CADTH Health Technology Review
Initiation of Biologic Disease-Modifying Antirheumatic Drugs for Rheumatoid Arthritis: A Budget Impact Analysis
Budget Impact Analysis
Authors: Jesse Elliot, Tessa Cornelissen, Bernice Tsoi, Karen Lee
Abbreviations
bDMARD
biologic disease-modifying antirheumatic drug
BIA
budget impact analysis
csDMARD
conventional synthetic disease-modifying antirheumatic drug
RA
rheumatoid arthritis
Key Messages
There is variation across Canadian jurisdictions in time to the initiation of biologic disease-modifying antirheumatic drug (bDMARD) therapy among adults with rheumatoid arthritis.
From a pan-Canadian perspective, harmonizing time to bDMARD initiation across jurisdictions may result in savings to publicly funded drug plans in some jurisdictions but increased drug expenditures in others.
The extent of savings or increased costs is dependent on jurisdiction, the number of new bDMARD users, and whether patients receive a biosimilar or originator bDMARD.
Executive Summary
Biologic disease-modifying antirheumatic drugs (bDMARDs) may improve outcomes for patients with rheumatoid arthritis (RA) but represent high costs to public drug plans.1 Reimbursement criteria for bDMARDs vary across Canadian jurisdictions, which may contribute to differences in the timing of initiating bDMARD therapy for patients with similar disease status. In a concurrent CADTH study of the utilization of bDMARDs for the treatment of RA across public drug plans in Canada, the longest mean time to a first bDMARD was observed in Alberta, Ontario, and British Columbia, with shorter times observed in Saskatchewan, Manitoba, and the Atlantic provinces. This budget impact analysis (BIA) examined the potential budgetary impact to Canadian provincial public drug plans of harmonizing the duration of conventional synthetic disease-modifying antirheumatic drug (csDMARD) use before initiating bDMARD therapy across jurisdictions.
An applied, policy-oriented BIA was developed to facilitate the consideration of alternative scenarios representing harmonized time to the initiation of bDMARD therapy from the perspective of the Canadian publicly funded drug plans. Three scenarios were considered: a reference scenario and 2 harmonization scenarios. The reference scenario reflects the status quo and was informed by the findings of the CADTH Utilization Study,2 which assessed the jurisdiction-specific duration of csDMARD use before the initiation of bDMARD therapy for RA across public drug plans in Canada. Notably, an approximately 4-month difference in csDMARD use before the initiation of bDMARD therapy was found between patients in the Atlantic provinces, Manitoba, and Saskatchewan and those in Ontario, Alberta, and British Columbia. The reference scenario was compared with 2 harmonization scenarios, which considered alternative durations of csDMARD use before the initiation of bDMARD therapy, such that time to a first bDMARD was harmonized across jurisdictions (i.e., mean duration of csDMARD use before bDMARD therapy was aligned within approximately 1 month across jurisdictions). The first harmonization scenario considers the impact of increasing the mean time to the initiation of bDMARD therapy by 4 months for patients in the Atlantic provinces, Manitoba, and Saskatchewan, while the second scenario considers the impact of reducing the mean time to the initiation of bDMARD therapy by 4 months for Ontario, Alberta, and British Columbia. In both harmonization scenarios, time to the initiation of bDMARD therapy was assumed to be unchanged for the remaining jurisdictions. The BIA considers a cohort of patients who are newly diagnosed with RA in 2018. Patients are assumed to initiate treatment with a csDMARD at the time of diagnosis and to continue csDMARD treatment until initiating bDMARD therapy, the timing of which is varied between the reference and harmonization scenarios. The cumulative time on csDMARD and bDMARD therapy was analyzed over the subsequent 3-year period to explore the impact to the public drug plans of initiating bDMARDs at various time points after diagnosis.
From a pan-Canadian perspective, CADTH estimated that increasing the time to the initiation of bDMARD therapy by 4 months in the Atlantic provinces, Manitoba, and Saskatchewan would result in a savings of $9,555,958 over a 3-year period. Conversely, decreasing the time to the initiation of bDMARD therapy by 4 months in Ontario, Alberta, and British Columbia would result in an incremental cost to the drug plans of $41,725,061 over 3 years. The projected impact of these changes varied by jurisdiction based on the number of new users. The findings were robust to most assumptions. However, when all new users were assumed to start on a biosimilar bDMARD (instead of an originator, if available), the impact of harmonizing the time to the initiation of bDMARD therapy was reduced by approximately 25% in both harmonization scenarios.
Key limitations include assuming that the market share of each bDMARD would remain unchanged over a 3-year analysis period, which does not account for the introduction of new bDMARDs, including new biosimilars. The introduction of new biosimilars may result in lower overall costs and reduce the differential between the reference and harmonization scenarios. Additionally, time to the initiation of bDMARD therapy was based on observed data for patients aged 67 years and older and was assumed to apply to all new adult users. The number of new bDMARD users with RA may be overestimated owing to the use of claims-based methodology in the Utilization Study, which may have captured patients receiving bDMARDs for the treatment of other rheumatic disease (e.g., ankylosing spondylitis, psoriatic arthritis). Individual results for the Atlantic provinces (i.e., Nova Scotia, New Brunswick, Prince Edward Island, Newfoundland and Labrador) are unavailable owing to low patient numbers in the concurrent Utilization Study. Finally, this analysis does not consider the health impacts of initiating bDMARD treatment at different times.
Harmonizing time to the initiation of bDMARD therapy across Canadian jurisdictions will have a budgetary impact on the public drug plans. From a pan-Canadian perspective, increasing time to the initiation of bDMARD therapy may result in savings to publicly funded drug plans, while decreasing time to bDMARD initiation may result in increased drug expenditures. The extent of savings or costs is dependent on jurisdiction, the number of new bDMARD users, and whether patients receive a biosimilar or originator bDMARD.
Budget Impact Analysis
Background
Rheumatoid arthritis (RA) is a chronic, systemic, autoimmune disease3,4 that affects physical function and health-related quality of life.5 Approximately 1.25% of Canadians are estimated to have RA, with a higher prevalence among women.6 The goal of treatment is remission and, if not possible, to reduce disease activity, control symptoms, prevent disability, and improve quality of life.7 First-line treatment of RA typically consists of conventional synthetic disease-modifying antirheumatic drugs (csDMARDs; also called traditional disease-modifying antirheumatic drugs), including methotrexate, sulfasalazine, hydroxychloroquine, and leflunomide.7 For those with an inadequate response to csDMARDs (i.e., moderate to high disease activity despite treatment with at least 2 csDMARDs), biologic DMARDs (bDMARDs) may be considered.7 The introduction of bDMARDs has resulted in increased public drug program spending and public health care payers have implemented criteria for their use in the treatment of RA.8 However, such strategies have been applied differently across provinces and territories,8 resulting in variation in public coverage for bDMARDs. A recent CADTH Environmental Scan of the coverage criteria for bDMARDs across Canadian public drug plans found that the reimbursement criteria are largely comparable across jurisdictions in the drugs covered but differ in the number of lines of prior therapy with csDMARDs required before accessing a bDMARD.8 As noted in the CADTH study of the utilization of csDMARDs and bDMARDs for the treatment of RA across public drug plans in Canada (hereafter referred to as the “Utilization Study”),2 the time to the initiation of bDMARD therapy among new users aged 67 years and older in Canada varies across jurisdictions, with the longest time to bDMARD initiation observed in Alberta, Ontario, and British Columbia, and shorter time to a first bDMARD in Saskatchewan, Manitoba, and the Atlantic provinces.
As part of the CADTH Technology Review of the harmonization of public coverage policies for biologic drugs for the treatment of RA, a budget impact analysis (BIA) was undertaken to explore the potential budgetary impact to Canadian public drug plans of harmonizing time to the initiation of bDMARD therapy across jurisdictions.
Decision Problem
The objective of this BIA was to address the following question:
What is the budget impact of harmonizing time to the initiation of bDMARD therapy across publicly funded drug plans in Canada compared to current jurisdiction-specific time to bDMARD?
Specifically, an applied BIA was constructed to facilitate the consideration of the financial impact of harmonizing time to bDMARD initiation for the treatment RA in adults. The BIA was conducted from the perspective of Canadian publicly funded provincial drug plans, excluding Quebec, over a 3-year time horizon. Owing to the drug plan perspective, only costs covered by the public drug plans were captured (i.e., drug costs). Time to bDMARD initiation was jurisdiction-specific (Table 2), as found in the accompanying Utilization Study.2
Two scenarios were considered, termed “harmonization scenarios.” In both, the duration of csDMARD use before the initiation of bDMARD therapy among bDMARD-naive patients was aligned so that mean time to a first bDMARD was within the same time frame (i.e., approximately within a 1-month range) in all jurisdictions. Other variables (e.g., prescribing behaviour, wait times) were assumed to be unaffected by the harmonization scenarios.
