CI, 0.54 to 0.96), and 0.52 (95% CI, 0.40 to 0.68) for sensitivity analyses 1, 2, 3, and 4, respectively.27
In addition to the pre-specified sensitivity analyses, an ad hoc analysis of PFS was conducted that excluded patients with protocol deviations identified by the sponsor as potentially affecting the efficacy results. The results of this analysis were consistent with those of the primary analysis of PFS (HR = 0.63; 95% CI, 0.46 to 0.86; P = 0.0011) with a median PFS of 7.03 months (95% CI, 4.97 to 9.37) for mogamulizumab and 3.13 months (95% CI, 2.87 to 4.30) for vorinostat.27
Additional (Subgroup) Analyses of PFS
Subgroup analyses of PFS during the randomized treatment period were performed based on investigator assessment for the ITT set and efficacy evaluable set. Results of subgroup analyses for PFS in subgroups of interest identified in Table 6 are summarized in Table 23.
In the subgroup of patients with SS, mogamulizumab was associated with a longer PFS compared with vorinostat (HR = 0.32; 95% CI, 0.21 to 0.49), with a median PFS of 13.30 months for mogamulizumab versus 3.13 months for vorinostat. In patients with stage III or IV disease, mogamulizumab was also associated with a longer PFS over vorinostat (HR = 0.36; 95% CI, 0.26 to 0.51), with a median PFS of 10.90 months for mogamulizumab versus 3.00 months for vorinostat.27
Table 23: Subgroup Analysis of PFS Based on Investigator’s Assessment (ITT)
Disease | Mogamulizumab (N = 186) | Vorinostat (N = 186) | HR (95% CI)b | P valuec | ||
|---|---|---|---|---|---|---|
N | Median (95% CI)a | N | Median (95% CI)a | |||
Disease type | ||||||
MF | 105 | 5.40 (3.97 to 7.57) | 99 | 3.10 (2.87 to 4.70) | 0.72 (0.51 to 1.01) | 0.0675 |
SS | 81 | 13.30 (7.70 to 17.07) | 87 | 3.13 (2.83 to 3.87) | 0.32 (0.21 to 0.49) | < 0.0001 |
Disease stage | ||||||
IB or II | 68 | 4.70 (2.90 to 7.47) | 72 | 3.90 (2.87 to 4.73) | 0.88 (0.58 to 1.35) | 0.7166 |
III or IV | 118 | 10.90 (7.03 to 15.03) | 114 | 3.00 (2.83 to 3.87) | 0.36 (0.26 to 0.51) | < 0.0001 |
CI = confidence interval; HR = hazard ratio; ITT = intention-to-treat population; MF = mycosis fungoides; PFS = progression-free survival; SS = Sézary syndrome.
aThe 95% CIs were obtained from SAS PROC LIFETEST using log-log transformation.
bHR based on Cox proportional hazards model with treatment, disease stage, and region as covariates.
cP value (2-sided) was obtained from a stratified log-rank test with disease stage and region as stratification factors.
Source: MAVORIC (0761 to 010) Clinical Study Report.27
Three post hoc analyses were identified evaluating PFS in patients with advanced-stage MF or SS28 by blood involvement29 and by number and type of prior therapy received.46 In the post hoc analysis of patients with advanced-stage MF or SS (stage IIB, IIIA, IIIB, IVA1, IVA2, or IVB; n = 150 in the mogamulizumab group and n = 137 in the vorinostat group), the median PFS was higher than the primary analysis but consistent with the subgroup analysis of patients with stage III or IV disease, with a median PFS of 9.40 months for mogamulizumab and 3.07 months for vorinostat.28
PFS by baseline blood classification (B0, B1, or B2) is summarized in Figure 10 in Appendix 3. The results of this post hoc analysis demonstrated that the PFS achieved with mogamulizumab increased with greater blood involvement. After a median efficacy follow-up of 17.0 months, median PFS was higher for mogamulizumab compared with vorinostat in patients with B2 (11.17 versus 3.30 months) and B1 (8.63 versus 2.53 months) blood involvement. Median PFS did not differ in patients with B0 blood involvement (4.70 months versus 4.37 months).29
Results of another post hoc analysis of PFS in patients in the mogamulizumab group of the MAVORIC trial by number and type of prior treatment received is summarized in Table 46, Table 47, and Figure 11 of Appendix 3. The results for PFS for mogamulizumab were consistent with the primary analysis regardless of the number of prior therapies received, ranging from 5.67 months to 10.30 months, or, based on therapeutic class received, ranging from 5.1 months to 10.3 months. Additionally, PFS was consistent with the primary analysis regardless of the specific systemic therapy received, ranging from 5.1 months for romidepsin to 11.2 months for interferon (Figure 11).46
PFS by Independent Review
PFS assessed by independent review was a secondary end point of the MAVORIC study. As of the data cut-off date, a total of 232 PFS events had been observed;110 (59.1%) in the mogamulizumab group and 122 (65.6%) in the vorinostat group. Results for PFS based on independent review also favoured mogamulizumab, with an HR of 0.64 (95% CI, 0.49 to 0.84). The median PFS by independent review was similar to the primary analysis, with a median PFS of 6.70 months (95% CI, 5.63 to 9.37) in the mogamulizumab group and 3.83 months (95% CI, 3.00 to 4.70) in the vorinostat group (P = 0.0007).27
Health-Related Quality of Life
HRQoL, as measured by the Skindex-29, FACT-G, and EQ-5D-3L up to cycle 5, was a key secondary end point of the MAVORIC trial.27 At baseline, 173 to 183 patients in the mogamulizumab group and 179 to 185 patients in the vorinostat group completed the HRQoL measures. Completion rates for the HRQoL measures were reduced at cycle 5, with 100 to 101 patients completing HRQoL measures in the mogamulizumab group, and 60 to 61 patients completing HRQoL measures in the vorinostat group.30
Skindex-29
The mean domain scores in the cross-sectional population and the change from baseline in the longitudinal-period population for the Skindex-29 are summarized in Table 24. Symptom scores were the highest at baseline for all domains in both the mogamulizumab and vorinostat arms, with higher scores indicating a higher impact of skin disease. The mean symptom domain scores at baseline for the emotional, functional, and symptom domains were 53.1 (SD = 24.47), 46.1 (SD = 25.50), and 61.2 (SD = 20.35) for mogamulizumab, and 46.3 (SD = 24.24), 40.5 (SD = 24.43), and 58.0 (SD = 19.80) for vorinostat. Overall, both the mogamulizumab and vorinostat groups reported an improvement in Skindex-29 scores for emotions, symptoms, and functioning from baseline to cycle 5 (Table 24).30
In general, the magnitude of the improvement from baseline to cycle 5 was greater with mogamulizumab compared with vorinostat for all domains and at all time points. For example, the greatest mean change from baseline in both the mogamulizumab and vorinostat groups at cycle 5 was in the symptom score domain, with mean changes of −18.0 (SD = 24.19), and −8.2 (SD = 20.73), respectively; the mean difference versus vorinostat was −9.8 (95% CI, −17.13 to −2.56) and favoured mogamulizumab (P = 0.0085).30
Functional Assessment of Cancer Therapy–General
Results for the FACT-G scores by domain and change from baseline are summarized in Table 24. In general, the mean FACT-G total score improved from baseline to cycle 5 for both the mogamulizumab and vorinostat groups. The score in the mogamulizumab group increased from 70.9 (SD = 16.87) to 76.8 (SD = 17.41); the score in the vorinostat group increased from 73.9 (SD = 16.94) to 74.1 (SD = 5.81).30
For the mogamulizumab group, the greatest improvement from baseline compared with vorinostat was seen in the physical well-being domain at cycle 1 (mean score of 0.8 versus −1.5), cycle 3 (mean score of 1.7 versus −2.4), and cycle 5 (2.1 versus −1.9). Similar results were shown for the emotional well-being and functional well-being domains; however, no differences were observed for the social well-being domain, with scores decreasing from baseline to cycle 5, indicating a worse QoL in this domain.30
3-Level EQ-5D
Results for the EQ-5D-3L at each time point and the change from baseline are summarized in Table 24. Results for the individual domains of overall mobility, self-care, usual activities, pain or discomfort, and anxiety were not reported. Based on the index score, HRQoL generally improved from baseline at each time point for both mogamulizumab and vorinostat.30
Mean VAS scores at baseline were 64.7 (SD = 21.53) for mogamulizumab and 60.8 (SD = 20.02) for vorinostat. Mean scores for the VAS were higher at cycle 5 compared with baseline for both mogamulizumab (69.0; SD = 20.30) and vorinostat (63.5; SD = 20.08) groups, with the mean difference versus vorinostat favouring mogamulizumab in cycles 3 and 5.30
Table 24: Analysis of Skindex-29, FACT-G, and EQ-5D-3L Scores by Time Point (CSP) and Change From Baseline (LPPa)
Score | Mogamulizumab | Vorinostat | Mean difference (95% Ci) | P valuec | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
n | Mean (SD) score | n | Mean (SD) CFB | P valueb | n | Mean (SD) score | n | Mean (SD) CFB | P valueb | |||
Skindex-29 | ||||||||||||
Emotional scale score | ||||||||||||
Baseline | 173 | 53.1 (24.47) | — | — | — | 178 | 46.3 (24.24) | — | — | — | — | — |
Cycle 1 | 163 | 44.3 (26.47) | 156 | −8.4 (17.31) | < 0.0001 | 170 | 42.8 (25.47) | 165 | −3.7 (17.29) | 0.0060 | −4.7 (−8.47 to −0.87) | 0.0163 |
Cycle 3 | 121 | 38.1 (27.20) | 113 | −14.7 (19.97) | < 0.0001 | 98 | 41.4 (24.92) | 94 | −6.9 (20.27) | 0.0013 | −7.8 (−13.29 to −2.21) | 0.0064 |
Cycle 5 | 100 | 37.8 (27.00) | 93 | −15.5 (23.35) | < 0.0001 | 60 | 42.4 (22.55) | 58 | −5.9 (18.02) | 0.0162 | −9.6 (−16.32 to −2.94) | 0.0051 |
Functional scale score | ||||||||||||
Baseline | 173 | 46.1 (25.50) | — | — | — | 178 | 40.5 (24.43) | — | — | — | — | — |
Cycle 1 | 163 | 39.7 (26.48) | 156 | −5.0 (16.75) | 0.0003 | 171 | 38.5 (24.22) | 165 | −1.8 (18.21) | 0.1980 | −3.1 − (0.98 to 0.70) | 0.1087 |
Cycle 3 | 121 | 34.3 (26.86) | 113 | −10.8 (18.97) | < 0.0001 | 98 | 36.6 (24.87) | 95 | −3.4 (18.73) | 0.0832 | −7.4 (−12.62 to −2.27) | 0.0050 |
Cycle 5 | 99 | 32.0 (25.93) | 92 | −13.2 (21.69) | < 0.0001 | 60 | 36.4 (22.37) | 59 | −1.9 (16.84) | 0.3785 | −11.3 (−17.53 to −5.08) | 0.0005 |
Symptoms scale score | ||||||||||||
Baseline | 173 | 61.2 (20.35) | — | — | — | 179 | 58.0 (19.80) | — | — | — | — | — |
Cycle 1 | 163 | 51.1 (23.02) | 156 | −9.0 (17.98) | < 0.0001 | 171 | 50.9 (20.85) | 166 | −7.5 (16.10) | < 0.0001 | −1.5 (−5.25 to 2.25) | 0.4325 |
Cycle 3 | 121 | 42.9 (22.90) | 113 | −16.8 (22.32) | < 0.0001 | 97 | 50.6 (23.83) | 95 | −7.7 (19.12) | 0.0002 | −9.1 (−14.78 to −3.45) | 0.0017 |
Cycle 5 | 100 | 44.1 (26.32) | 93 | −18.0 (24.19) | < 0.0001 | 60 | 49.9 (21.79) | 59 | −8.2 (20.73) | 0.0036 | −9.8 (−17.13 to −2.56) | 0.0085 |
FACT-G | ||||||||||||
FACT-G total score | ||||||||||||
Baseline | 177 | 70.9 (16.87) | — | — | — | 184 | 73.9 (16.94) | — | — | — | — | — |
Cycle 1 | 168 | 74.1 (17.10) | 164 | 3.0 (10.82) | 0.0005 | 172 | 72.8 (16.12) | 172 | −1.2 (10.95) | 0.1562 | 4.2 (1.87 to 6.54) | 0.0005 |
Cycle 3 | 123 | 76.1 (17.70) | 120 | 5.1 (13.62) | 0.0001 | 99 | 70.5 (18.62) | 97 | −4.5 (14.03) | 0.0020 | 9.6 (5.89 to 13.34) | 0.0000 |
Cycle 5 | 101 | 76.8 (17.41) | 99 | 5.1 (14.01) | 0.0005 | 61 | 74.1 (15.81) | 59 | −3.5 (13.63) | 0.0546 | 8.5 (4.05 to 13.02) | 0.0003 |
Emotional well-being domain | ||||||||||||
Baseline | 180 | 15.5 (4.91) | — | — | — | 185 | 15.8 (4.91) | — | — | — | — | |
Cycle 1 | 168 | 16.4 (4.73) | 165 | 0.9 (3.57) | 0.0011 | 175 | 16.2 (5.02) | 174 | 0.3 (3.35) | 0.2692 | 0.6 (−0.10 to 1.38) | 0.0901 |
Cycle 3 | 124 | 17.2 (4.29) | 121 | 1.5 (3.90) | < 0.0001 | 99 | 15.8 (5.39) | 98 | −0.2 (4.14) | 0.6827 | 1.7 (0.63 to 2.80) | 0.0020 |
Cycle 5 | 101 | 17.3 (4.60) | 99 | 1.4 (4.05) | 0.0007 | 61 | 16.2 (4.95) | 60 | −0.3 (4.21) | 0.5502 | 1.8 (0.41 to 3.10) | 0.0110 |
Functional well-being domain | ||||||||||||
Baseline | 179 | 14.7 (6.54) | — | — | — | 185 | 15.8 (6.34) | — | — | — | — | — |
Cycle 1 | 168 | 15.6 (6.76) | 165 | 0.8 (4.50) | 0.0262 | 174 | 15.9 (5.93) | 173 | 0.1 (4.69) | 0.7261 | 0.7 (−0.32 to 1.65) | 0.1864 |
Cycle 3 | 123 | 16.7 (6.70) | 120 | 1.7 (5.60) | 0.0009 | 99 | 15.4 (6.52) | 98 | −0.9 (5.29) | 0.1149 | 2.6 (1.13 to 4.05) | 0.0006 |
Cycle 5 | 101 | 16.6 (6.48) | 99 | 1.7 (6.04) | 0.0060 | 61 | 16.7 (6.05) | 60 | −0.4 (5.64) | 0.6005 | 2.1 (0.21 to 3.96) | 0.0293 |
Physical well-being domain | ||||||||||||
Baseline | 180 | 19.7 (5.97) | — | — | — | 185 | 20.3 (5.56) | — | — | — | — | — |
Cycle 1 | 169 | 20.8 (5.73) | 166 | 0.8 (4.87) | 0.0436 | 173 | 18.9 (5.77) | 173 | −1.5 (5.02) | 0.0001 | 2.3 (1.24 to 3.35) | 0.0000 |
Cycle 3 | 124 | 21.9 (4.85) | 121 | 1.7 (5.29) | 0.0005 | 99 | 18.5 (6.24) | 98 | −2.4 (5.42) | < 0.0001 | 4.1 (2.65 to 5.53) | 0.0000 |
Cycle 5 | 101 | 22.5 (4.99) | 99 | 2.1 (4.79) | < 0.0001 | 61 | 19.7 (5.03) | 60 | −1.9 (5.67) | 0.0110 | 4.0 (2.28 to 5.75) | 0.0000 |
Social well-being domain | ||||||||||||
Baseline | 181 | 21.0 (5.81) | — | — | — | 185 | 21.8 (5.66) | — | — | — | — | — |
Cycle 1 | 170 | 21.3 (5.36) | 167 | 0.5 (4.25) | 0.1159 | 173 | 21.6 (5.38) | 173 | −0.1 (4.33) | 0.7681 | 0.6 (−0.30 to 1.53) | 0.1859 |
Cycle 3 | 124 | 20.3 (6.31) | 121 | −0.1 (5.58) | 0.8263 | 99 | 20.7 (5.80) | 98 | −1.0 (5.00) | 0.0575 | 0.9 (−0.55 to 2.27) | 0.2313 |
Cycle 5 | 101 | 20.4 (5.83) | 99 | −0.2 (5.00) | 0.7302 | 61 | 21.5 (5.90) | 60 | −0.7 (4.61) | 0.2470 | 0.5 (−1.02 to 2.06) | 0.5039 |
3-level EQ-5D | ||||||||||||
Index scale score | ||||||||||||
Baseline | 180 | 0.742 (0.1948) | — | — | — | 183 | 0.748 (0.1872) | — | — | — | — | — |
Cycle 1 | 167 | 0.767 (0.2051) | 164 | 0.016 (0.1762) | 0.2511 | 174 | 0.768 (0.1829) | 171 | 0.011 (0.1656) | 0.3698 | 0.004 (−0.0323 to 0.0412) | 0.8117 |
Cycle 3 | 123 | 0.812 (0.1697) | 121 | 0.064 (0.1895) | 0.0003 | 100 | 0.767 (0.1871) | 98 | 0.009 (0.1565) | 0.5569 | 0.054 (0.0084 to 0.1006) | 0.0207 |
Cycle 5 | 101 | 0.815 (0.1559) | 101 | 0.068 (0.1881) | 0.0005 | 60 | 0.787 (0.1523) | 58 | 0.007 (0.1525) | 0.7189 | 0.061 (0.0064 to 0.1148) | 0.0288 |
Visual Analogue Scale | ||||||||||||
Baseline | 183 | 60.9 (22.10) | — | — | — | 180 | 60.8 (20.02) | — | — | — | — | — |
Cycle 1 | 168 | 64.7 (21.53) | 167 | 2.7 (21.47) | 0.1015 | 174 | 60.9 (21.08) | 169 | 0.2 (18.53) | 0.9109 | 2.6 (−1.73 to 6.88) | 0.2401 |
Cycle 3 | 122 | 68.3 (19.67) | 122 | 6.0 (20.60) | 0.0017 | 100 | 61.5 (20.59) | 97 | −1.0 (20.19) | 0.6233 | 7.0 (1.53 to 12.46) | 0.0124 |
Cycle 5 | 101 | 69.0 (20.30) | 101 | 7.2 (22.78) | 0.0019 | 61 | 63.5 (20.08) | 58 | −1.1 (18.27) | 0.6423 | 8.4 (1.83 to 14.88) | 0.0124 |
CFB = change from baseline; CI = confidence interval; CSP = cross-sectional analysis population; EQ-5D-3L = 3-level EQ-5D; FACT-G = Functional Assessment of Cancer Therapy–General; LPP = longitudinal-period population; QoL = quality of life; SD = standard deviation.
aThe LPP population consists of patients who have survived and maintained in the study from baseline to day 1 of cycle 1 to day 1 of the specified cumulative cycle and have available QoL data for that time point.
bP value from 1 sample t-test for change in score from baseline.
cP value and 95% CI from t-test for difference in mean change in Skindex-29, FACT-G, and EQ-5D-3L domain or overall scores from baseline between treatment groups.
Source: Sponsor’s internal QoL report.30
Pruritis Evaluation: ItchyQoL and Pruritis Likert Scale
Changes in pruritis via the Pruritis Likert Scale and ItchyQoL was a secondary outcome of the MAVORIC trial. Minimal differences between treatment groups were observed for the Pruritus ItchyQoL emotion, functional, and symptom scores with mean scores from baseline to cycle 5 decreasing for both mogamulizumab, and vorinostat groups.27,30
Table 25: Analysis of ItchyQoL and Pruritus Likert Scale Scores by Time Point
Detail | Mogamulizumab | Vorinostat | ||
|---|---|---|---|---|
n | Mean score (SD) | n | Mean score (SD) | |
ItchyQoL, mean (SD) | ||||
Emotional score | ||||
Baseline | 178 | 3.1 (1.03) | 184 | 2.9 (1.05) |
Cycle 1 | 166 | 2.8 (1.07) | 171 | 2.6 (1.06) |
Cycle 3 | 124 | 2.5 (1.07) | 100 | 2.5 (1.04) |
Cycle 5 | 102 | 2.4 (1.07) | 61 | 2.5 (1.00) |
Functional score | ||||
Baseline | 182 | 3.3 (0.93) | 185 | 3.2 (0.94) |
Cycle 1 | 167 | 3.1 (1.04) | 167 | 2.9 (1.01) |
Cycle 3 | 123 | 2.7 (1.02) | 100 | 2.7 (1.09) |
Cycle 5 | 102 | 2.7 (1.02) | 62 | 2.7 (1.03) |
Symptom score | ||||
Baseline | 182 | 3.1 (0.84) | 184 | 3.0 (0.83) |
Cycle 1 | 169 | 2.8 (0.87) | 169 | 2.7 (0.88) |
Cycle 3 | 124 | 2.5 (0.87) | 100 | 2.6 (0.93) |
Cycle 5 | 102 | 2.6 (1.02) | 62 | 2.6 (0.89) |
Pruritis Likert scale score, mean (SD) | ||||
Baseline | 180 | 6.2 (2.87) | 180 | 6.3 (2.72) |
Cycle 1 | 167 | 5.2 (2.74) | 170 | 5.0 (2.79) |
Cycle 3 | 120 | 4.3 (2.72) | 99 | 4.7 (2.94) |
Cycle 5 | 100 | 4.7 (2.93) | 61 | 4.3 (3.06) |
SD = standard deviation.
Source: Sponsor’s internal quality of life report.30
Overall Survival
OS was an exploratory end point of the MAVORIC trial and is summarized in Table 26 and Figure 3. At the time of data cut-off, 87 patients (23.4%) had died; 40 in the mogamulizumab group and 47 in the vorinostat group. There was no difference in survival advantage between mogamulizumab and vorinostat (HR = 0.93; 95% CI, 0.61 to 1.43). The median OS was NE for mogamulizumab and was 43.93 months (95% CI, 43.57 to NE) for vorinostat (P = 0.9439).27
Table 26: Summary of Overall Survival (ITT)a
Category | Mogamulizumab (N = 186) | Vorinostat (N = 186) |
|---|---|---|
OS event, n (%) | 40 (21.5) | 47 (25.3) |
Censored, n (%) | 146 (78.5) | 139 (74.7) |
Median OS, months (95% CI)b | NE (NE to NE) | 43.93 (43.57 to NE) |
HR (95% CI)c | 0.93 (0.61 to 1.43) | |
Log-rank P valuec | 0.9439 | |
OS rate, % (95% CI)d | ||
6 months | 94.2 (89.5 to 96.9) | 92.3 (87.3 to 95.4) |
12 months | 89.9 (84.3 to 93.6) | 85.3 (79.2 to 89.8) |
18 months | 80.7 (73.0 to 86.4) | 81.0 (74.2 to 86.2) |
24 months | 74.6 (65.6 to 81.6) | 76.4 (68.6 to 82.5) |
30 months | 67.1 (56.0 to 76.0) | 67.0 (57.2 to 75.1) |
36 months | 65.0 (53.3 to 74.4) | 64.5 (53.8 to 73.4) |
42 months | 52.9 (34.3 to 68.5) | 64.5 (53.8 to 73.4) |
CI = confidence interval; HR = hazard ratio; ITT = intention-to-treat population; NE = not estimable; OS = overall survival.
aDoes not include crossover.
bThe 95% CIs were obtained from the SAS PROC LIFETEST using a log-log transformation.
cHRs and 95% CIs are based on Cox proportional hazards model with treatment, disease type, disease stage, and region as covariates. P value (2-sided) was obtained from a stratified log-rank test with disease type, disease stage, and region as stratification factors.
dKaplan-Meier estimate.
Source: MAVORIC (0761 to 010) Clinical Study Report.27
Figure 3: Kaplan-Meier Curve of OS (ITT)
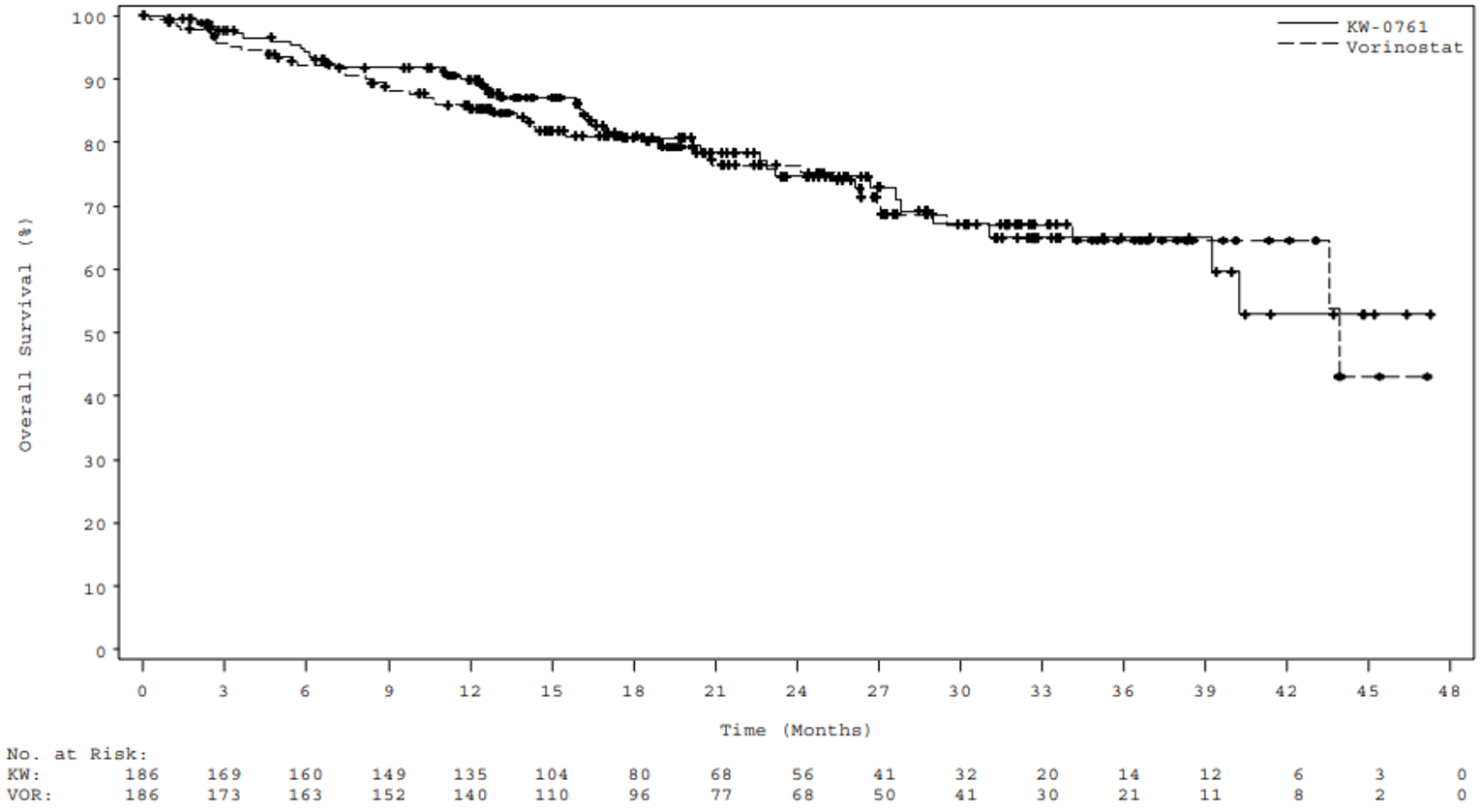
ITT = intention-to-treat population; KW = KW-0761 (mogamulizumab); OS = overall survival; VOR = vorinostat.
Source: MAVORIC (0761 to 010) Clinical Study Report.27
Additional analyses of OS adjusting for the 1-way crossover design using the RPSFT and IPCW methods are summarized in Table 48 of Appendix 3. The results of analyses adjusting for crossover demonstrated no difference in survival advantage for patients treated with mogamulizumab compared with vorinostat, with an HR of 0.739 (95% CI, 0.479 to 1.141) for the RPSFT method and an HR of 0.508 (95% CI, 0.172 to 2.905) for the IPCW method.35 When patients were censored at the time crossover occurred, the HR was 0.709 (95% CI, 0.405 to 1.242).
Time-to-Treatment Failure
TTF was an exploratory end point of the MAVORIC trial, defined as the time from the day of randomization until the discontinuation of randomized treatment due to any reason. The HR for TTF was 0.58 (95% CI, 0.47 to 0.72) in favour of mogamulizumab, with a median TTF of 5.80 months for mogamulizumab compared with 2.87 months for vorinostat.27 The reason for treatment failure is summarized in Table 15. No information on the proportion of patients undergoing a subsequent stem-cell transplant was provided.
Harms
Only those harms identified in the review protocol (Table 6) are reported subsequently.
Adverse Events
TEAEs occurring in the randomized portion (i.e., before crossover) of the MAVORIC trial are summarized in Table 27. A total of 179 (97.3%) and 185 (99.5%) patients had TEAEs with mogamulizumab and vorinostat, respectively. Gastrointestinal disorders were reported more frequently with vorinostat compared with mogamulizumab (93 versus 152 patients; 50.5% versus 81.7%). The most frequently reported TEAEs with mogamulizumab were IRRs (61; 33.2%), drug eruptions (44; 23.9%), diarrhea (43; 23.4%), and fatigue (43; 23.4%). IRRs and drug eruptions occurred at a notably higher rate with mogamulizumab versus vorinostat: there were 61 (33.2%) IRRs for mogamulizumab versus 1 (0.5%) for vorinostat while, for drug eruptions, there were 44 (23.9%) with mogamulizumab versus 1 (0.5%) with vorinostat. The most frequently reported TEAEs with vorinostat were diarrhea (115; 61.8%), nausea (79; 42.5%), fatigue (70; 37.6%), and thrombocytopenia (57; 30.6%).27
Overall, the incidence of specific TEAEs varied by SOC, with TEAEs of infections and infestations, skin and subcutaneous tissue disorders, musculoskeletal and connective tissue disorders, injury, poisoning, and procedural complications reported more frequently with mogamulizumab compared with vorinostat, and TEAEs associated with gastrointestinal disorders, general disorders or administration site conditions, nervous system disorders, investigations, metabolism and nutrition disorders, and blood and lymphatic disorders were reported more frequently in the vorinostat group compared with mogamulizumab, with variation in most of the TEAE preferred terms.27
The overall incidence of grade 3 or greater 3 TEAEs was similar between mogamulizumab (78; 42.4%) and vorinostat (85; 45.7%) and was generally similar across SOCs. However, the incidence of grade 3 or greater infections and infestations was higher with mogamulizumab (32; 17.4%) compared with vorinostat (19; 10.2%), while grade 3 or greater gastrointestinal disorders, general disorders and administration site conditions, and blood and lymphatic system disorders occurred more frequently with vorinostat when compared with mogamulizumab: 4 (2.2%) versus 17 (9.1%), 8 (4.3%) versus 17 (9.1%), and 3 (1.6%) versus 18 (9.7%), respectively.27
In patients who crossed over to mogamulizumab (n = 136), 127 (93.4%) experienced TEAEs of any grade; 47 (34.6%) of these TEAEs were grade 3 or greater.27
Table 27: TEAEs Reported by 5% or More of Patients During Randomized Treatment by System Organ Class and Preferred Term (Safety Analysis Set)
System organ class preferred terma | Mogamulizumab (N = 184) | Vorinostat (N = 186) | ||
|---|---|---|---|---|
All grades | Grade ≥ 3 | All grades | Grade ≥ 3 | |
Patients with any TEAEs, n (%) | 179 (97.3) | 78 (42.4) | 185 (99.5) | 85 (45.7) |
Gastrointestinal disorders | 93 (50.5) | 4 (2.2) | 152 (81.7) | 17 (9.1) |
Diarrhea | 43 (23.4) | 1 (0.5) | 115 (61.8) | 9 (4.8) |
Nausea | 28 (15.2) | 1 (0.5) | 79 (42.5) | 3 (1.6) |
Constipation | 21 (11.4) | 1 (0.5) | 34 (18.3) | 2 (1.1) |
Vomiting | 11 (6.0) | 0 | 24 (12.9) | 1 (0.5) |
Abdominal pain | 7 (3.8) | 0 | 21 (11.3) | 0 |
Dry mouth | 4 (2.2) | 0 | 17 (9.1) | 0 |
Abdominal pain upper | 1 (0.5) | 0 | 11 (5.9) | 1 (0.5) |
Dyspepsia | 1 (0.5) | 0 | 11 (5.9) | 0 |
General disorders and administration site conditions | 106 (57.6) | 8 (4.3)b | 126 (67.7) | 17 (9.1) |
Fatigue | 43 (23.4) | 3 (1.6) | 70 (37.6) | 11 (5.9) |
Edema peripheral | 27 (14.7) | 0 | 27 (14.5) | 1 (0.5) |
Pyrexia | 31 (16.8) | 1 (0.5) | 11 (5.9) | 0 |
Asthenia | 10 (5.4) | 0 | 27 (14.5) | 4 (2.2) |
Chills | 13 (7.1) | 0 | 14 (7.5) | 0 |
Infections and infestations | 118 (64.1) | 32 (17.4)c | 93 (50.0) | 19 (10.2) |
Skin infection | 17 (9.2) | 0 | 13 (7.0) | 3 (1.6) |
Upper respiratory tract infection | 19 (10.3) | 0 | 9 (4.8) | 2 (1.1) |
Nasopharyngitis | 12 (6.5) | 0 | 15 (8.1) | 0 |
Urinary tract infection | 12 (6.5) | 0 | 15 (8.1) | 0 |
Folliculitis | 13 (7.1) | 0 | 4 (2.2) | 1 (0.5) |
Cellulitis | 6 (3.3) | 4 (2.2) | 10 (5.4) | 4 (2.2) |
Oral candidiasis | 10 (5.4) | 0 | 1 (0.5) | 0 |
Skin and subcutaneous tissue disorders | 97 (52.7) | 10 (5.4) | 78 (41.9) | 9 (4.8) |
Alopecia | 13 (7.1) | 0 | 36 (19.4) | 0 |
Drug eruption | 44 (23.9) | 8 (4.3) | 1 (0.5) | 0 |
Nervous system disorders | 65 (35.3) | 2 (1.1) | 101 (54.3) | 7 (3.8) |
Dysgeusia | 6 (3.3) | 0 | 54 (29.0) | 1 (0.5) |
Headache | 23 (12.5) | 0 | 29 (15.6) | 1 (0.5) |
Dizziness | 12 (6.5) | 0 | 19 (10.2) | 0 |
Paresthesia | 5 (2.7) | 0 | 14 (7.5) | 0 |
Investigations | 65 (35.3) | 8 (4.3) | 95 (51.1) | 11 (5.9) |
Blood creatinine increased | 6 (3.3) | 0 | 52 (28.0) | 0 |
Weight decreased | 11 (6.0) | 1 (0.5) | 33 (17.7) | 2 (1.1) |
Platelet count decreased | 4 (2.2) | 0 | 19 (10.2) | 0 |
Aspartate aminotransferase increased | 8 (4.3) | 2 (1.1) | 12 (6.5) | 1 (0.5) |
Alanine aminotransferase increased | 10 (5.4) | 0 | 9 (4.8) | 1 (0.5) |
Weight increased | 14 (7.6) | 1 (0.5) | 2 (1.1) | 0 |
Metabolism and nutrition disorders | 59 (32.1) | 13 (7.1) | 77 (41.4) | 15 (8.1) |
Decreased appetite | 14 (7.6) | 2 (1.1) | 46 (24.7) | 2 (1.1) |
Hyperglycemia | 15 (8.2) | 2 (1.1) | 14 (7.5) | 2 (1.1) |
Hypokalemia | 10 (5.4) | 0 | 12 (6.5) | 2 (1.1) |
Musculoskeletal and connective tissue disorders | 67 (36.4) | 5 (2.7)d | 59 (31.7) | 6 (3.2) |
Muscle spasm | 9 (4.9) | 0 | 29 (15.6) | 2 (1.1) |
Back pain | 18 (9.8) | 1 (0.5) | 9 (4.8) | 1 (0.5) |
Arthralgia | 13 (7.1) | 1 (0.5) | 11 (5.9) | 0 |
Pain in extremity | 12 (6.5) | 0 | 9 (4.8) | 1 (0.5) |
Myalgia | 11 (6.0) | 0 | 8 (4.3) | 2 (1.1) |
Blood and lymphatic system disorders | 47 (25.5) | 3 (1.6) | 76 (40.9) | 18 (9.7) |
Thrombocytopenia | 21 (11.4) | 0 | 57 (30.6) | 13 (7.0) |
Anemia | 19 (10.3) | 2 (1.1) | 19 (10.2) | 2 (1.1) |
Neutropenia | 5 (2.7) | 1 (0.5) | 10 (5.4) | 3 (1.6) |
Injury, poisoning, and procedural complications | 81 (44.0) | 7 (3.8) | 28 (15.1) | 2 (1.1) |
Infusion-related reaction | 61 (33.2) | 3 (1.6) | 1 (0.5)e | 0 |
Fall | 11 (6.0) | 1 (0.5) | 3 (1.6) | 0 |
Respiratory, thoracic, and mediastinal disorders | 56 (30.4) | 7 (3.8) | 42 (22.6) | 7 (3.8) |
Cough | 18 (9.8) | 0 | 15 (8.1) | 0 |
Oropharyngeal pain | 10 (5.4) | 1 (0.5) | 5 (2.7) | 0 |
Vascular disorders | 29 (15.8) | 12 (6.5) | 38 (20.4) | 13 (7.0) |
Hypertension | 17 (9.2) | 8 (4.3) | 25 (13.4) | 12 (6.5) |
Eye disorders | 34 (18.5) | 3 (1.6) | 32 (17.2) | 0 |
Vision blurred | 8 (4.3) | 0 | 12 (6.5) | 0 |
Dry eye | 7 (3.8) | 0 | 11 (5.9) | 0 |
Psychiatric disorders | 32 (17.4) | 2 (1.1) | 28 (15.1) | 2 (1.1) |
Insomnia | 16 (8.7) | 0 | 14 (7.5) | 0 |
Depression | 11 (6.0) | 2 (1.1) | 6 (3.2) | 0 |
MedDRA = Medical Dictionary for Regulatory Activities; TEAE = treatment-emergent adverse event.
aMedDRA Version 15.1 was used for coding.
bIncludes 1 grade 5 TEAE (disease progression).
cGrade ≥ 3 infections and infestations TEAEs reported for patients in the mogamulizumab group but not shown in the table (i.e., reported by < 5.0% of patients in either group) include pneumonia (n = 4), sepsis (n = 3; 1 grade 5), bacteremia (n = 2), herpes simplex (n = 2), osteomyelitis (n = 2); all other events occurred in 1 patient each, including grade 5 pneumococcal pneumonia.
dIncludes 1 grade 5 TEAE (polymyositis).
eOne patient had an infusion reaction on day 1 of crossover to mogamulizumab treatment (17 days after the last dose of vorinostat) that was indicated as possibly related to vorinostat (and mogamulizumab).
