CADTH Horizon Scan
2023 Watch List: Top 10 Precision Medicine Technologies and Issues
Round Up
Authors: Sinwan Basharat, Andrea Smith, Nazia Darvesh, Tamara Rader
Authors
Sinwan Basharat, Andrea Smith, Nazia Darvesh, and Tamara Rader
Contributors
The authors are grateful to CADTH staff, Kristen Moulton, Francesca Brundisini, Barbara Greenwood Dufour, Aaron Naor, Kathleen Burns, Yan Li, Sharon Bailey, and Brandy Appleby for their involvement in the project, including providing feedback on the report, supporting the workshop, and/or project management.
Advisory Group
CADTH is grateful to the advisory group for the 2023 Watch List. Its group members provided project oversight, considerations about items to include, helped refine the short list, and reviewed earlier versions of the draft report.
Jonathan Kimmelman, PhD
Director, Biomedical Ethics Unit
Professor, McGill University
Valerie McDonald, MSWa
Patient Member
Ontario Palliative Care Network’s Models of Care Pediatric Working Group
Regulation Policy and Economics Matrix of the Canadian Pediatric Cancer Consortium
Yvonne Bombard, PhDa
Associate Professor, University of Toronto
Director and Scientist, Genomics Health Services Research Program, Unity Health Toronto
aThese members also participated in the final workshop.
Workshop Participants
CADTH is grateful to the workshop participants for their time, sharing their expertise and experiences, and providing input to help select the final items included in the 2023 Watch List. Their participation, insight, and willingness to collaborate were integral to the list.
Eesha Sharma, PhD
CEO and Cofounder, Lamar Health
Gregory Costain, MD, PhD
Physician, The Hospital for Sick Children
Assistant Professor, University of Toronto
Ian Stedman, MA, LLB, LLM, PhD
Assistant Professor, York University
Ivana Cecić, PhD
Director of Health Programs, Genome Canada
Ma’n H. Zawati, LLB, LLM, PhD (DCL)
Assistant Professor, McGill University
Executive Director, Centre of Genomics and Policy
Monette Greenway
Principal and Cofounder, Precision Rx-Dx Inc.
Maureen Smith, MEd
Patient Member, Ontario Health Technology Advisory Committee
Patient Member, Ontario Committee to Evaluate Drugs
Shantanu Banerji, MD, FRCPC
Director of Precision Oncology and Advanced Therapeutics, CancerCare Manitoba
Associate Professor, Department of Internal Medicine, Rady Faculty of Health Sciences, University of Manitoba
Tanya Nelson, FCCMG, PhD
Division Head, Division of Genome Diagnostics, BC Children’s Hospital and BC Women’s Hospital
Clinical Professor, Department of Pathology and Laboratory Medicine, University of British Columbia
Conflicts of Interest
Advisory group members declared the following conflicts: Johnathan Kimmelman has received speaking and consulting fees from 3 pharmaceutical organizations in the past, Valerie McDonald has a family member with past employment as a representative for a pharmaceutical organization, and Yvonne Bombard is a cofounder and the CEO of Genetics Adviser.
Workshop participants declared the following conflicts: 1 member received consultant fees from 2 precision medicine technology organizations, 1 member has academic research grants related to precision medicine technologies, 1 member received consultant fees from 1 pharmaceutical organization, 1 member received research funding from a precision medicine technology organization and has a family member who is employed at a precision medicine technology organization.
Abbreviations
HTA
health technology assessment
JLA
James Lind Alliance
NGS
next-generation sequencing
NSCLC
non–small cell lung cancer
PCR
polymerase chain reaction
Key Messages
CADTH’s 2023 Watch List presents the top 10 precision medicine technologies and issues that have the potential to make a significant and meaningful impact in transforming health systems in Canada over the next 5 years. These technologies and issues are likely to shape the future of health care.
The Human Genome Project was completed 20 years ago and offers new opportunities to use an individual's genetic information to manage their health care. Today, as health systems see an emergence of new precision medicine technologies that can use a person’s unique characteristics to inform and tailor their care, the 2023 Watch List unpacks the hype from hope, and indicates where there is likely real opportunity to improve patient care and health systems delivery.
As part of the 2023 Watch List, we identify and describe the 5 top precision medicine technologies; among them, examples such as liquid biopsies for informing cancer treatments and pharmacogenomics testing for mental health conditions that may shape the future of health care. To prepare health systems for their wider adoption and implementation, the 2023 Watch List explores some considerations for health care decision-makers about the impact and implications the technologies may have on care pathways, health human resources, health care infrastructure, and health equity.
The 2023 Watch List also identifies the top 5 issues that crosscut the broader scope of precision medicine technologies and could limit health systems from realizing the full potential of the technologies. Among them, issues such as increasing complexity related to interpreting test results and challenges in regulating precision medicine technologies are key issues that likely warrant more attention and will influence the wider adoption, diffusion, and implementation of precision medicine technologies.
Collectively, monitoring ongoing developments and evidence related to the top technologies and issues highlighted in the 2023 Watch List can help support health care stakeholders to future-proof health systems and guide health system planning in Canada.
Introduction
CADTH routinely identifies new and emerging technologies that are likely to have a significant and meaningful impact on Canada’s health care systems. Through regular monitoring of various health information resources, CADTH supports decision-makers by informing them about emerging health technologies to prepare for their potential introduction, availability, and wider adoption. CADTH’s annual watch lists1 specifically identify technologies that have the most potential to transform health systems and shape the future of health care in Canada. The annual list anticipates how innovations within technology may affect future health system needs and provides early signals to help guide health system planning. Building on previous lists that examined health technologies across different health conditions and aspects of care, the 2023 Watch List delves deeper into health technologies within a single theme, focusing on key developments within precision medicine technologies.
The Promise of Precision Medicine
Precision medicine2 is an approach that uses information about individuals’ unique biological, environmental, and health behavioural characteristics to inform and tailor their care. It enables a shift in certain aspects of health care away from a ‘one-size fits all’ approach to an alternative where a person’s unique characteristics and circumstances are used to inform their care. Since the completion of the Human Genome Project in 2003, precision medicine has opened up new opportunities to prevent, diagnose, and treat many health conditions using genomic information (DNA or RNA).3 For example, it has led to new technologies that can identify people with an increased risk of chronic health conditions such as Alzheimer disease, the development of tools that can improve the detection and diagnosis of rare diseases, and the proliferation of drug therapies targeted to specific mutations within cancer care.4-6 Advancements in sequencing technology and data analytics, along with investments into research and large genomic-based initiatives in Canada and internationally, are accelerating the pace of innovation of precision medicine technologies.4,7-10
While the increased development of precision medicine technologies raises an appealing proposition for delivering more targeted care that could improve outcomes, the transformation also brings more complexity to and for health systems. Should these technologies see increased adoption into routine clinical use, they are likely to change multiple aspects of care including pathways of care, health and human resources, health care infrastructure, patient outcomes and experiences, and raise equity considerations.4,11-13 To alert decision-makers to innovations within precision medicine and possible implications for health systems, the 2023 Watch List identifies new and emerging precision medicine technologies and crosscutting issues related to the technologies that are likely to have a significant impact on health care systems in Canada over the next 5 years.
Developing the 2023 Watch List
Given that precision medicine is a broad field, the scope of the 2023 Watch List is limited to medical devices, clinical interventions, or other health care technologies that can detect, characterize, or use people’s genomic or protein information to inform and tailor their care (refer to Appendix 1). It does not examine precision medicine therapies or other technologies that are defined as drugs by Health Canada (such as most stem cell products), or technologies that are not directly part of clinical care with patient outcomes (e.g., health care information technology software).
To be considered for the list, the technologies also had to be new or emerging; that is, not yet available or widely adopted in Canada beyond a limited number of health care centres or initiatives. Specific examples of technologies, ongoing research studies, or other initiatives are also included to illustrate the items in practice and are provided for general awareness. Items on the 2023 Watch List are not ranked in order of importance, potential impact, or timing of that impact, but some technologies are further along in their development than others. This year’s watch list also identifies the top issues; that is, the policy, social, legal, ethical, or regulatory considerations that crosscut the broader scope of precision medicine technologies and could affect their ongoing development, adoption, or implementation in Canadian health systems. Although effort was made to separate issues into discrete items, due to the interconnectedness of themes, there may be some overlap. The list is not an endorsement of any specific technology and is intended to be used for informational purposes only.
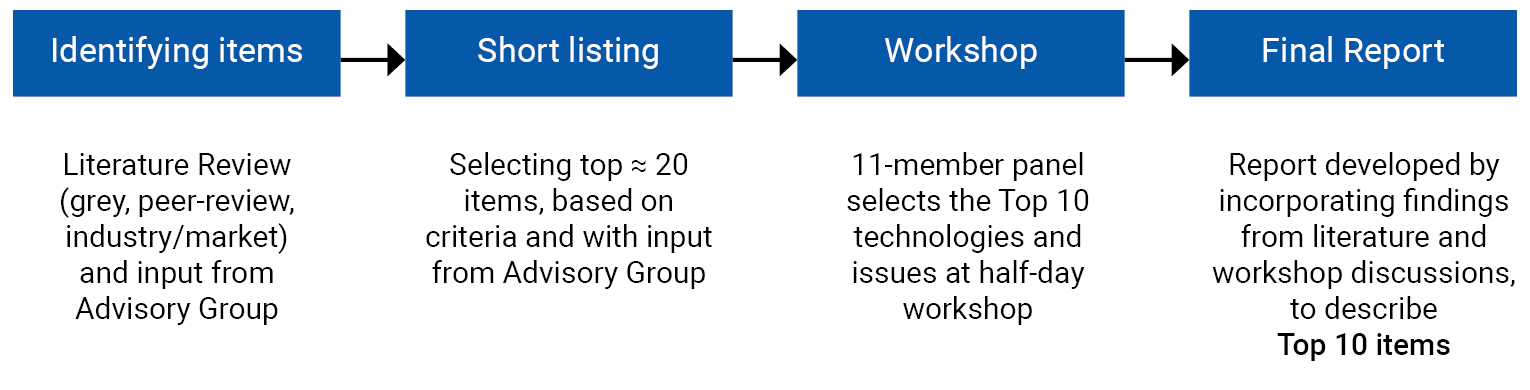
CADTH developed the 2023 Watch List using a modified James Lind Alliance (JLA) priority-setting approach. A short list of emerging technologies and issues was identified from published literature and industry news with input from an expert advisory group. The final items on the 2023 Watch List were selected by a panel through a consensus-based decision-making process. The panel brought together diverse views and experiences, and included patient partners, people with lived experiences, policy and legal experts, researchers, industry representatives, and health care professionals from across Canada. Further details about the selection and identification process of the items are described in Appendix 1.