Harmonization Scenario 1: Time to the initiation of bDMARD therapy was assumed to be increased by 4 months, compared to the jurisdiction-specific current time to bDMARD initiation, for the Atlantic provinces, Manitoba, Saskatchewan.
Harmonization Scenario 2: Time to the initiation of bDMARD therapy was assumed to be decreased by 4 months, compared to the jurisdiction-specific current time to bDMARD initiation, for Alberta, British Columbia, and Ontario.
Study Design and Methods
A BIA was conducted using an Excel-based tool developed for this project. This tool has the flexibility to conduct various scenario analyses and to report the estimates of budget impact disaggregated by jurisdiction. The ISPOR — International Society for Pharmacoeconomics and Outcomes Research — principles of good practice for BIAs were followed for the design and conduct of the BIA.9
Patient Population
The population considered within this analysis consists of bDMARD-naive RA patients. Specifically, the population included patients taking a bDMARD for the first time following the use of at least 1 csDMARD (termed “new users”) as defined in the Utilization Study.2 It was assumed that harmonizing the time to the initiation of bDMARD therapy would not change the current eligibility for bDMARD therapy as defined by the public drug programs and would thus not affect the number of people eligible for bDMARD therapy. No stratification of the target population was incorporated into the analysis.
A closed cohort was considered over the 3-year time horizon such that no new users were added after the first year and mortality was assumed to be negligible. The starting year for the cohort was 2018, consistent with the most recent year of utilization data available.2 Thus, in the CADTH base case, the results reflect the impact of harmonizing time to bDMARD initiation for a cohort of newly diagnosed patients initiating csDMARD treatment in 2018; the impact of initiating bDMARDs at different times on the overall cost of treatment over the subsequent 3 years was assessed. In a scenario analyses, alternative starting years were considered.
The population for the BIA (i.e., new bDMARD users) was defined based on data from a CADTH Utilization Study.2 Because the Non-Insured Health Benefits program, federal public drug plans, and Quebec were not captured in the Utilization Study, these programs are similarly not reflected in the BIA. In the Utilization Study, Nova Scotia, New Brunswick, Prince Edward Island, and Newfoundland and Labrador are grouped as “the Atlantic provinces” because of low patient numbers; this convention was similarly adopted in the BIA.
Harmonization Scenarios
Three scenarios were considered in the BIA: a reference scenario that reflects the 2018 utilization of bDMARDs and jurisdiction-specific mean time to bDMARD initiation for adults with RA; and 2 new harmonization scenarios in which the time to a first bDMARD is harmonized across jurisdictions. Time to bDMARD initiation was conceptualized as the duration of time between the initiation of a csDMARD to the initiation of a first bDMARD. The harmonization scenarios were defined based on data from the Utilization Study,2 which found an approximately 4-month difference in the mean duration of csDMARD use before bDMARD initiation between patients in the Atlantic provinces, Manitoba, and Saskatchewan and those in Ontario, Alberta, and British Columbia.
Reference Scenario
The reference scenario reflects the mean jurisdiction-specific time to the initiation of bDMARD therapy based on the concurrent Utilization Study.2 The study focused on the 67 years and older age group to capture a homogenous patient population across public drug plans. Patients aged 65 years and younger were assumed to potentially transition between multiple sources of coverage (e.g., public, private, out of pocket), which would impact the observed time to a bDMARD. In contrast, all patients aged 67 years and older are assumed to be eligible for public coverage. The jurisdiction-specific duration of csDMARD use before the initiation of bDMARD therapy observed among patients aged 67 years and older was assumed to be consistent across all adult new users in that jurisdiction.
Harmonization Scenarios
The harmonization scenarios were informed by the CADTH Utilization Study.2 Based on the findings of the Utilization Study, jurisdictions were grouped as having either a longer or shorter time to the initiation of bDMARD therapy. Group 1 comprised jurisdictions with a shorter mean time to bDMARD initiation (the Atlantic provinces, Manitoba, and Saskatchewan), whereas Group 2 comprised jurisdictions with a longer mean time to bDMARD initiation (Ontario, Alberta, British Columbia).
In Harmonization Scenario 1, the jurisdiction-specific mean time to bDMARD initiation (Table 2) was increased by 4 months for each jurisdiction in Group 1 (i.e., patients were assumed to remain on csDMARDs for an additional 4 months before initiating a bDMARD). Time to bDMARD initiation was assumed to be unchanged for the remaining jurisdictions.
In Harmonization Scenario 2, the jurisdiction-specific mean time to bDMARD initiation (Table 2) was decreased by 4 months for each jurisdiction in Group 2. The time to bDMARD initiation was assumed to be unchanged for the remaining jurisdictions.
Time Horizon
The time horizon of the analysis was 3 years (2018 to 2020). Based on data availability, 2018 was adopted as the starting year of the cohort, as this is the most recent year with available utilization data.2 No discounting was applied to the analysis.
Perspective
The perspective of this analysis was Canadian publicly funded drug plans, excluding Quebec and federal drug plans. Therefore, only costs covered by public drug plans were captured (i.e., broader health care system costs; costs to individuals and private payers were not incorporated).
Analytic Framework Description
An applied, policy-oriented economic BIA model was created to facilitate the consideration of alternative scenarios reflecting the timing of initiating bDMARD therapy. For consistency between the Utilization Study2 and the BIA, the starting year of the analysis was assumed to be 2018 and the number of new users and market share between bDMARDs was assumed to be static (i.e., no forecasting of market changes was undertaken). The model schematic is shown in Figure 1.
The Reference and Harmonization Scenarios were used to estimate the incremental changes (costs or savings) resulting from the adoption of each alternative time to bDMARD initiation. For each jurisdiction, the total cost of bDMARD treatment per patient over the 3-year analysis horizon was estimated for the Reference Scenario and for each harmonization scenario. The total cost for each scenario was calculated by summing the annual cost associated with each bDMARD by the total duration of bDMARD use across all new users during the analysis period.
The budget impact is represented by the incremental costs (or savings) between scenarios over the 3-year analysis horizon. The incremental budget impact was determined separately for each alternative harmonization scenario compared to the Reference Scenario.
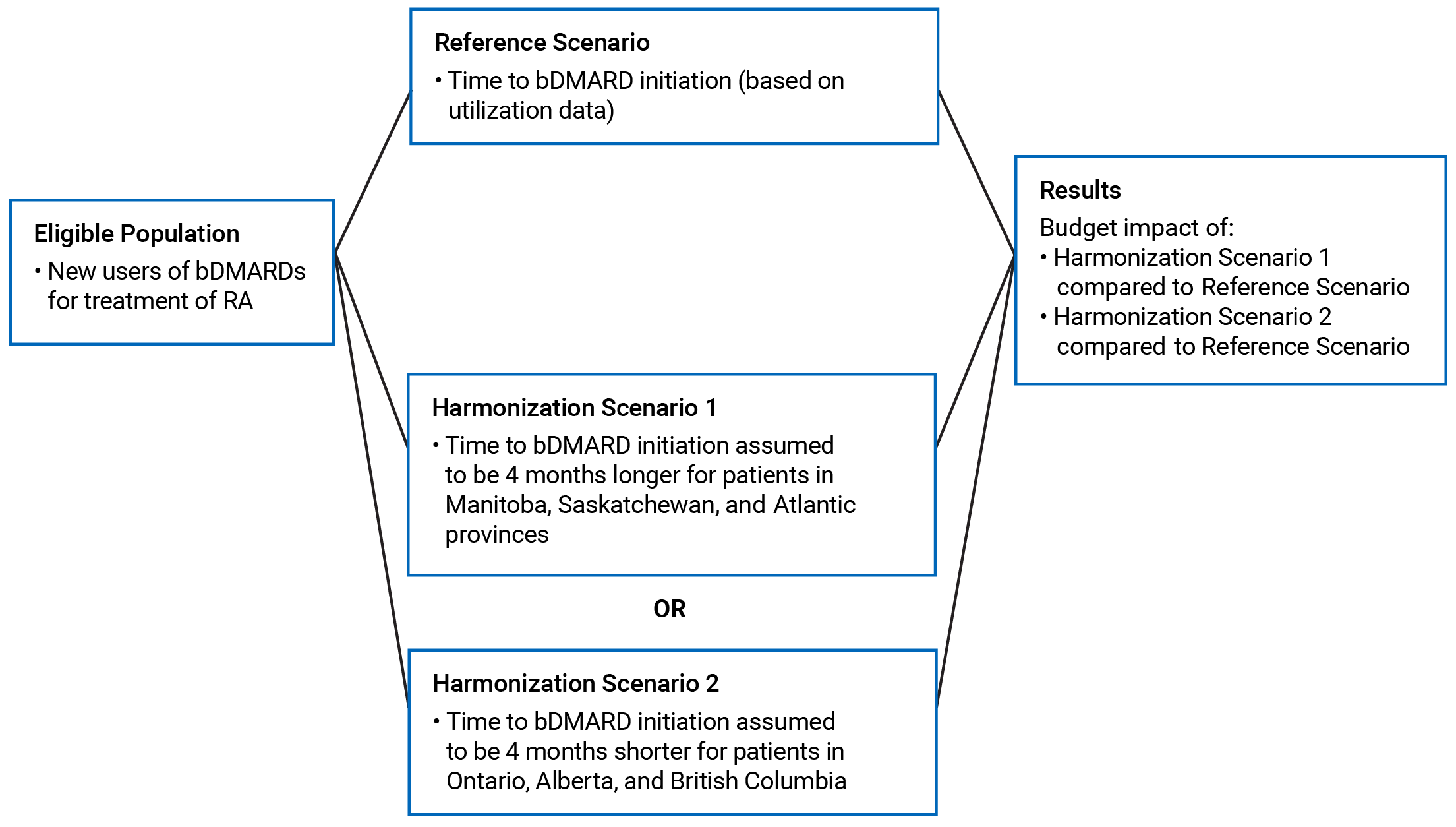
bDMARD = biologic disease-modifying antirheumatic drug; RA = rheumatoid arthritis.