Source: MAVORIC (0761 to 010) Clinical Study Report.27
Serious Adverse Events
SAEs in the MAVORIC trial are summarized in Table 28. The incidence of SAEs was higher in the mogamulizumab group (69; 37.5%) compared with vorinostat (46; 24.7%), driven by a higher incidence of AEs associated with the infections and infestations SOC (30 versus 20; 16.3% versus 10.8%) and general disorders and administration site conditions SOC (15 versus 8; 8.2% versus 4.3%). In the mogamulizumab group, the most frequently reported SAEs were pyrexia (8; 4.3%), cellulitis (5; 2.7%), and pneumonia (4; 2.2%) while, in the vorinostat group, the most frequently reported SAEs were cellulitis (6; 3.2%), pulmonary embolism (6; 3.2%), and sepsis (5; 2.7%). One patient in the mogamulizumab group experienced an SAE of acute myocardial infarction (grade 3).27
Of the 184 patients who were randomized to receive mogamulizumab, 5 (2.7%) had fatal AEs reported as of the data cut-off date. Of these, 2 died due to treatment-emergent events (pneumococcal pneumonia and polymyositis, and sepsis), 1 additional patient died due to pneumonia and sepsis that occurred more than 90 days after the last dose of study medication (not treatment-emergent, not related); and 2 patients died due to underlying disease. There were 9 (4.8%) deaths in the vorinostat group during the randomized treatment period.27
During the crossover portion of the study, 36 (26.5%) SAEs were reported in patients who crossed over to mogamulizumab, including 4 (2.9%) with IRRs and 2 (1.5%) with cellulitis. One patient also experienced a grade 3 myocardial infarction during the crossover period.27
Table 28: Treatment-Emergent SAEs Reported by 1% or More of Patients During Randomized Treatment (Safety Analysis Set)
System organ class preferred term, n (%)a | Mogamulizumab (N = 184) | Vorinostat (N = 186) |
|---|---|---|
Patients with any treatment-emergent SAEs | 69 (37.5) | 46 (24.7) |
Infections and infestations | 30 (16.3) | 20 (10.8) |
Cellulitis | 5 (2.7) | 6 (3.2) |
Sepsis | 3 (1.6) | 5 (2.7) |
Pneumonia | 4 (2.2) | 2 (1.1) |
Skin infection | 0 | 3 (1.6) |
Bacteremia | 2 (1.1) | 0 |
Bronchitis | 2 (1.1) | 0 |
Herpes simplex | 2 (1.1) | 0 |
Osteomyelitis | 2 (1.1) | 0 |
Upper respiratory tract infection | 0 | 2 (1.1) |
General disorders and administration site conditions | 15 (8.2) | 8 (4.3) |
Pyrexia | 8 (4.3) | 1 (0.5) |
Asthenia | 0 | 2 (1.1) |
Respiratory, thoracic, and mediastinal disorders | 9 (4.9) | 6 (3.2) |
Pulmonary embolism | 0 | 6 (3.2) |
Respiratory failure | 2 (1.1) | 0 |
Metabolism and nutrition disorders | 6 (3.3) | 4 (2.2) |
Hypercalcemia | 3 (1.6) | 0 |
Injury, poisoning, and procedural complications | 6 (3.3) | 2 (1.1) |
Infusion-related reaction | 3 (1.6) | 0 |
Fall | 2 (1.1) | 0 |
Musculoskeletal and connective tissue disorders | 6 (3.3) | 2 (1.1) |
Arthralgia | 2 (1.1) | 1 (0.5) |
Skin and subcutaneous tissue disorders | 4 (2.2) | 3 (1.6) |
Drug eruption | 2 (1.1) | 0 |
Blood and lymphatic system disorders | 1 (0.5) | 5 (2.7) |
Thrombocytopenia | 0 | 3 (1.6) |
Neutropenia | 0 | 2 (1.1) |
Renal and urinary disorders | 3 (1.6) | 3 (1.6) |
Renal failure acute | 2 (1.1) | 1 (0.5) |
Vascular disorders | 4 (2.2) | 1 (0.5) |
Embolism | 2 (1.1) | 0 |
Investigations | 2 (1.1) | 1 (0.5) |
Alanine aminotransferase increased | 2 (1.1) | 0 |
Aspartate aminotransferase increased | 2 (1.1) | 0 |
MedDRA = Medical Dictionary for Regulatory Activities; SAE = serious adverse event.
aMedDRA Version 15.1 was used for coding.
Source: MAVORIC (0761 to 010) Clinical Study Report.27
Withdrawals Due to Adverse Events
During the randomized treatment period, 35 patients (19.0%) in the mogamulizumab group and 43 patients (23.1%) in the vorinostat group discontinued treatment due to AEs (Table 29). The most frequent TEAE leading to discontinuation of mogamulizumab was drug eruption (13; 7.1%), while the most frequent TEAE leading to discontinuation for vorinostat was fatigue (8; 4.3%). During the crossover portion of the study, 30 patients (22.1%) who crossed over to mogamulizumab reported AEs leading to discontinuation, including 12 (8.8%) with drug eruptions and 4 (2.9%) with IRRs.27
Table 29: TEAEs Leading to Discontinuation in 1% or More of Patients During Randomized Treatment (Safety Analysis Set)
System organ class preferred term, n (%)a | Mogamulizumab (N = 184) | Vorinostat (N = 186) |
|---|---|---|
Patients with any TEAE leading to discontinuation | 35 (19.0) | 43 (23.1) |
General disorders and administration site conditions | 7 (3.8) | 12 (6.5) |
Fatigue | 1 (0.5) | 8 (4.3) |
Disease progression | 3 (1.6) | 1 (0.5) |
Asthenia | 0 | 3 (1.6) |
Skin and subcutaneous tissue disorders | 14 (7.6) | 3 (1.6) |
Drug eruption | 13 (7.1) | 0 |
Infections and infestations | 9 (4.9) | 2 (1.1) |
Skin infection | 2 (1.1) | 0 |
Gastrointestinal disorders | 0 | 8 (4.3) |
Diarrhea | 0 | 5 (2.7) |
Nausea | 0 | 4 (2.2) |
Vomiting | 0 | 3 (1.6) |
Metabolism and nutrition disorders | 3 (1.6) | 5 (2.7) |
Decreased appetite | 1 (0.5) | 2 (1.1) |
Musculoskeletal and connective tissue disorders | 3 (1.6) | 4 (2.2) |
Muscular weakness | 0 | 2 (1.1) |
Myalgia | 0 | 2 (1.1) |
Respiratory, thoracic, and mediastinal disorders | 3 (1.6) | 4 (2.2) |
Pulmonary embolism | 0 | 4 (2.2) |
Blood and lymphatic system disorders | 0 | 6 (3.2) |
Thrombocytopenia | 0 | 5 (2.7) |
Investigations | 0 | 6 (3.2) |
Weight decreased | 0 | 4 (2.2) |
Nervous system disorders | 1 (0.5) | 4 (2.2) |
Dysgeusia | 0 | 2 (1.1) |
Renal and urinary disorders | 3 (1.6) | 1 (0.5) |
Renal failure acute | 2 (1.1) | 0 |
Vascular disorders | 1 (0.5) | 2 (1.1) |
Deep vein thrombosis | 0 | 2 (1.1) |
MedDRA = Medical Dictionary for Regulatory Activities; TEAE = treatment-emergent adverse event.
aMedDRA Version 15.1 was used for coding.
Source: MAVORIC (0761 to 010) Clinical Study Report.27
Notable Harms
Infusion-Related Reactions
The incidence of IRRs is summarized in Table 27. IRRs occurred in 61 patients (33.2%) with mogamulizumab compared with 1 patient (0.5%) for vorinostat. The majority of IRRs were of grade 1 or 2 severity (31.5%), with only 1.6% of patients experiencing a grade 3 IRR. No cases of IRR led to discontinuation of treatment.27
During the crossover portion of the study, 50 patients (36.8%) who crossed over to mogamulizumab experienced IRRs. Most IRRs were of grade 1 or 2 severity (32.4%), while 4.4% were grade 3. Four (2.9%) patients had IRRs that were reported as SAEs during the crossover portion; of these, 2 patients discontinued treatment due to infusion reaction. Two additional patients also discontinued treatment due to non-serious infusion reactions.27
Infections
Infections and infestations are summarized by preferred term in Table 27. Infections and infestations occurred in 118 patients (64.1%) with mogamulizumab and 93 patients (50%) with vorinostat. Higher rates of upper respiratory tract infection (19 versus 9; 10.3% versus 4.8%), folliculitis (13 versus 4; 7.1% versus 2.2%), and oral candidiasis (10 versus 1; 5.4% versus 0.5%) were observed with mogamulizumab. The most frequent infections with vorinostat were nasopharyngitis and urinary tract infections (12 versus 15; 6.5% versus 8.1% for each). A total of 9 patients (4.9%) in the mogamulizumab group had treatment-emergent infections that led to discontinuation of the study compared with 2 patients (1.1%) in the vorinostat group.27
In the crossover portion of the MAVORIC trial, 15.4% of patients who crossed over to mogamulizumab experienced infections, of which 18 (13.2%) were grade 1 or 2, and 3 (2.2%) were grade 3. No infections led to discontinuation of treatment during the crossover portion of the trial.27
Immune-Related AEs
Though not expressly reported as a group of TEAEs in the MAVORIC trial, following a request for additional information, the sponsor provided the frequency of specific immune-related AEs (Table 30). The most frequently occurring immune-related TEAE with mogamulizumab was hypothyroidism, occurring in 5 patients (2.7%) compared with 1 patient (0.5%) in the vorinostat arm.27
Table 30: Immune-Related TEAEs in the MAVORIC Trial
Preferred term, n (%) | Mogamulizumab (N = 184) | Vorinostat (N = 186) |
|---|---|---|
Hypothyroidism | 5 (2.7) | 1 (0.5) |
Myositis | 2 (1.1) | 0 |
Myocarditis | 1 (0.5) | 0 |
Pneumonitis | 1 (0.5) | 1 (0.5) |
Polymyositis | 1 (0.5) | 0 |
TEAE = treatment-emergent adverse event.
Source: MAVORIC (0761 to 010) Clinical Study Report.27
Stevens-Johnson Syndrome
Despite warnings and precautions in the product monograph, no cases of Stevens-Johnson syndrome were reported in the MAVORIC trial.
Toxic Epidermic Necrolysis
Despite warnings and precautions in the product monograph, no cases of toxic epidermal necrolysis were reported in the MAVORIC trial.
Critical Appraisal
Internal Validity
The MAVORIC trial was an open-label, phase III RCT. Appropriate methods for randomization (via CTIVRS) treatment and allocation (via screening numbers) were employed. Randomized patients were stratified by disease type (MF or SS) and disease stage (IB or II versus stage III or IV). The methods for randomization were considered by the CADTH review team to result in a low risk of bias, as patients were randomly assigned a treatment group using technology that ensured that relatively equal proportions of patients would be randomized not only to each treatment group, but to each pre-specified stratification factor. In general, patients did not differ with regard to baseline characteristics, indicating that randomization was successful.
As of the data cut-off date, a total of 157 patients (84.4%) randomized to mogamulizumab discontinued treatment, which was considered to be high. There were no notable differences between patients discontinuing randomized treatment, with most patients in both groups discontinuing due to disease progression. The median time-to-treatment discontinuation was not reported. The clinical experts consulted by CADTH considered the rate of death in the randomized treatment phase of the MAVORIC trial to be low.
An open-label design was used for the MAVORIC trial, which could potentially increase the risk of bias in the reporting of outcomes that are subjective in measurement and interpretation, such as response, HRQoL, and AEs. The primary and key secondary end points of the MAVORIC trial were therefore subject to potential bias, given the open-label nature of the study. Disease progression was based on objective and quantitative assessments (CT, mSWAT, skin photographs, and flow cytometry) conducted at any time before the scheduled assessments, creating sources of assessment or measurement bias and interval censoring. Investigators also chose not to blind the treatment groups because of complexities related to differing routes of administration (IV versus oral), and the differing side effect profiles for mogamulizumab and vorinostat. The blinded independent review was instituted to assess for any potential investigator bias during the randomized treatment period in this open-label study.
The MAVORIC trial also included a 1-way crossover design, where patients whose condition had failed to respond to treatment with vorinostat were able to crossover to the mogamulizumab group. For patients who were crossed over to treatment with mogamulizumab, there had to have been at least 2 weeks between the last dose of vorinostat and the first dose of mogamulizumab, which was deemed by the CADTH review team to be sufficient for washout, given the mean terminal half-life for vorinostat. Any analyses for the 136 patients who crossed over were purely descriptive and should be interpreted with caution. Exposure to mogamulizumab was similar between those randomized to mogamulizumab and those who crossed over from vorinostat, suggesting that mogamulizumab remains efficacious after treatment with vorinostat.
In total, there were 10 protocol amendments to the original protocol spanning 2012 to 2018, many relating to the inclusion and exclusion criteria of patients as well as additional analyses of the data. It was unclear how many patients were enrolled at each amendment; thus, it is uncertain whether there were any impacts on the generalizability of the results or if any bias was introduced. The addition of exploratory analyses to the statistical analysis plan of PFS for patients with any mogamulizumab exposure compared with those who received vorinostat only, and analyses adjusting for crossover in OS, is expected to reduce the reliability of the methods.
The end points considered in the study were clinically appropriate for this population per the clinical experts consulted by CADTH and have been recommended by the ISCL, the United States Cutaneous Lymphoma Consortium, and the Cutaneous Lymphoma Task Force of the EORTC.31 Additionally, survival was noted to be an important outcome for patients. However, patients with clinical progression were censored in the PFS analysis, which may bias the results of PFS through reporting, assessment, or measurement bias. Acceptable methods to account for multiplicity were used in the MAVORIC trial to adjust for the overall studywise type I error rate for the key secondary end points of ORR and change from baseline in Skindex-29, FACT-G, and EQ-5D-3L using the Šidák adjusted P value method. Missing data were handled according to the scoring guidelines for the specific HRQoL questionnaires. Other secondary and exploratory end points were not controlled for either multiplicity or missing data. Though the choice of primary and secondary outcomes was appropriate, the MAVORIC trial was not powered to detect differences in OS between mogamulizumab and vorinostat. According to both the ISCL and the EORTC, evaluation of OS is not optimal in clinical trials of patients with MF or SS, and expected survival is far longer than the course of the study and the potential exists for survival to be impacted by treatment(s) given after study trial conclusion.31 Additionally, the analysis of OS was considered immature, with a median efficacy follow-up of 17.0 months, and was confounded by the 1-way crossover design, as outcomes measured after progression could be biased, and the open-label design and 1-way crossover may result in nonadherence to randomized treatment, potentially reducing the power of the ITT analysis. Moreover, given that OS was an exploratory end point, it must be treated as such, as it was not controlled for multiplicity and needs to be interpreted with consideration of type I error. A separate analysis of OS was conducted for the Health Canada Common Technical Document (section 2.7.3) using RPSFT and IPCW methods to account for crossover from vorinostat to mogamulizumab. Results for all adjustment methods suggest that mogamulizumab was associated with improved OS; however, no information was provided on the methodology used for these tests or how the most suitable crossover adjustment method was chosen. Thus, the true benefit of mogamulizumab on OS is uncertain.
Subgroup analyses of the MAVORIC trial were pre-specified; however, they were not controlled for multiplicity. The CIs for most subgroup analyses of interest suggested precision; however, those subgroups with a lower number of patients had wider, more imprecise CIs.
HRQoL, as measured by the Skindex-29, FACT-G, and EQ-5D-3L, was a key secondary outcome of the MAVORIC trial. In a separate report, 2 additional analysis populations were defined: the cross-sectional and longitudinal-period populations, which were based on the MAVORIC ITT population. The results demonstrated improvements in HRQoL domains at various time points in the analysis; however, per the definitions of these populations, HRQoL results were presented only as observed data and were not presented for the entire ITT population. As such, these patients can be assumed to be responders to treatment, as nonresponders would be expected to drop out or cross over, which may bias the results. Moreover, the open-label design of the MAVORIC trial may have led to bias in the reporting of HRQoL outcomes, though it is uncertain.
External Validity
In discussion with the clinical experts consulted by CADTH, the inclusion and exclusion criteria for the MAVORIC trial were generally appropriate to enrol patients with a rare disease. There were, however, no Canadian sites in the MAVORIC trial. The MAVORIC trial enrolled patients with MF or SS (stages IB, IIA, IIB, III, or IV), which was considered by the CADTH review team and clinical experts to be quite broad, given the differences in disease behaviour and prognosis at each stage. The clinical experts also noted that the trial included a high proportion of patients with SS compared with the overall incidence of SS in Canada. Additionally, only patients with an ECOG Performance Status of 0 or 1 were enrolled, which may limit the generalizability of results to patients with an ECOG Performance Status of 2, considering the heterogeneity of disease at varying stages. Moreover, the clinical experts consulted by CADTH expect there would be more patients with an ECOG Performance Status of 2 in Canadian clinical practice. Ultimately, the clinical experts stated that patients with an ECOG Performance Status of 2 should be treated with mogamulizumab only at the discretion of the treating physician. The clinical experts also expressed that the cell counts for eligibility in the MAVORIC trial were lower than what they may expect in practice; however, they noted this is generally dependent on stage and many other prognostic factors.
The MAVORIC trial excluded patients with stage IA disease as well as those with current evidence of LCT. In clinical practice, it is unlikely that patients with stage IA would receive systemic therapy; thus, they would not be eligible for treatment with mogamulizumab. However, the experts did indicate that if there was evidence of LCT in patients with stage IA, they would be eligible for systemic therapy; however, they would not use mogamulizumab on this population. Therefore, no generalizations can be made to this population. However, protocol amendment 6 allowed patients treated with vorinostat who developed LCT to cross over to mogamulizumab. In clinical practice, it would be reasonable to consider mogamulizumab in a patient with LCT requiring 2 or more lines of systemic therapy. Regardless, no results were presented specifically for these patients; thus, generalizability remains uncertain.
The overall MAVORIC population was heavily pre-treated, with 29% of all enrolled patients having received 5 prior systemic regimens. Among the patients randomized to mogamulizumab, 30.1% had received 5 prior systemic therapies. Overall, the experts considered that the number of lines of therapy received in the trial was reflective of clinical practice; however, there are fewer options available in Canada, as the proportion of patients in each arm receiving various prior systemic therapies for CTCL was more reflective of a US-based treatment approach. The experts noted that the proportion of patients receiving ECP in Canada could be higher than in MAVORIC, and that methotrexate is used less frequently in Canada. Results of post hoc analyses suggest that the number or type of prior treatments do not impact the efficacy of mogamulizumab. The post hoc analyses evaluating PFS and ORR by treatment were relatively consistent with the primary analysis; however, the results varied based on the treatment received and thus could not be generalized.
Though approved by Health Canada for the treatment of CTCL, vorinostat is not widely accessible in Canada and is generally limited to patients with private insurance. As such, the comparative efficacy of mogamulizumab in patients with MF or SS in Canada may not be generalizable, as this is not a standard treatment in Canada. The patient groups noted that access to treatment is critical in this population in Canada. Although the clinical experts agreed vorinostat is a relevant comparator compared with mogamulizumab, they highlighted that response with mogamulizumab seems to be most notable in patients with blood involvement, and acknowledged that response to vorinostat, or other drugs that are used in SS that are also aimed at blood responses, is not as favourable.
As previously mentioned, most patients randomized to vorinostat crossed over to treatment with mogamulizumab. Given that the analyses for this population were purely descriptive, the results cannot be generalized with regard to treatment switching; however, as noted earlier, the disposition of patients and the results for PFS2 and OS accounting for crossover were consistent with the primary ITT analysis.
As previously mentioned, HRQoL was a key secondary end point of the MAVORIC trial, and improved QoL was of importance to patients. The results for change from baseline to 6 months were controlled for multiplicity; however, high attrition rates were observed for all HRQoL measures. Comparing the assessments at 6 months versus baseline, the completion rate was 73 versus 82 (42.2% versus 44.8%) for mogamulizumab-treated patients, and 118 versus 125 (66.3% versus 67.03%) for vorinostat-treated patients. This resulted in uncertainty in the results and may impact the generalizability of the results.
Indirect Evidence
|||||||||| ||| ||||||| ||| ||| ||||||| || |||||||| ||||||||
||| ||||||||| || |||| ||||||| || || ||||||| || ||||||||| ||| ||||||| || |||||||| |||||||| |||| ||| ||||||||||||||||| ||| ||||||||| ||||||||||||| || |||||||||||| |||||||||
| ||||||| |||||||||| |||||| ||| |||| ||||||| |||| || || || ||| ||| || ||||||| ||| |||||| | || ||||||| ||| ||||| || |||||| |||| ||||||| || ||| ||||||| ||| |||||||||| |||||| |||||||||| ||| ||||| ||||| ||| ||| |||| ||| ||||||||||| ||||||||| ||| ||||||||||||||||| |||| ||||| ||| |||| || |||||| ||| |||||||||||||||| |||||| ||| ||||||||| ||| |||||||||||
||||||||||| || |||||||| |||||||||||||
||| ||||||| ||||||||| | |||| ||||| || | |||||||||| |||||||||| |||||| |||| |||||||| ||| ||| || ||| ||||||| ||||| || ||| ||||||||||| || |||||||| ||||| ||||||| ||| ||||||||||| || |||||||| || ||| ||||||||| || ||||| ||||||||| ||| |||||||| || || |||
||||||| || ||||||||||||||||||| |||||||| ||||||||||
||||||||||
||| ||||||||| || ||| ||||||||||||||||| |||||| ||| || |||||| ||| ||||||||||| || |||||||||| |||| || ||||||| ||||||||||||| |||||| |||| |||| ||| ||||||| |||||| |||||| |||||||| ||||||||||| || |||||||| ||| ||| |||||||| || |||| ||| |||| |||| ||| |||||||||||
|| |||||||| ||||| ||| ||| |||||||||| ||||||| ||||||||||||| |||||||| |||||| ||| ||| ||| |||| || ||| ||||||| ||||| |||||||||||| ||||||| ||| ||||||||||| |||||| ||||||||||||| ||| ||||||||||||| ||||| |||| ||| ||||| |||||||||| |||| |||||||| || || ||| |||| ||||||||||| || ||| |||||||||||||||||| ||||||
||||| ||||||||| |||||||
|| ||||||| ||||||||||| |||||||||| ||| ||||||||| ||||| || ||| |||||||| || | |||||||| |||| ||||||||| ||| ||||||||| |||||||| ||| ||| ||||||||| ||| ||| |||||||||| || ||||| 31| |||||||| |||||||| ||||||| |||||||| |||||||||| ||| ||||||||||||||| ||||| || || ||| |||||||| |||||| |||||||||||| |||||||| || ||| ||||||||| || |||||||| |||| |||| ||| || |||| ||||||||| ||||||||||| ||| ||| |||| ||||||||| || ||| ||||||| ||||||||| || |||||||||||| ||||||||||| ||||||||||| ||||||||||||| ||| |||||||||||| ||||| ||| || |||||||| |||| ||| ||| |||||||||| ||||||| | ||||| ||||| || |||||||| |||| || |||||||| ||| |||| || |||| |||| ||| ||||||| |||||| ||| ||||||||||| ||| |||||||| || ||| ||||||||| ||||||||| ||| |||||| || ||||| ||||||||| || |||| ||||||||||| || ||||||| |||||||||| ||||||||| || ||| |||||||| ||||||||
|||||||| | ||||||||| |||||||| | ||||||||| |||||||| |
|---|---|---|
|||||||||| | |||||||||||| ||||||||| || |||||||| |||| ||| ||||| || |||| |||||||||| ||||| |||| ||||||| |||||||| || ||| ||||| ||||||||||||| ||||||||| ||| |||||||| || ||| || | • |||||||||||| ||| ||||||||| || ||||| |||||||| ||||||||| |||| || ||||||||||||| |||||||| |||||||| ||||||||| |||||| |||| ||| |||| |||| || ||||||||||||||| ||| || ||| || |
||||||||||||||||||||||| | |||||||||||| ||||||||| || ||||||||||||||| || |||||||||||| ||||||||||||| ||| ||| ||||||||| || |||||||| |||| ||||| ||||||||| ||| |||||||| || ||| ||| | • |||||||||||| ||| ||||||||| || ||||||||||||||| || |||||||||||| |||||||||||||| |||||||| ||||||||| |||| |||| ||||||||||||||| |
||||||| | |||||||||||| ||||||||| ||| ||||||||| |||||||| |||| |||| |||| |||||| ||||||||| ||||||||| |||||||||| ||||||||||| ||||||| ||||| ||||| ||||| ||| ||||||||| ||| ||| ||| |||||| |||||||| ||| |||||||| ||||||||||| |||| |||||||||| || |||||||||||| ||||||||||||| ||||||||| ||||||| ||||||||||| ||||| ||||||||||| |||||||| |||||||||||| ||||||||||||||| |||||||| ||||||| ||||||| ||||||||||| |||||||| |||||||||||| ||||||| ||||||||| ||||||||| |||||||| ||||||| | |||||||||||| ||||||||| ||| |||||||| ||| |||||| || ||| ||||||||| |||||||| |
||||| |||||| | |||||||| ||||||| ||||||||| |||||||||| ||| |||||||||||||| |||||||| |||||| ||| |||| ||||| |||||||||||| || ||| ||| |||||| |||||||||||||||| ||||||| | • |||||| |||||||| |||||| | ||||||| |||||||| ||||||||||||| ||||||||||| ||||||| |||||||||||||||||||||||| ||||||||||||||||||||||||||| ||||||||||||| ||||||| |||||||||||| ||||||| ||||||| ||||||| |
||| |||||||| ||||||||| |||| | ||||||||| |||||| ||||||||| ||| | |||||||| || ||||||||| |||||||| | ||||||||||| |||||| |||||| | |||||||||| |||||||||| || |||||| ||||||| | |||||||| ||| | |||||||| ||||||||| ||||||||||| || | ||||||| |||||||||| ||| | ||||| ||||||||| ||||||||||| |||| ||||||| |||||||||||||| ||| | ||||||| |||||||| ||||| || | ||||||| ||||||||| || | ||||||||||| |||||||| ||| | |||||||||||||||| ||||||||| || | ||||||| ||||||||| ||| | ||||||| || ||||| || | |||||| |||||||||
Note: Table redacted as per sponsor’s request.
||||||| ||||||| ||||||||| ||||||
|||||||| ||||||||||||||||||| ||||||||||
| ||||||||||| |||||||||| ||| ||||||||| ||||| || ||| ||||||| |||||||| || ||| ||| || ||||||||| ||||||| | ||| ||| |||||||| || |||||||||| ||| ||||||| |||||||||||| || |||||||| ||||||| ||| ||| |||||| ||||||||| |||||||| |||| || |||||||||||| ||||||||| || ||||||||| |||||||| || |||||||| |||||| ||| || |||||||||| |||||||| ||||||||| ||| ||||||||| |||||||| ||| ||||| ||||| ||||||||||||||| ||| ||| || ||||||| ||||||||||||| ||||||||||| || |||||||| ||| |||||||||| || ||||| 32| ||||||||| || ||| |||||||| ||| ||||||| ||| ||||||| |||||| |||| || |||||||| ||||| ||| ||| |||||||
||| ||||||| ||||| ||| ||| |||| ||||| |||| |||||||||||| |||| || ||| ||| |||| |||||||| |||||||| ||||||||| |||||||| |||| |||||||||||| |||||||| |||| || ||| ||| ||| ||||||| |||||||| |||| ||| ||| ||||||| ||||| |||| |||||||||||| |||||||| |||| ||| ||||||| |||| ||||||||| ||||| ||| ||| |||||||||||| || ||| ||||||| ||||||
|| ||| |||||||| || ||||||| |||||||| || ||| |||| || ||||||| |||||||||||| ||||||||||| || |||||||| ||||||||||||| ||||||||||| |||||||||| ||| |||||||||||||| |||||| ||| |||||||| ||||||| ||| |||| |||||||||| |||||||| ||||| |||| || ||||||| |||||||||| ||||||||||| |||||||||| || ||| ||||
|||||||| |||||||| || ||| |||| |||| ||||||| |||||||| || ||| |||| |||| |||| ||| ||| ||| ||| |||| |||||||||| || ||||||||| || ||| |||| | ||||| || | ||||||| |||||||| || ||| | ||||||| |||||||| || |||||||||||||||| |||||||| |||||| ||| ||| ||| ||| ||||| |||||||| || |||| |||| ||||||| ||||| || ||||||||| |||||||| ||||||| ||| || ||||| |||| |||||| |||||| || ||| ||||||| ||||||||| || ||| ||| ||||||| |||| |||||||| || |||||||| ||| ||||||| ||| ||| |||||| ||| ||| ||||||||| ||| ||||||| ||| ||||||| |||||| |||||||| ||| ||| ||| ||||||||| || ||| |||||||| ||||||||| || ||||| ||||||| ||| ||||||||||| |||| || |||||| ||| |||||| |||||||||||| |||||| ||| |||||||| |||||||| ||| ||||||| ||||| ||||||| ||| || ||| |||||||||| || |||||||| ||| |||| |||||||||| |||||||||| || || ||| ||||| || ||| |||||||||||||| |||||||||| || ||||||| ||||||||| ||||| ||| |||||| ||||||||| |||||||| ||||| ||||||||| ||||||||| || ||||| |||||| ||||| |||||| ||| |||||||| |||| ||| |||||||||||| ||||||||| || ||| || |||| ||||||||||| |||||||||||| ||| | ||||||| || | |||||| ||| ||||||| ||||| ||||||| |||| || ||| |||||||||| || |||||||| ||||||||| || ||||||||| |||||| |||||||| |||||| |||||| ||||||||| ||| |||||||||||| ||||||||||| ||||||| ||||| ||| ||||| || ||| |||| ||||||||| ||| || ||||| |||| ||||||| ||| ||||||||||| ||||| |||| ||||||||||| |||||| |||||||||| ||||| || |||||||| ||||||||| ||| ||| ||||||||| |||||||||| || ||||| |||||| |||||||| ||||| ||||||| |||||| |||| ||| ||||||||| ||||||| ||||||| |||||||||| |||||||| |||||||| |||||| |||||| |||| ||| ||||| |||||||||| ||||| ||||| |||||||| || ||| |||||||| |||||||| || |||||||||| ||| |||||||||||||| |||||||| ||||||||||||||| |||||| ||| |||||| |||||| |||||||| ||| || |||||||| ||||||||||| || |||| ||| ||||| |||| |||||||| ||| ||||||||||| ||||| |||| ||||| ||| ||||||| ||||| |||||||| ||| ||||||||||| ||| |||||| |||||| ||||||| ||| ||| ||||||| ||| ||| ||| ||||||||||||||||| ||| ||||||| |||||||| || ||| |||| ||| |||||| ||| ||| ||||||| || ||| || |||||| ||| |||||||||| || ||||| |||||||| || ||| ||||||| |||||| |||| ||| || ||||| ||| ||| |||||||||| || |||||||| |||| || |||| || | |||||| |||| ||||| || |||||| |||| ||||||||| ||| |||| ||||| ||||||||| || ||| ||||| || ||| ||||| ||| |||||||| || | ||||||| ||| |||||| |||| | |||||| || || |||||||
||||| | ||||||||| |||||||| | |||||| |||| | ||||||||| |||| | ||||| ||||||||| | ||||||||| |
|---|---|---|---|---|---|
||||||| | ||| ||| || ||||||||||||| |||||| |||||||||||||| ||||||||| || || ||| |||||| ||| |||| |||| ||| || ||| |||||| || ||||| ||| ||||| |||||| || |||||||| ||||||| |||| ||| | ||| | ||||||||||||| | ||||| ||| | |||| ||| |||| ||| | |||||||| ||| ||||||||| |||||| |||||| |
||| | |||||||||| ||||| || | ||||
||||||| | ||| ||| |||||||| |||| ||||||||||||| || ||| ||| |||||||| || ||||| ||| |||||||| |||||||| |||||||| || ||||||||||||| |||||| ||| ||| |||||||| || ||||| ||| |||||||| |||||||| ||||||| || |||||||||||| |||||||| ||| ||| |||||||||| || |||| |||||||| |||||||||||| |||||||||| ||||||||| |||| |||||||||| | ||| | ||||||||||| ||||||| ||| ||||| ||| | ||||| ||| |||| ||| | |||||||| ||||||||| |||||||||| |||| ||||||| |||| ||| |
||| | |||||||||||| |||| || || || |||||||||| |||||||| || | ||||
||||||||||| | ||| ||| |||| ||||| |||||| |||| |||| ||| |||||||||| |||||| || |||| || ||||| ||||| || |||| |||||| |||||||| ||| |||| ||| || || |||||||||| || |||||||| || ||| ||||||| ||| |||||||||| || |||||||| || ||| ||||||| | ||||||||| || |||||| |||||||| ||||||| ||||| || ||||| ||| || ||| ||||||||| |||||||||| ||||| |||||||||| ||| |||||||||||| || ||||| |||| |||| |||||||| |||| ||||||||||| ||||||| || ||| ||||||| ||||||||||||| | ||| | |||||||||| ||| ||||| || | ||| | |||||||| ||||||||| ||| ||| ||| |
|||| | |||||||||| ||| ||||| || | ||||
||| | |||||||||| |||| ||||| || | ||||
|||||| |||| | ||| |||| ||||||||| |||| |||||||||| ||| |||| | || |||| ||||||||| | ||| | ||||||||| ||||||||| |||| || ||||| ||| ||||||||||| ||||||||| |||| |||||| |||||| || || | ||| | |||||||||| | || ||| | |||||||||||| || || ||| | ||| ||| | |||||||| ||||||||| || |
||| | |||||||||| | || ||| | ||| |||||||||||||| |||| || ||||| ||| | ||||
||||||||||| | ||||||||| ||||| || || ||| ||| | ||| ||||| || |||| |||||||||| ||||| |||||||| | ||| | |||| ||||||| |||||||||| |||| ||| | ||| | |||||||| ||||||||| |||||| |
||| | |||| ||||||| |||||||||| |||| ||| | |||||||||| ||| ||||| || |
||| | ||||| | ||||| |||| | ||||||||| |||||| ||||||||| ||| | |||||||||||| ||||||||| |||| | ||||||| ||||||||||| |||||||| |||||| ||||| | |||||||| |||||||||||| ||| |||||||| ||| ||||||||| || ||||||| ||| | ||||| ||||||||| |||||| ||| | ||||||||| ||||||||||| ||||||| || | ||||||| |||||||||| || | ||||||| |||||| || | ||| ||||||||| ||| | ||||||| |||||||| ||||| || | ||||||| ||||||||| |||||| | ||||||| ||||||||| |||||||||| ||||| |||| ||||||||| ||| | |||||||||||||||| ||||||||| |||| | |||||||| |||| ||||||||||| | |||||||||||| ||| | ||||| | |||||| ||| | ||||| | |||||| || | |||| | |||| || | |||| | ||||| || | |||||| ||||||||| ||| | ||||| ||||| | ||||| ||||| | ||||| |||| |||||||| |||| |||||||| ||| | ||||||||||||||||| |||||||| || | ||||||||||||
Notes: Table redacted as per sponsor’s request.
Redacted rows have been deleted.
|||| |||||||
||||| || ||| ||||||||||| || |||||||| |||||||||| || ||| ||||||||||| ||||||||||| || ||||||||| |||||||| |||| |||| || || |||||| |||| |||||||||||||| || ||||| || |||||||||| ||||||||||||||||| |||||||| |||||||||| |||||| ||| |||| || || ||||||||||| |||||||||| |||||| || ||||||| |||| |||||||||| ||||||||| |||| |||||||||||||| || |||||||||| |||||||||| ||| |||||||||| ||||||| ||||||| ||| ||||||| |||||| ||| || |||||||||| |||||||| || ||| |||||||| |||||| ||||||||||| || || ||| |||| |||||||||| || ||||||||| || ||||||||| || ||||||||||||| ||| ||||||||||| ||| ||||||||||| |||||| |||| ||| ||||||| ||||| || ||| ||||||| |||||||||| || ||| |||| ||| ||||||||||||| ||||||| ||||| ||||||| |||| || || ||| |||||| ||||||||| |||||||| |||| ||||||||| || ||||||| ||| |||| || ||| ||| ||||||| || ||| ||||||| ||||| || |||| ||| ||| ||| ||| || |||||||||||||| || ||| ||||||| ||||| ||||| ||| ||||||||||| || ||||||||||| ||||||||| ||||| ||| ||||||||||| || ||| ||||||||| || ||||||||||| || ||||||| ||| ||||||| ||| |||| ||| |||||||| ||| ||||||||| ||||| ||| ||||||| ||||| |||| ||||||||||| ||||| |||| |||||||| |||| |||| |||||||| || ||| |||| ||||||||| |||| ||||||| ||| |||||||||| || |||||||| || ||| || | ||| ||||||||||| ||||||||| |||||| | || || | ||| |||| ||||| ||| ||| || |||| ||||||| || ||||||| ||||||||||||| || ||| |||||||||||||||| || ||| || | ||| ||||||||||| || ||||||||| |||||| |||||| ||| ||| ||| |||| ||||||| || ||||||| ||||||||||||| || ||| ||||||||||||||| || ||| || |||||||||| || ||||||||| |||||| |||||| ||| ||| ||| |||| ||||||| || ||||||| ||||||||||||| || ||| |||||||||||||||| ||| ||| ||||||||||||| || ||| ||||||| ||||| ||| ||| ||||||||| |||| ||| ||| ||||||||||| |||| |||| || |||||||| ||||| |||||||||||| ||||||| ||||| |||| |||| ||| ||||||||| ||| ||| ||||||||||| ||||||||||| || ||||||||||| |||||| ||||||||||||| || ||||||||||| |||| ||| ||||||| |||||| |||||||||||| |||||| ||| || ||| ||| ||| ||||||||||| ||| |||||||||||| || |||||||||| |||| ||||||||| ||||| ||||||| ||||| ||||||||| || |||||| |||||||||| ||||| ||| ||||||||| || |||||| ||||| |||||||||| |||| |||||||||| ||||||||| ||| |||||||||||||||| ||| |||| ||| ||||||| |||||| ||| ||| |||||||||||| |||||| ||| ||||||||||| ||| ||||||||||| |||||| || ||| ||||||| ||||| |||| |||| ||||||||| ||| ||| ||| ||||||||||| || ||| ||| |||||||| || ||||||| || |||| ||| |||||||| |||||| || ||| || ||| ||| ||||||||||| ||||| || |||| || ||||||||| ||||||| ||| ||| ||| |||||| |||| | |||| ||||||||||||| || ||| || ||| ||||||| || ||| ||||||| || ||| ||| ||| |||| |||||| ||||||| ||||||||||| ||||||| ||| ||| ||| || |||||||||||| || ||| ||||||| |||| ||| ||| |||||||||||| ||||| ||| ||| ||| | |||| ||||||||||||| || ||| ||| || ||| || ||||||||||| ||| ||| || |||| |||||| ||| ||| ||||||||||| ||| ||||| |||| ||| ||||||| |||||| ||||| |||| |||||||| || ||| ||||||| |||||||||||||||| ||| ||| |||| ||||||| ||| |||||||| || |||| ||| ||| || |||||||||| ||| |||| ||||||| || || ||||||||||||| ||| |||| || ||| ||||| ||| ||| ||| ||||||||||| ||| |||| || |||||||| |||||||||| || ||||||||||||| ||| ||| |||||||||||| |||||||||| ||||||||||||||||||||| |||||||| |||| |||||||| ||||||||||| ||||||| |||| |||||||| ||| ||||| ||||| || |||||||||||||||||||| |||||||| || |||| |||||||| || ||| |||||||||| ||||||||| ||||| |||||||||| ||| ||||||| |||||||| ||||||||||||||| |||| |||||||| ||| ||||||||||||| ||| ||| ||||||||||| |||||||| |||||||||| |||||||||| ||||| |||||||| |||||||||| ||||| ||| ||||||||| ||| |||| ||||||||||| ||| ||||| ||||||||| ||| |||| || ||||| |||||||| |||| ||| ||||||||||||| ||||| || ||| |||||||||| |||||| ||| ||||| | |||||| || ||||||| ||| |||||||| || ||||| | |||||||||| ||||| |||||||| |||||||||| ||||| || || ||||||||| ||||||| ||| ||| ||| ||||||||||| |||||||| |||||||||||| ||||||| ||| |||| |||||||| ||||||||| || ||| ||||||||| ||||||||| |||| |||||||| || ||| ||||||| |||||| |||||| || ||||||||||||| || |||||||| ||| ||||| ||||||||| |||||| ||||||||| ||| |||||||||||||||||||||||| ||||||||||||||| |||||||||||||||| |||| || |||| ||||| || ||||||||||||| |||| ||||||| ||||| | || ||||| |||||||||||| ||| |||||||||| || ||| |||||||| ||||| |||||||||||| ||| ||||||||| |||| ||| |||| ||| ||| ||| || |||||| |||||||| ||| |||||| ||||||||||||| || |||| ||||||| ||| ||||||| || ||| ||| || ||| ||||| ||||||| ||| |||||||||| || ||| ||||| |||||| || ||||||||| ||||| |||||| ||||||| |||| |||| |||||||| ||| ||| ||| || ||||||||| |||||||||| || ||||||||| ||||||| | |||||||| ||||||| || ||||||||| || |||||| || |||||||| |||||| || ||||||||| |||||||| |||| ||||| || |||||||||||||||| || ||| ||||||||||||||| ||| |||||||| ||||||||| ||| ||||||||| ||| ||| ||||||| ||| |||| |||||||| |||||||| |||||||| ||||||| ||| |||||||| ||||||||| |||||| |||| |||||||| ||||| ||||||||||| ||||| |||| |||||||||||| ||| ||||||| |||||||| |||||||| |||||||||||| |||||| |||| ||||||||| ||| || ||| |||| |||||| |||||| ||||||||| ||||||||||||| ||| ||| ||||||||||| |||||||| ||||||| |||| ||||||||| ||||| |||||||| ||| |||||||||||| ||||||| ||||||| || ||||||||| ||||||||| ||| |||| || ||||||| ||| ||| |||||||||||||| ||||||||||| || ||||||||| ||||||||||||| ||| ||||||||| ||||| ||| ||||||||| |||||||||| ||||||||| || ||| ||||||||||||| |||||| |||| ||||||| |||| ||||||||||| || |||||||||||| ||||||||||||| |||| ||||||||| |||||||| | ||| || ||||||| ||| ||||||| ||||| ||| |||||||||| ||||| |||||||| |||||||||| ||||| ||| ||| |||||| || |||||||||||| |||| ||||||||| ||||||| ||| ||||||||||||| ||| || |||||||| || || ||| ||||||||| ||||| ||||||||| ||| |||||||| |||||||| ||||| ||||||| || |||||| | |||||||||||| || ||| ||| ||||| ||| ||||| ||| |||||| ||||||||||| |||| |||| || |||||||| ||| |||||| || | ||| | |||||||||||| ||||||| ||| ||| |||||||||||| ||| ||| || ||||| || ||||||||| |||||||| ||| ||||||| ||| |||||||||||||| ||||| ||| | ||||| || | || | ||||||| ||| || ||||||| |||||||| ||||||| || || ||| || ||||| | |||||||| |||| || |||| |||||||| ||| |||||||||| ||||| ||||||| ||||||| ||| |||| ||||||||||| ||| | |||| ||||| ||| |||| |||| || ||||||| ||| |||||||| || |||| || ||||||||||||| || |||| || ||| ||||||||||| |||||||||| ||||||||| || ||| |||| ||||| |||| ||||||| ||||| | ||||||| |||||||| || |||| |||| ||| ||| ||||
||||||| |||||||| || |||||||| |||||||
|||||||| ||||| || ||| ||||||| || ||| ||||||||||| ||||||||||| | ||||||| |||| |||||||| || ||| ||||||| ||||||||| |||||||||| ||||| ||| ||||||| |||||| ||| ||| ||||||| ||||| ||||||| 4|| ||| ||||||| ||||| ||||||||| ||||||||||||| | ||||| ||||| | ||||| ||| |||||||||| ||| || |||||| ||||| ||| ||||||| ||||| |||||||| ||||||||||| ||||||| ||| ||||| ||||| | ||||| || |||||||||||| |||| || |||||| || |||||||||| ||| ||||| ||||||

||| ||||| ||||||||||| ||||||| || ||||||| |||||||| || ||| |||||||| |||||||||| |||| ||||||||| ||| ||||||| ||||| ||| ||| ||||||| ||||||
||| | ||||| ||| |||||| ||| | ||||| | |||||| || | |||| |||||| || | |||| |||||||
Note: Figure redacted at the sponsor’s request.