Watch List Objectives
The 2023 Watch List aims to highlight the precision medicine technologies that are poised to have the most significant and meaningful impact to health care decision-makers and that are likely to shape the future of health care in Canada over the next 5 years. It also serves as a guide to separate some of the hype from hope about the prospect of these technologies and describes some of their related uncertainties. The top issues identified in the 2023 Watch List point decision-makers toward the policy, social, ethical, and legal considerations that underpin developments within precision medicine technologies. Contextualizing the technological developments alongside the broader issues can provide insight about the future of precision medicine in Canada and support health systems planning. Collectively, the top 5 precision medicine technologies and top 5 issues constitute the 2023 Watch List.
Top Precision Medicine Technologies to Watch
Digital Tools to Access Genetic Information and Navigate Care
A 2020 survey of people in Canada showed that there was a 35% increase in access to online health data since 2019, and that 79% of people wanted electronic access to their personal health information.14 Digital tools such as patient portals and other applications give clinicians, patients, and families direct access to individualized health information. In the context of precision medicine, these technologies can help people connect with their genetic test results, understand the implications of findings, and provide them with opportunities for meaningful engagement with health care professionals to discuss results that could help improve their health outcomes.15,16 With an emergence of tests such as whole genome sequencing or larger gene panels that collect information about hundreds of genes or other biomarkers, the size and complexity of information can be overwhelming. Tools that can help make sense of that complexity and provide patients, family members, and health care professionals with a platform to contextualize genetic information could help improve the patient experience, support shared decision-making, and support patient-centred care.17
A systematic review reported there are at least 70 different tools for delivering genetics care, including web-based portals, applications, and digital tools that may be used for clinical assessment, patient education, pretest and posttest counselling, and reporting results.18 Should some of these (or future) digital tools see increasing adoption and implementation over the next 5 years, they will likely raise several opportunities and considerations for health systems. For example, amid increasing demand for genetic testing, these technologies could help improve access to patient counselling services delivered by clinical geneticists, genetic counsellors, or primary care providers.17 Some digital technologies may improve workflow efficiencies by reducing administrative time spent preparing charts or other materials.18
Similar to the broader digital health space, the growth prospect of digital tools for genetics underscores the growing importance of patient data privacy and security, which are key concerns for people in Canada.19 Continuing to enhance data privacy infrastructures will be pertinent for addressing safety concerns, as genetic test results from medical records will need to connect seamlessly with patient-facing digital tools while still ensuring sensitive information, such as genetic risk factors, remains secure and confidential.16,20 The technologies’ wider diffusion across Canada will likely also be dependent on digital health equity considerations, including the potential benefits of technology to overcome geographical barriers to access, initiatives to improve health literacy, and supporting technology infrastructure (e.g., internet access in rural and remote areas).15
Examples of emerging digital tools to watch include:
Genetics Adviser21 (Toronto, Canada) is a digital genetic counselling and education support tool that is intended to guide patients throughout their pathways of care. It provides information to people before testing, helps describe the potential benefits and harms of different tests, and provides a guide to understanding test results and implications of findings, as well as a lifelong record of their genetic results and updates over time.21 The digital tool is adaptable to multiple genetic test types, populations, and clinical settings. A mixed-methods randomized control trial is under way to assess the effectiveness of the tool when accompanied with genetic counselling (compared with genetic counselling alone) on different outcomes such as impact on decision-making, patient experience, and economic costs.22 The project is supported in part by the Canadian Institutes of Health Research, St. Michael’s Foundation, and the McLaughlin Centre at the University of Toronto.22
ShareDNA23 (Seattle, US) is a free smartphone application that helps patients upload and share their genetic test results securely with others and learn how to interpret results without the need to input a large amount of personal data.24 An early user-testing study reported that people felt comfortable with using the application but also indicated a need to provide more information about how data are kept secure.24 The project is supported in part by the National Institutes of Health and the Kaiser Permanente Washington Health Research Institute in the US.24
Liquid Biopsy for Informing Cancer Treatments
Liquid biopsy25 is an emerging technology that detects genomic information in bodily fluids and could alter traditional pathways of care for cancer. While traditional approaches of precision-based testing rely on surgical biopsies, liquid biopsies could shift existing approaches toward less invasive testing technologies. Primarily aimed at detecting cell-free DNA in blood, these technologies may also be capable of detecting DNA from urine, saliva, or cerebrospinal fluid.26 The technology is based on growing evidence that among certain cancers, tumour cells can release DNA into bodily fluids.26 These DNA fragments may have a distinct genomic profile compared with DNA shed from healthy cells and, with the appropriate testing technology, can help describe features about the cancer that can be important for guiding treatment. Although different types of liquid biopsy–based testing technologies exist, tests that are used to guide treatment decisions after a cancer diagnosis, rather than tests used for screening, were specifically selected as 1 of the top precision medicine technologies to watch over the next 5 years. While the majority of these technologies that are currently available are single-gene assays or multigene panels that use some form of polymerase chain reaction (PCR) technology, next-generation sequencing (NGS) technologies that assess a multitude of genomic targets are increasingly being developed and adopted.26,27
Liquid biopsy can also provide information about suitable therapy targets (for example, as a companion diagnostic), therapeutic response (such as drug resistance), cancer progression (including evolution of the tumour over time), and signs of recurrence using minimally invasive approaches (i.e., blood based assays).28 Among people for whom surgical biopsies could be associated with elevated risks or when performing multiple surgical biopsies throughout a person’s clinical journey may not be feasible, liquid biopsies could provide an alternative or complement to tissue-based assays and help improve the patient experience and clinical outcomes.26,29 Although the technology has seen the greatest adoption for informing care for people with non–small cell lung cancer (NSCLC) because of the cancer’s heterogeneity and different possible treatment options, liquid biopsy may become more commonly used in the next 5 years for additional cancer types, such as colon, breast, ovarian, and prostate.29,30
Emerging research about the diagnostic accuracy (which is typically lower than that of tissue-based assays), clinical utility, and cost-effectiveness of liquid biopsy tests will be important to monitor. Should liquid biopsy be more widely adopted, health systems will likely need to consider its infrastructure requirements, including the upfront equipment investment, personnel training, and laboratory capacity needed to conduct such tests.29 Especially in the case of NGS-based tests, health systems likely need to consider whether centralization (compared to a decentralized or regional-based testing infrastructures) may be a more sustainable approach to implement testing and analysis.29 Evaluating liquid biopsy for reimbursement can be complex and learning from its early use and adoption for NSCLC can guide ongoing development.31 Continuing to engage with patients and health care professionals to understand perspectives about the technology and how it is used in care pathways can also help guide future adoption.32 Should the use of liquid biopsy expand from limited cancer types and a few tertiary research centres to more cancer types and as part of routine care in more centres, the wider diffusion of the technology may provide more equitable access within and across jurisdictions in Canada.
Examples of emerging technologies and their assessments include:
Comprehensive genomic profiling technologies are NGS-based, liquid biopsy tests that can characterize up to hundreds of genes, inform treatment selection, and monitor therapeutic response for NSCLC and other solid tumours with a single sample.31
Blood tests to detect EGFR mutations33 are PCR-based tests developed by different manufacturers. These tests identify mutations in the EGFR gene, which can confer resistance to certain drugs, to help guide treatment selection for NSCLC.
The PREDiCT trial ongoing in British Columbia34 is examining the detection rate, clinical outcomes, and cost implications of using liquid biopsy. The randomized open-label trial is comparing molecular characterization of tissue-based testing with liquid biopsy–based NGS testing (using FoundationOne Liquid CDx) to inform treatment decisions for multiple cancer types.
“Omics-Based” Sequencing Technologies Across Pathways of Care
Whole genome (the entire DNA code of an individual), exome (the DNA code of all protein coding regions), and transcriptome (global gene expression as measured by RNA) sequencing are precision medicine technologies that are referred to as “omics-based” technologies because they characterize the complete or comprehensive set of molecular information of individuals’ cells and tissues.35 This approach is in contrast to targeted approaches that examine a limited number of genes, genomic areas, or proteins. The comprehensive nature of omics-based sequencing technologies allows for a single test to be performed at the onset of clinical presentation rather than a series of individual tests, helping to improve diagnostic information and possibly increase speed of diagnosis.36-38 Although whole genome, exome, and transcriptome sequencing have been used extensively in research settings, in the next 5 years these technologies may see substantially more implementation into routine care due to their potential for providing a more precise and wider span of information compared to targeted- or single-gene tests.12 Their adoption across the continuum of care, from diagnosis to classification to treatment, could change health care in Canada.