Model Inputs
Population Inputs
The number of new bDMARD users (Table 1) and the duration of csDMARD use before bDMARD initiation (Table 2) across jurisdictions was obtained from the Utilization Study.2 New bDMARD users (aged3 18 years) were defined as individuals who had at least 1 RA biologic drug claim and no claims for any RA biologic drug in the 365 days before their first claim for an RA biologic drug (first prescription). The duration of csDMARD use before a first bDMARD was based on data pertaining to new users aged 67 years and older,2 and was assumed to be consistent for all adult RA patients.
Table 1: Number of New bDMARD Users, by Jurisdiction
bDMARD | Number of new users (3 18 years), 2018 | |||||
Atlantic Provinces | Ontario | Manitoba | Saskatchewan | Alberta | British Columbia | |
Abatacept (originator) | 23 | 90 | NRa | 14 | 50 | 96 |
Adalimumab (originator) | 278 | 1,374 | 189 | 148 | 564 | 645 |
Certolizumab (originator) | 36 | 112 | NRa | 24 | 49 | 89 |
Etanercept (originator) | 57 | 225 | 16 | 10 | 77 | 77 |
Etanercept (biosimilars) | 27 | 395 | 49 | 47 | 49 | 190 |
Golimumab (originator) | 118 | 238 | 38 | 79 | 142 | 132 |
Infliximab (originator) | 112 | 482 | 6 | NRa | 240 | 53 |
Infliximab (biosimilars) | 7 | 179 | 30 | NRa | 59 | 86 |
Rituximab (originator) | 29 | 272 | 53 | 60 | 50 | 266 |
Tocilizumab (originator) | 16 | 117 | 18 | 12 | 52 | 63 |
bDMARD = biologic disease-modifying antirheumatic drug; NR = not reported because of suppressed cell counts.
aSuppressed cells were assumed to include 5 patients in the base case.
Source: CADTH Utilization Study.2
Table 2: Duration of csDMARD Use Prior to bDMARD Initiation, by Jurisdiction
Scenario | Duration of csDMARD use,a mean days | |||||
Atlantic Provinces | Ontario | Manitoba | Saskatchewan | Alberta | British Columbia | |
Reference scenarioa | 664 | 748 | 668 | 681 | 792 | 749 |
Harmonization Scenario 1 | 786 | 748 | 790 | 803 | 792 | 749 |
Harmonization Scenario 2 | 664 | 626 | 668 | 681 | 670 | 627 |
bDMARD = biologic disease-modifying antirheumatic drug; csDMARD = conventional synthetic disease-modifying antirheumatic drug.
aBased on the average time to initiate biologic therapy for publicly insured rheumatoid arthritis new users ≥ 67 years old, 2015 to 2018.2
Market Share
The market share attributed to each bDMARD was based on the number of unique users of each bDMARD from the Utilization Study2 (Table 1) and was assumed to be constant for the 3-year analysis horizon.
Cost Inputs
The BIA incorporated drug acquisition costs for bDMARDs. Where possible, publicly available, jurisdiction-specific prices for bDMARDs were used to estimate the cost of each treatment approach; where this was not available, prices from Ontario were used as a proxy in the base case. For bDMARDs with initial loading or initiation dosing, separate costs for the first and subsequent years of treatment were included in the model. Drug administration, markup, and dispensing fees were not included in the analysis and drug prices were assumed to be constant over the analysis horizon.
The recommended dosage and administration schedule for each bDMARD for the treatment of RA was obtained from the Health Canada–approved product monograph (Table 9); these were used in the calculation of the number of units used per patient for each bDMARD. For bDMARDs with multiple biosimilars (e.g., infliximab), the most expensive was included in the cost calculation in the base case.
Table 3: Annual Cost of bDMARD Treatment
bDMARD | Average Annual Cost ($) Per Patienta,b,c,d | |||||
Atlantic Provincese | Ontario | Manitobae | Saskatchewan | Alberta | British Columbia | |
Abatacept (originator) | SC: 19,437 IV: 22,515 | SC: 19,437 IV: 22,515 | SC: 19,437 IV: 22,515 | SC: 19,437 IV: 22,515 | SC: 19,437 IV: 22,515 | SC: 20,409 IV: 23,641 |
Adalimumab (originator) | 20,422 | 20,422 | 20,422 | 20,422 | 20,422 | 21,443 |
Certolizumab (originator) | 19,935 | 19,935 | 19,935 | 19,935 | 19,935 | 20,932 |
Etanercept (originator) | 21,111 | 21,111 | 21,111 | 21,111 | 20,880 | 19,890 |
Etanercept (biosimilar) | 12,532 | 12,532 | 12,532 | 12,532 | 12,532 | 13,535 |
Golimumab (originator) | SC: 18,662 IV: 21,108 | SC: 18,662 IV: 21,108 | SC: 18,662 IV: 21,108 | SC: 18,666 IV: 21,108 | SC: 18,19 IV: 21,108 | SC: 40,013 IV: 21,108 |
Infliximab (originator) | 23,701 | 23,701 | 23,701 | 23,448 | 23,701 | 23,701 |
Infliximab (biosimilar) | 12,600 | 12,600 | 12,600 | 15,600 | 12,600 | 13,230 |
Rituximab (originator) | 19,292 | 19,292 | 19,292 | 19,292 | 19,292 | 20,257 |
Tocilizumab (originator) | SC: 9,332 IV: 11,882 | SC: 9,332 IV: 11,882 | SC: 9,332 IV: 11,882 | SC: 9,332 IV: 11,882 | SC: 9,332 IV: 11,882 | SC: 9,798 IV: 12,476 |
bDMARD = biologic disease-modifying antirheumatic drug; SC = subcutaneous.
aFirst year of treatment. Subsequent year costs were incorporated for bDMARDs with alternative initiation and maintenance dosing.
bFor biosimilars with multiple available products, the most expensive within each jurisdiction was included in the base case.
cDosages, administration schedules, and prices used in the determination of annual costs are available in Appendix 1.
dOntario costs were used as proxy costs for the Atlantic provinces and Manitoba, as bDMARD prices were not publicly available for these jurisdictions.
The cost of csDMARDs was not included in the analysis because it was assumed that csDMARD usage would not be affected by the harmonization of the time to bDMARD initiation (i.e., csDMARD usage would be similar between the reference and harmonization scenarios). The clinical expert consulted by CADTH for this BIA indicated that, for patients initiating a first bDMARD, the bDMARD would be added to a background of csDMARDs and that the number and dosage of csDMARDs would remain constant for at least the first 3 to 4 months of treatment. Any differences in the number or dosages of csDMARDs between scenarios were deemed unlikely to drive the conclusions of the analysis owing to the low price of csDMARDs relative to bDMARDs.
Analyses
Table 4 summarizes the key assumptions made in the base-case analysis of the BIA. Some base-case assumptions were tested in scenario and sensitivity analyses using a range of different assumptions. The scenarios explored and the inputs used for sensitivity analyses are presented in Table 5.