||| ||||||||| |||||||| ||| ||||||||| |||||||| ||||||||||||||| ||| ||||||| ||| ||||||| |||||| |||||||||| || ||||| 33| ||||| |||| ||||||| ||||||||||| ||||| || ||| ||||||||||||||||||| |||||||| ||| ||||||| ||| |||||||| |||||||||||| ||| ||||||||| ||||||| ||||||||||| || |||| |||||||| ||||| ||||||| |||||||| |||| || ||| || ||||||||| ||||| ||||||| |||||||| || ||| |||||| |||||||| ||| |||| ||| |||||||||||||| ||| ||||||| |||||| ||||| ||| ||||||| ||||| |||||||| |||||||| |||| |||||| ||| |||| |||| ||| ||| ||| ||||| ||| ||||||| ||||| |||||| || ||||| |||||||||||| || ||||| ||| |||||||| |||||||| |||| ||||| || |||||||| ||| ||||||| ||||| |||| ||||||||| |||| |||||||| ||| |||||||||| || |||| |||||||||||| ||| |||||||||| ||||||||| |||| ||||||||||| |||||||| || ||||||| |||| |||| ||||||| ||||||||||| |||| ||||| || ||||||| ||||||| ||||| || ||||| |||||||| | ||| ||| ||| ||||||| ||||| ||| ||| |||| ||||| |||| |||||||||||| |||| || ||| || |||||||||||| |||||||||| ||| |||||||| || ||||||| ||| || ||||| || || ||||| ||||||| |||||||||||||| |||| |||||| |||||||| ||||| ||||||||| ||||||| || |||||||| ||||| |||| || || |||||||| | ||| |||||||| ||| || || |||||| ||||||| ||||||||||||||| || |||||||| |||||||| ||| ||||||||||| || |||||||| ||||| ||| |||||||||| |||| ||||| || || |||||||||| ||| ||||||||| ||||| ||| |||||| || |||||||| |||| |||||||| ||||||| ||||||||| ||| ||||||| || ||||||| |||||||| |||| ||||||| |||| ||| |||||
|||||||||||||| | ||||||| | ||||||| |
|---|---|---|
||||||||||||||||||| |||||||| | ||
|||| ||||||| | || || || | || || |||||| |
||||||||||||| |||||| | |||| |||||| ||| |||||||| | ||||||||||||| |
||||||| ||||| | ||||| ||| |||| |||| ||| ||| || | || ||||| |||||||||||| | ||||||||| ||||| || || |||||||| |
||||| |||||| |||||| | || |||||||||||| || ||||| |||||||||||| | |||||||| |||| || ||||||| ||||| ||||| |||||| ||||||| |||| ||||||||| |
|||||||| | | ||||| | | ||||| |
||||||| | | |||||| | | |||||| |
|||||||| ||||||| | |||||| || ||||| ||| ||||| |||||| || |||||||| ||||||| | || | |||||||| || ||||| ||| |||||||| |||||||| ||||||||||||||| | || ||||| ||| |||||||| ||||||| || |||||||||||||||||||||| ||| |||||||||| || |||| |||||||| |||||||||||| ||| |||||||||| ||||||||| |||| ||||||||||| |
|||||||| ||||||||||||||| | ||
|||||| |||| | ||||||| | ||||||| |
||||||| | ||
|||||| ||||||| | || |||||||| | || ||||||| |
||||||| | ||
|||| | ||| ||||||| | || ||||| |
|||||| | ||| ||||||| | || ||||| |
|||| | ||||||||| |||||| ||||||||| |||| | ||||||| ||||||||||| |||||||| |||||| ||| | ||||||||||||| |||||| || | ||||||| |||||||||| || | ||| ||||||||| |||||| | ||||||| ||||||||| |||||||||| ||||| |||| ||||||||| || | ||||||||||| ||||||| || | |||||| |||||||||
| ||||||| ||||| || ||||||| || ||| || |||||||| ||||
Note: Table redacted as per sponsor’s request.
Redacted rows have been deleted.
|||| |||||||
|||||||| ||||| |||| |||||||||| ||||||||||| ||||||||||||| ||||||||||||| ||| || ||| ||||||| ||||| |||| ||| ||||||||||| ||| || ||| ||||||| ||||| || ||| ||| ||||||||||||||| ||||||||||||| ||| || ||| ||||||| ||||| |||| ||| ||||||||||| ||| || ||| ||||||| ||||| || ||| || ||||||||||||||| ||||||||||||| ||| || ||| ||||||| ||||| |||| ||| ||||||||||| ||||||| ||| || ||| ||||||| ||||| || ||| ||| ||||||||||| ||||| |||||||| || ||||| |||||||| ||||| || |||||||||||| |||||||||| ||| || ||| ||||||| ||||| |||| ||| ||||||||||| |||||| ||| || ||| ||||||| ||||| || ||| ||| ||||||||||||||| |||||||||| ||| || ||| ||||||| ||||| |||| ||| ||||||||||| |||||| ||| || ||| ||||||| ||||| || ||| || |||||||||||||||||||| ||||||||||||||| |||||| ||| ||||| |||||||| ||| ||| ||||| ||| |||||||||| || ||||| 34| ||| ||||||||| |||||| |||| || |||||| ||| ||||||| || ||| |||| ||| |||| ||||||||| ||| ||||| |||||||| |||||| |||| |||||||||| || |||||| |||||||||| |||||| |||||||||| |||| | | ||| |||||||||| |||| ||||| | || || |||||||| |||||||||| ||||| |||| ||||||||| ||| |||| |||||||||| || |||| ||| ||| |||||||||||| ||| || |||| ||||||||||| || ||| ||||||| ||| ||||||| ||||||| |||| ||||||||||||| ||| |||||||||| ||| || ||||||||| |||||| |||| || || |||| |||||||| || ||| |||||||||| ||| |||||||||||| ||||| ||| || |||||||||| ||| || ||||||||| |||||| |||| || || ||||| |||||||| || ||| |||||||||| ||| |||||||||||| ||| |||||||||| ||| |||||||||| ||| || ||||||||| |||||| |||| || || |||| |||||||| || ||| |||||||||| ||| ||||||||||| ||||||| ||||| ||| || |||||||||| ||| || ||||||||| |||||| |||| || || ||||| |||||||| || ||| |||||||||| ||| ||||||||||| ||||||||||| ||| ||| ||| ||| ||| || |||| || ||||||||||||| |||| |||||||| || ||||| || ||||||||||| ||||| ||| || |||| |||||||||| || |||||||| || ||| ||| |||||||||| ||| ||||||||||||| |||| ||| ||||||| |||||| ||| ||| ||| ||| ||| || |||| || |||||||||| |||| |||||||| || ||||||||||| ||||||| | |||||||||| ||| || ||||||| |||||||||| ||| ||||||||||| |||||| ||| ||| ||||||||| || |||| ||||||||| |||| |||| |||||||||| || ||||||||| |||||||||| |||| ||||| ||| || ||| ||| |||||||||||| ||| || |||||||||| ||| |||||| || || ||| |||| ||||||| ||||||| |||| ||| |||||||||| || ||||||||
|||||| ||| | ||||| | |||||| ||| | |||||||||| |||| | |||||||||| ||||| | |||||||||| |||| | | | |||||||||| ||||| | || || |
|---|---|---|---|---|---|---|
||||||||||||| | ||||||
||||||||||||| ||||| |||||| |||||||| | | | |||| | || | ||| | ||| | ||||| | ||| | ||||| | ||| | ||||| | ||| | ||||| | ||| |
||||||||||||| ||| ||||||||||| |||||| |||||||| | | | | | |||| | ||| | ||| | |||| | ||| | ||||| | ||| | ||||| | ||| | ||||| | ||| |
||||||||||||| ||||| |||||||| |||| ||| ||||||| ||||||||||| |||||||||| | ||| | || ||||| | ||| | ||| | ||||| | ||| | ||||| | ||| | ||||| | ||| | ||||| | ||| |
|||||||||| | ||||||
|||||||||| ||||| |||||| |||||||| | | | |||| | || | ||| | ||| | ||||| | ||| | ||||| | ||| | ||||| | ||| | ||||| | ||| |
|||||||||| ||| ||||||||||| |||||| |||||||| | | | ||| | || | ||| | ||| | ||| | ||| | |||| | ||| | ||||| | ||| | ||||| | ||| |
|||||||||| ||||| |||||||| |||| ||| ||||||| ||||||||||| |||||| |||||||||| | ||| | || ||||| | ||| | ||| | ||||| | ||| | ||||| | ||| | ||||| | ||| | ||||| | ||| |
|||| | ||||||| ||||||||||| |||||||| |||||| ||| | ||||||||| |||||| ||||| ||| | |||||||||||||||| || | ||||||| |||||||||| | | |||||| || |||||||||
Note: Table redacted as per sponsor’s request.
Source: Sponsor-submitted matching-adjusted indirect comparison.47
|||| ||||||| | |||||||||||||||| ||||||||
||||| 35 |||||||| ||| ||| ||| ||||||||||||| ||| |||||||||| ||| ||||||||||| ||| ||||||||||| |||||| ||||| ||||||| ||| |||||||||||| |||||| |||||| || ||| |||| ||||| || ||| |||||||| ||| |||| ||||||| ||||| ||| |||||||| ||| ||| ||||||||||||| ||| ||||||||||| |||| 2.21 (95% CI, 1.68 to 3.19) || ||| ||| ||||||||||| ||| 2.52 (95% CI, 1.78 to 3.75) || ||| || ||||||||||| || |||||| || ||||||||||| ||||||| 5|| ||||||||||||| |||| ||||||| || ||||| |||||||| ||||| || ||||||| ||| ||||||| |||| |||||||||| ||||||||| ||| |||| |||| ||| ||||| |||||| |||||| 6|| ||||||| ||| |||||||||| ||| ||||||||||| |||||| |||| ||||||| |||||| ||| ||||| ||||||||| |||||| ||||| ||| || |||||||||| ||||||| |||||||||| ||| ||||||||||| |||||| || |||||| ||| ||| || || ||||||||||| ||||||| 7||
||||||||| | |||||||||| | |||||||||| | |||||||||| || |||| ||| | |||||||| ||| |||| ||| |
|---|---|---|---|---|
||||||||||||| | ||||||||| | ||||||||||| | |||| |||||| ||||| | 2.21 (1.68 to 3.19) |
||||||||||||| | ||||||||| | ||||||||||| | |||| |||||| ||||| | 2.52 (1.78 to 3.75) |
||||||||||||| | ||| |||||| || |||||| || ||||| |||||||| ||||| || |||||||| || ||||||||| |||||||| || ||| |||||||| ||||||||| | ||||||||||| | |||| |||||| ||||| | |||| |||||| ||||| |
|||||||||| | ||||||||| | |||||||||| |||||| | |||| |||||| ||||| | ||| |||||| ||||| |
|||||||||| | ||||||||| | |||||||||| |||||| | |||| |||||| ||||| | |||| |||||| ||||| |
||| |||||||||| ||||||||| || | |||||| |||||| ||| | |||||||||||||||| || | ||||||| ||||||||||
Note: Table redacted as per sponsor’s request.
Source: Sponsor-submitted matching-adjusted indirect comparison.47

||| ||||| |||||||||||| |||||| ||| ||| |||| ||||| |||||||| ||||| ||| ||| |||||||| ||||||||||||| ||| |||| ||| ||| ||||||||||| ||| || |||| ||| ||| ||| || |||| ||||||||||| |||| |||||| |||||||||| || ||||||||||||| | |||||||
|| |||||||| ||| |||||||| |||||||||||| |||||| ||| ||| ||||| ||| ||| ||||||||||| |||| ||| ||||||| ||| ||||||| ||||||| || |||||||| ||| |||||||| |||||||||||| |||||| ||| ||| ||||| ||| || ||||||||||| |||| |||||||||| ||| ||||||| |||||||
Note: Figure redacted at the sponsor’s request.
||||||| ||||||| ||||||||| ||||||

||| ||||| |||||||||||| |||||| ||| ||| |||| ||||||| || ||||| |||||||| ||||| || ||||||| ||| |||||||||| |||| ||| ||||||| |||||||| || ||| ||| ||| || ||||||||||| |||| |||||||||| || |||||| || ||||||||||||| | |||||||
Note: Figure redacted at the sponsor’s request.
||||||| ||||||| ||||||||| ||||||

||| ||||| |||||||||||| |||||| ||| ||| || ||| ||| |||||||||| ||||| |||||||||| ||||||||||| |||| |||||||||| ||||||| || ||||||| |||||| |||| ||||||||||| |||||| ||||||| | ||| || ||||||| |||| ||| |||||| ||||| ||||||||| || ||||||||||| |||||| ||| ||||| ||||| |||| ||||||| || ||| || ||||||||||| ||| |||||| || ||||| |||||||||| ||| |||||| || ||||| || |||||||| ||| |||||| || ||||||| | ||| || |||||||
|| |||||||| ||| |||||||| |||||||||||| |||||| ||| ||| ||||| ||| ||| ||||||||||| |||| ||| ||||||| ||| ||||||| ||||||| || |||||||| ||| |||||||| |||||||||||| |||||| ||| ||| ||||| ||| || ||||||||||| |||| ||| ||||||| ||| ||||||| |||||||
Note: Figure redacted at the sponsor’s request.
||||||| ||||||| ||||||||| ||||||
|||| ||||||| | ||||||| ||||||||
||||| 36 |||||||| ||| ||| ||| ||||||||||||| ||| |||||||||| ||| ||||||||||| ||| ||||||||||| |||||| ||||| ||||||| ||| |||||||||||| |||||| |||||| || || ||||| ||| ||| ||||| ||| || |||||||||| ||||||| ||||||||||||| ||| ||||||||||| || |||| ||| ||| ||||||||| ||| 0.90; 95% CI, 0.62 to 1.27 || || ||||||||| ||| 0.79; 95% CI, 0.45 to 1.18 |||||||||||| |||||| ||| ||||| |||||||||| ||||||| 8|| ||||| ||| |||| || |||||||||| || || ||||||| ||||||||||||| ||| ||||||||||| |||| ||||||| || |||||| || ||||| |||||||| ||||| || ||||||| ||||||| 9||
||||||||| | |||||||||| | |||||||||| | |||||||||| || |||| ||| | |||||||| ||| |||| ||| |
|---|---|---|---|---|
||||||||||||| | ||||||||| | ||||||||||| | |||| |||||| ||||| | 0.90 (0.62 to 1.27) |
||||||||||||| | |||||||| | ||||||||||| | |||| |||||| ||||| | 0.79 (0.45 to 1.18) |
||||||||||||| | ||| |||||| || |||||| || ||||| |||||||| |||||| | ||||||||||| | |||| |||||| ||||| | |||| |||||| ||||| |
||| |||||||||| ||||||||| || | |||||| |||||| ||| | |||||||||||||||| || | ||||||| ||||||||||| ||||| |||||||||||||||||||||| ||||||| ||||||||| ||||||
Note: Table redacted as per sponsor’s request.

||| ||||| |||||||||||| |||||| || || || ||| ||| ||| || ||||||||||| |||||||||||| || |||||||||| ||||||| ||||||||||||| ||| |||||||||||| |||||||| ||||| |||| ||||||| ||| ||| |||||||| ||||||||||||| ||| ||| ||||||||||| ||||||| ||| ||| || ||||||||||| ||||||||||| ||||| |||||||| |||||| ||||| ||| || ||||||| ||||||||| || ||||||||||| ||||||| ||| |||||| || ||||||||||||| || |||||||
|| |||||||| ||| |||||||| |||||||||||| |||||| ||| || ||||| ||| ||| ||||||||||| |||| ||| ||||||| ||| ||||||| ||||||| || |||||||| ||| |||||||| |||||||||||| |||||| ||| || ||||| ||| || ||||||||||| |||| ||| ||||||| ||| ||||||| |||||||
Note: Figure redacted at the sponsor’s request.
Source: Sponsor-submitted matching-adjusted indirect comparison.47

||| ||||| |||||||||||| |||||| ||| || ||| ||||||||||||| ||| ||||||||||| |||| |||||||| || ||||| |||||||| ||||| || ||||||| ||| |||||||| || |||||||||| ||||||| |||||||| ||||| ||| ||| |||||||| ||||||||||||| ||| ||||||||||| |||||| ||| |||||| |||| || ||||| |||||||||| || ||||||
Note: Figure redacted at the sponsor’s request.
||||||| ||||||| ||||||||| ||||||
|||| ||||||| | |||||||| || ||||
|||||||| ||||| ||| ||||||||||| ||| ||||||||||| |||||| |||| ||||||||| ||| ||| || ||||||||||| ||| ||| |||||||| ||||||||||||||| |||| ||| ||||||||| ||| |||| ||||||||||| |||||||||| || |||||||| |||||||||| || ||| || ||||||||||| ||||| ||| || |||||||||| ||||| 37 |||||||| ||| |||| |||||| ||| ||| ||||||| |||| |||||||| || ||||||||||||| ||| |||||||||| ||| ||||||||||| ||| ||||||||||| ||||||| ||||||||||||| |||| |||||| || |||||||| |||| ||||||||||| ||| | |||||| |||||||| || |||| |||| ||||||||||||| || ||||||||||| || ||| ||| ||||||||||| ||||||||||||| ||| ||| |||||||| |||| ||||||||||| || |||| |||||||| |||||| |||| |||| |||| ||| ||||| ||||| ||| ||||| |||||||||| |||| |||| |||| ||| ||||| |||||| || ||| || |||| ||||||||||| ||||||| |||| |||||||||| |||| ||||||||||||| ||| |||||||| |||| ||||||||||| || |||| |||||||| |||||| |||| |||| |||| ||| ||||| |||||| ||| ||||| |||||||| |||| |||| |||| ||| ||||| ||||||| ||||| ||| || |||||||||| || |||| |||||||| ||||||| |||||||||| ||| ||||||||||| |||||| || |||||| ||| ||| || || ||||||||||| |||||| || ||||| |||||||||||
||||||||| | |||||||||| | |||||||||| | |||||||||| |||| ||||| |||| ||| | |||||||| |||| |||||| |||| ||| |
|---|---|---|---|---|
||||||||||||| | |||||| | ||||||||||| | |||| |||||| ||||| | |||| |||||| ||||| |
||||||||||||| | ||||| | ||||||||||| | |||| |||||| ||||| | |||| |||||| ||||| |
|||||||||| | |||||| | |||||||||| |||||| | |||| |||||| ||||| | |||| |||||| ||||| |
|||||||||| | ||||| | |||||||||| |||||| | |||| |||||| ||||| | |||| |||||| ||||| |
||| |||||||||| ||||||||| || | |||||| |||||| ||| | |||||||||||||||| || | ||||||| ||||||||||| ||||| ||||||||||||||
Note: Table redacted as per sponsor’s request.
||||||| ||||||| ||||||||| ||||||
|||||||| ||||||||| || ||| ||||||||||||||||| ||||
||| ||||||||||| |||||||||| ||| |||||||||| |||| |||| |||||||| || | |||| |||||||| || ||||||||||| ||| |||||||| || ||| ||| ||||||| |||| ||||||| || ||| ||||||||| ||||||||| ||| |||||| || ||||| ||||||||| || |||| |||||||||| |||||| ||||||||| ||||||||||| || ||||||| ||||||||||| |||| ||||| || |||||| || ||||||| || ||||||| ||||||||||| ||||||| |||| ||||| || |||||||| ||||||| ||| ||| ||||||||||| ||||||||||| ||| ||||||||||| |||||||||| ||| ||||||||| || ||| ||||| || ||| ||||||| |||||||||||| ||||||| ||| ||||||||||| || ||||||||| || |||| || ||| ||||| ||||||||||||||||||| |||||||| |||||| |||||||||||| ||||||||||| ||| ||||||| |||||||||||||||| ||| ||||| |||||||||| |||| ||||||| ||| ||||||||| || ||||||| |||| ||||||||| |||||| ||| || ||||||||||| || ||||||||||| ||| |||||||| |||||||||| |||||||| || || ||||||||| ||||||| ||| ||||||||| ||| ||||||||||| ||||| |||| | |||||| ||| ||| ||||||||| ||| ||| |||||||| ||||||| ||| ||||||| ||||| |||| ||| ||||| ||||||| ||||| ||| |||||||| || || ||| ||| ||||| ||| |||||||||| || || ||| || |||||||| || ||| |||||| ||||||| ||||| || ||| ||||||||| ||||||||| || |||||||| |||| ||| |||||||| |||| ||||||||| |||| |||||||||| ||| ||| |||| ||||| || |||||||| ||| ||| |||| ||||| || ||| ||||||||||| ||||||| |||| |||||||| ||| |||||||| || ||||| ||||||||||| || ||||| |||| ||| ||||||| ||| ||||||| |||||| |||| |||||||| ||| |||||||||| ||| ||| |||| ||| ||| || ||| ||||||| |||||||||| ||| ||||||||||| ||||| |||||| ||| |||||||| ||||||||||| |||||||||| |||||| ||| | ||||| ||||| |||||||| || |||||||| ||||||||| |||||| ||| ||||| |||| ||| ||||||||| || ||| ||||| |||| |||||| || ||||||| || |||| ||||||| ||||||| ||| ||||||| ||| ||||||||| ||||||||||| ||| |||| || |||||| ||||||||||| ||||| ||||| |||||||| |||| ||| ||||||||||| ||||||| ||| ||||||| ||| ||||||| |||||| |||| |||||| ||| ||||||||||||| || ||||||||||| |||||| ||||| |||||||| |||||||||||| ||| ||||||||| |||||||| || ||| ||||||| |||||| ||| |||||||| |||||||||||| ||| |||||||| |||||||||| || |||| || ||||||||||| || ||||||| |||||||| ||||||||||||||| |||| || |||| ||||||| ||||||| |||||| ||||| |||||||||||| ||||| ||||||||||| ||| |||||||||| || ||| |||||| ||||| |||||||||| | ||||| ||| ||||||||| |||||||| ||| ||| ||||| ||||| |||||| || ||| |||| || ||||||| |||| ||| |||||||||| |||||| || |||||||| ||| ||||||||| |||||||| || ||| ||||||| ||| ||||||| |||||| |||| |||||||||| |||||||| |||||||| ||| || ||| |||| ||||||||||| ||||||| ||||||| ||| ||||||| ||| ||| ||||||||| |||||||| ||||| ||| ||||||| ||||| |||||||| |||||||| |||| |||||||||||||| ||||||||| |||||| ||||| ||| ||||||| ||||| |||||||| |||||||| |||| ||||||||||||| || || ||||||| ||| || |||||||| |||| ||| |||| ||||||||||||| ||| ||||||||| ||| || ||| |||| || |||||||||| ||||||| |||||||| |||| ||| ||| |||||||||| || |||||||| || |||| || ||| || |||| |||||||||| || ||||||| || ||| ||||||| ||||||||||| |||||||| || ||| |||||||| |||||| ||||| || ||| ||||||||||||| ||| ||| || |||| ||||||||||| || ||| ||||||| ||||| ||||| |||||||| |||| ||| ||||||| ||||||||||| ||||||||||| ||| ||||||||||||| ||| ||| ||||||| || |||||| ||| |||||| ||||||||||||| ||| ||| |||||||||| || |||||||||| || ||||||||||| ||||||| ||| ||||||||||||| ||| ||| ||||||| || ||||||| ||| |||||| |||| ||| ||||||| ||| ||| || |||| |||||||||||| ||||||||||||| ||| |||||||||| || ||| ||| |||||||||| ||| ||| ||||||| |||||||||| || |||| |||||||||||| ||| ||||||||||| || ||| ||||||| ||||||||||| || ||| |||||||| |||||||| || || ||||||||| ||| |||| || |||| ||||||||| || ||| || ||||||||| || |||||||| ||||| || |||||||||| ||||||||||| || ||| |||| || ||||||||| ||| || ||| ||||||||| |||||||| ||||| ||||| ||| |||||| |||||||||||| ||||||||||||| ||||||| ||||||| ||||| ||| ||||||||| |||||||| || ||| ||||||||| |||||||| || ||| ||||||||||||||||||| |||||||| |||||||| ||||||| ||||||| ||| |||||||| ||||| || || ||||||||| |||||||||| || ||| |||||||| ||||||||| |||||| |||||||||| || ||| ||||||| || |||| ||||||||| ||| |||| ||| ||||||||| ||| |||||| || |||| || || |||||||||| |||| |||||| || || |||||||||||| |||||||||||| ||| ||||||| |||| ||| ||||||||||| ||| ||| || |||| |||||||||| ||||||||| ||||||| |||| ||| ||||||| ||| |||||| || |||||| || ||||||||||| ||||| |||| ||| || |||| |||||||||| || ||| | |||| |||||||||| || ||||||||||||| |||||||| || || |||| ||| ||||| ||||||| ||| |||||| |||||||||| ||| |||||||||||||| |||||||| ||||| ||||| ||||| |||| |||||| |||| || ||| |||||||| |||| ||| ||||||||||||| ||| || ||| ||||||| ||||| |||| |||||||| |||||| |||| |||| ||| ||||||||| || ||||||||| |||| || |||||||| ||| ||| ||||||| ||| ||| || ||||||||||||| || ||| |||||||| ||||| ||||||||||| |||| ||||||||||||||||| |||||| |||||||| | ||||||||||| || ||| ||||||||||||||| || ||| |||||||| |||||||| || |||| || ||||||||||| || |||||||| |||||| |||||| |||||| || ||| ||| ||||||||| || ||| ||||||| ||||| ||||| |||||| ||| |||||||| ||||||| ||||||||||||||| |||| |||||||||| ||||||| || |||||||||||||| ||| |||||||| |||||||| || ||| ||||||| ||| ||||||| |||||| |||| |||||||| ||||||| ||||| |||| |||||| ||||||||||| || ||| |||||||| || ||||||||||| || |||||||||| |||||||||| ||| |||||| |||||||| |||||| || || ||| ||||||| ||||| ||| |||| |||||| |||||||| || |||||| || |||||| || |||||||| ||||||||||| || ||||| |||||| |||||| || |||||||| ||| ||| ||| |||||||||| | |||||||||| || ||| |||| ||| ||| |||||| ||| |||||||||||||||| ||| |||||||||||||||| || ||| |||||||| ||||| |||| ||| |||||||| ||||||||||| ||||| |||| |||| ||||||||||| || ||| |||||||| ||||||||||||||| || ||| ||||||| ||| ||||||| |||||| ||||||||| ||||||||||| || ||| |||||||| || ||| |||| |||| ||| ||||| ||| |||||| || ||| ||| ||||||| ||||||| ||||| |||||| ||||| ||||| ||| |||| ||||| ||||||| ||||| ||| ||||| ||| ||||| |||||||||||| ||||| ||||| |||| || |||||||||||| || |||||||| ||| |||||||| |||| || ||||||| |||| |||||||| || |||||||| || ||| ||||||||| |||| ||||||||| |||| ||| |||||| |||||||| |||||||| || ||||||||| |||||| ||||||||||||| ||||||| || |||| ||||||| |||||||| || |||||||| |||| ||||| |||||||||||| |||||| ||| ||||| || ||| |||| |||||| ||| |||||||||| | ||||| ||| ||||||||||||| || |||||||| ||||||||||||||| ||| |||||||| |||| |||| |||||||||| ||| ||| ||||| || ||||| || || || |||||||| |||||||| ||| ||||||||||| |||||||| ||||| || ||||||||| ||| ||||| |||||||| |||| |||||| || ||||||||||||||| || ||| |||| |||||||| ||||| |||||||| |||||||| |||| |||||||||| ||| ||||||||| |||| |||| || ||| ||||||| |||||| || || || |||||||| |||| ||||||||| ||| ||||||||||||||| ||| ||| |||||||| || |||||||| |||||||||| ||| ||||||||||| || ||| ||||||||| ||| |||| |||| |||||||| || ||||||| | ||||| |||||||||| ||||||| |||||||||| || ||| ||||||| ||||| ||| |||||||||| |||||| |||||||||| || |||||||||||| || |||||||||| || ||| ||||||| |||||| ||| ||||||| || ||||| ||||||||||| ||||||||| |||| ||||| ||| || |||||||||| || |||||||| ||| |||| || |||||||| ||||||| |||||||||| ||| ||||||||||| ||||||| |||||||| || || ||||||||| ||||||| |||| |||||||||| ||||| ||||| || |||| |||||||||||| ||| ||||||||||| || |||| || |||||||||| ||| |||||| |||| || |||||||| |||||||| |||||||| ||||||||| || ||| |||||||| ||||||| ||||||||| || |||||| |||||||||| ||||||| |||||||| ||| |||||||||||||||| || ||||| |||||||| |||||||| ||| |||||||||| || ||| ||||||||||||||||| ||||| ||||| || | |||||||||| |||||||| || ||| |||||||||| |||||| || |||| || ||||||| |||| ||| |||||| ||||||||| ||| |||||||||| ||||||| ||| ||||||||| ||| || ||| |||||| |||| |||||||||| || ||||||| |||||||||| |||||||||| || |||| ||||||||| || ||| |||| ||| ||||||||| |||||||| |||||| || ||| ||||||| ||| ||||||||||||||||||| |||||||| |||||||||||| | |||| || ||| ||||||||| ||| ||||||||| ||| |||||||| ||||||||| |||| |||||||||| ||||| |||||||||| |||||| |||| |||||| | || ||| ||||| | || || |||||||| ||| |||||||| || ||||| ||||| || |||||||| ||||||| ||| |||| |||||||| || |||| || | |||||||| ||||||||| |||||| ||| ||||||| ||||||||| || ||||| ||||| ||||| || ||||||||| |||||||||| |||||||||||||||||||||||| |||||||||| || ||||||||||||| || |||||||| ||| ||| ||||||||| || ||||| ||||||||| ||| ||||||||| ||| || || ||||||||| ||||||| ||| ||| ||||||| |||| |||||||| || |||||||||| |||| ||| |||| || |||| || ||| |||||||| ||||||||| ||||||| ||| |||||||||| ||||| || |||||||||| ||||||||||| ||| |||| || ||||||||||| ||||||||| ||| ||||||| || ||| |||| ||||||| |||| ||||||||||| ||| |||||||| |||| ||||||||||||| ||| |||| |||||| ||| ||||| ||||||||||| ||||| ||||| ||| || |||||||||| ||||||| |||||||||| ||| ||||||||||| ||||||| ||||||| ||||| || ||||||||||| || ||| ||||||| ||| || ||| |||| |||| ||| ||| || |||||||||| ||| ||||| ||||||| ||| ||||||||||| |||||||| ||| |||||| |||||| || || ||| ||||||| ||||| ||| |||||||||||| || ||||||||| |||||| ||| |||| ||| ||||||| ||| |||| |||||||||| ||||||| ||| |||||||| || |||| |||| |||||||| ||||||||||| |||||||| || ||||||||||||| || |||| ||| ||| ||| || |||| |||||||||||| |||| |||||| ||| ||||| ||||||||||| ||||| ||||| ||| || |||||||||| ||||||| |||||||||| ||| ||||||||||| ||||||| |||||| ||| ||||||| ||||||||||| ||||||| ||||||| ||| ||||||| || ||||||||||||||||||| ||||||||| ||| ||||||||| |||||||||| ||| ||| ||||||||||| |||||||| || ||| ||||||||| ||| |||||||| |||||||||||||||| ||| ||||||||||| || ||||| ||||||| ||| ||| ||||| ||||||||| || |||| ||| ||||||| || ||| |||| ||| |||||||||| ||||||||| ||| ||| ||| || ||||||||||||||
Other Relevant Evidence
No long-term extension studies or other relevant studies were included in the sponsor’s submission to CADTH.
Discussion
Summary of Available Evidence
One phase III, open-label RCT, MAVORIC, was included in this review. The MAVORIC trial consisted of 372 patients with MF or SS randomized to receive mogamulizumab (n = 186) 1.0 mg/kg IV infusion on days 1, 8, 15, and 22 of the first cycle and on days 1 and 15 of subsequent cycles, or vorinostat (n = 186) 400 mg orally once daily on day 1 of each 28-day cycle until disease progression or unacceptable toxicity. The primary end point of the MAVORIC trial was PFS, with key secondary end points of ORR and HRQoL. OS was an exploratory end point of the MAVORIC trial.
Baseline characteristics in the MAVORIC trial were well balanced between the mogamulizumab and vorinostat groups, with 56.5% and 53.2% of patients in the mogamulizumab and vorinostat arms having MF, and 43.5% and 46.8% of patients in the mogamulizumab and vorinostat arms having a diagnosis of SS. The median age of all patients was 64 years, 58.1% were male, and 69.9% were White. The population in the MAVORIC trial was heavily pre-treated at baseline, with a median of 3 prior systemic therapies received in each treatment arm. The median follow-up of the MAVORIC trial was 17.0 months for efficacy, and 34.5 months for safety.
Interpretation of Results
Efficacy
The primary efficacy end point of the MAVORIC trial was PFS which is, according to the ISCL, United States Cutaneous Lymphoma Consortium, and the Cutaneous Lymphoma Task Force of the EORTC, a particularly useful primary end point in MF and SS and was considered by the clinical experts consulted by CADTH to be clinically relevant for this study. Although the median PFS was relatively short (7.70 months versus 3.10 months), mogamulizumab was associated with a significant improvement in PFS (HR = 0.53; 95% CI, 0.41 to 0.69; P < 0.0001), and was also considered clinically relevant by the clinical experts, given that MF and SS are incurable and given the younger age of the population with these diseases. The clinical experts considered the median PFS with vorinostat to be lower than expected, as PFS for chemotherapy is generally in the range of 4 months. Sensitivity analyses varying the definition of PFS were consistent with the primary analysis. Interestingly, the median PFS for patients who crossed over from vorinostat to mogamulizumab was greater than the results of the primary analysis (8.43 months). Results of the subgroup analyses for PFS were generally consistent with the primary analysis. There was a noticeably greater PFS benefit observed in patients with SS for mogamulizumab over vorinostat (13.30 versus 3.13 months). In patients with stage III or IV disease, the results were similar, favouring the PFS benefit with mogamulizumab (10.90 versus 3.00 months). The results were consistent in the 2 post hoc analyses for PFS in patients with advanced-stage MF or SS (stages IIB, IIIA, IIIB, IVA1, IVA2, and IVB) and patients with blood involvement (B1 and B2). It should be noted that these results were for the post hoc analyses and were not pre-specified. In the subgroup of patients with B1 disease, there were only 62 patients overall. As such, the results of the post hoc analyses should be interpreted with caution, considering the limitations. The clinical experts consulted by CADTH hypothesized that though mogamulizumab provides a survival benefit in patients with MF or SS, overall, it appears to provide greater PFS benefit in patients with more advanced stages of disease, given the strikingly improved PFS results.
The results for the key secondary end point of ORR were congruent with the findings of PFS, where the ORR for mogamulizumab is significantly greater than for vorinostat (28.0% versus 4.8%; P < 0.0001). The differences in response rates for mogamulizumab observed in MAVORIC were considered by the experts to be clinically meaningful. The majority of patients with MF or SS who respond to treatment achieve a PR, not a CR, as evidenced by only 4 (2.2%) confirmed CRs versus 47 (25.3%) confirmed PRs with mogamulizumab. Interestingly, more patients had stable disease with vorinostat compared with mogamulizumab. The clinical experts highlighted that in MF and SS, other than stable disease, a PR is most often observed with current treatments, which is in line with the results for mogamulizumab. Again, multiple subgroup and post hoc analyses demonstrated an improved response in patients with advanced-stage MF or SS and blood involvement. In patients with SS, the ORR with mogamulizumab was 37.0% compared with 2.3% for vorinostat. Conversely, for patients with MF, the ORR was 21.0% versus 7.1%. For patients with blood involvement, the proportion of patients achieving an ORR was also greater for mogamulizumab with a greater extent of blood involvement than with vorinostat (B0 = 15.6% versus 6.5%, B1 = 25.8% versus 6.5%, and B2 = 37.4% versus 3.2%, respectively). Furthermore, when measured in each compartment, mogamulizumab demonstrated a higher ORR in all cases; however, the greatest risk differences were observed in the blood compartment, where the ORR for mogamulizumab was 66.9% compared with 18.4% for vorinostat, followed by the skin compartment (41.9% versus 15.6%). The clinical experts noted that the response in the blood is aligned with the response in the skin, which is clinically impactful for these patients. The clinical experts consulted by CADTH and the clinician and patient groups all emphasized that any improvement in skin involvement is welcome in these patients and is considered an important outcome. Consistent with these findings was the median DOR of 14.07 months for mogamulizumab, which the clinical experts considered to be favourable for mogamulizumab at 14.07 months. DOR was also markedly higher for mogamulizumab in the blood (25.47 months) and skin (20.57 months), supporting the results for PFS and ORR.