Whole genome and exome sequencing offer an important value proposition for rare diseases, pediatric conditions, and adult conditions that affect different organ systems.39 For example, these technologies may reduce diagnostic delays for treating critically ill infants with unknown conditions, which could lead to better outcomes in acute care settings.40 Profiling tumours with these technologies can also help inform diagnoses and identify actionable mutations (i.e., genetic variants that can be targeted by therapy) for people with rare or unknown cancers.41,42 For more prevalent cancers, although there are clinical benefits of omics-based sequencing, the costs and interoperability reduce the feasibility of using the technology in routine care.43-45 With an increasing number of precision-based therapies targeting specific mutations, decreasing costs of sequencing, and the continuing growth of studies assessing implementation in real-world settings, the next 5 years may see omics-based technologies transform diagnosis and treatment care pathways.12,46
Should implementation of whole genome, exome, or transcriptome sequencing be further adopted into routine care, health care decision-makers will need to enhance or grow existing infrastructures to conduct these tests (including personnel training, equipment, and sample storage) and analyze test results, which are inherently more complex than results from traditional assays as they require greater bioinformatics expertise.12 Health systems also need to consider additional test result–related challenges that may need to be mitigated. These challenges could include uncertainty about identifying incidental findings that could affect patients and their families (e.g., potential for overdiagnosis and anxiety about results), interpreting complex findings, and being aware of limitations in genomic databases that may affect the relevance of findings due to underrepresentation of certain demographics in reference genomes.12,47,48
Examples of emerging technologies and initiatives to watch related to omics-based sequencing include:
Ultrarapid nanopore genome sequencing49 technology aims to speed up whole genome sequencing processes to deliver results in fewer than 8 hours. Preliminary study findings suggest the technology could be useful in critical care settings, such as neonatal intensive care units, where rapid sequencing could provide more timely results, reducing the need for a cascade of other diagnostic procedures.
The Marathon of Hope Cancer Centres Network50 has created interprovincial networks and is conducting pilot projects across Canada to expand the availability of whole genome and transcriptome sequencing and its necessary infrastructure to support the implementation of precision medicine technologies in routine care. Potentially thousands of people with different cancers who otherwise would not have had access to precision-based therapies may directly benefit. Continuing to watch the findings and implementation lessons from the programs will provide useful insights for health systems planning.
The Genome-wide Sequencing Ontario51 pilot project, launched in 2021, aims to assess the clinical performance, cost-effectiveness, and implementation of providing whole genome or exome sequencing for adults and children with suspected rare genetic disorders.52 Findings from the pilot project will help inform infrastructure development for the technology and its role as standard of care, as well as provide insight into the policy implications for its coverage.
Pharmacogenomics Tests for Mental Health Conditions
Pharmacogenomics testing characterizes genes that may influence how drug therapies are processed by an individual. This type of testing can provide information about suitable drug targets, interactions between different drugs, and who may be at an increased risk for side effects.53 Unlike some clinical areas, such as oncology, there have been relatively fewer developments of precision medicine technologies in the field of mental health care.53 However, with more developments in pharmacogenomics, there is a possibility that precision medicine technologies could lead to significant change over the next 5 years in terms of how drug-based therapies are prescribed for mental health conditions.54
Estimates indicate that 1 in 5 people55 in Canada may have a mental health condition and mental health care is a priority across jurisdictions. Although drug therapies are only a part of the treatment approach, in routine care, drug selection is largely based on clinical input and a sequential “trial and error”–based approach that leads to varied effectiveness.56,57 People who are prescribed drug therapies often have to wait to see if a treatment shows benefit and can experience side effects while doing so. This process of experientially finding a medication that is both effective at addressing symptoms and does not have side effects that impact quality of life can be drawn out, leading to prolonged symptoms and negatively affecting patients’ experiences.58 As such, there is growing interest in identifying biomarkers that predict treatment response and help guide a more tailored approach for selecting treatments.
Pharmacogenomics testing technologies are available as home sampling kits or laboratory-based genetic testing services and assess multiple genes that may be involved with drug metabolism.59 Commercially available tests often link to clinical decision support tools that use proprietary algorithms to provide assessments of the suitability of different medications for depression, psychosis, and attention-deficit/hyperactivity disorder (ADHD), among other conditions.60 As of yet, clinical adoption of the technology in Canada and across the globe remains limited due to uncertainties about clinical utility and relevance of test findings to individuals, among other barriers.58,60,61 Research examining the effectiveness of pharmacogenomics testing, in particular for guiding the treatment of depression, is emerging; however, to date, the findings remain mixed, with studies indicating both significant improvements and no difference in clinical outcomes compared with usual care.57,58,62 A qualitative study reported that people in Canada with lived experiences and health care professionals are hopeful about the technology but are also awaiting more robust clinical and economic evidence.63 Nevertheless, pharmacogenomics testing is being offered at some Canadian health care centres through research programs, and, increasingly, commercial tests are being offered to individuals to purchase out of pocket or with private insurance.59,60
Should emerging research show strong clinical benefit and testing becomes more widely available over the next 5 years, health care decision-makers will need to develop equitable approaches for providing access to tests — especially for people who may already face barriers in accessing mental health services. Given the high prevalence of mental health conditions, the potential volume of testing could impact laboratory infrastructure capacity and there may need to be additional training requirements for health care professionals on how to use results to inform care decisions.
Examples of emerging technologies include:
Inagene (Inagene Diagnostics, Canada), GeneSight (Myriad Genetics, US), and IDgenetix (Castle Biosciences, US) are targeted test services that analyze a swab sample provided by an individual and send a customized report about the genetic variants they may have. These variants may affect drug metabolism, interactions, and the efficacy of drugs used for treating a range of mental health conditions. The customized reports provide information that could be used by health care professionals to guide decisions about treatment plans.
myDNA (myDNA Life, Australia) and MatchMyMeds (DNALabs, Canada) are broader pharmacogenomics tests that are not specific to mental health conditions, but rather cover a range of drugs used for many different health conditions, including mental health. These tests provide drug compatibility reports that may provide recommendations for health care professionals about drug selection for mental health conditions.
Population-Based Genetic Screening Interventions
Screening aims to detect the early signs of specific health condition or assess the likelihood of having specific health conditions among people who are otherwise asymptomatic.64 Many screening interventions, including those that use genetic testing, are often limited to specific subpopulations with certain risk factors based on age, family history, or other health behavioural or demographic factors. For example, publicly funded genetic testing for BRCA genes is limited in Canada, with jurisdictions having varying eligibility criteria that often include having a known family history of mutations associated with an increased risk of cancer.65 However, using stringent criteria for genetic screening can miss a substantial proportion of people who may have high-risk mutations.66 It is possible that this existing targeted approach to screening may shift to become more population based. Driven by the decreasing costs of sequencing, increased availability of testing, and ongoing development of better analysis algorithmics, screening using genetic testing could see further expansion over the next 5 years and become more widely available at a population level for people of all ages, reducing the need for stringent eligibility criteria.66
Different pilot programs around the world67 are under way to assess the value, feasibility, and implementation considerations of expanding screening programs by offering genetic testing.68 If certain tests are made more widely available, they could facilitate the earlier diagnosis of health conditions, prevent some health-related complications, reduce delays in getting treatments, and help improve outcomes.69 Expanding genetic testing to a greater number of people may also support efforts to improve the size and diversity of genomic databases, which would help improve analyses and better inform care.70,71
Health systems also face several uncertainties related to expanding genetic screening systems, such as how test results could be used to inform clinical decision-making when there may be limited evidence about effective treatments, the impact of complex findings (e.g., identifying hereditary mutations associated with health conditions) for patients and families, the cost implications of follow-up testing, and determining optimal testing strategies (e.g., which genes to include and who to test).68,72 The infrastructure capacity required to process the volume of testing and the number and expertise of health care professionals needed to interpret test results will be key considerations for health systems planning. Assessing the acceptability of testing for diverse populations and equity considerations related to screening uptake will be needed to inform future policies.66 Screening is a complex health intervention with potential harms and benefits, and therefore, monitoring findings and lessons learned from ongoing pilot studies can help inform decisions about how best to expand genetic screening in Canada.
Some emerging pilot studies and initiatives to watch include:
The Screen Project73 by Women’s College Hospital (Toronto, Canada) is national program that launched in 2017 to offer genetic testing for BRCA1 and BRCA2 genes to any person older than 18 years. Testing costs $250 and results are delivered via a digital portal or by a genetic counsellor. The study aims to assess the feasibility of testing, examine people’s experiences, and estimate the clinical benefits of possible cancer prevention.74
The Newborn Genomes Programme75 by Genomics England aims to sequence 100,000 genomes of babies born in England starting in 2023. The pilot will screen for 200 actionable health conditions; that is, conditions for which there are existing treatments or interventions (an increase from the existing screening approach that identifies 9 conditions in the country) to inform the direct care of children.76 Although all jurisdictions in Canada have newborn screening programs, this specific program uses enhanced genetic testing and could inform the future of screening with emerging technologies.77 The program also aims to use deidentified data to further develop research about diagnostics and treatments.76 The pilot has a broader aim to assess the range of benefits, harms, and implications of genetic screening as participants may choose to be followed over their lifetime.76
In Our DNA SC78 by the Medical University of South Carolina (Charleston, US) and Helix79 (US) is a community health project that aims to enrol 100,000 participants over 4 years to provide free genetic testing for Tier 1 health conditions (i.e., conditions defined by the Centers for Disease Control and Prevention as having significant potential for genetic testing).80 The state-wide study is focused on examining implementation outcomes related to the participation rates, facilitators, barriers, and sustainability of offering population-wide genetic testing.80 Another goal of the study is to engage with diverse population groups.