Table 4: Key Base-Case Assumptions
Parameter | Assumption | Scenario analysis |
Analysis starting year | 2018.a | 2021: Additional assumptions about jurisdiction-specific population growth and market share were required (described in the following section). |
bDMARD market share | Assumed to remain constant for the 3-year analysis horizon. | New users were assumed to preferentially start on a biosimilar bDMARD (i.e., new users of infliximab were assumed to all receive the biosimilar form). Scenarios with a starting year of 2021 also incorporated new bDMARDs introduced since 2018. |
Patient cohort | The number of users in the cohort was assumed to be constant over the 3-year model horizon (i.e., no new users were added to the cohort, and mortality was assumed to be negligible). | None. |
Number of new users | For each cell with data suppressed owing to low patient counts (i.e., ≤5), it was assumed that there were 5 patients per cell. | Suppressed cells were assumed to have a zero count. |
bDMARD dosage | bDMARD dosage was assumed to not change based on clinical response. This is a simplifying assumption, as bDMARD dosage may be increased or stepped down depending on the treatment response. The clinical expert consulted by CADTH indicated that any dosage changes would not occur for at least the first 3 to 4 months after initiating a bDMARD. | None. |
bDMARD administration form | All new users of abatacept, golimumab, and tocilizumab were assumed to receive the subcutaneous form in the base case. | All new users of abatacept, golimumab, and tocilizumab were assumed to receive the IV form. |
bDMARD cost | Publicly available, jurisdiction-specific drug prices were used, where available. Prices for Ontario were used as a proxy for jurisdictions where public drug prices were unavailable. | Publicly available drug prices for British Columbia were used as a proxy for jurisdictions where public drug prices were unavailable. |
csDMARD usage | The number and dosage of background csDMARDs was assumed to not change with the addition of bDMARDs, or to be affected by the harmonization scenarios. | None. |
Duration of csDMARD use | The duration of csDMARD use before the initiation of the first bDMARD for patients aged 67 years and older was assumed to be consistent across all age groups. | None. |
Patient weight | Patient weight was assumed to be 75 kg (for bDMARDs with weight-based dosing). | Higher and lower patient weights were assumed in scenario analyses. |
Health impacts | Changes to health outcomes because of different durations of csDMARD use before bDMARD initiation were not modelled (i.e., changes to the timing of bDMARD initiation were assumed to have no effect on health outcomes or health care system costs). | None. |
bDMARD = biologic disease-modifying antirheumatic drug; csDMARD = conventional synthetic disease-modifying antirheumatic drug.
a2018 reflects the most recent data available from the CADTH Utilization Study2 regarding the number of new users by jurisdiction.
Uncertainty and Scenario Analyses
Sensitivity and scenario analyses were conducted to explore the impact of parameter uncertainty and assumptions made in the base case on the BIA results. These included adopting an alternative patient weight — assuming that new users would preferentially start on a biosimilar form — using price data from an alternative jurisdiction as a proxy for those without publicly available drug prices and adopting 2021 as the starting year of the cohort. Details of the analyses are available in Table 5.
Parameter | CADTH base case | CADTH scenarios |
1. Patient weight | 75 kg | 50 kg |
2. Patient weight | 75 kg | 100 kg |
3. bDMARD administration form (abatacept, golimumab, tocilizumab) | All new users assumed to receive the subcutaneous form | All new users assumed to receive the IV form |
4. Province used as proxy for bDMARD prices (for those without publicly available prices) | Ontario | British Columbia |
5. bDMARD biosimilar price (for those with multiple biosimilar forms) | Most expensive | Least expensive |
6. Suppressed cell counts | Assumed to represent 5 new users | Assumed to represent 1 new user |
7. Cohort starting year | 2018 | 2021a |
8. Cohort starting year | 2018 | 2021, with all new bDMARD users assumed to start on a biosimilara |
9. Cohort starting year | 2018 | 2021, with new bDMARDs introduced since 2018 incorporateda,b |
10. Duration of csDMARD use | Jurisdiction and harmonization scenario-specific: Assumed to increase by 4 months in Harmonization Scenario 1 and to decrease by 4 months in Harmonization Scenario 2, depending on jurisdiction (Figure 1) | Time to bDMARD initiation was harmonized across jurisdictions by assuming that jurisdiction-specific current time was concurrently increased by 2 months in the Atlantic provinces, Manitoba, and Saskatchewan, and decreased by 2 months in Alberta, British Columbia, and Ontario (Harmonization Scenario 3) |
bDMARD = biologic disease-modifying antirheumatic drug.
aThe number of new users was increased by jurisdiction-specific population growth rates; all other parameters were held constant unless otherwise stated.
b10% of new users were assumed to start on sarilumab, with market share taken from tocilizumab, on the basis of clinical expert opinion. All new users for adalimumab, etanercept, infliximab, and rituximab were assumed to start on the biosimilar form.
Results
The pan-Canadian results of the BIA are presented here. Full disaggregated results are presented in Appendix 1.
CADTH Base Case
Harmonization Scenario 1
From a pan-Canadian perspective (excluding Quebec), CADTH estimated that increasing the time to the initiation of bDMARD therapy by 4 months in each of the Atlantic provinces, Manitoba, and Saskatchewan would result in a savings of $9,555,958 over 3 years (Table 6). The projected savings were greatest in the Atlantic provinces (Table 10) because of highest number of new users (i.e., compared to the number of new users in Manitoba and Saskatchewan).
Table 6: Pan-Canadian Budget Impact Analysis Results — Harmonization Scenario 1
Scenario | Three-year total costs ($) |
Reference Scenario | 151,922,685 |
Harmonization Scenario | 142,366,726 |
Impact | –9,555,958 |
Note: Harmonization Scenario 1 — Time to the initiation of biologic disease-modifying antirheumatic drug therapy was increased by 4 months for the Atlantic provinces, Manitoba, and Saskatchewan.
Harmonization Scenario 2
From a pan-Canadian perspective (excluding Quebec), CADTH estimated that decreasing the time to the initiation of bDMARD therapy by 4 months in each of Ontario, Alberta, and British Columbia would result in an incremental cost of $41,725,061 over 3 years (Table 7). By jurisdiction, the projected costs were greatest in Ontario (Table 11) because of the highest number of new users (i.e., compared to the number in Manitoba and Saskatchewan).
Table 7: Pan-Canadian Budget Impact Analysis Results – Harmonization Scenario 2
Scenario | Three-year total costs ($) |
Reference Scenario | 151,922,685 |
Harmonization Scenario | 193,647,746 |
Impact | 41,725,061 |
Note: Harmonization Scenario 2 — Time to the initiation of biologic disease-modifying antirheumatic drug therapy was assumed to be decreased by 4 months for Alberta, British Columbia, and Ontario.
Scenario and Sensitivity Analyses
The pan-Canadian results of key scenario analyses are presented in Table 8 (jurisdiction-specific results are presented in Table 12 and Table 13). Of these, from a pan-Canadian perspective, none had a large effect on the expected impact of harmonizing time to bDMARD initiation among new users, with the exception of including new bDMARDs introduced since 2018 (i.e., biosimilars for adalimumab and rituximab, sarilumab originator) and assuming that all new users would receive a biosimilar form (Scenario 9). From a pan-Canadian perspective, the Harmonization Scenario 1 assumption that all new users would initiate on a biosimilar form (where available) resulted in an approximately 25% reduction in savings, while in Harmonization Scenario 2, this assumption resulted in approximately 24% lower costs.
In scenario analyses, CADTH explored the impact of a third harmonization scenario in which the duration of time to bDMARD initiation was harmonized across jurisdictions by increasing or decreasing the current mean time to a first bDMARD by 2 months, depending on the jurisdiction (i.e., changes in time to bDMARD initiation were implemented for all public plans). Specifically, time to the initiation of bDMARD therapy was assumed to be concurrently increased by 2 months in the Atlantic provinces, Manitoba, and Saskatchewan, and decreased by 2 months in Alberta, British Columbia, and Ontario. Compared to the reference scenario, this harmonization scenario resulted in higher overall costs to the drug plans from a pan-Canadian perspective because of the increased time on bDMARDs in Alberta, British Columbia, and Ontario ($16 million over 3 years). Jurisdiction-specific results for this scenario are presented in Table 12.