The range of disease stages included in the MAVORIC trial was considered by the CADTH review team and the consulted clinical experts to be quite broad, with the experts highlighting that the prognostic and clinical differences at various stages would impact the generalizability of the findings and the interpretation of the results. However, the results of the specific subgroup and post hoc analyses provide some insight into the efficacy of mogamulizumab across disease stages. The clinical experts highlighted that patients with SS have blood involvement labelled as B2, hence, the similar results for PFS and ORR in these subgroups were favourable. Though the results of these subgroup and post hoc analyses for PFS and ORR were not controlled for multiple testing or powered to detect differences, the clinical experts speculated that mogamulizumab shows the most promise in patients with later-stage disease and in patients with SS or blood involvement, given that most other current therapies are ineffective for blood involvement.
The other key secondary end point of MAVORIC was HRQoL, as assessed by the Skindex-29, FACT-G, and EQ-5D-3L. The clinical experts noted that the Skindex-29 is routinely used in their clinical practices for cutaneous lymphomas but may not be widely used in other provinces. Improvement in QoL was listed as an important aspect of treatment for patients. The results for all HRQoL measures were generally consistent, with improvements in QoL for both mogamulizumab and vorinostat; however, mogamulizumab was generally associated with significant improvements from baseline across several of the evaluated domains, which may reflect the open-label bias. The greatest change from baseline for mogamulizumab was noted in the symptom scale score of the Skindex-29; as previously noted, improvement in skin symptoms is of critical importance to patients. Furthermore, the clinical experts considered this improvement in skin symptoms from baseline to be clinically meaningful. The improvement in the FACT-G total score and the VAS for the EQ-5D-3L was also greater for mogamulizumab than for vorinostat. For the Pruritis Likert scale at baseline, patients reported scores of 6.2 and 6.3 for mogamulizumab and vorinostat, respectively. Though there was no discernable difference in scores between mogamulizumab and vorinostat from baseline to cycle 5, it is uncertain what these scores represent clinically in terms of itch, and how these scores correlate with those from the Skindex-29 tool. Overall, there remains uncertainty in the effect of mogamulizumab on HRQoL due to the high level of attrition between baseline and 6 months for all HRQoL measures.
OS was an exploratory end point of the MAVORIC trial. The median OS was not reached in the mogamulizumab group with 17.0 months follow-up for efficacy. The results of this end point were considered immature due to the short length of follow-up for this disease. Moreover, improved survival is important to patients. OS is generally not considered an appropriate or feasible outcome in MF and SS trials, as the expected survival is far longer than the course of the study and the potential exists for survival to be impacted by treatment(s) given after the conclusion of the study trial.31 OS was exploratory, and the results of this analysis are uncertain due to the limited follow-up time and confounded by the 1-way crossover; thus, the results should be interpreted with caution.
In the absence of comparative evidence, the sponsor submitted an MAIC that compared the mogamulizumab and vorinostat arms of the MAVORIC trial with relevant comparators for PFS, OS, and skin response in the treatment of MF and SS. The analysis was informed by a systematic literature review that identified evidence from 39 publications detailing 26 different studies: 14 parallel trials and 12 single-arm studies. In total, 2 trials, MAVORIC and ALCANZA, were included in the MAIC, comparing patients with MF or SS from MAVORIC with those with primary cutaneous anaplastic large-cell lymphoma in ALCANZA, given that brentuximab vedotin and physician’s choice treatments were expected to be the main comparators in the cost-effectiveness model. For the MAIC, individual patient data for the MAVORIC trial were weighted such that the mean baseline characteristics matched those of the ALCANZA study using both the MAVORIC ITT population and MF-only population. For PFS and response in skin, the results of the MAIC suggest that mogamulizumab is not favoured over brentuximab vedotin in either population, neither before nor after adjustment; however, the wide CIs for the PFS analyses suggest imprecision in these estimates. The results also suggest that vorinostat performed similarly to physician’s choice of treatments. There was no difference in terms of OS between mogamulizumab and brentuximab in either population before or after adjustment, and no analysis was conducted between vorinostat and physician’s choice. The key differences in the included populations of the MAVORIC and ALCANZA trials with regard to disease, stage, prior treatments, and some study design characteristics, result in multiple clinical limitations of the MAIC. Though in favour of the comparator, brentuximab vedotin, the results of the MAIC are uncertain and the results may not be generalizable to the MF and SS populations in Canada. The sponsor-submitted MAIC did not assess HRQoL outcomes.
Harms
The overall incidence of harms in the MAVORIC trial was well balanced between patients treated with mogamulizumab and vorinostat. There were some imbalances in the specific incidences of TEAEs experienced with mogamulizumab and vorinostat, despite the similar overall incidence. There was a notably higher incidence of gastrointestinal AEs associated with vorinostat compared with mogamulizumab (50.5% versus 81.7%), highlighting the unfavourable gastrointestinal disorders associated with vorinostat. Though the overall incidence of SAEs was higher in the mogamulizumab group (37.5% versus 24.7%), primarily due to increased rates in the SOCs of infections and infestations and general disorders and administration site conditions, which were more frequent with mogamulizumab (16.3% versus 10.8%; and 8.2% versus 4.3%, respectively), the clinical experts suggested that this was consistent with treatments in current clinical practice.
Notable harms such as IRRs were of interest for this review. Overall, the incidence of IRRs was higher with mogamulizumab compared with vorinostat (33.2% versus 0.5%), most of these IRRs were of mild to moderate severity and were not concerning. Infections were another notable harm of interest to this review, as patients expressed a need for treatments that reduce infections. The clinical experts noted that the incidence of infections was higher than expected in both the mogamulizumab and vorinostat groups (64.1% versus 50.0%, respectively), though the clinical experts considered the results between groups to be relatively comparable. The majority of infections were mild to moderate in severity. The management of these infections, through monitoring, antibiotics, and hospitalization, if necessary, was considered reasonable by the clinical experts consulted by CADTH. Immune-related AEs were included as a notable harm, as mogamulizumab may induce or exacerbate autoimmune disorders. According to the clinical experts consulted for this review, there was no clear signal of an increase in immune-related TEAEs. The eligibility criteria for the MAVORIC trial excluded patients with known active autoimmune disease, which may have resulted in a lower incidence of autoimmune exacerbations or AEs. Patients highlighted the importance of new treatments to control skin manifestations and reactions associated with treatments, given the nature of this disease and the emergence of mogamulizumab-associated rash. The incidence of drug eruptions was notably higher with mogamulizumab (23.9% versus 0.5%). This was a concern, given that this requires careful management through a multidisciplinary team to determine whether the manifestation is considered a drug eruption or disease progression in the skin requiring a skin biopsy and additional diagnostic differentiation studies. Other than the highlighted infections, there were no clear signals of an increased rate in the notable harms identified in this review.
Although the product monograph for mogamulizumab warns of Stevens-Johnson syndrome and toxic epidermal necrolysis, no evidence of these conditions was observed in the MAVORIC trial.
The sponsor-submitted MAIC did not assess safety outcomes.
Conclusions
Based on the clinical evidence from MAVORIC, a phase III, open-label RCT that was conducted in patients with MF or SS whose disease has failed to respond to at least 1 prior systemic therapy, mogamulizumab was associated with a clinically meaningful and statistically significant improvement in PFS and ORR compared with vorinostat; however, given the open-label design of the study, these results must be interpreted with caution due to the potential for reporting biases. Notably, these results were most pronounced in patients with advanced SS and blood involvement. Patients highlighted the need for accessible, alternative treatment options that improve survival, and HRQoL; however, due to high attrition rates, the short follow-up duration of the MAVORIC trial, and the high rate of crossover, there remains uncertainty in the effect of mogamulizumab on HRQoL and OS, as no difference between mogamulizumab and vorinostat was observed for OS. The sponsor-submitted MAIC was associated with significant clinical heterogeneity in the included studies that resulted in multiple limitations and notable uncertainty in the comparative efficacy of mogamulizumab. Given the difficulty in accessing treatment for MF and SS, the lack of a standard of care, and that vorinostat is not widely available in Canada, there is some uncertainty around the true significance of the comparative efficacy of mogamulizumab. Overall, the available evidence suggests that mogamulizumab provides an additional treatment option for patients with MF or SS whose disease has failed to respond to at least 1 systemic therapy that delays disease progression and provides a meaningful clinical response in the population of patients with MF or SS.
References
1.Hristov AC, Tejasvi T, Wilcox RA. Mycosis fungoides and Sézary syndrome: 2019 update on diagnosis, risk-stratification, and management. Am J Hematol. 2019;94(9):1027-1041. PubMed
2.Willemze R, Jaffe ES, Burg G, et al. WHO-EORTC classification for cutaneous lymphomas. Blood. 2005;105(10):3768-3785. PubMed
3.Bradford PT, Devesa SS, Anderson WF, Toro JR. Cutaneous lymphoma incidence patterns in the United States: a population-based study of 3884 cases. Blood. 2009;113(21):5064-5073. PubMed
4.Olsen E, Vonderheid E, Pimpinelli N, et al. Revisions to the staging and classification of mycosis fungoides and Sézary syndrome: a proposal of the International Society for Cutaneous Lymphomas (ISCL) and the cutaneous lymphoma task force of the European Organization of Research and Treatment of Cancer (EORTC). Blood. 2007;110(6):1713-1722. PubMed
5.Trautinger F, Eder J, Assaf C, et al. European Organisation for Research and Treatment of Cancer consensus recommendations for the treatment of mycosis fungoides/Sézary syndrome - update 2017. Eur J Cancer. 2017;77:57-74. PubMed
6.Criscione VD, Weinstock MA. Incidence of cutaneous T-cell lymphoma in the United States, 1973-2002. Arch Dermatol. 2007;143(7):854-859. PubMed
7.Agar NS, Wedgeworth E, Crichton S, et al. Survival outcomes and prognostic factors in mycosis fungoides/Sézary syndrome: validation of the revised International Society for Cutaneous Lymphomas/European Organisation for Research and Treatment of Cancer staging proposal. J Clin Oncol. 2010;28(31):4730-4739. PubMed
8.Imam MH, Shenoy PJ, Flowers CR, Phillips A, Lechowicz MJ. Incidence and survival patterns of cutaneous T-cell lymphomas in the United States. Leuk Lymphoma. 2013;54(4):752-759. PubMed
9.García-Díaz N, Piris MÁ, Ortiz-Romero PL, Vaqué JP. Mycosis fungoides and Sézary syndrome: an integrative review of the pathophysiology, molecular drivers, and targeted therapy. Cancers (Basel). 2021;13(8):1931. PubMed
10.Ghazawi FM, Netchiporouk E, Rahme E, et al. Comprehensive analysis of cutaneous T-cell lymphoma (CTCL) incidence and mortality in Canada reveals changing trends and geographic clustering for this malignancy. Cancer. 2017;123(18):3550-3567. PubMed
11.Mehta-Shah N, Horwitz SM, Ansell S, et al. NCCN guidelines insights: primary cutaneous lymphomas, version 2.2020. J Natl Compr Canc Netw. 2020;18(5):522-536. PubMed
12.NCCN guidelines for patients: mycosis fungoides/Sezary syndrome. Plymouth Meeting (PA): National Comprehensive Cancer Network; 2021: https://www.nccn.org/patients/guidelines/content/PDF/nhl-mycosis-patient.pdf. Accessed 2021 Oct 10.
13.Quaglino P, Pimpinelli N, Berti E, et al. Time course, clinical pathways, and long-term hazards risk trends of disease progression in patients with classic mycosis fungoides. Cancer. 2012;118(23):5830-5839. PubMed
14.Wieselthier JS, Koh HK. Sézary syndrome: diagnosis, prognosis, and critical review of treatment options. J Am Acad Dermatol. 1990;22(3):381-401. PubMed
15.Demierre MF, Gan S, Jones J, Miller DR. Significant impact of cutaneous T-cell lymphoma on patients’ quality of life: results of a 2005 National Cutaneous Lymphoma Foundation Survey. Cancer. 2006;107(10):2504-2511. PubMed
16.Clinical practice guideline: lymphoma. Edmonton (AB): Cancer Care Alberta; 2019: https://www.albertahealthservices.ca/assets/info/hp/cancer/if-hp-cancer-guide-lyhe002-lymphoma.pdf. Accessed 2021 Oct 21.
17.Scarisbrick JJ, Quaglino P, Prince HM, et al. The PROCLIPI international registry of early-stage mycosis fungoides identifies substantial diagnostic delay in most patients. Br J Dermatol. 2019;181(2):350-357. PubMed
18.Talpur R, Singh L, Daulat S, et al. Long-term outcomes of 1,263 patients with mycosis fungoides and Sézary syndrome from 1982 to 2009. Clin Cancer Res, 2012;18(18):5051-5060. PubMed
19.Jawed SI, Myskowski PL, Horwitz S, Moskowitz A, Querfeld C. Primary cutaneous T-cell lymphoma (mycosis fungoides and Sézary syndrome): part I. Diagnosis: clinical and histopathologic features and new molecular and biologic markers. J Am Acad Dermatol. 2014;70(2):205.e201-216; quiz 221-202. PubMed
20.Kim YH, Liu HL, Mraz-Gernhard S, Varghese A, Hoppe RT. Long-term outcome of 525 patients with mycosis fungoides and Sézary syndrome: clinical prognostic factors and risk for disease progression. Arch Dermatol. 2003;139(7):857-866. PubMed
21.Skov AG, Gniadecki R. Delay in the histopathologic diagnosis of mycosis fungoides. Acta Derm Venereol. 2015;95(4):472-475. PubMed
22.Larocca C, Kupper T. Mycosis fungoides and Sézary syndrome: an update. Hematol Oncol Clin North Am. 2019;33(1):103-120. PubMed
23.Tsambiras PE, Patel S, Greene JN, Sandin RL, Vincent AL. Infectious complications of cutaneous t-cell lymphoma. Cancer Control. 2001;8(2):185-188. PubMed
24.Axelrod PI, Lorber B, Vonderheid EC. Infections complicating mycosis fungoides and Sézary syndrome. JAMA. 1992;267(10):1354-1358. PubMed
25.NCCN Guidelines: primary cutaneous lymphomas version 2.2021. Plymouth Meeting (PA): National Comprehensive Cancer Network; 2021 Mar 4.
26.Poteligeo (mogamulizumab): 20 mg/5 mL single-use vial, 4 mg/mL for intravenous infusion [DRAFT product monograph]. Bedminster (NJ): Kyowa Kirin, Inc.
27.Clinical Study Report: 0761-010, amendment 01. Open-label, multi-center, randomized study of anti-CCR4 monoclonal antibody KW-0761 (mogamulizumab) versus vorinostat in subjects with previously treated cutaneous T-cell lymphoma (CTCL) [internal sponsor’s report]. Princeton (NJ): Kyowa Kirin Pharmaceutical Development, Inc; 2018 Nov 26.
28.Leoni M, Ito T, Jones T, Li J. PCN54. Efficacy and safety of mogamulizumab in previously treated, advanced stage mycosis fungoides and Sézary syndrome patients: a post-hoc analysis of the MAVORIC study. Value Health. 2019;22:S445-S446.
29.Cowan RA, Scarisbrick JJ, Zinzani PL, et al. Efficacy and safety of mogamulizumab by patient baseline blood tumour burden: a post hoc analysis of the MAVORIC trial. J Eur Acad Dermatol Venereol. 2021;17:17. PubMed
30.Quality of life analysis in subjects with cutaneous T-cell lymphoma treated with anti-CCR4 monoclonal antibody KW-0761 (mogamulizumab) versus vorinostat [internal sponsor’s report]. In: Drug Reimbursement Review sponsor submission: Poteligeo (mogamulizumab), 4 mg/mL (20 mg) for intravenous infusion. Bedminster (NJ): Kyowa Kirin Canada, Inc; 2021 Aug 5.
31.Olsen EA, Whittaker S, Kim YH, et al. Clinical end points and response criteria in mycosis fungoides and Sézary syndrome: a consensus statement of the International Society for Cutaneous Lymphomas, the United States Cutaneous Lymphoma Consortium, and the Cutaneous Lymphoma Task Force of the European Organisation for Research and Treatment of Cancer. J Clin Oncol. 2011;29(18):2598-2607. PubMed
32.Scarisbrick JJ, Kim YH, Whittaker SJ, et al. Prognostic factors, prognostic indices and staging in mycosis fungoides and Sézary syndrome: where are we now? Br J Dermatol. 2014;170(6):1226-1236. PubMed
33.Zinzani PL, Ferreri AJ, Cerroni L. Mycosis fungoides. Crit Rev Oncol Hematol. 2008;65(2):172-182. PubMed
34.Olek-Hrab K, Silny W. Diagnostics in mycosis fungoides and Sezary syndrome. Rep Pract Oncol Radiother. 2013;19(2):72-76. PubMed
35.Drug Reimbursement Review sponsor submission: Poteligeo (mogamulizumab), 4 mg/mL (20 mg) for intravenous infusion [internal sponsor’s package]. Bedminster (NJ): Kyowa Kirin Canada, Inc; 2021 Aug 5.
36.INTRON A (interferon alfa-2b): lyophilized powder with diluent: 10 million IU interferon alfa-2b/vial [product monograph]. Kirkland (QC): Merck Canada Inc; 2019 July 5: https://pdf.hres.ca/dpd_pm/00052048.PDF. Accessed 2021 Nov 22.
37.Accutane (isotretinoin): 10 mg, 40 mg capsule USP [product monograph]. Mississauga (ON): Hoffmann-La Roche Limited; 2019 Aug 21: https://pdf.hres.ca/dpd_pm/00052764.PDF. Accessed 2021 Nov 22.
38.Gemzar (gemcitabine hydrochloride): 200 mg or 1 g gemcitabine per vial for injection [product monograph]. Toronto (ON): Eli Lilly Canada; 2014 Apr 28: https://pdf.hres.ca/dpd_pm/00025004.PDF. Accessed 2021 Nov 22.
39.Zolinza (vorinostat): 100 mg capsules [product monograph]. Kirkland (QC): Merck Canada Inc; 2013 Oct 18: https://pdf.hres.ca/dpd_pm/00022508.PDF. Accessed 2021 Nov 22.
40.Adcetris (brentuximab vedotin): lyophilized powder for reconstitution with 10.5 mL of sterile water for Injection, USP 50 mg [product monograph]. Mississauga (ON): Seagen Canada Inc; 2021 Jun 11: https://pdf.hres.ca/dpd_pm/00061748.PDF. Accessed 2021 Nov 22.
41.McGowan J, Sampson M, Salzwedel DM, Cogo E, Foerster V, Lefebvre C. PRESS Peer Review of Electronic Search Strategies: 2015 guideline statement. J Clin Epidemiol. 2016;75:40-46. PubMed
42.Grey matters: a practical tool for searching health-related grey literature. Ottawa (ON): CADTH; 2019: https://www.cadth.ca/grey-matters. Accessed 2021 Sep 28.
43.Kim YH, Bagot M, Pinter-Brown L, et al. Mogamulizumab versus vorinostat in previously treated cutaneous T-cell lymphoma (MAVORIC): an international, open-label, randomised, controlled phase 3 trial. Lancet Oncol. 2018;19(9):1192-1204. PubMed
44.Kyowa Kirin Canada response to December 6, 2021 DRR request for additional information regarding Poteligeo (mogamulizumab) DRR review [internal additional sponsor’s information]. Bedminster (NJ): Kyowa Kirin Canada, Inc; 2021 Dec 9.
45.Geskin LJ, Scarisbrick J, Bagot M, et al. Efficacy of mogamulizumab in previously treated patients with less advanced mycosis fungoides: results from the MAVORIC study. J Clin Oncol. 2019;37(15_suppl):e19031-e19031.
46.Horwitz S, Zinzani PL, Bagot M, et al. Lack of impact of type and extent of prior therapy on outcomes of mogamulizumab therapy in patients with cutaneous T cell lymphoma in the MAVORIC trial. Leuk Lymphoma. 2021:1-10. PubMed
47.Mogamulizumab: report on indirect treatment comparisons: final version [internal sponsor’s report]. In: Drug Reimbursement Review sponsor submission: Poteligeo (mogamulizumab), 4 mg/mL (20 mg) for intravenous infusion. Bedminster (NJ): Kyowa Kirin Canada, Inc; 2020 May 21.
48.Uzelac D. Modified Severity-Weighted Assessment Tool (mSWAT). Birmingham (MI): Cutaneous Lymphoma Foundation; 2020: https://www.clfoundation.org/modified-severity-weighted-assessment-tool-mswat. Accessed 2021 Nov 2.
49.Stevens SR, Ke MS, Parry EJ, Mark J, Cooper KD. Quantifying skin disease burden in mycosis fungoides-type cutaneous T-cell lymphomas: the severity-weighted assessment tool (SWAT). Arch Dermatol. 2002;138(1):42-48. PubMed
50.Chren MM. The Skindex instruments to measure the effects of skin disease on quality of life. Dermatol Clin. 2012;30(2):231-236, xiii. PubMed
51.Chren MM, Lasek RJ, Flocke SA, Zyzanski SJ. Improved discriminative and evaluative capability of a refined version of Skindex, a quality-of-life instrument for patients with skin diseases. Arch Dermatol. 1997;133(11):1433-1440. PubMed
52.Porkert S, Lehner-Baumgartner E, Valencak J, Knobler R, Riedl E, Jonak C. Patients’ illness perception as a tool to improve individual disease management in primary cutaneous lymphomas. Acta Derm Venereol. 2018;98(2):240-245. PubMed
53.Demierre MF, Tien A, Miller D. Health-related quality-of-life assessment in patients with cutaneous T-cell lymphoma. Arch Dermatol. 2005;141(3):325-330. PubMed
54.Yost KJ, Thompson CA, Eton DT, et al. The Functional Assessment of Cancer Therapy - General (FACT-G) is valid for monitoring quality of life in patients with non-Hodgkin lymphoma. Leuk Lymphoma. 2013;54(2):290-297. PubMed
55.Yost KJ, Eton DT. Combining distribution- and anchor-based approaches to determine minimally important differences: the FACIT experience. Eval Health Prof. 2005;28(2):172-191. PubMed
56.Cella D, Hahn EA, Dineen K. Meaningful change in cancer-specific quality of life scores: differences between improvement and worsening. Qual Life Res. 2002;11(3):207-221. PubMed
57.Sinnott PL, Joyce VR, Barnett PG. Preference measurement in economic analysis. Menlo Park (CA): Health Economics Resource Center (HERC); 2007: https://www.herc.research.va.gov/files/BOOK_419.pdf. Accessed 2021 Nov 2.
58.Teckle P, Peacock S, McTaggart-Cowan H, et al. The ability of cancer-specific and generic preference-based instruments to discriminate across clinical and self-reported measures of cancer severities. Health Qual Life Outcomes. 2011;9:106. PubMed
59.Pickard AS, Neary MP, Cella D. Estimation of minimally important differences in EQ-5D utility and VAS scores in cancer. Health Qual Life Outcomes. 2007;5:70. PubMed
60.Jang YH, Kim SM, Eun DH, et al. Validity and reliability of itch assessment scales for chronic pruritus in adults: a prospective multicenter study. J Am Acad Dermatol. 2020;82(1):80-86. PubMed
61.Phan NQ, Blome C, Fritz F, et al. Assessment of pruritus intensity: prospective study on validity and reliability of the visual analogue scale, numerical rating scale and verbal rating scale in 471 patients with chronic pruritus. Acta Derm Venereol. 2012;92(5):502-507. PubMed
62.Schoch D, Sommer R, Augustin M, Stander S, Blome C. Patient-reported outcome measures in pruritus: a systematic review of measurement properties. J Invest Dermatol. 2017;137(10):2069-2077. PubMed
63.Desai NS, Poindexter GB, Monthrope YM, Bendeck SE, Swerlick RA, Chen SC. A pilot quality-of-life instrument for pruritus. J Am Acad Dermatol. 2008;59(2):234-244. PubMed
64.Zeidler C, Steinke S, Riepe C, et al. Cross-European validation of the ItchyQoL in pruritic dermatoses. J Eur Acad Dermatol Venereol. 2019;33(2):391-397. PubMed
65.Stumpf A, Pfleiderer B, Fritz F, Osada N, Chen SC, Stander S. Assessment of quality of life in chronic pruritus: relationship between ItchyQoL and Dermatological Life Quality Index in 1,150 patients. Acta Derm Venereol. 2018;98(1):142-143. PubMed
Appendix 1: Literature Search Strategy
Note that this appendix has not been copy-edited.
Clinical Literature Search
Overview
Interface: Ovid
Databases
MEDLINE All (1946–present)
Embase (1974–present)
Note: Subject headings and search fields have been customized for each database. Duplicates between databases were removed in Ovid.
Date of search: October 12, 2021
Alerts: Biweekly search updates until project completion
Search filters applied: No filters were applied to limit the retrieval by study type.
Limits
No date or language limits were used
Conference abstracts: excluded
Syntax | Description |
|---|---|
/ | At the end of a phrase, searches the phrase as a subject heading |
MeSH | Medical Subject Heading |
exp | Explode a subject heading |
* | Before a word, indicates that the marked subject heading is a primary topic; or, after a word, a truncation symbol (wildcard) to retrieve plurals or varying endings |
.ti | Title |
.ot | Original title |
.ab | Abstract |
.hw | Heading word; usually includes subject headings and controlled vocabulary |
.kf | Author keyword heading word (MEDLINE) |
.kw | Author keyword (Embase) |
.dq | Candidate term word (Embase) |
.rn | Registry number |
.nm | Name of substance word (MEDLINE) |
medall | Ovid database code: MEDLINE All, 1946 to present, updated daily |
oemezd | Ovid database code; Embase, 1974 to present, updated daily |
Multi-Database Strategy
(mogamulizumab* or Poteligeo* or amg 761 or amg761 or km 8761 or km8761 or “kw 0761” or kw0761 or YI437801BE).ti,ab,kf,ot,hw,rn,nm.
1 use medall
*mogamulizumab/
(mogamulizumab* or Poteligeo* or amg 761 or amg761 or km 8761 or km8761 or “kw 0761” or kw0761).ti,ab,kf,dq.
3 or 4
5 use oemezd
6 not (conference review or conference abstract).pt.
2 or 7
remove duplicates from 8
Clinical Trials Registries
ClinicalTrials.gov
Produced by the US National Library of Medicine. Targeted search used to capture registered clinical trials.
[Search -- Studies with results | mogamulizumab or Poteligeo]
Health Canada’s Clinical Trials Database
Produced by Health Canada. Targeted search used to capture registered clinical trials.
[Search terms -- mogamulizumab or Poteligeo]
EU Clinical Trials Register
European Union Clinical Trials Register, produced by the European Union. Targeted search used to capture registered clinical trials.
[Search terms -- mogamulizumab or Poteligeo]
Grey Literature
Search dates: September 29, 2021 to October 5, 2021
Keywords: mogamulizumab or Poteligeo, mycosis fungoides, Sézary syndrome, cutaneous T-cell lymphoma
Limits: None
Updated: Search updated before the completion of stakeholder feedback period
Relevant websites from the following sections of the CADTH grey literature checklist Grey Matters: A Practical Tool for Searching Health-Related Grey Literature were searched:
Health Technology Assessment Agencies
Health Economics
Clinical Practice Guidelines
Drug and Device Regulatory Approvals
Advisories and Warnings
Drug Class Reviews
Clinical Trials Registries
Databases (free)
Health Statistics
Internet Search
Appendix 2: Excluded Studies
Note that this appendix has not been copy-edited.
Reference | Reason for exclusion |
|---|---|
Kim YH, Bagot M, Pinter-Brown L, et al. Mogamulizumab vs. vorinostat in previously treated cutaneous T-cell lymphoma (MAVORIC): an international, open-label, randomized, controlled phase 3 trial. Lancet Oncol. 2018;19(9):1192 to 1204. | Duplicate |
Kim YH, Khodadoust M, de Masson A, et al. Patient characteristics of long-term responders to mogamulizumab: results from the MAVORIC study. Eur J Cancer. 2021 October;156(Supplement 1):S48-S49. | Outcome |
Musiek ACM, Whittaker S, Horowitz SM, et al. Characterization and outcomes in patients with mogamulizumab-associated skin reactions in the MAVORIC trial. Eur J Cancer. 2021 October;156(Supplement 1):S46. | Outcome |
Tamai H, Tajika K, Nakayama K, Arai A. Treatment of relapsed aggressive adult T-cell leukemia/lymphoma after allogeneic hematopoietic stem cell transplantation with mogamulizumab followed by lenalidomide. Bone Marrow Transplant. 2021 Aug 12;12:12. PubMed: PM34385616. | Study population |
Zinzani PL, Ortiz-Romero P, Lima M, Huen AO. Health-related quality of life effect of mogamulizumab by patient blood involvement. Eur J Cancer. 2021 October;156(Supplement 1):S65-S66. | Abstract only |
Fujimura T, Kambayashi Y, Ohuchi K, Amagai R, Muto Y, Aiba S. Successful treatment of CCR4+ mycosis fungoides palmaris et plantaris with mogamulizumab monotherapy. Dermatol Ther. 2020 07;33(4):e13731. PubMed: PM32474985. | Study design (case report) |
Lewis DJ, Rook AH. Mogamulizumab in the treatment of advanced mycosis fungoides and Sezary syndrome: safety and efficacy. Expert Rev Anticancer Ther. 2020 06;20(6):447 to 452. PubMed: PM32320304. | Review |
Amakata M, Teraki Y. Depletion of regulatory FoxP3 < sup > + < /sup > T cells in the pathogenesis of Stevens-Johnson syndrome induced by mogamulizumab. Int J Dermatol. 2019 Dec;58(12):e247-e249. PubMed: PM31334834. | Study design (case report) |
Bonnet P, Battistella M, Roelens M, et al. Association of autoimmunity and long-term complete remission in patients with Sezary syndrome treated with mogamulizumab. Br J Dermatol. 2019 02;180(2):419 to 420. PubMed: PM30328116. | Study design (case report) |
Johnson WT, Kartan S, Sokol K, Nikbakht N, Porcu P. Clinical characteristics and outcomes of black patients with mycosis fungoides and Sezary syndrome: a subgroup analysis of the phase III MAVORIC trial. Leuk Lymphoma. 2021 08;62(8):1877 to 1883. PubMed: PM33618592. | Study population |
Akilov O, Geskin L, Ito T, Dwyer K, Herr F, Musiek A. TCL-127: Impact of Concomitant Steroids on Mogamulizumab Efficacy in MAVORIC. Clinical Lymphoma Myeloma and Leukemia. 2020;20:S252-S253. | Outcome |
Bagot M, Dalle S, Sokol L, et al. Long-Term Clinical Benefit to Anti-CCR4 Mogamulizumab: Results from the phase 3 Mavoric Study in Previously Treated Cutaneous T-Cell Lymphoma (CTCL). Blood. 2018;132(Supplement 1):2901 to 2901. | Outcome |
Hudgens S, Dunn J, Floden L, Spencer G. Evaluation Of Meaningful Change In Patient-Related Outcomes For Patients With Cutaneous T-Cell Lymphoma. Value in Health. 2018;21:S38. | Duplicate |
Hudgens S, Dunn J, Floden L, Spencer G. PCN152 - Relationship Between Health State and Patient Reported Outcome In Cutaneous T-Cell Lymphoma: An Exploratory Analysis. Value in Health. 2018;21:S35. | Duplicate |
Hudgens S, Floden L, Leoni M, Nikonova E, Quaglino P. PCN350 - EVALUATION OF DISEASE-SPECIFIC SKIN SYMPTOM ITEMS ON SKINDEX-29 IN CUTANEOUS T-CELL LYMPHOMA PATIENTS TREATED WITH MOGAMULIZUMAB OR VORINOSTAT. Value in Health. 2018;21:S73-S74. | Duplicate |
Hudgens S, Porcu P, Quaglino P, et al. Evaluation of Symptom and Side Effect Bother in Cutaneous T-Cell Lymphoma Patients Treated with Mogamulizumab or Vorinostat. Blood. 2018;132(Supplement 1):3592 to 3592. | Duplicate |
Pro B, Kim YH, Ortiz-Romero PL, et al. Time to next treatment in patients with previously treated cutaneous T-cell lymphoma (CTCL) receiving mogamulizumab or vorinostat: A MAVORIC post-hoc analysis. Journal of Clinical Oncology. 2019;37(15_suppl):7539 to 7539. | Outcome |
Zinzani PL, Horwitz SM, Kim YH, et al. Efficacy of Mogamulizumab By Prior Systemic Therapy in Patients with Previously Treated Cutaneous T-Cell Lymphoma: Post Hoc Analysis from the phase 3 Mavoric Study. Blood. 2018;132(Supplement 1):1619 to 1619. | Duplicate |
Porcu P, Hudgens S, Horwitz S, et al. Quality of Life Effect of the Anti-CCR4 Monoclonal Antibody Mogamulizumab vs. Vorinostat in Patients With Cutaneous T-cell Lymphoma. Clin Lymphoma Myeloma Leuk. 2021 02;21(2):97 to 105. PubMed: PM33158772. | Duplicate |
Beylot-Barry M, Booken N, Weishaupt C, Medley M, Sun W, Rosen JP. Efficacy of mogamulizumab in mycosis fungoides by patient blood involvement and time to response analysis in mycosis fungoides and Sezary syndrome: a post hoc analysis of the MAVORIC study. Eur J Cancer. 2021;156(Supplement 1):S47-S48. | Duplicate |
Quaglino P, Iversen L, Dummer R, Musiek A, Rosen J-P. P139. The Correlation Between Skin Response and Blood Involvement with Mogamulizumab. Poster presented at the 16th EADO Congress. 12 to 14 October 2020, Vilnius, Lithuania. | Duplicate |
Scarisbrick J, Zinzani PL, Cowan R, et al. P138. Efficacy and Safety of Mogamulizumab by Patient Blood Classification. Poster presented at the 16th EADO Congress. 12 to 14 October 2020, Vilnius, Lithuania. | Duplicate |
Appendix 3: Detailed Outcome Data
Note that this appendix has not been copy-edited.
Table 40: Summary of Confirmed Response Rate by Disease Compartment (ITT; Independent Review)
Compartment | Independent Review | |
|---|---|---|
Mogamulizumab (N = 186) | Vorinostat (N = 186) | |
Blood, n | 130 | 133 |
ORR (confirmed CR + PR), n (%) | 77 (59.2) | 23 (17.3) |
95% CIa | (50.3 to 67.8) | (11.3 to 24.8) |
Risk Difference, Mogamulizumab vs. Vorinostat (95% CI)a | 41.9 (30.4 to 52.3) | |
P valueb | < 0.0001 | |
Skin, n | 186 | 186 |
ORR (confirmed CR + PR), n (%) | 73 (39.2) | 27 (14.5) |
95% CIa | (32.2 to 46.7) | (9.8 to 20.4) |
Risk difference, mogamulizumab vs. vorinostat (95% CI)a | 24.7 (14.5 to 34.6) | |
P valueb | < 0.0001 | |
Lymph Nodes, n | 158 | 153 |
ORR (confirmed CR + PR), n (%) | 15 (9.5) | 6 (3.9) |
95% CIa | (5.4 to 15.2) | (1.5 to 8.3) |
Risk difference, mogamulizumab vs. vorinostat (95% CI)a | 5.6 (−5.6 to 16.7) | |
P valueb | 0.0440 | |
Viscera, n | 12 | 13 |
ORR (confirmed CR + PR), n (%) | 1 (8.3) | 0 |
95% CIa | (0.2 to 38.5) | (0.0 to 24.7) |
Risk difference, mogamulizumab vs. vorinostat (95% CI)a | 8.3 (−30.3 to 44.6) | |
P valueb | 0.4795 | |
CI = confidence interval; CR = complete response; ORR = overall response rate; PR = partial response.
aThe 95% CIs for response rate are the exact 95% CIs. The 95% CI for difference is the exact 95% unconditional CI for the risk difference (mogamulizumab minus vorinostat).
bP value was obtained from Cochran-Mantel-Haenszel test adjusting for disease type, disease stage, and region. Adjusted P value (for ORR) was calculated using the Šidák method.
Source: MAVORIC (0761 to 010) Clinical Study Report.27
Table 41: Confirmed ORR by Number of Prior Systemic Therapies (ITT) as Assessed by Independent Review
Number of Prior Systemic Therapies | Mogamulizumab | Vorinostat | ||
|---|---|---|---|---|
n | ORR (95% CI)a | n | ORR (95% CI)a | |
0 | 0 | 0 | 1 | 0 (0.0 to 97.5) |
1 | 28 | 10.7 (2.3 to 28.2) | 40 | 2.5 (0.1 to 13.2) |
2 | 40 | 17.5 (7.3 to 32.8) | 38 | 10.5 (2.9 to 24.8) |
3 | 40 | 27.5 (14.6 to 43.9) | 37 | 2.7 (0.1 to 14.2) |
4 | 22 | 40.9 (20.7 to 63.6) | 18 | 5.6 (0.1 to 27.3) |
5 | 12 | 16.7 (2.1 to 48.4) | 21 | 0 (0.0 to 16.1) |
≥ 6 | 44 | 25.0 (13.2 to 40.3) | 31 | 0 (0.0 to 11.2) |
CI = confidence interval; ITT = intention-to-treat population; ORR = overall response rate.
aThe 95% CI for response rate is the exact 95% CI.
Source: MAVORIC (0761 to 010) Clinical Study Report.27
Table 42: Summary of BOR During Randomized Treatment Period (ITT) as Assessed by Independent Review
Category | Mogamulizumab (N = 186) | Vorinostat (N = 186) |
|---|---|---|
Responders (CR + PR) | 54 (29.0) | 13 (7.0) |
CR | 3 (1.6) | 0 |
Confirmed CR | 3 (1.6) | 0 |
PR | 51 (27.4) | 13 (7.0) |
Confirmed PR | 40 (21.5) | 7 (3.8) |
Stable disease | 88 (47.3) | 115 (61.8) |
Progressive disease | 2 (1.1) | 6 (3.2) |
Not assessableb | 42 (22.6) | 52 (28.0) |
CR = complete response; ITT = intention-to-treat population; PR = partial response.
aOne patient in the mogamulizumab group had a confirmed PR based on investigator’s assessment, but the BOR was an unconfirmed CR in this table at time of data cut. Since this patient had a CR with relapse only in skin, they should have been counted as confirmed CR since a relapse in skin does not constitute progression if it does not meet the specified criteria for progression.
bIf there was no post-baseline tumour assessment or the response for all post-baseline tumour assessments was not assessable or unable to evaluate, the best overall response was classified as not assessable.
Source: MAVORIC (0761 to 010) Clinical Study Report.27
Table 43: Summary of DOR During Randomized Treatment Period (ITT) as Assessed by Independent Review
DOR | Mogamulizumab (N = 186) | Vorinostat (N = 186) |
|---|---|---|
Number of Patients with Confirmed CR or PR | 43 | 7 |
Patients with PD or Death, n (%) | 16 (37.2) | 1 (14.3) |
Patients Censored, n (%) | 27 (62.8) | 6 (85.7) |
Median (95% CI) DOR (months)a | 16.07 (11.73 to NE) | NE |
CI = confidence interval; CR = complete response; DOR = duration of response; NE = not estimable; PD = progressive disease; PR = partial response.
aKaplan-Meier estimate.