Top Issues About Precision Medicine Technologies to Watch in 2023
Disparities in the Implementation of Precision Medicine Technologies
In many health systems, there are disparities in who can access and use existing precision medicine technologies.13,81 These disparities can result from a reduced ability to access health care because of costs, availability, geography, and awareness.13,81 They are also exacerbated by systemic racism in health care, which excludes and makes health care inaccessible to racialized people and Indigenous Peoples. Moreover, some populations have historically not been included in health research or the databases used to develop precision medicine technologies.13 As a result, the precision medicine tests developed using these databases are not necessarily applicable to or interpretable for those who are racialized or otherwise underrepresented in health research.13
Existing health disparities may become amplified with the further spread and implementation of precision medicine technologies. For example, inequalities based on geography may occur if emerging tests are reimbursed primarily through specialized research centres with higher levels of research funding, limiting access for people who do not receive care at those centres. Given the specialized resources required and the challenges faced when implementing precision medicine in higher-resource settings, there are unique challenges faced by lower-resource settings, including those with geographically dispersed populations.82
Health care decision-makers will need to ensure equitable development of and access to precision medicine technologies and mitigate the potential for precision medicine technologies to replicate or exacerbate existing health inequalities. Different health care systems are developing targeted approaches that could be important to watch, such as:
Investing in population-representative databases: To address bias and inequities within precision medicine technologies, health systems are investing in the collection and building of representative genomic databases. For example, within Canada, Genome British Columbia is funding the Silent Genomes Project, which aims to address the lack of First Nations, Inuit, and Métis genomic representation in cancer genetics databases for children.83 In the US, All of Us, an initiative funded by the National Institutes of Health Research, is attempting to achieve similar goals by building a database of social, environmental, and genomic information that is diverse and inclusive of all persons living in the US.84 Both initiatives involve active community engagement, participation, and oversight over the projects by the communities they are aiming to serve.
Reducing barriers to access: Pan-Canadian collaboratives such as PRecision Oncology For Young PeopLE (PROFYLE) by the Terry Fox Research Institute aim to make the technology that provides molecular profiling of tumours available to children and youth regardless of where they live. Additional initiatives being explored by health systems include reducing barriers to access due to geography and cost by developing travel support programs that enable cross-jurisdictional travel, ensuring that there is public reimbursement for precision medicine technologies within and between provincial and territorial health systems, and developing models of care that harness technologies such as at-home testing and virtual care to facilitate the remote delivery of care.85,86
Enhancing diversity, inclusivity, and engagement: Increasing diversity in health professionals through workforce planning and education may help mitigate health concerns and create more inclusive care.87 Other examples include the codesign (i.e., active partnering and engagement) of precision medicine health services with equity-deserving and underrepresented groups, and ongoing data collection related to disparities in access to and outcomes of precision medicine13,88,89
Increased Demands on Health System Capacity and Infrastructure From the Use of Precision Medicine
The emergence of precision medicine will put increased demands on the health care system, including physical resources, staff, and infrastructure. Physical resources include the freezers, hospital space, and equipment needed to store and process genetic samples.20 Treatments for certain cancers and other conditions may require ongoing testing and/or multiple tests throughout a person’s care, making the need for infrastructure to adequately store samples increasingly important. Infrastructure changes may include the health system’s capacity and workflow to conduct and process tests, maintain information databases, and provide strong information technology support.90 Having the necessary number and expertise of professionals to perform and analyze results from precision medicine technologies, such as sequencing tests that may have stringent quality control measures, will be necessary to adopt certain technologies. These increased demands may affect how precision medicine is adopted and implemented across Canada and its long-term sustainability.20 It is possible that with appropriate use, precision medicine technologies may reduce unnecessary care and improve patient outcomes, and over time result in more efficient and effective health care.4
In addition to meeting increased demands by adding new resources and infrastructure, there are opportunities to improve capacity for precision medicine through efficiencies. Developing health system capacity may include increasing the number of trained professionals, but also having streamlined workflows, and investing in technology-enabled clinical care and new models of care that deliver genetic-informed patient care.20,91
Some examples that aim to address health systems’ capacity include:
Scaling-up and spreading access to genetic testing services: For example, GenCOUNSEL92 is a collaborative, multidisciplinary project by the BC Children’s Hospital Research Institute that emphasizes patient and family engagement and has a number of initiatives that aim to increase the capacity to provide genetic counselling and services. It includes developing and implementing decision support tools to assist patients and providers when they order and interpret genetic test results, and identifying the current and future needs of genetic counselling services and providers.
Alternative models of care: In England, the NHS has developed the NHS Genomic Medicine Service93 and is investing in genomic medicine by mainstreaming workforce training, centralizing laboratories, and codesigning health services with patients and the public. Models of care that enable a centralized testing infrastructure and establish workflows20 and technology-enabled improved communication and coordination between health care professionals who order and interpret test results across care locations, such as e-consult,20,94,95 can help build a cohesive and streamlined system.20,94 This will also require technology to support and encourage clinicians to adopt and use patient portals and other digital platforms to enable information-sharing between clinics, health professionals, and patients.96
Evolving Roles and Scopes of Practice for Health Care Professionals
The mainstreaming of precision medicine and genetic testing across health conditions and health systems may lead to evolving roles and scopes of practice for many health care professionals. For example, nurses, pharmacists, public health professionals, primary care physicians, and specialists are increasingly using and tailoring patient care with precision medicine technologies.97-99 Many health care professionals who have not traditionally had direct contact with precision medicine are finding themselves adapting to ordering, interpreting, and/or consulting with patients about genetic tests. With the rapid pace of innovation within precision medicine technologies, limited formal training in precision medicine, and lack of clarity in roles, health care professionals report experiencing challenges in determining who orders tests (and at which point), who interprets the results, and who helps the patient navigate their treatment journey.100,101
There is an opportunity to clarify the roles and responsibilities of various health care professionals in delivering precision medicine to support the efficient and seamless delivery of services across the continuum of care.102,103 Supporting health care professionals to use their training and expertise to their optimal potential may lead to establishing new responsibilities (e.g., test ordering), shifting tasks (e.g., administrative duties), and facilitating the composition of care teams with the appropriate complement of professionals.96 Health systems may also need to consider alternative remuneration models for different health care professionals who may be involved at various stages of ordering or assessing test results and supporting patients and families through their care pathways.90,103
Examples of opportunities that may address issues relating to roles and scope of practice include:
Reviewing workflow and roles and responsibilities in genetic testing: There are opportunities to revise the roles and scope of practice and to formalize workflows to ensure efficiency in care delivery. For example, Cancer Care Ontario and Ontario Health’s Enhancing Clinical Cancer Genetic Service Delivery reviewed the provision of genetic services in Ontario and identified opportunities to shift tasks and responsibilities to ensure health professionals were able to optimize their expertise and training.96 While the report focuses on cancer care, it offers potential insight to other health areas and the types of opportunities that may be beneficial when implementing precision medicine more broadly.
Support and training for health care professionals in precision medicine: Many initiatives are under way to support health care professionals by offering training in genomic medicine. These types of activities enable professionals to work at their full scope of practice and integrate precision medicine into care. For example, the Canadian Nursing & Genomics initiative is supporting the integration of genomic medicine into nursing practice by considering the role of nurses and engaging nurses in developing a training strategy.104 Training initiatives in genomics education for health professionals benefits from systems-level support; for example, Genome British Columbia’s Genomics Education for Health Professionals Implementation Framework describes promising options that include developing centralized clinical guidelines, point-of-care supporting documents, and professional education that is linked to clinical practice.105
Complexity in Interpreting Test Results
The increase in availability of precision medicine tests across the pathway of care (e.g., from screening, to diagnosis, to treatment, to prognosis) raises challenges for health care professionals when ordering tests and interpreting their results. The clinical significance of precision medicine test results is not always clear and results are not always actionable (i.e., directly provide guidance about a certain therapy).106 Many health care professionals (e.g., primary care practitioners, pharmacists, oncologists) report having limited awareness and knowledge of how to order and interpret precision medicine test results.107,108 They also describe struggling to contextualize the results along with other phenotypic information, including the social and environmental determinants of health, in ways that make them clinically actionable.81,106,109 The difficulties around genetic test interpretation can be exacerbated when providing care to people who are underrepresented in health care.87 The potential for incidental and secondary findings (that is, additional information that may not be directly related to the reasons testing was undertaking), and their implications for patients and family members, adds to the complexity of testing.
The challenges of interpreting complex information can affect the extent to which health care professionals adopt precision medicine technologies and how care is organized to meet the needs of health care professionals and those accessing health care. Test ordering can be complicated in health systems where precision medicine is not embedded into the clinical workflow. For instance, ordering processes can vary across institutions and jurisdictions, depending on whether the test is publicly funded or reimbursed.82 Moreover, depending on the pathway of care, the provider ordering the test may not be the same person who interprets the test results and who communicates the results and follow-up information to the person undergoing testing, which adds complexity to the process of interpreting and delivering results.107
Furthermore, it is a challenge to develop evidence-based clinical guidelines in an area with increasingly complex tests. Decision support tools and electronic aids may offer valuable support to health care providers and, when embedded in electronic health records, these tools can help streamline decision-making and processes. However, it can be difficult to keep up with the rapid changes in the availability of testing as well as the tests themselves.90,110 Moreover, it is unclear to what extent decision support tools improve clinical decision-making and patient outcomes.110 Models of shared decision-making can help take patients’ perspectives and values into account when deciding to order tests and return results, as genetic information can involve incidental findings that may also affect family members.107
In addition to improving the capacity of health care professionals through broad training initiatives, further opportunities exist to address complexity in interpreting precision medicine test results. For instance:
Building patient-centred models of care: Patient-centred care can address test result complexity by ensuring that patients’ experiences guide the design of care pathways. This includes elements such as establishing which care professionals help patients navigate their testing journey, as well as enabling health care consultations to allow for the additional time needed to discuss ordering tests and returning results.107 Other opportunities include developing models of shared decision-making and tools to help order and interpret tests that keep pace with technological developments and clinical practice, and evaluating their impact on patients’ decision-making and outcomes.107,110
Developing processes related to incidental and secondary result: Ensuring patients are able to provide their informed consent to receive (or not) incidental or secondary results and guidance and recommendations to support their return if, for instance, they are medically actionable.106 This can include identifying processes to triage referrals and access specialized support through means such as e-consult and telehealth, when appropriate.