Table 8: Key Scenario Analysis Results
Analysis | Scenario | Harmonization Scenario 1a ($) | Harmonization Scenario 2b ($) |
Base case | Reference Scenario | 151,922,685 | 151,922,685 |
Harmonization Scenario | 142,366,726 | 193,647,746 | |
Impact | –9,555,958 | 41,725,061 | |
Scenario 1: Lower patient weight (50 kg) | Reference Scenario | 143,720,725 | 143,720,725 |
Harmonization Scenario | 134,523,616 | 183,178,719 | |
Impact | –9,197,109 | 39,457,994 | |
Scenario 2: Higher patient weight (100 kg) | Reference Scenario | 154,448,408 | 154,448,408 |
Harmonization Scenario | 144,749,350 | 196,904,986 | |
Impact | –9,699,057 | 42,456,578 | |
Scenario 3: All users assumed to receive IV forms of abatacept, golimumab, and tocilizumab | Reference Scenario | 152,391,754 | 152,391,754 |
Harmonization Scenario | 142,819,055 | 192,766,937 | |
Impact | –9,572,700 | 40,375,182 | |
Scenario 4: British Columbia drug prices adopted as proxy for Atlantic and Manitoba drug prices | Reference Scenario | 156,609,782 | 156,609,782 |
Harmonization Scenario | 145,728,151 | 198,334,843 | |
Impact | –10,881,631 | 41,725,061 | |
Scenario 5: Least expensive bDMARD price incorporated | Reference Scenario | 151,634,893 | 151,634,893 |
Harmonization Scenario | 142,093,761 | 193,284,146 | |
Impact | –9,541,131 | 41,649,253 | |
Scenario 6: Suppressed cell counts assumed to represent 1 new user | Reference Scenario | 151,565,711 | 151,565,711 |
Harmonization Scenario | 142,109,897 | 193,290,772 | |
Impact | –9,455,814 | 41,725,061 | |
Scenario 7: Cohort starting year assumed to be 2021c | Reference Scenario | 157,642,026 | 157,642,026 |
Harmonization Scenario | 147,857,893 | 201,101,733 | |
Impact | –9,784,133 | 43,459,707 | |
Scenario 8: Cohort starting year assumed to be 2021c and all new users assumed to start on a biosimilar (if available) | Reference Scenario | 144,067,076 | 144,067,076 |
Harmonization Scenario | 134,960,383 | 183,760,572 | |
Impact | –9,106,693 | 39,693,497 | |
Scenario 9: Cohort starting year assumed to be 2021,c all new users assumed to start on a biosimilar, and bDMARDs introduced since 2018 incorporatedd | Reference Scenario | 114,667,490 | 114,667,490 |
Harmonization Scenario | 107,487,961 | 146,208,669 | |
Impact | –7,179,528 | 31,541,180 | |
Scenario 10: Harmonization Scenario 3 (time to bDMARD initiation is adjusted by 2 months across jurisdictions)e | Reference Scenario | 151,922,685 | |
Harmonization Scenario | 168,193,140 | ||
Impact | 16,270,455 | ||
bDMARD = biologic disease-modifying antirheumatic drug.
Note: Negative values denote cost savings.
aHarmonization Scenario 1 — Time to a first bDMARD was increased by 4 months for the Atlantic provinces, Manitoba, and Saskatchewan.
bHarmonization Scenario 2 — Time to a first bDMARD was assumed to be decreased by 4 months for Alberta, British Columbia, and Ontario.
cThe number of new users was increased by jurisdiction-specific population growth rates; all other parameters were held constant unless otherwise stated.
dMarket share for sarilumab was estimated based on input provided by the clinical expert consulted by CADTH for this review.
eTime to bDMARD initiation was harmonized across jurisdictions by assuming that jurisdiction-specific current time was concurrently increased by 2 months in the Atlantic provinces, Manitoba, and Saskatchewan, and decreased by 2 months in Alberta, British Columbia, and Ontario.
Summary of Findings
In this BIA, CADTH explored the costs to Canadian public drug plans associated with harmonizing the duration of time on csDMARDs before the initiation of bDMARD therapy among new users across jurisdictions. As found in the CADTH Utilization Study2 of bDMARDs for the treatment of RA across public drug plans in Canada, the duration of csDMARD use before the initiation of a first bDMARD varies across jurisdictions. Notably, the Utilization Study found a divergence between British Columbia, Alberta, and Ontario compared to Manitoba, Saskatchewan, and the Atlantic provinces, with an approximately 4-month difference in time to bDMARD initiation among patients in the former and latter jurisdictions. Thus, in this BIA, CADTH explored the budgetary impact to the public drug plans of harmonizing the time to bDMARD initiation such that patients in all jurisdictions would have a similar duration of csDMARD use before initiating a first bDMARD.
Two harmonization scenarios were considered based on the findings of the Utilization Study, such that mean time to the initiation of bDMARD therapy would be similar (i.e., within 1 month) across jurisdictions.2 In the first scenario, jurisdiction-specific time to bDMARD initiation was assumed to be increased by 4 months, compared to current time to bDMARD initiation, for the Atlantic provinces, Manitoba, and Saskatchewan. In the second scenario, jurisdiction-specific time to bDMARD initiation was assumed to be decreased by 4 months, compared to current usage patterns, for Alberta, British Columbia, and Ontario. The total mean duration of csDMARD use before bDMARD initiation in the reference and harmonization scenarios by jurisdiction are provided in Table 2. As noted in the Utilization Study, the differences in duration of csDMARD use before initiating bDMARD therapy observed in the reference scenario may be owing in part to differences in coverage criteria in the number of lines of therapy that are required to be trialled before accessing publicly reimbursed bDMARD therapy. Both harmonization scenarios assumed that all variables other than time to a first bDMARD are constant between the reference and harmonization scenarios (e.g., no changes to prescribing behaviour, wait times).
In the first Harmonization Scenario, increasing time to the initiation of bDMARD therapy by 4 months for the Atlantic provinces, Manitoba, and Saskatchewan was associated with expected savings of $9,555,958 to the drug plans over the 3-year analysis period compared to the reference scenario. The projected savings were greatest in the Atlantic provinces because of the highest number of new users. In the second Harmonization Scenario, reducing time to bDMARD initiation by 4 months in each of Ontario, Alberta, and British Columbia was associated with increased costs of $41,725,061 to the drug plans over the 3-year analysis period, relative to the reference scenario. The projected savings were greatest in Ontario because of the highest number of new users.
Several scenario and sensitivity analyses were conducted to explore the uncertainty associated with these findings. These included adopting alternative patient weight, assuming that new users would preferentially start on biosimilar bDMARDs, using proxy price data from an alternative jurisdiction for those without publicly available drug prices, and adopting 2021 as the starting year of the cohort. The findings were robust to all scenarios considered, except when 2021 was adopted as the starting year and new biosimilar and originator bDMARDs introduced since 2018 were incorporated in the analysis (assuming all new users started on a biosimilar form, if available). This reduced the savings associated with Harmonization Scenario 1 by approximately 25% and reduced the costs associated with Harmonization Scenario 2 by approximately 24%. New bDMARDs introduced since 2018 were biosimilars for adalimumab and rituximab, as well as the originator sarilumab; however, the impact of this scenario analysis was largely driven by the introduction of adalimumab biosimilars, as adalimumab represented 40% of the market share.
Several key assumptions were incorporated in the BIA and potential limitations associated with these should be acknowledged. First, the starting year of the cohort in the base case was assumed to be 2018, which reflects the most recent year for which bDMARD utilization data were available from the CADTH Utilization Study.2 The market share of each bDMARD was assumed to remain unchanged over a 3-year analysis period (i.e., no forecasting was undertaken), which was a simplifying assumption. If new patients were preferentially started on biosimilar formulations, this would result in lower overall costs and reduce the differential between the reference and harmonization scenarios.
Second, time to the initiation of bDMARD therapy among new adult users in the BIA was assumed to be equivalent to the observed trends among patients aged 67 years and older in the Utilization Study.2 The cohort of patients aged 67 years and older was assumed to reflect patients eligible for public drug plans; in contrast, patients aged younger than 65 years may have public or private insurance and some patients may transition between these. The time to bDMARD initiation among new users aged 18 to 65 years who are eligible for public drug plan coverage is unknown. Additionally, individual results for the Atlantic provinces (i.e., Nova Scotia, New Brunswick, Prince Edward Island, Newfoundland and Labrador) are unavailable because of low patient numbers in the concurrent Utilization Study.
Third, because the starting year of the cohort was assumed to be 2018, bDMARDs approved since 2018 are not reflected in the base case. This includes biosimilars for adalimumab and rituximab, and the originator sarilumab. In scenario analyses, incorporating these new bDMARDs and assuming that new users would preferentially start on a biosimilar reduced the savings associated with Harmonization Scenario 1 and reduced the costs associated with Harmonization Scenario 2. Newer bDMARDs not currently reimbursed on public formularies (e.g., Remsima, a subcutaneous form of infliximab) are not included in this analysis.
Fourth, the cost of csDMARDs was not incorporated in the BIA. This was a simplifying assumption that is not expected to have an important effect on the budgetary impact of harmonizing time to bDMARD initiation. As indicated by the clinical expert consulted by CADTH for this BIA, bDMARDs are expected to be added to patients’ existing csDMARD regimen and their usage is not likely to be affected by altering time to the addition of a bDMARD. Further, the cost of csDMARDs is low compared to bDMARDs and changes to csDMARD regimens would not be expected to have an important budgetary impact.
Fifth, the health implications (including on health care system costs) of altering the duration of time that a patient spends on csDMARDs before initiating a first bDMARD was not considered in the budget impact analysis.