Source: MAVORIC (0761 to 010) Clinical Study Report.27
Table 44: Summary of Sensitivity Analyses for PFS by Investigator’s Assessment (ITT)
Sensitivity analyses | Mogamulizumab (N = 186) | Vorinostat (N = 186) |
|---|---|---|
Sensitivity analysis 1 | ||
PFS Event, n (%) | 106 (57.0) | 131 (70.4) |
Progressive disease | 103 (55.4) | 127 (68.3) |
Death | 3 (1.6) | 4 (2.2) |
Censored, n (%) | 80 (43.0) | 55 (29.6) |
Median PFS, months (95% CI)a | 8.43 (5.63,11.17) | 3.10 (2.87 to 4.07) |
HR (95% CI); log-rank P valueb | 0.52 (0.40 to 0.68); < 0.0001 | |
Sensitivity analysis 2 | ||
PFS Event, n (%) | 128 (68.8) | 135 (72.6) |
Progressive disease | 123 (66.1) | 134 (72.0) |
Death | 5 (2.7) | 1 (0.5) |
Censored, n (%) | 58 (31.2) | 51 (27.4) |
Median PFS, months (95% CI)a | 5.77 (4.63 to 8.43) | 3.07 (2.87 to 3.93) |
HR (95% CI); log-rank P valueb | 0.61 (0.47 to 0.78); < 0.0001 | |
Sensitivity analysis 3 | ||
PFS Event, n (%) | 110 (59.1) | 93 (50.0) |
Progressive disease | 104 (55.9) | 86 (46.2) |
Death | 6 (3.2) | 7 (3.8) |
Censored, n (%) | 76 (40.9) | 93 (50.0) |
Median PFS, months (95% CI)a | 9.67 (7.53 to11.50) | 4.70 (3.33 to 6.17) |
HR (95% CI); log-rank P valueb | 0.72 (0.54 to 0.96); 0.0148 | |
Sensitivity analysis 4 | ||
PFS Event, n (%) | 106 (57.0) | 131 (70.4) |
Progressive disease | 103 (55.4) | 127 (68.3) |
Death | 3 (1.6) | 4 (2.2) |
Censored, n (%) | 80 (43.0) | 55 (29.6) |
Median PFS, months (95% CI)a | 8.43 (5.63 to11.17) | 3.10 (2.87 to 4.07) |
HR (95% CI); log-rank P valueb | 0.52 (0.40 to 0.68); < 0.0001 | |
CI = confidence interval; HR = hazard ratio; ITT = intention-to-treat population; PFS = progression-free survival.
a95% CIs are obtained from SAS PROC LIFETEST using a log-log transformation.
bHR and 95% CI are based on Cox proportional hazards model with treatment, disease type, disease stage, and region as covariates. P value (2-sided) is obtained from a stratified log-rank test (1-sided test at 0.025 level or equivalent 2-sided test at 0.05 level) with disease type, disease stage, and region as stratification factors.
Source: MAVORIC (0761 to 010) Clinical Study Report.
Figure 10: Investigator-Assessed PFS in the Overall Population and by Blood Tumour Classification
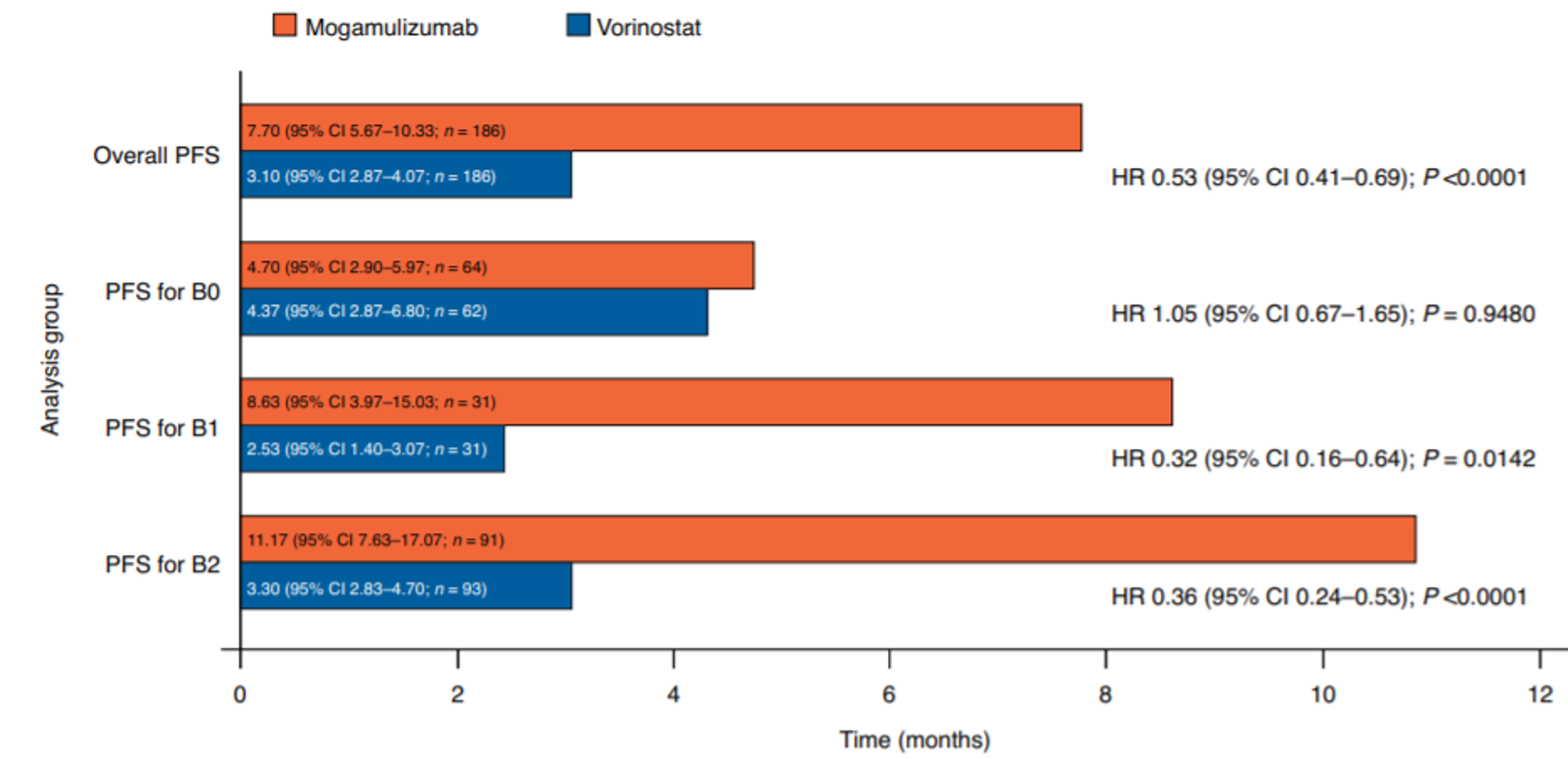
CI = confidence interval; HR = hazard ratio; PFS = progression-free survival.
Source: Cowan et al., 2021.29 This work is licensed under the Attribution-NonCommercial-NoDerivatives 4.0 International License.
Table 45: Investigator-Assessed ORR by Blood Classification
Blood classification | Mogamulizumab | Vorinostat |
|---|---|---|
B0 | ||
N | 64 | 62 |
ORR, % (95% CI) | 15.6 (7.8 to 26.9) | 6.5 (1.8 to 15.7) |
Risk difference (95% CI) | 9.2 (−2.4, 21.2) | |
P value | 0.0549 | |
B1 | ||
N | 31 | 31 |
ORR, % (95% CI) | 25.8 (11.9 to 44.6) | 6.5 (0.8 to 21.4) |
Risk difference (95% CI) | 19.4 (0.6, 38.6) | |
P value | 0.2758 | |
B2 | ||
N | 91 | 92 |
ORR, % (95% CI) | 37.4 (27.4 to 48.1) | 3.2 (0.7 to 9.1) |
Risk difference (95% CI) | 34.1 (22.9, 45.2) | |
P value | < 0.0001 | |
CI = confidence interval; ORR = overall response rate.
Source: Cowan et a., 2021.29
Table 46: Median PFS and DOR After Mogamulizumab Treatment by Number of Prior Systemic Therapies in Patients Randomized to Mogamulizumab (ITT)
Number of prior CTCL therapies | Mogamulizumab (N = 186) | ||
|---|---|---|---|
N | Median PFS, months | Median DOR, months (95% CI) | |
0 | 0 | — | — |
1 | 28 | 5.67 | 13.1 (3.8, 13.1) |
2 | 40 | 9.37 | 19.9 (6.1, 20.6) |
3 | 40 | 9.00 | 18.0 (4.7, 18.0) |
4 | 22 | 10.30 | 19.2 (3.8, –) |
5 | 12 | 5.77 | 14.0 (–, –) |
≥ 6 | 44 | 8.43 | 9.4 (4.5, –) |
CI = confidence interval; CTCL = cutaneous T-cell lymphoma; DOR = duration of response; PFS = progression-free survival.
Source: Horwitz et al., 2021.46
Table 47: ORR, PFS, and DOR by Immediate Prior Therapy Class in Patients Randomized to Mogamulizumab (ITT)
Type of prior systemic therapy | Mogamulizumab (N = 186) | |||
|---|---|---|---|---|
N | Median PFS (95% CI), months | ORR, % | Median DOR (95% CI), months | |
Therapy class | ||||
Immunostimulatory | 33 | 10.3 (4.67 to 19.03) | 21.2 | 18.0 (5.60 to 20.57) |
Immune-neutral | 55 | 8.4 (5.67 to 20.13) | 36.4 | 14.0 (12.20 to 19.23) |
Immunoinhibitory | 49 | 10.3 (4.7 to 15.03) | 20.4 | 8.5 (3.77 to NE) |
HDAC inhibitors | 13 | 5.1 (1.03 to NE) | 38.5 | NE (4.47 to NE) |
CI = confidence interval; DOR = duration of response; NE = not estimable; ORR = overall response rate; PFS = progression-free survival.
Source: Horwitz et al. (2021).46
Table 48: Exploratory Analyses of Overall Survival Adjusting for Crossover
Crossover Adjustment | HR (95% CI) |
|---|---|
As randomized | 0.93 (0.61 to 1.43) |
Censoring at crossovera | 0.709 (0.405 to 1.242) |
RPSFT modellingb | 0.739 (0.479 to 1.141) |
IPCW methodc | 0.508 (0.172 to 2.905) |
CI = confidence interval; HR = hazard ratio; IPCW = inverse probability censoring weighting; RPSFT = rank-preserving structural failure time.
aCrossed over censored at time crossover occurred (P value and CI by log-rank and Cox model with covariates stage, type, and region).
bP value and 95% CI by log-rank and Cox model with covariates of stage, type.
cThe 95% CI was based on 10,000 bootstrap samples with logistic models for the weight calculation, including terms of baseline ECOG score, age group, sex, stage, type, region, time-dependent disease progression status, and number of adverse events.
Source: Sponsor submission.35
Figure 11: ORR, PFS, and DOR by Prior Systemic Therapy and HDACi Exposure (ITT)
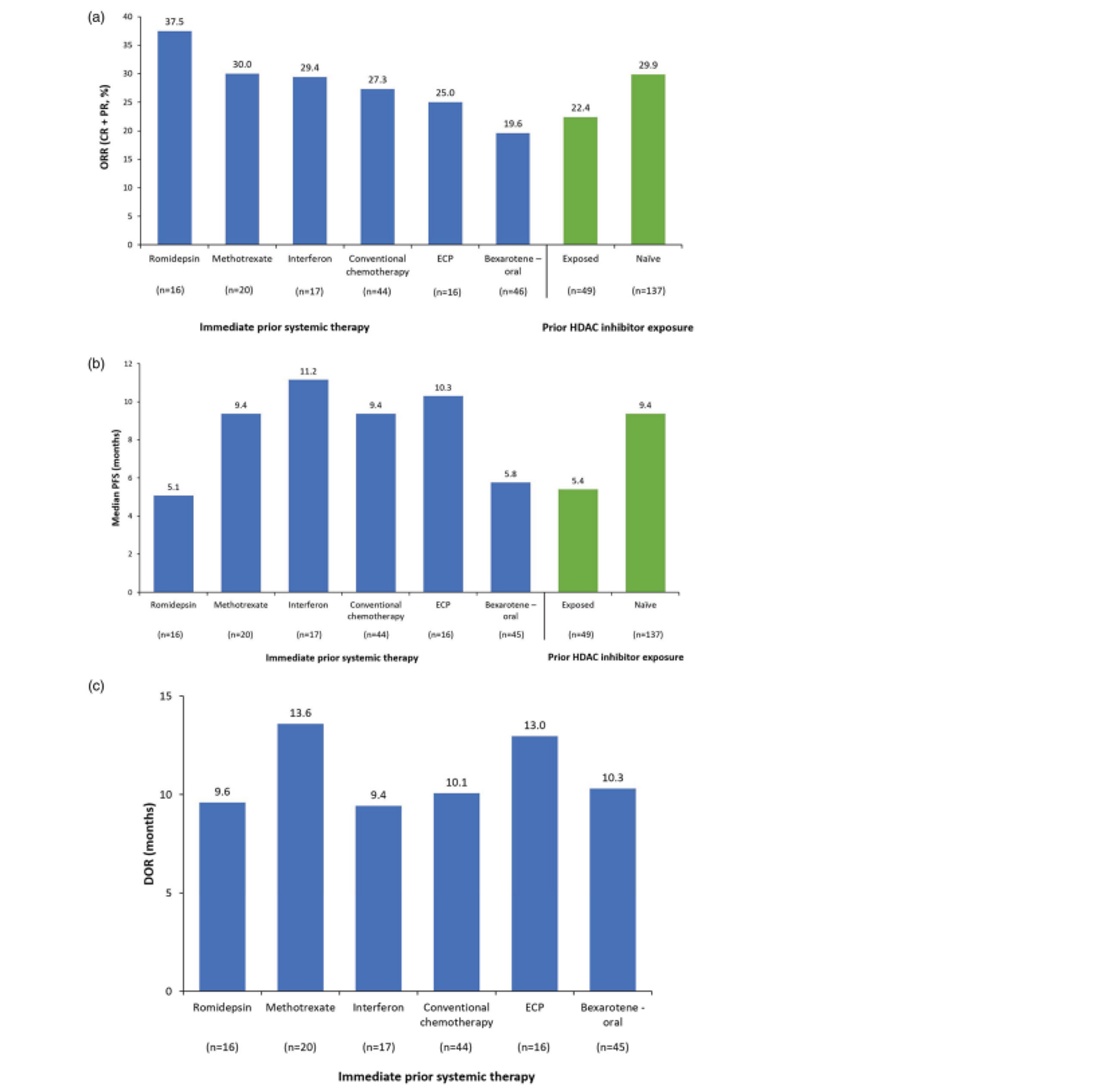
CR = complete response; DOR = duration of response; ECP = extracorporeal photopheresis; HDAC = histone deacetylase; ORR = overall response rate; PFS = progression-free survival; PR = partial response.
a) Confirmed global ORR; b) median PFS; and (c) DOR to mogamulizumab by immediate prior systemic therapy and prior HDACi exposure (ITT)
Source: Horwitz et al. (2021).46 This work is licensed under the Attribution-NonCommercial-NoDerivatives 4.0 International License.
Appendix 4: Description and Appraisal of Outcome Measures
Note that this appendix has not been copy-edited.
Aim
To describe the following outcome measures and review their measurement properties (validity, reliability, responsiveness to change, and minimal important difference [MID]):
Modified Severity-Weighted Assessment Tool (mSWAT)
Global Composite Response Score
Skindex-29
FACT-G
EQ-5D-3L
Pruritis Likert Scale
Itchy Quality of Life (ItchyQoL)
Findings
Table 49: Summary of Outcome Measures and Their Measurement Properties
Outcome measure | Type | Conclusions about measurement properties | MID |
|---|---|---|---|
mSWAT | Instrument for assessing the severity of disease based on percent BSA affected and weighted by lesion type (patch, plaque, or tumour). A higher score indicates greater disease severity. | Validity No evidence found for patients with MF or SS. Reliability No evidence found for patients with MF or SS. Responsiveness No evidence found for patients with MF or SS. | Not identified in populations with MF or SS. |
Global Composite Response Score | Definitions for complete response, partial response, stable disease, progressive disease, and relapse based on response in each compartment. | NA | NA |
Skindex-29 | A 29-item instrument for measuring how skin conditions affect HRQoL consisting of 3 domains (emotions, symptoms, and functioning) and scored on a 5-point Likert scale which is transformed to a linear numerical scale (0 to 100). A higher score indicates worse HRQoL. | Validity Face validity demonstrated for patients with MF and SS. Evidence of construct and content validity for patients with various skin conditions. Reliability Acceptable internal consistency for patients with MF, SS, or CTCL. Acceptable reproducibility for patients with various skin conditions. Responsiveness Evidence of responsiveness for patients who reported improved or worsened condition. | Not identified in populations with MF or SS. |
FACT-G | A 27-item instrument for assessing HRQoL in patients with cancer consisting of 4 domains (PWB, SWB, EWB, and FWB) and scored on a 5-point Likert scale. A higher score indicates better HRQoL. | Validity Some evidence of concurrent validity for patients with non-Hodgkin lymphoma. Reliability: Acceptable internal consistency for patients with non-Hodgkin lymphoma. Responsiveness: Evidence of responsiveness for the total scale, PWB, and FWB subscales for patients with non-Hodgkin lymphoma. | Not identified in populations with MF or SS. |
EQ-5D-3L | A patient-reported, generic, HRQoL instrument consisting of 5 domains (mobility, self-care, usual activities, pain/discomfort, and anxiety/depression) and ranked on a 3-point ordinal scale. A higher score indicates better HRQoL. | Validity: Moderate to strong concurrent validity for patients with cancer. Reliability: Acceptable reliability for patients with cancer. Responsiveness: No evidence found for patients with MF or SS. | Not identified in populations with MF or SS. |
Pruritis Likert scale | An 11-point NRS asking patients to rank their level of itch from 0 = no itch to 10 = worst itch imaginable. | Validity: Moderate concurrent validity and strong convergent validity for patients with chronic pruritus. Reliability: Acceptable test–retest reliability for patients with chronic pruritus. Responsiveness: Limited evidence of responsiveness for patients with chronic pruritus. | Not identified in populations with MF or SS. |
ItchyQoL | A 22-item instrument for assessing the impact of pruritus on HRQoL consisting of 3 domains (symptoms, functioning, and emotions) and scored on a 5-point Likert scale. A higher score indicates worse HRQoL. | Validity: Evidence of face and content validity. Evidence of strong construct and concurrent validity for patients with chronic pruritus. Reliability: Acceptable internal consistency and test–retest reliability for patients with chronic pruritus. Responsiveness: Some evidence of responsiveness for patients who reported improvement of pruritus. | Not identified in populations with MF or SS. |
BSA = body surface area; MID = minimal important difference; NRS = numeric rating scale.
Modified Severity-Weighted Assessment Tool
The mSWAT is used to assess the body surface area (BSA) of each lesion type (patch, plaque, and tumour) for each of the 12 body regions (listed in Table 50).31 The patient’s palm and fingers are considered to equal approximately 1% BSA. The sum of the BSA of each lesion type is multiplied by a weighting factor (1 for patches, 2 for plaques, and 4 for tumours). The total mSWAT score is the sum of the final weighted lesion BSA scores.27 Scores can be tracked over time and a lower score indicates less active disease, a stable score indicates stable disease, and a higher score indicates more active disease.48
The ISCL, the United States Cutaneous Lymphoma Consortium, and the Cutaneous Lymphoma Task Force of the EORTC published a consensus statement for clinical end points and response criteria for the treatment of MF and SS.31 The statement consists of recommendations for conducting clinical trials of MF and SS as well as a guidance for assessing disease severity and response in patients with MF and SS. The statement acknowledges the use of the SWAT or mSWAT for skin assessment and notes that there has been debate about the appropriateness of the weighting factor used for tumours considering their importance in prognosis. The authors discuss that the impact of tumours (both thickness of dermal infiltrate and proportion of neoplastic cells) is greater than 4 times that of patches. As a result, changes in tumours compared with other lesions may be underrepresented in the final SWAT or mSWAT score. The authors also note that investigator assessment of plaques versus tumours can be variable which further complicates the scoring method.
Table 50: The Modified Severity-Weighted Assessment Tool
Body region (% BSA) | Assessment of involvement: patch | Assessment of involvement: plaque | Assessment of involvement: tumour |
|---|---|---|---|
Head (7) | NA | NA | NA |
Neck (2) | NA | NA | NA |
Anterior trunk (13) | NA | NA | NA |
Arms (8) | NA | NA | NA |
Forearms (6) | NA | NA | NA |
Hands (5) | NA | NA | NA |
Posterior trunk (13) | NA | NA | NA |
Buttocks (5) | NA | NA | NA |
Thighs (19) | NA | NA | NA |
Legs (14) | NA | NA | NA |
Feet (7) | NA | NA | NA |
Groin (1) | NA | NA | NA |
Subtotal of lesion BSA | NA | NA | NA |
Weighting factor | x1 | x2 | x4 |
Subtotal of lesion BSA x weighting factor | NA | NA | NA |
BSA = body surface area; NA = not applicable.
Source: MAVORIC (0761 to 010) Clinical Study Report.27
There is some evidence of validity for the original SWAT in patients with MF (N = 323) showing correlation between SWAT score and total BSA (r = 0.95) as well as between SWAT score and physician global assessment (r = 0.60).49 No literature was found that assessed the mSWAT for validity, reliability, or responsiveness in patients with MF or SS.
No MID for the mSWAT was identified in patients with MF or SS.
Global Composite Response Score
Global response has been defined in the consensus statement by the ISCL, the United States Cutaneous Lymphoma Consortium, and the Cutaneous Lymphoma Task Force of the EORTC.31 The statement includes guidance for assessing skin, lymph nodes, viscera, and blood with definitions of response for each. Furthermore, the authors note that patients with a global objective response should have at least a PR in the skin when assessing MF and SS.
Skindex-29
The Skindex-29 is a 29-item instrument for measuring how skin conditions affect HRQoL.50 It has a 4-week recall period and has been translated into other languages. Items fall under 3 domains: emotions (10 items), symptoms (7 items), and functioning (12 items). Each item is scored on a 5-point Likert scale which is transformed to a linear numerical scale (never = 0, rarely = 25, sometimes = 50, often = 75, and all the time = 100). The scale score is calculated from the mean of a patient’s scores for a scale or domain, while the composite score is the mean of the 3 scale scores. A higher score indicates worse HRQoL.
Using mixture analysis methods, it has been suggested that the symptoms scale score be interpreted according to 5 categories: ≤ 3 = very little effect, 4 to 10 = mild effect, 11 to 25 = moderate effect; 26 to 49 = severe effect, and ≥ 50 = extremely severe effect.50 The thresholds indicating severe effect on HRQoL for each of the scales was estimated using anchor-based methods to be ≥ 39 for emotions, ≥ 52 for symptoms, and ≥ 37 for functioning.50
The original Skindex-61 was refined to the Skindex-29 and was assessed for validity, reliability, and responsiveness in a sample of adults with various skin conditions attending private practices in Ohio.51 Based on a sample of 692 patients, Chren et al. reported evidence of construct and content validity as demonstrated through patients with chronic skin conditions having higher scores than patients with isolated lesions, using principal axes factor analysis and correlating 3 factors to a priori scales, and having instrument items that addressed most patients’ concerns about their skin condition. Reliability was acceptable for each scale score (Cronbach alpha = 0.94, 0.87, and 0.96 for the emotions, symptoms, and functioning domains, respectively). Reproducibility was also high for each scale score (Pearson correlation coefficient = 0.88, 0.91, and 0.92 for the emotions, symptoms, and functioning domains, respectively) between patients’ initial response to the questionnaire and second response 72 hours later (N = 105). Responsiveness to change was assessed for 508 patients who completed the questionnaire initially and again 3 months later and were categorized based on their self-reported condition being worse, the same, or improved. The probability that the scale scores did not differ at the 2 time points was statistically significant for those who reported their condition was worse (n = 36, P < 0.01) and those who reported their condition was improved (n = 330, P < 0.001). For patients whose condition was unchanged, the results were not statistically significant (n = 142, P > 0.01).
A study of 92 adults with cutaneous lymphomas (53 with MF and 2 with SS) in Austria used the validated German version of the Skindex-29 to assess HRQoL.52 Internal consistency for the overall group of patients with cutaneous lymphomas was acceptable for the overall Skindex-29 score (Cronbach alpha = 0.958) and for each scale score (Cronbach alpha = 0.926, 0.817, and 0.924 for the emotions, symptoms, and functioning domains, respectively). Separate results for only patients with MF or SS were not reported. A separate study of adults with MF or SS (N = 22) reported acceptable face validity and internal consistency reliability for each subscale of the Skindex-29 (Cronbach alpha = 0.95, 0.89, and 0.94 for emotions, symptoms, and functioning domains, respectively).53
No MID was identified in populations with MF or SS.
Functional Assessment of Cancer Therapy–General
The FACT-G is a 27-item instrument for assessing HRQoL in patients with cancer and has a 7-day recall period.27 Items fall under 4 domains: physical well-being (PWB; 7 items), social/family well-being (SWB; 7 items), emotional well-being (EWB; 6 items), and functional well-being (FWB; 7 items).54 Each item is scored on a 5-point Likert scale where 0 = not at all, 1 = a little bit, 2 = somewhat, 3 = quite a bit, and 4 = very much. Response scores for negatively phrased questions are reverse scored after which a subscale score can be computed, and a total score is calculated from the sum of the 4 individual subscale scores (total range = 0 to 108). A higher score indicates better HRQoL.
Yost et al. assessed the psychometric properties of the FACT-G for adult patients with non-Hodgkin lymphoma who were participating in the Molecular Epidemiology Resource study.54 Concurrent validity of the FACT-G was investigated by calculating its correlation with other instruments: Brief Profile of Mood States (B-POMS), the “state” component of the State-Trait Anxiety Inventory (STAI), patient-reported ECOG Performance Status. and linear analogue scale assessment (LASA). Correlations were moderate and in the expected directions: B-POMS (r = −0.63, n = 201), STAI (r = −0.57, n = 226), ECOG PS (r = −0.43, n = 177), and LASA QoL (r = 0.63, n = 229). For assessing reliability, the authors reported acceptable internal consistency (Cronbach alpha > 0.7) for the total score as well as each subscale and was highest with the total score and FWB subscale and lowest with the EWB subscale (specific Cronbach alpha values were not reported). The FACT-G demonstrated good responsiveness for the total scale, PWB, and FWB scores among patients who showed change in ECOG PS and LASA QoL. The EWB and SWB subscales did not show good sensitivity which the authors suggested may be due to dissimilarity in constructs that the instruments measure or possible ceiling effects that were observed in this population. No literature was found that assessed the FACT-G for validity, reliability, or responsiveness in patients with MF or SS.
MIDs have been estimated to be 3 to 7 points55 for the total score and 2 to 3 points55,56 for each of the subscale scores in populations with different types of cancers. No MID was identified in populations with MF or SS.
EQ-5D-3L
The EQ-5D-3L is a generic HRQoL instrument consisting of a descriptive system and a VAS.27 Items fall under 5 domains (mobility, self-care, usual activities, pain/discomfort, and anxiety/depression) and are answered based on 3 levels (no problems, some problems, or extreme problems).
The EQ-5D index score is generated by applying a multi-attribute utility function to the descriptive system.57 Different utility functions are available that reflect the preferences of specific populations (e.g., US or UK). The lowest possible overall score for the 3L version (corresponding to severe problems on all 5 attributes) varies depending on the utility function that is applied to the descriptive system (e.g., −0.59 for the UK algorithm and −0.109 for the US algorithm). Scores less than 0 represent health states that are valued by society as being worse than dead, while scores of 0 and 1.00 are assigned to the health states “dead” and “perfect health,” respectively.
Teckle et al. conducted a study of patients with cancer (N = 184) at the Vancouver Cancer Clinic.58 There was evidence of moderate concurrent validity between the EQ-5D-3L and EORTC QLQ-C30 as well as between the EQ-5D-3L and EQ-VAS (Pearson correlation coefficient = 0.43 for both) and strong concurrent validity between the EQ-VAS and EORTC QLQ-C30 (Pearson correlation coefficient = 0.73). The EQ-5D-3L demonstrated acceptable internal consistency for all 5 functioning scales along with global health status (Cronbach alpha ranged from 0.77 to 0.82). No literature was found that assessed the EQ-5D-3L for validity, reliability, or responsiveness in patients with MF or SS.
Pickard et al. conducted a retrospective analysis of 534 patients with cancer to estimate a MID using distribution-based (SEM, one-half SD, and one-third SD) and anchor-based (ECOG PS) methods.59 After stratifying by ECOG PS, the mean weighted index score MID for all cancer patients was estimated to be between 0.07 and 0.11 for UK-index scores and between 0.05 and 0.08 for US-index scores. The VAS MID was estimated to range from 6 to 11 points for all patients with cancer. Sinnott et al. reported an estimated MID range of 0.033 to 0.074 for the index score from patients with a variety of conditions.57 No MID was identified in populations with MF or SS.
Pruritis Likert Scale
The Pruritus Likert Scale is an 11-point numeric rating scale (NRS) where patients are asked to rank their level of itch from 0 = no itch to 10 = worst itch imaginable.27
A study of 419 Korean adults with chronic pruritus (of various etiologies) for at least 6 weeks compared several itch-related scales (NRS, VAS, Verbal Rating Scale, and Itch Severity Scale) for validity and reliability.60 Jang et al. found evidence of moderate concurrent validity between the NRS and the ItchyQoL (Spearman correlation coefficient = 0.49 and 0.48 for initial and follow-up assessments, respectively). Good test–retest reliability was demonstrated between the initial NRS assessment and follow-up 3 hours later (intraclass correlation coefficient = 0.77). The NRS has also shown good test–retest reliability in another study of 250 adults with chronic pruritus between the initial assessment and when it was repeated 1 hour later (intraclass correlation coefficient = 0.80).61
A systematic review of patient-reported outcome measures for pruritus by Schoch et al. found evidence of convergent validity between the 11-point NRS and the horizontal VAS (r > 0.83) and verbal NRS-4 (r > 0.73) as well as adequate test–retest reliability (intraclass correlation coefficient = 0.76 to 0.86).62 The authors also found limited evidence for responsiveness. No literature was found that assessed the Pruritus Likert Scale for validity, reliability, or responsiveness in patients with MF or SS.
No MID was identified in populations with MF or SS.
Itchy Quality of Life
The ItchyQoL is a 22-item instrument for assessing the impact of pruritus on HRQoL and has a 1-week recall period for the first 15 questions and a 4-week recall period for the following 7 questions.27 Items fall under 3 domains (symptoms, functioning, and emotions) and are scored on a 5-point Likert scale (1 = never, 2 = rarely, 3 = sometimes, 4 = often, and 5 = always).60 The subscale score is the mean of the patient’s scores for a subscale, while the overall score is the mean of the scores of all items.27 A higher score indicates worse HRQoL.
During the development process of the ItchyQoL, Desai et al. tested the instrument’s psychometric properties and compared it to the Skindex-29 and Skindex-16 which were used to address frequency and bother, respectively.63 The study included adults attending a dermatology clinic who had active pruritus in the past 7 days and who answered the questionnaires at baseline, after 72 hours for test–retest reliability, and again at 2 months for instrument responsiveness. Face and content validity was assessed through interviews with patients with pruritus. The authors hypothesized that the bother and frequency items would cluster as 3 domains (symptoms, functioning, and emotions) which were tested by principal axis factor analysis methods and regression factor scores were compared with hypothesized subscale scores using Pearson correlation coefficients. Both frequency and bother items demonstrated strong construct validity (Pearson correlation coefficients ranged from 0.52 to 0.77 and from 0.86 to 0.93, respectively). The authors assessed discriminant validity by comparing the floor effect between the ItchyQoL and the Skindex-29 or Skindex-16 and found that the ItchyQoL had fewer insensitive items (i.e., fewer questions to which > 50% of patients responded “never”). The ItchyQoL overall score demonstrated acceptable internal consistency reliability (Cronbach alpha = 0.89 for bother and 0.92 for frequency) as well as good test–retest reliability (intraclass correlation coefficient = 0.87 for bother and 0.92 for frequency). Responsiveness was assessed using paired t-tests for patients who reported their condition to be improved, unchanged, or worsened after 2 months. Results showed that the ItchyQoL overall score was sensitive to patients who reported improvement, but not sensitive to patients who reported no change or worse condition though the authors acknowledged the limitation of testing responsiveness in a small number of patients (n = 23 for bother-type questions and n = 32 for frequency-type questions).
The ItchyQoL has demonstrated strong concurrent validity when compared with other skin- and pruritus-related instruments such as the Dermatological Life Quality Index (r = 0.7264 to 0.74565) and moderate concurrent validity with the NRS (r = 0.5164). Many studies have demonstrated acceptable internal consistency (Cronbach alpha ranging from 0.7662 to 0.9564) for the overall score and for each of the 3 domain scores (Cronbach alpha = 0.893, 0.814, and 0.89365 for emotions, symptoms, and functioning domains, respectively) for patients with chronic pruritus. Additionally, test–retest reliability was acceptable (intraclass correlation coefficient ranging from 0.87 to 0.92).62,64 There was limited evidence for responsiveness to change on the ItchyQoL with change in VAS (r = 0.35 to 0.46).62 No literature was found that assessed the Pruritus Likert Scale for validity, reliability, or responsiveness in patients with MF or SS.
No MID was identified in populations with MF or SS.
Pharmacoeconomic Review
Abbreviations
aSCT
allogeneic stem-cell transplant
CHOP
cyclophosphamide, doxorubicin, vincristine, and prednisolone
ECM
established clinical management
ICER
incremental cost-effectiveness ratio
IPCW
inverse probability of censoring weighting
MAIC
matching-adjusted indirect comparison
OS
overall survival
PFS
progression-free survival
PSM
partition survival model
QALY
quality-adjusted life-year
QoL
quality of life
TSE
2-stage estimation
Executive Summary
The executive summary comprises 2 tables (Table 1 and Table 2) and a conclusion.
Item | Description |
|---|---|
Drug product | Mogamulizumab (Poteligeo), vial for IV infusion (4 mg/mL), 20 mg single-use vial |
Submitted price | Mogamulizumab, 20 mg, IV infusion: $2,203.60 |
Indication | For the treatment of adult patients with mycosis fungoides or Sézary syndrome who have received at least 1 prior systemic therapy |
Reimbursement request | As per indication |
Health Canada approval status | NOC |
Health Canada review pathway | Standard |
NOC date | June 2, 2022 |
Sponsor | Kyowa Kirin Canada |
Submission history | Previously reviewed: No |
NOC = Notice of Compliance.
Table 2: Summary of Economic Evaluation
Component | Description |
|---|---|
Type of economic evaluation |
|
Target population | Adult patients with mycosis fungoides or Sézary syndrome who have previously received ≥ 1 systemic therapy |
Treatment | Mogamulizumab |
Comparator | ECM consisting of methotrexate, bexarotene, interferon alpha-2a, gemcitabine, CHOP, liposomal doxorubicin, etoposide, prednisolone, vorinostat, PUVA, extracorporeal phototherapy, total skin electron beam therapy, chlorambucil, purine analogues, pralatrexate, romidepsin, and alemtuzumab |
Perspective | Canadian publicly funded health care payer |
Outcomes | QALYs, LYs |
Time horizon | Lifetime (30 years) |
Key data source | MAVORIC |
Submitted results | ICER = $96,742 per QALY (incremental costs = $223,353; incremental QALYs = 2.31) vs. ECM |
Key limitations |
|
CADTH reanalysis results |
|
CHOP = cyclophosphamide, doxorubicin, vincristine, and prednisone; ECM = established clinical management; ICER = incremental cost-effectiveness ratio; LY = life-year; MAIC = matching-adjusted indirect comparison; OS = overall survival; PSM = partitioned survival model; PUVA = psoralen plus UVA phototherapy; QALY = quality-adjusted life-year; TSEBT = total skin electron beam therapy; vs. = versus.
Conclusions
The CADTH review of the clinical evidence suggests that mogamulizumab may improve progression-free survival (PFS) compared with vorinostat among patients with mycosis fungoides and Sézary syndrome who have received at least 1 prior systemic treatment; however, the pivotal clinical trial (MAVORIC) was not powered to detect differences in overall survival (OS), and interpretation of the OS data is confounded by the crossover of patients between treatment arms. The comparator in the MAVORIC trial, vorinostat, is not standard of care in Canada, and it is highly uncertain whether the effectiveness of established clinical management (ECM) would be equal to that of vorinostat. As a result, the comparative effectiveness of mogamulizumab is highly uncertain.
The sponsor submitted a model comparing the cost-effectiveness of mogamulizumab with ECM and reported a base-case incremental cost-effectiveness ratio (ICER) of $96,742 per quality-adjusted life-year (QALY). This result suggests that, even under the sponsor’s optimistic assumptions, the probability that mogamulizumab is cost-effective is 0% at a willingness-to-pay threshold of $50,000.
CADTH was not able to conduct a reanalysis due to critical limitations within the sponsor’s model and submitted evidence. Notably, the choice of a partition survival model (PSM) was inappropriate, given that the OS data are highly uncertain. The sponsor’s model predicts that 81% of the incremental benefit with mogamulizumab would be obtained after discontinuation of mogamulizumab by patients receiving subsequent treatments, which is not supported by the clinical evidence. The sponsor’s definition of ECM is not in line with Canadian clinical practice; therefore, there is uncertainty as to whether the incremental costs associated with mogamulizumab have been captured accurately. CADTH was unable to address these critical limitations (i.e., the apparent post-progression survival benefit, ECM costs, and effectiveness) due to constraints introduced by the submitted model structure and a lack of clinical data.
Due to the methodological limitations identified within the model and the highly uncertain comparative effectiveness data, the cost-effectiveness of mogamulizumab is unknown.
Stakeholder Input Relevant to the Economic Review
This section is a summary of the feedback received from the patient groups, registered clinicians, and drug plans that participated in the CADTH review process (specifically, information that pertains to the economic submission).
Patient input was received from Lymphoma Canada from 449 patients (10% from Canada) with mycosis fungoides or Sézary syndrome collected via an online survey. Patients described how the symptoms of mycosis fungoides and Sézary syndrome affect their quality of life (QoL), including the presence of raised, scaly, or discoloured patches; itchiness; rash-like skin redness; a feeling of pain or burning; thick raised lesions; fatigue; hair loss; and loss of sleep. Respondents also described stress, fear and worry, anxiety, and concerns with their physical appearance. Patients indicated there are many treatment options available with a wide variety of effectiveness and noted that treatments become less effective over time. Patients additionally noted that many currently available treatments are used in combination. Some patients reported that the number of clinic visits required for treatment negatively affects their QoL as well as their ability to work, travel, exercise, and maintain relationships. Some respondents from Canada described being unable to access treatment locally, which increased travel time, worry, and emotional distress. Adverse events associated with available treatments were reported to include fatigue, skin pain or burning, skin irritation or rash, itching, hair loss, skin discolouration, and peripheral neuropathy. Patients expressed a desire for a treatment that improves survival and QoL, allows for longer treatment-free periods, is easier to administer, and has fewer adverse events. Of the respondents with mogamulizumab experience, some noted that the number of hospital visits required for mogamulizumab treatment had a negative impact on their QoL, and some described discontinuing mogamulizumab because of adverse events (e.g., fatigue or weakness, skin rash or redness, hair loss, neutropenia); others noted that mogamulizumab had a positive impact on their QoL.
The clinician input received from the Canadian Cutaneous Lymphoma Providers and the Ontario Health Hematology Cancer Drug Advisory Committee noted that the treatment of mycosis fungoides and Sézary syndrome is individualized, based on extent of disease or stage, goals of therapy, patient tolerance, and practical considerations related to administration. Clinicians noted that systemic treatment in Canada includes pegylated interferon, isotretinoin, and methotrexate. Bexarotene was noted to be the standard of care outside of Canada but is not approved by Health Canada. Other treatment options include chemotherapy, alemtuzumab, pralatrexate, brentuximab vedotin, and vorinostat, some of which are not publicly reimbursed in Canada. Clinicians noted that total skin electron beam therapy may be used for the indicated population, but that access is severely limited. Clinicians noted that select patients with advanced-stage mycosis fungoides or Sézary syndrome may undergo an allogenic stem-cell transplant (aSCT). The goal of treatment for mycosis fungoides and Sézary syndrome is prolonged survival, overall response, improvement in skin-related symptoms, and QoL; however, the experts noted that some patients may continue to experience skin symptoms or reduced QoL or function despite improved objective parameters. The clinicians noted that mogamulizumab may be considered after chemotherapy, brentuximab (for patients with CD30-positive disease), or interferon (for those with advanced mycosis fungoides), or after interferon and/or isotretinoin for those with Sézary syndrome. Finally, the clinicians noted that mogamulizumab would be most suited for the treatment of advanced-stage mycosis fungoides with or without blood involvement (stage IIIB, IVA, IVB), or Sézary syndrome, and that mogamulizumab should not be considered for those with stage IA mycosis fungoides.