Equity-enhancing training and workforce initiatives: These can include, for instance, increasing health professional training and education that is dedicated to precision medicine testing and interpretation and addresses the needs of underserved or racialized populations,87 as well as developing supports that enable consistent interpretation to reduce potential inequalities in care.111
Challenges in Regulating and Evaluating Precision Medicine Technologies
Precision medicine technologies will likely create challenges for existing regulatory pathways and health technology assessment (HTA) processes and methods.112 Precision medicine tests, such as whole genome or multigene testing, often fit in a complicated health care pathway for very specific populations, and tend to be based on trials with small sample sizes and limited comparative data. This, along with the increasing volume of tests available and the rapid pace of innovation, makes it difficult to assess their value using traditional HTA methods and processes.112,113 Without new and consistent approaches for evaluating new precision medicine technologies, there may be inequalities in who has access to these technologies.82
Companion diagnostics and therapeutics often go through separate regulatory and reimbursement pathways.82 Some regulatory processes are federal and others, such as lab-developed diagnostic tests and diagnostic services, have provincial oversight.82 Similarly, reimbursement is sometimes at the provincial level, but can also be held by the regional health authority or hospital, depending on the technology and the jurisdiction. These system fragmentations lead to challenges in the funding and reimbursement of precision medicine technologies, and new evaluative approaches will be needed to help balance the need for evaluation and timely access for patients.20
Regulatory and HTA processes and methods are adapting by finding new ways of addressing uncertainty and heterogeneity and by harnessing the value of real-world evidence to help inform decision-making.112,114,115 Some examples of work in this area to watch include:
Generating and using real-world evidence: This can include generating and using real-world evidence through new models of care and health care design. For example, the Canadian Network for Learning Healthcare Systems and Cost-Effective 'Omics Innovation (CLEO) is focused on supporting developing frameworks for learning health systems that enable ongoing evaluations and HTA of precision medicine technologies.112
Developing new methods for HTA: For example, those who look at the life cycle of a technology, from research to obsolescence, could allow technologies to be reevaluated and foster ongoing data collection and frameworks for assessing the evidence.112 New approaches to economic evaluation are being developed, including instruments and approaches that account for value of precision medicine technologies, and the direct and in-direct costs and benefits of precision medicine technologies that are distributed across the health care system.20,113
Exploring new reimbursement and payment models arrangements: Alternative models of payment, such as outcome-based agreements and related approaches, could allow risks to be shared and provide opportunities to change purchasing and funding arrangements based on real-world evidence and ongoing evaluations.20,112,116
References
1.Mason J, Brundisini F, Hill S, Kumar D, Rader T. 2022 health technology trends to watch: Top 10 list. Can J Health Technol. 2022;2(3). https://canjhealthtechnol.ca/index.php/cjht/article/view/er0012. Accessed 2023 Jan 11.
2.National Library of Medicine. What is precision medicine? 2022. https://medlineplus.gov/genetics/understanding/precisionmedicine/definition/. Accessed 2023 Feb 03.
3.Phillips CJ. Precision medicine and its imprecise history. Harv Data Sci Rev. 2020. 10.1162/99608f92.3e85b56a. Accessed 2023 Jan 11.
4.Denny JC, Collins FS. Precision medicine in 2030 - Seven ways to transform healthcare. Cell. 2021;184(6):1415-1419. PubMed
5.Might M, Crouse AB. Why rare disease needs precision medicine-and precision medicine needs rare disease. Cell Rep Med. 2022;3(2):100530. PubMed
6.Behl T, Kaur I, Sehgal A, et al. The road to precision medicine: Eliminating the “One Size Fits All” approach in Alzheimer's disease. Biomed Pharmacother. 2022;153:113337. PubMed
7.Government of Canada. Consultation paper: Developing a pan-Canadian genomics strategy. 2022; https://ised-isde.canada.ca/site/genomics/en/consultation-paper-developing-pan-canadian-genomics-strategy. Accessed 2023 Jan 11.
8.Doxzen KW, Signé L, Bowman DM. Advancing precision medicine through agile governance: Bridging innovation and regulation for the greater good. 2022; https://www.brookings.edu/research/advancing-precision-medicine-through-agile-governance/. Accessed 2023 Jan 6.
9.Ahmed Z, Mohamed K, Zeeshan S, Dong X. Artificial intelligence with multi-functional machine learning platform development for better healthcare and precision medicine. Database (Oxford). 2020;2020:baaa010. PubMed
10.Rehm HL, Page AJH, Smith L, et al. GA4GH: International policies and standards for data sharing across genomic research and healthcare. Cell Genom. 2021;1(2):100029. PubMed
11.Liu G, Cheung WY, Feilotter H, et al. Precision oncology in Canada: Converting vision to reality with lessons from international programs. Curr Oncol. 2022;29(10):7257-7271. PubMed
12.Cuppen E, Elemento O, Rosenquist R, et al. Implementation of whole-genome and transcriptome sequencing into clinical cancer care. JCO Precis Oncol. 2022;6:e2200245. PubMed
13.Khoury MJ, Bowen S, Dotson WD, et al. Health equity in the implementation of genomics and precision medicine: A public health imperative. Genet Med. 2022;24(8):1630-1639. PubMed
14.Canada Health Infoway. Canadian digital health survey: What Canadians think. 2020; https://www.infoway-inforoute.ca/en/component/edocman/3856-canadian-digital-health-survey-what-canadians-think/view-document. Accessed 2023 Jan 13.
15.Bombard Y, Hayeems RZ. How digital tools can advance quality and equity in genomic medicine. Nat Rev Genet. 2020;21(9):505-506. PubMed
16.Ontario Health. Patient portals standards. 2022; https://www.ontariohealth.ca/system-planning/digital-standards/patient-portals-standards. Accessed 2023 Jan 13.
17.Shickh S, Rafferty SA, Clausen M, et al. The role of digital tools in the delivery of genomic medicine: Enhancing patient-centered care. Genet Med. 2021;23(6):1086-1094. PubMed
18.Lee W, Shickh S, Assamad D, et al. Patient-facing digital tools for delivering genetic services: A systematic review. J Med Genet. 2023;60(1):1-10. PubMed
19.Government of Canada. Digital health services survey: What we heard from Canadians. 2021; https://ised-isde.canada.ca/site/competition-bureau-canada/en/how-we-foster-competition/promotion-and-advocacy/advocacy-promoting-benefits-competition/digital-health-services-survey-what-we-heard-canadians#sec01. Accessed 2023 Jan 13.
20.Husereau D, Steuten L, Muthu V, et al. Effective and efficient delivery of genome-based testing - What conditions are necessary for health system readiness? Healthcare (Basel). 2022;10(10):2086. PubMed
21.Genetics Adviser. Genetics Adviser: Patients' digital guide to their genetic testing journey. 2021; https://www.geneticsadviser.com/. Accessed 2023 Feb 2.
22.Shickh S, Hirjikaka D, Clausen M, et al. Genetics Adviser: A protocol for a mixed-methods randomised controlled trial evaluating a digital platform for genetics service delivery. BMJ Open. 2022;12(4):e060899. PubMed
23.University of Washington. The ShareDNA App. http://sharedna.org/. Accessed 2023 Feb 2.
24.Jujjavarapu C, Anandasakaran J, Amendola LM, et al. ShareDNA: A smartphone app to facilitate family communication of genetic results. BMC Med Genomics. 2021;14(1):10. PubMed
25.Cleveland Clinic. Liquid biopsy. 2022; https://my.clevelandclinic.org/health/diagnostics/23992-liquid-biopsy. Accessed 2022 Dec 20.
26.Lone SN, Nisar S, Masoodi T, et al. Liquid biopsy: A step closer to transform diagnosis, prognosis and future of cancer treatments. Mol Cancer. 2022;21(1):79. PubMed
27.Jorgensen JT. The current landscape of the FDA approved companion diagnostics. Transl Oncol. 2021;14(6):101063. PubMed
28.Garcia-Pardo M, Makarem M, Li JJN, Kelly D, Leighl NB. Integrating circulating-free DNA (cfDNA) analysis into clinical practice: opportunities and challenges. Br J Cancer. 2022;127(4):592-602. PubMed
29.Ignatiadis M, Sledge GW, Jeffrey SS. Liquid biopsy enters the clinic - Implementation issues and future challenges. Nat Rev Clin Oncol. 2021;18(5):297-312. PubMed
30.Mauri G, Vitiello PP, Sogari A, et al. Liquid biopsies to monitor and direct cancer treatment in colorectal cancer. Br J Cancer. 2022;127(3):394-407. PubMed
31.IJzerman M, de Boer J, Azad A, et al. Towards routine implementation of liquid biopsies in cancer management: It is always too early, until suddenly it is too late. Diagnostics (Basel). 2021;11(1):103. PubMed
32.Shickh S, Oldfield LE, Clausen M, et al. “Game Changer”: Health professionals' views on the clinical utility of circulating tumor DNA testing in hereditary cancer syndrome management. Oncologist. 2022;27(5):e393-e401. PubMed
33.Health Quality Ontario. Cell-free circulating tumour DNA blood testing to detect EGFR T790M mutation in people with advanced non–small cell lung cancer: A health technology assessment. 2020; https://www.hqontario.ca/Evidence-to-Improve-Care/Health-Technology-Assessment/Reviews-And-Recommendations/Cell-Free-Circulating-Tumour-DNA-Blood-Testing-to-Detect-EGFR-T790M-Mutation-in-People-With-Advanced-NonSmall-Cell-Lung-Cancer. Accessed 2022 Dec 21.
34.Canadian Partnership Against Cancer (CPAC). PRecision Oncology Evidence Development in Cancer Treatment - Liquid (PREDiCTl). 2022; https://www.canadiancancertrials.ca/Trial/Default.aspx?TrialId=NCT05057234. Accessed 2022 Dec 20.