Finally, it should be noted that the data from the CADTH Utilization Study (i.e., new users, time to a first bDMARD) may include patients receiving bDMARDs for the treatment of other rheumatic disease (e.g., ankylosing spondylitis, psoriatic arthritis) owing to the use of claims-based methodology.2 CADTH further notes that, while the focus of this BIA was on estimating the budgetary impact of aligning time to the initiation of bDMARD therapy across jurisdictions, time to bDMARD initiation may be influenced by many factors (e.g., reimbursement criteria, prescribing behaviour, rural versus urban location, wait times); analyzing the impact of such particular factors was beyond the scope of this analysis.
References
1.Canadian Institute for Health Information (CIHI). Prescribed drug spending in Canada, 2019. 2019: https://www.cihi.ca/en/prescribed-drug-spending-in-canada-2019. Accessed 2021 Jun 2.
2.Dyrda P, Loncar M, Tadrous M. Utilization of csDMARDs and bDMARDs for the treatment of rheumatoid arthritis across public drug plans. 2021: https://www.cadth.ca/harmonization-public-coverage-policies-biologic-drugs-treatment-rheumatoid-arthritis. Accessed 2021 May 19.
3.Deane KD, Demoruelle MK, Kelmenson LB, Kuhn KA, Norris JM, Holers VM. Genetic and environmental risk factors for rheumatoid arthritis. Best Pract Res Clin Rheumatol. 2017;31(1):3-18. PubMed
4.Fazal SA, Khan M, Nishi SE, et al. A Clinical Update and Global Economic Burden of Rheumatoid Arthritis. Endocr Metab Immune Disord Drug Targets. 2018;18(2):98-109. PubMed
5.Aletaha D, Smolen JS. Diagnosis and Management of Rheumatoid Arthritis: A Review. JAMA. 2018;320(13):1360-1372. PubMed
6.Government of Canada. Canadian Chronic Disease Surveillance System (CCDSS). 2020: https://health-infobase.canada.ca/ccdss/data-tool/Index. Accessed 2021 May 17.
7.Bykerk VP, Akhavan P, Hazlewood GS, et al. Canadian Rheumatology Association recommendations for pharmacological management of rheumatoid arthritis with traditional and biologic disease-modifying antirheumatic drugs. J Rheumatol. 2012;39(8):1559-1582. PubMed
8.Comparative Assessment of Coverage Criteria for Biologic Disease-Modifying Antirheumatic Drugs Across Canadian Public Drug Plans: An Environmental Scan. Ottawa: CADTH; 2020: https://www.cadth.ca/comparative-assessment-coverage-criteria-biologic-disease-modifying-antirheumatic-drugs-across-0. Accessed 2021 May 11.
9.Sullivan SD, Mauskopf JA, Augustovski F, et al. Budget impact analysis-principles of good practice: report of the ISPOR 2012 Budget Impact Analysis Good Practice II Task Force. Value Health. 2014;17(1):5-14. PubMed
10.Ontario Ministry of Health, Ontario Ministry of Long-Term Care. Ontario drug benefit formulary/comparative drug index. 2021; https://www.formulary.health.gov.on.ca/formulary/. Accessed 2021 May 18.
11.Exceptional Access Program (EAP). Toronto (ON): Ontario Ministry of Health; Ontario Ministry of Long-Term Care; 2021: https://www.health.gov.on.ca/en/pro/programs/drugs/odbf/odbf_except_access.aspx. Accessed 2021 May 18.
12.Saskatchewan Drug Plan: search formulary. 2021; https://formulary.drugplan.ehealthsask.ca/SearchFormulary. Accessed 2021 May 18.
13.Government of British Columbia. BC PharmaCare formulary search. 2021; https://pharmacareformularysearch.gov.bc.ca. Accessed 2021 May 18.
Appendix 1: Budget Impact Analysis Tables
Table 9: Cost Table for bDMARDs for the Treatment of Rheumatoid Arthritis
bDMARD | Recommended dosage | Strength/ concentration | Price ($)a,b,c | |||
Ontariod | Saskatchewan | Alberta | British Columbia | |||
Abatacept (originator) | SC: 125 mg weekly; IV: 750 mge at week 0, 2, 4, then every 4 weeks | SC: 125 mg/mL IV: 250 mg | 373.7881 500.3411 | 373.7900 500.3400 | 373.7875 500.3400 | 392.4775 525.3582 |
Adalimumab (originator) | 40 mg SC every 2 weeks | 40 mg/0.2 mL 40 mg/0.8 mL | 392.7250 785.4500 | 392.7250 785.4500 | 392.7250 785.4500 | NA 824.7225 |
Adalimumab (biosimilar)f | 40 mg SC every 2 weeks | 20 mg/0.4 mL 40 mg/0.8 mL | 235.6400 471.2700 | NAg | 235.6400 471.2700 | 247.422 494.8335 |
Certolizumab (originator) | 400 mg SC at week 0, 2, 4, followed by 200 mg every 2 weeks | 200 mg/mL | 664.5100 | 664.5100 | 664.5100 | 697.7355 |
Etanercept (originator) | 50 mg SC weekly | 25 mg/vial 50 mg/mL | 202.9300 405.9850 | 202.9300 405.9900 | 200.7100 401.5400 | 764.9800 382.4940 |
Etanercept (biosimilar) | 50 mg SC weekly | 50 mg/mL | 241.0000 | 241.0000 | 241.0000 | 260.2800 |
Golimumab (originator) | SC: 50 mg every 4 weeks; IV 2 mg/kg at 0, 4 weeks, then every 8 weeks | SC: 100 mg/mL IV: 50 mg/4 mL | 1,555.1700 NA | 1,555.5000 NA | 1,516.0000 879.500 | 3,334.4430 NA |
Infliximab (originator) | 3 mg/kg IV at 0, 2, 6 weeks, then every 8 weeks | 100 mg/vial | 987.5600 | 977.0000 | 987.5600 | 987.5600 |
Infliximab (biosimilar) | 3 mg/kg IV at 0, 2, 6 weeks, then every 8 weeks | 100 mg/vial (Inflectra) | 525.0000 | 650.0000 | 525.0000 | 551.2500 |
Rituximab (originator) | One course is 2 IV infusions (1,000 mg; week 0, week 2); 2 courses per year | 100 mg/10 mL 500 mg/50 mL | 482.3080 2,411.5400 | 482.3050 2,411.5250 | NA NA | 506.4230 NA |
Rituximab (biosimilar)f | One course is 2 IV infusions (1,000 mg; week 0, week 2); 2 courses per year | 100 mg/10 mL | 297.0000 1,485.0000 | 297.0000 1,485.0000 | 297.0000 1,485.0000 | 320.7600 1,603.8000 |
Sarilumab (originator)f | 200 mg SC every 2 weeks | 150 mg/1.14 mL 200 mg/1.14 mL | 737.5800 | 737.5800 | 737.5800 | NAh |
Tocilizumab (originator) | SC: patients < 100 kg: 162 mg every other week; patients ≥ 100 kg: 162 mg weekly. IV: 4 mg/kg every 4 weeks | SC: 162 mg/0.9 mL IV: 80 mg/4 mL and 200 mg/10 mL | 358.9050 182.8000 457.0000 | 358.9050 182.8000 457.0000 | 358.9050 182.8000 457.0000 | 376.8503 191.9100 479.8500 |
IV = intravenous; NA = not applicable; SC = subcutaneous.
aFor biosimilars with multiple available products, the most expensive within each jurisdiction was included in the base case.
bA patient weight of 75 kg was assumed in the base case for biologic disease-modifying antirheumatic drugs with weight-based dosages.
cSource: Ontario Drug Benefit Formulary,10 Ontario Exceptional Access Program,11 Saskatchewan Drug Plan,12 Alberta Blue Cross, and British Columbia PharmaCare.13
dOntario costs were used as proxy costs for the Atlantic provinces and Manitoba in the base case, as bDMARD prices were not publicly available for these jurisdictions.
eDosage for patient with a body weight of 60 kg to 100 kg (for patients < 60 kg, 500 mg; for patients > 100 kg, 1,000 mg).
fIntroduced after the base case cohort starting year (2018); this bDMARD was therefore not included in the CADTH base case.
gNot currently covered by the public drug plan. In scenario analyses, the Ontario price was used as a proxy.
hReimbursed by the public payer per day of use. In scenario analyses where sarilumab is included, the Ontario price per unit was used as a proxy.