The CADTH-participating drug plans noted that the active comparator in the MAVORIC trial, vorinostat, is not publicly funded in Canada. The drug plans noted there is no standard of care for mycosis fungoides or Sézary syndrome in patients who have progressed after prior systemic therapy. Options for patients with relapsed disease, aggressive disease, or those with extracutaneous involvement include isotretinoin or alitretinoin, interferon, bexarotene, alemtuzumab, single-drug chemotherapy (methotrexate, gemcitabine, liposomal doxorubicin), or multi-drug chemotherapy (e.g., cyclophosphamide, doxorubicin, vincristine, and prednisolone [CHOP]). The plans noted that public funding of treatments varies across provinces and that patients may require several types of treatment and repeated courses of therapy. The drug plans indicated that brentuximab vedotin is funded in some provinces for the treatment of CD30-positive mycosis fungoides after prior systemic therapy. The drug plans also noted that patients in MAVORIC were highly pre-treated, while the requested reimbursement population comprises those with as little as 1 prior systemic treatment. The drug plans described concerns about infusion reactions and rashes, which may require additional monitoring and/or treatment.
Several of these concerns were addressed in the sponsor’s model:
PFS was incorporated into the model. The use of a cost-utility approach accounts for some issues related to QoL; however, the EuroQoL 5-Dimensions questionnaire (EQ-5D) is unlikely to capture all symptoms of mycosis fungoides and Sézary syndrome that were noted by the patients to affect QoL.
Costs related to the treatment of grade 3 or higher adverse events that occurred in at least 5% of trial participants were included.
CADTH was unable to address the following concerns raised in the stakeholder input:
The cost-effectiveness of mogamulizumab was compared with ECM, which was assumed to be a basket of treatments, some of which are not available or publicly reimbursed. The cost-effectiveness of mogamulizumab relative to any individual comparator is unknown.
The adverse events included in the sponsor’s model do not capture all adverse events deemed to be important to patients (e.g., hair loss).
The sponsor’s base case jointly considers mycosis fungoides and Sézary syndrome. While the sponsor provided scenario analyses that considered these populations separately, the composition of the ECM comparator did not change.
Economic Review
The current review is for mogamulizumab (Poteligeo) for the treatment of mycosis fungoides or Sézary syndrome among adults who have received at least 1 prior systemic treatment.1
Economic Evaluation
Summary of Sponsor’s Economic Evaluation
Overview
The proposed indication for mogamulizumab is for the treatment of mycosis fungoides or Sézary syndrome among adults who have received at least 1 prior systemic treatment.2 The sponsor submitted a cost-utility analysis of mogamulizumab compared with ECM, which was conceptualized as a basket of treatments (Table 9).1 The modelled population is consistent with the reimbursement request, i.e., patients with either mycosis fungoides or Sézary syndrome, and is based on the population of the MAVORIC trial, a phase III randomized controlled trial comparing mogamulizumab with vorinostat. The sponsor submitted scenario analyses to explore the cost-effectiveness of mogamulizumab in subgroups of patients with mycosis fungoides or Sézary syndrome.
Mogamulizumab is available as a 4 mg/mL solution for IV infusion at a submitted price of $2,203.60 per 20 mg vial. The proposed dosing regimen for mogamulizumab is 1 mg/kg administered on days 1, 8, 15, and 22 of the first 28-day cycle, and on days 1 and 15 of each subsequent 28-day cycle, until “disease progression or unacceptable toxicity.”2 The sponsor’s calculated cost, including wastage, for mogamulizumab is $8,703.43 per administration ($34,814 for the first cycle; $17,407 for subsequent cycles). The sponsor did not state the expected cost of ECM, and it was not directly calculable based on the structure of the sponsor’s pharmacoeconomic model. The treatments included in the ECM basket, including the dosage, unit price, and frequency of use assumed by the sponsor, are provided in Table 9. Based on these inputs, CADTH estimates the annual per-patient cost of ECM to be approximately $45,676.
The clinical outcomes of interest were QALYs and life-years. The economic analysis was undertaken over a 30-year horizon from the perspective of a publicly funded health care payer. Costs and outcomes were discounted at a rate of 1.5% annually.
Model Structure
The sponsor submitted a PSM that separately modelled the trajectory of patients assumed to undergo an allogeneic stem-cell transplant (aSCT) and those who do not undergo an aSCT. The inclusion of aSCT led to the modelling of 3 separate patient pathways that collectively reflect the model population: patients who do not undergo an ask (Figure 1), patients who undergo an aSCT after initial treatment with mogamulizumab or ECM (Figure 2), and patients who undergo an aSCT after subsequent treatment (Figure 3). For patients who do not undergo an aSCT, the model included the following health states: pre-progression (on or off treatment), post-progression (on or off subsequent treatment), and death. For patients who undergo an aSCT (after initial or subsequent treatment), the model included additional health states related to post-transplant disease status (disease-free, relapsed disease). Generally, patients entered the model in the pre-progression state on treatment with mogamulizumab or ECM. After the first 1-week cycle, patients could remain in the pre-progression state on their initial treatment (on treatment), discontinue treatment but remain progression-free (off treatment), experience disease progression, or die. Patients who experience disease progression while receiving mogamulizumab or ECM move to the post-progression state, where they can receive subsequent treatment or end-stage care, remain off treatment, or die. Patients who undergo an aSCT after initial treatment or subsequent treatment enter the aSCT “after current treatment” state or the aSCT “after subsequent treatment” state, respectively, where patients can remain disease-free or experience a disease relapse.
The movement of patients through the model was based on PFS, OS, time on treatment, next-treatment-free survival and, for patients who underwent an aSCT, disease-free survival and OS post transplant. For patients who do not undergo an aSCT, the proportion of patients who received subsequent treatment after disease progression on mogamulizumab or ECM was derived as the difference between the OS and next-treatment-free survival curves, while the proportion of patients with progressed disease who remained off treatment was derived as the difference between the next-treatment-free survival and PFS curves. Patients were assumed to receive end-stage care for 6 months before death, and the weekly probability of death was assumed to be no less than the age- and sex-equivalent portions of the general population.
Model Inputs
The modelled cohort’s characteristics were based on the MAVORIC trial (mean age of 63 years; body surface area if 1.91 m2, mean weight of 76.77 kg); however, CADTH notes that the mean weight in the pharmacoeconomic submission (76.77 kg) differs from that reported in the Clinical Study Report (78.96 kg). Separate data sources were used to inform the model for patients assumed to undergo an aSCT versus those assumed to never undergo an aSCT. The source of the clinical data and parametric functions adopted by the sponsor are summarized in Table 10. For patients who do not undergo an aSCT, data were obtained from the MAVORIC trial for PFS, OS, time on treatment, next-treatment-free survival, and adverse events for mogamulizumab. The sponsor assumed that effectiveness and adverse events for ECM would be equivalent to that observed for vorinostat in the MAVORIC trial (i.e., vorinostat data from MAVORIC were used in the model as a proxy for ECM). Kaplan-Meier estimates of OS, PFS, and next-treatment-free survival from MAVORIC were used to fit parametric survival curves to extrapolate the observed trial data (median follow-up of 17 months) over the entire model time horizon (30 years), with model selection based on statistical fit (Akaike Information Criterion [AIC], Bayesian Information Criterion [BIC]). OS data for the vorinostat arm of the MAVORIC trial were adjusted for crossover to the mogamulizumab arm by inverse probability of censoring weighting (IPCW).
For patients assumed to undergo an aSCT, data pertaining to disease-free survival and OS were based on digitized Kaplan-Meier curves from the National Institute for Health and Care Excellence (NICE) appraisal of brentuximab vedotin3 for patients who received minimal intensity management after an aSCT in the UK (median follow-up of approximately 60 months); these data were extrapolated over the 30-year model horizon. The sponsor assumed no difference in OS or disease-free survival based on the initial treatment received. The sponsor assumed that no patients would undergo an aSCT after treatment with mogamulizumab, while the proportion of patients assumed to undergo an aSCT after ECM (4.6%) was based on a survey of clinicians undertaken by the sponsor. The proportion of patients assumed to receive an aSCT after subsequent treatment was based on observations from the MAVORIC trial.
Health-state utility values were based on EQ-5D data collected during the MAVORIC trial and valued using UK tariffs. Pre-progression values were analyzed using longitudinal mixed models, and post-baseline utility values were regressed on fixed effects of baseline EQ-5D utility score, treatment group, and histology. The sponsor assumed that the health-state utility for patients in the post-progression state (either on or off treatment) would be equal to the last observed utility value after disease progression for patients in the mogamulizumab arm of MAVORIC. Disutilities were included for grade 3 or higher adverse events experienced by at least 5% of patients in MAVORIC for mogamulizumab or vorinostat (as a proxy for ECM). Adverse events were assumed to occur only in the pre-progression health state for patients on treatment and were assumed to last for 7 days. Disutility values were obtained from the literature.
The model included costs related to the acquisition of drugs and other treatment modalities, administration of chemotherapy drugs, the cost of aSCT, adverse events, health care resource use, and end-stage care. The cost of mogamulizumab acquisition was based on the sponsor’s submitted price, patient weight distribution in the MAVORIC trial, and dose banding to account for drug wastage.1 ECM was assumed to be a basket of treatments, with the components and frequency of use of each based on clinician input (Table 9). Subsequent treatment was assumed to be a basket of treatments, with the composition, distribution of treatments, and length of treatment based on clinician input. The costs of the components of ECM and subsequent treatment were obtained from the Ontario Drug Benefit Formulary,4 Alberta Drug Benefit List,5 Ontario Schedule of Benefits,6 Health Quality Ontario,7 ClaimSecure Drug Review, and from prior CADTH reviews. Administration costs were included for chemotherapy drugs.6,8 The cost of aSCT was obtained from Ontario’s Interprovincial Billing Rates for Designated High Cost Transplants.9 For patients with relapsed disease after an aSCT, the cost of subsequent treatment was assumed to be 41% of the cost of subsequent treatment for patients without an aSCT.3 Routine-care costs were assumed to be equal for patients with mycosis fungoides or Sézary syndrome and were based on a retrospective review of costs in the UK’s National Health Service.1 The costs associated with adverse events were obtained from the Ontario Case Costing Initiative,10 Alberta Health,11 and the literature.12 The cost of end-stage care for patients with lymphoma was obtained from the literature.13
Summary of Sponsor’s Economic Evaluation Results
All analyses were run probabilistically (5,000 iterations for the base-case and scenario analyses). The deterministic and probabilistic results were similar. The probabilistic findings are presented subsequently. Additional results from the sponsor’s submitted economic evaluation base case are presented in Appendix 3.
Base-Case Results
In the sponsor’s base-case analysis, mogamulizumab was associated with estimated costs of $419,654 and 4.96 QALYs over a 30-year time horizon. Treatment with mogamulizumab was both more costly and produced more QALYs than treatment with ECM (incremental costs: $223,353; incremental QALYs: 2.31), resulting in an ICER of $96,742 per QALY (Table 3). In the sponsor’s base case, mogamulizumab had a 0% probability of being the most cost-effective strategy at a willingness-to-pay threshold of $50,000 per QALY.
Results were driven by the predicted differences in total life-years between mogamulizumab and ECM (incremental life-years: 3.51 years), the continued gain of QALYs after disease progression (incremental QALYs post-progression: 1.88), and the increased drug acquisition costs associated with mogamulizumab (incremental costs: $196,761) (Appendix 3). In the sponsor’s base case in the mogamulizumab arm, 9% of patients who underwent an aSCT and 5% of patients who did not undergo an aSCT remained alive at the end of the 30-year horizon; in the ECM arm, 9% of patients who underwent an aSCT and 0% of patients who did not undergo an aSCT remained alive.
Table 3: Summary of the Sponsor’s Economic Evaluation Results
Drug | Total costs ($) | Incremental costs ($) | Total QALYs | Incremental QALYs | ICER vs. ECM ($/QALY) |
|---|---|---|---|---|---|
ECM | 196,301 | Reference | 2.65 | Reference | Reference |
Mogamulizumab | 419,654 | 223,353 | 4.96 | 2.31 | 96,742 |
ECM = established clinical management; ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year; vs. = versus.
Source: Sponsor’s pharmacoeconomic submission.1
Sensitivity and Scenario Analysis Results
The sponsor provided several scenario and sensitivity analyses, including:
adopting alternative time horizons
adopting alternative discount rates
assuming ECM is comprised entirely of pralatrexate (assumed to affect only costs)
adjusting for relative dose intensity (assumed to affect only costs)
adopting an alternative method to adjust OS estimates for crossover between treatment groups in the MAVORIC trial
applying hazard ratios to model the relationship between mogamulizumab and ECM for time on treatment, PFS, and OS
adopting an alternative model structure based on next-treatment-free survival, and
exploring the cost-effectiveness of mogamulizumab in patient subgroups (e.g., mycosis fungoides, Sézary syndrome).
Several scenarios resulted in notable increases to the ICER, including adopting alternative time horizons and assuming use in specific patient subgroups. Notably, the sponsor’s model was highly sensitive to the time horizon, with an ICER of $356,394 for mogamulizumab versus ECM when the analysis was limited to a 5-year horizon. The sponsor’s estimated ICER varied by patient subgroup: compared with the ICER for the full Health Canada indication, the ICER was higher for patients with Sézary syndrome ($159,509 per QALY) and among patients with advanced disease ($117,956), and lower for patients with mycosis fungoides ($70,377).
CADTH Appraisal of the Sponsor’s Economic Evaluation
CADTH identified several key limitations to the sponsor’s analysis that have notable implications for the economic analysis:
The comparative clinical efficacy of mogamulizumab versus ECM is highly uncertain. There has been no head-to-head trial of mogamulizumab and ECM. To inform the pharmacoeconomic model, the sponsor assumed that the effectiveness of ECM would be equal to that observed for vorinostat in the MAVORIC trial. The sponsor submitted a matching-adjusted indirect comparison (MAIC) between the vorinostat arm of MAVORIC and the “physician’s choice” arm of ALCANZA to support the assumption that ECM and vorinostat would be equally effective. Importantly, the results of the MAIC were not used in the sponsor’s model, and efficacy was based solely on the MAVORIC trial. The physician’s choice arm of ALCANZA comprised bexarotene (59%) and methotrexate (41%). Bexarotene is available through special access drug programs in some jurisdictions in Canada and, as noted by the clinical experts consulted for this review, is rarely prescribed, limiting the relevance of the results to clinical practice in Canada. As noted in the CADTH Clinical Review Report, there were several key differences between MAVORIC and ALCANZA that impact the comparability of studies (e.g., inclusion criteria, baseline characteristics [CD30 status, disease stage, blood involvement, prior treatments], and study treatments). Owing to such limitations, the results of the sponsor’s MAIC are considered uncertain, and it is highly uncertain whether the effectiveness of ECM is equal to that of vorinostat.
Owing to the limitations of the sponsor’s MAIC and the paucity of direct evidence, the cost-effectiveness of mogamulizumab relative to ECM is unknown.
The impact of the crossover adjustment on OS is uncertain. In the MAVORIC trial, approximately 73% of patients randomized to receive vorinostat were permitted to cross over to mogamulizumab, which confounds the interpretation of OS data from the trial. The sponsor attempted to address this through adjustment of the OS data via 3 methods: IPCW, 2-stage estimation (TSE), and rank-preserving structural failure time (RPSFT). These analyses were not transparently described by the sponsor. In its base case, the sponsor adopted the IPCW method. CADTH notes that IPCW assumes there are no unmeasured confounders and that it is important that the proportion of patients who were eligible to switch treatments but who did not switch is not too low; otherwise, large weights may be obtained for a small number of patients. This proportion, as well as the resulting weights, was not provided by the sponsor. CADTH further notes that, following IPCW adjustment, there was no significant difference in OS between the patients who received mogamulizumab versus vorinostat (hazard ratio [HR] = 0.45; 95% confidence interval [CI], 0.19 to 1.05). The finding of no difference between mogamulizumab and vorinostat was replicated by the sponsor using the TSE method (HR = 0.78; 95% CI, 0.51 to 1.19). No results were presented for the RPSFT method. The uncertainty associated with the choice of method for crossover adjustment is highlighted by the sponsor’s submitted scenario analyses, in which using the TSE method resulted in a considerably lower estimate of the incremental life-years gained with mogamulizumab (TSE = 2.97; IPCW = 3.51).
Owing to the high proportion of crossover in the MAVORIC trial, there is substantial uncertainty associated with the OS data. CADTH was not able to address this issue in reanalysis but examined it in an exploratory scenario analysis.
Limitations associated with the sponsor’s chosen modelling approach. The sponsor submitted a PSM in which treatment efficacy is represented by PFS and OS curves. For patients who do not undergo an aSCT, PFS and OS model inputs were based on observations from the MAVORIC trial, with vorinostat assumed by the sponsor to be a proxy for ECM. As noted in the CADTH Clinical Review Report, the median OS had not been reached in MAVORIC at the time of the data cut-off. Owing to the structure of PSMs, disease progression and OS are assumed to be independent; that is, any predicted gain in life-years is on the basis of OS, not PFS. This calls into question the sponsor’s predicted incremental gain of 3.51 life-years with mogamulizumab compared with ECM, given the high degree of uncertainty associated with the OS data from MAVORIC and a lack of statistically significant difference in OS between treatments after adjustment for crossover. Thus, any survival benefit obtained in the pharmacoeconomic model is assumed to be owing to benefits in PFS rather than to observed differences in survival. OS is additionally susceptible to potential confounding that may not affect PFS (e.g., choice of subsequent treatments after disease progression), and such confounding may attenuate an OS effect in clinical practice, despite the presence of a statistically significant effect of treatment on PFS in a clinical trial.14 The clinical experts consulted by CADTH indicated that the gains in PFS observed with mogamulizumab may translate to an overall incremental survival benefit in the range of 1 to 2 years (i.e., the predicted gain of 3.51 life-years was considered optimistic).
Results from the sponsor’s model suggest that the majority (81%) of the incremental benefits of mogamulizumab treatment are accrued in the post-progression health state (Table 11). This finding implies that the majority of the incremental benefit would be realized after patients have discontinued mogamulizumab. The clinical experts consulted by CADTH for this review noted there is no clear mechanism by which mogamulizumab would continue to provide clinical benefit after relapse. CADTH asked the sponsor to provide additional evidence to support the implied post-progression benefit (2.92 incremental life-years, 1.88 incremental QALYs). The sponsor noted that the observed results are the product of the selected parametric curves and are supported by a longer time-to-next treatment for mogamulizumab (11.0 months) and vorinostat (3.5 months) in the MAVORIC trial. The CADTH Clinical Review team examined this evidence and felt it was not sufficient to explain the post-progression survival difference observed in the sponsor’s model.
CADTH was unable to address these structural features of the sponsor’s pharmacoeconomic model and was unable to determine the extent to which the implied post-progression benefit was due to the effect of treatment versus due to structural bias within the PSM. CADTH was unable to address these issues in its reanalysis.
The long-term extrapolation of the effects of mogamulizumab is highly uncertain. The model’s efficacy data (i.e., PFS, OS) were based on observations from the MAVORIC trial, extrapolated over a 30-year horizon. As noted earlier, MAVORIC was not powered to estimate OS and maturity was not reached. As such, the incremental predicted a gain of 3.51 life-years with mogamulizumab is highly uncertain and is not supported by clinical trial data. CADTH notes that the extrapolated estimates for OS, as well as PFS, were highly variable and dependent on the assumed statistical distribution. The impact of alternative distributions was not explored by the sponsor. For PFS, the sponsor selected the log-normal distribution for both mogamulizumab and ECM on the basis of statistical fit (i.e., BIC and AIC), despite noting that the exponential and generalized gamma distributions, respectively, had better clinical fit. Statistical fit speaks only to the fit of the predicted data to the observed data within the trial period, not to the validity of predicted data for the extrapolated period. As such, the choice of parametric distribution for the extrapolation of data beyond the trial period should be based on clinical plausibility.
According to the clinical experts consulted on this review, the predicted OS obtained from the sponsor’s chosen parametric distribution (log-normal) lacks face validity. The log-normal distribution predicted that approximately 5% of patients with mycosis fungoides and Sézary syndrome who do not undergo an aSCT and 9% of patients who do undergo an aSCT would remain alive 30 years after initiating mogamulizumab, which clinical experts felt was too optimistic. The clinical experts consulted by CADTH additionally noted that the distribution of patients with mycosis fungoides and Sézary syndrome in the MAVORIC trial is not in keeping with the relative distribution of these diseases in clinical practice (i.e., Sézary syndrome was over-represented in the trial population) and that OS should be modelled separately, given the notable differences in the prognoses for these subgroups.
Given the high degree of uncertainty associated with the extrapolated data, the external validity of the sponsor’s predicted survival benefit with mogamulizumab is highly uncertain. As such, the predicted gain of life-years and QALYs associated with mogamulizumab relative to ECM is highly uncertain. CADTH was not able to address this limitation in its reanalysis.
The cost of ECM does not represent current clinical practice. The sponsor compared the effectiveness of mogamulizumab with a basket of therapies assumed to represent the ECM of mycosis fungoides and Sézary syndrome (Table 9), with the frequency of use of each therapy within the basket based on a survey of experts conducted by the sponsor. The sponsor assumed that the composition of ECM would be the same for patients with mycosis fungoides or Sézary syndrome, which lacks face validity, according to the clinical experts consulted by CADTH for this review. The clinical experts consulted by CADTH further noted that the treatment of mycosis fungoides and Sézary syndrome is variable and depends, at least in part, on disease stage and the prior treatments received, and may be guided by pathologic disease features.
The clinical experts further indicated that the composition of the ECM basket is not aligned with clinical practice in Canada, both in terms of the included treatments and their relative frequency of use. CADTH notes that some treatments included in ECM are not routinely available in Canada, owing to a lack of Health Canada approval (i.e., bexarotene), while access to other systemic therapies was noted to vary by jurisdiction. The clinical experts noted that interferon, retinoids, chemotherapy drugs, and histone deacetylase inhibitors are commonly used as first- and second-line systemic treatments, depending on jurisdictional coverage. In the sponsor’s model, interferon was assumed to account for 20% of the ECM usage, while retinoids were not included as part of ECM except for bexarotene which, as noted earlier, is not approved for use in Canada. The sponsor has further assumed that 30% of patients would receive methotrexate as part of ECM; however, the clinical experts consulted by CADTH noted that this is likely an overestimate, as methotrexate is infrequently used in their practices for the treatment of mycosis fungoides or Sézary syndrome, although this may depend on disease stage. Finally, the sponsor excluded brentuximab vedotin from the basket of ECM therapies “given that its target population is limited to only CD30+ patients, representing only a fraction of CTCL [cutaneous T-cell lymphoma] patients” and that brentuximab vedotin “would be prescribed before mogamulizumab would be considered.”1 The clinical experts consulted by CADTH noted that approximately 20% of patients with mycosis fungoides (60% of patients with mycosis fungoides and large-cell transformation) may have CD30-expressing disease; however, patients with large-cell transformation were excluded from the MAVORIC trial.
Expected clinical management, as conceptualized by the sponsor, likely does not reflect the actual impact the adoption of mogamulizumab would have on participating drug plans. As such, the cost-effectiveness of mogamulizumab relative to ECM and compared with relevant individual comparators is unknown. CADTH was unable to address this in its reanalysis. CADTH notes that the sponsor’s assumption that 30% of patients with mycosis fungoides or Sézary syndrome would receive methotrexate likely results in an underestimation of the cost of ECM, owing to the low cost of methotrexate relative to other treatments for mycosis fungoides and Sézary syndrome; however, the clinical experts consulted by CADTH for this review indicated that this represents an overestimation of methotrexate use.
The model lacked transparency. The sponsor’s submitted model included numerous IFERROR statements, which lead to situations in which the parameter value is overwritten with an alternative value without alerting the user to the automatic overwriting. The systematic use of IFERROR statements makes thorough auditing of the sponsor’s model impractical, as it remains unclear whether the model is running inappropriately by overriding errors.
CADTH was unable to address this limitation and notes that a thorough validation of the sponsor’s model was not practicable.
Additionally, the following key assumptions were made by the sponsor and have been appraised by CADTH (Table 4).
Table 4: Key Assumptions of the Submitted Economic Evaluation
Sponsor’s key assumption | CADTH comment |
|---|---|
Patients enrolled in the MAVORIC trial were assumed to be representative of the patients in Canada who would be eligible for mogamulizumab (age: 63.04 years; 41.9% female, weight: 76.77 kg; body surface area: 1.91 m2). | Reasonable, although the clinical experts consulted by CADTH for this review noted that the patients enrolled in MAVORIC represent a wide range of disease stages with various prognoses. The MAVORIC trial enrolled patients with stage IB and higher-stage disease and an ECOG PS of 0 or 1, which is narrower than the proposed indication population. |
Subsequent treatment was assumed to be the same regardless of when mogamulizumab or ECM was initially received. | Uncertain. The experts consulted by CADTH noted that treatment of mycosis fungoides and Sézary syndrome is guided by disease stage and the prior treatments received. Should mogamulizumab become reimbursed, patients whose disease progresses on ECM may receive mogamulizumab as a later line of treatment. Subsequent treatment was assumed to affect costs only, and the potentially confounding effect of subsequent treatment on overall survival was not considered. |
The sponsor assumed that the health-state utility for patients in the post-progression state would be equal for those who receive subsequent treatment and those who do not receive subsequent therapy. | Uncertain. The sponsor assumed that the utility value for all patients in the post-progression state would be equal to the last observed utility value after disease progression for patients in the mogamulizumab arm of the MAVORIC trial. This does not account for the effect of subsequent treatment on health-related quality of life (i.e., improvements owing to treatment effect, decrements owing to adverse events). Further, patients who do not receive subsequent treatment may be more ill than those who receive further treatment and, consequently, may have a lower health-related quality of life. |
Routine-care costs were based on an analysis of inpatient admissions, emergency department visits, and outpoint appointments at National Health Service hospitals in England and were assumed to be equal for mogamulizumab and ECM. | Uncertain. The sponsor undertook a retrospective study of the National Health Service’s Hospital Episode Statistics database, and the estimated costs were converted to Canadian dollars. Insufficient detail was provided to enable CADTH to evaluate whether the sponsor’s estimates are relevant in the Canadian context. The clinical experts consulted by CADTH indicated that patients receiving mogamulizumab may have more frequent outpatient visits to monitor for adverse events (e.g., drug eruptions) compared with patients on ECM. |
The sponsor modelled the initial time on treatment using Kaplan-Meier data from the MAVORIC trial for mogamulizumab and vorinostat (as a proxy for time on ECM). | Uncertain. The sponsor assumed that all patients in the model would discontinue treatment with mogamulizumab or ECM at the end of the observed MAVORIC trial data. The sponsor noted that the time on treatment for vorinostat in MAVORIC is shorter than what is seen in clinical practice with some of the components of ECM.1 |
Costs and disutilities related to grade 3+ adverse events with an incidence of at least 5% in the MAVORIC trial were included in the model. | Inappropriate. The sponsor selected an arbitrary threshold to capture the impact of treatment-related adverse events rather than selecting the most clinically meaningful adverse events to include within the model. As noted in the CADTH Clinical Review Report, drug eruption (all grades) was more common among patients who received mogamulizumab than in ECM. The inclusion of only grade 3+ drug eruptions in the pharmacoeconomic model may underestimate the cost of treatment associated with drug eruptions, as additional visits to a health care provider and drug treatments (e.g., topical corticosteroids) may be required. Additionally, the adverse events included in the sponsor’s model do not capture the range of adverse events deemed to be of special interest to clinicians (e.g., Stevens-Johnson syndrome, toxic epidermal necrolysis) or those noted in the patient input (e.g., hair loss) received by CADTH for this review. Adverse events related to aSCT were not included in the sponsor’s model. Increased transplant complications have been reported in patients who receive aSCT after mogamulizumab therapy, including grade 3 or 4 acute graft-vs.-host disease (GVHD), steroid-refractory GVHD, and transplant-related death.15 |
The adverse event profile for ECM was assumed to be equivalent to that of vorinostat. | Inappropriate. Each treatment included in the ECM basket of therapies would be associated with its own adverse event profile. The clinical experts consulted by CADTH noted that methotrexate (which accounts for 30% of ECM) would be expected to have a lower rate of gastrointestinal adverse events compared with vorinostat. The sponsor did not justify the assumption that the frequency and type of adverse events associated with vorinostat would be equal to those for ECM. |
Overall survival and disease-free survival for patients who undergo an aSCT were based on data pertaining to patients whose care was managed at a “supra-regional” cancer centre in the UK. | Uncertain. The sponsor notes that OS and disease-free survival were based on digitized Kaplan-Meier curves from the NICE appraisal of brentuximab vedotin,3 in which the original source of the data is cited as Palanicawandar (2017).16 As noted by NICE,3 these data pertain to 18 patients (mean age 47 years) who received a minimally intensive treatment protocol before aSCT (5-year survival: 55%),3 which is considerably younger than the MAVORIC trial population. Additional population characteristics and study design for the collection of these data were not described by the sponsor and thus could not be appraised by CADTH for methodological rigour. It is uncertain whether the management of patients at these centres is generalizable to the Canadian context. |
Premedication costs were not included. | Inappropriate, although unlikely to have an important effect on the ICER. The draft mogamulizumab monograph recommends administration of premedication (i.e., diphenhydramine, acetaminophen) with the first infusion and for subsequent infusions if an infusion reaction occurs.2 Infusion-related reactions were more common in the mogamulizumab arm than in the vorinostat arm of the MAVORIC trial (mogamulizumab: 33.2% of patients; vorinostat: 0.5% of patients). |
aSCT = allogeneic stem-cell transplant; ECM = established clinical management; ECOG PS = Eastern Cooperative Oncology Group Performance Status; GVHD = graft-vs.-host disease; ICER = incremental cost-effectiveness ratio; NICE = National Institute for Health and Care Excellence.
CADTH Reanalyses of the Economic Evaluation
Base-Case Results
As noted earlier, there are key limitations associated with the model structure, available clinical data, and the ECM comparator. Given that PSMs rely on mature OS data to produce reliable cost-effectiveness estimates, the use of a PSM is inappropriate. CADTH notes that the sponsor’s model predicts improbable estimates of incremental QALYs gained after disease progression, which was not supported by trial data. Further, as noted previously, the ECM comparator is not reflective of clinical practice in Canada and it is highly uncertain whether the effectiveness and safety of ECM is equivalent to that of vorinostat. Further, the incremental cost of mogamulizumab relative to ECM is highly uncertain, given that the sponsor’s ECM basket does not reflect Canadian clinical practice. The result of these limitations is that the costs and QALYs associated with the use of mogamulizumab are highly uncertain. These limitations represent fundamental problems for interpreting the results of the sponsor’s economic evaluation — since the costs and QALYs used to calculate the ICER are derived from an inappropriate model type and based on highly uncertain evidence — and for conducting any reanalyses using the sponsor’s model. As a result, any estimate of incremental cost-effectiveness would be misleading. Consequently, CADTH did not construct a reanalysis base case.
Scenario Analysis Results
Since no reanalysis was performed, price reduction analyses were conducted using the sponsor’s base-case assumptions only. This deterministic analysis — based on publicly available prices of the comparator treatments and subject to the key limitations of the sponsor’s model noted earlier — found that the price of mogamulizumab would need to be reduced by 51% for mogamulizumab to be cost-effective at a conventional threshold of $50,000 per QALY compared with ECM. It is important to note that this price reduction estimate is based on estimates of incremental OS and costs that are likely not representative of the true impact of mogamulizumab treatment. Consequently, the true price reduction that would be needed for mogamulizumab to be cost-effective remains unknown. The directionality of bias within the sponsor’s submission suggests that the price reduction would need to be higher than the estimated 51%.
Although CADTH did not conduct any formal reanalyses of the sponsor’s model, an exploratory analysis was undertaken to explore the impact of crossover adjustment and extrapolation of OS on the ICER. The key insight from this exploratory analysis is that the cost-effectiveness estimate of mogamulizumab is highly influenced by uncertainty in the OS data. (Details of this exploratory analysis are provided in Appendix 4.)
Issues for Consideration
The clinical experts consulted by CADTH indicated that mogamulizumab is likely to be used as third-line treatment after systemic treatment with interferon and retinoids as first- and second-line systemic treatments. However, the clinical experts indicated that mogamulizumab may be considered after first-line systemic treatment for some patients, depending at least in part on disease stage. Owing to the structure of the sponsor’s model and a lack of clinical data, CADTH was unable to explore the cost-effectiveness of mogamulizumab in different lines of therapy or based on prior treatment experience.
The clinical experts consulted by CADTH for this review, as well as the clinician and patient input, indicated that some treatments (e.g., phototherapy, total skin electron beam therapy, extracorporeal phototherapy) for mycosis fungoides and Sézary syndrome may be inaccessible for some patients (e.g., those living in remote regions) and associated with out-of-pocket costs (e.g., travel); for such patients, systemic treatments may be preferred. The availability of a new hospital-based treatment will likely affect these patient-borne costs and may lead to changes in treatment accessibility. These costs are not reflected in the estimates of cost-effectiveness.
In the patient input received by CADTH for this review, some patients reported a preference for fewer hospital visits for treatments and some patients with experience with mogamulizumab noted that the number of hospital visits required for mogamulizumab administration negatively affected their QoL. The impact of hospital visits on QoL was not captured as part of the sponsor’s submission.
Overall Conclusions
The CADTH review of the clinical evidence suggests that mogamulizumab may improve PFS compared with vorinostat among patients with mycosis fungoides and Sézary syndrome who have received at least 1 prior systemic treatment; however, the effects of mogamulizumab on OS are highly uncertain. Importantly, the comparator in the pivotal MAVORIC trial, vorinostat, is not the standard of care in Canada and it is highly uncertain if the effectiveness of ECM is equal to that of vorinostat. Additionally, the treatments included in the basket of ECM and their relative use are not reflective of Canadian clinical practice. As such, whether mogamulizumab is effective compared with ECM in the Canadian context is highly uncertain.
The sponsor submitted a model comparing the cost-effectiveness of mogamulizumab with ECM and reported a base-case ICER of $96,742 per QALY. This result suggests that, even under the sponsor’s baseline assumptions, there is a 0% probability that mogamulizumab is cost-effective at a willingness-to-pay threshold of $50,000.
CADTH was not able to conduct a reanalysis due to foundational limitations within the sponsor’s model and submitted evidence. Notably, the choice of a PSM was inappropriate, given that the OS data are highly uncertain (i.e., the MAVORIC trial was not powered to detect a difference in OS, the OS data are immature, and the OS data are confounded by patient crossover between treatment arms). Further, the sponsor’s model predicted that 81% of the incremental benefit with mogamulizumab would be accrued after the discontinuation of mogamulizumab by patients receiving subsequent treatment, which was not supported by the clinical trial evidence. Given that the sponsor’s conceptualization of ECM is not in line with Canadian clinical practice, there is a high level of uncertainty about the incremental costs associated with mogamulizumab treatment. CADTH was unable to address these critical limitations (i.e., the apparent post-progression survival benefit, ECM costs, and effectiveness) due to constraints introduced by the submitted model structure and a lack of clinical data.
The limitations identified within the submitted evidence interact with each other. The uncertainty in the MAVORIC trial’s ability to estimate OS for mogamulizumab contributed structural uncertainty to the PSM approach chosen by the sponsor. The comparative effectiveness of mogamulizumab versus ECM is confounded by issues related to the lack of head-to-head evidence and issues within the sponsor’s MAIC. This combination of data and model design limitations produced survival results that lacked face validity. This uncertainty in incremental effectiveness was matched by unknown incremental costs due to a definition of ECM that did not match current clinical practice. Consequently, the cost-effectiveness of mogamulizumab is unknown.
References
1.Pharmacoeconomic evaluation [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Poteligeo (mogamulizumab), 4 mg/mL (20 mg) for intravenous infusion. Bedminster (NJ): Kyowa Kirin Canada, Inc; 2021 Sep 13.
2.Poteligeo (mogamulizumab): 20 mg/5 mL single-use vial, 4 mg/mL for intravenous infusion [DRAFT product monograph]. Bedminster (NJ): Kyowa Kirin, Inc.
3.Brentuximab vedotin for treating CD30‐positive cutaneous T‐cell lymphoma [ID1190]. Pre-meeting briefing. London (GB): National Institute for Health and Care Excellence; 2018.
4.Ontario Ministry of Health, Ontario Ministry of Long-Term Care. Ontario drug benefit formulary/comparative drug index. 2021; https://www.formulary.health.gov.on.ca/formulary/. Accessed 2021 Sep 13.
5.Alberta Health Care Insurance Plan: medical price list as of 31 March 2020. Edmonton (AB): Government of Alberta; 2020: https://open.alberta.ca/dataset/30add047-29c2-4fc7-83b5-a8ab78605cdd/resource/48d0dd0b-bb33-478d-a85c-85a86d4555ad/download/health-somb-medical-price-list-2020-03.pdf. Accessed 2021 Sep 13.
6.Schedule of benefits for physician services under the Health Insurance Act: effective April 1, 2020. Toronto (ON): Ontario Ministry of Health; 2020: https://www.health.gov.on.ca/en/pro/programs/ohip/sob/physserv/sob_master20200306.pdf. Accessed 2021 Sep 13.
7.Extracorporeal photophoresis: an evidence-based analysis. (Ontario Health technology assessment series v.6 no.6). Toronto (ON): Ontario Ministry of Health and Long-Term Care; 2006: https://www.hqontario.ca/Portals/0/Documents/evidence/reports/rev_ecp_030106.pdf. Accessed 2021 Oct 21.
8.Shinder GA, Paradis PE, Posman M, et al. Patient and work flow and costs associated with staff time and facility usage at a comprehensive cancer centre in Quebec, Canada--a time and motion study. BMC Health Serv Res. 2012;12:370. PubMed
9.Interprovincial billing rates for designated high cost transplants effective for discharges on or after April 1, 2020. Toronto (ON): Ontario Ministry of Health. Ontario Ministry of Long-Term Care; 2020: https://www.health.gov.on.ca/en/pro/programs/ohip/bulletins/na_86/high_cost.pdf. Accessed 2021 Nov 8.
10.Ontario Case Costing Initiative (OCCI). Toronto (ON): Ontario Health and Long-Term Care; 2017: https://data.ontario.ca/dataset/ontario-case-costing-initiative-occi. Accessed 2021 Sep 13.
11.Alberta health. Hospital ambulatory care case costs - CACS - 2018 version. 2018; http://www.ahw.gov.ab.ca/IHDA_Retrieval/selectSubCategory.do. Accessed 2020 Nov 1.
12.Beaudry A, Ferguson TW, Rigatto C, Tangri N, Dumanski S, Komenda P. Cost of dialysis therapy by modality in Manitoba. Clin J Am Soc Nephrol. 2018;13(8):1197-1203. PubMed
13.de Oliveira C, Pataky R, Bremner KE, et al. Phase-specific and lifetime costs of cancer care in Ontario, Canada. BMC Cancer. 2016;16(1):809. PubMed
14.Msaouel P, Grivas P, Zhang T. Adjuvant systemic therapies for patients with renal cell carcinoma: choosing treatment based on patient-level characteristics. Eur Urol Oncol. 2021. PubMed
15.Mogamulizumab: drug information. UpToDate. Waltham (MA): UpToDate; 2021: http://www.uptodate.com. Accessed 2021 Sep 28.
16.Palanicawandar R, Morris S, Lozano-Cerrada S, et al. Allogenic stem cell transplantation for advanced cutaneous T-cell lymphoma with minimal-intensity conditioning. EORTC-CLTF; 2017; London (GB).
17.Budget Impact Analysis [internal sponsor's report]. In: Drug Reimbursement Review sponsor submission: Poteligeo (mogamulizumab), 4 mg/mL (20 mg) for intravenous infusion. Bedminster (NJ): Kyowa Kirin Canada, Inc; 2021 Sep 13.
18.Ghazawi FM, Netchiporouk E, Rahme E, et al. Comprehensive analysis of cutaneous T-cell lymphoma (CTCL) incidence and mortality in Canada reveals changing trends and geographic clustering for this malignancy. Cancer. 2017;123(18):3550-3567. PubMed
Appendix 1: Cost Comparison Table
Note that this appendix has not been copy-edited.