35.Hasin Y, Seldin M, Lusis A. Multi-omics approaches to disease. Genome Biol. 2017;18(1):83. PubMed
36.Lalonde E, Rentas S, Lin F, Dulik MC, Skraban CM, Spinner NB. Genomic diagnosis for pediatric disorders: Revolution and evolution. Front Pediatr. 2020;8:373. PubMed
37.Costain G, Walker S, Marano M, et al. Genome sequencing as a diagnostic test in children with unexplained medical complexity. JAMA Netw Open. 2020;3(9):e2018109. PubMed
38.Smedley D, Smith KR, Martin A, et al. 100,000 Genomes pilot on rare-disease diagnosis in health care - Preliminary report. N Engl J Med. 2021;385(20):1868-1880. PubMed
39.Shickh S, Mighton C, Uleryk E, Pechlivanoglou P, Bombard Y. The clinical utility of exome and genome sequencing across clinical indications: A systematic review. Hum Genet. 2021;140(10):1403-1416. PubMed
40.NICUSeq Study Group, Krantz ID, Medne L, et al. Effect of whole-genome sequencing on the clinical management of acutely ill infants with suspected genetic disease: A randomized clinical trial. JAMA Pediatr. 2021;175(12):1218-1226. PubMed
41.Horak P, Heining C, Kreutzfeldt S, et al. Comprehensive genomic and transcriptomic analysis for guiding therapeutic decisions in patients with rare cancers. Cancer Discov. 2021;11(11):2780-2795. PubMed
42.Cobain EF, Wu YM, Vats P, et al. Assessment of clinical benefit of integrative genomic profiling in advanced solid tumors. JAMA Oncol. 2021;7(4):525-533. PubMed
43.Malone ER, Oliva M, Sabatini PJB, Stockley TL, Siu LL. Molecular profiling for precision cancer therapies. Genome Med. 2020;12(1):8. PubMed
44.van de Ven M, Simons M, Koffijberg H, et al. Whole genome sequencing in oncology: using scenario drafting to explore future developments. BMC Cancer. 2021;21(1):488. PubMed
45.Duncavage EJ, Schroeder MC, O'Laughlin M, et al. Genome sequencing as an alternative to cytogenetic analysis in myeloid cancers. N Engl J Med. 2021;384(10):924-935. PubMed
46.Weymann D, Laskin J, Jones SJM, et al. Early-stage economic analysis of research-based comprehensive genomic sequencing for advanced cancer care. J Community Genet. 2022;13(5):523-538. PubMed
47.Costain G, Cohn RD, Scherer SW, Marshall CR. Genome sequencing as a diagnostic test. CMAJ. 2021;193(42):E1626-E1629. PubMed
48.Wickland DP, Sherman ME, Radisky DC, Mansfield AS, Asmann YW. Lower exome sequencing coverage of ancestrally African patients in The Cancer Genome Atlas. J Natl Cancer Inst. 2022;114(8):1192-1199. PubMed
49.Gorzynski JE, Goenka SD, Shafin K, et al. Ultrarapid nanopore genome sequencing in a critical care setting. N Engl J Med. 2022;386(7):700-702. PubMed
50.Marathon of Hope Cancer Centres Network. Creating the foundation for the creation of a pan-Canadian cancer centre network. 2021; https://www.marathonofhopecancercentres.ca/our-research/network-pilot-projects. Accessed 2022 Dec 20.
51.SickKids. Genome-wide sequencing pilot project helps Ontario families get answers. 2022; https://www.sickkids.ca/en/news/archive/2022/genome-wide-sequencing-pilot-project-helps-ontario-families-get-answers/. Accessed 2022 Dec 20.
52.Hayeems RZ, Marshall CR, Gillespie MK, et al. Comparing genome sequencing technologies to improve rare disease diagnostics: A protocol for the evaluation of a pilot project, Genome-wide Sequencing Ontario. CMAJ Open. 2022;10(2):E460-E465. PubMed
53.Centers for Disease Control and Prevention. Pharmacogenomics: What does it mean for your health? 2020; https://www.cdc.gov/genomics/disease/pharma.htm. Accessed 2023 Jan 13.
54.Manchia M, Pisanu C, Squassina A, Carpiniello B. Challenges and future prospects of precision medicine in psychiatry. Pharmgenomics Pers Med. 2020;13:127-140. PubMed
55.CAMH. Mental illness and addiction: Facts and statistics. https://www.camh.ca/en/driving-change/the-crisis-is-real/mental-health-statistics. Accessed 2023 Jan 20.
56.van Westrhenen R, Aitchison KJ, Ingelman-Sundberg M, Jukic MM. Pharmacogenomics of antidepressant and antipsychotic treatment: How far have we got and where are we going? Front Psychiatry. 2020;11:94. PubMed
57.Iosifescu DV. Pharmacogenomic testing for next-step antidepressant selection: Still a work in progress. JAMA. 2022;328(2):146-148. PubMed
58.Virelli CR, Mohiuddin AG, Kennedy JL. Barriers to clinical adoption of pharmacogenomic testing in psychiatry: A critical analysis. Transl Psychiatry. 2021;11(1):509. PubMed
59.Health Quality Ontario. Multi-gene pharmacogenomic testing that includes decision-support tools to guide medication selection for major depression: A health technology assessment. 2021; https://www.hqontario.ca/evidence-to-improve-care/health-technology-assessment/reviews-and-recommendations/multi-gene-pharmacogenomic-testing-that-includes-decision-support-tools-to-guide-medication-selection-for-major-depression. Accessed 2022 Dec 22.
60.Maruf AA, Fan M, Arnold PD, Muller DJ, Aitchison KJ, Bousman CA. Pharmacogenetic testing options relevant to psychiatry in Canada: Options de tests pharmacogenetiques pertinents en psychiatrie au Canada. Can J Psychiatry. 2020;65(8):521-530. PubMed
61.Maruf AA, Bousman CA. Approaches and hurdles of implementing pharmacogenetic testing in the psychiatric clinic. PCN Rep. 2022;1(2). https://onlinelibrary.wiley.com/doi/abs/10.1002/pcn5.26. Accessed 2022 Dec 22.
62.Darvesh N, Horton J, Lê M-L. Pharmacogenomic testing in depression: A 2021 update. Can J Health Technol. 2022;2(1). https://canjhealthtechnol.ca/index.php/cjht/article/view/rc1403. Accessed 2022 Dec 21.
63.Slomp C, Morris E, Edwards L, et al. Pharmacogenomic testing for major depression: A qualitative study of the perceptions of people with lived experience and professional stakeholders. Can J Psychiatry. 2022:7067437221140383. PubMed
64.Givler D, Givler A. Health screening. Treasure Island (FL): StatPearls Publishing; 2022: https://www.ncbi.nlm.nih.gov/books/NBK436014/. Accessed 2023 Jan 6.
65.Genetics Education Canada - Knowledge Organization. Hereditary breast and ovarian cancer (BRCA1/BRCA2). 2020; https://geneticseducation.ca/educational-resources/gec-ko-on-the-run/hereditary-breast-and-ovarian-cancer-brca1brca2/. Accessed 2023 Jan 6.
66.Mighton C, Shickh S, Aguda V, Krishnapillai S, Adi-Wauran E, Bombard Y. From the patient to the population: Use of genomics for population screening. Front Genet. 2022;13:893832. PubMed
67.Kaiser J. Sequencing projects will screen 200,000 newborns for disease. Science. 2022;378(6625):1159. PubMed
68.Foss KS, O'Daniel JM, Berg JS, et al. The rise of population genomic screening: Characteristics of current programs and the need for evidence regarding optimal implementation. J Pers Med. 2022;12(5):692. PubMed
69.Milko LV, Khoury MJ. Editorial: DNA-based population screening for precision public health. Front Genet. 2022;13:1061329. PubMed
70.Carress H, Lawson DJ, Elhaik E. Population genetic considerations for using biobanks as international resources in the pandemic era and beyond. BMC Genomics. 2021;22(1):351. PubMed
71.Lekstutiene J, Holm S, Gefenas E. Biobanks and individual health related findings: From an obstacle to an incentive. Sci Eng Ethics. 2021;27(4):55. PubMed
72.Shen EC, Srinivasan S, Passero LE, et al. Barriers and facilitators for population genetic screening in healthy populations: A systematic review. Front Genet. 2022;13:865384. PubMed
73.Women's College Hospital. The Screen Project. 2023; https://www.womensresearch.ca/active-studies/the-screen-project-study/. Accessed 2023 Jan 6.
74.Narod SA, Gojska N, Sun P, et al. The Screen Project: Guided direct-to-consumer genetic testing for breast cancer susceptibility in Canada. Cancers (Basel). 2021;13(8):1894. PubMed
75.Genomics England. Newborn Genomes Programme. 2023; https://www.genomicsengland.co.uk/initiatives/newborns. Accessed 2023 Jan 6.
76.Pichini A, Ahmed A, Patch C, et al. Developing a national newborn genomes program: An approach driven by ethics, engagement and co-design. Front Genet. 2022;13:866168. PubMed
77.Moore AM, Richer J. Genetic testing and screening in children. Paediatr Child Health. 2022;27(4):243-253. PubMed
78.Medical University of South Carolina. In Our DNA SC. https://web.musc.edu/inourdnasc/. Accessed 2023 Jan 9.
79.Helix OpCo LLC. Broadening the impact of genomics. https://www.helix.com/pages/about. Accessed Feb 03, 2023.
80.Allen CG, Judge DP, Levin E, et al. A pragmatic implementation research study for In Our DNA SC: A protocol to identify multi-level factors that support the implementation of a population-wide genomic screening initiative in diverse populations. Implement Sci Commun. 2022;3(1):48. PubMed
81.Erdmann A, Rehmann-Sutter C, Bozzaro C. Patients' and professionals' views related to ethical issues in precision medicine: A mixed research synthesis. BMC Med Ethics. 2021;22(1):116. PubMed
82.Cancer Care Survivor Network. A review of precision medicine companion diagnostics in Canada: Are we there yet? 2020; https://survivornet.ca/wp-content/uploads/2021/01/A-Review-of-Precision-Medicine-Companion-Diagnostics-in-Canada.pdf. Accessed 2023 Jan 16.