Table 10: Pan-Canadian Budget Impact Analysis Results by Jurisdiction — Harmonization Scenario 1
Scenario | Atlantic Provinces | Ontario | Manitoba | Saskatchewan | Alberta | British Columbia | Pan-Canadian impact |
Reference Scenario | $16,496,280 | $62,937,265 | $8,676,717 | $8,509,143 | $21,710,751 | $33,592,529 | $151,922,685 |
Harmonization Scenarioa | $11,895,957 | $62,937,265 | $6,213,669 | $6,016,555 | $21,710,751 | $33,592,529 | $142,366,726 |
Budget Impact | –$4,600,323 | $0 | –$2,463,047 | –$2,492,588 | $0 | $0 | –$9,555,958 |
Note: Negative values denote cost savings.
aTime to the initiation of biologic disease-modifying antirheumatic drug therapy was increased by 4 months for the Atlantic provinces, Manitoba, and Saskatchewan.
Table 11: Pan-Canadian Budget Impact Analysis Results by Jurisdiction – Harmonization 2
Scenario | Atlantic Provinces | Ontario | Manitoba | Saskatchewan | Alberta | British Columbia | Pan-Canadian impact |
Reference Scenario | $16,496,280 | $62,937,265 | $8,676,717 | $8,509,143 | $21,710,751 | $33,592,529 | $151,922,685 |
Harmonization Scenarioa | $16,496,280 | $84,436,745 | $8,676,717 | $8,509,143 | $30,276,695 | $45,252,167 | $193,647,746 |
Budget Impact | $0 | $21,499,480 | $0 | $0 | $8,565,944 | $11,659,637 | $41,725,061 |
aTime to the initiation of biologic disease-modifying antirheumatic drug therapy was assumed to be decreased by 4 months for Alberta, British Columbia, and Ontario.
Table 12: Pan-Canadian Budget Impact Analysis Scenario Results by Jurisdiction — Harmonization Scenario 1
Scenarioa | Atlantic Provinces | Ontario | Manitoba | Saskatchewan | Alberta | British Columbia | Pan-Canadian impact |
Base case | |||||||
Reference Scenario | $16,496,280 | $62,937,265 | $8,676,717 | $8,509,143 | $21,710,751 | $33,592,529 | $151,922,685 |
Harmonization Scenario | $11,895,957 | $62,937,265 | $6,213,669 | $6,016,555 | $21,710,751 | $33,592,529 | $142,366,726 |
Budget Impact | –$4,600,323 | $0 | –$2,463,047 | –$2,492,588 | $0 | $0 | –$9,555,958 |
Scenario 1: Lower patient weight (50 kg) | |||||||
Reference Scenario | $15,437,374 | $58,602,302 | $8,477,541 | $8,436,418 | $19,931,008 | $32,836,083 | $143,720,725 |
Harmonization Scenario | $11,121,171 | $58,602,302 | $6,068,619 | $5,964,434 | $19,931,008 | $32,836,083 | $134,523,616 |
Budget Impact | –$4,316,203 | $0 | –$2,408,922 | –$2,471,984 | $0 | $0 | –$9,197,109 |
Scenario 2: Higher patient weight (100 kg) | |||||||
Reference Scenario | $16,672,582 | $63,975,212 | $8,873,216 | $8,636,154 | $22,113,567 | $34,177,678 | $154,448,408 |
Harmonization Scenario | $12,022,485 | $63,975,212 | $6,354,173 | $6,106,236 | $22,113,567 | $34,177,678 | $144,749,350 |
Budget Impact | –$4,650,097 | $0 | –$2,519,043 | –$2,529,918 | $0 | $0 | –$9,699,057 |
Scenario 3: All users assumed to receive IV forms of abatacept, golimumab, and tocilizumab | |||||||
Reference Scenario | $16,843,804 | $64,037,774 | $8,798,489 | $8,738,322 | $22,292,357 | $31,681,009 | $152,391,754 |
Harmonization Scenario | $12,235,135 | $64,037,774 | $6,342,696 | $6,230,084 | $22,292,357 | $31,681,009 | $142,819,055 |
Budget Impact | –$4,608,668 | $0 | –$2,455,793 | –$2,508,238 | $0 | $0 | –$9,572,700 |
Scenario 4: British Columbia drug prices adopted as proxy for Atlantic and Manitoba drug prices | |||||||
Reference Scenario | $19,871,095 | $62,937,265 | $9,988,999 | $8,509,143 | $21,710,751 | $33,592,529 | $156,609,782 |
Harmonization Scenario | $14,318,685 | $62,937,265 | $7,152,365 | $6,016,555 | $21,710,751 | $33,592,529 | $145,728,151 |
Budget Impact | –$5,552,409 | $0 | –$2,836,634 | –$2,492,588 | $0 | $0 | –$10,881,631 |
Scenario 5: Least expensive bDMARD price incorporated | |||||||
Reference Scenario | $16,490,053 | $62,806,572 | $8,650,252 | $8,488,090 | $21,673,136 | $33,526,789 | $151,634,893 |
Harmonization Scenario | $11,891,401 | $62,806,572 | $6,194,396 | $6,001,467 | $21,673,136 | $33,526,789 | $142,093,761 |
Budget Impact | –$4,598,653 | $0 | –$2,455,856 | –$2,486,623 | $0 | $0 | –$9,541,131 |
Scenario 6: Suppressed cell counts assumed to represent 1 new user | |||||||
Reference Scenario | $16,496,280 | $62,937,265 | $8,494,282 | $8,334,604 | $21,710,751 | $33,592,529 | $151,565,711 |
Harmonization Scenario | $11,895,957 | $62,937,265 | $6,081,931 | $5,891,465 | $21,710,751 | $33,592,529 | $142,109,897 |
Budget Impact | –$4,600,323 | $0 | –$2,412,351 | –$2,443,139 | $0 | $0 | –$9,455,814 |
Scenario 7: Cohort starting year assumed to be 2021b | |||||||
Reference Scenario | $16,900,335 | $65,630,492 | $8,908,608 | $8,678,701 | $22,635,422 | $34,888,467 | $157,642,026 |
Harmonization Scenario | $12,187,333 | $65,630,492 | $6,379,734 | $6,136,445 | $22,635,422 | $34,888,467 | $147,857,893 |
Budget Impact | –$4,713,002 | $0 | –$2,528,874 | –$2,542,257 | $0 | $0 | –$9,784,133 |
Scenario 8: Cohort starting year assumed to be 2021b and all new users assumed to start on a biosimilar (if available) | |||||||
Reference Scenario | $14,831,277 | $58,421,044 | $8,656,589 | $8,530,157 | $19,767,883 | $33,860,125 | $144,067,076 |
Harmonization Scenario | $10,681,801 | $58,421,044 | $6,198,462 | $6,031,067 | $19,767,883 | $33,860,125 | $134,960,383 |
Budget Impact | –$4,149,476 | $0 | –$2,458,126 | –$2,499,090 | $0 | $0 | –$9,106,693 |
Scenario 9: Cohort starting year assumed to be 2021,b all new users assumed to start on a biosimilar, and bDMARDs introduced since 2018 incorporated | |||||||
Reference Scenario | $12,012,552 | $46,443,732 | $6,525,873 | $6,746,629 | $15,897,870 | $27,040,833 | $114,667,490 |
Harmonization Scenario | $8,658,859 | $46,443,732 | $4,674,926 | $4,771,741 | $15,897,870 | $27,040,833 | $107,487,961 |
Budget Impact | –$3,353,693 | $0 | –$1,850,947 | –$1,974,888 | $0 | $0 | –$7,179,528 |
Scenario 10: Harmonization scenario 3 (time to a first bDMARD is adjusted by 2 months across jurisdictions)c | |||||||
Reference Scenario | $16,496,280 | $62,937,265 | $8,676,717 | $8,509,143 | $21,710,751 | $33,592,529 | $151,922,685 |
Harmonization Scenario | $14,229,567 | $73,736,508 | $7,451,596 | $7,268,769 | $26,070,098 | $39,436,602 | $168,193,140 |
Budget Impact | –$2,266,713 | $10,799,243 | –$1,225,121 | –$1,240,374 | $4,359,347 | $5,844,072 | $16,270,455 |
Note: Negative values denote cost savings.
bDMARD = biologic disease-modifying antirheumatic drug.
aHarmonization Scenario 1: Time to bDMARD initiation was increased by 4 months for the Atlantic provinces, Manitoba, and Saskatchewan.
bThe number of new users was increased by jurisdiction-specific population growth rates; all other parameters were held constant unless otherwise stated.
cTime to bDMARD initiation was harmonized across jurisdictions by assuming that jurisdiction-specific current time was concurrently increased by 2 months in the Atlantic provinces, Manitoba, and Saskatchewan and decreased by 2 months in Alberta, British Columbia, and Ontario.