The comparators presented in the following table have been deemed to be appropriate based on feedback from clinical expert(s) CADTH-participating drug plans. Comparators may be recommended (appropriate) practice or actual practice. Existing Product Listing Agreements are not reflected in the table and as such, the table may not represent the actual costs to public drug plans.
Table 5: CADTH Cost Comparison Table for Systemic Treatments Indicated for Mycosis Fungoides and Sézary Syndrome
Treatment | Strength/ concentration | Form | Price ($) | Recommended dosage | Daily cost ($) | Average 28-day |
|---|---|---|---|---|---|---|
Mogamulizumab (Poteligeo) | 4 mg/mL | 20 mg vial | 2,203.60b | Cycle 1: 1 mg/kg on days 1, 8, 15, and 22 Cycle 2+: 1 mg/kg on days 1 and 15 | Cycle 1: 1,259 Cycle 2+: 630 | Cycle 1: 35,258 Cycle 2+: 17,629 |
Brentuximab vedotin (Adcetris)a | 50 mg | Vial | 4,840.0000c | 1.8 mg/kg every 3 weeks for up to 16 cycles | 691 | 14,520 |
For dosing that depends on weight or body surface area, CADTH assumed 76.77 kg or 1.91 m2, respectively, based on the MAVORIC trial. Wastage considered for single-use vials.
aFor the treatment of primary cutaneous anaplastic large-cell lymphoma or CD30-expressing mycosis fungoides.
bSponsor-submitted price; dosage based on the draft product monograph.
cDelta IQVIA wholesale price (November 2021).
Table 6: CADTH Cost Comparison Table for Systemic Treatments Used Off-Label for Mycosis Fungoides and Sézary Syndrome
Treatment | Strength/ concentration | Form | Price ($) | Recommended dosage | Daily cost ($) | Average 28-day |
|---|---|---|---|---|---|---|
Single-drug chemotherapy | ||||||
Gemcitabine | 1 g 2 g | Powder for IV infusion | 270.0000a 540.0000a | 1,000 mg/m2 given twice in a 21-day cycle | 51 | 1,440 |
Methotrexate (generics) | 2.5 mg | Tablet | 0.6325 | 15 to 30 mg weekly | 1 | 15 to 30 |
15 mg/0.3 mL 17.5 mg/0.35 mL 20 mg/2 mL 20 mg/0.4 mL 22.5 mg/0.45 mL 25 mg/0.5 mL | Pre-filled syringe | 24.5700 24.0000 12.5000 26.2500 26.5000 29.2500 | 4 to 7 | 98 to 197 | ||
Histone deacetylase inhibitor | ||||||
Romidepsin (Istodax) | 10 mg | Powder for injection | 2,582.0000a | 14 mg/m2 on days 1, 8, and 15 (28-day cycle) | 830 | 23,238 |
Other therapies | ||||||
Pegylated interferon alfa-2a (Pegasys) | 180 mcg/0.5 mL | Pre-filled syringe | 419.700c | 35 mcg to 210 mcg weekly | 60 to 120 | 1,679 to 3,358 |
Prednisone (generics) | 5 mg | Tab | 0.0220 | 20 mg daily | 0.09 | 2.46 |
Prices are from the Ontario Drug Benefit Formulary (accessed November 2021), unless otherwise indicated, and do not include dispensing fees. For dosing that depends on weight or body surface area, CADTH assumed 76.77 kg or 1.91 m2, respectively, based on the MAVORIC trial. Total cost estimates per regimen are based on the cheapest combination of the component drugs, with wastage considered for single-use vials or syringes.
The sponsor’s definition of ECM included a total of 16 possible medications, several of which were assumed to be used in fewer than 5% of patients. Following guidance from participating drug plans, only the most relevant drug treatments are included in this table. A full list of the treatments included in the sponsor’s ECM basket is available in Table 9.
aDelta IQVIA wholesale price (November 2021).
bOntario Exceptional Access Program (November 2021).
Table 7: CADTH Cost Comparison Table for Non-Pharmacologic Treatment of Mycosis Fungoides and Sézary Syndrome
Treatment | Price ($) | Recommended dosage | Daily cost ($) | Average 28-day |
|---|---|---|---|---|
Extracorporeal photopheresis (ECP) | $1,821.79 | Once every 2 weeks for 2 consecutive days; for patients with Sézary syndrome or mycosis fungoides with blood involvement | $260.26 | $7,287.16 |
CADTH was not able to validate this sponsor-submitted estimate of the cost of ECP.
Appendix 2: Submission Quality
Note that this appendix has not been copy-edited.
Description | Yes/No | Comments |
|---|---|---|
Population is relevant, with no critical intervention missing, and no relevant outcome missing | No | The established clinical management (ECM) comparator does not reflect clinical practice in Canada (i.e., included treatments, frequency of use). The sponsor’s model allowed consideration of clinically relevant subgroups; however, the composition of ECM was assumed by the sponsor to be the same for patients with mycosis fungoides or Sézary syndrome and to not vary by disease stage, which lacks face validity according to the clinical experts consulted by CADTH for this review. |
Model has been adequately programmed and has sufficient face validity | No | The model includes numerous IFERROR statements. The systematic use of IFERROR statements makes thorough auditing of the sponsor’s model impractical, as it remains unclear whether the model is running inappropriately by overriding errors. The sponsor modelled 3 separate patient pathways to reflect the possibility of patients receiving aSCT. For the cohort assumed to undergo an aSCT after initial treatment with mogamulizumab or ECM, immortal time bias was introduced, as all patients were assumed to remain alive until the time of aSCT, which is unlikely. |
Model structure is adequate for decision problem | No | A partitioned-free survival model was used which introduced structural constraints. A Markov model would have been more appropriate. |
Data incorporation into the model has been done adequately (e.g., parameters for probabilistic analysis) | No | Drug acquisition costs were assumed to be variable (in addition to varying with BSA). This is inappropriate, given that drug costs are fixed through negotiation by the health care payer. The sponsor provided CADTH with a revised model; however, this issue persisted. |
Parameter and structural uncertainty were adequately assessed; analyses were adequate to inform the decision problem | No | For some model parameters, the sponsor arbitrarily incorporated uncertainty, which does not reflect the true uncertainty around the model’s parameters possible values. |
The submission was well organized and complete; the information was easy to locate (clear and transparent reporting; technical documentation available in enough details) | No | Multiple discrepancies noted between the report and model file (e.g., % of patients assumed to receive aSCT after mogamulizumab). The source of data (e.g., outcome data after aSCT, recommended drug dosage) was not adequately described or referenced. As part of ECM, the sponsor included some drug strengths not available in Canada (i.e., 25 mg prednisolone). The original version of the model and pharmacoeconomic report inappropriately underestimated the cost of allogenic stem-cell transplant. A subsequent version was provided to CADTH during the review period in which this error was been corrected. |
Appendix 3: Additional Information on the Submitted Economic Evaluation
Note that this appendix has not been copy-edited.
Figure 1: Model Structure — Patients Who Do Not Undergo Allogeneic Stem-Cell Transplant
NTFS = next-treatment-free survival; OS = overall survival; ToT = time on treatment.
Source: Sponsor’s pharmacoeconomic submission.1
Figure 2: Model Structure — Patients Who Undergo Allogeneic Stem-Cell Transplant After Mogamulizumab or Expected Clinical Management
aSCT = allogeneic stem-cell transplant; OS = overall survival.
Source: Sponsor’s pharmacoeconomic submission.1
Figure 3: Model Structure — Patients Who Undergo Allogeneic Stem-Cell Transplant After Subsequent Treatment
aSCT = allogeneic stem-cell transplant; NTFS = next-treatment-free survival; OS = overall survival; ToT = time on treatment.
Source: Sponsor’s pharmacoeconomic submission.1
Table 9: Composition of Expected Clinical Management
Treatment | Cost ($) | Dosage form | Dose required per administrationa | Administration frequency | Cost per administrationb ($) | Frequency of use as part of ECM |
|---|---|---|---|---|---|---|
Pharmaceutical treatments | ||||||
Methotrexate | 0.6325 per 2.5 mg | Tablet | 23.44 mg | Once weekly | 6.33 | 30% |
Bexarotene | 261.90 per 75 mg | Vial | 600 mg | Daily | 2,095.20 | 1% |
Interferon alfa-2a | 419.70 per 180 mcg | Pre-filled syringe | 180 mcg | Once weekly | 419.70 | 20% |
Gemcitabine | 124.00 per 2,000 mg | Solution for infusion | 1,910 mg | Day 1 and 8 (21-day cycle) | 124.00 | 5% |
Cyclophosphamide | 0.47 per 50 mg | Tablet | 1,432.50 mg | Day 1 (21-day cycle) | 13.75 | CHOP:f 5% |
Doxorubicin | 5.60 per 50 mg | Vial | 95.50 mg | Day 1 (21-day cycle) | 11.20 | |
Vincristine | 31.00 per 2 mg | Vial | 2.67 mg | Day 1 (21-day cycle) | 62.00 | |
Liposomal doxorubicin | 341.50 per 10 mg | Vial | 38.20 mg | Twice per month | 1,366.00 | 3% |
Etoposide | 0.75 per 100 mg | Capsule | 343.80 mg | 5 days per month | 2.58 | 3% |
Prednisolone | 11.50 per 25 mg | Tablet | 76.40 mg | Day 1, 2, 3, 4, and 5 (21-day cycle) | 35.14 | 15% |
Vorinostat | 301.79 per 100 mg | Tablet | 400 mg | Daily | 1,207.16 | 3% |
Chlorambucil | 1.39 per 2 mg | Tablet | 7.68 mgd | Daily for 4 to 8 weeks initiallye | 5.56 | 1% |
Purine analogues (Fludara) | 40.08 per 10 mg | Tablet | 382.00 mgc | 5 days per 28-day cycle | 1,522.89 | 1% |
Pralatrexate (Folotyn) | 2,108.63 per 20 mg | Vial | 57.30 mgc | Once weekly for 6 weeks in 7-week cycles | 6,325.89 | 2% |
Romidepsin (Istodax) | 2,582.00 per 10 mg | Vial | 26.74 mgc | Day 1, 8, and 15 (28-day cycle) | 7,746.00 | 2% |
Alemtuzumab (Lemtrada) | 1,1045.83 per 12 mg | Vial | 60 mg | Two courses: 12 mg/day for 5 days, followed by 12 mg/day for 3 days after 12 months | 5,229.17 | 1% |
Other treatments | ||||||
Modality | Cost per session | Administration frequency | Cost included in model | Frequency of use as part of ECM | ||
PUVA | 80.00 | Twice per week for 14 weeks | 160.00 | 2% | ||
ECP | 1,821.79 | 2 days per 28-day cycle | 837.88 | 5% | ||
TSEBT | 913.84 | Four times per week for 4 weeks | 851.77 | 1% | ||
ECP = extracorporeal photopheresis; PUVA = psoralen plus UVA phototherapy; TSEBT = total skin electron beam therapy.
aSource of required doses not provided by sponsor.
bCost per administration was assumed to follow a gamma distribution in the sponsor’s probabilistic analysis.
cBased on mg/m2 dosing, assuming 1.91 m2 body surface area; assumed to be normally distributed in the sponsor’s probabilistic analysis.
dBased on mg/kg dosing, assuming 76.77 kg.
eInitial dose noted by the sponsor to be 0.1 to 0.2 mg/kg daily. Sponsor notes that, after the first 4 to 8 weeks, maintenance therapy is given either by a reduced daily dosage or intermittent courses of treatment. The required dose in the sponsor’s model assumes 0.1 mg/kg daily.
fCost of CHOP per 21-day cycle assumed to be $262.67. Assumed to include cyclophosphamide, doxorubicin, vincristine (each administered once per 21-days) and prednisolone (administered 5 times per 21-day cycle).
Table 10: Summary of the Clinical Data and Statistical Distribution for the Extrapolation of Observed Effects in the Sponsor’s Base Case
Outcome | Group | Source of clinical data | Statistical distribution |
|---|---|---|---|
All patients | |||
Time on treatment | Mogamulizumab | MAVORIC, post hoc analyses | KM curve; maximum time on treatment assumed to be 60 months |
ECM | MAVORIC, post hoc analyses (vorinostat arm) | KM curve; maximum time on treatment assumed to be 41 months | |
Next-treatment-free survival | Mogamulizumab | MAVORIC, post hoc analyses | Generalized gamma |
ECM | MAVORIC, post hoc analyses (vorinostat arm) | Generalized gamma | |
Progression-free survival | Mogamulizumab | MAVORIC | Log-normal |
ECM | MAVORIC (vorinostat arm) | Log-normal | |
Patients who do not undergo an aSCT | |||
Overall survival | Mogamulizumab | MAVORIC, post hoc analyses excluding patients who underwent with aSCT | Log-normal |
ECM | MAVORIC post hoc analyses (vorinostat arm) adjusted for crossover,a excluding patients who underwent an aSCT | Exponential | |
Patients who undergo an aSCTb | |||
Disease-free survival | Patients who received aSCT after mogamulizumab or ECM or after subsequent treatment after disease progression | London supra-regional centre | Gompertz |
Overall survival | As noted earlier | London supra-regional centre | Log-normal |
aSCT = allogeneic stem-cell transplant; ECM = established clinical management; KM = Kaplan-Meier.
aAdjusted by use of inverse probability of censoring weighting.
bFor patients who undergo an aSCT, the sponsor assumed that disease-free survival and overall survival would not differ, depending on previous treatment received.
Detailed Results of the Sponsor’s Base Case
Table 11: Disaggregated Summary of the Sponsor’s Economic Evaluation Results
Parameter | Mogamulizumab | ECM | Incremental (v. ECM) |
|---|---|---|---|
Discounted LYs | |||
Progression-free | 1.29 | 0.50 | 0.78 |
Post-progression — off treatment | 0.12 | 0.07 | 0.05 |
Post-progression — receiving subsequent treatment or ESC | 5.48 | 2.61 | 2.86 |
aSCT — disease-free | 0.61 | 0.76 | −0.16 |
aSCT — relapsed | 0.10 | 0.13 | −0.03 |
Total | 7.60 | 4.08 | 3.51 |
Discounted QALYs | |||
Progression-free | 0.94 | 0.37 | 0.58 |
Post-progression — off treatment | 0.09 | 0.05 | 0.04 |
Post-progression — receiving subsequent treatment or ESC | 3.42 | 1.58 | 1.84 |
aSCT — disease-free | 0.46 | 0.58 | −0.12 |
aSCT — relapsed | 0.04 | 0.07 | −0.03 |
Total | 4.96 | 2.65 | 2.31 |
Discounted costs ($) | |||
Drug acquisition | 213,432 | 16,671 | 196,761 |
Administration | 1,333 | 157 | 1,176 |
Monitoring/routine care | |||
Pre-progression — on treatment | 8,673 | 4,383 | 4,289 |
Pre-progression — off treatment | 4,385 | 704 | 3,681 |
Post-progression | 56,523 | 26,975 | 29,548 |
aSCT — relapsed | 917 | 837 | 80 |
End-stage care | |||
Progressed | 15,811 | 16,314 | −503 |
aSCT | 0 | 0 | 0 |
aSCT costs and monitoring DF | 15,776 | 19,729 | −3,953 |
Subsequent treatment | |||
Patients who have undergone aSCT | 22,924 | 28,950 | −6,026 |
Patients with no aSCT | 79,785 | 81,319 | −1,534 |
Adverse events | 96 | 261 | −165 |
Total | 419,654 | 196,301 | 226,081 |
ICER ($/QALY) | 97,960 | ||
aSCT = allogeneic stem-cell transplant; ICER = incremental cost-effectiveness ratio; LY = life-year; QALY = quality-adjusted life-year.
Source: Sponsor’s pharmacoeconomic submission.1
Appendix 4: Additional Details on the CADTH Reanalyses and Sensitivity Analyses of the Economic Evaluation
Note that this appendix has not been copy-edited.
Price Reduction Analysis
Since no reanalysis was performed, price reduction analyses were conducted using the sponsor’s base-case assumptions only. This deterministic analysis was subject to the key limitations of the sponsor’s model as noted in the CADTH Appraisal of the Sponsor’s Economic Evaluation section. In this analysis, based on the sponsor’s submitted model, the price of mogamulizumab would need to be reduced by 51% for mogamulizumab to be cost-effective at a conventional willingness-to-pay threshold of $50,000 per QALY compared with ECM. It is important to note that this price reduction estimate is based on estimates of incremental life-years (and hence QALYs) that are highly uncertain and may not be representative of the true incremental effect of mogamulizumab treatment. Consequently, the true price reduction that would be needed for mogamulizumab to be cost-effective remains unknown.
Table 12: Price Reduction Analyses of Sponsor’s Base Case
Price reduction | ICERs for mogamulizumab vs. ECM ($) | |
|---|---|---|
Sponsor’s base case | CADTH reanalysisa | |
No price reduction | 96,216 | NA |
10% | 87,068 | NA |
20% | 77,921 | NA |
30% | 68,774 | NA |
40% | 59,626 | NA |
50% | 50,479 | NA |
51% | 49,564 | NA |
ECM = established clinical management; ICER = incremental cost-effectiveness ratio; NA = not applicable.
Note: All analyses in this table are deterministic and are subject to limitations within the sponsor’s model.
aCADTH did not conduct a reanalysis due to limitations within the sponsor’s model.
Scenario Analyses
Although CADTH did not conduct any formal reanalyses of the sponsor’s model, the economic review team performed an exploratory analysis to explore the impact of crossover adjustment and extrapolation of OS on the ICER.
It should be noted that the key limitations of the sponsor’s base-case analysis noted earlier apply to this exploratory analysis, including the fundamental limitation that there is no direct evidence to support the equivalent effectiveness of vorinostat and ECM. Therefore, this exploratory analysis should not be interpretated as a formal CADTH reanalysis to which credence should be given to the results; in particular, the incremental QALYs and costs estimated as part of this exploratory analysis remains unlikely to be representative of the true effect of mogamulizumab treatment, such that the corresponding ICER is unlikely to be reflective of the true cost-effectiveness of mogamulizumab. The key insight from this exploratory analysis is that the cost-effectiveness estimate of mogamulizumab is highly influenced by uncertainty in the OS data.
Table 13: Summary of CADTH Exploratory Analysis — Full Health Canada Indication
Drug | Total costs ($) | Incremental costs ($) | Total QALYs | Incremental QALYs | ICER vs. ECM |
|---|---|---|---|---|---|
Sponsor’s submitted base case — Full Health Canada indication | |||||
ECM | 196,123 | Ref. | 2.64 | Ref. | Ref. |
Mogamulizumab | 420,968 | 224,845 | 4.97 | 2.34 | 96,216 |
CADTH scenario 1: Overall survival — unadjusted data | |||||
ECM | 246,911 | Ref. | 4.45 | Ref. | Ref. |
Mogamulizumab | 422,542 | 175,631 | 7.62 | 0.53 | 332,667 |
CADTH scenario 2: Overall survival — TSE crossover adjustment | |||||
ECM | 211,735 | Ref. | 3.34 | Ref. | Ref. |
Mogamulizumab | 420,978 | 209,243 | 4.97 | 1.63 | 128,138 |
CADTH scenario 3: Overall survival — Gompertz distributiona | |||||
ECM | 196,123 | Ref. | 2.64 | Ref. | Ref. |
Mogamulizumab | 395,774 | 199,651 | 3.49 | 0.85 | 235,085 |
ECM = established clinical management; ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year; TSE = 2-stage estimation; vs. = versus.
Note: all analyses deterministic
aThe least optimistic distribution for overall survival was chosen for this analysis to explore the impact of overall survival extrapolation on the ICER.
Table 14: Summary of CADTH Exploratory analysis — Mycosis Fungoides
Drug | Total costs ($) | Incremental costs ($) | Total QALYs | Incremental QALYs | ICER vs. ECM |
|---|---|---|---|---|---|
Sponsor’s submitted scenario — Mycosis fungoides | |||||
ECM | 192,349 | Ref. | 2.65 | Ref. | Ref. |
Mogamulizumab | 401,383 | 209,034 | 5.68 | 3.03 | 69,063 |
CADTH scenario 1: Overall survival — unadjusted data | |||||
ECM | 240,339 | Ref. | 4.21 | Ref. | Ref. |
Mogamulizumab | 402,978 | 162,639 | 5.68 | 1.47 | 110,891 |
CADTH scenario 2: Overall survival — TSE crossover adjustment | |||||
ECM | 207,860 | Ref. | 3.29 | Ref. | Ref. |
Mogamulizumab | 401,387 | 193,528 | 5.68 | 2.38 | 81,190 |
CADTH scenario 3: Overall survival — Gompertz distributiona | |||||
ECM | 192,349 | Ref. | 2.65 | Ref. | Ref. |
Mogamulizumab | 370,200 | 177,850 | 3.76 | 1.11 | 160,489 |
ECM = established clinical management; ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year; TSE = 2-stage estimation; vs. = versus.
Note: all analyses deterministic.
aThe least optimistic distribution for overall survival was chosen for this analysis to explore the impact of overall survival extrapolation on the ICER.
Table 15: Summary of CADTH Exploratory Analysis — Sézary Syndrome
Drug | Total costs ($) | Incremental costs ($) | Total QALYs | Incremental QALYs | ICER vs. ECM |
|---|---|---|---|---|---|
Sponsor’s submitted base case — Sézary syndrome | |||||
ECM | 195,153 | Ref. | 2.64 | Ref. | Ref. |
Mogamulizumab | 445,475 | 250,323 | 4.24 | 1.61 | 155,922 |
CADTH scenario 1: Overall survival — unadjusted data | |||||
ECM | 249,368 | Ref. | 4.71 | Ref. | Ref. |
Mogamulizumab | 447,048 | 197,680 | 4.24 | –0.47 | Dominated |
CADTH scenario 2: Overall survival — TSE crossover adjustment | |||||
ECM | 211,305 | Ref. | 3.41 | Ref. | Ref. |
Mogamulizumab | 445,476 | 234,171 | 4.24 | 0.84 | 280,292 |
CADTH scenario 3: Overall survival — Gompertz distributiona | |||||
ECM | 195,153 | 230,772 | 2.64 | Ref. | Ref. |
Mogamulizumab | 425,925 | 230,772 | 3.15 | 0.51 | 450,959 |
ECM = established clinical management; ICER = incremental cost-effectiveness ratio; QALY = quality-adjusted life-year; TSE = 2-stage estimation; vs. = versus.
Note: all analyses deterministic
aThe least optimistic distribution for overall survival was chosen for this analysis to explore the impact of overall survival extrapolation on the ICER.
Appendix 5: Submitted Budget Impact Analysis and CADTH Appraisal
Note that this appendix has not been copy-edited.
Table 16: Summary of Key Takeaways
Key Takeaways of the BIA |
|---|
|
Summary of Sponsor’s Budget Impact Analysis
The sponsor submitted a budget impact analysis (BIA) estimating the incremental budget impact of reimbursing mogamulizumab for the treatment of mycosis fungoides and Sézary syndrome among patients who have received at least 1 prior systemic therapy.17 The budgetary impact was estimated separately for mycosis fungoides and Sézary syndrome and combined to reflect the impact of reimbursing mogamulizumab for the full Health Canada population. The BIA was undertaken from the perspective of the Canadian public drug plans over a 3-year time horizon, and the sponsor’s pan-Canadian estimates reflect the aggregated results from provincial budgets (excluding Quebec). Key inputs to the BIA are documented in Table 17.
The sponsor estimated the eligible population using an epidemiologic approach, with the estimated prevalence of mycosis fungoides and Sézary syndrome based on commissioned research in the US17 and incidence based on a Canadian study.18 The sponsor assumed that 100% of patients would be eligible for public drug coverage for all jurisdictions. In the reference scenario, patients were assumed to receive ECM, comprised of a basket of pharmaceutical (methotrexate, bexarotene, interferon alfa-2a, gemcitabine, CHOP, liposomal doxorubicin, etoposide, prednisolone, vorinostat, chlorambucil, purine analogues, pralatrexate, romidepsin, alemtuzumab) and non-pharmaceutical strategies.
In the sponsor’s base case, costs related to drug acquisition for mogamulizumab and ECM were captured, as well wholesale mark-up. Costs related to non-pharmaceutical interventions that might be received as part of ECM excluded. The cost of mogamulizumab was based on the sponsor’s submitted price ($2,203.60 per vial; annual cost: $104,441). Drug costs for ECM were obtained from the Ontario Drug Benefit Formulary,4 Alberta Drug Benefit List,5 the Yukon Drug Formulary, wholesale acquisition costs, and from prior CADTH reviews. In the new-drug scenario, mogamulizumab was assumed to be reimbursed and to displace ECM. For mycosis fungoides, mogamulizumab was assumed capture 1%, 2%, and 3% of market share in years 1, 2, and 3, respectively. For Sézary syndrome, mogamulizumab was assumed to capture 20%, 30%, and 35% of the market share in years 1, 2, and 3, respectively.17 The sponsor assumed that 19.5 doses of mogamulizumab would be received, while ECM was assumed to be received for the full 3-year BIA horizon.17
Table 17: Summary of Key Model Parameters
Parameter | Sponsor’s estimate (Year 1 / Year 2 / Year 3) |
|---|---|
Target population | |
Projected jurisdictional population | 30,540,461 / 30,854,447 / 31,171,878 |
Prevalence | |
Mycosis fungoides | |||| ||| ||||||| |
Sézary syndrome | |||| ||| ||||||| |
Annual incidence ratea | |
Mycosis fungoides | 0.417 per 100,000 |
Sézary syndrome | 0.027 per 100,000 |
Annual mortality | |
Mycosis fungoides | 0.020 |
Sézary syndrome | 0.108 |
Proportion who received prior systemic treatment | |
Mycosis fungoides | ||| |
Sézary syndrome | ||| |
Number of patients eligible for drug under review | |
Mycosis fungoides | ||| | ||| | ||| |
Sézary syndrome | || | || | || |
Market Uptake (3 years) | |
Uptake (reference scenario) | |
Mycosis fungoides | |
Mogamulizumab | 0% / 0% / 0% |
Expected clinical management | 100% / 100% / 100% |
Sézary syndrome | |
Mogamulizumab | 0% / 0% / 0% |
Expected clinical management | 100% / 100% / 100% |
Uptake (new-drug scenario) | |
Mycosis fungoides | |
Mogamulizumab | 1% / 2% / 3% |
Expected clinical management | 99% / 98% / 97% |
Sézary syndrome | |
Mogamulizumab | 20% / 30% / 35% |
Expected clinical management | 80% / 70% / 65% |
Cost of treatment (per patient) | |
Mogamulizumab (19.5 dosesb) | $104,441 |
Expected clinical management (annual cost) | $40,607c |
aAssumed to be jurisdiction-specific; summary value presented here.
bIn the BIA, the sponsor assumed that patients would receive 19.5 doses of mogamulizumab.
cSponsor’s predicted annual drug cost of established clinical management (ECM), with the cost of each individual drug weighted by the sponsor’s predicted frequency of use. Non-pharmacologic interventions that may be received as part of ECM were excluded.
Summary of the Sponsor’s BIA Results
The sponsor estimated the net 3-year budget impact of reimbursing mogamulizumab for the treatment of mycosis fungoides after previous systemic therapy to be $3,448,534 (year 1: $566,794; year 2: $1,146,499; year 3: $1,735,241). The net 3-year budget impact of reimbursing mogamulizumab for the treatment of Sézary syndrome after previous systemic therapy to be $7,481,937 (year 1: $1,730,077; year 2: $2,633,848; year 3: $3,118,012). The budget impact for the full Health Canada indication was projected by the sponsor to be $10,930,471 over 3 years.
CADTH Appraisal of the Sponsor’s BIA
CADTH identified several key limitations to the sponsor’s analysis that have notable implications on the results of the BIA:
The number of patients eligible for mogamulizumab is uncertain. The sponsor used an epidemiological approach to estimate the target population size, with the prevalence of mycosis fungoides and Sézary syndrome based on sponsor-commissioned research involving US patients. As noted by the sponsor “the numbers may be expected to differ slightly from Canadian values.”17 The sponsor further notes that expected budgetary impact of reimbursing mogamulizumab is sensitive to the epidemiological inputs, in particular the estimated prevalence of Sézary syndrome.17 Between 1992 and 2010, a total of 6,685 cases were diagnosed in Canada, of which 2,620 were mycosis fungoides or Sézary syndrome.18 The incidence of cutaneous T-cell lymphoma increased during this period, and was observed to vary by region.18 Thus, the current number of patients with mycosis fungoides and Sézary syndrome in Canada is uncertain. CADTH additionally noted that the sponsor incorrectly calculated the number of eligible people in years 2 and 3, such that patients who receive mogamulizumab in year 1 (prevalent patients) would receive mogamulizumab again in years 2 and 3.
The sponsor assumed that 20% of patients and 65% of patients with mycosis fungoides and Sézary syndrome, respectively, would have received prior systemic therapy. The clinical experts consulted by CADTH noted that this was lower than expected in clinical practice. Of note, clinical experts indicated that up to 80% of patients with mycosis fungoides are likely to have had a prior systemic drug, while 100% of patients with Sézary syndrome are likely to have had a prior systemic drug.
CADTH corrected the sponsor’s BIA model such that years 2 and 3, only patients without mogamulizumab exposure in year 1 would be eligible for mogamulizumab treatment (i.e., incident patients). In the CADTH base case, 80% of patients with mycosis fungoides and 100% of patients with Sézary syndrome were assumed to have had prior systemic therapy. CADTH explored the impact of the prevalence and incidence of mycosis fungoides and Sézary syndrome in scenario analyses.
Limitations associated with the ECM comparator. The sponsor’s BIA considers costs related to mogamulizumab and ECM, which was assumed to be a basket comprised of pharmaceutical treatments (Table 9), with the frequency of use of each component of ECM based on a survey of experts conducted by the sponsor. As noted in the CADTH appraisal of the sponsor’s pharmacoeconomic submission, clinical experts consulted by CADTH for this review noted that the composition of ECM and the relative frequency of use of the components is not in line with clinical practice in Canada. The sponsor excluded brentuximab vedotin from the basket of ECM therapies; however, clinical experts consulted by CADTH indicated that brentuximab vedotin would be considered for patients with CD30+ disease. Of note, the reimbursement of several treatments included as part of ECM varies by jurisdiction.
CADTH was unable to address this limitation owing to a lack of data.
Uncertainty regarding the uptake of mogamulizumab. The sponsor assumed that, among patients with mycosis fungoides, the market share for mogamulizumab would be 1%, 2%, and 3% in year 1, year 2, and year 3, respectively, and, among patients with Sézary syndrome, the market share for mogamulizumab would be 20%, 30%, and 35%. The clinical experts consulted by CADTH for this review noted that this is likely a considerable underestimate of the uptake of mogamulizumab, especially among patients with Sézary syndrome, given the lack of efficacious alternative treatments. The clinical experts expect that, in each year of the BIA, 100% of patients with Sézary syndrome would receive mogamulizumab, while approximately 5% of patients with mycosis fungoides would receive mogamulizumab.
CADTH explored the impact of higher mogamulizumab uptake in scenario analyses.
Uncertainty regarding the duration of mogamulizumab treatment. In the calculation of drug costs, the sponsor assumed that all patients would receive 19.5 doses of mogamulizumab. As noted in the CADTH Clinical Report, the mean number of days of mogamulizumab exposure was 245.2 among those randomized to mogamulizumab, corresponding to approximately 8 months. . Based on this assumption, all patients who initiate mogamulizumab in Year 1 of the BIA would be assumed to discontinue mogamulizumab before the end of the first year (. This is in contrast with the sponsor’s pharmacoeconomic model, in which 8% of patients were assumed to remain on mogamulizumab 3 years after initiating treatment. CADTH notes that there was a wide range of durations of exposure among the patients randomized to mogamulizumab in the MAVORIC trial (minimum: 1 day; maximum: 1,328 days).
CADTH was unable to explore the impact of treatment duration owing to the structure of the sponsor’s model.
Costs associated with mogamulizumab were underestimated. As noted earlier, the sponsor assumed that patients would discontinue mogamulizumab after 19.5 doses were received. In the BIA model, no additional drug costs were incurred by these patients after discontinuation. This lacks face validity in that a proportion of patients who discontinue mogamulizumab would be likely to receive subsequent treatment (as assumed in the sponsor’s pharmacoeconomic submission). In the sponsor’s pharmacoeconomic model, subsequent treatment was assumed to comprise a basket of pharmacologic (bexarotene, interferon alfa, gemcitabine, CHOP, liposomal doxorubicin, prednisolone) and non-pharmacologic interventions (extracorporeal photopheresis, psoralen plus UVA phototherapy, total skin electron beam therapy), reflected by a one-time cost of $103,048 per patient ($83,377 excluding non-pharmaceutical interventions).
Drug costs associated with mogamulizumab were further underestimated owing to the exclusion of premedication costs. The mogamulizumab monograph2 recommends the administration of premedication (i.e., diphenhydramine, acetaminophen) for the first infusion and for subsequent infusions, if an infusion reaction occurs. In the MAVORIC trial, infusion-related reactions were more common with mogamulizumab compared with vorinostat (mogamulizumab: 33.2% of patients; vorinostat: 0.5%). Inclusion of such costs would increase the costs associated with mogamulizumab; however, CADTH notes that the reimbursement of diphenhydramine and acetaminophen is variable across jurisdictions.
In the CADTH base-case reanalysis, CADTH assumed that patients would receive subsequent treatment after the discontinuation of mogamulizumab.
CADTH Reanalyses of the BIA
CADTH revised the sponsor’s base case by adopting a higher proportion of patients with prior systemic treatment and assuming that patients receive subsequent treatment after discontinuation of mogamulizumab (Table 18).
Table 18: CADTH Revisions to the Submitted BIA
Stepped analysis | Sponsor’s value or assumption | CADTH value or assumption |
|---|---|---|
Corrections to sponsor’s base case | ||
1. Calculation of the number of eligible patients | Prevalent and incident patients were assumed to be eligible for mogamulizumab | In year 1, prevalent patients were assumed to be eligible for mogamulizumab. In years 2 and 3, incident patients were assumed eligible (i.e., prevalent patients would have received mogamulizumab in year 1 and would not be rechallenged) |
Changes to derive the CADTH base case | ||
1. Prior systemic treatment | Mycosis fungoides: 20% Sézary syndrome: 65% | Mycosis fungoides: 80% Sézary syndrome: 100% |
2. Treatment costs | Patients were assumed to receive no subsequent treatment after discontinuation of mogamulizumab | Patients were assumed to receive subsequent treatment after discontinuation of mogamulizumaba |
CADTH base case | Reanalysis 1 + 2 | |
BIA = budget impact analysis.
aThe cost of subsequent treatment was based on the sponsor’s predicted annual cost of established clinical management (excluding non-pharmacologic interventions).
Applying these corrections produced a 3-year budget impact estimate for reimbursing mogamulizumab for the treatment of mycosis fungoides and Sézary syndrome among patients with prior treatment exposure that was lower than the sponsor’s submitted base case. When CADTH applied changes to derive its base case, the total 3-year budget impact increased (i.e., was higher than the sponsor’s corrected base case). The results of the CADTH step-wise reanalysis are presented in summary format in Table 19 and a more detailed breakdown is presented in Table 20.
In the CADTH reanalysis, the 3-year budget impact of reimbursing mogamulizumab for the treatment of mycosis fungoides and Sézary syndrome among patients with prior treatment exposure is estimated to be $7,015,623. The disaggregated budget impact by patient subgroup is shown in Table 19.
Table 19: Summary of the CADTH Reanalyses of the BIA — by Patient Subgroup
Analysis | Three-year total ($) |
|---|---|
Submitted base case | |
Mycosis fungoides | 3,448,534 |
Sézary syndrome | 7,481,937 |
Full Health Canada indication | 10,930,471 |
Corrected sponsor base case | |
Mycosis fungoides | 774,661 |
Sézary syndrome | 2,181,755 |
Full Health Canada indication | 2,956,416 |
CADTH reanalysis 1a | 6,349,917 |
CADTH reanalysis 2a | 3,266,358 |
CADTH base case | |
Mycosis fungoides | 3,280,852 |
Sézary syndrome | 3,734,771 |
Full Health Canada indication | 7,015,623 |
BIA = budget impact analysis.
aPooled mycosis fungoides and Sézary syndrome.
CADTH also conducted additional scenario analyses to address remaining uncertainty, using the CADTH base case. Results are provided in Table 20.
Assuming a 10% higher annual incidence of mycosis fungoides.
Assuming a 10% higher prevalence of mycosis fungoides.
Assuming a 10% higher annual incidence of Sézary syndrome.
Assuming a 10% higher prevalence of Sézary syndrome.
Assuming that 20% of mycosis fungoides and 65% of Sézary syndrome patients have had prior systemic treatment, as submitted by the sponsor (i.e., applied to the total population of patients with mycosis fungoides and Sézary syndrome).
Assuming higher uptake of mogamulizumab, as indicated by clinical experts consulted by CADTH (100% of patients with Sézary syndrome are expected to receive mogamulizumab; 5% of patients with mycosis fungoides are expected to receive mogamulizumab).
Adopting a 51% price reduction of mogamulizumab.
Table 20: Detailed Breakdown of the CADTH Reanalyses of the BIA
Stepped analysis | Scenario | Year 0 (current situation) | Year 1 | Year 2 | Year 3 | Three-year total |
|---|---|---|---|---|---|---|
Submitted base case | Reference | 20,384,527 | 20,546,954 | 20,790,594 | 20,993,627 | 82,715,701 |
New drug | 20,384,527 | 22,843,825 | 24,570,941 | 25,846,880 | 93,646,172 | |
Budget impact | 0 | 2,296,871 | 3,780,347 | 4,853,253 | 10,930,471 | |
Corrected sponsor base case | Reference | 20,384,527 | 20,546,954 | 20,790,594 | 20,993,627 | 82,715,701 |
New drug | 20,384,527 | 22,793,472 | 21,127,022 | 21,367,097 | 85,672,117 | |
Budget impact | 0 | 2,246,518 | 336,428 | 373,470 | 2,956,416 | |
CADTH base case | Reference | 74,756,802 | 75,528,328 | 76,340,461 | 77,111,987 | 303,737,578 |
New drug | 74,756,802 | 81,062,984 | 76,978,141 | 77,955,274 | 310,753,201 | |
Budget impact | 0 | 5,534,655 | 637,681 | 843,287 | 7,015,623 | |
CADTH sensitivity analysis: Higher mycosis fungoides incidence | Reference | 75,162,869 | 75,975,001 | 76,746,527 | 77,518,053 | 305,402,450 |
New drug | 75,162,869 | 81,509,656 | 77,384,208 | 78,361,340 | 312,418,073 | |
Budget impact | 0 | 5,534,655 | 637,681 | 843,287 | 7,015,623 | |
CADTH sensitivity analysis: Higher mycosis fungoides prevalence | Reference | 81,375,683 | 82,269,028 | 83,121,768 | 83,974,507 | 330,740,985 |
New drug | 81,375,683 | 87,946,330 | 83,759,448 | 84,817,794 | 337,899,255 | |
Budget impact | 0 | 5,677,301 | 637,681 | 843,287 | 7,158,269 | |
CADTH sensitivity analysis: Higher Sézary syndrome incidence | Reference | 74,797,409 | 75,528,328 | 76,340,461 | 77,111,987 | 303,778,184 |
New drug | 74,797,409 | 81,062,984 | 77,013,380 | 77,996,809 | 310,870,582 | |
Budget impact | 0 | 5,534,655 | 672,920 | 884,823 | 7,092,397 | |
CADTH sensitivity analysis: Higher Sézary syndrome prevalence | Reference | 75,162,869 | 75,893,788 | 76,705,920 | 77,477,446 | 305,240,023 |
New drug | 75,162,869 | 81,685,206 | 77,343,601 | 78,320,734 | 312,512,409 | |
Budget impact | 0 | 5,791,418 | 637,681 | 843,287 | 7,272,386 | |
CADTH sensitivity analysis: prior systemic treatment (20% mycosis fungoides; 65% Sézary syndrome) | Reference | 20,384,527 | 20,546,954 | 20,790,594 | 20,993,627 | 82,715,701 |
New drug | 20,384,527 | 23,028,990 | 21,162,292 | 21,406,250 | 85,982,060 | |
Budget impact | 0 | 2,482,036 | 371,699 | 412,623 | 3,266,358 | |
CADTH sensitivity analysis: mogamulizumab uptakea | Reference | 74,756,802 | 75,528,328 | 76,340,461 | 77,111,987 | 303,737,578 |
New drug | 74,756,802 | 95,356,088 | 77,943,028 | 79,011,930 | 327,067,849 | |
Budget impact | 0 | 19,827,760 | 1,602,568 | 1,899,944 | 23,330,271 | |
CADTH scenario analysis: 51% price reduction | Reference | 74,756,802 | 75,528,328 | 76,340,461 | 77,111,987 | 303,737,578 |
New drug | 74,756,802 | 77,704,627 | 76,591,205 | 77,443,578 | 306,496,212 | |
Budget impact | 2,176,298 | 250,744 | 331,592 | 2,758,634 | 2,176,298 |
BIA = budget impact analysis.
aAssumed to be 2%, 3%, and 5% in year 1, year 2, and year 3, respectively, for mycosis fungoides and 100% in each year for Sézary syndrome.