83.Caron NR, Chongo M, Hudson M, et al. Indigenous genomic databases: Pragmatic considerations and cultural contexts. Front Public Health. 2020;8:111. PubMed
84.Mapes BM, Foster CS, Kusnoor SV, et al. Diversity and inclusion for the All of Us research program: A scoping review. PLoS One. 2020;15(7):e0234962. PubMed
85.Radich JP, Briercheck E, Chiu DT, et al. Precision medicine in low- and middle-income countries. Annu Rev Pathol. 2022;17(1):387-402. PubMed
86.Shemie G, Nguyen MT, Wallenburg J, Ratjen F, Knoppers BM. The equitable implementation of cystic fibrosis personalized medicines in Canada. J Pers Med. 2021;11(5):382. PubMed
87.Sharma Y, Cox L, Kruger L, Channamsetty V, Haga SB. Evaluating primary care providers' readiness for delivering genetic and genomic services to underserved populations. Public Health Genomics. 2021:1-10. PubMed
88.Lemke AA, Esplin ED, Goldenberg AJ, et al. Addressing underrepresentation in genomics research through community engagement. Am J Hum Genet. 2022;109(9):1563-1571. PubMed
89.Bombard Y, Baker GR, Orlando E, et al. Engaging patients to improve quality of care: A systematic review. Implement Sci. 2018;13(1):98. PubMed
90.Hooker GW. Building an infrastructure to enable delivery of genomic medicine. Am J Med Genet C Semin Med Genet. 2021;187(1):95-99. PubMed
91.Costa T, Gillies B, Oh T, Scott J. The Canadian genetic counseling workforce: Perspectives from employers and recent graduates. J Genet Couns. 2021;30(2):406-417. PubMed
92.BC Children's Hospital Research Institute. GenCOUNSEL Project. 2023; https://bcchr.ca/GenCOUNSEL. Accessed 2023 Jan 23.
93.NHS. Accelerating genomic medicine in the NHS. 2022; https://www.england.nhs.uk/long-read/accelerating-genomic-medicine-in-the-nhs/. Accessed 2023 Feb 3.
94.Horgan D, Curigliano G, Rieß O, et al. Identifying the steps required to effectively implement next-generation sequencing in oncology at a national level in Europe. J Pers Med. 2022;12(1):72. PubMed
95.Carroll JC, Liddy C, Afkham A, et al. Use of eConsult to enhance genetics service delivery in primary care: A multimethod study. Genet Med. 2022;24(10):2034-2041. PubMed
96.Cancer Care Ontario. Enhancing clinical cancer genetic service delivery in Ontario. 2020; https://www.cancercareontario.ca/en/guidelines-advice/types-of-cancer/67891. Accessed 2023 Feb 4.
97.Ielapi N, Andreucci M, Licastro N, et al. Precision medicine and precision nursing: The era of biomarkers and precision health. Int J Gen Med. 2020;13:1705-1711. PubMed
98.Khoury MJ, Holt KE. The impact of genomics on precision public health: Beyond the pandemic. Genome Med. 2021;13(1):67. PubMed
99.Luke MJ, Krupetsky N, Liu H, et al. Pharmacists as Personalized Medicine Experts (PRIME): Experiences implementing pharmacist-led pharmacogenomic testing in primary care practices. Pharmacy (Basel). 2021;9(4):201. PubMed
100.Truong TK, Kenneson A, Rosen AR, Singh RH. Genetic referral patterns and responses to clinical scenarios: A survey of primary care providers and clinical geneticists. J Prim Care Community Health. 2021;12:21501327211046734. PubMed
101.Slomp C, Morris E, Gen CS, Price M, Elliott AM, Austin J. The stepwise process of integrating a genetic counsellor into primary care. Eur J Hum Genet. 2022;30(7):772-781. PubMed
102.Blout Zawatsky CL, Leonhard JR, Bell M, et al. Workforce considerations when building a precision medicine program. J Pers Med. 2022;12(11):1929. PubMed
103.McGrath SP, Peabody AE, Jr., Walton D, Walton N. Legal challenges in precision medicine: What duties arising from genetic and genomic testing does a physician owe to patients? Front Med (Lausanne). 2021;8:663014. PubMed
104.Limoges J, Pike A, Dewell S, Meyer A, Puddester R, Carlsson L. Leading Canadian nurses into the genomic era of healthcare. Nurs Leadersh (Tor Ont). 2022;35(2):79-95. PubMed
105.Genome British Columbia. Genome British Columbia's genomics education for healthcare professionals: Implementation framework. 2021; https://www.genomebc.ca/wp-content/uploads/2021/12/Genome-BC-HCP-Education_Phase-II-Implementation-Framework_FINAL.pdf. Accessed 2023 Feb 3.
106.Austin-Tse CA, Jobanputra V, Perry DL, et al. Best practices for the interpretation and reporting of clinical whole genome sequencing. NPJ Genom Med. 2022;7(1):27. PubMed
107.Smith A, Loshak H. Pharmacogenomic testing for medication selection: A rapid qualitative review. Ottawa (ON): CADTH; 2020: https://www.cadth.ca/pharmacogenomic-testing-medication-selection-rapid-qualitative-review. Accessed 2023 Jan 16.
108.Basharat S, Farah K, Horton J. An overview of comprehensive genomic profiling technologies to inform cancer care. Can J Health Technol. 2022;2(8). https://canjhealthtechnol.ca/index.php/cjht/article/view/EH0106. Accessed 2022 Dec 19.
109.Petit C, Croisetiere A, Chen F, Laverdiere I. Are pharmacists from the province of Quebec ready to integrate pharmacogenetics into their practice. Pharmacogenomics. 2020;21(4):247-256. PubMed
110.Sebastian A, Carroll JC, Oldfield LE, et al. Effect of genetics clinical decision support tools on health-care providers' decision making: A mixed-methods systematic review. Genet Med. 2021;23(4):593-602. PubMed
111.CSER Consortium. Guide to interpreting genomic reports: A genomics toolkit. 2020; https://www.genome.gov/sites/default/files/media/files/2020-04/Guide_to_Interpreting_Genomic_Reports_Toolkit.pdf. Accessed 2023 Jan 16.
112.Regier DA, Pollard S, McPhail M, et al. A perspective on life-cycle health technology assessment and real-world evidence for precision oncology in Canada. NPJ Precis Oncol. 2022;6(1):76. PubMed
113.Tarride JE, Gould T, Thomas DM. Challenges of conducting value assessment for comprehensive genomic profiling. Int J Technol Assess Health Care. 2022;38(1):e57. PubMed
114.Pataky RE, Bryan S, Sadatsafavi M, Peacock S, Regier DA. Tools for the economic evaluation of precision medicine: A scoping review of frameworks for valuing heterogeneity-informed decisions. Pharmacoeconomics. 2022;40(10):931-941. PubMed
115.Mittmann N, Chan KK, Cheung M, et al. Platform, basket, and umbrella trial designs: Stakeholder perspectives of novel therapeutics. Can J Health Technol. 2022;2(7). https://canjhealthtechnol.ca/index.php/cjht/article/view/nm003/nm0003. Accessed 2023 Jan 17.
116.Tarride J-E, Cheung M, Hanna TP, et al. Platform, basket, and umbrella trial designs: Issues around health technology assessment of novel therapeutics. Can J Health Technol. 2022;2(7). https://canjhealthtechnol.ca/index.php/cjht/article/view/nm0002. Accessed 2023 Jan 17.
117.INAHTA. INAHTA position statement: Disruptive technologies. 2022; https://www.inahta.org/download/dis-tech-pos-stmt/?wpdmdl=10223. Accessed 2023 Jan 23.
118.Ahead of the curve: Shaping future-ready health systems — 2022-2025 Strategic plan. Ottawa (ON): CADTH; 2022: https://strategicplan.cadth.ca/. Accessed 2023 Jan 23.
Appendix 1: Approach
Project Overview
Advisory Group
An advisory group external to CADTH guided this project. In September 2022, we invited 3 experts to participate as members of the advisory group; these experts had diverse perspectives and expertise in patient experience and care pathways and the social and legal issues related to precision medicine, as well as an awareness of emerging technologies and issues facing health care systems in Canada. The advisory group provided guidance and input in refining the scope of the project, including project definitions, helping identify and refine items for the short list, reviewing the content of the draft report, and identifying potential participants for the workshop. Two members of the group also attended the workshop. All members reviewed the criteria and definitions, provided feedback, refined the items on the short list, and reviewed an earlier version of the final report. All members participated in at least 2 advisory group meetings and were offered an honorarium.
Project Scope and Definitions
We drafted definitions to assist with project scope and clarity and shared the drafts with the advisory group for their feedback; we revised the definitions to ensure accuracy and relevance to the topic of precision medicine and the purpose of the 2023 Watch List. Definitions are presented in Table 1.
Table 1: Definitions Used for Project Scope
Term | Definition used by project |
|---|---|
Precision medicine | An approach that uses the unique genomic information (i.e., genes, gene expression, and proteins) of people to tailor or customize the care they receive, from prevention to diagnosis to treatment to prognosis. |
Precision medicine technologies | Health technologies (any medical devices and diagnostic tests, but not technologies defined as drugs by Health Canada) that detect, characterize, or use the genomic information (i.e., genes, gene expression, and proteins) of a person to inform and tailor their care, from prevention to diagnosis to treatment to prognosis. |
Health technology | Any medical device or diagnostic test; any medical, surgical, or dental procedure; or any other nondrug clinical intervention. Drugs or technologies defined as drugs by Health Canada (such as most stem cell products) are not considered for the list. Technologies without direct patient-related outcomes (e.g., health human resources; heating, ventilation, and air conditioning systems) are excluded. |
New and emerging health technologies | Health technologies that are not yet available in Canada or have been available for clinical use for 1 to 2 years with limited use and are in the launch or early postmarket stage. It may also refer to technologies available in Canada but with diffusion or availability limited to a few health care facilities or centres. An existing technology with new indications could also be considered new or emerging. |
Issue relating to precision medicine technologies | Broader considerations (e.g., clinical, health systems planning, social, ethical, legal, or regulatory) that crosscut the broader scope of precision medicine technologies and could affect their ongoing development, adoption, or implementation in Canadian health systems. |
Key Steps
The process of developing the 2023 Watch List involved 4 key steps, which are briefly described in Figure 1.