Table 13: Pan-Canadian Budget Impact Analysis Scenario Results by Jurisdiction — Harmonization Scenario 2
Scenarioa | Atlantic Provinces | Ontario | Manitoba | Saskatchewan | Alberta | British Columbia | Pan-Canadian impact |
Base case | |||||||
Reference Scenario | $16,496,280 | $62,937,265 | $8,676,717 | $8,509,143 | $21,710,751 | $33,592,529 | $151,922,685 |
Harmonization Scenario | $16,496,280 | $84,436,745 | $8,676,717 | $8,509,143 | $30,276,695 | $45,252,167 | $193,647,746 |
Budget Impact | $0 | $21,499,480 | $0 | $0 | $8,565,944 | $11,659,637 | $41,725,061 |
Scenario 1: Lower patient weight (50 kg) | |||||||
Reference Scenario | $15,437,374 | $58,602,302 | $8,477,541 | $8,436,418 | $19,931,008 | $32,836,083 | $143,720,725 |
Harmonization Scenario | $15,437,374 | $78,743,577 | $8,477,541 | $8,436,418 | $27,826,053 | $44,257,757 | $183,178,719 |
Budget Impact | $0 | $20,141,275 | $0 | $0 | $7,895,045 | $11,421,674 | $39,457,994 |
Scenario 2: Higher patient weight (100 kg) | |||||||
Reference Scenario | $16,672,582 | $63,975,212 | $8,873,216 | $8,636,154 | $22,113,567 | $34,177,678 | $154,448,408 |
Harmonization Scenario | $16,672,582 | $85,838,662 | $8,873,216 | $8,636,154 | $30,841,275 | $46,043,098 | $196,904,986 |
Budget Impact | $0 | $21,863,450 | $0 | $0 | $8,727,708 | $11,865,420 | $42,456,578 |
Scenario 3: All users assumed to receive IV forms of abatacept, golimumab, and tocilizumab | |||||||
Reference Scenario | $16,843,804 | $64,037,774 | $8,798,489 | $8,738,322 | $22,292,357 | $31,681,009 | $152,391,754 |
Harmonization Scenario | $16,843,804 | $85,249,836 | $8,798,489 | $8,738,322 | $30,883,445 | $42,253,041 | $192,766,937 |
Budget Impact | $0 | $21,212,062 | $0 | $0 | $8,591,088 | $10,572,032 | $40,375,182 |
Scenario 4: British Columbia drug prices adopted as proxy for Atlantic and Manitoba drug prices | |||||||
Reference Scenario | $19,871,095 | $62,937,265 | $9,988,999 | $8,509,143 | $21,710,751 | $33,592,529 | $156,609,782 |
Harmonization Scenario | $19,871,095 | $84,436,745 | $9,988,999 | $8,509,143 | $30,276,695 | $45,252,167 | $198,334,843 |
Budget Impact | $0 | $21,499,480 | $0 | $0 | $8,565,944 | $11,659,637 | $41,725,061 |
Scenario 5: Least expensive bDMARD price incorporated | |||||||
Reference Scenario | $16,490,053 | $62,806,572 | $8,650,252 | $8,488,090 | $21,673,136 | $33,526,789 | $151,634,893 |
Harmonization Scenario | $16,490,053 | $84,265,104 | $8,650,252 | $8,488,090 | $30,224,900 | $45,165,746 | $193,284,146 |
Budget Impact | $0 | $21,458,532 | $0 | $0 | $8,551,764 | $11,638,957 | $41,649,253 |
Scenario 6: Suppressed cell counts assumed to represent 1 new user | |||||||
Reference Scenario | $16,496,280 | $62,937,265 | $8,494,282 | $8,334,604 | $21,710,751 | $33,592,529 | $151,565,711 |
Harmonization Scenario | $16,496,280 | $84,436,745 | $8,494,282 | $8,334,604 | $30,276,695 | $45,252,167 | $193,290,772 |
Budget Impact | $0 | $21,499,480 | $0 | $0 | $8,565,944 | $11,659,637 | $41,725,061 |
Scenario 7: Cohort starting year assumed to be 2021b | |||||||
Reference Scenario | $16,900,335 | $65,630,492 | $8,908,608 | $8,678,701 | $22,635,422 | $34,888,467 | $157,642,026 |
Harmonization Scenario | $16,900,335 | $88,049,984 | $8,908,608 | $8,678,701 | $31,566,194 | $46,997,912 | $201,101,733 |
Budget Impact | $0 | $22,419,491 | $0 | $0 | $8,930,771 | $12,109,445 | $43,459,707 |
Scenario 8: Cohort starting year assumed to be 2021b and all new users assumed to start on a biosimilar (if available) | |||||||
Reference Scenario | $14,831,277 | $58,421,044 | $8,656,589 | $8,530,157 | $19,767,883 | $33,860,125 | $144,067,076 |
Harmonization Scenario | $14,831,277 | $78,510,406 | $8,656,589 | $8,530,157 | $27,603,927 | $45,628,216 | $183,760,572 |
Budget Impact | $0 | $20,089,362 | $0 | $0 | $7,836,044 | $11,768,091 | $39,693,497 |
Scenario 9: Cohort starting year assumed to be 2021,b all new users assumed to start on a biosimilar, and bDMARDs introduced since 2018 incorporated | |||||||
Reference Scenario | $12,012,552 | $46,443,732 | $6,525,873 | $6,746,629 | $15,897,870 | $27,040,833 | $114,667,490 |
Harmonization Scenario | $12,012,552 | $62,333,096 | $6,525,873 | $6,746,629 | $22,179,779 | $36,410,740 | $146,208,669 |
Budget Impact | $0 | $15,889,364 | $0 | $0 | $6,281,908 | $9,369,907 | $31,541,180 |
bDMARD = biologic disease-modifying antirheumatic drug.
aHarmonization Scenario 2: Time to bDMARD initiation was assumed to be decreased by 4 months for Alberta, British Columbia, and Ontario.
bThe number of new users was increased by jurisdiction-specific population growth rates; all other parameters were held constant unless otherwise stated.
ISSN: 2563-6596
Disclaimer: The information in this document is intended to help Canadian health care decision-makers, health care professionals, health systems leaders, and policy-makers make well-informed decisions and thereby improve the quality of health care services. While patients and others may access this document, the document is made available for informational purposes only and no representations or warranties are made with respect to its fitness for any particular purpose. The information in this document should not be used as a substitute for professional medical advice or as a substitute for the application of clinical judgment in respect of the care of a particular patient or other professional judgment in any decision-making process. The Canadian Agency for Drugs and Technologies in Health (CADTH) does not endorse any information, drugs, therapies, treatments, products, processes, or services.
While care has been taken to ensure that the information prepared by CADTH in this document is accurate, complete, and up to date as at the applicable date the material was first published by CADTH, CADTH does not make any guarantees to that effect. CADTH does not guarantee and is not responsible for the quality, currency, propriety, accuracy, or reasonableness of any statements, information, or conclusions contained in any third-party materials used in preparing this document. The views and opinions of third parties published in this document do not necessarily state or reflect those of CADTH.
CADTH is not responsible for any errors, omissions, injury, loss, or damage arising from or relating to the use (or misuse) of any information, statements, or conclusions contained in or implied by the contents of this document or any of the source materials.
This document may contain links to third-party websites. CADTH does not have control over the content of such sites. Use of third-party sites is governed by the third-party website owners’ own terms and conditions set out for such sites. CADTH does not make any guarantee with respect to any information contained on such third-party sites and CADTH is not responsible for any injury, loss, or damage suffered as a result of using such third-party sites. CADTH has no responsibility for the collection, use, and disclosure of personal information by third-party sites.
Subject to the aforementioned limitations, the views expressed herein do not necessarily reflect the views of Health Canada, Canada’s provincial or territorial governments, other CADTH funders, or any third-party supplier of information.
This document is prepared and intended for use in the context of the Canadian health care system. The use of this document outside of Canada is done so at the user’s own risk.
This disclaimer and any questions or matters of any nature arising from or relating to the content or use (or misuse) of this document will be governed by and interpreted in accordance with the laws of the Province of Ontario and the laws of Canada applicable therein, and all proceedings shall be subject to the exclusive jurisdiction of the courts of the Province of Ontario, Canada.
The copyright and other intellectual property rights in this document are owned by CADTH and its licensors. These rights are protected by the Canadian Copyright Act and other national and international laws and agreements. Users are permitted to make copies of this document for non-commercial purposes only, provided it is not modified when reproduced and appropriate credit is given to CADTH and its licensors.
About CADTH: CADTH is an independent, not-for-profit organization responsible for providing Canada’s health care decision-makers with objective evidence to help make informed decisions about the optimal use of drugs, medical devices, diagnostics, and procedures in our health care system.
Funding: CADTH receives funding from Canada’s federal, provincial, and territorial governments, with the exception of Quebec.
Questions or requests for information about this report can be directed to requests@cadth.ca