Stakeholder Input
Patient Input
Lymphoma Canada, Canadian Skin Patient Alliance, and Cutaneous Lymphoma Foundation
About Lymphoma Canada, Canadian Skin Patient Alliance, and Cutaneous Lymphoma Foundation
Lymphoma Canada is a national Canadian registered charity that empowers the lymphoma community through education, support, advocacy, and research. Based out of Toronto (ON), we collaborate with patients, caregivers, healthcare professionals, and other organizations and stakeholders, to promote early detecting, find new and better treatments for lymphoma patients, help patients access those treatments, learn about the causes of lymphoma, and working together to find a care. Resources are provided for both English and French Canadians. For more information about our organization, please visit us at: www.lymphoma.ca
Information about the Canadian Skin Patient Alliance can be found at: https://www.canadianskin.ca/en/
Information about the Cutaneous Lymphoma Foundation can be found at: https://www.clfoundation.org/
Information Gathering
Lymphoma Canada (LC) in collaboration with CPSA and CLF, conducted an anonymous online survey of Cutaneous Lymphoma Patients, primarily Mycosis-Fungoides and Sezary Syndrome patients, between March 8, 2021 – September 21, 2021. Links to the surveys were sent via e-mail to patients registered through the LC database. The links were also made available via LC Twitter and Facebook accounts, Canadian and American Cancer Society message boards, Facebook groups for lymphoma patients and survivors, physicians specializing in cutaneous lymphoma across Canada and the USA, and lymphoma organizations’, primarily Cutaneous Lymphoma Foundation and the Canadian Skin Patient Alliance’s own contacts. The surveys had a combination of multiple choice, rating, and open‐ended questions. Skipping logic was built into surveys so that respondents were asked questions only relevant to them. Open ended responses to surveys that reflected the sentiment of a majority are included verbatim to provide a deeper understanding of patient perspectives.
There were 449 patients that responded to the survey that were diagnosed with either Mycosis Fungoides (MF) or Sezary Syndrome (SS). Of the patients diagnosed with MF/SS, there were 52 patients treated with Mogamulizumab that provided their experience. As this treatment is currently available in the USA, most responses were from this country. However, there were a few Canadian patients able to access this treatment and provide their experience. Of the patients with MF/SS who provided their demographic information (see Tables 1 and 2), 14% live in Canada, 54% are female, and 69% are ≥ 60 years old.
Table 1: Country of Survey Respondents (449 Respondents)
Respondents | CAN | USA | Europe | Other | Skipped | Total |
|---|---|---|---|---|---|---|
Patients WITHOUT Mogamulizumab experience | 42 | 189 | 19 | 18 | 129 | 397 |
Patients WITH Mogamulizumab experience | 4 | 44 | 3 | 0 | 1 | 52 |
Table 2: Gender and Age of Survey Respondents (Respondents)
Respondents | Age Range | Gender | Total | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
< 20 | 20 to 39 | 40 to 49 | 50 to 59 | 60 to 69 | 70 to 79 | 80 to 89 | Skipped | Female | Male | Prefer not to answer | Skipped | ||
Patients WITHOUT Mogamulizumab experience | 0 | 19 | 24 | 45 | 96 | 70 | 13 | 1 | 142 | 125 | 1 | 129 | 397 |
Patients WITH Mogamulizumab experience | 0 | 1 | 2 | 7 | 21 | 16 | 4 | 1 | 29 | 22 | 0 | 1 | 52 |
Disease Experience
Mycosis-Fungoides (MF) can be difficult to diagnose, with symptoms that can occur for many years before a diagnosis is achieved due to different presentations of the disease. Patient’s participating in this survey were for the majority diagnosed between 1-5 years ago (41%), with a portion of patients diagnosed over 10 years ago (26%). This means that there is a large portion of patients have been living with their disease for many years and have likely experienced a relapse or multiple relapses. Length of experience with disease could be a result of the number of treatment options available, indicating the important of having effective treatments in the relapsed/refractory setting.
Diagnosis can be very complicated for patients with CTCL and may take months to years to receive the correct diagnosis, as the symptoms of CTCL can masquerade as other more common skin conditions such as eczema or psoriasis. Patients were asked whether their MF was misdiagnosed as another skin condition before it was later correctly diagnosed as MF/SS. Only 23% of patients had their condition correctly diagnosed as MF/SS at presentation. The remainder of patients received diagnoses of eczema (40%), dermatitis (35%), psoriasis (21%) and allergic reaction (15%), among others. Further, 55% of patients took more than a year to receive their diagnosis, of which 22% received their diagnosis over 5 years after presentation of their symptoms. As such, patients may be diagnosed with more advanced stage disease due to difficulties in diagnosis and therefore there is a need for treatment options for advanced stage patients. The majority of patients (58%) have had their MF/SS relapse since diagnosis (449 respondents). These patients have experienced more patches over their body surface (39%), patches and/or raised plaques (20%), and lymph node involvement (11%). As two patients described:
“Because CTCL is a rare cancer and MDs were not considering CTCL as a diagnosis it took a while to reach a proper diagnosis as I was getting much worse.” – Anonymous Patient
“Symptoms started over 2 years ago, but my initial dermatologist just thought I had psoriasis and didn’t test until I went to the emergency.” - Anonymous Patient
MF/SS symptoms that most impacted affected patients’ quality of life at diagnosis (449 respondents) included visual patches or lesions (raised, scaly or discolored) (78%), itchiness of skin or lesions (57%), fatigue (28%), visual appearance of rash-like skin redness over the entire body (27%), pain or burning of skin or lesions (26%), and visual appearance of thick raised lesions (plaques) (26%).
Respondents were asked which aspects of their life, including mental and emotional problems, were NEGATIVELY impacted by their MF/SS symptoms and diagnosis. The majority of respondents (93%) had one or more symptoms negatively impact their quality of life (Table 3).
Table 3: Impact of MF/SS CTCL on Patients’ Mental and Emotional Well-Being (449 Respondents)
Impact | % of respondents | Impact | % of respondents |
|---|---|---|---|
Stress of Diagnosis | 70% | Difficulty sleeping | 36% |
Fear/worry of disease getting worse | 68% | self-conscious/embarrassment | 30% |
Anxiety/Worry | 59% | Depression | 30% |
Concerns with body image/physical appearance | 41% | Side effects such as fatigue | 29% |
Patients were asked about their current symptoms and impacts to their quality of life and wellbeing, as a change in disease and its impacts may have occurred since diagnosis. Symptoms that most commonly affected respondents’ quality of life currently (422 respondents) are similar to those at diagnosis and include visual appearance of skin patches or lesions (58%), itchiness of skin or lesions (52%), fatigue (32%), hair loss (23%), and pain or burning of skin or lesions (22%). Current wellbeing of patients has been impacted by fear/worry of disease progressing (58%), anxiety/worry (42%), side effects such as fatigue (36%), and stress of diagnosis (35%). Patients found that living with MF/SS has negatively impacted their personal image (32%), ability to sleep (27%), mental health (24%), and intimate relationships (23%) (rated 4-5 on a scale of 1 (no impact) to 5 (significant negative impact). Patients have reported:
“It's always with you. Makes you feel different from everyone else. No cure makes you feel like you never know what is going to happen.” – Anonymous Patient
“Emotional support is somewhat of an issue - because it's so difficult to explain, family & friends don't/can't understand what I'm experiencing.” – Anonymous Patient
“Prior to treatment the redness and scaly patches made me self-conscious of the appearance of my skin, especially when the disease progress to areas that were visible in summer clothes.” – Anonymous Patient
Experiences With Currently Available Treatments
Only 2% of patients were in a watch and wait period since there diagnosis with MF/SS, meaning that the remaining patients were receiving treatment (350 respondents). 327 patients provided information about their experience with MF/SS CTCL treatments. There are many treatment options that offer a wide variety of effectiveness but often do dimmish in effectiveness overtime. Such treatment options include systemic therapies, radiation treatment, light therapy and topical agents for patients, the top treatment options in each category have been summarized in Table 4. It should also be noted that many of these treatments are used in combination in order to provide greater efficacy.
Side effects of current treatments: The most common side effects respondents experienced by patients during their MF/SS CTCL treatments are listed in Table 5. 21% of patients did not experience side effects related to their treatments (350 respondents).
Table 4: Treatments for Patients With MF/SS CTCL (327 Respondents)
Topical Drug Treatment (105 not received / 327) | N (% of Respondents) | Light Treatment (84 not received / 274 | N (% of Respondents) | Systemic Treatment (92 not received / 211) | N (% of Respondents) | Radiation Treatment (115 not received / 181) | N (% of Respondents) |
|---|---|---|---|---|---|---|---|
Topical steroids | 294 (90%) | UVB light therapy | 145 (53%) | Interferon | 58 (27%) | Targeted skin electron bream therapy | 42 (23%) |
Retinoids | 92 (28%) | UVA light therapy | 57 (21%) | Methotrexate | 55 (26%) | Total skin electron beam (TSEB) therapy | 27 (15%) |
Compounded nitrogen mustard | 48 (15%) | PUVA light therapy | 56 (20%) | bexarotene (oral) | 50 (24%) | Local radiation therapy | 24 (13%) |
Mechlorethamine | 38 (12%) | Extracor-poreal Photopho-resis (ECP) | 48 (18%) | Mogamuli-zumab | 49 (23%) | — | |
Imiquimod | 26 (8%) | — | — | Romidepsin | 22 (10%) | — | |
Table 5: Side Effects From Treatment (350 Respondents)
Side effect (n) | % of resp. | Side effect (n) | % of resp. |
|---|---|---|---|
Fatigue (142) | 41% | Hair loss (39) | 21% |
Skin pain or burning (118) | 34% | Other (71) | 20% |
Skin irritation or rash (116) | 33% | Skin discoloration (81) | 23% |
Moderate/severe itching (106) | 30% | Peripheral Neuropathy (49) | 14% |
Respondents found fatigue, hair loss, severe itchiness, and skin burning and pain, to be the most difficult to tolerate side effects of treatment for their MF/SS (166 respondents).
Impact of treatments on quality of life: When asked about the impact of various aspects of treatment on their daily living (on a scale of 1 – 5, where 1= No impact and 5 = significant negative impact), respondents noted that the number of clinic visits and treatment-related fatigue had the most significant impact on their quality of life (Table 6).
Table 6: Impact of Treatment on Quality of Life (351 Respondents)
Treatment aspect | Minimal Impact (rating 2-3) | Significant negative impact (rating = 4-5) |
|---|---|---|
Number of clinic visits | 39% | 27% |
Treatment-related fatigue | 30% | 21% |
Number or frequency of infections | 19% | 7% |
Length of time of treatment administration | 35% | 19% |
Other side effects of treatment | 25% | 12% |
Patients were asked which areas of their life were negatively impacted by their treatments, using a similar rating scale as above. Patients rated work (21%), travel (21%), intimate relationships (19%) and ability to exercise (18%) to be most negatively impacted by their treatment. As patients have alluded:
“Couldn’t develop a new intimate relationship after a long marriage breaking up. Lesions, etc.” – Anonymous Patient
“Just all the multiple appointments made it difficult to work full time.” – Anonymous Patient
Access to treatment within the patients community is an important consideration. Certain treatments may only be available at specific tertiary cancer centers, and treatments may require frequent visits to the hospital which can impact patients quality of life and increase financial burdens. As treatment funding and therefore access can differ across Canadian provinces, and even within a province if only available at major cancer centres that are not accessible to rural patients, this can impact patients greatly. 79% of patients were able to access treatment within their local community, however 86% of respondents are from outside of Canada with different treatment access. A sub-analysis revealed that 32% of Canadians could not access treatment locally (47 respondents). For those that could not access treatment locally (21%), the impacts of not being able to receive treatment included long/exhausting travel to access treatment (28%), worry over survival/prognosis (25%), could not receive treatment (20%), and emotional hardship (18%) (338 respondents). As reported by two patients on their experience accessing treatment:
“[hospital] isn’t close, but I prefer going there than a local place with no specialist. Traveling there takes time and a lot of money in tolls and parking fees.” – Anonymous Patient
“I have to travel 7 hours to visit my doctor in Calgary who is a CTCL specialist. I live in a small town where we have a small local hospital, and no local doctors who specialize in CTCL.” – Anonymous Patient
Patients were asked about the financial impact that treatment has had. Out-of-pocket costs and costs related to treatment resulted in the greatest financial impact to patients, followed by travel and accommodation (Table 7).
Table 7: Financial Implications of Treatment for MF/SS Patients (338 Respondents)
Financial impact | Significant negative impact (rating = 4-5) |
|---|---|
Out of pocket costs | 24% |
Cost of treatment | 22% |
Prescription drug costs | 19% |
Travel/ accommodation for treatment access | 16% |
Time off work | 14% |
Improved Outcomes
Patients were asked whether there were enough treatment options available to them to manage/treat their MF/SS. 22% of patients were dissatisfied with both the number of treatment options available in both the frontline and relapsed/refractory setting (351 respondents). Though the majority state they do have access to enough treatment options, most patients responding to this survey are within the USA where there are more treatment options approved for use, including mogamulizumab. 81% of patients indicate it is extremely important to have an increased number of treatment options available (321 respondents). Further, it is important for patients to have a personal choice in their treatment (78%; 321 respondents). According to one patient: “I am always on the quest for more and better treatment options that will improve my overall quality of life and health.”
Patient preferences: Respondents were asked to rate, on a scale of 1 -5 (1 = not important; 5 = extremely important), the importance of various factors regarding a new drug or therapy for MF/SS CTCL. “Longer survival” and “better quality of life” compared to current therapies were rated as the most important outcomes for a new therapy to address (Table 8).
Table 8: Treatment Preferences (321 Respondents)
Treatment outcome or factor | Rating = 4-5 (Extremely important) |
|---|---|
Longer survival than current therapies | 82% |
Better quality of life than current therapies | 76% |
Longer treatment free periods than current therapies | 70% |
Easier or faster treatment application | 68% |
Fewer side effects than current therapies | 64% |
Respondents were asked if they would be willing to tolerate the side effects of a new treatment if they were short term. 68% (n=217) of respondents would be willing to tolerate potential short-term side effects, while 2% were not; the remaining were unsure (30%) for reasons depending on the type of side effect, duration, and cost-benefit ratio. Respondents were also asked if they would choose a treatment with known side effects, potentially serious, if their doctor recommended it was the best option for them. Of the 321 respondents who answered this question, 37% selected “Yes”, while only 9% selected “No”; the remainder were unsure for similar reasons above. Treatment side effects that patients do not want to tolerate for new treatments include viral reactivation (shingles) (48%), chemo-brain or fogginess (44%), increased number of infections (40%), and nausea/vomiting (38%) (321 respondents).
Symptoms of MF/SS that are most important for new treatments to control include itchiness (81%), skin ulcers and open sores (80%), pain or burning of the skin (77%), enlarged lymph nodes or nodules (72%), and skin patches or lesions (71%) (321 respondents).
Current gaps in accessing treatment according to patients included lack of resources and difficulties in accessing treatments:
“More studies in order to understand biological mechanism of [CTCL] and subsequent more effective treatment (progression free interval or complete healing).” – Anonymous Patient
“I need something to get rid of the red painful, itchy skin, but don't know where to turn. I am weary of 27 years of this, whether the redness is only the skin diseases or if it is mostly CTCL.” – Anonymous Patient
“If there are promising treatment options available, patients should have access to them. People need hope and the confidence that they will not be denied a potentially life- saving treatment.” – Anonymous Patient
“More treatments and treatments that don't have horrible side effects need to be available and need to be available to more doctors and in more facilities. If you live in a larger town, you are lucky. If you live in a small town, not so much.” – Anonymous Patient
Experience With Drug Under Review
52 patients (12% of respondents) received treatment with mogamulizumab. Patients largely accessed this treatment through private insurance (42%), public drug program (29%), compassionate access from a drug manufacturer (17%), and a clinical trial (8%). When specified, patients that have received mogamulizumab have received this therapy as their second line (14%), third line (24%), fourth line (24%), fifth line (14%), or 6th line or greater (24%) treatment (37 respondents).
12% of patients were able to complete their full course of mogamulizumab, while 44% are currently still receiving this treatment; 12% of patients had to stop this treatment due to side effects, while 10% did not have their symptoms controlled by this therapy (52 respondents). As a large percentage are still receiving treatment with mogamulizumab, data following treatment is limited. Only two respondents had their disease progress during treatment, while 25% were in disease remission (52 respondents).
Symptom Control and Treatment-Related Side Effects: Mogamulizumab was able to manage the major symptoms experienced by MF/SS patients including skin itchiness (62%), red skin patches/rash (56%), and skin pain (25%) (52 respondents). 36% of patients had all of their symptoms managed by mogamulizumab. The most commonly reported side effects of mogamulizumab treatment included fatigue/weakness (35%), skin rash or redness (29%), hair loss (19%), and neutropenia (19%).
Impacts to Quality of Life: 52 respondents provided details on whether their quality of life was impacted by various aspects of the treatment, rating this impact on a scale of 1 (no negative impact on my life) to 5 (significant negative impact on my life). The majority of patients did not experience significant negative impacts to their quality of life as a result of treatment and administration (Table 9).
Table 9: Impact of Mogamulizumab Administration on Patients’ Quality of Life (52 Respondents)
Aspect of mogamulizumab therapy | Rating 1-2 (Minimal Negative Impact (%) | Rating 4-5 (Significant Negative Impact) (%) |
|---|---|---|
Number of clinic/hospital visits required | 60% | 17% |
Length or Frequency of taking the drug | 62% | 15% |
Short-term side effects | 67% | 10% |
Long-term side effects | 62% | 19% |
Patients were asked about which aspects of their life were positively and negatively impacted by Mogamulizumab treatment and side effects (Table 10). The majority of patients were positively impacted by receiving mogamulizumab therapy.
Table 10: Impact of Mogamulizumab on Patients’ Quality of Life (52 Respondents)
Aspect of mogamulizumab therapy | Rating 1-2 (Negative Impact) (%) | Rating 4-5 (Positive Impact) (%) |
|---|---|---|
Ability to fulfill family obligations | 13% | 52% |
My personal image | 23% | 46% |
Ability to work or contribute to household finances | 12% | 42% |
Ability to exercise | 21% | 42% |
Ability to sleep | 21% | 42% |
Ability to perform household chores | 13% | 40% |
Ability to spend time with family & friends | 15% | 40% |
When asked how mogamulizumab treatment improved their overall health and well-being, 17% of patients indicated it was somewhat improved, with 50% stating it is greatly improved; 21% of patients were unchanged (52 respondents). As patients have commented:
“MOGA HAS MADE ME FEEL BACK TO NORMAL” – Anonymous Patient
“After 3 treatments my itching was gone and skin condition much improved.” – Anonymous Patient
Overall Experience and Recommendation Mogamulizumab Therapy: When asked to describe their experience with mogamulizumab, 69% of patients responded they had a good to excellent experience with the therapy, and 75% of patients mentioned they would take this treatment option again if available to them. Patients further commented:
“Mogamulizumab did everything I expected and managed my symptoms.” – Anonymous Patient
“Moga is the best treatment I have experienced.” – Anonymous Patient
Companion Diagnostic Test
There are no companion diagnostic tests to report on for this therapy.
Other Additional Information
N/A.
Patient Group Conflict of Interest Declaration — Lymphoma Canada, Canadian Skin Patient Alliance, and Cutaneous Lymphoma Foundation
To maintain the objectivity and credibility of the CADTH reimbursement review process, all participants in the drug review processes must disclose any real, potential, or perceived conflicts of interest. This Patient Group Conflict of Interest Declaration is required for participation. Declarations made do not negate or preclude the use of the patient group input. CADTH may contact your group with further questions, as needed.
Did you receive help from outside your patient group to complete this submission? If yes, please detail the help and who provided it.
Yes. |||||||||||||||||||| from the Cutaneous Lymphoma Foundation (CLF) and |||||||||||||||||||| from the Canadian Skin Patient Alliance (CSPA) both contributed to survey development, outreach to cutaneous lymphoma patients globally, and reviewed and provided feedback on the final report prior to submission.
Did you receive help from outside your patient group to collect or analyze data used in this submission? If yes, please detail the help and who provided it.
n/a
List any companies or organizations that have provided your group with financial payment over the past two years AND who may have direct or indirect interest in the drug under review.
Table 11: Conflict of Interest Declaration for Lymphoma Canada
Company | Check Appropriate Dollar Range | |||
|---|---|---|---|---|
$0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 | |
Kyowa Kirin | — | — | X | — |
Table 12: Conflict of Interest Declaration for Canadian Skin Patient Alliance
Company | Check Appropriate Dollar Range | |||
|---|---|---|---|---|
$0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 | |
No COI | — | — | — | — |
Table 13: Conflict of Interest Declaration for Cutaneous Lymphoma Foundation
Company | Check Appropriate Dollar Range | |||
|---|---|---|---|---|
$0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 | |
Kyowa Kirin | — | — | — | X |
Clinician Input
Ontario Health (Cancer Care Ontario) Hematology Cancer Drug Advisory Committee
About Ontario Health (Cancer Care Ontario) Hematology Cancer Drug Advisory Committee
Please describe the purpose of your organization. Include a link to your website (if applicable).
OH-CCO’s Drug Advisory Committees provide timely evidence-based clinical and health system guidance on drug-related issues in support of CCO’s mandate, including the Provincial Drug Reimbursement Programs (PDRP) and the Systemic Treatment Program.
Information Gathering
Please describe how you gathered the information included in the submission.
This input was jointly discussed at a DAC meeting.
Current Treatments
Describe the current treatment paradigm for the disease.
Current drug treatment options include: bexarotene, interferon, conventional chemotherapy, alemtuzumab (via compassionate access program), pralatrexate, brentuximab vedotin (BV), vorinostat, if the patient has not received them in prior treatment.
Other interventions include total skin electron beam radiation, chemo photopheresis and clinical trials.
Treatment Goals
What are the most important goals that an ideal treatment would address?
Disease control and improved health-related quality of life, including symptomatic improvement
Treatment Gaps (Unmet Needs)
Considering the treatment goals, please describe goals (needs) that are not being met by currently available treatments.
Many of the treatment options listed above are not publicly funded and may not provide sustained responses. Many patients eventually progress and require additional treatment.
Which patients have the greatest unmet need for an intervention such as the drug under review?
Patients who have progressed after at least 1 prior systemic treatment.
Place in therapy
How would the drug under review fit into the current treatment paradigm?
As per requested indication – after 1 prior treatment.
Please indicate whether or not it would be appropriate to recommend that patients try other treatments before initiating treatment with the drug under review. Please provide a rationale from your perspective.
There’s no standard of care/sequencing for this patient population. For MF patients with CD30+ disease and eligible, BV may also be recommended.
How would this drug affect the sequencing of therapies for the target condition?
Mogamulizumab would be an additional treatment option.
Which patients would be best suited for treatment with the drug under review?
As per the trial population.
How would patients best suited for treatment with the drug under review be identified?
No companion diagnostics required.
Which patients would be least suitable for treatment with the drug under review?
N/A
Is it possible to identify those patients who are most likely to exhibit a response to treatment with the drug under review?
No
What outcomes are used to determine whether a patient is responding to treatment in clinical practice?
Clinical improvement – e.g., regression of cutaneous disease; Sezary patients – improvement in pain, pruritus, blood work
What would be considered a clinically meaningful response to treatment?
Refer to previous answer.
Reduction in the frequency or severity of symptoms (provide specifics regarding changes in frequency, severity, and so forth)
Ability to perform activities of daily living
Improvement in symptoms
Stabilization (no deterioration) of symptoms
How often should treatment response be assessed?
Every cycle.
What factors should be considered when deciding to discontinue treatment?
Disease progression, adverse events, treatment-related toxicity
What settings are appropriate for treatment with the drug under review?
Outpatient clinic
For non-oncology drugs, is a specialist required to diagnose, treat, and monitor patients who might receive the drug under review?
N/A
Conflict of Interest Declarations
To maintain the objectivity and credibility of the CADTH drug review programs, all participants in the drug review processes must disclose any real, potential, or perceived conflicts of interest. This conflict of interest declaration is required for participation. Declarations made do not negate or preclude the use of the clinician group input. CADTH may contact your group with further questions, as needed. Please see the Procedures for CADTH Drug Reimbursement Reviews (section 6.3) for further details.
Did you receive help from outside your clinician group to complete this submission? If yes, please detail the help and who provided it.
OH-CCO provided secretariat support to the DAC in completing this input.
Did you receive help from outside your clinician group to collect or analyze any information used in this submission? If yes, please detail the help and who provided it.
No.
List any companies or organizations that have provided your group with financial payment over the past two years AND who may have direct or indirect interest in the drug under review. Please note that this is required for each clinician that contributed to the input — please add more tables as needed (copy and paste). It is preferred for all declarations to be included in a single document.
Declaration for Clinician 1
Name: Dr. Tom Kouroukis
Position: Provincial Head – Complex Malignant Hematology (OH-CCO)
Date: 16-Sep-2021
Table 14: Conflict of Interest Declaration for Ontario Health (Cancer Care Ontario) Hem DAC — Clinician 1
Company | Check Appropriate Dollar Range | |||
|---|---|---|---|---|
$0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 | |
Kyowa Kirin Canada — No COI | — | — | — | X |
Declaration for Clinician 2
Name: Dr. Jordan Herst
Position: Hematologist/oncologist
Date: 16-Sep-2021
Table 15: Conflict of Interest Declaration for Ontario Health (Cancer Care Ontario) Hem DAC — Clinician 2
Company | Check Appropriate Dollar Range | |||
|---|---|---|---|---|
$0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 | |
Kyowa Kirin Canada — No COI | — | — | — | — |
Declaration for Clinician 3
Name: Dr. Lee Mozessohn
Position: Hematologist/oncologist
Date: 16-Sep-2021
Table 16: Conflict of Interest Declaration for Ontario Health (Cancer Care Ontario) Hem DAC — Clinician 3
Company | Check Appropriate Dollar Range | |||
|---|---|---|---|---|
$0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 | |
Kyowa Kirin Canada — No COI | — | — | — | — |
Declaration for Clinician 4
Name: Dr. Mark Brown
Position: Clinical pharmacist
Date: 16-Sep-2021
Table 17: Conflict of Interest Declaration for Ontario Health (Cancer Care Ontario) Hem DAC — Clinician 4
Company | Check Appropriate Dollar Range | |||
|---|---|---|---|---|
$0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 | |
Kyowa Kirin Canada — No COI | — | — | — | — |
Canadian Cutaneous Lymphoma Providers
About Canadian Cutaneous Lymphoma Providers
Please describe the purpose of your organization. Include a link to your website (if applicable).
This Clinical Group is an expanding network of Canadian Cutaneous Lymphoma Providers. Due to the rarity of this subset of lymphoma, the heterogenous presentations, and challenging diagnoses, they are best managed in subspeciality clinic environments. An interdisciplinary collaborative approach is crucial comprising of hematologists/medical oncologists, dermatologists, radiation oncologists, and pathologists. Membership in international cutaneous lymphoma organizations expand our network and help gain expertise. Many of us participate in mentorships with other cutaneous lymphoma providers, wherein we review challenging cases or management issues.
Support and input for this submission has been provided by Dr. Lesley Street, Dr. Laurie Sehn, Dr. Gizelle Popradi, and Dr. Robert Gniadecki
Information Gathering
Please describe how you gathered the information included in the submission.
The information herein has been gained through discussions with colleagues at provincial tumour board or international cutaneous lymphoma meetings. As well, practical experience with the drug under review has been gained since the commencement of the special access program in January 2021.
Current Treatments
Describe the current treatment paradigm for the disease.
The management approach of mycosis fungoides/Sezary syndrome is individualized, based on extent of disease or stage, goals of therapy, patient tolerance, and practical considerations related to administration.
Limited stage mycosis fungoides is treated with skin-directed therapy including topical corticosteroids, ultraviolet light therapy, which is either narrow-band UVB, or PUVA (UVA combined with a photosensitizer), and less commonly topical carmustine or other topical agents. Some patients refractory or intolerant to these approached, unable to access light therapy, or with certain high risk pathological features, may require systemic therapy. In Canada, this generally is limited to interferon (recently alpha interferon was discontinued, so pegylated is now an alternative), isotretinoin (off-label use for MF), or oral methotrexate. Bexarotene which is a superior retinoid to isotretinoin, and standard of care outside of Canada, it is not approved by Health Canada, and not listed on provincial cancer formularies.
Advanced stage MF involving tumours may be treated with local radiotherapy, with similar systemic therapies as aforementioned. Advanced MF with extracutaneous nodal or visceral involvement requires systemic therapies such as chemotherapy (monotherapy with gemcitabine, or liposomal doxorubicin, if funding available. Many novel agents which are recommended by international guidelines are not funded in Canada, such as histone deacetylase inhibitors (vorinostat, romidepsin), or pralatrexate. Brentuximab was recently approved for use in CD30+ expressing MF, limited, or advanced stage, which has relapsed following one of these systemic approaches.
In Sezary syndrome, a frontline approach includes biweekly extracorporeal photopheresis, which is only available in some specialized urban centres. Systemic therapy such as interferon, and isotretinoin are typically added upfront, then further treatments are limited by available funded options. This may include romidepsin, alemtuzumab, or pembrolizumab.
Special access programs are currently available for mogamulizumab, in advanced stage MF (stage IIIB or higher), and Sezary syndrome. There is a special access program for romidepsin, although approved indications are limited to peripheral T cell lymphoma, which is a separate entity to MF/SS.
Another unique therapy which is available in 3 centres across Canada is Total Skin Electron Beam therapy, a highly specialized form of total skin radiation. It achieves excellent response rates, however, its broad application is limited by geography and interprovincial availability for this service (particularly during COVID-19).
Finally, select patients with advanced stage MF or SS and seeking potential curative intent treatment, may proceed to allogenic stem cell transplantation. Not all patients are eligible based on age, comorbidities, and donor availability. The long term disease free survival rates are generally less than 50% with this approach.
Treatment Goals
What are the most important goals that an ideal treatment would address?
Important clinical endpoints in the management of cutaneous lymphoma include prolonged survival, overall response rates, improvement in skin related symptoms, and quality of life.
Treatment Gaps (Unmet Needs)
Considering the treatment goals, please describe goals (needs) that are not being met by currently available treatments.
There are several treatment gaps for patients with MF and SS. These diseases have a prolonged but progressive course. Relapsed or refractory MF or SS is often characterized by extensive and disfiguring cutaneous involvement with intense skin related symptoms of itch, burning and pain. Many treatment options have suboptimal response rates (< 50%), or responses of limited duration (<4months). Often treatment selection in Canada is guided more by what drug is funded and available rather than necessarily the one with best chances of response, and fewest side effects. The treatments which must be delivered at specialized or urban centres (TSEB, ECP, or UV light) are practically not accessible to many patients living with cutaneous lymphoma. Some treatments may improve objective parameters, but patients may continue to experience skin symptoms, or impairment of quality of life or function.
Which patients have the greatest unmet need for an intervention such as the drug under review?
The patients with advanced stage MF or Sezary syndrome, as they have the greatest disease burden, typically, the greatest symptom burden, and the currently available treatment options have suboptimal response rates and duration. Also, these patients sometimes cannot access TSEB or ECP, so systemic therapy would be a better option. They are a subpopulation of the broader group of patients with limited stage MF, who predicably progress over many years to decades to advanced stages.
Place in Therapy
How would the drug under review fit into the current treatment paradigm?
Mogamulizumab is a first-in-class monoclonal antibody directed against chemokine receptor 4, targeting the underlying mechanism of lymphocyte trafficking to skin. In a phase III RCT it was demonstrated to prolong progression free survival and had superior response rates to the comparator. The drug was studied in second-line, following at least one prior systemic therapy. It would likely cause a shift in current therapy, and be considered after chemo-monotherapy, brentuximab (if applicable CD30+), or interferon for advanced MF, or following interferon and/or retinoid (isotretinoin) in Sezary syndrome. With the mechanism of action regulating malignant lymphocyte trafficking, it is compelling to consider its concurrent use with extracorporeal photopheresis which irradiates these circulating lymphocytes ex-vivo.
Please indicate whether or not it would be appropriate to recommend that patients try other treatments before initiating treatment with the drug under review. Please provide a rationale from your perspective.
It would be sensible to use mogamulizumab after failing the above-mentioned therapies. Firstly, mogamulizumab was not studied in the upfront setting, and some patients derive benefit from existing therapies, before ultimately progressing and requiring an alternate.
How would this drug affect the sequencing of therapies for the target condition?
The main impact of sequencing therapies following mogamulizumab is avoidance of allogeneic transplantation approximately 4 months after due to reports of increased graft-versus-host. This data is derived from mogamulizumab’s previous application in another lymphoma (adult T-cell Leukemia/Lymphoma) and likely more information on the GVH risk and timing will be gained with post-marketing surveillance experience. It would be unlikely a patient would be retreated with mogamulizumab based on existing evidence.
Which patients would be best suited for treatment with the drug under review?
The group most suitable for mogamulizumab are those with advanced stage MF and/or with blood involvement (Stage IIIB, IVA, IVB), or Sezary syndrome. These patients are also at highest need of intervention, have the worse prognosis, and are most likely to respond.
How would patients best suited for treatment with the drug under review be identified?
The diagnosis of CTCL (MF/SS) is complex and requires the clinicopathological correlation by experts in the field. Patients are most likely identified by a multidisciplinary group of doctors not limited to hematologists, medical oncologists, dermatologists, radiation oncologists, and hemato-/dermatopathologists. There is some overlap of clinical signs and symptoms which overlap with benign skin disorders. Accurate diagnosis requires thorough skin examination and appropriate documentation of morphology, skin biopsy with histopathological examination, along with ancillary testing which may include blood flow cytometry and/or molecular testing. Some labs are limited in their tools or expertise in establishing these diagnoses. For all of these reasons, there is typically significant delay or several years before a diagnosis of skin lymphoma is established. There are no pre-symptomatic testing that would be relevant for the drug under review.
Which patients would be least suitable for treatment with the drug under review?
Patients with stage IA MF were not included on the clinical trial, so should not be treated with mogamulizumab. Due to the mechanism of action of mogamulizumab, the responses outcomes demonstrated on study, those with stage IB or IIA/B are less likely to derive benefit.
Is it possible to identify those patients who are most likely to exhibit a response to treatment with the drug under review?
These patients would be identified by stage, and response to prior therapy.
What outcomes are used to determine whether a patient is responding to treatment in clinical practice?
Skin examination by individual with expertise, preferably mSWAT score (a validated assessment tool), quality of life assessments (e.g. Skin-Dex 29), serial diagnostic imaging in the case of nodal/extra-cutaneous disease, and CBC +/- flow cytometry for Sezary count. There are defined response criteria for MF/SS in the literature (Olsen et al, JCO 2011).
What would be considered a clinically meaningful response to treatment?
An objective improvement of skin typically corresponds with improvement of other measures of disease burden in blood, lymph nodes, and viscera. Improvement of patient quality of life is paramount in these malignancies, generally considered incurable. Subjective improvement in quality of life can be considered clinically meaningful endpoint. Improvement in specific symptom parameters such as pruritis (itch) is very crucial for some patients living with MF or SS.
How often should treatment response be assessed?
Treatment responses may be documented in accordance to published guidelines (Olsen et al, JCO 2011), as compartmentalized and composite responses. Close clinical follow-up is required to assess for infusion-related reactions, and the emergence of a well characterized mogamulizumab-related rash. Expert opinion and collaboration with dermatology is critical to distinguish this rash from underlying skin pathology. Biopsy is sometimes required.
What factors should be considered when deciding to discontinue treatment?
Disease progression is characterized in each of the disease compartments (skin, nodal, and blood) and there are published criteria for progressive disease (JCO 2011) in which case discontinuation advised. One may consider continuing therapy with stable disease if treatment tolerated. Adverse events, notably mogamulizumab -associated rash not responding to management algorithms, or other adverse events would be reason to discontinue.
What settings are appropriate for treatment with the drug under review?
This drug would typically be administered in an ambulatory setting at an oncology centre, or an infusion centre under the supervision of trained personnel.
For non-oncology drugs, is a specialist required to diagnose, treat, and monitor patients who might receive the drug under review?
N/A
Additional Information
Is there any additional information you feel is pertinent to this review?
N/A
Conflict of Interest Declarations — Canadian Cutaneous Lymphoma Providers
To maintain the objectivity and credibility of the CADTH drug review programs, all participants in the drug review processes must disclose any real, potential, or perceived conflicts of interest. This conflict of interest declaration is required for participation. Declarations made do not negate or preclude the use of the clinician group input. CADTH may contact your group with further questions, as needed. Please see the Procedures for CADTH Drug Reimbursement Reviews (section 6.3) for further details.
Did you receive help from outside your clinician group to complete this submission? If yes, please detail the help and who provided it.
No
Did you receive help from outside your clinician group to collect or analyze any information used in this submission? If yes, please detail the help and who provided it.
Lymphoma Canada helped to coordinate the group of clinicians for this submission, however they were not involved analyzing or adding feedback to any of the responses in this submission.
List any companies or organizations that have provided your group with financial payment over the past two years AND who may have direct or indirect interest in the drug under review. Please note that this is required for each clinician that contributed to the input — please add more tables as needed (copy and paste). It is preferred for all declarations to be included in a single document.
Declaration for Clinician 1
Name: Dr. Lesley E Street
Position: Hematologist, Tom Baker Cancer Centre. Co-Director Calgary Cutaneous Lymphoma Program; Clinical Associate Professor, University of Calgary
Date: 01-10-2021
Table 18: Conflict of Interest Declaration for Canadian Cutaneous Lymphoma Providers — Clinician 1
Company | Check Appropriate Dollar Range | |||
|---|---|---|---|---|
$0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 | |
Kyowa Kirin | — | X | — | — |
Seattle Genetics | X | — | — | — |
Recordati | X | — | — | — |
Declaration for Clinician 2
Name: Dr. Laurie Sehn
Position: Medical Oncologist, BC Cancer Agency
Date: 07-10-2021
Table 19: Conflict of Interest Declaration for Canadian Cutaneous Lymphoma Providers — Clinician 2
Company | Check Appropriate Dollar Range | |||
|---|---|---|---|---|
$0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 | |
No COI | — | — | — | — |
Declaration for Clinician 3
Name: Dr. Gizelle Popradi
Position: Hematologist, McGill University Health Centre
Date: 07-10-2021
Table 20: Conflict of Interest Declaration for Canadian Cutaneous Lymphoma Providers — Clinician 3
Company | Check Appropriate Dollar Range | |||
|---|---|---|---|---|
$0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 | |
Kyowa Kirin | X | — | — | — |
Servier | — | X | — | — |
Seattle Genetics | — | X | — | — |
Mallinckrodt | X | — | — | — |
Declaration for Clinician 4
Name: Dr. Robert Gniadecki
Position: Section Chief, Dermatology, AHS Edmonton Zone; Director, Division of Dermatology; Professor, University of Alberta
Date: 08-10-2021
Table 21: Conflict of Interest Declaration for Canadian Cutaneous Lymphoma Providers — Clinician 4
Company | Check Appropriate Dollar Range | |||
|---|---|---|---|---|
$0 to 5,000 | $5,001 to 10,000 | $10,001 to 50,000 | In Excess of $50,000 | |
Mallinckrodt | X | — | — | — |
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up-to-date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third-party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.