Step 1: Identifying Items for the Short List
The purpose of the 2023 Watch List development process was to identify and describe new and emerging precision medicine technologies and issues with the potential to have a substantial impact on health care delivery and planning in Canada in the next 5 years. This impact includes significant and meaningful changes in health and human resources, locations and pathways of health care delivery, patient experiences and outcomes, and health care equity and access. We selected these areas of change because of their relevance to health care policy and planning, including supporting system readiness to adopt and implement precision medicine technologies. We chose a time frame of 5 years to identify precision medicine technologies that were further along the research phase or have the potential to see greater adoption in Canada or similar health care contexts. This time period was intended to limit technologies that are in their early development phase, where their potential value proposition remains largely uncertain, or they are not likely to see substantial adoption outside of limited research settings. Similarly, the 5-year time period for identifying issues provides an opportunity for health care decision-makers to make changes that can guide health systems planning at various levels of health care delivery.
For clarity, we defined “significant and meaningful change” as that which would require the addition of substantial or new types of resources, policies, and procedures, as well as reorganization or transformations in health systems, to successfully adopt, implement, or diffuse technologies.
We identified these domains using information from other CADTH products and processes, the INAHTA position statement on disruptive technologies,117 CADTH’s Strategic Plan,118 and CADTH customer needs. We identified relevant and common criteria across these documents to build domains with prompts and key items that included health system, health care facility, and patient-level issues. We circulated and discussed these draft criteria with the advisory group, which provided input that enabled additional refinement. The final domains and considerations used to select the technologies and issues for the 2023 Watch List are presented in Table 2.
Table 2: Criteria for Selecting Items for the Short List
Domains | Area of significant and meaningful change |
|---|---|
Health care delivery and organization |
|
Health and human resources |
|
Patients’ outcomes and experiences |
|
Health equity and access |
|
CADTH priority themes |
|
We used the criteria in Table 2 to identify items from the literature review and select items for the draft short list by asking:
Does the technology (either as a discrete technology or as a category or type of technology) have a value proposition (i.e., what it is intended to do, what its proposed benefits are) that is anticipated or positioned to make a significant or meaningful change in 1 or more of the criteria in Table 2?
Is the issue anticipated or positioned to make a significant or meaningful change in 1 or more of the criteria in Table 2 and/or does it affect the ability for the value proposition of precision medicine to be realized by patients, clinicians, or health systems?
To identify and add a technology to the draft short list, the author team reviewed information sources (e.g., from market news, blogs, websites, start-ups, venture capitalists), published research, editorials, commentaries, and grey literature. We used the criteria in Table 2 to screen the information sources for eligible and relevant technologies and issues. We compiled information on potential issues and technologies electronically and included the name and a basic description of the technology or issue; its benefit or consequences to patients, clinicians, health systems, and payers; and the criteria for its inclusion. We drafted the extraction table a priori based on the selection criteria, piloted it between 3 authors, and revised the items for consistency and clarity. We met regularly to discuss findings, identify issues and trends in our observations, and note areas of consistency and discrepancies.
Step 2: Short Listing Items for the 2023 Watch List
Once there were approximately 20 items, we individually reviewed the full list of identified items by reflecting on the project scope, definitions, and selection criteria. We discussed the items further to refine definitions of items, and the collapsing, separation, and removal of some items. We shared the draft short list with the advisory group for validation and assessed their credibility (i.e., member checking). Based on the written and oral input of the advisory group, we added, removed, and modified items on the draft short list to create the final short list for step 3. The final short list included 9 technologies (Appendix 2 presents the items on the short list that were not selected for the final list) and 11 issues (Appendix 3 presents the items on the short list that were not selected for the final list).
Step 3: Workshop to Select the Top 10 Technologies and Issues for the 2023 Watch List
We used the process of the JLA Priority Setting Partnerships to guide the workshop and select the top 10 items for the 2023 Watch List. The JLA's principles align with CADTH's priorities of equal involvement and inclusivity (e.g., balanced representation from patients, clinicians, and other CADTH stakeholders), transparency (e.g., visible audit trail of submitted technologies and trends), and a commitment to using and contributing to the evidence base (e.g., using technologies and trends to inform future CADTH products).
Identifying and Recruiting Workshop Participants
We identified potential workshop participants through project scoping, the literature review, CADTH networks, and the advisory group’s recommendations. We invited individuals from a range of geographical settings (i.e., jurisdictions in Canada), and those who had a variety of professional experiences and expertise, such as clinicians and health care providers, patients and carers with lived experiences of precision medicine, funders, payers, health systems decision-makers, and industry representatives. At least 1 member with each type of expertise was invited to participate in the workshop, acknowledging that participants often represented more than 1 category (e.g., academia and clinical). CADTH recognizes the importance of diversity, equity, and inclusion, and the composition of the expert panel took these considerations into account. An invitation was sent to potential participants that described the purpose of the workshop and their role, and recruitment continued until 11 participants were confirmed for the half-day workshop.
The Half-Day Workshop to Select the Top 10 Technologies and Issues
Before the workshop, we provided participants with a workshop guide, the 20-item short list with summaries about each technology and issue, and a participant worksheet. Before attending the workshop, participants were asked to individually review and rank the technologies and issues on the short list using the participant worksheet.
The half-day workshop took place virtually on December 12, 2022. It brought together 11 external experts to identify and prioritize the top 10 items from the 20-item short list. Two CADTH team members, including 1 person who is an advisor with the JLA, facilitated the workshop sessions. The facilitators confirmed that all participants were actively included in the discussion, ensuring that JLA's principle of equal involvement was upheld. Additionally, 5 CADTH team members participated to provide support or take notes.
The workshop had 2 parts: In the first part, participants were split into 2 smaller groups and people shared their perspectives and individual rankings of the items in depth. In the second part, a group session was held with all participants, who worked together through a facilitated discussion, to order the items and select the top 5 technologies and top 5 issues. We chose to select 5 technologies and 5 issues, for total of 10, for consistency in process and ease in communication. These 10 items were not ranked and formed the 2023 Watch List.
Step 4: Preparing the Final Report
We prepared a draft of the final report that described each of the top 10 technologies and issues, their impact on health systems, and examples of the technologies or developments on the issue. The descriptions and examples provided in the report were created using the published literature identified during short listing, additional targeted internet searches, and information described or raised during the workshop. A draft version of the report was shared with the advisory group and was revised based on members’ input.
Appendix 2: Short List of Technologies Not Included in the Final 2023 Watch List
Table 3: Short List of Technologies Not Included in the 2023 Watch List
Technology (category or intervention) | Description |
|---|---|
Liquid biopsy for screening and early detection of cancer | Sequencing technologies that detect mutations or epigenetic markers of cancer from liquid biopsy–based samples (most commonly blood). Use next-generation sequencing and artificial intelligence to detect early signals of cancer in people who are otherwise asymptomatic. |
Liquid biopsy for diagnosing infectious diseases | Blood- or fluid-based next-generation sequencing technologies that can assess blood-borne pathogens, pulmonary infections, complex pneumonia, and urinary tract infections using genomics. |
Direct-to-consumer or at-home genetic testing technologies | Genetic tests delivered directly to consumers (purchased out of pocket by consumers or with private insurance) can provide information about a person’s family history, carrier status, potentially predictive risk factors, and certain pharmacogenomics reports (e.g., drug metabolism). Tests are largely intended to inform future health, using risk prediction. |
Single cell RNA or DNA sequencing technologies | Sequencing (gene or gene expression) of a single or a limited number of cells from a tissue could inform the diagnosis and treatment of reproductive, digestive, neurological, and immunological health conditions. Currently, the technology is being developed to monitor cancer and its treatment response. |
Appendix 3: Short List of Issues Not Included in the Final 2023 Watch List
Table 4: Short List of Issues Not Included in the 2023 Watch List
Issue | Description |
|---|---|
Biases in biobanks | Biobanks do not represent all people and are biased, underrepresenting people from racialized or other population groups historically underrepresented in health research. These biobanks form the basis of algorithms and technologies used to inform care with precision medicine. |
The need for big data | The development of precision medicine requires big data (i.e., huge volumes of data). Big data can come from new datasets (e.g., large, longitudinal cohorts) and/or by accessing and amalgamating existing data (e.g., electronic health records and addressing the interoperability of diverse databases). |
The uses of artificial intelligence in precision medicine | Artificial intelligence is increasingly used to advance the development of precision medicine technologies. The use of artificial intelligence magnifies some existing issues within precision medicine. For example, the use of big data that are biased and will replicate biases, and concerns about data security and privacy. |
The complexity of risk prediction and biomarker identification | There is uncertainty and challenges with the measurement and validation of biomarkers. With more testing being offered and made available to people, there are challenges in characterizing the meaningfulness and actionability of different mutations and biomarkers. For example: What is the associated risk of health conditions due to mutations in single or multiple genes? What factors influence that risk? Existing frameworks of assessing biomarkers may not be sufficient to inform care. |
The potential risks and harms of precision medicine for patients and their families | Precision medicine tests can provide information that might not be actionable at the moment. A greater number of people who are otherwise healthy and asymptomatic may be categorized as “at risk” — in particular, from tests that aim to inform future health or provide screening. These results may introduce risks of psychological harm and uncertainty about whether or not information may become actionable in the future. Patients could be given information that affects their biological relatives. |
Paying out of pocket for precision medicine | Given the limited reimbursement of precision medicine technologies by public health care, patients may only be able to access some forms of precision medicine through out-of-pocket payment or private insurance. |
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up to date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein are those of CADTH and do not necessarily represent the views of Canada’s federal, provincial, or territorial governments or any third-party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for noncommercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.
Questions or requests for information about this report can be directed to Requests@CADTH.ca.